

INSIDE:
Alberta’s AI health strategy
Alberta’s CMIO outlines the province’s approach to healthcare AI. Part of the plan includes building partnerships with clinicians, innovators, and healthcare organizations.
Page 4
HIFU in Sault Ste. Marie Sault Area Hospital has become the first in Canada to deploy the Neurolyser XR High-Intensity Focused Ultrasound system in a clinical setting. The system is being used for pain management.
Page 6
Future reading room
The diagnostic imaging reading room of the future will likely see radiologists talking to their computers like they are colleagues, asking for opinions on the results of MR and CT scans, and any number of tests.
Page 15

inventors honoured at UHN awards
An interdisciplinary group at UHN that was responsible for creating and launching the patient monitoring innovation called H.A.L.O (Human Attended Live Observation) has won the hospital’s Mission of Excellence Team Inventor of the Year Award. Pictured are H.A.L.O. clinical inventors and co-founders, Marijana Zubrinic and Dr. Shaf Keshavjee. SEE STORY ON PAGE 10.
Schroeder Ambulatory Centre aims to reduce backlogs
BY JERRY ZEIDENBERG
RICHMOND HILL, ONT. – The Schroeder Ambulatory Centre (SAC), a charitable non-profit facility in Richmond Hill, just north of Toronto, has been outfitted with operating rooms, CT and MRI scanners, and a host of other equipment and capabilities. The $300 million facility has begun offering services while awaiting final approvals from Accreditation Canada for diagnostic imaging and hip and knee surgical programs.
Eventually, it hopes to do even more –functioning as a comprehensive, not-forprofit ambulatory health centre.
But as a private medical facility, the first question people ask is, ‘aren’t you going to be moving surgeons, radiologists, nurses and technologists away from resource-starved, traditional hospitals and into your clinic?’
In response, chief medical officer Dr. Fabio Varlese told Canadian Healthcare Technology
that he and his colleagues have been sensitive to this issue for the four years the Schroeder Ambulatory Centre has been in the planning and construction stages. He said the key idea is to add capacity to the Ontario healthcare system, and not to diminish it.
“It was crucial for us to find a way to not
The
key idea is to add capacity to Ontario's healthcare system, not to diminish it.
simply shift capacity from the hospital system to facilities like ours,” he said.
So, when it comes to surgeries, he noted that most Ontario surgeons are only in the OR one or two days a week. SAC will enable them to do surgeries on other days, which will increase surgical capacity in the region. Dr. Varlese said they won’t be taking these surgeons away from their regular hospital
duties but will give them the resources to do more operations – to the benefit of patients.
As he puts it, “physicians will continue to deliver most of their care from their home hospitals.”
In July 2025, the Ontario government announced it will invest $155 million over two years to add 57 new community surgical and diagnostic centres licensed to deliver MRI and CT scans, and gastrointestinal (GI) endoscopy services across the province. The expansion will help 1.2 million people access publicly funded procedures faster, the Doug Ford government said.
For its part, the Schroeder Ambulatory Centre is receiving $14 million to provide MRI and CT scans, as well as GI endoscopy procedures to over 115,000 patients over two years, significantly reducing wait times in the region.
Adding private medical centres to the mix
Centre aims to reduce backlogs with high-quality DI, outpatient surgery
is a controversial issue, as it’s a modification of the public healthcare system that is sacrosanct to many people. Critics worry that private facilities will simply poach personnel from the public healthcare system.
As a result, centres like the SAC are under pressure to find solutions that don’t disrupt the existing healthcare system.
When it comes to the nurses that are needed, the centre is being creative. First, its recruitment strategy focuses on empowering healthcare professionals to extend their careers in a setting that prioritizes sustainable practice. By recruiting retirees, those seeking a more balanced pace, and practitioners looking for a fresh environment, SAC offers an alternative to traditional acute care.
This model provides a predictable schedule with defined hours and no overnight shifts, allowing professionals to work in the field on terms that work for their lives.
Secondly, it has struck up relationships with McMaster University and Toronto Metropolitan University to provide practical experience to their nursing students as part of their curriculum.
“We’re not doing the theory, we’re doing the practical part, and this has always been a big problem for academic institutions; they’re always looking for facilities that would deliver high-quality, practical
training and teaching, and we are becoming that,” said Dr. Varlese. “So, the ultimate outcome of that is that we’re adding to the system.”
He said SAC is using a similar recruitment strategy for radiologists and technologists and is providing extra hours to working radiologists and technologists who wish to fill up their schedules.
Dr. Varlese noted that SAC will be working closely with facilities in its area –Mackenzie Health is the closest hospital, North York General and Southlake Regional Health Centre are also nearby.
He said the Centre recently came to the assistance of a large regional hospital that is currently upgrading its MRI suites and needed to shut down its machines. During that time, it shifted patients to SAC, which has two Siemens MRI scanners installed –a 1.5T and a 3.0T machine.
Using equipment, they’ve been able to image 2,000 patients.
He said a partnership like this could be ongoing, with the Schroeder centre assisting area hospitals when they need extra capacity.
Dr. Varlese stressed that SAC is investing across the board in top-flight technologies that will not only help patients but will also integrate with the existing healthcare system and keep all physicians and allied healthcare professionals in the loop.
For example, they have acquired a cloud-

based MEDITECH Expanse electronic health record system that’s said to be the first of its kind in Canada. It is also acquiring a cloud-based Picture Archiving and Communications System (PACS) from Philips.
As Dr. Varlese noted, it’s important to give physicians and other healthcare professionals the tools they need track patient care and communicate with each other inside the organization.
These electronic solutions will also integrate with systems outside of SAC, ensuring that every clinician caring for a patient gets the feedback and information that they need.
“With high expectations of quality and outcomes, we knew we needed a hospitalgrade electronic record system,” said Dr.
Toronto-based GoAutomate develops powerful AI
BY JERRY ZEIDENBERG
Powerful AI agents that can improve DI patient flow and reduce stress and burnout among radiologists and technologists are being developed by GoAutomate, a Toronto-area company. GoAutomate originally came out of the banking sector, and it’s applying a great deal of expertise learned there to problems and bottlenecks in DI and other areas of healthcare.
For example, when it comes to diagnostic imaging, agents have been created that can read incoming requisitions – whether by fax or electronically – and automatically schedule the exams.
They could be MRIs, angiograms, or any number of tests performed at an imaging centre.
Agents can automatically send a message to the patient’s referring physician, letting them know that the exam has been booked – quickly eliminating a tedious but useful task for administrative staff.
Not only can the AI agents fill a medical centre’s DI schedule in this way, but they’re
also helping with the protocolling of exams – something that seems futuristic but is actually happening now.
“We’ve trained our system on hundreds of thousands of protocolling records,” said Jag Basrai, CEO of GoAutomate.
He explained that the agents have been trained to determine the right protocol based on the information presented in the
AI agents have been created that can read incoming requisitions and automatically schedule the exams.
requisition. What’s more, the system can be attuned to the nuances and preferences of the technologists and radiologists working at various facilities.
This can be done for any hospital or clinic using the system, said Basrai.
“It knows all the standard protocols and the specific nuances of Ontario hospitals,” said Basrai.
agents for DI
If there is an element of doubt about which procedure should be chosen, the agent will default to be reviewed by a technologist or radiologist.
When this happens, it takes the changes and uses it to further refine the AI model –a feedback loop that further improves the system.
“It goes back into the training,” said Basrai, explaining that the AI agents then become even more effective.
One customer that’s currently evaluating GoAutomate’s AI agents is the new Schroeder Ambulatory Centre in Richmond Hill, Ont., a private, not-for-profit medical centre that’s designed to perform medical imaging and some surgeries.
It has been outfitted with leading-edge technologies, including MRI scanners, as well as sophisticated software such as the cloud-based MEDITECH called MaaS (the first deployment in Canada) and a cloudbased PACS from Philips.
Dr. Fabio Varlese, medical director, told CHT that Schroeder has also created an AI
CONTINUED ON PAGE 23
Publisher & Editor
Jerry Zeidenberg jerryz@canhealth.com
Office Manager
Varlese. He said there were discussions about acquiring an EMR that’s more typically found in physician clinics, but the hospital systems have a higher capacity to connect and interoperate.
“We need full integration [with local hospital partners]. We’ve seen that disjointed care is a big problem. Even if the patient is receiving high-quality care, if it’s disjointed, it’s the biggest risk when it comes to patient care and outcomes,” he said.
That’s because unless all clinicians involved in a patient’s care are informed of tests and procedures, care for the patient can stall, and he or she may fall through the cracks. By using top-tier electronic records and communications, SAC intends to keep all allied professionals in the loop. By deploying advanced systems, the Schroeder centre intends to give professionals the tools they need.
To support this, the centre is actively integrating artificial intelligence across its systems to enhance the efficiency and precision of patient care.
“We want to be incredibly focused on the ability to continuously improve after analyzing the high-quality data we produce,” said Dr. Varlese.
“There’s a genuine interest in really looking at quality of care, patient outcomes, patient satisfaction and full integration with the healthcare system – which is going to be the backbone of improved services and outcomes.”
It’s already at work on AI projects, focusing first on foundational efficiencies such as deploying and refining AI scribes. The centre is also exploring solutions to streamline administrative processes and optimize scheduling to reduce cancellations and no-shows.
While technology is important, connections among real people are paramount. On this note, SAC has been creating partnerships and pathways with regional hospitals to work out procedures like patient transfers and ensure continuity of care.
There are close relationships in the works with local hospitals, but SAC is also working with centres like Trillium Health Partners, in Mississauga, Ont., and St. Michael’s, a downtown Toronto acute-care hospital.
As Dr. Varlese noted, hospitals across the Greater Toronto Area are seeking solutions to the growing demand for services in a time of fiscal constraints.
“Our goal is to be a dedicated partner within the healthcare ecosystem, providing tangible solutions to the most pressing challenges facing our province.
“Ultimately, we are here to help Ontarians bypass long wait times and access the care they need to reclaim their mobility and quality of life.”
Contributing Editors
Dianne Daniel dianne.l.daniel@gmail.com Will Falk will@wfalk.ca
Address all correspondence to Canadian Healthcare Technology, P.O. Box 907, 183 Promenade Circle, Thornhill ON L4J 8G7 Canada. Telephone: (905) 709-2330. Internet: www.canhealth.com. E-mail: info2@canhealth.com. Canadian Healthcare Technology will publish eight issues in 2026. Feature schedule and advertising kits available upon request. Canadian Healthcare Technology is sent free of charge to physicians and managers in hospitals, clinics and nursing homes. All others: $67.80 per year ($60 + $7.80 HST). Registration number 899059430 RT. ©2026 by Canadian Healthcare Technology. The content of Canadian Healthcare Technology is subject to copyright. Reproduction in whole or in part without prior written permission is strictly prohibited. Send all requests for permission to Jerry Zeidenberg, Publisher. Publications Mail Agreement No. 40018238. Return undeliverable Canadian addresses to Canadian Healthcare Technology, P.O. Box 907, 183 Promenade Circle, Thornhill ON L4J 8G7. E-mail: jerryz@canhealth.com. ISSN 1486-7133
Neil Zeidenberg neilz@canhealth.com
Dr. Sunny Malhotra Twitter: @drsunnymalhotra
Norm Tollinsky tollinskyn@gmail.com
Art Director
Walter Caniparoli art@canhealth.com
Digital Media Specialist
Rebecca Downer rebecca@canhealth.com
Dr. Fabio Varlese
Our most energy-efficient 1.5T MRI with DryCool Technology
siemens-healthineers.ca/magnetom-flow

