Guidance on The Management of Mpox Infections for Sexual Health Services
Version: V3
Ratified by:

Infection Prevention and Control Group
Date ratified: 23/03/2026
Job Title of author:
Reviewed by Committee or Expert Group
Related procedural documents
Sexual Health System Lead
Infection Prevention and Control Lead
Infection Prevention and Control Group
IPPOL21 Standard Precautions for Infection Prevention & Control including TBPs and Isolation Precautions
MMSOP32 Ordering, Storage, Transport and Disposal of Vaccines & Medicines Requiring Refrigerated Storage SOP (Cold Chain)
IPPOL03 Infection Prevention and Control Hand hygiene policy
Review date: 23/03/2029


It is the responsibility of users to ensure that you are using the most up to date document template – i.e. obtained via the intranet.
In developing/reviewing these guidelines Provide Community has had regard to the principles of the NHS Constitution.
Version Control Sheet
Version Date
V1 31st May 2022

V2 1st June 2022
Author
Chris Quinn: Clinical Nurse Specialist HIV/AIDS / Acting Service Lead for Essex& Thurrock Sexual Health
Chris Quinn Clinical Nurse Specialist HIV/AIDS / Acting Service Lead for Essex& Thurrock Sexual Health
V3 16th August 2024 Essex and Suffolk Sexual Healthservices
Status
Comment
New Guidance
V1 21st August 2024 Essex and Suffolk Sexual Healthservices
V2 2nd December 2024 Essex and Suffolk Sexual Healthservices
V3Updated 8th December 2025 Essex and Suffolk Sexual Healthservices
Updated comments and flowcharts
Updated guidance for management of Clade 1 Mpox in line with UKHSA notification for Outbreak of Clade 1 Mpox
Updated comments and guidance – Dr Malaki
Updated comments and guidanceNHSE and IPC
Updated as Clade I mpox virus (MPXV) and Clade 11 are no longer classified as a High Consequence Infectious Disease(HCID) in the UK, meaning all mpox is now managed as a non-HCIDinUK healthcare settings.
26/2/2026

Updatedtoadd appendix 1 on PPE, contacts and consent to share results withGP.


1. Background Information on Mpox virus (MPXV)
• There are two known clades of Mpox: Clade I and Clade II
• Transmission of Mpox to humans can be due to zoonotic transmission or personto-person spread
• Clade I Mpox was associated with zoonotic transmission and known to circulate in 5 African countries; Cameroon, Central African Republic, the DRC, Gabon and the Republic of Congo
• Clade I mpox virus (MPXV) and Clade 11 are no longer classified as a High Consequence Infectious Disease (HCID) in the UK, meaning all mpox is now managed as a non-HCID in UK healthcare settings, reflecting lower fatality rates and milder illness for the types seen in recent years, though it remains a significant public health concern
• Infection with Clade I Mpox has been reported to cause more severe mpox disease with a higher case fatality rate
• Clade I mpox virus (MPXV) has historically only been reported in five countries in Central Africa. There is now increasing transmission of clade I mpox in the Democratic Republic of Congo (DRC), and cases are also being reported from other surrounding countries in Central and East Africa
• Between 25 July and 5 August 2024, confirmed Clade I MPXV cases have been reported from Burundi, Kenya, Rwanda and Uganda for the first time, which has expanded the geographical footprint. Since August 2024, clade I MPXV has also been reported from multiple countries outside of the African region that had not previously reported it, including cases in the UK. This is mostly caused by clade Ib MPXV, and is spread mostly through close physical contact (including both sexual and non-sexual contact).
• Clade II cases have been reported from Benin, Cameroon, Cote D’Ivoire, Ghana, Liberia, Nigeria and South Africa in 2024
• There is evidence of sustained sexual transmission of clade I MPXV in the DRC.
• Sexual transmission should not be used to infer whether an mpox case is likely to be clade I or II
2. Mpox: Case Definition
This guidance has been developed to support Provide Sexual Health Services in the event of a suspected case of Mpox being identified within the sexual health clinic
When assessing patients for mpox, always take a travel history. Be alert to the possibility Mpox (Clade I) in all patients with suspected mpox including cases in the UK and where linked to a travel history to the Democratic Republic of the Congo or other specified countries in the African region https://www.gov.uk/guidance/clade-i-mpox-affectedcountries
For all suspected mpox cases
For patients being assessed for suspected mpox, infection prevention and control (IPC) measures should be undertaken per the NHS national infection prevention and control manual for England.
Clinicians should be aware that mpox is a notifiable disease

