Vision Beyond the Visible: Intuitive Ventures’ Murielle Thinard McLane and Terri Burke


Diagnostic Electrophysiology
Catheters, Hot Takes, and much more...



Diagnostic Electrophysiology
Catheters, Hot Takes, and much more...
February 2025
September 2025

March 16th - 20th, 2026
Waldorf Astoria, Monarch Beach, Dana Point, CA



Scott Pantel
Chief Executive Officer, Editor-in-Chief

Rebekah Murrietta
Vice President of Media, Contributing Author

Benny Tomlin Contributing Photographer

Henry Peck
Chief Business Officer, Editor-in-Chief

Blake Matrone Sr. Marketing Manager, Contributing Author

Kristin Karkoska Contributing Photographer

Tracy Schaaf
Managing Editor and Content Strategist, Lead Author

Gavin Kennedy Business Development, Contributing Author

Paul Streeto Graphic Designer

Brenna Hopkins Sr. Manager of Content Programs, Lead Author

Almonte Customer Service & Fulfillment

Nicholas Talamantes
Sr. Director of Market Intelligence, Contributing Author

Kelly Williams
Subscriptions & Enterprise Sales
714 847 3540 tel/fax email: info@ls-intel.com
Vol. 2, No. 9 September 2025
Created by LSI, The Lens leverages LSI’s deep industry relationships and proprietary market intelligence to help executives like you build lasting medtech companies. Start your Individual, Group or Enterprise-Wide subscription today!

Scan QR Code to activate your subscription.
The Lens is published monthly and available in print and electronic formats. Copyright 2025 by Life Science Intelligence, Inc. All rights reserved. Editor takes care to report information from reliable sources and does not assume liability for information published.
Interested in subscribing? email to: info@ls-intel.com
Interested in advertising? email to: kelly@ls-intel.com


Dear Readers,
Fresh off the heels of LSI Europe ’25, the momentum we are seeing in medtech is undeniable. Nearly 1,000 executives gathered in London this month, and thousands of meetings took place across three packed days. The energy was electric, and we are already hearing the rumblings of deals, partnerships, and investments that will soon emerge from the week. This is why we do what we do.
Our team is already back at work, planning the full 2026 slate: the return of our flagship LSI USA summit in Dana Point next March, the second year of LSI Asia in Singapore building on strong APAC relationships, and LSI Europe ’26 in Barcelona, expanding on a breakout year for our European summit.
If you joined us in London, thank you for being part of the community. If you missed it, now is the perfect time to get involved and build with us ahead of next year.
This issue of The Lens puts a spotlight on a firm that is building in a way that resonates deeply with us: Intuitive Ventures. Our cover story profiles Managing Partner Murielle Thinard McLane and Senior Partner Terri Burke, two leaders who bring deep operator experience to the venture table and an ability to “see around corners.” They are proving that bold ideas, backed by disciplined capital and relentless focus, will win.
The recent $1.5 billion exit of OrganOx, which they have been integral in, is a testament to their vision. But as you’ll read, Intuitive Ventures is about more than exits. It is about pairing capital with conviction, leveraging the Intuitive Surgical heritage to help founders accelerate innovation at the intersection of digital and physical patient care.
Another story we are proud to share is the journey of Centese, a company that exemplifies the relentless entrepreneurial spirit. From early days inside TheraNova’s incubator to treating over 15,000 patients with its Thoraguard intelligent recovery platform, Centese has turned nights-and-weekends ideation into national traction, with partners like Kaiser Permanente. Co-founders Evan Luxon and Randy Preston are the class of the LSI alumni community, and continue to contribute to (and derive value from) the LSI ecosystem. We are grateful to support their continued growth.
Centese’s story is why we built this platform: to convene the right people, catalyze the right conversations, and help innovative companies scale their impact.
Across the features, interviews, and insights in this issue, you will see a clear throughline: medtech’s future is being shaped by people who are not waiting for change. They are creating it.
Thank you for being part of this movement.
We cannot wait to build what is next, together.
All the best,
Scott Pantel & Henry Peck
Scott Pantel Chief Executive Officer, LSI Editor-in-Chief


Henry Peck Chief Business Officer, LSI Editor-in-Chief


Need fresh inspiration for your medtech company as we enter Q425? We’ve gathered insights from some of our LSI alumni thought leaders to help you stay ahead.
“I think we should learn to value ourselves more and price more aggressively, even though there might be an existing technology out there that has an existing framework…If you look at the predicates, whether it’s FARAPULSE or Shockwave, they’ve built billion-dollar new markets by pricing aggressively and understanding the value they were bringing to clinicians.”

— Cynthia Yee, Partner, Vensana Capital
LSI USA ‘25 Panel: Data Quality vs. Data Quantity — Getting to Market Fast or Getting to Market Right?
“The underlying optimism about ultimate demand for technology greatly exceeds reality most of the time.”

— Paul LaViolette, CEO & Co-Chairman, Pulse Biosciences
LSI USA ‘25 Workshop: The Best Practices Every Innovator Needs to Win in Medtech
“There’s a misperception of investors that money creates growth, and that’s not it; markets, customers, and timing the market right do. We just help enable that, and we help fund it.”

“If the incentives are not aligned and you hide from the investors, then it’s a dysfunctional board and you’re probably the first one out.”

“I always felt like you can’t develop a strategy or understand the business sitting at a desk. You need to be out there speaking to the reps and speaking to the customers.”

— Dave Amerson, Former President & CEO, Neotract
LSI USA ‘25 Signature Series: 1 on 1 with David R. Amerson, Former President and CEO of Neotract
— Tal Wenderow, Managing Partner, Star51 Capital
LSI USA ‘25 Panel: Investors — Do The Best Operators Really Need You?
— David Kereiakes, Managing Partner, Windham Capital Partners
LSI USA ‘25 Panel: Growth Investing in 2025 — Where Will Value Accrue in Later-Stage Medtech?
“You don’t get a prize for growth, and you don’t get a prize for profitability. You get a prize for growing enterprise value.”

— Ashley Friedman, Managing Director, Signet Healthcare Partners
LSI USA ‘25 Panel: Growth Investing in 2025 — Where Will Value Accrue in LaterStage Medtech?
“At BELKIN Vision, we never looked at an exit as a purpose. You look at an exit as a means to bring your technology to as many patients as possible.”

— Daria Lemann-Blumenthal, CEO, NanoDrops
LSI Asia ‘25 Panel: Bio-ConvergenceThe Future of Vision Tech


impacting our global LSI Alumni medtech community.





Biobeat has appointed Raymond Cohen as Chair of its Board of Directors, bringing one of the medtech industry’s most accomplished leaders to the helm. Best known as the co-founder and former CEO of Axonics, Cohen took the company public in 2018 and later led its $3.7 billion acquisition by Boston Scientific in 2024. Since retiring from Axonics, Cohen has joined the boards of multiple high-growth companies, including Nalu Medical, Kestra Medical Technologies, InspireMD,
and RxSight, while also serving as a venture partner with Andera Partners and Sherpa Healthcare Partners.
Cohen now brings his proven leadership and commercialization expertise to Biobeat, which develops a wearable “patch” for continuous, cuff-less ambulatory blood pressure monitoring. The company’s AI-enabled platform captures a broad range of physiological data, such as SpO2, respiration rate, HRV, ECG, temperature, and cardiac output, empowering clinicians with real-time insights. “Biobeat has accomplished what has been an elusive goal for many: a clinically validated and FDA-cleared cuff-less ABPM that has the potential to change the use paradigm for measuring ambulatory blood pressure,” said Cohen. “We see numerous applications for this technology in the new field of renal denervation (RDN) as well as long-term monitoring
of post-cardiac procedure and CHF patients following discharge from the hospital.”

Centerline Biomedical has appointed Jim Dillon as its new President and CEO and a member of its Board of Directors, ushering in a new chapter for the Cleveland Clinic Innovations spinout as it scales its intra-operative positioning platform, IOPS. Dillon brings decades of medtech leadership to the role, including prior President and
CEO positions at BiVACOR, BioVentrix, Reprieve Cardiovascular, and CardioRenal Systems. He succeeds Gulam Khan, who helped lead the company through second-generation product development, 510(k) clearance, and commercialization milestones.
The appointment comes as Centerline prepares to expand commercial adoption of IOPS, its platform for realtime 3D navigation during transcatheter procedures. IOPS reduces radiation exposure for patients and clinicians while improving visualization and device navigation. “It’s an honor to join Centerline at such a pivotal stage,” said Dillon. “The IOPS platform is poised for scaled growth, and I am grateful to have the opportunity to lead the company as it continues to innovate such remarkable technology for patients and caregivers.”

CMR Surgical has appointed Chris O’Hara as Commercial President and General Manager, U.S., as the company scales its commercial footprint following FDA marketing authorization for the Versius Surgical System. O’Hara brings more than two decades of experience in medtech, including senior roles at Virtual Incision, Intuitive Surgical, Globus Medical, and Cardiovascular Systems. He will report to CEO Massimiliano Colella and lead CMR’s U.S. commercial strategy as the company enters its next phase of expansion.
With over 36,000 Versius-assisted procedures completed globally and demand for surgical robotics accelerating, O’Hara’s appointment marks a key milestone in CMR’s U.S. market entry. “CMR Surgical is a hugely innovative business to be part of with an increasingly international presence,” said O’Hara. “This is an exciting time to be joining the company with a focus on growth in the U.S. and building a customer base across the country.”

CorNeat Vision has appointed Tzvika Fisher as COO, marking a pivotal addition to the company’s executive team. Fisher brings over 15 years of operational leadership experience across startups and growth-stage companies in Israel and the U.S. His background includes fractional COO roles, strategic advising, and the co-founding of COOperation, a community of 400+ operations leaders. At CorNeat, Fisher will lead operations, HR, and finance, helping the company align internal systems and teams with its mission to restore sight and improve lives through groundbreaking ophthalmic innovation.
CorNeat’s pipeline spans artificial corneas, synthetic scleral patches, and glaucoma drainage implants, all built on its proprietary EverMatrix platform for permanent tissue integration. Fisher’s appointment supports CorNeat’s transition from clinical success to global commercialization, with an emphasis on scalable infrastructure and high-performance execution.

ImmersiveTouch has appointed Ben Burnham as its new CEO. Burnham brings over 20 years of experience in the medtech industry, with leadership roles spanning craniomaxillofacial, ENT, neurosurgery, plastics, and foot and ankle. He has held key commercial positions at Stryker and most recently led commercial operations at Everis, focused on head and neck and ENT. His track record of scaling organizations, building high-performing teams, and driving strategic growth
will support ImmersiveTouch’s continued expansion.
Burnham joins the company at a key inflection point as it advances its category-defining virtual reality platform for surgical planning and simulation. The company’s advanced platform is reshaping surgical planning and education through FDA-cleared solutions that combine AI, computer vision, and extended reality. Its portfolio includes ImmersiveView 5, a VR planning tool that converts patient DICOM scans into real-time, interactive surgical models; ImmersiveAR, which overlays 3D anatomy onto the surgical field for intraoperative guidance; and ImmersiveEducation, a VR platform offering over 500 pre-loaded cases to support clinical training across specialties.

Keyron has expanded its leadership team with three high-profile appointments as it prepares to launch first-inhuman trials for its ForePass endoscopic weight loss device. The company has named Dr. David Feigal, former Director of the FDA’s Center for Devices and Radiological Health (CDRH), as Head of Regulatory Affairs; Joyce Peetermans,

a longtime Boston Scientific leader, as acting CTO; and Christopher Rowland, former President of Given Imaging, as Chair of the Board.
These appointments follow a series of successful large-animal studies demonstrating ForePass’s strong safety, tolerability, and metabolic efficacy. Designed to mimic the outcomes of metabolic

surgery without cutting or puncturing, ForePass is a fully removable endoscopic device aimed at patients with a BMI ≥35 and diabetes or a BMI ≥40. As Keyron advances toward clinical trials, the company plans to unveil preclinical data this summer showing superiority to semaglutide.
With the addition of Feigal, Peetermans, and Rowland, Keyron is positioned to accelerate development and scale a truly reversible, minimally invasive alternative to bariatric surgery.

