Solving
Wicked Problems at the Intersection of Healthcare and Technology Insights from Intuitive Foundation’s Catherine Mohr, MD

Hot Takes, Market Dives, LSI Alumni Innovator Spotlight, and more...

Wicked Problems at the Intersection of Healthcare and Technology Insights from Intuitive Foundation’s Catherine Mohr, MD

Hot Takes, Market Dives, LSI Alumni Innovator Spotlight, and more...
February 2025
February 2025


March 17th - 21st, 2025
Waldorf Astoria, Monarch Beach, Dana Point, CA
In 2024, LSI USA convened 1,500 executives from emerging companies, venture capital and private equity firms, family offices, global strategics, professional service providers, ecosystem partners and more.
Registration and applications to present are now open for LSI USA ‘25. Get in touch to learn more about the event.



Scott Pantel
Chief Executive Officer, Editor-in-Chief

Rebekah Murrietta Vice President of Media, Contributing Author

Kelly Williams Subscriptions & Enterprise Sales

Henry Peck
Chief Business Officer, Editor-in-Chief

Brenna Hopkins Sr. Content Manager, Contributing Author

Benny Tomlin Contributing Photographer

Tracy Schaaf
Managing Editor and Content Strategist, Lead Author

Matrone Sr. Marketing Manager, Contributing Author

Paul Streeto Graphic Designer

Nicholas Talamantes Sr. Director of Market Intelligence, Contributing Author

Almonte Customer Service & Fulfillment
Printed in the U.S.A.
A publication of Life Science Intelligence, Inc. (LSI) 17011 Beach Boulevard, Suite 500 Huntington Beach CA 92647
714 847 3540 tel/fax email: info@ls-intel.com
Vol 2. No. 2
February 2025
Created by LSI, The Lens leverages LSI’s deep industry relationships and proprietary market intelligence to help executives like you build lasting medtech companies. Start your Individual, Group or Enterprise-Wide subscription today!

Scan QR Code to activate your subscription.
The Lens is published monthly and available in print and electronic formats. Copyright by Life Science Intelligence, Inc. All rights reserved. Editor takes care to report information from reliable sources and does not assume liability for information published.
Interested in subscribing? email to: info@ls-intel.com
Interested in advertising? email to: kelly@ls-intel.com


Dear Readers,
To those of you reading this onsite at LSI USA ’25, or on your way — welcome.
This year’s event is our largest and most dynamic yet, bringing together over 1,500 top executives from across the medtech ecosystem: early and growth-stage companies, venture capital and private equity firms, multinational healthcare corporations, and beyond. As we gather for a week of partnerships, insights, and bold ideas, we look forward to capturing the most consequential discussions and innovations from the event in future editions of The Lens.
We step into this moment with both momentum and uncertainty. A new government administration is beginning to shape its approach to healthcare and business, and its early impact is already being felt across the industry. At LSI, like you, we remain steadfast in our mission to advance healthcare through innovation — which demands a regulatory environment that fosters efficiency and effectiveness, alongside business conditions that support long-term investment. We are closely monitoring the evolving landscape, not just to report on the ideas and policies shaping our industry, but to engage in the conversation and advocate for a strong, sustainable future for medtech innovation.
Amidst these shifts, this edition of The Lens brings us back to a fundamental question: what drives true innovation? AI breakthroughs dominate headlines daily, but Dr. Catherine Mohr’s framework for solving “wicked problems” reminds us that the most profound advancements begin with a deceptively simple question: Where do the real gaps exist between what clinicians want to do for their patients and what they can do today? And, importantly, how can we close those gaps with technology?
We also spotlight the “Power Moves” and achievements of leaders in the industry, and take a deep dive into two rapidly evolving markets:
● The cryoablation devices space, where pulsed field ablation (PFA) is reshaping treatment approaches in cardiac ablation and beyond.
● The neurovascular device market for ischemic stroke, where established leaders (Stryker, Penumbra, and Microvention/Terumo) control a significant share—but a wave of innovation stands to redefine the standard of care.
The road ahead is exciting, yet uncertain. In a fast-moving world, the only true constant is change. With this edition of The Lens, we invite you to pause, reflect, and refocus — before charging ahead with renewed optimism, clarity, and conviction.
We are honored to be on this journey with you.
All the best, Scott
Pantel and Henry Peck

Scott Pantel Chief Executive Officer, LSI Editor-in-Chief


Henry Peck Chief Business Officer, LSI Editor-in-Chief
Need fresh inspiration for your medtech company as we navigate Q125? We’ve gathered insights from some of our LSI alumni thought leaders to help you stay ahead.
“You need to believe. You need to have a plan and change your plan. So many times, I see people telling me, ‘No, I like my plan.’ I look at them and say, ‘Change your plan. You will like your new one.’ Plans have to be changed on a regular basis, not the vision.”

— Bernard Zovighian, CEO, Edwards Lifesciences
LSI USA ‘24 Keynote with the CEO of Edwards Lifesciences and Managing Partner & COO of SV Health Investors
“Relationships are more important than capital. In the face of uncertainties, strong collaboration between investors and founders builds trust and enables success, even in the toughest challenges.”

— Diana Saraceni, Co-Founder and Managing Partner, Panakès Partners
LSI Europe ‘24 Panel: Medtech’s Conundrum: Where Will Capital and Liquidity Come From?
“I’m going to steal a line from a comedian that said, ‘if you’re starving, go where the food is.’ If you don’t have money, go to the people that actually have it. Okay, so there’s a lot of medtech startups in the world. Why the hell aren’t you here?”

“Medtech has struggled as an asset class because we’ve accepted inefficiencies and failed to align with strategic buyers. To unlock our true potential, we must rethink our models and be relentless in creating value that lasts.”

— Duke Rohlen, CEO & Managing Partner, Ajax Health
LSI Europe ‘24 Keynote: The Middle Path to Innovation: Developing High-Growth Products in Slow-Growth Companies
“The opportunity to take information from the operating room, process it, and feed it back into the patient journey is one of the most powerful steps we can take to improve outcomes. What happens in the OR is no longer just one surgeon’s business—it’s everyone’s business to make care better for all.”

— Frederic Moll, Co-Founder & Partner at Sonder Capital
USA ‘23, Keynote: The Evolution of the Digital Operating Room
— Nicholas Pachuda, COO of Portfolio & Development, Peptilogics
LSI USA ‘24 Panel: Why the Heck Can’t I Raise Capital?
“Sometimes, the greatest ideas aren’t new, they’re the ones we revisit with a fresh lens, applying bold thinking to make what was once impossible a reality.”

— Alan Cohen, General Partner, DCVC
Europe
“The future of healthcare isn’t just about solving today’s problems, it’s about building a foundation for breakthroughs we can’t yet foresee. Staying true to a long-term vision is what changes the game.”

— Paul LaViolette, CEO and Co-Chairman, Pulse Biosciences; Managing Partner, SV Health Investors
LSI USA ‘24 Panel: Cracking the Code: Finding the Optimal Valuation for Successful Capital Infusion and M&A Deals
Highlighting recent leadership shifts and other announcements and accolades impacting our global medtech community.
Radiation therapy and radiosurgery company Accuray has appointed Leonel Peralta as chief operations officer (COO), effective February 3. Peralta will be responsible for leading worldwide manufacturing operations, including driving alignment of operations with corporate business goals, maximizing supply chain profitability and efficiency, and ensuring processes support customer service. He will report to President and CEO Suzanne Winter. Prior to joining the company, Peralta was VP and head of global operations for Siemens Healthineers’ ultrasound division. He’s also held roles of varying responsibility at Medtronic Diabetes, Hillrom (Baxter), KCI Medical (3M), and GE Healthcare.
Anaconda Biomed, a San Francisco Bay Area medtech company developing next-generation thrombectomy systems for the treatment of ischemic stroke, has named long-time medtech executive and LSI alumni Trent Reutiman as its new CEO. Reutiman will

establish a new footprint for the company in the U.S. as it works towards regulatory approval of its ANA5 advanced neurovascular access device, a catheter-based technology designed to enhance the efficiency of mechanical thrombectomy by facilitating the delivery of other intravascular devices such as stent retriever devices and catheters. Reutiman brings more than 25 years of executive, financial, market development, and sales operation leadership experience to Anaconda, with an extensive focus in the vascular space. He previously served as CEO of Mercator MedSystems, as VP of global
commercial operations for IDEV, and in leadership positions at ROX Medical, OmniSonics Medical Technologies, and RITA Medical Systems.
Baxter announced that José (Joe) E. Almeida, its chair, president and CEO, plans to retire, effective immediately.

Almeida, who took over as CEO in January 2016, plans to serve in an advisory capacity through Oct. 31, 2025. Baxter appointed lead independent director Brent Shafer—former CEO of Cerner with previous medtech leadership experience at Philips and Hillrom—as chair and interim CEO. In conjunction with his appointment, Shafer stepped down from his role as lead independent director, with director Nancy Schlichting tapped to assume this role. Alongside Almeida’s departure, Baxter announced that Heather Knight is taking on the post of EVP and COO, effective immediately.
LSI alum Capstan Medical has hired former Intuitive Surgical engineer Greg Dachs to lead the structural heart robotics startup’s research and development efforts as R&D head. Dachs joined Santa Cruz, CA-based Capstan Medical from 3D printing firm Carbon, where he was SVP of engineering. Dachs spent eight years at Intuitive, where he began as a research engineer in 2007, and rising to manager of mechanical engineering. Dachs’ wealth of experience in robotics, hardware, and technology scaling will be valuable in Capstan Medical’s next phase of development, according to the company.
Medtronic has appointed Thierry Piéton as chief financial officer, effective

March 3. He joins from Renault Group, where he served as CFO. Piéton brings experience in both healthcare and other highly regulated, operationally focused manufacturing companies, including Nissan Motor Co. Ltd., General Electric, GE Healthcare, and PricewaterhouseCoopers. As CFO, Piéton will be responsible for leading the Medtronic global finance organization and key supporting functions, including Treasury, Controller, Tax, Internal Audit, Investor Relations, Corporate Strategy, and Business Development. He replaces interim CFO Gary Corona, who will become senior vice president of corporate finance and remain on Medtronic’s executive committee.
Mendaera, a San Mateo, CA-based healthcare tech company and LSI alum creating a first-of-its-kind handheld

Josh DeFonzo, CEO (Source: LSI USA '23) robotic platform to revolutionize the delivery of prevalent interventional procedures, announced the appointment of Eric Davidson as its first Chief Commercial Officer. Davidson will lead commercialization efforts as the company prepares for market entry with its novel robotic technologies. Davidson joins Mendaera with more than two decades of leadership in medical robotics and

Eric Davidson, CCO, Mendeara disruptive healthcare technologies. During nine years at Intuitive Surgical, he held key leadership roles in marketing and product strategy, contributing to the evolution and global success of the da Vinci Surgical System. Following Intuitive, Davidson served as Vice President of Sales and Marketing at Auris Health, and played an important role in its acquisition for up to $5.8 billion by Johnson & Johnson (J&J).
Novadip Biosciences, a Belgiumbased, clinical-stage biotechnology company specializing in regenerative medicine, and LSI alumni company, announced the appointment of Drew Trivisonno to its board of directors as an independent board member. With three decades of experience in debt and equity financing, and 18 years as a recognized expert in M&A, Trivisonno has held vice president positions at venture capital firms and co-founded Precision Life Science Partners. Through this group, he has helped 15 early-stage life science companies navigate complex strategic and investor negotiations, supporting them in acquisitions, fundraising, exit planning and the development of commercial innovation and strategy. He has also held the role of CFO for multiple orthopedic-focused startups. His appointment will be critical in advancing the company’s 2025 strategy, including a Series C funding round targeting €50M and expansion in the U.S. orthopedics market.
NUA Surgical, an award-winning, women’s health-focused start-up spun out from the University of Galway, has welcomed Anna Nestor to the leadership team as VP of Quality. Nestor brings over two decades of experience to the role, having held senior leadership positions with major life science companies including Teleflex, Organon, Viatris, Covidien and Merck Sharpe & Dohme In addition, Anna has been instrumental in introducing innovative devices into women’s health, having managed the

Anna Nestor
effective transfer of the Jada System for postpartum haemorrhage from Alydia Health into Organon following the acquisition. Nestor will support NUA Surgical as it develops quality systems for its SteriCISION C-Section Retractor.
Onera Health, a company transforming sleep diagnostics and monitoring, has appointed Maria Sainz as

Maria Sainz (Source: LSI USA '23) the new Chair of the Board of Directors. Sainz's experience in the healthtech sector and her proven track record in strategic leadership will be instrumental as Onera Health pursues its mission to increase access to cost-effective home polysomnography through its innovative end-to-end Onera PSG-as-a-Service solution for at-home sleep diagnostics and monitoring. Sainz has more than 30 years of medtech industry experience, with senior roles in strategic planning, operations management, and business development. In addition to her skill in executing complex business strategies, Sainz is well-known for her successes in scaling medtech organizations, playing a key role in multiple exits. Most recently, Sainz was an independent board member of Shockwave Medical, which was acquired by Johnson & Johnson for approximately $13.1 billion.
Pulse Biosciences, an innovator in pulsed field ablation (PFA) technology, has appointed Jon Skinner as CFO. Skinner joins the company with significant financial experience across the healthcare sector. Most recently, he served as VP, FP&A and investor
relations at Copeland, a private equity-backed industrial company. Prior to Copeland, Skinner held the post of VP, finance and corporate development at Imperative Care, a stroke care technology developer. While at Imperative Care, Skinner also served as interim CFO of Kandu Health during its spinout and fundraising process. Other financial roles included stints at Teleflex and Axalta Coating Systems
Pulse Biosciences also announced that it has appointed LSI alumni Paul LaViolette as co-chair and CEO, to lead

Paul LaViolette (Source: LSI USA '24) the company’s strategic direction. With extensive experience scaling medtech companies and driving the adoption of innovative technologies, and also serving as Managing Partner at SV Health Investors, LaViolette will be a key part of the company’s next phase of growth.
LSI alumni company Single Pass has appointed Dan Hughes as its Commercial Lead. With over 30 years of experience in medical device sales, Hughes will lead the company’s U.S. launch with its distribution partners and build the commercial strategy for its biopsy closure device. Single Pass offers the first disposable device that prevents bleeding by cauterizing deep tissue through a guide needle.
SpectraWAVE, a medical imaging company and LSI alum focused on improving the treatment and outcomes for patients with coronary artery disease, announced that two seasoned executives are joining its leadership team. Brad Davis has been named Chief Commercial Officer (CCO), and Bill Kelly has been named Chief Financial Officer (CFO). Davis brings over 24 years of experience in cardiovascular medical devices, having worked for both large and small companies, including Guidant, Boston Scientific and CSI. Most recently, Brad was CCO at OpSens, where he led the scale-up of a similarly sized commercial team and rapidly

grew the business, successfully culminating in an acquisition by Haemonetics. Kelly will lead SpectraWAVE’s financial strategy, investor relations, and operational efficiency. He joins SpectraWAVE from Vicarious Surgical, where

Bill Kelly
he served as CFO and successfully took the company public, raising over $275 million through initial and follow-on public offerings. SpectraWAVE’s flagship HyperVue Imaging System combines next-generation DeepOCT images
and near infrared spectroscopy with workflows optimized for the cardiac cath lab, and serves as a central hub for future enhancements that will continue to empower interventionalists in their treatment decision making and optimization.
Stimdia Medical, another LSI alumni company, has appointed Allen Meacham as its new CEO, to lead the company’s growth and innovation efforts. He brings extensive neuromodulation experience to support the ongoing ReInvigorate clinical trial for the pdSTIM System. The pdSTIM System is a neurostimulation therapy that awakens and reconditions the diaphragm to support independent breathing and liberate patients more quickly from mechanical ventilation. Meacham previously served as Chief Revenue Officer for neurostimulation company Nalu Medical, and spent 15+ years at Boston Scientific, with leadership roles including VP of US Neuromodulation, and VP of US Sales.
Vitestro, a venture-backed company based in The Netherlands that has developed a unique, autonomous robotic phlebotomy device, has announced Bob Gerberich as its Chief Commercial Officer for North America. Gerberich will lead the company’s (that is an LSI alum) U.S. pre-launch market development


and commercialization efforts. With more than 30 years of leadership experience in medtech and diagnostics, he previously served as Chief Commercial Officer at Magnolia Medical Technologies, and Vice President of Sales and Marketing at Invuity.
Healthtech growth equity firm Windham Capital Partners has announced the appointment of former New York Medicaid Director, Jason Helgerson, and Alistair Erskine, MD, Chief Digital Officer and Chief Information Officer of Emory University, as Operating Partners. Helgerson and Dr. Erskine, both long-time Windham Advisors, will be joining the team to further enhance the firm's industry expertise and expand its network in healthcare innovation.
Helgerson, in addition to previously serving as New York's Medicaid Director where he oversaw a $70 billion program for over seven years and earned recognition as one of the nation's most effective healthcare leaders, is the Founder and CEO of healthcare consultancy firm, Helgerson Solutions Group. He also served as Wisconsin's Medicaid Director, where he led the state's nationally recognized Badgercare Plus program.
“LSI events are absolute game-changers and an unparalleled opportunity for us to engage with promising innovators and fellow strategics and investors in an efficient way."
Chris Eso VP, Global Head of Corporate & Business Development, M&A & Ventures, Medtronic
Dr. Erskine is Chief Information Officer and Chief Digital Officer at Emory University, including Woodruff Health Science Center and Emory Healthcare. Dr. Erskine was formerly Chief Digital Health Officer at Mass General Brigham, and brings decades of experience in digital health transformation to the Windham Capital team. At Geisinger Health System, he led the Division of Informatics, driving the implementation of advanced clinical systems and technologies. He has also served as Associate Dean of Medical Informatics at Virginia Commonwealth University and was appointed to the Virginia Governor's Health Information Technology Commission.
De-risk your biggest strategic decisions with our most comprehensive market data and projections (worldwide sales, share by supplier, CAGRs) on 27 major market segments and 200+ sub-segments, prepared with consistent methodologies and forecast periods.

