














Dear Friends,
Welcome to the Spring/Summer issue of Sulfuric Acid Today magazine. We are committed to bringing you the latest products and technologies impacting our industry, and we hope you find this issue both helpful and informative.
Inside, you’ll find several insightful articles covering cutting-edge technology and major projects. Our cover story highlights Lithium Americas Corp.’s development of the Thacker Pass project in Nevada, including a large on-site sulfuric acid plant to enable lithium extraction and generate power, with phase one targeting production by 2027 (page 7).
Acuity Commodities reports that sulfur prices reached their highest levels since 2008 in early 2026, driven by strong demand, particularly from Indonesian nickel projects, and constrained supply from Russia, structural shifts in refining, and Chinese export controls (page 10).
Elessent Clean Technologies explains that frequent shutdown–startup cycles in sulfuric acid plants cause thermal cycling, which damages catalyst structure and chemistry by cracking the porous support, altering vanadium oxidation states, and amplifying contamination effects (page 12).
Weir offers Lewis® pumps for molten sulfur and sulfuric acid that use specialized vertical centrifugal designs and carefully selected materials to withstand demanding conditions while delivering high efficiency,
durability, and reliable long-term performance (page 16).
Combustion Solutions reports that its advanced sulfur atomization nozzles improve combustion efficiency and spray uniformity, enabling faster sulfur-to-SO2 conversion (page 18).
CG Thermal presents a practical gasto-gas heat exchanger design using hotshell/cold-tube flow, radial variable-pitch tubes, and a dual-shell configuration to actively manage cold-end temperatures, minimize fouling and corrosion, and improve long-term reliability (page 20).
INTEREP notes that when industrial expansion joints fail, the most costly mistakes result from prioritizing price and lead time over downtime costs, operational constraints, and environmental risks (page 21).
Elessent Clean Technologies explains that modern sulfur plants must often choose between two main filtration options: traditional pressure leaf filters and self-cleaning candle filters (page 22).
Nemaska Lithium’s Whabouchi mine in Quebec installed Beltran Technologies’ advanced wet electrostatic precipitators (WESPs) to capture submicron particulates and acid mists, ensuring over 99.8% clean emissions and protecting both sulfuric acid purity and plant equipment (page 24).
Porter Process Technologies explains how optimal sulfur burner design balances complete sulfur combustion, minimal maintenance and safety risks, and reason-
able capital costs—keeping the system “between the ditches” of performance, cost, and reliability (page 26).
Topsoe’s VK69 catalyst, now more than 30 years old, continues to enable sulfuric acid plants to achieve ultra-low SO2 emissions, boost acid and steam production, improve energy efficiency, and reduce operational costs—with even greater performance when combined with the newer VK38+ catalyst (page 28).
We are pleased to welcome our new and returning Sulfuric Acid Today advertisers and contributors, including: Acid Piping Technology Inc., Acuity Commodities, BASF, Beltran Technologies, Central Maintenance & Welding, CG Thermal, Chemetics, Christy Catalytics, Clark Solutions, Combustion Solutions, Elessent MECS Technologies, INTEREP, Mercad Equipment Inc., NORAM Engineering & Constructors, Porter Process Technologies, Southwest Refractory of Texas, Spraying Systems Co., VIP International, and Weir. We are now assembling content for our Fall/Winter 2026 issue. If you have article suggestions or other contributions, please email me at kathy@h2so4today. com. I look forward to hearing from you.
Sincerely,
Kathy Hayward



AMMAN—The Indo-Jordan Chemicals Company (IJCC), a wholly owned subsidiary of the Jordan Phosphate Mines Company (JPMC), signed a $193 million strategic agreement with China’s East China Engineering Science and Technology Company to construct a sulfuric acid plant in the Shidiya area.
The new facility will have an annual production capacity of 900,000 tonnes of concentrated sulfuric acid and is scheduled to be completed within 30 months, in line with the highest international technical, engineering, and environmental standards, the Jordan News Agency, Petra, reported.
Under the agreement, the sulfuric acid produced will be used in phosphoric acid manufacturing as part of a new expansion phase, increasing IJCC’s phosphoric acid production capacity from 330,000 tonnes to 550,000 tonnes annually.
The agreement was signed in the presence of JPMC Chairman Mohammad Thneibat, and JPMC CEO Abdulwahab Ruwwad, while East China Engineering was represented by its CEO Meng Chenzhou.
Thneibat said the project reflects JPMC’s commitment to expanding investments and developing operations in line with royal directives and the economic modernization vision, noting that it will enhance value-added production in Jordan’s mining sector and create
direct and indirect employment opportunities during construction and operation.
Rawad stressed the project’s role in ensuring a stable supply of sulfuric acid to support phosphoric acid production, improving operational efficiency and strengthening competitiveness in regional and global markets.
For his part, Chenzhou expressed pride in the partnership, reaffirming his company’s commitment to delivering the project to the highest standards and expanding long-term industrial cooperation with Jordan.
For more information, visit www.ijcltd.com.
ESPOO, Finland—Metso has won a major order for the delivery of engineering and key process equipment for a new primary copper smelter investment in Asia.
At the same time, Metso has also booked the third part of the orders for JSC Almalyk Mining and Metallurgical Complex’s (Almalyk MMC) copper smelter project.
The planned production capacity of the new copper smelter complex is 300,000 tpa of copper cathodes and 1.1 million tpa of sulfuric acid.
The new copper smelting line is based on the licensed, well-proven Metso’s Outotec® Flash Smelting, PS Converting and gas handling technologies. It includes the design and supply of key process equipment for the main areas of the smelter complex, and the gas cleaning and sulfuric acid plant, copper electrolytic refinery, and precious metals refinery. The
delivery also comprises site services and spares.
“We are very pleased about this order. The Outotec® Copper Flash Smelting method, which is part of the Metso Plus portfolio, is the world’s most widely applied technology for large-scale copper smelting plants,” says Piia Karhu, President, Minerals at Metso.
At the ongoing Almalyk project, the first parts of the orders were booked in the fourth quarter of 2024, and the second part in the first quarter of 2025. The order value recorded in the Minerals segment’s orders in the fourth quarter of 2025 is $82.7 million. Possible additional orders will be announced once they have been signed and become effective.
Almalyk MMC is the main copper producer in Uzbekistan. The company produces refined copper, gold, silver, zinc, molybdenum, lead concentrate, and other products.
Metso’s scope of delivery to Almalyk MMC’s new copper smelter complex includes flash smelting and flash converting, gas cleaning, and sulfuric acid production technologies. The planned production capacity of the new copper smelter, which will be integrated with the company’s existing operations in Almalyk, is 300,000 t/y of copper cathodes and 1.8 Mt/y of sulfuric acid.
Metso is a leading technology supplier to the copper processing industry, offering comprehensive solutions that span the entire production chain, from concentrate processing to refined copper. For more information visit www.metso.com/ commodities/copper.
SANTIAGO—Chile’s state-run copper producer Codelco and Glencore, a Swiss trader and miner, have signed an initial agreement to collaborate on a smelter project in Chile.
The deal calls for Codelco to provide copper concentrate and for Glencore to build the smelter in the key mining region of Antofagasta, in northern Chile, with capacity to process about 1.5 million metric tons a year.
Industry experts say a project of this size would likely require a $1.5 billion to $2 billion investment.
Glencore will carry out a pre-feasibility study; and if the project moves forward, it would start construction in 2030 and begin operations between 2032 and 2033.
Codelco added that it selected Glencore after a competitive bidding process. It agreed to offer Glencore up to 800,000 metric tons of concentrate annually for at least a decade as part of the deal.
Chile accounts for just 6% of global smelting capacity, and sends much of its copper for processing to China, which holds half the world’s copper smelting capacity.
With China’s highly efficient smelters competing for limited concentrate, treatment charges (the fees paid by miners to convert concentrate into metal) have fallen below zero and forced some overseas smelters out of business.
Chile is aiming to increase its own smelting capacity, including a $1.7 billion smelter modernization led by state-run mining company ENAMI.

Codelco Chairman Maximo Pacheco said the Glencore smelter’s location in the Antofagasta region, where most of Chile’s copper is mined, is intended to allow for logistical benefits, and would be built with strong operational and environmental standards.
He added that increasing Chile’s smelting capacity was intended to benefit Codelco as well as help with “strengthening Chile’s sovereignty and strategic security.”
For more information, visit Codelco at www.codelco.com/en and Glencore at www. glencore.com.
SEOUL—Global metals producer Korea Zinc has announced its intention to work in cooperation with federal and local agencies to build a $7.4 billion smelting facility in Clarksville, TN. The smelter will convert metals production byproducts and electronic scrap into 13 different types of metal.
Seoul-based Korea Zinc has existing assets in the United States, including an existing smelter in Tennessee, a majority stake in New York-based electronics recycling firm PedalPoint (formerly Igneo) Technologies, and ownership of nonferrous recycling and trading company Kataman Metals, St. Louis.
“As geopolitical competition over natural resources intensifies globally and certain countries increasingly exert influence over critical mineral supply chains, the project is expected to strengthen U.S.-Republic of Korea (South Korea) economic security cooperation while contributing significantly to global supply chain diversification,” Korea Zinc says.
The company foresees annual production capacity in Clarksville of 540,000 combined tons annually of 13 different nonferrous metals, and says the proposed facility will be modeled after its existing Onsan smelter in South Korea.
The scrap includes printed circuit boards and other forms of electronic scrap that contain precious metals, nonferrous metals and rare earth elements (REEs).
The 13 metals planned for production at the Tennessee facility include zinc, lead, copper, gold, silver, antimony, indium, bismuth, tellurium, cadmium, palladium, gallium and germanium.
Korea Zinc expects construction to begin in 2026, with full-scale construction to begin in 2027 followed by phased commercial operations starting in 2029. The company estimates the sprawling complex will occupy approximately 160 acres (650,000 square meters) and will be built on the site of its current Nyrstar zinc concentrates smelter in Clarksville.
“Korea Zinc’s critical minerals project in Tennessee is a transformational deal for America,” Commerce Secretary Howard Lutnick says. “Our country will now produce, in volume, 13 critical and strategic minerals that are vital to aerospace and defense, semiconductors, artificial intelligence, quantum computing, autos, industrials and national security.”
While the company’s announcement refers to recycled content as feedstock for the smelter, a separate news release from the Tennessee Department of Economic and
Community Development (TNECD) refers to a closed zinc mine in that state that could be reopened in a related move.
Per TNECD, the company plans to reopen a former mine in Gordonsville, TN, bringing back lost jobs and industry to the community.
Tennessee Deputy Governor and TNECD Commissioner Stuart C. McWhorter said: “Korea Zinc’s decision to invest in Tennessee, which marks the single largest investment in state history, reinforces the importance of our strategic global recruitment efforts. The international relationships we’ve built over the years have been essential in securing projects like this one. We are grateful to Korea Zinc for investing in Tennessee and to our partners for helping demonstrate the strength of the Tennessee story and the volunteer spirit to companies around the world.”
Korea Zinc board chair Yun B Choi adds, “With its project in the U.S., Korea Zinc will solidify its position as a strategic partner supplying essential minerals for aerospace and defense. This will become a model case of strengthened U.S.-ROK economic security cooperation. Given the current geopolitical climate and strong U.S. support, now is the optimal moment for expansion into the American market.”
Founded in 1974, Korea Zinc is a leading comprehensive nonferrous smelter operation with initiatives in renewable energy and green hydrogen, critical materials for secondary batteries, and resource recycling efforts. For more information, visit www. koreazinc.co.kr/en.
VANCOUVER—Marimaca Copper has taken an uncommon approach to stabilizing the price of their inputs for their flagship Marimaca Copper project in Chile, announcing this morning that they have entered into an option agreement to acquire a sulfuric acid plant.
The plant, which has a capacity of 150,000 tonnes per annum, represents 30-40% of the total estimated needs of the Marimaca Oxide Deposit once fully operational. While it may not cover the entire need for the project, the acquisition of the plant would reduce the volatility of a key input cost, while also reducing the cost of sulfuric acid to the company by an estimated 30%.
Under the terms of the option agreement, Marimaca has agreed to pay US$2.5 million for the plant, which is expected to be moved to Mejillones. An exclusivity period of 3 months has also been arranged to enable the company to further review technical and engineering aspects, as well as operating cost estimates for the installation and operation of the plant. Similar equipment, if purchased new, is estimated to cost between US$35 and US$40 million, with total installation costs between US$50 and US$60 million.
If the option is exercised, the plant is expected to be mobilized to Marimaca’s site before June 30, 2026.
For more information, visit www. marimaca.com. q



By Acid Piping Technology, Inc.

SINCE 1991, ACID PIPING TECHNOLOGY, INC. HAS BEEN THE WORLD LEADER IN RELIABLE AND COST‐EFFECTIVE PRODUCTS FOR THE SULFURIC ACID INDUSTRY. MONDI™ PIPING WAS SPECIFICALLY DESIGNED FOR 30+ YEARS OF CONTINUOUS SERVICE IN HIGH CONCENTRATION SULFURIC ACID AT A FRACTION OF THE COST OF OTHERPIPING SYSTEMS. LEARN MORE ABOUT HOW MONDI™ PIPING CAN BENEFIT YOUR OPERATION AT WWW.ACIDPIPING.COM. CUT THE COST. INCREASE THE DEPENDABILITY.
By: April Smith, Editor

Lithium Americas (LAC) is building Thacker Pass, a sedimentary-based lithium project in Humboldt County, northern Nevada. LAC is focused on advancing phase 1 of Thacker Pass toward production, targeting nominal design capacity of 40,000 tonnes per year of battery-quality lithium carbonate (Li2CO3), with mechanical completion targeted for late-2027. Sulfuric acid is the key reagent in extracting lithium from ore and phase 1 will include an integrated sulfuric acid plant, one of the largest to be built in North America in the last 15 years.
“To provide enough sulfuric acid required for lithium production, plus take advantage of the waste heat generated, we’re building a conventional 2,250 metric tons per day (MTPD) dual contact dual absorption sulfuric acid plant,” said Mickey Jones, Project Director, Lithium Americas.
Thacker Pass is located in the McDermitt Caldera, an extinct 25x20 mile supervolcano formed about 16.3 million years ago from a hotspot underneath the Yellowstone Plateau. Following an initial eruption and concurrent collapse of the McDermitt Caldera, a large lake formed in the caldera basin. Volcanic activity subsequently uplifted the caldera, draining the lake and bringing the lithium-rich moat sediments to the
surface resulting in the near-surface lithium deposit.
In 2021, following years of drilling and environmental assessments, the Federal Bureau of Land Management issued a Record of Decision approving the plan of operation. In 2022, additional major environmental permits were obtained and LAC began securing engineering contractors to develop the Thacker Pass facility. Early construction commenced in February 2023 and today major construction continues with approximately 1,000 personnel on site, expected to increase to 1,800 at peak construction in late 2026.
Numerous contractors are involved in the project including engineering, procurement and construction management (EPCM) company, Bechtel Corporation, sulfuric acid plant engineering and procurement contractor, EXP OGC Group, and sulfuric acid technology provider, Elessent Clean Technologies/MECS Inc.
Since the 1970s, several companies have held the mineral rights to what is now known today as Thacker Pass and extensively surveyed the deposit over the years. In 2022 a feasibility study targeting nominal production capacity of 40,000 tonnes of lithium carbonate per year was published. This feasibility study included two phases to grow production from 40,000 to 80,000 tonnes per year of lithium carbonate.