Ushering in a new era of sustainable and helium-independent MRI, MAGNETOM Flow.Ace (60cm bore) and MAGNETOM Flow.Elite (70cm bore) are now available in Canada.
They empower you across the entire imaging process from start to finish with unmatched workflow simplicity, elevated patient experience and pioneering AI-enhanced imaging for high-quality results at unrivaled speed.
• Virtually helium-free 1.5T MRI with DryCool technology and sealed for life magnet
• Our most energy-efficient 1.5T MRI, bringing 30-40% energy savings
• No quench-pipe and an ultra-compact footprint of only 25 m2
• Complete patient-centric workflow with BioMatrix Contour coils
• Smart and intelligent AI guidance with Deep Resolve and myExam Companion
Core exams
Growing imaging
Oncology
Rising cancer cases
Cardiology
Increasing indications for CMR
Want to learn more in-person?
Visit us at the Canadian Association of Radiologists (CAR) Annual Scientific Meeting April 16-19, 2026 Montreal, Quebec
Scan for more event information
Alberta’s approach to clinical AI: evaluate, test, partner and scale
BY DR. JEREMY THEAL
As the chief medical information officer at Health Shared Services (HSS), formerly part of Alberta Health Services (AHS), I represent over 10,000 physicians/prescribers that use our province-wide clinical information system called Connect Care.
The frequency and intensity of calls I hear to “give us AI tools now!” has been ever-increasing over the last few years, so I am intensely aware that my colleagues see AI as a crucial release valve for burnout, and a transformative new way to address the ever-growing volume of waiting patients who need our help.
I work as part of a large digital health team that serves over 130,000 Connect Care users across Alberta. With our provincial scope and standardized system, we have a unique opportunity to scale clinical AI solutions rapidly.
However, while we want to move quickly to give our clinical teams the AI tools they want, we recognize there are inherent risks. Herein lies the dynamic tension with AI – how do we move quickly, but still proactively mitigate the potential negative impacts at a provincial scale?
How do our governance, policy, procurement, and implementation processes keep up with the dizzying pace of AI development, in a rapidly shifting marketplace?
Strategic approach: We see AI as a key strategic enabler in our pursuit of the healthcare quintuple aim, but the benefits can only be realized safely if we first inventory and mitigate the inherent risks. To achieve this goal, we established an interprofessional AI Steering Committee which
developed a framework of AI opportunities and risk management strategies. Using this framework, we have been refining our strategic approach to AI, which includes the following principles:
•Solution selection based on clinical value: We prioritize AI solutions that are vetted by clinicians and staff as having high value to their work in healthcare (such as reducing administrative burden, improving efficiency, reducing cost, readmissions, length of stay and/or mortality). We assign higher value to tools that help the widest scope of users (across client organizations, scopes of practice, and clinical specialties) and those tools that integrate directly into core clinical system workflows. Success metrics are defined upfront, aiming to verify and quantify the value that drove prioritization.
•Hybrid approach to AI solutions: We employ a hybrid (“build/buy”) AI solution approach, meaning for each need we evaluate capital and operating costs, solution fit, sustainability, workflow integration, and clinical utility when deciding whether to use internal resources and infrastructure to build our own solution, or to procure an externally developed one.
•Human-first approach: We design for AI that is human-centered, meaning understandable technology that works as a co-pilot, not a replacement, for humans. In this approach, AI refers to “augmented intelligence”, rather than artificial intelligence.
•Proactively mitigate risks: Our review of AI-associated risks was conducted by an internal interprofessional team of digital health, clinical, risk management, legal, and privacy experts, with input from industry third parties. Categories of risk we recog-

nize and manage include quality/safety (AI accuracy, hallucinations, omissions, unintended consequences), ethics (bias, discrimination, fairness, surveillance), HR (talent, skillsets, job displacement), technical infrastructure, data security/sovereignty, legal and privacy (legislative requirements, contracting, liability, intellectual property), financial (funding, sustainability, cost of cyberattacks), and environmental (climate change). A key insight we have reached is that not every AI tool carries the same risks. By stratifying the level of risk among different proposed AI solutions, we can identify those that can proceed more quickly, provided appropriate monitoring is in place.
•“Driver’s License” for AI: Users are required to receive training before using AI tools. They must have a basic understanding of how AI works, know the importance of data privacy and patient consent, and be aware that they remain responsible and accountable for all aspects of patient care. Users are required to be the “human in the loop”, ensuring all content and proposed decisions are verified and free of inappropriate inclusions, exclusions, and bias. Simultaneously, our Digital Health leadership and team members are rapidly upskilling in AI to ensure we make optimal governance and development decisions as we work in partnership with our clinical teams.
•Accelerated cadence: To keep pace with

the swiftly evolving AI landscape, we are pivoting to a rapid cycle execution approach, starting small and aiming to reach testing in real clinical practice as soon as it is safe and feasible. We start with small, trusted pilot groups for evaluation, iteratively adjusting solutions based on feedback, and gradually expanding scope of implementation as value is defined and risks are mitigated.
•Learning Health System approach: We build evaluation into each of our implementation plans, feeding the results into an iterative process that informs continuous improvement. This includes continual refinement of not only our solutions, but also our processes and overall strategic approach.
Our progress so far: Our earliest AI work started in 2021 when our Intelligent Automation group began using technologies such as process automation to reduce staff burden from repetitive manual tasks. Over the past four years, this team has deployed over 50 solutions that have saved over 275 years of staff time, or over $18M in staff costs.
Examples include automation of repetitive HR and finance back-office processes (job offers, matching staff to postings, purchase orders) and IT processes (desktop software deployment, license optimization). Clinically facing solutions are also in development using AI (such as automated processing of faxed patient referrals).





In 2024 we partnered with physician innovators to begin rollout of an internally designed and built AI scribe product. Today, hundreds of physicians across multiple specialties have signed on to use the solution, with further expansion underway. Also in 2024, our Data and Analytics team strengthened our AI solution capacity by providing analysts with foundational AI models and tools embedded within Alberta’s enterprise data warehouse.
On the horizon for 2026 is a conversational analytics application, initially focused on Emergency Department records, that will enable business users to query data in plain language and receive contextaware insights and data visualization, significantly reducing traditional analytics turnaround times.
In late 2025, we began working with clinicians to select key in-system AI tools that are low effort/cost to build and have high value for improving clinical efficiency. These tools are being deployed to small pilot groups and by the time you read this, will include generative AI solutions that summarize the care of inpatients (for clinician handover, discharge summaries) as well as outpatients (for more efficient and complete clinic visits).
Another priority area in 2026 is partnerships. The best AI solutions are not developed in a vacuum, but are the product of collaboration between clinicians, innovators, data scientists, analysts, health organizations, universities, researchers, companies, and government. We are currently exploring a model that proposes to integrate these groups, aiming to enable healthcare teams to prioritize the highest value innovations for development with cross-sector expertise.
Dr. Jeremy Theal




Sault Area Hospital deploys innovative HIFU system for chronic pain
BY FELICIA CLEMENT
Sault Area Hospital (SAH) has become the first hospital to integrate the Neurolyser XR High-Intensity Focused Ultrasound (HIFU) system into a clinical setting, marking a significant milestone in chronic pain management and hospital-based innovation.
This initiative reflects SAH’s leadership in redesigning care for underserved populations and advancing clinical practice through technology.
The Neurolyser XR HIFU system utilizes focused ultrasound energy to thermally ablate targeted peripheral nerves responsible for transmitting pain signals. The procedure is performed under precise imaging guidance, combining X-ray and optical navigation to ensure accurate localization of the treatment site.
Ultrasound beams are converged through intact skin – similar to the focal point created by a magnifying glass – delivering therapeutic heat to the nerve tissue without the need for needles, incisions or radiation exposure.
One of the most compelling clinical benefits is the rapid onset of pain relief, with some patients reporting significant improvement within 30 minutes posttreatment.
Additionally, the short recovery time allows most individuals to resume daily activities the same day.
The Neurolyser XR HIFU system also facilitates timely evaluation of treatment efficacy; if no relief is observed within seven days, clinicians can promptly adjust the pain management strategy.
This technology also improves safety by reducing radiation exposure during treat-
ment, making it a more accessible and effective option for a wider range of patients.
SAH’s adoption of the Neurolyser XR HIFU system underscores its commitment to expanding access to advanced care and driving innovation in hospital-based pain management.
This transformative technology was championed by locum physicians Dr. Kevin Smith and Dr. Michael Gofeld, whose clinical experience with the Neurolyser XR HIFU system in private practice since January 2025 demonstrated its effectiveness.
The method was introduced in Canada by Dr. Smith and Dr. Gofeld, who conducted pivotal studies that led to Health Canada’s approval of the treatment.
Their research included both preclinical and clinical trials, which confirmed the superb safety profile of the Neurolyser XR
HIFU system and demonstrated its superior effectiveness compared to traditional radiofrequency ablation (RFA).
Their advocacy through SAH’s Physician Recruitment and Retention program was instrumental in acquiring the system, with additional support from the Sault Area Hospital Foundation, making it possible to integrate this technology into a hospital setting for the first time worldwide.
“This is a proud moment – being able to offer a novel, non-invasive treatment to a greatly underserviced community is deeply meaningful,” said Dr. Smith. “In my line of work, treatment typically involves needles and invasive procedures that require healing and recovery. Now, with the Neurolyser XR HIFU system, we’re able to achieve the same, if not better, patient out-
comes without those burdens. This is a transformative step forward, setting a new standard for how we treat chronic pain conditions in a hospital setting.”
For his part, Dr. Gofeld said: “Many of the patients we see with chronic spinal arthritis are older adults who face significant limitations in their daily lives due to persistent pain. This new treatment option is non-invasive, offers extended relief, and helps patients return to a more active lifestyle without the burden of long recovery times. It’s a meaningful advancement in how we support comfort and mobility for this population.”
The integration of the Neurolyser XR HIFU system into the SAH Pain Clinic is more than a technological milestone, it is a reflection of Sault Area Hospital’s unwavering commitment to its mission of exceptional people working together to provide outstanding care in Algoma.
By pioneering this hospital-based application of a transformative pain management technology, SAH continues to fulfill its vision of being a trusted partner in delivering outstanding care. This advancement not only elevates the standard of treatment for chronic pain but also reinforces the hospital’s dedication to innovation, accessibility, and improving the quality of life for the communities it serves.
This initiative was made possible through the generous support of the Sault Area Hospital Foundation, whose dedication to funding transformative medical technologies continues to enhance patient care and outcomes in the Algoma region.
Felicia Clement is Communications and Digital Services Coordinator at the Sault Area Hospital.
Building a “Front Door” for personalized disease prevention in Canada
BY DR. ABBAS ZAVAR, MD, MPH, MHI (PRINCIPAL INVESTIGATOR)
AND DR. ROYA FARZANGEAN, MD, PHD (CO-INVESTIGATOR)
Canada’s healthcare system is primarily designed to treat illnesses, but it faces challenges in preventing them at an early and personal level. While preventive programs are available at federal, provincial, and community levels, they often rely on broad, population-based recommendations that do not adequately consider individual risk profiles, personal circumstances, and preferences.
As a result, the prevention model is challenging to implement proactively and in a patient-centred manner, especially in primary care, where prevention efforts must contend with time constraints and the complexities of managing multiple health conditions.
This is also where the “AI moment” in healthcare becomes significant. Advanced analytics and AI can only support personalized prevention if they have access to standardized, interoperable, patient-facing data.
However, Canada still faces ongoing
barriers to collecting that data at the point of care – fragmented information across jurisdictions, limited interoperability even where EMRs are common, and minimal integration of patient-reported and patient-generated health information into routine clinical workflows.
When risk information is incomplete or inconsistently captured, prevention shifts to a reactive approach: triggered by symptoms or late-stage clinical presentations, rather than through the systematic identification of modifiable risks and early intervention.
Why “personalized prevention” needs a practical entry point: Personalized Medicine (PM) offers a compelling framework for modernizing prevention. Drawing on established definitions from authoritative organizations and scientific literature, we define PM as “a medical model that characterizes individuals’ phenotypes and genotypes (integrating health information, molecular profiling, environmental exposures, lifestyle factors, and social determinants) to deliver the right clinical strategy (prevention, diagnosis, or treatment) to the right person at the right time.”
This prevention-focused approach is
essential: preventive measures should begin before disease onset, with risk stratification and early detection guided by multiple health determinants.
But to operationalize personalized prevention, primary care needs a reliable “front door”: a standardized intake approach that can efficiently, consistently, and equitably identify key risks across


diverse populations, and produce outputs that lead to action.
The Persoventa Project – a fourphase roadmap: The Personalized Preventive Plan (Persoventa) Project was designed to address this gap with a structured, evidence-based approach. Persoventa is a four-phase initiative that
moves from identifying validated assessment tools to building an interoperable knowledge base linking risk factors to diseases across 15 preventive domains.
Phase 1 concentrates on identifying reliable, preferably Canadian-validated, self-administered assessment tools across different domains. Selected instruments will be broken down into individual metrics, which will then be mapped to relevant risk factors and potential conditions (Phase 2).
The objective is to validate risk factors against Canadian clinical guidelines for the top 30 preventable diseases, ensuring consistency in risk factor alignment across various domains. (Phase 3).
Finally, Phase 4 will develop a unified, queryable knowledge base of risk factors and diseases using interoperable standards to support future analytics and AIdriven prevention efforts.
This structure is purposeful, creating a credible pathway from existing evidence to real-world implementation while establishing the data foundation required for connected care and future AI-enabled decision support.
What we did in Phase 1: Phase 1,
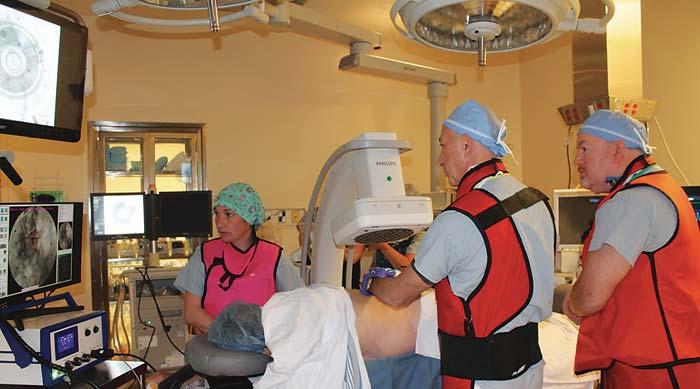
Dr. Michael Gofeld (centre) and Dr. Kevin Smith (far right) perform one of the first Neurolyser XR High-Intensity Focused Ultrasound (HIFU) treatments at Sault Area Hospital, assisted by nursing staff. This milestone procedure marks an innovative step in delivering non-invasive pain management care to patients.
Dr. Roya Farzangean
Dr. Abbas Zavar

