
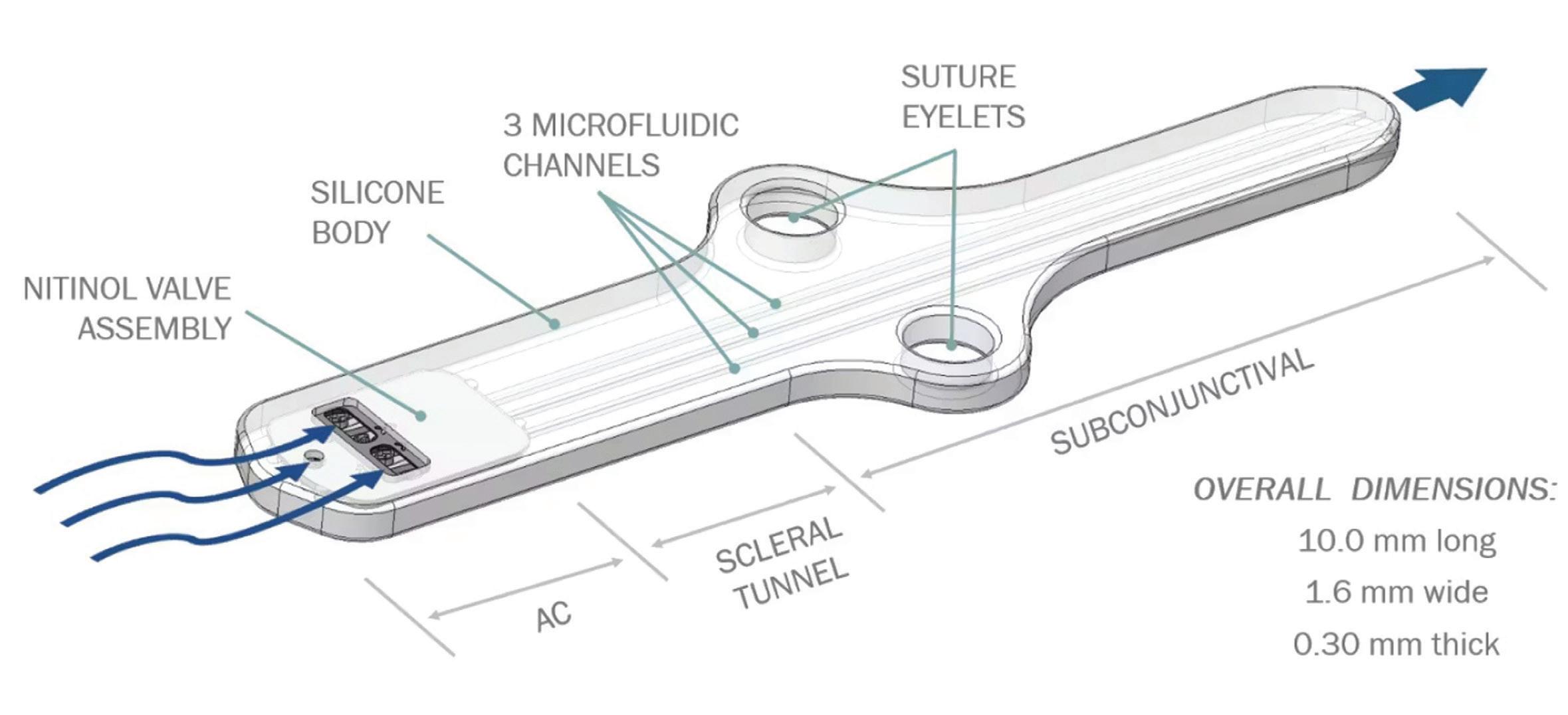
Engineering Services












Biocompatibility & Toxicology




Engineering Services












Biocompatibility & Toxicology




COMPREHENSIVE EXPERTISE: From early-stage product development and biocompatibility testing to scaled production, we provide the full spectrum of engineering, testing, and manufacturing services to ensure a seamless process and faster time-to-market.
CUTTING-EDGE CAPABILITIES: With advanced equipment and industry know-how, we offer specialized testing in drug stability, device verification, and biocompatibility services that few labs can match.
TAILORED SOLUTIONS: Whether you are a startup or an OEM, our team provides customized consultative support, guiding your product through its entire lifecycle with precision and care.















EDITORIAL
Editor in Chief Chris Newmarker cnewmarker@wtwhmedia.com
Managing Editor Jim Hammerand jhammerand@wtwhmedia.com
Senior Editor Sean Whooley swhooley@wtwhmedia.com
Associate Editor Skyler Rivera srivera@wtwhmedia.com
Editorial DirectorDeviceTalks Tom Salemi tsalemi@wtwhmedia.com
Managing EditorDeviceTalks Kayleen Brown kbrown@wtwhmedia.com
SALES
Ryan Ashdown 216.316.6691 rashdown@wtwhmedia.com
Jami Brownlee 224.760.1055 jbrownlee@wtwhmedia.com
CREATIVE SERVICES
VP, Creative Director Matthew Claney mclaney@wtwhmedia.com
VP, Marketing Annie Wissner awissner@wtwhmedia.com
Portfolio Marketing Manager McKenzie Burns mburns@wtwhmedia.com
LEADERSHIP TEAM
CEO
Matt Logan mlogan@wtwhmedia.com
SVP, Sales and Strategy Amanda Buehner abuehner@wtwhmedia.com
Mary Ann Cooke 781.710.4659 mcooke@wtwhmedia.com
Mike Francesconi 630.488.9029 mfrancesconi@wtwhmedia.com
PRODUCTION SERVICES
Customer Service Manager Stephanie Hulett shulett@wtwhmedia.com
Customer Service Rep Tracy Powers tpowers@wtwhmedia.com
Customer Service Rep JoAnn Martin jmartin@wtwhmedia.com
Customer Service Rep Renee Massey-Linston renee@wtwhmedia.com
Customer Service Rep Trinidy Longgood tlonggood@wtwhmedia.com
AUDIENCE DEVELOPMENT
Director, Audience Growth Rick Ellis rellis@wtwhmedia.com
Susan Powers 708.341.3370 spowers@wtwhmedia.com
Brian Toole 267.290.2386 btoole@wtwhmedia.com








888.543.2447








There’s no more electrifying field of technology right now than medtech.
It’s not automotive, where electric offerings from U.S. manufacturers are losing ground to overseas competitors. It’s not consumer tech, where VR and the metaverse (remember that?) have fizzled, and the next big thing has yet to materialize. And it’s not generative AI, which has failed to deliver on the hype.
But in the medical device industry, we’re using new materials, cuttingedge components and advanced manufacturing processes to save and improve lives. Minimally invasive and noninvasive medtech are the best examples of that.
Our annual minimally invasive medtech magazine focuses on device developers, engineers and product development experts working on these devices, systems and procedures.
This issue offers technical tips for materials like nitinol, shapememory polymers, expanded polytetrafluoroethylene (ePTFE) and textiles for heart valves. Beyond that, we cover high-voltage lithotripsy, pulsed-field technology for cardiac

ablation and other applications, including cancer, next-generation brain-computer interface design, and histotripsy technology that uses focused ultrasound acoustic liquefaction to enable incisionless surgery.
Also in this issue, we have an interview with Stanford Biodesign cofounder and Director Josh Makower, who warns of a “dire” threat to medtech innovation. And make sure to check out our preview of DeviceTalks Minnesota in Minneapolis on May 4.
Ahead of this issue, we’ve also launched our latest Nitinol Innovations Special report, diving into pulsed-field ablation systems through interviews with the OEM engineers and experts developing them. Download your free copy at wtwh.me/nitinolpfa.
Finally, I’m excited to welcome Skyler Rivera to our team as an associate editor. She’ll cover medtech innovation for Medical Design & Outsourcing online and in print, and medical device business news at our MassDevice website. Learn more about Skyler at wtwh.me/rivera.
As always, I hope you enjoy this edition of Medical Design & Outsourcing. Thank you for reading.
Jim Hammerand | Managing Editor
Medical Design & Outsourcing

HERE’S WHAT WE SEE:
High-energy minimally invasive systems are powering medtech
COMPONENTS:
Powering the future of lithotripsy technology with high voltage
ENERGY:
Galvanize aims pulsed electric fields at chronic bronchitis and cancer
MATERIALS:
Why medical device innovators must rethink PFAS now
POLYMERS:
Developing biocompatible shape-memory polymers
MEN’S HEALTH:
Francis Medical’s Vanquish system offers new features for prostate cancer ablation
TEXTILES:

How to optimize biomedical textile design for heart valves
TUBING:
Stereotaxis designed the MAGiC cardiac ablation catheter for consistent and safe tissue contact
DEVICETALKS:

IS MAKING NEXT-GEN GLAUCOMA IMPLANTS SAFER AND LESS INVASIVE
Minimally invasive glaucoma surgery is using nitinol to make symptom management for the progressive eye disease safer and increasingly personalized.


26 EPTFE MIGHT BE THE NEXT NITINOL-LIKE MEDTECH MATERIAL
If you’re looking for a medtech material enabling innovation in the same way that nitinol has, you don’t need to look far to find expanded polytetrafluoroethylene (ePTFE).


29 ‘IT IS DIRE.’ JOSH MAKOWER WARNS OF THE ‘BIGGEST CHALLENGE’ TO MEDTECH INNOVATION
“It’s getting worse, not better,” the Stanford Biodesign co-founder and director said in an interview.

32 CAN PETAL CUT THE CUTS FROM SURGERY?
Petal Surgical co-founder and CEO Prash Chopra envisions acoustic liquefaction for incisionless surgery and much more. 37 ABILITY NEUROTECH SAYS ITS BCI SYSTEM’S ELECTRODES AND SPEED SET IT APART
Brain-computer interfaces (BCIs) have made their mark on medtech and Ability Neurotech’s leaders hope their minimally invasive platform’s unique features propel it forward.

Engineered as an alternative to film-cast PTFE, PFX FlexTM Sub-Lite-WallTM is a next-gen catheter liner that delivers proven lubricity, enhanced bond strength, and expanded sterilization options – all without PFAS.














Off-the-shelf power supplies for lithotripsy systems can save months of development time if you select the right option early enough.

