What the TechMed Revolution Means for Healthcare
Alan Cohen of DCVC forecasts agile, data-driven innovation
Venture Report, Power Moves, Alumni Milestones, and more...

Alan Cohen of DCVC forecasts agile, data-driven innovation
Venture Report, Power Moves, Alumni Milestones, and more...
January 2025


Scott Pantel
Chief Executive Officer, LSI Editor-in-Chief

Rebekah Murietta Vice President of Media, LSI Contributing Author

Kelly Williams Subscriptions & Enterprise Sales, LSI

Henry Peck
Chief Business Officer, LSI Editor-in-Chief

Brenna Hopkins Sr. Content Manager, LSI Contributing Author

Benny Tomlin Contributing Photographer

Tracy Schaaf Managing Editor and Content Strategist, LSI Lead Author

Blake Matrone Sr. Marketing Manager, LSI Contributing Author

Nicholas Talamantes Sr. Director of Market Intelligence, LSI Contributing Author

Maricela Almonte Customer Service & Fulfillment, LSI
(LSI)
714 847 3540 tel/fax email: info@ls-intel.com Vol 2. No. 1 January 2025
Created by LSI, The Lens leverages LSI’s deep industry relationships and proprietary market intelligence to help executives like you build lasting medtech companies. Start your Individual, Group or Enterprise-Wide subscription today!

Scan QR Code to activate your subscription.
The Lens is published monthly and available in print and electronic formats. Copyright by Life Science Intelligence, Inc. All rights reserved. Editor takes care to report information from reliable sources and does not assume liability for information published.
Interested in subscribing? email to: info@ls-intel.com
Interested in advertising? email to: kelly@ls-intel.com


Dear Readers,
As we step into 2025, we are thrilled to share the latest edition of The Lens. The new year has been off to a fast start for many, and the medtech industry continues to lead the way as a hub of innovation, resilience, and collaboration. It is an extraordinary time to be part of this ecosystem (perhaps the most exciting time in the industry’s history) where today’s transformative breakthroughs will shape the trajectory and future of healthcare for generations to come.
Medtech mergers and acquisitions (M&A) have set a positive tone for the year, with Stryker’s $4.9 billion acquisition of Inari Medical reinforcing their strategic expansion into neurovascular care. Similarly, the broader healthcare sector is making waves with Johnson & Johnson’s $14.6 billion acquisition of Intra-Cellular Therapies, reflecting a robust appetite for strategic expansion into new indications beyond value-accretive M&A supporting core product portfolios. While these deals promise immediate gains, we are also closely observing the long-term implications of a more consolidated set of strategic acquirers. What does this mean for innovation and competition? It’s a question that will shape the trajectory of our industry in the years to come.
Adding to the dynamism, a new U.S. administration brings both opportunity and uncertainty to stakeholders across medtech, from clinicians to startups to multinational corporations. As the administration sets its priorities, our industry is uniquely positioned to adapt, respond, and innovate in the face of evolving policy landscapes.
This edition of The Lens captures the unprecedented moment medtech finds itself in—one defined by both challenges and boundless possibilities. Few advancements are as electrifying as the integration of artificial intelligence into healthcare. We are thrilled to feature insights from Alan Cohen, General Partner at DCVC, whose TechMed thesis explores the revolutionary impact of AI and healthcare data on the industry’s future.
We also take a moment to reflect on 2024, with original analysis from Jon Norris and the team at HSBC, presented in their HSBC Innovation Banking Venture Healthcare Report. Their findings highlight sector-specific growth in areas like neurology and neuromodulation, increased funding for urological and gynecological innovations, and the leadership role cardiovascular M&A continues to play in delivering life-changing technologies to patients.
As we embark on this journey into 2025, we remain steadfast in our commitment to equipping you with the insights, connections, and tools needed to thrive in this vibrant and ever-evolving industry. On behalf of our entire team, we are honored to be part of your journey and your success.
Here’s to a year filled with progress, purpose, and shared triumphs.
All the best,
Scott Pantel and Henry Peck

Scott Pantel Chief Executive Officer, LSI Editor-in-Chief


Henry Peck Chief Business Officer, LSI Editor-in-Chief

De-risk your biggest strategic decisions with our most comprehensive market data and projections (worldwide sales, share by supplier, CAGRs) on 27 major market segments and 200+ sub-segments, prepared with consistent methodologies and forecast periods.

HSBC’s Annual Venture Healthcare Report reveals a resilient VC-backed healthcare space in 2024, dominated by mega early rounds or derisked, later-stage deals. Med Device stood out with the top 10% of deals capturing 60% of all investment dollars. In 2025— which is starting off strong—a diversified investor pool will balance bold new bets with strategic portfolio support, saving dry powder for the kind of company everyone wants to have: high potential.

For HSBC Innovation Banking’s Managing Director, Jonathan Norris, the theme of his 2024 Venture Healthcare Annual Report, “Look What You Made Me Do,” is not only in homage to Taylor Swift’s hit song but it references the altered state of healthcare investing in 2023 to 2024, after the turbulent, record investment and aggressive valuations experienced from 2020 through the first half of 2022. That phase of furious market activity began counter-balancing in 2023, as public market struggles and the lack of significant private M&A activity led venture firms to prioritize portfolio triage, deciding which of their companies they need to support because they do not have enough dry-powder capital to support them all, Norris describes. Inside rounds prevailed in 2023. Then, in 2024, driven by large therapeutic deals, novel technologies like AI-powered diagnostics, minimally invasive therapies, and precision health tools, new deal activity resumed, with a 30% rebound in dollars being invested—albeit with a clear shift and investor focus.
“At some point investors have to be strategic with the dry powder they have left in their funds, which companies are they going to support or not? And that’s what I mean by, ‘look what you made me
do,’” Norris said in a recent conversation with The Lens. “There hasn’t been a ton of distributions for venture firms back to their limited partners (LPs), and so they’re very hesitant about going out and fundraising until they have those distributions. And how do they get them? By supporting their best companies, at the expense of companies they have questions about or are not doing as well,” he continues.
As detailed in the HSBC report, across healthcare as a whole—including the sectors of Biopharma, Med Device, Healthtech and Dx/Tools—the challenges of securing follow-on financing, particularly Series B rounds, dampened some early-stage activity in 2024, resulting in fewer Seed and Series A deals. Investors have instead leaned toward a “strength in numbers” play for Series A: larger syndications raising large early-stage rounds, including $100 million or larger mega rounds—which is much different from what we’ve seen in the past—or else investors have shifted their focus to later-stage, derisked opportunities. (Also see the LSI Market Intelligence Deals table on page 24 of this issue, and “Long Game Legends: Makower and Papiernik’s Playbook for Success,” The Lens, Dec. 2024.)
The pressure is on in 2025 for both companies and investors, says Norris. Many companies that raised add-on or inside rounds in 2023 are now “on the clock,” he says, as they are facing pressure to secure new investor-led financing or risk consolidation or shutdown. And, the increase in investment in 2024 comes as investors face pressure from LPs to demonstrate returns before raising their next fund. Distributions from exits have been challenging in recent years, and this casts uncertainty on the timing of future fundraising, according to the report.
All of this is not to say that healthcare investment is in a negative place—it definitely is not. A total of $57 billion was invested in venture-backed healthcare companies in 2024, up from $44 billion in 2023, though with only 77 more deals in 2024 (2,158 vs. 2,081 in 2023), according to the HSBC report. In fact, the top 10% of these healthcare deals (216 deals) accounted for $30.2 billion in investment, representing 53% of all venture healthcare dollars.
By sector, 13% of the venture capital healthcare dollars last year were in Med Device, compared to 49% Biopharma, 23% Healthtech, and 12% Dx/ Tools. This compares to the number of
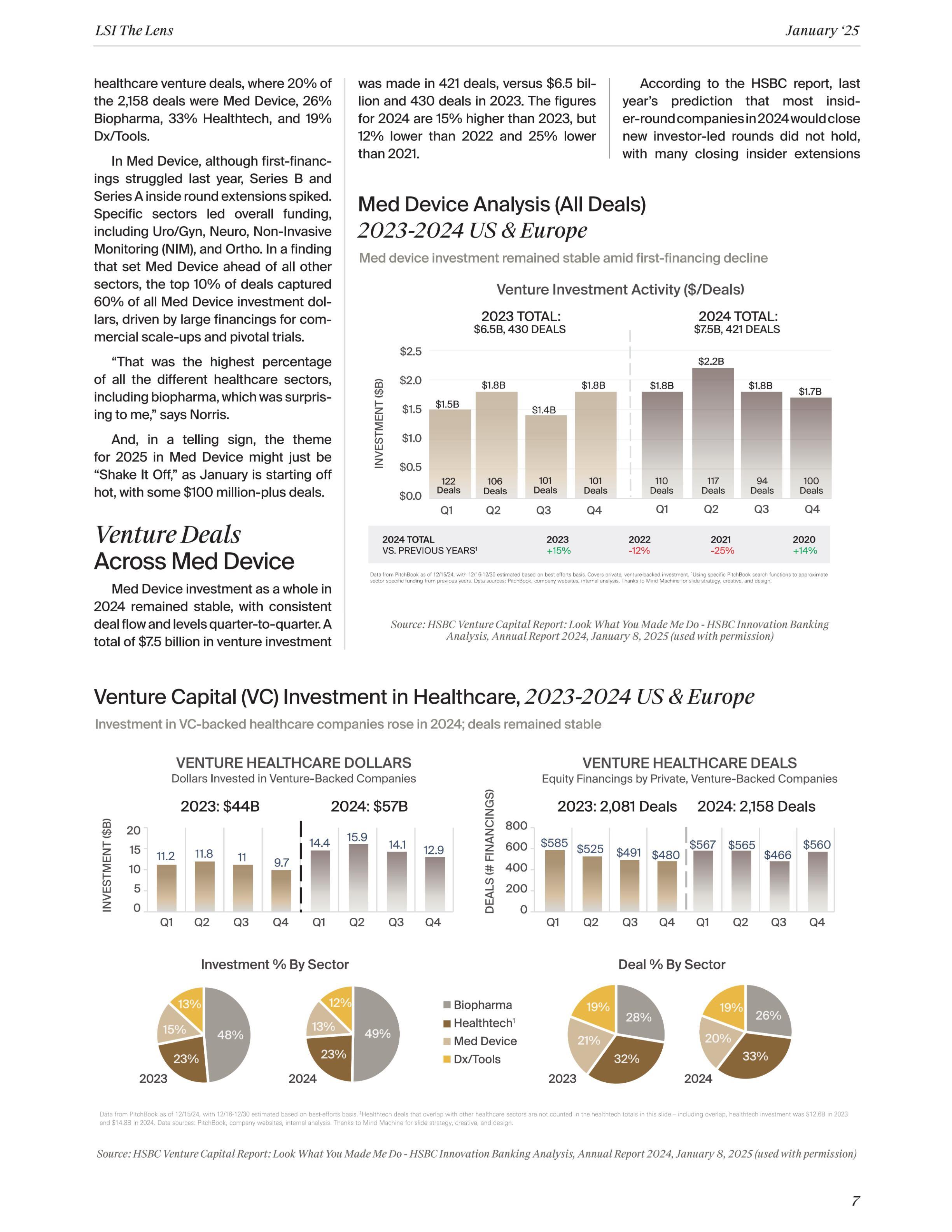


