Atul Butte, MD
Celebrating the Inspirational Life of a Pioneer in Data-Driven Healthcare Innovation


Cardiac Ablation Devices, Alumni Innovator Spotlights, Power Moves, and much more...

Celebrating the Inspirational Life of a Pioneer in Data-Driven Healthcare Innovation


Cardiac Ablation Devices, Alumni Innovator Spotlights, Power Moves, and much more...
February 2025
July 2025

September 7th - 11th, 2025
JW Marriott Grosvenor House, London
Now in its fourth year, LSI Europe has become a staple of the global Medtech and Healthtech community. The 2025 event in London will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Registration and applications to present are now open for LSI Europe ‘25. Get in touch to learn more about the event.



Scott Pantel
Chief Executive Officer, Editor-in-Chief

Rebekah Murrietta
Vice President of Media, Contributing Author

Benny Tomlin Contributing Photographer

Henry Peck
Chief Business Officer, Editor-in-Chief

Blake Matrone Sr. Marketing Manager, Contributing Author

Kristin Karkoska Contributing Photographer

Tracy Schaaf
Managing Editor and Content Strategist, Lead Author

Gavin Kennedy Business Development, Contributing Author

Paul Streeto Graphic Designer

Brenna Hopkins Sr. Manager of Content Programs, Lead Author

Almonte Customer Service & Fulfillment

Nicholas Talamantes
Sr. Director of Market Intelligence, Contributing Author

Kelly Williams
Subscriptions & Enterprise Sales
714 847 3540 tel/fax email: info@ls-intel.com
Vol. 2, No. 7
July 2025
Created by LSI, The Lens leverages LSI’s deep industry relationships and proprietary market intelligence to help executives like you build lasting medtech companies. Start your Individual, Group or Enterprise-Wide subscription today!

Scan QR Code to activate your subscription.
The Lens is published monthly and available in print and electronic formats. Copyright 2025 by Life Science Intelligence, Inc. All rights reserved. Editor takes care to report information from reliable sources and does not assume liability for information published.
Interested in subscribing? email to: info@ls-intel.com
Interested in advertising? email to: kelly@ls-intel.com



Dear Readers,
We are living in a moment where data, devices, and clinical decisions are converging faster than ever. Few understood that as well as Dr. Atul Butte, who unfortunately passed away on June 13, 2025, at just 55. This issue of The Lens is dedicated to his legacy.
Dr. Butte was more than a pioneer in biomedical informatics. He was a force of nature who helped move the industry toward a future where data not just supports medicine, but drives it. He challenged us to unlock the value hiding in plain sight and to act on it. He believed that open data leads to open opportunity. That mindset shaped the way he built companies, trained AI, unified health systems, and empowered others.
His message to share data, build with purpose, and scale with integrity feels especially urgent now.
In our cover story, we explore how Dr. Butte’s work laid the foundation for today’s datadriven health revolution. From launching diagnostics built on public databases to building UC Health’s vast real-world data engine, Dr. Butte both forecasted the future and built the infrastructure for it.
The smartest minds in medtech are still building on what he started, and you will see that momentum reflected throughout this issue.
In this issue, we also take a closer look at the cardiac ablation devices market. As new pulsed-field ablation technologies are reshaping how atrial fibrillation is treated, we see progress beyond just “better hardware.” Earlier intervention, shorter procedures, lower risk, and meaningful cost savings are becoming possible. The same principles of precision, efficiency, and smarter use of information that Dr. Butte championed are front and center in this space.
Throughout this issue, we highlight achievements from companies that reflect the strength of our medtech ecosystem through turbulence. Neuspera received FDA approval for the first battery-free sacral neuromodulation system. Atmo Biosciences gained clearance for its ingestible gut diagnostic capsule. Clairity made headlines with its AI platform that predicts breast cancer risk from a standard mammogram.
These are not incremental steps; these are major advances, and they are happening at scale. At our recent summits, we have seen the evidence. More startups are crossing regulatory finish lines, securing significant funding, and forming real partnerships with health systems that are ready to lead. Now, it is about execution.
The people and companies featured in this issue are solving real problems, grounded in data, moving with urgency, and setting a new standard for what medtech can accomplish. We are increasingly optimistic about the state of the industry and forever humbled to shine a light on the incredible work being done to benefit patients across the globe.
Thank you for being part of this community.
All the best,
Scott Pantel Chief Executive Officer, LSI Editor-in-Chief

Henry Peck Chief Business Officer, LSI Editor-in-Chief


Need fresh inspiration for your medtech company as we navigate Q325? We’ve gathered insights from some of our LSI alumni thought leaders to help you stay ahead.
“We have to make sure we’re serving patients [first], and then ask what else can we do alongside that. If your primary goal is data, you’re probably not going to be meaningful enough to the health system or to the patient to get the data you want in the first place.”

— Caitlin Morse, Co-Founder & CEO, BrainSpace
LSI USA ‘25 Panel: Data Quality vs. Data Quantity — Getting to Market Fast or Getting to Market Right?
“Asia now happens to be the world’s fastest-growing and second-largest market for medtech.”

— CJ Lim, Regional Director of Americas, Singapore EDB
LSI USA ‘25 Workshop: The Case for Asia — Why Should Your Business Be There?
“You don’t want to be that guy or gal that’s held on by the board when you shouldn’t be, because now your company isn’t realizing what it should.”

“You can have all the data in the world. If you don’t have the right people on the team, you’re going to struggle.”

“I think one of the reasons why they’re [China] able to scale and accelerate so quickly is that there’s just not as many barriers to getting data. Patient privacy is not really a thing over there.”

— Christopher Shen, Partner, Novo Holdings
LSI USA ‘25 Signature Series: Current Market Perspectives from Shifamed and Novo Holdings
— Andrew Morris, CCO, MY01
LSI USA ‘25 Workshop: How to Build a Clear Path to Revenue that Attracts Investors and Acquirers
— Daniel Hawkins, CEO, Vista.ai, Former Founder & CEO, Shockwave Medical
LSI USA ‘25 Panel: Founder ≠ CEO
“Culture eats strategy for lunch every single day.”

— Dave Amerson, Former President & CEO, Neotract
LSI USA ‘25 Signature Series: 1 on 1 with David R. Amerson, Former President and CEO of Neotract
“Top-line growth is everything in medtech. Strategics are willing to pay premium multiples for growth assets. That’s the name of the game in the sector.”

— Justin Montellese, Sr. Director of Corporate Business Development (M&A), Hologic
LSI USA ‘25 Panel: Demystifying Corporate Development — How to Avoid Common Pitfalls and Run a Best-In-Class Process

Celebrating recent leadership shifts and other announcements and accolades impacting our global LSI Alumni medtech community.



Approxima has appointed seasoned medtech entrepreneur Rick Geoffrion as an Independent Board Director to support the company’s next phase of growth as it advances a percutaneous right ventricular reshaping technology for the treatment of tricuspid valve regurgitation. Geoffrion brings over 35 years of executive leadership in cardiovascular devices, having co-founded and led multiple medtech companies to successful exits, including Mitralign (acquired by Edwards Lifesciences), CircuLite (acquired by HeartWare), and FlowMedica (acquired
by AngioDynamics). He also played a pivotal role in the strategic repositioning of Impella (acquired by Abiomed) and Pathway Medical Technologies (acquired by Bayer Healthcare). Currently President and CEO of Cyrano Therapeutics and Vice Chairman of the Mullings Group, Geoffrion also serves on the board of the Medical Device Innovation Consortium.
His appointment follows the successful first closing of Approxima’s Series A round and comes as the company prepares to launch pilot clinical trials of its novel reshaping technology. Designed for percutaneous, beating-heart deployment, Approxima’s implant is intended to restore physiological function to the tricuspid valve.
“Approxima has the opportunity to be that type of therapy,” said Geoffrion, citing the need for solutions that are both effective and simple to deploy. Chairman Umberto Pasquali and
Co-Founder and CEO Michal Jaworek both emphasized Geoffrion’s strategic value as the company advances toward first-in-human studies and long-term impact in the structural heart space.

Aspira Medical has appointed LSI Alumni Zaffer Syed as Co-Founder and CEO, a move that reinforces the company’s mission to transform ICU care through non-invasive, continuous monitoring for aspiration risk. Founded out of the Texas Medical Center with
exclusive IP licensed from Baylor College of Medicine, Aspira is advancing a wearable ultrasound platform to support early intervention and reduce the incidence of aspiration pneumonia, one of the most overlooked and deadly complications in the ICU. Syed joins President & Chief Medical Officer Todd Rosengart to lead the vision, strategy, and execution at this pivotal early-growth stage.
Syed brings two decades of medtech leadership experience, including recent roles as Entrepreneur-in-Residence at the Texas Medical Center Innovation Factory and as President and CEO of Saranas. His career spans clinical development, strategic marketing, and commercial leadership across companies such as Boston Scientific, St. Jude Medical, and OrthoAccel Technologies. At Aspira, Syed’s blend of clinical insight and executional expertise positions the company to scale its impact and improve outcomes for critically ill patients worldwide.

Biomed Simulation has appointed Denise Steinbring as CEO, formalizing a leadership transition that reflects the company’s growth and strategic momentum. A 30-year medtech veteran, Steinbring brings deep expertise in simulation-based education, go-to-market strategy, and operational excellence. She joins Founder and CTO Dr. Richard Tallman in steering the company’s next phase of growth.
Steinbring’s experience includes leadership roles at Medtronic, where she spearheaded global simulation-based learning and medical education programs, and her current role as Chief Learning Officer and Partner at Chief Outsiders. At Biomed Simulation, she will lead the company’s next phase of expansion in the ECMO and cardiopulmonary bypass segments. Her appointment highlights Biomed’s
commitment to advancing patient safety and transforming clinical education through scalable, high-impact experiential learning.

GT Medical Technologies has appointed James Leech as Chief Financial and Strategy Officer to support the company’s continued expansion of GammaTile, its FDA-cleared radiotherapy implant for treating brain tumors. Leech brings over a decade of leadership across commercial-stage life science companies, with expertise in financial strategy, investor relations, and corporate development. He most recently held the same title at orthopedic device company Moximed and previously served as CBO at Palette Life Sciences, helping lead its acquisition by Teleflex. Earlier in his career, he worked in life science investment banking and strategy consulting.
At GT Medical Technologies, Leech will drive cross-functional efforts to accelerate commercial adoption of GammaTile, which is now available in over 100 U.S. treatment centers. CEO Per Langoe praised Leech’s strategic and financial acumen, noting the company’s strong growth trajectory. Leech shared his enthusiasm for joining a mission-driven team focused on elevating care for brain tumor patients through innovative, bioresorbable implants that deliver immediate, targeted radiation therapy.

to support global commercialization of the Maestro System, a robotic surgical platform powered by Physical AI. Jonathan Conta joins as Chief Marketing Officer following 16 years at Intuitive Surgical, where he led product and marketing initiatives across multiport and single-port platforms. Jeff Semone was appointed Chief Quality and Regulatory Officer, bringing leadership experience from Siemens Healthineers and Varian Medical Systems. Rounding out the team is Jeff Driggs, Vice President of U.S. Sales, who previously led sales for the AirSeal platform at CONMED
These appointments follow a surge in adoption of the Maestro System, which has now supported over 1,600 procedures globally, 1,400 of which took place in the last year. CEO Anne Osdoit noted that the new leaders will play a key role as Moon Surgical transitions out of limited market release and into its next phase of global growth. Conta called Maestro “an exciting evolution in minimally invasive surgery,” emphasizing its intuitive, scalable, and intelligent design that supports high-volume outpatient care with seamless team integration.

