

Sustainability Meets Patient Adherence
Inside the packaging strategy behind Green Goo’s plant-based first aid revolution | 20
Distributing Pharma Products Along the Colder Chain | 12 How Babsy Reinvented the Children’s Toothbrush | 28
Global Labeling Challenges: Insights from Kite Pharma | 30
Healthcare Packaging’s Inaugural Annual Outlook Report | 34
Intelligent Data Capture for a Modern Healthcare Supply Chain | 38


SPECIAL SECTION: Leaders in Healthcare Packaging | 44










April 21 - 23, 2026
Javits Center
New York, NY
Booth #2040



MULTIVAC Health Packaging Automated Packaging Solutions for
Medical & Pharmaceutical
Products

MULTIVAC is your trusted automated packaging partner to develop custom integrated solutions that meet the highest requirements for clean-room compatibility, process reliability, and product traceability. Learn how our solutions enhance production with maximum reliability while providing the highest level of product protection and quality.
Contact the MULTIVAC Health Packaging specialists to get started today!


MATERIAL
DEVELOPMENTS
16 Great Earth Expands Use of Fiber-based Closures to Entire Line
Swedish nutraceutical brand rolls out paper screw caps across all supplement jars.
SUSTAINABILITY
20 Green Goo Brings Plant-based Healing to the First Aid Aisle
Inside the sustainable processing and packaging strategy behind Green Goo’s plant-based first aid revolution.
AUTOMATION
24 Fernway Cannabis Automates Its Labeling
Faced with fragmented packaging regulations and labeling inefficiencies, Fernway turned to Paxiom for a solution.
PACKAGE DESIGN
28 Building Better Brushers: How Babsy Reinvented the Children’s Toothbrush Through Design and Play
The packaging is made to look like a creative kit rather than a sterile healthcare product.
REGULATORY
30 Global Labeling Challenges: Insights from Kite Pharma
For cell therapy manufacturers, such as Kite Pharma, where every product is made for a single patient, labeling becomes a critical safety and traceability tool.
ANNUAL OUTLOOK REPORT
34 What’s in Store for Pharmaceutical Manufacturers in 2026 and Beyond?
Read results from our reader survey in this inaugural Annual Outlook Report.




TECHNOLOGY







38 Intelligent Data Capture: Paving the Path to a Modern Healthcare Supply Chain
This focus on standardization is helping to reduce integration complexity—an ongoing challenge for many healthcare organizations.
PROMINENT PEOPLE IN PACKAGING
42 Engineering for Integrity: How Evergen’s Ashim Moona Builds Packaging Systems that Protect Patients
Read the two aspects of sustainability, the importance of learning the standards, and how “burnout” can be a good thing.
BUSINESS INTELLIGENCE
88 Pharmaceutical Packaging’s Next Phase: Connectivity
Future-ready pharma packaging must integrate connectivity, automation, and material strategy into a coordinated approach.
COLUMNS & DEPARTMENTS
Courtesy: Green Goo










LEAK TESTING SOLUTIONS FOR PHARMA AND CCIT

www.healthcarepackaging.com
Content
EDITOR-IN-CHIEF Elisabeth Cuneo ecuneo@pmmimediagroup.com
CONTRIBUTING EDITOR Joseph Derr
DIRECTOR OF CONTENT Kim Overstreet
ART DIRECTOR Norman Smith
CREATIVE DIRECTOR David Bacho

Advertising
VICE PRESIDENT, SALES Reggie Lawrence rlawrence@pmmimediagroup.com
DIRECTOR, CLIENT SUCCESS & DEVELOPMENT Courtney Nichols cnichols@pmmimediagroup.com
SENIOR MANAGER, PRINT OPERATIONS Lara Krieger lkrieger@pmmimediagroup.com
FINANCIAL SERVICES MANAGER Janet Fabiano jfabiano@pmmimediagroup.com




PMMI Media Group
PRESIDENT David Newcorn
VICE PRESIDENT, DIGITAL Elizabeth Kachoris
SENIOR DIRECTOR, DIGITAL MEDIA Jen Krepelka
DIRECTOR OF MARKETING Amber Miller
DIRECTOR, AD TECH AND SEARCH Joan Jacinto
FOUNDING PARTNER AND EXECUTIVE VICE PRESIDENT, INDUSTRY OUTREACH, PMMI Joseph Angel
Questions about your subscription or wish to renew? Contact circulation@pmmimediagroup.com
PMMI Media Group 500 W. Madison, Suite 1000 Chicago, IL 60661 www.pmmimediagroup.com
PMMI The Association for Packaging and Processing Technologies 12930 Worldgate Dr., Suite 200, Herndon VA, 20170 Phone: 571.612.3200 | Fax: 703.243.8556 | www.pmmi.org
PLEASE RECYCLE THIS MAGAZINE Remove inserts or samples before recycling.
Healthcare Packaging® (ISSN # 21543666) is a registered trademark of PMMI, The Association for Packaging and Processing Technologies. Healthcare Packaging® is published twice annually by PMMI with its publishing office, PMMI Media Group, located at 500 W. Madison, Suite 1000, Chicago, IL 60661; 312.222.1010. Copyright 2026 by PMMI. All rights reserved. Materials in this publication must not be reproduced in any form without written permission of the publisher. Applications for a free subscription may be made online at hcpgo.to/9999. POSTMASTER; Send address changes to Healthcare Packaging®, 500 W. Madison, Suite 1000, Chicago, IL 60661. PRINTED IN USA by Quad. Volume 21, Number 1 The opinions expressed in articles are those of the authors and not necessarily those of PMMI. Comments, questions and letters to the editor are welcome and can be sent to: editors@healthcarepackaging.com. Mailing List: We make a portion of our mailing list available to reputable firms. If you would prefer that we don’t include your name, please write us at the Chicago, IL address.




















The Human Side of Healthcare Packaging: How Patient Safety and Centricity Reign Supreme

I have had the privilege of attending a handful of industry events already this year. I had the pleasure to sit in on sessions, meet industry leaders, and see the new materials and machinery progressing the pharmaceutical and medical device industries along.
It’s an exciting time with the emergence of new digital tools, sustainability breakthroughs pushing the limits, and new device features that promise a better user experience. But what always blows me away is not necessarily the technology—it’s the human element.
If I were to sum up a day at just about any trade show, seminar, or conference, I’d walk away with the sense that the patient is the “North Star” for these companies. For example, at Pharmapack this past January, I interviewed a woman from a global design and contract manufacturing company that expressed the best way to design a device is to truly understand the lived experience of the user. She did that through
remote diary studies using a digital platform that allowed participants to record videos and answer prompts in their own homes.
Then at PACK EXPO East, I spoke with a gentleman from a pharmaceutical company about the many considerations he must make when selecting equipment and materials, but ultimately, he said patient safety remains the core focus.
And within the pages of this very issue, you can read an interview with a medical device packaging engineer. From him I learned that patient care is his biggest “why.”
Behind every new technology, material, or software is the drive to improve the patient experience, and of course, ensure the utmost safety. I’m realizing that’s why I love this industry: it is the point where packaging and patient care come together.
Liz Cuneo is the Editor-in-Chief of Healthcare Packaging. She may be reached at ecuneo@pmmimediagroup.com.


















Q“I am extremely pleased with our commitment to invest more than $30 billion in the U.S. over the next five years ... it’s still a leading market in the world in terms of the launch of new drugs and vaccines.”
—DAME WALMSLEY, GSK’S CEO, SAID IN AN INTERVIEW ABOUT THE COMPANY’S RECENT INVESTMENT AND AMERICAN EXPANSION.
$38.38
BILLION—THE WORTH OF THE ELECTRONIC SKIN PATCH market size by 2032. These patches, which are attached to the skin using adhesive, serve as a platform for various types of sensors, primarily for medical monitoring purposes.
Source: Polaris Market Research
“
Clean design is about more than aesthetics. It’s about making sure your packaging protects the product safely through the supply chain—and that it has a life after the consumer uses it. That’s responsible design.”
—CHRIS LACORATA, FOUNDER OF GRAASI, A WELLNESS BEVERAGE BRAND, IN AN INTERVIEW ABOUT THE IMPORTANCE OF RESPONSIBLE MANUFACTURING AND PACKAGING.
“It’s clear
that specialty pharma
continues to
grow and now represents more than half of total pharmaceutical spending in the U.S., reinforcing its central role in our healthcare system.”
—PERRY FRI, PRESIDENT, HDA RESEARCH FOUNDATION, IN AN INTERVIEW ABOUT HDA FOUNDATION’S LATEST BENCHMARKING STUDY ON SPECIALTY PHARMACEUTICAL DISTRIBUTION.
59%
THE PERCENTAGE OF AMERICANS optimistic that single-use plastic will be largely phased out in the next 10 years.
Source: SmartLifeCo.
500%
THE RISE IN SPENDING on GLP-1s from 2018 to 2023.
Source: The American Medical Association
$196
BILLION—THE EXPECTED SIZE of the global medical device contract manufacturing market size by 2032, growing from reported roughly $79 billion in 2024.
Source: DevelInsight

Opportunities in the U.S. IV Bags Market
ResearchAndMarkets.com released a report titled, “United States IV Bags Market Report by Material Type, Capacity, Chamber Type, States and Company Analysis, 20252033.” According to this research, the market is expected to grow substantially, from $595.98 million in 2024 to $1.19 billion in 2033. Major growth drivers are a rise in healthcare requirements, technological improvements in IV bags, and the growing need for intravenous treatments. Another growth impetus is the shift from standard PVC-based IV bags to safer, more eco-friendly materials such as polyethylene and polypropylene. Phthalates and plasticizers migrating out of PVC products have caused regulatory authorities to tighten scrutiny, while hospitals increasingly prefer non-PVC alternatives, creating important opportunities for producers of high-performance polymer-based IV bags.
Community poll:
We asked, you answered: Which of the following factors most influence your packaging decisions?
59% Supply chain resilience
27% Regulatory compliance
8% Cost efficiency
5% Sustainability Answer the latest poll online at the HCP homepage and look for reader polls in our e-newsletter.



Demand for Advanced Drug Delivery Systems to Propel Pharmaceutical Packaging Market Growth
INCREASING AWARENESS of medication adherence and patient safety is accelerating adoption of packaging solutions with superior protection and usability.
The global pharmaceutical packaging market is projected to grow from $128.7 billion in 2025 to $299.1 billion by 2035, reflecting an increase of $169.6 billion, according to a new forecast by Future Market Insights. This robust growth underscores the escalating demand for advanced drug delivery systems, biologics and biosimilars adoption, and enhanced patient safety across prescription, overthe-counter, and specialty pharmaceutical products. By drug form, solid dosage packaging holds a dominant position, representing 45.2% of the market in 2025.
Lilly Partners with NVIDIA to Build the Industry’s Most Powerful AI Supercomputer

Eli Lilly and Company announced it is building the most powerful supercomputer owned and operated by a pharmaceutical company, in collaboration with NVIDIA. The supercomputer will power an “AI factory”—a specialized computing infrastructure that manages the entire AI lifecycle from data ingestion and training to finetuning and high-volume inference. Put into simple terms, the AI supercomputer ultimately will make discovering new drugs simpler and faster.

Global Medical Device Contract Manufacturing Market on the Rise
RISING DEMAND for advanced, cost-efficient medical technologies and the increasing shift by OEMs toward outsourcing is causing the market to take off. DelveInsight reports that the global medical device contract manufacturing market will grow to $190 billion by 2032. Expanding chronic disease burdens, rapid innovation in minimally invasive and digital health devices, and the need for scalable production are further accelerating market momentum. Additionally, regulatory complexities and the push for faster time-to-market are prompting manufacturers to rely more heavily on specialized contract partners.
CALENDAR OF EVENTS
Interphex (April 21-23) New York, NY the[PACK]out (May 12-14) Oxon Hill, MD
CPHI Americas (June 2-4) Philadelphia, PA
“The challenge in drug discovery is scale: The number of possible chemical compounds is astronomical, and evaluating each one as a drug candidate requires simulating complex molecular interactions—an extremely resource-intensive task,” says Thomas Fuchs, SVP and Chief AI Officer, Eli Lilly and Company. “Historically, limited computational power meant scientists could only process one molecular design or simulation at a time, forcing difficult trade-offs and slowing progress. With the supercomputer, Lilly has the potential to run numerous molecular design processes simultaneously, rapidly exploring chemical space that was previously inaccessible.”

Packaging Recycling Summit (June 15-17) Rosemont, IL
PACK EXPO International (Oct 18-21) Chicago, IL
MD&M Midwest (Oct 28-29) Minneapolis, MN
GS1 Healthcare Global Conference (Nov 3-5) Boston, MA





































































































Balancing Sustainability and Sterility: A Risk-Based Path Forward for Healthcare Packaging
Insights from the SPMC (Sterilization Packaging Manufacturers Council) on a risk-based sustainability assessment model grounded in regulatory compliance and patient safety.
From the Sterilization Packaging Manufacturers Council

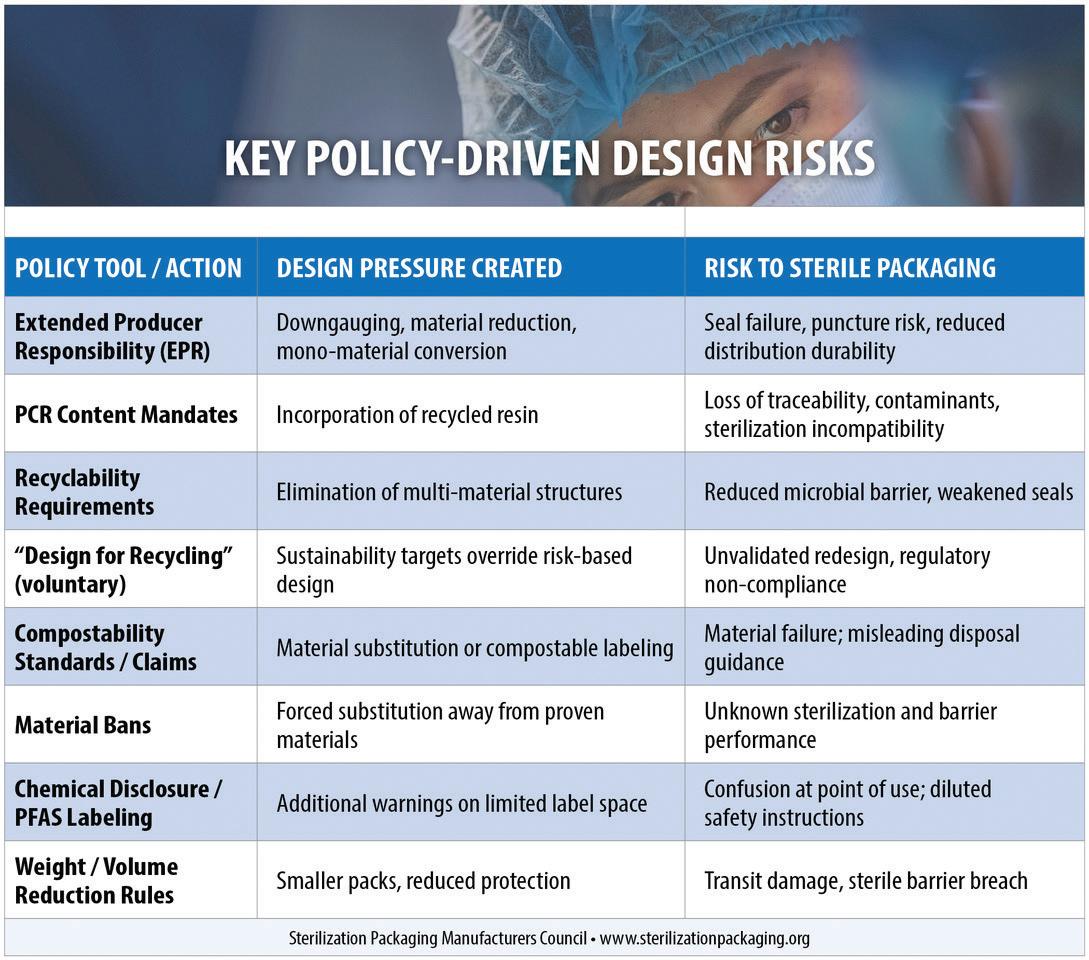
Sustainable packaging policy is reshaping packaging decisions across the United States, and despite some exemptions in certain cases, healthcare packaging is increasingly affected. Extended Producer Responsibility (EPR) programs, recycled content mandates, recyclability requirements, compostability standards, and chemical disclosure laws are designed to reduce waste and advance circularity. Yet for sterile medical device packaging, these policies introduce design pressures that can directly conflict with regulatory and performance requirements ensuring safety remains of paramount importance.
The Sterilization Packaging Manufacturers Council (SPMC) has examined these conflicts in depth and is working on a series of white papers addressing this topic, starting with Sustainability of Medical Device Packaging. The findings are clear: Sterile packaging is a safety-critical system governed by FDA regulations and international standards, and sustainability policies that fail to account for these requirements can introduce unacceptable risk.
Medical device packaging must meet stringent obligations under FDA Quality System Regulations and consensus standards, such as ISO 11607 for package integrity and ISO 10993 for biocompatibility. Policies requiring post-consumer recycled (PCR) content or universal recyclability are incompatible with these standards today. Mechanically recycled materials lack traceability, introduce contamination risk, and exhibit inconsistent performance that cannot be reliably validated for sterilization, seal integrity, or long-term barrier protection. In sterile packaging, even minor material variability can lead to seal failures, microbial ingress, and Class I recalls.
EPR laws illustrate how these pressures manifest in practice. Seven states—California, Colorado, Maine, Maryland, Minnesota, Oregon, and Washington—have enacted packaging EPR frameworks. While several states recognize the need to exclude sterile medical packaging, others leave ambiguity, particularly for secondary and tertiary packaging. Even where exemptions exist, EPR fee structures, recyclability metrics,
and corporate sustainability commitments often drive voluntary redesigns that reduce material thickness, favor mono-material structures, or encourage recycled content—sometimes without sufficient risk evaluation.
To address this challenge, SPMC advocates for a risk-based sustainability assessment model grounded in regulatory compliance and patient safety. This model begins by asking a fundamental question: Does a proposed sustainability change preserve sterility assurance and regulatory compliance across the entire lifecycle of the device? If not, it should not proceed.
Under this framework, sustainability initiatives must be evaluated against four core criteria: sterilization compatibility, microbial barrier performance, physical protection during distribution, and biocompatibility. Changes to materials, structures, or labeling should trigger formal validation and risk management processes consistent with FDA and ISO expectations.
systems, recognize where these policies can create failures and advocate for certain technologies and solutions to offer a safer pathway to circularity in the future.
Importantly, SPMC also recognizes the need for a strong advocate and educator on sterile medical packaging. Our industry needs to help legislators, regulators, and key stakeholders understand and protect the value of sterile packaging
Sustainable progress in healthcare packaging is both necessary and achievable—but only if policy, design, and sustainability goals are aligned with the realities of sterile packaging performance. As SPMC’s work underscores, patient safety must remain the non-negotiable foundation upon which all sustainability efforts are built.
FLEXIBILITY
A single
Columbia palletizer
can handle all of these products and more with ease.


