#MoreThanMyeloma
2026 Myeloma Action Month global campaign inspired over 60 million to show their support of the myeloma community




ALSO IN THIS EDITION: The remarkable progress in myeloma clinical trials and treatment options for today’s patients confirm that we are in an era of the new “normal”
Message from the IMF President & CEO
Dear Members of the Myeloma Community,
As we move into spring, a season that reminds us of renewal and progress, I find myself reflecting on the extraordinary momentum across the myeloma community. This year’s Myeloma Action Month was a powerful example of that momentum in action. Around the world, patients, care partners, healthcare professionals, and advocates came together to raise awareness, share their stories, and demonstrate the spirit of being #MoreThanMyeloma.
Myeloma Action Month is more than a campaign – it is a global movement that reflects the strength and determination of the myeloma community. At the IMF, we are driven by this momentum to continue advancing research, expanding education, and advocating for equitable access to care until no one faces myeloma alone. The energy, visibility, and engagement we saw throughout the month of March were truly inspiring and a testament to what our community can accomplish together.
At the same time, scientific and clinical progress in the field of myeloma continues at a remarkable pace. Recent FDA approvals and advances in immunotherapy, including

CAR T-cell therapies and bispecific antibodies, are expanding treatment options and helping many patients achieve deeper and more durable responses than ever before. These breakthroughs are reshaping what is possible in myeloma care and bringing new hope to patients and families worldwide.
Yet we must also acknowledge the challenges that remain. Myeloma is still considered an incurable disease, and many patients will face relapse. Disparities in diagnosis, access to cutting-edge therapies, and treatment outcomes continue to affect underserved communities, underscoring the importance of continued research, education, and advocacy.

Even so, the trajectory of progress has never been more encouraging. Each clinical trial, each new therapy, and each scientific discovery moves us closer to our ultimate goal: preventing myeloma and finding a cure.
The International Myeloma Foundation remains deeply committed to driving this progress – through research collaborations, global physician education, patient and care partner support, and advocacy efforts that ensure every patient has access to the best possible care. Thanks to the dedication of the members of this incredible myeloma community, we are moving forward together with determination, urgency, and hope.
With gratitude and optimism,
Heather Cooper Ortner