By Todd Huston Advanced Energy
Lithotripsy is a noninvasive medical procedure used to break down kidney stones and calcified tissues.
First developed in the 1980s, this technology has evolved significantly in terms of both safety and precision and is now being used for therapies at the intravascular level.
These evolutions are enabling the treatment of symptoms in a greater range of conditions and leading to shortened hospital stays and expanding outpatient treatment options.
These advances rely on highvoltage pulsed power systems. In this article, we’ll look at the challenges of implementing power supplies in lithotripsy and why power needs to be considered early in the design cycle.
Extracorporeal shock wave lithotripsy (ESWL)
ESWL remains the most widely used form of lithotripsy. It uses focused pressure waves generated outside of the body and
delivered to targets with millimeter-level precision for the fragmentation of kidney or urethral stones without harming the surrounding tissue. These shock waves are generated using three main types of lithotripsy systems: electrohydraulic, piezoelectric and electromagnetic.
Each of these methods have different retreatment rates, pain levels, and power profiles. Electrohydraulic systems are used for large (2 cm) stones and calcified lesions using shock waves that require rapid discharges of up to 25 kV. Piezoelectric systems are far gentler, with less collateral tissue damage and pain. Crucially, these systems can be deployed in outpatient settings to simplify treatment.
More recently, electromagnetic systems have been developed to enable more reproducible results via precisely controlled electromagnet coils or membranes that generate waves with a focal pressure of between 40 and 100 MPa (up to 14,000 psi) and a negative phase of around 5 to 10 MPa (up to 1,400 psi).
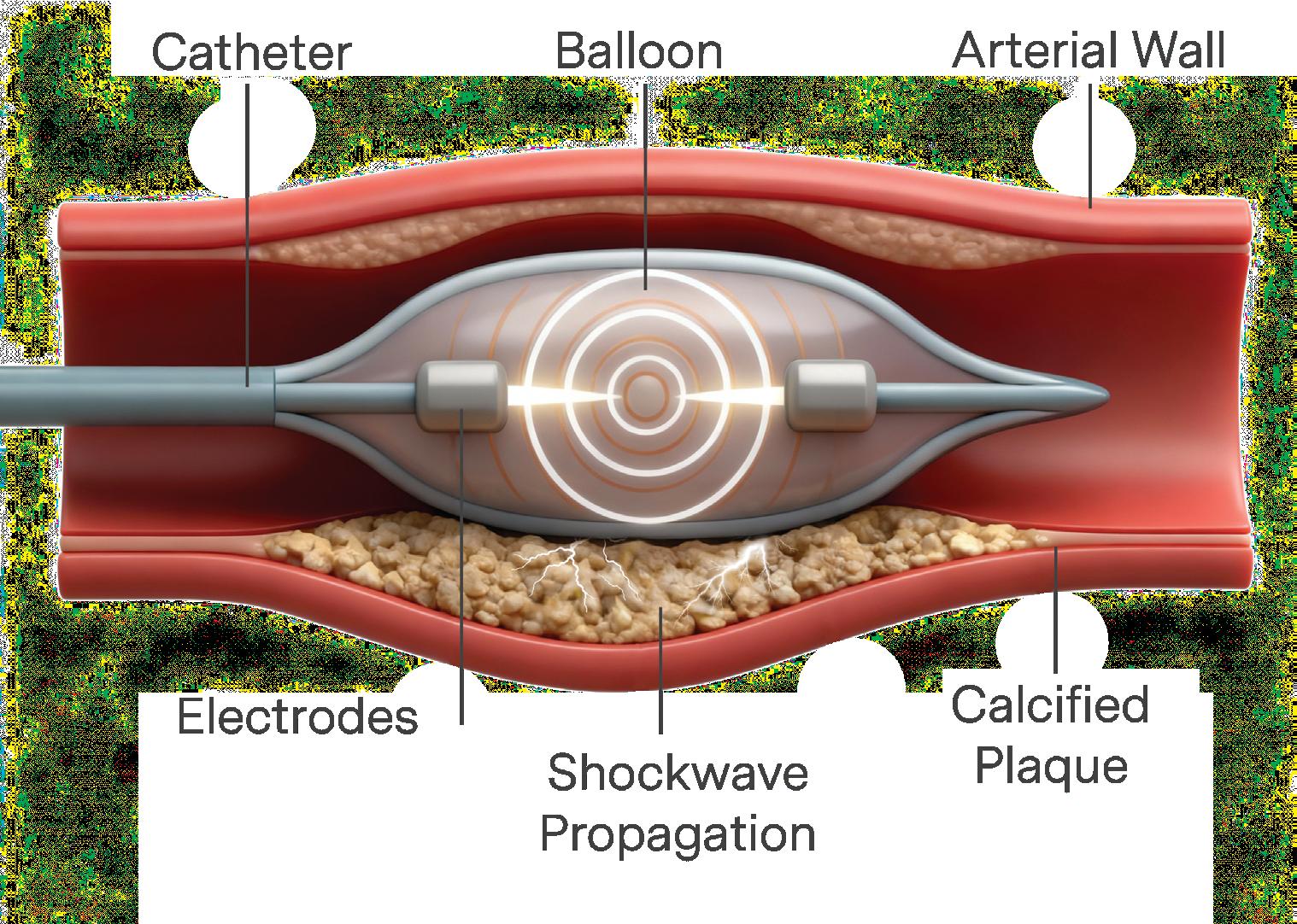
Intravascular lithotripsy (IVL)
Lithotripsy has also recently been applied to treat calcified arterial plaque, which is not only a symptom of age, but conditions such as diabetes. Unlike traditional ESWL methods, IVL delivers controlled, low-energy shock waves from directly inside the blood vessel via a balloon catheter. Shock waves soften and break up calcified lesions, making stent placement in coronary surgery safer, more effective, and more predictable with fewer failures.
There are still adoption barriers for IVL such as cost, training and access. However, this minimally invasive >>
technology is seeing considerable market growth and investment. One example is Shockwave Medical, a leading specialist developer of these systems that was acquired by Johnson & Johnson for $13.1 billion in 2024.
Lithotripsy power supply requirements
The ability to deliver power reliably is an engineering hurdle as well as a regulatory and liability challenge. Any failures could potentially lead to patient risk, device recalls and reputational damage.
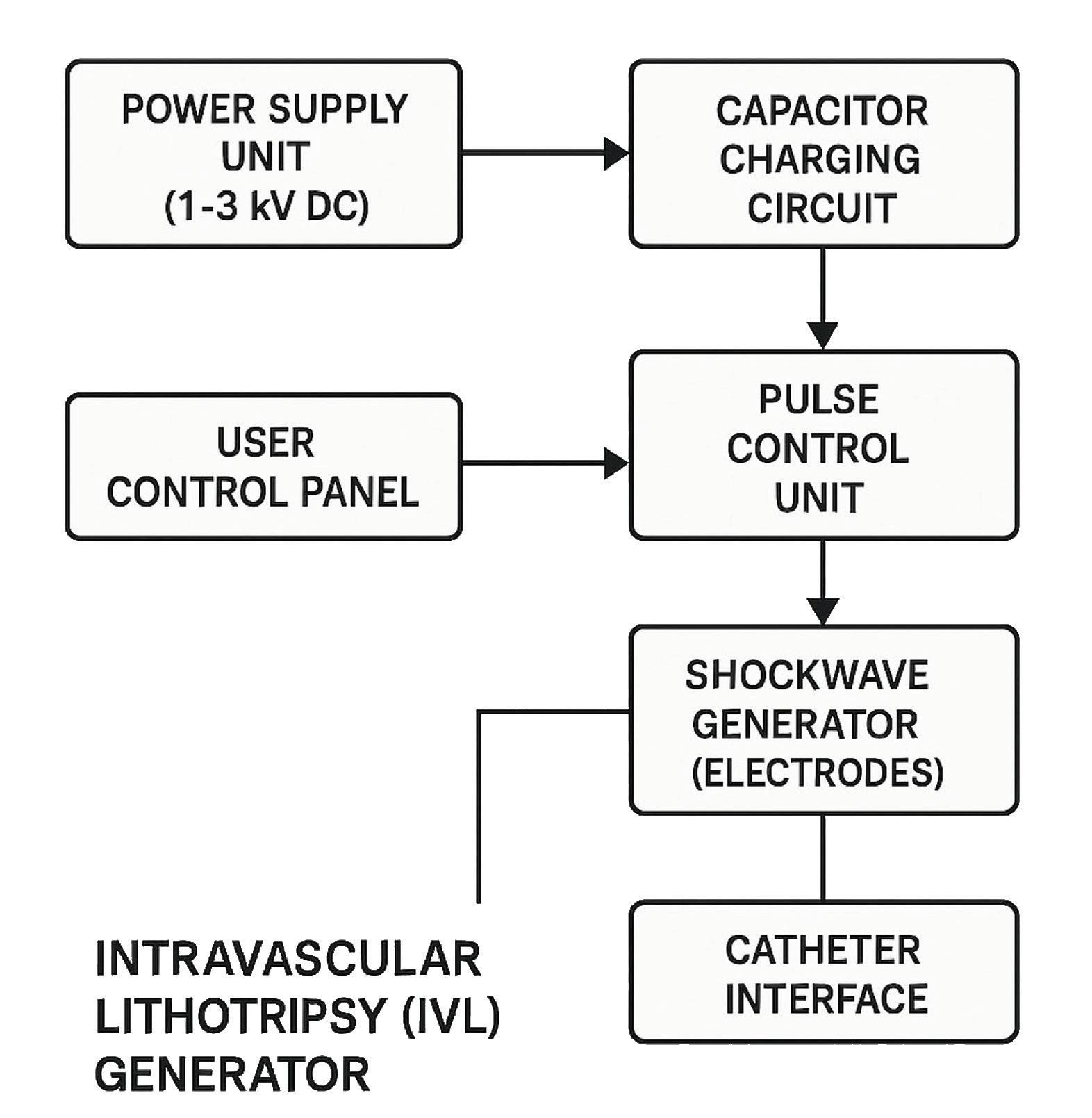
For power, the heart of both ESWL and IVL systems is a high-voltage capacitor charging circuit requiring a DC-DC power supply.
For ESWL systems, these typically operate between 3 kV and 25 kV. However, some piezoelectric systems will go as low as 1 kV. These systems need to be able to charge capacitors within milliseconds to enable repetition rates of up to 100 pulses per second and peak current discharges into a spark-plug-like electrode that exceed 100 A.
The entire system must also withstand the powerful electromagnetic pulses (EMP) generated during each discharge, requiring robust shielding and protection circuitry. For IVL, the technical characteristics are different, requiring controlled pulse timing and location with less powerful pulses to avoid damaging delicate vascular structures. The voltage range is therefore set to between 1 and 3 kV DC, with a pulse frequency of around 1 pulse per second (in cycles of 30 pulses and 10-second pauses) and a peak pressure of 5 to 10 MPa (700 to 1,400 psi). The electrode is also different, with needleplate electrodes used to generate nonoscillatory shockwaves.
Beyond delivering high voltage pulses quickly and reliably, these systems must meet stringent medical standards, implementing means of operator (MOOP) and patient protection (MOPP). As such, power supplies for both ESWL and IVL must satisfy IEC 60601-1 safety requirements, the global benchmark for medical electrical equipment, which sets out limits on leakage currents, insulation and isolation.
ESWL systems — where patient contact is external — are typically required to meet body floating (BF) standards. Therefore, power supplies must implement reinforced isolation from the mains, with patient leakage currents limited to below 100 µA under normal conditions and less than 500 µA in single fault conditions.
On the other hand, as IVL systems are catheter-based and deliver pulses directly inside coronary or peripheral vessels, power supplies for these must meet the most stringent classification of protection: cardiac floating (CF). For this, patient leakage current limits are set a full order of magnitude lower, less than 10 µA in normal and sub 50 µA in singlefault conditions.
Several engineering challenges remain after regulatory compliance. High-voltage isolation is essential, often requiring dielectric barriers that are rated in the tens of kilovolts to protect both patients and operators. As above, this is especially critical for IVL systems. Consistent, tightly regulated capacitor charging with minimal ripple is essential. Any variation in pulse delivery can lead to incomplete stone fragmentation in ESWL or damage to delicate vascular structures in IVL.
Furthermore, as each discharge also generates strong electromagnetic pulses, robust shielding, snubber circuits, and surge protections must be in place. Finally, thermal management must be considered, with the system’s high repetition rates and large peak currents producing considerable thermal loads. Air, liquid or hybrid cooling can be implemented. Failure to do so can reduce reliability, especially in more compact and portable systems.
Developing a power supply for lithotripsy
In the past, these systems used custom power supplies developed by the lithotripsy system provider or with outsourced power specialists, allowing precise specification control and compliance. However, this approach required certification testing, increasing development time and costs.
A more cost-effective alternative is to design in a commercial off-the-shelf module. Pre-certified BF-rated supplies are now well established, and CF-rated modules have now been on the market for at least a year. Advanced Energy has introduced CF-compliant supplies designed specifically for applications such as intravascular lithotripsy. These allow system developers to accelerate time-to-market by removing the need for separate supply certification.
Taking this route can reduce the development time by several months — and even up to a year — with the supply requiring only integration verification. However, power supplies are often considered relatively late in the design cycle, at which point size and integration constraints may rule out the use of standard modules. Depending on the project’s constraints, it may then be possible to use a standard-modified supply, but this is not guaranteed.
It is advisable to plan for the power architecture early, ensuring space is reserved and that the system’s electrical and mechanical requirements match those of pre-certified options where possible.
Expanded clinical applications of lithotripsy
The use of lithotripsy is being considered for orthopedics, neurology and oncology, among others. Future developments of this technology will place more demanding requirements on the underlying power supply: needing both the ability to deliver more tightly controlled, lower-energy pulses that avoid neurological damage, for example, and higher energy densities for deep tissue penetration. In parallel with these large hospital-based systems, the push toward portable and outpatient systems are driving a simultaneous need for compact, lightweight designs that can be run from batteries.
Meeting these challenges will open significant revenue streams from new applications and outpatient devices. However, this growth depends on continued advances in high-voltage power electronics, such as faster and more efficient capacitor charging, improved dielectric isolation in smaller footprints, integrated EMI suppression, and thermal solutions that scale to high repetition rates.

ABOVE: Advanced Energy’s High Power C DC-DC modules are pre-certified for BF applications and output voltages up to 60 kV.
BELOW: Advanced Energy’s NCF family are the industry’s first pre-certified CF-rated offthe-shelf supplies.
Ultimately, the evolution of lithotripsy will be paced as much by innovations in power supply design as by clinical discovery, with regulatory compliance and technical performance remaining inseparable.
Todd Huston is director of strategic marketing for medical power at Advanced Energy. He previously served as an electrosurgery product manager for the tumor ablation portfolio at a leading medical device company.
Images courtesy of Advanced Energy



Cadence isn’t just another supplier—we’re the MedTech & Pharma partner engineered for integrated, end-to-end solutions. One partner. One integrated process. From design through commercialization, we accelerate launches, reduce risk, and raise the bar on quality and compliance.
Cadence: delivering where it matters most.
Led by the former CEO of Shockwave Medical, this minimally invasive startup raised $100 million in 2025.
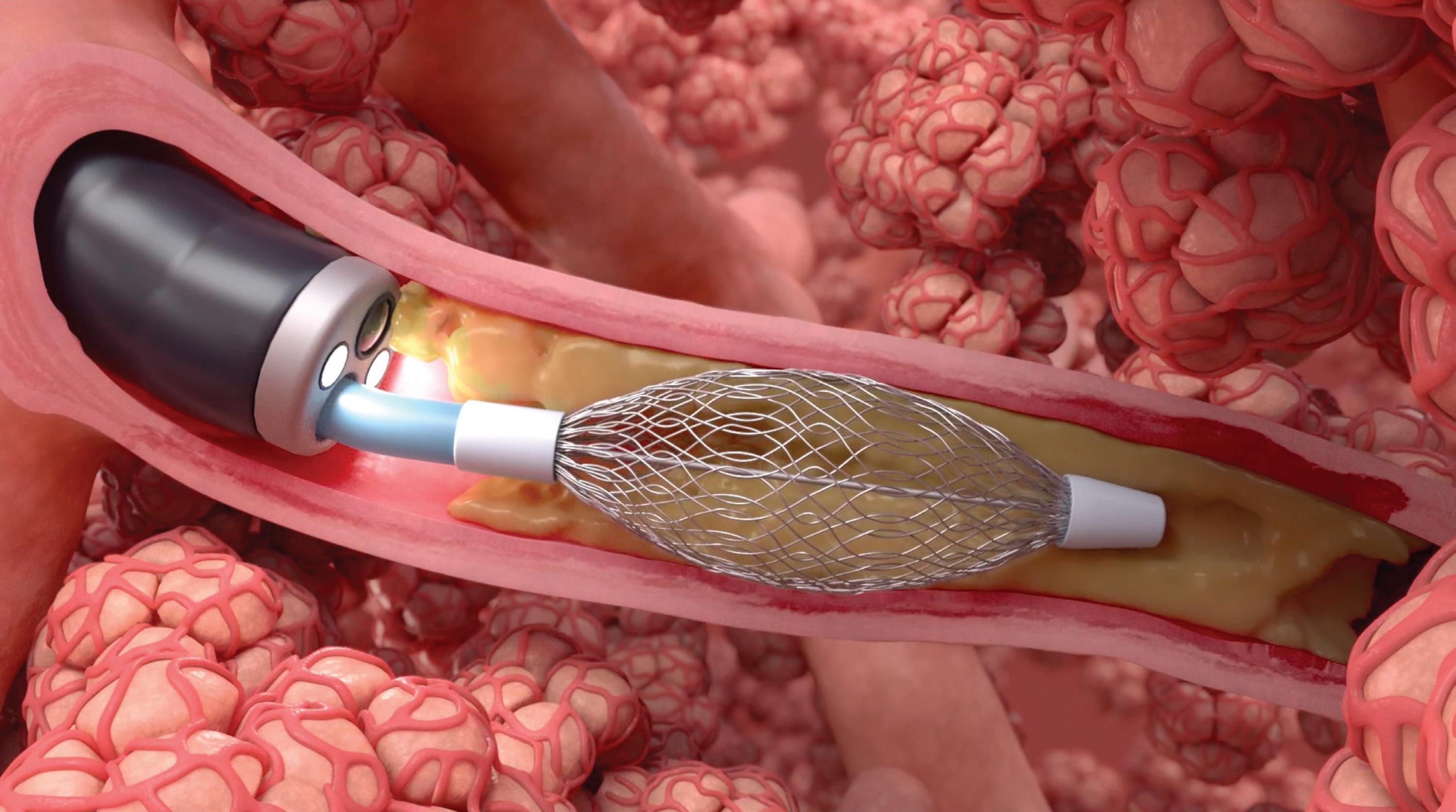

illustration depicts bronchial rheoplasty using the Galvanize Therapeutics RheOx catheter, which delivers energy to ablate abnormal mucus-producing cells in the airways.
Galvanize Therapeutics is making waves with a new CEO, $100 million of fresh funding and nonthermal, minimally invasive ablation technology.
The pulsed electric field (PEF) device developer announced its Series C funding round and the hiring of former Shockwave Medical CEO Doug Godshall as CEO of Galvanize in September 2025.

