


Volume 10, Number 1, February 2026




Volume 10, Number 1, February 2026

Clinicopathological Cases from the University of Maryland
1 70-year-old Woman with Chest Tightness and Shortness of Breath
RE Dunn, B Klucher, LJ Bontempo, JD Gatz
Case Report
7 Heed The Warning: A Case Report on Capsular Warning Syndrome
T Tarahomi, S Serio, AJ Scumpia
10 Amphetamine-induced Reverse Takotsubo Cardiomyopathy and Thrombosis: A Case Report
A Rabil, O Fakhereddine, A Rebeiz, T El Zahran
16 Sonographic Evaluation of an Inguinal Bubo from Bartonella henselae: A Case Report
JC Luna, RW Shaffe
20 Diagnosing Temporal Lobe Epilepsy in the Emergency Department: A Case Report
A Felker, J Tanner
24 Potassium Overdose in Patient with Chronic Kidney Disease on Losartan: A Case Report
A Naseem, M Schoenborn, J Scheidler, WH Barclay, G Volk
28 Pediatric Pisiform Dislocation: A Case Report
J Wahhab, IV Jeya, JR Huttner
31 Insect in the Ear—Response and Treatment of an Uncommon Prehospital Emergency: A Case Report
C Bashline, M Jester, C Morris
35 Ewing Sarcoma in the Cervical Spine Causing Left Lower Extremity Hemiparesis and Left
Upper Extremity Hemiplegia: A Case Report
A Adler, A Messman



Contents continued on page iii





About Us: Penn State Health is a multi-hospital health system serving patients and communities across central Pennsylvania. We are the only medical facility in Pennsylvania to be accredited as a Level I pediatric trauma center and Level I adult trauma center. The system includes Penn State Health Milton S. Hershey Medical Center, Penn State Health Children’s Hospital and Penn State Cancer Institute based in Hershey, Pa.; Penn State Health Hampden Medical Center in Enola, Pa.; Penn State Health Holy Spirit Medical Center in Camp Hill, Pa.; Penn State Health Lancaster Medical Center in Lancaster, Pa.; Penn State Health St. Joseph Medical Center in Reading, Pa.; Pennsylvania Psychiatric Institute, a specialty provider of inpatient and outpatient behavioral health services, in Harrisburg, Pa.; and 2,450+ physicians and direct care providers at 225 outpatient practices. Additionally, the system jointly operates various healthcare providers, including Penn State Health Rehabilitation Hospital, Hershey Outpatient Surgery Center and Hershey Endoscopy Center.
We foster a collaborative environment rich with diversity, share a passion for patient care, and have a space for those who share our spark of innovative research interests. Our health system is expanding and we have opportunities in both academic hospital as well community hospital settings.
Benefit highlights include:
• Competitive salary with sign-on bonus
• Comprehensive benefits and retirement package
• Relocation assistance & CME allowance
• Attractive neighborhoods in scenic central Pennsylvania



Indexed in PubMed and full text in PubMed Central
Rick A. McPheeters, DO, Editor-in-Chief Kern Medical/UCLA- Bakersfield, California
R. Gentry Wilkerson, MD, Deputy Editor University of Maryland School of Medicine
Mark I. Langdorf, MD, MHPE, Senior Associate Editor
University of California, Irvine School of Medicine- Irvine, California
Shahram Loftipour, MD, MPH, Senior Associate Editor
University of California, Irvine School of Medicine- Irvine, California
Shadi Lahham, MD, MS, Associate Editor Kaiser Permanente- Orange County, California
John Ashurst, DO, Decision Editor/ ACOEP Guest Editor Kingman Regional Health Network, Arizona
Anna McFarlin, MD, Decision Editor
Louisiana State University Health Science Center- New Orleans, Louisiana
Lev Libet, MD, Decision Editor Kern Medical/UCLA- Bakersfield, California
J. David Gatz, MD, Decision Editor University of Maryland School of Medicine, Maryland
Ezhilkugan Ganessane, MD, Decision Editor All India Institute of Medical Sciences (AIIMS) Madurai, India
Ryan Ley, MD, MBA, MS, Decision Editor Arrowhead Regional Medical Center, California
Amin A. Kazzi, MD, MAAEM
The American University of Beirut, Beirut, Lebanon
Anwar Al-Awadhi, MD
Mubarak Al-Kabeer Hospital, Jabriya, Kuwait
Arif A. Cevik, MD
United Arab Emirates University College of Medicine and Health Sciences, Al Ain, United Arab Emirates
Abhinandan A.Desai, MD
University of Bombay Grant Medical College, Bombay, India
Bandr Mzahim, MD
King Fahad Medical City, Riyadh, Saudi Arabia
Barry E. Brenner, MD, MPH Case Western Reserve University
Brent King, MD, MMM University of Texas, Houston
Daniel J. Dire, MD University of Texas Health Sciences Center San Antonio
David F.M. Brown, MD
Massachusetts General Hospital/Harvard Medical School
Amal Khalil, MBA
Edward Michelson, MD Texas Tech University
Edward Panacek, MD, MPH University of South Alabama
Erik D. Barton, MD, MBA Icahn School of Medicine, Mount Sinai, New York
Francesco Dellacorte, MD
Azienda Ospedaliera Universitaria “Maggiore della Carità,” Novara, Italy
Francis Counselman, MD
Eastern Virginia Medical School
Gayle Galleta, MD
Sørlandet Sykehus HF, Akershus Universitetssykehus, Lorenskog, Norway
Hjalti Björnsson, MD Icelandic Society of Emergency Medicine
Jacob (Kobi) Peleg, PhD, MPH Tel-Aviv University, Tel-Aviv, Israel
Jonathan Olshaker, MD Boston University
Katsuhiro Kanemaru, MD University of Miyazaki Hospital, Miyazaki, Japan
UC Irvine Health School of Medicine
Elena Lopez-Gusman, JD
California ACEP
American College of Emergency Physicians
DeAnna McNett, CAE
American College of Osteopathic Emergency Physicians
John B. Christensen, MD
California Chapter Division of AAEM
Randy Young, MD
California ACEP
American College of Emergency Physicians
Mark I. Langdorf, MD, MHPE
UC Irvine Health School of Medicine
Jorge Fernandez, MD
California ACEP
American College of Emergency Physicians University of California, San Diego
Peter A. Bell, DO, MBA
American College of Osteopathic Emergency Physicians
Baptist Health Science University
Robert Suter, DO, MHA
American College of Osteopathic Emergency
Physicians UT Southwestern Medical Center
Shahram Lotfipour, MD, MPH UC Irvine Health School of Medicine
Brian Potts, MD, MBA
California Chapter Division of AAEM
Alta Bates Summit-Berkeley Campus
Christopher Sampson, MD, Decision Editor University of Missouri- Columbia, Missouri
Joel Moll, MD, Decision Editor Virginia Commonwealth University School of Medicine- Richmond, Virginia
Alexander Scumpia, DO, MHA, MSc, Decision Editor HCA FL Aventura Hospital Florida
Steven Walsh, MD, Decision Editor Einstein Medical Center Philadelphia-Philadelphia, Pennsylvania
Melanie Heniff, MD, JD, Decision Editor University of Indiana School of Medicine- Indianapolis, Indiana
Austin Smith, MD, Decision Editor Vanderbilt University Medical Center-Nashville, Tennessee
Rachel A. Lindor, MD, JD, Decision Editor Mayo Clinic College of Medicine and Science, Minnesota
Jacqueline K. Le, MD, Decision Editor Desert Regional Medical Center
Arihant Jain, MD, Decision Editor All India Institute of Medical Sciences (AIIMS), New Delhi
Christopher San Miguel, MD, Decision Editor Ohio State Univesity Wexner Medical Center
Grace Hickam, MD, MEd, Decision Editor Virginia Commonwealth University, Virginia
Robert Suter, DO, MHA UT Southwestern Medical Center
Khrongwong Musikatavorn, MD King Chulalongkorn Memorial Hospital, Chulalongkorn University, Bangkok, Thailand
Leslie Zun, MD, MBA Chicago Medical School
Linda S. Murphy, MLIS University of California, Irvine School of Medicine Librarian
Nadeem Qureshi, MD St. Louis University, USA Emirates Society of Emergency Medicine, United Arab Emirates
Niels K. Rathlev, MD Tufts University School of Medicine
Pablo Aguilera Fuenzalida, MD Pontificia Universidad Catolica de Chile, Región Metropolitana, Chile
Peter A. Bell, DO, MBA Baptist Health Science University
Peter Sokolove, MD University of California, San Francisco
Robert M. Rodriguez, MD University of California, San Francisco
Robert W. Derlet, MD University of California, Davis
Rosidah Ibrahim, MD Hospital Serdang, Selangor, Malaysia
Samuel J. Stratton, MD, MPH Orange County, CA, EMS Agency
Scott Rudkin, MD, MBA University of California, Irvine
Scott Zeller, MD University of California, Riverside
Steven Gabaeff, MD Clinical Forensic Medicine
Steven H. Lim, MD
Changi General Hospital, Simei, Singapore
Terry Mulligan, DO, MPH, FIFEM ACEP Ambassador to the Netherlands Society of Emergency Physicians
Vijay Gautam, MBBS University of London, London, England
Wirachin Hoonpongsimanont, MD, MSBATS Siriraj Hospital, Mahidol University, Bangkok, Thailand
Ian Oliffe, BS Executive Editorial Director
Sheyda Aquino, BS WestJEM Editorial Director
Tran Nguyen, BS CPC-EM Editorial Director
Stephanie Burmeister, MLIS WestJEM Staff Liaison
Cassandra Saucedo, MS Executive Publishing Director
Isabella Choi, BS WestJEM Publishing Director
Alyson Tsai, BS CPC-EM Publishing Director
June Casey, BA Copy Editor
Official Journal of the California Chapter of the American College of Emergency Physicians, the America College of Osteopathic Emergency Physicians, and the California Chapter of the American Academy of Emergency Medicine



Available in MEDLINE, PubMed, PubMed Central, Google Scholar, eScholarship, DOAJ, and OASPA.

Editorial and Publishing Office: WestJEM/Depatment of Emergency Medicine, UC Irvine Health, 3800 W. Chapman Ave. Suite 3200, Orange, CA 92868, USA Office: 1-714-456-6389; Email: Editor@westjem.org
No. 1: February 2026 i Clinical Practice and Cases in Emergency
Indexed in PubMed and full text in PubMed Central
This open access publication would not be possible without the generous and continual financial support of our society sponsors, department and chapter subscribers.
Professional Society Sponsors
American College of Osteopathic Emergency Physicians
California ACEP
Academic Department of Emergency Medicine Subscribers
Albany Medical College Albany, NY
American University of Beirut Beirut, Lebanon
Arrowhead Regional Medical Center Colton, CA
Augusta University Augusta GA
Baystate Medical Center Springfield, MA
Beaumont Hospital Royal Oak, MI
Beth Israel Deaconess Medical Center Boston, MA
Boston Medical Center Boston, MA
Brigham and Women’s Hospital Boston, MA
Brown University Providence, RI
Carl R. Darnall Army Medical Center Fort Hood, TX
Conemaugh Memorial Medical Center Johnstown, PA
Desert Regional Medical Center Palm Springs, CA
Doctors Hospital/Ohio Health Columbus, OH
Eastern Virginia Medical School Norfolk, VA
Einstein Healthcare Network Philadelphia, PA
Emory University Atlanta, GA
Genesys Regional Medical Center Grand Blanc, Michigan
Hartford Hospital Hartford, CT
Hennepin County Medical Center Minneapolis, MN
Henry Ford Hospital Detroit, MI
State Chapter Subscribers
Arizona Chapter Division of the American Academy of Emergency Medicine
California Chapter Division of the American Academy of Emergency Medicine
Florida Chapter Division of the American Academy of Emergency Medicine
International Society Partners
INTEGRIS Health
Oklahoma City, OK
Kaweah Delta Health Care District Visalia, CA
Kennedy University Hospitals Turnersville, NJ
Kern Medical Bakersfield, CA
Lakeland HealthCare
St. Joseph, MI
Lehigh Valley Hospital and Health Network Allentown, PA
Loma Linda University Medical Center Loma Linda, CA
Louisiana State University Health Sciences Center New Orleans, LA
Madigan Army Medical Center Tacoma, WA
Maimonides Medical Center Brooklyn, NY
Maricopa Medical Center Phoenix, AZ
Massachusetts General Hospital Boston, MA
Mayo Clinic College of Medicine Rochester, MN
Mt. Sinai Medical Center Miami Beach, FL
North Shore University Hospital Manhasset, NY
Northwestern Medical Group Chicago, IL
Ohio State University Medical Center Columbus, OH
Ohio Valley Medical Center Wheeling, WV
Oregon Health and Science University Portland, OR
Penn State Milton S. Hershey Medical Center Hershey, PA
Presence Resurrection Medical Center Chicago, IL
California Chapter Division of AmericanAcademy of Emergency Medicine
Robert Wood Johnson University Hospital New Brunswick, NJ
Rush University Medical Center Chicago, IL
Southern Illinois University Carbondale, IL
St. Luke’s University Health Network Bethlehem, PA
Stanford/Kaiser Emergency Medicine Residency Program Stanford, CA
Staten Island University Hospital Staten Island, NY
SUNY Upstate Medical University Syracuse, NY
Temple University Philadelphia, PA
Texas Tech University Health Sciences Center El Paso, TX
University of Alabama, Birmingham Birmingham, AL
University of Arkansas for Medical Sciences Little Rock, AR
University of California, Davis Medical Center Sacramento, CA
University of California Irvine Orange, CA
University of California, Los Angeles Los Angeles, CA
University of California, San Diego La Jolla, CA
University of California, San Francisco San Francisco, CA
UCSF Fresno Center Fresno, CA
University of Chicago, Chicago, IL
University of Colorado, Denver Denver, CO
University of Florida Gainesville, FL
University of Florida, Jacksonville Jacksonville, FL
University of Illinois at Chicago Chicago, IL
University of Illinois College of Medicine Peoria, IL
University of Iowa Iowa City, IA
University of Louisville Louisville, KY
University of Maryland Baltimore, MD
University of Michigan Ann Arbor, MI
University of Missouri, Columbia Columbia, MO
University of Nebraska Medical Center Omaha, NE
University of South Alabama Mobile, AL
University of Southern California/Keck School of Medicine Los Angeles, CA
University of Tennessee, Memphis Memphis, TN
University of Texas, Houston Houston, TX
University of Texas Health San Antonio, TX
University of Warwick Library Coventry, United Kingdom
University of Washington Seattle, WA
University of Wisconsin Hospitals and Clinics Madison, WI
Wake Forest University Winston-Salem, NC
Wright State University Dayton, OH
Uniformed Services Chapter Division of the American Academy of Emergency Medicine
Virginia Chapter Division of the American Academy of Emergency Medicine
To become a WestJEM departmental sponsor, waive article processing fee, receive print and copies for all faculty and electronic for faculty/residents, and free CME and faculty/fellow position advertisement space, please go to http://westjem.com/subscribe or contact: Emergency Medicine Association of Turkey Lebanese Academy of Emergency Medicine MediterraneanAcademyofEmergencyMedicine
Stephanie Burmeister
WestJEM Staff Liaison
Phone: 1-800-884-2236
Email: sales@westjem.org
Sociedad Chileno Medicina Urgencia ThaiAssociationforEmergencyMedicine
Indexed in PubMed and full text in PubMed Central
Clinical Practice and Cases in Emergency Medicine (CPC-EM) is a MEDLINE-indexed internationally recognized journal affiliated with the Western Journal of Emergency Medicine (WestJEM). It offers the latest in patient care case reports, images in the field of emergency medicine and state of the art clinicopathological and medicolegal cases. CPC-EM is fully open-access, peer reviewed, well indexed and available anywhere with an internet connection. CPC-EM encourages submissions from junior authors, established faculty, and residents of established and developing emergency medicine programs throughout the world.
of Contents continued
39 Implanted in the Scar: A Case Report of Diagnosis and Management of Cesarean Scar Ectopic Pregnancy
H Schindler, L Keeler
43 Phantom Arm Pain and Tinnitus in a Patient with ST-Segment Elevation Myocardial Infarction: A Case Report
J Porter, CP Liedl, KP Steever, HD Lumby, CM Hanson, HR Vaden, AB Klassen
46 A Case Report of Acute Lymphoblastic Leukemia Presenting as Bilateral Knee Pain in a Healthy Runner
M Rosselli, A Sanoja, M Apicella
50 Acute Hypercapnic Respiratory Failure from Foreign Body Aspiration in a 16-Month-Old: A Case Report
S Lee, RD Shin, K Combs
55 Acute Aortic Dissection Masquerading as Testicular Torsion: A Case Report
BM Lo, MK Christensen, CE Byrns, B Chidester
59 A Case Report of Milk-Alkali Syndrome Secondary to Excessive Antacid Use
S Beso, L Abubshait
63 When STEMI Isn’t STEMI: Cardiac Arrest from Aortic Valve Papillary Fibroelastoma – A Case Report
M Kanaa, S Abbastanira, E Elameen, S Alsultan
68 Case Report: Pediatric Hallucinations and Anti-Neuronal Intermediate Filament Autoimmune Encephalitis
AH Bjornstad, N Oberhauser-Lim, T Phan, EJ Samones, T Young
72 Spontaneous Splenic Vein Rupture: Case Report of a Rare Presentation
B Franco, A Karski, K Boehm
76 Antimuscarinic Toxicity Safely Managed with High-Dose Transdermal Rivastigmine: A Case Report
CJ Watson, EM Burrill, WS Jaffee
81 Syncope in a Patient with Right Ventricular Compression from Severe Pectus Excavatum: A Case Report
MJ Christensen, J Foti
Policies for peer review, author instructions, conflicts of interest and human and animal subjects protections can be found online at www.cpcem.org.
Indexed in PubMed and full text in PubMed Central
Clinical Practice and Cases in Emergency Medicine (CPC-EM) is a MEDLINE-indexed internationally recognized journal affiliated with the Western Journal of Emergency Medicine (WestJEM). It offers the latest in patient care case reports, images in the field of emergency medicine and state of the art clinicopathological and medicolegal cases. CPC-EM is fully open-access, peer reviewed, well indexed and available anywhere with an internet connection. CPC-EM encourages submissions from junior authors, established faculty, and residents of established and developing emergency medicine programs throughout the world.
85 Medial Clavicle Physeal Fracture in a 15-Year-Old Male: A Case Report R Kofman, R Allen, AB Smartt, KM Drechsel, JR Pollock, D Rappaport
89 Carotidynia—A Rare Cause of Anterior Neck Pain: Case Report F Ibu, O Keller-Baruch, J Pelletier
93 Pleural and Pericardial Effusions Associated with Semaglutide: A Case Report M Stark, N Valentini
97 Recurrent Miller Fisher Syndrome: A Case Report l Vernier, GB Gilmore, K Van Housen, XC Zhang
Images in Emergency Medicine
101 Diagnosis of Bilateral Quadriceps Tendon Rupture Using Point-of-Care Ultrasound E Guo, A Duran, A Kuc, AB Cheng
Letter to the Editor
104 Syncope in a Patient with Takotsubo Syndrome: Additional Issues to Consider J Madias
Policies for peer review, author instructions, conflicts of interest and human and animal subjects protections can be found online at www.cpcem.org.

Robert E Dunn, MD*
Brianna Klucher, MD†
Laura J Bontempo, MD, MEd† J David Gatz, MD†
Section Editor: Joel Moll, MD
University of Maryland Medical Center, Department of Emergency Medicine, Baltimore, MD
University of Maryland School of Medicine, Department of Emergency Medicine, Baltimore, MD
Submission history: Submitted March 22, 2025; Revision received June 7, 2025; Accepted July 11, 2025
Electronically published December 7, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.47061
Chest tightness and shortness of breath are relatively common reasons for presentation to the emergency department (ED), often triggering protocolized workups and dispositions. A good history, however, can reveal additional elements that may dramatically alter the differential diagnosis and management. A 70-year-old woman presented to the ED complaining of subacute chest tightness with dyspnea on exertion. This case offers a thoughtful analysis of how to integrate key findings within a patient’s history, exam, and workup in the ED. The surprising final diagnosis and case outcome are then revealed. [Clin Pract Cases Emerg Med. 2025;10(1):1-6.]
A 70-year-old woman was brought into the emergency department (ED) by her family for intermittent chest tightness over the prior one month. The chest discomfort was nonradiating, but worsened with exertion, and was associated with fatigue and dyspnea on exertion. Her daughter reported that the patient’s heart rate had been bradycardic to 40 beats per minute (bpm) for a few months. On a review of systems, the patient endorsed decreased appetite, generalized abdominal pain, diarrhea, bilateral leg and back pain for months, and generalized weakness and fatigue.
Her medical history was notable for heart failure with reduced ejection fraction (HFrEF) of 20-25%, non-ischemic cardiomyopathy, coronary artery disease, atrial fibrillation, and dyslipidemia. Her home medications included atorvastatin, dabigatran, empagliflozin, famotidine, furosemide, metoprolol succinate, and sacubitril/valsartan. Her son noted the patient had been non-compliant with dabigatran for an unknown amount of time. Family history was significant for cardiomyopathy. The patient lived with her daughter, was a former smoker, and denied using drugs or alcohol. She had moved from Venezuela one year earlier.
On initial presentation, the patient’s vital signs were as follows: temperature, 36.6 ˚C; heart rate, 40 bpm; blood pressure, 103/54 millimeters of mercury; respiratory rate, 14 breaths per minute; and oxygen saturation, 99% on room air.
Her estimated body mass index was 20 kilograms (k) per square meter (reference range 18.5-24.9 k). On examination, the patient was well appearing and not in acute distress. Her head was normocephalic. Her neck had full range of motion and no jugular venous distension. Her heart had a regular but bradycardic rhythm. A soft gallop was heard on auscultation, but no other abnormal heart sounds were heard. Her lungs were clear to auscultation bilaterally, with some decreased air movement at the bases. On abdominal exam, she had right upper quadrant abdominal tenderness without a Murphy sign. The abdomen was otherwise soft without rebound tenderness or guarding. Examination of the extremities showed no edema. She appeared to be grossly neurologically intact, with appropriate strength in all extremities.
Laboratory studies were completed (Table 1). The complete blood count was notable for a low hemoglobin and hematocrit. The basic metabolic panel was notable for an elevated blood urea nitrogen (BUN) and creatinine (Cr). Liver function tests were notable for an elevated aspartate aminotransferase, elevated alanine aminotransferase level, and an elevated alkaline phosphatase with normal total bilirubin. Coagulation factors were notable for elevated prothrombin (PT) and activated partial thromboplastin times (aPTT), and an elevated international normalized ratio. An electrocardiogram (ECG) (Image 1), chest radiograph (CXR) (Image 2), and right upper quadrant ultrasound (Image 3) were
70-year-old Woman with Chest Tightness and Shortness of Breath
obtained as well.
The patient remained in no distress. Due to her history of heart failure and current presentation with chest pain, a cardiology consult was obtained while pursuing admission to the hospital. A test was subsequently ordered, and a diagnosis was made.
At first glance, this case seems deceptively straightforward. It is tempting to glance at the chief complaint and categorize this patient under the illness script of “older woman with fatigue and chest tightness.” Instinctively, one might begin theorizing whether the urinalysis showing a urinary tract infection or brain-natriuretic peptide indicating a congestive heart failure exacerbation will yield the first abnormal result. After all, how many septuagenarians with similar symptoms do we see every shift?
However, a closer look reveals a more complex scenario: Not only is this older adult with chronic cardiac disease presenting with chest tightness and fatigue, but also with bradycardia and persistent exertional chest pain with shortness of breath. These symptoms have developed over a subacute timeframe. Adding to the puzzle are her constitutional symptoms, generalized weakness and poor appetite, and an array of other complaints, including abdominal discomfort, diarrhea, and muscle aches. The differential diagnosis for this constellation of symptoms is vast, spanning from iatrogenic complications to infectious etiologies and everywhere in between. Her social history of recent immigration and medication nonadherence do not help to simplify this case. Diving into the details is crucial for narrowing down the possibilities and guiding diagnostic testing.
The patient’s physical exam is significant for bradycardia and what may be considered a “soft” blood pressure. She is normothermic. She has a cardiac gallop but otherwise reassuring cardiopulmonary exam including lack of jugular venous distension, basilar crackles, or peripheral edema. Of note, she does have right upper quadrant tenderness.
Her ECG reveals sinus bradycardia with poor R wave progression but without evidence of atrioventricular blocks. There are no prolonged intervals or U waves to suggest electrolyte deficiency as the etiology of her bradycardia. The ECG findings are confirmed by her laboratory values, which demonstrate no significant electrolyte derangements.
These results also steered me away from adrenal insufficiency as an etiology of her fatigue and bradycardia due to the absence of hyponatremia and hyperkalemia. Despite the patient’s cardiac complaints, the troponin level is normal, decreasing immediate concern for an acute injury, such as myocarditis or acute coronary syndrome. Although patients may experience an acute cardiac insult without troponin elevation, a subacute or chronic process with a more insidious onset seems more likely for this patient. She also has a normal white blood cell (WBC) count, although it is worth noting that
in the geriatric population an appropriate immune response may not be mounted to acute infection, leading to falsely reassuring normothermia and normal WBC. Thus, infectious processes must remain in play.
She also has increased PT and aPTT. While coagulation tests are not routinely performed for monitoring of direct oral anticoagulation agents, it is possible for dabigatran to cause elevations in PT and aPTT. It may be that our patient is more adherent to her anticoagulant regimen than reported by her family. Gastrointestinal (GI) bleeding from anticoagulation could lead to anemia, abdominal discomfort, diarrhea, and fatigue. Acute GI bleeding could additionally contribute to increased vagal tone and bradycardic episodes but would not explain her persistent bradycardia. Additionally, this patient’s diarrhea is not described as melena or hematochezia, making a GI bleed a less likely explanation for her symptoms.
The question of adherence to dabigatran could indicate that she has been nonadherent to additional medications and had progression or exacerbation of underlying illness such as her heart failure. Alternatively, incorrect dosing or poor understanding of her medications, such as metoprolol, could have led to bradycardia. This would, however, be less likely to cause right upper quadrant pain or transaminitis, and we are missing other signs of beta blocker toxicity such as hypotension or hypoglycemia.
Regarding her imaging, her CXR revealed cardiomegaly and small pleural effusion but without signs of pneumonia or interstitial edema. This is not particularly surprising in a patient with known non-ischemic cardiomyopathy and reduced EF. The right upper quadrant ultrasound was absent of pericholecystic fluid, sludge or stones, or a dilated common bile duct, which when combined with relatively mild transaminitis, makes acute cholecystitis, cholangitis, or primary biliary disorders, including autoimmune disease, less likely.
In the vein of autoimmunity, systemic lupus erythematosus with cardiomyopathy and nephropathy might explain her cardiac symptoms, kidney injury, muscle aches, and fatigue. That said, new-onset systemic lupus erythematosus in a 70-year-old woman would be unexpected as this disease occurs more commonly in younger patients with a peak incidence in patients in their 20s-30s. Furthermore, she lacks photosensitivity and rash, which are the most common presenting symptoms.
Ultimately, I must synthesize her history, symptoms, and workup thus far into a comprehensive picture. When viewing her presentation globally, it seems her exertional chest tightness/pressure and shortness of breath are most likely due to symptomatic bradycardia caused by subacute progression or a new insult to her chronic cardiac disease including heart failure. Her transaminitis and right upper quadrant pain may then be explained by congestive hepatopathy. This could also explain her kidney injury, with a BUN/Cr ratio > 20 suggesting a prerenal azotemia due to decreased renal perfusion from worsening cardiac disease and/or inadequate
Dunn et al. 70-year-old Woman with Chest Tightness and Shortness of Breath
Table 1. Initial laboratory results of a 70-year-old woman with chest tightness and bradycardia.
Complete Blood Count
Serum Chemistries
Hepatic Studies
Cardiac Studies Troponin I
dL, deciliter; g, gram; K, thousand; L, liter; mcL, microliter; mmol, millimole; mg, milligram; mL, milliliter; ng, nanogram; u, units.
oral intake (normal BUN/Cr ratio is between 10:1-20:1).
Generalized fatigue, poor appetite, and weakness could stem from worsening cardiac disease or as an additional finding of the etiology of her cardiac insult.
Hypothyroidism is common in older women and can contribute to bradycardia, fatigue, and other nonspecific symptoms such as weakness and poor appetite. Hypothyroidism may exacerbate underlying cardiac disease by reducing myocardial contractility and contributing to fluid retention and worsening existing heart failure. I do not yet have a thyroid stimulating hormone available for review; so this could be a reasonable test to consider as I work toward a final diagnosis. It is somewhat reassuring, however, that this patient is not hypothermic or hypotensive, which I might expect to see if she had hypothyroidism severe enough to cause bradycardia and heart failure.
Infiltrative cardiac diseases, such as sarcoidosis or amyloidosis, are important considerations in a patient with bradycardia, fatigue, and heart failure symptoms. These conditions can lead to conduction abnormalities, including bradycardia, as well as progressive myocardial dysfunction due to infiltration or inflammation of the cardiac tissue. These disorders more frequently are associated with diastolic dysfunction and preserved EF, however. This patient also lacks classical findings of these disorders such as perihilar lymphadenopathy in sarcoidosis or hypoalbuminemia and edema suggestive of nephrotic syndrome in amyloidosis. Infectious causes remain a critical final consideration in this patient’s presentation. Infective endocarditis can lead to conduction abnormalities, heart failure, and constitutional complaints like fatigue and poor appetite. However, the absence of fever and a normal WBC count are less typical,
even in older adults who may fail to mount an adequate immune response, and she lacks classic findings of murmur or embolic signs such as Janeway lesions. Other bacterial infections, such as those caused by Legionella or Mycoplasma, or viral illnesses like influenza and cytomegalovirus, fail to explain the full constellation of cardiac and systemic findings and are less consistent with her timeline and lack of respiratory symptoms.
The patient’s origin from Venezuela introduces important endemic infections into the differential diagnosis. Chronic Trypanosoma cruzi infection, the causative agent of Chagas
disease, is particularly relevant. Chagas cardiomyopathy is the most common cause of non-ischemic cardiomyopathy in endemic countries, including Venezuela. Over years to decades, it can progress and result in chronic bradycardia, conduction abnormalities, heart failure symptoms, and gastrointestinal complaints such as abdominal pain and diarrhea.1 Additionally, although less commonly seen, Chagas hepatopathy could explain her transaminitis and right upper quadrant tenderness independently of congestive effects. Other parasitic diseases, such as schistosomiasis, might also explain hepatosplenic involvement, but her findings are more specific for the cardiac involvement in Chagas disease.
Ultimately, I believe the patient’s symptoms align most closely with Chagas cardiomyopathy, a diagnosis that connects her cardiac history, regional background, and current presentation. While acute Chagas disease is typically diagnosed through blood smear or polymerase chain reaction to detect T. cruzi parasitemia, chronic Chagas disease is confirmed through serologic testing to detect antibodies (via enzyme-linked immunosorbent assay [ELISA] or immunofluorescence assay), as parasitemia usually becomes low or undetectable in the chronic phase.2 Cardiac manifestations of Chagas disease make chronic infection more likely in this patient. Given her clinical presentation and history, I would order serologic testing for T. cruzi to confirm a diagnosis of Chagas cardiomyopathy.
Cardiology consult sought to determine the etiology of the patient’s HFrEF. They felt her recent immigration from Venezuela, negative workup from an ischemia standpoint, and partial conduction delay on ECG (ie, QRS duration > 100 milliseconds) were all suspicious for Chagas cardiomyopathy.
An infiltrative process, such as sarcoidosis or amyloidosis, was also considered. The cardiology team specifically noted concern for a “cherry-on-top” pattern on the global longitudinal strain analysis of her echocardiogram, which can by suggestive of cardiac amyloidosis and suggested cardiac magnetic resonance imaging (MRI). A T. cruzi immunoglobulin G (IgG) antibody ultimately confirmed the diagnosis of Chagas cardiomyopathy.
Her inpatient cardiac MRI demonstrated worsening cardiac function. Her previous EF of 20-25% dropped to 16% in the setting of a severely enlarged left ventricle. Her right ventricle was mildly enlarged with moderately depressed systolic function. A thrombus was additionally identified, adhering to the left ventricular apex. The patient received an automatic implantable cardioverter defibrillator and was discharged after optimization on her guideline-directed medical therapy and anticoagulation.
She had one admission approximately two months after her discharge to a nearby hospital for intermittent chest pain and dyspnea that was felt to be from a viral syndrome, rather than a heart failure exacerbation. Following that admission, she had one additional medication refill from cardiology and was subsequently lost to follow-up.
Chagas disease is caused by a T. cruzi infection, which is a protozoan parasite transmitted by the triatomine or reduviid “kissing” bug. The fecal matter of the bug contains a parasite that inoculates the host through bite wounds or intact mucosal membranes. Vertical transmission is also a possibility and can present asymptomatic at birth. Finally, infection can also occur via infected organ transplantation or blood transfusion.3 The disease is named after Dr. Carlos Chagas, a Brazilian physician who first identified the causative agent and insect vector.
Epidemiology
Chagas disease is endemic in 21 continental Latin American countries with an estimated eight million people infected in the Americas, and an estimated 280,000 infected individuals living in the United States. As these bugs hide in thatch roofing and unfinished housing, infection is prevalent in rural areas. It is important to note that short-term travel to these endemic countries is not a risk factor to infection as it requires long-term stay in these rural areas where people are repeatedly bitten or exposed to the triatomine bugs.3
Infection starts with an asymptomatic incubation period of 1-2 weeks. The subsequent acute phase lasts 8-12 weeks and can also be asymptomatic or have non-specific symptoms such as fever, malaise, or anorexia. About 1% of acute phase patients will have complications such as a pericardial effusion
or meningoencephalitis. Once patients enter the chronic phase, after 12 weeks, they can begin to develop heart or GI disease.4 Chagas cardiomyopathy has four major manifestations: heart failure; cardiac arrhythmias; thromboembolism; and chest pain syndrome. The parasite’s presence in the body causes a chronic inflammatory state. In the heart, this leads to fibrosis of the ventricular wall, resulting in dilated cardiomyopathy, valvular regurgitation, and ultimately biventricular failure. Fibrosis can additionally occur in the conduction system, leading to atrioventricular blocks, ventricular rhythms, and other dysrhythmias.5 About 55-65% of deaths in patients with Chagas disease are secondary to sudden cardiac death from a dysrhythmia. The chronic inflammatory state also increases the risk of thromboembolism and strokes, with thrombi developing within the dilated cardiac chambers of the heart.6 Finally, patients can develop a chest pain syndrome that may mimic angina but is suspected to be due to myocardial microvascular abnormalities from the cardiac remodeling from the parasite’s presence.
In the acute phase of Chagas, a diagnosis is made by visualizing trypomastigotes in blood using microscopy. Unfortunately, outside existing screening programs, most patients are diagnosed after the relatively short acute phase. Chronic disease may be identified with IgG antibody testing for T, cruzi. These antibodies develop 2-8 weeks after infection. Due to antigenic diversity of T. cruzi, it is recommended to obtain two serologic tests to achieve adequate sensitivity. Immunoglobin G antibody testing using an ELISA methodology has a 99% sensitivity and 98% specificity in detection.7 Patients at risk of chronic disease should be assessed for potential cardiac complications, including an ECG for conduction abnormalities, CXR for cardiomegaly, and an echocardiogram if there is concern for valvular regurgitation, thrombus, and/or wall motion abnormalities.8
Acute Chagas disease is managed with antitrypanosomal medications. First-line treatment for adults is benznidazole, usually at 5 mg/k daily, administered in two divided doses for 60 days. It is worth noting that use of benznidazole for this purpose in patients > 12 years of age is off label but generally better tolerated than the primary alternative, nifurtimox. The role of antitrypanosomal treatment outside acute, congenital, or reactivated cases of T. cruzi is less clear.9
Historically, treatment of chronic Chagas cardiomyopathy has depended on the severity of the heart failure symptoms. A patient must meet the criteria of the American Heart Association Class A or B or the New York Heart Association Class 0 or 1 to receive antitrypanosomal therapy and find benefit. At this stage, this patient does not have evidence of
70-year-old Woman with Chest Tightness and Shortness of Breath
structural heart disease or heart failure symptoms and, as suggested by a 2015 randomized multicenter trial of benznidazole, will have a reduced incidence of death from cardiovascular events from Chagas cardiomyopathy.10 Antitrypanosomal therapy historically has not been thought to have mortality benefit after progression to symptomatic heart failure, although some authors have recently posited that a greater number of patients may benefit.8,11
All heart failure patients should be started on guidelinedirected medical therapy. Patients should have annual ECGs and cardiology visits. If patients have a normal left ventricular EF, they can usually receive an echocardiogram every 3-5 years, but those with an EF < 50% should receive annual studies. Clinicians can also use the Rassi score to help stratify mortality risk in patients with Chagas disease. The Rassi score is a prognostic assessment that incorporates factors such as the New York Heart Association class, presence of cardiomegaly on CXR, and segmental or global left ventricular systolic dysfunction. Severe cases may require cardiac transplantation. Evidence shows that cardiac transplant recipients have a survival benefit and that reactivation of T. cruzi is a rare cause of death.8
Chagas cardiomyopathy
KEY POINTS
1. Have a high index of suspicion for Chagas disease in a patient with heart failure and a history of residence in rural areas of continental Latin America.
2. Acute Chagas disease and some chronic Chagas cardiomyopathy patients can be treated with antitrypanasomal therapy.
3. As a patient’s Chagas cardiomyopathy progresses, their therapy is consistent with guideline-directed medical therapy including heart transplant, which has been shown to have a mortality benefit.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Dunn et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Echavarría NG, Echeverría LE, Stewart M, Gallego C, Saldarriaga C. Chagas disease: chronic Chagas cardiomyopathy. Curr Probl Cardiol 2021;46(3):100507
2. Schijman AG, Alonso-Padilla J, Britto C, Herrera Bernal CP. Retrospect, advances and challenges in Chagas disease diagnosis: a comprehensive review. Lancet Reg Health - Am. 2024;36:100821.
3. Santos É, Menezes Falcão L. Chagas cardiomyopathy and heart failure: from epidemiology to treatment. Rev Port Cardiol 2020;39(5):279-289
4. Saraiva RM, Mediano MFF, Mendes FS, et al. Chagas heart disease: an overview of diagnosis, manifestations, treatment, and care. World J Cardiol. 2021;13(12):654-675
5. Echeverría LE, Serrano‐García AY, Rojas LZ, Berrios‐Bárcenas EA, Gómez‐Mesa JE, Gómez‐Ochoa SA. Mechanisms behind the high mortality rate in chronic Chagas cardiomyopathy: unmasking a three‐headed monster. Eur J Heart Fail. 2024;26(12):2502-2514.
6. Lage TAR, Tupinambás JT, Pádua LBD, et al. Stroke in Chagas disease: from pathophysiology to clinical practice. Rev Soc Bras Med Trop. 2022;55:e0575-2021.
7. Suescún-Carrero SH, Tadger P, Cuellar CS, et al. Rapid diagnostic tests and ELISA for diagnosing chronic Chagas disease: systematic revision and meta-analysis. PLoS Negl Trop Dis. 2022 Oct 18;16(10):e0010860
8. Llerena-Velastegui J, Lopez-Usina A, Mantilla-Cisneros C. Advances in the understanding and treatment of chronic Chagas cardiomyopathy. Cardiol Res. 2024;15(5):340-349.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Address for Correspondence: J. David Gatz, MD, Department of Emergency Medicine, University of Maryland, 110 S Paca Street, 6th Floor, Suite 200, Baltimore, MD 21201. Email: jgatz@som. umaryland.edu.
9. Bern C. Antitrypanosomal therapy for chronic Chagas’ disease. N Engl J Med. 2011;364(26):2527-2534.
10. Morillo CA, Marin-Neto JA, Avezum A, et al. Randomized trial of benznidazole for chronic Chagas’ cardiomyopathy. N Engl J Med 2015;373(14):1295-1306.
11. Rassi A, Grimshaw A, Sarwal A, et al. Impact of antiparasitic therapy on cardiovascular outcomes in chronic Chagas disease. a systematic review and meta-analysis. eClinicalMedicine. 2025;79:102972
Targol Tarahomi, DO* Sean Serio, MD*†
Alexander John Scumpia, DO, MHA, MSc*
†
HCA Florida Aventura Hospital, Department of Emergency Medicine, Aventura, Florida
Baptist Health Baptist Hospital, Department of Emergency Medicine, Miami, Florida
Section Editor: Melanie Heniff, MD, JD
Submission history: Submitted April 16, 2025; Revision received June 10, 2025; Accepted July 28, 2025
Electronically published November 16, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem
DOI: 10.5811/cpcem.47259
Introduction: Evaluating patients with acute neurologic deficits is a regular occurrence in the emergency department (ED), but some presentations warrant increased concern. This case highlights the importance of repeat evaluations and how resolution of symptoms does not rule out a more ominous underlying pathology.
Case Report: A 59-year-old male with a past medical history of coronary artery disease and Human immunodeficiency viruses (HIV) presented to a Level II trauma and comprehensive stroke center for left-sided facial droop and left- sided hemiparesis. Computed tomography of the brain including angiography and perfusion was performed revealing no hemorrhage or large vessel occlusion. Given his National Institutes of Health Stroke Scale score of 11, he received alteplase and subsequently experienced several episodes of resolution and recurrence of his symptoms while in the ED. Magnetic resonance imaging revealed an acute ischemic infarct in the right basal ganglia and insular region, which along with his clinical presentation was consistent with capsular warning syndrome.
Conclusion: Capsular warning syndrome is a rare clinical entity with an incidence ranging from 1.5-5% in stroke patients. Its recognition is crucial when making decisions concerning management, as resolution of symptoms should still garner a high level of attention given that the the increased risk of stroke with permanent neurological disability is highest within the first 48 hours. The role of thrombolysis continues to be an area of focus as its benefit has not yet been determined but continues to be the mainstay therapy in the correct clinical setting. This is especially true in the cases of recurrent episodes post thrombolysis, which does not preclude the diagnosis of capsular warning syndrome but should heighten the need for acute management of these patients and close monitoring. This case illustrates its unique presentation and the need for increased recognition and understanding within the field of emergency medicine. [Clin Pract Cases Emerg Med. 2025;10(1):7-9]
Keywords: capsular warning syndrome; lacunar stroke; case report
INTRODUCTION
Capsular warning syndrome is a rare clinical entity characterized by three or more recurrent episodes of motor and sensory deficits occurring within a 24-hour period, with complete resolution between episodes.1 This syndrome represents a distinct subtype of lacunar strokes, predominantly involving the basal ganglia, subcortical white matter, and
pons, and is notable for the absence of cortical symptoms such as visual field deficits, neglect, and agnosia.
The pathophysiology of capsular warning syndrome is attributed to compromised blood flow in small, branching lacunar vessels, which typically results in unilateral motor or sensory deficits. The clinical significance of this syndrome cannot be overstated, as approximately one-third of transient
ischemic attacks, of which lacunar infarcts constitute a part, are associated with subsequent cerebral infarction.2 The incidence rate of capsular warning syndrome ranges from 1.5-5%, highlighting the critical need for prompt recognition and management to mitigate the risk of progression to more severe cerebrovascular events. Early intervention is essential for improving patient outcomes and preventing the potential transition from transient ischemic episodes to definitive strokes.
We present the case of a 59-year-old male with a past medical history significant for coronary artery disease managed with seven cardiac stents and a diagnosis of HIV, who presented to the ED as a stroke alert due to left-sided facial droop and hemiparesis, which began about two hours prior to arrival to the ED. Upon evaluation, the patient’s blood pressure was 155/80 millimeters of mercury, heart rate 63 beats per minute, and respiratory rate 20 respirations per minute, with an oxygen saturation of 100% on room air. The patient’s National Institutes of Health Stroke Scale (NIHSS) score. was 11, indicating partial facial paralysis, no effort against gravity in the left upper and lower extremities, and mild-to-moderate dysarthria.