The DVRd Regimen
This
standard-of-care quadruplet
combination is now FDA-approved for the majority of patients with newly diagnosed multiple myeloma
On January 27, 2026, the U.S. Food and Drug Administration (FDA) approved the use of the quadruplet (4-drug) combination of Darzalex Faspro® (daratumumab + hyaluronidase-fihj) + Velcade® (bortezomib) + Revlimid® (lenalidomide) + dexamethasone [DVRd] for adult patients with newly diagnosed multiple myeloma (NDMM) who are not eligible for an autologous stem cell transplant (ASCT). DVRd, a highly effective and well-tolerated combination therapy for patients with myeloma, was previously approved by the FDA in 2024 for patients who are eligible for ASCT.
The drugs in the DVRd regimen
Although it may seem that being treated with 4 drugs is a lot of therapy, each medication in the DVRd regimen is from a different drug class, each with its own way of attacking myeloma while enhancing the activity of the other drugs:
Darzalex Faspro, a newer formulation of Darzalex® (daratumumab), is a monoclonal antibody that targets and binds to the CD38 protein on the surface of myeloma cells and enlists the cells of the immune system to help attack and kill myeloma cells. Darzalex Faspro is given as a subcutaneous (SQ) injection under the skin of the abdomen at a doctor’s office in 3 to 5 minutes, while it also reduces the rate of an infusion-related reaction (IRR) to 13% vs. 34% with the original Darzalex intravenous (IV) formulation.
Velcade is a proteasome inhibitor that interferes with the normal function of a joined group (“complex”) of enzymes (“proteases”), causing damaged and unwanted proteins to build up in the myeloma cell, causing myeloma cells to die.
Revlimid is an immunomodulatory agent that can modify, enhance, or suppress the functioning of the immune system. It helps the cells of the immune system to recognize and destroy myeloma cells.
Dexamethasone is a steroid that can kill myeloma cells directly, increase the efficacy of other drugs used to treat myeloma, inhibit the survival of myeloma cells, and block the release of cytokines that promote myeloma cell growth.
It is because these four drugs work so well together that DVRd can lead to such good outcomes for many patients.
CEPHEUS clinical trial of DVRd
The FDA expanded approval of DVRd in 2026 was based on data from the CEPHEUS clinical trial in patients with NDMM who were ineligible for ASCT or who declined ASCT as frontline therapy, the initial treatment used in an effort to achieve response in a newly diagnosed patient. Of the 395 study patients, 197 were randomized to the DVRd arm and 198 to the VRd arm without Darzalex Faspro.
MRD and PFS efficacy measures
The CEPHEUS clinical trial demonstrated significant efficacy as measured by overall minimal residual disease (MRD)-negativity and progression-free survival (PFS).
MRD is the presence of residual cancer cells after treatment has been completed and complete response (CR) has been attained. MRD-negativity means that not even 1 myeloma cell was found in 100,000 or 1,000,000 sampled bone marrow plasma cells (depending on the test being used). In the DVRd arm of the CEPHEUS study, the rate of MRD-negativity was 52.3% vs. 34.8% in the VRd arm, a significant difference.
PFS is the length of time during and after treatment that a patient lives with myeloma but the disease does not relapse. At 54 months, approximately 68% of study patients in the DVRd arm remained progression-free vs. approximately 48% in the VRd arm. DVRd demonstrated a 43% lower risk of progression or death when compared to the VRd arm of the study. Median PFS, the midpoint in a set of data, was not reached for patients in the DVRd arm, indicating superior, long-lasting, durable responses compared to patients in the VRd arm of the study.
What DVRd means for patients
The CEPHEUS study demonstrate that DVRd is a new standard of care, a benchmark treatment that is widely accepted by medical experts as the most appropriate for patients with NDMM who are not proceeding to transplant as frontline therapy.
Similarly, the PERSEUS clinical trial, which was the basis of the 2024 FDA approval of DVRd in transplant-eligible patients with NDMM, showed significantly improved PFS and increased depth of response (DpR), demonstrating that DVRd is a new standard of care for ASCT-eligible patients with NDMM.
“DVRd has increasingly become the standard-of-care regimen for the majority of the transplant-eligible and fit or intermediate-fit transplant-ineligible newly diagnosed myeloma patients, following the paradigm of picking the best therapies to give all patients the opportunity to achieve best responses and survival outcomes,” says Dr. Saad Z. Usmani, principal investigator of the CEPHEUS clinical trial, myeloma specialist and cellular therapist at Memorial Sloan Kettering Cancer Center in New York City, as well as a member of the IMF Scientific Advisory Board and the IMF International Myeloma Working Group. MT
STAY INFORMED! Contact the IMF InfoLine with your myeloma-related questions and concerns. Phone lines are open 9 a.m. to 4 p.m. (Pacific) Monday through Friday at 1.818.487.7455 or you can email us at InfoLine@myeloma.org or visit mmsm.link/infoline to schedule a convenient time to talk with an IMF InfoLine Coordinator.
By Dr. Joseph Mikhael IMF Medical Advisor
The Amazing New “Normal” in
The progress in myeloma treatment for our patients has been and continues to be remarkable. In the past two decades, more than 20 new drugs have been approved by the U.S. Food & Drug Administration (FDA) for patients with myeloma. The emergence of numerous forms of immunotherapy have produced unprecedented outcomes. And all the while a greater emphasis continues to be placed on a patient’s quality of life.
These advances are not abstract – they have truly changed the standard of care and improved patient prognosis. In fact, we now expect most people with myeloma to live longer than 10 years after diagnosis, and this is something we couldn’t say until recently. A huge part of the improvement in patient survival is related to how we treat myeloma when it is first diagnosed and then at first relapse.
The new “norm” of myeloma frontline therapy and treatment at first relapse has radically changed with five very important developments:
1. Treatment of high-risk smoldering multiple myeloma,
2. Quadruplet (4-drug) therapies for frontline treatment,
3. CAR T-cell therapy at first relapse,
4. Bispecific antibodies at first relapse, and
5. The reintroduction of antibody-drug conjugates.
Treatment of HR-SMM
Smoldering multiple myeloma (SMM) is divided into two groups: standard-risk and high-risk. There are several models that define high-risk SMM (HR-SMM). The most commonly used model is “20/2/20” risk stratification criteria published by Mayo Clinic in 2018, in which patients with two or more of the following three factors are considered high-risk:
Bone marrow plasma cell (BMPC) infiltration > 20%,
Serum monoclonal protein (M-protein) > 2 g/dL, and/or
Ratio of involved-to-uninvolved serum free light chain (sFLC) > 20.
In 2014, the SLiM-CRAB criteria was developed to detect active myeloma earlier, ideally before damage occurs and is felt by the patient. The SLiM-CRAB criteria includes the following features:
S – Sixty percent (60%) plasma cells present in the bone marrow,
Li – Light chains ratio of involved-to-uninvolved serum free light chains (sFLC) of 100 or more,
