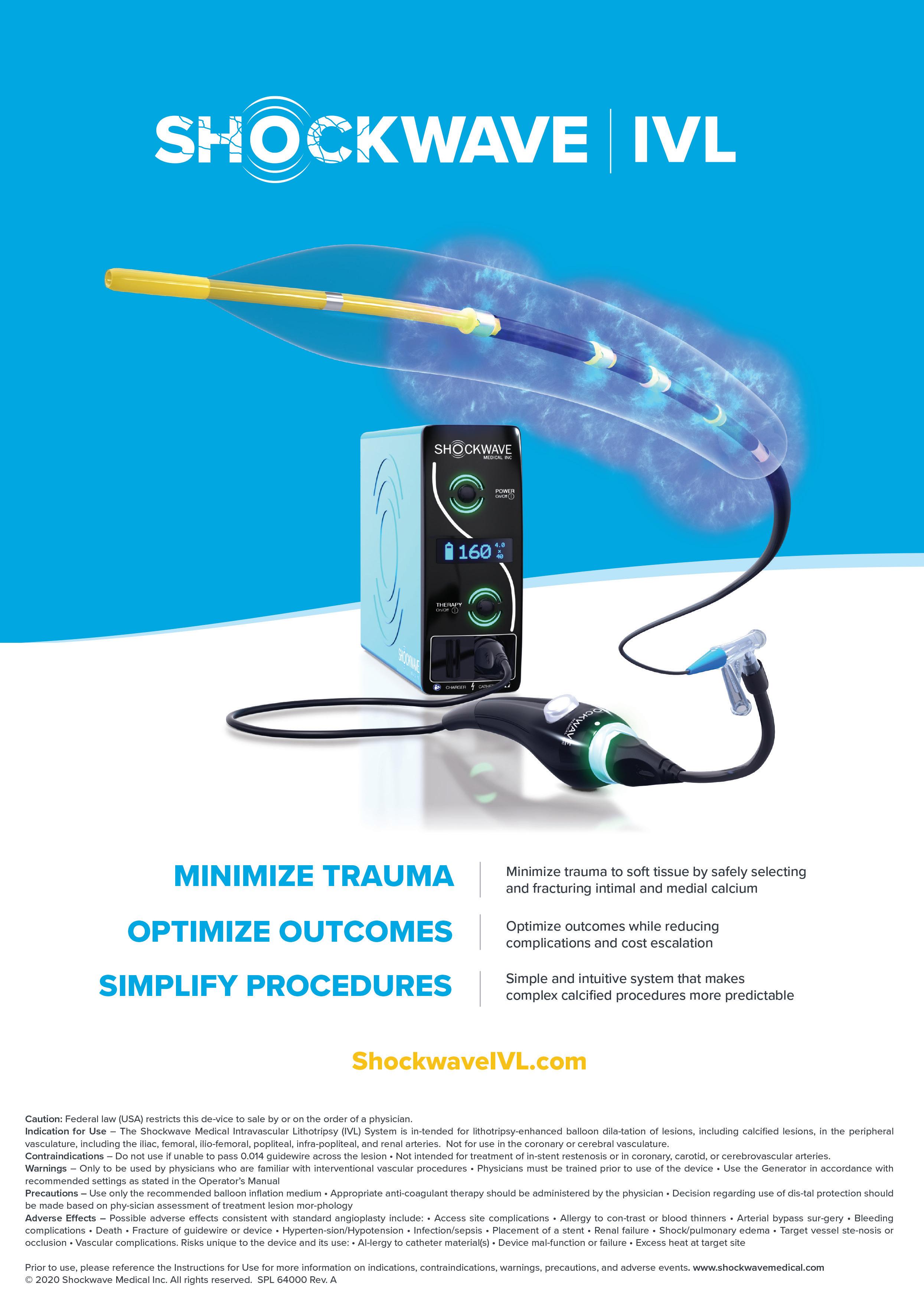
The emerging value and broad applicability of intravascular lithotripsy
An in-depth look at the role of IVL, across various anatomies, in a resourceconstrained healthcare environment
Interview
The value of IVL in a resource-constrained healthcare environment
Frank Arko discusses his experience with intravascular lithotripsy (IVL) and considers its value in a resource-constrained healthcare environment. He concludes that the technology has increased procedure predictability and provided an overall effective method for managing heavily calcified vessels.
When do you think about using IVL?
We increasingly begin to think about using IVL in a multitude of different vascular beds. In our area of the Carolinas, which is the western part of the state, there is a significant amount of both aneurysmal disease as well as very advanced critical limb ischaemia (CLI). We have had challenges in managing severely calcified plaque and have found that other technologies have had some limitations in this regard. So, for those with aneurysmal disease or iliac occlusive disease in which the vessels are heavily calcified, IVL has now become our go-to method. In the past, we managed this with a variety of different technologies. This included the doddering technique, plain old balloon angioplasty, atherectomy, endoconduits, and standard surgical conduits. Each of these worked to some degree, but they added a significant amount of time, cost, and morbidity, especially when utilizing open surgical exposure. The use of IVL has, in many cases, led to reduced overall procedure time, decreased cost of other devices historically used, decreased hospitalization cost, and has given us an effective method for managing these heavily calcified vessels. When treating other forms of peripheral arterial disease (PAD), we are limited by many of the same technologies and so have been encouraged with the results that we have seen with IVL in these applications as well.
For what types of procedures have you seen the economic benefits of IVL?
