

Dalman address: Meeting challenges of COVID-19, SVS future direction head-on
By Bryan Kay
In his introduction to the second of two Presidential Addresses on the 2021 Vascular Annual Meeting (VAM) slate (Aug. 18–21), new Society for Vascular Surgery (SVS) President Ali AbuRahma, MD, gave voice to that which seems virtually undeniable: that the SVS presidency of Ronald L. Dalman, MD, was defined by a double whammy of enormous challenges—both of which hit not only wider society but the world of medicine and vascular surgery.
Dalman spent much of his time at the SVS helm dealing with matters related to the COVID-19 pandemic. And the Stanford University vice chair for clinical affairs in the department of surgery was also holder of the SVS office when the diversity, equity and inclusion (DEI) torch was lit in the aftermath of a controversial paper published in the Journal of Vascular Surgery (JVS) in the back half of 2020.
Few might argue he did not meet these twin challenges head on. After a 2020 in which VAM was canceled in the thick of the pandemic, for a while it looked like the 2021 event might suffer a similar fate. Yet, there he stood at the VAM 2021 podium in San Diego on Aug. 20, accepting the gauntlet from AbuRahma to deliver his presidential swansong. On the DEI front, too, the SVS tackled head on the fallout from the JVS publication—popularly retermed #Medbikini in the Twittersphere, Dalman would remind his audience in his address.
So it was that Dalman’s address chimed with the zeitgeist of the times. “As a nation and as a professional medical association,
Doubling down on quality: Hodgson champions appropriateness of care
we have been through a lot this year,” he told those gathered for his Friday morning lecture. “But we’ve learned a lot as well. As Kim Brooks wrote in the New York Times, ‘Pandemics make visible what’s been hidden.’”
Dalman had just taken attendees on a journey through the storied history of both vascular surgery itself and the Society as a representative entity, as the SVS marked the beginning of its 75th anniversary—from birth as a singular specialty, through adolescence and maturation, and into the presence it bears today. “Courage and commitment were needed to launch a professional society dedicated to an entirely new medical
“As a nation and as a professional medical association, we have been through a lot this year”—
RONALD L. DALMAN
Kim Hodgson, MD, the 2019–2020 SVS president, challenges SVS members to confront key issue as he delivers his belated Presidential Address.


specialty,” Dalman said of the founding years. “But just as our nation has changed tremendously in the ensuing decades, so too has our Society and the specialty it created.”
There was “the breakthrough decade,” as Dalman described it. This saw the introduction of arterial reconstructive surgery; prosthetic grafts followed; and the SVS established its first guidelines committee. Later came non-invasive vascular imaging, then widespread adoption of vascular fellowship training. Into the 1980s, and the rapid evolution and improvement of endovascular technology dawned. The modern SVS emerged as a member services organization in tandem with the rise of multidisciplinary societies. And, more recently, there has been the rapid transition to outpatient treatment and a quality/outcome focus, Dalman added.
So to the present day, the endpoint of his presidency, and the portent of those difficult matters that helped define his time leading the SVS: “And now,” Dalman asked the crowd, “looking forward another 75 years, who and what will define this time, our time?”
He pointed to images depicted in a
continued on page 2
‘Let’s move on to solutions,’ keynote speaker tells diversity, equity and inclusion focus session
By Beth Bales
Lee Kirksey, MD, issued an unapologetic apology of sorts at the outset of his discussion of diversity, equity and inclusion (DEI) issues at VAM. “If I ruffle any feathers I apologize, but I think it’s a conversation that’s important to have,” said Society for Vascular Surgery (SVS) member Kirksey, of the Cleveland Clinic, in an Aug. 20 lecture. ”Let’s move beyond the problem we understand exists and let’s move on to solutions.”
KIRKSEY’S ADDRESS, “EQUITY challenges in vascular surgery: Closing the gap,” kicked off an hour-long session covering DEI issues, including a slew of scientific abstract talks. The coronavirus “unveiled longstanding inequities … and laid bare problems that have existed for many years for poor brown, Black and white Americans,” he said, adding that evidence of these inequities in medical care and outcomes converged amid a background of social unrest. He reviewed some of the history of addressing such issues with the SVS, including a report from 2010, the creation of the DEI Task Force (now a committee), its recommendations and other positive steps the SVS has taken in the past few years. The existence of an equity action plan demonstrates the Society’s focus, he said. “Inequality and inequity do not exist in a vacuum,” he said. “It impacts the way we care for patients.”
continued on page 2

Ronald L. Dalman
Dalman address: Meeting
challenges of COVID-19, SVS future direction head-on
continued from page 1
slide projected behind him on the auditorium screens—”my 1987 University of Washington Surgical Residency composite, the 1994 Executive Board of the Society for Vascular Surgery, with Fogarty as president, the 2001 composite of Stanford Vascular Surgery. Nearly all white—with or without the coats—nearly all male, and not remarkable in that way, in their respective contexts. But the world is changing.”
Dalman referenced the changing demographics of the U.S. Minorities will constitute a majority of the population by 2042, so the development of “a representative healthcare workforce now will help us better prepare for this future,” he said. “Hannah Valantine, a transplant cardiologist and faculty colleague, recently completed her term as chief officer for scientific workforce diversity at the National Institutes of Health. This slide summarizes her assessment: The health of underrepresented minorities is optimized when physicians from concordant backgrounds are involved in their care. This message was reinforced recently regarding COVID-19 vaccination, by SVS member Lee Kirksey and colleagues from the Cleveland Clinic.”
These demographic trends are mirrored in the vascular world, Dalman pointed out: Data from the 2021 member census showed that more than 50% of SVS members are younger than 50 years of age, and 23% are women.
More than a third of SVS members younger than 30 are women, as compared to 3% in his own age group, and more than a third are underrepresented minorities in medicine, regardless of age.
Dalman turned to the case of Samuel Kountz, MD, to illustrate his point.
talents like Kountz’s, how many others’ contributions have been overlooked or lost, to the detriment of our Society, our patients, our history?”
Dalman also scrutinized the Society’s history for its record on gender diversity. “It took 40 years following the founding of the SVS for female vascular surgeons to become members,” he pointed out. By 2013, when Julie Ann Freischlag, MD, became SVS president, women accounted for approximately 8% of the membership.
Ultimately, the SVS strives to be worthy of its mantle as serving the specialty, Dalman continued. “Despite everything we have accomplished, we can and must do better. We must minimize bias, implicit or otherwise, in the workings and processes of the SVS.”
So it was that, in March, the Society committed to achieving the objectives highlighted by the Diversity Task Force led by Bernadette Aulivola, MD, and John Eidt, MD, Dalman said. “The DEI Committee was commissioned as an element of SVS governance structure. Chaired by Vince Rowe, this Committee is already contributing substantially to the work of the SVS. We created a diversity position statement and updated our mission statement, core values and bylaws to reflect these priorities. We are developing DEI educational resources for SVS members, including implicit bias training for leaders throughout the organization.
“Despite everything we have accomplished, we can and must do better. We must minimize bias, implicit or otherwise, in the workings and processes of the SVS”—
RONALD L. DALMAN
Kountz was a pioneering vascular and kidney transplant surgeon in the 1960s and 1970s, he said. A prolific contributor to the vascular field, Dalman said he appears to have been the first Black surgeon to present his research before the SVS in June 1962 in Chicago.
But Kountz was ultimately lost to the vascular specialty as he pursued the role of chief of kidney transplantation at the University of California, San Francisco in 1967, eventually becoming personally responsible for 25% of all kidney transplants completed in the U.S. by the time of the 1970s.
“Intrigued by vascular surgery and vascular disease management as evidenced by his early investigations, Kountz’s career developed in another direction,” Dalman explained.
“I’m embarrassed to say that the records of the SVS don’t tell us who was the first Black member, but it wasn’t Dr. Samuel Kountz, because he never joined. Or more likely, was never asked to join, as far as we can tell, as membership was by invitation in the early years. By failing to encourage and include
“We completed the first-ever member census and developed metrics to track DEI progress across multiple domains annually. Since we cannot improve what we cannot measure, this year’s benchmark census will prove invaluable in the years going forward in helping us meet our goals. In fact, progress is being made on every priority outlined on this summary slide from the DEI report. Perhaps most representative is the recent evolution of our Appointments Committee process.”
New blood is important to the Society, Dalman reminded SVS members: “To that end, we are advancing plans to create a Young Surgeons Section within the SVS, limited to members in their first 10 years in practice.”
The Society must look ahead to continue getting better, Dalman declared as he brought his address and presidency to its final message.
“What brought our organization to this point in its history, impressive as our accomplishments are, won’t be enough to get us through the next 75 years,” he said. “The world will still need vascular specialists at the turn of the next century, and looking forward to our 150th anniversary in 2096, we must evolve to ensure that the SVS remains relevant and effective in the decades to come.”
‘Let’s move on to solutions,’ keynote speaker tells diversity, equity and inclusion focus session
continued from page 1

