MedTechRegulatoryInsightsAugust2025
REGULATORY RADAR
Team-NBClari�esSoftwareQuali�cationunderIVDR
Team-NBreleasedanupdatedpositionpaperclarifyingwhensoftwarequalifiesasanIVDunderRegulation(EU)2017/746 Theguidance highlightsstandalonesoftware,intendedpurpose,anddiagnosticfunctionalityaskeycriteria ItaimstoharmonizeNotifiedBodies interpretationsandsupportconsistentconformityassessmentdecisions,particularlyforborderlinecases ReadthePostionPaper(PDF)
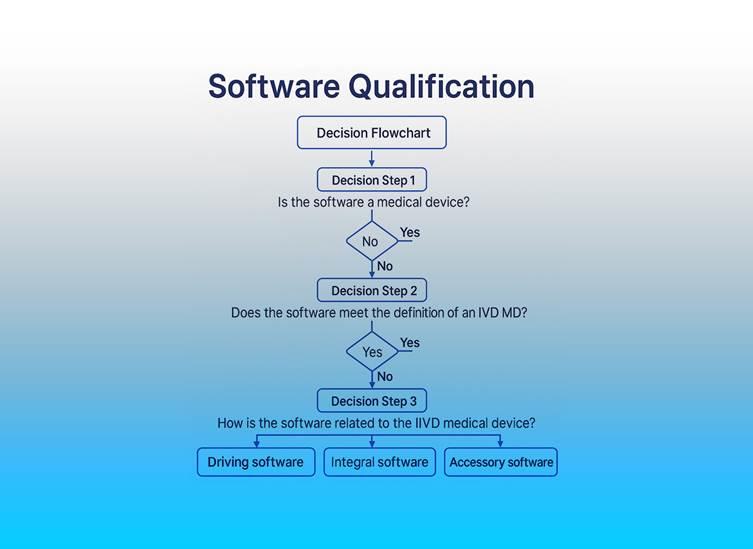
OrphanIVDDevicesGetNBAttention
Team-NBhaspublisheditsfirstpositionpaperfocusedonorphanIVDs emphasizingregulatoryflexibilityfordiagnosticstargetingrarediseases Itacknowledgestheuniquemarket,economic,andclinicalconstraintsfacedbymanufacturersandcallsforproportionateapproachesduring conformityassessment ThisdocumentencouragesdialogueonbalancingaccessandregulatorycomplianceundertheIVDR ReadthePostionPaper(PDF)

EUAddsNewExpertPanel&Clari�esAdminRules
TheEuropeanCommissionadoptedImplementingDecision 2025/1324toupdateadministrativeelementstiedtoexpert panelsundertheMDRandIVDR Thedecisionimproves clarityarounddocumentationhandling responsetimelines andconsultationprocesses Italsoformalizesthecreationof anadditionalexpertpanelinthemedicaldevicesectorto supportefficientevaluations
ReadtheO�cia Decison(PDF)
SingaporeHSARevisesChangeNoti�cationGuidance(GN-21,Rev.6)
HSAreleasedRevision6ofGN-21 updatingitsguidanceonchangenotificationforregisteredmedicaldevices Itincludesclearerdefinitionsof majorandminorchanges,illustrativeexamples,andnewdecision-makingflowcharts Therevisionhelpsregistrantsassesswhetherachange requiresnotification,enhancingregulatorypredictabilityandcomplianceforSingapore’sdevicemarket ViewtheGuidance(PDF)
India’sCDSCOPublishesSubjectExpertCommittee(SEC)Guidance India’sCDSCOpublishedguidancedescribingtheoperationsofSubjectExpert Committees(SECs) whichreviewhigh-riskandinnovativemedicaldevices
ThedocumentoutlinesSECcomposition submissionexpectations timelines andevaluationprinciples ThismovebringsgreatertransparencytoIndias regulatoryprocessandhelpsmanufacturersunderstandSECinvolvementin pre-submissionandreviewstages AccesstheGuidance(CDSCOsite)


FDALaunchesRegulatoryAcceleratorforDigital HealthInnovators
TheFDAintroducedtheRegulatoryAccelerator aninitiative tosupportdigitalhealthdevelopersnavigatingUS regulatorypathways ItcompilescuratedFDAresources tailoredtosoftware-baseddevices coveringsubmission prep qualityexpectations andreviewprocesses Thetoolis especiallyusefulforstartupsseekingfastermarketentry withcompliant,high-qualitysubmissions ExploreRegulatoryAccelerator
BostonScienti�cLaunchesAVVIGOGuidanceSystem
BostonScientifichasintroducedtheAVVIGO+Multi-ModalGuidanceSystem,designedtooptimizepercutaneouscoronaryintervention(PCI) The systemintegratesIVUS FFR andco-registrationcapabilitiestoprovidehigh-resolution real-timeimagingandphysiologicaldata enablingmore precisediagnosisandtreatmentofcoronaryarterydisease TheintuitiveinterfaceimprovesworkflowefficiencyduringPCIprocedures ExploreAVVIGO+
MedLumicsHitsMilestonewithAblaViewPFASystem
MedLumicsannouncedthat10patientshavebeentreatedinitsFirst-in-ManclinicaltrialfortheAblaViewPulsedFieldAblation(PFA)System The systemfeaturesreal-timevisualguidance,enhancingprecisionincardiacablationforatrialfibrillation Theannouncementfollowsleadership changes withanewCEOandBoardMembersteeringinnovation
Readthefulrelease
AITechforBreastCancerRiskGetsFDABreakthroughStatus