I think that we are starting to appreciate the economic benefit of IVL throughout a number of peripheral applications. We feel that especially with large bore access for TAVR, EVAR, and TEVAR in aortoiliac occlusive disease, that the safety and efficacy of gaining sheath access is significantly increased with the use of IVL. For these large bore procedures, the number of expensive additional devices outside of IVL that need to be added has been significantly decreased in our practice. In the past, we used a variety
of different atherectomy devices to improve or change the compliance of the vessels. By eliminating any of the ancillary devices I feel that we have significantly reduced our cost, in some cases up to $10,000 depending on the challenges we face with that procedure. Outside of large bore access procedures, I would also say that we have felt an improvement in the safety and effectiveness in calcified PAD procedures, both above- and below-the-knee, where we have had very good results with the use of IVL within our practice. In the past, we used a variety of different atherectomy devices but these can be hard to deliver and have not always been as effective as we would like in severe calcium and, in particular, medial calcium. Furthermore, these devices, especially in the tibioperoneal vessels, have been associated with an increased risk of embolization. We have not yet seen these complications with IVL in the periphery.
We do not need long-term data to validate the benefit of that because these patients will certainly at least get what they need to stay out of the hospital for an extended period, avoid prolonged hospital exposure, and free up scarce resources. If we need to treat them again down the road, we will always have that option with IVL.
How can IVL impact resource utilization or hospital stay for inpatient procedures?

Does IVL have a particular role in the post-COVID era?
In general, over the past several months, patients appear to be presenting at a much later stage of the disease process with advanced CLI, worsening gangrene, and worsening tissue loss as they try to avoid the hospital and the risk of developing COVID-19 while in the hospital. It also seems that patients are presenting at a later time frame with regards to aneurysms, with us seeing more symptomatic and/or ruptured aneurysms and for which access has been quite challenging. When we have performed these procedures, we have been trying to minimize the need for open surgery as that will almost always result in increased hospital length of stay versus an endo approach and potentially tie up other vital resources, such as ventilators and ICU beds. IVL has allowed us to treat more complicated aneurysmal and PAD patients with an uncomplicated and efficient endovascular approach and get them out of the hospital as quickly as possible.
We are faced with a much more challenging group of patients who are presenting at a later time frame. Sometimes we have no other options but to do open surgery and unfortunately this includes some amputations with patients who have just presented and are too advanced. Most of these tend to be those individuals who have less access to care or, at the moment, are not going to the hospital out of fear of contracting COVID-19. However, in the group of patients who are salvageable, I would say that there is a shift towards minimizing the need for surgery and IVL has been a valuable tool to help us achieve that. We have also been presented with the challenge of rapidly decreasing our length of stay as well as ICU admissions to the best of our ability to try to save as many hospital beds during this pandemic as we possibly can. I do believe that this has allowed us to innovate rather quickly and efficiently, at least in the short term, to match the acute problems that patients are presenting with. Time will certainly be a great judge, but hopefully some of these changes that we have instituted will also work well in the longer term.
What do you see as the overall impact of incorporating IVL into your facility?
In our patient population, the amount of severe calcium that we can treat with IVL has been quite significant. This has limited the number of ancillary devices that we have had to utilize for these patients and allowed for efficiency and predictability in procedures. We believe IVL has effectively improved hospital length of stay and decreased overall cost for inpatient procedures. I would say that I do believe that these changes that we have seen during this pandemic are most likely going to be here to stay and will be a part of the new normal and so the role of IVL will only grow.
Frank Arko, MD, is chief of Vascular and Endovascular Surgery at Sanger Heart and Vascular Institute, Charlotte, North Carolina.
Frank Arko
Discussion
Fracturing intimal and medial calcium with IVL
In this discussion, George Adams and Andrew Holden consider the benefits of fracturing intimal and medial calcium with IVL, detailing promising early results and eagerly awaited forthcoming data.
Why are both intimal and medial calcium important?
Holden: Arterial calcification is common and increasing in prevalence as more patients with risk factors for vascular calcification are treated, including the elderly, diabetics, and patients with chronic renal insufficiency. Calcified arterial segments have reduced compliance and respond poorly to most endovascular treatment strategies, including angioplasty and stenting. Increased rates of acute recoil, dissection, perforation, embolization, and incomplete stent expansion are seen after endovascular treatment of calcified arterial lesions. In addition, severe calcium acts as a barrier to antiproliferative drugs coated on balloons and stents reaching deeper layers of the vessel wall. It is not surprising to note that arterial calcification is one of the biggest challenges to endovascular treatment success in peripheral arterial disease.
Arterial calcification may occur in both intimal and medial layers of the artery wall. Intimal calcification is a common manifestation of atherosclerosis and occurs in most arterial beds. The location of intimal calcification immediately adjacent to the flow lumen means this calcification may be modified by treatment modalities, such as cutting and scoring balloons, as well as atherectomy technologies. However, these technologies are unpredictable in their ability to effectively and safely modify this type of calcium. Conversely, medial calcification occurs more frequently in certain vascular locations and medical conditions, such as the tibial arteries in patients with diabetes or renal impairment. Arteries containing medial calcification have increased arterial stiffness, which drives poor endovascular outcomes, including dissection and recoil. Most endovascular devices do not modify medial calcification.
How does IVL work?
Holden: The Shockwave IVL system involves a non-sterile generator, connector cable, and sterile IVL catheter. The catheter contains an integrated, miniaturized array of lithotripsy emitters placed on the catheter shaft within an angioplasty balloon. The angioplasty balloon is inflated at the


treatment site at low pressure (typically four atmospheres) to provide balloon to vessel wall apposition. On activation, electrical discharges at the emitters vaporize the salinecontrast solution within the angioplasty balloon to produce bubbles, which expand and collapse. These bubbles create sonic pressure waves that travel through the vessel wall, producing a localized field effect to fracture both intimal and medial calcification, but leave soft tissues unharmed. The controlled disruption of intimal and medial calcium changes the compliance of the vessel wall, allowing subsequent dilatation of the vessel to nominal diameter at low pressure.
tomography (OCT). The high spatial resolution of OCT clearly demonstrates controlled microfractures and dissections in both the intimal and medial layers of calcium without the uncontrolled spiral dissections often seen after plain balloon angioplasty (Figure 1).