Packaging & Distributing Pharma Products Along the “Colder” Chain
Logistical management of the cold chain has constantly evolved. How are today’s companies providing a “future-ready” environment?
By Joe Darrah, Contributing Editor
Some of the drug industry’s most sensitive and sophisticated products represent a diverse who’s who that are bound for the cold chain journey. Reconstituted antibiotics, insulin, messenger RNA (mRNA) vaccines, monoclonal antibodies (mAbs), cell and gene therapies (CG&Ts), and glucagon-like peptide-1 (GLP-1) agonists—each pharmacologic requires an array of storage and handling protocols. Due to a wider range of differences in chemical and biological makeup that inherently cause sensitivity to hot and cold temperatures, the stability of many drugs today can be more difficult to maintain. At the same time, demand for cold chain services continues to cause increased challenges to logistical planning and follow through. “Being able to mass produce the packaging and have it ready to go is more critical than in the past, when cold chain medications were more stable in their growth,”
Baseline controlled room temperature ranges from 15°C to 25°C and is the standard defined by the United States Pharmacopeia for proper storage of pharmaceuticals and other sensitive medical products.

Courtesy: DHL Customer Solutions and Innovations
Thermal modeling expertise provides actionable data to customers in order to make decisions proactively.
says Scott Dyvig, vice president of business development at Altor Solutions. Previously, he explains, volumes were easier to scale. Today, biologics and other temperature-sensitive products are expanding rapidly, including those still in clinical trials.
As more cold chain-necessary drugs come to market, and financial pressures related to global tariffs and the overall rising costs of healthcare persist, Altor and other companies are developing and launching new strategies to package, ship, and store items more intelligently and safely with the help of advanced technologies.
“Put simply, the cold chain is no longer made up of a select few products—it is the core platform for the next generation of personalized and advanced medicines,” explains Trevor McCormick, senior manager of global packaging systems tech operations at Marken, a healthcare logistics company. For many patients, he adds, a shipment may represent their only chance at survival.
Trends Impacting Cold Chain Logistics
Dyvig points to GLP-1 medications as a defining trend entering 2026. “GLP-1 medications are dominating the cold chain discussion,” he says. Surging prescriptions shipped directly to patients’ homes have shifted distribution dynamics. Compound pharmacies, historically operating at smaller volumes, now face unprecedented cold chain demand. “That has elevated their cold chain packaging demand,” Dyvig notes, as they adapt to mass distribution from comparatively small footprints.
Direct-to-consumer delivery also affects CG&Ts, particularly autologous therapies containing a patient’s own genetic material. “These types of products represent the evolution of drugs,” says Cael Pulitzer, managing director at Brown Gibbons Lang &
The surge in prescriptions that are going to patients’ homes has changed the dynamics of cold chain shipping.
Advanced phase change materials are being deployed in some instances as an alternative to potentially hazardous dry ice.
KEY TAKEAWAYS
Company. “They’re really changing the packaging requirements associated with shipping them.”
From Refrigerated to “Ultra” Cold
Once characterized by products that specifically require refrigeration while en route to their destination, the cold chain’s temperature depths have continued to evolve. “New cold chain technologies require special consideration because temperature requirements have expanded beyond traditional ranges,” says Thomas Ellmann, vice president of life sciences and healthcare specialty logistics at DHL Customer Solutions and Innovation. Baseline controlled room temperature ranges from 15°C to 25°C and is the standard defined by the United States Pharmacopeia for proper storage of pharmaceuticals and other sensitive medical products.
This is distinctly different from an environment’s ambient temperature, because active controls are required to prevent potentially dangerous temperature fluctuations that can be caused by external conditions. Standard cold chain refrigerated temperature typically has been 2°C to 8°C, with frozen products requiring -15°C to -25°C. As more innovative drugs such as mRNA vaccines, CG&Ts, and personalized medicines enter the market, it creates the need for “deep-frozen” temperatures (-25°C to -30°C), “ultralow” temperatures (-70°C to -80°C), and cryogenic temperatures (below -150°C and reaching as low as -200°C).
CG&Ts are among the most complex, requiring ultra-low to cryogenic. Gene therapies frequently require deep-frozen conditions and all are highly time sensitive. mRNA vaccines, while still requiring ultra-low storage, have evolved to tolerate slightly warmer conditions for short periods. Meanwhile, GLP-1 therapies are driving volume in the traditional refrigerated segment.
While refrigerated remains the vast majority of volume being distributed today, frozen is among the fastest growing segment, says Anthony Rizzo, chief commercial officer at Cold Chain Technologies. “With frozen products, historically the need was to only keep it frozen, and most companies simply put dry ice in an insulated box,” he explains. “Now many products have lower and upper limits while staying frozen due to lack of stability or primary packaging limitations. Some can’t go below -80C and some can’t go below -35C, but they have to be below -15C. As a result, packaging companies and logistics providers have developed specialized products and services to support these new requirements.”
As the changes in temperature specifics have emerged and created a demand for more specialized packaging and logistical solutions, it’s almost as if the term “cold chain” is becoming somewhat antiquated. “We’ve referred to it as ‘cold chain’ distribution, but
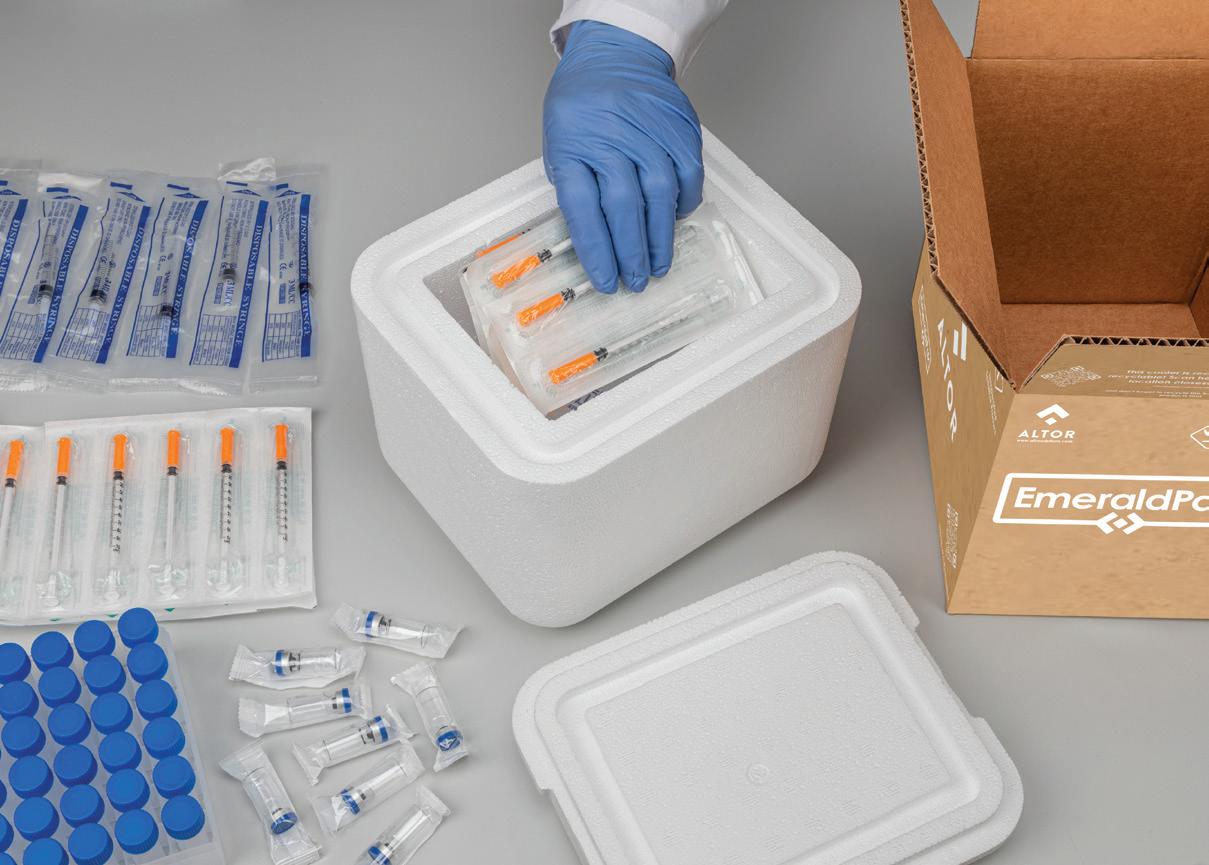
that term is really used today for anything that is temperature sensitive, whether it be too hot or too cold,” says Rizzo.
At Cold Chain Technologies, advanced phase change materials (PCMs) are being deployed in some instances as an alternative to potentially hazardous dry ice due to regulatory, health, and safety issues. Among these options is EcoFlex reusable solution, said to have the ability to maintain various precise temperature ranges for up to seven days.
Reusable Resources & Sustainability
As the volume of cold chain shipments grows, the need to consider sustainability and the conservation of resources become priorities, says McCormick. “We need to be asking ourselves what we can do to lower costs or prevent wasting other resources, such as dry ice, to reduce our carbon footprint,” he says.
Part of this commitment at Marken is an increase in real-time monitoring temperature-controlled containers embedded with next-generation data-monitoring tools that provide enhanced thermal stability and longevity with sustainable reconditioning to power reverse logistics, reliable data acquisition, and presentation for result verification. At Altor, sustainability efforts are best defined by a variety of expanded polystyrene (EPS) shippers, says Alex Arabea, senior brand manager, including the company’s newest innovation EmeraldPak, an EPS product made from recycled content. “This really is a new trend in the industry. Recycled content in EPS is going to be one of the big topics for the next couple years. Many people don’t think of EPS as historically recyclable, but it is. And sometimes the economics of these things are what drives the recyclability of the material,” says Arabea.
Visions of a Smarter Cold Chain Future
The ability of the cold chain to adjust to evolving demands of the various industries it serves has been evident since its origins of railroad distribution. There’s little chance that the need to be
As the volume of cold chain shipments grows, the need to consider sustainability and the conservation of resources also have become priorities.
excessively amenable is going to dwindle any time soon. “As a supply chain, the cold chain is probably the most future-ready in the world,” says Pulitzer.
McCormick says it’s been determined at Marken that nextgeneration therapies require real-time, multi-parameter, actionable data for what is happening with product throughout transit. “Whereas many traditional temperature logs have only provided forensic data to determine what happens after the fact, our realtime data solutions offer the ability to rapidly intervene when an adverse condition arises,” he says. “Smart tracking using Internet of Things (IoT) technology is another way we track packages faster and more reliably.”
One such IoT example at Marken is a digital, serialized security seal that’s connected to real-time monitoring devices. After any seal is cut, a reported record of where and what time the seal was broken is provided to customers to confirm a digitized chain of custody. These advancements increase operational complexity, requiring adaptation in both human and machine resources.
At Cold Chain Technologies, CCT Smart Solutions™ integrates connected technologies to provide greater visibility across the

logistics cycle. The company is integrating real-time monitoring and analytics into packaging, working with partners to turn data into actionable insights. “It’s leveraging the right package for the right shipment,” Rizzo explains, using risk-scoring models to enable proactive decisions.
McCormick envisions a future where blockchain and artificial intelligence (AI) are more significant as well. “The future-ready cold chain is digitally intelligent and predictive,” he says. “This could involve control towers enhanced with AI to predict demand fluctuations, shortages, and possible failures like flight delays, customs issues, weather, or labor shortages ...Furthering these efforts will be more decentralized manufacturing, a shift from large production centers to more regional—perhaps—modular, hubs.”

New cold chain technologies require special consideration because temperature requirements have expanded beyond traditional ranges.






















Great Earth Expands Use of Fiber-based Closures to Entire Line
Swedish nutraceutical brand rolls out paper screw caps across all supplement jars, a first large-scale commercial use of Blue Ocean Closures’ molded fiber technology.
By Matt Reynolds, Chief Editor, Packaging World
KEY TAKEAWAYS
Great Earth converted its entire line of supplement jars to fiber-based screw caps, marking one of the first commercial rollouts of molded fiber closures in the space.
FThe company’s goal is 100% circular packaging by 2030. The fiber lid is “just the first step” toward the goal.
ollowing a successful pilot that began in 2024, Swedish nutraceutical company Great Earth converted its entire line of supplement jars to fiber-based screw caps developed by Blue Ocean Closures (BOC). This marks one of the first, if not the first, full commercial rollouts of molded fiber closures in the health and wellness sector. According to Blue Ocean Closures CEO Lars Sandberg, the pilot launch on Great Earth’s top-selling magnesium supplement was “very positively received by both the market and other actors in the value chain.”
Sandberg says the move aligns closely with both companies’ sustainability ambitions. “This introduction—being first to market with an innovation—has been recognized as a strong move, in line with our shared vision to improve sustainability.”

BOC says its latest generation of fiber screw caps, shown here, can match or outperform fossil plastic in material cost while delivering significantly lower energy consumption during production.
Great Earth’s study revealed that 100% of participants found the new lid easy to use.
Scaling Up and Running on Existing Lines
Transitioning from pilot to full rollout, Great Earth and BOC worked to ensure the new closures could be adopted across the company’s entire range of more than 70 jarred products.
“Moving from pilot to scaling this solution to the full supplement range will take some time,” Sandberg noted, emphasizing the need for “continued close collaboration between us as well as with fillers and distributors.”
More importantly, the switch to fiber did not slow down production. Sandberg confirmed that “the capping line efficiency is not affected by the switch—it runs at the same speed,” though “some small adaptations may be necessary as we move to bigger volumes.” He added that ongoing testing for storage stability has shown “good results.” BOC’s design aims for drop-in compatibility with existing filling and sealing systems.
“No exchange of machinery or equipment [was required],” Sandberg says. “Small adjustments to the settings on the lines are made as we increase volumes.”
Torque performance and seal integrity are reported to be equivalent to conventional plastic closures.
Technical Details: Materials, Barrier, and Recyclability
The fiber caps feature a mechanically assembled induction seal liner, the same liner Great Earth used with its previous plastic caps. Sandberg explained that it “does not affect recyclability as it is attached to the bottle, not the cap.”
Courtesy: Blue Ocean Closures
The liner provides the necessary oxygen and moisture barrier, ensuring shelf life “is not affected.” The caps themselves are made from FSC-certified fiber, and each closure can be recycled in standard paper streams. For nutraceutical applications, BOC’s focus was on dry products rather than beverages.
“Blue Ocean Closures created a first design proposal, and final design discussions were held together with Great Earth,” says Sandberg. “Beverages are outside the scope of this project.”

Both companies also collaborated with fillers during validation and testing. Sandberg added that Blue Ocean Closures is “pursuing a wide range of applications and custom solutions,” including work on “fiber caps for beverage cartons—a huge segment of its own representing around 135 billion screw caps per year.”
Consumer and Retailer Response
On the consumer side, Great Earth conducted a survey of 500 participants to evaluate usability and perception. “The response on usability was unanimous—100% of participants found the fiber lid easy to open and user-friendly,” says Patrik Falk, Great Earth CEO.
Consumers rated the lid’s perceived strength at 4.3 out of 5, leading Falk to conclude that “a sustainable material can also be a robust, high-quality material.” Most strikingly, 95% said they would choose a product with the fiber lid again, which Falk described as “the ultimate validation” of the sustainable shift. Retailers also have responded positively. Sandberg cited a statement from Apotek Hjärtat, Sweden’s leading pharmacy chain, which read, “We need to reduce the use of plastic where we can and help our customers make conscious choices. The closure is recyclable and made from renewable resources, which is in line with our packaging strategy.”
Great Earth earned “Sustainable Supplier of the Year” from Apoteket AB, another major pharmacy chain, which praised the company for “combining sustainable innovation with a focus on health and well-being.”
The Business Case: From Cost to Circularity
For Great Earth, the shift is about long-term competitiveness as much as sustainability. Falk described the transition as “businesscritical,” noting that sustainable packaging has become “a competitive advantage.” He added that the recognition from major retailers underscores how packaging innovation can drive both environmental and commercial success.
“Today’s consumers want their wellness choices to reflect their values,” Falk says. “Sustainable solutions can’t carry a price premium, but when given the choice at the same price point, consumers consistently choose the sustainable option. Our fiber lids make that choice simple at the shelf.”
Falk says the company’s goal is 100% circular packaging by 2030, and the fiber lid is “just the first step.” He continues: “Each category requires its own tailored innovation, and we are already deep into that process. When we set our 100% circular goal for 2030, we didn’t have a perfect roadmap—we simply knew it was non-negotiable. That’s the power of setting a bold target without having all the answers: it forces innovation to happen.”
On the cost side, Sandberg confirms “there’s no difference in price at present” between the fiber and plastic caps, a fact bolstered by Blue Ocean Closure’s recent breakthrough in manufacturing efficiency. According to an October 2025 company announcement, BOC’s proprietary deep-forming technology enables production cycles below two seconds and energy consumption roughly one-tenth that of conventional plastic molding. The company now
Great Earth’s full supplement line now features molded fiber screw caps developed with Blue Ocean Closures, replacing plastic across more than 70 products.
Courtesy: Great Earth and BOC



reports that fiber-based closures “can match or even undercut fossil plastics in cost” while remaining recyclable and low-carbon.
“This is the moment when sustainability and profitability truly align,” Sandberg says.
Looking Ahead
As Blue Ocean scales up production for nutraceutical applications, Sandberg says brands across Europe are starting to adopt
Bottles misaligning


fiber cap solutions. “We are very fortunate to work with early adopters such as Great Earth and global brands such as The Absolut Company,” he says. “This is truly a game-changer for sustainability in packaging—and for the planet.”For Great Earth, the change represents both leadership and learning. “We’re not just meeting EU regulations,” Falk says, “we’re years ahead of them. That’s the difference between compliance and leadership.”
Editor’s note: The Blue Ocean Closures–Great Earth collaboration represents a rare commercial milestone in molded fiber packaging— a proof point that sustainability-driven innovation can achieve functional, regulatory, and cost parity with plastic. The nutraceutical sector, with its shelf-stable dry contents, may become the proving ground for paper-based closures before the technology expands into more demanding categories like beverages and liquid foods.




Courtesy: Blue Ocean Closures
Older prototype of Blue Ocean Closures, used during pilots with Pernod Ricard’s Absolut brand of vodka.




PIONEERING INNOVATION SINCE 1950
Ensure your products match the prescription. Quality Control. Inspection and foreign object detection systems that offer unparalleled sensitivity for pharmaceutical products to protect your

and your brand.



&
• CEIA high sensitivity metal detection; ferrous, non-ferrous, stainless steel
• Ishida X-ray for foreign object and product defects
• Ishida precision checkweighing for weight verification
• Powders, capsules, tablets, liquids
Heat and Control® offers a complete line of metal detectors, checkweighers, and X-ray inspection systems for pharmaceutical products from the leading manufacturers, Ishida and CEIA®. Technical support, demonstration, and testing is available in North, Central and South America.



Green Goo Brings Plant-Based Healing to the First Aid Aisle
Inside the sustainable processing and packaging strategy behind Green
Goo’s plant-based first aid revolution.
By Liz Cuneo, Editor-in-Chief
When Jodi Scott helped co-found Green Goo nearly 20 years ago with her sister and mother, the idea of a plant-based first aid line seemed unconventional—if not impossible. Today, their company stands at the forefront of sustainable healthcare and personal care, proving that natural, effective, and FDAcompliant formulations can coexist.
From Kitchen Creations to Clinical Use
KEY TAKEAWAYS
As a Certified B Corp, Green Goo proves that sustainability and clinical efficacy can thrive together.

