




The Broadway star discusses how his ALS journey catalyzed his Grammy-nominated album and research mission
Read more on Page 05
“While sequencing at birth will allow earlier diagnosis and intervention for numerous conditions, its potential reaches far beyond infancy.”
Mira B. Irons, M.D., President, and Michelle McClure, Ph.D., Director of Public Policy, American College of Medical Genetics and Genomics
“When patients and caregivers are engaged early in the research process and remain active partners throughout, diagnostic journeys shorten.”
The biopharma business model is misaligned with rare diseases, and children are paying the price.

RINTERVIEW WITH Craig Martin Founder, Orphan Therapeutics Accelerator (OTXL)
ecent policy and market changes have sparked a dramatic shift in investment toward therapies with broad commercial demand — leaving hundreds of rare disease programs shelved, often despite promising clinical data. Many of these conditions are pediatric, progressive, and fatal, making delays especially costly.
“There have been huge leaps forward in genetic medicines to treat rare diseases, but the business model to support their advancement hasn’t evolved at the same pace,” shared Craig Martin, who founded the Orphan Therapeutics Accelerator (OTXL) to reimagine how rare disease treatments are financed and advanced.
As a nonprofit, OTXL can incentivize companies to donate programs and negotiate deferred or reduced development costs with industry partners.
In partnership with the American Society for Gene and Cell Therapy, OTXL is also building an AI-powered marketplace to surface shelved assets and match them with aligned funders, opening the door to family offices, impact investors, and others historically excluded from this space.
Soon, OTXL plans to enable fractional investment — another pragmatic, scalable alternative to the current funding model that helps accelerate paths to patients.
Written by Emily Rose
To learn more, visit orphantxl.com
Rare diseases aren’t as rare as you might think. An estimated 1 in 10 Americans has one, and more than 50% are children, many of whom are still undiagnosed.
On average, it takes five years to get an accurate rare disease diagnosis. That’s five years of unanswered questions, misdiagnoses, and missed opportunities for early interventions that could change the trajectory of a child’s health and even save their life.
The National Organization for Rare Disorders (NORD®) is hoping to reduce the diagnostic journey by sharing steps to take when your child has unresolved health issues:
1. Trust your gut
No one knows your child better than you do. If something feels off — persistent symptoms, unusual developmental delays, or a pattern doctors can’t explain — listen to that inner voice. Parents and caregivers often recognize a problem long before it’s identified medically.
2. Partner with your physician
Your child’s pediatrician should be your first stop. Bring them a detailed symptom log, and ask questions about potential underlying conditions. If your concerns aren’t being addressed or you don’t feel heard, seek a second opinion.
3. Be your own advocate

Advocating for your child also includes taking care of yourself. NORD offers resources like patient assistance programs for nearly 80 rare diseases, as well as caregiver respite services to help lighten the load.

4. Educate yourself
Knowledge can help you feel more in control. If you or your child receives a rare disease diagnosis, the NORD® Rare Disease Database offers information on over 1,400 rare conditions, treatment options, and clinical trials with nearly 500 reports available in English and Spanish.
5. Build your support network
The diagnostic journey can be isolating, but you do not need to go at it alone. Connecting with other parents who have walked this path can provide comfort, guidance, and invaluable advice. There are many opportunities to link up with rare disease communities through social media and patient advocacy organizations.
6. Participate in research
Fewer than 5% of rare diseases have FDAapproved treatments. New treatments rely on families participating in research and drug development. Many breakthroughs have been driven by families involved in clinical trials, registries, and genetic studies.
7. Don’t give up
The rare disease journey can be long, filled with twists and uncertainties. Yet, progress is happening every day, and with persistence and the right support from organizations like NORD®, answers and help are possible.
Written by National Organization for Rare Disorders (NORD®)
My daughter, Juno, was diagnosed with a very rare neurodevelopmental condition called STXBP1-related disorder after a three-year diagnostic journey. When we received Juno’s diagnosis, I remember feeling utterly alone. I slowly began to meet other families, and together we started a foundation. We began driving forward collaborative research, community support, and awareness for STXBP1. This experience as a next-generation advocate led me to think about ways to advance work across rare diseases, which led me to become CEO of Global Genes.
Next-generation advocacy, the center
of Global Genes’ mission and vision, allows individuals to utilize the tools and resources needed to accelerate advocacy efforts and support opportunities to drive research and therapy development.
Next-gen advocates work in many ways to accelerate progress for their disorders, and their work spans diagnosis, becoming disease experts, starting a nonprofit organization, all the way to doing research and working with pharmaceutical companies.
Our scientific advisory board, partners, advocates, and mental health professionals are helping to identify the best approach and strategy for
Global Genes to support patients, caregivers, and advocates. In addition, health equity grants are open to Global Advocacy Alliance members to develop initiatives to improve outreach strategies and address challenges that affect underserved and underrepresented communities.
In the area of research enablement, we are catalyzing early work and research that will help advocates entice interest from research partners, including biopharma. There has never been more opportunity for patient advocates to get started with new therapies and treatments in the research readiness realm.