Lee Kirksey
He acknowledged issues within the SVS, including representation of various demographic groups. Women have made good strides; they now comprise 15% of SVS membership and hold 29% of committee and council member positions (including 25% of chair positions). Among trainees, 25% of vascular fellows and 34% of residents are women.
However, only 2% of SVS members are Black and just 6% are Hispanic. No Blacks or Hispanics are members of SVS councils, with only 3% of committee members from these demographic groups.
“Senior female leadership, with the energetic support of early- and mid-career female surgeons have made progress,” he said. “In terms of race, we have a long way to go.”
One issue is that only 60% of SVS members have identified their
Other tactics and possible solutions include maintaining accurate demographics, plus optimizing awareness of implicit bias (IB) amongst discordant providers, including IB training and expanding the definition of vascular care providers. Kirksey enumerated several ways bias—conscious or not—impacts care, including in revascularization procedures and amputations. “Vascular surgeons are involved in all levels of intervention and population health,” he said. “We must fully understand as a group and actively engage in conversations around the value-based care delivery model, which is predicated on thoughtful healthcare provision.”
Efforts to improve healthcare include a bill that has been introduced to amend a section of the Social Security Act “to provide for coverage of peripheral
“Senior female leadership, with the energetic support of early- and mid-career female surgeons have made progress. In terms of race, we have a long way to go”
— LEE KIRKSEY
race/ethnicity to the Society, he said. “DEI tactics start with this,” he stressed.
He urged development of a pipeline to spur those UnderRepresented in Medicine (URiM) and enhancement, specifically, of the vascular surgery URiM pipeline. He also advocated medicine identify a group specifically tasked with a strategic action plan to enhance the visibility of role model vascular surgeons amongst Hispanic, Black and Native American medical students
A “silver lining” during COVID-19 has been the creation of a network of Black vascular surgeons. Past President Ronald L. Dalman, MD and Executive Director Kenneth M. Slaw, PhD, enthusiastically supported this group working in concert with SVS, said Kirksey.
arterial disease [PAD] screening for at-risk beneficiaries, disallow non-traumatic amputation services without anatomical testing under the Medicare program, and for other purposes.” Another is a prospective trial on PAD now seeking National Institutes of Health funding.
“To date only two trials in the cardiovascular space have exclusively enrolled Black and Hispanic patients. This will be the first in the PAD space,” Kirksey said.
He ended his talk with a quote from author James Baldwin: “Not everything that is faced can be changed, but nothing can be changed until it is faced.”
“You decide if it applies,” Kirksey told the audience. “Discussing inequity is indeed a difficult conversation, but we should focus on solutions and avoid casting blame.”
Significant reintervention after F-BEVAR ‘does not negatively impact survival’
By Bryan Kay
Patients who underwent reinterventions after fenestrated and branched endovascular aneurysm repair (F-BEVAR) had improved long-term survival over those who did not undergo a secondary operation, a large study of all investigational device exemption (IDE) F-BEVAR procedures carried out at nine U.S. medical centers from 2005-2020 found.
The consortium of researchers further discovered after analysis that major, minor and low magnitude reinterventions were associated with improved survival but that high magnitude reinterventions were not. The investigators classified all reinterventions performed after the initial procedure as either open or percutaneous and as major or minor according to Society for Vascular Surgery (SVS) reporting standards. However, the consortium went further by designating the secondary procedures as either high or low magnitude depending on the physiologic impact of the intervention. Sara L. Zettervall, MD, an assistant professor of surgery at the University of Washington School of Medicine in Seattle, delivered the findings during Plenary Session 5 at VAM on Aug. 21.
There are limited data on long-term outcomes after F-BEVAR for thoracoabdominal and juxtarenal aneurysms, despite an increasing number of fenestrated and branched procedures being performed, said Zettervall. Against this backdrop, Zettervall and colleagues set out to evaluate mortality after secondary intervention following F-BEVAR,
the effect on long-term survival and which interventions carried the most risk by studying all patients enrolled in nine physician-sponsored IDEs. Of the 1,700 F-BEVAR cases included in the study, 23% were found to have involved a secondary intervention and 6% multiple.
Reintervention technical success was “very high at 96%,” she told VAM attendees, while mortality was recorded at just 1%. “When we looked at timing of these reinterventions, a quarter of all reinterventions occurred in the first 30 days, and this accounts for the majority of all high magnitude reinterventions,” Zettervall said. “Furthermore, 50% of all reinterventions happened within the first six months. When we looked at reinterventions over time, freedom from secondary intervention was 82% at one year. However, this decreased to 59% among patients with complete five-year follow-up.”
Those patients who ultimately required secondary intervention after F-BEVAR had undergone more complex initial procedures and had more complex anatomy, Zettervall and colleagues found.
In terms of secondary interventions carried out in a percutaneous fashion, the treatment of target vessels accounted for more than half.
“This was followed by proximal and distal extension, which is of particular importance because these account for large-bore sheath interventions,” said Zettervall, who explained that a key difference between the SVS reporting standards and the consortium’s high-low magnitude classification system involved large-bore sheath access. This type of access “accounts for a large proportion of major reinterventions according to SVS reporting standards but was

not considered a high magnitude reintervention in our new classification system,” she explained. As for open reinterventions, “access-type complications were by far the most common,” followed by bypass and bowel resection. Open, high magnitude and major reinterventions accounted for a small proportion of all interventions performed, Zettervall said, pointing out that the elimination of large-bore sheath access in high magnitude reinterventions accounted for about 10% of major reinterventions. In sum, long-term survival in patients who underwent secondary intervention was improved, the study found, with no difference detected in terms of aortic-related mortality.
“When we utilized our SVS reporting standards, we did find differences in aortic-related mortality as well as survival, with those patients who underwent no reinterventions performing worse,” said Zettervall.
“When we finally assessed using magnitude, the trends became even more clear: We found patients with low magnitude reinterventions performed by far the best among patients in terms of aortic-related mortality as well as all-cause survival. On adjusted analysis, again we saw that reinterventions were protective for all-cause mortality.


“However, when we looked specifically at aortic-related mortality, we found that high magnitude operations did have an adverse effect on survival, while low magnitude interventions were protective.”
Zettervall concluded that “a significant level of reintervention does not negatively impact survival. Rather, a reasonable rate of reintervention may be a marker for adequate follow-up.”
Sara L. Zettervall
Crawford
Forum on
multispecialty vascular
care: ‘We can work together’

By Bryan Kay
New Society for Vascular Surgery (SVS) President Ali AbuRahma, MD, put on an E. Stanley Crawford Critical Issues Forum at this year’s VAM focused on the role of multispecialty care in vascular and endovascular surgery, hoping to get at least some answers to the question: Can the competing specialties of the vasculature work together?
As the session unfolded on VAM opening day Aug. 18, he got the perspectives of providers from three other specialties involved in treating vascular disease. AbuRahma himself declared: “Vascular providers must have defined, dedicated vascular and endovascular training during their formal residency or fellowship. Multispecialty practice, if feasible, will enhance and improve vascular care. We can work together, regardless of our differences, since we all share common goals, of providing care for vascular patients.”
One of AbuRahma’s objectives with the forum was to highlight that those carrying out vascular procedures should be qualified to do so—and have the requisite competency and training.
He had earlier outlined how vascular surgeons are a minority in a crowded field of specialists who treat vascular disease, pointing out that the vascular surgical discipline is the only specialty dedicated to treating the totality of vascular disease.
Next at the podium was Mark Bates, MD, an interventional cardiologist who operates in the same multidisciplinary practice as AbuRahma at West Virginia University School of Medicine/ Charleston Area Medical Center in
Charleston, West Virginia. Bates erred on the side of what’s possible: AbuRahma heads up a vascular center of excellence in West Virginia that the pair held up as a model others could possibly follow.
“What are we doing well? I know there are maybe—perhaps—appropriate turf challenges out there around the country, but we are doing things together,” Bates said. “I think Dr. AbuRahma, in your leadership position—I’m looking forward to seeing some of these things come to fruition: Multispecialty guidelines; how we’re working together in
helping vascular patients, more senior specialists should “extend a hand, and help pull them up into this space so they work together.”
James F. Benenati, MD, of Miami Cardiac & Vascular Institute and a former president of the Society of Interventional Radiology (SIR), provided perspective from the standpoint of an interventional radiologist in a multispecialty group.
“Why are we even discussing this?” came his opening gambit. “Is cooperation absolutely necessary? Well, we’re going to argue yes. But to start with, many of you are in your own practices. So we could say that if you want to stay like that, you can. But you heard [Dr. AbuRahma] speak about the future, so for today— we’re in August 2021—you’re fine. Though I have the feeling in the next few
“When we eliminate turf, billing and what we make, and—if we can share in that—I think quality becomes a much bigger, important point for us, and patients do better” — JAMES F. BENENATI
training modules for people from different domains; looking at quality and outcomes.”
By coming together, “you’re going to accomplish more for the greater good, and not only your society, but for patients with vascular disease,” Bates told attendees. When providers from the different specialties see young physicians who have a “particular passion” for
years that’s going to change a lot.”
Physicians in many institutions are being commoditized, Benenati argued, “and the more we’re together, the more power we have.” The turf issues referenced by Bates, he said, can be eliminated—changing fee-for-service to quality outcomes. “When we eliminate turf, billing and what we make, and—if we can share in that—I think quality
becomes a much bigger, important point for us—and patients do better.” In his world, Benenati found working in concert made his group more profitable; they could provide comprehensive vascular care; and they could work to standardize quality.
“We’re in a situation now, like [Dr. AbuRahma] says, where we need offense and defense,” he said. “Collaboration is one way, with multiple specialties, to accommodate a shortened workforce, prevent burnout and provide a better lifestyle for all of us. We can talk about the advantages of improving outcomes; doing more complex cases; crosspollinization of training; the ability to be more efficient with procedure scheduling, cost-sharing; effective use of support services; taking money out of the conversation removes turf; and providing our administrators, regulators and patients with a comprehensive service line, with expertise in all areas.”
Representing interventional vascular medicine, Bruce Gray, DO, of the University of South Carolina in Greenville, made the case for combined, collaborative conferences between vascular medicine and vascular surgery like the ones he once saw take shape in his world. “In these meetings, it was really helpful—we both learned to understand disease in the same way,” Gray said. It was a collaborative culture that cultivated mutual respect—leading to impacts not only in patient care but also research efforts. “Let’s learn to learn together,” Gray added.
Mark Bates speaks from the podium during the VAM 2021 Crawford Forum

Analysis shows TCAR is more cost-effective than CAS for carotid stenosis
By Will Date
Transcarotid artery revascularization (TCAR) produces similar outcomes to the gold standard treatment for carotid stenosis—carotid endarterectomy (CEA)—but comes at a greater cost. This was among the conclusions presented by Sheila M. Coogan, MD, at VAM during Plenary Session 4 on Aug. 20, drawn from analysis of the cost-effectiveness of treatment modalities for nonemergent significant carotid artery stenosis. The analysis also showed that TCAR was incrementally more cost effective than carotid artery stenting (CAS).
“I THINK AS A SPECIALTY WE RECOGNIZE that carotid stenosis is a significant problem,” Coogan commented in the opening to her presentation, noting that a total of 34% of ischemic strokes result from carotid artery disease. Globally, she added, more than $11,621 million is projected to be spent on treatment of carotid artery disease in 2023, of which 37.8% will be within the U.S.
Working within the Memorial Hermann Health Care System (MMHS)—a 13-hospital network including community and academic facilities—Coogan and colleagues sought to determine which was the most cost-effective carotid revascularization treatment, using Vascular Quality Initiative (VQI) data to assess each of the three modalities.
A hospital cost program, EPSI (Enterprise Performance Systems Inc.), was also used to track the use of resources and assign estimates of cost based upon
payroll and general ledger expenses. “These are real dollars, and this is a real-time metric that hospitals are using,” commented Coogan.

