Annual Report 2025







The purpose of the Lundbeck Foundation is to create powerful ripple effects that bring discoveries to lives through investing actively in business and science at the frontiers of their fields.
Grants and Prizes
825DKKm grants awarded in 2025 Invest
We live our purpose every day through engaged ownership of our strategic ownerships and biotech portfolio companies, all of which strive to improve patients’ lives. Our investment activities enable us to continuously award significant independent research grants to advance Danishbased biomedical research with a specific focus on neuroscience. Established in 1954, the Lundbeck Foundation is one of Denmark’s largest enterprise foundations, with a net wealth at the end of 2025 of DKK 81bn.
2,905DKKm generated return in 2025 primarily driven by listed equities. Total market value at 31 December 2025 amounted to DKK 26,321m
In 2025, the Lundbeck Foundation maintained its strong support for outstanding biomedical research, with continued emphasis on talent development and both basic and clinical brain research.
In 2025, research supported by the Lundbeck Foundation resulted in 687 scientific publications. The examples below are based on articles published in Nature.

Parkinson’s disease can spread by three routes Researchers from Aarhus University have shown that Parkinson’s disease can start in the brain or in the body and enter the brain via two different routes

Special brain cells ensure safe behaviour in mice. Researchers from the University of Copenhagen have uncovered a previously unknown network of nerve cells that prioritise survival over both food and mating in mice. This might one day benefit people with movement disorders

The age of diagnosis reflects different course of autism. The genetic profile differs between patients diagnosed early with autism versus late, suggesting different underlying pathophysiology manifested in at least two different developmental trajectories. These insights come from researchers at Aarhus University

… and new strategic initiatives.


The Danish Sleep Center for Children and Adolescents has been established to advance knowledge of how sleep affects children’s development, disease progression and recovery. The centre is supported by a DKK 20m grant from the Lundbeck Foundation and is a collaboration between the Departments of Paediatrics at Herlev and Gentofte Hospitals and Mary Elizabeth’s Hospital. It will open in 2027
In 2025, we delivered solid financial results and maintained our grantmaking at a high level, while continuing to invest actively at the frontiers of business and science.
Group revenue grew by 9% to DKK 43,437m and our operating profit grew by 54% to DKK 7,046m, both exceeding expectations and at an all-time high. The Foundation’s financial investment activities delivered solid returns for the year of DKK 2,905m, and the profit for the year thus amounted to DKK 6,111m. As of 31 December 2025, the Foundation’s net wealth amounted to DKK 81bn, an increase of 13% compared to year-end 2024.
At the Lundbeck Foundation, we are driven by a clear purpose: Bringing discoveries to lives – for the benefit of people and society. This guides our decisions, shapes our priorities and constantly reminds us of the responsibility that follows from being one of Denmark’s largest enterprise foundations.
In a time when society faces growing health challenges, not least in the area of brain diseases, there is a strong need for scientific progress. We therefore genuinely believe that our long-term commitment matters more than ever – in business, in science and in society. Within all three areas, progress takes time, patience and willingness to invest ahead of outcomes
In 2025, the Foundation awarded grants totalling DKK 825m. A significant share (63%) of the grants was awarded through open calls, which remain the backbone of our support for public Denmark-based biomedical research with a specific focus on neuroscience
We aim to nurture talent, support ambitious research projects and foster strong scientific environments that encourage bold ideas and collaboration across disciplines in a shared effort to find new answers. For researchers and institutions, the ability to rely on stable and predictable funding is essential, and we aim to provide that continuity
At the same time, we pursue strategic philanthropic initiatives where scale and long-term engagement can help move entire fields forward. During the year, we reached important milestones, including the inauguration of the Lundbeck Foundation Parkinson’s Disease Research Centre (PACE) and the decision to establish a new national research centre for clinical psychiatry in partnership with the Capital Region of Denmark.

These initiatives are guided by our ambition to translate scientific insight into better understanding, prevention and treatment in areas of high unmet medical need. Meanwhile, initiatives such as The Brain Prize and Neurotorium serve as platforms for recognising excellence and advancing knowledge-sharing and collaboration within brain research.
Together, our open calls and strategic initiatives form a philanthropic approach shaped by our belief in scientific excellence, collaboration and the value of thinking across disciplines and institutions.
Financial strength is our basis for philanthropy
Our philanthropic aspirations rest on our ability to create value both as a long-term business owner and investor.
Through engaged ownership and disciplined financial and venture investments, we build financial strength that enables us to support stable grant-making and ambitious strategic initiatives – supporting continued investment in research, talent and scientific progress, while contributing to Denmark’s position as a strong life science nation.
In 2025, our strategic ownerships each took important steps to strengthen their businesses for the future and secure their long-term growth and value creation.
H. Lundbeck delivered significant growth driven by strong commercial performance and continued clinical advancement of its pipeline. At the same time, the
company reshaped parts of its business and established new commercial partnerships. It also completed the integration of the recently acquired Longboard Pharmaceuticals, reinforcing its strategic focus on differentiated neuroscience and future growth opportunities
Strong governance structures and transparency are essential to the way we work at the Lundbeck Foundation.
were implemented during the year to counter weaker demand in key markets and currency headwinds from a predominant exposure to US-dollar revenues.
In 2025, Lundbeckfonden BioCapital continued to drive clinical and scientific progress across its biotech portfolio, while also adding new companies, including Denmark-based Epoqe Pharma and QuantumCell. Reflecting the inherent risks associated with late-stage biotech development, certain investments did not develop as expected. BioCapital, however, maintained focus on the long-term development of the portfolio, facilitating access to new funding where needed. We remain convinced that supporting ambitious biotech ventures is an important part of our purpose of translating scientific discoveries into potential new therapies
We believe that strong governance structures and transparency are essential to the way we work at the Lundbeck Foundation and to the enterprise foundation model, and we will continue to engage openly in discussions about ownership, transparency and governance to ensure continued recognition of the Foundation’s valuable contribution to Danish society.
So, with confidence and aspirations for the future, we extend our sincere thanks to our partners for their close and valued collaboration throughout the year, and to the employees of the Lundbeck Foundation for their continued dedication and strong sense of purpose
ALK also continued its positive development in 2025, delivering solid growth and a steady margin increase. The strong execution in its core allergy immunotherapy business, including approvals for paediatric treatments and ongoing clinical progress, further strengthened confidence in the company’s strategy and growth prospects.
Falck saw a year of significant organisational changes. During the year, the company implemented a new structure aimed at reducing complexity and increasing efficiency, strengthening its operational foundation and regional anchoring.
Across our minority investments, Ellab, Ferrosan Medical Devices and WS Audiology different measures
The financial investment arm of the Lundbeck Foundation continued to deliver solid returns well above benchmark, despite sustained geopolitical unrest and volatility in financial markets. Building on previous years’ consistent value creation, in 2025 we further strengthened the Foundation’s financial robustness and capacity to support its long-term philanthropic and ownership ambitions
Looking forward, we will continue to pursue new life science business opportunities and nurture scientific excellence and collaboration through our philanthropic work and engaged ownership.

Søren Skou, Chair of the Board

Lene Skole, CEO
For the definitions of key figures, please see note 36.
2025 marked the fifth year since the launch of the Foundation’s 2030 strategy, ‘Bringing Discoveries to Lives’. With the strategy, the Foundation strives to create value across five key themes.





We create value when we fund Danishbased research that results in a better understanding of the brain and better prevention, diagnosis and treatment of brain diseases.
Our ambition is that, by 2030, Lundbeck Foundationfunded research will have led to a better understanding of the brain and to groundbreaking new programmes and treatments in clinical trials or on their way to the market.
We create value when we invest in research talents, entrepreneurs and innovation in Danish healthcare, and when we strengthen the collaboration between universities, hospitals and companies in Denmark.
Our ambition is that, by 2030, the Lundbeck Foundation will have contributed to innovative healthcare solutions and/or started new healthcare companies in Denmark.
We create value when, as a competent and engaged owner, we develop and grow healthcare companies to become international market leaders within their categories.
Our ambition is that, by 2030, we will be a long-term and significant owner of a portfolio of five to eight small or large healthcare companies, preferably in Denmark, that are international market leaders or on their way to becoming leaders
We create value when we develop and internationalise Danish healthcare research and business culture and when we improve society's understanding of the brain and its diseases
Our ambition for 2030 is that the Foundation has increased public understanding of the importance of brain health, improved the conditions for research in Denmark and further developed the role of enterprise foundations.
We create value when our return on investment enables us to increase our grants to society and secures our long-term financial robustness and growth.
Our ambition for 2030 is for our investment activities to deliver a competitive return equal to or better than that of relevant peers to at least double our net wealth, and double our average annual grants to at least DKK 1bn
The Lundbeck Foundation is an enterprise foundation engaged in a diverse range of commercial and philanthropic activities. While these activities vary greatly, they are interconnected and mutually reinforcing. The Foundation’s strategy provides a clear roadmap for value creation, with specific goals and ambitions across five thematic areas. In 2025, the Foundation continued to make progress across all themes, achieving several key milestones.

The Foundation’s scientific focus in 2025 remained centred on the tremendous complexity of the brain, one of humanity’s most challenging scientific topics. The lack of basic knowledge about the brain – both in terms of its normal functioning and its disorders – underscores the pressing need for new insights. By 2030, the Foundation therefore aims to double its average annual grants to at least DKK 1bn, with a strategic focus on neuroscience to generate knowledge that can improve prevention, diagnosis and treatment of brain disorders.
In 2025, the Foundation awarded DKK 825m. Of this total, 90% was dedicated to neuroscience research across all levels – from PhD students to professors –through strategic programmes, open calls and talent development initiatives. These efforts aim to generate knowledge that benefits patients, doctors and
therapists while further strengthening Denmark’s neuroscience community. This is achieved through a range of grants to support– or with the potential to support – research in neuroscience, and grants supporting talent development in biomedical and health-related science.
In 2025, the Foundation announced the first step in the Lundbeck Foundation’s National Strategy in Psychiatry Research, the establishment of a national centre for clinical psychiatry with its primary base at Bispebjerg Hospital in Copenhagen and satellite units across all regions of Denmark. The Foundation’s national strategy for psychiatry research has the overarching aim of providing better treatments for patients living with mental illness. The initiative comprises three closely aligned and collaborative centres: a national centre for clinical psychiatry research and two centres dedicated to basic biological psychiatry, with a focus on identifying the underlying mechanisms of psychiatric disorders.
This strategic initiative follows the first strategic initiative launched in 2024 to integrate basic and clinical research for better prevention, diagnosis and treatment of brain diseases with the establishment of the Lundbeck Foundation Parkinson’s Disease Research Center (PACE) at Aarhus University Hospital. In 2026, discussions are continuing with Danish universities and hospitals on Neurohubs and national strategic initiatives supporting translational research in neuroscience.
The Foundation also partnered with other foundations, universities and hospitals in 2025 to establish or strengthen several centres, including Brain Health Centre of Excellence, Danish Centre for Paediatric Sleep Disorders, the Nordic Centre for Glymphatic Biology and the Apollo Ancient Genomics Initiative for Evolutionary Understanding of Human Diseases.
The open knowledge platform Neurotorium, which develops and shares educational materials about the brain and its diseases, continued to grow strongly. In 2025, the platform attracted approximately 314,000 visits from 186 countries only three years after its launch in 2022. All content is curated by an international board of renowned neurologists and psychiatrists, ensuring high-quality resources.
Professor Michelle Monje and Professor Frank Winkler were awarded The Brain Prize for their pioneering contributions in incorporating neuroscience into cancer research, forming what is now called ‘Cancer Neuroscience’. The Brain Prize was again complemented by a series of seminars as part of the Foundation’s global scientific outreach programme.

Healthcare innovation and entrepreneurship require a strong connection between the scientific research community and the commercial business world. This dynamic is evident in the world’s most innovative
healthcare ecosystems, where talents seamlessly transition between science and business. However, this journey can be challenging in Denmark. Strengthening the dialogue and collaboration between Denmark’s academic research institutions and the local biotech and pharmaceutical communities is a key aspiration for the Foundation. The goal is to empower academic talent to explore, innovate and ultimately make the leap from science to business, enabling discoveries to scale and transform lives. The Foundation employs several instruments to achieve this ambition.
To bridge the gap between basic research and commercial viability, the Foundation introduced Frontier Grants in 2022. These grants support the transition of early-stage research projects into opportunities attractive to biotech investors. In 2025, four Frontier Grants were awarded to scientists working on projects related to exploring a new obesity drug, genome stability in neurodegenerative disorders, breast cancer and osteoporosis. In 2025, Seed Grants were introduced to support maturing projects to potentially receive a Frontier Grant with two projects in Parkinson’s and Alzheimer’s disease being supported.
Launched in 2022, Lundbeckfonden BioCapital is an evergreen life science venture fund that builds on the Foundation’s decade-long commitment to creating and financing biotech companies. Its focus is on translating pioneering research into innovations that address areas of high unmet medical need through novel and/or superior treatments
In 2025, Lundbeckfonden BioCapital continued to prioritise the local Danish biotech ecosystem, including two new seed investments in Denmark, Epoqe Pharma and QuantumCell The portfolio consist of 18 companies.

The engaged ownership of Danish healthcare companies lies at the core of the Foundation’s value creation. These companies develop products and treatments that benefit patients worldwide while generating financial returns that enable new investments in science and business development. As part of the 2030 strategy, the Foundation set a clear goal of expanding its portfolio to include five to eight Danish healthcare companies by 2030
In 2025, the Foundation’s portfolio of strategic ownerships includes a total of six listed and private healthcare companies headquartered in Denmark with majority stakes in H. Lundbeck, ALK-Abelló, Falck, and significant minority stakes in Ellab, Ferrosan Medical Devices and WS Audiology.
The Foundation’s ambition is to grow and develop its ownerships on a global scale, helping these companies become leaders in their respective industrial segments while bringing innovations, products and treatments to patients worldwide.
Bringing discoveries to lives is the common thread for our commercial and philanthropic endeavours

The aim of a proactive public presence is to enable our strategic objectives by raising awareness of brain health amongst decision makers and support better research conditions in Denmark and advance the enterprise foundation model as a responsible, sustainable approach to business ownership. To achieve this, the Foundation engages in a variety of activities spanning communication, partnerships and public affairs across its entire value chain.
As an enterprise foundation with significant ownership interests and a mission-driven investment approach, the Foundation operates at the intersection of research, business and society, and active engagement in public dialogue is therefore a strategic necessity.
From a business perspective, public affairs engagement helps safeguard stable and predictable framework conditions that enable long-term ownership,
responsible governance and sustainable value creation. Clear and future-proof regulation is critical for foundations to act as long-term owners, support innovation and reinvest returns into research and societal priorities. By engaging constructively with policymakers and authorities, the Foundation contributes knowledge and perspectives that support sound decision making and reduce regulatory uncertainty.
Promoting understanding of the Danish foundation model is a core element of this engagement. Enterprise foundations play a distinctive role in the Danish economy by combining commercial strength with longterm stewardship and targeted social investment. Through public affairs and communication activities, the Foundation works to ensure that the contribution of the foundation model, including stability, resilience and reinvestment in society, is recognised and reflected in policy development.
By communicating clearly about the Foundation’s purpose, governance and impact, as well as about brain research and brain health, we strengthen our legitimacy as both an owner and a philanthropic actor. Open communication also helps build public understanding of how long-term business ownership and philanthropic objectives can be mutually reinforcing.

The investment activities of the Foundation are split into three categories: Lundbeckfonden Strategic Ownerships, Lundbeckfonden Invest (financial investments) and Lundbeckfonden BioCapital (biotech investments)
The overarching ambition for all investment teams toward 2030 is to deliver annual financial returns that exceed those of market peers. This includes doubling the Foundation’s net wealth by 2030 compared to 2020 level and growing the Foundation’s philanthropic grants to an average of at least DKK 1bn annually by 2030 while ensuring long-term financial robustness.
Building on the momentum from 2024, solid returns were delivered in 2025, with the Foundation’s total wealth increasing to approximately DKK 81bn from DKK 71bn at the end of 2024


The Lundbeck Foundation is a long-term owner of multiple healthcare companies headquartered in Denmark. Our ambition is to position and maintain the companies as international market leaders within their industry segment. Business ownerships is a key element of the Foundation's financial and societal value creation and of building the route to market for new treatments and therapies for patients all over the world.
Financial investments ensure long-term financial stability and robustness, and allow the Foundation to invest in the future of healthcare and support new scientific research. The Foundation's biotech investments contribute to the financial value creation while also developing people and businesses in the Danish life science ecosystem.
• Supporting public research and science through grants and activities to bring forward new discoveries
• Disseminating knowledge through programmes and activities
• Strengthening a vibrant Danish ecosystem for life science, research, innovation, and economic growth
The Foundation channels profits back to society through grants and partnerships, which are anchored in the Foundation’s purpose of 'Bringing discoveries to lives'. Our strategic ambition is to improve the world's understanding of the brain and create better treatments of brain disorders while also developing the Danish life science ecosystem by improving conditions for science, innovation and economic growth in Denmark.
The Lundbeck Foundation’s ambition is to make Denmark a frontrunner in the field of neuroscience by funding Danish-based research that promotes an improved understanding of the brain as well as better prevention, diagnosis and treatment of brain disorders. The Foundation also supports activities to improve collaboration between research and business.
In the Foundation’s 2030 strategy, one of the main goals is to bring discoveries to lives by supporting a research environment that strengthens the collaboration between basic and clinical research, with the aim of better prevention, diagnosis and treatment of brain diseases. In 2025, the Foundation announced the first step in the Lundbeck Foundation’s National Strategy in Psychiatry Research, the establishment of a national centre for clinical psychiatry, in collaboration with Region Hovedstaden, located at Bispebjerg Hospital (see page 18).
National strategic initiatives involve two or more research centres with complementary competencies working towards a common goal.
In 2026, discussions are continuing with Danish universities and hospitals on neuroscience research hubs and national strategic initiatives supporting translational research in brain disorders.
Besides strategic initiatives, the Foundation continued its support of outstanding biomedical research, with a strong commitment to talent development and research in basic and clinical neuroscience.
Alongside research grants and prizes, the Foundation supported activities to strengthen and facilitate networks between scientists, as well as, to increase awareness about the brain and the importance of brain health among the Danish population.
The total grant sum amounted to DKK 825m, versus DKK 1,074m in 2024.
The Nordic Centre for Glymphatic Biology The Foundation awarded “Centre for Translational Neuromedicine” at the University of Copenhagen DKK 78m to the “Nordic Centre for Glymphatic Biology” for identifying and validating targets which can alter the glymphatic flow to treat neurological disorders, in particular Small Vessel Disease. The project will also include Nordic collaborators in the development of clinical biomarkers for glymphatic flow. The centre is led by professor Maiken Nedergaard.
Danish Centre for Paediatric Sleep Disorders at Rigshospitalet: The centre’s mission is to establish a research facility to study sleep mechanisms in children and adolescents to provide evidence-based solutions that promote better sleep. The centre will be led by researcher and neuropaediatrician Nanette Debes. The centre has been awarded DKK 20m.
Brain Health Center of Excellence: The Foundation has allocated a total of DKK 160m to a five-year partnership with BioInnovation Institute (BII) to strengthen the Danish ecosystem for brain health research, innovation and entrepreneurship. BII will support the programme with an equivalent amount.
The Brain Health Center of Excellence aims to create new groundbreaking treatments for brain diseases. It seeks to strengthen collaboration between the university environment and the start-up ecosystem by investing in research talent. Three projects were initiated in 2025 with a total grant of DKK 10m from the Lundbeck Foundation.
Total grants (DKKm)
825
Number of grants
185
Support for brain research (DKKm)
740
Number of full-time employees funded
736
Apollo Ancient Genomics Initiative for the Evolutionary Understanding of Human Diseases The Foundation awarded DKK 15m to the programme, which is an extension of “Centre for Disease Evolution”, which was previously funded by the Lundbeck Foundation. The programme will expand the previous data on ancient human DNA and microbial/pathogen DNA and - in combination with data from the Novo Nordisk Foundation funded programme AEGIS on ancient environmental data - seek to further uncover the evolutionary origins of human disease. The Novo Nordisk Foundation co-funds the project with DKK 15m.
Neuroscience Academy Denmark has awarded 16 new 1+3-year PhD fellowships. This is the fourth cohort out of the six cohorts granted by the Lundbeck Foundation.
The additional grants were distributed across the following categories:
Grants supporting research in neuroscience
Grants in this category support research aimed at solving a fundamental problem in neuroscience or research into brain-related diseases. The grants were:
• LF Professorships support the very best neuroscience researchers, thereby helping to build excellent research environments and programmes in Denmark. DKK 159m was awarded to five professors covering research in the ageing motoneuron-muscle axis, the dopamine-transporter complex, motor circuits, radioligand therapy for glioblastomas and cognitive enhancement across diseases.
• LF Collaborative Projects focus on complex, basic or clinical neuroscience projects that require collaborative efforts. LF Collaborative Projects alternate biannually with LF Professorships and will therefore be granted in 2026. The programme’s international profile will be strengthened by, for the first time, inviting European researchers, collaborating with Danish groups, to apply as principal investigators.
• LF Start-up Programme grants to support recruiting international or industry-based scientists to Danish universities. DKK 11m was awarded.
• LF Stipends for Sabbatical Leave, Visiting Professorships, and International Meetings and Conferences support internationalisation by enabling international exchange either out of or into Denmark, as well as Danish scientific gatherings open to international participants
Grants in this category broadly support research or technologies where the applicant can propose a potential benefit for neuroscience and therapies for nervous system disorders. The major grants were
• LF Postdoc grants support postdoctoral scientists who wish to strengthen their research career through changes in research environment or topics. DKK 50m was awarded to 17 postdocs, of which 12 bring international experience to Denmark and five gain
international experience by studying outside Denmark.
Lundbeck Foundation and Novo Nordisk Foundation Body-Brain Interdisciplinary Projects. The Foundation will, in collaboration with the Novo Nordisk Foundation, announce an international collaborative grant to study BrainBody interactions in 2026
• LF Ascending Investigator grants are for independent researchers leading smaller groups with projects aimed at expanding the groups and/or redirect their research. DKK 90m was awarded to 15 investigators (four professors, eight associate professors and three group leaders). Six grantees used the option to add an additional DKK 1m to support international exchange, whereas an additional six also had international collaborators as a part of their project proposal.
• LF Experiment. The call focused on funding high-risk, ground-breaking research ideas. DKK 58m was awarded to 26 projects.
• LFIN The Lundbeck Foundation Investigator Network facilitates collaboration and community building among 50 selected junior scientists from all over Denmark and received a grant of DKK 1m. LFIN held two meetings in 2025, as well as arranging several events, including a winter school for master students and open lectures for the public.
Grants supporting research in biomedical science
Grants in this category support the development of talented researchers in all fields of biomedical and health-related science. The grants were:
• LF Early-Career Clinician Scientist grants aim to support talented medical doctors who, concurrent with their clinical specialisation, want to maintain an active research career. DKK 18m was awarded to ten projects and four of the projects were in neuroscience (auditory dysfunction in schizophrenia, serotonin receptor levels in the brain, pain in idiopathic intracranial hypertension, effectiveness of psychopharmacological treatment in bipolar disorder).
• LF Fellows are talented, young researchers who wish to establish or expand their own independent research groups in Denmark. DKK 70m was awarded to seven scientists: three in neuroscience (migraine, proteinopathies and stress), and four in other areas (cellular metabolism, genome stability, biotechnology and colorectal cancer).
The Brain Prize is the world’s largest international neuroscience prize, dedicated to scientists who have made an outstanding contribution to neuroscience. His Majesty The King of Denmark presented The Brain Prize to the 2025 winners at an awards ceremony in Copenhagen. The DKK 10m Brain Prize 2025 was awarded to Professor Michelle Monje and Professor Frank Winkler for pioneering the field of Cancer Neuroscience. Working independently, they have transformed our understanding of the biology of glioblastomas, thereby pioneering a paradigm shift incorporating neuroscience into cancer research, forming what is now called ‘Cancer Neuroscience’.
Brain Prize outreach activities bring the outstanding science behind each year’s Brain Prize to the scientific community. Three webinars on “The neuroscience of Brain Cancers” were organised and chaired by the Brain Prize winners together with six other leading scientists who provided in-depth insights. A series of Brain Prize plenary lectures were also given at several international meetings. A film collection covering the winners’ scientific journey, patient case stories and educational films has been launched
The LF Scientific Enrichment Prize is a personal prize of DKK 100,000, awarded in 2025 to Professor Lene Niemann Nejsum, Aarhus University to recognise her efforts to prioritise inclusivity, respect and opportunity and to foster collaboration among researchers with different nationalities, genders, physical challenges and neurodivergence. She actively seeks to include and train people in “FlexJob” positions, creating roles tailored to the individual for them to advance the ongoing research. In addition, she collaborates with a range of scientific fields to bridge the gap between basic and clinical science.
The LF Young Investigator Prize of DKK 1m, for promising researchers under the age of 40, was awarded to Professor Anton Pottegård, Department of Public Health, University of Southern Denmark for his outstanding contribution to pharmacoepidemiology by developing analytical tools to identify patterns for additional and side effects of medicines used in clinical practice. The results of this research have led to changes in several clinical guidelines.
Five LF Talent Prizes of DKK 300,000 each for promising young researchers under the age of 30 were awarded to MD, PhD student Adelina Yafasova, Department of Cardiology, Rigshospitalet (interplay between multiorgan diseases and cardiovascular conditions); MD, PhD student Rune Häckert Christensen, Danish Headache Center, Rigshospitalet (migraine and post-traumatic headache); MD, PhD Mohamed Attauabi, Department of Gastroenterology and Hepatology, Herlev Hospital (Early prognostication of Inflammatory Bowel Disease); MD, Stine Johansen, Center for Liver Research, Odense University Hospital and University of Southern Denmark (alcohol-related liver disease) and MD, PhD student Mark Khurana, University of Copenhagen (infectious disease research).
The Foundation supports projects that inspire and engage young people to focus on science, particularly neuroscience, as a career and projects that communicate knowledge about the brain and brain disorders to the public. In 2025, a range of projects and festivals in Denmark were supported, including the Hearts & Minds Festival in Aarhus, Bloom Festival in Copenhagen, and several podcasts and documentaries, with a particular emphasis on reaching younger audiences and promoting brain health awareness. Notable initiatives include podcasts on child and adolescent brain development and psychiatric disorders, documentaries on postpartum depression and rare diseases, as well as public science events like Science & Cocktails and the “Neuroskib” at The People’s Meeting (Folkemødet), which aim to foster dialogue about
neurological conditions and mental health. In 2025, the Foundation granted DKK 31m to 15 projects, including DKK 16m to recurrent programs.
Frontier and Seed Grants serve a dual purpose by supporting advanced research ideas to a stage where they can become attractive for investors while at the same time supporting the training of the lead scientists. Four grants totalling DKK 19m were awarded to explore a new obesity drug, genome stability in neurodegenerative disorders, breast cancer and osteoporosis. Seed grants were introduced in 2025 to support maturing CNS projects to potentially receive a Frontier Grant. Two projects, in Parkinsons and Alzheimer’s disease, were supported with DKK 2m.
Neurotorium is an open, online platform (www.neurotorium.org) launched in 2022 aimed at clinicians and educators of clinicians, covering topics about the brain and its diseases and attracting approximately 314,000 visits in 2025.
Two new themes, Epilepsy and Anxiety disorders, have been added to the Neurotorium platform, now covering a total of ten themes. In addition, workshops, including an expert meeting on “Advances in Cognition in Brain Disorders”, and scientific symposia at conferences have been organised and supported. 13 international educational grants (DKK 2m) have been awarded.
Nadija Research Institute: DKK 7m has been awarded to research projects in Ukraine aiming at preventing or
reducing war traumas in Ukrainian children and, in addition, contribute globally to the understanding and treatment of childhood trauma in conflict zones.
Promote Danish life science: Healthcare Denmark has been awarded DKK 5m to develop new programmes to promote Danish life science. The aim is to develop Innovation District Copenhagen by enhancing the number of delegation visits and showcasing the Danish life science ecosystem internationally.
Research stipends to the Center for Health Research: DKK 2m has been awarded to support research training for healthcare professionals with a professional bachelor’s degree.
Pre-Graduate scholarships: Grants totalling DKK 9m were awarded to seven scientific societies that together fund approximately 35 individual pre-graduate scholarships. For the first time, the medical societies had the possibility to apply for additional funds for sending students to international research environments.
Scientific Enrichment: Three grants were awarded a total of DKK 1m for initiatives promoting diversity in the biomedical and clinical fields. The project “The Future of Health Research: Partnerships and Diversity among Researchers, Patients and Relatives” at Odense University Hospital will investigate the best practice of triaging researcher, patient and relative involvement when performing a research project - resulting in guidelines for more inclusive and diverse research
methods ensuring the research mirrors real-life complexity.
SPINE - Strengthening Pathways and Infrastructures for Navigating interdisciplinary collaboration in Existing academic structures: The SPINE project, led by Assoc. Professor Katrine Ellemose Lindvig, addresses the critical gap between the recognised importance of interdisciplinary research and practical challenges. The project will offer novel approaches to how interdisciplinary research is supported and sustained at the doctoral level. The project was awarded DKK 8m.
Innovation Discovery Network (ODIN)
projects: The Foundation has awarded DKK 14m to ODIN to support innovation within drug discovery/diagnostics to treat CNS disorders by facilitating co-creation of novel research projects between academia and industry/biotech. The grant provides funding for the academic project part.
The Foundation has implemented a qualitative approach to maximising the impact of LF funding, termed ”Roadmap to Impact”. Applicants are required to describe in concrete terms who will use their results, should the project turn out as anticipated, and how these users will benefit from the results. Focusing on the next steps after their own project stimulates the applicants to position their research project in the process of bringing discoveries to lives. Grantees must report on the progress of the ”Roadmap to Impact” in the annual status reports.
Learn more about our grants & prizes and what we have funded at lundbeckfonden.com
Research hubs are a central component of the Foundation’s strategic initiatives aimed at building strong and sustainable research environments in Denmark.
These hubs are designed to co-locate basic and clinical research, facilitating the close exchange of knowledge required to address major unmet medical needs.
An urgent need for better treatments Parkinson’s disease is the second-most common neurodegenerative disorder and the world’s fastestgrowing neurological disease in terms of prevalence, affecting more than six million people worldwide. It is a devastating condition for patients and their families. Consequently, there is an urgent need for diseasemodifying treatments and effective prevention strategies.
Discoveries should benefit patients within ten years
The focus of the Lundbeck Foundation Parkinson’s Disease Research Centre (PACE) will be to establish cohorts of patients and presymptomatic individuals in order to investigate novel treatments and better understand the mechanisms leading to Lewy body diseases, with the aim of identifying new therapeutic targets.
PACE aims to bring innovative diagnostic tools and therapies into early clinical testing within ten years.
Recent research has provided new insights into the underlying pathological mechanisms of Parkinson’s disease and other Lewy body diseases, which are driven by the propagation and aggregation of the protein αsynuclein in brain cells.
These advances have raised hopes for earlier and more biologically defined diagnoses of the different Lewy body diseases, and part of the research activities at PACE will focus on understanding how α-synuclein aggregation drives disease progression and how genetic and environmental factors influence disease development.
This work will involve deep and objective patient phenotyping to enable biological subtyping, biomarker discovery and the development of personalised treatment approaches. Through this integrated strategy, the centre aims to bring innovative diagnostic tools and therapies into early clinical testing within ten years.
PACE sets out to establish a unique environment by bringing together experts from many different research fields. Approximately 70 people are expected to work at the centre, which will be located at Aarhus University Hospital to ensure close integration with clinical activities. The centre will be part of the Danish Neuroscience Center.
The structure of the Foundation’s research centres is developed in collaboration with academic institutions and is based on research proposals submitted by worldleading scientists within the relevant research fields