M – MRI imaging of 2 or more focal lesions in bone marrow,
C – Calcium elevation due to myeloma,
R – Renal (kidney) insufficiency due to myeloma,
A – Anemia (low red blood cell count) due to myeloma,
B – Bone disease related to myeloma.
But we have now gone much further by intervening before the onset of active myeloma with limited therapy for HR-SMM. The AQUILA clinical trial demonstrated that 3 years of Darzalex® (daratumumab) therapy in people with HR-SMM can delay the diagnosis of active myeloma and even improve the overall survival (OS). Indeed, people with HR-SMM now have a choice: they can consider monitoring the disease (“wait and watch”) or they can elect to be treated.
In an era where we seek to intercept all cancers earlier, this is a fundamental shift. I think it signals that in the future we will be able to prevent so much of the damage caused by myeloma when we treat it earlier.
Quadruplets in frontline therapy
Frontline therapy is the initial treatment used in an effort to achieve response in a patient with newly diagnosed multiple myeloma (NDMM). In the past, we used to treat newly diagnosed patients with the one or two drugs that were available at the time, and responses were few and of limited duration. As novel agents with different mechanisms of action were developed, it became the norm to use doublet (2-drug) or triplet (3-drug) treatment regimens. But now we are in the era of quadruplet (4-drug) therapies.
A monoclonal antibody such as Darzalex or Sarclisa® (isatuximab), a proteasome inhibitor such as Velcade® (bortezomib) or Kyprolis® (carfilzomib), an immunomodulatory drug such as Revlimid® (lenalidomide) and the steroid dexamethasone have become the new standard of care for most patients. This is as a result of several large clinical trials that have demonstrated, time and time again, the benefit of quadruplets helping patients achieve a deeper and more durable response, while maintaining a patient’s quality of life.
Currently, we are seeing most patients achieve and remain in remission for more than 5 years after their first line of therapy! We have also learned that HOW we use these drugs together to maximize their benefits and minimize their side effects by dosing them carefully and tapering them over time is of critical importance. Although “more is better” when it comes to using 4 drugs together, we also know that “less is more” when it comes to using less medication over time to keep patients in remission while reducing their chances of developing side effects. That modality of “enough but not too much” is guiding researchers and clinicians as we develop even more effective approaches to frontline therapy.
CAR T-cell therapy at first relapse
Chimeric antigen receptor (CAR) T-cell therapy is an immunotherapy that involves collecting the patient’s T cells and multiplying engineered T cells for re-infusion into the patient to attack the patient’s own myeloma cells.
Myeloma is Here for Today’s Patients!
CAR-T has truly revolutionized the way we treat myeloma. For most patients, no other therapy delivers response rates of over 90% or lasts more than 3 years the way CAR-T can. The long-term follow up of the CARTITUDE-1 clinical trial shows that 33% of patients were still in remission with no active disease or treatment 5 years later. Keep in mind that this group of patients was very heavily pretreated, with 6 prior lines of therapy on average.
We know that patient outcomes can improve when treatment is given earlier. Indeed, this is what’s happening with CAR-T, which is now approved by the FDA for use in myeloma as early as first relapse. As we continue to make this modality of treatment safer and more conveniently delivered, even more patients with myeloma will be able to receive CAR-T earlier in their therapy. Furthermore, after a single infusion of CAR-T, myeloma patients typically do NOT require maintenance therapy, and they truly can be off treatment. Personally, I love giving “nada ” therapy!
Bispecific antibodies at first relapse
Bispecific antibodies bind to two (“bi”) targeted cells, the myeloma cell and the T cell, and activating the T cells to release cytotoxic granules to kill the myeloma cells. Much like CAR T-cell therapy, we have been using bispecific antibodies in late relapse, only after myeloma patients have been treated with 4 prior lines of therapy.
As of March 2026, the new “norm” is the ability to give bispecific antibodies to patients as soon as their first relapse. Bispecific antibodies are remarkable, allowing us to engage a patient’s T cells without the need of collecting their cells and later re-infusing them. Bispecific antibodies are “off the shelf” drugs that can be delivered to patients without delay.
The FDA granted approval for the use of Tecvayli® (teclistamabcqyv) in combination with Darzalex Faspro® (daratumumab + hyaluronidase-fihj), called “Tec-Dara” for short, as a second-line treatment for patients with relapsed or refractory multiple myeloma (RRMM). Second-line treatment refers to the next treatment used after patients receive their initial frontline therapy. Incredibly, 83% of patients enrolled in the clinical trial who received treatment with Tec-Dara were still in remission at 3 years.
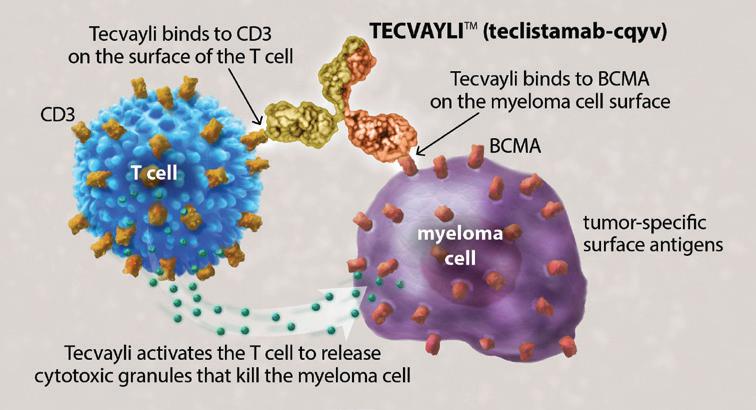
This is even more remarkable when you consider the comparator: the study patients in the group who received the typical triplet regimens had only 29% of patients in remission at 3 years. It is evident that immunotherapies are now becoming the new standard of care.
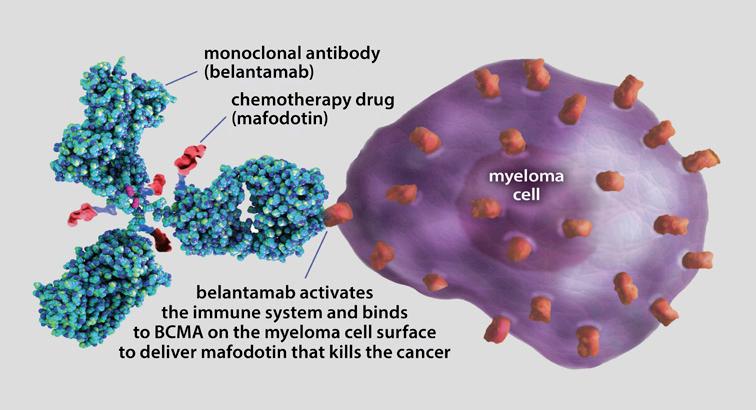
Antibody-drug conjugates
Blenrep® (belantamab mafodotin-blmf) is an antibody-drug conjugate (ADC). Blenrep was previously FDA-approved as a monotherapy in late relapse, before re-emerging as an option for earlier-line therapy in combination with either Velcade or Pomalyst® (pomalidomide). This unique treatment hooks on to the myeloma cell as an antibody but also delivers a toxin to enhance the destruction of myeloma cells. Blenrep is a very simple therapy to deliver, with most infusions given every 8–12 weeks. Side effects are controlled much better now than before. Blenrep offers yet another way to treat early relapse with a novel immunotherapy drug, giving patients even more choice.
In closing
It is genuinely hard to believe that these five significant developments have occurred so rapidly and that they have now become the new “norm.” I am so thankful for the many choices we can currently provide for our patients, and I am so enthusiastic about the newer approaches that are on their way. Stay tuned for news in smoldering myeloma, frontline therapy with CAR-T and bispecific antibodies, newer CAR-T agents and new bispecific antibodies –and even TRI-specifics, a new drug class of CELMoD agents, and much much more. The pace of research continues to accelerate; I am excited to see what the next new norm will be soon as we move closer and closer to a cure. MT
STAY INFORMED! Contact the IMF InfoLine with your myeloma-related questions and concerns. Phone lines are open 9 a.m. to 4 p.m. (Pacific) Monday through Friday at 1.818.487.7455 or you can email us at InfoLine@myeloma.org or visit mmsm.link/infoline to schedule a convenient time to talk with an IMF InfoLine Coordinator.
2026 MAM Global Campaign Demonstrates Over 60 million in 96 countries celebrate resilience,
By Peter Anton IMF Vice President, Marketing