In 2020, LAC built its original testing facility, and in 2022 built a state-of-the-art Lithium Technical Development Center (Tech Center) to validate the flowsheet from Thacker Pass ore via a continuous production process and all recycles in place. The Tech Center has been producing battery-quality lithium carbonate samples for customers, developing training and quality standards, and continually optimizing the flowsheet.
“Test work and feasibility studies indicated that we needed a larger sulfuric acid plant.” said Jones. “So, we went into major testing mode focusing on efficient use of the sulfuric acid to target the lithium we needed to recover.”
Testing and process improvements continued at the Tech Center. In 2024, the acid plant design was reconfigured from a heat
recovery system to a conventional plant but with the ability to reconvert to a heat recovery plant later. “The simplified design for first production allows for improved reliability on startup,” Jones said.
The result of the testing led to increasing sulfuric acid efficiency, which decreased the required acid plant size to the current 2,250 MTPD while maintaining the targeted nominal production of 40,000 tonnes per year of lithium carbonate.
In January 2025, LAC published the results in a National Instrument 43-101 Technical Report, positioning Thacker Pass as the largest known measured lithium resource (Measured and Indicated) and reserve (Proven and Probable) in the world. The report outlined potential for annual production capacity of up to 160,000 metric tons per year of battery-quality lithium carbonate over five phases. Production will operate 24 hours/day, 365 days/year with a projected overall availability of 88% and a mine life of 85 years. Over the entire life of the mine, production capacity is estimated at a total of 11.5 million metric tons of battery quality lithium carbonate.
The U.S. Department of Interior has listed lithium as a critical mineral that is vital for America’s national security and

economic resilience.
Currently, under one percent of the world’s lithium processing occurs in the United States, which makes the country dependent on foreign supply, most of which is fulfilled by China. Lithium production at Thacker Pass will enable a domestic lithium supply chain thereby reducing the country’s dependency on foreign suppliers. Thacker Pass production will provide a cornerstone for the supply of products that improve national defense, power resilience, and products used in daily life.
“Securing domestic lithium supply is essential to strengthening domestic energy independence, meeting rising electricity demand and ensuring the United States can win the global technology race,” said Jonathan Evans, CEO of Lithium Americas.
Project financing for phase 1 of Thacker

Pass includes a $2.23 billion loan from the U.S. Department of Energy (DOE) under the Advanced Technology Vehicles Manufacturing Loan Program and strategic investments from General Motors Holdings LLC (GM) totaling $960 million and a $250 million investment from funds managed by Orion Resources LP. GM’s investment represents the largest investment by an Original Equipment Manufacturer (OEM) in a U.S. lithium carbonate project. Additionally, the U.S. Department of Defense granted the Company $11.8 million to support an upgrade of the local power infrastructure and to help build a transloading facility. Thacker Pass is owned by a joint venture between LAC and General Motors (GM). The U.S. government has an option to acquire 5% of the project.
Liquid sulfur will be the most consumed reagent at Thacker Pass that will then be converted to sulfuric acid on-site and used in the leaching process. Each planned phase of Thacker Pass will have its own sulfuric acid plant, with phases 1-4 each capable of producing 2,250 t/d and Phase 5 capable of producing 3,000 t/d. The sulfur, which is the same type local farmers use as soil amendment, will be delivered to the site and unloaded by gravity into sulfur pits that will supply the sulfuric acid plants. Each acid plant will also generate power for lithium processing with any additional power needed supplemented from the local power grid.
The excess heat generated from the production of sulfuric acid will be captured to produce steam which, in turn, will be used to generate electrical power via a steam turbine generator. The generator’s power output is 25.2 MW. This power will be used in the lithium processing plant thereby reducing power consumption at the site.
A key design consideration for sulfuric acid product at Thacker Pass relates to stringent environmental emissions regulations.
“Although federal regulations allow 400 ppm of SO2 to be discharged to the atmosphere, the state of Nevada is requiring the Thacker Pass sulfuric acid plant restrict emissions to 7 ppm of SO2,” said John Horne, Business Development Manager at Elessent Clean Technologies, the project’s main technology provider.
“Few sulfuric acid plants in operation today achieve emissions that low,” said Horne. “The Thacker Pass sulfuric acid plant will aim to be one of the lowest emissions MECS® designs in the United States and around the world.”
For the phase 1 acid plant, Elessent Clean Technologies is supplying MECS® equipment that is proven in other locations to achieve these stringent requirements.
Beyond emissions regulations, many other considerations influenced LAC’s choice of equipment, including keeping up with 24/7/365 lithium processing and operating on a threeyear turnaround cycle.
“The converter is the heart of the sulfuric acid plant, so the team spent a considerable amount of time analyzing design options,” said Kevin Bryan, Sulfuric Acid Plant Consultant at LAC. Given the criteria of reliable, efficient and safe performance, it was a choice between a staid converter, with flat division plates and screens supported by posts and beams, or a catenary converter (also known as blooper plate) with catenary plates and selfsupporting screens.
In terms of cost and design preference, the two options weighed similarly. And both options can achieve a three-year turnaround cycle given a sufficiently large diameter, conservative catalyst bed depth, and room for additional catalyst.
Catalyst selection is instrumental regardless of converter choice. “Key to making the three-year turnaround is the use of MECS® GEAR® GR-330 catalyst in the converter’s first pass. The larger 13 MM catalyst pushes the process dust deeper into the catalyst bed reducing the pressure drop buildup over time,” said Horne.
When evaluating lifespan, the team found more examples of 30-year-old staid converters than the catenary type, which is a newer design.
“Staid types have thicker walls and division plates,” Bryan explained. “Plus, modern staid converters are all stainless steel with posts that are welded in place. We don’t have to contend with cast iron posts and grating that relies on gravity and plumbness to stay in place, like with the staid converters from 30 years ago. So we determined that the modern stainless steel staid converter had better chance of achieving a 40-year life than the catenary type,” he said.
Both designs will experience gas leaks if the insulation flashing is not designed and installed properly. The flat roof typical of large staid converters can have water ingress at the low point versus the domed roof of catenary style where water flows uniformly off all sides. “So we decided we could eliminate that disadvantage by utilizing a domed roof on a staid converter. Plus, the thicker plates used in the staid converter can provide insurance against gas leaks,” said Bryan.


Both designs could simplify troubleshooting by implementing an external rather than an internal gas heat exchanger.
“While operating, there is no easy way to differentiate whether the problem is with the converter or the heat exchanger and a cold shut down may be required to determine the cause. So we decided troubleshooting is equal between the two converter types as long as there are no internal heat exchangers,” Bryan said.
Based on the criteria, the team chose the staid converter with a domed roof and segmented rings at the division plates. They also specified that stresses at high converter temperature should be minimized, gas flow distribution optimized, and the insulation flashing design and installation should be closely monitored.

Other aspects of the plant’s design were influenced by the site’s remote location and particular local conditions. The elevation at Thaker Pass is 4,760 ft and the climate is arid. “Special provisions have been made for larger gas volumes due to elevation and for water conservation, such as the use of closed loop fin fan coolers for heat removal,” said Vulcan Mutler, Senior Vice President at EXP OGC Group, the contractor providing engineering and procurement services to the project.
Because of the limited electricity available from the local power grid, auxiliary generators are being supplied so that the acid and lithium processing plants can start up without grid power. “However, after the acid plant starts up,” Mutler said,” the steam turbine generator can be brought online to provide the power needed to operate the sulfuric acid plant at de-
sign rates and to provide surplus electricity to the remainder of the site facilities.”
Limited local electricity supply also prompted designers to allow for a future MECS® HRS™ system to capture high-temperature waste heat from the sulfuric acid production process and convert it into low-pressure steam. An MECS® HRS™system boosts energy efficiency, lowers operating costs, and may reduce greenhouse gases.
The remote location of the site means limited access to industrial facilities, so critical strategic spares, like the main blower rotor, were purchased as part of the initial capital expenditure.
“And there are very strict noise limitations,” said Mutler, “so we included silencers on all steam safety relief valves; and all major rotating equipment will have noise attenuated by either enclosed buildings or sound enclosures.”

One challenge is ensuring equipment is supplied to precise specifications. “LAC and EXP have spent a lot of effort to bring high quality equipment for this facility. And we have been performing inspections at supplier shops located internationally, including in China, Malaysia, and Turkey,” said Jones.
One of the most anticipated challenges is transporting oversized equipment from a west coast port to the site. But the plan is to ship in modules as large as possible within size limitations.
“A good example was the 44 ft diameter, 304H SS converter,” said Mutler. “Each pass was shipped to the site in two halves.”
From an operational standpoint, syncing lithium processing with the sulfuric acid plant




represents a significant milestone. “The biggest challenge is going to be getting the lithium processing plant to the minimum stable operating rate of the acid plant so that the number of acid plant starts and stops can be minimized,” said Mutler. “The current strategy is to postpone the startup of the acid plant, by importing acid, until the processing plant can operate at the minimum stable acid plant rate.”
The team is also working on how to attract experienced operators to the plant, train them and retain them.


Major construction is well advanced at phase 1 with all major foundations completed and equipment delivery continuing through early 2027. Mechanical completion is targeted for late 2027, with full ramp-up to production through 2028.
Community investment has been integral in the project's development. LAC is working closely with the local tribes on cultural resource management and providing employment opportunities throughout the region. LAC has also partnered with Humboldt County and local communities to address infrastructure needs.
year, targeting the new facility to be open for the 2027-2028 school year.
"This project demonstrates how industrial development can coexist with, and actively support community well-being," Jones said. "The new school will not only mitigate traffic impacts, but is an investment that demonstrates our long-term commitment to the community." q
“The key to the success of this project is the early engagement of the operations team,” said Matt Karins, Sulfuric Acid Plant Manager, Lithium Americas.
Toward that end, Elessent will be providing MECS® operator training using lifelike computer simulation. “The training allows operators to start and stop the plant, change rates, and train on simulated emergency scenarios such as boiler, economizer, and superheater leaks. The simulator will also be used to test the plant interlocks and instrumentation prior to starting up the sulfuric acid plants,” said Horne.

One significant community investment addresses school safety concerns. The existing Orovada K-8 school sits near the intersection of Highway 95 and State Route 293, where increased industrial traffic from Thacker Pass operations posed potential risks to students. To mitigate this impact, LAC partnered with the Humboldt County School District, the Bureau of Land Management, and the Orovada community to relocate the school to a safer site on BLM-donated land. The Community Foundation of Northern Nevada is helping ensure the project serves long-term community needs. Construction on the school begins later this

By: Fiona Boyd and Freda Gordon, Directors of Acuity Commodities
In our last article in the Fall/Winter issue of Sulfuric Acid Today, we noted that in 2026, emphasis would remain on tight concentrate availability, namely copper, sulfur price direction, and ongoing uncertainty in the geopolitical landscape. Since the last article, events have exacerbated the impact of some of these conditions, highlighting them as key issues during the first few weeks of 2026.
In terms of tight copper concentrate availability to feed smelters, this has overhung the market since 2024. Smelters continue to have to increase revenues from other product streams as they see limitedto-no revenue from treatment charges (TC) and refining charges (RC). Most annual benchmarks were set at zero, following the emergence of negative spot TC/RCs in 2025, which remains ongoing. This is as copper prices are firm because of supply disruptions, such as from the Grasberg mine in Indonesia as discussed in our last article, and firm demand with a bullish outlook on factors such as electrification and AI.
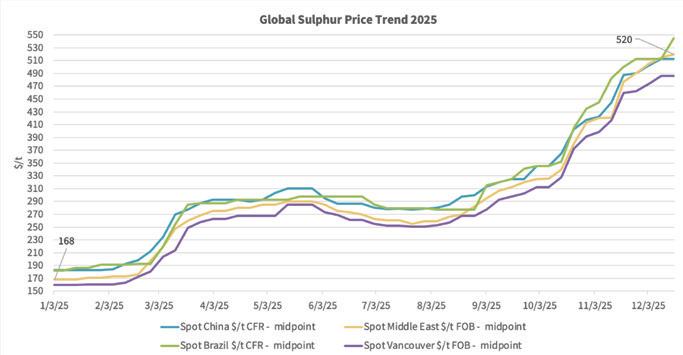
What has overshadowed the tight copper concentrate availability, however, is sulfur price direction, with recent pricing at levels not seen since 2008, the year of the global financial crisis. As an indication, the Tampa molten sulfur contract price for 1Q26 settled at $495.69/lt DEL, representing the highest price since 3Q08 at $617/lt DEL.
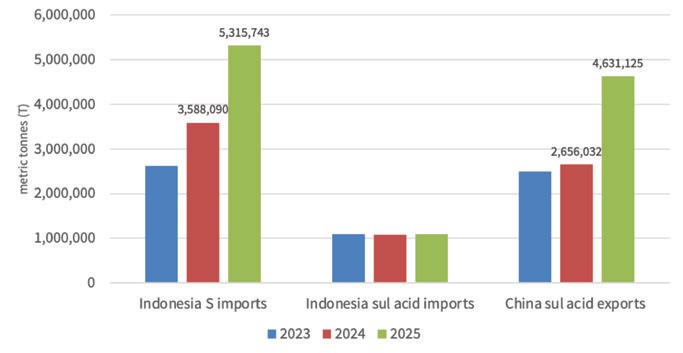
We noted in our last article that a key driver of the sulfur price run up, which saw prices move up mostly continuously since March 2025, was strong demand from Indonesia to support nickel leaching. Last year, Indonesia imported just over 5.3m t of sulfur, up an impressive 1.7m t from the volume imported in 2024.
But in addition to this higher demand, sulfur supply constraints have escalated since 4Q25, further providing price support. A key event has been Russia imposing a sulfur export ban as its own domestic production is reduced. Russia’s sulfur production has been constrained through Ukraine’s drone attacks on its refineries and other petrochemical complexes that impact upstream operations. Not only has this reduced sulfur production in Russia, but has resulted in some import sales, highlighting the shortness. This is an example of geopolitical uncertainty impacting the commodity markets.
With elevated sulfur prices sustained, it has certainly given sulfuric acid prices some support in tandem. However, participants in both markets are now increasingly focused on sulfur price direction moving forward.

