

SAMPLE EXAM PAPERS
BASED ON THE NEW SUBJECT SPECIFICATION FOR LEAVING CERTIFICATE



• 4 SEC Sample Papers
(2 Higher and 2 Ordinary)
• 11 4schools Sample Papers (8 Higher and 3 Ordinary)
• Student Guide to the Exam
• Full-colour papers to reflect the SEC exams
E-BOOK How to claim and access your e-Book
Code:
To claim your e-Book
1. Log in to 4schools.examcraftgroup.ie/user/login
2. Activate your e-Book using the code above at 4schools.examcraftgroup.ie/activatecode
3. Download the app at 4schools.examcraftgroup.ie/apps
4. Log in to the app (please use the same login and password you used on the 4schools website)
5. Download the e-Book
Notes
• Please ensure the email address used to complete the steps above is the email address of the student or teacher who will be using this e-Book.
• If the e-Book code was purchased using another email address, e.g., a parent’s, please register the email address of the e-Book user before completing the steps above. You can register the alternative email address here: 4schools.examcraftgroup.ie/user/register

Online Resources
Get access to exclusive content by scanning the QR code. https://4schools.examcraftgroup.ie/sample-papers-lc-chemistry


AND
BASED ON THE NEW SUBJECT SPECIFICATION
Published by 4schools
89F Lagan Road, Dublin Industrial Estate, Glasnevin, Dublin 11, D11 F98N, Republic of Ireland.
T: ++ 353 1 8081494 - E: info@4schools.ie
SAMPLE
ISBN 978-1-918341-06-5
All rights reserved. No part of this publication may be reproduced or transmitted in any form or by any means, including photocopying and recording, without the publisher’s written permission. Such written permission must also be obtained before any part of this publication is stored in a retrieval system of any nature. Permission requests should be directed to 4schools.
As of the time of publication, all web addresses listed were active and contained information relevant to the topics discussed in this book. 4schools does not accept responsibility for the content or opinions expressed on these websites and videos. Please note that the content, views, and web addresses may change, which is beyond the control of the publisher or the author. Students should always be supervised when reviewing websites and videos.
4schools has attempted to contact all proprietors of images, texts and graphics included in this book. Proprietors we were unable to reach are requested to contact us. We would be happy to make the necessary arrangements at the first opportunity.
Guide to the Leaving Cert Chemistry Exam
and AAC

Written exam
Overview
Duration: 2 hours 30 minutes
Marks: 300 marks (which is 60% of the overall grade). Each question is worth 50 marks
Levels: Ordinary Level and Higher Level
Question Types
• Structured Questions: Questions with several parts that check your understanding of key ideas.
• Application Questions: Real-life examples where you need to explain and solve problems.
• Data Analysis: Use of graphs, tables and experimental results.
• Mixed Topics: Questions may combine topics from different strands (e.g., Nature of Matter + Matter in Our World).

CHEMISTRY

Format:
• Students must answer Question 1 (mandatory) plus five other questions from the paper.
• Questions mix different topics and test skills like analysis, prediction, and evaluation
How the Exam Is Answered
The exam paper is a single bound booklet containing:
• Questions with answer spaces directly underneath each question.
• Students write answers directly into the space provided on the same page.
• Marking lines/boxes guide the expected length.
• No separate answer booklet is used for the written exam.
• Additional space can still be requested, but the structure encourages more concise, targeted responses.
Coimisiún na Scrúduithe Stáit
State Examinations Commission
Higher Level
2 hours 30 minutes
300 marks 2027L022A1ES1
Instructions
There are seven ques�ons on this paper. Each ques�on carries 50 marks.
Answer Ques on 1 and any ve other ques�ons.
Write your Examina�on Number and your Day, Month and Year of Birth in the boxes on the front cover.
Write your answers in blue or black pen. You may use pencil in graphs and diagrams only.
This examina�on booklet will be scanned and your work will be presented to an examiner on screen. All of your work should be presented in the answer areas, or on the given graphs, or diagrams. Anything that you write outside of these areas may not be seen by the examiner.
Write all answers into this booklet. There is space for extra work at the back of the booklet. If you need to use it, label any extra work clearly with the ques�on number and part.
The superintendent will give you a copy of the Formulae and Tables booklet. You must return it at the end of the examina�on. You are not allowed to bring your own copy into the examina�on.
Data from the Formulae and Tables booklet, including atomic numbers and rela�ve atomic masses, should be used wherever necessary.
You may lose marks if your solutions do not include relevant supporting work. You may lose marks if the appropriate units of measurement are not included, where relevant. You may lose marks if your answers are not given in their simplest form, where relevant.
Diagrams are generally not drawn to scale.
Write the make and model of your calculator(s) here:
Ques on 1
(a) The line emission spectra for elements can be modelled using the formula Em – En = hf.
(i) State the physical quan�ty represented by Em
(ii) State the physical quan�ty represented by En.
(iii) State the physical quan�ty represented by f.
Examine the line emission spectra below for salts of elements X and Y.