Data were analyzed using descriptive statistics, analysis of variance (ANOVA), linear and logistic regression, while incremental cost-effective ratio (ICER) was computed where more effective treatments were more expensive, and cost savings where more effective treatments were less expensive, the VAM audience heard. Between July 2017 to June 2020, MHSS performed a total of 1,143 non-emergent carotid revascularization procedures, the majority of which were treated via CEA (798, 69.8%), followed by TCAR (177, 15.5%) and CAS (168, 14.7%).
Researchers recorded rates of stroke and transient ischemic attack (TIA) for patients treated using each of the three modalities, with events recoded in 1.1% of CEA patients, 1.7% of TCAR patients and 4.2% of CAS
“Our institution was very reluctant to pay for TCAR because it was so much more expensive ... but we have demonstrated that even though they did not have any pushback for CAS procedures, TCAR was more cost-effective”—
SHEILA M. COOGAN
patients. Substantial differences were also observed in relation to the cost of treatment in each of the three groups, Coogan reported, the most costly of which was TCAR. “CEA was significantly less expensive for overall adjusted procedure time and length of stay,” she added. Breaking down the cost by category, Coogan told VAM attendees that the biggest cost differential related
TCAR has ‘statistically equivalent’ stroke and death outcomes to CEA, new study finds
By Bryan Kay
Silk Road Medical has announced positive results from an independent analysis of standard surgical risk patients undergoing carotid endarterectomy (CEA) and transcarotid artery revascularization (TCAR) for atherosclerotic carotid disease. The data were presented during Plenary Session 2 at VAM on Aug. 18.
OVER 20,000 STANDARD SURGICAL risk patients were included in the analysis (15,198 CEA and 5,066 TCAR) from the Vascular Quality Initiative (VQI) CEA and carotid artery stent (CAS) registries over four years (August 2016 to September 2020).
The results for symptomatic and asymptomatic standard surgical risk
patients undergoing CEA and TCAR for atherosclerotic carotid disease showed that TCAR had statistically equivalent stroke and death outcomes to CEA, while showing a nine-fold reduction in cranial nerve injury (2.7% vs. 0.3%, p=<0.001).
The data were delivered in a presentation by Patric Liang, MD, a vascular surgery fellow at Beth Israel
Deaconess Medical Center in Boston.
“The promise of safely preventing stroke while delivering benefits from a less-invasive approach is critically important as we consider an expanded patient population. This first ever large-scale, standard
to the cost of devices. Comparing the treatment modalities on a cost-bycost basis, the analysis showed that CEA vs. CAS saved roughly $5,000 per case, while CEA vs. TCAR was a $6,000 cost saving per case.
ICER, which is based upon the outcomes and the cost effectiveness of the procedure, determined that TCAR was incrementally more cost effective than CAS, at a rate of $640 saved per event prevented.
The study was limited by the fact that there was a low event rate among patients studied, and the fact that it was a single-center analysis, Coogan said.
Concluding her presentation, Coogan pointed to the finding that though CEA remains the gold standard for the treatment of carotid artery stenosis, TCAR produced similar outcomes, albeit at a higher cost.
“In the future I hope there will be a multicenter study using VQI data, which is readily available, and using realcost data from your institutions, and if we could find a way to collaborate with multiple institutions we could probably have enough outcome events that we would power this for the most cost-effective treatment for carotid stenosis,” she remarked.
Coogan also commented that further costeffectiveness analysis could provide greater insight in the guidance of treatment choices.
She said: “One of the reasons we proposed this was that our institution was very reluctant to pay for TCAR because it was so much more expensive than other procedures, but we have demonstrated that even though they did not have any pushback for CAS procedures, TCAR was more cost-effective.
“It highlights the fact that VQI is a really powerful way, both for contract negotiations with device companies, and also for analysis of what your institution might be willing to support financially.”

“These results reinforce the effectiveness of TCAR as an important treatment option for patients with carotid artery disease and standard surgical risk factors” —
MARC L. SCHERMERHORN
Depictions of carotid endarterectomy and stenting
surgical risk analysis demonstrates that TCAR delivers on that promise,” said Erica Rogers, president and chief executive officer of Silk Road Medical.
“These results reinforce the effectiveness of TCAR as an important treatment option for patients with carotid artery disease and standard surgical risk factors,” said Marc L. Schermerhorn, MD, chief in the division of vascular and endovascular surgery at Beth Israel Deaconess Medical Center.
“TCAR again shows equivalent risk to CEA of perioperative stroke, death, or myocardial infarction and ipsilateral stroke at one year in patients undergoing carotid revascularization with improved secondary outcomes such as cranial nerve injury.”

Sheila M. Coogan
Carotid disease
Arterial/Venous

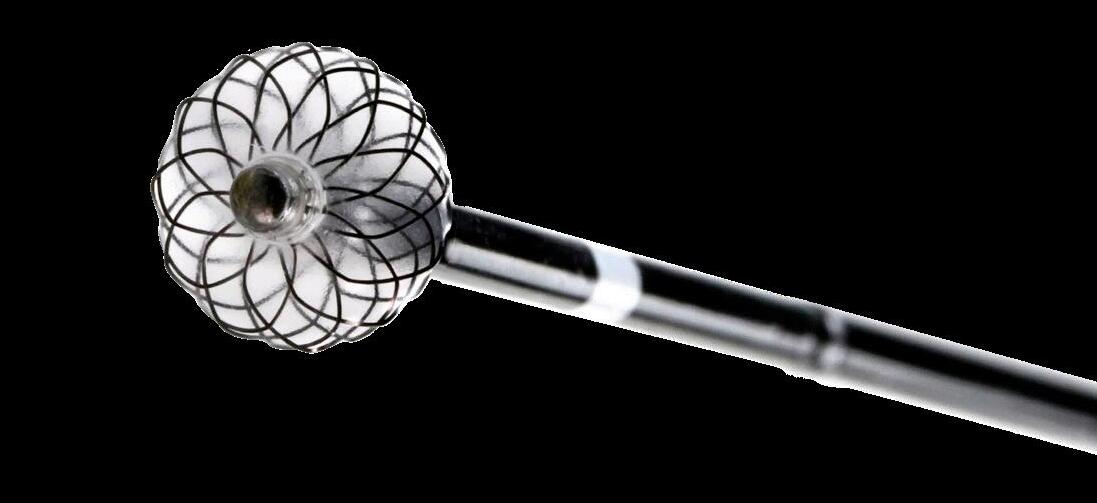
Venous disease
‘The
future is very bright for venous valve replacement’
By Bryan Kay
The progress of two “promising” new devices in the venous valve replacement space were outlined during a VAM special session co-hosted by the American Venous Forum (AVF) on Aug. 18.
Ellen Dillavou, MD, associate professor of vascular surgery at Duke University Medical Center in Durham, North Carolina, placed a lens over the BlueLeaf system (InterVene) and the VenoValve (Hancock Jaffe Laboratories) device in “Bringing what’s new in venous to you!”
“The history of venous valve replacement is long and usually not successful,” Dillavou told attendees. Both autogenous and non-autogenous valves have been tried for years to replace those that have been damaged or congenitally absent in the leg, she continued, but “on the horizon we now have two very promising new products.”
The BlueLeaf system, Dillavou explained, is an endovascular means to create a mono- or bicuspid valve. The system “takes the thickened intima of a postthrombotic vein that is damaged and, with a dissector, gets a wire through that thickened vein wall, and then uses hydrodissection, balloon dissection and a mechanical dissector to form a pocket,” she said, adding: “Intravascular ultrasound (IVUS) is critical during the procedure—both in choosing the vein site as well as looking at the final result.”
Early results, which are in publication, indicate a 93% technical success rate “of creating at least a monocuspid valve,” with the researchers testing the system producing a mean of 1.3 valves per patient. The investigators reported having 10 patients at one-year follow-up, with no thrombotic events recorded, Dillavou noted. “Although [the patients] didn’t have a difference in the popliteal reflux time, they did have a decrease in the Venous Clinical Severity Score (VCSS).” Additionally, nine out of nine venous ulcers either healed or improved at the last follow-up, she added.
Turning to the VenoValve—a moncusp biologic component valve mounted on a stainless steel structure, created from a porcine aortic valve—she described the device’s progress since its first-in-man placement by Jorge Ulloa, MD, of Universidad de los Andes in Bogota, Colombia, and the publication of early results in 2020.
A total of 11 patients with C5–C6 disease were studied, with Ulloa and colleagues reporting 100% technical success. One-year follow-up found that six out of 10 patients required patch venoplasty and five had adverse events, with the two-year results also presented (see adjacent story).
“At one year, the average reflux decreased by 54% across their patient population, their VCSS score decreased by an average of 56%, and the pain score had an average reduction of 76%,” said Dillavou.
The VenoValve team, now starting a pivotal trial in the U.S., is aiming to enroll about 100 patients, with 18 of 20 sites confirmed. Patients will be followed for one year, with five years of postmarket follow-up planned, she added. “The future is very bright for venous valve replacement,” Dillavou concluded.
“For the first time, we have two promising options for advanced venous stasis, human trials are in progress, and hopefully the next frontier will be honing patient selection and looking at long-term maintenance of these devices.”
Colombian researchers report positive 24-month results for VenoValve
Promising two-year results for an emerging potential prosthetic venous valve replacement— which is about to commence a U.S. pivotal trial—were announced during a VAM session on Aug. 19.
RESEARCHERS FROM BOGOTA, Colombia, revealed that patients treated with the bioprosthetic VenoValve (Hancock Jaffe Laboratories) were found to have seen a 63% improvement in venous reflux time, 60% improvement in venous clinical severity score (VCSS) score, and a 93% improvement in their recorded pain when compared with preoperative levels.
The data were presented by Sebastian Cifuentes, MD, a vascular surgery research fellow at Fundacion Santa Fe-Universidad de los Andes, Bogota, Colombia, during a VAM International Forum. Cifuentes is part of a research team led by principal investigator Jorge Ulloa, MD, of the same institution.
Ulloa and colleagues recently published the six-month data of their first-in-human study of chronic venous insufficiency patients with C5-C6 disease in the Journal of Vascular Surgery-Venous and Lymphatic Disorders
Eleven patients who were implanted with the device in Colombia completed one year of follow-up, with eight then followed out to 24 months. One patient died due to natural causes not related to the device, Cifuentes explained.