Nurami Medical has appointed Amirit Oshri-Chai as its new Vice President of Quality Assurance & Regulatory Compliance. A seasoned medtech executive, Oshri-Chai brings deep expertise in implantable devices, biomaterials, and complex systems that integrate software, hardware, and electronics. Her previous leadership roles include quality and regulatory positions at GE HealthCare, Medtronic, and HP.
Her appointment comes as Nurami scales the commercial rollout of ArtiFascia, its FDA-cleared dural repair graft, and advances development of ArtiFix, a suture-less, adhesive dural substitute designed for use in challenging spine and neurosurgical procedures. With Oshri-Chai leading QA and regulatory strategy, the company is well-positioned to bring its next-generation soft tissue repair solutions to broader markets and accelerate its innovation pipeline.

Planatome has named Keith Jeffcoat as its new President and Chief Innovation Officer, elevating the former CTO to lead the company’s strategic focus on product development and innovation. Jeffcoat brings over 20 years of experience across startups and global medtech leaders, including Johnson & Johnson and Merz Pharma, and will now guide Planatome’s efforts to scale its patented nano-polishing technology through new product offerings and licensing opportunities.

The company also announced the expansion of its Executive Board, with Bill Fender, a veteran executive with over 30 years in the medical device industry, transitioning into a board role, and Matt Likens, former CEO of GT Medical Technologies and Ulthera, joining as well. With this leadership structure in place, Planatome is actively forging strategic partnerships with major medical device companies to deploy its nano-polishing technology and accelerate its mission to enhance surgical performance and improve patient outcomes.

Preci-Health has appointed Dr. Paul Kirchgraber as Chairman of the Board,
bringing decades of global leadership in clinical diagnostics and laboratory services to the Swiss-based medtech and AI innovator. Dr. Kirchgraber previously served as CEO of Covance and Labcorp Drug Development, where he oversaw multi-billion-dollar clinical trial operations across more than 100 countries. In addition to his board leadership, he will provide operational support to accelerate Preci-Health’s strategic and commercial growth.
Dr. Kirchgraber’s appointment comes as Preci-Health advances its EZdraw platform, a needle-free blood collection solution designed for both clinical and at-home use. With deep expertise in global healthcare delivery, clinical trials, and laboratory operations, Dr. Kirchgraber is well-positioned to help guide Preci-Health through its next stage of innovation and expansion in the $80 billion in vitro diagnostics market.

Pulnovo Medical has appointed Dr. Francis Duhay, MD, MBA, FACS, as its new Global Chief Medical Officer, reinforcing the company’s clinical leadership as it expands internationally. A board-certified general and cardiothoracic surgeon, Dr. Duhay brings more than 25 years of experience across clinical medicine, regulatory strategy, and medtech innovation. He previously served as CMO at Edwards Lifesciences, where he played a pivotal role in the development and global adoption of Transcatheter Aortic Valve Replacement (TAVR), now one of the most successful cardiovascular therapies in history.
Dr. Duhay has led more than 80 clinical studies and has worked closely with regulatory bodies, including the FDA and CMS. At Pulnovo, he will guide the company’s clinical strategy as it advances its Pulmonary Artery Denervation (PADN) program and builds on its foundation of mechanism-driven therapies for pulmonary hypertension and heart failure.

Scalpel AI has appointed Jason Webster as its new Head of Commercial Operations and Customer Success, bringing more than two decades of orthopedic operations experience to the London-based medtech startup. Webster spent 24 years in senior roles at Johnson & Johnson, Biomet, and Zimmer Biomet, most recently serving as VP of Commercial Operations at Zimmer Biomet. His deep expertise in orthopedic workflow and operational strategy positions him to lead Scalpel AI’s commercial scale-up across the U.S. and global markets.
Webster will lead efforts to ensure seamless deployment, training, and long-term engagement. By combining computer vision and machine learning, Scalpel AI is redefining how surgical tools and implants are tracked from vendor warehouses to operating rooms and back.

Synchron has appointed Andy Rasdal as Chief of Staff and Mark Brister as Vice President of R&D, strengthening its executive leadership as the company advances its Stentrode brain-computer interface (BCI) through U.S. clinical trials and prepares for global market entry. The company says both leaders bring a strong track record of launching and scaling category-defining technologies, and their appointments reflect Synchron’s focus on building the infrastructure needed to lead in the rapidly evolving BCI space.
Rasdal, a former CEO of Dexcom and founding CEO of Obalon Therapeutics, also held senior roles at Medtronic Vascular and Arterial

Vascular Engineering. He joined Synchron’s board in 2023 and now transitions to a full-time executive role. Brister, a veteran product development leader with more than 100 product launches and over 450 patents, previously held R&D leadership positions at Guidant, Arterial Vascular Engineering, Medtronic, Dexcom, and Obalon. Their combined expertise positions Synchron to accelerate its mission of restoring independence to people with severe paralysis.

Tensive has appointed Dr. Bill Hunter, MD, as Chairman of the Board as the company advances toward pivotal clinical milestones and regulatory approval for its REGENERA and SOFTAG bioresorbable scaffolds. A veteran medical device innovator, Hunter is the founder and CEO of Canary Medical, and previously founded Angiotech Pharmaceuticals, where he led the development of one of the world’s first drug-eluting coronary stents. His appointment strengthens Tensive’s board leadership as the company prepares for commercialization in the U.S. and Europe.
Backed by strong first-in-human and interim pivotal trial data, Tensive’s platform is designed to restore breast shape by placing an implant that is gradually absorbed and replaced by natural tissue during lumpectomy surgery. The company is targeting the large unmet need among the 1.9 million patients per year who undergo lumpectomy without reconstruction. Hunter’s leadership is expected to support Tensive’s regulatory strategy, market entry, and global growth.

Vektor Medical has appointed Kelly Perez as CFO and Senior Vice President of Operations, adding seasoned leadership as the company scales adoption of its FDA-cleared arrhythmia mapping solution, vMap. Perez brings a proven track record in financial strategy and operational discipline from prior leadership roles at Omniome, Luxtera, and Sequenom
Her dual appointment comes as Vektor accelerates the rollout of vMap, a non-invasive software platform that uses 12-lead ECG data to localize arrhythmias in under a minute. Perez will support the company’s mission to drive efficient cardiac care, expand collaborations with health systems and medtech partners, and help lead the next phase of growth.

Vitestro has named Bas Nieuwenhuis as Vice President of Engineering to lead technical operations as the company scales deployment of Aletta, its CE-marked Autonomous Robotic Phlebotomy Device (ARPD). Nieuwenhuis brings nearly 30 years of product development and R&D leadership, including over a decade at BD Kiestra, where he led the development of its lab automation platform.
Nieuwenhuis succeeds Bert Wiggers, who is retiring after years of dedicated leadership at Vitestro. As VP of Engineering, Nieuwenhuis will guide the next phase of innovation and implementation as Vitestro prepares for U.S. regulatory approval and expands access to its robotic blood draw technology across international markets.
L&G (led), Allegis Capital (led), Lightrock (led), Oxford Science Enterprises, GV, Blue Venture Fund, Oxford University, UChicago Medicine’s venture investment vehicle, UPMC Enterprises
Majority stake acquisition by a syndicate of public and private investors, including K5 Global, Bezos Expeditions, Wellington Management, Alpha JWC Ventures, Alpha Wave Ventures, Venture Investors Health Fund, Lumira Ventures, Hatteras Venture Partners, Early Stage Partners, Amzak Health, HealthQuest Capital, Yonjin Venture, the State of Wisconsin Investment Board, the State of Michigan Retirement System, Johnson & Johnson – JJDC, Inc. (JJDC), and others
Elevage Medical Technologies (led), Ally Bridge Group (led), Northwell Health, SPRIG Equity, an undisclosed strategic investor, Norwest, New Enterprise Associates (NEA), Viking Global Investors, Action Potential Venture Capital, Abbott, Boston Scientific, Euclidean Capital, Richard King Mellon Foundation, Morgenthaler Ventures, ShangBay Capital, Ascendum Capital, Catalio Capital Management, Gilmartin Capital, Midas Capital 8/11/2025
Your one-stop-shop for global market sizing and analysis, procedure volume data, startup company and deal tracking, curated insights, and more.
Conformal Medical Series D Proprietary, conformable LAA device to reduce stroke risk
Reprieve Cardiovascular Series B
Fluid management technology for acute decompensated heart failure
$61M
Vivani Medical Other Drug and device implants to address unmet needs in chronic diseases $10M

Deerfield Management (led), Arboretum Ventures, Lightstone Ventures, Santé Ventures, Genesis Capital, Rex Health Ventures, Cadence Capital, an undisclosed strategic investor 8/13/2025
Share purchase agreement to sell approximately 8 million shares of common stock at $1.26 per share 8/14/2025 RIVANNA Medical Grant
Epidural needle guidance device utilizing automated 3D navigation
Better Medicine Other AI software for lesion detection in radiology
Vensana Capital (led), The Swiss Diabetes Venture Fund, Ascensia Diabetes Care, Winklevoss Capital, other undisclosed prominent investors
Investment Fund for Northern Ireland (IFNI) (led), Whiterock’s Growth Capital Fund, IQ Capital, Innovation Ulster Ltd, the Angel CoFund, private investors
8/19/2025
$1.2M Soulmates Ventures (led),
Venture Partners (led), aMoon Fund, Visionary Ventures, Sectoral Asset Management, Radius Special Situations Fund, Civilization Ventures 8/20/2025