(iv) Iden�fy element X.
(v) Iden�fy element Y.
(b) Hess’s law states that the enthalpy change (ΔH) for a chemical reac�on is independent of the path taken.
The ΔH values for three gas phase chemical reac�ons are as follows:
(1) 2OF2 O2 + 2F2 ΔH = –49.1 kJ mol–1 (2)
+ 2O2 Cl2O + 3OF2 ΔH = 533.4 kJ mol–1
+ O2 Cl2O + OF2 ΔH = 205.6 kJ mol–1 (3)
Using the data, calculate ΔH for the following chemical reac�on:
+ F2 ClF3
(c) Examine the rst ionisa�on energies of the elements on page 80 of the Formulae and Tables booklet.
(i) What is meant by the rst ionisa�on energy of an element?
First ionisa�on energies generally increase from le� to right across a period (row) of the periodic table. There are, however, excep�ons to this general trend.
(ii) Iden�fy and explain the excep�ons in the second period of the periodic table, i.e. Li to Ne.
SAMPLE
Leaving Cer cate Examina on – Sample 1 5 Chemistry – Higher Level
(d) Propanone (CH3COCH3) is the smallest ketone. It is an important solvent in industries, laboratories and homes. It mixes easily with both polar solvents like water and non‐polar solvents like benzene.
Draw a diagram to explain why propanone mixes easily with water.
Your diagram should show:
one molecule of propanone and one molecule of water,
displacements of charge (or dipoles) in each molecule,
intermolecular forces between specic parts of the molecules.
SAMPLE
Cer cate Examina on – Sample 1
(e) A student carried out an experiment to inves�gate the percentage of elemental iron in tablets containing iron compounds.
The student was given ve of the tablets.
Describe how the student could use the tablets to make up an exact volume of a solu�on of iron(II). Your answer should iden�fy all relevant chemicals and apparatus and describe how they were used.

SAMPLE
Leaving Cer cate Examina on – Sample 1 7 Chemistry – Higher Level
Ques on 2
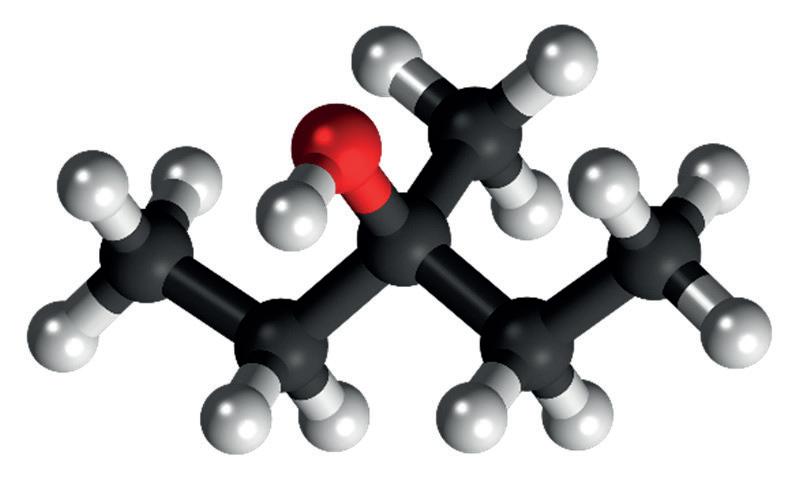
(a) The diagrams below show 3D models of two alcohols with the molecular formula C6H14O. The condensed structural formula for compound A is CH3CH(OH)CH2CH2CH2CH3
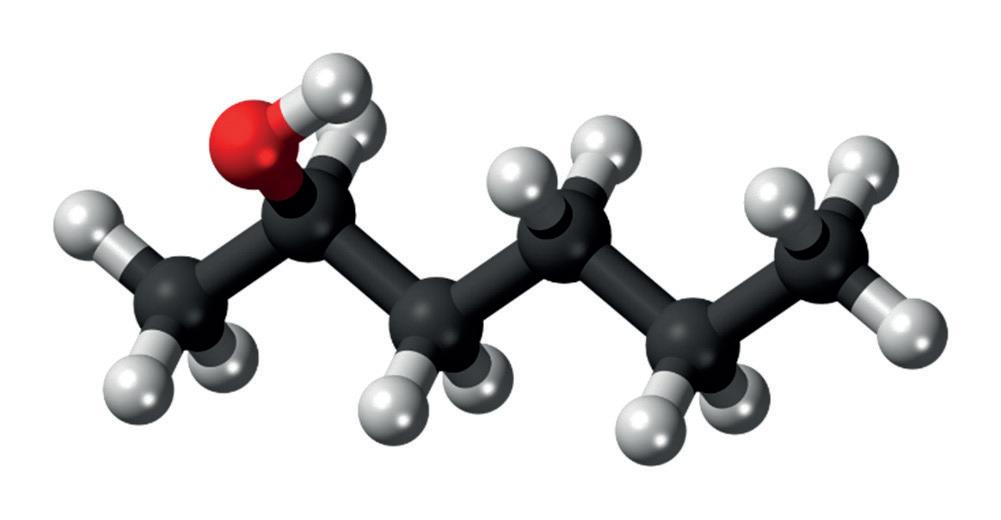
(i) State the IUPAC name for compound A.
(ii) Is compound A a primary, secondary or ter�ary alcohol? Jus�fy your answer.
(iii) State the IUPAC name for compound B.
(iv) Is compound B a primary, secondary or ter�ary alcohol? Jus�fy your answer.
(v) Write the condensed structural formula for compound B.
One advantage of using 3D models such as these is that the shape of the molecule can be be�er understood. For example, from the diagrams of compounds A and B it is clear that the bond angle at the oxygen atom is less than 180°.
(vi) Explain why the bond angle at the oxygen atom in compounds such as alcohols and water is less than 180°.
SAMPLE
(b) Another alcohol, methanol, reacts with ethanoic acid under reux condi�ons to produce an ester.
(i) State the IUPAC name for the ester produced.
(ii) Draw the molecular structure of the ester produced, including all atoms and bonds.
(iii) Iden�fy the other product of this chemical reac�on.
(iv) State one advantage of using reux condi�ons during the produc�on of an ester.
Leaving Cer cate Examina on – Sample 1
Chemistry – Higher Level
(c) Soaps may be produced using long chain esters such as triglycerides, which are natural oils or fats.
(i) Iden�fy the other reactant needed to produce soap from long chain esters.

