Joe
Kiani

Kiani
Reflections on Principle, Persistence, and Patient-Centered Innovation

March 2025 Hot Takes, Wearable Monitoring Devices, LSI Alumni Milestones, and more...
February 2025
June 10th - 13th, 2025
The Ritz-Carlton, Millenia Singapore
With the global community built through LSI USA and LSI Europe and our partnerships in Singapore, China, Korea, Japan and more, LSI Asia will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Like LSI USA and LSI Europe, LSI Asia will not be held in a chaotic convention center. We have identified a world-class venue in Singapore, the gateway to Southeast Asia, for our inaugural event where senior executives can do real business.



Thank you to our founding sponsors
As we launch in this new geography, we are offering a unique opportunity to a small set of value-add, mission-aligned partners looking to elevate their global presence and develop new business.

Scott Pantel
Chief Executive Officer, Editor-in-Chief

Rebekah Murrietta Vice President of Media, Contributing Author

Maricela Almonte Customer Service & Fulfillment

Henry Peck
Chief Business Officer, Editor-in-Chief

Brenna Hopkins Sr. Content Manager, Contributing Author

Kelly Williams Subscriptions & Enterprise Sales
Printed in the U.S.A.
A publication of Life Science Intelligence, Inc. (LSI) 17011 Beach Boulevard, Suite 500 Huntington Beach CA 92647
714 847 3540 tel/fax email: info@ls-intel.com
Vol. 2, No. 3
March 2025

Tracy Schaaf
Managing Editor and Content Strategist, Lead Author

Blake Matrone Sr. Marketing Manager, Contributing Author

Benny Tomlin Contributing Photographer

Nicholas Talamantes Sr. Director of Market Intelligence, Contributing Author

Gavin Kennedy Business Development

Paul Streeto Graphic Designer
Created by LSI, The Lens leverages LSI’s deep industry relationships and proprietary market intelligence to help executives like you build lasting medtech companies. Start your Individual, Group or Enterprise-Wide subscription today!

Scan QR Code to activate your subscription.
The Lens is published monthly and available in print and electronic formats. Copyright by Life Science Intelligence, Inc. All rights reserved. Editor takes care to report information from reliable sources and does not assume liability for information published.
Interested in subscribing? email to: info@ls-intel.com
Interested in advertising? email to: kelly@ls-intel.com


Dear Readers,
We are still wearing the kind of ear-to-ear smiles that only a week of unforgettable conversations and connections can bring. LSI USA ‘25, our ninth global summit and sixth in the United States, brought together more than 1,600 of medtech’s brightest minds. With hundreds of keynotes, panels, and presentations, thousands of scheduled meetings, and tens of thousands of spontaneous ones, we look forward to tracking the deals sparked in Dana Point—and spotlighting them in future editions of The Lens
One highlight of the event was the opening Keynote delivered by Joe Kiani, the Founder and Executive Chairman of Willow Laboratories, and previous Founder of Masimo, a global medical technology company renowned for noninvasive patient monitoring technologies. Joe took the stage before a packed ballroom with a speech in hand, but he never delivered it. Instead, he gave an unfiltered, unvarnished, emotional reflection of his life and story.
Joe offered a rare window into the making of one of medtech’s most influential companies—from a “garage start-up” in 1989 to a publicly traded, multi-billion-dollar enterprise with far-reaching products like their Masimo Signal Extraction Technology, the primary pulse oximetry at the top 10 hospitals in the United States and throughout the world. Now, he is leading Willow Labs and preparing to commercialize several new technologies with groundbreaking potential.
In addition to his insights on technology, commercialization, and financing, Joe issued a powerful reminder of the difficult responsibility and immutable opportunity our industry has to change people’s lives for the better. What he shared were more than tales of companies built and innovations developed, but an homage to the patients he served and the trust they put in him. As he said, the responsibility and opportunity to improve patient outcomes “is not easy, but it’s everything.”
Alongside Joe’s inspiring vision, this edition of The Lens features timely intelligence from LSI’s Global Procedure Volumes Database on general surgery, including 2023-2030 data and forecasts for colorectal surgeries, appendectomies, hernia repairs, and bariatric procedures. The latter is especially timely, as conversations around weight-loss treatments intensify with the rise of GLP-1 drugs like Ozempic and Wegovy.
We also explore the booming wearable monitoring device market, projected to reach $21.2 billion by 2028. From Dexcom and Abbott to Medtronic and Apple (whose Apple Watch now holds 86% of the global wearables market), the battle between medtech and consumer tech is heating up—and the implications are enormous.
With this edition of The Lens, we invite you to reflect on the responsibility and opportunity you have to serve patients.
We’re honored to be on this journey with you.
All the best,
Scott Pantel Chief Executive Officer, LSI Editor-in-Chief


Henry Peck Chief Business Officer, LSI Editor-in-Chief

Need fresh inspiration for your medtech company as we navigate 1H25? We’ve gathered insights from some of our LSI alumni thought leaders to help you stay ahead.
“I would choose debate and controversy over just complacency and yes, all the time. Actually, even when I’m in agreement with the CEO, sometimes on a board of directors, I disagree just for the fun of it, just for the learning of it. Let’s debate. Let me tell you a contrarian opinion. Let me ask you why. Let me ask you to go back to the drawing board and rethink this, even if I agree. I think that the boards that actually foster a culture of debate, including with their investors, in a constructive manner, are the ones that are building this partnership in the best way.”

— Marta G. Zanchi, Founder & Managing Partner, Nina Capital
LSI Europe ‘24 Fireside Chat: “Value-Add” is a Two-Way Street: Success Through Investor-Founder Partnerships
“You know, there’s high, there’s low, and then there’s the price at which a deal actually gets done.”

— Jay Watkins, Co-Chair & Partner, Sonder Capital
LSI USA ‘24 Panel: Cracking the Code: Finding the Optimal Valuation for Successful Capital Infusion and M&A Deals
“I think in a tough market, it’s actually very good for good companies with a good value proposition because the best funds all want in on the same deals.”

— Aidan Crawley, Co-founder & CEO, Amber Therapeutics
LSI Europe ‘24 Panel: Medtech Financing in Europe: Current State and Insights from Succesful Raises
“We’re at a moment of convergence, where transformational technology meets the realities of global health challenges. This isn’t just an opportunity; it’s a call to innovate in ways that truly enable a healthier world.”

— Rick Anderson, Chairman, Revival Healthcare Capital
LSI USA ‘24 Keynote: Reimagining How We Innovate in Medtech, with Alex Gorsky and Rick Anderson
“I’ve never seen more strategic interest in innovation. The onus is on us collectively to figure out new ways to align with these buyers. If you can do that, it’s going to open up a lot of efficiency and take away the black mark of medtech being a poor asset class for investment.”

— Duke Rohlen, CEO & Managing Partner, Ajax Health
LSI Europe ‘24 Keynote: The Middle Path to Innovation
“What happens when you become paralyzed is you lose your agency in the world, you become dependent on someone else, you lose your privacy, you lose your autonomy... The feeling the patients get when they can make a selection on a screen independently is very powerful.”

— Thomas Oxley, CEO, Synchron
LSI USA ‘24 Fireside Chat: Neurotechnology to Address the Limitations of the Human Body
Celebrating recent leadership shifts and other announcements and accolades impacting our global medtech community.
1MED SA, a Swiss provider of global expertise in the development of medical devices, novel technologies, and combination products, and an LSI alumni, recently announced two new executive appointments. The company has appointed David Bird as Chief Commercial Officer and John Thomas

as Director of Strategic Delivery. Bird brings 25 years of experience in commercial strategy and business growth across the medtech, biotech, and pharmaceutical sectors, while Thomas has over 20 years of global experience in the CRO, medical devices, and biotech sectors.
Accelus, a privately held medtech company and LSI alumni offering innovative expandable spinal implant technologies, announced that it has promoted Jim Fox to Vice President of U.S. Sales, expanding his leadership

role in driving nationwide commercial growth. Fox, who previously led U.S. East Coast sales for the FL-based company, and with past leadership roles at Medtronic, Biomet, Benvenue Medical, and Safe Passage Neuromonitoring, will focus on strengthening distributor partnerships and increasing adoption of Accelus’ spinal implants.
BrightHeart, a French developer of AI-based software for prenatal congenital heart defect screening, recently announced the addition of serial entrepreneur Amanda French to the team

as Chief Strategy Officer. Previously, Amanda ran Emme, a Stanford Biodesign spinout and women’s digital health company, from concept through commercial launch and successful acquisition. Amanda started her career at Edwards Lifesciences and Earlens before going on to the Stanford Biodesign Fellowship and subsequent executive roles at Emme and SimpleHealth, with clients including NXT Biomedical and Cardiex
CorVista Health has appointed Adrian Lam as President and CEO, succeeding Don Crawford as he retires

from his 42-year medtech career. Additionally, Tim Attebery—CEO of Cardiovascular Associates of America, former CEO of the American College of Cardiology, and current Board Director—was appointed Board Chairman. Bethesda, MD-based CorVista Health has developed the CorVista System, a non-invasive point-of-care solution intended to synchronously collect and apply machine learning to a symptomatic

patient’s cardiac and hemodynamic signals to predict the likelihood of cardiovascular diseases without the use of radiation, contrast agents, injections, fasting, or exercise.
Ditch Labs, a Montreal-based life sciences company developing innovative solutions for smoking and vaping cessation, has announced that Peter Bloch has joined the company as Chairman of the Board of Directors. Bloch,

whose previous executive roles include serving as CEO and Chairman of Bionik Labs, brings extensive leadership experience in the medical technology sector to guide the company’s strategic direction as it prepares for a pivotal trial in the UK and clinical trials in the U.S. The company’s flagship product, the DitchPen, combines medical-grade nicotine replacement therapy with a user-friendly digital app to deliver a personalized, automated cessation program.
Iantrek, a venture-backed medical technology company pioneering next-generation bio- and micro-interventional approaches for ophthalmic disease treatment, announced the appointment of LSI Alumni Adam Szaronos as CEO, where he will lead

the company’s next phase of growth. Szaronos, an ophthalmic industry executive, brings extensive experience in commercial strategy and medical device innovation.
Imaginostics, a healthtech startup developing a breakthrough longevity technology for precision medicine located in Orlando, FL, has appointed Co-Founder Valerie Gharagouzloo

as CEO. Imaginostics’ Quantitative Ultrashort Time-to-Echo Contrast-Enhanced MRI (QUTE-CE MRI) technology provides clinicians and researchers unprecedented quantitative insights into vascular health, enabling early disease detection, more effective treatments, and proactive management of health and longevity.
Mountain View, CA-based Levita Magnetics recently announced that Jon Beltrone has been appointed as