As the 2026 Myeloma Action Month (MAM) annual global awareness campaign came to a close, the International Myeloma Foundation (IMF) was truly grateful for the incredible support that helped make the event a worldwide success – reaching over 60 million on social media across 96 countries!
Every March, the IMF leads the MAM patient and care partner advocacy-driven program that sparks action to raise awareness, promote education, build community, and create change to benefit people with myeloma. During MAM, the global myeloma community of patients, care partners, healthcare providers, and supporters come together to share personal stories, inspire action, and raise awareness of this little-known blood cancer.
Taking action
During this year’s Myeloma Action Month, the IMF encouraged members of the myeloma community to demonstrate their spirit through the #MoreThanMyeloma global social awareness campaign by sharing how they are living with and not for myeloma. More than 60 million people were reached on social media!
Beyond the numbers, this year’s #MoreThanMyeloma global campaign celebrated progress, resilience, and hope through shared posts on the MAM Wall of Action. This active participation helped raise awareness, created positive change, and encouraged action in the fight against myeloma.

Celebrating progress
According to IMF President & CEO Heather Cooper Ortner, “Myeloma Action Month is more than a campaign – it is a global movement that reflects the strength and determination of the myeloma community. The powerful engagement we’ve seen through #MoreThanMyeloma reinforces that patients are living full, meaningful lives beyond their diagnosis. At the IMF, we are driven by this momentum to continue advancing research, expanding education, and advocating for equitable access to care until no one faces myeloma alone.”
IMF Director for the Global Myeloma Action Network (GMAN) and European & Middle Eastern Patient Programs Serdar Erdoğan had this to say about the global campaign: “Behind every diagnosis is a life, a story, and a strength that goes far beyond the disease. It’s not just about surviving – it’s about living fully, being seen, and being heard. Progress lights the way, hope fuels the fight, and our lives shine brighter than any diagnosis. During Myeloma Action Month and World Myeloma Day (March 26), we stand for hope, resilience, and a future where every voice matters.”
Events throughout MAM 2026
Lighting the World Red
To signify the global signal of unity, progress, resilience, and hope, 252 major landmarks across 108 cities in 17 countries were lit in red to honor all people impacted by myeloma. In the U.S., oncology centers and MAM partners across the country illuminated their campuses in red to show support. Members of the myeloma community also participated by illuminating their own homes, offices, and community spaces.
Across the globe, Global Myeloma Action Network (GMAN) member organizations lit the world red all month long, culminating on