Research in psychiatry is a key focus area for the Lundbeck Foundation, aimed at advancing understanding of the causes of mental illness and generating knowledge to improve treatment options for affected individuals.
The Foundation’s national strategy for psychiatry research
The Foundation’s national strategy for psychiatry research has the overarching aim of providing better treatments for patients living with mental illness. The initiative comprises three closely aligned and collaborative centres: a national centre for clinical psychiatry research and two centres dedicated to basic biological psychiatry, with a focus on identifying the underlying mechanisms of psychiatric disorders.
Mental illness is prevalent in society, and most individuals will, at some point in their lives, experience a mental illness requiring treatment. Mental illnesses are complex and encompass a wide range of conditions that profoundly impair the lives of affected individuals as well as their relatives.
Beyond the substantial impact on daily life function, mental illness is associated with reduced life expectancy and limited ability to work, and it constitutes a significant societal and socio-economic burden.
This unmet need underscores the urgency of developing new and better treatments, as well as improving early detection, intervention and prevention of onset of mental illness.
Denmark is uniquely positioned to advance research in clinical psychiatry, supported by comprehensive national registries, population-based biobanks, genetic databases and a nationwide healthcare system.
Together, these resources enable the integration of clinical, epidemiological and genetic data, creating exceptional opportunities for world-class research.
In 2025, the Foundation entered into a partnership with the Capital Region of Denmark to establish a nationally anchored research environment, with its primary base at Bispebjerg Hospital in Copenhagen and satellite units across all regions of Denmark.
The ambition is to establish a world-leading centre that delivers direct clinical benefits to patients, advances understanding of mental illness and attracts leading international scientific talent to Denmark. In the short term, this will lead to new research programmes and the initiation of novel treatments in clinical trials, while in the long term it will contribute to better treatment for patients with severe psychiatric disorders.
Through workshops and interviews the Foundation has engaged in extensive dialogue with Danish and international researchers, as well as with representatives from the Danish Health Authority, relevant ministries, universities, healthcare regions and patient organisations, to define the research focus for the coming centre and to ensure strong anchoring within the Danish research environment.
This collaborative process has resulted in the development of a scientific strategy with two key focus areas:
• Mental illness within a neurodevelopmental framework
• Treatment-resistant mental illness
The ambition is to establish a world-leading centre that delivers direct clinical benefits to patients, advances understanding of mental illness and attracts leading international scientific talent to Denmark.
During 2026, a clinical director will be recruited for the centre. The appointed individual will be responsible for developing a research strategy that defines the focus areas of the centre’s research activities.
Our proactive public presence is instrumental in achieving our strategic goals. By raising awareness about brain health among decision-makers and the general public, we aim to support better research conditions in Denmark and promote the enterprise foundation model as a responsible and sustainable approach to business ownership.
Raising awareness of brain health remains a key priority for the Foundation. Throughout 2025, the Foundation continued to engage decision makers and the wider public to strengthen understanding of how brain health can be promoted and what it means to live with a brain disorder.
The Foundation carried out a series of targeted outreach activities designed to translate scientific knowledge into public insight and dialogue. As part of the Brain Awareness Week, the Foundation hosted public events in Aarhus and Copenhagen. With a focus on ADHD, leading experts explored the biological and environmental factors behind the condition, differences between children and adults and the impact of gender in diagnosis and treatment. Under the overarching theme “Our Remarkable Brain”, the programme also
highlighted current research on sleep, memory, brain stimulation and pain.
At The People’s Meeting (Folkemødet) in Denmark, the Foundation organised events addressing key aspects of brain health, including the importance of brain breaks, sleep, recent advances in brain research, neurodiversity and how to foster more inclusive workplaces.
In October, during Copenhagen annual Culture Night, the Foundation hosted two events at Panum, the Faculty of Health and Medical Sciences at the University of Copenhagen. Here, members of the public were invited to engage with researchers and learn about the critical role of sleep for brain function and overall health.
Together, these activities reflect the Foundation’s continued commitment to bringing brain research closer to society and contributing to a more informed and inclusive public conversation on brain health
Throughout 2025, the Foundation maintained its emphasis on attracting international talent to Denmark. One initiative is Science Hub Denmark, which positions Denmark as a leading research destination in neuroscience to prestigious universities, mainly in the US. Guided by its strategy of Bringing Discoveries to Lives, the Foundation has also supported a number of new initiatives reflecting its commitment to innovation in life science, including its role as a founding member of Innovation District Copenhagen, an organisation
which aims to establish a hub in the Capital of Denmark for life sciences and quantum technology by building bridges between research environments and startup communities.
The foundation model and the role of enterprise foundations in society
In 2025, the Foundation continued its efforts to promote awareness and recognition of the unique Danish foundation model. Through active participation in public debate and key dialogue forums, we highlighted the societal value created by enterprise foundations through long-term ownership, stability and targeted social investment.
A consistent priority during the year was to contribute to the clarification and further development of the framework conditions governing the foundation model. While a final political resolution remains pending, we worked purposefully to ensure that the issue remained high on the public and political agendas. This included active engagement at The People’s Meeting as well as presentations and dialogue sessions with stakeholders such as PwC, Forenet Kredit and the Ministry of Industry, Business and Financial Affairs. In these forums, we emphasised the importance of clear and futureproof regulatory conditions that enable foundations to act as responsible and long-term contributors to society.

The Lundbeck Foundation engages people across Denmark through wellattended public events on the brain and brain health, from The People’s Meeting (Folkemødet) to lectures, fostering dialogues

A portfolio of six listed and private healthcare companies headquartered in Denmark.
The Foundation is a long-term owner of healthcare companies based in Denmark which are either global leaders in their respective market segments or striving for leadership positions. The Foundation invests in both public and private companies and can take minority as well as majority ownership positions. The ambition is to own between five and eight companies in this category by 2030. The Foundation is looking to invest in companies rooted in Denmark with the potential to become global leaders in their fields.
The Foundation’s aim is to develop both small- and large-scale healthcare companies and help them become global leaders. The Foundation can do this because skills have been built in the field of healthcare throughout the Foundation’s existence. Over the past decades, the Foundation has established itself as a competent and engaged owner.
As an enterprise foundation, the Foundation takes a long-term approach to the strategic ownerships and can ensure the right conditions for these companies to become global leaders in their respective fields. The enterprise foundation model represents a strong and engaged type of ownership that enables long-term value creation and contributes to society by - in the case of the Foundation - awarding grants to Danish-based
health sciences research, supporting the Danish biotech ecosystem and investing in Danish healthcare companies. The Foundation seeks to increase awareness of the enterprise foundation model among companies as a sustainable ownership model
What we invest in
• Healthcare companies headquartered in Denmark
• Companies leading in their field, or with the potential to become a leader and
• Companies delivering financially attractive returns that could contribute to growing the Foundation’s net wealth long term
The portfolio
• Majority-owned strategic ownerships
• H. Lundbeck
• ALK-Abelló
• Falck
• Minority-owned strategic ownerships
• Ellab
• Ferrosan Medical Devices
• WS Audiology

in the development of innovative therapeutics for brain disorders


Leading global developer and producer of a wider range of allergy treatments, products and services that meet the unique needs of allergy sufferers. Leading global player within high-end validation and monitoring solutions, as well as field service and consulting to primarily life science customers.

supplier of healthcare and emergency

Lundbeck is a global pharmaceutical company specialised in developing innovative treatments for brain diseases. The Lundbeck Foundation owns 69% of the share capital and 76% of the votes.
2025 was a truly remarkable year, marked by recordbreaking results and double-digit growth. In the relentless pursuit to advance brain health, Lundbeck has focused on transforming the business to secure longterm growth while improving patients’ access to innovative treatments within neurology and psychiatry.
In 2025, Lundbeck continued to execute the Focused Innovator Strategy, driving the transformation of the business, and fully integrating a focused innovator mindset. Strategically, Lundbeck focuses on neuro-rare and neuro-specialty conditions, expanding from the strong legacy within psychiatry and neurology.
During the year, Lundbeck transitioned to a partner-led commercial model in 27 markets, focusing resources where Lundbeck can make the greatest impact for patients and society. This partner-led model is part of the reallocation of funds for further investment in key growth areas and an innovative pipeline.
While reducing operational complexity and focusing the resources, the new commercial model means that Lundbeck said goodbye to 602 employees across the 27 countries affected. Lundbeck has supported the employees impacted in the best possible way to ensure a responsible and orderly process.
Equally important to securing long-term growth, Lundbeck continued building a sustainable pipeline set to deliver breakthrough products and long-term sustainable growth.
Lundbeck’s pipeline continues to evolve, with significant progress achieved in 2025. In the relentless pursuit to advance brain health, Lundbeck focuses on scaling its neuro-specialty position and building a neuro-rare franchise, expanding from the strong legacy within psychiatry and neurology.
Amlenetug (anti-α-synuclein) targeting MSA, and bexicaserin, targeting DEEs, are both progressing through Phase III trials. These assets represent promising opportunities to address brain disorders with high unmet needs
Also, in neuro-rare, notable pipeline milestones include the orphan drug designation granted to Asedebart (Lu AG13909, anti-ACTH) in both the U.S. and EU for the treatment of congenital adrenal hyperplasia. This development validates Lundbeck’s entry into targeted neuro-hormonal disorders, underscoring the ability to address medical needs in niche areas.
In the neuro-specialty area, results from two studies reinforced the clinical strength of Vyepti® in migraine treatment. Lu AG09222 (anti-PACAP), which represents a potential new therapeutic option for the treatment of migraine, also progressed in line with expectations.

Revenue (DKKm)
24,630
Operating profit (DKKm)
5,275
Net profit (DKKm)
3,192
Number of employees (FTEs)
In 2025, Lundbeck achieved a record revenue of DKK 24,630m (DKK 22,004m), driven primarily by strong performance in the U.S. and Europe. Revenue of Lundbeck’s strategic brands increased by 15% to DKK 19,011m, representing 77% of total revenue. Approximately 90% of the strategic brands growth was attributable to the strong performance of Vyepti® and Rexulti® in the U.S.
The U.S. constituted 56% of total revenue in 2025 and represented Lundbeck’s largest market. Sales of the strategic brands reached DKK 12,331m, increasing 20% and representing 93% of the revenue in this market. Vyepti® was the primary growth contributor, driven by an increase in demand underpinned by new patient starts, strong patient conversion, improved persistency and increased 300mg utilisation. Europe also saw robust revenue growth driven by higher sales across all strategic brands. International markets revenue declined, driven by generic competition for Brintellix® in Canada, Brazil and China
Cost of sales increased by 1% to DKK 4,265m in 2025, mainly driven by costs related to a manufacturing contract for amlenetug as well as an environmental provision, partly offset by the reversal of the Vyepti® provision for inventory obsolescence of DKK 389m, triggered by Vyepti®’s commercial performance. The gross margin was 82.7%, representing an increase of 1.9 percentage points compared to last year.
Sales and distribution costs reached DKK 7,743m, corresponding to a decrease of 5%. The S&D ratio decreased by 5.6 percentage points to 31.4%, primarily reflecting leverage from the strong revenue growth and improved cost efficiency. The S&D ratio development reflects the successful execution of the Focused Innovator Strategy in 2025 including the Trintellix® transition in the U.S., alongside disciplined resource allocation and capital reallocation.
Administrative expenses reached DKK 1,483m in 2025. The administrative expenses ratio decreased by 0.5 percentage points to 6.0%.
R&D costs reached DKK 4,895m in 2025, with an R&D ratio of 19.9%. The development in 2025 reflects the continued commitment to innovation and was primarily driven by advancing key pipeline programmes, including bexicaserin and amlenetug, as well as ongoing progress in anti-ACTH and anti-PACAP programmes.
Operating profit grew by 61%, thereby reaching DKK 5,275m in 2025. The operating profit margin reached 21.4% compared to 14.9% in 2024.
Net financial expenses amounted to DKK 788m, primarily driven by higher interest costs due to new debt raised in connection with the acquisition of Longboard as well as unfavourable foreign exchange effects mainly due to the USD depreciation.
The effective tax rate for 2025 was 28.9% compared to 15.5% for 2024. The tax rate is negatively impacted by a one-off item related to the conclusion of an agreement with the tax authorities for the transfer of Vyepti® product right as well as the effect of an impairment loss regarding the planned divestment of Lundbeck Pharmaceuticals Italy S.p.A. and the new commercial operating model, where it is not expected that tax deductibility can be utilised. Profit for 2025 reached DKK 3,192m (DKK 3,143m).
5,461 Figures for 2024 are shown in brackets
ALK is a global specialty pharmaceutical company focused on allergy and allergic asthma. ALK markets allergy immunotherapy (AIT) and anaphylaxis treatments for people with allergy. The Lundbeck Foundation owns 40% of the share capital and 67% of the votes.
ALK now has a solid earnings platform and is wellpositioned to continue delivering stable, high organic growth while maintaining an EBIT margin of around 25% in the coming years. As ALK continues to upscale operations, ALK’s strong financial position enables it to increasingly look at inorganic growth opportunities such as in-licensing and acquisitions, while also distributing excess cash to shareholders
Anaphylaxis
ALK delivered on its long-term financial targets in 2025 following strong execution of strategic initiatives across disease areas. Momentum is strong and the financial position solid. 2025 marked the seventh consecutive year of revenue and earnings growth in ALK. Results clearly exceeded initial expectations with revenue growing by 15% in local currencies and the EBIT margin improving by 6 percentage points to 26%.
Delivering on the 25-in-25 EBIT margin target was a significant milestone. ALK set this target in February 2021 on the back of the 2020 financial statements, which showed an EBIT margin of merely 4%. This means that ALK has been growing its EBIT margin by more than 20 percentage points, while still allocating significant resources to ongoing growth investments. This has only been possible exclusively owing to careful prioritisation and resource allocation to high-impact growth levers.
In 2025, ALK started launching the first-ever nasal adrenaline spray neffy® (branded EURneffy® in Europe) for emergency treatment of potentially life-threatening allergic reactions (anaphylaxis). Under a license agreement with US-based ARS Pharma, ALK holds exclusive rights to the product in all territories outside the U.S., Australia, New Zealand, Japan and China.
ALK’s roll-out initially focused on Germany and the UK, the markets expected to become the most important contributors to neffy® sales growth. EURneffy® was launched In Germany in late June and had gained a market share of close to 18% (by value) by the end of the year. The launch in the UK followed in October and attracted significant interest from healthcare professionals, key opinion leaders and the media. neffy® is currently under regulatory review in Canada and the outcome of this process is expected in the first half of 2026
In the U.S., ALK and ARS Pharma expanded their partnership with a co-promotion agreement where ALK
is responsible for selling neffy® to approximately 9,000 named paediatricians.
The paediatric roll-out of the house dust mite (HDM) tablet ACARIZAX®/ODACTRA® began in Europe in January and progressed market by market, following subsequent approvals and market access processes. By year-end, the house dust mite tablet was approved for paediatric use in 30 countries and launched in 21 of them.
The roll-out of the tree pollen allergy tablet ITULAZAX®/ITULATEK® for children and adolescents started in April, based on regulatory approvals from the EU and Canada. By year-end, the tablet was approved for children and adolescent use in 20 countries and launched in 12 of these markets

Revenue (DKKm)
6,312
Operating profit (DKKm)
1,654
Net profit (DKKm)
1,197
Number of employees (FTEs)
2,737
In China, ALK partnered with Changchun GeneScience Pharmaceutical Co. Ltd. (“GenSci”) to accelerate sales of ALK’s house dust mite products in the country with the highest number of house dust mite allergy sufferers worldwide. GenSci plans to allocate a significant sales force and conduct a wide range of market-building activities to promote ALK’s products and become AIT market leader.
ALK received DKK 244m in upfront payments from GenSci. Until 2039 (the expected lifetime of the partnership), ALK is further eligible for DKK ~300m in milestone payments related to regulatory progress with ACARIZAX® and commercial milestone payments of up to DKK ~780m, subject to future in-market sales in Mainland China. Furthermore, ALK will receive income from supplying GenSci with products
The Phase II trial for the peanut SLIT tablet is on track to deliver topline data in Q2 2026. Subject to these results, ALK plans to progress into Phase III, and preparations are ongoing. The U S. FDA has granted a Fast Track designation to the peanut programme, enabling ALK to benefit from more frequent interactions and additional guidance from the FDA.
ALK’s partner ARS Pharma initiated a Phase II clinical trial with patients in the U.S. and Europe to evaluate neffy® in the treatment of acute flares associated with chronic spontaneous urticaria. First read-outs from this trial are expected in 2026. ALK holds exclusive rights to
this and any other new indications for neffy® in the licensed territories.
2025 revenue grew by 15% in local currencies to DKK 6,312m (DKK 5,537m), following double digit growth in all sales regions. Respiratory tablets and anaphylaxis products (Jext® and neffy®) were key contributors to growth. Exchange rates impacted reported revenue growth negatively by approximately 1 percentage point.
Revenue in Europe grew by 14% to DKK 4,459m (DKK 3,914m) on broad-based growth across the region, including the region’s largest markets Germany and France. Revenue in North America increased by 19% in local currencies to DKK 1,037m (DKK 906m), fuelled by tablets and Anaphylaxis & other products. The U.S. business recovered from last year’s stagnation and reported double-digit growth, while growth was even in Canada, where tablets are the main product line.
Cost of sales increased by 5% in local currencies to DKK 2,078m (DKK 1,985m). The gross profit of DKK 4,234m (DKK 3,552m) yielded a gross margin of 67% (64%), driven by increased sales volumes, a more favourable sales mix and production efficiencies.
R&D, sales and marketing and administration costs increased by 6% in local currencies to DKK 2,581m (DKK 2,464m). R&D expenses increased by 15% to DKK 609m (DKK 531m), mainly reflecting funding of the peanut
tablet clinical trial, pre-clinical development projects and the bridging trial of ACARIZAX® in China. Sales and marketing expenses increased by 3% to DKK 1,584m (DKK 1,564m), driven by the launches of paediatric tablets and neffy®. Administrative expenses of DKK 388m (DKK 369m) increased by 4%.
Operating profit improved by 53% in local currencies to DKK 1,654m (DKK 1,091m), raising the EBIT margin to 26% from 20% in 2024. Progress was driven by higher sales, improved gross margin and a lower capacity costto revenue ratio of 41% (45%). Exchange rates impacted growth in reported EBIT negatively by approximately 1 percentage point.
Net financials showed a loss of DKK 19m related to interest expenses and exchange rate losses.
Tax on profit totalled DKK 438m (DKK 242m), and net profit increased by 43% in local currencies to DKK 1,197m (DKK 815m).
Figures for 2024 are shown in brackets
Falck delivers preventive, acute and rehabilitative healthcare services to supplement and support established healthcare systems. The Lundbeck Foundation owns 58% of Falck.
In a year characterised by a high level of uncertainty in all markets, Falck once again showed resilience in its operations and made progress on strategic priorities. However, financial results were less satisfactory. As a result, Falck initiated a restructuring to reduce complexity and increase efficiency and execution power.
In Europe, results were positively impacted by significant contract wins in the ambulance business in Denmark and Germany and higher demand for healthcare services in Scandinavia. Industrial Fire services also contributed positively with new contracts and improved profitability, whereas Falck’s travel and security assistance was negatively impacted by changing travel patterns during the second half of the year.
In the Americas, results in the U.S. were negatively impacted by lower revenue collection from ambulance trips compared to last year's extraordinary performance, salary increases resulting from new collective bargaining agreements and exchange rate headwinds. In Latin America, the decision by a major customer in Q3 2024 to insource services had a sustained negative impact during 2025. However, this was mitigated by increased
sales efforts, leading to the total number of subscriptions in Colombia surpassing 500,000.
In Q4, Falck consolidated the organisation from seven business units into four regions, based on its geographical strongholds: Scandinavia, Europe, the U.S. and Latin America. The purpose was to reduce complexity across service areas within these regions and make the organisation simpler, more efficient and more fit for purpose. The composition of the Executive Management Team changed accordingly.
Towards the end of the year, Falck also implemented changes at other levels of the organisation, aimed at further increasing efficiency and reducing costs. As a result, Falck said goodbye to a number of employees, primarily in central and administrative functions.
The organisational changes will not impact service levels for Falck’s customers.
Falck’s corporate strategy, Care for More ’27, remains unchanged and serves as the foundation upon which the company is evolving into a leading, global healthcare provider. The aim is to expand within four geographical strongholds, primarily through organic growth supported by carefully selected acquisitions, while growing Falck’s profitability.
In 2025, Falck made significant progress. In Europe, Falck successfully renewed major ambulance service
contracts in Denmark and Germany and began preparations for the new contract in Catalonia, Spain, which is set to launch in early 2026. In the U.S. Falck secured extensions for the ambulance service contracts in both Colorado and Alameda, California. LATAM operations saw significant subscriber growth, and in Scandinavia, Falck expanded its B2C and B2B businesses. In addition, Falck piloted multiple new healthcare services across markets.
Investments in digitalisation over the past few years have created a solid foundation for more streamlined and efficient back-office operations. Falck will now focus on reaping the benefits and lower the overall level of IT investments.

Revenue (DKKm)
12,495
Operating profit (DKKm)
333
Net profit (DKKm)
195
Number of employees (FTEs)
19,371
Financial performance
Sales
In 2025, revenue increased to DKK 12,495m (DKK 12,134m), corresponding to reported growth of 3.0%, primarily driven by strong performance in Europe. The growth in Europe stemmed from a combination of price increases, significant contract wins in the ambulance business and higher demand for both healthcare and Industrial Fire services. The Americas also contributed to organic growth, driven by price increases in the U.S. and by a combination of higher subscription levels and price increases in LATAM. Foreign exchange effects reduced revenue growth by 1.1 percentage points
Operating profit
Cost of services increased to DKK 9,535m (DKK 9,268m). The increase was primarily driven by salary inflation in the U.S. due to the new Collective Bargaining Agreement (CBA) and higher activity in general. This was partly offset by significant operational efficiencies in Europe. Overall, the OPEX ratio decreased by 0.1 percentage points to 75.9%.
Sales and administrative expenses increased to DKK 2,515m (DKK 2,404m). The increase was driven by salary increases in the U.S. and strategic investments in sales efforts in LATAM. These increases were partly offset by efficiency initiatives in Europe. In 2025, investments in digitalisation amounted to DKK 153m (DKK 213m), of which DKK 99m related to digital foundation work and DKK 54m to customer-oriented digital capabilities. Overall, the SG&A ratio increased to 20.1% (19.8%).
Operating profit decreased by DKK 127m to DKK 333m (DKK 460m), corresponding to an operating profit margin of 2.7% (3.7%).
Net profit
Net profit was DKK 195m (DKK 116m). Net financial expenses and income tax expenses decreased compared to last year, more than offsetting the lower operating profit.
The Lundbeck Foundation holds minority stakes in Ellab, Ferrosan Medical Devices and WS Audiology, three international leaders in their fields with exposure to global and local market dynamics.
Ellab
Ellab is the market-leading player within high-end validation, monitoring and calibration solutions, including field services primarily to life science customers where accurate and complete documentation is essential.
Ellab serves both small and large companies, including the top 40 pharma and top 20 biotech companies in the world. The company designs, develops and manufactures high-precision equipment and software for temperature, pressure and humidity validation and monitoring.
Ellab has solutions for applications such as sterilisation, freeze drying, environmental chamber testing, depyrogenation, warehouse mapping, pasteurisation and many more. The product portfolio comprises a comprehensive range of wireless data loggers, thermocouple systems, wireless environmental monitoring systems, calibration equipment, software solutions and accessories.
Ellab was established in Denmark in 1949. Today, Ellab is headquartered in Hillerød, Denmark, and has a worldwide presence, with R&D and production in Denmark, 24 sales and service centres and over 50 distributors around the globe. Ellab has close to 800 employees, of which approximately 240 are in Denmark.
The Foundation owns 34% of Ellab in a partnership with Novo Holding.
Ferrosan Medical Devices (FeMD) is an international medical device company that develops and manufactures medical devices used in surgical care. FeMD’s biomaterial devices are gelatine-based adjunctive haemostatic agents used by clinical professionals in the operation room to control intraoperative bleeding in a fast and effective manner, allowing surgeons to carry out surgery.

