Received:6November2019Revised:3December2019Accepted:5December2019
DOI:10.1002/jbio.201960177
Polarizedlighttherapy:Shiningalightonthemechanism underlyingitsimmunomodulatoryeffects
JackFeehan1,2* |NicholasTripodi3,4 |SarahFraser3 | KathleenMikkelsen3 |AprilThewlis1 |DimitriosKiatos1,3 |MajaHusaric3,4 | VassoApostolopoulos3*
1OsteopathyGroup,CollegeofHealthand Biomedicine,VictoriaUniversity,Victoria, Australia
2AustralianInstituteforMusculoskeletal Science(AIMSS),UniversityofMelbourne andWesternHealth,StAlbans,Victoria, Australia
3InstituteforHealthandSport,Victoria University,Victoria,Australia
4FirstYearCollege,VictoriaUniversity, Victoria,Australia
*Correspondence
JackFeehan,OsteopathyGroup,College ofHealthandBiomedicine,Victoria University,Victoria,Australia.
Email:jfeehan@student.unimelb.edu.au
VassoApostolopoulos,InstituteforHealth andSport,VictoriaUniversity,Victoria, Australia.
Email:vasso.apostolopoulos@vu.edu.au
Abstract

Thisstudyinvestigatestheimmunomodulatoryeffectsofpolychromatic polarizedlighttherapy(PLT)on humanmonocytecells.Whilethereis someevidencedemonstratingaclinicaleffectinthetreatmentofcertain conditions,thereislittleresearchinto itsmechanismofaction.Herein,U937 monocytecellswereculturedand exposedtoPLT.Thecellswerethen analyzedforchangeinexpressionof genesandcellsurfacemarkersrelatingto inflammation.Itwasnotedthat6hours ofPLTreducedtheexpressionoftheCD14,MHCIandCD11breceptors,and increasedtheexpressionofCD86.ItwasalsoshownthatPLTcauseddownregulationofthegenesIL1B,CCL2, NLRP3andNOD1,andupregulationof NFKBIAandTLR9.ThesefindingsimplythatPLThasthecapacityforimmunomodulationinhumanimmunecells,possiblyexertingananti-inflammatoryeffect.
KEYWORDS
inflammation,phototherapy,polarizedlight,polarizedlighttherapy
1 | INTRODUCTION
Inflammationisaprocessheavilyimplicatedinpathological statesofallkinds,fromallergyandneoplasia,toinfection. Despiteitscentralroleinthepathophysiologyofmany commonconditions,currentmethodsofmanaginginflammationarefraughtwithproblems,particularlyinachronic setting.Treatmenttypicallyinvolvespharmacologicalinterventions,manyofwhichexertarangeofunwantedeffects
inadditiontotheirtherapeuticaction[1].Thecontinuing evolutionanddevelopmentofnoveltherapeuticapproaches tothemanagementofinflammationisessentialinorderto advancepatientcare.Evaluationofnonpharmacological anti-inflammatorytreatmentsisakey,butrelativelyunderrepresentedpartofthisprocess.Asmallbutgrowingbody ofevidenceindicatesthatphototherapiessuchaspolarized lighttherapy(PLT)areapromisingavenueofexploration inthisarea[2,3].
ThisisanopenaccessarticleunderthetermsoftheCreativeCommonsAttribution-NonCommercialLicense,whichpermitsuse,distributionandreproductioninany medium,providedtheoriginalworkisproperlycitedandisnotusedforcommercialpurposes.
©2019TheAuthors. JournalofBiophotonics publishedbyWILEY-VCHVerlagGmbH&Co.KGaA,Weinheim
J.Biophotonics. 2019;e201960177.
www.biophotonics-journal.org 1of9 https://doi.org/10.1002/jbio.201960177
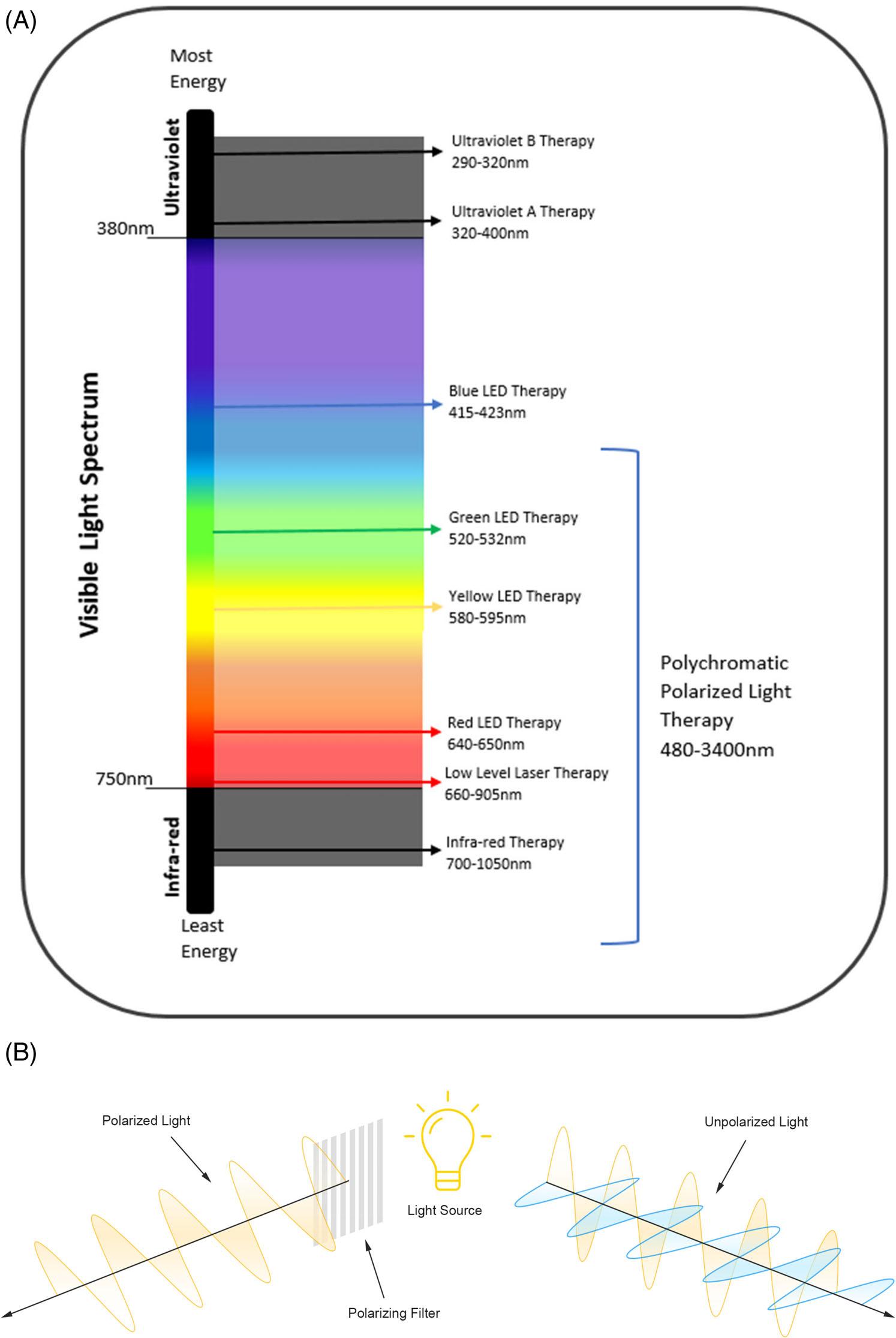
Thetherapeuticbenefitsoflighttherapyhavebeen reportedsincethelate1960swithapplicationsrangingfrom neonataljaundicetopsoriasisandvitiligo[3,4].Thetheoreticalbasisofphototherapyinvolvestheuseoflightto inducephysiologicalchangewithinatargettissue[5],with subsequenttherapeuticeffects.Thereareseveraltypesof phototherapiesdifferentiatedbythespecificphysicalqualitiesofthelightused,withthemostcommonbeinglowlevellasertherapy(LLLT)andultraviolet(UV)therapies. Inrecentyears,however,ithasbeenproposedthatabroad lightspectrumcoveringalllightcolors,andpolarization arecrucialelementsinlighttherapy[6].Polarizedlight (PL)isformedbythefilteringoflightwavessothatthey arealignedandvibratedinasingleplane(Figure1).Once polarized,lighthastheabilitytopenetratefurtherintotissuesthanitsunpolarizedcounterpart[7].
