The Rich History and Bold Future of Intuitive: Inside Gary Guthart’s Vision for Robotics and Healthcare

Predictable Winners, Hot Takes, All Eyes on Ophthalmology, and more...

The Rich History and Bold Future of Intuitive: Inside Gary Guthart’s Vision for Robotics and Healthcare

Predictable Winners, Hot Takes, All Eyes on Ophthalmology, and more...
February 2025
April 2025
June 10th - 13th, 2025
The Ritz-Carlton, Millenia Singapore
With the global community built through LSI USA and LSI Europe and our partnerships in Singapore, China, Korea, Japan and more, LSI Asia will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Like LSI USA and LSI Europe, LSI Asia will not be held in a chaotic convention center. We have identified a world-class venue in Singapore, the gateway to Southeast Asia, for our inaugural event where senior executives can do real business.



Thank you to our founding sponsors
As we launch in this new geography, we are offering a unique opportunity to a small set of value-add, mission-aligned partners looking to elevate their global presence and develop new business.

Scott Pantel
Chief Executive Officer, Editor-in-Chief

Hopkins
Sr. Content Manager, Contributing Author

Tomlin
Contributing Photographer

Henry Peck
Chief Business Officer, Editor-in-Chief

Matrone
Sr. Marketing Manager, Contributing Author

Kristin Karkoska
Contributing Photographer

Tracy Schaaf
Managing Editor and Content Strategist, Lead Author


Nicholas Talamantes
Sr. Director of Market Intelligence, Contributing Author

Customer Service & Fulfillment

Paul Streeto Graphic Designer
(LSI)
714 847 3540 tel/fax email: info@ls-intel.com
Vol. 2, No. 4 April 2025

Rebekah Murrietta
Vice President of Media, Contributing Author

Kelly Williams
Subscriptions & Enterprise Sales
Created by LSI, The Lens leverages LSI’s deep industry relationships and proprietary market intelligence to help executives like you build lasting medtech companies. Start your Individual, Group or Enterprise-Wide subscription today!

Scan QR Code to activate your subscription.
The Lens is published monthly and available in print and electronic formats. Copyright 2025 by Life Science Intelligence, Inc. All rights reserved. Editor takes care to report information from reliable sources and does not assume liability for information published. Interested in subscribing? email to: info@ls-intel.com Interested in advertising? email to: kelly@ls-intel.com


Dear Readers,
We find ourselves in a moment where the only certainty is change. Shifting policy, evolving tensions, and economic undercurrents are shaping the future of science, medtech, and global healthcare in real time. The stakes have never been higher.
In moments like this, trusted insight and enduring relationships matter more than ever. For us and our industry, LSI Asia ’25 couldn’t be launching at a better time (albeit, a challenging time). This June, we’ll bring together experts and executives from across Asia Pacific, including Singapore, China, Japan, Korea, Australia, and beyond, and our strong U.S. and European community to partner. The conversations happening at LSI Asia will reflect the complexity of our global moment, and the opportunity within it.
We’ll continue to monitor and analyze these trends, and share the moments of clarity with you, right here in The Lens.
One such moment of clarity came on March 20th in the Keynote delivered by Gary S. Guthart, PhD, CEO and Member of the Board at Intuitive at LSI USA ‘25. From his early days working with surgical robotics prototypes as the company’s 11th employee in the 1990s, to now leading Intuitive through the successful launch of its da Vinci 5 system and new era of surgical data science, Dr. Guthart has one of the most refined and unique perspectives on our industry.
In addition to his insights on robotics, imaging, and artificial intelligence, Dr. Guthart issued a powerful reminder that success stems not just from innovation, but from a strong culture of engaged, shoulder-to-shoulder leadership, clarity of mission, and a dedicated team. Intuitive built a leadership culture deeply rooted in a shared purpose of improving patient outcomes, and this cultural “magic dust,” as Dr. Guthart calls it, remains a pillar of the company’s strength. Their focus is not just on what has been done, but what remains to be done.
Alongside Dr. Guthart’s insights, this edition of The Lens features updated deals and financings in the industry, powered by Compass – LSI’s proprietary discovery engine for private companies, deals, and market opportunities in medtech. With noise and untrustworthy data bombarding us at every turn, our analyst team is committed to delivering trusted, vetted, up-to-date coverage of innovation and investment in our industry.
We also explore the Ophthalmology space, on the heels of Alcon, the largest eye care device company in the world, announcing its plans to acquire the fast-rising robotic cataract surgery innovator LENSAR in a deal worth up to $430M. It’s a bold move that signals further market consolidation, and a window into what the future of eye surgery may hold.
With this edition of The Lens, we invite you to take stock of your own mission, your team, and the impact you strive to create. Honor it. Share it. And then, as Dr. Guthart puts it, ask yourself “what remains to be done?”
We’re honored to be on this journey with you.
All the best,
Scott Pantel Chief Executive Officer, LSI Editor-in-Chief


Henry Peck Chief Business Officer, LSI Editor-in-Chief

Need fresh inspiration for your medtech company as we navigate 1H25? We’ve gathered insights from some of our LSI alumni thought leaders to help you stay ahead.
“Elon Musk’s Neuralink created unrealistic market valuations larger than entire established neuromodulation markets combined.”

— Bálint Várkuti, CEO & Co-Founder, CereGate
LSI Europe ‘24 Panel: Neuromodulation: Hurdles, Ethics, and Unmet Needs Driving Innovation and Investment
“If you’re in startup land and you’re not having one near-death experience a year, you’re not trying hard enough.”

— Colin Morrison, Senior Director of Venture Capital, Boston Scientific
LSI Europe ‘24 Panel: What Matters to Corporate VCs in 2024?
“Nothing good happens in five years. You want to get something really good to happen? It takes 10, if not 15 years.”

— Joe Kiani, Founder & Executive Chairman, Willow Labs, Founder, Masimo
LSI USA ‘25: Keynote with Joe Kiani, the Founder, Chairman, CEO of Willow Labs, and Founder of Masimo
“Sometimes having the wrong investor is worse than having no investor.”

— Ashley Seehusen, Venture Partner, Santé Ventures, CEO, Santé Accel
LSI Europe ‘24 Panel: Training & Supporting the Next Wave of Entrepreneurs Through Mentorship and Capital
“What I don’t want to see are five pivots over the course of a couple of years… I do think that speaks to inexperience.”

— Anita Watkins, Managing Director, Rex Health Ventures
LSI Europe ‘24 Panel: Light at the End of the Tunnel in Medtech
“Being a CEO is very different from being a surgeon. In surgery, I taught. As a CEO, I had to learn to ask questions and listen.”

“Better to be lucky sometimes than good.”
— Bruce Ramshaw, CMIO, Caresyntax
LSI Europe ‘24 Panel: Caresyntax: $180M to Accelerate the Future of AI and Precision Surgery
“People who are going to be successful are the ones who are willing to pay the price and do the hard work…. and it’s worth it.”

— Spencer Stiles, Group President, Orthopaedics, Stryker
LSI USA ‘25 Signature Series: To Build or To Buy? Stryker’s Thesis for AI, Robotics, and Growth

— Ray Cohen, Chairman of the Board, SoniVie, Former CEO, Axonics
LSI USA ‘25 Signature Series: Building and Exiting Companies
Celebrating recent leadership shifts and other announcements and accolades impacting our global medtech community.
LSI alumni AccuLine has appointed Amit Reches, PhD as its new Chief Technology Officer, to advance its non-invasive cardiovascular diagnostics platform. Dr. Reches brings over 25

years of expertise in medical devices and technology, including leadership roles at Nuvo and Firefly Neuroscience, with proven experience in developing and commercializing FDA- and CE-approved products.
LSI alumni Accuryn Medical has appointed Todd Dunn as Chief Executive Officer, to lead the company’s next

phase of growth and innovation in acute kidney injury predictive health. Dunn, previously Accuryn’s Chief Transformation Officer, brings extensive leadership experience from roles at Atrium Health, GE Healthcare, Cisco, Siemens, and McKesson.
Laguna Hills, CA-based Adagio Medical, an innovator in catheter ablation technologies for the treatment of cardiac arrhythmias, announced the appointment of Deborah Kaster as Chief Business Officer. Kaster brings more than 25 years of leadership experience in strategy, mergers and

acquisitions, corporate finance, investor relations, and strategic communications in the medtech sector. Most recently, Kaster served as Vice President of Investor Relations at Shockwave Medical, where she played a pivotal role in the company’s growth from IPO to its acquisition by Johnson & Johnson. As Director of Business Development at Kyphon, she led the company in a competitive bidding process for St. Francis Medical Technologies, which resulted in Kyphon’s successful $725 million acquisition of St. Francis and, almost immediately subsequent, the ultimate $4 billion acquisition of Kyphon by Medtronic.
French medical technology company Affluent Medical has recently strengthened its leadership team with the appointments of Prof. Howard Herrmann and Prof. Nicolas Barry Delongchamps as Strategic Chief Medical Officers in structural heart and urology, respectively. Additionally, Federica Azzimonti joined as Director of Clinical Operations, bringing 25 years of experience in managing international clinical studies. The company also added Liane Teplitsky, CEO of autonomous mechanical thrombectomy robotics company Artedrone, as a new Board member. Affluent Medical, founded by Truffle Capital, aims to become a global leader in the treatment of structural heart diseases and urinary incontinence.
CorWave, a French company developing innovative cardiac assist devices, has appointed Anne Reiser to its Board

of Directors as the representative of the EIC Fund (the venture investment arm of the European Innovation Council). Reiser’s leadership in heart failure therapies and medtech growth will help drive CorWave’s mission to bring its next-generation LVAD technology to patients. A member of the French Tech 120, CorWave was founded in 2012 by startup studio MD Start and is funded by investors including Bpifrance, Exor Ventures, EIC Fund, Financière Arbevel, M&L Healthcare, Novo Holdings, Seventure Partners, Sofinnova Partners, Ysios Capital, and Vlerick Group
Curespec, a company pioneering an innovative, non-invasive solution for patients with hypertension and chronic kidney disease, has appointed LSI alumni Avner Geva to its Board of Directors. Avner is the current co-founder and

CEO of Vensica Medical, a company that is developing the Vibe—a catheter that unlocks the power of therapeutic ultrasound to deliver drugs to the bladder wall.
Geva is also co-founder and a Board Member of the oncology startup, IntraGel Therapeutics, and Entrepreneur
in Residence and biotech principal at Yale Ventures. Curespec’s CE-marked Nephrospec system uses patented, electro-hydraulic acoustic therapy (eHAT) to improve kidney function, reduce blood pressure, and delay dialysis—addressing a major unmet need for millions of patients.
Dexcom, the global leader in glucose biosensing, has announced the appointment of Jon Coleman as Chief

Commercial Officer. In this role, Coleman will assume responsibility for Dexcom’s global commercial organization, including global sales, marketing, and customer experience. He joins Dexcom with more than 30 years of global commercial leadership experience across multiple healthcare segments and channels, including serving as an executive officer of Masimo, where he held roles of increasing responsibility across his 15-year tenure. He also held significant roles at Pfizer’s Consumer Healthcare business.
Dexcom also announced the appointment of Renée Galá to its Board of Directors, effective March 6. Galá is an

accomplished executive with significant financial, strategic, and operational leadership experience across the life sciences sector. She currently serves as President and Chief Operating Officer of Jazz Pharmaceuticals PLC, a global biopharmaceutical company with greater than $4 billion in total 2024 revenues. She brings to the Dexcom Board a strong track record of strategic
and financial leadership at high-growth companies, including oversight of key international expansion initiatives and business development collaborations.
Irvine, CA-based Diality, a device company that has developed the Modaflx Hemodialysis System, recently announced the addition of Dr. Chris Springer to its Board of Directors.

Springer brings 30 years of commercial and strategic experience in the pharmaceutical and biotech industries to Diality. As Chief Strategy Officer for Vifor Pharma, he created a joint renal-drug company between Vifor and Fresenius Medical Care as well as oversaw a series of licensing deals and acquisitions, culminating in the acquisition of Vifor Pharma by Australian biopharma CSL for $11.7 billion. Prior to Vifor Pharma, Springer also served in executive roles with Amgen. He sits on the board of multiple biotech firms, including Adrenomed, Nagi Bioscience, Quercis Pharma and HICT
Elucid, an AI medical technology company focused on providing physicians with a more precise view of atherosclerosis to drive patient-specific therapeutic decisions, announced that it has added several senior members to its clinical and commercial teams.
Amir Ahmadi, MD, has joined Elucid as a Chief Scientific Advisor and will lead the clinical strategy of the company. Dr. Ahmadi is a cardiologist with a passion for advancing cardiovascular care

through precision medicine, innovative research, and clinical excellence. He is a Clinical Associate Professor of Medicine (Cardiology) at the Icahn School of Medicine at Mount Sinai in New York where he serves as Director of Inpatient Cardiology and Co-Director of the Cardiac Intensive Care Unit at Mount Sinai Morningside.
Carol Holt, Elucid’s Vice President of Clinical Development, is an accomplished and multifaceted life sciences

executive with deep expertise in clinical trial management, professional clinical education, and business development for medical technologies including cardiovascular (heart failure) and interventional cardiology. Prior to joining Elucid, Holt served as Vice President of Clinical Affairs for Ventric Health, formerly Avicena. She also held roles at Cerus Endovascular, which was acquired by Stryker Neurovascular in 2023, and Uptake Medical, which was acquired by Broncus in 2016. Before Holt’s startup experience, she spent 14 years with Edwards Lifesciences and seven years in CV ICU at the Texas Heart Institute.
Kevin Mathews has joined Elucid’s leadership team as Senior Vice President of Marketing. Kevin is an experienced global commercial leader in

the device space with over 25 years of experience in segments including interventional cardiology, peripheral vascular, electrophysiology, and medicalsurgical. Prior to joining Elucid, Mathews was the Global Commercial Leader for Acutus, an arrhythmia mapping and
ablation company. Before Acutus, he held leadership roles in sales, marketing, and business development at Philips IGT Devices, a division created through Philips’ acquisitions of Volcano Corp. and Spectranetics. He also held leadership roles at Jomed, DeRoyal, and in KPMG’s healthcare practice.
Amy Pyke has joined Elucid as Senior Vice President of Market Access. Pyke has 30 years of experience devising and implementing strategies and tactics to secure favorable reimbursement and coverage with commercial and government payors in medical device and pharmaceutical organizations across multiple therapeutic areas. Prior to joining Elucid, she was head of Market Access and Reimbursement for IGTD & Systems at Philips. Previously, she led Stryker’s Instruments Market Access and Reimbursement Department and held a variety of market access and reimbursement positions at Edwards Lifesciences, GE Healthcare, and Alcon.
Endologix has appointed Andrew Davis as Chief Commercial Officer, to lead its global market expansion

and commercial strategy. With over 25 years in vascular and cardiovascular device sales, Davis will drive revenue growth, customer engagement, and strategic business development. Most recently, he served as the Chief Commercial Officer at Silk Road Medical where he successfully spearheaded the go-to-market strategy for a groundbreaking therapy to treat carotid artery disease. Previously, Davis was the Vice President of Sales and Marketing at 3M’s Advanced Wound Therapy Group, in addition to spending 15 years at Medtronic in various Vice President of Sales positions in Cardiac and Vascular Therapies to include TAVR, Endovascular EVAR/TEVAR, Peripheral Vascular, and Spine/Biologics.
Intuitive Ventures announced that it has appointed Terri Burke as Senior Partner, and Ross Jaffe, MD as Venture Advisor, to expand its investment

platform in minimally invasive care. With extensive experience in medtech investing and healthcare innovation, they will help scale Intuitive Ventures’ $250 million portfolio.
Most recently, Burke was a Venture Partner and Head of Medtech Investments at Epidarex Capital, where she led the firm’s Fund III US/UK investment strategy, deal flow and medtech brand. Burke also previously co-founded and served as CEO at Protaryx Medical, a pioneering company enhancing access and procedural efficiency in structural heart and electrophysiology interventions. Previously, she served in various senior leadership roles at Edwards Lifesciences, including Vice President of Global Marketing and Business Development, where she streamlined and re-focused the heart valve business to help drive growth.
Dr. Jaffe was one of the founders of Versant Ventures, where he remains a Legacy Managing Director, and was previously a General Partner at Brentwood Venture Capital, where he started his career as one of the first physicians in the venture capital industry. After stepping back from active investing for Versant, Jaffe also served as a Venture Advisor for New Enterprise Associates. He is currently a co-founder and Board Chair of Faro Health, a clinical trial software company.