The portfolio of haemostatic devices includes three formulations, flowable matrices, sponges and powder. The devices are sold under the trademarks SURGIFLO™ , SPONGOSTAN™ and SURGIFOAM™, and are all marketed and distributed in more than 100 countries through FeMD’s partnership with J&J Medtech (previously Ethicon, Inc., part of Johnson & Johnson).
In addition, FeMD develops and produces handheld biopsy devices used by physicians for breast cancer diagnostics. These are developed and distributed in collaboration with a global partner.
Today, FeMD manufactures the second-generation biopsy device at its manufacturing site in Poland
Since 1947, FeMD has developed and manufactured surgical haemostatic devices for use by healthcare professionals. Today, FeMD has approximately 550 employees; 400 employees at the headquarters in Søborg, Denmark, and 150 employees in Szczecin, Poland.
WS Audiology
WS Audiology (WSA) is a global leader in the hearing aid industry. The company was created through a merger between the family-owned Widex and EQT-owned Sivantos in 2019 and was until April 2024 owned by T&W Medical (51%) and EQT (49%). T&W Medical is owned by the founding families of Widex, Tøpholm and Westermann. In April 2024, T&W Medical entered an agreement with the Foundation, whereafter T&W Medical’s 51% stake in WSA was transferred to a company – CN8 – in which T&W Medical owns 85% and the Foundation owns 15%. The Foundation has through CN8 an indirect ownership of 8% in WSA.
Widex dates back to 1956, and Sivantos dates back to 1878 and was originally part of Siemens
WSA has its global headquarters in Lynge, Denmark, and has 7 production sites around the world, R&D hubs in Denmark, Germany, Singapore and India and global sales from 45 offices. WSA operates under several brands, Widex and Signia being the most well-known.
The mission of WSA - Wonderful Sound for All - is to address the growing challenge of hearing loss. With industry-leading innovation, a diverse portfolio of solutions and a strong global presence, WSA is very well positioned to help more people than any other hearing aid manufacturer.
WSA currently produces around 1/3 of all hearing aids in the world and has around 13,000 employees, of which approximately 1,000 are in Denmark.
The Foundation owns WSA in a partnership with T&W Medical and EQT
BioCapital is the Foundation’s evergreen life science venture capital fund. The fund creates and finances biotech companies based on pioneering research with the potential to deliver significant innovation to patients in areas of high unmet medical need.
Lundbeckfonden BioCapital experienced yet another eventful year where many of our portfolio companies made significant progress while some experienced setbacks, reflecting the inherent risks associated with biotech investments. During 2025, we continued to focus on the local Danish biotech ecosystem, and the year saw us close two new seed investments in Denmark.
2025 was also a year, where we accelerated our efforts to engage with the wider ecosystem by publishing three white papers covering topics from work culture & recruitment, best practises in the venture industry as well as capturing the Foundations experience from the first three years of pre-seed funding through the Frontiers grants.
From a global perspective, 2025 provided some relief in the public markets as the second half of the year saw a sustained rebound in share prices after four years of stagnation. Sustained positive public-market sentiment could lead to a reopening of the IPO window, supporting fundraising across both public and private companies.
In 2025, our portfolio companies delivered a succession of positive clinical results that, while not without challenges, all pointed to the future. For BioCapital, 2025 was also a year of resilience and learning, teaching us that drug development is not always a linear process, and that perseverance and longterm vision is a key determinant of success.
In an important year for Vesper Bio, the company reported positive results from a Phase Ib/IIa study in frontotemporal dementia (FTD). The study confirmed that the company’s lead product, a potentially first oral therapy for FTD, can normalise progranulin levels in the Cerebrospinal Fluid (CSF). Progranulin is a key biomarker, as a subset of FTD is caused by haploinsufficiency (a defect in one copy) of the progranulin gene leading to reduced levels of progranulin. Following
these results, the company closed an investment from The Association for Frontotemporal Degeneration (AFTD) and the Alzheimer’s Drug Discovery Foundation (ADDF). The funds will be used to extend the runway and prepare for a pivotal study. In related news, Vesper’s competitor Alector disclosed disappointing clinical data from a pivotal study in FTD of a sortilin degrading antibody and announced the termination of the programme. Vesper’s clinical candidate VES001 is a reversible sortilin inhibitor which, unlike sortilintargeting antibodies preserves many sortilin functions crucial for neuronal health not mediated by progranulin.
NMD Pharma received positive data from a Phase II study in Charcot-Marie-Tooth disease (CMT), an inherited, progressive and currently untreatable genetic disorder characterised by muscle weakness, atrophy and sensory loss. The results, which were announced in January 2026, showed that patients treated with NMD Pharma’s lead compound demonstrated improvements across multiple pre-specified secondary endpoints compared to placebo control. The results add to previously published positive Phase Ib data in myasthenia gravis, demonstrating the broad applicability of ClC-1 inhibitors across neuromuscular diseases. During the year, the company also executed on Phase II studies in Myasthenia Gravis and Spinal Muscular Atrophy scheduled to complete in 2026.
In 2025, SNIPR Biome initiated a Phase I/II study of SNIPR001 for the prevention of blood stream infections in haematological cancer patients, an area of significant unmet need with no currently approved therapies. In an
eventful year, the company also closed a EUR 35m series B funding round with participation from the Cystic Fibrosis Foundation and the German Federal Agency for Breakthrough Innovation (SPRIN-D) as well as existing investors. The funding will support the development of a CRISPR-Cas therapy specifically targeting airway infections in people with cystic fibrosis (CF) and enable the advancement of interventions designed to eliminate antibiotic resistance genes in humans across various bacterial species.
IO Biotech announced results from a pivotal Phase III trial in advanced melanoma investigating the company’s lead product Cylembio® in combination with KEYTRUDA®. The study demonstrated a clinical benefit but narrowly missed statistical significance on the primary endpoint. Notably, progression-free survival in the so-called PD-L1negative patient subpopulation, who normally respond poorly to KEYTRUDA®, increased with Cylembio®. IO Biotech was advised by the U.S. Food and Drug Administration (FDA) not to submit a Biologics License Application (BLA), and in January 2026 the company announced that it was exploring strategic alternatives.
Kvantify one of our more recent investments, released Qrunch, the first ever chemistry software for quantum computers. The software will allow customers to simulate complex chemistry problems on a quantum computer with unprecedented performance. Prior to launch, the software has been tested in a collaboration with Novonesis, where it successfully modelled enzyme catalysis. The software complements the suite of drug discovery solutions Kvantify offers on traditional
computers. The company also closed a new EUR 5m financing round, funding continued product development and commercial roll out.
Our international portfolio also continued to make progress as several companies reached significant value inflection points generating positive momentum in share prices and in some cases catalysing significant transactions.
scPharmaceuticals continued to see strong uptake following the successful launch of FUROSCIX®, reporting strong growth into the first half of 2025. The commercial success led to a trade sale to MannKind corporation in August 2025. scPharmaceuticals was not the only company in our portfolio that executed a transformative deal in 2025, as the year also saw VarmX sign a strategic collaboration and option agreement with CSL. Under the agreement, VarmX shareholders received USD 117m upfront and further potential milestone payments of up to USD 2.1bn. In addition, CSL will fully fund VarmX’s Phase III trial evaluating VMX-C001 in patients taking FXabased anticoagulants who require urgent surgery.
Enterome built on positive interim data from the ongoing Phase II trial in indolent Non-Hodgkin Lymphoma by raising USD 19m with participation from the Institute for Follicular Lymphoma Innovation (IFLI) as well as from existing investors. The funding will be used to continue the development of the Non-Hodgkin Lymphoma programme. In a separate programme, Enterome presented positive data from a Phase II colorectal cancer study.
Lexeo Trevi, Spero and Enliven all provided positive clinical updates from ongoing trials. During the year, Lexeo announced a positive update from their Phase I/II study of LX2006 in Friedreich Ataxia Cardiomyopathy, reporting clinically significant improvements in cardiac biomarkers as well as functional measures. The positive data was rewarded by the FDA, who granted Breakthrough Therapy Designation to the programme, allowing accelerated clinical development and regulatory review. Separately, Lexeo announced the first clinical data from the LX2020 arrhythmogenic cardiomyopathy programme, demonstrating transgene expression and a 67% reduction in premature ventricular contractions in the first participant evaluated.
Trevi reported positive data from a Phase IIb study in chronic cough in idiopathic pulmonary fibrosis (IPF), confirming earlier Phase IIa data. In a remarkable year for the company, they also presented positive results from a Phase IIa study in patients with refractory chronic cough, significantly expanding the potential market for Haduvio. Meanwhile, in May Spero Therapeutics announced that the pivotal Phase III study of Tebipenem HBr in patients with complicated urinary tract infections had met its end point sooner than expected and therefore been terminated prematurely. The successful study was followed by an NDA submission in December, paving the way for possible launch of the new antibiotic in 2026.
Enliven provided an update on their ongoing Phase I study in Chronic myeloid leukaemia demonstrating
promising and potentially best-in-class efficacy and tolerability data at 24 weeks.
During the year, four of our public companies capitalised on the positive sentiment on Nasdaq to raise additional funding to extend their runway and continue clinical development. Enliven Trevi and Lexeo leveraged positive clinical trial data to raise USD 230m, USD 115m and USD 234m, respectively, and Aura Biosciences closed a USD 75m financing round enabling the company to complete the ongoing studies in choroidal melanoma and non-invasive bladder cancer
There was also disappointing news from the portfolio, as Cytoki discontinued the development of their lead candidate, a lipidated IL-22 peptide, following the completion of a Phase IIa study in obese diabetics. The failure of the programme led to an impairment in the valuation of the company
The net return for the year was DKK -265m, versus DKK 108m in 2024. The negative result mainly reflected an impairment of Cytoki, necessitated by the discontinu-ation of their lead candidate, which was partly offset by an increase in the valuation of several portfolio companies driven by clinical progress and overall positive developments in public markets.
At year-end, the BioCapital portfolio comprised 18 companies, of which four were listed. The fair market value of the portfolio was DKK 2,576m at 31 December 2025, compared to DKK 2,884m at the end of 2024. The
complete portfolio at year-end is listed to the right and on the following page.

Aura Biosciences’ first product candidate, the laseractivated AU-011, is currently being investigated in a Phase III study for the treatment of patients with smallto-medium primary ocular melanoma

Enliven Therapeutics is developing best-in-class small molecule kinase inhibitors to treat cancer. The company currently has two compounds in Phase I

IO Biotech develops disruptive cancer immune therapies, targeting immunosuppressive cells in the local tumour environment.

Trevi Therapeutics is engaged in the late-stage development of Haduvio™ (nalbuphine ER) for the treatment of chronic cough for patients with idiopathic pulmonary fibrosis.

Akamis Bio is a clinical stage company developing genebased, immuno-oncology treatments for solid tumours, using its proprietary, intravenously administered T-SIGn virus platform

Epoqe Pharma is a pre-clinical-stage biopharmaceutical company focused on the development of novel treatments for serious cardiovascular diseases.

Cytoki Pharma is a biotechnology company focused on serious diseases caused by epithelial injury with unmet medical needs. The company’s lead programme is a long- acting interleukin-22 for use in metabolic and inflammatory disease, and other pathologies related to epithelial tissue injury.

Dania Therapeutics is a discovery-stage company developing first-in-class small molecule drugs to treat Acute Myeloid Leukaemia (AML) and other cancers.

Enterome is pioneering the development of novel microbiome-inspired peptide drugs. Enterome is conducting Phase II studies in several treatment lines in lymphoma
Folium Science uses CRISPR technology to eliminate harmful microbes and bacteria, while preserving the beneficial ones, resulting in a healthier animal gut. The technology has applications in the food and feed industries, and the first product is aimed at controlling salmonella in industrial chicken feed

Kvantify is a commercial-stage company developing quantum-ready software for computer-aided drug design (CADD) to accelerate drug discovery.

NMD Pharma develops innovative medical treatments for rare neuromuscular diseases based on world-leading electrophysiology science. The company has three ongoing clinical Phase II programmes.

Notify Therapeutics is a discovery-stage company developing hormone-free small molecule drugs for the treatment of infertility in women
Research, development and commercialisation of pharmaceuticals.

SNIPR Biome is a clinical-stage company developing new therapies based on the CRISPR gene technology. The company’s technology enables gene therapy of the microbiome or precision killing of bacteria. The lead clinical product is designed to prevent bloodstream infections caused by fluoroquinolone-resistant E. coli bacteria.

Sorriso is developing gastrointestinal tract-restricted antibodies to treat inflammatory bowel disease (IBD) and other inflammatory diseases. The lead candidate is in Phase II development.

VarmX is a clinical-stage company developing a novel reversal agent to safely and effectively restore haemostasis in case of bleeding or emergency surgery in patients taking oral factor Xa inhibitors

Vesper Bio is developing new products focused on central nervous system and peripheral diseases. The lead programme, targeting frontotemporal dementia, is currently in Phase II clinical development.
Invest generates returns with the primary purpose of securing sufficient reserves to protect the long-term ownership of the Foundation’s strategic ownerships, maintain philanthropic grant activities and increase the asset base. The financial investments are spread across a diversified investment portfolio.
2025 was another year marked by heightened geopolitical tensions, political uncertainty and continued market volatility.
The start of the year was positive for risk assets, but global trade policy tensions in April prompted both a significant stock market sell-off and substantial USD depreciation. Financial markets recovered, however, and global equity and credit markets advanced approximately 8% and 1% in 2025, respectively, measured in DKK, supported by falling central bank interest rates and reduced trade tensions that underpinned risk assets. The USD-DKK did not recover and dropped 12% in 2025. A traditional 60/40 (equity/bond) portfolio hence produced a gain of approximately 6% over the year (2024: 14%) measured in DKK. Against this backdrop, Invest generated a very satisfactory return in 2025 of 12%.
Global equity markets again delivered attractive gains, but the dominance of U.S. stocks and particularly a handful of U.S. mega-cap technology stocks (“Mag-7”) moderated. U.S. equities rose at a healthy pace, driven by resilient labour markets, firm household consumption and an ongoing, though gradual, easing of monetary policy. The annual total return of the S&P 500 ended at 17.9% in USD but only 4.1% when measured in DKK.
In Europe, the STOXX Europe 600 index increased by 20.8% in DKK and performed comparatively better than in 2024 and markedly better than global stock indices measured in DKK. The improvement was supported by declining inflation, a series of rate cuts by the European Central Bank and tentative signs of stabilisation in business activity. By year-end 2025, investors await for the deployment of infrastructure and defence funds to fuel industrial spending in the years ahead.
Consumers in the advanced economies held up relatively well. The U.S. labour market stayed tight for most of the year, and unemployment in Europe remained low by historical standards. Lower interest rates, accumulated savings and still solid wage growth helped offset the drag from higher prices and maintained support for private consumption, albeit being under pressure in some areas.
China, the world’s second largest economy, remained a source of concern in 2025. The country continued to wrestle with a slow moving housing correction, elevated leverage and subdued consumer confidence. These
headwinds weighed on Chinese equities, but despite that, MSCI China gained 16.1% in DKK terms.
Strong 12% return in 2025 reflected disciplined execution, high quality equity holdings, and resilient diversification –underscoring the strength of long-term investment strategy and active risk management.
Invest generated a solid positive return in 2025 of 12%, primarily driven by listed equities, with additional support from credit and the private equity portfolio. All asset classes contributed positively.
The return was primarily driven by listed equities delivering 20.3% in DKK terms, significantly outperforming the 8.8% return on global equity markets. Industrial, financial and utility sector investments contributed the largest positive returns in absolute terms, while selected consumer related holdings detracted from performance.
The investment in FLSmidth yielded a total return of 28%. FLSmidth completed a successful transformation and now stands as a pure-play supplier of technology and services to the mining industry
The bond portfolio generated a return of 1.9% versus the benchmark return of 1.2%. Invest had a cautious view on bonds and thus a low duration on the bond portfolio. Duration was further reduced in 2025.
The credit portfolio delivered a return of 5.5%, which was better than the benchmark portfolio yield of 4.6%. The corporate high yield portfolio generated the highest return, but overall returns were broad based. The externally managed private debt and multi-asset credit portfolio delivered satisfactory returns.
Private equity generated a return of 6.6%, which was 22%-points below the comparison benchmark portfolio
Real assets, including real estate and woodland, generated a positive return of 3.4%, which was 0.9%points above its benchmark.
Invest generates returns with the primary purpose of (a) securing sufficient reserves to protect the long-term future of the Foundation’s strategic ownerships; (b) increasing philanthropic grant activities; and (c) increasing the overall asset base.
Invest manages DKK 26bn, which is allocated to listed equities (56%), credit (9%), bonds and liquid funds (17%), private equity (12%) and real assets (6%). Illiquid assets represented 21% of the portfolio by year-end 2025
The majority (60%) of the assets are managed internally by the investment team. Listed equities are primarily managed internally, credit is managed equally by inhouse and external teams whereas real asset investments are mainly invested via partnerships (ObelLFI Ejendomme A/S and LFI Silva Investments A/S), while private equity is fully outsourced.
The investment philosophy is centred around a longterm fundamental investment focus with a balanced approach to risk, quality and valuation. All investments undergo a thorough due diligence process, and investments are selected based on absolute return potential and risk assessment.
Invest prefers quality companies whose revenues undergo secular growth driven by forces that will likely be in place for an extended period. Examples include energy transition, IT technology and healthcare. Invest also seeks investments in cyclical quality companies with deep intrinsic value.
Invest entered 2025 with a pro-risk portfolio view but with an expectation of persistently elevated market volatility due to the political and geopolitical landscape. Invest is a fundamental long-term investor, which enables it to exploit market volatility and short-term mispricing. However, new investments are only undertaken following a deep quality and margin of safety assessment including a multi-layered risk management framework. During the year, Invest added
quality exposure within cyclical segments of the equity market, especially within the industrials sector to increase exposure to the secular electrification theme.
Invest sustained its strong long-term performance in 2025, delivering annual total returns of 13.0% and 10.9% over the past three and five years, respectively – significantly outperforming the benchmark, which returned 10.7% and 6.2% over the same periods. Over five years, Invest’s cumulative return amounts to 67.5%, compared with 35.1% for the benchmark. Listed equities have been the main driver of this performance, achieving annual returns of 15.1% over the past five years.
The long-term returns endorse the investment philosophy of investing in high quality companies across different sectors with a goal of generating attractive long-term return without compromising strong nearterm risk management.
Invest manages both short- and long-term investment risks, ensuring adequate reserves to protect the longterm future of the Foundation's strategic ownerships.
The main risk in relation to long-term returns is the investment team's ability to select the right investments and external managers. Invest has developed a structured process and a solid fundamental due diligence investment framework for each asset class, with the key aim of opting out of weak investments and understanding the risks in each investment.
Portfolio construction considers the quality of the investments, expected return potential, risk assessment and overall knowledge level of the sector and company. The portfolio is well diversified.
Invest has continuously strengthened its risk management, primarily by improving the quality of the listed stock portfolio through enhanced investment processes and the incorporation of derivatives for hedging. Recently, Invest adjusted its investment strategy and process for credit and private equity.
Short-term risks mainly relate to external risks such as geopolitics and macro risk. Invest manages these risks ad hoc and seeks to take advantage of excessive market movements based only on a fundamental long-term view. During the year, Invest further strengthened its portfolio construction flexibility to capitalise on market volatility
In 2025, the Lundbeck Foundation Group delivered record revenue and operating profit.
Exceeding the Foundation’s forecast of Group revenue between DKK 42bn and DKK 43bn, the Group delivered revenue of DKK 43,437m, largely attributable to Lundbeck, which delivered record revenue of DKK 24,630m.
Group operating profit, at DKK 7,046m (DKK 4,569m), also exceeded expectations, which were between DKK 6.2bn and DKK 6.6bn.
Group profit for the year, at DKK 6,111m (DKK 6,567m), was mainly impacted adversely by financial expenses in subsidiaries, share of profit of associates and income tax.
Grants totalled DKK 825m (DKK 1,074m)
Operating activities
Reported revenue for the year increased to DKK 43,437m (DKK 39,675m), primarily driven by Lundbeck and ALK, which delivered record revenue of DKK 24,630m (+12%) and DKK 6,312m (+14%), respectively. Revenue at Falck increased by DKK 361m, corresponding to an increase of 3% on 2024.
Gross profit was DKK 27,559m, versus DKK 24,192m in 2024. The increase was primarily attributable to
Lundbeck, where gross profit increased by DKK 2,591m.
Gross profit at ALK and Falck increased by DKK 682m and DKK 94m, respectively.
The Group’s gross margin was 63%, up from 61% in 2024.
Revenue
+9%
DKK 43,437m (DKK 39,675m)
Reported revenue at ALK and Lundbeck increased by 14% and 12%, respectively, on 2024
Research and development (R&D) costs increased to DKK 5,543m (DKK 5,102m), corresponding to an increase of DKK 441m. In 2024, R&D costs included Lundbeck’s impairment loss of DKK 547m on part of the carrying amount of one of the MAGLi projects following a negative data read out.
Overall sales and distribution costs decreased to DKK 9,736m (DKK 10,087m), driven by lower costs at Lundbeck of DKK 403m primarily reflecting leverage from the strong revenue growth and improved cost efficiency. Overall sales and distribution costs represented 25% of total revenue in 2025 (25%).
Overall administrative expenses amounted to DKK 4,146m (DKK 4,006m), with the increase largely attributable to Falck and Lundbeck.
Group operating profit
+54%
DKK 7,046m (DKK 4,569m)
The increase in Group operating profit was primarily attributable to Lundbeck (+DKK 2,005m) and ALK (+DKK 563m).
Net wealth
+13%
DKK 80,709m (DKK 71,739m)
This was primarily attributable to an increase in ALK’s and Lundbeck’s share price and the return of the investment portfolio in Invest.
The Foundation’s own net administration and operating costs amounted to DKK 162m versus DKK 166m in 2024.
Total operating costs thus amounted to DKK 19,425m versus DKK 19,195m in 2024.
Other operating items, net, amounted to an expense of DKK 1,088m (expense of DKK 428m), primarily reflecting an impairment loss of DKK 639m as part of the planned divestment of Lundbeck Pharmaceuticals Italy S.p.A., and restructuring and redundancy cost in Lundbeck and Falck totalling DKK 523m. In 2024, other operating items, net, included transaction and integration costs of DKK 420m related to Lundbeck’s acquisition of Longboard.
The Foundation’s investment activities generated a profit of DKK 1,783m compared to a profit of DKK 3,006m in 2024.
The investment portfolio in Invest generated a profit of DKK 2,905m versus a profit of DKK 3,108m in 2024. In both years, the return was primarily driven by listed equities.
Meanwhile, the Foundation’s BioCapital portfolio delivered a net loss totalling DKK 265m (net profit of DKK 108m). The negative result mainly reflected an impairment of Cytoki, necessitated by the discontinuation of their lead candidate, which was partly offset by an increase in the valuation of several portfolio companies driven by clinical progress and overall positive developments in public markets.
The Group’s profit share of the strategic minority ownerships (associates) amounted to a loss of DKK 868m (loss of DKK 210m), of which DKK 757m related to impairment of goodwill.
Group operating profit increased to DKK 7,046m (DKK 4,569m). The improvement in profitability was primarily attributable to Lundbeck and ALK, which saw increases of DKK 2,005m and DKK 563m, respectively.
Tax on profit for the year amounted to DKK 1,754m (DKK 1,212m), corresponding to an increase of DKK 542m.
The effective tax rate of 22% (16%) is on par with the Danish tax rate, although it is affected by several opposing items, including deductible grants recognised directly in equity, changes in the valuation of net tax assets and prior‑year tax adjustments. In 2024, the
effective tax rate of 16% was primarily impacted by Lundbeck’s reversal of an uncertain tax provision related to a closed tax audit and by deductible grants recognised directly in equity.
The net effective tax rate of the three strategic subsidiaries was 27% (19%) impacted by changes in the valuation of net tax assets and prior year tax adjustments.
In 2025, overall profit for the Group was DKK 6,111m (DKK 6,567m). The Foundation’s share of Group profit was DKK 4,335m (DKK 5,068m).
The Foundation awarded a total of DKK 825m (DKK 1,074m) in grants in 2025.
Total assets at 31 December 2025 amounted to DKK 102,587m versus DKK 106,111m at the end of 2024.
Intangible assets amounted to DKK 42,204m at 31 December 2025 (DKK 47,495m). Product rights accounted for DKK 28,158m (DKK 32,379m), mainly driven by the negative impact from translation of foreign currencies of DKK 2,927m. Amortisation of product rights amounted to DKK 1,294m (amortisation of DKK 1,433m and impairment of DKK 547m). Goodwill at year-end amounted to DKK 11,448m (DKK 12,298m), mainly driven by the impact from translation of foreign currencies.
The Foundation’s financial assets at 31 December 2025 amounted to DKK 27,231m (DKK 25,290m), equivalent to an increase of DKK 1,941m compared to year-end 2024.
Cash and bank balances at 31 December 2025 amounted to DKK 5,563m (DKK 5,907m)
Equity and liabilities
Total Group equity at 31 December 2025 amounted to DKK 63,505m (DKK 61,043m), reflecting an increase of DKK 2,462m in 2025. The Foundation’s share of equity increased to DKK 52,053m (DKK 50,280m).
The Lundbeck Foundation’s share of equity and debt to financial institutions, etc.
At 31 December 2025, total debt to financial institutions etc., including lease liabilities, decreased to DKK 15,794m (DKK 21,635m). Consequently, net interest-bearing debt, excluding the Foundation’s financial assets, amounted to DKK 10,231m (DKK 15,728m).
The strategic ownerships’ contributions to net wealth differed from the contribution recognised and measured in accordance with the accounting policies of the Lundbeck Foundation Group. The fair value of the Foundation’s net assets is based on market prices, where available - for ALK and Lundbeck - and, for other companies, an estimated fair value based on a trading-multiple model.
Total cash flow from operating activities amounted to DKK 8,312m versus DKK 5,598m in 2024. The cash flow was affected by financial items and income tax by a total outflow of DKK 1,589m (inflow of DKK 170m). In 2024, cash flow was negatively impacted by the settlement of liabilities related to Longboard's longterm incentive programme and transaction costs associated with the acquisition.
Total cash flow from investment activities amounted to an outflow of DKK 1,254m (outflow of DKK 17,263m).
Excluding the purchase and sale of financial assets, cash flow from investment activities was an outflow of DKK 1,389m (outflow of DKK 17,983m), of which DKK 12m (DKK 15,862m) relates to acquisition of businesses.
In 2025, the total cash flow from grants paid and dividends paid to non-controlling interests amounted to an outflow of DKK 1,039m (outflow of DKK 845m).
Furthermore, cash flow from financing activities included a net outflow of DKK 6,229m regarding loan repayments (net inflow of DKK 12,414m). In 2024, cash flow was significantly affected by Lundbeck’s debt funding related to the acquisition of Longboard.
Net cash flow in 2025 amounted to an outflow of DKK 256m (outflow of DKK 174m). At the end of 2025, the Group’s cash balance totalled DKK 5,563m versus DKK 5,907m at the end of 2024
At 31 December 2025, the Group’s net wealth amounted to DKK 80,709m (DKK 71,739m). Net wealth was significantly affected by the increase in ALK’s and Lundbeck’s share price and the return of the investment portfolio in Invest, which had positive effects of DKK 6,238m, DKK 2,070m and DKK 2,857m, respectively.
The financial performance of the Group depends on developments in the commercial activities of Lundbeck, ALK and Falck, as well as returns generated by the Foundation’s investment activities, including strategic ownerships (Ellab, Ferrosan Medical Devices and WS Audiology) and the biotech portfolio. Returns provided by investment activities largely depend on the overall performance of the financial markets, whereas returns from the biotech portfolio also depend on the development of products and similar factors at the portfolio companies
For 2026, Group revenue is expected to reach between DKK 44.6bn and DKK 45.7bn. Group operating profit is expected to be between DKK 7.6bn and DKK 8.1bn. The expected increase in Group revenue and operating profit compared to 2025 is related to expected higher revenue and improvements at all subsidiaries.
These expectations are based on the exchange rates prevailing at the end of January 2026.
Please refer to the respective annual reports of Lundbeck, ALK and Falck for further details about the companies’ individual expectations.
Lundbeck Foundation (parent entity)
The Foundation’s profit for the year amounted to DKK 619m versus DKK 2,822m in 2024. The 2024 result was positively affected by dividends from Lundbeckfond Invest A/S amounting to DKK 1,545m. Net financial profit (excluding dividends) amounted to DKK 713m (profit of DKK 1,451m).
In total, grants awarded in 2025 amounted to DKK 825m versus DKK 1,074m in 2024. Net grants amounted to DKK 805m (DKK 1,060m) in 2025, as grants of DKK 20m (DKK 14m) were reversed or repaid during the year. Subsequently, equity at 31 December 2025 amounted to DKK 12,288m (DKK 12,474m).
Through its purpose and the 2030 strategy, ‘Bringing Discoveries to Lives’, the Foundation has articulated a clear commitment to making a positive societal impact. To achieve its purpose and execute its strategy, the Foundation engages in a range of activities that carry their own sustainability impacts, risks and opportunities, and sustainability is therefore managed throughout its business model and activities.
The Foundation strives to improve lives through healthcare innovation, with a specific focus on neuroscience. Over the years, it has grown to become one of Denmark’s largest enterprise foundations, managing close to DKK 81bn. Its commitment to sustainable development is rooted in its founding principles, strategy and business model. Sustainability is viewed both as a responsibility and a license to operate – now and in the future – as well as an opportunity to drive long-term value creation.
At the core of the Foundation’s business model is the principle that its ownerships and investments generate dividends and profits that are reinvested into society. In 2025, the Foundation awarded DKK 825m in grants and contributed DKK 95m in investments to the Danish life science ecosystem. As a Group, the Foundation employed close to 34,000 people in 2025, reached more than 40 million patients, and invested more than DKK 5.5bn in research and development. As a long-term owner, the Foundation aims to support sustainable business development and initiatives.
Research indicates that companies with enterprise foundation ownership achieve higher environmental, social and governance (ESG) performance, maintain ESG activities even during financial crises, and commit to more significant emission reductions in the post-Paris Agreement period.
In 2024, the Foundation conducted a double materiality assessment (DMA) based on its business model as both an investor and a long-term owner. This assessment enabled the Foundation to identify key impacts, risks and opportunities from a sustainability perspective. These insights were used to further integrate sustainability into our operations during 2025.