Fiona Boyd, Acuity Commodities
When sulfur price peaks were last seen in 2008 and 2022, they were quickly reversed by demand destruction from phosphate fertilizer producers, the largest sulfur-consuming sector from the phosphate fertilizer sector. Earlier we noted the Tampa molten sulfur price settling at the highest level since 2008. This quickly was followed by it moving down to $150/ lt DEL in 4Q08 and to $0/lt DEL by 1Q09 triggered by demand destruction from the phosphate fertilizer sector. While firm phosphate prices throughout most of 2008 supported firmer sulfur pricing, as those downstream prices eroded, production declined, and sulfur consumption slid.
What is different now than in both 2008 and 2022 is there is more competition for sulfur supply from the mining sector, as the growth in Indonesia illustrates, who may have a different price tolerance than other consumers.
Amid firm sulfur demand, supply remains snug. Although on paper sulfur production continues to grow, namely supported by growth in refining and natural gas processing capacity in the Middle East, less production in key hubs such as Europe, Russia, and the United States certainly has impacts on sulfur market trade flows and pricing. The situation in Russia is also more long-lasting than, for example, a temporary supply shortage triggered by
weather events, such as a US hurricane, further contributing to the price stability.
With the sulfur outlook firm, sulfuric acid prices were in catch up mode as 2025 was winding down, which carried into 2026. Against the backdrop of relying on revenue from acid amid the TC/RC environment, smelter acid producers have been able to point to sulfur price values to support price expectations.
Adding fuel to the fire, concerns around sulfuric acid exports from China emerged in November 2025, amid safeguarding national food security. This ultimately resulted in a quota system in place for sulfuric acid exports from China, now in place through April. China’s relevance in the market has increased in recent years alongside the growth in base metal smelting capacity. For illustration: China exported over 4.6m t of sulfuric acid in 2025, up a startling 2m t from the 2024 level.
With tighter smelter acid availability from China, other export prices are seeing a boost. This is despite still an ongoing lack of spot demand from Morocco’s OCP. OCP has largely been out of the market since August 2025; this initially resulted in northwest European FOB pricing dipping in 4Q25. However, with the supply picture in Asia becoming more complex, driving pricing there up, this allowed for European

Freda Gordon, Acuity Commodities
material to flow to other markets, namely Chile, which in turn drove that pricing up as well.
As we look to 2H26, outside of sulfur price direction, we are following several key factors on the supply and demand sides that will impact the market.
In terms of supply, on the smelter acid side, focus will be on if sulfuric acid exports from China increase after April. We will also be watching for any notable uplift in smelter acid availability from India and Indonesia pending smelter ramp ups there. Some of the latter will be tied to copper concentrate availability from domestic production, with the Grasberg mine expected to enter a phased restart in 2Q of an underground area, aiming for around 85% of production to be online in 2H. An improvement in smelter acid supply from China, India, and Indonesia, could pressure down export values, but other supply factors, and of course demand, will factor in.
Outside of smelter acid supply, we are tracking a restart of Minera Boleo’s copper operations in Santa Rosalia, Mexico, which includes a sulfur-based acid plant. It has been offline since July 2025 on an unplanned basis, with a restart targeted for 2Q. Excess sulfuric acid produced at the site is exported offshore, largely to Chile, as well as cabotage movements. Its absence from the market has been largely unnoticed given the supply conditions in the key export regions.
Meanwhile, in Brazil, Unigel continues to pursue start up of a sulfur-based plant in the Aratu region, despite its precarious financial position that continues to stall progress. Upon successful ramp up, the plant would reduce import needs into northern Brazil.
Finally on the supply side we note more steady supply from PetroPeru’s Talara refinery in Peru, which began to export sulfuric acid in 4Q25 after several delays. At least three cargoes had moved at the time of writing, suggesting more regular supply will be seen from this new production source in 2026.
Alongside any improvement in smelter acid supply, the restart of Minera Boleo, a ramp up of Unigel, and more regular supply from Talara signal a potential increase in supply availability.
On the demand side, we expect to see firm needs in Chile for 2H. While historically many large end users have covered significant amounts of their consumption needs with annual contracts, the disconnect between Asia FOB pricing and implied delivered pricing in Chile stalled discussions. Suppliers of Asian smelter acid were

By: Aida Barberena, Marketing Specialist, and Jeremy Schneider, Senior Technical Services Engineer, Elessent Clean Technologies
Across sulfur burning, metallurgical, spent-acid, and wet-gas sulfuric acid plants, catalyst performance remains central to achieving low SO2 emissions, regulatory compliance, high production rates, and on-stream time. Plant operators monitor SO2 emissions and, as the catalyst degrades, adjust converter temperatures to maintain SO2 emissions within compliance limits. When temperature adjustments no longer work, plant capacity must be reduced. Thermal cycling is one of the operational factors that has a long-term impact on catalyst health and performance. The impact of thermal cycling is persistently underestimated.
Every plant shutdown and restart results in a cooling and heating cycle. These thermal cycles, whether triggered by upstream interruptions, contamination-driven maintenance, or operational choices, exert physical and chemical stresses on the catalyst. These thermal cycles also heat and cool all the metal ducting and equipment in the plant, causing them to expand and contract. These expansions and contractions result in cracking, gas leaks, and more plant shutdowns and thermal cycles. Staying out of this repeated circle of thermal cycles is critical to long-term operation. Specifically for the catalyst, these thermal cycles result in degradation of the catalyst diatomaceous earth support. This degradation leads to increased pressure drop and loss of catalyst activity, which leads to reduced plant capacity, more cold plant shutdowns, and more required catalyst screening and replacement.
This article explains how sulfuric acid catalyst is engineered, how thermal cycling alters it over time, why shutdowns are occurring more frequently in some operating environments, and what practical steps plants can take to extend catalyst life without sacrificing production goals.
Modern sulfuric acid catalysts, such as the MECS® portfolio, are the product of over a century of development in materials science, pellet geometry, and vanadium chemistry. For 100 years, MECS® catalysts have reliably and economically converted SO2 to SO3 across diverse plant types. That longevity reflects continuous innovation
unwilling to match prices agreed between Chilean consumers and regional smelter acid producers. At the same time, the buyers were able to turn to Europe because of the absence of Morocco’s OCP from the market as noted earlier.
However, the European supply was not enough to cover full needs, and many suppliers of that material were unwilling to commit to annual contracts at fixed prices on the expectation that OCP will return to the import market in 2Q. As a result, there is demand left to be covered in Chile ahead, particularly with the lack of Asian supply. This is of course bullish in terms of demand.
Another bullish demand factor is more requirements in Australia due to more import needs into Kwinana stem-
around four performance characteristics: activity, pressure drop, hardness, and service life.
At its core, sulfuric acid catalyst uses a vanadium-containing molten salt, supported on a porous diatomaceous earth (DE) or silica-based substrate. This provides:
• High porosity and surface area to expose active sites
• Thermal stability across industrial temperature ranges
• Mechanical strength to resist crushing and dust formation
• Mass transfer efficiency, maintained by keeping open pathways for gas flow
Catalyst stability and durability depend on how the active vanadium salts are dispersed, a selection of DE that optimizes surface area, and how the pellet geometry manages both flow resistance and exposed area. The MECS® family of catalysts are designed for extended campaign duration and resilience, but even the best catalyst formulation and support media have limits when subjected to repeated heating and cooling cycles.
Sulfuric acid catalyst is affected by thermal cycles through three interconnected mechanisms:
1) Breakdown of the support structure through thermal shock: The diatomaceous earth (DE) or silica-based support is designed for high porosity and thermal stability, but it is not immune to repeated temperature cycles. Each time temperatures rise and fall, the support expands and contracts. Over enough cycles, this leads to:
• Microcracking and structural fatigue, because the pellet’s internal framework is stressed repeatedly.
• Collapse or partial melting of the support at higher temperatures can transform the once porous structure into a dense, amorphous mass.
• Loss of porosity, which means fewer pathways and fewer accessible active sites for SO2 oxidation.
• Increased pressure drop as the bed begins to compact or generate dust; and lower conversion as the effective surface area declines.
2) Vanadium oxidation state shifts during temperature swings:
ming from BHP putting its Nickel West operations on care and maintenance as reviewed in our last article.
On the other hand, while Glencore was able to secure the fate of its Mount Isa smelter for the next three years through a government support package, the fate of Phosphate Hill remains unknown. It processes metallurgical gas from the smelter to support fertilizer production and if no buyer is found by the end of 1Q, it is preparing for a September 2026 closure.
Also, alongside the bearish tone, around mid February, the time of writing, indications of quotas around nickel ore mining in Indonesia were emerging. This is as nickel production there has grown significantly and there is focus on reducing supply to provide price support.
For SO2 oxidation to proceed efficiently, vanadium must remain in its most reactive oxidation states. Thermal cycling disrupts this balance as follows:
• Temperature swings shift the oxidation state distribution, increasing the fraction of less reactive vanadium species. These chemical changes reduce catalytic performance even before any visible physical damage occurs.
• During cooling or dewpoint events with high acid mist or vapor present, the active vanadium-based melt phase can cool and redistribute unevenly, causing localized zones of reduced activity.
• If acid condensate forms, active salts can be leached or washed from the pellet surface, accelerating deactivation. Thus, even in the absence of structural failure, the catalyst can lose reactivity simply because temperature fluctuations destabilize the chemistry of the active phase.
3) Thermal cycling amplified by contamination: Thermal cycling rarely acts alone in real operating environments. In many plants, contaminants such as ash, dust, iron oxide, or trace elements are already present in the gas stream. These contaminants introduce mechanical and chemical stress on the catalyst.
• “99% pure” sulfur contains some solid contaminants. These solids become ash after sulfur combustion and lodge in the catalyst pore structure, increasing pressure drop and stressing the pellet.
• Maintaining an effective gas cleaning system is vital in metallurgical and spent acid plants to remove impurities from process feeds before they can interact with the catalyst, preventing reductions in conversion efficiency or mechanical strength. Even in sulfur burning plants, efficient filtering of the combustion air is critical to catalyst life.
• The need for frequent shutdowns to screen or replace contaminated catalyst introduces additional thermal cycles, which compound the existing degradation issues. The catalyst experiences not only thermal stress but also two simultaneous degradation pathways: thermal cycling and accumulated contaminants.
This could potentially impact demand for imported sulfur and sulfuric acid. Indonesia’s sulfuric acid imports were steady in 2025, despite its notable growth in sulfur-based capacity, which provided further demand and price support for Asian smelter acid.
Finally in demand, a slightly bearish tone around acid consumers in the industrial sector in Europe cannot be ignored as it continues to grapple with rising input costs and competition from producers with much lower costs of production, mainly in Asia.
Considering the above, it is evident that there are various levers on the supply and demand sides of the sulfuric acid market that will shape the rest of 2026. This is as all eyes will remain on sulfur
pricing to see if levels hang on, despite ongoing affordability concerns.
Acuity Commodities provides insight into the sulfur and sulfuric acid markets through price assessments, data, and supporting analysis. Offerings include weekly reports on the global sulfur and sulfuric acid markets. For North America, we offer a bi-weekly report on sulfur and sulfuric acid as well as a monthly report on industrial chemicals, including caustic soda and hydrochloric. We also have developed a database detailing battery and mining projects under development in North America that could consume products such as sulfuric acid. In addition, Acuity does bespoke consulting work. Please visit www.acuitycommodities.com for detailed information. q
In one observed case, a plant experienced nearly ten shutdowns within a single year. These repeated outages were not the result of poor operational practice within the acid plant itself, but rather frequent upstream process upsets that repeatedly interrupted the supply of hot process gas. Each interruption forced the catalyst through another temperature cycle. Over time, the cumulative impact severely weakened the catalyst structure, ultimately degrading it into essentially solidified dust, forcing the plant to replace the entire catalyst content prematurely. This anecdote illustrates how dramatically repeated thermal cycles can shorten the lifespan of a catalyst bed.

Most acid plants are designed and operated with the expectation of long continuous runs; but in many regions and applications, the number of interruptions can be much higher.
Acid plants dependent on metallurgical off-gas or power-related streams face outages when upstream conveyors, furnaces, or boilers are stopped or slowed. When hot SO2 gas ceases, the catalyst begins to cool unless the plant proactively maintains a high temperature.
High variation of sulfur quality available in the market can also contribute to shutdowns. If sulfur is not effectively filtered, solids will accumulate on the catalyst, increasing pressure drop. Accelerated pressure drop in the catalyst can result in more frequent shutdowns.
Lastly, to reduce downtime, some operations may shorten heat-up times or expose the catalyst to wet heat up gases while it is below the acid dew point. These choices might save hours but could cost months in catalyst life and increase screening losses by amplifying thermal shock and the likelihood of acid dewpoint occurrence.
Each of these drivers increases cycle frequency or severity, which means cumulative damage to the catalyst.
Catalyst is one of the most significant consumables in an acid plant. Not maintaining cycle discipline and exposing the catalyst to repeated thermal cycles will increase catalyst maintenance costs. Even small decisions around sulfur filtration, process gas cleaning or combustion air
filtering can have big cost consequences. Let’s look at a short case:
Material Costs:
With escalating cesium raw material prices, the cost to do maintenance on even a small portion of a cesium bed can reach several hundred thousand dollars. For larger units or multiple beds, costs multiply quickly.
Indirect Costs:
• Lost catalyst activity, resulting in reduced capacity to meet emissions targets
• Increased catalyst pressure drop, reducing plant capacity and increasing blower power and operating costs
• Increased downtime with associated production loss
• Higher emissions variability during restarts, increasing startup time and reducing plant capacity
These costs can be high when compared with the cost of maintaining temperature during short outages with an external heat source or adhering to disciplined startup procedures. From an OPEX standpoint, avoiding deep or frequent thermal cycles is one of the highest return operational policies available.
The encouraging reality is that damage from thermal cycling can be minimized. The following operational practices to increase the catalyst life are recommended:
1) Maintain temperature during outages:
Minimize heat loss during short outages by minimizing draft through the plant. Consider using an external heat to extend these short outage times when practical. Before cooling below the acid dew point, purge the plant of SO2 and SO3. Field experience shows that keeping beds warm for days is practical and economical compared to premature catalyst replacement. Keep in mind to:
• Purge the plant and then isolate to minimize draft through the plant
• Avoid rapid cooling and reheating
2) Follow startup/shutdown procedures rigorously. Follow the manufacturer’s procedures and plant standard operating procedures. In particular:
• Heat the catalyst above the acid dew point with dry air before exposing the catalyst to wet combustion gases.
• Observe temperature gradient limits between zones and over time. Note that bed inlet temperatures should not exceed 530°C and temperature differences across division plates should not exceed 140°C
3) Control contaminants at the source:
Every contamination-driven shutdown triggers at least one more thermal cycle. Try to reduce these by:
• Sampling incoming sulfur by lot and include an in-line real-time monitoring system
• Introducing a polishing filter to reduce ash below 8 ppm in sulfur burning plants
• Addressing upstream sources of solids in the sulfur or combustion air
• Using a properly designed and effective gas cleaning system for removing impurities in metallurgical and spent acid plants
4) Coordinate with upstream operations:
Where the plant relies on external SO2 sources, establish clear communication protocols so supplemental heat can be initiated proactively rather than reactively.
• Temperature trending: Focus on changes in per-pass temperature rise during stable windows.
• Pressure drop surveillance: A pressure increase can be an early sign of dust accumulation from repeated cycles. Investigate promptly. Plant pressure surveys are recommended monthly at a minimum.
• Converter gas sampling and analysis (such as MECS® PeGASyS™ testing): Periodic targeted analyses provide clarity on gas composition changes that can otherwise be mistaken for activity loss. These campaigns are especially useful after several cycling events to inform maintenance planning.
• Shutdown inspections: When outages occur, use the opportunity to inspect for fine accumulation, pellet edge wear, and evidence of moisture or acid mist contact. Collect catalyst samples from each bed for quantitative analysis of activity and hardness.