Life Seal Vascular has appointed Dr. Matt Thompson as CEO, leveraging his decades of experience as a vascular surgeon and medtech executive.

Thompson will lead the development and commercialization of the company’s endovascular solutions to prevent Type II endoleaks, a key challenge in aneurysm repair procedures. He previously held key leadership roles at Endologix, serving as Chief Medical Officer (CMO) and Chief Executive Officer. He currently continues to contribute to Endologix as CMO. Thompson also served as a leading vascular surgeon at St. George’s Hospital in London, UK, where he gained international recognition for his expertise in complex aortic repair.
LSI alumni company Lungpacer Medical has appointed Kevin Kearney as Chief Commercial Officer, to drive commercial growth and expand

access to its AeroPace System for ventilator weaning. With 20+ years of experience in commercial leadership, including a previous role as Vice President of Account Management Sales at Ceribell, where he led commercial strategy and significantly contributed to its growth in neurology and critical care. Kearney will also support the company’s clinical expansion of the investigational AeroNova System, aimed at mitigating multi-organ ventilator-induced injury.
LSI alumni organization MedtechWOMEN recently appointed Yvonne Bokelman, MBA, FACHE, as its new

Executive Director, to lead its mission of empowering and elevating women in the medtech industry. With leadership experience at Zimmer Biomet, Medtronic, and Alyve Medical, Bokelman’s passion for mentorship and advocacy will drive MedtechWOMEN’s growth and impact across the industry. She has also served as Chair of AdvaMed’s Payment & Policy Work Group and Co-Chair of the AdvaMed Orthopedic Work Group, influencing policies that drive industry-wide progress. Bokelman also brings extensive nonprofit and board experience, having served on multiple boards, and currently is Chairperson of the Board for PK MED, a French biotech company.
Ossiform has appointed LSI alumni Monica Wellejus as Chief Commercial Officer to lead the global strategy for its

3D-printed bone implant solutions. With experience in musculoskeletal innovation and medtech strategy, Wellejus will oversee all commercial and go-to-market activities.
Relief, a portfolio company of Panakes Partners that is developing a first-in-class device for the treatment of moderate to severe stress urinary incontinence, has appointed Michael Kujak and Steffen Hovard to its Board of Directors, and established a Medical Advisory Board led by Dr. Dean Elterman, with members including Enrico Finazzi Agrò, Roger Dmochowski, and Silvia Secco.


Kujak is a highly experienced executive with over 30 years in the medical device and urology industry. He is currently President and CEO of Francis Medical, where he has played a key role in securing a $80 million Series C funding round, having been the Chief Marketing Officer at NxThera (acquired by Boston Scientific) and General Manager at AMS (also acquired by Boston Scientific).
Hovard has over 25 years of leadership experience in the medical device, continence, and urology industry, having held leadership roles including CEO at Neuspera Medical, Chairman of UroMems and President of Coloplast Interventional Urology. He currently serves as Board Director at Butterfly Medical
Sense Neuro Diagnostics, an LSI alumni company developing non-invasive brain scanners that enable faster detection, triage, and continuous monitoring of brain injury to improve patient

outcomes, announced the appointment of Gary Kurdziel as Chief Executive Officer, effective April 1. He succeeds Geoff Klass, who is retiring after six years of service. Kurdziel brings extensive global leadership experience in the medical device industry, including roles at Medtronic, and will guide the company through key phases of FDA and Department of Defense collaboration.
Galway, Ireland-based Signum Surgical has appointed LSI alumni Carol Burns as Chief Executive Officer, to

lead the commercialization of BioHealx, the first bioabsorbable implant for anal fistula repair. She previously co-founded and served as CEO of both Cagent Vascular and Intact Vascular She has also held leadership positions at Embrella Cardiovascular (acquired by Edwards Lifesciences), Animas (acquired by Johnson & Johnson), Ventrica (acquired by Medtronic), and Guidant. With FDA De Novo clearance secured in 2024, the company will focus on U.S. market entry and post-market clinical validation.
San Diego, CA-based Vektor Medical, a leader in non-invasive, AI-powered arrhythmia analysis technology, recently announced key leadership appointments to support commercial expansion and market entry initiatives. Michael Andersen has joined

the company as Vice President, Sales, and Mihir Naik has been promoted to Vice President, Quality Assurance and Regulatory Affairs. These key moves
position Vektor for rapid growth following its recently oversubscribed $25 million Series A funding round, led by TVM Biosciences and Solas Bioventures
Andersen brings more than 20 years of commercial leadership experience in electrophysiology and medtech, with a strong track record of market development, sales execution, and strategic growth. Previously, he held senior roles at Hillrom, Abbott, and St. Jude Medical, where he successfully launched and expanded sales of advanced electrophysiology solutions.
Since joining Vektor Medical, Naik has played a key role in ensuring FDA compliance, and a robust quality and

compliance framework that supports commercial success while meeting global industry standards.
Vicarious Surgical, a next-generation robotics technology company, announced the appointment of Sarah Romano as Chief Financial Officer,

effective April 1. Romano will oversee Vicarious Surgical’s financial strategy, leveraging decades of financial leadership experience in capital markets. With exceptional strategic acumen, Romano has a proven record of driving growth and value creation in public companies, particularly in the healthcare sector, through successful fundraising and M&A initiatives.

Wellysis, a pioneering digital healthcare company spun off from Samsung SDS, has appointed Dominic P. Renda, Jr. as Chief Commercial Officer, to lead the global expansion of its AI-powered remote cardiac monitoring solutions. His experience in scaling digital health and medtech, including executive leadership positions at VivaLNK, InterSystems Corp., NetObjects, and Partnerpedia, will drive strategic partnerships and commercialization efforts. Wellysis is at the forefront of real-time monitoring, mobile cardiac telemetry, and AI-powered healthcare, with its flagship S-Patch wearable ECG monitor.

Highlights: Digitally compatible patient monitoring, epilepsy seizure detection approved for Apple Watch, novel mitral valve and transcatheter heart pump technologies, Alzheimer’s diagnostic, and much more.

Sibel Health, a Chicago, IL-based, award-winning medtech company, recently closed $30 million in new equity financing and its seventh FDA 510(k) clearance. The equity financing was led by Sibel’s existing investors, the Steele Foundation for Hope and Dräger, to accelerate commercial deployment of the ANNE One monitoring platform. The Steele Foundation for Hope previously led Sibel’s Series B round, adding an additional $20 million investment. Dräger, an international leader in medical and safety technology, previously led Sibel’s Series A round, contributing $10 million more in funding.
Enabled by advanced clinical-grade wearable sensors, the company’s ANNE One platform offers wireless monitoring of all vital signs for patients 12 years and older. The platform is one of the first medical devices to receive FDA clearance following the IEEE 11073 SDC standard, which supports open and secure communication between medical devices, replacing traditional proprietary networks. The ability to integrate wireless, wearable sensors into the digital acute care ecosystem via the new connectivity standard is an important milestone towards the future of patient monitoring along the entire patient care journey.
In March 2025, Sibel Health, a spinout of Northwestern University, is proud to also announce that Northwestern Medicine has acquired ANNE One systems to evaluate improvements to both nursing workflow and patient sleep quality with wireless sensors.
In early 2024, Sibel Health was awarded a $17.5 million award grant from the Gates Foundation. Later in 2024, Sibel Health and Dräger Denmark was selected by the Capital Region of Denmark to provide continuous wireless monitoring in multiple Copenhagen area hospitals. The company has an international office in Seoul, South Korea.

