

PDAC Approved
3 Year Warranty













PDAC Approved
3 Year Warranty











Mali Schantz-Feld, MA, CDE Editor in Chief
Joining the Dental Sleep Practice team as Editor in Chief represents a new opportunity — after 16 years as editor of MedMark dental specialty publications, it now will be my honor to be a part of conveying information to improve the health and quality of life for the 50 to 70 million Americans who suffer from some form of sleep-breathing disorder. Our publication’s goal is not only to help define dentists’ roles in this growing sector, but also to help develop awareness, discover evolving and established protocols, and educate about the many products and devices that can be a part of patients’ journey to overall better health.
We are very proud of Dental Sleep Practice’s growth into a reputable source for information on sleep-disordered breathing topics in the dental community. We have always covered medical topics as well, to inform our readers about the complex biological and anatomical synergies that affect those sleep processes. We view collaboration between medical and dental professionals as an imperative, and to illustrate that goal more strongly, we are broadening the branding identity of our publication. Going forward, we are thrilled to fully acknowledge that intersection and connection with our name change to Sleep Practice.
Partnerships and advocacy are at the heart of our mission. Treatment coordinated between dental professionals, medical doctors, and other members of the healthcare community can enhance patients’ understanding, acceptance, and compliance with the team approach that they so greatly need. This interaction extends past the clinical to the business side where dental/medical interaction can simplify referral systems and improve coordination. In clinical and business aspects, a joint approach reduces frustration and increases opportunities for all involved.
We will continue to cover the mental health issues, brain disorders, heart conditions, and chronic diseases that sleep disorders can cause in adults as well as the emotional,
behavioral, and developmental disorders that children and adolescents often face. We will also examine the physiology and functions of the human body that are involved in the process. An article titled “Sleep Deprivation, Sleep Disorders, and Chronic Disease” aptly summed up the key role of sleep medicine in overall wellness when it noted, “It is vital to recognize sleep as a fundamental pillar of public health.”*
Vision, energy, and incredible knowledge are hallmarks of our dental and medical sleep community. In 2026, I invite you to become involved in all aspects of dental/medical sleep. Authors, please email or call me with your ideas, and let your voice impact the lives of millions of people hoping for some relief. You can influence the future of this expanding area.
Shakespeare called sleep “nature’s soft nurse.” For some, this dream is out of reach. Their sleep-related disorder makes what should be a peaceful time of body and brain recovery into a harsh, unhealthy, and frustrating nightmare. The discoveries, perceptions, and triumphs presented in Sleep Practice can be life changing and lifesaving. I look forward to being an enthusiastic and productive steward of this publication and helping to spread the wakeup call.
Looking forward to future connections.
*Ramos AR, Wheaton AG, Johnson DA. Sleep Deprivation, Sleep Disorders, and Chronic Disease. Prev Chronic Dis. 2023;20:230197. doi: http://dx.doi.org/10.5888/pcd20.230197.
Cover Story
Airway Centric Dentistry Powers
Dr. Jerry Hu’s Mission
Dr. Hu practices practical and ethical airway centric dentistry.


Continuing Education
The Nose as a Diagnostic Organ by
Karen Parker Davidson, DHA, MSA, MEd, MSN, APRN
To find the first diagnostic checkpoint of breathing, follow your nose.
10

Clinical Focus Beyond First Latch by Lynda Dean-Duru, DDS and Krystle Dean-Duru, DDS
Even if you don’t treat littles, you need to know this.
2 CE CREDITS


Medical Insight Pharyngeal Biostimulation: A Nonsurgical Frontier in Treating Obstructive Sleep Apnea by Jamila Battle, MD Pharyngeal biostimulation represents the next non-surgical option for effective management of OSA.
Medical Insight The Silent Sleep Sabotage by Alison Kole, MD, MPH, FAASM, FCCP Dental sleep practitioners can play a crucial role in improving the lives of perimenopausal patients.

14 Bigger Picture The Role of the Dental Sleep Practitioner in Protecting Cognitive Health by Erela Katz Rappaport, DMD, D.ABDSM
Opening the airway is part of proactive intervention for long term health.

16 Expert View From Either/Or to Both/And: The CHOICE Trial and a New Era of Collaborative OSA Care by Sue Ellen Richardson, DDS
The CHOICE study compared the effectiveness of CPAP and MAS and their impact in the real world.
19 Product Spotlight Nylon flexTAP®: Revolutionizing Sleep Apnea Care by W. Keith Thornton, DDS, Founder and Chairman, Airway Management, Inc. Nylon flexTAP is the world’s first FDAcleared digitally printed midline oral appliance.

20 Cardio-Sleep Corner Sleep Deprivation and Increased Risk of SportsRelated Injuries by Lee A. Surkin, MD, FACC, FCCP, FASNC, FAASM
Sleep is a critical injury prevention factor for athletes.
34 Product Spotlight Make Dental Sleep Workflows More Efficient
The Nonin Health Platform is a complete system that simplifies dental sleep workflows.
36 Practice Management Why Every Dental Sleep Practice Needs an AI Consultant by Eric Stefanik, IT Security & AI Expert, Elliptic Systems Corporation
AI is everywhere — be careful how much you open your world to it.
38 Team Effort My Doctor Doesn’t Treat Airway – But I Still Do: The Hygienist’s Guide to Saving Breaths (and Lives) by Katrina Sanders, RDH, BSDH, MEd Dental hygienists are the key for monitoring health.
41 Seek and Sleep DSP Word Search
Editor in Chief
Mali Schantz-Feld, MA, CDE mali@medmarkmedia.com
Tel: (727) 515-5118
Associate Editor
Lee A. Surkin, MD, FACC, FCCP, FASNC drsurkin@n3sleep.com
Editorial Advisors
Jamila Battle, MD (Family/Sleep/Addiction)
Steven Bender, DDS
Jagdeep Bijwadia, MD (Pulmonary, Sleep)
Kevin Boyd, DDS
Saim Choudhry, DO (Internal Medicine, Sleep)
Alison Kole, MD, MPH, FCCP, FAASM (Sleep, Pulmonary, Critical Care)
Karen Parker Davidson, DHA, MSA, M.Ed., MSN, RN
Daniel Gartenberg, PhD (Behavioral Sleep)
Kristie Gatto, MA, CCC-SLP, COM
Amalia Geller, MD (Neurology, Sleep)
William Hang, DDS, MSD
Steve Lamberg, DDS, D.ABDSM
Ian Lalich, MD (ENT, Sleep)
Christopher Lettieri, MD (Pulmonary, Critical Care, Sleep)
Pat McBride, PhD, CCSH
Jyotsna Sahni, MD (Internal Medicine, Sleep)
Alan D. Steljes, MD (Cardiology, Sleep)
Laura Sheppard, CDT, TE
DeWitt Wilkerson, DMD
Scott Williams, MD (Psychiatry, Sleep)
Gy Yatros, DMD

Director of Sales & Business Development
Ty Maggard | ty@medmarkmedia.com
Director of Publishing
Amanda Culver | amanda@medmarkmedia.com
eMedia Coordinator
Michelle Britzius | emedia@medmarkmedia.com
Social Media
Felicia Vaughn | felicia@medmarkmedia.com
Website Support
Eileen Kane | webmaster@medmarkmedia.com
MedMark, LLC 400 Crossing Blvd. Bridgewater, NJ 08807 Tel: (480) 621-8955
www.DentalSleepPractice.com
Subscription Rate: 1 year (4 issues) $149

Dentists who practice airway-centric dentistry change more than smiles. They change sleep and daily function.
That is the path Dr. Jerry Hu followed. He first met dental sleep medicine as a patient who could not tolerate CPAP.
Oral appliance therapy helped him regain energy, lose more than 100 pounds, and earn a black belt in karate after 40. He later became a triple-boarded sleep dentist who says it plainly: “Airway trumps everything.” That shift now guides his clinical decisions and teachings on dependable oral appliance therapy.
His view was shaped by family history. He remembers gasping for air, acid reflux, and restless sleep that once caused little concern. With hindsight, he connects those signs to broader systemic risk. Given how many adults live with undiagnosed or untreated obstructive sleep apnea, the dental chair is a logical place to screen and guide care. Airway-first thinking is both practical and ethical.
Early in his career, Dr. Hu focused on full-mouth rehabilitation and esthetics. Over time, he saw a pattern. Restorations that looked excellent at delivery did not always do well at night. Snoring, apneas, and arousal-driven clenching put load on teeth and implants that they were never designed to carry. If sleep-disordered breathing is not managed, dentistry will be asked to endure physiology it cannot overcome. Screening moved from optional to standard, especially before complex restorative or implant plans. Health histories flag snoring, witnessed apneas, acid reflux, morning headaches, nocturia, and daytime sleepiness. Exams record tongue size and scalloping, Mallampati class, wear facets, and abfractions. When risk is present, testing proceeds through a physician. In his practice, an oral appliance always follows a diagnosis, not the other way around.
He also reframed success; a lower index means little if oxygen saturation remains low.
His goals are steady breathing and fewer arousals. When those improve, patients report energetic mornings, fewer headaches, and less depression and mood swings. Treat the airway, steady the breathing, then let physiology do its work.
Bruxism connected the old and new parts of his practice. The literature classifies sleep bruxism as a sleep-related movement disorder that often clusters around respiratory events and arousals. In many patients, parafunction reflects airway stress, not a primary dental problem. A flat-plane bruxism night guard can ignore the airway causes of clenching and, in some patients, make the airway more likely to collapse during sleep. Dr. Hu screens, phenotypes, and treats in collaboration with medicine so device choice, jaw position, and physiology support the same outcome.
The Case that Changed His Course
“Ignore the airway, and physics will find you.” That is how Dr. Hu describes the day a patient walked in and “opened her hand” to reveal half an implant fixture with bone and soft tissue attached. “If I gave you ten years,” he tells colleagues, “you could not break an implant in half with your bare hands.” The case had followed standard steps, including grafting, healing, placement, and osseointegration.
What he had not done was assess through an airway lens. Pre-op photos later showed abfractions and fractures that signaled heavy parafunction. He had not scored a Mallampati, assessed tongue volume, or screened for snoring, acid reflux, or morning headaches.
Eighteen months later, the implant failed. His analysis was direct. During sleep, respiratory events trigger arousals. The autonomic surge that follows can drive strong jaw activity. Occlusion that looks fine when the patient is awake does not describe what the system endures at two in the morning under hypoxic stress. Over months, off-axis loading and shear add up where articulating paper cannot measure them.
On the AAID (American Academy of Implant Dentistry) stage, he challenged the reflex to “protect the dentistry” with generic flat-plane splints. “Some splints can worsen the airway,” he said. A night guard that occupies vertical with-
out regard to mandibular posture, nasal airflow, and collapsibility can make breathing worse in susceptible patients. His message to implant and esthetic colleagues is consistent: screen for sleep, phenotype the patient, and involve a physician before major restorative work.
Good outcomes start with a careful beginning. Dr. Hu spends time capturing a therapeutic start position that respects joints, muscles, and airway, then validates when indicated with tools such as jaw tracking, surface electromyogram (EMG), or cone beam computed tomography (CBCT). Numbers guide judgment; the patient provides the context.
A physician establishes the diagnosis. Titration proceeds stepwise. Coaching covers sleep hygiene, nutrition, activity, and stress regulation. Two stories illustrate the approach.
“Airway-first thinking is both practical and ethical.”
“One patient weighed over 400 pounds, was jobless from nodding off during the day, and unable to climb a single flight of stairs. I shared my own before-and-after photos to encourage him. Months later, he’d lost about 160 pounds — and eventually more than 200 — sent me a video of himself taking the stairs, and told us he’d been promoted at work.”
“Another, a Department of Labor veteran, was a former nuclear test-site worker with severe breathing and oxygen problems, and his wife was very worried. His nighttime oxygen sat around 81%, which I explained would trigger a crash-cart response in a hospital. After titration with an oral appliance, his AHI reached 0.0 and
Jerry Hu, DDS, DASBA, DABDSM, DACSDD, is one of only a handful of triple board-certified sleep dentists in the world and the owner of Smiles of Alaska (www.smilesofalaska.com). He earned his combined Bachelor of Science and Doctor of Dental Surgery degrees from the University of Michigan at age 24.
Dr. Hu is internationally recognized for his expertise in dental sleep medicine, having lectured worldwide, published in peer-reviewed journals, and been named by Dentistry Today as a leader in continuing education for four consecutive years. He is a faculty member at the Medical University of South Carolina, where he helped create the first interdisciplinary sleep medicine curriculum.


REM rose above 25%. His wife said ‘I’m telling you, I don’t know what it is. It’s like a magic thing you put in his mouth. But he is night and day better.’ His cardiologist now takes the dental treatment seriously. “
Oral appliances do not replace CPAP. They offer a path for patients who cannot or will not use PAP when delivered within a diagnosisdriven, titration-guided plan.
Adaptation takes time. Medicare’s 90-day window before efficacy testing matches physiology. Patients leave their appliance delivery appointment with a checklist for cleaning, morning occlusal settling, side-sleeping, alcohol timing, bedroom temperature, and nasal care. Small details add up. Team members reinforce the plan in clear language (American Dental Association, 2017).
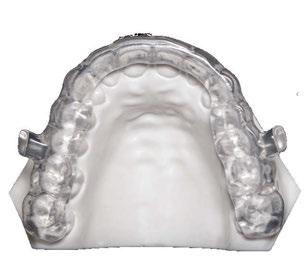
Airway-first care benefits from a partner that is consistent, reliable, and fast. Dr. Hu works with Panthera Dental for four reasons: BioMatch simplifies the ordering and appliance design process, drop-in fit at delivery, 8-day manufacturing, and a 360 Warranty.
BioMatch. A single step workflow ordering and design process that allows a clinician to order and upload patient files directly from the intraoral scanner, for reliable, fast delivery of a drop-in fit, patient-matched appliance.
Drop-in fit and materials. Appliances seat cleanly with no soft-tissue contact, ample tongue space, and a low profile that supports lip seal and nasal breathing. Automated production and a proprietary polishing process create a smooth feel. Devices are metal-free, printed from a medical-grade Type 12 PA2200 nylon that is light, durable, and biocompatible. As Dr. Hu notes, “You get a good scan and boom, it’s there.”

