Leading Medtech Innovation at Scale: Inside Lisa Earnhardt’s Vision for Abbott

Medtech Pulse Check, Alumni Innovator Spotlight, Drug Delivery Devices, and much more...

Leading Medtech Innovation at Scale: Inside Lisa Earnhardt’s Vision for Abbott

Medtech Pulse Check, Alumni Innovator Spotlight, Drug Delivery Devices, and much more...
February 2025
May 2025
June 10th - 13th, 2025
The Ritz-Carlton, Millenia Singapore
With the global community built through LSI USA and LSI Europe and our partnerships in Singapore, China, Korea, Japan and more, LSI Asia will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Like LSI USA and LSI Europe, LSI Asia will not be held in a chaotic convention center. We have identified a world-class venue in Singapore, the gateway to Southeast Asia, for our inaugural event where senior executives can do real business.


Thank you to our sponsors



















Scott Pantel
Chief Executive Officer, Editor-in-Chief

Rebekah Murrietta
Vice President of Media, Contributing Author

Benny Tomlin Contributing Photographer

Henry Peck
Chief Business Officer, Editor-in-Chief

Blake Matrone Sr. Marketing Manager, Contributing Author

Kristin Karkoska Contributing Photographer

Tracy Schaaf
Managing Editor and Content Strategist, Lead Author

Gavin Kennedy Business Development, Contributing Author

Paul Streeto Graphic Designer

Brenna Hopkins Sr. Content Manager, Lead Author

Almonte Customer Service & Fulfillment

Nicholas Talamantes
Sr. Director of Market Intelligence, Contributing Author

Kelly Williams
Subscriptions & Enterprise Sales
714 847 3540 tel/fax email: info@ls-intel.com
Vol. 2, No. 5 May 2025
Created by LSI, The Lens leverages LSI’s deep industry relationships and proprietary market intelligence to help executives like you build lasting medtech companies. Start your Individual, Group or Enterprise-Wide subscription today!

Scan QR Code to activate your subscription.
The Lens is published monthly and available in print and electronic formats. Copyright 2025 by Life Science Intelligence, Inc. All rights reserved. Editor takes care to report information from reliable sources and does not assume liability for information published. Interested in subscribing? email to: info@ls-intel.com
Interested in advertising? email to: kelly@ls-intel.com


Dear Readers,
Momentum is building. Across boardrooms and operating rooms, grand rounds and investment rounds, we’re seeing a surge of activity in medtech. It’s a moment shaped by scale, scarcity, and the rising urgency to innovate with purpose. And what we’re hearing from leaders, investors, and entrepreneurs is clear: the bar is higher, and so is the opportunity.
Few captured that more directly than Lisa Earnhardt, EVP and Group President of Medical Devices at Abbott, during her keynote at LSI USA ’25. Earnhardt leads nearly $20 billion in global medtech business, and she shared insights with the hard-earned clarity of a startup founder. She reminded us that at any scale, innovation is still personal. It starts with the patient, and it’s powered by staying close to the problem, focused on what matters, and brave enough to do what hasn’t been done before.
That ethos comes to life across this edition of The Lens.
We spotlight Earnhardt’s perspective on servant leadership, transformative product launches, and why “collaboration over competition” is the new winning strategy in medtech. From diabetes to heart failure to neuromodulation, Abbott is pushing healthcare closer to home and reimagining what it means to lead at scale.
We also take a hard look at the investment landscape in this edition. Q1 2025 marked the strongest quarter in three years for medical device venture funding, with $2.6 billion deployed across 132 deals. However, as Jonathan Norris of HSBC Innovation Banking points out, this momentum comes with caveats. Behind the dollars are deeper questions about sustainability, capital efficiency, and navigating an uncertain policy and regulatory environment. Big rounds are still happening, but with precision rather than abundance.
We also highlight exciting emerging breakthroughs, one of which comes from Fluid Biomed. The startup has developed a first-of-its-kind bioabsorbable brain stent designed to treat aneurysms and then disappear. It’s a bold rethinking of how we use implants, and a reminder that the best innovations often begin with deep frustration, relentless iteration, and a refusal to accept “good enough.”
These stories are more than case studies—they’re signals. Across the global medtech ecosystem, we’re seeing bold ideas and bullish support. We’re seeing a demand for clarity, strategy, and above all, impact.
So, as you read this edition, take a moment to reflect not just on what’s happening, but what’s possible. Where do you see the most urgent need? What’s your role in meeting it? And what’s the next step you’re ready to take?
We’re honored to be on this journey with you.
All the best, Scott Pantel & Henry Peck
Scott Pantel Chief Executive Officer, LSI Editor-in-Chief


Henry Peck Chief Business Officer, LSI Editor-in-Chief

Need fresh inspiration for your medtech company as we navigate Q225? We’ve gathered insights from some of our LSI alumni thought leaders to help you stay ahead.
“Not all ideas are worth pursuing, and I think we need to be really honest with ourselves about when something is not worth pursuing.”

— Ashley Seehusen, Venture Partner, Santé Ventures, CEO, Santé Accel
LSI Europe ‘24 Panel: Training & Supporting the Next Wave of Entrepreneurs Through Mentorship and Capital
“I believe patients want to be guided. I don’t think patients want to take ownership.”

— Thom Rasche, Managing Partner, Earlybird Health
LSI Europe ‘24 Panel: Consumerization in Medtech: Understanding Patient Behavior to Drive Real Value
“Medtech is hard. It’s really hard. If you’re interested in making big dollars, go to tech; it’s easier, and that’s hard, too. But I think the big thing that differentiates medtech from tech is the mission— the opportunity to have a real significant impact.”

— Todd Brinton, Chief Science Officer & Corporate VP, Edwards Lifesciences
LSI Europe ‘24 Panel: Behind the Scenes in Unlocking Disruptive Innovation
“Neuromodulation can treat depression better than drugs by mapping individual brain circuits associated with specific symptoms.”

Ana Maiques, CEO, Neuroelectrics
LSI Europe ‘24 Panel: Neuromodulation: Hurdles, Ethics, and Unmet Needs Driving Innovation and Investment
“Healthcare is a team sport, and we need to think more expansively about how we partner.”

— Lisa Earnhardt, EVP and Group President, Medical Devices, Abbott
LSI USA ‘25: Keynote with Lisa Earnhardt, EVP & Group President, Medical Devices at Abbott
“You want your investors to help you grow, not obstruct—but that’s not always the reality. Navigating complex investor conversations takes grit and belief in your vision.”

“You have to get rid of the weak people and promote the strong people.”

— Joe Kiani, Founder, Masimo, Founder & Executive Chairman, Willow Labs
LSI USA ‘25: Keynote w/ Joe Kiani, the Founder, Chairman, CEO of Willow Labs, and Founder of Masimo
— David Cubbin, CEO, Cure Capital Advisors
LSI Europe ‘24 Panel: Caresyntax: $180M to Accelerate the Future of AI and Precision Surgery
“You can’t wait for 100% of the information to make a decision.”

— Howard Levin, CEO & CMO, Deerfield Catalyst
LSI Europe ‘24 Panel: Behind the Scenes in Unlocking Disruptive Innovation
Celebrating recent leadership shifts and other announcements and accolades impacting our global medtech community.

Append Medical has appointed Shlomi Nachman as Chairman of the Board, reinforcing its commitment to transforming stroke prevention in atrial fibrillation patients. Nachman brings more than 25 years of medtech leadership, including senior roles at Johnson & Johnson, where he served as Company Group Chairman of Cardiovascular and Specialty Solutions and Vision Groups. He led global teams across electrophysiology, neurovascular, and cardiovascular portfolios—driving innovation, commercial success, and expansion into new markets. His appointment signals a pivotal phase for Append Medical as it scales its implantfree, vacuum-based left atrial appendage (LAA) occlusion technology.
CEO Nadav Agian highlighted Nachman’s “proven track record in driving commercial success and fostering innovation,” and noted his unique qualifications to help guide the company’s next stage of growth. Nachman shared his enthusiasm for the role, calling Append’s approach “disruptive technology” with the potential to significantly improve patient care. As Chairman, he will support the company’s strategy to eliminate device-related risks like thromboembolism and peri-device leakage and bring safer, more effective stroke prevention to patients worldwide.
Arsenal Medical has appointed Dr. Ray Baker to its Board of Directors, bringing over 35 years of interventional pain management expertise and executive leadership to support the company’s next phase of growth. Dr. Baker currently serves as CMO of

Neuromodulation at Boston Scientific and was previously CMO at Relievant Medical Systems, where he helped commercialize the Intracept procedure—a minimally invasive treatment for chronic low back pain due to degenerative disc disease. His career spans both clinical and operational leadership, with past roles as VP and Executive Medical Director at Evergreen Health and as Director of Interventional Pain Services at the University of Washington. A respected leader in policy and professional society engagement, Dr. Baker has chaired the CMS Multi-Society Pain Workgroup and held presidential roles at both the North American Spine Society and the International Pain & Spine Intervention Society.
His appointment comes as Arsenal advances its lead biomaterials-based products, NeoCast and ResQFoam, in the clinic and expands into new therapeutic areas such as neurointervention. With a career rooted in innovation and advocacy for minimally invasive care, Dr. Baker is well-positioned to help Arsenal transform how biomaterials are used to improve patient outcomes. “I am looking forward to joining Arsenal’s board during this exciting time for the company,” said Baker, emphasizing the potential for advanced materials to enhance both physician workflows and therapeutic efficacy.
CorWave has appointed Dawn Sadlowski-Buisserez as Vice President of Operations, bringing with her three decades of industrial leadership spanning medical devices, automotive manufacturing, and fiber optics.

Sadlowski-Buisserez most recently served as Director of Operations at the Sorin cardiac rhythm management facility in Clamart, France—one of Europe’s largest cleanrooms for active implantable devices—where she oversaw the production of pacemakers and implantable defibrillators. Following Sorin’s acquisition by MicroPort, she led key strategic initiatives, including duplicating a product line in China and navigating complex MDR re-approval processes. Her background includes global industrialization roles at Alcatel and continuous improvement leadership at Lear Corporation
At CorWave, she succeeds Michael Webb and will play a central role in scaling the company’s next-generation left ventricular assist device (LVAD) manufacturing ahead of clinical and commercial milestones. CEO Louis de Lillers called her “one of the few people in Europe with such in-depth knowledge of large-scale manufacturing of active implantable medical devices” and a reflection of the company’s transatlantic identity. With CorWave’s state-of-the-art urban factory in Clichy already producing close to 100 devices in validation testing, Sadlowski-Buisserez will lead the ramp-up of operations at a facility designed for €100 million in annual revenue, with the capacity to scale tenfold as demand grows.
FineHeart has appointed Sébastien Robitaille as Chief Financial Officer to guide the company through its next stage of commercial and industrial expansion. Robitaille brings more than 25 years of experience in

pharmaceuticals and biotechnology, with a track record that spans business transformation, corporate finance, and international growth. He has held leadership roles across both public and private companies, helping scale operations and secure capital on major exchanges, including Euronext and Nasdaq. Known for his strategic vision and financial acumen, Robitaille is well-positioned to help FineHeart unlock its full market potential as it advances the FLOWMAKER solution for patients with advanced heart failure.
In its announcement, FineHeart emphasized Robitaille’s role in supporting the company’s scale-up journey and its mission to address a critical unmet need in cardiac care. FLOWMAKER, the company’s next-generation fully implantable cardiac assist device, is designed to restore natural heart function without external components or driveline infections—an innovation that could redefine the standard of care for heart failure. Robitaille’s appointment reinforces FineHeart’s commitment to global growth, with his expertise expected to play a pivotal role in shaping strategic partnerships, commercial readiness, and long-term financial sustainability.

iCAD has appointed LSI alumni Mark Koeniguer as Chief Commercial Officer to lead the company’s global revenue growth strategy and expand adoption of its AI-powered breast health solutions. Koeniguer brings more than 30 years of executive leadership in
medical imaging, AI, and digital health, with a career spanning GE HealthCare, Volpara Health, and Stereotaxis. Most recently, he served as CEO of ScreenPoint Medical, where he transformed the company into a global commercial leader, launched two FDA-cleared products, and drove a 400% increase in recurring revenue. At iCAD, Koeniguer will oversee sales, partnerships, operations, and customer success, playing a pivotal role in shaping the next phase of commercial acceleration for the company’s ProFound Breast Health Suite.
In welcoming Koeniguer, iCAD emphasized his deep technical understanding of imaging workflows, consultative leadership style, and extensive experience across the breast diagnostics ecosystem. President and CEO Dana Brown described him as “an ideal leader for this new role,” while COO Michelle Strong highlighted his potential to bring iCAD’s mission—to create a world where cancer can’t hide—to more patients worldwide. With more than 40 million mammograms read using its technology in the past five years, iCAD is well-positioned for growth, and Koeniguer’s appointment reinforces its focus on global engagement, enterprise-wide adoption, and improved outcomes in breast cancer detection.

Minze Health has appointed Steffen Hovard as Executive Chairman of the Board, bringing more than 25 years of leadership in urology, medical devices, and digital health to the company. Hovard’s career includes executive roles such as CEO of Neuspera Medical, Chairman of UroMems, and President of Coloplast Interventional Urology, where he helped scale groundbreaking solutions in continence care. He currently serves on the boards of Butterfly Medical and Relief, reinforcing his continued influence in advancing patient-centric innovation across the urology landscape. At Minze, he will
play a critical role in scaling the company’s home-based digital solutions and supporting strategic initiatives in global expansion, commercialization, and regulatory execution.
In announcing the appointment, Minze Health highlighted Hovard’s deep expertise in guiding companies through growth and product adoption phases. His leadership is expected to bolster the company’s efforts to transform the diagnosis and treatment of urinary conditions through tools like the Minze Homeflow and Minze Diary Pod— solutions designed to support remote monitoring and personalized care for conditions such as benign prostatic hyperplasia (BPH) and overactive bladder (OAB). His appointment signals a strong commitment to global scale and clinical impact in the rapidly growing digital urology space.

Medical has appointed Raymond Cohen as Chairman of its Board of Directors, adding one of the medtech industry’s most accomplished leaders to guide its next stage of growth. Cohen co-founded Axonics, where he served as CEO until its $3.7 billion acquisition by Boston Scientific in 2024. Under his leadership, Axonics went public in 2018 and became one of the fastest-growing medtech companies in the Americas, earning the top spot on both the Deloitte Technology Fast 500 and Financial Times’ rankings for two consecutive years. Cohen also led the $600 million sale of SoniVie to Boston Scientific (announced in March 2025) and currently sits on the board of Kestra Medical Technologies, which recently completed a $232 million IPO.
Cohen brings over 40 years of executive and board-level experience in life sciences and was named the 2024 MedTech MVP and Orange County Business Journal’s Businessperson of the Year. His extensive track record in neuromodulation, commercialization, and public markets is expected
to accelerate the adoption of Nalu’s miniaturized neurostimulation system for chronic neuropathic pain. “We look forward to benefiting from Mr. Cohen’s broad experience in growing medtech companies,” said Nalu CEO Tom West, underscoring Cohen’s fit to help scale the company’s FDA-cleared peripheral nerve stimulation (PNS) and spinal cord stimulation (SCS) platform. As Nalu continues expanding market reach for the Nalu micro-IPG, Cohen’s leadership signals a new phase of strategic momentum. (Stay tuned for his cover story next month here in The Lens.)

NanoDrops has appointed Daria Lemann-Blumenthal as Chief Executive Officer, bringing two decades of ophthalmology leadership and a recent $466 million acquisition success to the helm. Lemann-Blumenthal co-founded and led BELKIN Vision, the company behind Direct Selective Laser Trabeculoplasty (DSLT), a breakthrough glaucoma treatment that streamlined laser therapy into a single, non-contact procedure. Under her leadership, BELKIN Vision was acquired by global eye care leader Alcon in 2024—a strategic deal that included upfront and sales-based milestone payments. At NanoDrops, she will guide the company’s next phase of clinical development and industry engagement, with a focus on advancing its nanoparticle-based, minimally invasive vision correction platform for presbyopia and myopia.
In announcing her appointment, NanoDrops highlighted Lemann-Blumenthal’s rare blend of regulatory, clinical, and commercialization expertise. She joins co-founder and CMO Prof. David Smadja, MD—an internationally recognized figure in refractive surgery— to form a leadership team uniquely equipped to drive bio-convergent innovation in eye care. “Daria’s exceptional leadership and proven ability to drive
strategic growth were demonstrated in her recent success,” said medical advisory board chair, Assoc. Prof. Arthur Cummings, MD. With Lemann-Blumenthal now at the helm, NanoDrops is poised to enter human clinical trials and shape a new model for personalized, adjustable vision correction.

