POLICY BRIEF
Corporate Influence in the Infant Formula Industry
A Comparative Analysis and Policy Recommendations
Dinesh Bojja * , Zoey Duan * , Georgio Maroun * , Emily Neumeyer * , Lilia Potter-Schwartz*, Danielle Ricketts*, Andrew Storino * , Daniel Benamouzig, Cara Kiernan Fallon, Catherine Panter-Brick
*co-first authors
EXECUTIVE SUMMARY
• The global infant formula industry represents a network of stakeholders lobbying to promote their products for financial gain.
• Breastfeeding is associated with reduced mortality among infants and women as well as multiple related health outcomes.
• The International Code on the Marketing of Breastmilk Substitutes has sought to protect breastfeeding and regulate the role of manufacturers, but corporations continue to drive the use of infant formula.
• Three case studies of France, Germany, and the United States (US) highlight the role of direct, indirect, and digital lobbying in the global infant formula industry.
• We recommend governments classify breastfeeding as care work, recognize breastmilk within food security frameworks, and fund independent research on breastfeeding accessibility, counseling, and health outcomes.

BACKGROUND
The global infant formula industry represents a complex web of stakeholders, from young families to transnational policy-makers and food industry giants (Figure 1). While breastfeeding was once commonplace, since the end of the twentieth century, shifting opinions across the public and health sectors have caused rates to decrease globally to 42%.2 Consequently, global infant formula sales have risen from one billion to fifty-five billion USD within the last fifty years.2,3 This increase has also been accompanied by an expanded geographical reach by marketing and lobbying leaders.3 Understanding the role of corporate political activity, public perception, and governance in the formula industry is crucial to protecting the rights of women and families.
Following a decline in breastfeeding rates in the 1970s, concerns rose over an increase in mortality and malnutrition among young infants.2,4 At the time, many suggested that these detrimental health effects, particularly among infants in the developing world, were exacerbated by the aggressive marketing of formula. In 1979, the World Health Organization (WHO) and United Nations Children’s Fund (UNICEF) responded by convening a meeting with experts on infant feeding.4 The meeting culminated with the establishment of The International Code on the Marketing of Breastmilk Substitutes (the Code), which sought to protect breastfeeding by making recommendations regarding the marketing of breastmilk substitutes and infant formula (Figure 2).4,5 Since the Code’s introduction, 70% of countries have adopted at least some provisions of the Code, but only 18% have adopted all provisions.3 In the Code, the WHO recommends that children be breastfed for at least two years, and exclusively for their first six months of life.6

While the related health outcomes vary, some estimates suggest that near-universal breastfeeding could save an estimated 823,000 deaths in children under five and 98,000 maternal deaths from cancer and type-2 diabetes. The averted deaths in children are attributed to reductions in the risk of all-cause mortality, diarrhoea, and respiratory infections due to the nutrients and antibodies in breastmilk.3 For women, breastfeeding can prevent cancer by lowering estrogen exposure, limiting the development of cancerous tissue, and triggering immune responses to remove damaged cells.7
The gendered nature of breastfeeding and its correlation to socioeconomic factors raise concerns about the research, implementation, and ethics of breastfeeding promotion. Many pediatric associations, including the American Academy of Pediatrics and seven European pediatric societies, have strongly encouraged the promotion of breastfeeding at an individual level.8 However, researchers have also found that health outcomes associated with breastfeeding are correlated with underlying systemic causes, such as socioeconomic status.2,8,9 A woman’s ability to breastfeed is similarly influenced by societal factors, such as parental leave.9 In fact, demographic health survey data from 38 low and middle-income countries (LMICs) demonstrated that a one-month increase in paid maternity leave may be associated with a 2.2-month increase in average breastfeeding duration.6 Therefore, it is crucial that social policies are considered alongside international and
national standards in a “collective social approach.” 6,9 This policy brief analyzes the intersection of stakeholders, identifies specific methods of lobbying in three countries, and offers recommendations to address the profit-driven corporate activity that interferes with human rights regarding breastfeeding.
ANALYSIS OF CORPORATE INFLUENCE
Commercial Determinants of Health
Large corporations play an influential role in determining the state of global health—leveraging influence to determine the state of markets and directly impacting consumer health. Scholars have defined the commercial determinants of health as “strategies and approaches used by the private sector to promote products and choices that are detrimental to health.”10 Understanding how corporate strategies interact with and influence changing global commercial landscapes is critical to exposing the role corporations play in determining global health.
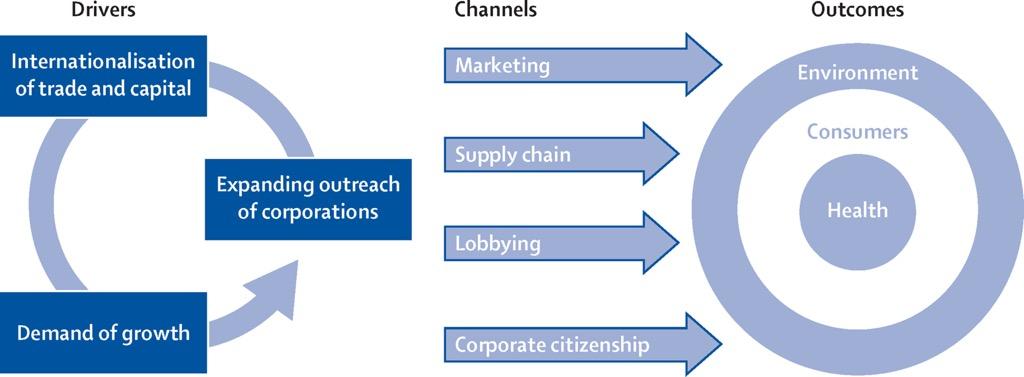
It is important to illuminate three main drivers of corporate power: expanding outreach strategies, internationalization of trade and capital, and demand of growth (Figure 3). These categories illustrate how corporations expand their reach, both driving and reinforcing rising demand, particularly in low- and middle-income countries. This favorable landscape creates an opportunity for large corporations to leverage product marketing to increase demand, build a positive reputation among consumers through social impact, and control markets through international supply networks (Figure 4).
infant formula industry use direct lobbying to advocate for political landscapes that work in their favor. For this industry and many others, favorable political landscapes lack regulation. This is exactly what direct lobbying seeks to accomplish by targeting high-level policy makers and influential agencies.




The role of marketing is a prime example of this. Marketing is used to increase product appeal to consumers and is a form of consumer outreach that the formula industry heavily relies on.12,14 Lobbying is used to ensure sustained freedom in infant formula advertising to increase global usage.12 Relatedly, corporations seek to counter regulatory actions from governments that might hinder this global scope. Expanding global reach and international trade agreements make it more difficult for governments to enforce national constraints. In this way, expansive international trade agreements work to the industry’s advantage.12,13 High-level policymakers are targeted to subvert efforts to support increased health regulations on formula use or recommended breastfeeding durations.13 These industry practices directly counter the recommendations of the Code.12 In combination with other strategic practices, they serve the ultimate goal of driving global breastmilk consumption to grow the industry and increase profits. These practices often directly contrast the expressed goals of the WHO and other agencies whose priorities lie in the “interests of its member states and their citizens,” according to the WHO’s 2001 Executive Director.13
Marketing and Advertising
Direct Lobbying
Lobbying is one tactic leveraged to influence the market environment of consumers, directly impacting consumption and health. The goal of lobbying is to allow corporations to maintain freedom from political interference in product promotion, manufacturing standards, and other efforts.12 This channel of corporate influence is employed by several large-scale industries, such as the tobacco industry, to maintain strong market influence.13 The infant formula industry is no exception to this, as there are skillful lobbying tactics being employed. Large corporations in the
Beyond direct lobbying efforts, multinational corporations often use indirect forms of influence to promote their products. For companies in the infant formula industry, this requires changing public perspectives regarding their products, often at the expense of informed and independent consumer decision-making.
