
MIDDLE EAST & AFRICA

MARKET TRENDS
Supply chain in Pharma manufacturing
EDITOR’S PICK
Catering in Dubai hospitals
DISEASE FOCUS
Kala-azar



MARKET TRENDS
Supply chain in Pharma manufacturing
EDITOR’S PICK
Catering in Dubai hospitals
DISEASE FOCUS
Kala-azar
Dr. Omar Ghanem, M.D., shares his passion for treating patients with obesity



www.manawards.fwafrica.net/pack

Medical Devices & Innovations

SUPPLY CHAIN IN PHARMA MANUFACTURING
Supply chain resilience in pharmaceutical manufacturing: Building a stronger backbone

COMPANY FEATURE: SCIVION LIMITED
Scivion Limited: Elevating pharmaceutical manufacturing in East Africa

EDITOR’S PICK:CATERING IN DUBAI HOSPITALS
Beyond the tray: How catering shapes the healthcare experience in Dubai

OPINION PIECE: AFRICA’S DIAGNOSTICS CRISIS
Enough talking. Africa’s diagnostics crisis demands emergency action

HOSPITAL FEATURE: MAYO CLINIC
Dr. Omar Ghanem, M.D., shares his passion for treating patients with obesity
DISEASE FOCUS: KALA-AZAR
Kala-azar: A deadly but preventable parasitic disease

TOPICAL FOCUS: PATIENT-CENTRIC CARE POSTRATE
From cure to care - the patient-centric revolution in Urology








EAST & AFRICA
Year 4 | Issue No. 18 | Jan-March 2026
FOUNDER & PUBLISHER
Francis Juma
SENIOR EDITOR
Alphonse Okoth
EDITOR
Vincent Moranga
EXTERNAL CONTRIBUTOR
Mark Adams, CMC Hospital
Dr Ifedayo Adetifa, CEO, FIND
BUSINESS DEVELOPMENT DIRECTOR
Virginia Nyoro
BUSINESS DEVELOPMENT ASSOCIATE
Johna Sambai
HEAD OF DESIGN
Clare Ngode
DESIGN
Monica Wachio
ACCOUNTS
Anita Kinyua

We publish some of the most influential magazines and websites in Africa & the Middle East regions. Please visit the websites below for more information about our publications

www.foodbusinessmea.com

www.horecamea.com

PUBLISHED BY: FW BRANDS MEA
P.O. Box 1874-00621, Nairobi Kenya
Tel: +254 725 34 39 32
Email: info@fwafrica.net
Company Website: www.fwafrica.net

HealthCare MEA (ISSN 2307-3535) is published 4 times a year by FW BRANDS MEA. Reproduction of the whole or any part of the contents without written permission from the editor is prohibited. All information is published in good faith. While care is taken to prevent inaccuracies, the publishers accept no liability for any errors or omissions or for the consequences of any action taken on the basis of information published.
www.healthcaremea.com
www.millingmea.com
www.dairybusinessmea.com www.sustainabilitymea.com