Why patient flow is a patient safety problem, not just an operational one
And why interoperability is the foundation Canada needs to get right.
Across Canada, patient flow is commonly framed as an operational challenge: bed capacity, emergency department overcrowding, alternate level of care patients occupying acute beds and systemwide pressure. These measures matter, but they are symptoms, not causes, and they only tell part of the story.
Patient flow is a patient safety issue. And increasingly, it is a clinical leadership issue, one that clinical digital leaders must actively own.
The scale of the problem demands it. According to CIHI, in 2024–25, more than 16.1 million unscheduled emergency department visits were recorded across Canada. Half of all patients admitted from an ED spent more than 16 hours waiting, one in 10 spent more than 48 hours.
Nationally, nearly one in six hospital days is consumed by alternate level of care patients who no longer require acute care but have nowhere else to go – in Ontario alone, this equates to more than 4,000 beds occupied every day. Meanwhile, 62 percent of Canadian ED directors describe overcrowding as a major or severe problem.
When flow breaks down, harm can follow. Access block – the inability to transfer admitted patients to an appropriate inpatient bed – can force hospitals to place patients on clinically inappropriate wards where there is a bed is available.
In a recent study in the UK, off-service patients are reported to have greater than twice the length of stay compared to patients managed on their home ward. Prolonged stays can expose patients to deconditioning, infection and medication-related harm and further compound hospital capacity issues.
Fragmented transitions can create gaps in oversight and missed opportunities for early intervention. These are not abstract consequences; they are daily realities in every Canadian province and territory.
We recently published Flow: A Handbook to Creating the Right Conditions for Patient Flow, in which we observe that improving patient flow requires a combination of technology, process improvement and cultural change. Drawing on that framework, this article explores seven levers that can drive meaningful improvement: achieving interoperability within and across care settings; building system-wide visibility of patients and resources; digitising clinical workflows and decision support; reducing unwarranted clinical variation; enabling new models of care; reducing administrative burden; and embedding clinical digital leadership to make changes enduring.
Interoperability – the foundation: With more than 10 different EMR systems in use in Ontario alone and limited interoperability between provincial and territory systems, clinical information frequently fails to follow the patient. But fragmentation also occurs within hospitals, with clinical data often siloed across laboratory, radiology, pharmacy and specialty applications that were never designed to share information in real time.
Canada has begun to recognise this must change. Canada Health Infoway estimates that improved interoperability could save $2.4 billion annually.
The Connected Care for Canadians Act (Bill C72), introduced in June 2024, and recently re-introduced in February of 2026 as Bill S-5, signalled clear legislative intent to require system interoperability and prohibit data blocking.
The Pan-Canadian Interoperability Roadmap, CA Core+ FHIR standard and PS-CA patient summary specification all confirm the direction of travel.
All healthcare leaders need to navigate multi-vendor environments. The practical question is how interoperability layers complement rather than compete with existing investments. The answer lies in platform architectures that consolidate data from disparate systems into a coherent, longitudinal view using open FHIR APIs, without requiring wholesale system replacement.
Visibility, workflows and variation: Interoperability creates the conditions for visibility, but visibility must extend beyond the patient record. Effective flow management requires real-time insight into two dimensions: the trajectory of every patient across their journey, and the availability of critical healthcare resources – most importantly staffed beds but also knowing other flow constraints such as imaging and pathology capacity.
When both views are connected, clinical and operational leaders can move from reactive to proactive
These models shift the definition of flow. It is no longer solely about moving patients through beds; it is about maintaining clinical oversight across settings. If patients become digitally invisible once they leave the hospital, if documentation is incomplete or data does not follow them, early deterioration signals may be missed.
Without interoperability and visibility of the longitudinal record across the continuum of care, virtual care inherits the same fragmentation risks experienced in inpatient settings, potentially with greater safety consequences.
From operational metric to safety discipline: The missing lever in many digital strategies is clinical leadership itself. Patient flow remains trapped in the language of bed management and operational metrics. In reality, it is a clinical safety function, one that demands the same rigour in documentation standards, data visibility and system design as any other patient safety domain.
For healthcare leaders, this represents both an opportunity and an obligation. The clinician who un-
capacity management across facilities and regions.
That visibility must be enabled and the value realised through digitised clinical workflows at the point of care. The goal is not simply to store information electronically, but to surface the right information to the right clinician at the right time. This includes structured documentation aligned to agreed clinical pathways, standardised assessments that surface risk early and consistent definitions of escalation and discharge readiness.
These same digitised workflows establish the data foundation for responsible AI. As the CMAJ has noted, a lack of reliable data is currently a bottleneck in developing AI technologies for healthcare. Without a standardised, interoperable data layer, AI risks amplifying the very fragmentation it is intended to resolve.
New models of care change the equation: Nowhere is the convergence of flow and safety more apparent than in virtual care. Globally remote patient monitoring, hospital in the home programs and virtual EDs are changing how care is delivered. The Ontario government continues to expand support for the HITH program, and programs in British Columbia and Quebec have reported positive engagement from patients.
derstands alert fatigue, who has navigated the politics of documentation standardisation and who can bridge the gap between clinical governance and IT architecture is precisely the leader patient flow demands.
Canada is at a defining moment. The interoperability standards being developed, the legislative direction of Bill S-5 and the growing adoption of virtual care all point towards a more connected health system. But connectivity without clinical purpose is not sufficient. Interoperability must be accompanied by platforms that translate connected data into safer clinical workflows, more reliable patient journeys and the conditions for AI to augment decision-making responsibly.
Patient flow, once measured in beds and wait times, is increasingly best understood as the ability to maintain safe, continuous oversight across the entire care pathway. When designed as a safety function, digital investment stops being about technology and starts being about outcomes.
To learn how Alcidion’s Miya Precision platform can support safer patient flow across your health system, connect with our team at Digital Health Canada ON26 on 19 March 2026, or reach out at info@alcidion.com.






























Data is infrastructure: The missing foundation of Canada’s healthcare
BY KAREN GAUTHIER
fter years of working with healthcare organizations across Canada and the U.S., I’ve seen digital transformation accelerate through rapid growth in virtual care, rising AI adop-
tion, and a steady stream of new platforms.
Yet despite all this momentum, one thing has remained stubbornly consistent: the experience of fragmentation. Patients still move through the system without their information following them. Clinicians still work without a full picture of the indi-
vidual in front of them. And organizations still struggle to connect systems that were never designed to speak to one another.
Through these experiences, I have come to believe that the real opportunity for change begins with a shift in perspective.
Data is not just the byproduct of adopting
Hypercare helps hospitals coordinate care, escalate faster, and reduce risk with one unified system.


the newest technologies. It is the foundation on which a modern, connected, patientcentered healthcare system must be built.
When data is standardized, governed, and connected, it becomes powerful. When it is not, even the most advanced technologies struggle to deliver meaningful results.
As I’ve worked with teams navigating these challenges, a clear structure has consistently emerged for building a connected ecosystem. It always begins with secure, standards-based access to data.
• Organizations need reliable ways to retrieve and share clinical and administrative information across different systems.
• Once that access is in place, the next step is building data networks, the “roads” that allow data to flow between regions, providers, and digital services.
• Finally, meaningful transformation requires data aggregation: normalizing and integrating information from multiple sources so that insights can fuel analytics, AI, and population-level intelligence.
These three layers of access, networks, and aggregation form a model I’ve seen work repeatedly. They turn fragmented inputs into shared intelligence.
A challenge that I often see is the number of organizations that still rely on older systems that were never built with interoperability in mind.




This is where organizations like ELLKAY have played an important role. ELLKAY’s technology acts as a kind of “data adapter,” transforming non-standard information into consistent, standards-based formats so that even the most outdated systems can participate in regional or national data exchange initiatives. This ensures no organization is left behind simply because of the age or limitations of their software.
When I see organizations treat their data as infrastructure by embracing standards, improving governance, and investing in connectivity, the impacts are significant.
Interoperability improves, data becomes cleaner, and redundancy drops. Clinicians gain clearer insights. Patients gain greater transparency, easier access to their information, and more trust in how their data is being managed. Leadership teams finally have the clarity they need to make decisions driven by reliable intelligence rather than fragmented snapshots.
Canada is at a moment of opportunity. The technology exists. The expertise exists. The demand from clinicians and patients exists. What is needed now is alignment: national data standards, interoperable networks, and tools that support organizations no matter where they are in their digital journey.
I’ve learned that healthcare transformation will never come from individual technologies alone. It comes from shared commitments, standards, infrastructure, and a shared understanding that data is one of our most powerful assets.
Karen Gauthier is VP, International at ELLKAY.
Karen Gauthier
Canadian Healthcare Technology magazine
Canadian Healthcare Technology offers the country's leading healthcare I.T. publications. And they're free to healthcare providers. Get your complimentary subscriptions now!
Canadian Healthcare Technology breaks the news about important projects, programs and technologies, and provides hospital executives and senior managers with an excellent source of information for improving the delivery of healthcare. It’s sent to over 3,900 readers in print format and to over 7,300 opt-in subscribers of the digital edition.
White Papers
eMessenger newsletter
Canadian Healthcare Technology’s e-Messenger contains breaking news about important deals, installations and developments. Four blasts are sent each month, via e-mail, to over 7,100 senior managers and executives in hospitals, clinics and health regions.
Canadian Healthcare Technology’s White Papers are sent out once a month, via e-mail, to over 5,500 senior managers and executives in Canadian hospitals and health regions. The monthly blast contains summaries and links to White Papers issued by various organizations, providing cutting-edge information about topics of interest to healthcare decision-makers.