Scott’s background bridges science and psychology, while her sister’s training as a midwife and herbalist brought deep botanical expertise. When her sister began experimenting with plant infusions at home, the family quickly noticed the results.
“We were living these very healthy, conscious lives—organic foods, clean shampoos—but if you opened our first aid cabinet, it was full of single-use plastics and chemical-laden ointments,” Scott recalls. “It was the one exception we were making, and it didn’t make sense.”
So, the sisters began crafting their own natural alternatives for burns, cuts, and skin irritations. What started as a personal experiment soon gained traction at local farmers’ markets. “Within months, we had the longest line,” says Scott. “People were telling us their son wasn’t embarrassed to go to school anymore because his eczema had cleared up. Others shared stories about pain relief or faster healing. That was the moment we knew—there’s a gap in this space.”
But scaling from kitchen batches to regulated, FDA-registered products was no small feat. The family was determined not to compromise the hands-on process that made their formulas effective. They continued bringing raw, whole herbs into their facility to extract oils themselves—an approach few manufacturers were willing to accommodate. “We met with so many manufacturers who said, ‘Can’t we just buy pre-made extracts?’” Scott recalls. “But the efficacy would be compromised. We refused to cut corners.”
The company’s manufacturing breakthrough came through an unlikely encounter. At a trade show, a manufacturing manager stopped by their booth, took home a sample for his son’s eczema, and called them a week later. “He said, ‘I manage a manufacturing facility, and my son has never experienced relief like this. The world needs these products. We’re going to make this happen.’”
That partnership led to the creation of a dedicated production line and team, built around Green Goo’s unique infusion process—a collaboration that continues today. The company’s early Dry Skin formula from that line is now used by Johns Hopkins’ Scleroderma Skin Condition Department, where it has been helping patients for more than a decade.
Packaging with Purpose
From Day 1, the Scotts viewed packaging not as an afterthought but as an extension of their values. They chose glass jars and aluminum tins—recyclable, stable, and ideal for oil-based
A family of scientists and herbalists built Green Goo to bring plant-based first aid products to market.
The company combines FDA-compliant production with sustainable packaging like aluminum tins and sugarcane bioplastics.
Jodi Scott is the co-founder of Green Goo, a plant-based first aid company.
Courtesy: Jodi Scott

products—over plastic tubes and one-time-use containers.
“We’ve tested every material that’s come along,” Scott says. “Fifteen years later, aluminum and glass are still the best performers. They’re renewable, they protect our formulations, and they’re easy for consumers to recycle.”
“SUSTAINABILITY IS A PROCESS, not a finish line. Every choice—from your supply chain to your shipping method—matters.
Green Goo also experimented with sugarcane-based bioplastics for its toothpaste and hand sanitizer lines, particularly during the pandemic when demand for sanitizers surged. “I couldn’t stand the idea of all that plastic going out the door,” says Scott. “So, we sought a renewable alternative that could withstand the high alcohol content. The sugarcane packaging held up beautifully—it also composted in a fraction of the time of traditional plastic.”
Although the toothpaste line is temporarily paused, Scott says the company is watching new advances in bioplastics with interest.
“We were one of the first to use sugarcane packaging,” she says. “It’s exciting to see how much progress has been made since.”
— Jodi Scott, Co-founder, Green Goo.
Science Meets Sustainability
Despite its natural ethos, Green Goo plays by the same regulatory rules as conventional OTC brands. All of its products are FDA-registered, meaning each formulation must use only recognized active ingredients and meet rigorous stability and quality standards.
“The FDA only acknowledges certain plants as active ingredients,” Scott explains. “If an herb isn’t listed, it becomes an inactive ingredient—even if it has powerful properties.”
Green Goo’s sustainability commitment extends beyond materials. As a Certified B Corporation and cruelty-free company, its mission includes community, fair trade, and environmental stewardship.
Green Goo’s tins, made from 100% recyclable aluminum, can be tossed into curbside recycling—or reused for crafts, travel kits, and storage.
Each ingredient undergoes certificate-of-analysis verification, followed by multiple in-process and post-production quality checks. Batch samples are retained for years, ensuring traceability and compliance with GMP-like standards.
Green Goo’s sustainability commitment extends beyond materials. As a Certified B Corporation and cruelty-free company, its mission includes community, fair trade, and environmental stewardship. Scott credits the B Corp process for helping the team identify new ways to improve. “Every year, we come out of recertification with five to 10 new action items,” she says. “It’s not just about meeting a score—it’s about being a better steward of the environment.”

initiatives. “Every purchase is a vote,” Scott adds. “If consumers keep voting for sustainable brands, the big players will have to follow.”
Beyond the Tin
One benefit to lidded aluminum packaging is that it can be reused over and over again.
That mindset has driven collaboration with suppliers and even competitors. “Sometimes smaller brands (like us) can pitch to manufacturers—‘I can’t buy 20,000 of these sugar cane tubes, but would you mind putting it in front of some of your other vendors to see if they would be interested too, so that we can make that happen?’” she says. “It’s about thinking outside the box so innovation can scale.”
The Market’s Slow Shift
While sustainable packaging has taken hold in beauty and personal care, Scott notes the over-the-counter (OTC) first aid market still lags behind.
“We’re still part of the plant-based renaissance,” she says. “Consumers in healthcare haven’t fully demanded sustainable alternatives yet—but it’s coming.”
She applauds retailers like Walmart for incentivizing greener suppliers and sees promise in Amazon’s carbon-reduction
“A man once told me his kids collect our tins, fill them with dirt, and play with them outside,” Scott laughs. “That’s exactly the kind of circular use we hope for.”
The tins, made from 100% recyclable aluminum, can be tossed into curbside recycling—or reused for crafts, travel kits, and storage.
Scott hopes her family’s journey offers lessons for both startups and established manufacturers looking to integrate sustainability. “Don’t give up,” she says. “Test every material, even if it doesn’t work the first time. And collaborate—if your order volume is too small, find others to join in. Shared innovation gets you access to new packaging technologies faster.”
Above all, she says, the key is persistence and partnership. “Sustainability is a process, not a finish line. Every choice—from your supply chain to your shipping method—matters. If we keep asking how to do better, we all move forward.”
Courtesy: Green Goo
MG America... the perfect choice for processing and packaging equipment.

A complete line of solutions from one trusted partner.
Processing
• Capsule fillers
• Capsule check-weighers
• Capsule banding machines
• Metal detectors for capsules & tablets

• Bin blending equipment
• Material handling equipment
• Washers
• Sterilizers
• Depyrogenation

Packaging
• Pouch fillers
• Cartoners
• Tray formers
• Case packers
• Palletizers
• Stick Pack
• Bottle unscramblers
• Packaging line integration

• Bottle fillers/cappers
- Tablets/capsules & liquids
• Bottle labelers
• Bottles: Carton coding, verification, serialization
• Blister machines
• Thermoformers

Fernway Cannabis Automates Labeling For Various Markets
Faced with fragmented packaging regulations and manual labeling inefficiencies, Fernway turned to Paxiom’s dual-head carton labeler for a solution.
By Matt Reynolds, Chief Editor, Packaging World
KEY TAKEAWAYS
For Fernway, packaging isn’t just a vessel for cannabis products—it’s a reflection of the brand’s identity and its promise of quality and care. Founded in 2019 by four friends in Massachusetts, Fernway has grown rapidly, expanding into New Jersey, New York, and Illinois. But with each new market comes a new set of labeling and regulatory hurdles.
“Every time we enter a new market, we do a regulatory analysis under the lens of packaging,” says Kevin Wu, Fernway’s COO and co-founder. “The goal is to make one carton or pack fit as many states’ regulations as possible.” Each state dictates its own rules for THC potency data, batch labeling, expiration dates, and even illustrations. “For example, Illinois doesn’t allow images of fruit on cannabis packaging [to avoid being mistaken for candy by children], so we replaced our strawberry illustrations with drawings of ferns.” Navigating that patchwork of rules became a logistical and operational challenge. But the bigger strain came from the labeling process itself.

From Hand Application to Automated Precision
Before automation, Fernway’s operators printed regulatory labels on Epson printers, loaded them onto manual dispensers, and applied each one by hand. That meant roughly one carton per minute—far too slow for a company scaling into multiple states.
“The human hand can only work so fast,” says Wu. “It wasn’t a big deal when we were selling a few thousand units a month, but once that turned into tens of thousands, it became unsustainable.”
Label accuracy was another pain point. “Having a premium brand means the product has to look the part,” Wu adds. “It’s hard to pull that off when the label is crooked or hanging off the edge of the box.”
That’s when Fernway turned to Paxiom for a more scalable solution. The company purchased its first dual-head carton labeler in 2022 for Massachusetts, then followed with additional units for New Jersey, Illinois, and New York as new facilities came online. A fifth system is planned for early 2026.
Paxiom’s two-headed carton labeler applies labels to two sides of a carton at up to 30 to 40 cartons per minute, a roughly 30x improvement over hand labeling. Operators feed cartons onto a conveyor manually, and as they move through the machine, each label head applies one of two labels in sequence—typically a front-facing regulatory label and a secondary back label for marketing copy, QR codes, or batch details.
The system features a stainless-steel frame, a split-belt conveyor with product guides, and a photo cell start/stop sensor to ensure precise timing and label placement.
The dual-head labeler applies regulatory and branding labels to opposite sides of premium cartons at up to 40 per minute.
Courtesy: Fernway
Fernway has grown rapidly, expanding into several states. But with each new market comes a new set of labeling and regulatory hurdles.
Manual labeling was both too slow for the company, and often inaccurate.
Paxiom’s two-headed carton labeler applies labels to two sides of a carton at up to 40 cartons per minute.








HIGH-SPEED DIGITAL INLINE LABEL PRINTING FOR ON-DEMAND PRODUCTION OF COMPLETE LABELS AND VARIABLE DATA CODING
The Greydon L-Max digital label printer is an advanced solution designed for high-quality label printing across various industries. It offers high-speed, on-demand printing and produces high-resolution markings, images, and text, making it ideal for the pharmaceutical and medical device industries.

DYNAMIC WEB HANDLING
Integrated servo-controlled web handling allows for continuous and intermittent label printing for complete production line integration flexibility.

INTELLIGENT UV CURING
Inline intelligent UV curing system ensures all printed labels are fully cured even under line stoppages and label material replenishments.

INTEGRATED SPLICE TABLE
Operator friendly built-in splice table provides easy access for fast and seamless label replenishments resulting in increased label printing throughput.
INCREASE YOUR DIGITAL LABEL PRINTING & THROUGHPUT


Fernway’s premium rigid cartons, used for edibles and vaporizer products, now carry consistent, compliant labeling in Massachusetts, New Jersey, Illinois, and New York.
“It’s accurate, fast, and flexible,” says Wu. “We even reduced the footprint of our packaging by 30% without having to modify the equipment. We just created new programs and recipes.”
Wu also praised the ease of training and changeover. “After a few weeks, someone can go from zero to fully understanding not just how to use the machine but also how to troubleshoot and reprogram it for different box sizes,” he says. “That’s critical for us because we’re constantly changing over—sometimes every thousand or two thousand cartons.”
Managing The Data Behind The Label
The variable data printed on each Fernway label is drawn from test results uploaded by independent labs to a platform called
Confident Cannabis, then integrated into the company’s Acumatica ERP system. That ensures the correct batch data— THC content, potency, lot number, and expiration date—flows seamlessly to each label. “Having the right software to manage all that data has been huge,” Wu says. “It’s what keeps us compliant across states.”
With automation, that data integrity now extends to the physical label application. The Paxiom equipment provides not just speed and precision, but consistency that human operators couldn’t match. “Occasionally we’d find a missing label or one upside down,” Wu says. “Those were experiences we just couldn’t afford to have.”
Training and Support
Each new machine installation is supported by an on-site Paxiom technician, who commissions the equipment and provides hands-on training. “They stay a couple of days to work with our team and make sure we’re comfortable running changeovers, handling maintenance, and troubleshooting,” Wu says. “By the time we got to our third and fourth machines, the process was seamless. Our technicians were already familiar with the workflows.”

ADVANCED INSPECTION TECHNOLOGY TRUSTED WORLDWIDE FOR PHARMACEUTICAL INTEGRITY & COMPLIANCE
Patient safety is on the line, so you need equipment that’s more than just good. Anritsu systems deliver precision, compliance, and low total cost of ownership. Plus, we back our advanced technology with unparalleled service and support.
Discover what you’ve been missing. Learn how to efficiently elevate safety and consistency at www.anritsu.com/product-inspection
Courtesy: Fernway



Fernway’s labeling cells are currently semi-automated—the cartons are hand-fed into the machine and case-packed manually after labeling—but Wu says the company is now exploring downstream automation with Paxiom to extend those gains further. “We’d love to fully automate everything,” he says, “but because every state requires its own facility, each investment gets multiplied across multiple locations.”
Even with those constraints, the investment in automation has paid off. “The payback has been roughly a year,” says Wu. “That’s equivalent to about two technicians’ worth of labor. It allows us to stay lean, give our team job security, and let them focus on higher-value work.”
Thinking Beyond The Consumer
Wu offers a piece of advice that applies to cannabis brands and CPGs alike: when designing packaging, think about the entire supply chain, not just the consumer experience.
“When we first designed our box, we were thinking only about our brand and the end consumer,” he says. “What we didn’t do was talk to our dispensary partners, and that was a mistake.”
Dispensaries in Massachusetts and New Jersey, for example, urged Fernway to reduce its package size to fit more inventory in limited vault space. “That feedback led us to cut our carton footprint by 30%,” Wu says. “It was a lesson in remembering that our ‘customer’ isn’t just the person who opens the box—it’s everyone who touches it along the way: transporters, dispensary staff, budtenders, and finally the consumer.”
For Fernway, packaging is both a brand expression and an operational necessity. And by investing in automation, the company is proving that even in a fragmented, heavily regulated market, consistency and quality can still scale.

Excellence in Precision Healthcare Packaging
ESS Technologies delivers robotic packaging solutions engineered for high performance and flexibility including:
• High precision pick and place
• Multi-format cartoners
• High efficiency case packers
• Modular palletizers


The stainless steel, split-belt conveyor system provides precise control for consistent label placement.
Courtesy: Fernway
Building Better Brushers: How Babsy Reinvented the Children’s Toothbrush
The packaging channels an arts-and-crafts aesthetic, made to look like a creative kit rather than a sterile healthcare product.
By Liz Cuneo, Editor-in-Chief
KEY TAKEAWAYS
From the ouset, the Leadof Studio team knew the key was to merge playfulness with professional credibility.
When pediatric dentists Dr. Zach Gelber and Dr. Dante set out to tackle one of parenting’s most universal struggles—getting kids to brush their teeth—they weren’t thinking about sleek tech or digital reminders. Instead they were thinking about empowerment and self-expression. Together with Jordan Diatlo, founder and creative director of Leadoff Studio, they created Babsy, a customizable toothbrush brand that turns a daily chore into a creative, confidence-building experience.
During their pediatric dental residencies, Dr. Gelber and his co-founder noticed a recurring theme: Nearly every parent asked the same question—how do we motivate our kids to brush their teeth, and how do we get them to want to? Gelber said regardless of the age of the kids, it was a common problem. The dentists saw firsthand how children gravitated toward products that reflected their personalities, from Crocs with collectible charms to T-shirts

In a broader healthcare context, Babsy reflects the growing trend toward personalized wellness products.
with beloved characters. That observation sparked an idea. “We wanted to put the power in kids’ hands,” says Gelber. “Instead of brushing being a battle, what if it became something they built and owned?”
This concept led to Babsy’s “Build-A-Brush” system—an interactive toothbrush that allows children to assemble and personalize their own brush using colorful, interchangeable parts.
Designed for Play and Purpose
To bring their vision to life, the Babsy team turned to Leadoff Studio, a New York-based industrial design firm known for consumer products that blend function and emotion. From the outset, Diatlo and his team understood the key was to merge playfulness with professional credibility.
“Parents expect something safe, effective, and dentist approved,” says Diatlo. “Kids, on the other hand, want fun, color, and creativity. Our job was to design a product that spoke to both.”
The resulting design channels an arts-and-crafts aesthetic, with packaging that looks like a creative kit rather than a sterile healthcare product. The outer box is made of natural cardboard and includes a “Created by Dentists” badge displayed prominently—a subtle but crucial reassurance for parents that playtime doesn’t come at the expense of performance.
Inside, the brush components are neatly arranged in an organized and intuitive layout—a moment of calm before what Diatlo calls “the explosion of color and fun” once kids start assembling.
Babsy is a customizable toothbrush brand that turns a daily chore into a creative, confidence-building experience.
Courtesy: Leadoff Studio
The packaging design for the Babsy Build-A-Brush encourages play and is scalable for future starter kits, brush heads, and expansion packs.