Each year on World Obesity Day, the global health community pauses to reflect on a growing epidemic that no country can afford to ignore. Once perceived as a challenge largely confined to high-income nations, obesity is now rising at an alarming rate across Africa and the Middle East.
Urbanization, changing diets, sedentary lifestyles and aggressive marketing of ultra-processed foods are reshaping public health profiles across the continent. For African governments already battling infectious diseases and strained healthcare systems, the rapid growth of non-communicable diseases linked to obesity, such as diabetes, cardiovascular disease and certain cancers, poses a double burden.
The lesson from World Obesity Day is clear: prevention must be prioritized. Investment in nutrition education, urban planning that promotes physical activity, fiscal policies that discourage unhealthy foods, and early screening programs are no longer optional. Africa has a narrow window to avoid the full-scale crisis now confronting many developed economies.
It is against this backdrop that we present Issue 18 of Healthcare Middle East & Africa magazine. In this edition, we spotlight institutions and innovators shaping the future of care delivery across regions.
Our company features include the globally renowned Mayo Clinic, whose model of integrated, patient-focused care continues to influence health systems worldwide. We also feature Scivion Limited, a pharmaceutical excipient distribution and technical support company playing a vital role in strengthening pharmaceutical value chains across emerging markets.
In our Disease Focus section, we turn the spotlight on Kalaazar, also known as visceral leishmaniasis—a neglected tropical disease that continues to affect vulnerable communities in parts of East Africa. Though preventable and treatable, it
persists due to late diagnosis, limited awareness and resource constraints. As Africa responds to rising lifestyle diseases such as obesity, it must not lose sight of endemic conditions that disproportionately impact the underserved. Balanced health priorities remain critical.
Our Topical Focus features insights from Mark Adams, CEO of CMC Hospital Dubai, who explores the evolving landscape of patient-centric care. In a post-pandemic era marked by digital transformation and heightened patient expectations, Adams underscores the need for healthcare providers to move beyond transactional interactions toward holistic, personalized experiences. Technology, transparency and empathy, he argues, are fundamental to rebuilding trust and delivering sustainable outcomes.
In the Trends section, we examine the pharmaceutical manufacturing supply chain, an area thrust into the spotlight by recent global disruptions. From sourcing raw materials to ensuring last-mile distribution, resilience and localization are emerging as strategic imperatives for African nations striving for medicine security.
Meanwhile, in our Opinion piece, Mark Adams reflects on catering in Dubai hospitals, reminding us that nutrition services, often overlooked, play a pivotal role in patient recovery and overall care quality.
As obesity rates climb and healthcare systems evolve, this issue reinforces a simple but powerful truth: prevention, innovation and patient-centred thinking must shape Africa’s next chapter in health.
Enjoy the read!
Alphonse Okoth Senior Editor HealthCare MEA
Duphat Dubai 2026 24th - 26th March 2026
Dubai World Trade Centre, Dubai, UAE www.duphat.ae/duphat-exhibition
Uganda Pharma & Healthcare International Expo 2026 26th - 28th March 2026
UMA Show Grounds, Kampala, Uganda www.ugandapharmahealth.com/index
Kenya Pharma & Health International Expo 2026 9th - 11th April 2026
Sarit Expo Centre, Nairobi, Kenya www.kenyapharmahealth.com
Medtec Kenya 2026 April 21st – 24th, 2026. Sarit Expo Centre, Nairobi, Kenya. www.expogr.com/kenyamedtec
India Pharma Expo 2026 23rd -25th April 2026
Hitex Exhibition Centre Hyderabad, India www.indiapharmaexpo.com/index.html
CPHI Middle East 2026 11th -13th May 2026
Riyadh Exhibition and Convention Centre, Malham, Saudi Arabia www.cphi.com/middle-east/en/home.html
Healthcare Innovation Summit Africa 27th – 29th 2026
Gallagher Convention Centre, Johannesburg, South Africa www.itnewsafrica.com/event/event/healthcare-innovation-summit-africa/
Pharma Futures East Africa 3rd – 4th June 2026
Sarit Expo Centre, Nairobi, Kenya www.pharmafutures-africa.com
Pharma India Expo 2026 7th - 9th July 2026
Bombay Exhibition Centre, Mumbai www.pharmaindiaexpo.com
Africa Pharma Manufacturing Expo (Eastern Africa / Kenya Edition)
15th - 17th July 2026
Sarit Expo Centre, Nairobi, Kenya www.pharma.afmass.com/east