Thermal cycling has significant consequences for catalyst degradation in sulfuric acid plants. The engineering behind modern sulfuric acid catalysts is impressive and proven, but cannot fully withstand repeated or poorly managed heating and cooling without accelerated wear.
The good news is that the most effective defenses are operational and practical: maintain temperature during short outages, abide by startup/shutdown procedures, control contaminants at their source, and coordinate closely with upstream operations. Plants that implement these practices consistently experience longer catalyst life, lower emissions, reduced maintenance frequency, and improved economics.
Minimizing thermal cycles and managing them with discipline when they do occur is one of the simplest, highest return strategies for protecting catalyst investments and ensuring reliable, efficient acid production over the long run.
For more information, please contact Jeremy Schneider, Senior Technical Services Engineer, Elessent Clean Technologies, at Jeremy.Schneider@elessentct.com. q

Heat stress is a serious and often underestimated occupational hazard in industrial environments. Facilities such as manufacturing plants, refineries, chemical processing units, construction sites, foundries, and warehouses frequently expose workers to high temperatures, radiant heat, humidity, and physically demanding tasks. When the body is unable to adequately regulate its internal temperature, heat stress can occur. Heat stress can potentially lead to heat exhaustion, heat stroke, reduced productivity, increased error rates, and even fatalities. With rising global temperatures and increased focus on worker well-being, managing heat stress is no longer optional. It is now a core component of an effective safety and health program. This article explores the causes of heat stress in industrial settings. It will also provide insight to the physical and mental impacts on workers and the practical strategies for prevention and control.
Prior to 2026, the Occupational Safety and Health Administration (OSHA) regulated heat stress through its General Duty Clause. Without specific regulations available for guidance, companies were left to come up with their own methods to manage heat stress. Particularly in the south, heat stress is a major hazard that our employees are exposed to daily. While exact percentages vary, heat related illnesses and injuries are a significant and often undercounted issue. According to OSHA, it is estimated that 1.18% of all workplace injuries were heat attributable above a 70-degree “F” index.
Although final regulations for 2026 aren’t set in stone, OSHA is moving toward a mandatory federal heat standard. The goal of this initiative is to provide employers with a detailed guideline for protecting employees from heat stress. The guideline will include Heat Injury and Illness Prevention Plans (HIPPS) for outdoor and indoor work settings with proposed rules requiring written HIPPS for all industries including heat triggers at 80°F. By the end of 2026, employers should expect regulation specifying mandatory water/shade/rest, acclimatization, training, medical monitoring, and paid breaks for high heat (90°F). OSHA will enforce these requirements by utilizing existing National Emphasis Programs (NEPS). As employers, we should be analyzing the programs currently in place and preparing for the new emerging heat stress requirements from OSHA.
Heat stress occurs when the body absorbs more heat than it can dissipate. The human body relies on sweating and increased blood circulation to the skin to regulate temperature. When environmental conditions or work demands overwhelm these mechanisms, internal body temperature rises which places strain on vital organs. Excessive heat in the workplace is known to cause an abundance of adverse health effects that include heat stroke and potentially death when proper precautions are not followed. Individuals who work outdoors, and even indoors, without the proper mitigations and climate controls are at risk of hazardous heat exposure. In certain settings, heat-generating processes that include machinery and equipment, such as furnaces, can add to hazardous heat risks when the proper cooling measures are not in place.
Industrial settings have various heat stress contributors. Some of these include extreme temperatures, high humidity, physical exertion, strenuous work tasks, upgraded PPE, and poor ventilation. Workers performing tasks near heat-generating equipment or outdoors under direct
By: Skyler Barker, CSP Safety Coordinator, VIP International
sunlight face compounded risk. The health effects of heat stress or heat related illnesses exist on a broad sequence. Symptoms derived from prolonged heat exposure escalate as the exposure increases.
Sulfuric acid plants present a unique challenge when it comes to managing heat stress. The handling of corrosive chemicals requires extensive uses of various PPE. The PPE required consists of acid-resistant suits, gloves and boots, face shields or goggles, and respirators. Acid suits are also typically worn on top of uniforms, that in some cases consist of flame resistant (FR) shirts and pants, which are not only long sleeve but usually thicker than normal clothing. The use of some PPE may increase the burden of heat on workers; and that is not limited to the summer months. Elevated temperatures exist year-round due to exothermic reactions, radiant heat, confined spaces, and maintenance conducted during turnarounds. When you combine these potential circumstances with the required acid-resistant PPE, respirators, and protective clothing used against chemicals, the risk of having heat-related illness significantly increases. Having the proper PPE and required chemical protection are essential; however, they also trap heat and increase body temperature even during moderate workloads.
Process generated heat sources include, but are not limited to, furnaces, boilers, ducting, and process piping. Even when these process components are properly insulated, they can substantially elevate the temperatures in work areas. Confined and enclosed workspaces have limited airflow which limits the ability for heat to dissipate and increases the likelihood of heat stress during maintenance activities. Heat stress does not only affect the employees’ health, but it can also compromise overall jobsite safety. Fatigued and overheated workers can exhibit reduced situational awareness, slower reaction times, and poor decision-making increasing the likelihood that procedures will not be followed. In an environment where following procedure is critical, the effects of heat stress could contribute to equipment damage, releases, and/or exposure events.
When mitigating heat stress, it is important to identify its different stages and their associated symptoms. The three stages include heat cramps, heat exhaustion, and heat stroke. Identifying the stage is important in determining the correct course of action. Distinguishing between the stages can be critical in ensuring the employee is receiving proper care. Typically, heat cramps are distinguished by muscle spasms or cramps. These normally occur in the legs, arms, or abdomen. Heat cramps can be remedied by stopping activity, cooling down, and hydrating. A more severe stage is heat exhaustion, shown by sweating, extreme thirst, elevated heart rate, nausea and/or vomiting, and dizziness. Heat exhaustion requires the employee to stop working, hydrate, and cool down with ice packs. The most severe stage of heat stress is heat stroke. Symptoms of heat stroke include: altered mental state such as confusion, fainting, seizures, sweating, red skin, and a high body temp. If an employee presents signs of a heat stroke, the facility ERT should be notified, and first aid should be given until they arrive, but only if you are trained to do so. Being able to identify these symptoms can make a difference in life and death, and it is imperative that both supervisors and
employees be able to identify these symptoms. It is crucial that, as employers, we train our employees to identify the signs and symptoms of heat stress. Staff should not only be able to identify the key indicators, but they should also know the steps to take once they are identified.
It is important that employers implement formal heat stress assessments to consider things like ambient temperature, humidity, radiant heat from equipment, air movement in the work area, work intensity, and the PPE required. These assessments may be jobsite specific and not a blanket tool, but should be used to establish a safe work and rest cycle to avoid employee burnout and fatigue. It is crucial to monitor and give special attention during summer operations, turnarounds, unplanned outages, non-routine maintenance, and any emergency response activities. Supervisors must closely monitor conditions and their employees to be able to identify the early signs of heat stress and intervene in a timely manner.
Engineering controls can also reduce risk. Engineering controls include but are not limited to: exhaust ventilation near heat sources, improved ventilation in operating areas, air-conditioned control rooms and rest areas, and temporary ventilation during maintenance activities. Other controls that can be put into place are things such as insulation and radiant heat reduction by maintaining insulation on hot surfaces and ducting as well as putting in reflective shielding where insulation may be impractical. In some scenarios, it may be feasible to automate samplings and valve operations which will reduce the need for prolonged presence near hot equipment.
While employee monitoring and engineering controls are important, it is also important to have administrative controls in place. Effective administrative controls include but are not limited to: implementing set work and rest schedules, increasing breaks during times when temperatures are higher, job rotation to limit exposure duration without delaying work, and scheduling high exertion tasks during cooler periods. Employees who are new to the industry may require a 7-to-14-day period to get acclimated. Their work intensity and duration should be limited, and they should be closely monitored by supervisors. Hydration management plays a critical part in managing heat stress. Employers should provide cool water near work areas and encourage water breaks frequently. Employees should also be given a safe and designated area to remove PPE, cool down, and hydrate.
Heat stress is a serious hazard in sulfuric acid plants due to the high-temperature processes, confined workspaces, and extensive PPE requirements. Without leadership and effective controls, heat stress can lead to severe health issues, increased errors, and elevated safety risks. Heat stress controls should be reviewed regularly to ensure effectiveness. These procedures should be reviewed particularly after extreme weather events, as well as process changes and expansions. By implementing an approach that prioritizes training, engineering controls, appropriate PPE, and a strong safety culture, sulfuric acid plants can significantly reduce heat stress exposure. Proactive management not only protects workers’ health, but it will strengthen operational reliability and reinforce the industry’s commitment to a safe and responsible work environment.
For more information, please visit www.vipinc.com. q

Lewis® molten sulfur and sulfuric acid share similar physical properties. Except for specific gravity, both fluids behave comparably to water at normal pumping temperatures. However, the higher specific gravity, typically 1.8 or greater, results in increased power requirements, influencing hydraulic efficiency and demanding rugged mechanical construction.
Pump designs optimized specifically for molten sulfur and sulfuric acid services, combined with carefully selected construction materials, ensure high operating efficiency, long service life, and reliable performance under demanding conditions.
Pump design
Lewis® vertical submerged process centrifugal pumps with a separate discharge pipe, known as VS4 and VS5 (API Standard), are commonly used for molten sulfur applications. This configuration eliminates the need for a pressurized shaft seal, which is typically required in horizontal pumps. The pumped fluid is bypassed back to the sump before reaching the seal cavity— reducing leakage risk.
Horizontal pumps equipped with packed stuffing boxes, mechanical seals, or dynamic seals have proven challenging due to sulfur leakage, which can lead to housekeeping and maintenance challenges.
A typical vertical pump design for clean molten sulfur, containing minor solid impurities includes:
• A closed impeller for optimal efficiency
• Replaceable wear rings to maintain hydraulic performance
• Efficiencies approaching 80–85% at best efficiency point (BEP) for larger units
Properly engineered wear rings help balance axial thrust loads on the impeller and extend thrust bearing life.
A submerged sleeve bearing and journal assembly is installed adjacent to the impeller to provide shaft support. Additional intermediate bearings may be used depending on shaft length, shaft diameter, bearing span, and operating speed. These bearings are lubricated by the pumped sulfur, which is forced upward through the shaft column by pump pressure. Excess sulfur is returned to the sump or tank through a bypass overflow orifice located above the uppermost submerged bearing.
Standard pumps are available with setting lengths up to 7 meters using a single-piece shaft. Longer settings can be accommodated using segmented shafts and rigid shaft couplings.
Contaminated sulfur applications
For sulfur containing abrasive solids, a vertical cantilever shaft pump is recommended. This design eliminates the need for close-clearance submerged
By: Marwan Karaki, Director of Outside Sales, Weir
bearings. Axial and radial hydraulic forces are transmitted through a large-diameter shaft to externally lubricated ball bearings.
Key design features include:
• Semi-open impeller capable of passing solids up to 6 mm diameter
• Externally adjustable wear plate clearance to maintain optimal efficiency
• Pump-out vanes on the impeller back shroud to reduce axial loads
• Double volute casing to minimize radial hydraulic forces and extend bearing life
Cantilever pumps are available up to a maximum setting length of 2.15 meters, with optional suction pipe extensions to increase effective operating depth.

Installation and heating
Molten sulfur pumps are typically mounted on flanged nozzles at the top of steel storage vessels or over concrete pits.
To prevent sulfur solidification above the operating liquid level, both the shaft column and discharge pipe are steam jacketed.
Lewis® materials of construction: molten sulfur
Molten sulfur is generally noncorrosive, making carbon steel and cast iron appropriate for clean sulfur service. Material selection focuses on adequate strength and ductility. Typical materials include:
• Impellers and wear parts: ASTM A48 Class 35 cast iron
• Pump shaft: AISI 1146 carbon steel
• Shaft column and discharge pipe: ASTM A106 Grade B pipe (or equivalent); ASTM A105 flanges (or equivalent)
These materials have been successfully used for recovering sulfur from natural gas and crude oil with trace hydrogen sulfide content and have demonstrated reliable long-term service performance.
For abrasive or dirty sulfur service, wear-prone components such as impellers and wear plates are manufactured from ASTM A743 Grade CA40 (12% chromium martensitic stainless steel), heat treated to a minimum hardness of 500 BHN to
maximize abrasion resistance.
In applications involving both corrosion and abrasion, AISI 316L stainless steel is typically used for casings and lowvelocity parts, while high-velocity rotating components are manufactured from Lewmet® alloy, described further in the sulfuric acid section.
Pump design
Lewis® vertical process centrifugal pumps are generally preferred for sulfuric acid applications. Design considerations are similar to those used for clean molten sulfur service, with additional emphasis on corrosion resistance and dimensional stability.

Lewis® materials of construction: sulfuric acid
Centrifugal pumps handling sulfuric acid face extremely demanding service conditions. Components must withstand both chemical corrosion and fluid velocities exceeding 30 m/s under highly turbulent flow. Maintaining tight dimensional tolerances is critical for achieving maximum hydraulic efficiency and long-term reliability.
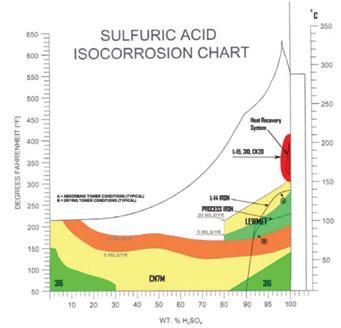
Sulfuric acid plants typically operate with acid concentrations ranging from 93% to 99.5%, at temperatures up to approximately 220°C. Within this narrow concentration range, corrosion behavior varies significantly.
Corrosion resistance in sulfuric acid is strongly linked to oxygen solubility, which decreases sharply as acid concentration drops below approximately 99%. Alloys with strong oxidation resistance—primarily due to chromium and silicon—offer superior performance in these environments. Additional alloying elements such as molybdenum and copper enhance resistance in mildly oxidizing or impurity-containing conditions.
Common alloy applications
• Chromium–molybdenum alloys: Used in 93–98% sulfuric acid at temperatures up
to approximately 140°C, these alloys rely on the formation of stable, adherent oxide films for corrosion resistance.
• Alloy 20: Introduced in 1940 and widely applied in sulfuric acid service, however, this alloy’s corrosion rates increase rapidly at temperatures above 90°C and fluid velocities exceeding 5 m/s in 98% acid.
• Lewmet ® alloy: Introduced in the early 1970s, this alloy provides excellent corrosion resistance even at high fluid velocities. It can be age-hardened to 500 BHN without compromising corrosion performance, offering superior galling and wear resistance for close-tolerance pump components.
• High-silicon stainless steels: Effective in 98% sulfuric acid at temperatures up to approximately 140°C, this material’s corrosion rates increase significantly at lower acid concentrations, above 99.5%, or in oleum service. These alloys are also sensitive to halogen impurities.
• High-chromium alloys: Applied in high-temperature heat recovery service (98–99.5% acid at 180–220°C), a slight decrease in acid concentration can dramatically increase corrosion rates, particularly at high velocities.
• Cast and specialty irons: Cast iron, ductile iron, and specialty irons continue to be used successfully in concentrated sulfuric acid service. L-14 iron, introduced in the mid-1980s, is suitable for components where dimensional stability is less critical, such as pump casings and discharge piping, at temperatures up to approximately 132°C.
Pump designs developed specifically for molten sulfur and concentrated sulfuric acid service, combined with properly selected materials of construction, provide reliable and efficient equipment for these demanding applications.
Specialized hydraulic and mechanical design minimizes corrosion, erosion, and mechanical wear, resulting in long service life and reduced risk of catastrophic failures in harsh operating environments. For more information, visit www. global.weir.com. q

In sulfuric acid production, major investments typically focus on converters, heat recovery systems, or emission control. Yet one of the most decisive performance factors remains comparatively small and often underestimated: the sulfur atomizer nozzle.
Sulfur combustion defines the thermal profile, SO2 generation rate, refractory lifetime, maintenance intervals, and ultimately the production capacity of the entire plant. When atomization quality improves, the effects cascade throughout the process. Recent retrofit projects by CS Combustion Solutions demonstrate how modern nozzle technology can serve as a true capacity lever without requiring large-scale structural modifications.
Efficient sulfur combustion depends on fine and uniform atomization of molten sulfur. Droplet size determines evaporation speed, mixing intensity with combustion air, and the completeness of conversion to SO2
If droplets are too large, evaporation slows down. Sulfur particles may travel deep into the furnace before reaction completes, creating localized hot spots when they impinge
on refractory surfaces. The consequences are well-known: thermal stress, refractory damage, unstable flame geometry, higher maintenance frequency, and operational limitations.
Traditional atomization technologies involve compromises. Rotary-cup systems generate relatively fine droplets but rely on moving components operating in high-temperature environments, leading to higher maintenance intensity and energy demand. Conventional pressure nozzles are mechanically simple and cost-efficient, but often produce coarser droplets and show greater sensitivity to clogging and wear.
Operators have therefore historically faced a trade-off between atomization quality and economic simplicity.

The SR-P sulfur atomizer nozzle was developed to eliminate this compromise. It

combines the fine droplet generation typically associated with ultrasonic systems with the robustness and cost efficiency of pressurebased designs.

