

RSV mRNA Vaccine Services
Respiratory Syncytial Virus (RSV) is a major cause of severe lower respiratory infections,especially in infants and the elderly.
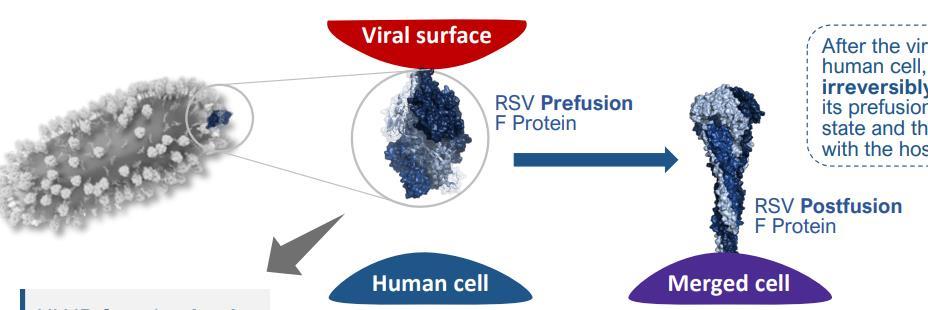
RSV Vaccine development focuses on the viralfusion (F) protein, with the prefusion (pre-F) form being the primary target due to itsabilityto elicita stronger protective immune response .
mRNA Product and Custom Services
Pipelines: mRNA-based RSV Vaccine
mRNA-1345
mRNA -1345 is an mRNA vaccine developed by Moderna for the prevention RSV . It received FDA approval for market authorization in 2024 It is the world's firstmRNA vaccine approved for a disease other than COVID -19.
Target:Prefusion (pre-F) Antigen
Composition: pre -F mRNA encapsulated in SM 102 LNP
Synonyms :mResvia (brand name)
More Clinical Pipelines
-1345 RSV pre -F
SP0256 Not available Phase 2
ABO1105 RSV pre -F
AFN0205 RSV pre -F
IN006 RSV pre -F, bi-valent
CL -0059, CL-0137 Not available
STR -V003 RSV pre -F
RSV mRNA vaccine Not available
SYS6016 RSV pre -F
NR222 Not available
LVRNA007 Not available
RV -1770 RSV pre -F
JCXH -108 RSV pre -F
Phase 2 Abogen (ψψ)
Phase 2
RNAlfa (ψψψ)
Phase 2 Innorna ( ψψ)
Phase 1/2 Sanofi
Phase 1/2 Starna (ψψψψ)
Phase 1 GSK
Phase 1
CSPC ( ψψ)
Phase 1 NanoRibo (ψψψ)
Phase 1 LiveRNA (ψψψ)
Phase 1 RNAimmune (ψψψψ)
Phase 1 Immorna (ψψψψ)
YKYY025 RSV pre -F IND Youcare (ψψψψ)
NT -INF -001 RSV pre -F
Not available RSV pre -F, bi-valent
Not available Not available
Not available Not available
Not available Not available
Update: Jan 2026
IND
IND
IND
Nextranslate (ψψψψ)
Rhegen (ψψψψ)
Aimbio (ψψψψ)
IND Sinovac (ψψψψ)
IND
Walvax (ψψψψ)