At Intuitive Ventures, Managing Partner Murielle Thinard McLane and Senior Partner Terri Burke bring a rare edge to medtech investing: the ability to see around corners. Drawing on decades of experience as operators, startup CEOs, and healthcare executives, they understand how today’s decisions reverberate years into the future. Recent wins, including an impressive $1.5 billion exit, illustrate the fund’s mission, uniquely positioned at the crossroads of bold financial returns and transformational impact on patient care.
Thinard McLane and Burke’s lived experience fuels the philosophy of the forward-thinking venture capital firm, Intuitive Ventures. Modeling its Intuitive Surgical heritage, the goal is to back bold teams where the physical and digital delivery of patient care meet, with grounded judgment, relentless focus, and a deep commitment to improving patient lives. With $250 million under management and access to Intuitive’s global reach, Intuitive Ventures’ focus spans medtech, diagnostics, and digital health. The team blends venture discipline with clinical empathy, spotlighting founders who aren’t just building companies, but reshaping the trajectory of patient care in parallel with Intuitive’s transformative impact on surgical robotics.
Despite shifting macro conditions, Intuitive Ventures’ ability to flex across company stages and therapeutic areas has allowed it to stay both nimble and disciplined, with in-house expertise seemingly tailor-made for today’s complex environment.
Together, Thinard McLane and Burke are co-creating the firm’s platform to be uniquely positioned for financial return as well as meaningful health transformation. Added to its past investments, the fund has made four recent deals, including with Apreo Health and OrganOx (the latter just acquired by Terumo for $1.5 billion), with the other two in stealth mode.
“We continue to be excited about medtech,” says Thinard McLane. “We’re architecting a fund that is financially motivated yet strategically bold, focused on the future of minimally invasive care. Our recent investments speak to the most transformative technologies out there that are addressing large unmet needs around the world, both from the patient and the physician’s point of view.”
“We are very busy. Venture capital really has no downtime,” noted Burke. “The best deals have a lot of interest and engagement, and they move quickly. At Intuitive Ventures, we pride ourselves on being able to move at the pace of the market. Investment fundamentals are strong, and we’re seeing both later-stage and transformative early-stage opportunities emerge. Good ideas still win.” The Lens caught up with the two powerhouse leaders recently to peer
inside their current venture strategy, powered by experience, agile flexibility, purpose, and a patient-first vision.
Thinard McLane and Burke are a rare breed in venture capital, as investors who have personally experienced the volatility, urgency, and grit of building healthcare companies.
“I’ve lived business cycles,” says Burke. “You select a strategy and have to live with the consequences. That teaches you to anticipate pitfalls and see around corners.”
“I’ve been in their shoes,” Thinard McLane echoed. “Raising money, navigating timelines, launching under pressure. It changes how you evaluate teams and plans.”
“Good ideas still win.”
— Terri Burke
That operator empathy is foundational to the duo’s approach at Intuitive Ventures. It shapes how they evaluate companies, engage with founders, and add value beyond capital.
Both Thinard McLane and Burke have stood on the frontlines of healthcare innovation. Thinard McLane, who joined Intuitive Ventures in 2023, brings a depth of experience across digital health tech, digital health, diagnostics, and biotech domains, leading investments in companies at the intersection of physical and digital delivery of patient care. She has spent over two decades as an investor, entrepreneur, and Fortune 500 executive, most recently serving as CEO of molecular diagnostics company, Ontera, and as co-founder of Nooma Bio, a life science tools platform focused on detecting cytogenetic abnormalities.
Thinard McLane started her foray into venture capital as an Executive in Residence for GE Ventures, where she helped launch Verana Health, which is assembling the largest clinical database in medicine empowering physicians and accelerating research
for patients. Previously, she was a General Manager at McKesson, where she oversaw multi-billion-dollar P&Ls spanning therapeutics, surgical supplies, services, and data analytics, and prior to that served in strategy and leadership roles at Genentech, Novartis, and Optum
Burke’s background is also wellrounded, with more than two decades of medtech investing and operational leadership experience. This includes roles as a medtech institutional investor and hands-on operator at both public companies and early-stage startups. Prior to joining Intuitive Ventures this past March, Burke was a venture partner and head of medtech investments at Epidarex Capital, where she led the firm’s Fund III US/UK investment strategy, deal flow, and medtech brand. She also previously co-founded and served as CEO at Protaryx Medical, a pioneering structural heart and electrophysiology company. Previously, Burke served in various senior leadership roles at Edwards Lifesciences, including Vice President of Global Marketing and Business Development, where she streamlined and re-focused the heart valve business to help drive growth.
Burke is also committed to supporting the women in the venture capital community, and is a co-organizer of the “Women in VC” networking event held at LSI and other medtech ecosystem events (see photo on the next page). At LSI USA ‘25, both Burke and Thinard McLane participated in the session and noted the growing momentum in gender diversity and the importance of creating space for connection.
“It was a wonderful launch of the Women in VC event at LSI, a big conference that has the critical mass of people that can then bring together a subset of investors,” says Burke (note that both Thinard McLane and Burke have participated in panel discussions at past LSI events).
“The idea behind it was simply to build a community across partner-level women in venture capital, so that we’re comfortable knowing each other and having conversations,” she continues. “I will say that the VC community is quite friendly and collaborative, but we found that there are fewer women partners across the ecosystem. With the support of LSI and Knobbe Martens, we’ve
been able to create a space where everyone can get together and share some energy around what we’re doing in this space. We’ve hosted a few events since then, and have met other women partners and leaders across funds. There’s power in those numbers. We’re building it fairly organically, but we like that it is casual and comfortable for a lot of the women in VC to participate in. We extend it to our male colleagues as well to join us, so it’s not exclusively just for women.”
Founded in 2020, Intuitive Ventures is the independent venture arm of Intuitive Surgical, the robotic surgery pioneer behind the da Vinci platform (see “The Rich History and Bold Future of Intuitive: Inside Gary Guthart’s Vision for Robotics and Healthcare,” The Lens, April 2025). Yet unlike traditional corporate venture capital models, Intuitive Ventures operates with the rigor and independence of a financial VC fund. With over $250 million in assets under management, the firm seeks to invest in early-stage companies that align with its mission to accelerate the growth of advanced healthcare, fueling breakthroughs with the capital needed to improve outcomes and deliver more efficient care.
“With the support of LSI and Knobbe Martens, we’ve been able to create a space where [women in VC] can get together and share some energy around what we’re doing in this space.”
— Terri Burke
“What Gary Guthart and Julian Nikolchev envisioned was a real fund that is return-driven, but one that could pursue truly transformational technology,” says Thinard McLane. “That resonated deeply with me, having been a founder myself.”
For entrepreneurs, this dual identity is powerful. It means access to worldclass clinical, technical, and commercial expertise, without the pressure or expectation of acquisition.
Every company in Intuitive Ventures’ growing portfolio reflects the firm’s core values: advancing positive patient outcomes, improving provider experience, and reshaping how care is delivered through technology and innovation.
Thinard McLane outlined the fund’s investment framework, focusing on three key areas: 1) expanding access and care coordination through solutions that bridge physical and digital barriers to improve patient outcomes, 2) precision diagnostics and intervention using technologies tailored for earlier detection and targeted treatment, and 3) big data and digital enablers that leverage data to power these strategic initiatives.
When assessing investments, the team starts with unmet clinical needs and the size of the market opportunity. They then evaluate defensibility (IP, differentiation) and, above all, the team.
“At the end of the day, you’re betting on the team to be successful,” she says. “A dealbreaker is if we don’t believe they can take it to the next stage, or if they’re not collaborative and willing to take feedback.”
So what are the non-negotiables when Intuitive Ventures evaluates a company?
1. Transformational potential: Does the innovation address a major unmet need? Can it touch a million lives?
2. Exceptional teams: Are the leaders humble, hungry, and able to execute? Are they coachable, bold, and resilient?
3. Clear milestones: Is there a line-of-sight to value inflection

points? Can the company secure follow-on capital?
4. Investor alignment: Are the co-investors aligned for the long haul, including the long timelines that PMA products can demand?
Thinard McLane and Burke also look for strong syndicate partners. “It’s increasingly important to have a good syndicate because rounds take a little longer to fill, and the timeline to exit is a little longer. Having expertise and deep pockets around the table really helps companies thrive,” says Burke.
“Transformation takes time and money,” adds Thinard McLane. “The best ideas need the right teams, syndicates, and patience to succeed.”
“What Gary Guthart and Julian Nikolchev envisioned was a real fund that is returndriven, but one that could pursue truly transformational technology.” — Murielle Thinard McLane
The Intuitive Ventures model is “capital plus”: funding but with a deep bench of value-added support across clinical development, regulatory strategy, commercial planning, and technical mentorship. Burke emphasized how unusual this level of engagement is in venture capital.
“We spend a lot of time with our portfolio companies,” she says. “Our investment committee members, even senior leaders with decades of experience, will jump in to help on clinical, regulatory, product development, and business strategy with our portfolio companies. That reach is a true differentiator.”
“We bring institutional rigor to what we do,” adds Thinard McLane. “We
partner well as a significant member in these deals, and we support companies all the way through to exit. We help them find lab space, map their reimbursement pathways, and connect with community physicians.”
The team even surveys its CEOs annually to measure the firm’s effectiveness, which is a rare practice among funds.
“We run our own NPS [Net Promoter Score] survey,” Thinard McLane explained. “That’s how serious we are. We want to know how we’re doing, where we can improve, and how to better support our CEOs. It’s not just a check. It’s about leveraging our network and expertise to help them succeed.”
The feedback has been overwhelmingly positive, with the fund ranked bestin-class in value-add by its portfolio leaders.
“We take that role seriously,” added Burke. “We want to be the kind of investor founders seek out for the long haul.”
Today, each startup in Intuitive Ventures’ portfolio represents the future of healthcare, with smarter, more precise, and more connected technology. They all share a minimally invasive ethos, platform potential, and teams capable of execution. Recently announced deals illustrate the team’s ability to spot and support exceptional technologies, and now, the firm has its name attached to a $1.5 billion exit.
In March, Intuitive Ventures joined OrganOx’s final private round to accelerate commercialization of its metra Normothermic Machine Perfusion (NMP) platform for donor livers, along with HealthQuest Capital, Lauxera Capital, and Terumo Ventures Together with the first closing of its private placement in February, OrganOx, which was spun out of the University of Oxford in 2008, raised $160 million in primary and secondary equity financing to accelerate growth opportunities for its metra technology platform in the large and rapidly expanding organ technology global market.
And now, this investment has just converted to one of the largest-ever deals in the UK medtech sector, and
one of the biggest Oxford spinouts. In late August, global medtech giant Terumo announced its strategic entry into the high-potential organ transplantation sector by entering a definitive agreement to acquire OrganOx for $1.5 billion.
OrganOx’s metra device was FDAcleared in 2021 and launched in the U.S. the following year. It has also obtained regulatory approvals in the EU, UK, Australia, and Canada, and is commercialized in all these regions. To date, metra has been used in over 6,000 liver transplant procedures worldwide. OrganOx is also developing a kidney NMP device, with commercialization targeted around 2030.
“OrganOx is transforming the way organ transplants are done, and is soon expanding beyond liver transplantation into kidneys. We’re proud to have been part of their journey to this incredible milestone,” commented Thinard McLane.
In further support of Intuitive Ventures’ keen ability to identify transformative bets early, its investments have extended globally into the areas of respiratory health, heart valve replacement, cancer, sleep apnea, metabolic
• Founded: 2020
• Assets Under Management: $250M+
• Fund Size: $100M Fund I, $150M Fund II
• Investment Stages: Seed → Growth
• First Check Size: $5-$10M
• Target Portfolio: 10-15 companies per fund
• Portfolio Sectors: Medtech, diagnostics, and digital health
• Value-Add: Board observer roles, regulatory/commercial mentorship, KOL access, technical expertise
disease, and other medtech markets with large unmet clinical needs. In August, the firm participated in Apreo Health’s $130M Series B, backing the company’s BREATHE Airway Scaffold for severe emphysema, a condition affecting 3 million patients in the U.S. with very limited treatment options.
Apreo Health, formed in 2021 out of medtech incubator (and LSI alum) The Foundry, has developed the BREATHE Airway Scaffold, a bronchoscopically delivered, self-expanding implant designed to gently release air trapped in the lungs of patients with severe emphysema. Severe emphysema, a debilitating form of chronic obstructive pulmonary disease (COPD), is a global health crisis, ranked by the World Health Organization as the fourth-leading cause of death and eighth-leading cause of poor health worldwide.
“We are quite excited to help support a technology that may completely transform this market, and patients affected by this deadly progressive disease,” says Thinard McLane.
The financing was co-led by new investors Bain Capital Life Sciences and Norwest, with participation from another new investor, F-Prime. Returning

