From The Chair
Dear Alumni and Friends,
As we draw near the end of 2025, I am filled with pride for the extraordinary strides made by our Carolina Chemistry community, especially in a year filled with many changes and uncertainties for our academic enterprise. Our department continues to grow in strength in people, in discoveries, and in impact thanks to the hard work and commitment of faculty, students, staff, and alumni & friends, like you.
This year, we were delighted to welcome three new faculty members: Netz Arroyo, Lina Quan, and Karen Scida colleagues whose evolving research programs deepen our strengths in materials, energy, sensing, and interdisciplinary chemistry. Parallel to this growth, we celebrated welldeserved promotions for a record number of our established faculty, a recognition of their sustained excellence in research, education, and service that continues to elevate Carolina Chemistry.

Our community’s scientific creativity again delivered powerful, real-world impact in 2025. Our very own Frank Leibfarth earned the 2025 Blavatnik National Awards for Young Scientists, the first ever such Laureate from UNC-Chapel Hill. His work developing new catalysts and polymer-chemistry approaches to upcycle plastic waste and extract persistent “forever chemicals” (PFAS) from water — stands at the interface of molecular-level chemistry and an urgent environmental challenge. A cross-disciplinary team of UNC researchers won a $1 million grant from the W. M. Keck Foundation to “decode the secrets of AIdesigned proteins,” combining state-of-the-art biochemical tools with computational design to better understand protein behavior with implications in drug design. Our Sustainable Energy Research Consortium (SERC) has relaunched and celebrated multiple successes SERC is leading a build-up in battery testing and research facilities supported by the NC Collaboratory and a UNC Creativity Hub award. It also plays an integral role in enabling the Center for Hybrid Approaches in Solar Energy to Liquid Fuels (CHASE), which recently received a 5-year renewal. This Department of Energy research center, led by Carolina Chemistry, brings together a multi-disciplinary team to learn how to generate liquid fuels from sunlight.
The department continues to push the frontiers of sustainable chemistry, materials, catalysis, and more, building on the strengths and vision that have long defined Carolina Chemistry.
Behind every publication, grant, and award is our community faculty mentoring students, staff and students keeping labs humming, alumni supporting new initiatives, and friends cheering on success. The achievements of 2025 are collective, and they reflect our shared values of excellence, collaboration, and responsibility to society
Looking ahead to 2026, I am more confident than ever that we are laying the groundwork for an even more impactful future. As we seek to deepen interdisciplinary collaborations, expand opportunities for our students and post-docs, and continue addressing global challenges through chemistry, I invite each of you to stay engaged in conversation, collaboration, and support
Thank you for all you do to make Carolina Chemistry a community of innovation, curiosity, and caring. I wish you a restful and joyous holiday season, and a New Year full of discovery and possibility.
With warm regards,
Jim Cahoon Chair, Department of Chemistry
University of North Carolina at Chapel Hill


UNC Chemist and Savvy Startup Spark a High-Tech Assault on National Drug Trafficking
BY DAVE DEFUSCO

The MX908, developed by UNC chemist J Michael Ramsey and 908 Devices, Inc., combines thermal desorption, chemical ionization and tandem mass spectrometry to detect hundreds of analytes at trace levels including illicit drugs, such as fentanyl, explosives and other threats
Two decades ago at UNC-Chapel Hill, a chemist fiddled with ion traps and high-vacuum systems, nudged by a question: What would happen if arguably the most powerful chemical detection tool in the world mass spectrometry could be shrunk to fit in the palm of your hand? Today, the answer is saving lives.
The MX908, a handheld mass spectrometer developed by 908 Devices Inc., is now deployed across the United States to fight the fentanyl crisis and detect chemical threats at borders and in local communities. Its origin story begins not in Silicon Valley, but in the UNC Department of Chemistry with Professor J. Michael Ramsey, and reaches its commercial realization under a small group of founders at 908 Devices, including Dr Kevin Knopp, co-founder and CEO.
Ramsey, a National Academy of Engineering member and pioneer in microfluidics, recalls the first inklings of the idea while at Oak Ridge National Laboratory in the mid-1990s He had already launched one company Caliper Technologies that developed seminal microfluidics products and began asking a radical question: What else can we shrink? The answer was mass spectrometry.
Ramsey and his students and postdocs began experimenting with miniaturized ion traps, exploring their theoretical performance at pressures orders of magnitude higher than traditional systems. A postdoc’s accidental success operating an electron multiplier at higher pressures than he should have opened the door to what would become High Pressure Mass
Spectrometry (HPMS). When Ramsey moved to UNC in 2004, he found a welcoming environment for innovation.
“UNC was very supportive of spinouts,” he said. “It was easier to form a company here than at Oak Ridge.”

The commercial leap came when Ramsey reconnected with Knopp, a successful entrepreneur who was a co-founder of Ahura Scientific, which was sold to Thermo Fisher Scientific in 2010. The two had collaborated previously on an NIH project. “We already knew each other and shared common values, which is crucial for co-founders,” said Ramsey.
In 2012, they launched 908 Devices, and Knopp took the reins as CEO From the start, he said the UNC connection was key. “Before day one of 908 Devices, we were working with Mike Ramsey,” he said. “We looked at technologies from around the world. What we found in Mike’s lab at UNC was so unique, we built the company around it.”
According to Knopp, Ramsey’s lab wasn’t just the source of ideas but also people. “Mike had a prolific lab, and many of his postdocs and students came over to 908 Devices. I can think of at least six to 10 who transitioned over time,” he said.
Transforming an academic prototype into a rugged field device was no small feat. “We had to work on every component: pumping systems, electronics, packaging,” he said “Then we had to bring all those together in a battery-powered, field-ready form factor.”
The MX908 combines thermal desorption, chemical ionization and tandem mass spectrometry to detect hundreds of analytes at trace levels including illicit drugs, such as fentanyl, explosives and other threats. Users place a swabbed sample under a lid, where it is heated and analyzed. Results appear in under a minute. But what makes it revolutionary is the user interface.
“Most of our customers don’t even know there’s mass spectrometry in the box,” said Knopp. “They see a named compound and next-step guidance. It’s not squiggly lines, it’s actionable information.”
The MX908 is now deployed in over 55 countries and used by agencies ranging from the Texas Department of Public Safety to U.S. Customs and Border Protection, and even Romanian customs. “We have about 600 accounts worldwide and nearly 3,000 mass spectrometers in the field,” he said “It’s amazing to see this go from a lab idea to a global public safety tool.”
In Texas, where fentanyl deaths surged 158% from 2020 to 2023, the device has become essential. “It provides a lab-grade answer in the field,” he said. “Whether it’s a police officer, firefighter or mailroom worker, they need immediate, accurate results to make critical decisions.”
The partnership between 908 Devices and UNC remains strong. “The reason I was very willing to have this conversation is because of how amazing our 13-year partnership with UNC has been,” said Knopp. “From Chance Rainwater in UNC’s Office of Technology Commercialization to researchers like Mike Ramsey, it’s been an enabler of our entire journey.”
J. Michael Ramsey, Minnie N. Goldby Distinguished Professor, holds the precursor to the MX908, the M908.
Rainwater said the partnership brought together a rare mix of strengths that often don’t align in university spinouts: the scientific ingenuity and entrepreneurial drive of faculty founders like Ramsey, the business savvy and operational leadership of Knopp and the 908 Devices team, and the intellectual property guidance and support of the university’s commercialization office. He said the combination created a solid foundation not just for a successful launch, but for long-term impact.
“Mike Ramsey combines world-class science with a genuine commitment to solving real-world problems, and he understands that commercialization requires engagement with business leaders beyond the university,” said Rainwater “The 908 Devices team takes a collaborative approach with OTC to find solutions to issues related to UNC intellectual property rather than treating them as obstacles.”
Knopp believes university chemistry departments like UNC’s have a crucial role in national security. “Academic labs can tackle big ‘R’ research proving the hard things—while companies like ours handle the big ‘D’ development,” he said. “You need both.”
In March, 908 Devices sold its bioprocessing desktop portfolio to Repligen Corporation to focus entirely on its handheld platform. “We called it the launch of 908 2.0,” he said. “We now have four handheld products, including new optical spectroscopy tools, giving our customers a full toolkit.”
The mission is clear: equip frontline workers with fast, reliable and robust chemical analysis tools “The fentanyl crisis is a national emergency,” said Knopp. “But this extends to homeland and military threats, too. If fentanyl is aerosolized, it’s no longer just a drug problem. It’s a weapon.”
Looking ahead, Knopp sees continued growth.
“The need is enormous,” he said. “From trace gases to carcinogens affecting firefighters, we’re building tools that can protect lives right at the point of need.”
Knopp has a final message for UNC students and faculty eyeing the entrepreneurial path. “Be passionate. Understand the customer. Think beyond the lab,” he said. “Even when you’re working on the fundamental science, always ask: Where could this go? Who could it help?”