instead. However, the unexpected surge in new investor-led, later-stage financings highlighted the ongoing disparity between “haves” and “have nots.” Incredibly, the top 10% of Med Device financing attracted 60% of all investment dollars, the highest percentage among all sectors covered in the HSBC report.
A strong mix of capital sources drove later-stage financing last year, including traditional VC, growth, private equity, and crossover investors. Additionally, corporates from pharma, diagnostics, and medtech returned to the Med Device market, funding both larger and smaller deals. This diversity represents the most varied funding sources for Med Device in a decade, which is a positive sign for the sector, says the HSBC report.
The 20 largest financings in 2024 supported both commercial and development-stage projects. Eleven deals focused on commercial 510(k) products, two funded commercial PMA/De Novo deals, and seven were pre-commercial financings, including three development-stage PMA and four PMA pivotal trials, notes the report.
Typically challenging Series B venture deals stood out in 2024, with 47 new financings closed during the year. Among the 27 deals with valuation data,
15 were up rounds, seven were flat, and only four were down rounds.
“For the companies that are doing really well, there is capital out there in the Series B,” says Norris. “When we say there were 47 deals, you have to put that into perspective because that comprised just 11% of all deals in 2024, which means there’s still a lot of Series A companies out there that are on insider rounds that have no ability to raise that Series B. They’re still hoping to hit some sort of value inflection point to get a new investor to come in.”
Looking at all Med Device investment by indication in 2024, NIM, Orthopedic, and Vascular attracted investment, while Neuro and Cardio remained stable. Neuro maintained its leadership in Med Device investment, with brain/computer interface technologies (Blackrock, Precision, and INBRAIN) securing three of the top eight largest neuro financings. Neurostim technologies dominated, featuring in 21 of 46 deals in this area, led by Nalu, SPR, ShiraTronics, and Cala Health. The HSBC report points out that despite strong investment, neurology saw more flat and down rounds, 11 combined, than up rounds (five).
NIM, sensor-based technology that captures important patient data, rebounded strongly, surpassing $1 billion investment in 2024, says the report. Cardiovascular and metabolic secured 11 NIM deals each, followed by six deals in vital sign monitoring and uro/gyn.
$48M/8Deals
$88M/13 Deals
$131M/21 Deals
$85M/10 Deals
$48M/8Deals
$46M/6 Deals
$88M/13 Deals
$154M/7 Deals
“Urology/gynecology is a really interesting area, and I feel like we’re seeing a lot more activity there. Not only in interventional technologies but also in imaging, and in noninvasive monitoring and pelvic floor training,” adds Norris.
$131M/21 Deals
$97M/11 Deals
$85M/10 Deals
$103M/14 Deals
$46M/6 Deals
$154M/7 Deals
$40M/11 Deals $126M/12 Deals
$97M/11 Deals
$103M/14 Deals
$36M/5 Deals $15M/1 Deal
$40M/11 Deals
$126M/12 Deals
$53M/6 Deals $27M/6 Deals
$36M/5 Deals $15M/1 Deal
$33M/8 Deals $13M/1 Deal
$53M/6 Deals $27M/6 Deals
$33M/8 Deals $13M/1 Deal
$48M/8Deals
$131M/21
$48M/8Deals
$88M/13 Deals
$46M/6 Deals $154M/7 Deals $97M/11 Deals
$36M/5
Deal
Vascular and orthopedics investment also grew in 2024, according to the report. Ortho deals focused on both implant technologies (19 deals) and surgical robotics (seven deals; for more details see the Orthopedic Surgery Market Dive, The Lens, December 2024), while vascular financing included five $50-million-plus deals: two for commercial 510(k) products and three in clinical stages.
/ 8 Deals
$36M/5 Deals $15M/1 Deal
$53M/6 Deals $27M/6 Deals #33M/8 Deals $13M/1 Deal
Looking at imaging, investment has experienced significant annual fluctuations ranging between $500 million and $1 billion a year since 2020, according to the HSBC report. With a crowded market and limited acquirers, the focus in imaging has shifted to technologies that enhance therapy planning and guidance. In 2024, oncology and cardiovascular were the leading areas for imaging investments. A few examples include CapsoVision, a company with an innovative endoscopic capsule technology that raised $65 million, wearable MRI company Openwater that raised $54 million, and cardiovascular imaging company SpectraWave that brought in $50 million.
Cardiovascular investment in 2024 remained steady compared to 2023, but was down 15-20% from 2020-2022 levels. There were seven deals exceeding $40 million, including four in development or feasibility trials, two in pivotal trials, and one commercial-stage deal, according to the HSBC report.
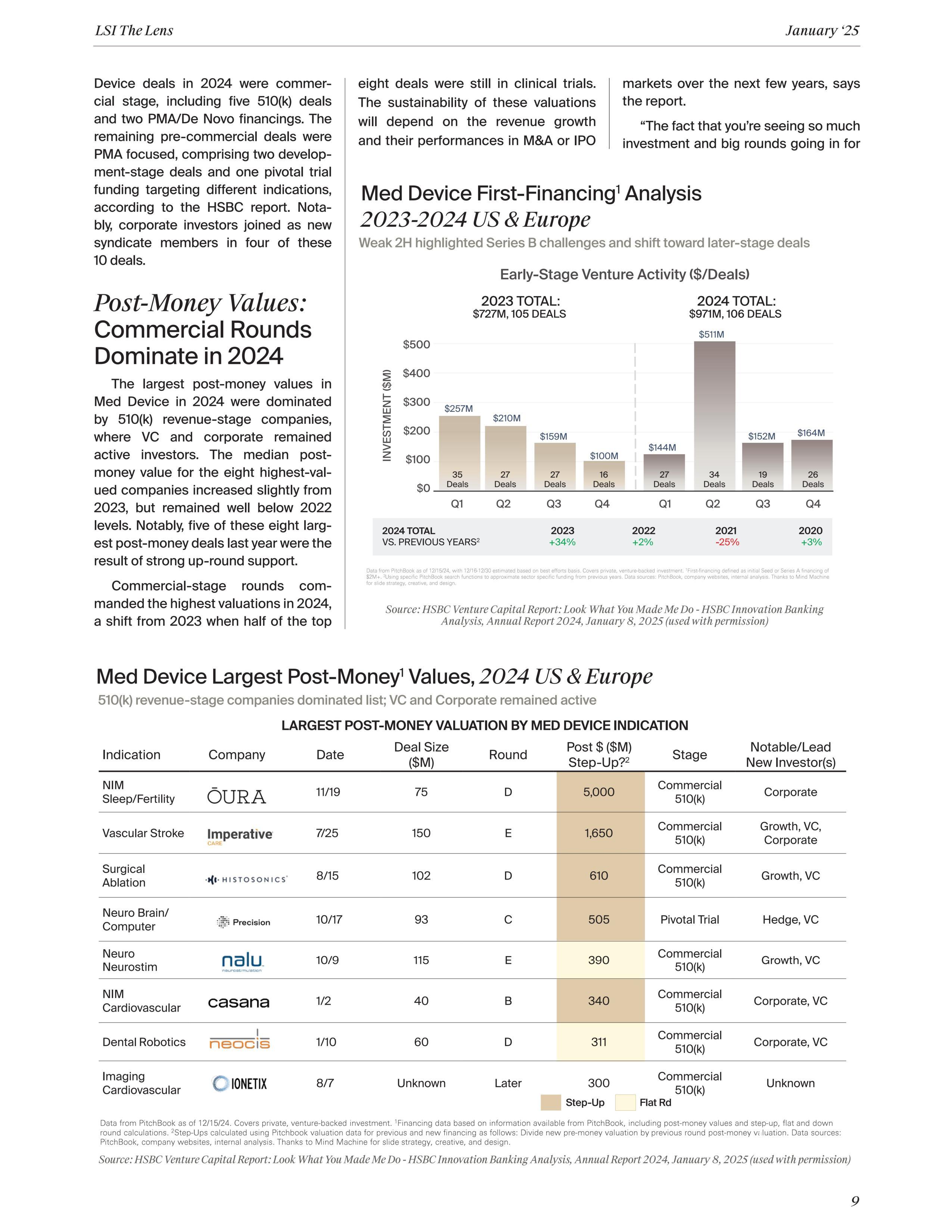
Speaking of commercial stage, these types of revenue-generating deals dominated larger Med Device venture investment in 2024, says the HSBC report. Seven of the 10 largest Med
Founded: 2021, as a spinout from the University of Oxford
Headquartered: London, UK
Technology: Fully implantable, adaptive neuromodulation therapy to treat mixed urinary incontinence
Financing: Series A
Amount: $100 million—one of the largest Series A rounds ever in Europe for a medical technology company
Deal Announced: June 10, 2024
Investors: The round was led by New Enterprise Associates (NEA) as part of a syndicate of new investors F-Prime Capital, Lightstone Ventures, and Intuitive Ventures, alongside existing investors Oxford Science Enterprises and 8VC
Founded: 2021
Headquartered: Amsterdam, The Netherlands Technology: Platform for AI-powered protein engineering; company reports that its technology has significantly sped up R&D projects between 1.2 and 12x, with cost reductions of up to 90%
Financing: Series B
Amount: $73 million
Deal Announced: November 26, 2024
Investors: Led by IVP with participation from previous investors Index Ventures and Kindred Capital
Founded: 2021, as a spin-out from UCLA
Headquartered: Malibu, CA
Technology: Microsurgical robotics; its flagship product, Polaris, combines state-of-the-art robotics, advanced medical imaging, and AI to push the boundaries of surgical precision, empowering surgeons to exceed current limitations
Financing: Series A
Amount: $30 million
Deal Announced: October 16, 2024
Investors: Led by ExSight Ventures and an undisclosed major corporate investor, with participation from Main Street Advisors and University of California (UC Investments), among others.
Sources: HSBC Venture Capital Report: Look What You Made Me DoHSBC Innovation Banking Analysis, Annual Report 2024, January 8, 2025; LSI Market Intelligence
these late-stage companies, I think it represents real optimism from folks who understand the market, that they believe that the IPO market will open up again. And I feel it’ll open again fairly soon for a small group of high performers,” says Norris.
Corporate investors participated in half of the top-valued deals in 2024, reflecting strong interest in supporting commercial growth. However, corporates are more willing to assume the dual risk of clinical and reimbursement challenges when the product aligns with their domain expertise, notes the HSBC report. Also, corporate syndicate members are more likely to acquire their device investment. In Med Device since 2018, M&A by a corporate that was also an equity investor yielded the highest percentage (15%), among healthcare sectors.
In 2024, a total of $971 million of private, venture money was invested in 106 total Seed or Series A Med Device deals of $2 million or more, according to the HSBC report. On the bright side, this dollar total was 34% higher than 2023, which saw $727 million invested in 105 first-financing deals. Looking back in time, however, the total invested in 2024 was just 2% above 2022, 25% less than 2021, and 3% higher than 2020.
What’s more, as noted in the figure, although investment levels stabilized in 2024, as compared to the decline in 2023, quarterly activity was inconsistent. The third and fourth quarters only saw 19 and 26 deals, respectively, for a total of $316 million, versus the anomaly that occurred in Q2, with $511 million invested in 34 deals (the three largest early-stage first financings of 2024, announced in Q2 and Q4, are detailed in the sidebar).
Annualized 2H 2024 investment shows total funding below 2023, trailing the prior three years by more than 40%, according to the HSBC report.
“The second half of 2024 was not very strong for early-stage Med Device,” says Norris. “And if you put that into context, it’s way behind previous years. The question is, is that a blip or is that the trend line? Will we see a billion-dollar investment year in 2025?”
New investors, drawn to the lower financing risk, attractive valuations, and potential for faster exits, showed a preference for later-stage investments last year, leading to an uptick in Series B investments in 2024, offering some relief to Series A early-stage companies funded in the previous few years. However, new Seed and Series A deals struggled to attract attention under these dynamics.
In a positive sign for 2025 and beyond, Norris notes that a lot of VCs have raised new funds, but they each tend to invest in just a few Med Device deals a year, he says. This includes investors like F-Prime Capital, NEA, Lightstone Ventures, Deerfield Management, and Vensana Capital. Vensana just announced in January the closing of Vensana Capital III, with $425 million in committed capital. The fund was oversubscribed at its hard cap with the support of the firm’s existing limited partners alongside select new institutional investors, and it brings Vensana’s total capital under management to approximately $1 billion.
“You’re seeing a lot of really good brand name VCs. You just don’t see them doing more than two or three deals,” says Norris. The problem for entrepreneurs is how do you thread the needle of
which VC firm still has an open slot looking for a company in your area, at your stage. It’s difficult. I think that we will see an uptick in large Series A syndicated investment in 2025, as investors band together to lead larger Series A deals with multiple investors. This will provide ample dry powder to help mitigate Series B financing risk.”
The rapid pace of medical device innovation is being powered by emerging technologies like AI and ML, to enhance early disease detection, improve diagnostic accuracy, and reduce the need for invasive procedures, among other game-changing benefits (also see the TechMed cover story featuring perspective from Alan Cohen of DCVC, and the BPH Market Dive in this issue). These advanced technologies are being supported by regulatory bodies such as CMS and FDA, with the FDA having approved approximately 1,000 medical devices driven by AI and ML to date for potential use in clinical settings. These and many other early-stage tech-enabled devices will be in high demand across the diagnostic and therapeutic landscape in the coming years, as illustrated by venture investment in 2024, and two of the three biggest early-stage deals, detailed previously.
According to the HSBC report, by Med Device indication, urology/gynecology, NIM, and neuro exceeded 2023 first-financing investment in 2024: uro/ gyn with $154 million in seven deals, up from $46 million in six deals in 2023; NIM saw a significant resurgence and reached $126 million in 2024, up from $40 million in 11 deals in 2023; and neuro deals totaled $88 million in 13 deals, compared to $48 million in 8 deals in 2023. Neurology reversed a four-year decline in early-stage funding, with half of the deals leveraging neurostim or brain/computer interface technologies.
In terms of valuation in Med Device in 2024, smaller Seed and Series A deals, particularly Software as a Service (SaaS) companies, commanded higher premoney valuations with four of the top five in NIM and imaging solutions, says
“I think that we will see an uptick in large Series A syndicated investment in 2025, as investors band together to lead larger Series A deals.”
– Jonathan Norris
the HSBC report. These include Dynocardia, BrainTemp, CairnSurgical, and Sonavex, all with pre-money valuations higher than $30 million.
Notably, four of the top six pre-money valuations in smaller Series A deals ($15M and smaller) were Series A extensions, underscoring the challenge of finding new Series B lead investors.
Neurostim and brain/computer interface technologies continue to attract substantial pre-money valuations for Series A deals, including Paradromics ($250 million pre-money), Invicta ($50 million pre-money), and Otolith ($46 million pre-money), all of which were Series A extensions, according to the report.
Orthopedic and NIM technologies both saw multiple financings with $70 million-plus pre-money valuations, including orthopedic deals Icotec and Cionic, and NIM deals Allez Health and Dynocardia. Two of these four deals were Series A extensions.
“What I’m finding is a lot of these higher pre-money valuation deals are Series A extensions. These companies should have raised their Series B and didn’t, but instead they raised an extension with their existing investors,” adds Norris. “These companies hope to use the additional capital to hit crucial value creation milestones to enable Series B investment by a new lead investor.”
Uro/Gyn interest continued to grow; neuro and NIM exceeded 2023 investment
Uro/Gyn








After a weak first half of last year for exits, Edwards Lifesciences and Stryker spurred a Q3 M&A rally, and the Ceribell IPO shined.

According to HSBC’s analysis, exits in the first half of 2024 were slow, with only three exits. In Q3, there was a significant uptick in M&A, with nine deals, including three led by Edwards. There were 14 venture-backed M&A deals in 2024, the highest since 2021.


Six deals in 2025 have already surpassed the $100 million mark, setting a positive tone for the year.
financing increased to 11.9 years in 2024, more than double 2021’s 5.8-year median. This extended timeline may be contributing to the decline in first financing, notes HSBC.
Edwards and Stryker drove M&A momentum in 2024 with three deals each, while Hologic completed two. (56)



While Q4 ‘24 saw limited private M&A activity with only two deals, it marked the year’s first Med Device IPO. Sunnyvale, CA-based Ceribell, in the NIM and Neuro categories with its AI-powered, rapidly deployable, point-of-care electroencephalography platform designed to address the unmet needs of patients in the acute care setting, priced its IPO on October 10, 2024. It






raised $180 million in IPO proceeds at a pre-money step-up from its last private round and has since traded to nearly two times its IPO post-money value, generating public market excitement for 2025. Ceribell’s IPO marked the first growth medtech IPO since October 2021. However, revenue predictability, and achieving profitability on IPO proceeds remain critical factors for future IPO candidates, says the HSBC report.



The report also points out that the median time to M&A exit from first
The new year is off to a brisk start in the Med Device fundraising space, with nearly $1 billion raised between January 1st and 15th, spanning a variety of market sectors. Incredibly, six deals in 2025 have already surpassed the $100 million mark, setting a positive tone for the year. What’s more, the new year already features a number of venture syndicates in action, that collectively are boosting many promising cutting-edge medical technologies over their clinical and regulatory hurdles.

After a weak 1H 2024, Edwards, Stryker spurred Q3 M&A rally; Ceribell IPO shined





















































Multiple acquisitions have already been announced in 2025, including Stryker’s planned acquisition of venous thromboembolism device company
Inari Medical for $4.9 billion, Boston Scientific’s $664 million purchase of Bolt Medical, and the completion of Hologic’s $350 million acquisition of Gynesonics. FIRE1, developer of the Norm heart failure management system, completed a $120 million financing round, led by Polaris Partners and Elevage Medical Technologies, joined by new investors Sands Capital and Longitude Capital, and existing investors Andera Partners, Gilde Healthcare, Gimv, the Ireland Strategic Investment Fund, Lightstone Ventures, Medtronic, NEA, Novo Holdings, and Seventure Partners (FIRE1 also received FDA Breakthrough Device Designation and has been accepted into the agency’s Total Product Lifecycle Advisory Program (TAP)).
Along with these deals, in January, Saluda Medical closed a $100 million financing to support its novel closedloop, dose-control neuromodulation platform for treating chronic pain. Existing investor Redmile Group led the round, with other existing investors Wellington Management, TPG Life Sciences Innovation, Fidelity Management & Research Company, Action Potential Venture Capital participating, and new investor Piper Heartland Healthcare Capital.
Wearable insulin delivery device company CeQur closed a $120 million financing round, investors undisclosed, to support its four-day wearable insulin patch technology.
In another hefty deal, Aspect Biosystems, a biotech company pioneering the development of bioprinted tissue therapeutics as a new category in regenerative medicine, closed a $115 million Series B financing round. The financing was led by Dimension, with participation from existing and new investors including Novo Nordisk, Radical Ventures, an undisclosed leading global investment firm, InBC, Pallasite Ventures, Pangaea Ventures, Rhino Ventures, and T1D Fund: A Breakthrough T1D Venture.
Also in January, Alleviant Medical, a privately held medical device company developing a no-implant atrial shunt for heart failure, announced a $90 million
financing to fund its second pivotal trial. Led by Gilde Healthcare, the round also adds Omega Funds and includes participation from existing shareholders S3 Ventures, RiverVest Venture Partners, Vensana Capital, Longview Ventures, Gilmartin Capital, TMC Venture Fund, and undisclosed strategic investors.
The new year already features a number of venture syndicates in action, that collectively are boosting many promising cuttingedge medical technologies over their clinical and regulatory hurdles.
Francis Medical, a privately held developer of an innovative and proprietary water vapor ablation therapy for the treatment of prostate, kidney, and bladder cancer, announced the completion of an oversubscribed $80 million Series C equity financing, the largest fundraising round to date for the company. Arboretum Ventures and Solas BioVentures co-led the Series C round. New investors Orlando Health Ventures and two additional strategics joined this round, along with previous investors Coloplast A/S and Tonkawa
The Series C proceeds will continue to fund the VAPOR 2 pivotal clinical study for the management of prostate cancer and the development and U.S. commercialization of its Vanquish Water Vapor Ablation proprietary prostate cancer treatment, expected by the end of 2025.
In another large deal announced in January, Cornerstone Robotics, a surgical robotics company, announced it raised more than $70 million in Series C funding. EQT led the round, with participation from Qiming Venture Partners, Alpha JWC Ventures, the Innovation and Technology Venture Fund, eGarden Ventures, CTS Funds, K2VC, and Long-Z Capital.
Also in the robotics space, Chinese robotics firm Fourier booked nearly $109 million in a Series E round, backed by Saudi oil giant Aramco's Prosperity7 Ventures. The Chinese asset management firm Peakvest and multiple investment companies from Shanghai, including the state-backed Guoxin Investment and Zhangjiang Science and Technology Venture Capital also participated. The company designs and produces exoskeleton and robotic rehabilitation products that are innovating the rehabilitation process. The company has developed upper body (arm) and lower-limb solutions, and it also has a humanoid robot.
Adding to the deals announced in January, Zeto, a medical technology company revolutionizing electroencephalogram (EEG) diagnostics with its cutting-edge devices and AI-driven cloud platform, announced the recent successful closing of a $31 million funding round. The round was led by MindWorks Global, a Michigan-based investment entity.
GT Medical Technologies, a medical device company focused on improving the lives of patients with brain tumors, completed a $37 million first close of a Series D financing round. The financing was led by Evidity Health Capital, alongside new investor Accelmed Partners. Also participating were existing investors MVM Partners, Gilde Healthcare, and Medtech Venture Partners. The funds will accelerate the completion of the ROADS clinical study that is focused on GammaTile for newly diagnosed brain metastases, and the GESTALT clinical trial for patients with newly diagnosed glioblastomas. In addition, the funds will support the continued commercialization of GammaTile, the company's FDA-cleared bioresorbable radiotherapy implant for the treatment of brain tumors.
LSI’s Market Intelligence Software Platform is your one-stop-shop for global market sizing and analysis, procedure volume data, startup company and deal tracking, curated insights, and more.
Scan the QR code to learn more!