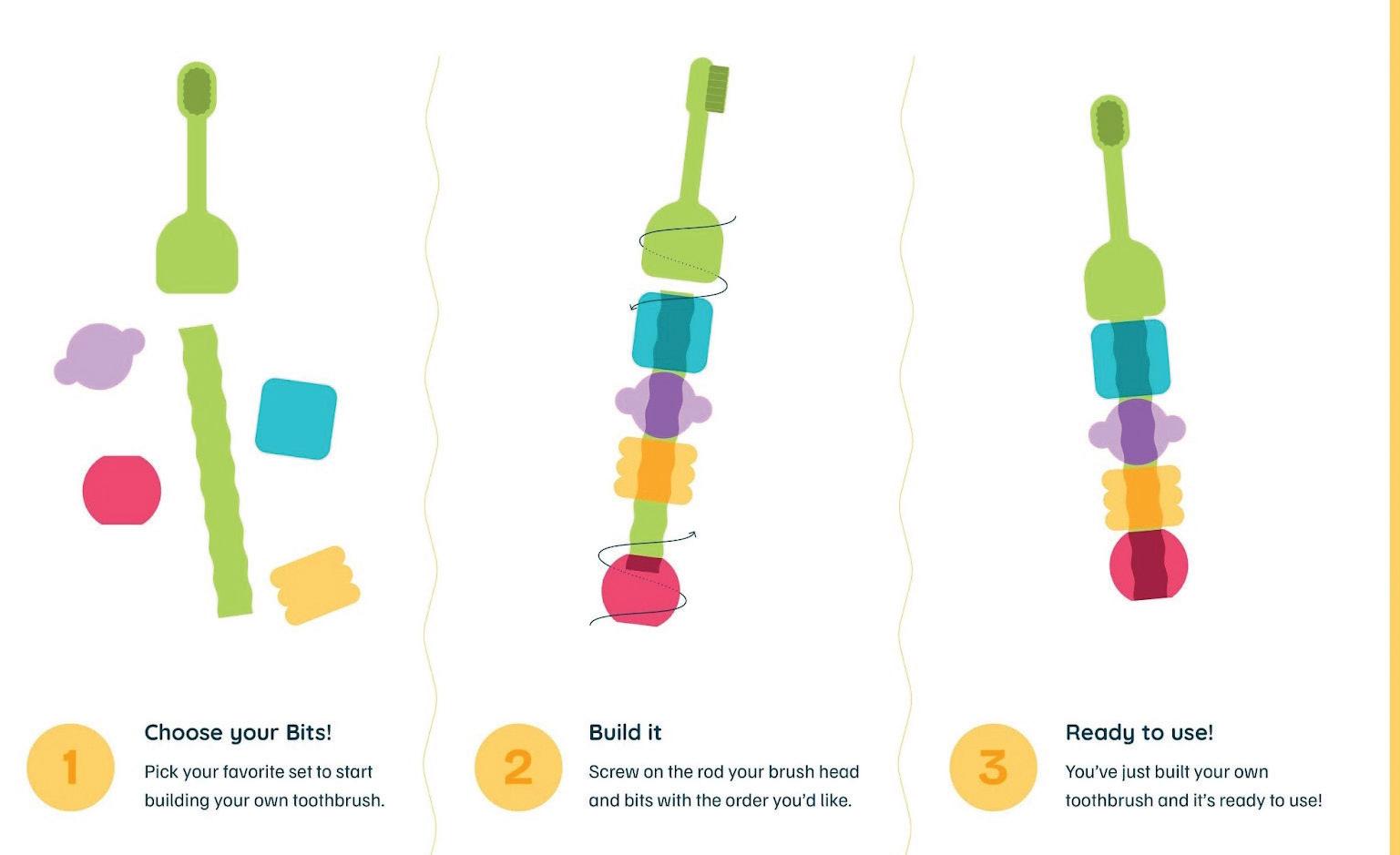
“We wanted it to be something that the second you open the box, kids know what it is and are wanting to take it out of the box and start using it right away,” says Gelber.
The packaging layout helps children instantly recognize what to do. The illustrated instructions use simple visuals rather than heavy text, allowing even toddlers to follow along.
Engineering for Imagination
Gelber, Dante, and Diatlo didn’t just rely on lab testing to see if the new toothbrush resonated with kids, they used their own children as product testers. Early testing included prototypes—3Dprinted in plain white plastic, which became hot commodities in their households.
“When I gave my kids a bag of test parts, they started fighting over who got which pieces,” Diatlo recalls. “That’s when I knew we had something special.”
Gelber echoes that sentiment. “It was exciting to see how much ownership they took over the process. Even younger kids who couldn’t assemble it completely still felt proud choosing the pieces they liked.”
While the “testing” was successful, the modular design posed unique manufacturing challenges for the company.
“One challenge was creating a standardized box that would fit all of the different bits in the expansion packs. For example, this one is a sphere, then a cylinder, and the spiral. That was the first challenge, just figuring out the manufacturing,” says Diatlo.
Each interchangeable piece—some with ears, spirals, or distinct shapes—had to fit into standardized packaging while staying costeffective for production. Leadoff Studio solved this by developing custom inserts and mold groupings, creating a scalable system that allows Babsy to expand its lineup with new refill packs and designs.
Personalization Meets Preventive Care
In a broader healthcare context, Babsy reflects the growing trend toward personalized wellness products. From tailor-made supplements to smart wearables, consumers increasingly want healthcare items that reflect their identities and lifestyles.
“It’s all about self-expression and motivation,” says Gelber. “If a child feels that their toothbrush represents who they are—even if that changes from a love for pizza one day to mermaids the next—they’re more likely to use it.”
The original goal of this project was to increase adherence— essentially to get kids to want to brush their teeth more.
“We were trying to figure out how to increase the efficacy through actually tapping into the motivations of a child and figuring out what the child is actually driven by: in this case, the self-confidence building and the sense of their self,” says Diatlo.
By merging design thinking with pediatric insight, the brand bridges the gap between healthcare and creativity.
“It’s not just a tool for a chore,” says Diatlo. “It’s an object of joy—something kids are excited to use. And if that excitement leads to better brushing habits, then we’ve achieved what we set out to do.”
On the Expansion Packs, the graphic design is scalable for future product releases.
Babsy’s “Build-A-Brush” system is an interactive toothbrush that allows children to assemble and personalize their own brush using colorful, interchangeable parts.
Courtesy: Leadoff Studio

Global Labeling Challenges in Pharma: Insights from Kite Pharma
For cell therapy manufacturers such as Kite Pharma, where every product is made for a single patient, labeling becomes a critical safety and traceability tool.
By Liz Cuneo, Editor-in-Chief
KEY TAKEAWAYS
Across regions, regulatory bodies continue to add more required content to pharmaceutical packaging.
Pharmac eu tical labeling has never been more complex—or more vital. As therapies become increasingly personalized and global regulatory requirements evolve, the role of packaging and labeling teams has expanded far beyond simple content placement. For cell therapy manufacturers like Kite Pharma—where every product is made for a single patient—labeling plays a critical role in safety and traceability tool.
Pharmaceutical manufacturers like Kite also must ensure regulatory compliance across global markets.
As in many industries, the future of pharma is going digital.
“That’s one of our biggest challenges.”
As pharmaceutical development becomes more global, personalized, and digitally connected, labeling teams sit at the intersection of compliance, safety, and patient experience.
To unpack the realities of today’s labeling landscape, I sat down with three labeling experts from Kite Pharma: Craig Vermeyen, senior director leading the packaging and labeling team; Sabrina Conner, who heads the clinical labeling team; and Josh Lipking, who oversees commercial labeling. Together, they offered a candid look at regulatory requirements, common pitfalls, global complexity, and the technology shaping the next decade of pharmaceutical labeling.
Regulation Is Expanding—But Labels Aren’t
Across regions, regulatory bodies continue to add more required content to pharmaceutical packaging. Yet, of course, blister packs, vials, and syringes maintain their original footprints. “Labels aren’t getting bigger,” Lipking explains. “A blister pack will remain the same size, but regulations require more and more data like barcodes, additional statements, and text visibility requirements.
Readability and design constraints compound the issue. Small containers still must meet type-size requirements and maintain human readability. Regulatory affairs teams often must negotiate with agencies on what goes where, especially as new mandates arrive.
“We face the same constraints in clinical labeling,” says Conner. “There’s only so much content we can fit, so there’s a lot of negotiation with regulators to determine what’s acceptable for patient supply.”
While that can be complex enough, pharmaceutical manufacturers like Kite also must ensure regulatory compliance across global markets. And label format, structure, and content hierarchy may shift dramatically between countries.
“We work closely with our regulatory partners,” says Lipking. “Often, the structure of the content may change from one region to the next. It’s our job to ensure that it translates well, that the hierarchy remains readable and understandable.”
One such example is the company’s U.S. commercial label compared to its Japanese label.


“There’s a striking difference—different languages, formats, and expanded statements—required in Japan,” says Lipking. “The whole structure shifts to meet those needs.”
According to Vermeyen, this requires regional expertise. “We’re fortunate to have Gilead affiliates embedded around the world who help us track local regulatory changes. Having someone who understands the regional regulatory climate is essential.”
Version control is another critical tool for keeping global variations in check. “Version control is big,” Conner notes. “We always want to make sure content version control is locked in because that helps us adhere to the right content.”
Speaking of global, a question on the minds of many: Is a harmonized global labeling standard on the horizon? Lipking sees some movement. “I’ve noticed more harmonization between FDA and EMA. It’s not perfect yet, but there’s progress.”
But while harmonization is a goal, regional differences remain entrenched. “Understanding the specific requirements for each jurisdiction is still a big part of our job,” says Vermeyen.
Common Pitfalls
As complex as labeling can be, the importance of accurate labeling is critical. Common pitfalls that companies face include inconsistent labeling due to multiple requirements, core data differences country to country, insufficient version control, and even translation issues.


Other pitfalls include safety changes not implemented quickly enough, serialization, and ineligibility due to lack of designer education.
“I’ve also noticed fewer people with design expertise to lay out label artwork,” says Lipking. “Print is unique. Many people can design for web and digital consumption, but print requires a different skill set.”
A lot of information needs to go on labels now, including serialization and traceability to track product from Kite all the way to the patient, but for Kite that is less of a concern than other pharma companies. While serialization can be complex, Kite’s patient-specific manufacturing model simplifies some aspects. “We manufacture a lot for one customer,” Lipking says.
But traceability remains critical—especially in cell therapy. “Kite has patient-specific therapy, which allows us to track and trace all lots going to each specific patient with more ease
than many big pharma companies that have giant lots with thousands of vials to track,” says Vermeyen.
While it may be easier, the importance is still there.
“
“We always maintain the chain of custody throughout handoffs to ensure the right patient receives the right dose,” explains Vermeyen. “Chain of identity ensures we know which patient’s cells are in that shipping container at all times. The labeling contents of that shipping container include patient-specific identifiers that can be tracked and documented at each handoff to ensure there’s never any mix-up.”
The Future of Pharma
As in many industries, the future of pharma is going digital. In the realm of labeling, that likely will include technologies such as e-labeling and QR codes.
“Imagine needing only a barcode on a product while all content is viewed digitally,” says Vermeyen. “That’s somewhere we definitely want to get to ... To support our initiative to maintain supply for life-saving therapies, it’s important to embrace technology as it emerges, even if it’s something we don’t yet
NEEDING only a barcode on a product while all content is viewed digitally. That’s somewhere we definitely want to get to ...
” IMAGINE
—
Craig Vermeyen, senior director leading the packaging and labeling team, Kite.
know exists.” Some regions are already experimenting. Kite participated in a Dutch government trial that eliminated leaflets in favor of digital content.
“We were excited to be at the forefront,” Lipking notes. “We’ve found many benefits in the simplicity it provides.”
Digital labels offer the benefit of real-time updates. “Instead of waiting for printed materials, patients could immediately access the most current information,” says Conner.
As pharmaceutical development becomes more global, personalized, and digitally connected, labeling teams sit at the intersection
Curious why experts choose Sepha over blue dye?
As a global specialist in non-destructive and deterministic CCIT solutions, we partner with all major pharma companies, including biopharma, generic drug manufactures, CMO's and speciality pharma organizations. These companies have chosen our innovative technologies over blue dye for:
• Non-destructive methods
• Accurate, rapid pass/fail results
• Vacuum decay & Pressure decay
• ASTM / USP 1207 compliance
• 21 CFR Part 11 reporting






Are you ready to switch to non-destructive and deterministic CCIT for better quality assurance?
Sepha North America 10100 Progress Way Harrison, OH 45030 Tel. 888-241-3179 info@sepha.com
What’s in Store for Pharmaceutical Manufacturers in 2026 and Beyond?
Key trends and insights reveal a focus on sustainability, smart packaging, and incorporating more digital tools.
By Liz Cuneo, Editor-in-Chief
The healthcare packaging landscape—and the broader medical device, pharmaceutical, and life sciences ecosystem—is evolving at an unprecedented pace. Healthcare Packaging’s inaugural Annual Outlook Report is designed to track and benchmark these shifts, providing insight into how packaging innovations, regulatory pressures, and market dynamics are shaping the future of healthcare delivery. This report will monitor key trends and changing perspectives as the industry continues to adapt to the demands of regulatory pressures, patient safety, and sustainability in an increasingly fast-moving environment.
Which of the following factors most influence your packaging decisions? (Select all that apply)
What’s driving packaging decisions? The top response is cost efficiency at 67%. Following close behind is supply chain resilience at 62%, followed by sustainability and eco-friendly materials (53%).
Supply
“We hear a great deal about patientcentricity as a guiding principle: the idea that packaging should not only protect medicines but actively support patient adherence, safety, and experience. Yet when industry professionals are asked to rank their top priorities, patient adherence trails far behind cost efficiency, supply chain resilience, and regulatory compliance. The gap between patient adherence and cost efficiency is striking and emblematic of the tension between industry rhetoric and the imperatives
“
THE LOW RANKING OF PATIENT ADHERENCE is especially telling. It reveals a structural disconnect we frequently observe: teams deeply care about patients, but the systems they operate in reward operational efficiency over human outcomes.
— Johanna Faigelman, HumanBranding
that shape actual investments,” says Vusi Moyo, Deputy Managing Director at APCO, an independent global advisory, public affairs, and strategic communications consultancy.
When asked which packaging innovations they’re currently exploring or adopting, respondents most frequently cited smart packaging technologies—such as NFC- and RFID-enabled solutions—at 73%. Recyclable or compostable materials followed at 57% each, reflecting strong interest in sustainability. Child-resistant packaging tied with recyclable materials at 57% and dose-controlled packaging at 34%, highlighting continued attention to patient safety and precision in drug delivery.
“The prioritization of smart packaging reflects a broader shift we’re seeing: Companies are moving from treating packaging as a static container toward treating it as an information touchpoint,” says Johanna Faigelman of HumanBranding. “What’s most interesting is that sustainability and child safety sit right below it, suggesting that manufacturers aren’t choosing between ‘innovation’ and ‘responsibility.’ Instead, they’re trying to solve multiple behavioral challenges at once: trust, safety, transparency, and waste reduction.”
What challenges are you facing when implementing new packaging solutions? (Select all that apply)
Material
Regulatory hurdles
Cost
Manufacturing
Supply chain limitations
Customer/patient
While the interest and drive is there to adopt new packaging solutions, there are, of course, challenges. The #1 challenge is material availability, followed closely by regulatory hurdles.
Compliance with stringent regulatory standards is a foundational pillar for all pharmaceutical packaging initiatives. “The challenges related to regulatory hurdles, restrictions, and approval delays are entirely understandable, given the need to ensure product safety and efficacy across diverse global markets,” says Moyo of APCO. “These findings underscore the necessity of a ‘glocal’ approach, or maintaining a broad global perspective, while being deeply attuned to the nuances and requirements of local markets, effortlessly navigating the patchwork of regulations, ensuring timely access to new therapies and packaging innovations worldwide.”
When asked how pharmaceutical packaging will change over the next 3-5 years, reader responses included more focus on sustainability, smart packaging, and personalized options.
“Over the next 3–5 years, I believe pharmaceutical packaging will undergo significant transformation driven by a mix of technology, sustainability, and patient-centric design,” said one reader from a global pharmaceutical brand.
“It will likely be characterized by increased focus on sustainability, smart packaging, and personalized medicine,” said a reader from an American multinational healthcare company.
“Manufacturers may be thinking more about sustainability—as this is currently an afterthought,” said one reader from a global pharmaceutical brand.
“It will move to patient-specific packaging. Unique SKUs for patients that are tied to their physician's database so that real time data can be shared about patients and treatments,” said one reader from a global biopharmaceutical company.
Which area(s) of digitalization do you believe will grow the most in the coming year? (Select all that apply)
The Future of Pharma
Over the past five years, pharmaceutical packaging has evolved.
First, packaging is becoming more intelligent: Technologies such as tamper-evidence, anti-counterfeit serialization, and smart labels or QR codes for traceability have gained ground.
Second, manufacturers are increasingly using reduced-material packaging, optimizing barrier films and lightweighting, and adopting take-back programs.
Third, packaging designs are becoming more patient-centric and flexible: from unit-dose and blister formats that improve adherence to modular and customizable packaging runs that accommodate smaller batches.
Finally, the rise of biologics, advanced therapies, and global distribution has meant that packaging must meet more stringent requirements for stability, cold chain, and barrier protection—leading to new materials and formats.
“
I’M NOT SURPRISED TO SEE THAT 63% of companies already have a digitalization strategy. As I talk with iA customers and others in the pharmacy industry, the general consensus is that embracing new technologies is an essential part of our strategy moving forward.
— Tom Utech, CEO of iA
Digitalization
The adoption of digital tools in healthcare manufacturing is transforming how medical devices, pharmaceuticals, and healthcare packaging are designed, produced, and monitored. The primary purpose is to improve efficiency, quality, compliance, and patient safety, though, it’s not surprising that 63% of respondents have a digitalization strategy.
The #1 motivation behind adopting digital tools is to reduce human error.
The No. 1 area where companies anticipate growth is data analytics, followed by AI, and predictive maintenance.
EVERY PACKAGE TELLS A STORY.
NOT JUST CO PLIANCE, CONFIDENCE.
Today’s pharmaceutical environment demands more than compliance. It demands visibility into every unit, control at every step, and assurance in every delivery.
End-to-End Serialization & Aggregation
Global regulatory readiness with centralized control.
AI-Driven Automated Inspection
High-speed defect detection for vials, syringes, and cartons.
Real-Time Production Intelligence
Actionable line data to improve uptime and reduce operational costs.
CONFIDENCE YOU CAN COUNT ON.
Scan to evaluate your packaging readiness. AntaresVisionGroup.com




Intelligent Data Capture: Paving the Path to a Modern Healthcare Supply Chain
This focus on standardization is helping reduce integration complexity— an ongoing challenge for many healthcare organizations.
By Liz Cuneo, Editor-in-Chief
Intelligent data capture—powered by 2D barcodes and global standards—is transforming logistics operations across industries.
KKEY TAKEAWAYS
Moving from 1D to 2D barcodes requires more than just buying new scanners. Manufacturers must update printers, imagers, and software.
nowledge is power. And within the healthcare supply chain, knowledge is known as data. But one cannot gain access to data without proper data capture. A recent GS1 US report offers companies a roadmap to modernization through intelligent data capture, highlighting how automation and data-driven systems can improve transparency, speed, and efficiency. The report, “Advancing Logistics Operations Through Intelligent Data Capture,” explores how intelligent data capture— powered by two-dimensional (2D) barcodes and global standards—is transforming logistics operations across industries.

Yet despite industry momentum, many organizations still face hurdles when updating legacy systems, especially in the healthcare space. To better understand these challenges and opportunities, I interviewed Tracy Nasarenko, Vice President of Customer Success for Healthcare at GS1 US. With more than two decades of experience in pharmaceutical manufacturing, Nasarenko provides insight into what it takes to transition from traditional barcodes to intelligent, connected systems that support end-to-end visibility.
Breaking Free from Legacy Systems
According to Nasarenko, two of the biggest barriers to modernization are cost and people. “Legacy systems tend to be deeply embedded in processes,” she explains. “That makes change complex. The most successful transitions begin with a thorough assessment—understanding how updating a system will impact every connected process.”
Mapping out every affected system and process is essential, as the DSCSA demonstrates the value of cross-supply-chain collaboration.
While many companies recognize the need to upgrade, hesitation often stems from the human side of transformation. “People are comfortable with what’s familiar,” she adds. “Even when the benefits are clear, change can be daunting.”
Moving from 1D to 2D barcodes requires some change, and it’s more than just buying new scanners. Manufacturers must update printers, imagers, and software—ensuring that every connected system can handle additional data points, such as lot numbers, batch codes, and expiration dates.
“These 2D barcodes allow companies to include far more information in a small space,” says Nasarenko. “But that introduces dynamic data, which must be managed seamlessly throughout packaging and distribution.”
For the pharmaceutical sector, this shift is also regulatory. The Drug Supply Chain Security Act (DSCSA) mandates four product identifiers—GTIN, lot number, batch number, and expiration date—encoded within a 2D data matrix. Similarly, the Unique Device Identification (UDI) rule governs medical device labeling, emphasizing traceability and accurate identification.
Cross-Sector Challenges and Collaboration
The overlap between pharmaceuticals and medical devices adds complexity for manufacturers producing both. “Companies like Johnson & Johnson that manufacture both prescription and over-the-counter products have extended 2D capabilities across
Courtesy: GS1 US
Tracy Nasarenko is the Vice President of Customer Success, Healthcare, GS1 US.