Because the patient was within the therapeutic window for administration of alteplase—a tissue plasminogen activator—a collaborative decision was made with neurology consult and the patient to proceed with thrombolytic therapy, which was initiated 22 minutes after the patient’s arrival. Following treatment, he exhibited rapid symptom resolution, achieving a NIHSS score of zero within 10-15 minutes. However, approximately 15 minutes after initial resolution he subsequently experienced four episodes of recurrent stroke symptoms, each lasting between 3-10 minutes, with complete resolution between episodes. During these episodes, his symptoms were identical to the initial presentation, and all four episodes were within a span of 48 minutes.
Neuroimaging, including computed tomography (CT) angiography of the head and neck and CT perfusion, revealed benign oligemia in the vertebral-basilar posterior circulation without evidence of penumbral regions or infarct core. The patient was then admitted to the intensive care unit for close monitoring. A magnetic resonance imaging study performed the following day identified an acute ischemic infarct in the right basal ganglia and insular region (Image). A follow-up CT conducted 24 hours later demonstrated hypodensity in the right putamen consistent with a subacute infarct.
Throughout his hospitalization, the patient underwent a bilateral extremity ultrasound, which was negative for deep vein thrombosis, and a transesophageal echocardiogram that ruled out the presence of thrombus in the left atrial appendage and aortic atheroma. Optimization of his medication regimen included the initiation of dual antiplatelet therapy with aspirin and clopidogrel for a duration of 21 days; the need for close monitoring was discussed with the patient. He remained stable
CPC-EM Capsule
What do we already know about this clinical entity?
Capsular warning syndrome represents a subtype of lacunar infarcts that involves episodic motor and sensory deficits within a 24-hour period.
What makes this presentation of disease reportable?
This description of episodes of capsular warning syndrome in the emergency department adds to the literature about this rare clinical entity.
What is the major learning point?
The transient nature of this syndrome can lead to incorrect recognition, thereby leading to improper management and disposition.
How might this improve emergency medicine practice?
Awareness of capsular warning syndrome could prevent the potential transition from transient ischemic episodes to definitive strokes.
without neurological episodes for six days and was subsequently discharged to a rehabilitation facility for continued recovery and management. At time of discharge, the patient’s modified Rankin Score for neurological disability was zero.
Lacunar infarcts result from the involvement of small penetrating lenticulostriate arteries that are affected by hypertensive arteriolar sclerosis.4 These arteries are branches of the middle cerebral artery and supply blood to critical regions, including the basal ganglia—comprising the striatum, nucleus accumbens, and globus pallidus—as well as the subcortical white matter, including the internal capsule and corona radiata, and the pons. These areas play integral roles in voluntary movement and feedback regulation to the cortex via the thalamus, specifically through its anterior, posterior, and genu limbs. Lesions in these regions can lead to abnormalities in the corticobulbar tract, corticospinal tract, and posterolateral thalamus.
Capsular warning syndrome represents a specific subset of lacunar infarcts characterized by three or more episodes of transient motor or sensory deficits affecting the face and extremities. Pure motor deficits are typically associated with
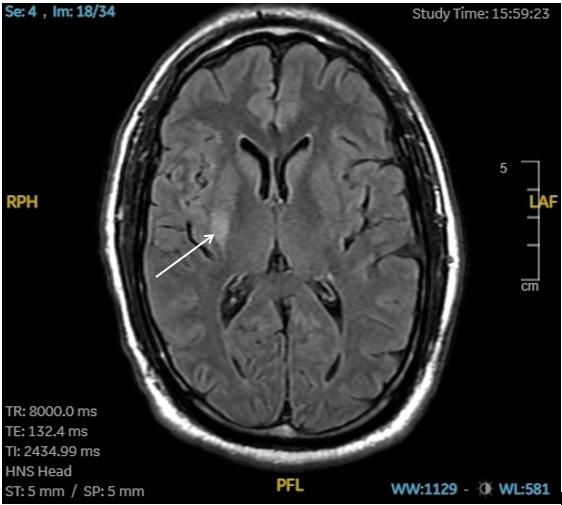
Image. T2 FLAIR of magnetic resonance imaging brain revealing an acute ischemic infarct in the right basal ganglia and insular region (white arrow).
FLAIR, fluid-attenuated inversion recovery.
lesions in the internal capsule, whereas pure sensory deficits correspond to lesions in the thalamus. Identifiable risk factors for this syndrome include hypertension, diabetes mellitus, dyslipidemia, and smoking.3 The incidence of capsular warning syndrome ranges from 1-5% of all transient ischemic attacks, highlighting a significant risk for subsequent stroke development.2 Notably, the risk of stroke associated with permanent neurological disability is highest within the first 48 hours, with up to a 60% likelihood of stroke occurring within the first week following the initial episode.3
Management strategies remain challenging to delineate. Current therapeutic approaches primarily involve tissue plasminogen activator and dual antiplatelet therapy, although studies are inconclusive as to the therapeutic efficacy of these modalities.5 The range of benefit specifically for thrombolytics ranges from complete resolution of symptoms and cessation of any further stuttering episodes to no benefit at all. 5 The need for further research into optimal management strategies is evident, given the high associated risk of impending cerebrovascular events.
Capsular warning syndrome serves as a critical clinical marker of impending lacunar infarcts, highlighting the need for heightened awareness among emergency clinicians.
Recognizing the transient nature of the neurological deficits, along with the significant risk factors such as hypertension, diabetes, dyslipidemia, and smoking, is essential for early intervention and management. The association between capsular warning syndrome and an elevated risk of subsequent stroke underscores the importance of timely diagnostic evaluation and therapeutic decision-making. Although current treatment strategies primarily involve tissue plasminogen activator and dual antiplatelet therapy, further research is necessary to optimize management and improve patient outcomes. Ultimately, early recognition and management of capsular warning syndrome are pivotal to preventing progression to disabling cerebrovascular events and improving long-term outcomes.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Address for Correspondence: Sean Serio, MD, HCA Florida Aventura Hospital, Department of Emergency Medicine, 20900 Biscayne Blvd, Aventura, FL 33180. Email: sserio18@gmail.com.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Tarahomi et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Martínez HR, Figueroa-Sanchez JA, Arreola-Aldape CA, et al. Capsular warning syndrome and its clinical awareness and therapeutic approach: two case reports and a systematic review of the literature. Front Neurol. 2023;14:1-11.
2. Ovbiagele B, Kidwell CS, Saver JL. Epidemiological impact in the United States of a tissue-based definition of transient ischemic attack. Stroke. 2003;34(4):919-24.
3. Paul NLM, Simoni M, Chandratheva A, et al. Population-based study of capsular warning syndrome and prognosis after early recurrent TIA. Neurology. 2012;79(13):1356-62.
4. Gore M, Bansal K, Khan Suheb MZ. Lacunar stroke. In: StatPearls [Internet]. March 10, 2024. Accessed March 22, 2025. https://www. ncbi.nlm.nih.gov/books/NBK563216/.
5. He L, Xu R, Wang J. Capsular warning syndrome: clinical analysis and treatment. BMC Neurology. 2019;19(1):1-7.
Anthony Rabil, MD*
Omar Fakhereddine, MD†
Abdallah Rebeiz, MD†
Tharwat El Zahran, MD*
Section Editor: John Ashurst, DO
American University of Beirut Medical Center, Department of Emergency Medicine, Faculty of Medicine, Beirut, Lebanon
American University of Beirut Medical Center, Department of Internal Medicine, Faculty of Medicine, Beirut, Lebanon
Submission history: Submitted January 21, 2025; Revision received March 31, 2025; Accepted May 21, 2025
Electronically published November 3, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem
DOI: 10.5811/cpcem.40059
Introduction: Takotsubo cardiomyopathy is characterized by stress-induced left ventricular dysfunction. The reverse form accounts for < 25% of all cases. We present a case of reverse Takotsubo cardiomyopathy in a young, otherwise healthy, woman following illicit drug use
Case Report: A 19-year-old female patient presented to the emergency department (ED) complaining of insomnia, left-sided chest pain, palpitations, and heightened energy levels after ingesting a significant quantity of small, rounded chocolate chips containing unidentified pills at a rave party the previous night. Her electrocardiogram revealed sinus tachycardia with ST-segment elevation in leads I and aVL. At the ED she developed respiratory distress and required oxygen supplementation. Her troponin level was 1.048 nanograms per milliliter (ng/mL) (reference range < 0.03 ng/mL), and her urine drug screen was positive for methamphetamines. Point-of-care transthoracic echocardiography showed moderately impaired left ventricular systolic function (ejection fraction approximately 35-39%) and hypokinesia of basal and mid-left ventricular segments accompanied by hyperkinesia of the apical segments, indicative of reverse Takotsubo cardiomyopathy. The patient was admitted to the cardiac care unit and showed clinical improvement after supportive treatment. However, 30 hours after discharge she presented back to the ED with epigastric pain and was found to have left renal artery thrombosis and an occlusive thrombus at the right internal iliac artery on computed tomography angiography.
Conclusion: Amphetamine intoxication is associated with the development of reverse Takotsubo cardiomyopathy, along with multiple vascular thromboses. [Clin Pract Cases Emerg Med. 2025;10(1):10-15.]
Keywords: case report; amphetamine; reverse Takotsubo cardiomyopathy; thrombosis.
Takotsubo cardiomyopathy (TTC), first described in 1980, is characterized by stress-induced left ventricular dysfunction that is reversible in most cases. It typically occurs in the setting of acute medical illness or during physical/emotional stress, hence its alternative name broken heart syndrome.1 Clinical presentation resembles that of acute coronary syndrome and is marked by electrocardiogram (ECG) changes, elevated troponin, and abnormal wall motion on echocardiography, despite the absence of obstructive
pericardial coronary artery disease.2 Several phenotypic variants of TTC have been reported, with the reverse form accounting for < 25% of all cases. Reverse TTC manifests as apical hyperkinesis alongside basal and midventricular hypokinesis or akinesis.3
Methamphetamine, a potent and addictive substance, is known for its modification from amphetamine through the addition of a methyl group, which enhances its lipid solubility and facilitates greater penetration through the blood-brain barrier.4 While the exact underlying pathophysiology is still
under investigation, hypothesized mechanisms include catecholamine cardiotoxicity, coronary artery spasm, coronary microvasculature impairment, and estrogen deficiency.2 We present a case of reverse cardiomyopathy in a young, otherwise healthy, woman following illicit drug use.
A previously healthy 19-year-old female, who was undergoing treatment for bronchitis with oral steroids, presented to the emergency department (ED) complaining of insomnia, left-sided chest pain, palpitations, and heightened energy levels following attendance at a rave party the previous night. She confirmed ingestion of marijuana edibles, a significant quantity of small, rounded chocolate chips containing unidentified pills, and multiple energy drinks approximately 12-14 hours prior to ED presentation. Her symptoms started three hours postingestion, characterized by temporary confusion, decreased concentration, incoherent speech, and severe left-sided chest and back pain radiating to her left shoulder.
Although these symptoms resolved, she continued to experience persisting mild chest pain. Additionally, she exhibited diaphoresis, tachycardia, headache, chest tightness, and nausea but was unable to vomit. Review of systems was otherwise unremarkable. Upon presentation, she was tachycardic with a heart rate of 137 beats per minute. She appeared anxious, complaining of mild dyspnea and rapid breathing. Her pupils were dilated and reactive. The rest of her physical exam was otherwise unremarkable. Intravenous fluids were administered along with symptomatic treatment. An ECG revealed sinus tachycardia with ST-segment elevation in leads I and aVL (Image 1).
A chest radiograph (CXR) displayed normal findings (Image 2A). As the patient awaited the results, she developed dyspnea, worsening tachycardia, and desaturation to 92%, while maintaining her airway. She reported increased severity of her left chest pain. Supplemental oxygen was administered,
CPC-EM Capsule
What do we already know about this clinical entity?
Reverse Takotsubo is a rare stress-induced cardiomyopathy variant linked to catecholamine surges, often misdiagnosed, especially in young or atypical patients.
What makes this presentation of disease reportable?
This rare case shows reverse Takotsubo with major arterial thromboses in a healthy 19-year-old after methamphetamine use, highlighting severe stimulant-related risks.
What is the major learning point?
Methamphetamine toxicity can cause reverse Takotsubo cardiomyopathy and large vessel thromboses. Clinicians must suspect cardiac issues and ensure close follow-up in young stimulant users.
How might this improve emergency medicine practice?
Early cardiac evaluation and awareness of delayed thrombotic risks in young stimulant users can improve diagnosis, monitoring, and safety in emergency care.
and a repeat ECG revealed no changes. A repeat CXR revealed bilateral bronchial wall thickening with patchy, interstitial-like opacities in the mid and lower lung fields (Image 2B). Intravenous (IV) hydration was withheld. Her troponin level was 1.048 nanograms per milliliter (ng/ mL) (reference range: < 0.03 ng/mL). The urine drug screen was positive for amphetamines and tetrahydrocannabinol. The
Amphetamine-induced Reverse Takotsubo Cardiomyopathy and Thrombosis: A Case Report
cardiology team was consulted. Point-of-care transthoracic echocardiography showed moderately impaired left ventricular systolic function with an estimated left ventricular ejection fraction (LVEF) of 35-39% (50-70%). There was hypokinesia of basal and mid-left ventricular segments, accompanied by hyperkinesia of the apical segments, indicative of reverse TTC.
The patient was admitted to the cardiac care unit, and her symptoms improved with IV diazepam as needed. Given her age, symptom improvement, and absence of concerning findings for obstructive coronary disease, cardiac catheterization was deferred. Subsequently, she was initiated on ivabradine five milligrams (mg) twice daily. Ivabradine lowers heart rate by selectively inhibiting the “funny” (If) current—a key pacemaker current in the sinoatrial node responsible for initiating spontaneous diastolic depolarization—without affecting myocardial contractility or blood pressure.5
She was discharged home 48 hours later after complete resolution of her symptoms. However, 30 hours postdischarge, the patient presented back to the ED with periumbilical pain exacerbated by worsening of pre-existing constipation. Her review of system was positive for nausea. Her physical examination revealed periumbilical tenderness.
Computed tomography angiography of the abdomen demonstrated left renal artery thrombosis, with complete occlusion of the anterior segment, partial occlusion of the posterior segment, and slight extension into the main renal artery (Image 3). Additionally, secondary renal infarction was observed, characterized by increased hypodensity involving the posterior aspect of the kidney and an occlusive thrombus at the right internal iliac artery (Image 4). On blood workup, she was found to have an acute kidney injury with a creatinine increase from 0.62 mg per deciliter (dL) on discharge day to 0.95 mg/dL (0.5-1.0 mg/dL). Ramipril was withheld to avoid possible worsening of renal function.
Vascular surgery and interventional radiology teams were consulted for possible embolectomy. The family was debriefed on the risks vs benefits of intervention. Both teams agreed with the family that the best course of treatment was conservative
management with therapeutic anticoagulation. The patient was started on acetylsalicylic acid and heparin drip and was admitted to the cardiac care unit for further investigation.
Point-of-care ultrasound showed normalization of left ventricular function that was still mildly dilated with a LVEF of 60%. In the investigation of thrombosis findings, a family history of deep venous thrombosis was found in the paternal uncle and aunt and from the mother’s side. (The father and mother were first-degree relatives.)
Thrombophilia workup was negative for lupus anticoagulant. Screening for factor V, factor II, and methylenetetrahydrofolate reductase (MTHFR) gene mutations using FV-II-MTHFR strip assay (reverse hybridization-sequence specific oligonucleotide probe) showed one heterozygous mutation of MTHFR (C677T) (Appendix 1). Since the patient’s creatinine level normalized, the hematology team recommended switching from heparin drip to subcutaneous enoxaparin and to follow up in clinic after discharge. She was discharged home on subcutaneous enoxaparin, bisoprolol, ivabradine, and acetylsalicylic acid.
Takotsubo cardiomyopathy, also referred to as stressinduced cardiomyopathy, is an acute but reversible form of left ventricular dysfunction that mimics the presentation of an acute myocardial infarction. It is believed to be triggered by physical and emotional stress, although cases without identifiable triggers have also been reported.6 The incidence is estimated to be 15-30 cases per 100,000 per year in the United States, but this figure may be underestimated due to confusion with acute coronary syndrome and subclinical cases in patients who do not seek medical attention.7 Takotsubo cardiomyopathy is more common in females, particularly post-menopausal women.6 Several variants of TTC have been described based on the location of ventricular wall motion abnormality (akinesis or hypokinesis). The most common type is the apical variant, accounting for 81.7% of cases, followed by the midventricular type (14.6% of cases) and, finally, the basal and focal variants (2.2% and 1.5 % of cases, respectively).6
Reverse TTC refers to the basal type of TTC characterized by akinesis or severe hypokinesis of the base with sparing of
Amphetamine-induced Reverse Takotsubo Cardiomyopathy and Thrombosis: A Case Report
the apex. Contrary to the apical type, reverse TTC is more commonly seen in younger rather than post-menopausal women.7-9 It has also been reported to be associated with amphetamine-type stimulants.9
The exact pathophysiology of TTC remains unclear. The most accepted theory is catecholamine-induced cardiotoxicity, where a surge of catecholamines and other stress hormones results in direct cardiotoxic effect and microvascular dysfunction.2,10
Methamphetamine belongs to the phenethylamine family. Its rapid onset of action and prolonged activity is related to the addition of a methyl group to amphetamine, giving it an enhanced lipophilicity.4 Methamphetamine acts by increasing the release of catecholamines (dopamine and norepinephrine), blocking their degradation by inhibiting the action of monoamine oxidase, and binding to various receptors of the cardiovascular system resulting in a prominent adrenergic stimulation.11
Reports suggest that methamphetamine causes cardiomyopathy through direct pathways such as increased production of free radicals, altered mitochondrial function and dysfunction in intracellular calcium hemostasis, and through indirect pathways by causing coronary vasospasm, hypertension, and tachycardia.9 Amphetamines can promote clot formation by increasing endothelial tissue factor expression, impairing natural anticoagulants and causing vascular inflammation.12 Moreover, although data remains limited, marijuana has been reported as a potential contributor to TTC, possibly also via catecholaminergic stimulation or autonomic dysregulation.13 Garakanidze et al found a correlation between MTHFR gene polymorphism and arterial thrombosis.14 These factors may have collectively contributed to the development of thrombosis in this patient.
Patients with TTC often present with symptoms similar to acute coronary syndrome, including angina-like chest pain, dyspnea, syncope, nausea, diaphoresis, and epigastric pain.2,8 Additionally, they may present in cardiogenic shock or dysrhythmias. On ECG, ST-segment elevations resembling those seen in ST-segment elevation myocardial infarction are common but are transient and often resolve within a few days. Other electrocardiogram findings include ST-segment depression, T-wave inversion, QT-interval prolongation, and a new bundle branch block.8 Patients with reverse TTC often present with ST-segment depression and QT-interval prolongation.2 Troponin and B-type natriuretic peptid levels are often elevated, reflecting both myocardial insult and high left ventricular pressure respectively.
Echocardiography can often distinguish between acute myocardial infarction and TTC. Cardiac catheterization with ventriculography remains the gold standard for diagnosis. The most commonly used diagnostic criteria is the revised Mayo Clinic Criteria, which include the following: transient hypokinesis, akinesis, or dyskinesis of the left ventricular midsegments with or without apical involvement with regional wall motion abnormalities extending beyond a single
epicardial vascular distribution; absence of obstructive coronary disease or angiographic evidence of acute plaque rupture; new ECG abnormalities (ST-segment elevation or T-wave inversion) or modest elevation in cardiac troponin; and absence of significant stressful event including pheochromocytoma, myocarditis, intracranial bleed, or recent significant head trauma.10
Management of TTC is focused on supportive care and management of complications, including cardiogenic shock and arrhythmias. According to the European Society of Cardiology, patients with Takotsubo syndrome should be admitted to a monitored unit and risk stratified into high or low risk. Risk stratification is based on several criteria as shown in Appendix 2. Low-risk patients are often managed conservatively with consideration of beta-blocker therapy and angiotensinconverting enzyme inhibitors in patients with LVEF < 45%. In high-risk patients, observation is recommended in a monitored unit for at least 72 hours to assess for complications including cardiogenic shock, pulmonary edema, thrombus formation, and arrhythmias.15 Beta blockers can be used for management of arrhythmias, left ventricular outflow tract obstruction (with a gradient more than 40 millimeters of mercury) and when LVEF is < 45%. Angiotensin-converting enzyme inhibitors are also recommended if LVEF is < 45%. Serial imaging is helpful to reassess for improvement in regional wall motion with followup in 3-6 months.15
Methamphetamine intoxication is associated with the development of reverse Takotsubo cardiomyopathy, along with multiple vascular thromboses. Takotsubo cardiomyopathy presents with symptoms resembling acute coronary syndrome; echocardiography can help differentiate between Takotsubo cardiomyopathy and acute myocardial infarction. Management of Takotsubo cardiomyopathy is focused on supportive care and management of complications including cardiogenic shock and arrhythmias.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Address for Correspondence: Tharwat El Zahran, MD, American University of Beirut Medical Center, Department of Emergency Medicine, Beirut, Lebanon, P.O.Box 11-0236, Riad El-Solh/Beirut 1107 2020 Lebanon. Email: te15@aub.edu.lb
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Amphetamine-induced Reverse Takotsubo Cardiomyopathy and Thrombosis: A Case Report Rabil et al.
Copyright: © 2025 Rabil et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Patankar GR, Choi JW, Schussler JM. Reverse Takotsubo cardiomyopathy: two case reports and review of the literature. J Med Case Rep. 2013;7(1):84.
2. Awad HH and McNeal AR. Reverse Takotsubo cardiomyopathy: a comprehensive review. Ann Transl Med. 2018;6(23):460.
3. Takafuji H, Arai J, Saigusa K, et al. Reverse Takotsubo cardiomyopathy caused by patent foramen ovale-related cryptogenic stroke: a case report. Eur Heart J Case Rep. 2020;4(6):1–6.
4. Reddy PKV, Ng TMH, Oh EE, et al. Clinical characteristics and management of methamphetamine-associated cardiomyopathy: state-of-the-art review. J Am Heart Assoc. 2020;9(11):e016403.
5. Riccioni G. Ivabradine: from molecular basis to clinical effectiveness. Adv Ther. 2010;27(3):160–7.
6. Templin C, Ghadri JR, Diekmann J, et al. Clinical features and outcomes of Takotsubo (stress) cardiomyopathy. N Engl J Med 2015;373(10):929–38.
7. Ramaraj R and Movahed MR. Reverse or inverted Takotsubo cardiomyopathy (reverse left ventricular apical ballooning syndrome) presents at a younger age compared with the mid or apical variant and is always associated with triggering stress. Congest Heart Fail 2010;16(6):284–6.
8. Ghadri JR, Cammann VL, Napp LC, et al. Differences in the clinical profile and outcomes of typical and atypical Takotsubo syndrome: data from the international takotsubo registry. JAMA Cardiol 2016;1(3):335–40.
9. Jafari Giv M. Exposure to amphetamines leads to development of amphetamine type stimulants associated cardiomyopathy (ATSAC). Cardiovasc Toxicol. 2017;17(1):13–24.
10. Medina de Chazal H, Del Buono MG, Keyser-Marcus L, et al. Stress cardiomyopathy diagnosis and treatment. J Am Coll Cardiol 2018;72(16):1955–71.
11. Kevil CG, Goeders NE, Woolard MD, et al. Methamphetamine use and cardiovascular disease: in search of answers. Arterioscler Thromb Vasc Biol. 2019;39(9):1739–46.
12. Gebhard C, Breitenstein A, Akhmedov A, et al. Amphetamines induce tissue factor and impair tissue factor pathway inhibitor: role of dopamine receptor type 4. Eur Heart J. 2010;31(14):1780–91.
13. Meera SJ, Vallabhaneni S, Shirani J. Cannabis-induced basal-midleft ventricular stress cardiomyopathy: a case report. Int J Crit Illn Inj Sci. 2020;10(Suppl 1):49–52.
14. Garakanidze S, Costa E, Bronze-Rocha E, et al. Methylenetetrahydrofolate reductase gene polymorphism (C677T) as a risk factor for arterial thrombosis in Georgian patients. Clin Appl Thromb Hemost. 2018;24(7):1061–6.
15. Lyon AR, Bossone E, Schneider B, et al. Current state of knowledge on Takotsubo syndrome: a position statement from the Taskforce on Takotsubo Syndrome of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail 2016;18(1):8–27.
Rabil et al.
Amphetamine-induced Reverse Takotsubo Cardiomyopathy and Thrombosis: A Case Report
Appendix 1. Thrombophilia workup done during admission for assessment of predisposing factors of thrombosis.
Tests
PTT, partial thromboplastin time; DRVVT, diluted Russell viper venom time; ACA, anti-cardiolipin antibodies; IgM, immunoglobulin M; IgG, immunoglobulin G; PT, prothrombin time; INR, international normalized ratio; mmHg, millimeter of mercury.
Appendix 2. Risk stratification in Takotsubo cardiomyopathy. Adopted from the European Society of Cardiology Guidelines.
Major
Age ≥ 75 years Age < 70-75 years
Blood pressure < 110 mmHg Physical stressor + LV function < 35% LV function
Pulmonary edema + Biventricular involvement +
Arrhythmias/ Syncope + Concomitant obstructive CAD +
LVOTO ≥ 40 mmHg NT- proBNP ≥ 2000 pg/ml
Moderate to Severe Mitral Regurgitation + BNP ≥ 600 pg/ml
Apical Thrombus + QTC ≥ 500 ms +
New/Contained VSD + Pathological Q-waves + Persistent ST-segment elevation +
High risk: at least 1 major + 2 minor criteria
LV, left ventricle; LVOTO, left ventricular outflow tract obstruction; VSD, ventricular septal defect; CAD, coronary artery disease; NTproBNP, N-terminal pro B-type natriuretic peptide; BNP, B-type natriuretic peptide; QTC, corrected QT-interval in milliseconds.
Julian Campillo Luna, MD, MHS
Robert W. Shaffer, MD
Section Editor: Joel Moll, MD
Arbor, Michigan
Submission history: Submitted May 25, 2025; Revision received July 18, 2025; Accepted August 4, 2025
Electronically published November 16, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.47934
Introduction: Emergency physicians can use point-of-care ultrasound (POCUS) to identify lymph nodes in certain clinical scenarios, and advanced users can determine significant information (such as concerns for malignancy or differentiating them from abscesses for incision and drainage) based on a large volume of literature and images associated with those pathologies.1 However, current literature does not contain a similar volume of images and cases of suppurative lymph nodes, or buboes, limiting the ability to make the diagnosis sonographically at the bedside.
Case Report: We report on a man who presented to the emergency department (ED) with a worsening inguinal mass that changed size with positioning, as well as a 20-pound weight loss occurring over the course of a month. Point-of-care ultrasound of the mass was concerning for a necrotic suppurative lymph node, which was further evaluated with cross-sectional imaging. The patient was admitted for a biopsy to rule out malignancy. He was discharged with serologies for Bartonella henselae pending, which later returned positive. The patient was then switched to azithromycin with significant improvement of his symptoms.
Conclusion: As POCUS becomes the modality of choice for rapid assessment of soft tissue masses in the ED, familiarity with less common variants of soft tissue infections such as buboes can help with medical decision-making, risk stratification, and further workup. This sonographic description of a bubo caused by a common zoonotic infection will enable clinicians to familiarize themselves with their appearance. [Clin Pract Cases Emerg Med. 2025;10(1):16-19.]
Keywords: ultrasound; emergency medicine; Bartonella; bubo; case report.
Buboes are suppurative and necrotic lymph nodes with significant associated tenderness. First described in the sixth century during the Justinian plague,2 they were frequently encountered as the manifestations of Yersinia pestis infections, a disease often referred to as “bubonic plague.” However, Y. pestis is not the only causative agent of buboes; they have been described in a range of diseases from venereal infections such as Chlamydia trachomatis, Haemophilus ducreyi, and syphilis,3 as well as mycobacterial infections (both classic tuberculosis and atypical mycobacteria),4 zoonotic diseases such as bartonellosis (cat scratch disease), filariasis, and tularemia,5 and non-infectious diseases such as cancer.6
Buboes often represent a natural progression of localized infection of bacterial pathogens into the lymphatic system, creating a localized response that then produces purulence secondary to neutrophil migration. The diagnosis of a bubo is made clinically, although imaging and histological examination can greatly aid in the diagnosis, as the differential for these lesions is broad. Their location, commonly near large neurovascular bundles (axillary, inguinal, and cervical), means clinicians must also rule out malignancy, vascular abnormalities such as pseudoaneurysms, and uncomplicated abscesses. Point-of-care ultrasonography (POCUS) in the hands of an apt clinician lends itself to rapid evaluation of most or all these etiologies.
Luna et al. Sonographic Evaluation of an Inguinal Bubo from Bartonella henselae: A Case Report
A 30-year-old male with no significant past medical history presented to the emergency department (ED) with a chief complaint of a soft-tissue mass on his abdomen that changed size depending on his positioning, getting bigger when he stood and decreasing in size when he would lie down. This mass was in the left groin area and had been growing for a month. Over the preceding week the patient had also noticed a small amount of erythema overlying the mass, and it had become painful at rest and tender to touch. He also reported approximately 20 pounds of weight loss over the prior month and a half, which made him worry that he had cancer.
He initially presented to his primary care physician (PCP), who evaluated him with sexually transmitted infection serologies, which were negative, as well as an outpatient ultrasound that demonstrated enlarged, hypoechoic, and hyperemic left-groin lymph nodes with a 3.7-cm complex fluid collection, which was concerning for an abscess. He did not undergo incision and draining. He then presented to his local ED where he was further evaluated with computed tomography (CT) of his abdomen and pelvis with intravenous contrast and found to have a 4.7-cm nonspecific, soft-tissue nodule in the left inguinal region, likely an infected or inflammatory lymph node. He was then discharged with seven days of cephalexin.
He presented to our ED approximately six days into his cephalexin course, with concerns that the mass kept growing despite the antibiotics. On exam he presented with an enlarged inguinal mass with mild erythema (Image).
The patient was evaluated with a basic laboratory panel including a complete blood count, a basic metabolic panel, a urinalysis, lactate dehydrogenase due to concerns for malignancy, C-reactive protein, and a set of blood cultures, all of which were within normal range. A radiology-based ultrasound was ordered, which re-demonstrated a complex fluid collection below the skin concerning for abscess. We also ordered computed tomography (CT) of the abdomen and pelvis to compare to the prior image to determine whether further growth had occurred, and to ensure that this was not a small hernia given its changes with position. The CT confirmed interval increase in soft tissue hypodense abnormality from 1.7 cm to 3.9 cm. The read was concerning for either a septation or vascularity, which led us to obtain POCUS. Using a high-frequency linear probe, the L20-5s (Mindray Medical International Ltd, Shenzhen, China), we were able to visualize the lesion in greater detail.
We saw a complex fluid collection, with a thin strip of echogenic material nearly bisecting it longitudinally. A small defect in that echogenic material had created a pathway for the slightly hypoechoic fluid inside to flow from deep to superficial, which we were able to demonstrate with gentle compression of the skin overlying the area of maximum fluctuance (Video). This was interpreted at bedside to be purulent fluid moving across the space.
These findings led us to believe it was not a simple abscess and to continue our workup prior to an incision and
CPC-EM Capsule
What do we already know about this clinical entity?
Buboes can present in multiple different illnesses, among them bartonellosis. Ultrasound can aid in rapidly evaluating soft-tissue masses.
What makes this presentation of disease reportable?
To our knowledge, this is the first description of the sonographic appearance of a bubo from Bartonella henselae.
What is the major learning point?
This report identifies the sonographic features of an inguinal bubo and how it differs from softtissue abscesses and masses.
How might this improve emergency medicine practice?
By empowering physicians to recognize the morphological appearance of these buboes, painful and unnecessary incision and drainage procedures can be avoided.
drainage. We became concerned that POCUS had instead revealed a bubo. Further questioning of the patient revealed that he had recently acquired a kitten, which would often scratch at his legs. No abrasions were noted to the legs of the patient to suggest a bacterial, soft tissue infection as the cause of lymph node reactivity, but this information increased our clinical suspicion for cat scratch disease, and B. henselae antibodies were sent at that time.
The patient was admitted to our short-stay medical unit where we completed the workup, specifically to rule out malignancy, while waiting for the Bartonella titers. He received an interventional radiology-guided needle biopsy of the mass, which was unrevealing. At that point, the patient was discharged with a seven-day prescription for trimethoprim-sulfamethoxazole (TMP-SMX) and instructed to follow up with the infectious diseases clinic as an outpatient. The day after discharge the patient’s titers returned positive for B, henselae immunoglobulin G at a 1:4096 titer (reference range: less than 1:128) suggesting recent/active infection. His tuberculosis serology was negative, and his needle biopsy showed lymph node tissue with no neoplastic changes. Fluid cultures were positive for Enterococcus faecalis in small quantities, favored to be a contaminant by the infectious diseases team following the case. The patient was informed of
Image. Photograph of patient’s left hip/inguinal region, with his feet facing the left of the image while supine. A 3-cm area of erythema and fluctuance was appreciated, corresponding to the patient’s source of pain (arrow).
Video. High-frequency linear probe at 2-cm depth showing the fluid collection (indicator facing the patient’s head). An echogenic strip at 0.75-cm depth corresponds to the hilum of the lymph node (white arrow), as well as a small defect through which pus can move (black arrow).
these findings; however, he missed his initial appointment with infectious diseases.
The patient called his PCP after the TMP-SMX course had been completed, with failure to improve. After a phone conversation with the infectious diseases team, the patient was switched to azithromycin (500 mg on day one and 250 mg daily for four additional days) per US Centers for Disease Control and Prevention recommendations. Follow-up a couple days later with his PCP showed that the patient’s pain was decreasing significantly. He presented to the infectious diseases clinic a week later with complete resolution of the symptoms. He has not since followed up or interacted with our medical system.
This case is a reminder that the bedside assessment for abscess and soft tissue infection using POCUS can go further than determining whether a collection of fluid exists or whether vascular features are present. As ultrasound equipment becomes more advanced and physicians become
more adept using it, POCUS can serve as a useful tool for further narrowing down a differential for these clinical cases. In this case ultrasonography was critical in accelerating care and eventually in accurately diagnosing the patient. The differential for bubo-producing illnesses includes significant diseases of public health note such as syphilis, chlamydia, and bubonic plague caused by Y. pestis. It can also be caused by lymphoma, as well as B. henselae.
In this case, the patient experienced changes in the size and protuberance of this mass with positioning and bearing down, which combined with the location and tenderness of the mass, made us significantly concerned for a hernia. Ultrasound demonstrated that the purulent material was traveling through a defect in the internal architecture of the lymph node, causing more pus to collect on the outside section as the patient’s intra-abdominal pressure increased. This is clearly seen in the video, during which graded compression achieved this same result in the opposite direction. Interestingly, the literature contains other cases of buboes masquerading as hernias due to this same effect, including one in which the organism was identified as Mycobacterium avium intracellulare.4
Ultrasonographic assessment of soft tissue masses and collections can go further than assessing for vascular structures in or over the planned incision-and-drainage site and help to significantly narrow the differential for an infectious process. This case is also a good reminder of the need to keep a broad differential for soft tissue masses. Emergency physicians should maintain buboes and their associated causal agents high in the differential for any abscess that fails to respond to traditional antibiotic therapy and have a low threshold for pursuing inpatient workups given the high-risk nature of these illnesses to both patient and public health.
Luna et al.
Sonographic Evaluation of an Inguinal Bubo from Bartonella henselae: A Case Report
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Address for Correspondence: Julian Campillo Luna, MD MHS, University of Michigan, Emergency Department, 1500 E Medical Center Dr, B1-380 Taubman Center, SPC 5305, Ann Arbor, MI 48109, United Sates. Email: Jucampil@med.umich.edu
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 First Author last name et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http:// creativecommons.org/licenses/by/4.0/
1. McCafferty J and Forsyth J. Point of care ultrasound made easy. CRC Press; 2020: 35-36
2. Raoult D, Mouffok N, Bitam I, et al. Plague: history and contemporary analysis. J Infect. 2013;66(1):18-26.
3. Viravan C, Dance DAB, Ariyarit C, et al. A prospective clinical and bacteriologic study of inguinal buboes in Thai men. Clin Infect Dis 1996;22(2):233-239.
4. Hodge K, Orgler R, Monson T, et al. Bubo masquerading as an incarcerated inguinal hernia. Hernia. 2001;5(2):97-98.
5. Oršolić M, Sarač N, Balen Topić M. Vector-borne zoonotic lymphadenitis—the causative agents, epidemiology, diagnostic approach, and therapeutic possibilities—an overview. Life 2024;14(9):1183.
6. Fried BM. Leukemic infiltrations of the central nervous system J Am Med Assoc. 1934;102(3):231.
Ashlynn
Felker, MD
Jason Tanner, MD, PhD
Section Editor: Anna McFarlin, MD
University of Utah Health, Department of Emergency Medicine, Salt Lake City, Utah
Submission history: Submitted February 26, 2025; Revision received June 18, 2025; Accepted July 29, 2025
Electronically published November 22, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.43526
Introduction: Temporal lobe epilepsy is a form of focal epilepsy that originates in the temporal lobes, often presenting with a variety of symptoms including altered consciousness, automatisms, and focal seizures with or without impaired awareness. Given such a diversity of manifesting symptoms, recognizing temporal lobe epilepsy in the emergency department (ED) can be challenging. Early identification is crucial for appropriate management, including timely initiation of antiepileptic therapy and differentiation from other neurological emergencies.
Case Report: A 50-year-old male with no prior history of seizures or neurological conditions presented to the ED after experiencing unusual sensations that had begun three days earlier. The patient described an intermittent sensation of warmth rising from his pelvis to his head, accompanied by an experiential déjà vu-like feeling he described as “dream reenactment.” His episodes had become progressively more frequent, occurring approximately once every 90 minutes within the first 24 hours, with two instances of brief loss of consciousness. Diagnostic workup, including neurology consultation and an electroencephalogram in the ED, revealed a 19-second, non-motor focal seizure originating from the left anterior temporal region, consistent with temporal lobe epilepsy. Magnetic resonance imaging showed no acute structural abnormalities. The patient was diagnosed with temporal lobe epilepsy, started on lacosamide, and discharged from the ED.
Conclusion: This case underscores the importance of recognizing temporal lobe epilepsy in the ED, particularly in patients with recurrent episodes of altered consciousness or unusual sensory experiences. Prompt diagnosis and treatment are critical to preventing further seizures and improving quality of life. [Clin Pract Cases Emerg Med. 2025;10(1):20-23.]
Keywords: seizure; temporal lobe epilepsy; electroencephalogram; déjà vu; case report.
Temporal lobe epilepsy is a common form of focal epilepsy that originates in the temporal lobes of the brain, leading to recurrent seizures that can present in various clinical forms, ranging from focal onset seizures with or without impaired awareness to more generalized convulsions.1-3 Given that the semiology of temporal lobe epilepsy can include various symptoms such as unusual sensory experiences, altered consciousness, automatisms, and postictal confusion, recognizing this neurological disorder in the emergency department (ED) can be
challenging. Prompt identification of temporal lobe epilepsy is crucial, however, for timely initiation of antiepileptic drug therapy, prevention of further seizure activity, and evaluation for possible underlying causes, such as brain lesions or structural abnormalities.4 Additionally, distinguishing between epilepsy-related seizures and other potential neurological emergencies, such as stroke or infection, is essential for reducing morbidity and improving patient outcomes in the ED setting.5 Therefore, emergency clinicians must be equipped with the knowledge and skills to accurately identify and manage temporal lobe seizures.
A 50-year-old male with no significant past medical history presented to the ED following an episode of altered mental status. The patient had no prior medical problems and no prior history of seizures, traumatic brain injury, or neurological conditions. His symptoms had begun suddenly three days prior to ED presentation when he noticed the onset of unusual sensations. He described experiencing a rising sensation, accompanied by a conscious “dream reenactment” and a sense of warmth that started in his pelvis and radiated upward to his head. These episodes were initially brief but progressively increased in frequency and duration. The patient had previously sought care at a local urgent care and was diagnosed with vasovagal syncope, but he presented to the ED due to persistent and worsening symptoms.
The episodes were characterized by staring spells with preserved awareness. He reported mild confusion after some, but not all, episodes. The episodes typically lasted 30-45 seconds, occurring randomly throughout the first two days, and became more frequent, occurring every 90 minutes during the day leading up to presentation. He also reported two instances of loss of consciousness, both of which were brief (less than 5 minutes) and only one of which had been witnessed. During his ED visit he experienced another episode of staring with preserved awareness, which was witnessed by ED staff; it was accompanied by a brief episode of sinus bradycardia of 40-50 beats per minute, with no other vital sign changes. There were no observed motor symptoms, either focal or generalized, and no pupillary changes noted during the episode.
On examination, the patient was alert, oriented, and able to engage in conversation. Neurological findings were otherwise unremarkable, with no signs of focal deficits or significant abnormality on the initial assessment. Given his persistent and worsening symptoms, and an episode witnessed in the ED, neurology was consulted. An electroencephalogram (EEG) was performed in the ED, which showed evidence of a 19-second, non-motor focal seizure originating from the left anterior temporal region. The seizure activity was suggestive of left anterior temporal lobe epileptogenicity, consistent with temporal lobe epilepsy. Magnetic resonance imaging of the brain was also obtained while in the ED, which showed no acute structural abnormalities.
The clinical presentation, together with the EEG findings, led to a new diagnosis of temporal lobe epilepsy. The left anterior temporal lobe was identified as the likely origin of the seizure activity, and the absence of prior seizure history and structural brain abnormalities further supported this diagnosis. The patient was initiated on intravenous lacosamide with a loading dose of 200 mg and was prescribed a maintenance dose of 100 mg orally twice daily. Outpatient neurology was arranged for ongoing management and adjustment of his
CPC-EM Capsule
What do we already know about this clinical entity?
Temporal lobe epilepsy (TLE) often presents with subtle sensory or experiential symptoms that can mimic syncope, anxiety, or migraine.
What makes this presentation of disease reportable?
Rapidly escalating focal seizures with preserved awareness and ictal bradycardia illustrate an uncommon presentation of new-onset TLE.
What is the major learning point?
Recurrent déjà vu, visceral warmth, or dreamlike sensations may represent focal seizures rather than benign or psychogenic events.
How might this improve emergency medicine practice?
Recognizing atypical seizure presentations allows prompt neurology consultation or referral, preventing misdiagnosis and delayed care.
treatment regimen as necessary, and the patient was discharged home.
Temporal lobe epilepsy is a significant cause of focal seizures, yet it is often under-recognized, especially in its early stages when symptoms can be subtle or atypical.1,2 Although diagnosis is usually made during childhood and adolescence, temporal lobe epilepsy is the most common focal epilepsy in adults, representing an estimated 60% of all focal epilepsies and approximately 30% of all epilepsy cases overall. Given that the prevalence of epilepsy is approximately 1% in developed countries, temporal lobe epilepsy affects nearly 0.3% of the general population.6,7 The disorder is characterized by focal seizures originating from the temporal lobe, which may manifest as a variety of sensory or experiential phenomena, or auras, such as flushing, déjà vu, olfactory or gustatory hallucinations, epigastric rising sensations, intense emotions (such as fear), and auditory or visual hallucinations.8 These auras often precede a seizure by seconds or minutes. During the aura, a patient retains awareness and can describe motor, sensory, autonomic, or psychic symptoms. These symptoms are subjective. Because they are not commonly recognized as presenting symptoms of epilepsy in the ED,
Diagnosing Temporal Lobe Epilepsy in the Emergency Department: A Case Report
they can be mistaken for non-epileptic events such as migraine aura, transient ischemic attack, psychogenic non-epileptic seizures, psychiatric disorders or other neurological emergencies such as transient global amnesia, vestibular disorders or sleep disorders, and near-syncope or syncope.8,9
The typical frequency of temporal lobe seizures is highly variable, ranging from multiple episodes per day to only a few per year.10 The rapid onset and increasing frequency of seizures in this case—progressing to every 90 minutes over a span of three days—was more abrupt than typically seen in temporal lobe epilepsy, making this presentation particularly noteworthy. Symptoms like the “rising sensation,” “dream reenactment” or déjà vu sensation, and preserved awareness during episodes are less commonly recognized in the ED setting as symptoms of epilepsy. While ictal tachycardia is common in patients with temporal lobe epilepsy, ictal bradycardia, as was seen in this case, is less frequent and typically associated with focal seizures with impaired awareness, mimicking or coinciding with syncope and further complicating diagnosis.11,12 This highlights the importance for emergency physicians to be vigilant and consider temporal lobe epilepsy in their differential diagnosis, particularly in patients presenting with unusual, recurrent episodes of altered mental status, staring, or altered awareness or perception.