Demonstrates the Spirit of #MoreThanMyeloma
resilience, strengthen connections, and inspire hope
March 26th, in celebration of World Myeloma Day. Major landmarks across the globe – in Canada, South America, New Zealand, Australia, South Korea, and all over Europe participated to send a clear message: being in solidarity with those impacted by the disease and signifying that they are #MoreThanMyeloma. See which landmarks showed their support through the MAM Global Map on myelomaactionmonth.org, and see the front cover of this edition of Myeloma Today for an image from this event.
Facebook LIVE Q&A sessions
The IMF hosted Facebook LIVE Q&A events with key figures in the myeloma community, including healthcare professionals and educators. Expert insights were shared about myeloma treatments, side effect management, and living well with myeloma –and questions from participants were answered in real time.
Q&A with Dr. Joseph Mikhael and Dr. Melissa Alsina
Q&A with Dr. Sagar Lonial
You can replay both recordings at videos.myeloma.org by clicking the Facebook LIVE tab.
In this edition of Myeloma Today, Dr. Mikhael shares the remarkable progress in the treatment options available to today’s patients and Dr. Saad Usmani discusses one of the promising clinical trials that may further expand these options in the near future.
Boca Raton Patient & Family Seminar
On March 13–14, the IMF held its annual Patient & Family Seminar in Boca Raton, Florida. Sessions presented at this highly successful and well-attended event included Hot Topics in Myeloma; Myeloma 101: The Big Picture Perspective with Q&A; Navigating Insurance and Medical Bills; Myeloma 202: Immunotherapy Made Simple, Understanding Clinical Trials, and much more. Another