Nia Therapeutics has appointed Prof. Michael Kahana as Chief Executive Officer, marking a major step forward in the company’s mission to develop closed-loop neurostimulation therapies for memory loss. A world-renowned expert in the science of human memory, Kahana previously served as Director of the Computational Memory Lab at the University of Pennsylvania and authored over 230 peer-reviewed publications, as well as the landmark textbook Foundations of Human Memory. His pioneering research on memory encoding, theta oscillations, and spatial and temporal coding in the brain laid the scientific foundation for Nia’s therapy. Among his many accolades, Kahana has received the Troland Award from the National Academy of Sciences and the Howard Crosby Warren Medal from the Society of Experimental Psychologists.
Kahana joins Nia full-time after co-authoring seminal studies alongside co-founder Dr. Dan Rizzuto and Dr. Youssef Ezzyat, which demonstrated the efficacy of closed-loop brain stimulation in enhancing memory. Conducted under DARPA’s Restoring Active Memory (RAM) program, this work produced the world’s largest dataset on AI-based memory decoding and neural modulation. Now at the helm of Nia, Kahana will lead efforts to translate this foundational research into clinical applications for patients with memory impairment due to traumatic brain injury (TBI) and other neurological disorders.
Novadip Biosciences has appointed Soumya Chandramouli as Chief Financial Officer, reinforcing the company’s

leadership team ahead of its €50 million Series C raise and planned commercial launch in 2027. Chandramouli brings 25 years of experience in international financial management, most recently serving as CFO of IBA Group, a €500 million revenue medtech company operating across North America and EMEA. At IBA, she led a team of over 50 and played a key role in a €93 million fundraising for a Belgian life sciences spin-off. Chandramouli’s deep expertise in private and public capital markets, coupled with her operational command of globally deployed organizations, positions her to steer Novadip’s financial strategy through late-stage clinical development and commercialization.
Chandramouli joins as Novadip prepares to launch pivotal Phase 3 trials for NVD-003—its autologous therapy for rare pediatric bone disorders such as congenital pseudarthrosis of the tibia— and a Phase 2b/3 trial for NVDX3, an allogenic product targeting spine fusion and trauma indications. CEO Denis Dufrane welcomed her appointment as a strategic asset in the company’s transition from clinical to commercial operations. With a seasoned executive team now in place, Novadip is well-positioned to build momentum toward market entry and long-term growth in its $13.5 billion target market.

Phagenesis has appointed Marc Korenberg as Chief Financial Officer, adding a seasoned finance leader with over 15 years of experience in
biopharma and medtech to its executive team. Korenberg has guided multiple companies through transitions from clinical development to commercial scale, bringing a strategic focus on enterprise-wide financial alignment and operational growth. Known for his people-first leadership style and cross-functional approach, he joins Phagenesis at a pivotal moment as it accelerates the adoption of Phagenyx, its non-invasive therapy for treating dysphagia through pharyngeal electrical stimulation (PES).
The company welcomed Korenberg as a strong cultural and strategic fit for its next stage of growth. With rigorous clinical validation and expanding global recognition, Phagenyx is redefining standards in the treatment of swallowing disorders, offering new hope to millions living with dysphagia. Korenberg’s appointment demonstrates Phagenesis’ readiness to scale its impact and operations, building on its commitment to innovation, patient outcomes, and commercial excellence in a field long underserved by therapeutic technologies.

Spiro Robotics has appointed Trae Gordon as Chief Commercial Officer, a strategic hire as the company moves from clinical validation to commercialization of its image-guided platform for difficult airway management. Gordon brings two decades of experience scaling emerging medtech technologies and driving go-to-market strategies in complex healthcare environments. His appointment follows a successful firstin-human clinical trial of Spiro-VISTA, a next-generation handheld intubation system designed to provide dual-camera visualization, servo-controlled navigation, and real-time feedback, specifically engineered to address the high-risk scenarios where conventional
tools often fail. Gordon will lead investor engagement and commercial planning as the company advances toward FDA 510(k) submission and a targeted early 2026 market launch.
The company described Gordon’s addition as a pivotal step in “building the commercial muscle” needed to scale operations and reach its ambitious goals. CEO Dimitri Sokolov praised Gordon’s track record and leadership, highlighting his role in bridging engineering-led development with commercial execution. With more than 30 million intubations performed annually in the U.S., and over six million considered difficult, Spiro’s technology aims to redefine a critical aspect of emergency and surgical care. Gordon joins a team led by both technical and clinical founders, working to deliver a platform that not only elevates airway management standards but also lays the groundwork for AI-guided automation in the near future. (See the February 8, 2025 edition of LSI’s The Memo featuring Spiro Robotics, on the LSI website at Resources > Research Desk > The Memo.)


Big deals, even bigger questions. Jonathan Norris of HSBC Innovation Banking explains why 2025 is already one for the books.

Venture investment across the medical device sector kicked off 2025 with a bang, posting $2.6 billion in first-quarter funding across 132 deals—the highest quarterly dollar mark in three years. According to Jonathan Norris, Managing Director at HSBC Innovation Banking, who writes comprehensive reports on the venture healthcare ecosystem, this surge marks a 20% rise in both capital and deal count compared to the 2024 average.
“Not only did we have more dollars, but there were more deals, which is really good to see,” Norris says, in a recent interview with The Lens. “I think the quick and easy takeaway is that more companies were getting funded in Q1, and a good percentage of the bigger deals in the quarter were PMA pivotal trial rounds. It looks like positive momentum for the sector to see a big quarter. That’s our 35,000-foot view.”
But beneath the celebratory surface lies a market under tension, and one that is bracing for impact with headwinds including controversial federal policies and regulatory changes, and a closed IPO window. However, despite this dynamic environment, resilient medtech companies are being funded and continue to develop life-saving technologies, and selective, strategic investors have funds available to invest in the most compelling device opportunities.
Here, we break down the Q1 medical device venture investment picture, including overall volume, the top venture deals of $50 million or more, and first financings, in what’s sure to be a year of reckoning in medtech.
From January to March of this year, while overall investment volume surged, in Norris’ view, the structure of the deals reflects an ongoing trend: bigger rounds, bigger syndicates, and shared risk.
Source: Jonathan Norris, Managing Director, HSBC Innovation Banking
During the quarter, 16 venture deals of $50 million or more accounted for $1.3 billion in funding—half of the quarter’s total (see pie chart). But no single deal dwarfed the field. The largest, at $142 million (organ tissue preservation company OrganOx), announced in February, was notable but not outsized compared to biotech, where deals north of $300 million aren’t uncommon.
“The top 10% of deals taking 50% of the dollars is a trend we had been seeing in 2024. I don’t think it’s necessarily new, but it’s a continuation of the bigger deals taking the majority of the capital that’s out here. It signals a continued investor strategy of caution,” Norris explains.
Investors are continuing to opt for a “safety in numbers” approach, building multiple syndicates with enough dry powder to weather potentially turbulent next rounds, notes Norris. If no new investor is found, the strategy is that a company could have enough capital from its pool of existing investors to stay afloat with an inside round if needed.
activity in 2024 and is a consistent source of strategic interest,” says Norris.
And this strategic interest is reflected in the capital being raised. Among the many deals that the LSI team tracks in Compass, LSI’s proprietary market intelligence solution for competitive intelligence, deal-flow analysis, and opportunity assessment, nearly every single one can find a corollary with recent M&A activity in the cardiovascular devices market. Put simply, people are doubling down on the bets that companies like Boston Scientific, Edwards Lifesciences, and Johnson & Johnson are making.
For good reason too. These are device markets, within the greater $62.5B cardiovascular devices market, that are seeing growth at between 2x to 6x the compound annual growth of the worldwide market for cardiovascular devices. For example, according to recent forecasts from LSI, pulsed field ablation (PFA), transcatheter mitral valves, and left ventricular assist devices (LVADs) are projected to increase at a CAGR of 32.8%, 22.0%, and 10.9%, respectively. (For more information on LSI’s extensive Market Intelligence offerings, visit the LSI website.)
Non-invasive monitoring (NIM) is another segment that stood out in Q1, according to Norris, with $419 million in 18 deals. For all of 2024, 54 NIM deals totaled approximately $1 billion.
PAUL - For a bar chart we’ll include here, please start with the 2024 Med Device All Deals table that you created in January, noted below, including these elements:
Title: Med Device Venture Investment Deals, 2024 vs. Q1 2025 Subhead above the bar chart: Venture Investment Activity ($/Deals)
In terms of overall Q1 device venture investments by indication, there were two segments that were either way ahead of pace or ahead of pace versus 2024 investment, according to Norris. Way ahead of pace is cardiovascular, with $500 million already invested in 16 deals. In all of 2024, 25 cardiovascular device deals secured funding totaling $542 million.
Once again, the investment is sound. As far as risks go, these devices typically have a lower risk classification, facing reduced clinical and regulatory barriers. This means that these products can validate commercial viability far more quickly than complex medical devices. In today’s M&A market, an established commercial model is often a prerequisite.
Below this please include the 2024 bar chart (circled below), showing each quarter and the 2024 total above it, then a dotted vertical line, then show a bar for each month of Q1 2025: $913 million in January, $790 million in February, and $882 million in March. Then please include the Q1 2025 TOTAL above, $2.6B, and 133 DEALS (note that for now I don’t have the # of deals by quarter, only the total - I will ask Jonathan Norris when he returns from vacation on 5/19)
Source: Jonathan Norris, HSBC Innovation Banking (data used with permission)
“Cardiovascular medical device investment in Q1 hit almost the entire dollar amount that was invested in the sector in all of 2024. I think that’s very interesting, and inherently makes sense, as the cardiovascular space had the most private M&A
Furthermore, the NIM market continues to be one seeing rapid growth. According to LSI’s Compass, the market for remote diagnostic and monitoring solutions will nearly double in the next five years, as the market grows at a CAGR of 11.4% from $26.2 billion to $45.7 billion.
With regard to total device venture investment by month, Norris notes that there was a fairly even distribution. In January, deals totaled $913 million, then a small dip to $790 million Source: Jonathan
in February, but then the total popped back to $882 million in March (see figure showing Q1 versus 2024 numbers on previous page). So fairly stable month-to-month activity—which wasn’t the case for early-stage deals, as Norris explains below.
Singling out the largest, most compelling deals in Q1—the 16 investments totaling $50 million or more by indication—a handful were cardiovascular, according to Norris. These include 4C Medical Technologies ($175 million), Supira Medical ($120 million), VitalConnect ($100 million debt and equity financing), Alleviant Medical ($90 million), and Cardiac Dimensions ($53 million).
In non-invasive monitoring (NIM), FIRE1 ($120 million) and VitalConnect ($100 million) were among the $50 million-plus deals announced in the first quarter.
On the neuro side, there were also several bigger deals in Q1, including MicroTransponder ($65 million; detailed below), Saluda Medical ($100 million), and Cala Health ($50 million). Norris notes that although Cala announced the initial closing of their financing in December 2024, the company upsized it a bit in 2025, so he is counting it in 2025.
Looking at the 16 $50 million-plus deals in Q1 by company stage, Norris notes that it was an even split between commercial and clinical.
“This is a shift from what we saw in 2024, where eight of the top 10 raises were for companies already on the market,” says Norris.
Norris also notes that many of the big deals this past quarter are pre-commercial, PMA-stage (and thus becoming prime M&A target) companies. The biggest med device deals in Q1 included:
● 6 commercial 510(k) companies
● 2 commercial PMA companies
● 6 PMA companies raising money for pivotal trials
● 2 PMA companies raising money for feasibility trials
“So far this year we are seeing companies successfully raise pivotal trial rounds, which is very important,” explains
Norris. “We see the opportunity for these PMA deals to get to a potential acquisition before they get approved. Typically, we see the exit zone present itself after they finish a pivotal trial. That’s where the big guys typically come in and say, we like this company, we like the market. Let’s just pick it up now, we’ll take it from there and handle the commercialization. It’s good to see venture investors taking those bets, and provides nearterm validation for early-stage PMA companies that funding will be available to fund pivotal trials.”
Norris explains that of the 16 biggest deals, eight had valuation information around them. Seven of the eight were either up or flat rounds, and one was down, Norris notes.
“We’re seeing larger rounds for companies that are doing very well in the eyes of new investors. An up-round means that new investors are ascribing value to what the company has done in their last round of fundraising,” says Norris. “Larger rounds are going to companies that investors see real value in—those companies have hit important milestones.”
“We’re seeing larger rounds for companies that are doing very well in the eyes of new investors.”
On the investor side, three large venture firms—Gilde Healthcare, Longitude Capital, and Ally Bridge Group took part in several $50-million-plus medical device syndicate deals this past quarter. Here we’ll detail a sampling of these multiple-investor deals, interestingly with several being cardiovascular deals (see next page for link to a compelling cardio investment panel from LSI USA ‘25).
In January, Gilde Healthcare participated as an existing investor and Longitude Capital as a new investor in Dublin, Ireland-based heart failure device solutions company FIRE1’s $120 million financing round. The deal was led by new investors Polaris Partners and Elevage Medical Technologies, joined by another new investor Sands Capital, and additional existing investors Andera Partners, Gimv, the Ireland Strategic Investment Fund, Lightstone Ventures, Medtronic, NEA (New Enterprise Associates), Novo Holdings, and Seventure Partners
Also in January, Gilde led a $90 million financing in Alleviant Medical, a privately held company developing a no-implant
In Q1, LSI Alumni raised over $1.37 billion across 45 deals. Our quarterly Alumni Funding Report features transactions made by these medical device startups and their investors, as well as links to our proprietary media and coverage of the companies.