“For the first time, we have two promising options for advanced venous statsis, human trials are in progress, and hopefully the next frontier will be honing patient selection” — ELLEN DILLAVOU
wound healing.”
The main two-year findings showed that venous reflux decreased from an average of 1.95 to an average of 0.72—a 63% improvement.
“We can see that after 30 days of implantation, we have seen a stable improvement of the reflux,” Cifuentes elaborated.
Disease manifestations—measured by revised VCSS—decreased from an average of 13.38 to an average of 5.38, a 60% improvement. “This means we took patients from severe disease to mild disease,” Cifuentes said. Pain—measured by visual analog scale (VAS) scores—decreased from an average of 7.25 to an average of 0.5. This represented “a dramatic improvement,” he added.
“These results allowed VenoValve to start a U.S. pivotal trial, the SAVVE [Surgical anti-reflux venous valve endoprosthesis],” Cifuentes explained.
“Our results show that VenoValve appears to be safe. Our patients had a 63% improvement in reflux time, 60% improvement in VCSS score and a 93% improvement in their pain levels compared with preoperative levels. The VenoValve has shown a good safety profile, with few complications, and benefits that outweigh the known risks.”


“Another was not able to attend appointments due to agerelated physical limitations, and we had one lost to followup.” Going forward, patients enrolled will be followed every six months for 48 months.
At 24 months after implantation, the research team found no device-related issues, Cifuentes continued. One patient developed contralateral ulcers, he said, but neither hospitalizations nor recurrence of ipsilateral ulcers were noted. “Although wound healing was not our primary outcome, we have seen significant improvement in ipsilateral
The SAVVE U.S. pivotal trial for the VenoValve will include 75 patients at up to 20 sites. The primary endpoints for the SAVVE trial will be the same as for the first-in-human trial: the primary safety endpoint is the occurrence of a major adverse event (MAE) in less than 10% of patients at 30 days post-VenoValve implantation, and the primary effectiveness endpoint is improvement of reflux equal to or greater than 30% at six months following VenoValve surgery.
MAEs are defined as the composite of allcause mortality, deep wound infection, major bleeding, ipsilateral deep vein thrombosis (DVT), or pulmonary embolism. Improvement of VCSS and VAS scores are also included in the SAVVE study as secondary endpoints.
The Food and Drug Administration (FDA) recently granted Breakthrough Device designation status to the VenoValve.
“We will continue to work with agencies like the FDA and [U.S. patent office], and other regulatory authorities throughout the world towards our ultimate goal of reaching the millions of patients suffering from CVI,” Hancock Jaffe CEO Robert Berman said in April as the company announced the first patent covering the VenoValve.-Bryan Kay
Top: VenoValve in action Above: Sebastian Cifuentes
VenoValve device
Appropriate care
Doubling down on quality: Hodgson champions appropriateness of care
By Bryan Kay

It was supposed to occur at the canceled 2020 Vascular Annual Meeting (VAM), but finally given his opportunity at the podium, 2019–2020 Society for Vascular Surgery (SVS) President Kim Hodgson, MD, wasted little time in conveying the message he has long sought to convey to SVS members: that the threat posed to vascular surgery by “bad actors” carrying out inappropriate procedures is real—and that it is long past time to address the issue.
Hodgson was speaking during the 2021 iteration of VAM on Aug. 19, delivering his Presidential Address a little over 12 months after he was scheduled to do so. The theme was appropriateness in care, a topic he kicked off during the E. Stanley Crawford Critical Issues Forum in 2019 as he began his presidential year. During that 2019 talk, he referenced the story of a cardiologist and cardiac surgeon whose rate of cardiac procedures and surgeries was 4–5 times the expected rate for the hospital and the population they served. The next day, Hodgson reminded VAM 2021 attendees, data were
Veterans with claudication who undergo revascularization
‘significantly more likely to have major amputation than those who do not’
Patients within the Veterans Affairs Administration healthcare system who underwent an intervention for intermittent claudication (IC) were consistently at a three-to-four times greater risk of undergoing subsequent amputation compared to those who did not, researchers at Stanford University report.
FURTHERMORE, INTERVENTION was associated with a 40% greater hazard of disease progression to chronic limbthreatening ischemia (CLTI), they found. The findings were revealed during a Plenary Session 6 presentation at VAM
presented by Caitlin W. Hicks, MD, assistant professor of surgery at John Hopkins Medicine in Baltimore, and colleagues at VAM 2019 on the overuse of early peripheral vascular interventions for claudication. Two years down the line, further research has built upon the charge that outliers are performing inordinate levels of certain procedures, including atherectomy carried out in the office-based lab (OBL) setting.
“As I lay out my case for changes here today, you might find yourself wondering, why should the SVS lead this charge?” Hodgson told those gathered. “To me,
2021 on Aug. 21 by Elizabeth L. George, MD, a vascular surgery integrated chief resident at Stanford in California.
George and colleagues looked into the association between intervention for IC, and progression to CLTI and amputation in light of data demonstrating “significant center-level variability” in adherence to guideline-recommended therapy. Both the Society for Vascular Surgery (SVS) and the American Heart Association (AHA) recommend intervention for IC only after a failed trial of medical and exercise therapy, she said.
With recent evidence suggesting intervention for claudication hastens progression of disease in as little as oneand-a-half years, “we sought to answer the question of whether intervention actually carries a risk for limb loss that is worse than the natural history of medicallymanaged claudication,” George told VAM attendees.
George and colleagues carried out an observational national cohort study of veterans diagnosed with IC between 2003–2012, identifying patients with incident peripheral arterial disease (PAD) and focusing on a diagnosis of claudication.
the answer is simple—because somebody has to address what should never have been allowed to get to this level of threat to us and our patients in the first place. And, because no organization is better positioned to lead the way than your SVS, by virtue of the portfolio of quality programs we have developed and the integrity of our members. That last point is critical—the integrity of SVS members—because doing our part to fix this problem is going to require each and every one of us to put our integrity on display by being transparent about who we are and what we do.”
The impact of the explosive growth of atherectomy on patients and healthcare finances is problematic and growing exponentially, Hodgson lamented. He turned to further findings from Hicks showing that of the roughly $267 million reimbursed by Medicare for firsttime femoropopliteal peripheral vascular interventions in 2019, 90% was for atherectomy, “leaving only $26 million to pay for all of the other therapies that actually have evidence showing that they work.”
But Hodgson said his message should not be construed as an attack on OBLs writ large. “The SVS recognizes the importance and value of the OBL to our members and their patients, which is precisely why we recently transitioned the OBL Committee into an OBL Section and selected exemplary community practice leaders, Drs. Robert Molnar and Cliff Sales, to lead it,” he continued. “It is also why we created a Community Practice Section, also led by exemplary community practitioners, Drs. Dan McDevitt and James Cravens. Not only do many of its members serve in other SVS leadership positions, including on the Executive Board, but just two months ago we held an election where one of the candidates for vice-president was a community practitioner.”
The SVS recognizes that the overwhelming majority of community practitioners suffer because of those who are practicing appropriately, Hodgson said. The SVS is about quality and appropriateness, “and is not anti-OBL, but rather anti-OBL abuse,” he added.
Hodgson called out what he dubbed “the enablers” of this type of practice—”people or constructs that encourage or enable negative behavior in another.”
The Centers for Medicare & Medicaid Services (CMS)
The primary and secondary outcomes of the study were, respectively, major amputation progression to CLTI.
A total 25,737 veterans were diagnosed with IC at 130 Veterans Affairs medical centers during the study period, with a third undergoing vascular intervention during follow-up. A further 755, or 2.9%, had a major amputation—493 of them within five years of diagnosis.