Can you detail the issue of BTK recoil and its potential relationship to calcium?
Adams: With the increased prevalence of an aging population, diabetes mellitus, and kidney disease, medial arterial calcium (Monckeberg’s medial sclerosis) is becoming more recognized as a significant challenge. Below-the-knee arteriosclerosis, typically found in critical limb ischemia (CLI) patients, is ridden with medial arterial calcium, making endovascular treatment difficult. We believe that medial calcification causes arterial stiffness leading to vessel recoil and ultimately restenosis. Anecdotally, recoil has been recognised by many peripheral interventionalists. Most notably, Baumann and colleagues evaluated elastic recoil
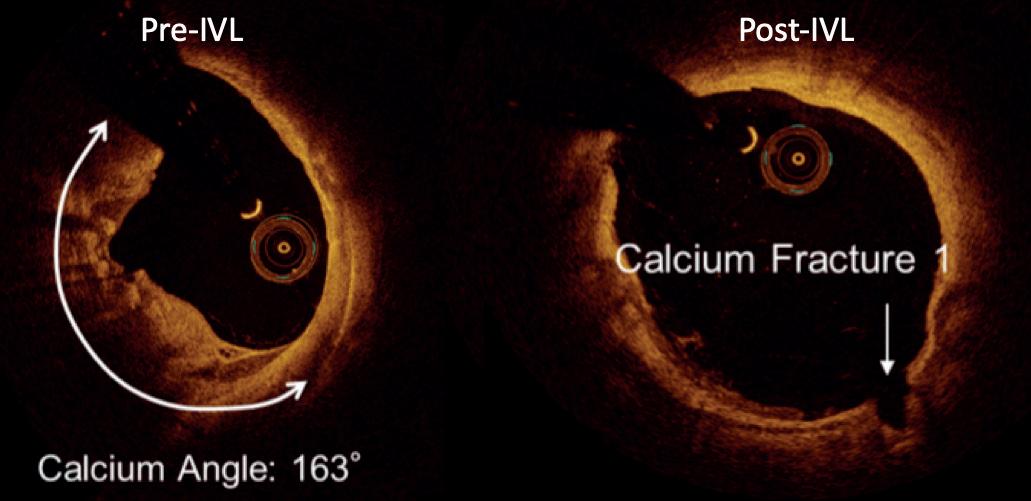
The phenomenon of low pressure dilatation of calcified vessels is a frequent and impressive finding after IVL. Confirmation of this controlled fracturing of vascular calcification has been obtained in preclinical cadaveric micro-CT studies. Clinically, this has also been demonstrated on intravascular ultrasound (IVUS) and optical coherence
Arterial calcification is common and increasing in prevalence.”
Continued
on page 4
George Adams Andrew Holden
Figure 1. OCT pre- and post-IVL
Discussion
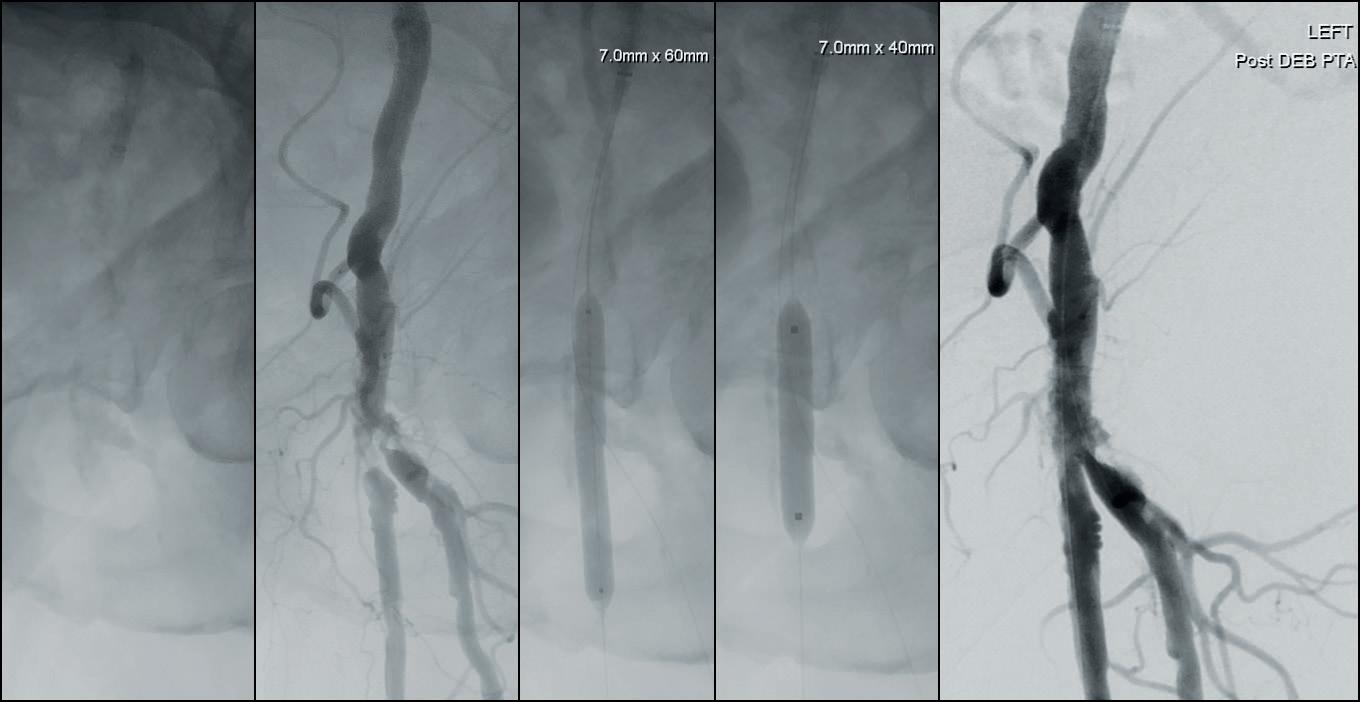
3. Treatment of a calcified stenosis of the left common femoral artery bifurcation with a 7.0x60mm IVL catheter, followed by a 7.0x40mm DCB
post-tibial balloon angioplasty and showed an angiographic mean luminal compromise of 29% only fifteen minutes post treatment (Baumann F, et al. Early recoil after balloon angioplasty of tibial artery obstructions in patients with critical limb ischemia. JEVT 2014; 21(1):44–51). This clearly remains a significant challenge in achieving acceptable luminal gain.