For advertising or editorial inquiries, contact Jerry Zeidenberg, Publisher, jerryz@canhealth.com
Sex differences could play key role in diagnosing diabetic retinopathy
VANCOUVER – Sex differences could play a key role in diagnosing diabetic retinopathy, according to the results of a groundbreaking study led by Vancouver Coastal Health Research Institute researcher Dr. Ipek Oruc. A condition that puts patients at risk of blindness, diabetic retinopathy may be more likely to progress along a given pathway depending on whether a patient was born male or female.
Diabetic retinopathy is the most common cause of blindness associated with diabetes, affecting around one million Canadians. The condition is characterized by changes to tiny blood vessels at the back of the eye.
In the early stages, symptoms of the disease may go unnoticed, such as with mild to moderate non-proliferative diabetic retinopathy. However, once the condition advances, various vision-threatening complications may arise.
Leakage from damaged blood vessels can lead to eye swelling – called macular edema – which causes vision loss primarily affecting central vision.
“Regular monitoring is crucial for detecting early signs of diabetic retinopathy and to prevent its progression to moderate or severe disease,” said Oruc. “However, aside from considerations during pregnancy – when females are at higher risk for progression – sex is not presently factored into disease diagnosis, management or treatment.”
Researchers trained, validated and tested an artificial intelligence (AI) algo-
rithm on a data set of 2,967 fundus images from 1,491 female and 1,476 male patients with diabetic retinopathy. The fundus is the inner area at the back of the eye comprising the retina, optic disc, fovea, macula and blood vessels.
The algorithm was trained to classify patient sex from retinal images of people with diabetic retinopathy. Male and female groups were matched for age, ethnicity, severity of diabetic retinopathy and hemoglobin A1c levels – a measure of blood sugar in the body – to prevent the model from using these potentially confounding variables when classifying a patient’s sex.
Oruc and her team used convolutional neural networks (CNNs) – AI algorithms specializing in image classification – to identify patterns of diabetic retinopathy presentation in the images. This approach
builds on the team’s prior research on the classification of retinas by sex.
One of the research team’s central objectives was to find an empirical method to identify potential differences in how diabetic retinopathy manifests in females and males.
To see whether there were sex-specific disease characteristics of diabetic retinopathy, Oruc and her team used Guided GradCAM saliency maps – an explainable-AI technique that can peer into the often mysterious ‘black box’ of CNNs. While CNNs can identify unique features in a series of images, the explanatory AI built into Guided Grad-CAM saliency maps helps researchers understand how the CNN arrived at its conclusions.
“For example, if we were to use explainable AI to identify what distinguishes a celebrity from other people, it may high-
light the celebrity’s nose and mouth as important features that set that celebrity apart,” Oruc said.
The Guided Grad-CAM saliency maps showed that the CNN focused on the macula in female fundus images and the optic disc and peripheral branching vasculature from the optic nerve in male fundus images.
“This pattern differed noticeably from the saliency maps generated by CNNs trained on healthy eyes, which did not highlight these particular regions, indicating that diabetic retinopathy may manifest differently by sex,” said Oruc.
“However, further research is needed to validate this hypothesis,” she adds, which is why her team is presently conducting a follow-up study.
The research team’s application of CNNs – interpreted using Guided Grad-CAM –was able to achieve a high level of accuracy using a smaller dataset for training the algorithm than is typical, Oruc noted. “This is particularly important, as large datasets are more costly and time-consuming.”
“The success of this approach with a small dataset puts studies like ours within reach of other research teams with limited resources.”
Dr. Ipek Oruc is the director of the NOVA Lab and a researcher with the Djavad Mowafaghian Centre for Brain Health. She is also an investigator with the Data Science Institute, an associate member of the School of Biomedical Engineering and an associate professor in the Department of Ophthalmology and Visual Sciences at the University of British Columbia.
HEALWELL AI reports on integrating companies and technologies
TORONTO – HEALWELL AI
Inc., a healthcare AI company focused on preventative care, says it has made significant progress integrating its AI capabilities and software platforms following the acquisition of several companies.
In recent years, HEALWELL AI has acquired an ecosystem of AI and electronic health record companies, including Khure Health, Pentavere, VersoSource, Intrahealth and Orion Health.
HEALWELL said it has successfully integrated its Khure and Pentavere AI capabilities into a unified AI engine powered by DARWEN, while also integrating its clinical operations.
Building on this progress, HEALWELL has expanded its offerings across the joint customer base through coordinated Orion-Verosource commercial initiatives.
“The integration of our AI platforms has been well received by customers and has translated directly into new opportunities for expanded life science partner agreements,” said James Lee, chief executive officer of HEALWELL AI. “This validates our focused strategy of building cohesive, enterprise-grade AI solutions that deliver real clinical and economic value.”
HEALWELL continues to embed AI capabilities directly into its software
platforms, creating differentiated, workflow-native solutions for healthcare customers. Amadeus AI represents the company’s next-generation AI-enabled digital care record, with active commercial opportunities across Canada, the United States, Europe, and the Middle East.
The integration of HEALWELL’s AI capabilities into Orion Health’s infrastructure is showing early commercial traction, the company said.
These offerings combine Orion Health’s more than 30-year track record in healthcare IT with HEALWELL’s clinically validated AI and, coupled with Verosource’s data science and analytics capabilities, deliver a broader AI and data science portfolio that is resonating well across Orion Health’s customer base.
HEALWELL’s award-winning DARWEN AI platform (a Prix Galien award recipient) continues to expand its disease-state coverage and life sciences customer base, with recent advancements in AI-driven label expansion and patient identification capabilities.
The company is also strengthening its validation and credibility in applied healthcare AI through a growing body of scientific publications, clinical collaborations, and expansion into additional disease areas, including complex and neurodegenerative conditions.
In the fourth quarter of 2025, HEALWELL had four publications accepted: one in The Journal of the Canadian Association of Gastroenterology (JCAG), two presented at United European Gastroenterology (UEG), and one presented at the European Crohn’s and Colitis Organisation (ECCO).
“Healthcare IT adoption requires trust, validation, and security, particularly in enterprise and public sector environments,” said Dr. Alexander Dobranowski, president of HEALWELL. “By
“Unlike many industries, we see AI as an opportunity, not a threat,” said HEALWELL’s CEO, James Lee.
embedding proven AI into established platforms with long-standing customer relationships, we believe we are uniquely positioned to drive adoption at scale.”
Following its refocused strategy, HEALWELL said it continues to execute against key integration milestones and unlock synergies to support improved profitability and economies of scale.
Building on the divestments announced on November 3, 2025, HEALWELL is also evaluating opportunities
for portfolio simplification.
As part of this ongoing review, the company is assessing its portfolio of strategic investments, which include a US$2 million investment in a dedicated investment vehicle that holds an interest in xAI, as previously announced on June 26, 2024, now part of SpaceX.
HEALWELL believes its progress on integration, expanding commercial traction, and alignment with structural healthcare tailwinds position the company well to execute on its long-term strategy to become a global AI and data science partner to life sciences companies, health systems, and public sector organizations.
CEO James Lee commented, “Our strategy is clear: we are building a global carriage network, to enable the adoption of AI assisted preventive care through early disease detection. Unlike many industries, we see AI as an opportunity, not a threat. Our long-standing customers and deep library of peer reviewed publications – now numbering over 50 – are unique, in our opinion.”
He added, “Everything about HEALWELL, including our M&A strategy, is deliberately focused on expanding our carriage network, and our capabilities, while keeping the patients and clinician’s outcome at the very core of what we do.”
AI agents interact with radiologists, technologists and referring physicians
BY JERRY ZEIDENBERG
CHICAGO – In a demonstration at the RSNA conference last December, Dr. Luciano Prevedello, a radiologist at Ohio State University’s Wexner Center of Medicine, chatted with his AI-enabled computer about a patient’s brain lesion as he created a report. He showed how he could discuss the case with an AI agent, much like he would with a colleague.
Not only did the AI agent offer expert opinions on the lesion’s type and location, but it could also bring up different patient exams on the computer screen, including CTs and MRIs that had been taken, with a variety of views.
That ‘natural language’ discussion was an example of how reading and reporting on radiological studies will change in the not-so-distant future. It was part of an exciting panel presentation at RSNA on ‘The Reading Room of the Future’, most of which focused on the role AI will play.
Dr. Prevedello discussed how AI agents are being deployed in the DI department of the hospital in a variety of ways – although much of the work is still in the early stages.
“We’re starting to see agentic AI introduced in multiple areas – ordering, protocols, acquisition, worklists and reporting,” he said.
Dr. Prevedello is also the vice-chair for medical informatics and augmented intelligence in imaging, division chief of medical imaging informatics and medical director of the 3D and Advanced Visualization Lab. He spoke remotely, by video call, as he was prevented from attending in person by the snowstorm that closed Chicago’s airports just before the conference.
Dr. Prevedello said that on the ordering side, AI agents are already able to autonomously schedule exams, calling patients and managing inbound and outbound phone traffic.
And when it comes to protocols, “they can select the most appropriate protocols, based on the free-text indications.”
“We’ve been working on this at Ohio State for a while now, experimenting with large language models to understand the indications, to guide ordering physicians and to help with protocol selection.”
On the acquisition side, he said that AI has been used to help position patients and to reduce anxiety. And for worklist and routing, “AI agents have been able to distribute the exams, based on complexity and sub-specialty, and to prioritize urgent cases,” he said.
But the most astounding changes are appearing on the reading and reporting side. Dr. Prevedello demonstrated work under development at Ohio State University’s medical center, where AI agents are analyzing entire imaging studies and providing full reports with diagnoses.
In an impressive demonstration, Dr. Prevedello discussed a patient’s images, in natural language English, with an AI agent.
The agent stated in a clear voice, “Okay, I see a screen with a presentation. It mentions progressive visual disturbance with bitemporal hemianopsia. And it shows it’s a 35-year-old female.”
The agent then asks, “Is there anything else you would like to discuss?”
Dr. Prevedello said, “What do you think about this history? The patient’s history is concerning.”
The agent responded, “Progressive visual disturbance with bitemporal hemianopsia suggests a lesion at the optic chiasma. Is there anything else you would like to discuss?”
“Yes, I would like to see the images. What do you see?”
“Yes, I would like to see the image of the bone window, okay?”
The agent and the doctor go on to look at
The AI agent brings up images and says, “I see a CT scan of the brain. It’s an axial view and I see a possible mass in the region of the sella. Would you like to see anything else.”

Governing generative AI: From permission to competency to measurement
AI is evolving. Neither drug nor device frameworks are an easy match for generative systems.
BY WILL FALK (W AI ASSISTANCE)
This article is condensed from a longer paper written by Will Falk, to be published by the CSA Public Policy Centre and available on their website. Will is a Fellow at four Canadian Think Tanks and Universities and a contributing editor to CHT.
Policy and regulation for generative AI (GenAI) in healthcare remain unsettled in Canada and internationally. What policies and practices have emerged are the product of a period of managed experimentation, shaped less by formal approval pathways than by professional accountability, institutional oversight, and pragmatic restraint. In Canada, this has produced a system that is permissive in practice but cautious in posture: clinicians are expected to supervise GenAI within scope of practice rules, organizations remain accountable for deployment, and patients experimenting with AI are expected to act responsibly.
Some have criticized Canadian governments for moving slowly. In early 2026, that criticism looks increasingly unfair as the UK, US, and Europe have each retrenched in early 2026 or late 2025 while Canada has used safe harbour rules and vendor approvals to allow adoption within physicians’ scope of practice.
Rather than forcing the use of legacy regulatory categories, Health Canada has taken a deliberately incremental approach, issuing principles and guidance while observing how GenAI is actually used in clinical settings. This was in part due to the change of government and the appointment of Evan Solomon as the first AI Minister; this has caused a policy pause.
Health Canada’s AI4Health document in 2024 signalled that AI adoption could proceed provided safety, accountability, and trust were maintained. In February 2025, updated pre-market guidance for machine-learning-enabled medical devices clarified expectations around evidence, lifecycle management, and predetermined change control plans.
These steps were modest by design and remain anchored in Canada’s existing Software as a Medical Device (SaMD)framework, introduced in 2018 for a different generation of deterministic and narrowly scoped software. That framework was never designed for large, probabilistic, continuously evolving systems embedded directly into clinical workflows. Health Canada has not attempted to retrofit it aggressively. Instead, it has left room to learn.
That restraint now appears justified. Regulators in the US, UK, and Europe each initially signalled that GenAI would be regulated through existing device pathways or even more aggressively – with some European jurisdictions forcing registration as at least level 1 medical devices. Each has since narrowed or softened those ambitions. Many have moved to selfcertification, which is largely performative. None has produced a stable, widely accepted framework. There are few global best practices to import.
Complement or substitute: The most important regulatory distinction is not whether a system uses generative AI, but whether it substitutes for clinical judgment. Is it a Complement or a Substitute.
Complementary GenAI systems support licensed professionals and operate within existing scopes of practice. Ambient scribes that draft clinical notes, second-screen clinical decision support (CDS) tools,
intake and referral systems, administrative automation, and patient-facing information tools all fall into this category. Accountability remains human. These systems have scaled rapidly because they fit within established licensure, liability, privacy and insurance structures. They reduce documentation burden, lower cognitive load, and improve workflow efficiency without displacing professional responsibility.
Substitutive GenAI systems are fundamentally different. They aim to initiate diagnoses or treatments, replace professional judgment, or act independently. These systems carry materially higher risk and require materially higher evidence and governance thresholds. Outside of medical imaging and a small number of tightly supervised pilots, substitutive generative AI remains largely in research environments in Canada.
tools are less safe, less useful and not limited to clinicians through a registration process.
The Competency-based Option: A competencybased framework offers a more coherent alternative. Healthcare already regulates human intelligence through scopes of practice, supervision, and progressive responsibility. Medical students, residents, and fellows are not approved once and left alone. They are hired, supervised, evaluated, audited, retrained, re-credentialed and fired or retired. Competence is maintained across the life cycle of the intelligence’s employment.
A competency-based model would apply this logic to AI systems. What is regulated is the class of capability exercised in a defined clinical context. Complementary systems operate under supervision. Near substitutive systems face progressively higher bars as autonomy increases. Interpretability is not required at every level, just as it is not required for trainees practicing under supervision. What matters is demonstrated performance, safety, and accountability.