This case also underscores the differences in available resources across various ED settings, highlighting the diagnostic challenges that emerge in resource-limited environments. While this case occurred in a tertiary-care center with in-house neurology and use of conventional full-montage EEG in the ED, such resources are often not readily available in many settings, making conditions such as temporal lobe epilepsy challenging to recognize and manage. In such environments, point-of-care EEG devices may offer some diagnostic utility; however, to our knowledge, these tools have not been specifically studied or validated for the diagnosis of this neurological disorder, and further research is needed to evaluate their effectiveness in this context. Additionally, patient transfer may be considered if the patient exhibits persistently altered mental status, otherwise unexplained focal neurological deficits, or other concerns for status epilepticus. For stable patients without such concerns, local management may be appropriate with timely neurology referral arranged.2,13
Specialist consultation also informs treatment decisions. In this case, lacosamide was selected based on neurology input. However, levetiracetam is also a safe and effective first-line option for new-onset temporal lobe epilepsy, as it is broadly available and is generally well tolerated. Studies suggest comparable efficacy and side-effect profiles between levetiracetam and lacosamide; therefore, treatment should be individualized based on patient factors such as comorbidities and potential drug interactions.2,14
Furthermore, temporal lobe epilepsy has been associated with increased intensity and frequency of psychiatric disorders such as anxiety and depression.15 Early recognition in the ED can lead to timely diagnosis and treatment, potentially preventing complications such as injury, cognitive decline, and psychological distress, while also improving long-term outcomes for patients.
This case illustrates a typical presentation with atypical frequency of newly diagnosed temporal lobe epilepsy in an adult, with characteristic symptoms and EEG findings of left anterior temporal lobe involvement. Early diagnosis and initiation of antiepileptic therapy are critical to controlling the patient’s seizures and improving quality of life. Regular follow-up with neurology and further diagnostic monitoring are essential to ensure appropriate management of this condition.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Address for Correspondence: Jason Tanner, MD, PhD, University of Utah Health, Department of Emergency Medicine, HELIX Building 5050, 30 N Mario Capecchi, Level 2, South, Salt Lake City, UT 84112. Email: jason.tanner@hsc.utah.edu
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Felker et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. McIntosh WC and Das JM. Temporal seizure. In: StatPearls, Tampa, FL: StatPearls Publishing; 2025. Accessed February 26, 2025. http:// www.ncbi.nlm.nih.gov/books/NBK549852/
2. Gavvala JR and Schuele SU. Epilepsy. JAMA. 2016;316(24):2686.
3. Frazzini V, Cousyn L, Navarro V. Chapter 27. Semiology, EEG, and neuroimaging findings in temporal lobe epilepsies. In: Miceli G, Bartolomeo P, Navarro V, eds. Handbook of Clinical Neurology, Vol 187, The Temporal Lobe; Amsterdam, Netherlands: RELX Group, Elsevier; 2022:489-518.
4. Devinsky O. Diagnosis and treatment of temporal lobe epilepsy. Rev Neurol Dis. 2004;1(1):2-9.
5. Mbizvo GK, Bennett K, Simpson CR, et al. Epilepsy-related and other
Felker et al.
Diagnosing Temporal Lobe Epilepsy in the Emergency Department: A Case Report
causes of mortality in people with epilepsy: a systematic review of systematic reviews. Epilepsy Res. 2019;157:106192.
6. Tatum WO. Mesial temporal lobe epilepsy. J Clin Neurophysiol 2012;29(5):356-365.
7. Crompton DE, Scheffer IE, Taylor I, et al. Familial mesial temporal lobe epilepsy: a benign epilepsy syndrome showing complex inheritance. Brain J Neurol. 2010;133(11):3221-3231.
8. Kasper BS, Kasper EM, Pauli E, et al. Phenomenology of hallucinations, illusions, and delusions as part of seizure semiology. Epilepsy Behav EB. 2010;18(1-2):13-23.
9. Asadi-Pooya AA and Farazdaghi M. Aura: epilepsy vs. functional (psychogenic) seizures. Seizure. 2021;88:53-55.
10. Durazzo TS, Spencer SS, Duckrow RB, et al. Temporal distributions of seizure occurrence from various epileptogenic regions. Neurology 2008;70(15):1265-1271.
11. Bianco M, Breviario S, Fraccalini T, et al. Epilepsy and syncope - a case report and narrative review of arrhythmias connected to temporal lobe epilepsy. J Electrocardiol. 2022;73:76-78.
12. Tinuper P, Bisulli F, Cerullo A, et al. Ictal bradycardia in partial epileptic seizures: autonomic investigation in three cases and literature review. Brain J Neurol. 2001;124(Pt 12):2361-2371.
13. Fisch L, Lascano AM, Vernaz Hegi N, et al. Early specialized care after a first unprovoked epileptic seizure. J Neurol 2016;263(12):2386-2394.
14. Hahn CD, Novy J, Rossetti AO. Comparison of lacosamide, levetiracetam, and valproate as second-line therapy in adult status epilepticus: analysis of a large cohort. Epilepsia. 2025;66(5):e73-e77.
15. Shehab AHIA, Simona T, Ion U, et al. Addressing the comorbidity between epilepsy and psychiatric disorders. Eur Psychiatry 2024;67(S1):S474-S475.
Ahmed Naseem, BS*
Mark Schoenborn, MD†
James Scheidler, MD†
William Hunter Barclay, MD‡
Garrett Volk, MD†
Section Editor: Steven Walsh, MD
McLaren Oakland Hospital, Department of Emergency Medicine, Pontiac, Michigan
J.W. Ruby Memorial Hospital, Department of Emergency Medicine, Morgantown
West Virginia
West Virginia University School of Medicine, Morgantown, West Virginia
Submission history: Submitted May 27, 2025; Revision received July 13, 2025; Accepted August 27, 2025
Electronically published December 7, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.47943
Introduction: Hyperkalemic emergencies can present with weakness, paralysis, sensorimotor deficits, and potentially fatal cardiac conduction abnormalities even in the absence of an elevated serum potassium. Common antihypertensive medications, such as angiotensin-converting enzyme inhibitors or angiotensin II receptor blockers, are associated with serum potassium elevations and can exacerbate hyperkalemia, especially in patients with renal impairment.
Case Report: We report a 49-year-old patient who presented to the emergency department six hours following an intentional ingestion of potassium supplements totaling 600 milliequivalents (mEq). The patient also reported chronic use of ibuprofen and losartan 50 mg. Symptoms on presentation included weakness, chest pain, and shortness of breath. Initial labs revealed a potassium > 10 mEq/L which was beyond the upper limit of assay detection for metabolic testing. Calcium gluconate, insulin with dextrose, albuterol, sodium bicarbonate, calcium chloride, fluids, and furosemide were sequentially administered. Initial electrocardiogram (ECG) showed tachycardia, a widened QRS complex without discernible P waves, and non-specific ST-segment changes. Following treatment, a repeat ECG demonstrated decreased heart rate, normal axis, and a decreased QT interval. Creatinine at presentation was 1.67 mg per deciliter (patient’s baseline) with repeat labs revealing a potassium of 9.6 mEq/L. Definitive treatment with placement of a central venous catheter for emergent dialysis was initiated.
Conclusion: This case illustrates how a patient’s regularly prescribed medication may complicate the management of an acute overdose. Prompt identification of a patient’s medications and supplements may expedite potentially life-saving interventions in a hyperkalemic emergency. [Clin Pract Cases Emerg Med. 2025;10(1):24-27.]
Keywords: hyperkalemic emergency; chronic kidney disease; case report.
INTRODUCTION
Hyperkalemic emergencies can be defined by various criteria related to elevated serum potassium and/or symptomatic presentation. Hyperkalemia is a common finding in patients with renal impairment and/or those who are prescribed renin-aldosterone-angiotensin-system inhibitors such as angiotensin-converting enzyme inhibitors (ACEi) or
angiotensin II receptor blockers (ARB). While hyperkalemia is generally defined as a serum potassium greater than 5.0 milliequivalents per liter (mEq/L), a serum potassium greater than 6.5 mEq/L is often considered a hyperkalemic emergency. The presence of muscle weakness/paralysis or cardiac conduction abnormalities can also be considered a hyperkalemic emergency regardless of the severity of serum
et al. Potassium Overdose in Patient with Chronic Kidney Disease on Losartan: A Case Report
potassium elevation.
Neurologic and cardiac manifestations commonly occur in the setting of acute rises in serum potassium < 6.5 mEq/L.1
Intentional drug overdoses, such as with this patient, are a common presentation in the emergency department (ED). However, reports of attempted suicide with potassium supplements in patients with chronic kidney disease (CKD) are rare and even rarer in those concomitantly taking medications that can inhibit potassium excretion.
A 49-year-old female with a history of hypertension, asthma, CKD stage III, stroke, and depression presented to the ED following a suicide attempt resulting in life-threatening hyperkalemia. Six hours prior to arrival, the patient intentionally consumed 30 pills containing 20 mEq of potassium. She also reported chronic use of nonprescribed ibuprofen and daily prescribed losartan 50 mg. The patient’s partner called emergency medical services after the onset of generalized weakness and confusion.
Upon arrival to the ED, the patient was weak but remained alert and oriented. She reported generalized weakness, chest pain, and shortness of breath. Vitals included the following: blood pressure, 131/83 millimeters of mercury; heart rate, 145 beats per minute (bpm); respiratory rate, 26 breaths per minute; and peripheral oxygen saturation, 100% on room air. Electrocardiogram (ECG) showed a heart rate of 68 bpm with right axis deviation, a significantly widened QRS complex without discernible P waves, and non-specific ST-segment changes (Image 1).
Serum potassium upon presentation was found to be greater than 10 mEq/L (reference range: 3.5-5.0 mEq/L) with an exact value beyond the upper limits of detection. The patient received intravenous (IV) calcium gluconate 2 grams (g), IV insulin 10 units, IV dextrose 50% 50 mL, nebulized albuterol 20 mg, IV sodium bicarbonate 100 mEq, IV calcium chloride 1g infusion, IV normal saline 1 L, and IV
Image 1. Pre-treatment electrocardiogram, prolonged QTc of 592 milliseconds, in a patient following intentional potassium overdose.
CPC-EM Capsule
What do we already know about this clinical entity?
Hyperkalemia management in emergency medicine is well-established, with progressive use of multiple agents providing timely, effective reduction of potassium levels.
What makes this presentation of disease reportable?
Our case highlights hyperkalemia management in a patient with renal impairment on a daily angiotensin II receptor blocker, causing rapidly rising potassium levels.
What is the major learning point?
Although standard therapies apply in an intentional potassium overdose with renal impairment, acuity rises due to rapid increases in potassium and subsequent poor excretion.
How might this improve emergency medicine practice?
Rapid recognition of hyperkalemia in renal impairment will aid emergency physicians in high-priority, complex cases.
furosemide 40 mg. Shortly after treatment, repeat labs revealed a serum potassium of 9.6 mEq/L. Repeat ECG showed a heart rate of 104 bpm, and the QRS decreased from 128 to 112 milliseconds (Image 2). Blood glucose was 220 mg per deciliter (dL). Complete blood count, prothrombin time, international normalized ratio, blood gases, and troponin levels were within normal ranges. Nephrology was consulted, hemodialysis was initiated, and the patient was transferred to the intensive care unit with subsequent recovery and discharge.
Nearly half of ED visits related to hyperkalemia result in mortality. Patients with CKD and/or those taking an ACEi/ ARB are at greater risk of developing hyperkalemia.2,3 Further, ACEi and ARB therapy have been associated with 10-38% of cases of hyperkalemia requiring hospitalization.2 These coexisting risk factors are of great importance in determining the prognosis of a hyperkalemic emergency given that taking higher doses of potassium does not always correlate with lethality. As illustrated by Simon, oral potassium doses of
Image 2. Post-treatment electrocardiogram, QTC decreased to 544 milliseconds, in a patient following management of her hyperkalemic emergency.
In contrast, whole bowel irrigation remains an option for many ingestions outside this short window. Whole bowel irrigation involves administering an osmotic laxative, such as polyethylene glycol, to help facilitate the expeditious removal of GI contents via the stool. However, while this method may be effective in reducing the total absorbable content of an acute ingestion, effectiveness is variable, and benefit in the literature remains debated. Importantly, this option would also be contraindicated in patients with hemodynamic instability or arrhythmia.9 Endoscopic removal has been described in the setting of a potassium salt bezoar. However, application of this technique is limited by the formulation of potassium ingested and the absence of clinically significant hyperkalemia in the report of this case.5
greater than 1,600 mEq/L can still be survivable while doses as low as 168 mEq/L may not.4
Management of hyperkalemia focuses on antagonizing the cardiac effects of potassium, shifting potassium into cells, and removing excess potassium from the body.5 Calcium, commonly administered as either calcium chloride or calcium gluconate, acts by directly antagonizing the cardiac membrane actions of potassium. Insulin’s role is to drive potassium into skeletal muscle cells via activation of the sodium potassium pump. If serum glucose is < 250 mg/dL, glucose should be administered to prevent hypoglycemia.6 Loop diuretics can also be added to promote potassium excretion via the urine. To then prevent further potassium absorption, oral potassium binding agents may be taken to promote excretion in the stool.7 Ultimately, hemodialysis may still be indicated for patients with acute or severe renal impairment.8
Although not used in this case because of proximity to rapid hemodialysis, gastrointestinal (GI) decontamination via both pharmacological and mechanical approaches remains a feasible way to limit continued potassium absorption in the setting of potassium ingestion. In cases of hyperkalemia resulting from enteral potassium exposure, decontamination represents one of the few treatment modalities capable of directly removing potassium from the body, whereas most standard therapies function by temporarily shifting potassium intracellularly.
The efficacy of GI decontamination is time sensitive and heavily dependent on the length of time from substance ingestion. Activated charcoal is often considered in the management of acute ingestion, but it is only effective in binding large, polarized organic compounds; generally it must be given within two hours of exposure, with the caveat of normal or near normal mental status to limit the risks of emesis and complications in airway management. Potassium is not bound by activated charcoal as it is a small, polarized, inorganic compound.
Long-acting, potassium binding agents remain an important adjunct for limiting the sustained absorption of extended formulation potassium supplements over a period of days.10 Examples more commonly used in practice include sodium polystyrene sulfonate and sodium zirconium cyclosilicate. Sodium polystyrene sulfonate is a non-specific cation-exchange resin that can be administered multiple times per day but carries a risk of GI complications, including colonic necrosis, and has questionable clinical efficacy.11 Sodium zirconium cyclosilicate, a selective potassium trapping agent, may be preferred as it is dosed daily and has a shorter onset of action.10
In this case, the use of sodium zirconium cyclosilicate was considered. However, its delayed onset of action—ranging from 1-4 hours post-ingestion—was deemed less favorable compared to immediate hemodialysis, which was readily available. Moreover, in the study by Amin et al, only 3.5% of patients had serum potassium > 6.5 millimoles per liter at baseline, and none had significant potassium ingestion as the etiology of their hyperkalemia.12 These differences limit the generalizability of those findings to our patient population. Despite these limitations, these pharmacologic agents may serve as a useful adjunct in cases where there is a delay in initiating dialysis or when managing ongoing enteral potassium absorption in conjunction with definitive therapy.
One of the unique features of this case report is that our patient presented with a baseline history of CKD and a creatinine of 1.67 mg/dL. Her chronic, nonsteroidal antiinflammatory drug use, daily ARB, and intentional overdose all contributed to the hyperkalemic emergency. Corroborating history from family, along with supporting ECG changes, allowed for prompt initiation of potassium-lowering therapies. As reported by Madan et al, intentional potassium overdoses requiring hemodialysis occur infrequently. In this case series following 11 patients over an 11-year period, only five presented with an initial potassium > 7 mEq/L.7 In a case detailing a patient with concomitant pacemaker failure, Muck et al furthered the idea of this rarity by reporting that very few isolated case reports exist in the literature with an acute serum potassium > 10 mEq/L.13
Naseem et al. Potassium Overdose in Patient with Chronic Kidney Disease on Losartan: A Case Report
Cases of intentional potassium overdose must be managed swiftly and aggressively to prevent life-threatening outcomes. Even without the availability of exact laboratory values, patients’ existing comorbidities and medication history can quickly enable clinicians to classify them as high risk and allow for prompt disposition determination.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Address for Correspondence: Garrett Volk, MD, J.W. Ruby Memorial Hospital, Department of Emergency Medicine, 1 Medical Center Drive, Morgantown, WV 26506, USA. Email: gavolk@hsc. wvu.edu.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Naseem et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Mount DB. Treatment and prevention of hyperkalemia in adults. In: Post TW, ed. UpToDate. UpToDate; 2025. Accessed March 13, 2025. https://www.uptodate.com/contents/treatment-and-prevention-of-
hyperkalemia-in-adults.
2. Raebel MA. Hyperkalemia associated with use of angiotensinconverting enzyme inhibitors and angiotensin receptor blockers. Cardiovasc Ther. 2012;30(3):e156-e166.
3. Davey M and Kuhn M. Slow-release potassium overdose: Is there a role for magnesium? Emerg Med Australas. 1999;11(4):263–271.
4. Simon G. Detection of fatal potassium overdose: a case report and review of the literature. Diagnostics (Basel). 2023;13(7):1339.
5. Guillermo PTJ, Carlos PHJ, Ivonne BAM, et al. Extended release potassium salts overdose and endoscopic removal of a pharmacobezoar: a case report. Toxicol Rep. 2014;1:209-213.
6. Harel Z and Kamel KS. Optimal dose and method of administration of intravenous insulin in the management of emergency hyperkalemia: a systematic review. PLoS One. 2016;11(5):e0154963.
7. Madan A, Morris C, Goggin A, et al. Oral potassium overdose: a case series. Clin Toxicol (Phila). 2021;59(11):963-968.
8. Bosse GM, Platt MA, Anderson SD, et al. Acute oral potassium overdose: the role of hemodialysis. J Med Toxicol. 2011;7(1):52-56.
9. Thanacoody R, Caravati EM, Troutman B, et al. Position paper update: whole bowel irrigation for gastrointestinal decontamination of overdose patients. Clin Toxicol (Phila). 2015;53(1):5-12.
10. Chaitman M, Dixit D, Bridgeman MB. Potassium-binding agents for the clinical management of hyperkalemia. P & T. 2016;41(1):43-50.
11. Sterns RH, Rojas M, Bernstein P, et al. Ion-exchange resins for the treatment of hyperkalemia: are they safe and effective? J Am Soc Nephrol. 2010;21(5):733-735.
12. Amin AN, Menoyo J, Singh B, et al. Efficacy and safety of sodium zirconium cyclosilicate in patients with baseline serum potassium level ≥ 5.5 mmol/L: pooled analysis from two phase 3 trials. BMC Nephrol. 2019;20(1):440.
13. Muck PM, Letterer S, Lindner U, et al. Beating the odds-surviving extreme hyperkalemia. Am J Emerg Med 2012;30(1):250.e1-250.e2504.
John Wahhab, MD
Iswarya Vimalan Jeya, BA
Jackson R. Huttner, MS
Section Editor: Joel Moll, MD
Chicago Medical School, Rosalind Franklin University of Medicine and Science, North Chicago, Illinois
Submission history: Submitted May 18, 2025; Revision received July 30, 2025; Accepted August 29, 2025
Electronically published November 16, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.47400
Introduction: Dislocations of the pisiform bone are rare, and literature on this injury is sparse. The uncommon nature of this condition, as well as limited data, makes recognition and diagnosis difficult, increasing the chances these injuries may be overlooked. Missing this diagnosis can lead to pain, reduced joint function, and nerve damage.
Case Report: We present a case of pediatric pisiform dislocation and discuss the diagnosis and treatment in an emergency department setting.
Conclusion: Prompt diagnosis and treatment of pisiform dislocations are vital to ensure favorable outcomes. [Clin Pract Cases Emerg Med. 2025;10(1):28-30.]
Keywords: pisiform bone; pisiform dislocation; pediatrics; wrist injuries; case report.
The pisiform bone is a carpal bone of the wrist within the flexor carpi ulnaris tendon, which functions to enhance flexion of the joint.1,2 Pisiform dislocations are rare, and there is little documentation in the literature. Diagnosing pisiform dislocations at an early stage can be difficult.1 These injuries often may go unnoticed, the consequence of which can result in significant pain for the patient and impairment of joint functionality.3,4 Cases in the current literature describe primarily adult patients.5 In this report we describe the diagnosis and treatment of a pediatric patient with a pisiform dislocation and a concomitant distal radius fracture, with the goal of highlighting the considerations when managing this rare condition.
An 11-year-old, right-handed boy presented to the emergency department (ED) after injury to his right wrist. He reported that he was at football practice doing a blocking drill when his teammate’s facemask hit his wrist. Emergency medical services responders placed a temporary splint and brought him to the ED. He complained of a sharp, stabbing pain in his wrist that was worse with movement and better with rest. Initial vital signs were temperature 98.7 °F; heart
rate 85 beats per minute; respiratory rate 18 breaths per minute; oxygen saturation 98% on room air; and blood pressure 120/67 millimeters of mercury. His physical exam revealed swelling, deformity, and tenderness of the right wrist, with light touch sensation intact distal to the injury. Range of motion of the wrist was decreased secondary to pain; there were no other signs of trauma on physical exam.
Radiographs of the right wrist were performed and showed an oblique fracture of the distal radial metadiaphysis with subcentimeter shortening and subcentimeter radial-sided displacement as well as mild anterior apex angulation. In addition, there was an ovoid, well-corticated ossific density that was suspicious for pisiform dislocation (Images 1 and 2). We obtained only two radiographic views, anterior-posterior and oblique. A lateral view could not be taken because of the patient’s inability to tolerate the imaging procedure due to pain.
Orthopedic consult did not recommend an attempt at closed reduction, instead recommending that he be transferred to a children’s hospital for urgent closed reduction. At the accepting hospital, pediatric orthopedic surgery was consulted. An attempt to reduce the pisiform dislocation by closed reduction was unsuccessful. He was placed in a splint and
discharged. The patient was asked to follow up in the hand surgery clinic; however, he was lost to follow-up.
DISCUSSION
The injury is described as a rupture of the anchoring ligaments of the pisiform, due to the powerful contraction of the flexor carpi ulnaris tendon during forced extension of the wrist.1 Pisiform dislocations are considered uncommon, with a lack of agreement on the preferred treatment method. In acute cases, pisiform dislocation injuries are addressed with closed reduction and immobilization in an attempt to stabilize the injury nonsurgically through the use of external support such as a plaster cast.6,7 Open reduction is only recommended when there are signs of neurological injury to the ulnar nerve, or when the patient has sustained serious crush injuries, or after the failure of attempted closed reduction.5,8 Open reduction is often accompanied by pinning or primary excision of the pisiform bone.4 Surgical removal of the pisiform offers better clinical outcomes due to a faster recovery and restoration of original function.6 A high incidence of pisiform instability and repeated dislocations have been reported after initial intervention, and they respond poorly to both open and closed reduction.4
Most cases in the literature involve adult patients and describe dislocations in the anterior and posterior directions,
CPC-EM Capsule
What do we already know about this clinical entity?
Reports of pisiform bone dislocation are sparse, leading to significant debate regarding optimal treatment.
What makes this presentation of disease reportable? We report a case of pisiform dislocation in a child with a concomitant distal radius fracture.
What is the major learning point?
Pisiform dislocations are frequently missed. Our goal was to shed light on this injury and how to diagnose and treat it.
How might this improve emergency medicine practice?
Timely management of pisiform dislocations is crucial to prevent chronic pain, disability, and nerve damage.
while distal dislocations are rarer.6,9,10 Our case differs from those reported, as it encompasses a pisiform dislocation in a pediatric patient,.5,7,9 Moreover, our patient had a concomitant oblique fracture of the distal radial metadiaphysis. The immediate identification and diagnosis of this pisiform dislocation in the ED should be noted, as it generated a focused treatment plan.
Pisiform dislocation injuries are reported to be misdiagnosed in emergency settings4 due to their infrequency and unfamiliar appearance on radiograph.1 The injury is often recognized only after prolonged wrist pain when the patient is directed to a specialist.4 In instances of delayed diagnosis, a direct excision of the pisiform is recommended.4 Further research and reporting of the results of different treatment options may improve diagnosis and reduce patient morbidity.
In this case report, we sought to address the management of this rare injury in a pediatric patient, to bring attention to the consideration of pisiform dislocation in hand and wrist trauma, and to emphasize the need for urgent treatment. Our initial management was conservative, based on the existing literature.6,7 We were fortunate to be able to make an expedient diagnosis and intervention, thus reducing the risk of pathological development of the joint. Identifying and managing the injury at an earlier stage can help avoid potential complications such as recurrence and instability of the
Image 2. Right wrist radiograph, oblique view, of a child with pisiform bone dislocation. Arrow indicates the dislocated pisiform.
Address for Correspondence: Jackson R. Huttner, MS, Chicago Medical School, Rosalind Franklin University of Medicine and Science, 3333 N Green Bay Rd 10 North Chicago, IL 60064. Email: Jackson.huttner@my.rfums.org.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Wahhab et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
pisiform bone.6,9,10 Missed diagnoses can contribute to difficulty associated with chronic wrist pain, ulnar nerve injury, and post-traumatic arthritis.11 Recurrent dislocations can result in neurological damage to the ulnar nerve and warrant an emergency reduction; such neurological injury may be avoided through a proper initial assessment.8
It is critical that all physicians be able to accurately diagnose and properly treat pisiform dislocations. Dislocation of the pisiform bone is a rare type of orthopedic injury, especially among the pediatric population, making it a frequently missed diagnosis. A missed or delayed diagnosis can lead to recurrent pain, decreased range of motion and functionality of the affected hand, permanent nerve damage, and need for surgical intervention. Conservative management including non-surgical intervention and closed reduction are recommended. Surgery should be reserved for cases that involve injury to the ulnar nerve or for chronic dislocations. Our report reveals the importance of prompt diagnosis and appropriate treatment of pediatric pisiform dislocations, thereby ensuring the most favorable patient outcomes and prevention of further complications.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
1. Mahmood F, Mehta M, Kakkar R. Pisiform dislocation. BMJ Case Rep. 2021;14(1):e237482.
2. Procházka O, Sánchez T, Kašpárková K. Fracture dislocation of the pisiform bone in 14-year-old boy—a case report. Medicina (Mex) 2024;60(4):532.
3. Saleh WR, Yajima H, Nakanishi A. Conservative treatment of the isolated dislocation of the pisiform bone. J Plast Surg Hand Surg 2014;48(4):283-284.
4. Petrou IG, Savioz-Leissing C, Gray A. Traumatic dislocation of the pisiform bone. J Hand Microsurg. 2018;10(1):37-40.
5. Lall A, Shephard N, Greenbaum S et al. Isolated carpal dislocation of the pisiform with distal radius fracture in two adults: a rare entity. J Hand Microsurg. 2020;12(3):215-218.
6. Afshar A, Tabrizi A, Shariyate MJ et al. Isolated pisiform dislocation after a seizure episode. Trauma Case Rep. 2024;52:101039.
7. Handoll HH and Madhok R. Closed reduction methods for treating distal radial fractures in adults. Cochrane Database Syst Rev 2003;2003(1):CD003763.
8. Giannetti A, Fidanza A, Passeri M et al. Pisiform bone dislocation in a pediatric patient: What is the best treatment? Case report and review of the literature. J Hand Microsurg. 2020;14(4):339-342.
9. Dias R, Alves-da-Silva T, Martinho C et al. Isolated distal pisiform dislocation: case report. Rev Bras Ortop. 2021;57(2):341-344.
10. Saglam F, Dagtas MZ, Sağlam S et al. Locked traumatic pisiform dislocation: a case report. JBJS Case Connect. 2019;9(4):e0278.
11. Choong Yoke Lin C and Hassan S. Atraumatic isolated dislocation of pisiform with ulnar nerve palsy. Cureus. 2023;15(9):e46042.
Colin Bashline, BS, NRP* Matthew Jester, MD†
Christopher Morris, DO†
Section Editor: Joel Moll,
MD
Ross/West View EMSA, Pittsburgh, Pennsylvania
Allegheny General Hospital, Department of Emergency Medicine, Pittsburgh, Pennsylvania
Submission history: Submitted June 18, 2025; Revision received August 9, 2025; Accepted August 29, 2025
Electronically published November 26, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.48649
Introduction: Foreign bodies in the external auditory canal are an uncommon presentation in emergency settings. Among adults, insects represent a frequent organic foreign body, often causing symptoms such as otalgia, tinnitus, vertigo, and anxiety. Prehospital management of such cases is rarely addressed in the medical literature, with minimal guidance available for emergency medical services (EMS) personnel. In this report we discuss their role in stabilizing patients and reducing discomfort through appropriate interventions.
Case Report: A 40-year-old male called EMS after a live insect entered his left ear, causing severe otalgia and distress. Prehospital medical personnel clinically confirmed the presence of the insect and assessed for signs of tympanic membrane perforation. A medical command physician authorized the use of 2% lidocaine to euthanize the insect, which alleviated movement-related discomfort within 20 seconds. Despite initial symptom relief, the patient experienced persistent fullness in the ear and was transported to a tertiary-care hospital. In the emergency department multiple removal attempts were made, with successful extraction using thin dressing forceps. No tympanic membrane perforation was noted, although minor trauma to the external auditory canal was present. The patient was discharged with ciprofloxacin-dexamethasone otic drops and return precautions.
Conclusion: Prehospital use of lidocaine for a live insect in the auditory canal may provide significant symptom relief while reducing the risk of further auditory canal trauma. This case underscores the importance of command-based support for EMS personnel to provide safe, evidence-based approaches for managing intra-aural insects in the field. [Clin Pract Cases Emerg Med. 2025;10(1):31-34.]
Keywords: foreign body; external auditory canal; prehospital care; emergency medical services; case report.
Foreign bodies of the external auditory canal are an infrequent but uniquely uncomfortable presentation to the emergency department (ED). Patients presenting with tinnitus, vertigo, otalgia, otorrhea, decreased hearing, hyperacusis and, in rare cases, persistent cough or hiccups should be evaluated for foreign bodies.1 Literature detailing the treatment of such patients is sparse and largely anecdotal. Prehospital guidance is limited, consisting of a handful of wilderness medicine guidelines.2,3
We present a case of a 40-year-old male who called emergency medical services (EMS) for a live insect that had flown into his left ear while he was in his garage. The patient complained of otalgia from the insect trying to move or fly. His spouse had unsuccessfully attempted to visualize the insect in the auditory canal prior to EMS arrival. Responders’ visual inspection showed no outward abnormalities of the ear and no drainage or bleeding from the auditory canal, but the
patient was restless and anxious. Persisting symptoms suggested that the insect was still in the auditory canal, prompting EMS to evaluate vital signs and contact a medical command physician for treatment orders.
After discussion with medical command to rule out signs suggesting a perforated tympanic membrane, such as hearing loss or bloody otorrhea, the EMS responders performed a lidocaine lavage. The patient’s head was rotated such that his left ear pointed up, and 2% lidocaine solution was instilled until the external auditory canal was filled. The patient was instructed to inform the EMS responders when the sensation of movement ceased. After approximately 20 seconds the movement ceased, and the patient’s head was then returned to anatomical position. The lidocaine drained by gravity and was absorbed with a paper towel. The external ear was inspected by flashlight, which yielded what appeared to be a single, detached antenna. Due to lack of visualization of the insect, the EMS responders made no attempt to remove it. While the patient’s distress and pain had subsided, a sensation of fullness remained in the left ear. He was transported to a nearby tertiary-care hospital for removal of the dead insect.
CPC-EM Capsule
What do we already know about this clinical entity?
Using lidocaine to kill a live insect in the ear prior to removal normally occurs in a hospital setting.
What makes this presentation of disease reportable?
We document prehospital treatment by emergency medical services responders who used lidocaine to kill an insect in a patient’s auditory canal before transporting him to the hospital.
What is the major learning point?
With a low clinical suspicion for tympanic membrane rupture, prehospital personnel should consult with medical command physicians and consider use of lidocaine.
How might this improve emergency medicine practice?
An animate foreign body in the ear is extremely uncomfortable. Killing the insect in the field minimizes suffering while the patient is transported to definitive medical care.
Additional history from the patient revealed that he initially attempted to remove the insect with his finger. This action caused the insect to be further wedged inside his external auditory canal. On arrival to the hospital, the patient was still very uncomfortable. An initial attempt to remove the insect with a cerumen loop was unsuccessful. A second, successful attempt entailed removing the insect (Image) with thin dressing forceps. There was no associated perforated tympanic membrane; however, the patient had minor trauma to the auditory canal. He was discharged with ciprofloxacindexamethasone 0.3-0.1% otic suspension drops and strict return precautions. Patient comfort during the extraction process had been improved as a result of the topical analgesia used by the paramedics.
Foreign bodies in the auditory canal are more common in pediatric patients and occur more frequently in males.1,4 While most of the foreign bodies seen in children are inorganic and iatrogenic, such as beads, toys and batteries, some of these objects are insects. As patient age increases the proportion of organic foreign bodies increases, with insects one of the most common culprits in adults.1 Insect foreign bodies tend to be isolated and accidental.
Insects pose a unique concern compared to other foreign bodies due to their ability to move. Insects tend to have difficulty walking backwards, which causes them to become trapped in the patient’s ear canal.5,7 After becoming stuck, the insects tend to move erratically, which can cause distressing sound and sensations, as well as pain. Multiple case reports have shown the significant and potentially fatal sequelae that insects in the ear can pose. Damage from insects in the middle and inner ear can lead to mastoiditis or facial nerve palsies either by direct trauma or localized swelling.6,8 A 2020 case
Bashline et al. Insect in the Ear—Response and Treatment of an Uncommon Prehospital Emergency: A Case Report
report from India described a patient who suffered from skull base osteomyelitis and mucormycosis that led to thrombi, neurological deficit, and death after two days of intensive treatment.7,9 While extreme, these complications illustrate the potential for animate foreign bodies to produce lifethreatening sequelae.
A key point to emphasize is the need for prompt referral to professional care. While emergency physicians are considered appropriate for initial removal attempts, there is strong preference for referral to otolaryngologists if initial attempts fail, or if timely initial referral can be made.4,6,8 While most patients do seek professional care, clinicians should be wary of the inclination to attempt home remedy; attempts at removal of insects have been reported to include cotton swabs, whole cloves of garlic, and hot oil poured into the auditory canal. These home remedies can result in worsening impaction, infection, burns, and trauma to the external auditory canal and tympanic membrane.4
While the need to remove the insect is obvious, the first step of treatment should focus on killing the insect to prevent further damage and reduce pain and distress.1,3,8,10 Multiple options exist for dispatching the insect, with the primary choices being mineral oil and lidocaine. A unique case report from Pittsburgh reported a patient with cockroaches in both ears.9,11 Clinicians used this opportunity to compare the efficacy of mineral oil vs lidocaine solution; the mineral oil swiftly killed the insect but made physical removal difficult.9,11 The lidocaine solution, by contrast, caused the roach to exit the ear canal “at a convulsive rate of speed.”9,11
The rates of efficacy of these two solutions have been evaluated by researchers under more controlled circumstances. Leffler et al evaluated 40 American cockroaches that had been administered mineral oil and varying concentrations of lidocaine; they concluded that mineral oil was significantly faster at killing the roaches, with lidocaine taking > 10 seconds longer.10,12 Differences in concentration did not seem to affect the speed at which lidocaine killed the roaches.10,12 Use of water was ruled out, as it took > two minutes to act.10,12 A study from Antonelli et al evaluated multiple solutions, finding ethanol and isopropyl alcohol to be the most effective agents for killing insects.11 However, mineral oil was still found to be more effective than lidocaine.11,13
Based on these studies and multiple anecdotal cases, mineral oil and 1-2% non-viscous lidocaine are the two most effective and readily available agents for killing an insect in the external auditory canal.2,8,10,11,13 Lidocaine is considered to have added benefit in its anesthetic properties. We recommend that caution and close discussion with medical command physicians be used to clinically evaluate risk of perforation of the tympanic membrane. Use of lidocaine with a perforated tympanic membrane can cause interactions with the membranous labyrinth, leading to severe vertigo and subsequent nausea and
vomiting.2,3,11,13 While both mineral oil and lidocaine would be effective in killing the insect and bringing immediate relief to the patients, mineral oil is not routinely stocked in ambulances; more importantly, lidocaine can be ototoxic if the tympanic membrane is perforated.12,14 Given this potential for toxicity, lidocaine cannot be empirically recommended for suspected aural insect foreign bodies.
Due to the presence of a live insect it would be difficult for EMS responders to examine for perforation of the tympanic membrane, even if an otoscope were available. In a study by Sousa et al that evaluated the clinical signs suggesting a perforated tympanic membrane, it was found that hypoacusis was the most frequent presenting symptom at 93.5% of the evaluated ears.13 Tinnitus was second at 16.4% of the sample population, followed by otorrhea at 9.4% and otalgia at 4.5%.13,15 If any of these symptoms are present, it would be recommended that lidocaine not be used, with prompt transport to the nearest ED the most appropriate course of action.
Once the insect has been killed, approaches to removal can vary depending on available equipment, position of the insect, and clinician preference. A strong light source and an otoscope are considered standard equipment for visualization. Alligator forceps, cerumen loops, Frazier suction tips, hooks, curettes, and tweezers have been suggested for foreign body removal, subject to availability.8,10 Referral to an otolaryngologist should be considered after a failed attempt at removal if the patient is immunocompromised or if there is pre-existing ear canal injury. Following removal, use of non-steroidal antiinflammatory drugs for pain control and fluoroquinolone otic drops for infection prevention are recommended.3
While case reports have documented in-hospital approaches to patients with intra-aural insects, prehospital literature is minimal, and little training exists for EMS personnel. Mineral oil and lidocaine can both be used to euthanize an intra-aural insect in consultation with a medical command physician. The benefits of prehospital intervention include pain control, relief of vertigo, and decreased risk of tympanic membrane rupture; these benefits are amplified for patients with extended transport times. While the risks and benefits of prehospital intervention are multifactorial, consideration should be given to the role that prehospital personnel can play in the treatment of intra-aural insect foreign bodies.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Insect in the Ear—Response and Treatment of an Uncommon Prehospital Emergency: A Case Report Bashline et al.
Address for Correspondence: Colin Bashline, BS, NRP, Ross/ West View EMSA, Ross/West View EMSA, 5325 Perrysville Ave, Pittsburgh, PA, 15229. Email: Cbashline@osteo.wvsom.edu
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Bashline et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Ansley JF and Cunningham MJ. Treatment of aural foreign bodies in children. Pediatrics. 1998;101(4):638-41.
2. Kizer KW. Intra-aural insects, lidocaine, and vertigo. Wilderness Environ Med. 2000;11(2):138-9.
3. Giltmier A, Aunins B, Ishman SL, et al. Management of live insects in the external auditory canal: a wilderness perspective. Wilderness Environ Med. 2022;33(3):318-23.
4. Ali YA and Irfan M. An annual audit of the ear foreign bodies in Hospital Universiti Sains Malaysia. Malays Fam Physician 2012;7(1):2-5.
5. Maddock DR and Fehn CF. Human ear invasions by adult scarabaeid
beetles. J Econ Entomol. 1958;51(4):546-547.
6. Chiu C-C and She Y-Y. Moth in the left ear. Ear Nose Throat J 2019;100(6):NP310.
7. Sikka K, Agrawal R, Devraja K, et al. Hazardous complications of animate foreign bodies in otology practice. J Laryngol Otol 2015;129(6):540-3.
8. Alex A, Philip A, Paulose AA, et al. Skull base osteomyelitis with secondary cavernous sinus thrombosis: a rare presentation of an animate foreign body in the ear. BMJ Case Rep. 2021;14(1):e236723.
9. Falcon-Chevere JL, Giraldez L, Rivera-Rivera JO, et al. Critical ENT skills and procedures in the emergency department. Emerg Med Clin North Am. 2013;31(1):29-58.
10. O’Toole K, Paris PM, Stewart RD, et al. Removing cockroaches from the auditory canal: controlled trial. N Engl J Med. 1985;312(18):1197.
11. Leffler S, Cheney P, Tandberg D. Chemical immobilization and killing of intra-aural roaches: an in vitro comparative study. Ann Emerg Med 1993;22(12):1795-8.
12. Antonelli PJ, Ahmadi A, Prevatt A. Insecticidal activity of common reagents for insect foreign bodies of the ear. Laryngoscope 2001;111(1):15-20.
13. Nader M and Saliba I. Ototoxicity of intratympanic docusate sodium and mineral oil in the guinea pig. Otolaryngol Head Neck Surg 2011;146(3):455-460.
14. Sousa LR, Fraga GA, Costa IS, et al. Diagnostic accuracy of the video otoscope in tympanic membrane perforation. Braz J Otorhinolaryngol. 2024;90(1):101336.
Alexander Adler, MD, MS
Anne Messman, MD, MHPE
Section Editor: Lev Libet, MD
Wayne State University School of Medicine, Department of Emergency Medicine, Detroit, Michigan
Submission history: Submitted May 5, 2025; Revision received August 7, 2025; Accepted October 16, 2025
Electronically published December 10, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem
DOI: 10.5811/cpcem.47332
Introduction: Ewing sarcoma is a relatively common neoplasm occurring in pediatric patients 10-20 years of age, commonly presenting with bone fracture, fever, and pain and swelling at the site of the primary tumor. Here we present an unusual case of Ewing sarcoma in the cervical spine leading to neurological symptoms including left lower extremity hemiparesis and left upper extremity hemiplegia.
Case Report: A 19-year-old Bengali-speaking male presented to the emergency department with a three-week history of left lower extremity hemiparesis and left upper extremity hemiplegia. Due to concern for spinal cord compression, a computed tomography of the cervical spine without contrast was obtained, which revealed a lucent lesion in the left fifth cervical (C5) vertebral body. Magnetic resonance imaging of the cervical spine revealed a left cervical extradural mass present from C3-C7. The patient subsequently underwent C3-C7 laminectomy with tumor decompression and fusion one week later. Surgical pathology revealed Ewing sarcoma. Following chemotherapy two months later the patient regained complete recovery of motor and sensory function in the left lower and left upper extremities.
Conclusion: It is important for emergency physicians to broaden their differential diagnosis when the physical examination reveals neurological deficits as exhibited in this case. A broader workup must be obtained that does not solely consist of head imaging but also includes imaging of the spine to prevent missing the diagnosis. [Clin Pract Cases Emerg Med. 2025;10(1):35-38.]
Keywords: Ewing sarcoma; neurological examination; upper motor neuron signs; case report.
Ewing sarcoma is a relatively common neoplasm occurring in pediatric patients, usually occurring between 10-20 years of age. The most common presenting symptoms include bone fracture, fever, and pain and swelling at the site of the primary tumor. The most common sites of Ewing sarcoma are the chest, legs, and pelvis, particularly around the growth plates.1,2 The cause is unknown, although accumulating evidence suggests a strong hereditary risk. The underlying mechanism in roughly 85% of cases involves a reciprocal translocation between chromosomes 11-22, which fuses the Ewing sarcoma breakpoint region 1 (EWSR1) gene of
chromosome 22 to the Friend leukemia virus integration 1 (FLI1) gene of chromosome 11. The resultant chromosomal translocation leads to the translation of a new EWS-FLI1 fusion protein, which can convert usually silent chromatin regions into fully active enhancers leading to oncogenesis.3
The most common symptoms associated with Ewing sarcoma involve pain and swelling over the primary tumor site with intermittent fevers and other symptoms common in inflammatory systemic illness. Here we present an unusual case of Ewing sarcoma in the cervical spine leading to neurological symptoms that included left lower extremity hemiparesis and left upper extremity hemiplegia.