Sponsored
Prader-Willi syndrome is a rare genetic condition marked by uncontrolled hunger and complex behavioral and medical challenges requiring lifelong management, supervision, and coordinated support.
Prader-Willi syndrome (PWS) affects an estimated 10,000 to 20,000 individuals in the United States. Caused by the lack of expression of paternally inherited genes on chromosome 15, PWS disrupts hunger regulation, behavior, cognition, and emotional well-being, often requiring lifelong, coordinated medical, behavioral, and social support.
The defining feature of PWS is hyperphagia, a chronic condition marked by extreme hunger, constant thoughts about food, and an urge to eat that cannot be satisfied. Unlike typical hunger, hyperphagia can drive compulsive food-seeking behaviors, emotional distress, and serious medical risks. Families often
maintain strict food security and constant supervision to ensure safety.
PWS also involves growth hormone deficiency, low muscle tone, endocrine disorders, and developmental delays. Often diagnosed at birth, ongoing care from an endocrinologist is critical, along with an individualized treatment plan. Access to information on PWS and specialists with PWS knowledge can shape long-term outcomes.
Stacy Ward, PWSA|USA CEO, notes, “Families impacted by PWS know they can count on specialist clinicians for answers, guidance, and real support. However, access to clinicians with experience in PWS remains inconsistent, leaving some to navigate with limited guidance.”
Families face ongoing emotional and social challenges, making education, advocacy, and connecting with others in the PWS community critical sources of support. PWS awareness helps ensure individuals and families are supported not just in clinical settings, but across schools, communities, and systems of care.
“The PWS community shows every day how powerful connection, advocacy, and shared support can be,” said Anish Bhatnagar, M.D., Soleno Therapeutics CEO. “Families and individuals living with PWS deserve understanding, access, and care that truly meets their needs.”
Sponsored By Soleno Therapeutics


Broadway star Aaron Lazar shares how an ALS diagnosis transformed his life, leading to a Grammy-nominated album and a new mission in research.
How has living with ALS shaped your life and career?
Before the diagnosis, I assumed ALS was a death sentence. Instead, despite suffering much loss, it’s been the opposite.
From the moment I was diagnosed, I listened to a quiet inner voice that told me healing was possible. I didn’t know how, but I followed that whisper. It sent me on a path to study the nervous system and healing through science and spiritual wisdom. Across disciplines and centuries, the message is the same: Health is not just physical — it’s mental, emotional, and spiritual.
ALS became a catalyst for the end of an ego-based identity I’d lived for decades and the beginning of discovering and embodying my true authenticity and autonomy. The paradox is that this new version of me feels more like myself than ever before. A stranger once wrote to me, “What if there’s nothing to heal? What if this is about remembering who you really are?” That landed. In many ways, that’s exactly what’s happened.
From the moment I was diagnosed, I listened to a quiet inner voice that told me healing was possible. I didn’t know how, but I followed that whisper.

