Ivermectin & Cancer Research with proposed Hybrid Orthomolecular Protocol
Journal of ANTIBIOTICS: https://www.nature.com/articles/ja201711
Title: “Ivermectin: enigmatic multifaceted “wonder” drug continues to surprise and exceed expectations”
Published: Feb 15, 2017
Abstract
Over the past decade, the global scientific community have begun to recognize the unmatched value of an extraordinary drug, ivermectin, that originates from a single microbe unearthed from soil in Japan. Work on ivermectin has seen its discoverer, Satoshi Ōmura, of Tokyo’s prestigious Kitasato Institute, receive the 2014 Gairdner Global Health Award and the 2015 Nobel Prize in Physiology or Medicine, which he shared with a collaborating partner in the discovery and development of the drug, William Campbell of Merck & Co. Incorporated. Today, ivermectin is continuing to surprise and excite scientists, offering more and more promise to help improve global public health by treating a diverse range of diseases, with its unexpected potential as an antibacterial, antiviral and anti-cancer agent being particularly extraordinary.
Quote in the article: “Since the prodigious drug donation operation began, 1.5 billion treatments have been approved.”
Disease-fighting potential identified to date (Feb 15, 2017) listed in the article:
Myiasis
Trichinosis
Disease vector control – toxic to 84 species of insections
Malaria
Leishmaniasis
African trypanosomiasis (sleeping sickness)
American trypanosomiasis (Chagas disease)
Schistosomiasis
Bedbugs
Rosacea
Asthma
Epilepsy
Neurological disease
Antiviral (e.g. HIV, dengue, encephalitis)
Antibacterial (tuberculosis and Buruli ulcer)
Anti-cancer
“shown to exhibit both anti-cancer and anti-cancer stem cell properties”
In human ovarian cancer and NF2 tumor cell lines, high-dose ivermectin inactivates protein kinase PAK1 and blocks PAK1dependent growth. ..
PAK1 is essential for the growth of more than 70% of all human cancers, including breast, prostate, pancreatic, colon , gastric, lung, cervical and thyroid cancers, as well as hepatoma, glioma, melanoma, multiple myeloma and for neurofibromatosis tumors.”
Ivermectin suppresses breast cancer by activating cytostatic autophagy, disrupting cellular signaling in the process, probably by reducing PAK1 expression…
It has been reported that ivermectin induces chloride-dependent membrane hyperpolarization and cell death in leukemia cells and it has also been suggested that ivermectin synergizes with the chemotherapy agents cytarabine and daunorubicin to induce cell death in leukemia cells, with researchers claiming that ivermectin could be rapidly advanced into clinical trials.
Cancer stem cells are a key factor in cancer cells developing resistance to chemotherapies and these results indicate that a combination of chemotherapy agents plus ivermectin could potentially target and kill cancer stem cells, a paramount goal in overcoming cancer.
Ivermectin inhibits proliferation and increases apoptosis of various human cancers.
Again, given that ivermectin is already approved for use in humans, its rapid development as an anti-mitotic agent offers significant promise.
Journal of ORTHOMOLECULAR MEDICINE: https://isom.ca/article/targeting-the-mitochondrial-stem-cellconnection-in-cancer-treatment-a-hybrid-orthomolecular-protocol/
Title: Targeting the Mitochondrial-Stem Cell Connection in Cancer Treatment: A Hybrid Orthomolecular Protocol
Published: Sept 19, 2024
RFK Jr Telegram Oct 16, 2024:
The first peer-reviewed protocol using Ivermectin and Fenbendazole for cancer treatment has finally been peer-reviewed and published.
This is huge. Dr. Makis says he is already seeing “incredible successes with these repurposed drugs.”
Here’s the quick summary of the study’s findings:
• Ivermectin targets cancer cells by disrupting their mitochondrial function, leading to cell death (apoptosis). It also blocks key energy pathways, showing significant tumor shrinkage, especially in pancreatic cancer.
• Fenbendazole and Mebendazole, both antiparasitic drugs, have been found to halt cancer cell growth, interfere with glucose metabolism, and trigger apoptosis. In some preclinical trials, these drugs have even outperformed standard chemotherapy in reducing tumor growth.
• Remarkably, three patients with stage IV genitourinary cancers were treated with high-dose Fenbendazole (1,000 mg, three times a week for several months) and experienced complete remission.
This is remarkable and especially hopeful for patients who have tried everything else with no luck. The next step will be further clinical trials to validate the protocol's effectiveness among larger populations.