A 19-year-old Bengali-speaking male presented to the emergency department (ED) with a three-week history of left-sided weakness. The history was predominantly obtained from the patient’s brother as the patient did not speak English and preferred to speak through his brother rather than through an interpreter. The patient’s brother stated that the patient had immigrated from Bangladesh several months prior to presentation and believed that he was lifting some heavy objects prior to symptom onset. Three weeks earlier the patient had developed significant difficulty moving his left arm and some difficulty walking. The symptoms had been progressing over the prior several weeks, which led him to visit his primary care doctor. The primary care doctor referred the patient to the ED out of concern for left lower extremity hemiparesis and left upper extremity hemiplegia, concerning for a cerebrovascular accident.
The patient had no known medical history. Physical examination revealed significant weakness in the left upper extremity, slight weakness in the left lower extremity, positive Babinski signs bilaterally, and a positive Romberg test. Due to the patient’s concerning neurological examination, a comprehensive metabolic panel, complete blood count, magnesium level, and computed tomography (CT) of the head and cervical spine without contrast were obtained. Neurology was consulted to evaluate the patient out of concern for potential central nervous system demyelination and spinal cord dysfunction.
Laboratory studies were significant for an elevated white blood cell count of 11,500 per microliter (μL) (reference range: 4,500-11,000/μL). All other lab values were within normal limits. Computed tomography of the cervical spine showed no acute findings; however, there was a lucent lesion in the left fifth cervical (C5) vertebral body extending into the transverse process (Images 1 and 2). Computed tomography of the head showed no acute intracranial hemorrhage or territorial infarct and subtle hypodensities along the bilateral precentral gyri superiorly.
Neurology subsequently evaluated the patient and requested magnetic resonance imaging (MRI) of the brain and cervical spine, both with and without gadolinium, to confirm the diagnosis before admitting the patient to their service for further evaluation and treatment. The MRI of the cervical spine revealed a left cervical extradural mass present from C3-C7 with mild homogenous enhancement measuring 7.1 x 4.9 x 3.8 cm. The mass expanded into the left neural foramina and effaced the thecal sac from the level of C3-C4 to C5-C6. It caused severe spinal canal stenosis from the level of the mid-C4 vertebral body to the mid-C5 vertebral body (Image 3).
The patient subsequently underwent C3-C7 laminectomy with tumor decompression and fusion one week later. Surgical pathology revealed Ewing sarcoma. Following the operation the patient was admitted to the neurocritical care unit and
CPC-EM Capsule
What do we already know about this clinical entity?
Ewing sarcoma commonly presents in patients from age 10 to 20 in the chest, legs, and pelvis.
What makes this presentation of disease reportable?
This case represents a unique case of Ewing sarcoma found in the cervical spine presenting with new focal neurological deficits.
What is the major learning point?
A complete neurological examination should be performed in patients presenting with a new onset of weakness to avoid missing the diagnosis.
How might this improve emergency medicine practice?
This case exemplifies the necessity of broadening the differential diagnosis to include spinal etiologies in patients presenting with focal neurological deficits.
Image 1. Transverse computed tomography of the cervical spine without contrast demonstrating lucent expansile lesion involving the fifth cervical vertebral body and transverse process (arrow), consistent with Ewing sarcoma.
discharged eight days later. He was instructed to return to the hematology oncology unit three days later for a multimodal chemotherapy regimen conducted as part of the Combination Chemotherapy in Treating Patients with Nonmetastatic Extracranial Ewing Sarcoma clinical trial.4 The patient received four cycles of chemotherapy, which he tolerated well. He was subsequently started on filgrastim for maintenance therapy. The patient was discharged approximately two months later with outpatient follow-up and a referral to radiation oncology. Physical examination conducted at the time of discharge documented complete recovery of motor and sensory function in the left lower extremity and the left upper extremity.
This case represents an unusual presentation of Ewing sarcoma that affected the cervical spine and caused neurological symptoms consisting of hemiparesis in the left lower extremity and hemiplegia in the left upper extremity. The patient had initially complained primarily of pain and discomfort in his left shoulder; he and his brother had assumed the pain was due to overuse from increased lifting at work. Due to the persistent nature of the symptoms the patient’s primary care doctor instructed him to present to the ED for a CT head due to concern for a cerebrovascular accident. Had the patient not received cervical spine imaging and only a CT head, as initially suggested by his primary care doctor and the neurology resident, the diagnosis would have been missed. This case highlights the importance of performing a thorough neurological examination in patients who present with weakness of any kind, and the importance of not becoming distracted by musculoskeletal complaints such as pain that often presents as the chief complaint. This patient
exhibited hemiparesis in his left lower extremity and hemiplegia in his left upper extremity indicative of a focal deficit necessitating further evaluation. Additionally, he exhibited a positive Babinski sign and positive Romberg test indicative of likely spinal cord compression affecting proprioception and upper motor neuron function. This was the rationale behind adding the CT of the cervical spine, which elucidated the underlying pathology.
Although involvement of the axial skeleton by Ewing sarcoma is rare, this case illustrates the importance of including it on the differential diagnosis when evaluating patients with musculoskeletal pain and neurological deficits. Few cases have been documented in the literature. Ilaslan et al conducted a retrospective review of 1,277 cases of Ewing sarcoma, 125 of which had a primary vertebral origin. Of these, four were found in the cervical spine. It is worth noting that in all the documented cases of primary vertebral origin, localized pain was the first symptom and was seen in all the cases; neurological deficits were present in 21 cases.5
Pain and swelling surrounding the primary tumor site are the cardinal symptoms of Ewing sarcoma.6 It is important that the emergency physician broaden the differential diagnosis in patients with these presenting complaints beyond musculoskeletal pathology and not be distracted by the patient’s chief complaint and history of present illness that may center around a potential musculoskeletal injury. This is particularly important when the physical examination reveals neurological deficits, as exhibited in this case, consistent with spinal cord compression. A broader workup must be obtained
EwS in the C-Spine Causing Left Lower Extremity Hemiparesis and Left Upper Extremity Hemiplegia Adler et al.
that does not solely consist of head imaging but also includes spine imaging to prevent missing the diagnosis. Additionally, it is important to keep in mind that pediatric osseous neoplasms do not always involve long bones and can involve the vertebral bodies as exhibited here.
1. Ewing Sarcoma Treatment. National Cancer Institute. Updated February 28, 2025. Accessed May 14, 2025. https://www.cancer.gov/ types/bone/patient/ewing-treatment-pdq#section/all
2. Goldman L and Schafer AI. Goldman’s Cecil Medicine. 24th ed. Philadelphia, PA: Elsevier Saunders; 2012:1326.
3. Riggi N, Suvà ML, Stamenkovic I. Ewing’s sarcoma. N Engl J Med 2021;384(2):154-64.
Address for Correspondence: Alexander Adler, MD, MS, CedarsSinai Medical Center, Department of Emergency Medicine, 8700 Beverly Boulevard, Los Angeles, CA 90048. Email: alexander.d.adler@gmail.com.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Adler et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
4. Leavey PJ, Laack NN, Krailo MD, et al. Phase III trial adding vincristine-topotecan-cyclophosphamide to the initial treatment of patients with nonmetastatic Ewing sarcoma: a Children’s Oncology Group report. J Clin Oncol. 2021;39(36):4029-38.
5. Ilaslan H, Sundaram M, Unni KK, et al. B. Primary Ewing’s sarcoma of the vertebral column. Skeletal Radiol. 2004;33(9):522-7.
6. Symptoms of Ewing’s Sarcoma. Bone Cancer Research Trust. Published October 2010. Accessed May 14, 2025. https://web. archive.org/web/20130130061013/http://bcrt.org.uk/bci_symptoms_ of_ewings_sarcoma.php.
Hanna Schindler, MS Leila Keeler, DO
Section Editor: Shadi Lahham, MD
Submission history: Submitted April 22, 2025; Revision received July 9, 2025; Accepted September 19, 2025
Electronically published December 10, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem
DOI: 10.5811/cpcem.47265
Introduction: Ectopic pregnancy is a serious pregnancy complication that occurs when a gestational sac implants outside the uterus, most commonly in the fallopian tubes. However, a rare form of ectopic pregnancy, the cesarean scar ectopic pregnancy, occurs within a prior cesarean section scar and is becoming more common as cesarean delivery rates continue to rise. Cesarean scar ectopic pregnancies are challenging to diagnose and pose significant risks, including rupture and hemorrhage, which can lead to maternal death.
Case Report: A 27-year-old woman presented to the emergency department with a 16-day history of abdominal pain and vaginal bleeding, initially believed to be her menstrual period. She had a history of one previous lower uterine segment cesarean section. On examination, her beta-human chorionic gonadotropin (β-hCG) levels were elevated, and transvaginal ultrasound revealed an empty uterus with a gestational sac within a cystic area of the cesarean scar. The patient was diagnosed with a cesarean scar ectopic pregnancy. Given the high rupture risk, she underwent laparoscopic surgery with dilation and curettage. Postoperative management included methotrexate, antibiotics, and analgesics. A follow-up β-hCG test showed a significant decline, confirming resolution of the ectopic pregnancy. At her two-week follow-up, the patient remained asymptomatic with no bleeding, and ultrasound confirmed no retained products of conception.
Conclusion: Cesarean scar ectopic pregnancies are a rare and life-threatening complication of pregnancy that require timely diagnosis and intervention. Early detection through transvaginal ultrasound and appropriate multidisciplinary management are critical to prevent adverse outcomes. This case highlights the importance of early recognition, classification, surgical decision-making, and standardized diagnostic protocols to improve outcomes and save lives. [Clin Pract Cases Emerg Med. 2025;10(1):39-42.]
Keywords: ectopic pregnancy; cesarean scar ectopic pregnancy; maternal morbidity and mortality; transvaginal ultrasound; women’s health.
An ectopic pregnancy occurs when a gestational sac implants outside the uterus; it is a known complication of pregnancy. This complication can present with vaginal bleeding and other non-specific and variable symptoms such as lower abdominal pain, nausea, and vomiting. Women in their first trimester who present to the emergency department (ED) with this complication have a prevalence for ectopic
pregnancy of 18%. It is easily misdiagnosed, sharing symptomatology with appendicitis, urinary calculi, early pregnancy loss, or trauma.1,2 Due to this symptomatic mimicry and resultant misdiagnosis, ectopic pregnancies are the leading cause of maternal mortality in the first trimester, with an incidence of 5-10% of all pregnancy-related deaths.2,3 Approximately 90% of ectopic pregnancies are implanted within the ampulla of the fallopian tube; however, a rare form
occurs when a gestational sac implants in a previous cesarean scar. Cesarean scar ectopic pregnancies occur in 4% of all ectopic pregnancies, as well as 1 in 2000 pregnancies in women who underwent at least one prior cesarean section.4 With the rate of cesarean deliveries steadily increasing and improved detection technology through sonographic imaging, the incidence of cesarean scar ectopic pregnancies has also risen.5
In this case report we highlight a challenging diagnostic situation that clinicians may encounter, which requires a high degree of clinical suspicion and often can require urgency. Cesarean scar ectopic pregnancies can present subtly and carry a high risk for uterine rupture and catastrophic hemorrhage, making early recognition critical in the emergency setting. This case report highlights key symptomatic features that support early diagnosis and guideline-based classification of cesarean scar ectopic pregnancy. It also emphasizes the value of clinical–imaging correlation in guiding treatment decisions, as well as the resulting management outcomes.
A 27-year-old female presented to the ED with a 16-day history of abdominal pain and vaginal bleeding, which she initially believed to be her menstrual period. Three months earlier, she had undergone a lower uterine segment cesarean section for fetal distress. Her post-surgical bleeding had resolved approximately two months prior, but the onset of these new symptoms prompted her to seek medical attention.
The patient’s full obstetric history included six total previous pregnancies, three term births, two vaginal deliveries, one lower transverse cesarean section previously disclosed, and two abortions. Her last menstrual period had been reported approximately one month prior to her presentation to the ED. She had no history of chronic medical illnesses and was not taking any medications at the time of her ED arrival.
General physical exam was normal with the only pertinent positive being suprapubic and lower left quadrant abdominal tenderness. Her blood pressure was 119/74 millimeters of mercury (mmHg), and her temperature was 37 °C. On admission, her beta-human chorionic gonadotropin (β-hCG) level was 9,623 international units per milliliter (IU/mL) (reference range: < 5 IU/L), and after admission, a retest 48 hours later, per the standard guideline set by the American College of Emergency Physicians (ACEP), showed the β-hCG value was 16,587 IU/mL, an increase of over 66%, consistent with early pregnancy progression. A transvaginal ultrasound revealed an empty uterine cavity, and a possible gestational sac within a cystic region of the cesarean section scar, near the endometrium. At this point, the patient was admitted for treatment of an ectopic pregnancy, likely within a previous cesarean section scar.
Treatment options of oral methotrexate and surgical resection were considered, and risks and benefits were discussed with the patient. Surgical resection was recommended due to the high risk of rupture and hemorrhage based on the location of the ectopic pregnancy. The patient opted for laparoscopic and
What do we already know about this clinical entity? Cesarean scar ectopic pregnancy is a rare but increasingly recognized complication in patients with prior cesarean delivery, carrying a significant risk of uterine rupture.
What makes this presentation of disease reportable? Subtle symptoms in a stable patient with a history of cesarean delivery led to early diagnosis and coordinated multidisciplinary care.
What is the major learning point?
Early recognition, classification, and prompt intervention are vital for reducing risk of rupture and maternal morbidity and mortality.
How might this improve emergency medicine practice?
Clinicians should have a heightened awareness in post-cesarean patients with abdominal pain or bleeding, emphasizing early ultrasound and multidisciplinary evaluation.
transvaginal ultrasound-guided removal via dilation and curettage. The patient underwent general anesthesia and sterile vaginal preparation. Local anesthesia was administered, followed by an infraumbilical incision. A secondary 5-mm trocar was inserted for visualization.
Upon laparoscopic visualization, dense adhesions were noted between the omentum and uterine fundus, as well as between the anterior peritoneal reflection and the lower uterine segment, likely sequelae of a prior cesarean section, and a known risk factor for cesarean scar ectopic pregnancy. Intraoperative transabdominal ultrasound confirmed a gestational sac implanted within the cesarean scar. Under ultrasound guidance the cervix was dilated, and a suction curette was introduced into the scar site. A total of eight suction passes were performed until transvaginal ultrasound confirmed evacuation of all products of conception (Image 1). Hemostasis was achieved, and laparoscopy confirmed no evidence of uterine perforation. Fascia and skin were closed in layers with absorbable sutures, and the incision was sealed with Dermabond™ (Ethicon, Inc, Somerville, NJ).
Postoperatively, the patient was given one dose of methotrexate and started on doxycycline 100 mg by mouth once daily for five days, methylergonovine maleate 0.2 mg by mouth every six hours for one day, and hydrocodone-acetaminophen, every six hours as needed for moderate pain for three days. The
et al. Implanted in the Scar: A Case Report of Diagnosis and Management of Cesarean Scar Ectopic Pregnancy
interpreted in conjunction with transvaginal ultrasound findings to determine pregnancy location and viability. A cesarean scar ectopic pregnancy was ultimately diagnosed after a hyperechoic, cyst-like structure was visualized within the cesarean scar tissue (Image 2) without anechoic or echogenic free fluid present in the pouch of Douglas.7,8
patient was also instructed to continue her combined oral contraceptive pill (ethinyl estradiol/norethindrone/ferrous fumarate) daily and cephalexin 500 mg by mouth four times daily for 10 days, which was previously started in the ED. A follow-up β-hCG test showed a significant decline, confirming resolution of the ectopic pregnancy. At her two-week follow-up, the patient remained asymptomatic with no further vaginal bleeding. Ultrasound imaging confirmed the absence of retained products of conception, and the β-hCG was negative at this time.
An ectopic pregnancy was considered among the differential diagnoses, alongside miscarriage, when the patient presented to the ED with an initial β-hCG level of 9,623 IU/ mL. At that time the patient was admitted. A repeat β-hCG measurement, 48 hours later, showed an increase to 16,587 IU/L, representing a rise of more than 66%, which is consistent with the expected increase seen in a normal early pregnancy. However, transvaginal ultrasound showed an empty uterus despite the patient’s β-hCG level being well above the discriminatory zone of 1,500-2000 milli-IU/mL, at which point gestational structures on transvaginal ultrasound are typically visible. These combined findings are what prompted the diagnosis of ectopic pregnancy.7
Serial β-hCG measurements are a valuable tool in assessing early pregnancy, as the rate of rise can inform clinical decision-making. Typically, a rise of at least 35-66% over 48 hours is expected in a viable intrauterine pregnancy. However, a plateau or suboptimal rise may suggest a nonviable pregnancy or ectopic pregnancy, although it is not confirmatory and transvaginal ultrasound is also needed. According to current clinical guidelines, including those from ACEP and the American College of Obstetricians and Gynecologists, serial β-hCG measurements should always be
Cesarean scar ectopic pregnancies head special consideration through typing based on the site of implantation. Type 1 occurs when the gestational sac develops in the myometrium and grows toward the cervico-isthmic space or uterine cavity, while type 2 occurs when the gestational sac grows toward the bladder and abdominal cavity.8 In the case of this patient, type 1 classification was determined due to the nature of ectopic positioning within the scar, growing toward the uterine cavity. Rupture and hemorrhage potential is of great concern in cesarean scar ectopic pregnancies due to increased vasculature within the uterine segment; thus, in the case of our patient, uterine rupture potential increased the emergent nature of her ectopic pregnancy presentation.9 Additionally, due to the β-hCG elevation > 5,000 IU/mL, methotrexate monotherapy was not an appropriate expectant management based on increased possibility of treatment failure at these levels in addition to the high risk of rupture due to positioning.7
Intrauterine excision and methotrexate as combination therapy was determined to be the necessary treatment modality. Dilation and curettage of the gestational sac with laparoscopic uterine visualization was indicated based on type-1 positioning and the patient’s good hemodynamic stability. The laparoscope was used to ensure the gestational sac did not breach the full width of the myometrium or the serosa of the uterus and for visualization during dilation and curettage to ensure no perforation occurred. If the gestational sac had perforated the uterine wall, laparoscopic removal or
Implanted in the Scar: A Case Report of Diagnosis and Management of Cesarean Scar Ectopic Pregnancy Schindler et al.
laparotomy could have been used for removal and to control intra-abdominal bleeding. Additionally, laparoscopic intervention and removal can be beneficial for reduction in recurrence due to the ability for scar revision by removal and reclosure of the scar tissue area; however, recurrence potential is still present, especially in patients without scar revision. Patients should be informed of potential risk and serious sequelae of a recurrence of cesarean scar ectopic pregnancy.10
Cesarean scar ectopic pregnancies are a rare, life-threatening maternal risk and a clinically challenging diagnosis. Given their non-specific presentation, ranging from mild abdominal pain to vaginal bleeding, a missed diagnosis could lead to delayed treatment resulting in rupture, hemorrhage, and maternal death. A training protocol that includes a multidisciplinary team approach consisting of emergency medicine, obstetrics and gynecology, radiology, and surgical specialists when needed can facilitate timely diagnosis and allow for earlier treatment and better patient outcomes. Early evaluation and point-of-care ultrasound should be considered with heightened suspicion of cesarean scar ectopic pregnancy with a presentation of fitting symptomatology and history of a previous cesarean delivery. Especially in resourcelimited settings, where access to advanced imaging and specialist consultation may be delayed, early point-of-care assessment and rapid response for available treatment could be life-saving.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Address for Correspondence: Hanna Schindler, MS, Orlando College of Osteopathic Medicine, 7011 Kiran Patel Drive, Winter Garden, FL 34787. Email: hschindler@students.ocom.org
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Schindler et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Vadakekut ES and Gnugnoli DM. Ectopic pregnancy. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2025. https:// www.ncbi.nlm.nih.gov/books/NBK539860/. Accessed June 3, 2025.
2. Mullany K, Minneci M, Monjazeb R, et al. Overview of ectopic pregnancy diagnosis, management, and innovation. Women’s Health (Lond). 2023;19:17455057231160349.
3. Fylstra DL. Ectopic pregnancy not within the (distal) fallopian tube: etiology, diagnosis, and treatment. Am J Obstet Gynecol 2012;206(4):289-299
4. Herman A, Weinraub Z, Avrech O, et al. Follow up and outcome of isthmic pregnancy located in a previous caesarean section scar. BJOG. 1995;102(10):839-841
5. Maheux-Lacroix S, Li F, Bujold E, et al. Cesarean scar pregnancies: a systematic review of treatment options. J Minim Invasive Gynecol. 2017;24(6):915-925.
6. Hahn SA, Promes SB, Brown MD; American College of Emergency Physicians Clinical Policies Subcommittee (Writing Committee) on Early Pregnancy. Clinical policy: critical issues in the initial evaluation and management of patients presenting to the emergency department in early pregnancy. Ann Emerg Med. 2017;69(2):241250.e20.
7. Morente LS, León AIG, Reina MPE, et al. Cesarean scar ectopic pregnancy—case series: treatment decision algorithm and success with medical treatment. Medicina (Kaunas). 2021;57(4):362.
8. Surette A and Dunham SM. Early pregnancy risks. In: DeCherney AH, Nathan L, Laufer N, Roman AS, eds. Current Diagnosis & Treatment: Obstetrics & Gynecology, 12th ed. Columbus, OH: McGraw-Hill Education; 2019. Accessed June 3, 2025. https:// accessmedicine.mhmedical.com/content.aspx?bookid=2559§ion id=206959029
9. Lin R, DiCenzo N, Rosen T. Cesarean scar ectopic pregnancy: nuances in diagnosis and treatment. Fertil Steril. 2023;120(3 Pt 2):563-572.
10. Shen F, Lv H, Wang L, et al. A comparison of treatment options for type 1 and type 2 caesarean scar pregnancy: a retrospective case series study. Front Med (Lausanne). 2021;8:671035.
John Porter, MSN, RN*
Chad P. Liedl, RN†
Kevin P. Steever, Paramedic†
Hunter D. Lumby, Paramedic†
Chase M. Hanson, EMT†
Hannah Rae R. Vaden, AuD‡
Aaron B. Klassen, MD, MA§
Section Editor: Lev Libet, MD
Mayo Clinic, Cardiac Catheterization Laboratory, Rochester, Minnesota
Mayo Clinic, Ambulance Service, Rochester, Minnesota
Mayo Clinic, Division of Audiology, Rochester, Minnesota
Mayo Clinic, Department of Emergency Medicine, Rochester, Minnesota
Submission history: Submitted March 31, 2025; Revision received June 25, 2025; Accepted August 28, 2025
Electronically published December 15, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.47095
Introduction: We present the case of a patient with the unusual occurrence of phantom arm pain and an acute exacerbation of chronic tinnitus during an ST-segment elevation myocardial infarction (STEMI).
Case Report: A 56-year-old man was having several classic symptoms associated with acute coronary syndrome, along with perceived pain in an arm lost years earlier in a traumatic accident and a sudden worsening of his chronic tinnitus. Emergency medical services responded and diagnosed a STEMI on scene. A 100% occlusion of his right coronary artery was rapidly identified in the hospital and treated with the deployment of two drug-eluting stents. After the procedure his symptoms resolved. He was discharged without incident two days later.
Conclusion: Whereas arm pain is a well-documented presenting symptom of acute coronary syndrome, phantom limb pain and exacerbation of tinnitus have been rarely reported in the literature. [Clin Pract Cases Emerg Med. 2025;10(1):43-45.]
Keywords: arm pain; case report; phantom limb pain; STEMI; tinnitus.
The diagnosis of acute coronary syndrome is not always straightforward. Although chest pain is the hallmark symptom, it can be accompanied by a spectrum of atypical symptoms. These can include nausea, vomiting, dyspnea, radiation of pain to the arm or jaw, diaphoresis, fatigue, lightheadedness, back pain, headache, otalgia, sore throat, hand pain, and anxiety, among others.1-4 Up to 44% of patients with non-ST-segment elevation myocardial infarction (STEMI) and 27% of patients with STEMI do not have chest pain.5 Our case describes a patient with STEMI who presented with phantom limb pain and exacerbation of chronic tinnitus.
A 56-year-old man with a history of acute coronary syndrome, obesity, and obstructive sleep apnea presented with worsening left arm pain. The patient had sustained a left upper extremity amputation up to his shoulder from a motor vehicle accident years prior. He also was a tobacco user and experienced chronic tinnitus and neuropathy in his lower extremities. Approximately 10 years earlier the patient underwent a cardiac stress test after having an episode of chest pain that he described as like “being punched in the chest.” This led to a hospital admission and a stress test at an outside facility, although he was unsure of the details of the stress test, and no records could be located. No interventions were performed or medications prescribed.
He had been experiencing chronic and unchanging paresthesias in his phantom limb since the amputation. On the day of presentation, a sudden onset of chest pain and pressure developed in the center of his chest, which he described as sharp and rated 7 (on a scale of 0-10). The pain radiated to his right arm and left shoulder. There was also substantial worsening of the chronic paresthesia in his left upper phantom limb, which he described as “someone trying to pull my arm off.” The chest pain decreased to 4 (on a scale of 0-10) after lying down, but he then became lightheaded, dyspneic, diaphoretic, pale, clammy, and reported “feeling weird.” In addition, he noted an “extreme” increase in his baseline tinnitus. He then had a syncopal episode witnessed by his significant other who contacted emergency medical services (EMS).
Electrocardiography obtained by EMS showed a STEMI (Image 1), and the cardiac catheterization lab was alerted from the field. Initial vital signs obtained by EMS revealed the following: blood pressure, 129/73 millimeters of mercury; heart rate, 110 beats per minute; respiratory rate, 32 breaths per minute; and oxygen saturation 94% on room air. Electrocardiography performed on arrival to the emergency department showed a worsening elevation in the inferior leads and atrial fibrillation. His troponin T value measured by Elecsys Troponin T Gen 5 STAT (Roche Diagnostics Corporation, Indianapolis, IN) was 608 nanograms/Liter (ng/L) (reference range: ≤ 15 ng/L).
He was found to have 100% occlusion of his distal right coronary artery, which was treated with two drug-eluting stents (Image 2). After percutaneous coronary intervention, his symptoms resolved, including improvement in his phantom limb paresthesia to baseline. During his hospital stay he was newly diagnosed with diabetes and hyperlipidemia and was discharged two days after admission. On follow-up, the patient continues to do well after significant lifestyle modifications and management of his chronic diseases.
This case is unique in that it illustrates yet another atypical symptom that can be present in a patient experiencing acute coronary syndrome. A lack of chest pain does not rule
CPC-EM Capsule
What do we already know about this clinical entity? The underlying pathophysiology and constellation of presenting symptoms in ST-segment elevation myocardial infarction (STEMI) are well defined.
What makes this presentation of disease reportable? We discuss a rare presenting symptom of arm pain in a phantom limb, along with a worsening of chronic tinnitus, in a patient with STEMI.
What is the major learning point?
Patients experiencing an episode of acute coronary syndrome may not present with symptoms more commonly associated with STEMI.
How might this improve emergency medicine practice?
This case highlights uncommon presenting symptoms in a patient with STEMI.
out a STEMI. Literature suggests that 20-30% of patients experiencing an episode of acute coronary syndrome have no chest pain at all.6, 7 Pain in the left arm is a known symptom of an MI.8 Stimuli arising in the myocardium during an acute coronary syndrome episode activate the same area in the brain that is activated by signals from somatic structures such as the arm. Arm pain is thought to arise from a convergence of inputs from cardiac and somatic inputs in the trigeminal nucleus located in the brainstem.9 Misinterpretation of these two inputs by the brain leads to the referred pain.8
Left-arm phantom pain, however, a symptom not
Porter et al. Phantom Arm Pain and Tinnitus in a Patient with ST-Segment Elevation Myocardial Infarction
previously seen by any member of the patient’s care team, is an uncommon symptom of an MI. The literature describing such a phenomenon is limited. Martin et al10 described a 49-year-old man with exertional chest pain that radiated into a phantom limb; the pain resolved after the patient had coronary artery bypass grafting surgery. Cohen and Jones11 reported two instances of patients with chest and phantom left-arm pain accompanied by evidence of damage to the myocardium.
Tinnitus is an awareness of sound with no external auditory stimuli and is usually a symptom of hearing loss or a change in hearing.12 A weak association between tinnitus and heart disease has been reported.13 Tinnitus has been correlated with a higher incidence of cardiac events but not of death due to cardiovascular disease.14 Arterial stiffness, which is a known predictor of cardiovascular events and mortality,15 is significantly associated with both the development and severity of tinnitus.16 Stress also is known to be related to the intensity of tinnitus symptoms. Neuroplasticity as an adaptation to stress is a key component in the progression of tinnitus,17 and stressful events can lead to a worsening of tinnitus.18, 19 We postulate that the increased stress, both conscious and subconscious, caused by the STEMI led to the worsening of our patient’s tinnitus from the chronic baseline level.
This case highlights the unusual symptoms that can manifest during a STEMI and provides a telling anecdotal report of the impact that rapid treatment, along with lifestyle changes, can have on a patient’s quality of life.
Patient consent has been obtained and filed for the publication of this case report.
Address for Correspondence: John Porter, MSN, RN, Cardiac Catheterization Laboratory, Mayo Clinic, 200 First St SW, Rochester, MN 55905. Email: Porter.John@mayo.edu.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The Scientific Pubcliations staff at Mayo Clinic provided editorial consultation, proofreading, and administrative and clerical support.
Copyright: © 2025 Mayo Foundation for Medical Education and Research. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/licenses/by/4.0/
1. Angerud KH, Sederholm Lawesson S, Isaksson RM, et al. Differences in symptoms, first medical contact and pre-hospital delay times between patients with ST- and non-ST-elevation myocardial infarction. Eur Heart J Acute Cardiovasc Care. 2019;8(3):201-7.
2. Bohamad AH, Buali HH, Aljasem JM, et al. Comparing gastroesophageal reflux disease (GERD) and non-GERD patients based on knowledge level of acute myocardial infarction symptoms, risk factors and immediate action taken in Eastern Province, Saudi Arabia. Cureus. 2023;15(2):e35309.
3. Schulte KJ and Mayrovitz HN. Myocardial infarction signs and symptoms: females vs. males. Cureus. 2023;15(4):e37522.
4. Khan IA, Karim HMR, Panda CK, et al. Atypical presentations of myocardial infarction: a systematic review of case reports. Cureus 2023;15(2):e35492.
5. Canto AJ, Kiefe CI, Goldberg RJ, et al. Differences in symptom presentation and hospital mortality according to type of acute myocardial infarction. Am Heart J. 2012;163(4):572-9.
6. Writing Committee M, Anderson HVS, Masri SC, et al. 2022 ACC/ AHA key data elements and definitions for chest pain and acute myocardial infarction: a report of the American Heart Association/ American College of Cardiology Joint Committee on Clinical Data Standards. J Am Coll Cardiol. 2022;80(17):1660-700.
7. Kwok CS, Bennett S, Holroyd E, et al. Characteristics and outcomes of patients with acute coronary syndrome who present with atypical symptoms: a systematic review, pooled analysis and meta-analysis. Coron Artery Dis. 2025;36(3):240-51.
8. Foreman RD, Garrett KM, Blair RW. Mechanisms of cardiac pain. Compr Physiol. 2015;5(2):929-60.
9. Kikuta S, Dalip D, Loukas M, et al. Jaw pain and myocardial ischemia: a review of potential neuroanatomical pathways. Clin Anat 2019;32(4):476-9.
10. Martin WR, Margherita A, Amsterdam E. Phantom angina. Chest 1994;105(4):1271-2.
11. Cohen H and Jones HW. The reference of cardiac pain to a phantom left arm. Br Heart J. 1943;5(1):67-71.
12. Makar SK. Etiology and pathophysiology of tinnitus - a systematic review. Int Tinnitus J. 2021;25(1):76-86.
13. Ausland JH, Engdahl B, Oftedal B, et al. Tinnitus and cardiovascular disease: the population-based Tromso Study (2015-2016). BMJ Public Health. 2024;2(2):e000621.
14. Zhang YP, Gao QY, Gao JW, et al. The association between tinnitus and risk of cardiovascular events and all-cause mortality: insight from the UK Biobank. Acta Cardiol. 2024;79(3):374-82.
15. Bonarjee VVS. Arterial stiffness: a prognostic marker in coronary heart disease. available methods and clinical application. Front Cardiovasc Med. 2018;5:64.
16. Bayraktar C and Tasolar S. Relationship between increased carotid artery stiffness and idiopathic subjective tinnitus. Eur Arch Otorhinolaryngol. 2017;274(5):2125-30.
17. Patil JD, Alrashid MA, Eltabbakh A, et al. The association between stress, emotional states, and tinnitus: a mini-review. Front Aging Neurosci. 2023;15:1131979.
18. Yıldırım N, Aksoy S, Erdoğan O. Role of stressful life events as the precipitating factor for chronic tinnitus. J Hear Sci. 2017;7(2):138.
19. Elarbed A, Fackrell K, Baguley DM, et al. Tinnitus and stress in adults: a scoping review. Int J Audiol. 2021;60(3):171-82.
Michael Rosselli, MD*
Alejandro Sanoja, MD†
Matthew Apicella, DO*
Section Editor: Joel Moll, MD
Mount Sinai Medical Center, Department of Emergency Medicine, Miami Beach, Florida University of Florida, Department of Emergency Medicine, Gainesville, Florida
Submission history: Submitted May 3, 2025; Revision received June 28, 2025; Accepted August 27, 2025
Electronically published January 5, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.47307
Introduction: Acute lymphoblastic leukemia (ALL) is typically a childhood disease but may present in older patients in rare occurrences. Due to its significant morbidity and mortality, early diagnosis is crucial. The symptoms of ALL may be non-specific, making the initial diagnosis difficult leading to delayed treatment.
Case Report: We present the case of a 34-year-old, healthy male runner presenting to the emergency department with a common complaint of bilateral knee pain, who was ultimately diagnosed with ALL with signs of tumor lysis syndrome leading to premature death.
Conclusion: We discuss the presenting symptoms of acute lymphoblastic leukemia, which may include joint or knee pain as well as leukemic arthritis. We further discuss the importance of clinicians maintaining a high level of suspicion for the “bounce-back” patient and avoiding taking cognitive shortcuts. [Clin Pract Cases Emerg Med. 2025;10(1):46-49.]
Keywords: acute lymphoblastic leukemia; sports medicine; emergency medicine; case report.
Acute lymphoblastic leukemia (ALL) accounts for 12% of all leukemia cases worldwide.1 It is normally a disease of childhood with typical presentation in children < 6 years of age, although a second peak of incidence typically occurs at ages > 60.1 The presenting symptoms of ALL are often vague but can include joint and bone pain.1,2 This case is unique in that the disease presented in an otherwise healthy, 34-year-old active individual.
We present the case of a 34-year-old, previously healthy male runner who presented to the emergency department (ED) for intermittent bilateral posterior knee pain for one month, worse in the morning after he awoke. The patient had been evaluated twice in the ED since the onset of his symptoms and discharged home each time with orthopedic follow-up, which he did not follow through with due to time constraints secondary to his job. Radiographs performed of the bilateral
knees on his previous visit were remarkable only for mild bilateral joint effusions, without fracture or evidence of arthritic changes. He also. Underwent an unremarkable duplex ultrasound of the bilateral lower extremities.
Considering he was an avid runner and worked in construction, the treating physicians on prior visits concluded that his symptoms were likely musculoskeletal in etiology. The patient denied a history of fever/chills, easy bleeding or bruising, night sweats, and weight loss. Physical exam findings on our evaluation were negative for any strength deficits, joint laxity, restricted range of motion, swelling, warmth, or skin changes. He stated that he took ibuprofen, which normally relieved his symptoms. When the pain persisted he returned to the ED a third time.
Bloodwork was drawn, and radiographs repeated on this visit were again found to be unremarkable. Blood analyses revealed a white blood cell count of 395,000 microliters (µL) with 33% blasts (reference range: 4.80–10.80 10*3/µL), hemoglobin of 3.9 grams per deciliter (g/dL) (12.0-16.0 g/dL),
et al A Case Report of Acute Lymphoblastic Leukemia Presenting as Bilateral Knee Pain in a Healthy Runner
and a platelet count of 38,000 µL (reference range: 150-450 10*3/µL). The patient was also noted to have an acute kidney injury with a creatinine of 1.35 milligrams (mg)/dL (0.55-1.02 mg/dL), potassium of 8.7 millimoles per liter (mmol/L) (3.5-5.1 mmol/L), phosphorus of 4.5 mg/dL (2.5-4.5 mg/dL), calcium of 8.0 mg/dL (8.5-10.1 mg/dL), and uric acid level of 11.7 mg/dL (reference range: 3.4-7.0 mg/dL). The electrocardiogram showed normal sinus rhythm with no significant T wave changes.
He was transfused with two units of packed red blood cells and treated for his hyperkalemia with one gram of calcium gluconate, 10 units of regular insulin with 1 ampule of D50, 50 milliequivalents of sodium bicarbonate, and 2.5 mg of albuterol nebulizer. He was also given 4.5 mg of rasburicase and two liters of normal saline for concern of tumor lysis syndrome. All medications and fluid boluses were given once. Oncology and nephrology were consulted from the ED. Both services agreed with current management and recommended admission to the intensive care unit (ICU) for continued management and close observation. The patient was admitted to the hospital and ultimately diagnosed with acute lymphoblastic leukemia based on cytology and positive Philadelphia chromosome.
Six days into his admission, the patient suffered a large, right-sided spontaneous intraparenchymal hemorrhage in the setting of severe thrombocytopenia; platelets were 7,000 µL He required emergent craniotomy and intubation. His course was further complicated by aspiration pneumonia. Bronchioalveolar lavage and computed tomography of the chest led to concern for cavitary aspergilloma, requiring intravenous antifungals, broad-spectrum antibiotics, and pressor support for septic shock. The patient ultimately required tracheostomy, percutaneous gastric tube placement, and inferior vena cava filter as he was bedridden and unable to take anticoagulants. After his prolonged ICU course, and prior to his discharge, he was started on methotrexate, nilotinib, and ponatinib.
Per family, the patient followed up with oncology after discharge and failed chemotherapy at an outside hospital. We were unable to obtain records of these failed treatments. The patient continued to have seizures after his discharge and was put on daily levetiracetam and lacosamide. He was unable to work in construction and became dependent on family for activities of daily living. He ultimately died two years after his diagnosis was made. He is survived by his parents who took the time to speak with us in detail about their son after he had died.
Acute lymphoblastic leukemia typically has a bimodal distribution, presenting in children < 6 years of age and adults > 60.3 The incidence of ALL in the United States is estimated
What do we already know about this clinical entity? Acute lymphocytic leukemia (ALL) is a cancer of lymphoid cells, most commonly affecting children < 6 and adults > 60, with the most common complaint being fatigue.
What makes this presentation of disease reportable? A 34-year-old male, status post multiple visits to the emergency department with the complaint of bilateral knee pain without fatigue, fever, bleeding or bruising, was diagnosed with ALL.
What is the major learning point?
Cognitive biases may contribute to delayed diagnosis when otherwise healthy “bounce-back” patients return to the emergency department with the same complaint.
How might this improve emergency medicine practice?
This case highlights the need to remain vigilant for cognitive biases and to generate a broad differential for all patients.
at 1.6 per 100,000 population, with > 1,400 deaths annually.3 The disease presentation is more devastating in adults.3 It involves the abnormal proliferation and differentiation of lymphoid cells, and clinical manifestations typically presents when these cells accumulate in the bone marrow, peripheral blood, and extramedullary sites.3
The presentation of ALL is often non-specific and includes classic “B symptoms”—fever, weight loss, and night sweats—along with symptoms of easy bleeding, bruising, lymphadenopathy, joint pain, and bone pain.3 Although our patient did not have classic B symptoms he did experience progressively worsening joint pain. Diagnosis is established by the presence of ≥ 20% lymphoblasts in the bone marrow or peripheral blood. Further testing typically includes bone marrow biopsy, cytology, and genetic testing.3 Coagulation studies, renal function studies, and electrolytes should concurrently be investigated as the patient should be evaluated for tumor lysis syndrome, as was seen in our patient, which is an oncologic emergency associated with underlying malignancies with a high degree of mortality. It should be treated immediately.4
A Case Report of Acute Lymphoblastic Leukemia Presenting as Bilateral Knee Pain in a Healthy Runner Rosselli et al
It should be noted that although joint aspiration was not done on our patient, it could be considered in cases of leukemia-associated joint pain to assess for leukemic arthritis.5 In our patient’s prior ED visit, he presented with mild effusions seen on knee radiographs. Joint aspiration could have been done, as immunohistology studies of synovial fluid may aid in diagnosis as would synovial biopsy.5 Prior cases of leukemic arthritis have shown blast cells within synovial fluid.5,6 Leukemic arthritis is a wellrecognized complication in children but may also be the presenting symptom in adults with a frequency of 14% and 4%, respectively; it is more commonly associated with larger joints such as knees and shoulders.5,7
It is also important to consider cognitive biases associated with delayed diagnosis in the otherwise healthy “bounceback” patient. Such biases include anchoring bias, halo effect, perception bias, and premature closure (Table).8 Anchoring bias is particularly important to consider in the ED. It has been shown that emergency physicians at all levels of training are susceptible to anchoring bias.8,9 Studies cite increased pressure to see patients rapidly, triage note documentation, and the high-pressure nature of the job as factors that may influence emergency physician decision-making.8-10 Zandbergen et al showed that anchoring bias in resident physicians may be limited by increased time spent deliberating a differential diagnosis. Our case exemplifies how cognitive bias may impact patient care and the discriminating features of ALL (joint pain in an otherwise healthy young male).
This atypical case presentation highlights the importance of having a high degree of suspicion for a more severe disease process in the bounce-back patient. The outcome of this case highlights the importance of doing so even in a seemingly benign complaint of bilateral knee pain in an otherwise healthy runner. It also underscores how delayed diagnosis may lead to poor prognosis and tragic outcomes, especially when we fail to consider cognitive biases in the medical decisionmaking process.
Consent of patient’s family has been obtained and filed for the publication of this case report.
Address for Correspondence: Michael Rosselli, MD, Mount Sinai Medical Center, Department of Emergency Medicine, 4300 Alton Road, Miami Beach, FL 33140. Email: mrosselli1430@gmail.com.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Rosselli et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
Table. Types of cognitive bias, Cognitive Bias Definition and example
Anchoring bias Too much weight is given to the early and most prominent feature of a patient’s history, physical, or test results. This causes diagnostic momentum in favor of a certain diagnosis. For example, a triage note states that a patient with heart failure has shortness of breath with leg swelling. The emergency practitioner fails to consider evaluating the patient for pulmonary embolism in history-taking and instead continues to do a work-up for heart failure.
Premature closure The clinician stops at a plausible diagnosis despite an incomplete work-up. An example would be a clinician who discharges a patient home with the diagnosis of a urinary tract infection based on the urinalysis alone, without considering acute appendicitis in that patient with right lower abdominal pain with urinary symptoms.
Halo effect Tendency to let the first impression of a person influence one’s thoughts about the individual overall. Example: A clinician in the office downplays a patient’s abdominal pain, stating “this patient tends to overreact with everything.”
Perception bias Tendency to stereotype and assume beliefs about a group of people and imposing those beliefs on an individual who belongs to that group. Example: A physician states, “Soccer players tend to be a histrionic group,” when discussing a patient.
Ascertainment bias In clinical practice, this refers to a clinician’s tendency to base their work-up on prior clinical experiences. Example: A chronic undomiciled patient with multiple presentations of alcohol use disorder arrives to the emergency department unconscious, and the clinician assumes it is acute alcohol intoxication due to prior experience and performs a limited neurologic exam only to find that the patient ultimately had a massive intracranial hemorrhage.
Rosselli et al A Case Report of Acute Lymphoblastic Leukemia Presenting as Bilateral Knee Pain in a Healthy Runner
1. Redaelli A, Laskin BL, Stephens JM, et al. A systematic literature review of the clinical and epidemiological burden of acute lymphoblastic leukaemia (ALL). Eur J Cancer Care (Engl). 2005;14(1):53-62.