Will Falk
Regulatory triggers should attach to substitution of clinical judgment, not to the mere presence of AI. Treating supervised ambient clinical copilot systems as if they were autonomous actors would unnecessarily slow the deployment of tools already delivering benefit. A broad class of complementary systems can continue to operate without formal premarket approval, provided accountability is clear, supervision is explicit, and harm thresholds remain low. Ambient scribes already reached 28 percent penetration by August 2025 according to a recent study by the CMA-CFIB. Safeharbour approaches developed for ambient scribes should be expanded and replicated, not treated as exceptional.
Why legacy approval frameworks fail in practice: Attempts to force generative AI into drug or medical device frameworks show that a new approach is needed.
•Drug approval pathways tolerate black boxes when safety and efficacy can be demonstrated, but drugs are approved as fixed interventions. Generative AI is adopted through learning, iteration, and realworld refinement. Requiring randomized controlled trials as a gating mechanism for deployment would delay adoption for years and suppress learning. Randomized trials will and should play a role as systems mature, particularly through academic centres, but they are learning studies, not approval studies.
•Medical device regulation poses a different mismatch. Even SaMD approvals assume fixed functionality, bounded performance, and predictable change cycles. Predetermined change control plans were a reasonable adaptation for earlier machine-learning tools. They are a poor fit for generative AI systems that are probabilistic, adaptive, and deeply embedded in workflows. Full interpretability may not be for some time. Transparency matters but insisting on devicelevel approval as the default either blocks useful tools or drives clinicians toward ungoverned alternatives. Jurisdictions that tightly constrained complementary tools have not eliminated AI use. They have displaced it into informal and unmeasured channels. The UK Royal College of Physicians report (late 2025) makes this clear. The lack of second screen CDS tools (like Doximity or OpenEvidence) has led to more grey usage of ChatGPT and other foundation models. This is very undesirable because the
This shifts the regulatory centre of gravity away from pre-market approval and toward continuous measurement.
Early debates about generative AI focused on risks such as hallucinations, bias, and instability. Early measurement relied heavily on standardized exams, most notably U.S. medical licensing tests. Those benchmarks were quickly exhausted. By mid-2025, frontier models were scoring above 90 percent. At that point, the metric ceased to be informative. But getting a 100 percent on the US MLE doesn’t make an AI a doctor any more than a calculator is a math teacher.
More recent evaluation frameworks represent a meaningful shift. Studies such as MAI-Dx by Microsoft’s Cambridge lab and the NOHARM work from Stanford-Harvard group moved beyond factual recall to test diagnostic reasoning, error patterns, and comparative performance against human clinicians in controlled settings. These studies consistently show that single-agent foundation models now perform roughly at general practitioner (GP) level on diagnostic tasks constructed by specialist panels. This group also published the excellent ARISE Report on AI in Clinical Practice at the start of 2026. New frameworks are being proposed that go beyond simple recall including MAST and MedHELM.
A practical framework should track:
•Transparency around model provenance and change history
•Error rates, override frequency, and escalation behaviour
•Bias and equity performance across populations
•Guardrails that constrain unsafe outputs
•Orchestration effects from multi-model and human-AI teaming
These are measurable properties. They can be audited. They align naturally with a competency-based approach. Under this model, regulation includes continuing medical education as well as licensure exams. They may not be familiar yet to digital health experts, but they will be! Model oversight, managing agentic resources, and local retraining will be much of the work of the future. This is exciting stuff and worth spending time understanding.
Canada’s current policies have allowed this logic to emerge. Complementary tools have scaled within existing accountability structures that addressed privacy,
Synthetic data breaks biggest bottleneck: Access to high-quality data
BY ANDREW FRANCES
The coronavirus pandemic, first detected in 2019, was a worldwide wake-up call to epidemiologists and clinicians. In Canada alone, more than 60,000 people had died from COVID-19-related illnesses by September 2024.
Seniors were particularly vulnerable; so were those with chronic lung or heart conditions and the immunocompromised. Specific jobs and circumstances that created certain patterns of interaction with the population at large also increased the risk of exposure.
In COVID’s wake, Canadian researchers prepared for the next pandemic by creating “synthetic populations” at the health region level – the level at which most public health decisions are made – using open data stores.
“The idea was to create a synthetic ‘Canadian world’ that reflects real population structure,” said Dr. Khaled El Emam, a professor of epidemiology and public health at the University of Ottawa and di-


rector of the Ottawa Medical AI Research Institute (OMARI).
These synthetic Canadians were assigned medical characteristics, daily routines, locations and other attributes. The model allowed researchers to simulate pandemic preparedness scenarios without using personally identifiable data.
“It’s a good illustration of how synthetic data can support large-scale public health modeling and planning, while avoiding many of the access barriers and privacy risks associated with using real health records,” Dr. El Emam said.
What is synthetic data? Synthetic data –data sets generated to mimic the properties, patterns and structures of real-world counterparts – are becoming increasingly valuable for healthcare settings. That’s because sometimes, doctors and data scientists can’t use actual datasets due to privacy regulations, or the research cohort is too small to be usable.
“In healthcare, it’s very common to hire data scientists and developers to innovate, only for them to spend months waiting for data access,” said Dr. El Emam. “Synthetic data can often be made available much more quickly, which lets teams build and test pipelines, develop and validate models, and explore data early.”
Synthetic data is not simply “made up”; it’s generated by training on original datasets to create datasets that mimic their behaviour. Those datasets could be from clinical trials, health system data from routine care, billing, payment and claims systems, research data held by institutions or
governments, such as the open data used to generate the “synthetic Canadian world”, and more.
By contrast, simulated data is created using human-specified statistical attributes, said Lorne Rothman, principal data scientist and healthcare lead for SAS
Canada, the Canadian arm of data and AI leader SAS. “A researcher might write a program to generate data that contains particular patterns, specific averages, spreads, and distributions of measures.”
To create synthetic data, machine learning algorithms analyze samples of realworld data, producing new data that mirrors patterns in the original data set.
“The most useful synthetic data typically comes from high-quality real data with strong statistical fidelity, diversity, longitudinal depth and balanced representation of

Lorne Rothman
Dr. Margarita Mersiyanova
Leading-edge hospitals in Canada creating AI-driven apps to solve real-world problems
Using AI, medical centres are taking on more ambitious problem-solving.
BY JERRY ZEIDENBERG
TORONTO – Numerous Canadian hospitals are testing the waters when it comes to AI, carefully establishing pilot projects and evaluating the technology. However, things are still in the early stages, and for the most part, hospitals have shied away from using AI to make clinical decisions about patient care. Many are playing it safe by sticking to AI for administrative purposes – like automating notetaking via AI scribes.
“I can say there has been almost no penetration of AI into the clinical environment to make patientcare decisions,” asserted Dr. Fahad Razak, an internist at St Michael’s Hospital and co- founder of GEMINI, the largest hospital research and quality improvement network in Canada.
“For the vast majority of physicians in Canada, the United States, and Europe, we have yet to use an algorithm to make a decision about a patient outcome, a therapeutic decision, or a prognostic decision that’s been informed by AI,” he added.
He said radiology departments may be the exception, where AI analysis has sped ahead – but radiologists have traditionally been the early adopters of advanced technologies.
Dr. Razak was part of an AI panel discussion at the HIMSS Eastern Canada conference, held last October.
At the same time, he said AI for patient care is definitely coming. “We know things are going to change,” he said, just as MRIs were once a technology with uncertain value among doctors, but are now the gold standard for many types of exams.
In the meantime, said Dr. Razak, there are many hurdles to jump as AI solutions emerge:
•There are questions of fairness in the use of AI – whether only the large centres like Toronto, Montreal, Vancouver, Calgary and Edmonton will benefit from the technology, while rural hospitals will not.
•There isn’t much work being done to assess AI solutions the way medications are – through randomized trials. Physicians will have more trust in AI applications if they are proven in this way.
•Legislators are having trouble keeping up with the fast rate of change in the AI sector. As Dr. Razak noted, a little over two years ago nobody had even heard of Large Language Models (LLMs).
tients become delirious in hospital, their risk of dying doubles. For those who survive, the odds of being admitted to long-term care goes up 2.5-fold. Moreover, the cost of care in hospital increases by $11,000.
“It’s incredibly consequential,” said Dr. Razak. He explained that his team has trained an AI algorithm on GEMINI data that has been able to identify 90 percent of delirium patients.
“So, it’s taken us from 25 percent to 90 percent … That is enough now to deploy into the healthcare system with a randomized trial.”
He said it’s currently the largest randomized trial of an AI healthcare solution in Ontario, and it is being conducted in partnership with Ontario Health.
Dr. Razak, who co-leads the GEMINI project at Unity Health, with Dr. Amol Verma, noted the system has data on 3 million Canadians. “It’s the most used data set for AI innovation in the country now,” he said. “It’s the primary data set for Vector [an AI centre of excellence based in Toronto]. And it’s leading the largest AI deployment in the country, across 12 Ontario hospitals.”
Meanwhile, at Ontario Shores Centre for Mental Health Sciences, in Whitby,
about 15 AI projects in the pipeline, said Duska Kennedy, vice president of strategy and digital health. The hospital has been careful to ensure that a robust governance strategy has been put into place, and that the projects can integrate with the hospital’s Oracle electronic health record system.
Moreover, the organization has started with lowrisk projects, “so we can learn and then expand from there,” said Kennedy. Still, she said these are not experiments, but systems that will deliver real-world value while educating staff and giving them the ability to take on even more challenging AI projects.
One of the systems NYG is currently working on is SmartER Zones, an AI-driven solution to support triage nurses in the emergency department. Like all EDs, it deals with high volumes of patients and nurses who are under a great amount of pressure.
Kennedy explained that patient flow in the ED revolves around the triage desk, where nurses sort patients into various levels of acuity and send them to different ‘zones’. “The placement of patients puts a very high mental load on the triage nurses, so with SmartER Zones, we’re equipping our emergency department clinicians with a tool that identifies patients who have been waiting a disproportionate amount of time and recommends alternative care zones. This helps move patients out of the waiting room faster and improves overall ED flow,” she said.

•Many other countries are far ahead of us in the application of AI to healthcare.
Still, some leading-edge organizations in Canada are moving ahead with AI, testing the solutions devised by private-sector companies and in some cases developing their own algorithms.
Dr. Razak noted that Unity Health Toronto, in partnership with Ontario Health, is hard at work on a project to demonstrate the effectiveness of an AI solution that identifies delirium in hospital patients.
He asserted that delirium is a serious problem –affecting as many as 500,000 Canadian hospital patients per year – but that it is often not recognized by clinicians or data in hospital records. Indeed, current data identifies only 25 percent of patients afflicted with delirium, and more accurate methods are labour-intensive.
It’s a serious problem, he said, because when pa-
Ontario, a team has been developing an AI-powered system that determines whether a mental-health therapy is working or not – faster than ever before.
“There’s a long wait list for services for people with depression and anxiety,” said Marsha Bryan, director of data, analytics and quality at Ontario Shores. “And sometimes, you may not have patient recovery with the treatment you’re providing.”
It would be far better for the patient, and for others waiting to see a clinician, to determine whether a given therapy is working and if not, to try another one.
“Do we really need to have someone go through 16 or 20 sessions?” she asked. Far better to use data to decide whether something is useful, which could shorten the therapeutic journey for the patient.
“We’re moving the needle forward, so we can get a signal maybe at week eight or nine,” said Bryan. “And what that allows us to do is adjust the treatment that you’re getting. We don’t want to wait another 10 weeks before we realize that it’s not going to be effective.”
At North York General, in Toronto, there are
“We’re trying to avoid having patients leave our emergency department unseen, which unfortunately happens far too often,” she continued.
The goal is also to reduce wait times and to have patients examined by doctors faster, bringing about better patient experience and safety.
“We grounded the model in historical data and validated it through simulations with experienced nurses and physicians to make sure it reflects real clinical practice and earns the trust of our frontline teams,” Kennedy explained.
Dr. Muhammad Mamdani, vice president, data science and advanced analytics at Unity Health, discussed the hospital’s creation of ChartWatch, an early warning system that detects when patients are crashing.
“It considers between 150 and 170 parameters, including demographics, vitals and labs. And it predicts if somebody’s going to die or go to the ICU in the next 48 hours,” he said.
ChartWatch has been used at St. Mike’s for about five years and has demonstrated a 26 percent reduction in patient mortality. When it detects a patient in need of attention, alerts go out and a team must respond. That has saved many lives at St. Mikes and at St. Joseph’s, in Toronto, as well.
Dr. Mamdani described another AI project at St. Michael’s Hospital that uses AI technology in radiology. The problem, he said, is that between 10 and 20 percent of brain bleeds are missed in emergency room patients. Now, he said, every CT head scan done in the emergency department will be read immediately by an AI system. “In just under two minutes, it will tell you not only if there’s a bleed or a different condition. It will also tell us whether a patient has to be in the operating room right away.”
Project could transform blood tests, saving billions of dollars worldwide
BY LISE DIEBEL
The director of a renowned Hamilton Health Sciences (HHS) research lab is harnessing the power of artificial intelligence (AI) to develop state-of-the-art precision blood tests that would radically reduce the cost of blood testing, saving billions of dollars worldwide. These new tests would also improve patient care by offering more insights into a person’s health.
“This project has the potential to transform blood testing, much like digital photography replaced film,” said medical biochemist Dr. Guillaume Paré, director of the HHS Clinical Research Laboratory and Biobank – Genetic and Molecular Epidemiology Laboratory (CRLB-GMEL). Paré launched the start-up company Pareon Biosystems to develop precision blood tests using AI.
Precision blood tests are advanced types of tests that give doctors more specific and personalized insights into a person’s health by measuring multiple tiny markers such as proteins, genes and other molecules to better monitor health and track diseases. Biomarkers are a protein, chemical or gene that can show if something’s wrong, such as an infection or illness. Biomarkers can also help doctors track how well a treatment is working.
Globally, over $134 billion is spent each year on blood tests alone, said Paré. “It’s a