Broad,visiblespectrumPLTdiffersfromotherformsof phototherapyasitusesamuchwiderrangeofwavelengths thanothermodalitiessuchas,LLLTorUV.Consequently, thedevicesusedinPLTaregenerallylessexpensiveand relativelyeasytouse.PLThasbeensuggestedbyadvocates anddevicemanufacturersforuseinseveralcontextssuch aspain,woundhealing,skinconditionsandinflammatory arthritis.Preliminaryclinicalevidenceindicatesefficacyin themanagementofskinulcers[8–10],burns[11–13],musculoskeletalinjuries[14–18]aswellassurgicalandnonhealingwounds[6,19–22].Despitethis,therearelittle scientificdataregardingthephysiologicalmechanisms underlyingthesechangesinvitroorinvivo[2].Ithas suggestedthattheinteractionsoflightwiththekeymitochondrialenzymecytochromeoxidaseCareresponsible forthebeneficialchangesobservedinLLLT[23–25];however,littleinformationhasbeendocumentedexploringthe mechanismsofPLT.
Withagingpopulations,inmostdevelopedeconomies, andtheassociatedincreaseindemandsonhealthbudgets,noninvasive,nonpharmacologicalavenuesofdisease treatmentwillberequiredtocountertheinevitableburdenofinflammatorydisease.PLThasthepotentialtobe aninexpensive,technicallysimpleandsafephototherapeuticintervention.Thisstudyaimedtodetermine whetherPLinducescellularchangestothehumanmonocytecellline,toprovideinsightintothepossiblemechanismsofactionunderpinningitsclinicaluse.Itwas hypothesizedthatadecreaseintheinflammatoryprofile ofimmunecellsmaybepartofthebiologicalactionof PLT.Specifically,thisinvolvedtheexposureofacultured U937monocyte/macrophagecelllinetoPL,andsubsequentassessmentofcellsurfacemarkersandgeneexpressionrelatingtoinflammation.Weshowedthatexposure toPLdecreasestheexpressionofproinflammatorycell surfacemarkersandcauseschangesingeneexpression consistentwithadecreasedinflammatoryprofile.
2 | MATERIALSANDMETHODS
AllproceduresinthisstudywereperformedinPC2laboratoriesattheWerribeecampusofVictoriaUniversity, andtheWesternCentreforHealthResearchandEducation(WesternCHRE)atSunshinehospitalunderstandardlaboratoryconditions.Allcellcultureworkwas doneunderasepticconditionsinaclassIIbiosafetycabinet.Trypanbluestainingwasusedbeforeandafterilluminationtoensurecellviabilitythroughoutthe experiments.Noethicswasrequiredforthisresearch.
2.1 | Cellcultureandcelldifferentiation
TheU937monocytecelllinewasculturedinRoswell ParkMemorialInstitutemedia,supplementedwith10% fetalbovineserum(InterpathServicesPty.Ltd.),1%penicillin/streptomycin(Sigma-Aldrich)and0.1%glutamine (Sigma-Aldrich),andincubatedat37 C,5%CO2 asper standardcultureprotocol.Thecellswerepassagedto50% to90%confluencyandmediareplenishedevery48hours untilanadequatenumberofcellswereobtained.Astock ofexperimentalcells(cellbank)wasthencreatedand storedinliquidnitrogen.Onevialfromthecellbankwas thawedeachtimetheexperimentwasrepeated,toensure allexperimentalcellswereatthesamepassagethroughouttheexperiments.U937cellsarepromonocyticcells andaredifferentiatedintomonocyte/macrophagecells viatheadditionofvitaminD3 tothemediatoafinalconcentrationof100nм for72hours[26].
2.2 | Polarizedlightexposure
ThetherapeuticPLsourceemployedwasusingaBioptron MedAllPAG-960lamp(Bioptron,Switzerland)witha wavelengthrangefrom400to3400nm.Thelightsource hasanaveragepowerdensityof40mW/cm2 evenlydistributedacrossthewavelengthspectrum.Theentireapparatus,includinglampandtripod,washousedcompletely withinanincubator(Figure2).Theincubatorandmedia temperaturesweremonitoredtoensuretherewasnooverheatingofthecultureduetothelightexposure.Thisisto ensurethatobservedeffectsstemmedfromcellularinteractionswithlight,ratherthanathermiceffect.Thelight fromthelampwasprojectedperpendicularlyontoa5cm cellcultureplateatadistanceof10cm,accordingtomanufacturer'sinstructions,withfulllightcoverageofthe sample.Thelidoftheculturedishcontainingtheexperimentalcellswasremoved,toavoidinterferenceofthe lightpassingthrough.Controlcellswerekeptinthesame incubator,separatedbyasolid,stainlesssteelpartitionto
FIGURE1 A,Summary figureofdifferentphysical propertiesofphototherapeutic light.B,Summaryoftheprocessof lightpolarization
ensurenospill-overlightinteractedwiththecontrolsample.Thelidofthecontrolcellplatewasalsoremovedto ensureinteractionswiththeatmospherewithintheincubatordidnotaccountforthechangesseen.Ininitial experiments,cellswereexposedfor5minutes,15minutes or6hours,followedbyincubationfor24hoursbefore beingpreparedforflowcytometricanalysisofcellsurface markerexpression.Thecellsusedforanalysisofgene expressionwereexposedtoPLfor6hours,immediately snapfrozen,andtheirRNAisolatedpriortotheir
preparationforPCRgenearrayanalysis.Allanalysiswas performedintriplicateastechnicalreplicates.