2. Sakata H, Nakao A, Matsuda K, et al. Acute leukemia presenting as bone pain with normal white blood cell count. Acute Med Surg 2014;1(4):249.
3. Terwilliger T and Abdul-Hay M. Acute lymphoblastic leukemia: a comprehensive review and 2017 update. Blood Cancer J. 2017;7(6):e577.
4. Ñamendys-Silva SA, Arredondo-Armenta JM, Plata-Menchaca EP, et al. Tumor lysis syndrome in the emergency department: challenges and solutions. Open Access Emerg Med. 2015;20;7:39-44.
5. Usalan C, Ozarslan E, Zengin N, et al. Acute lymphoblastic leukaemia presenting with arthritis in an adult patient. Postgrad Med J. 1999;75(885):425-427.
6. Aviña-Zubieta JA, Galindo-Rodriguez G, Lavalle C. Rheumatic
manifestations of hematologic disorders. Curr Opin Rheumatol. 1998;10(1):86-90.
7. Spilberg I, Meyer GJ. The arthritis of leukemia. Arthritis Rheum 1972;15(6):630-635.
8. Joseph MM, Mahajan P, Snow SK, et al; Committee on Pediatric Emergency Medicine, American College of Emergency Physicians Pediatric Emergency Medicine Committee, and Emergency Nurses Association Pediatric Committee. Optimizing pediatric patient safety in the emergency care setting. Pediatrics. 2022 1;150(5):e2022059674.
9. Mamede S, Zandbergen A, de Carvalho-Filho MA, et al. Role of knowledge and reasoning processes as predictors of resident physicians’ susceptibility to anchoring bias in diagnostic reasoning: a randomized controlled experiment. BMJ Qual Saf. 2024 16;33(9):563-572.
10. Ly DP, Shekelle PG, Song Z. Evidence for anchoring bias during physician decision-making. JAMA Intern Med. 2023;183(8):818-823.
Sabrina Lee, DO
Richard D. Shin MD, CHSE
Kallie Combs, DO
Section Editor: Melanie Heniff, MD, JD
Submission history: Submitted March 20, 2025; Revision received July 2, 2025; Accepted August 27, 2025
Electronically published December 7, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.47015
Introduction: Acute hypercapnic respiratory failure secondary to foreign body aspiration is a rare but severe complication seen in pediatric patients. Foreign body aspiration is one of the leading causes of death in children and requires prompt intervention and stabilization when definitive bronchoscopy is not readily available.
Case Report: We describe the case of a 16-month-old male who developed acute hypercapnic respiratory failure following the aspiration of a foreign body. On presentation to the emergency department, the child was in respiratory distress, appeared cyanotic, and had severely impaired oxygenation, all indicating respiratory failure. Initial management involved stabilization, advanced airway management, and ventilatory adjustments. Efficient communication with multiple specialists coordinated the appropriate transfer of the patient to a tertiary pediatric facility for bronchoscopy and ultimate successful foreign body removal without complications.
Conclusion: The report highlights challenges in the management of pediatric foreign body aspiration leading to severe hypercapnia, the importance of interdisciplinary coordination, and the management techniques used to stabilize the patient for safe transfer to a tertiary care center. [Clin Pract Cases Emerg Med. 2025;10(1):50-54.]
Keywords: foreign body aspiration; acute hypercapnic respiratory failure; pediatric resuscitation; case report.
Foreign body aspiration is a common cause of respiratory compromise in children and one of the leading causes of accidental deaths among children with morbidity ranging from 10% to 20% worldwide.1 In the United States, foreign body aspiration is the leading cause of accidental deaths in children under six years of age.1 The main symptoms of foreign body aspiration include acute onset of severe coughing, wheezing, dyspnea, or stridor. Often, as the foreign body ceases movement in the respiratory tract, these acute symptoms may subside or disappear, allowing this acute episode to bypass the adults’ attention.2 Additionally, the absence of these symptoms do not rule out foreign body aspiration when presented with the appropriate clinical presentation. A history of acute “choking” is
a critical indicator of respiratory distress and, thus, there should be high suspicion for foreign body aspiration.
Chest radiograph is often the main initial tool for airway foreign body assessment. Common findings include atelectasis, air trapping, pneumothorax, and emphysema. However, one retrospective study found that 32.2% of cases were associated with normal chest radiographs.3 Similarly, most foreign bodies that children aspirate on, such as toys, food, and balloons, are radiolucent on chest radiographs.4 The gold standard for detection and treatment is rigid bronchoscopy. With high suspicion of foreign body aspiration, bronchoscopy should be used despite the absence of symptoms, physical exam findings, or radiographic findings.
Acute hypercapnic respiratory failure is a rare but
potentially life-threatening complication of foreign body aspiration in the pediatric population. When a patient aspirates on a foreign body, there is varying obstruction to the upper and lower airways, impeding adequate ventilation and oxygenation. Poor ventilation leads to the inability to appropriately eliminate carbon dioxide (CO2) from the body, contributing to elevated levels of CO2 in the blood. Infants and younger children are more likely to experience acute respiratory failure due to a multitude of factors, including decreased respiratory drive in the early days of life; increased airway resistance secondary to reduced nostril and airway diameter; greater collapsibility of respiratory musculature; and lower threshold for respiratory fatigue.5 While the typical management involves bronchoscopy for foreign body removal, severe respiratory compromise leading to acute hypercapnic respiratory failure calls for immediate critical intervention.
The literature is limited regarding foreign body aspiration complicated by hypercapnic respiratory failure among pediatric patients. This case illustrates a unique presentation and management of foreign body aspiration in a boy who presented with acute hypercapnic respiratory failure, which required rapid diagnosis, early intervention, and appropriate transfer for specialized care. The patient was successfully treated with removal of the foreign body and had an uncomplicated recovery.
A 16-month-old male with no significant medical history presented to the emergency department (ED) following a witnessed accidental aspiration of a soft toy in the shape of a banana. According to his mother, the child turned purple and exhibited signs of choking shortly after the incident. The parents were unable to dislodge the object with back slaps and brought him to the ED within 10 minutes of the incident. On initial assessment, the child was cyanotic with severely compromised oxygen levels, showing a peripheral oxygen saturation (SpO2) of 68% on room air, as well as tachypnea with a respiratory rate elevated to 60 breaths per minute. The child appeared limp and obtunded, although he did have spontaneous eye-opening. Physical examination revealed facial petechiae, which is a clinical sign of significant respiratory effort, and diminished breath sounds on the right side.
The patient was quickly placed on a non-rebreather mask with a flow rate of 15 liters per minute, leading to an immediate improvement in his oxygen saturation to 100%. A chest radiograph was performed, which confirmed the presence of the foreign body lodged in the carina (Image). Initial arterial blood gas showed acidosis secondary to hypercapnia with a pH of 7.07 and partial pressure of arterial CO2 (PaCO2) of 72 millimeters of mercury (mm Hg) (Table 1). Due to his severe respiratory distress and hypoxia, the child required urgent intubation. Rapid sequence intubation with intravenous midazolam and rocuronium, using video laryngoscopy and a 3.5-mm cuffed endotracheal tube, was
CPC-EM Capsule
What do we already know about this clinical entity?
Respiratory failure secondary to foreign body aspiration in children is a life-threatening emergency, which requires prompt recognition and intervention.
What makes this presentation of disease reportable?
This case highlights the challenges in managing a difficult pediatric airway, involving patient stabilization, multi-disciplinary consultations, and transfer to a tertiary facility.
What is the major learning point?
Early diagnosis and airway management in pediatric foreign body aspiration is essential to ensure timely bronchoscopy and prevent lifethreatening outcomes.
How might this improve emergency medicine practice?
We review the methods to navigate hypercapnia in pediatric respiratory failure that can improve survival and reduce morbidity.
Acute Hypercapnic Respiratory Failure from Foreign Body Aspiration in a 16-Month-Old: A Case Report
Table 1. Initial arterial blood gas of a 16-month male upon arrival to the emergency department.
CO2, Total Arterial
O2 Saturation (Arterial, Calc)
CO2, carbon dioxide; HCO3, bicarbonate; O2, oxygen; pCO2, partial pressure of carbon dioxide; pO2, partial pressure of oxygen; mm Hg, millimeters of mercury; mmol/L, millimole per liter; mmol/dL, millimole per deciliter; WB, whole blood; W/B (A), whole blood arterial.
successful. During intubation, the foreign body was not visualized as it was near the carina, and moderate airway edema was observed, underscoring the difficulty of maintaining adequate ventilation.
Ventilatory management began with efforts to improve the patient’s oxygenation and reduce his hypercapnia. Initial ventilation attempts revealed hypoxia with SpO2 of about 80% and critically elevated end-tidal CO₂ (ETCO₂) levels remaining above 100 mm Hg (reference range: 35-45 mm Hg). Repeat arterial blood gas showed worsening of acidosis with a pH of 6.89 and severe hypercapnia of PaCO2 above 150 mm Hg (35-50 mm Hg) (Table 2). On reassessment, the patient had diffuse bilateral wheezing, and he was given IV dexamethasone and nebulized albuterol. To address breath stacking, which further compromised his oxygenation, the patient was disconnected from the ventilator periodically, and
Test Name Patient Value Reference Range
pH (Arterial) 6.89 7.32-7.45
pCO2 (Arterial) >150 35-50 mm Hg
pO2 (Arterial) 571 60-90 mm Hg
HCO3 (Arterial) cnc 17.0-23.0 mmol/L
CO2, Total Arterial cnc 17-24 mmol/L
Base Excess (Arterial) cnc -6.00-2.00 mmol/L
O2 Saturation (Arterial, Calc) cnc 95-98%
Glucose WB 312 50-80 mg/dL
Lactate W/B (A) < 0.30 mmol/L
CO2, carbon dioxide; cnc, could not calculate; HCO3, bicarbonate; mm Hg, millimeters of mercury; O2, oxygen; pCO2, partial pressure of carbon dioxide; pO2, partial pressure of oxygen; mm Hg, millimeters of mercury; mmol/L, millimole per liter; mmol/dL, millimole per deciliter; WB, whole blood; W/B (A), whole blood arterial.
manual sustained pressure was applied to the sternum. This manual decompression of the chest aided in temporary improvement in oxygenation.
When reconnected to the ventilator, volume control setting was employed at 4-6 milliliters per kilogram (mL/kg) tidal volumes, and pressure limits were set to manage airway pressures; however, peak pressure remained elevated to 55 cm of water pressure (cm H2O) (reference range: 30-35 cm H2O). Given the concern for risk of barotrauma, ventilator settings were then changed to pressure control with a maximum pressure of 40 cm H2O. Even on these settings, the ventilator returned only 5-10 mL of tidal volume, which was significantly suboptimal in a 10.3 kg child. However, this setting showed appropriate rise of SpO2 to about 90% and lowering of ETCO₂ to about 40 mm Hg, indicating improved oxygenation and ventilation; therefore, the patient was ultimately monitored on pressure control settings.
Due to ongoing agitation and poor ventilator synchrony, we administered continuous sedation with IV midazolam, pain control with fentanyl infusion, and paralysis with vecuronium infusion to maintain ventilator control. The patient’s need for rigid bronchoscopy necessitated consultation with pediatric otolaryngologist for foreign body removal. However, given the lack of pediatric bronchoscopy tools in-house, a transfer to a tertiary pediatric facility was organized. Additional planning included obtaining a pediatric chest tube kit from the neonatal intensive care unit, as the risk of pneumothorax was high due to elevated airway pressures. Following transfer to a specialized pediatric center, the patient underwent a successful rigid bronchoscopy, with removal of the foreign body from his airway. He was extubated post-procedure and continued to improve, ultimately being discharged with no further complications.
With foreign body aspiration being an unfortunate,
Lee et al. Acute Hypercapnic Respiratory Failure from Foreign Body Aspiration in a 16-Month-Old: A Case Report
common occurrence within the pediatric population, this case highlights the multiple steps involved in managing the clinical complexities of acute hypercapnic respiratory failure in pediatric foreign body aspiration. Firstly, early recognition is critical to prevent the progression to respiratory failure and allow for earlier targeted management. Foreign body aspiration is a preventable cause of death in children < 3 years of age, an age group that is prone to unwitnessed aspiration due to mouthing behaviors.6,7 Foreign bodies typically lodge in the bronchi and lower airway, although laryngotracheal obstructions can mimic upper airway conditions such as croup and asthma.8,9
In a retrospective study, Molla et al recommended maintaining a high index of suspicion for foreign body aspiration in cases of bronchopulmonary infections with an atypical course.6 Our patient had a witnessed aspiration, but in cases of unwitnessed events or sudden respiratory symptoms, clinicians should strongly consider foreign body aspiration.2,3 Sandhofer et al described a child treated for acute bronchitis with worsening symptoms and was found to have a concomitant peanut aspiration.10 Pradhan et al also identified a toy in the bronchus of a child evaluated for pnuemonia.11 These cases emphasize the importance of thorough historytaking in suspected aspiration cases.
Our patient had severe respiratory distress with hypoxia, prompting intubation with video laryngoscopy. Once on mechanical ventilation, standard ventilator settings resulted in high pressures and suboptimal oxygenation; the patient was then disconnected from the ventilator, and manual chest decompressions offered temporary relief. A similar technique has been used in children with severe asthma and bronchiolitis in the pediatric intensive care unit with significant air trapping. Intermittent manual external chest compression led to gradual but effective improvement in the children’s partial pressure of CO2, preventing intubation and allowing for ultimate discharge from the unit.12 With external manual decompression, optimal sedation was crucial for ventilator synchrony and oxygenation. Through these maneuvers, the patient was sufficiently stabilized for immediate transfer pending definitive treatment.
The child’s ability to tolerate extreme hypercapnia demonstrates that pediatric patients may withstand elevated CO₂ levels if oxygenation and circulation are adequately maintained. Following successful removal of the foreign body, the patient was extubated, improved clinically the next morning, and was ultimately discharged two days later without complications. Similar outcomes were seen in a case involving massive grain aspiration with severe hypercapnia (PaCO2 of 501 mm Hg), where preserved cardiovascular function allowed for the child’s full recovery.13 Likewise, Mazzeo et al reported a boy with sustained hypercapnia with a PaCO2 of 293 mm Hg for 14 hours during a near-fatal asthma attack, who remained stable secondary to adequate perfusion and oxygenation.14 These cases highlight the resilience of pediatric patients with extensive hypercapnia when oxygenation,
perfusion, and cardiovascular function are preserved. Finally, the need for coordinated, interdisciplinary care was underscored in this case, as successful management required the involvement of pediatric otolaryngology, intensive care, and ED teams. Timely communication between consultants and readiness for potential complications was critical in stabilizing the patient for transfer. The case further illustrates the importance of anticipating potential complications, such as pneumothorax, and preparing proactively to ensure patient safety during transport. With these modalities, our patient was promptly transferred to the tertiary pediatric facility and underwent rigid bronchoscopy with successful removal of the foreign body within two hours of the initial event. This case emphasizes the importance of both prompt intervention and recognition of foreign body aspiration in toddlers, particularly when severe respiratory distress and hypercapnia are present. Through ventilatory support adjustments, consultant coordination, and anticipation of potential complications, the patient was successfully stabilized for treatment and recovery.
In managing pediatric foreign body aspiration leading to severe respiratory distress, several key learning points emerged. First, addressing breath-stacking by disconnecting the patient from the ventilator and manually decompressing the chest can improve ventilation temporarily. Second, in addition to adjusting ventilator control settings, full paralysis can help achieve effective ventilator control in cases with severe respiratory distress. Third, in cases requiring advanced airway procedures, ensuring all necessary consultants are prepared with equipment for safe transfer is essential. Finally, clear communication and collaboration across disciplines can expedite care and improve patient outcomes in critical pediatric emergencies.
Patient consent has been obtained and filed for the publication of this case report.
Address for Correspondence: Sabrina Lee, DO, New YorkPresbyterian Queens, Department of Emergency Medicine, 56-45 Main St, Flushing, NY 11355. Email Address: sabrina.lee210@ gmail.com.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Lee et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
Acute Hypercapnic Respiratory Failure from Foreign Body Aspiration in a 16-Month-Old: A Case Report
1. Nasir ZM and Subha ST. A five-year review on pediatric foreign body aspiration. Int Arch Otorhinolaryngol. 2021;25(2):e193-e199.
2. Markov S, Markova P, Karavelikova I, et al. Foreign bodies in lower airway in children: brief review and clinical experience. Children (Basel). 2025;12(1):67.
3. Mallick M. Tracheobronchial foreign body aspiration in children: a continuing diagnostic challenge. African J Paed Surg. 2014;11(3):225.
4. Cramer N, Jabbour N, Tavarez MM, et al. Foreign body aspiration. [Updated 2023 July 31]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. Available at: https://www.ncbi.nlm. nih.gov/books/NBK531480/. Accessed January 21, 2025.
5. Panetti B, Bucci I, Di Ludovico A, et al. Acute respiratory failure in children: a clinical update on diagnosis. Children (Basel) 2024;11(10):1232.
6. Molla YD, Mekonnen DC, Beza AD, et al. Foreign body aspiration in children at University of Gondar Comprehensive Specialized Hospital, a two year retrospective study. Heliyon. 2023;9(10):e21128.
7. Karišik M. Foreign body aspiration and ingestion in children. Acta Clin Croat. 2023;62(Suppl1):105-112.
8. Punnoose SE, Victor J, Hazarika P, Ss M. C-MAC® video-
laryngoscope assisted removal of pediatric upper airway foreign body via apneic technique: two case reports. SAGE Open Med Case Rep 2019;7:2050313X18823088.
9. Kumar S, Al-Abri R, Sharma A, et al. Management of pediatric tracheo bronchial foreign body aspiration. Oman Med J 2010;25(4):e019.
10. Sandhofer MJ, Salzer H, Kulnig J. Foreign body aspiration – sometimes a tough nut to crack. Respir Med Case Rep. 2015;15:18-19.
11. Pradhan M, Gohil KM, Shinde SG, et al. Navigating challenges: diagnosis and management of foreign body aspiration in a child: a case report. J Family Med Prim Care. 2025;14(2):800-803.
12. Brooks R, Cohen-Cymberknoh M, Glicksman C, et al. Manual external chest compression reverses respiratory failure in children with severe air trapping. Pediatr Pulmonol. 2021;56(12):3887-3890.
13. Slinger P, Blundell PE, Metcalf IR. Management of massive grain aspiration. Anesthesiology. 1997;87(4):993-995.
14. Mazzeo AT, Spada A, Praticò C, et al. Hypercapnia: what is the limit in paediatric patients? A case of near‐fatal asthma successfully treated by multipharmacological approach. Pediatric Anesthesia 2004;14(7):596-603.
Bruce M. Lo, MD, MBA*
Megyn
K. Christensen, DO†
Coral E. Byrns, DO†
Benjamin Chidester, MD*
Editor: Rick Alan McPheeters, DO
Sentara Norfolk General Hospital/Eastern Virginia Medical School at Old Dominion University, Department of Emergency Medicine, Norfolk, Virginia Eastern Virginia Medical School at Old Dominion University, Department of Emergency Medicine, Norfolk, Virginia
Submission history: Submitted July 22, 2025; Revision received September 12, 2025; Accepted September 15, 2025
Electronically published January 5, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem
DOI: 10.5811/cpcem.49096
Introduction: Aortic dissection is a rare but life-threatening condition with a high mortality rate if diagnosis is delayed. Aortic dissection classically presents with sudden-onset, sharp pain in the chest or back. However, atypical presentations can also occur, which could lead to a delay in diagnosis.
Case Report: A patient initially presented to the emergency department (ED) with left testicular pain ongoing for several hours. On examination, he had tenderness in the left lower quadrant abdomen and left testicle. A testicular ultrasound revealed decreased blood flow to the left testicle, raising concern for testicular torsion. The patient was taken to the operating room, where no torsion was found, and he was subsequently discharged home. Several days later, the patient returned to the ED with worsening pain radiating to the back. A computed tomography revealed an acute type A aortic dissection extending to the iliac arteries. He was transferred for surgical repair and discharged 12 days later.
Conclusion: While acute aortic dissection (AAD) typically presents with chest or back pain, atypical presentations can occur. When initial findings do not fully explain a patient’s symptoms, AAD should remain on the differential. This case highlights an uncommon presentation of AAD initially mimicking a testicular torsion. [Clin Pract Cases Emerg Med. 2025;10(1):55-58.]
Keywords: aortic dissection; testicular torsion; case report.
Acute aortic dissection (AAD) is a rare but potentially life-threatening emergency. Timely diagnosis is critical, as delays are associated with significantly increased morbidity and mortality. Although AAD typically presents with suddenonset chest or back pain, it can manifest with atypical symptoms that contribute to frequent misdiagnosis. While atypical presentations such as acute stroke or ST-segment elevation myocardial infarction have been documented, it is rare for a type A AAD to present initially as testicular pain. We present a case of a patient whose type A AAD was initially misdiagnosed as testicular torsion, resulting in a delay in diagnosis and definitive treatment.
A 40-year-old male presented to the emergency department (ED) with sudden-onset left testicular and lower abdominal pain that began two hours prior to arrival. He denied any trauma or urinary symptoms, including hematuria or dysuria but reported pain and perceived swelling of the left testicle. On examination, the patient appeared uncomfortable, with tenderness in the left lower quadrant and left testicle, although no visible swelling was noted. He had no past medical history, nor did he have a family history of nephrolithiasis.
Initial vital signs were as follows: blood pressure, 101/58 millimeters of mercury (mm Hg); heart rate, 84 beats per
minute (bpm); and temperature, 98.7 °F. Morphine was administered for pain control. Laboratory testing included a white blood cell (WBC) count of 13.2 kilograms per microliter (K/μL) (reference range 4.0-11.0 K/μl); hemoglobin (Hg), 12.8 grams per deciliter (g/dL) (11.7-16.1 g/dL); blood glucose, 114 milligrams (mg)/dL) (70-90 mg/dL); and creatinine, 1.3 mg/dL (0.8-1.4 mg/dL). A testicular ultrasound with Doppler demonstrated decreased blood flow to the left testicle, raising concern for testicular torsion (Image 1). A concurrent computed tomography (CT) of the abdomen and pelvis without contrast showed no renal stones but a calcification within the aorta. The patient was taken to the operating room for surgical exploration, but no torsion was found. He was subsequently discharged home following the procedure.
Two days later, the patient returned with worsening left lower abdominal pain radiating to his back. He described the pain as sharp and tearing in nature. The patient denied any
CPC-EM Capsule
What do we already know about this clinical entity?
Acute aortic dissection is rare, life-threatening, and often misdiagnosed due to highly variable and sometimes atypical presentations.
What makes this presentation of disease reportable?
This case presented with testicular pain with abnormal ultrasound findings suggestive of a testicular torsion, a rare presentation of type A aortic dissection.
What is the major learning point?
Unusual pain patterns or unexplained imaging should prompt consideration of aortic dissection in the differential, even in younger patients without risk factors.
How might this improve emergency medicine practice?
Maintaining suspicion for aortic dissection in atypical presentations can reduce misdiagnosis and improve patient outcomes.
urinary complaints or drug use. Initial vital signs were as follows: blood pressure, 243/143 mm Hg; heart rate, 103 bpm; and temperature, 98.2 °F. Laboratory testing included a WBC count of 14.0 K/μL; Hg, 13.4 g/dL; blood glucose, 115 mg/dL, and creatinine, 1.1 mg/dL. A CT angiogram revealed a type A aortic dissection (AAD) extending from the distal ascending aorta to the iliac arteries (Image 2). The patient was then transferred for emergent surgical repair and was discharged 12 days later without complications.
Acute aortic dissection is a rare but life-threatening diagnosis that demands timely recognition and intervention. It is diagnosed in approximately one of every 12,000 ED visits and in one of every 1,000 patients who present with atraumatic chest pain.1 Approximately 67% of all dissections are classified as type A, involving the ascending aorta, while 33% are type B, involving a tear in the descending aorta.2 Data from the International Registry of Acute Aortic Dissection demonstrated that mortality for untreated type A dissections increases by 0.5% per hour, reaching 24% within 48 hours.3 The 30-day mortality rate for type A AAD is approximately 57%, whereas for
Image 2. Computed tomography angiography of chest, abdomen, and pelvis: A) dissection flap down to iliacs (red arrows); B) dissection flap starting at the ascending aorta (red arrow).
through the left testicular artery as it branches off the abdominal aorta, resulting in decreased perfusion on Doppler imaging and contributing to the initial misdiagnosis.
Computed tomography with intravenous contrast in arterial phase (angiography) remains the preferred imaging modality for diagnosis in the ED due to its rapid availability and high sensitivity. Other imaging modalities include pointof-care ultrasound, which can detect a dissection flap within an aorta but cannot rule out AAD. Additionally, clinical decision rules such as the Aortic Dissection Detection Risk Score along with D-dimer testing can assist in ruling out AAD in appropriately selected low-risk patients.10
Acute aortic dissection is a rare but life-threatening diagnosis. Although patients typically present with chest pain or back pain, atypical presentations can also occur with AAD and should be considered, especially if the initial diagnostic workup is not negative.
uncomplicated type B dissections it is around 10%. However, the mortality for type B AAD increases to 25% when complications such as malperfusion or rupture are present.2,4
Chest pain is the most common presenting symptom, reported in 79% of patients with type A dissections.2 Although hypertension is a known risk factor for AAD, only 36% of patients with AAD present with elevated blood pressure at the time of diagnosis.2 The majority of patients are older, with an average age of 61 years.2 Diagnosis can be particularly challenging due to the wide variability in presentation. A systematic review analyzing 12 studies found that approximately 34% of patients with AAD were initially misdiagnosed.5 Common misdiagnoses included myocardial infarction, cerebrovascular accident, pulmonary embolism, musculoskeletal pain, ureterolithiasis, and psychological disorders.
The diagnosis in this case was particularly difficult due to several atypical features. The patient was significantly younger than the average AAD patient and had no known risk factors such as hypertension, illicit drug use, or signs of connective tissue disease such as Marfan syndrome. His presenting symptoms of left lower abdominal pain and testicular pain were also highly atypical for type A AAD, particularly in the absence of chest or upper back pain.
Only a few cases of aortic dissection presenting as testicular pain have been documented in the literature,6-9 and only one of these involved a type A dissection.7 In some of these cases, the theory as to why patients presented with testicular pain was secondary to expansion of the aorta leading to compression of the ilioinguinal or genitofemoral nerves.7, 9 To our knowledge, this is the first reported case in which a patient with type A AAD had an abnormal testicular ultrasound suggestive of testicular torsion suggesting a vascular involvement causing the initial pain. It is plausible that the dissection transiently compromised blood flow
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Address for Correspondence: Bruce M. Lo, MD, MBA, Sentara Norfolk General Hospital/Eastern Virginia Medical School at Old Dominion University, Department of Emergency Medicine, 600 Gresham Drive, Room 304, Norfolk, VA, 23507. Email: brucelo1@ yahoo.com.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Lo et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Alter SM, Eskin B, Allegra JR. Diagnosis of aortic dissection in emergency department patients is rare. West J Emerg Med 2015;16(5):629-31.
2. Evangelista A, Isselbacher EM, Bossone E, et al. IRAD investigators. Insights from the International Registry of Acute Aortic Dissection: a 20-year experience of collaborative clinical research. Circulation 2018;137(17):1846-60.
3. Harris KM, Nienaber CA, Peterson MD, et al. Early mortality in type A acute aortic dissection: insights from the International Registry of
Acute Aortic Dissection. JAMA Cardiol. 2022;7(10):1009-15.
4. Isselbacher EM, Preventza O, Hamilton Black J 3rd, et al. 2022 ACC/ AHA Guideline for the Diagnosis and Management of Aortic Disease: a report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. Circulation. 2022;146(24):e334-e482.
5. Lovatt S, Wong CW, Schwarz K, et al. Misdiagnosis of aortic dissection: a systematic review of the literature. Am J Emerg Med 2022;53:16-22.
6. Jarvis S, Donohoe P, Huang D, et al. Unusual presentation of aortic dissection with bilateral testicular pain and rapidly deteriorating renal function. Urology. 2014;83(5):989-91.
7. Chan-Tack KM. Aortic dissection presenting as bilateral testicular pain. N Engl J Med. 2000;343(16):1199.
8. Siddiqui, M, Haider, I. Testicular pain, trouble voiding and hypertension: “dissecting the possibilities.” J Am Coll Cardiol. 2014, 63 (12_Supplement) A700.
9. Guarín-Loaiza GM, Nocua-Baez LC, Alfonso-Hernández G. Bilateral testicular pain as an acute aortic dissection symptom. Rev. Fac. Med 2016;64(3):571-4.
10. Promes SB, Westafer L, Byyny R, et al. Critical issues in the evaluation and management of adult patients with suspected acute nontraumatic thoracic aortic dissection. Ann Emerg Med. 2025; 86(1): e12-e26.
Samir Beso, MD*
Layla Abubshait, MD*†
Section Editor: Steven Walsh, MD
Jefferson Einstein Montgomery Hospital, Department of Emergency Medicine, East Norriton, Pennsylvania
†
Sidney Kimmel Medical College, Thomas Jefferson University, Philadelphia, Pennsylvania
Submission history: Submitted May 29, 2025; Revision received July 30, 2025; Accepted September 10. 2025
Electronically published January 1, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.48354
Introduction: Milk-alkali syndrome is characterized by the triad of hypercalcemia, metabolic alkalosis, and acute kidney injury resulting from excessive intake of calcium and absorbable alkali. Despite falling out of prominence with the advent of modern ulcer treatments, milk-alkali syndrome has experienced a resurgence with the widespread availability of over-the-counter calcium preparations, which now account for up to 10% of hypercalcemia cases.
Case Report: A 60-year-old man with multiple comorbidities presented to the emergency department with altered mental status after his scheduled kyphoplasty was canceled due to concerning neurological findings. Laboratory evaluation revealed severe hypercalcemia, marked metabolic alkalosis, and acute kidney injury. Further history revealed excessive antacid consumption for heartburn. The patient was diagnosed with milk-alkali syndrome, treated with intravenous fluids and calcitonin, and discharged home after 48 hours with complete resolution of signs and symptoms.
Conclusion: Milk-alkali syndrome represents an increasingly recognized cause of severe hypercalcemia in the emergency setting. This case demonstrates the importance of thorough medication history, early recognition of the classic triad, and prompt initiation of conservative management. With the growing use of calcium-based, over-the-counter preparations, emergency physicians must maintain vigilance for this potentially serious but readily treatable condition. [Clin Pract Cases Emerg Med. 2025;10(1):59-62.]
Keywords: milk-alkali syndrome; hypercalcemia; metabolic alkalosis; acute kidney injury; antacids; case report.
Milk-alkali syndrome is characterized by the triad of hypercalcemia, metabolic alkalosis, and acute kidney injury resulting from excessive intake of calcium and absorbable alkali. Although historically associated with milk and bicarbonate treatment for peptic ulcer disease, modern cases typically involve overconsumption of calcium-containing antacids or supplements.1 Despite falling out of prominence with the advent of modern ulcer treatments, milk-alkali syndrome has seen a resurgence with the widespread availability of over-the-counter (OTC) calcium preparations. It accounts for up to 10% of hypercalcemia cases and represents
the third most common cause of hospital-associated hypercalcemia after hyperparathyroidism and malignancy.2 The diagnosis can be easily missed, leading to unnecessary investigations and inappropriate treatment.
A 60-year-old man with type two diabetes on metformin, hyperlipidemia on rosuvastatin, hypertension on metoprolol, and prior myocardial infarction presented to the emergency department after his scheduled first lumbar vertebrae kyphoplasty was cancelled due to altered mental status noted by the interventional radiology team. The patient reported feeling
progressively disoriented over five days, accompanied by significant heartburn. Initially, he did not volunteer information about his antacid use. However, when the emergency physician specifically inquired about OTC medications and treatments for his heartburn symptoms, the patient revealed he had been self-medicating with calcium carbonate tablets, consuming more than 20 tablets daily during this period.
Vital signs revealed the following: heart rate, 110 beats per minute; blood pressure, 138/82 millimeters of mercury; temperature, 98.6 °F; respiratory rate, 18 breaths per minute; and oxygen saturation, 98% on room air. The patient appeared tired but remained alert and oriented. Physical examination was otherwise unremarkable with no focal neurological deficits. Computed tomography angiography of the head and neck was performed due to the acute onset of neurological symptoms and concern for cerebrovascular accident, which was negative for acute cerebrovascular pathology.
Laboratory studies and electrocardiogram were ordered, revealing multiple electrolyte abnormalities and prolonged corrected QT interval (Image) (Table).
The combination of severe hypercalcemia, metabolic alkalosis, and acute kidney injury suggested milk-alkali syndrome given the history of excessive antacid use, although thiazide diuretic use, primary hyperparathyroidism, and malignancy-associated hypercalcemia were also considered. Given the constellation of severe hypercalcemia, metabolic alkalosis, and the patient’s history of excessive antacid consumption, milk-alkali syndrome was strongly suspected. The emergency team initiated aggressive intravenous (IV) fluid resuscitation with 0.9% normal saline at 200 milliters per hour to promote calciuresis and correct volume depletion. To achieve more rapid calcium reduction, subcutaneous calcitonin 4 units/ kilogram was administered. While furosemide was considered
Image. Electrocardiogram showed sinus tachycardia at a rate of 110 beats per minute with QTc (corrected QT interval) prolongation (512 milliseconds). Notable findings include normal axis, normal QRS complex width, and no evidence of ischemic changes. The corrected QT interval prolongation is paradoxical given the severe hypercalcemia, which typically causes corrected QT interval shortening.
CPC-EM Capsule
What do we already know about this clinical entity?
Milk-alkali syndrome has resurged with widespread use of over-the-counter calcium preparations for heartburn.
What makes this presentation of disease reportable?
This case illustrates the challenge of diagnosis when patients don’t report antacid use, and the paradoxical electrocardiogram findings that can occur with concurrent metabolic derangements.
What is the major learning point?
Emergency physicians must specifically inquire about over-the-counter antacid consumption.
How might this improve emergency medicine practice?
Proactive medication reconciliation including specific questions about heartburn treatments can lead to faster recognition of milk-alkali syndrome.
for its calciuric effects, it was initially deferred due to concerns about further volume depletion in this already dehydrated patient. Given the severity of his hypercalcemia and altered mental status, the patient was admitted to the internal medicine service for continued monitoring and management.
The patient’s clinical response was reassuring and consistent with milk-alkali syndrome. Continued IV. hydration throughout hospital day (HD) 1 resulted in gradual improvement of his calcium levels to 12.1 milligrams per deciliter (mg/dL), However, recognizing that calcium remained significantly elevated despite aggressive hydration, the medical team administered furosemide 40 mg IV on HD 2 to enhance calcium excretion, with repeat calcium levels normalizing to 8.8 mg/dL. Concurrent with calcium normalization, the patient’s sodium levels improved to 134 milliequivalents/L as volume status was restored.
During his hospitalization, the patient experienced persistent cognitive signs and symptoms including speech slurring, which prompted concern for possible cerebrovascular pathology. Magnetic resonance imaging of the brain without contrast was obtained, revealing an incidental subacute infarct in the right frontal lobe. Neurology consultation was obtained, and the patient was prescribed apixaban 5 mg by mouth twice daily for secondary stroke prevention. Notably, his mental status improvements paralleled the correction of his
Table. Laboratory results during emergency department evaluation and hospitalization of patient with severe hypercalcemia. metabolic alkalosis, and acute kidney injury associated with excessive antacid consumption.
Test ED Results
Emergency Department Labs
Inpatient Labs
BUN, blood urea nitrogen; ED, emergency department; mEq/L, milliequivalents per liter; mg/dL, milligrams per deciliter; mcIU/mL, microinternational units per milliliter; ng/mL, nanograms per milliliter; pg/mL, picograms per milliliter; TSH, thyroid stimulating hormone; T4, thyroxine.
hypercalcemia, suggesting that the initial altered mental status was primarily attributable to the severe calcium elevation rather than the stroke itself.
Endocrinology consultation was also obtained to help guide further management and evaluate for underlying metabolic disorders. The endocrinology team confirmed that the patient’s normal-range parathyroid hormone levels were appropriate given the severe hypercalcemia, and they noted significant vitamin D deficiency, which likely contributed to the clinical picture. They recommended outpatient follow-up for thyroid ultrasound due to subclinical hypothyroidism, evidenced by low thyroid-stimulating hormone in the setting of normal triiodothyronine and thyroxine levels. The patient was discharged on HD 2 with instructions to avoid calciumcontaining antacids and follow up with his primary care physician and newly arranged endocrinology consultation.
This case exemplifies the classic presentation of milk-alkali syndrome in the modern era. The syndrome’s resurgence correlates with increased OTC calcium carbonate use, particularly among patients self-treating gastrointestinal (GI) symptoms.4 Recent literature continues to document cases of milk-alkali syndrome, emphasizing its ongoing clinical relevance despite advances in peptic ulcer disease management.5,6
The syndrome results from complex interactions between excessive calcium and alkali intake, leading to impaired renal excretion of both substances. Volume depletion, which can occur from various mechanisms including reduced oral intake
or GI losses, further exacerbates the condition by reducing glomerular filtration and enhancing calcium reabsorption. The pathophysiology involves a vicious cycle whereby hypercalcemia leads to nephrogenic diabetes insipidus and volume depletion, which in turn worsens calcium retention.
Clinical recognition relies on identifying key diagnostic features including a history of excessive calcium-containing antacid use, and the classic triad of hypercalcemia, metabolic alkalosis, and acute kidney injury, along with appropriately suppressed or normal parathyroid hormone levels. The rapid clinical improvement with conservative management serves as both a diagnostic and therapeutic confirmation. Physicians must maintain proactive medication reconciliation practices, as patients often do not spontaneously report OTC antacid use, viewing them as harmless supplements rather than medications with potential for toxicity.
Emergency management centers on immediate discontinuation of calcium-containing products as the most crucial intervention. Aggressive IV hydration remains the cornerstone of treatment, promoting calciuresis and correcting the volume depletion that perpetuates the syndrome. Calcitonin provides rapid but temporary calcium reduction, typically within four to six hours, making it useful for severe cases requiring immediate intervention. Loop diuretics should be used cautiously and only after adequate volume resuscitation to avoid worsening dehydration. Bisphosphonates are generally unnecessary given the rapid response to conservative measures, unlike other causes of severe hypercalcemia.
The paradoxical corrected QT interval prolongation observed
Too Much of a Good Thing: Milk-Alkali Syndrome from Excessive Antacid Use
in this case, typically shortened in hypercalcemia, likely resulted from concurrent metabolic derangements including severe hyperglycemia and ketosis. This finding illustrates the complex interplay of multiple metabolic abnormalities that can occur in severely ill patients. The incidental stroke finding, while unrelated to milk-alkali syndrome, emphasizes the importance of comprehensive evaluation in elderly patients with altered mental status, as multiple pathological processes may coexist.
Patient education regarding appropriate antacid use is crucial for preventing recurrence. Patients should be counseled on recommended dosing limits for OTC calcium preparations and advised to seek medical attention for persistent GI symptoms rather than escalating self-treatment. Physicians should consider concurrent medical conditions that may complicate the clinical presentation and require additional management, as demonstrated by this patient’s diabetic ketoacidosis and cerebrovascular disease.
Milk-alkali syndrome represents an increasingly recognized cause of severe hypercalcemia in the emergency setting. This case demonstrates the importance of thorough medication history, early recognition of the classic triad, and prompt initiation of conservative management. With the growing use of calcium-based over-the-counter preparations, emergency physicians must maintain vigilance for this potentially serious but readily treatable condition.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Address for Correspondence: Layla Abubshait, MD, Department of Emergency Medicine, Jefferson Einstein Montgomery Hospital, 559 W Germantown Pike, East Norriton, PA, 19403, USA. Email: Layla.abubshait@jefferson.edu
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Beso et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Rout P, Hashmi MF, Patel C. Milk-alkali syndrome. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024.
2. Medarov BI. Milk-alkali syndrome. Mayo Clin Proc. 2009;84(3):261267.
3. Beall DP, Henslee HB, Webb HR, et al. Milk-alkali syndrome: a historical reassessment of its current clinical utility. J Am Coll Cardiol 2006;47(10):2002-2008.
4. Patel AM and Goldfarb S. Got calcium? Welcome to the calciumalkali syndrome. J Am Soc Nephrol. 2010;21(9):1440-1443.
5. He J and Morton A. Milk-alkali syndrome. Aust J Gen Pract 2024;53(4):187-188.
6. Rizk MA, Abourizk N, Kovalenko I, et al. Milk-alkali syndrome: how electronic medical record open notes helped to rule out cancer. Am J Case Rep. 2022;23:e936969.
Mohammad Wasim Mohammad Zouhir Kanaa, MBBS
Shahd H I Abbastanira, MBBS
Esraa Mohamed Abdullah Elameen, MBBS
Sarah Emad Alsultan, MBBS
Section Editor: Christopher San Miguel, MD
Dubai Health, Department of Emergency Medicine, Dubai, United Arab Emirates
Submission history: Submitted July 26, 2025; Revision received October 6, 2025; Accepted October 21, 2025
Electronically published January 16, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem
DOI: 10.5811/cpcem.50489
Introduction: Cardiac arrest remains a major global cause of mortality, with both structural and non-structural cardiac abnormalities implicated. While ischemic heart disease is a common etiology, rare conditions such as papillary fibroelastoma can also result in life-threatening events through embolization or coronary obstruction. Timely recognition and advanced cardiac imaging, particularly transesophageal echocardiography, are essential in such atypical presentations.
Case Report: A 61-year-old female with a history of ischemic heart disease presented to the emergency department following an out-of-hospital cardiac arrest. Initial electrocardiogram (ECG) demonstrated anterior ST-elevation myocardial infarction, which resolved on repeat ECG, prompting reconsideration of the underlying cause. The patient achieved return of spontaneous circulation twice and eventually self-extubated. Further investigation, including cardiac imaging, revealed a mobile mass on the aortic valve intermittently obstructing the left main coronary artery. The mass was surgically resected and histologically confirmed as a papillary fibroelastoma. The patient recovered fully with no neurological or cardiac complications.
Conclusion: This case highlights the importance of maintaining a broad differential diagnosis in patients presenting with cardiac arrest, especially when ischemic changes are transient or unexplained. Although benign, papillary fibroelastoma can lead to sudden death due to embolization or coronary obstruction. Emergency physicians should be aware of such rare but treatable causes and consider early use of advanced cardiac imaging when standard presentations do not align with the clinical picture. [Clin Pract Cases Emerg Med. 2025;10(1):63-67.]
Keywords: papillary fibroelastoma; cardiac arrest; acute coronary syndrome mimic; transesophageal echocardiograph; case report.
Cardiac arrest remains a leading cause of mortality worldwide, with cardiac etiologies accounting for the majority of cases. While structural heart diseases such as cardiomyopathies, congenital anomalies, and heart failure are well-recognized contributors, non-structural conditions like Brugada syndrome and long QT syndrome also increase the risk of fatal arrhythmias.1 Rarely, external compression of coronary arteries by masses or malformations can lead to fatal
events, especially when classic risk factors are absent. Papillary fibroelastoma (PFE) is the most common primary tumor of cardiac valves, comprising approximately 75% of valvular tumors. Although histologically benign, its mobile and friable nature poses significant risk of embolization and dynamic obstruction, potentially resulting in stroke, myocardial infarction, or sudden cardiac death.2 Early detection is crucial, and transesophageal echocardiography remains the diagnostic modality of choice
When STEMI Isn’t STEMI: Cardiac Arrest from Aortic Valve Papillary Fibroelastoma – A Case Report Kanaa et
due to its superior sensitivity.3
We present the case of a 61-year-old woman with a history of ischemic heart disease and systemic lupus erythematosus who experienced out-of-hospital cardiac arrest. Her initial electrocardiogram (ECG) suggested anterior ST-elevation myocardial infarction (STEMI), which resolved on repeat testing. The patient achieved return of spontaneous circulation twice and ultimately self-extubated. Subsequent imaging identified a mobile aortic valve mass, later confirmed as papillary fibroelastoma.