Courtesy: stevecoleimages, Getty Images
To prevent downtime or relabeling errors during the shift, companies must manage dynamic data in real time. Nasarenko emphasizes that success depends on process design. “Processes must be designed to support those updates seamlessly,” she says. “In my experience, this often means adjusting workflows, staffing, and approval processes to ensure smooth transitions within packaging operations. The goal is to structure the system to realize the full benefits of the investment.”
A Roadmap for Modernization
According to Nasarenko, a typical modernization roadmap begins with defining objectives—whether improving stakeholder data exchange or internal efficiency. “The first step is always to define what you’re trying to achieve. Is it greater data exchange with stakeholders or improved internal efficiency? Your stakeholders should be involved from the start,” she says. “Projects fail when they happen in silos.”
all products for consistency,” Nasarenko notes. “But when some product categories face lighter regulation, uneven adoption can create roadblocks.”
Mapping out every affected system and process is essential, as the DSCSA demonstrates the value of cross-supply-chain collaboration. “When issues arise, no single party can fix them alone,” Nasarenko explains. “Manufacturers, hospitals, distributors, and tech providers must align on data needs and accessibility.”

The Drug Supply Chain Security Act mandates four product identifiers—GTIN, lot number, batch number, and expiration date— encoded within a 2D data matrix.
Through GS1’s Solution Provider Partner Program, technology providers are increasingly aligning their products with global standards. “We’re seeing strong engagement from software vendors, vision system developers, and printer manufacturers,” says Nasarenko. “By building systems on standardized frameworks, integration with ERP, warehouse, and quality systems becomes far simpler.”
This focus on standardization is helping to reduce integration complexity—an ongoing challenge for many healthcare organizations.
Technology alone isn’t enough. As Nasarenko said, one of the biggest barriers to modernization is people. That’s why workforce training and change management are critical components of intelligent data capture initiatives. “I come from a Lean background, where employee engagement is central,” she says. “When employees understand the ‘why’ behind a change and feel ownership, adoption rates rise dramatically.”
Too often, companies invest heavily in new tools but overlook the human factor. “It’s not just about learning to use a new scanner,” Nasarenko adds. “It’s about understanding how that technology improves accuracy, efficiency, and—most importantly—patient safety.”
Beyond Compliance:
Building a Data-Driven Future
Nasarenko cautions that companies should look beyond compliance when modernizing. For example, the upcoming expansion of the National Drug Code (NDC) from 10 to 12 digits will require major system updates.
“Changes like this can be catalysts for broader transformation,” she says. “When companies modernize, they should aim to improve data accuracy, collaboration, and overall efficiency— not just meet regulatory requirements.”
The healthcare supply chain is increasingly data centric. As Nasarenko put it, “One of the major takeaways from DSCSA is that companies aren’t just selling products anymore—they’re selling products plus data. Patients expect transparency, and that requires collaboration across the entire supply chain.”
As such, the path to intelligent data capture isn’t just about technology. It’s about people, processes, and purpose—working in harmony to deliver accurate, transparent, and actionable data. For the healthcare industry, these efforts ultimately translate into one critical outcome: better patient care.


























































Engineering for Integrity: How Evergen’s Ashim Moona Builds Packaging Systems That Protect Patients
In this Prominent People in Packaging profile, Moona shares two aspects of sustainability, the importance of learning the standards, and how “burnout” can actually be a good thing.
By Liz Cuneo, Editor-in-Chief
For Ashim Moona, packaging engineer at Evergen, the field of medical device packaging is far more than cartons, trays, and pouches. It is a tightly regulated discipline defined by ISO 11607, ASTM standards, process capability, validation activities, and—above all—the responsibility of protecting patients.
Patient care is the biggest why,” Moona says. “It has to be inherent to every single step of packaging development. If it’s not, you run into issues at the end-user stage—and you never want that in healthcare.”
With just over three years at Evergen, a global Contract Development and Manufacturing Organization (CDMO) focused on patient-first innovation, Moona has already become deeply embedded in the company’s new product development (NPD) pipeline. His journey into the field, however, began long before his first day on the job.
“I always knew I wanted to be an engineer,” he says. “In middle school I was learning Autodesk Inventor. I developed this big passion for 3D modeling and spatial visualization.”
After attending two summer camps in hospitals, this opened his eyes to a career in healthcare and different ways he can help serve patients. After starting college in aerospace engineering—and hitting burnout—he discovered the packaging systems and design program at Virginia Tech.
“It was engineering, design, materials, manufacturing,” Moona recalls. “My very first class had us redesign a soapbox in CAD. That moment I knew—I was back to doing what I loved.”
From there, everything aligned: his interest in engineering, his
desire to work in healthcare, and his talent for technical design converged into a clear career trajectory.
Inside Evergen’s NPD Workflow: Standards, Specs and Cross-Functional Pressure
Today, as part of Evergen’s operations engineering team, Moona supports packaging for multiple device programs. His work touches nearly every step of the NPD cycle.
“I work with R&D engineers, quality, regulatory—pretty much every function,” he says. “My job is to understand the packaging design requirements based on product needs, quality needs, and regulatory needs. For us that means ISO 11607-1 and 11607-2 are driving almost everything.”
Weekly cross-functional touchpoints are routine. “We’re always asking: what product iterations will impact packaging? What’s changing in the design history file? What do we need to update in our packaging specifications or process validations?”
While he’s not modeling trays himself, his background enables him to quickly evaluate supplier designs.
“I don’t build every 3D model anymore,” he explains. “But when a supplier sends a tray model, I can dissect it immediately because I’ve done that work. That experience really helps.”
Learning From the Work
One of Moona’s most formative technical challenges came early in his tenure: qualifying an additional sterile barrier pouch supplier amid ongoing supply chain disruptions. “Scoping that project was massive,” he says. “Meeting every requirement of ISO 11607 was overwhelming at first. I reme mber thinking,
‘Where do we even begin?’”
The effort required understanding seal strength testing, material characterization, transit simulation, aging studies, and sterile barrier integrity requirements.
“It became an evergreen teacher,” he says. “That project helped me understand not just the text of 11607-1 and 2, but how to apply them. I still reference those learnings on almost every packaging project I touch.”
The experience also taught him the value of asking questions. “People had gone through it before me,” he says. “Realizing that some work was already accomplished—especially on post-market packaging—was huge.”

For
Ashim
Moona, the field of medical device packaging is far more than cartons, trays, and pouches.
When asked how sustainability gets factored into medical design, Moona shared how it has two equally important dimensions.
1. Material & Environmental Sustainability
“When we talk sustainability, people think recyclable materials, circular economy,” he says. “That’s definitely part of it—especially in secondary and tertiary packaging.”
He cites RAPAC foams and molded pulp as materials that reduce environmental impact while offering protection in transit testing governed by ASTM D4169.
“Healthcare is stringent,” he adds. “Primary sterile packaging can’t always be recyclable. But where we can implement curbside-recyclable solutions, we absolutely should.”
2. Process Sustainability
The second, often overlooked dimension is operational. “Process sustainability is a huge factor,” Moona explains. “If you control the design from the earliest feasibility stage through mass manufacturing, it sets you up for long-term success.”
This includes ensuring proper tray tolerances, ergonomic loading forces, seal consistency, and operator usability—elements that directly affect process capability and packaging validation.
“You don’t want something so tight an operator can’t load it consistently,” he says. “That creates defects. Sustainability means
designing a process that can be repeated and improved.”
Advancing Patient Care Through Packaging Engineering
Packaging’s role in patient care is often underestimated.
“Everything we do—the shelf-life testing, the transit simulation, the sterile barrier checks—is about patient safety,” Moona says. “Packaging is part of the product.”
He points to a recent industry innovation: adhesive-coated Tyvek. “It reduces fiber tear, improves aseptic presentation, and— something I never thought about—reduces noise in the operating room. That can actually lower patient anxiety. It’s a perfect example of mechanical innovation having a human impact.”
Moona also highlights structural innovations such as paperboard inserts inside pouches, which stabilize products, reduce carbon emissions in transit, and streamline pre-surgical activation.
“Those solutions check so many boxes—performance, sustainability, usability, patient care,” he says. “That’s where the future is headed.”
Though early in his career, Moona offers grounded, technical guidance to students entering the field:
∙ Careers aren’t linear. “Burnout can be a sign you need a different direction.”
∙ Master the standards. “Understanding ISO 11607-1 and 2 puts you ahead immediately. Same with ASTM D4169 and D3A. These aren’t optional—everyone has to follow them.”
∙ Ask questions constantly. “People around you want to help. Take advantage of that.”
∙ Work through imposter syndrome. “Focus on your weaknesses and build from them.”
Above all, he encourages new engineers to embrace the learning curve. “I’ve only been in the industry three years,” he says, “but the potential I see—machine learning, better materials, smarter structural design—is incredibly exciting. Every improvement ultimately helps the patient. That’s what makes this work meaningful.”
Ashim Moona
This exclusive Leaders in Healthcare Packaging section includes profiles and product listings for a variety of companies serving the healthcare and pharmaceutical industry.
COMPANY INDEX
Amcor
Deerfield, Illinois

healthcare@amcor.com
amcor.com/healthcare
Healthcare packaging that makes a difference
Amcor Flexibles Healthcare North America is recognized globally as a premier supplier for medical and pharmaceutical packaging. The company has a broad technology base, specializing in coating, lamination, extrusion, thermoforming, and converting and printing technology. Amcor provides unparalleled customer focus, technical leadership, breakthrough innovation, responsive tailored solutions, industry-leading quality and the expertise to be your all-inclusive packaging partner.
Quality—in manufacturing and end-user experience
With eight ISO-certified facilities in North America, Amcor Flexibles Healthcare North America’s broad manufacturing network provides supply chain efficiency and continuity of supply. Our partnerships are built on a deep understanding of end-users to help you develop quality packaging that meets their needs. Our expertise helps reduce complexity and risk, increase efficiency, and ensure a safe, smoother and simpler route to market.
Partner with the packaging leader to enable innovation as your competitive advantage
Amcor has best-in-class R&D capabilities and teams to collaborate in making your pipeline a reality. Together we create better ways to move your business forward. From providing breakthrough innovation so you can proactively deliver solutions and rapid prototyping to validate usability to collaborating on more efficient production lines and supply chains, Amcor can help you enable scalable growth.




Anritsu Product Inspection & Detection
Elk Grove Village, IL

Anritsu Inspection & Detection is a global leader in precision product inspection and quality assurance solutions for the healthcare and pharmaceutical packaging industries. With decades of expertise in X-ray inspection, checkweighing, and metal detection technologies, Anritsu helps manufacturers safeguard product integrity, protect patients, and maintain regulatory compliance.
X-ray inspection is used to detect both foreign materials and quality defects. Anritsu advanced systems detect contaminants as well as missing tablets, compromised seals, and packaging defects even in foil or metallized packaging.
Precision checkweighers measure even the lightest products with sensitivity as fine as ±0.01 g, supporting dosage accuracy and compliance. Available in single or multi-lane configurations, they help prevent underfill, overfill, and product giveaway while processing thousands of units per hour.
Metal detectors identify ferrous and non-ferrous contaminants. Pharmaceutical-grade systems achieve high sensitivity levels while minimizing interference from static, vibration, and inverter noise common in production environments.
Additionally, Anritsu provides comprehensive support through our Performance 360° Service program, offering installation, training, preventative maintenance, and responsive technical service. This partnership approach helps manufacturers minimize downtime, improve operational efficiency, and maintain the highest standards of product quality.
From contaminant detection to weight verification, Anritsu Inspection & Detection delivers the confidence manufacturers need to protect both brand reputation and patient safety.




Antares Vision Group
Mount Laurel, NJ

Every pharmaceutical product tells a story of quality, safety, and trust. Antares Vision Group helps pharmaceutical manufacturers ensure that story is verified at every stage of production and across the global supply chain.
Antares Vision Group is a global provider of integrated inspection, serialization, traceability, and data management solutions that enhance product quality, protect patients, and support regulatory compliance. Our technologies connect physical products to secure digital identities, enabling companies to monitor, verify, and protect every product from production through patient use.
Our automated inspection systems deliver high-performance quality control for a wide range of pharmaceutical containers and dosage forms, including prefilled syringes, vials, and cartridges. Advanced vision technologies detect particles, cosmetic defects, and closure-integrity issues with exceptional precision, while intelligent handling systems help preserve product integrity at high production speeds.
Antares Vision Group is also a global leader in serialization, aggregation, and supply chain traceability, trusted by more than 80% of the world’s Top 20 pharmaceutical companies. Our solutions support global regulatory requirements, including DSCSA readiness, while improving visibility and resilience across pharmaceutical supply chains.
Antares Vision Group helps ensure every pharmaceutical product’s story is one of verified quality, trusted compliance, and patient safety from production line to patient.




BELL-MARK
Pine Brook, NJ
973-882-0202
connect@bell-mark.com
www.bell-mark.com

For over 65 years, BELL-MARK has been a global leader in the design and manufacture of innovative in-line coding and printing systems. Beyond its presence in the food, beverage, and converting industries, they focus heavily on the demanding and highly regulated medical device and pharmaceutical markets. By prioritizing engineering excellence and industry integration, BELL-MARK has become the standard for manufacturers seeking to bridge the gap between complex regulatory requirements and high-speed production efficiency.
BELL-MARK offers a diverse portfolio of printing solutions—including Piezo Inkjet, Thermal Inkjet, Thermal Transfer, and Flexographic systems—that integrate seamlessly into existing packaging lines. Users can print on a wide range of substrates, such as Tyvek, medical-grade paper, poly-films, foils, cartons and cases. With a wide range of solutions available, BELL-MARK ensures that every individual application is optimized for its unique substrate, line speed, and printing environment.
New innovations like the InteliJet HD3R Reel-to-Reel system and its push-button automated maintenance represent a new standard in package printing. Utilizing an in-house, print-on-demand workflow, users can pivot to market demands instantly, removing the burden of costly preprinted inventory that frequently becomes obsolete. This platform handles complex variable data and serialization with ease, ensuring high-density 2D barcodes meet the strictest UDI and EUMDR mandates. Through streamlined production and the elimination of downtime, these innovations provide significant cost savings and ensure the lowest total cost of ownership available.
BELL-MARK supports its global customer base through a network of factory-trained technicians and remote support capabilities, including recently expanded facilities in Europe. By focusing on precision, compliance, and operational efficiency, BELL-MARK remains a cornerstone of the healthcare packaging landscape, providing manufacturers the ability to deliver products with absolute confidence in the integrity of their package.


Chase-Logeman Corporation
Greensboro, North Carolina

Headquartered in North Carolina, Chase-Logeman Corporation has been designing and manufacturing monoblock filling and finishing equipment for more than 65 years. Built on a legacy of innovation and reliability, our systems deliver precision, efficiency, and performance for today’s demanding production environments.
To meet your exact production requirements, Chase-Logeman can integrate a complete production line to provide a fully turnkey process solution from filling through final closure.
Our Monoblock Filling, Stoppering, Capping, and Aluminum Sealing systems are engineered for fast, efficient multi-component changeover. Typical changeovers, including vial and stopper type, can be completed in less than 30 minutes. Our widely adopted “tool-free” design simplifies operation and is especially valued in sterile filling environments where efficiency and cleanliness are critical.
For containers requiring screw caps, our systems accommodate container sizes ranging from microtubes to 125 mL (4 oz). Eye dropper tips, dropper bulb closures, plugs, and caps are applied with precision to ensure a secure, leak-free package.
Each filling machine is controlled by an Allen-Bradley PLC with a full-color touchscreen HMI, providing intuitive operation and reliable process control. Hazardous-duty rated control packages are available to meet specific facility requirements.
Whether your application involves bottles with plugs and caps or vials with stoppers—filling volumes from microliters to 125 mL—Chase-Logeman brings decades of expertise to design, integrate, and support your complete filling line.



Colbert Packaging Corporation

Colbert Packaging is an independent, women- and family-owned business. As a premier provider of paperboard packaging for more than 65 years, we work each day to produce safe, smart and sustainable secondary packaging for our customers who include some of the biggest names in the pharmaceutical, healthcare and consumer goods markets. With extensive R&D capabilities, continual investment in the latest equipment and technology, and a solid reputation for excellence, Colbert Packaging provides customers with top quality products and services, in an environment where our employees find opportunity for career fulfillment.
Colbert Packaging is a dynamic high-volume producer of sustainable paper-based packaging. We produce offset and flexographic printed folding cartons, formed paper trays, pressure-sensitive roll labels and informational package inserts. Our in-house services include structural design, full-service prepress, paperboard sheeting, ink blending/matching and die making/cutting. We will help you meet your healthcare packaging goals, whether it’s an F=1 rating for child-resistant packaging, senior-friendly solutions, creative patient adherence features, or secure storage.
We’ll work with you to design earth-friendly, secondary packaging solutions that satisfy today’s sustainability requirements. Everything from right-sizing materials to 100% inline vision inspection for fully integrated quality control, sets you on a course to The Total Package. Add to the equation fully recyclable paperboard, along with water- and agri-based inks, coatings and adhesives, and you’ll have The Total Package in One Clean Carton®!




Columbia Machine
694-1501

Flexible Product Handling
Columbia’s conventional palletizers are the most flexible palletizing solutions available, with options that can simultaneously stack and stretchwrap a variety of package types including cases, trays, shrink bundles, display packs and more.
Industry Leading Standard Safety Features
Columbia’s standard safety package contains Category 3 electrical safety components, including dual circuit safety interlocks & full height light curtains. This safety package provides a fully guarded, Performance Level D palletizer to keep your employees safe while enhancing OEE and production demands.
Complete System Integration
Columbia Machine manufactures conventional high-speed, high-level, floor-level & robotic palletizers, load transfer stations and provides complete system design and integration. With the most flexible and modern palletizing solutions available, Columbia is the leading palletizer solutions provider in North America.
High Performance AND Sustainability
Columbia has a palletizing solution for virtually any application, each equipped with standard features designed to safely maximize OEE, throughput and uptime.




DT Engineering
Lebanon, Missouri
417-664-0120
contactus@dtengineering.com
www.dtengineering.com

DT Engineering delivers automation, robotics, and system integration solutions for medi cal device and pharmaceutical packaging. As regulations tighten and product complexity grows, we help manufacturers improve efficiency, quality, and reliability. Our flexible auto mation platforms scale seamlessly from early‑stage production to full commercial manu facturing, driven by modular tooling and adaptable control architectures. With Industry 4.0 connectivity, advanced material handling, and turnkey integration for assembly, packag ing, labeling, and inspection, we enable smarter, faster, and more compliant operations.




– A ProMach Product Brand

York, PA
717.848.3875
www.greydon.com Greydon@ProMachBuilt.com
Digital Printing Solutions for Healthcare Packaging
Greydon is a leading provider of integrated digital printing solutions for pharmaceutical, medical device, and healthcare packaging environments. Our high-resolution digital print systems enable manufacturers to eliminate pre-printed inventory, support late-stage customization, and maintain full regulatory compliance directly on the production line.
Our portfolio includes UV digital inkjet, inline flexographic, traversing thermal transfer, thermal inkjet, continuous inkjet, and code dating technologies. Designed for demanding healthcare applications, Greydon systems deliver precise, camera-readable 2D codes, serialized data, variable text, and graphics printed directly onto packaging substrates.
Built for modern packaging lines, our solutions support multiple SKUs, short runs, and rapid changeovers without sacrificing throughput. Inline and traversing configurations integrate seamlessly with blister packaging, flexible packaging, cartoners, and pouching systems, while ensuring compliance and compatibility with GS1 and HIBCC standards.
As part of ProMach Labeling & Coding, Greydon designs, manufactures, and integrates digital print solutions that strengthen traceability, improve operational agility, and support evolving healthcare packaging requirements.
Industries We Specialize In: Pharmaceutical, Nutraceutical, Medical Device, Personal Care, Animal Health, and Consumer Healthcare Packaging.