EpiWatch, a commercial stage company spun out from Johns Hopkins University, has announced FDA 510(k) clearance to market EpiWatch, a Continuous Seizure Monitor (CSM) platform that works with Apple Watch to help detect and alert for tonic-clonic seizures in people with epilepsy. As the first and only FDA-cleared seizure detection app on Apple Watch, EpiWatch is the product of a long-term collaboration between EpiWatch and Johns Hopkins Medicine
The integration of this proprietary algorithm for tonic-clonic seizure detection and alerting with a widely worn wearable device, Apple Watch, offers significant benefits, including enhanced safety and security of individuals living with epilepsy while also providing discreet monitoring—something that is important for those seeking to avoid the stigma often associated with traditional condition-specific wearable devices. Studies have shown that seizure detection and timely alerts can improve seizure management, safety, and emergency response for individuals, their families, and urgent response teams. Additionally, notification of seizure activity can help prevent Sudden Unexpected Death in Epilepsy (SUDEP), a leading cause of premature death in patients with epilepsy. EpiWatch is addressing the needs of the 3.4 million Americans living with epilepsy, 40% of whom experience seizures that are uncontrolled or poorly controlled by current medical therapies.
This FDA clearance follows the successful conclusion of the clinical trial, EpiWatch: Evaluation of a Non-EEG Physiologic Signal-Based Seizure Monitoring System, a multi-center study conducted at leading epilepsy centers. The results of the trial demonstrated that EpiWatch offers a high seizure detection rate with a low false alarm rate, making it a valuable tool in the ongoing management of seizures and epilepsy.
Virtual Incision, developer of the MIRA Surgical System, the world’s first miniaturized robotic-assisted surgery system, has announced plans to explore the use of the just-launched NVIDIA Isaac for Healthcare platform in developing their next-generation surgical robotics systems. Digital twin framework capabilities integrated with robotic learning tools will enable Virtual Incision’s developers to create highly realistic virtual environments to simulate surgical details and physiologic processes.
NVIDIA Isaac for Healthcare is a physical AI platform built on NVIDIA’s three computers for robotics: NVIDIA DGX, NVIDIA Omniverse, and NVIDIA Holoscan. It includes AI models finetuned for healthcare robotics that can understand, act, and see using enhanced vision and language processing. It also has a simulation framework for developers to accurately simulate medical environments and provides seamless deployment on NVIDIA Holoscan, an edge AI computing platform, to power robotic decision-making in the real world, in real time.
Virtual Incision will also continue to explore the NVIDIA Cosmos platform for the development of future surgical robots. The goal is to accelerate the product development cycle by leveraging comprehensive testing throughout digital prototyping, simulation, and physical deployment.
NVIDIA has announced that it will support other customers with Isaac for Healthcare for use cases including simulation environments. As an example, the tech giant recently announced that it will collaborate with GE HealthCare to advance innovation in autonomous imaging, focused on developing autonomous X-ray technologies and ultrasound applications. Neptune Medical and LSI alumni XCath are also harnessing NVIDIA’s AI robot development platform to enhance or develop their own systems.
Kirkland, WA-based Cardiac Dimensions, an innovator in minimally invasive treatments for heart failure and functional mitral regurgitation (FMR), today announced the close of an oversubscribed $53 million Series E financing round. The financing was led by Ally Bridge Group with significant participation from existing investors Hostplus, M.H. Carnegie, Horizon 3 Healthcare, Lumira Ventures, and a confidential
strategic investor, along with new investor Claret Capital Partners. The funding will support completion of the EMPOWER Trial U.S. pivotal study and continued commercialization of the innovative Carillon Mitral Contour System
The Carillon device is designed to restore natural mitral valve function without damaging the mitral valve leaflets. The simple, catheter-based procedure works by restoring the valve’s natural function and promoting favorable left ventricular remodeling. Clinical studies have shown Carillon reduces mitral regurgitation, improves quality of life, and has extended survival for a broad range of patients with FMR. Furthermore, patients with early-stage FMR may be treated with Carillon therapy as a front-line treatment option, as it does not prevent the use of other future therapies.
Functional mitral regurgitation is a prevalent condition among individuals with heart failure, which affects over 64 million people globally. Studies indicate that moderate or greater severity of FMR is present in up to 59% of heart failure patients. When left untreated, FMR can lead to increased hospitalization due to heart failure, reduced quality of life and higher mortality rates.
Samsung has announced that its companies will jointly invest $10 million in C2N Diagnostics, a U.S.-based biotech company specializing in blood-based Alzheimer’s disease diagnostics. C2N Diagnostics offers a blood test that is a safer, more cost-effective alternative to traditional diagnostic methods, such as amyloid PET-CT scans or cerebrospinal fluid tests, in accurately measuring Alzheimer’s-related proteins, including amyloid-beta. The company is seeking FDA approval for its Alzheimer’s disease-specific fluid biomarker tests, which could become a game-changer in early detection and treatment.
In September 2024, C2N Diagnostics secured a $15 million investment from the GHR Foundation to develop a tau tangles pathology test, which could further enhance the accuracy of Alzheimer’s staging. It also received $7 million from the Alzheimer’s Drug Discovery Foundation earlier last year.
Samsung C&T Corp., the de facto holding company of Samsung Group, is making the investment through the Samsung Life Science Fund, jointly created by Samsung C&T, Samsung Biologics Co. and Samsung Bioepis Co., and managed by their affiliated investment arm Samsung
Venture Investment Corp. The investment is expected to pave the way for Samsung’s venture into the diagnostics technology sector.
Evident Vascular, a San Jose, CA-based medtech company developing a best-in-class intravascular ultrasound (IVUS) platform powered by artificial intelligence, recently announced the successful closing of its Series B financing. New investors Shangbay Capital and two undisclosed multinational strategics joined founding investor Vensana Capital in the round. The funding will accelerate the development of the company’s advanced IVUS technology designed to enhance vascular imaging and patient outcomes, and support 510(k) FDA clearance ahead of U.S. market launch.
Evident Vascular’s vision is to transform vascular imaging through innovations that expand the adoption of IVUS by increasing its utility and accessibility in clinical practice. With the integration of artificial intelligence, the platform enhances image interpretation, streamlines workflows, and drives improved clinical outcomes. The flagship Evident Vascular Guidance System will deliver significant usability and image interpretation enhancements set to broaden IVUS adoption in peripheral and—soon thereafter—coronary applications.
Nanowear, a leader in home-based cardiometabolic digital diagnostics, recently announced that SimpleSense, a cloth-based, nanotechnology-enabled, wearable, mobile, and software diagnostics platform, will take data from Dexcom’s Dexcom G7 continuous glucose monitor (CGM), combining continuous glucose readings, blood pressure, ECGs, lung volume, respiration, and hemodynamics.
The integration of Dexcom G7 CGM data into Nanowear’s SimpleSense platform creates a first-of-its-kind, self-administered cardiometabolic assessment tool. By synchronizing glucose data with cardiovascular biomarkers, such as ECG, blood pressure, lung volume, respiration and hemodynamics, this collaboration offers to deliver unprecedented, real-world, real-time diagnostic insights.
Per the terms of the agreement, Nanowear will use glucose data from Dexcom G7 Continuous Glucose Monitoring Systems synchronously alongside Nanowear’s previously FDA-cleared cardiovascular biomarkers for investigational purposes.
With this recent deal, DexCom continues to strengthen its leadership in CGM integration. The collaboration also aligns with the company’s vision of expanding CGM applications beyond diabetes management, reinforcing the broader potential of continuous metabolic health monitoring. It also aligns with the growing demand in the pharmaceutical, medtech, biotech, and contract research organization (CRO) sectors, where real-world evidence is increasingly vital for regulatory and clinical decision-making.
Puzzle Medical Devices, a Montreal-based medtech startup developing a novel percutaneous heart pump to support cardiac and renal function in patients with advanced heart failure, has announced the successful closing of a CAD$43 million ($31 million) funding round. The round was led by KF Matheson and Desjardins Capital, with participation from Lumira Ventures, Longview Ventures, BDC Capital, and existing investors.
This deal represents one of the largest medical device raises in the past two years in Canada. The capital will be used to refine Puzzle Medical’s percutaneous heart pump and its novel features, advance clinical studies to assess its safety and efficacy, and scale the team to support the company’s growth. The funding will be primarily used to complete the company’s human feasibility study, which aims to evaluate the safety and efficacy of the percutaneous heart pump.
Founded in 2018, Puzzle Medical Devices is dedicated to improving patient outcomes through the development of a percutaneous heart pump for patients with advanced heart failure. The device’s modular design allows for safe implantation to support both renal and cardiac function through 4mm-pumps anchored in parallel in the descending aorta. This novel design results in low pump speeds, secure aortic anchoring, and positioning distal to the cerebral arteries. As a result, the device is expected to preserve blood integrity, reduce the risk of embolic events, and enable ambulatory and outpatient use. The long-term, percutaneous, transcatheter heart pump could also potentially provide a less-invasive alternative to more common left ventricular assist devices (LVADs).
Boston Scientific has entered into a definitive agreement to acquire SoniVie Ltd., a privately held device company that has developed the TIVUS Intravascular Ultrasound System
An investigational technology, the TIVUS system is designed to denervate nerves surrounding blood vessels to treat a variety of hypertensive disorders, including renal artery denervation (RDN) for hypertension.
As a strategic investor in SoniVie, Boston Scientific currently holds an equity stake of approximately 10%. The acquisition transaction consists of an upfront payment of approximately $360 million for the 90% stake not yet owned, and up to $180 million upon achievement of a regulatory milestone.
The catheter-based TIVUS system generates precise ultrasound energy that passes through the blood and into renal arteries supplying blood to the kidneys without anchoring to the artery wall, which allows for continual blood flow to cool the treatment area. This energy is designed to heat and ablate the bundles of nerves outside the arteries, stopping their ability to pass signals and reducing the sympathetic hormones that are released from the nerves. As a result, the blood vessels relax and the pressure within them is reduced.
Last year, SoniVie announced positive efficacy data from an IDE pilot trial of the TIVUS system in the U.S. and Israel. The company recently initiated the THRIVE global IDE pivotal trial of the device. The company has offices in Rehovot, Israel and Hoboken, NJ. (Also see Market Dive: Renal Denervation, The Lens, December 2024.)
Neuromodulation of the sphenopalatine ganglion (SPG) through transnasal evaporative cooling is a promising nonpharmacologic treatment for acute migraine, results of a randomized controlled trial with the Mi-Helper device suggest.
Mi-Helper, based in Baltimore, MD, has developed the Mi-Helper system, a drug-free, non-invasive approach to pain management, specifically designed to provide rapid relief from the comfort of home or anywhere it is needed. The investigational, semi-portable device, similar in size to a sleep apnea machine, uses dry, room-temperature air to cool the nasal cavity, which calms the SPG, relieving migraine pain and the need for medication. It consists of a durable tabletop device, tubeset, and drying cartridge. Ambient air is drawn into the device, dried using molecular sieve desiccant media in the cartridge, and delivered to the patient via the single-use tubeset. This flow of air is self-administered by the patient via the handpiece at the end of the tubeset. Air flows through the nasal pillow into their nostril and induces a phase change of moisture in the nasal turbinates. This process locally extracts energy from this area to cool certain nerve bundles, which have a clinically proven association to migraine pain and other symptoms.
The trial, with results recently reported at the American Academy of Neurology (AAN) 2025 annual meeting, enrolled 172 adults with episodic migraine, with 128 being treated with the study device and 74 analyzed for efficacy. Participants were randomly allocated to self-treat a single migraine attack at home with Mi-Helper at doses of 4 liters per minute (LPM; 14 patients), 6LPM (18 patients), 10LPM (17 patients), or sham (25 patients) for 15 minutes within one hour of onset of pain. No other treatment was allowed within that first hour.
The primary goal was to determine the most effective dose while also assessing efficacy, safety, and tolerability. The study found that the 10LPM dose was the most effective at relieving migraine pain, with approximately 47% of participants in this dose group achieving complete pain freedom at two hours compared with 16% of those in the sham group. This difference was statistically significant.
Mi-Helper is supported by private investors and has received grant support from renowned organizations such as the National Institutes of Health (NIH), Maryland Industrial Partnerships (MIPS), and the Department of Defense (DoD).
Fasikl, a neuro-AI company redefining the intersection of advanced bioelectronic medicine and AI, announced at the recent American Academy of Neurology (AAN) annual meeting that its pivotal TRANQUIL study met its primary endpoint of superiority for the company’s Felix NeuroAI wristband over a sham device for essential tremor (ET). The Felix wristband significantly reduced tremors and showed statistically and clinically significant improvement in the users’ ability to perform daily activities compared to those using the sham device.
At the end of the 90-day trial period, the Felix group showed clear superiority over the sham group. The Felix group experienced a statistically and clinically significant improvement in ET treatment, with a mean reduction of 6.9 points in modified Activities of Daily Living (mADL) scores compared to 2.7 points in the sham group (P < 0.0001). Nearly two-thirds of participants using Felix achieved at least a 20% reduction in mADL scores versus just one-quarter in the sham group. In addition, efficacy benefits were consistent across age, gender, tremor severity, and ET medication use.
Secondary outcomes further highlighted the impact of Felix. Both clinicians and patients reported greater symptom improvement with the device, and quality of life scores improved more substantially in the treatment group. Importantly,
no serious device-related adverse events were reported, confirming the device’s strong safety profile.
The Felix NeuroAI Wristband is a first-of-its-kind investigational wearable designed to deliver all-day, personalized relief for people with ET. Powered by advanced AI, Felix continuously adapts stimulation based on real-time feedback, providing dynamic, individualized therapy. Unlike surgical or pharmacologic interventions, Felix is non-invasive and wearable, connecting seamlessly to Fasikl’s cloud-based AI platform for ongoing optimization and learning.
A 2019 spin-off from the University of Minnesota, Fasikl leverages its proprietary platform to capture neural signals directly from peripheral nerves, enabling intelligent, adaptive therapy. The company is on a mission to transform the treatment landscape for movement disorders, paralysis, and other neurological impairments through cutting-edge neuromodulation innovations. Fasikl’s portfolio, currently under FDA review, includes the Felix NeuroAI Wristband and the MindForce Nerve-Computer Interface.
Irvine, CA-based Bright Uro, a medical device company with a mission to transform care for lower urinary tract dysfunction (LUTD) through innovations in urodynamics, today announced that the FDA has granted 510(k) clearance for the Glean Urodynamics System, a comprehensive system that enables wireless, catheter-free ambulatory urodynamics designed to support clinical decision making for patients with LUTD and other urological conditions.
An estimated 80 million Americans suffer from voiding problems, including overactive bladder, urinary incontinence, enlarged prostate (BPH) and neurogenic bladder. Urodynamics is the science of testing how well the bladder, sphincters, and urethra hold and release urine to determine the source of leaks or blockages.
The Glean System moves beyond current catheter-based urethral pressure testing, which is uncomfortable for patients and often generates imprecise data. Glean is easier for clinicians to use, delivers more accurate data to enable better-informed treatment plans, and offers a more comfortable experience for the patient.
Bright Uro was founded by Derek Herrera in 2021 with the goal of developing the most advanced diagnostic sensors and machine learning for urology. As a Marine Raider Special
Operations Officer, Derek was paralyzed from the chest down by a sniper’s bullet in Afghanistan. After recovering, Derek was inspired to develop products to address unmet clinical needs that he personally experienced. He began his career as a medical device entrepreneur in 2015.
Scientists from a biotechnology company that is revolutionizing the future of fertility care with end-to-end automation of the IVF lab, Conceivable Life Sciences, have reported that the first baby conceived using its fully automated, digitally controlled intracytoplasmic sperm injection (ICSI) system has been born.
Conceivable combines AI and robotics to automate more than 200 steps required to create embryos for IVF. Its workstation automates each of the 23 steps of the standard ICSI procedure. The ICSI system was developed and adopted into widespread use in the 1990s and is now routinely used for assisted conception. It achieves fertilization by injecting a single sperm cell into the center of a mature egg. Historically, ICSI procedures are performed manually by skilled embryologists operating microinjection systems, which can introduce variability in the fertilization process.
Full details are available in a new paper in Reproductive Biomedicine Online titled, “A digitally controlled, remotely operated ICSI system: case report of the first live birth.”
In January, the company announced the close of an $18 million Series A financing. ARTIS Ventures led the round, with participation from new investors including Atlantic Health Venture Studio, Muse Capital, and Scrub Capital. Existing investors include ACME Capital, Black Opal Ventures, Cadence Healthcare Ventures, Future Positive Capital, Stride.VC, and Time BioVentures. Conceivable has raised over $39 million to date.
Join our community of innovators and industry thought leaders at LSI’s premier global events. Secure your spot today for LSI Asia ’25 (June 10-13, Singapore), LSI Europe ’25 (Sept. 7-11, London), and LSI USA ’26 (March 16-20, Dana Point, CA). Details here:


Your one-stop-shop for global market sizing and analysis, procedure volume data, startup company and deal tracking, curated insights, and more.

Venture Partners (led), Orlando Health Ventures, Ballad Ventures, OSF Healthcare, WakeMed, Rex Health Ventures, CU Healthcare Innovations Fund, Sarnova
Ally Bridge Group (led), Claret Capital Partners, Hostplus, M.H. Carnegie, Horizon 3 Healthcare, Lumira Ventures, an undisclosed strategic investor 3/20/25 Asclepii Seed Funding Regenerative scaffolds for wound healing $500K Zentynel Frontier Investments 3/21/25
Alcon agreed to acquire all of LENSAR's outstanding shares for $14 per share plus up to $2.75 per share on the condition of 614,000 cumulative procedures with
Ronghui Capital (led), Hunan Pharmaceutical Development Fund, Guangzhou Bio-Island