How have you evaluated recoil after IVL?
Adams: Intravascular ultrasound (IVUS) may be a more efficacious modality to evaluate recoil over two-dimensional angiography. An example of using this tool to evaluate recoil was in a 50-year old gentleman who suffered from diabetes, chronic kidney disease, and HTN. He had a nonhealing wound on his right great toe and forefoot. We placed a ruler on the outside of the leg to make sure we were accurate in comparing IVUS images. I intervened on the 120mm right anterior tibial artery with plain old balloon angioplasty (POBA).
Thirty minutes after POBA, luminal gain had decreased by 39.1%.
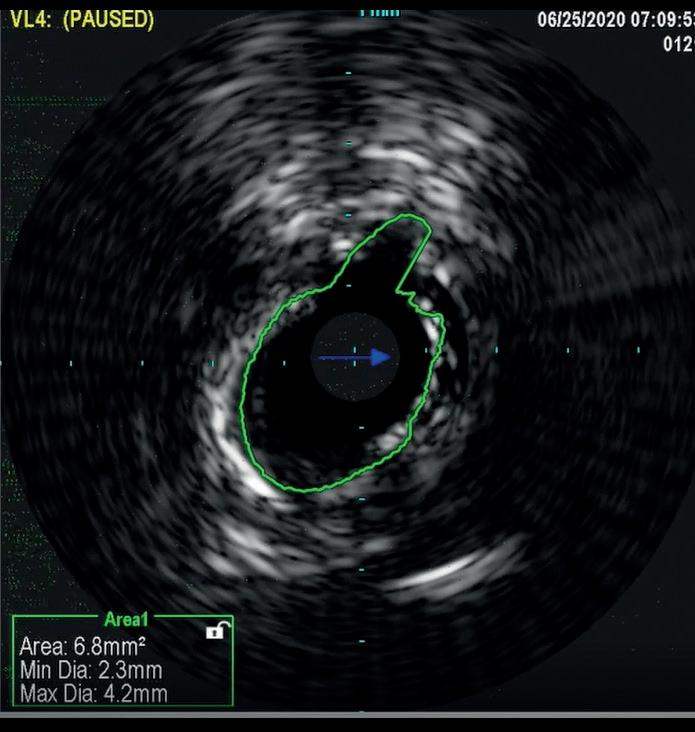
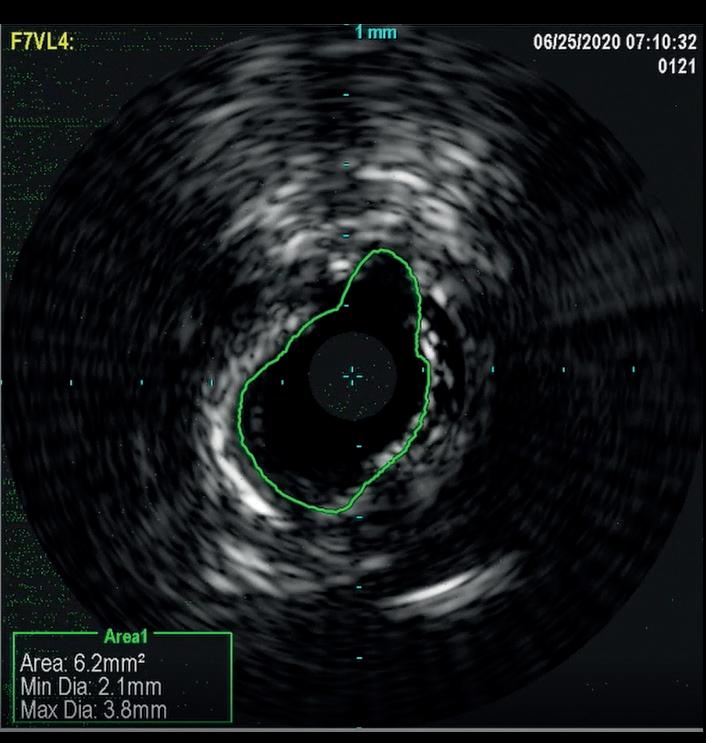
In a similar fashion, IVUS was used to evaluate elastic recoil following the use of IVL in a 61-year-old female patient with a history of diabetes, hypertension, high cholesterol, chronic kidney disease, and coronary artery disease. She presented with left lower extremity rest pain. Her baseline angiography showed two target areas with calcified stenotic disease that were treated (distal SFA and TPT). As shown in Figure 2, there was only an 8.8% loss of luminal gain in the max calcium portion of the TPT vessel 15 minutes after IVL treatment. Based on early and limited evaluations, IVL shows promising results with excellent luminal gain and with minimal luminal compromise, as evidenced on IVUS. Anecdotal cases have been encouraging and further studies are warranted to better understand if this mechanism of action provides the ability to ultimately produce significant luminal gain with minimal acute recoil. If this hypothesis holds true, this technology may lead to
meaningful reductions in the reintervention and amputation rates.