The most potent form of corporate influence on public perception occurs through direct advertising and manipulation of social media. While aggressive marketing from the baby food industry is discouraged according to the Code, this type of consumer marketing remains pervasive. A joint report by the WHO and UNICEF found that 51% of women surveyed across eight countries have been targeted by advertising from the baby food industry.15 Such messaging can be false and misleading, reinforcing myths such as how infant formula has ingredients that improve infant immunity,
that formula is necessary in the days immediately after childbirth, and that breastmilk does not have sufficient nutritional value.15
The rise of digital advertising has exacerbated the problem. The WHO found that between January and June 2021, a sample of four million posts about baby feeding practices reached 2.47 billion people, with 12 million likes, shares, and comments.16 Formula companies themselves post on corporate social media accounts nearly 90 times per day, reaching 229 million users.16 A content analysis of the websites of infant formula companies in the United States (US) showed that these multinational corporations intentionally compare their products to breastfeeding to promote sales. By framing infant formula as a simple and ubiquitous alternative to breastfeeding, corporations normalize, and even idealize, its use. This overwhelming and biased messaging prevents mothers from making informed decisions about their own infant feeding practices by pushing corporate narratives upon them.
Professional Organizations
Infant formula companies also pursue marketing within the healthcare industry, using nurses and pediatricians to promote their products. In a survey of 200 pediatricians in the US, 85.5% reported visits from major formula companies, and 90% reported receiving free samples of infant formula. These companies disproportionately visited high-income, suburban areas with a focus on private practices, as these were places with the highest rate of breastfeeding.17 By turning healthcare providers into unassuming advertisers, the infant formula industry further drives misinformation, coercing mothers into using infant formula without a fair and equal understanding of all available options.
Academic Research
Multinational corporations also pursue indirect influence by exerting control over scientific research. Corporations in the formula industry deploy a range of tactics to influence academics, including funding research and providing grants, positions on advisory boards, sponsorships of conferences and professional associations, and direct honorariums.18,19 As a result, researchers can be incentivized to produce findings favorable to infant formula companies. Sponsored studies with negative results are often not published, while those with favorable outcomes are published to disproportionately promote the benefits of infant formula and ignore the negative or neutral effects.20 An analysis of 307 trials for infant formula products found
that 80% of the trials were at high risk of bias due to “inappropriate exclusions from analysis and selective reporting.”20 By engaging in the practices of scientific research, these corporations influence the evidence base that allows healthcare providers and mothers to make educated decisions about infant care. These practices reinforce misleading health claims about the promise of infant formula as a complete replacement for, or even a better alternative to, breastfeeding.
GAPS IN GOVERNANCE
Current weaknesses in the regulation of infant formula marketing enable the direct and indirect channels of influence that are widely exploited. Despite the adoption of the WHO Code by the World Health Assembly and its implementation in over 136 countries, significant gaps persist across international regulatory frameworks.5
Five key gaps are notable in existing governance practices globally, and are discussed in detail below:
1. Deficiencies in lobbying transparency mechanisms,
2. Inconsistent enforcement of violation penalties,
3. Provisional discrepancies from the WHO guidelines,
4. Insufficient digital marketing controls,
5. Failures and barriers in social policy.
1) Deficiencies in Lobbying Transparency Mechanisms
First, one of the most critical gaps in the current regulatory landscape is the limited scope and efficiency of lobbying transparency systems worldwide. In the European Union, for instance, the EU Transparency Register—a public, online database for lobbyists and organizations to record and self-report lobbying activities in the European Parliament, Council, or Commission––has major blind spots.21 The register employs only ten full-time staff to manage and track over 12,400 registered organizations.22 Registration of activity is only required for high-level meetings, allowing contact with lower-level staff and individual Members of the European Parliament (MEPs) to occur without disclosure.23 Individual MEPs can also meet with unregistered
lobbyists entirely outside the system, with the content of both scheduled and informal communications going unreported.24 Additionally, the register relies on self-declared data without independent verification. Companies like Nestlé have been found to underreport their lobbying expenditures by nearly €850,000 following complaints, which then triggered €47.7 million in corrections of unreported expenditures across the entire register, but no imposed penalties.25 Even with the subsequent audit conducted to remedy underreporting, trade associations like Specialised Nutrition Europe continue to declare unrealistically low lobbying costs despite operating at scale across multiple EU institutions.25 Similar transparency challenges exist globally. The US lacks comprehensive federal lobbying disclosure requirements specific to infant formula marketing, with much activity occurring at the state level, where regulations vary widely (US Food and Drug Administration, 2024). In India, lobbying activities remain largely opaque with no public registry system or systematic tracking, despite documented involvement of formula companies in shaping nutrition policy.27
2) Inconsistent Enforcement of Violation Penalties
Different enforcement standards across countries further undermine international governance and public health efforts. Despite evidence showing that consistent implementation of the Code yields better breastfeeding outcomes, enforcement remains uneven
globally. In the EU, Norway’s coordinated oversight system achieves positive outcomes, with 64% of infants exclusively breastfed at four months, whereas the United Kingdom’s industry-funded model, which imposes no marketing prosecutions, corresponds to rates of only about 7%.28,29 The US presents another weak enforcement model, with the FDA and the US Federal Trade Commission exercising limited authority over formula marketing beyond safety standards and false claims.26
3) Provisional Discrepancies from WHO Guidelines
Beyond enforcement challenges, there are also fundamental gaps between the WHO Code provisions and national legal standards that create systematic opportunities for marketing exploitation. The WHO code discourages all advertising of breast-milk substitutes to the general public, but EU law only restricts advertising for infant formula (0-6 months) and explicitly allows marketing of follow-on formula (6-12 months). 30,31 As a result, products for children 12-36 months face no advertising restrictions at all in the EU, and companies can take advantage of “cross-promotion” strategies to bypass regulation (Figure 5). 32 By promoting infant formula, they can indirectly increase their brand recognition and advertise infant formula products. Research confirms that mothers cannot distinguish between formula categories for different age groups and thus fall victim to such marketing tactics.33