(ii) A soap is an example of a surfactant. Describe how soaps work as surfactants.
(iii) Other than soap, state one other use or applica�on of surfactants.
SAMPLE
Leaving Cer cate Examina on – Sample 1 11 Chemistry – Higher Level
Ques on 3
(a) Magnesium chloride is an inorganic compound with the chemical formula MgCl2. It is classied as an ionic compound.
(i) By reference to data on page 81 of the Formulae and Tables booklet, explain why magnesium chloride is classied as an ionic compound.
(ii) Write the arrangement of electrons in energy levels and sublevels for the Cl– ion. Explain why this is a stable electronic congura�on.
(iii) Explain why magnesium chloride is highly soluble in water.
(b) One aspect of electrochemistry is electrolysis. This is where an electric current is used to drive a redox reac�on. When an electric current is passed through molten magnesium chloride, it can undergo electrolysis to produce magnesium metal, as described by the following balanced chemical equa�on:
(i) Is magnesium metal produced at the anode or the cathode during this reac�on? Jus�fy your answer.
Another aspect of electrochemistry is the use of redox reac�ons to produce an electric current. This is called a galvanic cell.
(ii) Explain the opera�on of a simple galvanic cell. Your explana�on should iden�fy which chemical species is oxidised and which is reduced.
Leaving Cer cate Examina on – Sample 1
MgCl2 Mg + Cl2
(c) Magnesium chloride may be produced by the neutralisa�on reac�on between solid magnesium hydroxide and hydrochloric acid, as described by the following balanced chemical equa�on:
Mg(OH)2(s) + 2HCl(aq) MgCl2(aq) + 2H2O(l)
22.0 g of magnesium hydroxide reacts completely with 1.8 M hydrochloric acid to produce magnesium chloride and water.
(i) Calculate the mass of magnesium in 22.0 g of magnesium hydroxide.
SAMPLE
(ii) Calculate the minimum volume of 1.8 M hydrochloric acid needed to completely neutralise 22.0 g of magnesium hydroxide.
(iii) Calculate the concentra�on, in moles per litre, of the solu�on of magnesium chloride produced when the volume of 1.8 M hydrochloric acid that you calculated in part (ii) is added to the 22.0 g of magnesium hydroxide.
(You may assume that water has a density of 1.0 g cm–3.)
SAMPLE
Leaving Cer cate Examina on – Sample 1 15 Chemistry – Higher Level
Ques on 4
(a) Hydrogen peroxide (H2O2) decomposes to produce water and oxygen according to the following balanced chemical equa�on:
2H2O2 2H2O + O2
A student measured the volume of oxygen gas produced when a catalyst was introduced to a sample of hydrogen peroxide solu�on.
The data gathered are plo�ed on the graph on the next page.
(i) Calculate the average rate of reac�on over the rst 20 s.
(ii) Calculate the instantaneous rate of reac�on at 20 s.
(iii) Explain why the rate of this reac�on decreases over �me.
(iv) On the graph, show how the data would change if a more effec�ve catalyst had been used.
(v) Describe how the catalyst could be made more effec�ve.
SAMPLE
(b) Another student measured out 150 cm3 of a different solu�on of H2O2, which had a concentra�on of 5% w/v.
Calculate the volume of oxygen gas produced when this sample of H2O2 decomposed completely at standard temperature and pressure.
Leaving Cer cate Examina on – Sample 1
Chemistry – Higher Level
(c) H2O2 can be used in forensic science to detect blood at a crime scene. The H2O2 reacts with the iron (Fe2+) ions in blood in a redox reac�on.
(i) State the oxida�on number of oxygen in H2O2
(ii) Hence or otherwise, balance the following chemical equa�on: H2O2 + Fe2+ + H+ Fe3+ + H2O
SAMPLE
Leaving Cer cate Examina on – Sample 1
Chemistry – Higher Level
Ques on 5
(a) Uisce Éireann treats and monitors the water supply in Ireland to ensure we have sufficient safe water for our homes and businesses.
The water treatment process typically starts with screening, occula�on and sedimenta�on.
(i) Explain what is meant by occula�on and explain why it is carried out during water treatment.
Drinking water is regularly tested in line with EU legisla�on. Many contaminants are tested for, including bacteria, organic molecules and metal ions. Discussions are taking place at EU level about also tes�ng for microplas�cs, which are seen as a contaminant of emerging concern.
(ii) Describe how microplas�cs could come to be in our water supply.
Another step in the water treatment process is UV treatment.
(iii) Explain what is meant by UV treatment and explain why it is carried out during water treatment.
SAMPLE
(iv) Explain the advantage of using UV treatment instead of chlorina�on to treat water.
(b) Uisce Éireann provides over 1.5 billion litres of water each day. An industry of this size has a high energy demand and thus a signicant carbon footprint. In order to reduce carbon emissions, solar panels have been installed at some water treatment plants, such as the one in Thurles, pictured here.
(i) Describe how the use of solar panels can help reduce carbon emissions.
SAMPLE

