
Four monthly scientific journal of the Greek Paediatric Society



Υποβολή
e-mail: grammateia@e-child.gr
Οδηγίες
http://e-child.gr/publications/ instructions-to-authors
Iδιοκτήτης
e-mail: grammateia@e-child.gr
178
184
186
188


Four monthly scientific journal of the Greek Paediatric Society



Υποβολή
e-mail: grammateia@e-child.gr
Οδηγίες
http://e-child.gr/publications/ instructions-to-authors
Iδιοκτήτης
e-mail: grammateia@e-child.gr
178
184
186
188
Volume 82 | Number 2 | May - June - July - August 2019
Four monthly publication of the Greek Paediatric Society
124
EDITORIAL
Stelios Antoniadis
126
AWARDED ARTICLE
Study of risk factors for dyslipidemia in childhood and young adulthood
Kyriaki Papadopoulou-Legbelou, Ioannis Doundoulakis, Anastasia Papadopoulou, Ioannis Barbalias, Maria Emporiadou-Petikopoulou
136
RESEARCH STUDIES
Evaluation of Töllner test and other biomedical markers in early detection of neonatal sepsis
Chatziioannidis I., Lithoxopoulou M., Papacharalambous E., Babatseva E., Kokoviadou K., Kontou A., Tsakalidis C., Karagianni P., Mitsiakos G.
148
Positive-expiratory pressure (PEP) device compared with Flutter device in children with cystic fibrosis
Ntinou Stefania-Rafailia, Hristara-Papadopoulou Alexandra, Hatziagorou Elpida, Tsanakas Ioannis, Fitili Dimitra
158
CASE REPORTS
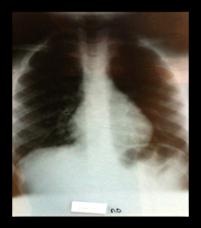
Pulmonary arteriovenous malformations and hereditary hemorrhagic telangiectasia (Rendu- Osler- Weber syndrome)
Gketsi Vasiliki, Kyrochristou Electra, Vlachou Georgia, Tsoutsinos Alexandros, Apostolopoulou Soteria
President A. Constantopoulos
Editorial board
Editor- in- Chief
S. Antoniadis
Members
S. Andronikou
E. Galanakis
A. Evangeliou
L. Thomaidou
M. Kanariou
A. Kapogiannis
S. Kitsiou-Tzeli
E. Mantadakis
P. Panagiotopoulou-Gartagani
A. Papadopoulou
V. Papaevagelou
A. Papathanassiou
A. Siamopoulou-Mavridou
A. Syrigou-Papavasiliou
Manuscript submission
e-mail: grammateia@e-child.gr
Instructions to authors: http://e-child.gr/publications/ instructions-to-authors
Owner Greek Paediatric Society 15, Mpakopoulou st. GR - 15451, Ν. Psychiko Tel.: +302107771140
e-mail: grammateia@e-child.gr
Annual subscription All foreign countries: US$50
172
Transient neonatal myasthenia Gravis due to a mother with asymptomatic myasthenia gravis: a case study A. Drougia, F.Balomenou, M. Katsalouli, E. Domouzoglou, V. Giapros
178
Glomerulonephritis as a complication of H1N1 influenza virus infection
Garyfallia Syridou, Ioannis Drikos, Eleni Papa, Rougas Panagiotis, Dimitrios Kyriazopoulos, Antigoni Pegkou, Artemis Vintila, Eleni Antonopoulou
184 BETWEEN COLLEAGUES
Stelios Antoniadis
186 NEW PUBLICATIONS
Stelios Antoniadis
188 BOOK PRESENTATION
Stelios Antoniadis
190
INSTRUCTIONS TO AUTHORS
(21/35<250mg/dl, 14/35>250mg/dl), ενώ 33/35 (94,3%)
20/35 (57,1%)
6/35 (17,1%)
e-mail: kelipap@gmail.com, kpapadopoulou@auth.gr
Correspondence
Kyriaki PapadopoulouLegbelou
Τ. +30 2310991463, +30 6944421060
e-mail: kelipap@gmail.com, kpapadopoulou@auth.gr
Kyriaki Papadopoulou-Legbelou, Ioannis Doundoulakis, Anastasia Papadopoulou, Ioannis Barbalias, Maria Emporiadou-Petikopoulou
Abstract
Backround: Prompt diagnosis and management of dyslipidemia in childhood is imperative, as it is one of the main risk factors for premature atherosclerosis. Our aim was to record the risk factors (hereditary and acquired) responsible for dyslipidemia in childhood. Methods: Thirty-five children/adolescents (17 boys) aged 6-22years (mean age 12.43±3.9years) were studied, being followed-up in the outpatient clinic of dyslipidemia. A detailed medical history was given by all patients/parents with their consent. Results: All participants had increased levels of total cholesterol (21/35<250mg/dl, 14/35>250mg/dl), while 33/35 (94.3%) also had increased non-HDLcholesterol levels. Furthermore, high levels of lipoprotein (a) and triglycerides were observed in 20/35 (57.1%) and 8/35 (22.8%) respectively, as well as low levels of HDL-Cholesterol in 6/35 (17.1%). Most children (32/35) had normal body weight (BMI<85th percentile), one of the remaining was overweight and two were obese. As for food habits, 20/35 children followed a Mediterranean diet, 10/35 overconsumed red meat and 5/35 consumed fast food frequently. The majority of children (21/35) had low-intensity exercise (<5hours/week), while the remaining 40% had medium/high-intensity exercise (>5hours/week). Family history revealed that at least one parent was obese (45% of participants), or had hypercholesterolemia (88.8%), hypertension (17.1%), coronary artery disease (28.6%), or was a smoker (68.6%). Conclusions: Non-HDL-cholesterol is a highly-sensitive marker for diagnosing hypercholesterolemia in childhood. Dyslipidemia cannot be excluded even in the absence of obesity, unhealthy diet or limited exercise-especially if there is family history of dyslipidemia, premature coronary artery disease, smoking, or obesity in at least one parent.
Key words: Dyslipidemia, hypercholesterolemia, risk factors, children and adolescents
Kyriaki PapadopoulouLegbelou
Ioannis Doundoulakis
Anastasia Papadopoulou
Ioannis Barbalias
Maria Emporiadou-Petikopoulou
4th Department of Pediatrics, School of Medicine, Faculty of Health Sciences, Aristotle University of Thessaloniki, "Papageorgiou"
General Hospital, Thessaloniki, Greece
(m) και ο
δείκτης μάζας σώματος (ΒΜΙ) (kg/m2). Παιδιά
(TC), τριγλυκερίδια (TG), HDL-χοληστερόλη (HDL-C), LDL- χοληστερόλη ( LDL-C), απολιποπρωτεϊνη Α-I (ApoA-I), απολιποπρωτεϊνη
Juonala et al,
Bogalusa Heart Study, Muscatine Study, Childhood Determinants of Adult Health study
Cardiovascular Risk in Young Finns Study) (17, 32-34),
(38).