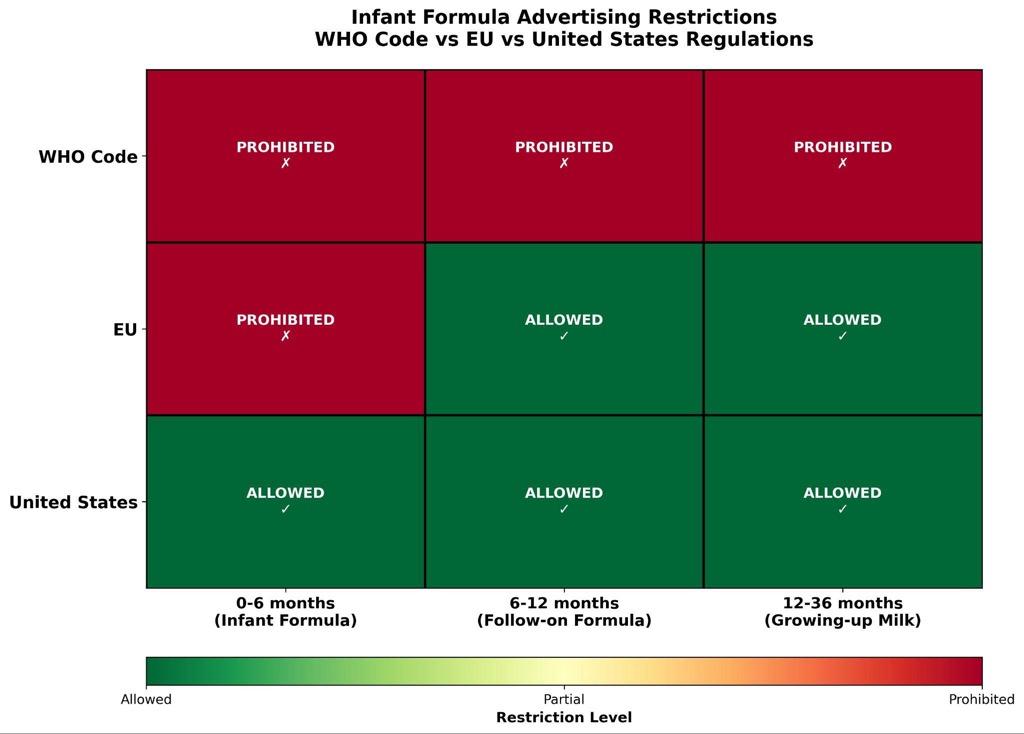

The US has not adopted the WHO Code at the federal level, allowing unrestricted marketing of all formula categories and permitting health and nutrition claims that would be prohibited under the Code.34 Australia and New Zealand implemented a voluntary Marketing in Australia of Infant Formula (MAIF) Agreement that covers only some Code provisions as well.35 The following WHO Code provisions are completely missing from most national legal frameworks: (1) Prohibiting health and nutrition claims on follow-on formula, (2) Prohibiting health and nutrition claims on follow-on formula, (3) Prohibiting health professional sponsorship by formula companies, (4) Comprehensive bans on health facility promotion, (5) Distribution of company materials in healthcare settings.36
4) Insufficient Digital Marketing Controls
The regulatory gap is even more severe in the digital space. The WHO Code was written before the rapid uptake of social media and algorithmic targeting in recent years, and fewer than 20% of countries explicitly prohibit digital promotion of breastmilk substitutes.37 No country has comprehensive digital marketing restrictions to date. The EU Digital Services Act, for example, doesn’t address formula marketing specifically and doesn’t classify pregnancy status as “sensitive data,” meaning parenting data collection remains completely legal.38 Formula companies can thus aggressively exploit this vacuum via unrestrained digital marketing strategies, as previously mentioned.
5) Failures and Barriers in Social Policy
Finally, inadequate social policies on parental leave and workplace support make it difficult for mothers to rely on breastfeeding and introduce vulnerabilities that formula marketing exploits. In France, breastfeeding breaks of one hour per day are legally mandated but unpaid—a violation of the European Social Charter.39 The US is the only high-income country without federally mandated paid maternity leave, creating significant barriers to breastfeeding continuation.40 India provides 26 weeks of maternity leave but lacks enforcement mechanisms for workplace lactation support, particularly in informal sectors where most women work.41 Formula companies position their products as solutions to such policy failures, capitalizing on vulnerable populations such as lower-income mothers whose choices are particularly constrained.42
Ultimately, the combination of weak advertising regulations, poor enforcement, and inadequate social support policies creates conditions where corporate marketing fills the void left by government shortcomings across diverse national contexts. While some countries demonstrate that strong Code implementation is achievable, the global reg-
ulatory landscape remains fragmented and insufficient to protect infant health from commercial interests.
To illustrate how these regulatory gaps manifest in practice and how corporations adapt their strategies to different national contexts, the following case studies examine three distinct examples: France, where lobbying efforts are centered on indirect marketing through scientific and professional capture; Germany, where strong regulations on paper are not met with the same commitment for enforcement; and the US, where corporations capitalize on trade and digital marketing spaces to exert influence.
CASE STUDIES
Case 1: France
France shows how corporate influence can take advantage of existing social dynamics to normalize the use of infant formula without direct lobbying. Instead, companies rely on scientific and professional capture and cultural normalization to increase the sales of infant formula.
France has one of the lowest rates of breastfeeding initiation in the world. After birth, only 69% of infants were breastfed in the maternity ward; after one month, this number dropped to 54%, with only 35% exclusively breastfed, and after six months, only 19% of infants were breastfed.43–45 The rate of breastfeeding initiation has decreased in recent years, with the adjusted rate of exclusive breastfeeding initiation decreasing by 9.6% and the adjusted rate of any breastfeeding initiation decreasing by 4.5% between 2010 and 2016.46
Structural and cultural factors have amplified this effect. Short maternity leaves and low or unpaid paternity leave make breastfeeding infants for a long period of time difficult, forcing parents to rely on formula to feed their babies when they are working.47,48 France also does not often provide a robust education on maternal care and breastfeeding after childbirth, pushing mothers to rely on the seemingly simpler approach of using infant formula instead. Other reasons for not breastfeeding are cultural. One survey showed that 41% of French women found breastfeeding in public to be embarrassing; additionally, some mothers reported “[wanting their] body back after nine months of pregnancy.”49 These factors have resulted in a voluntary reluctance to breastfeed, with 64.3% of mothers who use infant formula doing so by choice.50
With an already high reliance on infant formula, companies need not invest in direct lobbying efforts to increase the use of infant formula in France. Instead, they use commu-
nity-centric approaches to further normalize and promote infant formula use. A corporate political activity analysis of the baby food industry in France shows that manufacturers establish relationships with health-centric community groups, portraying themselves as experts in child nutrition and building alliances in civic society.51 Gallia, a baby food company, has sponsored events for SOS Préma, an organization advising parents of premature babies; Nestlé has partnered with the French Red Cross for a program to prevent early childhood obesity; Danone and Nestlé have both donated their products to charities in response to COVID-19.51 Through these and similar efforts, corporations have entrenched themselves into French society, establishing trust to make mothers more likely to buy their products.
Beyond community-level normalization, the infant formula industry has taken strides to cement infant formula as an alternative to breastfeeding in academia and healthcare: Danone established the Danone Institute to promote nutrition research with funding and prizes to pediatric researchers. Nestlé funded research on, and provided participants for, a survey on the relationship between doctors and parents in the context of infant feeding. Nestlé also invited medical students to seminars at their headquarters, where they aggressively promoted their products to future pediatricians. Professional associations such as the French Society of Perinatal Medicine and the Association of Juniors in Pediatrics have entered partnerships with multinational corporations for funding.51 Corporations then communicate potentially biased scientific findings to the public. Danone and Bledina formed the Grand Forum des Tout-Petits (Big Forum for Little Ones), a non-profit organization aimed at dispelling doubts about their products. Gathering experts and hosting annual conferences, the group pushes narratives on the importance of their products for infant health by educating mothers.51 These trends are not specific to the infant formula industry. Separate analyses on the corporate political activities of the food industry and dairy industry in France show that the majority of actions taken by companies like Nestlé to promote infant formula center around constituency building through community involvement and managing information by shaping the existing evidence base and reframing the narrative on relevant issues.52,53 The ubiquity of indirect influence across industries in France demonstrates the importance of addressing community- and information-focused practices.
