A SCIENTIST , A COOLER AND A

LIQUID HANDLING AND MACHINE LEARNING COMBO
LONG-HAUL FLIGHT BLOOD-CLOTTING DISORDER
IDENTIFIED







IDENTIFIED






6 A SCIENTIST, A COOLER AND A LONG-HAUL FLIGHT IN THE FIGHT AGAINST MND
Neuroscientist Dr Rachael Dunlop boarded a flight from Sydney to the US with a precious and unusual item of carry-on luggage.
14 LIQUID HANDLING WITH MACHINE LEARNING — THE PERFECT SCREENING COMBO?
A screening platform has been made public that promises a faster way to test drug combinations and doses.
16 PROTEOMIC APPROACH IDENTIFIES NEW BLOOD-CLOTTING DISORDER
25 PROJECT PEARL: SAFETY FINDINGS OF A LAB VENTILATION SYSTEMS COMPARISON
Could this independent comparison test of lab ventilation systems redefine standards for safety, efficiency and flexibility?
27 AI SPOTS HIDDEN OBJECTS IN CHEST SCANS
A new artificial intelligence tool can spot hard-to-see objects lodged in patients’ airways better than expert radiologists.
28 BEST PRACTICES FOR SAFE CENTRIFUGATION IN THE LABORATORY
Molecules produced by gut bacteria upon digestion of dietary fibre can improve the function of cancer-fighting immune cells. 14 28
Analysis of lethal antibodies has helped researchers identify a new blood-clotting disorder.
22 IMAGES REVEAL “INCREDIBLE” DETAIL OF INDUSTRIAL MICROCT SCANNER
Images of intricate biological structures and everyday items have revealed the capabilities of an industrial micro-CT scanner.
24 THE MICROBIOME HELPS TO FIGHT MELANOMA
The majority of all centrifuge accidents are understood to result from user error. These tips will help to ensure safe operation.
30 NANOPARTICLES SUPERCHARGE VINEGAR’S WOUNDHEALING POWER
Adding microscopic particles to vinegar can make them more effective against dangerous bacterial infections.
32 WHAT JOURNALISTS EXPECT FROM THE SCIENTISTS THEY SPEAK TO
Peer review is often treated as the end of the story, but for journalists it is usually the point where the story becomes possible.

Proteins perform a wide range of essential functions in the body and are vital for growth, development and the structure of every cell — but now scientists from the University of Surrey have revealed that proteins which play key roles in the development of diseases such as cancer and inflammation may also contribute to the risk of dying. Using data from the UK Biobank, the scientists analysed blood protein profiles from over 38,000 middle-aged and older adults, some of whom had suffered non-accidental death, within a 5- or 10-year period after blood samples were taken. The aim of the study was to determine if the risk for early non-accidental mortality is reflected by levels of circulating blood proteins.
The study discovered 392 proteins associated with an increased risk of death within a five-year timeframe and a further 377 proteins associated with dying within 10 years, even when adjusting for health and lifestyle factors, such as smoking or pre-existing disease diagnoses. Proteins responsible for cells communicating with each other, increasing the number of cells and processes of inflammation were most predictive of mortality within five years. For example, high levels of the protein SERPINA1, responsible for protecting the lungs against certain enzymes that cause
inflammation, was identified as being a strong predictor of five-year mortality.
It was also uncovered that five proteins were key indicators of mortality regardless of timeframe. This suggests these proteins play key roles in biological processes that result in conditions with poor outcomes. For example, elevated levels of SERPINA3 are associated with lower survival rates in several types of skin cancer and carcinomas. “Certain blood proteins reflect more gradual, chronic processes that affect longterm survival whilst others are reflective of more immediate, acute health risks, like troponin for heart disease,” said lead author Natalia Koziar, a PhD candidate at the University of Surrey. “The new information could be useful to clinicians in creating a scorecard of protein levels that help to identify individuals most at risk of developing conditions with poor outcomes.”
The researchers published their findings open access in PLOS One, so you can read more at doi. org/10.1371/journal.pone.0336845.
Proteomics is one of many topics explored in the February/March 2026 issue, which it gives me great pleasure to welcome you to as Interim Editor — looking after Lab+Life Scientist while Lauren Davis is on maternity leave. As my time so far at the Editor’s desk has shown me, there are a wonderful range of lab and life stories to report on, some fantastic technical insights from the experts to share with you, and a suite of new product
innovations, news and case studies to spotlight.
This time, some of our highlights include: an Australian neuroscientist’s adventure to Wyoming, cooler in hand, to develop a groundbreaking diagnostic blood test for motor neurone disease; a deep dive of a screening platform developed by St. Jude Children’s Research Hospital that, by combining acoustic liquid-handling protocols with machine learning, promises a faster way to test drug combinations and doses; and some interesting safety findings from Project PEARL (Performance for Efficient Airflow Regulation in Laboratories), which purports to be the world’s first to compare different lab ventilation and air distribution systems under controlled, real-world conditions.
I hope you enjoy the read.
Regards, Dr Joseph Brennan, Interim Editor LLS@wfmedia.com.au




Early in January 2026, neuroscientist Dr Rachael Dunlop boarded a flight from Sydney to the US with a precious and unusual item of carry-on luggage: a cooler containing blood samples from Australian patients with neurodegenerative diseases including motor neurone disease.
After navigating several transit points and border protection checks, the samples — and their personal escort — finally reached Dr Dunlop’s laboratory at the Brain Chemistry Labs in the stunningly picturesque town of Jackson, Wyoming. Now they are poised to make a valuable contribution to the development of a groundbreaking diagnostic blood test for motor neurone disease (MND).
At present, diagnosis of MND relies on careful clinical assessment of signs and symptoms by an experienced neurologist, together with the results of tests that exclude other neurodegenerative diseases with similar and overlapping presentations.
But there’s an urgent and overwhelming need for a fast, accurate and cost-effective test based on a specific “biomarker” that can be easily collected in a minimally invasive way, said Professor Dominic Rowe AM, who heads the MND clinical service at Macquarie University Hospital.
Patients with MND have typically seen several other medical specialists before they see a neurologist with MND experience; clinical studies have found an average time of eight months between the onset of symptoms and a definitive MND diagnosis. Rates of initial misdiagnosis are also high — up to two-thirds of patients in some studies — meaning many patients undergo unnecessary medical tests and procedures along the path to having an MND diagnosis confirmed.
And while there’s currently no cure for MND, earlier diagnosis and treatment offer potential to slow the progress of the disease, ease symptoms and improve patient quality of life.
A biomarker that’s truly a surrogate for disease activity would mean earlier diagnosis and earlier initiation of therapies that slow or halt the disease.
“A biomarker that’s truly a surrogate for disease activity would mean earlier diagnosis and earlier initiation of therapies that slow or halt the disease,” Rowe said. “It would allow us to measure disease progression far more accurately; it could even help to screen for MND.”
Tell-tale ‘fingerprint’
For several years, Dunlop and colleagues in the US have been working on a test based on short, single strands of ribonucleic acid (RNA) — so-called microRNAs (miRNAs) — found in body fluids like blood. RNA is the molecule in cells that helps turn genetic instructions encoded in DNA into proteins; miRNAs don’t code for proteins themselves but control how other genes are expressed.
“We’ve identified a combination of eight miRNAs that we’ve shown are diagnostic for both sporadic and genetic MND cases,” Dunlop told fellow MND researchers at Macquarie University in a presentation during her recent visit to Australia.
Dunlop’s team first demonstrated the accuracy and specificity of this approach to testing using miRNAs found in neural-enriched extracellular vesicles (NEE) — tiny, bubble-like packets released by brain and nerve cells that circulate in the blood carrying molecular “cargo”. Their latest published research has extended and validated their approach using miRNA collected from peripheral blood without the complex and time-consuming process needed to extract miRNA from NEE.
“If we can show this miRNA ‘fingerprint’ is present in the circulation before changes in proteins we know are there by the time MND symptoms first appear, this test might have predictive power,” says Dunlop. “That’s something we’re planning to evaluate by testing for miRNA

Dr Rachael Dunlop arrives in Jackson, Wyoming with a cooler full of patient samples from the Macquarie MND Biobank.
in people who carry genes associated with MND — before they’ve developed any symptoms.”
The specificity of the eight-miRNA combination has so far been replicated in five independent patient cohorts with a combined sample size of 788. Importantly, the test has been shown to identify MND in samples from newly diagnosed patients as well as in samples from patients at various stages of the disease, diagnosed by different neurologists, and stored in a biobank for many years.
And that’s where the samples from Australian MND patients come in.
Macquarie samples will broaden clinical base
Originally from Australia, Dunlop is a visiting research associate and long-time collaborator at Macquarie, and the specimens entrusted to her care were collected as part of the MND Research Centre’s Neurodegenerative Disease Biobank, established in 2013. All patients of the MND service and clinic at Macquarie, together with their families, are invited to contribute to the Biobank.
“MND is quite heterogeneous, so we’d like to ensure our diagnostic biomarker is useful across the full range of clinical settings, independent of variables like age of onset, stage of progression and disease severity,” Dunlop said. “The Macquarie Biobank gives us access to blood samples from a different patient population attending MND
clinics where extensive clinical data have also been carefully collected.”
Rowe said it’s exciting that the extensive patient experience from Macquarie is being used to further develop the test.
“The technology developed by Dunlop and her colleagues is unique and we hope her extraordinarily impressive initial results can be replicated using our Macquarie Biobank samples,” he said.
“It is really gratifying to see our Macquarie patient samples helping advance the global war on MND.”
*Stephen Downes is a Media Advisor at Macquarie University.