This journey has deepened my relationships with my children and my partner in profound ways. It’s also reshaped my career, leading to the creation of my debut album “Impossible Dream,” which earned me a Grammy nomination, and moving me from primarily acting and singing into keynote speaking, podcast hosting, and leading a company focused on ALS research through AI and data science.
How did you adapt to mobility changes, and what has helped you maintain independence?
I’m still adapting. Some changes are easier than others, and adaptation is ongoing. What helps most is love and support — from family, caregivers, friends, and even strangers who’ve connected through my work.
My transition to a wheelchair happened gradually. I’m now on my third one. It’s not
easy sitting all day, but the chair also gives me independence. With the right modifications, it allows me to move through the world and get where I want to go.
What types of care or patient support services have been most helpful?
The ALS Network has been extraordinary. They provide real, hands-on support for patients and families, much of it free, and they’ve been champions of my work from the very beginning.
Having the right live-in caregiver is essential. Medical equipment providers matter, too. Sherman Oaks Medical and my clinical team have been attentive, compassionate, and instrumental in helping me get the equipment and care I need to live and work.
How has your experience shaped your
perspective on early diagnostics, rare disease research, and advocacy?
It took nine months for me to be diagnosed — faster than many, but still far too long. Early diagnostics are critical, and rare disease research urgently needs more funding.
I consider myself fortunate. I can still speak, sing, and write — abilities many people with ALS lose early. That reality drove me to co-found the Impossible Dream Machine, using AI and data science to accelerate ALS diagnostics, treatments, and cures.
From the beginning, my clarity has been simple: We need to flip the script on this disease. We need to offer hope. We need to remind people how powerful and resilient they really are.
Two years ago, the Oxford-Harrington Rare Disease Centre (OHC) made a commitment to advance 40 rare disease drugs into clinical trials by 2034. The partnership already has over 50 drugs in development — reflecting the urgency of unmet need and the strength of its model.
Central to this effort is the OHC Rare Disease Scholar Award program. Each year, ten academic investigators from the United States, United Kingdon, and Canada are selected to receive funding and translational support on projects ranging from neurodevelopmental, neuromuscular and metabolic disorders to rare cancers. Uniquely, Scholars also receive strategic guidance on all aspects of drug development, with the goal of accelerating projects towards clinical trials and de-risking them for further development and investment.
One of the newest Scholar projects focuses on CASK, a rare genetic disease that affects normal brain development, leading to severe physical and intellectual impairment, and other debilitating symptoms. For families, the impact is devastating.
When Cindy Schulz’s daughter Noelle (“Noni”) was a toddler, developmental delays became impossible to ignore. Despite multiple appointments, the cause of Noni’s condition could not be identified, and Cindy was told that there was no treatment plan beyond supportive care.
Noni faced significant intellectual and physical challenges and, although she learned to walk and talk, many everyday skills remained out of reach. So, Cindy focused on building a joyful, fulfilling life for her daughter. Now 42, Noni lives happily with her parents and has enjoyed working at a local grocery store for nearly 20 years.
Noni was 30 when genetic testing finally identified the cause: a rare mutation in the CASK gene. At the time, only around 50 individuals had been diagnosed. Today, CASK is estimated to affect about 2,000 individuals and that number is growing as awareness and testing improve.


WRITTEN BY Matthew Wood, M.D., Ph.D. Director & Chief Scientific Officer, Oxford-Harrington Rare Disease Centre
WRITTEN BY Matthew Anderson, M.D., Ph.D. Co-Director, Oxford-Harrington Rare Disease Centre; Investigator, Harrington Discovery Institute
For many families, the hardest part of a rare disease diagnosis is the absence of a clear treatment path while promising academic discoveries often stall before becoming therapies, because of a lack of commercial incentives.
The OHC was created to bridge that gap
A first-of-its-kind partnership between the University of Oxford and Harrington Discovery Institute at University Hospitals in Cleveland, the OHC connects world-class academic science with drug development and industry expertise to help transform promising discoveries into therapies and cures.
Enter Dr. Mingshan Xue, a neuroscientist at Baylor College of Medicine.
Dr. Xue has long studied the genetic pathways that shape brain development and his interest in CASK began when he met a patient during his postdoctoral training. Confronted by its severity and the lack of effective treatments, he committed to finding a therapy. Through connections with families and advocacy groups, such as Project CASK, he understood the urgency.
As an OHC Scholar, Dr. Xue’s research is focused on developing a gene therapy to replace the dysfunctional gene with a

functional copy, thereby addressing the root cause of the disease rather than just managing symptoms.
“What attracted me to the OHC was access to deep drug development expertise,” Dr. Xue explains. “Gene therapy requires coordinated scientific, regulatory, and manufacturing planning. That integrated support is essential to move this toward patients.”
If successful, a disease-modifying therapy for CASK could significantly improve the development, health, and quality of life of affected individuals.
“When Noni was young, effective therapy wasn’t even part of the conversation,” Cindy reflected. “Now, researchers are working to change the course of CASK, raising hopes for a cure that families have been praying for. It’s extraordinary.”
Progress in finding new treatments and cures for CASK and for the many other rare diseases that collectively affect nearly 500 million people worldwide will take time, and not every program will succeed. But the convergence of determined families, focused science, and translational partnerships like the OHC is reshaping what is possible.