Celebrating recent leadership shifts and other announcements and accolades impacting our global medtech community.
Adagio Medical, a leading innovator in catheter ablation technologies for treatment of cardiac arrhythmias, announced that founder Olav Bergheim departed as CEO and Chairperson of the Board of Directors of the company, effective December 13, 2024. Concurrently, the company announced the

appointment of LSI alumni Todd Usen as CEO and Director of the company, and Orly Mishan as Chairperson of the Board of Directors, effective December 13, 2024. Before joining Adagio, Todd served as President and CEO of uterine health company and LSI alumni Minerva Surgical, where he successfully restructured the organization, completed successful financings, and drove significant top-line growth. Prior to that, he was CEO of Activ Surgical, also an LSI alumni company, overseeing FDA/CE clearance of advanced surgical imaging technology and leading four fundraising rounds totaling over $92 million.
Augmedics, a pioneer in augmented reality surgical navigation, has named Paul Ziegler as its president

and CEO, filling the vacancy created when Kevin Hykes left for neuromodulation company CVRx. With the appointment, LSI alumni Gwen Watanabe will

transition from interim CEO and serve as Vice Chair of the Board. Ziegler, a 20-year med device veteran, assumes leadership as the company enters a new phase of commercial growth. In November, Augmedics announced a new milestone of 100 patients treated in a single week, as well as FDA clearance for a new CT-Fluoro registration method for the xvision Spine System that greatly expands navigation access for spine surgeons.
Freyja Healthcare, founded in 2017, is focused on improving the standard of women’s health in surgical and in-office procedures to enable physicians to provide safe, effective care. The company recently appointed LSI alumni Kurt Azarbarzin, former CEO of EndoQuest Robotics and Verb Surgical, and founder of SurgiQuest, to its Board of Directors. Azarbarzin has also held senior roles at CONMED Corporation, U.S. Surgical, and Tyco Healthcare.
Hyperfine, an LSI alumni company that has redefined brain imaging with the first FDA-cleared, AI-powered, portable magnetic resonance (MR) brain imaging system, the Swoop system, announced the appointment of Chi Nguyen as Vice President of Office Strategy and Partnerships, and Rafael Donnay as Vice President of Hospital Strategy and Health Economics, to provide leadership in key growth areas. These strategic leadership appointments bolster the capability of Hyperfine to drive commercial adoption in hospital inpatient and outpatient settings and expand to neurology office settings.
Integra LifeSciences President and CEO Jan De Witte retired on January 6, 2025, with former 3M Health Care President Mojdeh Poul replacing him

at the LSI alumni tissue regeneration and neurological treatment technology company. De Witte joined Integra in December 2021. Before joining 3M, Ms. Poul held global business leadership positions of increasing responsibility at Medtronic and Boston Scientific. In these roles, she accelerated penetration of neuromodulation and emerging cardiovascular technologies and therapies through the deployment of multifaceted market development strategies.
Intuitive Surgical has announced three executive promotions to strengthen the surgical robotics market leader’s leadership team and position the company for continued growth. Henry Charlton, a 21-year Intuitive veteran

who took on the CCO role in 2022, is moving up to the role of EVP and chief commercial and marketing officer, after having served in key leadership roles across multiple geographies (U.S., Europe and APAC) and global distribution and commercial organizations, regional marketing, global customer
services and commercial enablement functions. Gary Loeb, who joined Intuitive in 2022 as SVP and general counsel, now shifts to the role of EVP and chief legal and compliance officer. In the post, he oversees legal and governance functions and ESG reporting while serving as chief compliance officer. Jamie Samath is taking on the position of EVP, CFO and enterprise technology leader. Having originally joined the company in 2013, Samath became CFO in 2022 and enterprise technology leader in 2024.
LSI alumni Lazurite, developers of the first wireless arthroscopic surgical camera system, ArthroFree, (powered with aerospace-grade batteries used in spacesuits, on the International Space Station, and in satellites, following early support from NASA), announced that Mark Froimson, MD, has been appointed as the new CEO, after joining the company in 2018 as Chairman of the Board. During this transition, Lazurite’s founder and former CEO, as well as inventor of ArthroFree, Eugene Malinskiy, will take on the role of Chief Product and Technology Officer, to advance the next generation of ArthroFree products, including ArthroFree S4k and T4K. Dr. Froimson is also principal of Riverside Health Advisors, specializing in financing and accelerating healthcare innovation. He serves as chief medical officer for AngelMD, chairs the boards of Lazurite and Thrive, and is a board member for Pacira Biosciences, Sintx Technologies, and several nonprofits including the Arthritis Foundation.
Moon Surgical, a pioneer in laparoscopic surgical innovation, announced the appointment of Chris Toth as an

independent member of its Board of Directors. With over 20 years of experience in scaling and managing commercial operations, Toth brings extensive expertise in commercial acceleration. He is currently Executive Vice President and Group President of Baxter
International’s Kidney Care segment, which will be named Vantive upon completion of its pending sale to Carlyle by Baxter. Toth is the named CEO of Vantive. Prior to his current role, Toth was the CEO of Varian, a Siemens Healthineers company (and LSI alumni), where he spent 20 years in a variety of leadership roles, including President of Global Commercial and Field Operations.
LSI alumni Moon Surgical’s Maestro System is pioneering a new category in soft tissue surgery, addressing the critical need for efficiency in smaller operating rooms and outpatient settings. With its ability to support high-volume procedures while maintaining surgeon control and operational consistency, Maestro is uniquely positioned to transform the care of the 20 million soft tissue procedures performed annually worldwide.
NeoPredics AG Switzerland, a leader in predictive analytics and clinical decision support for maternal and neonatal health and LSI alumni, announced that Stefan Verlohren, MD, PhD, will

join the company as Chairman of the Medical and Scientific Advisory Board, effective January 1, 2025. Prof. Verlohren is a Professor of Obstetrics and the Chairman and Director of the Department of Obstetrics and Fetal Medicine at University Medical Center Hamburg-Eppendorf, Germany, with over two decades of experience in maternal-fetal medicine and an extensive background in clinical innovation.
NeoPredics also announced the appointment of medtech industry vet and LSI alumni Todd Usen as Chairman of the Board. With career highlights that include leading Minerva Surgical through an organizational transformation, spearheading the commercialization of Activ Surgical’s groundbreaking AI-powered technology, and driving global growth initiatives at Olympus Medical, Usen’s influence on the healthcare industry is both broad and profound. His passion for advancing
women’s health aligns perfectly with NeoPredics’ mission to deliver innovative solutions that improve outcomes for mothers and children.
Mike Karim, a seasoned life sciences entrepreneur, has joined Singaporebased Synapto Ventures as Venture

Partner, where he will lead business development activities in the United Kingdom, Europe and the U.S., including client relationship management and fundraising execution. Karim is currently mentoring start-ups and entrepreneurs, and providing support to life science companies through his company Mike Karim Consulting. He also contributes to the healthcare and medical industry through leadership roles in NHS governance and industry associations. Previously he was the CEO and co-founder of Oxford Endovascular Ltd., a spinout from the University of Oxford, developing a next-generation brain aneurysm flow diverter treatment (Karim presented Oxford Endovascular at LSI USA ‘24).
Gal Noyman-Veksler, PhD, a Partner at Tel Aviv, Israel-based VC firm Lionbird and a behavioral researcher, investor, and entrepreneur focusing on innovation in tech and healthcare, has been recognized on the “40 Under 40” list for 2025 by TheMarker, a Hebrew-language daily business newspaper.
At LionBird, Dr. Noyman-Veksler invests in U.S.-focused early-stage tech and scientific-driven startups looking to reset healthcare’s relationship with technology. She is passionate about using AI and big data to understand human behaviors and finding digital solutions to help users lead a better, healthier, and happier life, manage chronic conditions and improve general well-being, as described on her LinkedIn profile.
As an LSI alumni, Dr. Noyman-Veksler participated on two panels at LSI Europe ‘24 in Portugal: AgeTech Headwinds and Training & Supporting the Next Wave of Entrepreneurs.


March 17th - 21st, 2025
Waldorf Astoria, Monarch Beach, Dana Point, CA
In 2024, LSI USA convened 1,500 executives from emerging companies, venture capital and private equity firms, family offices, global strategics, professional service providers, ecosystem partners and more.
Registration and applications to present are now open for LSI USA ‘25. Get in touch to learn more about the event.


Highlights: Novel AI and implantable brain-computer interface technologies gain traction, migraine neuromodulation implant moves to pivotal stage, and wearable robotics company makes a public offering.
Anteris Technologies Global Corp., founded in Australia with offices in Minneapolis, MN, is developing the balloon-expandable DurAVR transcatheter heart valve for the treatment of severe aortic stenosis. The DurAVR device is the first biomimetic valve, which is shaped to mimic the performance of a healthy human aortic valve and aims to replicate normal aortic blood flow.
The company recently announced the closing of its initial public offering (IPO) of 14.8 million shares of its common stock in the U.S., at a price of $6.00 per share, resulting in aggregate gross proceeds of approximately $88.8 million. An additional 2.2 million shares of common stock are issuable pursuant to the underwriters’ option to purchase additional shares, if exercised in full. ATGC’s common stock is listed on NASDAQ under the ticker symbol AVR.
The DurAVR device, developed in partnership with the world’s leading interventional cardiologists and cardiac surgeons, is made using a single piece of molded ADAPT tissue, Anteris’ patented anti-calcification tissue technology. ADAPT tissue, which is FDA-cleared, has been used clinically for over 10 years and distributed for use in over 55,000 patients worldwide. The DurAVR transcatheter heart valve (DurAVR THV) System consists of the DurAVR valve, the ADAPT tissue, and the balloon-expandable ComASUR Delivery System.
Anteris currently intends to use the net proceeds from its IPO, together with its existing cash and cash equivalents, primarily for the ongoing development of its DurAVR THV and the preparation and enrollment of a randomized global pivotal study of DurAVR THV for treating severe aortic stenosis, with the remaining for working capital and other general corporate purposes, including the repayment of amounts owed under its convertible note facility.
Cleerly has developed an artificial intelligence-driven tool that uses coronary computed tomography angiography (CCTA) to generate a 3D model of patients’ coronary arteries and noninvasively measure plaque, vessel narrowing and the likelihood of insufficient blood flow. The company has generated evidence that shows its system is better than experts at assessing vessel narrowing, plaque volume and composition, helping it to recently secure Medicare coverage for the technology. It has also received a CPT Category I code for advanced plaque analysis.
Cleerly recently raised $106 million in a Series C extension funding round, led by global software investor Insight Partners and joined by Battery Ventures, with participation from pre-existing investors. With this new funding, Cleerly will continue to scale its commercial growth and clinical evidence generation, helping healthcare professionals improve outcomes for patients across the coronary care pathway.
In October, five Medicare Administrative Contractors (MACs) officially approved Cleerly’s AI-Quantitative Coronary CT (AI-QCT) scans for Medicare recipients exhibiting stable and acute symptoms suspicious of coronary artery disease. The new coverage took effect on November 24, 2024, for Palmetto GBA, CGS Administrators, National Government Services, and WPS administrative groups, and took effect for Noridian on December 8, 2024, impacting approximately 67 million Medicare beneficiaries. The new Medicare coverage will include Cleerly Labs Plaque Analysis for patients with acute or stable chest pain and no known CAD, as well as CAD-RADS 1-3. Additionally, the American Medical Association recently approved a permanent CPT Category I code for Cleerly’s AI-QCT advanced plaque analyses, effective January 1, 2026.
Cleerly’s AI-QCT technology, in several studies comparing it with invasive gold standards, has been demonstrated to be highly accurate in the assessment of the presence, extent, and composition of coronary atherosclerotic plaque and similar to consensus readings of Level 3 Expert Physician Readers. Cleerly ISCHEMIA, an additional AI-enabled product, has also been proven to demonstrate high diagnostic performance compared to invasive fractional flow reserve (FFR) and higher performance than such legacy tests as nuclear SPECT imaging and FFRCT.
Tempus AI, a technology company advancing precision medicine through the practical application of artificial intelligence in healthcare, recently announced a new decision by CMS that will allow reimbursement for assessments of cardiac dysfunction using the Tempus ECG-AF algorithm. ECG-AF is one of just a few FDA-authorized medical technologies in the country to be impacted by the new CMS decision, and this milestone allows Tempus to more broadly support clinicians in identifying patients at increased risk of atrial fibrillation/flutter.
Per the CMS policy to allow payment for certain Software as a Service (SaaS) devices in the hospital outpatient setting, CMS has assigned associated procedure codes for assessments with assistive algorithms like Tempus’ ECG-AF (CPT 0764T and CPT 0765T) to APC 5734, which has a Medicare rate of $128.90, effective January 1, 2025. This ruling is expected to allow hospitals to receive reimbursement for using Tempus’ ECG-AF to help identify patients at increased risk of AF.
Earlier this year, Tempus received 510(k) clearance from the FDA for the Tempus ECG-AF device, which was the first FDA clearance for an AF indication in the category known as “cardiovascular machine learning-based notification software,” and paved the way for clinicians to use this innovative algorithm in the care of their patients.
In other cardiac AI news, Philips and Mayo Clinic have entered a collboration aimed at advancing MRI for cardiac applications through AI. Under the collaboration, Philips aims to leverage Mayo Clinic’s proprietary AI technology in combination with its own. This combination could help reduce MRI scan times and improve the efficiency needed to relieve the burden on healthcare professionals. With AI, the two organizations say less experienced radiographers could potentially perform complex cardiac MRI exams. In addition to CT scans, MRI can prove useful when treating congenital heart disease or disease affecting the heart muscle. However, high costs and limited availability often hinder access to high-quality MRI, according to Philips.
Additionally, the research partnership intends to look at the potential of lower-field-strength MRI solutions, developed by Philips, to enable MRI installations in a broader range of applications. They also provide safer scanning options for individuals with implants sensitive to high magnetic fields.
Axoft, a Cambridge, MA-based neurotechnology company, is on a mission to unlock new treatments for patients suffering from neurological disorders by producing implantable
brain-computer interfaces (iBCIs) that answer critical unmet needs. The company recently announced the approval of its first-in-human clinical study, which will take place early this year at The Panama Clinic. The study will demonstrate the usability of Axoft’s novel soft materials that mimic the mechanical properties of brain tissue and are tailored to improve the stability of the tissue-electronics interface for iBCIs. Axoft’s study marks the first time this type of bio-inspired material is authorized for human use. The study will include up to five patients undergoing existing brain resection surgery. Study participants will be awake during the testing of Axoft’s iBCI, allowing for the decoding of volitional brain activity.
Axoft grew out of research from Dr. Jia Liu’s Lab of Bioelectronics at Harvard University, and the company has secured a licensing contract with the institution. In 2024, Axoft published papers outlining the capabilities of its novel brain implant technology in Nature Nanotechnology and Nature Neuroscience
Minneapolis, MN-based ShiraTronics has designed a minimally invasive implantable neuromodulation system, the ShiraTronics System, that offers a new and potentially more effective treatment option to address the symptoms of migraines. The fully implantable, programmable device goes just beneath the skin in the head, where it delivers precise electrical pulses tailored to disrupt migraine pain signals. The device offers precise, 24/7 electrical neuromodulation to reduce the frequency and intensity of migraine attacks, and could allow patients to maintain their daily activities with fewer disruptions, according to the company.
The company recently initiated an FDA investigational device exemption (IDE) pivotal trial to evaluate the company’s neuromodulation therapy for chronic migraine. The RELIEV-CM2 Clinical Study will implant up to 148 patients across the U.S. and Australia to assess the long-term safety and potential efficacy of ShiraTronics implantable neuromodulation therapy. Unlike other approaches that primarily manage migraine symptoms, the ShiraTronics System is designed to provide preventive, sustained relief for those who experience 15 or more headache days per month and have not achieved success with other treatments. For many of the millions who live with the challenging effects of chronic migraine, this novel approach could represent a potential new therapy option.
The device won FDA breakthrough device designation in 2021. ShiraTronics completed enrollment for its RELIEV-CM pilot study for the device in February 2024. In October 2024, the company raised $66 million to support the launch of its trial.
Inotec AMD, based in Cambridge, United Kingdom, has developed continuous topical oxygen therapy (cTOT) solutions that enhance healing and improve patients’ quality of life The company recently closed a $33 million Series C financing round, led by existing investors Amadeus Capital Partners, Meltwind, Puhua Capital, and the Wealth Club. The
financing marks a major milestone in the company’s mission to heal every chronic wound and improve patients’ lives on a global scale.
The proceeds from this round will be strategically used to secure national reimbursement in the U.S. for the company’s flagship cTOT product, NATROX O₂, ensuring broader accessibility for patients across the US. Additionally, the funds will support the expansion of the company’s leadership team, strengthening its global capabilities to drive future growth and innovation.
NATROX O₂ delivers a continuous flow of oxygen directly to the wound, creating an optimal environment for healing. NATROX O₂ has set new standards in wound care, providing a non-invasive, wearable solution that enables providers to offer a simple, user-friendly therapy designed to accelerate healing, even in wounds that have persisted for years such as diabetic foot ulcers, venous leg ulcers, pressure injuries, and non-healing surgical or traumatic wounds.
More than 100 million people in the world suffer from wounds that will not heal. In the U.S., wound care costs the health system an estimated $97 billion annually, with chronic wounds making up $50 billion of that expense.