The MX908, a handheld mass spectrometer developed by 908 Devices Inc., reached its commercialization under CEO Kevin Knopp.
Netz Arroyo, who joined UNC in July after a highly successful tenure at Johns Hopkins University School of Medicine, is an expert in creating electrochemical biosensors— tiny, highly sensitive devices that can continuously monitor molecules in the body.

Imagine a tiny, wearable or implantable device that could track your body’s chemistry in real time, telling you whether your medication is working, if you’re low on a critical nutrient or even how your fitness routine is affecting your health. For Dr Netz Arroyo, the newest associate professor in Carolina’s Department of Chemistry, this is not science fiction, it’s his everyday work.
Arroyo, who joined UNC in July after a highly successful tenure at Johns Hopkins University School of Medicine, is an expert in creating electrochemical biosensors tiny, highly sensitive devices that can continuously monitor molecules in the body His research blends chemistry, engineering and biology to develop tools that could transform how we understand, track and improve health.
New UNC Professor Develops Wearable Devices Monitoring the Body’s Chemistry
BY DAVE DEFUSCO
“We want to make it possible for people to get detailed, real-time information about their bodies,” said Arroyo. “That information can empower better decisions about diet, exercise, treatments really, everything that affects health.”
The Arroyo Lab and his team design sensors that mimic the body’s natural ability to detect specific molecules. These devices can measure important compounds like drugs, proteins and other chemical signals without needing a blood draw or trip to the lab.
The potential applications are wideranging:
Better medication management by showing exactly how much of a drug is in the body at any moment. Personalized health monitoring, with wearable devices that give a detailed chemical snapshot of your well-being.
Smarter drug delivery systems that respond automatically to changes in the body.
of biosensors and DNA-based devices that can detect proteins and genetic material with incredible precision.
Beyond his research, Arroyo is committed to building a diverse and welcoming scientific community. His lab actively works to ensure that people from all backgrounds have access to research opportunities and that every voice is respected.

The Arroyo Lab shows off the pumpkins that they carved for Halloween.
His group’s recent work has even revealed surprising details about how medicines spread through the brain, and developed new tools for tracking HIV treatment and cancer biomarkers in real time.
Arroyo’s impact isn’t limited to the lab bench. In 2025, he became interim editor-in-chief of ECS Sensors Plus, an international journal that publishes cutting-edge research on sensing technologies. He has been recognized as a “Rising Star in Sensing” by ACS Sensors and has earned awards for both research and teaching, including Johns Hopkins’ Cecil H. Robinson Teaching Award in 2024.
His scientific reach spans collaborations on everything from COVID-19 antibody tracking to new materials for improving sensor performance. In the past two years alone, he’s co-authored studies on brain drug mapping, rapid prototyping
“Creativity and innovation come from diversity,” he said. “Different perspectives are what allow us to solve hard problems in new ways ”
Arroyo earned his undergraduate degree in chemical sciences from Tecnológico de Monterrey in Mexico, his Ph.D. in analytical chemistry from the University of Texas at Austin, and completed a postdoctoral fellowship at the University of California, Santa Barbara. He joined the faculty at Johns Hopkins in 2019 and quickly built a reputation as a creative researcher and inspiring teacher before making the move to Chapel Hill.
As he settles into his new role at UNC, Arroyo is looking forward to expanding his research and collaborations. His goal is clear: to create technologies that bring laboratory-level chemical analysis into everyday life.
“For patients, athletes, doctors and anyone interested in understanding their own biology,” he said, “the future as I envision it is one where chemistry quietly works in the background, helping people live healthier, more informed lives.”
Next Time You Crack Open a Cold One, Forever Chemicals May Be Lurking, Researcher Warns
BY DAVE DEFUSCO
When you crack open a cold beer, you might think about hops, barley or yeast, but probably not “forever chemicals ” Yet these synthetic substances, known as PFAS (per- and polyfluoroalkyl substances), may be hiding in your drink, and Carolina scientists are figuring out how to detect them.
PFAS are a large family of chemicals prized by manufacturers for being water- and oil-resistant. They’ve been used in everything from nonstick pans to waterproof jackets. The downside is that PFAS don’t easily break down in nature or in our bodies. Over time, they’ve spread into drinking water, soil and even rainfall. Studies have linked them to serious health problems, including high cholesterol, weakened immune systems, certain cancers and low birth weight.
“More than 99% of people have PFAS in their blood,” said Ashlee Falls, a Ph.D. student in the Baker Lab in the Department of Chemistry. “That means our exposure is happening in more ways than we realize, and beverages like beer could be one of those pathways.”
Falls recently presented her work at the American Society for Mass Spectrometry (ASMS) conference, focusing on how to accurately measure PFAS in beer brewed around the world. Her project aims to optimize methods for extracting and quantifying PFAS so researchers and brewers can better understand how much of these chemicals are making it from water and ingredients into the final product.

Ashlee Falls, a Ph.D. student in the Baker Lab, is focusing on how to optimize methods for extracting and quantifying forever chemicals in beer so that researchers and brewers can better understand how much of these chemicals are making it from water and ingredients into the final product
“Ashlee is very interested in water quality and how water contamination affects human health,” said Dr. Erin Baker, an associate professor and Falls’ Ph.D. advisor. “Her desire to increase health and help brewers create the best products possible inspired Ashlee to design the PFAS beer study and push through all the measurement difficulties she has encountered to date.”