2.3 | Flowcytometry
Thecellswerelabeledforflowcytometryaccordingto manufacturer'sinstructions,andstandardprotocols.All antibodiesweretitratedpriortotheexperimenttoensure optimalworkingconcentrations.Briefly,cellswereplated
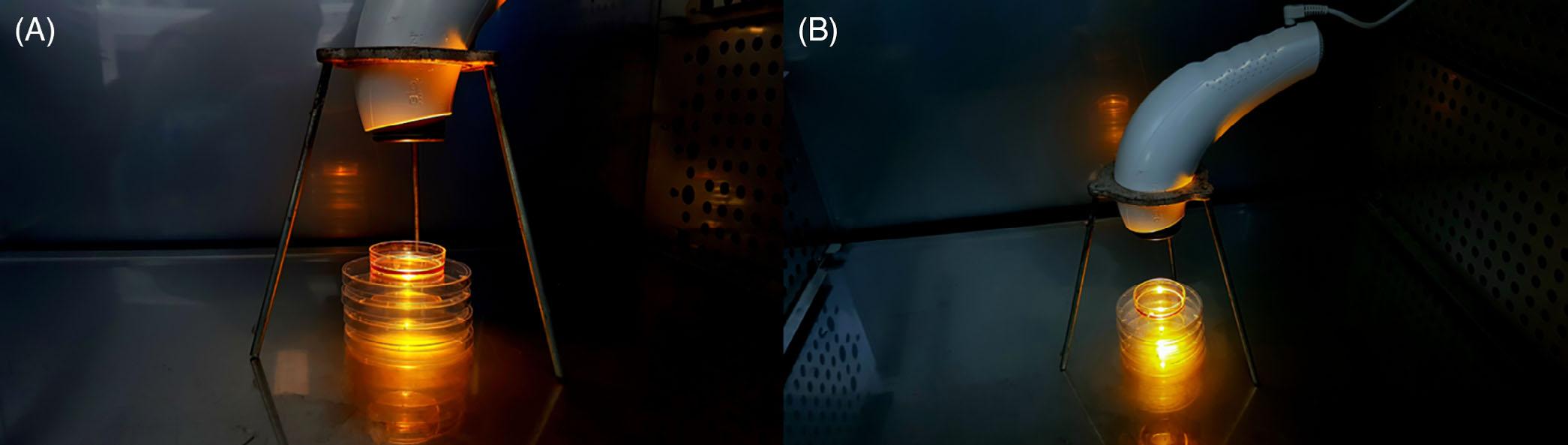
ina96-wellU-bottomplateandtreatedwithFcRblockingreagentfor10minutesatroomtemperature.The cellswerethenincubatedwiththeirspecificconjugated antibody(CD86-Alexa-Fluor488,MajorHistocompatibilityComplexClass[MHC]I,II-BV510,CD206-PE-Cy7, CB11b-PEorCD14-BV421)for45minutesinthedarkat 4 Catapredeterminedconcentration.Otherantibodies againstCD40,CD83andCD209werenegativeonthis celllineandweresubsequentlynotused.Cellswere transportedafterlabelinginthedarkonicepriortoanalysis.AnalysiswasperformedwithaBDFACSCantoII flowcytometer,withthreelasers(violet-405nm,blue –488nmandred – 633nm).Isotypecontrolswereusedto controlforbackgroundfluorescence.AllconjugatedantibodiesweresourcedfromBDBiosciences(SanJose,California)toensurecompatibilitywiththeflowcytometer used.AnalysisofdatawasdonewithBDFACSDivasoftware(Version3.0).Themonocytepopulationwasgated onforwardandsidescatterplotsandanalyzedforfluorescenceintensityagainstunilluminatedcontrolcells. Resultsarereportedasflowcytometricdotplotswithgatingstrategies,overlayhistogramsofcontrolandtreated cells,andmedianfluorescenceintensity,asrecommendedbytheInternationalSocietyfortheAdvancementofCytometrydatastandardstaskforce[27].
2.4 | Genearrays
AllreagentsandconsumablesusedinRNAextraction, cDNAsynthesisandgeneexpressionanalysiswereacquired fromQIAGEN(Hilden,Germany).Thearrayusedwasthe RT2 ProfilerPCRarray “HumanInnateandAdaptive ImmuneResponses” whichanalyses84genesrelatedto immuneandinflammatoryresponses,includingkeycytokines,chemokinesandimmuneactivesurfacemembrane receptors.RNAqualityandconcentrationwereassessedvia Agilent2100Bioanalyzer,Qubitfluorimeter(Invitrogen)and spectrophotometer(DeNovix).RNAextractionwas
performedusingtheRNeasyMinikitaccordingtothemanufacturer'sinstructions.Briefly,cellswerelysedinRLT buffercontaining β-mercaptoethanol(10 μL/mL)and centrifugedthroughaQIAshreddercolumn.Lysateswere passedthroughRNeasyspincolumns,treatedon-column withDNase,followingwhichtheRNAwascollectedin RNasefreewaterforpreparationofcDNA.cDNAwaspreparedinathermalcyclerusingtheQiagenRT2 FirstStrand kit.Briefly,RNA(500ng)wasincubatedat42 Cfor 2minutesinthepresenceofGEbuffer(containingDNase) toeliminateanyremaininggenomicDNA,beforebeing immediatelytransferredtoice.cDNAwasreversetranscribedfor15minutesat42 C,andat92 Cfor3minutesto terminatethereactions.cDNAwasthenaddedtotheRT2 SYBRgreenqPCRmastermixanddispensedtothe96-well genearrayplateaspermanufacturer'sinstructions.Thegene arraywasruninBioradCFX96real-timePCRcyclerwithan initialdenaturationstepof10minutesat95 C,followedby 40cyclesof15secondsat95 Cand1minuteat60 C.Once theRT-PCRwascomplete,meltcurveanalysiswasperformedtoensurethepresenceofasinglePCRproductfor eachgene.GenearraydatawereanalyzedusingCFXMaestro(BioRad).Anunpaired t testwasusedtocompare groups;a P value ≤.05anda2-foldchangeinexpression weresetasthresholdsforsignificance.Geneswereexcluded fromfurtheranalysisifthecrossingpoint(Cq)valuewas greaterthan34and/orauniquemeltingtemperaturewas notobserved.
3 | RESULTS
3.1 | Polarizedlightdecreasesthe expressionofcellsurfacemarkersrelated toinflammation
Gatingusedforanalysiswasperformedagainstappropriateisotypecontrolstoaccountforbackgroundfluorescence.ExamplegatingstrategyisshowninFigure3.