The evolution of Intuitive Surgical from a small Silicon Valley startup into a global leader in robotic-assisted surgery is more than a story of technological progress—it’s a masterclass in mission-driven innovation, long-term leadership, and relentless focus on clinical impact. At the helm of this transformation for more than a decade is CEO Gary Guthart. Under his leadership, Intuitive has not only pioneered the category of surgical robotics, but fundamentally reshaped what’s possible in the OR and beyond.
During a keynote presentation at LSI USA ‘25 in Dana Point, CA and a follow-up one-on-one interview with The Lens, Guthart reflected on Intuitive’s journey, its market leader momentum, and the bold new frontiers that lie ahead. His insights reveal not only what it takes to lead in the medical robotics space but also how to build a sustainable healthcare innovation ecosystem centered on patient outcomes, clinician experience, and equitable access.
“Even in 1993, even with these really primitive prototypes, I was like, ‘Wow, this is crazy better.’ And that fire, this could matter, that has persisted all the way through.”
Guthart’s path into medical robotics didn’t begin with a business plan, but with a question. How can we better solve hard problems in healthcare?
“I’ve been in technology really my whole life,” Guthart shared during the LSI interview. “I started as a math and science major growing up. I found my way into surgical robots in 1993. It was one of about five research teams in the world doing surgical robots at the time. That group had been founded at Stanford Research Institute in Menlo Park. I was a young mathematician, and when I saw a demo of their early prototype, it just lit my imagination.”
That initial spark grew into a decades-long commitment. Guthart joined Intuitive as the 11th employee and 10th engineer, drawn not only by the breakthrough potential of the technology but also by the need it addressed.
“If you had asked at the time if I would have anticipated how deeply our systems would permeate into the ecosphere, I probably would not have had the imagination to think it through,”
he says. “I think that the need part, the visceral need, you could tell was really clear from the very beginning. Even in 1993, even with these really primitive prototypes, I was like, ‘Wow, this is crazy better.’ And that fire, this could matter, that has persisted all the way through.”
Guthart’s leadership style is rooted in empathy, discipline, and strategic clarity. He credits mentors like Fred Moll and Lonnie Smith with showing him what it means to lead shoulder to shoulder.
He also believes in transparency— especially with shareholders.
“From day one, we’ve messaged unwavering belief in the long term, paired with honesty about near-term challenges. That builds trust.”
The elegance and impact of Intuitive’s technology today reflects the realization of the spark that Guthart felt more than three decades ago, and helped to nurture.
Intuitive’s da Vinci Surgical System, with the first-generation system cleared by the FDA in July 2000, and the fifth-generation da Vinci 5 cleared in March 2024, has ushered in a new era of minimally invasive surgery.
“It started with a clear mission,” explains Guthart. “We knew we were part of the surgical family. Robotic-assisted surgery was a natural extension of minimally invasive techniques. Surgeons who were champions of laparoscopy understood the potential immediately.”
That clarity helped Intuitive navigate early challenges as a pioneer opening a new frontier in medicine, including technical hurdles, regulatory complexities, and clinical skepticism. The leadership team assembled a multidisciplinary group of engineers, physicians, and entrepreneurs, backed by early investors who believed in the long-term vision.
But it wasn’t just about product development, says Guthart. A unique cultural DNA took hold early on—one that emphasized an unrelenting commitment to patient outcomes.
“We had really engaged leadership, leaders who were shoulder to shoulder with people doing the work, and deeply committed to the mission.”
“The right team was pulled together,” says Guthart. “The right mix of skills. The mix of entrepreneurs, professional leadership, and the early investors. They were patient, because these things take a long time. We had really engaged leadership, leaders who were shoulder to shoulder with people doing the work, and deeply committed to the mission.”
“And so that combination, of the right mission, the right mix of talent, and then a culture centered around a deep commitment and teamwork. Those have been the things that have been pillars of strength for the company,” he continues.
The impact of robotic-assisted surgery has been profound. Over 17 million procedures have been performed using Intuitive systems, with more than 2.7 million in the past year alone. Today, Intuitive supports care in more than 70 countries, across multiple clinical specialties.
But for Guthart, the impact is also deeply personal. During his keynote, he shared the story of his mother, a retired schoolteacher who faced a long and traumatic diagnostic and treatment journey after a suspicious lung nodule was detected.
“She went through a year of follow-up scans, a failed manual bronchoscopy, a diagnostic VATS [video-assisted thoracic surgery] procedure, and a full lobectomy,” Guthart recounted. “Six days in the ICU, two additional procedures to clear her lungs… it was brutal. She lived, and to the oncologist, that’s a win. But it was a tough run.”

He then reiterates a case several years later, with a similar patient—also a retired schoolteacher, also a prior smoker—who was treated with Intuitive’s Ion Endoluminal System. In three weeks, she had a biopsy, diagnosis, and definitive surgery in a single event. She was home within 48 hours.
“It’s rare to make that kind of change in medicine. But it’s possible.”
“The difference in time was a factor of ten,” Guthart said. “That’s what we’re trying to do. It’s rare to make that kind of change in medicine. But it’s possible.”
The fifth-generation da Vinci 5 is a testament to Intuitive’s commitment to continuous innovation, Guthart explains. The latest system incorporates new computational technologies, force-sensing instruments, and higher surgeon autonomy.
“We’re proud of it. It brings some really key capabilities to customers that are the result of a lot of years of learning,” he says. “It’s both iteration that is responding to their feedback, as well as disruptive innovation that has never been brought to market before.”
“First, it’s the highest precision system we’ve brought to market, and precision matters,” he continues. “How much fidelity is there in following surgeons’ ideas and commands. The second key capability is that we’re taking advantage of next-generation imaging and creating just spectacular 3D images, which we’ve been doing for a long time. This takes it to another level for us as well.
We’ve also learned a lot about surgeon workflow and what it takes to coordinate the team that’s in the room. The system leads to less stress and strain on the team and higher efficiency, greater throughput. The ergonomics are better. The goal is to improve workflow, reduce fatigue, and deliver even better outcomes.”
A standout innovation with da Vinci 5 is the system’s ability to measure and
reflect tissue force in real time—enabling a new era of surgical data science, says Guthart.
“For the first time, we can quantify force during surgery,” he says. “We’ve introduced something called Force Feedback technology, that allows surgeons to feel subtle forces exerted on tissue during surgery, something not offered by other surgical technologies. We’ve put tiny little sensors inside the body that sense contact forces with tissue, that surgeons can then measure and record so we can track what kind of forces are happening during surgery. But also they can feel it in their hands. So it’s both a tactile sense, a feeling sense, a haptic sense, and its measurement.”
Guthart explains that these capabilities are combined with enormous increases in processing power. “We use chips from our partner NVIDIA, and that set of processing power and new data sources allow surgeons to explore surgical technique in ways that have never been done before. We can now explore questions that go back to Halsted’s principles in the 1800s—how much force is too much? What does gentle surgery really mean in data terms, and in outcomes? That’s going to change things.
“Revolutionizing surgery is an ongoing task. It’s not just one time revolutionized, it is ongoing.”
I think it will really start to revolutionize surgery once again, in this sense from a surgical data science point of view, what makes great surgery as expressed in data. Revolutionizing surgery is an ongoing task. It’s not just one time revolutionized, it is ongoing.”
Alongside da Vinci 5, Intuitive’s da Vinci SP (Single Port) system is gaining global traction in narrow-access procedures such as head and neck surgeries. “SP is also a soft tissue platform and it enters the body differently and creates new opportunities for surgeons and technique,” says Guthart. “It’s also optimized for a single-incision surgery, where surgeons want to take a specimen out and make no other cuts in the body. We’ve seen really nice uptake where we have broad clinical clearances.” (As this issue of The Lens was going to press, Intuitive’s SP SureForm 45 surgical
stapler system was FDA-cleared for use with its da Vinci SP system, spanning minimally invasive thoracic, colorectal, and urologic procedures. Intuitive notes that the SP SureForm 45 is built off the same technology as its multiport da Vinci counterpart, including systems to monitor tissue compression before and during stapling to ensure a secure hold and reduce the risk of damage.)
Guthart also highlighted Intuitive’s new Ion endoluminal system, a first-ofits-kind flexible robotic platform that allows interventionalists and surgeons to enter tortuous tubular structures such as the lung. Ion’s ultrathin, ultramaneuverable catheter allows clinicians to reach small lesions in all 18 segments of the lung with more reach, precision, and stability. The system, FDA-cleared in 2019, is now used in more than a quarter of all lung cancer diagnoses in the U.S. (Also see “Solving Wicked Problems at the Intersection of Healthcare and Technology - Insights from Intuitive Foundation’s Catherine Mohr, MD,” The Lens, February 2025.)
“Ion’s first application and indication has been in the lung, and we’ve seen great uptake with regard to navigation for lung biopsies,” says Guthart.

While surgical robots remain the most visible manifestation of Intuitive’s innovation, Guthart is quick to clarify: “We don’t think of ourselves as a robotics company. Robots are tools—they’re dead things. What matters is helping our customers deliver better care.”
That philosophy is shaping Intuitive’s next frontier: surgical data science. By embedding sensors into instruments, leveraging cloud computing, and investing in advanced analytics, Intuitive is creating systems that don’t just perform—they learn. This is an exciting area because objective metrics in surgery haven’t really existed at scale, and the company’s robotic platform facilitates measurement of those. AI technology facilitates extracting clinically meaningful objective metrics that are interpretable by a surgeon around a particular surgical activity.
“We’ve been doing IoT for 15 years. Now we’re using that data to help care teams get better faster,” Guthart says. “We’re not interested in demo features that are easy to make but hard to validate. What matters is data that improves real-world decisions.”
“High-quality, differentiated data plus the ability to validate and integrate it into workflows. That’s where the real value is.”
He is also wary of AI hype. “Everyone’s weighing their data sets, but the differentiator isn’t mass—it’s quality. High-quality, differentiated data plus the ability to validate and integrate it into workflows. That’s where the real value is.”
Under Guthart’s leadership, Intuitive has adopted the Quintuple Aim, a
framework that prioritizes better patient outcomes, better care team experiences, better patient experiences, lower total cost per patient episode, and greater access to care.
These aren’t just aspirational goals. Intuitive has more than 43,000 peer-reviewed clinical studies—with 4,000+ added in just the last year—to measure its progress across all five aims.
“Well-run robotics programs aren’t just clinically valuable. They’re often the most profitable thing a hospital does. That surprises people, but the data is clear.”
“We measure ourselves in the language of the customer,” says Guthart. “Well-run robotics programs aren’t just clinically valuable. They’re often the most profitable thing a hospital does. That surprises people, but the data is clear.”
Despite leading a company with a $100+ billion market cap and commanding over 10,000 installed systems globally, Guthart maintains a mindset more akin to a researcher than a CEO.
“I wasn’t seeking a title. I wasn’t chasing compensation,” he says. “I was chasing the question: what can we do better?”
His advice to other innovators? “Stay mission-focused. Align your team, your customers, and your shareholders around a clear set of objectives. And always, always, start with the end in mind. Who are the beneficiaries of your work? Don’t fall in love with the technology. Fall in love with the problem to solve. Then assemble a team of people who can deliver on that mission, of customers who are excited by that mission, and of shareholders who see the vision and are aligned to the mission and objectives. If you can pull those together, you
“Don’t fall in love with the technology. Fall in love with the problem to solve.”
have the opportunity to make that lasting—and the world needs it.”
Intuitive’s future will be defined by this mindset. The company is investing heavily in organic R&D, partnerships, and a venture portfolio focused on surgical intelligence, diagnostics, and digital health. (Look for a feature article on Intuitive Ventures in an upcoming issue of The Lens.)
And while it may seem like Intuitive has reached the summit, Guthart sees only more hills to climb.
“Every patient deserves the best possible outcome,” he says. “If we ask ourselves the question, what interrupts that, that’s where the opportunity lies. If we stay focused on that, we’ll continue to add value.”
For Intuitive, innovation matters, and Guthart explained this central theme eloquently.
“You look to what needs to be done, rather than what has been done,” he says. “Sometimes you can look backwards with pride, but pride can get in the way. What you really want to do is look forward. What is the remaining opportunity? And the remaining opportunity is enormous.
I think what drives innovation is the goal of making the healthcare ecosystem centered around better patient outcomes. And that is true all over the place. You can see opportunities just about anywhere you look. That includes things that are currently being done with our products and systems today. We don’t look at them and say, well, we’re done. We look at things like cancer procedures and urology and say, hey, we can be more precise. We can do better at reducing positive surgical margins. We can help surgeons get better functional outcomes.
We can also look outside of the things we do today and say, where do our skills and attributes allow us to help a different
class or set of surgical procedures and a different population of patients. We have a real opportunity to help more patients in more places. I think that is a decades-long opportunity. I don’t think it goes away.”
Guthart remains committed to the idea that every patient deserves the best possible surgical outcome. By continuing to push technological boundaries, foster a culture of innovation, and prioritize patient well-being, Intuitive is poised to shape the next era of medical technology.
What’s more, reverberation from the company’s impact extends far beyond the operating room.
Intuitive has forever changed the face of surgery, and led to shift change in the medtech arena, setting a high bar for robotic-assisted surgery into the future and spurring the next wave of innovation and strategic investment. The company has played a major role in establishing the regulatory and reimbursement framework for robotic surgery systems, and reshaping hospital economics and reimbursement to demonstrate the long-term value of these technologies.
And when asked what keeps him pushing forward three decades in, Guthart’s answer is simple: “We’re not done. There’s so much more to do. Revolutionizing surgery isn’t a one-time thing. It’s ongoing. It’s the work.”
• COVER STORY: Solving Wicked Problems at the Intersection of Healthcare and Technology
- Insights from Intuitive Foundation’s Catherine Mohr, MD (February 2025 issue)
• Market Dive: GI Endoscopy Devices (January 2025 issue)
• Power Moves (January 2025 issue)

Gary S. Guthart, PhD, is chief executive officer at Intuitive and a member of the Board of Directors, roles he has held since 2010. He draws from more than 25 years of medical, engineering, scientific, and management experience.
Guthart joined the company as part of the first engineering team in 1996 as a control systems analyst. He was promoted to vice president of engineering in 1999, senior vice president of product operations in 2002, and four years later, he was appointed president and chief operating officer.
Before joining Intuitive, Guthart was part of the core team developing foundational technology for computer-enhanced surgery at SRI International (formerly Stanford Research Institute), a renowned nonprofit scientific research organization. While at SRI, he also developed algorithms for vibration and acoustic control of large-scale systems. Guthart’s first scientific experience came early in his career in a human factors lab at NASA, supporting a team studying human performance assessment of pilots.
Guthart is on the Board of Directors for Illumina.
Guthart received a BS in engineering from the University of California, Berkeley. He earned an MS and a PhD in engineering science from the California Institute of Technology.

September 7th - 11th, 2025
JW Marriott Grosvenor House, London
Now in its fourth year, LSI Europe has become a staple of the global Medtech and Healthtech community. The 2025 event in London will convene industry-leading startups, venture capital and private equity investors, strategics, and more.
Registration and applications to present are now open for LSI Europe ‘25. Get in touch to learn more about the event.


How precision, AI, strategic vision, and patient demand are reshaping the future of surgery involving the retina, cornea, cataracts, and glaucoma.