Dr. Erin Baker, an associate professor in the Department of Chemistry, is Ashlee Falls’ advisor
Beer is mostly water, or about 95% by volume. If local water supplies contain PFAS, there’s a good chance they could make it into your pint. The challenge is that most breweries don’t currently test for PFAS, and current methods are not optimized for dark beers like porters or stouts.
Testing is further complicated by beer’s chemistry Unlike plain drinking water, beer contains carbonation, proteins, sugars and other compounds from grains, hops and yeast. These extras can get in the way of detecting PFAS in the lab.
“Beer is a really complex liquid,” said Falls. “When you’re trying to measure tiny amounts of PFAS, all those other ingredients can interfere with the analysis.”

Falls demonstrates her techniques for testing beer.
To tackle the problem, Falls tested three variations of a lab process called QuEChERS, pronounced catchers, a method widely used to extract pesticides from food. She used special chemical cartridges designed to remove fats, sugars or both from beer before analyzing it. The idea was to clean up the beer sample just enough so that any PFAS present could be detected using high-tech approaches, such as combining liquid chromatography, ion mobility spectrometry and mass spectrometry (LC-IMS-MS) separations.
In her trials, she first spiked beer samples with known amounts of PFAS to see how much she could recover. The results weren’t encouraging;
current methods either didn’t remove those pesky “other ingredients” or they did remove them along with the PFAS. This meant that for the PFAS put into the beer at the beginning, only about 10-15% was observable at the end, far below the ideal 100% recovery rate.
The takeaway is that the usual tools for extracting PFAS from foods and water don’t work well for beer. “Our current methods just aren’t efficient enough,” said Falls. “We need to keep refining them.”
Falls and her colleagues are now testing a new solid phase extraction cartridge designed specifically for pulling PFAS out of complex liquids. Using an automated system, they hope to boost recovery rates to at least 75%, enough to reliably detect PFAS at very low concentrations.
Once the method works well, the team plans to analyze a wide range of beers lagers, ales, sours and more from breweries in North Carolina and around the world. That data will help them figure out how PFAS levels vary by region; whether the contamination comes mainly from water, grains or hops; and how brewing practices might influence PFAS levels. The ultimate goal is practical: give brewers an easy, low-cost way to test for PFAS and reassure consumers about what’s in their beer.
While most people think of PFAS as a problem in tap water, Falls says beverages like beer are part of the bigger picture of everyday exposure.
“Brewers take pride in the quality of their ingredients and the taste of their beer,” she said. “If PFAS are making their way into the brewing process, that’s something both brewers and consumers deserve to know.”
Her work could also help public health researchers map PFAS contamination worldwide, using beer and other drinks as a snapshot of local water quality.
“We want to make PFAS testing more accessible,” she said “Whether it’s beer, soda or spirits, our methods could give communities another tool for understanding and eventually reducing their exposure to these chemicals.”
In Redesigned Chemistry Course, Students Turn Pigments into Art Preservation Tools
BY DAVE DEFUSCO

Dr. Jade Fostvedt, standing second from right, a teaching assistant professor, has created a new course, “Chemistry, Conservation, and CURE: Pigments and Dyes of Antiquity,” and its goal is as colorful as its subject matter: give undergraduates a real-world research experience while contributing to the preservation of cultural heritage at UNC’s Ackland Art Museum. Beside her, standing left to right, are recently graduated chemistry students: Emerson Woolwine, Torrance (Tori) Jenkins and Dylan McEldowney, and kneeling, Philip Garside and Lilly Gornto.
In the UNC Chemistry Department, a new kind of experiment is taking shape one that melds the rigor of synthetic chemistry with the beauty of art conservation. At the heart of this innovation is Dr. Jade Fostvedt, a teaching assistant professor who has spent the past year redesigning the department’s senior capstone course, CHEM 550L: Synthetic Chemistry Laboratory, into a deeply interdisciplinary, research-driven experience.
The new course, “Chemistry, Conservation, and CURE: Pigments and Dyes of Antiquity,” was launched in Spring 2025 with a record enrollment of 71 chemistry majors. And its goal is as colorful as its subject matter: give undergraduates a realworld research experience while contributing to the preservation of cultural heritage at UNC’s Ackland Art Museum.
“The interface between chemistry and art is rich in connections,” said Fostvedt. “We want students to discover those connections not just in the lab, but while walking the museum galleries, thinking about history, culture and the chemistry that holds it all together.”

Cameron Grant, a recent graduate, is adding sodium hydroxide to a mixture of 2nitrobenzaldehyde and acetone to make indigo dye using a classic chemical method called the BaeyerDrewsen synthesis.
CHEM 550L has long served as a foundational course for senior chemistry majors an opportunity to synthesize knowledge through independent research. But until now, the CURE (Course-based Undergraduate Research Experience) component had remained largely unchanged since its inception in 2019. Students worked on catalyst design, a useful but isolated task.
Fostvedt saw the chance for something more dynamic. She partnered with Dr. Michel Gagné, a synthetic chemist; Dr. Kathleen Nevins and Dr. Tyler Motley, both lab curriculum specialists; and experts from the Ackland Art Museum to build a new vision That vision was supported with funding from the UNC System Undergraduate Research Program Award, making possible a full revamp of CHEM 550L into a living, evolving, multidisciplinary course.



In the first half of the semester, students synthesized five historical and culturally significant blue pigments Prussian blue, phthalo blue, smalt, indigo and the modern YInMn blue. These vibrant compounds were chosen not just for their Carolina-blue relevance but for their deep roots in both ancient and contemporary art traditions.
Students learned to quantitatively analyze color, determine what gives molecules their hues and investigate synthetic routes to both ancient and never-before-seen pigments. “These pigments span the full range of chemistry,” said Fostvedt, “from organic to inorganic compounds and even solid-state materials, which is a new frontier for our department.”
As an offshoot of the course, senior chemistry major Clara Lord collaborated with Fostvedt and Grace White, paper conservator at the Ackland Art Museum, to use infrared photography and spectral imaging to analyze unknown pigments in artworks from the Ackland. The museum, whose collection is more than 80% works on paper, had previously only analyzed a small fraction of its collection due to resource limitations.



Now, thanks to this partnership, students contribute directly to conservation science, helping identify unstable pigments and understand their long-term behavior critical for preserving delicate paper-based artworks vulnerable to light, air and time.
The second half of the course challenged students to take their work to the next level. Working in teams of four, they designed original research questions, including: How does the choice of copper source influence the color and structure of Egyptian blue pigments? Can organic-inorganic
Michel Gagné Kathleen Nevins
Tyler Motley
Clara Lord Jade Fostvedt Grace White
hybrid pigments like Maya blue be adapted to produce other colors? How do electronwithdrawing or -donating groups alter the color of indigo-based dyes?
With support from Dr. Josh Chen of the Chemistry X-Ray Core Lab and Dr. Bob Geil of the Chapel Hill Analytical and Nanofabrication Laboratory, students accessed advanced instrumentation, like powder X-ray diffraction and energy-dispersive Xray spectroscopy, to analyze their creations and trace elements in museum samples.