Highlights: Positive new pulsed field ablation studies, AI-powered cancer detection tools advance, robotics and endomicroscopy merge, a new no-implant atrial shunt for heart failure gains FDA and investor support, and much more.
Redwood City, CA-based spinal cord stimulation (SCS) company Nevro is being acquired by musculoskeletal solutions company Globus Medical for $250 million, in an allcash transaction.
The deal, which enables Globus to move into the markets for neuromodulation and pain management, is expected to close in Q2 ‘25. Nevro’s comprehensive HFX spinal cord stimulation (SCS) platform includes the Senza SCS system and support services for the treatment of chronic pain of the trunk and limb and painful diabetic neuropathy. Nevro also provides minimally invasive treatment options for patients suffering from chronic sacroiliac joint pain.
According to Dan Scavilla, President and CEO of Globus, in a Feb. 6 press release, “This transaction furthers our mission to become the preeminent musculoskeletal technology company in the world by enabling us to expand into new markets for future growth, while gaining access to world-class, differentiated technologies that can provide significant benefit in neuromodulation, enabling technologies, and future implant solutions. We believe our size and operational capabilities will accelerate market penetration of Nevro’s technology and bring much needed and clinically superior relief to patients suffering from chronic pain.”
Convergence Medical Robotics, an Australian pre-clinical startup developing the first arthroscopic surgery robot, has raised $3.14 million in a Series A financing round, led by U.S. investor Avicella Capital. The funds will support the
commercialization of the VO1 arthroscopic surgical robot, which aims to enhance precision in surgeries. The funding follows the VO1's acceptance into FDA's Breakthrough Devices Program.
The VO1 platform is equipped with three halo-mounted, high-precision robotic arms, featuring 21 degrees of freedom. All motors have onboard controllers facilitating continuous system mapping and advance speed and force control, and the system supports both RF ablation and shaving, providing a wide range of surgical actions. The company was founded by Chris Jeffery, MD, who specializes in orthopedic surgery and won the Deep Tech category at the 2024 Australian Young Entrepreneur Awards.
Convergence Medical will also use the new funding to scale production, expand global partnerships and advance research and development efforts in preparation for a soft launch of the V01 surgical robot this year. Five hospitals will take delivery of the V01 by November 2025, and the company has opened a U.S. office in San Francisco.
Hayward, CA-based Pulse Biosciences has reported positive results from a feasibility study of its nanosecond pulsed field ablation (PFA) technology for atrial fibrillation (AF), achieving a 100% lesion success rate and a 92.4% pulmonary vein isolation rate at three months. The company plans an investigational device exemption study for clinical validation this year.
The company’s proprietary CellFX nsPFA technology delivers nanosecond pulses of electrical energy to non-thermally clear cells while sparing adjacent noncellular tissue. The company is actively pursuing the development of its CellFX nsPFA technology for use in the treatment of atrial fibrillation and in a select few other markets where it could have a profound
positive impact on healthcare for both patients and providers. (See Power Moves, this issue, for recent executive announcements from Pulse Biosciences.)
Cardiac arrhythmia ablation company CardioFocus also recently announced the publication of the 12-month results from the ECLIPSE AF trial in Circulation: Arrhythmia & Electrophysiology, demonstrating an overall 90.2% 12-month freedom from clinically significant atrial arrhythmia among paroxysmal AF patients undergoing de novo pulmonary vein isolation (PVI) using three commercial contact-force sensing focal catheters with the Centauri System’s optimized WAVE1 PFA waveform.
The system, which has received CE mark and is commercially available within the EU and UK, where over 6,000 patients have been successfully treated, was used with the Abbott TactiCath Sensor Enabled, Boston Scientific INTELLANAV STABLEPOINT, and Johnson & Johnson THERMOCOOL
SMARTTOUCH ablation catheters. Paroxysmal and persistent AF patients were treated in the study and demonstrated an overall 80.2% 12-month freedom from clinically significant atrial arrhythmia. The waveform was optimized with invasive 90-day remapping and demonstrated an overall chronic PVI durability rate of 89%, per pulmonary vein, in patients treated within the optimized PFA cohorts.

Inflammatix has received FDA clearance for its TriVerity Acute Infection and Sepsis Test, an in vitro diagnostic system that quickly profiles the body's response to infections at the point of care. The test, which delivers results in about 30 minutes, helps emergency department clinicians differentiate between bacterial and viral infections and assess sepsis risk by analyzing 30 genetic biomarkers.
The Triverity test is designed to act like a triage system to quickly direct clinicians towards the best treatment pathway for each patient. By not specifically labelling patients as having sepsis it can also be used to ensure that people with severe infections that cause other health problems such as pneumonia or cellulitis are also identified.
In September 2024, the company closed $57 million in Series E financing, led by Khosla Ventures and Think. Health. The funds will support the company’s regulatory filing and early commercialization of the company’s lead product, TriVerity.
Median Technologies, developer of eyonis, a suite of artificial intelligence (AI)-powered Software as a Medical Device (SaMD) for early cancer diagnostics, and a globally leading provider of AI-powered imaging analyses and central imaging services for oncology drug developers, has secured up to $49.9 million to support the regulatory filing and commercialization of its AI-powered early lung cancer screening software, Eyonis LCS, in the US and EU. Up to $39.2 million of Median’s funding will come from a new loan facility with the European Investment Bank (EIB), and $10.5 million from an equity line with IRIS Capital Investment
ScreenPoint Medical has demonstrated the value of its AI-powered Transpara: Breast AI screening tool in a new study. The Netherlands-based company’s Mammography Screening with Artificial Intelligence (MASAI) study of 105,000 women found that Transpara increased cancer detection by 29% and reduced screen-reading workload for radiologists by 44%.
ScreenPoint’s data highlights that Transpara detected 338 cancers among 53,043 participants, which indicates a 29% detection increase, without increasing false positives, and a detection rate of 6.4 per 1,000 participants versus five per 1,000 in the control group. According to study researchers, the MASAI screen-reading procedure emphasized the importance of radiologists having access to breast AI lesion detection and risk information at screen reading to introduce a beneficial bias. Results from the study of Transpara, the latest version of which gained clearance from the FDA in December 2024, were published in The Lancet Digital Health

San Mateo, CA-based RapidAI recently received FDA 510(k) clearance for its Lumina 3D automated 3D imaging reconstruction solution, which is designed to replace manual workflows with an AI-powered approach.
Manual computed tomography angiography (CTA) reconstructions take considerable time, with turnaround stretched to hours at times, which can delay critical diagnoses and hinder radiologists and neurointerventionalists, according to the company. Lumina 3D can accomplish 3D imaging reconstruction in minutes, while running in real time.
RapidAI says it designed Lumina 3D for large-scale applications, ensuring 24/7 availability for hospitals of all sizes. As
part of the company’s Rapid Enterprise platform, Lumina 3D seamlessly integrates into existing workflows. It enables specialists to directly visualize compressed images of the aortic arch, carotid tortuosity, and entire neurovasculature on mobile devices. This enables faster, more accurate access planning and catheter selection. (RapidAI also offers a market-leading stroke portfolio; see Market Dive: Neurovascular Devices for Ischemic Stroke, this issue.)
San Jose, CA-based Vantis Vascular has reported the first commercial use of its CrossFAST integrated microcatheter advanced delivery system in 10 complex, high-risk coronary interventional procedures. The procedures were 100% successful with no device-related complications, according to the company. The CrossFAST System is the first and only dual monorail microcatheter advanced delivery system purpose-built for complex coronary and peripheral interventions. Unlike guide extension catheters, which have limited ability to reach targeted areas in high-risk PCIs and demonstrated risk of vessel trauma and damage to previously placed stents, the CrossFAST Advanced Delivery System incorporates an integrated microcatheter with a seamless transition to lower the risk of these adverse events during catheter delivery. The CrossFAST System is powered by DuoPRO Interlocking Technology, a novel mechanism coupling the microcatheter to the outer delivery catheter, allowing the system to be advanced as one unit for enhanced pushability and efficient navigation, a significant limitation of guide extension catheters, especially in complex high-risk procedures. The system is commercially available in the U.S. as part of a limited market release. Full market release is planned for later in 2025.
Mauna Kea Technologies, developer of Cellvizio, a multidisciplinary probe and needle-based confocal laser endomicroscopy (p/nCLE) platform, recently announced promising results in a new surgical indication published in Oral Oncology Reports. Surgeons at the University of Alabama at Birmingham have demonstrated the clinical feasibility and potential advantages of using the Cellvizio confocal laser endomicroscopy (CLE) platform during transoral robotic surgery (TORS), in combination with the da Vinci SP robotic surgical system by Intuitive Surgical to enhance the precision of margin assessment for head and neck cancers.
Head and neck cancers present unique surgical challenges, particularly in achieving clear margins while minimizing tissue loss to preserve critical functions such as swallowing and speech. Positive surgical margins—meaning cancer remains in the patient’s body after resection—occur in up to 15-20% of TORS procedures according to the study, and are associated with a significantly increased risk of cancer recurrence.
This study focused on the application of Cellvizio in assessing mucosal margins during TORS for cancers of the larynx, hypopharynx and oropharynx. The Cellvizio CLE platform directly addresses these challenges by providing high-resolution, real-time imaging of cellular structures and architecture. The company’s technology allows surgeons to visually delineate tumor boundaries intraoperatively, facilitating precise resection while sparing healthy tissue.
Autonomix Medical, based in The Woodlands, TX, is focused on changing the way nervous system diseases are diagnosed and treated. The company has recently finalized the design for its radiofrequency ablation catheter, intended to treat pancreatic cancer pain by ablating nerves transvascularly. The catheter uses a microchip sensing array to detect neural signals with high sensitivity. Autonomix Medical expects to submit an Investigational Device Exemption with the FDA and begin a clinical trial in the second half of the year, to support an FDA De Novo application.
The company’s first-in-class technology platform uses a catheter-based microchip sensing array antenna that has the ability to detect and differentiate neural signals with up to 3,000 times greater sensitivity than currently available technologies. Once target nerves are identified, Autonomix uses its proprietary RF technology to ablate nerves, enabling a precision-guided sense, treat, and verify approach to addressing a number of disease categories, from chronic pain management to hypertension and cardiology. Current approaches, primarily relying on opioids or invasive ethanol injections, can provide only limited relief and may lead to risky side effects.
Carlsbad, CA-based Nalu Medical announced that the FDA cleared expanded labeling of its peripheral nerve system to include whole-body MRI use. The clearance gives physicians
greater flexibility in treating chronic pain, minimizing MRIrelated limitations with whole-body MRI-conditional use.
Nalu offers FDA-cleared spinal cord stimulation and peripheral nerve stimulation technology, the Nalu Neurostimulation System, to give patients more options for managing intractable chronic neuropathic pain. It delivers gentle electrical pulses to the nervous system to modulate pain signals before they reach the brain. The system includes a fully featured, battery-free, miniaturized implantable pulse generator (IPG). An externally worn therapy disc wirelessly powers the IPG, and the user can control it through a smartphone-based app. The company says its IPG, despite its smaller size, delivers treatment capabilities similar to larger IPGs. Nalu’s system also has an expected service life of 18 years. With whole-body MRI conditionality, the therapy can reach a wider audience with fewer restrictions.
In January, Nalu also announced the publication of a study in the journal Pain Management showing that the use of Nalu PNS therapy in chronic pain patients is associated with lower healthcare resource utilization and a significant reduction in associated costs, including a 61% overall reduction in outpatient costs.
The independently analyzed real-world study conducted by research firm OM1 evaluated medical claims data in a comprehensive database of more than 340 million patient lives. The study compared patients’ healthcare-related cost estimates 12 months before versus 12 months after implant with a Nalu micro-IPG PNS System in patients receiving an implant between 2019 and 2023. Patients treated with the Nalu PNS System experienced a 50% reduction in total medical costs in the 12-month period post-implant compared to the 12-month period pre-implant. This led to cost savings of $13,776 per patient in the 12-month post-implant period.

Austin, TX-based Alleviant Medical, a privately held medtech company pioneering a no-implant atrial shunt for heart failure, today announced two significant milestones from the FDA. The agency has granted the company investigational device exemption (IDE) approval to begin a pivotal trial focused on patients with one of the most common forms of heart failure, reduced ejection fraction (HFrEF). The agency also designated the technology with breakthrough status for this patient population.
The company’s novel shunt technology, which leaves behind no permanent implant in the body, will now be inves-
tigated for patients with the full spectrum of heart failure— both HFrEF and preserved ejection fraction (HFpEF), for which it already has FDA breakthrough designation. More than 26 million patients worldwide suffer from some form of heart failure.
In January, the company announced a $90 million financing to fund its second pivotal trial. Led by Gilde Healthcare, the round also adds Omega Funds and includes participation from existing shareholders S3 Ventures, RiverVest Venture Partners, Vensana Capital, Longview Ventures, Gilmartin Capital, TMC Venture Fund, and undisclosed strategic investors.

IMVARIA, a Berkeley, CA-based health tech company pioneering AI-driven digital biomarker solutions, announced its second FDA authorization, securing 510(k) clearance for its ScreenDx solution. ScreenDx is an AI-powered healthcare tool to assist clinicians in assessing for interstitial lung disease (ILD).
Patients with ILD suffer from frequent diagnostic delays and often undergo multiple clinical and radiological assessments before getting diagnosed. ScreenDx was developed to augment referral pathways by automatically assessing for interstitial lung findings compatible with ILD, helping flag this deadly disease in a wide range of settings. It is intended as an incidental screening tool to set patients on a path to be given an appropriate diagnosis and treatment on a timely basis.
This regulatory milestone builds on IMVARIA’s success last year with the FDA’s authorization of IMVARIA’s Fibresolve, an adjunctive diagnostic solution that uses AI to guide safe, non-invasive diagnosis of idiopathic pulmonary fibrosis (IPF). In early 2024, this signified the first-ever FDA authorization of a diagnostic tool of any type in lung fibrosis, and the first FDA Breakthrough-Designated AI diagnostic tool with simultaneously adopted CPT billing codes by the American Medical Association in any disease.
In the U.S., ILD affects approximately 650,000 people and causes between 25,000 and 30,000 deaths every year, according to the JAMA Network. It is a type of lung condition that causes inflammation or fibrosis in lung tissue, characterized by shortness of breath and difficulty getting enough oxygen into the bloodstream. In some cases, ILD can eventually lead to a patient’s need for a lung transplant, if not detected and treated in an early stage.
Samsara Vision, a company focused on bringing vision and freedom back to patients with late-stage, age-related macular degeneration (AMD) and other untreatable retinal disorders through advanced visual prosthetic devices, reported intermediate-term visual and safety outcomes of the SING IMT (Smaller-Incision New-Generation Implantable Miniature
Telescope) in patients six months post-surgery. The SING IMT is an intraocular telescope approved for use in patients without previous cataract surgery and 55 years and older in CE-referenced countries and is under investigation in the U.S. Overall, researchers found that SING IMT implantation improved distance and near vision, with a low impact on the corneal endothelium cell density and manageable safety outcomes. These positive results were also published in the journal Heliyon.
AMD is a leading cause of permanent vision loss for people aged 50 and older, and the number one cause of blindness in people 65 and older. As many as 11 million Americans are affected by some form of macular degeneration, and this number is forecast to increase to 22 million by 2050. Nearly 2 million Americans have advanced forms of AMD with associated vision loss. Similarly, approximately 67 million people in the European Union are affected by AMD, and this number is expected to grow by 15% until 2050. While treatments exist to try to slow the progression of AMD, and there are assistive devices that can help people with reduced vision see better with magnification, many patients will progress in their disease.

The industry’s most comprehensive and trusted global procedure database, with coverage of 300+ diagnostic and therapeutic procedures across 37 countries.

Outset Medical Other All-in-one hemodialysis system for use from hospital to home
843,908 shares of Series A non-voting convertible preferred stock at $200 per share
Arcuro Medical Series A All-suture,"all-inside" meniscus repair system Undisclosed Varana Capital (led), Trendlines, Consensus
Medical Series C Water vapor endourology cancer ablation therapy
Remote heart failure monitoring
Arboretum Ventures (led), Solas BioVentures (led), Orlando Health Ventures, Coloplast A/S, Tonkawa, two strategic partners
Polaris Partners (led), Elevage Medical Technologies (led), Sands Capital, Longitude Capital, Andera Partners, Gilde Healthcare, Gimv, the Ireland Strategic Investment Fund, Lightstone Ventures, Medtronic, NEA (New Enterprise Associates), Novo Holdings, Seventure Partners
Pudong Venture Capital, Zhangjiang Kechuang Investment, Zhang Keyaokun Fund, Prosperity7, Junshan Capital, Peakvest
Dimension (led), Novo Nordisk, Radical Ventures, an undisclosed leading global investment firm, InBC, Pallasite Ventures, Pangaea Ventures, Rhino
Your one-stop-shop for global market sizing and analysis, procedure volume data, startup company and deal tracking, curated insights, and more.