Godshall has chaired the Galvanize board since 2021. In 2024, he led the $13.1 billion sale of Shockwave to Johnson & Johnson MedTech, which is now poised to become the world’s largest medical device company thanks in large part to growth from Shockwave’s intravascular lithotripsy platform.
Formed in 2022 by a threeway merger, Galvanize is backed by Sofinnova Partners — as was Shockwave — and Galvanize is also developing systems for catheter-delivered energy. Based in Redwood City, California, the startup already has a CE mark for
its RheOx system for treating chronic bronchitis symptoms and FDA 510(k) clearance for its Aliya and Inumi devices for soft tissue ablation.
Oncology is the next target, with technologies in the pipeline for lung cancer, hepatocellular cancer (the most common liver cancer), renal cell carcinoma (the most common kidney cancer) and breast cancer. Those programs are in the pre-clinical and/ or feasibility stages, according to the company’s website.
Pulsed-field energy is the nonthermal, minimally invasive technology behind the rush of pulsed-field ablation (PFA) systems for treating atrial fibrillation. Like those cardiac PFA catheters and generators, Galvanize has developed ablation systems that generate and deliver short-duration, high-voltage energy to create electrical fields that kill cells via electroporation while sparing nearby nerves and other structures.
The RheOx bronchoscopic system ablates “abnormal mucus-producing cells in the airways, leaving the extracellular matrix intact so that the epithelium may rapidly regenerate,” Galvanize says at its website.
Similarly, the Galvanize Aliya PEF System (which includes the Aliya Generator, the Aliya Ablation Device and the Ivy Biomedical Model 7600 cardiac monitor) and the Inumi Flex needle both deliver PFE to induce cell death.
Galvanize says its Aliya Generator has an algorithm that interprets trigger signals from the cardiac monitor and “will only allow the initiation and delivery of energy output when the patient’s heart rate is within acceptable limits.”
That energy is delivered percutaneously via the monopolar Aliya Ablation Device, which includes the Aliya Needle and the Aliya Electrode.
“Both the needle and electrode are designed for sterile, single use,” Galvanize explained in its FDA 510(k) submission. “The Aliya Needle consists of a 19-gauge, 20-cm needle with a
it the first endoscopic PEF ablation system. The Inumi Flex “has a 160cm working length and is compatible with endoscope working channels with a minimum of 2 mm diameter,” per Galvanize’s FDA filings.
Galvanize says its Aliya System (both percutaneous and endoscopic use) allow for precise, constant ablation zones without heat sink effects.
Galvanize’s cancer pipeline
Galvanize says data from its preclinical and early clinical studies “suggest the nonthermal cell death induced by PEF may release antigens from the dying tumor cells that may stimulate an immune response for a system effect beyond focal ablation. This may be synergistic with and additive to standard of care therapies.”
In August 2025, the company said it enrolled the first patient in the Propel Registry, a multicenter observational study evaluating the Aliya system for soft tissue
population outside of a prospective trial setting,” Galvanize Chief Medical Officer Dr. Bill Krimsky said in the announcement. “By systematically capturing procedural data, clinical outcomes, and physician experience, we aim to inform evidencebased integration of our PEF ablation into clinical care pathways.”
In May 2025, Galvanize shared results from its Incite ES study of PFE in non-small cell lung cancer (NSCLC) patients.
“Aliya PEF ablation showed 100% technical success and integrated seamlessly into existing care pathways,” study investigator Dr. Marcelo Jimenez at Salamanca University Hospital in Spain said in a news release. “By performing ablation and biopsy in a single anesthetic event, this study showed that Aliya PEF offered the ability to integrate Aliya into existing clinical workflows without requiring a second anesthetic event for early-stage NSCLC.”
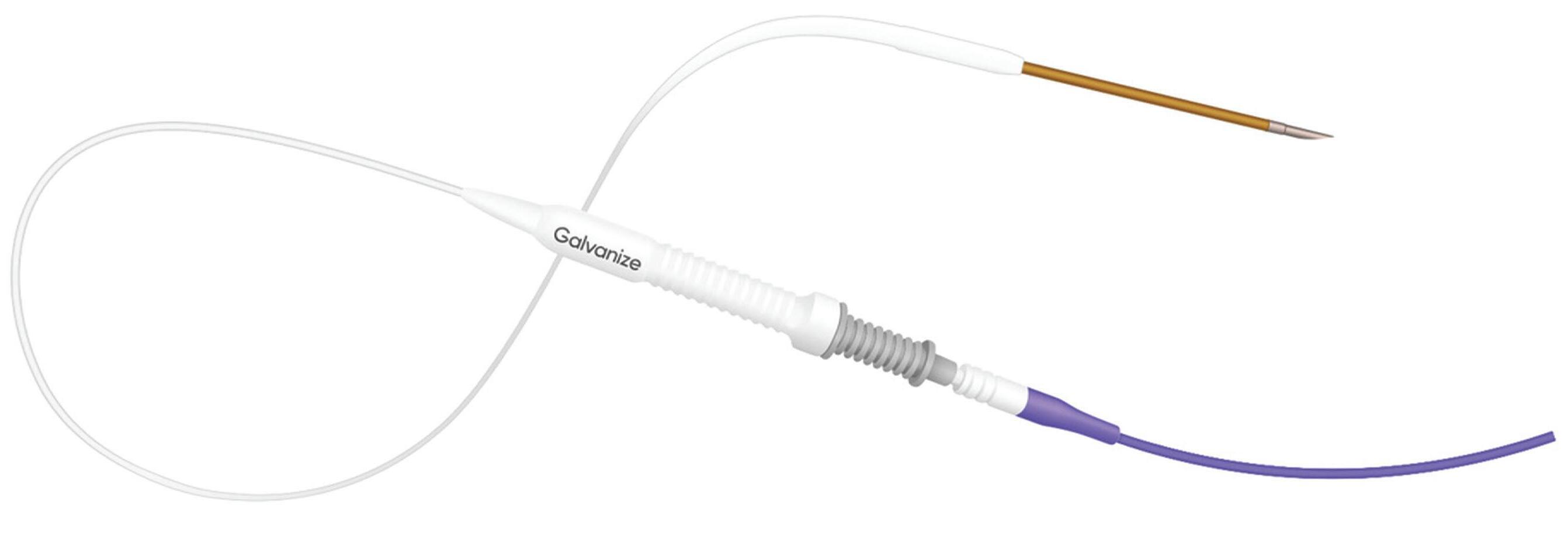
1-cm exposed tip and a nested stylet (which is removed once the Aliya Electrode is in position). The Aliya Electrode consists of a 21-cm probe that connects with the needle, a 3-meter cable, and a plug that connects with the Aliya Generator. When connected to the Aliya Generator, the Aliya Ablation Device delivers PEF energy to the target zone at the distal 1 cm of the needle.”
The Inumi Flex needle also works with the Aliya System, making
BY SYSTEMATICALLY CAPTURING PROCEDURAL DATA, CLINICAL OUTCOMES, AND PHYSICIAN EXPERIENCE, WE AIM TO INFORM EVIDENCEBASED INTEGRATION OF OUR PEF ABLATION INTO CLINICAL CARE PATHWAYS.”
“
lesions, including primary and metastatic lesions in the lung, liver and other organs.
“Interventional pulmonology has been focused on diagnosing lung cancer over the last few decades,” Director of Interventional Pulmonology and Associate Professor of Medicine at Duke University Medical Center Dr. Kamran Mahmood said in a news release after completing the first case. “Now we are shifting our attention to ablating the lesion.”
Galvanize said the registry would enroll up to 1,000 patients across up to 50 U.S. centers, following patients for up to two years.
“The Propel Registry is an important step in understanding how Aliya performs in a broad, heterogeneous patient
In Galvanize’s announcement of the Series C fundraise and his hiring as CEO, Godshall said he has “grown increasingly enthusiastic about the company’s prospects over the last year as the team continued advancing our portfolio and commencing our initial commercial efforts.
“Galvanize’s Aliya and RheOx technologies hold great promise in the fields of solid tumor oncology and chronic bronchitis,” he continued. “There are millions of patients who are underserved by today’s largely pharmaceutical approaches, and I feel fortunate to be able to join the team as we seek to meaningfully improve the outcomes of those who are suffering with these chronic diseases.”
medical device innovators must rethink PFAS now
Staying compliant and leading the market means planning for a future that’s already coming, and PFAS won’t be part of it.

By Nathan Rohner Lubrizol

PThermoplastic polyurethane (TPU) is a unique type of plastic bridging the gap between rubbers and plastics. The key to its versatility is that its hardness can be customized to be as soft as rubber or as hard as rigid plastics.
er- and polyfluoroalkyl substances (PFAS) are a huge family of compounds up to 15,000 strong.
These “forever chemicals” are in our water, soil, mascara, cheeseburger wrappers, and even the medical device supply chain, though not always in the way people think.
Known for their thermal stability, chemical resistance, lubricity and biocompatibility, polymers comprised or produced with PFAS have become the default for a wide range of medical applications. Fluoropolymers like polytetrafluoroethylene (PTFE) and fluorinated ethylene propylene (FEP) are used in catheter liners, lubricious coatings and heat-shrink tubing, just to name a few.
But this functionality comes with an environmental trade-off. Some PFAS are persistent, bioaccumulative and toxic, the “PBT” trifecta that drives today’s
regulatory pressure. These compounds don’t break down easily, can build up in the body and may be linked to certain health concerns. And while finished medical devices rarely leach PFAS, the upstream manufacturing and disposal (including incineration) of PFAScontaining materials are where real environmental risk shows up.
It’s the supply chain
Medical device exemptions still exist for now, particularly in the U.S., but that hasn’t stopped the industry from moving toward alternatives. Global suppliers are backing out of fluoropolymer markets and regulatory definitions are broadening. In the EU, anything with a carbon-fluorine bond is fair game under the Registration, Evaluation, Authorization and Restriction of Chemicals (REACH) regulation.

For medical device OEMs, the concern is regulatory and operational. If a supplier exits production of fluoropolymer-based raw materials for liners, shrinks and coatings, that leaves developers scrambling to qualify replacements on timelines that often exceed five to 10 years. Missing a materials window can mean losing an entire innovation cycle.
To stay ahead, many are already scouting alternatives, prototyping components and setting up validation programs for the next generation of devices. They’re not implementing changes yet, but they are watching for developments closely.

TPU as the challenger
Thermoplastic polyurethanes (TPUs) have entered the conversation as a viable PFAS alternative. Though sometimes perceived as “sticky” or overly soft, medical-grade TPUs have proven in head-to-head tests to match PTFE and FEP in dry and wet coefficient of friction against silicone, even before any specialty additives are introduced.
These materials are processed without fluorinated chemistry, meaning there are no intentionally added PFAS, and there’s no need for smallmolecule PFAS intermediates or processing aids. TPU synthesis relies on components like diols and alcohols where there are no carbonfluorine bonds involved. The result is strong extrusion quality, low-temperature processing, regulatory clarity and flexible compounding options that allow for customized performance tuning.
PFAS can show up as background contamination, even in city water. That’s why the industry is shifting toward terms like “no intentionally added PFAS” or “no measurable PFAS.” Lubrizol, for example, doesn’t add PFAS to its medical TPU materials and works with compounders to remove any residual additives that might come in via third-party inputs.
What’s slowing adoption?
Perception and risk aversion is slowing adoption, as TPU is often misunderstood. Even with data in hand, developers sometimes won’t believe in its performance until they feel it for themselves. There’s also strategic delay as OEMs are cautious about making early material changes. There’s a natural hesitation to invest in qualifying a new solution that could later be outpaced. Some teams are opting to watch how larger players approach PFAS replacement before making their own moves.
But supply chain disruptions don’t follow a schedule. When access to fluoropolymers disappears, either due to legislation or market shifts, retrofitting can become a scramble. Getting ahead of the curve means owning your innovation path instead of reacting to someone else’s.
TPUs won’t replace every use case. They won’t act like heat shrinks, and they won’t replicate the full thermal resistance of PTFE. But for liners, long-term implants, coatings and components where lubricity and biocompatibility are key, TPUs can deliver and are already being used successfully in many of these applications.
There is no miracle drop-in that checks every box PTFE does. The market offers additives that mimic lubricity or bio-based polymers pitched as sustainable PTFE replacements. But there remains no wholesale solution matching the full thermal-chemicallubricious package.
That said, Lubrizol has already identified TPU grades that closely match commercial fluoropolymer performance in key dimensions, and the next step is getting them into the hands of OEMs for testing in real-world devices.
It’s about preparedness. Whether regulations arrive in 2027 or 2030 or
beyond, the materials conversation is shifting fast, and developers who wait may find themselves locked out of critical markets or dependent on unstable supply chains.
Now is the time to test alternatives, run comparisons and identify gaps. In medtech, staying compliant and leading the market means planning for a future that’s already coming, and PFAS won’t be part of it.
Nathan Rohner is a senior scientist at Lubrizol, where he leads polymer innovation for pharmaceutical and medical device applications. With a background in biomedical engineering and more than a decade of experience in materials R&D, he specializes in the development of customized thermoplastic polyurethanes (TPUs) for combination products, implantable systems and controlled drug-delivery platforms. Rohner holds a doctorate in bioengineering from the Georgia Institute of Technology.

After solving several challenges for our implant, our team sees a promising future for shape-memory polymers in other medical devices.

Embolization Inc. received FDA 510(k) clearance in 2025 for a coil device using biocompatible shape-memory polymers. The Nitinol Enhanced Device (NED) is intended for arterial and venous embolization in peripheral vasculature, specifically achieving better vascular occlusion while minimizing artifacts in CT and MR imaging that occur with traditional metal devices.
Shape-memory polymers are key to the NED’s use and performance. While our efforts are focusing on applications within the venous system, what we learned from our work with these polymers could help medtech developers, others in the medical community, and ultimately, their patients.
Embolization Inc. makes its Nitinol Enhanced Device (NED) with a proprietary shape-memory polymer. Illustration courtesy of Embolization Inc.
The problem
Metals such as stainless steel and nitinol are commonly used biocompatible materials. Stainless steel offers excellent corrosion resistance in the body. It has been used in medical devices since the 1920s and in MRI applications since the 1980s. Nitinol, a nickel-titanium alloy, came to light in the medical device field in the early 1970s with the development of an intrauterine contraceptive device that could be deformed for insertion and then regain its shape. Nitinol is now widely used in medical devices due to its unique shape-memory and elasticity properties.
These characteristics make nitinol (and to a lesser extent, stainless steel) ideal for many minimally invasive procedures. As a metal, nitinol also shows up well in fluoroscopy. However, it can have significant artifacts (shadows) when
used in CT and MR imaging. The potential results — including compromised image quality and misdiagnosis — are significant.
The solution
Shape-memory polymers were developed in the 1980s, but it wasn’t until the 2010s that we started to see the first commercial devices incorporating them. These smart materials can “remember” a specific shape and return to it after being deformed, making them effective in a host of medical device applications.
Useful for many of the same procedures as nitinol, the advantage is that a shape-memory polymer does not have the same type of imaging artifacts because it’s not metallic.
Lessons learned in using shapememory polymers
As we worked through design and development of our coil device, we encountered and addressed four main challenges:
1. Transition temperature: Maintaining shape memory at the appropriate temperature is critical. That means body temperature, so that the material remains flaccid at other temps, but takes its pre-determined shape when it reaches body temperature.
2. Biocompatibility: If a polymer reaction was an absolute known, you could usually come up with some very good biocompatible materials. But unique and novel polymers (specifically the crosslinkers and initiators) are not known biocompatible materials. To meet biocompatibility standards, we learned we needed to define and take careful steps to monitor the percent of polymerization and create ways to remove non-polymerized materials.
3. Radiopacity balance with strength: Along with defining the correct amount, we identified a tradeoff between radiopacity and structural soundness, or strength. To make the polymer more radiopaque, you must add a filler. This is basically adding certain atoms in the molecular chain. But that action decreases strength. In practice, we had to consider how strong or weak our polymer was at given diameters (our polymer diameter is .018 in.) and then see what effects changes in radiopacity had on polymer strength. It’s a meticulous back-andforth process to achieve the right balance. Adding to the challenge is that anytime you start changing the polymer formulation, the transition temperature (that threshold where the material moves from flaccid to stiff) can easily change. We spent a lot of time balancing all of these factors.
4. FDA criteria: Along the way, we encountered changes in FDA criteria, particularly in the primary way it looks at biocompatibility. Initially, the criteria was based on cell culture and animal testing, then moved to extractables and leachables testing. One of the issues we encountered was that the solvents used in extractables and leachables testing can actually attack the shape-memory polymer. To fully meet all criteria, we conducted extensive animal testing as well as extractables and leachables testing. In doing so, we learned that we had to clean our product, leaching the product to remove any remains of unreacted materials.
For our particular technology, other applications include more form factors for use in more areas of the body. For instance, we’ve already developed and
tested a platform to help neurological embolisms clot.
The technology will lend itself to form factors for embolization beyond coils, such as with a plug, combination plugand-coil system, a long, skinny device, or even something more like a sponge.
While Embolization’s work is concentrated on applications within the venous system, we believe the future of shape-memory polymers is significant in many more areas of device development.
Jim Kasic is the CEO of Embolization Inc., chair of Boulder BioMed, and formerly served as president and CEO of Sophono (acquired by Medtronic) and president of OrthoWin (acquired by Zimmer Biomet). Kasic has more than 30 years of experience in the Class I, II, and III medical device industry and holds more than 40 U.S. and international patents.