Quantum technology company Archer Materials Limited has developed an early Biochip prototype designed to detect chronic kidney disease.
The company announced the completion of its alpha prototype and laboratory demonstration of its blood potassium sensor with integrated Biochip in January; a milestone, the company said in a statement, that “represents the first major step in translating Archer’s Biochip technology from individual laboratory devices into a product suitable for clinical workflows and, ultimately, point of care and at-home potassium monitoring”.
“For the first time, we have integrated our potassium sensing Biochip with fluidics and electronics into a single packaged system, while maintaining clinical-grade accuracy using patient-derived samples. This represents a major technical milestone for Archer’s Biochip program. The achievement significantly derisks the technology paving the way for developing a full (beta) prototype in 2026,” Archer CEO Dr Simon Ruffell said.
“Archer’s testing of the early prototype shows that the Biochip technology meets clinical accuracy requirements when integrated into a prototype cartridge capable of handling a drop of blood and the productready electronic readout system.”
Potassium measurement accuracy within ±0.3 mM in blood samples
was demonstrated by the Biochip prototype, Archer said, which is in alignment with Clinical Laboratory Improvement Amendments (CLIA) requirements for equivalent testing in a pathology lab and matches the previous laboratory experimental system results.
Incorporating the Biochip, fluidics and electronics into a single test system, Archer said its first integrated alpha prototype delivers the following outcomes:
• clinical-grade potassium measurement accuracy maintained while operating in a fully prototype environment;
• qualified micro-fluidics capable of performing accurate potassium testing using only 10µL of blood, a volume consistent with standard fingerprick tests and eliminating the need for finger ‘milking’, which is a key requirement for patient usability and eventual at-home adoption; and
• verified prototype electronics, confirming system-level functionality and stable performance across repeated measurements — ready for integration into final product.
The beta prototype will form the basis of negotiations with large MedTech companies regarding potential licensing deals as well as engagement with contract medical device manufacturers, the company said.
With the aim to jointly commercialise a global cryopreservation offering for farm animals and aquaculture, Vitrafy Life Sciences Limited has announced that it has entered a 12-month Strategic Commercial Agreement with IMV Technologies — a Frenchbased multinational conglomerate with over 500 million animal inseminations annually.
In a statement about the agreement, Vitrafy said the partnership is designed to integrate its next-generation cryopreservation technology with IMV’s globally recognised offering. “This agreement with IMV Technologies represents a significant step in Vitrafy’s commercialisation strategy,” Managing Director and CEO Brent Owens said.
“By partnering with the global leader in animal reproduction, we accelerate the validation and adoption of our cryopreservation technology at scale,” Owens added. “This Agreement creates immediate revenue opportunities but also positions Vitrafy for longterm growth through global market access, while we continue to advance our human health strategy in North America.”
Oliver Kohlhaas, CEO of IMV Technologies, commented: “Vitrafy’s unique nitrogen-free cryopreservation technology has the potential to improve post-thaw cell viability. We are excited to collaborate with Vitrafy to offer even better solutions for our customers.”
Vaxxas, a biotechnology company pioneering HD-MAP technology for future self-administered vaccine delivery, has appointed global biopharmaceutical executive David Peacock as CEO. Bringing more than 25 years of global vaccine and pharmaceutical leadership — most recently in roles as President of Merck Global Vaccines and President of MSD Asia Pacific — Peacock has been appointed to lead the commercialisation of Vaxxas’s proprietary high-density microarray patch (HD-MAP) vaccination technology.
“David’s appointment marks a clear inflection point for Vaxxas as we move from clinical validation towards commercial execution,” Vaxxas Chair Sarah Meibusch said. “He brings unmatched global vaccine commercialisation experience at precisely the moment the HD-MAP technology is transitioning from proof-of-concept to a scalable delivery platform with the potential to materially improve how vaccines are manufactured, distributed and administered worldwide.
“For global pharmaceutical partners, the HD-MAP offers a differentiated self-delivery administration option to enhance existing vaccine franchises, extend lifecycle value and address persistent challenges around cold-chain, dose efficiency and access. David’s leadership positions Vaxxas to engage at scale with governments and multinational vaccine companies as a long-term strategic partner.”
With a career including service on boards and industry bodies such as the US-ASEAN Business Council, The Association of the British Pharmaceutical Industry and Gavi, the Vaccine Alliance, Peacock said he is energised by the potential of Vaxxas’s HD-MAP technology to reshape

vaccine delivery and access. “Over the course of my career, I have seen first-hand the limitations of conventional vaccine delivery technologies in both developed, and developing, healthcare systems,” Peacock said. “I believe Vaxxas’s HD-MAP technology has the potential to fundamentally reshape vaccine access, distribution and administration.
“Vaxxas is now at a pivotal stage, with clinical validation, licensed manufacturing capability and a platform that can integrate with existing and future vaccine programs. I am excited to lead the company as we partner with pharmaceutical companies, research organisations and governments to translate this technology for global public-health and commercial impact.” Peacock will also be appointed as Executive Director to the Vaxxas board.
Vaxxas recently secured a manufacturing licence from the Therapeutic Goods Administration (TGA) for HD-MAP vaccine products at the company’s Brisbane biomedical facility.
Beckman Coulter Life Sciences has announced a strategic partnership with London-based lab automation company Automata that will integrate Beckman Coulter Life Sciences liquid handling, genomic and cell analysis technologies with Automata’s AI-ready automation platform with the aim of enabling faster, more consistent and more scalable experimentation for research organisations.

“This partnership reflects our relentless focus on empowering scientists to translate ideas into impact faster,” said Joe Fox, President of Beckman Coulter Life Sciences. “Agentic AI is poised to transform the way wet-lab screening studies are performed. Partnering with Automata allows us to combine our trusted life science tools with cutting-edge automation infrastructure that makes the lab of the future accessible today.”
Through the partnership, Beckman Coulter Life Sciences instruments will be integrated into Automata’s LINQ automation ecosystem — a modular, scalable platform composed of intelligent robotics, unified scheduling software and an agile workflow design environment. LINQ’s cloud-native orchestration engine is engineered to enable laboratories to standardise and automate multistep experimental processes, delivering structured, high-quality datasets that are essential for AI-driven discovery and analysis.
Beckman Coulter Life Sciences is a subsidiary of Danaher Corporation and the partnership is part of a larger investment in which Danaher Ventures will participate in Automata’s Series C funding round and join the company’s board of directors — a move intended to strengthen alignment and long-term support for joint innovation. iStock.com/jittawit.21


In today’s rapidly evolving life sciences industry, the importance of fast access to precise, reliable, and real-time process analytics cannot be overstated. As the demand for efficient and accurate measurements grows, METTLER TOLEDO is proud to announce the availability of Pendotech products in Australia and New Zealand, a move that signals a major advancement for the local biotech and pharmaceutical sectors. This development promises to bring advanced analytics technology and expert support closer to life science professionals in the region, helping them meet the increasing challenges of modern bioprocessing. These elements are vital for realising the full potential of the technology and ensuring smooth integration within existing workflows.
Pendotech has established itself as a global leader in downstream biotechnology applications, offering innovative singleuse sensors designed to deliver real-time measurement of critical parameters. These include pressure, UV absorbance, pH, conductivity, temperature, flow, and air-in-tube detection — all essential metrics for ensuring process integrity and product quality. Beyond the hardware, Pendotech’s product portfolio features sophisticated software solutions crafted to simplify measurement, monitoring, and comprehensive data collection, enabling more effective process control and analysis.
Since its inception in 2005, Pendotech has built a reputation for reliability and innovation, with its products widely trusted by pharmaceutical, biopharmaceutical, and bioprocess professionals worldwide. Technology plays an indispensable role in various applications such as research and development, filtration, concentration, chromatography, and bioreactor monitoring. These critical processes demand high precision and dependability, both of which Pendotech delivers through its advanced sensor technologies.
A defining feature of Pendotech sensors is their biocompatibility. Every sensor meets stringent USP Class VI standards before and after gamma irradiation, ensuring their suitability for sensitive bioprocess environments where contamination control is paramount. Additionally, most sensors are engineered to withstand high operating pressures — typically up to 75 psi — and incorporate IP67-rated cable connections that protect against dust and water ingress. This robust build quality ensures they perform reliably under the demanding conditions characteristic of bioprocessing operations.
The versatility of the Pendotech product line is another key advantage, supporting a wide spectrum of bioprocessing needs from early-stage R&D through to full-scale commercial production. This adaptability is critical in today’s biomanufacturing landscape, where processes and requirements can
shift rapidly. Having access to dependable, flexible analytics solutions allows companies to maintain consistent product quality and optimise process efficiency at every stage of development and manufacture.
In the highly competitive biomanufacturing environment, where innovation speed and cost management are critical factors, the efficiencies enabled by Pendotech’s advanced analytics solutions can yield significant benefits. Utilising single-use sensors for real-time data collection enables companies to streamline operations, minimise downtime, and enhance productivity. This empowers life science professionals to focus more on innovation and quality improvements rather than being bogged down by operational uncertainties and troubleshooting.
For professionals working across life sciences, biomanufacturing, and related fields, Pendotech offers a comprehensive toolkit tailored to address the complexities of modern bioprocessing. Its real-time monitoring and analytics capabilities provide critical insights that facilitate informed decision-making and process optimisation. By adopting these technologies, businesses are better positioned to meet the demands of dynamic markets while preparing for sustained growth and continued innovation.
To explore how Pendotech solutions can transform your organisation’s bioprocessing capabilities, visit mt.com or contact METTLER TOLEDO at 1300 659 761. Partnering with METTLER TOLEDO and Pendotech represents a strategic investment in enhanced process control, improved efficiency, and lasting innovation within the life sciences manufacturing sector. This step forward promises to equip life science professionals in Australia and New Zealand with the tools and support necessary to thrive in an ever-changing industry landscape.
Mettler-Toledo Ltd www.mt.com