With a characteristic droplet size of approximately 150 μm, the SR-P enables extremely rapid evaporation and oxidation of elemental sulfur. The accelerated transformation of sulfur into SO2 has a decisive technical consequence: SO2 is completed earlier within the furnace volume.
This faster reaction kinetics provides two major advantages:
First, it reduces the required effective combustion chamber length. Because sulfur conversion takes place in a shorter distance, furnace designs can be more compact in new installations. Smaller combustion chambers mean lower capital expenditure, reduced refractory surface area, and optimized thermal management.
Second—and even more relevant for existing plants—faster sulfur-to-SO2 conversion unlocks hidden capacity. In many brownfield facilities, the furnace volume limits throughput because combustion must be completed before the gas stream enters downstream equipment. By accelerating the reaction, the SR-P allows higher sulfur feed rates while maintaining complete combustion and stable temperature profiles. In practice, this enables measurable capacity increases without extending or replacing the furnace shell.
The homogeneous spray pattern further supports uniform flame geometry, minimizing localized overheating and protecting refractory linings. Reduced hot spots translate directly into longer lining lifetime and fewer unplanned shutdowns.
Another key advantage is operational flexibility. The SR-P operates reliably across a wide turndown range (approximately 20% to 110% load) allowing plants to adapt to variable production demands without compromising spray quality.
Importantly, the SR-P achieves these benefits without rotating components. The absence of moving parts reduces wear, simplifies maintenance, and lowers auxiliary energy consumption compared to rotary systems.
Field experience confirms the theoretical advantages.
In one retrofit project within an existing spray-gun facility, replacing conventional nozzles with advanced CS atomization systems enabled a throughput increase of approximately 25%, achieved with only minor equipment modifications. Beyond the pure capacity gain, operators reported
extended nozzle lifetime, longer maintenance intervals, improved operational stability, and more uniform furnace temperature distribution.
In another installation, an aging rotary-cup system was replaced by CS nozzle technology. Eliminating rotating equipment from the hot zone significantly reduced downtime and maintenance complexity. The improved spray performance stabilized combustion, minimized refractory hot spots, and extended run periods between shutdowns.
These examples illustrate a crucial point: capacity expansion does not necessarily require major structural revamps. In many cases, optimizing atomization is sufficient to remove the true bottleneck.
From an economic perspective, advanced atomization delivers value on multiple levels:
• Increased acid production capacity per unit investment
• Reduced downtime and maintenance cost
• Lower spare parts consumption
• Extended refractory lifetime
• Improved operational safety through stable combustion
Because the SR-P combines fine atomization with mechanical simplicity, both capital and operating expenditures remain controlled. The return on investment is particularly attractive for brownfield plants where large-scale furnace modifications would otherwise be required to increase throughput.
In new plant designs, the possibility of smaller combustion chambers further enhances project economics by reducing material requirements and installation effort.
In an industry under constant pressure to improve efficiency, reduce emissions, and optimize cost structures, operators must extract maximum performance from existing assets while ensuring long-term reliability.
Modern atomization technology proves that combustion optimization does not necessarily require large-scale revamps. By accelerating the conversion of elemental sulfur to SO2, improving spray homogeneity, and stabilizing thermal profiles, the SR-P nozzle enables higher throughput, smaller required furnace volume, extended refractory lifetime, and reduced maintenance effort—all through a targeted equipment upgrade.
The detailed technical background and industrial case studies behind these results will be presented by CS Combustion Solutions at the upcoming Sulfuric Acid Roundtable, where retrofit strategies and measurable performance improvements will be discussed in depth.
For more information, please visit www. comb-sol.com. q

Persistent fouling and cold-end corrosion are common challenges in gas-togas heat exchanger service, particularly where low metal temperatures at the cold tube sheet coincide with uneven thermal gradients and localized condensation. In these conditions, condensation promotes sulfate formation and particulate adhesion, accelerating fouling, increasing corrosion risk, and ultimately reducing heat transfer effectiveness. Over time, these mechanisms contribute to rising pressure drop, increased maintenance requirements, and shortened equipment life.
To address these issues, a refined exchanger configuration places the hot gas on the shell side while routing the cold gas through the tubes. This arrangement allows the hot shell-side gas to be used intentionally to warm the cold tube sheet through an integrated downcomer. Rather than relying solely on passive insulation or
By: Joan Bova, VP of Sales and Engineering, CG Thermal LLC
material upgrades, this approach actively manages metal temperatures at the cold end. By maintaining the cold-end tube sheet and surrounding metal temperatures above the gas dew point, the likelihood of condensation-driven fouling and corrosion is substantially reduced.

A gas-to-gas heat exchanger configured with shell-side hot gas and an integrated downcomer to actively manage cold-end metal temperatures above the dew point, reducing condensation-driven fouling and corrosion.
The design incorporates a variablepitch tube layout with radial shell-side flow, combined with a proprietary baffle system and downcomer to promote uniform heat distribution. Radial flow produces more symmetrical temperature profiles across the tube sheet and tube bundle. Radial flow also minimizes localized thermal gradients that can lead to high stress concentrations at tube-to-tube sheet connections. Variable tube pitch further improves flow distribution, reducing localized velocity peaks while enhancing thermal uniformity. Together, these features improve mechanical stability, reduce vibration risk, and promote predictable and uniform thermal expansion behavior under operating conditions.
Nathan Porter serves as Principal Engineer at Porter Process Technologies, an independent consulting firm based in Austin, Texas, specializing in sulfuric acid process technologies. The firm provides comprehensive services, including front-end engineering packages, process simulations, equipment specifications, debottlenecking studies, owner’s engineering, plant design, operations support, and modernization utilizing advanced sulfuric acid technologies.

Nathan Porter, Principal Engineer, Porter Process Technologies
Porter brings 20 years of sulfuric acid industry experience, including his first nine years as a process engineer at MECS (now part of DuPont), where he developed foundational expertise in sulfuric acid facilities. During his tenure at
MECS, he contributed to basic engineering designs, led commissioning and start-up activities for sulfuric acid plants, and spent time in the research group developing new process tools, technologies, and patented innovations. His project work encompasses all types of sulfuric acid plants, project phases, and plant renovations.
This depth of experience enables Porter Process Technologies to deliver integrated, high-value support to clients across consulting, detailed engineering, and project execution disciplines in the sulfuric acid industry.
For more information, visit porterprocess.tech. q
To further optimize material selection and fouling control, a dual-shell configuration can be employed. In this arrangement, the warmer shell section is exposed only to temperatures above 175.5 °C, allowing it to be fabricated from carbon steel without compromising corrosion resistance. A secondary 304 stainless steel shell incorporates a downcomer to ensure metal temperatures in the colder section remain safely above the dew point.
A key advantage of this dual-shell approach is the ability to tailor tube sizing to temperature-dependent fouling behavior. The warmer carbon steel shell utilizes smaller 1.5-inch tubes, supporting efficient heat transfer where fouling risk is minimal. In contrast, the colder stainless steel shell employs larger 3-inch tubes. The increased tube diameter reduces gas velocity and particulate impingement, significantly mitigating fouling and sulfate buildup in the most vulnerable section of the exchanger. This strategy directly addresses the root cause of cold-end fouling rather than relying solely on material upgrades or cleaning intervals.
In addition to fouling mitigation, the design allows the shell diameter and flow area to be adjusted to maintain combined

A variable-pitch tube sheet designed for radial shell-side flow, promoting uniform temperature distribution, reduced thermal stress at tube-to-tube sheet joints, and improved resistance to cold-end fouling.
shell-side pressure drop within acceptable limits. This flexibility enables performance targets to be met without introducing excessive fan or blower penalties.
Across the exchanger, the variablepitch radial-flow design accommodates differential thermal expansion between components. Uniform tube wall temperatures result in consistent tube expansion along the full tube length, minimizing stress at tube-to-tube sheet connections. When combined with a pre-stressed vessel expansion joint, operating stresses are further reduced, improving fatigue resistance and long-term mechanical reliability.
Development of this configuration requires detailed computational fluid dynamics (CFD) modeling guided by operating experience. CFD is used to evaluate temperature profiles, velocity distribution, pressure drop, and vibration potential, while engineering judgment informed by field experience ensures results are interpreted correctly and translated into practical design decisions. Together, these tools provide confidence that the configuration can deliver stable thermal performance, controlled pressure losses, and improved resistance to fouling and corrosion over extended service life.
For more information, please visit www.cgthermal.com. q
Across generations of people who helped build and rebuild American industry, one lesson tends to surface early: if you want to solve real problems, you’d better learn to ask real questions.
In industrial operations, the most expensive mistakes often happen when people stop asking “why?” too soon. That mindset becomes especially important when plants call with equipment problems that suddenly become urgent.
A maintenance planner recently called to ask for pricing and delivery for a replacement expansion joint. Actually, maybe two.
That word “maybe” is where the story begins.
The joints in question were 60-inch superheater expansion joints serving a high-temperature gas stream in a sulfuric acid plant. The plant suspected a leak in one of them and was worried the unit might have to come offline.
Superheater expansion joints operate in one of the harshest environments in the sulfuric acid process, where gas streams can exceed 800–1000°F and thermal cycling is constant. When components in these areas fail, plant teams often have to make decisions quickly.
Before talking about price or lead time, I started asking questions: How do you know one joint failed? Can the plant
keep running? Is there an environmental release? What does a day of downtime cost the plant?
That last question is where the conversation slowed down. At that point in the discussion, the economic impact of the situation hadn’t yet reached the people on the call.
Producers need to consider: what is the plant losing every day the unit is offline?
When equipment problems appear unexpectedly, most procurement conversations start the same way: What is your price? What is your lead time?
Those questions are understandable and are what buyers are trained to ask, but
By: Daniel A. Corral, Principal Engineer Operations & Systems, INTEREP
they rarely lead to the right decision.
Producers need to consider: what is the plant losing every day the unit is offline?
A plant losing $50,000 per day makes one set of decisions. A plant losing $2 million per day makes another. When the economic picture isn’t clear yet, the conversation usually defaults to the familiar procurement questions: price and lead time.
When expansion joints fail in hightemperature areas of sulfuric acid plants, there are usually several potential responses depending on plant conditions and urgency.
See Table 1.
In this case, the plant issued a purchase order for two replacement joints on an expedited schedule. Based on the information available at the time, that seemed like a reasonable middle ground.



We staged materials on Friday evening and planned to begin fabrication on Monday.
On Saturday morning, the calls began coming from senior leadership. The plant had calculated the cost of the outage cost. It was measured in millions of dollars per day. That changed the entire approach.
We called in the fabrication crew over the weekend, reassigned experienced welders from other projects, and began rolling and welding components immediately. By Sunday night, the joints were ready for dye penetrant testing. Early Monday morning, they were loaded and shipped to the plant.
When the economics become clear, decisions become clear.
From the outside, this story might sound like a manufacturing success story. It isn’t.
The real issue was that the most important information wasn’t available during the first conversation. The early discussion did not yet include the full picture of downtime economics, environmental exposure, or operational constraints. Once that information reached the right level within the organization, the path forward became obvious.
This is where experienced engineering and manufacturing partners can make a difference, helping plant teams think through the operational, economic, and technical implications before a crisis forces a rushed decision.
Expansion joints eventually fail. In high-temperature sulfuric acid service, that is simply part of operating industrial equipment under extreme conditions. How well a plant responds depends on understanding the economics of the situation. Plants that are aware of their downtime costs, environmental exposure, and operational constraints make better decisions when problems arise. When those plants contact vendors, the conversation is markedly better.
Instead of focusing solely on price and lead time, the discussion moves to the question that truly matters: What is the fastest and safest way to solve the problem?
When a unit is losing millions of dollars a day, the costliest question in the plant is often the one not asked soon enough.
For more information on INTEREP products and services, visit interep.us. q
By: Mathijs Sijpkes, Project Engineer, Elessent Clean Technologies
Selecting the appropriate filtration technology is critical to meeting today’s safety, performance, and operational reliability requirements. While traditional pressure leaf filters have supported the sulfur industry for decades, many plants are now reassessing their filtration systems in response to increasing performance expectations, more stringent HSE standards, and a growing emphasis on automation and digitalization.
This article provides an overview of sulfur filtration technologies and presents a comparison between conventional pressure leaf filters and modern self-cleaning candle filters. Ultimately, the optimal choice depends on the specific process requirements and operational objectives of each facility.
Pressure leaf filters have been around for nearly a century and have understandably become the standard choice across a wide range of industrial processes. Their versatility, ease of adapting or replacing filtration elements, and flexibility in precoat and element selection make them a dependable solution for the consistent removal of suspended solids.
At the time of their introduction, pressure leaf filters dramatically increased the volume of liquid that could be filtered, compared with the gravity filtration methods in use at the time. They also enhanced work environment safety by closing the system, hence reducing operators’ exposure to hazardous chemicals.
The process begins by feeding the process liquid into a vessel where multiple filter leaves are installed. Once the liquid reaches the top of the vessel, the outlet is opened and the system pressure pushes the liquid through the fine mesh or cloth on each leaf, which allows the filtered liquid through and into an outlet manifold, while the particles are retained on the filter leaf.
As these particles accumulate, they form a filter ‘cake’ which in itself acts as a filtration layer, sometimes in tandem with a filter aid. For application in sulfur, it is always required to apply such a layer of filter aid. This will continue until the maximum pressure drop or maximum cake thickness is reached.
Once filtration stops, the remaining liquid is drained, and the cake removal process begins. For the sulfur application, this can be done only by drying the cake layer and knocking it off the leaves manually.
This system offers the significant advantage of handling relatively high solids concentrations (0.01-3%), which makes it suitable for heavy-duty industrial process streams. The robustness of the vessel and leaf structures enables long-term service and strong wear resistance. The fact that the leaves can be customized in both layer configuration and number makes this filter a good choice for a wide range of industries.

Its global historical presence also translates into operational familiarity that plants can rely on, often reducing training requirements and providing a large base of industry know-how.
Finally, the standard pressure leaf filter is a competitive option on the market. While custom adaptations and technological enhancements may increase the initial investment, pressure leaf filters can significantly improve operational safety, efficiency, and long-term performance.
At Sulphurnet, for example, customizations include an improved design on the quick closing door mechanism, inversion of the traditional tank opening configuration, and a tailored drainage and filter leaf design, to name a few.
Of course, there is no single filtration system that completely fits all industrial needs. Like any system, the traditional pressure leaf filter has some disadvantages. To start, the cake-removal process requires a complete halt of
operations, creating downtime. Also, even with mechanical systems to help loosen the cake from the leaves, cleaning still requires operator involvement, which sometimes involves handling hot liquids or hazardous materials. This step in the process is one of the primary areas that modern self-cleaning systems were designed to eliminate.
Depending on the industry and operation, pressure leaf filters can also require a large plot for installation, operation, and cleaning.
Lastly, the hydraulics and electronics maintenance intensity of each filter can quickly increase costs and labor investment. Periodically inspecting and replacing the leaves and mechanical or sluicing systems, to prevent leaks or mechanical failures, helps ensure durability but still entails ongoing costs.
The limitations of traditional pressure leaf filters, along with advancements in technologies, led to the development of the self-cleaning candle filter. This has brought significant safety, regulatory, and efficiency improvements, particularly in the sulfur industry, where high temperatures, hazardous fumes, and labor-intensive cleaning make manual interventions risky.
The first of these filters was patented by E.I. DuPont in 1942, reflecting the industry’s early interest in automated cake discharge and more reliable continuous filtration. Over time, this drive for improved operability, combined with increasingly stringent HSE standards, contributed to the further development and refinement of this technology.
These filters are also pressure filters but use vertical candle elements to achieve fine solid-liquid separation. Technological advances enable candles to be produced from polypropylene, PVC, or stainless steel, allowing this type of filter to be used in a variety of applications.
The filtration process essentially works the same way. The vessel is filled with liquid, which flows from the outside to the inside of the candle and into an internal manifold. The solids are trapped