Minimally Invasive Device Solutions
Co-development
Wire & Catheter
Guidewires,









By Skyler Rivera Associate Editor
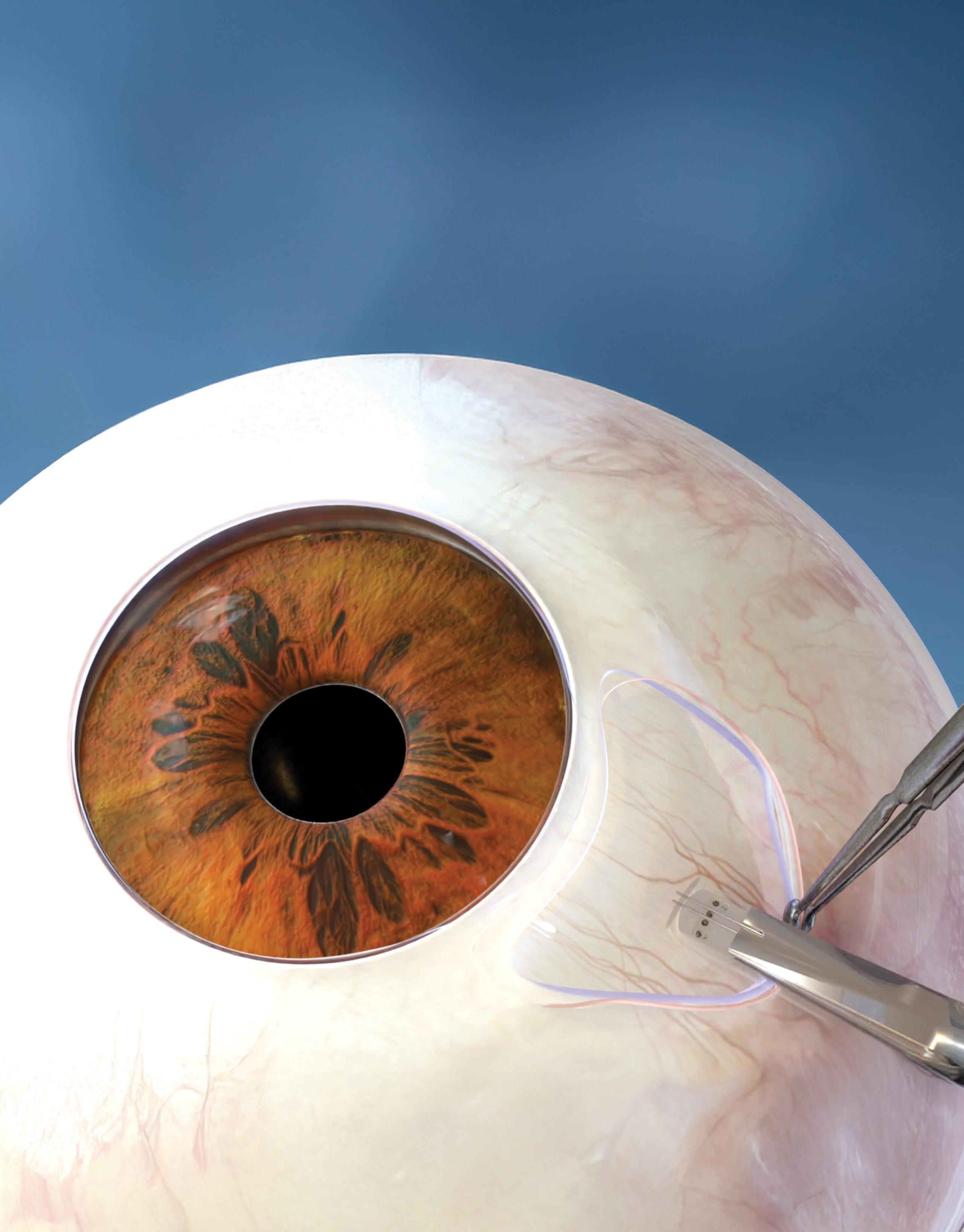
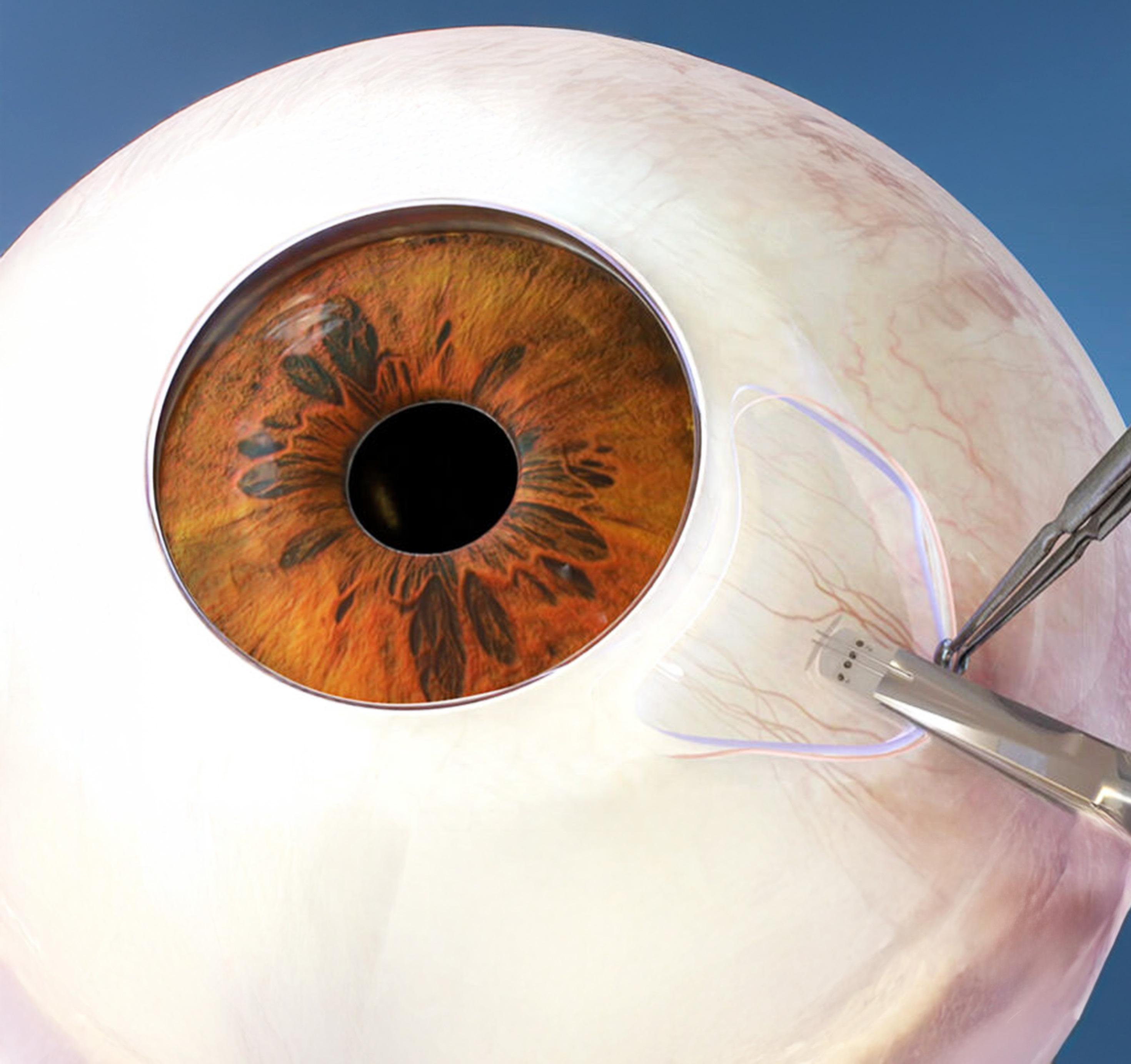
Nitinol is making next-gen glaucoma implants safer and less invasive
Alcon’s implant is already on the market, while a Shifamed startup is using shape-memory nitinol for a new system.
Minimally invasive glaucoma surgery (MIGS, also known as microinvasive glaucoma surgery) is using nitinol to make symptom management for the progressive eye disease safer and increasingly personalized.
Glaucoma is sometimes called the “silent blinder” because of its lack of noticeable symptoms in its early stages. It is the leading cause of irreversible blindness for people over 60 years old, according to the American Academy of Ophthalmology. Currently, there’s no cure for the disease, but treatment can slow or prevent vision loss.
What is glaucoma?
In a healthy eye, clear fluid called aqueous humor moves through the eye, maintaining intraocular pressure (IOP)
and providing nourishment. As aqueous fluid is constantly produced, it flows through the eye, traveling toward the drainage angle through a spongy web of tissue called the trabecular meshwork and exits through the Schlemm’s canal to be absorbed into the bloodstream.
Glaucoma occurs when there’s a problem in this drainage system. When the fluid doesn’t drain properly, eye pressure increases and can damage the optic nerve, leading to vision loss and eventually blindness. Openangle glaucoma, the most common type, occurs when the eye’s drainage angle appears open but the fluid has trouble draining through the trabecular meshwork. Most people don’t notice symptoms of glaucoma until they begin to lose vision, meaning significant optic nerve damage has already occurred. >>
Traditional glaucoma treatments
Treatment for glaucoma focuses on reducing intraocular pressure to limit damage to the optic nerve and preserve sight. Eye drops to reduce the amount of aqueous fluid are the first line of defense if caught early, but due to glaucoma’s silent onset, surgical treatments are commonly necessary. If the main pathway for drainage is blocked, surgeons will create a new pathway in an outpatient procedure called a trabeculectomy. The surgeon creates a tiny opening in the white part of the eye (sclera), allowing fluid to drain outside the eye to a small blister-like pocket called a bleb, where it’s absorbed back into the body. If a trabeculectomy is unsuccessful, the surgeon might consider a glaucoma tube shunt implant.

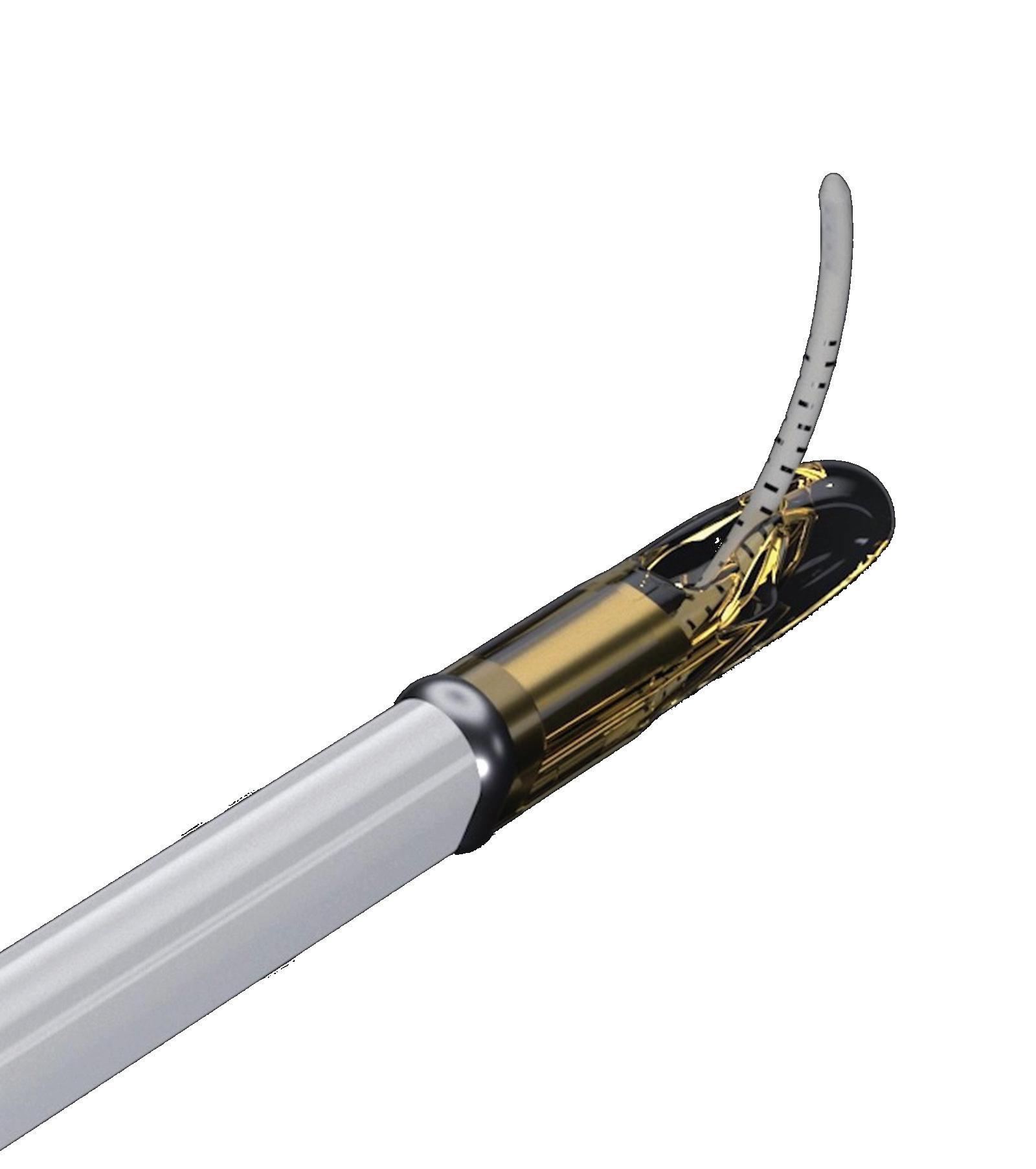
Alcon’s Hydrus Microstent is about the size of an eyelash and is laser cut from nitinol tubing to create an open-window scaffold design.
Image courtesy of Alcon
Glaucoma tube shunt surgery offers another option for reducing IOP by implanting a drainage shunt to divert excess aqueous fluid from inside the eye to a space outside the eye where it can be absorbed into the bloodstream. Glaucoma tube shunts are typically made of silicone or polypropylene since these biocompatible materials won’t break down in the body and are flexible. Sometimes, surgical interventions lower IOP to dangerously low levels (hypotony), causing blurry vision and pain. But new devices focus on managing aqueous flow with titratable implants and an implant within the trabecular meshwork itself, minimizing the risk of complications such as post-surgical hypotony and reducing the need for reintervention as eye pressure fluctuates.
Ivantis, for $475 million in 2022.)
Nitinol was chosen for its flexibility, biocompatibility and durability to support the tissue in the Schlemm’s canal. The implant is laser cut from nitinol tubing to create an open-window scaffold design with alternating “spines” for structural support and “windows” to provide outflow pathways.
The implant is heat-set to a curvature that matches the shape of the Schlemm’s canal, and is designed to occupy 90° of the canal to enhance aqueous outflow without puncturing conjunctival tissue like traditional therapies.
implant uses nitinol’s shape-memory properties, which are still underutilized in medtech, for glaucoma management.
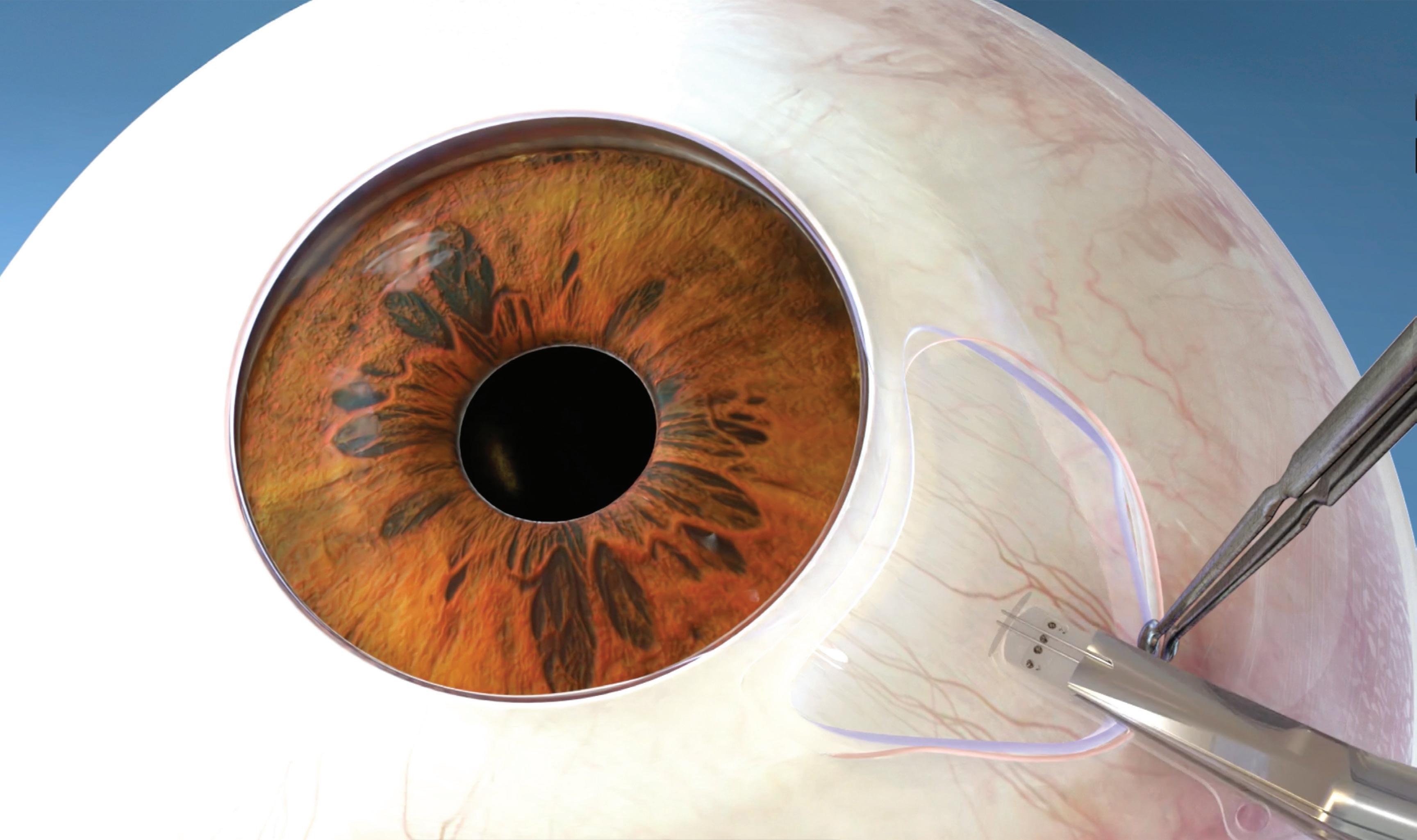
The investigational Calibreye system consists of three flow channels controlled by valves made of nitinol that can be repeatedly opened and closed using a slit-lamp green laser to provide reversible and customizable aqueous humor outflow control. The valves and its channels are encased in low-durometer silicone to conform to the patient’s eye shape and minimize the risk of erosion.
The nitinol valve assembly controls the fluid flow through the two outer channels, triggering changes
Alcon’s Hydrus Microstent
Alcon sells a nitinol implant to treat glaucoma in conjunction with cataract surgery. FDA-approved in 2018, the Hydrus Microstent is an 8 mm implant that bypasses the trabecular meshwork and dilates the Schlemm’s canal to decrease intraocular pressure. (Alcon bought the device and its developer,
Myra Vision’s Calibreye TGT Surgical System
Myra Vision — a Shifamed portfolio company — developed its Calibreye shunt to address fluctuating eye pressure with valve-controlled channels made of nitinol that allow physicians to titrate aqueous outflow resistance based on the patient’s needs. This novel
The Calibreye system’s nitinol valve assembly controls the fluid flow through the two outer channels.
Illustration courtesy of Myra Vision
in outflow resistance ranging from baseline to maximum. Nitinol was chosen because of its shape memory properties and superelasticity, allowing the valves to be repeatedly actuated with light energy from the green laser and change outflow resistance as needed in an outpatient setting.
The Calibreye device was welltolerated after six months of safety testing in rabbits. Myra Vision treated its first clinical trial patient in January 2026.
The future of nitinol in glaucoma therapies
Nitinol’s superelasticity is fueling researchers’ studies on new minimally invasive glaucoma treatments.
In August 2025, University of Oxford researchers published a study on a novel deployable microstent made out of nitinol that can shrink to less than a quarter of a millimeter in size with a diameter that fits within the needle of a hypodermic syringe for insertion, taking minimally invasive procedures to the next level. Once implanted, the stent expands, tripling its original size, to increase aqueous flow.
Initial studies in rabbits to evaluate safety and performance found a greater reduction in eye pressure when compared to standard tube stents, a promising sign for the future of glaucoma therapies.