Orthonika has appointed Kristina Bartolo as CEO as the company advances its Orthonika Total Meniscus Replacement (TMR) implant into the final stage of preclinical development. A biomedical engineer by training, Bartolo joined Orthonika as its first employee in 2017 and has held multiple leadership roles, most recently serving as CTO. She has been instrumental in the development of the company’s flagship synthetic meniscus, designed to fully replace the native meniscus with a durable, biocompatible alternative.
Her appointment comes as Orthonika secures a £460,000 Innovate UK Smart Grant, one of just 43 awarded out of more than 1,390 applications, providing 70% funding support to accelerate the company’s next phase.

Pylon Medical has appointed Dr. Elad Levy, an internationally recognized leader in stroke and neurovascular care, to its Medical Advisory Board. Dr. Levy is widely regarded as a pioneer in endovascular neurosurgery. His clinical insight and track record of shaping stroke intervention standards make him a high-impact addition as Pylon prepares for first-in-human trials.
Pylon is developing the first and only minimally invasive direct carotid access platform, an innovative percutaneous solution designed to improve treatment for patients with carotid artery obstructions and stroke. By replacing more invasive approaches with a streamlined, trans-carotid system, Pylon aims to expand access to life-saving interventions and address critical delays in acute stroke care. Dr. Levy’s appointment signals growing momentum behind the company’s mission to redefine carotid access.

Archimedes Vascular has appointed Raymond Cohen as Chairman of the Board of Directors. Cohen is a veteran medtech executive best known for co-founding Axonics and leading it through IPO and acquisition by Boston Scientific. He currently serves on the boards of Nalu Medical, Tulavi Therapeutics, Spectrum Vascular, Kestra Medical Technologies, and SoniVie, where he played a key role in the company’s recent acquisition. His leadership at Archimedes is expected to bring critical strategic insight as the company advances its novel implant for the treatment of severe hypertension.
The appointment follows the close of Archimedes’ $2.2 million Series A financing round, led by Sherpa Healthcare Partners. The company is developing the Barostent, a device implanted in the aortic arch to activate the baroreflex and reduce blood pressure in patients unresponsive to medications or renal denervation. Founder Dr. Farrell Mendelsohn emphasized the importance of Cohen’s involvement at this preclinical stage as Archimedes prepares for FDA clearance. (Also see our cover story featuring Cohen in The Lens, June 2025.)

Recor Medical has named Shon Chakrabarti, MD, MPH, as Chief Medical Officer. A board-certified interventional cardiologist with deep experience across both clinical medicine and medtech, Dr. Chakrabarti joins Recor to lead its medical strategy and support the continued rollout of the Paradise Ultrasound Renal Denervation (uRDN) System. Most recently, he served as Chief Medical Officer at LimFlow (acquired by Inari Medical) and Director of Clinical Research and Medical Affairs at Abiomed.
At Recor, Dr. Chakrabarti will advance the company’s vision to transform hypertension care through ultrasound-based renal denervation. The Paradise uRDN system is FDA-approved in the U.S. and CE-marked in Europe. It uses 360° ultrasound to target overactive sympathetic nerves in the renal arteries, a key contributor to uncontrolled blood pressure. His appointment reflects Recor’s commitment to delivering safe, effective, and widely accessible hypertension therapies.
THINK Surgical has appointed Chris Marrus as Executive Vice President. A seasoned leader in surgical robotics, Marrus brings more than two decades of experience scaling commercial operations at companies including Intuitive Surgical, MAKO Surgical (acquired by Stryker), and Synaptive Medical

At THINK, he will support President and CEO Stuart Simpson in advancing financing, product development, and operational growth initiatives, while accelerating market adoption of the company’s TMINI Miniature Robotic System.
Marrus previously led commercial growth at MAKO Surgical through its $100 million revenue milestone and continued as VP of Robotic Sales and Service at Stryker after the acquisition. He also served as CCO at Synaptive Medical. His appointment reflects THINK Surgical’s commitment to scaling its open-platform orthopedic robotics business ahead of an ambitious 2026 growth initiative.

Virtual Incision has appointed Jim Alecxih as CEO. A veteran commercial leader in surgical robotics, Alecxih brings over three decades of medtech experience, including key leadership roles at Intuitive Surgical, where he was instrumental in driving the adoption and growth of the da Vinci Surgical System. He most recently served as CEO of AI software company DH Medical.
At Virtual Incision, Alecxih will lead the company through the development and commercialization of M2, its next-generation surgical robot designed to expand access to robotic-assisted surgery in rural hospitals, HOPDs, and ASCs. His appointment follows a year of momentum for the company, including FDA De Novo authorization of the MIRA Surgical System and multiple innovation award recognitions.
Ship2B Ventures through its BSocial Impact Fund, Clave Capital through Clave Innohealth, Nara Capital, the EIC fund, CDTI Innvierte, undisclosed
Your one-stop-shop for global market sizing and analysis, procedure volume data, startup company and deal tracking, curated insights, and more.
Corvia Medical Other Atrial shunt system to reduce HF symptoms
Inverbío SGEIC, Verge HealthTech Fund, Impact Ventures

Third Rock Ventures, General Catalyst Partners, AccelMed, Lumira Ventures, two undisclosed strategic investors
Co-Founder Anne Wojcicki's TTAM Institute. This acquisition is an outbid to Regeneron's announced acquisition in May 2025.
YOFO Medical Technology raised a Series B round of tens of millions of CNY. The company is supported by Xingtai Capital and Xingtai Venture Capital.
Droplet IV Other Automated IV flushing $2M John Lindskog (former executive of ConvaTec), undisclosed members of medtech family offices and angel investors
InnovaHealth Partners (led), GLIN Impact Capital, Wealthing VC Club, Citrine Angels, Wavemaker Three-Sixty Health, Kimera Limited, Women’s Venture Capital Fund, Golden Seeds
Daring Capital, Social Tides, Lightbulb Trust, ULTRA VC, Dr. Helmut Schuster 6/18/2025
software for ultrasound
Racine² (led), BNP Paribas Développement, Épopée Gestion, Good Only Ventures, Better Angle, NACO 6/18/2025

Source: LSI USA ‘23
As a visionary biomedical informatician, pediatrician, and biotech entrepreneur, Dr. Atul Butte leaves behind an extraordinary legacy, defined not only by his groundbreaking work in data-driven medicine, but also by his contagious positivity and unwavering commitment to uplifting others. He was as fierce an advocate for human potential as he was for scientific progress, and the fields of translational and clinical bioinformatics that he pioneered will benefit for generations to come. Here, we pay tribute to his transformative work and passion for human health.
“Once we let data flow, lifelong discoveries become possible.”
This statement truly defines the transformational achievements and life
of Atul Butte, MD, PhD, a respected champion in the use of big data in science and health, and one of the most highly cited researchers in his field.
When he walked on stage at a recent LSI USA event in Dana Point, CA to give a compelling keynote address,
entitled “Precisely Practicing Medicine with a Trillion Points of Data,” he first grabbed everyone’s attention with his dazzling, friendly smile. Then, a single slide crammed with company logos appeared behind him. Dr. Butte noted that the list represented his “conflicts of
interest,” and grinned: “I wouldn’t blame you if you didn’t believe another word I say.”
What followed though was an unforgettably inspiring talk that was equal parts manifesto and master class, ranging from a rapid-fire review of the power of big data, to electronic health record treasure troves, and the story of three investor-backed startups he created out of public datasets: NuMedii, Carmenta, and Personalis, the latter which IPO’d at a peak $2 billion valuation. Dr. Butte hammered home a simple thesis: data, unlike oil, multiplies when shared.
“I hate the saying that data is the new oil,” he said. “You might take the same dataset and build a diagnostic; I might find a therapeutic. We can both win.”
That relentless optimism and drive, and the code, companies, and protégés that these traits unlocked, defined Butte’s life. When he passed away on June 13, 2025, at just 55, tributes poured in from University of California, San Francisco (UCSF) colleagues, Silicon Valley CEOs, and thousands of clinicians who now rely on the algorithms he once sketched on cocktail napkins.
“It’s a rare person who leaves their mark on a university like Atul,” noted UCSF Chancellor Sam Hawgood, in a tribute post on the university’s website. Under Dr. Butte’s leadership, UCSF became a global hub for artificial intelligence, informatics, and translational science, advancing healthcare in service of patients and discovery alike. And, his unparalleled impact on the UC Health system offers valuable lessons for all healthcare entities around the world.
Born in Philadelphia in August 1969, young Atul toggled effortlessly between Legos and BASIC code. He entered Brown University’s Program in Liberal Medical Education at 17, picking up a computer science degree and working as a software engineer at Apple and Microsoft, en route to his MD in 1995. A pediatrics residency and a fellowship in pediatric endocrinology at Boston Children’s followed, along with a PhD
from the Harvard-MIT Health Sciences and Technology program. Here, he met mentor Isaac Kohane, MD, PhD, who once urged him to narrow his research focus, as noted in the UCSF post. The story goes that Butte smiled back and said, “I’m very high-throughput. It takes a lot more to overwhelm me.”
A Stanford University faculty position in 2005 gave him the opportunity to prove it. There, he helped coin the term “translational bioinformatics,” referring to the convergence of molecular bioinformatics, biostatistics, statistical genetics, and clinical informatics. He also published a pair of seminal Science Translational Medicine papers showing how public bioinformatics data could identify new uses for approved drugs. One paper birthed his company NuMedii, which hunts for new uses for existing drugs within open access molecular data. Another seeded Personalis, which provides medical genome sequencing services. He also co-founded Carmenta, developer of a protein-based test for preeclampsia, a hypertensive disorder of pregnancy. In 2015, the company was acquired by Progenity, 18 months after it spun out of Stanford University (more on his datadriven companies later). He was also a co-inventor on 24 patents, including protein and gene biomarkers for rejection of organ transplants, biomarkers for ovarian cancer, methods of prognosing preeclampsias, and methods for diagnosis and treatment of non-insulin dependent diabetes mellitus.
Dr. Butte proudly served as the Priscilla Chan and Mark Zuckerberg Distinguished Professor of Pediatrics, Bioengineering and Therapeutic Sciences, and Epidemiology and Biostatistics at UCSF, inaugural director of the UCSF Bakar Computational Health Sciences Institute, and the first chief data scientist at the University of California (UC) Health System. In this role, Dr. Butte melded the clinical data from six UC medical centers into a single, de-identified data warehouse that comprises 9 million patients, 378 million encounters, 1.3 billion meds, 5.2 billion lab values, 44 million medical
device uses, and much more, over an 11-year span. And, all the data is centrally updated on a nightly basis.
Under his watch, pharmacists slashed unnecessary intravenous acetaminophen, saving millions; oncologists benchmarked biologic spending across campuses; and investigators used the data trove to secure an FDA nod for a cryo-nerve block in adolescents, without enrolling a single new patient.
Dr. Butte showed the LSI USA audience some of the powerful data that has been reaped from the centralized UC Health data warehouse, and how operational teams throughout the health system are benefitting from this data every day and saving millions of dollars in the process. Teams have been able to draw insights from billions of de-identified data points that have been aggregated and harmonized on an ongoing basis since 2012 from both UCSF (3 million patients) and the entire UC Health system (7 million patients), through the UCSF Center for Real World Evidence
“The future of evidence-based medicine is datadriven medicine.”
“The future of evidence-based medicine is data-driven medicine,“ he explained. “Instead of let’s try this treatment, then if that doesn’t work let’s try that, how about let’s use the power of data to get patients under better therapy sooner. We can do this across hospital systems, and across disease states. I think the future is not where we learn with our data, I think the computer is going to teach us automatically how to improve our care. If this seems farfetched, it’s really not. It’s happening now. It’s an amazing time for the power of clinical data to empower researchers, entrepreneurs, and communities.”
Dr. Butte didn’t just publish and speak on the power of data; he turned it into diagnostics, therapeutics, and