Source: LSI Europe ‘24
Dr. Catherine Mohr shares her journey with Intuitive Surgical as the surgical robotics leader evolved as a “29-year overnight success,” and offers lessons on how to approach what she calls the wicked problems burdening the world of healthcare and medtech. Now leading the Intuitive Foundation, she’s focused on solutions that go beyond technology, ensuring innovation meets real-world needs—for example addressing the global surgical training gap in emerging markets to save patient lives.
Dr. Catherine Mohr, President of the Intuitive Foundation and former VP of Strategy for Intuitive Surgical, where she held both research and strategy roles since 2006, brings a rare blend of VC insight and deep medtech product development experience. At LSI Europe ’24 in Sintra, Portugal this past September, she delivered a powerful keynote that spoke to Intuitive Surgical’s meteoric rise to surgical robotics market leadership—and the high-stakes impact of robotics technology on global healthcare and patient lives.
Her message, based on her two decades at Intuitive, was clear: Medtech must focus on solving “wicked problems.” She challenged the standing-room-only audience of medtech innovators to recognize the current environment, and ask where is the gap between what surgeons want to be able to do for their patients and what they can do? Also, can we fill that gap with technology?
“A lot of the problems at this current intersection of healthcare and technology are fundamentally about trying to address issues in patient care,” she notes. “These are the important problems that we are facing. But a lot of those issues have been consistent for a very long time.”
Here, we explore Mohr’s compelling ask for transformation-focused industry stakeholders, lessons learned from solving a wicked problem in lung cancer with Intuitive’s Ion Endoluminal System, and her world view on the problems that still need to be solved. Her work at the Intuitive Foundation is an inspiring example of how to deploy AI/ML technology in future-facing ways that help prevent avoidable patient deaths across the globe.
Going back to the 1990s at Intuitive, the company was always focused on solving what Mohr calls the wicked problems.
“We asked, what are the problems that we try to treat?” she says. “It’s not, ‘I have a robot, what do I apply it to?’ It’s ‘I have a really big clinical problem, the gap which I can close with robotic technology. What robot should I make to close that gap?’”
Intuitive was trying to solve the wicked problem of how to make minimally invasive surgery better for the patient. “The incision serves no therapeutic purpose,” Mohr says. “If you can make that incision as small as possible and do exactly the same surgery, it will always be better for the patient. That is the wicked problem we need to solve. Then Intuitive asked, can we do it with robotics?”
Mohr highlighted the long-runway perspective required for groundbreaking advancements in medical robotics. She traced Intuitive Surgical’s long evolution from an uncertain startup to a dominant force in minimally invasive surgery and pioneer of robotic surgery, emphasizing that success in medtech often depends on persistence, problem-solving, and strategic adaptation. And, she discussed the future-focused work going on at the Intuitive Foundation, which she leads as President, to scale surgical training so that surgeons and other healthcare professionals can save lives across the globe, including in emerging markets in Asia and Africa.
Mohr began by taking the LSI Europe ’24 audience on a journey through Intuitive Surgical’s product development. The company, founded in 1995 by Frederic Moll and Robert Younge, actually got its start in the 1980s, with the first prototypes developed by Stanford Research Institute (SRI), for military use. Moll and Younge then reworked the surgical telepresence system developed by Phil Green of SRI and developed Intuitive’s first robotic surgical prototype: Lenny, short for Leonardo. Intuitive’s second generation was Mona, in 1997, being their first robotic surgical system to be used in human trials.
In 1998, Intuitive brought into human use the first system, which is still the most successful robotic surgery platform today: da Vinci. The first da Vinci surgical system was FDA-cleared in 2000 for general laparoscopic procedures, becoming the first operative surgical robot in the U.S.
At that time, Intuitive was a start-up, with procedures numbering in the hundreds per year, says Mohr. “In 2003, there was a reverse stock split. Intuitive was struggling to keep its stock price
over $10,” she says. “Just doesn’t seem possible from where it is now. But understanding where we were in those days, we were building something that a lot of people didn’t understand, and didn’t see the future value of. But this is why I joined Intuitive Surgical. Not all the rest of the robot, the instrument, but because that was the big idea that was solving the big problem. And it’s my job to get that technology to the point where it can solve this beautiful problem. And so, after a few iterations, you arrive at elegant solutions to things. We think about robotics as futuristic, as modern— it’s really a 29-year overnight success.”
“We think about robotics as futuristic, as modern—it’s really a 29-year overnight success.”
– Catherine Mohr, MD
“Where Intuitive Surgical is now was not obvious, or even predictable from all of the stages that it was in along the way,” she continues. “There were plenty of things that could have killed the company over and over again,” she says. “By enabling robotic, single-port access, you’re putting different tools in the hands of the surgeons, who will teach you about how to use your products. And overall, the common goal has to be improving outcomes.”
Intuitive continues to create and refine robotic systems that extend the capabilities of surgeons’ eyes and hands, with the fifth-generation da Vinci 5 system FDA-cleared in March 2024. More than 14 million surgical procedures worldwide have been completed using da Vinci systems, spanning clinical specialties including urology, gynecology, thoracic surgery, general surgery, and transoral surgery.
“Today, almost every 17 seconds, someone has a da Vinci surgery,” says Mohr.
While many view the surgical robotics market today as a battle between industry giants, Mohr pointed out that the
real challenge is convincing healthcare providers to move away from traditional procedures. Rather than competing against other robotic systems, Mohr explained that Intuitive focused on improving existing surgical techniques. The goal was to minimize incisions and enhance patient outcomes, making robotic-assisted surgery a superior alternative to traditional methods.
“Your competition isn’t other companies—it’s the status quo,” she points out. “When you think about your competition, it’s generally not other companies that are trying to enter the space. It’s the established procedures of how that particular pathology is treated today.”
“Your competition isn’t other companies— it’s the status quo.”
Mohr described how Intuitive solved another wicked problem, that was more than two decades in the making. The company reached a breakthrough moment in 2019 with expansion into a new frontier: minimally invasive lung biopsy, with the introduction of the Ion Endoluminal System, a robotic-assisted platform.
The wicked problem that Intuitive was trying to solve was a surgical problem, but also a diagnostic one, describes Mohr: the big gap in lung cancer diagnosis. In lung cancer, time to intervention is critical. Collecting lung tissue samples for biopsy minimally invasively, to allow early diagnosis in this difficult-to-reach area of the anatomy, became possible only when precision robotics navigation technology advanced to the necessary level.
“We had the problem for years—we just had to wait until the technology existed to solve it,” she says. “It’s about iteration, and understanding what was great about the product that you had and then taking it to that next step.”
She explains that it all started when a new navigation technology was
developed: the shape sensor. Fiber optic shape-sensing technology measures the full shape of the endoluminal catheter hundreds of times per second, providing precise location, shape, and orientation information throughout the entire navigation and biopsy process.
“We already had our problem list, we already knew what we were trying to solve. When this technology came out, we knew what we were going to do with it. And we built a new robot around it to put the tip of the catheter in the right spot.”
Part of the problem came from difficulties going back decades with lung cancer screening, Mohr explained. In 2010, the National Lung Cancer Screening Trial was trying to do something that no trial had ever shown before, that it was cost effective and reasonable to do lung cancer screening.
“Every other trial in the past had shown that you harmed more patients than you helped if you did mass lung cancer screening,” Mohr says. “The workup associated with the nodule was so damaging that you actually hurt more people than you helped. In the early stages with lung nodules, nine out of 10 are entirely benign. And it can be very difficult to differentiate which is cancer and which is not. And so lots of people were getting surgeries, for nodules that didn’t have cancer in them about 30% of the time.”
Mohr explains that the goal of the Ion technology was to reimagine the lung cancer patient pathway, and provide definitive answers to anxious patients who learn they have a suspicious lung nodule in a more expeditious way than what typically occurs. Today, many patients are told to return months later for a follow-up CT scan to see if the nodule has grown. Patients may also require multiple biopsies prior to a lung cancer diagnosis, which can add months to a patient’s journey to care.
“Don’t underestimate the burden and the psychological burden of the workup,” says Mohr. “This is not just cost. This is real patients suffering associated with this. If the nodule has grown enough, often you’ve halved their life expectancy.”
The chief goal in designing Ion was to help shorten the patient journey by making it possible, for the first time, to biopsy small, difficult-to-reach nodules
in the peripheral lung, where more than 70% of cancerous lung nodules may be located. Ion’s ultrathin, ultramaneuverable catheter allows clinicians to reach small lesions in all 18 segments of the lung with more reach, precision, and stability, according to the company. With shape sensing, Ion provides the precision and stability needed to reach and biopsy small nodules in the periphery of the lung. Today, more than 100,000 Ion biopsies have been completed.
“And so that was a wicked problem that we had, and we had looked at for a while and we had been waiting until the technique showed up that let us get there. And so this, along with machine learning and imaging processing, is the Ion now.”
Dr. Mohr’s insights from this definitive time in Intuitive’s history offer valuable lessons for medtech innovators: the path to success is rarely linear, but a relentless focus on solving meaningful clinical problems will ultimately drive industry-changing advancements.
Taking Intuitive’s robotics technology a step further, Mohr says that the company sees a future of care that is increasingly technology-enabled and digitally informed. There’s tremendous potential to harness current and future innovations in robotics, artificial intelligence (AI), machine learning (ML), and advanced visualization to address stressors facing surgeons, physicians, care teams, and the institutions where they practice. And importantly, to deploy AI/ML technology in future-facing ways that help prevent avoidable patient deaths across the globe.
“Training is what I am most excited about using data for,” says Mohr. “Understanding how people go from being a novice to being an expert, how we think about the way of experts do something, so that we can think about best practices. We’ve been able to look at the way surgeons move, the way they optimize surgical paths, and we can think about ways in which we can bring people in that into that expert cycle.”
In Mohr’s view, training surgeons is a wicked problem that needs to be solved. And by solving this problem,
you drive adoption in a way that can be scaled worldwide, to help save lives.
In showing the Europe ’24 audience a 100-year-wide view on global health, she noted that as economies got healthier and wealthier over time, thanks to vaccinations and antibiotics available after World War II, big changes took place in surgery and surgical availability.
“If you don’t think that lack of access to surgery is a global health crisis, then you’re not looking at the same data that I’m looking at.”
“That’s resulting in about seven to 17 million avoidable deaths if somebody just had access to surgery where they were,” she says. “To put this in perspective, we have 3 million deaths a year from HIV, tuberculosis, and malaria combined. We have 10 million deaths a year globally from cancer. If you don’t think that lack of access to surgery is a global health crisis, then you’re not looking at the same data that I’m looking at.”
“It was these realizations as VP of Strategy, where I said we need to solve a much bigger problem, we need to start looking at the wicked problem of training this next generation of healthcare providers on da Vinci to meet those really critical needs, for example in Asia and Africa,” Mohr says. “Over the next 30 to 60 years, the focus will be in these rapid growth areas where we don’t have ways to train surgeons. And this is the wicked problem that I want to bring AI and ML into.”
Mohr says that in order to fully scale this goal, a shift in training philosophy has to take place. “Right now surgical training is ‘see one, do one, teach one, do one under observation,’” she says. “We need to shift it to ‘see one, teach yourself one with skill, observation and guidance, and then do one.”
This long-term wicked problem led Mohr to leave the strategy side and put on an Intuitive Foundation hat. One of the Foundation’s programs, the Surgical Education Learners Forum (SELF), develops and evaluates training modules for clinicians in low-resource settings. SELF training modules enable clinicians to independently learn and self-assess specific skills, with training modules being free and publicly available.
“We had whole hospitals in Cameroon in central Africa where everybody from the trainees all the way to the senior attendings all learned laparoscopy together without any instructors coming to the hospital. They are now doing nine to nine to 12 laparoscopic cases per week in that hospital. What’s exciting is that this year both the College of Surgeons of East Central and Southern Africa, and the West African College of Surgeons have formally adopted these methods into their training programs,” says Mohr. “When we think about trying to solve these wicked problems, a lot of the work we’re doing is looking at novel ways in which people are delivering content. We’re building these as an ecosystem, as a community of practice, and we’ve got partners from all over the world.”
Mohr describes that the economies of Asia and Africa will shape the future of healthcare, yet most training systems are designed for Western surgeon replacement economies, where clinicians are replaced with new clinicians that are coming in. Companies must rethink how they scale education and innovation globally.
“We did not build training programs for the economies of Asia and Africa that are growing. Moving forward the next 70 years, if you look at the economic development models, and the best modeling we’ve got on birth rates, and you don’t have a replacement economy. You’re looking in the wrong place for future customers if you think they’re all in the United States and Europe.”
As a closing message to the Europe ’24 audience, Mohr goes back to her initial question: what problems am I
trying to solve? She urges the audience to think about who their customers are going to be in the future, while always keeping an eye on what it is that they are doing for patients. She also asks them to think globally, recognize how the economies of the world are changing, and think carefully about what technologies they are bringing forward to address these needs and change the practice of surgery.
Mohr’s message was clear: The future of surgery is not just about building better robots—it’s about rethinking how we train surgeons, use AI intelligently, and ensure that life-saving procedures are accessible around the world. By tackling these wicked problems, the medical technology industry can drive meaningful global change.

Dr. Catherine Mohr is President of the Intuitive Foundation and previously the Vice President of Strategy for Intuitive Surgical, where she held both research and strategy roles since 2006. Prior to joining Intuitive Surgical, Dr. Mohr served in engineering project manager and new business development roles at AeroVironment. Holding a B.S. and M.S. in Mechanical Engineering from MIT and an M.D. from Stanford University, Dr. Mohr has a diverse background that covers surgery, medical technology, engineering, product design, healthcare, alternative energy, automotive, aerospace, global entrepreneurship, intellectual property litigation, U.S. Food and Drug Administration compliance, education, and product development.
The Intuitive Foundation was created in 2018 with the aim of reducing the global burden of disease and suffering through philanthropy, research, and education aimed at better outcomes for patients everywhere. The organization accomplishes this through a combination of research grants, philanthropic giving, and local volunteers worldwide.
The Foundation has committed more than $65 million to causes in support of its mission. In January, Intuitive Surgical gifted $45 million to the Foundation, bringing its total contributions to more than $170 million.
June 10th - 13th, 2025
The Ritz-Carlton, Millenia Singapore
With the global community built through LSI USA and LSI Europe and our partnerships in Singapore, China, Korea, Japan and more, LSI Asia will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Like LSI USA and LSI Europe, LSI Asia will not be held in a chaotic convention center. We have identified a world-class venue in Singapore, the gateway to Southeast Asia, for our inaugural event where senior executives can do real business.