Download our free special report featuring pulsed-field ablation advice and tips from OEM device experts at wtwh.me/nitinolpfa


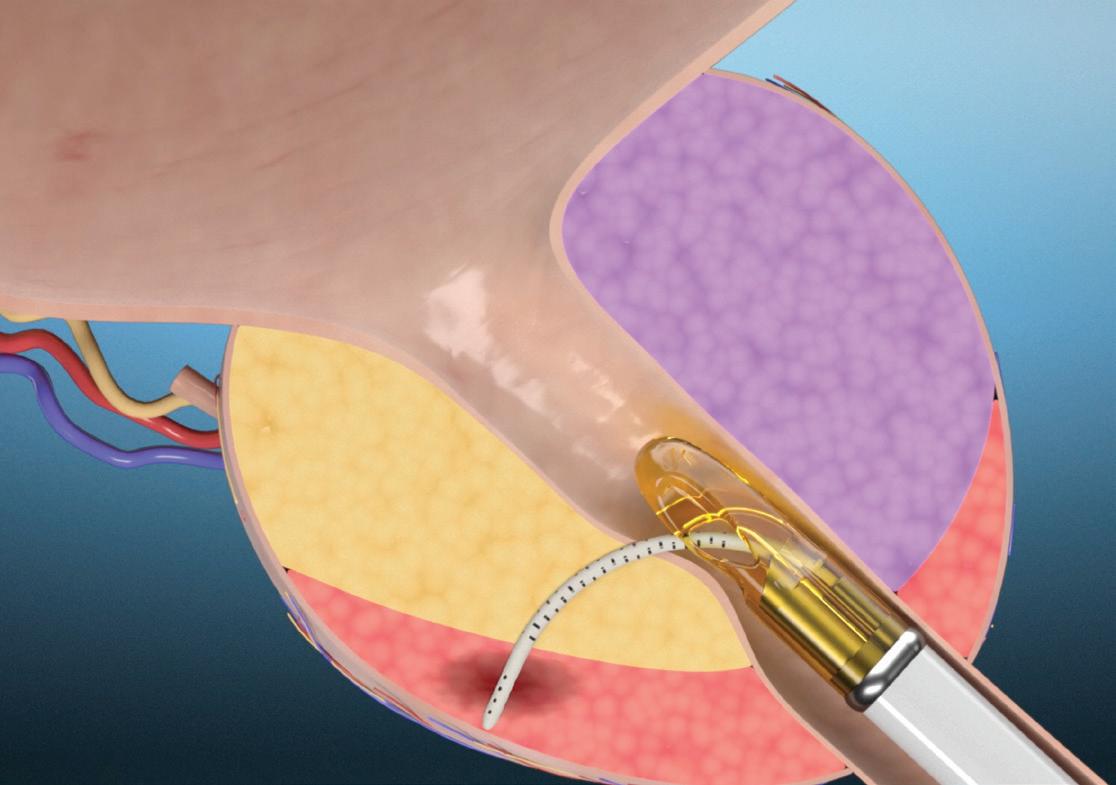
FThe Francis Medical Vanquish Water Vapor Ablation System is FDA-cleared for thermal ablation of targeted prostate tissue via a transurethral approach. Image courtesy of Francis Medical

Francis Medical designed its Vanquish Water Vapor Ablation System with electromagnetic guidance and hands-free stabilization.
rancis Medical recently won FDA 510(k) clearance for its Vanquish Water Vapor Ablation System, a new device for minimally invasive prostate ablation.
The Vanquish system uses water vapor for focal thermal ablation of cancerous tissue anywhere in the prostate. It includes a stabilizer system; electromagnetic needle guidance system; generator, cart and monitor; transrectal ultrasound cradle; and vapor delivery device. The system also has a user interface that displays real-time ultrasound and cystoscopy along with MRI images.
The transurethral ablation device uses transrectal ultrasound guidance to deliver sterile water vapor to the targeted prostate tissue, according to the company’s 510(k) summary.
A health provider inserts the Vanquish device into the patient’s urethra and advances it to the prostate. A transurethral needle with small holes at its tip deploys from the device and moves to the targeted cancerous tissue, applying hot water vapor to destroy tissue within the target location.
“Interstitial fluid-filled spaces surround prostate cells,” the company explains at its website. “During multiple treatments, vapor exits the catheter at a higher pressure than the extracellular environment, moving convectively through prostate tissue, displacing interstitial fluid, and reaching cell membranes. Rapid phase transformation of water vapor back to its liquid state (condensation) releases the stored latent heat of
vaporization directly onto the affected cell membranes, denaturing them and causing cell death.”
That vapor energy can also be applied to marginal areas around the cancer to ensure effective treatment. Water vapor remains within the zone it is released, making it different from other ablative procedures used for prostate cancer.
“When vapor is released within one of the prostate zones, it tends to ‘respect’ the boundaries of the prostate capsule and pseudo capsule by staying within the zone it was released,” the company says.
Containing the vapor’s energy to the prostate reduces the risk of common complications like urinary incontinence and erectile dysfunction that are more common in other prostate cancer treatments, Francis Medical said.
The Vanquish system’s 510(k) predicate is Boston Scientific’s Rezūm System for minimally invasive transurethral water vapor ablation of prostate tissue. Francis Medical says both systems have the same intended use and delivery mechanism.
But the Vanquish system has a couple of distinguishing features. Francis Medical says the systems have “slightly” different ablation energy levels, and that the Rezūm system lacks the Vanquish system’s hands-free stabilizer system and needle guidance system.
The stabilizer system is designed to facilitate freedom of user movement, while the needle guidance system works with the transrectal ultrasound to target prostate lesions.
Francis Medical said its Vapor 2 pivotal study is showing promising results. The study completed enrollment at 235 patients with intermediate-risk localized prostate cancer in February 2025.
The company included 12-month follow-up data on the first 110 patients in its FDA submission, reporting no serious device-related adverse events or unanticipated adverse device effects.
Nearly three fourths of those patients showed negative biopsy at six months, the company said, while the average prostate volume reduction at that time for all subjects (as measured by MRI) was 21%. One year after the treatment, the PSA reduction for 108 of the subjects was 52%. The company also reported low rates of urinary incontinence and erectile dysfunction.
Patients undergo general anesthesia for the outpatient procedure and can expect the entire appointment to take about a half day. One of the system’s benefits, the company says, is that it offers “flexibility for future procedures should it become necessary” for a patient.
“FDA clearance allows us to offer an alternative to patients with localized prostate cancer that better balances oncologic and quality of life risks,” Francis Medical President and CEO Mike Kujak said when announcing the FDA clearance.
The privately held medical device company said it plans to continue collecting data to understand longerterm outcomes in patients and to pursue premarket approval (PMA) for an expanded indication of the Vanquish System.
Harness our leading-edge vacuum technology . . . because lives depend on it.


Your high-value medical parts need special treatment. Solar’s leading-edge vacuum heat treating technology produces clean, bright, consistent results. From annealing to age hardening, rest assured knowing your life-critical parts were vacuum heat treated to your exact specs.
For your prosthetics, guide wires, stents, surgical tools, device and battery cases, hypodermics and hypodermic tubing, brazements for analytical devices...and more, trust Solar Atmospheres to provide you with uncompromising quality.


How to optimize biomedical textile design for heart valves
Custom
textiles can provide complex geometries, porosity, tensile strength and other critical characteristics for next-gen heart valve systems.


Biomedical textiles have been incorporated in vascular medical devices for more than 50 years. Thanks to their inherent compliance and flexibility, textiles can be custom engineered to achieve desired pore sizes and other specifications that inform performance and functionality, as well as be tailored to dimensions suited for devices delivered via catheter.
Implantable textiles have become more innovative, including complex and fully customizable geometries that support and promote healing and regeneration of damaged cardiovascular tissue. In heart valves, biomedical textiles may be used as components for skirts, ring covers, or even cables for delivery systems.
The evolution of biomedical textile manufacturing for heart valve fabrics Historically, textile machines were rudimentary, using oil and chain-links.
Their geared configuration significantly limited pattern and design capabilities.
Today’s textile machines can be fully electronic, which is much cleaner and allows for more predictable output of consistent and repeatable patterning for highly customizable characteristics.
Modern equipment coupled with engineering expertise can blend different raw materials together for further customization. Laser cutting offers precise dimensions and unique shapes, important for fitting the fabric over frames with complex geometries.
Knitted heart valve fabric structures are formed by interlocking loops of yarn in a weft or warp pattern to form flat, broad, or tubular structures with open pores to promote native tissue growth as needed. Warp knitting is particularly ideal for creating textile products for vascular applications like mitral heart valve replacement because it can produce low profile, dense textile structures that prevent blood leakage around the valve.


Porosity can be tailored to recruit the desired cell by size, creating specialized regions of tissue regeneration. Densities can be changed quickly so you can transition from dense to porous within a single fabric. For heart valve fabrics, this means blood leakage can be prevented inside the valve, while native tissue ingrowth is encouraged outside. Applications of knitted heart valve fabrics include vascular prosthesis, hemostasis, cardiac support devices, and valve sewing cuffs.
Woven fabric configurations include tubular, flat, tapered, fabrics that are beneficial due to their low porosity, dimensional stability and high-tensile strength.
One of the first things to consider when engineering a biomedical textile for use in a heart valve is what raw material to select.
Polyester (PET) has been used in heart valves since the first surgically implanted valve was developed in the 1960s. PET is still commonly used because the polymer is well researched, biocompatible, and results in a positive tissue response.
However, ultra-high molecular weight polyethylene (UHMWPE) may be a better option for applications that require high strength for a cardiovascular device designed for deployment and retrieval.
Polyester and polyethylene can both be braided, woven, or knitted. For heart valves, wovens are ideal for providing structure, while knits are well-suited for encouraging tissue growth. Components that incorporate both knitted and woven textiles mated together can be used on the same heart valve to achieve the desired result.
It is important that blood be able to flow through a valve, but surgeons generally want a seal around the edges of the valve to prevent paravalvular leakage. PET is known to have predictable coagulation cascade and can be placed around the circumference of the valve.
When a valve is inserted in the body, it is usually compressed and requires a force to allow it to spring open in situ. Shape memory materials such as nitinol are increasingly used for heart valves to maintain their shape and structure after catheter delivery and implantation in the body.
When a heart valve manufacturer is working with a textile partner, it is important to define the mechanical specifications early in the process to prioritize performance. Due to many modern heart valves being delivered via catheter, dimensions are critical for the design process.
Understanding the purpose of the finished product — such as providing structure, promoting tissue ingrowth, or preventing leakage — helps inform critical design decisions.
For example, if the overall goal of a device is to prevent paravalvular leakage, a knit with a high surface area would be desirable. Braids, on the other hand, provide versatility, but it is important to understand the end use and application, such as whether the braid is going to be sewn on the heart valve or used as a deployment mechanism in the catheter.
Porosity and tensile strength are also vital to performance. Porosity is an important consideration for textiles that support tissue ingrowth. Textiles can be tailored to range in porosity from watertight to open to promote cell ingrowth. Tensile strength is another critical design requirement for valves. Valves experience immense pressure as the heart pumps, so it is essential to engineer them to prevent bursting or deformation under force.
Customizable textiles enable thinner, lower profile and better performing devices, allowing surgeons to transition many open-heart surgeries to procedures where a device can be delivered via catheter instead.
By working in close collaboration with their textile partners and embracing innovation, heart valve manufacturers can continue to move the industry toward less invasive approaches that often result in faster recoveries and better patient outcomes.
Michelle Lishner is a new product design engineer at Cortland Biomedical. Lishner has experience in industry and academia in tissue engineering and polymer-based medical devices with awards in leadership, project management, innovation, and product design.