Article Abstract
The mitochondrial-stem cell connection (MSCC) theory suggests that cancer originates from chronic oxidative phosphorylation (OxPhos) insufficiency in stem cells. This OxPhos insufficiency leads to the formation of cancer stem cells (CSCs) and abnormal energy metabolism, ultimately resulting in malignancy. This concept integrates two well-established theories: the cancer stem cell theory and the metabolic theory. Drawing on insights from molecular biology, pharmacology, and clinical studies, this manuscript introduces a hybrid orthomolecular protocol targeting the MSCC. The protocol includes 7 therapeutic recommendations, consisting of ortho molecules, drugs, and additional therapies. The aim of this hybrid orthomolecular protocol is to achieve additive and synergistic effects to enhance OxPhos, inhibit the primary fuels of cancer cells (glucose and glutamine), target CSCs and metastasis. Thus, numerous experiments suggest that targeting MSCC could be a potential therapeutic approach for cancer treatment.
Introduction paragraph
…Thus, after reviewing the literature on various therapies capable of targeting the MSCC, we selected, based on in vitro and in vivo studies, several ortho molecules, drugs, and additional therapies that have demonstrated an ability to enhance OxPhos, reduce fermentable fuels, and target CSCs and metastasis. Furthermore, when supported by scientific literature, we included case studies of cures using monotherapy in humans. From this combination, we developed a hybrid orthomolecular protocol, which is proposed as a new therapeutic strategy for cancer.
Key Points – please read
Orthomolecular Medicine for Targeting the MSCC
Vitamin C
Vitamin D
Zinc
Potential Drugs for Targeting the MSCC
Several pharmaceutical agents can primarily target genetic pathways associated with CSCs, including Vismodegib, Glasdegib, MK0752, OMP-54F28, and Selinexor (Zhou, et al., 2021). Other pharmaceutical agents have been proposed to target mitochondria, such as Metformin for OxPhos (Ward, et al., 2017; Zheng, et al., 2023) Doxycycline, Tigecycline, and Bedaquiline for mitochondrial biogenesis; Mdivi-1 drug in mitochondrial dynamics; and 188Re-liposome and the inhibitor liensinine to block mitophagy (Jagust, et al., 2019; Praharaj, et al., 2022). Most of the time, these agents do not restore mitochondrial homeostasis (Liu, Y., et al., 2023), as their specific actions alter or only partly restore dysfunction. The alteration of mitochondrial function with pharmaceutical agents must be considered with caution, as it can be very dangerous for healthy cells (Vuda & Kamath, 2016).
Repurposed (off-label) Drugs for Targeting the MSCC
Ivermectin
Benzimidazoles
DON (6-diazo-5-oxo-L-norleucine
Dietary Interventions for Targeting the MSCC
Fasting
Ketogenic Diet and Ketone Metabolic Therapy (KMT)
Additional Therapeutic Considerations
Press—Pulse Therapy
Physical Activity
Hyperbaric Oxygen Therapy (HBOT)
Proposed Hybrid Orthomolecular Protocol
Based on our review of the scientific literature, the following protocol combining ortho molecules, drugs and additional therapies for targeting the MSCC in cancer treatment is proposed:
1. Intravenous Vitamin C
Intermediate- and high-grade cancers: Dose of 1.5g/kg/day, 2-3x per week (Fan, et al., 2023).
Established as a non-toxic dose for cancer patients (Wang, F., et al., 2019).
.
2. Oral Vitamin D
All cancer grades: Dose of 50,000 IU/day for patients with a blood level ≤ 30ng/mL; 25,000 IU/day for levels 30-60ng/mL; and 5000 IU/day for levels 60-80ng/mL.
Established as a non-toxic dose (Cannon, et al., 2016; Ghanaati, et al., 2020; McCullough, et al., 2019).
It is necessary to reach a blood level of 80 ng/mL of vitamin D (25-hydroxyvitamine D (25(OH) D) (Kennel, et al., 2010; Mohr, et al., 2014; Mohr, et al., 2015). This level is non-toxic (Holick, et al., 2011). Once this level is reached it must be maintained with a reduced daily dosage of ≈ 2000 IU/day (Ekwaru, et al., 2014). The vitamin D blood concentration should be measured every two weeks for high doses and monthly for lower doses.
3. Zinc
All cancer grades: Dose of 1 mg/kg/day is established as a non-toxic dose for cancer patients (Hoppe, et al., 2021; Lin, et al., 2006). The reference range for serum zinc concentration is 80 to 120 μg/dL (Mashhadi, et al., 2016; Yokokawa, et al., 2020). Once this level is reached it must be maintained with a reduced daily dosage of 5mg/day (Li, et al., 2022). The zinc blood concentration should be measured monthly.