(ii) Other than the use of solar panels, suggest one way of reducing carbon emissions from the water treatment industry, without affec�ng the quality of the water.
(c) The chlorina�on of water results in the forma�on of hypochlorous acid (HOCl). HOCl is a weak and unstable acid that par�ally dissociates, forming an equilibrium with the hypochlorite anion (ClO ), as described by the following balanced chemical equa�on: HOCl ⇌ H+ + ClO–
(i) Explain what is meant by the term ‘chemical equilibrium’.
(ii) Write an expression for the equilibrium constant (Kc) for this reac�on.
At a pH of 7.53 and a temperature of 25 °C, HOCl and ClO– have equal equilibrium concentra�ons.
(iii) Calculate the value of Kc.
SAMPLE
Leaving Cer cate Examina on – Sample 1
Chemistry – Higher Level
(iv) State Le Châtelier’s principle.
(v) Sodium hypochlorite (NaOCl) was added to the equilibrium mixture. Will the pH increase, decrease or stay the same?
Explain your answer with reference to Le Châtelier’s principle.
Leaving Cer cate Examina on – Sample 1
Chemistry – Higher Level
Ques on 6
(a) Arrhenius, Brønsted and Lowry are notable scien�sts who each helped to develop and modify our understanding of acids and bases.
(i) Dis�nguish between an acid and a base.
(ii) Dis�nguish between a strong acid and a weak acid.
A student carried out a number of acid‐base �tra�ons. She used a pH meter to measure how the pH of the solu�ons changed during each �tra�on. She generated the pH curves shown on the next page.
(iii) Fill in the table to match the pH curves to the different types of reac�on.
type of reac on pH curve (A, B, C or D)
strong acid – strong base
strong acid – weak base
weak acid – strong base
weak acid – weak base
(iv) Explain why it is difficult to carry out an accurate weak acid – weak base �tra�on.
Leaving Cer cate Examina on – Sample 1
(v) Explain why the student used a pH meter rather than universal indicator to measure the pH.
(vi) Name one variable which the student should have kept constant to make sure that she could make a fair comparison between the �tra�ons.
(b) A different student carried out a �tra�on using a weak acid (HA) and a standard solu�on of a base (XOH). The reac�on is described by the following balanced chemical equa�on:
HA + XOH XA + H2O
(i) Explain the importance of using a standard solu�on in a �tra�on.
The XOH solu�on had a concentra�on 0.08 M. At the start of the �tra�on, the student transferred a 20.0 cm3 por�on of the base into a conical ask.
(ii) Calculate the number of moles of XOH in the conical ask.
SAMPLE
The student carried out a number of accurate �tra�ons. The average value for the endpoint was found to be 11.4 cm3 of HA.
(iii) Name the piece of equipment the student used to measure this volume of HA.
Leaving Cer cate Examina on – Sample 1
Level
(iv) How many moles of HA did the student add?
(v) Calculate the concentra�on of HA.
The acid dissocia�on constant (Ka) for HA is 1.74 × 10– 9 .
(vi) Calculate the pH of HA.
Leaving Cer cate Examina on – Sample 1
Chemistry – Higher Level
Ques on 7
(a) But‐2‐ene is the name given to two different hydrocarbon compounds that have the molecular formula C4H8 But‐2‐ene exists as two geometric isomers, cis‐but‐2‐ene and trans‐but‐2‐ene, which have similar physical and chemical proper�es.
(i) Iden�fy one signicant industrial use of hydrocarbon compounds.
(ii) Draw the molecular structure of cis‐but‐2‐ene, including all atoms and bonds.
As part of a research inves�ga�on on hydrocarbons, a student learned that cis‐but‐2‐ene and trans‐but‐2‐ene have similar heats of combus�on. However, the student also learned that the conversion between these two geometric isomers has a high ac�va�on energy, approximately 262 kJ mol–1.
(iii) Explain why cis‐but‐2‐ene and trans‐but‐2‐ene have similar heats of combus�on.
(iv) Write a balanced chemical equa�on for the complete combus�on of but‐2‐ene, or any compound with the molecular formula C4H8.
(v) Explain why the conversion between cis‐but‐2‐ene and trans‐but‐2‐ene has a high ac�va�on energy.
In addi�on to cis‐but‐2‐ene and trans‐but‐2‐ene, there are a number of other isomers with the molecular formula C4H8. One such isomer is but‐1‐ene.
(vi) Explain why but‐1‐ene does not exhibit geometric isomerism.
SAMPLE
Leaving Cer cate Examina on – Sample 1
(b) As part of your studies in Chemistry, you should have conducted an experiment to dehydrate ethanol so as to prepare and collect a sample of the hydrocarbon compound ethene (C2H4) in your school laboratory.
(i) Draw a labelled diagram of the arrangement of apparatus and chemicals you used during the laboratory prepara�on and collec�on of ethene.
(ii) When ethene is bubbled through a dilute pink‐purple solu�on of acidied potassium permanganate (KMnO4), the solu�on becomes colourless. Explain why this happens.
SAMPLE
Leaving Cer cate Examina on – Sample 1
(iii) Describe the reac�on mechanism for the ionic addi�on of chlorine (Cl2) to ethene. Your answer should include a drawing of the structure of any intermediate(s) formed and should show the movement of electrons during the reac�on.
SAMPLE
(iv) State the IUPAC name for the compound produced by the ionic addi�on of chlorine to ethene.
Leaving Cer cate Examina on – Sample 1
Chemistry – Higher Level
Page for extra work.
Label any extra work clearly with the ques�on number and part.
SAMPLE
Page for extra work.
Label any extra work clearly with the ques�on number and part.
SAMPLE
Page for extra work.
Label any extra work clearly with the ques�on number and part.
SAMPLE
Page for extra work.
Label any extra work clearly with the ques�on number and part.
Acknowledgements
SAMPLE
Images
Image on page 3: bitesize.co.uk/bitesize/guides
Image on page 5: examina�ons.ie
Image on page 7: healthcastle.com
Images on page 8: wikipedia.org
Image on page 11: istockphoto.com
Image on page 21: water.ie/projects/local‐projects
Leaving Cer cate Examina on – Sample 1
Do not write on this page
Copyright notice
This examination paper may contain text or images for which the State Examinations Commission is not the copyright owner, and which may have been adapted, for the purpose of assessment, without the authors’ prior consent. This examination paper has been prepared in accordance with section 53(5) of the Copyright and Related Rights Act, 2000. Any subsequent use for a purpose other than the intended purpose is not authorised. The Commission does not accept liability for any infringement of third-party rights arising from unauthorised distribution or use of this examination paper.
Leaving Certificate Examination – Higher Level
SAMPLE
Chemistry
Sample 1
2 hours 30 minutes
Leaving Certificate Examination
Sample A Chemistry
Higher Level
2 hours 30 minutes 300 marks
Examination Number
Date of Birth For example, 3rd February 2005 is entered as 03 02 05
Centre Stamp
You can download this paper’s marking scheme by scanning the QR code:

Instructions
There are seven questions on this examination paper. Each question carries 50 marks.
Answer Question 1 and any five other questions.
Write your details in the boxes on the front cover.
Write your answers in blue or black pen. You may use pencil for sketches, graphs and diagrams only.
This examination booklet will be scanned and your work will be presented to an examiner on screen. All of your work should be presented in the answer areas, or on the given graphs, or diagrams. Anything that you write outside of the answer areas may not be seen by the examiner.
You are not required to use all the space provided. There is space for extra work at the back of the booklet. If you need to use it, label any extra work clearly with the question number and part.
The superintendent will give you a copy of the Formulae and Tables booklet. You must return it at the end of the examination. You are not allowed to bring your own copy into the examination.
Data from the Formulae and Tables booklet, including but not limited to fundamental physical constants, particle physics data and electrical circuit symbols should be used wherever necessary.
You may lose marks if your solutions do not include relevant supporting work.
You may lose marks if the appropriate units of measurement are not included, where relevant.
You may lose marks if your answers are not given in their simplest form, where relevant.
Diagrams are generally not drawn to scale.
Write the make and model of your calculator(s) here:
Question 1
(a) In the mid-19th century, scientists were beginning to recognise patterns in the properties of elements, but there was no universally accepted system for organising them. Dmitri Mendeleev, a Russian chemist, made a groundbreaking contribution in 1869.
(i) State the law that Mendeleev used when arranging the elements in his periodic table.
(ii) Why did Mendeleev leave gaps in his periodic table?
SAMPLE
(iii) Mendeleev reversed the order of iodine and tellurium in his periodic table because iodine fitted better with the halogens. What atomic structure characteristic of iodine matched the halogens?
(b) Enthalpy of formation can be used to calculate enthalpy changes in chemical reaction via Hess’s Law.
(i) What is meant by enthalpy of formation?
The formation of methanol can be described using the following equation:
2CH4(g) + O2(g)
2CH3OH(g)
(ii) Use the bond enthalpy data in the table below to calculate ΔH for this reaction.
SAMPLE
(c) Aluminium oxide is a white compound used in things like abrasives and ceramics.
(i) Predict the type of bonding found in aluminium oxide.
(ii) Would you expect aluminium oxide to be a solid, liquid or gas at room temperature?
(iii) Aluminium oxide is amphoteric. Explain what amphoteric means.
(iv) Aluminium oxide reacts with nitric acid in a neutralisation reaction because aluminium oxide is amphoteric. Balance the equation:
SAMPLE
(d) Discuss the use of organic compounds in natural products for medicinal use.
(e) PCl5 decomposes to form PCl3 and Cl2 in the following equilibrium reaction.
PCl5 (g) ⇋ PCl3 (g) + Cl2 (g) ΔH = 87.9 kJ mol-1
(i) How, if at all, would the equilibrium concentration of PCl5 change if you (a) remove Cl2 and (b) increase the volume of the reaction vessel? Explain your answer in both cases.
SAMPLE
(ii) How is the value of K c affected if the temperature is decreased?
Question 2
(a) Astronomers detect elements in space using spectroscopy, which reveals how atoms interact with light. Sodium is especially easy to identify because it produces strong lines in the visible spectrum.
(i) Which of the following emission line spectrums is the emission spectrum for sodium? X or Y?

