Long Game Legends: Makower and Papiernik’s Playbook for Success

Medtech M&A, Overheard, Market Dives, LSI Alumni Innovator Spotlight, and more...

Long Game Legends: Makower and Papiernik’s Playbook for Success

Medtech M&A, Overheard, Market Dives, LSI Alumni Innovator Spotlight, and more...
December 2024

Scott Pantel Chief Executive Officer, LSI Editor-in-Chief

Rebekah Murietta Vice President of Media, LSI Contributing Author

Kelly Williams Subscriptions & Enterprise Sales, LSI

Henry Peck Chief Business Officer, LSI Editor-in-Chief

Brenna Hopkins Sr. Content Manager, LSI Contributing Author

Tracy Schaaf Managing Editor and Content Strategist, LSI Lead Author

Blake Matrone Sr. Marketing Manager, LSI Contributing Author

Nicholas Talamantes Sr. Director of Market Intelligence, LSI Contributing Author

Maricela Almonte Customer Service & Fulfillment, LSI
Printed in the U.S.A.
A publication of Life Science Intelligence, Inc. (LSI) 17011 Beach Boulevard, Suite 500, Huntington Beach CA 92647 714 847 3540 tel/fax email: info@ls-intel.com www.lifesciencemarketresearch.com
Vol. 1, No. 2

Scan QR Code to activate your subscription.
The Lens is published monthly and available in print and electronic formats. Copyright by Life Science Intelligence, Inc. All rights reserved. Editor takes care to report information from reliable sources and does not assume liability for information published.
Interested in subscribing? email to: info@ls-intel.com
Interested in advertising? email to: kelly@ls-intel.com
Dear Readers,
As we close out an eventful 2024, we are thrilled to present the second issue of The Lens. In a year which was in many ways defined by global challenges and political uncertainty, the medtech industry has served as both a sanctuary for those of us who work in it and a beacon of hope for the world we aim to improve. It is a place where innovation thrives, where we come together to tackle the world’s most pressing health challenges, and where our shared mission remains steadfast: building a healthier and happier future for our parents, contemporaries, children, and generations to come.
This issue reflects the resilience of our industry. Two of our articles highlight pivotal themes that are not only shaping the present but also setting the stage for an exciting future.
First, insights from the fireside chat with Josh Makower, MD, and Antoine Papiernik at LSI Europe offer a masterclass in what it takes to achieve transformative success in medtech. Their reflections underscore the importance of people, a long-haul mindset, and a collaborative spirit needed to move mountains in medtech. The $13.1 billion acquisition of Shockwave Medical by J&J serves as a shining example of these principles, demonstrating how persistence, teamwork, and a relentless commitment to innovation can create life-changing outcomes for patients.
Second, our analysis of the medtech M&A landscape reveals how 2024 has been a year which thawed the ice, setting the stage for continued momentum in 2025. With over $47 billion in acquisitions through the third quarter alone, the industry has experienced robust activity across markets like cardiovascular, neuromodulation, and more.
As we head into the final innings of 2024, we are closely watching advancements in artificial intelligence, charting the renewed venture and private capital deployment, and noting seminal FDA approvals and clearances. More than just milestones, these are signals of an ecosystem that is rising to meet the challenges ahead and seize opportunities towards an extraordinary future.
This moment also calls for thoughtful reflection. Meeting the challenges ahead will require more than ambition—it will demand strategic foresight, careful planning, and a dedication to cultivating key relationships and reliable intelligence. This is at the core of what LSI and The Lens magazine strive to deliver: access to the people and insights that enable our industry to build breakthrough businesses, efficiently and repeatedly.
With the holiday season upon us and the energy that a new year brings, we hope this issue provides you with insight, inspiration, and a sense of connection to the vibrant and growing medtech and healthtech ecosystem. On behalf of our entire team, we are proud to play a small role in your growth and journey.
Here’s to a new year filled with progress, purpose, and shared success.
All the best,
Scott Pantel and Henry Peck

Scott Pantel Chief Executive Officer, LSI Editor-in-Chief


Henry Peck Chief Business Officer, LSI Editor-in-Chief
Looking for inspiration for your medtech company as we head into 2025? Here’s what’s on the minds of some thought leaders in our space.
“We believe value creation in surgery and acute care is foundationally human. It flows from respect for and understanding of patients and care teams, their needs and their environment.”
“They’re spending more money on research for erectile dysfunction than menstruation. One is affecting 20% of men, and one is affecting 50% of the population.”

— Gary S. Guthart Chief Executive Officer and Member of the Board of Directors, Intuitive Surgical
On the company’s Q3 2024 earnings call, October 17, 2024
“To sustain something you need the portfolio, but people catch up and sometimes someone jumps ahead of you. The portfolio by itself is not ever going to be enough to have sustained high performance. It’s also the culture and the sales force.”

— Kevin Lobo Chair and Chief Executive Officer, Stryker
On the company’s Q3 2024 earnings call, October 29, 2024

“I’m loving that big tech from the consumer side and big pharma are going to bring more patients in.”

— Michael Farrell Chairman of the Board, Chief Executive Officer, ResMed
Speaking on the company’s Q1 2025 earnings call, October 25, 2024
“We’re constantly looking at the portfolio, to make sure it’s constructed in a way that’s aligned with our mission, aligned with our strengths… it’s a competitive world out there. We want to make sure we’re playing to our strengths, and we’ve got a number of those.”

— Geoff Martha Chairman and CEO, Medtronic
Speaking on the company’s Q2 2025 earnings call, November 19, 2024
— Yahel Halamish Early Stage Investor, Nina Capital
Speaking during the LSI Europe ‘24 Panel Discussion, “An Open Discussion on the State of Women’s Health”
“If I had a magic wand, I would have us all change from a system of sick care to truly health care … that will require moving patients and care outside the four walls of the hospital.”

— Lisa Earnhardt
Executive Vice President and Group President, Medical Devices, Abbott
Speaking at Milken Institute’s Future of Health Summit, “Innovation for All: Strategies for Maximizing Impact” panel discussion, November 13, 2024
“People often think that the only thing investors care about is big, meaning lots of patients. I completely disagree. Believe in the impact you can ultimately have on patients.”

— Todd Brinton Chief Scientific Officer & Corporate Vice President, Edwards Lifesciences
Speaking during the LSI Europe ‘24 Panel Discussion: “Behind the Scenes in Unlocking Disruptive Innovation”
Highlights: Boston Scientific expands its AFib platform, innovative cancer, urology, diabetes and dialysis companies secure later-stage funding, and early-stage neurotech, AI healthtech, and robotics innovators gain runway.
Series C: 2
Series D: 1
Series E: 3
Distribution Agreements: 1
Acquisitions: 1
Medtech innovation is hot!
Our sampling of medtech deals this past month shows a high volume of early-stage investment rounds
Series B: 2
Series A: 2
Grant/Pre-Seed/ Seed Funding: 7
Note: Data above is not indicative of total market activity.
Santa Clara, CA-based Cortex, which is developing a comprehensive platform for the treatment of atrial fibrillation (AFib), the world’s most common cardiac arrhythmia, is being acquired by Boston Scientific. Cortex’s technology will use pulsed field ablation in conjunction with diagnostic mapping capabilities to simplify workflows and improve procedural outcomes in patients being treated for AFib. Boston Scientific is pitching the company’s technology as a tool for physicians to develop ablation procedure strategies in complex AFib cases, and expects to close the deal for an undisclosed amount in the first half of 2025.
About 10.5 million U.S. adults—or 5% of the population—have AFib, according to new prevalence estimates published in September 2024 in the Journal of the American College of Cardiology. The findings suggest AFib is three times more common than previously thought. An aging population along with increasing hypertension, diabetes, and obesity rates in the U.S. have led to increased prevalence of the condition in recent years. AFib doubles the risk of mortality and is one of the most common causes of stroke, in addition to increasing risk of heart failure, myocardial infarction, chronic kidney disease, and dementia, and lower quality of life, according to study researchers. What’s more, it costs the U.S. healthcare system $26 billion annually, according to the American Heart Association.
In the thrombectomy space, Menlo Park, CA-based Inquis Medical closed a $40 million Series B financing round to support its advanced thrombectomy technology. Marshall Wace led the oversubscribed funding round, with strong participation from existing investors ShangBay Capital, Yu Star, EnPointe Ventures, and Pierre Lamond.
The privately held, clinical-stage company’s flagship Aventus Precision Thrombectomy System features proprietary tissue-sensing technology that provides operators with real-time information on the tissue composition in contact with the device. The system offers enhanced precision and control during procedures, according to Inquis.
Earlier this year, the company won FDA 510(k) clearance for the system, along with Inquis’ blood return system for peripheral indications. Inquis also indicated it is nearing enrollment completion in an FDA investigational device exemption (IDE) trial evaluating pulmonary embolism (PE) patients. The company expects the new funding to support completion of the IDE trial and support preparations for the launch of Aventus in 2025.
In the U.S., approximately 900,000 individuals are affected by venous thromboembolism (VTE) each year, with over 50% of deep vein thrombosis cases leading to PE, which is the third leading cause of cardiovascular death. Current lytic-free aspiration thrombectomy procedures are often limited by significant blood
loss and procedural inefficiencies, making Inquis Medical’s innovations a promising advancement in the field, according to the company.
Cardiff, UK-based LaennecAI, a pioneering medical AI company, has announced the successful completion of its pre-seed funding round, for an undisclosed amount. The company has secured investment from SFC Capital and OVC Ventures, along with grant funding from the Welsh Government, Innovate UK, and the National Institute for Health Research.
Laennec AI is focused on developing AI-powered, smartphone-based digital stethoscopes and companion applications for healthcare professionals and patients. The company aims to enhance the diagnostic capabilities of traditional stethoscopes by integrating artificial intelligence to analyze heart and lung sounds effectively. The company is currently in the early stages of product development and aims to target a global market that includes doctors, nurses, paramedics, and medical students.
PrognomiQ, a clinical-stage spin-out of Seer focused on developing a diagnostic system for early cancer detection and treatment, announced Series D financing of $34 million led by Seer, with participation from a new, large strategic investor in the diagnostics segment as well as from existing investors, including Catalio Capital Management, aMoon, Invus, and Bruker. The closing of this round brings PrognomiQ’s total funding to more than $135 million since the company was founded in 2020.
PrognomiQ will use the new funding to advance development of an early detection test for lung cancer, which the company plans to provide as a lab-developed test (LDT) and later as an in vitro diagnostics test. PrognomiQ was recently granted a Clinical Laboratory Improvement Amendment (CLIA) license to support the development of the LDT product, and it is currently enrolling a prospective 15,000-subject study in lung cancer, initiated in 2023 to support registration of the IVD product.
The company’s multi-omics platform combines proteomic data with genomic and metabolomic information, allowing for comprehensive insights into a person’s biological state. By leveraging advanced technologies, including Seer’s Proteograph Product Suite, PrognomiQ aims to overcome the historical barriers of broad-scale proteomics analysis, which has limited clinical applications until now.
Zap Surgical Systems has announced the successful closing of a $78 million Series E funding round, led by Qingdao Baheal Medical, with participation from other strategic investors. The new financing will advance the commercialization of the company’s ZAP-X gyroscopic radiosurgery platform, which delivers high-dose stereotactic radiosurgery (SRS) to non-invasively ablate brain tumors and other select intracranial conditions.
To deliver the radiation necessary for SRS, ZAP-X utilizes a modern linear accelerator to obviate the historical use of Cobalt-60 and the related costs and challenges of handling radioactive isotopes. ZAP-X also incorporates a unique self-shielded
design that typically eliminates the need for costly radiation vaults. By offering more cost-effective care, ZAP-X strives to bring world-class non-invasive radiosurgery to many more patients. The company was founded by John Adler, MD, inventor of the CyberKnife and the related field of image-guided therapeutic radiation. Since 1987, Dr. Adler has served on the faculty at Stanford University, being appointed the Dorothy and TK Chan Professor of Neurosurgery and Radiation Oncology in 2007.
Precision Neuroscience, a brain-computer interface (BCI) developer, closed a $93 million Series C funding round, from 21 unidentified investors. The round brings the device developer’s total funding to approximately $146 million.
The New York City-based company also recently reported positive results from a pilot study of its Layer 7 Cortical Interface
The flexible, thin-film implant, one-fifth the thickness of a human hair, conforms to the surface of a patient’s brain, with embedded electrodes for high-resolution mapping of brain activity and stimulation. The device was designed for a neurosurgeon to implant or retrieve without damaging brain tissue.
The implantation procedure—differentiated from the BCI device from Elon Musk’s company, Neuralink—does not require a craniotomy. Instead, physicians place the device with a novel, minimally invasive “micro-slit” technique using 400-micron-wide incisions in the skull.
UK-based Reneural Technologies Ltd., formerly known as CardioCrown, has raised £200,000 (approx. $259,000) in a pre-seed funding round led by SFC Capital, alongside undisclosed investors, with an additional £275,000 (approx. $356,000) in grant support from Innovate UK. The company is addressing the critical needs of stroke survivors by offering a comprehensive neurorehabilitation platform that integrates cutting-edge technologies including virtual reality (VR), robotics, electromyography (EMG), and functional electrical stimulation (FES). The platform is designed to improve patient outcomes, reduce healthcare costs, and ease the burden on clinicians by supporting remote care. (For more on the emerging neurotech market, check out the “Mind Blowing Progress: The State of Neurotech” panel discussion from the LSI USA ‘23 conference in Dana Point, CA).
London-based MintNeuro, developer of low-power semiconductor technology for minimally invasive neural implant applications, raised £1 million (approx. $1.3 million) in a funding round led by Empirical Ventures. Additional investment came from Parkwalk Advisors (through the Imperial College Enterprise Fund), Imperial College London, Plug & Play, Excellis Holding, and several prominent angel investors.
Neurological disorders impact over a billion people globally, and pose a significant social and economic burden. Current treatments, especially pharmaceuticals, are often ineffective or fail to provide long-term solutions. Neural implants offer a promising alternative to traditional technologies that can be bulky, power-intensive, and reliant on invasive surgeries that limit accessibility and increase patient risk, according to MintNeuro. MintNeuro’s technology was developed at Imperial College London.
ZenFlow, a South San Francisco, CA-based device company developing a minimally invasive treatment for urinary obstruction caused by enlarged prostate, or benign prostatic hyperplasia (BPH), has closed a $24 million Series C financing round. The round includes new investor Cook Medical, as well as existing investors Invus Opportunities, F-Prime Capital, Medical Technology Venture Partners and others. Wilson Sonsini Goodrich & Rosati advised Invus, F-Prime, and MedTech Venture Partners on the transaction.
Funds will be used to support the company’s PMA submission and to prepare for commercialization upon FDA approval of its Spring System, a minimally invasive treatment option for patients who suffer debilitating symptoms due to enlarged prostate. The Spring technology was designed with the patient experience in mind, and relies on a small spring-like coil that gently props open the urethra, restoring its normal function while preserving the natural anatomy. (We’ll dive into the dynamic BPH space in the next issue.)
Irvine, CA-based Beta Bionics, maker of advanced diabetes management solutions, has closed a $60 million Series E Preferred Stock financing round. The funding was led by new investor Wellington Management. Previous investors Eventide Asset Management LLC, certain funds managed by RTW Investments, Sands Capital, Soleus Capital, Omega Funds, Perceptive Advisors, and Marshall Wace also participated.
Beta Bionics’ flagship product, the iLet Bionic Pancreas, is an autonomous insulin delivery system that streamlines diabetes management and reduces the burden on patients, caregivers, and physicians. When iLet users “GO BIONIC” with their diabetes management, there is no carb counting or calculating insulin corrections throughout the day – the iLet determines 100% of the insulin doses and continuously learns and adapts to the user. The only input required to get started on the iLet is the user’s weight.
Proceeds from the financing will support expanded commercialization of the iLet and development of the product pipeline as
Beta Bionics works towards improving health outcomes and the quality of life of children and adults living with diabetes and other conditions of glycemic dysregulation.
In an agreement announced in September, the company will integrate Abbott’s glucose sensing technology with the iLet Bionic Pancreas automated insulin delivery system. The iLet will connect to Abbott’s FreeStyle Libre 3 Plus continuous glucose sensor, where readings every minute will help the iLet calculate insulin doses.
Quanta Dialysis Technologies, a Warwick, UK-based company dedicated to transforming kidney care through its Quanta Dialysis System has raised $60 million in a Series E round, led by Novo Holdings, Glenview Capital, and b2venture, with participation by existing shareholders.
The company designed its dialysis system to be compact but with the performance of traditional machines. In 2022, the FDA cleared the system to perform intermittent hemodialysis, sustained low-efficiency dialysis, and CRRT without needing bags. Recently, there was an additional FDA clearance for home hemodialysis, offering a high dialysate flow (500 ml/minute) system.
The company’s proprietary cartridge-based system, SC+, is a portable and compact system providing clinical efficacy utilizing high flow rates typically used on traditional machines. SC+ is designed to offer ease-of-use and flexibility, facilitating self-care and supporting dialysis patients across the entire continuum of care.
Quanta intends to use the funds to expand its commercial presence in the U.S. and continue to invest in innovation within the sector.
Hangzhou, China-based ROBO Medical, a developer of products for robotic endoscopy and lower limb rehabilitation, reported a Series A funding round, amount undisclosed, led by Hangzhou Guoshun Jianheng Venture Capital. The company’s Gastrointestinal Endoscopic Surgery Assistant Operating System is its primary endoscopic platform and is compatible with traditional endoscopes. The system has an independent robotic arm that can perform basic tasks in order to assist endoscopic surgeons without changing their operating habits. The company is also investigating a multi-channel robotic endoscopy system to provide a better field of vision during surgery while reducing physical strain on the surgeon.
ROBO Medical’s Jiexing Lower Limb Rehabilitation Exercise
Trainer is a wearable exoskeleton device for the leg, designed to treat patients suffering from hemiplegia, knee joint injury, or abnormal gait.
According to an article in the journal Nature (June 24, 2020), China has stepped up research efforts into medical robotics systems in the past two decades, driven by the clinical utilization of the systems; increased funding levels driven by national planning needs; advances in engineering in areas such as precision mechatronics, medical imaging, artificial intelligence, and new materials for making robots; and global competition.
Johnson & Johnson MedTech announced a strategic commercial distribution agreement with Minneapolis, MN-based Responsive Arthroscopy to expand its sports soft tissue repair solutions. The agreement strengthens Johnson & Johnson MedTech’s sports platform, enhancing its soft tissue portfolio to meet the rising demand for advanced solutions in the rapidly growing sports soft tissue repair market.
Responsive Arthroscopy offers a range of medical devices for arthroscopic procedures, focusing on knee, shoulder, hip, and foot and ankle surgeries. The company’s product lineup includes several suture anchor systems, including Valor (a knotless suture anchor with a unique SMART CINCH tension mechanism), Hornet (standard threaded suture anchors), and Super Hornet (which incorporates suture tape). The Falcon system is specifically designed for biceps tenodesis repair, featuring a unique nitinol securing fork with multiple screw diameter and length options. The company also offers the Lightning and Nighthawk suture anchors, which are designed to reduce surgical procedure costs and provide advanced fixation techniques.
OptraSCAN, a digital pathology solutions company, closed a $30 million Series B funding round, led by Molbio Diagnostics. The new investment will drive OptraSCAN’s continued innovation in digital pathology, enabling the expansion of its AI-driven solutions to healthcare providers worldwide, and reinforcing its commitment to making diagnostic technology affordable and widely available.
The company’s flagship product, the OS-Ultra Whole Slide Scanner, can scan a 15x15 mm area at 40x magnification in under 60 seconds, providing high-resolution images for comprehensive analysis and remote consultations. The OS-SiA Cytology Scanner integrates AI to enable real-time analysis of cytology slides, expediting the diagnostic process by delivering immediate
feedback and insights. The InnoQuant fluorescence scanner enables advanced imaging with multiplexing capabilities, catering to diverse research applications.
With the new funding, OptraSCAN aims to enhance its portfolio of cloud-enabled digital pathology whole slide scanning systems and AI algorithm suites designed to provide seamless, accurate diagnostics. It also plans to accelerate research and development, expand global sales and marketing efforts, and support the company’s mission of breaking down barriers to healthcare equity through advanced diagnostic technology. The company’s technology is designed to make digital pathology adoption affordable, providing flexible pricing models and scalable options to ensure that even small laboratories can access it.
GI Windows Surgical, a clinical-stage device company based in Westwood, MA, has secured $37 million in additional funding to support the continued clinical development and planned commercialization of the first fundamental breakthrough in anastomosis technology in both delivery and tissue fusion, its Flexagon self-forming magnet.
The Flexagon magnets are delivered by endoscope or similar devices to the site where they need to be positioned as a segmented string, where they then form into the ringlike formation needed to create the anastomosis. The magnets in a magnetic anastomosis compress toward each other, to cause necrosis in the tissue between them. As the tissue between the magnets dies, the tissue around them fuses between the two pieces of the organ in which they are placed.
This most recent financing was led by Orlando Health Ventures and joined by Rex Health Ventures, with participation from existing investors including Johnson & Johnson Innovation–JJDC, JCInvestco, Fred Moll, MD, and an additional strategic investor.
The company is developing a revolutionary technology employing magnetic anastomosis to enable less-invasive surgery, marking the first significant breakthrough in this field in over 40 years. According to Brian Tinkham, CEO of GI Windows Surgical, the company is targeting enrollment of an FDA IDE clinical trial and commercial launch in the U.S. in 2025.
Hi-Tech
Berlin, Germany-based Quantune Technologies, developer of a miniaturized laser spectrometer for continuous, non-invasive health monitoring, announced that it has secured €13.5M (approx. $14.3 million) in seed financing round. Out of the total, €8.5M (approx. $8.9 million) was in equity financing, led by U.S.-based Point72 Ventures and Munich-based Vsquared Ventures.
This funding comes on top of more than €5M (approx. $5.3 million) in public grants from the European Innovation Council and the Investment Bank of Berlin through the ProFIT program. The company will use the funds to support product development to meet regulatory standards toward global market entry, and to expand its detection capabilities and fine-tune the spectrometer’s design for even better compactness and user experience. Based on Quantune’s patented Quantum-Cascade Laser spectrometer, the wristworn device provides lab-quality precision in measuring biomark-
ers like glucose, lactate, and ketone bodies, making proactive and personalized healthcare accessible to all.
Perspectum, a leader in medical imaging quantification techniques, has been awarded a 2024 U.S. Food and Drug Administration Drug Development Tool (DDT) Research Grant to evaluate a novel biomarker aimed at addressing the unmet needs of patients with rare diseases. The nearly $250,000 research grant will fund Perspectum’s research in partnership with the Consortium for Autoimmune Liver Disease (CALiD) to investigate the efficacy of a novel biomarker which shows promise in preliminary research.
CALiD is one of the largest U.S. databases for primary sclerosing cholangitis (PSC), a disease which affects just over 36,000 in the U.S. Although rare, PSC is the fourth-leading cause of liver transplant. This biomarker could revolutionize the way rare diseases, including PSC, are monitored and treated, offering new hope to patients who currently have limited therapeutic options.
Impilo, a Philadelphia, PA-based digital health company, has raised $11.5M in a Series A funding round, led by Construct Capital with participation from GC 1 Capital, One Way Ventures, 2048 Ventures, and strategic angel investors.
Impilo offers a digital health platform designed to enhance athome healthcare delivery. The company’s services include remote patient monitoring device logistics, which streamline the procurement and distribution of medical devices and supplies directly to patients’ homes. The platform also features a unified data API that integrates health data from various connected devices, facilitating seamless communication between patients and healthcare providers. Additionally, Impilo provides robust patient support services, ensuring personalized assistance throughout the care journey. This integrated ecosystem aims to make high-quality healthcare more accessible and efficient for patients nationwide.
Soombit.ai, a South Korea-based medical artificial intelligence company, has raised $3.6 million in a seed round of funding, led by Silicon Valley VC firm Altos Ventures. The funding will support Soombit.AI in advancing AI solutions for radiology. The startup also plans to expand its technology to other imaging modalities, including CT. Soombit.AI develops AI-powered tools to assist radiologists with medical imaging tasks, particularly in reading chest X-rays.
Its flagship product, AIRead-CXR, generates preliminary draft readings for chest X-rays, helping radiologists detect abnormalities quickly and accurately. Soombit.AI leverages generative AI technology to improve the speed, accuracy, and efficiency of radiology workflows.
The industry’s most comprehensive and trusted global procedure database, with coverage of 300+ diagnostic and therapeutic procedures across 37 countries.