4. Ivermectin
Low-grade cancers: Dose of 0.5mg/kg, 3x per week (Guzzo, et al., 2002).
Intermediate-grade cancers: Dose of 1mg/kg, 3x per week (Guzzo, et al., 2002).
High-grade cancers: Dose from 1 mg/kg/day (de Castro, et al., 2020) to 2 mg/kg/day (Guzzo, et al., 2002).
All these doses have been established as tolerable for humans (Guzzo, et al., 2002).
5. Benzimidazoles and DON
Low-grade cancers: Mebendazole: Dose of 200 mg/day (Dobrosotskaya, et al., 2011).
Intermediate-grade cancers: Mebendazole: Dose of 400 mg/day (Chai, et al., 2021)
High-grade cancers: Mebendazole dose of 1,500 mg/day (Son, et al., 2020) or Fenbendazole 1,000 mg 3x per week (Chiang, et al., 2021).
All these doses have been established as tolerable for humans (Chai, et al., 2021; Chiang, et al., 2021; Son, et al., 2020).
Benzimidazoles can be replaced or combined with DON, administered without toxicity; intravenously or intramuscularly: 0.2 to 0.6 mg/kg once daily; or orally: 0.2 to 1.1 mg/kg once daily (Lemberg, et al., 2018; Rais, et al., 2022). Benzimidazoles are much easier to obtain than DON. However, for metastatic cancers, which rely heavily on glutamine (Seyfried, et al., 2020), a combination of DON and Benzimidazoles should be considered (Mukherjee, et al., 2023).
6. Dietary Interventions
All cancer grades: Ketogenic diet (low carbohydrate-high fat diet, 900 to 1500 kcal/day) (Weber, et al., 2020).
Ketone metabolic therapy consists of approximately 60-80% fat, 15-25% protein and 5-10% fibrous carbohydrates.
Adequate hydration and single-ingredient whole food ketogenic meals are necessary to achieve a glucose ketone index (GKI) score of 2.0 or below (Meidenbauer, et al., 2015; Seyfried, Shivane, et al., 2021). GKI should be measured 2–3 hours postprandial, twice a day if possible (Meidenbauer, et al., 2015; Seyfried, Shivane, et al., 2021).
Intermediate- and high-grade cancers: The ketogenic diet should be coupled with a water fast for 3 to 7 consecutive days in advanced cancers (Phillips, et al., 2022; Arora, et al., 2023). The water fast should be repeated several times (≈ every 3-4 weeks) throughout the treatment (Nencioni, et al., 2018), but fasting needs to be undertaken cautiously in individuals using certain drugs and those with < 20 BMI, to prevent loss of lean body mass. For patients who can not fast, the Fasting-
Mimicking Diet (300 to 1,100 kcal/day of broths, soups, juices, nut bars, and herbal teas) can be used (Nencioni, et al., 2018).
.
7. Additional Therapeutics
All cancer grades: Moderate physical activity, 3x per week. Increased heart and respiratory rate for a period of 45 to 75 minutes (Bull, et al., 2020) with activities such as cycling, running, swimming, etc.
Intermediate- and high-grade cancers or individuals who are unable to engage in physical activity: Hyperbaric oxygen therapy, 1.5 to 2.5 ATA for 45 to 60 minutes 2-3x per week (Gonzalez, et al., 2018; Poff, et al., 2015).
The protocol should be followed for an average duration of 12 weeks, regardless of cancer type. The analysis of the interactions between each of the molecules revealed no contraindications to the combination of these substances (ANSM, 2023; CRAT, 2024; Lemberg, et al., 2018; Vidal, 2024). The treatment dosage and duration can be adjusted by the physician according to the individual patient, their ability to obtain the various molecules, and the treatment results. Adaptation of the protocol to include additional molecules to restore health, could be considered by the physician. These may include: vitamin K2 (Xv, et al., 2018), vitamin E (Abraham, et al., 2019), coenzyme Q10 (Liaghat, et al., 2024), methylene blue (da Veiga Moreira, et al., 2024), niacinamide (Yousef, et al., 2022), riboflavin (Suwannasom, et al., 2020), Artemisinin + 5-aminolevulinic acid (to cause porphyrin accumulation) (Adapa, et al., 2024), melatonin (Mocayar, et al., 2020), NADH (Medjdoub, et al., 2016), and magnesium (Ashique, et al., 2023), as examples. However, antioxidant dosages should be avoided.