What are the broad applications of IVL technology?
Holden: The safety and acute effectiveness of Shockwave IVL has been shown in the DISRUPT PAD I & II trials in the femoropopliteal segment as well as the DISRUPT BTK trial in tibial arteries. These studies confirmed excellent luminal gain in heavily calcified arteries with a very low incidence of dissection and no safety concerns such as embolization. Longer-term follow-up revealed restenosis that was improved with optimal IVL energy delivery.
Since IVL does not address neointimal hyperplasia, it was hypothesized that the addition of a drug-coated balloon (DCB) plus IVL may have the potential to further improve long-term results in this challenging patient population. DISRUPT PAD III is a randomized study designed to assess that strategy. This trial has completed enrollment and results are eagerly awaited. Today, Shockwave IVL has been used to treat calcified peripheral arterial occlusive disease in a wide range of anatomic locations. A large observational registry has provided independent Core Laboratory adjudication of acute procedural outcomes and confirmed excellent safety and efficacy. Interestingly, IVL is usually followed by an adjunctive therapy but this differs depending on the anatomic location. In the femoral artery, IVL is most commonly followed by DCB angioplasty; in the iliac artery, IVL is usually followed by stenting. The aortoiliac segment has been an area of recent interest where IVL has been used to successfully dilate calcified iliac arteries to facilitate EVAR or in aortoiliac occlusive disease. The common femoral artery has also been a new and successful application for IVL (Figure 3).
Adams: In our practice, we treat a very high volume of CLI patients and we have used IVL technology to safely, efficiently, and effectively treat many difficult iliac, CFA, SFA, pop, and BTK lesions over the past several years. Now that Shockwave S4, the dedicated BTK device, is available, we have been able to treat even more challenging CLI patients than before.
Andrew Holden, MBChB, is director of Interventional Radiology at Auckland City Hospital, Auckland, New Zealand.
George Adams, MD, is director of Cardiovascular and Peripheral Vascular Research at Rex Hospital, Raleigh, North Carolina.
August 2020
Figure
Figure 2a. IVUS image of the most calcified aspect of TCT lesion immediately post-IVL
Figure 2b. IVUS image 15 minutes after IVL treatment
Case study
Minimal below-the-knee vessel recoil after IVL
In this case study, Peter Soukas demonstrates the benefits of IVL in a below-the-knee vessel, including decreased resource utilization, cost-effectiveness, and reduced length of hospital stay.
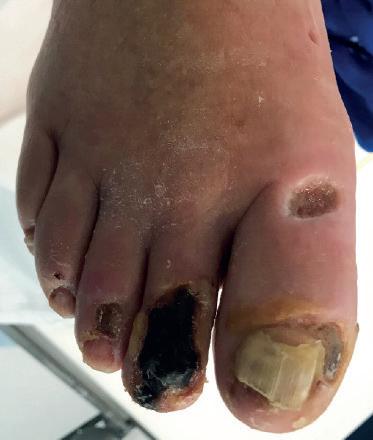
THE PATIENT IS A VIGOROUS 91year-old male with a past medical history of hypertension, hyperlipidaemia, and type II diabetes mellitus with resultant stage III chronic kidney disease. He is S/P coronary drug-eluting stent for symptomatic coronary artery disease and has a history of prostate cancer and prior right lower extremity deep vein thrombosis (DVT) complicated by postthrombotic syndrome with chronic edema. He developed ulcers on the right hallux as well as the second to fourth toes (Figure 1). A computed tomography angiography (CTA) of the lower extremity was notable for occlusion of all three infrapopliteal vessels. He was scheduled for a right below-the-knee amputation and came to see us for a second opinion. He was subsequently referred for angiography/intervention.
Ultrasound-guided access was obtained in the left common femoral artery and right lower extremity angiography confirmed nonobstructive inflow disease with tandem calcified 70 and 80% P1 as well as P2 extending into P3 disease. The anterior tibial
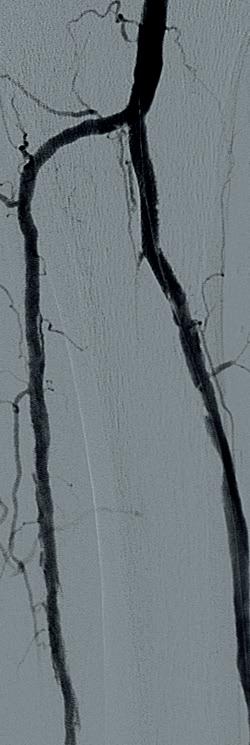

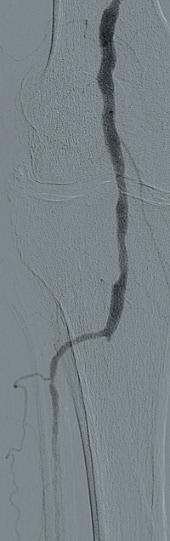
artery was noted to have a highgrade stenosis proximally with severe diffuse disease throughout its course, with essentially calcified occlusive disease distally with diffuse disease of the dorsalis pedis artery. The TP trunk was occluded proximally with complete occlusions of the TP trunk peroneal and posterior tibial vessels, with reconstitution of the posterior tibial artery above the ankle via geniculate collaterals. There was moderate diffuse disease of the plantar vessels as well (Figure 2a–c).

