










The Ottawa Heart Institute is advancing coordinated, research-driven care for rare cardiac conditions across Canada. Canadian Centre for Rare Cardiac Conditions

Rare heart conditions affect millions of people worldwide, yet for years they received limited attention due to gaps in research, diagnosis, and treatment. That’s changing at the Ottawa Heart Institute, where the Canadian Centre for Rare Cardiac Conditions (CCRCC) is leading a new, coordinated approach to care.
Most rare heart diseases have a genetic cause and are a significant contributor to heart failure and sudden cardiac death in younger adults. Diagnosing them can be challenging, often requiring specialized genetic testing and advanced imaging.
Under the leadership of Dr. Ian Paterson, a cardiologist and director of the CCRCC, patients benefit from integrated care that brings diagnosis, treatment, research, and education together. “We identified a significant gap in coordinated care for patients with rare cardiac conditions across Canada,” says Dr. Paterson.
By combining cutting-edge science with national collaboration and education, the Ottawa Heart Institute is improving access to care and offering new hope for people living with rare forms of heart disease.


In Canada, about 1 in 12 people live with a rare disease, including Duchenne muscular dystrophy, a severe and progressive neuromuscular disorder. For families affected by Duchenne, time matters. Irreversible muscle loss continues daily, and delays in treatment can have lasting consequences.
This Rare Disease Day, Defeat Duchenne Canada is calling on communities to stand together and advocate for affordable, timely access to treatments.
In October 2025, Health Canada approved AGAMREE

This Rare Disease Day 2026, Canadian families living with rare disease have much to celebrate — and even more to look forward

T(vamorolone), the first treatment for Duchenne in Canada, marking a long-awaited milestone. But approval is only the first step. Reimbursement decisions are still needed to ensure equitable access across the country.
While multiple Duchenne therapies are available in other countries, Canadian families continue to wait. These delays affect mobility, independence, and quality of life.
Rare Disease Day is a moment to turn awareness into action and push for meaningful, timely change.

Durhane Wong-Rieger President & CEO, CORD
hrough Canada’s National Strategy for Drugs for Rare Diseases, patients with serious and life-threatening rare diseases are gaining faster access to innovative therapies.
For those living with rare conditions, time matters. Earlier access to treatment can reduce the risk of irreversible organ or tissue damage, preserve function, slow or stabilize disease progression, prevent relapses, and, in many cases, prolong survival. The nine therapies currently on the new strategy’s common list of drugs — with more to come — are not incremental advances. They’re breakthroughs: safer, more effective treatments and, in some cases, options where none previously existed.
Faster and more equitable access also creates opportunity. As patients across Canada begin treatment earlier, health systems are better positioned to observe, measure, and understand the full impact of these innovations. The next phase of Canada’s National Strategy for Drugs for Rare Diseases must focus on collecting real-world evidence that documents how these therapies improve outcomes, extend lives, reduce complications, and enhance quality of life.
Turning access into lasting impact
The common thread across the list of rare disease therapies is clear: each addresses serious unmet needs and delivers meaningful patient value. Demonstrating that value clearly and credibly will help ensure sustained and equitable access across the country.
Similarly, Ontario’s Funding Accelerated for Specific Treatments (FAST) program reflects a broader evolution in policy thinking — that timely access and evidence generation can proceed together. While not yet applied to rare-disease drugs, FAST signals how complementary accelerated pathways could further strengthen national progress.
On Rare Disease Day, we celebrate momentum — and commit to sustaining it. Canada’s National Strategy for Drugs for Rare Diseases has shown that faster access is possible when patients are placed at the centre of decision-making. The task now is to ensure that this progress endures, so that timely access to life-changing therapies becomes standard practice, not a rare exception.


