BENTHICFORAMINIFERALDYNAMICSINREEF-ASSOCIATEDSEDIMENTSOF JOBOSBAY,PUERTORICO:2018–2019
ANGELIQUE ROSA MARÍN1,2,*,PAMELA HALLOCK2 AND MICHAEL MARTÍNEZ-COLÓN1
ABSTRACT
JobosBay,southeasternPuertoRico,experiencesstrong environmentalgradientsbetweenanhistoricallyimpacted coastal-plainandoligotrophicCaribbeanwaters.Thecoastal zoneisdynamicbothseasonallyandinterannually.During 2018–2019,waterquality,sediments,andbenthic-foraminiferalassemblageswereassessedfromfore-andback-reefsites offthreecaysthatseparateJobosBayfromCaribbean waters.Temperatureandsalinityreflectedseasonalvariations,inorganicnitrogenindicatedterrestrialrunoff,andsedimenttexturereflectedinfluenceofwindsandwaveenergy. Foraminiferalassemblagesinthefore-reefweredominated by Amphistegina whiletaxasuchas Quinqueloculina and Discorbis dominatedback-reefsediments.Lowtestdensities reflectedtheinfluenceofwaveenergyinpredominantlysiliciclasticsediments.Interannualdifferencesinsedimenttexturesandassemblagescollectedduringcomparablemonths reflectedtimingofstormpassages.Theprevalenceofalgal symbiont-bearingtaxainfore-reefsitesindicatedsuitable waterqualityforreefaccretion.ThisstudyofJobosBay foraminiferalassemblagesprovidesbaselinedataonseasonal andinterannualvariability.
INTRODUCTION
BACKGROUND
TropicalcoralreefsareamongEarth’smostvitalmarine ecosystems.Suchreefsprovideawidearrayofecosystemservicesthatcontributetoenvironmentalandsocietalinterests (e.g.,Moberg&Folke,1999;Eddyetal.,2021).Despitetheir importance,reefsarethreatenedbyawiderangeofglobaland localenvironmentalstressors,bothnaturalandanthropogenic (e.g.,Beyeretal.,2018;Woodheadetal.,2019).Increasing concentrationsofcarbondioxideintheatmospherearedriving oceanacidificationandclimatechange,includingrisingseasurfacetemperatures(e.g.,Hoegh-Guldbergetal.,2007, 2018).Terrestrialrunoffincreasesaftertropicalstormsand hurricanes,resultinginnutrientpollutionandwater-quality changes(e.g.,increasedwaterturbidity)oncoralreefs(Takesueetal.,2021).Humaninfluencesfromagriculture,industrial,andcoastaldevelopmentpracticescontributetoincreases inturbidity,nutrients,andotherchemicalpollution(e.g., Wooldridge&Done,2009;Lesser,2021).Inthepast50years, coral-bleachingeventsassociatedwithwarmersea-surface temperatureshaveincreasedfromrareoccurrencesinthe 1970s,totwosignificanteventsintheCaribbeanin1983 (Jaap,1985)and1987(Langetal.,1992),tomultiplemass
1 FloridaAgriculturalandMechanicalUniversity,1601S.Martin LutherKingJr.Blvd.,Tallahassee,FL32307
2 CollegeofMarineScience,UniversityofSouthFlorida,8301st St.S., St.Petersburg,FL33701
*Correspondenceauthor.E-mail: angeliquerosamarin@gmail.com
bleachingeventssincetheworldwideeventin1997–1998 (e.g.,McClanahan,2022;Shlesinger&vanWoesick,2023).
InPuertoRico,coral-reefecosystemsdirectlyinfluencethe economiesofcoastalcommunities,primarilythroughtourism andrecreationalactivities.Atthesametime,coralreefsare threatenedbyhumanactivities(e.g.,Hernández-Delgado& Ortíz-Flores,2022)thatpromoteterrigenoussedimentinflux (Ramos-Scharrónetal.,2015;Ramos-Scharrón,2021),nutrientpollution(Larsen&Webb,2009),sewagepollution(Hernández-Delgadoetal.,2011),non-pointpollutionsources (Bonkoskyetal.,2009),anddirectandindirectimpactsfrom recreationalactivities(Webler&Jakubowski,2016).
Benthicforaminifershavebeenusedformorethan50years toprovideinsightintoenvironmentalchanges,pollution,and climatechange(e.g.,Alve,1995;Frontalini&Coccioni,2011; Barbosaetal.,2012).Theyareusefulasbioindicatorsbecause oftheirdiversityinestuarine,coastal,andmarinehabitats,combinedwiththeirproductionofagglutinatedorcalciumcarbonate(CaCO3)shells(commonlyknownastests).Thekindsof shellsfoundinsedimentscanprovideinformationaboutpast environmentalconditions(BarragánMontilla&SanchezQuiñonez,2021),typesandratesofchanges,andpresentconditions inahabitat(e.g.,Oliveretal.,2014;Mathesetal.,2022).A varietyofmetrics(e.g.,speciesrichnessandotherdiversity indices)arecommonlyusedtoevaluateforaminiferalassemblages(e.g.,Murray,1973,2006;Hayek&Buzas,2010;Gonzalesetal.,2022).Inaddition,severalindicesspecificallyusing benthicforaminiferstoevaluatethehealthofcoastalenvironmentshavebeendeveloped(e.g.,Hallocketal.,2003;Schönfeldetal.,2012;ElKatebetal.,2020;Prazeresetal.,2020).
Twostudies(Donnelly,1993;Oliveretal.,2014)offLa Parguera,PuertoRico,specificallycontributedtothedevelopmentandapplicationofindicesusingbenthicforaminifersin coralreefecosystems.Hallocketal.(2003)usedsamplesfrom LaParguera(Donnelly,1993)andtheFloridareeftract (Cockeyetal.,1996)todevelopawater-qualityindexknown as “ForaminiferainReefAssessmentandMonitoring”,abbreviatedastheFoRAMIndexorFI.TheFIreflectshowreef waterqualityinfluencestheoccurrencesofbenthicforaminifers.Oliveretal.(2014)subsequentlycomparedtheFIwith otherenvironmentalmetrics,includingcoralcover, fishcommunities,andmacrobenthicfauna,forLaParguerawaters, findingthatreefsexposedtomoresignificanthumandisturbanceshadlowerFIvalues(rangeof2–3)indicativeofdeterioration,whilethosefartheraway(withlesshumandisturbance) hadhigherFIvalues(4),indicatingbetterwaterqualityforcoral survivalandreefaccretion.
STUDY SITE:JOBOS BAY NATIONAL ESTUARINE RESEARCH RESERVE
JobosBayNationalEstuarineResearchReserve(JBNERR), offshorefromSalinas,PuertoRico,ispartoftheU.S.National EstuarineResearchReservesystem(Fig.1A).JobosBay, locatedonthesoutheastcoast,isthesecondlargestestuaryin
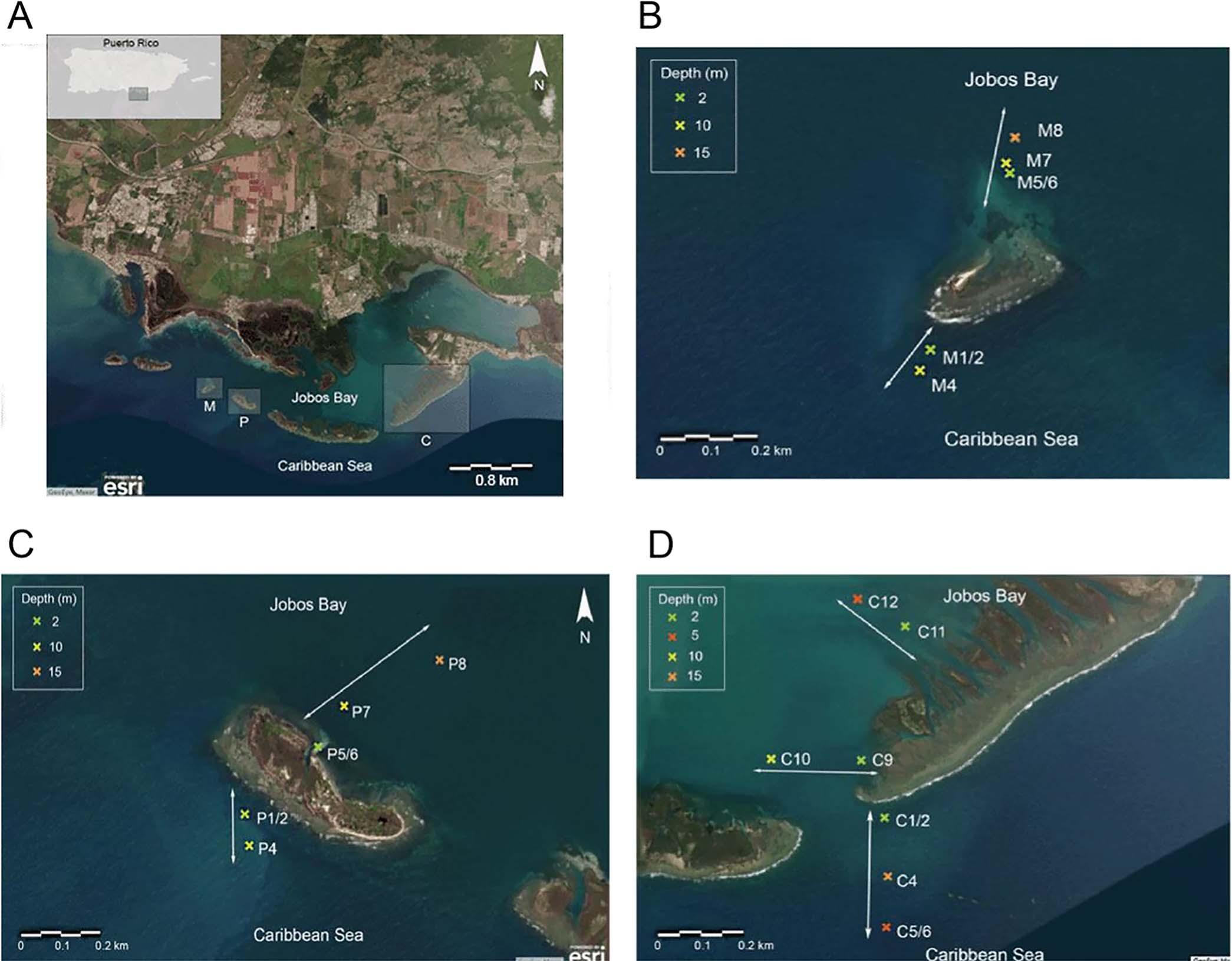
FIGURE 1.JobosBayNationalEstuarineResearchReserve,Salinas,PuertoRico.A)JobosBayandvicinity(insetPuertoRicowithshadedareashowinglocationofthelargerimage).LocationsofsamplingsitesinB)CayoMorillo,C)CayoPajaros,andD)CayoCaribe.
