2025 Accelerator Impact Report TRANSLATING


Letter from
Leadership
Fifteen years ago, the Dana-Farber Accelerator launched with a bold vision: to speed the journey from the discovery of new cancer therapies to patient care. Today, that vision is becoming reality. Our portfolio includes technologies now in clinical trials and a pipeline poised to deliver the next breakthroughs.

Tolikas, PhD, MBA SVP & Chief Innovations Officer
Despite the challenges our scientific community is facing, over the past year, DFCI researchers, clinicians, and partners remained focused to overcome obstacles and drive remarkable progress. Together, we advanced projects across the spectrum of cancer therapeutic development from novel strategies to enhance the host anti-tumor immune response to approaches that inhibit the function of oncogenic drivers. One Accelerator-funded therapy moved into Phase I trials

Christiana Iyasere, MD, MBA Senior Director Accelerator
for hard-to-treat cancers, a milestone that reflects the power of collaboration across academic medical centers and philanthropic organizations.
Rooted in this collaborative foundation, the Accelerator combines state-of-the-art science with business and IP strategy to move the further development of discoveries forward faster. Looking ahead to 2026, we will enable deeper partnerships among faculty within Dana-Farber foster interinstitutional collaboration and continue to engage industry leaders and investors to help us catalyze the translation of innovative science into therapies.
This report highlights the groundbreaking projects funded by the Accelerator, including those on the cusp of significant clinical impact. We are deeply grateful to our entire DFCI community, generous donors, and industry partners. Your support makes this progress possible towards fulfilling DanaFarber’s mission to advance our understanding of cancer and accelerate the development of new treatments and cures for patients everywhere. Thank you for your partnership.
Sincerely,
MaryTolikas
ChristianaIyasere


Accelerator Funded Technologies

Manipulating the Immunopeptidome for Rapid Tumor Elimination
Ellis Reinherz, MD
Advancing Small Molecule Therapies to Target MUC1-C in Cancer
Donald Kufe, MD
A New CAR T Strategy for Kidney Cancer Heads to Patients
Wayne Marasco, MD, PhD
2021 Project Update
Advancing CAR-NK Cell Therapy for LongTerm Cancer Control
Eric Smith, MD, PhD
Accelerator Success Story
Liquid Biopsy for Myeloma: From Discovery to Patient Care
Irene Ghobrial, MD

Accelerator Projects
Small Molecule
2010
2013
Bromodomain inhibitors for cancer therapy 2015 Novel inhibitors of the deubiquitinating enzymesto target leukemia oncogenes
Targeting novel oncogenic kinases in breast cancer
DUB Inhibitors in cancer therapy
2016 Targeted inhibitors for breast and ovarian cancers
2017 Small molecule degraders for the treatment of mantle cell lymphomas
Development of a novel p300/CBP inhibitor targeting oncogenic transcription 2017
2019 Chromatin reader protein inhibitor as novel therapyin Bcell neoplasms
2020
Chemical manipulation of “undruggable” proteins to treat obesity and cancer
2020 Discoveryof mutant-selective allosteric inhibitorsof cancer targets
Development of next generation allosteric EGFR inhibitors
Targeting TIRR dependencyin p53 heterozygouscancers
Small moleculestargeting chromatin regulatory complexes
Targeting creatine kinase in colon cancer
Small molecule targeting oncogenic driver
Manipulating the Immunopeptidome for Rapid Tumor Elimination
Accelerator Projects
Translation of Galectin-1 blockade to the clinic
Anti-IL23R-mediated cancer immunotherapy
Novel CART Cell factories to achieve B-CLL cures
Gene modified hematopoietic stem cellsas as CAR factories
CD4+ cytotoxic T lymphocyte (CTL) therapy to cure refractory B-cell malignancies
Dual-targeted CAR to cure Multiple Myeloma
2022 Antibody discoveryplatform in ovarian and pancreatic cancer
Engineered NK CARsfor enhanced immunotherapy 2023
TransTAC platform for targeted protein degradation
platform for degrading autoimmune targets
Memory like NK cells to eradicate MRD cellsin high-risk myeloid malignancies
Armored CAR T for renal carcinoma
Engineered
Dynamic BH3 profiling as predictive biomarker
Stabilized mRNA for improved gene therapy and vaccines
Predicting disease byimmune and circulating tumor cell profiling
Investment for Impact
$9 million of funding to the Accelerator since 2010 has enabled four new companies, four clinical trials and more than $26 million in follow-on funding.



32 Projects Funded



4 Clinical Trials




35 IP Filings




4 Companies Launched




Total Company Valuation >$1Bn


Enabling Funding $26M


Manipulating the Immunopeptidome for Rapid Tumor Elimination
Despite major advances in immunotherapy, many cancers still evade immune attack. Ellis L. Reinherz, MD, a researcher at Dana-Farber and Professor of Medicine at Harvard Medical School, and his team aim to change that by rethinking how T cells recognize tumors. Backed by a SPARK Accelerator grant, they’re leveraging decades of expertise in T cell biology along with chemical synthesis and structural biology to design a novel strategy that strengthens the immune system’s ability to target cancer. Their approach focuses on enhancing the role of T lymphocytes, particularly cytolytic T cells (CTLs), which recognize and destroy tumor cells displaying “foreign” antigens. These antigens are short protein fragments presented by HLA molecules—specialized proteins on the cell surface that show these fragments to T cells so they can detect and respond to threats. By improving this recognition process, the team hopes to overcome key limitations of current immunotherapies and expand their reach to more patients.
This work builds on a central principle in cancer research: harnessing the immune system to eradicate tumors. T lymphocytes have been at the heart of many immune-based therapies, including checkpoint inhibitors drugs that release the “brakes” on T cells and have transformed treatment for cancers like melanoma and lung cancer. Reinherz’s strategy seeks to push this paradigm further by improving how T cells identify and attack cancer cells, opening new possibilities for tumors that currently resist even advanced immunotherapies.
Understanding Tumor Immune Evasion
Understanding why tumors evade immune detection is critical to this effort. Many cancers manipulate the peptides they display to avoid recognition. Some suppress potential foreign targets from the outset, while others adaptively restrict mutant peptide expression once CTLs begin their attack. Using mass spectrometry, the team analyzed data from more than 5,000 patient samples and found that few, if any, tumor-associated antigens or neoantigens they assessed are displayed. This means the immune response is severely limited for most cancers, underscoring the need for new strategies to overcome this barrier.
Modifying the Immune Response to Cancer
To address the stealthiness of cancer cells, the team is developing a method to increase the diversity of
David Barbie, MD
Reinhold, PhD Team Members