massive international market in need of better solutions, and our tests could dramatically decrease this cost.”
How savings work: Traditional bloodwork involves multiple tests to check for diseases and give an overall picture of a person’s health. On average in North America, about 25 blood tests are done per person, per year, amounting to about 10.5 billion tests each year.
While many blood tests are inexpensive on an individual basis, when combined they add up to tens of billions of dollars worldwide. Pareon Biosystems uses AI to find a wider range of patient health information using customized precision blood tests that check for multiple biomarkers in one sample.
“Just three years ago, our tests wouldn’t have been possible,” said Paré. “But rapid advancements in AI have allowed scientists to innovate at a much faster pace.”
A winning idea: In support of this project, HHS awarded Paré, and Pareon Biosystems, a 2025 DRIVE Spark grant of $50,000 at October’s Research & Innovation Awards of Excellence event.
DRIVE Spark funding grants from HHS support original, new projects aimed
at improving health-care delivery. DRIVE – which stands for Dare, Research, Innovate, VenturE – cultivates the development of HHS research and innovation into commercially viable medical technologies and solutions.
“We’re currently at the starting point,”
Friday, April 24, 2026 8am 5pm
Liuna Station
360 James St. N. Hamilton, Ontario
Register now
Early bird pricing until April 1
$100 individual / $80 per person for groups (5+)
Breakfast + lunch + coffee/snacks included
Learn more & register: www.miit.ca

said Paré, adding that he sees enormous potential for improved patient care down the road. “For example, a hospital emergency department could test every patient coming through their doors, and with a single blood sample that checks for multiple biomarkers, and triage patients based on their test results for much faster, targeted care.”
Paré was one of three innovators to receive a 2025 DRIVE Spark grant of up to $50,000 to support commercialization of new, game-changing healthcare innovations.

MIIT 2026
Canada’s Medical Imaging Informatics Conference.
Local. Practical. Connected.
Learn what’s next in imaging informatics – right here in Canada. Join us for a full day of practical insight, emerging tech, and realworld problem solving – built for imaging leaders, informatics teams, IT, and clinicians.
FEATURED KEYNOTE
Dr. Woojin Kim (Chief Strategy Officer & CMIO, HOPPR; CMO, ACR DSI)
“The Promise and the Pitfalls: Navigating Generative and Agentic AI in Medical Imaging”
ON THE AGENDA
• Agentic AI & foundation models in radiology
• Imaging informatics best practices & career planning
• PanCanadian updates, cybersecurity, and more
• Vendor talks + networking (with prize draw)
• Interoperability standards that actually work
NETWORK + LEARN IN A WELCOMING COMMUNITY
Meet – and learn from – leaders, faculty, and peers in an inclusive, welcoming environment. Stay after the meeting for the networking event and keep the conversation going.
Dr. Guillaume Paré PHOTO:
Trillium Health Partners deploys clinical AI to provide faster, safer care
BY NORM TOLLINSKY
Trillium Health Partners, a community-based academic health system serving Mississauga, Ont., and surrounding communities, is advancing a system-level approach to clinical AI as embedded infrastructure for faster, safer care.
“At Trillium Health Partners, we see AI as foundational infrastructure – not simply a standalone technology project – enabling better quality, access, equity, and system integration across hospital, community, and home-based care,” said Dr. Ivan Diamond, program chief and medical director of the Cator Family Diagnostic Imaging Program at Trillium Health Partners.
In June 2025, Dr. Diamond’s team launched a clinical AI application designed to accurately identify and notify physicians of intracranial hemorrhage (ICH) in realtime. Running in the background on head CT scans, the tool strengthens clinical decision-making for radiologists and emergency physicians while maintaining a clear focus on patient safety, quality, and experience.
The AI application was acquired via Sectra Amplifier Services, which features 100+ validated third-party AI apps for radiology, pathology, and cardiology that integrate with the Sectra diagnostic viewer.
“The tool flags the possibility of a bleed right away, immediately triaging urgent cases to the top of the queue, helping us deliver high-quality care faster,” said Dr. Diamond. “Particularly in high-volume and time-sensitive settings like the emergency department, where saved time can mean the difference between perma-
nent harm and a successful recovery.”
In outpatient settings where CT scans are routinely reviewed the next day, the algorithm can identify potential intracranial hemorrhages earlier, prioritizing those patients for prompt review. Additionally, for ED physicians, who look at these cases initially themselves, AI gives them more confidence in their diagnoses.
The selected application also detects and flags cases of midline shift, mass effect and skull fractures. Midline shift is the displacement of brain structures away from their normal central position, while mass effect occurs when a tumor, bleeding, swelling or infection takes up extra space in the skull, pushing on and compressing brain tissue.
Sectra Amplifier Services has been live since 2022 with adoption in Europe. Trillium Health Partners’ acquisition of the ICH detection application is the first time it has been deployed in Canada.
“Obtaining AI applications through Sectra Amplifier Services guarantees that the AI is baked into the workflow,” said Stephen Harrold, Sectra Canada’s director of strategy and innovation. “All the AI applications within Sectra Amplifier Services are also pre-validated for compliance with the regulatory agencies.”
Prior to going live, Trillium Health Partners validated the application’s performance using historical scans from its own archives. “Because we serve one of Canada’s most diverse communities, it was important to evaluate and validate the application against local data to ensure it performs as intended for our patient population,” explained Dr. Diamond. Local validation was performed by the AI Deployment and Eval-
uation Lab at Trillium Health Partners’ Institute for Better Health.
Sectra and the selected AI vendor representatives were also onsite at the hospital to assist with training radiologists, technologists, and ED physicians, who were coached on how to use the application and made aware of its limitations.
When evaluating the performance of a diagnostic test, sensitivity and specificity are crucial metrics that help determine the test’s ability to identify positive and nega-

tive cases. Sensitivity measures how well a test correctly identifies positive cases, while specificity measures how well it correctly identifies negative cases. Both metrics are essential for making informed decisions.
Consistent monitoring of the application is necessary because its performance could ‘drift’ if significant changes occur, such as major upgrades to the hospital’s CT scanner or substantial shifts in the hospital’s patient population.
While it’s still too early to have all the necessary data on the algorithm’s performance, Dr. Diamond estimates his team is seeing three to four cases per month where the algorithm detects a subtle finding that may not have been seen by a human.
“We are impressed by the algorithm’s ability to correctly prompt cases for earlier review, which is enhancing decision-making, as well as improving care,” said Dr. Diamond. Like all diagnostic AI applications, the product is locked, meaning that the AI application doesn’t learn to improve its performance between scheduled updates to the algorithm. “We only see changes in the tool once they’ve been approved by Health Canada,” he said.
Trillium Health Partners is investing in AI across the organization as a key enabler of its plan to 2030, including a lung nodule AI algorithm that comes with a scanner the hospital uses, as well as a bone AI application developed in-house.
Harrold notes that AI adoption in Canada is accelerating as hospitals across the country undertake major refreshes of their PACS systems. As these upgrades move forward, Sectra’s market share in Canada has expanded in recent years to more than 30 percent.
“Because our goal is to never miss a bleed, we optimized sensitivity over specificity. This gives our highly experienced radiologists additional areas to target and rule out,” said Dr. Diamond. “Working with Sectra, the ability to provide feedback is built directly into the clinical workflow. That makes it easy for our teams to contribute to continuous learning and improvement as part of everyday care.”
AI procurement requires new ways of evaluating providers and data
BY LAURIE LAFLEUR AND SHIRLEY FENTON
Artificial Intelligence has quickly shifted from a futuristic concept to a clinical and operational necessity across Canadian healthcare institutions. However, as healthcare leaders look to integrate these tools, they are discovering that the traditional “IT procurement playbook”, which was designed for commodity software with predictable workflows, is no longer sufficient.
Procuring AI in a Canadian context requires a fundamental shift in how we assess value, risk, and data. For innovators and health system leaders alike, the challenge is not just “buying a tool,” but managing a dynamic, data-dependent lifecycle that demands a new level of strategic oversight.
Why AI procurement is different: In traditional healthcare IT, procurement typically follows a “purchase and implement” model. For instance, you set out to purchase a healthcare IT system, ensure vendors can meet clinical requirements and underlying technical standards, then manage the deployment of the selected application. AI, however, introduces three distinct shifts that complicate this linear path:
•The “narrow AI” fragmentation: Unlike the enterprise (like the EHR) or departmental systems (such as a PACS), AI is highly fragmented. Most models are designed for specific, “narrow” use cases like detecting lung abnormalities on a chest X-ray or predicting sepsis in the ICU. For leaders, this means procurement is not a one-off event, but a strategy for managing a growing ecosystem of many diverse algorithms across the continuum of care.
•From on-premise to cloud-first: While many Canadian hospitals have historically preferred on-premise servers, high-performance AI models often require cloud hosting for necessary compute power. This not only involves a shift in how IT teams integrate with these systems and vendors; it also necessitates rigorous assessment of data residency to ensure sensitive patient information remains under Canadian legal jurisdiction and adheres to federal and provincial privacy acts.
•Data as an infinite resource: In traditional IT, data is an output. In AI, it is the fuel for training and refinement. This creates another unique challenge; vendors often require access to your data not just to run the tool, but also to refine the model. Leaders must decide early on
if they will allow institutional data to be used for global model training and under what ethical terms.
Core challenges in the Canadian landscape: Canada faces a unique set of hurdles that can stifle innovation if not navigated with precision:
•Fragmented governance: Procurement requirements often vary wildly between


provinces and even between health authorities. Without a standardized model, vendors navigate a “patchwork” of compliance standards, which increases costs and slows deployment.
•The Black Box problem: Leaders often struggle with “explainability”. If an AI system suggests a treatment path, how do we procure a system that allows clinicians to
see the “why” behind the decision to meet their standard-of-care obligations?
•Innovator barriers: Small Canadian AI startups often find the “Standard RFP” process prohibitive. The high cost of meeting broad security requirements – originally designed for multi-billiondollar corporations – can lock out the very innovators the Canadian ecosystem is intended to foster.
Strategies for success: Leveraging national frameworks: To overcome these hurdles, Canadian healthcare leaders should look toward emerging standardized models and specific strategic frameworks that have been proven both domestically and abroad:
1. The Infoway pre-approval model: A significant development is the move toward national pre-qualification. Canada Health Infoway has worked to propose key elements of an AI governance framework aimed at helping delivery organizations adopt these technologies responsibly. Whenever possible, leverage “Pre-Qualified Vendor” lists. These vendors have already undergone vetting for cybersecurity, interoperability, and privacy, drastically reducing the local administrative burden.
2. The CAN Health Network “Edge”
Dr. Ivan Diamond, DI medical director
Laurie Lafleur
Shirley Fenton
Radiology shifting from AI adoption to the AI Quality Framework
BY JEFF VACHON
Across academic and enterprise health systems, AI tools are now routinely embedded within imaging workflows, supporting triage, image detection, quantification, reporting, and operational coordination. The more pressing challenge today is not adoption but understanding how well these systems perform once they become part of daily clinical reality.
As AI assumes a more central role in radiologic practice, it increasingly influences diagnostic decisions, workflow and clinical management. In this environment, traditional approaches to AI evaluation, often based on static, retrospective accuracy metrics, are no longer sufficient.