To learn more, visit oxford harrington.org
Advances in genomic sequencing offer the potential to significantly increase the number of genetic conditions that can be identified at birth and treated.
Each year, newborn screening (NBS) saves thousands of lives by identifying babies who appear healthy at birth but have serious medical conditions requiring early intervention. Without timely treatment, these conditions can lead to irreversible harm or even death. Today, nearly every baby born in the United States undergoes NBS within the first few days of life, regardless of birthplace, geography, or socioeconomic factors. The screening consists of a hearing test, a pulse-oximetry test to check for congenital heart conditions, and a heel prick to check for a panel of genetic conditions. While each state runs its own NBS program, these efforts are supported by federal resources, including funding, guidance, and quality standards.
Despite its decades-long success, the current NBS includes fewer than 40 genetic conditions. However, advances in genomic sequencing, a powerful technology that examines the majority of an individual’s DNA, offer the potential to significantly increase the number of conditions that can be identified at birth and treated. Clinical studies have shown the benefits of incorporating genomic sequencing into NBS, also referred to as genomic NBS (gNBS). These studies have evaluated 200-500 actionable conditions, far beyond the current standards.
A new era begins
In 2025, Florida became the first state to offer gNBS for all newborns. Families who choose to participate can have their baby’s genome sequenced at no cost, enabling screening for approximately 500 genetic conditions. Federally, the National Institutes of Health Common Fund Venture Program has funded the first national initiative to assess the
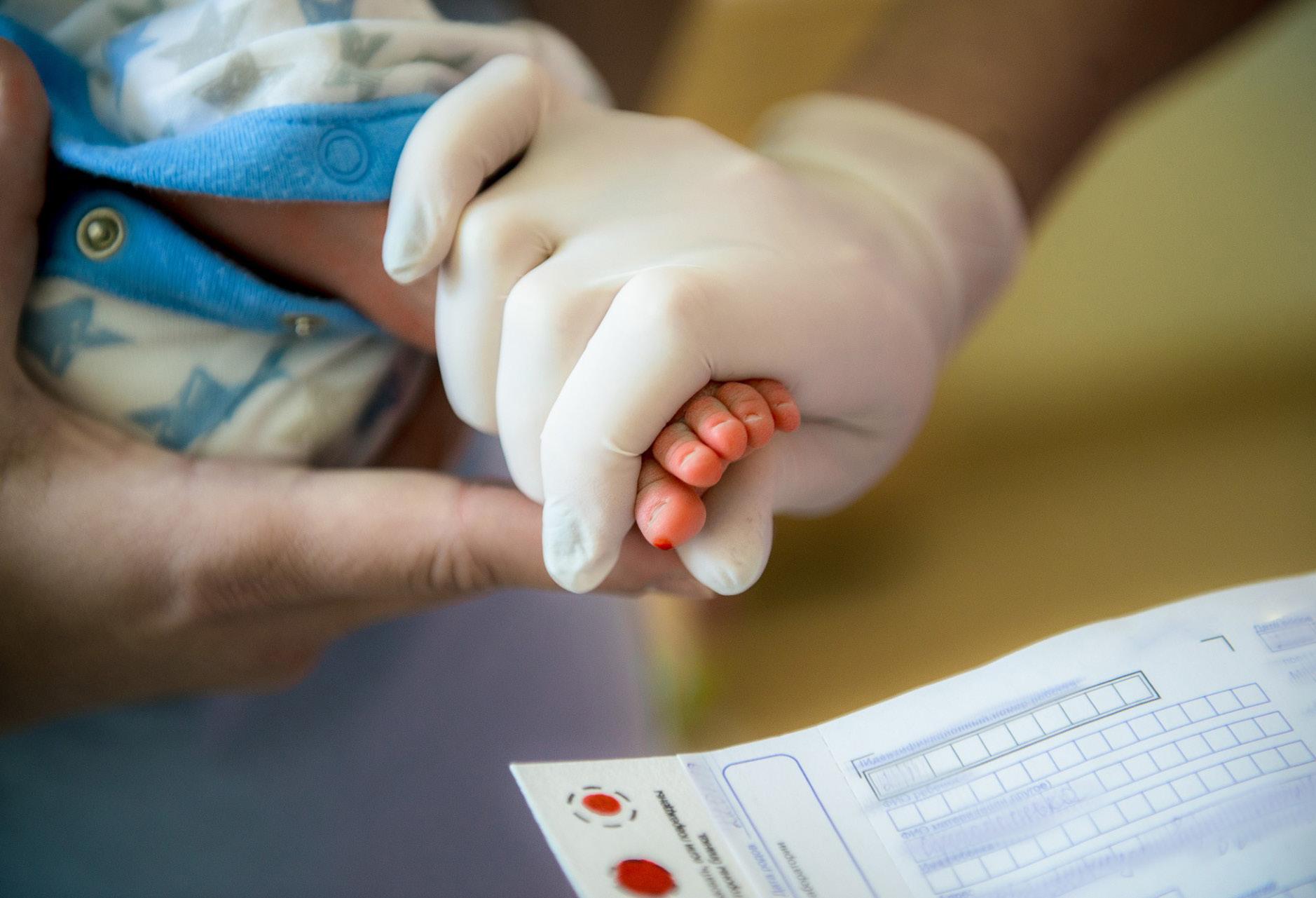
feasibility of incorporating gNBS into all public health programs throughout the United States. The study, referred to as Building Evidence and Collaboration for GenOmics in Nationwide Newborn Screening (BEACONS), will also examine public perceptions and ethical considerations surrounding gNBS.
While sequencing at birth will allow earlier diagnosis and intervention for numerous conditions, its potential reaches far beyond infancy. Because a person’s DNA does not change, genomic information can support health decisions throughout life, guiding surveillance, preventive care, and treatment as new therapies emerge. Together with advances in targeted genetic therapies, gNBS represents a transformative step toward a future where healthcare is proactive, personalized, and available to all.
However, integrating genomic sequencing into NBS also presents challenges. These include managing large amounts of data, ensuring storage in medical records, addressing privacy concerns,
and preventing genetic discrimination. Policymakers, clinicians, and ethicists must also determine what types of results should be shared and at what age.
Another critical challenge is workforce capacity. There is a significant shortage of physicians who are formally trained in genetics. Expanding screening without ensuring accessible clinical expertise risks overwhelming an already strained system.
As we stand on the brink of this new era in public health, genomic sequencing offers an unprecedented opportunity to redefine how we approach lifelong health. By expanding the conditions detected at birth and empowering families with actionable information, gNBS can create a foundation for truly personalized care. Continued collaboration among states, federal agencies, clinicians, researchers, patient advocates, and the general public will be essential to ensure that gNBS is implemented ethically and effectively. The future of NBS is here, and it promises a healthier tomorrow for generations to come.