PuertoRico.Theexpansionofcoastalurbanareasaroundthe watershedexertstremendousecologicalpressureonthebay. Illegalcoastalconstructionbringsland-basedcontaminantsto thebay,affectingwaterqualityandbiota(e.g.,Alegríaetal., 2016;Martínez-Colónetal.,2021).Inaddition,JBNERR innerregionscontinuouslyreceivewarmwaterfromanearby ThermoelectricPowerPlant,apredominantsourceofthermal pollution,runoff,andoilspillcontamination.FormeragricultureandpharmaceuticalindustrieswithinSalinasandGuayama municipalitiesremainamongthenon-pointsourcesofchemical pollution.Asthedistancefromtheshorelinetothereefsison theorderof1–3km,gradientsfromturbid,nutrient-enriched terrestrialrunofftoveryclear,nutrient-poorCaribbeanwaters areverystrongandshiftwithweatherchanges,especiallyseasonally.Thus,thecoralreefsofJobosBayaresubjecttothe influenceoflocalstressors.
SeasonalityinPuertoRico,asinmanytropicalenvironments, isassociatedwithrainfall.PuertoRicotypicallyexperiencesa cooldryseasonfromDecemberthroughMarch,followedbya rainyseasonstartinginApril.Tropicalstormsystemsarecommon,especiallyinAugustandSeptember,producingheavy
rainfall,aswellasstrongwinds,waves,andcurrents.Forexample,in2017,twomajorstorms,hurricanesIrma(Category4) andMaría(Category5)devastatedPuertoRico.RainfallassociatedwithHurricaneMaríaresultedinasmuchas55cmofrainfallintheuplandwatershedand11–22cmintheimmediate vicinityofJobosBay(https://www.weather.gov/sju/maria2017).
CoralreefsarevitalecosystemsfortheprotectionofJobos BayandtheirmanagementisapriorityforJBNERR.Practices tobettermanagereefecosystemssubjecttoincreasingstressors,includingincreasingpollution,oceanacidification,and climatechange,areneededforeffectiveresourceconservation. TheJBNERRisanappropriatesystemtoutilizeindicesbased onforaminiferalassemblages,includingtheFI,aslong-term bio-monitoringtoolsforthereserve.Characterizingtheparametersinfluencingforaminiferalassemblagescanbekeyto understandingwhichecosystemscanpersistinthereserveand morebroadlyalongCaribbeancoastlines.
Theobjectivesofthispaperwere:a)tosummarizeenvironmentalconditionsseasonallyandspatially(i.e.,forereefand backreef)atselectedsitesatJBNEERin2018and2019;b)to collectandevaluateforaminiferalassemblagesatthosesites;
TABLE 1.Environmentalparameters[mean(standarderror)]inback-reefsitesbymonthandyear.BD 5 concentrationbelowdetection;DO 5 dissolved oxygen;TSS 5 totalsuspendedsolids.Asterisks(*) 5 nodata.
EnvironmentalParameters Mar-18Sept-18Dec-18Mar-19Aug-19Dec-19
#sites849886
Temperature(°C)26.63(0.11)29.81(0.08)27.43(0.13)26.43(0.03)29.56(0.07)27.47(0.09)
Salinity37.42(0.08)35.77(0.02)34.30(0.18)35.24(0.14)35.24(0.02)37.72(0.00) pH8.18(0.02)8.02(0.01)8.36(0.01)8.20(0.00)8.04(0.02)8.11(0.02) DO(%)103.2(2.7)106.7(3.0)102.1(2.5)101.6(1.2)99.4(2.4)99.6(1.2)
Totalchlorophyll(mg/L)1.11
TSS(mg/L)7.34(1.55)6.76(0.87)1.51(0.04)12.48(1.35)12.89(0.40)*
NH4þ(mM)0.02(0.01)0.0(0.05)*0.01(0.01)0.03(0.01)*
PO43 (mM)0.39(0.13)BD****
NOx (mM)1.24(0.54)5.10(0.33)*0.15(0.00)0.18(0.00)*
CaCO3 (%)22.4(5.7)18.7(2.9)21.8(4.4)23.1(4.9)27.1(3.8)24.3(3.7) Mud(%)70(13)82(6)74(8)75(11)91(13)66(11)
andc)totestthehypothesisthatassemblagesreflectseasonal changes(rainyvs.dryseasons)inenvironmentalparameters. TheoriginaldatasetisavailableinRosaMarín(2022).
METHODS
FIELD SAMPLING
ThesamplescollectedatJBNERRwerefromjustoffshore CayoMorillo,CayoPájaros,andCayoCaribe(Fig.1).At CayoMorilloandCayoPájaros,twotransectsweresampled offeachcay:oneeachintheforereefandbackreef.AtCayo Caribe,threetransectsweresampled:twoattheback-reefand oneatthefore-reef.Overall,eachtransectincludedtwoto threestationsbetween2–5mand10–15m,foratotalof17 stationsamongallseventransects(Fig.1B–D).Thegoalwas toconductsamplingin2018and2019atintervalsrepresenting rainy(August,September)anddryseasons(March,December),thoughweatherconditionspreventedfore-reefsampling inMarchandDecember2019(Table1).
Ateachstation,in-situwater-qualityparametersweremeasuredatthesurfaceandbottomofthewatercolumnusinga multiparametersonde(YSI-EXO2®).Theparametersmeasured weretemperature(°C),salinity(measuredasconductivityand reportedunitless),dissolvedoxygen(%DO)andpH.Two3.8L surfaceseawatersampleswere filteredonsitethroughaprecombusted0.44 mmglassmicrofiber filter(Whatmanglass microfiber filters,GradeGF/F)usingapump-linesystem.If weatherconditionsdidnotallowforon-site filtration,water sampleswerecollectedinNalgenepolypropylenecontainers and filteredattheJBNERRlaboratoryfacilities.The filters werestoredinaluminumfoilandfrozen( 80°C)untilfurther analysisfortotalchlorophyllandtotalsuspendedsolids(TSS).
Inaddition,three filteredwatersubsamples(0.5Leach) werestoredinautoclavedandpre-combustedamberbottles andfrozenat 18°Cfornutrientanalysisincludingorthophosphate(PO43 ),ammonium(NH4þ),andnitrateþnitrite(NOx). Forsedimentanalyses,approximately50gwerecollectedper stationandthesampleswerekeptinNalgenepolypropylene containers(0.5L).Dependingoncoralcoverage,thesediment sampleswerecollectedeitherbySCUBAdivingorusinga petite-ponargrab.Allsedimentsampleswerestoredat4°C
untilanalyzedinthelaboratoryforgrain-size,carbonatecontent(CaCO3%),andforaminiferalassemblages.
SEAWATER NUTRIENT ANALYSES
Seawatersampleswereanalyzedfor[PO43 ],[NH4þ],and [NOx].SpecificcolorimetricmethodswereusedforeachnutrientandtheabsorbancewasmeasuredusingaSpectraMaxM5 spectrophotometer(MolecularDevices,SanJose,CA).Orthophosphateanalysesfollowedthemodifiedprotocolofammoniummolybdate/potassiumantimonyltartrate/ascorbicacid methodofStrickland&Parsons(1972)formicroplateassay. Fivestandards(2.5 mM,5 mM,10 mM,15 mM,and20 mM) werecreatedusingtheprimarystandard(KH2PO4)anddeionized(DI)water.Tocompletetheassay,1.4mLofeachstandard andwatersamplewereaddedtoeachwellina48-microwell platefollowedby140 mLoftheworkingreagent.The[PO43 ] (mM)wasdeterminedbymeasuringtheabsorbanceat885nm. Themodifiedsalicylate-hypochloritemethodfromBower &Holm-Hansen(1980)wasusedtodetermine[NH4þ]inseawatersamples.Fourstandards(5 mM,10 mM,25 mM,50 mM) werecreatedusingtheprimarystandard(NH4Cl)withDIwaterfora100mL(finalvolume)usedforthecalibration curve.The[NH4þ](mM)wasdeterminedbymeasuringthe absorbanceat625nm.The[NOx]wasdeterminedcalorimetricallyusingtheprocedureofSchnetger&Lehners(2014).Five standardswerecreatedusingtheKNO3 asprimarystandard. ThentheNOx reagent(450 mL)and540 mLofeachstandard orsamplewereaddedtomicrocentrifugetubes(2mL).Each microtubewasmanuallymixedfor10seconds,thenheatedat 45°C 6 5°Cfor60mininanoven,andsubsequentlymixed againfor10additionalseconds.The[NOx](mM)inthesampleswasdeterminedbymeasuringtheabsorbanceat540nm. The[NH4þ]and[NOx]togetherindicatedrelativeavailability ofdissolvedinorganicnitrogen(DIN).