Atul Butte, MD, on stage at LSI USA ‘23
real-world solutions. His entrepreneurial efforts reflected his belief that open datasets could be mined not only for insight, but for impact.
“If you want to change the world, you can’t just keep writing papers about it. The science continues in the startup companies.”
Each of his companies began with a deceptively simple question: What if we could put existing data to better use? For Dr. Butte, the answers fueled not just academic discovery, but tangible progress toward more precise, affordable, and impactful care. Across each venture, he demonstrated how
public science could be translated into patient-centered innovation.
NuMedii, founded in San Mateo, CA in 2008, discovers and de-risks effective new drugs by translating life sciences big data into therapies with a higher probability of therapeutic success. The company’s proprietary and dynamic big data technology, developed in Dr. Butte’s lab at Stanford University and licensed exclusively to NuMedii, consists of hundreds of millions of human, biological, pharmacological, and clinical data points that the company normalized and annotated.
Dr. Butte’s team realized that thousands of gene-expression studies mandated by the National Institutes of Health now lived online, free for the taking. By matching disease signatures to drug profiles, they generated a
conveyor belt of new uses for old drugs, and developed proprietary Artificial Intelligence for Drug Discovery (AIDD) technology. NuMedii’s AIDD technology employs deep learnings of human biology, coupled with proprietary machine learning and network-based algorithms to discover and advance precise, effective new drug candidates, as well as biomarkers predictive of efficacy for subsets of patients across a broad spectrum of therapeutic areas.
The company formed active R&D collaborations with several global pharmaceutical and biotech companies and patient-centric organizations, including Yale School of Medicine and Brigham and Women’s Hospital, to create pipeline and new products in multiple therapeutic areas including inflammation, oncology, and, rare diseases. NuMedii raised its first $10 million and pivoted to idiopathic pulmonary fibrosis, an orphan disease that is chronic, progressive, and usually fatal, pulling a potential therapy from the digital ether in just months.
“Every one of you can come up with a new diagnostic for a different disease and we’d never step on each other’s toes. We need that many diagnostics in medicine today.”
Preeclampsia kills 70,000 pregnant women worldwide each year. Carmenta began by downloading every publicly available placental dataset, hunting for convergent biomarkers. By analyzing every available dataset on preeclampsia, Dr. Butte’s team developed a blood-based diagnostic test for this life-threatening pregnancy complication. Within 18 months, the company went from GitHub repo to $2 million in funding, to acquisition by Progenity, validating Dr. Butte’s creed that “open data is venture fuel.”
“The data is out there, the papers are out there. It’s up to you to realize this need is coming, and ask, what can I do about it now to address it in three to five years?”
In 2008, a San Jose high schooler in Dr. Butte’s lab manually curated 50 genetics papers to build a list of spots on the DNA associated with diseases. The exercise snowballed into the world’s most comprehensive variant
disease catalogue, and Personalis. The company IPO’d in 2019, reached a $2 billion market cap, and its cancer monitoring assay now guides therapy for thousands of patients. Its technology even underpins the Defense Advanced Research Projects Agency’s (DARPA’s) genomic surveillance program.
Cancer slowed but never silenced him. Even in treatment, Dr. Butte spoke of “maps of death indices,” which are animated trajectories of Californian lives arcing from wellness to organ failure to sepsis. The point wasn’t morbidity; it was agency: if we can predict the next 90 days, we can intervene today. His team is continuing to push that work forward.
“I want to know how our patients are going to die, so we can change the ending.”
Dr. Butte has been recognized with numerous scientific accolades over the years. By 2013, he was an Open Science Champion of Change at the White House. In 2015, he was elected to the National Academy of Medicine. In 2024, he was honored with the Morris F. Collen Award of Excellence, the highest honor in biomedical informatics. He also received the Association for Molecular Pathology’s Award for Excellence for his pioneering work in molecular diagnostics and computational health sciences. In April of this year, he was recognized for his outstanding career in the computational and health sciences with induction into the American Academy of Arts and Sciences, one of the most prestigious and oldest honor societies in the U.S.
By any metric, from citations, to startups, to colleagues and patient lives touched, Dr. Atul Butte leaves a super-
cluster of impact. Yet friends say he measured success differently: in the number of students and colleagues who he inspired, and in the terabytes of public health data waiting to be mined.
“He brought this incredibly optimistic view of what can be accomplished,” Chancellor Hawgood noted in the UCSF post, “and it’s changed the way the entire institution undertakes digital transformation.” Hawgood’s statement, also joined by Dr. Talmadge E. King, Jr., Dean of UCSF School of Medicine, Suresh Gunasekaran, President and CEO at UCSF Health, and Dr. David Rubin, Executive Vice President at UC Health also notes: “While we mourn this profound loss, we also celebrate a life lived at the intersection of compassion and computation. Dr. Butte showed us what is possible when we unite data, medicine, and purpose. We honor him by continuing the work he championed: unfreezing data, advancing equitable innovation, and ensuring that discovery always serves humanity.”
A memorial fund now supports cancer research, and Bakar Computational Health Sciences Institute has planned an annual Butte Lecture for computational health disruptors.
Meanwhile, every time a clinician at UC Health swaps an EHR-driven insight for unnecessary guesswork, or a biotech repurposes an old molecule for a new disease, a quiet echo of Dr. Butte’s inspirational quote from the LSI stage lives on: “Data multiplies when shared.”
Special note: All of us here at LSI have been deeply impacted by Dr. Butte and his extraordinary vision, and we offer our deepest condolences to his wife, Gini Deshpande, their daughter, and his family and friends.
• LSI USA ‘23 Panel Discussion: Powering Up Innovation in a Digital, Connected World, featuring Atul Butte, MD, PhD, Gretchen Purcell Jackson - Intuitive, Josh Makower - NEA, Todd Brinton - Edwards Lifesciences, and Joe Smith - BD
• What Happens When We Mix Real Doctors, Big Data, and AI? Dr. Atul Butte’s Tedx Talk, December 2018

Atul Butte, MD, on stage at LSI USA ‘23
Atul Butte, MD, on stage at LSI USA ‘23
the reports daily. The entire healthcare system (and California) owes him a debt of gratitude. I’m going to miss the insightful conversations. But most of all, I’m going to miss sitting at our local sandwich shop and watching Atul come around the corner getting his steps in while on a call. We’d wave to each other as he passed by and I knew he was helping solve a problem for someone. It was always motivation for me to pick up the phone and call someone to see if I could help someone. Thanks for being an inspiration Atul.”
- DJ Patil, Investor, entrepreneur, public policy, former U.S. Chief Data Scientist (excerpted from his June 2025 LinkedIn post)
“Atul has had an immeasurable impact on my career and my life. I met Atul nearly 20 years ago when I was interviewing for graduate school and he was giving a job talk at Stanford. I was blown away by his infectious excitement, charisma, and the potential impact of the work he was proposing. I was fortunate to join his lab in 2006 and after a brief stint at Pfizer join him back in academia first briefly at Stanford and then at UCSF in 2015.
From all corners of the Life Sciences, the heartfelt tributes, fond memories, and inspiring stories of Dr. Butte are too numerous to list. A few poignant testimonials gathered by The Lens are noted below.
From all corners of the Life Sciences, the heartfelt tributes, fond memories, and inspiring stories of Dr. Butte are too numerous to list. A few poignant testimonials gathered by The Lens are noted below.
“The passing of Atul Butte still weighs heavy on me. There are some people that are just special. He was one of those people … his thinking and approach to healthcare and medicine was ahead of its time. And, frankly, I couldn’t have done my job at the White House without his input.
When I found myself back in public service during COVID, of course whose email showed up with what the entire UC System was seeing in regards to COVID? Yep. Atul’s email. He mobilized fast and gave us insights that were essential. And then he figured out how to automate the whole thing to get us
Nearly everything that I know about doing science I learned from Atul. He taught me how to ask questions, how to think big, how to mentor and bring collaborative teams together. He taught me the importance of communication and telling a story. But I’m only one of hundreds if not thousands of people that he has inspired. He was a true innovator and a champion. He was very supportive of getting students excited about science early in their career and had numerous high school interns in his lab throughout the years. He was a fierce advocate of women in science. He had a unique ability to elevate people around him, making them feel good about themselves and the work that they did which in turn inspired them to grow. He was incredibly loyal and dedicated - to his work and his family. He was an optimist and his positive outlook and energy was contagious.
He was a fighter and one of the strongest people I know. The world has lost an amazing person. He was truly one of a kind. Atul Butte, I’m incredibly thankful for the opportunity to work with
you and learn from you over the past 19 years and I will do my very best to continue your legacy at University of California, San Francisco and beyond. Rest in peace and may your memory be an inspiration to many generations to come.”
- Marina Sirota, PhD, Professor and Acting Director, Bakar Computational Health Sciences Institute, UCSF (excerpted from her June 2025 LinkedIn post)
“Atul Butte was my mentor, colleague, and close friend for 20 years across two institutions. I loved everything about him - his huge smile, contagious laugh, passion for our field, and amazing ideas. He dreamt up concepts like ‘machine learning healthcare systems’ and ‘AI as scalable privilege’ and he inspired me and countless people around the world to think bigger. I will miss him but my life is immeasurably better for knowing him and I am grateful for the time we spent together. May his memory be a blessing to all who knew him and his legacy inspire those who didn’t.”
- Chris Longhurst, MD, Chief Clinical and Innovation Officer; Executive Director, Jacobs Center for Health Innovation, UC San Diego Health (excerpted from his post on Kudoboard)
“Atul had that rare gift of turning complex science into something captivating and deeply human. I first saw him speak about AI and data at a packed global event, and he had the entire audience spellbound. Not just learning, but laughing, inspired, and moved to act. He was a brilliant communicator, a connector, and a deeply principled leader who never lost sight of impact. You didn’t just want to hear him speak, you wanted to be part of his orbit. He was the real deal: physician, data scientist, entrepreneur, educator, and just an all-around good human being. We are all better for knowing him.”
- Lisa Carmel, Chair - Business Development, Mayo Clinic (excerpted from a recent interview with The Lens)
and medicine. He had an infectious joy in discovery, an unshakable belief in the power of open data, and a gift for showing us all that the future of healthcare wasn’t just possible, it was inevitable, and we all had a role to play.
What made him truly remarkable was not just his vision, but how generously he shared it. He didn’t seek credit; he invited others in. Long before AI became a buzzword, Atul was already connecting machine learning, molecular biology, and public health to improve lives. His time with us was too short, but his legacy will endure in the tools he built, the problems he helped solve, and most of all, in the people he inspired.”
- Joe Smith, MD, PhD, Senior Vice President & Chief Scientific Officer, BD (excerpted from a recent interview with The Lens)
“In honor of my dear friend Atul Butte, whose fierce authenticity and magnetic enthusiasm lit every room; whose courage in speaking uncomfortable truths, boundless innovation, and visionary service to science carved highways in the tapestry of human possibility. I miss you!
The Spark Ideas lit the room, he spoke, and silence listened.
Fire glowed within his eyes, he filled the air and hearts then left them brighter.
Even data danced, when he looked at it with joy.
Whispers in the code, echoes in the minds he moved.”
- Yves Lussier, MD, Chair of Biomedical Informatics at University of Utah School of Medicine (excerpted from his post on Kudoboard)
in our field was both grounding and inspiring. He will be deeply missed, but his impact will continue to resonate in all of us who had the honor of knowing him and well beyond. My deepest condolences to Gini, Kimi, and Atul’s extended family.”
- Genevieve Melton-Meaux, MD, PhD, FACMI, FACS, FACSRS, professor in the Department of Surgery and Institute for Health Informatics, Senior Associate Dean of Health Informatics and Data Science, and Director of the Center for Learning Health System Sciences at the University of Minnesota (excerpted from her post on Kudoboard)
“I am saddened by the loss of one of my scientific mentors and one of the great physician-scientists of this era. I will be forever grateful that Atul took a chance on me during a time when I was struggling. I would not be the physician-scientist I am today (in fact, I might not have even made it at all) without his support and mentorship.
I will carry with me his infectious optimism and enthusiasm, his chase of the big important questions, his belief that it is the role of a good scientist to apply their findings to real world benefit of others, and his teaching that credit is infinitely divisible. You can see his imprint on my career as a pediatric immunologist and genome editor designing genetic therapies for children with rare diseases.
Above the science was Atul’s genuine passion for lifting up others and his love of family. When I updated him about my faculty appointment and the birth of my son, you can tell from his very characteristic response where his heart lay: “... but even more important, congrats on your new Large Language Model, and so great to see exponential growth in those neurons!!”
“Atul was a rare and catalytic force at the intersection of science, engineering,
“It’s hard to put into words the loss of Atul Butte. Through AMIA and the field of informatics, I had the privilege of calling him a colleague, a mentor, and a friend. Atul had a rare gift—his clarity of thought could cut through complexity, his wit could light up a room, and his ability to connect with people on multiple levels was nothing short of extraordinary. He offered me career advice that shaped my path, and his presence
Warp speed, Atul.
- Matthew Kan, MD, PhD, Butte Lab postdoc 2016-2017, Assistant Professor of Pediatrics, UCSF (excerpted from his post on Kudoboard)
Note: Quotes used with permission, or cited from public sources.
Cardiac ablation devices are seeing rapid adoption as advances in pulsed-field ablation (PFA) and forcesensing catheter technology reshape the treatment of atrial fibrillation. Following FDA approval of Boston Scientific’s Farapulse PFA system in January 2024, the market is experiencing strong momentum driven by more accurate atrial fibrillation (AFib) detection, increasing procedure volumes, and a shift toward ablation over long-term drug therapy.