Harpak-ULMA Packaging
Taunton, MA
800.813.6644
info@harpak-ulma.com
www.harpak-ulma.com
Look to Harpak-ULMA Packaging to prototype, design, build, implement and maintain packaging automation solutions for today’s complex packaging landscape.
Harpak-ULMA packaging systems can handle all your medical primary and secondary packaging equipment requirements – from single components through completely automated systems. Our full-service solutions address installation, training, spare parts, service and customer support, while capabilities span robotics and automation, thermoforming, tray sealing, filling, flow pack, stretch, blister, skin pack, and vacuum. Our secondary equipment can prepare products for retail ready displays, create multipacks for bundling of products, and erect and load cartons to get your product out the door. We provide the total solution – from beginning to end, product to pallet – for the medical industry.
Our medical packaging solutions offer maximum product protection, hermetic sealing, the ability to customize the package shape, blister packs for retail sale, hygienic individual and multipacks, easy open options, and protection during transport and handling. These solutions meet the strictest standards required by the medical sector. We can meet them all, like ISO 11607, EN 868, ASTM D1585, ASTM F2097 and ASTM F3475-11. Packages are suitable for sterilization processes and maintain these conditions until opened. You can also expect to be in accordance with CFR 21 Part 11 for documentation.
We have in-process controls like vision inspection for product and printing, product in pack-age height detection, and automatic rejection. Process monitoring includes alarm conditions for critical parameters out of range like time, temperature and pressure. There’s also a data integrity option for track and trace of operator in the HMI, plus trending of critical parameters. When it comes to calibration, our critical parameter equipment comes calibrated from the factory, and access points are provided for routine calibration.

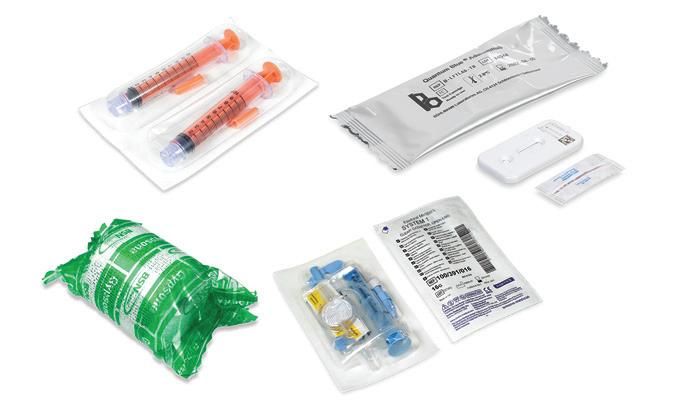




Heat and Control, Inc.
Hayward, California
1.800.610.9674
www.heatandcontrol.com info@heatandcontrol.com
Detect metal contaminants with confidence by choosing the world’s leading metal detection solution. CEIA®, is a leading innovator of industrial metal detection systems for products such as powders, capsules, tablets, and liquids. Quality control is at the core of CEIA development of the most advanced electronic and mechanical technologies for detecting contaminants accidentally present in products. CEIA metal detection systems meet high level industrial metal contaminant detection standards.
Find what you don’t see with the latest Ishida X-ray solutions. The photon counting dual energy IX-PD Series X-ray machine employs an alternative sensor and accompanying image processing technology to give our highest sensitivity and accuracy of low-density and minute foreign object contaminant detection. This technology differentiates with high accuracy between product and foreign objects, reducing the rate of erroneous detection. All models offer exceptionally sensitive foreign body contaminant detection and additional benefits such as the ability to identify damaged and missing products or components, helping the pharmaceutical industry achieve a rapid return on investment.
Providing safeguards that help ensure your products match the prescription. Anywhere along the line, protect your consumer and your equipment. Efficient detection of foreign objects is critical to consumer safety, brand survival, and will also protect machinery and prevent downtime. We offer a complete line of metal detectors, checkweighers, and X-ray inspection systems from our strategic partners: CEIA and Ishida.
Providing sales, service and spare parts expertise across the globe for metal detection and X-ray anywhere along a production line.



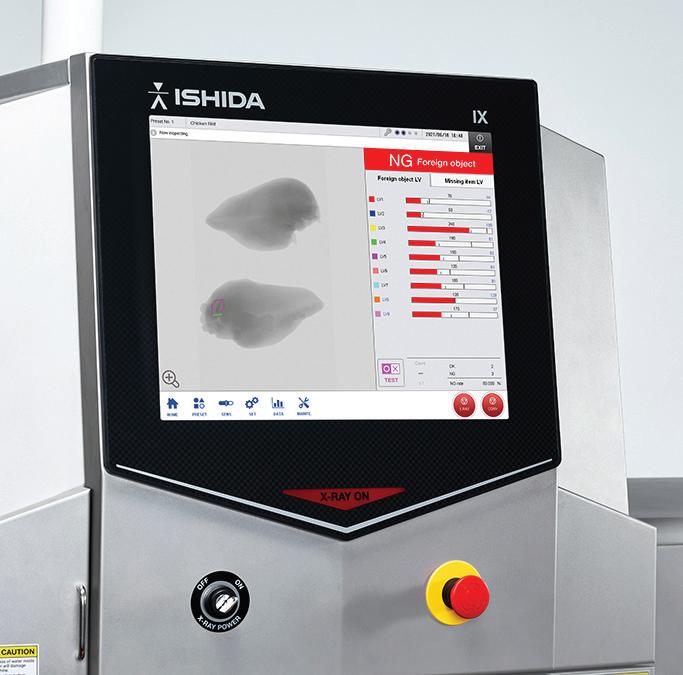
12420 Gold Flake Court, Rancho Cordova, California, USA
800.504.7120
sales@hightekusa.com
www.hightekusa.com
Automated Weighing & Packaging Equipment Designed Specifically for the Pharmaceutical and Nutraceutical Industries
Our High Tek USA MINI Weigher was designed to weigh and accurately dispense pharmaceutical and nutraceutical products, with accuracies down to one-hundredth of a gram.
Our automated pharmaceutical weighing and packaging Mini Weigher provides the speed and accuracy needed for efficient operation of packaging lines in the pharmaceutical industry, outperforming traditional methods for weighing and packaging Pharmaceuticals and Nutraceuticals.
We are experts in manufacturing our custom High Tek USA brand of Multi-head Weighers, Combination Weighers, Vertical Form Fill & Seal (VFFS) Baggers, Pre-made Pouch Machines, Rotary Pouch Machines, Inline Checkweighers and Checkscales, Scale Platforms, Bucket Elevators, Incline Conveyors, Take-away Conveyors, Rotary Pack-off Tables (Lazy Susans), High-Speed Scales & Baggers, Product Distribution & Seasoning Systems, and Stand-alone Combo Units.
We offer 24-hour technical support, parts, and service for all our equipment, along with preventive maintenance agreements.





James Alexander Corp.
Blairstown, NJ
908/362-9266
info@james-alexander.com
www.james-alexander.com
Located in northern New Jersey, James Alexander Corporation (JAC) is a leading contract manufacturer and custom filler of single-use crushable glass and plastic ampoules. Its manufacturing facility features unique, company-designed equipment and produces its patented plastic ampoules, among other product offerings.
James Alexander Corp. has expanded its operations floorspace, adding 18,000 square feet to its warehousing capacity and 2,000 feet apiece to manufacturing and office space. The expansion provides additional capacity for servicing key markets including pharmaceutical (OTC & Rx), medical devices, health & beauty products, first aid and diagnostics.
JAC’s patented single-use plastic ampoules, which have undergone various enhancements since their initial market introduction, are available in a variety of colors and with an array of applicators, offering singlehanded activation in a customizable format. Meanwhile, the company’s glass ampoules can be filled and assembled in single-use swab or dropper packages. JAC also recently introduced a winged device, THE ACTI-VATOR™, which provides easier activation for these glass formats. Other services include autoclave sterilization for glass ampoules, blister packaging and formula compounding.
Plastic Unit-Dose Dispensing Systems
James Alexander Corp.’s revolutionary plastic ampoule combines style and ease of use through single-handed activation. With just a gentle squeeze, the inner membrane ruptures, allowing the contents to be dispensed by the user. The plastic ampoule is available in sizes up to 5ml, as well as a range of colors and applicators.
Unit-Dose Glass Swabs
James Alexander Corp.’s unit-dose swabs offer the stability of glass in one- or two-part systems allowing for convenient application of pharmaceuticals and health aids. JAC also produces single-use glass ampoules for inhalation and dropper tip assemblies for the dispensing of liquids.



Massman Companies
Alexandria, MN
320.554.3611
sales@massman.com
www.massman.com

Massman Companies builds automated packaging machinery for healthcare, pharmaceutical, and nutraceutical manufacturers, delivering efficient, reliable, and scalable packaging lines for regulated environments.
Driven by a mission to provide the BEST-RUNNING MACHINES IN THE FACTORY, Massman Companies has the collective expertise to support packaging technologies across your production line while working with a single partner. This coordinated approach simplifies procurement, machine connectivity, and long-term service for healthcare packaging operations.
New England Machinery (NEM), a Massman Company, is a globally recognized packaging equipment specialist with more than 50 years of experience serving pharmaceutical and nutraceutical manufacturers. NEM’s pharmaceutical line includes container Unscramblers, Cappers, Orienters, Scoop Feeders, and Pluggers hygienically designed for cleanroom applications designed to meet your strict regulatory requirements in a small footprint.
Massman Automation and ADCO Manufacturing, also Massman Companies, further support healthcare packaging needs with secondary packaging and end-of-line solutions, including Cartoners and Case Packers designed for efficient and reliable operations across the line.




Metsä Board

Norwalk, CT
203.229.7480
www.metsagroup.com/metsaboard americas.metsaboard@metsagroup.com
Metsä Board is a leading producer of premium, lightweight paperboards made from fresh, renewable wood fiber. As part of Metsä Group, the company focuses exclusively on fresh‑fiber paperboard solutions designed to deliver high performance, material efficiency, and a lower environmental footprint across a wide range of demanding packaging applications.
Metsä Board’s portfolio includes folding boxboards, food service boards, barrier boards, and white kraftliners used globally in consumer, retail‑ready, food, and healthcare packag‑ ing. Manufactured using fully traceable raw materials from responsibly managed Nordic forests, Metsä Board paperboards are recyclable and produced with a high share of fossil‑ free energy, supporting the transition to a circular, low‑carbon economy. For pharmaceutical and healthcare packaging, Metsä Board offers a combination of purity, consistency, and reliability that is critical in regulated environments. Its fresh‑fiber paperboards are naturally clean and free from unknown substances often associated with recycled fibers, making them well suited for prescription and over‑the‑counter medicine packaging, nutraceuticals, and medical wellness products. The boards provide excellent surface quality for high‑resolution printing, 2D matrix coding, braille, and anti‑counterfeit‑ ing features, while maintaining shade consistency and visual uniformity across production runs.
Lightweight yet strong, Metsä Board paperboards support efficient converting and pack‑ ing operations, helping reduce material use without compromising protection or perfor‑ mance. This material efficiency not only improves operational performance but also contributes to meaningful reductions in packaging‑related carbon footprint—an increas‑ ingly important consideration for pharmaceutical brands managing Scope 3 emissions. Beyond materials, Metsä Board works closely with customers through expert support and fact‑based sustainability services, providing transparent data and life‑cycle insights to support informed packaging decisions. By combining fresh‑fiber purity, advanced material science, and close collaboration, Metsä Board helps brands meet high standards for safety, sustainability, and brand integrity—today and into the future.




METTLER TOLEDO

METTLER TOLEDO delivers proven product inspection solutions that help manufacturers protect their brands, profits, and productivity by reducing the risk of unsatisfactory products reaching the market.
Safeline metal detection systems are extremely sensitive, easy to use, and help prevent costly recalls by removing ferrous, non-ferrous and stainless-steel contaminants which can be introduced during processing.
Safeline X-ray inspection systems can detect contaminants including stone, glass, metal and dense plastics, as well as detect mass and check for missing or damaged product inside closed packages.
Hi-Speed checkweighers are available in a wide range of sizes and configurations to handle nearly any container type or package design at high speeds to ensure accurate product delivery, reduced giveaway, and an optimized production process.
CI-Vision vision systems perform accurate label and package quality inspections on all types of packaging, including label quality, 1D, 2D, and alphanumeric text, proper sealing, cap and lid placements, and tamper band presence.
ProdX™ software facilitates the collection and storage of production data across multiple lines and facilities. All data is easily accessible and exportable to other systems, facilitating compliance with all regulatory requirements.
Our wide range of solutions are supported by the most comprehensive engineering solutions, product testing, and training in the industry, with 24/7 telephone support and nationwide field service.




MG America, Inc.

Headquartered in Fairfield, New Jersey, MG America is a subsidiary of MG2 of Bologna Italy, a company that was founded in 1966 and today is one of the world’s three leading manufacturers of capsule filling equipment.
MG America is a leading supplier of an innovative family of precision-crafted processing and packaging machinery, including capsule fillers, material handling, sterilization equipment, primary packaging equipment, secondary packaging equipment, checkweighing/ weight control systems, tablet & capsule inspection, and line integration solutions. From sales, field service, and spare parts to machine trials and local service/support representation, MG America offers a true “Partnership for Success.”
Packaging equipment from MG America can be found throughout North America in industries such as pharmaceutical, medical device, diagnostics, nutritional products, and OTC products. Our lineup of premier, European made machinery has earned a global reputation for reliability, precise performance, and superior craftsmanship.




MULTIVAC Health Packaging
Kansas City, Missouri
800.800.8552
sales@multivac.com
www.multivac.com/us

MULTIVAC Health Packaging is your partner for medical and pharmaceutical packaging, labeling, inspection, and handling solutions.
MULTIVAC is one of the world’s leading suppliers of packaging solutions for a wide variety of medical and pharmaceutical products, as well as consumer and industrial goods. Our portfolio covers virtually all packaging design, performance, and resource efficiency requirements. In addition, it encompasses an extensive range of packaging technologies, automation solutions, and labeling and quality control systems.
All MULTIVAC equipment can be integrated into holistically designed systems thanks to our comprehensive line solutions. To ensure maximum line operating and process reliability, high efficiency, and low Total Cost of Ownership (TCO). With this in mind, we are consistently driving forward digitalization through real-time analysis tools such as MULTIVAC Smart Services, targeted towards predictive maintenance and machine learning. Thinking and acting sustainably, we are dedicated to the responsible use of resources and energy in terms of manufacturing our products and their use at your site.
MULTIVAC is your equipment and technology partner who makes a decisive contribution to your business success – whether you are a start up or a large company, when you decide on a MULTIVAC packaging solution, you opt for the highest level of quality and efficiency, comprehensive customer care, and perfect service. As a worldwide group with 85 subsidiaries, we are closely linked with our customers and their markets, allowing us to identify trends and developments early. As our customer, you benefit from our highly qualified sales and service team in your area.




Nita Sentient Labeling Systems
Waconia / Minnesota / USA - Terrebonne / Québec / Canada
855/668-6482
www.NitaLabeling.com Sales@NitaLabeling.com

Welcome to NITA Sentient Labeling Systems, the TRULY ORIGINAL100% Servo Inline and Rotary Labelers that dramatically REDUCE OPERATOR TOUCH-TIME and increase productivity, all while integrating the latest next-gen technology, the most intuitive user experience, best-in-class responsive support, and truthfully, pure labeling joy.
We love UPTIME. We’re so driven by it we’ve designed all the features on every NITA to create it. How? By meticulously analyzing your labeling pain points, then solving them, with innovation, technology, and flawless execution. And how do we keep you running? By building these incredible UPTIME features on every NITA:
· Parts ordering directly from the machine via built-in 3D drawings that scale down to your specific required item
· A self-diagnosing system that identifies possible issues and suggests fixes, or when parts need changing or servicing before they become a problem
· Ultra-fast & precise color-coded changeovers with in-screen 3D location maps or even our incredible Servo-driven FULLY AUTOMATIC product changeover system at the push of a button
· Daily Preventative Maintenance schedules and video tutorials with tiered-level tasks
· NitaCare Wi-Fi connectivity live remote tech support built right into your screen
Nita Labeling PROMISES to do anything and everything required to make your labeling life as easy and productive as it can be. It is why we exist. Experience for yourself what the best labeling company and labeler will bring to your productivity numbers.
For the love of labeling!
Your NITA Team




NJM, A ProMach Product Brand

NJM Packaging is a long‑established leader in automated pharmaceutical packaging and labeling systems, with more than a century of experience serving pharmaceutical, nutra ceutical, biotech, and personal care markets. The company offers a comprehensive portfo lio that spans stand‑alone machines to fully integrated turnkey packaging lines.
Their solutions cover every major function on a solid‑dose or liquid packaging line, includ ing bottle unscramblers, desiccant dispensers, tablet counters, cottoners, cappers, labelers, outserters, vial filling and closing systems (through partner Dara), track‑and‑trace/serializa tion systems, print‑and‑apply labelers, cartoners, bundlers, case packers, and palletizers. NJM is also known for its robust line integration expertise—designing custom layouts that combine ProMach equipment, third‑party machines, and existing assets into cohesive, high‑efficiency lines.
Built with cGMP design principles, stainless‑steel construction, and advanced controls, NJM equipment emphasizes reliability, accuracy, and long service life. As part of ProMach Pharma Solutions, NJM provides end‑to‑end support, from engineering and integration to aftermarket service and parts.




Pacteon Group

Pacteon Group delivers a responsive and integrated approach to complete packaging line solutions. Through the acquisition of companies with complementary products and shared values, Pacteon provides a unified portfolio that simplifies complexity across the packaging process.
Pacteon brands include Schneider Packaging Equipment, Phoenix Wrappers, Descon Conveyor Systems, and our pharmaceutical division, ESS Technologies.
ESS Technologies is a leader in advanced pharmaceutical and healthcare packaging solutions that prioritize efficiency, precision, compliance, and flexibility. Our equipment is designed to reduce labor demands, adapt seamlessly to evolving production needs, and maintain stringent quality standards with IQ/OQ documentation.
• Multi-format cartoners: Engineered for precision handling of bottles, tubes, pouches, cartridges, vials, and more—our cartoners deliver exceptional accuracy across formats. Their flexibility supports fast changeovers, short runs, and minimal downtime.
• High performance case packers: Available in top-load, side-load, and bottom-load configurations, our case packers integrate seamlessly with serialization, track-and-trace technologies, product inspection and rejection systems, and leaflet placement.
• Scalable palletizing systems: From collaborative palletizing solutions and ultra-compact modular cells to fully integrated systems with high-speed stretch wrappers, labelers, and AMRs, our palletizing lineup scales effortlessly with your operational needs.