A 6F Ansel sheath (Cook Medical) was placed in the right CFA, and the anterior tibial occlusion was crossed with an Abbott Command 0.014” wire, followed by treatment of the popliteal lesions with a 6.0x60mm IVL catheter followed by drugcoated balloon (DCB) angioplasty with a Bard 6x150 Lutonix balloon, with reduction to a <30% residual. The right anterior tibial artery was treated with a 3.5x60mm

IVL catheter along a ~20cm segment of the lesion, followed by percutaneous transluminal angioplasty (PTA) with a 3x3.5x210mm Nanocross balloon (Medtronic), with an excellent result without recoil, despite the heavy calcific burden of disease. Antegrade traversal of the TP trunk and posterior tibial arteries was unsuccessful despite an escalating wire strategy, so
the posterior tibial artery was accessed in a retrograde manner with ultrasound guidance. The 0.014” Abbott Command wire was eventually delivered to the common femoral artery using a Quick-Cross 0.014” support catheter (Spectranetics) and exteriorised using a 7mm goose neck snare (Microvena). IVL of the TP trunk and proximal posterior (approximately 20cm of treatment length) tibial artery was performed with a 3.5x40 S4 IVL catheter in less than five minutes. This was followed by PTA distally with a tapered 3x3.5x210mm Nanocross balloon. A 4x38mm Medtronic Resolute Onyx DES was placed to treat a residual dissection in the TP trunk with final angiography demonstrating three-vessel runoff to the foot (Figure 3a–c). Remarkably, the anterior tibial IVL/PTA result showed no appreciable recoil, despite the fact that it took over 90 minutes to complete the TP trunk and posterior tibial reconstruction. He was discharged later that day. He later underwent second toe amputation and successfully healed his wounds.
This case illustrates the utility of IVL not only for superior acute luminal gain in calcified vessels, but also for the lack of recoil that is commonly seen with PTA. Despite the length and complexity of the multiple lesions, treatment required less than five minuted per catheter.
Reduced rates of dissection and recoil seen with IVL result in decreased resource utilization by limiting the need for bailout stenting. Unlike atherectomy devices that typically require the additional costs of embolic protection devices, IVL may be used as standalone therapy, or combined with DCBs.
Moreover, additional interventions for distal embolization or perforations seen by atherectomy are largely avoided with IVL, limiting those costs. In spite of often complex calcified disease, most patients treated with IVL are predictably discharged the same day.
Peter Soukas, MD, is director of Vascular Medicine at The Miriam and Rhode Island Hospitals, Providence, Rhode Island.
Peter Soukas
Figure 3a Figure 3b Figure 3c
Figure 1
Figure 2a
Figure 2b Figure 2c
Case study
IVL as a new endovascular option for calcified common femoral artery stenosis
Michael Lichtenberg discusses IVL as a new endovascular option for calcified femoral artery stenosis. Based on case experience, he suggests this “may be a viable option for patients who are not good surgical candidates,” adding that percutaneous intervention may improve hospital efficiency and cost-effectiveness when compared to a surgical approach.
CALCIFICATION IS A KEY underlying factor in common femoral artery (CFA) disease. Endarterectomy (CFE) is the standard of CFA stenosis and associated with good long-term patency. However, not all patients are candidates for CFE, as it is not a benign procedure in general and is also associated with extended length of stay.1
Endovascular interventions, including IVL, are growing in acceptance, as they have high technical success rates and can be offered as an alternative for high-risk patients with
comorbidities. Typical high-risk patients are those with histories of healing wound problems, severe obesity, or who have undergone prior CFA interventions.
was the only patent artery below the knee, as the anterior tibial artery showed a long occlusion from the proximal third of the vessel. Ankle brachial index (ABI) on the left side was 0.55. No wounds were present on the right extremity.
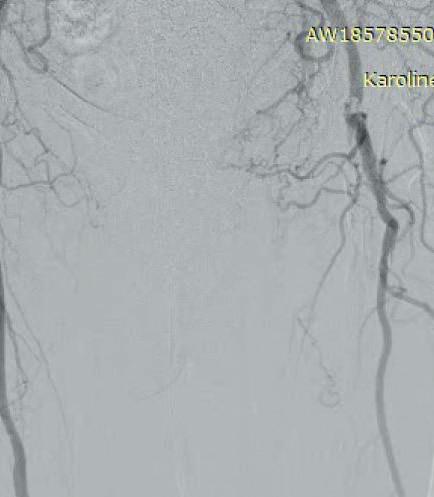
Subsequent diagnostic angiography confirmed the high grade left CFA stenosis (Figure 1) and the long SFA occlusion with diminished contrast flow below the knee. The deep femoral artery showed no significant stenosis within the first centimetres. For
In this case example, a 79-year-old man with severe claudication of the left leg was admitted to our angiology department. Cardiac risk factors included poorly controlled diabetes, renal insufficiency (GFR 27ml/min), severe obesity (body mass index [BMI] 38) and hyperlipoproteinaemia. On duplex ultrasound, a severe calcified short 90% CFA stenosis and a long 30cm occlusion of the superficial femoral artery (SFA) from the origin to popliteal artery (PA) segment one was diagnosed. The posterior tibial artery

DCB PTA catheter, BD Bard) which was inflated for three minutes at 8atm. Subsequent angiography showed a very good lumen gain without significant restenosis within the target lesion (Figures 3 and 4). We finalized the intervention with full recanalisation of the SFA using three more DCBs and one bailout stent implantation using the new triaxial, 4F Pulsar-18 T3 nitinol self-expanding stent system (6x150mm; Biotronik). After implantation of the Pulsar-18 T3 stent and a 6mm balloon post dilatation, a brisk flow was seen within the whole length of the target vessel and a straight flow into the foot via the patent posterior tibial artery. Follow-up examination of the patient the next day showed an ABI of 0.9 on the left side.