FastWave Medical is a clinical-stage device company developing a portfolio of highly deliverable intravascular lithotripsy (IVL) catheters to treat artery calcification. The company, founded in 2021 and headquartered in Minneapolis, MN, recently announced the close of a $19 million funding round, increasing the total capital invested into the company to over $40 million. The latest investment was led by Epic Venture Partners, with participation from M&L Healthcare Investments and the company’s existing investors. The funds will fuel FastWave’s development momentum and provide support for ongoing regulatory and clinical initiatives for its product portfolio.
In May, the company announced successful 30-day results from the first-in-human study of its intravascular lithotripsy (IVL) technology. The prospective, single-arm study aimed to assess the IVL system’s safety and feasibility in patients with peripheral arterial disease (PAD) affecting the superficial femoral artery (SFA) and popliteal artery with moderate to severe calcium.
According to a news release, investigators successfully treated eight patients with moderate to severe calcified occlusions in the SFA and popliteal arteries. Evidence supported early safety and feasibility of the IVL system. Findings included 100% procedural success and 0% peri-procedural adverse events. FastWave reported 5.9% mean residual diameter stenosis post-therapy. At 30 days, the company also reported zero major adverse events, 100% patency and 0% revascularization.
Calcific plaque, which is prevalent in about 30% of CAD patients, resists vessel expansion, making it difficult to treat with traditional interventional modalities. FastWave Medical states on its website that it is elevating IVL delivery with a reduced crossing profile, optimized shaft for better trackability, a sleek, rupture-resistant design, and hands-free energy delivery with an improved generator UI that offers real-time procedural feedback.
LaNua Medical, a start-up spun out of University College Dublin (UCD), is developing an innovative technology that makes it easy, safe, and cheaper for doctors to treat internal bleeding, vascular malformations and many benign and malignant tumours.
The company has raised $6.3 million in seed funding to develop Ecore, a device that targets embolization procedures and aims to help improve patient outcomes, lower hospital costs, minimize duration of hospital stays, and reduce stress and procedure times for practitioners.
The funding will accelerate product development and market access, with a focus on the U.S. The funding round was co-led by Elkstone and Atlantic Bridge, with participation from Enterprise Ireland and Furthr VC. The funding will be used to accelerate product development and market access.
LaNua’s pioneering Ecore device allows doctors to restrict blood flow in a targeted segment of veins or arteries while still allowing ancillary medical tools such as guidewires and catheters to pass through it. The innovative device design will safely complement liquid and microparticle embolization, including localized intravascular radiation procedures performed by interventional radiologists worldwide. Exposure to x-ray radiation is also reduced for both patient and practitioner, and lowers the risk of human errors like inadvertently blocking blood flow to healthy adjacent organs. The company says the device will be adaptable for use in many areas of the human body to treat multiple disease states.
LaNua was founded in 2024 by interventional radiologist Dr. Cormac Farrelly, Tom Fitzmaurice (ex-Medtronic VP) and UCD biomedical engineers Dr. Eoin O'Cearbhaill and Dr. Sajjad Amiri.
Myomo, a Boston, MA-based wearable medical robotics company that offers increased functionality for those suffering from neurological disorders and upper limb paralysis, announced that it has priced an underwritten public offering of 3,000,000 shares of its common stock at a public offering price of $5.00 per share. Myomo expects the gross proceeds from this offering to be approximately $15 million, before deducting the underwriting discount and other offering expenses.
Myomo has developed and is marketing the MyoPro product line. MyoPro is a powered upper-limb orthosis designed to support the arm and restore function to the weakened or paralyzed arms of certain patients suffering from cerebrovascular accident (CVA) stroke, brachial plexus injury, traumatic brain or spinal cord injury or other neuromuscular disease or injury. It is currently the only marketed device in the U.S. that, sensing a patient’s own EMG signals through non-invasive sensors on the arm, can restore an individual’s ability to perform activities of daily living, including feeding themselves, carrying objects and doing household tasks. Many are able to return to work, live independently, and reduce their cost of care.
Sotelix Endoscopy is a Baltimore, MD-based startup developing next-generation therapeutic endoscopy devices for minimally invasive procedures dependent on the closure of gastrointestinal (GI) tract tissue, without the need for incisions.
The company recently announced the closing of a $1.7 million seed round financing, from individual investors, among them some of the world’s leading endoscopists, experienced medical device entrepreneurs, and other investors with clinical, technical, and business expertise. The investment will fund
the company’s research and development efforts, including in vivo testing of its innovative device.
Sotelix Endoscopy was founded by Dr. Mouen Khashab, a Professor of Medicine at Johns Hopkins University School of Medicine and one of the world’s leading endoscopists. The leadership team includes co-founder Dr. Venkata Akshintala, an associate professor at Johns Hopkins University School of Medicine and pioneer in gastrointestinal medical device development; chairman Mohamad Khachab, a successful entrepreneur and investor; and CEO John Schellhorn.
Marlborough, MA-based CardioFocus, a device company dedicated to advancing ablation treatment for cardiac arrhythmias, recently announced the first series of patients treated with the investigational OptiShot Pulsed Field Ablation (PFA) System for the treatment of paroxysmal atrial fibrillation, as part of the VISION AF clinical trial. The first-in-human trial will treat up to 50 patients in the coming months with 12-month follow-up planned, including critical remapping procedures to validate the efficacy of this novel technology.
The OptiShot balloon catheter is unique among the advanced generation of PFA catheters, with its ability to deliver circumferential lesions to the pulmonary veins with endoscopic visual confirmation of electrode-tissue contact, according to study researchers.
CardioFocus is taking a portfolio approach to PFA. In addition to OptiShot, CardioFocus will continue clinical trials evaluating the investigational QuickShot PFA System, a large area focal ablation catheter that integrates with various navigation technologies. In the EU, CardioFocus has treated over 6,000 patients with the Centauri PFA System, which uses a proprietary monopolar waveform with marketed contact-force sensing focal ablation catheters and mapping systems.
NanoVibronix, a device company that produces the UroShield, PainShield and WoundShield Surface Acoustic Wave (SAW) Portable Ultrasonic Therapeutic Devices, today announced it has renewed its exclusive distribution agreement with Ultra Pain Products, Inc. (UPPI) for the distribution of the company’s PainShield for another five years, with a minimum purchase commitment of $12 million. The company’s
decision to renew the distribution agreement comes in recognition of UPPI’s growth over the past four years and its evolution into a leading force in the non-opioid pain management and injury recovery industry.
NanoVibonix’s proprietary technology allows for the creation of low-frequency ultrasound waves that can be utilized for a variety of medical applications, including for disruption of biofilms and bacterial colonization, as well as for pain relief. The devices can be administered at home or in any care setting, without the continuous assistance of medical professionals.
MeMed, a developer in the emerging field of advanced host response technologies, recently announced that the FDA has granted Breakthrough Device Designation to its innovative MeMed Severity test, used to help manage patients with suspected acute infections and suspected sepsis by empowering clinicians with timely, data-driven, clinical insights.
Building on the scaling adoption of the FDA-cleared MeMed BV test, which redefines how clinicians differentiate between bacterial and viral infections, MeMed Severity aims to aid clinicians in rapid risk stratification and predicting disease progression, in conjunction with clinical assessments and other laboratory findings.
The test is based on advanced host-response technology that measures multiple proteins from a blood sample and uses machine learning to stratify the risk of a patient with a suspected acute infection deteriorating to severe outcomes within 72 hours or death within 14 days. Designed for emergency department settings, it has the potential to support critical decisions on triage, treatment, and optimized patient disposition.
With easy-to-interpret results in under 15 minutes, compatibility with high-throughput analyzers and minimal blood volume requirements, MeMed Severity will integrate into clinical workflows, driving value for both care providers and patients. The Breakthrough Device Designation will accelerate MeMed Severity's path to market, supporting reimbursement strategies, and ensuring that this critical tool reaches healthcare providers as quickly as possible.

Irish maternal health company NUA Surgical has secured €6.5M ($6.6 million) in Series A funding, led by EQT Life Sciences. The round was also supported by new investors Kidron Capital and the Texas Medical Center Venture Fund, and existing investors including Enterprise Ireland and business veterans from Ireland and the US. The proceeds will be used to drive the regulatory clearance and early commercialization of the company’s product, the SteriCISION C-Section Retractor, specifically designed to address the unique challenges of Caesarean-section (C-section) surgery.
With this new investment, the Nua Surgical Board will be strengthened with several experienced female board members. Anne Portwich, Partner at EQT, and Anula Jayasuriya, Co-Founder of Kidron Capital, will join as Board Directors, while Gabrielle Guttman of TMC Venture Fund and Prashanthi Ramesh of EQT will serve as Board Observers.
C-sections are the most common major surgical procedure globally, with over 30 million performed each year. The SteriCISION C-Section Retractor is ergonomically designed to provide fast, adjustable, and safe retraction, enabling clinicians to deliver the baby, repair tissue, and crucially, identify bleeds. As a single-use sterile device, it aims to reduce the risk factors that lead to surgical complications, benefiting the patient, clinician, and the healthcare system. The new investment will support Nua Surgical's next phase of development and the early commercialization of SteriCISION.
“This is a meeting where we find the best quality—size of the meeting, the venue—it’s conducive to proper one-on-one meetings and multiple interactions with entrepreneurs. The entire ecosystem shows up at the LSI conference."
Dr. Luc Marengère
Managing Partner,
TVM Capital Life Science

The industry’s most comprehensive and trusted global procedure database, with coverage of 300+ diagnostic and therapeutic procedures across 37 countries.

Fred Alger Management, T. Rowe Price Associates, Mumtalakat, Parkway Venture Capital, Breyer Capital, Tizvi Traverse, S32, US Innovative Technology Fund, Ava Investors, Eric Schmidt, Marc Benioff, Yann LeCun, and others
Your one-stop-shop for global market sizing and analysis, procedure volume data, startup company and deal tracking, curated insights, and more. Scan QR code for details.

Fund (led), Nexus NeuroTech Ventures (led), Action Potential, Johnson & Johnson, Lightstone, Lux, Google Ventures (GV), OSF Ventures, Ascension Ventures, TriVentures, Reimagined Ventures, Peak6,
The integration of advanced AI, machine learning, and computer vision tech with medical devices, dubbed the TechMed revolution, is a bold, empowering new chapter happening now in healthcare, and differentiated from MedTech, describes Alan Cohen, General Partner at DCVC. This data-driven new era is poised to improve patient outcomes worldwide.
Not all that long ago, groundbreaking medical technology began to change healthcare as we know it. Advanced, hardware-centric devices impacting the lives of patients during the MedTech Revolution, ranging from transcatheter heart valves to knee replacement implants, have improved patient outcomes and quality of life for problematic and prevalent health conditions. Today, the traditional, $500-billion-a-year
Source: LSI Europe ‘24
medtech industry is being overshadowed by the transformational power of data, in a bold new era in patient-centered healthcare, coined by Alan Cohen, General Partner at deep tech venture capital firm, DCVC as TechMed.
Cohen, whose keynote address at LSI Europe ‘24 in Sintra, Portugal in September 2024, “The TechMed Movement Has Arrived,” offers a thought-provoking
view on the emerging but current impact of data and deep tech in healthcare. This tech-enabled movement represents a shift from traditional medical technology, to a data-first, innovation-driven approach aimed at empowering healthcare professionals and transforming patient outcomes, describes Cohen.
"TechMed is a speciation event—a new species emerging from medtech,

driven by a technology-first approach," he told the audience.
Cohen’s tech background and role at DCVC parallels his futuristic, but realistic view. He been a successful entrepreneur, technology executive, and board member for over 25 years for a range of companies including DCVC-backed Illumio, Nicira (acquired by VMware), Airespace (acquired by Cisco), Cisco (where he led the $25 billion enterprise marketing and solutions organization), General Growth Properties, and IBM
The TechMed paradigm represents the evolution of traditional medtech by integrating advanced technologies such as sensors, machine learning (ML), and proprietary datasets, says Cohen. This approach prioritizes technology as the foundation for medical applications, rather than retrofitting existing medical disciplines. By blending advanced artificial intelligence (AI), ML, and computer vision with hardware, TechMed companies—differentiated from their MedTech brethren—are poised to tackle some of the healthcare industry’s most difficult and long-standing challenges.
Advancements in the use of AI/ML are enabling the identification of population trends and the ability to forecast health outcomes by evaluating enormous amounts of medical data, including images, test results, and patient records. These powerful, novel capabilities promise to offer healthcare professionals a whole new level of diagnostic accuracy, individualized treatment strategies, and effective patient monitoring, that hold the potential of democratizing access to high-quality care, reducing costs, and improving patient quality of life worldwide.
And, the term TechMed is catching on big time within industry. NVIDIA, a leader in AI hardware and software, promoted the term at the recent annual J.P. Morgan Healthcare Conference in San Francisco. Tom Oxley, CEO of Synchron, described in a LinkedIn post and in presentations that there is a new, fast-growing and high-potential sector that is data-first in healthcare, TechMed, that they are calling Digital Devices, that is comprised of robotics and Brain Computer Interfaces (BCI). This is akin to the “'TechBio” subset of Biotech that is data-first, according to Oxley. To the Medtech analysts and VCs, he says this is real, it is coming, and it is big.
Here we take a look at the main takeaways from Cohen’s keynote and his vision of TechMed, which he presented to the LSI Europe ‘24 audience as offering unprecedented opportunity for the MedTech and healthcare ecosystem.
The seamless integration of hardware and software is a driving force behind transformative medical technologies, enabling more accurate diagnostics, streamlined workflows, and improved treatment outcomes, notes Cohen. As a powerful example, innovative medical devices are combining AI with imaging tools to identify anomalies with a level of precision that was once considered impossible. Robotic-assisted surgery platforms use 3D imaging and haptic feedback to allow surgeons to perform minimally invasive procedures with enhanced precision. This fusion is also being used to improve workflow efficiency, such as automating data collection and analysis in hospitals to reduce the burden on clinicians and increase operational efficiency.
"Hardware and software are fusing in transformative ways, changing how we approach healthcare," he says.
A central theme of the TechMed revolution is its ability to empower rather than replace healthcare professionals, says Cohen. Tools like AI-assisted imaging systems enable radiologists to interpret scans faster and more accurately, while wearable technologies provide nurses with real-time patient vitals, allowing for timely interventions.
TechMed innovation focuses on providing tools that enhance the capabilities of medical practitioners, ranging from sonographers to surgeons. For instance, AI-powered guidance systems allow non-specialists to perform tasks like capturing diagnostic-quality images, helping to alleviate workforce shortages. This democratization of expertise means that care can reach more patients, particularly in underserved areas.
“The goal is to give superpowers to people to do their job better, to be more effective,” emphasized Cohen.
AI and machine learning are unlocking new possibilities across the healthcare continuum. Examples include Caption Health’s use of AI to guide imaging and diagnostic tools (more about Caption Health on the next page), as well as semantic AI, which creates hyper-detailed anatomical models. These innovations are revolutionizing areas like personalized medicine, where algorithms analyze a patient’s unique characteristics to tailor treatments.
• More actionable, precise, and accurate care
• Reduced costs through scalable technology
• Broader global accessibility
• Integration of care into everyday life
"Machine learning and neural networks collect information from the periphery and bring it back—just like what we do in medicine," notes Cohen.
Despite AI's transformative potential, its adoption in healthcare lags behind other industries, he says. Cohen emphasized the urgency for the medical device sector to harness AI for diagnostics, digital therapeutics, and clinical insights.
He also introduced the concept of "Semantic AI," focusing on using AI to create highly detailed anatomical models and improve the understanding of the body's interactions with drugs, devices, and procedures. Semantic AI is poised to revolutionize healthcare by integrating and visualizing diverse health data streams, including lifestyle,
medical interventions, and environmental factors.
"Semantic AI will enhance our ability to understand human anatomy and its interactions with medical interventions down to the millimeter," he describes.
Data is the backbone of the TechMed revolution. And, the transition from static, point-in-time measurements to continuous, real-time data flows is enabling more proactive and personalized care, says Cohen. Wearables, implants, and monitoring devices are now capable of generating dynamic datasets that allow clinicians to detect issues early and intervene proactively. For instance, platforms like Sickbay aggregate data from multiple devices to monitor for conditions such as sepsis or heart failure, enabling faster and more informed clinical decisions, he notes.
"Using AI to enhance healthcare outcomes is dramatically more important than building models to write marketing copy," says Cohen.
In effect, Cohen notes, both in his keynote address and in his thesis entitled “The TechMed movement has arrived,” that TechMed companies cultivate a “data river” rather than a data moat, a continuous and dynamic flow of data that spans the entire patient lifecycle, from diagnostics to intra-procedures, and post-procedure phases. The depth, breadth, and constant updating of these datasets ensure they remain comprehensive and relevant, with each new data input enhancing the model’s capabilities. This contrasts with traditionally “siloed” institutional data stores that are fragmented, narrow in scope, and quickly become stale, Cohen notes.
This concept aligns with the growing trend of real-time health monitoring and adaptive care models. For the medical device industry to thrive, leveraging continuous, real-time data from implants, wearables, and other devices is crucial, Cohen continues. "To fulfill the promise of TechMed, we must move from static snapshots to dynamic, real-time insights."
TechMed is as much about purpose as it is about innovation. Many TechMed pioneers are inspired by deeply personal experiences, driving them to create solutions with lasting societal impact. For Cohen, when his father, a Navy corpsman providing medical care during the Korean War, was diagnosed with glioblastoma in 2016, he talked Cohen into leaving the tech industry and doing something that would have a more long-lasting impact on society. Based on his father’s impact on troops during the war, he chose the field of medicine.
“To fulfill the promise of TechMed, we must move from static snapshots to dynamic, real-time insights.”
– Alan Cohen
This mission-driven mindset fosters TechMed innovations that prioritize accessibility, affordability, and meaningful change. For instance, autonomous robotics, like those being developed by Remedy Robotics, leverage advanced robotics inspired by self-driving cars, and aim to provide precision care in remote settings. The implications of this advancement can also help address long-standing concerns with global disparities in healthcare access, says Cohen.
Drawing from his work with AI pioneers, Cohen underscores the role of neural networks and machine learning in advancing medical technologies. Two DCVC-backed companies exemplify the TechMed thesis: Proprio and Caption Health, he describes.
Proprio is a surgical navigation and intelligence company focused initially
on spine procedures like posterior spinal fusions to correct curvature of the spine. The company’s flagship product, the Proprio Paradigm platform, revolutionizes the surgical process by providing AI-guided visualization and data-driven insights that improve precision, efficiency, and patient outcomes. It relies on an advanced array of sensor technologies including lightfield imaging, which captures visuals with far more depth and dimensionality than traditional cameras. The company’s system gives surgeons real-time 3D guidance akin to “surgical GPS,” continuously mapping and adjusting the operative field to ensure optimal implant placement and spinal alignment. It leverages computer-vision AI models built from analyzing hundreds of prior procedures by world-class surgeons across 250+ gigabytes of multi-modal operating room data per procedure. This proprietary data pipeline allows Proprio’s system to improve with every surgery, learning new techniques and adjusting for any intraoperative changes.
In 2023, Proprio secured landmark FDA 510(k) clearance as the first such AI-guided surgical navigation system for spine surgery. With a robust pipeline of further FDA submissions across other spine procedure types and ultimate ambitions to expand into orthopedics, neurosurgery, and more, Proprio is looking to become the default “co-pilot” platform, augmenting surgeons across myriad therapeutic areas.
Companies like Proprio demonstrate the strength of diverse expertise, blending machine learning, medical device engineering, and clinical knowledge to advance surgical technology. "The fusion of disciplines at Proprio shows how collaboration builds entirely new platforms for the industry," describes Cohen.
Caption Health, acquired by GE Healthcare in 2023, took a similar datadriven AI approach but for the imaging diagnostic realm. Its breakthrough product was an AI guide that enabled minimally trained operators to capture diagnostic-quality ultrasound image sequences on par with expert sonographers or cardiologists. By studying tens of thousands of prior exams, Caption’s AI learned to identify standard views and provide real-time instructions on properly manipulating the ultrasound wand.
For conditions like heart disease, which require assessing the heart’s ejection fraction, Caption’s “virtual instructor” democratized the ability to administer these studies at the point of care, rather than having to rely on scarce, trained staff constantly shuttling between imaging rooms and offices. Caption subsequently expanded its product into other use cases like lung imaging and maternal health, creating AI-guided imaging protocols for a variety of acute and chronic conditions, describes Cohen.
The human and economic impact of the TechMed movement is multifaceted and transformative, says Cohen. Unlike the slower innovation cycles of traditional MedTech companies, TechMed players like Proprio and Caption more resemble agile tech industry disruptors, he notes.
This new revolution is not just a technological shift but a profound transformation of how healthcare is delivered. By harnessing the power of data and deep tech, this movement holds the promise of democratizing access to high-quality care, reducing costs, and improving lives worldwide. And, it's already happening.
Ultimately, TechMed stands at the forefront of a healthcare transformation, promising better patient care, more efficient medical practices, and a more economically sustainable healthcare system, says Cohen. It also means that the traditional medical device business model faces disruption. Startups and incumbents alike must rethink how they deliver value, says Cohen. Subscription-based and value-driven pricing are emerging as sustainable models, aligning costs with benefits for providers and patients.
"If you provide a compelling solution for users, they will help pull you through on the business model," he describes. Cohen concludes his address with a call to action for MedTech innovators: the time to lead this change is now, and to work with tech-forward companies to not only innovate but to profoundly enhance human health. For those ready to embrace the TechMed vision, the possibilities for impact are immense.