Cardiac ablation, which uses radiofrequency, cryoablation, or PFA to eliminate aberrant conduction pathways, has become a mainstay in the treatment of arrhythmias like AFib. Between 2016 and 2021, procedure volumes in the U.S. Medicare population grew at an average annual rate of 16.2%. Growth continued post-pandemic, with volume rising 76% from 2021 to 2024. This surge reflects broader adoption, better diagnostics, and improvements in procedural safety and efficacy.

The most significant development of the past couple of years was the FDA approval and subsequent adoption of Boston Scientific’s FARAPULSE PFA System. The technology generated over $1 billion in global revenue in 2024 alone, illustrating the rapid uptake of PFA. Unlike traditional thermal ablation methods, PFA uses non-thermal energy to precisely target cardiac tissue while minimizing damage to surrounding structures. PFA also significantly reduces ablation procedure times, leading to meaningful cost savings. Current clinical data shows a
30% to 50% reduction in procedure duration, with these savings likely to grow as next-generation technologies are developed and the training curve flattens.
As more companies bring PFA platforms to market, the shift away from thermal ablation is expected to accelerate. Johnson & Johnson MedTech’s Biosense Webster unit and other leading electrophysiology players are actively developing competitive PFA systems, which could further expand the market and improve access to care.
The cardiac ablation device market is projected to grow at a compound annual growth rate of 16.4% from 2024 to 2029, outpacing the overall medtech market. Global sales are expected to rise from $4.0 billion in 2024 to $8.5 billion in 2029, with unit volume more than doubling over the same period. Despite this growth, average selling prices are expected to remain relatively stable, increasing only modestly from $3,622 to $3,660.
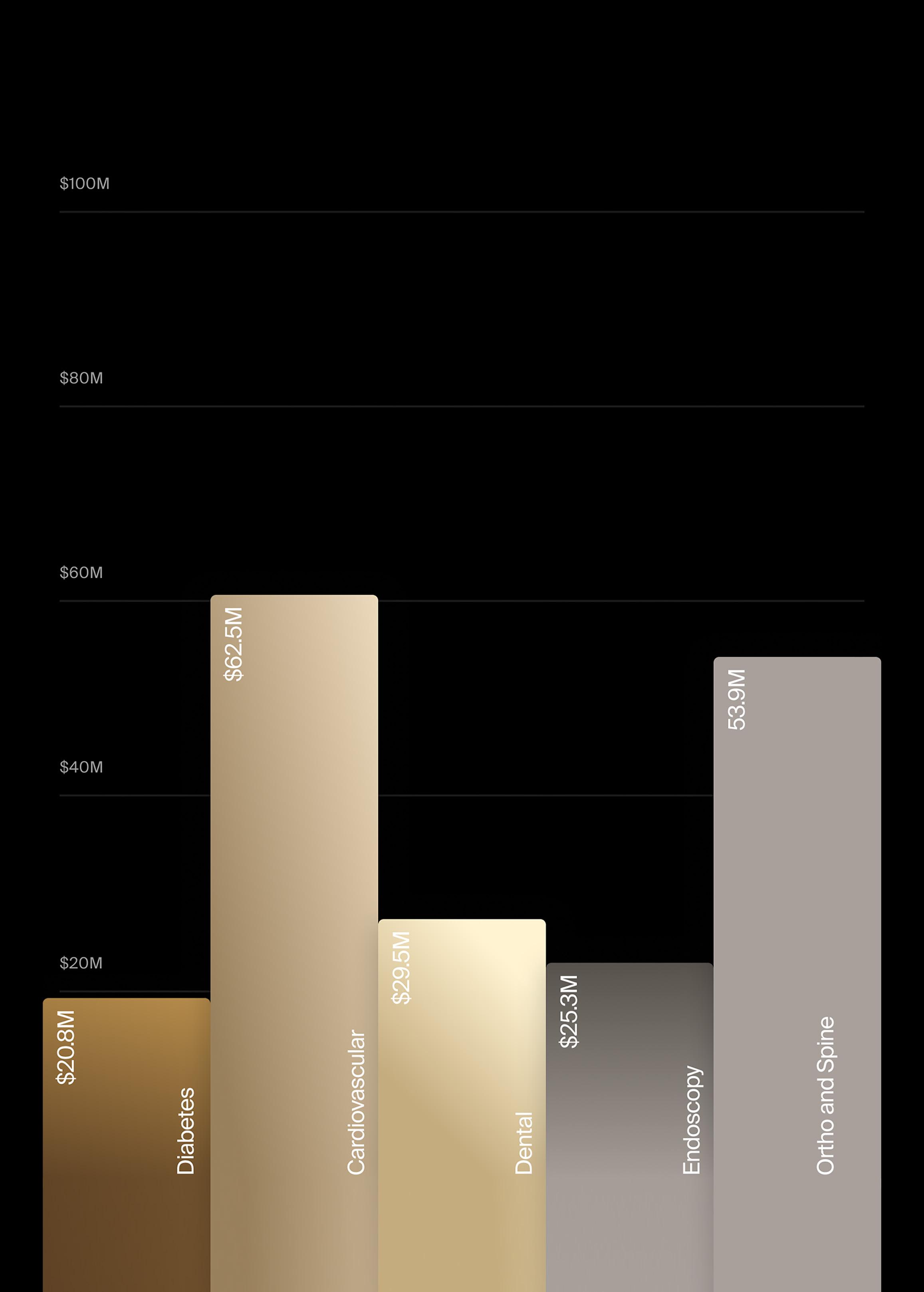
• J&J/Biosense: $2.39B
• Boston Scientific: $1.02B
• Medtronic: $197M
• AtriCure: $188M
• Abbott: $124M
• Stereotaxis: $22M
• Others (BIOTRONIK, AngioDynamics): $30M
Increased detection of AFib through wearable and ambulatory devices like AliveCor’s KardiaMobile is helping clinicians identify arrhythmias earlier, enabling more patients to benefit from ablation. Simultaneously, advancements in mapping, navigation, and catheter design are improving procedural accuracy, reducing risks, and shortening recovery time.
These trends are pushing ablation therapy toward earlier use in the treatment algorithm. Historically reserved for patients who failed drug therapy, ablation is increasingly being considered as a first-line intervention.
The cardiac ablation market continues to accelerate with notable regulatory wins, product approvals, and public funding announcements.
In October 2024, Medtronic achieved FDA approval for its Sphere-9 Catheter
and Affera Mapping and Ablation System. Designed to treat both AFib and typical atrial flutter, the system allows physicians to toggle between pulsedfield and radiofrequency energy. In a 477-patient clinical study, the system demonstrated non-inferior outcomes to the THERMOCOOL SMARTTOUCH SF Catheter, with nearly 74% of patients free from arrhythmia recurrence at 12 months.
In January 2025, Johnson & Johnson MedTech announced CE Mark approval for its Dual Energy THERMOCOOL SMARTTOUCH SF Catheter, which integrates both pulsed-field and radiofrequency ablation in a single device. Early results from the SmartfIRE clinical trial showed a 100% acute success rate with first-pass isolation achieved in 96.8% of veins. The company also completed enrollment for the SmartPulse and PulseSmart trials in the U.S., Canada, and Australia, signaling a strong push toward global market access.
In April 2025, Adagio Medical received FDA Breakthrough Device Designation for its vCLAS cryoablation