(ii) How would the absorption spectrum for sodium differ from the emission spectrum?
(iii) The emission line spectrum gives us information about the structure of the atom. How did Niels Bohr explain the appearance of the coloured lines?
SAMPLE

(iv) The Bohr model of the atom explained hydrogen’s emission spectrum. However, it could not fully account for the spectra of more complex atoms like sodium. How did the idea of orbitals improve our understanding of where electrons are in an atom?
(v) Draw a diagram of a p orbital.
SAMPLE
(b) (i) Define first ionisation energy of a neutral gaseous atom in its ground state.
(ii) The approximate values for the first eleven ionisation energies of sodium are given in the following table.
Outline how these values give further evidence for the existence of energy levels.
(iii) Explain why sodium has a higher first ionisation energy than potassium but a lower first ionisation energy than magnesium.
(iv) The mass spectrum of sodium shows that it has two stable isotopes as shown below:
Use the data given to calculate the relative atomic mass of sodium correct to two decimal places.
(v) Write the electronic configuration for the sodium ion Na+
Question 3
(a) In a redox titration, a chemist analyses a solution of sodium nitrite (NaNO₂). A 20.0 cm³ sample of the solution is reacted with 33.0 cm³ of a 0.018 M solution of potassium chlorate(V) (KClO₃) in acidic conditions. The balanced equation for the reaction is:
2ClO3 - + 5NO2 - + 2H+
Cl2 + 5NO3 - + H2O
(i) Calculate the number of moles of chlorate (V) ions used.
(ii) Determine the number of moles of nitrite ions that reacted.
(iii) Calculate the concentration of sodium nitrite in the original solution in g/L.
(iv) One of the products of the reaction is chlorine gas (Cl₂). Calculate the volume of chlorine gas produced at room temperature and pressure (assume 1 mol of gas occupies 24.0 dm³).
(v) Calculate the number of molecules of sodium nitrate (NaNO3) produced. How many nitrate ions did this quantity of sodium nitrate contain?
(b) Chlorine gas, a product of this reaction, is used in the treatment of water.
(i) What is the purpose of adding chlorine to a drinking water supply?
(ii) Give one advantage of microfiltration over sand filtration in water treatment.
(iii) Give an example of a substance that can be added to water if the pH is too high.
(iv) One of the products of the above reaction is sodium nitrate. Outline tests that can be carried out to confirm the presence of (a) the sodium ion and (b) the nitrate ion.
SAMPLE
Question 4
(a) A student carried out an experiment to determine the heat of combustion of propan-1-ol. A spirit burner containing propan-1-ol was used to heat 100 g of water in a copper calorimeter. The temperature of the water increased from 20°C to 58°C. The mass of the spirit burner before and after the experiment was 155 g and 152 g, respectively.
(i) Calculate the amount of heat energy absorbed by the water.
(ii) Calculate the mass of propan-1-ol burned.
(iii) Calculate the heat of combustion in kJ mol-1
(iv) Suggest two sources of error.