Alan Cohen is a partner focusing on an array of AI-enabled health tech, energy, security, and enterprise opportunities that are transforming industries. In addition to his board roles, he works across our portfolio on product/market fit, go-to-market strategy, and executive leadership development, and led DCVC’s successful exits at Element AI, Evolv (Nasdaq: EVLV), and Caption Health.
He has been a successful entrepreneur, technology executive, and board member for over 25 years for a range of iconic companies, including DCVCbacked Illumio, Nicira (acquired by VMware), Airespace (acquired by Cisco), Cisco (where he led the $25 billion enterprise marketing and solutions organization), General Growth Properties, and IBM. He has authored over 200 articles, undertaken over 1,000 press interviews, and delivered over 100 keynotes at industry conferences. In addition to his DCVC responsibilities, Cohen is a member of the Advisory Committee of the Secure Community Network, a mentor at the NYUbacked Endless Frontier Lab, and an adviser to Black Women on Boards.
He received a bachelor’s degree in English from SUNY Buffalo, a master’s degree in English from the University of Vermont, a master’s degree in international affairs and economics from the American University School of International Service, and an MBA from New York University.
June 10th - 13th, 2025
The Ritz-Carlton, Millenia Singapore
With the global community built through LSI USA and LSI Europe and our partnerships in Singapore, China, Korea, Japan and more, LSI Asia will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Like LSI USA and LSI Europe, LSI Asia will not be held in a chaotic convention center. We have identified a world-class venue in Singapore, the gateway to Southeast Asia, for our inaugural event where senior executives can do real business.


As we launch in this new geography, we are offering a unique opportunity to a small set of value-add, mission-aligned partners looking to elevate their global presence and develop new business.
Get in touch with us to learn more and become a founding sponsor.

Thank you to our founding sponsors
The field of gastrointestinal (GI) endoscopy is undergoing a technological revolution, with new devices and systems incorporating AI/ML and robotics that can significantly enhance diagnostic accuracy, therapeutic capabilities, and patient comfort.
Endoscopy—a cornerstone of modern medicine—enables the minimally invasive visualization, diagnosis, and treatment of upper and lower gastrointestinal (GI) conditions using a flexible fiberoptic scope. From detecting colorectal cancer to managing chronic GI disorders, this critical procedure is being revolutionized by new technologies that promise to enhance diagnostic precision, improve patient outcomes, and lower costs, all while expanding access to care.
The stakes in GI care are immense. In the U.S. alone, an estimated 60 to 70 million adults suffer from GI conditions, yet many delay seeing a physician until their symptoms become severe. This delay is particularly deadly for colorectal and esophageal cancers, which are often diagnosed in the advanced, hard-to-treat stages. Colorectal cancer, the second leading cause of cancer-related deaths in the U.S., is rising in incidence—a trend that underscores the urgency for transformative, patient-centered innovation in GI endoscopy.
Emerging tools like artificial intelligence (AI) and machine learning (ML) are revolutionizing healthcare, and they are reshaping GI imaging. ML can process every frame in real-time, while AI aids in detecting and classifying polyps, diagnosing disease, predicting cancer risk, and minimizing missed diagnoses. While still evolving, these technologies are set to become indispensable in the fight against “silent killers” like colorectal cancer, especially as the demand for GI procedures soars.
Ahead, we’ll examine procedure volumes, competitive dynamics, and cutting-edge innovations poised to redefine the future of GI endoscopy and transform patient care.
During the forecast period 20232028, global GI endoscopy procedure volumes are projected to increase at a CAGR of 5.7%, according to LSI Market Intelligence. GI surgery, whether diagnostic or interventional, is one of the most commonly performed procedures worldwide. This is largely due to the fact that many GI conditions are more common in older age, as is the case with cardiovascular and orthopedic conditions.
Total sales of GI endoscopy devices— including GI endoscopes for imaging and endoscopic instruments for intervention, for the purposes of this analysis—are projected to increase at a CAGR of 6.5% during the forecast period. While market growth is subjected to intense downward competitive pressures, the introduction of advanced robotic systems will provide an opportunity for companies to market higher value consumables that are compatible with their systems.
Growth in this market is driven by continued conversion of open surgical procedures to less-invasive laparoscopic
or endoscopic procedures, enabled by device advances including:
• Robotic surgery systems providing enhanced capabilities for performing endoscopic surgery procedures,
• Improved endoscopic imaging and guidance technologies,
• Advances in endoscopic surgical instruments including instruments requiring smaller-diameter access ports, and
• More effective energy-based laparoscopic surgery tools.
Global GI Endoscopy Devices, Unit Volumes, 2023—2028
GI Endoscopy Devices, Market Forecast, 2023—2028
Source: LSI Market Intelligence, GI Endoscopy Devices Market Snapshot, Published June 2024
As of 2023, three powerhouses— Medtronic, Ethicon/J&J, and Olympus— dominated the global GI endoscopy market, commanding a combined 57% market share, with Medtronic leading the pack at 21.5%. However, the remaining 43% has become a battleground for a dynamic cohort of challengers, including Applied Medical, B. Braun, Boston Scientific, Conmed, Integra Lifesciences, Intuitive Surgical, Karl Storz, Purple Surgical, Richard Wolf, Stryker, and Teleflex, among others, according to LSI Market Intelligence.
What’s driving the competition? Robotics and AI. These rapidly evolving technologies are redefining the rules of engagement in GI endoscopy, delivering breakthroughs in maneuverability, diagnostic precision, and therapeutic capability. By enhancing the tools available to endoscopists, these innovations are reshaping the competitive landscape and setting the stage for a new era of smarter, faster, outcomes-impacting GI care.
In this fiercely competitive market, the ability to leverage robotics and AI is becoming more than an advantage—it’s
the key to staying relevant. And as these technologies continue to evolve, so too will the expectations of patients, providers, and the industry (and investors) as a whole.
In 2023, Medtronic increased its market share in the GI endoscopy space, edging out J&J subsidiary Ethicon as the global market leader. Medtronic has made bold moves into robotics and AI, including with its Hugo robotic-assisted surgery (RAS) system and associated consumables, that are part of its Medical Surgical Portfolio.
Along with sales of Hugo outside the U.S., Medtronic has indicated that sales of its GI Genius Intelligent Endoscopy Module bolstered overall revenue attributable to endoscopy solutions in 2023. GI Genius was the first commercially available computer-aided detection (CADe) system using AI to identify colorectal polyps. Its significant impact has been well-researched with more than 30 academic and real-world publications across a combined total of 23,000+ patients, according to Medtronic. Based on the current global installed base, the GI Genius system has the potential to impact 2.7 million patients annually.
In April 2024, Medtronic released ColonPRO, the latest generation software for the GI Genius intelligent
Notes: Figures may not sum due to rounding. “Others” include Applied Medical, B. Braun, Boston Scientific, Conmed, Integra Lifesciences, Intuitive Surgical, Karl Storz, Purple Surgical, Richard Wolf, Stryker, and Teleflex, among others
Sources: LSI Market Intelligence, GI Endoscopy Devices Market Snapshot, Published June 2024; annual company reports; SEC filings; and investor relations materials
endoscopy system, that allows the system to take a significant leap forward in AI-driven polyp detection by introducing an enhanced algorithm supported by a dataset that is double the size of the previous one, resulting in a notable improvement in its detection capabilities. Specifically, there's been a 9% reduction in false positives, according to the company. The algorithm improvements also made ColonPro better at detecting the most challenging lesions, like tiny, flat polyps hiding in the corner of the camera’s field of vision that the system might have missed previously.
The updated ColonPro software automatically summarizes colonoscopy procedures with quality metrics and other details, including withdrawal time, bowel cleanliness (Boston Bowel Preparation Scale) throughout the procedure, and key anatomical landmarks visualized within the patient’s colon.
Also in April 2024, Medtronic announced a collaboration with Modernizing Medicine (ModMed), a pioneer in healthcare technology and electronic health records (EHR), further emphasizing its commitment to a synergistic approach to the future of endoscopic care. ModMed's gGastro EHR software complements Medtronic’s GI Genius system, offering robust documentation solutions.
Added to this success, last May, Medtronic received FDA clearance for the next-generation PillCam Genius SB capsule endoscopy kit. PillCam capsule endoscopy has been in use for more than 20 years. Medtronic won FDA clearance for the PillCam Small Bowel (SB) 3 system for remote endoscopy procedures in late 2021.
The new PillCam Genius SB kit offers flexibility through simplified equipment management, compared to the previous generation system. It enables the procedure to take place at a hospital, clinic, or the home. Medtronic notes that its newest offering features a link device that replaces traditional data recorders as well. The wearable, single-use device efficiently stores images transmitted by the capsule, via an adhesive patch. It also alerts patients via haptic vibrations, audible signals, and LED lights when they complete the procedure.
While J&J/Ethicon reported overall increases in revenue that is in part attributable to sales of the company’s endoscopic solutions within the Ethicon portfolio, innovative products from J&J that take advantage of current market demands for solutions that enhance clinical decision-making and robotics have lagged behind other major competitors in this market.
J&J is still in the process of obtaining regulatory approval for its OTTAVA robotic system for general surgery. In November 2024, J&J announced that the FDA had approved the OTTAVA robotic surgical system investigational device exemption (IDE), which allows a clinical trial to begin at U.S. sites. J&J’s Ottava has been designed with six arms to allow for enhanced control, flexibility, and patient access during surgery, with plans that the system will be able to perform a variety of procedures that overlap with Intuitive Surgical’s da Vinci system, including GI. J&J invested in the growing robotic surgery market by acquiring Auris Health and its Monarch system for bronchoscopic procedures in 2019.
Olympus is distinguished from both Medtronic and J&J as it offers both imaging and interventional endoscopic devices. In 2023, Olympus launched the EVIS X1 endoscopy system that features Texture and Color Enhancement Imaging (TXI) technology, designed to increase the visibility of lesions and polyps by enhancing image color and texture during endoscopic screening. It also features Red Dichromatic Imaging (RDI) technology, designed to enhance the visibility of deep blood vessels and bleeding points, and Brightness Adjustment Imaging with Maintenance of Contrast (BAI-MAC), that corrects the brightness levels in dark areas of the endoscopic image, while maintaining the brightness of lighter areas, to increase the total distance view.
Intuitive Surgical is another major competitor in the GI endoscopy devices market, as the global robotic-assisted surgery leader, with an estimated market share of 9.1% and corresponding product sales of $2,202.9M in 2023, according to LSI Market Intelligence. In March 2024, Intuitive obtained FDA 510(k) clearance for its fifth-generation robotic system, da Vinci 5. Da Vinci 5 builds
on Intuitive’s da Vinci Xi’s highly functional design, which surgeons and care teams around the world have used in more than 7 million procedures to date. The new system includes more than 150 enhancements, including improved accuracy and precision, next-generation 3D display and image processing, and first-of-its-kind force-sensing technology. The company’s da Vinci surgical systems are designed to help surgeons perform minimally invasive surgery and offer surgeons high-definition 3D vision, a magnified view, and robotic and computer assistance.
The field of GI endoscopy has attracted many start-ups looking to address unmet needs in the space or improve on current technology with single-use endoscopic solutions that eliminate the need for costly disinfection and reprocessing, features that enhance user control, and wireless endoscopic solutions. A few of these promising innovations from our LSI alumni are highlighted below.
Wales, UK-based IQ Endoscopes is pioneering sustainable endoscopy technologies for the early diagnosis of diseases, helping to create a world where everyone has access to sustainable sterile endoscopy. Using a novel and patented engineering approach, the company’s range of single-use endoscopes will deliver much needed change within endoscopy, addressing the huge demand for the service globally. Their mission is to help endoscopists and their service providers improve their way of working, giving them greater flexibility, choice, and control so they can increase capacity and ultimately save patients’ lives. The company’s first generation single-use gastroscope has received regulatory approval from the UKCA and FDA.
Endostart, founded in 2018, is focused on the development and manufacturing of a new generation of GI endoscopy devices. In October 2024, it received CE marking for the expanded use of its flagship product, Endorail, extending the device’s application to enteroscopy, making this challenging procedure more accessible and widely
available, significantly advancing GI endoscopy and improving patient care. Endorail leverages a magnetic balloon system, providing enhanced endoscope control in the small and large bowel. The system uses a magnetically anchored balloon to stabilize and guide the endoscope, reducing looping and improving maneuverability. This innovation simplifies the colonoscopy process and improves enteroscopy by aiding both balloon-assisted and push enteroscopy procedures. Endorail is already available in Italy, UK, and France. With this new CE marking, Endorail offers a solution for enteroscopy, which is critical for diagnosing and treating small bowel conditions, such as cancer, bleeding, and chronic inflammatory diseases.
Founded in 2019, Hayward, CA-based Endiatx is on a mission to reshape the future of medicine through innovative micro-robotics, and simultaneously improve the early detection of cancers, ulcers, and more. Its product, PillBot, is the world’s first low-cost, disposable, remote-controlled, high-quality endoscopy pill. The company’s micro-robot is designed to navigate the human stomach, providing a cost-effective, non-invasive solution for GI diagnosis and treatment. Following the completion of clinical trials, Endiatx anticipates FDA clearance for PillBot in 2025. The commercial launch in the US is expected in early 2026, with plans for international expansion, particularly in developing nations where access to GI medicine is limited. The ongoing clinical trials have shown promising results, demonstrating PillBot’s efficacy and safety in real-world scenarios.
Motus GI, launched out of the New Generation Technology (NGT) incubator in Nazareth, Israel, in 2008, has developed the Pure-Vu System, a medical device that has been cleared by the FDA to help facilitate cleansing of the gastrointestinal tract to improve visualization during an upper GI endoscopy or a colonoscopy. The Pure-Vu System has received a CE Mark in the EU for use in colonoscopy. The system integrates with standard and slim colonoscopes, as well as gastroscopes, to improve visualization during colonoscopy and upper GI procedures while preserving established procedural workflow and techniques. Through irrigation
and evacuation of debris, the Pure-Vu System is designed to provide better-quality exams. The company began market introduction in the U.S. of its fourth-generation system the Pure-Vu EVS, which received FDA clearance in Q4 2023 for use in both the upper and lower GI tract, and is currently targeting early-adopter hospitals.
Pristine Surgical is a medical device company with a mission to simplify endoscopy. The company combines single-use endoscopes with cloudbased software to make minimally invasive visualization more efficient, consistent, and safe. The company’s Summit 4K single-use surgical arthroscope is a fully integrated single-use scope with 4K resolution that simplifies arthroscopic procedures and packs the latest in minimally invasive visualization technology into a sterile, cost-effective, ready-to-use package. Summit 4K was developed to address the challenges inherent to conventional surgical visualization—which relies on reusable arthroscopes that become obsolete, are difficult and costly to maintain and
are prone to wear, damage, and potential infection.
Pristine Surgical offers a subscription business model with transparent pricing and automated inventory management, delivering improved workflow and reduced costs to the endoscopy suite. It is developing a full portfolio of rigid and flexible endoscopes, to address 125 million procedures worldwide each year. In May 2024, Pristine Surgical added medtech industry veteran Christopher Cleary, previously Medtronic’s Senior Vice President of Corporate Development, to its Board of Directors.
Privately held EndoSound, based in Portland, OR, is developing the EndoSound Vision System (EVS), that converts any standard flexible endoscope into an endoscopic ultrasound system (EUS). EUS combines endoscopy (a visual modality using video imaging) and ultrasound to create images of the digestive tract and its surrounding organs and tissues. With these two technologies, the physician can create more detailed images of the digestive
tract, without the need for expensive and complex new scopes. In June 2024, EndoSound was granted a Transitional Pass-Through (TPT) code from CMS, effective July 1, 2024, for the company’s EVS.
Data in this article is partially sourced from the LSI Market Intelligence Platform, including the Global Surgical Procedure Volumes (SPV) database and the Gastrointestinal Endoscopy Devices Market Snapshot (published June 2024).
Looking for more global orthopedics market intelligence and insight?
Access LSI’s continuously updated Market Intelligence Platform.
Scan QR code for details and to request a demo:


1.4M
BPH procedures globally in 2024
8.1% CAGR
BPH implants growth rate
6.8% CAGR
Minimally invasive surgical therapies (MISTs) growth rate
1.9% CAGR
Transurethral resection of the prostate (TURP) growth rate
Driven by innovations in implants, tissue ablation techniques, and other therapies, the minimally invasive BPH treatment landscape is being transformed, with more diverse and personalized options for patients than ever before.