The University of Melbourne’s Laureate Professor Sam Berkovic AC FAA FAHMS FRS has been elected as the 21st President of the Australian Academy of Science. Formerly Director of the Epilepsy Research Centre at Austin Health, Berkovic is considered one of the world’s most respected neurologists, whose pioneering work changed the way we think about epilepsy.
Epilepsy is a chronic noncommunicable disease of the brain that affects around 50 million people worldwide and is characterised by recurrent seizures. 30 years ago, doctors regarded most forms of epilepsy as acquired rather than inherited — that was until Berkovic, together with molecular genetic collaborators in Adelaide and Germany, discovered the first gene for epilepsy in 1995.
Berkovic’s own discoveries revolutionised basic scientific research into epilepsy. His research established the connection between genetics and epilepsy, with major global efforts now focused on gene discovery and understanding how genetic abnormalities cause seizures.
Outgoing Academy President Professor Chennupati Jagadish AC said he is delighted that Berkovic has been elected as his successor.
“Professor Berkovic’s journey from the child of Holocaust survivors to a world-leading researcher and clinician is powerful,” Jagadish said.
“Sam is well known for his mentorship of young researchers and has put Australia at the forefront of global research into epilepsy while improving the lives of people around the globe.”
Berkovic was elected a Fellow of the Academy of Science in 2005 (and has previously served on the Academy’s Council as a Member for Biological Sciences), a Fellow of the Royal Society in 2007, and a Member of the US National Academy of Medicine in 2017 for his outstanding contributions to science. In 2014, he won the Prime Minister’s Prize for Science alongside Professor Ingrid Scheffer for their groundbreaking work in epilepsy and its genetic links. The following year, he was recognised as the Victorian Senior Australian of the Year.
With his presidency set to officially commence in May 2026, Berkovic said he feels honoured to have the opportunity to lead the Academy and thanked the Academy Fellowship for the trust they have placed in him.
“Bringing science to the service of the nation so that everyone can benefit from it will be paramount during my presidency,” he said. “Australians are experiencing mass disruption in the form of rapid technological change, climate impacts and shifts in the way we engage with the world due to geopolitical tensions. Science is central to each of these, and it will be important that the Academy rises to these challenges.
“I am conscious that I take the reins of this esteemed institution at a time when investment in research and development is at a historic low.” he said. “I look forward to building on the advocacy of President Jagadish on this issue and to propose solutions so Australia can build an R&D system able to support our national interests.”
HaemaLogiX Ltd, a clinical stage Australian biotech developing novel immunotherapies for patients with blood cancers and B-cell diseases, has appointed Dr Chris Baldwin as Chief Executive Officer and Managing Director, having commenced 5 January 2026.
Holding a PhD in Chemical Engineering from the University of Cambridge, for which he was selected as British Marshall Scholar, Baldwin brings more than two decades of experience spanning scientific research, operational leadership, capital markets and commercial strategy across Australia, Asia–Pacific, Europe and the United States.
Having begun his career in academia as an Associate Professor of Chemical Engineering at Iowa State University, Baldwin then transitioned into industry, joining McKinsey & Company in New York. Subsequent roles have spanned C-suite and founder positions across ASX-listed and international biotechnology and precision therapeutics companies.
“HaemaLogiX has built a compelling immunotherapy platform with the potential to address significant unmet needs for patients with blood cancers,” Baldwin said. “I am excited to join the Company at this important stage of growth and to work with the Board and team to advance our clinical programs, build strategic partnerships and create longterm value for patients and investors.”

To advance a pioneering molecule for autoimmune diseases, global immunology company argenx has signed a strategic partnership with Monash University. Having opened its Australian affiliate office in early 2025, the partnership marks the company’s first partnership with Monash and its second research and development partnership in Australia.
The collaboration will see argenx and Monash advance an antibody molecule in pre-clinical development, with the shared ambition of one day addressing diseases with significant unmet need for patients. “Australia has built a world-class environment for innovative research,” argenx Australia General Manager Kathryn Evans said.
“This collaboration with Monash University reflects our continued interest in engaging deeply with Australia’s research community and learning from the scientific rigour and creativity that exists here. I’m looking forward to partnering with a team that values open exchange, long-term perspective and a shared commitment to advancing science in service of patients.”
Dr Ingmar Wahlqvist, Acting Chief Commercialisation Officer at Monash University, said: “This partnership is an excellent demonstration of what’s possible when leading researchers and industry partners come together with a shared commitment to achieving impact in areas of significant unmet need.”
Wahlqvist added: “We are excited by this combination of Monash’s research expertise and argenx’s global development capabilities. It highlights Monash’s commitment to industry collaboration, especially when it has the potential to deliver positive benefits for patients worldwide, and long-term value for both organisations.”
Initiated as part of argenx’s Immunology Innovation Program (IIP), the partnership will span key milestones from early-stage research, with the goal of reaching clinical trials and registration. Through its IIP, argenx works closely with leading scientists around the world to source novel biology that may lead to treatments for patients living with rare and underserved immunological conditions.





In December, scientists at St. Jude Children’s Research Hospital in the US made public Combocat — a screening platform that, by combining acoustic liquid-handling protocols with machine learning, promises a faster way to test drug combinations and doses.
Combinations of drugs — whose effects are more than the sum of their parts — are required to create effective treatment regimens for many diseases, including cancers. However, according to St. Jude Children’s Research Hospital in the United States, as the number of new drugs and potential combinations has ‘exploded’, classical screening methods have been made impractical.
Combocat, an open-source screening platform
That’s why, to address this need, the hospital’s researchers — dedicated to advancing cures and means of prevention — created Combocat, a platform that combines specialised liquid-handling technology with machine learning, to enable larger combination screens and make discovering effective drug combinations easier.
“The field of drug discovery has lacked a way to deal with the sheer number of potential combinations, which would require impractical amounts
of experimental materials to screen,” said senior co-corresponding author Dr Paul Geeleher from the St. Jude Department of Computational Biology.
“We designed Combocat to use minimal resources and enable scientists to test massive numbers of drug combinations, rapidly nominating those most likely to have synergistic effects to explore further,” Geeleher added.
The Nature Communications paper
In December 2025, the team detailed the platform’s capabilities in a paper titled ‘An open-source screening platform accelerates discovery of drug combinations’ — published open access in the journal Nature Communications (doi: 10.1038/s41467-025-66223-8).
About the platform, first and co-corresponding author Dr Charlie Wright from the St. Jude Department of Computational Biology said: “By tying together leading methods in machine learning and drug dispensing technology to power high-throughput combination screening, we performed experiments on a scale that was not feasible before.”
For proof of principle of the platform’s capabilities, 9045 pairs of drugs were tested by the researchers against a neuroblastoma cancer cell line. Multiple drug pairs with strong synergistic effects were uncovered by the


(L) First and co-corresponding author Charlie Wright, PhD and (R) co-corresponding author Paul Geeleher, PhD, both of the St. Jude Department of Computational Biology.
We designed Combocat to use minimal resources and enable scientists to test massive numbers of drug combinations, rapidly nominating those most likely to have synergistic effects to explore further.
The confidence test
The sparse mode model was trained on hundreds of drug combination experiments generated with the platform’s dense mode approach; confidence in the approach coming when the researchers compared the machine learning predictions to measured values — finding them to be highly consistent.
“We created two screening ‘modes’, with something of a trade-off between them,” Geeleher said. “We optimised the sparse mode approach for scale, but it trades detail for efficiency, while we optimised the dense mode to obtain ultra-reliable measurements, which can’t scale. However, we showed that Combocat can combine them to analyse more combinations and validate them more rapidly than traditional approaches.”
screen, and the top findings were confirmed with additional experiments.
What the results demonstrated, according to St. Jude, is that Combocat can efficiently uncover promising drug combinations at scale.
The path to scale
Miniaturised drug dispensing with machine learning was the means of achieving scale with the platform, with sonic technology enabling customised and efficient experimental layouts for drug dispensing.
“We incorporated acoustic liquid handlers that use sound waves to transfer tiny droplets of drugs very precisely,” Wright explained. “They use the exact minimum of each liquid you need, allowing for the use of far less material than conventional pin or pipette-based techniques, and increasing the number of testable combinations.”
By using one of Combocat’s two modes, ‘dense mode’, to inform its ‘sparse mode’, machine learning helps fill in the picture. In dense mode, the researchers measured every possible dose pairing for each drug combination. Alongside this dense mode, a sparse mode allows for even tighter resource management by predicting the full results from only a small fraction of the original data.
The potentialities of open source
According to St. Jude, the platform continues a legacy of drug combination therapy innovation at the research hospital, providing a way to easily screen combinations not just for cancer researchers, but for any disease in need of new treatments.
“We’ve created a platform that’s free, open-source and highly usable that could become a strong standard in the drug combination discovery field,” Geeleher said.
“Combocat can help expedite the identification of potentially safe and effective drug combinations, which could ultimately yield useful and potentially practice-changing new drug combinations in the clinic,” Geeleher added.
The study’s other authors — Min Pan, Gregory Phelps, Jonathan Low, Duane Currier, Ankita Sanjali, Marlon Trotter, Jihye Hwang, Richard Chapple, Xueying Liu, Declan Bennett, Yinwen Zhang, Richard Lee and Taosheng Chen — were all from St. Jude, with the study having been supported by grants from National Institute of General Medical Sciences, National Cancer Institute, National Human Genome Research Institute and ALSAC, the fundraising and awareness organisation of St. Jude.

Australian and international researchers have identified a new bloodclotting disorder that has been found to have certain similarities to vaccine-induced immune thrombocytopenia and thrombosis (VITT) — a rare but aggressive clotting disorder that was caused by certain discontinued COVID-19 vaccines.
Led by Professor Ted Warkentin — professor emeritus in the Department of Pathology & Molecular Medicine at McMaster University, Canada — the study demonstrates, in the words of clinical team leader Professor Tom Gordon from the College of Medicine and Public Health and SA Pathology: “This is a new disorder identified by researchers at McMasters University and is of considerable importance to all physicians.”
The newly identified disorder has been termed VITT-like monoclonal gammopathy of thrombotic significance (VITT-like MGTS) — the Warkentin-led study revealing that certain patients can develop severe blood clotting due to
antibodies that closely resemble those that cause VITT, even in the absence of known triggers for such antibodies, such as blood thinners (heparin) or prior vaccination.
The “major jump” of a VITT-like chronic condition
Linked to the AstraZeneca COVID-19 vaccine in early 2021 and leading the Australian Government to restrict its rollout to those aged over 65, VITT is also associated with the Johnson & Johnson vaccine Janssen — although the vaccine is not currently included in Australia’s vaccination program.
“We all remember those difficult times during the pandemic when the vaccine was linked to the rare, sometimes lethal clotting complication called vaccine-induced immune thrombocytopenia and thrombosis or ‘VITT’,” Gordon said. “This was
initially thought to be self-limiting over days and weeks.”
Gordon added: “The major jump in knowledge coming from this new study is that a highly similar chronic condition — over months and years — can occur with patients presenting with intermittent clotting episodes.”
“Proven” via a proteomic approach
The study included a detailed analysis of cases exhibiting unusual blood-clotting despite patients being on full-dose blood thinners. Including multinational collaboration, data was collected from five patients who had unexplained VITT-like antibodies that were detectable for a year or more and who were treated at institutions in Canada, France, Germany, Spain and New Zealand.
The presence of M (monoclonal) proteins (which typically indicate plasma cell disorders) was identified by the analyses, which together with the persisting VITT-like reactivities over at least 12 months (which is highly unusual for most anti-PF4 antibodies), pointed to an ongoing pathological process rather than a short-term anomaly.