The purpose, business model and 2030 strategy of the Foundation provide a strong platform and clear direction for its societal impact, focusing on health, innovation, economic growth and good governance.
The Foundation’s role as a societal partner presents both unique opportunities and significant responsibilities. Its societal impact, risk and opportunities are evident throughout its diverse activities.
The Foundation is the engaged owner of six strategic ownerships with just under 48,000 employee, investing close to DKK 7bn in research & development and directly reaching approximately 44 million patients globally through medicine, products and services.
The strategic ownerships entail a responsibility to foster sustainable economic growth, create jobs and be a responsible employer, ensuring a healthy and inclusive working environment. The Foundation remains committed to supporting long-term development and helping its companies achieve global leadership in their fields while emphasising innovation and social aspects such as talent development, employee engagement, pay equality and diversity
The Foundation’s Double Materiality Assessment identified the following material sustainability matters, pertaining to the role as long-term owner and investor:
Advancing health and especially brain health is a key strategic pillar for the Foundation, through funding of research, biotech investments, long-term ownerships of healthcare companies and support for collaboration in the research ecosystem.
Sound working conditions and inclusive corporate cultures with focus on employee engagement and development are crucial for retaining and attracting talent for the Foundation and our portfolio companies.
As a long-term investor and asset owner our capital can be attributed towards the release of greenhouse gas emissions in our portfolio. Climate change poses long-term risks to health, society and economic stability, including the sustainability of healthcare systems.
Business ethics and good governance are central to the Foundation’s governance practices with a strong focus on fostering ethical decision making across portfolio companies, as well as in investment and grant-giving processes.
Sustainability is a cornerstone of the Foundation’s ownership model. While decisions are ultimately made by the companies’ boards and leadership teams, the Foundation actively encourages the integration of sustainability measures and initiatives, as reflected in their respective reporting. Through its board representation, the Foundation promotes sustainability agendas and ensures compliance with its sustainability policy’s minimum standards.
All six strategic ownerships have greenhouse gas reduction targets that are approved by the Science Based Targets initiative. This framework provides a defined path for reducing emissions. The Foundation also leads initiatives to facilitate knowledge sharing and promote sustainable practices across its ownerships, including efforts to improve access to healthcare
Through new investments and its existing portfolio of 18 international and Danish life science companies, the Foundation supports innovation in healthcare by investing in people, businesses and science at the forefront of their fields. Supporting the growth of Danish life sciences and investing in the future of healthcare are central to the Foundation’s biotech investments.
The Foundation identifies companies and teams with strong commercial potential that address unmet medical needs through novel and/or superior treatments. By taking a proactive approach that combines guidance, policies, advisory support and
resource allocation, the Foundation works to embed sustainable practices into the early stages of these companies’ commercial development.
In 2025, the Foundation continued to develop and implement its Sustainability Programme. This involved a contribution to the Life Sciences VC Sustainability initiative (LSVC). LSVC is a peer-led collaboration between European life science venture funds focused on concrete practical steps to strengthen the early-stage life science ecosystem in Europe.
The Foundation’s financial investments ensure longterm financial stability and robustness and allow the Foundation to invest in the future of healthcare and support new scientific research through grants and partnerships.
The Foundation’s investment policy incorporates environmental, social and governance (ESG) factors into asset management and due diligence processes. All financial investments must comply with ESG principles. The listed equity portfolio undergoes a biannual screening to ensure alignment with these standards
Partner to Society: Supporting and disseminating new knowledge
Brain diseases affect over 3 billion people worldwide, causing significant personal and socio-economic challenges. Despite these impacts, brain health remains an under-prioritised research area. Addressing this is at
the core of the Foundation’s purpose and grant activities.
In its grant activities, the Foundation is committed to integrity, transparency and accountability. The Foundation aims to support world-class scientific research that drives positive outcomes and advances the frontiers of knowledge. Best practices – such as the Danish Code of Conduct for Research Integrity – and environmental, social and governance (ESG) considerations are integrated into the Foundation’s grant processes. The Foundation also integrates impact measurement into its grant requirements to enhance the translation of research into tangible patient outcomes.
Applications are handled and evaluated through a rigorous assessment process, which includes peer reviews conducted by international third-party expert panels. These panels operate in compliance with the Foundation’s principles of impartiality, ensuring a fair and transparent evaluation.
The Foundation actively works to build partnerships and use its public voice to support its mission of driving positive societal change. The Foundation’s public voice is planned and conducted with strong attention to principles of transparency, accountability and integrity, and is shared publicly on the Foundation’s website: https://lundbeckfonden.com/about-us/socialresponsibility-and-transparency/active-public-voice.
Good governance is central to integrating sustainability considerations into day-to-day business decisions and strategy. As owner, investor and philanthropist, the Foundation has many opportunities and a responsibility to promote sustainable practices both inside and outside its organisation. The Foundation does this with a clear governance model based on four key principles:
• Compliance with Danish’s Recommendations on Foundation Governance
• Board director independence
• Transparency in reporting and grant decisions
• Internal checks and balances in decision-making
The Foundation has implemented a clear sustainability governance framework that is anchored in the Board of Directors with the CEO having overall responsibility and clear responsibilities distributed throughout the organisation.
Moreover, the Foundation has developed policies to guide key activities: grant administration, investments, communication, tax, remuneration and sustainability.
The sustainability policy applies to the Foundation, grant recipients, investments and ownerships, encouraging the strategic ownerships and direct investments to pursue ambitious sustainability agendas. The goal is for the Foundation to fulfil a role in business and society in a way that is environmentally, socially and financially sustainable.
All policies can be found on the Foundation’s website. The Foundation has also implemented a whistleblower system, which can be used by external and internal parties in case of concerns involving legal or other serious risks. The whistleblower system can also be accessed via the Foundation’s website. In 2025, one report was made via the whistleblower system regarding a portfolio company. The case has been closed
Learn more about sustainability at the Lundbeck Foundation Group
For more information on the Lundbeck Foundation’s impact as well as important developments in 2025, please refer to the Foundation’s Sustainability Report 2025
(https://www.lundbeckfonden.com/en/sustainability).
This report is the Lundbeck Foundation’s statutory statement on sustainability in accordance with sections 99b and 99d of the Danish Financial Statements Act.
For information about Lundbeck, ALK and Falck’s approaches to sustainability, please visit their websites.
Key ESG figures for the Foundation and initiatives
Gender diversity
In 2025, the Foundation’s Board of Directors comprised eight board members elected according to the statutes. Seven of the eight board members were considered independent. The eight board members counted two women and six men, excluding the four employee representatives who represent the Foundation’s three subsidiaries.
The gender balance does not meet the guidelines on gender equality issued by the Danish Business Authority (‘Guidelines on target figures, policies and reporting on the gender composition of management’), and addressing this imbalance on the Board of Directors is therefore a priority going forward.
When electing new members to the Board of Directors, the board strives to ensure diversity in competencies as well as gender. The board will continue to pursue equal representation in the coming elections.
At 31 December 2025, the Management Team of the Foundation comprised seven members, including the CEO, and counts two women and five men. The Foundation’s employee group counts 27 women out of a total of 57 Diversity and inclusion will remain focus areas for the Foundation in the years to come.
While the Foundation’s primary purpose is advancing health, it recognises that its activities contribute to the release of greenhouse gas. The Foundation’s climate impact is primarily driven by scope 3 activities regarding the investment portfolio. With regards to our investment portfolio, the reporting covers most of our assets. Please refer to the sustainability report for more details, including accounting policies and methodologies.
In 2025, emissions from international business travel totalled 358 tonnes of CO2. To offset these emissions, the Foundation purchases high-quality carbon credits.
1) These key ESG figures are based on recommendations by the Chartered Financial Analysts’ (CFA) Society Denmark, the Association of Danish Auditors (FSR) and Nasdaq Copenhagen. They represent a first step in a process that, over the coming years, will enable the Foundation to provide stakeholders with further insights into the Foundation’s ESG profile.
2) Comparative figures for business travel and investments are not presented. The full greenhouse gas inventory, presented from 2025, covers significant categories of own operations and supply chain, as well as the investment portfolio, excluding government and mortgage bonds and real assets
3) The ESG metrics have been restated from previous years due to an updated methodology; Scope 1 to include company owned vehicles, energy consumption to use MwH as opposed to GJ, and renewable energy share to reflect the energy mix from purchased electricity and district heating.
For information on accounting policies and methodologies applied to ESG-related figures, reference is made to the Sustainability Report.
The Lundbeck Foundation’s risk management framework provides a systematic approach to identifying, monitoring, managing and reporting risks and opportunities in a changing environment.
The Lundbeck Foundation strives for a reasonable balance between value creation and risk exposure in its work to deliver on its longer-term strategic targets.
Risk assessment is an important part of the Foundation’s business procedures, allowing it to respond appropriately to changing circumstances and to integrate risk management into the development of the Foundation’s strategy. The risk management framework includes a ‘top-down’ as well as a ‘bottom-up’ approach to risk mapping, which identifies key risks that the Foundation faces.
The most important risks relate to the business risks of the Foundation’s strategic ownerships – particularly its subsidiaries – as well as the financial risks related to its portfolio of other investments. Assessing and mitigating these risks is important for long-term value creation, as well as for the ability of the Foundation and its subsidiaries to execute on the strategy.
As a foundation with significant positions in the Danish business and research communities, reputational risk is
an important factor. In relation to grant-making, there is a strong governance framework in place to cover these activities, setting high standards designed to protect the Foundation from excessive risk
As a business owner and investor, risks are a natural and integral part of the Foundation’s activities. However, by incorporating risk management at both entity and group level into the risk assessment framework, and by balancing its capital allocation, the Foundation mitigates risk to what it considers to be an acceptable level.
The Foundation is faced with several types of risks, including business, operational, cyber and market risks –which cover fluctuations in interest rates, share prices, exchange rates and credit spreads – as well as reputational risks. Based on an understanding of its ownerships and of its own internal operations, other investments and grant activities, the Foundation aggregates the various risk factors and identifies the most important ones in terms of probability and potential impact.
A risk analysis report – including proposed mitigating actions – is prepared and submitted biannually to the Foundation’s Board of Directors. More frequently, updates on exposure to risk factors, such as industry and geographical concentrations, are submitted to the Investment Committee, which meets on a quarterly basis. The Foundation’s Management Team monitors
the development of current and potential risks on an ongoing basis.
The overall risk level is assessed in the risk reports. The Foundation has a long-term horizon for its activities, and the current strategy extends through 2030. Similarly, the Foundation takes a long-term view of potential risks and risk management, spanning at least 10 years.
Diversification across the Foundation’s different investments and activities is a central part of the Foundation’s approach to risk management, and this is adjusted continuously, depending on the risk assessment, to ensure that there are sufficient financial resources to support the Foundation’s activities.
The risk assessment exercise has resulted in the identification of a range of strategic, operational, financial, political and reputational risks that could potentially pose a threat to the Foundation. A key priority for the subsequent work has been to ensure that no single risk can materially damage the Foundation, and that the Foundation would be able to continue its operations and strategy should any one or more of the risk factors materialise. To ensure this is the case, each of the risks identified has an assigned owner who is responsible for actively managing and mitigating the risk.
The key risks that have been identified are presented in the table on pages 43-44 together with details of the mitigation measures that have been put in place. The
key risks relate to business risks in the strategic ownerships, investment risks across the whole portfolio, compliance, IT and legal risks, and grant and reputational risks.
Risks related to the strategic ownerships
Business and financial risks associated with the operation and performance of the Foundation’s six strategic ownerships, Lundbeck, ALK, Falck, Ellab, Ferrosan Medical Devices and WS Audiology, are most effectively managed within each business. Consequently, the boards of directors and management teams at each individual company define their own enterprise risk management.
The Foundation is represented on the board of each strategic ownership and monitors the business performance of the companies closely. Descriptions of each company’s approach to risk management are given by each of the companies in their own annual reports
Risks related to the Foundation’s other investments
The Board of Directors defines the Foundation’s investment policy, while compliance with the policy is monitored by the Investment Committee.
The Foundation manages the market risk of its investments by having limits for its exposure to individual asset classes and their underlying assets that also take into account the sectoral and geographic exposure related to the strategic ownerships. This policy covers the entire portfolio, including investments by BioCapital.
To manage interest rate risk, limits for the duration of bond investments are defined. Derivative financial instruments, such as swaps, options and forward contracts, are used for risk management purposes and as an alternative to buying the underlying assets.
The investment policy governs the use of such instruments regarding maturity, quantity and counterparty requirements.
Weekly portfolio performance reports are prepared for the CEO.
Finally, all investments must comply with the Foundation’s environmental, social and governance (ESG) principles. The portfolio investments are monitored for issues related to ESG principles. An ESG status report is reviewed and approved by the CEO and the Investment Committee twice a year. In 2025, no investments were identified as being in violation of the ESG principles.
In recent years, several major cyber-attacks have been launched against companies and organisations around the world.
Each of the ownerships and the Foundation rely on their IT platforms to run their businesses and the Group owns various types of intellectual property rights. Consequently, the Group is a potential target for cyberattacks or industrial espionage, and it is a strategic priority to continuously improve cyber-security.
As IT platforms differ across the Group, cyber-security is managed by each company and the Foundation separately. Each has programmes designed to improve resilience against cyber-attacks. In addition, cyber-risk awareness campaigns are conducted regularly throughout each organisation to minimise risks resulting from phishing emails and similar threats.
The Foundation’s risk reporting and management framework is illustrated in the figure to the right
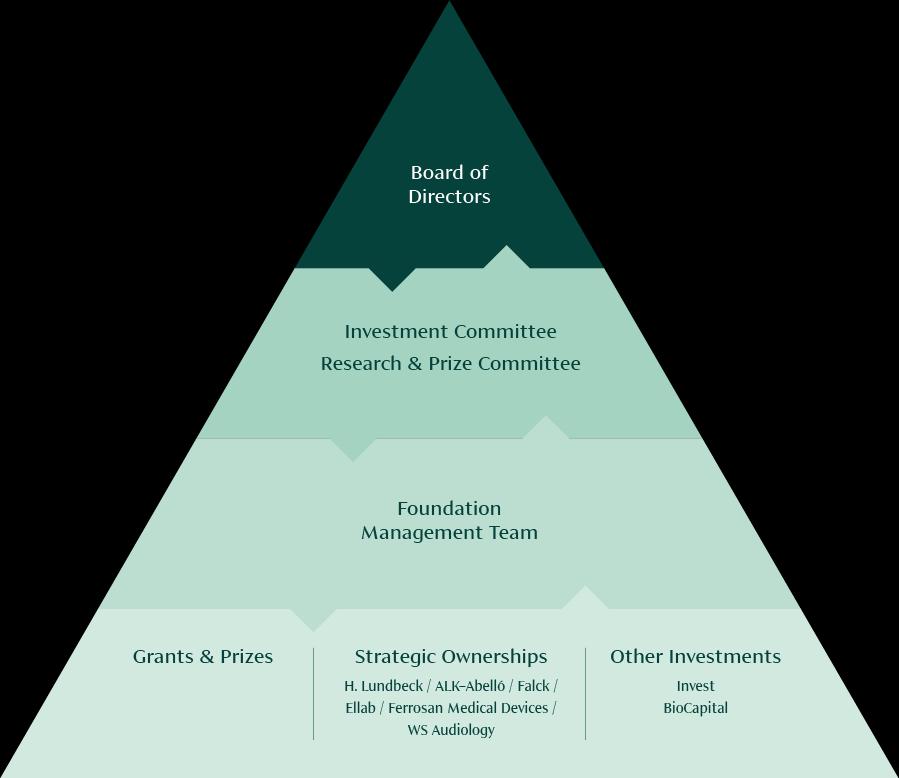
Risk Poor business performance in the Foundation’s strategic ownerships
Context
Mitigation
Performance, ultimately resulting in value destruction of the Foundation’s shareholdings in the strategic ownerships accounting for 67% of the Foundation’s total net wealth
• The Foundation is an engaged owner with a focus on:
•The overall strategy and key activities
•Board and executive management composition to ensure the right competencies
•A sound capital structure to support the strategy
•Good governance with delegation of duties
•Representation on the boards
•Diversification through several different companies
•The companies’ own risk management procedures and their mitigating actions
Research and development risk in pharmaceutical subsidiaries and changes in the key markets
Lack of a successful pipeline in pharmaceutical subsidiaries will limit long-term growth and result in value destruction
Regulatory or market dynamics, including impacts from geopolitical instability, affecting key markets
• • In strategic ownerships:
•Focus on research and development in areas with unmet medical needs & ongoing evaluation of the product pipelines and product benefits
•Monitor political and regulatory developments in key markets
•Cooperation with healthcare authorities to document the value of the companies’ products
•Stringent process for managing price changes
• The Foundation is an engaged owner through:
•Board and executive management composition
•Representation on the scientific committees in the subsidiaries
•A sound capital structure to support research and development
Disruption of production and supply, and defects in product quality or safety, and potential loss of license to manufacture or sell pharmaceuticals
• Financial turmoil and possible economic recession that affects the financial investments which account for 33% of the Foundation’s total net wealth
• Unsuccessful development and/or commercial failure of biotech portfolio companies accounting for 3% of the Foundation’s total net wealth
• • Supply chain and production resilience:
•Initiatives to ensure resilience in production and supply chain include monitoring, production forecasting, safety stock supply of key products, alternative supply strategy, long-term contracts, reducing manufacturing complexity and management of key third parties
•Rigorous pharmacovigilance system, policies, procedures and surveillance to ensure quality and safety of products
•
• Diversified portfolio
• Close monitoring of financial and non-financial risks in the portfolio and dynamic hedging and adjustment of exposure
• Limits on market and concentration risks through investment policy
•
• Close monitoring of risks in the portfolio and active board participation
• Diversified investment in a broad portfolio of life science companies
• Understanding risk in portfolio companies and investing in tranches
Risks and Mitigation (2/2)
The Foundation’s key investment risks
Exposure to the healthcare sector
The Foundation purpose and strategy result in a risk profile that is skewed towards the healthcare sector, which accounts for 63% of the Foundation’s total net wealth
• Geographic concentration risks across the Foundation’s asset classes
Key Group compliance, legal and IT risks
• Non-compliance with applicable laws, industry standards and regulations
Loss, expiration or infringement of intellectual property rights
Regulatory changes may affect future profit and level of grants
Cyber-attacks and cyber-fraud
System down-time
• Key grant and reputational risks for the Foundation
• Misuse of grants
Mitigation
• • Diversified financial investment portfolio regulated in the investment policy
• Ensuring competencies and resources to be an engaged owner in the healthcare sector
• • Ongoing monitoring of geographic exposure
• Hedging against exchange rate risk in key currencies and financing of companies in key currencies (strategic ownerships)
• • Code of Conduct, compliance programmes and employee training
• Whistleblower system
• Internal reviews and monitoring of compliance with laws and industry standards
• Policies and processes to safeguard intellectual property rights
• Close monitoring and evaluation of legislative proposals
•
• • IT policies and procedures are in place to safeguard systems and data
• Cyber-defences are tested on a regular basis
• Fraud or scientific misconduct by grant recipients
• • Transparent grants allocation processes
• Peer reviews by external scientists
• Status reports and budget follow-ups
• Code of conduct signed by grant recipients
The Lundbeck Foundation is committed to transparency and integrity, operating under a clear governance framework and relevant policies that define the direction of the operating model for the Foundation’s broad range of commercial investments and philanthropic grant activities.
Enterprise foundations hold a unique and influential position in Danish society. As majority owners of some of Denmark’s largest companies and as significant contributors to philanthropy, these foundations have the capacity to influence social and economic development in Denmark. Such influence carries responsibility.
The Lundbeck Foundation is committed to transparency and operates with a clear governance model that defines the framework for both its commercial investments and philanthropic grant activities. As an enterprise foundation and majority owner of two listed companies, the Foundation recognises its broad responsibility and the need for robust governance practices.
The Foundation’s Board of Directors has established a governance structure with clear roles, responsibilities, and policies. The policies are regularly reviewed and updated to ensure they remain relevant and effective.
The Foundation follows the recommendations issued by The Committee on Foundation Governance, with two exceptions:
• Board members’ tenure: In accordance with the Foundation’s statutes, Board members are elected for one year at a time, with the possibility of re-election. This approach allows for regular evaluation of each member’s contribution and ensures the Board’s continued effectiveness
• CEO bonus model: The Board of Directors has decided to include a bonus component in the remuneration of the CEO, who is also the CEO of the Foundation’s holding company, Lundbeckfond Invest A/S. The bonus is based on a combination of objective criteria, including the financial results of the subsidiaries, the Foundation’s investment activities and the execution of the Foundation’s strategy. The bonus elements are considered relevant to ensure alignment between the CEO’s remuneration and the Lundbeck Foundation Group’s short and long-term development
For a full overview of the Foundation’s compliance with the Recommendations on Foundation Governance, cf. section 77a of the Danish Financial Statements Act, please use the following link: (https://www.lundbeckfonden.com/en/foundationgovern ance).
Further details on the Foundation’s governance principles can be found on the Foundation’s website and on the right-hand side of this page.
The Lundbeck Foundation’s four governance principles
Compliance
• As an enterprise foundation, the Foundation strives to adhere to the Recommendations on Foundation Governance issued by the Danish Committee on Foundation Governance
• Where the Foundation does not follow these recommendations, this is explicitly stated and explained
Independence
• The Foundation is governed by the Board of Directors. The Board of Directors consists of 12 members, of which eight are elected according to the statutes and four are employee-elected representatives elected by Group employees
• Seven of the eight statute-elected board members (including the Chair) are independent
• Former executives of the Foundation and its subsidiaries cannot be elected to the Foundation’s Board of Directors
• In each strategic subsidiary, the Foundation’s Board of Directors is represented by one board member elected according to the statutes
• The Chair of the Foundation cannot hold board positions in the Foundation’s subsidiaries
Transparency
• The Foundation publishes its financial statements in alignment with listed companies
• All key policies and governance documents are publicly available on the Foundation’s website
• Major decisions on philanthropic grants are proactively communicated via the website, social media and the media
Checks and balances
• The Foundation’s organisational set-up is structured to safeguard a balanced allocation of responsibilities and to avoid undue concentration of decision-making authority
• All significant grant-making decisions are subject to external review by international experts to ensure appropriate quality standards are met
The Lundbeck Foundation Group consists of the Lundbeck Foundation (parent entity) and entities controlled by the Foundation, including its whollyowned investment and holding company Lundbeckfond Invest A/S. The Foundation receives dividends from Lundbeckfond Invest A/S, which are used to support the Foundation’s philanthropic grant activities.
Lundbeckfond Invest A/S’ income is derived from:
• Dividends and returns from its portfolio companies
• Returns on financial investments
The Foundation’s commercial activities and grantawarding activities are managed separately. The Board of Directors of the Foundation determines the overall financial strategy, investment framework and governance principles for the Group. Lundbeckfond Invest A/S manages the Foundation’s commercial activities within the framework defined by the Foundation’s Board of Directors.
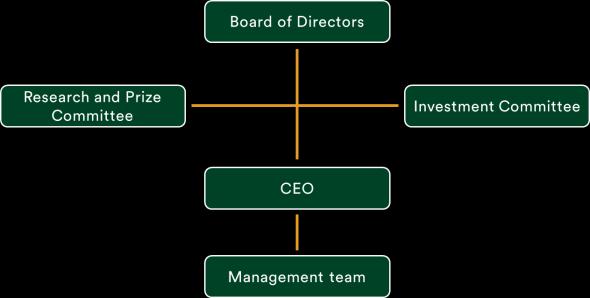
The Lundbeck Foundation is managed by the Board of Directors in collaboration with the CEO
In accordance with the statutes, the Foundation is managed by a Board of Directors consisting of 6 to 12 members elected under the statutes, as well as any members elected by Group employees. At least one of the members of the Board of Directors must be a lawyer or a prominent personality within trade or industry, within the Danish medical profession or within medical research.
The members of the Board are presented on pages 51-53.
The Board’s primary responsibilities include:
• Setting the Foundation’s overall strategy
• Making decisions of major significance or an exceptional nature
• Making final decisions on the allocation of grants
• Supervising the organisation to ensure that the Foundation is managed appropriately and in accordance with applicable laws and the Foundation’s statutes
• Approving the Foundation’s key policies, including those for investments, communication and remuneration
• Appointing the Foundation’s CEO
To ensure that the Board collectively possesses the necessary competencies and experience to act in the
Board member’s attendance at meetings in 2025
Søren Skou, Chair of the Foundation and Lundbeckfond Invest A/S and Chair of the Investment Committee since 12 March 2025
Lars Holmqvist, Deputy Chair of the Foundation and Lundbeckfond Invest A/S and member of the Investment Committee since 12 March 2025
Morten Hummelmose member of the boards of the Foundation and Lundbeckfond Invest A/S and the Investment Committee since 12 March 2025
Naval Ouzren member of the boards of the Foundation and Lundbeckfond Invest A/S and the Investment Committee since 12 March 2025
Michael Kjær, member of the Research Committee
Susanne Krüger Kjær, Chair of the Research Committee
Svend Andersen, member of the Investment Committee
Thomas Mears Werge, member of the Research Committee
Henrik Sindal Jensen, employee representative from H. Lundbeck A/S
Katja Barnkob, employee representative from ALK-Abelló A/S
Mikkel Helmer Nielsen, employee representative from Falck A/S
Morten Egholm Aagaard, employee representative from Falck A/S
Steffen Kragh, former Chair of the Foundation and Lundbeckfond Invest A/S and member of the Investment Committee, stepped down on 12 March 2025
● Attended ○ Did not attend □ Momentarily conflicted
best interests of the Foundation, the Board of Directors has developed three competency profiles that are used when recruiting new members. These profiles are designed to support the Foundation’s purpose and strategic objectives and are defined as Commercial, Finance and Science profiles.
Independence is a key principle in the composition of the Board of Directors. The majority of the members of the Board of Directors, including the Chair, are
independent, and none have previously held executive roles in the Foundation or its subsidiaries.
The Board of Directors meets at least four times a year and holds an annual seminar to review, discuss and refine the Foundation’s strategy.
To support its work, the Board of Directors has established two committees: the Research and Prize Committee and the Investment Committee. These
committees meet regularly to prepare and discuss matters related to the Foundation’s grant and prize activities and investment activities, respectively.
Active involvement in strategic ownerships
The Lundbeck Foundation is a long-term strategic shareholder in six companies: Lundbeck, ALK, Falck, Ferrosan Medical Devices, Ellab and WS Audiology. The Foundation actively monitors the performance of the companies and contributes to value creation as a longterm owner, exerting influence through representation on the companies’ boards.
Independence is fundamental to the Foundation’s approach to strategic ownership. Board representation in the Foundation’s strategic ownerships is structured to ensure objective oversight and a clear separation of roles. In the Foundation’s three strategic subsidiaries –Lundbeck, ALK and Falck – the Foundation is represented on the board by the CEO (who serves as Deputy Chair) and a member of the Foundation’s Board of Directors. In Falck, the representative is a former Board member. In the Foundation’s other strategic companies – Ferrosan Medical Devices, Ellab, and WS Audiology – a member of the Foundation’s management team serves on the board
The Board of Directors has established a clear and robust framework for the governance, administration and oversight of all grant-making activities within the Lundbeck Foundation through a dedicated grant governance policy. Grant governance is a central element of the Foundation’s overall governance structure.
The grant governance framework is designed to ensure that all grants are awarded in accordance with the Foundation’s statutes and strategic objectives, and is guided by four core principles:
• Compliance: All grant activities comply with applicable laws, the Foundation’s statutes and relevant codes of conduct, including research integrity
• Transparency: All grant processes and decisions are documented and, where appropriate, communicated publicly
• Fairness and equal treatment: All applications are assessed consistently and impartially
• Accountability: Clear roles and responsibilities are defined for all parties involved, with appropriate oversight and reporting
The Board of Directors holds ultimate responsibility for all grant allocations. The Foundation operates under a governance model, approved by the Danish Business Authority, which allows the Board of Directors to
delegate grant mandates to executives, committees or panels established by the Board of Directors. These mandates authorise the implementation of the Board of Director’s grant decisions within clearly defined boundaries. Each body operates under formal rules of procedure approved by the Board of Directors, and all members are appointed by the Board of Directors and subject to strict rules on impartiality and conflict of interest
Committees, panels, and executives report regularly to the Board of Directors, typically through the Research & Prize Committee, on their activities, decisions, and use of mandates. The Board of Directors may establish ad hoc evaluation committees and panels or consult with international experts as required.
Research and Prize Committee
To facilitate grant and prize activities, the Foundation has established a committee that serves as advisory body, implements the Board of Director’s grant decisions and submits recommendations according to mandate set by the Board of Directors.
The Research & Prize Committee consists of three to five Board members appointed for their research expertise, in accordance with the statutes. The committee meets at least four times a year, or as required by special circumstances The CEO and the Senior Vice President, Grants & Prizes, Director of Science, neither of whom are members of the Research and Prize Committee, assist the committee in its work. The members of the Committee, who are also members
of the Grants and Prizes Panel (see below), are tasked with:
• Advising and submitting recommendations for decisions to the Board of Directors regarding applications, prize nominations and strategic research policy issues
• Implement the Board of Director’s grant decisions
• Supervising the implementation of the Foundation’s strategy within grants and prizes
The selection committee for The Brain Prize
This committee reviews nominations for The Brain Prize. All members are external and independent of the Foundation, representing the highest level of expertise in brain research.
The Foundation has established four expert panels to evaluate applications and implement decisions on grants.
This panel comprises three to four members of the Board of Directors appointed for their research expertise according to the statutes as well as three external experts from within the field of neuroscience. The panel evaluates applications and implements decisions on grants according to a grant mandate issued by the Board of Directors of the following instruments:
• LF Fellows
• LF Professors and
• LF Collaborative Projects
This panel comprises 13 – 17 external biomedical research experts. The panel evaluates applications for:
• LF Postdocs
• Early-Career Clinician Scientist
• LF Ascending Investigators
• LF Experiments.
The Scientific Enrichment Prize Panel comprises three members with experience in handling diversity policies in a variety of research environments. The panel evaluates nominations for the Lundbeck Foundation Scientific Enrichment Prize.
This panel comprises six to eight members with expertise in neuroscience and cardiometabolic diseases. The panel evaluates and selects the applications for approval by the Research & Prize Committee.
The Board of Directors issues annual grant mandates to selected executives – the CEO and the Senior Vice President, Grants & Prizes, Director of Science – to implement the Board of Directors’ decisions on specific grant activities. These mandates define the scope of authority, evaluation and recommendation processes, and reporting requirements.
The Senior Vice President, Grants & Prizes, Director of Science, is responsible for managing day-to-day operations in the Grants & Prizes department
The Board of Directors has established an Investment Committee to support the governance and oversight of the Foundation’s investment activities. The Committee consists of three to five members of the Board of Directors, appointed for their investment and business experience. Members are appointed annually by the Board of Directors.
The Investment Committee meets four times a year, or as required by special circumstances. Its main responsibilities are to:
• Prepare decisions and recommendations for consideration and approval by the Board of Directors
• Approve investments within the framework of the investment policy
• Monitor the Foundation’s investment activities and performance in accordance with the investment policy approved by the Board of Directors
• Provide guidance to management on significant investments and market developments
The Board of Directors retains overall responsibility for investment management and annually reviews and approves both the investment policy and the terms of reference for the Investment Committee
Each year, the Board of Directors discusses and approves several policies which are a vital part of the framework for the Foundation’s activities.
Please find the Foundation’s policies at the Foundation’s website: https://www.lundbeckfonden.com/en/policies
The Lundbeck Foundation’s grant activities are guided by the following principles:
• Grants are awarded in accordance with the Lundbeck Foundation’s statutes, applicable law and the Lundbeck Foundation’s strategic objectives
• The grant policy ensures transparency regarding the purposes, main categories and recipients of grants
• The Board of Directors is responsible for all grant decisions and for ensuring that grants are awarded in a manner that supports the Foundation’s purpose and societal value
The Foundation’s grant activities are anchored in its statutes, which set out two main purposes
a. To consolidate and expand the activities of the Lundbeck Group
b. To make distributions to the purposes set out in Article 6 of the statutes
In accordance with the statutes, grants are allocated within the following main categories, reflecting Article 6 of the statutes:
• Social causes
• Research
• Cultural purposes
• Relatives of the Founder
• Employees of the Lundbeck Group
• Subsidiaries
The Board of Directors exercises discretion to decide which of the listed purposes to support, and to what extent, within the framework set by the statutes and applicable law. This means that grant allocations may vary from year to year, and some purposes may receive support in one year while others may not. This flexibility allows the Foundation’s grant activities to be adapted to strategic priorities and societal needs, as long as they remain within the overall framework of the statutes.
Article 6a (Relatives of the Founder)
Article 6b (Employees of the Lundbeck Group)
Article 6c (Research)
Article 6d (Research)
Article 6e (Subsidiaries)
Article 6f (Research)
Article 6g (Research)
Article 6h (Research)
Article 6i (Social causes)
Article 6j (All categories)
The grant objectives listed under Article 6 of the statutes are categorised as follows
The Foundation may make grants to descendants of the parents of the founder and her spouse.
The Foundation may provide financial support to, and otherwise make distributions for the benefit of, existing and former employees of the Lundbeck Group, including for training and education, for holidays and for holiday purposes. In so far as there is no need in each individual case to make distributions at any other time of the year, such distributions will only be made once a year on 20 July, which is the birthday of the deceased Mr Hans Lundbeck, manufacturer
The Foundation may grant honorary awards to physicians, scientists and others.
The Foundation may grant support for scientific purposes, primarily for specific projects.
The Foundation may support special research projects within the Lundbeck Group.
The Foundation may grant support to hospitals and for disease prevention.
The Foundation may provide support for training and education in the widest sense of the words, if relevant, by way of interest-free loans.
The Foundation may make grants for nurses.
The Foundation may distribute up to 3% of the profit for the year, before tax, less retained earnings, in subsidiaries and associates, including LFI a/s, for the support of old and/or sick people, as well as those in need.
The Foundation may support other purposes, as decided by the Board of Directors. However, support for such purposes may only account for 25% of the total annual distributions in each calendar year.
The Board of Directors has established a strategy for fulfilling the Foundation’s statutory purpose. In line with this strategy, the Board has decided that the Foundation’s grant activities will primarily support research in the health sciences, with the aim of advancing understanding of the brain and promoting better prevention, diagnosis and treatment of brain diseases. This is directly reflected in the Foundation’s 2030 strategy and five strategic value flags:
• Top-tier neuroscience: Prioritising research that advances understanding of the brain and the development of new treatments for brain diseases.
• Close collaboration between research and business: Supporting initiatives that foster collaboration between universities, hospitals and companies.
• Leading healthcare companies: Developing and maintaining international market leaders in healthcare through active and engaged ownership.
• Attractive financial returns: Balancing grant-making with the need for consolidation and robustness, ensuring the Foundation can maintain and increase its grant levels over time.
• Active public voice: Contributing to public debate and increasing awareness of brain health and the importance of research
The Foundation primarily operates with the following types of grants:
• Personal research grants within the health sciences, focusing on projects that lead to increased understanding of the brain and to better prevention, diagnosis and treatment of brain diseases
• Strategic/thematic research grants within the health sciences, aimed at advancing understanding of the brain and improving prevention, diagnosis and treatment of brain diseases
• Awards for outstanding researchers
• Grants for science communication and teaching within national sciences and neuroscience subjects from upper secondary level and onwards
Grants are mainly awarded through open competition based on applications submitted in response to public calls. However, grants may also be awarded without a public call. Grant opportunities, criteria and deadlines are published on the Foundation’s website. Applications are assessed based on scientific quality, relevance to the Foundation’s purpose and scientific and societal impact
All grant recipients must comply with the Foundation’s General Terms and Conditions for Research Grants.
The Foundation expects researchers who receive funding to conduct their research according to recognised codes of Good Research Practice, including the Danish Ministry of Higher Education and Science’s Code of Conduct for Research Integrity, the Medical Research Council’s Good Research Practice and the International Society for Pharmacoepidemiology’s Guidelines for Good Pharmacoepidemiology Practice. This applies to all aspects of research, including collaboration, data collection and analysis, funding applications, publication of results and recognition of contributions.
It is a prerequisite that researchers who receive funding from the Foundation are affiliated with institutions that have their own published codes and guidelines for Good Research Practice, and that grant recipients comply with such guidelines. In addition these institutions must have formally described procedures for handling any suspicion of scientific dishonesty.
To be eligible for a grant from the Foundation, the grant recipient, their host institution, and all researchers and third parties affiliated with the research project must comply with all laws and rules relevant to the research project