on the outer surface of the filter media, as in a traditional pressure leaf filter, forming a filter cake that, in turn, becomes a filtration layer.
The filtration cycles are also determined by the differential pressure or cake thickness; once these limits are reached, the vessel is drained, and the cake’s discharge process begins. This is when the back flush—or reverse pressure—is applied. Pressured steam is applied from the inside of the candle to the outside, causing the filter media to expand and the cake to crack and detach from the candle. The cake is discharged into the bottom and then through a valve.
Even with the technical improvements, expanded material options, and the integration of stricter design codes, these filters are not the standard for the sulfur industry. Sulphurnet is the first and only player in the market to offer a fully automated configuration, having created a design that not only competes with traditional filters but also improves certain aspects of the operation.
Sulphurnet’s self-cleaning candle filters can operate in manual, semiautomatic, or fully automatic modes and can be controlled by PLC control systems. This enables automatic filling, filtration, and back-flush cleaning cycles with virtually no manual intervention, resulting in continuous operation and reduced operator exposure.
Lastly, installing a closed system reduces exposure to dangerous vapors, high-temperature cakes, and iron sulfiderelated fire risks; thereby facilitating compliance with modern HSE and labor condition requirements.
With the growing shift of plant operations from manual on-site to automated and digital, having a reliable closed filtration system is imperative. Also, it is important to note that each application, process, and organization has different chemical requirements and operational challenges, which makes it even more important to invest in custom systems that will outlast the familiarity and expected durability of traditional one-size-fits-all solutions.
At first glance, installing a traditional pressure leaf filter without an automation system and a standard opening configuration (moving the vessel head and its attached leaves) may result in lower CAPEX than installing the equivalent amount of self-cleaning candle filters to manage the same flow rate.
However, when considering the technical and digital implementations that can be added to the traditional pressure leaf
filter, on top of a custom design tailored to the application’s characteristics, plus the operational costs of manual cleaning and maintenance, the investment difference is not as large as initially estimated.
Another big budget consideration is the cost of the downtime between cycles for the pressure leaf filter. In comparison, a configuration of several self-cleaning candle filters operating in parallel, along with their back-flush cleaning system, will minimize production interruptions and reduce downtime-related losses.
This comparative discussion demonstrates that both filters have advantages and disadvantages, and that an in-depth analysis of the needs and challenges of each plant is required to determine which filter system is most appropriate.
ties, plant age, and the specifics of each process will determine whether the familiar and robust pressure leaf filter remains a viable filtration option.
Our recommendation when implementing a closed self-cleaning candle filter system is to analyze and custom-design a configuration of several filters working in parallel, which allows a higher flow if needed for the application.
If pressure leaf filters are determined to be the best fit for a plant, it is recommended to incorporate the latest technological advancements—such as improved opening configurations, quick-closing door designs, and optimized draining schemes—to ensure the highest possible level of operational safety.
At the end of the day, both systems have reliable results, and it will depend on individual conditions to determine which is the better fit.
Comparative
While self-cleaning candle filters are the more advanced solution, market reali-
We specialize, in reliable, end-to-end heavy haul transportation for industrial, construction, and specialty equipment across the Gulf Coast and beyond.
For more information, please contact Mathijs Sijpkes, Project Engineer, Elessent Clean Technologies at Mathijs.Sijpkes@ elessentct.com. q

swrt-us.com info@swrt-us.com
By: Gary Siegel, Marketing Director, Beltran Technologies, Inc.
Nemaska Lithium’s Whabauchi mine, located in the Eeyou Istchee James Bay territory in Nord-du-Quebec, is one of the largest high-purity lithium deposits in North America and Europe. The mine has contracted with Beltran Technologies Inc., an international pollution control manufacturer, for two wet electrostatic precipitators (WESPs) to capture and control emissions before the gas stream is released through the stack to the atmosphere. The WESPs will be placed after a scrubber system reducing submicron particulate emissions to more than 99.8% clean.
The Nemaska operation consists of a spodumene mine and a concentrator at the Whabauchi complex in Northern Quebec. Also, Namaska has a processing plant in Becancour to produce lithium hydroxide for batteries, with lithium production supplied by the Whabauchi mine.
Lithium extraction from spodumene requires a lot of sulfuric acid. The extraction process entails initial crushing, then roasting at 2120 F, more grinding, and sulfuric acid leaching at 4820 F. The S2SO4 is typically sourced from sulfuric acid plants that are downstream plant operations derived from metallurgical non-ferrous smelters processing copper, zinc, lead, nickel, molybdenum, zirconium, and gold ores.
WESPs are used in metallurgical acid plants to protect the catalyst beds. WESPs are also used in spent acid recovery sulfuric acid plants where reprocessed or “spent” acid is converted into SO2 feedstock for the formation of new sulfuric acid. Another application for WESPs is to protect the sulfuric acid plants that are used to reduce SO2 and SO3 emissions from heavy oil and boilers where the fuel has high concentrations of sulfur.

WESPs efficiently collect submicron dusts and acid mists. These fine particulates usually contain heavy metals, such as arsenic, lead, zinc, cadmium, and other metals, depending on the content of the ores. Emissions from these metallurgical processes can contain flotation oils used to separate the various constitutes in the ore, such as sulfides. These oils evaporate in the high temperature of the metallurgical process and, in the quenching section of the gas cleaning plant, are condensed into mists, which are then collected by the WESP. WESPs are also used for tail gas cleaning where it is necessary to remove particulate, mists, and aerosols, and reduce emissions beyond safe levels before entering the stack to the atmosphere.
For the purest form of market-ready sulfuric acid, an efficient sulfuric acid manufacturing process requires the strict removal of contaminants from the input gas streams, especially fine and submicron particulates and acid mists, such as those emitted from metal ore roasters and smelters, petroleum refineries, and coal-fired industrial boilers. This is necessary for protecting downstream components such as catalyst beds from corrosion, fouling, and plugging, as well as for preventing the formation of a “black” or contaminated acid end-product. Proper gas cleaning also results in lower maintenance and operating costs for the manufacturing plants.
The advantages of using a WESP versus other types of gas cleaning equipment is that WESPs have high efficiency collection of submicron particulate, mists, and aerosols and low-pressure loss since the internal structure is open tubes, which are not easily plugged to restrict gas flow. Another advantage is that WESPs can remove dusts that are conductive or have high resistivity, which are problematic for dry ESPs. Since a lot of metallurgical dusts have high resistivity, the wet environment of the WESP coats the particulate with moisture, which makes the dust conductive and its collection highly efficient.
WESPs operate by charging and collecting particulate, mists, and aerosols with a corona formed by the collector surfaces and the sharp pointed discharge electrodes. High voltage power charges the WESPs at usually between 30 and 75 kilovolts, depending on the WESP design and the process gas conditions. The WESP is usually formed with a collector of tubes or plates with discharge electrodes held in the center of the collector structure by a high voltage frame, supported by non-conducting insulators. Since the process gases are saturated and contain electrically conductive mists and aerosols, the insulators are operated dry, being purged by dry, clean, and heated purge gases, usually ambient air. The WESP
can be operated by the collection of liquid acid droplets, mists, and aerosols, flushing the collector plates, or with the operation of continuous fogging sprays into the collector section. WESPs usually have deluge or wash nozzles mounted to periodically wash the WESP of solids and collected particulate, which might not be removed by the draining acid/water collected by the WESP.
Beltran WESPs are engineered with the use of advanced materials of construction that utilize collector tubes, such as hexagonal and square tubes. The most efficient design when considering collection efficiency, compactness, and economics is the square tube collector configuration. The square tube collector completely utilizes the cross-section of a square or rectangular vessel and can be effectively fitted in a round or hexagonal vessel. Due to the square tube’s high utilization of the vessel’s cross-section, it can be operated at a lower velocity so that the required tube length is lower, making it more efficient and easier to wash, since the wash sprays penetrate the collector. The high voltage frame is also more rigid, does not swing, and stays more accurately aligned, resulting in more efficient and reliable performance. Because of the shorter tube length, lower stabilizing insulators are not required, and the insulators can be mounted on the clean gas side of the WESP, reducing the requirement for heated purge air and resulting in more reliable WESP operation.
Since WESP collection efficiency is increased with increasing corona power, Beltran engineers designed multi-point star discharge electrodes, which maximize corona power and WESP operating efficiency. Multi-point star discharge electrodes overcome the problems of current suppression of space charge effect, whereby the corona power is significantly reduced by the high concentration of submicron particles, mists, and aerosol present in the process gases. This reduces the corona power of the WESP operation and can lower collection efficiency. The multi-point stars charge and repel some of the submicron particles and then enable the next star to increase its corona power, repeating this phenomenon almost 100 times as the gases flow up the tube. This type of electrode can produce considerable efficiency in the single or multiple pass WESPs commonly used in acid plants.
WESPs are being built of metal al-
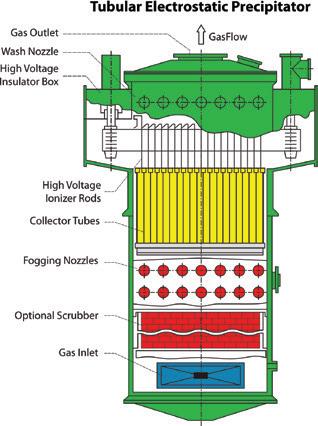
loys, thermoplastic materials, thermosetting materials, and conductive graphite composite materials. Metal alloys are expensive and have extended delivery time, but their biggest disadvantage is the unreliable performance with regard to corrosion. The sulfuric acid WESP operates in highly corrosive environments, including sulfuric, hydrochloric, and hydrofluoric acid and other impurities, as well as increased temperature. Because of the high cost of more robust chrome-nickel-molybdenum alloys, such as C-276, C-22 and C-2000, designers are attempting to employ less corrosion resistant alloys, like AL6XN and SMO 254, resulting in corrosion problems in some applications and conditions.
WESPs can be set up in various configurations: single WESP; two WESPs in series; two WESPs in parallel; and multiWESPs in parallel and two in series. Smaller gas flows are usually treated with one WESP. This also depends upon the efficiency requirements. However, one WESP unit can produce reliable service at 99.5% efficiency for smaller flows. Typically, plants have two WESPs in series so that either one WESP can be washed while one operates, or the two can operate simultaneously.
The Beltran WESPs delivered to Nemaska Lithium are manufactured with FRP housing and conductive graphite composite materials for the collector. The advantages are:
• Highly corrosion resistant
• Have good mechanical properties
• Electrically conductive
• Homogenous
• Do not require water/acid film to ground WESP
• Fire retardant and thermally robust
• Cost effective For more information, please contact Beltran Technologies, Inc. at (718) 3383311 or info@beltrantechnologies.com; or visit the company website at www.beltrantechnologies.com. q