We all produce and need cholesterol. But high amounts of the wrong kind can put us at risk of cardiovascular disease.
Anne Papmehl

Dr. Amanda Berberich Assistant Professor, Western University & Endocrinologist, Division of Endocrinology & Metabolism, St. Joseph’s Health Care
Cholesterol is a fat that’s found in our blood. Our bodies make it naturally, but it can also be increased through our diet. Cholesterol comes in two forms: low-density lipoprotein (LDL-C) and high-density lipoprotein (HDL-c).
LDL-C is often referred to as “bad cholesterol” and for valid reason.
“LDL particles carry high concentrations of cholesterol that can enter the blood vessel walls and cause plaque buildup. Over time this can cause narrowing of the arteries, which increases the risk for heart attack and stroke,” says Dr. Amanda Berberich, Assistant Professor, Western University, and Endocrinologist, Division of Endocrinology & Metabolism, St. Joseph’s Health Care in London, Ontario. HDL-c, on the other hand, is often referred to as “good cholesterol” because it works to remove excess cholesterol from your body.
Cholesterol is measured through a blood test. A LDL-C reading below 3.0 mmol/l for children, adolescents and adults is considered ideal, while anything above 4.0 is considered high.
Once cardiovascular disease develops, it’s much harder to reverse or even stabilize the process. Conversely, if treatment begins early and cholesterol levels are well controlled, the risk is minimized.
Genetically inherited high LDL-C conditions
Numerous factors can contribute to high cholesterol, such as nutrition, physical activity levels, and other medical conditions. However, some people have an inherited genetic condition known as familial hypercholesterolemia (FH) which makes them unable to remove the LDL particles from their bloodstream. “As a result, they can have more than double the normal amounts of LDL-C, which puts them at risk for early heart attack and stroke,” says Dr. Berberich. There are two types of FH — heterozygous hypercholesterolemia (HeFH) where the patient has one defective gene (HeFH affects about one in 250 Canadians 1) and homozygous familial hypercholesterolemia (HoFH) where the patient has two defective genes (this is rarer and affects about one in 300,000 Canadians, although French Canadians have a higher prevalence).
Complicating matters is that patients with FH may be undiagnosed until they’ve had their first cholesterol screening at age 40. “By this point significant disease has often
already developed and the window to intervene early to prevent disease onset has lapsed,” says Dr. Berberich. “Even when a level is checked, not all providers are aware of this potential diagnosis and the dangers it poses, and levels may be left unaddressed,” she says.
Few visible symptoms of LDL-C
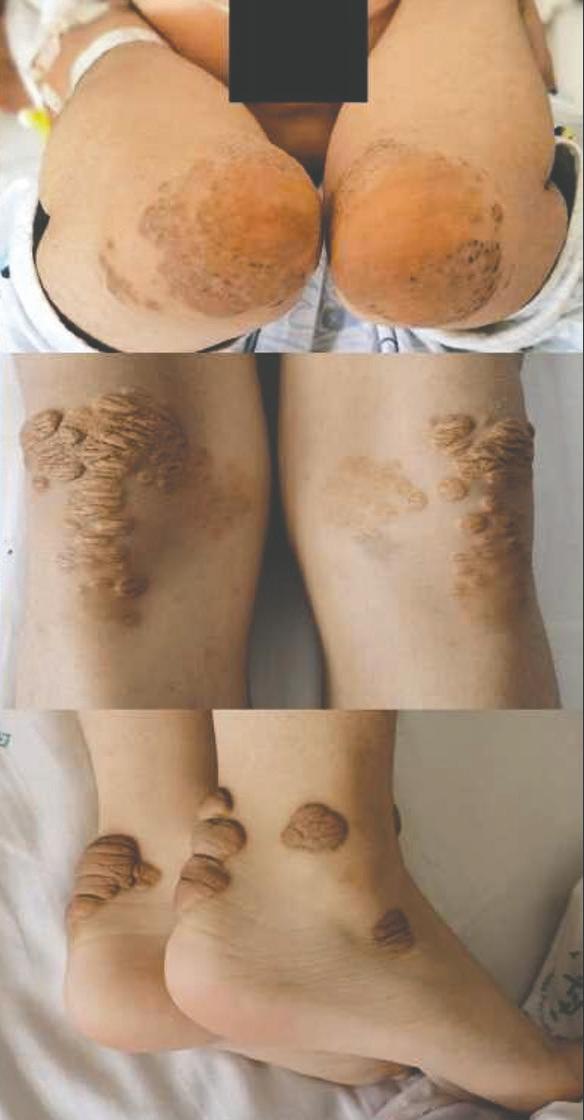