Søren Skou Chair



Other board and management positions
· Chair of Bygma Gruppen A/S, Controlant hf., C.W. Obel A/S, Danish Crown AmBA, The Mærsk Mckinney Møller Center for Zero Carbon Shipping, HES International B.V.
· Senior Advisor, Blackrock Global Infrastructure Partners and chair of two portfolio companies (VTG GmBH and Skyborn Renewables GmBH)
Education and competencies
BSc (Econ) from Copenhagen Business School and MBA from IMD in Switzerland
· Member of the boards of H. Lundbeck A/S, ALK-Abelló A/S and Vitrolife AB (Sweden)
Extensive global leadership experience and expertise within strategy and corporate development, capital markets, risk management, investments, organisational development and sustainability Degree in Business Administration
Experience in management, finance, sales and marketing in international life science companies, including medtech and pharmaceutical companies
in ALK-Abelló A/S
· Professor, Chief Physician, DMSc. Head of Institute of Sports Medicine, Department of Orthopaedic Surgery, Bispebjerg Hospital
· Coordinating Professor (for Bispebjerg-Frederiksberg Hospital), Institute of Clinical Medicine, University of Copenhagen
Scientific production within musculoskeletal and metabolic research. Skills in research, development and research evaluation. Experience in research management and international scientific board work
· Founder and member of the board of the Frececo Foundation and CEO of Frececo ApS Board Member of Human Rights Watch
· Member of the Investment Committee in the EQT Foundation
MSc in Economics (Cand. Polit) from University of Copenhagen and a master’s degree in finance from London University
Extensive international experience in strategic leadership, as well as private equity, investment and fundraising, gained from a career at EQT Partners

Considered independent

independent
Elected
Board committee(s)
Member of the Investment Committee
Member of the Investment Committee

Considered independent
Chair of the Research and Prize Committee and the Grants and Prizes Panel
Shareholdings 1,182 shares in ALK-Abelló A/S
Other board and management positions
CEO at Sensorion, a Euronext-listed biopharmaceutical company (France)
· Non-Executive Director of the CRODA board
Education and competencies
M.Sc. in Chemical Engineering from the University of Technology of Compiègne (UTC)
More than 20 years of expertise in clinical development, operational and strategic management roles within the pharmaceutical industry
Investor and advisor
Advisor for London-based private equity companies
Professor, Copenhagen University Hospital
· Head of Research, Unit of Virus, Lifestyle and Genes, Danish Cancer Institute
· Member of the Steering Committee of the Mermaid project
Member of the board of Johannes Clemmesens Research Foundation
· Member of the Human Papillomavirus Prevention and Control Board
· Visiting professor, Johns Hopkins University, Baltimore, USA

Considered dependent
Elected 2023
Member of the Research and Prize Committee and the Grants and Prizes Panel
Professor at the Institute for Clinical Medicine, University of Copenhagen
· Research Chief at the Inst. Biol. Psychiatry, Mental Health Services, Copenhagen University Hospital Member of the working group of the National Genome Centre, and Member of the Strategic Research Council, Copenhagen University Hospital
· Principal Investigator, NIH & EU Initiatives on mental disorders
· Expert Witness (WADA) CAS, Court of Arbitration for Sport, Lausanne (CH)
· Member of the board of Sputnik Holding JAC ApS
BSc in Economics and Business, and Graduate Diploma in Business Administration, Sales & Marketing and Organisation
Extensive international leadership experience in several pharmaceutical sectors with a proven track record of creating sustainable value
MD and DMSc
Substantial scientific output within oncology. Skills in research development, clinical cancer epidemiology, research evaluation and innovation. Longstanding experience within international research management
B.Sc. Biochemistry; M.Sc. Mol. Oncology; PhD, Mol. Neuroscience
Substantial scientific output on Psychiatry and Neurosciences. Skills in Genetics and epidemiology of complex traits and disorders. Longstanding experience in large-scale, international initiatives








in H. Lundbeck A/S and 23,000
ALK-Abelló A/S
Board positions Chair of the board of Ørsted A/S and deputy chair of the boards of H. Lundbeck A/S*, ALK-Abelló A/S*, Falck A/S* and Nordea Bank Abp
* Board positions included in the position as CEO of the Lundbeck Foundation
Chair of the board of Cresco Capital Services A/S and deputy chair of the boards of Obel-LFI Ejendomme A/S and CN8 A/S
Member of the boards of Ferrosan Medical Devices Group, Ellab Group WS Audiology A/S and LFI Silva Investments A/S
Chair of the boards of SNIPR Holdings ApS, SNIPR Biome ApS and CytoKi Pharma ApS
Member of the boards of IO Biotech, Inc, NMD Pharma A/S and QuantumCell ApS