SOUTHWEST REFRACTORY DELIVERS THE HIGHEST QUALITY ACID PROOFING, REFRACTORY, INDUSTRIAL MASONRY AND FIREPROOFING WORK.
With in-depth engineering support, meticulous craftsmanship and extensive planning down to the finest details, we’re turning industry into artistry.
AND
By: Nathan
Sulfur burners are a critical component in the production of sulfuric acid. The technology within the sulfur burning furnace—where molten sulfur combusts with oxygen to create sulfur dioxide (SO2)— comes with a variety of design options and inherent trade-offs in performance, maintainability, and capital costs.
Sulfur burner technology has evolved significantly, driven by the need for improved efficiency, reduced emissions, and enhanced operational reliability, particularly as capacities and equipment sizes have increased. This article explores the historical progression of sulfur furnace designs, recent technological advancements, inherent limitations, and a crucial cost-benefit analysis of modern innovations.
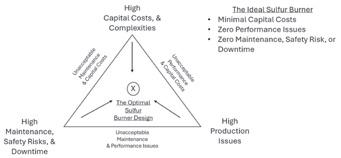
All engineering inherently involves trade-offs. A common, practical phrase in engineering is “keep it between the ditches,” meaning find a solution that satisfies all the necessary, often conflicting, constraints without veering into failure. For a sulfur burner, the “ditches” define the boundaries of an unacceptable design, as illustrated in Fig. 1. An optimal sulfur burner design (marked ‘X’ in the figure) must steer clear of the triangle’s three failure points: High Capital Costs & Complexities, High Maintenance, Safety Risks, & Downtime, and High Production Issues (i.e., poor performance). An optimal sulfur burner aims to mitigate anything that creates costs. This includes mitigating capital costs, performance issues, maintenance costs, safety risks, or production downtime.
The analysis presented here explores the following questions:
• What are the performance & design trade-offs in some recent modern sulfur burners?
• What commercial and technical improvements drove the sulfuric acid industry to accept incremental technology changes over time?
• What are the inherent weaknesses in most modern sulfur burner designs, and what still needs to be improved?
The constraints of an optimal design can be categorized into three critical areas that form the vertices of the design tradeoff triangle.
Achieving peak performance is nonnegotiable, as incomplete combustion will immediately plug the downstream equipment causing emissions, inhibiting conversion, and prolonging shutdowns.
Complete combustion of all sulfur
The primary goal is the quantitative and efficient conversion of molten sulfur to sulfur dioxide, ideally producing minimal sulfur trioxide in the furnace itself. Incomplete combustion leads to unburned sulfur vapor, which forms solid sulfur disposition on all cool surfaces (typically blinding off the mist eliminators as the first point of failure), plugging and clogging catalyst pores, and reducing the overall plant production rate until the operators are forced to take an unplanned shutdown and remove the sulfur from the equipment. Extreme examples involve using jack-hammers to remove sulfur-laden catalyst from the converter passes.
The sulfur burner achieves complete combustion of all sulfur molecules by optimizing three primary design parameters: atomization, residence time, and heat transfer. The sulfur combustion process proceeds as follows:
1. The spray nozzles atomize liquid sulfur into droplets of approximately 10 microns in diameter.
2. The droplets rapidly vaporize and transition into the gas phase.
3. The first chamber in the sulfur burner mixes the sulfur vapor with a controlled quantity of dry air, which initiates ignition.
4. Adequate process gas residence time in the sulfur furnace at elevated temperatures ensures that 100% of the sulfur oxidizes into sulfur dioxide before the combustion products leave the furnace.
The need to achieve a complete sulfur combustion drove the development of modern sulfur injection and sulfur burning technologies over many decades.
Manage thermal-cycling
Sulfuric acid plants often experience planned or unplanned shutdowns, leading to significant thermal-cycling of the sul-
fur furnace. Refractory linings—the inner protective layer of the furnace—are highly susceptible to damage (cracking, spalling) from rapid or repeated heating and cooling cycles. A good design must manage heat distribution and material selection to minimize these thermal stresses, thus preserving the refractory and extending equipment life.
There is significant interaction between refractory lining and steel shell and thus the thermomechancial behavior of the system must be considered. The temperature profile and acid dew point remains the strongest contributor of corrosion in a sulfur furnace.
nitrogen oxide (NOx) generation
While the furnace’s main purpose is to produce sulfur dioxide, the high-temperature environment is conducive to the formation of nitrogen oxides (NOx), primarily from the combustion air. Nitrogen oxides are a significant air pollutant and a target of environmental regulation.
One counterintuitive design aspect of sulfur burners that mitigates NOx formation is that the feedstock flows entering the sulfur furnace are significantly cooler than the sulfur combustion and flame temperatures. Sulfur auto-ignites in air at temperatures above 400°F (204°C); however, starting a sulfur burner at temperatures below approximately 800°F (427°C) is not advisable, as radiative heat transfer from the brickwork into the sulfur droplets is critical for efficient sulfur combustion. Liquid sulfur flows optimally at around 275°F (135°C)—any higher, and it begins to polymerize, increasing viscosity. Combustion air typically enters at 180–230°F (82–110°C, approximately). The relatively low combustion air temperature requires additional furnace residence time, as the cooling effect prolongs the heat transfer needed for sulfur vaporization before ignition can occur. The majority of NOx forms at high flame temperatures, often referred to as “kinetic NOx” (as opposed to thermal NOx), which typically range from 2,100–2,800°F (1,150–1,540°C, approximately). Operational experience shows that increasing the plant’s production rate raises the main compressor discharge temperature, which in turn elevates the peak flame temperature, significantly increasing NOx formation.
The maintenance and operational profile of the burner determine its long-term total cost of ownership and reliability.
Refractory maintenance challenges
The refractory lining is the most vulnerable component of a sulfur furnace. In addition to thermal-cycling, chemical attack from molten sulfur can occur, requiring careful material selection (e.g. highalumina bricks or specialized castables). Repair or replacement of the refractory is a costly, time-consuming, and major cause of downtime. Designs that simplify refractory geometry or use more durable materials, even if at a higher initial capital cost, often prove less expensive in the long run.
life expectancy
The longevity of mechanical parts, such as the atomizing device (nozzle or spinning cup), fans, and air registers, directly impacts maintenance costs. Choosing robust, high-temperature alloys and designs with easy access for inspection and replacement is vital to prevent unscheduled shutdowns.
Hazards of working on the equipment
Molten sulfur is handled at a temperature of ~275°F, posing burn risks. Equipment design must prioritize worker safety through features like lockable atomizing systems, controlled depressurization procedures, and robust sealing to prevent fugitive emissions. Maintenance tasks are inherently hazardous, demanding simplified designs to minimize exposure time.
Initial capital expenditure must be judiciously weighed against projected operational savings and performance gains.
Is the sulfur burner sized appropriately?
The “Goldilocks’ style” principle applies here: the burner must be sized just right. An undersized burner compromises production capacity and efficiency, while an oversized one incurs unnecessary costs for larger vessels, more refractory material, a larger footprint, as well as higher thermal losses and reduced steam production. Accurate modeling of heat release and residence time is necessary to avoid both extremes.
Are certain aspects of a sulfur burner design even necessary?
Engineers should carefully evaluate options to incorporate “state-of-the-art” features, as complexity often correlates with higher initial costs and increased maintenance requirements. For example, a spinning cup atomizer, while effective for sulfur atomization, introduces additional rotating equipment that requires ex-
tra maintenance and vigilance, as it must be removed from the furnace during a hot shutdown to prevent damage. In contrast, steam-jacketed spray nozzles, despite offering lower sulfur atomization performance and requiring a furnace approximately 33% larger, can remain inside the furnace during a hot shutdown. The continuous flow of steam through the nozzles prevents overheating or melting, simplifying operation and maintenance.
Cost/benefit analysis of additional features
Every incremental design improvement—from a specialized alloy to an added baffle—requires a rigorous economic justification. The benefit (e.g. an increase in conversion efficiency or a reduction in annual refractory costs) must clearly outweigh the additional cost and complexity. This constant push-pull between innovation and economy has shaped the industry’s acceptance of new technologies.
The journey toward the optimal design is clearly seen in the evolution of how molten sulfur is introduced into the furnace.
Sulfur spray nozzles
The development of sulfur spray nozzles revolutionized combustion precision compared to earlier, simpler injection methods. By forcing molten sulfur through a restrictive orifice, these nozzles atomize the liquid into fine, high-velocity droplets. The primary benefit of small drops is higher surface area to volume ratio of the suspended drops which increases their evaporation from convective and radiative heat transfer. This action ensures a more intimate and quicker mixing with the combustion air, leading to more complete combustion and higher sulfur dioxide yields, reducing waste and improving process control. However, nozzles are prone to plugging from impurities in the molten sulfur (especially if the sulfur pit isn’t well filtered) and suffer from wear due to the abrasive nature of the flow, requiring periodic replacement. Pooling of sulfur leads to refractory spalling from quenching and acid dew point corrosion of the shell from the local cold spot.
Spinning cup atomizers
Further refinements came with the spinning cup atomizer. This technology, which uses a high-speed rotating cup (often driven by a motor or air turbine) to centrifugally fling the molten sulfur into a finely dispersed mist, provides even finer and more uniform droplet dispersion than simple pressure nozzles.
The spinning cup technology enhances combustion uniformity and efficiency, particularly in high-capacity furnaces. Its
Atomization Quality Good
Maintenance/Wear
Prone to plugging; nozzle wear is common
Capacity Range Limited turn-down ratio
Capital Cost Lower
Excellent; very fine, uniform mist
Less prone to plugging; cup and drive system require precision maintenance
Wide operating range/turndown ratio
Higher and more complex; often requires technology licensing fee
Furnace Size Standard 1.7 – 2.5 seconds of residence ~33% smaller
Operational Challenges Fewer
Minimum Furnace Start-Up Temperature ~ 800°F
ability to handle a wider turndown ratio (i.e. operating efficiently across a broad range of production rates) also aligns with the industry’s need for flexible plant operation. The trade-off is higher initial capital cost and greater mechanical complexity associated with the high-speed drive system.
The structure of the furnace itself has also evolved to manage heat transfer and ensure uniform combustion. This provides additional mechanical stability since the baffles are tongue and groove and permit axisymmetric designs (circular and ring shaped baffles) which are more efficient by reducing furnace “dead” volume.
The adoption of hexagonal baffles (or similar internal partitioning systems) improved furnace durability and heat distribution. These hexagonal (or other complex shape) baffles or thermal walls serve several critical functions:
1. Enhance turbulence and mixing: By disrupting the straight flow of the gas, they promote greater turbulence, ensuring the sulfur droplets and air are thoroughly mixed, allowing for more complete and rapid combustion. This directly addresses performance. The first baffle is important in establishing a recirculation cell (like an orifice or sudden expansion into a large space) to move heat back to the front of the furnace for drop evaporation. Ideally, all droplets should be evaporated before the first baffle.
2. Heat transfer: The large surface area of the baffles acts as a radiant heat sink and source, helping to stabilize the flame temperature and provide a uniform heat profile. Typically, the front of the furnace operates at 100–200°F / 38–76°C hotter than a furnace with traditional baffles or no baffles.
3. Refractory protection: They shield the outermost refractory from the direct, highvelocity flame, reducing thermal stress and
Refractory failure and thermal stress
Inherent weakness: The fundamental problem remains the refractory-molten sulfur/high-heat interaction. No refractory material is immune to the combination of extreme temperature and chemical exposure over decades. Thermal cycling remains the single biggest cause of refractory failure, leading to costly and lengthy shutdowns.
Can require removal from furnace during a hot shutdown.
< 800°F due to increased atomization
enhancing the refractory’s life, thus addressing maintenance and downtime concerns. However, baffles add to the complexity and capital cost of the furnace, and they create more refractory surface area that must be inspected and maintained, representing a maintenance trade-off. Their placement must be modeled carefully to avoid creating zones of stagnation that could lead to NOx formation or incomplete combustion.
The sulfuric acid industry, by nature, is risk-averse. Plant sizes can be enormous, and unscheduled downtime is unacceptably expensive. Therefore, the industry has historically favored incremental changes over revolutionary leaps. The key drivers for accepting new technology include:
• Scale economies: As global sulfuric acid demand grew, burners needed to handle significantly higher capacities without massive increases in footprint or refractory thickness. Larger furnaces sometimes justify few baffles as the larger volume to surface area in the furnace reduces walleffects and wall-channeling effects.
• Operational cost reduction: Improvements that reduce the need for manual cleaning, decrease refractory spalling, or extend maintenance cycles are often adopted because labor and downtime are the largest operating expenses. Hexagonal baffles and more durable refractory castables fit this profile.
In essence, the industry accepted changes that offered a high return on investment (ROI)—a substantial reduction in OpEx or a regulatory necessity—with a low risk of operational failure.
Despite significant advancements, most modern sulfur burner designs still face inherent weaknesses that represent the remaining “ditches” to avoid.
Need for improvement: Research into novel, non-ceramic refractory materials (e.g. highly stable composite materials) or advanced cooling jacket designs that better manage the heat flux across the refractory wall could revolutionize furnace longevity.
High temperature vs. maintenance trade-offs
Inherent weakness: The high temperatures required for perfect sulfur combustion are exactly what drives undesired NOx formation. Any attempt to significantly lower the flame temperature risks incomplete sulfur combustion.
Mechanical complexity of atomization
Inherent weakness: While the spinning cup atomizer offers superior performance, it introduces a high-speed mechanical device into a high-temperature, corrosive environment. This complexity inherently increases the risk of mechanical failure, leading to unscheduled downtime and challenging, hazardous maintenance.
Need for improvement: The ideal solution is a method of ultra-fine atomization that is purely static (no moving parts) or relies on non-mechanical, low-wear energy sources (e.g. ultrasonic atomization or advanced fluidic oscillators), thus driving the design toward the “Zero Maintenance, Safety Risk, or Downtime” goal.
The pursuit of the perfect sulfur burner design is an ongoing effort to keep the technology “between the ditches” balancing minimal capital costs, zero performance issues, and zero maintenance risk. The industry’s evolution from simple nozzles to sophisticated spinning cups and complex furnace geometries with hexagonal baffles represents a continuous, incremental optimization driven by economic necessity and environmental stewardship.
Modern burners have successfully mitigated many risks, but the fundamental challenges of refractory life, the cost vs. efficiency trade-off, and the mechanical complexity of high-performance atomization persist. Future innovations will likely need to be un-intuitive and create substantial benefits without negatively impacting potential down-side risks, thus steering the industry closer to the elusive center point ‘X’ of the trade-off triangle and cementing the reliability of sulfuric acid production for decades to come.
For more information, please visit porterprocess.tech. q
By: Martin Alvarez, Solution Specialist Sulfuric Acid Catalyst, Topsoe
Some 30 years ago Topsoe launched the VK69, a novel catalyst specifically tailored for the lean gases present after the intermediate absorption tower in double absorption plants. Initially envisioned as a cost-effective solution to help producers cut stack emissions, it has proven very effective in helping to address several other, new and eternal, key concerns as well. For example, +9% in specific steam production, +7% higher acid production, double-digit SO2 emissions, or slashing emissions in half, are some of the results customers have achieved by installing VK69.
This catalyst is uniquely suited to help sulfuric acid plant operators address many of the new or tightened requirements so important today, such as improving energy efficiency, reducing waste, or achieving ultra-low SO2 emissions without reducing production rates.
In 1996, Topsoe introduced the cesium-promoted VK69 catalyst, which was specifically designed for lean SO2 gases typically found in the lower passes of the converter in double absorption plants. Today, with more than 130 installations successfully running, VK69 has proven to be an unmatched solution for reducing SO2 emissions and increasing production rates.
Upon the introduction of VK69, it was already established that the use of cesium as a promoter could enhance the catalyst performance in the low-temperature region by keeping soluble V-complexes in solution and thereby active in catalytic cycles. However, Topsoe understood that the performance of the catalyst in the lower passes of the converter was restricted not only by low temperatures but also by the lean SO2 gases, where diffusion was the limiting factor.
To address this limitation, Topsoe designed the VK69 catalyst with a new and unique 9 mm daisy shape, a revised support material, and an optimized melt composition. The result was a high-vanadium cesiumpromoted catalyst that combined a very low ignition temperature with a significant activity advantage in the entire operating temperature range.
Since the first commercial installation in 1996, VK69 has been widely adopted by the industry. Today, VK69 is installed in more than 130 sulfuric acid units worldwide and has successfully helped operators to
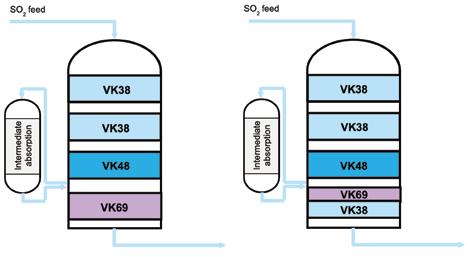
achieve the following benefits:
• Increased acid and steam production rates by using a higher SO2 strength, without increasing the plant’s pressure drop
• Improved energy efficiency of the unit and reduction in carbon footprint
• Reduction of up to 50% in SO2 emissions when used in the last bed of double absorption plants
• Reduction or avoidance of scrubber chemical consumption and disposal costs of byproducts
• Reduction of SO2 emissions during startups
• Flexibility to handle upsets in the plant saving fuel and reducing downtime
As shown in Fig. 1 depending on the application and the case, VK69 can be used in a full bed or as an ignition layer in the lower passes.
The following case studies illustrate some of the operating experience gained by Topsoe over the last 27 years. Actual operating data and SO2 measurements accompany all the cases shown.
The following industrial example shows the performance of a metallurgical unit in North America designed originally with bed 4 fully loaded with 51 m³ of VK69. Its features include:
• SO2 source: Metallurgical, Zn
• Configuration: 3+1 DCDA
• SO2 strength: 7.50-8.50%
• Design Capacity: 1,040 MTPD
Following the startup, the plant’s performance was excellent. SO2 measurements were conducted on multiple occasions, all of which showed emission levels below 43 ppm.
very high-grade SO2 gas without exceeding the maximum temperatures for the catalyst and equipment. The plant’s features are summarized as follows:
• SO2 source: Metallurgical, Cu
• Configuration: 3+2 DCDA with LUREC
• SO2 strength: 14.0-16.5%
• Design Capacity: 2,250 MTPD
Since the Chinese government had been constantly tightening SO2 emissions in many regions, this customer chose a long-term approach during the design phase and decided to install VK69 in the fifth pass of the converter with a LUREC configuration. In this way, they could process very strong gas and comply with the legislation, without depending on a scrubber in the years to come.
SO2 measurements have been conducted on numerous occasions in this unit, and all measurements have consistently shown that the emission level never exceeded 43 ppm, even with an SO2 gas strength above 15%.
The VK69 screening history in this unit is illustrated in the following timeline and shows that not only has the plant achieved an excellent conversion level, but it has done so over a prolonged time with very little replacement catalyst needed. Over the first 12 years, no work was done in the last bed, and after that, only 18% of the catalyst was replaced. Since that replacement, the unit has been running very well, with the usual very low emissions and no new catalyst work done in bed 5.
The case illustrates that if a plant is designed with the right conditions to take full advantage of VK69, it is possible to reach very low emissions. Additionally, it shows that the low emission level can be maintained for 4 years without the need to do any catalyst work in bed 4.
The next industrial example consists of a 2,250 MTPD Chinese plant based on copper smelting with a LUREC process. The LUREC technology consists of a 3+2 double absorption layout with a recirculation loop of a small gas stream that is branched off after the third pass. This recirculation stream mixes with the feed gas before entering the converter, enabling the processing of
This industrial case study also provides valuable insight into the potential cost savings that can be achieved by choosing a VK69 solution over a standard catalyst solution with a tail gas scrubber.
In a hypothetical case in which the customer had chosen a standard catalyst solution with VK48 instead of VK69 loaded in bed 5, the emission achieved with the standard VK48 solution would have been 113 ppm instead of 43 ppm. Assuming a typical scrubber installa-
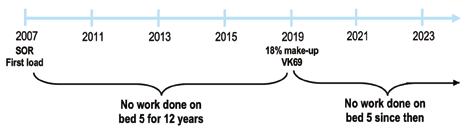
tion cost of around USD 400,000 and an annual operational cost of 44,500 USD (detailed in Table 3), the cost of operating an H 2O2 scrubber over 16 years would have been approximately USD 1,112,000.
Emissions outlet converter with VK48 case 113 ppm
Emissions outlet scrubber with VK69 case 43 ppm
Chemical consumption of H2O2 @ 27.5 wt% 325 ton/year
H2O2 @ 27.5 wt% price (China) 137 USD/ton
Total operating cost 44,500 USD/year
In contrast, choosing VK69 over VK48 for the fifth bed would have required a one-time extra investment of 594,000 USD. The 18% make-up done in 2019 would have added an extra cost of 106,000 USD totaling a sum of 700,000 USD of catalyst extra cost over 16 years.
By choosing VK69 over a standard solution with a scrubber, the customer was able to recover the investment in less than 5 years and save around USD 412,000 over 16 years.
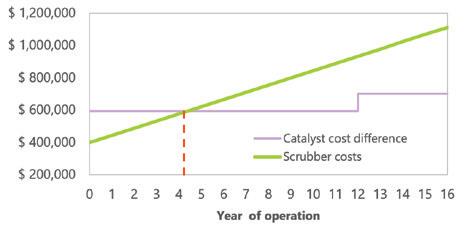
An S-burner located in Africa with a 3+1 DCDA layout was designed originally with VK38 catalyst in the last pass. With this design, the plant was able to burn 36.5 MT/h of sulfur and produce around 2,675 MTPD of sulfuric acid with an emission level of around 110 ppm. The export steam rate at this condition was estimated to be approximately 135 MT/hr.
Five years later, the customer considered increasing the production of the unit. A bottleneck study was conducted that identified several constraints that would need to be addressed to increase capacity. These constraints included:
• Insufficient catalyst in the converter for the new capacity
• Lack of extra room for additional catalyst
• Insufficient air supply from the blower
• Inlet temperature to bed 4 that was too hot to achieve an acceptable conversion at the new capacity due to equilibrium limitations
These constraints made it clear that the plant needed a catalyst solution that was not only able to work at lower temperatures but also offered considerably more activity to compensate for the lack of room in the converter and the stronger gas that would have to be processed when burning more sulfur without increasing the airflow. Topsoe observed that this was a perfect match for VK69 and recommended it replace the last bed originally loaded with VK38.
Once the VK69 was installed in bed 4, several SO2 measurement campaigns were carried out. The measurement confirmed that the unit could increase acid production capacity while maintaining low emission levels and producing more HP steam. Table 4 presents a comparison between two sets of SO2 measurements taken before and after the installation of VK69.
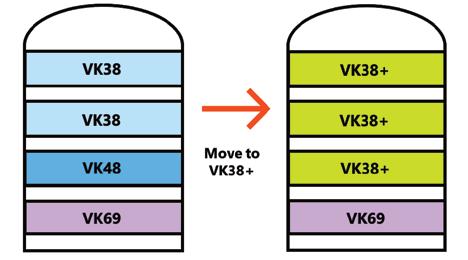
combined with the VK69 to achieve even lower emissions levels. Operators can also utilize the flexibility that the extra activity of the combination VK38+/VK69 delivers to target specific bottlenecks.
Fig. 6 demonstrates how the new VK38+/VK69 scheme can achieve improved emissions levels compared to the previous VK38/VK48/VK69 design. The example is based on an S-burner unit with feed gas of 11% SO2 and 1,000 MTPD capacity. If the unit is already using VK38/VK48 and VK69 catalysts and has a full converter with an emission level of 120 ppm, the unit can easily cut emissions by 20% and achieve levels below 100 ppm simply by changing the catalyst from VK38/VK48 to VK38+ and adjusting the inlet temperatures by ±10°C.
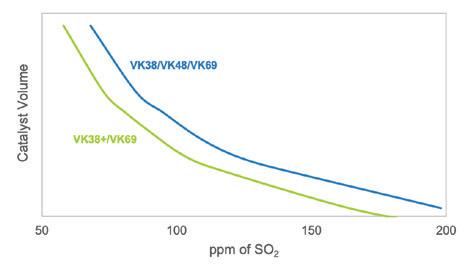
Fig. 4 shows the benefits not only in terms of acid production but also in terms of steam production (and reduction in carbon footprint) of upgrading catalyst with VK69.
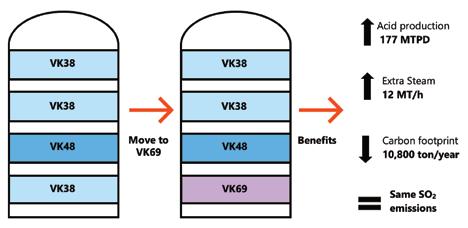
As operators face more demanding conditions, they may be wondering what the next step is to maintaining low emission levels without compromising productivity. In 2020, Topsoe launched the VK38+, a new non-cesium catalyst that offers higher activity than any other potassium-promoted catalyst in the market. This catalyst can be
The cases analyzed in this paper demonstrate that over the last few decades, VK69 has helped operators not only to cope with ever tightening SO2 emission legislation, but also boost their acid production, maximize energy efficiency, and experience cleaner and faster startups. In addition, implementing VK69 has proved to be very cost-effective, since upgrading catalyst loading does not require additional chemicals, generate waste, or require costly, extensive revamps.
Finally, for those plants that have more demanding requirements for emissions or production capacity, and have already installed VK69, a new combination of VK38+/VK69 is now available. This combination allows sulfuric operators to achieve even higher conversion levels or deal with specific bottlenecks that can be solved with extra activity. With the decarbonization and clean air trend of the present day, VK69 is indeed more relevant than ever.
For more information, please
Held from September 29 to October 2, 2025, in the city of Gramado, the 13th Brazilian Sulfuric Acid Congress–COBRAS 2025 once again established itself as an important and influential technical event for the sulfuric acid industry in Latin America.
Since 1993, the Brazilian Sulfuric Acid Congress (COBRAS) has been recognized as a leading forum for technical discussion and knowledge exchange in the sulfuric acid sector in the Southern Hemisphere. In its 13th edition, the event brought together more than 185 participants, with over 70% representing sulfuric acid producers, reinforcing its strong technical and strategic character.
COBRAS 2025 stood out for fostering a highly qualified environment for the exchange of experiences among producers, technology suppliers, industry specialists, and sector leaders.
The event provided a platform for high-level technical discussions, presentation of industrial case studies, sharing of operational best practices, and debates on innovation, efficiency, safety, and sustainability.
More than just an event, COBRAS has consolidated itself as a key integration platform for the sulfuric acid