VP of Global Sales, where he will expand access to the company’s MARS Surgical System. With experience driving the adoption of robotic surgery systems at Stryker and Momentis, Beltrone will lead the company’s commercialization
efforts. MARS, a surgical platform that leverages Levita’s proprietary Dynamic Magnetic Positioning technology, has FDA approval for use in high-volume abdominal surgeries including laparoscopic bariatric surgeries, cholecystectomy, prostate, and colorectal procedures.
Seoul, South Korea-based medical robotics company LN Robotics has announced that Donghee Kim has

joined the company as CEO. Leveraging her 20 years of experience at multinational medtech companies, Kim will drive strategic partnerships and global growth initiatives. Kim previously served as CEO of Philips Korea, led the Health Systems business for Philips Asia-Pacific, led Edwards Lifesciences’ transcatheter aortic valve implantation business in major Asia-Pacific markets, and led marketing for drug-eluting stents and intracardiac ultrasound products at Boston Scientific. In February 2023, LN Robotics’ AVIAR became the first coronary intervention robot to receive approval from South Korea’s Ministry of Food and Drug Safety. Since then, clinical trials have been undergoing at several university hospitals.
Nina Capital, a Barcelona-based VC firm dedicated to healthcare transformation through technology, announced that LSI Alumni Nadin Youssef has

been promoted to Associate at the firm. Youssef, an alumni in Medical Device Design & Entrepreneurship at Imperial College London, was previously Senior Analyst at Nina Capital, where her dedication and contributions were
instrumental to Nina Capital’s continued success in supporting need-driven health tech founders.
Novuson Surgical, a Bothell, WAbased company dedicated to advancing surgical precision and safety through innovative energy-based solutions, has appointed Mart Pearson as

its Executive Vice President of Business Development. Pearson brings over three decades of medical device commercialization experience and will play a pivotal role in driving the commercialization of Novuson’s Direct Therapeutic Ultrasound technology for surgical applications. Pearson previously played a key role in the successful commercialization of the Aquamantys system prior to its acquisition by Medtronic
NOxy Health Products, a healthcare company driven to developing innovative medicines that harness the natural restorative power of nitric oxide to heal chronic wounds, has announced the addition of Sarah Griffiths Langbord, PhD as its Chief Scientific Officer. Griffiths Langbord will accelerate the

clinical development of NOX1416, the company’s topical nitric oxide drug for chronic wounds, and expand real-world data for NOxyDERM. Additionally, she will lead the company’s R&D innovations to expand its indication platform, help guide NOxy’s strategic vision, and identify top talent who are passionate about the impact nitric oxide can make in wound-healing and dermatology at large.
Scottsdale, AZ-based NurseIO announced the appointment of LSI alumni Shane Brown as CEO, to lead the company’s expansion in the healthcare

staffing industry. Under Brown’s leadership, NurseIO will further develop its technology-powered staffing solutions and expand its network of healthcare professionals and facilities in the U.S.
Orchestra Biomed, a biomedical company accelerating high-impact technologies to patients through riskreward sharing partnerships, has appointed Vivek Reddy, MD as Executive Chairman of the BACKBEAT Study

Steering Committee and Chairman of its Bioelectronic Therapies Scientific Advisory Board. In these roles, Dr. Reddy will provide critical leadership in continuing to execute the BACKBEAT global pivotal study and further develop scientific evidence and clinical value for the company’s bioelectronic therapies portfolio. The company’s lead product candidate is atrioventricular interval modulation (AVIM) therapy for the treatment of hypertension. It is also developing the Virtue Sirolimus AngioInfusion Balloon (SAB) for the treatment of atherosclerotic artery disease.
Orchestra Biomed also announced that it had appointed Christopher Cleary, former Medtronic SVP of Corporate Development, to its Board of Directors. Cleary brings over three decades of experience in corporate development, mergers, acquisitions, strategic investments and structured

R&D collaborations, having significantly impacted the medical technology industry through his strategic leadership and visionary approach, including his role in shaping Orchestra BioMed’s collaboration with Medtronic for AVIM therapy. (As announced in the January 2025 issue of The Lens, Cleary also recently joined Pristine Surgical’s Board of Directors.)
Osseointegrative ceramic bone graft substitute company Ossiform announced that it has appointed Jackie Fielding to its Board of Directors. With

over 30 years in medical device commercialization, including 18 years at Medtronic, Fielding brings extensive expertise to support Ossiform’s mission of advancing 3D-printed, bio-integrative, patient-specific bone implants.
Memphis, TN-based OsteoRemedies has brought on LSI alumni Ted Bird

as Vice President of Spine Commercial to lead its new U.S. spine division. Bird, a global medtech executive specializing in commercialization strategy, business development, education and product marketing, will oversee the launch of
sterile-packaged spine procedure kits and drive strategic growth in outpatient spine surgery.
Brussels, Belgium-based Synergia Medical announced that Charles Nolet has succeeded Attila Borbath

as CEO, effective February 1, 2025. Nolet brings significant strategic and operational expertise, including from his tenure at McKinsey & Company He also served as Investment Manager at Lumine Capital Advisors, where he supported entrepreneurs and start-ups, particularly in the medtech sector. Most recently, he served as Chief Strategy Officer for Synergia Medical, where he played a key role in refining the company’s strategy and guiding management during a pivotal phase. Synergia Medical pioneers optoelectronic neurostimulation, offering a fully MRI-compatible, rechargeable vagus nerve stimulation (VNS) device that is revolutionizing epilepsy treatment with advanced safety and reliability.
The Netherlands-based ViCentra, developer of Kaleido, one of the world’s smallest, lightest, and most precise insulin patch pump and automated insulin delivery (AID) systems, recently appointed Tom Arnold as its

CEO to lead its next phase of growth. Arnold brings extensive experience in diabetes technology and will oversee the expansion of the Kaleido insulin pump and automated insulin delivery system.
Highlights: Neuromodulation, mobile concussion testing, cryoablation, pediatric vascular access, continuous hormone monitoring, and other innovations advance with fresh new funding, regulatory approvals, and clinical evidence.
Commercial-stage neuromodulation company Saluda Medical recently presented new clinical data at the 2025 North American Neuromodulation Society (NANS) Annual Meeting, held from January 30-February 1, 2025, in Orlando, FL. Data from 11 total abstracts and four oral presentations, two of which received the “Top Oral Abstract” award, underscore the significance of objective neurophysiological measurements in spinal cord stimulation (SCS) with evoked compound action potential (ECAP) dose-controlled closedloop therapy, and the potential clinical benefits of Saluda Medical’s recently FDA-approved automated patient programming platform, EVA.
EVA is a biomarker-based, automated patient programming platform in SCS, representing a significant advancement for the therapy. The new programming platform is compatible with all commercially implanted Evoke System patients in the U.S. EVA is designed to improve the SCS patient programming experience by automating manual programming steps and autonomously scanning and analyzing a patient’s nerves to optimize therapy settings. The automated programming workflow has the potential to minimize the burden of care by substantially reducing the time element of programming and elevating the overall patient experience, while simultaneously improving daily clinic throughput in busy pain practices.
In the EVA Automated Programming study, 94% of all programming sessions using the automated programming platform successfully generated a closed-loop program; there was less than a 13-minute median time to generate each patient-specific automated program; and ≥90% of patients were satisfied or very satisfied with the automated programming platform experience.
Highlighting results from the Top Oral Abstracts, the ECAP IDE Study builds on the EVOKE IDE Study and represents the largest SCS study executed in a real-world cohort of 220 patients. Real-world data demonstrated profound improvement across all pain subgroups in the study including pain associated with failed back surgery syndrome, non-surgical back pain, complex regional pain syndrome, diabetic peripheral neuropathy, and upper limb pain.
Levee Medical, a Durham, NC-based device company focused on improving outcomes for prostate cancer surgery patients, announced the successful close of its Series B financing, totaling more than $14 million including note conversions. The proceeds will accelerate the development of the Voro Urologic Scaffold, an absorbable device designed to reduce post-prostatectomy urinary incontinence. The funding will also support ongoing clinical studies, including an upcoming U.S. pivotal trial and current ARID studies.
In March, the company also announced that the FDA has granted approval to initiate the ARID II (A Pivotal Study of VoRo UrologIc ScaffolD for the Treatment of Post Prostatectomy Stress Urinary Incontinence) clinical trial, under an Investigational Device Exemption (IDE). ARID II is a multicenter, single blind, randomized controlled study designed to demonstrate the safety and effectiveness of the Voro Urologic Scaffold in adult men undergoing robotic assisted radical prostatectomy as compared to control. Arvin K. George, MD, Director of Prostate Cancer Programs at Johns Hopkins School of Medicine, will serve as the national principal investigator for the ARID II trial.
The Voro Urologic Scaffold is designed to be placed during a prostatectomy procedure to manage the geometry of the bladder neck and maintain urethral length, which are critical factors in preventing post-operative incontinence. This innovative, absorbable device aims to markedly reduce the incidence of stress urinary incontinence, providing a much-needed solution for patients undergoing radical prostatectomy.
Prostate cancer is the second most common cancer among men, and the primary type of surgery to handle the disease is radical prostatectomy. Nearly all men experience urinary incontinence after the procedure, and about 15% of patients develop a chronic problem. Current therapies for post-prostatectomy incontinence are invasive, inconvenient and inadequate, according to Levee.
Neuvotion is a Stamford, CT-based early-stage medtech company developing AI-driven neuromodulation technologies and products for use in the neurorehabilitation, brain-computer interface (BCI), and physical therapy markets. The company recently received FDA 510(k) clearance for their first product, NeuStim, a non-invasive, surgery-free wearable that electrically stimulates muscles dynamically and with high-precision. NeuStim supports hand movement recovery after stroke or spinal cord injury and allows clinicians to quickly scan and pinpoint stimulation targets through an innovative touchscreen interface. Stimulation profiles can be quickly set up and saved for each user through a graphical user interface on a lightweight mobile device. Neuvotion plans to launch NeuStim in the coming year.
Combining AI, highly focal stimulation, and BCI-compatible technology, the company’s approach will deliver unparalleled therapeutic value for users. Neuvotion’s devices will allow clinicians to facilitate and monitor patient progress in-person or remotely, while providing leading-edge care. (Also see Market Dive: Wearable Monitoring Devices, this issue.)
Navi Medical Technologies, a Melbourne, Australia-based medical device company focused on pediatric healthcare innovation, recently announced that its Neonav ECG Tip Location System has received 510(k) clearance from the FDA. This milestone represents a significant step forward in vascular access care for critically ill newborns and children worldwide.
The Neonav is the first device of its kind specifically designed for neonatal and pediatric care. By using real-time ECG signal analysis, it aids accurate placement of central venous access devices, significantly reducing the risks associated with misplacement and migration which can cost U.S.
hospitals up to $1 billion per year. This breakthrough innovation minimises reliance on confirmatory chest X-rays, will help in reducing delays in care, as well as lowering unintended complications for vulnerable patients.
The Neonav system has the broadest indication for use of any tip location system on the market, covering preterm newborns through to adults. Importantly, the system can be used to place tiny 1Fr catheters used in very small newborns, and is cleared for placement of catheters both above and below the heart. The system has also been developed to enable ongoing surveillance of catheters after the initial placement, an important first which can help to prevent potentially serious healthcare complications resulting from undiagnosed movement of the tip of the catheter.
Navi Medical Technologies’ investors and partners include Breakthrough Victoria, The Royal Women’s Hospital, the Australian Federal and Victorian Governments, MTPConnect, and the U.S. FDA Pediatric Device Consortia program.
Northernmost Medical Devices, a Canadian medtech startup developing a next-generation perfusion machine, the NoMo Kidney Pump, is the recipient of the first investment in 2025 for the National Kidney Foundation (NKF) Innovation Fund. NKF will help fund development of Northernmost’s new technology, a system designed to modernize the preservation and transport of donor kidneys for transplantation.
Currently, 71% of donor kidneys are preserved in static cold storage, a no-tech method that risks organ damage, limits safe storage time, and causes delayed graft function and other adverse events. Perfusion machines circulate fluid through the kidney, optimizing preservation and providing realtime data on organ viability. But despite decades of clinical evidence demanding continuous perfusion, all the way from donor to recipient, today’s machines are too large, unwieldy, and expensive to do the job routinely. With NoMo’s small, simple, and smart design, Northernmost intends to eliminate ice coolers altogether.
With nearly 90,000 people currently on the U.S. kidney transplant waitlist, improving how donor organs are handled is critical. Research indicates that adopting continuous perfusion systems like NoMo could reduce kidney discards by more than 50% and save tens of billions in healthcare costs over the next decade.
This strategic investment aligns with NKF’s commitment to advancing innovations that improve kidney transplantation outcomes, reduce organ discard rates, and expand access to life-saving transplants.
Also in February, Northernmost announced a collaboration with Mayo Clinic to test and advance its technology and services. Mayo Clinic is the largest integrated transplant provider in the U.S., performing nearly twice as many solid-organ transplants as any other center. Northernmost leadership, engineers, and industrial designers will be collaborating with surgeons and administrators at Mayo Clinic transplant programs in Rochester, MN; Phoenix, AZ; and Jacksonville, FL.
IceCure Medical, an Israel-based developer of minimally invasive cryoablation technology that destroys tumors by freezing as an alternative to surgical tumor removal, announced it has filed for regulatory approval with the Medical Device Division of Israel’s Ministry of Health for its next-generation single cryoprobe cryoablation system, the XSense System and its cryoprobes.
The filing contains a request for approval of all indications for which ProSense has already received approval in Israel, including general surgery, dermatology, neurology, including cryoanalgesia, thoracic surgery, ENT, gynecology, oncology (including benign and malignant breast tumors), proctology, and urology.
IceCure Medical develops and markets advanced liquid-nitrogen-based cryoablation therapy systems for the destruction of tumors (benign and cancerous) by freezing, with the primary focus areas being breast, kidney, bone and lung cancer. Its minimally invasive technology is a safe and effective alternative to hospital surgical tumor removal that is easily performed in a relatively short procedure. The company’s flagship ProSense system is marketed and sold worldwide for the indications cleared and approved to date including in the U.S., Europe, and China.
San Francisco, CA-based Averto Medical, a clinical-stage medical device company pioneering minimally invasive gastrointestinal care, recently announced that the FDA has granted Breakthrough Device Designation for its ColoSeal Intraluminal Colonic Diversion (ICD) System. This designation
underscores the potential of ColoSeal to significantly improve outcomes for patients undergoing colorectal surgery by eliminating the need for a temporary ostomy, enhancing patient recovery and reducing complications associated with traditional surgical methods.
The FDA’s Breakthrough Device Program is designed to expedite the development and review of medical devices that offer substantial advantages over existing alternatives in treating or diagnosing life-threatening or irreversibly debilitating diseases. With this designation, Averto Medical will benefit from prioritized FDA interactions and a streamlined review process, facilitating timely access to this innovative solution for patients and healthcare providers.