Case 2: Germany
Lessons from Germany show that transparency alone is insufficient to curb harmful corporate behavior. Germany is often seen as a high-regulation country, with EU directives governing formula composition and safety standards, strict labeling requirements, a National Strategy for the Promotion of Breastfeeding, and a federal lobbying register.54,55 These frameworks establish what companies must do with respect
to product content and mandatory information, but say little about the manner in which formulas may be promoted. In practice, weak enforcement and heavy reliance on voluntary compliance create a regulatory architecture where companies can meet formal legal requirements while still engaging in practices the WHO discourages. German and EU rules set minimum standards for safety and labeling, but do not fully incorporate the WHO Code’s broader guidance to restrict marketing, leaving a large gap between legal compliance and Code compliance.
The Access to Nutrition Initiative (ATNI) found that none of the 153 breast-milk substitutes and complementary foods it assessed in Germany were fully Code-compliant; 100% of labels violated at least one provision, often through health and nutrition claims, suggesting that formula can match or replicate breastmilk, and cross-promotion.56 Experts in the German Society for Child and Adolescent Medicine have criticised such claims by companies as “unacceptable idealization,” noting, for example, that synthetic oligosaccharides add branding value but no proven clinical benefit over standard formula.57 Civil-society monitoring similarly highlights gaps in national Code monitoring, conflicts of interest, and the absence of effective sanctions.58,59
These governance challenges are reinforced by how industry influence is organized. Regulation concentrates heavily on product-level labeling and composition requirements, while digital marketing, branding strategies, and cross-promotion remain largely unregulated (Table 1). Formula manufacturers work through the Federal Association for Special Foods (DIÄTVERBAND), a trade association representing major infant-formula and medical-nutrition companies. Registered in the Bundestag’s lobbying database, DIÄTVERBAND reports substantial spending and engages systematically in consultations on food law, advertising, and digital marketing.54,60 Its submissions typically question WHO guidance as methodologically weak or contextually inappropriate, argue that existing rules are sufficient, and warn that tighter restrictions would limit “information access” for parents and health professionals.56 This framing encourages regulators to treat advertising as informational rather than promotional, which normalizes behaviors the WHO Code discourages.
Because enforcement bodies focus on checking narrow legal requirements rather than WHO Code standards, companies can use brand advertising, follow-on and toddler milks, digital channels, and bottle and teat marketing to build loyalty and normalize formula use.57,61 Germany thus exemplifies a “legalistic non-compliance” model: strong laws on paper, minimal penalties in practice, and corporate influence that is institutionalized through expert committees and trade associations rather than overt political lobbying.62
While lobbying registers and public strategies create the appearance of accountability, they do little without
Table 1
Covered under German/EU Regulation
Product label composition + nutrient standards
On-pack health and nutrition claim restrictions
Mandatory safety + preparation information
Packaging + labeling format requirements
Loopholes and Marketing Workarounds
Digital marketring campaigns (Instagram, parenting apps, influencers)
Brand-level promotion not tied to a specific product
Cross-promotion across product stages (infant, follow-on, toddler)
Advertising of follow-on and toddler milks as a proxy to promote infant formula
Table 1: Comparison of regulatory scope and marketing loopholes in Germany.
enforcement bodies empowered to monitor and penalize violations. Effective regulation must go beyond label text to include digital marketing, brand-level promotion, and cross-promotion across product lines. Furthermore, trade associations such as DIÄTVERBAND act as central nodes of influence, meaning that governing corporate lobbying requires not only disclosure but also rules addressing conflicts of interest, committee participation, and association-based representation. In short, transparency without teeth leaves underlying incentive structures intact and allows harmful practices to continue largely unchecked.
Case 3: The United States of America
The trade power and regulatory influence of the United States of America (US) influence global policy environments to reflect the commercial formula industry’s interests, particularly in LMIC. Formal political structures such as trade agreements and diplomatic negotiations are used by US trade entities and corporations to advance their interests, ultimately allowing US formula companies and trade officials to set global precedents that weaken breastfeeding protections worldwide.
Between 1996 and 2019, the US was the top filer of World Trade Organization (WTO) “interventions” challenging infant formula nutrition and labeling rules, often pressuring governments, particularly those of LMICs, to revise or repeal breastfeeding-protective policies.63 The US has filed nearly twice as many formula interventions as compared to Europe,
which works to frame breastfeeding-protective measures as restrictive overreach (Figure 6).63 The US effectively uses the WTO as a venue to discipline countries that try to implement stronger breastfeeding protections with the Code, reframing public health regulations as obstacles to commerce, which can deter governments, and especially LMICs, from proposing stronger laws. In practice, this helps shift the center of authority away from WHO guidance and Codex Alimentarius (the collection of international food standards and guidelines) to narrow the policy space available to countries that want to regulate infant formula marketing in the interests of child and maternal health.
This trade-based leverage is also paired with diplomatic pressures, where US officials can leverage their trade power, often in close alignment with major formula manufacturers such as Abbott and Mead Johnson. US delegations have pressured LMIC to dilute or drop WHA resolutions that call for stricter regulation of formal marketing. For example, US trade officials threatened to withdraw military aid from Ecuador in 2018 unless it denied a WHO breastfeeding resolution.64 US trade power places smaller nations at risk for economic consequences for adopting stronger breastfeeding protections.
Structural factors within the US magnify the extent of corporate overreach. The US remains the only high-income country without federally mandated paid maternity leave, and workplace protections for breastfeeding are inconsistent and under-enforced.65 Only 17 of 57 federal policy changes
Figure 6
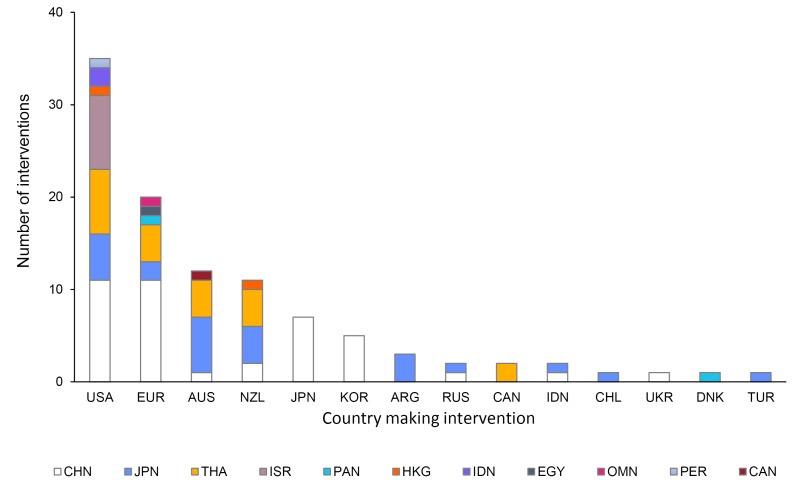
Figure 6: Countries Making WTO Interventions Involving Commercial Milk Formula from 1996-2019.63
between 2014 and 2023 aligned with core breastfeeding policy priorities, leaving major gaps in parental leave, lactation support, and public funding.66 These gaps create the conditions that incentivise formula use, particularly for low-income mothers.