Investigational transplacental “bridging” therapy, followed by rapid postnatal treatment, aims to protect a baby’s motor neurons during a critical window before and immediately after birth.
When Stacy Auker found out she was pregnant, she knew a diagnosis of spinal muscular atrophy (SMA) was a possibility. “We have relatives who have been affected, so I got tested during pregnancy with each of my kids,” Auker explained.
This time, her baby did test positive for SMA type 1, a severe neurodegenerative genetic condition that typically results in death in early childhood. Despite the news, Auker and her husband, Clyde, remained optimistic, knowing that treatment options were now available.
Stacy’s physician referred her to maternal-fetal medicine specialists at Nationwide Children’s Hospital, the same place where researchers led the development of Zolgensma®, a one-time gene therapy approved by the U.S. Food and Drug Administration (FDA) in 2019 to treat SMA type 1.
The timing was nearly perfect when the specialists met with the Aukers. Adolfo Etchegaray, M.D., chief of Fetal Medicine at Nationwide Children’s, was aware of a new approach for prenatal treatment of SMA recently published in The New England Journal of Medicine. The investigational therapy involved risdiplam, a medication for the production of SMN protein, given orally to mothers during the third trimester. He approached the Aukers with this experimental, but exciting, new treatment option.
Upon Stacy’s consent, team members pursued investigational approval from the FDA for use
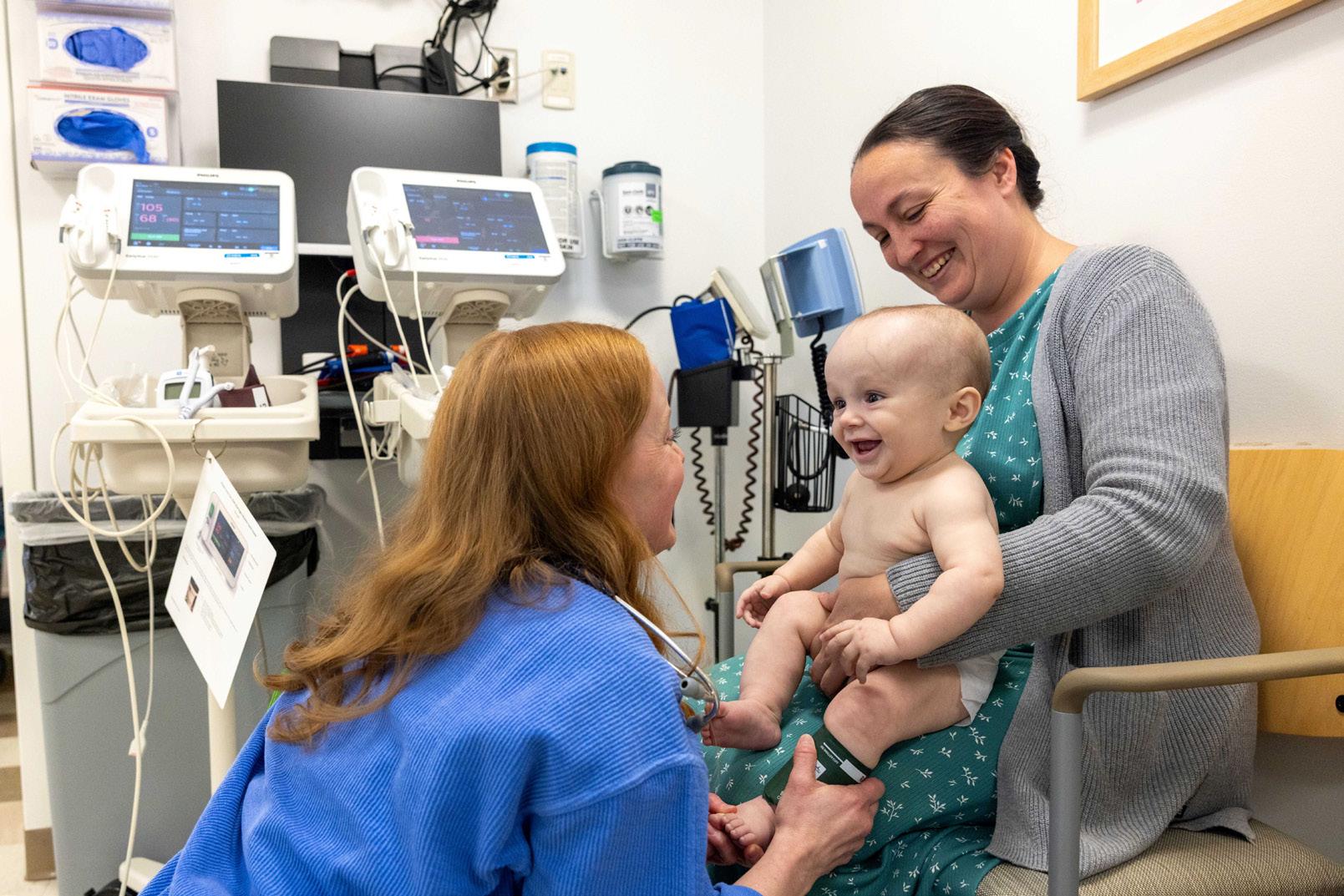
of risdiplam before birth. The Genentech Patient Foundation provided the drug free of charge, and Stacy started taking risdiplam at Week 34 of her pregnancy.
At Week 38, their son Bryson was born, apparently healthy and with normal muscle tone, reflexes, and motor function. He was given his first postnatal dose of risdiplam the day after delivery. He then received gene therapy at 10 days old, making him one of the youngest patients ever to be given Zolgensma.
“Cases like these are changing the paradigm of managing some monogenic conditions,” Dr. Etchegaray said. Nationwide Children’s offered the Auker Family the full continuum of care from its Maternal-Fetal Medicine Center to its Gene Therapy Center of Excellence to its SMA Clinic.
Dr. Etchegaray and his
colleagues presented their findings at the 2025 meeting of the Society for Maternal-Fetal Medicine and plan to publish updated findings in a case series with a handful of other institutions that have initiated risdiplam therapy prenatally.
“This is an example of how fetal medicine is personalized, preventative, and participative,” Dr. Etchegaray noted.
The Aukers continue to provide samples for research with the Nationwide Children’s team and follow up with specialists in the SMA Clinic. Bryson continues with daily risdiplam and, as of the age of 8 months, shows no signs of SMA.
“His siblings adore him — they love to make him laugh. We are hopeful Bryson can run and play with them in the future, Lord willing,” said Stacy, who understands
this outcome would have been impossible prior to the development of these treatments.
Well known for studying and treating rare diseases, Nationwide Children’s researchers have developed three FDA-approved gene therapies: Zolgensma® and Itvisma® for SMA, and Elevidys®, a gene therapy for Duchenne muscular dystrophy approved in 2023.
Written by Katie Brind’Amour, Ph.D., M.S., CHES, Science Writer, Nationwide Children’s Hospital
Discover more about Nationwide Children’s rare disease research at nationwidechildrens.org/ research