Tulsa, OK-based Sway Medical has received FDA 510(k) clearance as a Computerized Cognitive Assessment Aid for Concussion under Section 882.1471. This clearance expands on Sway Medical’s previous FDA clearance for balance testing in head injuries, officially recognizing Sway as the first fully integrated tool that combines both cognitive and balance testing into one product for concussion management.
This milestone reinforces Sway’s commitment to delivering clinically validated, real-world solutions for concussion assessment, aligning with the latest Concussion in Sport Group Consensus Statement, which highlights multi-modal testing as the gold standard for concussion care.
Concussions are complex injuries requiring a multi-faceted assessment approach. While many existing tools focus only on cognitive testing, Sway combines balance and cognitive evaluations in a single, FDA-cleared platform, providing a complete and objective approach to concussion care. Sway transforms any mobile device into a Class II medical device that leverages patented algorithms and advanced motion analysis technology. This innovative platform detects subtle changes in patient performance following a concussion, enabling healthcare professionals to assess, track, and manage concussions with greater accuracy and efficiency.
Pleasanton, CA-based Movano Health has commenced a blood pressure clinical trial with its new radiofrequency-enabled cuffless wrist wearable. The company designed its updated device to monitor pulse pressure waveforms. The
study, expected to include at least 70 participants, is utilizing Movano’s device in comparison with a hospital-grade blood pressure device.
In addition to blood pressure, Movano developed the device to collect data on a number of other vital signs including pulse rate, blood oxygen saturation (SpO2), respiration rate and ECG waveforms.
The company’s latest study follows three clinical trials that took place in 2023 and 2024. Those studies used non-invasive devices to collect pulse pressure waveform data from more than 60 participants of varying gender, age, ethnicity and BMI. During each session, participants wore Movano’s wrist-worn device along with an FDA-cleared, hospital-grade vital signs monitor with cuffed blood pressure measurements.
In mid-2025, the company plans to execute an additional 100 to 150-participant continuous blood pressure study using an arterial line, with results expected to help finalize the algorithm used in a potential FDA pivotal trial in the future. (Also see Market Dive: Wearable Monitoring Devices, this issue.)
Kirkland, WA-based Kestra Medical Technologies, a wearable medical device and digital healthcare company, recently announced the closing of its upsized initial public offering of 11.8 million common shares at a public offering price of $17.00 per share. The total gross proceeds from the offering, before deducting underwriting discounts and commissions and other expenses, were approximately $202 million.
Kestra’s common shares began trading on the Nasdaq Global Select Market on March 6, under the ticker symbol KMTS. Kestra was founded in 2014 by leaders from the external and implantable defibrillation industries.
The ASSURE system from Kestra is a wearable cardioverter defibrillator (WCD) offering integrated sensing, autonomous detection, and highly effective defibrillation therapy, designed to protect users from sudden cardiac arrest (SCA). The vest-like system includes the Kestra CareStation remote patient data platform, designed to provide robust management of at-risk cardiac patients, and the ASSURE patient app to engage patients in their own recovery and care. The components work in concert to promote patient compliance while offering protection during normal patient activities.
Heart failure (HF) patients and patients with low left ventricular ejection fraction (low LVEF) of 35% or below are at elevated risk of SCA while waiting for their condition to improve or until eligible for subsequent therapy. WCDs are non-invasive,
monitor around the clock, and can protect patients who are at known risk for SCA. (Also see Market Dive: Wearable Monitoring Devices, this issue.)
Danish company Cercare Medical, a leader in perfusion software for stroke and neurodegenerative diseases since 2013, recently announced that it has raised €7.4 million ($7.7 million) in its latest funding round, led by UK-based West Hill Capital. The funding will be used to expand operations in key markets, with a focus on the U.S., the UK, and selected European countries.
Cercare Medical’s cutting-edge software provides advanced perfusion maps for both CT and MRI imaging, equipping clinicians with a comprehensive tool to assess brain tissue pathologies. Over recent years, the company has achieved yearly triple-digit growth and has been adopted by prominent stroke and oncology facilities in more than 23 countries. Its groundbreaking technology has also been featured in leading medical journals and cited in more than 30 key clinical publications. As part of its expansion strategy, Cercare Medical is deepening its collaborations with key industry partners, including Microsoft, Siemens, Blackford, Aidoc, SECTRA, TeraRecon, and Viz.AI
With fully automated and multi-vendor compatible software for CT and MRI perfusion imaging post-processing, Cercare Medical is committed to delivering cutting-edge technology that enhances patient care and advances medical diagnostics across various neurological conditions, including stroke, oncology, Alzheimer’s, dementia, and COVID-19. The company’s proprietary biomarkers allow for better, more precise imaging maps, improving decision-making in critical care, and enhancing neuroimaging.
Onc.AI, a Canadian digital health company developing AI-driven oncology clinical management solutions using advanced deep learning applied to routine diagnostic images, announced that it was awarded FDA Breakthrough Device Designation for its Serial CTRS (Serial CT Response Score) AI model, an artificial intelligence (AI)-based prognostic tool that uses deep learning to analyze CT scans and stratify patients into high- or low-mortality risk categories.
A multi-institutional study demonstrated that Serial CTRS improved the prediction of overall survival for patients
receiving immunotherapy compared to traditional imaging assessment tools. This highlights the model’s ability to deliver more accurate, data-driven insights into patient outcomes, enabling more precise, personalized cancer care.
The company’s platform is applied at the point of care to medical oncologists. Onc.AI is backed by premier institutional investors, including Sandbox/Blue Venture Fund, Action Potential Venture Capital, MassMutual Alternative Investments, Accomplice, Digitalis, KdT, and Life Extension Ventures. Onc.AI is also supported by the National Cancer Institute SBIR program.
Atreon Orthopedics, a Columbus, OH-based start-up innovator in tissue healing and regenerative technologies, recently announced FDA 510(k) clearance and the full market launch of BioCharge Autobiologic Matrix, a bioresorbable synthetic implant designed to address biological failure modes in rotator cuff repair while improving repair integrity and long-term patient outcomes.
The company was also FDA 510(k)-cleared to expand the use of its ROTIUM Bioresorbable Wick to all tendon repair, building on its proven success in rotator cuff procedures.
Atreon’s electrospun nanofiber platform technology distinguishes itself from traditional augment devices, which rely on animal-processed collagen or human dermal allografts that carry inherent patient compatibility risks and have a costly manufacturing process. With 17 years of tissue remodeling research, Atreon’s design mimics the natural structure of tissues and drives cellular attachment and proliferation more effectively than market leading collagen-based products
London-based healthtech startup Level Zero Health recently announced a €6.6 million ($7.2 million) pre-seed funding round to enable remote and continuous hormone monitoring, using unique DNA-based sensors. The round was led by Redalpine Venture Partners, HAX (SOSV), Entrepreneur First, with industry experts also contributing to the round.
According to Level Zero Health, although hormones play a critical role in our health, clinicians have relied solely on laboratory blood tests, which only capture a single moment in time
and require specialized equipment and expertise. The innovator is focused on changing this paradigm with technology that enables remote and continuous hormone monitoring, opening up new possibilities for understanding the body’s chemistry. Instead of invasive blood draws, Level Zero Health’s DNAbased sensors detect hormone levels in interstitial fluid–the fluid surrounding cells and tissues. In just under a year, the company has validated its DNA-based sensors in simulated samples across 98% of the human clinical range, reportedly outpacing industry standards.
Founded in 2023 by Ula Rustamova, ex-Palantir Enterprise Tech Lead and wearable startup founder, alongside Irene Jia, who developed medical devices at Philips, Level Zero Health focuses on wearable technology innovation and hormone health. Its technology uses proprietary processes for remote intermittent and continuous hormone monitoring.
Medtech start-up Emboa Medical has created a patent-pending microstructured catheter that mimics a boa constrictor’s teeth to grab onto blood clots and increase extraction forces without tearing. Its patent-pending device, called TRAP, or thrombus retrieval aspiration platform, has been validated to improve outcomes for stroke patients. The company’s biomimetic design has demonstrated a 200% increase in blockage removal force compared to a traditional catheter.
Emboa Medical was founded by clinical experts at Goodman Campbell Brain and Spine and NYU Langone Health, and device researchers at Purdue University’s College of Engineering. TRAP was designed by Angel Enríquez and Hyowon Lee, professor in Purdue’s Weldon School of Biomedical Engineering and director of the Center for Implantable Devices. They disclosed TRAP to the Purdue Innovates Office of Technology Commercialization (OTC), which submitted a patent application to protect the intellectual property. OTC also issued a license to Emboa Medical to commercialize the IP. The company plans to conduct an in vivo porcine model study to preliminarily validate TRAP’s safety and effectiveness by the end of Q1 2025.
Since its launch, Emboa Medical has been awarded more than $150,000 in nondilutive funding through winning pitch competitions such as Medtech Color, Purdue Innovates’ New Venture Challenge and Startup Prize: Focus on Health. The company is also a Seedling award recipient for the National Institutes of Health’s Blueprint Medtech program.