For many students, it was their first time conducting truly independent research and the experience was transformative. Being able to see their pigments showcased in artworks the Ackland’s collection and then to have their own lab-synthesized creations on display made them feel like their work mattered. It wasn’t just a class; it was a contribution.
Beyond the technical skills, Fostvedt’s course encourages belonging. By using art a subject that connects deeply with culture and identity—she hopes to draw a more diverse array of students into chemistry and keep them there.
“We want students to see themselves in the science,” she said. “Whether that means studying pigments from indigenous traditions or exploring dyes linked to their own heritage, art allows us to personalize the lab in a way traditional experiments don’t ”
It’s part of a larger movement in STEM to not only teach content but to cultivate inclusion. Research shows that when students can connect their
learning to personal or cultural experiences, their engagement and retention in the field increases.
Dr. Elizabeth Manekin, head of university programs and academic projects at the Ackland, is already curating exhibitions to support future classes, and the museum has plans to showcase student findings in upcoming displays a literal gallery of student scholarship.
“Through our collaboration,” said Fostvedt, “we’re not just training chemists We’re training thoughtful scientists who can communicate across disciplines, solve complex problems and recognize the value of their work in the world around them.”







Josh Chen
Bob Geil Elizabeth Manekin
Professors Help Flip the Script on Chemistry Education with Atoms-First Textbook
When Carribeth Bliem first stepped into a lecture hall of first-year chemistry students at the University of North Carolina at Chapel Hill, she didn’t know she would one day help write a textbook that would be used nationwide What she did know deeply and instinctively was that chemistry wasn’t just about equations and reactions. It was about showing students that the discipline touches every corner of their lives.
“I really like teaching first-years,” said Bliem, a teaching associate professor in the Department of Chemistry. “Convincing them that chemistry impacts their daily lives, that’s the heart of it. After years of adapting the pedagogy we use at UNC for large-enrollment classes, the book was just the next logical step. Honestly, it felt like kismet.”
Her co-author, Anna Curtis, also a teaching associate professor, shares that same instinct for asking “why?” and not just in science. Curtis began teaching at UNC in 2019 after earning both her undergraduate degree there and her Ph.D. at the University of Colorado Boulder. Early on, she found herself questioning the traditional sequencing of general chemistry topics.
“I was teaching with a colleague for the first time, running through the traditional order, and she asked me, ‘Why do we teach it this way?’” said Curtis. “If our goal is to help students solve problems in new contexts, not just memorize, we have to start with the fundamentals the why behind what they’re learning. That’s where the atoms-focused approach comes in ”
Traditionally, general chemistry courses begin with broad concepts like stoichiometry and reactions,
BY DAVE DEFUSCO
only later diving into the atomic and molecular principles that underpin them. The atoms-focused approach flips that order, starting with electrons, nuclei and bonds, then building up to the macroscopic world.
“When you tell students to memorize the charges on common cations in chapter two, but you don’t explain why until chapter seven, it’s frustrating and counterproductive,” said Bliem “In the atomsfocused order, we teach the why first.”

Bliem and Curtis were thrilled when the department adopted the previous edition of the textbook in Spring 2022, though they never imagined becoming part of the author team That summer, while developing course materials for the new edition, they met Norton’s editor at a chemistry education conference and offered suggestions for enhancing Chemistry: An AtomsFocused Approach, 3rd Edition, drawing on their expertise as both physical chemists and educators.
For example, one theme Bliem and Curtis emphasize throughout their courses is the central role of energy in shaping chemical intuition about atoms. When the editor invited them to audition as co-authors, they demonstrated how introducing energy concepts earlier could strengthen and expand the book’s opening chapters.
“Even if nothing came of the audition process, I’d at least shared ideas I think are essential for student growth,” said Bliem. “But it all fell into place.
When the editor invited Carribeth Bliem, standing, and Anna Curtis to audition as co-authors, they demonstrated how introducing energy concepts earlier could strengthen and expand the book’s opening chapters

Both professors structured, active-learning style a method that engages students in problem-solving rather than passive note-taking. Translating that into a textbook meant making it more than a static reference. The fourth edition of Chemistry: An Atoms-Focused Approach introduces two signature features:
Stepwise Figures, formulated by Bliem, break complex visuals into smaller, ordered steps, modeling for students how to demystify visuals by asking guiding questions at each stage. ChemTours, developed by Curtis, use sequential questions paired with interactive visuals and animations to guide students toward a deeper understanding of course concepts
Stepwise Figures and ChemTours: Guided Inquiry are found within Norton’s learning system, which
includes Smartwork online homework and an interactive e-text. In Smartwork, instructors can search a vast question bank by topic, learning objective or Bloom’s taxonomy level or use premade assignments, adaptive modules and mathprep sets to meet students where they are. The result is a fully integrated learning system textbook, modules, activities and assessments all aligned in art and vocabulary.
“That consistency is vital for novice learners,” said Curtis. “As chemists, we can say the same thing five different ways and know we mean the same thing. But to a student, that’s five different concepts. Consistency removes that barrier.”
When students can recognize themselves and their career aspirations in course materials, they are more motivated to learn. To support this, the text highlights scientists from diverse backgrounds and showcases a wide range of career paths. The cover of the fourth edition features Professor Sonja Salmon and a member of her research team, Siyan Wang, who developed a textile recycling method at North Carolina State University. Real-world contexts, from textile chemistry to environmental science, help students connect classroom learning to their daily lives and future professions.
In many ways, Bliem and Curtis’s work on the new textbook is a love letter to teaching chemistry blending curiosity, rigor and inclusivity drawn from more than 30 years of experience and over 15,000 students. They credit their co-authors, Tom Gilbert and Stacey Lowery Bretz, whose expertise and collaboration were essential in bringing their shared vision for the edition to life.
“We want to give students the tools to think critically, solve problems and connect chemistry to their daily lives,” said Bliem, “and we want them to believe they can do it.”
Curtis agrees. “With a growth mindset and the right learning environment, all students are capable of learning chemistry. That’s the foundation of everything we do.”
Each holiday season, UNC chemist Erin Baker and her lab trade data analysis for presents rallying fellow Tar Heels to raise money for Toys for Tots and Coats for Kids. Their efforts show that science isn’t their only way of making an impact in the community.
“I know many families struggle just to cover everyday expenses, and the holidays bring even more financial and emotional stress,” said Baker. “If we can help even in a small way by providing a toy, I know we are making a meaningful difference in a child’s life and the families’ holiday memories.”

The Baker Lab of UNC Chemistry during their annual charity shopping trip.
Baker Lab of UNC Chemistry
Calls for Donations for Toys and Coats for Kids
BY AMIE SOLOSKY
This year, the Baker Lab went on its big shopping spree for coats and toys on November 24 . th
Another way you can help is by donating to the Toys for Tots box directly. The lab will have a box stationed outside their office, Caudill 010, where students, faculty and staff can drop off new, unwrapped toys of their choosing anytime between 9 a.m.–5 p.m. from November 27 to December 8.
Toys for Tots is hosted by the Marine Corps Reserve and provides support to disadvantaged children during the holiday season, primarily through organizing toy donations. According to the organization’s website, Toys for Tots has distributed over 708 million toys since 1947.
Coats for the Children, a collaboration with The Salvation Army and WRAL, distributes coats, hats and other warm clothing to families in need. This drive began in 1989 and has collected over 176,650 coats and more than $2 million to help clothe local children.


Last year, the lab raised $2,000 and split the money raised in half between coats and toys. This year, the team is hoping to raise even more by adding the Toys for Toys box in Caudill Labs

“We want to give people the ability to donate toys they picked out and also make it really easy for the department to give,” said Baker. “Coming together for this cause is also so important since buying in bulk helps us secure better discounts from the stores and allows us to reach and support even more families in need.”