Top medtech investors from Deerfield Management, KCK Medtech, Broadview Ventures, and Orchestra BioMed discuss key trends, strategies, and opportunities shaping cardiovascular investments for 2025. (Visit LSI’s YouTube channel.)
This panel featured: Bill Little — Orchestra BioMed; Michael Hurley — Deerfield Management; Viral Gandhi — KCK Medtech; and Daniel Gottlieb — Broadview Ventures.
atrial shunt for heart failure, to fund its second pivotal trial. The round also added Omega Funds and includes participation from existing shareholders S3 Ventures, RiverVest Venture Partners, Vensana Capital, Longview Ventures, Gilmartin Capital, TMC Venture Fund, and undisclosed strategic investors.
In February, ambulatory cardiac monitoring company VitalConnect closed $100 million in financing through a combination of equity and debt capital. Equity financing was led by new investor Ally Bridge Group, with significant participation from existing investors including EW Healthcare Partners, MVM, and Revelation Partners, while Trinity Capital provided the debt financing.
In March, Gilde and Longitude participated as new investors in a $65 million Series F financing round for commercial-stage implantable vagus nerve stimulation company MicroTransponder, along with existing investors US Venture Partners, Osage University Partners, Action Potential Venture Capital, GPG Ventures, The Vertical Group, and Exceller Hunt Ventures
Also in March, Cardiac Dimensions, developer of minimally invasive treatment modalities to address heart failure and associated cardiovascular conditions, closed an oversubscribed $53 million Series E financing round led by Ally Bridge Group. New investor Claret Capital Partners and existing investors Hostplus, M.H. Carnegie, Horizon 3 Healthcare, Lumira Ventures, and a confidential strategic investor also significantly participated in the round.
Along with overall Q1 device venture investments, first financings—defined by HSBC as seed and Series A venture rounds totaling at least $2 million—also surged in the quarter. In medical devices, 25 deals were reported, totaling $289 million.
“While the total sounds small compared to overall, it was the second biggest quarter for early stage in the last four years,” says Norris. “Q2 of last year was a big quarter, coming in at $511 million. That was the biggest quarter we had ever really seen in early-stage device. But Q1 2025’s $289 million is close to double any other quarter in 2024, which ran at about $150 million a quarter. So this is a really strong early-stage number.”
In terms of deal size, there were no first-financing deals over $50 million, but there were four deals exceeding $20 million, and then another five deals that raised between $10 and $20 million, according to Norris. “Those are good-sized early-stage rounds for the device sector, and will typically give companies some room to hit important value inflection milestones,” he says.
However, in terms of the investors that stepped up to the plate for first financings in the quarter, Norris says the typical, traditional players stayed on the bench.
“I noted one deal each from OrbiMed, the Mayo Clinic, ShangBay Capital, and Catalyst Health Ventures. But most
“It was the second biggest quarter for early-stage [device investments] in the last four years.”
of the other folks that are typical major players in early-stage device were quiet in Q1.”
Norris says that this relative radio silence begs the question: why would investors want to invest in Series A medical device companies right now?
“There’s uncertainty in the market at the moment around not only what’s going on within the FDA, NIH, and the financial markets, but also about the ability to find a new Series B investor. And that’s been a longstanding concern in the med device arena,” he explains. “Typically, when a company gets to Series B, it hasn’t taken substantial technology risk off the table, and doesn’t have a substantial amount of patient data. While companies have made progress, new investors often do not see enough progress to enable a strong step-up in valuation.”
What Norris is seeing is a bit of a push and pull in the market.
“It’s a great opportunity for investors to come in and do a Series A in terms of finding great companies and actually negotiating favorable valuations. But a lot of these investors are just not willing to do it right now because it is so difficult to find new Series B investors,” he says. “Many typical early-stage players have pulled back Series A activity, in favor of later-stage deals with more technology risk off the table and closer to exit.”
Looking at first financings by indication, in 2024, cardiovascular didn’t get much investment. But in Q1 of this year, four cardiovascular deals came in for a total of $60 million, notes Norris.
The story becomes a bit more telling when examining the monthly totals. January and February appeared stable, with $150 million invested in January and $113 million in February. This compares to $144 million in Q124, an impressive $511 million in Q2, $152 million in Q3, and $164 million in Q424.
But then came March of this year, when funding plunged to just $26 million.
“Early-stage medical device funding dropped off a cliff in March. But because these numbers are not really big, and one month does not constitute a trend, I offer caution about concerns with how light the month was. It will be very interesting to see where dollars go over the next few months,” says Norris.
While Q1’s numbers paint an overall positive picture, the undercurrents, as noted with first financings, are anything but stable. Medical device venture investors are selective, cautious, and protective—and startups must go above and beyond to prove not just their technology, but their long-term viability in a market where fear of the next round is driving every decision.
As Norris predicted in his 2024 year-end outlook, 2025 is going to be a “rubber hitting the road moment” for a lot of companies that were on insider rounds that might not be able to find a new investor. (See “Look What You Made Me Do: Novel Tech Deals Dominate Device Venture Investment in ‘24, Set Stage for a Brisk ‘25,” The Lens, January 2025.)
“It’s also a function of the fact that we had such a buoyant 2020 and 2021, with a lot of companies launched during that period of time,” he says. “Many companies that scraped by on insider rounds over the past two years now face a brutal reality. Find a new investor or fold. With limited support left in existing funds, consolidations and asset sales could rise in some segments.”
As seen in the Q1 numbers, investors are showing that they are cautious, but as always, opportunistic. The best deals are happening, but only for those who can prove they’re ready.
“The best investments are made when the market is down,” says Norris. “But when you don’t know where the floor is, it’s hard to commit. It’s a market watching itself, unsure whether to speed up or slow down. Keep in mind that the device sector has always been resilient, and it will be able to navigate this down cycle,” Norris concludes.

Jonathan Norris joined HSBC’s Innovation Banking Division in April 2023 from SVB, where he was a Managing Director for 17 years. At HSBC, he manages healthcare venture relationships and works with investors and companies on commercial banking and debt products. In addition, for more than a decade, Norris has written comprehensive reports on the venture healthcare ecosystem, covering venture fundraising, investment, valuations, and exits. These reports have been widely cited in the ecosystem, and he often speaks at major investor and industry conferences. He has more than 20 years of healthcare banking experience. Norris earned a B.S. in business administration from the University of California, Riverside, and a J.D. from Santa Clara University.
You can reach Norris at Jonathan.Norris@us.hsbc. com. View his 2024 Annual Venture Healthcare Report at www.hsbcinnovationbanking.com/us/en/business-stage/ hsbc-healthcare-reports, and stay tuned for his first-half 2025 venture healthcare report in July.



via the eyes
closed $1.2m in one week following their announcement of a $3m Series A raise. They are reporting commercial growth in Houston, Denver, San Antonio, North Carolina, Tennessee and Ohio and are hiring sales professionals in those locations.
Ottobock (led), Breakout Ventures, Draper Associates, LionBird Ventures, Time BioVentures, Risk and Return, Actual VC, METIS Innovative, e1 Ventures, Jumpspace, MainSheet Ventures, Brown Advisory 4/15/2025
disorders $6M Kickstart Fund (led), other current and new investors 4/18/2025
Aopeng Medical Series C Fully autonomous robotics for interventional surgery $13.8M* Jinan International Medical Center Industrial Development (led), Jinan Huaiyin District Guorong Holdings (led), Shanlan Capital 4/20/2025 AnthroTek Seed Artificial silicone body parts for medical & surgical training, prosthetics, & robotics $1.99M* Undisclosed 4/21/2025 MgShell Seed Magnesium-based ophthalmic drug delivery plaform for retinal diseases $1.29M Capital Cell (led), Andrea Giani, MD (led), members of Club degli Investitori through Simon Fiduciaria, Italian Angels for Biotech, Bios Line Holding 4/22/2025
Biolinq Series C Needle-free continuous metabolite monitoring technology
$113.2M Alpha Wave Ventures (led), RiverVest Venture Partners, AXA IM Alts, LifeSci Venture Partners, M Ventures, Hikma Ventures, Aphelion Capital, Taisho Pharmaceutical, Features Capital 4/22/2025
Field Medical Series A Pulsed field ablation solution for cardiac arrhythmia treatment $40M Undisclosed strategics, venture capital firms, and family offices 4/22/2025
H'ability Seed VR rehab for physiotherapy & occupational therapy
Your one-stop-shop for global market sizing and analysis, procedure volume data, startup company and deal tracking, curated insights, and more. *Currency converted to $USD for data consistency (5/5/25 exchange rate). Company

$1.2M* YEAST, BAMA, Bpifrance, Banque Populaire, CIC 4/22/2025
Mendaera Strategic Partnership Medical robot that blends imaging and AI to improve access to interventions NA Cook Medical - collaboration combining Cook's EchoTip needle with Mendaera's ultrasound guided handheld robotics 4/23/2025
Coologics Seed Drug-free device for vaginal infections
Conavi Medical Public Offering Technologies to help guide minimally invasive procedures, starting with cardio
Anumana Series C AI algorithms for detecting rare and chronic conditions
SonoMotion Grant Non-invasive solutions for the treatment of kidney stones
Araceli Biosciences Seed Assay scanning systems for drug discovery
Prevail Partners (led), Dream Ventures (led), Ohio State Early Investor Network, Illinois Ventures Illini Angels Program 4/23/2025
$14.5M* Public offering of 32,500,000 common shares and 17,500,000 pre-funded warrants 4/23/2025
NA Boston Scientific, Matrix Capital, Mayo Clinic, Matrix Partners, NTTVC
$2.2M Fastrack NIH SBIR
$11.2M Bill Cortelyou (led)
Core Medical Series D Lightweight, low-power LVAD $100M ZhenFund (led), Zhongguancun Special Fund for Independent Innovation of the National Social Security Fund (managed by Legend Capital) (led), Prosperity7 Ventures under Aramco Ventures (led), CStone Capital (led), Delian Capital, Lianxin Capital
Nuclivision Seed AI for PET scan enhancement
Cacto Health Seed Remote monitoring for early lymphedema detection
4/24/2025
4/24/2025
4/24/2025
4/24/2025
$5M* LUMO Labs (led), Heran Partners (led), imec.istart Future Fund 4/24/2025
$455K* EIFO, Innovation Fund via Innobooster, undisclosed business angels 4/24/2025
Cipher Surgical Series A Intrabdominal lens cleaner for laparoscopic instruments $10M Medical Solutions (led)

Lisa Earnhardt (Source: LSI USA ‘25)
Lisa Earnhardt, EVP and Group President of Medical Devices at Abbott, leads nearly $20 billion in global businesses with a vision shaped by startup grit and fueled by customer focus, patient outcomes, and bold collaboration. From cardiovascular disease to diabetes, Abbott’s goal is to democratize healthcare by staying grounded in what matters most.
On a sunny March morning along the Southern California coast at LSI USA ’25, Lisa Earnhardt, Executive Vice President and Group President of Medical Devices at Abbott, stepped on stage and delivered a memorable and inspiring talk, rooted in real-world experience.
Not just about Abbott’s innovation pipeline or the future of AI-powered technologies, but about what it really takes to lead transformation in healthcare—through every boom, bust, and detour in between.
Earnhardt joined Abbott in 2019 and was appointed to her current role in December 2023, previously serving as Abbott’s Executive Vice President, Medical Devices. But she wasn’t always a Fortune 100 executive managing nearly $20 billion in global medtech businesses. In fact, she grew up in the cornfields of Illinois, is an engineer by training, and fell—quite deliberately— after business school at Northwestern University’s Kellogg School of Management into a career that bridged her dual passions: technology and health.
In her keynote conversation at LSI USA ‘25 with Juliana Elstad, CEO of Vibrato Medical, Earnhardt summed up lessons learned in her career:
“I went from big corporation to startup back to big corporation. But my overarching mantra has always been the same: if you do what’s right for the customer, if you do what’s right for the patient, the rest will follow.”
Prior to her tenure at Abbott, Earnhardt held senior roles at both Guidant and Boston Scientific, where she led global product launches and drove adoption of new medical technologies. She served as President of Boston Scientific’s Cardiac Surgery division while it was in the process of being spun out to the Getinge Group (in 2007). Then, influenced by the tremendous amount of medtech innovation happening in the California Bay area, she decided to make the leap into a startup.
“So much of innovation happens in the startup arena. If I’m ever going to do it, now is the time,” she recalls.
“My overarching mantra has always been the same: if you do what’s right for the customer, if you do what’s right for the patient, the rest will follow.”
Earnhardt was employee #14 at the early-stage sinus stent technology startup, Intersect ENT, in March of 2008, as it charted new territory in the ENT device space. She came aboard just as the company had maxed out its funding runway, unsure if it could even make payroll in three months. “Naïveté as a first-time CEO really worked to my advantage,” she says now with a smile.
As is familiar to startup execs, she did a bit of everything: ran clinical trials, scrubbed into cases, was the receptionist, chased venture capital, washed dishes, and occasionally made morale runs to Dairy Queen. It was not the leadership style of a corner office exec—it was a master class in servant leadership and team-building from the ground up.
The technology was a novel, bioresorbable scaffold to treat sinus conditions. The market was uncertain. The regulatory path was murky. The financing was unstable. In the first few months of her tenure, Earnhardt and team had to secure a Series B round as the company was operating on emergency bridge funding. Then came the real hurdles: a surprise PMA requirement from the FDA, a full re-do of the pivotal trial, and several near-death financial scenarios.
“We planned our trial unblinding over Labor Day weekend because if the data didn’t work out, at least we had a couple days to figure out what we were going to do if it wasn’t positive,” recalls Earnhardt.
But she and her team made it through the highs and lows and eventually took the company public. Eleven years after she joined, Intersect ENT was an established player with FDA approval and solid commercial traction. In 2022,
Medtronic acquired Intersect ENT for $1.1 billion in an all-cash transaction.
Those early experiences, in both the large strategic and startup world, shaped everything Earnhardt does at Abbott. Today, she leads eight businesses tackling some of the most costly and debilitating chronic conditions, including cardiovascular disease, diabetes, and chronic pain. With over 40,000 global employees under her leadership, the scale is vastly different, but her north stars haven’t changed.
First, in a key lesson from her startup life, Earnhardt says that nothing happens without a deep understanding of the customer. And she stays true to this belief to this day. She still scrubs into procedures when she travels, sits beside physicians observing cases, and listens to patients—not just to validate ideas but “to hear the unsaid.” She believes frontline observation is the surest path to meaningful innovation.
She also abides by the rule of staying focused. She describes that at an emerging startup, the focus is on survival, and on earning the right to do a clinical trial, and earning the right to commercialize, based on financing. And this progression is based on staying focused to demonstrate what you said you would do.
“We had a ton of ideas. But we had to ask: what are the one or two things that matter right now?”
“We had a ton of ideas,” Earnhardt says when reflecting on previous startup experience. “But we had to ask: what are the one or two things that matter right now? And then that earns us the right to do the next thing.”
Today at Abbott, when her much larger team brings forward their big ideas, Earnhardt continues to apply those lessons of discipline and focus to