Radiology now requires a framework that can continuously assess how intelligently AI systems operate within real-world clinical environments. This need has given rise to the concept of the AI Quality Framework (AIQ): an evidence-driven approach to measuring AI performance as it is experienced by radiologists, departments, and patients. From point solutions to embedded intelligence: Early generations of radiology AI were characterized by narrowly focused algorithms designed to address specific diagnostic tasks. These tools often existed outside the core imaging workflow and were accessed selectively, requiring additional interfaces or manual steps. While clinically promising, their impact was inherently constrained.
Today, AI has migrated into the foundational infrastructure of radiology. Algorithms are increasingly embedded directly into AI platforms, reporting systems and enterprise workflow engines.
Rather than acting as optional add-ons, AI systems now operate continuously in the background – prioritizing studies, generating structured findings, surfacing quantitative measurements and supporting operational decision-making.
In many departments, AI has effectively become part of the digital backbone of imaging services.
As AI becomes pervasive, radiology must move beyond asking whether an algorithm is accurate in isolation and instead evaluate how intelligently it performs within the broader context of a complex diagnostic system guided by human expertise.
Why traditional AI metrics are no longer sufficient: Most AI systems enter clinical use on the strength of validation studies conducted in controlled research environments. These studies typically report sensitivity, specificity, and related statistical accuracy measures derived from curated datasets with known ground truth.
While essential for regulatory clearance and early assessment, these metrics represent only potential performance.
Clinical radiology, however, is dynamic.
Image quality varies, disease prevalence shifts, biases become transparent, patient populations differ across jurisdictions and over time.
Workflow integration influences how AI outputs are interpreted and acted upon. Human judgment remains central, and ra-
diologist interpretation itself introduces variability. Under these conditions, AI performance cannot be assumed to remain static after deployment.
An algorithm that performs well during development may drift, degrade, or behave unpredictably once exposed to real-world complexity. Static accuracy metrics cannot capture these changes.
Intelligence, by contrast, implies consistency, adaptability, and contextual awareness. Measuring AI intelligence therefore requires continuous observation within
Building a “Front Door” for personalized disease prevention
funded by Canada Health Infoway, focused on a key question: Which self-administered questionnaires are validated and feasible for preventive intake in primary care?
To address this issue, Persoventa employed a tailored, systematic search strategy across 15 domains, including cancer screening, environmental health exposures, ethnic susceptibility, geriatric and aging health, mental health, nutrition, occupational health, oral/dental conditions, physical inactivity, sexual health, sleep health, socioeconomic determinants, spiritual/religious influences, substance use/dependency, and youth health concerns.
Fifteen specialized teams, each focused on a specific domain and supported by 65+ methodologists, clinicians, and subject-matter experts, searched scientific databases and grey literature. Their focus was on studies published between 2014 and 2025, prioritizing tools relevant to preventive assessments in populations aged 15 and older.
The review examined approximately 43,000 records and used a standardized selection process to find tools that met evidence and feasibility standards. The result was a final list of 31 self-administered questionnaires – one or more per domain – designed for universal, front-line preventive screening in primary care settings.
From “Validated” to “Implementable” – closing the readiness gap: A key insight from Phase 1 was that prevention tools often fail not because they lack scientific credibility, but because they are not de-
AI agents interact
CONTINUED FROM PAGE 15
several more CT and MRI sequences, with the same kind of back-and-forth discussion. Eventually, the AI agent suggests a pituitary adenoma as the most likely diagnosis.
While all this was astounding, Dr. Prevedello stated that the agent’s final diagnosis was incorrect. “It was actually a very rare case of a chordoma simulating a sella lesion,” he said.
Still, the system’s ability to work with the radiologist in natural language, and to call up images and point out areas of concern, was remarkable. “What’s even more impressive,” said Dr. Prevedello, “was that the agent was not specifically trained for radiology. It was a generalpurpose model.”
He then demonstrated two more agents that diagnosed sets of images.
signed for “front door” use. Many well-validated instruments are too lengthy, lack clear cut-points, or do not offer triage guidance – leaving clinicians and patients with results that are difficult to interpret and even harder to act on.
To address the challenges in evidence readiness and intake readiness, Persoventa implemented a dual-index approach:
•The Screening Proof Score (SPS) captures a tool’s foundational readiness to
Persoventa employed a tailored, systematic search strategy across 15 domains, including cancer screening.
screen by assessing (i) self-administration suitability, (ii) psychometric strength, and (iii) Canadian relevance/validation.
•The Persoventa Suitability Index (PSI) assesses front-door fitness, focusing on (i) risk-factor coverage, (ii) feasibility (length, complexity, accessibility), and (iii) patient usability/acceptability.
Importantly, PSI was enhanced through feedback from Canada Health Infoway’s Patient Advisory Committee, which reviewed recommended tools for clarity, cultural appropriateness, comfort with sensitive topics, and perceived relevance. This ensured that the final recommendations were not only evidence-based but also practical and acceptable for diverse populations.
Recommendations and conclusion: Phase 1 establishes a solid foundation for a unified preventive intake pathway and
One of them analyzed a large series of CT exams in less than two minutes. One of the agents got the diagnosis correct, while the other didn’t.
“I think the technology is still not there yet, especially for rare conditions,” said Dr. Prevedello. However, it’s early days for these systems – what’s coming will be even more formidable. Even in the future when these agents
The system’s ability to work with the radiologist in natural language was remarkable.
are much more accurate, Dr. Prevedello asserted they will always require human supervision, as there is always a chance of error. “The computer is incredibly fast, accurate and stupid. But man is unbelievably slow and accurate and bril-
clarifies the next steps required to translate tools into an actionable system.
•First, optimize usability where evidence is strong. Tools with strong SPS but weaker PSI should be enhanced by bundling brief measures, shortening or reordering items, and adding clear thresholds and referral pathways to ensure results lead to actionable next steps.
•Second, strengthen Canadian readiness where usability is high, but evidence is incomplete. High-PSI tools without Canadian psychometrics should be prioritized for validation studies, bilingual development, and culturally suitable adaptation, while addressing licensing constraints early to support scaling.
•Third, embed equity and governance from the outset. Prevention cannot be personalized without careful handling of identity and socioeconomic data. Persoventa’s upcoming phases will require governance informed by equity and community partnerships to ensure tools support care improvement rather than reinforcing inequities.
•Finally, design for interoperability to enable AI. Phases 2-4 will align questionnaire items and outputs to standards, validate risk factors against Canadian guidelines, and develop a semantic risk factor–disease knowledge base.
Dr. Abbas Zavar is Adjunct Faculty, Master’s in Health Informatics Program, University of Toronto and Digital Health Research Lead, OntarioMD. Dr. Roya Farzangean is a Visiting Researcher at Toronto Metropolitan University.
liant. And the marriage of the two is beyond calculation.”
Dr. Nancy Pham, a neuroradiologist at Stanford University, highlighted several areas in which AI is helping radiologists. She noted right away that imaging volumes are increasing at a rate of 3 percent to 12 percent annually, while there are only about 2 percent more radiologists each year. “So, that means we have a large and unsustainable workload gap,” she said. The fast growth in DI exams is also leading to burnout and the possibility of more diagnostic errors. For these reasons, she said, AI is being harnessed to improve efficiency and workflow automation.
There are some real success stories already, she said. In a trial of AI reading for breast cancer, 80,000 women were screened and there was a 29 percent increase in cancer detection using an AI tool. There was also a 44 percent reduction, she said, in radiologist workload.
Jeff Vachon
Radiology shifting from AI adoption to the AI Quality Framework
clinical practice rather than episodic retrospective review.
Defining the AI Quality Framework: The AI Quality Framework (AIQ) reframes how radiology evaluates AI performance. Rather than focusing solely on technical accuracy, AIQ assesses how consistently AI outputs align with radiologistreported findings, how reliably the system avoids false positives and false negatives, and how much incremental value it adds to clinical interpretation.
AIQ is derived from a composite of clinically meaningful performance indicators calculated directly from routine imaging workflows. By comparing AI-generated findings with finalized radiology reports on a case-by-case basis, AIQ reflects how AI behaves in the same clinical context as the radiologist. Importantly, the framework allows institutions to weight these indicators according to clinical priorities, recognizing that intelligence may
be defined differently for screening, triage, or diagnostic augmentation.
In this way, AIQ functions not as a single abstract score, but as a structured measure of real-world clinical intelligence.
Measuring operational impact as a component of AI intelligence: As AI becomes embedded in daily workflows, its value must also be assessed through its impact on radiology performance and productivity. Within AIQ, operational metrics are intentionally evaluated alongside diagnostic KPIs to provide a unified view of how AI influences both patient care and departmental function.
Radiologist read-time is one of the most direct indicators of AI’s operational impact. AI systems that automatically identify findings, generate measurements, or populate structured report elements are designed to reduce cognitive and manual workload.
Within AIQ, changes in read time are interpreted in parallel with concordancebased KPIs such as overall concordance,
positive and negative concordance rate. Consistent reductions in read times with stable or improving AIQ metrics indicate that AI is effectively supporting clinician decision-making, while drops in diagnostic alignment may point to unsafe speedups or workflow issues.
Turnaround time offers a broader view of AI’s influence on care delivery. AI-dri-
These metrics provide evidence that AI is not merely accelerating workflows but actively improving diagnostic insight.
ven triage and prioritization tools are intended to surface critical findings earlier in the workflow, particularly in emergency and high-acuity settings.
Improvements in turnaround time that coincide with stable AIQ scores indicate successful workflow integration. Conversely, gains in speed accompanied by de-
Red Rover’s approach creates a platform for innovation
BY MICHAEL BILLANTI
Digital transformation is reshaping healthcare, but a persistent challenge slows progress: integration. Healthcare systems have long relied on traditional HL7-style interfaces, resulting in rigid, time-consuming, and resource-intensive integration efforts that limit speed, innovation, and scalability.
Red Rover Health is changing that narrative.
Instead of requiring a system-wide overhaul, the company uses a more open, flexible, and future proof approach to interoperability. Integration is reimagined as a launchpad for a connected ecosystem, not just a way to link systems. With a secure, scalable, and API-driven platform, healthcare systems and applications can move faster, deploy smarter, and connect better.
The healthcare sector has relied on HL7 for health data exchange and transfer for a long time. As it shifts to an APIdriven environment, these point-topoint interfaces are proving to be less efficient and scalable. Each new digital solution often requires a custom, one-off interface build. However, this increases complexity for IT teams and significantly delays the time to value.
At its core, Red Rover Health provides a RESTful platform designed to streamline integration through a one-tomany approach. Rather than building unique connections for each new application, the platform creates a centralized, reusable connection layer. This layer supports multiple applications, functioning much like an “app store” for healthcare.
This model delivers several key advantages:
•Faster Implementation: It gets solutions up and running in a fraction of the time.
•Simpler Testing: Utilizes a secure sandbox environment to test and vali-
date integrations efficiently.
•Seamless Data Flow: Enables bidirectional data exchange without reinventing the wheel for every application.
With security and compliance at the forefront of the platform, healthcare organizations trust every connection with confidence, with SOC 2 Type II certification, CSSAP approval from Oracle Health, and best practices for security integrated into its architecture.
Operationally, this shift delivers tangible benefits. IT teams deal with fewer support tickets, reduce vendor-specific maintenance, and gain greater visibility through centralized monitoring and governance.