For the period 1 January – 31 December
For the period 1 January – 31 December
Basis of preparation - continued
of preparation
Reporting entity
Lundbeckfonden (the Lundbeck Foundation) is domiciled in Denmark. The Foundation’s registered office is at Scherfigsvej 7, 2100 Copenhagen. These consolidated financial statements comprise the Parent Foundation and its subsidiaries.
Basis of accounting
The consolidated financial statements have been prepared in accordance with IFRS Accounting Standards as adopted by the EU and additional requirements of the Danish Financial Statements Act. The consolidated financial statements were approved by the Board of Directors on 26 March 2026.
Details of the Group’s accounting policies are included in this note and note 36 Material accounting policy information.
Functional and presentation currency
Items included in the financial statements of each of the Group’s entities are measured using the currency of the primary economic environment in which the entity operates (the functional currency).
The consolidated financial statements are presented in Danish kroner (DKK), which is also the functional and presentation currency of Lundbeckfonden (the Parent Foundation). All amounts have been rounded to the nearest DKK million, unless otherwise indicated.
Principal accounting policies
Apart from the general accounting policies, which are described in note 36 Material accounting policy information, some other relevant information is
specified in each of the individual notes to the consolidated financial statements. The accounting policies have been applied consistently in the preparation of the consolidated financial statements for all the years presented.
On 2 December 2024, Lundbeck announced the successful acquisition of Longboard Pharmaceuticals, Inc. (’Longboard’). Through this transaction, Lundbeck obtained control of Longboard by acquiring 100% of Longboard’s share capital. The purchase price allocation was finalised in 2025, and, consequently, the 2024 comparative information has been restated to reflect final fair value of the Longboard’s net assets at the acquisition date. The restatement affected only the Statement of financial position (Balance sheet) and had no impact on Statement of profit or loss or equity. For further information see note 31.1 Acquisitions of businesses, etc.
In preparing the consolidated financial statements, Management has made estimates and judgements that affect the application of the Group’s accounting policies and the reported amounts of assets, liabilities, income and expenses. Actual results may differ from these estimates.
Estimates and underlying assumptions are reviewed on an ongoing basis. Revisions of estimates are recognised prospectively. Management believes that the below accounting estimates, assumptions and judgements are significant to the consolidated financial statements.
Changes in material accounting policy information
New and amended standards adopted by the group New and amended IFRS Accounting Standards and interpretations issued by the IASB and endorsed by the EU, effective on or after 1 January 2025, have not had a significant effect on the consolidated Financial Statements.
New standards and amendments issued but not yet effective
Furthermore, new or amended IFRS Accounting Standards and interpretations issued by the IASB that have not yet become effective are generally not adopted until they become effective and are endorsed by the EU. Management does not anticipate any significant impact on the consolidated Financial Statements in the period of initial application from the adoption of these new standards and amendments, except for IFRS 18 Presentation and Disclosure in Financial Statements, which replaces IAS 1 Presentation of Financial Statements and is effective from 1 January 2027. IFRS 18 is expected to change the presentation of the income statement by differentiating earnings from operating, investing, and financing activities. IFRS 18 will also require additional disclosures but is not expected to affect the Group’s accounting policies for recognition and measurement and, accordingly, is not expected to impact reported net results.
* Abilify long-acting injectable (LAI) franchise comprises the following products: Abilify Maintena®, Abilify Maintena® 960 mg, and Abilify Asimtufii®.
Lundbeckfonden
The Executive Management of Lundbeckfonden participates in a short-term incentive programme that provides an annual bonus for the achievement of predetermined targets. Bonuses under the programme amounted to DKK 3.7m in 2025 (DKK 4.0m in 2024), which was equivalent to 81% of the maximum, and will be paid out with a delay of 12 months.
In addition, Lundbeckfonden has incentive programmes to be able to attract and retain skilled and qualified employees. The costs related to the programmes are recognised as employee costs when there is certainty about the amount due and the time of payment.
To attract, retain and motivate key employees and align their interests with those of the shareholders, a number of incentive programmes have been established in Lundbeck, ALK and Falck. The Group uses short-term incentive programmes that provide an annual bonus for the achievement of pre-determined targets for the financial year, as well as long-term, equity-based and debt-based programmes, as described below.
Lundbeck
Lundbeck uses equity- and cash-settled programmes.
Equity-settled programmes
As from 2023, the Group has established a performance share units (PSU) programme in substitution for the previous restricted share units (RSU) programme for Lundbeck’s Registered Executive Leadership Team and
key employees, as part of Lundbeck’s recurring longterm incentive programme. The general terms and conditions for the PSU programme are similar to those applying to the RSU program. In 2024, the Registered Executive Leadership Team and some key employees were granted PSUs. The total number of units granted to the above-mentioned employees is disclosed below. The price of the granted shares is referenced to the price of B-shares. The participants were selected based on job level. All the PSUs/RSUs vest three years after grant. Vesting is subject to the Board of Directors’ decision on vesting, to Lundbeck achieving certain strategic and financial targets specified by the Board of Directors, and to continuing employment with the Group during the vesting period. The fair value of PSUs and RSUs has been calculated based on the share price reduced by an expected dividend yield of 2.00% p.a. The fair value is disclosed below for each date of grant. At 31 December 2025, a total of 1.6 million instruments (1.4 million at 31 December 2024) were outstanding for key management, including all ongoing programmes.
In 2025 and 2024, the cash-settled programmes consisted of performance cash units (PCUs) and restricted cash units (RCUs). The cash-settled programmes cannot be converted into shares, as these programmes are settled in cash.
As from 2023, the Group has established a PCU programme in substitution for the RCU programme for a few key employees in the subsidiaries. The general terms and conditions for the PCU programme are similar to those applying to the RCU programme. In 2025, their programme was re-established to only cover an RCU programme. At 31 December 2025, the RCUs granted to key employees totalled 11,258 RCUs (18,842 PCUs for the 2024 programme). All PCUs and RCUs will vest three years after grant. Vesting is subject to the Board of Directors’ decision on vesting, to Lundbeck achieving certain strategic and financial targets specified by the Board of Directors, and to continuing employment with the Group during the vesting period. The size of the amount depends on the value of the Lundbeck share on the vesting date. The fair value at the time of the initial grant was DKK 40.5 per RCU
(DKK 31.07 per PCU for the 2024 programme). The RCUs granted in 2022 vested in 2025, after which the programme was settled. The RCUs granted in 2021 vested in 2024, after which the programme was settled.
Fair value, liability and expense recognised in the income statement
The PSUs/RSUs granted are recognised in profit or loss for 2025 and 2024 at an expense corresponding to the fair value at the time of grant for the part of the vesting period attributable to each one. The total expenses recognised in respect of equity-settled programmes amounted to DKK 44m (DKK 45m in 2024). At 31 December 2025, the grant date fair value of unvested equity settled awards outstanding was DKK 221m (DKK 192m at 31 December 2024).
The PCUs/RCUs granted are recognised in the Statement of profit or loss at an expense for the year arising from remeasurement of the fair value of the cash settled liability. The total expenses recognised in respect of cash-settled programmes amounted to DKK 16m (DKK 2m in 2024) and cover all cash-settled programmes in force at 31 December 2025. At 31 December 2025, the total liability in respect of cash-settled programmes was DKK 17m (DKK 2m at 31 December 2024). The total expenses recognised in profit or loss for all incentive programmes amounted to DKK 60m in 2025 (DKK 47m in 2024).
The incentive plans consist of share options, performance share units, and restricted stock units that are considered sufficiently covered by treasury shares.
The share options entitle the holder to acquire one existing B share of DKK 0.5 nominal value in the company per share option. The performance share units and restricted stock units entitle the holder to receive one existing B share per performance share unit or restricted stock unit free of charge.
The vesting period for both share options, performance share units, and restricted stock units is three years after grant. Vesting of share options and performance share units is conditional upon certain targets being met and upon the participant not having resigned. Target achievement is met upon fulfilment of strategic key performance indicators. In case performance is below the threshold there will be no units vesting, and if above target, a multiplier is applied that can increase the vesting by up to 100%. Vesting of restricted stock units is conditional upon continued employment.
The exercise of share options is possible in the trading windows following the release of annual and interim reports conditional upon the share option holder not having resigned at the time of exercise. For performance share units and restricted stock units, the final transfer of ownership takes place at vesting three years after the grant.
From the 2023 plans and onwards, a cap applies to the maximum total value gain from share options, performance shares, and restricted stock units at exercise and/or vesting, respectively, granted in a calendar year. The cap is four times the annual base salary at the time of award of the share options, performance share units, and restricted stock units concerned.
Expensed in the income statement
In 2025, the total cost of share-based payments included a financial expense of DKK 0 due to the exercise and cash settlement of share options (2024: Financial income of DKK 3m). The total cost included DKK 18m related to adjustment in the share options and performance share units (2024: DKK 22m).
Specification of outstanding share options, performance share units, and restricted stock units:
The Board of Directors decided for three trading windows in 2025 to settle share options by cash and a total of 270,970 share options were exercised and total cash payments amounted to DKK 20m.
In 2025 the Board of Directors decided not to settle share options by shares and no share options were exercised.
The Board of Directors decided for two trading windows in 2024 to settle share options by cash and a total of 616,647 share options were exercised and total cash payments amounted to DKK 33m. For two trading windows in 2024 the Board of Directors decided to settle share options by shares and a total of 77,168 share options were exercised
Outstanding share options, performance share units and restricted stock units have the following characteristics
Fair value of share options, performance share units, and restricted stock units granted
Share options
Fair value at grant date is measured in accordance with the Black & Scholes model for valuation of share options, using the following assumptions:
In 2025, performance share units have been granted at DKK 140 per share (2024: DKK 126 per share).
No restricted stock units were granted in 2025 or 2024.
The remuneration of the members of the Executive Committee consists of three key components: base salary, a short-term incentive plan and a long-term incentive plan.
Falck’s short-term incentive programme is a cash-based one-year programme with focus on financial, commercial and ESG KPIs.
The long-term incentive programme is a cash-based incentive on a three-year performance period with an 50% EBIT target, 40% on the implementation of strategic Care for More '27 initiatives, and 10% on CO2 reduction at the end of the three-year period.
Note 5
The amounts include gains and losses on disposal of intangible and tangible assets.
Note 6
Fees to auditors appointed at the annual meeting
PwC audits the consolidated financial statements of the Lundbeck Foundation and a majority of the subsidiaries’ financial statements.
The fee for non-audit services provided to the Group by PricewaterhouseCoopers Statsautoriseret Revisions-
Other operating items, net
partnerselskab, Denmark, consisted of limited assurance of the sustainability statements, consultancy services, other assurance services and other accounting and tax advisory services.
In December 2025, the Lundbeck’s Board of Directors approved a plan to divest a non-core production site in Italy, a wholly owned subsidiary. The divestment of the production site is expected to be completed within one year from the reporting date. As a result of this decision, an impairment loss of DKK 639m has been recognised in other operating expenses, net, in 2025, as the carrying amount exceeded the recoverable amount determined. The recoverable amount was based on fair value less costs of disposal and was determined based on information available at the reporting date. The fair value measurement is categorised as level 2 in the fair value hierarchy.
A breakdown of the impairment loss by asset class is presented below
See note 31.1 for information on transaction costs related to acquisitions and divestments and integration costs.
In 2025, the change in value of biological assets and related land included a reversal of impairment on property and land of DKK 3m (impairment of DKK 8m in 2024).
In 2024, as part of the business combination, Lundbeck entered into a deal-contingent forward (foreign exchange contract) to mitigate the foreign exchange risks associated with the acquisition of Longboard. The derivative was designated at fair value through profit or loss and amounted to DKK 380m. The contract was entirely settled with the closing and cash payment of the acquisition of Longboard.
9
The Group operates in a multinational tax environment. Complying with tax rules can be complex, as the interprettation of legislation and case law may not always be clear or may change over time. In addition, transfer pricing disputes with tax authorities may occur. Management’s judgements are applied when estimating the expected outcome of disputes or interpretational uncertainties. Provisions for uncertain tax positions are determined by using the ‘most probable outcome’ or ‘single best estimate’ method, depending on the type of uncertainty.
At 31 December 2025, uncertain tax positions comprise a liability of DKK 363m (DKK 449m at 31 December 2024) and an asset of DKK 120m (DKK 128m at 31 December 2024). Uncertain tax positions are recognised as current tax. Management believes that the provision is adequate. However, the actual obligation may differ from the provision made and depends on the outcome of litigations and settlements with the relevant tax authorities.
The Group is within the scope of the OECD Pillar Two model rules, and it applies the IAS 12 exception to recognise and disclose information about deferred tax assets and tax liabilities related to Pillar Two income taxes. The Group will incur top-up taxes due to the Pillar Two legislation that became effective on 1 January 2024. Under the legislation, the Group is liable to pay a top-up tax for the difference between its GloBE effective tax rate
in each jurisdiction and the 15% minimum rate, where transitional Safe Harbour requirements are not met.
The Group has estimated that the majority of the jurisdictions in which it operates meet the transitional Safe Harbour requirements, except for six countries. Considering the impact of the specific adjustments in the Pillar Two legislation, the Group recognised a current income tax expense of DKK 1m (DKK 14m
which is
in the
(DKK 138,105 in 2024).
assets - continued
Product rights at 31 December 2025 included product rights not yet commercialised amounting to DKK 18,280m.
In 2025, an impairment loss of DKK 16m was recognised in connection with the planned divestment of a noncore production site in Italy, as disclosed in note 7 Other operating items, net
Other intangible assets includes intangible assets in progress, and individual development projects running for short-term periods. In 2025, Patent licence of DKK 994m, capitalised in 2024, was taken into use.
Other intangible assets includes intangible assets in progress, and individual development projects running for short-term periods. In 2024, ALK has entered a Collaboration, License and Distribution Agreement with ARS Pharmaceuticals Operations Inc. where the asset of DKK 994m is not ready for use due to pending regulatory and other approvals.
Product rights at 31 December 2024 included product rights not yet commercialised amounting to DKK 20,155m.
Intangible assets acquired as part of the acquisition of Longboard in 2024 amounted to DKK 21,249m at the acquisition date. This amount represents the final fair value measurement at the acquisition date, and accordingly, the 2024 comparative figures have been restated to reflect the final purchase price allocation, as disclosed in note 31.1 Acquisitions of businesses, etc.
Please refer to note 31.1 for further information about additions on acquisitions
As required by IFRS, intangible assets with indefinite useful lives, intangible assets not yet available for use and goodwill acquired in a business combination are tested for impairment annually, irrespective of whether there is any indication of impairment.
Management has performed impairment tests of goodwill related to the investments in the subgroups.
The impairment test for 2025 did not result in recognition of any impairment losses on goodwill.
Based on sensitivity analyses related to the impairment test for 2025, it is Management’s opinion, that no probable change in any key assumptions would result in impairment losses.
The impairment test for 2024 did not result in recognition of any impairment losses on goodwill.
- continued
Methodology used for Lundbeck
At 31 December 2025, goodwill related to Lundbeck amounted to DKK 7,008m.
Goodwill has been tested at an aggregated level for Lundbeck as one cash-generating unit (CGU).
In the impairment test of the CGU, based on the fair value less cost of disposal, the market price of Lundbeck is compared with its carrying amount.
Methodology used for ALK
At 31 December 2025 goodwill related to ALK amounted to DKK 553m.
Goodwill has been tested at an aggregated level for ALK as one CGU.
In the impairment test of the CGU, based on the fair value less cost of disposal, the market price of ALK is compared with its carrying amount.
Methodology used for Falck
At 31 December 2025, goodwill related to Falck amounted to DKK 3,887m.
Impairment tests are carried out per business segment which is the lowest level of the CGUs to which the carrying amount of intangibles, i.e. goodwill and customer contracts, can be allocated and monitored with any reasonable certainty.
Impairment tests are carried out on the business segments Societal Care Europe, Individual Care Europe, Industrial Fire services, Societal Care US and Individual Care LATAM.
The carrying amount of the Falck brand is tested at Group level based on Group-wide cash flows (aggregated cash flows determined for each CGU), less the total carrying amount of the goodwill and other non-current assets. The impairment test shows significant headroom from comparing the value in use to the carrying amount of all assets in the Falck Group.
Keyassumptionsintheimpairmenttest
The recoverable amounts for the CGUs are determined based on the value-in-use.
In the impairment tests, the discounted values of the future net cash flows of each of the CGUs value-in-use are compared with their carrying amounts. The value-inuse is calculated using certain key assumptions for the expected future cash flows and applied discount factor.
The cash flow projections are grounded in financial budgets and business plans approved by Management. These projections involve judgement and estimates, which, while based on experience, carry some uncertainty. To calculate the present value, discount rates are applied using a weighted average cost of capital (WACC), which incorporates both estimates and external sources.
The primary assumptions included in the value-in-use calculation are:
• Revenue growth in the forecast period
• EBIT margin before special items (b.s.i.)
• Net working capital
• Discount rates
• Growth rate in terminal period
Falck’s climate related transition plans are not assessed to impact the future operating margins of the CGUs materially.
Revenue growth projections in the financial forecast for 2026-2030 are estimated based on current operations and the expected market development for the individual CGU.
For Societal Care Europe, new contracts in Spain and Sweden represent a significant increase in revenue and in the longer term the strategic focus on key European markets is unchanged.
For Individual Care Europe, an increase in revenue is expected through organic growth and launch of new products in line with the development in 2025.
The expected growth for Industrial Fire services is moderate in line with Falck’s strategy plan.
Societal Care US expects an increase in revenue driven by organic growth resulting from new initiatives in 2025 that target additional states and introduce new products. Additionally, revenue growth is expected through price increases on current contracts.
During the forecast period Individual Care LATAM expects revenue growth driven by increased number of subscribers, alongside entry into new markets.
When estimating the CGUs’ EBIT margin b.s.i. in the financial forecast for 2026-2030, past experiences are taken into consideration.
The EBIT margin b.s.i. in the forecasting period in Societal Care Europe is expected to steadily increase, driven by both a new contract in Spain and execution of profitability improvement initiatives in accordance with the Falck Group strategy plan.
Individual Care Europe expects an improvement in EBIT margin b.s.i. driven by the introduction of new products and the implementation of efficiency initiatives in 2025.
For Industrial Fire services, an improved EBIT margin b.s.i. is expected in the forecasting period due to operational efficiency improvements.
The EBIT margin b.s.i. in Societal Care US is expected to be relatively stable in the forecasting period due to continued focus on revenue cycle management and other operational efficiency initiatives. The operational efficiency initiatives are expected to compensate for other impacts e.g. from changes to contracts.
For Individual Care LATAM, an increasing EBIT margin b.s.i. is expected mainly due to an increased number of subscribers.
Net working capital is based on historical experience for each CGU and thus increases on a linear basis as the level of activity increases.
The discount rates for 2025 impairment testing purposes are based on a calculation of weighted average cost of capital (WACC).
The cost of equity is calculated using Capital Asset Pricing Model (CAPM). The beta applied at year-end 2025
assets - continued Note 11
is obtained from comparable peers. Market risk premiums have been added based on observed market data.
The cost of debt is based on Falck’s debt margin plus the risk-free interest to Falck’s spread plus the risk-free interest of the countries in which the CGU operates.
The terminal growth rates are equal to the International Monetary Fund (IMF) projections of inflation in 2030.
Carryingamountsandkeyassumptions
The carrying amount of goodwill and customer contracts, and the key assumptions used in the impairment testing at 31 December are presented for each CGU:
Impairment testing of other intangible assets
Other intangible assets in use with indefinite useful lives are tested for impairment if there is any indication of impairment. Furthermore, prior impairment losses are reviewed for possible reversal at each reporting date.
Lundbeck
Materialproductrights
Vyepti®
The eptinezumab product rights (Vyepti®), which are investigational monoclonal antibody (mAb) for migraine prevention targeting the calcitonin gene-related peptide (CGRP), were acquired in 2019. The value of those
product rights was DKK 13,421m at the time of acquisition. At 31 December 2025, the carrying amount of the Vyepti® product rights, net of amortisation, amounted to DKK 8,497m (DKK 10,154m at 31 December 2024). The remaining amortisation period of the Vyepti® product rights is around 10 years.
Rexulti®
Rexulti® is a prescription medication used as an adjunctive therapy to antidepressants for the treatment of major depressive disorder (MDD) and as a treatment for adults with schizophrenia in certain markets. Rexulti® is co-marketed in a partnership collaboration
with Otsuka Pharmaceuticals Co., Ltd. The carrying amount of the Rexulti® product rights, net of amortisation, amounted to DKK 1,381m at 31 December 2025 (DKK 1,762m at 31 December 2024). The remaining amortisation period of the Rexulti® product rights is around four years.
A family of compounds; a first-in-class, small-molecule inhibitor of monoacylglycerol lipase (MAGLi/MGLL) currently being investigated in clinical trials for the treatment of neurological disorders, along with various compounds in the preclinical phase, was acquired
- continued
in 2019. The value of the family of compounds recognised as product rights was DKK 1,853m at the time of acquisition.
At 31 December 2025, the carrying amount was DKK 1,324m (DKK 1,324m at 31 December 2024) and refers to Lu AG12947, the remaining molecule from the acquisition after the impairment recognised in 2024. As described in the 2025 Testing outcome section below, no further impairment has been identified in 2025. Lu AG12947 is not yet commercialised; consequently amortisation has not commenced.
Bexicaserin
Bexicaserin, a novel 5-HT2C agonist in development for the treatment of seizures associated with developmental and epileptic encephalopathies (DEEs), including Dravet syndrome, Lennox-Gastaut syndrome, and other rare epilepsies, was acquired in December 2024 along with the Longboard acquisition, see note 5.1 Business combination. The value of the product rights was DKK 18,729m at the time of acquisition. The carrying amount of DKK 16,584m at 31 December 2025 (DKK 19,037m at 31 December 2024) was affected by developments in the USD/DKK exchange rate. Bexicaserin is not yet commercialised; consequently amortisation has not commenced.
The recoverable amount of the specific product right is determined as the higher of its fair value less costs of disposal and its value in use. Under both methods, the discounted expected cash flows of the asset are compared with its carrying amount. The expected future cash flows are based on a forecast period, which is the
period used by Management for decision-making, with due consideration of patent expiry. For impairment testing of product rights using fair value less costs of disposal methodology, the fair value measurement is categorised as level 3 in the fair value hierarchy.
The assumptions used in the impairment test are based on benchmarked external data and historical trends. The key parameters used in the estimation of the recoverable amount are revenue, probability of success, earnings, working capital, discount rate, and the preconditions for the cash flow period.
• Financial elements: Prices, rebates, quantities, patient population, market shares, competition, fill rates, prescription rates and Lundbeck costs (including promotion costs)
• Market elements: Healthcare reforms, price reforms, market access, pharma restrictions, launch success, product positioning, competing pharmaceuticals and generics on the market
• R&D elements: R&D spend, collaborations, pipeline success rate, product labelling and liaison with regulatory bodies
• Other elements: Supply chain effectiveness and strength and abilities of partners
Significant assumptions and estimates are applied to the discounted expected future cash flows from the product rights. The assumptions are based on experience, external sources of information, and industry-relevant observations for each product right.
The four category elements in the table below are considered when determining the key parameters for the impairment test.
The impairment tests for product rights are based on a weighted average discount rate, pre-tax, of 9.90% (8.34% in 2024).
Lundbeck
The impairment tests performed in 2025 did not result in the recognition of any impairment loss
Sensitivity analysis of impairment tests focuses on changes in discount rate (WACC) and revenue growth, while all other factors are held constant. Based on these analyses, Management assesses that no reasonably possible change in any key assumption would cause the carrying amounts of the product rights to exceed their recoverable amount as of 31 December 2025.
Lundbeck
During 2024, an impairment loss of DKK 547m was recognised as a result of the negative read-out of a compound of the MAGLi family (Lu AG06474 and Lu AG12947) that was acquired in 2019 through a business combination. Management decided to discontinue the development of the molecule Lu AG06474 after the read-out, as results did not support additional studies, resulting in the individual asset being impaired. The impairment loss was recognised in research and development costs in the Statement of profit or loss.
Impairment testing in 2024 resulted in an impairment of production and administrative software of DKK 2m.
Impairment testing total
The impairment losses per intangible asset category can be specified as follows:
The impairment loss has been recognised in the
statement as follows:
Impairment
plant and equipment - continued
Impairment amounted to DKK 444m of which DKK 430m was recognised in connection with Lundbeck’s planned divestment of a non-core production site in Italy, as disclosed in note 7 Other operating items, net
Furthermore, DKK 14m related to impairment of production equipment in ALK and Lundbeck.
Impairment amounted to DKK 12m, of which, DKK 8m was attributable to land and buildings related to forestry activities in Scotland. Impairment of production and administrative equipment in ALK amounted to DKK 4m.
- continued
The total cash outflow from recognised lease agreements amounted to DKK 561m (DKK 612m in 2024) and includes repayment of lease liabilities and interest.
For disclosures of the lease liabilities, see note 26.3 Lease liabilities.
Biological assets comprise forestry activities in Scotland. The fair value of the biological assets was determined based on a calculation of the present value of future expected cash flows from the forests and independent market valuation from the chartered surveyors
Goldcrest Land & Forestry Group (level 3 input from the fair value hierarchy). The cash flows were calculated based on harvesting volumes according to the Group’s current plan and assessments of future price and cost changes. The cash flows were discounted using discount rates of 2.05 - 3.15% (2.05 – 3.15% at 31 December 2024).
At 31 December 2025, the Group owned 3,414 hectares of forest farming land being managed actively.
The carrying amount of land related to forestry activities is recognised under property, plant and equipment.
The investments in WS Audiology was completed end April 2024. The Group has significant influence through representation on the Boards of Directors of the companies. Consequently, the investments are classified as associates.
The Group’s financial investments classified as financial assets at fair value through profit or loss primarily relate to Invest’s investments. These investments are made based on an investment policy approved by the Board of Directors. The strategy aims for an appropriate diversification of investments across different asset classes and geographical markets to achieve an appropriate diversification of interest rate, exchange rate, credit and equity risks on the financial investments. The purpose is to reduce the risk of losses but also to retain the prospect of gaining a long-term return on the investments.
Financial assets include investments in listed and unlisted equity instruments and securities, including life science investments recognised at their fair value. Investments in unlisted equity instruments and securities amounted to DKK 8,196m at 31 December 2025 (DKK 7,484m at 31 December 2024).
The assessment of fair value of unlisted investments is subject to considerable uncertainty. This applies especially to life science investments because the value of these businesses is linked to the companies’ often long-term investment in the development of new pharmaceuticals and technologies. Please refer to note 32.3 for information about applied valuation methods for the determination of fair value.
Credit risks relating to the Group’s financial investments primarily relate to investment in bonds and unlisted funds investing in loans to businesses.
To limit the credit risk, a proportion of this asset class has been invested in Danish government and mortgage bonds with a high credit rating. To achieve a higher return, the Group also invests in corporate bonds.
Equity risks relate to the Group’s holding of listed and unlisted shares, including private equity funds, as part of the Group’s investment operations. Most of these investments are placed in listed shares.
To limit the risk of losses on these shares, the investments are diversified across different geographical regions and sectors in accordance with the applicable investment policy. Derivative financial instruments are used to manage the equity risk. The instruments can be used both for risk management purposes and as an alternative to selling or buying the underlying assets.
Other things being equal, a 10% decrease/increase in equity prices would decrease/increase profits after tax by DKK 1,619m and DKK 1,573m respectively (decrease by DKK 1,430m and increase by DKK 1,372m in 2024).
For further information on risks relating to the Group’s financial investments, see note 21 Liquidity and note 32 Financial risks and financial instruments.
Financial assets included in Invest and BioCapital are part of the Group’s investment strategy and measured at fair value through profit or loss. Other receivables are measured at amortised cost. At 31 December 2025, investments in associates included in Lundbeckfonden’s investment strategy amounted to DKK 2,799m (DKK 3 047m at 31 December 2024).
Please refer to note 32 for information about exchange rate and interest rate risks.
Note 17
Deferred tax - continued
Deferred tax
Temporary differences between the carrying amount and the tax base:
Management estimates future income according to budgets, forecasts, business plans and initiatives scheduled for the coming years, which support the recognition of deferred tax assets. When forecasting the utilisation of tax assets, the Group applies the same assumptions as for impairment testing.
At 31 December 2025, all deferred tax assets relating to tax losses carried forward in Denmark were fully utilised (DKK 452 at 31 December 2024)
U.S. tax losses and tax credits stemming from acquisitions have been recognised at an amount of DKK 814m (DKK 486m at 31 December 2024), equalling the expected utilisation within a foreseeable future, whereas an amount of DKK 26m (DKK 20m at 31 December 2024) has not been recognised in the balance sheet.
Unrecognised deferred tax assets primarily relate to net operating losses and tax credits not expected to be utilised. within the foreseeable future. The majority of the tax losses have no expiry date.
- continued
At 31 December 2025, write-downs consist of DKK 151m for Vyepti® obsolescence and DKK 169m associated with the planned divestment of a non-core production site in Italy, as disclosed in note 7 Other operation items, net. In 2025, following changes in accounting estimates, reversals of previous write-downs related to Vyepti® of DKK 389m were recognised in cost of sales.
Inventories of DKK 2,627m (DKK 2,506m at 31 December 2024) are expected to be recovered after more than 12 months, mostly due to the Vyepti® fixed batch quantity supply agreement, which ended in 2023.
The Group’s products are sold primarily to distributors of pharmaceuticals, pharmacies and hospitals. Services are sold to public authorities, other large customers and small individual customers.
No single customer contributed 10% or more to total revenue. The Group has no significant reliance on specific customers. Internal procedures for evaluating
specific credit risks from new customer relationships, and changes to the risk profile of existing relationships, ensure that the risk of loss is reduced to an acceptable level.
Securities are classified as financial assets measured at fair value through profit or loss.
With the present capital structure, the Group is wellconsolidated. The Group aims to retain adequate cash resources to support business development and flexibility in case of changes to the market situation, potential acquisition activities and product in-licensing opportunities. This is achieved through a combination of liquidity management, ultra-liquid assets and guaranteed and unguaranteed credit facilities. The capital structure is considered appropriate relative to the Group's strategic plans.
The credit risk of cash, bank balances and derivatives (forward exchange contracts, currency options, interestrate options and share options) is limited because the Group only deals with banks with a high credit rating. To further limit the risk of loss, internal limits have been defined for the credit exposure accepted towards the banks with which the Group collaborates. Pursuant to the Group’s policies, the credit lines are presented to the Board
groups, for approval. Furthermore, the Group
maintain counterparty diversification to avoid material concentration at individual counterparties. The Group also uses collateral agreements (e.g., International Swaps and Derivatives Association (ISDA) and Global Master Repurchase Agreement (GRMA)) and exchange of collateral with counterparties with which the Group has hedging business.
The present statutes of Lundbeckfonden were approved by the Board of Directors on 12 March 2025. The Danish Business Authority acts as supervisory authority.
Of the Foundation’s profit before tax less nondistributed earnings in the subsidiaries and associates, at least 20% must first be allocated to the capital base.
23
Currency translation of foreign subsidiaries and currency translation concerning additions to net investments in foreign subsidiaries and tax related to these items amounted to a net loss of DKK 2,884m (net profit of DKK 826m in 2024), and are recognised in the currency translation reserve in equity. Other items and tax related to such items are recognised in reserve for hedging transactions at a net profit of DKK 279m (net loss of DKK 272m in 2024).
The minority shareholder’s share of goodwill in acquired businesses in Falck is recognised in the consolidated financial statements. However, goodwill of DKK 1,791m (DKK 1,791m at 31 December 2024) arising at the time of the Foundation’s acquisition of Falck is not recognised in the consolidated financial statements.
Lundbeckfonden's subsidiaries with significant noncontrolling interests include the following:
- continued
25.1 Pensions and similar obligations
Defined contribution plans
In defined contribution plans, the employer is obliged to pay a certain contribution to a pension fund or the like but bears no risks regarding the future development in interest, inflation, mortality, disability rates, etc., regarding the amount to be paid to the employee.
The cost of defined contribution plans, representing contributions to the plans, amounted to DKK 846m in 2025 (DKK 783m in 2024).
The defined benefit plans guarantee employees a certain level of pension benefits for life. The pension is based on seniority and salary at the time of retirement. The Group bears the risks regarding the future development in interest, inflation, mortality, disability rates, etc. regarding the amount to be paid to the employee.
The Group has defined benefit plans in a few countries. The most important plans comprise current and former employees in France, Germany, Switzerland and the UK.
The following were the key actuarial assumptions at the reporting date.
Pensions and similar obligations – continued
Assumptions regarding future longevity are set based on actuarial advice in accordance with published statistics and experience in each country. The longevities underlying the values of the defined benefit obligation for the most significant plans were as follows:
Assumed life expectations on retirement age for current pensioners
The most significant assumptions used in the calculation of the obligation for defined benefit plans are discount rate, inflation rate and assumed life expectations on retirement age for current pensioners. An increase in the discount rate of 0.25 of a percentage point would result in a decrease in the obligation of approximately DKK 19m, before tax (DKK 20m at 31 December 2024) and vice versa. An increase in the inflation rate of 0.25 of a percentage point would result
in a decrease in the obligation of approximately DKK 1m, before tax (DKK 2m at 31 December 2024) and vice versa. An increase in the life expectancy of 1 year would result in an increase in the obligation of approximately DKK 20m, before tax (DKK 20m at 31 December 2024) and vice versa. The sensitivity analysis indicates how a change in the individual assumptions would change the obligation. However, the assumptions will most likely be correlated and consequently result in a different obligation.
Assumed life expectations on retirement age for current employees (future pensioners)
Shares and bonds are measured at fair value based on quoted prices in an active market. Insurance contracts and other assets are not based on quoted prices in an active market.
Pensions and similar obligations – continued
The expected contribution for 2025 for the defined benefit plans was DKK 33m (DKK 25m in 2025).
An obligation at 31 December 2025 of DKK 91m (DKK 109m at 31 December 2024) was recognised to cover other obligations of a retirement benefit nature, which primarily include post-employment benefits in a number of subsidiaries. The benefit payments are conditional upon specified requirements being met.
be specified as follows:
Discounts and rebates
The most significant sales deductions are in the U S and comprise discounts and rebates given in connection with sales under U S Federal and State Government Healthcare programmes, primarily Medicaid.
Management’s estimate of discounts and rebates is based on a calculation which includes a combination of historical product/population utilisation mix, price increases, program/market growth and state-specific information. Further, the calculation of rebates involves legal interpretation of relevant regulations and is subject to changes in interpretive guidance from governmental authorities. The obligations for discounts and rebates are incurred at the time the sale is recorded; however, the actual rebate related to a specific
sale may be invoiced by the authorities six to nine months later. In addition to this billing time lag, there is no statute of limitations for states to submit rebate claims; thus, rebate adjustments in any particular period may relate to sales from a prior period. Moreover, when a product loses exclusivity, shifts in payer mix may cause Medicaid claims/estimates to be more volatile.
At 31 December 2025, other comprised liabilities for legal disputes DKK 642m (DKK 423m at 31 December 2024), restructuring programmes DKK 259m (DKK 288m at 31 December 2024), long-term incentive programmes in USA DKK 55m (DKK 77m at 31 December 2024) and provisions from acquisition DKK 38m (DKK 94m at 31 December 2024).
Fair value was calculated by applying the market value of the underlying bonds at 31 December and therefore measured by level 1 input.
26.2 Bank debt and bond debt, etc. DKKm
Bank debt and bond debt by maturity:
Falck
Falck’s primary debt financing is a syndicated bank loan facility of EUR 310m, expiring in January 2029, and a DKK 300m term loan with Nordic Investment Bank where proceeds were dedicated to investments in digitalisation.
In December 2025, Falck entered a EUR 75m term loan with Nordea dedicated to finance investments in Falck’s operations in Spain. During 2025, Falck made a voluntary repayment of mortgage loans amounting to DKK 254m. Partly refinancing corresponding to DKK 133m was made.
Lundbeck
In 2019, Lundbeck entered into a revolving credit facility (RCF) of EUR 1.5bn with its strategic banks. The RCF was extended in 2025 by one additional year and now expires in 2027. The flexible structure of the RCF enables Lundbeck to repay the debt in full at short notice, normally not more than three months, while still maintaining the facility until expiration of the credit commitment.
To hedge the risk of the drawdown on the RCF, Lundbeck swapped EUR funding into DKK by a crosscurrency swap with an amortised profile matching the expected payback profile of the underlying loan. The floating DKK debt amount has been swapped into fixed interest by an interest rate swap with the same amortised profile and an average fixed interest rate of 2.20%.