Benign prostatic hyperplasia (BPH), commonly known as an enlarged prostate, is a condition that impacts the lives of more than 500 million men worldwide. By their 60s, over half of men experience symptoms, and by their 70s and 80s, that number skyrockets to 90%, according to the National Institute of Diabetes and Digestive and Kidney Diseases. This progressive, non-cancerous enlargement of the prostate can disrupt daily life with lower urinary tract symptoms (LUTS) like frequent urges, weak flow, and difficulty starting or stopping urination.
While pharmaceutical therapies remain the first line of treatment, only about 0.2% of men with BPH—approximately 1.4 million globally, as tracked by LSI’s Global Surgical Procedure Volumes database—pursue surgical solutions annually. However, the past two decades have seen a revolution in BPH management. Life-changing innovations in minimally invasive surgical techniques, implants, and other advanced therapies are reshaping the treatment landscape, offering patients more targeted, effective, and personalized solutions than ever before.
In this article, we’ll dive into the evolution of BPH management, the driving trends behind these advancements, and the emerging technologies poised to define the future of care for millions of men.
For much of the 20th century, transurethral resection of the prostate (TURP) reigned supreme as the gold standard
Minimally invasive implants are shaking up the BPH market, emerging as a hotbed of innovation and rapid growth. With an aging male population and a growing demand for alternatives to medication and invasive surgery, this segment is thriving. As in many other
for BPH surgery. Introduced in the 1930s, TURP was groundbreaking for its time, but it wasn’t without its challenges. The procedure often came with a heavy price: significant bleeding, risk of infection, potential sexual complications, and long recovery times.
Modern BPH surgery aims to do more than just open the prostatic urethra for better urinary flow and bladder emptying—it’s about doing so with precision, personalization, and minimal disruption to a patient’s quality of life. Today’s minimally invasive techniques eliminate the need for incisions, targeting the prostate through the urethra. Surgical choices depend on factors like prostate size, symptom severity, and the patient’s priority to preserve sexual function. However, larger prostates still pose challenges, with limited options and an increased risk of complications like incontinence and sexual side effects.
While early minimally invasive innovations like prostatic stents from the 1990s have largely fallen out of favor, a new wave of medical technology is redefining BPH treatment. From robotic systems and laser therapies to high-pressure water jets and advanced implants, these cutting-edge solutions focus on minimizing side effects while addressing the root causes of LUTS—and prioritizing sexual function preservation. The future of BPH surgery is as much about innovation as it is about delivering personalized care that truly puts patients first.
areas of medicine, patients are no longer settling for the status quo—they want effective, low-risk solutions that fit seamlessly into their lives.
According to LSI’s analysis, the momentum isn’t slowing down anytime soon. Global sales of these cutting-edge implants are set to skyrocket, with an impressive compound annual growth rate (CAGR) of 8.1%. By 2028,
the market is projected to surpass an impressive $532 million, signaling a new era in BPH management (see figure)
As minimally invasive implants continue to redefine patient care, they’re becoming a cornerstone of the evolving BPH treatment landscape—delivering precision, convenience, and outcomes supported by real-world clinical evidence.
The proliferation of innovative minimally invasive options to provide symptom relief from BPH today, both on the market and under development, are designed to minimize complications and maximize durable patient outcomes using a variety of techniques and materials. The progression of the disease itself often determines the most appropriate intervention, with factors including the size and shape of the prostate, and patient preference. Minimally invasive options include:
• Laser Therapies: Techniques like holmium laser enucleation of the prostate (HoLEP) and GreenLight Laser Vaporization, also known as photoselective vaporization of the prostate (PVP), offer precise tissue removal with reduced bleeding and faster recovery times. PVP is performed under anesthesia by placing a telescope into the urethra up to the level of the prostate. The laser fiber is guided through the scope and the prostate tissue is then quickly and gently vaporized.
• Prostatic Urethral Lift (e.g., the UroLift System from Teleflex):
An implant placed through the urethra via a minimally invasive, reversible procedure that mechanically pulls the prostatic lobe away from the urethra without removing tissue. This procedure, which dominates the global BPH implant market, is ideal for smaller prostates.
• Additional Implants: Developers in this innovative space include Olympus/ Medi-Tate (iTind procedure, temporary implantable nitinol device), Butterfly Medical (Butterfly Medical in-office implantable
device), ProVerum (ProVee nitinol implant), ProArc Medical (Omega biocompatible arc-shaped implant), and ZenFlow (Spring System coil that gently props open the urethra).
• Convective Water Vapor Energy Therapy (Rezum from Boston Scientific): Uses water vapor thermal therapy (steam) to ablate excess prostate tissue, aimed at preserving surrounding structures and sexual function.
• Aquablation:
The flagship approach of PROCEPT BioRobotics, the therapy employs high-pressure water jets guided by robotic systems to precisely remove tissue, offering a balance of precision and minimal invasiveness. Research has been promising for this new treatment, with significant improvement in urinary symptoms and good durability with low retreatment rates of less than 3% at the five-year mark. Aquablation has also been shown to be successful in patients with acute urinary retention who are catheter dependent. After the procedure, many of these patients are catheter-free and able to void completely on their own.
• Mechanical Dilation: The Optilume BPH Catheter System from Optilume, FDA PMA-approved in 2023, combines mechanical dilation with concurrent localized delivery of paclitaxel for treating BPH. Mechanical dilation with proprietary double-lobe balloon technology achieves an anterior commissurotomy (split), releasing the constricting lateral lobes, while the delivery of paclitaxel prevents re-fusion of the lobes and maintains patency during healing.
• Transurethral Microwave Thermotherapy (TUMT): An outpatient procedure that is less invasive compared to surgery and involves using microwaves (heat) to shrink prostate tissue that is enlarged.
• Prostate Artery Embolization (PAE): A newer treatment option for BPH, PAE involves a percutaneous endovascular approach, with selective embolization of each prostatic artery with microparticles, resulting in a gradual decrease in prostatic volume, leading to a decrease in LUTS.
• Robotic-Assisted Prostatectomy: Provides enhanced precision for larger prostates or complex cases, including clinically localized prostate cancer.
By 2023, Teleflex’s UroLift reigned supreme in the global BPH implant market, commanding an impressive 90% market share (see figure). With a decade of clinical evidence and over 145 peer-reviewed publications to its name, UroLift has set the gold standard in minimally invasive BPH treatments. Backed by robust product development and expansion strategies, Teleflex is making it clear: they’re here to stay at the forefront of this competitive landscape.
Meanwhile, Olympus/Medi-Tate’s iTind is carving out its own niche with a solid 10% share as of 2023, according to LSI Market Intelligence. This innovative outpatient or office-based procedure uses a temporary nitinol implant to reshape the prostatic urethra—no burning, no cutting. Upon removal of the implant after five to seven days at home, patients have reported rapid,
Innovators in the high-stakes minimally invasive BPH implant space have recently achieved critical milestones in their efforts to provide patients with the latest generation of devices.
Teleflex’s UroLift System, introduced in 2013 by Neotract (acquired by Teleflex in October 2017 for an upfront cash payment of $725 million and $375 million in additional milestone payments), is a minimally invasive treatment option for BPH that can help men get off BPH medications and avoid major surgery. It is the only leading enlarged prostate procedure that does not require heating, cutting or destruction of prostate tissue, according to the company. The prostatic urethral lift procedure involves the insertion of one or more permanent implants into the prostate, which retract prostatic tissue and maintain an expanded urethral lumen, hence increasing the size of the urethral opening and reducing obstruction to urine flow. According to the company, as of December 2024, 450,000 men have
effective symptom relief. In March 2024, Olympus scored a significant win, securing broader U.S. patient access for iTind through major group purchasing organization (GPO) contracts, signaling its ambition to challenge the competition.
This innovative field is continuing to heat up. New players poised to enter the BPH implant arena during the forecast period are set to challenge UroLift’s dominance, potentially reshaping the market dynamics. The race for patient-centered innovation in BPH
treat-ment is just getting started.
Source: LSI’s Benign Prostatic Hypertrophy Implants Market Snapshot, published July 2024
been treated with the UroLift System in select markets worldwide. The procedure is also recommended for the treatment of BPH in both the 2021 American Urological Association (AUA) and 2022 European Association of Urology clinical guidelines.
The company presented new data at the April 2024 European Association of Urology Congress showing that men with LUTS from BPH had better outcomes with the implant than with a commonly prescribed drug. Then in May 2024 at the AUA annual meeting, Teleflex presented findings from two randomized head-to-head trials, showcasing excellent patient experience with the UroLift System for BPH. The company also introduced the UroLift 2 System with Advanced Tissue Control (ATC), which has recently received FDA clearance. The UroLift 2 ATC System offers physicians enhanced confidence, easier control of obstructive tissue, and targeting accuracy through tissue control wings and laser-etched needle markers designed to make tissue manipulation and implant delivery more precise. A streamlined delivery system, utilizing typically one handle
per procedure and individual implant cartridges, promotes increased physician comfort and improved efficiency during the procedure.
South San Francisco, CA-based device company ZenFlow has developed the Spring System, a minimally invasive treatment option for patients who suffer debilitating symptoms due to enlarged prostate. ZenFlow’s Spring technology was designed with the patient experience in mind, and relies on a small spring-like coil that gently props open the urethra, restoring its normal function while preserving the natural anatomy. And, its approach has investor support.
In November 2024, ZenFlow closed a $24 million Series C financing round, including new investor Cook Medical, as well as existing investors Invus Opportunities, F-Prime Capital, Medical Technology Venture Partners and others. Wilson Sonsini Goodrich & Rosati advised Invus, F-Prime, and MedTech Venture Partners on the transaction. Funds will be used to support the company’s PMA submission and to prepare for commercialization upon FDA approval of the Spring System.
Source: LSI Market Intelligence

1,400,000
BPH procedures will be performed worldwide in 2024. More than 90 million men worldwide are living with benignprostatic hyperplasia (BPH).
According to LSI’s Global Surgical Procedure Volumes (SPV) database, 1.4 million BPH procedures are projected to be performed globally in 2024 (see page 42 in this issue for our full Urological Procedure Volumes forecast for 2023-2030). Of these, TURP still accounts for the majority—approximately 85% of all procedures.
While TURP remains dominant, MISTs are gaining ground. These less-invasive approaches are projected to grow at a faster pace, with implants like UroLift expected to increase at a CAGR of 6.8% from 2023 to 2028, compared to TURP’s slower growth rate of 1.9%.
Despite the rise of MISTs, TURP has stayed on top in BPH treatment for several reasons:
• Proven track record: Decades of clinical data validate TURP’s safety, efficacy, and durability.
• Rapid symptom relief: Patients experience immediate and lasting improvements in LUTS.
• Accessibility: TURP is widely available and familiar to most urologists, with a lower learning curve than newer techniques like HoLEP.
• Efficiency: TURP offers shorter operative times compared to complex procedures like robotic prostatectomy or laser enucleation.
We expect to see new techniques and implants continue to challenge TURP and aggressively chip away at its dominance, particularly in markets where patients and physicians prioritize minimally invasive options with faster recovery times.
Key trends we're watching in the future of the BPH treatment landscape:
• Advances in imaging and robotics: Enhanced precision and targeting will improve outcomes across all procedure types.
• Durable, minimally invasive options:
A wide variety of implants and techniques are expected to gain significant traction, offering a balance of safety, effectiveness, and convenience.
• Better patient selection and personalized treatment: Improved diagnostics will help tailor treatments to individual needs, ensuring optimal outcomes.
Ultimately, maintaining a diverse array of treatment options for BPH patients will be critical. From long-established procedures like TURP to cutting-edge approaches like Aquablation, the goal remains the same: improving quality of life for millions of men worldwide.
Data in this article is partially sourced from the LSI Market Intelligence Platform, including the Global Surgical Procedure Volumes (SPV) database, and LSI’s The Numbers e-newsletter - market data and insights from LSI, every Saturday on LinkedIn, November 23, 2024.
Looking for more global urology market intelligence and insight? Access LSI’s continuously updated Market Intelligence Platform.

Data sourced from LSI’s Global Procedure Volumes Database, which provides trusted coverage of 300+ diagnostic and therapeutic procedures across 37 countries. Scan QR code to request a demo!

Celebrating the recent wins of our LSI presenting company and speaker alumni community.

Altra has engineered a novel, dual-chamber infusion pump, inspired by the human heart, to address adverse events and challenges associated with drug delivery. The Altra Infusion System incorporates numerous advancements designed to enhance patient safety and improve infusion reliability, including state-of-the-art air-in-line detection, and an occlusion detection system which provides timely alerts and reduces risks during therapy.
The Redmond, OR-based company recently announced that its Altra Infusion System has been accepted into the FDA’s Safer Technologies Program (STeP). Altra’s inclusion in this program will play a critical role as the company prepares its 510(k) submission to secure market clearance.
FDA’s collaborative STeP program, launched in 2021, is designed to streamline the regulatory review process for medical devices that significantly
enhance patient safety, but that target an underlying disease or condition associated with morbidities and mortalities less serious than those eligible for the Breakthrough Devices Program. The program provides select companies with the opportunity to collaborate closely with the agency, offering real-time feedback, interactive communication, and customized regulatory guidance.
San Mateo, CA-based Cala Health is a bioelectronic medicine company transforming the standard of care for

Deanna Harshbarger, CEO, Cala Health (Source: LSI USA '24)
chronic disease. The company's wearable neuromodulation therapies merge innovations in neuroscience and technology to deliver individualized peripheral nerve stimulation, and its direct-tohome digital durable medical equipment platform is reshaping the delivery of prescription therapies. Cala Health's products are the only FDA-cleared, clinically validated noninvasive devices for the relief of hand tremors. In December, the company completed the initial closing of an oversubscribed $50 million growth round, co-led by Vertex Growth Fund and Nexus NeuroTech Ventures. All existing Cala investors participated in the round, including Action Potential Venture Capital, Johnson & Johnson (through its corporate venture capital organization, Johnson & Johnson Innovation – JJDC, Inc.), Lightstone Ventures, Lux Capital, GV (Google Ventures), OSF Ventures, Ascension Ventures, TriVentures, Reimagined Ventures, Peak6 and Fiscus Cala is continuing to build strong clinical evidence to expand patient
access for its neurostimulation therapy. The results of a study by CVS Health, presented at the Movement Disorders Congress this year, concluded that TAPS therapy offers substantial benefits for patients with essential tremor (ET) in reducing ET-related healthcare resource utilization, healthcare costs, and improving clinical outcomes.
The Cala kIQ System with TAPS (Transcutaneous Afferent Patterned Stimulation) Therapy is the only non-invasive, FDA-cleared, wearable device that delivers effective therapy for action hand tremor relief in people with essential tremor and Parkinson's disease. The TAPS device is clinician-prescribed and clinically proven to safely and effectively reduce action hand tremors. The Cala kIQ device treats tremor by sensing each patient's unique tremor signature and delivering individualized stimulation to the nerves on the wrist that then reach the brain to counteract tremor.
ET impacts over 7 million people in the U.S. and is characterized by uncontrollable movement that can make critical daily activities very difficult. Drugs are most used to treat ET, but they can cause a range of unwanted side effects such as fatigue, nausea and depression and may not provide desired outcomes. Surgical options like deep brain stimulation or focused ultrasound ablation can be effective but are invasive procedures that carry the risk of potentially irreversible side effects.
California-based Capstan Medical is creating a new standard for minimally invasive heart valve intervention, with a