INDIA — India has confirmed two cases of Nipah virus infection in West Bengal’s North 24 Parganas district, marking the country’s seventh documented outbreak of the deadly pathogen.
The patients, both 25-year-old nurses working at a private hospital in Barasat, developed symptoms in late December 2025 that quickly progressed to severe neurological complications. They were isolated in early January as
their conditions deteriorated.
Initial detection was carried out by the Viral Research and Diagnostic Laboratory at a government hospital in Kalyani on January 11, with confirmation provided two days later by the National Institute of Virology in Pune.
Following confirmation, authorities launched extensive contact tracing, identifying 196 individuals who had contact with the infected nurses.
All contacts have remained asymptomatic and tested negative, offering cautious optimism as containment efforts continue.
This is West Bengal’s third Nipah outbreak, following earlier incidents in 2001 and 2007. The affected districts border Bangladesh, where outbreaks occur almost annually, heightening concerns about cross-border transmission.
The national government has since deployed a response team to strengthen surveillance, testing, and infection
prevention measures.
Regional governments have also intensified precautionary actions. Thailand has introduced passenger screening at three airports receiving flights from West Bengal, while Nepal has activated health protocols at Kathmandu airport and land border crossings.
Singapore’s Communicable Diseases Agency is closely monitoring developments and has classified Nipah as a notifiable disease requiring immediate reporting.
The World Health Organization lists Nipah among its top priority diseases due to its epidemic potential.
Transmitted by fruit bats and through contaminated food or close human contact, the virus carries a fatality rate of 40% to 75%.
There is currently no licensed vaccine or specific antiviral treatment, making early supportive care critical for improving survival outcomes.
USA — Boston Scientific has agreed to acquire California-based medtech firm Penumbra for US$14.5 billion, marking its largest deal in two decades and the first major healthcare transaction of 2026.
The acquisition significantly expands Boston Scientific’s cardiovascular device portfolio and signals its return to the neurovascular market, more than a decade after divesting that business to Stryker.
CEO Mike Mahoney described the deal as a “home run” and “financially compelling,” highlighting strong strategic alignment and long-term growth potential.
Executives believe Penumbra’s products will strengthen both revenue and profit margins, particularly as
demand for heart and vascular devices rises globally.
Boston Scientific recently raised its annual profit forecast, citing sustained growth in its cardiovascular segment.
The move comes as analysts anticipate a resurgence of large-scale healthcare mergers, supported by a more favorable regulatory climate and easing interest rates.
Truist analyst Richard Newitter noted the acquisition restores Boston Scientific’s presence in several highgrowth markets while addressing a gap in its neurovascular offerings.
Mahoney added that the company had long viewed neurovascular as attractive but waited to re-enter with sufficient commercial scale and pipeline depth.
Penumbra brings a broad portfolio of devices used to treat pulmonary embolism, stroke, deep vein thrombosis, acute limb ischemia, heart attack, and aneurysms.
The company expects to generate approximately US$1.4 billion in revenue in 2025.
Under the agreement, Penumbra shareholders will receive either US$374 per share in cash or 3.8721 Boston Scientific shares, reflecting a 19.3% premium.
The transaction is structured as roughly 73% cash and 27% stock and is expected to close in 2026. Penumbra CEO Adam Elsesser will join Boston Scientific’s board upon completion.







IRELAND – Novo Nordisk is investing €432 million (USD 470 million) to expand its tabletting facility in Monksland, Athlone, enhancing production of oral GLP-1 treatments for diabetes and obesity.
The upgrade will modernize operations with advanced technology and sustainable practices, strengthening Ireland as a strategic manufacturing hub for international markets.
Kasper Bødker Mejlvang, Executive VP of Product Supply, said the expansion enables the company to meet growing global demand while maintaining high-quality standards.
Construction, already underway, is expected to create up to 500 jobs, complementing the existing 260 employees.
The project, scheduled for completion between late 2027 and 2028, underscores Novo Nordisk’s commitment to innovation, patient care, and economic development in the Athlone region.
West Africa launches innovative roadmap to ensure equitable access to Lassa fever
NIGERIA - West Africa has launched the Lassa Fever End-to-End (E2E) Access Roadmap, a firstof-its-kind, regionally led framework to ensure equitable access to Lassa fever vaccines once approved.
Developed with support from the Coalition for Epidemic Preparedness Innovations (CEPI) and led by the West African Health Organization (WAHO), the roadmap connects every stage from vaccine development to sustained delivery.
The framework addresses research, regulatory readiness, manufacturing, financing, procurement, and distribution, prioritizing uninterrupted supply chains and early planning.
It reflects lessons from past epidemics and emphasizes regional ownership, coordination, and long-term sustainability.
Officials hope the roadmap will maximize readiness for the first vaccine approvals anticipated within five years, strengthening epidemic preparedness and health security across West Africa.
ETHIOPIA — The Africa Centres for Disease Control and Prevention (Africa CDC) has inaugurated its first dedicated warehouse in Addis Ababa, Ethiopia, strengthening the continent’s ability to respond swiftly to public health emergencies.
The 1,000-square-meter facility enables Africa CDC to procure, store, and rapidly deploy critical medical supplies during disease outbreaks.
Equipped with advanced cold storage systems, the warehouse ensures the safe preservation of temperature-sensitive products such as vaccines and other essential medical commodities.
This addresses a long-standing gap in Africa’s emergency response infrastructure, where delays in accessing supplies can determine whether outbreaks are contained or escalate.
Dr. Raji Tajudeen, Africa CDC’s Deputy Director General, described the facility as a strategic milestone, noting that timely access to medical products is vital for efficient outbreak response and containment across the continent.
The project was financed by the Mastercard Foundation through Mpox emergency response funding.
The United Nations World Food Programme (WFP) provided technical support, including design, engineering supervision, logistics planning, and compliance with safety standards.
WFP’s Acting Director of the African Union Global Office, Lydie Kouame, highlighted the initiative as a strong example of effective international collaboration in strengthening health systems.
The Addis Ababa warehouse is a key component of Africa CDC’s broader Supply Chain Framework, aimed at expanding warehousing and logistics capacity across Africa.
Additional infrastructure is underway, including a regional warehouse in Douala, Cameroon, funded by the African Development Bank.
According to Tesfaye Hailemichael, Head of Africa CDC’s Supply Chain Division, effective emergency response depends on preparedness.
These investments ensure that life-saving supplies are coordinated, readily available, and rapidly deployable when needed most.