Ventures (led), Investissement Québec, CABHI (Centre for Aging + Brain Health Innovation), Cedars-Sinai

September 7th - 11th, 2025
JW Marriott Grosvenor House, London
Now in its fourth year, LSI Europe has become a staple of the global Medtech and Healthtech community. The 2025 event in London will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Registration and applications to present are now open for LSI Europe ‘25. Get in touch to learn more about the event.



25)
Joe Kiani stood before the packed ballroom at LSI USA ’25 in Dana Point with a speech in hand. But he never delivered it. Instead, he told a story—unscripted, emotional, raw. It was a story not just of companies built or innovations commercialized, but of the angels who showed up along the way, the systems that tried to shut him out, and the patients whose lives gave meaning to every battle he fought.
“We have a real responsibility and an opportunity to change people’s lives for the better,” Kiani tells the audience. “And it’s not easy. But it’s everything.”
This is the story of Joe Kiani—a founder whose first company, Masimo, grew from a “garage startup,” as he calls it, to a global medical technology powerhouse, and whose new company, Willow Laboratories, is set on reimagining diabetes care. But more than that, it’s the story of a leader guided not by market share or IPO timing, but by something far more enduring: the North Star of doing what’s best for patient care.
Just days before his keynote, Kiani had a chance encounter at the BNP Paribas Open tennis tournament in Indian Wells, CA—a moment that would reframe his keynote message.
For 10 years, he’d been greeted at the tournament by the same woman, a graceful, 75-year-old fan of the sport— and of his story. This time, she handed him a small angel figurine.
“You’ve been an angel for so many people,” she said. “I think you need one to look after you.”
That quiet act of recognition cracked something open. “It touched me,” Kiani said. “But it also made me think. Maybe I’ve had angels watching over me this whole time.”
The more he reflected, the more names came to him: mentors, early team members, fearless IP attorneys, and angel investors who bet on him when logic said not to. This angel wasn’t just a gift. It was a reminder.
“Sometimes the unintended consequences of what we do are greater than the consequences we planned. But if you keep your compass set on doing what’s right for patients, good things can happen,” says Kiani.
Irvine, CA-based Masimo’s origin story is the kind of myth-making that only happens to those who refuse to quit. Kiani started Masimo from his
“Sometimes the unintended consequences of what we do are greater than the consequences we planned. But if you keep your compass set on doing what’s right for patients, good things can happen.”
– Joe Kiani
garage with a $40,000 loan and a mission: solve the problem of pulse oximeters that failed when patients moved. It was a niche problem—but a critical one. In NICUs, faulty readings were leading to blindness in premature infants.
The solution—Masimo SET (Signal Extraction Technology)—today the industry-leading, clinically proven pulse oximetry technology that enables accurate monitoring of oxygen saturation and pulse rate of more than 200 million people a year—broke new ground. It filtered motion artifacts and saved lives.
But it ran up against a wall: group purchasing organizations (GPOs) that had locked up hospital contracts in the U.S. for a fee, shutting out innovation in favor of incumbent industry giants.
“It’s not easy to make a true difference. Our product was saving babies’ eyesight—and we still couldn’t break into U.S. hospitals because of greed,” says Kiani. So Kiani did the unthinkable. He went public. Not with an IPO—but with the truth.
In 2002, he exposed the GPO stronghold in a series in The New York Times, with multiple Pulitzer prize-winning investigative reporter Walt Bogdanich, who years ago broke the infamous story about tobacco companies adding addictive nicotine to cigarettes. Kiani then testified before Congress, triggering Senate hearings that would crack open the market for Masimo—and countless other innovators.
“Our business grew 10,000% in three years. But more importantly, the market
opened up to other innovative medical technology companies, which benefits patients,” Kiani told the LSI USA ‘25 audience.
Victory wasn’t easy. The Masimo story is marked by years of legal battles in the early 2000s—against Nellcor, Covidien, and even Apple. Kiani didn’t just protect his patents. He protected a principle: that ideas, when built to serve patients, deserve to see the light of day.
Masimo won its patent and antitrust cases and walked away with an astounding billion dollars in damages and royalties. But that wasn’t the end. Kiani did something few founders do.
Instead of buying out his early investors, as advised, he paid them dividends. He handed $35 million in bonus checks to employees as if they were shareholders. And then he went public, ensuring that everyone who believed in the mission had a stake in its future.
“The investors that believe in us are at least as important as the ideas we try to bring to life,” Kiani says. “It’s important that we give back to those people. We walked into the office with a suitcase of checks. Some people used them to buy their first homes. It was really fun to see the fruits of the labor of our whole team come together. That was a good day.”
Despite the success, there were hard lessons. Chief among them: never give up control.
After going public, Kiani found himself at odds with some of his investors—many of whom had large holdings in companies like Apple. When Masimo sued Apple and won, Apple’s stock fell 2%. Masimo’s rose 10%. The market sent a message. So did some of his shareholders.
“They killed products that helped babies. They let go hundreds of amazing people.”
Kiani’s advice? Structure your company to protect your mission. Dual-class

shares. Clear governance. Investor alignment. Because in healthcare, staying in control isn’t about ego. It’s about outcomes.
Kiani’s advice?
Structure your company to protect your mission.
Today, Kiani is channeling that same purpose into Willow Labs—a stealthmode startup located at the University of California, Irvine’s Research Park office campus, aiming to reinvent diabetes care. The goal: prevent the disease before it starts. Empower patients before they fall through the cracks.
The company’s first digital health product, Nutu (Latin for “nudge”), is designed to do exactly that—help people with prediabetes avoid the full-blown condition. Research shows that losing just 5% of body weight can reverse prediabetes. Willow wants to make that not just possible, but engaging. (Nutu, introduced last month, is an innovative health and wellness app that empowers individuals to take charge of their health thanks to its unique Nutu Score, a tool within the app designed to help nudge users into optimal balance. It also offers a Healthy Living Program and a Diabetes Prevention Program, all starting with a prediabetes risk assessment.)
Kiani offers a thought-provoking question to the LSI USA ‘25 audience: “We spend so much money at the end of our lives trying to live another year or two. What if we invested that energy earlier?”
Willow is already home to 90 employees and growing. The team is full of “purists,” many from Masimo, says Kiani—engineers, clinicians, and operators who care more about the mission than the money.
He won’t reveal what’s next in the product portfolio. But if history is any guide, it will challenge the status quo—and demand a different kind of leadership.
Kiani has built companies, fought monopolies, battled in courtrooms, and still sees himself as someone who hasn’t done enough.
“We haven’t done anything yet. The future is bright—and we’ve got to get there fast,” he says.
So what does it take to lead like Joe Kiani?
• Attract Purists: Surround yourself with people better than you— people obsessed with their craft, not their paycheck.
• Stay Principled: When the market tells you to compromise, don’t. Principles don’t just protect your mission. They protect your legacy.
• Work Like It’s Life or Death: Because in healthcare, it often is. “The only time success comes before work is in the dictionary,” he says (sage advice that is enshrined as his senior quote in his high school yearbook).
• Remember Why You Started: Kiani’s “why” was personal. His sister suffered a preventable injury as a child. He spent summers in hospital waiting rooms while she underwent surgeries, watching not just patients suffer, but families.
“That’s what got me into medical technology. That’s what made me care about patient safety. It’s not just the patient. It’s the people who love them,” he says.
“The only time success comes before work is in the dictionary.”
In a moment of quiet defiance, Kiani closed his keynote with a nod to a 17-year-old tennis champion at the recent Indian Wells tournament who had the confidence to thank herself.
“I’m going to thank myself,” he said. “For not giving up.”
The audience rose to their feet and applauded. It was more than deserved. Because in a world of quarterly reports and diluted missions, Joe Kiani is proof that you can still build something lasting by doing the right thing— over and over again.
Even if it takes a decade. Even if you have to go to court. Even if you lose sleep and friends and titles along the way.
“Don’t fall in love with the technology. Fall in love with the problem to solve. Then fight like hell to fix it,” he advised the global medtech stakeholder audience.
It’s fitting to close Kiani’s story with insightful gems from his recent post on LinkedIn:
“Healthcare innovation isn’t just about technology. It’s about caring people. It’s about ensuring every patient feels seen, heard, and cared for, no matter where they are. The real measure of success isn’t how advanced our tools are but how much in the hands of skilled and caring hands they improve lives.
Telehealth and remote monitoring are improving lives—no more long commutes to appointments or waiting weeks for a check-in. Now, patients can connect with their providers from home, track their health in real time, and get the care they need before small issues become big problems.
The result is:
• Happier patients
• Better outcomes
• Healthcare experiences that feel more personal, even when powered by technology.
The future of healthcare isn’t just about innovation—it’s about making care more human, more accessible, and more proactive. When technology enhances connection rather than replaces it, we create a system where every patient feels valued and supported. A truly patient-first approach doesn’t just treat illness—it empowers health, transforms experiences, and ensures care is always within reach.”
Joe Kiani Founder and Executive Chairman Willow Laboratories

(Source: Willow Laboratories)
Joe Kiani is committed to transforming yesterday’s impossibilities into today’s realities. This is precisely what he accomplished at Masimo and continues to achieve with Willow Laboratories, which was founded in 1998.
Kiani founded Masimo in 1989, a global medical technology company renowned for noninvasive patient monitoring technologies. Under his leadership as Chairman and CEO, Masimo transformed from a “garage start-up” into a publicly traded, multi-billion-dollar enterprise listed on NASDAQ (MASI). To name just one successful invention, Masimo Signal Extraction Technology (SET) is the primary pulse oximetry that can be found at the top 10 hospitals in the United States and throughout the world. Today, Kiani is leading Willow Laboratories as the team prepares for the commercialization of several new groundbreaking technologies.
Kiani has received numerous awards for his work in technology and patient safety. In 2024, his W1 Medical Watch was named one of TIME’s Best Inventions of 2024. In 2023, he received the Robert Wears Patient Safety Award from University of Florida Health. He has also received the Hubert H. Humphrey ‘Dawn of Life’ Award (2014), which is presented by the Newborn Foundation to individuals who have significantly impacted infant mortality rates. In addition, Kiani was honored with IP Champion Award in December 2018, the Society of Critical Care Medicine Innovation Award in 2000 and Ernst & Young’s Entrepreneur of the Year: Life Sciences Award in 2012.
Kiani received his Bachelor’s in Electrical Engineering from San Diego State University (SDSU). He also earned his Master’s in Electrical Engineering from SDSU. Kiani has received Honorary Doctorate Awards in Science from SDSU and Chapman University. He holds more than 600 patents related to patient monitoring technology, and his technology is used to monitor more than 200 million patients worldwide each year. Kiani remains tirelessly dedicated to improving the lives of patients, their safety, and the healthcare system they interact with.
This month, our featured Global Procedure Volumes data spotlights general laparoscopic surgical procedures, and open procedures for comparison.
LSI’s Global Surgical Procedure Volumes (SPV) Tracker provides trusted coverage of 300+ diagnostic and therapeutic procedures across 37 countries. Scan QR code to request a demo:


Let’s face it, we’re all tired of the pop-ups, clickbait, and ads.
That’s why we created The Lens — well-curated insights and coverage, from the analysts and industry insiders you trust.


$599 for your first year
Billed annually and includes 12 editions $699.00
Evolving over the last decade from simple step and fitness trackers to AI-powered, always-on health diagnostics, wearable monitoring devices are transforming how chronic conditions such as diabetes, and various heart and neurological diseases are managed—and even treated.