The 13th edition of COBRAS 2025 brought together over 185 participants from around the world.
production chain, stimulating the advancement of national engineering and the continuous improvement of industrial plants across Brazil and Latin America.
This edition featured 25 sponsors, bringing together the main suppliers of technologies, equipment, and specialized services for the sulfuric acid market.
The strong presence of leading companies reinforces
From November 3-5, 2025, the Sulphur + Sulphuric Acid Conference brought together professionals in the sulfur and sulfuric acid industry from around the world for three days of technical learning, innovation showcases, and global networking at the Woodlands Waterway Marriott Hotel & Convention Center in The Woodlands, Texas.
The annual forum, organized by CRU Group, attracted more than 375 registered attendees from around the world, representing producers, operators, technology developers, equipment suppliers, and researchers focused on sulfur and sulfuric acid markets and technologies.
The event included a Sulfuric Acid Troubleshooting workshop. Presenters included Graeme Cousland, Begg Cousland Envirotec; Douglas Azwell, Elessent MECS Technologies; Helen Cardwell, Elessent MECS Technologies; Steve Puricelli, EXP; Bill Goodell, Topsoe; and Jack Harris, VIP International
A dual-track technical program for sulfur and sulfuric acid explored new technology and best practices. Sulfuric acid sessions included the following presentations:
• “Innovations in sulfuric acid production technology,” by Dr. Ben Egelske, Recalibrate
• “A 500kt/a sulfur-burning acid plant in Africa,” by Yijiao Zhou, Wylton (China) Chemical Co., Ltd.
• “Advanced strategies in the filtration process to extend the operation of the converter,” by Mathijs Sijpkes, Elessent MECS Technologies
• “Maximizing the ROI of your sulfuric acid plant,” by Benjamin Senyard, Worley Chemetics
• “Circular economy approach in the Gresik Petrochemical Complex: Utilization of silica by-product as a sustainable alternative to diatomaceous earth for sulfur filtration in sulfuric acid production,” by Faizal Alianto and Jawad Farisi, PT Petrokimia Gresik
• “Safe & reliable production of high-purity sulfur trioxide (SO3) for the semiconductor industry,” by Justin Thiems, Elessent MECS Technologies
• “Advanced molten sulfur spraying: Ensuring operational reliability through precise temperature and droplet management,” by Ashwin Patni, Lechler, Inc
• “Keeping a 40-year-old converter going,” by Stuart Hinze, Simplot
• “Advancements in SO2 scrubber/quench gas coolers, using ceramics to resist corrosion,” by Ethan Schrader, CG Thermal LLC
• “A tale of two towers,” by Steve Puricelli, EXP
• “Improving reactor performance at the Ilo Copper Smelter acid plant,” by Alvaro Jara, Southern Peru
• “Maintaining acid quality and plant efficiency in metallurgical offgas operations,” by Collin Bartlett, Metso Canada Inc.
• “Integrated surface protection and reliability strategies for sulfuric acid assets: from tank linings to predictive maintenance,” by Michael Labbe, REMA TIP TOP AG
• “Staged upgrade and capacity expansion of a sulfuric acid
the credibility of the conference and highlights its importance as a strategic venue for technical networking and the generation of new business opportunities.
Over four days of intensive programming, COBRAS 2025 reaffirmed its commitment to knowledge dissemination, technological innovation, and the development of a more efficient, competitive, and sustainable sulfuric acid industry.
The event once again demonstrated its ability to gather key professionals and industry players in a collaborative, technically driven, and results-oriented environment.
Continuing its trajectory of growth and relevance, the next edition of COBRAS will take place in Manaus, in the heart of the Brazilian Amazon, in 2027.
The industry now looks ahead to another chapter of integration, knowledge exchange, and strengthening of the sulfuric acid sector in Latin America.
For more information, please contact: marketing@ clarksolutions.com q

Graeme Cousland of Begg Cousland Envirotec shares his experience with mist carryover during the Sulfuric Acid Troubleshooting workshop. Other presenters included Steve Puricelli, EXP, Bill Goodell, Topsoe, Helen Cardwell, Elessent MECS Technologies, Douglas Azwell, Elessent MECS Technologies, and Jack Harris, VIP International.
plant,” by Nesho Plavsic, NORAM Engineering & Constructors
• “De-bottlenecking spent sulfuric acid regeneration unit for sustained economic and environmental benefits,” by Blake Stapper, Messer
• “Hydrogen generation and mitigation strategies in sulfuric acid plants,” by Payton Wanstreet, Pegasus TSI Inc
• “Platinum promoted honeycomb catalysts–a versatile instrument for acid making” by Johannes Hofer, P&P Industries AG
• “Fertilizer producer and Topsoe partner to boost acid production,” by Martin Alvarez, Topsoe
• “AI-troubleshooting in sulfuric acid production: A modern upgrade for a mature process,” by Jesse Mikulec, ControlRooms.ai
• “Update on sulfuric acid dew point monitoring–applications and case study,” by Cal Lockert, Ohio Lumex The next Sulphur + Sulphuric Acid Conference will be held November 3-5, 2026, in Berlin. For more information, please visit: www.crugroup.com. q








3–5,


Marouane El Hajjam, Brahim Nait Telhak, Amal Bellarbi, Youssef Daafi, and Hamza Allam of OCP Group attend CRU’s Sulphur + Sulphuric Acid Conference in The Woodlands, Texas.


and


Acid Conference.




Catching up at the Sulphur + Sulphuric Acid Conference, from left, are Jonas Gabriel Linder of SmartScope; Collin Bartlett of Metso Canada Inc.; Singh Ashish Kumar and Prasad Shashikran of Pacific Refractories Ltd.; and Martyn Dean of Begg Cousland Envirotec Ltd.




Elessent Clean Technologies hosted a dinner for their customers celebrating the 100th anniversary of the company’s sulfuric acid catalyst manufacturing during the Sulphur + Sulphuric Conference in The Woodlands, Texas. Seated from left, are Rick Mason of Ecoservices, Jacob Carpenter of Nevada Gold Mines, Stephan Hörning of Röhm GmbH, John Varnum of CMW, and Rachel Thapalia of Elessent/MECS® Technologies. Standing are, left to right, Ricky Jaswal of Weir, Brad Varnum of CMW, and Kelly Hopkins of CMW.


of




Stephan Hörning of Röhm GmbH, left, Verka Stoyanova of Aurubis Bulgaria, and Ivaylo Ivanov of Aurubis Bulgaria enjoy the hospitality at Elessent Clean Technologies’ catalyst centennial celebration.



At Elessent Clean Technologies’ catalyst centennial celebration are, from left, Vulcan Mutler of EXP, Mickey Jones of Lithium Americas, John Horne of Elessent/MECS® Technologies, Kevin Bryan of Lithium Americas, and Matt Karins of Lithium Americas standing in front of a Ford Model T made in 1915 —the year the catalyst company was founded.



Marcelo Kascheres of Elessent/MECS ® Technologies, left, Alex Knoll of Acid Piping Technology, and Robert Schlegel of Ecoservices attend Elessent Clean Technologies’ 100 years of sulfuric acid catalyst manufacturing dinner during the Sulphur + Sulphuric Acid Conference.

The Sulphur World Symposium 2026, organized by The Sulphur Institute, is an annual industry conference focused on the global sulfur and sulfuric acid value chain. It will be held April 28-30, 2026, in Vancouver, Canada, and will bring together producers, refiners, fertilizer companies, agronomists, logistics firms, and technology providers.
The symposium serves as a global forum for professionals involved in sulfur and sulfuric acid production, marketing, logistics, and end-use industries. The event will focus on:
• Market outlooks for sulfur and sulfuric acid
• Demand drivers such as fertilizers, mining, and energy
• Technical and operational innovations
• Agronomic uses of sulfur in crop nutrition
• Networking among decision-makers across the industry value chain
For more information, please visit the event’s website, www.sulphurinstitute.org.
For the past 48 years, members of the AIChE Central Florida Section and colleagues from around the world have gathered to share ideas on chemical process technology— particularly the production of phosphoric acid, phosphate fertilizers, sulfuric acid, and related topics in the chemical
process industry. The convention also offers an excellent opportunity for a relaxing getaway with friends and family, great food, and plenty of fun.
This year’s event will be held at the TradeWinds Resort on St. Pete Beach on Friday, June 5 and Saturday, June 6, 2026.
Friday’s workshops will include the returning Sulfuric Acid Workshop and the biannual PE Laws, Rules, and Ethics Workshop. Attendees can earn up to 8 PDH by participating in both Friday and Saturday sessions.
If you have any questions or would like more information about the convention, please contact Convention Chair Jordan Waters at vicechair@aiche-cf.org or Section Chair Michelle Navar at chair@aiche-cf.org, or visit the event website at www.aiche-cf.org/annual-conference.
Holtec’s 2026 Roundtable for Sulfuric Acid Plants will be held on September 6-10, 2026, in Punta Arenas, Chile. The conference will take place at the Dreams Hotel, conveniently located in the heart of Punta Arenas.
The Sulfuric Acid Plants Roundtable is a technical meeting that, for more than a decade, has brought together operators, maintenance professionals, engineers, and acid plant leaders from major operations around the world.
Over the five days of the event, there will be technical sessions featuring presentations, discussions, and roundtables, as well as networking and social activities. Each session will address current challenges and best practices in a variety of areas including operation, maintenance, environmental control, safety, and energy
efficiency in metallurgical and sulfur-burning sulfuric acid plants.
For more information, please visit the event’s website, www.mesaredondachile.com
CRU’s Sulphur + Sulphuric Acid conference continues to be an essential annual forum for the global sulfur and sulfuric acid community to learn, connect, and do business. Knowledge sharing is at the event’s core via the respected dual-streamed technical program, and the connections made with industry experts and solution providers attending.
The conference will take place November 3-5, 2026 in Berlin, Germany.
In November, you can expect an expanded market outlooks agenda, including expert insights from CRU’s analysis teams on major supply and demand markets, including sulfur and sulfuric acid, plus additional industry updates from key players from across the supply chain.
Industry-leading presentations will cover technical updates on the production and processing of sulfur and sulfuric acid and will feature new innovations in process, technology, materials and equipment developments, as well as practical case studies highlighting operational experience and improvement.
Running alongside the agenda will be an exhibition of world-class solution providers serving the sulfur and sulfuric acid industries.
For more information, please visit the event’s website, www.crugroup.com q










Designed to match your exact production needs, our modular sulfuric acid plants can be placed next to your main facility, reducing installation costs and ensuring reliable acid supply.























With over 130 years experience, Lewis® pumps and valves are engineered with genuine Lewmet alloys, meaning they provide better corrosive resistance than anyone else. Our team is focused on one thing, and one thing only, creating the most durable products in the world. It’s no surprise we’re market leaders, there’s just nothing as strong as Lewis® pumps and valves. Learn