In October 2020, Lundbeck issued a seven-year Eurobond in the amount of EUR 500m with a fixed coupon of 0.875%. During 2025, Lundbeck swapped the EUR debt into DKK via a cross-currency swap (fixed/fixed), providing a lower fixed interest rate of 0.689%.
In June 2025, Lundbeck issued a four-year Eurobond in the amount of
with a
coupon of 3.375%.
The net proceeds from the bond were used to refinance a EUR 500m bridge facility that was established in October 2024 in connection with the acquisition of Longboard Pharmaceuticals. Hence, the bond issuance was considered to be leverage-neutral. Both bonds have been issued under Lundbeck’s Euro medium-term note (EMTN) programme of EUR 2bn and are listed on Euronext Dublin.
Lease liabilities
In 2025 the Group paid DKK 561m (DKK 612m in 2024) for lease agreements, of which, interest expenses amounted to DKK 61m (DKK 61m in 2024). See also note 13 Right-of-use assets.
26.4 Development in mortgage debt, bank debt and bond debt, etc.
break down as follows:
At 31 December 2025 other payables included debt related to salaries and holiday payment of DKK 2,096m (DKK 2,056m at 31 December 2024), VAT and other taxes of DKK 559m (DKK 634m at 31 December 2024) and discounts and rebates of DKK 959m (DKK 888m at 31 December 2024).
Contingent consideration recognised through acquisitions
As part of the acquisition of Alder BioPharmaceuticals, Inc. (subsequently renamed Lundbeck Seattle BioPharmaceuticals, Inc.), Lundbeck has recognised a contingent consideration liability related to sales milestones dependent on predefined milestones being reached. At 31 December 2025, the fair value of this contingent consideration related to this acquisition amounted to DKK 357m (DKK 306m at 31 December 2024).
As part of the acquisition of Abide Therapeutics, Inc. (subsequently renamed Lundbeck La Jolla Research Center, Inc.), Lundbeck has recognised a contingent consideration liability related to sales milestones
dependent on pre-defined milestones being reached. At 31 December 2025, the fair value of this contingent consideration related to this acquisition amounted to DKK 30m (DKK 33m at 31 December 2024).
Contingent considerations are recognised at fair value.
The calculation of the fair value is based on the discounted cash flow method (DCF method), which comprises significant assumptions and estimates. Expected timing of payment (using a specific discount rate) and probability of success are key inputs to the fair value of the contingent considerations.
The fair value adjustment of all contingent considerations amounted to a net loss of DKK 89m (net gain of DKK 21m in 2024), comprising DKK 90m (DKK 39m in 2024) of financial expenses and DKK 1m (DKK 60m in 2024) of financial income. The liability was impacted by favourable exchange variations of DKK 41m (DKK 23m at 31 December 2024).
The amounts in the table represent the purchase price allocation to the identifiable assets and liabilities, and consequently also to goodwill at the acquisition date.
Purchase price of DKK 2m related to Falck, and paid contingent considerations of DKK 10m related to the ALK’s acquisition in 2024
On 30 December 2025, Falck signed an agreement to acquire all membership interest (LLC-units) in the American company The Morin Group LLC in Texas. The acquisition was completed by payment of the agreed consideration of DKK 2m on 31 December 2025. No additional deferred considerations or contingent payments has been agreed.
The purpose behind the acquisition was to secure a Texas Emergency Medical Service Provider License covering the entire state, and this move supports Falck’s strategy of expanding its presence into new states within the U.S.
Except for a minor bank account holding less than DKK 1m, The Morin Group LLC had no net assets. The total consideration was attributed to the license, identified as an intangible asset, with no goodwill recognised other than the deferred tax asset tied to the license. The recognised goodwill was not tax deductible.
No transaction costs were incurred related to the acquisition.
Net assets, goodwill, contingent assets and liabilities recognised at the reporting date are to some extend still provisional. Adjustments can be made for at period of 12 months from the acquisition date
On 2 December 2024, Lundbeck completed the acquisition of Longboard Pharmaceuticals, Inc. (herein denominated ’Longboard’), a U.S.-listed entity, at a total purchase consideration of USD 2.35bn fully paid (DKK
16.6bn), on a fully diluted basis. Lundbeck obtained control of Longboard by acquiring 39,168,546 of issued shares, representing 100% of Longboard’s share capital. Under the terms of the agreement, Lundbeck paid an amount of USD 60 per share.
Longboard is a clinical-stage biopharmaceutical company focused on developing novel, transformative medicines for neurological diseases. Its lead asset, bexicaserin, has shown encouraging anti-seizure reduction to date in preclinical and clinical studies, with its next-generation superagonist mechanism specifically targeting 5-HT2C receptors, which supports bexicaserin’s potential to offer a highly differentiated and best-inclass profile. Bexicaserin is now being evaluated in a global Phase III clinical programme (the DEEp programme).
The acquisition of Longboard marked a strategic milestone for Lundbeck, enhancing and complementing our Focused Innovator Strategy and advancing our goal of building a neuro-rare disease franchise.
Through the acquisition of Longboard, Lundbeck gains access to bexicaserin, a novel 5-HT2C agonist in development for the treatment of seizures associated with developmental and epileptic encephalopathies (DEEs), including Dravet syndrome, Lennox-Gastaut syndrome, and other rare epilepsies. This aligns with Lundbeck’s expertise in delivering innovative treatments and re-establishes our scientific and commercial leadership in rare epilepsies. Bexicaserin has entered a global Phase III trial (DEEp SEA program) evaluating bexicaserin for the treatment of seizures associated with Dravet syndrome in participants two years of age and older. The DEEp SEA study is part of a broader DEEp
31.1 Acquisitions of businesses, etc. – continued programme (DEEp SEA, DEEp OCEAN, and DEEp OLE), which is planned to take place across approximately 80 sites globally and include approximately 480 participants with a range of DEEs. Bexicaserin has received a breakthrough therapy designation (BTD) from the U.S. FDA and is set to become a cornerstone of Lundbeck’s new neurorare disease franchise. Recent nine-month open-label data further supports the derisked nature of its 5-HT2C mode of action, highlighting its superior target product profile.
As a result of the acquisition, Longboard has become a wholly owned subsidiary of Lundbeck, and the common stock of Longboard has been delisted from the NASDAQ Global Market.
The net assets recognised in the 2024 Financial Statements were based on a provisional assessment of the fair value of the net assets acquired. During 2025, Lundbeck finalised the purchase price allocation in accordance with IFRS 3, including the identification and measurement of acquired intangible assets, assumed liabilities, and related deferred tax effects. The identifiable assets acquired and liabilities assumed are presented in the table to the right. The table sets out the final amounts of net assets acquired and goodwill recognised at the acquisition date.
During the measurement period, the provisional amounts recognised in 2024 were updated based on new information obtained about facts and circumstances that existed at the acquisition date. These updates primarily related to the final valuation of
intangible assets and the associated deferred tax liability. Consequently, the final fair values were determined to be DKK 19,255m for intangible assets, DKK 4,621m for deferred tax liabilities, and DKK 1,994m for goodwill. The 2024 comparative figures have been restated to reflect these measurement period adjustments.
The intangible assets recognised by the Group comprise product rights of DKK 18,729m and know-how of DKK
526m. The valuation methods for measuring the fair value of the acquired intangible assets were as follows:
• Product right (Bexicaserin): The fair value of the Bexicaserin product right is determined using an income-based multi-period excess earnings method (MEEM) approach, reflecting the total expected revenues from Bexicaserin sales and the product’s estimated economic life
• Know-how: The fair value of know-how is determined using the cost method, reflecting the estimated cost to reproduce or replace the asset at current market prices
Goodwill represents the value of the acquired workforce and the expected synergies arising from the acquisition. None of the goodwill is expected to be deductible for income tax purposes.
On 2 January 2024, the ALK Group acquired the operating assets of AllerQuest for a total cash consideration of DKK 125m. The consideration amount included an escrow amount of DKK 10m which served as reserve for potential indemnifications over 18 months from acquisition date.
AllerQuest was a U.S.-based company dedicated to manufacturing PRE-PEN® Skin Antigen Test. This acquisition makes ALK the sole manufacturer and distributor of PRE-PEN in the U.S. and Canada, with global ownership rights to all assets of AllerQuest. PREPEN is the only FDA-approved diagnostic skin test for the evaluation of penicillin allergy and is indicated for the assessment of sensitisation to penicillin in patients suspected to have clinical penicillin hypersensitivity.
AllerQuest was previously a supplier of the ALK Group and integration was completed in 2024.
The transaction was on a debt and cash free basis. No liabilities were transferred.
In June 2024, Falck signed an agreement to acquire all shares in two Colombian companies: Asistencia Médica S.A.S. and Servicio de Asistencia Médica Inmediata S.A.S. The acquisition was completed by payment of the agreed consideration on 14 June 2024, from which point Falck assumed control.
The consideration paid on the closing date amounted to DKK 26m and an additional amount of DKK 18m was paid in Q4 2024. An additional deferred payment of DKK 3m was agreed bringing the total consideration to DKK 47m. Adjusted for cash of DKK 4m, the net consideration amounted to DKK 43m. The deferred consideration did not depend on the receivers’ continued employment in Falck, which means that this part of the consideration was included in the total consideration amount. No further additional contingent consideration was agreed.
Transaction costs related to the acquisition amounted to DKK 1m, which was included in other operating items, net.
Besides the value of customer contracts amounting to DKK 7m, no assets or liabilities were identified which were not recognised in the companies acquired on the date of acquisition. Goodwill of DKK 21m was calculated. The recognised goodwill was not tax deductible.
31.1 Acquisitions of businesses, etc. – continued
Goodwill mainly represents the value of operational synergies from the integration of the acquired companies into the existing operation in Colombia as well as the strategic leverage for the B2I segment in Colombia.
During 2025, the provisional Purchase Price Allocation has been completed. Final consideration and opening balance were not adjusted and no other adjustments were applied to the provisional amounts.
In 2024, Falck divested the Industrial Fire services company in Brazil and an ambulance activity in Cuxhaven, Germany.
An amount of DKK 4m of exchange rate adjustments from divestments was recirculated from the currency translation reserve.
Adjustments amounting to DKK 14m from previous years' divestments mainly related to the write-off of aged receivables from companies that had been divested in prior years. Furthermore, adjustments concerning costs related to disputes which were settled.
The total loss from divestment of enterprises of DKK 15m was recognised in the income statement in financial expenses
The Group’s business activities imply that the results and balance sheet may be affected by various financial risks. The management of these risks is decentralised and handled in Lundbeckfonden and in Lundbeck, ALK and Falck based on policies and guidelines approved by the Board of Directors or the boards of directors in the subsidiaries.
See also note 16 Financial assets, note 19 Trade receivables, and note 21 Liquidity for descriptions of risks and the management thereof and note 32.5 for information about derivative financial instruments.
32.1 Exchange rate risks
Exchange rate risks arise because the Group’s expenses and income in different currencies do not match and because the Group’s assets and liabilities denominated in foreign currency do not balance, among other things, due to BioCapital’s and Invest's investment assets. The management of these risks is focused on risk mitigation.
The Group applies various derivative financial instruments to manage these risks. Some of these instruments are classified as hedging instruments and meet the accounting criteria for hedging future cash flows. Changes in the fair value of these contracts are recognised in the statement of comprehensive income under other comprehensive income as they arise and, on invoicing of the hedged cash flow, transferred from other comprehensive income for inclusion in the same item as the hedged cash flow. Hedging contracts that do not meet the hedge criteria are classified as trading contracts, and changes in the fair value are recognised
as financial items as they arise. The need for hedging is assessed separately in Lundbeck, ALK and Falck and in Invest.
Estimated impact from financial instruments on profit for the year and equity from a 5% increase in year-end exchange rates of the major currencies:
are located. The profit impact is limited as the largest liabilities are exchange rate adjusted in other comprehensive income, being part of the Group’s hedging structure.
The equity impact includes financial instruments that remained open at the balance sheet date and which are exchange rate adjusted in other comprehensive income. The equity effects in 2025 and 2024 primarily consist of exchange-rate adjustments in USD on outstanding cash flow hedging contracts.
Due to Denmark’s long-standing fixed exchange rate policy against the euro and the expected continuation of this policy, the foreign currency risk for the euro is considered immaterial, and the euro is therefore not included in the table.
32.2 Interest rate risks
The shown sensitivities only comprise impact from financial instruments and reflect a relative change of the exchange rates at 31 December 2025 and 2024.
The sensitivity analysis includes derivatives, bank loans, trade receivables, trade payables, intercompany lending and borrowing, as those are the financial instruments where the Group has the most currency exposure.
The profit impact comprises financial instruments that remained open at the balance sheet date and which have an impact on profit in the current financial year. It includes foreign exchange differences relating to intragroup balances that are not eliminated in the consolidated financial statements. The calculation of the estimated impact is based on the functional currency of the entities where the financial instruments
Interest rate risk relates to the Group’s interest-bearing assets and liabilities and, principally, to the Group’s bonds classified as financial assets measured at fair value through profit or loss. See note 26 Bank debt, bond debt and borrowings and note 32.3 Fair value hierarchy for financial assets and liabilities, measured at fair value.
The duration of the investments when selecting financing and investment instruments is used to manage the interest rate risk. In addition, the Group uses derivative financial instruments to mitigate the interest rate exposure. The use of financial instruments to manage interest rate risk does not qualify for hedge
accounting, and the changes in fair value are therefore recognised as financial income or expenses in an ongoing process.
At 31 December 2025, the Group’s portfolio of bonds had a duration of 2.88 years (2.41 years at 31 December 2024). Other things being equal, an increase of 1 percentage point in interest rates would increase the Group’s profit after tax by DKK 92 (increase of DKK 104m at 31 December 2024).
At 31 December 2025 and 2024, there were no derivatives to manage interest rate risks because the distribution of investments carrying floating and fixed interest at the given times was deemed to be satisfactory.
Lundbeck’s exposure to interest rate risk is low, as both outstanding bonds have fixed coupon rates and the drawdown on the revolving credit facility is swapped into fixed interest rates. For more information see note 26.2.
Falck’s exposure to interest rate risk relates to the part of the mortgage loans which carry a floating interest rate, and the bank debt of DKK 2,530m, which carries variable interest.
ALK does not hedge its interest rate exposure, as this is not considered to be financially viable.
32.2 Interest rate risks - continued
An interest rate change on bank debt and bond debt, including interest rate swaps, of +/-1 percentage point would decrease/increase profit for the year after tax by DKK 30m (DKK 65m in 2024) and increase/decrease equity by DKK 49m in 2025 (DKK 61m at 31 December 2024) on an annual basis.
32.3 Fair value hierarchy for financial assets and liabilities, measured at fair value
Level 1 includes financial assets for which the fair value is measured based on quoted prices (unadjusted) in active markets for identical assets. Level 2 includes financial assets and financial liabilities for which the fair value is measured based on directly or indirectly observable inputs other than the quoted prices included in level 1. Level 3 includes financial assets for which the fair value is measured based on valuation methods which include inputs not based on observable market data.
The requirement for reclassifications between the levels is evaluated continually during the year. For individual financial assets and liabilities, it is evaluated whether the most critical input variable in connection with determination of fair value has changed from unobservable to observable or the other way round. If this is the case the asset or liability is reclassified from the recent relevant level to new level from the time when the change in input variable occurs.
Level 3 liabilities are determined on the basis of profit prior to the right being exercised, multiplied by an already agreed multiple, typically less net debt in the relevant companies. When recognised in the balance sheet, this liability is made up on the basis of earnings and net debt at the time when the non-controlling interests are expected to exercise their right to sell their shares to the Group. The calculated liability typically assumes an increase in earnings and a decrease in net debt in the relevant companies as compared with the value recognised in the financial statements.
The fair value of derivatives is determined by applying recognised measurement techniques, whereby assumptions are based on the market conditions prevailing on the balance sheet date.
The fair value of contingent consideration is calculated as the discounted cash outflows from future milestone payments, taking probability of success into consideration.
Applied valuation methods for the determination of fair value of the material categories above are as follows:
Note 33
Contractual obligations, contingent assets and liabilities, and collaterals - continued
The Group is involved in pending legal proceedings arising out of the normal conduct of its business. While provisions that Management deems to be reasonable and appropriate have been made for probable losses, there are inherent uncertainties connected with these estimates
In June 2013, Lundbeck received the European Commission’s decision that agreements concluded with four generic competitors concerning citalopram violated competition law. The decision included fining Lundbeck EUR 93.8m (approximately DKK 700m). Lundbeck paid and expensed the fine in the third quarter of 2013. In March 2021, the European Court of Justice rejected Lundbeck’s final appeal of the European Commission’s decision. So-called ’follow-on claims’ for reimbursement of alleged losses resulting from violation of competition law often arise when decisions and fines issued by the European Commission are upheld by the European Court of Justice. The below-mentioned ’follow-on claims’ are ongoing or threatened. Lundbeck disagrees with all claims and intends to defend itself against them.
At the end of the first quarter of 2023, the UK health authorities served their claim form on Lundbeck and several generic companies, and Lundbeck filed its
defence in the third quarter of 2023. The hearing on whether the claim is time-barred was held in the second quarter of 2024, and the Competition Appeal Tribunal subsequently issued a decision in favour of the UK health authorities. Lundbeck was granted permission to appeal the decision to the Court of Appeal, and the Court of Appeal issued a decision in favour of the UK health authorities in the second quarter of 2025. In October 2025, the Supreme Court refused Lundbeck’s application for permission to appeal the ruling on timebarring.
In late October 2021, Lundbeck received a writ of summons from a German healthcare company claiming compensation for an alleged loss of profit plus interest payments, allegedly resulting from Lundbeck’s conclusion of agreements with two of the four generic competitors, which were covered by the EU Court of Justice ruling. Lundbeck filed its first defence in May 2022, and the parties have subsequently exchanged additional pleadings. The first instance court hearing was held in the second quarter of 2024, and Lundbeck currently expects a first instance court ruling in 2026. The first instance court ruling may be appealed, and it may take several years before a final conclusion is reached by the German courts.
Note 33
Contractual obligations, contingent assets and liabilities, and collaterals - continued
In October 2024, Lundbeck received a claim form from the health authority in one of the regions (comunidades autónomas) in Spain, and in November 2024 Lundbeck filed its defence. The first instance court hearing was held in the second quarter of 2025, and a first instance ruling was issued in the third quarter of 2025. The court dismissed the health authority’s claim based on timebarring. The health authority has appealed the decision.
Lundbeck has been informed about potential claims in several European countries, however, it is still uncertain whether the potential claims will be actively pursued.
In Canada, Lundbeck is involved in a product liability class-action lawsuit against several Selective Serotonin Reuptake Inhibitor (SSRI) manufacturers (including Lundbeck), alleging that SSRIs (Celexa®/Cipralex®) induce autism birth defects. Lundbeck strongly disagrees with the claim. Other product liability claims concerning Rexulti®, Abilify Maintena®, and Celexa® have all been closed.
Otsuka and Lundbeck received paragraph IV certifications from Sun Pharma, Apotex, and Alvogen with respect to certain patents listed for Abilify Maintena® in the U.S. and commenced patent infringement proceedings against all three companies. The case with Sun Pharma has now been settled, whereas the cases against Apotex and Alvogen are scheduled for trial in October 2026. The FDA will stay approval to Apotex and Alvogen until 30 months from receipt of the respective paragraph IV certifications or a court decision in Apotex’s or Alvogen’s favour.
Lundbeck received a civil investigative demand (’CID’) from the U.S. Department of Justice (’DOJ’) in March
2020. The CID seeks information regarding the sales, marketing, and promotion (including the promotional speaker program) of Trintellix®. Lundbeck is cooperating with the DOJ.
In June 2022 in the U.S., several entities created for the purpose of receiving assignment of claims from payors providing health insurance coverage pursuant to Medicare Parts C and D and Medicaid filed a complaint against Lundbeck and others. The complaint alleged that Lundbeck and the other defendants conspired to increase the unit price and quantity dispensed of Xenazine®. The case was dismissed with prejudice in 2023, all appeals have been exhausted, and the case is closed.
In June 2023 in the U.S., Humana Inc., an insurer, filed a complaint against Lundbeck U.S. legal entities. The complaint alleges that Lundbeck engaged in an illegal kickback scheme to increase the sales and sale price of Lundbeck’s Xenazine®. The complaint alleges that Lundbeck’s activities targeted Humana Inc. and other private Medicare insurers, who were forced to bear the costs of the alleged illegally subsidised drug sales. Lundbeck denies the allegations in the complaint and intends to defend itself.
The Group has entered into a number of agreements relating to research and development of new products and intellectual property rights from acquisitions, as well as other collaborations. Under the agreements, Lundbeck is committed to paying for the research and development services provided by third parties. The obligation amounts to DKK 2,979m. Further, Lundbeck is
committed to paying certain milestones related to achieving different research, development, and regulatory milestones. Such amounts entail uncertainties in relation to the period in which payments are due because a proportion of the obligations is dependent on the milestone achievements.
At 31 December 2025, potential future milestone payments amounted to DKK 2,883m (DKK 770m at 31 December 2024).
The Group is committed to paying certain commercial sales milestones, royalties, or other payments based on a percentage of sales generated from the sale of goods following marketing approval. These amounts are excluded from the contractual obligations because of their contingent nature, being dependent on future sales
The Group has product return obligations that are normal for the industry. Management does not expect any major losses from these obligations apart from the amount already recognised.
Usual representations and warranties are made in connection with the divestment of enterprises and operations. There are no significant outstanding claims that are not sufficiently recognised in the balance sheet.
The Group has undertaken purchase obligations relating to property, plant and equipment in the amount of DKK 444m (DKK 637m at 31 December 2024). Contractual obligations for intangible assets, excluding
commitments with R&D milestones and collaborations, amounted to DKK 5m (DKK 24m at 31 December 2024), and other obligations relating to licensing agreements and lease obligations for signed but not yet commenced lease agreements as per commencement date in accordance with IFRS 16 amounted to DKK 25m at 31 December 2025 (DKK 7m at 31 December 2024).
The contractual obligations not recognised in the Balance sheet represent contractual payments and are neither discounted nor risk-adjusted.
PFAS pollution has been identified at Lundbeck’s site in Lumsås. Pollution to the soil and water has occurred from the use of PFAS-containing firefighting foam in the factory’s fire extinguishing system, which was used until 2011 in compliance with applicable law and following recommendations by the fire authorities at that time. The case is being managed in accordance with the requirements set by the authorities and in line with Lundbeck’s HSE, Compliance, and Sustainability policies. Since the pollution was detected, Lundbeck has been engaged in a close dialogue with the Danish Environmental Protection Agency (EPA) regarding the mapping and remediation of the pollution. Lundbeck has proactively taken steps to reduce PFAS levels while continuously engaging with neighbours and the municipality to address concerns in the local community. As remediation and mitigation action advances, our knowledge and understanding of PFAS pollution will continue to evolve, and we are committed to continuing to take further steps to reduce PFAS levels in the area.
Note 33
Note 34
Related parties - continued
obligations, contingent assets and liabilities, and collaterals - continued
Falck has received information requests from municipalities, and might in the future be involved in further investigations regarding PFOS contamination in the portfolio of properties owned by Falck. Falck does not consider it probable that the investigations will result in a liability for Falck.
Land and buildings provided as security for mortgage debt amounted to DKK 714m (DKK 562m at 31 December 2024) out of mortgage debt of DKK 367m (DKK 518m at 31 December 2024).
Lundbeckfonden is an enterprise foundation established by Grete Lundbeck in 1954. As a foundation, no party controls Lundbeckfonden.
Related parties to Lundbeckfonden:
• The Foundation’s Executive Management and Board of Directors, including their close family members and related undertakings
• Associates
See note 3 Employee costs for information about remuneration received by the Board of Directors and the Executive Management.
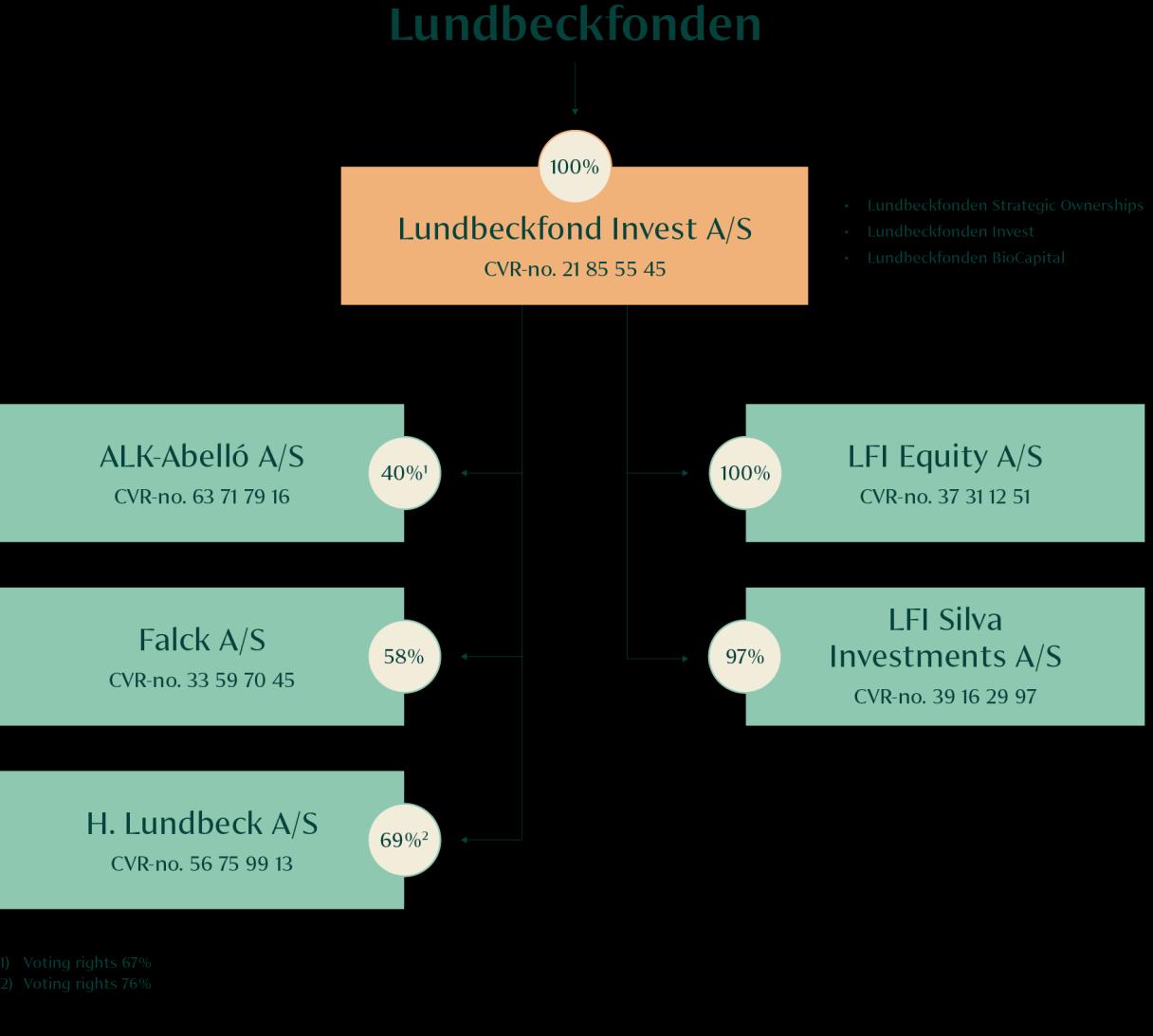
Subsidiaries of H. Lundbeck A/S
Lundbeck Argentina S.A.
Lundbeck Australia Pty Ltd, including
- CNS Pharma Pty Ltd
Lundbeck Austria GmbH
Lundbeck S.A.
Lundbeck Brasil Ltda.
Lundbeck Canada Inc.
Lundbeck Chile Farmacéutica Ltda.
Lundbeck (Beijing) Pharmaceuticals Consulting
Lundbeck Colombia S.A.S.
Lundbeck Croatia d.o.o.
Lundbeck Czech Republic s.r.o.
Lundbeck Export A/S
Lundbeck Pharma A/S
Lundbeck Eesti A/S
OY H. Lundbeck
Lundbeck
Sofipharm SAS,
Elaiapharm
Lundbeck GmbH
Lundbeck Hellas S.A.
Lundbeck HK Limited
Lundbeck Hungária KFT
Lundbeck (Ireland) Ltd.
Lundbeck Israel Ltd.
Lundbeck Italia S.p.A.
Lundbeck Pharmaceuticals, Italy S.p.A.,
- Archid S.A.
Lundbeck Japan K.K.
Lundbeck
SIA Lundbeck
UAB Lundbeck Lietuva
Lundbeck Malaysia SDN. BHD.
México, SA de CV
Prexton Therapeutics B.V., including
H. Lundbeck AS
Lundbeck Pakistan (Private) Limited
Lundbeck America Central S.A.
Lundbeck Business Service Centre Sp.z.o.o.
Lundbeck Poland Sp.z.o.o.
Lundbeck Portugal - Produtos Farmacêuticos Unipessoal Lda
Lundbeck Romania SRL
Lundbeck RUS LLC
Lundbeck Regional Headquarters
Lundbeck Singapore PTE. LTD.
Lundbeck Slovensko s.r.o.
Lundbeck South Africa (Pty) Limited, including
- H. Lundbeck (Proprietary) Limited
Lundbeck España S.A.
H. Lundbeck AB
Lundbeck (Schweiz) AG
Lundbeck İlaç Ticaret Limited Şirketi
Lundbeck Group Ltd. (Holding), including
- Lundbeck Limited
- Lundbeck Pharmaceuticals Ltd.
- Lifehealth
- Lundbeck UK LLP b)
Lundbeck USA Holding LLC, including
- Lundbeck LLC, including
- Chelsea Therapeutics International, Ltd., including
- Lundbeck NA Ltd.
- Lundbeck Pharmaceuticals LLC
- Lundbeck Research USA, Inc.
- Lundbeck La Jolla Research Center, Inc., including
- Abide Therapeutics (UK) Limited
- Lundbeck Seattle BioPharmaceuticals, Inc., including
- Alder Biopharmaceuticals Pty., Ltd.
- Alderbio Holdings LLC ("ANEV")
- Longboard Pharmaceuticals, Inc.
Lundbeck de Venezuela, C.A.
Subsidiaries of ALK-Abelló A/S Europe
ALK-Abelló Allergie-Service GmbH
ALK-Abelló Nordic A/S
S.A.S.
ALK-Abelló Arzneimittel GmbH
ALK-Abelló B.V.
ALK-Abelló Sp. z o.o.
ALK Slovakia s.r.o.
ALK-Abelló S.A., including
- ALK-Abelló S.p.A.
ALK AG (in liquidation)
ALK-Abelló AG
ALK-Abelló Ltd.
ALK-Abelló Pharmaceuticals, Inc.
ALK-Abelló, Inc., including
- OKC Allergy Supplies, Inc.
ALK-Abelló Source Materials, Inc., including
ALK (Shanghai) Medical Technology Co., Ltd., including
- ALK (Guangzhou) Medical Technology Co., Ltd.
Tasfiye Halinde ALK Ilac ve Alerji Ürünleri Ticaret Anonim Sirketi (in liquidation)
Subsidiaries of Falck A/S
Falck (Victoria) Pty. Ltd.
Falck Pty. Ltd.
Falck Fire Services BE NV
Falck Brasil 747 Participações Ltda.
Falck Global Assistance (China) Limited
Empresa de Medicina Integral EMI S.A. Servicio de Ambulancia Prepagada – Grupo
Servicios Logisticos S.A.S.
Haces Inversiones y Servicios S.A.S
Asistencia Médica S.A.S. Prepagado
Servicio de Asistencia Médica Immediata S.A.S.
Falck Danmark A/S
Falck Digital Technology Denmark A/S
Falck Global Assistance A/S
Falck Healthcare A/S
EMI Ecuador S.A.- Emergencia Medica Integral
EMI El Salvador S.A. de C.V.
Falck Global Assistance Oy
Falck France SAS
Falck Arbeitsgemeinschaft Rettungsdienst Plauen GmbH & Co. OHG
Falck Deutschland GmbH
Falck Fire Services DE GmbH
Falck Notfallrettung und Katastrophenschutz gGmbH
Falck Notfallrettung und Krankentransport Dortmund GmbH
Falck Notfallrettung und Krankentransport GmbH
Falck Operations Services DE GmbH
Falck Rettungsdienst Hanse GmbH
Falck Rettungsdienst Nord GmbH
Falck India Pvt. Ltd. (India)
Falck Servizi Industriali di Emergenza S.r.l.
Falck Brann og Redningstjeneste AS
Falck Global Assistance Norway AS
Falck Helseformidling AS
Falck Norge AS
EMI Holdings Management S.A.
EMI Panama S.A.
Falck Fire Services Polska Sp. z.o.o.
Falck Digital Technology Poland Sp. z.o.o.
Falck Medycyna Sp. z.o.o.
Falck SCI Portugal – Segurança Contra Incêndios, SA.
Falck Fire Services S.R.L
Falck Global Assistance Singapore Pte. Ltd.
Falck Fire Services a.s.
Falck Ambulance s.r.o
Falck Ambulance Sourcing, S.L.
Falck Global Assistance Spain S.L.
Falck SCI, S.A.
Falck Servicios Sanitarios, S.L.
Falck Sverige AB
Alviva AB
Falck Ambulans AB
Falck Global Assistance AB
Falck Healthcare AB
Falck Hälsopartner AB
Falck Räddningstjänst AB
Falck Global Assistance (Thailand) Ltd.
Falck Global Assistance Ltd.
Falck Eurasia B.V.
Falck Holding B.V.
Falck Sağlık AŞ
Falck Fire Services UK Limited
Luvtel S.A.
UCM Uruguay S.A.
Falck USA, Inc.
Falck Mobile Health Corp.
Falck Northern California Corp.
Falck Northwest Corp.
Falck Rocky Mountain, Inc.
Falck HealthNet, Inc.
Falck Southwest Corp.
Falck Global Assistance, LLC
The Morin Group LLC
The Group has consistently applied the following accounting policies to all periods presented in these consolidated financial statements, unless otherwise mentioned (see note 1 Basis of preparation).
The consolidated financial statements comprise the Parent Foundation Lundbeckfonden and entities controlled by the Parent Foundation.
Subsidiaries are all entities over which the Group has control. The Group controls an entity when it is exposed to, or has rights to, variable returns from its involvement with the entity and has the ability to affect those returns through its power to direct the activities of the entity. Subsidiaries are fully consolidated from the date on which control is transferred to the Group. They are deconsolidated from the date that control ceases.
The acquisition method of accounting is used to account for business combinations by the Group. Intercompany transactions, balances and unrealised gains on transactions between group companies are eliminated. Unrealised losses are also eliminated unless the transaction provides evidence of an impairment of the transferred asset. Accounting policies of subsidiaries have been changed where necessary to ensure consistency with the policies adopted by the Group.
Non-controlling interests in the results and equity of subsidiaries are shown separately in the consolidated income statement and of comprehensive income, statement of changes in equity and balance sheet, respectively.
Acquisitions are evaluated to determine whether they constitute a business combination in accordance with IFRS 3 Business Combinations or a purchase of individual assets and liabilities.
Acquired assets and liabilities that do not constitute a business are recognised at cost, i.e., no goodwill or negative goodwill is recognised.
Newly acquired or newly established companies or operations are recognised in the consolidated financial statements from the date of acquisition or establishment. The date of acquisition is the date when control of the company actually passes to the Group.
The acquisition method of accounting is used to account for all business combinations. The consideration transferred for the acquisition of a subsidiary comprises:
• fair values of the assets transferred
• liabilities incurred to the former owners of the acquired business
• equity interests issued by the Group
• fair value of any asset or liability resulting from a contingent consideration arrangement
• fair value of any pre-existing equity interest in the subsidiary. Identifiable assets acquired, and liabilities and contingent liabilities assumed in a business combination are, with limited exceptions, measured initially at their fair values at the acquisition date
The Group recognises any non-controlling interest in the acquired entity on an acquisition-by-acquisition basis, either at fair value or at the non-controlling interest’s
proportionate share of the acquired entity’s net identifiable assets. Acquisition-related costs are expensed as incurred.
The excess of the consideration transferred, amount of any non-controlling interest in the acquired entity, and acquisition-date fair value of any previous equity interest in the acquired entity over the fair value of the net identifiable assets acquired is recognised as goodwill. If those amounts are less than the fair value of the net identifiable assets of the business acquired, the difference is recognised directly in the income statement as a bargain purchase.
Companies divested or discontinued are recognised in the income statement until the date of divestment or discontinuation. Divested activities are shown separately as discontinued operations.
Gains and losses on divestment or winding up of subsidiaries and associates are stated as the difference between the sales price and the carrying amount of the net assets, including goodwill at the time of sale, accumulated foreign exchange adjustments recognised in other comprehensive income and anticipated disposal costs. In addition, any retained non-controlling interests are measured at fair value. Gains and losses on the disposal and the effect of renewed measurement of any retained non-controlling interests are recognised in the income statement.
On initial recognition, transactions denominated in foreign currencies are translated at standard rates which approximate the exchange rates at the
transaction date. Exchange differences arising between the exchange rates at the transaction date and the exchange rates at the date of payment are recognised in the income statement under financial items.
Receivables, payables, and other monetary items denominated in foreign currencies that have not been settled at the balance sheet date are translated at the exchange rates at the balance sheet date. The differences between the exchange rates at the time of recognition and the exchange rates at the balance sheet date or recognition of settlement are recognised in the income statement under financial items.
On translation of foreign subsidiaries having a functional currency different from the one used by the Group items in the income statement are translated at monthly average exchange rates, and non-monetary and monetary items are translated at the exchange rates at the balance sheet date. Exchange differences arising when translating the income statements and the balance sheets of foreign subsidiaries are recognised in other comprehensive income.
Exchange gains/losses on translation of receivables from, or payables to, subsidiaries that are considered part of the Group’s net overall investment in the subsidiaries are recognised in other comprehensive income.
Exchange gains/losses on that part of the bank debt in foreign currency which is used for hedging the net investments in subsidiaries, and which provides an effective hedging of the exchange gains/losses of the net investments, are recognised in other comprehensive income.
Forward exchange contracts, interest rate swaps, equity options and other derivatives are initially recognised in the balance sheet at fair value at the contract date and subsequently remeasured at fair value at the balance sheet date. The fair value of derivatives is determined by applying recognised measurement techniques, whereby assumptions are based on the market conditions prevailing at the balance sheet date. Positive and negative fair values are included in other receivables and other payables, respectively.
Changes in the fair value of derivatives classified as hedging instruments and meeting the criteria for hedge accounting are recognised in other comprehensive income. On recognition of hedged items, income and expenses related to such hedging transactions are transferred from other comprehensive income and recognised in the same line item as the hedged item.
Changes in the fair value of derivatives classified as cash flow hedging instruments and meeting the criteria for hedging the fair value of a recognised asset or liability are recognised in the income statement together with changes in the value of the hedged asset or liability.
Changes in the fair value of derivatives used for hedging net investments in foreign subsidiaries or associates and that otherwise meet the relevant criteria for hedging are recognised in other comprehensive income.
Changes in the fair value of derivatives not qualifying for hedge accounting are recognised in the income statement under financial items as they arise.
Securities, equity investments recognised in other financial assets, derivatives, and contingent consideration measured at fair value are classified according to the fair value hierarchy as belonging to levels 1-3 depending on the valuation method applied.
Share-based incentive programmes (equity-settled share-based payments) which comprise share option plans, conditional share plans, and performance shares are measured at the grant date at fair value and recognised in the income statement under the respective functions over the vesting period and offset in equity.
The fair value of share options is determined using the Black-Scholes model.
If the share option agreements entitle the Group to demand cash settlement of the options, the cashsettled share options are recognised as other liabilities and adjusted to fair value when the Group has an obligation to settle in cash. The subsequent adjustment to fair value is recognised in the income statement under financial items.
Warrants regarding warrant programmes for the executive management boards of subsidiaries are issued at the market value on the date of grant. Payments received and made in relation to the warrant programmes are recognised in equity.
On initial recognition, non-controlling interests are measured either at fair value (including the fair value of goodwill related to non-controlling interests in the
acquired company) or at the non-controlling interests’ proportionate share of the acquired company’s identifiable assets, liabilities and contingent liabilities measured at fair value (excluding the fair value of goodwill related to non-controlling interests in the acquired company). The measurement basis for noncontrolling interests is selected for each individual transaction.
Increases and reductions of non-controlling interests are accounted for as transactions with shareholders, in their capacity as shareholders. Thus, any differences between adjustment to the carrying amount of noncontrolling interests and the fair value of the consideration received or paid are recognised directly in equity.
Revenue from the sale of goods is recognised when Lundbeck/ALK have transferred control of products sold to the buyer and it is probable that Lundbeck/ALK will collect the consideration to which they are entitled for transferring the products. Control of the products is transferred at a single point in time, typically on delivery.
Revenue is measured at the amount of consideration to which the Groups expect to be entitled in exchange for transferring the products. Revenue is recognised net of sales deductions, including product returns as well as discounts, rebates and sales-based taxes.
Moreover, revenue includes licence income and royalties from out-licensed products as well as non-refundable down payments and milestone payments relating to research and development collaborations and income from collaborations on the commercialisation of products.
Sales-based licensing income and royalties from outlicensed products is recognised in profit or loss under revenue, when the Groups provide access to its product rights as they exist throughout the licence period. Revenue from sales-based licensing income is recognised when the performance obligation is satisfied, i.e., when transferred to the customer. For royalties, revenue is recognised when the subsequent sale occurs.
When the Groups provide a customer the right to use the product rights as they exist at the point in time at which the licence is granted, revenue is recognised at a point in time when control is transferred to the licensee, and the licence period begins when the customer's right to the intellectual property is transferred. Nonrefundable down payments and milestone payments received relating to research collaborations are recognised in profit or loss under revenue.
The Group may enter into consignment arrangements under which finished goods are delivered to a partner while the Group retains control of the goods. Revenue is recognised only when control of the finished goods transfers to the partner in accordance with the terms of the arrangement.
Revenue includes services and goods delivered together with invoiced subscriptions attributable to the financial
period. Revenue is recognised in the income statement if the control of the services or goods are transferred to the customer. Services are recognised over time when the customer receives and consumes the benefits as the service is delivered by Falck.
For long-term contracts with fixed price and subscriptions, revenue is recognised on a straight-line basis over the contract period. For pay-by-use contracts, revenue is recognised in connection with the actual delivery of the individual service.
Services related to long-term contracts with fixed price and subscriptions are usually prepaid by the customers. Prepayment usually occurs between one and twelve months before the start of the service period. For payby-use contracts, the payment term is usually between one and three months after the service has been delivered.