Source: LSI Market Intelligence
system, which targets drug-refractory, recurrent, sustained monomorphic ventricular tachycardia (VT) in patients with ischemic or nonischemic structural heart disease. The vCLAS system, already CE Marked in Europe, is now being evaluated in the pivotal U.S. FULCRUM-VT IDE study and aims to improve the safety and efficacy of VT ablations through ultra-low-temperature cryoablation technology.
In May 2025, LuxMed Systems, a Massachusetts-based startup, was awarded a $100,000 START grant from MassVentures to advance its sensor-based PFA platform. The company’s technology enables real-time metabolic tissue feedback for direct lesion assessment and gap detection. LuxMed was one of 26 companies selected across deep tech sectors, including cleantech, biotech, advanced materials, and quantum computing.
David Neale
Argá Medtech is developing a next-generation PFA system powered by its proprietary Coherent SineBurst Electroporation (CSE) Ablation System. The company’s single-shot catheter can deliver circular, linear, and focal lesions to make AFib treatment faster, safer, and more efficient than current solutions. In February 2024, Argá raised a €54 million oversubscribed Series B to support clinical activities.
Initial results from the BURST-AF study, presented at HRS 2025 and published in Heart Rhythm Journal, showed >98% lesion durability at three months and high freedom from
arrhythmia at six months, 94.7% in paroxysmal AFib and 90% in persistent AFib patients. With strong clinical momentum and a differentiated technology platform, Argá is emerging as a major contender in the competitive PFA landscape.
CEO: Steven Mickelsen
Field Medical is developing a purpose-built PFA system engineered for ventricular use. Unlike first-generation platforms optimized for the atria, Field’s system combines PFA with contact force sensing, electromagnetic localization, and AI-enabled mapping to overcome the unique challenges of the ventricle’s thicker, highly mobile tissue.
In December 2024, Field Medical received both FDA TAP Pilot acceptance and Breakthrough Device Designation for its FieldForce Ablation System. The company closed a $40M Series A in April 2025 and followed with a $35M Series B in July 2025.
CEO: Jessie Lian
Pulnovo Medical is advancing interventional treatment for pulmonary hypertension (PH) and heart failure with its pulmonary artery denervation (PADN) technology. The company’s radiofrequency-based system targets the pulmonary vascular endothelial sympathetic nerve to reduce pulmonary artery pressure and slow disease progression. The technology is already commercially available in China and has received FDA Breakthrough Device designation.
In March 2025, Pulnovo closed a nearly $100 million Series C to support FDA trials for Group I and II PH and expand international operations. With global clinical trials underway, Pulnovo is positioning PADN as a first-line interventional therapy for PH worldwide.
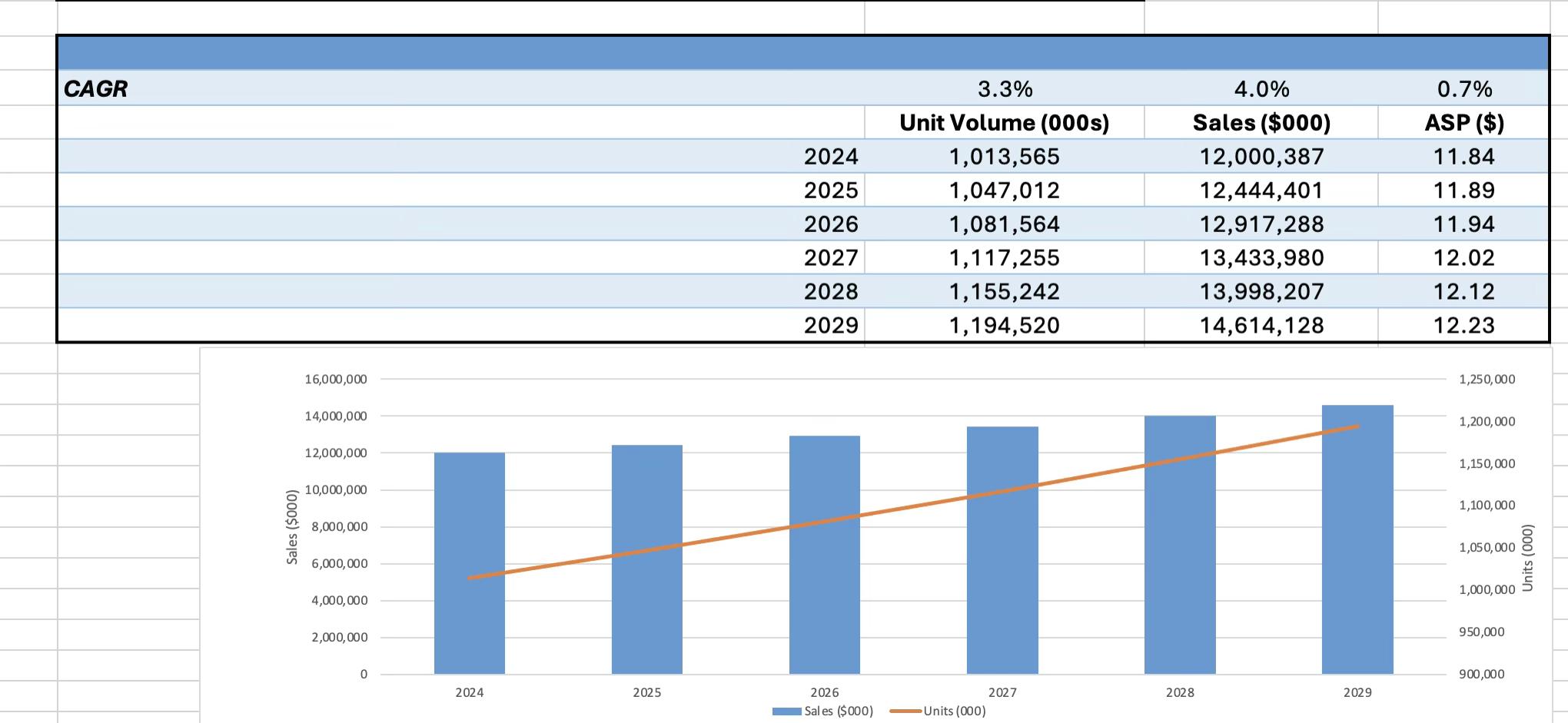
The global hemodialysis catheters and dialyzers market continues its upward climb, driven by the expanding burden of end-stage renal disease (ESRD), rising rates of diabetes and hypertension, and the continued reliance on hemodialysis as the primary treatment modality. With nearly 90% of the world’s 3.8 million dialysis patients receiving hemodialysis, demand for vascular access catheters, cannulae, and dialyzers remains strong and steadily growing.


In 2024, global sales of hemodialysis catheters and dialyzers reached $12.0 billion across more than one billion units. The market is projected to grow at a compound annual rate of 4.0% over the 2024–2029 forecast period, with unit volume increasing 3.3% annually and average selling prices rising modestly at 0.7%. Aging populations and the global rise in chronic illnesses are key drivers of demand. According to the U.S. Renal Data System, the U.S. ESRD population nearly doubled between 2002
and 2022, and more than 60% of new ESRD cases in 2022 were associated with diabetes. Globally, the prevalence of diabetes and hypertension, the two leading causes of ESRD, is projected to rise dramatically through 2045.
As access to care expands, particularly in Asia and other emerging markets, more patients are staying on dialysis longer, increasing demand for consumables like catheters and dialyzers
in hospital, clinic, and home settings. Yet, the clinical and economic burden remains high. Each year, over 500 million dialysis treatments are performed globally, but long-term use of catheters carries risks. A recent study found that patients with long-term tunneled-cuffed catheters (L-TDCs) experience persistently elevated inflammatory markers such as C-reactive protein, highlighting the need for improved access solutions and innovation in vascular access technologies
Meanwhile, research groups and startups are exploring next-generation materials to enhance catheter
performance. Cerulean Scientific, for instance, received an NIH grant to develop a slippery omniphobic coating designed to resist biofilm and fibrin sheath formation, two major causes of catheter dysfunction and infection. These advances could reduce catheter-related complications and improve patient outcomes over time.
The 2024 supplier landscape was dominated by Baxter, Nipro, and Fresenius, who collectively accounted for over 75% of global sales. Baxter’s $3.8 billion divestiture of its Baxter Kidney Care business to Carlyle in 2024, now operating as the standalone company Vantive, marked a significant shift. This move is expected to refocus Vantive’s strategy on core innovation and global expansion in renal care.
Additional momentum came from Fresenius, which received FDA 510(k) clearance in March 2025 for its FX CorAL 40 and 50 dialyzers. These highflux dialyzers are single-use, high-flux hemodialyzers that are steam sterilized and supplied sterile for the blood pathway and free of pyrogens. These devices facilitate the exchange of water and solutes between the blood and dialysate through semipermeable hollow fiber membranes.
New paradigms for dialysis membranes are beginning to emerge. A Vanderbilt-led team recently developed carbon nanotube-based membranes that demonstrated significantly higher hydraulic permeability and improved removal of middle molecules, an area where conventional filters often fall
short. Their approach could enable smaller, more efficient dialysis kits in the future and lay the groundwork for home or even implantable systems. These incremental innovations, though often overshadowed by larger infrastructure changes, represent critical gains in patient care.
Despite its maturity, the hemodialysis catheters and dialyzers market continues to evolve. Epidemiological trends point to sustained demand, while recent M&A activity and regulatory clearances reflect ongoing industry investment. As access expands, especially in underserved regions, and new technologies address longstanding complications like infection and inflammation, the market presents a stable yet strategically significant growth opportunity across global medtech.
Source: LSI Market Intelligence
De-risk your biggest strategic decisions with our most comprehensive market data and projections (worldwide sales, share by supplier, CAGRs) on 27 major market segments and 200+ sub-segments, prepared with consistent methodologies and forecast periods.

Every week, LSI’s Market Intelligence team tracks the standout shifts, strategies, and signals shaping the global medtech industry. From robotic energy tools and next-gen imaging to disruptive cardiac ablation and regional access trends, The Numbers offers a clear, data-driven lens on what matters most. Here’s what our analysts followed in June across surgical energy, startup innovation, pulsedfield ablation (PFA), and the European medtech market.


Electrosurgical devices have powered modern surgery since the early 20th century, but their role is rapidly evolving as surgical robotics gains traction globally. These technologies are no longer standalone instruments;
they are becoming integrated tools within next-generation robotic systems. (Also see: Market Dive: Electrosurgical Devices, The Lens, June 2025.)
• Global market size (2024): $10.2 billion
• Projected market size (2029): $14.7 billion
• CAGR (2024–2029): 7.6%
• Global soft tissue surgical procedures (2024): 151.7 million
• Projected (2029): 174.9 million
• CAGR for soft tissue surgical procedures (2024–2029): 3.0%
Intuitive Surgical has rapidly grown its share of the electrosurgical devices market, rising from 16% in 2020 to an estimated 25% in 2024. At this pace, it is projected to become the market leader in 2025. This shift reflects a broader transition toward
bundled device-platform ecosystems. Medtronic and Johnson & Johnson are racing to build competing systems with integrated energy platforms.
• Medtronic’s Hugo system pairs modular robotics with its Valleylab energy platform.
• J&J’s Ottava is still in development, but its Ethicon portfolio positions it well for surgical energy integration.
• US Medical Innovations: Canady Helios Cold Plasma System
• Creo Medical: CROMA Advanced Platform powered by Kamaptive technology
As robotic systems continue to gain global market share, electrosurgical tool-
makers must adapt or risk becoming obsolete. The winners will be those who recognize energy tools as a foundational piece of the surgical workflow, not just an accessory.

At the inaugural LSI Asia summit, 75 early- and growth-stage companies presented their technologies. Here, LSI’s Market Intelligence team spotlights five of these companies for their innovative approaches and compelling market opportunity.
• Presenter: Grace Katzschmann
• Stage: Series A

• Currently Raising: $10 million
• Focus: Remote robotic navigation for ischemic stroke
• Market Insight: In the U.S., only 184,200 ischemic stroke interventions occurred in 2024 out of over 700,000 cases. That means nearly 70% go untreated.

• Presenter: Bo Peng
• Stage: Seed
• Currently Raising: $4.5 million
• Focus: Intravascular lithotripsy (VIL) pre-treatment for aortic stenosis (AS) to improve transcatheter aortic valve replacement (TAVR) outcomes
• Market Insight: Over 19 million people over 75 globally have severe AS, yet calcification increases procedural risks and has been shown to increase costs by 20–60%.

• Presenter: Tomer Ben-David
• Stage: Series A
• Currently Raising: $12 million
• Focus: Cloud-based, 3D ultrasound imaging for point-of-care use
• Market Insight: The global ultrasound market is projected to grow at a CAGR of 5.9%, reaching $13.3 billion by 2029.

• Presenter: Ella Fung
• Stage: Seed
• Currently Raising: $5 million
• Focus: Digital PCR-based noninvasive prenatal testing (NIPT)
• Market Insight: The global digital PCR market is expected to double to $1 billion by 2028. Around 3% of live births are affected by congenital abnormalities.

• Presenter: Yi-Kai Lo
• Stage: Series C
• Currently Raising: $55 million
• Focus: Noninvasive neuromodulation for paralysis (ExaStim)
• Market Insight: The $6.0 billion neuromodulation market remains dominated by implants, but less
invasive alternatives are gaining ground. (Also see: Market Dive: Neuromodulation Devices, The Lens, September 2024.)
These innovators are reshaping categories from diagnostics and neuromodulation to robotic stroke therapy, helping meet unmet needs across aging, chronic, and underserved patient populations in APAC and beyond.
PFA is upending the atrial fibrillation (AFib) treatment landscape with unprecedented speed. First approved in Europe in 2021, PFA is gaining clinical and commercial ground due to its selective electroporation, shorter procedure times, and promising safety profile.
• Global PFA market (2024): $1.0 billion
• Projected market (2029): $4.1 billion
• CAGR (2024–2029): 32.2%
• Comparative CAGR for radiofrequency (RF) and cryoablation: 8.1%
By 2030, LSI projects that PFA will become the dominant ablation modality.
New indications: expansion into persistent, long-standing, and complex AFib cases (as well as being explored beyond AFib)
Combination devices: incorporating mapping and hybrid energy-delivery to enhance the flow of these procedures.
Boston Scientific nearly doubled its market share from 2023 to 2024, while J&J’s Biosense Webster lost 10% due to a delayed rollout. This competitive volatility reflects the rapid evolution of the field. (Also see Market Dive: Cardiac Ablation Devices, this issue.)
• Argá Medtech: Raised $68.4 million Series B to develop Coherent Sineburst Electroporation (CSE) for linear and focal ablation
• LuxMed Systems: Developing optically enabled ablation catheters with real-time feedback
• Field Medical: Raised $40 million Series A for an AI-enabled, contact-sensing PFA platform
The next phase of growth will focus on expanding indications beyond AFib, combining imaging and ablation, and earning a place in global clinical guidelines. The technology is not just replacing thermal ablation; it’s redefining the rhythm care continuum.