help them make the best decisions for patients. Earnhardt also firmly believes in protecting innovation and getting game-changing products through the gauntlet. Some of Abbott’s most successful launches in recent years, for example, leadless pacemakers, transcatheter aortic valve implantation (TAVI) devices, and continuous glucose monitors, all went through a rigorous evaluation process to identify the best innovations to meet the needs of patients and fuel future growth.
Earnhardt’s keynote traced a clear path toward healthcare’s transformative next iteration: decentralization into patient’s homes and other settings outside of the hospital. That means everything from advanced remote monitoring to app-guided wellness coaching, and critical reimbursement for these products and services—and Abbott is investing in all of it.
“We’re now finally at a point where we’re moving from a system that I would call ‘sick care,’ to more well care, or truly ‘health’ care,” she says. “As an industry, medtech is really leading the way, with things like remote monitoring and management.”
Earnhardt cited the Abbott CardioMEMS HF System as an example of this innovation within the company’s heart failure business. CardioMEMS is a pulmonary artery (PA) pressure monitor the size of a paperclip. Implanted into the heart’s vasculature, it remotely monitors changes in PA pressure—an early indicator of the onset of worsening heart failure—to help keep patients out of the hospital and living their best lives. The device has demonstrated a significant reduction in hospitalizations and cost of care by helping physicians adjust medications based on real-time physiologic signals.
Earnhardt also discussed NeuroSphere, Abbott’s digital health platform with pioneering remote neuromodulation programming technology and in-app video chat capabilities. Patients with chronic pain or movement disorders like Parkinson’s disease in
the U.S. (and approved countries outside the U.S.) no longer have to travel hours to centers of excellence for device adjustments. A pandemic-era
Helping to ensure that patients have access to this technology, on January 13, 2025, the Centers for Medicare & Medicaid Services (CMS) issued a National Coverage Determination (NCD) to cover implantable pulmonary artery pressure sensors (IPAPS) for heart failure management, under Coverage with Evidence Development (CED).
The CardioMEMS HF System CED study is now approved by CMS for the IPAPS NCD. The NCD with CED means that Medicare beneficiaries will have expanded access to care for both the Traditional Medicare and Medicare Advantage Plans if appropriate criteria are met.
Source: Abbott
pivot enabled Abbott to launch a remote therapy management platform years ahead of schedule. Now, clinicians can program and adjust stimulation protocols from afar (note: anywhere with a cellular or Wi-Fi connection and sufficiently charged patient controller). It’s a game-changer for patients who struggle with mobility or caregiver availability.
“We realized the need wasn’t five years away. The need was now. So we moved the timeline up.”
“During the pandemic, we realized the need wasn’t five years away. The need was now. So we moved the timeline up,” says Earnhardt. By making this change, Abbott was able to accelerate its plans and introduce NeuroSphere in 2021.
Abbott isn’t just a medtech powerhouse, Earnhardt tells the LSI USA ‘25 audience. It’s one of the few companies that truly understands consumers, thanks to its presence in direct-to-consumer diagnostics, nutrition, and diabetes care. That advantage comes to life in its flagship continuous glucose monitor (CGM) biowearable, the FreeStyle Libre, which has redefined diabetes management for 7 million people, with many more who could benefit.
With a tiny sensor worn on the arm and real-time data delivered to a smartphone, the FreeStyle Libre portfolio has transformed how diabetes is managed—from finger pricks and guesswork to easy-to-use, personalized insight and preventive action. For Earnhardt, it was her first real foray into consumer tech. She was hooked.
Now, Abbott is leveraging this consumer focus and expanding its reach with the new Lingo CGM biowearable system, available over the counter to U.S. consumers who are interested in improving their overall health and wellness. Lingo, FDA-cleared in June 2024, is a metabolic wellness product that turns CGM data into behavior coaching,
translating glucose patterns into actionable micro-decisions. The Lingo biosensor, which can be worn for 14 days, tracks real-time glucose levels while the accompanying app provides personalized coaching insights to help people create healthy habits, retrain their metabolism, and improve their overall well-being.
“I call it my accountability partner, my little buddy. After 30-plus years in healthcare, I still need help making good choices,” laughs Earnhardt.
Earnhardt believes this model—integrating personal data with technology providing real-time feedback and behavioral AI—is the powerful future of preventive health. And she sees Lingo as an important step.
When Earnhardt talks about innovating with data insights, it’s not hypothetical. To date, more than 50 billion hours of glucose data has been collected from Abbott’s biowearables— data that is already fueling real-world decision-making.
In cardiology, the company’s AI-powered optical coherence tomography (OCT) imaging platform uses pattern recognition to identify calcium and vessel diameter, helping guide stent size choice and placement with surgical precision. It’s built on the expertise of tens of thousands of interventional cardiologists and millions of patients over time, all embedded into one platform.
Lisa Earnhardt’s Medtech Manifesto, in Four Lessons
1. Do what’s right for the patient, and everything else follows.
2. Stay close to the problem and listen to what’s not being said.
3. Focus earns you the right to grow.
4. Collaboration is the new competitive advantage.
This is what Earnhardt calls “democratizing healthcare.” By encoding expert experience into accessible algorithms, Abbott is utilizing the power of data to improve patient care across healthcare systems.
But the real game-changer, she says, is collaboration. Abbott, a company that has been around for an incredible 137 years, isn’t going it alone. In fact, one of its newest partnerships, announced last August, is with Medtronic—a longtime competitor. The two are co-developing integrated insulin pump and CGM systems for diabetes, offering users a seamless experience for managing diabetes and prioritizing patient needs over turf wars.
“Innovation at scale requires everyone at the table.”
“Gone are the days of going it alone. Innovation at scale requires everyone at the table. It’s the right thing for customers, and it’s the right thing for patients,” she says.
For Earnhardt, collaboration has become a key strategy in today’s medtech ecosystem. In fact, it’s a survival skill in today’s complex ecosystem of care delivery, regulation, payment, and technology.
As she wrapped up her keynote session, Earnhardt issued a call to action for the industry. Not just to innovate faster, invest smarter, or stay focused, but to convene. To come together in spaces like LSI’s events and others around the globe.
“It’s important to connect within our medtech ecosystem to hear new ideas and meet with innovators and investors across the space,” she says.
Bringing together founders, funders, regulators, and strategics—not to compete, but to connect and collaborate to serve patients—is now mission-critical.
“Healthcare is a team sport, and we need to think more expansively about how we partner. It’s never needed more teammates than it does right now,” Earnhardt advises the LSI USA ‘25 audience.

September 7th - 11th, 2025
JW Marriott Grosvenor House, London
Now in its fourth year, LSI Europe has become a staple of the global Medtech and Healthtech community. The 2025 event in London will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Registration and applications to present are now open for LSI Europe ‘25. Get in touch to learn more about the event.


Once limited to traditional surgical interventions, the market for rotator cuff repair suture anchors is now evolving, driven by an aging global population and advances in minimally invasive surgical techniques. As the demand for rotator cuff repair procedures steadily increases, suture anchors are becoming a cornerstone in restoring function and mobility to millions suffering from rotator cuff injuries. With ongoing innovations and competitive pressures, the market is set to see modest growth in the coming years.


Driven by advances in surgical technology and a rapidly aging population, the global rotator cuff suture anchors market is poised for steady expansion, with its 2024 valuation of $100.9 million projected to reach $104.2 million by 2028, according to LSI Market Intelligence. This growth rate of 0.8% CAGR over the 2023-2028 forecast period is reflective of the ongoing demand for rotator cuff repair procedures, a key element in orthopedic surgery. Despite the market’s slow pace, key demographic shifts and emerging technologies indicate a promising outlook for this segment, particularly in aging populations.
A torn rotator cuff, a critical injury among aging populations, is commonly the result of degenerative wear and tear rather than acute injury. Repairing such injuries through minimally invasive procedures—which utilize suture anchors to reattach torn tendon tissue—has become standard practice in sports medicine and orthopedic care.
Suture anchors, typically small screws placed in the bone during surgery, are critical in holding sutures that reattach torn rotator cuff tendons. Over the years, these products have

evolved, with innovations aimed at improving ease of use, reducing surgical complications, and enhancing the longevity of repairs. From 2023 to 2028, the market for suture anchors used in these procedures is expected to grow at a slow pace, largely due to the decline in the average selling price for rotator cuff suture anchors, according to LSI Market Intelligence.
The growing number of individuals aged 65 and older—who experience a higher rate of rotator cuff tears—plays a crucial role in fueling the demand for rotator cuff repairs. According to LSI’s Global Surgical Procedure Volumes database, the volume of rotator cuff repair procedures is expected to increase at a steady rate, aligning closely with the rising number of elderly individuals globally, particularly in Japan, China, and South Korea. The rate of growth in rotator cuff procedures is expected to align with the 2.9% annual increase in the 65+ population, which continues to be a key driver of this market.
Despite the demand for rotator cuff suture anchors, the market faces hurdles that could limit more aggressive growth. Recent studies have revealed mixed patient outcomes following rotator cuff repair, dampening some enthusiasm for adopting this procedure universally. Additionally, the continued pressure from lower-cost suppliers, particularly from China, has introduced price erosion, putting further strain on market expansion. Despite these challenges, suture anchors remain essential to surgical repairs, and as a result, the overall market size will likely remain stable.
The competitive landscape is led by DePuy Synthes, part of the Johnson & Johnson orthopedic family, which dominates the market with its broad portfolio of suture anchors. Smith+Nephew and Zimmer Biomet also maintain notable shares in this market.
DePuy Synthes, with an estimated 60.4% market share in 2023, has maintained its dominance in the rotator cuff suture anchor market thanks to its robust market position in orthopedic and sports medicine devices. The company’s extensive clinical backing and long-established presence across key surgical markets ensure its continued market leadership. However, as more cost-effective and innovative solutions enter the market, including from smaller, agile competitors, this market dynamic may shift slightly (see market share table on the next page).
The competitive environment is further complicated by legal and regulatory challenges. In August 2023, Arthrex faced a class action lawsuit in the United States alleging design and manufacturing defects in its suture anchors, which raised questions regarding the reliability and safety of anchor products. Nonetheless, this is a typical part of a market populated with high-stakes medical devices, and companies that continue to innovate will maintain their competitive edge.
While the market’s growth remains slow and steady, technological advances are expected to contribute significantly to future demand. Innovations in suture anchor materials and designs aimed at improving ease of implantation and reducing post-surgical complications could provide a strong competitive edge for companies in the coming years. A shift towards more bioresorbable materials, which naturally degrade over time, and anchors designed for greater anatomical fit could see increased adoption among surgeons seeking more reliable and efficient solutions.
Another important trend is the ongoing growth in emerging markets. As Asia-Pacific markets continue to develop and healthcare infrastructure improves, countries like China, Japan, and South Korea are expected to become critical to medtech market growth. These regions are experiencing particularly high growth in rotator cuff repair procedure volumes, which is expected to drive corresponding growth in unit volumes of rotator cuff suture anchors.



DePuy Synthes/J&J
Smit & Nephew
Zimmer Biomet
Others
Tensor Surgical, led by President and CEO Justin Anderson, is revolutionizing rotator cuff repair with its TransOs Tunneler system, which eliminates traditional suture anchors. This anchorless approach preserves bone integrity, reduces pain, and eliminates complications like anchor pull-out, all while cutting procedural costs by up to $1,500 per case. With 10,000 successful procedures and national contracts, Tensor is set for global growth. Its AI-driven platforms and focus on value-based care make it a major disruptor in the orthopedic space.
Reach Orthopaedics, led by CoFounder and CEO Christina Goudy, is addressing a gap in care for middle-
aged patients with massive rotator cuff tears. Their subacromial spacer implant offers a minimally invasive solution that preserves shoulder anatomy, reducing pain and improving function without requiring bone removal. With FDA Breakthrough Device Designation and successful cadaver studies, Reach is on track for a first-in-human study in 2025, offering a promising alternative to joint replacement for younger patients.
15,304.7
Tetrous, led by Co-Founder, Chairman, and CEO Bradley Patt, is advancing rotator cuff repair with its EnFix RC and EnFix TAC products, which utilize demineralized bone fiber (DBF) technology to promote healing at the tendon-to-bone interface. This biologic approach addresses a common failure point in rotator cuff surgeries, improving tendon-to-bone healing and reducing the risk of re-tear. Early studies show
stronger fixation and better outcomes compared to traditional suture anchors, positioning Tetrous as a leader in orthopedic regenerative solutions. Recently, the company closed a successful Series A financing round, raising $6.5 million to expand market penetration and clinical data. Additionally, Tetrous signed a distribution agreement with Alpha Medical in New Zealand, marking a key step in its Asia-Pacific expansion.

Mesh Suture, led by CEO Adom Dumanian, is innovating soft tissue repairs with its Duramesh, which flattens like a ribbon to evenly distribute tension, reducing tissue damage. Clinical studies show superior outcomes with fewer complications and faster healing compared to standard sutures. With FDA clearance, CE mark approval, and a growing presence in the U.S. and Europe, Mesh Suture is poised to make a significant impact in soft tissue repair across multiple surgical specialties.

Notes: Figures may not sum due to rounding. The “Others” category includes Anika Therapeutics, Arthrex, ConMed, Stryker, and Wright Medical, among others. Source: Annual company reports, SEC filings, LSI Market Snapshot and Investor Relation Materials
Despite modest projected growth, the rotator cuff suture anchor market remains vital to orthopedic surgery, driven by rising global procedure volumes and demographic shifts. As innovation continues and new players enter the space, companies that can differentiate on clinical outcomes, ease of use, and pricing will be best positioned to compete.

Theglobaldrugdeliverydevices marketisexperiencingsteadygrowth asinnovationsinbiologics,smart technologies,andpatient-centric designtransformhowtreatments areadministeredacrosshealthcare settings.Frominfusionsystems andinsulinpumpstowearable solutions,thesedevicesare playinganincreasinglyvitalrole inmanagingchronicconditions andenhancingcare.



The drug delivery devices market, valued at approximately $18.7 billion in 2023, is on track to reach $24.6 billion by 2028, growing at a CAGR of 5.7% over the 2023-2028 forecast period. This market, integral to modern healthcare, is evolving to accommodate new biological drugs and increasingly sophisticated delivery technologies, reshaping the landscape for the treatment of both acute and chronic conditions.
Drug delivery devices—which include injection devices, infusion pumps and sets, gravity administration sets, vascular access catheters, enteral solutions, and external insulin pumps—are used across a variety of healthcare settings.
From hospitals administering fluids to patients in need of emergency care to outpatient treatments for chronic conditions like diabetes, these devices form the backbone of patient care for millions globally. The market’s growth is fueled by a combination of factors, including the adoption of newer, high-cost biologic drugs, which require intravenous administration, and the increasing prevalence of chronic conditions in aging populations.
In particular, the market is seeing a rise in demand for smart pumps, which help reduce medication errors and automate drug administration. These devices are playing a crucial role in hospitals and care facilities, increasing both efficiency and patient safety. The growing adoption of these devices has led to an increase in the sales dollar volume, albeit at a pace slightly slower than the rate of medical device price inflation.
One of the most significant developments in the drug delivery devices market is the increasing adoption of smart infusion systems and smart pumps. These devices not only help reduce medication errors but also offer enhanced features like automated drug administration documentation, contributing to more reliable patient care.
However, this technological shift is not without its challenges. Several smart infusion systems have faced recalls due to issues such as improper medication administration and cybersecurity vulnerabilities. This, in turn, has led to increased scrutiny from regulators, which may slow down the widespread adoption of these systems.
The trend towards biologic drug delivery is expected to continue driving growth in the drug delivery devices market. As more biologics require intravenous or subcutaneous administration, the demand for devices that can safely and efficiently deliver these treatments will rise. This includes products like insulin pumps, infusion pumps, and syringes, which are becoming increasingly sophisticated. Smart pumps and biosensors that monitor and adjust dosage in real time are expected to play a pivotal role in the future of drug delivery.
In addition to biologic drugs, patientcentric innovations are gaining traction. Companies are increasingly focusing on user-friendly devices, such as patch pumps and wearable drug delivery systems, which allow patients to manage their treatments at home with minimal medical supervision. These devices are particularly attractive to patients with chronic conditions, such as diabetes,
The “Others” category includes Ace Medical, Ambu, Avanos, Avid Medical, Cane, Infutronix, Iradimed, Laborie Medical, MicroPort Scientific, Moog, Nouvag, Stradis, Teleflex, Walkmed, and Zyno Medical, among others.
Source: LSI’s Drug Delivery Devices Market Snapshot, published August 2024
who require continuous medication delivery. (Also see Market Dive: Wearable Monitoring Devices, The Lens, March 2025.)
Another notable trend is the growing market for injectable drug delivery systems designed for chronic disease management, such as diabetes and rheumatoid arthritis. These devices, including insulin pens and jet injectors, are becoming more advanced, offering patients a more comfortable and efficient way to manage their conditions at home.
The market’s volume growth is projected at a CAGR of 4.9%, correlated strongly with hospitalization rates, which are increasing as the population continues to age and develop chronic conditions requiring drug delivery products to manage. The global unit volume for drug delivery devices is projected to increase from 9.7 million units in 2023 to 12.3 million units in 2028.
As the market grows, Medtronic, Baxter, and BD continue to dominate the competitive landscape, holding approximately 48% of the market share in 2023. Smaller players like ICU Medical have also gained ground, with ICU Medical increasing its market share from 5% to 8.6% in 2023 via the acquisition of Smith Medical’s drug delivery business.
As in many other segments of the medtech market, the drug delivery devices market remains highly competitive and fragmented, with no single player holding more than 20% market share (see figure). In addition, smaller medtech companies like Medtronic, Baxter, and BD continue to dominate, but smaller medtech companies are emerging with innovative technologies. ICU Medical, for example, has successfully expanded its share through strategic acquisitions, such as Smith Medical’s drug delivery business. Other companies like B. Braun and Teleflex are also pushing for a larger slice of the market, focusing on both innovative product design and global market penetration.

Altra is redefining the infusion pump landscape with its Altra Infusion System, a breakthrough technology that delivers unparalleled accuracy and precision. Unlike traditional pumps that rely on squeezing tubes, The Altra Infusion System uses a two-chamber system that remains unaffected by factors like pressure or rate. The result? 10 to 100 times the accuracy of existing systems, making it ideal for neonatal units and emergency situations. The pump also significantly reduces nuisance alarms, a common issue in ICU settings, and is portable, waterproof, and compact, opening the door for use in military, ambulance, and field care.
Recently, Altra achieved a major milestone by having its dual-chamber infusion system accepted into the FDA’s Safer Technologies Program (STeP). This acceptance will help Altra as it prepares its 510(k) submission for market clearance.
ViCentra is tackling one of the most pressing challenges in diabetes care with its Kaleido insulin pump—a small, wearable device designed for a seamless user experience. The Kaleido pump stands out due to its compact size (just 19 grams), accuracy, and reusability, making it more affordable than competitors like the OmniPod. With its sleek

design available in 10 colors, ViCentra is making insulin pumps more personalized and user-friendly for patients. ViCentra achieved a significant milestone by securing CE Mark Certification for the Kaleido insulin pump under the EU Medical Device Regulation (MDR) in October 2024.
In another key development, ViCentra appointed Tom Arnold as CEO in February 2025. With nearly 25 years of leadership experience in the medical technology sector, Arnold is poised to scale manufacturing, drive European expansion, and guide the company as it prepares to introduce the Kaleido insulin patch pump system to the U.S. market.