“Low-level serum M (monoclonal) protein are often identified in patients who have VITT-like MGTS,” said research team leader and co-first author Dr Jing Jing Wang from the College of Medicine and Public Health and Flinders Health and Medical Research Institute (FHMRI). “By using our proteomic approach developed at Flinders Proteomics Facility, we have proven that the M proteins are the pathological VITTlike antibodies.
“Despite these M proteins being in relatively low concentrations, they are highly pathological VITT-like proteins, which explain the patients’ severe symptoms,” Wang said. That each of the patients had failed blood thinning treatment yet showed some benefit with unusual treatments — such as high-dose intravenous immunoglobulin (IVIG), Bruton tyrosine kinase inhibitors (ibrutinib), and plasma cell-targeted myeloma therapy — was a key observation.
Developing more effective treatment strategies
For their part, Flinders researchers played a key role in analysing the specific antibodies that are involved in VITT-like MGTS. “We examined the antibodies to see how they are constructed by our immune system and what makes this new disorder different from the classic VITT cases we saw
This is a new disorder identified by researchers at McMasters University and is of considerable importance to all physicians.

during the pandemic and to improve our overall understanding of this condition,” Wang said.
“Our findings indicate that chronic anti-PF4 disorders, such as VITT-like MGTS, have distinct immunological features and require tailored diagnostic and therapeutic approaches,” Wang added. “The chronic nature of these disorders often leads to severe clinical outcomes, necessitating comprehensive management strategies.”
With the potential to improve patient outcomes and the expectation that their findings will influence how doctors test for and treat patients with unusual or recurrent blood clotting, it is believed that the existence of this novel blood clotting disorder will have important implications for how healthcare providers will evaluate patients who develop unusual or difficult-to-treat blood clots in the future.
In the words of co-first author Warkentin: “By understanding how to diagnose VITT-like MGTS, we can develop more effective treatment strategies that go beyond traditional anticoagulation.”
The Cayman Chemical Off-Patent Drug Screening Library (96-Well) consists of eight plates and contains approximately 607 compounds in a 96-well Matrix tube rack format as 10 mM stock solutions in DMSO. Featuring a curated collection of FDA-approved compounds that are no longer covered under composition-of-matter patents in the United States, the library is intended for drug repurposing screening. The library contains compounds from a variety of therapeutic areas, including oncology, gastroenterology, neurology, immunology, endocrinology, ophthalmology, pulmonology, infectious diseases and cardiology, among others. It is distributed in Australia and New Zealand by Sapphire Bioscience. Sapphire Bioscience www.sapphirebioscience.com

Camfil’s CleanSeal AU is a robust, fully welded ceiling housing, specifically engineered for the Australian market. Designed to deliver reliable performance and ease of use, it is suited to cleanrooms, laboratories and life science facilities where precision airflow and contaminant control are critical. The housing can be customised to suit specific finishes, formats or fully bespoke ceiling integrations, providing flexibility to meet the unique requirements of any facility.
Equipped with a full suite of measurement ports, CleanSeal AU enables accurate filter validation and monitoring, making it suitable for both supply and exhaust air applications. The premium integrity variant supports ISO 14644-3 in-situ testing, offering a complete installation kit for high-performance cleanroom environments.
CleanSeal AU is compatible with both PU and GEL gaskets and features a universal mounting system, allowing for suspended or compression installations. Its pre-positioned filter retainers and patented adjustable clamping system accommodate all Camfil Megalam HEPA filter sizes (MG/ MD/MX), facilitating quick upgrades to maintain pressure cascades and enhance energy efficiency.
Key advantages include: stainless steel filter retaining section for gasket or gel seal HEPA filters and diffusers; quick front plate locking for immediate filter access; perforated diffuser standard, with alternative options available; powdercoated plenum standard, optional stainless-steel finish; and optional adjustable damper for precise room airflow control.
CleanSeal AU is engineered to combine durability, flexibility and compliance with stringent cleanroom standards, making it a reliable solution for maintaining optimal airflow, pressure control and energy-efficient operation in demanding life science and pharmaceutical environments.
Camfil Australia Pty Ltd www.camfil.com.au
Visualising intact biological structures in three dimensions remains a major challenge for researchers. Conventional histology techniques require thin tissue slicing, which can distort spatial relationships and make it difficult to interpret complex cellular networks. Thick or whole-tissue imaging, on the other hand, is hindered by light scattering caused by lipids, leading to opaque samples and poor imaging depth. There is a growing need for standardised clearing methods to clear tissues efficiently without damaging structure or losing fluorescent signals.
The X-CLARITY system from Logos Biosystems addresses these challenges by automating and standardising the tissue clearing process. Using proprietary electrophoretic tissue clearing (ETC) technology, the system rapidly and uniformly removes lipids from fixed tissues, rendering them optically transparent while maintaining molecular and structural integrity.
The X-CLARITY tissue clearing system, together with dedicated hydrogel and mounting solutions, provides an all-in-one, easy-to-use workflow from start to finish. The process can clear a mouse brain in just a few hours, eliminating manual variability and improving consistency across samples.
Once cleared, tissues can be imaged using confocal, two-photon or light sheet microscopy to reveal intricate 3D architecture — from neuronal pathways and vascular networks to tumour microenvironments. By simplifying and accelerating tissue clearing, the system empowers researchers in neuroscience, developmental biology and pathology to gain deeper insight into biological systems.
ATA Scientific provides demonstrations and expert support to help laboratories adopt this transformative workflow.
ATA Scientific Pty Ltd www.atascientific.com.au

OGT has launched its next-generation sequencing (NGS) panel for measurable residual disease (MRD): the SureSeq Myeloid MRD Plus NGS Panel. Designed using OGT’s 30 years of expertise in hybrid capture technology, the panel detects ultralow-frequency variants in key MRD-associated biomarkers in acute myeloid leukaemia (AML). This means users can obtain detailed genomic information, to provide an earlier and more complete picture of their sample’s MRD status.
Detection of MRD is a pivotal component in advancing clinical research and improving our understanding of therapeutic strategies for AML. Evidence consistently demonstrates that MRD-negative status is associated with significantly improved long-term outcomes and the ability to sensitively identify even a small number of residual leukemic cells at earlier time points. This provides clinicians and researchers with a more detailed snapshot of disease status.
With targeted coverage of 16 biomarkers associated with AML, including FLT3 and NPM1, the panel is optimised for MRD characterisation. Users benefit from robust detection of complex and challenging mutations such as FLT3 internal tandem duplications (ITDs) exceeding 300 base pairs, so they can obtain comprehensive genomic insights from their samples. Additionally, its focused design enables high-precision variant detection without the increased sequencing costs typically associated with broader panels and, by leveraging SureSeq’s refined bait design strategy, the panel achieves high sensitivity, detecting variants at allele frequencies as low as 0.01%.
Complementing the panel is Interpret, OGT’s powerful bioinformatics solution provided at no additional cost, which includes longitudinal visualisation of MRD dynamics over time — including SNVs, indels and ITDs. Available as a secure cloud-based platform or local installation, it offers user-friendly preconfigured analysis pipelines which streamline laboratory workflows and accelerate implementation. Additionally, customisation options allow users to tailor their analysis to meet their specific clinical research needs.
The panel and bioinformatics solution are supported by OGT’s dedicated team of experts, who provide comprehensive assistance from initial set-up through to advanced troubleshooting, enabling seamless adoption of MRD detection. Sysmex Australia Pty Ltd www.sysmex.com.au
Dharmacon ON-TARGETplus siRNAs by Revvity combine a patented dual-strand modification pattern with the SMARTselection algorithm to deliver efficient and specific gene silencing, making the product suitable for RNAi experiments.
Benefits are said to include: up to 90% reduction in off-target effects compared to unmodified siRNA; guaranteed target silencing, with at least three of four individual siRNAs in each SMARTpool performing effectively; and full sequence information provided with every siRNA purchase for transparency and reproducibility.
The siRNAs are available for human, mouse and rat targets, with users able to choose the format that best fits their workflow. SMARTpool is a potent and specific mixture of four siRNAs combined in one reagent for good knockdown. Alternatively, users can purchase aliquots of four individual siRNAs targeting their gene of interest. Single siRNAs can also be selected for targeted experiments.
The siRNAs are designed to give researchers the confidence to interrogate gene function with precision and specificity. Experimental success can be maximised by pairing ONTARGETplus with Dharmacon non-targeting and positive controls, along with DharmaFECT transfection reagents or electroporation for efficient intracellular delivery.
Millennium Science Pty Ltd www.mscience.com.au