In the high-stakes world of medical technology, the eye is a frontier like no other.
Tiny, intricate, and unforgiving, the structures of the human eye demand surgical precision measured in microns. It’s a space where the slightest tremor or delay can mean the difference between restored sight and irreversible loss. Which is exactly why robotic-assisted technology—engineered to improve upon human capabilities by amplifying control, enhancing accuracy, and reducing variability—is emerging as the next great leap in ophthalmology.
And in a telling sign, one of medtech’s biggest players just went all in.
In March, Alcon, the largest eye care device company in the world, announced it would acquire the fast-rising robotic cataract surgery innovator LENSAR in a deal worth up to $430 million. It’s a bold move that signals more than market consolidation—it’s a bet on the future of eye surgery itself.
This substantial deal spotlights a surgical market in transition, toward unparalleled precision, punctuated by demographic shifts and patient demand. And Alcon isn’t alone: a new wave of innovators is racing to reimagine ophthalmological microsurgery, with robotics and AI at the core.
Alcon’s acquisition of LENSAR includes a $356 million upfront payment, with an additional $74 million contingent on performance milestones. The star of the deal is LENSAR’s flagship ALLY Adaptive Cataract Treatment System, the first platform to combine world-class imaging and a next-generation, dual-pulse femtosecond laser in a single system, to enable precise, efficient, all-in-one cataract procedures.
In practical terms, ALLY enables surgeons to perform the entire femtosecond laser-assisted cataract surgery (FLACS) in a single sterile setting—streamlining workflow, increasing throughput, and enhancing safety.
But in strategic terms, this is Alcon doubling down on the integration of robotics and AI into its market-dominant surgical portfolio.
Almost in the blink of an eye, ophthalmology has become one of the most dynamic and high-volume segments in medical devices. Strategic deals, like Alcon’s acquisition of LENSAR, are accelerating innovation and bringing cutting-edge technologies to market. Additionally, VC funding and R&D
investment in AI-powered diagnostics and robotic systems are reshaping the surgical landscape.
According to LSI’s Global Surgical Procedure Volumes database, nearly 30 million ophthalmic procedures were performed worldwide in 2024—surpassing even cardiovascular surgeries. Refractive procedures, in particular, are seeing a steep rise, with a projected 4.9% CAGR through 2030. These procedures are often elective, premium-priced, and driven by patient demand for life-enhancing vision correction (see page 31 for the full global, U.S., and EU procedure volumes forecast).
LSI’s Market Intelligence platform, Compass, estimates global ophthalmic device sales reached $14.9 billion in 2024, with Alcon commanding over 35% of that share. With LENSAR’s ALLY system already accounting for an estimated 10% of the FLACS market, Alcon is positioning itself to extend its lead and future-proof its surgical franchise.
In addition to technological advancements such as robotic-assisted surgery, the growth in ophthalmologic surgery is being driven by a powerful mix of demographic shifts and rising patient expectations:
• Cataracts, glaucoma, and age-related macular degeneration (AMD) are more prevalent in older adults. As global life expectancy increases, the demand for vision-restoring surgeries rises.
• Cataract surgery is the most common elective procedure worldwide, with over 30 million surgeries per year.
• By 2030, it’s estimated that over 1 billion people worldwide will be over 65, fueling higher surgical volumes.
• Myopia (nearsightedness) is reaching epidemic levels, particularly in Asia. Procedures like LASIK, SMILE, and phakic intraocular lens (IOL) implants are increasingly sought after.
• Lifestyle shifts (more screen time, less outdoor activity) are contributing to this rise globally.
• Emerging markets are improving access to ophthalmological care through public health initiatives and private investment.
• Mobile clinics and teleophthalmology are reaching previously underserved populations.
• Patients want better-than-normal vision post-surgery.
• Demand for premium IOLs—such as multifocal, toric, and extended depth-of-focus (EDOF) lenses—is growing.
• Cosmetic expectations are influencing refractive and cataract surgery markets.
• Diabetes-related eye diseases, especially diabetic retinopathy, are on the rise and driving demand for surgical interventions like vitrectomy.
• The global diabetes epidemic is expected to significantly impact ophthalmologic surgery demand.

If someone were to design the perfect application for robotic systems, eye surgery would be near the top of the list. The anatomy is complex. The tolerances are tight. And the margin for error is razor-thin.
“Robotics are a natural fit in ophthalmology,” says one surgical robotics executive. “When you’re operating on a space the size of a pinhead, even the steadiest hands have limits.”
Whether it’s performing subretinal injections for gene therapy or executing ultra-fine maneuvers on the retina, robotic tools offer a level of control and consistency that human dexterity alone can’t match. In eye surgery, precision isn’t a niceto-have—it’s everything.
With Alcon now investing heavily, the LSI Market Intelligence team expects robotic platforms to expand beyond cataract surgery into retinal, glaucoma, and other ophthalmic subspecialties.
Alcon isn’t the only ophthalmology player eyeing the future. A wave of fast-moving innovators is racing to reimagine microsurgery with robotics and AI at the core. Among them:
Innovator Technology/Recent Development
AcuSurgical Focused on delicate retinal operations, AcuSurgical is developing robotics-assisted systems to help surgeons navigate and treat the back of the eye with new levels of control. In July 2024, the company announced the successful completion of its first clinical study with the Luca surgical robot for vitrectomy procedures, at Gent University Hospital in Belgium.
ForSight Robotics
Developing a hybrid robotic ophthalmic platform using AI-based algorithms, advanced computer vision, and miniaturized mechanics, ORYOM offers 14 degrees of maneuverability for high-precision eye surgeries.In February 2025, the company appointed Dr. Fred Moll, co-founder of surgical robot giant Intuitive Surgical, to its board of directors.
Developing Polaris, a robotic microsurgery system that integrates advanced imaging and AI to optimize cataract surgery outcomes. In October 2024, the company closed a $30M Series A financing, led by ExSight Ventures and a major corporate investor, with participation from Main Street Advisors and University of California (UC Investments), among others.
B.V.
Innovating robotic solutions to assist with vitreoretinal surgeries, enhancing precision in this specialized field. In March 2022, the company was acquired by Carl Zeiss Meditec AG and is now integrated into the ZEISS group.
Source: LSI Market Intelligence
These companies, along with Alcon and others, are pushing the boundaries of robotic eye surgery technology, expanding its potential applications and improving outcomes for patients undergoing cataract, retinal, and glaucoma surgeries.
Labs around the world are also testing new robotic innovations for eye surgery. In one example, researchers at the University of Utah have developed a helmet-like robotic platform to assist in subretinal injections—even when patients move unexpectedly during surgery. With robotic stability, more surgeons could access and safely deliver advanced therapies such as gene editing for macular degeneration.
For all the excitement, robotic ophthalmology isn’t without its challenges.
The cost of development and regulatory approval remains high. Surgeon training will need to evolve. And hospitals must weigh capital investment against immediate clinical ROI.
Still, for companies willing to think long-term, the prize is clear: a scalable, intelligent surgical solution for one of the world’s most common—and in-demand—procedures.
Alcon’s LENSAR acquisition is not only a headline, it’s a harbinger. It marks the moment robotic ophthalmology moved from promising to inevitable.
It’s also a wake-up call to the medtech industry: the future isn’t just about seeing better. It’s about doing better.
With vision care volumes rising as the population ages, surgical complexity increasing, and the demand for precision at an all-time high, robotics offers more than innovation—it offers transformation.
All data in this article is sourced from LSI’s Market Intelligence Platform, and the April 1, 2025 edition of LSI’s The Numbers on LinkedIn and on the LSI website under Resources > Research Desk: “Robotic Eye Surgery and Alcon’s Strategic Acquisition.”
Access LSI’s continuously updated Market Intelligence Platform.
Scan QR code for details and to request a demo.

The industry’s most comprehensive and trusted global procedure database, with coverage of 300+ diagnostic and therapeutic procedures across 37 countries.

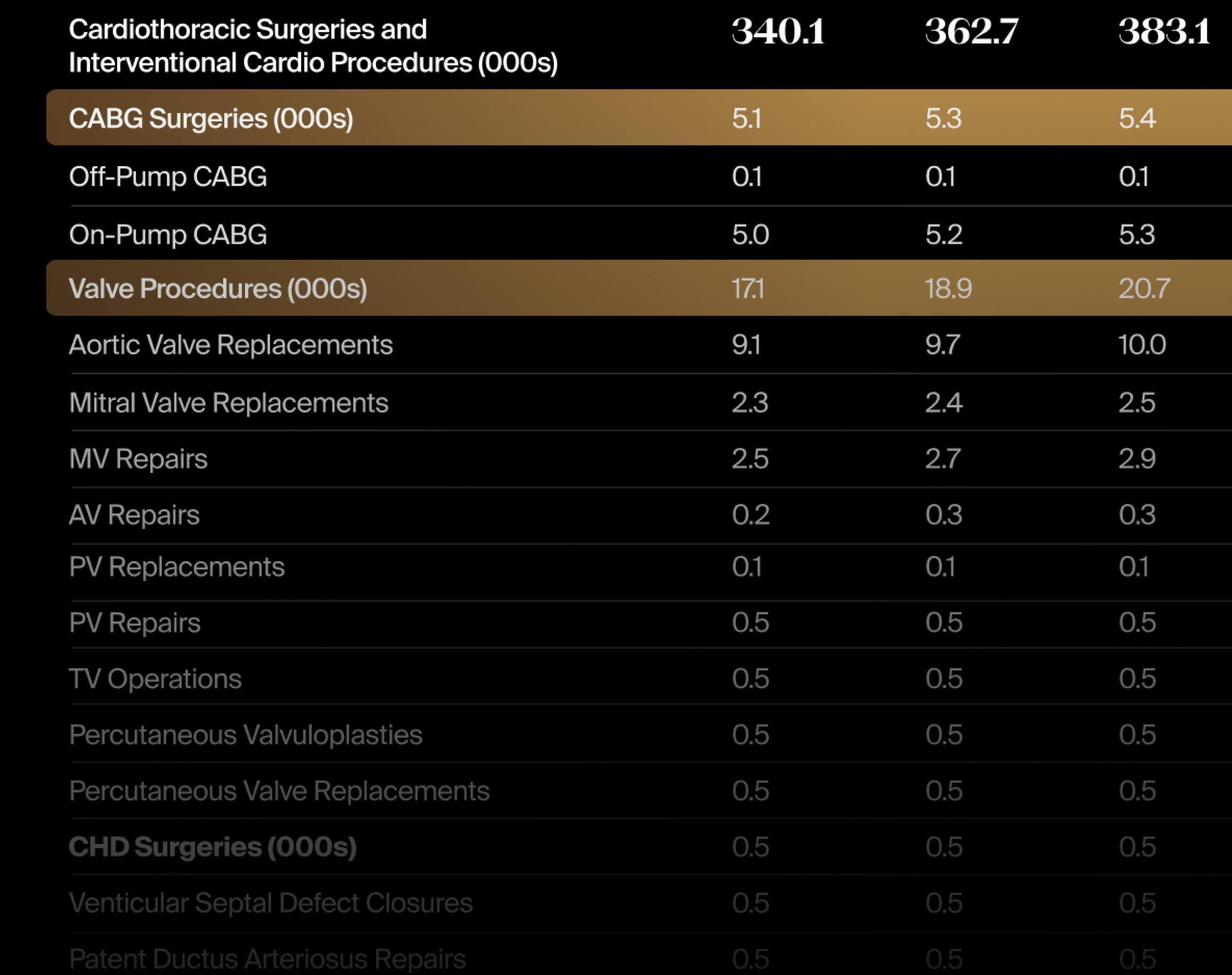

This month, to accompany our Market Dive on ophthalmology innovations, we detail the latest LSI Market Intelligence data on the global, U.S., and European forecast volume for select ophthalmological surgical procedures, by type.
LSI’s Global Surgical Procedure Volumes (SPV) Tracker provides trusted coverage of 300+ diagnostic and therapeutic procedures across 37 countries. Scan QR code to request a demo:

LSI’s SPV Tracker includes these geographies: Argentina, Australia, Belgium, Brazil, Canada, Caribbean, Chile, China, Colombia, Costa Rica, Denmark, Dominican Republic, Finland, France, Germany, Guatemala, India, Italy, Japan, Malaysia, Mexico, Netherlands, New Zealand, Norway, Panama, Poland, Russia, Singapore, South Africa, South Korea, Spain, Sweden, Switzerland, Thailand, Turkey, United Kingdom, and the United States.