(b) Propan-1-ol can be converted to propanal by means of an oxidation reaction.
(i) Identify suitable reagents for this conversion.
(ii) A few drops of Fehling’s reagent was added to a sample of propanal. What was observed upon this addition?
(iii) What was observed when Fehling’s reagent was added to some propanone? Explain this observation.
SAMPLE
(c) Propanoic acid is a product of the reaction of propanal and Fehling’s reagent. The boiling points of propanoic acid, propanal and propan-1-ol are shown in the table below.
(i) Explain why propanoic acid has a higher boiling point than propan-1-ol.
(ii) Propanal and propan-1-ol have similar relative molecular masses yet the boiling point of propan-1-ol is almost twice the boiling point of propanal. Explain why this is the case.
(iii) A student prepared an ester by reacting propanoic acid and propan-1-ol react together using a reflux method. (a) Name the ester formed in the reaction and (b) explain the purpose of using a reflux method.
Question 5
(a) A student investigates the reaction between magnesium ribbon and ethanoic acid. The experimental setup is shown below:
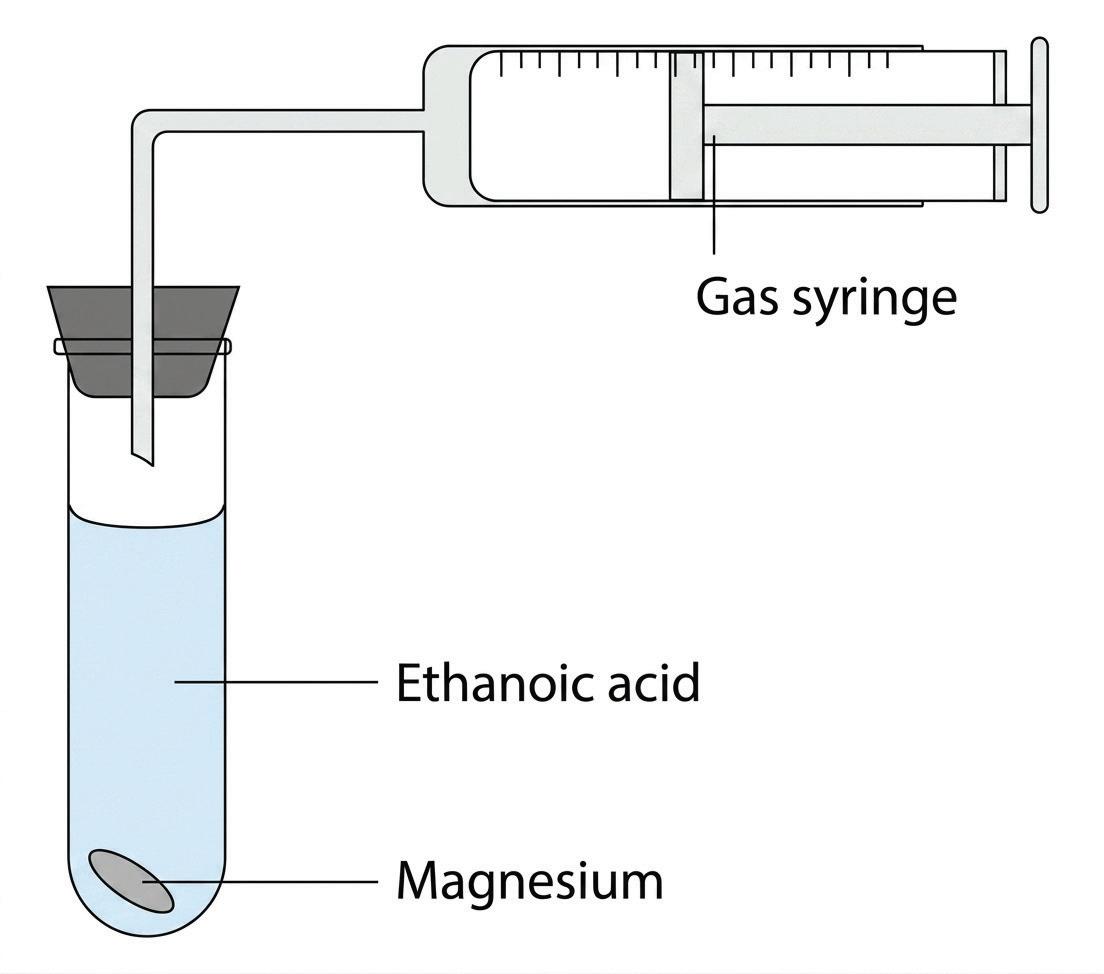
(i) What is observed in the boiling tube as the reaction proceeds?
(ii) Hydrogen is produced by this reaction. Describe a test that can be carried out to show that hydrogen gas is produced.
(iii) Write a balanced equation for the reaction between magnesium and ethanoic acid.
AI Generated Image
(iv) Ethanoic acid is a weak acid. Calculate the value of K a for a 0.075 M solution of ethanoic acid with a pH of 3.06.
(v) Describe the influence of the inductive effect on the acidity of ethanoic acid.
(b) The volumes of hydrogen gas collected at room temperature and pressure at intervals over 2 minutes are recorded in the table below.
Time (seconds) 0102030405060708090100110120
Volume of H₂ (cm3) 0122230 36 4145 48 5051525252
(i) Plot a graph of volume versus time for the reaction between magnesium and ethanoic acid.
(ii) Use your graph to determine the instantaneous rate at 20 seconds (in cm³ H₂ per second at room temperature and pressure).
(iii) Mark clearly on your graph above the curve you would expect to obtain if the reaction were repeated using hydrochloric acid instead of ethanoic acid.
(iv) Use the collision theory to explain the effect using hydrochloric acid instead of ethanoic acid will have on the rate of reaction.
SAMPLE
(v) Calculate the maximum mass of hydrogen produced in the reaction given the reaction was carried out at room temperature and pressure.
Question 6
(a) The Haber process for the manufacture of ammonia involves reacting nitrogen and hydrogen. Chemical equilibrium is established according to the following balanced equation.
2 (g) + 3H2 (g) ⇋ 2NH3 (g)
(i) This reaction is an example of a dynamic equilibrium. Explain what this means in terms of concentrations of reactants and products.
(ii) Write an expression for the equilibrium constant Kc for this reaction.