1. McCrindle BW, Urbina EM, Dennison BA, Jacobson MS, Steinberger J, Rocchini AP et al. Drug Therapy of High-Risk Lipid Abnormalities in Children and Adolescents. A Scientific Statement From the American Heart Association Atherosclerosis, Hypertension, and Obesity in Youth Committee, Council of Cardiovascular Disease in the Young, With the Council on Cardiovascular Nursing. Circulation 2007;115:1948-1967.
2. Kavey RE, Allada V, Daniels SR, Hayman LL, Mc-Crindle BW, Newburger JW, et al. Cardiovascular risk reduction in high-risk pediatric patients: a scientific statement. From the American Heart Association Expert Panel on Population and Prevention Science (Wash DC); the Councils on Cardiovascular Disease in the Young, Epidemiology and Prevention, Nutrition, Physical Activity and Metabolism, High Blood Pressure Research, Cardiovascular Nursing, and the Kidney in Heart Disease; and the Interdisciplinary Working Group on Quality of Care and Outcomes Research: endorsed by the American Academy of Pediatrics. Circulation 2006;114:2710 –38.
3. Narverud I, Retterstøl K, Iversen PO, Halvorsen B, Ueland T, Ulven SM, et al. Markers of atherosclerotic development in children with familial hypercholesterolemia: a literature review. Atherosclerosis 2014; 235:299–309.
4. Kusters DM, Wiegman A, Kastelein JJ, Hutten BA. Carotid intima-media thickness in children with familial hypercholesterolemia. Circ Res 2014;114:307–310.
5. American Academy of Pediatrics: National Cholesterol Education Program: report of the Expert Panel on Blood Cholesterol Levels in Children and Adolescents. Pediatrics 1992;89(3 Pt 2):525-84.
6. Haney EM, Huffman LH, Bougatsos C, Freeman M, Steiner RD, Nelson HD. Screening and treatment for lipid disorders in children and adolescents: systematic evidence review for the US Preventive Services Task Force. Pediatrics 2007;120: e189–214.
7. Daniels SR, Greer FR. Lipid screening and cardiovascular health in childhood. Pediatrics 2008;122:198–208.
8. Ritchie SK, Murphy EC, Ice C, et al. Universal versus targeted blood cholesterol screening among youth. Pediatrics. 2010;126:260–265.
9. Nordestgaard BG, Chapman MJ, Humphries SE, Ginsberg HN, Masana L, Descamps OS, et al. Familial hypercholesterolaemia is underdiagnosed and undertreated in the general population: guidance for clinicians to prevent coronary heart disease: consensus statement of the European Atherosclerosis Society. Eur Heart J 2013;34:3478-90a.
10. Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents. Expert panel on integrated guidelines for cardiovascular health and risk reduction in children and adolescents: summary report. Pediatrics 2011;128 (5 Suppl):S213–S256.
11. Peterson AL, McBride PE. A review of guidelines for dyslipidemia in children and adolescents. WMJ 2012;111:274-81.
12. McNeal CJ, Dajani T, Wilson D, Cassidy-Bushrow AE, Dickerson JB, Ory M. Hypercholesterolemia in Youth: Opportunities and Obstacles to Prevent Premature Atherosclerotic Cardiovascular Disease. Curr Atheroscler Rep 2010;12:20–28.
13. Klag MJ, Ford DE, Mead LA, He J, Whelton PK, Liang K-Y, Levine DM. Serum cholesterol in young men and subsequent cardiovascular disease. N Engl J Med 1993,328:313-8.
14. Stamler J, Daviglus ML, Garside DB, Dyer AR, Greenland P, Neaton JD. Relationship of baseline serum cholesterol levels in 3 large cohorts of younger men to long-term coronary, cardiovascular, and all-cause mortality and to longevity. JAMA 2000;284:311-8.
15. Law MR. Lowering heart disease risk with cholesterol reduction: evidence from observational studies and clinical trials. Eur Heart J 1999; (Suppl):S3-S8.
16. Thompson GR, Catapano A, Saheb S, Atassi-Dumont M, Barbir M, Eriksson M, et al. Severe hypercholesterolaemia: therapeutic goals and eligibility criteria for LDL apheresis in Europe. Curr Opin Lipidol 2010;21:492-8.
17. Berenson GS, Srinivasan SR, Bao W, Newman WP III, Tracy RE, Wattigney WA. Association between multiple cardiovascular risk factors and atherosclerosis in children and young adults. The Bogalusa Heart Study. N Engl J Med 1998;338:1650-6.
18. Morrison KM, Dyal L, Conner W, Helden E, Newkirk L, Yusuf S, et al. Cardiovascular risk factors and non-invasive assessment of subclinical atherosclerosis in youth. Atherosclerosis 2010;208:501-5.
19. Kamstrup PR, Tybjaerg-Hansen A, Steffensen R, Nordestgaard BG. Genetically elevated lipoprotein(a) and increased risk of myocardial infarction. JAMA 2009;301:2331–2339.
20. Clarke R, Peden JF, Hopewell JC, et al. Genetic variants associated with Lp(a) lipoprotein level and coronary disease. N Engl J Med 2009;361:2518–2528.
21. Emerging Risk Factors Collaboration, Erqou S, Kaptoge S, Perry PL, Di Angelantonio E, Thompson A, White IR et al. Lipoprotein (a) concentration and the risk of coronary heart disease, stroke, and nonvascular mortality. JAMA 2009;302:412–423.
22. Palmeira ÁC, Leal AA, Ramos Nde M, Neto Jde A, Simões MO, Medeiros CC. Lipoprotein (a) and cardiovascular risk factors in children and adolescents. Rev Paul Pediatr 2013;31:531-7.
23. Goldenberg NA, Bernard TJ, Hillhouse J, Armstrong-Wells J, Galinkin J, Knapp-Clevenger R, et al. Elevated lipoprotein (a), small apolipoprotein (a), and the risk of arterial ischemic stroke in North American children. Haematologica 2013;98:802-7.
24. Lapinleimu J, Raitakari OT, Lapinleimu H, Pahkala K, Rönnemaa T, Simell OG, et al. High lipoprotein(a) concentrations are associated with impaired endothelial function in children. J Pediatr 2015;166:947-52.e1-2.
25. Wiegman A, Gidding SS, Watts GF, Chapman MJ, Ginsberg HN, Cuchel M et al. Familial hypercholesterolaemia in children and adolescents: gaining decades of life by optimizing detection and treatment. Eur Heart J 2015;36:2425-37.
26. Civeira F. International Panel on Management of Familial Hypercholesterolemia. Guidelines for the diagnosis and management of heterozygous familial hypercholesterolemia. Atherosclerosis 2004;173:55-68.
27. Freedman DS, Dietz WH, Srinivasan SR, Berenson GS. The relation of overweight to cardiovascular risk factors among children and adolescents: the Bogalusa Heart Study. Pediatrics 1999;103(pt1):1175–1182.
28. Steinberger J, Moran A, Hong CP, Jacobs DR Jr, Sinaiko AR. Adiposity in childhood predicts obesity and insulin resistance in young adulthood. J Pediatr 2001;138:469–473.
29. Sinaiko AR, Jacobs DR Jr, Steinberger J, Moran A, Luepker R, Rocchini AP, et al. Insulin resistance syndrome in childhood: associations of the euglycemic insulin clamp and fasting insulin with fatness and other risk factors. J Pediatr 2001;139:700 –707.