Thank you to our founding sponsors
As we launch in this new geography, we are offering a unique opportunity to a small set of value-add, mission-aligned partners looking to elevate their global presence and develop new business.
The field of ischemic stroke treatment, where “time is brain,” is being transformed by nextgeneration hybrid thrombectomy devices and emerging robotics-assisted and AI-driven platforms, with a focus on improving clinical outcomes and reducing disability.
Stroke isn’t just a health concern—it’s a global crisis. As the second-leading cause of death and a top driver of longterm disability, stroke devastates millions of lives each year. Ischemic stroke, the most common type, occurs when a blood clot blocks oxygen flow to the brain, which can trigger severe neurological damage. With more than 77 million cases worldwide annually, a high rate of recurrence, and a staggering 3.3 million deaths, its impact extends far beyond individual patients—placing immense strain on healthcare systems, economies, and families. Adding to this burden, researchers
are warning of a rising incidence of ischemic stroke in the coming decade. The need for faster, more effective stroke interventions is growing, especially since time to treatment is critical for the survival and health outcomes of these patients. The race is on for life-saving innovations that can change the trajectory of this deadly condition.
To address this urgent need, the neurovascular device market for ischemic stroke has seen an exciting wave of innovation in recent years. Market growth is being driven by an expanding patient population and the development of
Global Neurovascular Devices for Ischemic Stroke Market Forecast, 2023-2028
Source: LSI's Neurovascular Devices for Ischemic Stroke Market Snapshot, published July 2024
new technologies—including advanced AI-driven platforms— designed to address critical gaps in patient care and improve outcomes, as an adjunct to tissue plasminogen activator (tPA), the gold-standard thrombolytic treatment.
Recent device advancements include next-generation stent retrievers and aspiration catheters designed for enhanced clot engagement and faster revascularization. Hybrid thrombectomy devices, which combine both aspiration and stent retrieval, are gaining traction for their ability to optimize clot removal efficiency. Miniaturized microcatheters and distal access catheters are improving navigation in complex cerebrovascular anatomy, allowing for better reach in small, distal vessels. Emerging robotic-assisted thrombectomy systems promise greater precision and faster procedural times, while AI-powered imaging platforms are revolutionizing stroke diagnostics and clinical decision support by enabling real-time clot characterization and automated triage. These and other innovations are reshaping ischemic stroke care and expanding access to life-saving treatments worldwide.
With this level of market excitement and innovation as a backdrop, total global sales of neurovascular devices for the
The global market for neurovascular devices for the treatment of ischemic stroke consists of several large players holding about 75% combined market share, and a growing field of other device, robotics, and AI-focused innovators.
As of 2023, Stryker was the leading supplier in this market (see figure). For over a decade, the company has built up a comprehensive global portfolio of ischemic and hemorrhagic stroke solutions through a combination of strategic mergers and acquisitions, and product development.
Another leading market player, Penumbra, offers a broad portfolio of thrombectomy products, including computer-assisted vacuum thrombectomy (CAVT). The company is focused on removing blood clots from head to toe with speed, safety, and simplicity. It has achieved strong adoption by clinicians, with sales for the Penumbra neurovascular system increasing more than four-fold from 2014 to 2023 to $373 million, and rising 20% just from 2022 to 2023.
Last year, Penumbra received FDA clearance for and launched the Lightning Flash 2.0, its next-generation CAVT system to remove venous thrombus and
treatment of ischemic stroke are projected to increase at a compound annual growth rate (CAGR) of 5.1% during the forecast period covered by this analysis, from about $1.5 billion in 2023 to more than $2 billion in 2028 (see figure on previous page), according to LSI Market Intelligence. Products included within the scope of this analysis include:
• Neurovascular microcatheters, guidewires, guide catheters, and sheaths
• Neurovascular stents
• Thrombectomy devices
• Stent retrievers
• Aspiration devices
Strong market adoption is reflective of the evolving approach to the treatment of acute ischemic stroke, with a focus not just on removing thrombus but on improving clinical outcomes and reducing disability. Revenues are also being driven by growth of the at-risk global population, related to trends in modifiable risk factors such as high blood pressure, diabetes, obesity, smoking, and sedentary lifestyle.
treat pulmonary emboli. Lightning Flash 2.0 features advanced Lightning Flash algorithms, designed for increased speed and sensitivity to thrombus and blood flow. These new advancements combined with Penumbra’s novel catheter technology allow physicians to better navigate the body’s complex anatomy and deliver
high power for clot removal with possible minimal blood loss. In January, the company launched its Element Vascular Access System, the first laser-cut hypotube sheath designed for venous thromboembolism (VTE). The system is compatible with Lightning Flash 2.0, effectively completing Penumbra’s VTE platform.
Notes: Figures may not sum due to rounding. The “Others” category includes Abbott, Cerenovus/J&J, Cordis, InNeuroCo, and MicroPort Scientific, among others.
Sources: Annual company reports, SEC filings, investor relations materials, and LSI Market Intelligence
Attracted to a large and growing global market and the potential to further impact ischemic stroke patient outcomes and cognitive function in novel ways, a large number of medtech and healthtech start-ups are innovating in this space. A few of these promising companies, including our LSI alumni, are highlighted below.
Anaconda Biomed, a medtech company developing next-generation thrombectomy systems for the treatment of ischemic stroke. At the heart of its product portfolio is the ANA Funnel Catheter, which is designed to assist in neurovascular procedures by facilitating the delivery of other intravascular devices (i.e., stent retriever devices). The device consists of a radiopaque braid covered with a polymeric coating to enable local flow arrest. (Also see Power Moves, this issue.)
Israel-based Inretio Medical Device is developing the PREVA clot retriever for the treatment of ischemic stroke. PREVA features a distal basket to ensnare and encapsulate clots in the brain and protect the brain from sub-clots breaking off during the thrombectomy procedure.
Julier Medical SAS is a Paris-based, privately held, preclinical medical device company that is developing a next-generation mechanical thrombectomy solution for the removal of clots and revascularization of patients experiencing acute ischemic strokes. The device’s thin and flexible structure allows for easy navigation through the brain's tortuous vessels, and can be expanded and collapsed at will for more effective clot removal through larger aspiration diameters, according to the company.
Magneto Thrombectomy Solutions, established as part of Incentive, Peregrine Ventures' Incubator, is evaluating a catheter-based solution to transform the way thrombi are retrieved. The company's eTrieve system is based on the understanding of the electric properties of clots, which allow the Magneto catheter to function as a magnet for clots through the use of electric

attractive forces. An immediate benefit of this electric-based approach is the device does not place radial force on the blood vessel. The device will enable the safe and efficient removal of all types of clots from all types of different blood vessels.
Miami, FL-based Navigantis recently announced the first successful neurovascular clinical cases performed with its Vasco interventional robotic platform. The company designed its Vasco platform for patients with neurovascular disorders, including acute ischemic stroke. It conducted first-in-human cases in 2022. In those cases, the platform’s previous generation successfully performed robotic embolization of liver tumors. Navigantis said its neurovascular interventional robotic clinical trial includes patients undergoing diagnostic cerebral angiogram, brain tumor embolization and mechanical thrombectomy for ischemic stroke.
Irvine, CA-based Neurovasc Technologies is focused on developing a portfolio of novel catheter-based technologies to facilitate broad treatment options for patients suffering stroke and other neurovascular diseases. The NeuroVasc ENVI-SR is a next-generation clot retrieval device for use in patients experiencing acute ischemic stroke, that was developed as a stent-retriever, and the unique articulating segmental design allows the device to remain open under tension. The device has received CE Mark and is currently under investigation in China.
Progressive NEURO, a Silicon Prairie Center-incubated company, is designing devices that can capture clots in a single pass, prevent clots from escaping to unaffected territory, and minimize vessel wall trauma.
Miami, FL-based RapidPulse is currently undergoing FDA-approved clinical
trials for its innovative cyclic aspiration system, the RapidPulse system, designed to significantly improve the success rate of treating acute ischemic stroke by achieving high first-pass reperfusion rates. RapidPulse has developed a complete system including a proprietary console to deliver precise, pulsed aspiration, highly trackable catheters, and a smart tubing set which ensures that the company’s proprietary cyclic algorithm is tuned to each catheter size. RapidPulse’s technology is a spin-out of the medical device incubator, Syntheon 2.0.
On the diagnostics side, Sense Neuro Diagnostics is leading the way in creating scalable technology to detect, measure and monitor the brain. As CEO

Geoffrey Klass frames the problem, there has historically not been technology to triage people effectively in the field or monitor their condition in the neuro ICU. And when “time is brain,” these are people that need to receive treatment in minutes, not hours. Enter Sense Neuro, and their non-invasive technology aiming to enable rapid, comprehensive detection of stroke by subtype and traumatic brain injury, and enable continuous brain injury monitoring. View the company’s presentation on “Non-Invasive Brain Scanners” and their Studio Interview from LSI USA '24 in the Resource Hub on the LSI website.
San Mateo, CA-based Route 92 Medical is focused on improving outcomes for patients suffering from acute ischemic stroke. It offers a portfolio of neurovascular solutions, including the FreeClimb access catheter, the Base Camp sheath, and Tenzing delivery catheter to streamline access to diseased segments of the brain. In September 2024, the company announced a new $50 million extension to its previously announced oversubscribed Series F financing, bringing the total raised to $82 million.
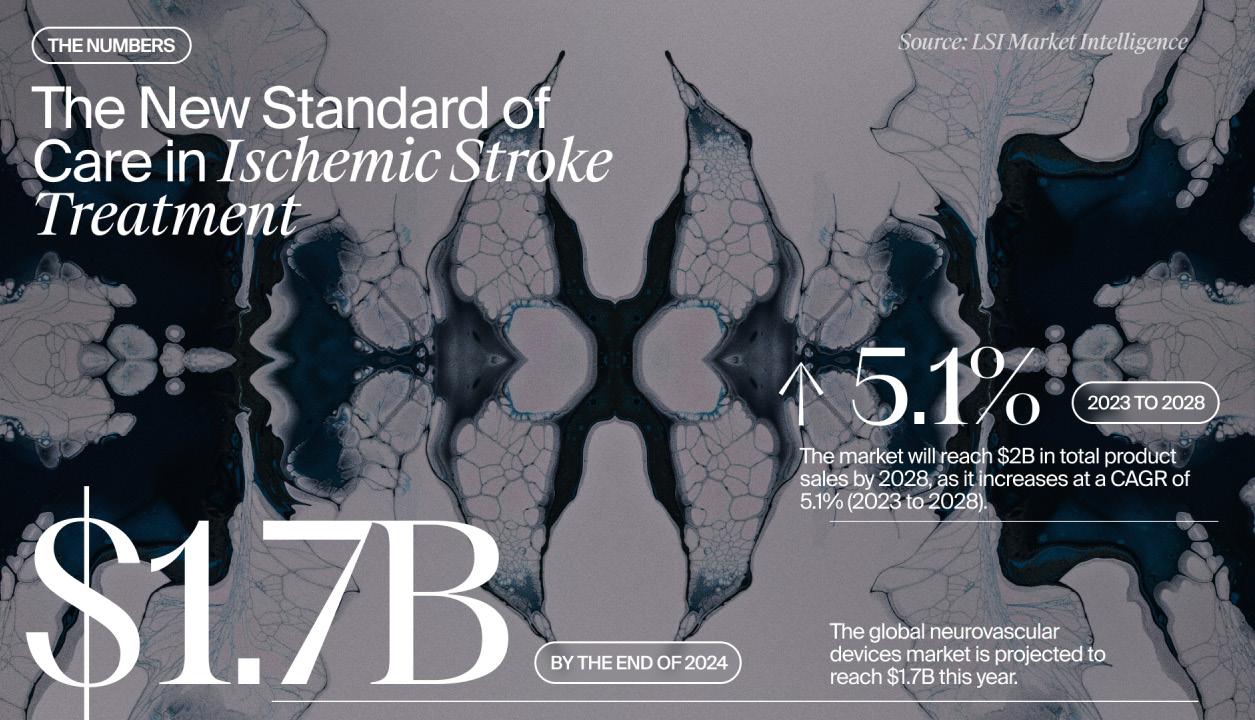
Leveraging data from LSI's Market Intelligence Platform, our analysts estimate that the global neurovascular devices market will reach $1.7B by 2024, and eclipse $2B in total product sales by 2028, as it grows at a CAGR of 5.1% over the 2023 to 2028 forecast period.
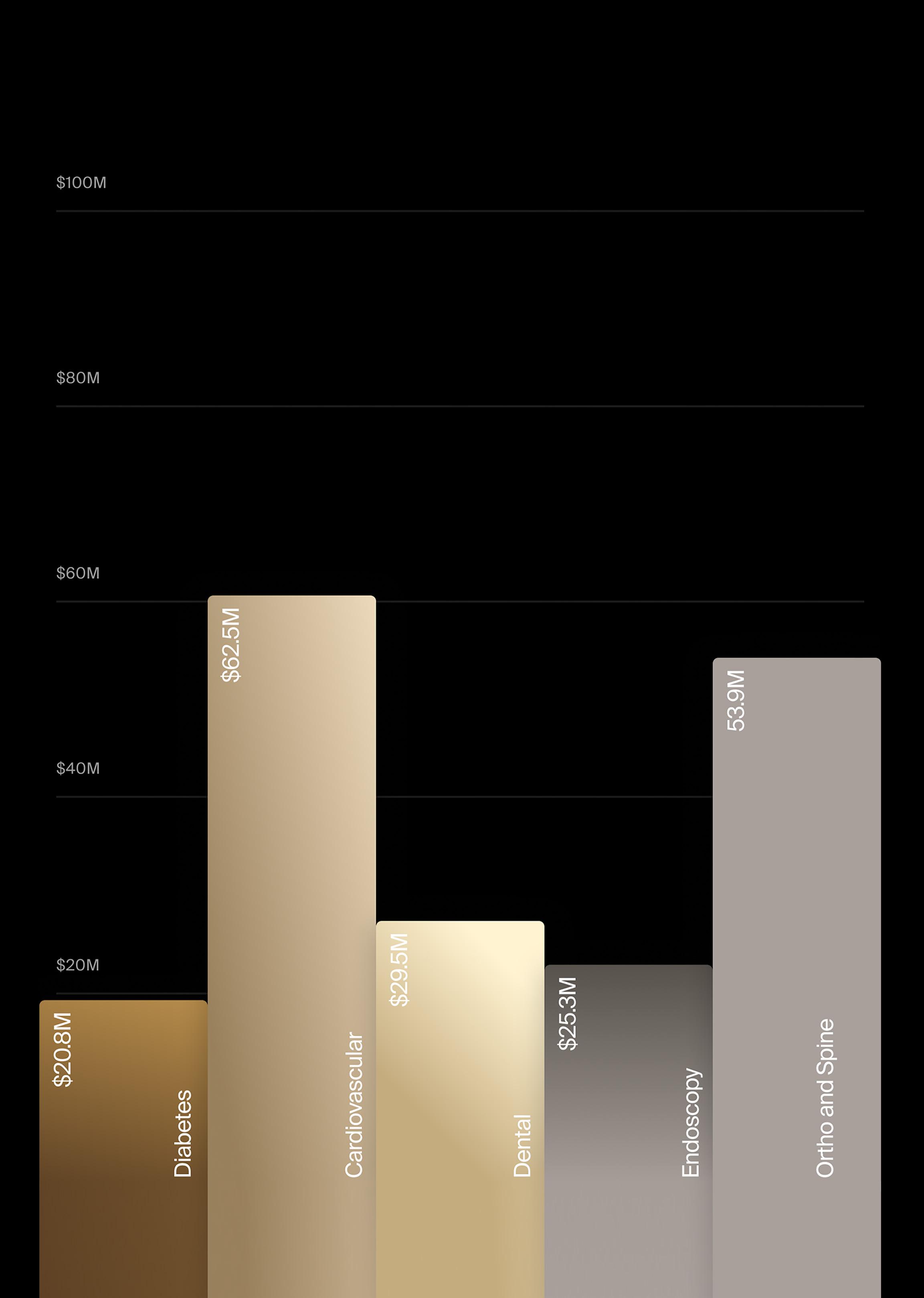
Recently updated data in LSI's Global Surgical Procedure Volumes Database projects that approximately 168,000 ischemic stroke procedures will be performed in the U.S. in 2024, a fraction of the 700,000+ annual ischemic strokes (for more procedure volumes data, see pages 36-37 in this issue).
Limited access to, and delayed arrival at, stroke-ready facilities remains a significant barrier for patients to access effective and timely treatment.
• Among the U.S. stroke procedures performed, mechanical thrombectomy is seeing increased adoption and projected to continue growing at a 4.0% CAGR (2023–2028).
• Neurovascular stents, which can be placed in tandem with thrombectomy, are seeing even faster domestic growth, with U.S. volumes growing at a 6.4% CAGR (2023–2028).
• Most importantly, outcomes for thrombectomy patients are relatively better, with 50%+ of thrombectomy patients achieving functional independence compared to 20–30% with thrombolysis alone.
In stroke care, every minute counts in protecting and saving the brain. Towards this end, innovations in stroke care reflect rapid evolution of devices and digital technologies, pharmaceuticals, and procedural strategies to enable faster, precision treatment and management.
• Imaging-Based Selection: Techniques like CT perfusion and diffusionweighted MRI help clinicians identify salvageable brain tissue and expand treatment eligibility.
• Mobile Stroke Units (MSUs): Ambulances and mobile vehicles with CT scanners, stroke specialists, and other portable technologies reduce prehospital delays, and improve access to timely intervention.
• Next-Generation Thrombolytics: Faster, safer clot-dissolving agents that may extend time windows for administration are under investigation.
• AI-Driven Workflow Optimization: AI tools will help prioritize and streamline decision-making, accelerating clinical decisions and treatment delivery in emergencies.
Robotics-assisted and AI-driven platforms are areas of transformative innovation in stroke management, with products designed to assist healthcare professionals in the swift and accurate assessment and treatment of ischemic stroke patients. A few game-changing technologies on the market or in development, including from our LSI alumni, are outlined here.
Since launching as a spin-out from the University of Oxford, Brainomix has pioneered the development of an AI platform that automates validated imaging biomarkers to improve both diagnosis and treatment decisions, and transform stroke treatment. Brainomix 360 is a comprehensive, FDA-cleared platform powered by the most advanced and clinically validated AI algorithms built around basic imaging—most notably through the company’s flagship e-ASPECTS module. The patented AI tool assesses non-contrast CT scans to automatically generate an ASPECTS score and features a unique heatmap that enables a more nuanced assessment of each region.
In February at the International Stroke Conference, Guilford, CT-based Hyperfine, developer of the world’s first FDA-cleared AI-powered portable magnetic resonance (MR) brain imaging system—the Swoop system and LSI alumni, announced the presentation of two studies which provide evidence supporting the value of AI-powered portable MRI in acute ischemic stroke triage and diagnosis.
Notably, this includes patient data in the Acute Ischemic Stroke Detection with Portable MR (ACTION PMR) study. Ohio State University, an ACTION PMR site, presented its data from the study showing that physicians were able to use Swoop system images to diagnose 90% of acute infarcts seen on conventional highfield MRI—the standard of care—and also demonstrated DWI-FLAIR mismatch, a key method of identifying acute ischemic stroke. Researchers from Massachusetts General
Hospital also presented data on stroke detection, showing that images from the Swoop system enabled accurate differentiation of stroke versus stroke mimics with strong agreement to highfield MRI, indicating the Swoop system’s value in evaluating patients with suspected stroke and transient ischemic attack. (Read more about Hyperfine in Power Moves, this issue.)
Zurich, Switzerland-based Nanoflex Robotics, an LSI alumni company, is building a next-generation telerobotics interventional platform to enhance