Pfeiffer Vacuum+Fab Solutions

Pfeiffer Vacuum+Fab Solutions, part of the Busch Group, provides advanced leak detection equipment for the medical and pharmaceutical industries. Leak testing plays a critical role in ensuring the safety and integrity of medical devices and pharmaceutical packaging, particularly for applications requiring reliable container closure integrity testing (CCIT).
With decades of experience supporting medical manufacturers and research institutions worldwide, Pfeiffer Vacuum+Fab Solutions delivers tailored vacuum and leak detection solutions designed for highly sensitive healthcare applications. The company’s portfolio supports CCIT and device testing using three deterministic technologies: mass extraction, helium mass spectrometry, and optical emission spectroscopy.
From development through production, Pfeiffer Vacuum+Fab Solutions offers highsensitivity, non-destructive CCIT solutions capable of detecting extremely small leaks in container closure systems. These technologies help manufacturers maintain sterile barrier integrity, meet global regulatory requirements, and ensure consistent pharmaceutical product quality.
For more information about our CCIT solutions, visit www.pfeiffervacuum.com under Solutions. www.pfeiffervacuum.com




Pharmaworks, A ProMach Product Brand
Odessa, FL
www.Pharmaworks.com Sales@Pharmaworks.com 727.232.8200

Pharmaworks, part of ProMach Pharma Solutions, is the leading North American manu facturer of pharmaceutical blister machines, known for delivering innovative, flexible, and dependable packaging solutions. With more than 20 years of experience, the company designs and builds a full range of thermoforming blister machines—from semi‑automatic units to high‑output systems—as well as cartoners, feeding systems, and product handling equipment.
Beyond new machinery, Pharmaworks is also a major provider of rebuilds and upgrades for many OEM blister machines and cartoners, offering controls modernization, full refurbish ments, and tooling solutions for both its own platforms and third‑party equipment. Their portfolio extends into advanced vision inspection systems, including exclusive distribution of Scanware technologies in the U.S. and Canada.
Serving pharmaceutical, nutraceutical, medical device, and consumer product markets, Pharmaworks combines engineering expertise with operator‑focused design to deliver reliable performance, fast changeovers, and cGMP‑compliant packaging lines.



PMI KYOTO Packaging Systems
Elk Grove Village, IL 60007 USA
847-437-1427
sales@pmikyoto.com
www.pmikyoto.com
Precision Meets Speed
Based in Chicagoland since 1993, PMI KYOTO has worked with our customers to solve their hardest packaging challenges. Specializing in cartoning and case packing, our engineering and manufacturing teams at both our US and Japanese factories have the decades of experience required to provide a reliable end-of-line system that will meet your secondary packaging needs now and operate on your floor for decades.
From robust, high-speed cartoning of 200+ products per minute, flexible robotic systems that automate infeed and material loading, to innovative machines that reduce packaging material needs, our machines are built and assembled in Elk Grove Village, IL to meet your requirements. With internal machining and fabrication, our machines are truly made from the ground up by us. This means when in the future, if a spare part is needed and we don’t already have it in stock, our team can have it machined and shipped within 1-2 days, so production isn’t waiting around for a part to arrive from some far-off supplier.
We take pride and go the extra mile to make sure your machine meets and exceeds your expectations. Our user-focused philosophy of designing rugged, well-crafted machines that make life easy on operators and maintenance will assure your highest efficiencies with minimum cost of ownership while your machine runs for decades. Following our high production standards and engineered for easy future updates as production requirements change, it’s common to find PMI KYOTO machines running for 20 or more years. It’s the PMI KYOTO way.


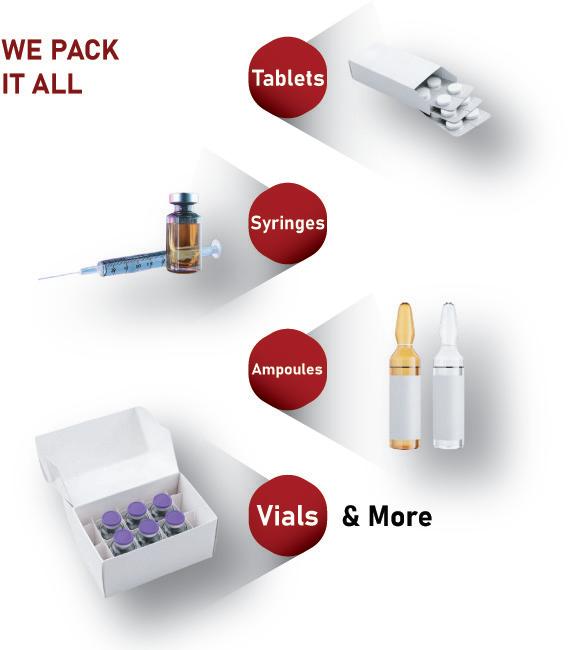

Missouri

ProSys is a premier manufacturer of semiautomatic and fully automatic equipment for filling, Squeeze Tubes, Syringes, Airless Pumps, Cartridges, Jars, Custom Containers & Hot Melt applications for the Health Care industry. A global supplier of filling equipment since 1985 with U.S. sales, manufacturing and customer service facilities located in Southwest Missouri and Durham North Carolina.
FEATURES & BENEFITS
• Fill Accuracy of +/- 0.1% by Volume
• Turnkey & Custom Designs
• Air-Free Vertical Bottom-Up Filling
• Custom Mix Solutions (Eliminates Batching)
• Drum & Pail Presses
• SERVO Solutions
• Explosion Proof Controls (Class 1 Division 1&2, ATEX 0&1)
• Tool-free Release System for Simple Changeovers
• “Digital Readout Indicators” for Fast & Accurate Adjustments
• Multiple Service Technicians for Less Down Time & Preventive Maintenance
• On-line Service & Support
• Recipe Storage & Recall
• Creams, Lotions & Viscous Pastes to 3 Million Centipoise
• Designed & Built in the U.S.A.




Sepha Ltd.
Harrison, OH
888-241-3179
info@sepha.com
https://sepha.com
Sepha provides a range of innovative leak testing solutions, deblistering machines, and small-scale blister packing machines for pharmaceutical and healthcare products.
CCIT & Leak Test Solutions
Sepha offers a range of non-destructive and deterministic CCIT & Leak Test solutions for various types of parenterals, blister packs and medical device packaging. Key features:
• Enable to identify defects as small as 1μm
• In line with ASTM standards & USP 1207 guidelines
• Can form part of 21 CFR Part 11 compliant system
Blister Packaging Solutions
Sepha offers a range of small scale blister packaging machines, that can handle thermoform and cold-form materials, to package tablets, capsules, medical devices and other products. Designed with full 21 CFR Part 11 capabilities. Ideal for:
• Clinical trials and stability studies
• Package development
• Low volume production of specialist drugs
Deblistering Solutions
Sepha provides a range of manual and automatic deblistering machines for the safe recovery of drugs from all blister pack types. Key characteristics:
• Suitable for push-through, child resistant and peelable blister packs
• Deblistering capacity up to 60 blister packs per minute
• Handles thermoform/alu and coldform/alu blisters
• No product or foil contamination




Simco-Ion
Hatfield, Pennsylvania
215.822.6401
customerservice@simco-ion.com
www.simco-ion.com

Simco‑Ion, an Illinois Tool Works (ITW) company, is the global leader in static control solu tions, delivering proven technologies that enhance product quality, process consistency, and contamination control throughout healthcare packaging operations. Since 1936, Simco‑Ion has engineered innovative ionization and particulate‑management systems designed to solve electrostatic challenges across highly regulated, clean manufacturing environments.
With decades of research and field expertise, Simco‑Ion provides a comprehensive port folio of static neutralizing bars, ionizing blowers, air guns, web cleaners, and monitoring devices used to optimize medical and pharmaceutical packaging lines. These solutions help reduce particle attraction, support sterility requirements, improve film and material handling, and minimize defects caused by electrostatic charge—critical factors for health care packaging where product integrity and compliance are paramount.
Serving industries that include medical device, pharmaceutical, and sterile barrier packag ing, Simco‑Ion’s application specialists work closely with OEMs and end‑users to integrate tailored static‑control strategies into extrusion, converting, form‑fill‑seal, assembly, and cleanroom processes. With global manufacturing, rapid delivery, and a commitment to continuous innovation, Simco‑Ion provides reliable, high‑performance solutions that help healthcare packagers achieve greater safety, efficiency, and regulatory confidence.
Driven by close collaboration with customers, Simco‑Ion continually develops new prod ucts and application‑specific innovations to address emerging challenges in healthcare packaging. This customer‑focused development process enables healthcare packaging teams to maintain precision, cleanliness, and consistency as their production environ ments grow more sophisticated.




Starview Packaging Machinery

Starview Packaging Machinery, Inc. is the leading manufacturer of packaging machinery for high-visibility packaging and medical device packaging with over 30 years of providing standard and custom packaging systems to our customers and distributors.
We engineer, design, and manufacture a comprehensive line of manual, semi-automatic, and automatic sealing machines. Available in shuttle, rotary, carousel, and inline configurations, with a variety of standard and custom sealing areas to meet exact requirements and maximize productivity. Machines are configured to suit specific applications such as retailcarded packages, sterile medical device packages, pharmaceutical wallet packages, OTC pharmaceutical packages and food tray packaging.
Starview offers many value-added features for our machines such as: marking / printing systems, vision verification systems , robotic product loading, product detection / sensing, automatic packaging materials loading, automatic inline fold-over, finished package unloading with reject feature for non-compliant packages, RFID tooling identification and hydropneumatic cold seal presses.
Machines are offered worldwide through a network of Authorized Distributors who provide sales and service as well as the packaging materials used in conjunction with our machines.
Leading the industry with the most complete and innovative packaging machines for: Blister & Clamshell Packaging, Medical & Pharmaceutical Packaging, Systems with Automation & Integration, Food Tray Packaging, Skin Packaging & Die Cutting, Stretch Pak Packaging, Thermoforming Equipment, Customized Packaging Equipment.





Uhlmann Packaging Systems L.P.
Towaco, NJ
(973) 402-8855
www.uhlmannpackaging.com info@uhlmann-usa.com
How Intelligent Packaging Design Elevates Parenteral Delivery
Packaging is key to ensuring the integrity and safe delivery of life saving parenteral products. Uhlmann Pac-Systeme demonstrates how deep design competence turns packaging challenges into patient-centric solutions.
As the pharmaceutical market sees unprecedented growth in parenteral drugs, especially self-administration devices like injector pens, new challenges arise. Packaging must guarantee product integrity, securing glass vials, pre-filled syringes, and complex pen devices against shock and vibration-all while ensuring perfect safety and ease of handling. In addition, material selection becomes a critical balancing act, driving shifts toward recyclable mono-materials or fiber-based trays.
The Solution: Integrating Design and Automation
These complexities require holistic approaches that seamlessly integrate packaging design with machine performance. Effective solutions emerge when manufacturers partner with packaging experts possessing both design acumen and deep engineering knowledge.
Uhlmann’s specialized competence centers allow custom packaging concepts-including features like intuitive thumb-push openings, clear perforations, and tamper-evident cartons-to be prototyped, tested, and validated on production-level machinery, guaranteeing seamless transitions from initial ideas to full-scale manufacturing.
By merging automation with profound design expertise, Uhlmann delivers secure, efficient, user-friendly solutions, that bring vital medicines to patients safely and reliably.




www.wipotec.com/us

Wipotec is a global leader in innovative weighing and inspection technology, delivering exceptional accuracy, speed, and reliability to the pharmaceutical industry. Our solutions provide 100% in-line control, ensuring product safety and full regulatory compliance.
At the core is our TQS (Traceable Quality System) – a modular Track & Trace solution offering serialization and aggregation to meet strict global regulations. With TQS, each package is traceable back to the manufacturer, helping combat counterfeiting and ensuring process integrity.
Wipotec systems are designed with compact, modular architecture, allowing easy integration into existing packaging lines. All machines meet stringent hygiene and validation standards, making them ideal for cleanroom environments.
Our high-precision EMFR Weigh Cells enable dynamic weighing at high speeds, used for individual tablet weighing, completeness checks, and verifying package contents—including leaflets and inserts. Wipotec checkweighers, such as the HC-A series, achieve throughputs of up to 600 units per minute with unmatched accuracy.
Key features include:
• Intuitive ConfigureFast interface for easy setup and fast changeovers
• Open XML interfaces for seamless integration with Level 3 systems
• Secure remote service tools for efficient troubleshooting and minimal downtime
With 85% in-house production, Wipotec ensures rapid delivery, high quality, and tailored solutions. Trusted worldwide, we bring precision, speed, and safety to every stage of your pharmaceutical production.




WLS, A ProMach Product Brand

WLS (Weiler Labeling Systems) is a leading manufacturer of advanced pressure‑sensitive labeling and label‑printing solutions, serving pharmaceutical, biotech, medical device, and other regulated industries. With more than 30 years of experience, WLS designs, builds, integrates, and supports high‑performance rotary and in‑line labeling systems known for exceptional precision, reliability, and long service life.
Their portfolio includes:
• Plunger rod insertion + labeling systems, including award‑winning NEW technologies
• Rotary and in‑line pressure‑sensitive labelers for bottles, vials, syringes, and other containers
• Label printing systems for high‑accuracy serialization and variable data
• Device stabilization and specialty labeling solutions for complex formats
WLS is recognized for engineering excellence, robust construction, and deep expertise in pharmaceutical compliance. As part of ProMach Pharma Solutions, they also provide full lifecycle support—parts, service, integration, and customization—to ensure maximum uptime and performance.









Amcor HealthCare™ Thermoformed Trays
Packaging engineered to protect with customized solutions to fit all your healthcare packaging needs, comprehensive material choice, and award winning design service. Learn more about precision packaging you can trust at amcor.com/products/healthcare

Every Pharmaceutical Product Tells a Story
From automated inspection to end-to-end serialization, Antares Vision Group ensures the quality, safety, and traceability of every pharmaceutical package - from production line to patient. Discover more: AntaresVisionGroup.com

BELL-MARK
Print Entire Rolls of Film On Demand & Eliminate Preprinted Inventory
Take complete control of your production line with the InteliJet HD 3R Reel-To-Reel Printer. Print your entire rolls of film, including die-cut labels, in-house, in full-color CMYK. Reduce your lead times for preprinted materials from weeks to minutes with the ability to print exact quantities anytime. ZERO waste!

Chase-Logeman Corporation
Innovative Monoblock Fill-Finish Equipment
Chase-Logeman manufactures innovative fill-finish equipment solutions providing equipment that transforms once-manual processes into efficient, automated operations. Our systems increase productivity, reduce waste, and enhance workplace safety for manufacturers across a wide range of industries.

We know the most important thing is what goes INSIDE. Colbert PaCkaging
Independent and women-owned Colbert Packaging provides safe, smart and sustainable paperboard packaging for healthcare and consumer goods. With our expertise in folding cartons, package inserts and labels, what packaging challenge can we help you solve today?

AntAres Vision Group
47




DT EnginEEring
Automation That Adapts as Fast as Healthcare Changes
DT Engineering delivers scalable automation solutions built for modern medical device and pharmaceutical packaging. We combine robotics, end-of-arm tooling, industrial automation, material handling, and smart system integration to boost efficiency and precision across regulated environments.

The Greydon C-MAX integrates UV-cured digital inkjet directly into your continuous web line, delivering high-resolution 2D codes, text, graphics, and variable data without pre-printed inventory. Achieve compliant, production-ready output for pharmaceutical and medical device packaging.

Harpak-ULMa packaging
Reliable. Reproducible. Traceable.
Validation-ready product to pallet automated medical packaging solutions that meet all required standards. Maximum product protection, hermetic sealing, customizable package shapes, hygienic individual or multi-packs, and easy open options.

Heat and Control, InC. See our profile on page 55
Inspection Solutions: Metal Detection and X-ray
Take advantage of the world’s only multi-spectrum metal detection technology with CEIA®, and have peace of mind that your products are thoroughly inspected for the full spectrum of foreign bodies with Ishida.

ALEXANDER CORPORATION See our profile on page 57
We’ll help you develop a customized dispensing system for your formulations with a variety of tips and in a range of different sizes/colors. You get: Extended shelf life, formulation stability, tamper resistance, quality assurance and two-part mixing. PROUDLY MADE IN THE USA. www.james-alexander.com

Digital Full-Color Blister Film Printing at Production Speed
Greydon – A ProMAch Product BrAnd See our profile on page 53
JAMES




Purpose-Built Systems for Regulated Environments
From container unscramblers and orienting systems to cappers, scoop feeders, and plug applicators, NEM, a Massman Company, provides integrated solutions that support accuracy, efficiency, and product integrity. Explore our machines at neminc.com
Designed for Strength, Precision and Peace of Mind
Pack with Confidence - Pharmaceutical integrity starts with the box. Metsä Board provides traceable, fresh fiber packaging engineered for uncompromising hygiene and strength. Our lightweight solutions ensure secure supply chains and clear, compliant labeling - all while reducing your carbon footprint.
METTLER TOLEDO

ProdX: Providing The Next Level in Product Safety
ProdX is a complete inspection data management and connectivity software solution that delivers full digital management of your product inspection equipment for real-time monitoring and food safety compliance. Save time, money, and keep your data safe. www.mt.com/prodX

Packaging equipment from MG America can be found throughout North America and across the life sciences industry. Our lineup of premier, European-made machinery has earned a global reputation for reliability, precise performance, and superior craftsmanship.

MULTIVAC Health Packaging provides customized solutions for healthcare packaging, labeling, inspection, handling, and more. In addition, our solutions encompass an extensive range of packaging technologies, automation solutions, digital services, and quality control systems.





NEW | BRONCO® 4 Versatile Pressure Sensitive Labeler
Flexible for every format. Smart, simplest operation. Future-proof and reliable.
Designed with you, not for you.
Flexible End-of-Line Automation Pacteon GrouP
ESS Technologies, A Pacteon Company, delivers high precision, flexibility, and cutting-edge features in a compact footprint.
• Pick and Place Robotics • Cartoners • Case Packers • Palletizers


Reliable CCIT solutions for pharmaceutical and medical packaging
With helium mass spectrometry, optical emission spectroscopy, and mass extraction technologies, our CCIT solutions provide high-sensitivity leak detection for critical pharmaceutical and medical packaging.

Leading Pharmaceutical Blister Machine Manufacturer
Innovation, flexibility, and dependability drive every blister packaging solution we deliver. Along with our own full line of blister machines and equipment, we also rebuild and upgrade systems from other OEMs.

ProSys is a premier manufacturer of semiautomatic and fully automatic equipment for filling, Squeeze Tubes, Syringes, Airless Pumps, Cartridges, Jars, Custom Containers & Hot Melt applications for the Pharmaceutical industry. FLEXIBLE, RELIABLE & ACCURATE
FILLING SYSTEMS






Is Static Compromising Your Product Quality?
The Phoenix 2.0 delivers powerful ionization to keep healthcare packaging lines efficient, stable, and debris‑free. Automatic ion balance and superior airflow ensure rapid static neutralization where precision matters most. Simple controls and quick‑clean features keep uptime high and contamination low.

The recently updated SB/PH1-1012 sealer now comes with a color HMI screen to give you full access to on screen heat and optional pressure calibrations. Includes exclusive data validation/data download port and class 100 clean room filter. Operational log is CFR21 Part 11 compliant

Packaging SyStemS l.P.
Next Level Packaging: Your parenteral journey
From concept to a running line-turnkey with a trusted partner. Experience next level packaging throughout the entire line. Uhlmann offers the right services, machines and digital solutions to turn complex challenges into patient-focused success.

The modular design allows customization for product size, weight, and speed with full system integration, and it scales for larger items like shipping cartons, including Track & Trace for serialization and aggregation. Smart Modular Design. Seamless Traceability.