highlight was a Fireside Q&A Chat on What is the Future of Myeloma with Dr. Carl Ola Landgren and Dr. Joseph Mikhael. Patients, care partners, and family members were genuinely grateful for the learnings they gained from world-renowned myeloma experts while bonding with others in the myeloma community. Faculty presenters were equally excited about sharing the latest information and direct interaction with the myeloma community.
Miracles for Myeloma 5K Run/Walk
Myeloma Action Month is all about taking action, and we quite literally took strides for myeloma research with the IMF Miracles for Myeloma 5K Run/Walk, held on March 21 at Griffith Park in Los Angeles, California. The 5K Run/Walk encouraged everyone to participate in finding a cure for myeloma. The 5K Run/Walk brought together the local community to inspire, transform, and shift the narrative for patients and their loved ones – from a place of uncertainty to an embracing myeloma community, united in hope.
Many runners and walkers were there in person while others joined virtually from around the world. The IMF Miracles for Myeloma 5K Run/Walk raised more than $60,000 thanks to the incredible support from nearly 200 in-person and virtual participants, making it a special and collaborative global event. Please see the front cover of this edition of Myeloma Today for a photo of 5K Run/Walk participants.
The IMF extends its warmest thanks to the co-chairs of the Miracles for Myeloma 5K Run/Walk in Los Angeles: Dr. Murali Janakiram (Myeloma Specialist, City of Hope); Lani Beggs (8-year myeloma survivor); Donna Cunningham (Leader, San Fernando Valley Support Group); Malcolm Katz (Leader, San Fernando Valley Support Group and 17-year myeloma survivor); Todd & (continues on next page)


Diane Kennedy (Leaders, City of Hope-Duarte Support Group and 9-year myeloma survivor and care partner); Jackie Sarlitt (Leader, Orange County Support Group and 8-year myeloma survivor); and Mike Verla (Leader, Los Angeles Support Group and 12-year myeloma survivor).
Video billboard in Times Square
During the entire month of March, the IMF Myeloma Action Month digital billboards were displayed in Times Square at 42nd Street & 7th Avenue in New York City, New York. See photo on the front cover of this edition of Myeloma Today
Myeloma Community Workshop
This in-person Myeloma Community Workshop took place on March 28 in Kansas City, Missouri.
“Hill Day” myeloma advocacy
During MAM, the IMF brought a group of myeloma advocates to Capitol Hill to ensure that the voices of the myeloma community are heard directly by policymakers in Washington, D.C. Myeloma patients, care partners, and advocates met with Members of Congress and their staff to share personal stories and deliver a simple but powerful message: We Are #MoreThanMyeloma
Beyond the month of March, the IMF continues to work to ensure that patient voices remain at the center of policy decisions.
2026 MAM is a global success
This year’s Myeloma Action Month #MoreThanMyeloma campaign shined a global spotlight on the people living with this disease. To drive awareness, we lit landmarks red around the world and ran a Times Square video display in New York City all day, every day throughout March.
At its core, the #MoreThanMyeloma campaign reflects the voices of those who are affected by myeloma; people living with

this disease, not for it. The campaign connects their experiences to action, bringing more people into the conversation, supporting those navigating myeloma every day, and recognizing the progress being made for early detection, improved outcomes, and moving closer to a cure each and every day.
The IMF is honored and grateful to have partnered with GMAN member patient advocacy organizations from around the world to bring global attention to myeloma during Myeloma Action Month. Together, we made Myeloma Action Month a viral movement and showed the world the strength of the myeloma community!
The IMF is grateful to all of our sponsors for supporting the extremely successful 2026 Myeloma Action Month #MoreThanMyeloma global campaign:

Adaptive Biotechnologies
Amgen
Binding Site / Thermo Fisher
Bristol Myers Squibb GSK
Johnson & Johnson
Karyopharm Therapeutic , Kite Pharma / Arcellx
Legend Biotech
Pfizer
Regeneron Sanofi
For additional information about Myeloma Action Month, please visit myelomaactionmonth.org. MT


We Are #MoreThanMyeloma on Capitol Hill
By Danielle Doheny IMF Director, Public Policy & Advocacy
During Myeloma Action Month (MAM) in March 2026, the International Myeloma Foundation (IMF) brought patients, care partners, and advocates to Capitol Hill, ensuring that the voices of the myeloma community are heard directly by policymakers in Washington, DC.
The 2026 MAM advocacy efforts were grounded in a simple but powerful message: #MoreThanMyeloma. The campaign emphasizes that patients are more than a diagnosis, they are individuals with full lives, responsibilities, and goals, all of which are impacted by their ability to access timely, affordable care.
Over the course of the MAM Hill Day, IMF advocates met with Members of Congress and their staff to share personal stories, highlight ongoing challenges, and reinforce the urgent need for policies that reflect the realities faced by myeloma patients across the country. These conversations made clear that while scientific progress in myeloma has been significant, barriers to access continue to shape patient outcomes and quality of life.
Policy priorities for the myeloma community
The 2026 MAM Hill Day focused on the following key policy priorities aimed at improving access, affordability, and innovation:
Cancer drug parity
Myeloma advocates urged Congress to advance the Cancer Drug Parity Act to ensure that oral cancer medications are covered on par with intravenous and injectable treatments. For people with myeloma, many of whom rely on oral therapies, disparities in coverage can lead to significantly higher out-of-pocket costs and create unnecessary barriers to care.
Prior authorization reform
To streamline prior authorization in Medicare Advantage, the IMF is advocating for passage of the Improving Seniors’ Timely Access to Care Act. Delays can be especially harmful for patients managing a complex and relapsing disease like myeloma.
Clinical trial access
The Clinical Trial Modernization Act would reduce financial and logistical barriers to study participation by allowing support for expenses (e.g., travel, lodging). Myeloma advocates are highlighting the critical need to expand access to clinical trials so that all patients can benefit from emerging therapies.
Federal investment in research
Hill Day meetings also reinforced the importance of sustained funding for the National Institutes of Health (NIH) and the National Cancer Institute (NCI) as well as the Congressionally Directed Medical Research Program (CDMRP). Continued federal investment remains essential to advancing innovative treatments, including immunotherapies and cellular therapies that are transforming outcomes for myeloma patients.
Elevating the patient voice
A defining feature of IMF Hill Day is the role of the participating patient and care partner advocates who shared firsthand experiences navigating myeloma diagnosis, treatment, and survivorship, bringing real-world perspective to policy discussions.

These stories underscored that patients are not defined by their disease. They are parents, partners, professionals, and community members whose lives are deeply affected by policies that determine access to treatment, affordability, and care delivery. For many, Hill Day is not only an opportunity to educate lawmakers, but also a meaningful way to ensure that policy decisions reflect the lived experience of patients.
A call to action
While Myeloma Action Month provides an important moment to raise awareness, the needs of the myeloma community extend far beyond the month of March. Hill Day serves as a reminder that advocacy is essential to ensuring that patients are seen not just as diagnoses, but as individuals. Policies must support the whole person, not just the condition. Together with our dedicated team of myeloma advocates, the IMF is working to ensure that patient voices remain at the center of policy decisions, today and beyond.
If you are interested in becoming a myeloma advocate and sharing your story with policymakers, the IMF offers its Advocacy Master Class, a training program designed to equip patients and care partners with the tools and the confidence needed to engage in advocacy. No prior experience is needed, only a willingness to drive meaningful change. MT
Reach out to us at advocacy@myeloma.org, visit advocacy.myeloma.org to learn more about our activities and how we support our advocates, and subscribe to the IMF Advocacy Newsletter at subscribe.myeloma.org.