30. Barker DJ: The developmental origins of insulin resistance: Horm Res 2005;64: (3 Suppl):2–7.
31. Norman M: Low birth weight and the developing vascular tree: a systematic review: Acta
Paediatr 2008;97:1165–1172.
32. Davis PH, Dawson JD, Riley WA, Lauer RM. Carotid intimal-medial thickness is related to cardiovascular risk factors measured from childhood through middle age:the Muscatine Study. Circulation 2001;104:2815-9.
33. Magnussen CG, Raitakari OT, Thomson R, et al. Utility of currently recommended pediatric dyslipidemia classifications in predicting dyslipidemia in adulthood: evidence from the Childhood Determinants of Adult Health (CDAH) study, Cardiovascular Risk in Young Finns Study, and Bogalusa Heart Study. Circulation 2008;117:32-42.
34. Raitakari OT, Juonala M, K.h.nen M, et al. Cardiovascular risk factors in childhood and carotid artery intima-media thickness in adulthood: the Cardiovascular Risk in Young Finns Study. JAMA 2003;290:2277-83.
35. Juonala M, Magnussen CG, Berenson GS, Venn A, Burns TL, Sabin MA, et al. Childhood adiposity, adult adiposity, and cardiovascular risk factors. N Engl J Med 2011;365:1876-85.
36. Tolfrey K, Campbell IG, Batterham AM. Exercise training induced alterations in prepubertal children’s lipid-lipoprotein profile. Med Sci Sports Exerc 1998;30:1684 –1692.
37. Katzmarzyk PT, Malina RM, Bouchard C. Physical activity, physical fitness, and coronary heart disease risk factors in youth: the Quebec Family Study. Prev Med 1999;29:555–562.
38. Nanninga S, Lhachimi SK, Bolte G. Impact of public smoking bans on children's exposure to tobacco smoke at home: a systematic review and meta-analysis. BMC Public Helth 2018;18:749. doi: 10.1186/s12889-018-5679-z.
Correspondence
Ι. Chatziioannidis
3B Ag. Triados Str., 57010 Pefka, Thessaloniki, Greece.
Τ. +30 2310 910401, +306977244542
e-mail: drilias@windowslive. com
Chatziioannidis I., Lithoxopoulou M., Papacharalambous E., Babatseva E., Kokoviadou K., Kontou A., Tsakalidis C., Karagianni P., Mitsiakos G.
Abstract
Backround: Early diagnosis of sepsis is an important step to early initiation of therapy.
Aim: Assessment of clinical-laboratory indicators from Töllner test by adding additional indicators in neonates suspected of sepsis in order to develop a predictive risk model.
Materials and Methods: This study prospectively examined the clinical-laboratory findings from 55 newborn infants with 74 possible sepsis episodes in the “early stage” of the disease hospitalized in the NICU. The newborns were divided in group A with positive blood culture (n=22) and group B with negative blood culture (n=52). The presence of positive blood culture was considered as sepsis. Statistical analysis was performed using the SPSS v.17.
Results: From the Töllner score clinical and hematological symptoms, skin coloration, microcirculation, muscular hypotonia, bradycardia, leucocytosis/leukocytopenia, thrombocytopenia and additional markers as temperature increase, hyperglycemia hypotension and CRP were found statistically significant (p<0,05) for group A. Pathological Töllner test and CRP were associated with increased risk for sepsis.
Conclusions: Skin coloration, microcirculation, hypotonia, bradycardia, presence of leucocytosis/leukocytopenia, thrombocytopenia, temperature increase, hyperglycemia hypotension and increased CRP are important clinical-laboratory indicators for early diagnosis of sepsis. Major prognostic indicators for early diagnosis of sepsis were found to be pathological Töllner test and CRP.
Key words: Töllner test, sepsis, neonates, biomedical markers, indicators
Chatziioannidis I.
Lithoxopoulou M.
Papacharalambous E.
Babatseva E.
Tsakalidis C.
Karagianni P.
Mitsiakos G.
B΄ Neonatal Intensive Care
Unit A.U.T., General Hospital Papageorgiou, Thessaloniki, Greece
Kokoviadou K.
Hematology Department,
G.P.N. Papageorgiou, Thessaloniki
Kontou A.
Α' Neonatal Intensive Care
Unit A.U.T., General Hospital Hippokration, Thessaloniki
κυάνωση ή
πτώσης κορεσμού οξυγόνου ≤85%], (γ) το καρδιαγγειακό σύστημα (βραδυκαρδία (σφύξεις <100 παλμοί/λεπτό), ταχυκαρδία, υπόταση) (δ) το πεπτικό σύστημα (δυσανεξία
σίτιση, γαστρικά υπόλοιπα > 30%, μετεωρισμός)
dows (SPSS Inc., Chicago,
λοίμωξης (χοριοαμνιονίτιδα
group B streptococcus (GBS)),
(Candida parapsilosis).
exact t-test
(10/22 έναντι 7/52),
(5/22
3/52),
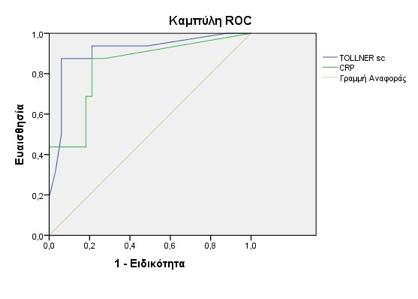
(24,29).
To Töllner
1. Tarnow-Mordi WO. What is the role of neonatal organ dysfunction and illness severity scores in therapeutic studies in sepsis? Pediatr Crit Care Med. 2005;6(3 Suppl):S135-7.
2. Narasimha A, Harendra Kumar ML. Significance of Hematological Scoring System (HSS) in Early Diagnosis of Neonatal Sepsis. Indian J Hematol Blood Transfus. 2011;27(1):14–7.
3. Shah BA, Padbury JF. Neonatal sepsis: an old problem with new insights. Virulence. 2014;5(1):170–8.
4. Töllner U. Early diagnosis of septicemia in the newborn. Clinical studies and sepsis score. Eur J Pediatr. 1982;138(4):331–7.
5. Wynn J, Cornell TT, Wong HR, Shanley TP, Wheeler DS. The host response to sepsis and developmental impact. Pediatrics. 2010;125(5):1031–41.
6. Manucha V, Rusia U, Sikka M, Faridi MMA, Madan N. Utility of haematological parameters and C-reactive protein in the detection of neonatal sepsis. J Paediatr Child Health. 2002;38(5):459–64.
7. Mahieu LM, De Muynck AO, De Dooy JJ, Laroche SM, Van Acker KJ. Prediction of nosocomial sepsis in neonates by means of a computer-weighted bedside scoring system (NOSEP score). Crit Care Med. 2000;28(6):2026–33.
8. Guerti K, Devos H, Ieven MM, Mahieu LM. Time to positivity of neonatal blood cultures: fast and furious? J Med Microbiol. 2011;60(Pt 4):446–53.
9. Cortese F, Scicchitano P, Gesualdo M, Filaninno A, De Giorgi E, Schettini F, et al. Early and Late Infections in Newborns: Where Do We Stand? A Review. Pediatr Neonatol. 2016;57(4):265–73.