Formula companies have capitalized on these dynamics through corporate political activity. Nestlé’s US operations, for example, used information management and direct engagement with policymakers to manipulate public narratives and research agendas around infant feeding.67 These strategies of funding formula organizations and framing formula as a technologically advanced solution mirror tactics across other food sectors in shaping evidence and public perspective. This broader environment of a “political economy of infant feeding” allows for commercial interests to influence policy and public health responsibilities. Just as trade power enables the US to control international standards, digital marketing allows formula companies to influence parental decision-making domestically, reinforcing corporate narratives about infant feeding.
Digital marketing creates the intersection for the structural barriers and trade dynamics to translate into environments for parents to engage with daily. In the US, this happens through e-commerce platforms: A content analysis of the 26 top-selling infant formula products on Amazon, Target, and Walmart found that 0% complied with WHO
Code guidelines through the use of idealizing imagery, unsubstantiated health claims, and narrative themes such as purity and innovation to frame formula as superior to breastfeeding.68 These techniques were amplified by the rapid shift toward digital media. A global review of the Code implementation shows that since 2016, infant formula promotion has moved toward social media and parenting apps, with widespread advertising of lightly regulated products like infant milks that fall outside of national laws.69 Within the US, online baby-product sales grew over 13% per year from 2018-2023, and the Code-aligned legislation is largely absent to allow for a highly profitable, poorly regulated online marketplace for formula companies to target parents’ pain points, such as infant sleep and the ability to return to work.69 These messages work to pose formulas as products with superior health and reliability, regardless of approved FDA evidence.
At the same time, digital markets reinforce formula use as personal choice rather than the failures of structural inequities, such as workplace protections. Infant formula lobbying and the elimination of breastfeeding protections are not solely legislative issues; they are fundamentally interconnected within global regulation and trade. Therefore, national policy is not in isolation when it comes to multinational corporations, since these global standards set by the US and its trading partners are interconnected.
POLICY RECOMMENDATIONS
From this comparative analysis, we offer three sets of recommendations to improve breastfeeding rates in light of disproportionate corporate influence:
1. Classify breastfeeding as care labor and provide financial support
2. Recognize breastmilk within food security frameworks
3. Fund independent research on breastfeeding accessibility, counseling, and health outcomes
1) Classify breastfeeding as care labor and provide financial support
To combat corporate influence and increase breastfeeding rates, governments should classify breastfeeding and breastmilk production as a public health good and a critical form of care labor. Specifically, governments can (1) create or expand economic accounting systems to pursue data collection on breastfeeding and breastmilk production as unpaid care labor and (2) reform labor laws and financial welfare programs to support the practice of breastfeeding.
Infant formula advertising capitalizes on the lack of recognition and support for breastfeeding as a form of gendered care work.70 This lack of recognition is an urgent reminder of the gaps between knowledge production, policymaking, and public health practice. To close these gaps, governments should invest in making the financial- and labor-related barriers to breastfeeding more legible. The global “care movement” has called for the acknowledgement of care work in the labor market as a matter of gender and economic justice.71
The European Union and the US have developed survey-based accounting programs that recognize work in the “informal” care economy using analytical methods such as time-use analysis.72 These interventions should index breastfeeding and breastmilk production in particular to situate the practice within unpaid care work and allow for more detailed descriptive studies and more effective policymaking.
A better understanding of breastfeeding within labor economics will allow for informed parental leave labor regulations, maternal-child welfare programs, and campaigns to compensate informal care work. Multi-country studies show that parental leave durations have a strong correlation with breastfeeding initiation and duration.9 Beyond maternity leave in particular, a growing area of research at the intersec-
tion of welfare economics and public health suggests that financial support for breastfeeding is effective in increasing breastfeeding rates.73 At a broader level, the practice and its challenges should be acknowledged and prioritized in campaigns to compensate unpaid and often gendered care labor.
2) Recognize breastmilk within food security frameworks
Countries should recognize breastmilk as an important component of food security and monitor accordingly. Both EU countries and the US frame breastfeeding as an individual, private practice recommended for its benefits to maternal and infant health, but frame infant formula as a public health good distributed by government programs.74 This distinction makes infant formula provision a more visible site of public health intervention than breastfeeding promotion.74 Governments, as well as NGOs and academic researchers, can level the playing field by integrating breastmilk into food surveillance networks at the national, regional, and global levels. In the US, the national Pregnancy Risk Assessment Monitoring System (PRAMS) provides data on breastfeeding rates and early cessation of breastfeeding, although state participation is uneven.75 Governments should pursue standardized metrics integrated within broader food security monitoring.
3) Fund independent research on breastfeeding accessibility, counseling, and health outcomes
While economic reform is crucial to classify breastfeeding and breastmilk production as a public health good, such reform cannot exist in a vacuum. Rather, there must be further financial investment into independent research on breastfeeding accessibility, counseling services, and health outcomes. Research-informed activities can identify local barriers to breastfeeding that may impede women from feeling safe, comfortable, and able to breastfeed for extended periods.76 Such efforts may include offering employers resources to comply with government regulations, increasing the number of lactation support providers in communities with low breastfeeding rates, and surveying existing counseling services that are available to families.76 However, this research must be funded by independent sources instead of corporations that have a financial stake in the expanded use of infant formula. Bias within scientific research, particularly in the form of conflicts of interest, continues to fuel the bil-
lion-dollar infant formula industry.77 Trials funded by the industry are more likely to produce results in favor of the sponsor, causing a detriment to patient care.77 International guidelines for primary care have been funded by formula manufacturers, with the majority of authors declaring interests with formula manufacturers in recent years.77 The need for unbiased research in scientific literature extends beyond basic science research to include studies that identify societal barriers to breastfeeding. Taken together with the economic reforms described previously, independent research to assess breastfeeding accessibility, counseling services, and health outcomes will mitigate the lobbying power and influence of infant formula manufacturers.
CONCLUSION
Across France, Germany, and the US, our analysis shows that infant formula companies exert influence
not through a single channel, but through an interconnected system of political, scientific, digital, and cultural tactics that influence both individual decision-making and national policy environments. Despite the WHO Code’s longstanding guidance, gaps in enforcement, transparency, and social policy have allowed commercial interests to normalize formula use and undermine breastfeeding protections across diverse contexts.
Strengthening breastfeeding outcomes will require governments to move beyond narrow product-level regulation toward a holistic approach that addresses the political economy of infant feeding. Recognizing breastfeeding as care labor, integrating breastmilk into food security networks, and funding independent research represent necessary steps to reorient public policy around maternal and child health rather than corporate priorities (Figure 7). Taken together, these reforms can help ensure that families make feeding decisions based on unbiased information, meaningful support, and structural conditions that promote rather than constrain healthy infant feeding practices.