Source: LSI Europe ‘25
investors Lightstone Ventures and Santé Ventures also participated in the round. The Series B will primarily support BREATHE-3 (Bronchoscopic RElease of Air Trapped in Hyperinflated Emphysematous Lung, Study 3), a multicenter, randomized controlled trial designed to evaluate the safety and efficacy of the Apreo BREATHE Airway Scaffold. Additionally, the Series B financing will fund regulatory activities related to BREATHE-3 and early commercialization efforts to demonstrate strong market potential. The company’s device was granted FDA Breakthrough Device Designation in May 2024.
Apreo Health emerged from stealth mode this past May to unveil its airway scaffold device and first-in-human sixmonth clinical data, with results including a 92.4% technical success rate in device placement and no post-procedural pneumothoraxes observed.
Intuitive Ventures’ other portfolio companies have also made significant strides in recent months. Earlier this year, Capstan Medical achieved the world’s first robotic-assisted mitral valve replacement in a human, in Chile. In October 2023, Intuitive Ventures participated in an oversubscribed $31.4 million Series B funding round in Capstan, led by Eclipse. (LSI alum Maggie Nixon,
CEO of Capstan, brings more than 20 years of experience at Intuitive Surgical, and co-founder and CTO Dan Wallace’s background includes senior roles at Intuitive, Hansen Medical, Auris Health, Cephea Valve Technologies, and others.)
Burke brings deep operating and investment experience in the cardiovascular space, and sees this as an important area for Intuitive Ventures to build upon. “I saw a statistic from the American Heart Association stating that every 34 seconds a patient dies from cardiovascular disease,” she says. “Despite first-rate medications, therapies, and devices, we still haven’t solved it. Those are the types of things we look for, transformative therapies and markets that are really in need of better patient and physician solutions.”
Intuitive Ventures is also focused on early cancer diagnosis, as a specialty area in need of transformative solutions. In October 2022, the fund participated in a $14 million Series A round in Optellum, developer of AI-driven lung nodule detection and early lung cancer diagnosis technology, led by Mercia. This past April, Optellum signed a landmark partnership with Bristol Myers Squibb to advance AI-driven early detection of lung cancer, one of today’s deadliest
Thinard McLane joined Intuitive Ventures in 2023 and brings a depth of experience across digital health tech, digital health, diagnostics, and biotech domains to lead investments in companies that are shaping the future of minimally invasive care throughout the patient continuum.
She is a seasoned operator and executive with over 20 years of experience building and scaling organizations as an investor, entrepreneur, and Fortune 500 executive. She most recently served as CEO of molecular diagnostics company, Ontera, and as co-founder of Nooma Bio, a life science tools platform focused on detecting cytogenetic abnormalities. She started her foray into venture capital as an Executive in Residence for GE Ventures, where she helped launch Verana Health, the largest clinical database in medicine empowering physicians and accelerating research for patients. While there, she also supported investment in Genome Medical, a telehealth provider of genomic medical insights, and Arterys, the first cloud-based deep learning imaging software startup to receive FDA clearance. Previously, she was a General Manager at McKesson, where she oversaw multi-billion-dollar P&Ls spanning therapeutics, surgical supplies, services, and data analytics, and before that served in strategy and leadership roles at Genentech, Novartis, and Optum.
Passionate about bridging the gender gap in the C-suite of venture-backed companies, she served on Springboard Enterprises’ executive council for pharmaceuticals and digital health, a network dedicated to building high-growth, technology-oriented companies led by women.
Thinard McLane holds an MBA from Wharton and an MS in Finance and the Science of Management from Université de Lyon, and started her career in financial services.
diseases. Under the agreement, Optellum will apply its AI-powered imaging and clinical decision support platform to evaluate the real-world impact on patient outcomes. The Oxford-based medtech company has been the first and only to achieve FDA 510(k) clearance for the application of AI decision-support software in lung cancer diagnosis, that is reimbursable under the CMS New Technology Ambulatory Payment Classification.
Other Intuitive Ventures portfolio companies and technologies include:
• Amber Therapeutics: The first adaptive neuromodulation therapy for mixed urinary incontinence in women.
• Endogenex: Device therapies for GI and metabolic disease processes.
• Flywheel: Comprehensive data management platform accelerating the future of biomedical research and collaboration.
• MedCrypt: Cybersecurity platform protecting software-enabled medical devices.
• SURGE: Platform for extended local delivery of cancer immunotherapy in the context of tumor resection.

Source: LSI USA ‘25
• XII Medical: Neuromodulation for obstructive sleep apnea.
“Each of these companies represents a different piece of the future we’re trying to build,” says Burke. “Not just better tools, but better outcomes.”
With one-third of its portfolio based in Europe, Intuitive Ventures remains globally minded. At the LSI Europe ‘25 event in London earlier this month, Thinard McLane participated on an insightful panel discussing investment trends in the U.S. versus the EU.
“There is great technology in Europe,” she says. “OrganOx proves these companies can win, not only for patients but for investors as well. The goal is to be the partner of choice for the most mission-aligned entrepreneurs in healthcare. We want to be known not just for capital, but for conviction.”
As the fund looks ahead, both leaders emphasize cadence and momentum. “We pride ourselves on being active in the market and in the community,” says Burke. “We get to imagine what’s possible, and we get to make it happen. Stay
tuned, we have a very active cadence here and we expect a few more investments this year.”
The Lens’ sit-down with these two leaders ended by asking: What advice would they give to early-stage medtech entrepreneurs?
“Dream big, but focus. Be persistent and resilient. Do your homework. Know your investor fit, and be able to clearly communicate your value. And as Terri says, good ideas will win,” offers Thinard McLane.
“Know your market. Be ready to adapt,” adds Burke. “Be willing to make mistakes, and to course-correct. And stay mission-focused through the hard stuff, because it’s coming.”
“The Rich History and Bold Future of Intuitive: Inside Gary Guthart’s Vision for Robotics and Healthcare,” The Lens, April 2025
“Solving Wicked Problems at the Intersection of Healthcare and Technology: Insights from Intuitive Foundation’s Catherine Mohr, MD,” The Lens, February 2025
Burke is Senior Partner at Intuitive Ventures, where she manages and grows the firm’s investment platform by leading investments that advance minimally invasive care. She leverages her deep investment and operating experience to foster the firm’s brand and collaborate with the team to refine its investment strategy.
Burke has more than 20 years of strategic business and investment leadership experience delivering exceptional results in complex markets. Previously, she led the medtech investment strategy and portfolio for Epidarex Capital’s Fund III US/UK. She is also co-founder and former CEO of Protaryx Medical, where she successfully built the company and raised several rounds of financing. Additionally, Burke was a member of the Senior Leadership Team at Edwards Lifesciences (NYSE: EW) and held multiple vice president positions in the heart valve business, including managing a cross-functional technical development program, leading global marketing, and overseeing business development for several strategic acquisitions and partnerships. Burke has prior experience in product marketing with Guidant Corporation and in health economics and reimbursement as a consultant with Covance.
Burke serves as a Board Advisor for Raydiant Oximetry and a Trustee for the Roessler-Chadwick Foundation, a Director for the Roessler-Chadwick Foundation Group, and is an early member of MedtechWOMEN. Terri previously served as a Board Director for Ryme Medical, Radiant Oximetry, Protaryx Medical, and Flashback Technologies (dba Impact Vitals).
Burke holds a B.S. in Biology from Duke University and an MBA from the University of Southern California. She was a member of the Division I field hockey team at Duke and remains active in mentoring student-athletes.




As cardiac ablation procedures surge, the market for diagnostic electrophysiology (EP) catheters is experiencing accelerated growth. Driven by pulsed-field ablation (PFA) adoption, rising arrhythmia incidence, and an expanding product mix, this segment is evolving rapidly. Recent FDA approvals and strategic product launches from top competitors signal a new era of innovation in EP mapping and diagnostics.
Diagnostic EP catheters serve as critical tools in the diagnosis and treatment of cardiac arrhythmias. These devices record electrograms from within the heart to assess electrical activity, guide treatment decisions, and support pro-
cedures such as cardiac ablation. Driven by the rapid increase in utilization of cardiac ablation procedures for heart rhythm disorders like atrial fibrillation (AFib) and supraventricular tachycardia, demand for diagnostic EP catheters is accelerating across global markets.
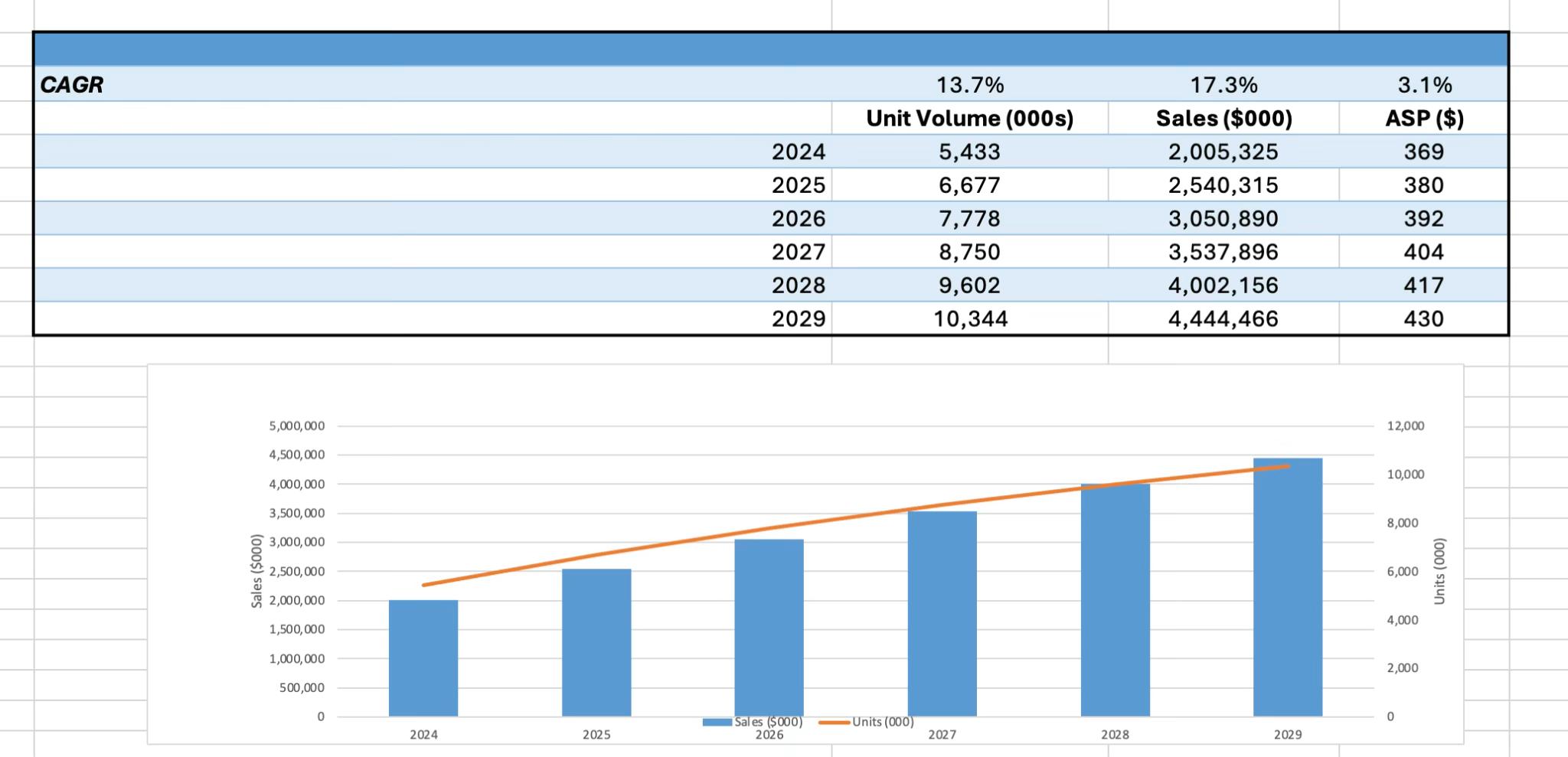
In 2024, diagnostic EP catheters reached $2.01 billion in worldwide sales,
supported by 5.4 million unit sales. The market is projected to grow to more than $4.44 billion by 2029, representing a 17.3% compound annual growth rate (CAGR). Unit volume is forecast to grow at a 13.7% CAGR, while average selling prices (ASPs) are expected to climb modestly from $369 to $430 over the forecast period. Growth is outpacing
prior forecasts, driven largely by the impact of PFA and a continued shift toward higher-cost, high-performance mapping catheters.
Beyond procedural growth, one of the most significant contributors to market expansion is the shift in product mix toward advanced diagnostic technologies. While traditional fixed-curve catheters still serve as the baseline, steerable and high-density mapping catheters are gaining traction. These products can cost up to seven times more than conventional offerings, and their increasing use in complex procedures is lifting ASPs and total revenue.
In parallel, diagnostic EP catheters play an important role in procedures
beyond ablation. Device implantations, including pacemakers, implantable cardioverter defibrillators (ICDs), and cardiac resynchronization therapy (CRT), routinely require intracardiac electrogram monitoring. As these therapies expand globally, so too does the need for real-time, high-fidelity diagnostic capabilities.
2024 marked a turning point for the diagnostic EP catheters market, with major competitors racing to integrate their mapping systems with PFA platforms. Boston Scientific’s entry into the U.S. market with its FARAPULSE PFA System drove more than $1 billion in revenue and accelerated the shift
toward single-shot ablation paired with diagnostic mapping. To support this transition, the company launched its FARAWAVE NAV Ablation Catheter and FARAVIEW Software, designed for seamless integration with the OPAL HDx Mapping System. The new tools combine therapy delivery and real-time visualization to streamline PFA workflows.
Abbott also advanced its diagnostic EP footprint with FDA clearance for the Advisor HD Grid X Mapping Catheter, Sensor Enabled. This system features an innovative electrode configuration that improves signal resolution regardless of catheter orientation, an important feature for accurately mapping complex arrhythmias. Paired with Abbott’s EnSite X mapping system and its upcoming Volt PFA System, the company’s diagnostic tools are designed to offer physicians more precise control and visualization during ablation.
Source: LSI Market Intelligence

