

HSV mRNA Vaccine Services
Herpes simplex virus type 2 (HSV -2) infects approximately 13% of people aged 15–49 worldwide, can cause genital herpes Current treatments only alleviatesymptoms but cannot cure the infection. There are currently no preventative vaccines, highlighting an urgent need for breakthroughs in prevention strategies.
Glycoproteins, including gB ,gC ,gD ,gE , gH , and gL , play crucial roles in viral attachment, entry, and immune evasion, and represent criticaltargets for herpes simplex vaccine development .
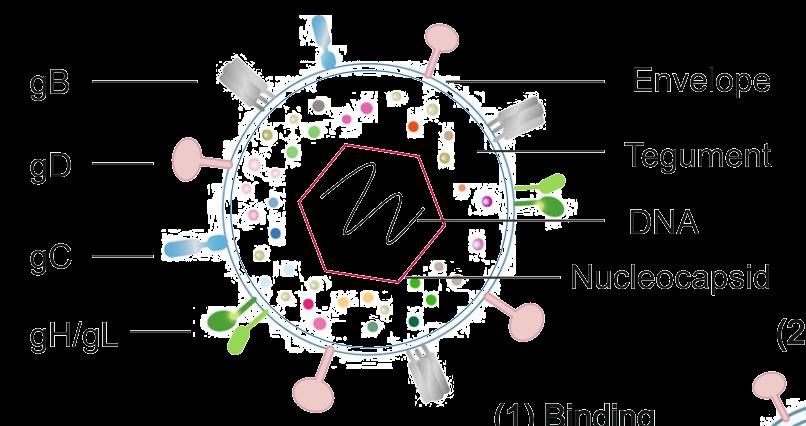
Source: Molecular Biomedicine
RNASci provides HSV glycoprotein mRNA Products and Custom Services.
mRNA Product
Product Coding
BNT163 (BioNTech )
Truncated gC protein: gC2 (27–426 AA)
Truncated gD2 protein: gD2(26 –331 AA)
Truncated gE2 protein: gE2 (24–405 AA)
Naked mRNA or mRNA -LNP (ALC -0315)
Custom Services
Sequence Design & Gene
US20240024463A1, PMID: 31541030
Pipelines: mRNA-based HSV Vaccine
BNT163
BNT 163 isa mRNA -based vaccine candidate for the prevention of genital lesions caused by HSV -2 and potentiallyHSV -1
Target :HSV -2 gC ,gD ,and gE
Institutions:BioNTech and University of Pennsylvania
More Clinical Pipelines
BNT163
Truncated gC ,gD , andgE from HSV -2
LVRNA101 multi-antigen of HSV -2
GSK -4108771A gE/gI(SAM)
mRNA -1608
Not available
Phase 1
Phase 1
BioNTech
LiveRNA (–––)
Phase 1 (termination) GSK
Phase 2 (termination) Moderna
Update: Jan 2026