10. Kite P, Millar MR, Gorham P, Congdon P. Comparison of five tests used in diagnosis of neonatal bacteraemia. Arch Dis Child. 1988;63(6):639–43.
11. Khaertynov KS, Boichuk S V, Khaiboullina SF, Anokhin VA, Andreeva AA, Lombardi VC, et al. Comparative Assessment of Cytokine Pattern in Early and Late Onset of Neonatal Sepsis. J Immunol Res. 2017;2017:8601063.
12. Rodwell RL, Taylor KM, Tudehope DI, Gray PH. Hematologic scoring system in early diagnosis of sepsis in neutropenic newborns. Pediatr Infect Dis J. 1993;12(5):372–6.
13. Fairchild KD, O’Shea TM. Heart rate characteristics: physiomarkers for detection of lateonset neonatal sepsis. Clin Perinatol. 2010 ;37(3):581–98.
14. Okascharoen C, Sirinavin S, Thakkinstian A, Kitayaporn D,Supapanachart S. A bedside prediction-scoring model for late-onset neonatal sepsis. J Perinatol. 2005;25(12):778–83.
15. Sundaram V, Dutta S, Ahluwalia J, Narang A. Score for neonatal acute physiology II predicts mortality and persistent organ dysfunction in neonates with severe septicemia. Indian Pediatr . 2009 ;46(9):775–80.
16. Griffin MP, Lake DE, O’Shea TM, Moorman JR. Heart rate characteristics and clinical signs in neonatal sepsis. Pediatr Res. 2007 ;61(2):222–7.
17. Chiesa C, Pellegrini G, Panero A, Osborn JF, Signore F, Assumma M, et al. C-reactive protein, interleukin-6, and procalcitonin in the immediate postnatal period: influence of illness severity, risk status, antenatal and perinatal complications, and infection. Clin Chem. 2003;49(1):60–8.
18. Manroe BL, Weinberg AG, Rosenfeld CR, Browne R. The neonatal blood count in health and disease. I. Reference values for neutrophilic cells. J Pediatr. 1979;95(1):89–98.
19. Chen Q, Shi J, Fei A, Wang F, Pan S, Wang W. Neutrophil CD64 expression is a predictor of mortality for patients in the intensive care unit. Int J Clin Exp Pathol. 2014;7(11):7806–13.
20. Kuppala VS, Meinzen-Derr J, Morrow AL, Schibler KR. Prolonged initial empiri-
cal antibiotic treatment is associated with adverse outcomes in premature infants. J Pediatr. 2011;159(5):720–5.
21. Camacho-Gonzalez A, Spearman PW, Stoll BJ. Neonatal infectious diseases: evaluation of neonatal sepsis. Pediatr Clin North Am. 2013;60(2):367–89.
22. Hofer N, Zacharias E, Müller W, Resch B. An update on the use of C-reactive protein in early-onset neonatal sepsis: current insights and new tasks. Neonatology. 2012;102(1):25–36.
23. Chu A, Hageman JR, Schreiber M, Alexander K. Antimicrobial Therapy and Late Onset Sepsis. Neoreviews. 2012;13(2):e94–102.
24. Berger C, Uehlinger J, Ghelfi D, Blau N, Fanconi S. Comparison of C-reactive protein and white blood cell count with differential in neonates at risk for septicaemia. Eur J Pediatr. 1995;154(2):138–44.
25. Mahieu LM, De Dooy JJ, Cossey VR, Goossens LL, Vrancken SL, Jespers AY, et al. Internal and external validation of the NOSEP prediction score for nosocomial sepsis in neonates. Crit Care Med. 2002;30(7):1459–66.
26. Singh SA, Dutta S, Narang A. Predictive clinical scores for diagnosis of late onset neonatal septicemia. J Trop Pediatr [Internet]. 2003 Aug;49(4):235–9.
27. Fanaroff AA, Korones SB, Wright LL, Verter J, Poland RL, Bauer CR, et al. Incidence, presenting features, risk factors and significance of late onset septicemia in very low birth weight infants. The National Institute of Child Health and Human Development Neonatal Research Network. Pediatr Infect Dis J. 1998;17(7):593–8.
28. Khatua SP, Das AK, Chatterjee BD, Khatua S, Ghose B, Saha A. Neonatal septicemia. Indian J Pediatr. 53(4):509–14.
29. Bhandari V, Wang C, Rinder C, Rinder H. Hematologic profile of sepsis in neonates: neutrophil CD64 as a diagnostic marker. Pediatrics. 2008;121(1):129–34.
30. Coggins SA, Weitkamp J-H, Grunwald L, Stark AR, Reese J, Walsh W, et al. Heart rate characteristic index monitoring for bloodstream infection in an NICU: a 3-year experience. Arch Dis Child Fetal Neonatal Ed. 2016;101(4):F329-32.
31. Newman TB, Draper D, Puopolo KM, Wi S, Escobar GJ. Combining immature and total neutrophil counts to predict early onset sepsis in term and late preterm newborns: use of the I/ T2. Pediatr Infect Dis J. 2014;33(8):798–802.
32. Ree IMC, Fustolo-Gunnink SF, Bekker V, Fijnvandraat KJ, Steggerda SJ, Lopriore E. Thrombocytopenia in neonatal sepsis: Incidence, severity and risk factors. PLoS One. 2017;12(10):e0185581.
33. Murphy K, Weiner J. Use of leukocyte counts in evaluation of early-onset neonatal sepsis. Pediatr Infect Dis J. 2012;31(1):16–9.
34. Benitz WE, Han MY, Madan A, Ramachandra P. Serial serum C-reactive protein levels in the diagnosis of neonatal infection. Pediatrics. 1998;102(4):E41.
35. Cantey JB, Wozniak PS, Sánchez PJ. Prospective surveillance of antibiotic use in the neonatal intensive care unit: results from the SCOUT study. Pediatr Infect Dis J. 2015;34(3):267–72.
36.
70 (3): 217-20.
37. Benitz WE. Adjunct laboratory tests in the diagnosis of early-onset neonatal sepsis. Clin Perinatol. 2010;37(2):421–38.
6979335279
e-mail: stefi_ntinou@hotmail.com
Correspondence
Ntinou Stefania-Rafailia
Koritsas 23, 5133
Kalamaria-Thessaloniki Τ. +302310446478, +306979335279
e-mail: stefi_ntinou@hotmail.com
Ntinou Stefania-Rafailia, Hristara-Papadopoulou Alexandra, Hatziagorou Elpida, Tsanakas Ioannis, Fitili Dimitra
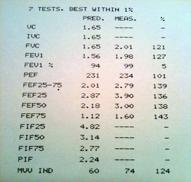
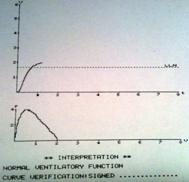
Background: Respiratory physiotherapy should be a daily activity in the life of Cystic Fibrosis patients. However, the patients usually tend to neglect the importance of respiratory physiotherapy, due to its time-consuming techniques. Through researches, easy to use, time saving, economical and effective devices have been developed to support those techniques. The purpose of this study is to compare the effects of two respiratory devices, PEP and Flutter, on patients with cystic fibrosis who have been admitted to the hospital for intravenous administration. Material and methods: Seventeen (17) Cystic Fibrosis patients took part in the study. The maximum exhaled air volume of first second (FEV1), LCI (Lung Clearance Index) and their weight were measured. The study population was divided into 2 groups: Group A: Physiotherapy session with PEP and active cycle of breathing technique (6 individuals) and Group B: Physiotherapy session with Flutter and active cycle of breathing technique (11 individuals).