This additive and synergistic effect of this combination of ortho molecules, drugs, and additional therapies targets the MSCC by increasing OxPhos activity in healthy mitochondria, offering protective action for these cells. However, in cancer cells, both CSCs and non-CSCs, the pro-oxidant effect of the combination induces apoptosis. Additionally, this protocol specifically targets fermentable fuels, CSCs and macrophages, and thus metastases. In brief, the key points of the MSCC. Therefore, comparative studies need to be conducted in both animals and humans to evaluate the effectiveness and safety of this hybrid protocol against standard therapies.
Conclusion
National LIBRARY OF MEDICINE: https://pmc.ncbi.nlm.nih.gov/articles/PMC7505114/
Title: “Ivermectin, a potential anticancer drug derived from an antiparasitic drug”
Published: Sep 21, 2020
Abstract:
Ivermectin has powerful antitumor effects, including the inhibition of proliferation, metastasis, and angiogenic activity, in a variety of cancer cells. This may be related to the regulation of multiple signaling pathways by ivermectin through PAK1 kinase. On the other hand, ivermectin promotes programmed cancer cell death, including apoptosis, autophagy and pyroptosis. Ivermectin induces apoptosis and autophagy is mutually regulated. Interestingly, ivermectin can also inhibit tumor stem cells and reverse multidrug resistance and exerts the optimal effect when used in combination with other chemotherapy drugs.
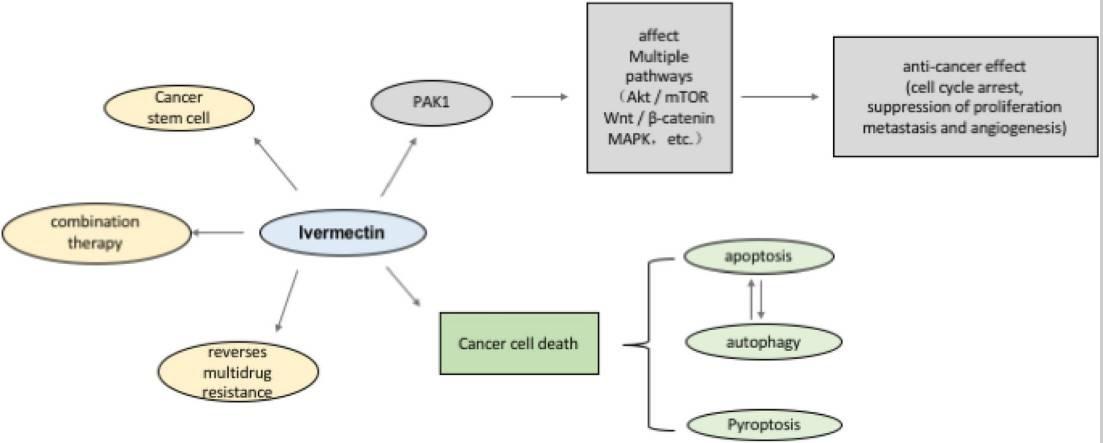
1. Introduction
2. The role of IVERMECTIN in different cancers
2.1 Breast Cancer
2.2 Digestive system cancer
2.3 Urinary system cancer
2.4 Hematological cancer
2.5 Reproductive system cancer
2.6 Brain glioma
2.7 Respiratory system cancer
2.8 Melanoma
3. IVERMECTIN-induced programmed cell death in tumor cells and related mechanisms
3.1 Apoptosis
3.2 Autophagy
3.3 Cross talk between IVM-induced apoptosis and autophagy
3.4 Pyroptosis
4. Anticancer effect of IVM through other pathways
4.1 Cancer stem cells
4.2 Reversal of tumor multidrug resistance
4.3 Enhanced targeted therapy and combined treatment
5. Molecular targets and signaling pathways involved in the anticancer potential of IVM
6. Summary and outlooks
One paragraph: As mentioned above, the broad-spectrum antiparasitic drug IVM, which is widely used in the field of parasitic control, has many advantages that suggest that it is worth developing as a potential new anticancer drug. IVM selectively inhibits the proliferation of tumors at a dose that is not toxic to normal cells and can reverse the MDR of tumors. Importantly, IVM is an established drug used for the treatment of parasitic diseases such as river blindness and elephantiasis. It has been widely used in humans for many years, and its various pharmacological properties, including long- and short-term toxicological effects and drug metabolism characteristics are very clear. In healthy volunteers, the dose was increased to 2 mg/Kg, and no serious adverse reactions were found, while tests in animals such as mice, rats, and rabbits found that the median lethal dose (LD50) of IVM was 1050 mg/Kg [112] In addition, IVM has also been proven to show good permeability in tumor tissues [50]. Unfortunately, there have been no reports of clinical trials of IVM as an anticancer drug. There are still some problems that need to be studied and resolved before IVM is used in the clinic.