Ellis Reinherz, MD
Project lead Professor Chief of Immunobiology Lab


Bruce
Director of the Lowe Center for Thoracic Oncology, DanaFarber Professor of Medicine, Researcher, Dana-Farber Principal Associate in Medicine
peptides associated with HLA molecules on tumor cells, thereby improving the likelihood that our immune system can recognize tumors as foreign and mount an immune attack. Their approach involves the identification of drug-like small molecules that can bind to HLA molecules and dramatically alter the array of peptides presented to CTLs. This increases the chance that immunogenic peptides will be expressed and subsequently recognized and killed by our immune system. Hundreds of new peptides may be expressed on tumor cells following treatment with novel small molecules identified by Reinherz’s team, potentially generating a large, diverse T cell response and increasing the probability of therapeutic benefit.
Preclinical data indicate that treatment with newly identified small molecules can cause a significant shift in the immunopeptidome the full set of peptides presented by HLA molecules on cancer cells. Proof-ofconcept has been achieved with one HLA molecule. Using AI-guided drug design, the team plans to extend this approach to a total of 40 HLA molecules, covering virtually all racial and ethnic populations globally.
Targeted Delivery with Antibody-Drug Conjugates
To ensure the specific delivery of these compounds only to tumor cells, the team is developing antibodydrug conjugates (ADCs) designed to bind to specific cancer cell surface markers and release the small molecule as a pharmaceutical payload. This targeted delivery system will alter peptide display selectively on tumor cells. By creating ADCs that target the 12 most common cancers, the team is building a generic toolkit for cancer therapies.
Toward a Broadly Applicable Therapeutic Platform
With this conjoint approach—combining peptide modulation and targeted delivery—the team aims to change the landscape of cancer treatment. In particular, the researchers hope to make cancers generally thought to not respond to immune based therapy such as glioblastoma, pancreatic, ovarian, and breast cancers, among others, immunotherapy-responsive. The project benefits from Dana-Farber’s broad cancer expertise and collaborative research environment, involving oncologists and researchers across multiple cancer types.

Andrew Parkins, PhD
Research Fellow in Medicine, DanaFarber



Koji Haratani, MD, PhD
Research Fellow in Medicine, Dana-Farber

Jonathan DukeCohan, PhD
Principal Researcher, Dana-Farber Principal Associate in Medicine

Robert Mallis, PhD
Instructor in Dermatology, Dana-Farber
Lyn Jones, PhD
Director of the Center for Protein Degradation, DanaFarber Biological Chemistry and Molecular Pharmacology
Advancing Small Molecule Therapies to Target MUC1-C in Cancer
Donald Kufe, MD, Distinguished Physician at Dana-Farber and Professor of Medicine at Harvard Medical School, has spent decades investigating the MUC1-C protein, one of cancer’s most elusive drivers, and has gleaned essential insights into how certain cancers grow, evade treatment, and resist immune attack. Today, his lab is translating that foundational research into a new therapeutic strategy developing small molecule inhibitors aimed at disabling MUC1-C’s cancer-promoting functions. This work has been selected for funding by the Dana-Farber Accelerator.
Kufe first identified the MUC1 gene and revealed that the encoded C-terminal subunit, MUC1-C, acts as an oncoprotein. It disrupts normal cell architecture, activates growth-promoting signals, and fuels inflammation all of which contribute to cancer progression and resistance to therapy. “MUC1-C drives malignant transformation across pan-cancers,” Kufe explains. “We’re now working to block its activity with small molecules.”
A New Therapeutic Strategy
The Kufe Lab is focused on designing small molecules that target the intracellular domain of MUC1-C— the region responsible for its oncogenic signaling. Unlike antibody-based therapies or CAR T-cell approaches directed against proteins on the cancer cell surface, these small molecules are intended to work inside the cell, where MUC1-C exerts its effects.
This approach represents a shift in strategy, offering a potentially more direct way to interfere with MUC1C’s role in cancer biology. By focusing on the intracellular domain, researchers hope to address mechanisms of resistance and tumor survival that are not targeted by existing therapies.
Why MUC1-C Matters
MUC1 and particularly MUC1-C is overexpressed in at least 8–10 major tumor types, including breast, lung, pancreatic, ovarian, colorectal, prostate, gastric, renal, and head and neck cancers. Together, these represent the majority of adenocarcinomas and many aggressive solid tumors. Some estimates suggest that MUC1 is overexpressed in more than 80% of adenocarcinomas, translating to millions of cases worldwide.
This is significant, since MUC1-C supports cancer cell survival by activating inflammatory pathways and promoting a stem-like state that makes tumors harder to treat. It also contributes to resistance against targeted therapies, such as those aimed at EGFR and HER2.
Team Members


Donald Kufe, MD
Distinguished Physician, Dana-Farber Professor of Medicine
2024 Accelerator Project
Importantly, MUC1-C is linked to processes like epithelial-mesenchymal transition (EMT), epigenetic reprogramming, and self-renewal hallmarks of aggressive and treatment-resistant cancers. These features make it a compelling target for drug development, especially in cancers where conventional therapies have limited impact.
Translating Discovery into Impact
The project was initially conducted by Atrayee Bhattacharya, PhD, who oversaw early development efforts. Mai Moriya has now taken over performing these studies, which has completed its SPARK-funded phase and is advancing toward further preclinical development. Specifically, the team is working to refine its approach and move toward testing in relevant cancer xenograft models. While details of the molecules under development remain confidential, the Lab’s goal is to identify inhibitors that can be used across multiple cancer types and potentially in combination with other therapies.
Complementing Existing Therapies
Other therapies targeting MUC1-C such as antibody-drug conjugates and CAR T-cell therapies are in various stages of clinical and pre-clinical development. These approaches primarily focus on the extracellular domain of the protein. Small molecule inhibitors offer a complementary strategy, one that could be used alongside existing treatments or in cancers where immune-based approaches are less effective.
Given MUC1-C’s role in maintaining the cancer stem cell state and promoting resistance, these inhibitors could have broad applications. They may be especially valuable in cancers that are refractory to current therapies, offering a new option for patients with limited treatment choices.
Looking Ahead
With growing recognition of MUC1-C’s role in cancer resistance and progression, the development of targeted small molecules represents a critical step forward. For patients with few options, this research could open the door to more effective and personalized therapies.
Kufe’s decades-long commitment to understanding MUC1-C is now converging with a translational push to bring new treatments to the clinic. “We’ve spent years defining the biology,” he says. “Now we’re focused on turning that knowledge into therapies that can make a difference for patients.”