HOW TO CITE
Bojja B*1, Duan Z*1, Maroun G*1, Neumeyer E*1, Potter-Schwartz L*1, Ricketts D*1, Storino A*1, Benamouzig B2, Fallon CK1, Panter-Brick C1 (2026). Corporate Influence in the Infant Formula Industry: A Comparative Analysis and Policy Recommendations. Policy Brief, Global Health Studies Program, Jackson School of Global Affairs, Yale University.
*Co-first authors
1 Jackson School of Global Affairs, Global Health Studies Program
2 Centre for the Sociology of Organisations, Sciences Po, Paris, France
REFERENCES
1. Barbhuiya, T. (n.d.). A Close-Up Shot of a Scooped Formula Milk Powder [Photograph]. https://www.pexels.com/ photo/a-close-up-shot-of-a-scooped-formula-milk-powder-11501481/
2. Stevens, E. E., Patrick, T. E., & Pickler, R. (2009). A History of Infant Feeding. The Journal of Perinatal Education, 18(2), 32–39. https://doi.org/10.1624/105812409X426314
3. Baker, P., Russ, K., Kang, M., Santos, T. M., Neves, P. A. R., Smith, J., Kingston, G., Mialon, M., Lawrence, M., Wood, B., Moodie, R., Clark, D., Sievert, K., Boatwright, M., & McCoy, D. (2021). Globalization, first-foods systems transformations and corporate power: A synthesis of literature and data on the market and political practices of the transnational baby food industry. Globalization and Health, 17, 58. https://doi.org/10.1186/s12992-021-00708-1
4. Lee, R. (2019). Commodifying Compassion: Affective Economies of Human Milk Exchange. International Journal of Feminist Approaches to Bioethics, 12(2), 92–116. https://doi.org/10.3138/ijfab.12.2.06
5. Weltgesundheitsorganisation (Ed.). (1981). International code of marketing of breastmilk substitutes. World Health Organization.
6. Gribble, K. D., Smith, J. P., Gammeltoft, T., Ulep, V., Van Esterik, P., Craig, L., Pereira-Kotze, C., Chopra, D., Siregar, A. Y. M., Hajizadeh, M., & Mathisen, R. (2023). Breastfeeding and infant care as ‘sexed’ care work: Reconsideration of the three Rs to enable women’s rights, economic empowerment, nutrition and health. Frontiers in Public Health, 11 https://doi.org/10.3389/fpubh.2023.1181229
7. Stordal, B. (2023). Breastfeeding reduces the risk of breast cancer: A call for action in high-income countries with low rates of breastfeeding. Cancer Medicine, 12(4), 4616–4625. https://doi.org/10.1002/cam4.5288
8. Bognar, Z., De Luca, D., Domellöf, M., Hadjipanayis, A., Haffner, D., Johnson, M., Kolacek, S., Koletzko, B., Saenz de Pipaon, M., Shingadia, D., Tissieres, P., Titomanlio, L., Topaloglu, R., & Trück, J. (2020). Promoting Breastfeeding and Interaction of Pediatric Associations With Providers of Nutritional Products. Frontiers in Pediatrics, 8. https://doi. org/10.3389/fped.2020.562870
9. Colen, C. G., & Ramey, D. M. (2014). Is Breast Truly Best? Estimating the Effect of Breastfeeding on Long-term Child Wellbeing in the United States Using Sibling Comparisons. Social Science & Medicine (1982), 109, 55–65. https://doi. org/10.1016/j.socscimed.2014.01.027
10. The commercial determinants of health—ClinicalKey. (n.d.). Retrieved December 10, 2025, from https://www. clinicalkey.com/#!/content/playContent/1-s2.0-S2214109X16302170?returnurl= https:%2F%2Flinkinghub.elsevier. com%2Fretrieve%2Fpii%2FS2214109X16302170%3Fshowall%3Dtrue&referrer=
11. Infant Nutrition Market Analysis | Industry Trends, Size & Forecast Report, 2030. (2025, June 26). Mordor Intelligence. https://www.mordorintelligence.com/industry-reports/infant-nutrition-market
12. Globalization, first-foods systems transformations and corporate power: A synthesis of literature and data on the market and political practices of the transnational baby food industry | Globalization and Health. (n.d.). Retrieved December 10, 2025, from https://link.springer.com/article/10.1186/s12992-021-00708-1#ref-CR42
13. Granheim, S. I., Engelhardt, K., Rundall, P., Bialous, S., Iellamo, A., & Margetts, B. (2017). Interference in public health policy: Examples of how the baby food industry uses tobacco industry tactics. World Nutrition, 8(2), 288–310. https://doi.org/10.26596/wn.201782288-310
14. Selling second best: How infant formula marketing works | Globalization and Health. (n.d.). Retrieved December 10, 2025, from https://link.springer.com/article/10.1186/s12992-020-00597-w
15. How the marketing of formula milk influences our decisions on infant feeding. (n.d.). Retrieved November 28, 2025, from https://www.who.int/publications/i/item/9789240044609
16. Scope and impact of digital marketing strategies for promoting breastmilk substitutes. (n.d.). Retrieved November 28, 2025, from https://www.who.int/publications/i/item/9789240046085
17. Werner, K. M., Mercurio, M. R., Shabanova, V., Hull, S. C., & Taylor, S. N. (2023). Pediatricians’ Reports of Interaction with Infant Formula Companies. Breastfeeding Medicine, 18(3), 219–225. https://doi.org/10.1089/ bfm.2022.0217
18. Rollins, N., Piwoz, E., Baker, P., Kingston, G., Mabaso, K. M., McCoy, D., Neves, P. A. R., Pérez-Escamilla, R., Richter, L., Russ, K., Sen, G., Tomori, C., Victora, C. G., Zambrano, P., & Hastings, G. (2023). Marketing of commercial milk formula: A system to capture parents, communities, science, and policy. The Lancet, 401(10375), 486–502. https://doi.org/10.1016/S0140-6736(22)01931-6
19. Helfer, B., Henke-Ciążyńska, K., & Boyle, R. J. (2025). Commercial influences on infant and young child feeding. Future Healthcare Journal, 12(2), 100264. https://doi.org/10.1016/j.fhj.2025.100264
20. Helfer, B., Leonardi-Bee, J., Mundell, A., Parr, C., Ierodiakonou, D., Garcia-Larsen, V., Kroeger, C. M., Dai, Z., Man, A., Jobson, J., Dewji, F., Kunc, M., Bero, L., & Boyle, R. J. (2021). Conduct and reporting of formula milk trials: Systematic review. The BMJ, 375, n2202. https://doi.org/10.1136/bmj.n2202
21. European Union. (n.d.). Transparency register. European Commission. https://transparency-register.europa.eu/index_en
22. Specialised Nutrition Europe. (2023). EU Transparency Register annual report https://ec.europa.eu/ transparencyregister
23. Nestor, R., & Valentin, M. (2022). Transparency without accountability: The structural limitations of the EU Transparency Register. European Journal of Public Policy, 29(4), 612–628.
24. Greer, S. L., & Löblová, O. (2021). Informal lobbying in the European Parliament: How unregistered influence shapes health policy. Health Policy, 125(6), 743–751.
25. Corporate Europe Observatory. (2020). Nestlé’s lobbying correction triggers €47.7 million register-wide audit. CEO Publications.