Speed and assurance. Panthera Sleep’s 8-day manufacturing keeps cases moving, especially when year-end schedules tighten. The 360 Warranty offers three years of coverage. As Dr. Hu puts it, “we have a wonderful warranty. Panthera Sleep is so confident in the product that they can give you this warranty.” That promise reinforces both perceived and real value, which helps patients commit to nightly use.
Dr. Hu relies on two mandibular advancement devices from Panthera Sleep because they fit his goals: protect joints and teeth, support the airway, and keep visits focused on coaching not adjustments.
Panthera Classic. A E0486 medicare-reimbursed low-profile device in Type 12 PA2200 nylon. Minimal tooth contact and ample tongue space support comfort and lip seal. The 3.0 rod system offers a broad range and holds position. Panthera X3. A dorsal pathway with fins for guidance and clip-on advancement modules for small changes without tools. Notches allow for elastics to promote mouth closure and nasal breathing in open-mouth sleepers. Also produced in Type 12 PA2200 nylon.
Both devices can be designed with BioMatch, produced in an automated environment with rigorous quality checks, manufactured in 8 days, and covered by the 360 Warranty. Selection follows phenotype and goals — Panthera Classic when rod-based traction and minimal soft-tissue contact serve the plan. Panthera X3 when a dorsal pathway, clip-on titration, and elastics will reduce arousal-driven mouth opening. In both paths, the aim is the same: restore oxygen, reduce arousals, and protect the dentistry by protecting the airway.
1. Screen before planning and coordinate testing with a physician when risk appears.
2. Plan restorations through a sleep lens; bruxism often reflects airway stress.
3. Match devices to phenotype, and collaborate on titration targets.
4. Standardize digital intake with BioMatch to reduce handoffs and speed delivery.
5. Track oxygen saturation and arousals alongside AHI; set expectations for a 90-day adaptation window.
6. Document like a medical device provider when billing medical insurance, including E0486 where applicable.




role of
in
by Lynda Dean-Duru, DDS and Krystle Dean-Duru, DDS

The connection between breastfeeding and craniofacial development begins remarkably early, setting the foundation for proper sleep and breathing patterns throughout life.
Many babies have restrictions that make it difficult, sometimes impossible, to breastfeed. Typically, these situations involve anatomic, functional, and sometimes neurological issues with the baby. The tongue acts as a natural pump while breastfeeding and drives the development of the face, jaws, and airway, and when tongue function is compromised, breastfeeding, breathing, sleep, and the growth of the face and jaws are adversely affected.
At 4 weeks gestation, the neural tube is specializing, mesoderm growing the vascular system and seeding the skeletal, and endoderm invaginating into the gut tube. The endoderm is also forming the pharyngeal pouches, which contain the neuro/ meso/skeletal blueprints for the face, mouth, and throat. Neural crest cells are migrating through all tissues, illuminating cranial nerve sprouts and roots and wiring underlying autonomic brainstem circuits (Courtesy of Neurovascular Institute, Inc. ©2019). Before week four, the tongue is functional/peristaltic. This core principle, Rhythmic Integration,
begins early in the embryo, gains sophistication during fetal development, facilitates recovery perinatally, and continues throughout life, unless interrupted by unresolved trauma or toxic exposure (Dr. Darick Nordstrom). At 8 weeks gestation, genetic blueprints are established, the embryo transitions to fetus and continues to grow and develop for another 32 weeks.
The brainstem has three main functions: as a signal conduit between body and brain, as “home base” for our cranial nerve function, and as an integrative switchboard for signals transmitting between the body and the brain (Courtesy of Neurovascular Institute, Inc. ©2019).
The brainstem is a stalk-like projection of the brain extending caudally from the base of the cerebrum, bridging communication between the cerebrum with the cerebellum and spinal cord.1,2 It has three sections: midbrain, pons, and medulla oblongata, combining to maintain functions necessary for life, such as breathing, consciousness, maintaining blood pressure, heart rate, and sleep regulation.
The brainstem contains both collections of white and grey matter: The grey matter consists of nerve cell bodies and includes important brainstem nuclei; for example, 10 of the 12 cranial nerves nuclei originate there. The white matter tracts involve neuron axons traveling between the cerebrum, cerebellum, and spinal cord to the peripheral nervous system. These tracts carry information to the brain (afferent pathways, such as the somatosensory pathways) and from the brain (efferent pathways such as the corticospinal tract).3
People are born obligate nose breathers. For survival, the baby needs to be able to suck-swallow, and breathe. The important steps include: first breath, breast crawl, and breastfeeding, which all help decompress the cranium, setting the stage for graceful transition of the autonomic nervous system as well as loosening the fascia connections. The healthy swallow is most critical in re-igniting and maintaining rhythmic motion and, with breathing, coordinates fascia, visceral motion, and the brainstem.
It follows then, that intrauterine factors, birth trauma, and intervention could affect these important areas and compromise function and structure. These include the normal process of descent through the birth canal, abnormal position of the baby, and interventions such as epidural and pitocin, which increase compressive forces via stronger contractions. Others are Cesarean births, vacuum, and/or forceps-assisted deliveries. These can lead to somatic dysfunction, notably, occipital condylar compression/dysfunction.5
With extra compressive forces, the skull bones and surrounding fascia can affect the function of the nerves and muscles. The occipital bone, which at this stage is in four parts, is in close proximity to the vagus and hypoglossal nerves, which partially control tongue function, breathing, rest, and digestion. Cranial nerves IX (glossopharyngeal) and XI (accessory nerve) are in close proximity as well.
Occipital condylar compression manifests through various interconnected symp-
toms affecting multiple bodily functions. Common manifestations include difficulties with nursing, sucking, and swallowing, often accompanied by digestive issues such as reflux, vomiting, and colic. Respiratory challenges frequently arise, and patients may experience constipation and bloating. Physical manifestations can include torticollis, and many infants display notable irritability as a result of these combined challenges.
Somatic function, early cranial nerve mapping, and integration are most critical for successful breastfeeding, and problems are best detected and treated within 24-48 hours after birth. Breastfeeding is a baby’s innate function and should be effortless. Suck, swallow, and nasal breathing set the founda-

Dr. Lynda Dean-Duru pursued a degree in dental surgery from the University of Benin in Nigeria and continued her training in the United States, completing her general practice residency at Howard University Hospital and pediatric dentistry specialty training at Children’s National Medical Center. She holds board certification from the American Board of Pediatric Dentistry, Mastership from the World Clinical Laser Institute, and Fellowship Laser Certification from the Academy of Laser Dentistry. In 2000, Dr. Lynda established Ashburn Children’s Dentistry in Virginia and now, Womb2Grow Wellness LLC with a mission to provide comprehensive, holistic dental care for children and promote overall well-being. From the very beginning, she focused on early intervention, starting by helping moms with breastfeeding and nursing difficulties through tongue and lip-tie releases using laser technology. Dr. Lynda was a pioneer, bringing laser dentistry to Ashburn and mentoring countless dentists and dental professionals along the way. She is certified in Advanced Light Functionals (ALF) therapy, a treatment that uses brain-friendly custom-made appliances to promote proper neurology, craniofacial development and optimal airway function, and is a member of the prestigious Gnathos Orthodontic Education Group and a Fellow at the United States Dental Institute.

Dr. Krystle Dean-Duru followed in her mother’s footsteps. With a bachelor’s degree in psychology from Columbia University and a dental degree from Virginia Commonwealth University, Dr. Krystle further honed her skills during her specialty pediatric residency at the Interfaith Department of Dental Medicine in Brooklyn, New York, where she served as Chief Resident. Her specialized training includes pediatric dental sleep medicine, airway orthodontics, and functional frenuloplasty. She is a Diplomate of the American Board of Pediatric Dentistry.
“Suck, swallow, and nasal breathing set the foundation of good function and flow throughout the body.”
tion of good function and flow throughout the body.
Infants experiencing compromised tongue function display a complex array of feeding-related symptoms. During breastfeeding, they often exhibit clicking sounds, chomping or gumming behaviors, and struggle to maintain proper flange formation. The latch is typically shallow and frequently slides off the nipple, leading to extended feeding times and frequent sleep episodes at the breast. These babies commonly show signs of acid reflux, notably clicking sounds and air swallowing during nursing, accompanied by frequent feeding sessions and excessive spit-up.
Digestive issues manifest through gassiness and hiccups, while nursing blisters may develop. The inadequate milk transfer often results in slow weight gain and, in severe cases, failure to thrive. Physical manifestations include a preference for feeding on one side and development of a high vaulted palate. Breathing difficulties become apparent through open mouth posture, heavy breathing patterns, and inability to retain a pacifier. Sleep disturbances often present as congestion upon waking, along with snoring. Skin manifestations such as baby acne or rash may also occur.
Mothers experiencing breastfeeding challenges due to infant tongue dysfunction face
a range of physical and emotional difficulties. Physical symptoms predominantly affect the breast and nipple area, including soreness and discomfort, creased or lipstick-shaped nipples, and flattened nipples. More severe complications can develop, such as plugged ducts, open wounds, and mastitis. Many mothers struggle with low milk supply and may need to resort to using nipple shields. The presence of inverted nipples can further complicate the feeding process. The persistent physical challenges, including ongoing nipple pain and repeated episodes of mastitis, often contribute to low milk supply and can lead to depression.
The challenges in breastfeeding can stem from various underlying conditions and factors that require careful consideration for proper diagnosis. Primary physical factors include fascial restrictions resulting from in-utero positioning or traumatic birth experiences, along with occipital condylar compression. Oral structure issues such as restrictive tethered oral tissues, which can be either functional or structural in nature, play a significant role. Additionally, conditions like hypotonia, laryngomalacia, and Pierre Robin sequence can impact feeding ability. Structural abnormalities such as cleft palate and/or cleft lip must be considered, as well as complications related to prematurity. Maternal metabolic issues can also contribute to breastfeeding difficulties, highlighting the importance of examining both infant and maternal factors in the diagnostic process. The importance of early intervention and proper diagnosis is foundational in a child’s lifelong overall health. It’s critical that healthcare providers, particularly dentists, understand these developmental processes as they directly impact craniofacial development for the children we treat, so we can set them up for a healthy and happy life.
1. Haines DE. Fundamental neuroscience for basic and clinical applications. 4th ed. Elsevier Saunders; 2012.
2. Netter FH. Atlas of human anatomy 6th ed. Elsevier Saunders; 2014.
3. Basinger H, Hogg JP. Neuroanatomy, Brainstem. [Updated 2023 Jul 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK544297/.
4. Nolte J. The Human Brain: An Introduction to its Functional Anatomy. 3rd ed. Mosby; 1993.
5. Tobey AH, Kozar AJ. Frequency of Somatic Dysfunction in Infants With Tongue-Tie: A Retrospective Chart Review, AAO Journal. 2018;28(4):10–14. https://doi.org/10.53702/2375-5717-28.4.10
6. Bonuck K, Freeman K, Chervin RD, Xu L. Sleep-disordered breathing in a population-based cohort: behavioral outcomes at 4 and 7 years. Pediatrics. 2012 Apr;129(4):e857-865. doi: 10.1542/peds.2011-1402.




by Erela Katz Rappaport, DMD, D.ABDSM
s dental sleep practitioners, we understand that the airway is not just a mechanical pathway for breathing, it is central to neurologic, cardiovascular, and systemic health. Increasingly, evidence is pointing to a powerful triad: oral health, sleep health, and cognitive function. For those of us practicing at the intersection of airway dentistry and systemic wellness, this connection provides both a clinical responsibility and an opportunity for leadership in prevention.
We’ve seen patients who “pass” their sleep study but wake unrefreshed and cognitively dull.
Chronic oral inflammation – particularly from untreated periodontitis – acts as a systemic inflammatory burden that may play a direct role in the pathogenesis of dementia. Studies have identified periodontal pathogens such as P. gingivalis in the brain tissue of Alzheimer’s patients, where they appear to promote amyloid production and tau protein hyperphosphorylation (Dominy et al., Science Advances, 2019).
As DSM clinicians, many of us are already evaluating inflammation during oral exams. By incorporating discussions about cognitive risk when we observe signs of chronic periodontitis, we can help patients understand that resolving oral inflammation isn’t just about saving teeth — it’s about protecting the brain.
Sleep-disordered breathing (SDB) — including OSA and UARS – can severely impair glymphatic clearance, oxygenation, and memory consolidation. Neuroimaging studies show that untreated OSA is associated with cortical
thinning, hippocampal atrophy, and accelerated neurodegeneration (Yaffe et al., Neurology, 2011).
In my practice, airway screening is routine. We use structured interviews, CBCT, and sleep studies (home or in-lab) to evaluate airway patency. When OSA is diagnosed, oral appliance therapy (OAT) is a powerful tool – not only to reduce AHI, but to restore the sleep architecture necessary for neurocognitive resilience. We regularly educate patients on how fragmented or hypoxic sleep may be contributing to daytime brain fog, poor memory, or early cognitive decline.
It’s important to remember that many patients with cognitive complaints fall outside traditional OSA thresholds. They may have UARS, positional or REM-related obstruction, or simply poor tongue posture and compensatory mouth breathing. For these cases, a more comprehensive DSM approach — including myofunctional therapy, nasal breathing optimization, and collaborative care such as CBTi — is essential.
We’ve seen patients who “pass” their sleep study but wake unrefreshed and cognitively dull. When we address tongue ties, collapsed lateral walls, or oral dysfunction, we often see significant improvement in subjective sleep quality, energy, and focus.
Educating patients about the sleep-brain connection opens new doors. Discussing
Alzheimer’s prevention may be far more motivating for a middle-aged patient than treating snoring or clenching. By reframing OAT, myofunctional therapy, and inflammation control as part of a broader brain health strategy, we create context for deeper patient engagement and long-term follow-through.
In my office, we routinely explain how sleep and oral inflammation affect brain aging. We align treatment goals not just with AHI reduction, but with enhanced oxygenation, reduced arousals, and long-term cognitive protection. This holistic message resonates deeply — especially among health-conscious adults, caregivers, and high performers.
The dental sleep community is uniquely positioned to contribute meaningfully to cognitive health prevention. We have the tools, the training, and the access to detect airway dysfunction early — often years before neurocognitive symptoms appear. By integrating
airway, inflammation, and brain function into our diagnostic thinking, we shift from reactive care to proactive prevention.
In short, we don’t just help patients sleep better — we help them think clearer, age slower, and live fuller.