Atrayee Bhattacharya, PhD
Mai Moriya, MD, PhD Assistant Professor, The University of Texas Health Science Center at Tyler
Research Fellow in Medicine, Dana-Farber
A New CAR T Strategy for Kidney Cancer Heads to Patients
CAR T cell therapy is a form of immunotherapy in which a patient’s own T cells are genetically engineered with a chimeric antigen receptor (CAR) that enables them to recognize and kill cancer cells. This approach has produced dramatic remissions in certain blood cancers, leading to several FDA approvals. But for solid tumors the vast majority of cancers success has been far more limited. Funding from the Dana-Farber Accelerator helped support manufacturing expenses at Dana-Farber’s Cell Manipulation Core Facility costs not covered by other grants.
“Solid tumors are not the same battlefield as leukemias,” says Wayne Marasco, MD, PhD, Principal Investigator, Cancer Immunology and Virology, Dana-Farber Cancer Institute, who has been studying new ways to battle solid tumors with CAR T therapies for over two decades. “You have to penetrate tissue, overcome tumor heterogeneity where tumor markers are shared on normal cells, and deal with an immunosuppressive microenvironment. All three make the challenge much greater.”
Clear cell renal cell carcinoma (ccRCC), the most common form of kidney cancer, accounting for ~85% of all kidney cancer cases, exemplifies the problem. For these patients, existing treatments offer limited options once the disease has spread.
DFIR CARs: A Triple Innovation
To tackle these barriers, Marasco’s lab has developed a novel therapy called DFIR CARs short for dualtargeted, fine-tuned, immune-restoring CAR T cells with a primary focus on treating ccRCC. The design combines three breakthroughs in one therapy:
• Dual targeting of two renal cancer proteins to cover tumor heterogeneity and minimize tumor escape.
• Fine-tuned receptor design that recognizes only high-density tumor-associated antigens (TAA) on the cancer cells but not low-expressing TAAs on normal tissue.
• Immune restoration through secretion of immune checkpoint inhibitors (ICIs) directly into the tumor microenvironment to revive anti-tumor activity where it is typically suppressed.
“In one cell, we combined three solutions to three of the biggest obstacles in solid tumor CAR T therapy,” Marasco says.
A Professional—and Personal—Mission
Team Members

Wayne Marasco, MD, PhD
Professor, Department of Cancer Immunology and Virology, DanaFarber

Danielle Braun, PhD
Research Scientist, Dana-Farber

2024 Accelerator Project
Marasco has been steadily refining a CAR T design aimed specifically at solid tumors. “The whole idea began in a hospital bed right after I was diagnosed with renal cell carcinoma,” Marasco explains, adding that he wrote the first grant for this work while in the hospital.
He and his team have worked to refine the DFIR CAR strategy, publishing foundational studies and testing the approach in animal models and patient-derived tumor samples. Analysis of more than 200 patient tumors confirmed the relevance of the dual-target strategy in all four stages of renal cell carcinoma and the safety of the fine-tuned receptors.
Preparing for the First Clinical Trial
The team plans to finalize its Investigational New Drug (IND) application for the FDA, looking to launch a Phase I clinical trial in the near future. The study will focus on patients with advanced ccRCC, who progressed after prior treatment with at least one PD-1/PD-L1 immune checkpoint inhibitor, as these therapies rarely achieve lasting remission, even when included in combination therapies.
Collaborators now preparing for the trial include clinical leadersAnthony D’Amico, MD, PhD, chief of Genitourinary Radiation Oncology at the Brigham and Women's Hospital (BWH) and Dana-Farber, and Vincent Su, MD, medical oncologist and clinical/translational investigator inthe Lank Center for Genitourinary Oncology at Dana-Farber. Other essential team members include pathologist and principal investigator Sabina Signoretti, MD, of Dana-Farber and BWH, and instructor Yufei Wang, PhD, who has led much of the preclinical science from Marasco’s lab.
“This is truly a homegrown therapy, conceived here and now heading to patients,” Marasco says. “The Accelerator has been essential in getting us to this point.”
While Accelerator support has been crucial, Marasco emphasizes that the upcoming trial will require additional fundraising to cover the full scope of costs. Still, the therapy’s path forward is now clear.
“Renal cell carcinoma changed my life,” he reflects. “My hope is that this work will change the lives of patients who are facing what I once faced.”


Jane Liang, PhD
Theodore Huang, PhD Researcher/Biostatistician, Kaiser Permanente Northern California, Division of Research
Yufei Wang, PhD
Instructor in Medicine Immunology and Virology, Dana-Farber

Principal Biostatistician, Vertex Pharmaceuticals


Advancing CAR-NK Cell Therapy for Long-Term Cancer Control
In 2021, the Dana-Farber Cancer Institute (DFCI) Accelerator provided critical funding for a groundbreaking project led by Eric Smith, MD, PhD, Director of Translational Research, Immune Effector Cell Therapies at Dana-Farber. The project aimed to solve a major challenge in CAR T cell therapy: the limited persistence of CAR effector cells, which often leads to relapse. While CAR T therapies have revolutionized treatment for advanced blood cancers, their long-term efficacy is constrained by the eventual disappearance of therapeutic cells. Smith’s team is pioneering a novel approach leveraging hematopoietic stem cells (HSCs) to create a durable, in vivo CAR-Natural Killer (NK) cell therapy platform.
“By using hematopoietic stem cells, we wanted to think differently about how to create a therapy that could provide continuous, long-term pressure on tumors,” Smith explains.
The Problem: Persistence and Safety in CAR Therapies
CAR T cell therapies engineer a patient’s immune cells to attack cancer but face two hurdles: limited persistence and safety concerns like cytokine release syndrome (CRS) and neurological toxicities. Natural killer cells offer a safer alternative due to lower toxicity risk, but they struggle even more with persistence. “NK cells are great at killing tumors, but they don’t stick around,” Smith says. His team sought to address these issues by developing a platform that continuously generates CAR-NK cells in vivo, using the body’s own hematopoietic stem cells as a “factory.”
The Solution: A CAR Factory Platform
The CAR Factory Platform uses patented gene-engineering technologies. By introducing a diseasespecific CAR gene into hematopoietic stem cells, the platform enables these stem cells to selectively differentiate into CAR-NK cells over time. This mimics natural hematopoiesis and ensures a steady, longterm supply of CAR-NK cells to target and destroy tumor cells. “Hematopoietic stem cells are incredibly powerful because they can repopulate the entire immune system,” says Smith. “By engineering these stem cells to produce CAR-NK cells, we’ve created a system where new immune effector cells are made every day, potentially surveilling for recurrence of the tumor for the rest of the patient’s life.”
To ensure safety, the team developed a proprietary synthetic promoter system that activates the CAR gene only after stem cells become NK cells. “This was critical,” Smith notes. “We didn’t want CAR
Team Members
Eric Smith, MD, PhD

Director of Translational Science, Immune Effector Cell Therapies, and Faculty Director, IMmunotherapy
Platform for Antibody and CAR Therapeutics discovery and Translation (IMPACT2), Dana-Farber