Red Rover Health is advancing the integration model; not by replacing HL7, but by simplifying complexity with modern APIs, enabling healthcare systems and applications to innovate freely without constraints on integration. Simplifying integration: As health systems modernize their digital ecosystems, they increasingly seek partners who simplify complexity, rather than add to it. One such example is TransForm Shared Service Organization (TSSO), a non-profit shared service organization supporting healthcare providers in southwest Ontario through shared infrastructure, clinical priorities, and health system needs.
TransForm was looking for a partner that not only understands integration but truly understands interoperability.
“Supporting our patients, families, clinicians, and staff requires far more than simply moving data from point A to point B. What is required is the ability for data to move seamlessly between
clining negative concordance or increasing false-positive alerts may reflect prioritization inefficiencies that warrant governance review.
At the departmental level, throughput metrics such as studies interpreted per shift or relative value units (wRVU) generated provide insight into AI’s effect on capacity management.
Within an AIQ-aligned model, throughput is never evaluated in isolation. Increases in volume are assessed alongside AIQ trends, drift indicators, and radiologist interaction metrics to ensure that productivity gains do not come at the expense of diagnostic intelligence or clinician trust.
Augmented diagnostic capability and clinical value: Beyond efficiency, AI’s most meaningful contribution may be its ability to enhance diagnostic capability. AIQ captures this dimension through measures such as augmented findings rate, which quantify AI’s incremental contribution relative to baseline interpretation. These metrics provide evidence that AI is not merely accelerating workflows but actively improving diagnostic insight.
systems – arriving in the right place, in the right format, ready to be used to support patient care without forcing teams to re-enter information or navigate unnecessary steps,” said Lyn Baluyot, CEO, TransForm Shared Service Organization.
She adds, “Red Rover Health recognizes this need and shares our commitment to strengthening the systems we rely on today, without constant rebuilds, replacements, or costly reinvestments.”
Enabling ecosystems, not just connecting systems: In today’s health-tech landscape, one thing is clear: interoperability is not just about exchanging information, it is about enabling new and efficient workflows on either side of those connections.
Red Rover Health’s neutral, secure, and developer-friendly infrastructure serves as a powerful connective layer, that enables healthcare systems and applications to:
•Collaborate across diverse EHR types without rebuilding integrations for each instance
•Embrace a best-of-breed application model while reducing operational overhead
•Accelerate the journey from concept to deployment
•Allow integration to enable configurable workflows
•Uphold data security, compliance, and operational simplicity
•Make it easy for AI vendors and developers to integrate into large EMR’s
While legacy integration models focused on linking individual systems, Red Rover Health is focused on powering entire ecosystems, helping healthcare organizations unlock the full value of the digital tools they choose to deploy.
We believe it’s not just improving integration but is enabling a new era of innovation in healthcare.
Michael Billanti is VP, Global Business Development, Red Rover Health.
The need for continuous monitoring: A defining characteristic of radiology is that it evolves over time. AI systems are no exception. Changes in patient populations, imaging protocols, scanner technology, or clinical workflows can subtly influence performance long after deployment. Without continuous monitoring, these changes may remain undetected until they manifest as clinical risk.
AIQ addresses this challenge by enabling longitudinal tracking of AI performance across diagnostic and operational dimensions. Trends in AIQ scores, concordance metrics, and productivity indicators provide early warning of drift or instability.
This allows radiology departments to intervene proactively – adjusting workflows, retraining models, or restricting use when necessary – rather than reacting to failures retrospectively.
Implications for radiologists and radiology leadership: As AI becomes more deeply embedded in practice, the role of the radiologist is evolving from sole generator of findings to supervisor of AIaugmented interpretation. For this model to succeed, trust must be supported by evidence. AIQ provides that evidence by making AI behavior transparent and measurable.
For radiology executives, AIQ offers a strategic governance tool. It supports informed decisions about AI procurement, deployment, scaling, and retirement, while providing defensible documentation for quality oversight and regulatory readiness. By integrating clinical and operational intelligence into a single framework, AIQ enables leadership to manage AI as a core component of radiology performance rather than as an isolated technology initiative.
AIQ as the foundation of intelligent radiology: Radiology is no longer questioning whether AI works, but how intelligently it performs in real-world clinical practice. AIQ offers a framework to ensure AI remains trustworthy, clinically aligned, and beneficial to patient care as AI diagnostics become the norm.
Jeff Vachon is the president of Bialogics.
Michael Billanti
Synthetic data
variables – demographics, diagnoses, treatments,” said Dr. Margarita Mersiyanova, health and life sciences industry consultant with SAS. Synthetic datasets are most often generated using statistical and probabilistic methods, machine learning generators, or a hybrid of the two, she said.
Building trust: Synthetic data can be particularly valuable for specific use cases, Rothman said. For example, the cost to collect or purchase real data might be prohibitive; existing real data might require timeintensive, error-prone labeling; existing data might be biased or lack adequate representation of groups or segments; or next to no data might exist for some scenarios.
But using synthetic data comes with risks. Mersiyanova points to a possible problem common to generative AI technologies: hallucinations. These are the false, fabricated pieces of content that large language models can produce, while amplifying original biases within the data set.
“If we don’t have good underlying data to start with, it will impact any data that is synthetically generated,” Dr. El Emam said. “This is a broader issue in healthcare, where data collection tools are primarily designed for administration, and data is spread across various systems.
Governing AI
CONTINUED FROM PAGE 16
liability, and practice scope. Early safe-harbour work by OntarioMD and Canada Health Infoway created space for responsible deployment. Health Canada’s restraint avoided premature rigidity. Take the win. Even if it wasn’t 100 percent planned.
Federal–provincial roles, trade, and sovereignty: These regulatory questions do not sit neatly within existing constitutional lines. Healthcare delivery and professional regulation are provincial responsibilities. AI governance, competition policy, privacy, and trade are federal domains. Generative AI cuts them.
Provinces control procurement, deployment, and day-to-day clinical governance for digital health systems (if anyone does). The federal government sets the legal, eco-
AI procurement
CONTINUED FROM PAGE 20
model: The CAN Health Network offers an integrated marketplace that helps Canadian innovators bypass typical procurement hurdles. Their model allows hospitals to partner with homegrown tech companies to validate and scale solutions in a real-world setting, providing a sandbox for discovery and validation before full-scale procurement or implementation.
3. Drafting your requirements: When drafting your RFP, three areas require specific AI-focused clauses:
•Data governance: Define policies for data collection and sharing early on. Ensure Right to Audit clauses are included for algorithmic performance.
•Privacy and security: Organizations should complete an Artificial Intelli-
“Before any generative AI-assisted tool touches care delivery, you need guardrails that focus on safety, accountability and trust.”
And, as always with healthcare, privacy is the elephant in the room.
On the one hand, synthetic data can be a valuable tool for de-identification. In Ontario, regulatory guidance specifically
“If we don’t have good underlying data to start with, it will impact any data that is synthetically generated.”
acknowledges how it can reduce re-identification risk in structured data.
On the other hand, there’s a potential risk of “over-fitting,” which occurs when a generative model is trained on a data set so closely that it reproduces records, leading to identity disclosure. Proper training practices and post-generation testing can ameliorate that risk, Dr. El Emam explains, but generated data can never be a zero-risk proposition.
“Zero risk is not achievable or expected in practice, and if you transform data so aggressively that you destroy its utility, there’s no point in transforming it at all,” he said.
Rothman describes the creation and use of synthetic data as a “privacy-accuracy balancing act.”
nomic, trade, and competitive conditions under which these systems operate.
Generative AI amplifies scale and affects FPT dynamics. Provinces cannot counter global platform power on their own. Global vendors will not navigate a fragmented patchwork of provincial rules. Only federal leadership will prevent fragmentation, regulatory arbitrage, and uneven standards.
A lean, AI-ready national capability could support measurement frameworks, oversee AI capability classification, manage shared datasets and synthetic testbeds, and coordinate competition policy with the Competition Bureau. Health Canada has renewed Bill C-72 by introducing Bill S-5 on patient data, interoperability and anti-blocking. Infrastructure needs to be built in Canada by Canadians for models, training data and synthetic data for all data and specifically for health data.
gence Fairness and Privacy Impact Assessment (AIFPIA) for all AI programs. This addresses AI-specific risks like model exfiltration or data poisoning.
•Ethical guidelines: Explicitly require that AI serves as augmented intelligence that supports, rather than replaces, human judgment. Require vendors to disclose the diversity of datasets used to train models to ensure the AI performs equitably across Canada’s diverse populations.
Establishing a clear ROI model: A potential pitfall for many AI projects is rooted in a lack of defined success metrics. Unfortunately, the path to ROI for AI in healthcare is rarely linear – it requires careful evaluation of both quantitative and qualitative benefits – like efficiency gains, quality improvement, cost management, and user satisfaction, while considering that AI does not yet have the clear budgets reimburse-
“There’s a trade-off. The more realistic a synthetic data set is, the less privacy it preserves,” said Rothman. “Adding more ‘noise’ to protect privacy makes the data set less realistic.”
Human oversight is essential. Generative AI tools should support clinicians, not replace them, said Dr. El Emam. There must be clear rules about how these tools are used, when a human must review or override the output, and who is ultimately responsible for decisions. It also means paying attention to how the tool is actually used in practice, not just how it performs in testing.
“For tools that influence care, it’s not enough to show that a model works in a lab setting. You need evidence that it performs reliably in the real clinical context, and you need to test for known failure modes of generative systems, such as hallucinated outputs, inconsistent behaviour across populations, and performance drift over time,” he said.
Looking ahead: Rothman said there is
growing interest in synthetic data, but its use across Canada is still rare.
“Initial interest focuses on the creation of structured tabular data, tables of health information,” he said. “It also shows promise for generating synthetic medical images to support research and diagnostic applications.”
Many of those applications require multiple tables in relational databases. Maintaining those data relationships in synthetic data can be challenging.
Adding to that is the longitudinal nature of much healthcare data. Synthetic data generators must preserve the relationships of data through points in time. Has a patient’s condition improved? Is the disease’s representation in various populations changing?
“If a synthetic data set does not look and behave like real-world data, then it won’t be useful, and in the worst cases, it can be counter-productive or even harmful,” Rothman said.
GoAutomate develops powerful AI agents
physicians and other colleagues.
department to help attain further efficiencies and to optimize patient care and safety. The agents and bots from GoAutomate are part of this effort – among the most advanced of their kind in a Canadian medical centre.
The team at Schroeder noted they’re still in the early stages of evaluating and deploying bots and AI agents.
Basrai noted that the AI agents are especially helpful in reducing the amount of te-
AI agents are especially helpful in reducing the amount of tedious, repetitious work that staff members perform. CONTINUED FROM PAGE 2
dious, repetitious work that staff members normally perform in any diagnostic imaging centre.
In the case of protocolling, it means that technologists and radiologists can focus on higher-value work – such as discussing care with patients, diagnosing exams and communicating with referring
ments structures used for more traditional IT systems.
To ensure a successful AI project, healthcare leaders must establish clear success criteria and KPIs that align with the goals and requirements of their respective organizations. These should be defined and refined across procurement phases, like pre-RFP (RFI), pilot, imple-
A potential pitfall for many AI projects is rooted in a lack of defined success metrics, both quantitative and qualitative.
mentation, and beyond. By building performance-based milestones into the procurement process – where full payment is tied to reaching specific ROI targets – organizations can mitigate the risk of AI hype.
“Some DI departments spend 10 hours a day on protocolling,” said Basrai. “You can reduce or eliminate those hours by using agents.”
Many people may be concerned that computerized bots are making decisions about their care. But Jason Daly, chief technology officer at GoAutomate, asserted that agents already have an accuracy rate “in the high nineties”.
And when there is any question about a form, document or decision, it’s flagged for human review.
As Basrai observed, a “human-machine collaboration” is emerging in the healthcare system through the use of AI. Humans are guiding the AI and people are in charge and making the final decisions.
By automating many processes, AI can not only reduce staff burnout and overworked frontline staff, it can also reduce the need to hire more staff for clerical functions, but it can also improve efficiencies, reduce errors, and speed up the process of booking diagnostic tests.
“Anywhere that people are spending a lot of time on paperwork, there are pain points that can be addressed,” Basrai said.
The path forward: AI procurement is not just another IT task; it is a strategic leadership function. By moving away from static procurement models and embracing a multidisciplinary, riskbased approach, Canadian healthcare leaders can ensure they are not just buying technology, but are instead investing in a more efficient, equitable, and intelligent future for patient care.
Learn more: You can learn more about AI procurement in National Institutes of Health Informatics (NIHI) courses on AI. For more information, see: www.nihi.ca.
Laurie Lafleur is a healthcare innovation consultant, a professor of health information science at Conestoga College and an instructor for NIHI. Shirley Fenton is president of the National Institutes of Health Informatics and a cofounder of Waterloo MedTech.