A 61-year-old female with a history of ischemic heart disease, hypertension, and systemic lupus erythematosus was brought to the emergency department (ED) following an out-of hopsital cardiac arrest with ongoing cardiopulmonary resuscitation (CPR). According to her daughter, she collapsed suddenly at approximately 9 pm with agonal breathing. Bystander CPR was initiated immediately, and paramedics arrived about 10 minutes later, providing Advanced Cardiac Life Support (ACLS). During prehospital resuscitation, she underwent 14 cycles of CPR, achieved return of spontaneous circulation twice, and experienced two episodes of unstable ventricular tachycardia requiring defibrillation. The estimated total downtime before sustained return of spontaneous circulation was approximately 38 minutes.
Upon arrival to the ED, the patient remained in cardiac arrest with ongoing CPR and a laryngeal mask airway in place. Return of spontaneous circulation was achieved in the ED, after which she was intubated for airway protection and connected to a mechanical ventilator. Induction was performed with etomidate and succinylcholine, and sedation was maintained with midazolam and fentanyl. The initial ECG, obtained post-return of spontaneous circulation, demonstrated anterolateral STEMI (Image 1), prompting cardiology consultation and activation of the STEMI code. A
Image 1. Initial electrocardiogram demonstrating ST-segment elevation in leads V1-V4 (arrows), consistent with anterior STsegment elevation myocardial infarction.
CPC-EM Capsule
What do we already know about this clinical entity?
Papillary fibroelastomas (PFE) are rare cardiac tumors that can cause embolization or obstruct coronary ostia, leading to myocardial ischemia or arrhythmias.
What makes this presentation of disease reportable?
Intermittent obstruction of the left main coronary ostium by a mobile PFE causing recurrent cardiac arrest and transient ST-Elevation Myocardial Infarction (STEMI) is extremely rare.
What is the major learning point?
Transient STEMI or unexplained cardiac arrest should prompt evaluation for structural causes like PFE when common etiologies do not fully explain the presentation.
How might this improve emergency medicine practice?
Emergency medicine clinicians should consider structural lesions in unexplained arrest or transient electrocardiogram changes and use early advanced cardiac imaging to avoid missed diagnoses.
repeat ECG five minutes later showed resolution of the ST elevations and a new right bundle branch block (Image 2). Shortly thereafter, the patient experienced another cardiac arrest with a non-shockable rhythm; return of spontaneous
Image 2. Repeat electrocardiogram post return of spontaneous circulation shows resolution of the ST-segment elevations and a new right bundle branch block (arrows).
Kanaa et al. When STEMI Isn’t STEMI: Cardiac Arrest from Aortic Valve Papillary Fibroelastoma – A Case Report
circulation was regained after two cycles of CPR. She subsequently developed hypotension, prompting discontinuation of midazolam and fentanyl and initiation of a norepinephrine infusion. Peak troponin reached 390 nanograms per liter (ng/L) (reference range: < 34 ng/L), indicating significant myocardial injury.
After achieving return of spontaneous circulation, the patient regained consciousness and self-extubated, demonstrating spontaneous breathing and intact neurological function. A non-contrast brain computed tomography (CT) was unremarkable, and point-of-care echocardiography demonstrated normal left ventricular systolic function without regional wall motion abnormalities. Due to transient ECG changes and hemodynamic instability, pulmonary embolism was considered and ruled out via a negative CT pulmonary angiogram (CTPA). Aortic dissection was also considered but deemed unlikely due to the transient STsegment elevation on ECG and the lack of radiological evidence of dissection on CTPA.
The patient was admitted under cardiology for further management and evaluation with percutaneous coronary intervention. Coronary angiography revealed non-flowlimiting stenoses in the mid left anterior descending (LAD) and distal right coronary arteries (RCA). A departmental echocardiogram identified an aortic root mass, further characterized by transesophageal echocardiography as a 1.5 × 1.0-cm lesion attached to the left coronary cusp of the aortic valve (Image 3), later identified as PFE. The mass was surgically resected, and the patient made a full recovery without post-arrest neurological deficits.
Sudden cardiac arrest refers to the abrupt cessation of cardiac activity, triggered by malignant ventricular arrhythmias such as sustained pulseless ventricular tachycardia and ventricular fibrillation,4 resulting in hemodynamic collapse. Coronary artery disease, the underlying cause in over 75% of sudden cardiac death cases in developed countries,5 often leads to these arrhythmias. While the causes of sudden cardiac death vary by age, acute coronary syndrome accounts for nearly 50% of cases in individuals ≥ 40 years of age.6
This case highlights a rare and diagnostically challenging cause of sudden cardiac arrest. The initial suspected cause of the arrest was STEMI, based on the patient’s presentation and past medical history. This prompted immediate resuscitation as per the ACLS protocol and ACS management of acute coronary syndrome. However, after the second return of spontaneous circulation, a repeat ECG showed resolution of ST elevations, prompting reconsideration of the diagnosis and broadening the differential. Hypoxia was excluded, as the patient was intubated and mechanically ventilated. She experienced fluctuating blood pressure and several episodes of cardiac arrest. As sedation was tapered, she regained consciousness and self-extubated. These events, with the evolving ECG findings and fluctuating hemodynamic status, prompted consideration of other potential causes, including pulmonary embolism, which was ruled out via a negative CTPA, and aortic dissection, which was also excluded given the transient ST-segment elevation on ECG and the absence of dissection on CTPA. After stabilization, angiography showed non-flow-limiting LAD and RCA stenosis. Transesophageal echocardiography revealed a 1.5 × 1.0-cm mobile mass on the left coronary cusp intermittently obstructing the left main ostium, confirmed through histopathology as a PFE and was resected surgically.
Recurrent ventricular tachycardia and cardiac arrest were likely due to intermittent obstruction of the left main coronary ostium by the mobile PFE. This mechanism is supported by previous reports where transient ischemia from fibroelastomainduced obstruction triggered arrhythmias.7 Return of spontaneous circulation between arrests likely indicates intermittent resolution of the obstruction, permitting transient restoration of coronary perfusion.
Image 3. Transesophageal echocardiography showing a 1.5 × 1.0 centimeters, highly mobile lesion attached to the left coronary cusp of the aortic valve (arrows). The mass has a very small neck and intermittently obstructed the left main coronary ostium.
Similar cases of papillary fibroelastoma causing coronary obstruction have been reported in the literature. Talari et al described a 73-year-old woman with recurrent cardiac arrests and an inferior STEMI caused by a PFE obstructing the anterior coronary cusp, ultimately requiring surgical resection.8 Raheela et al reported a 68-year-old woman who developed acute myocardial infarction due to a PFE on the right coronary cusp, with distal coronary embolization resulting in ST-segment elevation.9 Ismaiel et al
When STEMI Isn’t STEMI: Cardiac Arrest from Aortic Valve Papillary Fibroelastoma – A Case Report Kanaa et al.
described a 39-year-old male presenting with inferior STEMI and a right coronary cusp PFE detected by echocardiography and surgically removed.10 Ramirez et al reported a 63-yearold woman presenting with angina; myocardial perfusion imaging showed anterior ischemia, and cardiac CT angiography revealed a PFE on the left coronary cusp causing intermittent left main coronary artery occlusion during systole and early diastole, which resolved after surgical resection.11 Most previously reported cases involve the right coronary artery or its branches, whereas obstruction at the left main ostium, as seen in our patient and in Ramirez et al, is less commonly reported. These cases highlight the importance of considering structural cardiac lesions, such as PFE, in patients presenting with transient ischemic ECG changes or unexplained cardiac arrest.
Although primary cardiac tumors are rare (0.02-0.45%), their potential to cause fatal events through embolization or coronary obstruction warrants heightened clinical awareness. Papillary fibroelastoma is the most common valvular tumor and second only to myxoma overall.12 Despite being benign and slow-growing, PFEs are clinically significant due to their high mobility and embolic risk.13 Rarely, they may intermittently obstruct coronary ostia, triggering arrhythmias and cardiac arrest,7 as demonstrated in this case. Transesophageal echocardiography remains the diagnostic gold standard, offering near-100% sensitivity, especially for small tumors.14 Surgical resection is recommended for mobile or symptomatic PFEs to prevent recurrence and sudden death.15.
This case underscores the importance of maintaining a broad differential diagnosis in cases of sudden cardiac arrest and incorporating advanced cardiac imaging early in the evaluation of unexplained cases of cardiac arrest, especially when there is rapid evolution in clinical status, ECG findings, and hemodynamic parameters. Rare structural causes such as PFE should be considered when common etiologies are ruled out or clinical findings are inconsistent with typical presentations.
This case highlights the importance of maintaining a high index of suspicion for rare structural causes of cardiac arrest when common diagnoses like acute coronary syndrome do not fully explain the presentation. Although papillary fibroelastoma is histologically benign, it can cause sudden death through coronary obstruction. Early use of advanced cardiac imaging, particularly transesophageal echocardiography, is essential for timely diagnosis. Emergency physicians should remain especially vigilant when there is rapid evolution in clinical status or ECG changes to identify and manage these rare but potentially treatable causes of cardiac arrest.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Patient consent has been obtained and filed for the publication of this case report.
Address for Correspondence: M. Wasim Zouhir Kanaa, MD, Specialist Registrar in Emergency Medicine, Dubai Academic Health Corporation, Dubai, United Arab Emirates; Email: wasim91k@hotmail.com.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Kanaa et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Patel K and Hipskind JE. Cardiac arrest. In: StatPearls. Treasure Island, FL: StatPearls Publishing. 2023. Available from: https://www. ncbi.nlm.nih.gov/books/NBK534866/. Accessed July 10, 2025.
2. Grolla E, Vestra MD, Zoffoli G, et al. Papillary fibroelastoma, unusual cause of stroke in a young man: a case report J Cardiothorac Surg.2017;12(1):33.
3. Neupane G, Sharma R, Parajuli R, et al. Cardiac papillary fibroelastoma and cerebrovascular events: a systematic review. CJC Open.2024;6(11):1259–73.
4. Myerburg RJ and Junttila MJ. Sudden cardiac death caused by coronary heart disease. Circulation.2012;125(8):1043–52.
5. Yow AG, Rajasurya V, Sharma S. Sudden cardiac death. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2019. Available from: https:// www.ncbi.nlm.nih.gov/books/NBK507854/. Accessed July 10, 2025.
6. Waldmann V, Karam N, Bougouin W, et al. Burden of coronary artery disease as a cause of sudden cardiac arrest in the young. J Am Coll Cardiol.2019;73(16):2118–20.
7. Maludum O, Ugoeke N, Mahida H, et al. Papillary fibroelastoma on the aortic valve presenting as multiple cardiac arrests from electrical storm due to ischemia in patient without previous cardiac history. HeartRhythm Case Rep. 2018;5(3):134–7.
8. Talari MP, Freitas A, Patel R, et al. Cardiac papillary fibroelastoma: a case report review. J Hosp Med. 2024;19(S1):Abstract 877.
9. Raheela F, Talpur AS, Rasmussen M, et al. Cardiac papillary fibroelastoma: a rare cause of ST-segment elevation myocardial infarction: a case report. Ann Med Surg (Lond). 2023;85(11):5800-3.
10. A Ismaiel M, Salah Shehata M, Khaleel Taha A, et al. Accidentally discovered aortic valve papillary fibroelastoma in a patient presenting
Kanaa et al. When STEMI Isn’t STEMI: Cardiac Arrest from Aortic Valve Papillary Fibroelastoma – A Case Report with inferior myocardial infarction. Cureus 16(10): e72783.
11. Ramirez AY, Kashuba-Shanahan KM, Cavallaro JA, et al. Mobile papillary fibroelastoma leading to severe chest pain via left main occlusion. JACC Case Rep. 2025;30(16):103535.
12. Kumar V, Soni P, Hashmi A, et al. Aortic valve fibroelastoma: a rare cause of stroke. BMJ Case Rep. 2016;2016:bcr2016217631.
13. Ikegami H, Andrei AC, Li Z, et al. Papillary fibroelastoma of the aortic valve: analysis of 21 cases, including a presentation with cardiac arrest. Tex Heart Inst J.2015;42(2):131–5.
14. Neupane G, Sharma R, Parajuli R, et al. Cardiac papillary fibroelastoma and cerebrovascular events: a systematic review. CJC Open. 2024;6(11):1259–73.
15. Westhof F, Chryssagis K, Liangos A, et al. Aortic valve leaflet reconstruction after excision of a papillary fibroelastoma using autologous pericardium. Thorac Cardiovasc Surg. 2007;55(3):204–7.
Amanda H. Bjornstad, MD
Natalie Oberhauser-Lim, MD
Tammy Phan, BS, CCRP
Emmelyn J. Samones, CCRP
Timothy Young, MD
Section Editor: Melanie Heniff, MD, JD
Loma Linda University Medical Center, Department of Emergency Medicine, Loma Linda, California
Submission history: Submitted June 11, 2025; Revision received July 23, 2025; Accepted September 10, 2025
Electronically published January 1, 2025
Full text available through open access at http://escholarship.org/uc/uciem_cpcem
DOI: 10.5811/cpcem.48533
Introduction: Patients with psychiatric concerns often present to an emergency department (ED) for medical evaluation prior to inpatient psychiatry placement. One diagnosis to consider prior to disposition is autoimmune encephalitis (AIE). This report describes a pediatric patient who presented with psychiatric symptoms that required inpatient admission and workup to diagnose a rare form of AIE.
Case Report: A 16-year-old female with no known past medical history presented as a transfer from an outside hospital for medical evaluation of two days of auditory and visual hallucinations. Initial labs and imaging were unremarkable. Due to the acuity of her symptoms and abnormal vital signs, she was admitted to the hospital for further medical workup. After almost three weeks inpatient and multiple specialist consultations, she was diagnosed with anti-heavy chain neuronal intermediate filament AIE. The next month of admission included treatment with immunomodulators, antibiotics for associated infections, and malignancy evaluation. Symptoms resolved, and the patient was discharged. The patient remained asymptomatic on immunotherapies, and without psychiatric medications, the following year.
Conclusion: During evaluation of psychiatric concerns in the ED, it is essential to consider organic causes of behavioral changes, which can be difficult to discern. Autoimmune encephalitis can be subtle. Features such as autonomic dysregulation, acute or subacute symptom onset, recent infection, autoimmune or malignancy history, cognitive deficits, or focal neurologic findings should raise clinical suspicion. For patients with psychiatric symptoms, the role of an emergency physician is not to diagnose autoimmune encephalitis, but to recognize nuances in patient presentations to best direct proper workup, treatment, and disposition. [Clin Pract Cases Emerg Med. 2025;10(1):68-71.]
Keywords: Autoimmune encephalitis; pediatric; hallucinations; neuronal intermediate filament; case report.
Mental health concerns are common in pediatric emergency departments (PED).1 Often, children are sent to PEDs for medical stabilization prior to admission to inpatient psychiatric facilities. One uncommon, but vital, diagnosis to consider prior to disposition is autoimmune encephalitis (AIE). Autoimmune encephalitis is a central nervous system (CNS) inflammatory disease process caused by an autoimmune response toward
various CNS antigens.2-4 Presenting symptoms are vast and variable, including gastrointestinal (GI) upset, headache, seizures, movement disorders, and behavioral changes that can resemble psychosis.2,3,5
If left unrecognized or untreated, AIE can have devastating effects, including persistent cognitive dysfunction, neurologic deficits, and death, with mortality rates as high as 6%.2 Although AIE is uncommon, with an incidence of five to
et al. Case Report: Pediatric Hallucinations and Anti-NIF Autoimmune Encephalitis
eight cases per 100,000 people in the general population, prompt identification, workup, and disposition are essential, as early diagnosis and treatment have been shown to significantly improve outcomes.2-4 Emergency physicians will frequently be the first to recognize symptoms and determine which patient presentations require further investigation.
A 16-year-old, fully vaccinated female in eleventh grade with no past medical history presented as a transfer from an outside hospital for acute onset auditory and visual hallucinations. The patient endorsed hallucinations that started in seventh grade; contrary to this, her parents reported no psychotic symptoms prior to the 48 hours before presentation. The patient reported auditory and visual hallucinations of a flying green dinosaur, hearing voices that told her to hurt herself, and a sense that people were following her. Throughout the interview, the patient responded to internal stimuli and reacted to her visual hallucinations of the dinosaur.
Per the patient’s mother and father, within the prior month the patient’s excellent grades declined, and her interest in social interaction and participation in extracurricular activities diminished. Her parents brought her to the PED due to worsening bizarre behavior and actively interacting with hallucinations for two days, which they had not previously observed. The patient and her family traveled to the Philippines three weeks prior, and multiple members of the family, including the patient, developed a cough during this time. Per parents, the patient did not have a history of substance use, emotional or physical trauma, psychiatric diagnoses, or need for psychotherapy. Additionally, there was no family history of schizophrenia or other psychiatric disorders.
Upon presentation to the PED, the patient was afebrile, normotensive, persistently tachycardic to the 140s beats per minute, tachypneic to the 20s breaths per minute but saturating well on room air. Of note, she was febrile to 100.6º Fahrenheit at the transferring hospital. Physical exam was notable for tremulousness, diaphoresis, no respiratory distress or abnormal lung sounds, tracking and interacting with internal stimuli, normal neck range of motion, normal pupils without significant miosis or mydriasis, and non-focal neurologic exam, including normal gait. The patient’s abnormal vital signs, ill appearance, and rapid symptom onset raised concern for organic etiology.
Initial workup at the transferring hospital had the following pertinent findings: no leukopenia/leukocytosis; no anemia; no thrombocytopenia/thrombocytosis; no electrolyte derangements; normal thyroid-stimulating hormone; negative urine drug screen; urinalysis with no infection, hematuria, proteinuria, or glucosuria; and chest radiograph with no abnormalities. In the PED, a non-contrast computed tomography (CT) head was unremarkable with no mass, edema, or hemorrhage. Lumbar puncture was attempted in the PED without return of cerebrospinal fluid (CSF). No
CPC-EM Capsule
What do we already know about this clinical entity?
Patients with psychiatric concerns commonly present to the emergency department (ED) for medical evaluation; underlying organic causes can be difficult to identify.
What makes this presentation of disease reportable?
This patient presented with a common pediatric ED concern of hallucinations but with signs of autoimmune encephalitis (AIE).
What is the major learning point?
History and exam findings that favor AIE versus psychiatric diagnosis include acute or subacute presentation, recent infection, and history of autoimmunity.
How might this improve emergency medicine practice?
Enhanced clinical suspicion for AIE in patients with psychiatric concerns could improve patient outcomes.
antibiotics were started in the PED due to low suspicion for meningitis given the absence of meningismus, lethargy, seizures, or significant lab or CT findings. However, due to her persistent ill appearance and vital sign abnormalities, the PED team determined she was not medically stable for psychiatric facility admission; therefore, the general pediatric team was consulted for admission.
Upon admission, an electroencephalogram was completed, per the recommendation of the pediatric neurology team, with no abnormal findings. A rapid response was called on admission day two due to the patient demonstrating complete catatonia, for which the consulting psychiatry team recommended treatment with lorazepam; catatonic symptoms subsequently improved over a period of days. The psychiatric team’s initial differential diagnosis included psychosis but emphasized the need to rule out organic etiology. Additional unremarkable workup included magnetic resonance imaging (MRI) of the brain with and without contrast, respiratory viral panel, pregnancy test, human immunodeficiency virus (HIV) testing, hepatitis panel, and herpes simplex virus types 1 and 2 serum polymerase chain reaction.
A lumbar puncture was performed by the interventional radiology team, and the following CSF studies were normal: cell count; glucose; protein; meningitis panel (viral, bacterial,
Case Report: Pediatric Hallucinations and Anti-NIF Autoimmune Encephalitis
and fungal); and culture. On hospital day 4, a GI panel resulted positive for cryptosporidium, for which she was treated with nitazoxanide. On hospital day 8, the consulting psychiatry team recommended inpatient psychiatry transfer. The patient was deemed medically stable by the primary pediatrics team, but she developed difficulty with independent feeding that precluded transfer. Serum and CSF autoimmune encephalitis panels were pending at this time. Per the recommendation of the consulting psychiatry team, olanzapine was added to her medication regimen on hospital day 11 for treatment of persistent psychosis.
On hospital day 18, a CSF autoimmune encephalitis panel showed positive cell-based assay and titer (1:16) for antiheavy chain neuronal intermediate filament antibody (anti-NIF Ab). Given the strong association of anti-NIF Ab with paraneoplastic processes, MRI of the chest, abdomen, and pelvis were completed but showed no evidence of malignancy. She ultimately received intravenous (IV) steroids, IV immunoglobulin (IVIG), rituximab, and five sessions of plasmapheresis over the next month. Due to the association between ehrlichiosis and anti-NIF Ab, she was empirically treated with doxycycline. On hospital day 57, the patient was discharged home on prednisone, olanzapine, lorazepam, and monthly IVIG. Outpatient positron emission tomography showed no evidence of neuroendocrine tumor or other malignancy. The patient now follows in outpatient clinic with reported continued resolution of symptoms more than one year after index presentation, continued immunomodulation therapy, and discontinuation of psychiatric medications.
Autoimmune encephalitis can be difficult to diagnose due to its rarity. Also, much of pediatric AIE data is restricted to anti-N-methyl-d-aspartate receptor Ab (anti-NMDAR Ab), as it is the most commonly isolated auto-antibody and best characterized AIE syndrome in the pediatric population.2-4 However, many additional auto-antibodies have been identified as causes of pediatric AIE.2,3,5 Despite the uniqueness of pediatric anti-NIF autoimmune encephalitis, the post-infectious presentation is similar to many other pediatric AIE cases.
Infections documented to precede AIE include herpes simplex virus (strongly associated with anti-NMDAR autoimmune encephalitis), Haemophilus influenzae, enterovirus, mycoplasma, streptococcus, varicella zoster, cytomegalovirus, Ebstein-Barr, adenovirus, and rickettsial pathogens.2,3,5 Cases of post-infectious adult anti-NIF autoimmune encephalitis include anaplasma, HIV, ehrlichiosis, and severe acute respiratory syndrome coronavirus 2.6-8 Post-infectious anti-NIF autoimmune encephalitis is less common than paraneoplastic anti-NIF autoimmune encephalitis.7
Recognizing AIE, especially in an acute setting, is challenging due to symptom variability and subtlety. Common
pediatric symptoms include autonomic dysfunction, movement disorders (eg, dystonia), language disorders (eg, mutism), sleep/wake cycle disturbances, and neurologic dysfunction (eg, seizures). Only 60% of pediatric patients present with psychiatric symptoms.2-5 This is different from adults who present with neuropsychiatric symptoms as a defining AIE feature.4 Cognitive dysfunction (eg, memory loss, inattention, etc.) and viral prodrome (eg, fever, headache, etc.) commonly occur in both populations.4,9,10 Anti-neuronal intermediate filament AIE, specifically, has three distinct phenotypes in adults: encephalopathy predominate; cerebellarataxia predominant; and myeloradicular neuropathies.7
In patients with psychiatric symptoms, differentiating psychological etiology from organic causes is clinically complex, and the two diagnoses may not be mutually exclusive. Psychiatric patients tend to have more anxious symptoms than AIE patients, although this may be difficult to clinically discern. “Red” and “yellow” flag symptoms that should raise clinical suspicion for AIE in psychiatric presentations are epileptiform activity, facial dystonia, bulbar symptoms, focal neurologic deficits, autonomic dysfunction, hyponatremia, headache, history of autoimmune disorders, and rapid psychosis progression despite treatment.11,12 Acute or subacute presentation may also be more suggestive of organic vs psychiatric etiology.13,14 In this particular presentation, the patient’s sustained tachycardia, diaphoresis (autonomic dysregulation), and symptom acuity raised suspicion for organic etiology.
A diagnostic approach exists for adult AIE, and although validated through some studies, there is opportunity to broaden inclusion criteria given significant variability in patient presentation.14,15 The current diagnostic criteria are considered highly sensitive and specific in pediatric populations, but most patients do not fulfill criteria until two weeks after symptom onset.11 Cellucci and colleagues have proposed modified pediatric criteria and an algorithm to account for differences from adult presentations.3 For an emergency physician evaluating a potential AIE patient, the proposed adult and pediatric criteria are beneficial to consider; however, it is essential to remember that AIE is not ruled out if criteria are not fulfilled completely. Diagnosis of AIE requires clinical suspicion, concordant history, extensive workup, and consideration of the most up-to-date diagnostic criteria and research
Autoimmune encephalitis is rare and can be difficult to recognize. When determining whether to pursue an AIE workup, important historical data to acquire include the following: acuity of behavioral changes; recent infection; recent travel; personal or family history of autoimmune disorders and/or malignancy; prior psychiatric history; toxic ingestions; sleep cycle changes; and neurologic symptoms. Physical exam should include multiple sets of vitals to
Bjornstad et al. Case Report: Pediatric Hallucinations and Anti-NIF Autoimmune Encephalitis
evaluate for autonomic dysregulation, thorough neurologic exam, and cognitive testing. Current AIE criteria and algorithms can also be helpful to support proper management but should not be used to rule out an AIE diagnosis. It is important to understand that a final diagnosis of AIE is not required in the emergency department, but it is a crucial diagnosis to consider and appropriately work up for safe disposition and treatment to improve patient outcomes.
The authors attest that their institution does not require Institutional Review Board approval. Patient consent has been obtained and filed for the publication of this case report.
Address for Correspondence: Emmelyn J. Samones, CCRP, Loma Linda University Medical Center, Department of Emergency Medicine, 11234 Anderson St., Room A890A, Loma Linda, CA 92354. Email: Esamones@llu.edu.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Bjornstad et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Sheridan DC, Spiro DM, Fu R, et al. Mental health utilization in a pediatric emergency department. Pediatr Emerg Care 2015;31(8):555-9.
2. Garg D, Mohammad SS, Sharma S. Autoimmune encephalitis in children: an update. Indian Pediatr. 2020;57(7):662-70.
3. Cellucci T, Van Mater H, Graus F, et al. Clinical approach to the diagnosis of autoimmune encephalitis in the pediatric patient. Neurol Neuroimmunol Neuroinflamm. 2020;7(2), e663.
4. Esposito S, Principi N, Calabresi P, et al. An evolving redefinition of autoimmune encephalitis. Autoimmun Rev. 2019;18(2):155-163.
5. Li Q, Fu N, Han Y, et al. Pediatric autoimmune encephalitis and its relationship with infection. Pediatr Neurol. 2021;120:27-32.
6. Merati M, Rucker JC, McKeon A, et al. A case of opsoclonusmyoclonus-ataxia with neuronal intermediate filament IgG detected in cerebrospinal fluid. J Neuroophthalmol. 2022;42(2):278-81.
7. McKeon A, Shelly S, Zivelonghi C, et al. Neuronal intermediate filament IgGs in CSF: autoimmune axonopathy biomarkers. Ann Clin Transl Neurol. 2021;8(2):425-439.
8. Clinical and paraclinical features of non-paraneoplastic NIF-mediated disease associated with concurrent SARS-CoV-2 Infection. Neurology. Accessed April 19, 2025. https://www.neurology.org/
9. Newman MP, Blum S, Wong RCW, et al. Autoimmune encephalitis. Intern Med J. 2016;46(2):148-157.
10. Brenton JN and Goodkin HP. Antibody-mediated autoimmune encephalitis in childhood. Pediatr Neurol. 2016;60:13-23.
11. Ho ACC, Mohammad SS, Pillai SC, et al. High sensitivity and specificity in proposed clinical diagnostic criteria for anti-N-methyl-Daspartate receptor encephalitis. Dev Med Child Neurol 2017;59(12):1256-1260.
12. Herken J and Prüss H. Red flags: clinical signs for identifying autoimmune encephalitis in psychiatric patients. Front Psychiatry 2017;8:25.
13. Abboud H, Probasco JC, Irani S, et al. Autoimmune encephalitis: proposed best practice recommendations for diagnosis and acute management. J Neurol Neurosurg Psychiatry. 2021;92(7):757-768.
14. Graus F, Titulaer MJ, Balu R, et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol. 2016;15(4):391-404.
15. Orozco E, Valencia-Sanchez C, Britton J, et al. Autoimmune encephalitis criteria in clinical practice. Neurol Clin Pract 2023;13(3):e200151.
Brittany Franco, DO
Amanda Karski, MD
Kevin Boehm, DO, MSc
Section Editor: Joel Moll, MD
Submission history: Submitted May 5, 2025; Revision received July 8, 2025; Accepted September 22, 2025
Electronically published January 13, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.47345
Introduction: Spontaneous splenic vein rupture is a rare condition associated with a high risk of mortality. Only a few cases have been documented, all of which have been linked to underlying predisposing conditions. In this case, however, we present a previously healthy patient with no identifiable risk factors.
Case Report: A 64-year-old male presented to the emergency department with acute-onset abdominal pain and nausea. Physical exam revealed a rigid abdomen with diffuse tenderness and guarding. Serial labs revealed a progressively decreasing hemoglobin and red blood cell count. Computed tomography demonstrated a ruptured splenic vein with a large volume of retroperitoneal hemorrhage. Laparotomy identified a large retroperitoneal hematoma with hemoperitoneum, and embolization was performed by interventional radiology. The patient recovered after one week in the surgical intensive care unit and was subsequently discharged home with no complications to date.
Conclusion: This case underscores the importance of maintaining a high clinical suspicion in patients with acute, unexplained abdominal pain, and emphasizes the crucial role of emergency physicians in the timely recognition and management of such conditions. Given the scarcity of existing literature, this case provides valuable insight into the presentation and management of spontaneous splenic vein rupture in previously healthy individuals, thereby enhancing clinical awareness of this rare and life-threatening condition. [Clin Pract Cases Emerg Med. 2025;10(1):72-75.]
Keywords: splenic vein; spontaneous rupture; retroperitoneal hemorrhage; case report.
INTRODUCTION
Spontaneous splenic vein rupture is a rare and potentially life-threatening condition characterized by the sudden rupture of the splenic vein, leading to significant retroperitoneal hemorrhage and hypovolemic shock in the absence of preceding trauma. The etiology remains unclear; however, predisposing conditions may include splenic vein aneurysm or thrombosis, liver cirrhosis, and pregnancy.1-5 In this report we present a rare case of spontaneous splenic vein rupture in a previously healthy patient with no identifiable predisposing factors. With only a few documented cases in the medical
literature, recognizing this condition requires a high index of suspicion when evaluating patients with unexplained abdominal pain. This case underscores the critical role of emergency physicians in the timely identification and management of spontaneous vascular injuries to prevent potentially fatal complications.
A 64-year-old male with a medical history of hypertension, hyperlipidemia, and benign prostatic hyperplasia presented to the emergency department (ED) for acute-onset
abdominal pain and nausea for three hours. The patient stated that he had been bicycling earlier that day and explicitly denied any trauma, including a bicycle accident or handlebar injury. Social history was negative for tobacco, alcohol, or illicit drug use. He denied fever, chills, vomiting, diarrhea, constipation, melena, hematochezia, hematemesis, dysuria, hematuria, syncope, or weakness. Upon arrival to the ED, his vital signs were as follows: heart rate, 76 beats per minute; blood pressure, 124/85 millimeters of mercury; oxygen saturation, 94% on room air; and temperature, 97.9 ° Fahrenheit. Neurologic examination was non-focal. The mucosa was pink and moist, and there was no conjunctival pallor. Cardiopulmonary examination was unremarkable. Pulses were equal in bilateral upper and lower extremities. Abdominal exam revealed a rigid abdomen with diffuse tenderness, more pronounced in the epigastrium and left upper quadrant, and with involuntary guarding. There was no costovertebral angle, tenderness, and McBurney and Murphy signs were negative.
Initial labs included a complete blood count (CBC), comprehensive metabolic panel, lipase, troponin, magnesium, type and screen, prothrombin time and international normalized ratio (INR), Thromboelastogram, and urinalysis. CBC was remarkable for mild normocytic anemia (hemoglobin 12.8 grams per deciliter (g/dL) (reference range: 13.0-17.3 g/dL) without leukocytosis or thrombocytopenia. Repeat CBC (three hours later), was notable for a decrease in hemoglobin to 10.9 g/dL. Comprehensive metabolic panel, lipase, and troponin were unremarkable. The INR was 1.3. thromboelastogram, demonstrated a shortened R-time of 3.7 minutes (4.6-9.1 minutes) but was otherwise normal.
Initial computed tomography (CT) of the abdomen and pelvis with intravenous contrast demonstrated abnormal dilatation of the splenic vein with large volume surrounding fluid of varying densities, suggesting splenic vein rupture with extensive retroperitoneal hemorrhage (Image 1). Computed tomography of the abdomen and pelvis with venous and delayed venous phases was subsequently ordered and confirmed splenic vein rupture at the level of the midpancreatic body with active extravasation (Image 2). There was no evidence of splenic artery aneurysm or other vascular anomaly noted (Video).
The patient initially received one liter of lactated Ringer’s, ondansetron, famotidine, and morphine without resolution of his symptoms. He grew increasingly uncomfortable, prompting the administration of hydromorphone. After receiving the results of the CT abdomen and pelvis, urgent surgical and interventional radiology consultations were placed. The patient was admitted to the surgical intensive care unit (SICU) in stable condition. Given the venous and retroperitoneal nature of the bleed and the patient’s stable hemodynamic status at the time of admission, the surgical team opted for conservative management and close monitoring to allow tamponade in the
CPC-EM Capsule
What do we already know about this clinical entity?
Spontaneous splenic vein rupture is rare, usually linked to trauma, cirrhosis, thrombosis, or pregnancy.
What makes this presentation of disease reportable?
A previously healthy patient with no identifiable risk factors presented with spontaneous splenic vein rupture.
What is the major learning point?
Maintain high suspicion for spontaneous vascular rupture in patients with acute abdominal pain, even when they are stable and without risk factors.
How might this improve emergency medicine practice?
Awareness of spontaneous vascular rupture in unexplained abdominal pain will enable clinicians to play a critical role in early imaging and timely intervention.
retroperitoneum. However, the patient’s hemoglobin levels continued to decline, and his clinical status deteriorated, necessitating emergent surgical intervention.
Emergent laparotomy revealed a large retroperitoneal hematoma with hemoperitoneum. The patient was stabilized after receiving two units of packed red blood cells and fresh frozen plasma. Interventional radiology then performed an embolization of an un-named branch of the right gastroepiploic artery. The abdomen was irrigated and left open with a wound vacuum-assisted closure in place. The patient was transferred back to the SICU in stable condition.
Two days later, he was returned to the operating room for a reopening laparotomy, washout, and wound closure. This operation revealed a stable, non-pulsatile retroperitoneal hematoma without active extravasation. No additional abdominal exploration was performed. The abdomen was then irrigated, and the wound was closed. The patient tolerated this procedure well, requiring no additional blood product administration.
He remained hemodynamically stable throughout his SICU stay. After 10 days of steady recovery with stable hemoglobin levels and no complications, he was discharged home with a structured outpatient management plan and surgical follow-up. To date, the patient has had no reported complications.
Splenic vein rupture with subsequent intra-abdominal hemorrhage is a rare condition, typically associated with
trauma.6 This case presents a patient with spontaneous splenic vein rupture occurring without trauma or underlying predisposing factors, providing valuable insight into this unique condition. The exact pathophysiology of spontaneous splenic vein rupture remains uncertain, although reported risk factors include splenic vein aneurysm or thrombosis, liver cirrhosis, and pregnancy.1-5 Documented examples include rupture of a splenic vein aneurysm during pregnancy1 and splenic vein rupture in the setting of liver cirrhosis,2 both resulting in massive intra-abdominal hemorrhage. In such cases, the proposed mechanism is largely attributed to portal hypertension. In pregnancy, the combination of mechanical compression of the inferior vena cava and portal system by the gravid uterus, increased splanchnic venous pressure due to pregnancy-induced hemodynamic changes, and hormonal alterations— particularly, elevated estrogen and progesterone— remodeling can compromise the vessel wall integrity and increase susceptibility to rupture.3-4
In patients without predisposing risk factors, as demonstrated in this case report, the underlying cause of rupture remains unknown. The rarity of spontaneous splenic vein rupture in the absence of underlying pathology cannot be emphasized enough. Of the documented cases, the clinical presentation includes acute abdominal pain, hypotension, and signs of hemorrhagic shock.1-5 This may reduce the suspicion of intra-abdominal hemorrhage in hemodynamically stable patients as seen in this case. This highlights the necessity to maintain a high index of suspicion for spontaneous vascular rupture in seemingly healthy patients with acute-onset abdominal pain in the absence of hemodynamic instability.
Imaging studies are crucial for diagnosis. Ultrasound and CT are the primary modalities used to identify intra-abdominal hemorrhage and vascular pathology.7,9 Point-of-care ultrasound (POCUS) enables rapid identification of intraperitoneal free fluid.7 Despite its clinical utility, POCUS has limitations, including reduced sensitivity in detecting retroperitoneal injuries and its reliance on the volume of free fluid for accurate detection. Experienced operators may detect volumes as low as 200 milliliters (mL), but the average volume required for detection is 619 mL.8 Computed tomography, specifically angiogram with venous and delayed venous phases, can provide detailed images to confirm the presence of a splenic vein rupture, identify the source of bleeding, and assess the extent of hemoperitoneum.9
The management of spontaneous splenic vein rupture is largely dependent on the patient’s hemodynamic status. Conservative treatment is appropriate for hemodynamically stable patients, but surgical intervention is necessary in cases of instability or rapid decompensation.10 In this case, the patient was initially managed conservatively, but as his condition deteriorated, laparotomy and embolization via interventional radiology were necessary for stabilization.
Spontaneous splenic vein rupture is a rare but potentially fatal condition characterized by the sudden rupture of the splenic vein, leading to significant intra-abdominal hemorrhage and shock. Few cases of splenic vein rupture have been reported, all of which have been linked to underlying predisposing conditions. This case report contributes to the limited body of research on spontaneous splenic vein rupture in previously healthy individuals with no identifiable predisposing factors. It emphasizes the importance of maintaining a high clinical suspicion when evaluating patients with acute, unexplained abdominal pain. This case highlights the critical role of emergency physicians in the prompt recognition and management of spontaneous vascular injuries to improve patient outcomes and prevent further morbidity and mortality.
Address for Correspondence: Brittany Franco, DO, Broward Health North Hospital, Department of Emergency Medicine, Broward Health North, Emergency Medicine Program, 201 E Sample Rd, Deerfield Beach, FL 33064. Email: bfranco@ browardhealth.org.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Franco et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Turan N, Oghan F, Boran T. Spontaneous rupture of splenic vein in a pregnant woman during a religious ritual. J Forensic Leg Med. 2007;14(7):4403.
2. Fleury L and McEnnan C. Fatal hemoperitoneum secondary to a spontaneous splenic vein rupture. Acad Forensic Pathol. 2024;14(3):112-118.
3. Abbadi K, Shrateh ON, Musleh A, et al. Life-saving intervention following spontaneous rupture of splenic vein in a young patient: a case report and review of the literature. Int J Surg Case Rep. 2023;112:108952.
4. Kane D, Keaney JM, Tunney E, et al. Splenic vein rupture in pregnancy: a case report and systematic review of the literature. Int J Gynaecol Obstet. 2025;168(1):63-68.
5. Nomura T, Keira N, Urakabe Y, et al. Fatal splenic laceration in a young woman caused by idiopathic isolated splenic vein thrombosis Intern Med. 2009;48(11):907-10.
6. Cai H, Zhang L, Xiang H. Expert consensus on interventional therapy for traumatic splenic bleeding. J Intervent Med. 2020;3(3):109-17.
7. Genthon A, Frasure S, Kinnaman K, et al. Bedside ultrasound diagnosis of a spontaneous splenic hemorrhage after tissue plasminogen activator administration. Am J Emerg Med. 2014;32(12)1553.e1-1553.e2
8. Richards JR and McGahan JP. Focused assessment with sonography in trauma (FAST) in 2017: what radiologists can learn. Radiol. 2017;283(1):30-48.
9. Verma N, Steigner ML, Suranyi PS, et al. ACR Appropriateness Criteria® Suspected Retroperitoneal Bleed. J Am Coll Radiol. 2021;18(11 Suppl):S482-S487.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
10. Leung E, Maingard J, Yeh J, et al. Contemporary endovascular management of splenic vascular pathologies. Clin Radiol 2020;75(12):960.e23-960.e34.
C. James Watson, MD*†‡
Emilie M. Burrill, PharmD*§
William S. Jaffee, DO‡¶
Maine Medical Center, Department of Emergency Medicine, Portland, Maine
Northern New England Poison Center, Portland, Maine
Tufts University School of Medicine, Boston, Massachusetts
Maine Medical Center, Acute Care Pharmacy Services, Portland, Maine
Maine Medical Center, Adult Inpatient Medicine, Portland, Maine
Section Editor: Steven Walsh, MD
Submission history: Submitted August 2, 2025; Revision received August 24, 2025; Accepted September 12, 2025
Electronically published January 16, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.49089
Introduction: Antimuscarinic toxicity, which can cause delirium and unsafe behavior, may result from an adverse effect of prescribed medications or from non-medical substance use. Physostigmine shortages have prompted use of transdermal rivastigmine for management of antimuscarinic toxicity; however, symptom control is equivocal at standard dosing.
Case Report: A patient with antimuscarinic toxicity was treated with physostigmine and transitioned to 26.6 milligrams/24 hours transdermal rivastigmine for sustained symptom control. He experienced no adverse effects and tolerated floor admission.
Discussion: There is mechanistic plausibility supporting safe, sustained control of antimuscarinic toxicity with high-dose transdermal rivastigmine. Central distribution is more rapid than serum distribution and higher doses correlate with a shorter time to peak concentration. [Clin Pract Cases Emerg Med. 2025;10(1):76-80.]
Keywords: drug overdose; muscarinic antagonists; physostigmine; rivastigmine; case report.
Physostigmine is the gold standard antidote for antimuscarinic toxicity (AMT).1,2 In 2023, persistent shortages of US-manufactured physostigmine led the US Food and Drug Administration to authorize importation of a German alternative Anticholium—physostigmine salicylate (Dr. Franz Köhler Chemie GmbH, Bensheim, Germany).3,4 The imported physostigmine remains scarce, however, and the shortage is ongoing. To manage AMT without requiring critical care admission or physostigmine re-dosing, physicians are using other centrally acting acetylcholinesterase inhibitors for symptom management.5–9
Transdermal rivastigmine is one such option. It is formulated in 4.6 milligrams (mg) per 24 hours, 9.5 mg/24 hour, and 13.3 mg/24 hour patches with a maximum approved dose of 13.3 mg/24 hours for Alzheimer dementia and Parkinson disease.10 Existing literature on transdermal
rivastigmine for AMT challenges its efficacy and time to clinical effect, particularly given the slower onset of the transdermal formulation as compared to oral rivastigmine. However, the literature reports the same dosing for Alzheimer dementia.5–9 To our knowledge there have been no prior published reports of higher dose transdermal rivastigmine for the management of AMT.
Patients with AMT are, by definition, experiencing direct muscarinic receptor antagonism. We hypothesize that this cohort requires transdermal rivastigmine in higher doses than used for Alzheimer dementia to achieve adequate symptom control. We argue that previous studies of transdermal rivastigmine for AMT were limited by insufficient dosing. We present a novel case of AMT requiring physostigmine for initial symptom control that was promptly, effectively, and safely maintained without critical care admission on high-dose 26.6 mg/24 hour transdermal rivastigmine.
A 42-year-old, 63-kilogram man with polysubstance use disorder presented to the emergency department (ED) from an inpatient detoxification center for abnormal behavior. He had enrolled in the center one day prior for medically supervised management of withdrawal from fentanyl, freebase cocaine, and ethanol. Earlier on the day of ED presentation, he exhibited erratic behavior described as kicking walls, thrashing, and moaning. His behavior was presumed related to substance withdrawal, and he was treated at the detox center with 20 mg oral olanzapine, 50 mg oral hydroxyzine, 1,500 mg oral methocarbamol, and unspecified doses of oral gabapentin, trazodone, dicyclomine, and clonidine. He had worsening mentation, nonsensical speech, and frequent unsuccessful attempts to urinate on the floor, prompting his ED evaluation.