Cedric Louvet, PhD Instructor, Dana-Farber
Yunxin Chen, MBBS, MRCP(UK), FRCPath(UK) Senior Consultant, Hematology, Singapore General Hospital
Erin Rosenberg Medical Student, Boston University
Emma Bucey Master's Student, Johns Hopkins University
2021 Accelerator Project Update

expression in the wrong cell type. Our system ensures normal immune lineage differentiation and function while reducing toxicity.”
Progress and Impact of Accelerator Funding
Accelerator funding in 2021 was instrumental. “It allowed us to generate preliminary data to secure additional grants from the Mathers Foundation and ultimately an R01 from the NIH,” says Smith. “It was truly the launchpad for this work.”
Using the funding, the team screened hundreds of DNA regulatory elements they identified, in combinations they term “assemblies,” to ensure high CAR expression in NK cells while remaining silent in stem cells. They validated findings in humanized mouse models, showing engineered HSCs could differentiate into many immune lineages with CAR expression limited to daughter NK cells. They further demonstrated that these HSC CAR-NK factories effectively controlled tumor growth.
Results have been promising. In preclinical models of multiple myeloma, CAR-NK cells derived from engineered HSCs showed strong anti-tumor activity and long-lasting protection. The team is now optimizing the platform and preparing for an Investigational New Drug (IND) filing, with plans to initiate clinical trials in multiple myeloma by 2027
Vision for the Future
Smith envisions this CAR Factory Platform transforming treatment for blood cancers, solid tumors, and autoimmune diseases. “This platform has the power to generate a ‘pipeline in a product’ opportunity while addressing unmet needs across many diseases,” he says. “We’re excited about integrating this approach into existing stem cell transplant protocols, which are already standard of care for some cancers.” The long-term goal is to simplify therapy, potentially delivering the CAR gene directly into patients’stem cells through in vivo viral delivery, without transplant conditioning making it more accessible.
Smith credits the success to his team, including Cedric Louvet, PhD, and external collaborators like Richard Koche, PhD, at Memorial Sloan Kettering Cancer Center. “This has been a true team effort,” Smith emphasizes. “The Accelerator funding was the catalyst that made this possible, and we’re excited to see where it takes us next.”




Evelyn Mesler
PhD Student, Albert Einstein College of Medicine
Douglas Russo, MS
Computational Biologist, Dana-Farber
Felicia Ciuculescu, MD
Tech Transfer Director Cell Manipulation Core Facility, Dana-Farber
Myriam Armant, PhD
Director of the Therapeutic Innovation Lab, Boston Children's Hospital




Richard Koche, PhD
Director of Computational Epigenetics, Memorial Sloan Kettering Cancer Center
Justin Bagger
Research Associate, Dana-Farber
Poulami Das, PhD Scientist, Dana-Farber
Rafael Irizarry, PhD
Chair and Professor, Department of Data Science, Dana-Farber
Liquid Biopsy Reaches Patients
Liquid Biopsy for Myeloma: A Game-Changing Innovation Now Serving Patients
From groundbreaking research to real-world care—an exciting leap forward in cancer diagnostics
Last year, we featured this technology as a promising innovation poised to transform care. Today, we are thrilled to share an exciting update: the liquid biopsy technology for multiple myeloma, developed by Irene Ghobrial, MD, Director of the Center for Early Detection and Interception of Blood Cancers and CoLeader of the Lymphoma/Myeloma Cancer Center Program at Dana-Farber, and her team is now licensed to Predicta Biosciences, where it is available to patients in a CLIA-approved laboratory setting.
This breakthrough supported by Dana-Farber’s Accelerator Program and partners including the Broad Institute, The Engine, and Illumina Ventures marks a major step forward in precision medicine. Instead of relying on painful bone marrow biopsies, patients can now benefit from a simple blood draw that delivers a comprehensive genomic and immune profile, helping clinicians select the right therapy the first time.


Why This Matters
Multiple myeloma is one of the most complex blood cancers to diagnose and treat. Current diagnostic methods, such as bone marrow biopsies and FISH (fluorescence in situ hybridization), are invasive and often fail to capture the full genomic picture of the disease. Despite more than 20 approved treatments, clinicians struggle to identify which therapy will work best for each individual, leading to toxic side effects and frequent relapses.
The new test changes that equation. Using whole-genome sequencing of circulating tumor cells and single-cell immune profiling, it identifies key genomic alterations—including mutations in BCMA and GPRC5D that predict whether patients will respond to advanced therapies such as CAR T or bispecific antibodies. It also meets International Myeloma Society and IMWG standards for identifying high-risk disease. Unlike FISH, which misses many critical variants, this approach provides a comprehensive genomic and immune map from either blood or bone marrow samples.
From Discovery to Clinical Impact
This technology grew out of Ghobrial’s global effort to track precursor conditions like MGUS and smoldering myeloma.These insights revealed that disease progression begins years before symptoms appear. These discoveries laid the foundation for a non-invasive test that could diagnose active myeloma and identify high-risk precursor states, enabling earlier intervention and potentially preventing full-blown cancer. Importantly, the findings also demonstrated how integrating genomic data with immune signatures can predict which patients are likely to benefit from immunotherapies. This dual approach genomics plus immune profiling—sets the technology apart from traditional diagnostics and positions it as a future standard for precision oncology.
Expanding the Horizon
Licensed to Predicta Biosciences in 2024, the technology is now being deployed in early clinical studies. Predicta is building an AI-powered database to reveal patterns of disease progression and uncover new therapeutic targets. Next steps include launching minimal residual disease (MRD) monitoring and ImmunoPredicta, a platform that characterizes the immune system to further personalize care. Predicta also aims to expand beyond multiple myeloma to other blood cancers and autoimmune diseases.
Bringing Innovation to Patients
For patients, this milestone means fewer invasive procedures and a better chance of receiving the right therapy the first time. For clinicians, it means a powerful new tool to guide precision treatment decisions.
“This is what innovation is all about—taking cutting-edge science and making it available to patients,” says Ghobrial. “Our goal is to improve the patient experience and outcomes by replacing invasive procedures with a simple test that gives us more information than ever before.”
Team Members