CHINA — AstraZeneca has entered an US$18.5 billion licensing agreement with China-based CSPC Pharmaceuticals to develop next-generation weightloss and type 2 diabetes treatments, intensifying competition in the fastgrowing obesity drug market.
Under the deal, AstraZeneca will pay US$1.2 billion upfront for access to eight development programs. CSPC could receive up to US$3.5 billion in additional milestone payments tied to development and regulatory progress, plus royalties on approved products.
The agreement also includes up to US$13.8 billion in sales-based milestone payments, bringing the total potential value to US$18.5 billion.
At the center of the partnership is
SYH2082, a once-monthly injectable candidate that combines GLP-1 receptor agonist (GLP-1RA) and GIP receptor agonist (GIPR) mechanisms.
The drug is set to enter Phase I clinical trials and is among four programs expected to advance through earlystage development. Three additional preclinical candidates complete the initial portfolio.
AstraZeneca will lead latestage development and global commercialization, excluding Greater China. CSPC retains exclusive rights to develop and market the treatments in mainland China, Taiwan, Hong Kong, and Macau.
The deal bolsters AstraZeneca’s obesity pipeline, which already includes
elecoglipron, an oral GLP-1RA, alongside weekly injectable candidates and preclinical assets.
Extended dosing intervals, particularly monthly formulations, are increasingly viewed as a competitive advantage in improving patient adherence and convenience.
The agreement follows AstraZeneca’s US$5.2 billion partnership with CSPC in June 2025 focused on chronic disease research, signaling deepening strategic ties.
Industry-wide competition is also accelerating, highlighted by Pfizer’s high-profile acquisition of Metsera amid bidding interest from Novo Nordisk.