Dr. Erela Katz Rappaport is a Diplomate of the American Board of Dental Sleep Medicine and a Qualified Dentist with the American Academy of Dental Sleep Medicine. Based in San Diego, she brings decades of clinical experience and a deep commitment to advancing dental sleep medicine through both patient care and innovation. Originally from New York, Dr. Rappaport earned her BA in Biology from SUNY Binghamton and her DMD from the University of Pennsylvania School of Dental Medicine. Her professional journey has included practice in Philadelphia and Washington, D.C., as well as research contributions ranging from chemical caries removal techniques to the development of a dental implant registry with the Office of Veterans Affairs. An active member of the ADA, CDA, and San Diego County Dental Society, Dr. Rappaport remains passionate about integrating evidence-based sleep solutions into dentistry and improving long-term outcomes for patients with sleep-disordered breathing.
The only OAT device engineered to dilate the velopharynx and the oropharynx.
The Insight 94% of airway collapse involves the velopharynx1. Protruding the mandible dilates the oropharynx but has only indirect effects on the velopharynx.
The Solution
EVO Guided is engineered to prevent airway collapse by repositioning the hyo-glossal complex through guided jaw positioning, tongue posturesupport, and mouth closure—customized from your prescription.
Why EVO Guided?
Prescribe EVO Guided as your frontline device. Engineered to optimize efficacy, patient satisfaction, and adherence, EVO Guided supports improved outcomes in value-based care. Its precision design helps mitigate side effects and reduce costly non-responder cases, making it a smart choice for both patients and practices.
Lingual-less design offers up to 14x more tongue space than predicate devices
1Watanabe et al. (2002). Contribution of body habitus and craniofacial characteristics to segmental closing pressures of the passive pharynx in patients with sleep-disordered breathing. AJRCCM; 165:260-265.
Instructions for Use and User Manual, including safety information, available at https://prosomnus.com/instructions/.


by Sue Ellen Richardson, DDS
Obstructive sleep apnea (OSA) remains one of the most prevalent and undertreated chronic health conditions worldwide. Its consequences — daytime sleepiness, impaired cognitive function, cardiovascular risk, and diminished quality of life — are well known to both physicians and dentists. Although continuous positive airway pressure (CPAP) has long been recognized as the standard of care for reducing respiratory indices, oral appliance therapy (OAT), delivered through mandibular advancement splints (MAS), has demonstrated substantial benefit for many patients by improving symptoms, quality of life, and adherence.
Traditionally, the care pathway has positioned CPAP as the first therapy offered, while OAT was often reserved for patients who could not tolerate PAP therapy. The recently published CHOICE Trial (2024) challenges this long-standing sequencing. It provides compelling evidence that patients may achieve the best outcomes when CPAP and OAT are not viewed as competing therapies but as complementary tools that can be used individually, interchangeably, or in combination. This represents a shift from an either/or approach to a both/and model of OSA care — one that highlights the power of interprofessional collaboration.
The CHOICE study was a multicenter, doublerandomized clinical trial conducted across three Canadian universities, an 11-year ‘labor of love’. The trial recruited 81 treatment-naïve adults with moderate to severe OSA (BMI <35). The trial included 18 months of participation for each patient, with 15 total visits. After initial titration for both CPAP and MAS, participants completed a crossover phase where each therapy was trialed for one month. Importantly, patients were then provided with their own individualized efficacy results for both treatments. This disclosure allowed participants to understand how well each therapy worked for them personally before entering a 6-month “real-world” observational phase, in which they could freely choose between CPAP, MAS, or both on any given night.
The study’s objectives were threefold: to evaluate usage patterns when patients were empow-
ered with choice, to compare the effectiveness and adherence of CPAP and MAS in real-world conditions, and to assess the impact of these treatments on clinical outcomes including blood pressure.
As expected, CPAP was the most effective at reducing respiratory events, lowering the respiratory event index (REI) from a baseline of ~22 events/hour to ~2.2 events/hour. MAS therapy reduced REI to approximately 12 events/hour. Despite this difference in physiologic efficacy, both therapies produced substantial and nearly equivalent improvements in patient-centered outcomes, including daytime sleepiness (Epworth Sleepiness Scale), fatigue (Chalder Fatigue Questionnaire), and daily functioning (FOSQ10, SF-36).
Adherence patterns yielded some of the most striking findings. Patients using MAS monotherapy demonstrated higher average nightly use compared with CPAP monotherapy (6.0 vs. 5.3 hours/night). When given the opportunity to choose between treatments, participants not only used therapy for more hours per night but also on more nights per week. This flexibility translated into a substantial adherence benefit: 84% of participants achieved ≥4 hours of use per night, a threshold often cited as a key clinical benchmark. In other words, allowing patients to select their preferred therapy markedly improved adherence across the board.
Patient preferences over the 6-month observational phase revealed that no single therapy met the needs of all patients. Approximately 20% relied exclusively on CPAP, 9% used only MAS, and a majority — 55% — alternated between the two. This alternation occurred both across nights and, in some cases, within the same night. Preferences shifted during the observation phase, and many participants who predicted they would favor one therapy ultimately chose the other or alternated between both. Only 8% were partially adherent (1–4 hours/night) and 8% were non-adherent (<1 hour/night).
Participants, particularly those with hypertension, experienced significant reductions in
both systolic and diastolic blood pressure. Notably, alternating therapy produced greater reductions in blood pressure than CPAP alone, underscoring the value of sustained adherence made possible by multimodal care.
Adverse effects were documented for both therapies — mask discomfort and air leaks with CPAP, jaw soreness, and chewing difficulty with MAS. However, symptoms diminished over time, and patients who alternated therapies often found relief by switching between devices, enabling them to maintain adherence.
Together, these results highlight that multimodal therapy preserves the physiologic benefits of CPAP while incorporating the adherence advantage of MAS.
Clinical Translation: CHOICE in Practice
The CHOICE Trial has important implications for clinical practice in both medicine and dentistry. Its findings highlight that long-term success depends not only on the therapy itself, but on how well patients are educated, engaged,

Sue Ellen Richardson, DDS, D.ABDSM, FAASM — A single lecture on sleep opened her eyes to how powerfully dental appliances can improve a patient’s health and daily life. Discovering that an oral appliance can not only change — sometimes even save — a life, but also help couples sleep side by side again marked a complete paradigm shift from traditional dental care. This profound insight ignited a curiosity and passion that has guided her career ever since. That passion became personal when her mother, diagnosed with sleep apnea, was terrified of using CPAP and refused to wear it. Hoping simply to help her mother rest, Dr. Richardson made her first oral appliance — an experience that ultimately led her into a field she now loves. Today, Dr. Richardson’s practice is devoted exclusively to oral appliance therapy for obstructive sleep apnea. She works closely with sleep physicians and teaches sleep medicine fellows at Baylor College of Medicine and McGovern Medical School, helping the next generation understand the importance of collaboration in patient care. She also serves as Co-Chair of the AASM Dental Assembly and supports ongoing AADSM and AASM efforts to advance oral appliance education and interdisciplinary communication. Her clinical work includes caring for Houston-area veterans as an approved VA provider, a role she considers a privilege. Dr. Richardson is a Diplomate of the American Board of Dental Sleep Medicine and a Fellow of the American Academy of Sleep Medicine.
and supported in their choices. By disclosing personalized efficacy results and offering both CPAP and OAT, clinicians can create a care environment in which patients are engaged and active participants in their therapy.
Patients valued this flexibility so highly that many were willing to invest in both therapies when given the opportunity. Their willingness to pay for dual treatment options reinforces how much they perceived the benefit of choice in supporting comfort, long-term use, and better health outcomes. This insight points directly to several practical strategies for implementation: Practical strategies include:

“Dentists can build bridges rather than silos, reinforcing their role as essential partners in OSA management.”
• Providing individualized efficacy data to patients from each treatment to support informed decision-making and treatment ownership.
• Encouraging flexibility by presenting multimodal regimens as viable, evidence-based treatment options.
• Measuring success with broad outcomes, including daytime function, fatigue, and blood pressure — not AHI alone.
• Supporting adherence through choice, allowing patients to alternate therapies as needed for comfort and sustainability. Encourage the patient to use a wearable monitor to maintain motivation towards treatment.
For dental sleep professionals, the CHOICE Trial offers more than validation of oral appliance therapy — it provides a powerful opportunity to strengthen collaboration with physicians. The study demonstrates that OAT is not simply a fallback option but a partner therapy that complements CPAP.
Three key messages emerge for dentists engaging medical colleagues:
• Position OAT as a partner therapy. OAT enhances overall adherence when used alongside CPAP, ensuring patients stay in treatment longer.
• Highlight adherence. Physicians already recognize CPAP’s efficacy; CHOICE shows that the addition of OAT leads to
more consistent use and better patient outcomes.
• Focus on shared goals. Both professions aim to reduce symptoms, maintain adherence, and improve health. CHOICE confirms that these outcomes are most achievable when therapies are combined. By presenting OAT in this way, dentists can build bridges rather than silos, reinforcing their role as essential partners in comprehensive OSA management.
The CHOICE Trial reframes OSA care from a binary decision between CPAP or MAS to a collaborative model where patients benefit from both/and. Alternating CPAP and OAT is not only feasible but beneficial, enhancing adherence, supporting patient preferences, and improving outcomes — including meaningful reductions in blood pressure.
For dentists, this evidence represents a call to collaboration. By using CHOICE as a foundation for dialogue, dental sleep professionals can approach physicians with data that validates oral appliance therapy, not as competition, but as a tool that strengthens patient success.
Ultimately, everyone benefits. Physicians gain partners who help their patients stay adherent. Dentists gain credibility and deeper interprofessional connections. Patients gain flexibility, comfort, and improved long-term health. The message is clear: the future of OSA care is not either/or, but both/and. Together, physicians and dentists can offer patients the choice — and the adherence — they deserve.
For a more comprehensive clinical walkthrough of this groundbreaking research, watch Dr. Nelly Huynh’s 2025 AADSM Annual Meeting presentation, where she brings these concepts to life with striking visuals and practical case examples.
And don’t miss the European Respiratory Journal Podcast (April 2025): Continuous Positive Airway Pressure and Mandibular Advancement Splints, featuring Dr. Fernanda Almeida, who shares her expert insights.
1. Hamoda MM, Huynh N, Ayas NT, Rompré P, Bansback N, Masse JF, Arcache P, Lavigne G, Series F, Fleetham JA, Almeida FR. Continuous positive airway pressure and mandibular advancement splints: the CHOICE multicentre, open-label randomised clinical trial. Eur Respir J. 2025 Apr 3;65(4):2401100. doi: 10.1183/13993003.01100-2024. PMID: 39638418; PMCID: PMC11965959.
by W. Keith Thornton, DDS Founder and Chairman, Airway Management, Inc.

As the founder of Airway Management Inc., I’ve spent over three decades advancing oral appliance therapy to help patients with sleep-disordered breathing achieve restful sleep. My mission has been to deliver effective, patient-centered solutions. On September 16, 2025, we launched Nylon flexTAP®, the world’s first FDA-cleared digitally printed midline oral appliance, a milestone that embodies our commitment to innovation through our patented Vertex Technology.