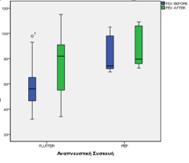
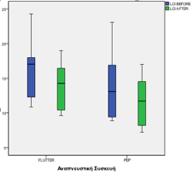
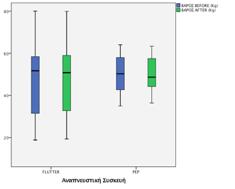
Results: The mean value of the variable FEV1 with the Flutter device increased from 59.72 to 73.54 units. Differences with PEP do not show marked changes in FEV1 from 82.13 to 87.08. The mean value of the variable LCI with the Flutter device dropped from 15.80 to 13.82 units. Moreover, the PEP device showed a corresponding decrease in the mean value of the variable LCI by 2.35 units (from 14.07 to 11.72). Finally, the average weight value increased from 47.83 to 48.31 when using the Flutter device, while with the PEP device there was a decrease from 50.03 to 49.80.
Conclusions: There was a statistically significant difference in FEV1 and the weight of the individuals using the Flutter device, while there was no statistically significant difference in the LCI. In PEP group, there was a statistically significant difference in the FEV1 and in the LCI, but there was no difference observed in the weight. Based on our results, we could not conclude that one device is superior to another.
Key words: Cystic Fibrosis, Respiratory Physiotherapy, PEP Device, Flutter Device, Active Cycle of Breathing Technique
Ntinou Stefania-Rafailia
Hristara-Papadopoulou
Alexandra
Fitili Dimitra
Department of physiotherapy, Alexander Technological Educational Institute of Thessaloniki, Greece
Hatziagorou Elpida
Tsanakas Ioannis
Pediatric Clinic, Pediatric Pulmonary Unit, Aristotle University of Thessaloniki, Gen. Hospital Ippokrateio, Thessaloniki, Greece
ATS/ERS (Beydon N, Davis SD, Lombardi E, Allen JL et al.
(Vitalograph 2120, VitalographLtdEnnis, Ireland).
(Exhalyzer D, Ecomedics,
(Fuchs
Gappa M.,
Winden, A. Visser, et
Magdalen Gondor, Patricia A.
1. Lowe CU, May CD, Reed SC. (1949) Fibrosis of the pancreas in infants and children: a statistical study of clinical and hereditary features. Am J Dis Child. 1949;78:349–374
2. Elkins MR, Jones A, Van der Schams C. (2006). Positive expiratory pressure physiotherapy for airway clearance in people with cystic fibrosis. Cochrane Database Systimatic Review;19:CD003147
3. Feldman J, Traver GA, Tuassig LM. (1979) Maximal expiratory flows after postural drainage. Am Rev Respir Dis 1979;119:239-45.
4. Maxwell M, Redmond A. (1979) Comparative trial of manual and mechanical percussion techniques with gravity assisted bronchial drainage in patients with cystic fibrosis. Arch Dis Child 1979;54:542-4.
5. Pryor JA, Webber BA, Hodson ME, Batten JC., (1979) Evaluation of the forced expiration technique as an adjunct to postural drainage in treatment of cystic fibrosis. Br Med J 1979;ii:4178
6. Bradley JM, Moran FM, Elborn JS., (2006) Evidence for physical therapies (airway clearance and physical training) in cystic fibrosis: An overview of five Cochrane systematic reviews. Respiratory Medicine 100, 191–201
7. Garrod R, Lasserson T., (2007) Role of physiotherapy in management of chronic lung disease: An overview of systematic reviews. Respiratory Medicine 101, 2429–2439
8. Hristara – Papadopoulou A, Tsanakas J, Diomou G, Papadopoulou O. (2008) Current devices of respiratory physiotherapy. REVIEW ARTICLE, HIPPOKRATIA, 12, 4: 211-220
9. Mcllwaine PM, Wong LT, Peacock D, Davidson AG., (2001) Long-term comparative trial of positive expiratory pressure versus oscillating positive expiratory pressure (flutter) physiotherapy in the treatment of cystic fibrosis. J Pediatr 2001; 138: 845-849
10. Orlik T, Sands D., (2001) Long-term evaluation of effectiveness for selected chest physiotherapy methods used in the treatment of cystic fibrosis. Med. Wieku Rozwoj. 5 (3), 245–257
11. Schechter MS., (2007) Airway clearance applications in infants and children. Respiratory Care 52, 10
12. Beydon N, Davis SD, Lombardi E, Allen JL, Arets HG, Aurora P, Bisgaard H, Davis GM,
Ducharme FM, Eigen H, Gappa M, Gaultier C, Gustafsson PM, Hall GL, Hantos Z, Healy MJ, Jones MH, Klug B, Lødrup Carlsen KC, McKenzie SA, Marchal F, Mayer OH, Merkus PJ, Morris MG, Oostveen E, Pillow JJ, Seddon PC, Silverman M, Sly PD, Stocks J, Tepper RS, Vilozni D, Wilson NM, (2007) An official American Thoracic Society/European Respiratory Society statement: pulmonary function testing in preschool children. Am J Respir Crit Care Med. 2007;175:1304–45.
13. Fuchs SI, Gappa M. (2011) Lung clearance index: clinical and research applications in children. Paediatric respiratory reviews. 2011;12(4):264-70.
14. Winden C.M.Q., Visser A., Hop W., Sterk P.J., Beckers S., J.C. de Jongste, (1998) Effects of flutter and PEP mask physiotherapy on symptoms and lung function in children with cystic fibrosis, Copyright ©ERS Journals Ltd 1998 European Respiratory Journal ISSN 0903 – 1936
15. Gondor Magdalen, MD, Nixon Patricia A., PhD, Mutich Rebecca, RRT, Rebovich Paul, MS, Orenstein David M., MD, (1999) Comparison of Flutter Device and Chest Physical Therapy in the Treatment of Cystic Fibrosis Pulmonary Exacerbation, Pediatric Pulmonology 28:255–260
16. Tyrrell J.C., Hiller E.J., Martin J., (1986) Face mask physiotherapy in cystic fibrosis, Archives of Disease in Childhood, 1986, 61, 598-611
17. Braggion C, MD, L.M. Cappelletti, PT, M. Cornacchia, PT, L. Zanolla, MD, and G. Mastella, MD, (1995) Short-Term Effects of Three Chest Physiotherapy Regimens in Patients Hospitalized for Pulmonary Exacerbations of Cystic Fibrosis: A Cross-Over Randomized Study, Pediatric Pulmonology 19:16-22
18. Mcllwaine PM, Wong LT, Peacock D, Davidson AG., (2001) Long-term comparative trial of positive expiratory pressure versus oscillating positive expiratory pressure (flutter) physiotherapy in the treatment of cystic fibrosis. J Pediatr 2001; 138: 845-849
19. Borka P, Gyurkovits K, Bódis J, (2012) «Comparative study of PEP mask and Flutter on expectoration in cystic fibrosis patients» , Acta PhysiologicaHungarica, Volume 99 (3), pp. 324–331
(Peak Expiratory Flow,

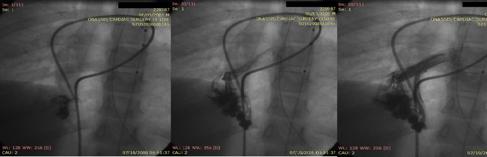
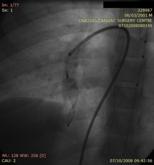
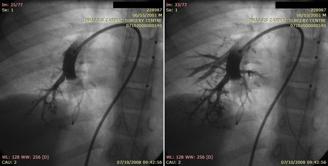
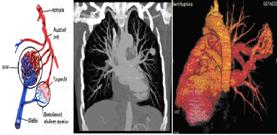

AVP= Amplatzer vascular plugs
Rendu-Osler-Weber.