Based on our case experience and the existing literature, the conclusion could be made that IVL may be a viable option for patients that are not good surgical candidates.2 IVL does not prohibit future
recanalization, a crossover approach from the right groin was performed using an 8F crossover Destination reinforced introducer sheath (Terumo). Using an 0.014” Glidewire Advantage guidewire we were easily able to pass the high-grade CFA stenosis with the guidewire.
A 7x60mm IVL catheter was delivered across the lesion and the integrated balloon was expanded to 4atm to facilitate efficient energy transfer. After four calcium modification cycles, we used the integrated balloon to dilate the lesion at 6atm pressure in order to maximize luminal gain. The IVL catheter was then exchanged to an 8x40mm drug-coated balloon (DCB; Lutonix 035
surgical interventions if required and it may improve hospital efficiency and cost effectiveness with a reduced length of stay compared to surgical interventions.
References
1. Nguyen, Bao-Ngoc H, et al. Most complications of common femoral endarterectomy occur after hospital discharge. Journal of Vascular Surgery, Volume 58, Issue 4, 1152.
2. Brodmann M, Schwindt A, et al. Safety and feasibility of intravascular lithotripsy for treatment of common femoral artery stenoses. J Endovasc Ther, 2019 Jun;26(3):283–287.
Michael Lichtenberg, MD, is director of the Angiology Department at the Vascular Center Clinic Arnsberg, Germany.
Figure 1
Figure 2
Figure 3
Figure 4
Michael Lichtenberg
Case study
Simple IVL-facilitated EVAR access
Mazin Foteh discusses the clinical and economic value of IVL for endovascular aneurysm repair (EVAR), concluding that the technology has “revolutionized” his approach to aortic procedures.
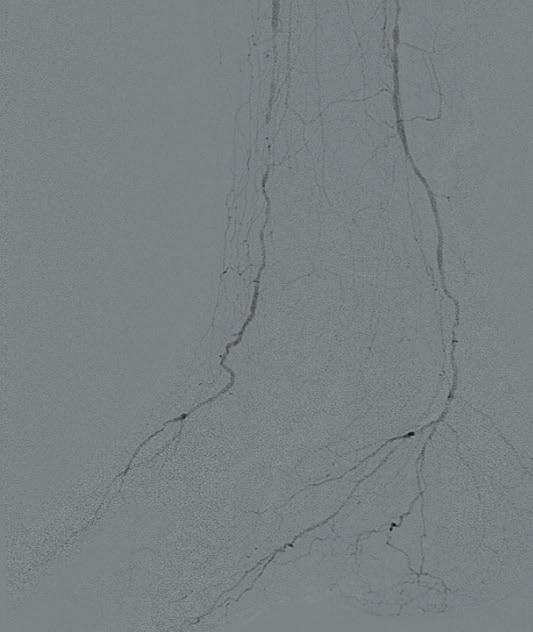
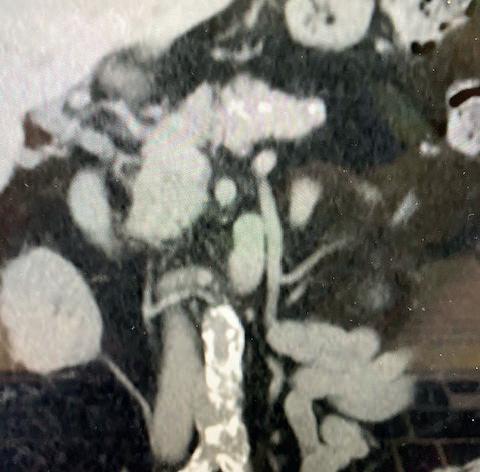
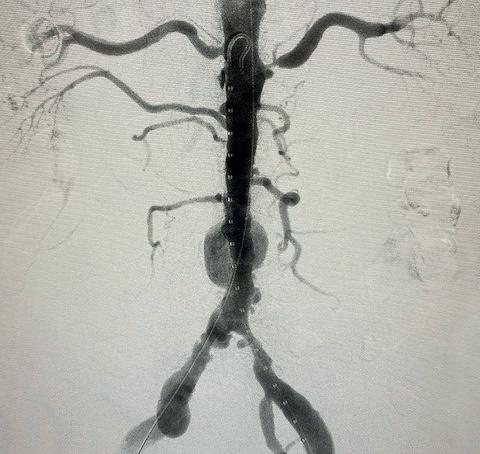
THE PATIENT IS AN 81-YEAR-OLD man with a past medical history of hypertension, hyperlipoproteinaemia, and remote history of smoking, who presents with an expanding saccular abdominal aortic aneurysm (AAA) and severe hip claudication. A preoperative computed tomography angiography (CTA) reveals dense circumferential calcified 90% stenosis of the bilateral common iliac arteries (Figures 1 and 2). In addition, the patient had a mid-aortic highly calcified stenosis. The patient desired a minimally invasive approach and given the underlying conditions, an EVAR was the best option to treat the AAA, as well as the bilateral common iliac artery stenosis.
Preoperative planning focused on three main areas of concern: the first is whether or not the stent graft could advance through the iliacs; secondly, would the stent graft limbs open to nominal diameter within the iliacs; and lastly, whether or not the stent graft would open fully within the AAA.