Perceptive Medical is addressing a critical issue in healthcare with its innovative solution aimed at improving blood pressure management in critical care settings. The company’s BaroHawk system is designed to automate the titration of blood pressure medications, reducing the need for manual intervention by healthcare providers. This system has already demonstrated significant clinical benefits, including
reducing hypotension time and severity by 95% or more. With a focus on improving patient safety and reducing provider workload, the BaroHawk system aims to enhance outcomes in ICUs and emergency settings. The BaroHawk has received FDA Breakthrough Device Designation and is progressing through a PMA regulatory path.

Liquet Medical is transforming the treatment of pulmonary artery blood clots with its groundbreaking Versus Catheter. This innovative device enables Hemodynamics-Led Thrombolysis (HLT), allowing clinicians to personalize thrombolytic therapy by monitoring real-time pulmonary artery pressure. Recently, Liquet received FDA 510(k) clearance for the Versus Catheter, marking a significant milestone for the company. The device’s dual-tip design allows simultaneous treatment of both lungs via a single catheter, improving treatment efficiency. With its data-centric approach, Liquet’s technology has the potential to improve patient outcomes while minimizing risks and reducing healthcare costs.
While the drug delivery devices market faces some regulatory and technological hurdles, it remains one of the most essential segments in healthcare. Driven by demographic changes and increasing chronic disease management, the market is set to experience steady growth. As innovations continue and companies push for smarter, more user-friendly devices, the market presents ongoing opportunities, particularly for those companies that can deliver high-quality, reliable solutions at
Every week, LSI’s Market Intelligence team dives into a different topic shaping the future of medical technology. From disruptive startups to fast-growing device segments, we connect the dots between clinical demand, market dynamics, and innovation pipelines. Here’s what the LSI Market Intelligence team tracked this past month across electrophysiology (EP), transcatheter mitral valve replacement (TMVR), and the U.S. and APAC medtech markets.

Heart rhythm disorder management is entering a new era. Once dominated by basic monitoring tools like the surface ECG, EP now encompasses a robust ecosystem of AI-powered diagnostics, leadless pacemakers, non-invasive ablation technologies, and real-time navigation software.
Today, over 1.5 million pacemaker implantation procedures and 1.4 million cardiac ablation procedures are performed annually. Demand is growing, driven by technological refinement, an aging global population, and rising awareness of arrhythmias like atrial fibrillation (AFib).
• CEO: Juan Jimenez
• Stage: Seed
• Raise to date: $3.2M
• Innovation: AI-driven ECG analytics for 13 heart rhythm disorders
• Potential Impact: Reduces time to diagnosis and supports both remote and ambulatory patient monitoring
• CEO: Edouard Fonck
• Stage: Series A
• Raise to date: ~$18.5M
• Innovation: A leadless, self-sustainable pacemaker powered by the heart’s own kinetic energy
• Potential Impact: Improves the device’s efficiency and durability
• CEO: Amir Soltanianzadeh
• Stage: Series A
• Raise to date: $4.5M
• Innovation: Non-invasive neuromodulation wearable targeting the median-vagus axis
• Potential Impact: Offers a personalized therapy for reducing AFib burden and symptoms in a market of more than 60 million patients (Also see LSI Alumni Innovator Spotlight: CardiaCare CEO Amir Soltanianzadeh, The Lens, February 2025.)
• CEO: Marina Izzo
• Stage: Series A
• Raise to date: ~$22.0M
• Innovation: Non-invasive arrhythmia ablation using synchronized ultrasound and proton beam therapy
• Potential Impact: Eliminates catheterization, potentially broadening access to arrhythmia care
• CEO: Théophile Mohr Durdez
• Stage: Series B
• Raise to date: ~$69.0M
• Innovation: AI-powered mapping software compatible with most EP systems
• Potential Impact: Improves precision during ablation procedures
According to LSI Market Intelligence, the global mapping and ablation consoles and catheters market is already worth $6.5 billion and growing at a 9.3% CAGR, outpacing most of medtech. The convergence of non-invasive technology, intelligent software, and growing clinical need is fueling a wave of innovation across the EP space.
The TMVR market has reached an inflection point. With CE Mark approval for Edwards Lifesciences’ SAPIEN M3, a transfemoral replacement system for moderate-to-severe or severe mitral regurgitation, the field is finally seeing a commercially viable, minimally invasive TMVR solution.
This is more than a product milestone. It represents a turning point for the entire structural heart landscape.
• Global Procedure Volumes: From 58,000 in 2024 to 128,000 by 2028 (22% CAGR)
• Market Value: From $1.65B in 2024 to $5.47B by 2029 (27.0% CAGR)
• Clinical Demand: Over 24 million people globally suffer from mitral valve disease; a majority remain untreated or ineligible for surgery.
If the trajectory of aortic valve replacement (TAVR) is any indication—growing from high-risk to broad indications in under a decade—the TMVR market may see even faster expansion.
Source: LSI’s Global Procedure Volumes Database
Edwards Lifesciences is forecasting $2 billion in sales from its transcatheter mitral and tricuspid therapies segment by 2030. Meanwhile, Abbott retains a dominant 75% share of the mitral repair and replacement market. However, with over three dozen companies developing TMVR solutions according to LSI’s Compass, competitive dynamics are intensifying.
• CEO: Maggie Nixon
• Stage: Series C
• Latest Raise: $110M
• Solution: Catheter-based surgical robot to deliver heart valve implants for the repair and replacement of diseased heart valves
• CEO: Mike Dineen
• Stage: Later stage
• Currently Raising: $40M
• Solution: Transcatheter valve replacement experience that is procedurally simpler and less invasive than the current mitral or tricuspid approaches
• CEO: David Wilson
• Stage: Series C
• Currently Raising: $17.6M
• Solution: Saturn trans-septal TMVR System addressing many challenges, such as left ventricular outflow tract (LVOT) obstruction and annular size and shape, identified by other companies in early feasibility studies focused on addressing MR
• CEO: Wei Sun
• Stage: Series A
• Currently Raising: $8M
• Solution: Minimally invasive solution for treating MR, featuring a posterior leaflet replacement designed to coapt with the native anterior mitral leaflet, effectively eliminating MR while maintaining a minimal footprint in the heart

In the world’s largest medtech market, accounting for more than 45% of global medtech revenue, the numbers reveal a different narrative than the headlines—one of quiet strength and consistent growth. While macroeconomic pressure and policy changes dominate media coverage, surgical procedure volumes in the U.S. continue to trend upward.
• 2024 Actuals: Revised up from 92.3M to 95.7M
• 2025 Forecast: 97.8M procedures
• 100M Milestone: Expected as early as 2026
The top three procedure categories by volume in the U.S. are:
1. Aesthetic, Dermatological, and Plastic Surgery
2. General Surgery
3. Urological Procedures
A closer look at high-growth individual procedures from 2024–2029 highlights the broader strength of the U.S. market:
• Extracranial Radiotherapy (excl. lung/spine): 17.3%
• Shoulder Replacement: 11.5%
• Aortic Valve Repair: 10.4%
• Ischemic Stroke Mechanical Thrombectomy: 10.0%
• Nephrectomy: 6.0%
This acceleration is being driven by shifts in clinical demand and structural changes across care delivery, particularly

the rise of Ambulatory Surgical Centers (ASCs). ASCs enable faster, lower-cost care and are accelerating demand for devices that are compact, interoperable, and cost-effective. (Also see Market Dive: Orthopedic Surgery, The Lens, December 2024.)
• AI integration is helping clinicians stratify patients more accurately, reducing unnecessary interventions.
• The U.S. remains a first-choice market for medtech innovation, given faster commercialization timelines, ASC-friendly design, and payor alignment.
• In contrast, Europe’s MDR environment and fragmented reimbursement landscape continue to delay the adoption of novel technologies.
As Intuitive CEO Gary Guthart reminded the audience during his keynote at LSI USA ’25, “What I know is that next year people will need surgery, and I know the year after that people will need surgery, and I know that five years from now they’ll need surgery … that means that we’ll get through the turbulence.” The need is constant—and the data supports the demand. (Read more about Guthart’s insightful perspective in our April 2025 cover story, “The Rich and Bold Future of Intuitive: Inside Gary Guthart’s Vision for Robotics and Healthcare.”)
For many companies, particularly those based in the U.S., the Asia-Pacific (APAC) region is now the second-most important commercialization priority after the domestic market. With over 4.5 billion people, rapidly expanding healthcare systems, and increasing regulatory clarity in key nations, APAC is no longer a future opportunity—it’s a current imperative.

• Medtech Revenue (2024): $143.6B
• Global Share: 22.9%
• CAGR (2024–2029): 8.4% vs. global medtech market of 6.6%
• Surgical Procedure Volumes (2024): 212.6M
• Procedure CAGR (2024–2029): 4.8%
• Population: ~1.4B
• Estimated Procedure Volumes (2024): ~89.0M
• Market Characteristics: APAC’s largest medtech market, driven by government-backed innovation, rapid healthcare infrastructure development, and a growing middle class. Domestic giants like Mindray and MicroPort are gaining international traction.
• Market Access Difficulty: Challenging. Stringent localization requirements, complex regulatory hurdles, and a strong preference for domestic suppliers can limit opportunities for Western firms. Source: Welcome to

• Population: ~125M
• Estimated Procedure Volumes (2024): ~14.8M
• Market Characteristics: A highly developed market with global influence in diagnostic imaging. Companies like Olympus and Terumo maintain leadership positions.
• Market Access Difficulty: Moderate. Japan’s PMDA regulatory process is strict but transparent, offering a reliable path to entry for companies that meet its standards.
• Population: ~1.4B
• Estimated Procedure Volumes (2024): ~60.0M
• Market Characteristics: A high-growth market with increasing healthcare investment. Historically reliant on imports, India is now building domestic medtech capacity through its “Make in India” initiative.
• Market Access Difficulty: Moderate to challenging. Pricing controls, complex approval processes, and domestic manufacturing preferences present hurdles for Western firms.
• Population: ~52M
• Estimated Procedure Volumes (2024): ~9.4M
• Market Characteristics: An emerging powerhouse in digital health, biotech, and advanced diagnostics, with major players like Samsung expanding into medtech.
• Market Access Difficulty: Moderate. The country has a well-structured regulatory system, the Ministry of Food and Drug Safety (MFDS), but pricing pressures and cost sensitivity can impact market entry and growth.
• Population: ~26M
• Estimated Procedure Volumes (2024): ~5.9M
• Market Characteristics: A high-income, innovation-driven market with robust demand for cutting-edge medical technologies.
• Market Access Difficulty: Easier relative to China and India. Australia’s Therapeutic Goods Administration (TGA) is globally respected and closely aligned with the FDA and the EU MDR, simplifying cross-market strategies.
• Population: ~675M
• Market Characteristics: Includes a range of markets at different stages of healthcare development. Countries like Thailand, Malaysia, and Indonesia are seeing rapid adoption of both local and imported medtech solutions.
• Market Access Difficulty: Highly variable. Singapore offers one of the most streamlined and innovation-friendly environments, while others, such as Indonesia, present more complex regulatory and importation challenges.
Western companies exploring APAC must understand that market access strategies are not one-size-fits-all. From localized manufacturing to public-private R&D collaborations, success in the region depends on tailoring technology and commercialization to local needs.
All data in this article is sourced from LSI’s Market Intelligence Platform, and recent additions of LSI’s The Numbers on LinkedIn and on the LSI website.
Access LSI’s continuously updated Market Intelligence Platform.
Scan QR code for details and to request a demo.



The industry’s most comprehensive and trusted global procedure database, with coverage of 300+ diagnostic and therapeutic procedures across 37 countries.

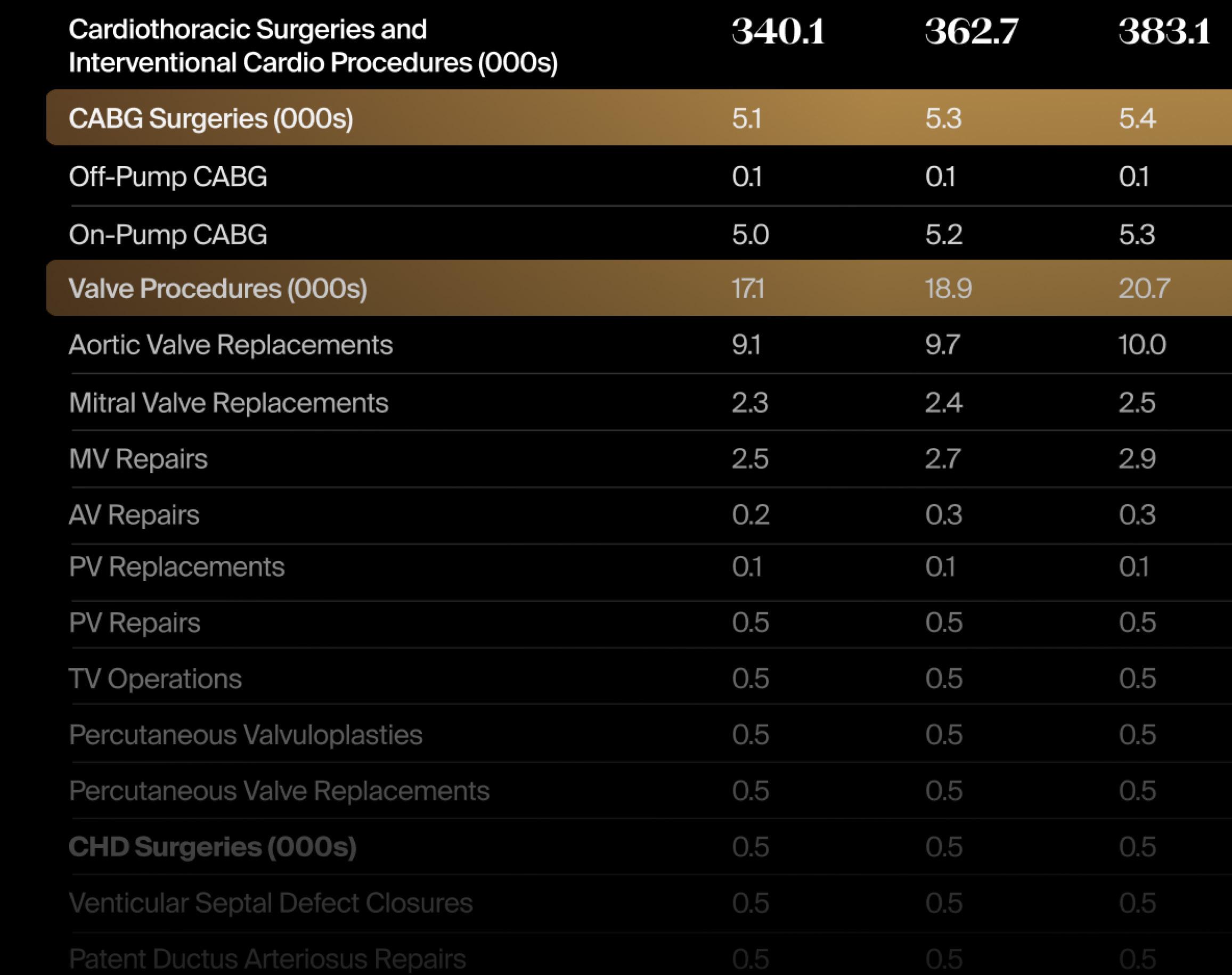

This month, to accompany our Innovator Spotlight on Fluid Biomed, we detail the latest LSI Market Intelligence data on the global, Asian, European, and U.S. forecast volume for select neurovascular stroke procedures.
LSI’s Global Surgical Procedure Volumes (SPV) Tracker provides trusted coverage of 300+ diagnostic and therapeutic procedures across 37 countries. Scan QR code to request a demo:

LSI’s SPV Tracker includes these geographies: Argentina, Australia, Belgium, Brazil, Canada, Caribbean, Chile, China, Colombia, Costa Rica, Denmark, Dominican Republic, Finland, France, Germany, Guatemala, India, Italy, Japan, Malaysia, Mexico, Netherlands, New Zealand, Norway, Panama, Poland, Russia, Singapore, South Africa, South Korea, Spain, Sweden, Switzerland, Thailand, Turkey, United Kingdom, and the United States.