Irene Ghobrial, MD
Senior Vice President for Experimental Medicine, Dana-Farber Professor,
Kenneth Anderson, MD, PhD
Program Director, Jerome Lipper
Multiple Myeloma Center, DanaFarber, Professor,
Gad Getz, PhD
Director, Cancer Genome
Computational Analysis Group, Broad Institute, Professor, Harvard Medical School
Romanos SklavenitisPistofidis, MD, PhD
Instructor of Medicine, Dana-Farber
Translational Pipeline
Beyond funding several innovative technologies emerging from Dana-Farber research labs, the Dana-Farber Accelerator contributes to the business development and marketing efforts for the entire Dana-Farber intellectual property portfolio. Working closely with the Institute's faculty, the Belfer Office for Dana-Farber Innovations and Office of General Council's IP team, the Accelerator team helps develop strategy for dozens of technology programs in multiple modalities. The examples below are just a fraction of the innovations ready for partnering and commercialization. We invite collaborators to join us in transforming the future of cancer treatment and diagnosis.
A New Platform Expands Cancer Drug Discovery
A new chemical biology platform being developed by Lyn Jones, PhD, Faculty Member of the Chemical Biology Program and the Department of Pediatric Oncology at Dana-Farber, is expanding the boundaries of what’s considered “druggable” in cancer. Many cancer-driving proteins are difficult to target with conventional therapies because small molecule ligands that bind to these proteins often lack the potency and selectivity needed for effective treatment. In other words, a protein may be ligandable able to bind a molecule but not truly druggable, meaning the interaction isn’t strong or specific enough to produce a therapeutic effect.
Jones’s lab is overcoming this by designing covalent modulators specialized molecules that form strong, lasting bonds with proteins. Unlike most approaches that focus on cysteine (a reactive amino acid but rarely available for targeting), this strategy addresses previously inaccessible sites. The team combines novel protein-labeling chemistry and structure-guided drug design with chemoproteomics, which maps how reactive chemical probes bind proteins across the cell. These tools also help identify cryptic and allosteric sites regions that influence protein function in subtle ways, such as rewiring cancer cell signaling.
What makes this platform transformative is its scale and precision. By systematically profiling protein reactivity, Jones’s group has uncovered more than 600 previously undruggable sites, creating opportunities to target proteins long considered beyond reach. This work is fueling collaborations across Dana-Farber and industry partners to accelerate drug discovery, paving the way for a new class of therapies for cancers that resist conventional drugs.



A Targeted Approach to Autoimmune Disease
Mohammad Rashidian, PhD, a faculty member of Dana-Farber’s Department of Cancer Immunology and Virology and Associate Professor of Radiology at Harvard Medical School, is developing a therapeutic platform designed to selectively eliminate B cells that produce disease-causing antibodies in autoimmune conditions. These antibodies mistakenly attack healthy tissues, driving chronic inflammation and organ damage. Current treatments often rely on broad immunosuppression, which can leave patients vulnerable to infections and other complications.
Rashidian’s approach is different. It uses engineered biologics that bind to both the autoantibodies and the B cell receptors responsible for their production, creating a molecular “bridge” that triggers targeted removal of the pathogenic B cells. By focusing only on the cells causing disease, the strategy aims to preserve healthy immune function a major advantage over conventional therapies.
The lead candidate is being developed for Pemphigus Vulgaris, a painful and potentially life-threatening condition that affects mucosal tissues and skin. In preclinical studies, the therapy has shown promising results in depleting pathogenic B cells without inducing broad immunosuppression. Because the platform is modular, it can be adapted to other B cell–mediated autoimmune diseases with well-defined targets, including Myasthenia gravis and Graves’ disease.
If successful, this approach could represent a new class of precision immunotherapies—addressing the root cause of disease rather than broadly suppressing immunity. Rashidian’s work underscores the potential of targeted biologics to improve outcomes and quality of life for patients living with autoimmune disorders.
Tracking Hodgkin’s Lymphoma Through Blood
Margaret Shipp, MD, Douglas S. Miller Chair in Lymphoma, Chief of the Division of Hematologic Neoplasia in the Department of Medical Oncology at Dana-Farber, and Professor at Harvard Medical School, is advancing a blood-based diagnostic approach for Hodgkin’s lymphoma using circulating tumor DNA (ctDNA) fragments of genetic material shed by cancer cells into the bloodstream. This innovative method enables clinicians to detect and monitor disease activity without invasive biopsies, offering a less burdensome and more dynamic alternative to traditional imaging.
By analyzing ctDNA, Shipp’s team can identify genetic mutations associated with Hodgkin’s lymphoma, track treatment response in real time, and potentially detect relapse earlier than scans alone. Unlike imaging, which may take weeks to reveal changes, ctDNA provides a rapid and sensitive measure of tumor burden. This is especially valuable in cases where tumors are difficult to access or slow to show progression on imaging.
Translational Pipeline
Beyond early detection, the approach could transform risk stratification and guide personalized treatment decisions. For example, patients who respond quickly at the molecular level might avoid unnecessary chemotherapy cycles and their associated toxicities, while those with persistent ctDNA could receive timely intervention. Shipp’s work is part of a broader effort to integrate liquid biopsy technologies into lymphoma care, aiming to improve outcomes and quality of life for patients. If validated in larger studies, this strategy could become an important tool for monitoring Hodgkin’s lymphoma alongside existing clinical practices.
Editing Cancer’s Code to Reveal New Drug Targets
Matthew Oser, MD, PhD,Associate Professor of Medicine at Dana-Farber, is using CRISPR base editing a precise gene-editing tool to uncover weak spots in small cell lung cancer (SCLC), an aggressive cancer with few targeted treatments. Unlike traditional CRISPR, which works by cutting DNA, base editing makes tiny changes without breaking the strands. It works by changing a single base or “letter” in the DNA code, allowing researchers to study how specific changes affect cancer cell survival with greater accuracy.
Oser’s team is focusing on two key proteins, ASCL1 and POU2F3, which act like “on switches” for different subtypes of SCLC and are essential for tumor growth. These proteins have long been considered “undruggable” because they don’t have obvious places for drugs to latch onto. To tackle this, the team combines base editing with special reporter systems tools that show how cells respond when these proteins are altered. By making precise edits and watching what happens, they can pinpoint critical regions of the proteins and identify potential drug-binding sites.
This work could open the door to the first therapies that directly target these drivers of SCLC. It’s part of a broader effort to bring precision medicine to a cancer type that urgently needs better options, moving beyond one-size-fits-all treatments toward strategies tailored to the biology of each tumor. The insights gained may also help researchers understand why certain subtypes respond differently to existing therapies, guiding future drug development across the field.


Universal Platform for Localized Cancer Immunotherapy
Solid tumors have resisted the transformative gains of CAR T cell therapy seen in blood cancers, largely because T cells struggle to reach, persist, and function inside the hostile tumor microenvironment. The Universal Platform for Localized Solid Tumor Cancer Immunotherapy, developed by Thanh Barbie, MD, and colleagues at Dana-Farber, tackles this problem with a simple yet radical idea: repurpose an FDA-approved PEG hydrogel biopsy marker—already used in breast cancer imaging as a programmable, tissue-anchored depot that orchestrates immunotherapy exactly where it’s needed. This “grain of rice” marker, radiographically visible and stable in human tissue for up to 11 months, absorbs and releases payloads without triggering foreign body reactions, enabling sequential, localized delivery of agents that prime and empower anti-tumor immunity.