Las Voces de Mieloma – Grupo Virtual Un grupo de apoyo voluntario para pacientes interesados, se anima a los cuidadores, familiares y amigos a unirse a nosotros. Se trata de una excelente manera de conocer e interactuar con otros supervivientes y cuidadores para conocer los últimos avances en el tratamiento y la gestión del mieloma.
Para más información: lasvoces@imfsupport.org lasvoces.support.myeloma.org
SABER ES PODER !
IMF NEWS
Stephen L. Houff, MD
Joins the Board of
Directors

In January 2026, the International Myeloma Foundation (IMF) Board of Directors welcomed its newest member, Stephen L. Houff, MD.
Dr. Houff is a highly regarded physician, an innovative business leader, and a myeloma survivor who is passionate about improving patient outcomes by bridging the gap between cutting-edge research and community healthcare settings.
“Dr. Houff brings an extraordinary combination of clinical expertise, healthcare leadership, and lived experience as a myeloma patient,” said Heather Cooper Ortner (IMF President & CEO). “His journey from physician to patient and advocate gives him a deep understanding of both the science and the human impact of this disease. We are honored to welcome him to the Board.”
After earning his degree from the University of Maryland School of Medicine and completing a residency at the University of Massachusetts Medical Center, Dr. Houff practiced clinical medicine for more than 15 years before moving exclusively to the business side of medicine. Dr. Houff grew his hospital-based practice in internal medicine into a national company encompassing 1,000 clinicians and serving 140 healthcare systems across 38 states. Following the sale of the Hospitalists Management Group, a business Dr. Houff founded and led as CEO, he served in a variety of leadership roles in hospital clinical outsourcing companies.
“[Dr. Houff’s] experience leading large, complex healthcare organizations, combined with his firsthand understanding of myeloma, will strengthen our oversight and help guide the Foundation’s strategy as we work to ensure that scientific progress translates into better outcomes for patients everywhere,” said Dr. S. Vincent Rajkumar (Chairperson, IMF Board of Directors). MT
Founders
Board of Directors
Chairperson S. Vincent Rajkumar, MD
Christine Battistini
Loraine Alterman Boyle
Martine Elias, MSc
George T. Hayum
Stephen Houff, MD
Jason Katz
Andrew Kuzneski, III
Sagar Lonial, MD
Nikhil Munshi, MD
Charles Newman, MS
Kent Oliver Poornima Parameswaran, PhD
Matthew Robinson, MBA
Sanjay Singh
Maria Whitman, MBA
IMF Executive Team
Heather Cooper Ortner President & Chief Executive Officer
Peter Anton Vice President, Marketing
Diane Moran Senior Vice President, Strategic Planning
Brian D. Novis • Susie Durie • Brian G.M. Durie, MD IMF Team
Nikki Arends Senior Planner, Meetings & Events
Betty Arevalo Inventory Control Manager
Katie Atkins Associate Director, Support Groups
Becky Bosley Director, Support Groups
Brittnay Brandon Senior Coordinator, Meetings & Events
Michelle Carroll Director, Prospect Development
Lisette Contreras Administrative Assistant, Meetings & Events
Danielle Doheny Director, Public Policy & Advocacy
Jon Fitzpatrick Senior Manager, Meetings & Events
Simona Grace Director, Development
Paul Hewitt Coordinator, InfoLine
Kevin Huynh Coordinator, Tech Solutions
Marya Kazakova Senior Director, Editor-in-Chief, Publications
Missy Klepetar Coordinator, InfoLine
Sapna Kumar Marketing Strategist
Jason London Senior Manager, Marketing & Communications
Lisa Paik
Executive Vice President, Research & Operations
Kristina Mease Director, Meetings & Events
Jim Needham Publication Design
Selma Plascencia Senior Director, Operations
Joy Riznikove
Database Administrator
Cecilia Romero Project & Technology Manager, Support Groups
Miko Santos Senior Manager, Tech Solutions
Narmeen Shammami
Research Project Manager
Brando Sordoni Senior Associate, Accounting & Distribution
Rafi Stephan Senior Administrative Support
Daria Tabota Associate, Marketing & Communications
Joi Tisdale Project Manager
Jennifer Wieworka Director, Support Groups
Sandy Wilkes Grants Manager
Yara William
Associate Director, Support Groups
Robin Tuohy Vice President, Patient Support imfteam.myeloma.org
Ed Yerke-Robins
Gift Processing & Donor Data Specialist
Marquela Zepeda Grants Specialist

International Myeloma Foundation
4400 Coldwater Canyon Avenue, Suite
Studio City, CA 91604 USA
myeloma.org
800.452.CURE
Change Service Requested
myeloma.org