Matt Curran, CEO & Co-Founder, Nanoflex Robotics (Source: LSI USA '24) access to life-saving procedures. The first target application of the company’s Remote Intervention System is to enable telerobotic mechanical thrombectomies in acute ischemic stroke patients where timely intervention is critical. The system combines unique advanced magnetic navigation technology and ultra-flexible endoluminal robotic tools to give physicians greater control and dexterity over procedures, allowing for safer and more precise interventions.
The RapidAI stroke portfolio combines advanced imaging and workflow to enable physicians to make faster, more accurate triage or transfer decisions. The company’s market-leading RapidAI software analyses brain images to help decide whether a patient needs an operation or medication to remove a blood clot. The technology automatically detects large vessel blockages and shows blood circulation to the brain in detail. The software then gives physicians a better idea of a patient’s neurovascular status.
In February, the San Mateo, CAbased company received FDA 510(k) clearance for Lumina 3D by RapidAI— an industry-first, automated 3D imaging reconstruction solution designed to replace time-consuming manual workflows with an AI-powered approach
based on the latest groundbreaking AI technology advances.
In May 2024, Rapid Medical announced the first successful robotic thrombectomies with its Robotic TIGERTRIEVER, a unique endovascular thrombectomy device that adapts autonomously to the patient’s anatomy. With the press of a button, the AI-activated TIGERTRIEVER captures the blockage with active integration. The device’s radial force is then automatically reduced to minimize trauma to the brain as it is retrieved.
This January, the company announced the first patient enrolled in its COGNITIVE Study—the first to examine a link between mechanical thrombectomy and cognitive improvement. The study is looking to show that its TIGERTRIEVER device may uniquely preserve cognitive function with a tailored approach to removing thrombus while limiting impact to the natural vessel. Rapid Medical plans to enroll up to 450 patients at 50 sites.
During a cardiovascular emergency such as a stroke, fast treatment is critical. San Francisco, CA-based Remedy Robotics, which is presenting at the upcoming LSI USA ‘25 event in Dana Point, CA (Mar. 17-21), is building the world’s first remotely operated endovascular robot to enable specialists to treat all patients immediately regardless of their location.
Viz.ai, the pioneer in the use of AI algorithms and machine learning to increase the speed of diagnosis and care across 1,700+ hospitals and health systems in the U.S. and Europe, recently announced results from a clinical study recently published in The Neurohospitalist highlighting the transformative impact of the Viz Connect care pathway on post-stroke patient care. The study demonstrates that the incorporation of Viz Connect drove improvements in cardiac monitoring utilization, care efficiency, healthcare provider experience, and patient satisfaction.
These data stem from a recent collaboration between Viz.ai and Medtronic to leverage AI-driven solutions to streamline communication between neurology and cardiology teams and accelerate critical interventions for stroke patients. The AI-powered Viz.ai One is an intelligent care coordination solution that
identifies more patients with a suspected disease, informs critical decisions at the point of care, and optimizes care pathways and helps improve outcomes.
XCath, based in Houston, TX and also an LSI alumni company, is developing next-generation endovascular robotic

systems and steerable guidewires to treat cerebrovascular disorders and other life-threatening conditions. In May 2024, the company announced the successful completion of a first-of-its-kind live telerobotic trial for stroke treatment, which took place between Abu Dhabi, United Arab Emirates (UAE) and Seoul, South Korea, at Abu Dhabi Global Healthcare Week. (Also see LSI’s February 16, 2025 edition of “The Memo” on LinkedIn, featuring insights from Eduardo Fonseca on his dual leadership roles at XCath and EndoQuest Robotics.)
Data in this article is partially sourced from the LSI Market Intelligence Platform, including the Global Surgical Procedure Volumes (SPV) database and the Neurovascular Devices for Ischemic Stroke Market Snapshot (published July 2024).
Presenter photos are from recent LSI conferences; visit lifesciencemarketresearch.com for details on upcoming events.
Looking for more global Neuro market intelligence and insight?
Access LSI’s continuously updated Market Intelligence Platform. Scan QR code for details and to request a demo:


Minimally invasive cryoablation, that harnesses extreme cold to destroy cancerous and other abnormal tissue across a variety of specialties, is heating up the ablation market, thanks to precision image-guided advancements and innovative new devices.
Cryoablation, the targeted freezing of tissue to produce selective cellular necrosis, is widely used as an alternative to surgical resection or thermal therapies throughout the body. The minimally invasive modality, also associated with reduced side effects compared to surgery, has gained additional traction with the use of precision image-guided techniques and innovative new devices, according to LSI Market Intelligence. It also has distinct advantages over other modes of ablation such as radiofrequency, microwave, and ultrasound, in that providers have greater control of the shape of the ablation zone and ability to mitigate pain, according to research (Kwak K., et al. Theranostics, 2022 Feb 14;12(5):2175-2204).
Cryoablation—also referred to as cryosurgery or cryotherapy—is currently being used across multiple clinical applications, including tumor ablation for both palliative and therapeutic use (primarily liver, prostate, and kidney tumors), heart arrhythmias including atrial fibrillation, skin cancer removal, menorrhagia treatment, malignant endobronchial obstruction, and other conditions. It is considered a prime candidate to achieve synergy with adjuvant therapies (e.g., chemotherapy, immunotherapy, radiotherapy, etc.) for complete local destruction of cancer tissue.
Market growth drivers include trends in cancer incidence, particularly for liver, prostate, and kidney cancer (+2.6%/ year, 2022-2030); skin lesion incidence trends (+2.8%/year for melanoma and non-melanoma skin cancer); trends in menorrhagia, which are primarily related to the size of the female population of child-bearing age (+0.2%/ year); and trends in incidence of diseases such as Barrett's esophagus, according to analysis by LSI Market Intelligence.
Increased levels of adoption for cryoablation are reflected in projections both for unit volume and device sales. During the 2023-2028 forecast period covered by this analysis, global unit volumes for cryoablation devices (excluding those indicated for use in cardiac ablation; see the sidebar on page 33 for commentary on innovations in this segment) are projected to increase at a CAGR of 7.7%, from approximately 470,000 units to 683,000 in 2028, according to LSI Market Intelligence. Unit volume growth is strongly correlated with epidemiologic factors, such as the rising incidence of diseases applicable to this technology.
Total global sales of cryoablation devices are projected to increase at a CAGR of 7% during the forecast period, from approximately $195.8 million in
2023, to over $275 million in 2028 (see figure below). The market is growing at a slightly slower rate compared to the projected unit volume growth, as a result of a steady decrease in the average selling price of cryoablation devices. A primary reason? Competition is heating up. More than 15 companies compete in the global cryoablation device space, with a host of startups working to bring innovative new technologies to providers and patients.
In 2023, Boston Scientific was the leading global supplier for cryoablation devices, with a 37.9% share and $74.3 million in product sales. The company’s market dominance is attributed to its 2019 acquisition of the former global market leader, BTG plc.
Two established cryoablation players, Easton, PA-based CryoConcepts and Marietta, GA-based Erbe, each hold approximately a 13.8% share. Other manufacturers in this space include AtriCure, Brymill Cryogenics, Cryoprobe, CryoSurgery, Cooper Surgical, Endocare, Grand Cryo, KryoLife, and Mectronic Medicale, among others.
1.4M Procedures
According to LSI's Surgical Procedure Volumes database, approximately 1.4 million cardiac ablation surgeries were performed worldwide in 2024.
8.8% CAGR (‘24-’29)
Global cardiac ablation procedure volumes are forecast to expand at more than double the growth rate for cardiac surgeries.
Source:LSIMarketIntelligence
The FARAPULSE PFA System used in over 200,000 patients worldwide to date, was FDA-cleared in January 2024.

THE NUMBERS PulseSelect PFA system FDA-cleared in December 2023. Only PFA system indicated for both paroxysmal and persistent AFib.
Source:LSIMarketIntelligence
Cardiac ablation is making headlines again, and again.
Especially in the treatment of atrial fibrillation (AF), we are witnessing a transformative shift in how new technologies are being utilized. Over the past four decades, innovations in cardiac ablation have redefined the atrial fibrillation (AF) treatment landscape, offering safer and more effective options with improved outcomes.
For decades, radiofrequency (RF) ablation and cryoablation have dominated AF treatment. Introduced in the mid-1980s and early 2000s respectively, these energy modalities transformed AF management by enabling electrophysiologists to precisely target and isolate arrhythmogenic tissue in the pulmonary veins.
• Radiofrequency Ablation: Harnesses thermal energy to create controlled lesions that disrupt erratic electrical signals. Although highly effective, it carries the risk of collateral damage to surrounding structures.
• Cryoablation: Uses extreme cold to freeze tissue, offering a different method of lesion creation. While it simplifies pulmonary vein isolation, it sometimes falls short in terms of precision and durability.
A departure from thermal approaches, PFA utilizes nonthermal electric fields to selectively target cardiac cells while preserving the surrounding tissue.
The rapid adoption of pulsed field ablation (PFA) is fueled by multiple compelling advantages over RF and cryoablation:
• Enhanced Safety: PFA significantly reduces the risk of damaging adjacent structures, such as the esophagus and phrenic nerve, addressing major concerns with traditional ablation methods.
• Faster Procedures: Early studies indicate that PFA can significantly shorten procedure times compared to RF and cryo, boosting efficiency in high-volume electrophysiology labs.


The VARIPULSE PFA System received FDA approval in November 2024, and has been used in more than 3,000 commercial cases.
Abbott plans to file an FDA submission, receive CE Mark approval and launch its Volt PFA System outside the U.S. in 2025.
• Superior Efficacy: Promising durability in early clinical trials means fewer repeat ablations over time, especially notable when 20%-40% of current catheter ablation patients require a second procedure.
• Regulatory Momentum: Major strategics are aggressively developing and acquiring PFA technologies, underscoring its potential to become the new industry standard.
With large-scale clinical trials underway and increasing regulatory approvals, PFA has all the tailwinds to overtake traditional ablation methods as the dominant technique for AF treatment. And, it will fuel global cardiac ablation market growth at an 8.8% CAGR through 2029, according to LSI projections.
Along with a number of large strategics, as noted in the image below, the LSI Market Intelligence team is also tracking a number of start-ups that are developing their own ablation technologies. PFA innovators on the LSI radar include:
• AtriAN Medical: Gentle energy, micro Pulsed Electric Fields (mPEF), to only ablate hyperactivity; other tissues such as the myocardium are spared. (See CEO Ken Coffey’s talk at LSI Europe '23 in our Video Library on our website.)
• Field Medical: Purpose-built system that incorporates PFA, contact force sensing, electromagnetic localization, and artificial intelligence-enabled mapping capabilities (see CEO Steven Mickelsen’s talk at LSI Europe '24 in our Video Library on our website.)
• Healium Medical: Focused on advancing atrial fibrillation treatment with an innovative, no-contact guided ablation technology that allows simultaneous tissue ablation and lesion monitoring. (See CEO Ran Sela’s talk at LSI Europe '23 in our Video Library on our website.)
• Kardium: Developing a complete solution that combines advanced mapping and single-shot PFA into a single catheter, consists of 122 electrodes in a spherical array that enables single-position pulmonary vein isolation.
The innovative, rapidly expanding field of cryoablation has attracted many start-ups looking to address unmet needs in the space or improve on current technology, across a variety of health conditions. Some offer single-use endoscopic solutions that eliminate the need for costly disinfection and reprocessing, wireless endoscopic solutions, and devices that enhance patient safety. A few of these promising innovations from our LSI alumni, developed for use in cardiac, women’s health, hypertension, gallstone disease, and obesity applications, are highlighted below.
Adagio Medical, founded in 2011 and based in Laguna Hills, CA, is an early commercial-stage, publicly traded company with a vision to improve ablation outcomes in patients with complex cardiac arrhythmias, including paroxysmal and persistent atrial fibrillation, atrial flutter, and ventricular tachycardia. It is pioneering ultra-low temperature cryoablation (ULTC) and pulsed field cryoablation (PFCA) technologies designed to produce durable, contiguous, transmural lesions anywhere in the heart. LSI alumni Todd Usen was recently appointed as CEO and Director of the company; see Power Moves, The Lens, January 2025.
Seed-stage Ananya Health, founded in 2020 and headquartered in Oakland, CA, is building a self-contained, portable, battery-operated cryoablation platform to freeze abnormal cells in the cervix and prevent them from becoming cervical cancer, without any consumable cryogen. The CRCL device's compact design ensures that it can be used in any clinic in order to expand global access to preventative treatment.
Berlin, Germany-based Cryovasc is the first to apply cryoablation technology for catheter-based renal denervation to treat arterial hypertension. With the company’s product, sympathetic nerves can be ablated comprehensively, as the procedure is endpoint-controlled; i.e., renal denervation can be applied until the nerves are fully interrupted.
Ictero Medical, founded in 2018 as part of the Texas Medical Center’s Biodesign Fellowship program, has developed a novel catheter-based cryoablation system to treat high-risk patients with gallstone disease, the CholeSafe System. Ictero’s technology leverages existing interventional techniques to deliver cryoablation therapy to effectively defunctionalize the gallbladder without the need to remove it. The CholeSafe device allows physicians to offer their patients the benefits of surgical treatment without the risk associated with general anesthesia.
Munich, Germany-based Sedivention, founded in 2021, aims to transform the way obesity is treated. It has developed a gastric balloon equipped with cryoablative technology to ablate the gastric nerves inside of the stomach, in a one-time outpatient procedure similar to gastroscopy. The technology is being evaluated as a means of turning off the “hunger nerves” to help patients realize improvements in weight management and diabetes control.
Data in this article is partially sourced from the LSI Market Intelligence Platform, including the Global Surgical Procedure Volumes (SPV) database, the Cryoablation Devices Market Snapshot 2024-2028 (published August 2024), and LSI’s The Numbers LinkedIn e-newsletter “State of the Cardiac Ablation Market” article (published February 15, 2025).
Looking for more global Cryoablation and Cardiovascular medtech market intelligence and insight?
Access LSI’s continuously updated Market Intelligence Platform.
Scan QR code for details and to request a demo:

Notes: Figures may not sum due to rounding. The “Others” category includes AtriCure, Brymill Cryogenics, Cryoprobe, CryoSurgery, Cooper Surgical, Endocare, Grand Cryo, KryoLife, and Mectronic Medical, among others.
Sources: Annual company reports, SEC filings, investor relations materials, and LSI’s Cryoablation Devices Market Snapshot, published August 2024
March 16th - 20th, 2026
Waldorf Astoria, Monarch Beach, Dana Point, CA


This month, our featured Global Procedure Volumes data spotlights some of the segments covered in our two Market Dives, plus much more!
Rhythm Procedures (000s)
LSI’s Global Surgical Procedure Volumes (SPV) Tracker provides trusted coverage of 300+ diagnostic and therapeutic procedures across 37 countries. Scan QR code to request a demo:


Soltanianzadeh’s medtech journey has taken him from start-up entrepreneur, to investor, to CEO of CardiaCare: an innovative AI-based neuromodulation therapy company working to revolutionize atrial fibrillation treatment. In the midst of a Series A raise, he will be presenting at the March LSI USA ‘25 event in Dana Point, CA.
For Amir Soltanianzadeh, once you’ve been bitten by the medtech entrepreneurial bug, building a business around transforming the lives of as many patients as possible becomes part of your identity. And when the rare opportunity presents itself to disrupt one of the biggest markets in medtech, you must take it.
Soltanianzadeh began his journey in 2016 while earning his MSE degree at the Johns Hopkins Center for Bioengineering Innovation and Design (CBID) program. Through this unique Biodesign-esque program, he started his first company, Spine Align. Spine Align developed a platform for measuring the intraoperative 3D biomechanics of the spine during complex spine surgeries via advanced sensors and computer-vision algorithms that complement navigation and robotic systems. Amir led the company from its founding to an M&A exit in late 2021 to one of the leading musculoskeletal companies, Globus Medical
After becoming active in the investing world post-exit, Soltanianzadeh launched his boutique VC firm, Solardis Health Ventures, to invest exclusively in medical device innovations. One of Solardis’ core investments in 2023 was CardiaCare, a breakthrough neuromodulation therapy for atrial fibrillation (AF). The technology is a wearable, non-invasive device that combines proprietary neurostimulation of the median nerve with AI-enabled cardiac monitoring. Soltanianzadeh was searching for a disruptive innovation in AF, one of the fastest growing medtech markets in need of innovation— and CardiaCare was just that.
This January, Soltanianzadeh stepped back into the operator role and joined the CardiaCare leadership team as its new CEO.
Originating from Israel, CardiaCare was founded by Amos Ziv (CSO) in 2015 to provide an alternative therapy modality for AF patients that are limited by the current standard of care (i.e., antiarrhythmic drugs, cardioversion, and/or an ablation procedure). The solution was cleverly designed to modulate the body’s own autonomic nervous system to reduce AF burden, mitigate arrhythmia triggers, and improve patient quality of life and symptoms. Ziv was then joined by co-founder Tsahi Holand (CTO) who helped develop the first-of-its-kind wearable neurostimulation device.
CardiaCare has recently completed a RCT pilot feasibility trial and is now raising a Series A financing to fund pivotal trials for both indications (paroxysmal AF and post-ablation), with targets to achieve FDA clearances in 2027.
Recently, LSI met with Soltanianzadeh to discuss his roles as both investor and medtech CEO, what it takes to translate a novel technology into a disruptive product, his team’s achievements and upcoming milestones, advice to other entrepreneurs, and his perspective as a long-time LSI event alum.
Q. How did you first get involved with CardiaCare?
“I first heard of CardiaCare through the Chairman of the Board, Ken Nelson, who is a prolific executive with an impressive track record of commercializing cardiac monitoring technologies. Ken has commercialized three of the four largest cardiac monitoring companies in iRhythm, Biotelemetry, and Bardy, and thus his involvement caught my attention.
He shared that he joined CardiaCare’s board because he had already helped AF become more widely diagnosed and monitored with his past commercial successes, and that he was very excited to finally help a breakthrough therapy get to market to help those diagnosed AF patients.
After meeting the leadership team and learning more about the technology, I was blown away by what the team had developed, which is completely out of left field. Instead of another invasive procedure or implant, CardiaCare created a wearable, non-invasive, patient-centric device. It’s a wearable form factor, but is very much not another monitoring or wellness tracker device.
I said to myself, there’s no way this wrist-worn, non-invasive therapy could actually work. But then I dove into the clinical data and literature and saw how much impressive work this small team had done on the technology and clinical science. I talked to some of the leading cardiologists in the world, and with their support, I quickly fell in love with the possibilities of this technology. It can potentially help avoid costly and unwanted drugs and additional interventions, and make existing therapies more effective and durable. It’s great for patients, it’s great for providers, it’s great for payors, and can really be a scalable breakthrough.
Solardis led the Seed round in July 2023, and then I became heavily involved as a board observer and advisor to the company. At that time, I realized how much I was enjoying the investor journey—gaining a broad view of the market and witnessing groundbreaking innovations firsthand. However, as an operator at heart with a deep entrepreneurial drive, it was impossible to ignore the rare and transformative opportunity that the CardiaCare team had built.
And so, I discussed with the team about how I could be more involved and add more value, and we mutually decided that I could help advance the team’s mission as CEO. This is one of those rare opportunities in one’s career to disrupt one of the biggest markets in medtech with a transformative technology, and so it was a no-brainer for me to take the role. I’m all in.”
“While researching alternative therapies for cardiac arrhythmias, founder Amos Ziv, a clinical research scientist and entrepreneur, uncovered compelling preclinical data highlighting the median nerve as a promising therapeutic target. Recognizing the opportunity to leverage the body’s own nervous system to influence cardiovascular function, he saw a novel path forward: using neuromodulation to indirectly stimulate the vagus nerve, a key regulator of heart rate and blood pressure, via transcutaneous stimulation of the median nerve.
The app that accompanies our technology transforms AF management by providing therapy management, real-time ECG tracking and arrhythmia alerts, continuous vital sign monitoring, and personalized insights into symptoms and triggers. Given AF’s multifactorial nature—often influenced by medical conditions, medications, alcohol, caffeine, sleep apnea, stress, and genetic predisposition—this comprehensive platform for understanding and managing each AF patient with a
personalized approach is critical. In addition, a cloud-based dashboard enables clinicians to remotely monitor patient disease progression, treatment effectiveness, device compliance, trigger patterns, and overall cardiovascular health.
“It’s great for patients, it’s great for providers, it’s great for payors, and can really be a scalable breakthrough.”
What sets this technology apart is its seamless integration of closed-loop monitoring and machine learning-driven algorithms with proprietary stimulation parameters, refined through years of development and clinical know-how. And in terms of user experience, it’s as simple as wearing an Apple Watch. Our sophisticated digital health platform empowers patients to manage their condition at home, non-invasively, instead of enduring treatments at the hospital. We hope to shift the paradigm away from reactive hospital-based treatment to proactive, patient-centric care. With this technology, CardiaCare is not just improving AF management, it’s redefining how we think about digital therapeutics and neuromodulation in cardiology.
While pulsed field ablation in cardiac electrophysiology has seen recent dramatic growth, this is still a small fraction of the AF market and is not a silver bullet solution. Not only does our technology offer a non-invasive alternative that physicians can offer patients outside of invasive ablation procedures, but interestingly, it is being studied for its use as a post-ablation adjuvant therapy to improve patient outcomes and the longterm durability of these interventions.
This technology really opens the aperture for how many AF patients we can treat and better manage in our healthcare system, from the earlier stages to the later stages, and whether they are pre- or post-ablation.”
Q. What stage has CardiaCare reached in its clinical development, and what key milestones have been achieved so far?
“CardiaCare’s technology is designed to address two primary indications of AF: paroxysmal AF, an early stage of the disease, and post-ablation therapy, to enhance the procedural durability of cardiac ablation and reduce the need for repeat interventions. Currently, the redo rate for ablation is significant— approximately 20-30% within one year—which presents a major clinical and economic challenge in need of more solutions.
For the paroxysmal AF indication, we successfully completed our first-in-human study in 2022. This single-arm trial evaluated ten patients who underwent eight weeks of home-based neuromodulation therapy using the CardiaCare device. The results demonstrated a strong safety profile, a significant acute reduction in AF precursor arrhythmia events—such as premature atrial complexes (PACs)—and a long-term decrease in AF recurrence. Notably, patient compliance with both the monitoring and therapeutic aspects of our device was exceptionally high.
Building on this, we recently completed a double-blinded, sham-controlled, randomized feasibility study in 31 patients. The results were compelling, showing a significant reduction in AF burden that is comparable to leading drugs on the market, and with no significant side effects. In addition, we observed a tremendous improvement in severe AF symptoms and quality of life questionnaires, as assessed by blinded physicians and patients, respectively. This is very promising, as regulatory bodies, payors, and providers are increasingly focused not just on AF burden reduction but also on tangible improvements in patient-reported quality of life.
With strong clinical results, we are now advancing directly into the pivotal trial stage. Planning is well underway for our pivotal study in paroxysmal AF, with patient enrollment set to begin by the end of this year.
For our second indication—post-ablation therapy—we recently launched a pilot study under the leadership of Dr. Vivek Reddy, a globally recognized cardiac electrophysiologist and a long-time advocate of our technology. This study is evaluating the effects of immediate post-procedure neuromodulation, where patients receive the CardiaCare device in the recovery room and begin therapy right away. The pivotal study for this indication is expected to commence in 2026 after the paroxysmal AF pivotal study is enrolled.”
Q. Can you describe challenges you have faced along the way, and how did you overcome them?
“One of our biggest challenges has been overcoming skepticism around non-invasive therapies. Historically, many medical wearables have underdelivered, leading to hesitancy from clinicians and investors alike. There’s a prevailing belief that meaningful therapeutic impact requires an interventional approach. But we believe healthcare is at an inflection point— one where external, patient-friendly solutions can and should play a much larger role.
The reimbursement landscape is evolving to support non-invasive, at-home therapies, and we see an opportunity to reshape the narrative around external neuromodulation. CardiaCare is at the forefront of that shift, proving that an external device can drive real clinical outcomes. Our approach isn’t just about treating AF—it’s about redefining how medtech integrates with the digital health ecosystem to deliver more accessible, scalable, and cost-effective care.
If we ask ourselves, where is healthcare headed in the next 10, 20, or 50 years, it’s clear that it won’t be dominated solely by interventional procedures. The future lies at the intersection of the digital and physical worlds, where technologies like ours enable proactive, continuous disease management. That’s the vision we’re committed to realizing.”
Q. What advice do you have for the medtech entrepreneurs out there, and lessons learned along the way?
“One of the most important lessons I’ve learned is that you will never have all the answers. As an entrepreneur, you have to
strike a balance between unwavering belief in your vision and a deep understanding of what stakeholders—investors, strategics, providers, and patients—truly need.
A common mistake, and one I’ve made myself, is assuming that because something makes sense to you, it will naturally make sense to others. But investors think in terms of market impact, risk-reward balance, and short- and long-term value creation. Physicians have a clear threshold for adopting new technologies. Patients need usability and demonstrated benefits. Payors must know why the cost of a device is a worthwhile investment to the healthcare ecosystem, and not another fancy technology. Understanding these perspectives early on is critical.
Another key takeaway is that medtech must learn from the consumer mindset. While our industry is highly regulated, we should embrace the same principles that drive consumer markets—innovation, cost-efficiency, and seamless user experience. Healthcare is moving in that direction, whether we
acknowledge it or not, and the companies that recognize this shift will be the ones that thrive.”
Q. How does your experience as an entrepreneur, investor, and as a strategic come into play in your new role as CEO of CardiaCare?
“Going into this role, I assumed that being a second-time CEO would give me greater confidence and insight. What I didn’t fully appreciate was how my experience as a full-time investor in between these leadership roles would double my perspective.
Now, I approach challenges with a much broader view— anticipating what investors will prioritize, understanding what strategics seek in partnerships and acquisitions, and critically evaluating how our solution fits into the clinical workflow.
• Neuromodulation involves the stimulation of various nerves in the central nervous system, peripheral nervous system, autonomic nervous system, or deep cell nuclei of the brain that lead to the “modulation” of its activity.
• Total global sales of neuromodulation devices are projected to increase at a compound annual growth rate (CAGR) of 7.7% during the forecast period 2023-2028. Expansion into new indications and more sophisticated technology, which translates to increased product prices, are major factors driving market growth.
• Technology trends include the introduction of closed-loop/responsive systems, device rechargeability, and the reduction in the invasiveness of systems.
• Closed-loop systems have offered significant advantages to patients, including reduced power consumption, a reduction in erroneous stimulation, and more effective, “on-demand” delivery of therapy during physiological states necessitating stimulation.
• There is a significant amount of innovation being driven by early-stage, precommercial companies.
• The market is highly compelling given the number of indications and patients that fail to respond to pharmacological management of their condition that are in search of effective therapies.
Source: LSI’s Neuromodulation Devices Market Snapshot 2024-2028, published June 2024

Balancing all of these factors while staying agile as a startup is a complex challenge, but one that’s essential.
Had I transitioned directly from my first CEO role to this one, I wouldn’t have had the same depth of understanding. My time as an investor has been invaluable in shaping how I now lead and position CardiaCare for long-term success.”
Q. Can you talk a bit about the LSI events, and your experience both as an alum and a speaker at the upcoming LSI USA ‘25 conference. Have the events and the connections you’ve made impacted your company’s trajectory?
“LSI has been an incredible platform for the medtech ecosystem. I’ve attended several events, both in the U.S. and Europe, and what the LSI team has built is invaluable. It’s a true nexus—bringing together early-stage startups, growth-stage companies, strategic investors, and family offices, all in one setting.
The relationships I’ve built through LSI have been transformative. I now work daily with people I met through these events—connections that otherwise might have taken years to establish. What sets LSI apart is that it’s not just a networking forum; it’s where real business happens. I know firsthand of deals that originated at LSI, and as the event expands globally, its impact will only grow.
I’m incredibly proud to have been involved since the early years. Attending as an investor was a great experience, but now, returning as an entrepreneur, it’s even more meaningful.
This is where medtech will continue to evolve, and for anyone serious about this industry, LSI is a must-attend event.”
“We are preparing to launch our pivotal studies by the end of this year. This is a major step forward in bringing our technology to market.
In parallel, we’ve kicked off our Series A fundraising, which will fully fund our go-to-market strategy for both indications, covering pivotal trials, FDA clearance, and the establishment of an initial reimbursement pathway.
The response so far has been strong, with significant interest from venture capital firms and strategic investors. We are on track to bring CardiaCare to market for our first two clinical indications by mid-to-late 2027, delivering a much-needed breakthrough therapy for AF patients. Our goal is not just to launch a product but to redefine how AF is treated for millions of patients globally.
The next chapter is about scaling, executing, and delivering on this vision. I joined this team specifically to make this vision a reality, and we will pursue that mission relentlessly.”
Note: Images in this article used with permission from Amir Soltanianzadeh. Watch his LSI Europe ‘24 in-studio interview, and Amos Ziv’s presentation at LSI USA ‘24, among other presentations, on the LSI website at Resources > Video Library.
Is your organization interested in having your journey and accomplishments featured in an upcoming issue of The Lens? Contact our Managing Editor and Content Strategist, Tracy Schaaf, at tracy@ls-intel.com.
Celebrating the recent wins of our LSI presenting company and speaker alumni community.


New York-based AccurKardia, an innovator in ECG-based diagnostics technology, has received FDA Breakthrough Device Designation for its ECG-based, AI-powered AK+ Guard hyperkalemia detection software. The investigational technology uses Lead I ECG data to alert patients and clinicians of moderate to severe episodes of hyperkalemia (excess potassium in the blood) that can lead to sudden cardiac arrest.
AK+ Guard is designed to work with a wide range of FDA-cleared consumer and clinical wearables that currently
capture Lead I ECG data (e.g., smartwatches), enabling hyperkalemia monitoring outside of the clinic and earlier intervention for high-risk populations. These include those with end-stage renal disease, chronic kidney disease, and other risk factors.
AK+ Guard was also recently accepted as one of only 62 devices in the FDA Total Product Life Cycle Advisory Program (TAP). The TAP program provides earlier and more frequent engagement with the FDA, helping accelerate the regulatory process, improve the quality and timeliness of device evaluations, and ultimately speed commercialization.
The AK+ Guard news follows AccurKardia’s recently received Breakthrough Device Designation for its Aortic Valve Stenosis (AVS) screening software, AK-AVS. The software is designed to leverage the ubiquity of the electrocardiogram (ECG) to identify potential cases of aortic valve stenosis within millions of ECGs already present in healthcare system electronic health records. AK-AVS software aims to help identify
and prioritize which patients should receive echocardiograms for definitive diagnosis.

In another $100M+ deal announced in early January, Aspect Biosystems, a biotech company pioneering the development of bioprinted tissue therapeutics as a new category in regenerative medicine, closed a $115 million Series B financing round.
The financing was led by Dimension, with participation from existing and new investors including Novo Nordisk, Radical Ventures, an undisclosed
leading global investment firm, InBC, Pallasite Ventures, Pangaea Ventures, Rhino Ventures, and T1D Fund: A Breakthrough T1D Venture
Proceeds from the Series B enable Aspect to advance multiple bioprinted tissue therapeutics towards the clinic, with a mission to deliver a new class of cellular medicines and functional cures for people living with serious metabolic and endocrine diseases. Additionally, the funds will fuel the expansion of Aspect’s industry-leading, full-stack tissue therapeutic platform, which integrates proprietary AI-powered bioprinting technology, computational design tools, therapeutic cells, and advanced biomaterials.
This financing builds on Aspect’s recent momentum, including a CA$200 million partnership with the governments of Canada and British Columbia, and a partnership with Novo Nordisk focused on diabetes and obesity. (Also see “Look What You Made Me Do: Novel Tech Deals Dominate Device Venture Investment in ‘24, Set Stage for Brisk ‘25,” The Lens, January 2025; and LSI Alumni Milestones, The Lens, September 2024.)

Capricorn Partners, a Leuven, Belgium-based independent European manager of venture capital and equity funds, has launched the Capricorn Healthtech Fund II, with a first closing of €51 million ($53.4 million).
This fund marks a significant milestone in the firm’s ongoing commitment to advancing personalized healthcare and promoting value-based health initiatives across Europe. The fund will target investment in 10-15 late-seed to Series B stage companies, in digital health or medtech, with a proven technology base and a health economic benefit.


Caresyntax, a global leader in realworld evidence (RWE) and surgical intelligence, and Qaelon Medical, a medtech pioneer in surgical leak detection technologies, have announced a partnership to build the first RWE platform to address the devastating impact of surgical leaks.
The initial phase of the partnership will focus on building the first-ever standardized and transparent patient database to establish baseline data on the incidence and outcomes of surgical leaks using Qaelon Medical’s proprietary intra-operative data. The second phase will capture RWE to demonstrate the life-saving benefits of Qaelon’s realtime surgical leak detection on patient outcomes and healthcare costs.
In the third phase, Caresyntax and Qaelon will incorporate predictive clinical decision-making support for surgical teams to reduce the incidence and impact of surgical leaks. Across all phases, Qaelon and Caresyntax will be actively engaged with leading surgeons and health systems. In addition, the organizations are seeking industry partners in the medtech ecosystem to collaborate and share data that will accelerate learning and insights around surgical leaks while fostering greater transparency in surgical care.
Headquartered in San Francisco and Berlin, Caresyntax software is used in more than 4,200 operating rooms worldwide and supports surgical teams in more than three million procedures per year.
Qaelon Medical is pioneering the first and only platform that combines real-time detection of life-threatening surgical leaks and state of the art surgical insufflation. The company’s breakthrough solution empowers surgical teams with definitive leak test results during surgery so they can take immediate corrective action before the patient leaves the operating room, thereby improving patient outcomes while reducing $1.1 billion spent annually in the U.S. to treat surgical leaks. Qaelon builds upon existing insufflation technology used in almost every minimally invasive surgery enhanced with proprietary controls, digital sensors, software, and data algorithms.
In January, Qaelon also announced that it had been accepted into the @NVIDIA Inception Program, a prestigious initiative designed to support startup companies pioneering advancements in AI and data science. As the first real-time, objective performance indicator for surgical leaks, access to NVIDIA’s cutting-edge technologies and global resources will help accelerate Qaelon Medical’s life-saving mission to reduce surgical leaks using proprietary data and advanced surgical intelligence. The company also recently secured a new patent in China for its real-time surgical leak detection platform, adding to its existing patents in the European Union, the United States, and Japan.