Photo of the toys and coats donated in 2023.
Photos from previous years.
UNC Chemistry Department Welcomes Three New Faculty Members
BY DAVE DEFUSCO

The UNC Department of Chemistry is proud to welcome three outstanding new faculty members whose expertise and energy promise to enhance our vibrant scientific community: Dr. Netz Arroyo, Dr. Lina Quan, and Dr. Karen Scida. These new additions bring diverse research strengths and a shared commitment to mentoring, innovation, and collaborative discovery.

Professor Netz Arroyo joins UNC Chemistry’s faculty from the Johns Hopkins University School of Medicine, where as an Associate Professor he led a highly regarded research program focused on the development of biology-inspired electrochemical sensors that support real-time, continuous measurements of a wide range of physiologically-important molecules in vivo. His interdisciplinary approach bridges analytical chemistry, biophysics, and bioengineering, with broad implications for diagnostics and personalized medicine. Dr. Arroyo’s pioneering work in wearable and implantable sensors has already made a significant impact on this field of research, and we are excited to see how his innovative and mentoring spirit will enrich UNC’s exploratory landscape.
Professor Lina Quan comes to Chapel Hill Virginia Tech, where she led her lab as an Assistant Professor. Her research centers on the optical and electronic properties of emerging semiconductors such as perovskites to control the absorption or emission of light and/or manipulate the transport and recombination of energized charge carriers in these materials. With expertise in techniques such as ultrafast spectroscopy and materials synthesis chemistry, Dr. Quan is poised to help expand UNC’s initiatives in energy, sustainability, and health. Her creative and interdisciplinary approach will contribute meaningfully to both research and graduate training.


Research Associate Professor Karen Scida arrives to us from the Lieber Institute for Brain Development, where she made notable contributions to analytical chemistry, its applications, as well as integration to microfluidic and biological systems. Her research at the Institute focused on the development of in vitro and in vivo neurotransmitter-specific methods to evaluate the performance of the drugs created to understand and treat schizophrenia As a Research Associate Professor, Dr Scida will bring a dynamic perspective to UNC Chemistry, with plans to explore new methods of chemical discovery and therapeutic intervention.
We are thrilled to have them join our department. Their arrival reflects UNC Chemistry’s commitment to scientific excellence, diversity of thought, and impactful research that addresses society’s most pressing challenges.
Hill!
Katelyn Kitzinger, who earned a Ph.D. in organic chemistry, holds aloft a 3D molecular model of diethyl ether, a 19th-century chemical that was once the world’s most common anesthetic. She joined a team of UNC researchers in chemistry and medicine to explore an unexpected question: Could doctors make their own anesthetic in the field using simple materials?
BY DAVE DEFUSCO
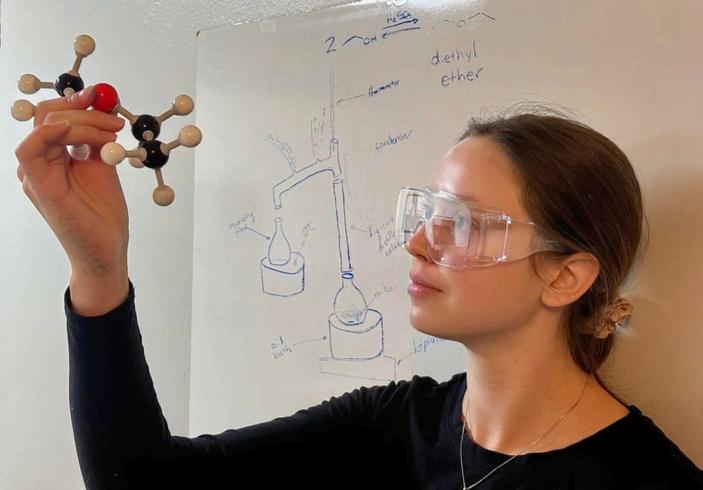
Katelyn Kitzinger Helps Revive an Old Anesthetic for Modern Emergencies
When earthquakes, wars or hurricanes devastate communities, hospitals can disappear overnight. Doctors may have no electricity, few medicines and no safe way to put patients to sleep for surgery In those moments, something as routine as an appendectomy can become impossible.
That urgent problem led Katelyn Kitzinger, who holds a Ph.D. in organic chemistry from UNCChapel Hill, to join a team exploring an unexpected question: Could doctors make their own anesthetic in the field using simple materials? The answer, she and her colleagues found, might lie in a 19th-century chemical called diethyl ether, once the world’s most common anesthetic Ether was replaced long ago by safer, less flammable drugs, but it has two big advantages in a crisis: it works and it’s easy to make.
“It’s one of the simplest anesthetics ever used,” said Kitzinger, now a senior scientist at AbbVie, a global pharmaceutical company. “Chemically, it’s made from alcohol. If you can produce it safely, it could be a lifesaver where modern anesthetics just can’t reach.”
Her study, “Is Small-scale Production of Diethyl Ether for Anesthetic Use Possible?” published in the Journal of Special Operations Medicine, set out to prove that point with the design of a small-scale experiment using materials that might be available in almost any country.
A key figure behind the project was Sandeep Dhanjal, an assistant professor of anesthesiology in the UNC School of Medicine who focuses on global health. He approached Kitzinger and Jeffrey Johnson, A. Ronald Gallant Distinguished Professor, with a pressing question: how could doctors in resource-limited or disaster-stricken settings safely produce anesthetics when commercial supplies were unavailable?
Sandeep Dhanjal, an assistant professor of anesthesiology in the UNC School of Medicine, whose vision first brought the idea to the chemistry team.

Acting as both the catalyst and clinical partner, Dhanjal worked closely with Kitzinger to run the experiments after Johnson’s group explored the chemical feasibility Their collaboration became the project’s defining strength, showing how people from very different parts of the university could combine their expertise to tackle a realworld humanitarian challenge.
The idea was straightforward: start with ethanol the same type of alcohol found in hand sanitizer or spirits— and add sulfuric acid, a common chemical that’s found in some household drain cleaners. Heat the mixture to about 145 degrees Celsius, or 290 degrees Fahrenheit, and a chemical reaction occurs that transforms some of the alcohol into diethyl ether and water.
The next challenge was to separate and purify the ether so that it could be safely used as an anesthetic. “The tricky part is that diethyl ether evaporates very quickly,” said Kitzinger. “If you leave it sitting out, it’ll be gone in minutes ”
To keep it from disappearing, the team used a simple setup: a glass flask connected to a cooling tube that collected the ether vapors into a second container chilled in an ice bath. “That’s how we kept it from escaping,” she said. “It’s not high-tech. You could imagine doing something similar in a basic lab setup ”
Kitzinger draws a diagram of the simple setup.
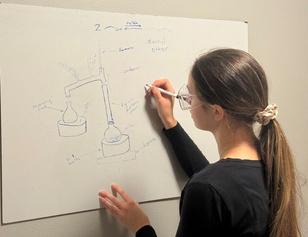
When the team analyzed what they’d made, they found that the sample was 97% pure ether, with the remaining 3% being leftover ethanol. “We were happy to see that the impurity was just ethanol,” said Kitzinger. “It’s something that’s already safe for the body, so we weren’t introducing anything dangerous.”
To confirm what they had made, Kitzinger used a tool called proton nuclear magnetic resonance spectroscopy, or proton NMR This method works a bit like fingerprinting; it produces a unique pattern for each chemical. “Every molecule has its own signature,” she said. “When we saw the pattern for ether, we knew exactly what we had.” Still, ether’s flammability poses a real risk, especially in field conditions. Kitzinger said the way to minimize that danger is by keeping the apparatus sealed, cooling the collection container and avoiding open flames “It’s about containment,” she said “If you can prevent vapors from getting into the air, you greatly reduce the fire hazard.”
For Kitzinger, the project was a reminder of why she became a scientist. “It was really exciting to take something I learned in an undergraduate chemistry class and use it to address a humanitarian problem,” she said. “I got into science because I wanted to make a positive impact, and this felt like one of those rare projects that connects chemistry directly to helping people.”
She credits Johnson, her graduate mentor, with nurturing that mindset. “He was a great example of what it means to be a good scientist curious, encouraging and willing to take on tough questions,” she said “He gave me the freedom to explore new ideas, even if they seemed a little unconventional.”