The medtech M&A landscape in 2024 has soared past the benchmarks of 2023, with innovation, VC and FDA support acting as the engines driving its ascent. As the industry gains altitude, the flight path indicates a bustling and strategically pivotal 2025.


Medtech ecosystem stakeholders, please fasten your seatbelts.
Mergers and acquisitions (M&A) as we approach the runway to 2025 are expected to be turbulent, with tailwinds provided by accelerated innovation in compelling frontiers such as AI, that promises to revolutionize healthcare, VC investment, and continued regulatory support of novel device technology. The industry will be navigating through pockets of high-stakes negotiations, where every acquisition feels like a daring ascent, and every partnership, a critical adjustment to stay on course. Buckle up—standing still isn’t an option at this altitude.
Just like the friendly skies, the medtech space is filled with companies determined to reach their destination: market leadership, propelled by innovation, strategic and synergistic alignments, and the pursuit of transformative patient outcomes. But in this race, only those who navigate the turbulence with adaptability and strategic collaboration will beat the competition. This year has seen some elevation in the M&A space, with high-dollar bets along with room for improvement as we prepare to mark a quarter of the way through this century.
Medtech venture investments and M&A are signaling a positive year in 2024, with activity reported through the third quarter set to beat full-year 2023 numbers, according to the Q3 2024 Medtech Licensing and Venture Report from J.P.Morgan and DealForma.
From Q1 to Q3 of this year, medtech venture activity totaled $16.1 billion across 554 funding rounds, according to the J.P.Morgan and DealForma report (see chart).
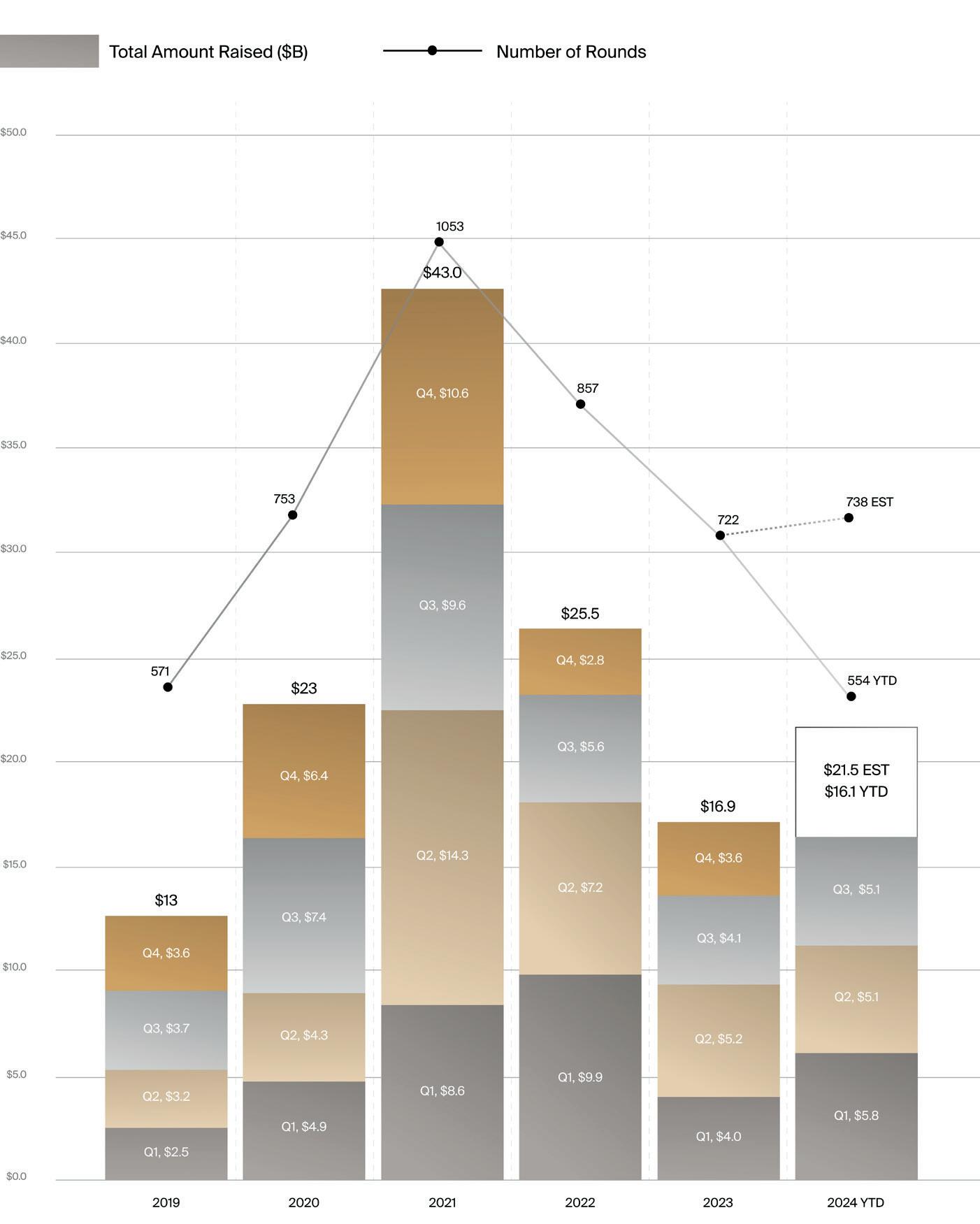
In Q3 2024 alone, venture investments totaled $5.1 billion across 154 rounds, up approximately $1 billion compared to the same period in 2023. Robust venture activity is helping 2024 to post over 27% annual growth.
Through Q3 2024, 195 medtech acquisitions totaled $47 billion in value. This compares to FY 2023, when 128 medtech M&A deals were announced, totaling over $50.1 billion.
In terms of medtech licensing and partnerships, the 744 medtech deals announced through Q3 2024 have disclosed $4.7 billion in total deal value, with $400 million in upfront payments, according to the J.P.Morgan and DealForma report.
Initial public offerings also showed signs of life this year, with a handful of companies raising over $700 million on NASDAQ and NYSE. This follows no U.S. medtech IPOs in 2023. The companies going public thus far in 2024 include Fractyl Health with $110 million in Q1, Tempus AI with $411 million in Q2, and CeriBell with $188 million in Q3.
Venture investment and licensing partnership upfront check sizes in the medtech sector remained relatively modest through the first three quarters of 2024, according to the J.P.Morgan and DealForma report. Most venture funding rounds for medtech companies came in at under $50 million, and upfront payments for R&D licenses concentrated below $10 million.
A cadre of large strategics have shelled out big bucks for highgrowth opportunities, primarily in the cardiovascular space, in 2024. Hefty deals include Johnson & Johnson’s $13.1 billion purchase of Shockwave Medical (this acquisition alone accounted for 23% of the annual total M&A investment, according to EY), the industry giant’s purchase of heart failure company V-Wave for $1.7 billion, and Edwards Lifesciences’ $1.2 billion dual purchase of JenaValve Technology, with a transcatheter valve aimed at aortic regurgitation, and implantable heart failure monitor company, Endotronix (see table below).
J&J is banking on big returns from Shockwave, with its successful intravascular lithotripsy technology used to treat atherosclerotic cardiovascular disease by applying sonic waves to disrupt plaques. Shockwave’s attractiveness lies in its value proposition: it reported $730.2 million in revenue for the full year 2023, representing an impressive 49% increase year-over-year. In July, J&J increased its operational sales guidance for the full year 2024 by $500 million to reflect the completion of the Shockwave acquisition. Company representatives also reported that Shockwave is well-positioned to become J&J’s thirteenth business with sales in excess of $1 billion annually. (For more on Shockwave’s journey to this epic exit, see the cover story in this issue, “Long Game Legends: Makower and Papiernik’s Playbook for Success.”)
383 of the 486 disclosed medtech venture rounds through Q3 2024 have been under $50 million, though there have been 36 so far, valued at over $100 million.
Some of the largest medtech venture rounds in Q3 2024 include the private rounds for Element Biosciences at $277 million (Series D led by Wellington Management with participation from new and existing investors, including Samsung Electronics, Fidelity, Foresite Capital, funds and accounts advised by T. Rowe Price Associates, Inc, and Venrock, among others) and women’s health-focused digital consumer app company Flo Health, at $200 million (Series C investment from global growth investor General Atlantic).
Medtech partnership and licensing upfront payments finished the first three quarters of 2024 with 11 deals under $10 million.
Note: Data shown here current as of November 22, 2024. Source: LSI Market Intelligence
Rather than continuing to expand and diversify their product lines, in this past year some of the sector’s biggest players streamlined their business and concen-
trated their portfolios in high-growth areas. As a result, divestments have become a major theme of recent industry dealmaking, according to the EY report.
As one example, in June, Becton, Dickinson and Co. (BD) acquired Edwards Lifesciences’ Critical Care product group, focused on advanced monitoring
solutions with advanced AI algorithms serving millions of patients globally, for $4.2 billion in cash, to enhance BD’s portfolio of smart connected care solutions. Prior to the acquisition, the Critical Care group had approximately 4,500 employees, with most based in Irvine, CA, and 2023 revenues totaling more than $900 million. For Edwards, the sale of Critical Care reflects the company’s sharpened focus as a structural heart innovator.
In August, Baxter International announced a definitive agreement under which global investment firm Carlyle will acquire Baxter’s Kidney Care segment, to be named Vantive, for $3.8 billion. Baxter first announced its intention to create a standalone kidney care company in January 2023, but a little over a year later, in March 2024, the company was in discussions to explore a potential sale of the segment. After reviewing the financial impact of potential separation pathways, management and the Baxter Board determined that selling the business to Carlyle should maximize value for Baxter stockholders and best position Baxter and Vantive for long-term success. Vantive offers products and services for peritoneal dialysis, hemodialysis, and organ support therapies, with more than 23,000 employees globally and 2023 revenues of $4.5 billion.
One reason to be positive about the industry’s underlying fundamentals going into 2025 is the record-breaking pace of new product FDA approvals. The FDA’s Center for Devices and Radiological Health (CDRH) noted in its 2023 Annual Report that it authorized 124 novel, firstof-their-kind devices in 2023 (excluding emergency use authorizations)—the highest on record in the Center’s history. CDRH continues to see high numbers of novel devices coming to market, with an almost five-fold increase in novel device authorizations since 2009.
The number of 510(k) approvals in 2023 rose for the fourth consecutive year, to reach a record 3,325, according to FDA, while pre-market approvals (PMAs) grew by a monumental 77% as review and approval timelines recovered from any lingering bureaucratic backlog following the COVID-19 public health emergency.
In another sign of an innovation-themed
future ahead, in 2023, CDRH granted Breakthrough Device designation to 167 devices, and granted marketing authorization to 29 Breakthrough Devices. Since the launch of the Breakthrough Devices Program, the agency has granted this special status to 921 devices as of the end of 2023, including devices originally designated under the Expedited Access Pathway program that started in 2015.
Medtech is also progressing into bold new frontiers, as ongoing breakthroughs in artificial intelligence (AI) hold the promise of making devices smarter and more personalized.
The FDA’s Digital Health Center of Excellence continues to foster innovation for new and emerging digital health technologies, including AI-machine learning (AI-ML)-enabled devices. To date, CDRH has authorized over 700 AI/ML-enabled medical devices, and more are under development.
Look out for a review of CDRH’s upcoming 2024 annual report here in The Lens!
As we embark on a new year, the medtech landscape is brimming with momentum. As illustrated over the past few years, medtech M&A has charted a consistent and steady course, with 2024 bringing fresh gains in both venture investments and deal-making—a trajectory poised to soar even higher in the year ahead. The engines driving this growth include advancements in transformative technologies like AI, robust FDA support for innovative devices, the global challenge of an aging population, and the urgent demand for cost-effective healthcare solutions. These forces have solidified medtech as a magnet for strategic buyers and investors, positioning the sector as a hub of opportunity and innovation. The very real challenges coming from global economies, geopolitical uncertainties, healthcare system constraints, and rising competition, among other factors, will test the resiliency and persistence of all medtech players, but also offer opportunities and rewards for companies agile enough to innovate and align with shifting market demands.
Even amid expected turbulence, the medtech industry has exemplified remarkable innovation, keeping it firmly on a flight plan toward its goals of financial success and improved patient outcomes into 2025. The spirit of patient-centered innovation
remains the industry’s jet fuel, and we can’t wait to see how the ecosystem will continue to soar to new heights to meet the evolving needs of patients.
Join us at our three upcoming LSI events in 2025 to meet the innovators driving our industry to the next level!