At ED triage (time 00:00), the following vital signs were recorded: heart rate, 98 beats per minute; blood pressure, 135/90 millimeters of mercury; respirations, 19 breaths per minute; temperature, 36.8 °Celsius; pulse oximetry, 97%; and capillary glucose, 137 mg per deciliter. Promptly after arrival he was evaluated by the ED resident and attending physician, who was also a consultant toxicologist with the regional poison center. The patient provided no history, and no additional information was available from the detox center. His exam was notable for restlessness, trying to climb over the gurney railing, grabbing at space, picking at his telemetry leads, and biting at his pulse oximeter. He vocalized with nonsensical, muffled, mumbling sounds. He had nonreactive 3-millimeter pupils bilaterally, flushed skin, dry axillae, tachycardia, and mild tachypnea. There was no clonus, and patellar reflexes were 1+ bilaterally. There was no tremor and no tongue fasciculations. He repeatedly grabbed at his genitals in unsuccessful attempts to urinate. Qualitative ultrasound demonstrated a large bladder volume. Laboratory results from 00:21 (Table) were reassuring against acute metabolic disturbance or common co-ingestion.
Clinical course is shown in the Figure. The patient received two benzodiazepine doses between 00:20 and 00:29 without effect. His presentation was consistent with AMT, and he received physostigmine for diagnostic and therapeutic purposes. Pre-administration, hyperactivity prevented an interpretable electrocardiogram (ECG); however, the QRS interval by telemetry was 80-100 milliseconds (ms). At 00:38, he received 2 mg intravenous (IV) physostigmine (Köhler) over five minutes. He subsequently had improved attention and ability to remain in bed. Otherwise, his speech, picking movements, and urinary retention were unchanged. At 01:01, he received another 2 mg IV physostigmine (Köhler) over five minutes followed shortly by abrupt, complete resolution of encephalopathy, hyperactive movements, and urinary retention. There were no findings consistent with alcohol or opioid withdrawal. He recalled being at the detox center previously, and that he had been given “a whole bunch of
CPC-EM Capsule
What do we already know about this clinical entity?
Transdermal rivastigmine (tdR) can be used to treat antimuscarinic toxicity (AMT) in the absence of physostigmine.
What makes this presentation of disease reportable?
Following high-dose tdR (26.6 mg/24hours) for AMT after initial physostigmine reversal, the patient’s symptoms did not recur while admitted on telemetry.
What is the major learning point?
High-dose tdR for AMT may be an effective means of maintaining symptom control. We recommend telemetry monitoring for those receiving high-dose tdR.
How might this improve emergency medicine practice?
High-dose tdR may improve AMT and reduce intensive care admissions in the era of physostigmine shortages.
pills” from an unknown source and without additional detail. An ECG at 01:13 showed sinus tachycardia at 106 beats per minute, QRS of 99 ms, and QTc (Bazett) of 475 ms.
Anticipating AMT recrudescence and given limited physostigmine availability, transdermal rivastigmine was administered. Due to the presenting illness severity and the bedside physician’s experience with incomplete symptom control from 13.3 mg/24 hour transdermal rivastigmine, two 13.3 mg/24 hour transdermal rivastigmine patches were placed on the upper back at 01:36 (total 26.6 mg/24 hour).
Appreciating the possible time to clinical effect, application was expedited. The patient was admitted to a telemetry hospitalist service, but he continued boarding in the ED. Given the novel use of high-dose rivastigmine, monitoring for muscarinic toxicity was discussed with the admitting hospitalist, and arrangements were made for the poison center to follow the case.
The patient developed no further AMT or muscarinic toxicity. At 13:20, both rivastigmine patches were removed. There was no concern for suicidal intent. He was discharged at 35:21 after receiving subcutaneous, extended-release buprenorphine. Serum testing performed at 16:08 resulted
Antimuscarinic Toxicity Safely Managed with High-Dose Transdermal Rivastigmine: A Case Report
post-discharge. Hydroxyzine/cetirizine were detected at 27 nanograms per milliliter (ng/mL) and 220 ng/mL (5 ng/mL and 50 ng/mL reporting limits, respectively). Diphenhydramine was undetectable (50 ng/mL reporting limit). Olanzapine concentrations were not collected. The patient provided informed consent for the publication of this case.
We present the case of an adult with AMT resolved with IV physostigmine, whose symptom control was maintained outside a critical care setting using high-dose transdermal rivastigmine without adverse effect. Acute AMT is classically managed with physostigmine, which has a short time to effect of < 10 minutes but is limited by a short duration of effect of one to two hours.11,12 The physostigmine shortage has resulted in substitution with more widely available, centrally acting acetylcholinesterase inhibitors including rivastigmine.5–9
Transdermal rivastigmine has benefits compared to the oral formulation: 1) easier administration in those intolerant of oral medications or enteric tubes; 2) no dependence on normal gastrointestinal absorption kinetics; and 3) once-daily dosing with more consistent tissue concentrations.6,13 However, two retrospective cohort studies found that transdermal rivastigmine was less effective than oral rivastigmine at controlling AMT. Greene found that among 22 AMT patients, those receiving transdermal rivastigmine alone (9.5-13.3 mg/24 hours) had a five-hour median time to symptom control compared to those also receiving oral rivastigmine (two hours).8 Similarly, Chiew et al found that among 50 AMT
Watson et al.
Table 1. Laboratory values on presentation in a patient suspected of having antimuscarinic toxicity.
Laboratory Test Value (reference range)
Hemoglobin 14.6 g/dL (12.3-16.9)
Leukocytes 8.6 thousand/μL (3.6-11.8)
Platelets 385 thousand/μL (142-390)
Sodium 142 mEq/L (135-145)
Potassium 4.2 mEq/L (3.5-5.1)
Chloride 106 mEq/L (96-108)
Magnesium 2.3 mg/dL (1.6-2.6)
Carbon dioxide 23 mEq/L (21-30)
Anion gap 13 mEq/L (7-16)
Blood urea nitrogen 14 mg/dL (6-20)
Creatinine 0.87 mg/dL (0.5-1.3)
*Glucose 141 mg/dL (70-99)
Aspartate transferase 16 U/L (8-48)
Alanine transferase 17 U/L (7-55)
Bilirubin (Total) < 0.2 mg/dL (< = 1.2)
Lipase 26 U/L (13-60)
Creatine kinase 93 U/L (39-308)
Lactate 1.1 mmol/L (0.5-2.0)
Acetaminophen < 5 μg/mL
Salicylate < 10 mg/L
Ethanol < 10 mg/dL
*Abnormal value.
dL, deciliter; g, gram; L, liter; mEq, milliequivalent; mg: milligram; mmol: millimole; μg: microgram; μL: microliter; U: unit.
Figure. Clinical course: Shortly after arrival, the patient received a clinical diagnosis of antimuscarinic toxicity. He was treated with both lorazepam and diazepam with minimal effect and subsequently received two doses of IV physostigmine 2 mg and had complete resolution of his symptoms. Transdermal rivastigmine (26.6 mg/24 hour) was placed. The patient developed no recurrent antimuscarinic toxicity nor did he develop any muscarinic adverse effects from the rivastigmine; his heart rate remained in the normal range. The rivastigmine was discontinued, and the patient was discharged on hospital day two. IV, intravenous.
Watson et al. Antimuscarinic Toxicity Safely Managed with High-Dose Transdermal Rivastigmine: A Case Report
patients, 73% of those receiving transdermal rivastigmine (mode 9.5 mg/24 hours) required additional sedation compared to 32% of those receiving oral rivastigmine.9 Both investigators proposed that transdermal rivastigmine’s slower time to serum peak concentration (TMAX; 8-16 hours10) was contributory to its inferior effects.8,9
Our patient, however, transitioned from requiring multiple doses of physostigmine to high-dose transdermal rivastigmine without recurrent AMT. This may indicate that transdermal rivastigmine was clinically effective within one to two hours of being placed, as it is mechanistically plausible that higher doses facilitate more rapid, complete symptom control. Transdermal rivastigmine’s serum TMAX shortens as the dose increases.14 Furthermore, rivastigmine’s rapid central distribution results in a cerebral spinal fluid (CSF) TMAX of only 1.4-2.6 hours despite the lagging serum TMAX 10 Rivastigmine’s clinical effects in patients with Alzheimer dementia correlate directly with CSF acetylcholinesterase activity, which in turn correlates with rivastigmine dose.15 Therefore, we theorize that the higher transdermal rivastigmine dose contributed to more rapid CSF acetylcholinesterase inhibition and onset of clinical effect before the physostigmine was no longer effective.
Safety—that is, monitoring against muscarinic toxicity— was a primary consideration when implementing high-dose rivastigmine. While not requiring critical care, the patient underwent telemetry monitoring for the duration of his rivastigmine treatment, and atropine was immediately available. We were comfortable pursuing a high dose due to the following considerations: 1) the tolerability profile of transdermal rivastigmine in patients with Alzheimer dementia even at 17.4 mg/24 hour10; 2) the absence of documented adverse events attributed to transdermal rivastigmine in previous AMT cases5,6,8,9; 3) strong communication with a collaborative hospitalist team with oversight by the regional poison center; and 4) the toxicodynamic principle that developing muscarinic symptoms from higher rivastigmine doses would be unlikely given the clear muscarinic receptor antagonism our patient was already experiencing. While this patient demonstrated no muscarinic toxicity, we still recommend telemetry monitoring in future cases.
An unexpected benefit of the physostigmine shortage has been the development of treatment protocols requiring less-frequent dosing than is allowed by IV physostigmine, an issue previously raised by Dawson and Buckley.12 Physostigmine has a one to two hour duration of effect and is typically administered in either an ED or critical care setting given the historic risk of precipitous muscarinic toxicity or seizure.12 In the case presented here, transitioning to transdermal rivastigmine was viewed as protective against the need for additional physostigmine or critical care services; therefore, the patient was admitted to a telemetry floor. Without durable AMT control, the admitting team would have requested an intensive care unit admission. If physostigmine
becomes broadly available again, physostigmine bolus followed by transdermal (or oral) rivastigmine maintenance should be considered for resource conservation.
This case report has limitations. First, generalizability is limited and causative conclusions cannot be made. Second, while it is possible that the patient would have had a similar course without transdermal rivastigmine or with a dose lower than 26.6 mg/24 hour, the need for two physostigmine doses for initial symptom control and the lack of any muscarinic effects with a high rivastigmine dose argue against this possibility. Third, while the patient’s examination and response to physostigmine clearly support AMT, he never mounted tachycardia to the degree generally expected with this toxidrome. This may be related to clonidine administered prior to transfer from the detox center. Fourth, there is no definitive causative agent for the patient’s symptoms. Hydroxyzine and cetirizine were detected late in the clinical course at inconclusive concentrations;16 it is unknown how these compounds contributed to the initial presentation or whether other antimuscarinic agents (olanzapine) were involved. The clinical toxidrome and its resolution with physostigmine, however, support this as a case of AMT.
High-dose transdermal rivastigmine (26.6 mg/24 hour) may provide efficient, effective, and safe symptomatic control following physostigmine bolus in carefully selected patients. Those receiving high-dose transdermal rivastigmine require telemetry monitoring and prompt availability of atropine, but they may not need critical care admission. Larger cohort studies are indicated to identify the appropriate transdermal rivastigmine dose for this population.
The authors attest that their institution does not require Institutional Review Board approval. Patient consent has been obtained and filed for the publication of this case report.
Address for Correspondence: C. James Watson, MD, Maine Medical Center, Department of Emergency Medicine, 22 Bramhall Street, Portland, Maine 04102. Email: james.watson@ mainehealth.org
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. Support for author CJW was provided by the National Center for Advancing Translational Sciences, National Institutes of Health, Grant Number K12TR004384. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Copyright: © 2025 Watson et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
Antimuscarinic Toxicity Safely Managed with High-Dose Transdermal Rivastigmine: A Case Report Watson et al.
1. Arens AM, Shah K, Al-Abri S, et al. Safety and effectiveness of physostigmine: a 10-year retrospective review. Clin Toxicol 2018;56(2):101-7.
2. Boley SP, Olives TD, Bangh SA, et al. Physostigmine is superior to non-antidote therapy in the management of antimuscarinic delirium: a prospective study from a regional poison center. Clin Toxicol 2019;57(1):50-55.
3. Whitledge JD, Soto P, Glowacki KM, et al. Shortages of agents used to treat antimuscarinic delirium. Am J Emerg Med. 2023;67:163-7.
4. Temporary importation of Anticholium® (physostigmine salicylate) ampules with foreign, non-U.S. labeling to address supply shortage. Published online September 2023. Accessed May 1, 2025. https:// www.fda.gov/media/173483/download?attachment%20or%20https:// www.fda.gov/drugs/drug-shortages/october-31-2023-temporaryimportation-available-physostigmine-injection.
5. Fratta KA, Ginder M, Haggerty DA. Oral and transdermal rivastigmine for the treatment of anticholinergic delirium: a case report. J Emerg Med. 2023;65(4):e366-e368.
6. Whitledge JD, Watson CJ, Simpson MD, et al. Diphenhydramineinduced antimuscarinic delirium treated with physostigmine and transdermal rivastigmine. J Med Toxicol. 2023;19(2):219-223.
7. Yakey B, Vohra V, Martin A, et al. Treatment of pediatric antimuscarinic delirium with oral rivastigmine. Oxf Med Case Reports 2023;2023(9):330-2.
8. Greene SC. Rivastigmine use in the treatment of antimuscarinic delirium. J Med Toxicol. 2023;19(3):284-287.
9. Chiew AL, Holford AG, Chan BSH, et al. Rivastigmine for the management of anticholinergic delirium. Clin Toxicol. 2024;62(2):82-7.
10. EXELON® PATCH (Rivastigmine Transdermal System) [Package Insert]. Novartis Pharmaceuticals Corporation; 2018. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/ label/2007/022083lbl.pdf
11. Hartvig P, Wiklund L, Lindström B. Pharmacokinetics of physostigmine after intravenous, intramuscular and subcutaneous administration in surgical patients. Acta Anaesthesiol Scand 1986;30(2):177-182.
12. Dawson AH and Buckley NA. Pharmacological management of anticholinergic delirium ‐ theory, evidence and practice. Br J Clin Pharmacol. 2016;81(3):516-24.
13. Lefèvre G, Sędek G, Huang HA, et al. Pharmacokinetics of a rivastigmine transdermal patch formulation in healthy volunteers: relative effects of body site application. J Clin Pharmacol. 2007;47(4):471-8.
14. Lefèvre G, Sędek G, Jhee SS, et al. Pharmacokinetics and pharmacodynamics of the novel daily rivastigmine transdermal patch compared with twice-daily capsules in Alzheimer’s disease patients. Clin Pharmacol Ther. 2008;83(1):106-114.
15. Giacobini E, Spiegel R, Enz A, et al. Inhibition of acetyl- and butyryl-cholinesterase in the cerebrospinal fluid of patients with Alzheimer’s disease by rivastigmine: correlation with cognitive benefit. J Neural Transm. 2002;109(7-8):1053-1065.
16. Baselt R. Disposition of Toxic Drugs and Chemicals in Man. 12th ed. Seal Beach, CA: Biomedical Publications; 2020.
Matthew J. Christensen, MD Jennifer Foti, DO
Section Editor: Jacqueline Le, MD
Naval Medical Center San Diego, Department of Emergency Medicine, San Diego, California
Submission history: Submitted August 10, 2025; Revision received September 15, 2025; Accepted September 24, 2025
Electronically published January 14, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.50611
Introduction: Pectus excavatum (PEX) is the most common congenital chest wall deformity, characterized by posterior depression of the sternum and lower costal margin. While often asymptomatic, severe PEX can lead to compression of the heart and great vessels, potentially causing right ventricular dysfunction, syncope, and other cardiovascular symptoms. Syncope due to right ventricle compression in PEX is rare but can significantly impact quality of life and may require surgical intervention.
Case Report: An 18-year-old female presented to the emergency department after an unwitnessed syncopal episode. The patient reported feeling lightheaded while showering, followed by collapse and brief loss of consciousness. Diagnostic testing revealed normal neurological and metabolic parameters including point-of-care glucose, electrocardiogram, serum troponin, electrolytes, and head computed tomography. Chest imaging showed severe PEX with concerns of right ventricular compression. Transthoracic echocardiography demonstrated normal cardiac function, and exercise stress testing showed no ischemic changes. Additional laboratory studies revealed iron deficiency anemia.
Conclusion: This case underscores the potential for PEX to cause distorted cardiac morphology, including right ventricular compression, which can lead to syncope in severe cases. The absence of cardiac ischemia, arrhythmias, or metabolic derangement suggests postural changes compounded by undiagnosed anemia and underlying PEX as the most likely cause of this patient’s syncope. Given the patient’s symptoms and anatomical findings, referral for surgical evaluation was made to discuss definitive management options. This case highlights the importance of considering structural chest wall abnormalities in the differential diagnosis of syncopal events, particularly when standard causes are excluded. [Clin Pract Cases Emerg Med. 2025;10(1):81-84.]
Keywords: syncope; pectus excavatum; right ventricle compression; case report.
Pectus excavatum (PEX) is a congenital chest wall deformity characterized by posterior depression of the sternum and lower costal margin.1 This condition is the most common structural anomaly of the anterior chest, occurring in approximately 1 in 300-400 live births.1 Females with PEX typically experience more severe chest wall deformity than males, although this does not lead to worse exercise intolerance or overall cardiopulmonary function.2 In severe cases, the chest wall deformity can lead to physical
compression of mediastinal structures including the heart and great vessels, which has been implicated in cardiovascular pathology including ventricular arrhythmias,3,4 palpitations,5 right ventricle dysfunction,6,7,8 and sudden cardiac arrest.9,10 While PEX does not cause left ventricle dysfunction,11 the most notable hemodynamic impact of the resultant distorted right ventricle morphology is the potential for clinically significant ventricular compression, which can predispose to syncope or near-syncopal events.12,13 In severe cases of PEX, the posterior displacement of the sternum can compress the
right ventricle and inferior vena cava, which reduces venous return and impairs right ventricular filling.14,15 This functionally decreases cardiac output, particularly during physical exertion or in postural changes, when the heart’s ability to adapt to changes in intrathoracic pressure becomes strained. This impaired cardiac output may cause a drop in cerebral perfusion, leading to syncope or near-syncopal episodes. Although syncope in PEX patients is less common than other symptoms such as exercise intolerance, chest pain, or dysrhythmias, it is an important clinical manifestation that can significantly impact quality of life and may indicate a need for surgical intervention.
An 18-year-old female with no prior medical history was brought by ambulance to the emergency department after an unwitnessed syncopal event that occurred at her home. The patient reported that she was in the shower, felt lightheaded, stepped out of the shower, and then collapsed into the bathroom counter. Her roommate heard the noise and discovered her on the floor, with loss of consciousness lasting approximately one minute. No convulsive activity or post-ictal state was witnessed. Physical exam revealed no focal neurologic deficits or sequelae of head trauma, although there was mild tenderness to palpation of the lateral left chest wall. Point-of-care blood glucose was normal. Electrocardiogram showed no dysrhythmias and no ischemic changes. Serum troponin-I level was undetectable.
Complete metabolic panel demonstrated normal kidney function and normal liver function, and was without electrolyte derangement. Complete blood count revealed a stable microcytic anemia (hemoglobin 8.9 grams per deciliter (g/dL) (reference range: 12.0-16.0 g/dL) and hematocrit 26.7% (36%-46%) (compared to hemoglobin 8.7 g/dL and hematocrit 26% measured seven months prior) without leukocytosis, leukopenia, or thrombocytopenia. Urine pregnancy testing was negative. Non-contrast computed tomography (CT) of the head was obtained due to the traumatic mechanism and reported headache, which showed no intracranial pathology.
A two-view chest radiograph was obtained due to the reported left-sided rib pain with tenderness on exam, which showed no apparent rib fractures, pulmonary contusion, or pneumothorax, but did reveal severe PEX with concern for right ventricular compression (Image 1). Contrast-enhanced CT of the chest was ordered for further characterization, which demonstrated significantly reduced anterior-posterior diameter of the chest and right sternal torsion with resultant compression of the right ventricle (Images 2 and 3). The patient was admitted to the cardiology service for additional diagnostic testing, telemetry monitoring, and risk stratification.
Transthoracic echocardiogram revealed left ventricular ejection fraction of 60% with normal right ventricle and left ventricle function and no evidence of right ventricular
CPC-EM Capsule
What do we already know about this clinical entity?
Pectus excavatum is the most common congenital chest wall deformity, and has been implicated in cardiovascular pathology including ventricular arrhythmias and cardiac arrest.
What makes this presentation of disease reportable?
This is the first published case report of syncope caused by right ventricular compression from severe pectus excavatum.
What is the major learning point?
Consider atypical etiologies such as cardiac compression from structural chest wall deformity in patients with syncope when standard causes have been excluded.
How might this improve emergency medicine practice?
Broadening our differential diagnoses for common emergency department presentations will improve the care we provide to our patients.
collapse, right atrial dilation, or elevated pulmonary artery pressure. Exercise stress testing showed no ischemic changes during exercise or recovery. Continuous telemetry revealed no paroxysmal dysrhythmias during her hospital course. Iron studies revealed iron deficiency as the likely cause of the identified microcytic anemia. The patient was ultimately discharged after a three-day hospitalization with outpatient referral to cardiothoracic surgery to discuss surgical and nonsurgical management options.
Several clinical learning points are demonstrated throughout this case. It highlights the importance of maintaining a broad differential diagnosis in syncope, the impact of PEX on cardiac morphology, and the potential compounding effect of an underlying microcytic anemia in PEX that may predispose to syncopal events even in the absence of overt cardiac dysfunction.
Syncope, defined as a transient loss of consciousness due to insufficient cerebral perfusion,1 requires a comprehensive workup to identify the underlying cause. Although syncope or near-syncopal episodes in young and otherwise healthy
3. Contrast-enhanced chest computed tomography (axial plane) demonstrating right ventricular compression from severe pectus excavatum (black arrows). Thoracic cavity measurements are included in red to highlight the abnormal anterior-posterior diameter of this patient’s chest wall.
patients are often the result of benign vasovagal or orthostatic etiologies, this case emphasizes the importance of considering a broad differential diagnosis even in the healthy population. This patient’s normal physical exam, normal point-of-care glucose, unremarkable electrocardiogram, normal serum troponin-I, and unremarkable head CT all lowered the likelihood of classic emergent causes of syncope such as hypoglycemia, dysrhythmias, cardiac ischemia, or intracranial hemorrhage. Syncopal events may also be provoked by anemia or impaired cardiac function. This patient’s radiographic evidence of severe PEX with right ventricle compression along with laboratory evidence of chronic microcytic anemia likely had compounding effects that contributed to her syncope.
Severe PEX may cause physical compression of critical cardiac structures, particularly the right ventricle. 6,7,8 This case demonstrates how imaging studies such as a chest radiograph or contrast-enhanced chest CT can reveal underlying structural abnormalities in the appropriate clinical scenario, which might otherwise be overlooked. Resultant right ventricle compression may impede normal blood flow and cause syncope, especially during activities that decrease venous return. This patient’s syncopal event occurred in the shower, potentially due to postural changes or increased intrathoracic pressure, and was likely compounded by the underlying chronic microcytic anemia.
Another key element in this case is the identification of microcytic anemia. The patient’s stable hemoglobin (8.9 g/dL) and hematocrit (26.7%) compared to values from seven months prior (hemoglobin 8.7 g/dL and hematocrit 26%), as well as the new diagnosis of iron deficiency anemia based on inpatient iron studies, suggest that a previously undiagnosed
chronic anemia likely contributed to her syncope. Acute blood- loss anemia is less likely in this patient without reported menorrhagia and without melena or hematochezia to suggest occult gastrointestinal bleed. Anemia can impair oxygen delivery, especially in the context of structural cardiac abnormalities such as PEX, which further reduces cardiac output and may exacerbate symptoms. This case underscores the importance of considering anemia as a contributing factor in patients with unexplained syncope, and the value of routine laboratory tests such as a complete blood count and iron studies when indicated.
The patient’s management involved careful risk stratification including cardiac imaging, echocardiography, and exercise stress testing, which revealed no major functional abnormalities or ischemia. Given the absence of acute cardiac compromise, she was discharged with outpatient follow-up for cardiothoracic surgical consultation, which may alleviate the structural compression and mitigate the risk of future syncopal episodes. This case demonstrates the importance of multidisciplinary management, as the etiology of her iron deficiency anemia will require additional outpatient workup and the decision between surgical and nonsurgical management of PEX will require input from both cardiology and cardiothoracic surgery specialists.
This case demonstrates the importance of a broad differential diagnosis and thorough workup in patients presenting for syncope. Chest wall deformities such as pectus excavatum may lead to cardiovascular compromise, and a comprehensive approach that considers both structural and non-structural causes of syncope is essential.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Address for Correspondence: Matthew J. Christensen, MD, Naval Medical Center San Diego, Department of Emergency Medicine, 34800 Bob Wilson Drive, San Diego, CA 92134 Email: matthew.j.christensen11.mil@health.mil.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Christensen et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http:// creativecommons.org/licenses/by/4.0/
1. David VL. Current concepts in the etiology and pathogenesis of pectus excavatum in humans-A systematic review. J Clin Med 2022;11(5):1241.
2. Casar Berazaluce AM, Jenkins TM, Garrison AP, et al. The chest wall gender divide: females have better cardiopulmonary function and exercise tolerance despite worse deformity in pectus excavatum. Pediatr Surg Int. 2020;36(11):1281-1286.
3. Chan Wah Hak Y-S, Lim Y-P, Liew R, et al. Pectus excavatum: uncommon electrical abnormalities caused by extrinsic right ventricular compression. J Cardiovasc Electrophysiol 2014;25(3):324-327.
4. Ferraz L, Carvalho D, Carvalho S, et al. Pectus excavatum with right ventricular compression-induced ventricular arrhythmias. Br J Cardiol. 2023;30(1):4.
5. Sandhu ZA, Shaikh S, Buckley A. An unusual case of palpitations: pectus excavatum. Clin Med Rev Case Rep. 2019;6(10):288-289.
6. Park S-Y, Park T-H, Kim J-H, et al. A case of right ventricular dysfunction caused by pectus excavatum. J Cardiovasc Ultrasound 2010;18(2):62-65.
7. Underwood K, Vorsanger M, Saric M, et al. Positional right ventricular obstruction in pectus excavatum. Am J Cardiol 2017;119(8):1288-1289.
8. Carroll MK, Powell AW, Hardie WD, et al. Pectus excavatum: the effect of tricuspid valve compression on cardiac function. Pediatr Radiol. 2024;54(9):1462-1472.
9. Rachwan RJ, Purpura AK, Kahwash BM. Sudden cardiac arrest in a young patient with severe pectus excavatum. Am J Med Sci 2018;356(6):570-573.
10. Moossdorff M, Maesen B, den Uijl DW, et al. Case report: ventricular fibrillation and cardiac arrest provoked by forward bending in adolescent with severe pectus excavatum. Eur Heart J Case Rep 2021;5(10):1-6.
11. Saleh RS, Finn JP, Fenchel M, et al. Cardiovascular magnetic resonance in patients with pectus excavatum compared with normal controls. J Cardiovasc Magn Reson. 2010;12:73.
12. Borrhomée S, Lenoir M, Gouton M, et al. Syncope caused by right ventricular obstruction by pectus excavatum. J Thorac Cardiovasc Surg. 2016;151(4):e67-9.
13. De Feria AE, Bajaj NS, Polk DM, et al. Pectus excavatum and right ventricular compression in a young athlete with syncope. Am J Med 2018;131(11):e451-e453.
14. Rodriguez-Granillo GA, Raggio IM, Deviggiano A, et al. Impact of pectus excavatum on cardiac morphology and function according to the site of maximum compression: effect of physical exertion and respiratory cycle. Eur Heart J Cardiovasc Imaging. 2020;21(1):77-84.
15. White JA, Fine NM, Shargall Y. Pectus excavatum with compression of the inferior vena cava: a rare cause of recurrent syncope. Circulation. 2009;120(17):1722.
Rochelle Kofman, BS* Ryan Allen, BA*
Addison B. Smartt, BS*
Kevin M. Drechsel, MD†
Jordan R. Pollock., MD, MBA*‡
Douglas Rappaport, MD*†
Section Editor: Grace Hickam, MD
Mayo Clinic Alix School of Medicine, Phoenix, Arizona
† ‡
Mayo Clinic Arizona, Department of Emergency Medicine, Phoenix, Arizona
Mayo Clinic Arizona, Department of Radiology, Phoenix, Arizona
Submission history: Submitted August 13, 2025; Revision received October 8, 2025; Accepted October 24, 2025
Electronically published January 14, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.50643
Introduction: Sternoclavicular joint injuries are rare and potentially life-threatening injuries due to their proximity to vital mediastinal structures. In adolescents, skeletal immaturity can add complexity to the injury due to potential involvement of the physis. A physeal fracture with displacement can appear as a dislocation on imaging, also known as pseudo-dislocation. Additionally, this anatomic area is difficult to visualize with plain radiographs, which can result in misdiagnosis and delayed treatment.
Case Report: We present a case of a 15-year-old male athlete who presented to the emergency department with severe right clavicular pain four hours after sustaining a football injury. Plain radiographs obtained at an outside facility as well as repeat plain radiographs at our facility showed no evidence of fracture or dislocation. The patient’s degree of pain and physical exam findings prompted further imaging with computed tomography (CT), ultimately revealing a physeal fracture of the medial right clavicle with posterior and superior displacement.
Conclusion: Sternoclavicular joint injuries in skeletally immature patients are complex and require immediate diagnosis and intervention. Plain radiographs are often unreliable in recognizing these injuries and, in our case, the physeal fracture with displacement was not radiographically apparent on two separate occasions. Advanced imaging with CT revealed the diagnosis, highlighting the importance of a detailed physical exam and for physicians to maintain a high index of clinical suspicion when evaluating adolescents with high-impact trauma, even in the setting of negative plain radiographs. [Clin Pract Cases Emerg Med. 2025;10(1):85-88.]
Keywords: sternoclavicular joint; pediatric sports injury; physeal fracture; case report.
Sternoclavicular joint (SCJ) injuries are relatively uncommon, constituting less than 1% of all clavicular injuries.1 In adolescents as many as 75% of SCJ injuries are posterior, which can be potentially serious or even lifethreatening due to damage to vital underlying structures including the trachea, esophagus, and great vessels.1, 2 Computed tomography (CT) is the imaging modality of choice
for injuries of this nature, given poor visualization of mediastinal structures with plain radiographs, as well as poor sensitivity of plain radiographs for detecting SCJ fracture or dislocation.3-5 While CT and magnetic resonance imaging (MRI) have higher diagnostic accuracy and sensitivity evaluating for SCJ injury, clinicians may not be aware of the need for advanced imaging in the setting of a negative plain radiographs for this injury.6-8
In pediatric populations still undergoing skeletal development, the incidence and categorization of this type of injury are often unclear. Typically, these injuries, described as either SCJ dislocation or medial clavicular fracture, occur during contact sports.8, 9 Some studies suggest that fractures are far more common than dislocation in the setting of posterior SCJ injury, although one retrospective review of 48 adolescent patients suggests the two have prevalence.8 Cases of SCJ dislocation with accompanying fracture are exceedingly rare, although some are described in the literature and require advanced imaging such as CT or MRI to properly diagnose.10, 11
Here, we present a case of medial clavicle physeal fracture with posterior and superior displacement in a 15-yearold patient after a football injury requiring open reduction and internal fixation.
A 15-year-old male presented to the emergency department (ED) with right clavicular pain approximately four hours following a football injury. The patient described the event as falling after jumping for a ball, with another player falling on top of him. He broke the fall with his right elbow, which resulted in immediate pain in the right clavicular area. Initial plain radiographs at an outside facility did not reveal any fractures. However, further care was pursued due to the severity of his pain.
Upon arrival to the ED, vitals were within normal limits. The patient described the pain as 8/10 in severity and localized to the right clavicle, with significant tenderness and mild right anterior neck pain. On physical examination, there was tenderness and a palpable deformity over the right mid-toproximal clavicle. A depressive deformity was noted at the SCJ. No crepitus or subcutaneous emphysema was noted. The right radial pulse was intact, and grip strength was preserved. There was no tenderness over the right acromioclavicular joint or anterior shoulder. No midline cervical spine tenderness or tracheal deviation was appreciated. Pulmonary exam showed normal breath sounds, and the patient was in no acute respiratory distress.
Consistent with the radiographs obtained earlier, clavicle radiographs obtained in the ED again showed no sign of fracture or dislocation, with reports of normal acromioclavicular and coracoclavicular intervals and unremarkable appearance of acromioclavicular and glenohumeral joints. Further investigation with chest CT was pursued due to persistent pain and concern for deeper injury, which revealed a right SCJ dissociation with approximately 1.4 cm of posterior displacement of the medial clavicle, along with a possible small adjacent avulsion fragment suspicious for physeal fracture (Images 1-2). A small hematoma was noted anteroinferior to the right clavicular head, without evidence of pneumothorax, pulmonary contusion, mediastinal hematoma, or vascular injury.
CPC-EM Capsule
What do we already know about this clinical entity?
Posterior sternoclavicular joint (SCJ) injuries are rare, often subtle on imaging, and can be life-threatening due to proximity to vital mediastinal structures.
What makes this presentation of disease reportable?
A physeal fracture with posterior SCJ displacement in an adolescent, which was missed on initial plain radiograph, was confirmed with computed tomography (CT) due to physician’s clinical suspicion.
What is the major learning point?
High clinical suspicion is critical; CT is the imaging modality of choice, even in the setting of negative plain radiographs in suspected SCJ trauma.
How might this improve emergency medicine practice?
Earlier CT use in the emergency department and orthopedic consult for at-risk mechanisms could improve outcomes in pediatric SCJ trauma cases.
Orthopedic and thoracic surgery teams were consulted and ultimately recommended the patient be evaluated at a pediatric center. After discussion with the accepting tertiarycare pediatric facility, the patient was placed in a shoulder immobilizer and determined to be stable for transfer by private vehicle. Upon arrival to the pediatric ED, the evaluating pediatric orthopedic team proceeded with immediate surgical intervention, including open reduction and internal fixation of the SCJ injury. Physeal fracture with posterior superior displacement of the clavicle was confirmed intraoperatively. The operative course was uncomplicated, and he tolerated the procedure and anesthesia well. He was discharged the same day in a soft sling for immobilization, with plans to follow up and begin physical therapy six weeks postoperatively. A full recovery was anticipated after three months.
Posterior sternoclavicular joint displacements are rare injuries that carry a significant clinical risk due to the proximity to vital mediastinal structures such as subclavian and brachiocephalic vessels, trachea, and esophagus. These
Image 1A and 1B. Superior displacement of the right medial clavicle relative to the sternum shown by arrow one on coronal computed tomography (CT) without contrast (A) and three-dimensional CT reconstruction (B). Relative to the displaced clavicle, the epiphyseal ossification is in the expected anatomic location, suggesting fracture of the physis with displacement of the ossified clavicle, shown by arrow two. The unaffected left clavicle is anatomically aligned, shown by arrow three.
2. Axial computed tomography without contrast showing posterior displacement of the right medial clavicle relative to the left clavicle, noted by arrow.
are all at risk of compression or damage in this type of injury.1,2 Posterior SCJ injuries carry a higher risk for lifethreatening complications such as vascular injury and airway injury.12,13 The critical nature of these complications makes prompt diagnosis of this injury essential and management by multidisciplinary care teams necessary to prevent potentially fatal complications.
Initial imaging with plain radiographs is often insufficient in identifying posterior SCJ injuries because of the joint’s deep anatomic location and overlapping of the clavicle with thoracic structures.5 A high index of clinical suspicion for SCJ injury should prompt further advanced imaging even in the setting of negative plain radiographs. Computed tomography is the gold standard imaging modality for SCJ injuries as it allows for clear delineation of joint alignment, physeal involvement, and clear views of mediastinal structures.5,13
Despite this, the diagnosis is sometimes still missed or delayed in the setting of negative plain radiographs, with missed diagnosis rates up to 23-25% in pediatric cohorts. This may be due to physicians’ lack of awareness of the limitation of plain radiographs for SCJ injuries and lack of standardized protocols for such injuries.8, 14 Retrospective reviews have shown that CT is often delayed until after orthopedic consultation, rather than being obtained at the initial presentation, which further contributes to diagnostic delays.15 The early use of CT in our patient confirmed the diagnosis, ensured the proper specialists were involved, and facilitated an effective and safe transfer of the patient to a tertiary-care pediatric center.
Skeletal immaturity added complexity to this patient’s injury and, in general, makes interpreting SCJ injuries in the pediatric population more challenging. The medial clavicular
physis typically fuses between 22-25 years of age; therefore, it may be difficult to radiographically distinguish physeal fractures from dislocations in adolescents.8 In this patient, CT revealed a small bony fragment adjacent to the displaced clavicle, which suggested physeal involvement. Differentiating between physeal fractures and joint dislocations is crucial, as their treatment and management are vastly different, particularly in children.9,10 However, sometimes this distinction cannot be made even with advanced imaging, and the diagnosis must be confirmed intraoperatively. In this patient, surgery confirmed the presence of a physeal fracture with posterior displacement.
The mechanism that led to this patient’s injury is classic for a SCJ injury with displacement. Axial loading through a fall onto the elbow aligns with the literature that describes the most common etiologies of this injury.9,14 Contact sports such as football and wrestling are the leading sources of pediatric SCJ trauma.6,8 In young athletes presenting with clavicle pain following high-energy impacts, physicians should carefully examine the SCJ, even in the setting of negative initial plain radiographs and the absence of obvious vascular or airway compromise.
Management of suspected posterior SCJ displacements or physeal fractures with posterior displacement requires a multidisciplinary approach due to its complex nature and risk profile. Thoracic surgery should be consulted due to the risk of mediastinal injuries, as well as orthopedic surgery. Closed reduction is preferred within 48 hours of injury and should be performed with cardiothoracic or vascular surgery backup. Open reduction is indicated if closed reduction fails or is
Medial Clavicle Physeal Fracture in a 15-Year-Old Male: A Case Report
delayed. While non-operative management may be considered if alignment is preserved and there are no complications, most displaced injuries require surgical stabilization.2, 9, 14 In our case, consultation with a pediatric care team was needed due to the skeletally immature nature of this patient, and prompt transfer to a pediatric center was safely coordinated. This case contributes to the growing literature on pediatric SCJ injuries. While similar cases of dislocation-fractures and pseudo-dislocations have been reported, this remains a rare injury and, importantly, highlights the significance of the emergency physician’s clinical suspicion to pursue further imaging, despite negative initial films. This case reinforces the key point that a negative initial plain radiograph does not rule out SCJ injury. Clinical suspicion and awareness of these injuries must prompt further investigation with more advanced imaging early in the care timeline to prevent delays in diagnosis and treatment.
This was a rare case of a posterior sternoclavicular joint displacement with physeal involvement in an adolescent football player; a diagnosis originally missed on initial plain radiographs. Computed tomography is often underused in emergency settings for SCJ injuries, despite the known limitations of plain radiographs in identifying posterior displacement, and the diagnosis is often missed or delayed. This case reinforces the need for early advanced imaging, even in the setting of negative plain radiographs, for prompt diagnosis and treatment of pediatric SCJ injuries.
The authors attest that their institution does not require Institutional Review Board approval. Patient consent has been obtained and filed for the publication of this case report.
Address for Correspondence: Douglas Rappaport, MD, Mayo Clinic Arizona, Department of Emergency Medicine, 5777 E Mayo Blvd, Phoenix AZ 85054. Email: Rappaport.Douglas@mayo.edu
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Kofman et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Kraus R, Zwingmann J, Jablonski M, Bakir MS. Dislocations of the acromioclavicular and sternoclavicular joint in children and adolescents: a retrospective clinical study and big data analysis of routine data. PLoS One. 2020;15(12):e0244209.
2. Groh GI and Wirth MA. Management of traumatic sternoclavicular joint injuries. J Am Acad Orthop Surg. 2011;19(1):1-7.
3. Morell DJ and Thyagarajan DS. Sternoclavicular joint dislocation and its management: a review of the literature. World J Orthop. 2016;7(4):244-250.
4. Jacob M, Snashall J, Dorfman A, Shesser R. X-ray-negative posterior sternoclavicular dislocation after minor trauma. Am J Emerg Med. 2013;31(1):260.e3-260.e5.
5. Thomas DP, Davies A, Hoddinott HC. Posterior sternoclavicular dislocations—a diagnosis easily missed. Ann R Coll Surg Engl. 1999;81(3):201-204.
6. Hellwinkel JE, McCarty EC, Khodaee M. Sports-related sternoclavicular joint injuries. Phys Sportsmed. 2019;47(3):253-261.
7. Laur O, Ha AS, Bartolotta RJ, et al. ACR Appropriateness Criteria® acute shoulder pain: 2024 update. J Am Coll Radiol. 2025;22(5S):S36–S47.
8. Lee JT, Nasreddine AY, Black EM, et al. Posterior sternoclavicular joint injuries in skeletally immature patients. J Pediatr Orthop. 2014;34(4):369-375.
9. Chaudhry S. Pediatric posterior sternoclavicular joint injuries. J Am Acad Orthop Surg. 2015;23(8):468-475.
10. Vitali M, Drossinos A, Pironti P, et al. The management of SalterHarris type II fracture with associated posterior sternoclavicular joint displacement using a locking compression plate: a 14-year-old adolescent’s case report. Medicine (Baltimore). 2019;98(51):e18433.
11. Beckmann N and Crawford L. Posterior sternoclavicular Salter-Harris fracture-dislocation in a patient with unossified medial clavicle epiphysis. Skeletal Radiol. 2016;45(8):1123-1127.
12. Marcus MS and Tan V. Cerebrovascular accident in a 19-year-old patient: a case report of posterior sternoclavicular dislocation. J Shoulder Elbow Surg. 2011;20(7):e1-e4.
13. Groh GI, Wirth MA, Rockwood CA Jr. Treatment of traumatic posterior sternoclavicular dislocations. J Shoulder Elbow Surg. 2011;20(1):107-113.
14. Tepolt F, Carry PM, Taylor M, Hadley-Miller N. Posterior sternoclavicular joint injuries in skeletally immature patients. Orthopedics. 2014;37(2):e174-e181.
15. Tepolt F, Carry PM, Heyn PC, Miller NH. Posterior sternoclavicular joint injuries in the adolescent population: a meta-analysis. Am J Sports Med. 2014;42(10):2517-2524.
Faith Ibu, MBBS* Olivia Keller-Baruch, MD† Jessica Pelletier, DO‡
Section Editor: Joel Moll
, MD
Mayo Clinic, Department of Emergency Medicine, Rochester, Minnesota Ichilov Medical Center, Department of Emergency Medicine, Tel-Aviv, Israel University of Missouri-Columbia, Department of Emergency Medicine, Columbia, Missouri
Submission history: Submitted July 19, 2025; Revision received August 29, 2025; Accepted October 2, 2025
Electronically published January 20, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.48896
Introduction: Carotidynia is a rare, often under-diagnosed condition characterized by idiopathic inflammation around the carotid artery, presenting with unilateral neck pain that typically resolves within two weeks.
Case Report: A 32-year-old male presented with intermittent right anterior neck pain for two years, with no other associated symptoms. Computed tomography revealed carotid perivascular inflammation, consistent with carotidynia.
Conclusion: Although self-limited, carotidynia should be considered in patients with unexplained neck pain, as its recognition is crucial to differentiate it from more serious conditions and to avoid mismanagement or unnecessary interventions. [Clin Pract Cases Emerg Med. 2025;10(1):89-92.]
Keywords: carotidynia; neck pain; carotid inflammation; case report.