Starting over a decade ago, Fitbit (now part of Google), Apple’s Apple Watch, and Samsung’s Galaxy Watch made wearable fitness tracking mainstream. Now, the medtech industry and health monitoring-first companies have seized on the opportunity, with smart health technology including wrist-worn devices, patches, sensors, headbands, and other products.
From continuous glucose monitors (CGMs) to AI-driven cardiac patches and sleep apnea sensors used in a variety of healthcare settings, from tech and medtech giants such as Dexcom, Abbott, Medtronic, iRhythm, AliveCor, Omron, ResMed, and BrainBit, among many others including emerging innovators, cutting-edge wearable monitoring devices aren’t just for tracking. They’re now helping to diagnose, predict, guide, and even treat chronic conditions like diabetes and heart disease. Packed with AI-powered analytics and personalized, real-time insights, they’re shifting healthcare from reactive to proactive, slashing chronic disease hospitalizations, and redefining patient outcomes.
With demand for proven always-on health wearable monitoring devices for a variety of conditions surging, and clinical evidence supporting their efficacy, the future of the global wearables market is heading toward more advanced, multi-condition monitoring with deeper AI integration, making healthcare more proactive, personalized, and accessible.
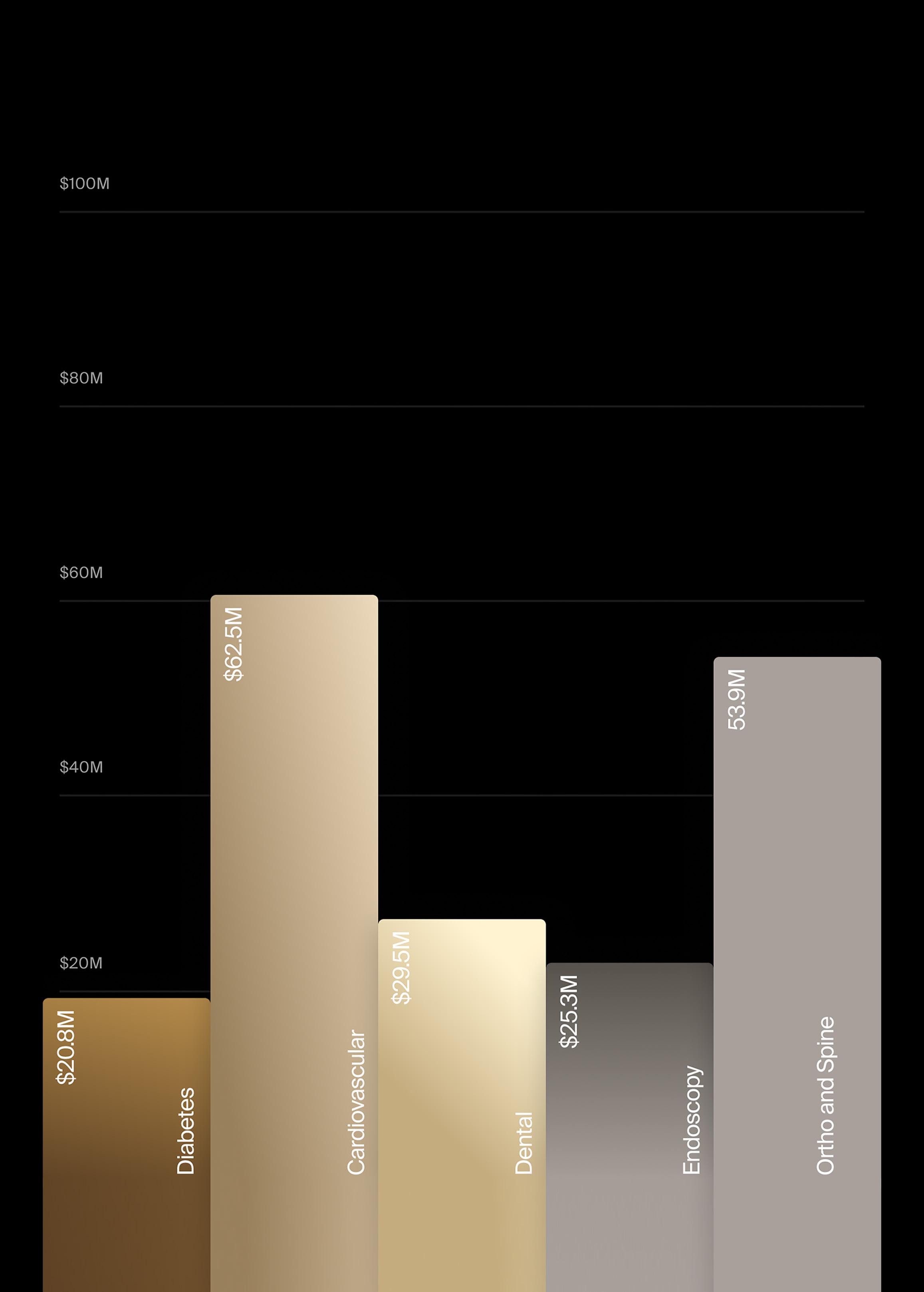
The market for wearable monitoring devices has grown continuously since its inception in 2009 with the launch of Fitbit, with a dramatic expansion in 2015 with the introduction of the Apple Watch, followed by the Samsung Galaxy Watch in 2018. The Apple Watch now accounts for about 86% of the global wearables market. These easy-to-use wearable devices have seen pervasive utilization, with some estimates indicating that over 30% of adults in the U.S. regularly use wearables (Roos & Slavich, 2023).
Total sales of wearable monitoring devices are projected to increase at a CAGR of 3.6% during the forecast period, from $17.8 billion in 2023 to $21.2 billion in 2028, according to LSI Market Intelligence (see figure on next page). Market growth is primarily driven by increases in price, as newer models featuring advanced software and sensor technologies are introduced, with growth in unit volume contributing 1.5% annually. Since their market entry, new sensors and applications have been implemented in subsequent generations of devices that increase the amount of healthcare data being captured and analyzed.
Products included within the scope of this LSI analysis include: Wearable devices for physiological monitoring of

heart rate, electrocardiograms (ECGs), oxygen saturation, blood pressure, sleep apnea, exercise/exertion, respiration, body temperature, and others.
• Continuous Glucose Monitors (CGMs): Considered by providers and patients to be essential for diabetes management, CGMs from companies such as Dexcom (Dexcom G7 ), Abbott (Freestyle Libre systems), and Medtronic (MiniMed 780G system) provide realtime glucose readings, trend analysis, and alerts to help patients maintain optimal blood sugar levels and prevent health complications. Most CGM devices are designed to help patients manage their Type 1 or 2 diabetes with fewer fingerstick checks. A sensor just under the skin measures glucose levels 24 hours a day, and a transmitter sends results to a wearable device or cell phone to allow users to track changes to their glucose level in real time. Recently, the potential market for CGMs has expanded significantly with the FDA approval of over-the-counter (OTC) CGM devices from Dexcom (the Dexcom Stelo Glucose Biosensor System) and Abbott (Lingo and Libre Rio) in 2024. These devices are marketed toward the general population who are looking to monitor their glucose levels for overall health and wellness.
• Cardiac Monitoring Wearables: Devices like ECG patches (e.g., iRhythm’s Zio, AliveCor’s Kardia 12L and other models, Hillrom/Bardy Diagnostics’
Global Wearable Monitoring Devices Market Forecast, 2023-2028
Carnation Ambulatory Monitor [CAM Patch]) and smartwatches with ECG functionality (e.g., Apple Watch and Withings ScanWatch) help detect arrhythmias, atrial fibrillation (AFib), and other cardiac irregularities to aid in reducing stroke risk and enabling early intervention.
• Wearable Blood Pressure Monitors: Continuous blood pressure tracking devices, such as those from Omron (the Omron Healthcare Heart Guide BP8000-M Blood Pressure Monitor) and Aktiia (the Aktiia bracelet, cuff, and mobile app), and cuffless technology under development, provide hypertension patients with round-the-clock insights, aiding in better disease management and medication adherence.
• Respiratory and Sleep Monitors: Devices like ResMed’s AirView-connected continuous positive airway pressure (CPAP) systems and wearable pulse oximeters help manage conditions such as chronic obstructive pulmonary disease (COPD) and sleep apnea, reducing hospital readmissions and improving quality of life.
• Neurological Wearables: Smart headbands and electroencephalogram (EEG) wearables, such as those by NeuroMetrix, BrainBit, and Epitel, assist in monitoring neurologic conditions, e.g., stroke (for potential arrhythmia capture), epilepsy, Parkinson disease, and sleep disorders. The Quell Fibromyalgia device from NeuroMetrix is the first and only FDA-approved medical device that helps reduce the symptoms of fibromyalgia.
As a tool for monitoring patients, there are many factors influencing market growth in this space. Drivers include a greater volume of data for clinical decision-making, and reduced healthcare costs.
There is an increasing body of evidence indicating that wearable technologies can reduce healthcare expenditures, through a combination of preventative care (e.g., lowering the impact of lifestyle-related risk factors), reductions in healthcare visits, and a transition from more costly monitoring alternatives. A systematic review by De Sario Velasquez et al. (2024) found that across a broad spectrum of wearable monitoring devices, the probability of these technologies being cost-effective for their respective uses ranged from 66% to 100%. The case for utilizing wearable monitoring devices is expected to increase as more studies are conducted.
Game-changing, venture-backed wearables in development, such as the “lab in a patch” from Nutromics, which uses DNA sensor technology to track multiple targets in the body such as disease biomarkers and hard-to-dose drugs, offer a glimpse into the potential for this market.
Another new innovation, Google’s Pixel Watch 3, includes a newly FDAcleared feature called Loss of Pulse Detection, which can detect impending death due to a stopped heart.
Continuous monitoring through nextgeneration wearables represents a transformation in the way patients can be cared for, which would translate to significant market growth if the technology can be actualized.
The challenge with these technologies remains in helping clinicians decipher the large volume of data to inform, data quality, user retention, and reimbursement, as well as not overwhelming the care delivery process.
Perhaps the most consistently highlighted challenge to the wearable mon-
itoring devices market is the quality of the data being collected. According to broker research by The Benchmark Company, one of the most significant barriers to increased adoption of wearable monitoring devices is the lack of consistent, high-quality data from these devices.
Like other interventions, user retention or compliance is another challenge that limits the success of this market— but it also provides avenues for innovation. Device makers and providers are both invested in ensuring that patients continue to utilize these devices to maximize their benefits. Payors recognize the benefit of wearable monitoring devices, however reimbursement considers the time and resources necessary to monitor and review this large volume of data.
Other limiters of the wearable monitoring devices market include the relatively high price for wearable devices, which limits market penetration particularly in less developed regions, and market saturation as the pool of consumers interested in personal health monitoring devices but who have not adopted the technology gradually shrinks.
With an estimated 86% market share (see figure), Apple unequivocally leads the market for wearable monitoring devices as the most popular smartwatch in the world. As of 2023, nearly 80% of iPhone users were also Apple Watch owners, according to industry research.
Of all the consumer-focused competitors, Apple has been the most aggressive in obtaining FDA approval for various sensors and monitoring capabilities used in their devices, such as the detection of arrhythmias including Afib. The company clearly has its eyes on making a consumer-focused device that can bring medical-grade data for various vital signs to physicians.
In terms of units, having reached a peak of 53.9 million units in 2022, sales of Apple Watches declined the following year. In 2023, the company reported that 38.3 million units were sold. Other major market leaders, like Google/Fitbit and Zepp Health (with the Amazfit smartwatches and wearables), have not committed nearly as aggressively towards the positioning of their devices
Notes: Figures may not sum due to rounding. The “Others” category includes AliveCor, Braebon Medical, Biobeat Technologies, BioIntelliSense, BodiMetrics, COSINUSS, Current Health, Empatica, Intelesens, LETI-CEA, Lifewatch Technologies, Medtor, Medtronic, Movano Health, Neopenda, Nuubo, Oura Health, PCL Health, Preventice Solutions, Qardio, Sensium Healthcare, SHL Telemedicine, Sibel Health, Spire Health, VitalConnect Masimo, VitalTracer, Vitis, Vivalink, Waire Health, Wellue, WHOOP Labs, Withings, and Zoll, among others.
Sources: Annual company reports, SEC filings, investor relations materials, and LSI’s Wearable Monitoring Devices Market Snapshot, published March 2025
as medical-grade wearables. Like Apple and its Apple Watch, both companies have reported declining sales of their devices.
Given its accessibility, connectivity to the wider Apple products ecosystem, and focus on implementing additional healthcare-focused features, Apple and its Apple Watch platform is the most dominant wearable monitoring device The market leader has received FDA approvals for several health-related features on its devices, including the Apple Watch’s AFib history feature, irregular rhythm notification feature, the ECG app, and sleep apnea detection. A new model, the Apple Watch Ultra 3, with enhanced features like hypertension detection, blood pressure tracking, and satellite connectivity, is potentially planned for introduction in September 2025.
As this market has become saturated with competitors and mature from a unit volume growth perspective, LSI projects that clinical-grade devices are driving underlying market growth in terms of unit and dollar volumes. There are several reasons for this, including clinical-grade devices commanding a higher average selling price and being approved for monitoring and managing some of the most burdensome types of chronic disease (e.g., cardiovascular disease and diabetes).
While the overall market, which includes consumer- and clinical-grade devices, is projected to expand at a slow rate, the dollar volume of the medical-grade devices market is projected to increase rapidly over the next five years as healthcare providers and payers seek to implement more efficient ways to manage patients.
Market leaders remain focused on incorporating more advanced health monitoring features into consumer-based devices to transform them into clinical-grade wearables and maintain market share. The most targeted features are for continuous electrocardiogram (ECG) monitoring and blood glucose monitoring. The latter is far more challenging to implement without a more invasive sensor (e.g., microneedles). However, the implementation of diabetes monitoring and therapeutic capabilities would result in being able to service approximately 10% of the global adult population.
Attracted to a large and growing market for wearable monitoring devices and the potential to impact health outcomes for a variety of chronic diseases in novel ways, many medtech and healthtech start-ups are innovating in this space. A few of our promising LSI alumni presenters are highlighted below.
Glucomodicum, founded in 2018 as a spin-out of the University of Helsinki in Finland, is transforming glucose