WashingtonUniversityresearchershavereceivedFDABreakthroughDeviceDesignationforanAI-basedbreastcancerriskpredictiontool The modelanalyzesmammogramdataandpatientrecordstopredictcancerlikelihoodwithgreateraccuracy particularlyforunderservedpopulations
Thisinnovationmayenableearlierdetectionandmorepersonalizedscreeningstrategies
Learnmore
ArterialCannulaRecall:EdwardsLifesciencesActsonWireExposureRisk
EdwardsLifesciencesrecalledspecificarterialcannulasduetoadefectthatmayexposeinternalwires,riskingvesselinjury TheFDAclassified thisasaClassIrecallduetothepotentialforseriousharmduringcardiacprocedures
FDArecallnotce
EarlyAlert:BaxterFlagsInfusionPumpMalfunction
Baxterissuedanearlywarningaboutinfusionpumpissuesthatcouldcauseinaccuratedosing Thepotentialmalfunctionmayresultinover-or under-infusion Usersareurgedtomonitordevicescarefullyandfollowinterimsafetyinstructions
FDAalert
CranialDrillRecall:RiskofDeviceDisassembly
IntegraLifeSciencesrecalledCodmanDisposablePerforatorsduetoriskofdisassemblyduringcranialprocedures ThisClassIrecalladdresses potentialpatientharmorsurgicaldisruptioncausedbythedrillheaddetachingfromtheshaft
FDArecalldetails
StaplerCartridgeCorrection:LockoutIssuePromptingFieldFix
EthiconEndo-SurgeryissuedacorrectionforEndopathEchelonstaplercartridgesduetounintentionallockoutsmid-surgery Thoughnotaformal recall,affectedlotsmustbereviewed,andnewhandlingguidancefollowed Noinjuriesreported
FDAcorrectionnotice
ASKTHEEXPERT
WhilepreparingClinicalEvaluationReport(CER)fortheCEmarkingofabloodglucosemonitoringsystem(ClassIIadevice)thatusesaglucose, howdoyoudeterminethecorrectclinicalevaluationroute?
Considerations for route of evaluation:
1 ClinicalEvaluationBasedonExistingData(Article61(10)ofMDR)
Ifthedevicetechnologyisalreadyclinicallyvalidatedinsimilardevices,existingclinicaldata(eg,publishedstudies)canbeusedtodemonstrate safetyandperformance
2 ClinicalEvaluationBasedonNewClinicalInvestigations(Article61(1)ofMDR)
Ifthedeviceintroducesnewfeatures clinicalinvestigationsmustbeconductedtogatherfreshsafetyandperformancedata ForallclassIII devicesandfortheclassIIbdevicesitismandatory
Forabloodglucosemonitor chooseOption1ifthesensoriswell-established Option2ifthesoftwareisinnovative
TRIVIA
PHAvs.FMEAinMedicalDeviceRiskManagement
Didyouknow?PreliminaryHazardAnalysis(PHA)andFailureModesandEffectsAnalysis(FMEA)arebothusedinmedicaldeviceriskmanagement butatdifferentstagesandfordifferentpurposes PHAistypicallyappliedearlytoidentifybroadsystem-levelhazards,whileFMEAdigsinto specificfailuremodesduringdesignandproduction
Areyouabletodifferentiatebetweenthetwo?
Letsdecodethisfurtherinourlatestpost
VisittheTriviaPostonLinkedIn
GujaratMedicalExpowillconvenetop-tiermedicalprofessionals innovators andindustryplayerstospotlightadvancementsindiagnostics medicaldevices,andhealthcareinfrastructure TheeventpromotescollaborationandgrowthwithinIndiasMedTechlandscape 13–15September2025| VigyanBhavan Ahmedabad India Registerhere
9thAnnualMedicalDeviceRegulatoryandQualitySummit2025
Thissummitwillgatherregulatoryexperts,qualityleaders,andMedTechprofessionalstodiscussIndiasevolvingregulatoryframework,global harmonization,clinicalevaluations,andpost-marketobligations Amust-attendforRA/QAprofessionals 21–22August2025|PridePlazaHotel Aerocity NewDelhiAirport
Eventdetals
Conclusion:
Thecorrectclinicalevaluationroutedependsonthedevicesriskclass novelty legacystatusandclinicalevidenceavailability guidingthe approvalprocessinalignmentwithMDRArticles61andMDCGrecommendations
Read further:
https://wwwlinkedincom/posts/neujin