Maternal stress during pregnancy can speed up the timing of teeth eruption, which may be an early warning sign of faster biological aging.
Milk teeth begin to develop for children in the womb around the sixth week of gestation, and gradually come out (‘erupt’) between six months and three years after birth. However, there is considerable variation in this timing, due to genetics, geography, and an infant’s overall health and nutritional status. Now, researchers from the US have shown that maternal stress during pregnancy can speed up the timing of teeth eruption, which may be an early warning sign of faster biological aging.
The University of Rochester’s Dr Ying Meng and colleagues studied a cohort of 142 mothers in the US from socioeconomically disadvantaged backgrounds, who were pregnant between 2017 and 2022 and enrolled through the university’s medical centre. In the late second and third trimesters of pregnancy, each woman gave a saliva sample, in which the concentration of the hormones cortisol, oestradiol, progesterone, testosterone, triiodothyronine and thyroxine was measured.
All children in the study were born at full term. At one, two, four, six, 12, 18, and 24 months after birth, each mother–child pair visited the clinic, where dentists assessed which milk teeth had erupted. The results were published in Frontiers in Oral Health
By six and 12 months of age, respectively, 15% of children had between one and six erupted teeth while 97.5% had between one and 12. Importantly, women with higher levels of the stress hormone cortisol in their saliva had offspring with a greater number of erupted teeth by six months of age. Infants of mothers with the highest level of cortisol had on average four teeth more at this age than infants of mothers with the lowest cortisol levels.
“High maternal cortisol during late pregnancy may alter fetal growth and mineral metabolism, including the regulation of levels of calcium and vitamin D — both essential for mineralisation of bone and teeth,” Meng said. “Cortisol is also known to influence the activity of so-called osteoblast and osteoclast cells, responsible for building up, shaping and remodelling bone.
“These results are further evidence that prenatal stress can speed up biological aging in children. Premature eruption of teeth could thus serve as an early warning sign of an infant’s compromised oral development and overall health, associated with socioeconomic deprivation and prenatal stress.”
The authors also found an association between a mother’s level of the sex hormones oestradiol and testosterone and a greater number of erupted teeth in her child at 12 months after birth, but this link appeared to be weaker. Similar weak yet statistically significant positive associations were found between a mother’s level of progesterone and testosterone and her child’s number of teeth at 24 months, and between her level of the thyroid hormone triiodothyronine and the child’s number of teeth at 18 and 24 months.
Oestradiol, progesterone and testosterone are known to play important roles in fetal development and birthweight, which is how high levels of these hormones might speed up tooth eruption.
“We still have key questions that need answering; for example, which maternal hormones or downstream developmental pathways drive the change in the timing of tooth eruption, what the exact relationship is between accelerated eruption of teeth and biological aging and development, and what such speeding up says about a child’s general health,” Meng said.
Monash University has launched what is claimed to be Victoria’s most advanced, research-dedicated MRI scanner to support scientists as they accelerate breakthroughs in critical health challenges like dementia, schizophrenia, addiction, epilepsy and cancer.
The Siemens Healthineers MAGNETOM Cima.X 3T MRI scanner, housed at Monash Biomedical Imaging (MBI) in Clayton, is designed to utilise multimodal imaging capabilities to deliver scans more quickly and accurately than ever before. This critical research infrastructure replaces an existing system at MBI and was delivered through $2 million in matched co-investment from Monash University and the National Imaging Facility, with funding from the Department of Education’s National Collaborative Research Infrastructure Strategy (NCRIS) program.
Accelerating translation from bench to bedside, the outcomes delivered by the Cima.X scanner are set to strengthen Australia’s global leadership in research innovation and the development of the next generation of therapeutics. Monash University Pro ViceChancellor (Research Infrastructure) Professor Jacek Jasieniak said the scanner is critical to ensuring continued innovation and development of the university’s globally impactful research.
“The major technological advancements offered by the Cima.X were specifically developed to drive innovation and new discoveries,” Jasieniak said.
“In addition to accelerating our capability for much faster and greater impact in the future, the scanner will attract new research collaborations and partnerships and strengthen Australia’s international competitiveness for translational research.
“This will give our researchers and clinicians even more support to deliver the life-changing and -saving work they do every day.”
The scanner is especially suited to neuroscience and mapping the intricate structure of the brain, thanks to its unprecedented resolution and short scan times; it is particularly useful for imaging the brain in vulnerable populations including pregnant women and
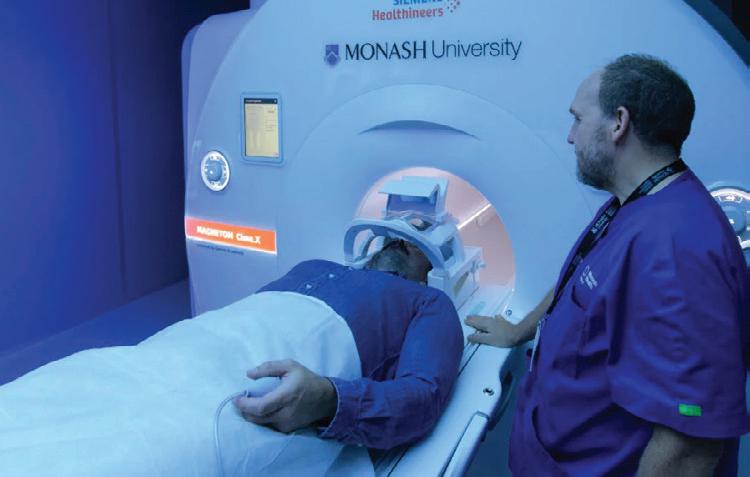
children. It will therefore be pivotal in supporting flagship university projects like the Monash Brain and Behaviour Project, which is recruiting thousands of patients for detailed neuroimaging and psychiatric assessments, and the Monash Autism and ADHD Genetics and Neurodevelopment project to map brain changes across autism and ADHD.
“The unparalleled capacity of the Cima.X to capture the intricate microstructure of the brain will lead to a step change in our understanding of neurological and psychiatric disorders,” said Associate Professor Sharna Jamadar, Deputy Director of MBI.
“I am particularly excited about how the improved precision will benefit researchers like Prof Antonio Garcia-Verdejo, who is using the Cima.X to identify individualised treatment targets for alcohol use disorder, and Prof Adeel Razi, who is using the Cima.X to refine psychedelic therapies.”
With the MBI facility co-located in the same precinct as the Monash Velos Accelerator alongside the university’s MAVERIC supercomputer, the Cima.X offers a novel opportunity for AI researchers to collect high-resolution and signal-to-noise imaging data for advanced artificial intelligence algorithms development. MBI Director Professor Christoph Hagemeyer said the scanner will ultimately deliver better patient care and outcomes, with foundational research being translated sooner into better care.
“So much of today’s diagnosis and treatment relies on MRI scans,” Hagemeyer said.
“The Cima.X instrument has the highest gradient strength in the world, which means better resolution faster than ever before, and much better patient comfort.
“Australian clinicians are aiming to diagnose many conditions earlier and improve treatments, and the Cima.X supports that critical work.”
Siemens Healthcare Pty Ltd www.healthcare.siemens.com
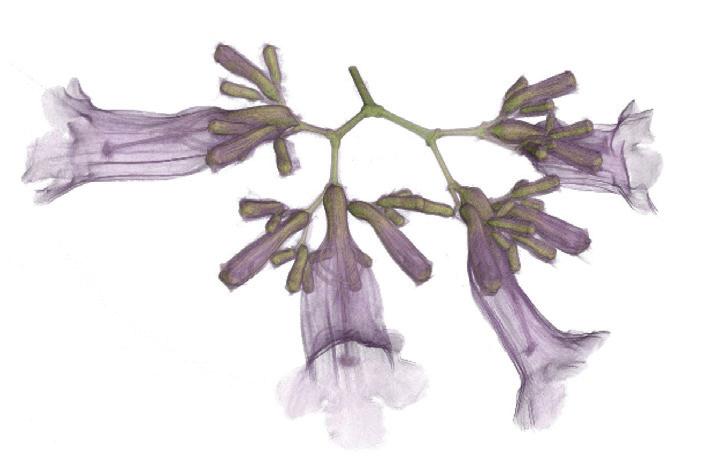
Images of intricate biological structures and everyday items have revealed the capabilities of The University of Queensland’s Yxlon FF35 micro-CT scanner.
The University of Queensland’s (UQ) seven-tonne industrial micro-CT scanner can handle tiny samples — down to 1 mm, or as large as 400 mm — and offers “huge scope” for use in research across both industry and academia, according to Associate Professor Gary Cowin, National Imaging Facility Fellow at UQ’s Australian Institute for Bioengineering and Nanotechnology (AIBN).
“The level of detail that can be achieved is incredible, from visualising the internal structure of a toothpick to mapping the tiny brains of insects,” Cowin said. “These images demonstrate the scientific and commercial potential of this technology.
“Agricultural researchers have used the micro-CT scanner to study root growth under different soil conditions,” Cowin explained; also saying that the technology supports disciplines as diverse as archaeology, biology, engineering and materials science.

“Materials scientists and engineers have used it to assess porosity and cracks in carbon fibre for advanced manufacturing and verify the accuracy of 3D-printed components,” Cowin added. “We’ve scanned everything from mining ore samples to praying mantis brains.”
Colour-enhanced scans of native flowers and the timber of a matchstick are among the more striking outputs — revealing hidden cellular structures in vivid detail. “Being able to zoom in at the micron level allows you to see patterns and textures that are both scientifically valuable and visually interesting,” Cowin said.
Funded through contributions from the National Imaging Facility (NIF), in addition to researchers at UQ and elsewhere, the micro-CT scanner is accessible to commercial partners through the national facility.
“We can provide scanning services or train users to operate the system themselves, whether they’re developing new materials, investigating biological systems or need high-resolution imaging for quality control,” Cowin said. “The idea is for the technology to be as accessible as possible.”
Based at UQ’s AIBN, a range of research capabilities are said to be provided, including human imaging, pre-clinical MRI and molecular imaging, high-resolution MRI microscopy, industrial micro-CT imaging, NMR spectroscopy, EPR Spectroscopy, radio chemistry and a cyclotron for radioisotope production.
Companies and researchers interested in using the micro-CT scanner can contact the National Imaging Facility-Qld at AIBN.

Dr Gary Cowin and micro-CT scanner.