Backed by a 100% success rate in its first-in-human trial, an oversubscribed funding round, and a game-changing image-guided platform for airway management, Spiro Robotics is redefining how clinicians approach difficult intubations. CEO Dimitri Sokolov shares how engineering precision, clinical validation, and a clear path to FDA clearance are positioning the company to transform emergency and surgical care.
For Dimitri Sokolov, the mission is personal. The CEO of Spiro Robotics is leading the charge to make difficult tracheal intubations safer, faster, and more predictable, because when seconds count, there’s no room for error.
With more than 30 million OR, ICU, and ED intubations performed annually in the U.S. alone and over six million considered difficult, the stakes are high. Current tools often fall short in complex cases, leaving too much to chance.
“When an airway is difficult, failure rates can reach as high as 42% in high-risk patient populations,” Sokolov explained. “Major complications leading to severe hypoxia, brain damage, and
even death continue to happen when intubation is delayed or unsuccessful.”
“When an airway is difficult, failure rates can reach as high as 42% in high-risk patient populations.”
It’s this reality that led to the development of Spiro-VISTA, a handheld image-guided intubation system engineered to improve outcomes in the
highest-risk scenarios. The device has already completed a 30-patient clinical study in Lithuania, where it achieved a 100% success rate with zero complications or adverse events, even in patients with multiple predictors for difficult airway access.
According to a January 2025 press release, the trial demonstrated the exceptional performance of SpiroVISTA in patients where conventional video laryngoscopy would typically be expected to fail. Sokolov commented, “The success of this trial confirms the potential of Spiro-VISTA to elevate airway management practices worldwide.”
Spiro-VISTA is more than a new device—it’s a reimagined platform for airway access. Designed to bridge the gap between advanced visualization and intuitive control, the system empowers clinicians to manage even the most challenging intubations with confidence.
The system is designed to provide:
• Dual-camera full airway visualization for airway assessment with real-time feedback to the operator.
• A servo-controlled flexible endoscope for precise airway maneuverability, allowing for navigation of complex patient anatomy.
• A predictable, reproducible solution that aims to reduce costs.
• A disposable system to reduce the risk of cross-contamination and facilitate ease of use.
And that’s just the beginning. With a future AI-guided mode already validated in cadaver labs, Spiro Robotics aims to fully automate intubation navigation, helping every provider, regardless of experience, perform like an expert.
Following its first-in-human study, Spiro Robotics announced the close of a $3 million oversubscribed second seed round, bringing total funding to $9 million to date. That capital has enabled the company to develop a commercial-ready device, complete preclinical and clinical validation, and prepare for regulatory submission with remarkable efficiency.
Now, Spiro is raising an additional round to support its FDA 510(k) submission, targeted for Q3 2025, and launch an early commercial rollout in collaboration with key clinical champions in early 2026. The funds will also support the development of Spiro-VISTA’s AI-guided mode, which has already shown success in cadaver testing.
In parallel, the company recently welcomed Trae Gordon as its CCO. With two decades of experience scaling medtech technologies, Gordon is helping bridge Spiro’s engineering-led
development with go-to-market execution. (Also see Power Moves, this issue.)
“Trae’s deep industry insight and proven track record of commercial excellence make him an ideal addition to our leadership team,” said Sokolov.
The company was founded by Stanford anesthesiologist Dr. Vladimir Nekhendzy during the height of the COVID-19 pandemic. Sokolov—a veteran medical device engineer with ~28 patents and a background at Johnson & Johnson, Stryker, and Potrero Medical—joined the team to help bring the idea to life.
His role has since evolved from R&D leadership to full-time CEO, where he has guided Spiro Robotics through preclinical testing, product development, and clinical validation. “I’ve always loved building things,” Sokolov said. “Now, instead of just building a product, I’m building a company.”
“I’ve always loved building things. Now, instead of just building a product, I’m building a company.”
Sokolov is clear-eyed about what it takes to succeed in medtech.
“There are easier ways to make money,” he joked. “It’s a marathon, not a sprint. Fundraising is hard. Development always takes longer than you think. But if you’re solving a real problem—and building something that matters—it’s worth it.”
His advice? “Stay scrappy. Embrace the process. And build with the enduser in mind.”
“A lot of devices live or die based on user experience,” he said. “Our clinicians didn’t just like using the device— they asked if they could keep it.”
Sokolov first presented at LSI USA in 2022 and has returned almost every year since, with increasingly compelling data and progress. He’s slated to attend again in 2026. Over the years, he has come to view LSI as a key venue for building investor relationships and accelerating strategic conversations.
“What I love about LSI is that it’s all medtech. Every conversation is relevant. It’s big enough to meet the right people, but small enough to build real relationships.”
“What I love about LSI is that it’s all medtech. Every conversation is relevant.”
Spiro Robotics is focused on securing FDA clearance, launching a soft commercial rollout with key clinical partners, and progressing toward its AI-enabled platform. The company is also advancing work under a U.S. Air Force grant and laying the groundwork for broader applications. In fact, in mid-May, the company was awarded an $1.8M AFWERX grant to support post-market clinical studies. This isn’t just non-dilutive capital; it also unlocks eligibility for TACFI, potentially adding another $2M in 1:1 matching funds alongside any private investment. That’s up to $3.8M in non-dilutive funding, dramatically accelerating the clinical and commercial trajectory.
“We’re building the commercial muscle now,” said Sokolov. “This year is about laying the foundation. Next year, we’ll be ready to scale.”
• Dimitri’s presentation at LSI USA ‘24
• Spiro’s feature in LSI’s weekly newsletter, The Memo
• Dimitri’s recent State of MedTech podcast with Omar Khateeb
• Dimitri’s recent interview with The Mullings Group

Inside John Wong, MD and team’s expectation-defying journey with their startup’s bioabsorbable brain stent technology, that has the worthy aim of reimagining how brain aneurysms are treated.
The brain is the most delicate terrain in the human body—an intricate, highstakes network of vessels, neurons, and impulses where seconds or millimeters can mean the difference between a future regained and a life lost. For Drs. John Wong and Alim Mitha, neurosurgeons-turned-founders, this is not a metaphor. It’s the daily reality that propelled them to launch their Calgary, Canada-based company, Fluid Biomed, that is advancing what could be a seismic shift in the neurovascular field: the world’s first bioabsorbable, polymer-based stent for aneurysm treatment.
Unlike traditional metal stents that remain in the brain permanently—alongside the risks and lifelong medications they entail—Fluid Biomed’s ReSolv stent is designed to do its job and then the
majority of it slowly dissolves, allowing blood vessels to progressively heal.
“We have solved the riddle of curing aneurysms,” says Wong in his March 19 presentation at LSI USA ’25 in Dana Point, CA. “We’ve taken the best of metal and the best of polymer and combined them into one product.”
And, the company has defied expectations all along the way, not only by developing a technology previously thought impossible but also by doing so outside the geography of traditional medtech hubs.
Fluid Biomed didn’t get its start in a boardroom or a business accelerator. It began in the operating room, with
two innovation-minded neurosurgeons deeply frustrated by the limitations of their tools.
Wong and Mitha met over two decades ago at the University of Calgary. Wong, newly recruited as a faculty neurosurgeon, saw in Mitha—then a resident—an unusual combination of surgical rigor and creative spark. “Even as a trainee, he was already thinking about how we could make the tools we used better,” Wong recalls.
Mitha took a detour to Harvard to earn a degree focused on biomedical engineering while completing his neurosurgical training. When he returned to Calgary, he and Wong launched a biomedical engineering lab and began exploring ways to move beyond the limitations of metal stents.
“Alim is one of those rare people you meet on your professional journey who completely changes what you think is possible,” Wong says. “He literally completed a highly technical degree at Harvard at the same time as learning the art and science of brain surgery, in order to build better tools and technology for patients.”
Wong, who also holds an MBA from Wharton School of Business and trained in both neurosurgery and interventional neuroradiology, helped lead the transition from concept to company. Today, Wong serves as CEO; Mitha is president and CTO.
Brain aneurysms are bubble-like weaknesses of blood vessels, with a risk of rupture that can lead to stroke, disability, or death. For many aneurysms, standard treatment relies on the placement of metal flow-diverting stents, which help clot off the aneurysm and reinforce the blood vessel. However, metal stents have significant drawbacks.
“They’re biologically inert, meaning the body never really accepts them,” says Wong. “And they’re permanent, so they interfere with CT and MRI scans, limit future options, and require blood thinners, sometimes for life. And, not all aneurysms are eligible for treatment with metal stents.”
Along with these concerns, many aneurysms only partially heal using available metal stents. Up to 40% of aneurysms don’t fully cure even a year after metal stent placement.
In Wong’s view, despite the urgent need for solutions and the expanding market opportunity, existing tools for managing brain aneurysms fall short.
“Today’s devices are better than what we had, but they’re not good enough,” says Wong. “Too many patients require lifelong medication or multiple interventions.”
Fluid Biomed’s bioabsorbable ReSolv stent could change that equation. “We felt physicians need a solution that fixes the aneurysm but allows the blood vessel to heal and function without significant foreign material indefinitely left
behind,” he continues. “Our stent is radically different. As it starts to disappear, it gets out of the way and gives the body a chance to heal.”
“Our stent is radically different. As it starts to disappear, it gets out of the way and gives the body a chance to heal.”
And because the polymer is invisible to CT and MRI, it enables clearer follow-up imaging and eliminates the need for costly and invasive angiograms that harbor both pain and risk to the patient.
As Wong described at LSI USA ‘25, Fluid Biomed’s bioabsorbable polymer-based technology is a hybrid— combining the familiar utility and structural confidence of self-expanding stents with the breakthrough potential of bioabsorbable materials.
“We designed something transformative but still very familiar for physicians to use that fits into current interventional workflows,” he says. “This isn’t just an incremental improvement, it’s a shift in philosophy from permanent implants to temporary healing scaffolds.”
Fluid Biomed’s design choice is strategic. The stent is flexible enough
to reach complex neurovascular anatomy, potentially allowing the treatment of a wider range of aneurysms in areas metal stents can’t address. It is also compatible with coiling, allowing physicians to combine therapeutic strategies that were previously siloed.
“Coiling can be combined with our stent, something that is very challenging for current metal designs. That’s a game changer,” adds Wong. “We’re not segmenting the market, we’re bringing it together and expanding the opportunity. We can treat more types of aneurysms, in more locations, with fewer trade-offs.”
Fluid Biomed’s stent is more than a concept—it’s a validated clinical reality.
Preclinical data have been compelling. Over 100 animal implantations, including long-term studies now up to five years, bolstered the company’s confidence to advance to human trials.
Fluid Biomed’s first-in-human trial launched just two years after closing seed funding, an exceptional timeline for a Class III neurological implant. The results, including technical feasibility, safety, and early signs of efficacy, were strong.
“In our brain aneurysm patients treated with the ReSolv stent, we demonstrated aneurysm cure remarkably quickly compared to our experience with current metal stents. We’ll be exploring this value proposition in future clinical studies,” says Wong. “From our
earliest animal data, we knew our invention was something special. Our scientific groundwork gave us the courage to move efficiently, but responsibly.”
Fluid Biomed has been busy building momentum. It launched in 2019 with under $1 million on hand, and secured seed financing from established venture capital firms in 2021, which allowed it to quickly advance from laboratory development to clinical testing.
As a major milestone, in December 2024, the company raised an oversubscribed $27 million round, in one of the largest Series A financings in medtech despite the challenges of a tough investment climate. The deal was co-led by Amplitude Ventures and an undisclosed major strategic partner. New participants in the investment syndicate include IAG Capital Partners and LifeArc Ventures, as well as returning investors, ShangBay Capital and METIS Innovative. Fluid Biomed has also built a team of 20+ full-time employees.
“We’ve been capital efficient, clinically rigorous, and focused on what matters: building something that works in the real world,” says Wong. “We have NDAs signed with many potential partners. There’s strong interest because this space hasn’t seen a meaningful leap forward in years.”
Wong adds that in today’s challenging macroeconomic environment, the Fluid Biomed team is fortunate to have several factors in their favor. “We have an intimate understanding of the clinical imperative as physicians, a deep understanding of the limitations of existing treatments as surgeons, and the ability to sequentially iterate and test the device in our own engineering lab as inventors. Added to this, when we
“We’ve been capital efficient, clinically rigorous, and focused on what matters: building something that works in the real world.”
thought our technology was ready, we have the conviction to move rapidly to a first-in-human study as clinical trialists,” he says. “Combined with an appealing and growing market in the exciting neurovascular space, the intersection of those factors was propitious and led to our successful fundraise with first-rate financial and strategic VCs.”
The company is now preparing a U.S. pivotal trial and applying for FDA Breakthrough Device designation. Wong emphasized the importance of this phase.
“As clinician-scientists, we are fortunately very familiar with conducting clinical trials. Our eye is on the prize of a pivotal study, which will help bring our technology to many more patients in need.”
Given the challenges in detecting unruptured aneurysms, it is estimated that tens of millions of people worldwide have an unruptured aneurysm. Of these, it is estimated that 1 in 10,000 people worldwide experience a ruptured aneurysm, which translates to 500,000 to 600,000 cases per year globally. According to the Brain Aneurysm Foundation, one in 50 Americans, or approximately 6.7 million people, has an unruptured brain aneurysm. Annually,
about 30,000 Americans experience a brain aneurysm rupture.
Fluid Biomed estimates that the market for devices for the treatment of brain aneurysms was valued at $1.57 billion in 2024 and will increase at a brisk compound annual growth rate (CAGR) of 9.8% through 2029. Segment expansion is projected to exceed the growth of the wider neurovascular devices market, which, according to LSI’s Market Analysis and Projections database, will increase at a CAGR of 5% during the same period (see figure below; also see the Surgical Procedures Volumes table on page 40). Market growth is largely being driven by improved diagnostic and therapeutic tools, along with increasing awareness, rather than an underlying increase in the incidence of ruptured brain aneurysms.
It’s also a high-value space. Neurovascular devices command margins several times higher than cardiovascular equivalents, and recent M&A activity signals appetite from acquirers With reimbursement codes already in place and about a $15,000+ average selling price in the neurovascular space, Fluid Biomed’s commercial model is strong.
“Brain devices are unique,” Wong says. “The stakes are high, the margins are strong, and the impact is real.”
Emerging treatment strategies for brain aneurysms offer a transformative opportunity to prevent catastrophic ruptures. With cutting-edge imaging techniques and AI-driven diagnostic tools enabling earlier detection of vulnerable aneurysms, clinicians are poised to intervene before a rupture occurs. Combined with minimally invasive approaches such as endovascular coiling, intrasaccular devices, and flow diverting stents, the potential for significantly reducing aneurysm-related morbidity and mortality is massive.
When asked for his advice to medtech innovators embarking on their own journeys, Wong says that it’s all about delivering an authentic story that resonates. For Wong and team, this includes participation at LSI events to build his company’s network and connect to the global medtech ecosystem.
“An early advisor wisely told me that as CEO, I am not selling a device but the entire startup,” he says. “They advised that I needed to convey a story that encapsulated not just the technology, but our company’s people and potential. And, the message needs to be delivered in an engaging manner that captures the attention of the potential investor so that they will be receptive to another conversation. The right message at the right time can inspire your employees to surmount obstacles and achieve what has never been done before.”
Wong also explained that traveling to meet his investor teams in person has been critical in growing those relationships, along with his team’s participation at LSI events over the past several years.
“I am a firm believer that physical and repeated contact builds connection and remains the crux of human relationships,” he says. “I came to appreciate the value of events that connect investors and startups, and based on first-hand experience, I believe the epitome is the LSI conference in
Dana Point. Scott Pantel and his team bring together high-caliber investors, entrepreneurs, and service providers in a splendid venue, support them with great infrastructure and organization, and the result is a super productive and most enjoyable conference! Many of the people you meet at LSI become close acquaintances and some, close friends.”
Fluid Biomed’s journey isn’t just about building a new tool. It’s about rethinking how, and why, we treat disease. Wong and Mitha’s backgrounds as practicing neurosurgeons give them a grounded sense of what matters in the clinic—and a boldness to imagine better.
“Ultimately, we’re trying to give patients a chance at a better outcome so they can move forward with their lives,” says Wong.
When asked what’s next, Wong says that its Series A financing has given the company fuel to accelerate and drive the company to new heights. “Our new investors are very supportive and have opened doors for us to evolve as a company. We’re scaling up our manufacturing, driving ahead with our regulatory strategy, and planning our U.S. pivotal clinical trial. Stay tuned, it’s going to be an exciting ride!”
“Every patient deserves the best possible future. Our job is to identify what stands in the way and carve a better path forward,” he concludes.
John Wong, MD, MSc, MBA
CEO and Co-founder

Dr. John Wong is co-founder and Chief Executive Officer. He is an associate professor in the Departments of Clinical Neurosciences and Radiology at the Cumming School of Medicine at the University of Calgary. He serves as Director of Cerebrovascular and Endovascular Neurosurgery and maintains a clinical practice dedicated to treating brain aneurysms and stroke.
Dr. Wong received his medical education and neurosurgical training at the University of Alberta. After fellowship subspecialization in neurovascular surgery at Yale University and interventional neuroradiology in New York City, he was recruited to Calgary in 2001.
Dr. Wong completed an MBA at The Wharton School at the University of Pennsylvania with a double major in Finance and Entrepreneurship & Innovation, and was recognized with the Dean’s Leadership Award.