26. U.S. Food and Drug Administration. (2024, February 11). Regulations and information on the manufacture and distribution of infant formula https://www.fda.gov/food/infant-formula-guidance-documents-regulatory-information/ regulations-and-information-manufacture-and-distribution-infant-formula
27. Yale School of Public Health. (2018). India’s Infant Milk Substitutes Act, monitoring, and enforcement. Yale University. https://ysph.yale.edu/news-article/indias-infant-milk-substitutes-act-monitoring-and-enforcement/
28. Norwegian Directorate of Health. (2022). Breastfeeding statistics and Code implementation in Norway. Norwegian Health Authority.
29. Rollins, N. C., Bhandari, N., Hajeebhoy, N., Horton, S., Lutter, C. K., Martines, J. C., & Victora, C. G. (2016). Why invest, and what it will take to improve breastfeeding practices? The Lancet, 387(10017), 491–504.
30. World Health Organization. (2016). Guidance on ending the inappropriate promotion of foods for infants and young children. WHO Press.
31. Changing Markets Foundation. (2019). How formula companies lobby across Europe: Undermining breastfeeding and overriding public health policy. Changing Markets Foundation.
32. Berry, N. J., Jones, S. C., & Iverson, D. (2012). Toddler milk advertising in Australia: Infant formula advertising in disguise? Australasian Marketing Journal, 20(1), 24–27.
33. Pereira, C., Ford, R., Feeley, A. B., Sweet, L., Badham, J., & Zehner, E. (2016). Cross-sectional survey shows that follow-up formula and growing-up milks are labelled similarly to infant formula in four low and middle income countries. Maternal & Child Nutrition, 12(S2), 91–105. https://doi.org/10.1111/mcn.12269
34. Soldavini, J., & Taillie, L. S. (2017). Recommendations for Adopting the International Code of Marketing of Breastmilk Substitutes Into U.S. Policy. Journal of Human Lactation : Official Journal of International Lactation Consultant Association, 33(3), 582–587. https://doi.org/10.1177/0890334417703063
35. Australian Government Department of Health and Aged Care. (2025). Marketing infant formula in Australia https:// www.health.gov.au/topics/pregnancy-birth-and-baby/breastfeeding-infant-nutrition/marketing-infant-formula
36. World Health Organization, United Nations Children’s Fund, & International Baby Food Action Network. (2022). Marketing of breast-milk substitutes: National implementation of the international code, status report 2022. WHO.
37. Hastings, G., Angus, K., Eadie, D., & Hunt, K. (2020). Selling second best: How infant formula marketing works. Globalization and Health, 16(1), 1–12.
38. Regulation (EU) 2022/2065 of the European Parliament and of the Council on a Single Market For Digital Services (Digital Services Act), L 277/1 Official Journal of the European Union ___ (2022).
39. Victora, C. G., Bahl, R., Barros, A. J., França, G. V., Horton, S., Krasevec, J., & Rollins, N. C. (2016). Breastfeeding in the 21st century: Epidemiology, mechanisms, and lifelong effect. The Lancet, 387(10017), 475–490.
40. Livingston, G., & Thomas, D. (2019, December 16). Among 41 countries, only U.S. lacks paid parental leave. Pew Research Center. https://www.pewresearch.org/short-reads/2019/12/16/u-s-lacks-mandated-paid-parental-leave/
41. Ramesh, N., Siddaiah, A., & Joseph, B. (2023). Promoting Workplace Breastfeeding Practices. Indian Journal of Occupational and Environmental Medicine, 27(4), 275–277. https://doi.org/10.4103/ijoem.ijoem_309_23
42. Abrahams, S. W. (2021). Milk and social media: Online cultures of infant feeding. Social Science & Medicine, 293, 114691.
43. Taux d’allaitement maternel à la maternité et au premier mois de l’enfant. Résultats de l’étude Épifane, France, 2012. (2012). Journal de Pédiatrie et de Puériculture, 25(6), 364–372. https://doi.org/10.1016/j.jpp.2012.09.008
44. Wagner, S., Kersuzan, C., Gojard, S., Tichit, C., Nicklaus, S., Geay, B., Humeau, P., Thierry, X., Charles, M.-A., Lioret, S., & de Lauzon-Guillain, B. (2015). Durée de l’allaitement en France selon les caractéristiques des parents et de la naissance. Résultats de l’étude longitudinale française Elfe, 2011. Bulletin Epidémiologique Hebdomadaire, 29, 522–532.
45. Trends in breastfeeding practices and mothers’ experience in the French NutriNet-Santé cohort | International Breastfeeding Journal. (n.d.). Retrieved November 29, 2025, from https://link.springer.com/article/10.1186/s13006021-00397-x
46. Guajardo-Villar, A., Demiguel, V., Smaïli, S., Boudet-Berquier, J., Pilkington, H., Blondel, B., Salanave, B., Regnault, N., & Pelat, C. (2022). Trends and disparities in breastfeeding initiation in France between 2010 and 2016: Results from the French National Perinatal Surveys. Maternal & Child Nutrition, 18(4), e13410. https://doi.org/10.1111/ mcn.13410
47. Maternal employment characteristics as a structural social determinant of breastfeeding duration—PMC. (n.d.). Retrieved November 29, 2025, from https://pmc.ncbi.nlm.nih.gov/articles/PMC11516972/
48. The influence of family policies on fertility in France. (n.d.). Retrieved November 29, 2025, from https://www.un.org/ development/desa/pd/sites/www.un.org.development.desa.pd/files/undp_egm_201511_policy_brief_no._7.pdf
49. Administrator, W. A. F. (2016, October 13). The Stigma of Breastfeeding in France. Women Across Frontiers Magazine. https://wafmag.org/2016/10/stigma-breastfeeding-france/
50. Courtois, F., Péneau, S., Salanave, B., Andreeva, V. A., Roland-Cachera, M. F., Touvier, M., Galan, P., Hercberg, S., & Fezeu, L. K. (2021). Trends in breastfeeding practices and mothers’ experience in the French NutriNet-Santé cohort. International Breastfeeding Journal, 16, 50. https://doi.org/10.1186/s13006-021-00397-x
51. Cossez, E., Baker, P., & Mialon, M. (2022). ‘The second mother’: How the baby food industry captures science, health professions and civil society in France. Maternal & Child Nutrition, 18(2), e13301. https://doi.org/10.1111/mcn.13301
52. Corporate political activity of the dairy industry in France: An analysis of publicly available information | Public Health Nutrition | Cambridge Core. (n.d.). Retrieved November 29, 2025, from https://www.cambridge.org/core/ journals/public-health-nutrition/article/corporate-political-activity-of-the-dairy-industry-in-france-an-analysis-ofpublicly-available-information/400B6107C8859B9E8B62110E0B5D5EF6
53. Mialon, M., & Mialon, J. (2018). Analysis of corporate political activity strategies of the food industry: Evidence from France. Public Health Nutrition, 21(18), 3407–3421. https://doi.org/10.1017/S1368980018001763
54. Consolidated version of the Lobbying Register Act. (n.d.). German Bundestag. https://www.bundestag.de/resource/ blob/991942/Konsolidierte-Fassung-LobbyRG-2024_EN.pdf