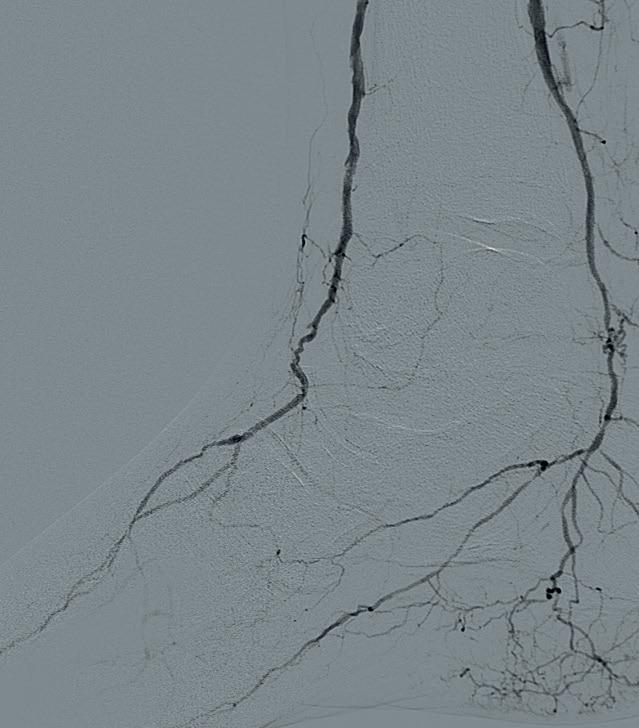
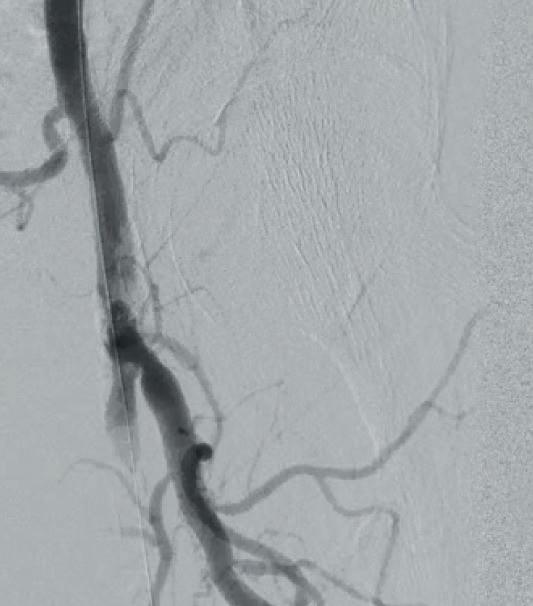
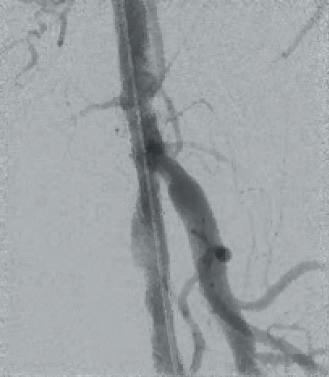
We used a percutaneous approach. Initially, we were unable to advance the 16Fr DrySeal sheath for the main graft on the right or the 12Fr DrySeal sheath on the left side for the contralateral limb (Figure 3). At this point, we performed IVL with a 7.0x60mm IVL catheter, of both the right and left common iliac arteries. We delivered 150 pulses to each vessel at 4atm and subsequently carefully dilated to nominal pressure of 6atm. Both arteries dilated easily. The Gore Excluder graft was delivered, and the limbs were placed within the common iliac arteries. Post-implantation balloon angioplasty was completed and revealed complete seal of the AAA and no residual stenosis in the iliacs (Figure 4). The patient was found to have a 90% stenosis of the left renal artery, which was treated with a balloon-expandable stent. His postprocedural CT scan revealed a very nice result, a sealed AAA, wide iliac arteries, and no residual stenosis.
From many aspects, this case had a number of pitfalls that would complicate a potential EVAR. The access was very difficult and, in the past, the options for advancement of devices would include predilatation with high pressure balloons, which carries a high rupture risk. Alternatively, we could create a short

2. Right
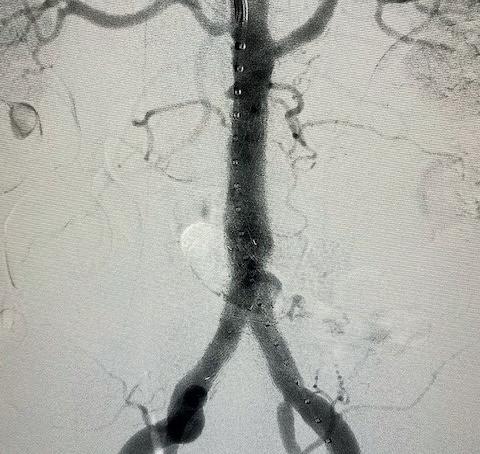

endoconduit by placing covered stents in the iliacs, but this adds tremendous cost and does not eliminate the risk of rupture. An open conduit is anatomically not an option and would not solve the issue with limited access. In an era of medicine in which cost has become omnipresent, cost effectiveness is now one of the very first considerations when designing a plan for complicated cases. IVL allows for the elimination of between $6,000 and $10,000 in additional cost.
For this patient, an open aortic reconstruction would have carried a much higher surgical risk, including a 5.5% risk of death. On a more intangible level, it would
have also led to a much more significant and costly hospital stay, a prolonged recovery, and much longer return to function. We must take all of these considerations into account and offer the best options for our patients.
IVL has been a wonderful addition to my practice and, in many ways, has revolutionized my approach to complex aortic procedures. In an effective and safe manner, we are now able to conquer calcified blockages. Where in the past we may have opted to only proceed with the most invasive operation, IVL creates new opportunities to the patients once deemed suitable only for open surgery.
Mazin Foteh, MD, is a vascular surgery specialist in Austin, Texas.
Figure 3. Initial intraoperative angiogram
Figure 1. Preoperative CTA revealing dense calcified stenosis of the iliac bifurcation
Figure 4. Completion arteriogram, areas of stenosis completely resolved
Figure
common iliac luminal diameter of 3mm, left common iliac diameter of 5mm
Mazin Foteh