When it comes to revolutionizing medtech, two names carry heavyweight status: Josh Makower, MD, and Antoine Papiernik. These visionaries, both powerhouses in innovation and venture capital, dish out straight talk following Shockwave Medical’s epic exit on what it really takes to succeed in medtech. For entrepreneurs and investors aiming to make waves, they say the magic formula is all about grit, teamwork, a long-haul mindset, and a relentless commitment to a new form of investor collaboration.

Source: LSI Europe ‘24
As the sun set over Sintra, Portugal at LSI Europe ‘24, with wine in hand and a Mediterranean breeze in the air, medtech icons Josh Makower, MD, and Antoine Papiernik shared hard-won insights with the large audience gathered for their fireside chat. From what it takes to nail today’s device investments to lessons from Shockwave Medical’s monumental exit—and even a few thoughts on mountain climbing—these two mavericks served up the ultimate roadmap for medtech success.
Makower and Papiernik, who have known each other for more than 20 years and have both played key roles in the evolution of the medical device industry throughout their impactful careers, are now
collaborating at powerhouse European VC firm Sofinnova Partners, with Makower joining as an Advisory Venture Partner in early 2024. Sofinnova, still aglow from the monumental acquisition of intravascular lithotripsy (IVL) company Shockwave Medical by Johnson & Johnson (J&J) earlier this year, after becoming an early and committed investor more than a decade ago, was a key theme of their discussion.
In today’s dynamic medtech investment landscape, this blockbuster deal sends a clear message to innovators and investors that success hinges on a few key pillars. Makower and Papiernik lay it out: it’s all about the people, a mindset built for the long haul, and an unstoppa-
ble commitment to a new wave of collaboration reshaping medtech. Shockwave’s incredible journey—from startup in 2009, to its successful IPO in 2019, to a staggering $13.1 billion exit in 2024—is the kind of
It’s all about the people, a mindset built for the long haul, and an unstoppable commitment to a new wave of collaboration reshaping medtech.

Source: LSI Europe ‘24
victory that every innovator dreams of, and it’s proof that delivering on the right formula can turn ambition into industry-shaking success.
To kick off the evening, Makower asked Papiernik, Sofinnova’s Chairman and Managing Partner, to describe the magic formula that comes into play when he’s making medtech investment decisions. Both agreed that people are everything.
“Companies go through ups and downs, and technology goes through ups and downs, but the people really are at the heart of it,” says Papiernik, who joined the Shockwave Board of Directors in early 2014, at the time of its $12.5 million series
A financing. “The team at Shockwave, including the first person we met, co-founder Todd Brinton [MD; now Chief Scientific Officer and Corporate Vice President at Edwards Lifesciences; see his keynote with Makower at LSI Europe ‘24], impressed us. I think the most beautiful thing for me is really the path, and the incredibly talented people, including [CEO] Doug Godshall who executed on the plan.”
Ecosystem relationships build trust, and Brinton has worked closely with both Papiernik and Makower over the years. In addition to co-founding Shockwave along with Daniel Hawkins (whose entrepreneurial DNA has led him back to the helm of another early-stage venture in Khosla Ventures-backed Vista.ai) and John Adams, Brinton worked alongside Makower and team at Stanford Byers Center for Biodesign (now the Stanford Mussallem Center
for Biodesign) for 18 years in various roles, including as Innovation Fellowship Director. Brinton is also an Adjunct Professor of Medicine at Stanford University, where he has held multiple roles in cardiovascular medicine for almost 25 years.
Makower agrees that the decision to invest is always all about the people, including patients. “Find those people that you can build something amazing with. If you’re an investor, if you are an entrepreneur, find the investor that you can partner with and see your way through. Build something together to last, and a journey
“Find those people that you can build something amazing with.”
– Josh Makower, MD
multiple successful roles as a venture, general and special partner at NEA, to co-founding and growing Stanford Biodesign’s Innovation Process into a global approach to patient-centered health technology innovation, to being the founder and executive chairman at ExploraMed, a medical device incubator.
Papiernik touched upon another critical point to keep in mind when navigating company growth: when to grow the team.
“People are everything, and you need to understand when you need to grow the team, and be able to bring the appropriate skill set to the company. To grow Shockwave from seven early patients that showed that the technology was possible, to go from there to a billion-dollar company, required huge execution skills. To be able to find those is what’s difficult. Bringing on the right people at the right time is key.”

Source: LSI Europe ‘24
that is transformative—not only for your team and your company, but for the patients. That’s always going to be where the real goal is, that’s why we’re all here. We’re trying to do something good for patients.”
Makower’s passion stems from his
Makower spoke from his own experience as a serial founder, investor, and innovation builder. “The way to win is with patience and perseverance. I can remember when Impella CardioSystems went bankrupt twice, then it was reborn and over time
it was eventually bought by Abiomed. Abiomed itself was purchased for one of the biggest exits in the history of medtech at the time,” he says. (It was acquired by J&J in 2022 for about $17.3 billion.)
“For entrepreneurs, the story there is if you have a technology you believe in, and you have the data, you just have to stick with it. That’s the message to investors as well. That’s how real value is created. In this business, it’s not created overnight,” Makower continues.
“Shockwave took about 12 years, but some of the other companies that ultimately went there took longer than that, and I think we both have examples of that. Persistence is absolutely key,” added Papiernik.
“Shockwave took about 12 years… persistence is absolutely key.”
– Antoine Papiernik
Makower told the audience that the model for building medtech companies is unusual. “It’s not like other industries where there’s a markup, and then another markup, and another. This is really about derisking capital over time, and as you continue to invest even though the values don’t necessarily click up that much. You’re having a greater opportunity to get in early, and have the chance to have potentially in the long run the largest position in the company. But you only achieve that by following through.”
The Rise of a Powerful Investor Ecosystem
Both Makower and Papiernik noted a powerful phenomenon happening today in medtech investing that wasn’t the case 30 years ago: consortiums of investors. Makower noted that Sofinnova has always been excellent at attracting great investor partners to join a device company financing, and that has been a key ingredient in the firm’s recipe for success.
“If you look at this particular meeting [LSI Europe ‘24] and the list of investors, along with the sheer number of people at this meeting, 30 years ago there was not a single meeting like this, or an ecosystem or community of investors,” says Papiernik. “Today there are dozens and dozens of medtech investors at this conference, it’s

Source: LSI Europe ‘24
made a huge difference to the ecosystem. For start-ups, as you know it’s difficult to raise money, we all know that. But companies actually raise tens of millions and hundreds of millions today, thanks to the robust investment community that was built over the last 20-plus years.”
Makower also has an incredible passion for the industry and for patient-centered innovation, and has had an influential role in the creation of a diverse and thriving device ecosystem over the past 30 years. And, his vast and varied experience gives him a perspective of the space that not many have.
“The real opportunity I can offer others is to be able to see around some corners, and help guide CEOs or other people navigating it so that they can avoid the obvious pitfalls and also to speak the truth about what the challenges are going to be,” he says.
“The thing I love so much about this medtech community is that so many of you are really here to help each other,” he continues.
“There’s something special about medtech where we recognize it’s a challenge, we stand together, and we truly are helping each other. Even when there are direct competitors, it’s more about respect for the opportunity to create value for patients, and a recognition that there can be more than one that services a tremendously large need. Just stepping forward and helping the next generation, being a part of that community of backing early projects and then working together. It’s not VCs competing against each other, it’s about VCs collaborating to ultimately amass the funding necessary to get these deals to a tremendous value.”
“There’s something special about medtech where we recognize it’s a challenge, we stand together, and we truly are helping each other.”
– Josh Makower, MD
The two thought leaders also offered advice to the LSI Europe ’24 audience by comparing medtech innovation and company growth to the challenge of conquering a mountain.
“It’s about appreciating the journey you’re on, and being ready for it,” says Makower. “It’s like if there’s Mount Kilimanjaro or Everest in front of you. You don’t want to just be looking at Base Camp. You want to be able to look up to the top and appreciate, okay, there’s where we’re going. You bring the right partners or sherpas along with you, bring the right amount of food, the right protective clothing, and then you can scale it. If I can play some of that role, and help entrepreneurs prepare, that’s what gets me excited.”
Papiernik provided his investment viewpoint, pointing to the true scale of global company evolution.
“I remember back in the day thinking Base Camp was actually the top of Everest,” he says. “I thought yes, this is where we need to go, it was all about CE Mark, then you maybe have a path to the U.S., that’s the ultimate, the top of Everest.
But then you get there and you think blimey, absolutely not. The goal posts have kept being pushed back over the last 30 years. If you think that’s the top of Everest, you’re going to be disappointed.”
For some companies, handing their company off to a strategic acquirer might be the summit. But Makower describes a profound quote from Bill George, former chairman and CEO of Medtronic, who led the company for many years to become a growth story.
“I remember him saying on stage to an audience just this, ‘you know we are benefiting from all the investment that you guys are making, and you’re selling your companies way too early.’ He was taking those great businesses and then creating multi-billion-dollar franchises out of them. I think he was right, but it is an ecosystem, right? So, recognizing that the venture model has an arc to it, it’s about handing off
to the next arc, and that might be the summit for a venture investor. But then it becomes an opportunity for the strategics.”
Papiernik describes that he had several experiences where in hindsight his firm may have sold too early, one being CoreValve, with its transcatheter, transfemoral aortic valve replacement device. “Medtronic is probably happy that we sold CoreValve to them when we did. What if we had stayed? What if we had found the money? But hindsight is 20/20, of course.” (The 2009 deal included an initial payment of $700 million plus additional payments contingent upon the achievement of agreed milestones, with Sofinnova, its greatest shareholder, having led the Series A.)
However, with Shockwave, Sofinnova’s perseverance and faith paid off, and Papiernik and team got to reach the summit. Papiernik left the board after the company’s acquisition by J&J, when sales
boomed from $13 million to over $900 million at the time of the IPO. (Side note: The J&J acquisition is already paying off, with FDA clearance of the Shockwave Javelin Peripheral IVL Catheter this October, followed in November with the company unveiling positive first clinical outcomes with the device.)
Speaking to the CEOs in the audience, Papiernik says that kind of surreal deal sounds impossible, but obviously it is possible.
“I’ve lived it once in my career, but this one was a monumental exit. And, it’s a bellwether of what can be if you have the combination of the technology, the market, and the people. You can actually do this and you can find the money to get it done. This deal has transformed the way we look at companies, and we want more of that.”

Josh Makower, MD
The Yock Family Professor, Professor of Medicine and of Bioengineering, Stanford University Schools of Medicine and Engineering, The Byers Family Director and Co-Founder, Stanford Mussallem Center for Biodesign
Dr. Makower is the Yock Family Professor of Medicine and Bioengineering at the Stanford University Schools of Medicine and Engineering, and the Byers Family Director and Co-Founder of the Stanford Mussallem Center for Biodesign, a program he co-founded in 2000. He is also the Founder and Executive Chairman of ExploraMed, a medical device incubator that has created 10 companies over the past 20 years. Dr. Makower was also a longtime partner in NEA’s healthcare practice, advising the healthcare team and medtech/healthtech investing practice. In early 2024, he joined Sofinnova Partners, a leading European life sciences venture capital firm, as an Advisory Venture Partner, and Patient Square Capital as a Senior Advisor. Dr. Makower currently serves on the boards of Elevage, ExploraMed, Moximed, X9, Willow, and Coravin.
Dr. Makower holds over 300 patents and patent applications for various medical devices in the fields of cardiology, ENT, general surgery, drug delivery, plastic surgery, dermatology, aesthetics, obesity, orthopedics, women’s health, and urology. He received an MBA from Columbia University, an MD from the NYU School of Medicine, and a bachelor’s degree in Mechanical Engineering from MIT. Dr. Makower is a Member of the National Academy of Engineering, a Fellow of The National Academy of Inventors and The American Institute for Medical and Biological Engineering, and was awarded the Coulter Award for Healthcare Innovation by the Biomedical Engineering Society in 2018.

Antoine Papiernik
Chairman and Managing Partner, Sofinnova Partners
Papiernik is Chairman and Managing Partner at Sofinnova Partners, where he actively where he actively invests for Sofinnova Capital Strategy, the firm’s flagship investment strategy. He has been an initial investor and active board member in a number of publicly listed companies including Actelion (ultimately sold to J&J),ProQR, NovusPharma (sold to CTI), Movetis (sold to Shire), and Shockwave Medical (sold to Johnson & Johnson).
Papiernik’s trade sales of private companies include CoreValve (sold to Medtronic), Fovea (sold to Sanofi Aventis), Ethical Oncology Science (EOS, sold to Clovis Oncology), and Recor Medical (sold to Otsuka).
Papiernik has an MBA from the Wharton School of Business, University of Pennsylvania. He has been selected twice for the Forbes Midas List, an annual ranking recognizing the world’s top venture capital investors.
Founded in 1972, Sofinnova Partners is a deeply established venture capital firm in Europe, with 50 years of experience backing over 500 companies and creating market leaders around the globe. Today, Sofinnova Partners has over €2.8 billion under management.
Registration and applications to present at LSI USA ‘25, March 17-21 at the Waldorf Astoria, Monarch Beach, Dana Point, CA are now open!
Get in touch to learn more about this event, along with LSI Asia ‘25 and LSI Europe ‘25.