Today, Kitzinger applies that same curiosity and approach to problem-solving in AbbVie’s process chemistry department, where her team develops large-scale synthetic routes for chemicals of pharmaceutical importance. Their focus is on making these reactions safe, efficient and reproducible while ensuring the highest product purity. To do that, they rely on a wide range of analytical tools and reaction-monitoring techniques.
“Scaling up a reaction, from a small vial in the lab to a large reactor in a manufacturing plant, can change its behavior in surprising ways,” said Kitzinger, “and part of my job is figuring out why. Understanding how molecules transform from starting materials to final products is critical, and we build that knowledge through both experimental work and insights drawn from the scientific literature.”
As for the future of ether production in crisis medicine, Kitzinger hopes others will build on her proof-of-concept study to test safer equipment, find better starting materials and develop simple field tests to confirm the quality of the ether produced.
“If we could come up with a way for doctors to verify what they made without fancy lab equipment, that would be huge,” she said. “It could make surgery possible in places where it currently isn’t. Sometimes the solutions we need are already in the past. We just have to look at them with fresh eyes.”
Jeffrey Johnson,
James Custer’s Ph.D. ‘Scientific Bet’ Launches an Unexpected Career in Clean Energy Innovation
When James Custer arrived at UNC-Chapel Hill to begin his Ph.D. in chemistry, he didn’t know he was about to launch a career that would take him from highstakes scientific discovery to global clean energy negotiations, and eventually to the front lines of climate technology and entrepreneurship. But he did know one thing about himself—he was curious. Deeply, relentlessly curious.
That curiosity has been the thread running through Custer’s life and career “When I chose to study chemistry in undergrad, I didn’t really know what I was doing,” he said. “I figured if I could understand chemistry from the atoms up, I could build the rest from there.”

What started as an instinct gradually grew into a scientific lens that shifted from chemistry to materials science, then expanded even further into technology commercialization, business strategy, startups and now climate tech leadership. UNC, he said, was the place where that approach took shape.
“My education at UNC helped develop my whole way of problem-solving,” he said. “It gave me the confidence to be curious about anything, even things totally outside my domain.”
Custer’s doctoral research, completed under Professor James Cahoon, was a turning point. His work in materials science led to a first-author paper in the journal Science, a rare achievement for a graduate student and one that came with risk
James Custer, a Ph.D. graduate of the Department of Chemistry, is working on several major projects across the deep tech and clean tech ecosystems in Colorado.
“My Ph.D. was kind of a scientific bet,” he said. “Publishing in Science is extremely difficult. If that paper hadn’t been accepted, things would have turned out very different.”
The gamble paid off, and so did the years of struggle behind it. Cahoon’s mentorship was key. “Jim was great for me,” said Custer. “He had the right balance of being available while also giving you the space to struggle and grow.”
Even while refining his scientific skills, Custer realized the lab wasn’t where he wanted to stay. He was fascinated by the idea of technology leaving the university and entering the world. “Academic work is incredibly important, but the timelines can be very long. I found myself wanting to influence impact on a shorter timeframe ”
That curiosity pulled him toward entrepreneurship. Through UNC’s Kenan-Flagler Business School, he completed a certificate in entrepreneurship and technology commercialization a three-year program he describes as a “mini MBA for STEM graduate students.” There he worked with startups ranging from a kombucha company to a photonics spinout from NC State It was his first real taste of building something new.
That training led directly to his first role after graduation at 8 Rivers, a climate and clean energy technology company. Custer joined as chief of staff and quickly became deeply involved in business strategy and global partnerships He helped secure $100 million in funding, led work in hydrogen and direct air capture, negotiated international deals and advised on projects around the world.
“I treated the business side the same way I treated science,” he said. “Be curious, ask good questions and learn how things work.”
Today, Custer is applying that same mindset to plastics recycling and deep tech entrepreneurship Through Columbia

Custer said Professor James Cahoon’s mentorship at UNC has been key to his success.
University’s ClimateTech Expertise Network, he serves as fractional chief commercial officer for Estercycle, a company developing a next-generation recycling technology. Traditional mechanical recycling can only reuse plastics a few times before the material degrades. Compostable plastics rarely get composted. Estercycle’s approach can take a mixed stream of traditional and bio-based plastics and break them down to their basic building blocks, producing material that behaves like new plastic and can be recycled again and again.
“It solves several problems at once,” said Custer. “It reduces emissions, cuts down on sorting, makes recycling economical and keeps valuable materials in the supply chain.”
At the same time, Custer is building Deep Tech Trailhead, a new open-access resource mapping all the labs, investors, incubators and partners that support deep tech companies across Colorado. “When I moved to Colorado, it felt like excellent resources existed, but they were hard to navigate at times,” he said. “Some people were building maps and lists, but there was no public-facing, deep-tech ecosystem map. I thought, why not just make something that helps everyone?”

In his first role after graduation from UNC, Custer joined 8 Rivers, a climate and clean energy technology company, as chief of staff. He helped secure $100 million in funding for work in hydrogen and direct air capture.
For students considering a path outside traditional academia, Custer offers simple advice: show up. “Networking, more than anything else,” he said. “Just go to things that sound even vaguely interesting. My internship at 8 Rivers came from a random event I went to three years before. Most career transitions come from loose connections, not close friends ”
Looking back, he credits UNC for giving him the mindset that underlies all of it: curiosity, resilience and a willingness to push into the unknown
“UNC taught me how to think, how to ask questions and how to build something new,” said Custer “Everything since has grown from that.”
UNC Scientists Win $1 Million W. M.
Keck Foundation Award to Decode the Secrets of AI-Designed Proteins
BY DAVE DEFUSCO
When you think of technology reshaping the future, you might picture robots, self-driving cars or smart devices. Some of the biggest breakthroughs, however, may come from a much smaller world the world of proteins, which are the molecules that carry out almost every task inside cells. Two scientists at UNC-Chapel Hill, Gary Pielak and Brian Kuhlman, have just received a $1 million Science and Engineering Research Award from the W.M. Keck Foundation to study these molecules in a new way.
Gary Pielak, left, Kenan Distinguished Professor of Chemistry, Biochemistry and Biophysics, and Brian Kuhlman, Oliver Smithies investigator and Professor of Biochemistry and Biophysics.