Jokke Maki (Source: LSI Europe ‘24) monitoring with precise, needle-free magnetohydrodynamic technology. The company’s new CGM sensor, the Talisman, measures glucose molecules diffused through the skin. By integrating ultra-sensitive and low-cost biosensors, Talisman enables continuous, painless, and accurate monitoring of glucose levels. Review Co-Founder and Medical Director Jokke Maki’s LSI Europe ‘24 presentation on the LSI website (Resources > Video Library).
Goleta, CA-based Laxmi Therapeutic Devices was founded in 2016 to improve the quality of life for people living with diabetes. The company

Sumita Pennathur (Source: LSI USA ‘24) is developing a wearable diagnostic device that supports people living with Type I diabetes with real-time, accurate measurements. Review Co-Founder
and CEO Director Sumita Pennathur’s LSI USA ‘24 presentation on the LSI website (Resources > Video Library).
Fisher Wallace Labs is a developer of wearable brain stimulation technologies for the treatment of depression, anxiety, insomnia, and other mental health conditions. Its OAK device uses

Kelly Roman (Source: LSI USA ‘24)
Transcranial Alternating Current Stimulation (tACS), which modulates brain function and cognitive processes by entraining neural oscillations and inducing long-term synaptic plasticity. Review Co-Founder, CEO and Director Kelly Roman’s LSI USA ‘24 presentation on the LSI website (Resources > Video Library).
Onera Health is developing diagnostic and monitoring solutions to help people with sleep-related ailments. The company’s Onera STS wearable sleep system collects comprehensive sleep data which allows providers to optimize patient care and reduce costs. Review Founder and CEO Ruben de Francisco’s LSI Europe ‘22 presentation on the LSI website (Resources > Video Library). (Also see Power Moves and LSI Alumni Milestones, The Lens, February 2025.)
CARI Health is developing a remote medication monitor that will enable clinicians to view medication levels in realtime and customize dosages for every patient. The pain-free, small, wireless sensors are worn on the patient’s skin and continuously send data about medication levels to the CARI Cloud. If medication levels fall outside of the

Partik Schmidle (Source: LSI USA ‘24) desired range, then alerts may be sent to clinicians, patients, and family members. Review CEO Partik Schmidle’s LSI USA ‘24 presentation on the LSI website (Resources > Video Library).
CardiaCare is developing a first-ofits-kind wearable monitoring device that can also provide treatment. Its digital neuromodulation device is designed

Amir Soltanianzadeh (Source: LSI Europe ‘24) for patients with atrial fibrillation (AF), that combines proprietary, closed-loop neurostimulation of the median nerve with AI-enabled cardiac monitoring. The app that accompanies the device transforms AF management by providing therapy management, real-time ECG tracking and arrhythmia alerts, continuous vital sign monitoring, and personalized insights into symptoms and triggers. Given AF’s multifactorial nature—often influenced by medical conditions, medications, alcohol, caffeine, sleep apnea, stress, and genetic predisposition—this comprehensive platform for understanding and managing each AF patient with a personalized approach is critical.
The company has recently completed a pilot feasibility trial and is now raising a Series A financing to fund pivotal trials for two indications for its technology, paroxysmal AF and post-ablation, with targets to achieve FDA clearances in 2027. Review CEO Amir
Soltanianzadeh’s LSI Europe ‘24 presentation on the LSI website (Resources > Video Library). (Read more about Soltanianzadeh’s story and the company’s technology in “LSI Alumni Innovator Spotlight: CardiaCare CEO Amir Soltanianzadeh,” The Lens, February 2025.)
Pulsify Medical is developing a wearable patch, the Pulsify Patch, that utilizes ultrasound to continuously monitor cardiac output and performance. The flexible patch can be worn on the chest for hours or days, and is the first device to continuously and directly monitor cardiac performance, employing an ultrasound sensor. The company is a spinoff from both imec and the Catholic University of Leuven’s Medical Imaging Center. Review the company’s LSI USA ‘23 presentation on the LSI website (Resources > Video Library).
San Jose, CA-based VitalConnect, a leader in biosensor technology, offers the only cardiac monitoring technology, VitalPatch RTM, that sends all

Nersi Nazari (Source: LSI USA ‘22)
patient data instantaneously to a secure cloud, enabling monitoring for 21 different cardiac arrhythmias and real-time monitoring for four important patient vitals. The flexible, water-resistant, fully disposable VitalPatch RTM requires no assembly or charging, and allows patients to go about their normal daily routine, optimizing compliance over the monitoring period.
In February, the company closed $100 million in financing, through a combination of equity and debt capital. The equity financing was led by new investor Ally Bridge Group, with participation from existing investors including EW Healthcare Partners, MVM and Revelation Partners, while Trinity Capital provided the debt financing. Review
Founder and Executive Chairman Nersi Nazari’s LSI USA ‘22 presentation on the LSI website (Resources > Video Library).

Washington, DC-based Otolith Labs is developing prescription medical devices for the treatment of vestibular disorders, which impact millions of Americans. Otolith’s patented non-invasive head-worn technology, which received Breakthrough Device Designation from the FDA for the treatment of chronic vertigo, uses precisely tuned vibrations to interact with the acceleration sensors in the inner ear, mitigating the symptoms of vertigo.
Otolith is conducting clinical trials to evaluate its technology as an immediate symptom-relief treatment for chronic vertigo associated with vestibular disorders. The company recently announced results from an exploratory user study investigating the effect of vestibular stimulation on cybersickness from virtual reality (VR) experiences. The study demonstrated that subjects utilizing the Otolith technology had improved virtual environment experiences with lower reported nausea and longer VR durations. Review Founder & CEO Sam Owen’s LSI USA ‘23 presentation on the LSI website (Resources > Video Library).
Data in this article is partially sourced from the LSI Market Intelligence Platform, including the Wearable Monitoring Devices Market Snapshot, published March 2025, and the extensive LSI event Video Library. Access LSI’s continuously updated Market Intelligence Platform.

Scan QR code for details and to request a demo.
De-risk your biggest strategic decisions with our most comprehensive market data and projections (worldwide sales, share by supplier, CAGRs) on 27 major market segments and 200+ sub-segments, prepared with consistent methodologies and forecast periods.

Celebrating the accomplishments of a few of the many bright stars in our LSI presenting company and speaker alumni community.
Be sure and check out presentations from these and many other innovative companies from our recent LSI events on our website, at Resources > Video Library.



Accelus, a leader in expandable spinal implant technology, has announced that its FlareHawk Interbody Fusion System has been granted regulatory approval by the Brazilian Health Regulatory Agency (ANVISA).
The FlareHawk Interbody Fusion System, with more than 22,000 devices implanted in 24 countries worldwide, represents a significant advancement in spinal interbody fusion. It utilizes Accelus’ proprietary Adaptive Geometry technology to expand in width, height, and lordosis after insertion with a minimal profile. FlareHawk’s multi-
directional expansion is designed to reduce subsidence, restore foraminal height and reestablish sagittal balance from a posterior approach.

Milan, Italy-based cardiovascular start-up Approxima Srl announced that it has secured €7.3M ($7.9 million) in the first closing of its Series A financing round, to advance its percutaneous right ventricular reshaping technology for tricuspid valve regurgitation (TR). The round was co-lead by ENEA Tech e Biomedical and Panakès Partners, with strong continued support from current shareholders Club degli Investitori
through Simon Fiduciaria and other business angels. The round remains open for subscription from other co-investors in the coming months.
Approxima’s technology is being developed to offer a transformative approach to treating patients with TR by addressing its underlying cause, ventricular dilation, restoring thus the tricuspid valve function under a straightforward and efficient percutaneous implantation procedure. Its impact extends beyond the valve itself, improving also the right ventricle performance while maintaining all bailout options feasible, including native valve repair or valve replacement. The device is intended to be implanted into the heart via a totally percutaneous and straightforward beating-heart procedure. The financing will support preclinical verification, first-in-human trials, and a U.S. early feasibility study.
Arcuro Medical, a portfolio company of The Trendlines Group, has received FDA 510(k) clearance for its

SuperBall-RC system for rotator cuff indications. The SuperBall-RC system is based on the company’s SuperBall technology, successfully used in over 5,000 meniscus repairs, and is designed to enable safe, easy fixation of rotator cuff augmentation grafts.
Current rotator cuff re-tear rates following surgery can range from 20-40% in patients over 50, according to Arcuro. Rotator cuff augmentation with biologic or biosynthetic grafts can mitigate this risk but can be technically demanding with current surgical techniques.
The SuperBall-RC will enter a limited user release in the second quarter of 2025, in anticipation of a full launch in the second half of the year, ahead of schedule due to the timely FDA clearance.