Ten years ago, babies born with spinal muscular atrophy (SMA) rarely lived past their second birthday. Today, thanks to breakthrough treatments, some of those same children are walking, attending school, and imagining their futures.
Once the leading genetic cause of infant death, SMA is no longer a death sentence, but living longer has introduced new challenges that must now be addressed.
SMA affects about 1 in 15,000 babies born in the United States and touches families in all 50 states. The rare neuromuscular disease weakens muscles used for breathing, eating, crawling, and walking. Just a decade ago, children born with the most severe form of SMA often did not survive early childhood. Today, access to treatments has dramatically changed that outlook, with the SMA mortality rate dropping by roughly 80%.
Research shows that timing
matters. Babies with the most severe form of SMA who receive treatment within the first two weeks of life have extraordinary outcomes. Studies supported by Cure SMA show that 100% of these infants were able to walk independently by age 2.
Medical progress alone is not enough. Many people with SMA face outdated policies and systems that limit independence and financial security. Nearly 1 in 5 SMA households live on less than $20,000 a year, and some adults report food insecurity, unstable housing, or skipped medications and medical appointments due to cost.
Technology and innovation are helping bridge some of these gaps. Robotic arms that assist





with eating, eye-gaze systems that enable communication, and other assistive tools already make daily tasks easier. Looking ahead, accessible autonomous vehicles and advanced robotics could dramatically improve mobility and independence if people with disabilities are included from the earliest stages of design.