Dr. Alim Mitha is co-founder, President, and Chief Technology Officer. He is a specialist neurosurgeon and associate professor at the University of Calgary.
He graduated with an gree he University of Michigan in Ann Arbor, a medical doctorate from the University of Alberta, and completed his neurosurgical residency training at the University of Calgary. During that time, he also completed a Master of Science degree with a focus on Biomedical Engineering at Harvard University and a research fellowship at the Massachusetts General Hospital in Boston. Thereafter, he completed an additional two years of clinical subspecialty training at the Barrow Neurological Institute in Phoenix, AZ.
Since 2011, he has been practicing as a Cerebrovascular, Endovascular, and Skull Base Neurosurgeon at the Foothills Medical Centre in Calgary. Registration is Officially Open

Celebrating the accomplishments of a few of the many bright stars in our LSI presenting company and speaker alumni community.
Be sure to check out presentations from these and many other innovative companies from recent LSI events on our website, at Resources > Video Library.


Netherlands-based AMT Medical has raised $25 million in Series B funding to advance its ELANA Heart Bypass System—a minimally invasive, sutureless technology designed to eliminate the need for open-chest coronary artery bypass surgery. The round was led by Bender Analytical Holding, with participation from Invest-NL, the European Innovation Council, and existing investors.
The ELANA system connects blood vessels using a proprietary clip and laser, enabling beating-heart procedures without the need for a heart-lung machine. This approach may significantly reduce stroke risk, shorten recovery time, and lower procedural costs, especially in robotic-assisted surgeries.
Proceeds will support European first-in-human trials, pursuit of CE Mark approval, and initiation of U.S. clinical studies. AMT aims to transform bypass surgery into a faster, safer, and more scalable intervention for millions of patients worldwide.

ANEUVO has secured CE Mark certification and achieved ISO 13485 compliance for its ExaStim Stimulation System, a non-invasive neurostimulation therapy designed to enhance motor function in individuals with chronic spinal cord injury. These milestones signal ANEUVO’s readiness for European market entry and affirm the company’s commitment to global quality and safety standards.
ExaStim delivers targeted electrical pulses to the spinal cord using a portable device and multi-electrode pad, offering a novel therapy without surgery or implants. The CE Mark enables rollout to initial partner sites across Europe, supporting ANEUVO’s broader mission to expand access to innovative neuromodulation treatments worldwide.

Atmo Biosciences has strengthened its intellectual property position with the full acquisition of foundational patents for its Atmo Gas Capsule from RMIT University. Under the new agreement, RMIT has transferred all associated IP to Atmo in exchange for equity, reinforcing a long-term partnership between the two organizations.
Originally licensed in 2018, the capsule technology was developed through research at RMIT and has since been advanced by Atmo into a clinically validated platform for diagnosing gastrointestinal motility disorders. The company is now preparing for FDA submission following the completion of a pivotal study last year.
With full ownership of its core IP and expanded patent filings across diagnostic and therapeutic use cases, Atmo is well-positioned to bring its realtime gut health monitoring platform to market.

Axoft has completed the first four implants in its first-in-human clinical study, FINESSE, evaluating the safety and signal quality of its novel implantable brain-computer interface (iBCI). Made with Axoft’s proprietary Fleuron material—10,000 times softer than conventional polyimide used in existing iBCIs—these implants are designed to reduce tissue disruption and improve access to deeper brain regions.
The implants were performed in patients undergoing brain tumor resection at The Panama Clinic. In each case, Axoft’s iBCI reliably recorded high-resolution neural activity across cortical and subcortical areas. Fleuron’s ultrasoft properties enabled stable recordings without drift and allowed for detection of single-neuron signals and a biomarker of consciousness, all within a 20-minute surgical window.
These early results are supported by a new research article outlining how Fleuron enables scalable, high-density, tissue-wide interfaces. Axoft plans to continue its study at The Panama Clinic and is preparing for a second trial using a next-generation device. (Stay tuned for Axoft’s LSI Alumni Innovator Spotlight next month.)

Belgium-based Azalea Vision has raised €9 million in the first closing of its €15 million Series A round to advance the development of its intelligent, connected vision platform. The funding will support the company’s first-in-human pilot clinical trial and regulatory preparations as it works toward FDA IDE approval and future commercialization.
Azalea is building a medical-grade smart lens designed to adapt in real time to the wearer’s visual needs, integrating stretchable electronics, liquid crystal optics, and wireless communication into a lens-embedded system. The platform aims to address complex vision disorders while enabling remote diagnostics and targeted ocular therapy.
The company also announced the appointment of industry veteran Robert Dempsey as Chairman of the Board. With a track record of driving major ophthalmic innovations and transactions, Dempsey will guide Azalea’s strategic expansion in the U.S. and European markets.

San Diego-based Biolinq has raised $100 million in a Series C financing round to advance its biowearable glucose monitoring platform toward commercialization. The round was led by Alpha Wave Ventures and included continued support from
major healthcare and venture capital investors.
Biolinq’s first product is an intradermal glucose sensor designed for people with Type 2 diabetes who are not on insulin. Worn on the upper forearm, the biosensor provides real-time feedback through a color-changing indicator and integrates activity and sleep data to deliver a more complete picture of metabolic health.
Proceeds from the raise will support regulatory submissions and prepare the company for launch following completion of its U.S. pivotal trial. Biolinq’s platform aims to unlock broader applications in multi-analyte sensing and redefine the future of biowearables through simplicity, precision, and accessibility.

Cala Health presented new clinical data at the 2025 American Academy of Neurology meeting, reinforcing the effectiveness of its FDA-cleared Cala TAPS Therapy for essential tremor (ET). The studies highlight that consistent home use of TAPS improves underlying tremor severity, modulates brain activity in key tremor-related regions, and reduces ET-related healthcare costs over 12 months.
Data from over 2,000 patients— including real-world evidence, randomized control trials, and mechanism-ofaction studies—demonstrate Cala’s commitment to advancing bioelectronic medicine. Cala’s system remains the only FDA-cleared, wearable neuromodulation device commercially available for tremor relief in ET and Parkinson’s disease patients, with access pathways through the VA and Medicare.
Since December 2024, Cala Health has raised in excess of $50 million in funding rounds, co-led by Vertex Growth Fund and Nexus NeuroTech
Ventures. All existing Cala investors also participated, including Action Potential Venture Capital, Johnson & Johnson (through its corporate venture capital organization, Johnson & Johnson Innovation – JJDC), Lightstone Ventures, Lux Capital, Google Ventures, OSF Ventures, Ascension Ventures, TriVentures, Reimagined Ventures, Peak6, and Fiscus Global Brain, through its Kirin Health Innovation Fund and Meiji Yasuda Future Innovation Fund L.P. (Meiji Yasuda FIF), also invested in the company in March. (Also see “Q1 2025 Medical Device Investment Roundup: A Record-Breaking Start to a Year of Reckoning,” in this issue.)

CereVasc has received its second FDA Breakthrough Device Designation for the eShunt System—this time for treating communicating hydrocephalus in pediatric patients aged 12 and older. The designation was supported by data from pilot studies and new research published in the Journal of NeuroInterventional Surgery, which found that the majority of children over age one met anatomical criteria for endovascular shunt placement using eShunt.
The eShunt system is a minimally invasive, endovascular approach designed to treat hydrocephalus without the need for open surgery. It offers a novel alternative to traditional interventions by using a percutaneous transvenous-transdural technique to implant a cerebrospinal fluid shunt.
This latest designation will allow CereVasc to engage more closely with the FDA as it advances clinical studies for pediatric use. It builds on recent positive data from the company’s U.S. investigational study in older adults with normal pressure hydrocephalus, where 97% of treated patients showed clinical improvement at 90 days.

Toronto-based Conavi Medical has raised $20 million through an upsized public offering to advance the development of its Novasight 3.0 intravascular imaging platform. Proceeds will support preclinical testing and regulatory preparations ahead of a planned 510(k) submission to the U.S. FDA in Q3 2025, along with general corporate and working capital needs.
The company’s Novasight platform combines intravascular ultrasound (IVUS) and optical coherence tomography (OCT) to deliver simultaneous, co-registered coronary imaging. The Novasight Hybrid System has already received regulatory approvals in the U.S., Canada, China, and Japan. The 3.0 version aims to enhance imaging performance and procedural guidance in complex cardiovascular interventions.
This latest raise reflects growing investor confidence in Conavi’s imaging technologies and positions the company for its next stage of commercial and clinical progress.

CorWave, a France-based developer of cardiac assist devices, has successfully completed key preclinical studies for its next-generation left ventricular assist device (LVAD), clearing the path for its first-in-human trial. The company’s device leverages a novel wave membrane pump technology that
delivers physiological, pulsatile blood flow—an advancement designed to reduce complications commonly associated with rotary blood pumps.
In a chronic in vivo study, the CorWave LVAD functioned successfully for six months, including nine ovine implants followed for 60 days without chronic anticoagulant therapy. No device failures or thrombosis were observed. The pump operated in synchrony with the heart’s rhythm, demonstrating hemocompatibility and stable performance with a self-adaptive control algorithm.
These outcomes mark the final stage of preclinical development and suggest potential clinical advantages in safety and patient quality of life. With a fully equipped urban manufacturing facility capable of producing up to 1,000 devices annually, CorWave is now preparing to initiate clinical trials and bring its smart, adaptive LVAD to patients.

EBR Systems has received FDA approval for the WiSE System, the world’s first and only leadless left ventricular endocardial pacing (LVEP) device for cardiac resynchronization therapy (CRT). This milestone introduces a new option for heart failure patients who cannot benefit from traditional lead-based CRT systems due to anatomical challenges or prior lead failures.
The WiSE System delivers endocardial pacing from inside the heart via an ultrasound-powered electrode, eliminating the need for transvenous LV leads. Results from the SOLVE-CRT trial demonstrated significant improvements in reverse remodeling and functional outcomes, including a 16.4% reduction in LV end-systolic volume and symptom improvement in over half of the treated patients.
With FDA approval, EBR transitions from a pre-commercial to a commercial-stage company, aiming to broaden access to CRT for patients previously deemed untreatable.

Egg Medical has raised $14.3 million in a round led by TVM Capital Life Science to support the growth of its EggNest system—an innovative shielding solution designed to reduce X-ray scatter exposure during interventional procedures.
Developed by interventional cardiologist Robert Wilson, MD, the EggNest is a carbon-fiber protective enclosure that surrounds the patient and has been shown to reduce scatter radiation exposure to medical staff by over 90%. Unlike traditional lead aprons, the system offers lightweight, full-room protection without the ergonomic and health drawbacks associated with wearable shielding.
With more than 200 units sold and sales doubling annually, the Arden Hills, MN-based company is scaling rapidly to address what it sees as a long-overlooked opportunity in procedural radiation safety.

Elixir Medical has announced the CE Mark approval and European commercial launch of its LithiX Hertz Contact (HC) Intravascular Lithotripsy
System (IVL). LithiX is a first-of-itskind mechanical IVL device designed to treat moderate to severely calcified coronary lesions using Hertz Contact Stress, without requiring an external energy source.
The system’s unique mechanism, featuring a balloon-mounted array of metallic hemispheres, delivers force directly to calcium-rich plaque to create wide, deep fractures while minimizing trauma to adjacent soft tissue. Early users across Italy, France, and Germany have reported strong procedural outcomes, including precise lesion modification and improved workflow during percutaneous coronary intervention (PCI).
LithiX adds a novel mechanical option to the company’s growing coronary portfolio, building on six-month results from the PINNACLE I study that demonstrated effective lesion treatment and favorable stent expansion outcomes.

Field Medical has raised $40 million in Series A financing to accelerate the development of its next-generation pulsed-field ablation (PFA) system, with $20 million in new capital and $20 million in converted seed debt. The funding will support ongoing pilot studies in ventricular tachycardia (VT) and atrial fibrillation (AF), as well as operational scale-up ahead of the company’s pivotal VT trial, VERITAS.
Field’s technology includes the FieldForce Ablation System, featuring the first built-for-purpose, contact force focal PFA catheter designed for rapid, precise energy delivery. Backed by the company’s proprietary FieldBending technology, the platform is intended to treat the full spectrum of cardiac arrhythmias with improved safety and efficiency.
The raise positions Field Medical to move from early clinical development
into commercial readiness, building on recent FDA Breakthrough Device Designation and inclusion in the TAP Pilot Program.

Dutch medtech company Hy2Care has received FDA Investigational Device Exemption (IDE) approval to initiate its first U.S. clinical trial for the CartRevive hydrogel implant, a minimally invasive solution for treating traumatic cartilage injuries in the knee. The IDE approval follows the implant’s FDA Breakthrough Device Designation in 2023 and builds on positive results from the company’s ongoing European clinical trial.
CartRevive is designed to enable optimal cartilage repair through a minimally invasive approach, addressing a major unmet need in orthopedic care. The upcoming U.S. trial is expected to begin in early 2026 in collaboration with top-tier orthopedic centers in both the U.S. and Europe.
With CE marking targeted for 2026, Hy2Care continues advancing toward commercialization in both markets as it aims to transform outcomes for patients with joint injuries.

illumicell AI, a Swiss-American startup redefining cell-based diagnostics, has raised $2 million in pre-seed funding to accelerate the development of its portable, real-time AI platform for male
fertility testing. The company’s “lab scanner” analyzes cells and biomarkers within minutes, offering clinical-grade, point-of-care diagnostics.
Co-founded by Dr. Michel Bielecki, Jeyla Sadikova, and Loup Cordey, illumicell AI is addressing the imbalance in fertility diagnostics, where men are under-evaluated despite contributing to nearly half of infertility cases. The platform has already amassed over 400,000 datapoints and is expanding its capabilities beyond semen to fluids like urine and cerebrospinal fluid. FDA submission is planned for 2026.

LEM Surgical, a Swiss developer of robotic technologies for hard tissue surgery, has received FDA 510(k) clearance for its Dynamis Robotic Surgical System. Designed to improve precision and control during spine procedures, Dynamis integrates real-time imaging, dynamic guidance, and adaptable instrumentation compatibility into a compact, navigation-based robotic platform.
The system features three robotic arms—two for surgical guidance and one for optical navigation—consolidated into a single cart that partially fits beneath the operating table. Dynamis supports a broad range of surgical instruments through adjustable end-effectors and intraoperative calibration, streamlining workflow while enhancing accuracy.
This milestone marks the U.S. market debut of what LEM Surgical calls the first-of-its-kind multi-arm robot for hard tissue surgery. Initial select hospital installations are expected this year, with broader commercialization planned for 2026. Headquartered in Bern, with U.S. offices in Tampa, the company aims to redefine robotic spine surgery with intelligent, next-generation systems. (Also see LSI’s The Memo: LEM
Surgical Bringing the Next Generation of Spine Surgery Innovation to Life.)