INTRODUCTION
Carotidynia first emerged as a medical term in the late 20th century and has since been attributed varying definitions and diagnostic criteria, leading to confusion and controversy within the medical community.1 In 1988, carotidynia was categorized by the International Classification of Headache Disorders as an atypical headache syndrome separate from idiopathic neck pain2; the diagnosis required unilateral neck pain, focal carotid tenderness, absence of structural lesions, and spontaneous resolution within 14 days for diagnosis.3 However, in 2004, carotidynia was excluded from the classification system due to concerns over diagnostic validity, and it was redefined simply as a nonspecific symptom of other diseases.4 More recently, however, there has been recognition of characteristic radiologic findings that favor carotidynia as a distinct entity, now more commonly referred to as transient perivascular inflammation of the carotid artery (TIPIC) syndrome.3,5 While it remains a rare disorder, reported cases suggest a slight female predominance, with peak incidence
in the fifth and sixth decades of life.5 The disease is thought to be driven by an underlying transient inflammatory process of the carotid adventitia, although its exact pathogenesis remains debated.
A 32-year-old male presented to the emergency department (ED) with acute exacerbation of chronic right anterior neck pain. He reported that the pain had been intermittent for approximately two years. He reported severe pain in the same area, lateral to the hyoid bone on the right. He denied fevers or infectious symptoms around the onset of the pain or during any recurrences. He reported exacerbation of pain with leaning forward and sometimes lying flat to sleep, with no change with swallowing. He denied any systemic symptoms such as fever, dysphagia, trismus, neurologic, respiratory, or gastrointestinal complaints. He had taken acetaminophen and ibuprofen with only temporary relief. The current flare-up began five days prior and had not improved despite taking acetaminophen, ibuprofen, amoxicillin, and
acetaminophen-hydrocodone, which he had available at home. He was seen in the ED the previous day, diagnosed with suspected hyoid-related musculoskeletal pain, and discharged after declining osteopathic manipulation and additional analgesics. His pain had worsened, prompting his second ED visit. In the ED, vitals were within normal limits, and he was overall well-appearing. Physical examination revealed no swelling or asymmetry, marked tenderness to palpation over the right anterior neck around the area overlying the common carotid bifurcation. He had no abnormal neurological findings. There was no pharyngeal erythema or swelling, or evidence of dental infection, and the physical examination was otherwise non-contributory.
Lab workup was within normal limits, including complete blood count and inflammatory markers, making an acute infectious process less likely. A computed tomography scan of the soft tissues of the neck with intravenous contrast demonstrated circumferential inflammation of the right carotid and extracranial internal carotid arteries (Images 1 and 2), correlating with the patient’s region of tenderness.
Given the patient’s clinical presentation in concordance with these characteristic imaging findings, a diagnosis of carotidynia was made. The patient was counseled on the
CPC-EM Capsule
What do we already know about this clinical entity?
Carotidynia is a rare, self-limiting condition characterized by idiopathic inflammation around the carotid artery that more often occurs in middle-aged women.
What makes this presentation of disease reportable?
We present the case of a young male diagnosed with this condition who was successfully treated with Nonsteroidal Anti-Inflammatory Drugs.
What is the major learning point?
While the exact etiology of carotidynia remains obscure, it presents with distinct radiologic findings that allow for accurate diagnosis.
How might this improve emergency medicine practice?
Recognizing carotidynia enables the clinician to distinguish it from more serious pathologies and to prevent unnecessary interventions and potential repeat visits.
diagnosis and likely disease course and was treated with scheduled nonsteroidal anti-inflammatory drugs (NSAID) and a short course of steroids. A follow-up appointment was scheduled with vascular surgery. On follow-up about two months later, he reported complete resolution of symptoms. He had a CT angiography and a carotid ultrasound with resolution of prior findings. No additional interventions were provided, and the patient was counseled to follow up as needed for recurrent symptoms.
Carotidynia is a rare clinical entity with a reported prevalence of about 2.8% of patients presenting with acute neck pain in one study.5 Over the years, since its first description in 1927, there has been controversy over this diagnosis, further adding to its obscurity.1,3,5 Nonetheless, its characteristic radiographic findings suggest it to be a distinct clinical entity. As in this case, most patients report suddenonset, throbbing, unilateral neck pain that may radiate to the jaw, ear, or ipsilateral eye and may be exacerbated with swallowing or head movement. Patients may also report bilateral symptoms and recurrent, self-resolving episodes.5 On
physical examination, the cardinal finding is exquisite focal tenderness upon palpation of the carotid artery bifurcation.5
In most studies, no neurologic symptoms have been reported; however, Lecler et al reported varying transient neurological symptoms such as dizziness, vertical diplopia, facial nerve palsy, dysesthesia, and motor deficit in about 17% of patients (eight of 47).5 Differential diagnosis is critical in managing carotidynia due to its symptom overlap with other conditions; it involves considering the patient’s entire clinical picture, including history and symptom progression. Carotid artery dissection, for instance, presents with neck pain and
tenderness but often includes focal neurological symptoms, which are absent in carotidynia.6 Similarly, thyroiditis may cause neck pain but is accompanied by systemic symptoms and thyroid dysfunction, which can be distinguished through lab tests.7
Imaging proves invaluable in differentiating carotidynia or TIPIC syndrome from these conditions. On ultrasound (US), carotidynia presents with hypoechoic wall thickening at the region of tenderness along the carotid artery with associated mild luminal narrowing.3,5 Ultrasound can also detect flow disruption or stenosis, which is not expected in carotidynia.3 The perivascular thickening around the carotid artery that is characteristic of the condition can also be distinguished on both CT and magnetic resonance imaging (MRI).5 Additionally, these imaging modalities can aid in visualizing vascular integrity and ruling out structural anomalies. The exclusion of these other potential causes is essential to arrive at a diagnosis of carotidynia, guiding appropriate treatment and preventing unnecessary interventions.
Regardless of modality (US, CT, MRI), typical imaging features of carotidynia include eccentric thickening and enhancement of the wall of the distal common carotid artery, bulb and proximal internal carotid artery, mild luminal narrowing, and fat-stranding in surrounding tissues.5 Unfortunately, the sensitivity and specificity of imaging studies for diagnosing carotidynia have not been established in the literature, and there is no validated gold standard modality for diagnosis. It is important to note that, like the self-limiting nature of the symptoms associated with the disease, the imaging abnormalities are also transient in nature, further complicating the diagnostic process.5
In the past, antimigraine medications such as propranolol, tricyclic antidepressants, ergotamine, and methysergide were used for the treatment of carotidynia.8 However, this trend decreased once carotidynia was removed from the International Classification of Headache Disorders in 2004. Now, given that most cases self-resolve, treatment typically includes rest, reassurance, and NSAIDs or aspirin. Symptom resolution is expected within one to two weeks. Additional treatments like corticosteroids, low-dose benzodiazepines, and calcium channel blockers have shown some success in recurrent cases.9
In emergency medicine, the ability to rapidly identify and differentiate carotidynia from other potentially life-threatening conditions is paramount. Imaging modalities such as CT, ultrasound, and MRI can be used to visualize the integrity of the carotid artery and surrounding structures. While carotidynia is a self-limiting disease, other causes of unilateral neck pain that are more severe and may even be lifethreatening, such as carotid dissection, thyroiditis, giant cell arteritis, sialadenitis, or cervical arthrosis, must first be excluded. The role of imaging in carotidynia extends beyond
diagnosis, contributing to a deeper understanding of its inflammatory nature and helping guide clinical management to ensure timely and effective treatment.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Address for Correspondence: Faith Ibu, MBBS, Mayo Clinic, Department of Emergency Medicine, 200 1st Avenue SW, Rochester, MN 55905. Email: ibu.faith@mayo.edu
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2026 Ibu et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Fay T. Atypical neuralgia. Arch Neurol Psychiatry. 1927;18:309–315.
2. Headache Classification Subcommittee of the International Headache Society. Classification and diagnostic criteria for headache disorders, cranial neuralgias and facial pain. Cephalalgia. 1988;8(suppl 7):1-96.
3. Abrahamy M, Werner M, Gottlieb P, et al. Ultrasound for the diagnosis of carotidynia. J Ultrasound Med. 2017;36(12):2605-2609.
4. Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders. 2nd ed. Cephalalgia 2004;24:9–160.
5. Lecler A, Obadia M, Savatovsky J, et al. TIPIC syndrome: beyond the myth of carotidynia, a new distinct unclassified entity. AJNR Am J Neuroradiol. 2017;38(7):1391-1398.
6. Arnold M. Pain as the only symptom of cervical artery dissection. J Neurol Neurosurg Psychiatry. 2006;77(9):1021-1024.
7. Yuan J, Qi S, Zhang X, et al. Local symptoms of Hashimoto’s thyroiditis: a systematic review. Front Endocrinol (Lausanne). 2023;13:1076793.
8. Clark HV, King DE, Yow RN. Carotidynia. Am Fam Physician. 1994;50(5):987-990.
9. Stanbro MD, Gray BH, Kellicut DC. Carotidynia: revisiting an unfamiliar entity. Ann Vasc Surg. 2011;25(8):1144-1153.
Maggie Stark, DO, MS
Nicholas Valentini, MD
Section Editor: Steven Walsh, MD
Brown University, Department of Emergency Medicine, Providence, Rhode Island
Submission history: Submitted July 2, 2025; Revision received September 15, 2025; Accepted October 2, 2025
Electronically published January 21, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.48864
Introduction: Semaglutide, a glucagon-like peptide-1 receptor agonist, has gained increasing popularity for managing both type 2 diabetes mellitus and obesity. However, as its use increases, new adverse events are emerging. This case report presents a 70-year-old patient who developed pleural and pericardial effusions likely related to semaglutide use.
Case Report: Four weeks after being prescribed semaglutide, a 70-year-old woman presented to the emergency department (ED) with shortness of breath. Diagnostic testing in the ED and hospital revealed that she had both pericardial and exudative pleural effusions, along with a positive antinuclear antibody and elevated inflammatory markers. Her signs and symptoms improved with steroid administration, and no other etiology was identified.
Conclusion: The patient was diagnosed with drug-induced lupus, likely triggered by semaglutide. This case underscores the importance of recognizing and investigating uncommon drug-related complications. With the growing use of semaglutide, clinicians must remain vigilant for rare adverse effects such as pleural effusions to ensure prompt diagnosis and treatment. [Clin Pract Cases Emerg Med. 2025;10(1):93-96.]
Keywords: semaglutide; pleural effusions; drug-induced lupus; case report.
Obesity has become an increasing problem within the United States. Due to the significant comorbidities associated with obesity, pharmaceutical solutions have been in great demand. Semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, has gained increasing popularity for managing both type 2 diabetes mellitus and obesity. It has been shown to be effective for weight loss and improving cardiometabolic risk factors when compared to a placebo.1 It is known to most commonly cause gastrointestinal side effects1; however, as its use increases, new adverse events are emerging. In this case report we discuss a patient who developed pleural and pericardial effusions likely related to semaglutide use.
A 70-year-old woman with a past medical history significant for hypertension, hyperlipidemia, Raynaud phenomenon, and seasonal allergies presented to the
emergency department (ED) for shortness of breath and chest tightness. Her symptoms had progressively worsened over three weeks. She had started semaglutide six weeks prior for weight loss and began experiencing nausea, shortness of breath, and chest discomfort three weeks after initiation. She was advised to discontinue the medication in week four, but her symptoms persisted, prompting an ED visit.
On physical exam, her vitals were as follows: oxygen saturation, 95%; heart rate, 99 beats per minute; blood pressure, 127/63 millimeters of mercury; respiratory rate, 18 breaths per minute; and temperature, 36.7 °C. Physical exam was notable for pitting edema to the lower extremities, left greater than right, and coarse rales at the bilateral lung bases. A complete blood count, complete metabolic panel, brain natriuretic peptide (BNP), D-dimer, electrocardiogram, troponin assay, and chest radiograph (CXR) were ordered. Ultrasonography was performed including a lower extremity vascular ultrasound of the left lower extremity and a point-of-care ultrasound of the
heart and lungs. The vascular ultrasound showed no evidence of deep vein thrombosis in the lower extremities.
Her point-of-care echocardiography demonstrated a small, circumferential pericardial effusion without evidence of decreased ejection fraction and without right heart strain. Her CXR and point-of-care lung ultrasound demonstrated bilateral pleural effusions, more significant on the right (Images 1 and 2). Her D-dimer was elevated at 1,102 nanograms per milliliter (ng/mL) (reference range < 230 ng/mL). Subsequent computed tomography (CT) angiography of the chest did not demonstrate any pulmonary embolism; however, it did show moderate bilateral pleural effusions greater on the right (Image 3). The remainder of her lab testing was within normal limits.
Rheumatology was consulted with concern for an autoimmune process contributing to her pleural effusions. Rheumatology recommended that additional labs be collected. Pertinent results included an elevated C-reactive protein (CRP) at 110.9 milligrams per liter (mg/L) (< 10.0 mg/L) and an erythrocyte sedimentation rate at 66 mm/hour (0-30 mm/hour). She had a positive anti-nuclear antibody (ANA) with a speckled pattern and 1:160 titer (< 1:40) and anti-cyclic citrullinated peptide (anti-CCP) antibody level of 131 units (< 20 units). Additional autoimmune studies (anti-double stranded DNA, anti-histone, anti-Smith, anti-Ro antibody, anti-La antibody, complement 3, complement 4, and rheumatoid factor) were negative. The patient was then admitted to the hospital on the internal medicine service for further testing.
While admitted, the patient underwent a diagnostic thoracentesis with fluid studies suggestive of an exudative process. Her serum protein measured 6.3 grams (g)/dL (6.1-8.3 g/ dL) and her pleural fluid protein measured 4.2 g/dL. Her serum
CPC-EM Capsule
What do we already know about this clinical entity?
Semaglutide, a glucagon-like peptide-1 agonist, is effective for weight loss, but the spectrum of side effects is still being discovered.
What makes this presentation of disease reportable?
A patient who developed pleural and pericardial effusions after semaglutide use was diagnosed with drug-induced lupus despite negative antihistone antibodies.
What is the major learning point?
Semaglutide has a developing side-effect profile, including rarely seen pathology such as druginduced lupus. Point-of-care ultrasound can aid in rapid diagnosis.
How might this improve emergency medicine practice?
Awareness of atypical semaglutide side effects helps clinicians broaden differentials and identify serious complications early.
lactate dehydrogenase (LDH) measured 217 IU/L (119-265 IU/L) while her pleural fluid LDH measured 145 IU/L. Cytology of the pleural fluid showed mixed, non-specific inflammatory changes without evidence of malignancy. Culture of the pleural fluid including bacterial, fungal, and acid-fast bacilli did not produce any growth suggesting against an infectious etiology. Cardiac studies including a cardiology-based echocardiogram did not show any evidence of heart failure. Cardiology specialty consultation did not believe her pleural effusions were related to structural heart disease or heart failure. Infectious disease was also consulted and did not believe her pleural effusions originated from an infectious etiology.
Ultimately, following multispecialty discussions, the patient was placed on a prolonged oral steroid taper with a provisional diagnosis of drug-induced lupus despite negative anti-histone antibodies. At outpatient follow-up three months after hospitalization with rheumatology, the patient had marked improvement in symptoms with resolution of the pleural and pericardial effusions on follow-up imaging. She continued to do well from a clinical perspective.
Atraumatic pleural effusions may be caused by several different clinical entities such as heart failure, parapneumonic effusion, cirrhosis, infection, and pulmonary embolism (PE) with less common etiologies including malignancy, renal disease, and drug-induced and
autoimmune factors. In this case, a 70-year-old woman presented with symptoms of shortness of breath, chest tightness, and nausea, which were temporally associated with her initiation of semaglutide. The evaluation of pleural effusions, along with her clinical course and lab results, required a systematic approach to rule out various potential causes. She had no recent history of infection, antibiotic use, fever, travel, cough or associated symptoms, making an infectious cause of her pleural effusion less likely. Pleural fluid cultures did grow any pertinent organisms.
Given her age and risk factors, including hypertension, new-onset heart failure was initially considered high on the differential diagnosis, although this was not supported by subsequent point-of-care ultrasound and lab evaluation, namely a finding of a preserved ejection fraction and low BNP. A confirmatory cardiology-based echocardiogram performed later in the patient’s hospital course was also normal. Given the shortness of breath, PE and resultant reactive effusion was also considered. A D-dimer was elevated prompting a CT demonstrating the bilateral pleural effusions without evidence of PE.
The temporal relationship between the initiation of semaglutide and the onset of the patient’s symptoms raised suspicion for drug-induced lupus. Semaglutide has been associated with immune-mediated side effects, including lupus-like reactions.2,3 While the patient did not present with the classic symptoms of lupus, such as a butterfly-shaped rash or photosensitivity, the positive ANA and anti-CCP antibodies, along with a raised CRP, were indicative of an inflammatory process. By the Light criteria, the pleural fluid protein and LDH values have a 98% sensitivity and 83% specificity for exudative physiology.4 The exudative nature of the pleural effusions and the negative workup for other autoimmune diseases (eg, negative anti-dsDNA, anti-Smith antibodies) further supported this diagnosis. The patient’s improvement after stopping the offending medication, a prolonged steroid course, and supported by follow-up imaging and clinic visits reinforced the diagnosis.
Drug-induced lupus presents without anti-histone antibodies in approximately 25% of cases.5,6
There is emerging evidence that newer medications have a lower association with anti-histone antibody production despite the same clinical presentation.7 Semaglutide, a new medication widely prescribed for weight loss, has become increasingly popular. A 2022 meta-analysis reported a two-fold likelihood ratio of adverse events in patients taking semaglutide when compared to placebo.8 Patients were also 1.6 times more likely to experience severe adverse outcomes including death and prolonged hospitalization.7 Drug-induced lupus from semaglutide has been previously reported, although it was associated with a positive anti-histone antibody test.9 However, the patient presented with symptoms of acute liver failure and autoimmune hepatitis with intra-abdominal ascites rather than the primary thoracic pathology identified in this case report.9
Pleural and Pericardial Effusions Associated with Semaglutide: A Case Report Stark et al.
More study is needed to establish a definitive association between semaglutide and the autoimmune phenomena described. This case highlights the need to consider atypical causes of pleural effusion, especially in patients with recent medication changes, and to remain vigilant for rare adverse effects that can mimic more common pathologies.
Exudative pleural and pericardial effusions associated with semaglutide.
The authors attest that their institution does not require Institutional Review Board approval for publication of this case report. Patient consent has been obtained and filed for the publication of this case report.
Address for Correspondence: Maggie Stark, DO, MS, Brown University, Department of Emergency Medicine, 55 Claverick Street, Providence Rhode Island 02903. Email: mstark@ brownhealth.org.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2026 Stark et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Amaro A, Sugimoto D, Wharton S. Efficacy and safety of semaglutide for weight management: evidence from the STEP program. Postgrad Med. 2022;134(sup1):5-17.
2. Rivera FB, Arias-Aguirre E, Aguirre Z, et al. Evaluating the safety profile of semaglutide: an updated meta-analysis. Curr Med Res Opin. 2024;40(9):1495-1514.
3. Ruder K. As semaglutide’s popularity soars, rare but serious adverse effects are emerging. JAMA. 2023;330(22):2140-2142.
4. Light RW, Macgregor MI, Luchsinger PC, et al. Pleural effusions: the diagnostic separation of transudates and exudates. Ann Intern Med 1972;77(4):507-513.
5. Epstein A and Barland P. The diagnostic value of antihistone antibodies in drug-induced lupus erythematosus. Arthritis Rheum 1985;28(2):158-162.
6. Solhjoo M, Goyal A, Saliba F, et al. Drug-induced lupus erythematosus. In: StatPearls. Treasure Island (FL): StatPearls Publishing; April 3, 2023. Available at: https://www.ncbi.nlm.nih.gov/ books/NBK441889/. Accessed February 26, 2025.
7. Lee AYS. Clinical use of anti-histone antibodies in idiopathic and drug-induced lupus. Immunol Med. 2022;45(4):180-185.
8. Tan HC, Dampil OA, Marquez MM. Efficacy and safety of semaglutide for weight loss in obesity without diabetes: a systematic review and meta-analysis. J ASEAN Fed Endocr Soc. 2022;37(2):65-72.
9. Castellanos V, Workneh H, Malik A, et al. Semaglutide-induced lupus erythematosus with multiorgan involvement. Cureus 2024;16(3):e55324.
Larry Vernier, MD*
Griffin Barnes Gilmore, MD†
Kelsey Van Housen, MSPAS, PA-C‡
Xiao Chi Zhang, MD, MS§
Montefiore Einstein, Department of Emergency Medicine, The Bronx, New York
Mount Sinai Elmhurst, Department of Emergency Medicine, Queens, New York
Sidney Kimmel Medical College, Thomas Jefferson University, Philadelphia, Pennsylvania
Thomas Jefferson University, Department of Emergency Medicine, Philadelphia, Pennsylvania
Editor: R. Gentry Wilkerson, MD
Submission history: Submitted May 27, 2025; Revision received July 8, 2025; Accepted September 19, 202
Electronically published January 20, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem
DOI: 10.5811/cpcem.47931
Introduction: Guillain-Barré syndrome can manifest with progressive paralysis, requiring careful monitoring and treatment with steroids or intravenous (IV) immunoglobin. While this disease can be devastating and require intensive care unit level of care, there are few incidences of relapses.
Case Report: A 67-year-old man with a past medical history of Miller Fisher syndrome variant of Guillain-Barré syndrome, with complete recovery, presented to the emergency department (ED) with ataxia, ophthalmoplegia, dysphonia, and ambulatory dysfunction. The patient noticed his neurologic symptoms after waking up, and he presented to the ED with physical examination positive for difficulty with upward gaze, mild right lower facial droop, dysmetria with left finger to nose testing, and unsteady gait. A lumbar puncture revealed albuminocytologic dissociation (an elevated cerebrospinal fluid protein count without elevation in white blood cell count) and he was diagnosed with recurrent Miller Fisher syndrome. The patient completed a five-day course of IV immunoglobin with marked improvement of his symptoms. Unfortunately, the patient continued to have ambulatory difficulty, requiring inpatient rehabilitation with residual deficits including ophthalmoplegia and mild dysphonia.
Conclusion: Miller Fisher syndrome is an uncommon variant of Guillain-Barré syndrome with atypical neurologic findings that can lead to respiratory distress; it requires high levels of suspicion and diagnostic evaluation. This disease process can recur in a patient’s lifetime, contrary to what has been reported in the literature. [Clin Pract Cases Emerg Med. 2025;10(1):97-100.]
Keywords: Guillain-Barré syndrome; Miller Fisher syndrome; neurological emergencies; case report.
Miller Fisher syndrome is an uncommon variant of Guillain-Barré syndrome that can result in progressive paralysis and has been found to have relapses, contrary to common medical knowledge.1,2 Guillain-Barré syndrome is an autoimmune disease caused by an antibody attack of the myelinated sheath of peripheral nerves in the body resulting from molecular mimicry.3 The symptoms typically present as ascending lower motor nerve dysfunction starting in the lower extremities. Miller Fisher syndrome is a rare variant of Guillain-Barré syndrome that initially presents as cranial and facial nerve dysfunction causing symptoms of ophthalmoplegia, ataxia, and areflexia, before further progressive neurologic deterioration resulting in chest wall
weakness and severe respiratory distress.1,4 While most cases resolve within 8-12 weeks without recurrence when treated appropriately with intravenous (IV) immunoglobulin,3 recurrence of any variant of Guillain-Barré syndrome, including Miller Fisher syndrome, is rare.
A 67-year-old man with a past medical history of hypertension, Crohn colitis, basal cell carcinoma, and GuillainBarré syndrome presented to the emergency department (ED) with several hours of ophthalmoplegia, speech changes, and ambulatory dysfunction. He first noticed these symptoms upon waking in the morning and promptly presented to the ED. He reported diplopia, headache, voice changes, and bilateral lower
extremity paresthesias. Review of systems was notable for cough and congestion for the previous few days. He noted that these neurologic symptoms mirrored his original diagnosis of Miller Fisher syndrome five years prior, specifically the GQ1bpositive Miller Fisher variant, with complete resolution within four months after hospitalization.
Upon ED arrival, his vital signs were as follows: heart rate, 86 beats per minute; blood pressure, 155/73 millimeters of mercury; respiratory rate, 16 breaths per minute; oxygen saturation, 97% on room air; and temperature, 98.9 °Fahrenheit. On examination, he was alert and oriented times four, and his speech was regular without dysarthria or dysphagia. His cranial nerve exam revealed symmetric facies, intact hearing, and symmetric palate. His motor exam showed normal bulk and tone, 5/5 strength in all extremities. A detailed neurologic exam, however, revealed mildly diminished sensation over fingertips, left finger-to-nose dysmetria, truncal ataxia, and inability to ambulate without assistance. He was noted to have dysphonia with hypernasality and hoarseness without aphasia. Fund of knowledge was appropriate. All other exam findings were within normal limits.
Laboratory results showed a complete blood count with a white blood count of 7,000 cells/microliter (µL) (reference range: 4,500-11,000 cells/µL) and a hemoglobin of 13 grams per deciliter (g/dL) (14-18 g/dL). His metabolic panel was notable for a glucose of 135 (70-99); his chest radiograph and negative inspiratory force study were normal. A non-contrast computed tomography (CT) of the brain showed no acute intracranial hemorrhage or mass effect; however, there were findings suggesting a new but not acute left parietal white matter infarct. Negative inspiratory force was reassuring. A lumbar puncture revealed an opening pressure of 14 cm H2O (10-25); clear cerebrospinal fluid with 2 white blood cells per µL (0-5 µL); 0 red blood cells/µL (0-10 µL); glucose, 65 mg/ dL (45-80 mg/dL); protein, 56 mg/dL (15-45 mg/dL); and negative cerebral spinal fluid gram stain. The patient was diagnosed with recurrent Guillain-Barré syndrome, Miller Fisher variant, and admitted to the neurology service.
During his hospitalization, the patient underwent magnetic resonance imaging (MRI) that showed no acute infarct but probable subacute to chronic infarct in the left corona radiata. Antibody studies confirmed positive Gq1b, similar to his previous antibody testing five years prior, confirming his recurrent Miller Fisher syndrome diagnosis. The patient completed a five-day course of IV immunoglobulin with marked improvement of his symptoms. Unfortunately, the patient continued to have ambulatory difficulty, requiring inpatient rehabilitation with residual deficits including ophthalmoplegia and mild dysphonia.
Guillain-Barré syndrome and Miller Fisher syndrome are uncommon but treatable causes of progressive paralysis.5
CPC-EM Capsule
What do we already know about this clinical entity?
Miller-Fisher syndrome (MFS) is an uncommon variant of Guillain-Barré Syndrome with ophthalmoplegia, ataxia, and areflexia.
What makes this presentation of disease reportable?
Miller-Fisher syndrome recurrences are extremely rare, and our patient presented with a recurrence of MFS with similar symptoms after completing initial treatment.
What is the major learning point?
Miller Fisher Syndrome can recur in a patient’s lifetime.
How might this improve emergency medicine practice?
Miller Fisher Syndrome can lead to respiratory distress that requires high levels of suspicion and diagnostic evaluation.
Guillain-Barré syndrome is a post-infectious, immunemodulated neuropathy that can lead to symmetrical ascending paralysis. There is an incidence of 0.4 to 2 per 100,000 with two-thirds of cases caused by an antecedent infection of campylobacter jejuni, leading to molecular mimicry that causes the immune system to target gangliosides in the neurons and slowing neuron conduction.6 The disease reaches its nadir usually around four weeks and recurrence is uncommon, estimated to happen in < 10% of all cases.7
Miller Fisher syndrome is a variant of Guillain-Barré syndrome characterized by at least two of the following: ataxia, areflexia, and ophthalmoplegia3; it is believed to be responsible for 5-25% of cases of Guillain-Barré syndrome.6 Miller Fisher syndrome is thought to be caused by molecular mimicry due to a preceding infection (approximately twothirds of case presentations), reaching a nadir around four weeks, similar to Guillain-Barré. While Guillain-Barré syndrome targets primarily peripheral nerves, resulting in progressive paralysis, Miller Fisher syndrome causes demyelination in cranial nerves as well, generally cranial nerves III, IV, and VI, resulting in ophthalmoplegia and diplopia. In most cases, Miller Fisher syndrome does not recur; however, variants have been observed that recur many times over several years.8
It is common for Guillain-Barré syndrome and Miller
Fisher syndrome to overlap in presentation.6 A textbook presentation of either disease is very unlikely. For example, some people with typical Guillain-Barré syndrome will have ophthalmoplegia, and patients with Miller Fisher syndrome sometimes report ascending numbness.
Fortunately, Guillain-Barré syndrome and Miller Fisher syndrome require similar diagnostic workups and treatment. Advanced brain images such as CT or MRI may be obtained but are not sufficient to diagnose either of these etiologies of progressive neurological disease.6 The gold standard is to obtain cerebrospinal fluid CSF analysis; a positive result would demonstrate increased protein levels without an increase in mononuclear or polynuclear cells, a finding described as albuminocytologic dissociation.5,6 Although this finding is most indicative of Guillain-Barré syndrome and its subtypes, if the clinical history is consistent enough it is still possible to diagnose Guillain-Barré syndrome and its variants without this finding.
Guillain-Barré syndrome and Miller Fishe syndrome are not the only causes of ascending paralysis. Diseases such as tick-borne paralysis and electrolyte abnormalities have similar presentations, including hypomagnesemia and hypokalemia, and should be considered on the differential diagnosis for ascending paralysis.9 Wernicke encephalopathy, characterized by nystagmus, ataxia, and confusion, has been shown to be commonly confused as Guillain-Barré syndrome.9 Other infectious causes of peripheral polyneuropathy such as HIV could be confounded with Guillain-Barré. Finally, neuromuscular junction diseases such as myasthenia gravis and Lambert-Eaton syndrome that present with persistent weakness have been known to be confused as Guillain-Barré or Miller Fisher syndrome.9
The treatment primarily consists of IV immunoglobulin or plasma exchange with respiratory rescue as indicated. Diagnostically, antibody tests for precedent infections can be useful in diagnosing Guillain-Barré syndrome; specifically, anti-GQ1b antibodies are present in 90% of Miller Fisher syndrome cases. Electrodiagnostic studies can also be helpful in diagnosis.6
Patients at risk of imminent respiratory collapse or severe cardiovascular dysfunction will require intensive care unit (ICU) admission.6 Criteria requiring ICU admission include rapid progression of respiratory muscle weakness, evolving respiratory distress, and severe dysautonomia and dysphagia.10 Up to 22% of patients admitted for Guillain-Barré syndrome require mechanical ventilation; therefore, there should be a low threshold for admitting patients to the ICU who appear to be at risk of respiratory distress or cardiovascular collapse. Rapid decline of the expiratory forced vital capacities to < 15 mL/kilogram of ideal body weight, or of the negative inspiratory force to < 60 cm/H2O, each indicate the need for urgent intubation and mechanical ventilation.11
Albuminocytologic dissociation is usually diagnostic for both diseases; however, patients with concerning clinical
Recurrent Miller Fisher Syndrome: A Case Report
history for Guillain-Barré syndrome and its variants should be worked up despite a normal cerebrospinal fluid analysis. These patients require neurological consultation and possible admission for IV immunoglobulin or plasma exchange. There should be a low threshold for ICU evaluation and admission in patients diagnosed with these diseases.
Furthermore, although Guillain-Barré syndrome and Miller Fisher syndrome are established to have a low chance of recurrence, it is not impossible to have multiple courses of these diseases. A history of Miller Fisher syndrome should not necessarily preclude a recurrence.3,7
Guillain-Barré syndrome and its variant, Miller Fisher syndrome, are uncommon causes of progressive paralysis that emergency physicians should be aware of. It is important to keep in mind that they can recur in a patient’s lifetime, as seen in the presented case of Miller Fisher syndrome. Work-up with CT can be non-diagnostic, and a lumbar puncture should be performed. Patients diagnosed with Guillain-Barré syndrome require admission, respiratory evaluation, and ICU admission if there are signs of respiratory distress, severe dysautonomia or dysphagia, or imminent cardiovascular collapse.
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
Address for Correspondence: Kelsey Van Housen, MSPAS, PA-C, Thomas Jefferson University, Department of Emergency Medicine Sidney Kimmel Medical College at Thomas Jefferson University, 1025 Walnut Street, Philadelphia, PA, 19107. Email: kelseyvanhousen@gmail.com
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2026 Vernier et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Chida K, Nomura H, Konno H, et al. Recurrent Miller Fisher syndrome: clinical and laboratory features and HLA antigens. J Neurol Sci. 1999;165(2):139-143.
2. Barbato F, Di Paolantonio A, Distefano M, et al. Recurrent miller fisher: a new case report and a literature review. Clin Ter 2017;168(3):e208-e213.
3. Rocha Cabrero F and Morrison EH. Miller Fisher syndrome. [Updated
Recurrent Miller Fisher Syndrome: A Case Report
June 26, 2023]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi. nlm.nih.gov/books/NBK507717/. Accessed August 22, 2025.
4. Ooi ST, Ahmad A, Yaakub A. Recurrent Miller Fisher syndrome. Cureus. 2022;14(6):e26192.
5. Nguyen TP, Taylor RS. Guillain-Barre syndrome. [Updated February 7, 2023]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; January 2025–. Available from: https://www.ncbi.nlm.nih. gov/books/NBK532254/. Accessed August 22, 2025.
6. Barbagallo G, Caggiula M, Lupo A, et al. Recurrent Miller-Fisher syndrome overlapping Guillain-Barrè syndrome and Bickerstaff brainstem encephalitis: a case report. Clin Neurol Neurosurg
2021;206:106691.
7. Kuitwaard K, van Koningsveld R, Ruts L, et al. Recurrent GuillainBarré syndrome. J Neurol Neurosurg Psychiatry. 2009;80(1):56-59.
8. Ayas ZÖ, Aras YG, Güngen BD. An unusual recurrence of Miller Fisher syndrome: three times in eight years. Noro Psikiyatr Ars 2019;57(1):78-79.
9. Tham SL, Prasad K, Umapathi T. Guillain-Barré syndrome mimics. Brain Behav. 2018;8(5):e00960.
10. Shang P, Feng J, Wu W, et al. Intensive care and treatment of severe Guillain-Barré syndrome. Front Pharmacol. 2021;12:608130.
11. Yuki N and Hartung HP. Guillain-Barré syndrome. N Engl J Med 2012;366(24):2294-2304.
Edward Guo, MD*
Akaysha Duran, MD†
Alexander Kuc, MD‡
Alfred B. Cheng, MD‡
Section Editor: Shadi Lahham, MD
* † ‡
Jefferson Health Northeast, Department of Emergency Medicine, Philadelphia, Pennsylvania University of Pittsburgh Medical Center (UPMC) Memorial, Department of Emergency Medicine, York, Pennsylvania
Cooper University Health Care, Department of Emergency Medicine, Cooper Medical School of Rowan University, Camden, New Jersey
Submission history: Submitted June 7, 2025; Revision received July 18, 2025; Accepted September 10, 2025
Electronically published January 5, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.40087
Case Presentation: A healthy 32-year-old man presented to the emergency department with bilateral knee pain after landing from a jump. He was unable to extend his knees and had pain to palpation superior to the patella. Bilateral quadriceps tendon rupture was confirmed using point-ofcare ultrasound, and the patient underwent operative repair the next day.
Discussion: Bilateral quadriceps tendon rupture is exceedingly rare, which often leads to misdiagnosis. Magnetic resonance imaging is the gold standard diagnostic imaging study but has multiple disadvantages, especially in emergency settings. Point-of-care ultrasound is an excellent tool to screen for this injury and prevent morbidity from delay in surgical repair. [Clin Pract Cases Emerg Med. 2025;10(1):101-103.]
Keywords: bilateral quadriceps tendon rupture; point-of-care ultrasound.
CASE PRESENTATION
A 32-year-old man with no past medical history presented to the emergency department (ED) for bilateral knee pain. He had been jumping on a trampoline when he landed in a squat position, felt pain in both knees, and was then unable to bear weight. He had an athletic build and body mass index of 32.1 (reference range 18.5-24.9) kilograms per square meter. Physical examination was significant for bogginess and tenderness to palpation to the distal femur bilaterally with inability to extend either knee. The patient denied medication or drug use and had no previous orthopedic surgeries.
Plain radiographs of the bilateral knees demonstrated trace amounts of bilateral suprapatellar joint effusions with no fracture or dislocation (Image 1). Point-of-care ultrasound (POCUS) of the suprapatellar regions revealed discontinuity of the bilateral quadriceps tendons with adjacent hematomas (Image 2). Dynamic POCUS of the quadriceps tendons while the patient attempted to extend at the knee further
supported a diagnosis of bilateral quadriceps tendon rupture (Video). No further diagnostic imaging studies were obtained. The patient underwent operative repair the following day and was confirmed to have complete rupture of the bilateral quadriceps tendons.
Bilateral quadriceps tendon rupture is rare with just over 100 reported cases in the literature. It is commonly misdiagnosed at initial presentation due to its rarity and the inability to compare the affected limb to the unaffected limb.1 Risk factors include chronic renal disease, diabetes mellitus, obesity, and steroid use.2 Complete quadriceps tendon rupture is typically caused by forceful contraction of the quadriceps muscles with the knee in a flexed position while regaining balance such as in the case of our patient. Orthopedic consultation is indicated as patients suffer ambulatory dysfunction from injury to the extensor mechanism of both lower extremities. Delay in surgical repair for complete
CPC-EM Capsule
What do we already know about this clinical entity?
Quadriceps tendon rupture is uncommon; bilateral cases are rare and frequently missed, delaying surgical repair and worsening patient outcomes.
What is the major impact of the image(s)?
Video of left and right suprapatellar regions with the patient attempting to extend his knees demonstrates bilateral quadriceps tendon rupture.
How might this improve emergency medicine practice?
Dynamic point-of-care ultrasound may improve recognition of tendon rupture and expedite a rapid, accurate diagnosis.
rupture is correlated with quadriceps retraction, muscle atrophy, and decreased functional outcomes.3
Plain radiographs may show indirect signs of quadriceps tendon rupture but are rarely diagnostic. Magnetic resonance imaging (MRI) of the knee is the gold standard imaging study but has several limitations including cost, time, and availability. Physical examination paired with radiologyperformed ultrasonography has been used to diagnose quadriceps tendon rupture with sensitivities reported as high as 100%.4 Thus, ED POCUS is an excellent screening modality to assess for bilateral rupture and prevent delay in diagnosis, treatment, and potential morbidity. It was particularly valuable in our case due to the inability to compare findings to an unaffected limb. Furthermore, to our knowledge, no prior peer-reviewed case report has included annotated, dynamic POCUS images visualizing quadriceps tendon rupture.
Our images provide novel educational value, demonstrating a practical, real-time imaging technique to improve recognition of this rare injury. Advantages of POCUS include universal availability and rapid utility, making it an ideal screening tool for quadriceps tendon rupture in the ED that may also be diagnostic. Its major limitation is operator dependence. In cases with any doubt of the diagnosis, an MRI should be obtained given its superior specificity. 4,5
Video. Dynamic point-of-care ultrasound of the left and right suprapatellar regions in sagittal plane with the patient attempting to extend his knees, demonstrating bilateral quadriceps tendon rupture (arrows).
L QUAD, left quadriceps; R QUAD, right quadriceps.
Address for Correspondence: Edward Guo, MD, Jefferson Health Northeast, Department of Emergency Medicine, 10800 Knights Road, Philadelphia, PA 19114. Email: edward.guo@jefferson.edu.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Copyright: © 2025 Guo et al. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
The authors attest that their institution requires neither Institutional Review Board approval, nor patient consent for publication of this case report. Documentation on file.
1. Abduljabbar FH, Aljurayyan A, Ghalimah B, et al. Bilateral simultaneous quadriceps tendon rupture in a 24-year-old obese patient: a case report and review of the literature. Case Rep Orthop. 2016;2016:4713137.
2. Camarda L, D’Arienzo A, Morello S, et al. Bilateral ruptures of the extensor mechanism of the knee: a systematic review. J Orthop 2017;14(4):445-453.
3. Hak DJ, Sanchez A, Trobisch P. Quadriceps tendon injuries. Orthopedics. 2010;33(1):40-46.
4. Perfitt JS, Petrie MJ, Blundell CM, et al. Acute quadriceps tendon rupture: a pragmatic approach to diagnostic imaging. Eur J Orthop Surg Traumatol. 2014;24(7):1237-1241.
5. LaRocco BG, Zlupko G, Sierzenski P. Ultrasound diagnosis of quadriceps tendon rupture. J Emerg Med. 2008;35(3):293-295.
John E. Madias, MD
Icahn School of Medicine at Mount Sinai, New York, New York Elmhurst Hospital Center, Department of Cardiology, Elmhurst, New York
Section Editor: Rick McPheeters, DO
Submission history: Submitted November 17, 2025; Revision received November 22, 2025; Accepted November 24, 2025
Electronically published January 14, 2026
Full text available through open access at http://escholarship.org/uc/uciem_cpcem
DOI: 10.5811/cpcem.53978
To the Editor:
Virella et al¹ reported on a 66-year old woman who sustained a fall with injury consequent to syncope in the setting of takotsubo syndrome (TTS), triggered while she was participating in a Zumba dance class, with subsequent fast recovery, and discharged after four days’ hospitalization. The authors reported that point-of-care ultrasound (POCUS) revealed apical hypokinesis with thickened basal septum, left ventricular outflow obstruction (LVOTO), and systolic anterior motion of the mitral valve (SAM-MV), which they attributed to a Venturi effect. The authors should be congratulated for the management of their patient.
The objective of this letter is to draw the authors’ attention to some issues needing further consideration, to further enhance the value of their work and contribute to the followup care of their patient. Consequently, I would appreciate the authors’ response to the following comments/questions, in view of the inclusion of reference #5 in their report:2 1) Was there any intraventricular pressure gradient recorded during POCUS or subsequent conventional echocardiograms (CECHO), during hospitalization? 2) Did the systolic murmur1 persist until discharge, or at follow-up? 3) Did the patient have a post-discharge CECHO? 4) Does the patient have underlying “sigmoid septum”2-5 on POCUS or CECHO? 5) Does the patient have underlying latent/overt hypertrophic cardiomyopathy,2-5 in view of the fact that the patient “reported experiencing intermittent lightheadedness over the prior one to two weeks while walking, which resolved with rest”?1 6) The authors should also consider an alternative to the “Venturi effect” mechanism for the LVOTO, the SAMMV, and apical hypokinesis,1 described in detail in their reference #5.2 7) According to the above, one could surmise that this patient suffered an “increased afterload/segmental
left ventricular coronary mismatch”-based TTS, rather than the “neurohumoral TTS type,”3-5 described in reference #5.2 Indeed, we should even consider that the presented patient with “apical ballooning” did not have TTS, after all; let’s not forget that the pathophysiology of TTS is still elusive!
Address for Correspondence: John E. Madias, MD, Department of Cardiology, Elmhurst Hospital Center, 79-01 Broadway, Elmhurst, NY 11373. Email: madiasj@nychhc.org.
Conflicts of Interest: By the CPC-EM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The author disclosed none.
Copyright: © 2026 Madias. This is an open access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY 4.0) License. See: http://creativecommons.org/ licenses/by/4.0/
1. Virella A, Jose S, Mirro J, et al. Not just another broken heart: a case report of takotsubo cardiomyopathy causing syncope. Clin Pract Cases Emerg Med. 2025;9(4):467-470.
2. Citro R, Bellino M, Merli E, et al. Obstructive hypertrophic cardiomyopathy and takotsubo syndrome: how to deal with left ventricular ballooning. J Am Heart Assoc. 2023;12(21):e032028.
3. Madias JE. Frequent POCUS and auscultation for an earlier diagnosis of takotsubo syndrome and unraveling of its pathophysiology: the possible crucial role of LVOTO. Curr Probl Cardiol. 2024;49(5):102482.
4. Madias JE. Takotsubo syndrome in the elderly: frequent auscultation and POCUS. QJM. 2024;117(7):547.
5. Madias JE. Left ventricular outflow tract obstruction/hypertrophic cardiomyopathy/takotsubo syndrome: a new hypothesis of takotsubo syndrome pathophysiology. Curr Probl Cardiol. 2024;49(8):102668.



BENEFITS
- Western Journal of Emergency Medicine Subscription
- CAL/AAEM News Service email updates - Discounted AAEM pre-conference fees
- And more!
CAL/AAEM NEWS SERVICE
- Healthcare industry news
- Public policy - Government issues - Legal cases and court decisions