$68.9Bn
Size of the global orthopedic surgery market by 2030
30.5M
Global orthopedics procedures in 2024 (up 4.5% from 2023)
The fastest-growing orthopedic surgery procedure, at 9.4% CAGR worldwide
Driven by an aging population, robotic surgery innovations, sensors and other technologies to detect costly periprosthetic joint infection, and a move to ambulatory surgery centers, the global orthopedic surgery market is on track to reach a value of nearly $70 billion by 2030.
The orthopedic surgery market, one of the largest medical device markets in the world, is evolving and expanding, with a global forecast value of nearly $70 billion by 2030, according to LSI’s Market Analysis and Projections (MAP) database. Device and surgical innovations are addressing unmet needs in this space, leading to a transformation in healthcare delivery, improvement in patient outcomes, and an enhancement in the overall quality of life for a growing patient population whose lives are limited by bad knees, hips, shoulders, and other joints.
Demographic factors contributing to demand for device innovation include a growing population over age 65 with degenerative joint diseases and injuries that are requiring prosthetic implants in order to maintain a high quality of life, and the obesity epidemic. Joint replacement is typically considered once other treatment options such as medication, physical therapy, and injections have been exhausted and a person is still experiencing significant pain and limited function in a joint due to conditions like arthritis—meaning it is considered a last resort when other alternatives are no longer effective. Better and more personalized implant technology and computer-assisted surgery systems including robotics and 3D printing, plus the cost-effective transition of many orthopedic surgeries to the ambulatory surgery center (ASC) setting, are also fueling demand and growth. In 2024, 30.5 million orthopedic surgical procedures will be performed, a 4.5% increase from 2023, according to
LSI’s Global Surgical Procedure Volumes database. Orthopedic surgeries rank as the second most-performed procedure globally, behind general surgeries at 75 million, and cardiovascular procedures coming in at number three with 24 million globally projected for 2024, according to LSI’s Market Intelligence Platform.
The top orthopedic surgery performed globally, by far, is extremity fracture repair, with a projected total of 15 million for 2024, that is more than double that of sports medicine arthroscopies at 6.4 million. Following that is knee arthroplasties (4 million; total, partial, or revision), hip arthroplasties (3.6 million; total, partial, or revision), and other joint arthroplasties (740,000).
During the forecast period 2023 to 2028, the fastest growing orthopedic procedures worldwide are forecast to be major joint arthroplasties, with shoulders growing at the greatest rate, a 9.4% compound annual growth rate (CAGR), according to LSI research. This growth rate is partially because they are the smallest volume procedure today (421,200 estimated procedures worldwide in 2023) in comparison to knees and hips, and also due to the rapid adoption of reverse shoulder arthroplasty for proximal humerus fractures (the second-most common fragility fracture in older adults), that has expanded the number of patients eligible to receive this procedure. Knee replacement procedures are also increasing rapidly, at a 6.7% CAGR, and hip replacements at a 5.5% CAGR, according to LSI projections.
In terms of total orthopedic procedure
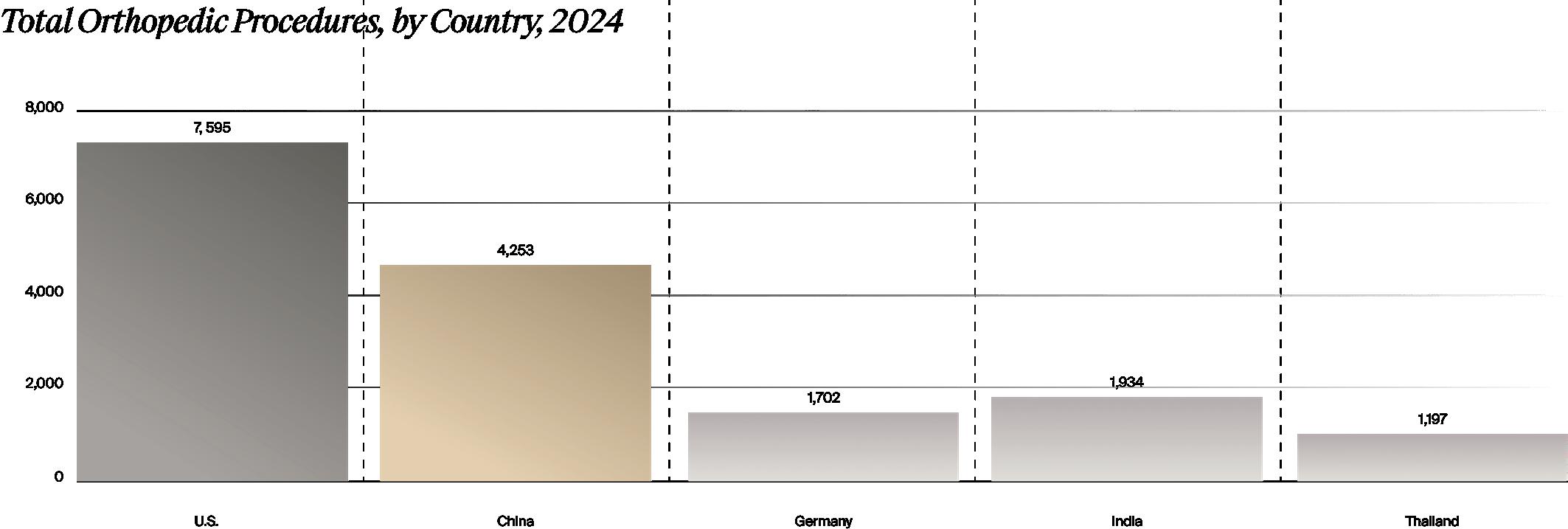
volumes by country, the U.S. comprises the highest share, with 7.8 million total orthopedic procedures forecast for 2024, followed by China at 4.7 million, then India at 2 million, Germany with 1.7 million, and Thailand at 1.2 million, according to LSI’s global procedure volumes data. However, with a 10.2% annual growth rate, China is projected to surpass the U.S. in orthopedic procedure volumes after 2030, according to LSI (see figure)
Several factors are at play in the U.S., in addition to an aging population: the country has a well-developed healthcare system that makes procedures accessible, and also the growth of same-day ASCs. The number of surgical cases shifting from hospitals to ASCs continues to increase, particularly in the field of orthopedics, and these lower-cost settings represent a major growth driver for orthopedic surgeries over the next few years, as discussed later in this article.
Drilling down into the U.S. numbers, LSI expects orthopedic procedures to grow at a moderate rate of about 3.3% from 2023 to 2028. The fastest growing procedures include elbow, knee, and shoulder arthroplasties.
The four largest joint replacement players globally are Zimmer Biomet, Stryker, DePuy Synthes, and Smith & Nephew. The dominance of these players can be attributed to their diversified portfolios of joint implants for both upper and lower extremities, and their aggressive stance on new product launches and tuck-in acquisitions of complementary technologies to maintain their market position.
The U.S. remains the largest country in terms of volume for orthopedic procedures. As a common topic of interest in the industry, we are not projecting China to surpass the U.S. in overall orthopedic procedure volumes until after 2030.
Source: LSI Market Intelligence Platform
10.2%
China is projected to see the fastest growth from 2023 to 2028
Device and surgical innovations to improve the accuracy and longevity/durability of implant placements, and reduce costly revision surgeries and implant-related infections, are among the growth drivers in the global orthopedic device market. In the following sections, we examine some key enabling technologies and examples of companies that are working to shape the future of orthopedic surgery.
Robotic arm-assisted arthroplasty was first introduced in orthopedics in 2006, and since that time the technique has been advancing to overcome technical challenges associated with manual procedures, increasing accuracy in alignment, and impacting clinical outcomes—in the knee in particular.
LSI and the device industry remain bullish on robotic-assisted orthopedic surgery, as the rapidly evolving innovation has already impacted general, gynecologic, urologic, and cardiothoracic surgery, to name a few. According to LSI’s estimates, the global orthopedic surgical robotics market in 2024 will reach $679 million.
Robotics has allowed for a number of advantages compared to traditional open surgery in orthopedics, particularly in the knee. This includes improved precision in surgery, as robotic arms provide real-time feedback and fine adjustments to ensure implants are positioned correctly and aligned with the patient’s anatomy. Robotics also enables personalized care, as the technology can tailor procedures to individual anatomical characteristics. Robotics can also minimize tissue damage and blood loss, especially with miniaturized technology, and facilitate minimally invasive procedures that can lead to fewer complications and revision surgeries (especially in combination with the use of customized implants), shorter hospital stays, and faster patient recovery. Robotics can also extend to postoperative rehabilitation, using exoskeletons and motion-capture systems to optimize mobility and strength recovery.
Advancements in near-field camera technology, device miniaturization, adaptive software algorithms, and AI and deep learning techniques for the optimization of surgical planning—including intraoperative plan adjustments—are poised to
enhance all robotic surgery capabilities, including orthopedic, in clinically relevant ways. The overarching goal, according to LSI alum THINK Surgical, a Fremont, CA-based developer of a miniature, wireless, open (meaning that it is implant- and vendor-agnostic) robotic system for total knee replacement, is to drive efficiency, reduce the physical and cognitive burden on surgeons and operating room staff and, most importantly, ensure improved and consistent patient outcomes. In November, THINK Surgical announced that its TMINI Miniature Robotic System had received FDA 510(k) clearance for use with the Freedom Total Knee, Freedom Titan Knee, and Freedom Primary PCK Systems; the MOBIO Total Knee System; and the LinkSymphoKnee. THINK Surgical’s implant-agnostic approach is a patient-centered trend and a real differentiator in this market niche, as individualized surgical plans for implant positioning and a more personalized approach to implant selection based on patient need aligns with the goal of robotic systems to improve patient outcomes.
Another LSI alum innovator in this space, the French company eCential Robotics, has developed and received FDA 510(k) clearance for an open, unified, and scalable robotic surgery solution adapted to bone surgery procedures. Its platform is designed as a single system integrating 2D/3D imaging, navigation and robotics.
The leading suppliers of orthopedic robotic systems include some of the largest orthopedics companies in the world: Stryker, Zimmer Biomet and Smith & Nephew. Stryker’s Mako system (developed by Mako Surgical Corp., which was acquired by Stryker in 2013 for $1.65 billion) is a robotic arm-assisted platform designed for knee and hip replacements. It combines 3D planning with robotic precision to help surgeons create personalized surgical plans and execute them accurately and efficiently. Stryker has continued to develop its Mako system, enhancing its capabilities for total knee, partial knee, and hip replacements. The company is also investing in research and development to bring new robotic solutions to market.
Zimmer Biomet offers the ROSA Knee System, the ROSA Partial Knee System, and the ROSA Hip System, a robotic-assisted platform that helps surgeons personalize knee and hip replacement procedures, and improve procedure accuracy and efficiency. Zimmer Biomet acquired the ROSA robotics portfolio, which included spine and brain applications, as part of
its 2016 acquisition of French robot-assisted surgery firm, Medtech. Zimmer Biomet then developed the knee application internally. In February of this year, the company received FDA 510(k) clearance for its ROSA Shoulder System for robotic-assisted shoulder replacement surgery, the world’s first robotic surgery system for this application.
Smith & Nephew, through its acquisition of Blue Belt Technologies in 2016, offers the Navio Surgical System, a handheld robotic system that assists surgeons in partial knee replacements. It uses a CTfree, image-guided approach for precise implant placement and alignment.
Robotic surgical systems for orthopedics and other specialties are continuously evolving to meet the needs of customers, including models that are transforming the same-day-surgery/ASC environment. Challenges still to be overcome include the high cost of purchasing and maintaining robotic systems, the often longer operating times, and the need for specialized training. In addition, a robotics approach is more often used for knee arthroplasties currently, versus hip and other joints. Larger, long-term, randomized controlled trials are needed to confirm their comparative effectiveness to other surgical techniques, and both short-term and long-term outcomes, and drive orthopedic procedure volumes in the coming years.
Affecting over 40,000 people in the U.S. annually, periprosthetic joint infection (PJI) is a rare and potentially devastating complication of joint replacement surgery in which pathogenic bacteria colonize the joint prosthesis, forming difficult-to-remove structures called biofilms. Biofilm infections are challenging to resolve, requiring long, invasive and expensive treatments that are often unsuccessful, resulting in high rates of permanent disability and early death. Recent retrospective analyses demonstrate that the current gold standard for treatment of PJI, twostage exchange arthroplasty, takes an average of 16 weeks and has a success rate under 50% after 12 months, highlighting the unmet need for faster and more efficacious treatment options.
Worldwide, the cost of PJIs totals over $3.3 billion, to manage the 1-2% of joint replacement procedures that develop infec tions, according to LSI (see the sidebar
for a compelling LSI “By the Numbers” examination of the estimated cost of infections in knee replacements alone).
PJIs represent a significant health challenge, with standard of care treatment involving a costly two-stage revision surgery and prolonged rounds of antibiotics, which can profoundly disrupt a patient’s life. There is a need for better methods to treat these rare but significant complications.
One novel solution to detecting joint replacement health and spotting complications early is orthopedic implant sensors. These devices are embedded in implants and monitor a patient’s condition and the implant’s performance in real time. The sensors can detect changes in the patient’s physical environment, such as pressure, temperature, strain, and alignment. They can also detect changes in the implant’s biochemical environment.
Orthopedic implant sensors can be used for a variety of applications, including providing real-time information to help surgeons position the implant, post-operative monitoring for help optimize the healing process, detect implant loosening or failure, and reduce the need for hospital visits and secondary procedures, infection detection through the use of thermal sensors, and fracture healing monitoring. The data from the sensors is transmitted to an external device such as a computer, using Bluetooth or radiofrequency waves. The data can then be analyzed by clinicians to help them monitor the patient’s treatment and make timely interventions.
One innovator in this area, LSI alum Canary Medical, is giving a “voice” to implantable medical devices through the use of proprietary integrative sensor technology. Its Canturio platform “listens” to implanted devices and collects data to monitor device performance. The company has strategically partnered with ortho implant leader Zimmer Biomet and its Persona IQ Smart Knee
A number of innovative companies are targeting the management of implant-related infections in different ways. These include Reselute, Osteal Therapeutics, and Solenic Medical
Start-up Reselute, which is developing new approaches to deliver antibiotics locally to sites of orthopedic infection, closed a seed financing round in May 2023 led by Duke Capital Partners, the early-stage venture investment arm of Duke University.
Osteal Therapeutics, whose CEO David Thompson presented at LSI Europe ‘23, is a clinical-stage biopharmaceutical
LSI BY THE NUMBERS

knee replacements (i.e. total knee arthroplasties) were performed worldwide in 2023
5.2
As explained by the LSI Market Intelligence team in an October 26, 2024 edition of The Numbers, our weekly LinkedIn publication, the global cost to treat periprosthetic joint infections (PJIs), in just knees alone, may run into the billions of dollars
Every year, millions of joint replacement procedures are performed worldwide While advances in surgical techniques and implants have improved outcomes, there’s still a significant post-op risk that looms large—PJI.
How significant is the problem?
The literature places PJI incidence between 1% and 2% for hip and knee replacements.
So...not that much, right?
Let’s dig into
According to LSI’s Global Surgical Procedure Volumes database, 3.8 million knee arthroplasties/replacements were performed worldwide in 2023. Over a fiveyear forecast period, global knee arthroplasty procedure volumes are projected to increase at a CAGR of 6.5%, reaching an estimated 5.2 million procedures worldwide by 2028.
Even at a seemingly small rate of 1% to 2% of these procedures resulting in a PJI, that translates to 38,000 to 72,000 infections annually. Treating PJI isn’t cheap either. The average cost to directly treat PJI after TKA ranges from $19,476 to $46,000 (systematic review by Szymski et al., 2024), depending on the approach.
Two-stage revisions are considered the standard of care for both hip and knee PJI. For knees, the mean cost of a two-stage revision PJI is ~$38,000.
That adds up fast.
At the low end, we’re talking a $701 million direct cost to treat—but the real figure could soar to $3.3 billion, especially when standard-of-care two-stage revision surgery is factored in.
And that’s just for knees! Hip replacements, though slightly less frequent with an estimated 3.4 million performed globally in 2023, carry similar costs. Small incidence, but a hefty bill, and impact on patients.
company developing a new category of combination drug/device therapies for orthopedic infections. It is leveraging the ability of concentrated, locally delivered antimicrobials to treat the bacterial biofilms typically responsible for musculoskeletal infections while minimizing off-target tissue exposure and associated adverse effects.
Focusing on implant preservation, Solenic, whose CEO James Lancaster also presented at LSI Europe ‘23, is developing a non-invasive medical device that utilizes alternating magnetic fields to eradicate biofilms that form on implants. Solenic’s work on a non-invasive solution to tackle PJI has already captured the attention of heavy hitters like Johnson & Johnson. In July 2023, Solenic closed a $5.1M Series A led by Johnson & Johnson Innovation–JJDC, Inc.
Advanced imaging and implant technologies, and minimally invasive surgical techniques have made ambulatory (aka outpatient or same-day) orthopedic surgery safer, more comfortable, and more cost-effective overall, and a replacement for hospital-based care in the majority of cases. In fact, initially fueled by surgical site of care challenges learned during the pandemic, orthopedics is one of the main categories of high-demand procedures being shifted to ASCs.
Another crucial driver for the move to ASCs: the Centers for Medicare & Medicaid Services (CMS) has recently approved reimbursement for total shoulder replacements, total ankle replacement, hip tendon incision, meniscal knee replacement, and hip and knee arthroscopy, to name only a few of the musculoskeletal conditions when performed in these non-hospital settings.
Over the next 10 years, orthopedic surgical procedures at ASCs are projected to grow by at least 25%, primarily due to cost. Common procedures performed in hos-
pital outpatient departments cost significantly more than in an ASC. In fact, an ASC can perform the same procedures for up to 144% less, due to increases in healthcare provider workflow by focusing on one specialty, and by avoiding hospital room charges, nursing services, and other hospital-related costs, according to studies.
And, these trends mean significant savings for the healthcare system. Non-Medicare patients choosing ASCs for procedures (all procedures combined, not just orthopedic) create $42.2 billion in annual savings in healthcare spending, while Medicare patients being cared for in an ASC setting result in $4 billion in annual savings, according to the Ambulatory Surgery Center Association (ASCA).
Medical device companies are also fostering growth in this area by catering to their ASC customers, including financing plans for equipment, special payment plans for pricier devices such as surgical robots, and dedicated sales teams.
While facing challenges similar to robotics, chiefly higher cost and lack of clinical data, in addition to regulatory and quality control concerns, 3D printing represents an accessible approach to custom implants designed to the patient’s specific physiology. By combining medical images from X-rays, CT, MRI, or ultrasonic scanning, 3D printing can be used to create patient-specific implants with almost the same anatomical structures as the injured tissues.
Potential benefits to this emerging specialty include faster recovery time for patients due to the precise fit of the custom implant, improved range of motion, better osteointegration due to the implant being made of alternatives to solid metal such as metal lattice material that promotes osteointegration, increased bone strength, and lower implant cost. 3D printing technology has been used to create implants for a variety of orthopedic procedures, including knee replacements, spine implants, and
ankle replacements.
One innovator in this space is LSI alum restor3d, that has developed 3D printed, personalized orthopedic implants for use across the entire human body. The company leverages a proprietary porous implant architecture to encourage bone growth through the implant and maximize graft packing.
Another innovative approach to improving patient outcomes and quality of life, specifically in patients with mild to moderate osteoarthritis who want to preserve their knee joints, has been developed by Fremont, CA-based Moximed
The company’s FDA-cleared technology, the MISHA Knee System, is the first implantable shock absorber for the treatment of medial compartment knee osteoarthritis, bridging the gap between chronic knee pain and total knee arthroplasty. The polymer implant is placed on the medial knee and moves with the natural joint, reducing about 30% of the peak force on the knee with every walking step, according to the company.
And, investors believe in Moximed’s approach. This past August, Moximed closed on a $61 million Series D preferred stock financing, with an option to close on up to an additional $30 million. The round was led by Elevage Medical Technologies, a Patient Square Capital platform, with participation from new investors Cormorant Asset Management and Warren Point Capital and existing investors New Enterprise Associates (NEA), Future Fund, Advent Life Sciences, Gilde Healthcare, Vertex Ventures HC, GBS Venture Partners, and Morgenthaler Ventures.
Driven by the aging population, there is a huge unmet need to introduce solutions that, like Moximed’s technology, orthobiologics, and other innovative strategies, bridge the gap between chronic knee pain and joint replacement.
The content in this article is based on LSI’s Orthopedic Market Dive webinar, presented on November 13, 2024 by LSI’s Senior Director of Market Intelligence, Nick Talamantes, with contributions by Henry Peck, LSI’s Chief Business Officer.
Looking for more global orthopedics market intelligence and insight? Access LSI’s continuously updated Market Intelligence Platform




Recent green lights from the FDA and CMS are finally igniting this long-awaited market that 10 years ago faced an uncertain future, unlocking an innovative potential treatment for millions battling uncontrolled hypertension.
Current global market share leader $52.5M
RDN catheters, worldwide market forecast, 2024 22%
CAGR (2023- 2028)