Medtronic joined the fray with the FDA approval of its Affera Mapping and Ablation System and the Sphere-9 Catheter, which delivers dual-energy therapy while simultaneously enabling high-density mapping. By offering both radiofrequency (RF) and PFA capabilities in a single catheter, Medtronic aims to simplify workflow and provide physicians with greater flexibility to tailor treatment based on patient needs.
In May 2025, a California jury ruled that Johnson & Johnson subsidiary Biosense Webster, the current market leader, violated antitrust rules by withholding clinical support from hospitals using reprocessed catheters. The verdict resulted in $147 million in damages awarded to reprocessing company Innovative Health. A federal judge then tripled the damages to $442 million and granted Innovation Health’s motion for a permanent injunction.
Elsewhere in the industry, CoreMap received Investigational Device Exemption (IDE) approval from the FDA to extend its INvENI study into the U.S. The company’s EP mapping system uses a dense array of micro-scale electrodes and novel algorithms to identify AFib drivers, potentially enabling more tailored ablation strategies.
Another notable regulatory milestone came from Kardium, which received FDA premarket approval for its Globe Pulsed-Field System. The Globe platform uniquely combines single-shot PFA and high-density mapping in a single device, streamlining pulmonary vein isolation (PVI) while offering real-time visualization. The device’s clinical data, presented at the 2025 Heart Rhythm Society annual meeting, showed 78% freedom from atrial arrhythmia at one year in paroxysmal AFib patients with zero device-related primary safety events.
Together, these regulatory milestones and next-generation product launches highlight just how quickly the diagnostic EP catheter landscape is evolving. As mapping technologies grow more integrated with PFA and other ablation systems, the bar is rising for precision, performance, and clinical versatility.
With high-growth procedural trends, increasing reliance on sophisticated mapping systems, and the convergence of diagnostic and therapeutic platforms, the diagnostic EP catheter market is undergoing a transformative phase. As AFib and other arrhythmias continue to affect millions globally, diagnostic EP catheters will remain essential to advancing treatment and improving outcomes.
Peripheral vascular guidewires are a foundational technology for transcatheter interventions, enabling safe and precise navigation through the vasculature to treat peripheral vascular disease (PVD). Global demand for these devices is rising steadily, driven by a growing aging population, increased PVD diagnosis, and advancements in access techniques, such as transradial intervention.
Peripheral vascular guidewires are used in diagnostic and interventional procedures to navigate vessels and guide treatment devices, such as stents or catheters. Their utility spans both routine and complex interventions, with wire selection based on key characteristics such as core diameter. Smaller diameter wires have increased flexibility, while larger diameter wires offer increased device support.
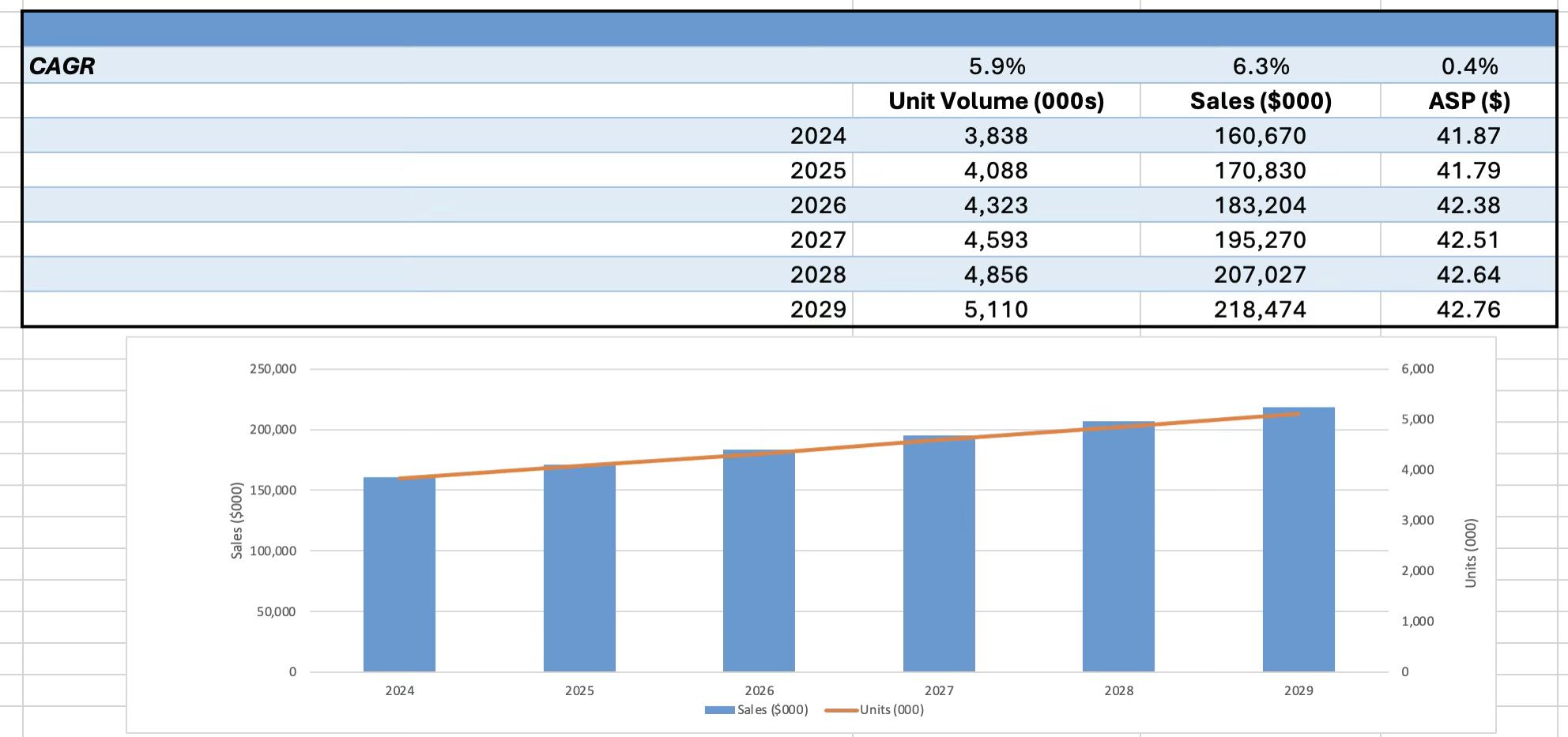
According to LSI Market Intelligence, global peripheral vascular intervention procedure volumes are expected to grow at 5.2% annually from 2024 to 2029. This uptick reflects an increase over last year’s projection and corresponds directly with increased unit demand for peripheral vascular guidewires. Although pricing is forecasted to rise more modestly, just 0.4% per year due to reimbursement pressure, dollar volume growth is still projected to reach 6.3% annually over the same period.
Behind the growth in procedures is a convergence of epidemiological and clinical trends. The incidence of peripheral vascular disease is increasing as the population aged 65 and older expands worldwide, with the U.S. Census Bureau forecasting this demographic to grow at 3.1% per year from
2024 to 2029. In tandem, broader adoption of vascular screening is surfacing more patients in need of intervention. According to a 2023 study in The Lancet Global Health, the global population affected by peripheral artery disease nearly doubled from 65.8 million in 1990 to 113 million in 2019, representing a 1.9% annual increase.
Boston Scientific is the dominant player in the peripheral vascular guidewire market, holding nearly 80% of global share. Its broad portfolio, longstanding brand recognition, and deep integration across peripheral vascular workflows have cemented its leadership
position. However, other players continue to expand their presence through strategic partnerships and the introduction of new products.
ASAHI INTECC recently made headlines with a strategic distribution move in Europe. The company assumed European distribution rights for the Medikit Super Sheath, ensuring continued access to a product known for its kink resistance, smooth transition, and vessel-friendly design.
Medtronic, meanwhile, expanded its U.S. portfolio through an exclusive agreement with Future Medical Design, a Japanese manufacturer with a longstanding reputation in guidewire innovation. The deal brings 10 new F-14 and F-18 stainless steel peripheral guidewires to the U.S. market, including the nation’s first 400 cm 0.018” wire
Source: LSI Market Intelligence
optimized for transradial access. Available in 300 cm and 400 cm lengths and in 0.014” and 0.018” diameters, these guidewires are designed to provide crossability, trackability, and tip retention across both routine and complex cases. Features such as stainless-steel cores and multiple tip loads equip clinicians with precise control, particularly when navigating tortuous or occluded vessels.
As procedural volumes climb and transcatheter techniques evolve, peripheral vascular guidewires remain
an essential cornerstone of devicebased therapy. Companies that can offer a diverse range of wire configurations, support transradial strategies, and deliver reliable performance in increasingly complex cases will be best positioned to capture growth in this segment.
With a global aging population and improved diagnostic rates driving procedure demand, the guidewire market is expected to expand consistently through 2029. While pricing pressure remains a headwind, ongoing innovation, product differentiation, and targeted distribution agreements, like those seen from ASAHI INTECC and Medtronic, highlight how leading players are adapting to maintain a competitive edge and support clinicians worldwide.
Supplier Sales & Market Shares, 2024
Supplier Sales and Market Shares, 2024

Every week, LSI’s Market Intelligence team tracks the signals, strategies, and standout stories shaping the global medtech industry. August brought big moves across M&A, venture, and public markets, with corporates leaning into organ transplant innovation, build-to-buy models gaining ground, mega-rounds stacking up, and IPOs picking up pace. Here’s what defined the pulse of medtech this past month.