One of the challenges of high LDL-C is that it presents with few signs or symptoms. However, one physical manifestation is yellowish, fatty deposits on the tendons of the hand, Achilles tendon, other exterior tendons (tendon xanthomas) or in the corners of the eyes (xanthelasmas). “These often appear as yellowish, irregular bumps within or on the surface of the tendon,” says Dr. Berberich. “A yellow-whitish crescent in the lower portion of the cornea in an individual under age 45 can also be a sign,” she says.
For many individuals, however, there are often no signs of high cholesterol until it causes a heart attack or stroke. This may even be preceded by many years of silent, asymptomatic accumulation of plaque on the artery walls. “As plaque volume accumulates to a critical level, some patients start to develop chest tightness or shortness of breath when they become more active or experience cramping in the leg or back of the calf,” she says.
High LDL-C can indicate other, treatable conditions
While high LDL-C levels can be a sign of other treatable medical conditions such as hypothyroidism and diabetes, they most commonly signal a risk for cardiovascular conditions. The only way to get an accurate diagnosis is by having your cholesterol levels checked through a blood test.
An early and accurate diagnosis is critical to allow for early intervention and improved lifelong outcomes. “Risk is directly proportional to the amount of exposure to high cholesterol, so the higher an individual’s cholesterol and the longer they’re exposed, the greater the risk for disease development,” says Dr. Berberich. “Once cardiovascular disease develops, it’s much harder to reverse or even stabilize the process. Conversely, if treatment begins early and levels are well controlled, the risk is minimized,” she says.
What to do if you have high LDL-C It’s important for everyone, including children, to get their LDL-C levels checked once. If you have high LDL-C, you want to address it immediately. Standard treatment typically involves
lifestyle changes and medication. Eating a healthy diet, exercising, not smoking, maintaining a normal weight, and stress management are a good place to start but may not be sufficient on their own.2 “Most individuals will be started on a statin medication, which decreases cholesterol production in the liver,” says Dr. Berberich. “Other options, such as cholesterol inhibitors or PCSK9 inhibitors, which improve the clearance of cholesterol from the blood, may also be required if cholesterol levels start very high. For some of the rare genetic syndromes like HeFH and HoFH, we have targeted management strategies,” she says.
Before any treatment can begin, you need to know your LDL-C levels.
“For individuals who may have FH, this is especially important as many are undiagnosed,” says Dr. Berberich. Because children with HoFH are at higher risk of early coronary events and death from heart attack than adults, early diagnosis and treatment are crucial to ensuring long-term survival. 3
This complex rare disease highlights why patient advocacy is critical to accessing better treatments and increasing clinical understanding overall.
Sonya Friesen