Hypertension, the stealthy killer behind cardiovascular and kidney disease, affects 1.3 billion adults worldwide and costs the U.S. healthcare system a staggering $131 billion annually. Despite lifestyle changes and medication, millions suffer from treatment-resistant hypertension, a condition that defies even aggressive pharmaceutical regimens.
Enter catheter-based renal denervation (RDN) — a novel interventional approach promising relief where pills fail. Once a subject of both excitement and skepticism following a high-profile clinical stumble in 2014, RDN is back in the spotlight with renewed momentum. Over the past year, landmark FDA and CMS approvals have reignited interest and in-
vestment in this space, following years of product modifications and clinical studies, paving the way for this innovative therapy to gain traction and potentially reshape hypertension care.
Let’s unpack how RDN works, how the market has evolved and where it stands, and the questions still on the horizon.
The sympathetic nervous system is an important contributor to hypertension, partly through renal sympathetic nerve activity. Antihypertensive medications that work by sympathetic nervous system modulation are often ineffective in inhibiting sympathetic outflow, and that’s where RDN comes in.
RDN is an innovative, minimally invasive procedure targeting drug-resistant, uncontrolled hypertension. It targets the nerves around the kidneys, which play a key role in regulating blood pressure. The procedure involves the percutaneous insertion of a steerable ablation catheter to the renal artery through a femoral sheath and guide catheter, treating each renal artery in sequence. By disrupting these nerve signals, RDN aims to consistently lower blood pressure, for patients with inadequate blood pressure control despite lifestyle modification and antihypertensive drug therapy.
Different modalities have been developed to treat the renal nerves, with the two leading methods being radiofrequency (RF) ablation and ultrasound (US) ablation. Medtronic’s Symplicity Spyral system uses heat generated by radiofrequency energy to ablate renal nerves, while US ablation, employed by ReCor Medical’s (parent company Otsuka Medical Devices Co., Ltd.) Paradise system, delivers high-frequency ultrasound waves to target renal nerves, causing localized damage.
Others developing RDN technologies include Ablative Solutions, that is using its Peregrine System Infusion Catheter to deliver neurolytic agents such as alcohol to targeted areas as an alternative to traditional thermal energy , along with Mercator MedSystems, and DeepQure, with its laparoscopic RDN device, HyperQure The large strategics Abbott, Boston Scientific, and Johnson & Johnson are also assessing the technology.
It’s important to touch upon how far the market for RDN systems (catheters and generators) has come over the last decade, as significant challenges have paved the way for today’s successes. The global market for RDN systems was growing and appeared promising in 2013-2014, prior to publication of the
SIMPLICITY HTN-3 pivotal trial results that involved the Medtronic Symplicity Spyral system. It was the first randomized, sham-controlled study evaluating RDN for resistant hypertension. The highly anticipated study found no significant difference in blood pressure reduction between the RDN group and the sham procedure group after six months, raising serious questions across the clinical community about the therapy’s efficacy, and highlighting the need for refined techniques and patient selection criteria.
These confounding results caused a major disruption in the RDN market. Following the trial, skepticism grew about the efficacy of the technique as a treatment for resistant hypertension, with some citing that it could be the end of the road for the controversial therapy. This led to a pause in RDN device development, a decline in investor confidence, and a significant slowdown in clinical adoption. At least 20 companies worldwide were marketing or developing RDN at the former height of the market, but following the publication of the SIMPLICITY HTN-3 results, three of the four largest companies involved in RDN dropped out of the market or truncated their development programs, including Boston Scientific, Covidien (now a unit of Medtronic), and St. Jude Medical (now a unit of Abbott).
As we all know and love, the medical device community is known for its persistence, and sometimes innovation takes time. This reality check in RDN prompted a reevaluation of study designs, patient selection criteria, catheter design, and procedural techniques. Subsequent trials, including SPYRAL HTN-OFF MED and RADIANCE-HTN, were designed with stricter protocols and demonstrated positive results, restoring confidence in the technology.
These positive developments, followed by evolution in the space, have helped the RDN market recover and gain renewed momentum in recent years. Adding further validation, 2023 European Society of Hypertension treatment guidelines recommend RDN as a Class II treatment option for patients with poorly controlled hypertension, meaning it can be considered as a potential treatment choice for individuals whose blood pressure remains high despite other medications and lifestyle modifications. And, it has recently gained landmark support of U.S. regulators.
In November 2023, a huge milestone was achieved: the FDA cleared both the Medtronic and ReCor Medical systems, based on multiple positive sham-controlled trials. Both products, that were given Breakthrough Device designations, have broad indications for the treatment of hypertension when lifestyle modification and medications fail to control blood pressure.
The Medtronic Symplicity Spyral RDN System is a minimally invasive procedure that delivers radiofrequency energy to nerves near the kidneys that can become overactive and contribute to high blood pressure. After sedation, the doctor inserts a single catheter into the artery leading to the kidney. Once the tube is in place, the doctor administers energy to the system to calm the excessive activity of the nerves connected to the kidney. The tube is removed, leaving no implant behind.
Recor Medical’s Paradise Ultrasound RDN System treats hypertension by ablating the nerves around the renal arteries, disrupting the overactive sympathetic nerves that can cause hypertension. The system employs the SonoWave 360 transducer that delivers a complete 360-degree ring of energy, reaching approximately 80% of the sympathetic nerves around the renal artery. In addition, a HydroCooling system circulates sterile water through the balloon during the procedure for a cooling effect that helps protect the arterial wall, according to the company.
As further support for the “new and improved” field of RDN, Medtronic and Recor Medical recently announced Medicare reimbursement for their respective technologies.
In August 2024, the Centers for Medicare & Medicaid Services (CMS) issued the Final Rule for FY2025 Hospital Inpatient Prospective Payment Systems, which includes a New Technology AddOn Payment (NTAP) for RDN procedures using ReCor Medical’s Paradise ultrasound renal denervation and Medtronic’s Symplicity Spyral catheter. The NTAP went into effect on October 1, 2024, and will be in place for three years.
Then, on November 1, 2024, the Centers for Medicare & Medicaid Services (CMS) granted transitional passthrough (TPT) payment for procedures using the Medtronic Symplicity Spyral RDN catheter and Recor Medical’s Paradise Ultrasound Renal Denervation system, under the Medicare Hospital Outpatient Prospective Payment System. Going into effect on January 1, 2025, TPT offers incremental reimbursement payments for outpatient procedures performed with RDN for Medicare fee-for-service beneficiaries.
The market for RDN catheters, relatively small at the moment due to the emerging US market, is projected to increase at a CAGR of 22% globally from $47.6 million to $128.7 million, during the forecast period 2023 to 2028, according to the LSI Renal Denervation Devices Market Snapshot 2024-2028. Market growth will be driven by trends in procedure volumes and corresponding unit growth, fueled by recent U.S. FDA and CMS approvals, the growing body
of clinical evidence, and increasing prevalence of resistant hypertension. Growth will be somewhat restrained by downward pricing pressure, due to the effect of increased market competition over the forecast period.
At present it appears that the target patient population for RDN will be relatively small in comparison to the size of the population with treatment-resistant hypertension, restraining long-term market potential, although market growth is forecast to be in the double digits over the forecast period 2023-2028.
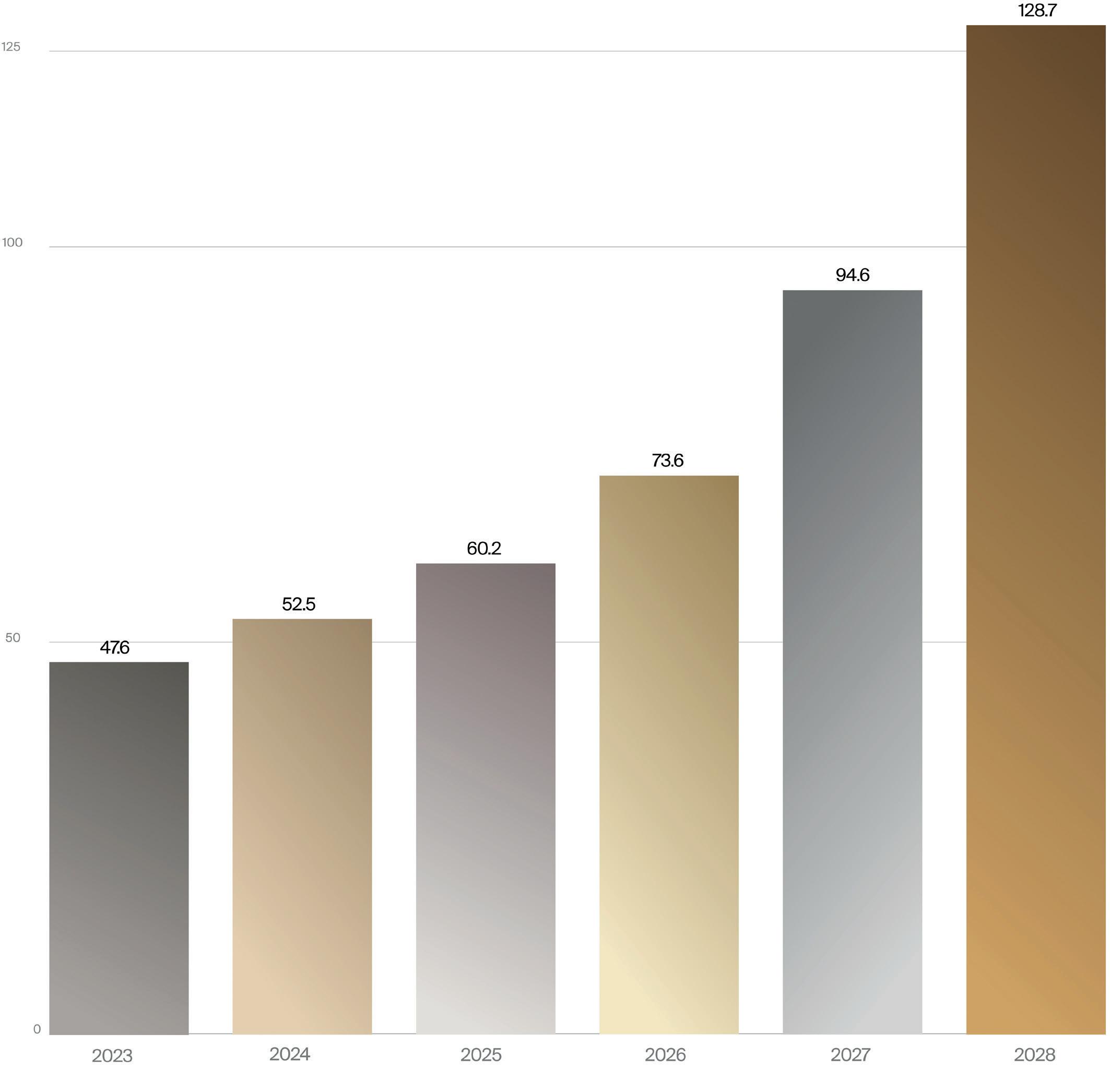
Source: LSI (From the Renal Denervation Devices Market Snapshot 2024-2028)
As of 2023, Medtronic accounted for the largest share of the global market for RDN catheters, according to the LSI report. The company has remained steadfast in its commitment over time to develop its RDN program in order to capture the significant opportunity that is present in the treatment-resistant hypertension patient population.
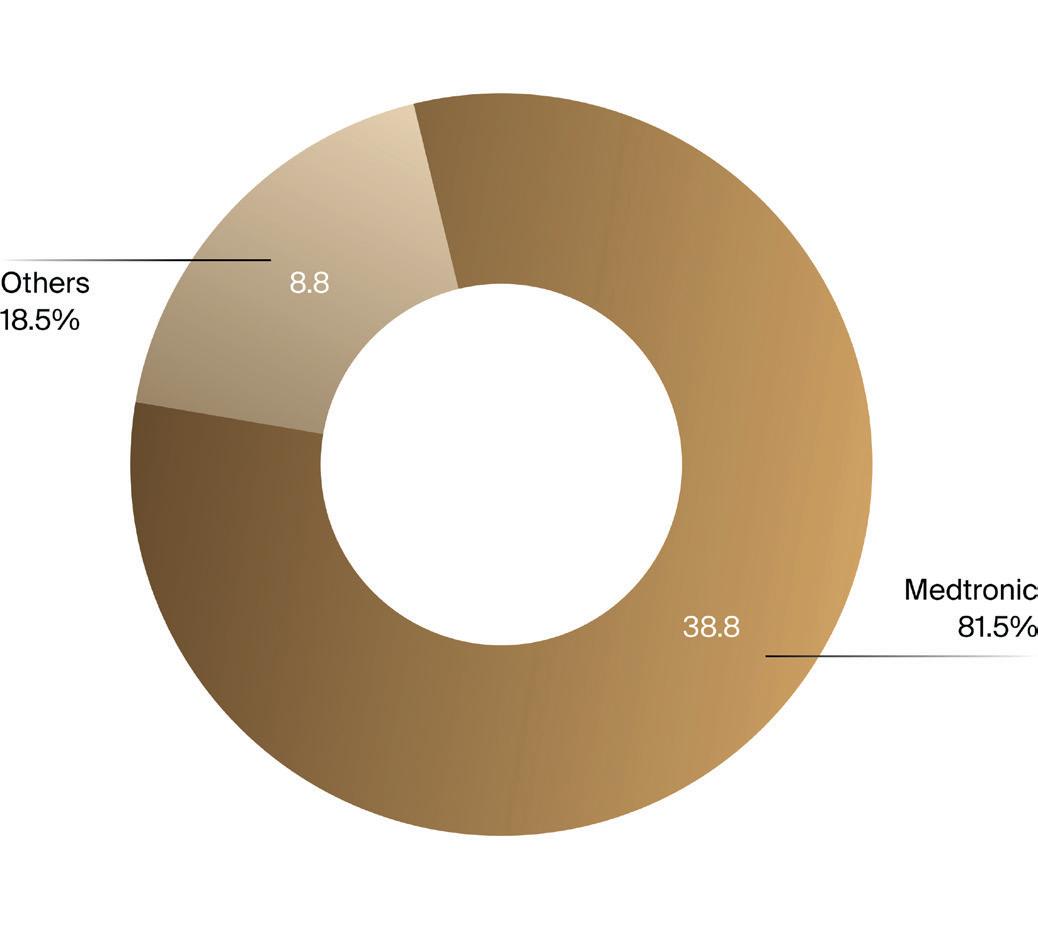
Source: LSI (From the Renal Denervation Catheters Market Snapshot)
Recent positive clinical results over the last year have provided further evidence, and fuel for market growth, on the efficacy and safety of current RDN systems for managing treatment-resistant hypertension.
The RADIANCE II trial, with results published in February 2023, demonstrated that ultrasound-based RDN significantly reduced blood pressure in patients, even without the use of antihypertensive medications. Similar results were seen in the SPYRAL HTN-OFF MED and RADIANCE-HTN SOLO trials, which showed reductions in blood pressure comparable to the effect of a single antihypertensive drug. These studies reported no significant safety concerns .
Real-world data from the Global SYMPLICITY Registry and the Global Paradise Registry reinforce these findings. In the SYMPLICITY Registry, a reduction in systolic BP of 13.2 mmHg at six months and 16.7 mmHg at three years was observed. These registries have also confirmed the safety and durability of RDN.
At TCT in late October, Medtronic announced new data from its global, randomized, sham-controlled SPYRAL HTN-ON MED trial showing that subjects who underwent the RDN procedure had significantly greater reductions in 24-hour ambulatory systolic blood pressure (ABPM) and office-based systolic blood pressure (OSBP), compared to sham patients at two years.
The clinical spotlight on RDN is burning brighter than ever, with mounting evidence cementing its potential to tackle stubborn hypertension and its promise for widespread adoption. Yet, the story isn’t finished—ongoing trials are sharpening the focus on its long-term effectiveness and uncovering which patients will benefit most, setting the stage for the future of RDN in treating this difficult health condition.
In addition to the clarity that future clinical investigations will bring, there are still other open questions about renal denervation.
One area of major interest is how to predict which patients will respond to the intervention, with studies showing that 20% to 30% do not have lowered blood pressure after undergoing denervation.
Ongoing concerns with RDN and future focus areas are perhaps best summarized by conclusions cited in this comprehensive review article, published by the American Heart Association in August 2024: “Renal Denervation for the Treatment of Hypertension: A Scientific Statement from the American Heart Association”:
Although further research is needed, particularly in the realms of patient selection and long-term efficacy, RDN is a promising new therapeutic approach for some patients with uncontrolled hypertension, particularly patients with RH or who have multiple medication intolerances. Ideally, individual characteristics that predict response will be identified to enhance the success of the procedure.
A multidisciplinary team approach that includes hypertension specialists and proceduralists is important both for identifying the right candidates for RDN and for following them after the procedure.
Efforts to educate patients and healthcare practitioners about how to realistically assess the benefits and risks of RDN for each individual person will be crucial for its uptake.

Data sourced from LSI’s Global Procedure Volumes Database, which provides trusted coverage of 300+ diagnostic and therapeutic procedures across 37 countries.

Connection, collaboration, community, and transformative opportunities. LSI events bring it all together, with incredible medtech innovators from around the world, in world-class venues like the Ritz-Carlton, Penha Longa Resort in Sintra, Portugal.
Relive a few moments from the unforgettable LSI Europe ‘24 event this past September, and stay updated on our three upcoming 2025 events at lsiusasummit.com.

An Open Discussion on the State of Women’s Health Panel Discussion
Yahel Halamish, Nina Capital; Andrew Pieprzyk, Hologic; Regina Atim, Clinicians Touch Healthcare Solutions; Sharon Handelman-Gotlib, Shela Health

Medtech’s Conundrum: Where Will Capital and Liquidity Come From? Panel Discussion
Michael Wasserman, Elevage Medical; Bill Little, Orchestra BioMed; David Kereiakes, Windham Venture Partners; Alexander Schmitz, Endeavour Vision; Diana Saraceni, Panakes Partners

Burton Tripathi, Beyeonics Vision

Lasry, FaceHeart

“Value-Add” is a Two-Way Street: Building Success Through Strong Investor-Founder Partnerships - Fireside Chat
Marta Zanchi, Nina Capital; Christina Vallgren, Terapet SA

SIA Story: the Good, the Bad, and the Ugly of M&A Panel Discussion
Todd Cruikshank, SIA; Lauren Lacagnino, Integra LifeSciences; Alexei Mlodinow, SIA