Nylon flexTAP builds on the TAP Sleep Care® system, backed by over 50 peer-reviewed studies — more than any other oral appliance. Crafted from medical-grade nylon using advanced digital printing, it offers a precise, ultra-thin, custom-fit design that’s BPA-free and metal-free. With a 17 mm range of advancement in 1/3 mm increments, it eliminates the need for bite registration, streamlining clinical workflows. The included Mouth Shield helps achieve lip seal and promotes nasal breathing, a feature proven by research to enhance treatment outcomes for mild to moderate obstructive sleep apnea (OSA). Our AM Aligner® morning repositioner is also included with every case.
The heart of Nylon flexTAP is Vertex Technology, a design I’ve championed to address the limitations of traditional mandibular advancement devices. Its dualaxis adjustability — vertical and horizontal — enables precise positioning via angled post hardware, creating additional tongue space without excessive forward protrusion. For patients with larger tongues or narrower airways, this maximizes airway patency
while minimizing discomfort, a critical advancement for effective OSA treatment.
Clinical evidence underscores Vertex Technology’s impact. Studies on TAP® appliances with vertical adjustability show significant reductions in the apnea-hypopnea index (AHI), often requiring 2-3 mm less protrusion than fixed-vertical devices. This reduces jaw pain, TMJ strain, and bite changes, improving patient compliance. Patients report less morning soreness and better sleep quality, while clinicians note enhanced efficacy, especially for CPAP-intolerant patients. A TAP-specific study demonstrated improved AHI reduction and oxygen saturation, aligning therapy with individual anatomy for superior outcomes.
Clinicians trialing Nylon flexTAP have called it the most effective TAP yet, particularly for complex airways. By optimizing airway space multidimensionally, Vertex Technology addresses anatomical variations that single-axis devices overlook, making it a game-changer for OSA management.
This FDA clearance marks a proud moment for Airway Management, reflecting my vision as a founder to empower clinicians with innovative tools. Manufactured in the U.S.A. and available through Airway Labs (PDAC coverage E0486 pending), Nylon flexTAP offers a 4-year warranty and unmatched reliability. I invite dental and medical professionals to explore how Vertex Technology can elevate their practice, transforming lives by helping patients achieve restorative sleep and improved health.
Learn more at tapintosleep.com/.
by Lee A. Surkin, MD, FACC, FCCP, FASNC, FAASM
Agrowing body of research shows that athletes who obtain insufficient sleep have significantly higher injury rates. For example, a recent narrative review reports that athletes sleeping fewer than 7 hours per night exhibit roughly 1.7 times the musculoskeletal injury risk of well-rested peers.1 In a prospective cohort of 340 adolescent elite athletes, those averaging >8 hours of sleep on weekdays had 61% lower odds of new injury (OR≈0.39) than those with shorter sleep.2

College athletes sleeping 6–9 hours (versus less) show significantly reduced incidence of injury.3 In male NCAA Division I basketball players, each additional hour of sleep was associated with a 43% drop in next-day injury risk.4
Studies show that sleeping less than 6–7 hours severely affects performance and doubles injury rates. A meta-analysis of sleep and performance further confirms that even short-term sleep loss significantly degrades reaction time, accuracy, strength, and endurance in athletes5 – deficits that likely contribute to more injuries.
Several cohort and team studies illustrate the sleep–injury link:
• In a study of 24 collegiate women’s soccer players, those sustaining multiple injuries averaged significantly fewer nightly sleep hours than uninjured teammates.6
• A study of 19 male NCAA basketball players found sleep duration was the only significant predictor of in-season timeloss injuries.7
• In women’s volleyball, a multi-month tracking study (17 players, 54 injuries) showed every injury was preceded by a night of shorter sleep, leading authors to declare sleep loss an “independent risk factor” for musculoskeletal injury.8 Even extreme cases highlight the effect: 12 collegiate men who played an uninterrupted 61-hour volleyball game (with almost no sleep) suffered widespread muscle overstrain injuries.9
• In younger athletes, insufficient sleep strongly predicts concussion risk. One prospective study of ~600 student-athletes grouped by sleep found those sleeping ≤5.8 h (vs. >7.0 h) were nearly twice as likely to sustain a sport-related concussion (15.7% vs. 8.8% incidence).10
Biological effects of sleep loss help explain
the injury link. Chronic sleep restriction disrupts endocrine and metabolic processes critical for recovery. Studies show that sleep deprivation elevates cortisol and other stress hormones, driving muscle protein breakdown and impairing glycogen repletion. As a result, muscle recovery is inhibited, and athletes experience higher perceived effort and fatigue during training. Even modest sleep reduction raises pro-inflammatory cytokines in blood, creating a systemic catabolic environment that can weaken connective tissues and delay healing. Poor sleep also alters metabolic appetite signals (ghrelin/leptin), potentially disrupting nutrition and energy balance needed for tissue repair. In female athletes, inadequate sleep may compound hormonal variability (e.g. estrogen fluctuations) and impair neuromuscular control, further elevating injury risk.
Athletically, optimal sleep is when growth hormone and anabolic processes are maximally active (deep NREM sleep). Without this restorative sleep, microtrauma from training accumulates. In short, sleep deprivation blunts the body’s repair and regeneration capacity. One review concludes that any significant shortfall in sleep “inhibits muscle recovery” and predisposes athletes to overtraining and injury.11
Sleep loss also undermines the brain’s control of movement and cognition. Numerous laboratory studies have documented that even acute sleep deprivation slows reaction times, reduces coordination, and degrades decision-making — all critical for avoiding injury. For example, the Rygielski 2019 study notes “clear negative effects” of sleep loss on athletes’ reaction time, accuracy, vigor, and cognitive functions (judgment, decision-making). According to the Vitale 2019 study, mechanistically, <7 h of sleep impairs vigilance and alertness, making athletes slower to perceive and respond to on-field hazards. Motor learning is also compromised: sleep-deprived athletes exhibit poorer technique and balance, increasing biomechanical stress during play. In practice, a sleep-deprived player is more likely to misjudge a jump, mistime a tackle, or lose balance — events that directly raise injury odds.
Summary
These converging lines of high-quality evidence indicate that sleep is a critical injury-
prevention factor. Athletes sleeping sufficiently recover faster, maintain neuromuscular precision, and sustain fewer injuries than their sleep-deprived counterparts. Coaches and clinicians should therefore prioritize sleep hygiene as part of athlete care and injury prevention protocols.
1. Rygielski, A., Melnyk, B., Latour, E., Latour, M., Judek, R., Kowalczyk, Z., Płudowska, K. (2024). The Impact of Sleep on Athletes Performance and Injury Risk: A Narrative Review. Quality in Sport, 19, 54333. https://doi. org/10.12775/QS.2024.19.54333
2. Rygielski, A., Melnyk, B., Latour, E., Latour, M., Judek, R., Kowalczyk, Z., Płudowska, K. (2024). The Impact of Sleep on Athletes Performance and Injury Risk: A Narrative Review. Quality in Sport, 19, 54333. https://doi. org/10.12775/QS.2024.19.54333
3. Rygielski, A., Melnyk, B., Latour, E., Latour, M., Judek, R., Kowalczyk, Z., Płudowska, K. (2024). The Impact of Sleep on Athletes Performance and Injury Risk: A Narrative Review. Quality in Sport, 19, 54333. https://doi. org/10.12775/QS.2024.19.54333
4. WOJTCZAK, Karolina, SKUPIŃSKA, Olga, ANTCZAK, Joanna, FENRYCH, Urszula, LIPSKA, Julia, HAMERSKA, Laura, HAMERSKA, Julia, BOCIANIAK, Bogumił, RUTA, Damian and KAJKA, Anna. Sleep Deprivation as a Risk Factor for Injuries in Athletes. Quality in Sport. 2024;20:53480. eISSN 2450-3118.
5. Kenneth C. Vitale , Roberts Owens , Susan R. Hopkins , Atul Malhotra, Sleep Hygiene for Optimizing Recovery in Athletes: Review and Recommendations. Int J Sports Med 2019; 40(08): 535-543. DOI: 10.1055/a-09053103.
6. Rygielski, A., Melnyk, B., Latour, E., Latour, M., Judek, R., Kowalczyk, Z., Płudowska, K. (2024). The Impact of Sleep on Athletes Performance and Injury Risk: A Narrative Review. Quality in Sport, 19, 54333. https://doi. org/10.12775/QS.2024.19.54333
7. Rygielski, A., Melnyk, B., Latour, E., Latour, M., Judek, R., Kowalczyk, Z., Płudowska, K. (2024). The Impact of Sleep on Athletes Performance and Injury Risk: A Narrative Review. Quality in Sport, 19, 54333. https://doi. org/10.12775/QS.2024.19.54333
8. WOJTCZAK, Karolina, SKUPIŃSKA, Olga, ANTCZAK, Joanna, FENRYCH, Urszula, LIPSKA, Julia, HAMERSKA, Laura, HAMERSKA, Julia, BOCIANIAK, Bogumił, RUTA, Damian and KAJKA, Anna. Sleep Deprivation as a Risk Factor for Injuries in Athletes. Quality in Sport. 2024;20:53480. eISSN 2450-3118.
9. WOJTCZAK, Karolina, SKUPIŃSKA, Olga, ANTCZAK, Joanna, FENRYCH, Urszula, LIPSKA, Julia, HAMERSKA, Laura, HAMERSKA, Julia, BOCIANIAK, Bogumił, RUTA, Damian and KAJKA, Anna. Sleep Deprivation as a Risk Factor for Injuries in Athletes. Quality in Sport. 2024;20:53480. eISSN 2450-3118.
10. Halstead ME, Walter KD, Moffatt K; Council on Sports Medicine and Fitness. Sport-related concussion in children and adolescents. Pediatrics. 2018;142(6):e20183074. doi:10.1542/peds.2018-3074
11. OJTCZAK, Karolina, SKUPIŃSKA, Olga, ANTCZAK, Joanna, FENRYCH, Urszula, LIPSKA, Julia, HAMERSKA, Laura, HAMERSKA, Julia, BOCIANIAK, Bogumił, RUTA, Damian and KAJKA, Anna. Sleep Deprivation as a Risk Factor for Injuries in Athletes. Quality in Sport. 2024;20:53480. eISSN 2450-3118.

Lee A. Surkin, MD, is the Chief Medical Officer of Nexus Dental Systems. A private practitioner in cardiology, sleep medicine, and obesity medicine, he is one of a small group of physicians to be triple board certified in cardiology, sleep medicine, and nuclear cardiology. In 2009, he created Carolina Sleep – the only dedicated sleep medicine practice in eastern NC. Dr. Surkin has created a cardiovascular and sleep healthcare model that includes a multi-faceted diagnostic and treatment approach that is enhanced by a network of relationships with physicians, dentists, respiratory therapists, sleep technologists, and public officials who recognize the important role that sleep medicine has in our daily life. In 2012, Dr. Surkin founded the American Academy of Cardiovascular Sleep Medicine which is a not-for-profit academic organization dedicated to educating healthcare providers, supporting research, and increasing public awareness of the convergence between cardiovascular disease and sleep disorders. In 2014, Dr. Surkin created a new multi-specialty practice called Carolina Clinic for Health and Wellness which combines his specialties with primary care, gynecology, behavioral health and a medical spa. Dr. Surkin is married, has three daughters, a golden retriever, and resides in Greenville, NC.

A summary of the article “Integrative
Apnea: Principles and Practice” by Drs. G. Dave Singh and Jamila Battle, published in the Journal of Clinical Sleep Medicine (Vol. 21, No. 9, 2025)
by Jamila Battle, MD
In the article titled “Integrative Treatment of Obstructive Sleep Apnea: Principles and Practice” in the Journal of Clinical Sleep Medicine, my co-author Dr. G. Dave Singh and I emphasized a core idea: effective management of OSA requires collaboration across specialties — medical, dental, and behavioral. As the field advances, that same integrative model continues to shape the development of emerging therapies.
One promising innovation aligned with this philosophy is pharyngeal biostimulation — a nonsurgical, non-pharmacologic approach designed to enhance upper airway muscle tone through targeted daytime stimulation. This therapy represents the next frontier for airway-focused clinicians seeking to address the underlying physiology of OSA rather than relying solely on nighttime mechanical support.
OSA is a heterogeneous disorder. While some patients present with obesity-driven airway collapse, others exhibit neuromuscular weakness or craniofacial limitations. This variability explains why a “one-sizefits-all” model — such as CPAP alone — often falls short in real-world adherence and outcomes.
Pharyngeal biostimulation seeks to address one of these root contributors: reduced neuromuscular tone of the upper airway. By retraining airway dilator muscles during waking hours, it may be possible to reduce collapsibility during sleep — analogous to physiotherapy for the pharyngeal muscles.
of
To explore this concept, Singh and Battle conducted a pilot study evaluating non-invasive daytime biostimulation in adults with OSA. Three participants (two men and one woman, ages 37–62) completed approximately 2 months of treatment.
Protocol highlights:
• Device: A handheld extraoral stimulator applied beneath the chin in the submandibular region.
• Schedule: 15–20 minutes per day, seven days per week
• Duration: 8 weeks
Measurements:
• Distance from skin to tongue base (DST)
• Cross-sectional area (CSA) of the retroglossal airway
• Sleep indices: Apnea-Hypopnea Index (AHI), Oxygen Desaturation Index (ODI), and oxygen saturation (SpO2).
Following 2 months of regular use:
• DST increased by roughly 1.6 cm, suggesting caudal traction.
• CSA increased by about 1.8 cm², implying greater airway patency.
• AHI improved modestly (from 39 to 34 events/hour).
• ODI decreased (from 37 to approximately 32).
• Mean SpO2 improved from 95% to 97%.
While preliminary, these findings demonstrate early physiological and structural benefits that may translate into functional improvement.
Dentists have long been on the front lines of airway health. Routine oral evaluations reveal craniofacial features often linked to sleep-disordered breathing — narrow palates, retrognathia, and restricted tongue posture. Pharyngeal biostimulation aligns naturally with this airway-aware lens.
For the dental community, this technology offers a potential adjunct to mandibular advancement therapy, orthodontic expansion, or myofunctional rehabilitation. The focus shifts from passive mechanical correction to active neuromuscular training — an exciting paradigm for functional dentistry.
Still, this therapy is not a replacement for comprehensive medical assessment. Its role is most powerful when integrated within an interdisciplinary team.
Sleep medicine specialists remain the cornerstone of care. Their responsibilities include:
• Conducting the initial consultation and symptom review.
• Determining medical necessity for diagnostic testing.
• Confirming OSA severity and identifying phenotypic contributors.
• Evaluating and treating other sleep disorders.
• Coordinating medical management of comorbidities and follow-up evaluation.
Equally vital in an integrative care model is the behavioral health perspective. Cognitive Behavioral Therapy for Insomnia (CBT-I) remains the gold-standard, evidence-based approach for addressing the behavioral and psychological contributors to poor sleep. Incorporating CBT-I within OSA management helps patients overcome insomnia, improve adherence to PAP or oral appliance therapy, and reinforce healthy sleep behaviors — bridg-
ing the gap between physiology and behavior.
The Singh and Battle pilot was intentionally small, designed to test feasibility and generate hypotheses. Larger randomized controlled trials are now needed to determine long-term efficacy, optimal treatment parameters, and phenotype-specific responses.
If validated, pharyngeal biostimulation could represent a significant step forward in daytime adjunctive care for OSA — especially for patients who struggle with CPAP or who seek multimodal, non-invasive solutions.
Pharyngeal biostimulation exemplifies the integrative philosophy first outlined in “Integrative Treatment of Obstructive Sleep Apnea: Principles and Practice” — collaboration between medical and dental professionals to deliver individualized, physiologic, and patient-centered care.
As evidence grows, airway-focused dentists and sleep physicians together can lead a new era of treatment — one that combines innovation, accessibility, and the shared goal of restoring restorative sleep without unnecessary invasiveness.