Scientific breakthroughs have transformed what it means to live with SMA. With continued research, smarter public policy, and inclusive innovation, the future holds even greater promise, one where people with SMA not only survive but thrive.
Written by Cure SMA








As a parent, I’ve seen firsthand that scientific breakthroughs alone aren’t enough. Bold, strategic investment is what turns hope into real progress.
When my son was diagnosed with Duchenne muscular dystrophy, I asked the question many families ask: Why isn’t there a treatment? And if researchers are working on one, why is it taking so long?
For anyone affected by a rare disease, the hardest part is waiting for promising research to advance, clinical trials to begin, and new drugs to be approved. Too often, we’re waiting for early-stage funding to begin this process.
Bringing a therapy to market is expensive. A 2020 JAMA study found the median cost of developing a new drug was $1.1 billion, with many programs exceeding that due to failures, regulatory hurdles, and long clinical timelines. For rare diseases like Duchenne, which often require the design of novel clinical endpoints, new delivery methods, and complex manufacturing processes, the costs are higher.
But that’s not the whole story. These billions don’t show up all at once. Every breakthrough starts with an early-stage discovery that needs capital to move out of the lab and into development. This is where many stall — not because the science isn’t strong, but because there’s no funding to push it forward.
The early-stage funding gap
This early-stage gap is one of the biggest obstacles in advancing rare disease research. Many traditional venture capital firms wait until a program is far along and safer to invest in. Pharmaceutical companies often
prioritize diseases with larger patient populations. Government research grants don’t cover the full cost of development.
At CureDuchenne, we saw this gap as an opportunity. It’s why we created a venture philanthropy model to fund promising early-stage science and help advance it far enough to attract larger, later-stage investment. To date, we’ve funded 19 research projects that advanced to clinical trials, some now FDA-approved. Because today’s treatments don’t treat all Duchenne patients, our support continues to fuel the pipeline. Several research projects we funded years ago are showing promising clinical results and are expected to be submitted for FDA approval in the coming year.
These outcomes show what is possible when promising science is supported. They also show how much more is needed for Duchenne and the thousands of other rare diseases still overlooked.
Rare diseases affect 1 in 10 people, yet 95% still lack an FDA-approved treatment. That’s not due to a lack of science; it’s because we lack funding to back it. Families deserve bold, targeted investments — not someday, but now. Good science without funding is just potential, and without investment, that potential may never reach the people who need it most.


DMD steals the health of thousands of young boys. Atossa Therapeutics is researching a new approach to treatment that may offer hope.
Families dealing with Duchenne Muscular Dystrophy (DMD) experience the heartbreak of watching a child suffer from a debilitating, incurable disease.
A different approach
DMD occurs due to a genetic mutation primarily affecting boys, inhibiting production of a protein called dystrophin, which maintains and repairs muscle cells. This results in a steady decline in muscle function and shortened life expectancy. Atossa Therapeutics is evaluating whether impacting a protein called utrophin, which is functionally similar to dystrophin, may replace the shortened or missing dystrophin and could result in impactful treatment for DMD. This approach may also benefit female carriers of the DMD mutation, who often experience skeletal muscle loss later in life.
Early research leading to future clinical trials is encouraging, offering something families desperately need: Hope.
Written by Jeff Somers
Debra Miller Founder and CEO, CureDuchenne

Our panel of experts discusses gaps in patient care and how new technology is making the development of rare disease treatments more accessible.

Where do you see the biggest gaps in the rare disease patient journey today?
Charlene Son Rigby: Rare disease patients face critical challenges, starting with delayed diagnosis. Patients often endure years of misdiagnoses and diagnostic odysseys before receiving accurate answers, leading to prolonged suffering and potentially irreversible disease progression. Once diagnosed, patients struggle with a lack of recognition and support from healthcare systems not designed for rare conditions, resulting in fragmented care, social isolation, and difficulty accessing specialized expertise or connecting with other patients facing the same challenges.
PANELIST Tim Guilliams Co-Founder and CEO, Healx Ltd