Backed by fresh NIH grants, Series A funding, a CPT code, FDA IDE pivotal trial approval, and crowned with a Best Invention of 2024 award, Sonavex exemplifies the playbook for medtech success. CEO David Narrow reveals how embracing challenges, laser-focused execution, and prioritizing market access, reimbursement, and adoption-driving clinical evidence have propelled the company—with the goal of revolutionizing care for hemodialysis patients and their vascular access lifeline.
Built at the crossroads of innovation and ambition, Baltimore, MD-based Sonavex—spun out of Johns Hopkins’ Center for Bioengineering Innovation and Design—is poised to shake up the hemodialysis market and the critical area of vascular access.
The company’s CEO, David Narrow, who began his career as a biomedical engineer and received his Masters from Johns Hopkins University, is a driven entrepreneur and proud dad. He was included in Forbes’ “30 Under 30 in Healthcare” list in 2016 and in the Baltimore Business Journal’s “40 Under 40” list in 2017. He previously worked with multinational device companies to commercialize their technologies and provide long-term business strategy as a healthcare consultant at Health Advances, LLC.
He counts among his mentors and supporters, in addition to his family, Sonavex co-founder and William B. Kouwenhoven Professor of Computer Engineering, Radiology and Biomedical Engineering at Hopkins, Jerry Prince, PhD; Sonavex co-founder and microvascular reconstruction surgeon Devin O’Brien-Coon, MD, MSE; Sonavex Board Director and former VP at Becton Dickinson, Bob Hallenbeck; and kidney patient advocate Terry Litchfield.
LSI caught up with Narrow in early November, just after Sonavex’s EchoMark, an ultrasound-visible marker implanted to monitor a hemodialysis fistula and reduce dependency on problematic central venous catheters (CVCs), was recognized by Time magazine as a Best Invention of 2024. Joining trailblazing tech like Dexcom’s Stelo OTC glucose monitor and the BiVacor total artificial heart, Sonavex is breaking down barriers in the high-need field of vascular access for dialysis patients, standing tall among groundbreaking advances in gene therapy, robotics, AI, and beyond.
Here, Narrow discusses what it takes to pursue a novel technology, his team’s achievements and upcoming milestones, his advice to other medtech entrepreneurs, and his experience as an LSI event alum.
Q: What is the unmet clinical need for your technology?
“The area that we focused on with our first solution is reliance on central venous catheters (CVCs) for dialysis patients. The vast majority of dialysis patients in this country crash into dialysis in the hospital, and that’s where they discover they have end-stage renal disease [ESRD], or kidney failure. Immediate, life-saving dialysis is delivered through a CVC that is placed from their neck into their heart. These catheters are designed to be very short-term stopgap solutions, because of the host of dangerous and expensive complications associated with them. They often cause serious bloodstream infections, leading to costly hospitalizations, and the mortality rate is multiple-fold greater than a patient on a more permanent type of dialysis access with an arteriovenous fistula, or AVF, in which a vein is surgically connected to an artery in the arm. Unfortunately, once placed, fistulas aren’t ready to use out of the gate. They take time to undergo a process called maturation, where that vein will enlarge and enable the flow rate to increase, such that the dialysis machine can be connected to the fistula using needles for regular dialysis therapy. Surprisingly, the current av-
erage time to transition from a catheter to a fistula is about seven months, and during that time, life-threatening complications can occur with the CVC. It’s a major issue for patients and a significant burden on the healthcare system.
What surprised us during our discovery process is that vascular remodeling happens quickly, but a key driver for the long delay in fistula use is that an ultrasound exam is typically needed to inform the care team of the status of a patient’s fistula, or if there’s a problem that requires an intervention. The standard of care right now is for the patient to go to a separate appointment in a specialized lab, where a trained sonographer, registered vascular technologist, or physician completes an ultrasound exam of the arm. This is due to the fact that the staff responsible for delivering dialysis to the patient multiple times per week, or regular check-ups if they are doing dialysis at home, is not trained in ultrasound. For a host of reasons, it is often challenging for these patients to commute to a specialized vascular lab for their ultrasound appointments. The impact here is that the interval for getting the ultrasound data, and transitioning from CVC to long-term fistula use for dialysis, is unnecessarily delayed. The use of ultrasound to assess fistula maturation during existing patient touchpoints is an ideal pathway to improving patient outcomes, but its availability has been limited by the specialized skill set required to operate conventional ultrasound.
What we’re aiming to do is bridge that gap by empowering incenter dialysis techs, who are already frequently engaging with these patients, to obtain this ultrasound data that the care team needs to assess fistula status. We’ve created a solution that empowers these dialysis techs, without any ultrasound training, to get a vascular lab-quality ultrasound to assess AVF maturation in a matter of seconds.”
Q: How does Sonavex’s technology work?
“Our solution empowers any member of the patient’s care team to collect real-time AVF volumetric flow rate, diameter, and depth information. Our small, highly echogenic, bioresorbable polymeric implant, EchoMark, is placed underneath the fistula at the time of fistula creation, where it acts as an ultrasound reflector. Its placement adds only a few minutes to the patient’s procedure.
It works with our companion device called the EchoSure, an automated, 3D Duplex ultrasound system designed to allow any member of the care team to collect AVF volume flow, diameter and depth information. The system uses AI and other image analysis algorithms, such that the technician can simply press a button and capture the data. That data is then sent through our HIPAA-compliant cloud to the physician, so that they can then inform the dialysis center about a patient’s fistula status and determine cannulation suitability.
By empowering members of the care team who routinely care for dialysis patients to collect this essential information, EchoSure facilitates early decision-making to remove the catheter at the earliest possible time.”
Q: Walk us through the incredible progress your company has made just in the past few months.
“Over the past few months there were several announcements in a short period of time, but the reality is that this news was the culmination of a lot of hard work over many years that just came together all at once. Much of the traction was catalyzed by the clinical data that we were able to publish from a 20-patient U.S. trial,

Source: Sonavex (used with permission)
which demonstrated the safety profile of EchoMark, and the ability for non-sonographers to capture ultrasound data with EchoSure comparable to data captured by trained sonographers. Most importantly, when ultrasound is performed more frequently, we can reduce the time to AVF maturation from seven months to several weeks. That was a meaningful milestone for us that catalyzed some of these other exciting developments.
Shortly after that data arrived, in October 2023, we received our first Sonavex-specific CPT code from AMA [the American Medical Association]. This was something our team was working on in parallel for quite some time.
It’s critically important to ensure that there are both appropriate codes to describe your technology, and have the data necessary for long-term coverage, to ensure incentive alignment. The CPT process was an interesting and eye-opening process for us, but ultimately we were very fortunate to have support from a key physician specialty society to drive success.
After that news, in December we received IDE approval from the FDA to run a 304-patient randomized control trial, MAFASA [Maturation of Arteriovenous Fistula With Automated Sonography Assessments]. This trial is designed to provide Level 1 data to demonstrate how the use of EchoMark and EchoSure will reduce time to AVF maturation, and reduce the costs of managing dialysis patients as compared to standard of care, by providing more timely fistula measurements.
At this point, we know that in a smaller cohort of patients, we can achieve the results we desire. However, we felt that it was important to have a randomized trial demonstrating the clinical benefit, but something that would show the health economic benefit in a statistically significant manner. We believe you need to show both clinical and economic benefit to drive widespread adoption and ensure patient access for a new technology.
That study is now actively enrolling and has been an exciting endeavor for our team. We’re really fortunate to be working with many of the key opinion leaders in this space, and collaborating with them is a great privilege for everyone on our team.
During the pre-submission process, FDA noted that our trial design was more extensive and comprehensive than what the
agency typically sees. Our philosophy was to structure a single trial to satisfy all regulatory, coding, and coverage requirements we expect to encounter, to expedite widespread and permanent access to patients.
It struck me as a much more efficient path to getting to where we want to be, from both a time and a capital perspective. I think that’s better for patients. I think it’s better for all of our financial stakeholders. It’s better for our team who have been working so hard on this.”
Q. In parallel with these achievements, you also have another product in the works. Can you tell us about that?
“In our continued engagement with nephrologists and surgeons, we discovered another related problem that we decided to pursue. Once AVFs mature and are ready for use, they are notoriously challenging for patient care technicians [PCTs] to cannulate successfully. This is due to the inability for them to see the fistula when attempting to place large needles into the vessel. Needle misplacement happens frequently and leads to serious and expensive complications such as hematoma, infection, and aneurysm formation. The physicians asked if there was a way we could adapt our ultrasound technology to help minimize the risk of cannulation injury or infiltration, as it would have a meaningful impact on these patients.
So we started thinking about ways that we could address that issue. We were very fortunate that the National Institutes of Health [NIH] awarded us a $1.7 million grant about two years ago to start working on a 3D ultrasound solution that provides volumetric fistula imaging to aid technicians in AVF cannulation, that we call EchoGuide, as a compliment to EchoMark and EchoSure.
That product development process went very well, and our user testing was incredibly successful. That enabled us to apply for the next SBIR [Small Business Innovation Research] Phase IIB Grant from NIH, which was awarded in September for $2 million. That grant provides us with the funding necessary to pursue our
regulatory clearance and conduct a study with a large dialysis organization to better understand how our solution can impact care.
Our goal is to have a series of ultrasound-oriented solutions to help across the various stages of the dialysis patient pathway. We want to get fistulas ready, get catheters out sooner, and then keep fistulas humming. The idea is to keep these patients out of the hospital, avoid unnecessary interventions, and improve the quality of life for folks who have a challenging experience as a dialysis patient.”
Q. These wins all set Sonavex up for its $15M Series A-2 financing in October. How did you accomplish that?
“We kicked off this financing when the market was at rock bottom. There was concern with high interest rates, a potential recession, and we observed that many venture funds were struggling to raise new funds. The VCs that did have capital were prioritizing the needs of their portfolio companies, and were deprioritizing new opportunities.
One of the key catalysts for raising this financing was executing on key milestones with a capital-efficient team, in parallel with the due diligence. It was a way for us to be able to stand apart. The team’s ability to secure strong clinical data, a new CPT code, FDA IDE approval for the MAFASA pivotal trial, and grant funding to support FDA clearance of our pipeline product in a short period of time was instrumental in attracting the new capital.
We are proud that the round brought in several new institutional investors, strong participation from existing shareholders, and industry veterans in vascular and dialysis. New investors include Unorthodox Ventures, GenHenn Capital, JSTAR Capital, Oakwood Circle Ventures, Riptide Ventures, and Partners Investment.
I’m most proud of our team and how each individual contributed meaningfully across all key functions. We had so many disparate workstreams running in parallel and were admittedly a little stretched, but all that hard work paid off. We’re in a really nice position to be able to realize our mission.”
Q. Speaking of accomplishments, Sonavex’s solution was recently recognized by Time magazine as a Best Invention of 2024. What does this award mean to you and your team?
“It’s incredibly flattering and we were not expecting it at all. It was out of left field, but we’re grateful for the exposure it has provided. The primary reason we’re excited about it is that it’s another avenue to build awareness around kidney disease and solutions that can help patients.
The publicity has been a great avenue for people to take the extra few minutes to read more about the technology, navigate to the website, and even reach out. We’re grateful for that. It’s also a testament to the numerous serial inventions that were necessary to make this whole solution a reality.”
Q. What advice do you have for the medtech entrepreneurs out there?
“This industry requires a ton of perseverance. It takes a lot of consistent effort over a long period of time, and there will inevitably
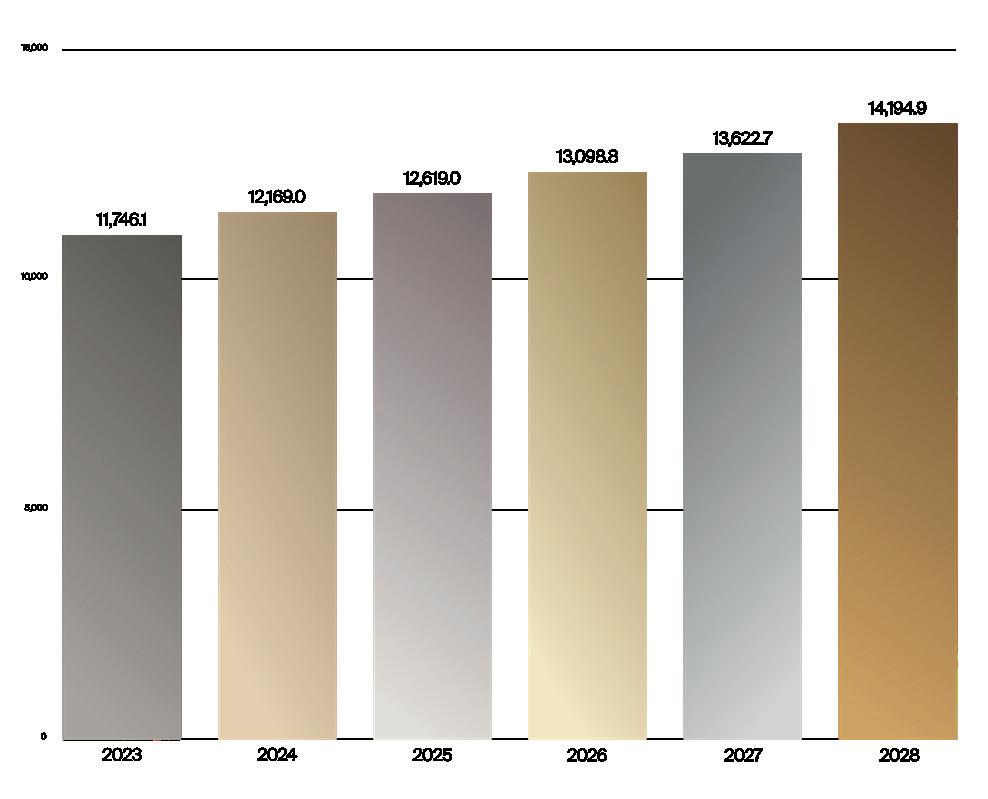
Global Hemodiaysis Catheters & Dialyzers, Market Forecast, 2023 - 2028
Hemodialysis is a treatment that involves the removal of toxic waste products and excess fluid from the body by use of a device that functions as an artificial kidney.
4.1 million patients worldwide require regular dialysis, of which 89% receive hemodialysis - and this figure is rising with the incidence of end-stage renal disease.
About 70% of the global sales for hemodialysis devices come from consumables likecatheters and dialyzers for filtering and cleansing blood.
These devices are projected to grow at a CAGR of 3.9% from 2023 to 2028, to over $14 billion in global sales by the end of the forecast period.
Surgically created arteriovenous fistulas, a critical vascular access for managing regular dialysis treatments in any treatment setting, fail to mature properly in up to 50% of p tients, delaying care and increasing the cost burden.
As the population of patients requiring dialysis is projected to double, the need for effective solutions to assess fistula maturation in a timely and efficient manner is urgent.
Innovations that improve patient outcomes from the initiation of treatment, like Sonavex’s suite of products, are essential in both lowering costs and enhancing the quality of care.
Source: LSI’s Market Analysis and Projections database; Hemodialysis Catheters and Dialyzers Market Snapshot, published August 2024
be setbacks. It’s about minimizing the impact of those setbacks and figuring out how to reorient and keep moving forward. It’s never a straight line, and you just have to have that mindset.
It’s also about the people you surround yourself with. Some don’t fully appreciate it, but each individual can have such a major impact. Everybody always gives so much credit to the CEO, whose job is to raise money, determine the strategy, and execute on the strategy, but the CEO can’t and doesn’t do it all themselves. It’s all about the people, whether it’s the full-time team, Board, advisory board members, consultants, partners, physician collaborators, all these folks. It’s that greater team that is responsible for all those milestones.
I’m incredibly fortunate. We get to work with some of the brightest, hardest-working people and we genuinely enjoy spending time together. We’re a small group of people, but everyone has complementary skill sets, trusts each other, and everyone’s aligned on our mission.
Lastly, in order to have that team, you need money. So you need to be clever and scrappy. Until our latest equity fundraise, we brought in more than one dollar of non-dilutive funding for every dollar of equity financing that we raised. Investors love that. Frankly, that non-dilutive capital was instrumental to get us through the early days. The medical device ecosystem is not throwing a lot of capital at early-stage, non-clinically validated technology right now.
Early on, most companies need to focus on sources of capital outside of traditional ventures. We were fortunate, we had an array of supporters including physicians, angel investors, super angels, family offices, and federal government agencies that understood the vision and believed that our team could execute on the opportunity ahead of us. These investors aren’t limited by the constraints of 10-year fund cycles, and therefore can be a little more patient than traditional VCs.
Another important element of what we did was formalize a Board of Directors and Advisory Board. We were fortunate to find people who were aligned with our vision, were well-respected, could open doors for us, and had a reputation for backing technologies for the right reasons. They provided a routine source of feedback and really extended our reach. The tricky part is finding the right people.
You also need to really understand the market access and reimbursement landscape, which is something that a lot of people ignore. It’s complex and rarely feels like the most urgent issue for many CEOs and founders. It was one of the things that we spent a lot of time on early, and the investor community recognized that we were prioritizing the right things.”
Q. Can you talk about the LSI events, and your experience as an alum. Have the events and connections you’ve made impacted your company’s trajectory?
“LSI has done an incredible job attracting a large, diverse group of decision makers across startups, strategics, investors, and the needed service providers that enable the industry to function. Scott and the team have intentionally created a forum that allows for people to be their organic selves and build strong, lasting relationships. The brilliance behind LSI events, in my mind, is the amazing venues that are isolated from the rest of the city or town. Nearly everyone stays on site for the entire conference, which is a very unique aspect of the LSI experience.
Several of the relationships that I’ve cultivated at LSI resulted in warm introductions to the people that ultimately participated in a Sonavex financing. I’ve later found that some of the individuals I became friendly with ended up serving as a reference for me as part of a diligence process.
So, hats off. I think LSI is the place to be if you’re a medtech entrepreneur trying to establish and maintain great relationships in the industry. No one has done it better.”
“The key things we’re focused on are executing our MAFASA randomized trial that’s currently enrolling, and in parallel getting our pipeline product, EchoGuide, FDA cleared and gathering the necessary clinical evidence. We still have a lot of work to do, but we’re proud of what we’ve been able to accomplish this past year.”

Note: Images in this article are used with permission from David Narrow. See David’s presentation from this past March, on LSI’s YouTube channel, LSI USA ‘24 Emerging Medtech Summit at the Waldorf Astoria Monarch Beach in Dana Point, CA. You can reach him at dnarrow@sonavex.com.
Have you presented at an LSI event and are interested in having your journey and technology featured in an upcoming issue of The Lens? Contact our Managing Editor and Content Strategist Tracy Schaaf at tracy@ls-intel.com.
De-risk your biggest strategic decisions with our most comprehensive market data and projections (worldwide sales, share by supplier, CAGRs) on 27 major market segments and 200+ sub-segments, prepared with consistent methodologies and forecast periods.


June 10th - 13th, 2025, The Ritz-Carlton, Millenia Singapore
With the global community built through LSI USA and LSI Europe and our partnerships in Singapore, China, Korea, Japan and more, LSI Asia will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Like LSI USA and LSI Europe, LSI Asia will not be held in a chaotic convention center. We have identified a world-class venue in Singapore, the gateway to Southeast Asia, for our inaugural event where senior executives can do real business.


Thank you to our founding sponsors
As we launch in this new geography, we are offering a unique opportunity to a small set of value-add, mission-aligned partners looking to elevate their global presence and develop new business. Get in touch with us to learn more and become a founding sponsor.
Celebrating the recent wins of our LSI presenting company and speaker alumni community.

Helsingborg, Sweden-based Acorai is developing a non-invasive intracardiac pressure monitoring device, the SAVE Sensor System, to improve heart failure management for more than 64 million
patients worldwide. The system received FDA Breakthrough Device Designation in 2023, enabling Acorai’s participation in the FDA’s Total Product Life Cycle Advisory Program (TAP).
The company recently announced the successful completion of patient enrollment for its CAPTURE-HF study. The study is a two-phase data acquisition study designed to evaluate the accuracy, safety, and usability of Acorai’s device in assessing cardiac and pulmonary pressures, with patients compared against a right heart catheterization gold standard.
Acorai enrolled a total of 1,600 patients across 20 clinical sites in six countries, with 1,300 participants in Phase 1 for further product development and over 300 participants in Phase 2 for validation, with over half of the patients enrolled from U.S.
sites. The study’s comprehensive design will support regulatory submissions across Europe and the U.S. and inform future developments of Acorai’s technology. CAPTURE-HF study results are expected in the first half of 2025, providing the foundation for Acorai’s regulatory submissions across the U.S. and Europe, and future innovations in non-invasive cardiac care.
The prevalence of heart failure is a significant concern worldwide, affecting more than 64 million people. In the United States alone, around 1 million of the 6 million individuals with heart failure are hospitalized each year due to their condition. Unfortunately, 30% of patients return to the emergency room within less than a month of being discharged, resulting in poor quality of life and a cost of over $100 billion to the U.S. healthcare system.
Intracardiac pressure monitoring (ICPM) is the single most effective way to manage heart failure patients, but within the hospitalized and clinic environment, only invasive methods exist to assess this vital parameter accurately. The Acorai device’s non-invasive handheld form factor could improve access to ICPM and potentially reduce time and cost. It has been shown that access to these vital signs can improve the quality of care for patients, yield better health outcomes, and result in significant cost savings. Acorai aims to be able to enable the deployment of ICPM across the broader healthcare system for use by healthcare professionals to aid the assessment and personalized treatment of patients with heart failure.
Acorai is backed by Solardis Health Ventures, KHP Ventures, GA Investors, and Life Science Invest, with a seasoned medical device team and corporate advisory board with experiences from GE Healthcare, Verily, and Abbott, and has established clinical partnerships with world-leading hospitals.