Europe remains one of the world’s largest and most influential medtech markets, but regulatory complexity,
Guidelines: while end-users recognize the potential of PFA (as demonstrated by market growth), it is still not to the point of being recognized as the standard of care.
Source: LSI Market Intelligence
Access: PFA requires new capital investments and training requirements, which is delaying the rollout of the technology in resource-limited settings.
demographic shifts, and cost pressures are driving a new wave of recalibration.
• Population: 740 million
• Surgical procedures performed (2024): 63 million
• Projected (2029): 70 million
• CAGR for procedure volume: 2.3%
• Medical device sales (2024): $125.3 billion
• Share of global medtech market: 20%
• CAGR for European sales (2024–2029): 4.1%
• Global medtech market CAGR for comparison: 6.6%
Europe’s population peaked in 2020 and is now slowly declining, but an aging demographic supports sustained demand. Regulatory tightening under the EU Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR) is pushing some innovators to seek first approvals in the U.S. instead.
• Tightening regulatory standards are influencing go-to-market strategies.
• Adoption of digital and AI-driven health solutions is rapidly advancing.
• Talent and labor shortages are impacting tech adoption and implementation.
• Rising healthcare costs are driven by aging populations and the increasing burden of chronic diseases.
While growth is moderate, Europe remains a hub of clinical rigor, regulatory influence, and medtech innovation. Success in this market hinges on navigating complexity while addressing local needs. (See the Surgical Procedure Volumes forecast for Europe’s Big Five on the next page.)
Across all four topics tracked this month, we see medtech’s ongoing evolution toward:
• Platform integration over standalone tools
• Cross-border access with regional adaptation
• Consumer-centric, procedureagnostic design
• Disruption that balances safety with scalability
Whether it’s electrosurgical dominance shifting toward robotics, breakthrough technologies reshaping cardiac care, or APAC startups tackling entrenched barriers to access, the pulse of medtech is beating faster.
All data in this article is sourced from LSI’s Market Intelligence Platform, and recent additions of LSI’s The Numbers on LinkedIn and on the LSI website.
Access LSI’s continuously updated Market Intelligence Platform.
Scan QR code for details and to request a demo.


This month, to accompany our Innovator Spotlight on Solenic Medical, we detail the latest LSI Market Intelligence data on the global, Asian, European, and U.S. forecast volume for knee replacement procedures.
LSI’s Global Surgical Procedure Volumes (SPV) Tracker provides trusted coverage of 300+ diagnostic and therapeutic procedures across 37 countries. Scan QR code to request a demo:

This month, to accompany our Innovator Spotlight on Fluid Biomed, we detail the latest LSI Market Intelligence data on the global, Asian, European, and U.S. forecast volume for select neurovascular stroke procedures.
LSI’s Global Surgical Procedure Volumes (SPV) Tracker provides trusted coverage of 300+ diagnostic and therapeutic procedures across 37 countries. Scan QR code to request a demo:

LSI’s SPV Tracker includes these geographies: Argentina, Australia, Belgium, Brazil, Canada, Caribbean, Chile, China, Colombia, Costa Rica, Denmark, Dominican Republic, Finland, France, Germany, Guatemala, India, Italy, Japan, Malaysia, Mexico, Netherlands, New Zealand, Norway, Panama, Poland, Russia, Singapore, South Africa, South Korea, Spain, Sweden, Switzerland, Thailand, Turkey, United Kingdom, and the United States.

Let’s face it, we’re all tired of the pop-ups, clickbait, and ads.
That’s why we created The Lens — well-curated insights and coverage, from the analysts and industry insiders you trust.

$995.00

PRINT SUBSCRIPTION
$895 for your first year
Billed annually and includes 12 editions

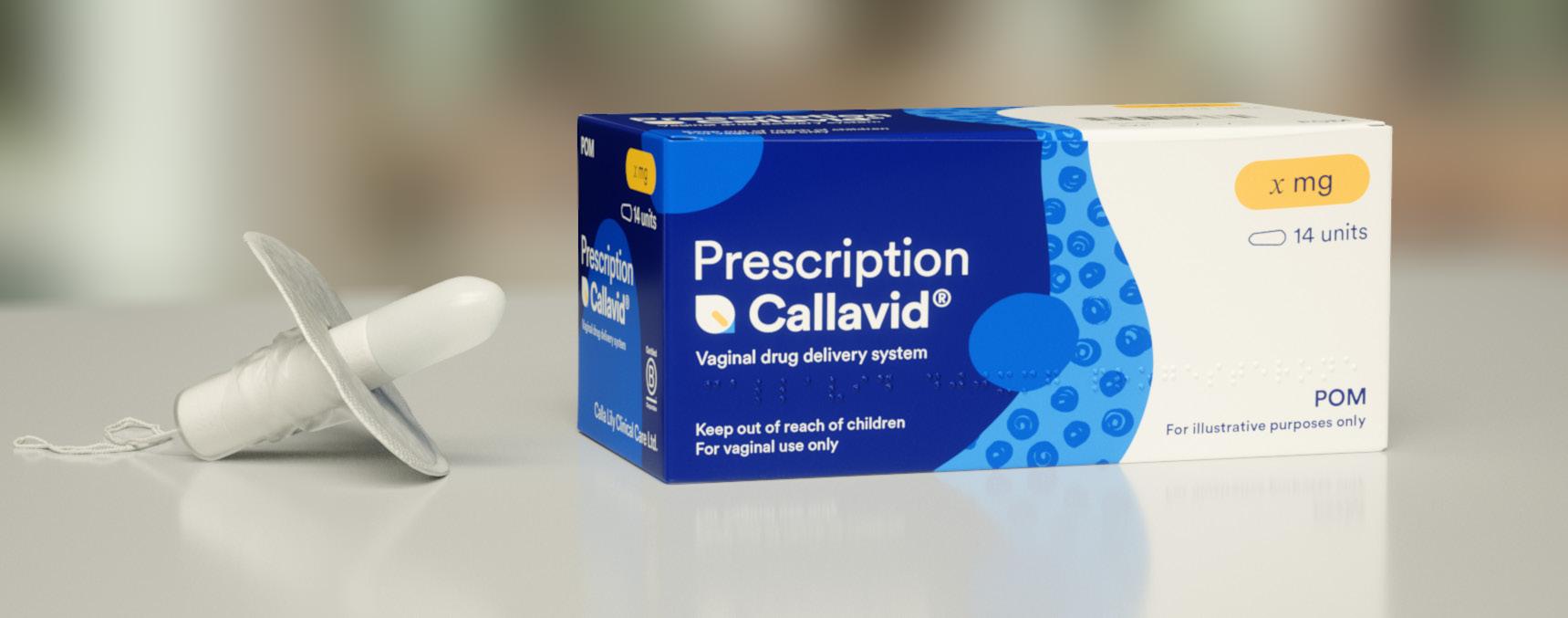
Calla Lily Clinical Care is challenging outdated norms in women’s health with Callavid, a first-of-its-kind vaginal drug delivery platform designed to replace painful daily injections and leaky pessaries. Co-founded by pediatric emergency physician-turned-entrepreneur Dr. Lara Zibners and private equity investor and medtech entrepreneur Thang Vo-Ta, the company is addressing the often-overlooked shortcomings of fertility and gynecologic care.
Dr. Lara Zibners didn’t set out to become a startup founder. As a board-certified pediatric emergency medicine specialist and trauma educator, she spent her career treating patients, not pitching to investors. But seven failed IVF rounds and the physical and emotional toll of progesterone treatment changed everything. “It was, by far, other than not getting pregnant, the most traumatic part of IVF,” Zibners said.
Her transition to entrepreneurship began when she was advising a com-
pany with a leak-free period care product, Tampliner. What intrigued her most was the underlying potential of the platform as a basis for vaginal drug delivery. “What I went through is very traumatic, but the possibilities of what we can do are so endless and amazing. I just believe.”
Her experience with painful daily intramuscular injections and leaky vaginal pessaries propelled her to team up with Thang Vo-Ta, a former Goldman Sachs private equity investor and serial entrepreneur who had previously developed and commercialized the Tampliner two-in-one tampon and mini panty liner combo. Together, they relaunched as Calla Lily Clinical Care, with a focus on the medical opportunity.
She describes her role with characteristic humility: “I call myself the Chief Cocktail Officer. I’m not the technical person … I bring the real, lived experience.” As she and Thang began their journey together, she started dreaming, literally, of a better way. “I started having dreams, nightmares actually, about injecting progesterone. I don’t want other women to go through that,” she said.
Across IVF and the prevention of threatened miscarriage and preterm birth, vaginal progesterone delivery offers a clinically sound, less invasive alternative to injections. But current
options come with serious drawbacks: leakage, anxiety over placement, uncertainty about receiving the full dosage, and emotional distress. “Vaginal drug delivery is a uniquely unaddressed area because women don’t want to talk about it and men don’t want to hear about it,” Zibners said.
She’s committed to changing that stigma head-on. “If nasal drugs leaked the way vaginal drugs did, that problem would have been solved years ago because men wouldn’t want stuff running down their face,” she quipped.
The company’s solution, Callavid, was designed to address every pain point in the standard of care. It provides leakage protection, placement and dosage confidence, comfort and convenience, mess-free insertion and removal, and reduced variability in drug absorption.
The initial focus is a 400mg progesterone Callavid for IVF luteal phase support and threatened miscarriage. What sets it apart is its user-centric design. “You want to get the right medication to the right spot in the right dose, without the systemic side effects,” said Zibners.
By eliminating leakage and building confidence in the treatment process, Callavid reduces a major source of stress and uncertainty during critical moments in care, offering not just clinical improvement but emotional relief.
Callavid isn’t a single product; it’s a platform. The technology behind Callavid is adaptable. Each drug has different physical properties and clinical requirements, and the platform can be tailored to meet them. Development

has started for Callavid to be used in dozens of future indications beyond IVF and miscarriage prevention, including:
• Prevention of preterm birth and preeclampsia
• Menopause hormone replacement therapy (HRT)
• Vaginal microbiome therapies
• Pre-cancerous cervical lesions, HPV infections, and endometriosis
• Other women’s health conditions, including vaginal candidiasis and vulvovaginal atrophy
“I see a world where eventually every woman takes vaginal progesterone at some point in her life. We’d like to be the company to make that possible,” Zibners said. “Progesterone is just the tip of the iceberg; there are so many other therapeutics that could and should be given vaginally.”
The initial market opportunity for Callavid spans $22+ billion, including:
• $2.7B for IVF and miscarriagerelated progesterone delivery
Source: Calla Lily Clinical Care
• $5.5B for preterm birth and preeclampsia prevention
• $14.7B for microbiome-related therapies
The company’s progress to date includes:
• $1.3M NIHR funding to prepare for and conduct a clinical feasibility study
• Confirmed approval pathway with MHRA (UK), with a pivotal 55-patient study to achieve the IVF and miscarriage prevention indications, demonstrating bioequivalence with an existing progesterone product
• FDA 510(k) clearance for the underlying consumer technology
• Manufacturing readiness, including proprietary automation in place
• Patents granted across Europe, the U.S., Canada, Japan, South Korea, Singapore, China, India, and Brazil
For Zibners, Calla Lily is more than a company. It’s a chance to prevent others from reliving her pain. “Women are willing to do almost anything to have a baby,” she said. “But they shouldn’t have to suffer to do it.”
And she’s not backing down.
“You need someone like me who’s willing to say the word vagina 50 times a day on LinkedIn to change the conversation,” she says.
Zibners isn’t just changing the conversation; she’s building the platform that will deliver the future of women’s health.