Their work centers on a surprising puzzle. Proteins designed by artificial intelligence once considered almost science fiction now exist in labs around the world These AI-designed proteins can fold into beautiful, compact shapes and sometimes act as enzymes, the tiny “chemical robots” that speed up the reactions that make life possible. But there’s a catch: AI-designed proteins often behave differently from natural ones, even when they look similar on paper.
“Something unusual is going on inside these designed proteins,” said Pielak, Kenan Distinguished Professor of Chemistry, Biochemistry and Biophysics. “They’re often far more stable than proteins found in nature and, in many cases, their interiors look almost molten or unusually flexible. We want to understand why.”
Natural enzymes are marvels of evolution. They’re shaped over millions of years to be just stable enough to do their job, but not so rigid that they can’t move or adjust when they need to. This balance of structure and motion is what gives them their extraordinary speed and precision. AIdesigned enzymes, however, don’t always follow nature’s rules. Many stay folded even at temperatures near boiling water. Others seem soft or flexible at their core. Yet, these “unnatural” behaviors come from a design process that learns from natural proteins. Page 24
Understanding these differences matters, not just for science but for industry. Enzymes could one day replace many chemical processes that rely on toxic metals or harsh solvents. If researchers can learn how to design enzymes to be as efficient as natural ones, it could transform the pharmaceutical and chemical industries with cleaner, greener technologies. Pielak and Kuhlman, Oliver Smithies investigator and
Professor of Biochemistry and Biophysics, will explore the mystery from two angles.
First, they will study two AI-designed enzymes created in the laboratory of Nobel Prize-winning scientist David Baker. One of these proteins, called LuxSit-i, stays folded even at or above the boiling point of water. Early data suggests that it may have a soft, flexible center.
The second enzyme, Dad t2, works like a natural enzyme but is far less efficient. Pielak’s team will use a powerful technique called nuclear magnetic resonance spectroscopy (NMR) to look inside these proteins and measure how rigid or flexible they are, how stable they remain under heat and how well they perform their catalytic tasks.
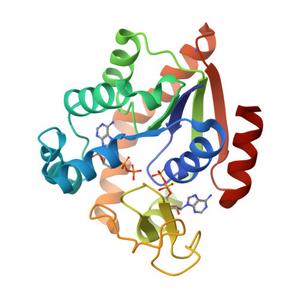
Above, the structure of a natural enzyme, called adenylate kinase which the researchers will redesign using AI.
Credit: Protein Data Base entry 7APU
“NMR lets us see proteins at the level of individual atoms,” said Pielak. “We can measure how each part of the molecule moves and how that movement relates to stability and, perhaps, function. It’s like watching the heartbeat of a protein.”
Second, the researchers will take a natural enzyme called adenylate kinase and redesign it using AI. They want to see whether the redesigned version becomes super-stable, like many AI-created enzymes, and whether that added stability comes with a molten or flexible core.
Comparing the redesigned enzyme with natural versions from cold-loving, moderate-temperature and heat-loving organisms will help reveal how evolution tuned these proteins for different environments.
“We’ll be testing whether AI pushes proteins into states that evolution never chooses,” said Kuhlman, “and, if so, why? What trade-offs is the machine making that nature avoids?”
If AI-designed enzymes are too soft or too rigid on the inside, that may explain why they often don’t match the incredible speed and precision of natural enzymes. Understanding this gap could help scientists design better molecules ones that combine the stability of AI creations with the fine-tuned performance of natural proteins. The payoff could be huge. Enzymes are environmentally friendly: they work in water, avoid toxic metals and use less energy. Better designed enzymes could clean up industrial processes, unlock new therapies and create sustainable ways to build essential chemicals.
“We want to make the next generation of enzymes smarter, greener and more reliable,” said Pielak. “But to do that, we first have to understand how nature works and why AI sometimes breaks the rules.”
Pielak brings more than 40 years of experience probing protein stability and dynamics. He pioneered the use of incell NMR, revealing how proteins behave inside living cells, which is a far more chaotic environment than the test tube. Kuhlman is internationally recognized for his work in protein design, including the first design of an entirely new protein fold. Together, their expertise bridges evolution and computation, experiment and theory.
“The Keck Foundation recognized that we’re at a turning point,” said Kuhlman. “AI is advancing fast, but we’re missing key data about how these designed proteins behave This project fills that gap If we’re successful, we’ll identify ways that AI-based protein modeling can be improved to design proteins with important applications in medicine, industry and research.”
Based in Los Angeles, the W M Keck Foundation was established in 1954 by the late W. M. Keck, founder of the Superior Oil Company. The Foundation’s grant making is focused primarily on pioneering efforts in the areas of medical research and science and engineering. The Foundation also supports undergraduate education and maintains a Southern California Grant Program that provides support for the Los Angeles community, with a special emphasis on children and youth For more information, visit www.wmkeck.org.
Page 25
On Tuesday, Oct. 7, Frank Leibfarth spent the evening at the American Museum of Natural History in New York City, 501 miles from his home base in Carolina’s Caudill Labs.
That’s slightly farther than the combined length of the Deep River, the Haw River and the Cape Fear River that form a unified watershed serving more than 1.5 million North Carolinians. It’s the largest river basin in North Carolina.
It’s also among the most polluted.
Leibfarth’s innovative solution to help remove toxic chemicals from the Cape Fear River Basin was among many reasons he was in New York at a celebration hosted by the Blavatnik Family Foundation and the New York Academy of Sciences. There, he was named the 2025 Blavatnik National Awards for Young Scientists Laureate in the Chemical Sciences, an award that comes with a $250,000 prize.

Frank Leibfarth receives prestigious prize for early-career researchers
BY CALLEY JONES, COLLEGE OF ARTS & SCIENCES
Inspired by the Nobel Prizes, the Blavatnik National Awards for Young Scientists are the largest unrestricted prizes for early-career researchers in the United States. Leibfarth joins two other 2025 Laureates Philip J. Kranzusch from Dana-Farber Cancer Institute, who won the prize in the life sciences category, and Elaina J. Sutley from the University of Kansas, who won the prize in the physical sciences and engineering category.
This year’s laureates were selected from a pool of 310 nominees representing 161 academic and research institutions across 42 states. The award is “designed to empower them with the freedom to continue to explore bold ideas, driving scientific innovation forward,” said Len Blavatnik, head of the Blavatnik Family Foundation.
“We are tremendously proud of Professor Leibfarth for this well-deserved recognition of his groundbreaking work in the chemical sciences,” said Chancellor Lee H. Roberts. “Being named a Blavatnik laureate is an extraordinary achievement and a historic first for Carolina, reflecting the far-reaching impact of his research and innovation.”
Leibfarth, the Royce Murray Distinguished Term Professor of Chemistry and the inaugural Institute for Convergent Science faculty fellow, was awarded the prize “for pioneering approaches to upcycle plastic waste and remove toxic ‘forever chemicals’ from water by developing reactions and catalysts that selectively control the structure and function of polymers.”