Derry Wilcox Treasurer, Canadian Association for Porphyria
Acute Hepatic Porphyrias (AHP) is a group of genetic disorders that presents differently in every patient. AHP is a misunderstood, misdiagnosed condition. Patients may experience sudden attacks including severe symptoms like confusion, insomnia, abdominal pain, extreme fatigue and tachycardia making it difficult to complete even the simplest of daily tasks. With a mean diagnosis delay of up to 15 years, awareness is critical to ensuring patients understand their treatment options, which can help alleviate symptoms.
Acute Hepatic Porphyrias disorders are a direct result of the body’s inability to properly produce heme, a molecule integral to iron production, which carries oxygen, produces energy and detoxifies chemicals. It may be just a tiny piece of our body’s puzzle, but when heme production is defective, the result is a rare genetic disorder (like AHP) that appears when toxic substances — porphyrins — accumulate, damaging our nervous system.
Understanding is critical for prevention It is unclear how rare AHP is. While current estimates range from 1 in 500 to 1 in 50,000, AHP is often genetic and can remain dormant leading to low diagnosis rates. AHP is characterized by sudden ‘attacks’ which vary in severity but typically appear as extreme abdominal pain, brain fog, and tachycardia. For those experiencing these symptoms, it’s important

to discuss porphyria with a physician as the condition can often go misdiagnosed. Sometimes as quickly as they come, attacks disappear, making formal diagnosis challenging. For Derry Wilcox, Treasurer for the Canadian Association for Porphyria, while attacks caused an inability to complete even the basic functions of day-to-day life, it was the chronic symptoms that made life especially difficult: “Between attacks, I often experience chronic pain, nausea and fatigue. It is a constant daily struggle to just make it through.”
Like any rare disease, AHP suffers from a lack of awareness. Attacks can be sporadic but, in a particularly vicious cycle, are also necessary to the diagnostic process. During an AHP attack, heme precursor levels are highest, meaning that diagnostic tests can only be completed reliably while someone is experiencing an attack. Once it subsides, levels go back to normal. While this urgency is necessary for proper diagnosis, clinical disease understanding is low, amounting to further diagnostic challenges: “One limitation is the lack of familiarity with ordering and administering medications which can cause delays in treatment,” explains Derry.
A brighter future for AHP treatment
Typically, treatment for AHP attacks is IV therapy, which replenishes heme while suppressing the overproduction of toxic substances. It’s given reactively and requires patients to present
From years-long diagnostic journeys to breakthrough treatments using existing drugs, Dr. Luke Chen explains how collaboration, persistence, and the next generation of physicians are helping bring clarity—and hope—to patients living with rare diseases.
Many people living with rare diseases spend years searching for answers. From your experience, why are rare conditions so difficult to diagnose—and what can patients do when they feel something is being missed?
Rare diseases are difficult to diagnose because many physicians may have never seen them or heard of them. I established the Coastal Rare Inflammatory Diseases Program to improve the care of patients with rare inflammatory blood diseases like HLH, Castleman disease, and histiocyte disorders.
For patients, sometimes a simple internet search combining key medical problems can provide useful information from a reputable source or highlight specialists with expertise in a rare condition. If you find relevant information from a credible source, bring it to your physician. If you do not have access to a local specialist who can help, it may be reasonable to reach out to the relevant society or international expert for the condition in question.
You’re involved in repurposing existing medications to treat rare diseases through collaborations like Every Cure. What does drug repurposing mean in simple terms, and why can it be life-
Between attacks, I often experience chronic pain, nausea and fatigue. It is a constant daily struggle to just make it through.
at hospital during an attack. While offering patients relief from their symptoms, IV therapy doesn’t treat the underlying cause. However, newer once-monthly therapies now exist that may prevent recurrent attacks, which could reduce hospitalizations and the need for IVs. New therapies are effective, but patients must still meet specific criteria to access, including documented evidence of attacks at a hospital level. Unfortunately, attacks cause such extreme pain that many patients either can’t get to the hospital or choose to weather the worst at home.
While hope is on the horizon and new treatments like gene therapy continue to be explored, regulatory challenges make the availability of these treatments difficult. In Canada, these processes are known to be long and even once approved, expensive.
As with many rare conditions, patient advocacy continues to be important not only by increasing awareness for AHP, but by putting increased pressure on reimbursement bodies to make these essential treatments available to patients who desperately need them.

changing for patients with few treatment options?
Many rare diseases do not have a dedicated medication, and so we need to repurpose existing drugs. I collaborate with Dr. David Fajgenbaum and Every Cure, who leverage artificial intelligence, traditional science, and clinical data to uncover and validate new uses for existing drugs. One of our collaborations, led by Dr. Emily Leung at UBC, highlights lenalidomide for the treatment of Rosai-Dorfman disease (RDD). Traditional treatments for RDD are expensive and hard to access in low income countries; we have shown that lenalidomide, which is inexpensive and widely available, is also an effective option.
Many rare diseases do not have a dedicated medication, and so we need to repurpose existing drugs.


You also mentor young physicians who are helping solve rare disease cases early in their careers. How does training the next generation of clinicians ultimately improve care and outcomes for patients?
I’ve been very fortunate to have philanthropic support from the Hsu & Taylor Family in Vancouver to supervise student and resident research across Canada. For example, Dr.
Mariam Goubran (Vancouver) and Dr. Steven Rowe (Memorial) discovered simple blood tests that helped us distinguish TAFRO from HLH. Dr. Caroline Spaner and Dr. Derrick Chong (Toronto) have done fantastic work using blood tests and skin biopsies to diagnose Still’s disease. Dalhousie science students (and varsity soccer players) Richard Schwarzkopf and Andrew Chen described repurposing ruxolitinib to treat severe immune neutropenia — the first new treatment for this condition in over 30 years!
These are just a handful of the many trainees I’ve been privileged to mentor, and I’m so excited about the future when I see the energy and enthusiasm they bring.