Armed with a joint-mimicking hydrogel and a track record of engineering success, Leo Smit is leading Hy2Care into a new era of orthopedic care, where restoring true cartilage is no longer a costly dream but a scalable solution. With IDE approval now in hand and CE mark submission underway, Hy2Care is preparing to enter pivotal markets in the U.S., Europe, and beyond.
For Leo Smit, the mission behind Hy2Care is personal. “My mother lived with chronic pain from arthritis until the end of her life,” he shared. “It’s deeply rewarding to create something that gives people their lives back.”
Smit brings decades of medtech experience to his role, including the creation of high-strength polyethylene fibers now used in implantable sutures across orthopedics. But his decision to join Hy2Care as CEO in 2019 was sparked by something more: a belief that cartilage repair could, and should,
be simpler, more affordable, and more widely accessible.
“It’s deeply rewarding to create something that gives people their lives back.”
That belief is shared by Dr. Sanne Both, Hy2Care’s co-founder and current Director of Clinical Applications. Before she turned 24, Both had undergone three failed cartilage surgeries and knew firsthand the shortcomings
of current treatments. Her decision to pursue a PhD in regenerative biology and develop a new solution led to the founding of Hy2Care in 2014 alongside Prof. Marcel Karperien. Smit and COO Sanna Severins, who had worked together previously at DSM Biomedical, joined the company during its early funding stages in 2019 to bring that solution to life.
“We don’t just have the science,” said Smit. “We have a team that knows how to deliver on time, on budget, and with heartfelt dedication, even through two and a half years of intermittent lockdowns during the COVID19 pandemic.”
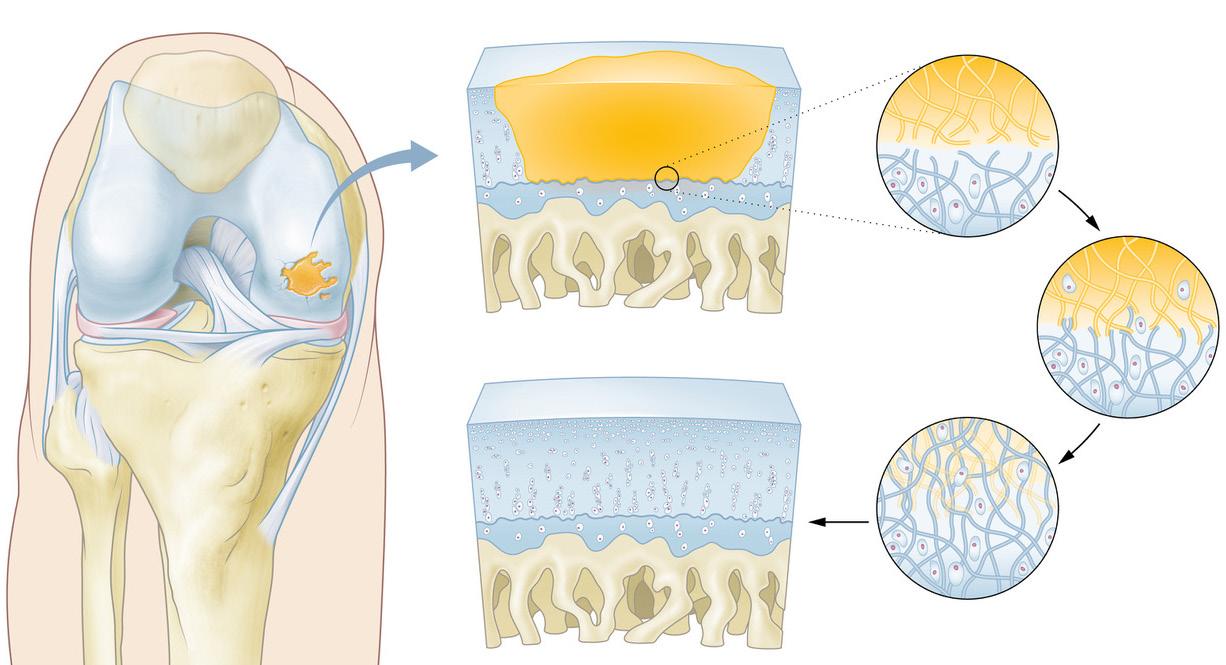
The company’s lead product, CartRevive, is a hydrogel implant designed to restore natural cartilage in focal knee defects. Unlike conventional implants that require cutting and fixation, CartRevive is delivered as a liquid that molds perfectly to the defect before solidifying into a tissue-integrating scaffold within 60 seconds.
A clinical illustration provided by Hy2Care (see below) shows this process in action. Smit explained, “When cartilage is damaged, it creates a defect in the smooth joint surface, leading to pain and reduced mobility. Our in-situ forming hydrogel precisely fills that defect, adheres to the surrounding tissue, and forms a protective matrix. This matrix supports the body’s natural healing process and creates an ideal environment for chondrocytes to migrate, proliferate, and build new, healthy cartilage while the gel resorbs naturally.”
The result is a seamless fit within the joint’s delicate architecture. “The knee is like a Swiss clock,” Smit explained. “Any mismatch throws it off. Because CartRevive molds exactly to the shape of the defect, the clockwork keeps running.”
That biological precision is paying off. In a European clinical trial, 46 patients have been treated with CartRevive. Results show significant improvement in KOOS scores, a measure of pain and mobility, reaching 2.5 times the threshold for clinical relevance after just 12 months. Patients are not only painfree, but many are returning to sports within a year.
At LSI Asia ’25, Smit shared the story of Bob, a 35-year-old patient who had been told to “learn to live with the pain” after an MRI indicated a severe cartilage injury. “After nine months with CartRevive, Bob was running again, and after a year, he was back to playing football and tennis,” Smit told the audience. “That’s what we want, for every patient.”
What sets CartRevive apart is its performance relative to cost. “We match
the tissue quality of cell therapies,” said Smit. “This efficiency comes from a fundamentally different approach, but at 15% of the cost. It’s a return to biology, made scalable.”
Hy2Care’s regulatory progress reflects its momentum. The company secured FDA Breakthrough Device Designation in 2023 and received IDE approval in April 2025 to begin its U.S. clinical trial. A CE mark submission is already underway in Europe, with market approval targeted for 2026.
“After nine months with CartRevive, Bob was running again, and after a year, he was back to playing football and tennis.”
In May 2025, Hy2Care closed a €4.5 million funding round, led by Brightlands Venture Partners and supported by the European Innovation Council Accelerator. This round fuels preparations for the U.S. trial and supports early commercial planning.
To fully fund the U.S. trial and global expansion, the company is currently raising €35 to €40 million, with an initial €15 million tranche targeted for completion this year.
“We’re a small team with global ambitions,” said Smit. “We’re looking for partners to help us scale, whether distributors or licensees. Our mission is to make this technology available to everyone who needs it.”
Hy2Care’s leadership isn’t just technically strong; it’s intentionally diverse. “We’re balanced across age, gender, nationality, and expertise,” said Smit. “You’ll find Baby Boomers and Gen Z working side by side here, and that’s not an accident. It’s a strength.”
That cohesion has helped the team hit every major milestone, despite launching just before the COVID-19 pandemic. “We’ve never missed a deadline,” said Smit. “That’s a testament to the people behind the product.”
Looking ahead, Hy2Care sees broader potential for its platform. Future applications could include larger defects, patellar defects, and eventually other joints and orthopedic indications.
Cartilage Repair with CartRevive
Source: Hy2Care website
Celebrating the accomplishments of a few of the many bright stars in our LSI presenting company and speaker alumni community.
Be sure to check out presentations from these and many other innovative companies from recent LSI events on our website at Resources > Video Library.



Accelus has received 510(k) clearance from the FDA confirming that its FlareHawk Interbody Fusion System can be safely subjected to MRI scanning in certain conditions. The clearance follows extensive ASTM-standard testing, which evaluated magnetic field interactions, radiofrequency-induced heating, and image artifacts. MRI compatibility addresses a critical clinical need, giving both patients and surgeons added confidence when selecting spinal implants.
The implants feature a small insertion profile that minimizes neural re-
traction during placement, followed by multidirectional expansion in width, height, and lordosis. More than 32,000 FlareHawk cages have been implanted across 25 countries, reflecting its clinical adoption. With a growing portfolio that includes the LineSider Spinal System and the Toro-L Interbody Fusion System, Accelus continues to drive innovation in expandable spinal technologies for both hospital and ASC environments.

AliveCor has partnered with the British Heart Foundation in a yearlong initiative to raise funds for cardio-
vascular research through a causerelated marketing campaign. For every KardiaMobile 6L device sold in the UK through May 2026, AliveCor will donate £3 to support the charity’s mission of diagnosing, treating, and preventing cardiovascular disease. This collaboration aligns two organizations with shared goals: empowering individuals to take charge of their heart health while accelerating research that could save lives.
KardiaMobile devices, which have recorded more than 300 million ECGs globally, offer clinically validated, AIpowered arrhythmia detection from the convenience of a smartphone. Already recommended by NICE for use within the NHS, the technology enables early detection of conditions like atrial fibrillation and helps bridge gaps in care. With the British Heart Foundation funding over £100 million in cardiovascular research annually, this partnership represents a united effort to improve cardiac outcomes both today and in the future.

Atmo Biosciences has received 510(k) clearance from the FDA for its Atmo Gas Capsule System, a first-of-itskind ingestible diagnostic designed to aid in the evaluation of gut motility disorders. This clearance allows the company to market the capsule in the U.S. for use in assessing regional and whole gut transit times in patients with conditions like gastroparesis and slow transit constipation, chronic disorders that often go undiagnosed or misdiagnosed for years. The device offers a patient-friendly, radiation-free alternative to traditional diagnostic tools such as gastric scintigraphy and radio-opaque marker studies.
The decision was backed by a 209patient pivotal clinical study across 12 sites, which demonstrated equivalence to the predicate SmartPill device in measuring gastric emptying and colonic transit times. With a commercial launch in the U.S. set for July 2025, Atmo aims to bring its non-invasive solution to a broader clinical audience. According to CEO Malcolm Hebblewhite, the capsule provides “comprehensive, clinically valuable information” that empowers clinicians to diagnose and manage gut motility disorders more efficiently and accurately.

Brainomix has partnered with 3DR Labs to bring its FDA-cleared Brainomix 360 Stroke platform to U.S. hospitals,
advancing stroke care through AI-powered imaging and clinical workflow solutions. Brainomix 360 is the only stroke AI tool clinically validated to increase mechanical thrombectomy rates, with real-world data showing a greater than 50% boost in intervention where the platform is adopted. By integrating Brainomix into its AI Labs platform, 3DR Labs aims to streamline access to actionable imaging insights, reducing time-to-treatment and improving patient outcomes.
3DR Labs supports radiology and neurology teams with advanced postprocessing and AI-integrated workflows, backed by more than 250 certified radiologic technologists and proprietary stroke-specific certifications. This collaboration will provide health systems with a turnkey solution for stroke triage and decision-making, enhancing diagnostic precision and enabling earlier intervention. The partnership marks a major step forward in making high-impact stroke imaging tools more accessible across the U.S.