TOTAL CHLOROPHYLLAND SUSPENDED SOLIDS
Thetotalchlorophyllconcentrationsweredeterminedfollowingthe fluorescencemethodofArar&Collins(1997). Each filterfromeachsamplewasmanuallybrokendowninto piecesandplacedina15mLpolypropylenecentrifugetube with1mLof90%acetonesolution.Anadditional10mLof theacetonesolutionwasaddedpriortocentrifugation(1,000
rpmfor10minutes)toseparatethesolutioncontainingchlorophyllfromthe filterdebris.Tomeasurethe fluorescenceof eachsample,adilutionof2:1(2mLofthe90%acetonesolutionand1mLofthechlorophyllsolutionsample)wasmade incentrifugalglassvials(2mL).Then, fluorescencewasmeasuredusingaFluorometerTD-700(TurnerDesigns)and recordedasabsorbance(FSU).
ThemethodofVanderLinde(1998)wasusedtodetermine totalsuspendedsolids(TSS)inseawater.Priorto fieldwork andwater filtration, filterswerepre-combustedat550°Cfor fourhoursinafurnacetodeterminethedryandempty weights.After filtration,thewet filtersweredriedat60°Cfor 24hoursandthenre-weighed.TodeterminetheTSS(mg/L) contentinthewatersample,weightdifferencewascalculated anddividedbythetotalvolume filteredforeachsample.
SEDIMENT COMPOSITION
Muddysampleswereprocessedbybothwetanddrysieving; sandysampleswereonlydrysieved.Forthemuddysamples, 10–20g(dependingonavailablesamplesize)werewetsieved (,63 mm)toremovethemud(silt þ clay)particles.Afteroven drying(60°C)for24hours,thesampleswerere-weighedto determinetheamountofmudcontentbymassdifference.The residualsediments(.63 mm)weredrysievedthrough2-mm, 1-mm,500-mm,250-mm,125-mmand63-mmmeshsieves.For sandysamples,10gofsedimentwasdrysievedusingthesame meshsievesasabove.TheWentworthgradescalewasusedfor grain-sizeclassification(Wentworth,1922).
Todeterminecalciumcarbonate(CaCO3%)content,each drysedimentsubsample(1g)wasplacedinapre-cleanedand pre-weighedcrucible.Toensurecompletedryness,thesubsamplesweredriedinanovenat105°Cfor24hoursand,after coolinginadesiccatortoroomtemperature,crucibleswerereweighed.Eachsubsamplewas “baked” inamufflefurnaceat 550°Cforfourhoursand,aftercoolinginadesiccatortoroom temperature,crucibleswerere-weighed.Finally,CaCO3%was determinedbyheatinginamufflefurnaceat1000°Cforone hourandsubsequentlyre-weighingaftercoolinginadesiccatortoroomtemperature.
BENTHIC FORAMINIFERAL ANALYSES
Amicro-splitterwasusedtoreducethebulksedimentintoan evenlyrepresentativesubsampleforremovalofforaminiferal specimens.A1-gsubsampleisrecommendedintheprotocols ofHallocketal.(2003)andPrazeresetal.(2020).However, giventhepredominanceofsiliciclasticsedimentsandscarcity offoraminiferalspecimens,3gofsubsamplewereusedfor analysis.The3gwerewetsievedtodiscardanymud-sized (,63 mm)sedimentsandtheremainingsediment(.63 mm) wasdriedintheovenat60°Cfor24hours.Usingastereomicroscope,theforaminiferalspecimenswerepickedfromthe sedimentusinga0/18paintbrush.Specimenswerepickedfrom eachsubsampleuntilamaximumof200individualswerecollectedortheentire3gwereexamined.TheworkofLoeblich& Tappan(1987),theWorldRegisterofMarineSpecies(https:// www.marinespecies.org/index.php),andothersourceswere usedfortaxonomicclassification.
Foraminiferaldensity(FD 5 #/g)wascalculatedasthe numberofindividualsfounddividedbythemassofsediment
examined(3g).Speciesrichness(S)isthenumberofspecies identifiedinasample.TocalculatetheShannon-WienerIndex [H(S)]thefollowingequationwasused:
H 5 X S n 1 pi lnpi (e.g.,Hayek&Buzas,2010).ThePaleontologicalstatistics softwarepackageforeducationanddataanalysis(PAST; Hammeretal.,2001)wasusedtocalculatetothediversity indicesusingrawforaminiferalcounts.
ToapplytheFoRAMIndex(FI),theforaminiferalspecimens weredividedintothreefunctionalgroupsdefinedbyHallock etal.(2003),asmodifiedbyCarnahanetal.(2009).Thesefunctionalgroupsare:symbiont-bearing(s),stress-tolerant(o),and othersmalltaxa(h).TocalculatetheFI,thefollowingequation wasused:
FI 5 10 3 Ps ðÞ þ 2 3 Ph ðÞ þ Po ðÞ
wherePs 5 theproportionofsymbiont-bearingspecimensusing thetotalnumberofforaminiferalspecimens(T)inthesample, Ph 5 theproportionofOSF(othersmallforaminifers)inthe sample,andPo 5 theproportionofSTF(stress-tolerantforaminifers)inthesample.Forexample,Po 5 STF/Trepresentsthe numberofstress-tolerantbenthicforaminifersdividedbythetotal specimensfoundinthatsample;Ph andPs werecalculatedsimilarly.Afterthecalculation,thecriteriafortheFIvalues(Hallock etal.,2003)wereusedtoindicateifwater-qualityconditionssupportcoral-reefaccretioninJBNERR.
RESULTS
ENVIRONMENTAL PARAMETERS
Datafortemperature,salinity,pH,anddissolvedoxygen collectedatthetimesofsamplingweresummarizedbymonth, year,andreeflocation(Tables1,2).Temperaturesranged from »26°CinMarch,tohighsof29–30°CinAugust/Septemberforbothyears.SalinitiesinMarch2018,whichisnear theendofthedryseason,were .37.Inthelate-summerrainy season,salinitiesrangedfrom »34.6–35.6,withsimilarsalinitiespersistingintoDecemberin2018andMarch2019(Tables 1,2).ThepHvaluesalsoshowedaseasonaltrend,withintermediatevalues(8.1–8.2)inMarch,slightlylowervaluescoupledwiththelowersalinitiesinlateAugust/September,and highestvaluesinDecember2018(8.3–8.5).Dissolvedoxygen wasgenerallynearsaturationorsupersaturated.
Nutrientanalysesindicatedthatduringthelate-summerrainy seasonavailableDINwaslikelyinexcess,while[PO43 ]was belowdetection.InMarch,[PO43-]wasdetectableandDINwas lower,indicatingapossibleshifttonitrogenlimitation(Tables 1,2).Chlorophyllconcentrationsandtotalsuspendedsolidsare indicatorsofwatertransparencyandvariability.Totalchlorophyllconcentrationswererelativelyconsistentwithinyears (average:2018 5 7.0 3 10 4 mg/L;2019 5 9.0 3 10 4mg/L). ConcentrationsinAugust/Septemberwerelowerandlessvariablethaninotherseasons.Suspendedsolidsingeneralwere higherin2019(10.0–17.0mg/L)versus1.0–8.0mg/Lin2018.
TABLE 2.Environmentalparameters[mean(standarderror)]infore-reefsitesbymonthandyear.BD 5 concentrationbelowdetection;DO 5 dissolved oxygen;TSS 5 totalsuspendedsolids.Asterisks(*) 5 nodata.
EnvironmentalParameters Mar-18Sept-18Dec-18Aug-19
#sites6454
Temperature(°C)26.32(0.04)*27.32(0.00)29.54(0.02)
Salinity37.28(0.01)*34.46(0.01)34.58(0.01)
pH8.19(0.01)*8.36(0.01)8.03(0.00)
DO(%)98.0(1.1)*96.5(0.2)100.1(0.4)
Totalchlorophyll(mg/L)9.32
TSS(mg/L)4.61(0.67)3.90(0.28)1.21(0.09)11.82(0.56)
NH4þ(mM)0.04(0.02)0.15(0.12)*0.03(0.01)
PO43 (mM)0.34(0.16)BD**
NOx (mM)0.81(0.32)5.07(0.43)*0.18(0.00)
CaCO3 (%)33.0(4.9)30.2(2.6)40.5(1.7)39.0(1.3)
Mud(%)24(15)33(20)30(17)7(1)
ConcentrationsofsuspendedsolidsinDecember(bothyears) werelowerthaninothermonths(Tables1,2).
Grain-sizeanalysisrevealedthatback-reefsitesgenerally weredominatedby finesediments,whilesedimentsonthe fore-reefweremostlygranulesandcoarsesand(AppendixA).
Calciumcarbonate(CaCO3%)insedimentvariedsomewhat byseason,withlowervaluesinAugust/Septemberandhighest inDecember(Tables1,2).
BENTHIC FORAMINIFERAL ASSEMBLAGES
Fromatotalof64surface-sedimentsamples,4,753foraminiferaltestswerepickedandatotalof117taxawereidentified (AppendixB).Fromthe64samples,45back-reefsamplesand 19fore-reefsampleswereanalyzed.Intheback-reefatotalof 3,533foraminiferaltestswerepickedandatotalof115taxa wereidentified.Inthefore-reefatotalof1,220foraminiferal testswerecountedfromwhich96taxawereidentified.Ingeneral, Amphisteginagibbosa (Am.gibbosa)testsdominatedin fore-reefsamples,whilesmallerrotaliidssuchas Rotorbinella rosea and Discorbis spp.,andmiliolids,especially Quinqueloculina spp.,dominatedinback-reefsamples(Figs.2,3).In MarchandDecember2019,inclementweatherpreventedsedimentsamplingatfore-reefstations.