Nochangetocellsurfacemarkerexpressionwasseen after5or30minutesofPLexposure.Thecellsurface markerexpressionchangeafter6hoursexposuretoPL isshown(Figure4).SixhoursofexposuretoPLcaused ameandecreaseinthemedianfluorescenceintensityof 23%forCD11b,39%forCD14,27%inMHCIand35% inMHCII,thoughMHCIIexpressionwaslowatbaseline(Figure4).Conversely,therewasameanincrease inthemedianfluorescenceof20%inCD86.Therewere noconsistentchangesseeninCD206expression.
3.2 | Polarizedlightdecreasesgenes relatedtoinflammation
Allexperimentalandcontrolsamplespassedtheinbuilt qualitycontrolmeasuresinthegenearray.Normalizationwasperformedagainstthetwomoststablehousekeepinggenes GAPDHandRPLP0.Cutoffpointswere setat2-foldupordownregulation,and P valuescalculatedwithsignificancelevelsetat P <.05.Theresultsare summarizedinFigures5and6.Figure5showsgenes withsignificantchangeinregulation,andmorethan 2-foldregulation,andthetotalchangesforallexpressed
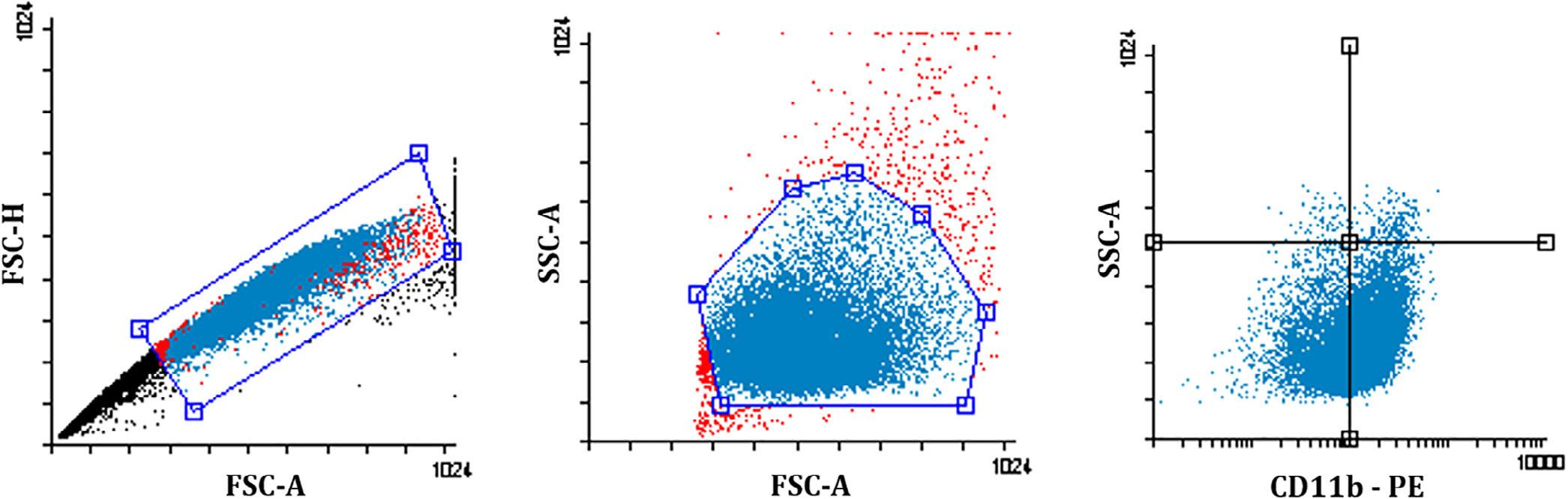
FIGURE3 Examplegatingstrategy.Left-handpanel,doubletdiscriminationstrategy;middlepanel,monocytesgatedusingsizeand density;right-handpanel,fluorescenceintensityofthegivenantibodywithquadrantsforvisualinspection.FSC-A,forwardscatterarea; FFC-H,forwardscatterheight;SSC-A:Sidescatterarea
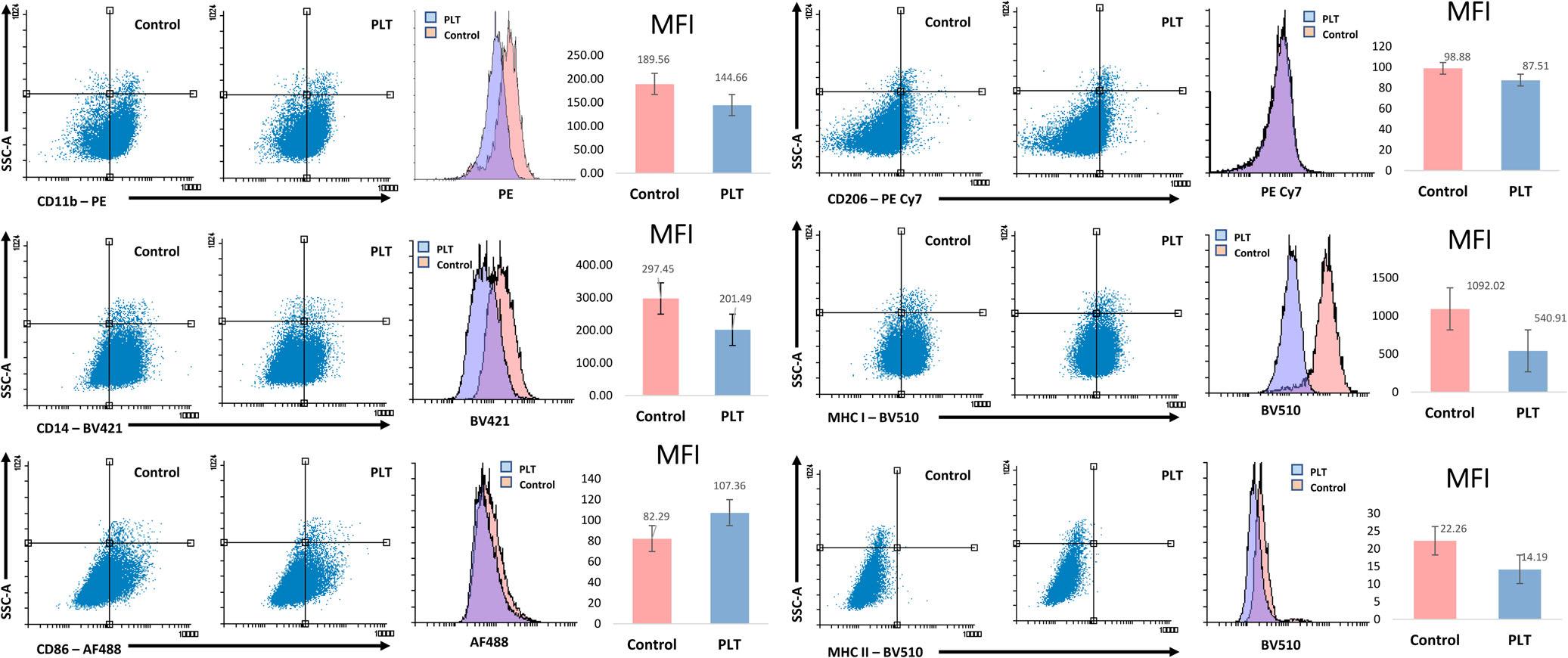
FIGURE4 Changesincellsurfacemarkerexpressionasassessedbyflowcytometryfollowing6hoursexposuretopolarizedlight therapy.LivecellsweregatedonFSCvsSSCprofileandisotypecontrolantibodieswereusedasbackgroundcontrol.Shownarevalues abovethebackgroundisotypecontrols.Experimentswereperformedintriplicate;representativesamplesaredisplayedindotplotsand histograms.MFI,medianfluorescenceintensity
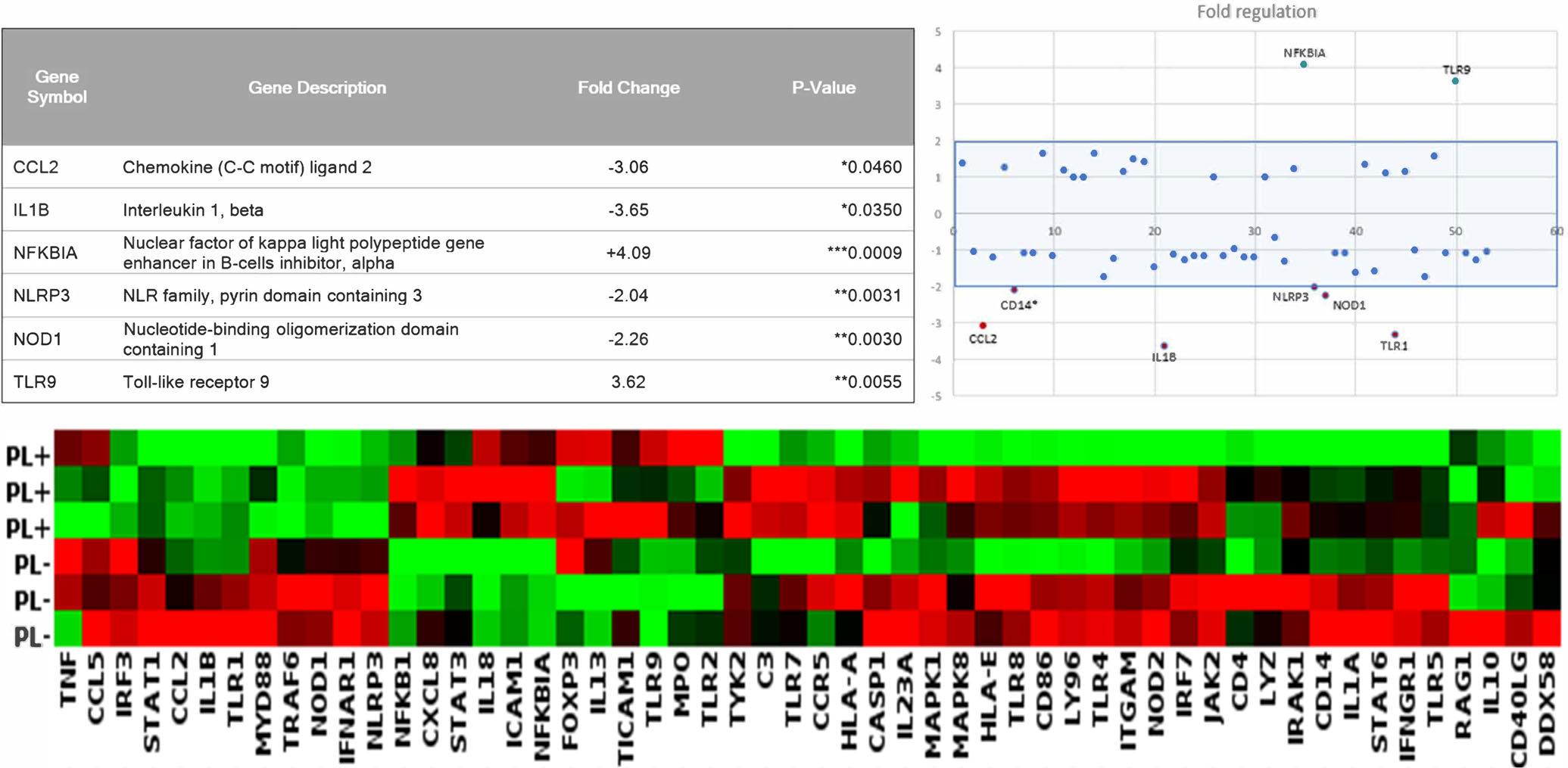
FIGURE5 Geneexpressionchangesfollowing6hoursofPLT.TopLeft,foldregulationandsignificanceofgenes;topright,scatterplot offoldregulation;bottom,clusteredheatmap.Experimentswererepeatedthreetimesandmeanofthreerepeatsareshown
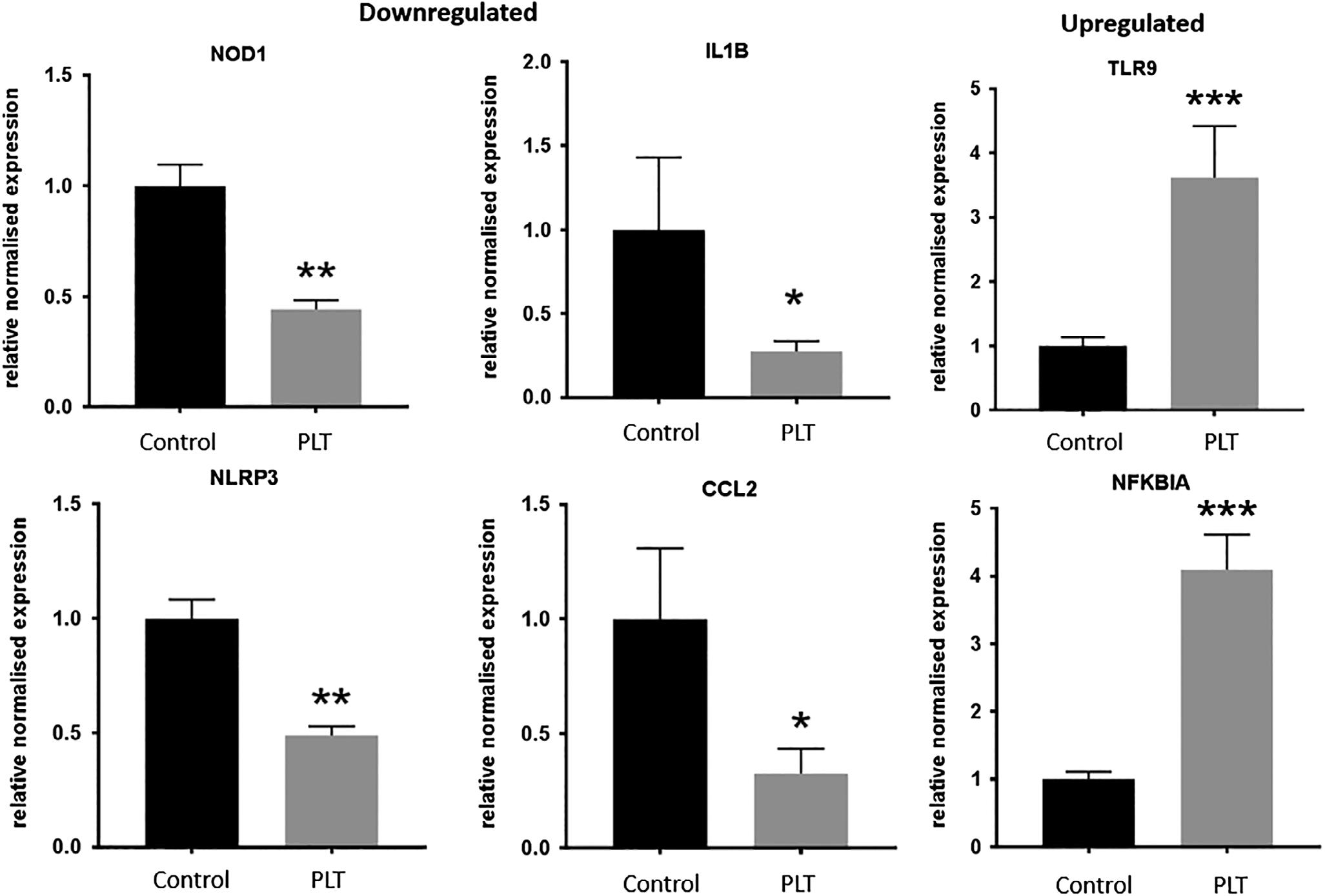
FIGURE6 Bargraphsofrelativenormalizedfoldregulationofsignificantgenescomparedtoreferencepointof1.0. *P <.05; **P <.01; ***P <0.001