Dr. Jamila Battle is a triple board-certified physician specializing in sleep medicine. She is dedicated to improving lives through comprehensive sleep evaluations and holistic, patient-centered care. With expertise spanning Obstructive Sleep Apnea (OSA), insomnia, circadian rhythm disorders, and other sleep-related challenges, Dr. Battle offers personalized and innovative solutions tailored to each patient’s unique needs. Dr. Battle is passionate about empowering patients with options that go beyond traditional therapies. While she is experienced with Positive Airway Pressure (PAP) therapy, she is also knowledgeable about alternatives like oral appliance therapy, epigenetic orthodontics, and myofunctional therapy. Her integrated approach addresses the vital relationship between sleep and overall wellness. Dr. Battle’s philosophy is grounded in her education and personal journey. A graduate of Duke University, UNC Chapel Hill School of Medicine, and the University of Michigan Family Medicine, she incorporates the pillars of health into her practice. She is also a #1 Amazon bestselling author of from Abuse to Abundance and the creator of RebootRepairRebirth®, a system for healing and fostering self-love. Dr. Battle actively contributes to improving care pathways for managing sleep-related disorders. Her use of technology complements her personalized approach, ensuring patients benefit from the latest advancements in sleep medicine. She brings empathy and dedication to every aspect of her work, always seeking to grow and improve the care she provides.


by Alison Kole, MD, MPH, FAASM, FCCP

Perimenopause and menopause mark a transformative period in women’s health, profoundly impacting sleep quality and overall well-being. For dental sleep practitioners, understanding the interplay between hormonal changes and sleep disturbances is crucial — not only for effective patient care but also for identifying overlapping symptoms of sleep-disordered breathing that can masquerade as perimenopausal complaints.
Up to 80–90% of women experience a range of symptoms related to fluctuating and declining hormones including estrogen, progesterone, and testosterone, during perimenopause, with sleep disturbance being a major quality-of-life concern.1 Typical complaints include:
• Insomnia (difficulty falling or staying asleep)
• Frequent early awakenings
• Interrupted sleep from vasomotor symptoms (night sweats, hot flashes)
• Sleep-related breathing disorders
• Movement disorders such as restless leg syndrome
Prevalence of sleep disorders escalates from 16%–47% during perimenopause, rising to 35%–60% in postmenopausal women. These sleep issues can be exacerbated by mood changes, decreased melatonin production, and circadian rhythm alterations associated with aging.1
Fluctuations in estrogen and progesterone underlie much of the sleep turbulence during the menopausal transition. In particular, progesterone acts as a neuroactive steroid with sedative, anxiolytic, and hypnotic effects on the central nervous system. Progesterone’s metabolites (e.g., allopregnanolone) interact with GABA and benzodiazepine receptors to promote restorative sleep, reduce anxiety and irritability, and even stimulate respira-
tion – thereby potentially supporting upper airway tone.2,3 Studies show oral micronized progesterone (MP) can:1,4
• Decrease night sweats
• Improve perceived sleep quality
• Enhance sleep efficiency
A recent open-label pilot study in Japanese women receiving hormone replacement therapy with estradiol and micronized progesterone demonstrated significant short-term improvements in sleep quality, particularly among participants with low baseline sleep efficiency, highlighting the need for larger, controlled trials to confirm these findings.5
Vasomotor symptoms, particularly night sweats and hot flashes, are critical disruptors of sleep in perimenopausal and menopausal women. Many women report that calming these symptoms — often through hormone therapy — produces the greatest improvement in sleep. Still, not all patients are eligible for or comfortable with hormone replacement, and risks must be individually assessed, especially for those with a personal history of hormone-sensitive cancers.1,6
Recent advances and updated guidelines now offer a spectrum of non-hormonal therapies for menopausal symptoms and related sleep disturbances.1 These include:
• Neurokinin B antagonists: New on the scene, this class targets vasomotor symptoms and shows promise for sleep and mood issues.
• Gabapentin: Well-established for night sweats and insomnia, particularly in women who cannot use HRT.
• SSRIs/SNRIs (e.g., low-dose paroxetine): Effective for hot flashes and may modestly improve sleep.
• Melatonin: Promotes sleep, although efficacy varies across studies.
Lifestyle modifications — like regular exercise, stress reduction, and sleep hygiene — remain foundational. Cognitive Behavioral Therapy for Insomnia (CBT-I) and Mindfulness-Based Interventions (MBI) are both well-supported for alleviating nonphysiologic symptoms, including depression, anxiety, cognitive changes, and overall quality of life.7 CBT-I has been shown to improve both
insomnia and vasomotor symptoms, particularly in women who cannot use or choose not to take hormone replacement therapy.1
Sleep-disordered breathing — especially obstructive sleep apnea (OSA) — becomes much more common as women transition through menopause and lose the protective effects of estrogen and progesterone. Studies confirm menopausal status is independently associated with increased prevalence of OSA, even after controlling for body weight. In fact, women may develop OSA despite only mild weight gain or having a petite body type due to hormonal shifts and changes in upper airway anatomy. As many as 47%–67% of postmenopausal women are estimated to have OSA.8,9
Unlike the classic male presentation — loud snoring, witnessed apneas, and significant daytime sleepiness — women with OSA may report subtle symptoms that frequently overlap with perimenopausal complaints: chronic insomnia, unrefreshing sleep, morning headaches, and low energy. This atypical presentation means that OSA — and the closely related entity of COMISA (co-morbid insomnia and sleep apnea) — can easily go undiagnosed in women. Evidence shows that between 30%–50% of OSA patients indicate insomnia-type complaints.10 Therefore, anytime insomnia is part of a woman’s sleep complaints, practitioners should have a low threshold for assessing for underlying OSA — using in-lab polysomnography or multi-night home sleep studies, both of which are becoming more accessible.10
Sleep-disordered breathing — especially obstructive sleep apnea (OSA) — becomes much more common as women transition through menopause and lose the protective effects of estrogen and progesterone.


Dr. Alison S. Kole is Board Certified in Sleep, Pulmonary, and Critical Care Medicine. She is currently host of the Sleep Is My Waking Passion podcast, Medical Director of the Oak Health Center Sleep Telemedicine program, and Director of Medical Affairs and Strategy for TotalCME. Dr. Kole believes that empowerment and advocacy through patient and provider education and interdisciplinary collaboration are key components to improving sleep health globally.

As diagnostic approaches become more tailored, dental sleep practitioners are uniquely positioned to play a crucial role in enhancing quality of life for women navigating the menopausal transition.
For women who cannot tolerate CPAP, oral appliance therapy (OAT) — custom-fitted by dental professionals — offers an effective alternative. OAT has been shown to reduce snoring and breathing disturbances, while also improving oxygen levels, daytime alertness, and overall sleep quality, particularly in those with mild-to-moderate OSA. Research suggests that women have similar-to-improved response rates and adherence rates to OAT compared to men.11,12
Ultimately, dental sleep practitioners must recognize that women may not always present with typical OSA symptoms. If insomnia is a central complaint — especially in the menopausal transition — COMISA should be considered, and OSA actively screened for, to ensure accurate diagnosis and tailored treatment that may include CPAP, oral appliances, surgical options, or future medication breakthroughs.
Effective patient care requires a collaborative approach. Partner with the medical community, including menopause specialists, to advocate for sleep testing when patients fail to improve with standard therapies or when OSA symptoms are suspected, even if they present atypically.
Patient education should encompass the full spectrum of OSA treatment options, including oral appliances. This comprehensive

approach ensures that women receive individualized care that addresses both their sleep disorders and broader health needs during this critical life transition.
Sleep disorders in perimenopausal women are multifactorial and demand a personalized, interdisciplinary approach. While both hormonal and non-hormonal treatments offer benefits, clinicians must carefully weigh efficacy, safety, and patient preferences when developing treatment plans. As nonhormonal modalities advance, and diagnostic approaches become more tailored, dental sleep practitioners are uniquely positioned to play a crucial role in enhancing quality of life for women navigating the menopausal transition. By recognizing atypical presentations, collaborating with specialists, and offering comprehensive treatment options, we can better serve this underdiagnosed population.
1. Troìa L, Garassino M, Volpicelli AI, et al. Sleep Disturbance and Perimenopause: A Narrative Review. J Clin Med. 2025 Feb 23;14(5):1479.
2. Belelli D, Hogenkamp D, Gee KW, et al. Realising the therapeutic potential of neuroactive steroid modulators of the GABAA receptor. Neurobiol Stress. 2019 Dec 23;12:100207.
3. Lancel M, Faulhaber J, Holsboer F, et al. Progesterone induces changes in sleep comparable to those of agonistic GABAA receptor modulators. Am J Physiol. 1996 Oct;271(4 Pt 1):E76372.
4. Prior JC, Cameron A, Fung M, et al. Oral micronized progesterone for perimenopausal night sweats and hot flushes a Phase III Canada-wide randomized placebo-controlled 4 month trial. Sci Rep. 2023 Jun 5;13(1):9082. Erratum in: Sci Rep. 2024 Jul 27;14(1):17229.
5. Ogawa M, Makita K, Takamatsu K, et al. Changes in Sleep Quality after Hormone Replacement Therapy with Micronized Progesterone in Japanese Menopausal Women: A Pilot Study. J Menopausal Med. 2025 Apr;31(1):45-50.
6. Baker FC, Lampio L, Saaresranta T, et al. Sleep and Sleep Disorders in the Menopausal Transition. Sleep Med Clin. 2018 Sep;13(3):443-456.
7. Spector A, Li Z, He L, et al. The effectiveness of psychosocial interventions on non-physiological symptoms of menopause: A systematic review and meta-analysis. J Affect Disord. 2024 May 1;352:460-472.
8. Jehan S, Auguste E, Zizi F, et al. Obstructive Sleep Apnea: Women’s Perspective. J Sleep Med Disord. 2016;3(6):1064. Epub 2016 Aug 25.
9. Wang Y, Liu H, Zhou B, Yet al. Menopause and obstructive sleep apnea: revealing an independent mediating role of visceral fat beyond body mass index. BMC Endocr Disord. 2025 Jan 26;25(1):21.
10. Sweetman A, Lack L, Bastien C. Co-Morbid Insomnia and Sleep Apnea (COMISA): Prevalence, Consequences, Methodological Considerations, and Recent Randomized Controlled Trials. Brain Sci. 2019 Dec 12;9(12):371.
11. Vecchierini, MF., Attali, V., Collet, JM. et al. Sex differences in mandibular repositioning device therapy effectiveness in patients with obstructive sleep apnea syndrome. Sleep Breath 23, 837–848 (2019).
12. Fransson A, Nohlert E, Tegelberg Å, et al. Gender differences in oral appliance treatment of obstructive sleep apnea. Sleep Breath. 2024 Aug;28(4):1723-1730.
• Whole-Team Training
• Ongoing Coaching
• Streamlined Protocols
• Precision Bite Technology
• Patient Ed ucation
• In-Network Medical Billing