The platform’s innovation is twofold. First, it converts the tumor microenvironment from immunosuppressive to immunostimulatory through sustained, local release of STING agonists, overcoming the rapid clearance and systemic toxicities that limited prior trials. Second, it delivers CAR T cells directly into the tumor via the hydrogel, bypassing trafficking barriers and reducing off-target risks. In triple-negative breast cancer mouse models, this approach drove robust
intratumoral CAR T expansion, inhibited tumor growth, prevented relapse when preconditioned with a STING agonist (ADU-S100), and strikingly controlled distant metastases and blocked new lesions, suggesting the induction of systemic immune memory with minimal systemic toxicity.
Because the platform is device-agnostic and payload-agnostic, it is truly universal: a plug-and-play strategy for CAR T, NK cells, checkpoint inhibitors, and small molecules across diverse solid tumors. It fits seamlessly into existing clinical workflows via minimally invasive, image-guided placement, and multiple markers can be deployed to treat oligometastatic disease. By merging a proven, FDAapproved biomaterial with cutting-edge immunotherapies, this platform reframes solid tumor treatment from systemic exposure to precision, localized immune engineering opening a clear path to first-in-human studies and a new paradigm in cancer care.

TIGER Platform: A Trojan Horse Approach to Turn Cancer Cells against Themselves
Solid tumors remain stubbornly resistant to today’s immunotherapies because dense tumor masses and an immunosuppressive microenvironment block immune cell access and function. Traditional targeted therapies often chase shifting mutations and escape pathways, but tumors evolve faster than our ability to target them. The TIGER (Tumor Immunotherapy by Gene Circuit Engineered Response) platform from Dana-Farber introduces a Trojan Horse strategy, developed by Ming-Ru Wu, MD, PhD, of the Department of Cancer Immunology & Virology, that turns cancer cells into factories for their own destruction. Rather than forcing external immune cells to fight their way in or adapt to cancer’s changes, TIGER programs malignant cells to broadcast danger signals and present targets that recruit and activate the patient’s immunity precisely within the tumor. This paradigm recasts solid tumors from immune deserts into self-marked, self-amplifying signals for clearance, while sparing healthy tissue.
Translational Pipeline
Solid tumors remain stubbornly resistant to today’s immunotherapies because dense tumor masses and an immunosuppressive microenvironment block immune cell access and function. Traditional targeted therapies often chase shifting mutations and escape pathways, but tumors evolve faster than our ability to target them.
The TIGER (Tumor Immunotherapy by Gene Circuit Engineered Response) platform from Dana-Farber introduces a Trojan Horse strategy, developed by Ming-Ru Wu, MD, PhD, of the Department of Cancer Immunology & Virology, that turns cancer cells into factories for their own destruction. Rather than forcing external immune cells to fight their way in or adapt to cancer’s changes, TIGER programs malignant cells to broadcast danger signals and present targets that recruit and activate the patient’s immunity precisely within the tumor. This paradigm recasts solid tumors from immune deserts into self-marked, selfamplifying signals for clearance, while sparing healthy tissue.
At its core, TIGER uses synthetic gene circuits encoded in DNA and gated by cancer-specific transcription factor signatures. Highly selective synthetic promoters act as sensors, remaining silent in normal tissue and switching on only inside malignant cells.After systemic delivery via viral vectors or plasmids, the circuits trigger localized production of immune-stimulatory payloads chemokines, cytokines, universal antigens, and even intratumoral checkpoint inhibitors—creating a potent, tumor-confined combination therapy from within. Boolean logic enables multi-input discrimination and tunable specificity, while an AIenabled design engine optimizes sensor performance across tumor types. The result is programmable, modular precision that targets hard-to-drug signatures conventional biologics cannot reach.
In preclinical ovarian cancer and melanoma models, TIGER provoked robust immune infiltration, tumor regression, and durable protection even when few tumor cells received the circuit highlighting powerful bystander amplification. Because the therapy is genetically encodable, its payloads and logic can be reconfigured to match diverse indications and synergize with FDA-approved immunotherapies, positioning TIGER as both a standalone and combination backbone. Beyond oncology, the same sensing framework could be adapted to autoimmune and other diseases defined by pathological transcriptional programs.


ImmunoBac:
Bacteria-Based Microbots as a Novel Platform for Immunotherapy
The story of ImmunoBac begins with a stubborn problem: most advanced solid tumors are “cold.” They hide in hypoxic pockets, keep immune cells out, and drain the few that get in. Checkpoint inhibitors and cell therapies can work in some cancers, but often stall at the tumor’s gates. Patients need options that deliver strong, localized immunity inside the tumor’s most resistant spaces.
A clue came from an unexpected source: certain E. coli naturally colonize hypoxic tissues the same conditions inside solid tumors.At Dana-Farber Cancer Institute, a team led by Rizwan Romee, MD, engineered a safe, gut-derived strain—E. coli K12 DH5α—to carry immune-activating cytokines on its surface. Instead of releasing drugs that diffuse away, these “microbots” display signals on their outer membrane, creating a persistent “on-switch” for immunity where the tumor lives.
In preclinical studies, E. coli displaying a decoy-resistant version of IL18 (DR18) outperformed checkpoint inhibitors alone, achieving 50–60% cure rates in mice with solid tumors. When combined with checkpoint therapy, the bacteria amplified the effect. Treated tumors filled with activated CD8+ T cells and natural killer (NK) cells, converting cold, suppressive masses into immune-active sites. In a mesothelioma model that resists NK therapy, the bacteria also boosted mesothelin-targeting CAR NK cells acting like a “tumor GPS” to improve trafficking and activation.
Safety was central to the design. The chosen strain is non-pathogenic, lacks known toxins, and is sensitive to common antibiotics, providing an immediate off switch.Across multiple tumor models, including mesothelioma, the team observed strong anti-tumor activity without overwhelming infection or dangerous cytokine surges.
By enlisting tumor-homing bacteria as localized immune activators, ImmunoBac addresses trafficking and exhaustion barriers that limit current therapies and pairs naturally with checkpoint inhibitors and adoptive cells. With robust preclinical validation and a clear path to early-phase trials, this approach represents a novel strategy for tumors that have resisted immunotherapy.
Translational Pipeline
The X Factor in Kidney Cancer: Targeting TFE3 Fusions to Transform tRCC
Translocation renal cell carcinoma (tRCC) is an aggressive kidney cancer with no approved standard of care and a median survival of just 2–3 years in the metastatic setting. It accounts for 5% of adult and 50% of pediatric RCC.A defining feature and core vulnerability of tRCC is oncogenic fusions involving the transcription factor TFE3. Work from Dana-Farber Cancer Institute, led by Srinivas R. Viswanathan, MD, PhD, has shown that these TFE3 fusions are exquisite dependencies and revealed why tRCC is more common in females: TFE3 can fuse from either the active or the inactive X chromosome, creating a 2:1 female-to-male fusion ratio and broad epigenetic consequences when the inactive X is involved. Together, these insights open the door to truly sex-informed precision oncology.
Targeting TFE3 fusions represents a first-in-class opportunity to drug a transcription factor driver with a potentially wide therapeutic window. TFE3 deletions show no discernible phenotypes in mice or non-tRCC cells, suggesting tumorselective sensitivity. The unique fusion structures also enable straightforward, scalable diagnostics for patient
other rare cancers harboring TFE3 fusions, expanding the impact beyond kidney cancer. selection across tRCC and
To capitalize on this biology, the Dana-Farber team has completed target validation and built a purposedesigned discovery engine. They developed and benchmarked a high-throughput, cell-based DNA displacement assay that reports on TFE3-fusion occupancy at DNA addressing a class historically considered “undruggable.”A 25,000-compound pilot screen has already yielded candidate hits now undergoing validation. Next, the program will launch a biochemical screen for direct TFE3-fusion binders.