“Veterans who underwent revascularization were overall significantly more likely to have major amputation than patients who did not [5.6% vs. 1.5%; p<0.001], and this was true at the fiveyear mark as well,” George revealed.
“In adjusted Cox proportional hazard modeling, intervention was associated with three times higher hazard of amputation [adjusted hazard ratio (aHR)
3.60, 95% confidence interval (CI) 2.97–4.39; p<0.001].”
Prescription of an antiplatelet agent (aHR 0.77, 95% CI 0.60–0.99; p<0.001] but not statin medication was associated with significantly lower hazard of amputation, George continued.
On multivariable logistic regression, she said, intervention was “significantly associated” with three times greater odds of amputation at five years (adjusted odds ratio [aOR] 3.84, 95% CI 3.14–4.69; p<0.001). “But prescription of statins and antiplatelets were not associated with amputation,” she added. “A number of comorbidities were significantly associated but, in both models, current smoking status was not.”
George et al also found on Kaplan-Meier analysis that veterans who underwent intervention experienced significantly
Elizabeth L. George
“The SVS recognizes the importance and value of the OBL to our members and their patients, which is precisely why we recently transitioned the OBL Committee into an OBL Section” —
KIM HODGSON
was one, with “distorted reimbursement incentives” contributing to the problem, he argued. “The OBL, if properly incentivized, would be a very positive development for patients and physicians, but CMS policies have instead incentivized inappropriate behavior.”
The situation is already being addressed, Hodgson went on. With the growth in atherectomy bringing peripheral vascular interventions under increased scrutiny, reimbursement for atherectomy is going to plummet, Hodgson said. This will come on top of the Medicare fee schedule cuts recently announced, which appear to specifically target vascular surgery and interventions, he explained. “You think maybe CMS is sending the message that if we don’t do something to curb runaway utilization, then they will?” Hodgson posited. “And as always, it is going to be the responsible physicians practicing appropriately who are going to be hurt the most, because they won’t respond by just doing more unnecessary procedures to make up the difference as the less scrupulous—those who have created this situation— are likely to do.”
Another enabler is the Food and Drug Administration (FDA), Hodgson said, calling for target lesion revascularization (TLR) to be “tossed as a legitimate endpoint for peripheral vascular disease treatment success in FDA approval trials.” Hodgson also pointed to vascular surgery’s industry partners. He said they should be “the canary in the coal mine for inappropriateness. You are uniquely positioned to spot abuse early—before patients
worse amputation-free survival at five years (p<0.001).
THE RESEARCHERS DELVED DEEPER.
“When we used a three-level independent variable—time to diagnosis to intervention less than or greater than six months compared to no intervention—our model revealed that intervention when performed more than six months out from diagnosis was associated with a greater hazard of major amputation compared to earlier intervention,” George explained.
“This unexpected finding was also confirmed on Kaplan-Meier analysis and prompted us to perform a post-hoc analysis to incorporate the secondary outcome of progression to CLTI to address the
are harmed. You know every one of the outliers in vascular care.”
Lastly, he turned to the OBL itself as the ultimate enabler. The OBL is an inanimate structure, he said, so “it’s not guilty of anything. Nor are the overwhelming number of vascular surgeons, in both clinical and academic practice, providing great care in OBLs by prioritizing ethical clinical values over business values in their clinical decisions.” Yet, their lack of meaningful oversight enables the unscrupulous to misbehave, Hodgson said.
He referenced the 2015 SVS Presidential Address delivered by Peter Lawrence, MD, which was also devoted to appropriateness. The talk contained a list of proposed measures aimed at addressing the problem of overtreatment of vascular disease. “First on his list was the need to accredit OBLs,” recalled Hodgson. Lawrence had noted that “there needs to be an accreditation system that establishes standards for quality in the office, as occurs in the hospital,” Hodgson said.
To Lawrence’s point, continued Hodgson, this raises a new SVS initiative—the prospect of the Vascular Center Verification and Quality Improvement Program (VCV&QIP), which the SVS has developed in partnership with the American College of Surgeons (ACS), helping to clean up vascular care (see related story in September’s Vascular Specialist). “Modeled after other successful ACS Quality Improvement programs, the vascular program verifies a facility’s compliance with evidencebased care guidelines, peer review, and continuous quality improvement,” Hodgson explained, as he revealed the future launch of the program.
Hodgson also touched on another feature of his presidential year—the development of an SVS Supervised Exercise Therapy (SET) app. Held up as a simple, inexpensive means of treatment, contrasted against expensive interventions, it lingers large over discussion of inappropriate care and overutilization of certain procedures.
“That supervised exercise therapy, or SET, works is perhaps the most solidly supported therapy in vascular care,” he said. “A 2017 Cochrane Review determined that ‘High-quality evidence shows that exercise programs
possibility that these later interventions were actually due to rapid progression of disease.”
Using a composite endpoint of CLTI and major amputation, the investigators found that intervention was associated with a 40% greater hazard of disease progression in adjusted Cox proportional hazard modeling.
“Intervention for IC is associated with significantly less freedom from disease progression during five-year follow-up on Kaplan-Meier analysis,” George said. “Overall, the rates of disease progression in the veteran population included in this study were quite high, with progression of disease from IC to CLTI noted in 60% of veterans who did not undergo
“As one of the only risk-based studies that looks at the natural history of claudication, we found that within the VA system 33% of claudicants underwent invasive vascular interventions and, overall, 2.9% of veterans received a major amputation”— ELIZABETH L. GEORGE
provide important benefit compared with placebo or usual care in improving both pain-free and maximum walking distance in people with leg pain from intermittent claudication.’ SET has also been shown to have considerable potential for savings in healthcare spending.”
Why, asked Hodgson—with mounting evidence of its effectiveness—isn’t SET used more often? “I think we all know that lack of reimbursement is just a minor part of the answer. Traditional SET requires patients to schedule an appointment, get at least a little bit cleaned up, travel to a facility, find a parking spot, and, because it’s done in a gym setting, can be boring.”
These types of drawbacks to traditional SET prompted the SVS leadership to establish a Health Information Task Force, now a standing Committee, and ultimately the SVS SET app, Hodgson said, adding that enrollment in a randomized controlled trial to demonstrate the efficacy of the app is currently underway. Ultimately, though, Hodgson said it is up to vascular surgeons themselves to use the tools the SVS has developed in order for appropriateness in care to be addressed.
“We can play whack-a-mole every time the bad actors surface until the cows come home, but that leaves a trail of harmed patients and wasted resources,” he told VAM attendees. “So how do we distinguish the good operators from the opportunists in our space? How do we prevent vascular practices from becoming profit centers because of profiteering?
“As I see it, our only pathway to success is to spotlight those practicing appropriately; you don’t think the news media are going to write exposés on physicians practicing appropriately, do you?
“Your SVS leaders, past, present, and future, have spent countless volunteer hours developing a portfolio of tools to help you distinguish yourself through quality practice. Some of them, like clinical practice guidelines and appropriate use criteria, are pretty basic in that they require little from you other than reading them and practicing accordingly; that’s your job. A future SVS Improving Wisely program will give you a snapshot of where you fall on the spectrum of a given practice characteristic. Hopefully you will review it and, if you are an outlier in either direction, you will reflect on that.”
a revascularization and 75% who did undergo a revascularization. Our post-hoc hypothesis that earlier interventions are associated with more rapid progression of disease appears to be confirmed. And we again see that the progression to amputation and CLTI is much slower in veterans who do not undergo an intervention for claudication.”
George conceded that her research team’s analysis suffers from a number of limitations, including a lack of granular data on symptomatology either at presentation or during follow-up; the homogenous patient population served by the Veterans Affairs system; the absence of data on supervised exercise therapy (SET); and the fact the covariates in their models do not vary by time. George further pointed out that the study does not account for different practice patterns.
CITING THE SVS PRESIDENTIAL
Address given by Kim Hodgson, MD, at VAM earlier in the week, George touched on the controversial use of invasive vascular interventions as a firstline therapy for claudication. “As one of the only risk-based studies that looks at
the natural history of claudication, we found that within the VA [Veterans Affairs] system 33% of claudicants underwent invasive vascular interventions and, overall, 2.9% of veterans received a major amputation. Amputation risk for those who did not undergo an intervention was 1% at five years,” she said.
Concluding, George added: “Using three different statistical methods as points of comparison, veterans who underwent revascularization for IC were consistently at three-to-four times greater hazard, odds, risk of undergoing subsequent amputation compared to veterans who did not receive revascularization for IC. And, intervention was associated with a 40% greater hazard of disease progression to CLTI. Earlier use of medical and exercise therapy along with appropriate preoperative patient counseling regarding increased amputation risk with revascularization for IC is warranted.”
Future research is required in order to “identify mechanisms and risk factors for accelerated disease progression following intervention, as well as to figure out how procedural approach factors into the equation,” George also noted.—Bryan Kay