Stereotaxis designed the MAGiC cardiac ablation catheter for consistent and safe tissue
“Our mission is to advance robotics in endovascular interventions,” Stereotaxis CEO and Chair David Fischel says.
tereotaxis designed its robotically navigated MAGiC cardiac ablation catheter with a few features to maintain consistent heart tissue contact while mitigating safety risks of the minimally invasive procedure.
The FDA approved the MAGiC radiofrequency ablation catheter in January 2026. MAGiC is short for “magnetic interventional ablation catheter,” an abbreviation Stereotaxis also uses for its FDA-cleared MAGiC Sweep robotically navigated high-density electrophysiology (EP) mapping catheter

The MAGiC cardiac ablation catheter uses robotics and six magnets along its 8.5-Fr shaft for precise and stable navigation, winning FDA approval to treat supraventricular tachycardia (SVT) in congenital heart disease patients who can’t undergo cardiac ablation with conventional manual catheters.
“Our mission is to advance robotics in endovascular interventions,” Stereotaxis CEO and Chair David Fischel said.
In a Medical Design & Outsourcing interview ahead of the latest regulatory milestone, Fischel discussed the MAGiC catheter’s design and features and how it fits into the broader
Stereotaxis portfolio of robotic magnetic navigation technology.
For the thousands of children born with congenital heart disease every year in the United States, their odds of survival into adulthood are much better than decades ago thanks to advances in cardiac surgery. But the vast majority of those patients will have arrhythmias as they grow older due to their anatomical anomalies and all the surgeries they had as children.
“When you have hearts that are not typical and you have vascular flow that is not typical, a traditional catheter isn’t really built for that,” Fischel said. “A robotically steered catheter, by contrast, is steered directly from the distal tip and able to reach areas with precision and stability and safety. … With a robotically steered catheter, you can do pretzels — to some extent — around obstructions and reach where you need to get into the heart without having to puncture functional, useful implants.
“Our technology was designed specifically to overcome the natural limitations of manual pull-wire catheters,” he later continued. “Those manual limitations are that you’re trying to

perform surgery at the tip of the catheter, but you only have control a meter away at the proximal handle. By using robotically actuated magnetic fields, we can steer a very soft, gentle catheter directly from the distal tip, and that allows you to get places and to hold the tip with with a stability and with a precision that is otherwise impossible.”
The Stereotaxis MAGiC catheter’s features for ablation
Getting to the target is only the first part of cardiac ablation. The MAGiC catheter has a rounded, gold tip for consistent heart tissue contact during ablation. That contact is “necessary and critical” for both radiofrequency ablation and pulsed-field ablation (PFA) inside the heart, Fischel said.
“What you have with manual catheters is this challenge where you’re trying to create contact between the catheter and the tissue in a beating heart,” Fischel said. “If you think about placing a rigid device on the beating heart, you’re going to have some denting in the tissue, you’re going to have forces between the catheter and the tissue that is volatile based on where you are in the heartbeat cycle.
“If you want to create consistent contact, you have to push a little bit into the tissue,” he later continued. “That pushing introduces the risk that you might have too high of a force at certain points in the heart cycle, and that creates the risk of a perforation.”
The rate of perforation is around 1% even in the most modern manual catheters, Fischel said, but that’s still significant for a major adverse event. To minimize that risk, the Stereotaxis MAGiC catheter has a soft, gentle shaft and is steered directly from the tip.
“When you steer the tip into the tissue, as you start to advance the catheter forward into the tissue, the back shaft buckles at forces typically around 1020 grams, which is far below what what is typically needed to perforate the heart,” Fischel said. “That’s an intrinsic design characteristic of robotically steered catheters generally and the MAGiC catheter specifically which helps avoid the risk of perforation while ensuring consistent contact with the tissue.”
Fischel declined to name the specific materials in the MAGiC catheter, but said they’re the same as in traditional catheters.
“Other than that, we take out a pull wire and introduce solid magnetic material in the distal end of the catheter and then we use softer durometer plastics and different coils and braidings,” he said.
The MAGiC catheter also has a helical irrigation fluid channel designed for efficient and uniform cooling of the catheter tip to avoid char and coagulation.
“Historically, physicians needed to deliver something like 30 mL per minute of saline to the tip to keep the tip cool, and we reduced that by 66% to 10 mL a minute,” Fischel said.
“Many cardiac patients also have renal disorders, and the more fluid you deliver to a patient with renal disease, the more you can have fluid overload and other renal complications,” he continued. “Being able to deliver lesions and to treat these patients without that risk is one of the nice improvements of the MAGiC catheter.”




Gore Medical’s Tag thoracic branch endoprosthesis (TBE) uses nitinol and ePTFE. The late Bob Gore invented ePTFE in 1969.
you’re looking for a medtech material enabling innovation in the same way that nitinol has, you don’t need to look far to find expanded polytetrafluoroethylene (ePTFE).
Implants like stents, stent grafts and replacement heart valves often use nitinol — a uniquely useful nickel-titanium alloy — wrapped or covered with ePTFE. These two materials are increasingly being paired together for minimally invasive medical devices and systems.
Based on the synthetic fluoropolymer PTFE, ePTFE is stretched to create a strong, durable, microporous membrane with a low coefficient of friction. EPTFE is chemically inert and biocompatible. “We have never had a confirmed allergic response to ePTFE,” says Gore Medical, which invented the material. “PTFE is the most inert substance known and is very biocompatible.”
BY JIM HAMMERAND MANAGING EDITOR
While PTFE and ePTFE are perand polyfluoroalkyl substances (PFAS, more commonly known as “forever chemicals”), the FDA has said it sees no reason to restrict their use in medical devices.
EPTFE can be customized for variable porosity, uniaxial or biaxial stretching, and thickness. EPTFE can be thinner than a single human hair and layered for more thickness. EPTFE can also be engineered to be hydrophobic to repel water or hydrophilic for filtering.
This material can be sterilized with methods such as ethylene oxide (EO or EtO), steam autoclaving and vaporized hydrogen peroxide (VHP).
In transcatheter heart valves like the Medtronic Evolut family, ePTFE helps minimize paravalvular leaks (PVL). The Edwards Lifesciences Sapien M3 mitral valve replacement system uses ePTFE for the nitinol dock that anchors the valve.

“WE HAVE NEVER HAD A CONFIRMED ALLERGIC RESPONSE TO EPTFE. PTFE IS THE MOST INERT SUBSTANCE KNOWN AND IS VERY BIOCOMPATIBLE.”

The unique material properties of ePTFE “allow us to achieve some of the characteristics we were hoping to achieve on the dock, such as the slippery nature of the PTFE resin, but also the padded compatibility of the porous nature,” Edwards Lifesciences VP of Engineering Darshin Patel, who led the Sapien M3’s development, said in a Medical Design & Outsourcing interview.
“We try to respect the anatomy and make sure that when we anchor our devices, we’re not putting a bunch of point loads on our devices that could interact with the anatomy and cause tears,” he continued. “We want to spread that load as much as possible. One of the things that we utilized was ePTFE in different porosities. Depending on what we’re trying to achieve, if you have a very small porosity, you’re going to have a little bit more rigid of an ePTFE, versus if you have larger porosity it’s more compressible and it affects its relationship to ingrowth.”


Adona Medical’s Delphi adjustable heart shunt implant (shown here with a quarter for scale) is made of ePTFEcoated nitinol and has pressure sensors on both sides of the heart.
Photo courtesy of Adona Medical
Adona Medical’s Delphi adjustable heart shunt implant is another novel cardiac device combining ePTFE with nitinol (in this case, tapping nitinol’s shape memory properties, which is less commonly used than the alloy’s superelasticity). Delphi’s ePTFE coating is meant to encourage complete endothelialization within months of implantation to reduce the likelihood of thromboembolic events.
Another new system combining nitinol and ePTFE is Morphic Medical’s endoscopically delivered Reset device, a CE-marked, nonsurgical implant designed to be anchored with nitinol >>
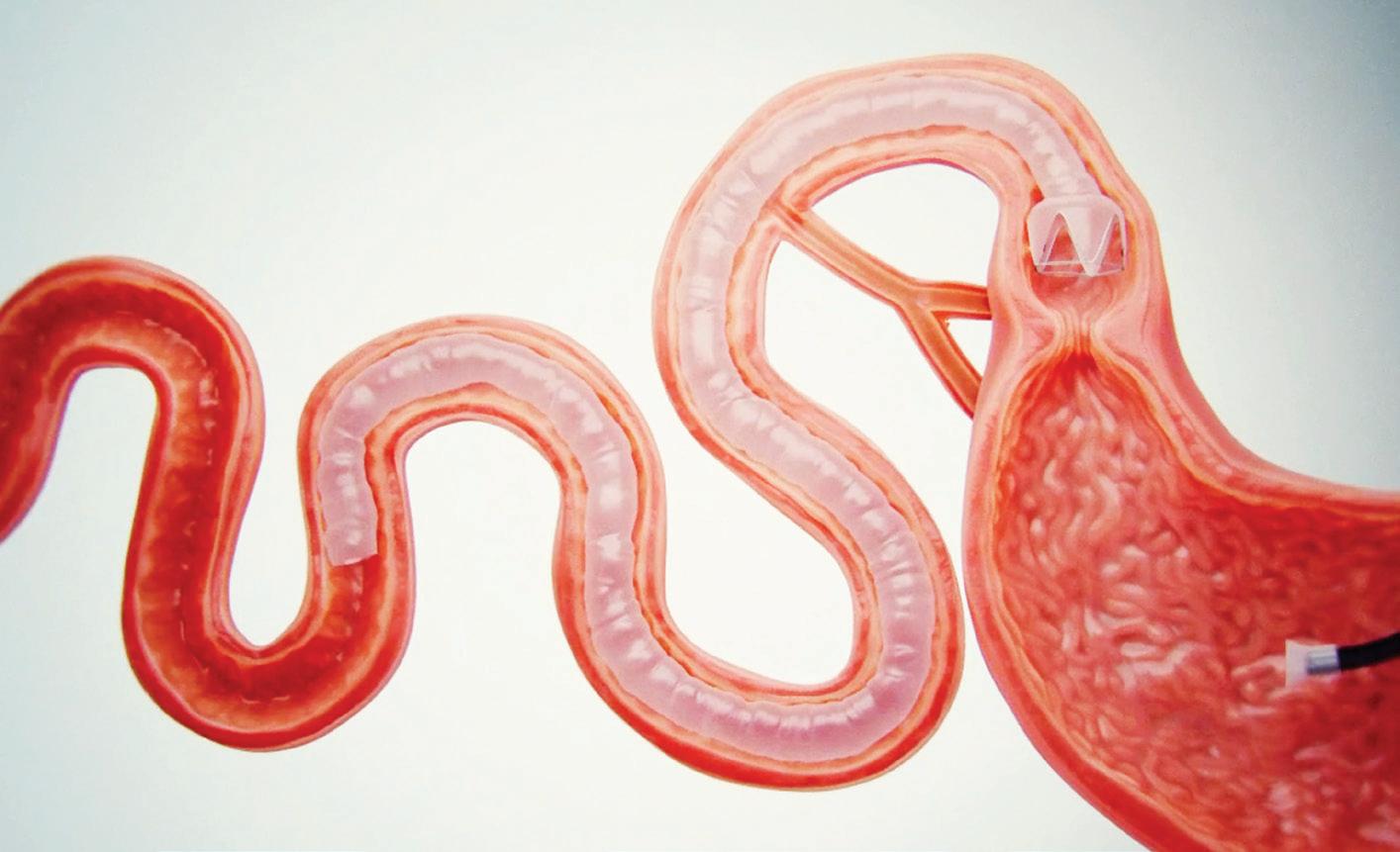
in a type 2 diabetes patient’s duodenum for up to a year.
“Once in place, Reset conforms to the shape and movement of the intestine and may begin to work immediately by creating a physical barrier between receptors in the intestinal wall and food that has been shown to directly affect key hormone levels,” resulting in lower blood sugar levels, weight loss and lower cardiovascular risk, the company says.
EPTFE can also be used without nitinol for medical applications such as pericardial and vascular patches, heart leaflets and heart strings, meshes, negative pressure wound therapy, venting, filtration and diagnostics.
Intravenous (IV) filters can use ePTFE to evacuate gas bubbles while containing fluids and maintaining sterility, Gore Medical says. “It consistently delivers high performance and enables a stable integration process for high volume production, unlike other materials.”
Much like with nitinol, device developers are still learning how to make the most of ePTFE’s properties. Contract development and manufacturing organizations offer ePTFE capabilities to their device OEM customers.
Working with such a thin, light material can be challenging. It’s prone to static cling, and the slightest air movement can cause the material to wave or ripple even inside the walls of a
controlled clean room. On one medical device facility tour, Medical Design & Outsourcing saw cleanroom employees shielding their workspaces with physical barriers to block breezes from others walking past.
Gore Medical warns that ePTFE grafts must be handled with care to prevent mechanical damage or graft disruption.
Boston Scientific used ePTFE when it developed its Reliance defibrillation leads to prevent tissue ingrowth, but later learned that cell debris, proteins and minerals could pass through the membrane and cause dystrophic calcification that increased low-voltage shock impedance (LVSI) and reduced shock efficacy.
The late Bob Gore invented ePTFE in 1969, a decade after moving to Minnesota to earn his master’s degree and doctorate in chemical engineering from the University of Minnesota. In an experiment, he rapidly stretched heated rods of PTFE and created the microporous material that would later be launched as Gore-Tex by his family’s business, W. L. Gore & Associates.
“One night in October 1969, Bob was researching a new process for stretching extruded PTFE into pipethread tape when he discovered that the polymer could be ‘expanded,’” the story goes, according to W. L. Gore & Associates.
Morphic Medical’s Reset implant uses ePTFE for the sleeve and nitinol to anchor the device in the small intestine.
Illustration courtesy of Morphic Medical
“The discovery followed a series of unsuccessful experiments in which he was attempting to stretch heated rods of PTFE by about 10 percent,” the company continued. “As it turned out, the right conditions for stretching PTFE were counterintuitive. Instead of slowly stretching the heated material, he applied a sudden, accelerating yank that unexpectedly caused it to expand to nearly 1000 percent. This resulted in the transformation of solid PTFE into a microporous structure that was mostly air.” Gore Medical launched in 1975 with the first U.S. sale of its Gore-Tex vascular graft. The next year, Bob Gore succeeded his father as CEO and president of W. L. Gore & Associates.