Hidden in Plain Sight: Decoding Cryptic Neoantigens for Off-theShelf Cancer Immunity
Most solid tumors, especially pancreatic ductal adenocarcinoma (PDAC), lack clear, shared targets for the immune system, which is why many immunotherapies struggle. William Freed-Pastor, MD, PhD, at DanaFarber Cancer Institute, built a newimmunotherapy platform for the identification and targeting of cryptic neoantigens to solve that problem. The idea is simple and powerful: cancers often make “hidden” protein fragments from parts of the genome that aren’t supposed to code for proteins. These cryptic neoantigens can be unique to cancer cells and crucially shared across many patients, opening the door to off-theshelf vaccines and T cell therapies instead of costly, bespoke treatments.
This platform is end-to-end. It discovers these hidden signals in real patient tumors, confirms they’re truly cancer-specific, and then turns them into treatments. As a proof-of-concept, the team focused on PDAC, one of the hardest cancers to treat. They used lab-grown mini-tumors from patients to map what protein fragments are actually displayed by cancer cells. They found a large number of previously overlooked, cancer-made fragments; about 30% appeared only in cancer and not in healthy tissues, and many were shared across patients exactly what’s needed for scalable therapies. When the team trained T cells to recognize these targets, the cells powerfully attacked and killed the pancreatic cancer mini-tumors in the lab and in animal models.
The benefits are clear: this platform expands the universe of targetable cancer signals, prioritizes those that are shared and safe, and delivers a practical route to off-the-shelf therapies. It can generate building blocks for vaccines, T cell engagers, and T cell receptor (TCR) therapies, with patient selection guided by straightforward tests.
2025 Accelerator Scientific Symposium



Accelerator Scientific Symposium
In 2025, the Dana-Farber Accelerator Scientific Symposium highlighted ground-breaking translational science developed at the Institute and brought together thought leaders in the scientific and business communities dedicated to the advancement of cancer care. Speakers discussed novel science in the fields of immunotherapy, RNA therapeutics, small molecule degraders, and more. The Dana-Farber Accelerator team hosts the event every spring to showcase the amazing science of that year.
2025 Symposium Speakers




Moderator: Lee Greenberger, PhD
President, LMG Bio1 Consulting, Former Chief Science Officer, The Leukemia & Lymphoma Society
Irene Ghobrial, MD; Unlocking Precision Therapeutics
Professor, Harvard Medical School, Dana-Farber, Senior Vice President for Experimental Medicine Dana-Farber
Rizwan Romee, MD; Harnessing Innate NK Cell Memory for Enhanced Immunotherapy in Acute Myeloid Leukemia
Associate Professor, Harvard Medical School,Dana-Faber, Director, Haploidentical Donor Transplantation Program




Keynote: Eric Fischer, PhD
Professor, Harvard Medical School, Dana-Farber, Joseph Deitch Family Chair, Dana-Farber, Director, Chemical Biology Program
Ellis Reinherz, MD; Leveraging TCR Mechanobiology and Ligand Recognition Ground Truth for Tumor Targeting by Cytotoxic Lymphocytes
Professor, Harvard Medical School, Dana-Farber, Chief, Laboratory of Immunobiology

Roger Belizaire, MD; A Novel Targeted Therapy for VEXAS syndrome
Assistant Professor, Harvard Medical School, Dana-Farber, Associate Medical Director, Division of Transfusion Medicine and Pathology BWH

Megan Insco, MD, PhD; Modulation of Nuclear RNA Surveillance as a Novel Therapeutic Strategy for the Treatment of Cancer
Instructor, Harvard Medical School, Dana-Farber
Loren Walensky, MD, PhD; Triple Action PROTACs for Cancer Therapy
Professor, Harvard Medical School, Dana-Farber, Director, Harvard/MIT MDPhD Program
The Accelerator
Driving Therapeutic Discovery
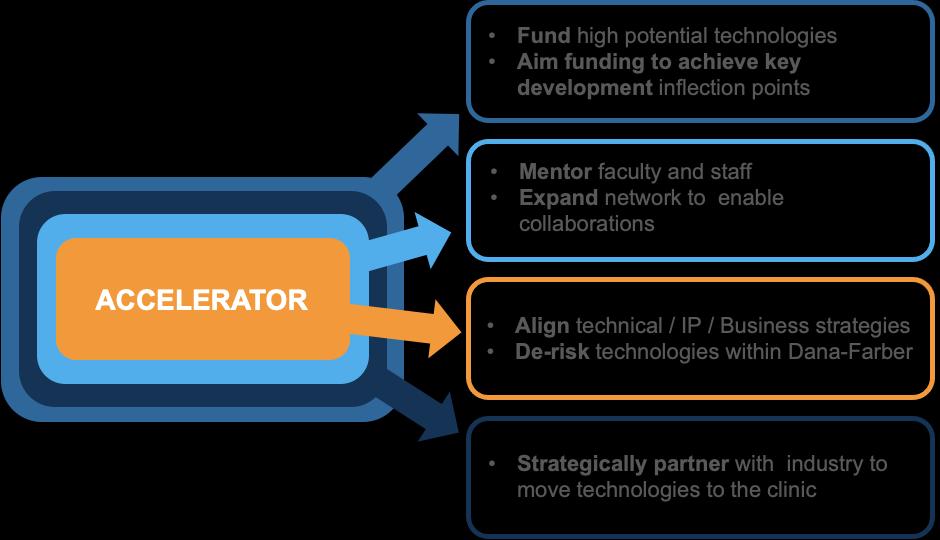
The Dana-Farber Accelerator is focused on the translation of scientific discovery from institute investigators into first-in-class technologies with the potential to create significant impact for patients worldwide. We identify investigators taking on high risk, novel science, who, with early-stage funding and mentorship, can address key developmental questions to transition from idea to potential novel technology.Accelerator funding is focused on critical inflection points in technology development that allow investigators to pressure test assumptions and answer critical questions about the technology's potential to translate into a therapeutic or diagnostic for patients with cancer or related diseases. We align technical, business, and intellectual property strategies to enable subsequent collaboration with the best partner who will further the next stage of development towards commercialization.