Low Profile without compromise
Relay®Pro offers a low profile system without compromising performance. The platform utilizes the same stent design, materials and foundational Dual Sheath Technology of the proven Relay®Plus.
The Relay®Pro system has a wide range of sizes and tapers allowing each patient access to the right solution, every time.
IDENTICAL TO TREO®
‘Tremendous advances in imaging possibilities’ signal further innovation, says leading vascular chief
By Bryan Kay
It was a complex repair in the thoracoabdominal region of the aorta around 10 years ago, and Matthew Eagleton, MD, and his surgical team were in the process of cannulating several target vessels. They were being guided by what, for the period, was state-of-theart imaging technology. When the chief in the division of vascular and endovascular surgery at Massachusetts General Hospital in Boston looks back on the overlay image—a computed tomography (CT) scan—now, he almost balks.
“Ithink this was one of the original cases I did in Cleveland,” Eagleton says, displaying a slide of the imaging. “Even though this seems really clunky now compared to some of the overlays that are built into systems today, this was cutting edge at the time. Now I look at it and say, ‘How can you tell wherever the hell you were going? It makes absolutely no sense to me.”
Eagleton was speaking before a VAM session exploring the digital transformation in the delivery of comprehensive vascular care, with his contribution zeroing in on advances in intraoperative 3D image guidance on Aug. 21.
During the talk, he highlighted three key players in the imaging space pushing the boundaries of image guidance in endovascular therapy: Cydar Medical, Philips and Centerline Biomedical—the
last for which Eagleton serves as chairman of the company’s scientific advisory board, he disclosed. “Currently, imaging is the key component of endovascular therapy,” Eagleton told attendees. “We use fluoroscopy as the gold standard. It gives us a real-time image, but it’s only twodimensional, it’s associated with radiation exposure and, as procedures get more complicated, the amount of radiation goes up. With increased complexity of endovascular procedures, we need corresponding improvements in imaging.”
The products available now stand in stark contrast to the operating room experience Eagleton described from all those years ago. “A lot of leaps forward were made almost a decade ago when we started to look at 2D fluoroscopy being converted to 3D-guided interventions, predominantly with something called
The Society for Vascular Nursing thanks the following companies for their generous support of the 2021 SVN Annual Conference
EDUCATIONAL GRANTS
3M Healthcare Boston Scientific Corporation Gore Hanger Medtronic Smith & Nephew Silk Road Medical
SPONSORSHIP
Osborn Health, Rooke ‘Best of Awards’
THANK YOU!
fusion imaging, which is just a CT scan overlaid on a fluoroscopy image,” he said.
In that vein, Eagleton outlined a tableau of options whose stages of development demonstrate a “tremendous amount of advancement in our imaging possibilities.”
Cydar EV intelligence maps have improved overlay technology, as well as image position and accommodation, he said. Likewise, Philips’ Fiber Optic RealShape (FORS) has made improvements in the tracking of endovascular tools through the FORS overlay system, while Centerline’s Intra-Operative Positioning System (IOPS) pushes forward 3D imaging and endovascular tracking, Eagleton continued.
Looking ahead, Eagleton pointed out Cydar’s EV maps—dynamic, patientspecific 3D models that can be modified intraoperatively and through deep learning and artificial intelligence—have improved the predictability of patient outcomes. “There is initial intelligenceenhanced planning so that when a clinical user is planning a new case, they are informed by the outcomes of previous patients with similar disease who have had similar procedures,” he said.
and the U.S. as it continues to undergo clinical study. The technology has CE mark approval in Europe and is Food and Drug Administration (FDA) 510(k) cleared.
The developers behind IOPS, meanwhile, are working on improving the applicability of the system’s technology, Eagleton revealed. “IOPS interactive 3D vascular imaging and electromagnetic tracking of our endovascular tools within that vascular tree is based on a mathematical model for vascular image construction generated from CT data. The model is tested by assessing the relative geometry of the aortic branches.”
With 510(k) clearance in the U.S. and the system currently in use at select sites, Eagleton describes technology with “a lot of room for further innovation.”
“With increased complexity of endovascular procedures, we need corresponding improvements in imaging” —
MATTHEW EAGLETON
As for FORS, Eagleton disclosed having no first-hand experience with the technology, instead relating knowledge gleaned from Tilo Kölbel, MD, a professor of vascular surgery at the University of Hamburg, Germany, and Marc L. Schermerhorn, MD, chief in the division of vascular and endovascular surgery at Beth Israel Deaconess Medical Center in Boston.
The platform enables real-time 3D visualization of the full shape of devices inside the body without the need for stepping on the fluoroscopy pedal, sending pulses of light through hair-thin optical fibers within minimally invasive devices. Though the core equipment is set to remain largely the same, related Eagleton, the development of a FORS 3D hub that can recognize several commercial catheters is due in the near future. Currently, the system is limited to two catheters and one guidewire. FORS is currently available at a limited number of centers in Europe
The system now uses holographic digitalization technology “with a headset display so you can look at a 3D picture of the aorta that you’re working on,” he says. In the long run, Eagleton hopes further breakthroughs in these types of technology lead to a zero-radiation world for patients and clinicians. “There has been a tremendous amount of advancement in our endovascular tools, and a tremendous amount of advancement in our imaging possibilities for all of these endovascular procedures,” he said. “I hope at some point we are able to get rid of the fluoroscopy and X-ray exposure completely.”
Yet, Eagleton remains realistic. One attendee queried him on operator feedback when using such devices: How do clinicians know, for instance, if a wire is bending? “Our laudable goal in the beginning was to get rid of all fluoroscopy and that’s probably an unrealistic goal in the shortterm, and it’s just to reduce the amount of fluoroscopy and the amount of contrast used, because you still have to check,” he admitted. “But it’s obvious from a visual standpoint when you’re not where you’re supposed to be.”
Matthew Eagleton
Elsie Gyang Ross, MD, assistant professor of surgery and medicine at Stanford University, California, raised the broader point of which particular technology specialists should buy into as developments unfold: Will most hospital systems be able to acquire multiple intraoperative visualization systems, or will they have to choose one and hope the company behind their selection continues to innovate? “I suspect you’ll probably have multiple,” Eagleton answered. “In some ways, they’re competing, but in others complementary. There are some parts that overlap, some that don’t.”
Eagleton envisages a coming decade filled with innovation and ample room for application: “And there’s going to be more people who are going to come to the table.”
Five-year STABLE II results show Zenith system makes the cut for acute complicated TBAD treatment
By Urmila Kerslake
Joseph V. Lombardi, MD, reported long-term outcomes favoring the continued safety and effectiveness of a composite device (proximal covered stent graft plus distal bare stent) deployed for the endovascular repair of acute, type B aortic dissection (TBAD) complicated by aortic rupture and/or malperfusion.
“STABLE II RESULTS PLACE FREEDOM FROM all-cause mortality at 80.3% at one year, and 68.9% at five years [with the use of the Zenith Dissection Endovascular System]. Further, there were two procedure-related deaths, and the freedom from dissection-related mortality remained 97.1% at one year through five years,” he told VAM 2021 delegates via Zoom on Aug. 19 during Plenary Session 3.
The Zenith Endovascular System includes a proximal covered stent graft to cover the primary entry tear plus a distal bare metal stent, built on a nitinol Z-stent platform, for true lumen support. The bare metal component provides expansible support along the length of the dissected aorta without blocking critical branch vessels to provide a scaffolding to aid adjunctive interventions and facilitate favorable aortic remodeling of the entire dissected aorta.
Gore Excluder IBE ‘effectively prevented common iliac artery aneurysm enlargement’
By Bryan Kay
Five-year results from the U.S. prospective, multicenter study evaluating endovascular repair of iliac aneurysms using the Gore Excluder iliac branch endoprosthesis (IBE) were presented at VAM on Aug. 19, confirming the safety, efficacy and durability of the IBE for treatment of aortoiliac and iliac artery aneurysms, investigators revealed.
“THE IBE DEVICE EFFECTIVELY prevented common iliac artery aneurysm [CIAA] enlargement while maintaining patency of the internal iliac artery and avoiding complications associated with internal iliac artery sacrifice,” they found. “Although CIAA enlargement was rare, abdominal aortic enlargement was more common, suggesting that outcomes of EVAR [endovascular aneurysm repair] may be different in patients with and without associated CIAAs.”
Outlining the adverse events seen in the study, Lombardi commented that notable adverse events at one year were one instance of aortic rupture, one instance of conversion to open repair and two instances of stroke—there were no instances of paraplegia or paraparesis. “Freedom from secondary intervention was 88.2% in one year, and 70.7% at five years and secondary interventions were used to treat a number of presentations, including distal aortic growth. Aortic growth distal to the stent graft suggests the need for continuous monitoring,” he added.
STABLE II is a prospective, single-arm, multicenter study that enrolled 73 patients between August 2012 and January 2015 at institutions in the United States and Japan. The mean age of patients was 60.7 years with nearly 66% being male with acute TBAD complicated by malperfusion (78.1%), rupture (27.4%). Patients were treated with either a composite device (proximal covered stent-graft + distal bare stent, 79.5%) or the proximal stent-graft alone (no distal bare stent, 20.5%). Dissections were more extensive in patients receiving the composite device (408.9 ± 121.3mm long, with 47.4% of dissections extending into the iliac arteries) compared with those who did not receive the distal bare stent (315.9 ± 100.1mm long, with 6.7% of dissections extending into the iliac arteries). Fiveyear follow-up was available for 86.1% of eligible patients.
and the dissected stent region. “Complete false lumen thrombosis rate increased (75% of patients at five-year vs. 51.4% post-procedure), with a higher complete thrombosis rate at five years in patients who received the composite device (81%) compared with the rate in patients who did not receive the distal bare stent (57.1%). There were no incidences of device fracture or infolding reported and these five-year data suggest a positive influence of composite device use on false lumen thrombosis,” he summarized.
Complete thrombosis rate at five years:
The researchers also reported that proximal dissection events were reported in seven patients, including four patients with retrograde progression of dissection and three patients with a new tear/type A dissection. Importantly, Lombardi called out the progressive false lumen thrombosis seen within the stent graft region
Five-year primary patency of the internal iliac artery and external iliac artery IBE limbs was 95.1% and 100%, respectively.
Furthermore, no patients experienced new onset buttock claudication on the IBE side or new onset erectile dysfunction.
Additionally, there were no type I or type III endoleaks and no device migrations identified by the core lab, and freedom from secondary intervention was 88.2%. The device offers a treatment option that allows patients the benefits of endovascular therapy yet preserves pelvic perfusion.
Darren B. Schneider, MD, chief of the division of vascular surgery and endovascular therapy in the University of Pennsylvania Health System, Philadelphia, presented the findings during Plenary Session 3.
“The positive outcomes from this fiveyear study validate the effectiveness and durability of the Gore Excluder IBE for the treatment of iliac artery aneurysms,” said Schneider, the study’s national principal investigator.
“Importantly, the IBE reduces the risk of common iliac artery enlargement and rupture while maintaining patency of the internal iliac artery to prevent complications associated with internal iliac artery sacrifice.”
Used in conjunction with the Gore Excluder abdominal aortic aneurysm (AAA) endoprosthesis, the IBE isolates the common iliac artery from systemic blood flow and preserves blood flow in the external iliac and internal iliac
To a question posed on how the decision was made to deploy or not deploy the bare metal stent, Lombardi clarified: “We left that up to our investigators to determine the need for additional stenting using the bare metal stent. A lot of the dissections were very focal on the thoracic aorta and some thought that an additional dissection stent was not needed to support normal aorta distally. So, this was a primary reason why dissection stents weren’t deployed, but the fact that we didn’t have dissection stents in a small cohort of patients gave us a nice control group to look at afterwards.” When pressed on recommendations regarding when to use the dissection stent and tips on how far to place it, especially when the dissection was particularly distal, Lombardi explained: “The data is very supportive of the distal aortic remodeling using the dissection stent. And oftentimes, as you saw on the data, secondary interventions are not infrequent, and we’ll have to go back and reintervene for distal aortic growth to remedy ongoing expansion. The use of the dissection stent in that situation for me and for a lot of our colleagues has been very, very helpful in mitigating a continued false lumen flow.”