Launching a new medical technology is always a gamble, but according to Ilya Trakhtenberg and Stuart Jackson of L.E.K. Consulting, a systematic, step-by-step approach to innovation can manage risk and unlock the full potential of breakthrough innovation for organizations—far surpassing industry benchmarks.
Most innovations fail. And most companies struggle with innovation. Case in point: Nearly all the growth in the S&P 500 (prior to 2025 YTD) has been driven by just seven companies, the so-called “Magnificent Seven”—Alphabet, Amazon, Apple, Meta, Microsoft, NVIDIA, and Tesla. Between 2013 and 2019, they delivered 19% compound returns. The other 493 companies in the index delivered just 2% in that time period. And medtech is no different: the top five market cap growers accounted for 60% of all of the industry’s market cap growth
from 2015 to 2024, and a full 80% of it since 2019. Studies show that of the thousands of product launches across industries each year, 70-90% fail.
So how do you bridge that gap if you’re a medtech company looking to succeed with a novel technology? And not only that, but build an enduring franchise?
According to Ilya Trakhtenberg, a partner at L.E.K. Consulting and co-author of the new book, Predictable Winners: A Handbook for Developing, Forecasting, and Launching New Products and Services, in a recent conversation with The Lens, along with his co-author and Vice Chair of L.E.K. Consulting, Stuart Jackson, success is a learnable, doable process.
But this isn’t a book review. It’s a field guide for founders, operators, and commercial leaders who know that FDA
clearance alone won’t get your product into the hands of clinicians, or into the standard of care.
“While there may be certain things that are special about the Magnificent Seven, and other successful entities, the way that they pursue innovation is a little bit different, and it is learnable,” Trakhtenberg told The Lens. “Our mission was to show that success in innovation isn’t about luck, it’s about de-risking at every stage of the journey—from concept through commercialization. Our philosophy is akin to the movie Ratatouille, but instead of anyone can cook, we believe anyone can innovate.”
The key is identifying your organization’s weak spots and closing the gaps.
Drawing from decades of consulting experiences and hundreds of product launches across industries, Trakhtenberg and Jackson’s view is that
innovators—be it medtech innovators or those in other industries—need to systematically eliminate the sources of failure from step to step.
“We essentially outline what good looks like at each step, what are the common pitfalls, and how to avoid them,” says Trakhtenberg. “We go through that for the whole journey. Our point is you need to think about it as systematically managing the entire innovation process.”
The authors’ lessons are particularly relevant for the medtech ecosystem, based on the increasingly complex but yet essential nature of life-impacting innovation. For those launching new products and services in this arena— and especially those with novel breakthrough products where the pathway to market is more uncertain and difficult—they emphasize the importance of structured execution, organizational readiness, and strategic commercialization to drive adoption and long-term impact. These qualities can help separate sustained medtech innovation from fleeting success.
To make innovation more predictable, Trakhtenberg and Jackson lay out a five-stage journey for product development, in which each stage helps reduce uncertainty and builds momentum:
1. Configuring for success:
Embracing a systematic approach to managing every step of the innovation journey, with constant reevaluation in light of new information and with the right personal mindset on the part of those leading innovation teams.
2. Developing product and service concepts:
Sourcing and developing highpotential innovation ideas, taking advantage of lean approaches to concept development and assessment, including tactics large organizations can use to embrace breakthrough innovation.
3. Forecasting revenue:
Developing a realistic forecast and
business case for an innovation— including understanding opportunity size, target customers, adoption expectations, competitive threats, and optimal pricing and pricing models. If the forecast isn’t robust enough to convince management or investors, or if the necessary product or service concept doesn’t appear to be achievable, then you’re better off walking away at the outset.
4. Ensuring commercial success:
Taking the key steps and overcoming the hurdles involved in going from fully developed concept to successfully launching and generating revenue, including proactively identifying and addressing adoption barriers, creating a launch plan, and preparing the organization for successful launch execution.
5. Creating value for the long-term:
Avoiding the traps that cause many organizations to destroy value after the launch, and using product innovation as part of the broader corporate development agenda for different types of organizations.
Here, Trakhtenberg shares a few takeaways from Predictable Winners for each stage, as food for thought to medtech stakeholders looking to improve their innovation strategy and execute with confidence.
Between 70 and 90% of new product launches fail. And most companies never figure out why, say Trakhtenberg and Jackson. In medtech, where capital is scarce and clinical complexity is high, the odds can feel even steeper.
“Our industry depends on a pipeline of real, transformative innovation,” says Trakhtenberg. “But the easy wins are gone. What’s left are the tough problems. And there are plenty of unmet needs.”
Tighter capital, complex regulations, and often longer product development
timelines mean there’s no room for sloppy execution.
“We can’t afford to guess anymore,” he adds. “We need structure, discipline, and courage.”
Success, says Trakhtenberg, isn’t about getting lucky. It’s about managing risk in a disciplined way. “Our mission is to show that innovation success is learnable. It’s about de-risking at every stage of the journey.”
Medtech companies often depend on acquisitions to stay innovative. But integrating those technologies successfully is the real challenge. The real problem they face lies in generating an attractive return on investment in product and service innovations, say Trakhtenberg and Jackson.
“You can’t throw products out there and hope they stick—especially in medtech.”
“It’s not enough to just buy stuff and hope it works,” Trakhtenberg continues. “With factors such as regulation, science, and market complexities, you can’t throw products out there and hope they stick—especially in medtech.”
The most effective innovators take a long view, thinking across the entire journey—from concept to commercialization and beyond. Trakhtenberg and Jackson describe what “good” looks like at each step and outline how to avoid the most common mistakes.
The result? More disciplined investment decisions, fewer false starts, and a portfolio tilted toward winners, not waste. The goal isn’t perfection. It’s progress—through structure, iteration, and a repeatable process that any medtech company can adopt.
In the first few chapters of their book, Trakhtenberg and Jackson recommend that companies manage risks by evaluating multiple indicators for success:
• There is no single ingredient that guarantees success; even the
best ideas will fail if other elements of the innovation journey are not executed successfully.
• Leverage multiple sources of evidence such as product analogs, market research, and live trials.
• Be aware of the strengths and prejudices of your own organization. Just because you have a strong market insights group or a world-class rapid prototyping team does not mean that you should ignore other sources of information.
• Take time to experiment, but make sure those experiments translate into actionable insights.
Innovation often stagnates in large organizations due to risk aversion and organizational inertia, says Trakhtenberg. They can struggle with the kind of breakthrough innovation that’s more often associated with nimble startup enterprises.
What makes startups special in this regard? They tend to flourish when there is an established ecosystem of talented entrepreneurs and willing backers, say Trakhtenberg and Jackson. This creates a reinforcing cycle for exchanging and refining ideas; attracting, developing, and motivating talent; and recycling financial gains from successful exits into new opportunities.
But what if you’re an established business trying to develop breakthrough innovation?
You can always outsource it, or you can foster an environment within your organization to focus on specific innovations. A key point that Trakhtenberg and Jackson emphasize in Predictable Winners is the importance of instilling a startup mentality within medtech giants.
“One of the biggest challenges is maintaining the magic of a startup inside a larger organization,” says Trakhtenberg. “There are a lot of organizational best practices that large organizations can onboard from startups to try to
recreate much of the feel, but in a real and actionable way. You need to create an environment where risk-taking is encouraged, and failure is seen as part of the process.”
Broad recommendations on how to get there:
• Build a dedicated team
• Reward milestone-driven progress, not just headcount
• Create internal venture-style funding mechanisms (both positive and negative)
• Shield breakthrough teams from legacy bureaucracy
• Incentivize learning and experimentation
He points to Intuitive Surgical as a rare example of how to do this well. (Read more about Intuitive Surgical’s journey and CEO Gary Guthart in this issue’s Cover Story.)
Trakhtenberg advises that large medtech firms improve their internal innovation capabilities rather than relying solely on external acquisitions.
The table below, excerpted from Predictable Winners, summarizes the key organizational challenges and potential solutions for early-stage development of breakthrough innovations in large organizations.
Predictable Winners also highlights the necessity of integrating digital capabilities into medtech strategies, not as a separate initiative but as a fundamental part of business operations.
“Talking about digital strategy today is like talking about internet strategy 25 years ago,” says Trakhtenberg. “It’s not a nice-to-have—it’s foundational. Digital is here to stay and you have to be thoughtful about how you’re incorporating it.”
He offers a digital value hierarchy, demonstrating how data becomes exponentially more valuable when used to drive real-time decision-making, as seen in today’s advanced closed-loop insulin delivery systems and, going forward, in AI-assisted robotic surgery:
1. Detect: Capture raw data
2. Analyze: Spot patterns and anomalies
3. Act: Drive automated or guided clinical actions
“The more autonomous your solution, the more value it delivers. Using the example of closed-loop insulin delivery systems, it is also disrupting an industry, in the way patients are empowered to monitor themselves,” he notes.
Challenges for In-House Startups
Lack of focused resources
People lacking creativity and risk tolerance
Insufficient incentives given career risks
Lack of recognition and ownership stake
Slow decision-making
Inconsistent support from divisions/functions
Lack of feedback from fundraising cycle
Solutions for Large Organizations
Create small, dedicated teams
Provide coaching; be a home for varied talent
Increase rewards; make it a rite of passage
Create legacy-like recognition
Delegate decision-authority, within boundaries
Encourage support, but allow go-arounds
Tie resources to progress against milestones
Source: Jackson, SE, Trakhtenberg, I. Predictable Winners: A Handbook for Developing, Forecasting, and Launching New Products and Services
A compelling revenue forecast and business case for a new product or service concept are critical to winning support from investors and other stakeholders, say Trakhtenberg and Jackson. What’s more, a rigorous research and forecast development process covering opportunity size, target customers, realistic adoption expectations, competitive threats, and pricing will generate conviction in a revenue forecast and lay the groundwork for a successful commercial launch.
The authors say that one of the most common issues they see in medtech is a false understanding of who the early adopters should be for a company.
“Most medtechs try to sell to their friends and family—advisors, KOLs, longstanding customers, investigators in trials. These are not necessarily the best early adopters—you will inherently run out of friends and family,” says Trakhtenberg. “You need adopters who love your product and are willing to become champions and references. Being pickier with early adopters is often critical to successfully segmenting customers.”
Another common challenge is underestimating the competition. An important part of assessing competitive threats is to think like a competitor, and assume that they are at least as smart as you are.
Many medtech startups, and larger companies, underestimate how quickly and aggressively competitors will react to their innovations. Trakhtenberg stresses the importance of strategic foresight.
“One thing that we recommend executives spend more time on is war gaming, where you roleplay as different competitors,” he says. “It’s a tactic that can be extremely helpful for really putting yourself in the shoes of your competitors to think through, what are they realistically going to do? If you were running that business, what would you do? You end up with a far more nuanced understanding of what to expect and then you can, as an innovator, plan. People don’t actually do that very often, or
they’re not thoughtful enough about it.”
Trakhtenberg notes that by proactively considering how rivals will adjust pricing, messaging, and product positioning, innovators can strengthen their market strategy prior to launch.
One last point on competition: don’t forget to look ahead.
“Just because you are the disruptor today doesn’t mean you won’t be the disrupted tomorrow,” says Trakhtenberg. Think one year post-launch, three years, five years, and beyond.
“Just because you are the disruptor today doesn’t mean you won’t be the disrupted tomorrow.”
Trakhtenberg and Jackson’s research shows that one of the biggest gaps when it comes to the entire innovation journey is when people start preparing for launch. Poor commercialization—not poor technology—is the most common reason for launch failures.
“The best practice is to understand your adoption barriers, which ones matter the most, and how you can overcome them as much as physically possible before you’re on the market. Once you launch, it’s too late to fix fundamental issues,” says Trakhtenberg.
One of the most overlooked aspects of a successful medtech launch is proactively identifying and mitigating adoption barriers, notes Trakhtenberg. Without thorough preparation, even the most innovative technologies can struggle to gain traction in the market.
“The best innovators don’t just launch a product; they actively track and address adoption hurdles—whether it’s surgeon training, workflow integration, or reimbursement alignment,” he says.
Questions for innovators to ask include understanding how their customers will look at the product, he
continues. “How is this going to fit in their workflows? Do they get the economics? Are the incentives aligned for the clinicians, for the administrators, for your reps?”
Trakhtenberg cites Stryker’s Mako and Procept BioRobotics as companies that effectively identified and mitigated obstacles with their innovations, ensuring smoother market entry.
Understanding why potential customers abandon a product at various stages of the buying journey is critical to improving adoption rates, say the authors of Predictable Winners.
“A structured leakage analysis helps companies pinpoint where they’re losing potential or existing customers, and why—whether it’s lack of awareness, misaligned incentives, patient referral issues, workflow challenges, or maybe the footprint in the OR is wrong. We saw that with some of the robotics technologies,” says Trakhtenberg.
“Understanding these barriers and what you can do to address them, then prioritizing which ones are worth your time, effort, and money to go after, is something that I can’t tell medtech clients to do more of,” he continues.
• Leverage multiple sources of insight to evaluate innovation potential
• Identify and validate the riskiest assumptions early
• Design experiments that drive actionable learning
• Fund innovation in tranches, not waterfalls
• Incentivize reps for long-term success, not one-time sales
• Equip commercial teams with decision-making tools, not just decks
This approach also helps ensure that innovations keep being used, and don’t end up gathering dust in a hospital storage room. Placement or getting a product on the shelves doesn’t ensure utilization.
Trakhtenberg notes that very few companies do a good enough job of understanding the barriers to adoption and coming up with effective strategies for overcoming those barriers before they’re on the market. “That’s how you end up with surprises like, ‘Well, adoption isn’t going as quickly as we thought,’” he says. “And for a startup, that can be the difference between life and death.”
With tougher regulatory hurdles, higher-risk innovation, and funding challenges, Trakhtenberg warns that medtech must build stronger internal capabilities to drive future growth.
Key considerations at this stage of the commercial success journey include paying careful attention to these five key areas: operations, competition, international expansion, resourcing, and managing investor expectations. The authors note that innovators need to understand how hard it is to maintain success and growth beyond the first product, and be prepared to invest accordingly.
From a long-term perspective, incremental developments can be utilized to complement breakthrough innovation, and partnerships and acquisitions are powerful tools for accelerating innovation value.
“Be a garage startup with a Fortune 500 business plan.”
“Focus on finding the best ideas, whether those come from internal teams or potential acquisition targets,” advise the Predictable Winners authors. “Be a garage startup with a Fortune 500 business plan.”
For medtech teams navigating longer timelines, tighter margins, and rising complexity, Predictable Winners offers not just insights—but an actionable blueprint.
“Our goal is to offer time-tested best practices that give innovators some pause, and to think about applying these principles to their own journeys. The key takeaway is that the companies that
succeed will be the ones that improve their ability to manage breakthrough innovation—and maximize their odds of success,” says Trakhtenberg.
“Innovation is the lifeblood of this industry, but it’s getting harder,” he continues. “The companies that succeed will be the ones that improve their internal ability to manage breakthrough innovation and maximize their odds of success.”

Ilya Trakhtenberg (on the right in the photo) is a Partner and Managing Director in L.E.K. Consulting’s Chicago office and a leading partner in its healthcare practice. In his more than 15 years with L.E.K., he has led hundreds of engagements with a wide range of corporate and private equity clients to develop and launch innovative new products, accelerate business growth, and deliver superior investment returns. He has published more than a dozen journal articles, serves as a regular speaker at industry conferences, and founded L.E.K.’s MedTech Launch Center of Excellence. Trakhtenberg is also a past recipient of M&A Advisor’s Emerging Leaders Award and a board member of L.E.K.’s global Sustainability Centre of Excellence.
Trakhtenberg studied Economics and Political Science at Northwestern University and was awarded an MBA with Honors from the Kellogg School of Management.
Stuart Jackson (on the left in the photo) is Vice Chair of L.E.K. Consulting, where he advises clients on organizational performance, governance issues, and strategic growth. He previously served as L.E.K.’s Global Managing Partner for nine years and has held key leadership roles, including Americas Region Head and Chicago office head and the firm’s Board of Directors.
Over his career, Jackson has worked with hundreds of clients across industries such as medtech, manufacturing, and consumer products, helping them achieve higher growth, increase profit margins, expand product offerings, and complete over 100 M&A transactions.
Jackson is a past recipient of Consulting Magazine’s Top 25 Consultants award. He is the author of the highly acclaimed book “Where Value Hides: A New Way to Uncover Profitable Growth for Your Business” and dozens of articles published in journals such as the Wall Street Journal, the Journal of Business Strategy, Forbes and Harvard Business Review. Before joining L.E.K., Jackson worked as a manufacturing engineer with BAE Systems. He holds an engineering degree from Cambridge University and earned his MBA with distinction from INSEAD, where he was awarded a NATO Fellowship.
De-risk your biggest strategic decisions with our most comprehensive market data and projections (worldwide sales, share by supplier, CAGRs) on 27 major market segments and 200+ sub-segments, prepared with consistent methodologies and forecast periods.


Let’s face it, we’re all tired of the pop-ups, clickbait, and ads.
That’s why we created The Lens — well-curated insights and coverage, from the analysts and industry insiders you trust.


$599 for your first year
Billed annually and includes 12 editions $699.00
Celebrating the accomplishments of a few of the many bright stars in our LSI presenting company and speaker alumni community.
Be sure to check out presentations from these and many other innovative companies from our recent LSI events on our website, at Resources > Video Library.


Minneapolis, MN-based 4C Medical Technologies, a device company dedicated to advancing minimally invasive therapies for structural heart disease, announced the closing of a Series D financing round, resulting in gross proceeds of up to $175 million, led by Boston Scientific, with participation from certain new and existing shareholders. The financing will be utilized to accelerate the clinical development and commercialization of the AltaValve System, the company’s next-generation transcatheter mitral valve replacement (TMVR) technology. The proceeds will primarily be used to support the ongoing global pivotal trial, ATLAS (A
Transseptal Left Atrial System for Treatment of Mitral Regurgitation), in the U.S. and Europe. 4C Medical commenced U.S. enrollment of the ATLAS trial in October 2024.
Mitral regurgitation (MR) is the most common valvular heart disease, affecting nearly 10% of Americans aged 75 years and older, that occurs when the mitral valve leaflets do not close properly, allowing blood in the left ventricle to leak back into the left atrium. It is a progressive disease and if left untreated, MR can lead to atrial fibrillation, pulmonary hypertension, heart failure, and death.