Olympus made headlines with a strategic build-to-buy agreement to acquire Swan EndoSurgical for up to $458 million. Co-incubated with Revival Healthcare Capital, the startup is developing
a robotic platform for endoluminal surgery. Olympus contributed an initial investment of at least $65 million and secured exclusive acquisition rights.
The deal highlights a growing trend in medtech M&A: structured partnerships that allow strategics to back early-stage technologies with defined exit paths. These “build-to-buy” models reduce risk for buyers, support startups with operational independence, and align incentives across all parties.
Other recent examples include:
• Medtronic and Twelve (2014)
• Abbott and Cephea Valve Technologies (2015)
• Olympus and Veran Medical (2020)
• Ajax Health/Boston Scientific and Cortex (2024)
The GI market offers fertile ground for this approach. In 2024, an estimated 40 million GI endoscopic procedures were performed globally. Procedure volumes are projected to grow at a 2.9% CAGR through 2029. Most procedures remain diagnostic, but the real opportunity lies in enabling therapeutic interventions for complex conditions like inflammatory bowel disease (IBD), GI cancers, and GI defects.
Olympus already leads in endoscopic imaging and instruments. This move lets it expand deeper into procedural robotics, extend treatment capabilities, and fend off competitors like Johnson & Johnson, Medtronic, and Intuitive
Surgical. The strategic alignment, early-stage collaboration, and large market potential make this a signal deal for the future of endosurgical robotics.

August was a strong month for medtech financing, with multiple megarounds signaling healthy investor appetite for disruptive platforms. These rounds reflect a renewed willingness to back companies with breakthrough potential and regulatory momentum.
• HistoSonics, a University of Michigan spin-out behind non-invasive ultrasound therapy for tumors, was acquired in a management-led majority stake deal by a consortium including K5
Global, Bezos Expeditions, and Wellington Management, valuing the company at $2.25 billion.
• Heartflow, an AI-powered cardiac imaging company, completed its upsized IPO, raising $316 million. Heartflow’s shares surged as much as 47%, closing with a valuation near $2.27 billion.
• Alcon agreed to acquire STAAR Surgical and its implantable collamer lenses (ICLs) for vision correction for $28 per share in cash (a ~51% premium to its August 4th closing price), valuing the company at about $1.5 billion.
• Apreo Health raised $130 million in a Series B to advance its Breathe Airway Scaffold for emphysema. The round was backed by Bain Capital Life Sciences, Norwest Venture Partners, F-Prime, Intuitive Ventures, Lightstone Ventures, and Santé Ventures
• SetPoint Medical secured $140 million across its Series C and D rounds to support neuromodulation-based treatments for autoimmune diseases. Investors include Elevage Medical Technologies, Ally Bridge Group,
Northwell Health, SPRIG Equity, Abbott, Boston Scientific, and more.
• Orchestra BioMed raised $111 million in later-stage financing, including strategic transactions and public and private equity offerings, to support its hypertension and coronary disease programs. Notable investors include Ligand Pharmaceuticals and Medtronic.
After a subdued 2024, medtech IPOs are making a comeback in 2025. Six U.S. device companies have already gone public this year.
• Product: iLet Bionic Pancreas
• Raise: $234.6 million

Source: LSI Market Intelligence

• Day 1 pop: +39%
• Key watchpoint: pediatric expansion and manufacturing scale
• Product: Capsule endoscopy (GI)
• Raise: $27.5 million
• Day 1 movement: Flat
• Key watchpoint: commercial execution and AI pipeline
• Product: Personalized spine implants
• Raise: $100.5 million
• Day 1 movement: −3.3%
• Key watchpoint: surgeon adoption
• Product: Noninvasive AI-based coronary diagnostics
• Raise: $316.7 million
• Day 1 pop: +47–66%
• Key watchpoint: scale of AI integration and payer traction
• Product: Wearable for sudden cardiac arrest
• Raise: ~$202–232 million
• Day 1 pop: +28%
• Key watchpoint: adherence and post-acute partnerships
• Product: InSet shoulder arthroplasty
• Raise: $75 million
• Day 1 close: Slight decline
• Key watchpoint: competition with ortho strategics
Public markets are rewarding clear TAM stories, validated clinical data, and scalable economics. Hardware-intensive businesses are facing tighter valuations, while software-enabled, service-light models are outperforming. Overall, IPO momentum is building, and more listings are expected in Q4.
In one of the year’s largest disclosed medtech M&A transactions, Terumo announced it will acquire OrganOx for $1.5 billion. The move builds on Terumo’s long-term focus on cardiopulmonary and perfusion technologies and marks a significant bet on organ transplant innovation.
OrganOx develops normothermic machine perfusion (NMP) platforms that preserve donor livers at body temperature. Its flagship metra system is intended to improve organ viability and allows real-time functional assessment. From FY’23 to FY’24, OrganOx’s revenue grew from £12.4 million to £55.2 million, and the company moved from loss to profitability.
• Global organ preservation devices market size: $1.9 billion
• Main transplant drivers: Kidney and liver, representing 90% of global transplant volume
• U.S. waitlist: ~100,000 patients, including 89,800 for kidneys and 9,400 for livers
• Organ discard rates: ~30% for kidneys, ~10% for livers
• Cost per transplant prep: $30,000 to $100,000
Terumo joins a wave of strategics entering this space. Getinge acquired Paragonix Technologies in 2024 for $477 million. Terumo’s deal extends its portfolio from blood and cell processing, aortic and vascular grafts, and neurovascular devices into transplant viability.
OrganOx’s kidney preservation system is in development, with commercialization targeted for 2030. This acquisition deepens Terumo’s strength in perfusion, unlocks new markets, and aligns with its vision of improving physiologic support across the continuum of care. (Also see Intuitive Ventures cover story.)

This month, to accompany our Innovator Spotlight on Solenic Medical, we detail the latest LSI Market Intelligence data on the global, Asian, European, and U.S. forecast volume for knee replacement procedures.
This forecast accompanies our Market Dive: Diagnostic Electrophysiology Catheters, which starts on page 24.
Germany (000s)
LSI’s Global Surgical Procedure Volumes (SPV) Tracker provides trusted coverage of 300+ diagnostic and therapeutic procedures across 37 countries. Scan QR code to request a demo:

LSI’s SPV Tracker includes these geographies: Argentina, Australia, Belgium, Brazil, Canada, Caribbean, Chile, China, Colombia, Costa Rica, Denmark, Dominican Republic, Finland, France, Germany, Guatemala, India, Italy, Japan, Malaysia, Mexico, Netherlands, New Zealand, Norway, Panama, Poland, Russia, Singapore, South Africa, South Korea, Spain, Sweden, Switzerland, Thailand, Turkey, United Kingdom, and the United States.
accompanies our Market Dive: Peripheral Vascular Guidewires, which starts on
This global forecast accompanies our Market Dive: Femoral Closure Devices, that starts on page 23.
This month, to accompany our Innovator Spotlight on Solenic Medical, we detail the latest LSI Market Intelligence data on the global, Asian, European, and U.S. forecast volume for knee replacement procedures.2023 2024 2025 2026
and
LSI’s Global Surgical Procedure Volumes (SPV) Tracker provides trusted coverage of 300+ diagnostic and therapeutic procedures across 37 countries. Scan QR code to request a demo:

LSI’s SPV Tracker includes these geographies: Argentina, Australia, Belgium, Brazil, Canada, Caribbean, Chile, China, Colombia, Costa Rica, Denmark, Dominican Republic, Finland, France, Germany, Guatemala, India, Italy, Japan, Malaysia, Mexico, Netherlands, New Zealand, Norway, Panama, Poland, Russia, Singapore, South Africa, South Korea, Spain, Sweden, Switzerland, Thailand, Turkey, United Kingdom, and the United States.

Under the leadership of Co-Founder and CEO Evan Luxon and CBO Randy Preston, Centese is rewriting the playbook for recovery following heart and lung surgery. With more than 15,000 patients treated to date, Centese’s intelligent surgical recovery platform, Thoraguard, is accelerating recovery times, reducing complications, and reshaping the postoperative experience for cardiothoracic patients.
Centese originated inside San Francisco-based medical device incubator TheraNova, where Evan Luxon worked as Partner in new product development. While researching the clinical benefit of an automated urine output monitoring device, a chance conversation with cardiac surgeons sparked a new idea.
“They asked us if we could do something similar for chest drains,” Luxon recalled. “Those devices were even harder to manage, played an integral role in highly complex procedures, and hadn’t seen innovation in decades. That was the spark.”
TheraNova founder Dan Burnett, MD, introduced Luxon to Randy Preston, an experienced, commercially focused operator with experience in
cardiovascular markets. “Evan and I dove in, initially on nights and weekends,” said Preston. “We validated the clinical need, talked with leading surgeons from around the country, and explored whether innovating in this field could make a real impact in the lives of patients and healthcare more broadly.”
With early support from the National Science Foundation and non-dilutive state funding, Centese began to take
shape. Luxon returned to his hometown of Omaha to advance product development, set up pilot manufacturing, and grow the company, while Preston stayed in San Francisco to lead market development, clinical affairs, and commercial strategy.
Thoraguard replaces outdated plastic canisters and large rigid tubes with a digital platform that uses continuous monitoring of patient data and proprietary tubes to actively optimize fluid drainage with less pain.
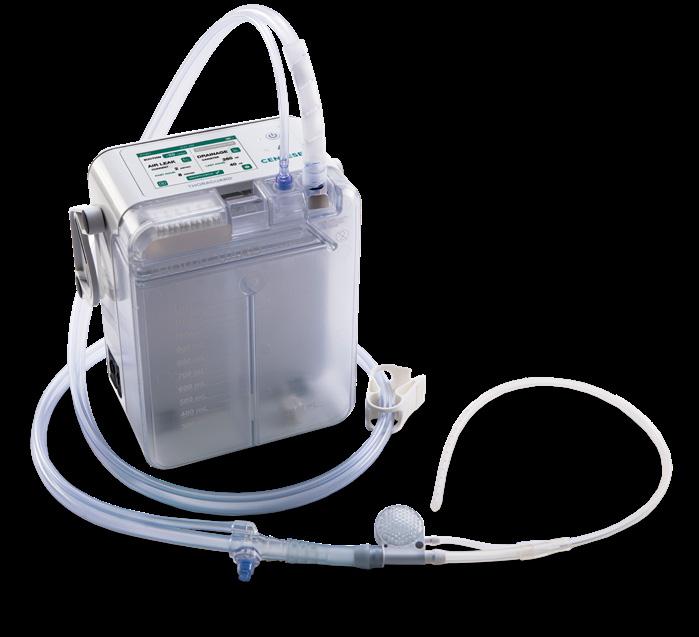
Source: Centese
Antiquated post-surgical fluid management undermines patient recovery
“It’s hard to overstate the clinical importance of drainage after surgery. It directly affects complications, pain, and how soon patients can go home.”
“It’s hard to overstate the clinical importance of drainage after surgery,” said Luxon. “It directly affects complications, pain, and how soon patients can go home.”
The system collects high-resolution data on post-operative bleeding and surgical site healing, giving surgeons real-time insights. If a complication occurs, it flags it faster. That visibility enables better decision-making, enhanced recovery protocols, and often earlier discharge with fewer residual complications.
“We’ve had clinicians take the length of stay after lung cancer surgery from five days down to under 24 hours,” said Preston. “Hospitals are reducing their costs by $15,000 per patient on these high-volume procedures, while patients are recovering faster and safer. It’s
Antiquated post-surgical fluid management undermines patient recovery
“Hospitals are reducing their
costs
by
$15,000 per patient on these high-volume procedures,
while patients are recovering faster and safer. It’s win-win, and that’s the kind of impact we aim for.”
win-win, and that’s the kind of impact we aim for.” Clinical outcomes have exceeded expectations, including a 40% reduction in post-operative atrial fibrillation in cardiac surgery patients.
Thoraguard is now used in over 40 hospitals across the United States, including leading centers such as NYU Langone, Stanford Health Care, Emory University, and the Mayo Clinic.
Thoraguard: The intelligent recovery platform enabling superior outcomes
Thoraguard: The intelligent recovery platform enabling superior outcomes
Key Limitations Key Features