AliveCor is pioneering the creation of FDA-cleared machine learning techniques to enable proactive heart care. Its FDAcleared KardiaMobile is the most clinically validated mobile ECG solution on the market and is recommended by leading cardiologists and used by people worldwide for accurate ECG recordings. This simple-touse mobile device and app-based service provides instant analysis for detecting atrial fibrillation (AFib) and normal sinus rhythm in an ECG. KardiaPro is the first AI-enabled platform for doctors to monitor patients for the early detection of atrial fibrillation, the most common cardiac arrhythmia that leads to a five times greater risk of stroke.
The company has announced the activation of its services and solutions, designed specifically for U.S. veterans, on
the Veterans Affairs (VA) Federal Supply Schedule and Defense Logistics Agency (DLA) ECAT Contract System. Veterans are able to get prescriptions for AliveCor’s FDA-cleared devices including KardiaMobile 6L, the world’s first pocket-sized sixlead personal ECG device, and KardiaMobile Card, the world’s first credit card-sized single-lead personal ECG device, with two years of KardiaCare membership included as a potentially covered benefit.
AFib, the most common type of treated heart arrhythmia, is associated with a highly elevated risk of stroke. With more than 4.4 million veterans living in rural areas, often far from a VA cardiologist, and more than 1 million veterans treated for atrial fibrillation at VA centers in the past decade alone, there is a significant need to bring innovative tools to this population. AliveCor’s Kardia devices and KardiaCare services facilitate remote heart monitoring by detecting six common arrhythmias, including AFib, and provide board-certified cardiologist reviews of ECGs within a day, empowering veterans to manage their heart health remotely, while giving physicians timely, actionable cardiac data outside care settings.

ARC is a privately held device company focused on preventing surgical adhesions – a large unmet medical need. Internal adhesions are the most common complication of many types of surgeries and can lead to serious medical issues, including chronic pain, immobility, infertility, and small bowel obstruction – often necessitating an expensive procedure to release the adhesions.
ARC’s high molecular weight, polymer-based, liquid medical devices are designed to prevent or reduce adhesions following common surgeries. The company’s devices are easily and rapidly applied into the surgical site at the end of an arthroscopy, laparoscopy or open laparotomy sur-
gery; and then flow and form a temporary, physical, polymer barrier film that coats the tissues and mechanically prevents adhesions throughout the entire surgical area. ARC’s lead medical devices, JOCOAT and IPCOAT, are currently orthopedic, gynecologic, and abdominal surgeries, respectively.
The company announced the successful completion of a $3 million financing, with funds to be used to support two new clinical trials to further demonstrate the efficacy and safety of JOCOAT and IPCOAT:
- JOCOAT Orthopedic Clinical Trial: Led by Prof. Dr. Robert Litchfield, ARC’s Chief Medical Officer and an experienced orthopedic surgery principal investigator, the company’s JOCOAT device will be evaluated in a clinical trial with 20 patients undergoing knee surgery for anterior cruciate ligament (ACL) repair.
- IPCOAT Gynecological Clinical Trial: Led by Prof. Dr. Rudy De Wilde, a worldwide key opinion leader in gynecological surgical adhesion reduction, ARC’s IPCOAT device will be evaluated in a clinical trial with 20 patients undergoing gynecological surgery for the treatment of endometriosis. In addition to the clinical trials, the funding provides additional runway for ARC to continue current discussions towards a Japanese partnership agreement.

Arsenal Medical is a clinical-stage company that creates innovative biomaterials to solve challenging and underserved medical problems, with its lead products targeting neurovascular and trauma conditions. Its NeoCast embolic agent is a first-of-its-kind, shear-responsive, non-adhesive, solvent-free, liquid embolic material designed to be injected through a neurovascular catheter and deeply penetrate target vessels to achieve complete vascular occlusion.
The company has announced the initiation of the EMBO-02 clinical study of NeoCast to treat chronic subdural hematomas (cSDH), with the first patient enrolled in the trial. The study will evaluate the safety and feasibility of NeoCast in expanding the patient population utilizing middle meningeal artery embolization (MMAe). The study is enrolling up to 10 subjects at three sites in Australia; the first patient has been enrolled at Monash Health, Victoria, Australia.
cSDH is the persistent bleeding of an injured vessel in the membrane that protects the brain (dura). It is typically caused by a fall or injury to the head (including minor head trauma) and can result in significant physical and cognitive decline. It is the most common neurosurgical condition in those 65 and older, and has been categorized as a sentinel health event in the elderly. MMAe is an emerging minimally invasive procedure to treat and prevent the recurrence of cSDH.
Biometry Raises $5M in Seed Funding

Biometry Inc. (myBiometry), an asthma and COPD diagnostic and remote monitoring company, raised $5 million in an oversubscribed seed funding round. The round was led by Dexcom Ventures, with participation from CareSource Indiana, Elevate Ventures, and other investor groups.
This funding will advance the development of the company’s fenoTRACK solution, a breakthrough device designed to empower asthma and COPD patients by providing data to enable early detection of exacerbations. fenoTRACK is a portable, non-invasive breath test that measures type 2 airway inflammation via fractional exhaled nitric oxide (FeNO).
Asthma and COPD impact over 500 million patients globally, and 41 million in the U.S. Exacerbations occur in approximately 40-50% of patients annually, leading to millions of ER visits and hospitalizations each year in the U.S. alone. Frequent
attacks can lead to progressive loss of lung function, permanent airway remodeling, and in COPD, or death. Type 2 inflammation is the underlying driver in 80% of patients with asthma and 30-40% of patients with COPD, according to Biometry.

BMI OrganBank is developing novel organ preservation systems with potential to significantly reduce the waitlist for lifesaving organ transplants, beginning with kidneys.
The organization has been awarded a $3.5 million Phase II Small Business Innovation Research (SBIR) grant from the National Institutes of Health (NIH). The funding will support further development of the company’s groundbreaking kidney preservation technology with the goal of preparing it for clinical trials and FDA approval.
This grant follows promising preclinical results from BMI OrganBank’s partnership with Duke University’s Ex Vivo Organ Laboratory (DEVOL), which demonstrated the potential of the company’s warm perfusion system to enhance organ quality assessment and extend ex-vivo (outside the body) preservation times for donated kidneys.
Millions of registered organ donors die every year, but only a small fraction have their kidneys transplanted. As a result, almost 90,000 Americans are on the waiting list for lifesaving kidney transplants. This imbalance in supply and demand could be addressed by the greater use of kidneys from older donors and donors after circulatory death (DCD). With current organ preservation technologies, which use cold temperatures, it can be difficult to evaluate the quality of these kidneys before transplant. Furthermore, they are vulnerable to significant injury during the storage period before transplantation. As such, clinicians and patients are reluctant to use them.
BMI OrganBank’s warm perfusion plat-
form offers a solution by providing a more effective method of preserving kidneys outside the body. This technology allows for new methods of organ quality assessment and extended preservation times, potentially increasing the number of kidneys available for transplant and helping to address the critical shortage of organs.
BMI has research facilities in the Innovation Accelerator at the Wake Forest Institute for Regenerative Medicine in Winston-Salem, NC and in Research Triangle Park, NC. BMI OrganBank has multiple medical devices in development that use its patented and proprietary technologies.

BrightHeart, an innovator in AI obstetric and pediatric cardiology technology, is a Paris-based, privately held medical device company and part of Sofinnova MD Start, Sofinnova Partners’ medical device accelerator.
The company has received FDA 510(k) clearance for its first artificial intelligence software, designed to transform prenatal ultrasound evaluations of the fetal heart. This milestone marks a significant breakthrough in one of the most challenging and critical areas of prenatal care.
Fetal heart ultrasound exams are essential for detecting congenital heart defects (CHDs), which are the most common type of birth defect, affecting nearly 1 in 100 newborns. However, misdetection or delayed diagnosis can lead to severe consequences, including missed opportunities for life-saving interventions. BrightHeart’s AI software offers an innovative solution to these challenges, enabling clinicians to improve the detection of morphological abnormalities suggestive of CHDs, in resource-constrained practices.
BrightHeart’s technology is designed to integrate seamlessly into existing workflows, reducing the burden on over-
worked sonographers and OB/GYNs. By automating key aspects of the evaluation process, the software has the ability to save time and improve accuracy, in increasingly resource-constrained practices. BrightHeart builds on an idea by Dr. Marilyne Levy and Dr. Bertrand Stos, two leading fetal cardiologists based in Paris, France.

Clairity, a pioneer in AI-based predictive tools for breast cancer, has appointed Jeffrey R. Luber, JD, MBA, as President, Chief Executive Officer and Director. Luber, an award-winning healthcare leader with extensive experience across oncology, genetic sequencing, infectious disease testing, and digital health, will guide Clairity through market launch and global expansion.
Luber’s extensive career includes leading multiple healthcare companies through the complete arc of maturation from research entities, through pre-clinical work, FDA submission and clearance, market launch, commercial growth, and company exit. Regarded as a builder of enduring relationships and value-enhancing partnerships, Luber served most recently as CEO of binx health, where he led the company to first-in-market status in a $2B category for infectious disease point-ofcare testing and also founded the firm’s athome digital health arm, reaching millions in underserved areas and generating tens of millions in revenue. Luber continues to serve as binx Chairman today.
His previous roles also include CEO of Good Start Genetics (sold to Invitae in 2017) and President and CEO of Exact Sciences, where he helped establish DNA-based testing for colorectal cancer as a new standard for millions, winning endorsement from the American Cancer Society and World Gastroenterology Organization under his leadership.
Clairity’s mission is to improve breast cancer screening for everyone, regardless of race, ethnicity, or geography. Allix5, the company’s first product, uses AI-derived analysis of a screening mammogram designed to identify patients at increased risk of developing breast cancer. The platform draws on vast and diverse imaging data and clinical outcomes from around the world that the company believes will offer novel opportunities for risk prediction.


Echopoint Medical Ltd., a London-based medtech innovator dedicated to addressing gender bias in the diagnosis of ischemia with non-obstructive coronary arteries (INOCA), has appointed Nancy Briefs as Chair of the Board. Nancy brings extensive experience in medtech innovation, strategic growth, and leadership, aligning with Echopoint’s mission of advancing cardiovascular diagnostic solutions.
Nancy is an Independent Board Member, serial CEO, advisor, and venture partner at StageNext. Nancy is a strategic business leader with extensive experience creating value, driving strategy, and launching products and drugs in digital health, medtech, and pharma. With her deep general management and fundraising expertise, she has a remarkable ability to turn innovation into commercial reality. Having raised more than $500M in equity, including an IPO, her companies are a living testament to her entrepreneurial spirit. She currently serves on the boards of AltrixBio and BiomeBank, and she previously served on six corporate boards with multiple successful exits.
Her previous affiliations are EY Entrepreneur of the Year, Northeast, EY National Judge for Life Sciences Entrepreneur of the Year, Mass Ventures, Start Program,
Former Chairman of the Medical Device Manufacturing Association (MDMA), Mass Life Science Center (MLSC) Milestone Achievement Program, Coach for Mass Next Gen Initiative, and MedExecWomen.
Echopoint Medical is actively preparing for the U.S. launch of iKOr, its innovative diagnostic tool for INOCA. Echopoint’s iKOr platform uses pulses of light in fine optical fibres inserted into the blood vessels to measure coronary flow-rates in patients who suffer from chest pain, with ischemia and no obstructions. It enables therapy decisions to be guided by one device in one ‘intuitive’ step, facilitating easy to use early intervention, and more effective management of heart diseases associated with angina (heart pain).
With FDA market clearance in place, the company is poised to undertake clinical trials that will further validate iKOr’s effectiveness in real-world settings. This important step reflects Echopoint’s commitment to creating accessible, patient-centered solutions that empower clinicians and improve patient outcomes globally.
INOCA affects two-thirds of cardiac patients and four times as many women as men, and in most cases is caused by microvasculature dysfunction and/or vasospasm of the coronary arteries. Because the diagnosis requires specialized expertise and testing, it is frequently missed, but leaving it undiagnosed can be dangerous for patients as INOCA is associated with repeated emergency hospital admissions and increased incidence of cardiovascular events.

EndoQuest Robotics is a leading innovator in the development of transformative endoluminal robotic technologies. The company is focused on addressing unmet needs in gastroin-
testinal (GI) medicine and endoluminal surgery by enabling physicians with unprecedented surgical access, precision, and flexibility. Its proprietary technology is designed to navigate and perform scar-free procedures through the body’s natural orifices, allowing for the least invasive surgical approach.
EndoQuest Robotics has recently submitted an Investigational Device Exemption (IDE) application for its pivotal colorectal clinical study. This IDE will enable EndoQuest to begin their Prospective Assessment of a Robotic-Assisted Device in Gastrointestinal Medicine (PARADIGM) study, which is a prospective, multi-center, open-label study. With an expected enrollment of 50 subjects, the PARADIGM study will be conducted at five leading U.S. healthcare facilities, including Brigham and Women’s Hospital (Boston), Mayo Clinic (Scottsdale), Cleveland Clinic (Cleveland), AdventHealth (Orlando), and HCA Healthcare (Houston).
As part of the PARADIGM study, globally recognized gastroenterologists and colorectal surgeons will perform robotic endoscopic submucosal dissection (ESD) procedures for removal of colorectal lesions using the EndoQuest Robotics Endoluminal Surgical (ELS) System. Results from a preclinical ESD study presented earlier this year at Digestive Disease Week (DDW2024) demonstrated that the ELS System had superior procedural outcomes, a shorter learning curve, and reduced procedure time versus the conventional technique.
The IDE submission is subject to review by FDA and must be approved prior to initiating this study. The review process is expected to take a minimum of 30 days, with the total duration of the review to depend on a variety of factors, including the extent of potential comments, questions, and any additional information requested by the FDA.
“There is a significant unmet need for improved treatment of colorectal disease in the U.S.,” says Dr. Todd Wilson, Chief Medical Officer and Chairman of the Scientific Advisory Board of EndoQuest Robotics. “The robotic ESD procedure provides the opportunity to clearly demonstrate the potential benefits of our flexible robotic system in terms of outcomes, complications, healthcare costs, and much more, and should ultimately pave the way for the system to revolutionize a wide and growing range of other procedures across the globe.”


Enspectra Health is an emerging, privately held healthtech company pioneering the virtual biopsy to transform the way physicians evaluate a broad spectrum of skin conditions. The company’s novel cross-modal technology combines reflectance confocal and multiphoton laser scanning microscopy to instantaneously digitize cellular images of skin noninvasively.
The company recently announced the publication of its innovative cross-modal imaging technology in Scientific Reports, a journal of the Nature Research family. The publication, titled “Handheld multiphoton and pinhole-free reflectance confocal microscopy enables noninvasive real-time, cross-sectional imaging of skin,” was published online. Easily portable and handheld, cross-model imaging digitizes histopathology directly from a patient’s skin to provide actionable health insights for dermatologists.
The scientific publication outlines how cross-modal technology overcomes the limitations of prior imaging modalities such as monochromatic images and rigid positioning of patients. The revolutionary cross-modal imaging technology delivers high-resolution, multi-color (Tetrachrome) results in an intuitive, user-friendly system making it a practical solution for the in-office dermatology practice setting. The performance of the technology was evaluated with healthy human participants ranging in age from 9–81 years and in skin type diversity from Fitzpatrick I-V. Ethnic representation was a strength of the study; 43% of participants identified as Hispanic/ Latino, Asian, Black/African American, or as multiracial. Additionally, feasibility performance was evaluated in lesions suspected of skin cancer with observational results consistent with traditional H&E histopathology.
EpiBone, Inc. is a privately-held regenerative medicine company focused on skeletal reconstruction. Sitting at the intersection of biology and engineering, the company harnesses the power of cells to create living solutions that have the potential to integrate seamlessly into patients’ bodies. EpiBone is currently developing a pipeline of bone, cartilage, and other skeletal tissue products.
The company recently announced the successful close of a convertible note funding round led by Kendall Capital Partners, with participation from LifeSpan Vision Ventures, EMV Capital plc, and others. This additional funding will propel EpiBone’s ongoing pipeline development, furthering its mission to revolutionize skeletal reconstruction on a global scale.
EpiBone’s pipeline includes:
BONE: EB-CMF tissue-engineered autologous bone graft: A facial bone product indicated for craniofacial defects resulting from trauma, congenital defect or degenerative bone disease. It is customized for patients via a CT scan so that precise anatomical shape can be cultivated, and with stem cells extracted from patients’ abdominal fat. The company completed a Ph I/IIa clinical trial for this product in 2023.
JOINT: EB-OC tissue-engineered osteochondral autograft: EB-OC is an allogenic, tissue-engineered osteochondral plug for treating high-grade cartilage lesions in healthy joints. It’s composed of condensed mesenchymal cells from donor bone marrow, encapsulated in hydrogel, and cultivated on a bone scaffold in a bioreactor. Cyclical mechanical loading and perfusion optimize tissue development. The bone layer anchors the plug, eliminating the need for hardware. The company has successfully tested it in horses, and the FDA approved an IND for a Phase I/IIb trial with 36 patients.
CARTILAGE: EB-IAC injectable cartilage filler: EB-iAC is an allogenic, injectable cartilage product for treating low-grade cartilage lesions that don’t involve underlying bone. Stem cells from a cell bank are processed into condensed mesenchymal cells primed for cartilage formation. The cells are combined with a hydrogel carrier and delivered to the patient. The capability to store product components for up to two years ensures flexibility and convenience in scheduling treatment procedures.