Mendaera, a Silicon Valley-based robotics company, has announced a strategic collaboration with Cook Medical to advance needle-based interventions using handheld robotics. The partnership integrates a trocar needle with Cook’s EchoTip technology— known for its high-visibility ultrasound guidance—into Mendaera’s in-development robotic platform for percutaneous procedures.
Mendaera’s platform combines robotics, real-time imaging, and connectivity to enhance precision and ease of use in accessing hard-to-reach anatomical targets. By pairing its system with EchoTip technology, the company aims to improve workflow and reproducibility in urological and other needle-based procedures.
The collaboration reflects both companies’ commitment to transforming minimally invasive care. While Mendaera’s robotic system is not yet FDAcleared, the partnership positions the company to deliver streamlined, highvalue solutions that meet the growing demand for precision intervention across care settings.

NeuroSigma, a Los Angeles-based bioelectronic medicine company, has
announced a new clinical trial evaluating its Monarch external Trigeminal Nerve Stimulation (eTNS) system for treating ADHD symptoms in children with fetal alcohol syndrome (FAS). The two-year trial will be conducted at UCLA, led by Professor Joseph O’Neill, and supported by a $350,000 grant from the National Institute on Alcohol Abuse and Alcoholism (NIAAA).
The Monarch system, FDA-cleared for pediatric ADHD, delivers non-invasive neuromodulation therapy and offers a drug-free alternative to stimulant medications. Children with FAS often exhibit ADHD-like symptoms— including hyperactivity, impulsivity, and executive function deficits—but are typically less responsive to standard treatments.
Phase I will enroll up to 30 children ages eight to 12 with fetal alcohol exposure. Participants will receive nightly therapy for four weeks, with parents tracking symptom progression, sleep habits, and tolerability. If successful, the study will expand into a larger, double-blind randomized crossover trial.
This milestone reflects NeuroSigma’s ongoing efforts to expand the Monarch system’s clinical applications. The company’s pipeline also includes potential uses in autism spectrum disorder, epilepsy, and learning disabilities.

NovApproach Spine has partnered with Lovell Government Services to expand access to its OneLIF interbody spinal fusion system across U.S. federal healthcare systems, including the Veterans Health Administration (VHA), the Military Health System (MHS), and the Indian Health Service (IHS). Through this partnership, OneLIF is now available on multiple government purchasing platforms, streamlining acquisition
and supporting service-disabled veteran-owned small business goals.
Designed to accommodate a range of patient anatomies, OneLIF supports anterior spinal fusion through multiple surgical approaches—including ALIF, SupineATP, and Lateral Anterior-to-Psoas. The partnership reflects NovApproach’s growing role in improving spinal care for veteran and military patient populations.

London-based OSSTEC has raised £2.5 million in funding, led by Empirical Ventures, to bring its next-generation joint replacement implants to market. Built on a decade of research at Imperial College London, the company’s first product is a knee implant that combines innovative 3D-printed articulating surfaces with biomimetic cementless fixation for enhanced functionality, superior fixation, and a long-lasting bone foundation.
With the new capital, OSSTEC plans to scale its impact through strategic partnerships and expand its presence in the orthopedic implant space. The company’s patented design aims to improve implant integration and durability, addressing long-standing challenges in joint replacement surgery.

Iceland-based digital health and therapeutics company Sidekick Health
has secured a €35 million venture debt facility from the European Investment Bank (EIB) to accelerate its AI-driven platform innovation and therapy development. The financing is backed by the European Commission’s InvestEU initiative and marks the EIB Group’s first venture debt transaction in Iceland.
In parallel, Sidekick also raised an additional €7 million capital injection from existing and new investors to support its commercial expansion. The combined funding will be used to advance digital solutions across chronic and specialty care areas, including oncology, cardiovascular disease, women’s health, and inflammatory conditions.
Sidekick partners with major pharmaceutical companies and health insurers to deliver regulated, AI-powered digital therapeutics aimed at improving outcomes and reducing healthcare costs. With this latest funding, the company plans to scale R&D efforts and expand its global footprint.

Baltimore-based Sonavex has received FDA 510(k) clearance for EchoMap, a new ultrasound device designed to simplify vascular access for dialysis patients. EchoMap combines 3D imaging and AI algorithms to help dialysis technicians visualize arteriovenous fistulas and grafts before cannulation, without requiring prior ultrasound experience.
Traditional cannulation can be challenging, especially when access points are deep or tortuous. These difficulties often lead to needle misplacement, increasing the risk of hematoma, infection, and other complications. While ultrasound has proven effective in expert hands, its complexity and training requirements have limited broader adoption in dialysis clinics.
EchoMap addresses these barriers with automated imaging in the coronal plane, offering a top-down view of vascular anatomy and minimizing the need for manual image interpretation. This makes it easier for technicians to locate the access site and avoid complications, with minimal training required.
A post-market study supported by the National Institutes of Health (NIH) will launch later this year in partnership with a large dialysis organization, further evaluating the system’s performance and impact. EchoMap is the latest advancement from Sonavex, a Johns Hopkins spinout focused on developing ultrasound technologies that deliver critical vascular insights at the point of care.

Dallas-based Spark Biomedical has closed a $15 million Series A round to advance its wearable neurostimulation platform, which delivers non-invasive, drug-free therapies across neurological health, women’s health, and hemostasis. The round was led by WAVE Ventures and included strategic support from Pathway to Cures, a venture philanthropy fund focused on bleeding disorders.
Spark’s technology uses targeted mild electrical pulses to stimulate the vagus and trigeminal nerves, modulating neural pathways to reduce symptoms and support physiological balance. This bioelectronic approach is being developed as an alternative to pharmaceuticals for a range of complex conditions.
With this funding, Spark plans to expand its clinical programs and engineering pipeline to address unmet treatment needs through cutting-edge, science-backed neuromodulation solutions. The company continues to explore new therapeutic applications that improve patient outcomes and transform care delivery.

THINK Surgical has announced the first use of Maxx Orthopedics’ Freedom Total Knee implant with its TMINI Miniature Robotic System, marking a key milestone in expanding access to open-platform robotic-assisted surgery. The procedure was performed by David Cashen, MD at Coastal Orthopedics Surgery Center in Florida.
The TMINI system is a wireless, handheld robotic device designed to support implant placement with greater precision and efficiency. Its compatibility with Maxx’s Freedom and Titan Knee systems allows surgeons to personalize procedures while using implants they already trust.
Following FDA clearance in late 2024, this first clinical use demonstrates the growing appeal of compact, flexible robotic platforms and reinforces THINK Surgical’s commitment to supporting surgeon-preferred implants across its technology.

Dever-based Transverse Medical has completed a $10 million+ Series B2 financing round to support continued development of its POINT-GUARD cerebral embolic protection device for use during transcatheter procedures, such as transcatheter aortic valve replacement (TAVR). The round was led by the
company’s legacy lead investor, a large family office, with additional participation from existing Series B1 backers.
Funds will advance the ongoing feasibility clinical study at Victorian Heart Hospital in Melbourne, expand operational capacity, and prepare the company for pivotal trials and FDA IDE submission. The POINT-GUARD system is designed to deliver full-brain protection by preventing embolic debris from reaching cerebral circulation during high-risk cardiovascular procedures.
The company’s next-generation platform aims to address procedural stroke risk through a user-friendly design that accommodates a variety of aortic arch anatomies without interfering with the procedure. The device is currently under clinical investigation in Australia.

Vivasure Medical has received CE Mark approval for its PerQseal Elite vascular closure system, the first fully absorbable, sutureless closure solution designed specifically for largebore vessel closure. The device will be launched in select European markets this summer and is intended for use following structural heart procedures such as TAVR and endovascular aortic repair (EVAR).
Unlike existing closure devices, PerQseal Elite requires no pre-procedure steps and is deployed from inside the vessel, simplifying placement and eliminating the need for permanent materials like metal implants, sutures, or collagen. The system is engineered to return the vessel to its natural state, aiming to reduce complications and improve patient outcomes.
The CE Mark represents a major milestone for the Galway-based company, which is developing a portfolio of polymer-based, minimally invasive closure technologies. PerQseal Elite builds
on the success of Vivasure’s original platform and advances the standard of care in large-bore cardiovascular interventions.

San Diego-based Vektor Medical has launched IMPRoVED AF, a randomized, controlled, multi-center clinical study evaluating whether AI-powered mapping can improve ablation outcomes in patients with persistent and recurrent AF. The trial will compare standard pulmonary vein isolation (PVI) alone to PVI plus additional ablation guided by Vektor’s FDA-cleared vMap platform.
vMap uses data from a standard 12-lead ECG to non-invasively localize electrical hotspots responsible for arrhythmias. By identifying non-pulmonary vein drivers, the platform aims to reduce repeat ablation procedures and improve long-term freedom from AF. Up to 384 patients will be enrolled across 15 electrophysiology centers in the U.S. and Europe.
In parallel, Vektor also initiated the VITAL-EP registry to collect real-world data on the clinical utility and procedural efficiency of vMap-guided ablation. Interim results from IMPRoVED AF are expected in 2026, with final results anticipated in 2027. Together, these studies aim to validate vMap’s potential to redefine the standard of care in arrhythmia treatment.
LSI events produce results. Check out inspiring stories from attendees on our website:

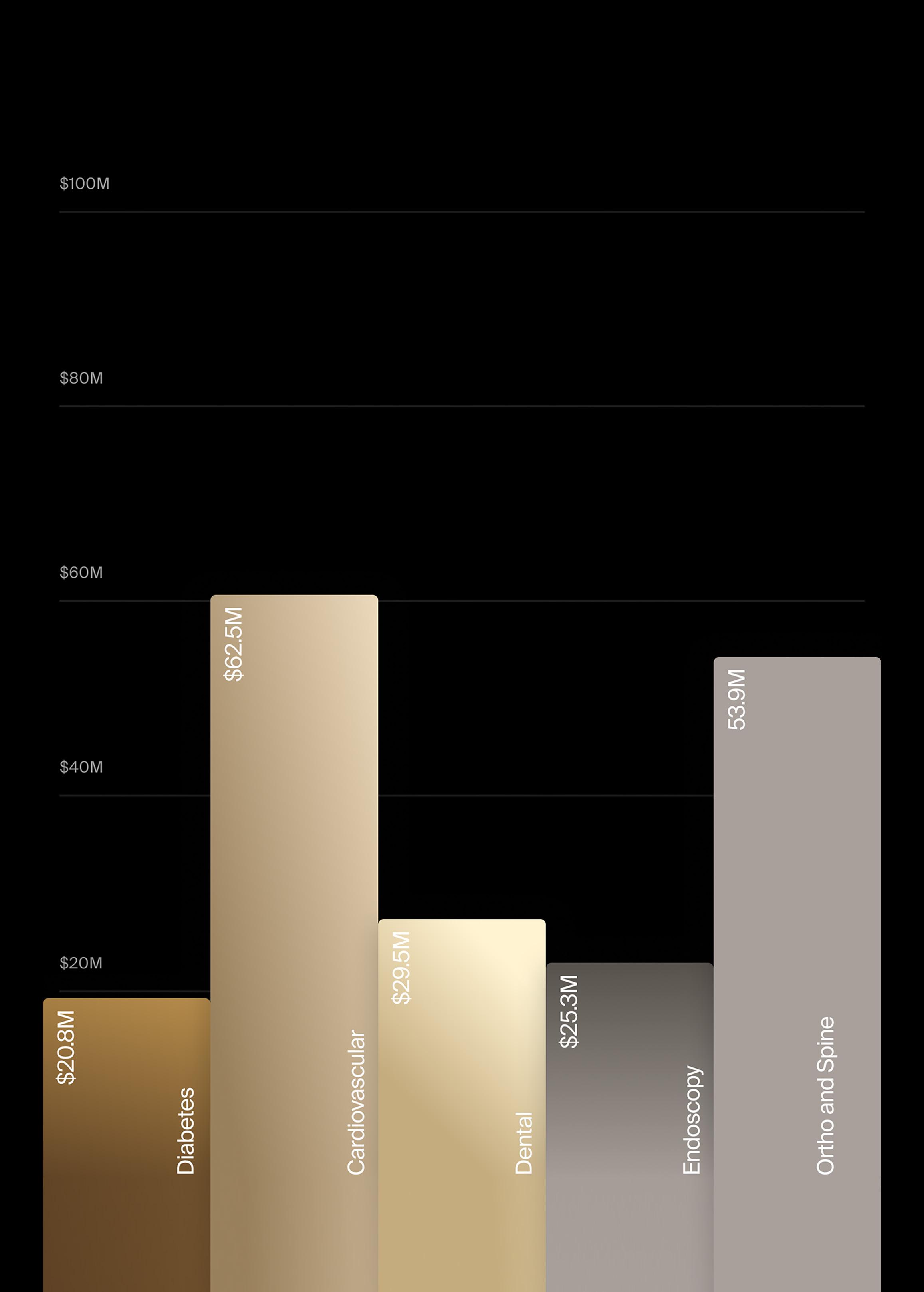
De-risk your biggest strategic decisions with our most comprehensive market data and projections (worldwide sales, share by supplier, CAGRs) on 27 major market segments and 200+ sub-segments, prepared with consistent methodologies and forecast periods.

4C Medical Technologies 16
22 Hor Invest 20
50 Partners Capital Health 20
Abbott 8, 22, 23, 24, 25, 36, 56
AccurKardia 21, 34
Action Potential Venture Capital 17, 50
Actual VC ...................................................... 20
Afrimobility 21
Alcatel 9
Alcon 11
Alim Mitha 44
Alleviant Medical 16
Ally Bridge Group 16
Alpha Medical 30
Alpha Wave Ventures 20, 49
Alven 20
Amir Soltanianzadeh 34
Amplitude Ventures 46
AMT Medical 20, 48
Ana Maiques 8
Andera Partners 16
Andrea Giani, MD 20
Andrew Glass 54
ANEUVO........................................................ 48
Annaida Technologies 21
AnthroTek 20
Anumana 21
Aopeng Medical 20
Aphelion Capital 20
Append Medical 9
Araceli Biosciences 21
Aramco Ventures 21
Arsenal Medical 9
Arthur Cummings 11
Ascension Ventures 50
Ashley Seehusen 8
Atmo Biosciences 48
Avenue Venture Opportunities Fund ....... 21
AXA IM Alts 20
Axoft 49
Axonics........................................................... 10
Azalea Vision 21, 49
B
Baiyun Financial Holdings......................... 20
BAMA 21
Banque Populaire 21
BDC Capital 20
BELKIN Vision 11
Bender Analytical Holding 48
Bill Cortelyou 21
Biolinq 20, 49
Bios Line Holding 20 Blast 21
Boston Scientific 9, 10, 15, 21, 23
Bpifrance 21
Breakout Ventures 20
BrightCap Ventures 21
LifeSci Venture Partners 20
Lighthouse Ventures 20
Lightstone Ventures 16, 50
LionBird Ventures 20
Lisa Earnhardt 8, 22, 23, 24, 25
Longitude Capital ........................................ 16
Longview Ventures 17, 20
Louis de Lillers 9, 50
Loup Cordey 52
Luminate Medical ........................................ 21
Lumira Ventures 17, 20 LUMO Labs 21 Lumus Investment Collective 21
Maxx Orthopedics 54 Mayo Clinic 17, 21
MedTechSyndicates 20
Medtronic 16, 23, 25
Mendaera 21, 52
METIS Innovative 20, 46 MgShell 20
M.H. Carnegie 17
Michael Kahana 11
Michel Bielecki 51, 52 Michael Webb 9 Michelle Strong 10 MicroPort 9, 37
MicroTransponder 16, 17 Mike Dineen 36 Mindray 37

March 16th - 20th, 2026
Waldorf Astoria, Monarch Beach, Dana Point, CA