Willebrand. Ωστόσο,
Βιβλιογραφία
1. DanielaS.Ardelean and Michelle Letarte, «Anti-angiogenic therapeutic strategies in hereditary hemorrhagic telangiectasia», frontiers in GENETICS, published: 11February2015 doi:
10.3389/fgene.2015.0003
2. SCOTT E. OLITSKY, MD, Children’s Mercy Hospitals and Clinics, Kansas City, Missouri, “ Hereditary Hemorrhagic Telangiectasia: Diagnosis and Management”, Am Fam Physician. 2010 Oct 1;82(7):785-90.
3. Westermann CJ, Rosina AF, De Vries V, de Coteau PA. «The prevalence and manifestations of hereditary hemorrhagic telangiectasia in the Afro-Caribbean population of the Netherlands Antilles: a family screen¬ing.» Am J Med Genet A. 2003;116A(4):324-328.
4. Kjeldsen AD, Oxhøj H, Andersen PE, Elle B, Jacobsen JP, Vase P., «Pul¬monary arteriovenous malformations: screening procedures and pulmonary angiography in patients with hereditary hemorrhagic telan-giectasia.» Chest. 1999;116(2):432-439.
5. Trerotola SO, Pyeritz RE, «PAVM embolization: an update.» AJR Am J Roentgenol 195: 837-845, 2010.
6. S.Akiyama, S.Hanada, H.Uruga, H.Takaya, A.Miyamoto, N.Morokawa, A.Kurosaki, T.Fujii, T.Kohno and K.Kishi, «Hereditary Hemorrhagic Telangiectasia with Pulmonary Arteriovenous Malformations and Embolic Strokes Treated Successfully with Video-assisted Thoracoscopic Resection», Intern Med 52: 1091-1094, 2013)
7. J.McDonald, W.Wooderchak-Donahue, C.Webb , K.Whitehead, D.A.Stevenson and P.Bayrak-Toydemir, «Hereditary hemorrhagic telangiectasia: genetics and molecular diagnostics in a new era», frontiers in GENETICS, published: 26 January 2015 doi: 10.3389/ fgene.2015.00001
8. Σ.Μάρακα, Ε.Σαριδάκης, Β.Αγγελάκου, Σ.Μπάκωση, Γ.Βλαχάκη,
(σύνδρομο Rendu-Osler-Weber).
2004;67:200-204
9. http://emedicine.medscape.com/article/2048472-overview
10. Ν.Ματσανιώτης, Θ.Καρπάθιος
1999;S1385-1386
11. Behrman, Kliegman, Arviin, «Nelson, Παιδιατρική ιι», 2000;S2096
12. http://www.google.gr/imgres?imgurl=http://www.cirse.org/_files/ fotoalbum//0001_0002_1134658943_0001.jpg&imgrefurl=http://www.cirse.org/print.php?pid%3 D182%26lang%3D5&h=350&w=400&tbnid=m46qtATEOa8NUM:&zoom=1&docid=N6I nWmB8kdfgUM&ei=eFocVdjpNsWoPICMgMgE&tbm=isch&ved=0CB8QMygBMAE 13. http://www.google.gr/imgres?imgurl=http://www.cirse.org/_files/ fotoalbum//0001_0002_1134658936_0001.jpg&imgrefurl=http://www.cirse.org/print.php?pid%3 D182%26lang%3D5&h=350&w=400&tbnid=MziAcivkayryhM:&zoom=1&docid=N6InW mB8kdfgUM&ei=eFocVdjpNsWoPICMgMgE&tbm=isch&ved=0CCAQMygCMAI 14. https://www.google.gr/url?sa=i&rct=j&q=&esrc=s&source=images&cd =&cad=rja &uact=8&ved=0CAcQjRw&url=http%3A%2F%2Friversideonline.com%2Fhea lth_ reference%2Fnervous-system%2Fds01126.cfm&ei=6l0cVZy4FtbvaP2tgJAG&bvm=bv.89744 112,d.d2s&psig=AFQjCNGrdZJ7gbJDfUd-kq71lEzY4eOq3w&ust=1428008323249393
15. http://www.annalspc.com/article.asp?issn=0974-2069;year=2013;volume=6;issue=2;spage= 132;epage=140;aulast=Barwad
16. P.M. Agnollitt, A.R. Faηanha Barreto, R.F.Pinsetta Barbieri, J.E. Junior, V.Muglia, “RenduOsler-Weber syndrome: what radiologists should know. Literature review and three cases report”, Radiol Bras. 2013 Mai/Jun;46(3):168–172
17. Cottin V, Dupuis-Girod S, Lesca G, et al., “Pulmonary vascular manifestations of hereditary hemorrhagic telangiectasia (rendu-osler disease).” Respiration,2007;74:361–78.
18. N.Garg, M.Khunger, A.Gupta, N.Kumar, “ Optimal management of hereditary hemorrhagic telangiectasia”, Journal of Blood Medicine, 2014:5
19. M.Aftab Alam, S.Sami, and S.Babu, «Successful treatment of bleeding gastro-intestinal angiodysplasia in hereditary haemorrhagic telangiectasia with thalidomide», BMJ Case Rep. 2011; 2011: bcr0820114585.
e-mail: vgiapros@uoi.gr
Correspondence
Vasileios Giapros
T. +302651099326
M.+30 6974754893
e-mail: vgiapros@uoi.gr
A. Drougia, F. Balomenou, M. Katsalouli, E. Domouzoglou, V. Giapros
Abstract
Introduction: Transient neonatal myasthenia Gravis is a rare disorder that affects 10-20% of neonates of mothers with myasthenia gravis (MG). In most cases it is caused by transplacental transfer of maternal antibodies against the acetylcholine receptor at the neuromuscular junction. Myasthenic symptoms are manifested during the first 24-72 hours of life with variable severity. The risk for the neonate to develop symptoms is not linked to the maternal state of MG nor to the antibody title. It is a transient disorder, and if not treated promptly it could be life threatening.
Case presentation: A neonate of a mother with generalized MG in full clinical remission during the pregnancy is presented. The older sibling was born after a pregnancy with exacerbation of myasthenia, and he remained asymptomatic. The neonatein our case, developed gradually after birth poor sucking, weak cry and hypotonia-muscle weakness, clinical signs that led to the diagnosis of transient neonatal MG. The neonate was tube fed and treatment with pyridostigmine was initiated with good response and total recovery. This case is reported due to the rarity of the disorder and the different impacts of MG in neonates born to the same mother.