“IT’S GETTING WORSE, NOT BETTER,” THE STANFORD BIODESIGN CO-FOUNDER AND DIRECTOR SAID IN AN INTERVIEW.
BY KAYLEEN BROWN DEVICETALKS MANAGING EDITOR
Reimbursement now represents the largest source of downstream risk. For emerging medtechs without clear coding or coverage pathways, delays can persist long after regulatory clearance, with Stanford Biodesign’s Josh Makower cautioning that without a payment framework in place, adoption may be slow and uncertain.
“It’s going to be a while,” he said.
During a keynote interview at our DeviceTalks West 2025 conference, Makower expressed concern that the
current reimbursement landscape is “throttling” medtech innovation.
Makower, who is the Yock Family Professor of Medicine and Bioengineering at Stanford University School of Medicine and School of Engineering, as well as director and co-founder of the Stanford Mussallem Center for Biodesign, also offered his perspective on FDA and regulatory predictability, as well as on how the current investment climate is shaping the path from approval to adoption for medical technologies. >>
Reimbursement, coding, and coverage realities
The problem is not theoretical. Data generated by Makower’s team at Stanford, led by Sandy Ruggles, showed that the average time for breakthrough medical technology to move from FDA clearance or approval to even nominal coverage in the U.S. is 5.7 years.
“That is too long. That is a call to action. We need to fix it,” Makower said.
Even devices that clear the FDA with strong clinical evidence may not become commercially available before venture capital funding runs out.
“VCs have, in most of their arrangements, a 10-year life … and if it really is a 5.7-year average, which we believe it is, it could break the venture model.”
One of the biggest contributors to that delay is the lack of clear coverage standards.
“There is no definition for widespread use. I mean, literally, no definition. It’s in the eye of the beholder,” said Makower.
That ambiguity is a problem and can cause payors to hesitate to cover new technologies because they’re not widely used, creating adoption issues for providers due to uncertain coverage.
“If everybody takes that position, nothing new is ever going to get covered,” he said.
Still, there are some encouraging signs.
“Historically, if you went for a Category I CPT [current procedural terminology] code and … you lost that battle, they’d push you, whether you liked it or not, into Category III. And that could be a place where you would live for five years before you could get out of it.”
Recent changes at the American Medical Association have reduced that risk.
“They have now taken that out. … If you don’t want to [go Category III], you will not be forced to take it, which is a huge win,” said Makower.
Makower also noted, “One of the first wins we got was to allow innovators to use foreign data in support of their CPT I application.”
This shift could reduce duplicate trials and help with capital requirements, especially for medtechs running early feasibility studies outside of the U.S.
On CPT codes, Makower left us with this thought: “For all CPT codes, over time, the payment goes down. Many innovators … don’t adopt a new code for a new technology because they’re afraid they’re going to lose the code and lose payment on the existing technologies. Our data is showing you’re going to lose those payments anyway over time.
“So if you really want to protect your specialty, embrace innovation and
Verify compliance with industrial certifications Pinpoint the ideal material for your application






get new codes that bring new, higher payments, and you’ll be able to preserve the overall income for the physicians in your specialty rather than the reverse. … That encourages innovation and, as a result, brings value and dollars.”
When asked if artificial intelligence could help, Makower said yes.
“There [are] a lot of qualifiers … but the truth is that reviewers have to digest a ton of information, and AI is pretty good about digesting a ton of information. If it’s trained on a solid basis and has reviewer oversight — or some knowledgeable person’s oversight — then it could really facilitate and move things along.
“[VENTURE CAPITALISTS] HAVE, IN MOST OF THEIR ARRANGEMENTS, A 10YEAR LIFE … AND IF IT REALLY IS A 5.7YEAR AVERAGE, WHICH WE BELIEVE IT IS, IT COULD BREAK THE VENTURE MODEL.”
It’s not all bad news on the FDA and regulatory front. The agency is under pressure but still mission-driven, he said. The FDA is structurally intact, and its leadership remains under Center for Devices and Radiological Health Director Dr. Michelle Tarver, in the face of a volatile policy environment.
“Thus far, medtech has been very modestly impacted,” said Makower. “Michelle Tarver’s doing a great job, and her team is pretty much intact. On balance, the majority of people are getting a great service from the FDA. They are doing their jobs well, and meeting their timelines per MDUFA,” referring to the Medical Device User Fee Amendments agreements for FDA funding from device submissions.
But regulatory success is not just about timelines, it’s about predictability. And for device makers, predictability matters more than speed. Even when timelines are met, Makower said, changes in expectations mid-process can be destabilizing for emerging medtechs. He also shared concern with how applications are being reviewed.
“It has to do with consistency reviewer-to-reviewer, and developing an overall quality approach towards the way that they manage reviews,” he said. “It doesn’t matter who you get. They will always give you the same level of service.”
“It could also be bad because if it winds up being less people and less oversight, and you’re dealing with a robot telling you your application’s not approved or whatever, who do you appeal to? Those things need to be worked out,” he continued. “But the use of technology to create greater efficiencies would be potentially a good outcome.”
Reflecting on the current state of the FDA, Makower reminded us, “They’re not the enemy. They’re trying to protect citizens and patients and if we can align on some reasonable and predictable processes around whatever application, that’s the goal.”
Though the investment climate goes through cycles, one truth never wavers: identifying unmet clinical and system needs is a must. This is the only durable foundation upon which to build a successful medical technology.
design with reimbursement and costefficiencies in mind from day one. After all, medtech is still a business.
“’Let’s get a better clinical outcome at any cost’ doesn’t play anymore,” said Makower. “We need to think about the cost-effectiveness as we conceive of the problems that we’re trying to solve.”
Capital still exists, but the investment climate is becoming more restrained.
“We are seeing a real lack of early- and mid-stage VCs these days. As these funds have grown in assets, they have moved to later-stage, and now you’re seeing total pile on, so it’s either feast or famine.”
As a result, emerging medical device companies are being forced to accept new funding models.
“A lot of people think that there’s going to be a lot more build-to-buys with bigger companies. I think that’s probably true,” said Makower. “Partnering with corporations to do those could be a nice early model, but it’s tough out there for founders and early-stage companies.”
Makower reinforced the real problem: Without earlier paths to revenue, fewer investors are willing to engage early.
“The real focus is, ‘How do we improve reimbursement?’ Because if we can shorten the time to getting these companies more accretively commercial, then we’ll enhance the market for venture capital early-stage. And people
“ ‘LET’S GET A BETTER CLINICAL OUTCOME AT ANY COST’ DOESN’T PLAY ANYMORE.”
“The No. 1 thing is you really need to make sure you’re solving the right problem and that you’re really tuned into the need,” said Makower. “That sounds simple, but there still is a lot of technology in search of application. … If you’re not solving the real problem, then you’re going to have challenges downstream.”
However, it isn’t just about seeking the best clinical outcomes. We must
will start going earlier because they can realize they can exit earlier.”
With these fundamental challenges facing medtech, Makower offered advice to innovators.
“You just have to be perseverant and believe in yourself, believe in your technology, believe in the clinical data and just use the clinical data as your shield,” he said. “If you have good clinical data, you will win.”

Petal Surgical co-founder and CEO Prash Chopra envisions acoustic liquefaction for incisionless surgery and much more.

BY TOM SALEMI DEVICETALKS EDITORIAL DIRECTOR
The promise of incisionless surgery continues to draw the capital and interest from some of the biggest names in medtech.
A few months after the $2.25 billion private-equity-fueled buyout of HistoSonics, Petal Surgical emerged as a new player with marquee name investors — including Dr. Fred Moll, the father of surgical robotics — and a grand vision to completely upend the health care industry.
Petal Surgical hasn’t shared many details about its approach. But in a Medical Design & Outsourcing interview, Petal Surgical co-founder and CEO Prash Chopra explains how a painful loss drove him and other high-powered medtech veterans to develop a way to “take away cuts from surgery so the next father, the next loved one will avoid the bad things that happen during surgery.”
The company is working with the same class of ultrasound energy used by HistoSonics and others. Petal Surgical is employing the expertise and experience of co-founders to expand the technology’s reach beyond ablation into a new class of surgery.
Like Chopra, who previously worked at Intuitive and Verb Surgical, the leadership team includes pivotal figures in surgical robotics and spine surgery:
• Co-founder Dr. Bowen Jiang is a neurosurgeon specializing in minimally invasive, robotic-assisted spine surgery who leads the Providence St. Jude Neuroscience Institute and a robotic spine center in California.
• Co-founder Dr. Nicholas Theodore, a Johns Hopkins neurosurgeon and robotics pioneer, co-founded spine surgery company Excelsius Surgical.
When we founded Petal, we did not know the how. We knew we didn’t want to just become another company with a piece of technology in hand, another hammer looking for a nail.”

• Moll, listed as a Petal Surgical investor, founded Intuitive and Auris Surgical and sits on the board of many surgical robotics companies.
• Co-founding advisor Rony Abovitz is a co-founder of Mako Surgical, which was purchased by Stryker for $1.65 billion in 2013.
The following has been lightly edited for clarity.
MDO: Can you tell us about Petal Surgical’s origins? Chopra: “I was frustrated. Every company I worked at was not fundamentally trying to help people like my father, who passed away when he was only 52. Petal came together out of personal frustration. I reached out to the three co-founders. All three said, ‘Let’s take away cuts from surgery so the next father, the next loved one will avoid the bad things that happen during surgery.’“ >>
How did your father pass away?
Chopra: “He passed away because of a surgical complication that turned into an infection. Within 72 hours his body went into sepsis.”
How does Petal approach this problem?
Chopra: “When we founded Petal, we did not know the how. We knew we didn’t want to just become another company with a piece of technology in hand, another hammer looking for a nail. With our combined experiences — two surgeons (Jiang and Theodore), Abovitz and myself — we spent six months examining what we could do. We weren’t just looking at tissue manipulation, but ways to fundamentally transform surgery by removing incision, excision and suturing. The answer came down in two buckets: radiation or ultrasound. Another nine months of exploration and experiments led us to focus on ultrasound and histotripsy. We then took two more years to refine our end-to-end approach, not just around the act of tissue intervention itself, but identifying everything that would enable this socioeconomically, workflow-wise, business-model-wise.”
How is Petal different from other ultrasound-based surgical systems in the market?
Chopra: “We use a refined form of histotripsy. We said we won’t be happy with just an ablative approach where we are helping a category of patients potentially for cancer treatments, palliative care. Instead, we’ve designed an end-toend solution which starts with our form of histotripsy but then integrates everything we know, from robotics and AI to image guidance to the biology of disease states. We want to create an incisionless outpatient surgical experience that can scale, so we also avoided using active, big machine guidance like CT or MRIs.”
How are you impacting tissue?
Chopra: “Broadly speaking, histotripsy is about harnessing naturally present gas bubbles and popping them to destroy tissue. We’re thinking about the level of precision and control surgeons need from histotripsy, so our distinctive approach focuses on predictable shapes and sizes for each treatment. Instead of just relying on bubbles to mechanically destroy tissue, we’re adding more control. The mode of action enables us to create one vapor filled cavity at
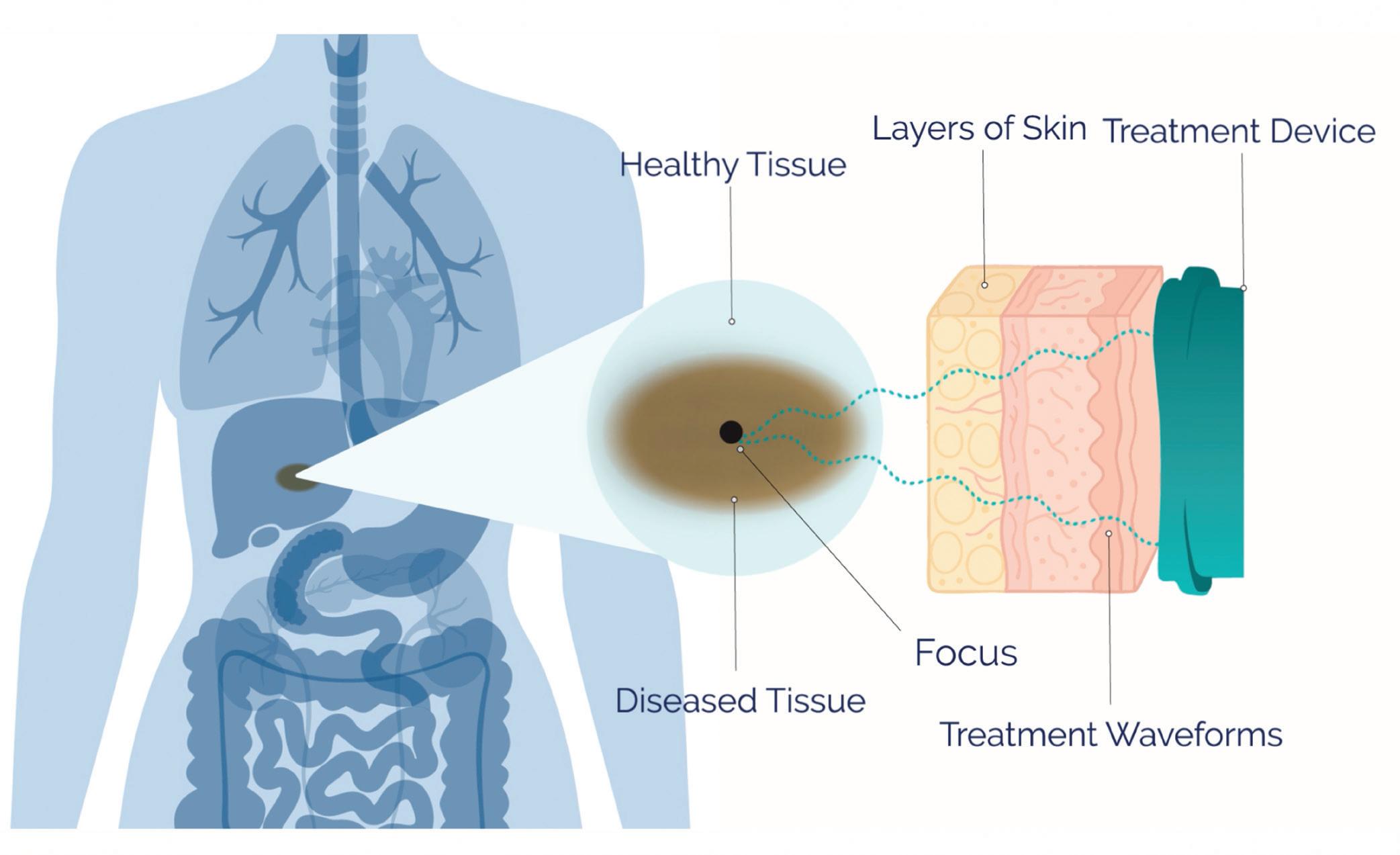
a time, the size of which is only limited by biology and physics. If you are a few centimeters beneath the skin surface of a patient, you’re talking rice grains or smaller. These vapor-filled cavities then become like micro universes for tiny hurricanes that we call jetting.”
What kind of imaging is used to guide the procedure?
Chopra: “We can use a multitude of imaging modalities, so it depends on the disease state and the organ system, but an example would be ultrasound. And then a couple other modalities mixed in as well, because we have to make sure what we are looking at is reliable with margins and predictable for surgeons to use.”
What disease states have you identified as your first targets for treatment?
Chopra: “We’re starting with cancer first, but we’re designing our platform so we aren’t bound to ablative therapies alone. These are only a few examples, but other potential areas for Petal’s platform include softening plaque in the arteries in the heart, dissolving blood clots, and affecting bulging discs for low back pain treatment.”
Where can this go?
Chopra: “What I personally get very excited about is the potential of immunomodulatory properties, broadly speaking, from histotripsy. As an example, imagine a day where we are not just relieving a cancer patient’s pain and debilitating conditions by doing palliative care alone. Imagine us being able to tell a cancer patient we have treated a third of their tumor and because we didn’t burn the tissue, the rest has disappeared over a period of weeks to months, that it disappeared because the body’s immune system got the help it needs. We can think about not just destruction, but subcellular modulation of things we may not even be able to see today, protein level, or denaturing certain cells or tagging them. So if we open a whole new suite, not just for destruction of tissue, I can’t even imagine where the world will go with that.”
What specialists or surgeons do you envision using Petal’s system?
Chopra: “We will learn that together, but it’s our current belief that the next
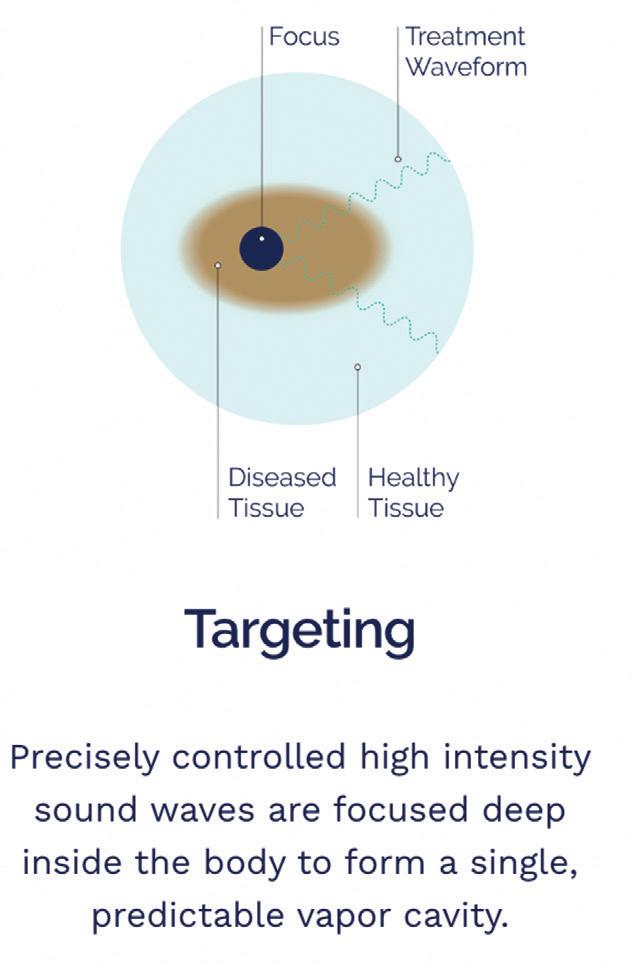
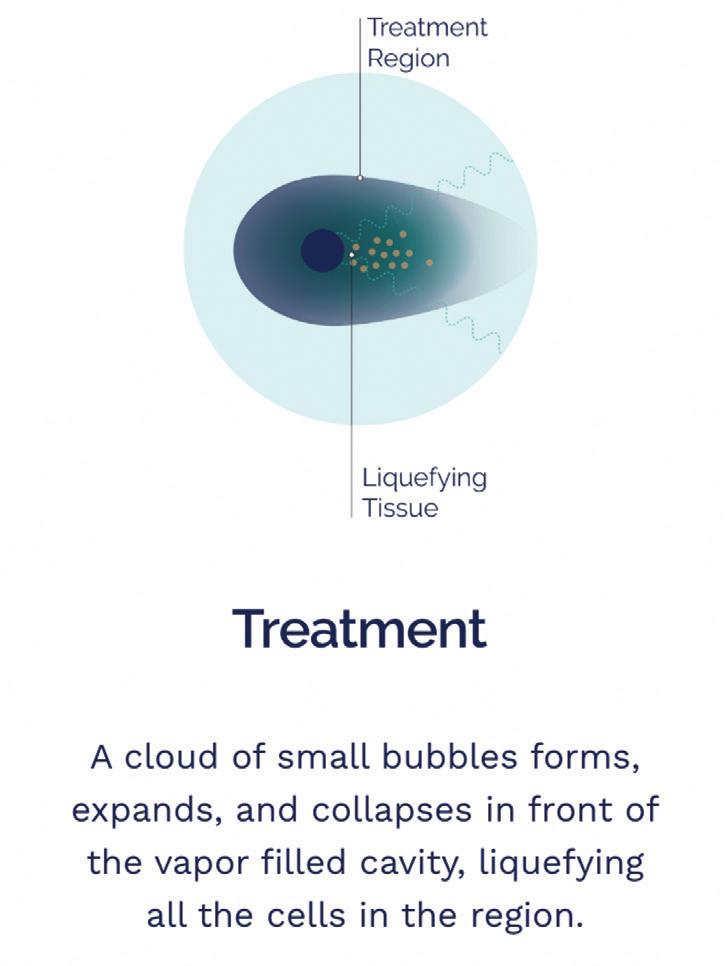