Manual operation
Manual operation
Subjective assessments
Subjective assessments

Automated active drainage
Automated active drainage
Continuous digital monitoring
Continuous digital monitoring
Missed complications Increased complications
Foundational data platform
Foundational data platform
Resulting Impacts Resulting Benefits
Resulting Impacts Resulting Benefits
Missed complications Increased complications
Prolonged hospital stays
Prolonged hospital stays
Significant nursing burden
Significant nursing burden
Source: Centese
Reduced complications
Reduced complications
Shorter hospital stays
Shorter hospital stays
Reduced nursing burden
Reduced nursing burden
“We’ve seen record usage quarter over quarter,” said Preston. “And that growth is almost entirely driven by wordof-mouth from surgeons and nurses who see the impact firsthand.”
In a recent milestone, Centese signed a national Master Service Agreement with Kaiser Permanente, validating both its clinical and economic value at the highest level. “This agreement opens the door to bring the best care to a large consolidated healthcare system like Kaiser Permanente across the U.S., significantly expanding our opportunity to help patients,” said Luxon. “It’s a huge step forward.”
“One in three patients who undergo openheart surgery develops post-operative AFib. Our data shows we can reduce that by as much as 40%.”
To support scale, Centese has brought on key leadership, including new COO Hari Agarwal, to accelerate
production to high-volume contract manufacturing and Matt Kirby as VP of US Sales, to lead the go-to-market strategy and build out the company’s sales infrastructure.
As long-time LSI Alumni, Luxon and Preston credit the community for its lasting impact.
“LSI is a reunion of sorts,” said Luxon. “We see familiar faces, share real conversations, and always learn something new. Moonlight Chats last year were particularly memorable.”
For Preston, the value goes deeper.
“Medtech startups are really hard. It’s a long road to success,” he said. “LSI has become the center of the ecosystem. It’s one of the few places where you can swap war stories, get feedback, and connect with people who understand what we’re facing.”
“LSI has become the center of the [medtech] ecosystem.”
With the first close of a $6.5 million funding round completed and commercialization efforts accelerating, Centese is preparing for its next chapter. This includes regulatory expansion into Europe and the introduction of new features for Thoraguard, such as wireless connectivity and decision support through advanced data analysis to enable remote patient monitoring.
The company is laying the groundwork for future generations of its platform. The next generation of Thoraguard, expected in 2026, will incorporate additional AI-powered insights and remote monitoring to enable predictive analytics, earlier discharge, and even hospital-at-home applications. Looking further ahead to 2027 and beyond, the system will provide in-line diagnostics and personalized recovery by detecting biomarkers, such as infections and bleeding, to guide highly tailored care.
“We’re building something that helps patients, hospitals, and surgeons,” said Luxon. “And we’re doing it by listening, adapting, and staying focused on realworld impact.”
Centese will present its next phase of growth at LSI USA ‘26 in Dana Point, CA.

Evan Luxon and Randy Preston (Source: LSI USA ‘24)




Backed by 30 years of research and a world-first Nature publication, Heart Biotech is transforming the future of heart valve replacement with a regenerative scaffold that grows a living valve inside the body.
Under the leadership of CEO Francis White, Heart Biotech is developing the Yacoub Heart Valve, a groundbreaking solution designed to eliminate the compromises of today’s standard of care. Rather than implanting a mechanical or animal-derived valve, the company’s platform uses a resorbable scaffold that prompts the patient’s body to regenerate fully functional, native cardiac tissue in situ
Heart Biotech was founded in 2014 to translate decades of research led by legendary cardiothoracic surgeon, Prof. Sir Magdi Yacoub, into clinical reality.
Yacoub, who established the Magdi Yacoub Institute at Harefield Heart Science Centre, spent over 30 years exploring how to use tissue engineering to regrow living structures inside the human body.
“Sir Magdi had been working in science as well as surgery, which is quite rare for a surgeon, and he spent 30 years trying to crack the code,” said White. “The goal has always been to regrow living tissue inside a patient, rather than implanting metal or plastic that’s permanent and carries long-term risks and compromises.”
White joined the company in 2024, bringing decades of medtech leadership to a field he describes as transformative. “Most medical devices are
“The goal has always been to regrow living tissue inside a patient.”
compromises; they alleviate symptoms but don’t restore what’s lost,” he said. “This is fundamentally different. This is real repair.”
Today’s heart valve replacement options fall into two categories: mechanical valves, which require lifelong anticoagulation and lifestyle adjustments, and tissue valves, which don’t last beyond 10 to 15 years. Neither is
ideal, particularly for children with congenital heart disease who may face three or more open-heart surgeries before adulthood.
“A child may require surgery every two to three years just to keep pace with their growth,” said White. “It’s traumatic for families, costly for health systems, and inaccessible for much of the world.”
White believes the regenerative approach could make single-surgery solutions possible. This would represent a major leap forward in both care and equity. “If we can implant a small valve that regenerates and grows with the child, it’s one and done,” he said. “That’s life-changing.”
“If we can implant a small valve that regenerates and grows with the child, it’s one and done.”
Heart Biotech’s solution is a ready-touse, cell-free scaffold. Once implanted, it attracts local stem cells and triggers the regrowth of a new, living heart valve. Unlike competitors that rely on electrospinning or ex vivo cell seeding, Heart Biotech utilizes a proprietary jet-spraying method that produces a scaffold with heterogeneous fiber sizes that better mimics natural elastin and collagen.
“That biomimicry is the difference,” said White. “It attracts the right cells, in the right sequence, and enables them
to differentiate into all the tissues that form a valve.”
In 2023, the company published six-month preclinical data in Nature, demonstrating the regrowth of not only structural tissue but also nerve tissue, blood vessels, and adipose tissue.
“[The biomimicry] attracts the right cells in the right sequence, and enables them to differentiate into all the tissues that form a valve.”
Heart Biotech is preparing for a regulated GLP animal study to validate the technology under conditions that mimic human use. The team has finalized its manufacturing processes and is targeting first-in-human trials in 2028. The company is also pursuing Breakthrough Device Designation with the FDA.
Recent milestones include:
• Securing additional funding from high-net-worth individuals, including the company’s first SAFE agreement
• High level of activity, including due diligence, with venture capital firms and family offices from Europe, the U.S., and the Middle East
• A partnership with BMG Financial Group for representation in Saudi Arabia
• Application for EIS Advance Assurance
• Simplification of company structure through CARTA
Heart Biotech is currently raising a $15 million Series A round, which will fund completion of the GLP study and support regulatory engagement. The team has already raised $5 million in equity and secured $2 million in UKRI grants.
According to LSI data, Tetralogy of Fallot procedures, many of which include pulmonary valve replacement, are among the fastest-growing congenital heart defect surgeries performed globally, expected to reach 70,400 procedures by 2029. Heart Biotech is initially targeting pulmonary valve replacement in adult Ross patients and pediatric congenital heart patients, representing a $1.6 billion beachhead within a broader $37 billion global market that includes heart valves, vascular grafts, and cardiac patches.
As Heart Biotech continues advancing toward the clinic, White remains focused on execution, not hype. “We’re building something the world has never seen,” he said. “But we’re doing it by standing on the shoulders of three decades of work and listening to what nature already knows how to do.”

Source: Heart Biotech
Celebrating the accomplishments of a few of the many bright stars in our LSI presenting company and speaker alumni community.
Be sure to check out presentations from these and many other innovative companies from our recent LSI events, available exclusively in our Compass platform


Autonomix Medical has completed the final design review for its proprietary intravascular nerve-sensing catheter, a major milestone in preparation for human clinical trials. Engineered with a proprietary microchip-based sensing antenna array, the catheter is the first of its kind designed to detect and differentiate nerve signals in real time from inside the vasculature.
The company’s novel “sense, treat, and verify” approach enables real-time nerve mapping, precise ablation guidance, and immediate confirmation of treatment efficacy. Autonomix is initially targeting pancreatic cancer pain through an upcoming FDA IDE submission, with additional applications across cardiology and resistant hypertension, interventional pain management, and pulmonary and gastrointestinal disorders.
Berg Capital Partners has announced a strategic partnership with Simpson Interventions, a medtech innovator advancing treatments for coronary chronic total occlusions (CTOs). As Chief Financial Officer and Capital Markets Advisor, Berg will provide financial leadership and capital strategy support as Simpson scales its FDA-designated

Breakthrough Device, the Acolyte Image-Guided Crossing and Re-Entry Catheter System. The technology uses real-time optimal coherence tomography (OCT) to improve guidewire placement and revascularization.
Through this partnership, Berg Capital Partners will help optimize Simpson’s financial operations and lead fundraising efforts across its investor network, including venture capital, private equity,
and family office channels. The collaboration reflects Berg’s mission to support transformative medtech companies and highlights Simpson’s growing role in the minimally invasive cardiovascular space.

BMI OrganBank has secured FDA Breakthrough Device Designation for its novel room temperature machine perfusion (RTMP) system, a platform designed to assess and preserve kidneys for transplantation. Traditional cold storage methods often limit the ability to evaluate kidney function and contribute to unnecessary organ discard. BMI OrganBank’s portable RTMP device aims to overcome these challenges by enabling functional assessment at room temperature and extending preservation time.
Developed in collaboration with top transplant surgeons, the technology has the potential to increase the number of viable kidneys available for transplant and address persistent waitlist backlogs. The company plans to initiate clinical studies in early 2026. With facilities in Winston-Salem and Research Triangle Park, BMI OrganBank is also developing additional perfusion systems to transform organ and tissue preservation more broadly.

Ciliatech has received CE mark approval under the EU MDR for Intercil,
its nonpenetrating implant designed to treat glaucoma by increasing uveoscleral outflow. Unlike conventional approaches, Intercil achieves intraocular pressure (IOP) reduction without entering the anterior chamber of the eye, potentially minimizing the risk of corneal endothelial cell loss and simplifying post-operative care.
Labeled as the first cilioscleral interpositioning device (CID), Intercil features 360-degree positioning for optimal placement and suitability across multiple glaucoma subtypes, including primary open-angle and angle-closure glaucoma. With CE mark approval in hand, Ciliatech plans to launch commercial efforts in the European Union, expand into the UK and other global markets, and roll out a U.S. strategy backed by newly secured funding.

DeepSight Technology has received its first FDA 510(k) clearance for the NeedleVue LC1 Ultrasound System, a device designed to bring sub-millimeter precision and enhanced clarity to ultrasound-guided interventional procedures. The system is the first in a broader platform that includes DeepSight’s proprietary NeedleVue and OnPoint technologies, which aim to improve visualization, efficiency, and clinical confidence in procedures that rely on real-time imaging.
The clearance represents a major milestone for DeepSight as it moves toward commercial adoption and clinical expansion. The company showcased the technology at LSI Europe ‘25 in London and CIRSE 2025 in Barcelona. Founded in 2019, DeepSight is advancing a new era of procedural ultrasound with a platform that will span diagnostic, therapeutic, surgical, and interventional applications.

Egg Medical has announced the publication of new clinical data in JSCAI validating the performance of the EggNest Complete system. In a head-tohead comparison with the Rampart IC shielding system, EggNest not only reduced radiation exposure for primary operators and assistants but also offered significantly more protection for staff at the head of the bed and nursing positions, an advantage not observed with the Rampart IC system. The study reinforces Egg Medical’s goal of protecting all personnel in the cath lab while maintaining workflow and patient access.
Radiation exposure in interventional cardiology has long posed occupational risks, including cancer, cataracts, and musculoskeletal injuries from traditional lead aprons. This new data supports EggNest Complete as a safer, more comprehensive solution that aligns with the ALARA (As Low As Reasonably Achievable) principle. With increasing awareness of cumulative radiation exposure, the study highlights EggNest as a potential new standard in lab-wide protection for complex, x-rayguided procedures.