Waltham, MA-based Arsenal Medical, a clinical-stage company developing medical devices from innovative biomaterials, recently announced the complete enrollment of an initial cohort of 10 patients in its EMBO-02 clinical study of NeoCast to treat chronic subdural hematomas (cSDH), across three sites in Australia.
NeoCast is a next-generation, nonadhesive, solvent-free, liquid embolic material designed for deep distal penetration. EMBO-02 is an open-label, multi-center, prospective clinical trial to
evaluate the early safety and feasibility of NeoCast to embolize target vessels of the subject’s middle meningeal artery (MMA) to treat cSDH. All MMA embolization (MMAe) procedures in the trial were successfully completed with NeoCast, and all patients passed the 30-day safety endpoint with no device-related serious adverse events. On average, investigators were “very satisfied” with NeoCast’s acute performance and usability, highlighting the material’s ease of use. (Also see Power Moves, this issue; and LSI Alumni Milestones, The Lens, December 2024.)
cSDH is the persistent bleeding of an injured vessel in the membrane that protects the brain (dura). It is typically caused by a fall or injury to the head, including minor head trauma, and can result in significant physical and cognitive decline. It is the most common neurosurgical condition in those 65 and older and has been categorized as a sentinel health event in the elderly.

Berlin Heals Holding AG, a Swiss late-stage medtech company, announced that it has secured over CHF 7M ($7.8 million) in funding to support first-in-human studies for less-invasive implant approaches of its electronic C-MIC (Cardiac Microcurrent) device, for the treatment of heart failure. The C-MIC is a small implantable device that propagates reverse remodeling of the cardiac muscle tissue by delivering a constant but minimal electrical direct current (DC) to the heart.
The funds will also advance CE submission for the first-generation device and prepare for a pivotal trial to achieve global regulatory and reimbursement approvals. Berlin Heals was incorporated in 2014 with the mission to establish a novel therapeutic approach to not only delay the progression of heart failure disease, but to virtually cure it.

Brainomix, a privately held pioneer in stroke AI imaging software launched as a spin-out from the University of Oxford, has announced a strategic partnership with medtech leader Medtronic Neurovascular. The collaboration aims to enhance stroke care for patients across Western Europe by integrating cutting-edge AI solutions into clinical practice, expanding access to life-changing treatments and improving patient outcomes.
By combining Brainomix’s AIpowered imaging technology with Medtronic’s expertise in neurovascular treatments, the partnership represents a significant step forward in stroke care. The collaboration will support faster, more accurate diagnoses, enabling healthcare professionals across the region to make better-informed decisions and deliver life-saving interventions more efficiently.
The Brainomix 360 Stroke platform is powered by state-of-the-art AI algorithms that provide real-time interpretation of brain scans to aid treatment and transfer decisions for stroke patients. Brainomix 360 Stroke was studied in the largest real-world evaluation of stroke AI—involving data from more than 450,000 patients across England over a 5-year period—with results showing a doubling of thrombectomy rates in Brainomix sites, and an additional 37% increase compared with non-evaluation sites. Brainomix is a private company backed by leading healthtech investors, including Boehringer Ingelheim Venture Fund, Parkwalk, and Tencent Holdings
In March, Brainomix announced the completion of a £14 million ($18 million) Series C investment round. The round was co-led by existing investors Parkwalk Advisors, the UK’s largest growth EIS fund manager, and the Boehringer
Ingelheim Venture Fund, along with new investor Hostplus via the IP Group Hostplus Innovation Fund. Hostplus is a leading Australian industry superannuation (pension) fund with over AUD$120 billion in funds under management. The round was supported by LifeSci Capital.
Cagent Vascular, the exclusive developer of serration technology for vessel dilation in endovascular interventions, recently announced the launch of its newest product, the Serranator SL-PRO PTA Serration Balloon Catheter. Serranator SL-PRO is purpose-built for pedal intervention as well as challenging-to-cross tibial disease. Serranator SL-PRO utilizes Cagent’s proven Serration Technology on a redesigned platform offering improved deliverability and trackability, while offering Serranator’s proven 1,000 times increase in point force versus plain balloon. Its innovative mechanism of action allows for predictable and controlled lumen gain with minimal dissection.
The company also secured $15 million in growth capital funding from Trinity Capital to expand commercialization efforts, supporting the continued rollout of the company’s Serranator angioplasty balloon. This capital augments $45 million of equity financing led by US Venture Partners in 2024.

Copenhagen, Denmark-based CathVision, a medtech company developing advanced electrophysiology solutions, has received CE Mark for its breakthrough product, ECGenius System. This regulatory milestone allows CathVision to market and distribute ECGenius System across the European Economic Area (EEA), bringing state-of-the-art
electrophysiology solutions to healthcare providers and patients.
ECGenius System is a cuttingedge medical device designed to provide accurate and real-time electrogram recording. The device leverages advanced signal processing and AI-driven analytics that empower clinicians with actionable insights. The product is FDA 510(k) cleared.
CathVision’s mission is to empower physicians to make informed clinical decisions by providing precise cardiac electrical signals that aid in diagnosing, characterizing, and treating heart rhythm disorders. Founded in 2013, CathVision has a U.S. office in Minnesota.

Boston, MA-based CereVasc, a clinical-stage device company developing novel treatments for neurological diseases, reported initial results of its pilot clinical study of the eShunt System in the treatment of elderly patients with normal pressure hydrocephalus (NPH). CereVasc reported the study results in 30 elderly patients treated with its eShunt System in the U.S., in which the Primary Safety and Primary Efficacy Endpoints were both achieved.
The company’s FDA-approved Investigational Device Exemption (IDE) study is a prospective, non-randomized, open-label, multi-center study being conducted at 11 academic medical centers in the U.S. in subjects with NPH for whom a traditional cerebrospinal fluid (CSF) shunt implant is indicated. The objective of the study is to evaluate the safety and efficacy of the eShunt System procedure. No serious adverse events were reported in the first 90 days of the study. Published data on conventional open surgical shunt procedures for NPH have documented a 25.15% rate of complications or hospital
readmission within 30 days of ventricular shunting.
Also in February, the company received approval from Argentina’s National Administration of Drugs, Food and Medical Devices (ANMAT) to initiate the STRIDE trial, a clinical study evaluating CereVasc’s eShunt System as a treatment for normal pressure hydrocephalus (NPH). The trial will evaluate the safety and effectiveness of the eShunt System versus the current standard of care, the ventriculo-peritoneal (VP) shunt, in draining accumulated cerebrospinal fluid from the brain in elderly patients. The results of the STRIDE trial will serve as the basis for CereVasc’s anticipated submission to regulatory agencies for approval to market the eShunt System.
The Hydrocephalus Association estimates that 800,000 older Americans may be living with NPH, and it is estimated that more than 80% of cases remain unrecognized or untreated. Without appropriate diagnostic testing, NPH is often misdiagnosed as Alzheimer’s disease or Parkinson’s disease, the result of a stroke, or other neurodegenerative conditions. NPH is, however, one of the few treatable forms of dementia.

Menlo Park, CA-based Endovascular Engineering (E2) announced that it has secured $42 million in an oversubscribed Series B financing to advance its next generation clot removal technology platform for venous thromboembolism (VTE). The round was co-led by 415 Capital and S3 Ventures, with participation of Panakès Partners, M&L Healthcare, and strong support from founding investor Santé Ventures. Two global strategic investors made substantial commitments alongside
Cordis, which continues its strategic partnership with E2.
The capital injection accelerates E2’s mission to transform VTE treatment, placing an initial focus on advancing novel solutions for addressing pulmonary embolism (PE). As the company approaches completing enrollment in its ENGULF pivotal trial to evaluate the Hēlo PE Thrombectomy System, the additional capital strengthens E2’s position to address critical unmet needs in interventional medicine while pursuing a new standard of care for both patients and physicians.

Exero Medical, a U.S.- and Israel-based company focused on early detection of post-operative complications using tissue healing data, has secured up to $12.7 million in funding to complete its FDA trial and launch its breakthrough-designated xBar system in the United States. The round includes $10 million in upfront funding and additional milestone-based warrants, as well as $2.7 million in non-dilutive grants. The round was led by Longevity Venture Partners, Tech Council Ventures, and Edge Medical Ventures. The funding will be used to support clinical and commercial activities.
Mortality rates in patients with severe complications after gastrointestinal surgery can be up to 20% due to challenges in identifying these complications early. The xBar system uses sensors placed at the surgical site to collect data that can assist clinicians in early detection of major, life threatening complications after gastrointestinal surgery. Exero Medical was founded in 2018 by MEDX Xelerator
Gyder Surgical, a medtech company specializing in intuitive, orthopedic


navigation solutions, recently announced 510(k) FDA clearance of its GYDER Hip System, the world’s first commercially available non-invasive (pin-less) and image-less solution for the accurate positioning of the acetabular cup during anterior hip arthroplasty. The FDA’s clearance decision is the second significant regulatory milestone for the GYDER Hip System, which received Australia’s TGA regulatory approval previously. Surgical cases have already been successfully performed in Australia and India.
Improving acetabular cup placement accuracy is clinically important with significant potential to reduce post-surgery complications, thereby improving patient outcomes and lowering health system cost burden. An estimated 700,000 hip procedures are performed annually in the U.S., with growing demand driven by aging demographics and more active lifestyles. Unlike traditional hip navigation approaches, the GYDER Hip System’s patented technology does not use invasive metallic pins. Developed by Gyder Surgical, the innovation also does not rely on pre-operative or intra-operative imaging for anatomical landmark registration. One-minute calibration and quick registration make the system a fast, easy-to-use computer-assisted navigation solution that seamlessly integrates into existing surgical workflows. The company is based in Mulgrave, Australia and Palo Alto, CA.
Ibex Medical Analytics, a Boston, MA-based leader in AI-powered cancer diagnostics, recently announced that it has received FDA 510(k) clearance for Ibex Prostate Detect, an in vitro diagnostic medical device that harnesses AI to generate heatmaps identifying small and rare missed prostatic cancers, acting as a safety net that assists
pathologists in ensuring patients receive an accurate diagnosis.
Ibex Prostate Detect is a software only device that analyzes scanned histopathology whole slide images from prostate core needle biopsies prepared from hematoxylin & eosin (H&E) stained formalin-fixed paraffin embedded tissue. The device is intended to identify tumors that may have been missed by the pathologist. If tissue suspicious for prostate cancer is identified, the system provides case- and slide-level alerts, and includes a heatmap directing the pathologist to areas likely to contain cancer.
In March, Royal Philips, a global leader in health technology, announced the expansion of its partnership with Ibex and a new release of Philips IntelliSite Pathology Solution (PIPS) to further accelerate the adoption of AI-enabled digital pathology, that addresses the pressure pathologists face because of the global shortage of pathologists and the growing number of cancer patients.

Lymphatica Medtech recently announced the publication of its latest research, “Advancing women’s health with a pioneering implant to treat breast cancer-related lymphedema,” in the European Journal of Internal Medicine
Lymphedema is a chronic condition resulting in a local swelling, often of one arm or one leg, caused by the build-up of lymph under the skin. The condition, as well as the limb swelling, is permanent. On top of the swelling, it causes pain, recurrent skin infections and impaired movements, in many cases resulting in a state of disability for the patient. The company’s product, LymphoDrain, is a permanent implant designed to recirculate lymph inside the body to restore physiological fluid equilibrium.
The study represents a significant milestone as a first-in-human investigation of LymphoDrain, providing crucial first insights into its safety and effectiveness. Lymphatica Medtech is a spin-off company out of the Angiology Division of the Lausanne University Hospital and of the Laboratory of Lymphatic and Cancer Bioengineering of the Swiss Federal Institute of Technology in Lausanne, Switzerland.