Cognito Therapeutics has released promising new data from its OVERTURE study, highlighting the ability of its Spectris AD device to slow Alzheimer’s progression through daily, non-invasive neuromodulation. The Spectris system uses gamma frequency light and sound stimulation to evoke EEG-confirmed neural oscillations disrupted by Alzheimer’s disease. In the six-month randomized phase of the study, patients experienced up to 4.83 months of preserved daily function, 4.56 months of delayed cognitive decline, and 4.09 months of delayed whole-brain atrophy compared to sham controls.
The benefits became even more apparent during the 12-month openlabel extension, reinforcing the potential of Spectris AD to meaningfully delay disease progression and improve quality of
life. The at-home device has received FDA Breakthrough Device designation and represents a new, physics-based approach to treating central nervous system diseases. Cognito’s CEO, Christian Howell, emphasizes that restoring the brain’s natural rhythms through neuromodulation may offer a powerful alternative to traditional pharmacologic therapies for Alzheimer’s patients.

Clairity has made history with FDA De Novo authorization for CLAIRITY BREAST, the first AI-powered platform that predicts a woman’s five-year risk of developing breast cancer based solely on a routine screening mammogram. Trained on millions of images and validated in over 77,000 mammograms across diverse populations, CLAIRITY BREAST analyzes subtle pixel-level patterns invisible to the human eye, delivering a personalized risk score to clinicians through existing infrastructure. This milestone marks a major advance for precision prevention and equitable access in women’s health.
Unlike traditional risk models that rely heavily on family history and demographic data, often missing the majority of women who develop breast cancer, CLAIRITY BREAST brings a validated, image-based approach to stratify risk earlier and more accurately. The platform is designed to scale seamlessly across hospitals and screening centers, enabling providers to tailor follow-up care and screening pathways before cancer even appears. Clairity’s groundbreaking technology represents a shift from reactive treatment to proactive, AI-enabled prevention.
Endovascular Engineering has completed enrollment in the pivotal cohort of its ENGULF IDE study evaluating

the Hēlo PE Thrombectomy System, a novel device for removing life-threatening blood clots in the lungs. Conducted at 19 leading U.S. centers, the ENGULF trial follows a successful feasibility study and aims to demonstrate the safety and efficacy of its dual-action clot removal system, which combines aspiration with mechanical disruption. The system’s Flow Mitigation Technology is designed to optimize clot clearance while minimizing blood loss, a key safety consideration in treating pulmonary embolism (PE).
PE remains a leading cause of cardiovascular mortality, with treatment options often constrained by trade-offs between effectiveness and safety. Endovascular Engineering’s Hēlo platform seeks to address these limitations, and the company anticipates releasing results from the pivotal trial later this year. With this milestone, the company moves closer to making its investigational technology available to patients across the U.S., supported by strong clinical collaboration and growing interest in next-generation PE interventions.

FineHeart has received authorization from the French National Agency for the Safety of Medicines and Health Products (ANSM) to initiate a first-inhuman clinical trial of its fully implantable cardiac output accelerator, FlowMaker. This prospective, non-randomized study will enroll patients with
advanced heart failure at specialized French cardiac surgery centers to evaluate the safety, feasibility, and early clinical performance of the device. The study builds on successful initial implants completed in 2024 in Prague and marks a major step forward in FineHeart’s clinical and regulatory roadmap.
FlowMaker is a next-generation left ventricular assist device (LVAD) designed to work in synchrony with the heart’s natural rhythm. Unlike traditional LVADs, it is fully intraventricular, operates without an aortic bypass, and does not require any percutaneous connection to external batteries. Instead, the device is recharged via transcutaneous energy transfer, reducing infection risk and improving patient comfort. Minimally invasive to implant and adaptable to disease progression, FlowMaker could benefit over 200,000 patients with advanced heart failure annually.

GI Windows Surgical has received FDA 510(k) clearance for its Flexagon self-forming magnet technology, a sutureless, staple-free solution for creating anastomoses using magnetic compression. The laparoscopic and endoscopic delivery system enables a minimally invasive approach to surgical tissue fusion that leaves no permanent implants behind. According to the company, the Flexagon platform represents a major advance in anastomotic technology, achieving strong clinical outcomes without leaks, bleeds, or obstructions attributed to the procedure.
The company integrates laparoscopic, endoscopic, and robotic capabilities to streamline anastomosis, reduce operating time, and minimize complications. With this regulatory milestone, GI Windows is now poised to bring its magnetic platform to more surgeons and patients in need of less
invasive solutions. Backed by $37 million in funding raised last fall, the company is entering its next phase of growth as it works to standardize and expand the use of magnet-based surgical technologies.

InspireMD has received CE Mark approval under the EU MDR for its next-generation CGuard Prime carotid stent system, designed to prevent stroke in patients with narrowed carotid arteries. Built on the foundation of the original CGuard platform, the new system features improved deliverability and deployment, along with the company’s proprietary MicroNet mesh technology that traps embolic debris and minimizes plaque prolapse.
The company developed CGuard Prime for patients at elevated surgical risk who require a less invasive treatment option. The device’s open-cell stent frame and fine-pore mesh offer both embolic protection during and after the procedure, with clinical evidence demonstrating durable performance and stroke prevention for more than five years. InspireMD plans to bring the platform to the U.S. pending FDA approval expected later this year.

Neurent Medical has received FDA 510(k) clearance for its next-generation
NEUROMARK System, a non-surgical radiofrequency platform designed to treat symptoms of chronic rhinitis by targeting overactive posterior nasal nerves. The upgraded system enhances procedural precision by providing real-time feedback, guiding electrode placement, and confirming treatment delivery, empowering ENTs to treat symptoms like congestion and rhinorrhea with greater accuracy and confidence.
The device’s flexible shaft and atraumatic leaflets conform to varied nasal anatomies, improving access to challenging regions while maximizing treatment coverage. Following a successful commercial validation phase in collaboration with ENT leaders across private and academic settings, Neurent is preparing for broader U.S. rollout. Chronic rhinitis affects nearly one in four Americans, and the company’s latest milestone marks a significant step in reshaping care standards through minimally invasive innovation.

Neuspera Medical has received FDA approval for its integrated sacral neuromodulation (iSNM) system, the first of its kind to treat urinary urge incontinence (UUI) without the need for implanted batteries. The miniaturized neurostimulator is paired with a wearable external disc, offering the therapeutic benefits of traditional SNM with fewer complications and improved patient comfort. In a 128-patient Phase II pivotal trial, 84.2% of patients experienced a ≥50% reduction in UUI episodes, with 42% of responders achieving complete dryness.
This approval introduces a new standard in UUI care, offering a discreet, patient-friendly option for millions affected by overactive bladder symptoms. By eliminating the surgical burden of battery replacements and enabling wireless daily therapy, Neuspera’s iSNM
system delivers clinically meaningful results with fewer trade-offs. The company is now positioned to advance neuromodulation through smarter, more adaptable solutions that enhance both patient outcomes and quality of life.

Novadip Biosciences and Cliniques universitaires Saint-Luc have announced positive results in four pediatric patients with congenital pseudarthrosis of the tibia (CPT) treated under Belgium’s compassionate use programs. Using NVD003, Novadip’s autologous bone graft material, all four children achieved complete fracture healing with no recurrence over four to seven years of follow-up. The results mark a significant step forward in treating CPT, a rare and debilitating disease that can result in amputation due to non-healing fractures.
NVD003 is a scaffold-free, tissue-engineered bone implant developed from the patient’s own stem cells and tailored to restore lost bone volume. Following these early successes, the company is planning a phase 3 clinical trial in CPT. The company aims to bring NVD003 to market by 2027, offering a durable, regenerative solution for children with few other treatment options.

OSSIO has expanded its OSSIOfiber platform with the launch of 2.5mm
suture anchors designed to improve soft tissue fixation in procedures like Brostrom repair for lateral ankle instability. The new anchors are smaller, stronger, and metal-free, offering 55% greater pull-out strength than the market-leading 2.4mm bio-composite suture anchor. The OSSIOfiber material enables implants to integrate into bone while eliminating the longterm complications of metal fixation. With configuration options and familiar instrumentation, the new system supports procedural flexibility and broad clinical adoption.
The OSSIOfiber suture anchor is indicated for use across a range of orthopedic procedures involving the foot, ankle, knee, shoulder, hand, wrist, and elbow in both adults and children (2–12 years). Backed by Intelligent Bone Regeneration Technology, OSSIOfiber implants combine mechanical strength with natural healing in a fully bio-integrative design. More than 50,000 OSSIOfiber implants have been used in the U.S. to date, demonstrating growing surgeon demand for strong, absorbable alternatives to traditional metal fixation.

Paradromics has successfully implanted its Connexus Brain-Computer Interface (BCI) in a human for the first time, marking a major milestone for the neurotech startup. The procedure took place during epilepsy surgery at the University of Michigan and demonstrated the device’s ability to be safely implanted and record neural activity. Removed after 20 minutes, the implant confirmed the company’s data-driven confidence in its human compatibility and paves the way for a clinical trial later this year.
Founded in 2015, Paradromics aims to enable communication for people with severe motor impairments (e.g., paralysis) by translating brain signals into digital speech. Its BCI system is
designed to record activity at the level of individual neurons, offering granularity that sets it apart from other BCI platforms. With nearly $100 million in funding and a recent strategic partnership with Saudi Arabia’s Neom, Paradromics now enters the clinical stage of development, joining the ranks of Neuralink, Synchron, and Precision Neuroscience in the competitive BCI space.

STENTiT has successfully enrolled the first patient in its VITAL-IT 1 clinical trial, evaluating a novel fully bioresorbable stent designed to treat chronic limb-threatening ischemia (CLTI) below the knee. The company’s Resorbable Fibrillated Scaffold (RFS) System was implanted at the Medical University of Graz, Austria, in a procedure that
restored blood flow to the foot using an endovascular approach. The scaffold provides temporary structural support while promoting natural healing, as its porous microfiber design enables the patient’s own cells to regenerate vascular tissue and gradually replace the implant.
The single-center, prospective, nonrandomized feasibility study (NCT07006467) will enroll up to 10 patients and monitor them over 24 months. CLTI is a severe stage of peripheral artery disease that affects an estimated 3.5 million people across Europe and the U.S. and results in over 250,000 amputations annually. This milestone marks STENTiT’s transition into clinical-stage development, backed by the European Innovation Council and a 2025 MedTech Innovator cohort selection.
TYBR Health has received FDA 510(k) clearance for its B3 GEL System, a bioresorbable, flowable gel barrier designed to protect soft tissue planes during the early stages of healing after musculoskeletal surgery. Composed of a naturally derived extracellular matrix, B3 GEL is delivered via TYBR’s proprietary integrated mixer-applicator,

conforming to complex anatomy in both open and minimally invasive procedures. The gel temporarily separates tissues to reduce binding and preserve mobility, then fully resorbs without leaving behind bulk or interfering with the body’s natural healing process.
The clearance marks a major milestone for the company, positioning B3 GEL as a novel solution in surgeries involving tendons, ligaments, and skeletal muscle. Preclinical data demonstrated improved range of motion and reduced tissue binding in treated models, supporting the product’s potential to enhance functional recovery. TYBR Health plans to begin commercial sales by the end of 2025, addressing a significant unmet need in musculoskeletal procedures where post-op complications can limit outcomes and drive up costs.


Atmo

March 16th - 20th, 2026
Waldorf Astoria, Monarch Beach, Dana Point, CA