GT Medical Technologies has announced a major clinical milestone, with
more than 2,000 patients now treated using GammaTile, its FDA-cleared, bioabsorbable radiation therapy implant for operable brain tumors. The 2000th patient received treatment at Jersey Shore University Medical Center, a GammaTile Center of Excellence. Since its full U.S. market launch in March 2020, GammaTile has been adopted at over 200 leading cancer centers, hospitals, and institutions, reflecting growing demand for immediate, localized radiation at the time of tumor resection.
GammaTile is designed to eliminate the delay between surgery and radiation by delivering treatment directly at the resection margin, when residual cancer cell levels are at their lowest. This approach has gained traction among neurosurgeons and radiation oncologists aiming to improve local control while minimizing harm to healthy brain tissue. The milestone highlights GT Medical Technologies’ commitment to expanding access to innovative care for patients with operable brain tumors.

Interventional Systems and HICREN have received Class III Medical Device Registration approval from China’s National Medical Products Administration (NMPA) for their jointly developed robotic surgical platform, marking the first regulatory clearance for a miniature orthopedic surgical robot in China. The approval follows fast-track entry into the NMPA’s innovative medical device expedited review pathway last year and supports the upcoming commercial launch of the system in September. Developed through the companies’ joint venture, Sino-European MicroRobotics (SEMR), the system is commercially known as the Intelligent SpinePecker and is built on Interventional Systems’ Micromate technology platform.
The palm-sized robotic arm weighs just 1.63 kg and is housed in a compact
console occupying only 0.32 m² of space, with setup taking less than 10 minutes. The system delivers 0.2 mm positioning accuracy and allows for the placement of a single pedicle screw in approximately one minute. With intelligent algorithms that reduce surgical complexity and shorten the learning curve, the platform is positioned as a scalable solution. The companies anticipate strong demand in China and cite the approval as a major milestone in international collaboration and the evolution of micro-invasive surgical robotics.

Nalu Medical has unveiled its next-generation Therapy Disc, a smaller, wearable external power source for its battery-free, miniaturized implantable pulse generator (IPG). Designed for patients with chronic neuropathic pain, the Nalu neurostimulation system delivers targeted spinal cord and peripheral nerve stimulation. The new Therapy Disc is 39% smaller than its predecessor and works in tandem with a smartphone-based app to give patients more control. Despite its compact form, the Nalu system offers performance comparable to larger IPGs and has an expected service life of 18 years.
The latest update reinforces Nalu’s commitment to improving the patient experience while expanding access to peripheral nerve stimulation (PNS). Earlier this year, the company received FDA clearance for whole-body MRI compatibility of its PNS system, making it more versatile for patients with imaging needs.
Nyxoah has received FDA approval for its Genio system, a leadless hypoglossal nerve stimulation (HGNS) therapy

for patients with moderate to severe obstructive sleep apnea (OSA), with an Apnea-Hypopnea Index (AHI) of ≥15 and ≤ 65. Powered by a wearable component, Genio avoids implanted batteries and allows for future upgrades without surgery. In the DREAM pivotal trial, the system achieved a 63.5% AHI responder rate and showed strong efficacy regardless of sleeping position, a key differentiator in the OSA space.
With U.S. commercialization now underway, Nyxoah is offering a novel alternative for patients who have failed, refused, or are unable to tolerate positive airway pressure therapies, such as CPAP. The FDA milestone builds on Genio’s CE Mark, IPOs, and expanding indications, positioning the company to compete in a growing global sleep apnea market.

ONWARD Medical has secured FDA IDE approval to initiate Empower BP, a pivotal study evaluating its ARCIM neuromodulation system for managing blood pressure instability after spinal cord injury (SCI). The study will enroll patients with injuries at spinal cord levels C2–T6, injury severities of AIS A–D, and blood pressure instability across approximately 20 centers in the U.S., Canada, and Europe.
Empower BP marks the first pivotal evaluation of the fully implantable ARCIM platform, which includes the Onward Neurostimulator and ARCIM
thoracic lead. A first-in-human implant has already been performed in Switzerland, and further clinical feasibility studies are underway. With this IDE milestone, ONWARD continues its mission to restore autonomic function and improve the quality of life for people living with movement disabilities.

Plexāā has been selected to join Johnson & Johnson’s global incubator network, JLABS, the world’s largest open innovation ecosystem in healthcare. As a virtual member company, Plexāā will gain access to mentorship, strategic resources, and a powerful network across pharma and medtech to advance its mission of improving surgical outcomes through supraphysiological preconditioning. The company’s flagship device, BLOOM⁴³, is the world’s first fully wearable device worn next to the skin before breast surgery and has demonstrated potential to prevent wound healing complications in proofof-concept clinical trials.
Backed by a growing body of clinical data, Plexāā’s platform stimulates the release of Heat Shock Proteins to improve blood flow and promote healing. BLOOM⁴³ is initially focused on breast surgery, where wound complications affect one in three patients, and the company is actively exploring additional applications in orthopedics, obstetrics, and general and vascular surgery. With support from JLABS, Plexāā is moving closer to establishing supraphysiological preconditioning as a new standard of care in surgical recovery.
Pulse Biosciences has announced the publication of first-in-human feasibility study results for its nanosecond pulsed-field ablation (nsPFA) platform in the treatment of benign thyroid nodules. Conducted by Dr. Stefano Spiezia

at Ospedale del Mare in Naples, Italy, the study evaluated the company’s CellFX percutaneous electrode system, showing an average 86% nodule size reduction at one year and symptom improvement as early as two weeks post-procedure. The findings were published in the journal Thyroid.
The study reported no intranodular fibrosis or scarring on follow-up ultrasounds, highlighting the platform’s favorable safety profile. These results support Pulse Biosciences’ growing body of evidence that PFA may expand beyond cardiac indications into soft tissue applications with significant unmet need.

SpinaFX has received FDA Breakthrough Device Designation for Triojection, its flagship image-guided, minimally invasive treatment for contained lumbar disc herniations. The procedure delivers a proprietary oxygen-ozone mixture into the disc to relieve pressure and nerve compression, offering a cost-effective outpatient alternative to invasive surgery. The designation acknowledges Triojection’s potential to improve recovery time, reduce complications, and expand access to effective care, especially in under-resourced settings.
With FDA engagement now accelerated, SpinaFX aims to bring Triojection
to a global patient population suffering from chronic pain and disability caused by disc herniation. The company is also exploring expanded indications and applications for its platform in other disc-related conditions and musculoskeletal disorders.

SurgiBox has received CE Mark certification under MDR for its SurgiField System devices, a milestone that enables commercial launch across Europe and key global markets. The ultraportable system integrates a sterile barrier (SurgiBubble) and a previously approved electronic filtration system to create a safe surgical environment outside of traditional ORs.
Designed for use in both high- and low-resource settings, the system protects patients and providers from cross-contamination with bodily fluids while expanding surgical access at the bedside, in ambulatory centers, or even in field conditions.
This approval follows years of development, testing, and collaboration with clinicians worldwide, and positions SurgiBox to meet rising demand for flexible surgical solutions. With an estimated five billion people chronically lacking consistent access to safe surgery, and growing logistical challenges in existing systems, the company aims to transform how and where surgery happens. Global sales have begun, and SurgiBox is scaling operations to fulfill a growing pipeline while raising funds to support its next phase of impact.
A
Abbott 14, 25, 29–31
Abbott Vascular .................................... 29
Action Potential Venture Capital 14
AIT Studio 14
Ajax Health 30
Alcon 6, 14, 31
Allegis Capital ........................................ 14
Ally Bridge Group 14, 31
Alpha JWC Ventures 14
Alpha Wave Ventures ........................... 14
Amber Therapeutics 21
American Heart Association 20
Amirit Oshri-Chai 12
aMoon Fund 15
Amsel Medical ....................................... 10
Amzak Health 14
Andera Partners 10
Andy Rasdal .......................................... 13
Angel Co-Fund 15
Angiotech Pharmaceuticals 13
Apreo Health 6, 14, 17, 20, 31
Arboretum Ventures 15
Arterial Vascular Engineering ............ 13
Arterys 20
ASAHI INTECC 28, 29
Ascendum Capital ................................ 14
Ascensia Diabetes Care 15
Ashley Friedman 8
Auris Health 20
Autonomix Medical 44
AVASA ...................................................... 15
Axonics 10
B
Bain Capital Life Sciences ..... 14, 20, 31
Bas Nieuwenhuis 13
BD Kiestra 13
Ben Burnham .......................................... 11
Berg Capital Partners 44
Bert Wiggers 13
Beta Bionics 31
Better Medicine 15
Bezos Expeditions .......................... 14, 31
Bill Fender 12
Bill Hunter, MD 13
Biomet ..................................................... 13
Biosense Webster 26
BioVentrix 11
BiVACOR
K
K5 Global .......................................... 14, 31
Kaiser Permanente 40
Kardium .................................................. 26
Keith Jeffcoat 12
Kelly Perez .............................................. 13
Kestra Medical Technologies 10, 32
Keyron ................................................ 11, 12
Knobbe Martens 17
Korea Investment Accelerator ........... 14
L
Labcorp Drug Development 12
Lauxera Capital 19
L&G 14
Ligand Pharmaceuticals 14, 31
Lightrock 14
Lightstone Ventures 14, 15, 20, 31
Lumira Ventures 14
Luna Diabetes 15 Luxtera 13
M
Magdi Yacoub 42
Magdi Yacoub Institute at Harefield Heart Science Centre 42
Maggie Nixon........................................ 20
Maj Invest 15
Mark Brister ............................................ 13
Massimiliano Colella 11
Matt Kirby............................................... 40 Matt Likens 12
Mayo Clinic ............................................ 39 McKesson 17, 20
MedCrypt ................................................ 21
MedtechWOMEN 21
Medtronic ...................... 12–14, 26, 28–31
Mercia 20
Merz Pharma.......................................... 12
Method AI 15
Midas Capital ......................................... 14
Mike Teodorescu 47
Morgenthaler Ventures 14
Movac 15
Murielle Thinard McLane 5, 6, 16–21
MVM Partners 15 MYSC 14 N
Nalu Medical 10, 46 NanoDrops 8 National Science Foundation 38 Neotract 8
Neurolief 15
Neurovalens 15
New Enterprise Associates 14
Nooma Bio 17
Northwell Health 14, 31
Norwest ...................................... 14, 20, 31 Novartis 17, 20
Nurami Medical ..................................... 12
NYU Langone 39
Nyxoah ................................................... 46
O
Obalon Therapeutics ........................... 13 Olivier Benoit 45 Olivier Taelman ..................................... 46
17, 20 OrbiMed 15
Orchestra BioMed 14, 31
OrganOx 15, 17, 19, 21, 32
Ospedale del Mare 47 Oxford Science Enterprises ............... 14 Oxford University .................................. 14
Paige ........................................................ 15
Paragonix Technologies 32 Partners Group 15 Paul Kirchgraber 12 Paul LaViolette 8, 47 Pedro Costa 46 Per Langoe 45 Planatome 12 Plexāā 47 Preci-Health


June 23rd–26th, 2026
Shangri-La Singapore Hotel
With the global community built through LSI USA and LSI Europe and our partnerships in Singapore, China, Korea, Japan, and more, LSI Asia will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Like LSI USA and LSI Europe, LSI Asia will not be held in a chaotic convention center. We have identified a world-class venue in Singapore, the gateway to Southeast Asia, for our second LSI Asia event, where senior executives can do real business.