Roivios, a clinical-stage medical device company, and Caresyntax, a global leader in RWE and surgical intelligence, have joined forces to revolutionize renal
care. This partnership aims to accelerate the integration of clinical and economic insights during product launch, driving the development of innovative medical technologies to improve patient outcomes and reduce healthcare costs.
Central to this collaboration is the pivotal trial of Roivios’ JuxtaFlow Renal Assist Device (RAD), an investigational technology specifically engineered to sustain or enhance kidney function for patients with renal impairment undergoing an on-pump cardiac surgery. This first-of-its-kind device has the potential to transform kidney care for high-risk patients. (Also see LSI Alumni Milestones, The Lens, January 2025.)
Caresyntax’s clinical data as a service (CDaaS) platform will capture RWE to complement the pivotal trial and help Roivios measure and optimize the value of the JuxtaFlow RAD. The partnership will establish a new model in renal disease management—one that prioritizes actionable clinical insights and financial efficacy to deliver superior patient care.

Digital health innovations company CathWorks announced that it has successfully completed enrollment in the Advancing Cath Lab Results with FFRangio Coronary Physiology Assessment (ALL-RISE) randomized controlled trial. This landmark study is evaluating the clinical and economic benefits of the CathWorks FFRangio System in diagnosing and treating coronary artery disease (CAD), offering potentially groundbreaking evidence that could redefine the standard of care.
The ALL-RISE Study included over 1,924 patients across 59 sites in North America, Asia, Europe, and the Middle East. Participants presenting with coronary stenoses of intermediate significance and requiring physiology
assessment were randomized to receive either FFRangio-guided treatment or invasive pressure wire-guided treatment. Notably, this was the first-ever randomized controlled trial in the U.S. to assess clinical outcomes using an angiography-based tool for physiologic lesion assessment.
The CathWorks FFRangio System combines artificial intelligence (AI) and advanced computational science, transforming how cardiovascular disease—the leading cause of death in the U.S.—is diagnosed and treated. The FFRangio System obtains physiologic information from routine angiograms, eliminating the need for drug stimulation and invasive pressure wires. It provides physicians with quick and reliable intraprocedural FFRangio values for the entire coronary tree.

Wearable insulin delivery device company CeQur closed a $120 million financing round, investors undisclosed, to support its four-day wearable insulin patch technology, CeQur Simplicity. The funds will support the continued growth of the company’s sales force and expansion of its clinical team, to ensure that more people living with diabetes can benefit from convenient, discreet, and injection-free dosing.
One CeQur Simplicity patch holds up to 200 units of rapid-acting insulin administered in two-unit increments and replaces, on average, twelve daily mealtime injections over four days. Clinical research has shown that nearly 90% of patients using CeQur Simplicity reported following their insulin regimen better as compared to multiple daily injections. The Patch is clinically proven to improve glycemic control, with patients achieving significantly improved A1C and time-in-range goals, according to the company.
CeQur has seen increasing adoption of its device, with over 6,000 patients currently using it. (For a wrap-up on the 2024 device venture investment picture and the biggest financings so far in 2025, with CeQur included on the list, see “Look What You Made Me Do: Novel Tech Deals Dominate Device Venture Investment in ‘24, Set Stage for Brisk ‘25,” The Lens, January 2025.)

Geneva, Switzerland-based medtech startup EBAMed, specializing in a guidance system for non-invasive heart arrhythmia treatment using proton therapy, has successfully closed a Series A financing round, raising EUR 1.6 million. This new investment, contributed by CDP Venture Capital and the European Union, supports the development of EBAMed’s CardioKit system for non-invasive treatment of heart arrhythmias using proton therapy.
EBAMed had initially secured EUR 14.35 million in June 2023, led by Panakès Partners, with support from the EIC Fund, Swiss EFI Lake Geneva Ventures, LIFTT Venture Capital Italia, OCCIDENT, and existing backers like Mayo Clinic. With the Series A funding now complete, EBAMed is focused on advancing the CardioKit system to the final stages of clinical trials, marking a crucial step toward commercial deployment.
Heart arrhythmias impact 50 million people globally, with numbers projected to rise as populations age. EBAMed’s CardioKit enables non-invasive treatment of ventricular tachycardias (VTs) using proton therapy. The system enhances precision by automatically adapting radiation delivery to cardiac motion, offering an alternative to drugs and catheter-based ablation while minimizing radiation exposure to healthy heart tissue.

Field Medical has raised $26.87 million in funding, following FDA Total Product Life Cycle Advisory Program (TAP) Pilot acceptance and Breakthrough Device Designation for its FieldForce Ablation System. FieldForce is the first and only pulsed-field ablation (PFA) system designed specifically for ventricular arrhythmia ablation. The company’s proprietary FieldBending technology and comprehensive product portfolio, including the FieldForce Catheter, FieldForce Ablation System, and FieldFlex Sheath, are designed to expand PFA applications beyond atrial fibrillation to treat complex ventricular arrhythmias.
The Cardiff-By-the-Sea, CA-based company is led by Steven Mickelsen, MD, a pioneer in pulsed electric field technology. Dr. Mickelsen’s foundational work in PFA established the basis for modern advancements in the technology, which Field Medical continues to refine with its groundbreaking solutions. Mickelsen was previously the founder of PFA technology company Farapulse, which he led to an acquisition worth at least $460 million by Boston Scientific in 2021.

Francis Medical, a privately held developer of an innovative and proprietary water vapor ablation therapy for the treatment of prostate, kidney,
and bladder cancer, announced the completion of an oversubscribed $80 million Series C equity financing, the largest fundraising round to date for the company.
Arboretum Ventures and Solas BioVentures co-led the Series C round. New investors Orlando Health Ventures and two additional strategics joined this round, along with previous investors Coloplast A/S and Tonkawa The Series C proceeds will continue to fund the VAPOR 2 pivotal clinical study for the management of prostate cancer and the development and U.S. commercialization of its Vanquish Water Vapor Ablation proprietary prostate cancer treatment, expected by the end of 2025. (Also see “Look What You Made Me Do: Novel Tech Deals Dominate Device Venture Investment in ‘24, Set Stage for Brisk ‘25,” The Lens, January 2025.)
The Vanquish device’s thermal water vapor energy technology is a breakthrough therapy designed to use phase shift energy stored in sterile water vapor to convectively transfer thermal energy to cancerous tissue, causing cell death. Through this process, damage to surrounding structures is minimized or eliminated by respecting the prostate’s natural boundaries. Vanquish aims to be a revolutionary cancer therapy that is tough on cancer yet gentle on patients.

GT Medical Technologies, a medical device company focused on improving the lives of patients with brain tumors, today announced the company has completed a $37 million first close of a Series D financing round. The financing was led by Evidity Health Capital, alongside new investor Accelmed Partners. Also participating were existing investors MVM Partners, Gilde Healthcare and Medtech Venture Partners
The funds will accelerate the completion of the ROADS clinical study that is focused on GammaTile for newly diagnosed brain metastases, and the GESTALT clinical trial for patients with newly diagnosed glioblastomas. In addition, the funds will support the continued commercialization of GammaTile, the company’s FDA-cleared bioresorbable radiotherapy implant for the treatment of brain tumors. By delivering tile-based radiation therapy directly into the surgical cavity at the time of tumor removal, GammaTile provides immediate, localized treatment. This approach targets remaining cancer cells when they are at their lowest levels to help prevent regrowth while minimizing radiation exposure to healthy brain tissue. (Also see Look What You Made Me Do: Novel Tech Deals Dominate Device Venture Investment in ‘24, Set Stage for Brisk ‘25, The Lens, January 2025.)


Surgical training tech companies Inovus Medical and UpSurgeOn have announced that they are joining forces in a global partnership aimed at expanding access to high-quality surgical training for thousands of surgeons worldwide.
A key part of the partnership is the integration of UpSurgeOn’s mixed reality surgical simulation technology into Inovus Medical’s Totum digital surgery
platform. This combination will create the first-ever “digital synthetic cadaver” for cranial, spine, ENT, and plastic surgery. By merging UpSurgeOn’s lifelike anatomical and tissue simulations with Totum’s advanced digital tools, the partnership sets a new standard for multidisciplinary surgical training.
Beyond technology integration, Inovus Medical will also take on a commercial role for UpSurgeOn in several international markets, helping to expand its reach and support its growing user base. In addition, UpSurgeOn will offer access to its cranial, spine, ENT, and plastic surgery training modules in regions where Inovus doesn’t have a direct presence—bringing this cutting-edge technology to surgeons in more than 100 countries.

St. Paul, MN-based iotaMotion, a leader in advanced robotic-assisted systems for cochlear implant surgery, announced a major milestone in its mission to advance cochlear implant surgery beyond human capability. Following the first full year of commercialization, there are 25 centers across the U.S. now offering the iotaSOFT Insertion System. The company also announced that it secured Series B funding, with details undisclosed, that will be used to scale commercial operations while driving further innovations in the field of cochlear implantation.
As the first and only robotic-assisted system available in the U.S. for cochlear implantation, the iotaSOFT Insertion System offers a groundbreaking approach to cochlear implant surgery by providing enhanced control beyond the capability of a human hand during electrode array insertion, resulting in less trauma to the cochlea when compared to manual insertions. iotaMotion aims to provide hearing professionals
with tools to improve patient outcomes and preserve intracochlear structures. The company was founded in 2015 by otolaryngologists from the University of Iowa Hospitals and Clinics.

Lumicell, a leader in developing innovative fluorescence-guided imaging technologies for cancer detection, recently announced the U.S. commercial availability and first commercial use of LumiSystem. The system combines two recently FDA-approved products: LUMISIGHT, an optical imaging agent that fluoresces at sites of suspected cancerous tissue, and Lumicell Direct Visualization System (DVS), an imaging device that provides real-time fluorescence guidance for scanning the breast cavity during lumpectomy surgery. (Also see LSI Alumni Milestones, The Lens, December 2024.)
Unlike other detection systems that only evaluate tissue after it has been surgically removed, LumiSystem allows for real-time visualization of cancer inside the breast. This enables surgeons to identify and immediately resect any suspicious tissue during the surgery, improving the likelihood of achieving a more complete resection and reducing the need for follow-up procedures.
With 84% diagnostic accuracy, LumiSystem improved surgical outcomes in 10% of patients treated in the INSITE pivotal trial. By identifying cancerous tissue completely missed by surgeons and pathologists, LumiSystem offers a more comprehensive approach to breast-conserving surgery and ultimately enhances patient care.
Finnish company Maculaser has announced that it is now part of PhotonMed, a €32 million European Union Chips Joint Undertaking (Chips

JU)-funded initiative spanning nine countries. The ambitious collaboration brings together leading research institutes and industrial partners to boost the development and adoption of advanced nano-electronic chip technologies and systems manufactured in Europe.
As part of this initiative, Maculaser is integrating innovative photonic technologies into its treatment platform to enhance outcomes for patients with macular degeneration and other retinal conditions.
The company’s technology employs non-damaging retinal heating, in order to harness the regenerative effects associated with temperature elevation, promoting healing without causing harm to delicate retinal tissues. Its solution employs retinal temperature monitoring designed to ensure safe, effective, and consistent treatments for every patient, taking into account inter-patient variability and differences across retinal locations.

Cleveland, OH-based Neuronoff, a pioneer in minimally invasive neuromodulation solutions, announced two major funding milestones totaling $5 million to advance its innovative Injectrode technology. The Department of Defense (DoD) has recommended $1.73 million to fund a clinical randomized controlled trial for the treatment of
neurogenic bladder following spinal cord injury, while the National Institutes of Health (NIH) has awarded $3.2 million to support preclinical research in overactive bladder (OAB) treatment. The company’s flagship Injectrode product offers a drug-free and non-opioid alternative for conditions ranging from chronic pain to autonomic, bladder, cardiac, and inflammatory disorders.
The DoD funding will support a clinical randomized controlled trial in collaboration with the University of Texas Health Science Center in Houston, TX. This three-year clinical evaluation will assess the safety and efficacy of the Injectrode when implanted along the tibial nerve in individuals with spinal cord injury.
The preclinical research focused on OAB is a collaborative work between the University of Wisconsin, Case Western Reserve University, and Massachusetts General Hospital. The investigation aims to improve treatment for people suffering from OAB using the Injectrode’s two-component, passive injectable electrode system for direct nerve interfacing and improved target engagement. The study teams anticipate that this method will dramatically improve the reliability of the nerve interface, resulting in better long-term stability while reducing complications.

NEVENT is focused on making retained guidewires one less risk patients take when they receive medical care. NEVENT’s newly patented Safety Sheath eliminates the patient harm and medical system costs of retained guidewires after vascular catheter placement.
The company recently announced that it has secured a new patent in India for its Safety Sheath technology, adding to its U.S. patent issued
in November 2023. As product development progresses through Phases 1 and 2, the company plans to submit for FDA approval later in 2025, to advance its solution for preventing retained guidewires.
Each year, the medical error of retained guidewires impacts over 1,500 patients, with a serious morbidity and mortality rate of 10.5%. Recognized by the Joint Commission as a Never Event in 2018, retained guidewires not only endanger lives but also inflict an additional $830 million in costs on physicians and healthcare systems annually. Current prevention strategies typically only identify such errors after they have occurred. In contrast, NEVENT has engineered the first effective device designed specifically to prevent the occurrence of guidewire retention.

Mont Saint-Guibert, Belgium-based Novadip Biosciences, a clinical-stage biotech company specializing in regenerative medicine, has announced the completion of the 12-month post-surgical visit of the last patient implanted with its investigational tissue regeneration product, NVD003, in the phase 1b/2a clinical trial (NCT05693558) which treated four patients with congenital pseudarthrosis of the tibia (CPT), a rare pediatric bone condition. Novadip is developing NVD003, an autologous therapy derived from adipose stem cells, as a potential single treatment to save limbs and restore mobility in patients with CPT.
Combining the 12-month results from this trial together with those from four other children previously treated with NVD003 in two compassionate use programs in Belgium, 88% of patients, most of whom had prior failed surgeries, achieved healing of their fractures with NVD003.
In February, the company also released the promising 12-month results of its first-in-human, proof-of-concept study of NVDX3 in distal radius fractures in adults, following the interim results unveiled in October 2024 NVDX3-CLN01 (NCT05987033) was a single-arm, open-label clinical trial aimed at evaluating the safety and preliminary efficacy of NVDX3, an allogeneic bone grafting material created from human osteogenic adipose tissue-derived mesenchymal stem cells. This trial focused on adults with distal radius fractures involving the wrist joint with multiple bone fragments.
The participants, aged 28-84 years, were treated with NVDX3 with follow-ups conducted at 3, 6 and 12 months. At the 12-month follow-up visit, 100% of patients had bone union of their fractures, regardless of age and complexity of fractures treated, with the majority of patients achieving this by the six-month visit. No adverse events were deemed by the investigator to be related to NVDX3.

Onera Health, a leader in transforming sleep medicine and remote monitoring, recently announced the publication of one of the largest sleep diagnostic trials of the decade, a multicenter validation study of its Onera Sleep Test System (STS) in the Journal of Clinical Sleep Medicine. The study demonstrated that the patch-based Onera STS home-polysomnography (hPSG) performs comparably to gold-standard in-lab polysomnography (PSG) for both sleep staging and respiratory event detection.
The international multicenter study involved the analysis of 206 participants across seven clinical sites in Germany, and a core laboratory at Johns Hopkins University in Baltimore, MD.
The study found high concordance between the Onera STS and traditional PSG across essential sleep variables and key respiratory parameters, as well as substantial epoch-by-epoch agreement in sleep staging and comparable inter-scorer reliability between the two systems, which is essential for physicians to make accurate diagnoses and treatment decisions in sleep medicine.

Onkos Surgical, a provider of innovative solutions for complex orthopedic procedures, announced that its revolutionary NanoCept Antibacterial Technology has been used successfully in multiple first-in-human surgical procedures. The company also secured a $35 million venture loan facility from Horizon Technology Finance to expedite manufacturing and commercialization.
Orthopedic implant-related infections are clinically challenging and have significant negative effects on patients. Periprosthetic joint infection (PJI) has a lower survival rate than several forms of cancer, and is expected to cost the U.S. healthcare system up to $1.8 billion by 2030, according to the company. It is estimated that 60-70% of all early and delayed-onset PJI (less than 3 months and 12-24 months post-surgery) are initiated at the time of the procedure through intraoperative contamination.
Onkos received De Novo market authorization from the FDA in April 2024 for its NanoCept Antibacterial Technology. Data submitted to support the De Novo application showed that NanoCept has up to a 99.999% kill rate in standardized in vitro testing of bacteria that are commonly found in the operating room environment. The initial FDA authorization for NanoCept was for the company’s ELEOS Limb Salvage System, which is a modular total joint system used for patients with bone loss.

Pristine Surgical recently announced that its Summit single-use arthroscopy platform has secured a Defense Logistics Agency (DLA) ECAT contract, thanks to its partnership with Veterans Healthcare Supply Solutions. This milestone grants access to its innovative products and strengthens the company’s mission to simplify endoscopy for caregivers serving veterans and military members, both in the U.S. and abroad.
Summit is a fully integrated, singleuse scope with 4K resolution that simplifies arthroscopic procedures and packs the latest in minimally invasive visualization technology into a sterile, cost-effective, ready-to-use package. The company’s Summit platform combines single-use endoscopes with cloudbased software to improve minimally invasive visualization. Summit integrates with a surgery center’s existing endoscopy tower, allowing ASCs to upgrade to 4K without major capital investment. (Also see Market Dive: GI Endoscopy Devices, The Lens, January 2025.)