Back-reefstationsshowedstrongvariabilityacrosssamplingevents.InMarch2018, R.rosea madeup19%ofthe testscounted,with Quinqueloculina spp.makingup15%,and twosymbiont-bearingtaxa[Am.gibbosa 11%and Archaias angulatus 6%(subsequently Ar.angulatus)]foundwithsimilar abundancesasthetwocommonstress-tolerantgenera(Elphidium 7%and Ammonia 6%;Fig.2-A).Otherforaminiferal taxawithrelativeabundances .5%comprised36%ofthose foundand19%comprisedaccessorytaxathatweredefinedas anytaxaforwhichrelativeabundancewas .1%intheassemblage.InSeptember2018,tworotaliids, D.mira and R.rosea, togethercomprised36%ofthespecimens,followedby Am. gibbosa (13%), Quinqueloculinaspp. (11%), Ammonia (7%), and Ar.Angulatus (5%)(Fig.2-C).Otherforaminiferaltaxa withrelativeabundance ,5%comprisedtheremaining28%, fromwhich12%wereaccessorytaxa. Quinqueloculina spp (26%)dominatedtheback-reefsamplesinDecember2018, followedby Triloculina spp.At12%.Inaddition, Am.gibbosa and R.rosea werefoundat6–7%,otherscomprisedoftaxa (49%)thatdidnotexceed5%thetotal(Fig.2-E).
Smallertaxadominatedtheback-reefsamplesinMarch 2019,makingup57%( Quinqueloculina spp.33%, Pyrgo spp.18%,and Discorbis spp.6%);symbiont-bearing Am. gibossa (8%)andthestress-tolerant Ammonia (7%)were alsocommon.Theotherforaminiferaltaxathatindividually didnotexceed5%comprised29%ofspecimenscounted (Fig.2-B).InAugust2019, Quinqueloculina spp.againdominated(34%),followedby Am.gibbosa (15%), Discorbis spp.(10%),and Pyrgo spp.(7%).Otherforaminiferaltaxa presentat ,5%comprisedtheremaining34%ofthosefound (Fig.3-D).InDecember2019, Discorbis spp.(24%)and Quinqueloculina spp.(24%)co-dominated,whilesymbiontbearingtaxaincluded Am.gibbosa (13%)and Ar.angulatus (5%;Fig.2-F).Stress-tolerant Ammoniabeccarii (6%)and Elphidium spp.(3%)werealsopresent.
Infore-reefstations,theproportionof Am.gibbosa in March2018was37%,followed Quinqueloculina spp.(15%) and R.rosea (14%;Fig.3-A).Theproportionof Am.gibbosa inSeptember2018was39%,withtworotaliids, R.rosea (26%)and Discorbis spp.(10%)togethercomprising36%of thespecimens,followedbysymbiont-bearing Soritesmarginalis (5%;Fig.3-B).Theproportionof Am.gibbosa waslowest inDecember2018(29%),followedby Quinqueloculina spp. (23%), Discorbis spp.(10%),and Ar.angulatus (9%;Fig.3C).InAugust2019, Am.gibbosa (49%)dominated,followed byrotaliidsandmiliolids,including Quinqueloculina spp. (19%)and Discorbis spp.(10%;Fig.2-D).
BENTHIC FORAMINIFERAL ECOLOGICAL INDICES
Densitiesofforaminiferalspecimensinthesedimentswere low(Tables3,4),consistentwithrelativelylowproportionsof CaCO3 (Tables1,2).Back-reefsitesaveragedfrom3to67 individualspergram,fore-reefstationsaveragedfrom1to46 individualspergram.Consistentwithlowdensities,species richnesspersamplewaslow;mostofthe127specieswere rare.Inback-reefsamples,meanspeciesrichnessrangedfrom alowof11inSeptember2018,downfromthehighof24in March2018,thenincreasingto23inDecember2018(Table 3).Incontrast,themeanspeciesrichnessinfore-reefstations waslowestinMarch2018andhighestinDecember2018 (Table4).
MeanShannonDiversityIndex(SDI)valuesforback-reef sitesin2018rangedfrom1.9inSeptember2018to2.9in
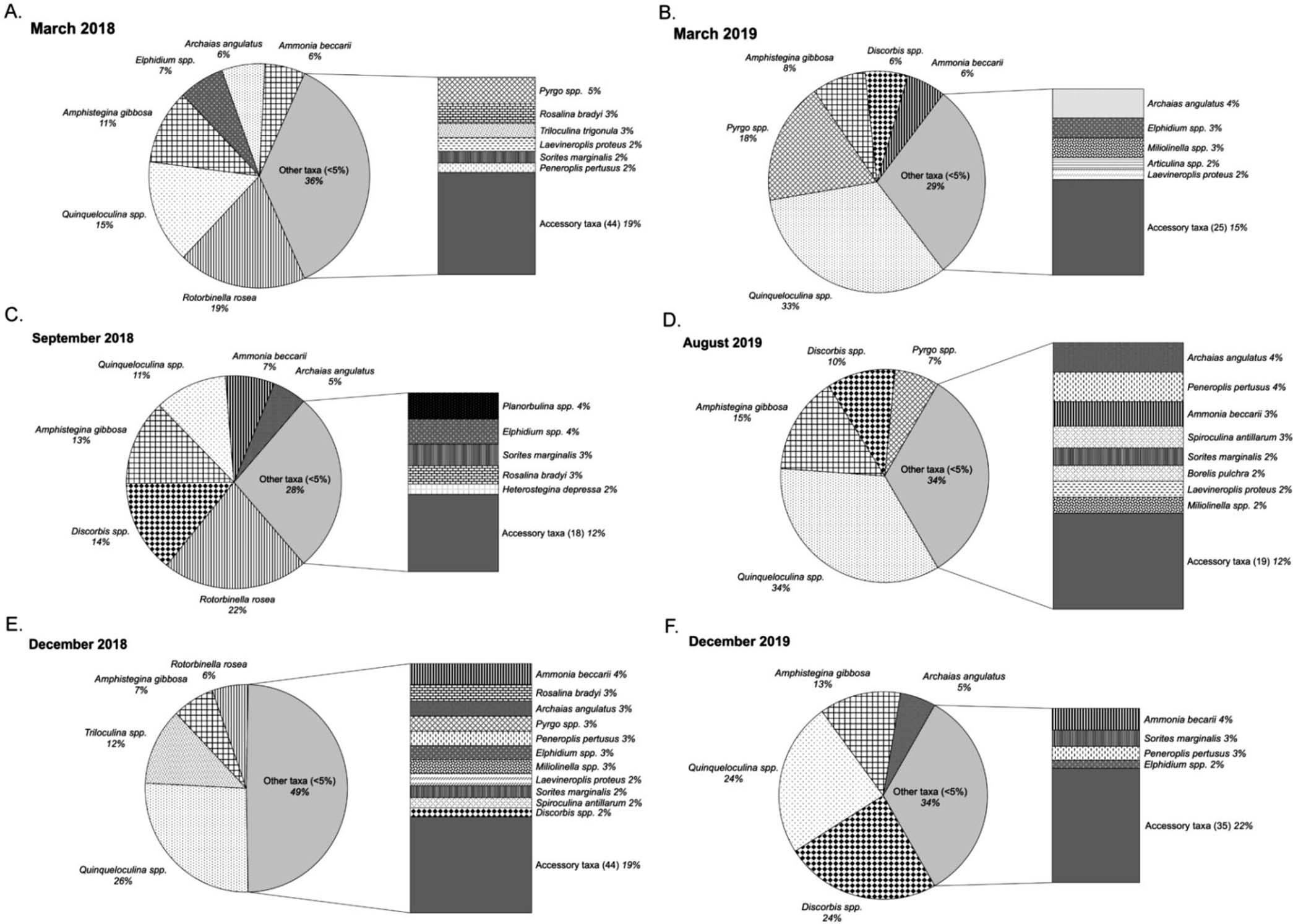
FIGURE 2.Relativeabundancesinback-reefsitesbymonthandyear:A)March2018,B)March2019,C)September2018,D)August2019,E) December2018,andF)December2019.Thepiechartsshowtaxawhoserelativeabundanceswere
$5,whilethebarsshowtaxapresentat ,5%and accessorytaxapresentat ,1%(numberofspeciesshowninparentheses).
December2018(Table3).MeanSDIvaluesinfore-reefsites werelowestinMarch2018(1.2)andagainhighestinDecember2018(1.7).Notethatfore-reefstationswerenotsampled inMarchandDecember2019becauseofinclementweather conditions.ThemeanFoRAMindicesreflectedtheconsistent presenceofsymbiont-bearingforaminifersinbothback-reef andfore-reefsamples.TheFIintheback-reefsiteswereinthe 3–4range,whilefore-reefvalueswereinthe6–8range.
DISCUSSION
SEASONAL VARIATIONSIN ENVIRONMENTAL PARAMETERSAND ECOLOGICAL INDICES
Theoriginalobjectivesofthisstudyweretoassessforaminiferalassemblagesandenvironmentalconditionsseasonallyand spatiallyaroundcaysthatdefineJobosBay,PuertoRico,totest thehypothesisthatthoseassemblagesreflectseasonalchanges (rainyvs.dryseasons).ThechoicetosampleinDecember,at thebeginningofthedryseason,andMarch,attheendofthe dryseason,wasoriginallyplannedtocharacterizedryseason conditions.Incontrast,AugustandSeptemberarecommonly thepeakoftherainyandtropicalstormseason.
InMarchbothyears,theback-reefenvironmentalparametersandtheforaminiferalassemblagesweregenerallysimilar (inclementweatherpreventedfore-reefsamplingin2019). Thehighest[PO43-]wererecordedinMarch,indicatingpossibleinorganicnitrogen(DIN)limitation([NOx]and[NH4]). Temperatureswerethecoolest,thoughsalinitywasnotably lowerin2019(37in2018,35in2019;seeTable2).