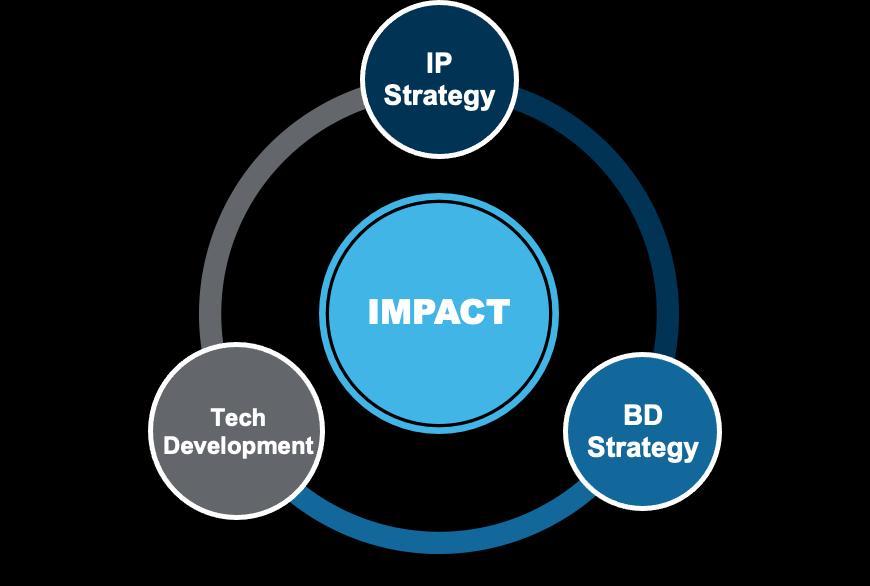
An Integrated Strategy
Successful development of nascent technologies requires a multidisciplinary approach that takes into account the unique opportunity presented by the technology, integrating in-depth understanding of the science, the competitive landscape into which the technology will be introduced, and an intellectual property strategy that strengthens the business case, all focused on maximizing the potential impact of the technology. To be able to do such work, the Accelerator draws upon the diverse expertise of the DanaFarber community.

Importantly, the Accelerator team operates out of the Belfer Office of Dana-Farber Innovation (BODFI), a group of business development, licensing, and contracts professionals whose collective mission is to expedite the translation of discoveries made in the labs at Dana-Farber. This requires efficient collaboration between the primary investigator, the Accelerator team, the larger BODFI Office, and the Dana-Farber Office of General Council to develop and implement an effective strategy for technology development.

The Team
Operations
• Christiana Iyasere, MD, MBA, Senior Director, Business Development
• Harry Rogoff, PhD, Director, Business Development (Therapeutics)
• Shankar Parajuli, PhD, Business Development Director – Startups
• Linda van Weele, PhD, Business Development Specialist
• Javier Vinals Camallonga, PhD, Business Development Data Analyst
• Liz Robinson, Accelerator Pipeline Marketing Manager
Business Development Council
• Steven Caltrider, Esq., Vice President and Chief Intellectual Property Officer, Dana-Farber
• Emy Chen, PhD, Vice President of Innovation, Dana-Farber
• Benjamin Ebert, MD, PhD, President and Chief Executive Officer Dana-Farber
• William Hahn, MD, PhD, Executive Vice President and Chief Operating and Transformation Officer, Dana-Farber
• Kevin Haigis, PhD, Chief Scientific Officer, Dana-Farber
• Kostas Kaloulis, PhD, Chief Executive Officer and Board Member, EpiCure Biotechnologies
• Hillary Mankin-Kufe, Esq, Senior Counsel, Dana-Farber
• Christopher Mirabelli, Chairman, Leap Therapeutics
• Steven Neier, PhD, Principal at Binney Street Capital
• Martin Seidel, PhD, Chief Executive Officer, IFM Therapeutics
• Alice Shaw MD, PhD, Chief, Strategic Partnerships, Dana-Farber Cancer Institute
• Mary Tolikas, Senior Vice President and Chief Innovation Officer, Dana-Farber
Scientific Review Committee
• David Barbie, MD, Professor, Harvard Medical School, Dana-Farber
• Myles Brown, MD, Professor, Harvard Medical School, Dana-Farber
• Sara Buhrlage, PhD, Associate Professor, Harvard Medical School, Dana-Farber
• Kathleen Burns, MD, PhD, Professor, Harvard Medical School, Dana-Farber
• Edward Chouchani, PhD, Professor, Harvard Medical School, Dana-Farber
• Michael Eck MD, PhD, Professor, Harvard Medical School, Dana-Farber
• Eric Fischer PhD, Professor, Harvard Medical School, Dana-Farber
• Irene Ghobrial MD, Professor, Harvard Medical School, Dana-Farber
• Ruud Hulspas PhD, Director Process Development, Dana-Farber
• Jeffrey Meyerhardt, MD, MPH, Professor, Harvard Medical School, Dana-Farber
• Jerome Ritz, MD, Professor, Harvard Medical School, Dana-Farber
• David Scott, PhD, Principal Scientist, Director of Medicinal Chemistry Core
• Eric Smith, MD, PhD, Assistant Professor, Harvard Medical School, Dana-Farber


How to Get Involved
INTERACT
Want to learn more about how to work with the Accelerator?
Accelerator support is open to all Dana-Farber faculty engaged in translational science who are looking for mentorship and support towards pursuing a commercial opportunity. We hold requests for proposals in the fall and spring, focused independently on therapeutics and diagnostics. Individuals may engage with the Accelerator team at any time; we are always eager and interested to hear how we can help enable the important work of Dana-Farber investigators.
Email: DFCI_Accelerator@Dana-Farber.harvard.edu
PARTNER
Interested in the science behind some of these projects?
Accelerator funding is one small step in part of a long pathway toward the creation of successful technologies and impact; our goal is to demonstrate the potential of the science as a solution to elicit follow-on funding for the next stage of development. Please reach out if you are interested in hearing more about the science or want to learn about other opportunities in Dana-Farber’s technology portfolio. Here we have highlighted only a small fraction of the exciting science that is part of the Accelerator and the larger technology landscape of Dana-Farber.
Email: DFCI_Accelerator@Dana-Farber.harvard.edu
DONATE
Looking to support the Accelerator with the potential to change cancer care?
Generous philanthropy created and continuous to support the DFCI Accelerator and the projects we fund. We are interested in high-risk, high-reward translational science that can change the face of cancer therapy. Please contact our team.
Email: JimmyFundContactUs@dfci.harvard.edu
Thank you to our donors for enabling our mission and making the work of the Dana-Farber Accelerator program possible.