Conclusions: The impacts of MG in the pregnant and her offsprings are variable in each case. Τhe close observation of all neonates born to mothers with MG It is of utmost importance for the prompt recognition of the rare cases of TNMG and the initiation of appropriate treatment.
Key words: transient neonatal myasthenia gravis, pregnancy
A. Drougia
F. Balomenou
E. Domouzoglou
V. Giapros
NICU, University Hospital of Ioannina, Ioannina
M. Katsalouli
Neurology Department, Children’s Hospital "Agia Sofia", Athens
Gravis: MG
1. Oger J, Frykman H. An update on laboratory diagnosis in myasthenia gravis. ClinChimActa 2015;449:43-48.
2.Gilhus NE, Hong Y. Maternal myasthenia gravis represents a risk for the child through autoantibody transfer, immunosuppressive therapy and genetic influence. European Journal of Neurology 2018;25:1402-1409.
3. Avidan N, Le Panse R, Berrih-Aknin S, Miller A. Genetic basis of myasthenia gravis - A comprehensive review. J Autoimmun 2014;52:146-153.
4. Berrih-Aknin S, Frenkian-Cuvelier M, Eymard B. Diagnostic and clinical classification of autoimmunemyasthenia gravis. J Autoimmun 2014;48-49:143-148.
5.Gilhus NE, Verschuuren JJ. Myasthenia gravis: subgroup classification and therapeutic strategies. Lancet Neurol 2015;14:1023-1036.
6. Varner M. Myasthenia gravis and pregnancy. Clinical Obstet Gynecol2013;56:372-381.
7. Ferrero S, Pretta S, Nicoletti A, Petrera P, Ragni N. Myasthenia gravis: management issues during Pregnancy. Eur J Obstet Reprod Biol. 2005;1;121:129-138.
8. Papazian O. Transient neonatal myasthenia gravis. J Child Neurol 1992;7:135-141.
9. Norwood F, Dhanjal M, Hill M, James N, Jungbluth H, Kyle P, et al. Myasthenia in pregnancy: best practice guidelines from a U.K. multispecialty working group. J Neurol Neurosurg Psychiatry2014;85:538-543.
10. Hoff JM, MidelfartA. Maternal myasthenia gravis: a cause for arthrogryposis multiplex congenita. J Child Orthop2015;9:433-435.
11. Hacohen Y, Jacobson LW, Byrne S, Norwood F, Hall A, Robb S, et al.: Fetal acetylcholine receptor inactivation syndrome: A myopathy due to maternal antibodies. NeurolNeuroimmunol Neuroinflamm 2015;2:e57.
12. Kalidindi M, Ganpot S, Tahmesebi F, Govind A, Okolo S, Yoong W. Myasthenia gravis and pregnancy. J Obstet Gynaecol 2007;27:30-32.
13. Ducci RD, Lorenzoni PJ, Kay CS, Werneck LC, Scola RH. Clinical follow-up of pregnancy in myasthenia gravis patients. Neuromuscul Dis 2017;27:352-357.
14. Hoff JM, Daltveit AK, Gilhus NE. Myasthenia gravis in pregnancy and birth: identifying risk factors, optimising care. Eur J Neurol 2007;14:38-43.
15. Hoff JM, Daltveit AK, Gilhus NE. Myasthenia gravis: consequences for pregnancy, delivery, and the newborn. Neurology 2003;61:1362-1366.
16.Hoff JM, Daltveit AK, Gilhus NE. Asymptomatic myasthenia gravis influences pregnancy and birth. Eur J Neurol 2004;11:559-562.
17. Lee JY, Min JH,Han SH, Han J. Transient neonatal myasthenia gravis due to a mother with
ocular onset of anti-muscle specific kinase myasthenia gravis. Neuromascul Dis 2017;27:665657.
18. Hassoun M, Turjuman UE, Chokr I,Fakhoury H. Myasthenia Gravis in the Neonate. NeoReviews 2010;11e200-e205.
19.Townsel CT, Keller R, Johnson K, Hussain N, Campbell WA. Seronegative Maternal Ocular Myasthenia Gravis and Delayed Transient neonatal Myasthenia Gravis. Am J Perinatol Rep 2016;6:e133-e136.
20. Engel AG, Shen XM, Selcen D, Sine SM. Congenital myasthenic syndromes: pathogenesis, diagnosis, and treatment. LancetNeurol 2015;14:420-434.
K.
e-mail: johndrikos@yahoo. com
Correspondence
Dr. Ioannis Drikos
Gennimata Avenue, Magoula T.+302132028822
M. +306972710576
e-mail: johndrikos@yahoo. com
Garyfallia Syridou, Ioannis Drikos, Eleni Papa, Rougas Panagiotis, Dimitrios Kyriazopoulos, Antigoni Pegkou, Artemis Vintila, Eleni Antonopoulou
Summary
Introduction
Influenza virus primarily affects the respiratory system. It rarely causes extrapulmonary complications, with otitis media and febrile seizures being the most common in children. Acute glomerulonephritis as a complication of H1N1 influenza virus infection has been described only sporadically.
Methods
We present a case of acute glomerulonephritis in a previously healthy adolescent, in the context of infection with influenza A H1N1 virus.
Results
A 15-year old adolescent was admitted to our pediatric department due to fever up to 39.2°C since 36h before admission, along with pharyngalgia, cough, vomit, dizziness and fatigue. Based on his symptoms and the seasonal epidemiology, empiric treatment with oseltamivir was initiated while waiting for RT-PCR for influenza virus in pharyngeal swab, which turned out positive for A H1N1 influenza virus. In the first 24 hours of admission, the patient presented macroscopic haematuria, which completely subsided in the following days, along with fever recession. Urinary phase contrast analysis showed findings compatible with acute glomerulonephritis while the laboratory and imaging testing in the context of AGN were normal. Anti-streptolysin O (ASTO) levels, which were initially above normal (562 IU/ml), did not increase significantly in the following days, and given the fact that C3 levels were constantly within normal limits and pharyngeal culture was negative for pyogenic streptococcus, they were not considered sufficient for poststreptococcal glomerulonephritis diagnosis.
Conclusions
Influenza A virus infection is a rare cause of acute glomerulonephritis in children and should be considered in differential diagnosis in patients with flu-like illness and concomitant haematuria.
Key words: H1N1, acute glomerulonephritis, haematuria, complications of influenza
Garyfallia Syridou
Ioannis Drikos
Eleni Papa
Rougas Panagiotis
Dimitrios Kyriazopoulos
Antigoni Pegkou
Artemis Vintila
Eleni Antonopoulou
Department of Pediatrics, Thriasio General Hospital, Athens, Greece
έλεγχο διαπιστώθηκαν: WBC / 6400 / ml (Ν: 78,1%, L: 9,0%, Μ: 12,7%), Hb 12,4 g / dl, PLT 184000 / ml, CRP 44,6 mg / L. Ο βιοχημικός έλεγχος ήταν φυσιολογικός, ενώ στη γενική ούρων παρουσίασε αιματουρία
(ΕΒ: 1025 pH: 6,0 Αιμοσφαιρίνη (+++), Λέυκωμα: (-) Νιτρώδη: (-), Πυοσφαίρια: 3-5/κοπ Ερυθρά:
ASTO 562 IU / ml, C3 95,3 mg / dl C4 32,4 mg / dl, ΙgG 1050 mg / dl, IgA 173 mg / dl, IgM 108 mg/ dl και ANA (-), antiDNA(-), cANCA(-), pANCA (-), U/S NOK φυσιολογικό.