Inback-reefandfore-reefsites,temperaturewashighestin AugustandSeptember,salinitywasintermediate(34.6–35.8),and DINwasinexcess,while[PO43-]wasbelowdetectionin2018(no [PO43-]dataareavailablefor2019),possiblyindicatingeffectsof runoff.Theforaminiferalassemblagesin2018wereleastdiverse (10–11speciespersample,SDI 5 1.9).Incontrast,inAugust 2019,densitywasstilllow(14–26individuals/g),butspeciesrichness(13–17)andSDI(2.4)intheback-reefwerenotablyhigher (seeFig.2-C,D).Moreover,in2018theback-reefsampleswere dominatedby R.rosea,aspeciesoftenfoundinhigherenergysettings,whilein2019theassemblagesweredominatedby Quinqueloculina spp.Intheforereef,samplesweredominatedby Am. gibbosa and R.rosea, bothspeciesthatoccurinhydrodynamic environments(e.g.,Trifflemanetal.,1991;Crevisonetal.,2006). InDecember2018,salinitieswerelowestinbothback-reef andfore-reefsites(»34.4),indicatingtheinfluenceofterrestrial
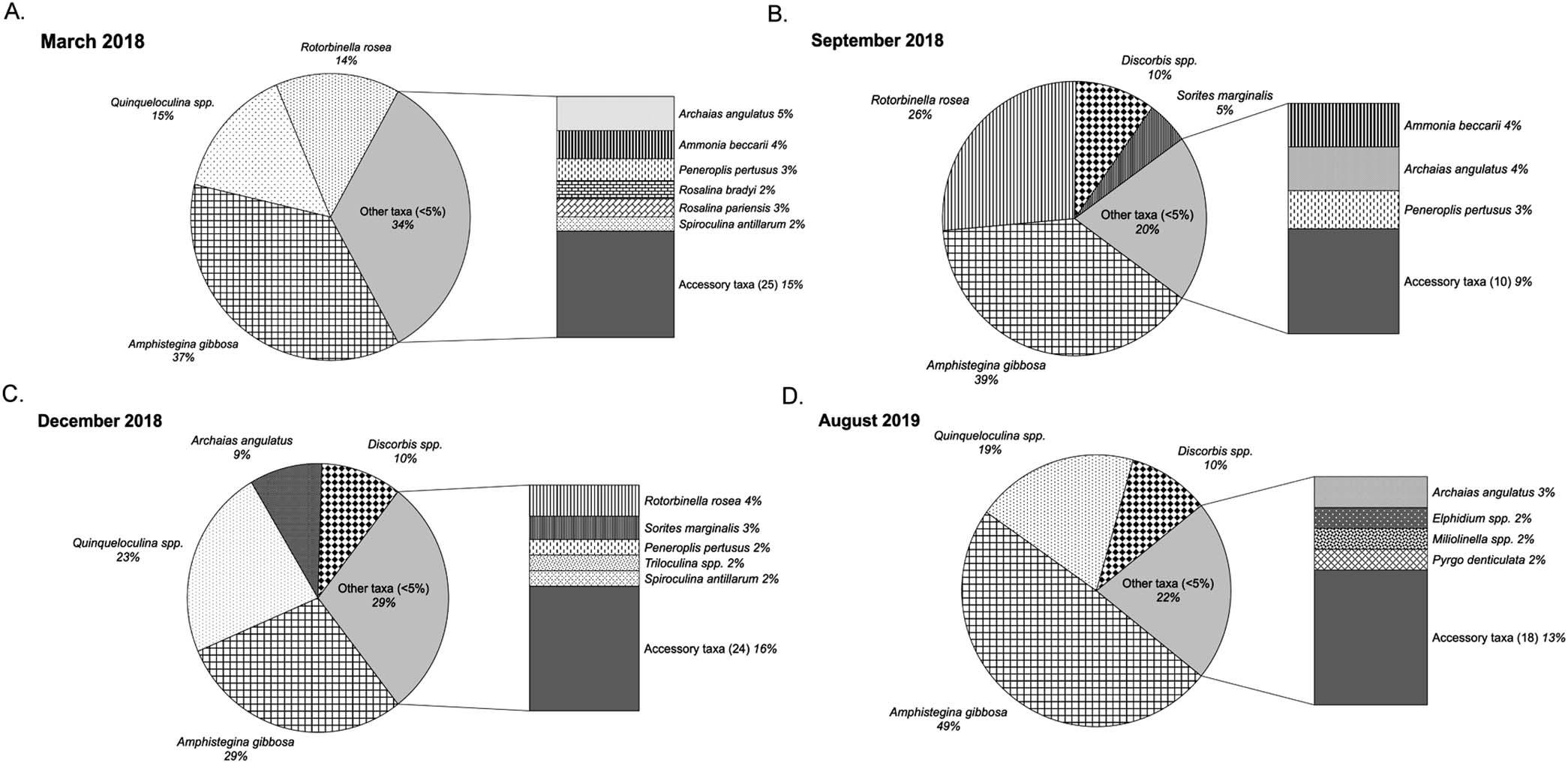
FIGURE 3.Relativeabundancesinfore-reefsitesbymonthandyear:A)March2018,B)September2018,C)December2018,andD)August2019. Thepiechartsshowtaxawhoserelativeabundanceswere $5%,whilethebarsshowtaxapresentat ,5%andaccessorytaxapresentat ,1%(numberof speciesshowninparentheses).
runoff.Theforaminiferalassemblagesintheback-reefsites werequitediverse,withanaverageof23speciespersample andthehighestmeanSDI(2.86).InDecember2019,incontrast,salinitieswerethehighestfound,highereventhanMarch 2018,andspeciesrichness,SDI,andassemblagecomposition werealsomoresimilartoMarchassemblages.
Fortunately,nomajorhurricanesdirectlyhitPuertoRicoin 2018or2019.TheonlynotablestormreportedtoaffectsoutheastPuertoRicoin2018occurredinJuly.TheremnantsofHurricaneBerylpassedsouthofPuertoRico,deliveringheavyrain ontheeastsideofthemainisland.Thus,samplinginSeptember2018occurredaftertheonlymajorstormin2018(https:// www.nhc.noaa.gov/archive/2018/).In2019,samplingoccurred inmid-August,priortoHurricaneDorianpassingjusteastof PuertoRicoinlateAugust,withwindsandheavyrain.Roughly amonthlater,TropicalStormKarenalsopassedalongtheeast coastofPuertoRico(https://www.nhc.noaa.gov/archive/2019/). Thus,thedifferencesintheforaminiferalassemblagesbetween September2018andAugust2019mayreflectdifferencesin
timingofstormactivity.SamplinginSeptember2018occurred afterHurricaneBeryl’spassageandmayreflecttheinfluenceof windandstormwaves,withlowerspeciesrichnessandhigh relativeabundancesofhyalinetaxa.Porcelaneoustaxasuchas Quinqueloculina spp.areoftensmall,relativelyfragile,and readilywinnowedbywaveaction(e.g.,Wetmore,1987).Those arethetaxathataremuchmoreprevalentinsamplesfrom August2019,whichwerecollectedbeforemajorstormpassage.
ThedifferencesinassemblagesbetweenDecember2018and 2019showtheoppositetrendofthelatesummersamples.In 2018,afteralesshydrodynamicbutrainylatesummerand autumn,theforaminiferalassemblagesweremuchmorediverse anddominatedby Quinqueloculina spp.andothersmallermiliolids.In2019,afteramoreactivetropicalstormseason,hyalinetaxamadeuphalftheassemblage,though Quinqueloculina spp.werealsoabundant.
TheeffectsofstormactivitymayalsoprovideinsightintodifferencesinMarchassemblages.Theback-reefassemblagesin March2018were .40%hyalinetaxa.Therelativeabundanceof
TABLE 3.Foraminiferalassemblagesandecologicalindices[mean(standarderror)]persiteinback-reefstationsbymonthandyear.Functional-group percentagesincludeaccessorytaxa(presentin ,1%).
ForaminiferalAssemblages Mar-18Sept-18Dec-18Mar-19Aug-19Dec-19 #samples849896 #countedpersample131(12)81(19)81(14)68(15)43(7)75(20) Density(individuals/g)43(5)27(6)29(4)68(13)14(2)75(20) #taxa24(3)11(2)23(2)18(3)17(2)20(4) SDI2.55(0.21)1.91(0.26)2.86(0.15)2.62(0.17)2.45(0.10)2.42(0.23) Evenness0.61(0.07)0.69(0.08)0.82(0.04)0.87(0.08)0.76(0.04)0.65(0.08) FI4(1)4(1)4(0)3(1)4(0)4(1) Stress-toleranttaxa13%11%7%9%4%6% Othersmallertaxa65%62%75%74%68%71% Symbiont-bearingtaxa23%26%19%17%28%24%
TABLE 4.Foraminiferalassemblagesandecologicalindices[mean(standarderror)]persiteinfore-reefstationsbymonthandyear.Functional-grouppercentagesarealsoshownincludingaccessorytaxa(presentin ,1%).
ForaminiferalAssemblages
Mar-18Sept-18Dec-18Aug-19 #samples7354 #countedpersample37(14)71(0)68(27)78(21) #species7(3)10(1)15(3)13(3) Density(individuals/g)17(6)24(0)54(29)26(7) SDI1.22(0.40)1.66(0.04)1.74(0.48)1.52(0.17) Evenness0.81(0.13)0.57(0.08)0.72(0.13)0.40(0.05) FI8(1)6(1)6(2)7(1) Stress-toleranttaxa5%5%2%2% Opportunistictaxa48%42%52%39% Symbiont-bearingtaxa67%53%46%57%
R.rosea (19%)andrelativelyhighspeciesrichnessmayreflect residualeffectsoftheextreme2017hurricaneseason,whentwo majorstorms,hurricanesIrma(Cat.4)andMaría(Cat.5)devastatedPuertoRico.InMarch2019,afteraquietstormseasonin 2018,relativeabundanceofhyalinetaxawas »20%ofthe assemblages,andproportionsofporcelaneous Quinqueloculina weredoublethosein2018.
Additionalenvironmentalparametersmustbeconsidered,especiallywhenconsideringsedimentwinnowing.Highproportionsof CaCO3 andlowproportionsofmudinAugust2019,alongwith coarsersedimentsincludingcoralandshellfragments,reflect hydrodynamicevents.Butoverall,sedimentsofJobosBayand reefsaremixedbiogeniccarbonatesandterrigenoussiliciclastics, withthelatterdominating,especiallyinthebackreef.Becausesiliciclasticsaregenerallymuchharder(6–7onMohsscale)compared tobiogeniccarbonates(3–4),wavewinnowingalsopredominantly breaksdownmorefragilecarbonategrains,includingthetestsof smallerforaminifers(e.g.,Wetmore,1987).