by Karen Parker Davidson, DHA, MSA, MEd, MSN, APRN

This self-instructional course for dentists aims to reposition the nose as a clinically meaningful diagnostic organ in dental sleep medicine, by demonstrating how airflow patterns and nasal resistance act as functional biomarkers, and by showing how integrating these measurements can improve intervention planning, therapy tolerance, and patient outcomes.
Dental Sleep Practice subscribers can answer the CE questions online at https://dentalsleeppractice.com/continuing-education/ to earn 2 hours of CE from reading the article. Correctly answering the questions will demonstrate the reader can:
1. Describe the multifaceted physiological roles of the nose beyond simple airflow.
2. Interpret how nasal airflow patterns and resistance serve as functional “fingerprints” of nasal health and dysfunction.
3. Differentiate among airflow states and correlate them with clinical phenotypes in dental sleep patients.
4. Apply the concept of “useful resistance” and the Goldilocks Zone to clinical decision-making in dental sleep practice, CPAP/ oral appliance tolerance, and nasal intervention planning.
The nose is often dismissed as a simple conduit for air, yet its role in health and disease extends far beyond basic anatomy. In reality, the nose is the first diagnostic checkpoint of breathing, a barometer of systemic health, and the gateway to understanding why patients succeed, or fail, in sleep and airway therapies.
For dental sleep medicine providers, this distinction is crucial. When the nose falters, compensatory mechanisms ripple throughout the orofacial complex. In children, this can alter craniofacial growth; in children and adults, it can drive clenching, bruxism, and airway instability; across the lifespan, it perpetuates sleep-disordered breathing and related respiratory disorders.
This article introduces the nose as a diagnostic organ, highlighting how airflow patterns and resistance serve as functional “fingerprints” of nasal health. By reframing nasal function and resistance as measurable, classifiable, and clinically actionable, dental sleep practitioners can better recognize hidden drivers of dysfunction and tailor interventions that restore not just the airway, but also the quality of sleep and systemic health. Think of nasal resistance as the “Fifth Vital Sign”; this is not the first you have heard me propose this concept.
Before air reaches the lungs, it must be conditioned, filtered, and regulated by the nasal passages. The nasal turbinates and mucosa create a vast surface area designed for evaporative and conductive heat transfer. Inspired air is warmed toward near-alveolar temperature, humidified to protect the lower airways from dehydration, and filtered to trap particulates and pathogens. Far from trivial, this “air-conditioning” system reduces bronchial reactivity, stabilizes mucosal physiology, and modulates immune signaling to the lungs.
Equally critical is nitric oxide (NO), produced in high concentrations within the paranasal sinuses. Nasal and sinus-derived NO acts as an endog-
enous antimicrobial, antiviral, and vasoregulatory molecule. It enhances mucociliary clearance, promotes bronchodilation, and directly influences pulmonary gas exchange when inhaled into the lungs. Reduction of nasal NO, whether from chronic rhinosinusitis, polyps, or obstruction, impairs these defenses and has measurable impact on infection risk, ventilatory efficiency, and overall sleep physiology.
Neuroregulation further underscores the nose’s complexity. Through the trigeminal nerve, the nose communicates with the central nervous system, regulating airflow reflexes, autonomic tone, and even cortical arousal thresholds during sleep. This sensory integration explains why nasal obstruction not only limits airflow but also heightens sympathetic drive, fragments sleep, and increases cardiovascular strain. The nervous system feeds off the nose, and the nose, in turn, feeds off the gut, an emerging concept I coined as the “gut–nose axis.”
Together, these physiological and host-defense functions establish the nose as the indispensable first gateway of sleep breathing, one that cannot be bypassed without consequence to systemic and sleep health.
The function of the nose lies in the aerodynamics of respiration. Traditional discussions of nasal obstruction emphasize anatomy; septal deviation, turbinate hypertrophy, and valve collapse, but anatomy tells only part of the story.
Furthermore, resistance in the upper airway is needed to maintain a Functional Reserve Capacity (FRC) of the lungs; we need a certain amount of nasal resistance for optimal function, call it the “Goldilocks Zone” — not too much, not too little, just enough for homeostasis of the airway. This concept was presented as far back as 1973 at the 3rd Congress of the International Rhinologic Society. Since then, computerized rhinomanometric technology has been developed in Germany by Dr. Klaus H. Vogt, an ENT, OMF, and DDS who became frustrated with subjectiveness of his surgical outcomes.1
Research from our ENT airway laboratories has since confirmed that the nose is not merely a passive conduit for airflow but an active participant in the aerodynamics of respiration.2-4 In healthy individuals, total airway resistance, distinct from nasal resistance, averages approximately 2.8 cm H2O/L/sec (≈0.275 Pa/cm³/sec),
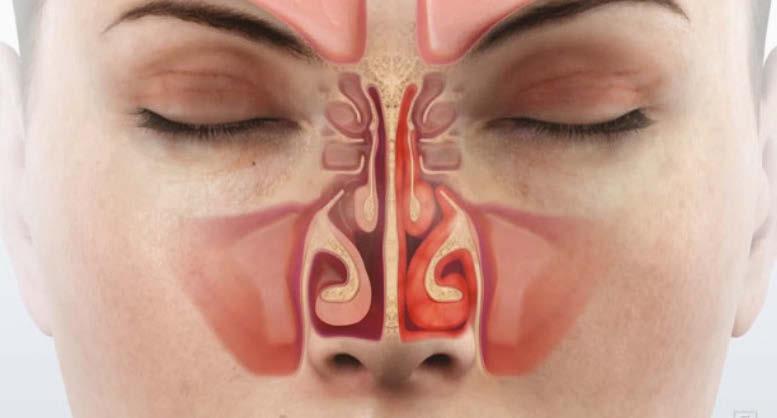
with the nasal passages contributing nearly half of this load (Table 1). This “useful additive resistance” plays a physiological role in stabilizing alveoli against collapse by counterbalancing surface tension forces, thereby supporting functional residual capacity.
Patients with chronic nasal obstruction often leads to adaptive mouth breathing which increases resistance throughout the upper airway and alters respiratory mechanics. Evidence from computational fluid dynamics and clinical