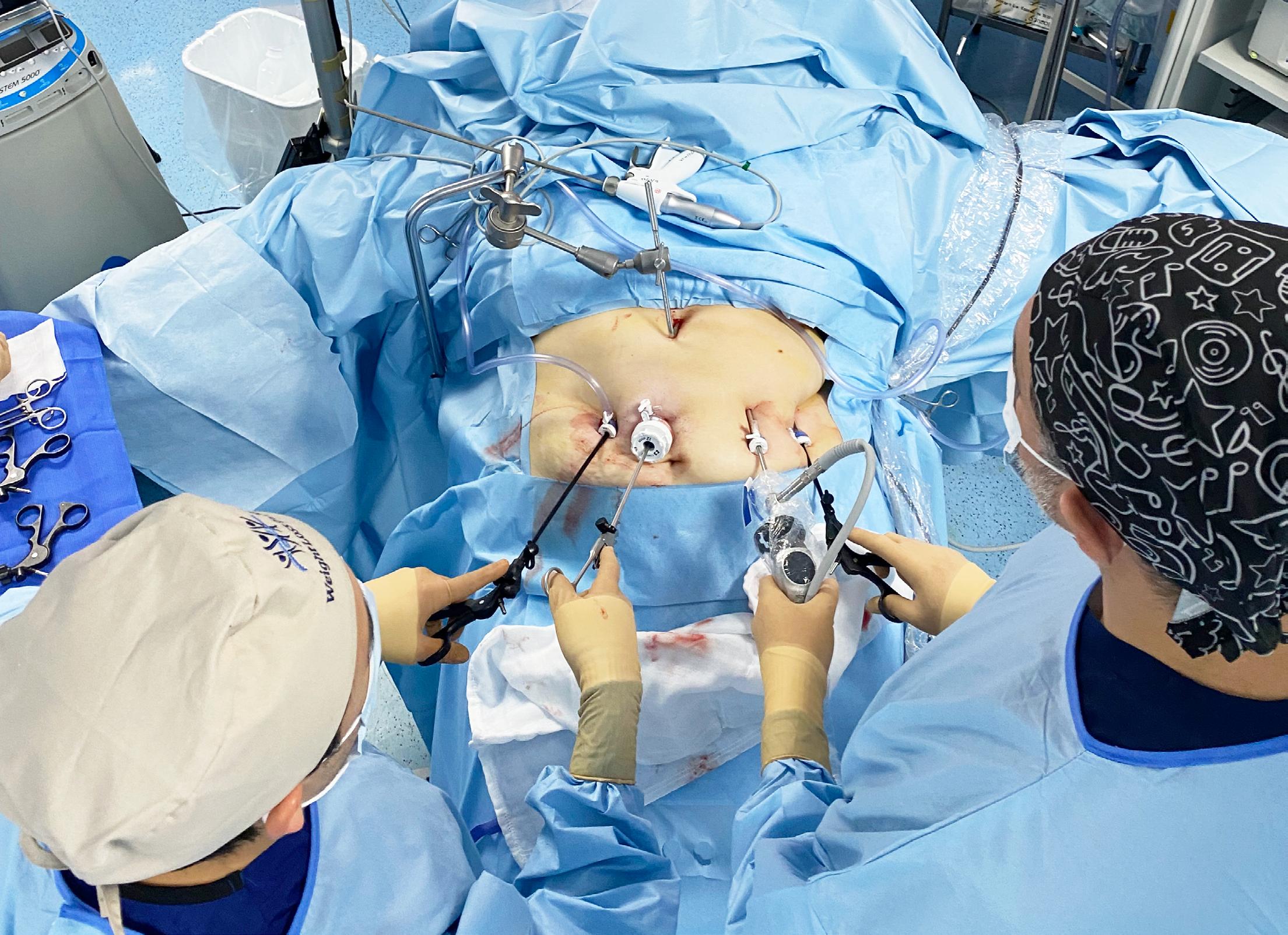
Teaching and Referral Hospital (JOOTRH) has commissioned a state-ofthe-art cryogenic liquid oxygen plant to ensure uninterrupted supply of medical oxygen across its facilities, significantly strengthening emergency and critical care services.
The hospital’s Chief Executive Officer, Dr. Joshua Clinton Okise,
formally received the system from Gatewell Solutions Ltd during a handover ceremony, following successful technical trials and staff training led by chief engineer Joseph Wananga.
At the heart of the installation is a 20,000-litre cryogenic liquid oxygen tank—an upgrade from conventional Pressure Swing Adsorption (PSA) plants.
While PSA systems typically produce oxygen at 93% purity, the new plant delivers medical-grade oxygen at 99.9% purity, enhancing patient safety and treatment outcomes.
The system stores oxygen in liquid form at extremely low temperatures. When needed, a specialized vaporizer converts the liquid into gas, which is then distributed through the hospital’s central pipeline network to wards, intensive care units, theatres, and other high-dependency areas.
To prevent supply disruptions, JOOTRH has integrated a 10×2 automated
manifold backup system holding 20 compressed oxygen cylinders.
The backup activates instantly if liquid levels in the main tank fall, ensuring continuous oxygen flow to critical patients.
The hospital now operates multiple oxygen sources simultaneously: the new cryogenic tank, an existing PSA plant, the automated manifold cylinder system, and a secondary BOC tank.
This layered infrastructure significantly enhances resilience against emergencies or supply shocks.
The project was implemented under Kenya’s Ministry of Health through the COVID-19 Health Emergency Response Project, funded by the World Bank.
Gatewell Solutions Ltd has provided a one-year warranty, while JOOTRH engineers have undergone specialized training to manage daily operations, refilling, and routine maintenance, ensuring the plant operates efficiently around the clock.
LIBERIA — John F. Kennedy Medical Center has unveiled a USD132.8m transformation plan, the most ambitious overhaul in its 50-year history, aimed at modernizing Liberia’s premier referral hospital.
However, only USD9.5 million in government funding has been secured, leaving a USD123.3 million financing gap.
Chief Executive Officer Dr. Linda A. Birch presented the 2025–2029 strategy under the theme “Reimagining Excellence: A New Era for John F. Kennedy Medical Center.”
The plan focuses on infrastructure renewal, service expansion, digital transformation, and long-term financial sustainability.
Built in 1971 to serve a population of under one million, the hospital
now supports more than 5.5 million Liberians.
Aging facilities, marked by cracked walls, leaking ceilings, damaged sewage systems, and outdated electrical networks, struggle to meet rising demand.
Equipment shortages, limited biomedical systems, and workforce gaps further constrain service delivery.
JFK has expanded beyond its traditional specialties to include cardiology, urology, orthopedics, neonatal intensive care, and dialysis.
Plans are underway to conduct Liberia’s first cardiac surgery by November 2026. New investments include MRI installation, digital X-ray systems, CT scanners, and upgraded ultrasound services, reducing reliance on external diagnostic providers.
Administrators plan to replace paperbased records with an integrated digital health information system to streamline patient registration and reduce waiting times.
Financial reforms have consolidated revenue collection points, strengthened banking partnerships, and established an in-house pharmacy.
Despite these efforts, government salary allocations consume 92% of the national budget for JFK, leaving only about US$600,000 annually for operations. Uncompensated emergency care adds financial strain.
House Speaker Richard Nagbe Koon described the plan as a “social contract” with Liberians, while international partners signaled support, underscoring the urgent need for sustained funding and political commitment.