“The positive outcomes from this five-year study validate the effectiveness and durability of the Gore Excluder IBE for the treatment of iliac artery aneurysms” —
DARREN B. SCHNEIDER
Darren B. Schneider presents five-year results on the Gore Excluder IBE
arteries. Its design allows for a two-staged repositionable deployment for precise placement and it has a precannulated internal iliac gate and bi-femoral delivery for ease of use.
The device was approved by the Food and Drug Administration (FDA) in February 2016. Gore says it was the first off-the-shelf aortic branch device approved in the U.S.
Medical therapy linked with low early mortality in type B aortic intramural hematoma patients
By Bryan Kay
Medical management of type B aortic intramural hematomas is associated with low early mortality but a 19% risk of aortic-related intervention—primarily for proximal descending thoracic aneurysms, researchers from the Mayo Clinic in Rochester, Minnesota, discovered.
THE INVESTIGATORS REPORT THAT ELEVATED creatinine, the hematoma thickness and aortic diameter at presentation portend eventual failure of medical management. The findings were delivered during Plenary Session 4 at VAM on Aug. 20 by Parvathi W. Balachandran, MD, on behalf of a team of colleagues.
The study aimed to evaluate the natural history of medically treated aortic intramural hematomas and identify factors linked to faltering medical management. Balachandran and colleagues gathered data from
consecutive patients diagnosed with type B hematomas from 1995 to 2019. A total of 91 were identified. All patients received antihypertensive medications and pain control. Initial management was best medical therapy in 85% (n=77) and surgery in 13% (n=12) of patients. Two patients declined intervention and died within 48 hours, “hence were not included in the study,” said Balachandran.
The primary outcome of the study was 30day mortality. Secondary outcomes included five-year all-cause and aortic-related mortality, as well as aortic-related surgical intervention for dissection or aneurysm. Balachandran et al found that 30-day mortality was 5% (n=4) among the best medical therapy group and 17% (n=2) in the surgery treatment group.
All-cause five-year mortality was comparable between groups (57% vs 50%, p=0.8), with a median survival of 6.8 years after therapy and 5.9 years after surgery. “However, five-year aortic-related mortality was higher in the surgical group at 25% compared to best medical therapy due to high perioperative mortality,” Balachandran said.
Of the 77 patients who were managed with best medical therapy, Balachandran told VAM attendees, 19% (n=15) worsened and eventually required surgery for dissection (n=3) and aneurysm (n=12)—13% had surgery within two weeks of initial diagnosis and 87% required a late surgical intervention. Of the 81% who continued on the best medical therapy track, 2% succumbed to early in-hospital death.
The median freedom from intervention was 4.6 years. On univariate analysis, coronary artery disease, hypertension, baseline creatinine level, peripheral artery disease, active smoking, intramural hematoma thickness
and maximum aortic diameter were independent risk factors for subsequent need for surgery after medical management, the research team further discovered.
On multivariate analysis, chronic kidney disease with a creatinine >1.2mg/dL, intramural hematoma thickness (>7.7mm) and aortic diameter (>42mm) were predictive risk factors.
Meanwhile, reintervention after initial surgical management was required in 25% (N=3), for aneurysm, endoleak and infected endograft, with a median freedom from reintervention of 5.2 (IQR 3-7.9) years.
Balachandran concluded: “Given the nearly five-year time between type B aortic intramural hematoma and need for surgery beyond 30 days, diligent follow-up is needed. These findings provide useful information to guide patient expectations and optimal treatment.”


for
Parvathi W. Balachandran
VOYAGER PAD: Rivaroxaban plus aspirin should be considered after lower-extremity bypass regardless of conduit type
By Jocelyn Hudson
“A strategy of adding rivaroxaban 2.5mg twice daily to aspirin should be considered after lower extremity bypass regardless of conduit type,” concluded Nicholas Govsyeyev, MD, during the William J. von Liebig Forum at VAM on Aug. 18. Govsyeyev was giving an update on the VOYAGER PAD trial, addressing the efficacy of rivaroxaban and aspirin in peripheral arterial disease (PAD) patients with venous and prosthetic surgical bypass conduits.
“The optimal antithrombotic therapy following infrainguinal bypass is not known,” Govsyeyev began.
He noted that two major trials have examined the role of dual antiplatelet therapy (DAPT) and oral anticoagulation following infrainguinal bypass—CASPAR and DUTCH BOA. While both these trials had neutral results, the presenter detailed, there was significant heterogeneity by conduit type, with the CASPAR trial showing the potential benefit of DAPT in prosthetic conduits and the DUTCH BOA trial demonstrating the potential benefit of anticoagulation in venous conduits. Both trials, however, had a significant increase in major bleeding.
“The VOYAGER PAD trial provides a large, prospective database to further examine the optimal antithrombotic regimen following infrainguinal bypass,” Govsyeyev remarked. This trial randomized 6,554 patients with symptomatic PAD undergoing lower extremity revascularization and receiving standard of care to rivaroxaban 2.5mg taken twice daily or placebo, with standard of care consisting of aspirin, clopidogrel use per the investigator’s discretion, and the encouragement of statin use.
Govsyeyev summarized the key findings: “The trial demonstrated a 15%
“The trial demonstrated a 15% reduction in the primary composite endpoint of acute limb ischemia, major amputation, myocardial infarction, ischemic stroke, and cardiovascular death”
NICHOLAS GOVSYEYEV
reduction in the primary composite endpoint of acute limb ischemia, major amputation, myocardial infarction, ischemic stroke, and cardiovascular death; bleeding was increased, but overall incidence was low with rivaroxaban; there was no significant interaction for efficacy or safety on the basis or surgical or endovascular revascularization; and surgical patients’ risk for primary endpoint events was reduced by 19% with rivaroxaban.”
The objectives of the current study, Govsyeyev explained, were to compare limb outcomes with venous versus prosthetic conduits in VOYAGER PAD patients who underwent bypass, and to evaluate whether the efficacy of rivaroxaban is consistent across conduit types.
In order to assess this goal, the researchers used a multivariate adjusted model to determine the risk of limb outcomes in patients with venous and prosthetic conduits, and Cox proportional hazards to assess for efficacy.
Govsyeyev detailed that a total of 1,448 (66%) patients underwent bypass in the VOYAGER trial. Breaking this down by conduit type, he noted that prosthetic use was slightly more frequent (773 patients; 54%) than venous use (646 patients; 46%).
In terms of baseline characteristics, the speaker communicated that the median age of the venous cohort was 65

(59–70) compared to 66 (61–72) in the prosthetic group, and that the number of female patients in the venous group was slightly lower than in the prosthetic group, at 17 and 22, respectively. Medical comorbidities were not different between the two groups, he added.
With regard to distribution by target artery, Govsyeyev stated that venous conduits were more likely to be used below the knee, whereas three-quarters of prosthetic conduits had an above-the-knee target artery.
Govsyeyev reported that, in the placebo group, the cumulative incidence of unplanned limb revascularization at three years from randomization was 29.6% in the prosthetic cohort (n=388) compared to 13.1% in the venous cohort (n=314), with a risk difference of 16.5%. Regarding acute limb ischemia, the figures were
13.6% and 5.4%, respectively, with an 8.2% risk difference.
The presenter then outlined results related to the efficacy of rivaroxaban on the primary composite endpoint by conduit type. “Prosthetic patients were at higher risk for primary endpoint events, and this was reduced with rivaroxaban, with an absolute risk difference of 6.4%,” Govsyeyev revealed. Among the venous conduits, the data showed an absolute risk difference of 7.9% with rivaroxaban. Govsyeyev added that there was no significant interaction by conduit type and overall bleeding was quite low in the rivaroxaban and placebo groups.
Furthermore, the speaker informed the audience that there was consistent benefit of rivaroxaban on limb outcomes regardless of conduit type.
Thirty-year experience indicates SMA aneurysms are ‘relatively stable’
Based on 30 years of experience, researchers have concluded that aneurysms of the superior mesenteric artery (SMA) are “relatively stable” and that those <20mm may be safely monitored with surveillance imaging. The experience also suggests that operative treatment should be considered if the aneurysms are symptomatic and that patients with vasculitis of fibromuscular dysplasia may need closer follow-up.
PRESENTING THESE FINDINGS AT VAM DURING THE WILLIAM J. VON LIEBIG Forum on Aug. 18, Grayson S. Pitcher, MD, of Mayo Clinic, Rochester, Minnesota, notes that the study represents one of the largest series on aneurysms of the SMA and its branches.
“SMA aneurysms are rare and the SVS currently recommends repair of all SMA aneurysms and pseudoaneurysms regardless of size,” Pitcher stated. However, he stressed that the rupture rates of true aneurysms versus pseudoaneurysms vary significantly, with a much lower risk for true aneurysms.
The presenter detailed that two recent, small series suggest SMA aneurysms <20mm may be safe to observe. In light of this, Pitcher and colleagues at the Mayo Clinic aimed to review the centre’s 30-year experience with aneurysms of the SMA and its branches, determine their growth rate, and evaluate predictors of growth.
The research team conducted a retrospective review of all patients diagnosed with an aneurysm of the SMA or one of its branches—excluding pseudoaneurysms and mycotic aneurysms—in the period 1988–2018. Demographics, presentation, aetiology, anatomic characteristics, and treatment modalities were taken into consideration. Endpoints included outcomes, growth, and predictors of growth.
Pitcher communicated that a total of 131 patients, or 144 aneurysms, were included in the study. The average age of the patients was 60, 84 (64%) were male, and 66% of the aneurysms were degenerative, representing the most common aetiology. The majority of the aneurysms were found incidentally, with only 27% being symptomatic.
Growth rate of the aneurysms was first estimated using linear regression. However, only 16% had a growth rate of ≥1mm per year. Due to the large number of aneurysms without any change in size, aneurysms were dichotomized by slope and a categorized analysis was performed using logistic regression.
Addressing the VAM audience, Pitcher reported: “Initial aneurysm size was significantly associated with growth rate for fusiform aneurysms only, meaning initial size is only predictive of growth for fusiform aneurysms”. The odds ratio (OR) for the fusiform aneurysm growth rate was 1.13 (95% confidence interval [CI], 1–1.3), with a p-value of 0.02. Initial aneurysm size was not significantly associated with saccular (OR, 0.91; 95% CI, 0.76–1.1; p=1.1) or dissection-associated aneurysms (OR, 1.2; 95% CI, 0.91–1.5; p=0.2).
The team then used logistic regression models to assess the effect of multiple variables on growth. Pitcher said that while the team evaluated a range of variables, none were associated with growth.
The presenter revealed that symptomatic status was significant and that acute abdominal pain (OR, 5.91; 95% CI, 1.56–22.41; p=0.0009) and chronic abdominal pain (OR, 3.76; 95% CI, 1.06–13.13; p=0.04) were significantly associated with growth rate.
Finally, Pitcher informed the audience that there were only two ruptures in the team’s series. One of these ruptures occurred in a 24mm saccular branch aneurysm and the other in a 12mm fusiform branch aneurysm. He added that only one of the ruptures occurred in an aneurysm less than 20mm in size out of a possible 106 possible aneurysms.—Jocelyn Hudson
VAM briefs
SVS announces winners of Scavenger Hunt, Social Media Challenge
By Kristin Crowe
COMPETITION WAS IN THE AIR at this year’s Vascular Annual Meeting (VAM) in San Diego. Attendees had the opportunity to compete in both the Exhibit Hall Scavenger Hunt and the VAM21 Social Media Challenge.
The Exhibit Hall Scavenger Hunt required attendees to download the SVS Events App and visit the seven sponsors’ booths in the exhibit hall, scan the booth’s QR code and then answer the resultant question. Participants tested their knowledge about the number of SVS members, which product of a sponsor’s company was of best use in a particular vascular procedure and more.
Jenna Kazil, MD, received first prize of $3,000 in American Airlines vouchers; Leigh Ann O’Banion, MD, took second prize, a $1,500 Apple gift card; and Mariya Kochubey, MD, won a $500 Amazon gift card.
O’Banion quipped on Twitter that her prize “definitely made running around the booths in 4-inch heels worth it!”
The Social Media Challenge—with both in-person and at-home versions— utilized Twitter and Instagram. Those at VAM completed seven individual challenges, posting photos of their VAM21 experience while using the #VAM21 hashtag.
At-home participants completed three challenges, sharing their livestreamed experience with their own social media followings, with completed challenges becoming an entry into a random drawing to win SVS swag.
Of those participating in person, Matthew Smeds, MD, won first prize of free 2022 VAM registration; Bernadette Aulivola, MD, won second place of a $100 Visa gift card, and Meryl Logan, MD, won third place of a $50 Visa gift card. Jason Chuen, MD—participating all the way from Melbourne, Australia—won the at-home challenge.