(iii) The initial concentrations of N2 and H2 are 0.25 mol L-1 and 0.85 mol L-1 respectively. At equilibrium it was found that there was 0.4 mol L-1 of NH3 in the equilibrium mixture. Calculate the value of K c at 454 oC.
N
(b) An iron catalyst is used in the Haber process.
(i) Describe the role of the iron catalyst in the Haber process.
(ii) Explain why finely divided iron is used instead of solid iron blocks.
(iii) The catalyst does not affect the position of equilibrium. Explain why.
(iv) Using the surface adsorption theory, explain how the iron catalyst increases the rate of reaction.
(c) Some of the ammonia produced in the Haber process is used as a reactant in the production of urea, an important nitrogen-based fertiliser. In one step of the process, ammonia reacts with carbon dioxide as follows:
A fertiliser plant has 3.6 x 107 cm3 of ammonia gas stored at a pressure of 4.8 x 102 kPa and a temperature of 330K.
Calculate the number of moles of urea that can be produced from this amount of ammonia, assuming carbon dioxide is in excess.
2NH3 (g) + CO2 (g)
NH2CONH2 (s) + H2O(g)
Question 7
(a) Compound A can be converted to compound B, as shown below: Reaction 1
(i) Give the systematic (IUPAC) name for compound A.
(ii) Draw the structure of a molecule of compound A showing all of its atoms and all of its bonds.
(iii) What organic reaction type is involved in Reaction 1?
(iv) State the reagents required to bring about Reaction 1.
SAMPLE
(v) Compound B is a primary alcohol. Explain why it is classified as a primary alcohol.
(b) Compound A can undergo an elimination reaction as shown in Reaction 2 below.
Reaction 2
(i) What properties of an elimination reaction are seen in Reaction 2?
(ii) The product of this reaction C2H4 undergoes an addition reaction to produce C2H6. How does the carbon geometry change in this reaction?
SAMPLE
(c) Reaction 3 is shown below.
(i) State the reagent and conditions required to bring about Reaction 3.
(ii) The initiation stage of this reaction is know as a photochemical reaction. Explain why.
(iii) One of the propagation steps in this mechanism involves setting up a chain reaction. Describe the reaction mechanism for this step. You should show the movement of electrons during the reaction.
(iv) Why are only trace amounts of butane formed in the reaction?
SAMPLE
Page for extra work. Label any extra work clearly with the question number and part.
Page for extra work. Label any extra work clearly with the question number and part.
Do not write on this page
Copyright notice
This examination paper may contain text or images for which the Examcraft Group is not the copyright owner, and which may have been adapted, for the purpose of assessment, without the authors’ prior consent. Any subsequent use for a purpose other than the intended purpose is not authorised. The Examcraft Group does not accept liability for any infringement of third-party rights arising from unauthorised distribution or use of this examination paper.
Leaving Certificate Examination - Higher Level
SAMPLE
Chemistry
Sample A
2 hours 30 minutes
You can download this paper’s marking scheme by scanning the QR code:


SAMPLE