Petal Surgical explains acoustic liquefaction as “a new class of focused therapeutic ultrasound that delivers precise, controlled tissue manipulation without incisions, heat, or toxicity.”
Illustration courtesy of Petal Surgical
generation of tissue manipulation experts will be broken into two categories. The first category would be the traditional surgeons who might only be comfortable when they’re physically looking at tissue while manipulating it. But we believe that the second category will slowly become the primary users of our technology, interventional radiologists or people who are more comfortable with indirect image guidance in an outpatient setup. There is precedent. We’ve seen a portion of cardiothoracic surgeries become interventional cardiology procedures, for example. The transition took time, but the
primary setting for a technology like Petal, because the push is already there. We’re sitting at the juncture to make it happen.”
How are you going about building the company and raising capital?

Petal
Surgical co-founder Dr. Bowen Jiang
“We’re starting with cancer first, but we’re designing our platform so we aren’t bound to ablative therapies alone.”
summary answer is it’s going to be a new class of physicians who have been trained in deep surgery, but who are comfortable operating with a virtual tool, trusting the real-world evidence and outcomes as if it’s a surgical tool and not just a therapy.”
Would this be used in larger clinical hospitals or ambulatory surgery centers?
Chopra: “This is a subjective vision statement. We’ll have to do our work to prove it, right? I believe in 15-20 years ambulatory surgical centers will become the
Chopra: “We wanted to be very sure that the science we started to talk about is well understood by our team and repeatable in multiple clinical tissue targets. We want to have the big picture story settled and the strategic vision in place. We want to be able to talk about going from one to 100, not just zero to one. We are actively in talks with the next round to build Petal’s mission out in three steps. The first step is going to the first human trials to prove to ourselves, the societies and the market that we have a modality that works repeatedly. The second step will be commercialization of the platform with one example use case: Here’s what incisionless surgery means, here’s the value proposition and prove the fit. And the final one will be we start to think of Petal.Health, not just Petal Surgical.”
What does Petal.Health mean?
Chopra: “The last part means starting to harness the potential of acoustic liquefaction beyond just destroying tissue. So we could talk about completely noninvasive or incisionless diagnosis, biopsies and — this is where I get
tingles down my spine — curative care. So it starts with cancer, but we believe the world is moving toward needing a high-tech yet cost-effective healthcare model at a fast rate. Starting in the U.S., what we have today is not sustainable. I have a 15-year-old daughter. I want to leave her a world where health insurance is less critical because it’s going to be less expensive for her to take care of acute illnesses. We’ll start with surgery and then cover the gamut: diagnosis, preventive care, all the way up to curative care. You’re relieved of cancer, you’re free of cancer, you’re free of heart disease, you’re free of stroke, it’s not going to show up again. But it’s a vision, so we’ll see how far we go.”
Do you see this moving more quickly than perhaps a traditional medical device startup would?
Chopra: “We are not worrying about how long it will take. What we are concerned about is getting started the right way. So hopefully the rest of the industry will take note because all stakeholders’ interests will be aligned with a company like Petal when we go out there. We are prepared for a long haul, and that’s okay because that’s how big transformations take place. That said, we’re also focused on right now so we can prove that our vision is viable.”


Are tariffs causing you to reassess your medical power cord suppliers? You shouldn’t have to pay more for rising imports when you can get them right here in the United States. Interpower cords are durable and reliable, and they come with 1-week U.S. lead times. Our North American and international hospital-grade cord sets provide the correct amperages and voltages for medical devices such as portable CT scanners, X-ray machines, surgical robots, or the medical office.
North American and Japanese hospital-grade plugs and receptacles bear the green dot, signifying they have passed the rigorous UL 817 Abrupt Removal Test (UL 817, 18.2.4.1) for hospital-grade cords. All Interpower hospital-grade conductors inside the plug are anchored by a stainless steel ring that ensures electrical continuity when you need AC power in critical medical settings.
Interpower cords and components are manufactured in accordance with its product quality plan: hipot testing, continuity testing, and ground testing with multiple inspections.
• 1-week U.S. manufacturing lead times
• Same-day shipping on in-stock products
• No tariffs, no surprises




Ability Neurotech CEO Rotem Kopel
Electrodes sit on the cortex, on the surface of the brain. The implant is placed on the skull and the skin is closed over the skull, because the thin form factor is opened in the wearable docks by magnets that are kept in place. Then, the data transfer is 50 megabits per second (Mbps) via the laserbased system that we’ve developed over the past eight years.
“The key thing here is 5 mm thickness. Anything more than 5 mm, you need to host it externally or have a chest-mounted implant. We wanted to make sure we could keep it below 5 mm. Then, it could be placed on the skull without any need for bone work, then the skin can be closed over.”
MDO: What does Ability’s technology do?
Kopel: “We are recording data from the brain. Most of the translation and decoding application is done externally. In order to have the maximum capacity that we could have, to have the best decoding, to have the most flexibility on designing today’s application and tomorrow’s application, to understand the brain better to understand the disease better, the intention of Ability was to extract all the data we record. By doing that, we are recording raw data like everyone else that we are also extracting from the brain, from the implant, to the brain-computer interface.”

Ability Neurotech Chief Business Officer Craig Cook
Kopel: “On the topic of safety and patient simplicity, we chose not to have a battery in the implant. The battery has a coil and the implant is really being powered by the external device, inducing power to the implant. By [doing] that, we can supply more power to the implant to do everything we need. Also, sometimes you need to change your battery and on the pacemaker at the beginning, it was something that was recurring very fast. It’s a full-brain surgery. You can’t just replace the battery. It means you have to open the brain again, take out the electrodes and implant them back. … Our choice was to avoid that risk of replacing the battery.”

MDO: How does it achieve this?
Kopel: “You have many channels. Your information is the brain. We work at a very high pace in order to do what we do. We speak, we move, we have so much different sensing information that goes in. All of that is happening super fast. It’s not only that you have many channels, but the information is being conducted in the brain at a very high pace. To extract that out, you cannot use a common communication system like Bluetooth or RF. All that is not enough to extract our data. To maintain all those principles, we have developed an optical communication. We have a laser from the implant to the wearable and the principle is that when you’re shooting light via skin, it doesn’t show very well, but light is actually transferring very well over the skin. So when you have a laser within the implants and you’re modulating the information on top of a laser, an external device that actually captures it could send highspeed data and a lot of capacity over that link. Our technology is extracting the raw data of our brain recording. That allows us to not compromise. It allows us to have the full information of everything we record.”
MDO: Were there any significant challenges in the design process at Ability?
Kopel: “From the principle of understanding that we need to extract everything through how we designed the system, having that communication and supporting that with a brain implant, that is a very challenging task for a
medical device. It’s not a router that sits on the table and you could have as much power [as you’d like] and you don’t care for the heat. You have to think about power, you have to think about the temperature and you have to think about putting all that in less than a 5mm-thick device. That’s a complex task.
“What do we do different is we believe in raw data that allows us to have the full system, from recording to how we communicate out to how we define our decoding. … We don’t discriminate any information, and that’s unique. The device itself is set to have full safety for patients with the electrodes sitting on the surface of the brain. They’re not penetrating the brain.
When you are penetrating the brain with electrodes — and some companies are doing that — you’re risking a different reaction between the brain and those electrodes. Most of the time, you lose the quality of data. Over even one year, you dramatically lose the amount of data you have because of scarring around the tissue.
“The wearable sits on top of the brain. It’s 5 mm thick, so patients do not look funny. They don’t have something coming out, they don’t have wires running through the chest. The flexibility and the robustness of the device are there for the patient for a long time. As a concept, data and patient safety really guided us through all the design choices we made.”
MDO: What are the key differences between your technology and the competition?
Kopel: “Because each one of us took such a different approach to BCI by the way we designed the system, even though we target the same population it created a very different solution. In the end, we have chosen to use surface electrodes to get a very wide coverage of different areas. … For the patient, it’s less invasive. It will be stable for a long duration of time, because we know that the electrode could stay [working] for 10 years or more. When we look at longterm implantation, it ticks all the boxes.
“Second … we don’t transmit at 1 Mbps. We transmit at 50 Mbps. The real secret is what a 50 Mbps transmission allows us to transmit. The brain is super efficient. … You have to develop a very unique communication system that would run 50 Mbps and up in order to extract everything. With Ability at 50 Mbps, it allows us to transmit movement, sight, speech, everything. When we are looking at what we can offer from the electrodes, the network analysis that we could have the durability of the electrodes, the placement of the implant, and the communication system that extracts raw data. All that is a full package.”

There’s no place like medtech’s home

DPlease
join us in Minneapolis this May for DeviceTalks Minnesota 2026.
oes medtech have a hometown?
From a historical perspective, one might point to Edison’s fluoroscope in New Jersey in 1896, or the founding of Revra DePuy’s orthopedics company in Warsaw, Indiana, a year earlier.
Others might argue that the first truly modern medical device emerged in 1949, when Earl Bakken and Palmer Hermundslie built the first wearable pacemaker and founded Medtronic.

Abbott
EVP and Medical Devices
Group
President Lisa Earnhardt
HistoSonics
CEO and Chair
Mike Blue
Solventum
Chief Commercial Officer
Heather Knight
But the historical argument is inconclusive. Too many great things have happened in too many places. Perhaps the better approach is a less fact-driven one: What’s the definition of home?
The immediate answer may be cliché, but it resonates: “Home is where the heart is.”


If we apply that axiom — the source of which is undetermined — the answer is clear.
Minnesota is medtech’s home.
I’ve covered medtech for close to three decades, and very few days have passed without me thinking, writing or talking with someone from that state’s deep and diverse community of medical device companies and professionals.


That vision of Minnesota is what drew us to relaunch DeviceTalks Minnesota last year at the University of Minnesota’s McNamara Alumni Center for a truly remarkable day filled with light, energy and innovative insight. We
immediately committed to returning this year, and we will on May 4 at the same spectacular location.
I simply can’t wait to be back.
To be sure, I’m excited to sit down with keynotes, industry experts, and attendees to discuss the latest developments shaping our industry. We’ll be joined by many local and national leaders from Abbott, HistoSonics, Medtronic, Solventum and others.
But over the past few weeks, a different force has pulled my heart and mind to the region: a desire to stand with people who are defending their neighbors and communities.
Minnesota’s extraordinary courage and compassion against the federal government’s overreach has inspired all of us.
So while my professional eye is still drawn to the exemplary work being done by medical device companies across the region, my heart is drawn there through LinkedIn posts from medtech leaders and videos from Minnesota documenting previously unimaginable trauma.
If home is where the heart is, then to me, Minnesota is medtech’s home.
I can’t wait to be with you on May 4.
Go medtech,


The experts in design, development, and scalable manufacturing of interventional catheter-based devices and implants. Your trusted partner for bringing current and future medical devices to market
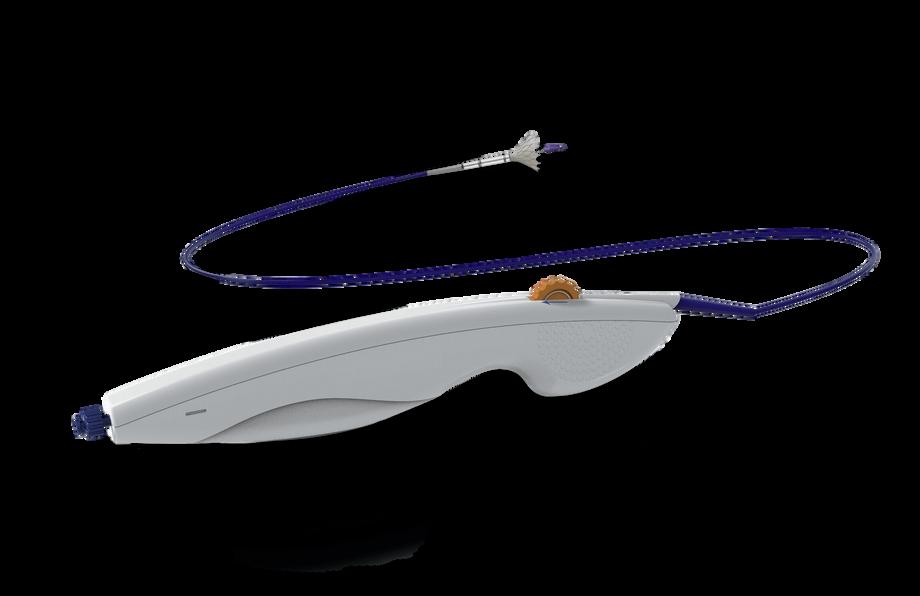



Learn more about our offerings at www.confluentmedical.com or email sales@confluentmedical.com for a custom quote.