THE FORAMINDEX
The “ForaminiferainReefAssessmentandMonitoring” , abbreviatedastheFoRAMIndexorFI,wasdevelopedasan indicatorofwhetherwaterqualityissufficientlyoligotrophic tosupportcarbonatesedimentaccumulationbyorganismsthat hostalgalendosymbionts,suchasreef-buildingcoralsand largerbenthicforaminifers.Hallocketal.(2003)usedforaminiferalassemblagesinsamplesfromLaParguera,PR(Donnelly,1993)andtheFloridaReefTract(Cockeyetal.,1996) todevelopthisindex.Oliveretal.(2014)appliedtheFIinLa Parguera,concludingthatforaminiferalassemblageswerethe mostresponsiveindicatortogradientsofanthropogenicinfluence.ThoughdevelopedbasedonwesternAtlanticandCaribbeanforaminiferalassemblages,theFIhassincebeenapplied worldwide(Prazeresetal.,2020,andreferencestherein).
Overall,theFIvalueswere3–4forback-reefsitesand6–8for fore-reefsitesofthethreeJoboscays.Giventheproximityofterrestrialinfluence,thesevaluesindicatethattheinfluenceofCaribbeanwatersallowswaterqualitytosupport Am.gibbosa,whichis aprevalentsymbiont-bearingforaminiferalspeciesthroughoutthe westernAtlanticandCaribbean.Inaddition,temporalchangesin theoverallassemblagessuggestthatwaveactionassociatedwith tropicalstormactivitymayhelpdiluteandpossiblyremovepollutantsassociatedwithterrestrialrunoffandgroundwaterdischarge.
Twoadditionalfactorsexplainthepersistenceof Am.gibbosa asakeymemberoftheforaminiferalassemblages,despite intermittentunfavorableconditions:dormancyandpropagules. Forexample,Ross&Hallock(2019)reportedthatindividuals ofthisspeciescansurviveandrecoverafter20monthsinaphoticconditions.Andmoreimportantly,Alve&Goldstein(2002, 2003,2010)demonstratedthattinypropagules,primarilyproducedbysexualreproduction,provideakeydispersalmechanism forbenthicforaminifers. Amphistegina propagulesundoubtedly arecarriedinwarmwesternAtlanticandCaribbeanwaters,and whentheysettleintoasuitablehabitat(orintoanintermittently suitablehabitat),theycandevelop,grow,andasexuallyreproduce.
Hallocketal.(2003)discussedthelimitationofapplying theFIinshallowwaterbecausehigh-energyenvironments generallyhavewell-sortedsediments,fromwhichsmallerheterotrophictaxahavebeenbrokendownandwinnowedout. Prazeresetal.(2020)discussedwaystostandardizetheFIprotocol,suggestingtheeliminationofsitesshallowerthan3m andthosewithpredominantlycoarsetextures(.2mm).Prazeresetal.(2020)furtherrecommendedagainstusingsamples containing ,50specimenspergramofsample.Althoughterrigenoussedimentsarementionedinpreviousstudies,the dilutionandprobabilityofdestructionofcarbonatesediments werenotaddressed.
Thus,sedimentsfromthevicinityofJobosreefshaveavarietyofcharacteristicsthatdon’t fittheprotocolsforuseoftheFI asrecommendedbyPrazeresetal.(2020).Avarietyofdepths canbeencounteredinatransect ,100mlong.Thecarbonate fractionsareclearlydilutedbyterrigenoussiliciclasticsediments,whichiswhy3-gsampleswereassessedratherthanthe standard1-g.Insuchsediments,even5-gsampleswouldnotbe unrealistic.
TheJBNERRpresentsanexcellentexampleofwhystudysites oftendon’t fit “standard” definitions.Thereefsborderacoastalplainestuaryfromwhichbothsurfaceandgroundwatersdischarge. Thecoastalareahasalonghistoryofanthropogenicdisturbance, thoughsinceestablishmentofthereserve,muchofthecoastlineis vegetatedbymangroves.Thereefsareonlyafewkilometersoffshorefromthemuddymangroveshoreline.And,asnotedabove, thesediments,evenaroundthereefs,arepredominantlysiliciclastic. Indeed,foraminiferaldensitiesinfore-reefandback-reef sitesofJoboscayslimittheFIinterpretation.Nevertheless, theforaminiferalassemblagesshowedthepredominanceof smallheterotrophictaxain finersedimentsfromtheback-reef sitesandsymbiont-bearingtaxafromfore-reefsiteswith
coarsergrainsandhigherproportionsofCaCO3.Inaddition, thechangesinproportionsofhyalinetaxasuchas R.rosea, alongwith Am.gibbosa,providedadditionalinsightintothe influenceofhydrodynamicsintheJobosBaysystem.
CONCLUSIONS
TheessentialcharacteristicthatallowsthereefstobepresentatJobosBayistheproximityofclear,oligotrophicCaribbeanwater.Thesalinitiesrecorded,eveninback-reefareas, consistentlyexceeded34andmostlyexceeded35.Theproximityof,andexchangewith,Caribbeanwatersprovidesthe reefs,cays,andthebackreefsnearthecayswithwatersthat supportforaminifersandcoralsthathostalgalsymbiontsand areimportantproducersofcarbonatesediments.Thevariabilityintheforaminiferalassemblagesrecordedinthistime seriesindicatethattropicalstormsenhance flushingofbackreefandlagoonwaters,limitingeutrophicationandallowing theforaminifershostingalgalsymbiontstomaintainsufficient presencethattheFIvaluesof4predominatedinback-reef sites,withFI $6infore-reefsites.
Finally,thisworkdemonstratesthevalueoftime-seriesstudiesthatprovideessentialinsightintovariabilitythatnaturally occursintropicalcoastalwaters.Theoriginalhypothesiswas thatassemblagesinsamplescollectedduringdry-seasonmonths ofDecemberandMarchwouldcontrastwiththosefoundduring therainyseason.Therealityismorecomplex,andtheinfluence oftropical-stormsystemsisanessentialconsideration.
ACKNOWLEDGMENTS
ThankstoJobosBayNationalEstuarineResearchReserve forallowingustodo fieldworkandcontributetotheneedsof thereserve.ThankstoÁngelDieppa,MiltonMuñozHincapié, LuisD.OrtizSerrano,andAitzaPabónfortheircollaboration inthe fieldwork,instruments,andfacilities.Wearegratefulto JohannysJiménezCollazo,LexaMedero,Zakiya,Marcus, Kyle,andDantefortheirassistanceindatacollectingand processing.ARMisgratefultotheGeoLatinasorganization forthespacetotestandsolidifyideasandforsupport.This researchwaspossiblebyfundingfromSeaGrantPuertoRico (#NA18OAR4170089)andNOAAEducationalPartnership ProgramwithMinorityServingInstitutionsCooperative Agreement(#NA16SEC4810009).
REFERENCES
Alegría,H.,Martinez-Colon,M.,Birgul,A.,Brooks,G.,Hanson,L.,and Kurt-Karakus,P.,2016,HistoricalsedimentrecordandlevelsofPCBs insedimentsandmangrovesofJobosBay,PuertoRico:Scienceofthe TotalEnvironment,v.573,p.1003–1009.
Alve,E.,1995,Benthicforaminiferalresponsestoestuarinepollution:A review:JournalofForaminiferalResearch,v.25,p.190–203,DOI: 10. 2113/GSJFR.25.3.190
Alve,E.,andGoldstein,S.T.,2002,Restingstageinbenthicforaminiferal propagules:Akeyfeaturefordispersal?Evidencefromtwoshallowwaterspecies:JournalofMicropalaeontology,v.21,p.95–96,DOI: 10.1144/jm.21.1.95
Alve,E.,andGoldstein,S.T.,2003,Propaguletransportasakeymethod ofdispersalinbenthicForaminifera(Protista):LimnologyandOceanography,v.48,p.2163–2170,DOI: 10.4319/lo.2003.48.6.2163
Alve,E.,andGoldstein,S.,2010,Dispersal,survivalanddelayedgrowth ofbenthicforaminiferalpropagules:JournalofSeaResearch,v.63,p. 36–51,DOI: 10.1016/j.seares.2009.09.003
Arar,E.J.,andCollins,G.B.,1997,Method445.0InVitroDetermination ofChlorophyll-a andPheophytin a inMarineandFreshwaterAlgaeby Fluorescence:U.S.EnvironmentalProtectionAgency,Washington, D.C.,accessedat https://cfpub.epa.gov/si/si_public_record_report. cfm?Lab5NERL&dirEntryId5309417
Barbosa,C.,Ferreira,B.,Seoane,J.,Oliveira-Silva,P.,Gaspar,A.L.B., Cordeiro,R.C.,andSoares-Gomes,A.,2012,Foraminifer-basedcoral reefhealthassessmentforsouthwesternAtlanticoffshorearchipelagos, Brazil:JournalofForaminiferalResearch,v.42,p.169–183,DOI: 10. 2113/GSJFR.42.2.169
BarragánMontilla,S.,andSanchezQuiñonez,A.,2021,PaleoenvironmentalinferencesbasedonbenthicforaminiferafromtheUitpaFormation(Guajira,Colombia):Micropaleontology,v.67,p.459–81, DOI: 10.47894/mpal.67.5.03
Beyer,H.L.,Kennedy,E.V.,Beger,M.,Chen,C.A.,Cinner,J.E., Darling,E.S.,Eakin,C.M.,Gates,R.D.,Heron,S.F.,andKnowlton, N.,2018,Risk-sensitiveplanningforconservingcoralreefsunder rapidclimatechange:ConservationLetters,v.11,e12587,DOI: 10. 1111/conl.12587
Bonkosky,M.,Hernández-Delgado,E.A.,Sandoz,B.,Robledo,I.E., Norat-Ramírez,J.,andMattei,H.,2009,Detectionofspatial fluctuationsofnon-pointsourcefecalpollutionincoralreefsurroundingwaters insouthwesternPuertoRicousingPCR-basedassays:MarinePollution Bulletin,v.58,p.45–54,DOI: 10.1016/j.marpolbul.2008.09.008
Bower,C.E.,andHolm-Hansen,T.,1980,Asalicylate-hypochloritemethod fordeterminingammoniuminseawater:CanadianJournalofFisheries andAquaticSciences,v.37,p.794–798,DOI: 10.1139/f80-10
Carnahan,E.A.,Hoare,A.M.,Hallock,P.,Lidz,B.H.,andReich,C.D., 2009,ForaminiferalassemblagesinBiscayneBay,Florida,USA: Responsestourbanandagriculturalinfluenceinasubtropicalestuary: MarinePollutionBulletin,v.59,p.221–233,DOI: 10.1016/j.marpolbul. 2009.08.008
Cockey,E.,Hallock,P.,andLidz,B.H.,1996,Decadal-scalechangesin benthicforaminiferalassemblagesoffKeyLargo,Florida:Coral Reefs,v.15,p.237–248,DOI: 10.1007/BF01787458
Crevison,H.,Hallock,P.,andMcRae,G.,2006,Sedimentcoresfromthe FloridaKeys(USA):Isresolutionsufficientforenvironmentalapplications?:JournalofEnvironmentalMicropaleontology,Microbiology, andMeiobenthology,v.3,p.61–82.