For contracts with predetermined price reductions, the transaction price will be recalculated to an average price covering the total contract period.
For contracts where Falck acts as an agent (mainly related to claims handling for insurance companies), revenue is recognised as the net amount that Falck is entitled to retain in return for its services as agent. For contracts where Falck acts as a principal, revenue is recognised as the gross amount to which Falck expects to be entitled.
Revenue is measured at the fair value of the agreed consideration excluding VAT and other taxes collected on behalf of third parties. All discounts granted are recognised in revenue.
Contracts with variable considerations are measured using the most likely amount and remeasured on a monthly basis.
of sales
Cost of sales comprises the cost of goods and services sold. Cost includes the cost of raw materials, transportation costs, consumables and goods for resale, direct labour and indirect costs of production, including operating costs, amortisation/depreciation and impairment losses relating to product rights and manufacturing facilities. Moreover, cost of sales includes royalty payments for in-licensed products, expenses for quality assurance of products and write-downs to net realisable value of obsolete and slow-moving goods.
Cost of sales also includes external assistance used to generate the revenue for the year.
Research and development costs comprise costs incurred for the Group’s research and development functions, i.e., employee costs, amortisation/ depreciation and impairment losses, and other indirect costs as well as costs, relating to research and development collaborations.
Research costs are always recognised in the income statement as they incur.
Due to a very long development period and the significant uncertainties inherent in the development of new products, development costs are expensed as incurred in line with industry practice. Consequently, the development costs do not qualify for capitalisation as intangible assets until marketing approval by a
regulatory authority is obtained or considered highly probable.
Sales and distribution costs comprise costs incurred for the sale and distribution of the Group’s products sold during the year. This includes costs incurred for sales campaigns, training and administration of the sales force and direct distribution, marketing and promotion. Sales and distribution also include salaries and other costs for the sales, distribution and marketing functions, amortisation/depreciation and impairment losses, and other indirect costs.
Administrative expenses comprise expenses incurred in the year for the management and the administration of the Group, i.e., salaries and other expenses relating to for example management, HR, IT and finance functions, as well as amortisation/depreciation and impairment losses, and other indirect costs.
Other operating items comprise other income and expenses relating to operating activities of a secondary nature to the Group. Other operating items include integration and transaction costs relating to material acquisitions, transaction costs relating to divestments, income and expenses relating to legal settlements and material gains and losses on the sale or retirement of items of intangible assets and property, plant and equipment.
Special items comprise significant income and expenses of a special nature in terms of the Group’s revenue-
generating operating activities such as impairment of goodwill and product rights.
Results of investments in associates measured using the equity method
The proportionate share of the results of associates is recognised in the income statement after tax.
Financial items comprise:
• Interest income and expenses
• Net gain or loss on financial assets, including dividends
• Fair value adjustment of contingent consideration
• Fair value adjustment of other financial liabilities
• Interest expenses and income related to uncertain tax positions are recognised on the balance sheet as tax liabilities and tax assets, respectively, upon the receipt of rulings from the tax authorities, and correspondingly reflected in the income statement as financial items, net
• Foreign currency gain and loss
• Other financial income and expenses
Interest income and expenses are accrued based on the principal and the effective rate of interest.
Lundbeckfonden’s Danish subsidiaries are jointly taxed with Lundbeckfond Invest A/S as the administration company. The current Danish corporate income tax liability is allocated among the companies of the tax pool in proportion to their taxable income (full allocation subject to reimbursement in respect of tax losses).
Lundbeckfonden has the option to use section 3(4) of the Danish Corporation Tax Act. Under these rules, the taxable income of Lundbeckfond Invest A/S is considered to have been earned by Lundbeckfonden if the taxable income is distributed as a dividend to Lundbeckfonden. Since Lundbeckfonden’s taxable income is regularly offset against grants for the year and tax provisions for future grants, no deferred tax asset or liability is recognised owned by Lundbeckfonden.
Tax for the year, which consists of the year’s current tax and the change in deferred tax, is recognised in the income statement as regards the amount that can be attributed to the net profit or loss for the year, in other comprehensive income as regards the amount that can be attributed to items in other comprehensive income, and in equity as regards the amount that can be attributed to items in equity. The effect of foreign exchange differences on deferred tax is recognised in the balance sheet as part of the movements in deferred tax.
The Group has determined that the global minimum top-up tax, which it is required to pay under Pillar Two legislation, is an income tax in the scope of IAS 12. The Group has applied a temporary mandatory relief from deferred tax accounting for the impacts of the top-up tax and will account for it as a current tax when it incurs.
The current income tax charge is calculated based on the tax laws enacted or substantively enacted at the end of the reporting period in the countries where the Group operates and generates taxable income. Management periodically evaluates positions taken in
tax returns with respect to situations in which applicable tax regulation is subject to interpretation and considers whether it is probable that a tax authority will accept an uncertain tax treatment. The Group measures its tax balances based on either the most likely amount or the expected value, depending on which method provides a better prediction of the resolution of the uncertainty.
Current tax for the year is calculated based on the income tax rates and rules applicable at the reporting date.
Current tax payables and receivables, including contributions payable and receivable under the Danish joint taxation scheme, are recognised in the balance sheet, computed as tax calculated on the taxable income for the year adjusted for provisional tax paid.
Deferred tax is recognised on all temporary differences between the carrying amounts of assets and liabilities and their tax bases. However, deferred tax is not recognised on temporary differences arising either on initial recognition of goodwill or from a transaction that is not a business combination, if the temporary difference ascertained at the time of the initial recognition affects neither the financial results nor the taxable income and does not give rise to equal taxable and deductible temporary differences. The tax value of the assets is calculated based on the planned use of the individual assets.
Deferred tax is measured based on the income tax rates and tax rules in force in the respective countries at the balance sheet date. Changes in deferred tax resulting
from changed income tax rates or tax rules are recognised in profit or loss.
Deferred tax assets, including the tax value of tax loss carry-forwards, are recognised in the balance sheet at the value at which the assets are expected to be realised, either through an offset against deferred tax liabilities or as net tax assets to be offset against future positive taxable income.
Changes in deferred tax concerning expenses for sharebased payments are generally recognised in profit or loss. However, if the amount of the tax deduction exceeds the related cumulative expense, it indicates that the tax deduction relates not only to an operating expense but also to an equity item. In such a case, the excess of the associated current or deferred tax is recognised directly in equity.
Deferred tax in respect of recaptured losses previously deducted in foreign subsidiaries is recognised based on a specific assessment of each individual subsidiary.
On initial recognition, goodwill is measured and recognised as the excess of the cost over the fair value of the acquired assets, liabilities and contingent liabilities. On recognition, the goodwill amount is allocated to those of the Group’s activities that generate separate cash flows (cash generating units).
Development costs are recognised in the income statement as they incur unless the conditions for capitalisation have been met. Development costs are
capitalised only if the development projects are clearly defined and identifiable and where the technical rate of utilisation of the project, the availability of adequate resources, and a potential future market or development opportunity can be demonstrated. Furthermore, such costs are capitalised only where the intention is to manufacture, market or use the project, when the cost can be measured reliably and when it is probable that future earnings can cover production, sales and distribution costs, administrative expenses and development costs.
After completion of the development work, development costs are amortised over the expected useful life. The maximum amortisation period for development projects protected by intellectual property rights is consistent with the remaining patent protection period of the rights concerned. Ongoing development projects are tested for impairment at least annually or when there is an indication of impairment.
Acquired intellectual property rights in the form of product rights, patents, licences, know-how, customer relationships, brands and software are measured at cost less accumulated amortisation and impairment losses. The cost of software comprises the cost of planning, labour costs, and costs directly attributable to the project. Subsequent milestone-related expenditures are considered contingent consideration and the Group follows the cost accumulation approach.
Product rights are amortised over the economic lives of the underlying products, which in all material aspects follow the patent terms, currently between five and 15 years. Other intangible assets are amortised over the
useful life/period of agreement Intangible assets acquired on acquisition are amortised over the expected economic life, estimated to be three-to-10 years. Software is amortised over the expected economic life, estimated to be three-to-five years. Amortisation commences when the asset is ready to be brought into use.
Amortisation is recognised in the income statement under cost of sales, and research and development costs, respectively.
Borrowing costs to finance the manufacture of intangible assets are recognised in the cost price if such borrowing costs relate to the production period. Other borrowing costs are expensed.
Gains and losses on the disposal of development projects, patents and licences are measured as the difference between the selling price less cost to sell and the carrying amount at the time of sale. Gains and losses are recognised in profit or loss; normally in other operating items, net, or, if considered immaterial to the understanding of the consolidated financial statements, in the same line item as the associated amortisation. In general, amortisation methods, useful lives, and residual values are reviewed at each reporting date and adjusted if appropriate.
Items of property, plant and equipment are measured at cost less accumulated depreciation and impairment losses. Land is not depreciated.
Cost includes the cost of acquisition and expenses directly attributable to the acquisition until the asset is
ready for use. The cost of self-constructed assets includes costs directly attributable to the construction of the asset. Borrowing costs to finance the manufacture of property, plant and equipment are recognised in the cost price, if such borrowing costs relate to the production period. Other borrowing costs are recognised in the income statement.
Right-of-use assets are initially measured at cost, which comprises the initial amount of the liability adjusted for any lease payments made at or before the commencement date, plus any initial direct costs incurred and an estimate of costs to dismantle and remove the underlying asset or to restore the underlying asset or the site on which it is located, less any lease incentives.
Subsequently, the right-of-use asset is depreciated using the straight-line method from the commencement date to the end of the lease term unless it is reasonably certain that a purchase option will be exercised at the end of the lease term. In that case, the assets are depreciated over the full, expected useful life.
Items of property, plant and equipment are depreciated on a straight-line basis over the expected useful lives of the assets:
Plant and machinery
Vehicles according to category
Dispatch centres, radio systems, major administrative systems and networks
Fire extinguishers and similar equipment installed at customers’ locations
Other fixtures and fittings, tools and equipment 3-10
Leasehold improvements, max. 10
Depreciation methods, useful lives and residual values are reassessed annually and adjusted if appropriate.
Costs incurred that increase the recoverable amount of the asset are added to the value of the asset as an improvement and are depreciated over the estimated useful life of the improvement.
Gains or losses on the sale or disposal of items of property, plant and equipment are calculated as the difference between the carrying amount and the selling price less cost to sell or discontinuance costs. Gains and losses are recognised in profit or loss; normally in other operating items, net or, if considered immaterial to the understanding of the consolidated financial statements, in the same line item as the associated depreciation.
Forest assets are divided into growing forests, which are recognised as biological assets at fair value less cost to sell, and land, which is measured at cost. The valuation
of biological assets is based on discounted cash flow models.
Changes in the fair value of biological assets are recognised in the income statement under other operating items.
Intangible assets with indefinite useful lives and intangible assets not yet commercialised are not subject to amortisation and are tested annually for impairment, or more frequently if events or changes in circumstances indicate that they may be impaired. The annual impairment test is performed irrespective of whether there is any indication of impairment.
Intangible assets and property, plant and equipment in use with finite useful lives are tested for impairment whenever events or changes in circumstances indicate that the carrying amount may not be recoverable.
Impairment losses are reversed only if the assumptions and estimates underlying the impairment calculation have changed. Indications of impairment or reversal of impairment include the following:
• Research and development results for a product
• Changes in expected cash flows due to lower sales expectations
• Changes in technology
• Changes in assumptions about future use
• Changes in market and legal risks
• Changes in cost structure
• Changes in discount rate
An impairment loss is recognised for the amount by which the asset’s carrying amount exceeds its recoverable amount. The recoverable amount is the higher of an asset’s fair value less costs of disposal and value-in-use. For the purpose of assessing impairment, assets are grouped at the lowest levels for which there are separately identifiable cash inflows that are largely independent of the cash inflows from other assets or groups of assets (cash-generating unit). Non-financial assets other than goodwill that have suffered an impairment are reviewed for possible reversal of the impairment at the end of each reporting period.
Investments in associates Investments in associates, except for investments in associates that are included in Lundbeckfonden’s investment strategy, are measured in the consolidated financial statements using the equity method and recognised at the proportionate share of the equity of the relevant enterprise, made up in accordance with the Group’s accounting policies, with the addition of values added on acquisition, including goodwill.
Investments in associates that are included in Lundbeckfonden’s investment strategy are measured at fair value and presented together with the investment assets. Both realised and unrealised gains and losses are recognised in the income statement under financial items.
Financial assets
At initial recognition, securities that are included in the group’s investment strategy are measured at its fair value. Transaction costs of financial assets are expensed under financial items.
Subsequently, securities are measured at fair value at the balance sheet date. Both realised and unrealised gains and losses are recognised in the income statement under financial items.
Bonds with a term to maturity of less than one year are recognised in current assets. Bonds forming part of repo transactions, i.e., the selling of bonds to be repurchased at a later date, remain on the balance sheet as financial assets, and the amount received on repo transactions is recognised as repo debt. Returns on such bonds are recognised under financial items.
The fair value of listed investments is calculated using market prices at the balance sheet date. The calculation of fair value of unlisted investments, including biotech investments, is made on the basis of relevant valuation methods based on discounted cash flows or trading multiples. If the fair value cannot be determined with sufficient reliability, the investments in question are recognised at cost at recent transaction, or price at financing round, taking into account whether or not the companies live up to predefined value triggers/business plans.
Equity investments that are not included in the Group’s investment strategy are classified as other financial assets. On initial recognition, these investments are measured at cost, corresponding to fair value. They are subsequently measured at fair value at the balance sheet date, and changes to the fair value are recognised in the income statement or other comprehensive income according to an individual decision for each equity investment.
Inventories are measured at the lower of cost and net realisable value. Cost is determined using the FIFO method. Work in progress and finished goods manufactured by the Group are measured at cost, comprising raw materials and consumables, direct labour, and production overheads. Indirect production costs include materials, labour, maintenance, and depreciation of production equipment and facilities, as well as factory administration and management. Indirect production costs are allocated based on the normal capacity of the production facilities.
Inventories are written down to net realisable value when this is lower than cost. Net realisable value is the estimated selling price in the ordinary course of business less costs of completion and costs to sell. Net realisable value is determined by marketability, obsolescence, and expected selling price developments.
Trade receivables are initially recognised at transaction price and subsequently measured at amortised cost using the effective interest method, less allowance for bad debts.
Other receivables recognised in financial assets are financial assets with fixed or determinable cash flows that are not quoted in an active market and are not derivative financial instruments
On initial recognition, other receivables are measured at fair value and subsequently at amortised cost, which usually corresponds to the nominal value less writedowns to counter the risk of losses. Write-downs are calculated using the full lifetime-expected credit losses
method, whereby the likelihood of non-fulfilment throughout the lifetime of the financial instrument is taken into consideration. A provision account is used for this purpose
Contract assets comprise the Group’s right to consideration regarding completed services which have not been invoiced at the reporting date.
Impairments for lifetime expected credit losses (ECL) are recognised in the income statement upon initial recognition of the contract asset. The expected credit losses are calculated according to the portfolio of contract assets and are grouped by credit risk characteristics. A provision matrix is established based on the historical development in contract assets and historical credit losses, and adjusted for forward-looking factors specific to the debtors and the economic environment. The expected credit losses are recognised in sales and distribution costs.
Prepayments comprise prepaid costs which are measured at cost.
On initial recognition, securities including the bond portfolio, which are included in the Group’s investment strategy for excess liquidity, or bonds with a term to maturity of less than one year, are recognised under current assets and measured at cost, corresponding to fair value. The securities are subsequently measured at fair value at the balance sheet date. The fair value is based on publicly quoted prices of the invested assets. Both realised and unrealised gains and losses are recognised in the income statement.
36
Equity
Authorised grants
Grants are considered equity movements and are recognised as a liability at the time when the grant has been authorised by the Board of Directors and announced to the recipient. Authorised grants not yet disbursed are recognised in non-current or current liabilities, respectively.
Reserve for future grants
In accordance with the Danish Act on Commercial Foundations, a reserve for future grants has been set up in order for Board of Directors to be able to donate grants during the period until the approval of the annual report for the subsequent financial year. The reserve does not have to be used, but is continuously reduced with donated grants. Every year at the annual meeting, the Board of Directors will re-evaluate the size of the reserve.
Hedging reserve
Hedge transactions that meet the criteria for hedging future cash flows, and for which the hedged transaction has yet to be realised, are recognised in equity through other comprehensive income under the hedging reserve.
Foreign exchange adjustments concerning hedging transactions used to hedge the Group’s net investment in such entities are recognised in equity through other comprehensive income under the hedging reserve.
Currency translation reserve
Foreign exchange adjustments arising on the translation of financial statements for subsidiaries and associates which are not part of Lundbeckfonden’s investment strategy and have a functional currency
other than DKK, and foreign exchange adjustments relating to financial assets and liabilities representing a part of the Group’s net investment in such entities, are recognised in equity through other comprehensive income under the currency translation reserve.
On full or partial realisation of a net investment, foreign exchange adjustments are recognised in the income statement.
Treasury shares in subsidiaries
Acquisition and sale of treasury shares held by subsidiaries as well as dividends are recognised directly in equity under retained earnings.
The proportionate shares of the profit and equity of subsidiaries attributable to non-controlling interests are recognised as a separate item under equity. On initial recognition, non-controlling interests are recognised as described under ‘Business combinations’.
Share-based incentive programmes in which shares are granted to employees and in which employees may opt to buy shares in H. Lundbeck A/S and ALK-Abelló A/S (equity-settled programs) are measured at the equity instruments’ fair value at the date of grant and recognised as employee costs over the vesting period as employees earn the right to receive or purchase the shares. The offsetting item is recognised directly in equity under retained earnings.
Share price-based incentive programmes in which employees have the difference between the agreed price and the actual share price settled in cash (cash-settled
programmes) are measured at fair value at the date of grant and recognised as employee costs over the vesting period as employees earn the right to the cash settlement. The cash-settled programmes are subsequently remeasured at each balance sheet date and upon final settlement, with any changes in fair value recognised in employee costs. The offsetting item is recognised under liabilities until the time of the final settlement.
Payments to defined contribution plans are recognised in the income statement at the due date, and any contributions payable are recognised in the balance sheet under current liabilities.
The present value of the Group’s liabilities relating to future pension payments under defined benefit plans is measured on an actuarial basis once a year based on the pensionable period of employment up to the time of the actuarial valuation. The calculation of present value is based on assumptions of future developments of salary, interest, inflation, mortality and disability rates and other factors. Present value is computed exclusively for the benefits to which the employees have earned entitlement through their employment with the Group. Pension expenses, finance costs and administration fees are recognised in the income statement under employee costs. Actuarial gains and losses are recognised in other comprehensive income as they are calculated and cannot subsequently be recycled through profit or loss.
The present value of the defined benefit plan liability is recognised less the fair value of the plan assets, and any net obligation is recognised in the balance sheet under
non-current liabilities. Any net asset is recognised in the balance sheet as a financial asset, considering, where relevant, the provisions of IFRIC 14 The Limit on a Defined Benefit Asset, Minimum Funding Requirements and their Interaction.
Provisions mainly consist of provisions for discounts and rebates, product returns, pending lawsuits, environmental, restructuring and integration provisions A provision is a liability of uncertain timing or amount.
Unsettled discounts and rebates are recognised as provisions when the timing or amount is uncertain. Where absolute amounts are known, the discounts and rebates are recognised as trade payables.
Return obligations imposed on the Group are recognised as provisions in the balance sheet.
Amounts relating to provisions are recognised when the outflow is probable, and the amount is measured as the best estimate of the costs required to settle the liabilities at the balance sheet date.
In connection with restructurings in the Group, provisions are made only for liabilities set out in a specific restructuring plan based on which the parties affected can reasonably expect that the Group will carry out the restructuring, either by starting to implement the plan or announcing its main components.
Prepayments mainly include accrued subscriptions and prepayment according to contracts. Contract liabilities also comprise the recalculated transaction price from
predetermined price reductions, where the service is transferred over time, and are recognised at the same average consideration over the term of the contract.
Mortgage debt, bank debt and bond debt are recognised at the time of the raising of a loan/issuing of bonds at the fair value of the proceeds received less transaction costs paid. In subsequent periods, the financial liabilities are measured at amortised cost, which is equivalent to the capitalised value when the effective rate of interest is used. The difference between the proceeds and the nominal value is recognised in the income statement under financial items over the loan period. Debt included in the short-term financial liquidity is measured at amortised cost in subsequent periods.
Repo debt relates to bonds included in repo transactions. Repo debt is recognised at amortised cost, and accumulated repo interest has been accrued.
Other payables
Other payables include employee costs payables, contingent consideration, derivative financial instruments, debt to public authorities, etc.
Contingent consideration is recognised as part of the business combination and is recognised at fair value considering the passage of time and changes in the applied probability of success. The fair value is assessed at each reporting date and the effect of any adjustments relating to the timing of payment and the probability of success is recognised under financial income or financial expenses.
Other debts, which include trade payables and debt to public authorities, etc., are measured at amortised cost.
On initial recognition, lease liabilities are measured as the present value of future payments, including payments relating to reasonably certain extensions. The lease payments contain fixed payments less any lease incentives receivable and variable lease payments that depend on an index or a rate.
On subsequent recognition, lease liabilities are measured at amortised cost. The difference between the present value and the nominal value of lease payments is recognised in the income statement over the term of the lease as a finance charge.
If the interest rate cannot be determined in the agreement, the lease payments are discounted using the Group’s incremental borrowing, adjusted for the functional currency and length of the lease term. The lease liability is remeasured if, or when, the future payment or lease term changes.
Changes to lease agreements after initial recognition are accounted for either as a modification to an existing agreement, a separate agreement or a partial disposal, depending on the nature of the change. Changes will result in changes to both the lease liability and the right-of-use asset.
The Group has decided not to recognise right-of-use assets and lease liabilities for short-term leases and lowvalue asset leases. The Group recognises the lease payments associated with these leases as an expense over the lease term.
The cash flow statement is presented in accordance with the indirect method commencing with the operating profit.
Cash flows include cash flows from companies acquired as from the date of acquisition and cash flows from companies divested until the date of divestment.
Cash flows denominated in foreign currencies, including cash flows in foreign subsidiaries, are translated at the average exchange rates for the year as they approximate the actual exchange rates at the date of payment. Cash and cash equivalents at year-end are translated at the exchange rates at the balance sheet date, and the effect of exchange gains/losses on cash and cash equivalents is shown as a separate item in the cash flow statement.
Key figures
The key figures are calculated according to the Danish Finance Society’s Recommendations & Financial Ratios.
Operating profit: Defined as the profit before special items, financial items and tax
Operating profit margin: Operating profit x 100 / Revenue
Return on equity: Profit for the year x 100 / Average equity
Lundbeckfonden’s net wealth is estimated based on fair value at the balance sheet date. For the valuation of Lundbeckfonden’s investment activities, please refer to
the accounting policies above under financial assets. The fair value of Lundbeckfonden’s shares in H. Lundbeck A/S and ALK-Abelló A/S is based on market prices. The fair value of Lundbeckfonden’s shares in other strategic ownerships are an estimated value based on a trading multiple model using historical accounting numbers for the relevant company and a peer group.
Note 37
Events after the balance sheet date
No events of importance to the annual report have occurred during the period from the balance sheet date until the presentation of the consolidated financial statements.
For the period 1 January – 31 December
of
for the period 1 January – 31 December
The financial statements for Lundbeckfonden (the Parent Foundation) for 2025 have been prepared in accordance with the Danish Financial Statements Act for large reporting enterprises class C.
Other securities and investments are accounted for using the fair value through the income statement and accounted for in accordance with IFRS 9.
The financial statements are presented in Danish kroner (DKK), which is also the functional currency of Lundbeckfonden. All amounts have been rounded to millions, unless otherwise indicated.
The accounting policies are unchanged from the previous year.
Differences relative to the accounting policies for the consolidated financial statements
The Parent Foundation’s accounting policies for recognition and measurement are consistent with the policies for the consolidated financial statements with the exceptions stated below.
Investments in subsidiaries
Investments in subsidiaries are measured at cost. Costs include the consideration at fair value plus direct acquisition costs.
Subsequently, investments in subsidiaries that suffered an impairment are reviewed for possible reversal of the impairment at the end of each balance sheet date. Reversal of impairment is recognised in financial income.
If there is an indication of impairment, an impairment test is performed. Where the carrying amount exceeds the recoverable amount, the investments are written down to this lower value. The recoverable amount is the higher of fair value less costs to sell and value-in-use.
Dividends are recognised in the income statement.
With reference to section 86(4) of the Danish Financial Statements Act and the consolidated financial statements of Lundbeckfonden, the parent has not prepared a cash flow statement.
2 Financial income and expenses
Members of Executive Management and the Board of Directors, who also serve as board members in subsidiaries also receive board remuneration directly from such subsidiaries. For a complete description hereof, see note 3 in the consolidated financial statements.
Other external costs include fees to auditors appointed by the Board of Directors.
When calculating the taxable income, Lundbeckfonden has deducted grants and tax provisions for future grants. No deferred taxes are recognised for accounting purposes concerning tax provisions for future grants as these are not expected to materialise. Deferred tax
Note 12
Payable grants, short-term amount to DKK 676m (DKK 728m at 31 December 2024). Payable grants, long-term amount to DKK 1,847m (DKK 1,738m at 31 December 2024), of which DKK 239m (DKK 230m at 31 December 2024) are due after more than five years.
Note 15
Lundbeckfonden has contractual capital contribution obligations amounting to DKK 24m at 31 December 2025 (DKK 27m at 31 December 2024).
Lundbeckfonden’s related parties are:
• the Foundation’s Board of Directors and Executive Management, including their close family members and related undertakings
• the Foundation’s wholly owned investment and holding company, Lundbeckfond Invest A/S, and its subsidiaries: H. Lundbeck A/S, ALK Abelló A/S, Falck A/S, LFI Equity A/S, LFI Silva Investments A/S, DySIS Medical, Inc. and Vesper Bio ApS, together with their respective subsidiaries and associates
Lundbeckfond Invest A/S shares the same address as Lundbeckfonden, and there is duality of membership between the Executive Management, administration (partly) and Boards of Directors. Lundbeckfonden receives dividends from Lundbeckfond Invest A/S, which are recognised in the income statement.
In 2025, Lundbeckfonden received dividend from Lundbeckfond Invest A/S of DKK 0 (DKK 1,545m)
Lundbeckfonden received payment for administration costs, net amount DKK 10m in 2025 (DKK 10m in 2024) from Lundbeckfond Invest A/S. At 31 December 2025
Lundbeckfonden has a payable to Lundbeckfond Invest A/S of DKK 88m (Receivable of DKK 553m at 31 December 2024) and a receivable from Vesper Bio ApS of DKK 270m (DKK 242m at 31 December 2024).
Lundbeckfonden received interest on receivables from Lundbeck Invest A/S and Vesper Bio ApS of DKK 4m (DKK 86m in 2024) and DKK 20m (DKK 14m in 2024), respectively.
For information on remuneration paid to the members of the Executive Management and Board of Directors, see note 3 in the consolidated financial statements.
The Foundation has purchased electricity for DKK 0.1m from Ørsted, in which CEO, Lene Skole, serves as chair of the board of directors.
Other than the above, there have only been a few transactions of immaterial importance with related parties.
The Foundation has not entered into any transactions with related parties that were not on an arm’s length basis.
Note 17
Events after the balance sheet date
No events of importance to the annual report have occurred during the period from the balance sheet date until the presentation of the financial statements.
The Executive Management and the Board of Directors have today considered and adopted the annual report of Lundbeckfonden for the financial year 1 January31 December 2025.
The Consolidated Financial Statements have been prepared in accordance with IFRS Accounting Standards as adopted by the EU and further requirements in the Danish Financial Statements Act, and the Parent Foundation Financial Statements have been prepared in accordance with the Danish Financial Statements Act.
In our opinion, the Consolidated Financial Statements and the Parent Foundation Financial Statements give a true and fair view of the financial position at 31 December 2025 of the Group and the Parent Foundation, and of the results of the Group’s and the Parent Foundation’s operations and the Group’s cash flows for 2025.
In our opinion, Management Review includes a true and fair account of the development in the operations and financial circumstances of the Group and the Parent Foundation, of the results for the year and of the financial position of the Group and the Parent Foundation as well as a description of the most significant risks and elements of uncertainty facing the Group and the Parent Foundation.
We recommend that the Annual Report be adopted at the Board of Directors Meeting.
Copenhagen, 26 March 2026
Opinion
In our opinion, the Consolidated Financial Statements give a true and fair view of the Group’s financial position at 31 December 2025 and of the results of the Group’s operations and cash flows for the financial year 1 January to 31 December 2025 in accordance with IFRS Accounting Standards as adopted by the EU and further requirements in the Danish Financial Statements Act.
Moreover, in our opinion, the Parent Foundation Financial Statements give a true and fair view of the Parent Foundation’s financial position at 31 December 2025 and of the results of the Parent Foundation’s operations for the financial year 1 January to 31 December 2025 in accordance with the Danish Financial Statements Act.
We have audited the Consolidated Financial Statements and the Parent Foundation Financial Statements of Lundbeckfonden for the financial year 1 January - 31 December 2025, which comprise income statement, balance sheet, statement of changes in equity and notes, including material accounting policy information, for both the Group and the Parent Foundation, as well as statement of comprehensive income and cash flow statement for the Group (“financial statements”)
We conducted our audit in accordance with International Standards on Auditing (ISAs) and the additional requirements applicable in Denmark. Our responsibilities under those standards and requirements are further described in the “Auditor’s Responsibilities for the Audit of the Financial Statements” section of our report. We are independent of the Group in accordance with the International Ethics Standards Board for Accountants’ International Code of Ethics for Professional Accountants (IESBA Code) and the additional ethical requirements applicable in Denmark, and we have fulfilled our other ethical responsibilities in accordance with these requirements and the IESBA Code. We believe that the audit evidence we have obtained is sufficient and appropriate to provide a basis for our opinion.
Management is responsible for Management Review.
Our opinion on the financial statements does not cover Management Review, and we do not express any form of assurance conclusion thereon.
In connection with our audit of the financial statements, our responsibility is to read Management Review and, in doing so, consider whether Management Review is materially inconsistent with the financial statements or our knowledge obtained during the audit, or otherwise appears to be materially misstated.
Moreover, it is our responsibility to consider whether Management Review provides the information required under the Danish Financial Statements Act
Based on the work we have performed, in our view, Management Review is in accordance with the Consolidated Financial Statements and the Parent Foundation Financial Statements and has been prepared in accordance with the requirements of the Danish Financial Statements Act. We did not identify any material misstatement in Management Review.
Management is responsible for the preparation of Consolidated Financial Statements that give a true and fair view in accordance with IFRS Accounting Standards as adopted by the EU and further requirements in the Danish Financial Statements Act and for the preparation of Parent Foundation Financial Statements that give a true and fair view in accordance with the Danish Financial Statements Act, and for such internal control as Management determines is necessary to enable the preparation of financial statements that are free from material misstatement, whether due to fraud or error.
In preparing the financial statements, Management is responsible for assessing the Group’s and the Parent Foundation’s ability to continue as a going concern, disclosing, as applicable, matters related to going concern and using the going concern basis of accounting in preparing the financial statements unless Management either intends to liquidate the Group or the Parent Foundation or to cease operations, or has no realistic alternative but to do so
Our objectives are to obtain reasonable assurance about whether the financial statements as a whole are free from material misstatement, whether due to fraud or error, and to issue an auditor’s report that includes our opinion. Reasonable assurance is a high level of assurance, but is not a guarantee that an audit conducted in accordance with ISAs and the additional requirements applicable in Denmark will always detect a material misstatement when it exists. Misstatements can arise from fraud or error and are considered material if, individually or in the aggregate, they could reasonably be expected to influence the economic decisions of users taken on the basis of these financial statements.
As part of an audit conducted in accordance with ISAs and the additional requirements applicable in Denmark, we exercise professional judgement and maintain professional scepticism throughout the audit. We also:
• Identify and assess the risks of material misstatement of the financial statements, whether due to fraud or error, design and perform audit procedures responsive to those risks, and obtain audit evidence that is sufficient and appropriate to provide a basis for our opinion. The risk of not detecting a material misstatement resulting from fraud is higher than for one resulting from error as fraud may involve collusion, forgery, intentional omissions, misrepresentations, or the override of internal control.
• Obtain an understanding of internal control relevant to the audit in order to design audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Group’s and the Parent Foundation’s internal control.
• Evaluate the appropriateness of accounting policies used and the reasonableness of accounting estimates and related disclosures made by Management
• Conclude on the appropriateness of Management’s use of the going concern basis of accounting in preparing the financial statements and, based on the audit evidence obtained, whether a material uncertainty exists related to events or conditions that may cast significant doubt on the Group’s and the Parent Foundation’s ability to continue as a going concern. If we conclude that a material uncertainty exists, we are required to draw attention in our auditor’s report to the related disclosures in the financial statements or, if such disclosures are inadequate, to modify our opinion. Our conclusions are based on the audit evidence obtained up to the date of our auditor’s report. However, future events or conditions may cause the Group and the Parent Foundation to cease to continue as a going concern.
• Evaluate the overall presentation, structure and contents of the financial statements, including the disclosures, and whether the financial statements represent the underlying transactions and events in a manner that gives a true and fair view.
• Plan and perform the group audit to obtain sufficient appropriate audit evidence regarding the financial information of the entities or business units within the group as a basis for forming an opinion on the Consolidated Financial Statements. We are responsible for the direction, supervision and review of the audit work performed for purposes of the group audit. We remain solely responsible for our audit opinion
We communicate with those charged with governance regarding, among other matters, the planned scope and timing of the audit and significant audit findings, including any significant deficiencies in internal control that we identify during our audit.
Hellerup 26 March 2026
PricewaterhouseCoopers
Statsautoriseret Revisionspartnerselskab CVR No 33 77 12 31