Molecules produced by gut bacteria upon digestion of dietary fibre can improve the function of cancerfighting immune cells, according to new research.
Scientists at The Peter Doherty Institute for Infection and Immunity, in collaboration with the Peter MacCallum Cancer Centre and Monash University, say they have uncovered how the gut microbiota help the immune system fight melanoma, explaining why patients with a fibre-rich diet and balanced gut bacteria tend to respond better to cancer immunotherapies.
The team’s study, published in the journal Immunity, showed that molecules produced by gut bacteria upon digestion of dietary fibre can improve the function of cancer-fighting immune cells. The research team found that these digestive by-products influence melanoma progression by naturally boosting killer T cell function in preclinical cancer models, filling important gaps in our knowledge about how the gut microbiota regulate melanoma immunity.
“Melanoma patients undergoing immunotherapy can benefit from a fibre-rich diet and previous studies suggested that what we eat affects the immune system; however, how that works wasn’t clear,” said Dr Annabell Bachem, a Senior Research Fellow at the Doherty Institute and co-first author on the paper.
“When gut bacteria break down dietary fibre and other nutrients, they produce small molecules called metabolites. In this study, we found that the process involved in the production of short-chain fatty acids, a type of metabolite, plays a key role in how cancer-fighting T cells function.
“These metabolites act like messengers and fuel — directing how T cells
function and powering them with the necessary energy so that they can fight cancer more effectively.”
Senior author Professor Sammy Bedoui, Laboratory Head at the Doherty Institute, said the work highlights the importance of discovery research.
“This project began by us asking very fundamental questions about how killer T cells function,” he said. “Once we identified how killer T cells responded to the microbiota-derived metabolites, it became obvious that resolving these fundamental questions about how T cells operate might also help understand why only some patients with melanoma respond effectively to immunotherapies.”
Together with Professor Shahneen Sandhu, a medical oncologist at Peter Mac and close collaborator of the Doherty research team, they applied their experimental findings to melanoma patients and discovered that the gut microbiome from advanced melanoma patients responding to immunotherapy also shows signs of increased short-chain fatty acid production.
“Our discovery could help explain why recent clinical studies have shown that patients with advanced melanoma who follow a high-fibre diet and have a healthy gut microbiome respond better to immunotherapy,” Bedoui said.
“T cells can become exhausted and lose the ability to properly fight cancers. Our study showed that the microbiota-derived metabolites preserve the function of T cells and thereby improve their capacity to kill melanoma cells.
“Resolving these links between diet and the killer T cell response could help improve treatment for melanoma.”
A $5 million NHMRC Synergy Grant has since been awarded to Sandhu, Bedoui, Dr Vanessa Marcelino (Doherty Institute), Professor Tony Papenfuss (WEHI), Dr Lavina Spain and Professor Grant McArthur (both Peter Mac) to address this further and test their experimental findings in clinical studies of melanoma patients.
“Collectively, our research will provide new insights into how microbial metabolites may regulate immune responses against melanoma and to identify new ways to treat melanoma patients and improve their outcomes,” Sandhu said.
Could this independent comparison test of lab ventilation systems under real-world stress conditions redefine standards for safety, efficiency and flexibility in life sciences labs?
Project PEARL (Performance for Efficient Airflow Regulation in Laboratories) — which purports to be the world’s first to compare different lab ventilation and air distribution systems under controlled, real-world conditions — has released results. Released by Siemens, the “comprehensive and independent study” on laboratory ventilation in the area of life sciences was conducted by Siemens together with H. Lüdi + Co. AG — a supplier of core components and system solutions for modern laboratories and a Siemens Xcelerator partner — between November 2024 and February 2025, with Lucerne University of Applied Sciences and Arts in Lucerne, Switzerland contracted as the independent research and testing institution to execute all measurement activities.
The study proceeded as follows. During the test phase, three different air supply systems were controlled and measured in seven different configurations under comparable situations and were stressed to their limits. A unique set of data insights for each system resulted, where specific situations can be compared, addressing various elements like safety and contamination control, user comfort, system flexibility, adaptability, and ventilation efficiency. According to Siemens, what was found is that precise control of airflow is paramount, leading to safer, more efficient, and comfortable lab environments.
“In fact, oversupplying air can disrupt controlled conditions, elevate costs and increase CO2 emissions,” Siemens stated in a release of findings. “Remarkably, some scenarios required less than half the volume of air, achieving 45% better ventilation efficiency and demonstrating superior removal of dangerous gases and heat.

Better-controlled airflow also led to up to 29% faster recovery times after simulated spills, directly improving user safety and comfort.”
Project PEARL advocates a shift in lab space design — from designing for a single, specific use to creating flexible, next-generation environments capable of scaling from zero up to 300 watts per square metre. Such advanced systems are, the project believes, right-sized and well-controlled, delivering the exact amount of air needed to maintain safety and comfort while simultaneously saving energy and reducing emissions. As physical tests were conducted, a digital model of the complete project set-up was built by Siemens, including a simulation of the tests themselves.
“Comparing our project PEARL digital model with real-world measurements revealed amazing accuracy,” said Tim Walsh, Global Solution Director for Life Sciences at Siemens Smart Infrastructure. “This validated knowledge allows us to fine-tune and optimise performance, safety and comfort for future lab designs directly within their digital twin.”
Siemens says it has improved the Smart Lab Ecosystem (SLE) — a modular infrastructure kit that allows for the creation of highly adaptable and scalable lab environments that can be tailored to specific research needs, from basic research to high-containment facilities — based on the insights from project PEARL. The SLE enables, according to Siemens, up to 80% faster design and configuration of all types of lab environments up to bio-safety level 2.
On the project’s significance, Walsh deemed it “a game-changer for the laboratory industry”. “For the first time, we have real-world data that not only confirms our digital models but allows us to refine them and design laboratories that are truly optimised for safety, comfort and efficiency.”
Fuelled by R&D investments, biotech innovations and a growing pipeline of new drugs, the demand for laboratory space is on the rise in all key geographies. In the United Kingdom alone, for example, to cover predicted research needs estimates call for one million square metres of additional lab space in the next five years. This study’s findings, Siemens believes, help address the critical need for safer, more flexible and energyefficient laboratories.
The Seer Proteograph product suite is intended to represent a meaningful shift in how researchers access biological insight. Rather than being limited by the inherent complexity and dynamic range of proteomes, the suite aims to ensure scientists can explore biology with greater depth, reproducibility and scale. The Proteograph platform is built on a simple but powerful idea: use engineered nanoparticles to uncover proteins that traditional workflows often miss, enabling a more comprehensive and unbiased view of the proteome.
What sets the Seer Proteograph product suite apart is its end-to-end approach. From automated sample preparation to data-rich protein discovery, the suite integrates seamlessly into modern laboratory workflows. This design allows researchers to move beyond incremental gains and instead achieve step-change improvements in throughput and consistency — critical factors for large-scale studies and translational research.

As proteomics increasingly underpins advances in biomarker discovery, drug development and precision medicine, the suite recognises that need for scalable and robust solutions has never been greater. The Proteograph suite aims to meet this demand by transforming complex biological samples into actionable datasets, empowering scientists to ask better questions and get clearer answers.
Bio-Strategy - Part of DKSH Group www.bio-strategy.com
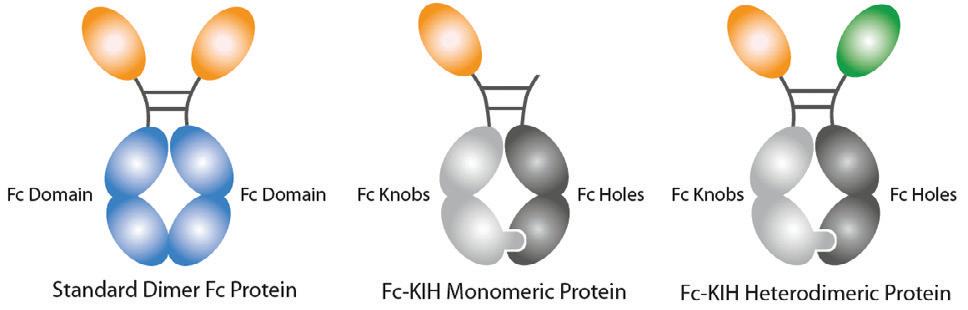
InVivoKines are recombinant fusion proteins for immunotherapeutic, preclinical and translational in vivo research, developed in-house by AdipoGen Life Sciences. The company has leveraged knobs-into-holes (KIH) technology to develop monomeric, heterodimeric or homodimeric Fc fusion proteins. Features include: native conformation (production in HEK 293 or CHO cells); production under animal-free conditions; high bioactivity tested by ELISA/ cell-based assays; verified purity and homogeneity by SEC; low endotoxin levels; and batch-to-batch consistency.
InVivoKines are distributed in Australia and New Zealand by Sapphire Bioscience.
Sapphire Bioscience www.sapphirebioscience.com
Alithea Genomics has launched MERCURIUS Spheroid DRUG-seq — a novel, extraction-free RNA sequencing library preparation kit built for 3D spheroid models. The product extends Alithea’s MERCURIUS DRUG-seq technology into physiologically relevant 3D systems, bringing simplicity, sensitivity and throughput to drug screening in more in vivo-like cellular environments.
The kit enables researchers to bypass conventional RNA extraction and pre-amplification, enabling direct processing of spheroids in an optimised lysis buffer. The streamlined, one-day workflow delivers full transcriptome coverage in a multiplexed manner, making the method suitable for large-scale mechanistic and toxicity studies in 3D models.
The kit has successfully detected >14,000 genes per sample at ~1.7 million reads and is compatible with Illumina and AVITI sequencers (with broader compatibility upon request). It is expected to unlock new layers of transcriptomic discovery in drug screening and toxicology, enabling researchers to explore disease biology in architectures far closer to human tissue.
Custom Science Australia Pty Ltd customscience.com.au
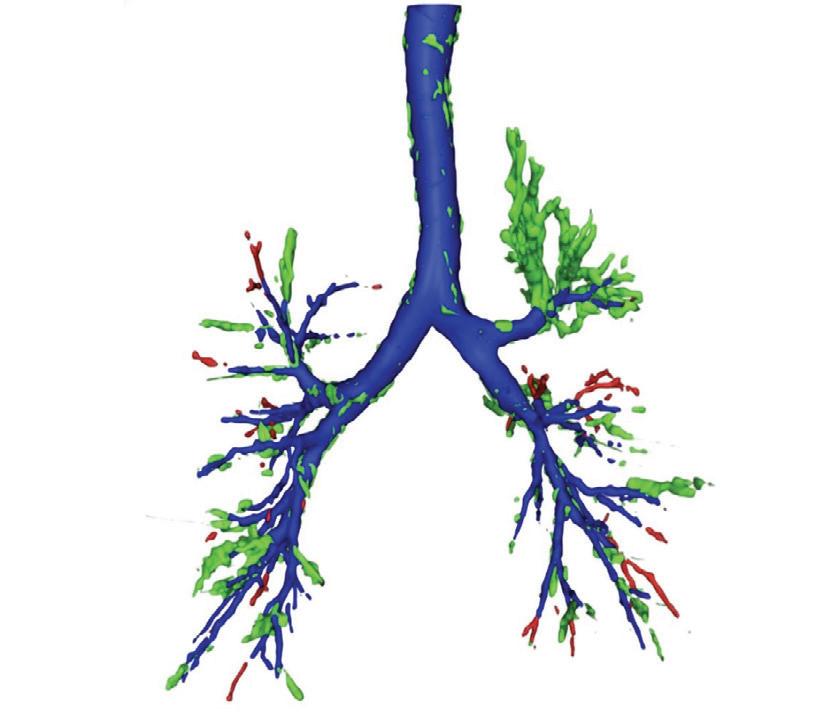
A new artificial intelligence tool can spot hard-to-see objects lodged in patients’ airways better than expert radiologists.
Researchers at the University of Southampton have developed an artificial intelligence (AI) tool that can spot hard-to-see objects lodged in patients’ airways better than expert radiologists, as demonstrated in a study published in npj Digital Medicine — highlighting how AI can support doctors in diagnosing complex and potentially life-threatening conditions.
Foreign body aspiration (FBA) occurs when an object — often food or a small piece of material — becomes lodged in the airways. These accidentally inhaled objects can cause coughing, choking and difficulty breathing, and sometimes lead to more serious complications if not treated properly.
The problem is, when such objects are radiolucent (invisible on X-rays and faint even on CT scans), they can be very difficult to detect. This often leads to missed or delayed diagnoses, putting patients at risk of serious complications. Up to 75% of FBA cases in adults involve radiolucent foreign bodies.
“These objects can be extremely subtle and easy to miss, even for experienced clinicians,”
said PhD researcher Zhe Chen, co-first author on the study.
To address this challenge, the research team created a deep learning model that combines a high-precision airway mapping technique (MedpSeg) with a neural network that analyses CT images for hidden signs of foreign bodies. The model was trained and tested using three independent patient groups, consisting of over 400 patients, in collaboration with hospitals in China.
To put the model to the test, researchers compared its performance to that of three expert radiologists, each with over 10 years of clinical experience. The task was to examine 70 CT scans, 14 of which were cases of radiolucent FBA, confirmed by bronchoscopy.
When the radiologists detected a case of radiolucent FBA, they did so with total precision — there were no false positives. In comparison, the AI model did so with 77% precision, detecting some false positives.
However, the radiologists missed a large portion of FBA cases, identifying just 36% of them and highlighting the difficulty humans have in spotting such cases. The AI model, on the other hand, was able to spot 71% of cases, meaning far fewer FBA cases slipped through the net. When it
came to the F1 score, which balances precision and recall, the model outperformed the radiologists with a score of 74% vs 53%.
The researchers emphasised that the system is designed to assist, not replace, radiologists, providing an additional layer of confidence in complex or uncertain cases. As noted by Chen, “Our AI model acts like a second set of eyes, helping radiologists detect these hidden cases earlier and more reliably.”
“The results demonstrate the real-world potential of AI in medicine, particularly for conditions that are difficult to diagnose through standard imaging,” added Dr Yihua Wang, lead author of the study. The researchers now aim to conduct multi-centre studies with larger and more diverse populations to improve the model and reduce the risk of bias.
A high-speed centrifuge is a common sight in laboratories. They are essential for liquid and gas sample separation based on density. Scientists rely on them when studying cells, proteins, viruses and more. However, there are concerns that necessitate both careful selection of a highquality centrifuge and safety training to ensure an accident-free laboratory.
If used improperly, centrifuges can pose safety risks because of the high centrifugal forces generated by the rotor during use. Improper installation of the rotor or sample imbalance can result in damaged equipment, ruined samples and, at worst, serious injury. Sample container breakage is a very real concern as broken glass can cut or otherwise injure the operator, leaked liquids may cause burns or skin irritation and released aerosols can be harmful if inhaled.
Safety first
According to guidance issued by OSHA (Occupational Safety and Health Administration of the United States), the majority of all centrifuge accidents result from user error. These tips from OHAUS will help to ensure safe operation. Before operation:
• Make sure centrifuge bowls and tubes are dry and the spindle is clean.
• Do not overfill tubes or containers.
• Use safety cups/buckets to contain potential spills and prevent aerosol leakage.
• Ensure that the rotor is properly seated on the drive shaft.
• Make sure that tubes or containers are properly balanced in the rotor.