Dr. Karen Parker Davidson, APRN, is a globally recognized expert, healthcare executive, educator, and author specializing in nasal physiology and objective airway measurements, with nearly four decades of clinical, research, and leadership experience spanning critical care, ENT, and sleep medicine. A former United States Air Force Reserve Flight Nurse and Critical Care Air Transport (C-CAT) member, she is the founder of the International Society of Airway Function, Diagnostics, and Therapeutics, and serves as CEO of MedTec Research, Inc., where she advances innovations in nasal physiology, airway diagnostics, and AI-driven airway assessment tools. She is the Clinical Director and Educator for Intake Breathing and a member of the American College of Healthcare Executives and the National Association of Clinical Nurse Specialists.
Dr. Davidson is a National Science Foundation award–winning innovator who holds multiple patents and copyrights related to nasal resistance as a biomarker, advanced data logic and interpretation, and genome sequencing for airway disorders. She is the inventor of the DAFNE Score Software, the creator of the Davidson Maneuver and MAVERICK, and has co-invented several forthcoming medical devices poised to transform clinical practice. As a dedicated educator, she teaches health policy and medical coding at Liberty University and Central Michigan University, inspiring the next generation of healthcare leaders and researchers. Residing near Annapolis, Maryland, Dr. Davidson continues to advance translational airway research, education, and innovation while shaping clinical practice globally, bridging science, technology, and patient-centered care.
sleep studies confirms that nasal obstruction, especially when persistent, raises airway collapsibility and load. For example, in obstructive sleep apnea (OSA) patients, oral breathing is associated with significantly higher resistance and negative static pressures downstream, compared to nasal breathing.
These insights help clarify several long-standing clinical puzzles: why nasal obstruction often feels more pronounced at rest than during exertion; why over-resection of the turbinates can lead to persistent symptoms despite anatomical patency; why some individuals with complete obstruction remain functionally well adapted while others experience significant distress; and why symptoms of Upper Airway Resistance Syndrome (UARS) may recur even after apparent resolution of sleep fragmentation and obstructive nasal breathing through maxillary expansion or nasal bone widening, likely due to a downstream shift in airway resistance known as pseudo UARS (p-UARS) rather than restoration of optimal flow dynamics.1
Modern studies employing computational fluid dynamics and 4-phase rhinomanometry demonstrate that nasal resistance is not inherently detrimental but serves as a key modulator of airflow distribution, lung recruitment, and upper–
DefinitionOpposition to airflow throughout the entire respiratory tract (nose, pharynx, larynx, trachea, bronchi, bronchioles).
MeasurementPulmonary function testing measuring pressure–flow relationship (ΔP/flow).
lower airway integration. The emerging concept of “useful resistance” aligns with contemporary evidence showing that optimal nasal resistance supports ventilatory drive, pulmonary compliance, and even sleep physiology. High-resolution simulations further reveal that it is not resistance itself, but turbulent or unstable flow states, that most often drive pathology. Collectively, this growing body of evidence calls for a fundamental shift in perspective: the nose should be regarded not merely as a passive conduit or site of obstruction, but as a dynamic organ of respiratory regulation and diagnostic significance.
Within this framework, nasal airflow can be classified into distinct categories spanning normal states, resistance-driven flows, pathological dysfunctions, neuroregulated cycles, and extreme disease states. A few illustrative examples:
• Laminar flow: Smooth, efficient airflow with minimal turbulence, seen in healthy noses.
• Turbulent flow: Chaotic, swirling airflow, hallmark of septal deviation or narrowed valves; linked to noisy breathing and snoring.
• Asymmetric partition flow: Unequal right–left distribution (e.g., 70/30 split), often due to deviation or valve collapse.
Resistance to airflow only within the nasal passages (nasal cavity, turbinates, valves, septum, choanae).
Rhinomanometry measuring transnasal pressure difference versus flow. Unit
Scope of Measurement Global measure — effort to move air from mouth/nose to alveoli.
LocalizationWhole respiratory system.
Clinical Relevance Pulmonary medicine (asthma, COPD, restrictive diseases).
Diagnostic Value Doesn’t identify the site of obstruction.
Proportional Contribution N/A
H₂O/L/sec or Pa/cm³/sec (same unit, but restricted to nasal segment).
Limited measurement — isolates the nasal component of total airway resistance.
Just the nose.
ENT, sleep medicine, dentistry, and orthodontics — nasal dysfunction affects patient stratification, identification, and diagnosis, orthodontic treatment, CPAP use, oral appliance compliance, pre and post surgical outcomes, and post-treatment stability.
Pinpoints nasal obstruction; quantifies impact; rhinomanometry is uniquely powerful.
Nose contributes up to ~50% of total airway resistance in normal physiology; high Rn disproportionately affects downstream airflow.
Plain Terms Total “drag” lungs face. “Front door lock” often the rate-limiting step of airflow.
• Jetting flow: High-velocity streams through narrowed internal nasal valve, perceptible as whistling or localized mucosal trauma.
• Bypass flow: Mouth breathing as compensation, often leading to malocclusion, craniofacial growth disturbance, disturbed oral microbiome, and appliance failure.
• Adaptive flow: A natural fluctuation in nasal resistance that may be exaggerated in disease or reduced in cases of autonomic dysfunction.
• Critical flow limitation: A plateau of inspiratory airflow despite increased effort, mirroring the pathophysiology of obstructive sleep apnea.
Each flow state functions like a diagnostic fingerprint, revealing the underlying drivers of dysfunction: structural, inflammatory, neurologic, developmental, environmental, or systemic. Where a CT scan might show anatomy, and a sleep study might show apnea, airflow analysis connects physiology to clinical outcomes.
Nasal resistance is not only a measure of airflow but can serve as a window into systemic health. Thyroid dysfunction, particularly hypothyroidism, may manifest as increased nasal resistance due to mucosal edema, thickened secretions, and decreased neuromuscular tone, narrowing the nasal passages and elevating airflow impedance.5 Similarly, systemic and local conditions such as allergic rhinitis, chronic sinusitis, nasal polyps, asthma, and COPD can alter nasal resistance by influencing mucosal swelling, airway caliber, or bronchial tone.
The gut–nose axis exemplifies shared immune and epithelial barrier mechanisms linking the nasal and intestinal mucosa. Emerging evidence suggests that disruptions in gut health or microbiome balance, through dysbiosis or systemic inflammation, can manifest in the nose as mucosal edema, congestion, and increased resistance.6,7 These changes may worsen breathing difficulty, sleep fragmentation, or CPAP intolerance. This highlights the nose as a sensitive organ that reflects broader systemic and mucosal health, extending its diagnostic value beyond local structural factors.
Pharmacologic influences are equally revealing: antihistamines, decongestants, and corticosteroids tend to reduce nasal resistance by
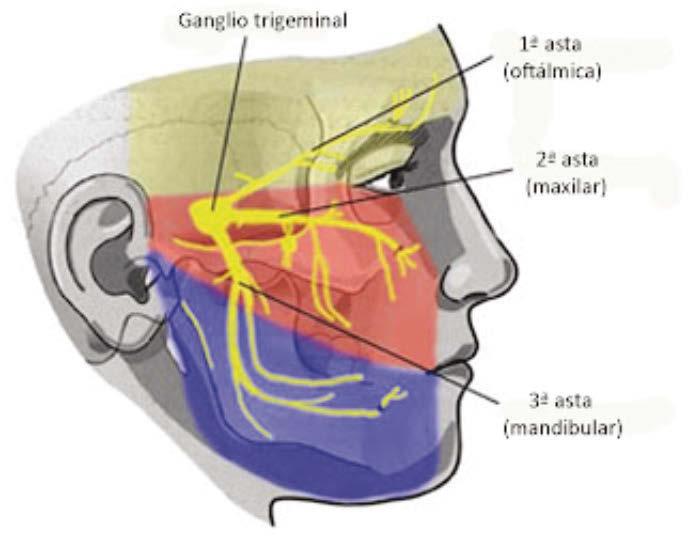
mitigating inflammation, whereas beta-blockers or sedatives may increase resistance by diminishing neuromuscular tone.8-10 Accordingly, careful measurement of nasal resistance can alert clinicians to metabolic disturbances or the physiologic impact of medications, positioning the nose positioning the nose as a sensitive, signaling organ that reflects broader systemic or pharmacologic influences.
For dental sleep medicine, nasal function is not peripheral, it is foundational. Several domains illustrate why:
1. Nasal Dysfunction Drives Mouth Breathing: When resistance rises, patients shift to oral breathing. This compensation alters jaw posture, tongue position, and craniofacial growth. In children, mouth breathing promotes long-face syndrome, high-arched palates, and malocclusion. In adults, it perpetuates clenching, bruxism, TMJ strain, and reduced appliance efficacy, and impacts oral health.
2. Nasal Resistance Predicts Therapy Success and Stability of Outcomes: Oral appliance tolerance, CPAP adherence, and even surgical outcomes correlate strongly with nasal resistance. Patients with unresolved turbulent or jetting flow often fail CPAP due to discomfort, leak, or intolerance. Similarly, oral appliances are less effective when nasal obstruction forces persistent mouth breathing.
3. Diagnostic Potential Across Systems: Airflow patterns reflect not just local
“Every breath whispering through the nose is a signal, not just of life, but of balance, resilience, and health.”
obstruction but systemic disease. Congested flow may signal allergic or infectious inflammation; paradoxical or oscillatory flows may point toward neurologic dysregulation; critical flow limitation highlights overlap with OSA physiology. Recognizing these patterns allows dentists to identify when referral, co-management, or airway testing is warranted.
4. Objective Testing Validates Clinical Insight: Tools such as 4-phase rhinomanometry and Peak Nasal Inspiratory Flow (PNIF) meter rhinometry allow for measurement of resistance, partition flow, and pressure responses in the nasal cavity. When paired with clinical exam and patient-reported symptoms, they transform subjective impressions into objective diagnostics, bridging dental, medical, and sleep disciplines.
In sleep-disordered breathing, the difference between laminar and turbulent airflow may determine whether therapy succeeds or fails
• Case 1 — Appliance Intolerance: A 48-yearold male with OSA failed oral appliance therapy due to persistent mouth breathing. Rhinomanometry revealed asymmetric partition flow (80/20), consistent with nasal valve collapse. After valve repair, appliance tolerance and AHI improved significantly.
• Case 2 — Pediatric Malocclusion: A 10-yearold girl presented with high-arched palate and crossbite. Evaluation showed chronic bypass flow (mouth breathing) driven by adenoid hypertrophy and turbinate enlargement. Adenoidectomy and orthodontic expansion restored nasal breathing, stabilizing dentofacial growth.
• Case 3 — CPAP Intolerance: A 55-year-old female discontinued CPAP due to “air leak.” Testing revealed turbulent expiratory airflow through narrowed nasal valves and concurrent hypothyroidism. After septoplasty with turbinate reduction and levothyroxine adjustment, CPAP tolerance improved, adherence increased, and symptoms resolved. These cases highlight how recognizing airflow patterns can change the trajectory of treatment.
For too long, nasal evaluation has been relegated to the ENT clinic, considered peripheral to dentistry, orthodontics, and sleep medicine. But the evidence is clear: the nose is the diagnostic compass of airway health. Its flow patterns and resistance provide real-time, physiologic insight into why patients mouth breathe, fail therapy, or relapse after orthodontic treatment.
For dental sleep practitioners, adopting a nasal-centric lens means:
• Expanding differential diagnosis beyond the oropharynx.
• Identifying hidden barriers to appliance and CPAP success.
• Recognizing systemic and neurologic contributions to airway disease.
• Partnering in multidisciplinary care with ENT, pulmonology, pediatrics, and sleep medicine.
As sleep-disordered breathing continues to challenge patients and providers alike, the opportunity lies not in adding more appliances or machines, but in re-centering on the body’s own diagnostic gateway.
The nose is not just the first step of breathing, it is the roadmap to understanding airflow, function, and ultimately, the success of sleep and dental airway care.
1. Parker Davidson K. Breathe Through Your Nose, Don’t Pay Through It: The Impact the Healthcare Industry Has on Nasal Function and How We Breathe. 1st ed. PeeDee Publishing; 2022. Revised ed. 2024.
2. Bauer SM, Vog K, Wernecke KD, Rasp, Davidson KP, Roesch S. Quantitative measurement of the flow depending nasal valve function by elastography with electro-optical distance sensors. A pilot study. Journal of Biomechanics. Published online 14 September 2024, 112326. https://doi.org/10.1016/j.jbiomech.2024.112326
3. Vogt K, Parker-Davidson K. Measurement of nasal obstruction: rhinomanometry as basic method in functional rhinology. Facial Plast Surg. Published online January 15, 2024. doi:10.1055/a-2218-5883
4. Bauer SM, Vogt K, Wernecke KD, Rasp G, Davidson KP, Roesch S. Elastometry - The biomechanical analysis of the lateral nasal wall. J Mech Behav Biomed Mater. 2024;150:106284. doi:10.1016/j.jmbbm.2023.106284
5. U P S, Kuruwatti A, Nasser A. Relationship Between Rhinitis, Nasal Obstruction and Hypothyroidism: A Follow Up Study with Pre-post Treatment with Levothyroxine. Indian J Otolaryngol Head Neck Surg. 2023;75(3):1394-1398. doi:10.1007/s12070-023-03547-1
6. Chandra RK, Lin D, Tan B, et al. Chronic rhinosinusitis in the setting of other chronic inflammatory diseases. Am J Otolaryngol. 2011;32(5):388-391. doi:10.1016/j.amjoto.2010.07.013
7. Book DT, Smith TL, McNamar JP, Saeian K, Binion DG, Toohill RJ. Chronic sinonasal disease in patients with inflammatory bowel disease. Am J Rhinol. 2003;17(2):87-90.
8. Daley-Yates PT, Larenas-Linnemann D, Bhargave C, Verma M. Intranasal Corticosteroids: Topical Potency, Systemic Activity and Therapeutic Index. J Asthma Allergy. 2021;14:1093-1104. Published 2021 Sep 8. doi:10.2147/JAA.S321332
9. Alromaih S, Alsagaf L, Aloraini N, et al. Drug-Induced Rhinitis: Narrative Review. Ear, Nose & Throat Journal. 2022;104(9):582-590. doi:10.1177/01455613221141214
10. Cingi C, Ozdoganoglu T, Songu M. Nasal obstruction as a drug side effect. Therapeutic Advances in Respiratory Disease. 2011;5(3):175-182. doi:10.1177/1753465811403348
AGD Code: 730
Date Published: January 31, 2026 Course Expires: January 31, 2029
Legal disclaimer: Course expires 3 years after publication date. The CE provider uses reasonable care in selecting and providing accurate content. The CE provider, however, does not independently verify the content or materials. Any opinions expressed in the materials are those of the author and not the CE provider. The instructional materials are intended to supplement, but are not a substitute for, the knowledge, skills, expertise and judgement of a trained healthcare professional.
Each article is equivalent to two CE credits. Available only to paid subscribers. Free subscriptions do not qualify for the CE credits. Subscribe and receive up to 8 CE credits for only $149 by visiting https://dentalsleeppractice.com/subscribe/.
n To receive credit: Go online to dentalsleeppractice.com/continuing-education/, click on the article, then click on the take quiz button, and enter your test answers
To provide feedback on this article and CE, email us at education@medmarkmedia.com.
by Karen Parker Davidson, DHA, MSA, M.Ed., MSN, APRN
1. Which of the following best describes airway resistance (Raw)?
a. The resistance to airflow only within the nasal passages
b. The opposition to airflow throughout the entire respiratory tract, from nose/mouth to alveoli
c. The resistance created by the larynx only
d. The pressure drop across the nasal valves
2. Nasal resistance (Rn) is most accurately assessed by ___________.
a. Spirometry measuring FEV₁/FVC
b. Peak flow meter readings
c. Rhinomanometry measuring transnasal pressure vs. airflow
d. Pulse oximetry
3. Which factor does NOT directly affect nasal resistance?
a. Turbinate size
b. Nasal septum deviation
c. Bronchospasm
d. Autonomic tone
4. What proportion of total airway resistance is typically contributed by the nose in normal physiology?
a. 10%
b. 25%
c. 50%
d. 75%
5. Dynamic flow in the nasal passages refers to ________________.
a. Constant, unchanging nasal resistance
b. A physiologic fluctuation in nasal resistance that can be exaggerated in disease or blunted in autonomic dysfunction
c. The airflow measured only during forced expiration
d. Airflow through the mouth when the nose is obstructed
6. How can hypothyroidism affect nasal and airway resistance?
a. By directly enlarging the nasal valves
b. Through mucosal edema, thickened secretions, and reduced
neuromuscular tone
c. By decreasing lung compliance only
d. It has no effect on airway resistance
7. Which of the following can increase nasal resistance?
a. Chronic sinusitis or nasal polyps
b. Use of intranasal corticosteroids
c. Antihistamine therapy
d. Bronchodilator therapy
8. How can nasal resistance serve as a diagnostic indicator beyond local nasal obstruction?
a. It only reflects structural nasal abnormalities and cannot indicate systemic conditions.
b. Increased nasal resistance may reflect underlying metabolic disorders, such as hypothyroidism, or the physiologic effects of medications.
c. Nasal resistance is solely influenced by allergic rhinitis and sinus disease.
d. Measuring nasal resistance cannot provide clinically useful systemic information.
9. Which respiratory condition primarily increases total airway resistance rather than isolated nasal resistance?
a. Nasal polyps
b. Asthma
c. Deviated septum
d. Allergic rhinitis
10. Which of the following medications is most likely to increase nasal resistance?
a. Beta-blockers
b. Sedatives
c. Intranasal corticosteroids
d. Tricyclic antidepressants


Obstructive sleep apnea (OSA) impacts over 30 million Americans.1 Oral appliance therapy offers many patients a path to better sleep and improved health when the devices are properly adjusted.2 Capturing accurate pulse oximetry data is critical for proper titration and ultimately, improved outcomes.3 Yet manual workflows can delay this treatment, potentially compromising care.
The Nonin Health® platform delivers a new level of efficiency, speeding workflows and allowing providers to deliver treatment more quickly with fewer manual workflows.
1. Connect: Pair the WristOx2 with the Nonin Health app.
2. Collect: Capture overnight SpO2 and pulse rate data.
3. Sync: Securely transmits data upon test completion.
4. Review: Access and analyze results anytime, anywhere.
5. Report: Use insights to guide therapy and optimize care.
The Nonin Health platform relays secure overnight oximetry data immediately upon completion of the overnight test, without the delays of manual uploads or complex software. Seamlessly integrated with the WristOx2 ® Model 3150BLE pulse oximeter, the cloud-based platform helps dental sleep professionals evaluate treatment effectiveness, optimize therapy, and accelerate care decisions.
Personalized adjustments in oral appliance therapy lead to more effective treatment and significant improvements in sleep quality, daytime functioning, and overall patient outcomes.2
• Cloud-Based Data Management — Secure, encrypted storage eliminates manual transfers. Data is available
anytime, anywhere through the Nonin Health web portal.
• Patient-Centered Accuracy — Built for patient comfort and real-world usability, the WristOx2 pulse oximeter helps ensure accurate4 data is available for oral appliance titration.
• Scalable & Flexible Design — Easily integrates with existing workflows — no steep learning curve or custom setup required.
The Nonin Health platform is more than a data portal. It’s a complete system designed to simplify dental sleep workflows, saving time for providers and accelerating therapy for patients. Enabling early intervention and tailored adjustments supports stronger outcomes.2
Learn more about how the Nonin Health platform can streamline your oximetry workflows at nonin.com/nonin-health.
1. American Academy of Sleep Medicine; https://sleepeducation.org/get-involved/count-on-sleep/public-health/ national-indicator-report/
2. Sutherland K; Vanderveken OM; Tsuda H; Marklund M; Gagnadoux F; Kushida CA; Cistulli PA; on behalf of the ORANGE-Registry. Oral appliance treatment for obstructive sleep apnea: an update. J Clin Sleep Med 2014;10(2):215-227. https://pmc.ncbi.nlm.nih.gov/articles/PMC3899326/
3. Metz JE, Attarian HP, Harrison MC, Blank JE, Takacs CM, Smith DL and Gozal D (2019) High-Resolution Pulse Oximetry and Titration of a Mandibular Advancement Device for Obstructive Sleep Apnea. Front. Neurol. 10:757. doi: 10.3389/fneur.2019.00757 https://www.frontiersin.org/journals/ neurology/articles/10.3389/fneur.2019.00757/full
4. Nonin Medical, Inc. Data on file
The Nonin Health® platform is an adjunct system requiring interpretation of the results by a medical professional. It does not suggest a course of treatment or generate a diagnosis.


The Nonin Health® platform provides you with fast, accurate1 oximetry data to help optimize therapy and accelerate care.
Patient-Centered Accuracy
Comfortable, noninvasive testing for accurate1 titration data.
Scalable for Any Practice
Seamless integration into existing workflows.

Cloud-Based Data Management
Secure, remote access to overnight SpO2 data.
Reduce complexity and improve efficiency with no manual uploads, saving time and operational costs.
by Eric Stefanik, IT Security & AI Expert, Elliptic Systems Corporation
Artificial intelligence (AI) is rapidly reshaping dental sleep medicine, transforming how practices diagnose sleep apnea, streamline operations, and enhance patient care.
However, while dentists recognize AI’s potential, many feel uncertain about its practical implications or fear unintended exposure of protected health information (PHI). This is precisely why your dental sleep practice needs an AI consultant, someone who understands your curiosity, clears confusion, and mitigates your security fears.
AI Beyond ChatGPT: Understanding Its Practical Value
Dentists today are familiar with AI primarily through applications like ChatGPT, but the true value of AI extends far beyond text generation. In dental sleep medicine, AI
significantly enhances clinical efficiency and patient outcomes by:
• Speeding Up Diagnostics: AI-powered systems, such as Sleepifi and EnsoSleep, accelerate sleep apnea diagnosis by accurately analyzing patient data from home sleep tests. This reduces waiting times, accelerates treatment, and boosts patient satisfaction.
• Customizing Patient Treatment: AIdriven design software enables rapid and precise creation of personalized oral appliances, significantly improving patient comfort and compliance.
• Automating Administrative Tasks: AI technologies streamline scheduling, billing, and patient follow-ups, freeing your staff from repetitive tasks and allowing more focus on patient care.