55. National Strategy for the Promotion of Breastfeeding. (2021). Federal Ministry of Food and Agriculture (BMEL).
56. Germany country study: Marketing of breast-milk substitutes and complementary foods. (2024). Access to Nutrition Initiative. https://accesstonutrition.org/app/uploads/2024/08/Germany-Country-Study-2024-Final.pdf
57. Bührer, C., Ensenauer, R., Jochum, F., Kalhoff, H., Koletzko, B., Lawrenz, B., Mihatsch, W., Posovszky, C., & Rudloff, S. (2022). Infant formulas with synthetic oligosaccharides and respective marketing practices: Position Statement of the German Society for Child and Adolescent Medicine e.V. (DGKJ), Commission for Nutrition. Molecular and Cellular Pediatrics, 9(1), 14. https://doi.org/10.1186/s40348-022-00146-y
58. Rouw, E., Hormann, E., & Scherbaum, V. (2015). The high cost of half-hearted breastfeeding promotion in Germany. International Breastfeeding Journal, 9(1), 22. https://doi.org/10.1186/s13006-014-0022-5
59. World Breastfeeding Trends Initiative—Assessment Report: Germany. (2018). International Baby Food Action Network (IBFAN) Asia & WBTi Germany team.
60. Lobbyregistereintrag “Bundesverband spezielle Lebensmittel (DIÄTVERBAND) e.V.” (n.d.). Lobbyregister beim Deutschen Bundestag. Retrieved December 12, 2025, from https://www.lobbyregister.bundestag.de/suche/R001228
61. Theurich, M. A., Ziebart, M., & Strobl, F. (2024). National survey of infant feeding bottles in Germany: Their characteristics and marketing claims. Maternal & Child Nutrition, 20(3), e13632. https://doi.org/10.1111/mcn.13632
62. Brettschneider, A.-K., Steindl, J., Matthes-Bolz, B., Lehmann, I., Hilger-Kolb, J., Roskosch, E., Strauß, G., Hoffmann, M., & Ensenauer, R. (2025). Development and results of the German National Strategy for the Promotion of Breastfeeding: A participatory process. Frontiers in Public Health, 13, 1555139. https://doi.org/10.3389/ fpubh.2025.1555139
63. Russ, K., Baker, P., Byrd, M., Kang, M., Siregar, R. N., Zahid, H., & McCoy, D. (2021). What You Don’t Know About the Codex Can Hurt You: How Trade Policy Trumps Global Health Governance in Infant and Young Child Nutrition. International Journal of Health Policy and Management, 10(12), 983–997. https://doi.org/10.34172/ijhpm.2021.109
64. Vogel, H. (2024, March 21). How the U.S. waged a global campaign against baby formula regulation. The Lund Report. https://www.thelundreport.org/content/how-us-waged-global-campaign-against-baby-formula-regulation
65. Hecht, H. K., Nguyen, A.-M. N., & Harley, K. G. (2024). Association of Maternity Leave Characteristics and Postpartum Depressive Symptoms among Women in New York. Maternal and Child Health Journal, 28(11), 1990–1999. https://doi.org/10.1007/s10995-024-03997-1
66. Harrigan, P. B., Schenk, T., Volpe, S. L., Hedrick, V. E., Khan, T., & Misyak, S. A. (2025). A comparison of U.S. infant feeding policy changes to Global Breastfeeding Collective policy priorities. Frontiers in Public Health, 13. https://doi. org/10.3389/fpubh.2025.1653377
67. Tanrikulu, H., Neri, D., Robertson, A., & Mialon, M. (2020). Corporate political activity of the baby food industry: The example of Nestlé in the United States of America. International Breastfeeding Journal, 15(1), 22. https://doi. org/10.1186/s13006-020-00268-x
68. Froley, S., Watkins, C., & Tomori, C. (2025). Infant Formula in the Digital Age: How US Online Formula Marketing Targets Parents. Maternal & Child Nutrition, 21(3), e70034. https://doi.org/10.1111/mcn.70034
69. Topothai, C., Cetthakrikul, N., Howard, N., Tangcharoensathien, V., Chong, M. F.-F., & van der Eijk, Y. (2024). Outcomes of implementing the International Code of Marketing of Breast-milk Substitutes as national laws: A systematic review. International Breastfeeding Journal, 19(1), 68. https://doi.org/10.1186/s13006-024-00676-3
70. Pérez-Escamilla, R., Tomori, C., Hernández-Cordero, S., Baker, P., Barros, A. J. D., Bégin, F., Chapman, D. J., Grummer-Strawn, L. M., McCoy, D., Menon, P., Neves, P. A. R., Piwoz, E., Rollins, N., Victora, C. G., & Richter, L. (2023). Breastfeeding: Crucially important, but increasingly challenged in a market-driven world. The Lancet, 401(10375), 472–485. https://doi.org/10.1016/S0140-6736(22)01932-8
71. Zhou, M., Wu, Y., & Kan, M. (2025). Using Time Use Data to Investigate Gender Inequalities: Enduring Patterns and New Dimensions. Sociology Compass, 19(11), e70132. https://doi.org/10.1111/soc4.70132
72. Nivakoski, S., Baggio, M., & European Foundation for the Improvement of Living and Working Conditions (Eds.). (2025). Unpaid care in the EU. Publications Office. https://doi.org/10.2806/5387374
73. Hoskins, K., & Schmidt, H. (2021). Breastfeeding, Personal Responsibility and Financial Incentives. Public Health Ethics, 14(3), 233–241. https://doi.org/10.1093/phe/phab020
74. Baker, P., Smith, J. P., Garde, A., Grummer-Strawn, L. M., Wood, B., Sen, G., Hastings, G., Pérez-Escamilla, R., Ling, C. Y., Rollins, N., & McCoy, D. (2023). The political economy of infant and young child feeding: Confronting corporate power, overcoming structural barriers, and accelerating progress. The Lancet, 401(10375), 503–524. https:// doi.org/10.1016/S0140-6736(22)01933-X
75. Dinour, L. M., Rodas, E. I. R., Amutah-Onukagha, N. N., & Doamekpor, L. A. (2020). The role of prenatal food insecurity on breastfeeding behaviors: Findings from the United States pregnancy risk assessment monitoring system. International Breastfeeding Journal, 15(1), 30. https://doi.org/10.1186/s13006-020-00276-x
76. CDC. (2025, May 16). Strategies for Continuity of Care in Breastfeeding. Breastfeeding. https://www.cdc.gov/ breastfeeding/php/strategies/public-health-strategies-for-continuity-of-care-in-breastfeeding.html
77. Overdiagnosis and industry influence: How cow’s milk protein allergy is extending the reach of infant formula manufacturers | The BMJ. (n.d.). Retrieved December 10, 2025, from https://www.bmj.com/content/363/bmj.k5056
78. High Angle Portrait of a Cute Asian Baby Feeding from the Breast of Her Mother. Intimate Moment Between New Mother and Infant Showing Motherly Love, Tenderness, and Unconditional Affection. (2023). [Photograph]. https:// www.istockphoto.com/photo/high-angle-portrait-of-a-cute-asian-baby-feeding-from-the-breast-of-her-mothergm1514034392-524127912?utm_source=pexels&utm_medium=affiliate&utm_campaign=sponsored_photo&utm_ content=srp_inline_portrait_media&utm_term=breastfeeding