genesarepresentedasascatterplotandheatmap. Figure6showsthespecificchangeinexpressionofthe sixgenesthatreachedgreaterthan2-foldup-ordownregulation,andstatisticalsignificance.SixhoursofPLT causedupregulationofNFKBIAandTLR9,anddownregulationofIL1B,CCL2,NLRP3andNOD1.NFKBIA isakeyinflammatoryinhibitorofcytokineproduction, andTLR9codesfortheproductionofthetoll-likereceptor9,akeymembranereceptorinvolvedinpathogen recognitionandimmuneactivation.IL1BandCCL2are importantcytokines,whileNLRP3andNOD1aremembranereceptorsinvolvedininflammatorysignaling.All othergenesassessedinthearraydidnotreachthesignificanceandfoldregulationcutoffs.
4 | DISCUSSION
ThisstudydemonstratesthatPLTcanexertameasurable effectonthehumanimmunesystem,givingmuch neededmechanisticevidenceforitsclinicaleffects.While phototherapiessuchaslow-levellaserhavebeenpreviouslyshowntohaveasuppressiveeffectoninflammation andimmuneresponse[2],verylimitedevidenceexists investigatingwhetherPLThasthesamecapacity.The resultsofthisstudydemonstratechangesingeneexpressionandcellsurfacemarkerexpressionindifferentiated U937cellsfollowing6hoursexposureofPL.ThissuggeststhatPLThasthecapacitytocausemodulationof thebehaviorofactivatedmonocytesinvitro.Thiscould aidinexplainingthedemonstratedeffectsofPLTin improvingwoundhealinganddecreasingpainand inflammationinvivo.
Thecellsurfacemarkersstudiedhereareknownto beinvolvedinarangeofprocessesvitaltotheimmune andinflammatoryresponse,includingrecognitionofself, Tcellactivation,recognitionandphagocytosisofpathogensandimmunecellinfiltrationandaccumulation.Itis difficulttopredicthowtheobservedchangesdemonstratedinthisresearchtranslateintotheinfinitelymore complexcellularenvironmentsurroundinganinflammatoryorhealingprocess,howeverpossibleinvivoeffects maybeinferred.TheMHCIandMHCIIreceptorsare involvedinantigenpresentation[28]andCD11breceptor ininflammatorycellaccumulation[29]andtheCD14 receptorisassociatedwiththedetectionandphagocytosis ofbacteriallipopolysaccharides,andotherproteinaceous debrisbymacrophages[30].Theirsuppressionmaysignifyadecreaseintheinflammatoryactivityofthesecells andhavetheneteffectofdampeningtheintensityand hasteningtheresolutionofinflammationinvivo.Similar changesinimmunecellmarkerexpressionhavebeen showninothercelltypesfollowingexposuretoLLLT,