Aescape announced that it has secured $83 million in strategic funding
to expand its AI-powered robotic massage technology. The round was led by Valor Equity Partners, an early investor in Tesla and SpaceX. The round also saw participation from existing investors, including Alumni Ventures, and NBA Champion and All-Star Kevin Love. With this latest investment, Aescape’s total funding now stands at $128 million, enabling the company to scale operations, expand into new markets, and grow its partnerships with global brands. Using advanced AI technology, the company is delivering the world’s first fully automated, customizable massage experience for personalized wellness and recovery. Since debuting with Equinox, Aescape has announced partnerships with industry leaders in hospitality, corporate wellness, luxury real estate, and professional sports— including select Four Seasons Hotels and Resorts, Marriott, and Ritz-Carlton properties throughout North America.
Founder and CEO Eric Litman participated in a Fireside Chat at LSI USA ‘25, entitled “Wellness 3.0: Building and Scaling a Robotics Startup in a New Era of Well-Being,” where he discussed Aescape’s journey, vision, and the future of technology-driven wellness.



Startup Ajax Health, a turnkey growth solution for commercial-stage medtech companies founded by Duke Rohlen in 2017, has applied its innovative funding model to FlowMod, a new platform in collaboration with KKR and Boston Scientific for the development of a novel heart failure treatment system. The new entity will be led by Dr. Philippe Marco, former President and Chief Operations Officer of Epix Therapeutics and CV Ingenuity, both Ajax-led companies that obtained pre-market approval for groundbreaking cardiovascular devices. FlowMod aims to accelerate the clinical validation and regulatory approval of the heart failure technology to address the 64 million people affected by the condition worldwide.
The new company, FlowMod, is an example of an innovative new collaborative funding model that Rick Anderson, Chairman and Founding Managing Director of Revival Healthcare Capital, cites as a build-to-buy strategy with a pre-negotiated exit. This type of emerging partnership between strategics and investors involves investors putting money in for a set time period in a thoughtful way, with a pre-negotiated exit. (Also see Reimagining How We Innovate in Medtech: Alex Gorsky and Rick Anderson Offer Predictions of a Disruptive Future, The Lens, September 2024.)
This deal follows the November 2024 transaction in which Boston Scientific entered into a definitive agreement to acquire Ajax Health company, Cortex. Cortex is a privately held medtech company focused on the development of a diagnostic mapping solution which may identify triggers and drivers outside of the pulmonary veins that are foundational to atrial fibrillation, a heart rhythm disorder affecting nearly 38 million people globally. Ajax Health is based in Menlo Park, CA.
San Clemente, CA-based AtaCor Medical, a privately held device company focused on transforming cardiac rhythm management, has initiated its ASCEND EV study. The prospective, non-randomized international study aims to evaluate the safety and performance of AtaCor’s all-in-one Extravascular Implantable Cardioverter Defibrillator (EV-ICD) Lead System in combination with leading commercially available transvenous ICDs. The initial cases were performed at a single clinical site and demonstrated successful sensing and defibrillation of induced ventricular fibrillation using the Atala lead in connection to a range of commercially available ICDs. Early cases successfully demonstrated sensing and defibrillation of induced ventricular fibrillation, with the company planning to expand the study to additional clinical sites.
The AtaCor EV-ICD Lead System aims to deliver life-saving ICD benefits without the complications associated with transvenous leads or the pacing discomfort associated with other extravascular ICD systems. The all-inone system facilitates both antitachycardia pacing and shock delivery from a single lead and with one procedure—all without entering the heart or vascular system. Implanted via a streamlined parasternal approach, the system is designed for compatibility with commercially available transvenous ICDs and will accommodate either a lateral or left-pectoral pulse generator pocket placement.
Avatar Medical, an immersive medical imaging innovator, has received FDA 510(k) clearance for brAIn SPS, a cloudbased AI-powered shoulder positioning system developed in collaboration
with FX Shoulder Solutions. The brAIn SPS offers advanced 3D visualization of both bone structures and soft tissues, enhancing surgical planning accuracy and improving patient outcomes in shoulder arthroplasty.
For orthopedic surgeons, brAIn SPS offers unparalleled insights by allowing the visualization in stunning 3D detail. The technology features automatic segmentation, humeral and glenoid planning, and can allow surgeons to gain insights on the potential impact of implant positioning on the soft tissues.
Avatar Medical also announced that it has obtained CE marking for its innovative solution, Avatar Medical Vision, that enables the real-time conversion of patient medical images into stunning 3D and XR renderings, enhancing visualization and understanding.
Founded in 2020 as a spin-off from the Institut Pasteur and Institut Curie, Avatar Medical democratizes access to medical imaging through advanced 3D visualization software. With offices in Mountain View, CA, Paris and SaintMalo, France, Avatar Medical redefines patient-doctor consultations by enhancing understanding, reducing anxiety, and improving decision-making.

Santa Cruz, CA-based Capstan Medical announced that it has completed the world’s first robotic-assisted transcatheter mitral valve replacements, demonstrating unprecedented
catheter stability and precision. This marks the first time a minimally invasive robotic structural heart platform has been used in clinical practice, successfully delivering a novel mitral valve into two human patients. The procedures were performed at Hospital Clínico Universidad Católica in Santiago Chile, by Dr. Gonzalo Martinez, MD MPhil, Director Endovascular Therapy Center at Pontifical Catholic University of Chile, along with Dr. Santiago Garcia, Director, Structural Heart Program at The Christ Hospital in Cincinnati, Ohio, who supported the cases.
This follows the company’s oversubscribed $110 million Series C funding in December 2024, led by Eclipse, with participation from existing investors Yu Galaxy and Intuitive Ventures, and new investment from Gideon Strategic Partners. Capstan is advancing toward pivotal trials to expand access to minimally invasive heart valve interventions.
The company also recently hired former Intuitive Surgical engineer Greg Dachs as R&D head (see Power Moves, The Lens, February 2025). Founded in 2020, Capstan Medical is creating a new standard for minimally invasive, heart valve intervention, with a suite of next-generation heart valve implants and a first-of-its-kind, catheter-based robotic delivery platform.

Caresyntax, a leader in surgical intelligence and automation technologies, and Pristine Surgical, a medical device company dedicated to Simplifying Endoscopy, recently announced a partnership to enhance surgical intelligence for Pristine’s Summit 4K single-use digital arthroscope and its Pristine Connect digital platform.
Through this collaboration, the companies will work alongside world-class

arthroscopy surgeons leveraging Caresyntax’s Clinical Data as a Service (CDaaS) platform. The partnership will focus initially on real-world data from orthopedic procedures performed in outpatient ambulatory surgical centers (ASCs). This partnership will help drive surgical innovation and collect realworld data to increase efficiency, lower costs and provide key insights for hospitals and ASCs to improve patient care.
With a surgical intelligence network spanning more than 4,200 operating rooms worldwide, Caresyntax has pioneered data-driven insights for hospitals. This partnership will expand the reach of Caresyntax’s surgical intelligence to ASCs, laying the foundation for further data-driven analysis in non-hospital settings. (Also see LSI Alumni Milestones: Wins Announced in January, The Lens, February 2025.)

Cleveland, OH-based Centerline Biomedical, an innovator in cardiovascular navigation and visualization systems, announced that it has received FDA 510(k) clearance of expanded indications for its Intra-Operative Positioning System (IOPS). This news is complemented by the recent 510(k) clearance of the IOPS Fiducial Tracking Pad, a second-generation, lower-profile device useful in broader applications of the IOPS platform.
During minimally invasive surgeries, clinicians typically rely on fluoroscopy, or live X-ray, to see anatomy and medical devices inside the body. Use of fluoroscopic imaging exposes treating clinicians and patients to X-ray radiation, which aggregated over time, introduces risks for DNA damage and other medical complications. IOPS was designed to minimize the need for prolonged fluoroscopy use and promote the safety of both clinicians and patients, achieved through advancements in both device navigation and visualization.
The IOPS platform makes use of patient anatomical images or scans already captured for diagnosis and procedure planning to create multi-color, 3D vascular maps. These interactive maps of the anatomy, including arteries and veins, can be displayed in multiple views and highlight the finer features of diseased vessels; including calcifications, occlusions, dissections, and other anatomical and soft tissue formations not clearly visible when using traditional, 2D, grayscale X-ray fluoroscopy. During procedures, the maps guide IOPS proprietary, sensorized guidewires and catheters during real-time navigation of the vasculature, while minimizing dependence on fluoroscopy.

Canadian ultrasound device innovator, Flosonics Medical, has secured $7.5 million CAD ($5.4 million) in venture debt financing from RBCx. The financing and specialized support will accelerate Flosonics Medical’s mission to improve patient outcomes through non-invasive, data-driven solutions that empower clinicians to make faster, more informed decisions at the bedside.
Sepsis is a critical health challenge, affecting over 75,000 patients annually in Canada and 1.7 million in the U.S., with mortality rates that make it one of the leading causes of in-hospital deaths in
the U.S. Traditional Doppler ultrasound machines, which can assess blood flow and vascular conditions, can be cumbersome and time-intensive, making them less accessible in fast-paced environments such as emergency departments and intensive care units. FloPatch is a wireless, wearable Doppler ultrasound device that conducts rapid, repeatable hemodynamic assessments to understand a patient’s fluid responsiveness, allowing clinicians to deliver timely and effective interventions. By streamlining critical decisions, FloPatch helps healthcare providers optimize sepsis care, reduce fluid-related complications, and improve patient outcomes.
RBCx is the first tech and innovation banking arm under a Canadian bank with a dedicated life science credit advisory team that provides debt financing to pre-revenue, venture-backed life sciences companies. This type of unique servicing model provides companies like Flosonics Medical with access to capital while they scale operations and refine their innovative solutions. RBCx’s venture debt financing is designed to support venture-backed life science startups at every stage of growth, from pre-clinical to commercial. In doing so, RBCx is helping to address the significant investment gaps and funding resources available in the capital-intensive sector.

Gleamer, an artificial intelligencepowered medical imaging solutions innovator, recently announced that its advanced chest X-ray interpretation solution, ChestView, has received FDA clearance.
This announcement marks a groundbreaking advancement in chest X-ray interpretation, with a computer-aided detection (CADe) model cleared by the FDA for simultaneously detecting multiple findings, enabling direct highlighting
of regions of interest on images. This capability significantly enhances explainability and confidence in detections compared to traditional triage-focused CADt solutions on chest X-rays, setting a new standard in AI-assisted radiological detection.
ChestView further solidifies Gleamer’s leadership in AI medical imaging, uniquely covering all core imaging modalities, including X-ray, mammography, CT, and MRI. By identifying anomalies such as pleural space abnormalities, lung nodules, and other critical pathologies, ChestView aims to significantly enhance detection accuracy, reduce diagnostic delays, and improve patient outcomes. The introduction of CADe technology, such as ChestView, represents a significant advancement in medical imaging, enabling healthcare providers to manage increasing workloads efficiently, reduce oversight errors, and enhance the overall quality of patient care.
The company also acquired Pixyl and Caerus Medical, expanding its AI-powered imaging portfolio to include MRI solutions for neurology and musculoskeletal diagnostics.

LEADOPTIK, a Silicon Valley-based medical imaging company pioneering AI-enhanced real-time optical imaging at the point of biopsy, recently announced that it has entered into a research collaboration with Stanford Medicine to explore the use of AI-enhanced real-time optical imaging in lung biopsy procedures. This partnership will evaluate how high-resolution imaging can improve lesion assessment and biopsy accuracy while also contributing to the advancement of smart tools designed to support clinical decision-making.
Lung cancer remains the leading cause of cancer-related deaths world-
wide, with survival rates heavily dependent on early detection and timely intervention. However, diagnosing small, peripheral lung nodules remains a significant challenge, often leading to inconclusive biopsies and treatment delays. This collaboration aims to integrate real-time optical imaging into biopsy workflows, providing physicians with immediate insights at the point of care and enabling a deeper understanding of tissue microstructure that could support future advancements in AI-driven analysis.
Stanford Medicine’s Interventional Pulmonology Program will play a leading role in evaluating this technology’s potential for lung biopsy procedures. As a recognized leader in minimally invasive pulmonary diagnostics, Stanford will assess how high-resolution microstructural visualization, combined with emerging AI-powered image interpretation, can refine sample collection strategies and improve patient outcomes. By analyzing detailed optical imaging data in real-world clinical settings, the collaboration aims to enhance diagnostic precision and lay the foundation for more intelligent, image-guided decision-making.

Lucid Diagnostics, a commercial-stage cancer prevention medical diagnostics company, and subsidiary of PAVmed, announced that it has secured the first commercial insurance coverage for its EsoGuard Esophageal DNA Test, from Highmark Blue Cross Blue Shield. This coverage milestone supports non-invasive screening for esophageal precancer and cancer, setting a precedent for further insurance approvals.
Additionally, the company recently announced the closing of its previously announced underwritten public offering of 14,375,000 shares of its common
stock, including 1,875,000 shares issued pursuant to the exercise of the underwriters’ option to purchase additional shares, at a public offering price of $1.20 per share. The total net proceeds from the offering, after expenses, total approximately $16.1 million. The company intends to use the net proceeds from this offering for working capital and general corporate purposes.
Lucid is focused on the millions of patients with gastroesophageal reflux disease (GERD), also known as chronic heartburn, who are at risk of developing esophageal precancer and cancer. Lucid’s EsoGuard Esophageal DNA Test, performed on samples collected in a brief, noninvasive office procedure with its EsoCheck Esophageal Cell Collection Device, represent the first and only commercially available tools designed with the goal of preventing cancer and cancer deaths through widespread, early detection of esophageal precancer in at-risk patients.