• Only check O-rings on the rotor if you are properly trained.
• Follow the manufacturer’s guidelines for maintenance and operation. During operation:
• Never exceed the rotor’s maximum run speed.
• Keep the centrifuge lid closed.
• Check that the centrifuge is operating normally before leaving the area.
• Do not open the lid until the rotor has come to a complete stop.
• If a spill occurs, use appropriate decontamination and clean-up procedures; use tweezers to remove broken glass.
After operation:
• Clean rotors and accessories using neutral cleaning solutions and a soft cloth.
• Do periodic visual inspections of rotors to check for residue and corrosion.

• Inspect the rotor for cracks or nicks and replace rotors that show any signs of wear or damage.
To ensure safe operation, reliability and long life of your centrifuge, it is essential to take proper care of the unit and rotors between usage. For maintenance tips, refer to the OHAUS Centrifuge and Rotor Maintenance & Cleaning Guide.
Safe, reliable centrifuges
OHAUS Frontier centrifuges are designed for safe use and ease of operation with a range of smart features. The series covers a range of sizes and features to suit basic and advanced centrifugation applications. To determine which model will best suit your needs, refer to the OHAUS Frontier 5000 Rotor Guide.
Proteintech’s FlexAble Oligo-Ready Labeling Kits are used for generating Oligo-FlexLinker conjugates, which enable species-specific and high-affinity labelling of antibodies, providing a fast, easy way to label primary antibodies with Oligos.
The kits are designed to offer maximum flexibility, allowing users to label only what they really need. There is no need to label 100 µg of antibody even though atypical immunofluorescence (or other) workflow may only use a few micrograms. With the FlexAble Oligo-Ready kit, users gain full flexibility: prepare FlexLinkers, select the antibodies the user wants to modify, and label as little as 0.5 µg of antibody.
The innovative FlexAble technology allows for easy multiplexing, even with same species antibodies.
It’s also designed to make it simple for the user to build their own barcode library. A single FlexAble Oligo-Ready Antibody Labeling Kit allows the user to prepare up to three Oligo-FlexLinkers with three different oligos, enabling the user to create their own custom barcode library. The resulting OligoFlexLinkers are stable and can be stored long-term at −20°C. From there, labelling antibodies with the user’s barcode takes only 15 minutes.
For further information, contact United Bioresearch Products, which distributes the full range of Proteintech products in Australia.
United Bioresearch Products Pty Ltd www.unitedbioresearch.com.au
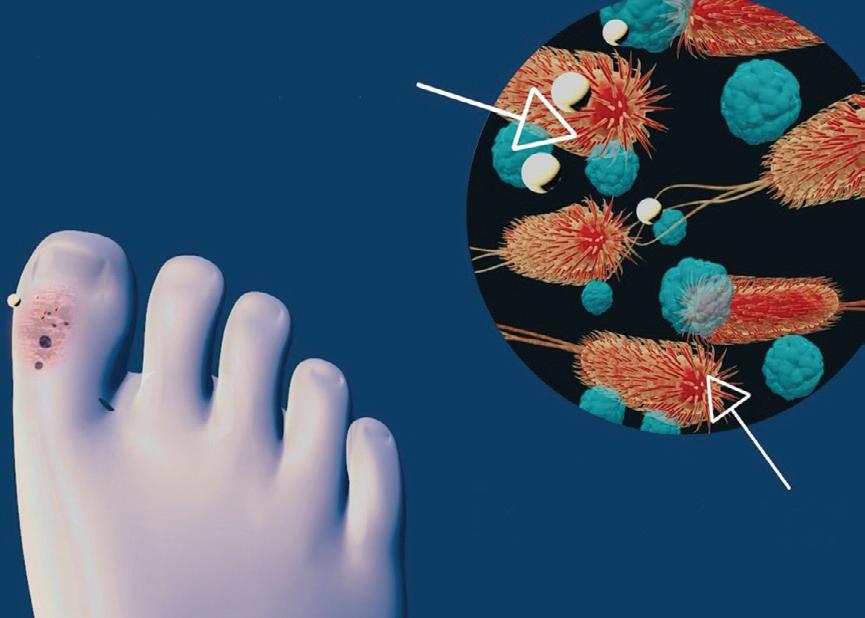
Adding microscopic particles to vinegar can make them more effective against dangerous bacterial infections — and the combination could help combat antibiotic resistance.
Researchers from QIMR Berghofer, Flinders University and the University of Bergen have found that adding microscopic particles to vinegar can make them more effective against dangerous bacterial infections, with hopes that the combination could help combat antibiotic resistance. The team’s findings have been published in the journal ACS Nano
Top arrow points to vinegar and cobalt-containing nanoparticles. Bottom arrow points to the wound bacteria.
Wounds that do not heal are often caused by bacterial infections and are particularly dangerous for the elderly and people with diabetes, cancer and other conditions. And while acetic acid (more commonly known as vinegar) has been used for centuries as a disinfectant, it is only effective against a small number of bacteria and it does not kill the most dangerous types.
The research team has now managed to boost the natural bacterial-killing qualities of vinegar by adding antimicrobial nanoparticles made from carbon and cobalt. Molecular biologists Dr Adam Truskewycz and Professor Nils Halberg, both from the University of Bergen, found these particles could kill several dangerous bacterial species, and their activity was enhanced when added to a weak vinegar solution.
As part of the study, Truskewycz and Halberg added cobalt-containing carbon quantum dot nanoparticles to weak acetic acid to create a potent antimicrobial treatment. They used this mixture against several pathogenic species, including the drug-resistant Staphylococcus aureus, Escherichia coli and Enterococcus faecalis
Truskewycz, who is also based at Flinders University, said the acidic environment from the vinegar made bacterial cells swell and take up the nanoparticle treatment.
“Once exposed, the nanoparticles appear to attack dangerous bacteria from both inside the bacterial cell and also on its surface, causing them to burst,” he said. “Importantly, this approach is non-toxic to human cells and was shown to remove bacterial infections from mice wounds without affecting healing.”
Halberg, who is also based at QIMR Berghofer, said the study shows how nanoparticles could be used to increase the effectiveness of traditional bacterial treatments.
“Combination treatments such as the ones highlighted in this study may help to curb antimicrobial resistance,” he said. “Given this issue can kill up to 5 million people each year, it’s vital we look to find new ways of killing pathogens like viruses, bacteria and fungi or parasites.”