suggestingthepossibilityofcommonmechanismsforthe actionsofphototherapies[31,32].CD86hasbeenshown toplayanimportantroleintheamplificationandmaintenanceofaninflammatorystateinvivo[33,34].This couldresultinfasterclearanceofdebrisandpathogens andahasteningoftheresolutionoftheinflammatory response,leadingtoenhancedhealingtimes,correlating withtheobservedclinicaleffectsofPLT.
Whilethereductionincellsurfacemarkerssuggestsa functionalimmunomodulatoryeffect,changesingene expressionprovideamechanisticinsightintothecellular responsetoPLT.ThedownregulationofIL1B,CCL2, NLRP3andNOD1suggestsadampeningoftheimmune andinflammatoryresponse.Thesegenesandtheirassociatedproductsareimplicatedinlymphocytedifferentiationandproliferation,inflammasomeactivation[35], systemicinflammation[36],immunecellmigrationand accumulation[37],antigenrecognition[38,39]and phagocyteactivity[39].Othershavedescribeddecreases inIL1BinmurinewoundmodelsandaorticsmoothmusclecellsfollowingexposuretoLLLT[40,41]andLED therapy[42].Additionally,LLLThasbeenshownto increaseordecreasetheexpressionofCCL2atdifferent intensitiesintheTHP-1monocytecellline,suggestinga dose-dependenteffect[43].Thefindingsinthisstudyprovideatheoreticalmechanismfortheirinvivofindings. Decreasingtheexpressionofthesegenesinactivated monocyte/macrophagecellsislikelytocauseafunctional reductionininflammation.Theobservedincreasein NFKBIAfitswithinthepictureofimmunesuppression, asthisgenehasbeenshowntomodulateanddecrease theeffectsofmalignant[44],inflammatory[45]and auto-immunedisease[46].TheoutcomeofthePLmediatedincreaseinTLR9expressionmightplayislessclear. Itisinvolvedintherecognitionandbindingofbacterial andviralDNAandhasbeenassociatedwithautoimmunediseaseandinflammation[47],buthowan upregulationoftheseprocessesmightexertanantiinflammatoryorincreasedhealingstateisunclear.