Chemical Sciences Laureate Frank Leibfarth speaks to the audience about using polymer chemistry to reduce the damaging effects of plastics on our environment and human health. (Right photo, from The New York Academy of Sciences.)
“Frank is an incredible asset to the College of Arts and Sciences and the Carolina community,” said Jim White, the Craver Family Dean of the College of Arts and Sciences. “His intuitive scientific insights and passion for service are making an impact on the state of North Carolina and inspiring students, staff and fellow faculty alike. We’re delighted to see him receive this well-earned and remarkable honor ”
ABSORBING PFAS
The 2016 finding that per- and polyfluoroalkyl substances, better known as PFAS, had been detected in the Cape Fear River Basin shocked the communities that draw their water from the basin These “forever chemicals” are nearly impossible to break down so they instead accumulate in the environment and in living tissue, where they can increase the risk of cancer, birth defects and other health issues.
Almost a decade later, researchers across disciplines are still working to make sense of what these chemicals mean for their communities and how to mitigate the risk. Leibfarth joined the fight by drawing inspiration from an unconventional source: diapers.
Leibfarth studies polymers, or long chains of repeating chemical structures that make up materials from plastic and rubber to fingernails and DNA The absorbent hydrogels inside diapers are also polymers that keep users dry by clinging tightly to liquids. When a colleague asked Leibfarth why no one had made a hydrogel that could soak up PFAS, he set to work designing one.
(Left to right) Nicholas B. Dirks, Academy President and CEO; 2025 National Laureates: Frank Leibfarth, Elaina J. Sutley, Philip J. Kranzusch; 2025 Regional Laureates: Valentin Crépel, Veena Padmanaban, Xiao Xie; Subra Suresh, Presenter of Ceremonies, President of the Global Learning Council, and Former Director of the US National Science Foundation. (Left photo, from The New York Academy of Sciences.)

Although the hydrogel proved highly effective in a laboratory environment, Leibfarth needed help to scale up his resin for use at water treatment plants.
Enter the North Carolina Collaboratory, an innovative group based at UNC-Chapel Hill that provides state and private support to projects aiming to better North Carolina through research.
The Collaboratory established NC Pure, a research project embedded in its larger PFAS Testing Network and helmed by Leibfarth and his collaborator Orlando Coronell, a professor of environmental sciences and engineering in the UNC Gillings School of Global Public Health and an adjunct professor of applied physical sciences at Carolina Through NC Pure, Leibfarth, Coronell and their team have conducted pilot tests in four municipal water treatment utilities in North Carolina and provided critical data to inform their decision-making around PFAS management. Leibfarth and Coronell cofounded Sorbenta Inc., a start-up company focused on implementing this PFAS removal technology more broadly.
REVITALIZING PLASTIC WASTE
Plastic is another pollutant accumulating in the environment at an alarming rate. While many types of plastic can be recycled after use, the resulting products are less valuable than their starting materials, making plastic recycling an economically disadvantageous process
Two of the most common types of plastic, polyethylene and polypropylene, are notoriously difficult to break down in part because of the strong bonds that hold the molecules together. Leibfarth’s expertise in polymer chemistry gives him unique insights into how researchers might modify and rearrange those bonds to produce new, more valuable materials, thereby “upcycling” the plastic waste.
His lab has been working on chemical reagents that can be added to existing recycling workflows to transform the plastic particles instead of simply heating and grinding them. So far, they’ve found some potential reagents, but they’re effective only on pure polyethylene or polypropylene. Mixed plastics and food residue both common in current recycling pipelines complicate the process. Leibfarth has collaborated with numerous industrial partners to explore avenues to translate this work from a laboratory environment to a commercial setting, with work ongoing to address key economic and environmental limitations.

Leibfarth shows an AI-predicted polymer that he created in collaboration with faculty from other universities
With such enterprising ideas, it is easy to see why the Blavatnik Award is far from Leibfarth’s first notable honor. Some of his other accolades include a Sloan Research Fellowship in Chemistry, a Cottrell Scholar Award, the UNC Tanner Award for Excellence in Undergraduate Teaching, a Barry Goldwater Scholarship, a Presidential Early Career Award and being named a “Brilliant 10” earlycareer scientist by the magazine Popular Science
Leibfarth, however, is quick to mention the contributions of fellow scientists to his awardwinning research “Since starting at UNC in 2016, I have had the privilege of working with some of the most talented and ambitious scientists internationally who care deeply about making plastics more sustainable and removing forever chemicals from our environment,” he said. “The Blavatnik award recognizes their hard work and dedication during their educational journey.”
Leibfarth continued, “I am especially proud to receive the Blavatnik Award for Young Scientists for work at a state university. Being able to do groundbreaking research that serves the students and citizens of the state is immensely fulfilling and is a responsibility I take seriously.”
Awards and Recognitions

ErinBaker
Inaugural Fellow of the American Society for Mass Spectrometry (ASMS)
2025 Chapman Family Teaching Award


CarribethBliem DivisionalFacultyDevelopment Facilitator,AssociateDean,Natural SciencesandMathematics
JoshuaBeaver
Awards and Recognitions

LizBrunk
·PhRMAFacultyStarterGrantinDrugDiscovery
·BoehringerIngelheimEarlyCareerInvestigator
·SelectedforaNationalSynthesisCenterfor EmergenceintheMolecularandCellular Sciencesworkinggroup
NitaEskew
2025TannerAwardforExcellencein UndergraduateTeaching


Awards and Recognitions

2024FellowoftheAmerican PhysicalSociety
FrankLeibfarth
·2024CleantechInnovationAwardFinalist
·PresidentialEarlyCareerAwardinScienceand Engineering(PCASE)
·2025PolymerChemistryLectureshipaward
·2025BlavatnikNationalAwardsforYoungScientists







Connect with Fellow Alumni at UNC Chemistry Events!
Attention UNC Chemistry Alumni! We’re excited to announce a series of exclusive events tailored just for you. These gatherings are designed to enhance your professional network and reconnect with Fellow Alumni, Faculty and Industries linked to UNC Chemistry.
Spring and Fall Event Series
Our Spring Events are conveniently located near ACS Spring Conference venues. Fall Events will be held on UNC Chapel Hill Campus. These events are focused on providing a great opportunity for our Chemistry Alumni to gather, celebrate, and establish valuable connections.
Upcoming Spring Event: Atlanta, GA
�� Date: Monday, March 23, 2026
�� Location: Ray’s in the City, Atlanta, GA
�� Time: 11:00 a.m. – 1:00 p.m.
Complimentary heavy hors d'oeuvres will be served.
Join your fellow alumni in Atlanta for a luncheon filled with stimulating discussions and networking opportunities with industry leaders affiliated with UNC Chemistry. This event aligns perfectly with the ACS Spring Conference, making it an ideal addition to your schedule.
Save the Date: Fall 2026 on Campus
Date: Fall 2026
Location: UNC Chapel Hill Campus
Mark your calendars for an extraordinary event back on our Chapel Hill campus! This fall gathering will be a wonderful chance to explore familiar spots, witness new developments, and build meaningful connections with industry insiders.
More information will be shared as the date approaches. We’re excited to welcome you back to where it all began!
Registration: Scan the QR code or click here!
Stay Connected: Follow us on LinkedIn and email chemadmin@unc.edu to be added to future communication.
We look forward to seeing you soon and celebrating the Tar Heel Chemistry spirit together!