The company recently closed an oversubscribed $110 million Series C round led by Eclipse. Existing investors Intuitive Ventures, Yu Galaxy, and new investor Gideon Strategic Partners, participated in the round. Last year, Capstan closed an oversubscribed $31.4 million Series B funding round, which Eclipse also led.
While many cardiovascular treatments have improved and become less invasive, traditional heart valve surgery remains highly invasive and is associated with lengthy recovery times. Given the risks associated with open-heart surgeries, many patients delay treatment, unwittingly increasing their health risks as well as the chances they may be too weak later on for open heart surgery. Capstan's minimally invasive solution seeks to overcome limitations of current treatment options by bringing together novel heart valve implants and advanced catheter technology, fully enabled through a robotic platform, to treat a broader set of patients.
Centerline Biomedical, a cardiovascular navigation and visualization systems innovator, has received FDA 510(k)

live x-ray, to see medical devices inside the body. Using fluoroscopic imaging exposes the treating clinicians and staff to x-ray radiation, which aggregated over time may introduce risks for DNA damage and other medical complications. IOPS was designed without the need for prolonged fluoroscopy during endovascular procedures to promote the safety of clinicians and patients. This is achieved through advancements in both device navigation and visualization.
IOPS advancements in visualization, including the novel Spintegration feature, enables multicolor 3D visualization of the arteries and their branches before, during, and after stent graft placement. The result is unparalleled vascular mapping with colorful, 3D images displaying the fine features of diseased arteries; including calcifications, occlusions, dissections, and other anatomical formations not as visible when using traditional 2D grayscale x-ray fluoroscopy. In addition, new software features, only available on IOPS by Centerline, add the capability to create and display 3D wireframe vessel models for procedure mapping.
Founded in 2015 and headquartered in Cleveland, OH, Centerline Biomedical’s commercially available platform, IOPS, has 510(k) clearance from the US FDA for use in the descending aorta.
Distalmotion’s mission is to empower robotic surgery excellence across all sites of care, including hospital outpa-
suite of next-generation implants and a first-of-its-kind, catheter-based, robotic delivery platform. Established in 2020, Capstan Medical came out of the Santa Cruz incubator, Occam Labs.
clearance for its new IOPS Guidewire Handle. This second-generation device is the most recent addition to the company’s novel, patented IOPS (Intra-Operative Positioning System) technology portfolio. The IOPS Guidewire Handle is used with the sensorized IOPS Guidewire and new Viewpoint Catheter to support 3D navigation of a patient’s vasculature. When combined with the proprietary algorithms of the IOPS software, clinicians are able to clearly visualize these devices in real-time and with minimized dependence on fluoroscopy during procedures.
During endovascular procedures, clinicians typically rely on fluoroscopy, or

tient and ambulatory surgery centers, by expanding access and improving care with robotic-assisted surgery. The Dexter Surgical Robot is designed to address the growing demand for cost-effective and efficient solutions for high-volume procedures in these settings. Its mobile design, small form factor, and open platform design make it particularly well-
suited to outpatient environments, enabling hospitals and ASCs to optimize resources while still accessing the latest technologies and best-in-class surgical tools. Distalmotion recently announced that its Dexter Surgical Robot has successfully treated the first U.S. patients in an outpatient setting. Two adult inguinal hernia repairs were completed with Dexter on November 26, 2024, less than 30 days after receiving FDA de novo approval for its Dexter surgical robot for this indication. Dexter seamlessly integrates into existing operating workflows and is fully compatible with current operating room equipment, protecting hospitals’ existing investments. Additionally, its single-use instruments remove the complexities of reprocessing, further enhancing workflow efficiency. The system can work natively with third-party 3D imaging systems, energy devices, vessel sealers and other laparoscopic devices. It features a suite of fully wristed, single-use instruments. These deliver dexterity, precision of movement, reliable instrument performance and lower reprocessing requirements.
Unique to Dexter, the surgeon remains sterile at the console, enabling direct and quick access to the patient at any point during the procedure. This empowers surgeons to flexibly choose the best technique for each step of the procedure. Dexter is designed to be the “surgeon’s robot” that provides the physician complete control of the procedure to optimize patient outcomes.
The company raised $150 million last year to progress its regulatory efforts in the U.S., and reports more than 1,400 patients successfully treated in Europe.
Houston, TX-based EndoQuest Robotics designed its flexible endoluminal robotic technologies to address unmet needs in gastrointestinal and other endoluminal surgeries. The proprietary technology navigates and performs procedures within the body’s lumina spaces, via a trans-oral or trans-anal approach, allowing for minimally invasive interventions, while offering precision and flexibility to improve patient outcomes.
The company recently announced it received FDA investigational device exemption (IDE) for its pivotal colorectal clinical study, Prospective Assessment of a Robotic-Assisted Device in
Gastrointestinal Medicine (PARADIGM). The landmark multi-center, open-label study will examine the Endoluminal Surgical (ELS) System’s safety and performance in robotic endoscopic submucosal dissection (ESD) procedures for the removal of colorectal lesions. EndoQuest expects 50 subjects to participate in the study. It will take place across five leading U.S. healthcare institutions: Brigham and Women’s Hospital (Boston), Mayo Clinic (Scottsdale), Cleveland Clinic (Cleveland), AdventHealth (Orlando), and HCA Healthcare (Houston).
The ELS System is designed to solve the technical challenges and decrease the learning curve associated with complex endoluminal procedures such as ESD. The system enables endoluminal procedures with the visualization, precision, and main tenets of conventional surgical technique, allowing for surgical tasks and maneuvers not currently possible in advanced endoscopy. A pre-clinical ESD study presented at Digestive Disease Week in 2024 demonstrated that the ELS System had superior procedural outcomes, a shorter learning curve, and reduced procedure time versus the conventional technique.
The company, which is currently led by interim CEO Eduardo Fonseca, raised $42 million to support the platform a year ago.
Fluid Biomed is a clinical-stage medical device start-up founded by two neurosurgeons, Dr. John Wong and Dr. Alim

Mitha, at the University of Calgary. The pair has developed a novel polymer-based stent, ReSolv, that can heal a blood vessel by diverting blood flow away from a weak brain aneurysm. Working together for the past two decades to help people with stroke, their drive and commitment to improve patient health sparked a vision
to create a novel stent technology to cure a wide variety of aneurysms with greater efficiency and safety.
The company recently completed a $27 million oversubscribed Series A equity financing round to advance the development of its device. The round was co-led by Amplitude Ventures and an undisclosed major strategic partner. New participants in the investment syndicate include IAG Capital Partners and LifeArc Ventures, as well as returning investors, ShangBay Capital and METIS Innovative. Fluid Biomed plans to use the proceeds to further validate the ReSolv stent and its proprietary delivery system in expanded patient studies.
Fluid Biomed’s bioabsorbable, polymer-based ReSolv stent has been validated by years of scientific research with functional benefits demonstrated in a first-in-human clinical study to treat brain aneurysms. The self-expanding stent is engineered to be highly flexible to navigate into delicate blood vessels of the brain, and provides a scaffold for endothelial cells to promote blood vessel healing. After the aneurysm is cured, ReSolv is gradually integrated into the vessel wall, leaving very little footprint behind. Because the polymer is invisible to x-rays and eventually dissolves away, there is minimal interference with follow-up CT and MRI scans, so patients are not required to undergo invasive angiogram tests or take life-long blood thinners.
About 6.7 million people in the U.S. have unruptured brain aneurysms and more than 30,000 rupture and bleed each year, often leading to disability and death.
InkSpace Imaging’s vision is to transform the magnetic resonance imaging (MRI) experience for healthcare professionals, radiologists, MRI technologists,

and patients by providing superior comfort and performance at a lower cost. Its lightweight, ultra-flexible MRI coils are designed with groundbreaking versatility, utilizing advanced printed electronics to conform effortlessly to each patient’s unique anatomy. This innovative approach not only enhances patient comfort but also optimizes signal quality, resulting in sharper imaging and more efficient scan times, ultimately improving the diagnostic experience for both clinicians and patients.
The company, based in Pleasanton, CA, recently announced that its Small Body Array has achieved FDA clearance for use with the Siemens Healthineers Magnetom 1.5T platform. This milestone highlights the robust capabilities of InkSpace Imaging's technology and underscores its potential to expand accessibility and efficiency in the broader 1.5T MRI market. By addressing a key gap in patient comfort and diagnostic precision, this clearance positions the company to accelerate its commercialization efforts and make a meaningful impact in clinical imaging.
Founded in 2020, Lauxera Capital Partners is an independent asset management company and healthcare spe-

cialist investor based in Paris, France and San Francisco, CA, that is working to promote the growth of European healthtech companies.
The company announced the first close of Lauxera Growth II at €300 million ($315 million), representing 75% of the target and already exceeding the size of its first fund. In February 2022, Its first fund, Lauxera Growth I, gathered €262 million ($275 million) of capital from investors and had invested in 12 fast-growing healthtech businesses
in France, the UK, Germany, Sweden, Denmark, and the U.S.
Lauxera Capital Partners also announced the acquisition of Galen Data, a leading provider of secure, cloudbased connectivity solutions for medical devices. This transaction follows Lauxera’s 2022 acquisition of Matrix Requirements, a leading application lifecycle management and electronic quality management software solution for medical device R&D and Quality teams.
Berlin, Germany-based Lindera, founded in 2017, has developed AI-powered mobility analysis tools utilizing smart-

phone cameras to perform 3D motion tracking and assess fall risk, primarily aiding elderly individuals.
The Lindera Mobility Analysis app allows individuals to determine their risk of falling, easily and precisely. For the first time, a 3D analysis of gait movement is possible using a simple smartphone camera. The precise 3D motion tracking accurately calculates step patterns as well as body inclanation – even with the use of walking aids. Using a short video recording and a questionnaire, patented algorithms determine the risk of falling, and provide personalized recommendations for preventing falls and maintaining mobility in old age. System integration allows results and suggested measures to be automatically transferred to care documentation software. The repeated use of the app is associated with significant decreases in fall risk. Study results show a fall risk reduction of 18% after the second analysis and an overall improvement of the clinically relevant gait parameters.
The company has published a new groundbreaking study in the journal JMIR Aging, validating its AI-driven fall
risk prediction system, based on realworld data from over 600 seniors. The retrospective cohort study evaluated the prognostic validity of the Fall Risk Score (FRS), a multifactorial fall risk scale generated by the mHealth app Lindera Mobility Analysis. The study included 617 older adults (857 observations) residing in German nursing facilities. The FRS was assessed at the initial evaluation and correlated with fall frequency during follow-up evaluation. The study concluded that FRS exhibits good prognostic validity for predicting future falls, particularly in specific subgroups. The findings support a stratified fall risk assessment approach and emphasize the significance of early and personalized intervention.
Lungpacer Medical is a neurostimulation company dedicated to developing innovative interventional therapies

to free patients from mechanical ventilation and improve diaphragm, lung, heart, and brain health.
In early December, FDA approved Lungpacer Medical’s AeroPace System, a first-of-a-kind temporary diaphragm activation system placed with the AeroPace Catheter, indicated to improve ventilator weaning success in patients 18 years or older. The AeroPace System previously received a Breakthrough Device designation
Muscles necessary for breathing, such as the diaphragm, may be inactive while a patient is mechanically ventilated. As a result, a type of muscle atrophy, known as ventilator-induced diaphragm dysfunction, may occur. The AeroPace System uses neurostimulation via an electrode-containing, cardiovascular-catheter and software-controlled system to contract and strengthen the diaphragm in patients on mechanical ventilation. The
system’s FDA-approved indication is to improve weaning success – increase weaning, reduce ventilator days, and reduce reintubation – in patients ages 18 years or older on mechanical ventilation ≥ 96 hours and who have not weaned.
A growing body of research including more than 100 scientific presentations, 30 peer-reviewed publications, and clinical studies in over 400 patients at 50 international centers, is building evidence and data to demonstrate the value of Lungpacer’s AeroPace System and the investigational AeroNova System, currently in clinical trials. Together, these therapies can address approximately 2.5 million mechanically ventilated critical care patients in the U.S.
Founded in 2014, MedLumics is developing fiberoptically guided platform technologies, with an initial focus

on the treatment of atrial fibrillation (AF). MedLumics’ product, the AblaView Optically Guided Pulsed Field Ablation (PFA) System, is a catheter-based, pulsed field ablation (PFA) technology that optically guides catheter positioning and ablation treatment strategy using optical coherence reflectometry (OCR). AblaView proprietary multibeam technology provides real-time, direct visual confirmation of catheter contact, contact stability, and predicts lesion durability during tissue ablation, with the potential to reduce recurrence rates and reduce procedure technical complexity while increasing procedural safety. Although AF patients are expected to benefit most from this innovation, AblaView is a platform technology with multiple therapeutic applications using OCR and PF ablation.
The company recently announced completion of its first-in-man study, with successful remapping at three months, demonstrating the efficacy of its AblaView system.
AF is the most common type of heart arrhythmia. An estimated 100 million people worldwide currently suffer from AF, and prevalence of the disease is expected to increase as the number of people over the age of 65 continues to expand, and the greatest risk factor is age. Although many technologies have emerged over the last several years to improve the catheter ablation treatment of AF, the ablation procedure has been elevated to first-line therapy in European and North American clinical guidelines, but outcomes remain difficult to predict. Broad registries reveal recurrence rates of up to 60% at the one-year follow-up time point.
Nervonik is a CA-based medical device startup focusing on peripheral nerve stimulation (PNS) for the treatment of chronic pain—a rapidly growing therapy space within the multibillion-dollar chronic pain market that involves applying highly controlled electrical currents to nerves in the body, which modulate pain signals and provide relief.
The company announced that it has successfully completed a first-in-human clinical study, using its proprietary Nervonik PNS device. The implantable device utilizes proprietary technology, originally developed in Dr. Aydin Babakhani’s laboratory at the University of California, Los Angeles, to enhance therapy outcomes. The implants rely on next-generation, neural-sensing integrated circuits to deliver an optimized therapy with minimal therapeutic resistance. The implants’ small size and PNS targeting offer new hope for managing intractable discomfort and suffering, and represents a new, long-term, narcotic-free solution for chronic pain.
Headquartered in Galway, Ireland, Neurent Medical develops innovative non-surgical treatments for chronic inflammatory sino-nasal diseases. By targeting and safely disrupting the hyperactive autonomic nerves that drive underlying inflammation, their revolutionary treatment provides effective relief to patients. Neurent has its U.S. office in Boston, MA.
The company recently announced that it has secured €9 million ($9.3

million) in growth capital from the Claret European Growth Capital Fund III (CEGCF III), as part of a larger fundraising to further develop its product portfolio and expand its presence in the U.S. market.
Neurent closed the €18.2 million ($18.9 million) deal with equity investors, which included a drawdown of €3 million ($3.1 million) from the Claret Capital facility. It was one of the biggest Irish venture funding deals of 2024.
Approximately one in four Americans suffer from chronic rhinitis, a common condition that results in persistent symptoms including congestion, rhinorrhea, sneezing and nasal itching. Chronic rhinitis significantly decreases quality of life and can have an adverse effect on sleep quality, mood, and daily activities.
The traditional rhinitis patient journey is often full of frustration due to limited treatment options. The NEUROMARK System is a groundbreaking, minimally invasive treatment option designed to target the underlying drivers of chronic rhinitis. The NEUROMARK System enables the physician to simultaneously treat multiple nerve branches with a high degree of precision, safety, and patient comfort.
Barcelona, Spain-based Neuroelectrics is a pioneer in high-definition brain stimulation and EEG technology.

Its products are being used worldwide for both neuroscience and clinical research in hundreds of universities and academic medical centers. The company’s vision is to create a powerful neurotechnology platform upon which verticals can be developed to improve the lives of as many people as possible. Clinical applications are being developed to diagnose and treat brain diseases as well as improve brain health (e.g., in epilepsy, neuropathic pain or mood disorders) and cognitive function (e.g., memory in dementia or executive function in children with ADHD).
The company recently announced a partnership with Neuronostics, an innovator in advanced digital biomarkers and electroencephalography (EEG) analysis, to deliver an integrated EEG solution tailored for pharmaceutical companies, contract research organizations (CROs), and academic partners. This collaboration combines Neuroelectrics’ cutting-edge EEG hardware with Neuronostics’ proprietary analytics to accelerate clinical trials with advanced EEG solutions, providing pharmaceutical and device companies with tools to optimize participant selection, protocol design, and data analysis, and unlock new possibilities in neurological research. The collaboration addresses a critical need in clinical trials for high-quality, actionable data, by providing an integrated platform that accelerates research and improves outcomes.
Woburn, MA-based OSSIO is a device company focused on evolving orthopedic surgery with bio-integrative fixation technology. Founded in 2014, the company’s vision is to provide the first credible replacement for metal implants in the multibillion-dollar global orthopedic fixation market, with its OSSIOfiber Intelligent Bone Regeneration Technology. OSSIOfiber implants provide strong fixation while leaving nothing permanent behind, which avoids the risk of common post-surgical complications and secondary procedures associated with metal and similar materials. After serving their fixation function, they fully integrate and are replaced with the patient’s natural bone.
The company recently closed on $27.6 million in equity funding to rapidly scale
all aspects of its commercial efforts, including a new product pipeline, sales channel and medical education, as well as building out a flagship U.S. manufacturing site and surgeon training center in Florida. Questa Capital, which focuses on investments in expansion-stage healthcare companies, led this latest round of financing, with participation from existing investors MVM Partners, OCV, Ortho-Vision, and Courage Capital
Since the company’s first product launch in 2019, upwards of 50,000 OSSIOfiber implants have been used across a broad spectrum of orthopedic procedures. The company presented at LSI’s USA ‘20 event.
PhotoniCare has secured $4.6 million in Series B financing for its first-inclass, FDA-cleared OtoSight Middle