Whilethisresearchprovidesproofofconceptfora physiologicalmechanismbywhichPLTmayexertitsclinicaleffects,manyquestionsremain.Firstly,whilstmonocyte/macrophagecellsareimportantcontributorsto inflammationandhealingprocesses,thereareahostof othercellsinvolvedinthenaturalsetting.Furtherstudies toevaluatewhetherPLTexertsachangeonotherimportantimmune,connectivetissuesandstemcellsarenecessarytogiveamoreintegratedpictureoftheeffectsofPLT ontissuehealing.Additionally,whilecelllinesareaconvenientandusefulresearchtools,theirbehaviormaynot perfectlyreflectthatoftheirinvivocounterparts.Consequently,evaluationofhowPLTmayaffectthediverse rangeofnativecellularinteractionsinvolvedinhealing
andinflammationshouldbeexaminedthroughanimal modelresearch.Theeffectoflightpenetrancemustalso beevaluatedtoidentifywhethertheeffectsshowninthis studyareequivalenttothosethatmightoccurinaclinical settingwheretheincidentlightisobstructedbyoverlying tissues.WhilePLhasbeenshowntopenetratetoadepth ofupto5centimeters[7],itiscurrentlyunclearifitsabilitytoexertaphysiologicalchangeisaffectedbydistance fromthelightsourceorthelighttransmittingproperties ofthetissuebeingtreated.Finally,thedoseresponseof PLTmustbeevaluatedfurther.Thisstudyidentified 6hoursofPLTasbeingabletogenerateacellular response,butfortranslationintoclinicaluse,minimum effectivetimeframesmustbeidentified,andtheeffectsof repeateddosagesmustalsobeexploredfurther.
5 | CONCLUSION
Noninvasive,nonpharmacologicalinterventionstotreat inflammationandtoassistintheprocessoftissuehealing havethepotentialtorevolutionizethetreatmentofa widerangeofconditions.Thisstudyprovidespreliminary evidencesuggestingthatPLTcouldbeacandidatetofill thisgapintherapeuticapproaches.
Thisstudyprovideskeymechanisticevidenceforthe capacityofPLTtoimpartaphysiologicalchange.The changesingeneandcellsurfacemarkerexpression observedafter6hoursofPLTweresuggestiveofimmune andinflammatorysuppression,providingpotentialfor clinicaluseinahostofillnessesandinjuries.ThesefindingssupportthereportedresultsofPLTseeninclinical practice,whereitisusedtoassistinthehealingof woundsandinjuries.Futureresearchonothercelltypes shouldbeperformedtoestablishaclearmechanism,and doseresponseexperimentsdonetoidentifyoptimaltreatmentpractices.Further,studiescomparingtheeffectof theindividualwavelengthsthatmakeupPLTcould allowformoretargetedtherapeuticapproaches.
CONFLICTOFINTEREST
Theauthorshavenoconflictofinteresttodeclare.
AUTHORCONTRIBUTIONS
J.F.,N.T.andK.M.performedtheexperimentsunderthe supervisionofV.A.,S.F.andM.H.;J.F.,D.K.and V.A.wrotethearticleandJ.F.,V.A.,S.F.,M.H.,D.K.and A.T.editedandreviewedthearticle.
ACKNOWLEDGMENTS
TheauthorswouldliketothanktheImmunologyand TranslationalGroupwithintheInstituteforHealthand Sport,VictoriaUniversity,forsupportanddiscussions.
V.A.wouldliketothanktheVictoriaUniversityCollege ofHealthandBiomedicinestart-upfundsforfinancial support.J.F.wassupportedbytheAustralianGovernmentResearchTrainingProgramandMKbytheVictoria UniversityVice-ChancellorsPostgraduateScholarship.
ORCID
JackFeehan https://orcid.org/0000-0002-9627-1299 VassoApostolopoulos https://orcid.org/0000-0001-67882771
REFERENCES
[1]I.Cascorbi, Clin.Pharmacol.Ther. 2017, 102(4),564.
[2]J.Feehan,S.P.Burrows,L.Cornelius,A.M.Cook, K.Mikkelsen,V.Apostolopoulos,M.Husaric,D.Kiatos, Maturitas 2018, 116,11.
[3]J.M.Bjordal,M.I.Johnson,V.Iversen,F.Aimbire, R.A.B.Lopes-Martins, Photomed.LaserTher. 2006, 24(2),158.
[4]T.Gambichler,S.Terras,A.Kreuter, Clin.Dermatol. 2013, 31 (4),438.
[5]S.Jahanshahifard,M.Ahmadpour-Kacho,Y.Z.Pasha, J.Clin. Neonatol. 2012, 1(3),139.
[6]S.Monstrey,H.Hoeksema,K.Depuydt,V.Maele,K.Van Landuyt,P.Blondeel, Eur.J.Plast.Surg. 2002, 24(8),377.
[7]A.DaSilva,C.Deumié,I.Vanzetta, Biomed.Opt.Express 2012, 3(11),2907.
[8]P.Iordanou,G.Baltopoulos,M.Giannakopoulou,P.Bellou, E.Ktenas, Int.J.Nurs.Pract. 2002, 8(1),49.
[9]A.Durovic,D.Maric,Z.Brdareski,M.Jevtic,S.Durdevic, Vojnosanit.Pregl. 2008, 65(12),906.
[10]L.Medenica,M.Lens, J.WoundCare 2003, 12(1),37.
[11]S.Monstrey,H.Hoeksema,H.Saelens,K.Depuydt, M.Hamdi,K.VanLanduyt,P.Blondeel, Br.J.Plast.Surg. 2002, 55(5),420.
[12]C.A.Karadag,M.Birtane,A.C.Aygit,K.Uzunca, L.Doganay, J.BurnCareRes. 2007, 28(2),291.
[13]P.C.Oliveira,A.L.B.Pinheiro,I.C.deCastro,J.A.Reis Junior,M.P.Noia,C.Gurgel,M.C.TeixeiraCangussú, L.M.PedreiraRamalho, Photomed.LaserSurg. 2011, 29(9),619.
[14]D.Stasinopoulos, Photomed.LaserTher. 2005, 23(1),66.
[15]D.Stasinopoulos,C.Papadopoulos,D.Lamnisos, I.Stasinopoulos, Disabil.Rehabil. 2017, 39(5),450.