The Azure 300 from Azure Biosystems is a versatile multichannel imager designed for fluorescence, visible light and UV detection. Fully upgradeable to a fluorescent western blot imaging system, it has been designed to combine the speed and simplicity of film-based detection with the precision, resolution and quantitative capabilities of digital imaging.
The system enables rapid chemiluminescent detection with sensitivity, dynamic range and linearity sufficient for rigorous quantitative blot analysis, replacing traditional darkroom workflows while delivering reproducible results. Digital acquisition provides sensitivity comparable to film while eliminating the associated time and handling challenges: simply place a blot in the system and capture high-quality images with a single click.
The product features an intuitive touchscreen interface that provides complete control over imaging settings, including exposure time, focus, lens aperture, distance to the camera and image overlays. Researchers can use predefined protocols for standard detection methods or create custom protocols tailored to specific experimental requirements. Fluorescent imaging capabilities can be added as research needs evolve; when equipped with the green channel, the Azure 300Q is compatible with total protein stains such as TotalStain Q, AzureRed and Ponceau, which should facilitate accurate normalisation and quantitative analysis.
Measuring just 42 x 56 x 33 cm, thus saving valuable bench space, the compact system offers flexible connectivity through WiFi, Ethernet or USB and is fully compatible with Microsoft Windows 11. High-resolution CCD cameras, fast lens options, adjustable illumination and pre-calibrated auto-focus enable high-quality imaging across fluorescence, chemiluminescence, UV, visible and true-colour applications, achieving a dynamic range of 4.8 OD.
By streamlining imaging workflows, accelerating data acquisition, and providing the precision and flexibility required for modern molecular biology research, the product supports publication-quality results and reproducible quantitative analysis.
SciTech Pty Ltd www.scitech.com.au
Bacterial extracellular vesicles (bEVs) are critical for understanding microbial communication, antibiotic-resistance transfer and host–pathogen interactions. However, isolating EVs from bacterial culture media has remained technically challenging, typically requiring ultracentrifugation or precipitation methods that are time-consuming, low-yield and difficult to standardise.
Norgen Biotek, considered a leader in EV isolation from bodily fluids such as urine, blood and saliva, has now released its first commercially available kit specifically designed to isolate extracellular vesicles from bacterial samples. The Bacterial EV Isolation Kit enables researchers to isolate extracellular vesicles directly from bacterial culture media using a purpose-built workflow that eliminates the need to adapt mammalian EV protocols.
Powered by Norgen’s silicon carbide resin technology, the kit rapidly purifies extracellular vesicles ranging from 30 to 400 nm using a streamlined slurry-based method that requires no ultracentrifugation. Its flexible workflow accommodates input volumes between 5 and 35 mL, supporting stable performance across sample volumes and enabling researchers to increase EV and RNA yield simply by processing more starting material.
Purified vesicles are suitable for various downstream molecular applications, including real-time PCR and NGS when paired with Norgen’s Exosomal RNA Isolation Kit for EV RNA purification. This workflow provides consistent results, high EV and RNA yields, and detection of bacterial EV-sized particles. Overall, the kit provides researchers with an accessible and reproducible platform for advancing microbiome research, host–pathogen studies, and diagnostic applications.
Millennium Science Pty Ltd www.mscience.com.au
Richard Stone*
Science journalists spend a lot of time explaining work that is still in a position where it can be challenged as part of the scientific process, often to audiences who expect neat conclusions. That tension shapes how reporting actually happens, and it quietly shapes what journalists expect from the scientists they speak to. Here, the founder of a science PR agency outlines what those expectations look like in practice.
Anyone who has been through peer review knows that the most useful comments are rarely the most comfortable. They focus on what still needs explaining, often circling places where confidence has crept ahead of the evidence. Science journalists tend to approach spokespeople in much the same way, not as critics looking for faults, but as readers trying to understand what holds up once the work is examined closely — as illustrated by recent coverage in The Guardian questioning how evidence around microplastics is interpreted and presented.1

Peer review is often treated as the end of the story, but for journalists it is usually the point where the story becomes possible.
You can usually tell within the first few minutes how the conversation is going to go. Journalists listen out for how a scientist talks about their own work when it’s placed alongside what came before it, and alongside the probability of future critique, corrigendum or even retraction, and whether they seem at ease admitting what still isn’t clear. When that understanding comes through naturally, it gives a reporter enough confidence to keep moving, rather than stopping to double-check every claim later.
That moment often comes when a journalist needs to slow the conversation down, not to challenge the work, but to make sure they understand it well enough to explain it accurately.
Explanation, after all, is not the same as simplification. Journalists tend to expect scientific
spokespeople to recognise that distinction, because it shapes whether a piece helps readers understand the work, or merely compresses it into something thinner than it should be.
That same instinct carries through once the interview ends. Journalists often come back with a short follow-up email, sometimes asking for a source, sometimes asking for a clarification that feels obvious to the scientist who wrote the article or paper or made the product claim. Those questions tend to surface when a journalist is thinking ahead to how a statement will be read once it is published.
Journalists also expect scientists to be able to talk about uncertainty without trying to smooth it away. As Ed Yong has written for The Atlantic, “uncertainty is not a flaw in science. It

is a fundamental feature of it”.2 What matters in practice is whether a scientist can explain that uncertainty clearly enough for a reporter to decide how a finding should be framed, rather than leaving that judgement to implication.
Another expectation shows up once the work is public. Journalists assume that findings will be read closely and may be questioned by people who know the field well. A scientist who is comfortable with that level of attention, makes it easier for a reporter to reflect the work accurately without over-defending it on the page.
There is also an expectation around timing that shapes how accurately a story can be reported. Journalists often need to confirm a detail or sense-check an interpretation within a narrow window. When a scientist is available at that
point, it reduces the chance that a reporter has to rely on inference or another source to keep the piece moving. Alternatively, they might simply pull the piece.
Another expectation is that scientists are able to stay close to what the work actually supports. Journalists are often trying to understand where a finding stops being consensus and starts becoming a subject for debate, especially when a result has implications beyond its immediate field. A spokesperson who can hold that line clearly makes it easier to report the work without stretching it into claims it was never designed to carry.
Peer review is often treated as the end of the story, but for journalists it is usually the point where the story becomes possible. Reporting sits alongside that process, not after it, which means
conversations with scientists tend to happen while questions are still open rather than once they have been resolved sufficiently to be communicated. What journalists expect from scientific spokespeople is an understanding of that moment, and a willingness to talk about the work as it stands, without pushing it into a certainty it has not yet earned.
*Richard Stone is the founder of science PR agency Stone Junction.
1. Mohdin A. ‘Thursday briefing: is your body really full of microplastics?’ The Guardian. 15 January, 2026. Accessed 29 January, 2026. https://www.theguardian. com/world/2026/jan/15/thursday-briefing-is-yourbody-really-full-of-microplastics
2. Yong E. ‘Why the coronavirus is so confusing,’ The Atlantic. April 29, 2020. Accessed 29 January, 2026. https://www.theatlantic.com/health/archive/2020/04/ pandemic-confusing-uncertainty/610819
TSANZSRS 2026
March 27–31, Perth
The Thoracic Society of Australia and New Zealand and the Australian and New Zealand Society of Respiratory Science invite you to Perth for the 2026 TSANZSRS Annual Scientific Meeting.
The theme for The Thoracic Society of Australia and New Zealand 2026 Annual Scientific Meeting is ‘Lungs Under the Microscope’. This theme delves into the intricate landscape of pulmonary science, revealing the lung’s complexities at all levels — from cellular and molecular through to daily functioning.
The theme for the Australian and New Zealand Society of Respiratory Science 2026 Annual Scientific Meeting is ‘Breathing Through the Ages: From Infancy to Late Adulthood’. This theme will explore lung health across the lifespan, examining how lung function changes in response to aging, environment and respiratory disease. tsanzsrsasm.com
World Science Festival Brisbane 2026
March 20–29, Brisbane worldsciencefestival.com.au
Science Meets Parliament 2026 March 25–26, Canberra scienceandtechnologyaustralia.org.au/sciencemeets-parliament-2026
TSANZSRS 2026
March 27–31, Perth tsanzsrsasm.com
Quantum Australia Conference 2026 April 29–30, Adelaide www.qac2026.com/quantum-australia-2026
AusMedtech 2026
May 19–21, Perth www.ausmedtech.com.au
ASID Annual Scientific Meeting 2026
May 27–30, Hobart www.asidasm.com.au
ASM National Meeting 2026
June 15–18, Melbourne www.theasmmeeting.org.au
2026 RACI National Congress July 5–10, Perth www.racicongress.org.au
Accreditation Matters 2026 July 7–8, Melbourne nata.com.au
74th CSANZ Annual Scientific Meeting August 6–9, Sydney www.csanzasm.com
National Science Week 2026 August 15–23, Australia-wide www.scienceweek.net.au
Science at the Shine Dome 2026 September 15–17, Canberra www.science.org.au/news-and-events/events/ science-shine-dome/science-at-the-shine-dome-2026
AIMS National Scientific Meeting 2026 September 16–18, Perth aimsnsm2026.com
ComBio 2026 September 29–October 1, Sydney www.combio.org.au
Westwick-Farrow Media
A.B.N. 22 152 305 336 www.wfmedia.com.au
Head Office
Unit 5, 6-8 Byfield Street, (Locked Bag 2226) North Ryde BC NSW 1670, AUSTRALIA Ph: +61 2 9168 2500
Editor
Dr Joseph Brennan, PhD Interim Editor, Lab+Life Scientist LLS@wfmedia.com.au
Publishing Director/MD Janice Williams
Art Director/Production Manager Linda Klobusiak
Circulation Alex Dalland circulation@wfmedia.com.au
Copy Control Ashna Mehta copy@wfmedia.com.au
Advertising Sales
Sales Manager: Kerrie Robinson Ph:0400 886 311 krobinson@wfmedia.com.au
Tim Thompson Ph: 0421 623 958 tthompson@wfmedia.com.au
If you have any queries regarding our privacy policy please email privacy@wfmedia.com.au