Chicago, IL-based Madison Scientific (MadSci), a private medtech company developing smart electro-mechanical solutions for patients with neurologic conditions, announced that it has secured $7 million in a fully subscribed seed financing round. The round was co-led by WARF Ventures, the Isthmus Project, and a successful medical device entrepreneur/investor, with participation from other institutional investors, family offices, and angel investors. Funds will be used to grow the MadSci team, expand preclinical testing and advance prototype development of the company’s innovative SmartShunt Hydrocephalus Management System.
The company’s SmartShunt System is designed to integrate diagnostic and therapeutic functions to enable
personalized and informed hydrocephalus management. The system features a proprietary valve, state-of-the-art intracranial pressure (ICP) sensor, and advanced algorithms to optimize shunt drainage to meet each patient’s needs. The in-development SmartShunt System aims to leverage ICP readings to reduce life threatening shunt failure, while also facilitating on-demand and informed patient management, reducing uncertainty for patients and their families.
In addition to this financing, the company was previously awarded several non-dilutive grants, including a Phase I Small Business Innovation Research Program (SBIR) grant from the National Institutes of Health (NIH) and a Phase 2 SBIR Grant from the National Science Foundation (NSF).
Hydrocephalus is an incurable neurological condition impacting approximately one million children and adults in the U.S. today. The condition is caused by a buildup of cerebrospinal fluid (CSF) in the brain’s ventricles, which can increase intracranial pressure and lead to neurological problems or even death if untreated. The current standard of care is the surgical placement of a permanent shunt to drain CSF from the central nervous system to other parts of the body. However, traditional shunts, which have seen limited innovation since their introduction almost 75 years ago, are prone to failure, with nearly 40% failing within the first year and the majority failing within two to three years.

Austin, TX-based stroke company MicroTransponder has announced a $65 million Series F financing round, The round was led by US Venture Partners and the syndicate of current investors—Osage University Partners, Action Potential Venture Capital, GPG Ventures, The Vertical Group,
and Exceller Hunt Ventures, as well as two new investors, Gilde Healthcare and Longitude Capital.
The company’s FDA-approved Vivistim Paired VNS System is the first and only FDA-approved intervention clinically proven to help chronic ischemic stroke survivors regain two to three times more upper extremity function than high-intensity stroke therapy alone, after six weeks of in-clinic occupational or physical therapy.
Approved with the Breakthrough Device Designation, Vivistim is an implanted device that a therapist pairs with vagus nerve stimulation (VNS) during high-repetition, goal-oriented therapy tasks. Paired VNS Therapy helps increase neuroplasticity, so stroke survivors can make unprecedented gains with their hands and arms during activities of daily living. Real-world data presented at the 2025 International Stroke Conference by two independent comprehensive stroke centers complements the published clinical studies, proving the effectiveness of Paired VNS Therapy for chronic stroke survivors.

Moon Surgical, a French-American pioneer in surgical innovation, recently announced FDA clearance for ScoPilot. Enabled by NVIDIA Holoscan, a real-time sensing platform designed to develop and deploy applications based on AI in the operating room, and running locally on Moon Surgical’s Maestro System, ScoPilot is the first AI application to run intraoperatively on any commercially available surgical robotic platform with Holoscan.
ScoPilot ensures a stable and ideal view during surgery, and aims to enhance control, safety, and efficiency in the operating room. On-demand and easy to use, ScoPilot allows the laparoscope attached to a Maestro System to follow
a desired instrument tip seamlessly. The surgeon controls laparoscope positioning, without disengaging from the instruments in their hands, helping maintain surgical flow and focus.
Moon Surgical’s commercial Maestro System, FDA-cleared in June 2024, has been used to treat over 1,100 patients across the U.S. and Europe, spanning general, bariatric, gynecologic, and urologic surgeries. With its unique and extensive ambient sensing, paired with NVIDIA-accelerated computing and AI, Maestro generates an unprecedented surgical dataset including multi-modal sensing and kinematics—thereby driving the development of next-generation physical AI capabilities that enhance surgical care both in the operating room and across the broader perioperative workflow. (Also see LSI Alumni Milestones: June and July 2024, The Lens, September 2024; and Power Moves, The Lens, January 2025.)

Haifa, Israel-based Nurami Medical, developing products for neurological surgeries, specifically addressing cerebrospinal fluid leakage, announced that it has closed a $30 million funding round. The proceeds from the financing will be used for the commercialization of their FDA 510(k)-cleared biomimetic scaffold product, ArtiFascia, that enhances tissue ingrowth to address cerebrospinal fluid (CSF) leaks, and for the clinical development of ArtiFix, a three-in-one sealant patch with a scaffold layer for use in cranial and spinal repair.
The new investment will be used to nearly double the company’s workforce and expand its operational capabilities. Plans include the construction of a commercial manufacturing facility, a new R&D laboratory, and high-grade clean rooms that will support production. These enhancements are expected
to position Nurami Medical to scale its operations as it transitions from development to full-scale production and commercialization.
Nurami Medical’s technology addresses a critical challenge in neurosurgery—CSF leakage—which can prolong hospital stays and increase the risk of complications. With a significant market in neurosurgical procedures valued at approximately $1 billion and potential applications in additional clinical areas, the company’s advancements could have far-reaching implications in the field of medical innovation. The company was founded in 2014 by Nora Nseir and Dr. Amir Bahar.

Powerful Medical, a leader in AIdriven cardiovascular diagnostics, announces that its PMcardio STEMI AI ECG model has been granted Breakthrough Device Designation by the U.S. FDA. This designation recognizes PMcardio as a breakthrough technology for the detection of ST-elevation myocardial infarction (STEMI) and STEMI equivalents—a life-threatening cardiac condition requiring immediate intervention.
Every 40 seconds, someone in the U.S. suffers from a heart attack, where rapid diagnosis is crucial to saving heart muscle and preventing long-term damage, often leading to higher mortality rates. The electrocardiogram (ECG) remains the primary tool for rapid diagnostics, yet the standard of care often fails to detect heart attacks accurately and timely, resulting in critical delays in treatment.
PMcardio is the only solution capable of detecting both STEMI and STEMI equivalents on the ECG—aligning with the emerging emphasis on a paradigm shift towards occlusion myocardial infarction and bridging a vital gap in
early, precise heart attack diagnosis. The company’s platform helps ensure rapid and precise patient triage, driving better outcomes and optimizing operational efficiency across the healthcare system.

Shanghai, China-based Pulnovo Medical, which is innovating medical devices for pulmonary hypertension (PH) and heart failure (HF), has announced the closing of nearly $100 million Series C financing. The round was co-led by Qiming Venture Partners and existing shareholder Lilly Asia Ventures, with existing investors OrbiMed and Gaorong Capital participating on a super pro-rata basis. The financing round was oversubscribed by two times, marking one of the largest fundraises in the innovative Asia-Pacific medtech sector in recent years.
The proceeds will be used to advance Pulnovo Medical’s global clinical trials, international business expansion, and strategic initiatives. The company has two upcoming FDA trials for Group I PH and Group II PH (chronic heart failure combined with PH) planned this year.
Founded in 2013, Pulnovo Medical is a pioneering medtech company focused on developing superior therapeutic solutions for various stages of HF, to enhance patient outcomes. Pulnovo Medical’s core product, PADN (Pulmonary Artery Denervation) technology, is an innovative and effective minimally invasive treatment for PH that has successfully obtained market approval.
PH is a condition that can lead to heart failure and mortality, with limited treatment options. PADN uses radiofrequency ablation to target the pulmonary vascular endothelial sympathetic nerve, effectively reducing pulmonary artery pressure and slowing disease progression.

Qaelon Medical, a pioneer in real-time surgical leak detection and insufflation, and RevMedica, an innovator in surgical stapling solutions, have announced a non-exclusive data partnership to improve stapler performance and clinical outcomes in gastrointestinal surgery.
By integrating RevMedica’s advanced stapler technology with Qaelon’s realtime leak detection and performance data, the collaboration aims to accelerate product development and product validation; assess the impact of varying tissue properties on staple line performance; and assist with regulatory approvals and claims development.
Qaelon Medical is pioneering the first and only platform that combines real-time detection of life-threatening surgical leaks with state-of-theart constant flow surgical insufflation. The company’s breakthrough solution empowers surgical teams with definitive leak test results during surgery, enabling immediate corrective action before the patient leaves the operating room.
RevMedica’s new Endo Stapling Platform provides digital precision and real-time feedback for minimally invasive procedures by optimizing staple formation and tissue management. Qaelon Medical’s unique solution provides real-time, quantifiable leak detection data that empowers surgeons to take immediate corrective action before the patient leaves the operating room.
Both RevMedica and Qaelon Medical technologies have been selected for the FDA’s Safer Technologies Program (STeP), reserved for “medical devices that are reasonably expected to significantly improve the safety of currently available treatments.” (Also see LSI Alumni Milestones: Wins Announced in January, The Lens, February 2025.)

Grand Rapids, MI-based Shoulder Innovations, a leader in shoulder replacement technology, recently announced the closing of a $40 million Series E equity financing round. The financing was led by existing investor U.S. Venture Partners, with support from additional existing investors including Gilde Healthcare Partners, Gilmartin Capital, and Aperture Venture Partners. New investors Arboretum Ventures and Sectoral Asset Management also participated.
Proceeds from the financing will be used primarily to advance commercialization of the company’s InSet Total Shoulder and Reverse Shoulder Arthroplasty Systems and support new product development, as well as for general corporate purposes.
Leveraging its breakthrough, patented InSet glenoid design, Shoulder Innovations developed a shoulder replacement implant system focused on improving outcomes related to the greatest cause of anatomic shoulder replacement failure: glenoid loosening. InSet technology has been shown in testing to significantly reduce glenoid implant micro-motion and simplifies the surgical technique, potentially reducing complications or increasing implant longevity. The company has also developed the anatomic reverse shoulder system, designed to work with and maximize the available rotator cuff instead of substituting for it.
Sofinnova Partners, a leading European life sciences venture capital firm based in Paris, London, and Milan, had announced it has raised €1.2 billion across its platform of investment strategies over the past year, including more than €1 billion secured in the last quarter. This achievement brings

the firm’s assets under management (AUM) to more than €4 billion, solidifying its position as a leading backer of transformative innovation in healthcare and sustainability. With the new funds, the firm plans to back 50 to 60 new companies and leverage Sofinnova.ai to accelerate disruptive life sciences investments.
Sofinnova’s latest funds have attracted significant commitments from leading institutional investors, sovereign wealth funds, corporates, and family offices, underscoring strong investor confidence in the firm’s ability to identify and nurture high-potential opportunities. Today, the firm comprises over 80 professionals, including 25 investment partners with deep sector expertise. It is an international team, spanning 24 nationalities across three European offices, with presence in key innovation hubs worldwide. (Also see Long Game Legends: Makower and Papiernik’s Playbook for Success, The Lens, December 2024.)

Brussels, Belgium-based Synergia Medical has announced that its NAO. VNS device, successfully implanted in five patients at UZ Gent and Cliniques universitaires Saint-Luc in Belgium, has met the primary safety endpoint in the AURORA first-in-human clinical study with no serious adverse events and a 100% implantation success rate over three months. Synergia is pursuing
its first indication to treat drug-resistant epilepsy (DRE) via vagus nerve stimulation (VNS). Three months post-implantation, results confirm strong safety, robust device performance, and positive patient outcomes. Synergia plans to now move into a pivotal FDA and CE trial.
Synergia Medical’s technology is a next-generation neuromodulation platform that eliminates the main metal components of traditional devices, ensuring immunity to electromagnetic interference (EMI). It enhances patient safety and care with full compatibility across MRI and other EMI sources (e.g., electrosurgery, defibrillators) and features a fast-recharge battery with an expected lifetime of 15 years with just one minute of daily charging. The NAO platform holds the potential to optimize therapy by remaining fully active during imaging, allowing physicians to adjust treatment in real time while observing brain responses, while its long-lasting battery supports energy-intensive stimulation for more effective therapeutic outcomes.
Epilepsy affects 50 million people globally and is the fourth most common neurological disorder. When two anti-epileptic drugs fail, DRE (drug-resistant epilepsy) is diagnosed, affecting 30% of patients with severe health, personal, and economic impacts. DRE patients face a 10 times higher risk of premature death and increased depression. VNS, which involves the implantation of a small pacemaker that delivers mild electric signals to the vagus nerve, is a clinically proven solution for DRE that helps reduce seizures, improve mood, and enhance quality of life.
Synergia Medical has been supported by both public and private investors since its founding in 2015, including funding from the Wallonia Region and the European Union’s EIC program.
London-based THENA Capital, the UK’s first specialized early-stage medtech venture fund led by an all-female team, has announced the first close of its £50M fund, securing £27M from notable investors. The firm aims to address critical funding gaps in early-stage medtech while bringing muchneeded diversity to the venture capital landscape.

Founded by Tatum Getty, Pamela Walker Geddes, and Esther Reynal de St Michel Richardot, THENA Capital represents a convergence of three different nationalities (French, American, and Canadian) with complementary backgrounds in building and scaling health, wellness, and healthcare companies. The founding team conceived the fund five years ago with a mission to change the face of venture capital and transform the future of healthcare.
THENA Capital will target British startups developing digital healthcare solutions and medical devices, with a particular interest in innovations addressing women’s health challenges. Their portfolio already includes several notable femtech investments including Daye and Samphire Neuroscience
The fund has attracted significant support from institutional and individual investors, including the British Business Bank’s Enterprise Capital Funds programme, Firebird Collective, This Day Foundation, Patrick Healy of Hellman & Friedman, Lastminute.com co-founder Baroness Martha Lane Fox, and various individuals from private equity and pharmaceutical firms.

Valcare Medical, a leading innovator in transcatheter-based mitral solutions, has announced that the FDA has approved its AMEND TransSeptal System for investigational device
exemption (IDE) application to commence an early feasibility study.
The AMEND Trans-Septal System is a trans catheter device intended for treatment of mitral insufficiency with a semi rigid, closed, D-shaped annuloplasty ring that mimics today’s surgical gold standard.
The EFS study will evaluate the safety and functionality of the AMEND Trans-Septal System in the treatment of subjects with symptomatic moderate-to-severe (3+) or severe (4+) functional mitral regurgitation that are anatomically suitable for transcatheter mitral annuloplasty. The EFS will enroll up to 15 subjects in seven investigational centers in the U.S. The study is expected to begin treating patients in the first half of 2025, paving the way for a pivotal study anticipated in 2027.
The primary and secondary endpoints of this trial include safety and device performance assessments such as technical success of ring delivery, implantation, and delivery system retrieval. Patient outcomes such as stroke, myocardial infarction, non-elective cardiovascular surgery for device-related complications, and all-cause mortality are to be reported at 30 days and six months postimplantation.

Vektor Medical recently announced that it had closed an oversubscribed $25 million Series A funding round to scale vMap, its AI-powered arrhythmia mapping technology. The round was led by TVM Biosciences and Solas Bioventures. The investment will support commercialization efforts, additional clinical studies, and expansion into the European market. (Also see Power Moves, this issue; and LSI Alumni Milestones: Wins Announced in November, The Lens, December 2024.)
3M
Alcon 11, 21, 30
Asclepii
Bpifrance 9

March 16th - 20th, 2026
Waldorf Astoria, Monarch Beach, Dana Point, CA