[16]D.Stasinopoulos,I.Stasinopoulos, Clin.Rehabil. 2006, 20(1),12.
[17]D.Stasinopoulos,I.Stasinopoulos,M.Johnson, Photomed. LaserTher. 2005, 23(2),225.
[18]D.Stasinopoulos,I.Stasinopoulos,M.Pantelis, K.Stasinopoulou, Photomed.LaserSurg. 2009, 27(3),513.
[19]M.Fenyo,Theoreticalandexperimentalbasisofbiostimulation bylaserirradiation. Opt.LaserTechnol. 1984, 16(4),209.
[20]A.L.B.Pinheiro,D.H.Pozza,M.G.D.Oliveira, R.Weissmann,L.M.P.Ramalho, Photomed.LaserTher. 2005, 23(5),485.
[21]P.Iordanou,E.G.Lykoudis,A.Athanasiou,E.Koniaris, M.Papaevangelou,T.Fatsea,P.Bellou, Photomed.LaserSurg. 2009, 27(2),261.
[22]K.Tada,K.Ikeda,K.Tomita, J.Trauma 2009, 67(5),1073.
[23]Y.Wang,Y.-Y.Huang,Y.Wang,P.Lyu,M.R.Hamblin, Biochim.Biophys.ActaGen.Subj. 2017, 1861(2),441.
[24]X.Wang,F.Tian,S.S.Soni,F.Gonzalez-Lima,H.Liu, Sci. Rep. 2016, 6,30540.
[25]X.Wang,F.Tian,D.D.Reddy,S.S.Nalawade,D.W.Barrett, F.Gonzalez-Lima,H.Liu, J.Cereb.BloodFlowMetab. 2017, 37 (12),3789.
[26]N.Y.Rots,A.Iavarone,V.Bromleigh,L.P.Freedman, Blood 1999, 93(8),2721.
[27]J.A.Lee,J.Spidlen,K.Boyce,J.Cai,N.Crosbie,M.Dalphin, J.Furlong,M.Gasparetto,M.Goldberg,E.M.Goralczyk, B.Hyun,K.Jansen,T.Kollmann,M.Kong,R.Leif, S.McWeeney,T.D.Moloshok,W.Moore,G.Nolan,J.Nolan, J.Nikolich-Zugich,D.Parrish,B.Purcell,Y.Qian,B.Selvaraj, C.Smith,O.Tchuvatkina,A.Wertheimer,P.Wilkinson, C.Wilson,J.Wood,R.Zigon,InternationalSocietyfor AdvancementofCytometryDataStandardsTaskForce, R.H.Scheuermann,R.R.Brinkman, CytometryA 2008, 73 (10),926.
[28]U.Gazi,L.Martinez-Pomares, Immunobiology 2009, 214 (7),554.
[29]D.A.Solovjov,E.Pluskota,E.F.Plow, J.Biol.Chem. 2005, 280 (2),1336.
[30]R.L.Kitchens, Chem.Immunol. 2000, 74(1),61.
[31]A.C.Chen,Y.Y.Huang,S.K.Sharma,M.R.Hamblin, Photomed.LaserSurg. 2011, 29(6),383.
[32]D.E.King,H.Jiang,G.O.Simkin,M.O.Obochi,J.G.Levy, D.W.Hunt, Scand.J.Immunol. 1999, 49(2),184.
[33]D.M.Sansom,C.N.Manzotti,Y.Zheng, TrendsImmunol. 2003, 24(6),313.
[34]R.Rutkowski,T.Moniuszko,A.Stasiak-Barmuta,B.KosztylaHojna,M.Alifier,K.Rutkowski,A.Tatarczuk-Krawiel, Arch. Immunol.Ther.Exp. 2003, 51(6),421.
[35]B.Vandanmagsar,Y.-H.Youm,A.Ravussin,J.E.Galgani, K.Stadler,R.L.Mynatt,E.Ravussin,J.M.Stephens, V.D.Dixit, Nat.Med. 2011, 17(2),179.
[36]J.W.Haukeland,J.K.Damås,Z.Konopski,E.M.Løberg, T.Haaland,I.Goverud,P.A.Torjesen,K.Birkeland,K.Bjøro, P.Aukrust, J.Hepatol. 2006, 44(6),1167.
[37]B.-Z.Qian,J.Li,H.Zhang,T.Kitamura,J.Zhang, L.R.Campion,E.A.Kaiser,L.A.Snyder,J.W.Pollard, Nature 2011, 475(7355),222.
[38]O.Takeuchi,S.Sato,T.Horiuchi,K.Hoshino,K.Takeda, Z.Dong,R.L.Modlin,S.Akira, J.Immunol. 2002, 169(1),10.
[39]R.S.Mahla,C.M.Reddy,D.Prasad,H.Kumar, Front. Immunol. 2013, 4,248.
[40]I.S.Sayed,A.Saafan,F.K.Abdel-Gawad,T.A.Harhash, M.A.Abdel-Rahman, J.Dent.Lasers 2015, 1(9),23.
[41]L.Gavish,L.Perez,S.D.Gertz, LasersSurg.Med. 2006, 38 (8),779.
[42]D.F.Martins,B.L.Turnes,F.J.Cidral-Filho,F.Bobinski, R.F.Rosas,L.G.Danielski,F.Petronilho,A.R.Santos, Neuroscience 2016, 324,485.
[43]C.H.Chen,C.Z.Wang,Y.H.Wang,W.T.Liao,Y.J.Chen, C.H.Kuo,H.F.Kuo,C.H.Hung, MediatorsInflamm. 2014, 2014,625048.
[44]M.Bredel,D.M.Scholtens,A.K.Yadav,A.A.Alvarez, J.J.Renfrow,J.P.Chandler,I.L.Yu,M.S.Carro,F.Dai, M.J.Tagge, N.Engl.J.Med. 2011, 364(7),627.
[45]T.Yoshioka,R.Nishikomori,J.Hara,K.Okada,Y.Hashii, I.Okafuji,S.Nodomi,T.Kawai,K.Izawa,H.Ohnishi, J.Clin. Immunol. 2013, 33(7),1165.
[46]W.Klein,A.Tromm,C.Folwaczny,M.Hagedorn,N.Duerig, J.T.Epplen,W.H.Schmiegel,T.Griga, Int.J.ColorectalDis. 2004, 19(2),153.
[47]F.S.Wong,C.Hu,L.Zhang,W.Du,L.Alexopoulou, R.A.Flavell,L.Wen, Ann.N.Y.Acad.Sci. 2008, 1150(1),146.
Howtocitethisarticle: FeehanJ,TripodiN, FraserS,etal.Polarizedlighttherapy:Shininga lightonthemechanismunderlyingits immunomodulatoryeffects. J.Biophotonics .2019; e201960177. https://doi.org/10.1002/jbio.201960177