Ear Scope. The new funding is the first close in an expected $9 million round.
The company also formed a manufacturing partnership with Gentex Corporation for its OtoSignt Middle Ear Scope, and fiinalized the relocation of its headquarters from Champaign, Illinois to Grand Rapids, Michigan. Founded in 2013, PhotoniCare’s mission is to improve clinical outcomes and serve unmet patient and provider needs by translating novel light-based technologies into objective medical practice, starting with the OtoSight Middle Ear Scope. The FDA-cleared device was developed for healthcare providers seeking a superior solution for pediatric middle ear issues. With remarkable 90+% reader accuracy in determining the presence or absence of fluid in the middle ear, the device eliminates subjectivity and speculation by providing patients with a non-invasive and comprehensive assessment of middle ear fluid, even in the presence of significant earwax.
Middle ear infections are the leading cause of hearing loss, surgery and antibiotic use, especially in children. Affecting more than 80% of children, they are misdiagnosed up to 50% of the time using the current gold standard, examination via otoscope. Children can suffer from recurring ear infections for six to 12 months before they are referred to an ENT specialist, and are often prescribed increasingly potent antibiotics during this time. Often, the endpoint of the disease is insertion of tympanostomy tubes in the eardrum. More than 1 million tube surgeries are performed in the U.S. alone every year.
Lowell, MA-based RevBio is a clinical-stage device company developing and commercializing TETRANITE, a

patented, synthetic, injectable, self-setting, and osteoconductive adhesive biomaterial. This novel technology will be indicated for use in dental, cranial, and broader orthopedic applications as well as animal health.
The company recently announced it has received FDA approval to expand its ongoing clinical trial to immediately fixate cranial flaps using TETRANITE. RevBio also received reimbursement coverage from CMS for the use of TETRANITE to replace metal plates and screws.
Previously, RevBio received FDA approval to initiate a first-in-human clinical study for an initial five patients to restore cranial flaps following craniotomy procedures and to repair extradural use cases where intentional durotomies are not required. The approval of this clinical trial expansion is a result of the successful demonstration of safety in the first five patients treated with TETRANITE. The neurosurgeon investigators involved in this study will now be able to use TETRANITE in intradural use
cases, such as tumor resection surgeries and other cranial procedures, which require intentional durotomies.
Metal plates and screws can produce radiographic artifacts which make it difficult to accurately interpret patient imaging following surgery. What’s more, traditional cranial flap closure methods do not lead to the full osseous reintegration of the flap with the skull, resulting in issues with flap movement, patient pain, and the potential for post-surgical infection. Because TETRANITE eliminates radiographic artifacts and results in osseous union of bone flaps with the surrounding skull, it could become the new standard of care for cranial surgery, according to company representatives.
Roivios and its wholly owned R&D subsidiary, 3ive Labs, are clinical-stage medical device companies striving to

sustain or enhance kidney function during times of acute stress. The company is initially focused on providing a first-to-market solution to sustain or enhance kidney function for patients with impaired kidney function during a cardiac surgery with cardiopulmonary bypass (CPB).
The company recently received FDA investigational device exemption (IDE) approval to conduct a pivotal trial for its renal assist device (RAD), the JuxtaFlow RAD, for treating cardiac surgery patients with renal insufficiency. This IDE follows FDA breakthrough device designation for JuxtaFlow in April. The GRADIENT trial aims to address the need for renal support among patients on CPB. The study will be a prospective, multicenter, randomized, controlled, open-label trial. Roivios plans to look at JuxtaFlow’s safety and effectiveness in sustaining or enhancing renal function during and following CPB surgery. The
trial marks a critical step in validating the safety and effectiveness of its innovative renal therapy designed to enhance outcomes for patients with kidney disease.
Bahamas-based Roivios designed JuxtaFlow to improve treatments for patients facing kidney disease during cardiac surgery. JuxtaFlow, which combines a catheter and pump system, uses a unique, gentle negative pressure technique on the kidneys’ urine-collecting system. It enhances function and shields against hypoxia-induced damage under acute conditions.
The company believes the technology can reduce ICU stays, minimize the need for emergency dialysis, and enable savings for healthcare providers. Roivios expects a U.S. launch in late 2025.
Panakes Partners' portfolio company SamanTree Medical SA has announced promising results from the SHIELD study, presented by researchers on December 12 at the 2024 San Antonio Breast Cancer Symposium. The findings show that the company’s flagship product, the Histolog Scanner, a confocal laser microscope, reduced reoperation rates during breast-conserving surgery (BCS) from 30% to 10%—representing a 67% reduction. The innovative imaging device also demonstrated significant accuracy in detecting positive margins with high sensitivity and specificity.
The prospective study, conducted at St. Vincenz Hospital in Paderborn, Germany, evaluated the impact of the Histolog Scanner on reducing reoperation rates during BCS. Fifty patients with invasive breast cancer and/or ductal carcinoma in situ (DCIS) were enrolled, and reoperation rates were compared to historical data from the POLARHIS study, a retrospective observational study performed at the same breast center by the same surgical team using standard intraoperative margin assessment techniques. The Histolog Scanner outperformed standard techniques by achieving a sensitivity of 80.9% and a specificity of 99.5% for positive margin detection, compared to the combined sensitivity of 17.4% and specificity of 97.3% achieved
with standard methods, as seen in the SHIELD study.
SamanTree Medical’s Histolog Scanner features massive parallel confocal microscopy, a cutting-edge technology that expands the imaging field up to 40,000 times larger than standard confocal microscopy. The breakthrough device enables surgeons and pathologists to visualize large, fresh tissue samples in real-time with exceptional resolution and accuracy, making it uniquely suited for intraoperative environments in oncologic surgery.
Headquartered in Liège, Belgium, with U.S. regional headquarters in Boston, SamanTree Medical SA is dedicated to improving oncologic surgery through innovative imaging solutions. Founded in 2010 based on innovations from the École Polytechnique Fédérale de Lausanne (EPFL), a leading Swiss research institute renowned for engineering and technological advancements, SamanTree Medical is committed to improving surgical precision and outcomes by enabling realtime fresh tissue imaging.
Sensome, developer of microsensing technology for instant intra-operative tissue analysis, announced

that results from two studies of its FDA Breakthrough Device-designated Clotild smart guidewire system demonstrated the device’s ability to identify “fresh” clot—thrombus rich in red blood cells (RBCs)—in peripheral artery disease (PAD) and differentiate it from other tissue encountered during PAD procedures.
Results from the SEPARATE and E-SEPARATE studies were presented in a late-breaking trial session at the Paris Vascular Insights Course 2024, held December 12-14 in Paris, France.
According to the company, the prospective, single-arm SEPARATE study was composed of 17 patients who were treated at AZ Sint Blasius Hospital in Dendermonde, Belgium. In a postprocedure analysis, the study showed that there was a high level of agreement between the technology’s identification of fresh clot, the expert’s assessment of fresh clot, and the treatment decisions appropriate for fresh clot.
The E-SEPARATE study demonstrated the technology’s ability to differentiate fresh clot from other tissue collected from these PAD patients and examined ex vivo. It also demonstrated the correlation between the technology’s ability to determine the RBC content of clots collected from PAD patients with subacute and chronic lesions and a histological analysis of the same clot by an outside core lab, according to the company.
The investigational Clotild device is based on electrical impedance spectroscopy, which measures the electrophysiologic characteristics of fluid or tissue in the 360 degrees surrounding the sensor, and analyzed by Sensome’s predictive models. Sensome has miniaturized the technology to fit on the distal part of a standard 0.014-inch guidewire, directly behind a soft, atraumatic tip. The company stated that the technology can be integrated into devices commonly used during PAD procedures, such as guidewires and catheters, without changing current workflow.
SpinaFX Medical is a Toronto, Ontario, Canada-based device company working to bring innovative, minimally invasive, image-guided treatments to patients with back pain due to contained herniated discs.

The company recently announced the successful completion of its Series B funding round, raising its last tranche of CAD $5.3 million ($3.7 million), led by an unnamed strategic investor. The financing will support the company’s continued efforts to secure regulatory approvals, drive commercialization, and expand innovations with its next-generation solution as the company seeks to advance spinal care.
After failed conservative treatments for back pain, surgery is often the only treatment option. SpinaFX is addressing this therapeutic gap with a minimally invasive, image-guided therapy for contained herniated discs, called Triojection, an investigational therapy, which delivers an ozone-oxygen mixture into the disc. Preliminary data from studies indicates the potential of this outpatient procedure to address disc volume and related nerve pressure, pending further clinical validation and regulatory approval.
Galway, Ireland-based medtech company SymPhysis Medical has developed releaze, a minimally invasive home-use drainage device that offers a novel, and patient-empowering approach to managing malignant pleural effusion, which occurs as fluid accumulates in the chest due to cancer. The condition can cause severe discomfort, pain, and shortness of breath, often requiring frequent hospital visits. The device is manufactured in Letterkenny, Co. Donegal, by Phillips Medisize.
The company recently secured €2.2 million ($2.3 million) in funding from a combination of investors, including Irrus Investments, MedTech Syndicate, Boole Investments, Enterprise Ireland, and a new private investor. This funding positions the company for its next major milestone: obtaining regulatory clearance in the U.S. for its innovative releaze device. With a total fundraising goal of €6.5 million ($6.7 million), SymPhysis Medical plans to conduct preclinical studies, usability tests, and seek FDA clearance by 2026.
Following FDA approval, the company aims to be treating the first patients at leading institutions, including MD
Anderson Cancer Center in Houston, TX and Mayo Clinic in Rochester, MN. These partnerships, developed over years of collaboration, will help demonstrate releaze’s effectiveness and collect clinical data for broader adoption.
Synova Life Sciences is a privately held biotechnology company that develops medical devices and method-

ologies that harness the body’s natural healing ability to address symptoms of degenerative conditions. The company helps physicians and patients harvest autologous stem cells and use their regenerative ability to enhance quality of life.
The company is currently in development with a device, the Synova WAVE, that extracts a patient's own stem cells from their fat (adipose tissue), using a novel, non-enzymatic process to provide patients, doctors and researchers with fast, safe, sterile, high-yield access to stem cells - a solution that allows personalized autologous therapies to scale. The company recently submitted a 510(k) application to the FDA for its technology.
The Synova WAVE Adipose Processing Device is the world’s fastest fat processing system. This breakthrough product offers a 3-minute cycle yielding twice as many stem cells as the industry-standard enzyme, collagenase. From the same sample of fat, cells derived from the WAVE Device are also healthier and more uniform in size.
3ive Labs 50
3M Healthcare 14
4growth VC 25
5Point Foundation............... 25
6 Dimensions Capital 25
15th Rock 25
120 Capital 25
A
Action Potential Venture
Capital.................... 13, 25, 44
Activ Surgical 14
Accelmed Partners 13, 25
Acute Medical Innovation 24
Adagio Medical..................... 14
Adam MacDougall 25
Agnes Health 25
Airespace 27
Alan Cohen ........................... 26
Alcon 12
Alleviant Medical 13
Alle Health 11
Allez Health 11
Alpha JWC Ventures ........... 13
Altra 44
Amadeus Capital Partners 19, 25
Amber Therapeutics ...... 10, 11
Amplitude Ventures 24, 46
Ana Maiques 48
Andera Partners 13
AngelMD................................. 15
Anteris Technologies 18
Anula Jayasuriya 18
Aramco 13
Arboretum Ventures ............ 13
Artelon 12
Ascension Ventures 25, 44
Attune Medical 12
Anne Portwich 22
Aspect Biosystems.............. 13
Aspen Medical 24
Atlantic Bridge 20, 24, 25
Augmedics 14
Ava Investors ........................ 24
Axoft 19
B
Battery Ventures 18, 25
Bausch & Lomb 25
Baxter International 15
Belkin Vision .......................... 12
Big Pi Ventures 24
Blackrock 8
Bolt Medical 13
Boole Investments 51
Boston Scientific 12, 13, 14, 39
Bpifrance 25
Brian Hess 49
BrainTemp 11
Brian Shields ........................ 48
Breakthrough Victoria 25
Breyer Capital 24
Edwards Lifesciences 12
eGarden Ventures 13
eHealth Ventures 24
electroCore........................... 24
Elevage Medical Technologies 13
Elios Vision 25
Elkstone .......................... 20, 24
Elron Ventures 24
Endiatx 35
EndoGastric Solutions 12
Endomag ............................... 12
EndoQuest Robotics 14, 46
EndoSound 36
Endostart 35
ENISA Digital Entrepreneurs 25
Enterprise Ireland 20, 22, 24, 25, 51
Epic Venture Partners 20, 24
EQT Life Sciences .....13, 22, 25 Eric Schmidt 24
Ethicon/J&J 34
Eugene Malinskiy 15
European Women Tech EU grant 24
Eurostars 25
Evidity Health Capital 13
Exor ......................................... 25
Faber ...................................... 25
Far UV Tech. 24
FastWave Medical 20, 24
Fidelity Management & Research Co. ............... 13, 24
Finn Murphy 25 FIRE1 13
Fiscus 25, 44
Fize Medical 24
Flow Medical......................... 24
Fluid Biomed 24, 46 Fourier 13
F-Prime Capital 10, 40
Francis Medical .................... 13
Fred Alger Management 24
Freyja Healthcare 14
Furthr VC 20, 25
GT Medical Technologies 13
Gabrielle Guttman............... 22
Galen Data 45
Gal Noyman-Veksler, PhD 15
Gary Loeb 15
GE Healthcare 28
General Growth Properties 27
Gentex Corporation 25, 49
24, 45
ECM Therapeutics 11
École Polytechnique
Fédérale de Lausanne 50
Eduardo Fonseca 46
Getinge
John Erbey, PhD 50
John Wong, MD ................... 46
Johnson & Johnson ..4, 24, 44
Johnson & Johnson Innovation – JJDC, Inc. 44
K
K2VC 13
Kevin Hykes 14
Kidron Capital ............... 22, 25
Kurt Azarbarzin ..................... 14
L
LaNua Medical 20, 24
Lauxera Capital Partners 47
Lazurite 15
LifeArc Ventures ........... 24, 46
LIFTYA .................................... 25
Lightstone ............................. 25
Lightstone Ventures. 10, 13, 44
Lindera 47
Lionbird 15
LongeVC 25
Longitude Capital 13
Longview Ventures 13
Long-Z Capital 13
Luminar Ventures 24
Luminate Medical 25
Lungpacer Medical 47, 48
Lux 25, 44
M
Maggie Nixon 45
Marc Benioff 24
Mark Froimson, MD 15
Mark Webber 25
Matrix Requirements 47
Mayo Clinic 19
Medical Technology Venture Partners ..............40
MedLumics ........................... 48
MedTech Syndicate 51
Medtech Venture Partners 13, 50
Medtronic 13, 14, 34, 36
Meltwind 19, 25
MeMed 22
METIS Innovative 24, 46
Merit Medical 12
Michigan Capital Network 25
Mike Karim Consulting 15
MindWorks Global 13
Minerva Surgical................... 14
Miyazaki Taiyo Capital ........ 24
M&L Healthcare Investments 20, 24
Modernizing Medicine (ModMed) 34
Mojdeh Poul 14
Moon Surgical 15
Motus GI 35
MoveR 25
MTIP 25
Mumtalakat 24
MVM Partners 13, 49
Myomo 21
N
Nalu 8, 9
21, 22
.......................... 25
24
Investment
27
Nico Corp. 12
Nisshinbo Holdings 24
Nitinotes 24
Novacuff 11
Novo Holdings 13
Novo Nordisk......................... 13
NUA Surgical ................. 22, 25
NVIDIA 27
Occam Labs ......................... 45
OCV......................................... 49
Olav Bergheim 14
Olympus Medical 15, 34, 35 Olympus/Medi-Tate 39
8
39
Biomed ................... 24 Orlando Health Ventures 13 Orly Mishan 14 Ortho-Vision 49
OSF Ventures 25, 44 OSSIO 49 Otolith 11
Oura .................................... 9, 24
Oxford Endovascular Ltd. ... 15
Pacira Biosciences 15
Pallasite Ventures................. 13
Panakes Partners ................ 50 Panda Surgical 25
Pangaea Ventures 13 Paradromics 11 Paragonix 12
Parkway Venture Capital 25 Paul Ziegler 14
Peak6............................... 25, 44
Peakvest ................................. 13
Peter Fischer, PhD 46
Philips 19
Phillips Medisize 51
PhotoniCare 25, 49
Piper Heartland Healthcare Capital ........... 13
Polaris Partners..................... 13
Prashanthi Ramesh 22
Precision 8, 9
Precision Neuroscience 24
Pristine Surgical 36
ProArc Medical 39
PROCEPT BioRobotics...... 39
Profound Medical ................ 25
Proprio.................................... 28
Prosperity7 Ventures 13
Proton Intelligence 24
ProVerum 39
Public-Private Collaboration Projects 24
Puhua Capital 19, 25
Qiming Venture Partners 13
Questa Capital 49
Radical Ventures .................. 13
Rafael Donnay ....................... 14
Rapha Capital....................... 24
Redmile Group...................... 13
Reimagined Ventures 25, 44
Remedy Robotics 28
RevBio 49
Rhino Ventures 13
Riverside Health Advisors 15
RiverVest Venture Partners 13
Roivios 50
Ryan Shelton, PhD 49
S
S3 Ventures 13
S32 24
Sagin Capital & Consulting 24
Saluda Medical 13
SamanTree Medical SA 50
Samuel Levy 47
SandboxAQ 24
Sands Capital ........................ 13
Saventic Health ................... 25
Sensome ......................... 50, 51
Seventure Partners 13
ShangBay Capital 24, 46
ShiraTronics 8, 19
Sickbay 28
Siemens Healthineers 15, 25, 47
Sintx Technologies 15
Solas BioVentures 13
Sonavex 11
SOSV 24
Sotelix Endoscopy 21, 25
SpectraWave .......................... 8
SpinaFx Medical ............ 24, 51
SPR ........................................... 8 Sprint Ventures 25
Stefan Verlohren, MD, PhD 15 Steve Pacelli 25
Stryker 4, 12, 13 SurgiQuest 14 SymPhysis Medical 51 Synapto Ventures 15 Synova Life Sciences 51
T1D Fund: A Breakthrough T1D Venture 13

September 7th - 11th, 2025
JW Marriott Grosvenor House, London
Now in its fourth year, LSI Europe has become a staple of the global Medtech and Healthtech community. The 2025 event in London will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Registration and applications to present are now open for LSI Europe ‘25. Get in touch to learn more about the event.



Scan QR Code to activate your subscription.
The Lens is published monthly and available in print and electronic formats. Copyright by Life Science Intelligence, Inc. All rights reserved. Editor takes care to report information from reliable sources and does not assume liability for information published.
Interested in subscribing? email to: info@ls-intel.com