Barcelona, Spain-based Neuroelectrics, a global leader in noninvasive brain stimulation and monitoring technologies, has launched its advanced 24-hour EEG recording system, marking a significant step forward in the diagnosis and management of neurological disorders, particularly epilepsy. The system enables continuous, longterm brain activity monitoring, offering deeper clinical insights beyond the capabilities of traditional EEGs—all while seamlessly integrating into patients’ daily lives.
EEG has long been a cornerstone in diagnosing and managing neurological conditions. Unlike standard EEG sessions, which typically last 20 to 40 minutes, long-term EEG allows neurologists to detect sporadic or infrequent events that might otherwise go unnoticed,
offering deeper insights into a patient’s brain function. Neuroelectrics’ products are being used worldwide for both neuroscience and clinical research in hundreds of universities and academic medical centers. The company’s vision is to create a powerful neurotechnology platform upon which verticals can be developed to improve the lives of as many people as possible.

Retia Medical, a White Plains, NY-based innovator in AI algorithms for patient monitoring and management, announced that it has entered into an agreement with Medtronic to distribute the Argos Cardiac Output Monitor designed to provide healthcare professionals with highly accurate hemodynamic data to support the treatment of high-risk surgical and critically ill patients.
In the operating room and intensive care unit, circulatory shock, often diagnosed as hypotension, is a primary cause of complications and requires intervention within one to two minutes to prevent damage to vital organs. Hemodynamic monitoring detects important changes in a patient’s hemodynamic status, and enables clinicians to intervene earlier, ensuring sufficient oxygen delivery and preserving end organ function. In addition, patients had one- to two-day shorter stays in the ICU when preemptive hemodynamic intervention was used.
The Argos Cardiac Output Monitor offers a unique advanced algorithm called Multi-Beat Analysis (MBA), that was developed at the Massachusetts Institute of Technology (MIT) and Michigan State University. Unlike other monitors that analyze only one beat at a time, the MBA algorithm analyzes multiple heartbeats from the blood pressure signal to model a patient’s
vascular resistance directly. Thus it provides accurate hemodynamic data on 10 comprehensive parameters even during changes in vasomotor tone, low cardiac output, and arrhythmia.

Cambridge, MA-based Sinaptica Therapeutics, a clinical-stage company leading the development of a new class of personalized neuromodulation therapeutics to treat Alzheimer’s and other primary neurodegenerative diseases, recently announced that its second-generation dual stimulation technology, the SinaptiStim System, is now being tested in a newly launched phase 2 study. The trial has enrolled its first patients and will enroll up to 60 with early-stage Alzheimer’s into this randomized, double-blind study, at multiple centers in Italy.
The second-generation technology builds on the company’s original patented, personalized and precision-delivered rTMS therapy, and adds simultaneously delivered transcranial alternating current stimulation. The idea to combine the two modalities was based on over a decade of foundational research conducted by Sinaptica’s scientific co-founders into the mechanisms of memory and methods of potentiating neuroplasticity in the Default Mode Network, including the hippocampus.
The SinaptiStim System is an investigational new approach to treating Alzheimer’s disease using non-invasive personalized precision neuromodulation. Calibrated to each individual’s brain, the therapy is delivered in weekly sessions in a recliner ranging from 20 minutes with the first-generation system to six minutes with the second-generation system. The technology was granted Breakthrough Device Designation by the FDA in 2022.



Solaris Endovascular has announced an enrollment milestone in its DEScover clinical trial, assessing a sirolimus-eluting covered stent graft—Solaris DE—for the treatment of patients facing challenges with dialysis access dysfunction and peripheral arterial disease (PAD).
In a press release, the company announced that trial enrollment is ahead of schedule and has successfully enrolled 42 patients out of the planned 120 patients to date.
The trial’s objective is to evaluate the safety and efficacy of the Solaris drug-eluting endovascular stent-graft in patients with arteriovenous fistulae (AVF) and arteriovenous grafts (AVG). A total of 80 participants with AVF are randomized in a 1:1 ratio to receive either the Solaris DE device or standard balloon angioplasty, with a single-arm design for AVG patients (n=40) treated with Solaris DE. The trial’s safety endpoints include the proportion of patients free from serious adverse events at 30 days, and efficacy will be determined by target lesion primary patency at six months.
With healthcare costs for end-stage kidney disease surpassing $59 billion in 2020, effective dialysis access management is more critical than ever. Vascular access failures, particularly restenosis, remain unresolved. Solaris DE seeks to address this unmet clinical need by combining a mechanical barrier of the next-gen impermeable electrospinning PTFE with a biological barrier to block cell proliferation.
Strados Labs, a medtech company focused on improving respiratory health, is proud to announce a new collaboration with Beth Israel Deaconess
Medical Center (BIDMC). The upcoming study will evaluate the use of Strados Labs’ RESP Biosensor, a wearable stethoscope technology for the early detection of pulmonary edema in critically ill patients on mechanical ventilation (MV). This pilot study will enroll 20 patients with an anticipated duration of 12 months.
This single-center, prospective, observational study will evaluate whether abnormal lung sounds captured by the RESP Biosensor precede currently observed markers and signs of pulmonary edema—a common and life-threatening complication in critically ill patients. The device’s performance will be compared to existing monitoring methods, including daily chest X-rays, intermittent clinical assessments, and fluid intake and output tracking.
The RESP Biosensor, an FDA 510(k)cleared, CE-marked wearable device, is designed to continuously capture lung sounds such as cough, wheeze, and crackles associated with worsening respiratory health. The technology has been used in several patient populations and disease states including chronic cough, COPD, asthma, idiopathic pulmonary fibrosis, and heart failure. This study will be the first time the RESP Biosensor will be used to monitor patients on MV to detect pulmonary edema.
SurgiBox, a Cambridge, MA-based medical device startup that aims to revolutionize mobile surgery with its flagship SurgiField product that ensures safe and sterile conditions anywhere in the world, recently announced it has received two U.S. patents for its core technology. The first involves an innovative airflow technology for clean surgery and is a utility patent, while the second is a design patent on its userfriendly, single-button, automated Smart
Control Module technology. SurgiBox’s SurgiField product combines an innovative inflatable sterile drape with an automated, battery-powered, ultraportable HEPA air filtration system. The system is protected by patents in over 20 countries.
Committed to saving lives, SurgiBox empowers medical professionals by providing cutting-edge, accessible surgical environments in even the most challenging locations

Canadian medtech startup Vena Medical has been granted Breakthrough Device designation from FDA for its MicroAngioscope, marking what the company describes as a “historic leap forward” in neurovascular care and stroke treatment.
For the first time ever, Vena claims, direct intracranial angioscopy is now possible—with real-time, high-definition imaging inside brain blood vessels provided before, during, and after neurointerventional procedures.
Traditional imaging methods provide only indirect views, often leaving clinicians without immediate feedback during critical procedures, according to Vena. In contrast, the MicroAngioscope has the potential to offer direct visualization via high-definition imaging of lesions, vessel walls and devices during neurointerventional procedures,
enhancing stroke diagnosis; optimized decision-making, with immediate confirmation of diagnoses and treatment success, reducing procedural risks and potentially improving long-term patient outcomes; and real-time feedback, boasting the ability to identify thrombus, endothelialization and proper wall apposition for intracranial stents, arming physicians with greater precision during stroke interventions.

Vesteck, a clinical-stage company based in West Chester, PA, recently announced that it won the Transformative Endovascular Decisions (TED) 2025 Ideas and Innovations Competition, held in February in Scottsdale, AZ, with its Suture-Tight endovascular catheter. The catheter delivers preloaded sutures, ensuring secure attachment of endovascular aortic repair grafts (EVAR/ TEVAR) to the aorta during the initial implant or repair procedure.
Vesteck is dedicated to revolutionizing the aortic repair space through the development of an advanced endosuturing platform. The goal of the Suture-Tight is to eliminate graft migration and endoleaks that lead to complex and expensive repair procedures. In alignment with Vesteck’s physician-centric approach, the Suture-Tight Delivery System and Suture are designed to fit seamlessly into existing procedural workflows.
With promising clinical results from 15 preclinical studies, the company is currently in a design freeze and is ready to move forward with a 100-patient 510(k) clinical trial with plans to raise $16 million to fund the study in a Series B funding round. (Also see LSI’s The Memo on LinkedIn, September 14, 2024.)
Hoag Hospital in Southern California

has become the first in the Western U.S. to offer Virtual Incision’s MIRA Surgical System, the world’s first miniaturized robotic-assisted surgery (miniRAS) device. Its small, sleek form factor is designed to offer the benefits of RAS during colectomy procedures without the logistical inefficiencies of traditional mainframe robotics. The easily accessible device, FDA-cleared in February 2024, weighs approximately two pounds (less than one kg) and offers internal triangulation with shoulders, arms, and infinite wrist roll inside of the body. It can be used in any operating room—a dedicated mainframe room is unnecessary. With its drape- and dock-free design and portability, MIRA is quick to set up, clean up, and move between cases.
The MIRA Surgical System was partially funded by Hoag Innovators, a group of community leaders dedicated to catalyzing innovation at Hoag through the power of collective giving. MIRA’s broader commercialization and expansion of its roadmap for future indications includes gynecology, general surgery, urology and other soft tissue and solid organ surgery.
Minneapolis, MN-based Visura Technologies, a privately held device company dedicated to delivering state-of-the-art visualization solutions to improve the safety and success of transesophageal echocardiogram (TEE) probe intubations, announced the initial release of case-level data collected from over 50 physicians across 15 US sites using TEECAD to assist TEE probe placement. The findings demonstrate that the TEECAD system was able to successfully visualize and navigate the esophageal inlet in the vast majority of cases of TEE intubation.
The use of 3D TEE represents a significant advance in ultrasound imaging guidance and is the mainstay of imaging
in catheter-based structural heart interventions given its ability to deliver high-quality and real-time images without the need for additional x-ray exposure. TEE probe placement failure associated with difficult probe placement has been reported to range from 0.18% to 1.9% including catastrophic complications of pharyngeal and esophageal perforation. The TEECAD system travels with the probe to provide a real-time, dynamic image of the oropharyngeal and esophageal anatomy to support safe and efficient TEE probe placement.

Santa Fe, NM-based Vitazi.ai, a leader in oculomics, recently announced an agreement with Eye Associates of New Mexico, the largest ophthalmology organization in the state. Eye Associates sees between 1,400 and 1,500 patients daily at its 11 clinics throughout the state. In addition to these sites, Eye Associates has three surgery centers and 10 optical shops. This collaboration marks a significant step in harnessing artificial intelligence to revolutionize retinal diagnostics and early disease detection.
Oculomics, the study of the retina using AI, has emerged as a groundbreaking field with the potential to detect systemic diseases—including cardiovascular disease, neurodegenerative disorders, and diabetes—years before clinical symptoms appear. By leveraging this partnership, Vitazi.ai Chief Technology Officer, Dr. Jeffrey Wigdahl, PhD, and his team of retinal researchers will research and develop new Oculomics tools on OculoInsights, Vitazi.ai’s revolutionary AI-powered retinal diagnostic and early disease detection platform, designed to empower retinal specialists and healthcare providers with advanced screening capabilities.
The industry’s most comprehensive and trusted global procedure database, with coverage of 300+ diagnostic and therapeutic procedures across 37 countries.


1MED
Action
Aktiia
Anges
Lymphatica
Medtronic ........... 9, 10, 11, 18, 30, 31, 40
Medtronic

March 16th - 20th, 2026
Waldorf Astoria, Monarch Beach, Dana Point, CA