FluidAI, formerly known as NERv Technology, is a Canadian medical technology company that uses AI to aid with the early detection of postoperative complications. From general to gastrointestinal surgeries, FluidAI’s mission is to empower healthcare providers with data-driven solutions that improve patient outcomes and advance postoperative care.
The company recently announced a collaboration to access de-identified clinical data through Mayo Clinic Platform_Discover. This collaboration will enable FluidAI to accelerate the development of predictive models aimed at enhancing clinical decision-making and improving patient outcomes.
FluidAI’s Stream Platform is a software suite that leverages AI to analyze data from disparate sources to build a comprehensive understanding of the patient’s condition and alert surgeons to potential complications and risks before and after surgery. Stream Platform combines data from novel sensors, including FluidAI’s Origin sensor platform, with comprehensive and contextualized medical record data to generate insights in real-time that may help physicians predict clinical outcomes.
Mayo Clinic Platform_Discover offers AI solution developers access to diverse, de-identified clinical data, including demo-
graphics, diagnoses, imaging, lab results, clinical notes, and pathology reports. This diverse data set enables FluidAI to design predictive models on real-world retrospective data to facilitate faster model development.

GT Medical Technologies, Inc., a medical device company focused on improving the lives of patients with brain tumors, today announced it had reached two clinical milestones that will help expand the use of GammaTile, the company’s FDA-cleared innovation for the safe delivery of brain brachytherapy.
GammaTile was implanted in the 1,500th patient at Brooke Army Medical Center in Fort Sam Houston, Texas. Additionally, the Registry of Patients With Brain Tumors Treated With STaRT (GammaTiles; NCT04427384), a large observational study to further demonstrate the safety and efficacy of GammaTile, enrolled the 400th patient. The registry will collect follow-up information on local control, overall survival, quality of life, functional status, and surgical and radiation-associated adverse events over five years. The data will help demonstrate why GammaTile should be considered as a preferred option for patients with operable brain tumors.
The device is a bioabsorbable collagen implant embedded with Cesium-131 seeds that delivers radiation immediately after the tumor is removed. It is implanted into the tumor cavity and targets residual cancer cells while limiting the dose to healthy brain tissue.
GammaTile is FDA-cleared as a treatment for patients with newly diagnosed malignant intracranial neoplasms and patients with recurrent intracranial neoplasms. Since its full market release in the United States in March 2020, GammaTile has been offered in over 100 leading in-
stitutions, with more centers being added each month.

Hyperfine, Inc. (Nasdaq: HYPR) is a health technology company that has redefined brain imaging with the Swoop system—the first FDA-cleared, portable, ultra-low-field, magnetic resonance brain imaging system capable of providing imaging at multiple points of professional care. The mission of Hyperfine is to revolutionize patient care globally through transformational, accessible, clinically relevant diagnostic imaging.
The Swoop Portable MR Imaging system is FDA cleared for brain imaging of patients of all ages. It is a portable, ultralow-field magnetic resonance imaging device for producing images that display the internal structure of the head where full diagnostic examination is not clinically practical. When interpreted by a trained physician, these images provide information that can be useful in determining a diagnosis.
The compact and versatile Swoop Portable MR Imaging system is designed for use in any professional healthcare setting, including medical offices. Its ultra-low field strength of 0.064T eliminates the need for costly shielded MRI rooms. Its plug-andscan functionality and user-friendly tablet interface enable medical staff to easily operate the system.
The company recently announced CE approval of its latest generation of AI-powered Swoop system software, under the European Medical Device Regulation (MDR, EU No. 2017/745). Hyperfine also recently announced the issuance of new MRI standards by the Intersocietal Accreditation Commission (IAC), a leading CMS-approved accrediting body. The new standards, effective immediately, now include portable MR scanning at the point of care.
Earlier in 2024, Hyperfine significantly expanded its global distribution network with partnerships established in 13 European countries, including the five major European markets. This strategic expansion aims to enhance access to advanced portable brain MR imaging technology across diverse healthcare settings worldwide.
Founded by Dr. Jonathan Rothberg in a technology-based incubator called 4Catalyzer, Hyperfine scientists, engineers, and physicists developed the Swoop system out of a passion for redefining brain imaging methodology and how clinicians can apply accessible diagnostic imaging to patient care.

Lumicell is a privately held company focused on enabling a more complete resection of cancer by advancing the development and commercialization of its innovative fluorescence-guided surgery technology.
The company’s lead products are LUMISIGHT (pegulicianine) and Lumicell DVS, which are approved for use in combination to illuminate cancerous tissue within the breast cavity during the initial lumpectomy procedure, as an adjunct to the standard of care. Lumicell’s proprietary, pan-oncologic optical imaging agent, LUMISIGHT, is also being explored for further development across a wide variety of solid tumor indications.
Lumicell recently announced that LUMISIGHT was granted a permanent Healthcare Common Procedure Coding System (HCPCS) code for Hospital Outpatient Prospective Payment System (OPPS) by the Centers for Medicare & Medicaid Services (CMS).
Effective on January 1, 2025, this new code, A9615 Injection, pegulicianine, 1 mg, will enable streamlined billing and reimbursement for healthcare providers and
patients. Transitional pass-through (TPT) payments will continue and will be associated with this permanent HCPCS code, A9615, instead of the temporary C-code, C9171, that was granted in September 2024.
HCPCS codes are established by CMS to facilitate accurate Medicare claims processing. The assignment of HCPCS code A9615 marks a major milestone for Lumicell as it seeks to improve patient access to its new and innovative technology while supporting healthcare providers by simplifying the claims submission process.
The company also recently announced the appointment of Howard Hechler as Chief Executive Officer. Hechler joined Lumicell as SVP of Corporate Development & Strategy and was then promoted to President and now CEO. His prior experience spans investment banking, management consulting and extensive partnering/M&A work at large medical device and biopharmaceutical companies, including Medtronic, Smith & Nephew, and Mallinckrodt Pharmaceuticals. He graduated cum laude with a bachelor’s degree from Harvard University and received his MBA and juris doctorate from the University of Virginia.
granted a key patent covering Navigation Sciences’ NaviSci System for the minimally invasive, precision localization, and resection of lung and other cancers with real-time surgical margin control. The patent, “System for a Tissue Resection Margin Measurement Device,” the company’s first issued under the International Patent Cooperation Treaty, is being validated in the U.K., France, Germany, and Switzerland (including Lichtenstein). The company has four issued U.S. patents and additional international and U.S. patents pending.
The NaviSci System has undergone hardware development, preclinical testing, and a successful 25-patient clinical trial at Brigham & Women’s Hospital. The trial results confirmed that the System is useful in providing real-time measurement for surgical resection of lung nodules, that it is safe and effective, and that its distance measurements correlate with the pathology lab. The company is working to complete development in preparation for an FDA submission in 2025.
To support the NaviSci System’s development, the company has raised $5.5 million. In addition, the company was awarded a $400,000 National Cancer Institute SBIR grant to support the development of the NaviSci EndoMarker

Navigation Sciences is a clinical-stage company developing the NaviSci System for the tissue conserving removal of lung cancer and other tumors. The NaviSci System integrates augmented reality and advanced software with surgical instruments to guide precise surgical resection by enabling real-time in vivo margin measurement for the first time. It aims to improve surgical outcomes with cutting-edge technologies that reduce recurrence risk, conserve lung function, shorten hospital length of stay, and enhance surgical workflow.
The European Patent Office recently


OncoHost, a technology company headquartered in Binyamina, Israel, and Cary, North Carolina, is transforming the approach to precision medicine for improved patient outcomes. OncoHost’s proprietary platform, PROphet, is a plasma-based, proteomic pattern analysis tool whose initial offering in non-small cell lung cancer (NSCLC) uses a single blood sample to guide first-line immunotherapy decision-making.
The PROphet NSCLC test provides clear clinical utility by offering physicians
crucial guidance on the optimal first-line immunotherapy treatment plan for each individual patient, with a significant effect on overall survival.
The company recently announced that Ron Andrews has joined the company as Strategic Consultant to the CEO. In this role, Andrews will support OncoHost’s growth strategy, while also providing strategic leadership in fueling the company’s development and expansion initiatives.
With over 35 years in the diagnostics and molecular diagnostics industry, Andrews has successfully led organizations ranging from Fortune 500 divisions to innovative startups. His executive experience spans global industry leaders including Abbott Diagnostics, Roche Molecular Diagnostics, and LifeTechnologies/Thermo Fisher, as well as public CEO roles at Clarient Inc and Oncocyte Inc. He currently serves as a trusted advisor to prominent venture capital firms, as well as guiding emerging molecular technology companies through critical product development and fundraising stages. His financial acumen has driven over $600M in successful capital raises and more than $15B in strategic exits over the course of his career.
Led by an experienced team of entrepreneurs and industry experts and supported by a large-scale prospective clinical trial with over 40 sites and 1,700 patients recruited worldwide, OncoHost is well-positioned to lead precision diagnostics and biomarker development to the next stage.

Sense Neuro Diagnostics is a medical technology company involved in improving outcomes for stroke and brain injuries.
Founded in 2017 by UC physicians including Opeolu Adeoye, MD, George Shaw, MD, PhD, and Matthew Flaherty, MD, as one of the first initiatives funded by the University of Cincinnati’s Technology Accelerator
for Commercialization, Sense Neuro Diagnostics is pioneering a brain injury detection device.
The company has developed a noninvasive brain scanner, the NeuroHawk device, that utilizes advanced, low-power radiofrequency (RF) technology to detect and monitor traumatic brain injury (TBI) and brain hemorrhage in seconds.
According to Sense Neuro Diagnostics, the quick, impartial diagnosis capability could improve triage and treatment decisions on the battlefield, thereby reducing the risk of secondary injury and improving outcomes for those with the most acute needs.
The company was recently awarded a $2 million contract by the Department of Defense (DoD), Congressionally Directed Medical Research Programs (CDMRP) via the Traumatic Brain Injury and Psychological Health Research Program (TBI PHRP). The aim of the funding is to support clinical trials for Sense’s NeuroHawk device.
Set to begin in June 2025, the clinical trial is designed to evaluate the capabilities of the NeuroHawk device to diagnose TBI and localize brain hemorrhage, offering critical insights in high-pressure surroundings.
With a period of performance of 30 months, the contract marks the second direct military funding the company has received. The first came through the Medical Technology Enterprise Consortium to build the device’s technology, including deriving initial algorithms for hemorrhage detection.
TBI has become a hallmark injury in modern warfare, affecting over 420,000 service members in the past two decades. As warfare continues to evolve, with emerging challenges like drone and trench combat in conflicts such as the ongoing war in Ukraine, the need for effective, point-of-injury diagnosis tools is more urgent than ever. Traditional methods of TBI diagnosis are limited and often rely on subjective measures, which may delay critical care.
The NeuroHawk device is also being tested for use in hospital and EMS markets, where it could be used to provide rapid diagnosis and continuous monitoring for stroke and brain injury patients. Sense Neuro Diagnostics plans to file for FDA clearance in the first quarter of 2025, then start a full commercial launch in mid-2025.
Sense Neuro has grown substantially since it launched, receiving over $18 million from various sources, such as the U.S. Army Medical Research and Development Command, the Cleveland Clinic, St. Elizabeth Healthcare, and Queen City Angels.

THINK Surgical is a privately held U.S.based technology innovator that develops and markets orthopedic robots. The company believes that by placing implant choice at the heart of the robotic procedure, together with surgeons and implant partners, its robotics can create a transformative momentum in orthopedics.
THINK Surgical’s TMINI Miniature Robotic System recently received 510(k) clearance from the FDA for use with the LinkSymphoKnee from Waldemar Link GmbH & Co. KG, Germany, under a collaboration agreement between the two companies. The LinkSymphoKnee has also been added to THINK Surgical’s ID-HUB, a proprietary data bank of implant modules for use with the open platform version of its TMINI System in the United States.
In addition, the TMINI Miniature Robotic System received FDA 510(k) clearance for use with the Freedom Total Knee, Freedom Titan Knee, and Freedom Primary PCK Systems from Maxx Orthopedics (Norristown, PA), under a collaboration agreement between the two companies.
The Freedom Total Knee and Freedom Titan Knee Systems feature a multi-radius design that accommodates individual patient anatomy, potentially leading to improved alignment and positioning for longterm success. Clinically, it has demonstrated strong performance and durability with a 10-year survivorship of 98.3%, according to the company.
THINK Surgical also announced that its TMINI Miniature Robotic System has received FDA 510(k) clearance for use with the MOBIO Total Knee System from b-ONE Ortho Corporation (Cedar Knolls, NJ), under a collaboration agreement between the two companies.
For more information on THINK Surgical and the rapidly evolving orthopedic robotics space, see the Orthopedic Surgery Market Dive in this edition of The Lens.

Cardiac arrhythmias cause 10% of global deaths, and over 25% of adults over 40 will develop a serious arrhythmia. Untreated arrhythmias not only raise the risk of death but are also associated with serious co-morbidities such as stroke and dementia.
Headquartered in San Diego, CA, Vektor Medical is on a mission to revolutionize arrhythmia care for millions affected by atrial fibrillation and other arrhythmias. Through its AI-based, non-invasive arrhythmia analysis technology, vMap, the company aims to improve cardiac ablation outcomes, optimize workflows, and increase procedural efficiency. vMap is the only FDA-cleared, non-invasive AI-based solution for arrhythmia localization and analysis, seamlessly leveraging only a 12-lead ECG. It is FDA cleared for use on the following arrhythmias: atrial and ventricular tachycardia, fibrillation, pacing, premature complexes (PACs and PVCs); and orthodromic atrioventricular reentrant tachycardia.
In November, the company announced the presentation of new data showcasing the significant capabilities of vMap, presented at the American Heart Association (AHA) Scientific Sessions 2024. This data highlights vMap’s impressive 91.1% accuracy rate in differentiating epicardial from endocardial ventricular tachycardia. Researchers analyzed 136 arrhythmia or pacing episodes from 72 patients, split evenly between epicardial and endocardial sources. The vMap algorithm demonstrated an impressive sensitivity of 97.1%, significantly outmatching the top-performing visual ECG methods (62.5% accuracy).
In September, the company presented findings of two studies at the Heart Rhythm Society HRX 2024 meeting, showcasing how Vektor Medical’s advanced technologies uniquely leverage AI and deep learning to assess wall thickness and scar tissue in ventricular tachycardia patients, providing crucial insights for electrophysiologists performing ablation procedures.

Vitara Biomedical, a privately held company based in Philadelphia, PA that is developing breakthrough technology intended to transform neonatal care, announced the closing of a $50 million funding round. Sands Capital led the Series B financing, with participation from Google Ventures (GV), First Spark Ventures, Flerie, and Khosla Ventures.
The company also announced the appointment of Kim Rodriguez, an experienced global healthcare and medical device industry professional, as Chief Executive Officer. She has 25 years of experience, most recently as CEO of BrainCheck, a digital health company supporting early identification of dementia and Alzheimer’s disease.
The company is developing a special life support system designed to improve outcomes for babies born prematurely. The Vitara System creates a fluid environment that mimics what a baby experiences in the mother’s womb and is designed to help support healthy growth and development of organs for babies born prematurely. Studies to date with fetal lambs have demonstrated that both mid-and late term gestation can be supported in the system for 10 to 28 days without harm, with continued development of organ systems.
The company is pursuing a first-in-human clinical study of the Vitara System. The company’s technology was initially developed at Children’s Hospital of Philadelphia (CHOP), one of the leading centers in the world for clinical and scientific innovation in neonatology.
Approximately 15 million babies are born preterm annually worldwide, indicating a global preterm birth rate of about 11%. This results in an estimated cost of $26 billion in economic burden in the U.S. alone, according to Vitara Biomedical. Extreme prematurity remains a major cause of neonatal mortality and severe long-term morbidity.

Xeltis, founded in Switzerland in 2006 as a University of Zurich spin-off, seeks to address the limitations of currently available options for the millions of people requiring hemodialysis access grafts or cardiovascular replacements every year. The company’s proprietary endogenous tissue restoration (ETR) platform, driven by Nobel prize-winning breakthroughs in polymer technology, utilizes an advanced polymer implant which regenerates the patient’s own tissue before gradually being absorbed and leaving new, living, and long-lasting vessels in place.
Xeltis’ most advanced product currently under clinical development is aXess, an implantable blood vessel for hemodialysis vascular access, that combines the safety and patency of an arteriovenous fistula (AVF) with the speed to treatment of an arteriovenous graft (AVG). The aXess vascular access conduit offers an improved dialysis patient experience and avoids the frequent reinterventions and complications, such as infections, faced by renal disease patients.
In November, the company announced that FDA granted aXess Breakthrough Device Designation status. Concurrently, following IDE approval from the FDA, the first patient in aXess’ U.S. pivotal trial has now been enrolled and treated at the Flow Vascular Institute in Houston, Texas, by Dr. Karl Illig, marking a significant clinical milestone for Xeltis.
A first-in-human trial of aXess demonstrated a significant improvement in performance compared to hemodialysis vascular access solutions. A pivotal trial of aXess is currently underway in Europe.
Xeltis’ investors include DaVita Venture Group, EQT Life Sciences, Kurma Partners, VI Partners, Ysios Capital, Grand Pharma Group, the European Innovation Council, and Invest-NL, in addition to other public and private investors.
Lung
Breakthrough technology regulator
Insulin pump maker
Sofinnova Partners portfolio company
Denervation for hypertension
Abbott, Medtronic and Dexcom tech
New Acting Director of CDRH
Leading medtech trade association
Omnipod maker
Protection of health data from breaches
Leader in the healthcare AI space
Real-time surgical visual tech: ________ Reality
Surgical
See page 54 for answers. This
Health tech devices worn on the body
Opened up valve replacement to more patients
Vaccine delivery method
Cortex acquirer ________ Scientific
AI plaque analysis company
Computer-assisted vacuum thrombectomy company
ExploraMed portfolio company
Tiny tech improving drug delivery
Tech for calcified aortic stenosis
J&J MedTech/Shockwave Medical device
Key asset: Intellectual __________
Technology advancing diagnostics and imaging
Brain disorder that has a new biomarker
Pulsed field ablation indication
Ogilopoly market with high barriers to entry
For relatable jokes on our ever-evolving industry and the times in which we’re living, we’ve got the memes you need to brighten your day!









Stuck on a tricky clue? Don’t worry—we’ve got you