Clinicians treating patients with suspected mpox should discuss the case with local infection specialists. Infection specialists may wish to discuss possible mpox cases with the UKHSA Imported Fever Service (IFS) on 0844 778 8990 for clinical advice, for example in patients who are severely immunocompromised or pregnant, paediatric patients, or patients from a high risk setting such as shared accommodation.
Test for MPXV, the causative agent of mpox, using an appropriate testing pathway. See also Mpox diagnostic testing guidance
Undertake testing for other diagnoses if clinically appropriate and if not done already.
Possible Case
A possible case is defined as anyone who fits one or more of the following criteria:
• A febrile prodrome (fever >38 degrees C, chills, headache, exhaustion, muscle ache, joint pain, backache and swollen lymph nodes) compatible with mpox infection, where there is a known prior contact with a confirmed case in the 21 days before symptom onset
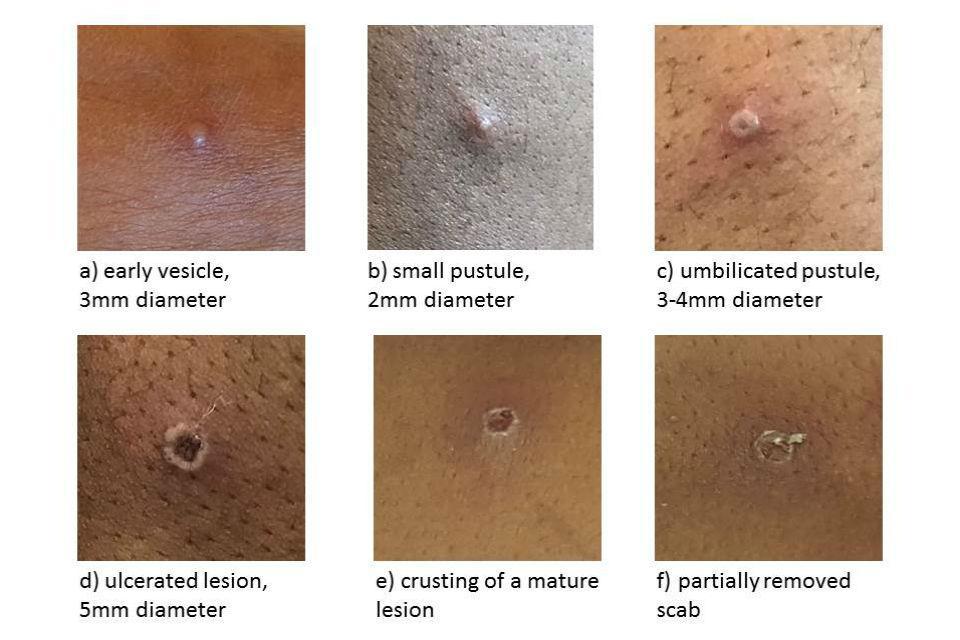
• An illness, where the clinician had a suspicion of mpox, such as unexplained lesions, including but not limited to
➢ Genital, ano-genital or oral lesion(s)- e.g. ulcers, nodules
➢ Proctitis- e.g. anorectal pain, bleeding
Probable Case
A probable case is defined as anyone with an unexplained rash or lesion(s) on any part of the body (including genital/perianal, oral) or protitis (anorectal pain, bleeding) who:
• Has an epidemiological case to a confirmed, probable or highly probable case of mpox in the 21 days before symptom onset OR
• Identifies as gay, bisexual or other man who has sex with men OR
• Has had one or more sexual partners in the 21 days before symptom onset
Actions for a possible or probable case:
• Have a low threshold for testing for mpox in patients with clinically compatible presentations with a travel history irrespective of sexual history, using designated testing pathways.
• Undertake additional contemporaneous tests to rule out alternative diagnoses if clinically appropriate and if not already done
• Be alert to the possibility of clade I mpox in all patients with suspected mpox if there is a link to the specified countries in the African region (as listed above). Patients with a travel or exposure history indicating possible Clade 1 mpox should be discussed with the Rare and Imported Pathogens Laboratory (RIPL) on 01980 612348 (available 9am to 5pm, Monday to Friday) and the clinical team as soon as possible via the 24/7 Imported Fever Service helpline (0844 778 8990).
• Isolate patients meeting the following criteria as a high consequence infectious disease and contact the Imported Fever Service to discuss urgent testing and typing:
➢ Confirmed or clinically suspected mpox cases but clade not yet known
➢ travel history to the DRC or specified countries where there may be
a risk of clade I exposure including the UK https://www.gov.uk/guidance/clade-i-mpox-affected-countries
➢ link to a suspected case from those countries (listed above), within 21 days of symptom onset and/or there is an epidemiological link to a case of Clade I mpox within 21 days of symptom onset.
Actions required by Clinicians:
Isolate patients meeting the following criteria- (All suspected Mpox cases)
Contact the Imported Fever Service (IFS) (0844 778 8990): - to discuss urgent testing and typing of:-
• Confirmed or clinically suspected mpox cases but clade not yet known and there is a travel history to the DRC or specified countries where there may be a risk of clade I exposure, or a link to a suspected case from those countries (listed above or on updated UKHSA list) https://www.gov.uk/guidance/clade-impox-affected-countries, within 21 days of symptom onset and/or there is an epidemiological link to a case of Clade I mpox within 21 days of symptom onset
• Discuss any patient with suspected mpox and severe or disseminated disease with Imported Fever Service (IFS) (0844 778 8990), even if no travel history is identified.
• Notify the local Health Protection teams on suspicion of Clade I mpox Cases where the clade is unknown, but who have a travel or contact history as above, should be discussed with the Imported Fever Service (0844 778 8990) as soon as possible to ensure appropriate testing and escalation.
Operational case definition of Clade I and Clade II
Clade I - Clade II -
Confirmed mpox case where clade I has been confirmed
Confirmed or clinically suspected mpox case but clade not yet known

TravelhistorytotheDRCorspecifiedcountries where there may be a risk of clade I exposure, or a link to a suspected case from those countries (listed above), within 21 days of symptom onset and/or there is an epidemiological link to a case of Clade I mpox within 21 days of symptom onset
GiventherapidspreadofCladeIintheAfrican region, check the UKHSA mpox pages regularly for any updates to the countries included
Confirmed as Clade II MPXV
Confirmed or clinically suspected mpox but clade not known, and all of the following conditions apply: -
There is no history of travel to the DRC or specified surrounding countries (as above) within 21 days of symptom onset
There is no link to a suspected case from the DRC or specified surrounding countries within 21 days of symptom onset