WLS
Plunger Rod Insertion and Labeling
The NEW, patented PR-12 and PR-24 from WLS perform high-speed, high-control plunger rod insertion, and syringe labeling in one, single, compact-footprint unit. The PR from WLS provides a generational leap in technology, productivity, and control over prior plunger rod insertion technologies.

SB/PH1-1012 Updated Table Top Medical Device Tray Sealer
Starview Packaging Machinery inc. See our profile on page 72
Uhlmann
Wipotec
74


Cold Chain Insulated Shipper Contains 30% Recycled Content
ReGenX Technology
All Atlas DuraTherm insulated shippers are now being manufactured with ReGenX Technology.
ReGenX Technology represents a major step forward in sustainable material innovation for the cold chain and insulation industries.
Developed to conserve resources and reduce waste, ReGenX Technology closes the loop on product life cycles, turning used materials into new, high-performance recycled content products.
Containing 30% recycled content, ReGenX enables the creation of more sustainable cold chain packaging and insulation products that perform as well as those made from virgin materials, at no added cost.
Ice Packs for Pharma Cold Chain Efficiency
Hydropac
Hydropac’s HydroFreeze is a next-gen water-based ice pack that delivers faster freezing, longer thaw protection, and improved sustainability for stricter cold chain requirements.
HydroFreeze is engineered to support pharmaceutical and life sciences logistics with greater thermal stability, faster conditioning, and enhanced environmental compliance.

As medical supply chains tighten and regulatory expectations evolve, the icepacks offer cold chain stakeholders a highperformance, low-impact solution to support strict temperature standards while reducing energy and waste.
Compared to the original HydroFreeze (water-based ice pack):
• 51% faster to freeze.
• 110% longer thaw protection.
• 34% reduction in freeze down time.

Auto Tensile Tester
Labthink
Labthink’s C610G is a multi-station system designed to perform a wide range of strength and seal integrity tests across flexible packaging, medical, and industrial materials.
The C610G Auto Tensile Tester is a significant advancement in material testing technology. Designed for applications involving plastic films, flexible packaging, adhesives, medical patches, rubber, and paper, this system provides a comprehensive, all-in-one solution. It supports an extensive range of tests, including tensile strength, peel strength, tear resistance, heat seal performance, and puncture force. Equipped with Labthink’s latest innovation, Multi-axis Positioning Technology, the system enables all six testing stations to operate simultaneously.
Intermittent Cartoner
ELMACH
ELMACH’s WKH-100 Intermittent Motion Horizontal Cartoner is a servo-driven system that offers precise, reliable, and GMP-compliant cartoning for bottles, blisters, and tubes.

The WKH-100 is engineered for reliable and versatile cartoning applications across the pharmaceutical and cosmetic industries. Designed to accommodate a wide range of packaging needs, it combines servo-driven precision, modular construction, and userfriendly operation in a compact footprint.
It is equipped to handle bottles, blisters, tubes, and other solid-dose formats, providing manufacturers with high flexibility for diverse product portfolios. Its intelligent in-built pre-breaking carton erection system ensures consistent carton opening, while mechanical transfer and product insertion guarantee stable operation and accurate placement at high speeds.
Sustainable Blister Packaging Machine
IWK Packaging Systems, Inc.
The IWK CABLIblue 870 Blister System features “card-to-card” blister technology in which both the cardboard blister and its backing are not only securely sealed but comprise the same material. The result is a robust packaging solution that is 100% recyclable in paperboard streams—a premium selling point as companies continue to embrace sustainability. Featuring a modular design easily reconfigured to suit various blister shapes and sizes, the IWK CABLIblue 870 can produce as many as 22 large-capacity blister packages per minute.

VRC Solutions Driving Uptime and Process Efficiency
PFlow Industries
PFlow Industries highlights its VRCs for the pharmaceutical sector.
Material handling equipment, including the PFlow M Series and F Series, are engineered specifically to enhance efficiency, safety, and uptime in pharmaceutical manufacturing and distribution environments.

The PFlow M Series 2-Post Mechanical Material Lift offers versatility, supporting high-speed, high-cycle, and automated environments. Capable of lifting up to 10,000 pounds at speeds up to 400 feet per minute, the M Series moves materials fast—and safely.
Its ability to reach multiple levels, advanced safety features compliant with ASME B20.1, and custom construction options, including straddle and cantilever designs, make it adaptable for seamless, controlled workflows that reduce manual material handling.
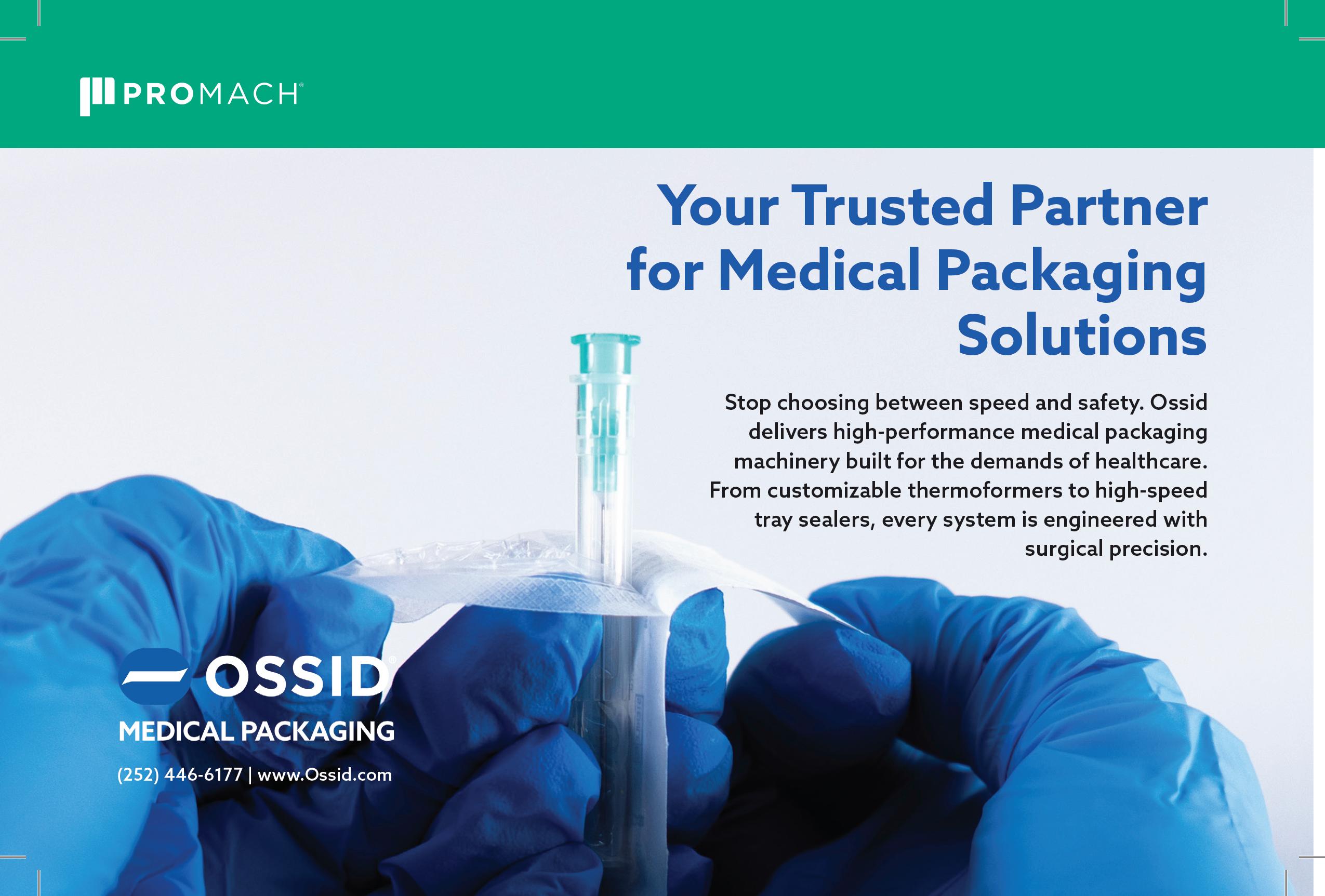
Tethered Cap
Sanner
Sanner expands its sustainable effervescent tablet packaging portfolio with a new Tethered Cap that improves recyclability without disrupting existing filling processes.

Together with Sanner BioBase, the solution adds to the company’s portfolio for sustainable effervescent tablet packaging. The Tethered Cap stays attached, enabling improved recycling and thus a more environmentally friendly overall product.
Manufacturers of effervescent supplement tablets are increasingly confronted with rising demands regarding the sustainability of their products and packaging. The issue also plays an increasingly important role for consumers. This is where Sanner’s new Tethered Cap closure comes in: The TE safety ring remains attached to the cap. This means that all plastic components can be recycled, reducing the environmental impact caused by microplastics.
Pharmaceutical Systems for Advanced Septic Manufacturing
Shibuya Corporation
Shibuya Corporation’s systems include integrated isolators, sterilization technologies, and filling solutions that deliver sterility, reliability, and efficiency for global pharma production.

Shibuya’s pharmaceutical systems encompass a broad portfolio of Sterility Testing Isolators, isolator-based containment systems for high-potency products, such as oncology products, as well as Isolator Systems for all aseptic manufacturing applications. Shibuya’s aseptic processing is engineered to ensure state-of-the-art sterility assurance fully compliant with EU Annex 1 and global cGMP expectations. Shibuya isolators provide optimum air particulate quality and environmental decontamination for applications such as parenteral injections, vaccines, combination products, cell culture, and cytotoxic compounding.
To create state-of-the-art control of microbiological risk, Shibuya provides Sterilization and Decontamination Systems, utilizing fully integrated Vapor Phase Hydrogen Peroxide Decontamination Systems for isolators, closed RABs, and Cleanroom Decontamination Systems. These solutions leverage advanced Shibuya hydrogen peroxide vapor and other proven sterilization/decontamination technologies to provide reliable microbial inactivation for both equipment and packaging materials.
In addition, Shibuya offers one of the most comprehensive ranges of pharmaceutical filling systems in the world, supporting Vial Filling Systems, Ampoule Filling Systems, Syringe Filling Systems, Nested Syringe and Vial Filling Systems, Drop-Dose Solution Filling Systems, Lyophilized product manufacturing, and Aseptic powder filling in a variety of glass, plastic, or flexible packaging systems.

Smart Shrink Sleeve Label
Sleever and Avery Dennison
Sleever and Avery Dennison launch INVENTRA, an intelligent shrink sleeve label with integrated RFID technology that enables item-level traceability, anti-counterfeiting, and inventory accuracy for beauty products without compromising design or efficiency.
Sleever and Avery Dennison launched INVENTRA in response to the rapid evolution of distribution models, rising regulatory requirements, and the growing number of product references in the beauty and makeup market. This intelligent product that integrates RFID technology into the shrink sleeve label is designed to support the sector’s future transformations.
INVENTRA reflects a shared conviction between the two partners: Packaging is set to become a strategic vector for data, traceability, and performance, without compromising the aesthetic or industrial standards of brands.

New Safety Syringe Launches in the U.S.
Terumo Medical Corporation
This retractable safety hypodermic line offers enhanced safety, reliability and ease of use while minimizing patient discomfort.
Terumo Medical Corporation, a wholly-owned subsidiary of Terumo Corporation, announces the U.S. launch of its SurTract Safety Syringe, powered by SafeR passive safety technology and developed by Roncadelle Operations S.r.l. The SurTract Syringe will be distributed across hospitals, clinics, and emergency-care settings beginning in 2026.
SafeR technology provides the SurTract Syringe with a fully passive, automatic retraction mechanism that withdraws the needle into the plunger immediately after use—helping reduce the risk of needle-stick injuries for physicians and providing patients with a smooth and comfortable injection due to less plunger pressure.
The syringe system also preserves standard clinical technique while offering enhanced safety, reliability, and ease of use.



High-Speed Inspection Platform
Syntegon

Syntegon’s AIM9, which offers output of up to 600 vials per minute and container sizes up to 250 ml, is future-proof thanks to integrated visual inspection and leak detection.
Syntegon launched the AIM9 inspection platform based on decades of experience and proven technology. It combines visual inspection and leak detection with outputs of up to 600 vials per minute, helping pharmaceutical manufacturers and CMOs achieve the highest inspection and product quality.
“Efficiency is key in the fast-paced pharmaceutical industry,” says Bernd Barkey, President of Syntegon Japan and Global Head of Inspection. “The new AIM9 enables our customers to boost productivity and meet ambitious delivery targets.”
A further challenge for pharmaceutical manufacturers and CMOs lies in handling multiple container sizes and products. Therefore, Syntegon designed the new AIM9 with enhanced flexibility. The platform accommodates vial sizes from 2 to 250 milliliters and enables easy changeovers as well as fast adaptation for new products.
Prefillable Syringe System Delivers a Fully Verified System-Level Solution
West Pharmaceutical Services West Pharmaceutical Services, Inc. announces the launch of its West Synchrony Prefillable Syringe (PFS) System.

This solution accelerates syringe selection through its comprehensive performance and regulatory data packages and delivers a fully verified system-level solution designed specifically for biologics and vaccines.
It sets a new standard in drug delivery by accelerating syringe selection through its comprehensive performance and regulatory data packages.
Key benefits include:
• Design and System-Level Performance: a single design verification and characterization package from West ensures the form, fit, and function of the entire PFS system.
• Regulatory Ease with Comprehensive Submission: a streamlined submission process with one system-level drug master file and regulatory package tailored for comprehensive regulatory needs.
• Single Source Supply: single supplier approach, offering make-tostock and make-to-order supply with low minimum order quantities, ensuring reliability and flexibility.




Harpak-ULMA offers validation-ready medical packaging solutions that meet all of the standards required, helping to provide maximum product protection, hermetic sealing, customizable package shapes, hygienic individual or multi-packs, and easy-open options.
Learn More



















Amcor Flexibles North America 3, 45, 76 www.amcor.com
Anritsu 26, 46 www.anritsu.com
Antares Vision Group 37, 47, 76 www.antaresvisiongroup.com
BELL-MARK 48, 76 www.bell-mark.com
Chase-Logeman Corporation 49, 76 www.chaselogeman.com
Colbert Packaging Corporation 39, 50, 76 www.colbertpkg.com
Columbia Machine, Inc. 11, 51 www.palletizing.com
Dispense Works Inc. 14 www.dispenseworks.com
DT Engineering 52, 77, 89 www.dtengineering.com
Greydon 25, 53, 77 www.greydon.com
Harpak-ULMA Packaging, LLC. 54, 77, 85 www.harpak-ulma.com
Heat and Control, Inc. 19, 55, 77 www.heatandcontrol.com
High Tek USA 56 www.hightekusa.com
James Alexander Corporation 31, 57, 77 www.james-alexander.com
Massman Companies 15, 58, 78 www.massman.com
Metsa Board 40, 59, 78 www.metsagroup.com
METTLER TOLEDO 60, 78, 78 www.mt.com/pi
MG America, Inc. 23, 61, 78 url
MULTIVAC Group IFC, 62, 78 www.multivac.com/us
Nita Labeling Systems 4, 63 www.nitalabeling.com
NJM Packaging 9, 64, 79 www.njmpackaging.com
Ossid 83 www.ossid.com
Pacteon Group 2, 65, 79 www.pacteon.com
Pfeiffer Vacuum+ Fab Solutions 27, 66, 79 www.pfeiffer-vacuum.com/global/en
Pharmaworks 5, 67, 79 www.pharmaworks.com
PMI KYOTO 41, 68 www.pmikyoto.com
PMMI 81 www.pmmi.org
ProSys Fill LLC. 32, 69, 79 www.prosysfill.com
Sepha Ltd. 33, 70 www.sepha.com
Simco-Ion 18, 71, 80 www.simco-ion.com/industrial
Starview Packaging Machinery Inc. 72, 80, 84 www.starviewpackaging.com
Uhlmann Packaging Systems L.P. 7, 73, 80 www.uhlmann.de
Wipotec 74, 80, OBC www.wipotec.com
WLS 75, 80, 87 www.weilerls.com

NEW PR-12 & PR-24
Plunger Rod Inser ion nd L beling
Inser ing he plunger rod, orquing i in o pl ce, nd l beling he syringe using our new, p en ed ATR echnology
The NEW, p en ed PR-12 nd PR-24 from WLS perform high-speed, high-con rol plunger rod inser ion nd syringe l beling in one, single, comp c -foo prin uni .







































Pharmaceutical Packaging’s Next Phase: Connectivity
Future-ready pharma packaging must integrate connectivity, automation, and material strategy into a single, coordinated approach rather than treating them as separate initiatives.
By Brianna Guntz, Associate Editor
Pharma ceut ical packaging is entering a more connected and compliance-driven era, where digital functionality is becoming as critical as barrier protection. New industry data shows manufacturers accelerating adoption of temperature monitoring, scannable codes, and RFID—while simultaneously balancing sustainability mandates and tighter regulatory oversight.
Currently, monitoring and scannable features dominate packaging features. According to PMMI’s 2026 Trends and Challenges in Pharmaceutical Manufacturing report, 69% of respondents already utilize temperature and humidity monitoring and 67% use QR codes or other scannable features.

Other technology features like RFID (33%) and wireless sensors (13%) are looming but not yet mainstream.
“RFID technology is emerging as a major requirement,” an OEM VP of Product Development said. “Major store brands are requiring RFID tags be embedded in products, and pharma is adopting this for inventory management. We’ve been asked to work on embedding RFID tags in shrink sleeves. We’ve been testing with film manufacturers who embed tags in each label. We haven’t sold equipment for this yet but expect it to become huge soon.”
While monitoring tools such as temperature indicators and QR codes are now common in pharmaceutical packaging, the next wave of investment is shifting toward deeper connectivity.
According to the report, 27% of CPGs plan to add RFID tags, 25% expect to incorporate wireless packaging sensors, and 23% are looking at augmented reality features.
“RFID TECHNOLOGY IS EMERGING as a major requirement. Major store brands are requiring RFID tags be embedded in products, and pharma is adopting this for inventory management.”
The drivers are both regulatory and operational. Serialization mandates and anti-counterfeiting pressures are pushing manufacturers toward technologies that enable item-level traceability and authentication. Unlike traditional printed codes, RFID and near-field technologies can store more data and enable automated tracking throughout the supply chain. For manufacturers managing cold chain distribution or high-value injectables, that added visibility can help reduce disputes, verify the chain of custody, and support compliance documentation.
For more insights from PMMI’s Business Intelligence team and to find reports—including Trends and Challenges in Pharmaceutical Manufacturing and Knowledge Transfer for Machine Operators: From Paper to Digital Intelligence—visit https://www.pmmi.org/ business-intelligence.

From Plant to Patient Safety Through RFID
Today’s healthcare market demands more than quality—it demands accountability, safety, and transparency. The Traceable Quality System (TQS) helps you deliver all three.
TQS seamlessly integrates RFID technology with high-precision printing and inspection—ensuring every vial is clearly marked, automatically verified, and fully traceable down to the dose. All components (printer, camera, RFID) are centrally synced, so there's no manual setup or operator input.
For your customers, that means safer medication handling, fewer errors, and full compliance. For your brand, it means a future-ready solution that reinforces trust and leadership in pharmaceutical safety.
US AT: JAVITS CENTER, NYC APRIL 21 - 26, 2026
BOOTH: 1021