Yet, despite these clear benefits, many dentists hesitate. The reason often boils down to two factors: uncertainty around integrating AI into existing workflows, and legitimate security fears about data exposure.
AI can seem intimidating because integrating these advanced technologies into a busy dental practice feels overwhelming. Dentists naturally wonder:
• Which AI tools are right for my practice?
• Will the learning curve disrupt my workflow?
• Is the initial investment in AI worth the return?
An AI consultant directly addresses these concerns by assessing your specific practice needs, recommending the right AI tools, and developing straightforward integration plans. Rather than a one-size-fits-all approach, consultants tailor AI solutions precisely to your operational structure, minimizing disruptions, and maximizing immediate benefits.
The greatest hesitation among dental professionals when considering AI is the potential exposure of sensitive patient information. With dental sleep medicine increasingly involving multiple medical offices and extensive data sharing, the risk of cyberattacks and inadvertent data leaks grows exponentially. Without specialized guidance, integrating AI can inadvertently expose practices to regulatory violations, costly data breaches, and damage to patient trust. An AI consultant with expertise in healthcare cybersecurity is uniquely equipped to:
• Evaluate Risk: Identify potential vulnerabilities in your current systems and recommend secure AI tools specifically designed for healthcare environments.
• Ensure Compliance: Guide your practice through HIPAA and related regulatory frameworks, ensuring your AI integrations are secure and fully compliant.
• Implement Secure Practices: Establish rigorous data security protocols and continuous monitoring strategies to
safeguard patient data from unauthorized exposure.
By addressing these concerns directly, an AI consultant turns hesitation into confident action.
An AI consultant doesn’t merely recommend new software. They actively demystify AI, answer your specific questions, train your team, and ensure that everyone – from practice administrators to clinical staff – understands and benefits from these technologies.
Imagine an AI solution that automatically screens patients at risk for sleep apnea, instantly sends recommendations for further testing, securely handles all PHI, and even predicts scheduling needs months in advance. This isn’t futuristic — it’s achievable today, with guided integration.
AI adoption in healthcare is inevitable; practices that hesitate risk being left behind. The decision isn’t merely about technological advancement, it’s about enhancing patient care, operational efficiency, and maintaining competitive advantage in your market.
Working with an AI consultant ensures your practice can confidently embrace technology, secure patient information, simplify complex integrations, and unlock AI’s full potential for clinical excellence and business growth.
In dental sleep medicine, AI is transformative but understandably intimidating. With an experienced AI consultant, you don’t have to navigate this transformation alone. Address your curiosity, clear your confusion, and remove security anxieties – your practice and your patients will greatly benefit.

Many dentists fear unintended exposure of protected health information.
Eric Stefanik is a recognized cybersecurity authority and AI consultant with over two decades of experience. As the founder of Elliptic Systems Corporation, he specializes in helping healthcare practices integrate AI solutions securely and effectively. For information, visit https://IsAIForYou.com.
by Katrina Sanders, RDH, BSDH, MEd, RF
The Epidemic You Can’t See Airway disorders are everywhere — and yet almost no one is talking about them. As dental hygienists, we spend our careers with a front-row seat to the changes of the mouth, the muscles of the oropharynx, and subsequently the breath — but how often are we encouraged to look beyond the prophy and into the passageways of the airway itself?
It’s estimated that 1 in 5 adults has some form of sleep-disordered breathing, and most of them don’t even know it. They’re undiagnosed, untreated, and unfortunately, deteriorating.
What begins as loud snoring or mouth breathing can evolve into fragmented sleep, chronic fatigue, and systemic inflammation — all of which are intimately linked to various systemic conditions such as hypertension, myocardial infarction, cerebrovascular accident, Type 2 diabetes, and even Alzheimer’s Disease. While these patients may not be rushing to their physicians complaining about tired jaws or scalloped tongues, they are sipping triple-shot lattes just to make it through their day.
We live in a world that glorifies the grind and caffeinates the crash. Our patients fall asleep at the wheel — literally and figuratively — struggling to concentrate at work, failing to achieve in school with diagnoses of “ADHD,” and slowly surrendering to a chronic health crisis that’s masquerading as “just tired.”
Most medical doctors receive less than two hours of education on sleep medicine in the entirety of their medical training despite what we know about the importance of
proper sleep. After all, poor sleepers become poor healers. When patients finally mention fatigue or brain fog, they often leave with a prescription for a sleep aid instead of a rootcause investigation.
Dental hygienists: where we come in!
As dental hygienists, we are trained and effective preventive professionals. We do not wait for pathology to show up — we screen for risk, we educate, and we act accordingly. We are trained to assess not just for disease, but for the deep-rooted underlying causes of it. This makes us uniquely positioned to recognize the signs of airway dysfunction in our patients long before a diagnosis ever comes.
If your doctor is not currently screening for or actively treating airway, that does not mean you cannot. Here is how to begin assessing risk through four primary lenses:
• High blood pressure, especially if poorly controlled
• Type 2 diabetes or insulin resistance
• History of GERD or acid reflux
• ADHD (especially in children), depression, or anxiety
• TMD or frequent migraines
• Frequent nighttime urination (nocturia)
Vital
• Elevated blood pressure or pulse
• Low oxygen saturation (if using pulse oximetry)
• Obesity or high BMI
• Large neck circumference (>17” in men, >16” in women)
Head and Neck Observations
• “Allergic Shiners” of bags or dark circles under the eyes indicating poor oxygenation during sleep
• Droopy eyelids and poor skin tone
• Mouth-breathing or open lip posture
• Scalloped tongue or noted tongue thrust
• Enlarged tonsils or ankyloglossia (restricted tongue movement)
• Forward head posture and curvature of the spine
• High, narrow palate or visible malocclusion
• Bruxism, wear facets or fractured restorations
Patient-Reported Symptoms
• Chronic fatigue or excessive daytime sleepiness
• Snoring (self or reported by partner)
• Waking up gasping or choking
• Dry mouth in the morning
• Trouble falling or staying asleep
• Poor concentration, memory lapses, or irritability
Many dental hygienists are surprised to realize they’re already gathering much of the information needed to screen for airway issues without even thinking about it. From reviewing medical histories and taking vital signs to observing oral posture, scalloped tongues, and dried oral tissues, these findings are often already part of a standard hygiene assessment.
Adding airway awareness does not typically require a complete overhaul of your existing workflow — it’s simply a matter of connecting the dots. When you begin listening differently, looking with intention, and asking focused questions, airway screening becomes a natural extension of what you are already doing. The goal is not to diagnose or treat — it is to identify red flags early, educate your patients, and advocate for the care they may not even know they need.
You do not need a full sleep lab in your op to start airway conversations. These simple screening tools help identify patients at risk:
STOP-BANG
A quick and validated screening tool for
obstructive sleep apnea (OSA), using eight yes/no questions. A score of 3 or more suggests moderate to high risk for OSA.
• S – Snoring: Do you snore loudly (louder than talking or loud enough to be heard through closed doors)?
• T – Tiredness: Do you often feel tired, fatigued, or sleepy during the daytime?
• O – Observed Apnea: Has anyone observed you stop breathing or choking/ gasping during your sleep?
• P – Pressure: Do you have or are you being treated for high blood pressure?
• B – BMI: Is your Body Mass Index over 35?
• A – Age: Are you over 50 years old?
• N – Neck Circumference: Is your neck circumference greater than 17 inches (men) or 16 inches (women)?
• G – Gender: Are you male? (Men are at higher risk for sleep apnea.)
Score of 3 or more = further evaluation is recommended.
This is a visual assessment of airway space using tongue position (see figure 1). For an accurate Mallampati Score assessment, the patient should be positioned as follows:
• Seated upright with the head in a neutral position (not tilted).
• Mouth fully open as wide as possible.
• Tongue maximally protruded without phonating (no speaking or saying “ahh”).
• Patient should be breathing quietly during the assessment.
• Class III or IV may suggest compromised airway space.

“We are trained to assess not just for disease, but for the deep-rooted underlying causes of it. This makes us uniquely positioned to recognize the signs of airway dysfunction in our patients long before a diagnosis ever comes.”
A clinical dental hygienist, author, and international speaker, Katrina Sanders, RDH, BSDH, M.Ed, RF, is passionate about elevating the dental profession by creating an undeniable movement that educates, encourages, and empowers the profession to rise in its power. Known as the “Dental WINEgenist™,” she pairs her desire for excellence in the dental industry with her knowledge and passion for wine. She is the Clinical Liaison for Hygiene Excellence at AZPerio, founder of Sanders Board Preparatory and has been published in various publications including RDH Magazine and Today’s RDH. Recently, Katrina proudly received the University of Minnesota Distinguished Alumni Award and the 2024 Sunstar Award of Distinction. Follow her @TheDentalWINEgenist.
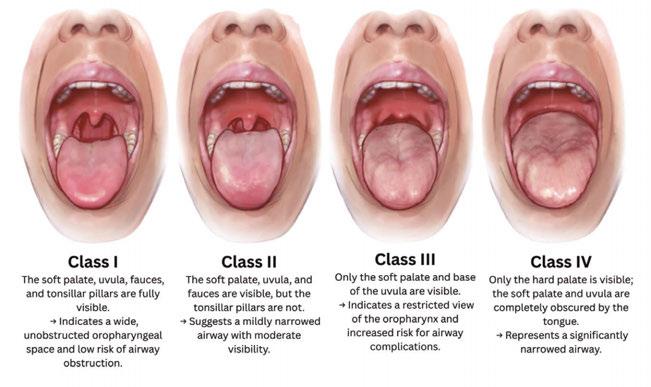
In today’s tech-savvy world, many patients are already wearing devices like the Oura Ring, Apple Watch, or Fitbit that track sleep quality, oxygen saturation, heart rate variability, and even breathing patterns during the night. These wearable technologies often give early insight into disrupted sleep or signs of potential airway issues — sometimes before the patient even realizes there’s a problem. By opening up a conversation about their sleep data or wearable insights, we may discover that patients have already noticed concerns like frequent waking, low sleep scores, or restless nights. These conversations not only validate the importance of airway screening but also position us as collaborative partners in our patients’ overall wellness.
What do you do when your doctor isn’t interested in sleep or airway treatment? You advocate anyway.

• Screen and Educate: Start the conversation. Patients trust you. Your role is to inform, not diagnose.
• Refer with Purpose: Collaborate with airway-aware providers: sleep dentists, ENTs, myofunctional therapists, or sleep physicians.
• Champion Myo: Consider myofunctional therapy referrals for children and adults with oral habit concerns, poor posture, or tongue dysfunction.
• Document It: Note clinical findings in the chart — especially risk factors or
observations that may require future follow-up.
• Build a Network: Connect with professionals who do treat airway, so you have a go-to referral source when your patient needs help beyond your scope.
There are many educational opportunities available to help interested professionals explore airway management and help empower themselves to success such as:
• Free Podcasts: A few of my favorites include Beauty of Breathing with Renata Nehme and Airway, Sleep & TMD Podcast with Jamison Spencer
• Free Educational Programs: Educational organizations like Airway Health Solutions offer content through their “AHS Conversations” platform.
• Deep Dive Programs: Institutions like The Vivos Institute have created comprehensive and immersive programs designed for a comprehensive approach to airway management.
• Certifications: Consider pursuing a certification in Myofunctional Therapy. Organizations like the Academy of Orofacial Myofunctional Therapy (AOMT) or the International Association of Orofacial Myology (IAOM) offer in-depth training programs that can transform how you evaluate and co-manage oral function and structure.
You do not need your dentist’s buy-in to start noticing. You do not need a prescription pad to educate. And you do not need a CPAP machine to save a life.
You just need the confidence to trust your clinical intuition and the courage to ask one simple question: “Are you breathing well while you sleep?”
Because your patient’s next breath may depend on it.
This article is dedicated to my mother, Linda Marie Sanders, whose beautiful life was taken too quickly from an airway obstructive disorder that remained untreated despite visiting her ENT the day she tragically collapsed. May her story transform from tragedy to legacy in the ways her preventable death inspires others to rise above and serve their suffering patients with excellence. We miss you, Mom.
ADHERENCE AIRWAY-CENTRIC ATHLETES
BIOSTIMULATION
BRUXISM COGNITION

WORD
CRANIOFACIAL DATA DIGNITY DYSFUNCTION
NEURODEGENERATION
NEUROMUSCULAR NOSE
PERIMENOPAUSE
TELEDONTICS
TONGUE
For the solution, visit www.dentalsleeppractice.com.
• In-Network options with major payors and plans
• Out-of-Network when appropriate
• Reliable, timely, predictable results with best rates
• Access to the Nexus Specialty Physician Network
• Nexus billing por tal ensures error-free claim submission and tracking
• Personal billing representative
• Complete solution for:
º Sleep Medicine
º Airway Expansion
º TMJ/TMD
º Botox
º CBCT

Getting reimbursement from medical insurance can be a nightmare for dentists. With the Nexus Bill system, you can avoid the common pitfalls of inconsistent payments, endless delays, constantly fighting with insurance companies, and patients refusing care because you are not “In-Network” for medical.