Finally, limited research investment and small patient populations make developing new therapies challenging, leaving many rare diseases without effective treatments and forcing patients to navigate an underfunded landscape where therapeutic options remain desperately scarce.
What approaches are most promising for addressing unmet needs in rare diseases?
Tim Guilliams: Target-agnostic, AI-enabled discovery approaches that focus on novel biology and first-in-class mechanisms are particularly promising in rare diseases, where validated targets often don’t exist. Just as important is
Charlene Son Rigby CEO, Global Genes
deep collaboration with patient foundations, which bring essential disease insight, data, and urgency to the development process.
What is the biggest opportunity to improve patient outcomes in rare disease research?
TG: The biggest opportunity is aligning advanced AI with patient-relevant biology early in development to increase the likelihood of clinical success. When discovery, translational science, and patient insight are integrated from the start, we can accelerate timelines and deliver treatments that are safer, more effective, and meaningful for patients.
With 70-90% of primary immunodeficiency cases undiagnosed, and diagnostic journeys averaging 9-15 years, early detection is critical to prevent permanent damage and improve outcomes.
Primary immunodeficiencies (PI) are a group of more than 550 rare, chronic conditions where a part of the body’s immune system is missing or does not function correctly. PI conditions are caused by genetic variants and can affect anyone, regardless of age, gender, or ethnicity. Some disorders cause symptoms in infancy or early childhood, while others may not present until mid- or even late adulthood. This creates a dangerous combination: a disease that is invisible, rare, and chronic.
The benefits of early detection Without proper treatment, PI can
lead to permanent organ damage throughout the body. Infections can occur in any organ system, and patients may experience a range of complications.
Patients with undiagnosed PI are also at increased risk for developing additional immune-related conditions and certain types of cancer. Beyond physical health, delayed diagnosis takes a substantial toll on quality of life, with patients missing work or school, struggling with chronic fatigue, and facing daily limitations.
When PI is identified early, treatment options can be highly effective. Immunoglobulin replacement therapy,
prophylactic antibiotics, and other targeted interventions can dramatically reduce infection frequency and severity. Early diagnosis also provides families with crucial information for genetic counseling and helps identify other at-risk family members. If you routinely experience multiple infections each year or have been referred to multiple specialists without receiving a clear diagnosis, ask your doctor about your immune system. The path to diagnosis may require persistence, but identifying and treating PI promptly can prevent irreversible damage, improve quality of life, and, in some cases, save lives.

Across the United States, 1 in 10 individuals are affected by rare diseases, yet our healthcare system still isn’t built to serve rare disease patients.
Every family living with a rare disease has a story, and nearly all begin the same way: with questions that take years to answer.
Across the United States, 1 in 10 individuals is affected by a rare disease. This is 30 million Americans — a larger number than those affected by cancer. Yet, most Americans have never heard this statistic, and our healthcare system still isn’t built to serve rare disease patients.
The road to diagnosis averages six years — six years of unanswered tests, mounting medical bills, misdiagnoses, and profound isolation. When an answer finally comes, families face another sobering reality: 95% of rare diseases have no approved treatment. Families are left to navigate a fragmented system, often paying out-of-pocket for specialists, genetic testing, or experimental care.
Patients as partners
These are not just medical challenges; they are human ones. Patients become advocates, testifying before regulators and fundraising for clinical trials. Parents become researchers, poring over scientific articles late into the night. Communities form from necessity and love, not luxury. Behind every discovery in rare disease is a family that refused to give up, and behind many breakthroughs in modern medicine are lessons first learned through rare disease science. At Global Genes, we’ve seen this transformation firsthand through our work over the past two decades. When patients and caregivers are engaged early in the research process and remain active partners throughout, diagnostic

journeys shorten, research becomes more inclusive, and treatments reach people sooner.
Building a better system
Today’s rare disease patients and patient organizations are jumping in to fund research, run natural history studies, hire scientists, and, in some cases, launch biotech companies. For families on this journey, success takes scientific knowledge, funding, credibility, and courage through an extremely challenging process. Yet, families are proving that even the rarest diseases can become treatable.
Behind many breakthroughs in modern medicine are lessons first learned in rare disease science. Gene therapy, personalized medicine, patient-driven research — these innovations emerged from the rare disease community’s refusal to accept “nothing can be done.”
These families aren’t just fighting for their own children. They’re building a healthcare system that works for everyone — one that listens to patients, values lived experience, and refuses to leave anyone behind.
Rare diseases affect more than 400 million people globally, yet the vast majority still lack approved treatments. AI may be able to improve outcomes for these rare patients.
One of the biggest challenges facing rare disease patients is that many conditions are poorly understood at a biological level, making traditional drug discovery slow and uncertain.
Artificial intelligence is beginning to change that. By analyzing complex biological, clinical, and patient-derived data, AI can uncover disease mechanisms and therapeutic opportunities that would be difficult to identify using conventional approaches alone. This systems-level understanding is particularly powerful in rare diseases, where validated drug targets are often missing.
At Healx, an AI-enabled rare disease biotech, this approach is already being translated into the clinic. By combining AI-driven discovery with deep partnerships across patient foundations and clinical experts, Healx is advancing first-in-class therapies designed around the needs of patients — not just the disease.
For the rare disease community, the opportunity is clear: When advanced technology, human expertise, and patient insight come together, scientific breakthroughs can move faster and deliver meaningful improvements in patient outcomes.