Healthtech company Quibim, which is pioneering the use of imaging biomarkers for precision medicine, has announced the closing of a $50 million Series A financing. With this financing, Quibim is establishing a strong U.S. presence, and building on its success in Europe through strategic
collaborations with hospitals and pharmaceutical companies.
The financing was led by Asabys (through its fund Sabadell Asabys II) and Buenavista Equity Partners (through the BHG I fund, created in partnership with Columbus Venture Partners), and joined by UI Investissements, GoHub Ventures and CDTI Innovación, through its program Innvierte, as new investors. It was also joined by current investors, Amadeus Capital Partners, APEX Ventures, Partech, Adara Ventures, Leadwind (KFund’s early growth fund), Tony Fadell’s Build Collective, and individual investor Jonathan Milner, PhD, founder of Abcam plc.
Quibim is dedicated to the ambitious mission of unlocking a complete, non-invasive understanding of every tissue point in the human body, at any moment in time. To achieve this, the company develops foundational AI models applied to imaging that extract actionable insights from MRI, CT, and PET scans. These insights enable precise characterization of phenotypes and prediction of outcomes in areas such as oncology, immunology, neurology, and metabolic disorders. According to the company, this is the pathway toward achieving human digital twins: dynamic models that not only reflect and allow health monitoring but also enhance patient stratification, improve the success rates of drug development programs, and enable treatment testing before application. Today, the company is building digital twins at the organ and lesion level, exemplified by solutions like QP-Brain, QP-Prostate, and QP-Liver

Robeauté, a Paris, France-based medtech startup developing neurosurgical microrobots, announced that it has raised $28 million in funding. The round was led by Plural, Cherry Ventures,
and Kindred Ventures. Other investors included LocalGlobe, Think.Health, APEX Ventures, and Brainlab
Led by CEO Bertrand Duplat and COO Joana Cartocci, Robeauté is developing neurosurgical microrobots that can move along curved routes through the extracellular matrix to safely reach multiple sites of interest and carry out whichever task is required locally. Their innovative microrobots, smaller than a grain of rice, aim to provide surgeons with enhanced access to delicate areas of the brain and other hard-to-reach regions of the body. The devices can also embark on specific missions depending on the pathology, making them the ideal tool to deliver molecules, implant an electrode and collect cell or live-data samples through sensors to transform the understanding of and ability to treat neuropathologies.
The funds will support continued development, preparation for human trials in 2026, and the establishment of U.S. operations ahead of FDA approval and market entry.

Campbell, CA-based Simpson Interventions, a medtech company specializing in cardiovascular interventional devices, recently announced that its Acolyte Image-Guided Crossing and Re-Entry Catheter System has been granted an Investigational Device Exemption by the FDA, after having been designated as a Breakthrough Device earlier this year. The Acolyte system is intended to provide interventional cardiologists with a more reliable and predictable tool to facilitate revascularization of coronary chronic total occlusions. A significant percentage of all patients with coronary artery disease, whether managed medically or referred for bypass surgery, have CTOs.
Additionally, the company also announced that its Shadow Catheter received 510(k) clearance from the FDA. The single-use/disposable Shadow Catheter is intended for use in conjunction with steerable guidewires to access discrete regions of the coronary and peripheral arterial vasculature. This access helps facilitate placement and exchange of guidewires and other interventional devices for use during procedures, permitting simultaneous use of two guidewires and/or to subsequently infuse or deliver diagnostic or therapeutic agents.

Cambridge, MA-based Sinaptica Therapeutics, a clinical-stage company leading the development of a new class of personalized neuromodulation therapeutics to treat Alzheimer’s and other primary neurodegenerative diseases, today announced that co-development of the SinaptiStim Neuromodulation System is underway, with planned March 2025 delivery of the first clinical system for validation testing and use in forthcoming clinical trials.
Partner Nexstim is customizing the system to Sinaptica’s specifications, incorporating high resolution, 64-channel electroencephalography (EEG) from industry leader Bittium to enable precision calibration of the therapy for each Alzheimer’s patient. The new system will be used in Sinaptica’s upcoming clinical trials in Alzheimer’s patients and beyond, with multiple trials scheduled to begin in 2025
Sinaptica’s new non-invasive neuromodulation therapy (nDMN) to treat Alzheimer’s disease uses personalized precision repetitive transcranial magnetic stimulation targeting the Default Mode Network (DMN)—the primary functional brain network most impacted by
Alzheimer’s disease. Sinaptica’s novel approach incorporates the patient’s MRI and concurrent EEG information in order to determine personalized treatment parameters. (Also see Market Dive: Neuromodulation Devices, The Lens, September 2024.)

Spiro Robotics, a medical device innovator based in the California Bay Area, has announced the successful completion of its first-in-human trial for the Spiro-VISTA, a groundbreaking image-guided tracheal intubation device designed to revolutionize difficult airway management.
Out of an estimated 30 million tracheal intubations performed annually in the U.S., 6 million are difficult, representing a large unmet medical need and market opportunity.
In the trial involving 30 patients with highly complex and challenging airways, all participants were successfully intubated on the first attempt without adverse events or complications. Many of these patients had multiple predictors for what would otherwise have been a difficult conventional video laryngoscopy, highlighting the exceptional performance of the Spiro-VISTA device.
Spiro-VISTA uniquely combines the capabilities of video laryngoscopy and flexible endoscopy in a single, handheld, user-friendly device. By enabling operators to navigate diverse airway challenges with precision and confidence, Spiro-VISTA sets a new standard in airway management, potentially transforming the field for both novice and experienced clinicians. (Also see the February 18, 2025 edition of The Memo, a weekly LinkedIn newsletter published by Life Science Intelligence, spotlighting Spiro Robotics’ origin story, technology, progress, and milestones.)

Milan, Italy-based StarTric announced that it has successfully finalized the first phase of pre-clinical testing of its TriClover technology. TriClover is a novel triple edge-to-edge transcatheter repair technology designed to provide a minimally invasive solution for treating tricuspid regurgitation.
StarTric’s mission is to provide a truly accessible, safe and effective treatment for tricuspid regurgitation, a very serious need for millions of patients, which remains largely unmet also after the introduction of a first generation of minimally invasive repair and replacement devices.
The company’s technology has been conceived to replicate, through a minimally invasive transcatheter device, the repair concept, results, and clinical outcomes of the Clover open-heart technique. It consists of a device that can be safely implanted in the beating heart by means of a transfemoral catheter. StarTric is the first transcatheter tricuspid valve repair technology based on a successful surgical technique conceived specifically for the tricuspid valve, unlike other repair technologies derived from mitral valve devices.

THINK Surgical, an innovator in the field of orthopedic surgical robots, recently announced the successful first
use of Medacta International’s GMK Sphere Medially Stabilized Knee utilizing the TMINI Miniature Robotic System by Matthew Beal, MD, an Adult Reconstruction Surgeon at the Ohio State University Wexner Medical Center in Columbus, OH.
The GMK Sphere and GMK SpheriKA knees are an innovative implant addition to THINK Surgical’s ID-HUB proprietary implant Data Bank used by the TMINI Miniature Robotic System. The GMK Sphere and SpheriKA systems are the only medial ball-in-socket design knee systems available for use with the TMINI Robot, and the GMK Sphere knee has been shown to provide high stability throughout the entire range of motion. GMK SpheriKA is the latest addition to Medacta’s knee portfolio and is the first femoral component specifically optimized for the kinematic alignment technique. Both knee systems also offer a metal-sensitive option with SensiTiN coating and cementless applications with MectaGrip coating. THINK Surgical received FDA 510(k) clearance for use of the Medacta GMK Sphere and SpheriKA knee implants with the TMINI Miniature Robotic System in August 2024.
THINK Surgical robots are open platforms providing support for implant brands from multiple manufacturers, enabling the choice of implant to be driven by the surgeon. The company’s TMINI Miniature Robotic System has been utilized in over 500 total knee arthroplasty (TKA) procedures in the U.S. (Also see Market Dive: Orthopedic Surgery, The Lens, December 2024.)

New Zealand-based Wellumio recently announced that it is expanding its global footprint by launching a new subsidiary in California—a strategic move that lays the groundwork for planned U.S.-based clinical trials to validate its
innovative Axana 0.1-tesla portable MRI device and accelerate its mission to bring this portable, life-saving technology to frontline stroke teams nationwide.
The company also announced that it has enrolled the first patient in a feasibility, safety, and usability assessment of its Axana device, in Melbourne, Australia. The study will be a twopart, dual-center, feasibility/observational study, with the primary objective being to assess the safety and feasibility of the Axana device in a hospital setting with healthy controls and stroke patients. Secondary objectives include evaluating the device’s usability, acquiring physiological data from healthy controls and hospital patients, and investigating the reproducibility of MRI scans within and between participants.
Unlike conventional MRIs, which rely on superconducting magnets and large coils to generate magnetic gradients for imaging, Axana’s innovative technology is powered by Pulsed Gradient Free Mapping (PGFM). The company says that this groundbreaking approach eliminates the need for bulky components, enabling the device to track the magnetic resonance properties of brain tissue and identify clinically proven stroke biomarkers, such as molecular diffusion, in a compact, portable design.
Axana’s portability and simplicity make it ideal for bedside use, where it enables fast detection of stroke-related damage, and provides critical insights into its severity and extent, Wellumio claims, also noting that—by delivering this information quickly—Axana supports the prioritization of advanced imaging and guides urgent treatment decisions.
LSI events produce results. Check out inspiring stories from attendees on our website:

3M 8
A
Abbott 33
Abcam 49
Accelmed Partners .......... 19, 46
Accuray 8
AccurKardia 43
Action Potential Venture Capital 18
Adagio Medical 34
Adara Ventures, 49
Advanced Research and Invention Agency 19
Advent Life Sciences 18
AerWave Medical 18
Alan Cohen 7
Alistair Erskine, MD 10
Allen Meacham 10
Alleviant Medical 15, 18
Allumin8 19
Alpha JWC Ventures 18
Alydia Health...............................9
Amadeus Capital Partners 49
Amos Ziv 39
Amplitude Vascular Systems 19
Amzak Health 19
Anaconda Biomed 8, 28
Ananya Health 34
Andera Partners ....................... 18
Angel Alberich-Bayarri 49
Anhui Kunhua Fund 19
Anna Nestor 9
APEX Ventures 18, 49, 50
Arboretum Ventures 18, 46
ARCS 19
Arcuro Medical 18
Asabys 49
Ashish Kacholia 19
Aspect Biosystems .......... 18, 43
AtriAN Medical 33
AtriCure 32
Auris Health 9
Autonomix Medical 14
Avicella Capital 12, 19
Aviva Ventures 19
Axalta Coating Systems...........9
B
Bardy 39
Baxter ...........................................8
Benny Dilmoney 28
Bernard Zovighian 7
Bertrand Duplat 50
Beta Bionics 19
Bill Kelly 9
Biosense Webster 33
BioStar Capital 19
Biotelemetry 39
Bittium 50
Bob Gerberich .......................... 10
Bolt Medical 18
Ventures 18
Bryan Lord 49
Brymill Cryogenics 32
BTG plc...................................... 32
Buenavista Equity Partners 49
Build Collective 49
BVP Investments 19
Capricorn Partners 44 Capstan Medical 8
8
38-42
13
44
EBAMed 19, 45 Eduardo Fonseca 30 Efung Capital 19 eGarden Ventures 18 EIC Fund 45 Elevage
18
Ventures 18, 49
Chongqing Changsheng Shenghe Investment Fund . 19 Chris Eso 10 Chris Kaufmann, MD 47 Christopher Arapoff 45
Coloplast A/S 18, 46 Columbus Venture Partners 49
Sokolov 50
Dr. Catherine Mohr 20-23 Drew Trivisonno 9
Boston Scientific 9, 10, 18, 32, 33, 46 Brad Davis 9, 10 Brainlab ............................... 18, 50
Healthcare 15, 18, 19, 46
Capital 15, 18
Kandu Health 9
Kardium 33
KCI Medical .................................8
Kegg 18
Ken Mariash 50
Ken Nelson............................... 39
KFund 49
Khosla Ventures 13
Kindred Ventures .............. 18, 50
KryoLife 32
Kuros Biosciences 18
L Leadwind 49
Leonel Peralta 8
Liam Burns 44
LifeHealthcare 18
Lifeward 18
LIFTT Venture Capital Italia 45
Lightstone Ventures 18
LocalGlobe......................... 18, 50
Longitude Capital 18
Longview Ventures 15, 18
Long-Z Capital ......................... 18
Lucius Partners 18
Lumicell 47
Lumira Ventures....................... 19
M
Maculaser 47
Magneto Thrombectomy Solutions 28
Magnolia Medical Technologies 10
Manfred Franke 47
Maria Sainz 9
Marina Izzo 45
Massachusetts General Hospital ................................ 48
Mass General Brigham 10
Matt Curran 30
Mauna Kea Technologies ...... 14
Mayo Clinic 45
Mectronic Medicale 32
Medacta International 51
Medasense Biometrics 19
MedEdge Innovations 19
Median Technologies 13
Medtech Venture Partners 19, 46
Medtronic 8, 10, 18, 30, 33
Medtronic Diabetes 8
Mendaera 8
Mercator MedSystems 8
Merck Sharpe & Dohme 9
Mike Kujak 46
MindWorks Global 18
Mirai Souzou 19
Mizuho Capital Investments 19
Motif Neurotech 19
MVM Partners 19, 46
N
Nalu Medical 10, 14
Nancy Schlichting 8
Nanoflex Robotics 30
National Reconstruction Fund Corporation (NRFC) 19
Navigantis 28
NEA (New Enterprise Associates) 18
Neo Modulus 19
Neuronoff 47
Neurovasc Technologies ...... 28
NEVENT 48
Nevro 12
Nexstim ..................................... 50
Next Generation EU Initiative 19
Nicholas Pachuda 7
Nissan Motor Co. Ltd. ...............8
Novadip Biosciences 9, 48
Novo Holdings 18
Novo Nordisk 18, 43, 44
Novus Diagnostics 19
NUA Surgical 9 NVIDIA 18
O
OCCIDENT 45
Ohio State University Wexner Medical Center 51
Omega Funds..................... 15, 18
OmniSonics Medical Technologies 8
Onera Health .......................9, 48
Onkos Surgical 18, 49
Ontario Brain Institute 19
Ontario Centre of Innovation ..19
OpSens 9
Organon 9
Orlando Health Ventures 18, 46
Outset Medical 18
Owlstone Medical 19
OYMotion Technologies 19 P
Pacific Health Investment 18
Pallasite Ventures 18, 44
Panakès Partners 45
Pangaea Ventures ............ 18, 44
Partech 49
Patrick Treacy 49
Paul LaViolette ....................... 7, 9
Peakvest 18
Penumbra 27
Perceptive Technologies 19
Peregrine Ventures 28
Per Langoe 46
Phil Green 21
Philips 8
PhotonMed 47
Piper Heartland
Healthcare Capital 18
Plural 18, 49
PNRR (Piano Nazionale di Ripresa e Resilienza) 19
Polaris Partners 18
Precision Life Science Partners 9
PricewaterhouseCoopers 8
Pristine Surgical 49
Progressive NEURO 28
Proprio ........................................ 18
Prosoma 19
Prosperity7 18
Proxima Ventures .................... 19
Pudong Venture Capital 18
Pulse Biosciences 9, 12 Q
Qaelon Medical 44
Qiming Venture Partners 18, 19
Quibim 49
Radical Ventures 18, 43
Ramin Mousavi 45
RapidAI 13, 30
Rapid Medical 30
RapidPulse ............................... 28
Redmile Group 18
Relentless Consumer Partners 18
Relief Cardiovascular 18
Remedy Robotics ................... 30
Renault Group 8
Resmonics 19
Rhino Ventures 18, 44
Rhodel Dacanay 48
RITA Medical Systems .............8
RiverVest Venture Partners 15, 18
Robeauté 18, 49
Robert Younge 21
RocGene Technology ............. 19
Roivios 44
Route 92 Medical 28
ROX Medical 8
Ruben de Francisco 48 S
S3 Ventures 15, 18
Saluda Medical 18
Samsara Vision ........................ 16
Sands Capital 18
Sanmei Investment 19
ScreenPoint Medical 13
Sedivention 34
Sense Neuro Diagnostics .... 28
Sequoia China 19
Seventure Partners 18
Shenzhen Capital Group 19
Shieak Tzeng 51
Shockwave Medical ..................9
Shunbai Fund 19
Siemens Healthineers 8
Silicon Prairie Center 28
Simpact Ventures 19
Simpson Interventions .......... 50
Sinaptica Therapeutics 50
Single Pass 9
Skandalaris Center Global Impact Award 19
Smartlink Partners ................... 19
Solardis Health Ventures 39
Solas BioVentures 18, 46
Sound Blade Medical 19
SpectraWAVE 9
Spine Align ............................... 39
Spiro Robotics 50
Stanford Research Institute 21
StarTric 51
Steven Mickelsen, MD 46
Stimdia Medical ....................... 10
Stryker 6, 27
Stuart Simpson 51
Suzanne

September 7th - 11th, 2025
JW Marriott Grosvenor House, London
Now in its fourth year, LSI Europe has become a staple of the global Medtech and Healthtech community. The 2025 event in London will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Registration and applications to present are now open for LSI Europe ‘25. Get in touch to learn more about the event.