SVS Foundation awards presented during VAM
The Society for Vascular Surgery (SVS) Annual Business Meeting held during the Vascular Annual Meeting (VAM) saw a number of SVS members receive SVS Foundation awards.
SVS FOUNDATION AND AMERICAN COLLEGE OF SURGEONS MENTORED CLINICAL SCIENTIST RESEARCH CAREER DEVELOPMENT AWARD K08:
■ Andrea T. Obi, MD, University of Michigan Medical School: “Myeloid TLR4 epigenetic regulation and signaling in accelerating venous thrombus resolution”
SVS FOUNDATION MENTORED PATIENT-ORIENTED RESEARCH CAREER DEVELOPMENT AWARD (K23):
■ Caitlin W. Hicks, MD, Johns Hopkins University: “Risk factors and outcomes associated with late-life diabetic peripheral neuropathy”
■ Tze-Woei Tan, MD, University of Arizona: “Determinants in limb preservation in Hispanics and Native Americans”
E.J. WYLIE TRAVELING FELLOWSHIP:
■ Shipra Arya, MD, Stanford University School of Medicine
CLINICAL RESEARCH SEED GRANT:
■ Katharine L. McGinigle, MD, University of North Carolina: “Using precision medicine to define adaptive treatment strategies for patients with chronic limb-threatening ischemia”
■ Cassius Iyad Ochoa Chaar, MD, Yale University School of Medicine: “Genetic variants associated with premature peripheral arterial disease”
RESIDENT RESEARCH AWARD:
■ James Melvin, MD, general surgery resident, University of Michigan Medical Center: “Coronavirus induces diabetic macrophage-mediated inflammation via IFNßregulation of SETDB2.” Mentor: Katherine Gallagher, MD
VISTA PILOT PROJECT GRANTS:
■ Misty Humphries, MD, University of California Davis: “Targeted outreach in vascular deserts to improve outcomes for chronic limb-threatening ischemia”
■ Kelly Kempe, MD, University of Oklahoma School of Community Medicine: “Disparities in access to care for limb salvage: The Oklahoma Project”
■ Joseph Mills, MD, Baylor College of Medicine: “A patientcentered approach to reduce diabetes-related lower-extremity amputations among underrepresented minorities”
Submit session proposals for VAM 2022 by Sept. 29
VASCULAR SURGEONS ENCOUNTER SITUATIONS daily that vex or perplex them. Propose one today for examination at the 2022 Vascular Annual Meeting (VAM) in Boston.
The Society for Vascular Surgery (SVS) is seeking ideas and proposals from SVS members for invited sessions for VAM 2022, set for June 15 to 18, 2022. Invited sessions include postgraduate courses, breakfast and concurrent sessions and "Ask the Expert" small-group sessions. SVS opens the submission process to the entire membership, seeking the widest range of topics of interest to its members, who work in a variety of practice settings. Those submitting topics should provide the educational value of a session, a short outline of the program topic, session goals and target audience, among other information. The deadline is Sept. 29. Learn more at vascular.org/VAM.—Kristin Crowe



FROM THE PRESIDENTS: At top, SVS Immediate Past President Ronald L. Dalman, MD (pictured third from left), distributes Presidential Citations at the close of his term. At center, Dalman appears with new Distinguished Fellows of the SVS. And at bottom, Past President Kim Hodgson, MD (center), also awarded Presidential Citations as part of his 2019–2020 term.
3
WIN POSTER CONTEST AT VAM
FROM AMONG 100-PLUS POSTERS IN THE SVS POSTER Competition, the audience selected three participants as winners in the competition’s championship round on the final day— Aug. 21—of the 2021 Vascular Annual Meeting (VAM).
The winners were, in first place, Juliet Blakeslee-Carter, MD,( $1,500 prize) for “Resource utilization and Medicare payment analysis of completion endoleaks following infrarenal endovascular abdominal aortic aneurysm repair”; in second place, Jessica Nguyen ($1,000 prize) for “The use of ‘spin’ in the analysis of non-significant findings in randomized trials in vascular surgery”; and, in third place, Ben Li, MD, ($500 prize) for “Gender differences in faculty rank among academic physicians: A systematic review and meta-analysis.”
Each of the 10 finalists, selected from the full roster of posters a few days earlier, received $300 for advancing to the Championship Round.—Beth Bales
VAM 2021 featured two versions of a social media competition
VAM 2021 Opening Ceremony video captures SVS progress
IMMEDIATE PAST PRESIDENTS
Ronald L. Dalman, MD, welcomed back attendees during the VAM 2021 Opening Ceremony by screening a video that charted the three-quarter century of progress made by the Society.
“This meeting marks the dawn of a new era of learning combining streaming with in-person learning to a worldwide audience, offering discussions on critical issues like diversity, equity and inclusion, and exploring new advances and novel ideas for practice,” said Dalman.
VAM 2021 also marked the beginning of the 75th anniversary of the SVS.
“To achieve this milestone at such a pivotal moment in history is an excellent opportunity to honor both the many innovations that have defined our specialty while simultaneously keeping our eyes on the future as we advance the science in our field,” said Dalman.
The challenges that we face will continue to evolve. Given all that we have accomplished since 1946, I’m confident we are up for whatever comes next,” added the 2020–2021 SVS president.—Bryan Kay
Save the date: Gala celebration is June 17, 2022
IT’S OUR PARTY AND, UNLIKE IN THE Lesley Gore classic song, there will be no crying. And there also will be far more than “16 Candles” on the cake. In fact, there’ll be 75.
The celebration of the Society for Vascular Surgery’s 75th anniversary officially commenced Friday evening, Aug. 20, at the President’s Reception and 75th anniversary kick-off, complete with a festive cake and plenty of people on hand.
Revelries will continue for the next 10 months, culminating at the 2022 Vascular Annual Meeting (VAM) in Boston (June 15 to 18, 2022).
Members, be sure to save the date of June 17, 2022, for the SVS Foundation “Vascular Spectacular” Gala, the 75th anniversary/birthday party for the Society. All proceeds will benefit the SVS Foundation.—Beth Bales

AI and transforming vascular surgery

The VAM 2021 Roy Greenberg Distinguished Lecture saw delegates hear about the potential for artificial intelligence (AI) to transform vascular surgery. Elsie Gyang Ross, MD, assistant professor of surgery at Stanford University, told attendees during her Aug. 19 talk: “We have the ability to shape how this technology can be used to transform vascular care, whether that be developing systems to decrease late referrals, to better manage vascular population health, or improving our operative abilities. Ultimately, the quest for better vascular care will always include integration of our hard-won intelligence, married with excellent use cases from which we can build AI to improve care, decrease inefficiencies and waste.”—Bryan Kay
SVS honors community practitioners
The Society for Vascular Surgery (SVS) honored six members at VAM on Aug. 19 with the Excellence in Community Service award.
This award, in only its third year, honors surgeons in community practice for their outstanding leadership within their community as a practicing vascular surgeon, who have exceptional personal integrity and reflect the highest standards of the profession.
Because of the cancellation of the 2020 Vascular Annual Meeting (VAM), the three 2020 recipients were honored along with the celebrants from the 2021 group.
The 2020 honorees were Krishna Jain, MD, Russell Samson, MD, and William Shutze, MD.
Meanwhile, the 2021 honorees are John Kirkland, MD, of Rome, Georgia; Daniel McGraw, MD, of New Braunfels, Texas; and Kenneth Schwartz, MD, of Purchase, New York.—Beth Bales

Simple innovations to the VenaSeal™ closure system plus new techniques mean greater* impact for you and your practice.
n Greater Efficiencies
n Greater Control
n Greater Ease of Use

To learn more visit medtronic.com/impact