Donnelly,K.B.,1993,Characterizationoftheforaminiferalassemblage offthesouthwestcoastofPuertoRicoatLaParguera:M.Sc.Thesis, UniversityofSouthFlorida,Tampa,Florida,p.1–272.
Eddy,T.D.,Lam,V.W.Y.,Reygondeau,G.,Cisneros-Montemayor, A.M.,Greer,K.,Palomares,M.L.D.,Bruno,J.F.,Ota,Y.,and Cheung,W.W.L.,2021,Globaldeclineincapacityofcoralreefsto provideecosystemservices:OneEarth,v.4,p.1278–1285,DOI: 10. 1016/j.oneear.2021.08.016
ElKateb,A.,Stalder,C.,Martinez-Colon,M.,Mateu-Vicens,G., Francescangeli,F.,Coletti,G.,Stainbank,S.,andSpezzaferri,S,2020, Foraminiferal-basedbioticindicestoassesstheecologicalqualitystatus oftheGulfofGabes(Tunisia):EcologicalIndicators,v.111,DOI: 10. 1016/j.ecolind.2019.105962
Frontalini,F.,andCoccioni,R.,2011,Benthicforaminiferaasbioindicatorsofpollution:AreviewofItalianresearchoverthelastthree decades:RevuedeMicropaléontologie,v.54,p.115–127,DOI: 10. 1016/j.revmic.2011.03.001
Gonzales,M.B.,Heyres,L.J.,Monteclaro,H.M.,delNorte-Campos,A.G., andSantander-deLeon,S.M.S.,2022,BenthicforaminiferaasbioindicatorofcoralreefconditioninNogasIsland,Philippines:RegionalStudiesinMarineScience,v.52,DOI: 10.1016/j.rsma.2022.102352
Hallock,P.,Lidz,B.H.,Cockey-Burkhard,E.M.,andDonnelly,K.B., 2003,Foraminiferaasbioindicatorsincoralreefassessmentandmonitoring:TheFoRAMIndex:EnvironmentalMonitoringandAssessment,v.81,p.221–238.
Hammer,Ø.,Harper,D.A.T.,andRyan,P.D.,2001,PAST:Paleontologicalstatisticssoftwarepackageforeducationanddataanalysis:PalaeontologiaElectronica,v.4,p.1–9.
Hayek,L.-C.A.,andBuzas,M.A.,2010,SurveyingNaturalPopulations: ColumbiaUniversityPress,NewYork,590p.
Hernández-Delgado,E.A.,andOrtíz-Flores,M.F.,2022,Thelongand windingroadofcoralreefrecoveryintheAnthropocene:Acasestudy fromPuertoRico:Diversity-Basel,v.14,DOI: 10.3390/d14100804
Hernández-Delgado,E.A.,Hutchinson-Delgado,Y.M.,Laureano,R., Hernández-Pacheco,R.,Ruiz-Maldonado,T.M.,Oms,J.,andDíaz, P.L.,2011,Sedimentstress,waterturbidity,andsewageimpactson threatenedelkhorncoral(Acroporapalmata)standsatVegaBaja, PuertoRico:Proceedingsofthe11thInternationalCoralReefSymposium,p.83–92,accessedat http://aquaticcommons.org/id/eprint/ 15512
Hoegh-Guldberg,O.,Mumby,P.J.,Hooten,A.J.,Steneck,R.S., Greenfield,P.,Gomez,E.,Harvell,C.D.,Sale,P.F.,Edwards,A.J., Caldeira,K.,Knowlton,N.,Eakin,M.,Iglesias-Prieto,R.,Muthiga, N.,Bradbury,R.H.,Dubi,A.,andHatziolos,M.E.,2007,Coralreefs underrapidclimatechangeandoceanacidi fi cation:Science,v.318, p.1737 –1742,DOI: 10.1126/science.1152509
Hoegh-Guldberg,O.,Kennedy,E.V.,Beyer,H.L.,McClennen,C.,and Possingham,H.P.,2018,Securingalong-termfutureforcoralreefs: TrendsinEcologyandEvolution,v.33,p.936–944,DOI: 10.1016/j. tree.2018.09.006
Jaap,W.C.,1985,Anepidemiczooxanthellaeexpulsionduring1983in thelowerFloridaKeyscoralreefs:Hyperthermicetiology:ProceedingsoftheFifthInternationalCoralReefCongress,v.6,p.7. Lang,J.C.,Lasker,H.R.,Gladfelter,E.H.,Hallock,P.,Jaap,W.C., Losada,F.J.,andMuller,R.G.,1992,Spatialandtemporalvariability duringperiodsof “recovery” aftermassbleachingonWesternAtlantic coralreefs:AmericanZoologist,v.32,p.696–706.
Larsen,M.C.,andWebb,R.M.T.,2009,Potentialeffectsofrunoff, fluvialsediment,andnutrientdischargesonthecoralreefsofPuerto Rico:JournalofCoastalResearch,v.251,p.189–208,DOI: 10.2112/ 07-0920.1
Lesser,M.P.,2021,Eutrophicationoncoralreefs:Whatistheevidence forphaseshifts,nutrientlimitation,andcoralbleaching:Bioscience,v. 71,p.1216–1233,DOI: 10.1093/biosci/biab101
Loeblich,A.R.,andTappan,H.,1987,ForaminiferalGeneraandTheir Classification:VanNostrandReinholdCompany,NewYork,2013p., DOI: 10.1007/978-1-4899-5760-3
Martínez-Colón,M.,Alegría,H.,Kubra-Gul,H.,andKurt-Karakus,P., 2021,Bioaccumulationandbiomagnificationofpotentialtoxicelements(PTEs):An Avicenniagerminans-Ucarapax trophictransfer storyfromJobosBay,PuertoRico:EcologicalIndicators,v.121,DOI: 10.1016/j.ecolind.2020.107038
Mathes,G.H.,Steinbauer,M.J.,andCotton,L.,2022,BenthicforaminiferaasbioindicatorsforassessingreefconditioninKane’oheBay, O’ahu,Hawai ’i:Paci ficConservationBiology,v.29,p.238–245, DOI: 10.1071/PC21027
McClanahan,T.R.,2022,Coralresponsestoclimatechangeexposure:EnvironmentalResearchLetters,v.17,DOI: 10.1088/1748-9326/ac7478
Moberg,F.,andFolke,C.,1999,Ecologicalgoodsandservicesofcoral reefecosystems:EcologicalEconomics,v.29,p.215–233,DOI: 10. 1016/S0921-8009(99)00009-9
Murray,J.W.,1973,DistributionandEcologyofLivingBenthicForaminiferids:Crane,Russak,NewYork,274p.
Murray,J.W.,2006,EcologyandApplicationsofBenthicForaminifera: CambridgeUniversityPress,Cambridge,426p.
Oliver,L.M.,Fisher,W.S.,Dittmar,J.,Hallock,P.,Campbell,J.,Quarles, R.L.,Harris,P.,andLoBue,C.,2014,Contrastingresponsesofcoral reeffaunaandforaminiferalassemblagestohumaninfluenceinLa Parguera,PuertoRico:MarineEnvironmentalResearch,v.99,p.95–105.
Prazeres,M.,Martínez-Colón,M.,andHallock,P.,2020,Foraminiferaas bioindicatorsofwaterquality:TheFoRAMIndexrevisited:EnvironmentalPollution,v.257,DOI: 10.1016/j.envpol.2019.113612
Ramos-Scharrón,C.E.,Torres-Pulliza,D.,andHernández-Delgado,E.A., 2015,Watershed-andislandwide-scalelandcoverchangesinPuerto Rico(1930s–2004)andtheirpotentialeffectsoncoralreefecosystems: ScienceofTheTotalEnvironment,v.506–507,p.241–251,DOI: 10. 1016/j.scitotenv.2014.11.016
Ramos-Scharrón,C.E.,2021,Impactsofoff-roadvehicletracksonrunoff, erosionandsedimentdelivery – Acombined fieldandmodeling approach:EnvironmentalModelling&Software,v.136,p.104957, DOI: 10.1016/j.envsoft.2020.104957
RosaMarín,A.,2022,Environmentalassessmentofcoralreefecosystems atJobosBay,PuertoRico:FloridaAgriculturalandMechanical UniversityProQuestDissertationsPublishing,accessedat https://www. proquest.com/dissertations-theses/environmental-assessment-coral-reefecosystems-at/docview/2778394123/se-2
Ross,B.J.,andHallock,P.,2019,Survivalandrecoveryoftheforaminifer Amphisteginagibbosa andassociateddiatomendosymbiontsfollowing upto20monthsinaphoticconditions:MarineMicropaleontology,v. 149,p.35–43.