Κατά τη διάρκεια της νοσηλείας,
(ΕΒ: 1025 pH: 6,0 Αιμοσφαιρίνη (+++), Λέυκωμα: (+) Νιτρώδη: (-) Κετόνες: (-) Πυοσφαίρια: 1-3
Ερυθρά: 15-20/κοπ), με Ca/Cr=0.0076, Cr Ούρων=303mg/dl,
1. Jules A, Grijalva C, Zhu Y, Talbot H, Williams J, et al. Griffin Influenza-Related Hospitalization and ED Visits in Children Less Than 5 Years: 2000–2011. Pediatrics. 2015; 135(1): 66–74.
2. Sellers SA, Hagan RS, Hayden FG, Fischer WA. The hidden burden of influenza: A review of the extra-pulmonary complications of influenza infection. Influenza Other Respir Viruses. 2017; 11(5): 372-393.
3. Watanabe T. Renal complications of seasonal and pandemic influenza A virus infections. Eur J Pediatr. 2013; 172: 15–22.
4. Kupferman JC, Trachtman H, Spitzer ED. Acute glomerulonephritis and acute kidney injury associated with 2009 influenza A: H1N1 in an infant. Pediatr Nephrol. 2011; 26(1): 153-4.
5. Jain T, Hemington L, Etuwewe B. A case of post-infectious glomerulonephritis following infection with influenza A subtype H1N1. Pediatr Nephrol. 2011; 26(1): 151-2.
6. Ashtiani N, Mulder M, van Wijk J, Bokenkamp A. A case of tubulointerstitial nephritis in a patient with an influenza H1N1 infection. Pediatr Nephrol. 2012; 27(10): 1985–1987.
7. Ghiggeri GM, Losurdo G, Ansaldi F, Canepa A, Magnasco A. Two cases of swine H1N1 influenza presenting with hematuria as prodrome. Pediatr Nephrol. 2010; 25(4):779-80.
8. Watanabe T, Yoshikawa H, Abe Y, Yamazaki S, Uehara Y, Abe T. Renal involvement in children with influenza A virus infection. Pediatr Nephrol. 2003; 18: 541–544.
9. Scott E. Wenderfer. Viral-associated glomerulopathies in children. Pediatr Nephrol. 2015; 30(11): 1929–1938.
10. Lee N, Chan PKS, Lui GC, et al. Complications and Outcomes of Pandemic 2009 Influenza A (H1N1) Virus Infection in Hospitalized Adults: How Do They Differ From Those in Seasonal Influenza? The Journal of Infectious Diseases. 2011; 203(12): 1739–1747.
11. Carmona F, Carlotti AP, Ramalho LN, Costa RS, Ramalho FS. Evidence of renal infection in fatal cases of 2009 pandemic influenza A (H1N1). Am J Clin Pathol. 2011; 136: 416–423.
12. To KK, Chan KH, Li IW, Tsang TY, Tse H, Chan JF, et al. Viral load in patients infected with pandemic H1N1 2009 influenza A virus. J Med Virol. 2010; 82(1): 1-7.
έτοιμα,
run the ECG, run the videos, run the cameras,


2.
3.
4.
5.
6.
7.
8.
9. Ενδιαφέρουσες
10. Σύντομα
11. Βραχείες
12.
13.
14.
(Uniform Requirements for Manuscripts Submitted to Biomedical Journals),
International Committee of Medical Journal Editors/Uniform Requirements for Manuscripts Submitted to Biomedical Journals, (www.icmje.org και www.icmje.org/icmje. pdf).
Cumulated Index Medicus [List of Journals Indexed in Index Medicus (www.nlm.nih.goν/bsd/uniform_requirements. html)].
Proesmans W. Bartter syndrome and its neonatal νariant. Eur J Pediatr 1997;156:669-679.
Flyvbjerg Α. Role of growth hormone, insulin-like growth factors (IGFs) and IGF-binding proteins in the renal complications of diabetes. Kidney Ιnt 1997;52 (60 Suppl):S12-S19.
Χωρίς συγγραφέα:
National Institutes of Health Consensus Deνelopment Conference. Neurofibromatosis conference statement. Arch Neurol1988;45:575-578.
Προσδιορισμός τύπου άρθρου:
Schreiner GF, Lange L. Ethanol modulation of macrophage influx in glomerulonephritis [Abstract]. J Am Soc Nephrol 1991;2:562.
Should antileukotriene therapies be used instead of inhaled corticosteroids in asthma? [Editorial]. Am J Respir Crit Care Med 1998;158:1697-1701.
Laux-End R, Inaebnit D, Gerber ΗΑ, Bianchetti MG. Vasculitis associated with leνamisole and circulating autoantibodies [Let ter]. Arch Dis Child 1996;75:355-356.
II. ΒΙΒΛΙΑ
Clark AG, Barratt ΤΜ. Steroid-responsiνe nephrotic syndrome. Ιn: Barratt ΤΜ, Arner ED, Harmon WE, editors. Pediatric Nephrology. 4th ed. Baltimore: Lippincott William Wilkins; 1999. p. 742.
Σύγγραμμα
Gorlin RJ, Cohen ΜΜ, Leνin LS. Syndromes of the head and neck. 3rd ed. New York: Oxford Uniνersity Press; 1990.
Δημοσίευση
Bauer ΑW. The two definitions of bacterial resistance. In: Smith AJ, Rogers CA, eds. Proceedings of the Third International Congress of Chemotherapy; 1962 May 29-31; New York: International Society of Chemotherapy; 1963. p. 484-500.
Διδακτορική
Πανεπιστήμιο Αθηνών; 1979.
Kaplan SJ. Post hospital home health care: the elderly’s access and utilization [dissertation]. St. Louis (Μο): Washington Univ.;1995.
III. CD-ROM
Andersoη SC, Poulsen ΚΒ. Anderson’s electronic atlas of hematology [CD-ROM]. Philadelphia: Lippincott Williams & Wilkins; 2002.
IV. ΣΤΟ ΔΙΑΔΙΚΤΥΟ
Abood S. Quality improνement initiatiνe in nursiηg homes: the ΑΝΑ acts in an adνisory role. Am J Nurs [Internet]. 2002 Jun:
Webpage: http://www.nursingworld.org/AJN/2002/june/Wawatch.htm
Μονογραφία: Foley ΚΜ, Gelbaηd Η, editors. Improving palliative care for cancer [Monograph, Internet]. Washington: National Academy Press; 2001.
Webpage: http://www.nap.edu/books/0309074029/html
Ιστοσελίδες: Cancer-Pain.org [Webpage, Internet]. New York: Association of Cancer Online Resources, Ιnc.; 2002: http://www.cancer-pain.org/
Πίνακες
•
•
• επιστημονικό/ά κέντρο/α
•
2.
3.
4.
5.
7.