Schnetger,B.,andLehners,C.,2014,Determinationofnitrateplusnitrite insmallvolumemarinewatersamplesusingvanadium(III)chloride asareductionagent:MarineChemistry,v.160,p.91–98.
Schönfeld,J.,Alve,E.,Geslin,E.,Jorissen,F.,Korsun,S.,andSpezzaferri, S.,2012,TheFOBIMO(FOraminiferalBIo-MOnitoring)Initiative Towardsastandardisedprotocolforsoft-bottombenthicforaminiferal monitoringstudies:MarineMicropaleontology,v.94–95,p.1–13,DOI: 10.1016/j.marmicro.2012.06.001
Shlesinger,T.,andvanWoesik,R.,2023,Oceanicdifferencesincoralbleachingresponsestomarineheatwaves:ScienceoftheTotalEnvironment,v.871,DOI: 10.1016/j.scitotenv.2023.162113
Strickland,J.D.H.,andParsons,T.R.,1972,APracticalHandbookof SeawaterAnalysis:FisheriesResearchBoardofCanadaBulletin, Ottawa,2ndEdition,310p.
Takesue,R.K.,Sherman,C.,Ramirez,N.I.,Reyes,A.O.,Cheriton, O.M.,Ríos,R.V.,andStorlazzi,C.D.,2021,Land-basedsediment sourcesandtransporttosouthwestPuertoRicocoralreefsafterHurricaneMaria,May2017toJune2018:Estuarine,CoastalandShelfScience,v.259,DOI: 10.1016/j.ecss.2021.107476
Triffleman,N.J.,Hallock,P.,Hine,A.C.,andPeebles,M.W.,1991,ForaminiferaofSerranillaBank:DistributionofforaminiferaltestsinsedimentsofSerranillaBank,NicaraguanRise,SouthwesternCaribbean: JournalofForaminiferalResearch,v.21,p.39–47.
VanderLinde,D.W.,1998,Protocolfordeterminationoftotalsuspended matterinoceansandcoastalzones:JRCTechnicalNoteI,v.98,p.182. Webler,T.,andJakubowski,K.,2016,Mitigatingdamagingbehaviorsof snorkelerstocoralreefsinPuertoRicothroughapre-tripmedia-based intervention:BiologicalConservation,v.197,p.223–228,DOI: 10. 1016/j.biocon.2016.03.012
Wentworth,C.K.,1922,Ascaleofgradeandclasstermsforclasticsediments:JournalofGeology,v.30,p.377–392.
Wetmore,K.L.,1987,Correlationsbetweenteststrength,morphologyand habitatinsomebenthicForaminiferfromthecoastofWashington: JournalofForaminiferalResearch,v.17,p.1–13.
Woodhead,A.J.,2019,CoralreefecosystemservicesintheAnthropocene:FunctionalEcology,v.33,p.1023 –1034.DOI: 10.1111/13652435.13331
Wooldridge,S.A.,andDone,T.J.,2009,Improvedwaterqualitycanameliorateeffectsofclimatechangeoncorals:EcologicalApplications,v. 19,p.1492–1499,DOI: 10.1890/08-0963.1
Received31July2023
Accepted29October2023
APPENDIX A.Dominantphi-sizeofsedimentsfromallsitesby fieldseason.Phi-sizes: 1 5 gravel,0 5 verycoarsesand,1 5 coarsesand,2 5 mediumsand,3 5 finesand,and4 5 very finesand.Asterisks(*)indicatenodata.
ReefStation Mar-18Sept-18Dec-18Mar-19Aug-19Dec-19
CayoMorrilloF-M1/2010* 1*
F-M41***1*
B-M5/6211011
B-M7012001
B-M8*40122
CayoPájarosF-P1/2000*0* F-P4*42*2* B-P5/6121413
B-P7444444
B-P8*44444
CayoCaribeF-C1/2000*0* F-C4000***
F-C5/64*****
B-C9244444
B-C10*43*44
B-C11424444
B-C12444444
APPENDIX B.Listoftaxapresentinsuperficialsedimentsamplesfrom coralreefsatJobosBay,PuertoRico.
Taxaname
Af
finetrina sp. Łuczkowska,1972
Ammoniabeccarii (Linnaeus,1758)
Amphisteginagibbosa d'Orbigny,1839
Archaiasangulatus (Fichtel&Moll,1798)
Articulinamexicana Cushman,1922
Articulinapacifica Cushman,1944
Asterigerinacarinata d'Orbigny,1839
Bolivina sp.d'Orbigny,1839
Borelispulchra (d'Orbigny,1839)
Cornuspiraplanorbis Schultze,1854
Cornuspira sp.Schultze,1854
Cycloforinacollumnosa (Cushman,1922)
Cycloforinacf.columnosa (Cushman,1922)
Cycloforinagranulocoastata (Germeraad,1946)
Dentostominabermudiana Carman,1933
Discorbismira Cushman,1922
Elphidium sp.Montfort,1808
Hauerina sp.d'Orbigny,1839
Heterosteginadepressa d'Orbigny,1826
Hoeglundina sp.Brotzen,1948
Laevipeneroplisproteus (d'Orbigny,1839)
Miliamminafusca (Brady,1870)
Miliolinellacf.oblonga (Montagu,1803)
Miliolinellacircularis (Bornemann,1855)
Miliolinella sp.Wiesner,1931
Nonion sp.Montfort,1808
Peneroplispertusus (ForsskålinNiebuhr,1775)
Planorbulina sp.d'Orbigny,1826
Planurbulinaacervalis Brady,1884
Pseudohauerinaoccidentalis (Cushman,1946)
Pseudoaurila sp.Ishizaki&Kato,1976
Pyrgo sp.Defrance,1824
Pyrgocf.denticulata (Brady,1884)
Pyrgocf.murrhina (Schwager,1866)
Pyrgodenticulata (Brady,1884)
APPENDIX B.Continued. Taxaname
Pyrgoelongata (d'Orbigny,1826)
Pyrgomurrhina (Schwager,1866)
Quinqueloculina sp.d'Orbigny,1826
Quinqueloculinaagglutinans d'Orbigny,1839
Quinqueloculinabicarinata d'OrbignyinTerquem,1878
Quinqueloculinabicostata d'Orbigny,1839
Quinqueloculinabidentata d'Orbigny,1839
Quinqueloculinabosciana d'Orbigny,1839
Quinqueloculinabradyana Cushman,1917
Quinqueloculinacandeiana d'Orbigny,1839
Quinqueloculinafrigida Parker,1952
Quinqueloculinalaevigata d'Orbigny,1839
Quinqueloculinalamarckiana d'Orbigny,1839
Quinqueloculinaparkeri (Brady,1881)
Quinqueloculinapoeyana d'Orbigny,1839
Quinqueloculinapolygona d'Orbigny,1839
Quinqueloculinapseudoreticulata Parr,1941
Quinqueloculinaseminulum (Linnaeus,1758)
Quinqueloculinasubpoeyana Cushman,1922
Quinqueloculinatenagos Parker,1962
Quinqueloculinatransversestriata (Brady,1881)
Quinqueloculinatricarinata d'Orbigny,1839
Quinqueloculinacf.bicarinata d'OrbignyinTerquem,1878
Quinqueloculinacf.bicostata d'Orbigny,1839
Quinqueloculinacf.brayana Cushman,1917
Quinqueloculinacf.crassa d'Orbigny,1850
Quinqueloculinacf.intricata Terquem,1878
Quinqueloculinacf.lamarckiana d'Orbigny,1839
Pseudotriloculinacf.inneiana (d'Orbigny,1839)
Quinqueloculinacf.multimarginata Said,1949
Quinqueloculinacf.parkeri (Brady,1881)
Quinqueloculinacf.philippinensisCushman,1921
Quinqueloculinacf.poeyana d'Orbigny,1839
Quinqueloculinacf.polygona d'Orbigny,1839
Quinqueloculinacf.seminulum (Linnaeus,1758)
Quinqueloculinacf.tenagos Parker,1962
Quinqueloculinacf.tricarinata d'Orbigny,1839
Quinqueloculinacf.venusta Karrer,1868
Quinqueloculinacf.garrettii Andersen,1961
Quinqueloculinacf.akneriana d'Orbigny,1846
Reussella sp.Galloway,1933
Rosalinabradyi (Cushman,1915)
Rosalina floridana (Cushman,1922)
Rosalinapariensis d'OrbignyinDeshayes,1832 †
Rotorbinellarosea (d'OrbignyinGuérin-Méneville,1832)
Schlumbergerinaoccidentalis Cushman,1929
Sigmoihauerina sp.Zheng,1979
Siphonina sp.Reuss,1850
Siphoninapulchra Cushman,1919
Soritesmarginalis (Lamarck,1816)
Spirolocammina sp.Earland,1934
Spiroloculina sp.d'Orbigny,1826
Spiroloculinaantillarum d'Orbigny,1839
Spiroloculinaarenata Cushman,1921
Spiroloculinacomumnis Cushman&Todd,1944
Spiroloculinacorrugata Cushman&Todd,1944
Spiroloculinadepressa d'Orbigny,1826
Spiroloculinaplanulata (Lamarck,1804)
Spiroculinasoldanii Fornasini,1886
Sigmoliscf.arenata (Cushman,1921)
Spiroloculinacf.caduca Cushman,1992
Spiroloculinac.f.planulata (Lamarck,1804)
Textulariaagglutinans d'Orbigny,1839
Triloculinacf.oblonga (Montagu,1803)
Triloculinacf.trigonula (Lamarck,1804)
Triloculina sp.d'Orbigny,1826
APPENDIX B.Continued. Taxaname
Triloculinaangusteoralis (Wiesner,1923)
Triloculina fiterrei Acosta,1940
Triloculinalinneiana d'Orbigny,1839
Triloculinaoblonga (Montagu,1803)
Triloculinaplanciana d'Orbigny,1839
Triloculinarotunda d'OrbignyinSchlumberger,1893
Triloculinatricarinata d'OrbignyinDeshayes,1832
Triloculinatrigonula (Lamarck,1804)
Wiesnerellaauriculata (Egger,1893)