USA — Eli Lilly and Company has chosen Huntsville, Alabama, as the site for a new US$6 billion manufacturing facility dedicated to producing orforglipron, its oral treatment candidate for obesity and type 2 diabetes.
Construction is expected to begin in 2026, marking a major step in the company’s expanding U.S. footprint.
The project is set to create around 3,000 construction jobs during the building phase and more than 450 permanent positions once operational, including roles for engineers, scientists, operations specialists, and laboratory technicians.
The Huntsville plant will produce both small-molecule synthetic medicines and peptide-based therapies, with orforglipron positioned as a key product within Lilly’s glucagon-like peptide-1 receptor agonist (GLP1RA) portfolio.
The company plans to integrate machine learning, artificial intelligence, and advanced digital automation across the site to enhance production efficiency, strengthen supply chains, and improve overall productivity.
This digital-first strategy builds on Lilly’s recent collaboration with NVIDIA to develop a high-powered supercomputer aimed at accelerating drug discovery and development.
The Alabama facility forms part of Lilly’s broader US$27 billion domestic manufacturing expansion plan.
The company intends to build four new U.S. plants, including a US$6.5 billion orforglipron site in Texas and a US$5 billion complex in Virginia focused on bioconjugate and monoclonal antibody production.
It is also investing US$1.2 billion to expand its existing orforglipron operations in Puerto Rico.
Globally, Lilly is strengthening its European manufacturing network with a US$3 billion orforglipron plant in the Netherlands and a US$1 billion expansion in Limerick, Ireland.
The company plans to submit a new drug application for orforglipron to the U.S. Food and Drug Administration, with analysts projecting potential annual sales of up to US$13 billion by 2031.
USA – Johnson & Johnson has announced a USD 1 billion investment to build a next-generation cell therapy facility in Montgomery County, Pennsylvania, creating around 500 permanent jobs and over 4,000 construction roles.
The move is part of the company’s broader USD 55 billion U.S. manufacturing expansion, aimed at strengthening domestic production and mitigating potential pharmaceutical tariffs.
The investment also includes a new drug product facility in North Carolina, complementing existing biologics operations and partnerships with contract manufacturers like Fujifilm Biotechnologies.
J&J’s expansion supports its CAR-T therapy, Carvykti, which has seen rapid sales growth but faces supply constraints, reflecting the company’s commitment to advanced cell therapies and securing domestic production for high-demand treatments.
KENYA — The government has established the National Food Environment Policies Steering Committee to tackle the country’s rising burden of diet-related diseases.
Convened by the Ministry of Health, the committee held its inaugural meeting on February 26, 2026, in Nairobi, chaired by Dr. Joel Gondi on behalf of Director-General for Health Dr. Patrick Amoth.
Health officials say Kenya faces a dual challenge of persistent malnutrition alongside increasing cases of obesity, diabetes, cardiovascular diseases and cancers.
The committee will develop policies including front-of-pack nutrition labelling, restrictions on marketing unhealthy foods to children, elimination of industrial trans fats, and fiscal measures to encourage healthier diets.
It will also promote product reformulation and healthy food procurement in public institutions, strengthening existing national nutrition and NCD strategies

Dr. Alicia López de OcárizCorporate Medical Director, Cinfa Group

Diabetes has become one of the most pressing health challenges worldwide, with the MENA region standing at the epicenter. According to recent data, this region reports the highest global prevalence—17.6% of adults live with diabetes, and the number of cases is projected to nearly double by 2050, reaching 163 million.
Notably, one in three cases remains undiagnosed, leaving millions at risk of severe complications due to late detection.1
In response, Cinfa, the leading laboratory in the Spanish pharmaceutical market and with presence in over 100 countries worldwide, is reinforcing its dedication to improving chronic disease care in the Gulf, building on a strategy that has been implemented for over 10 years, focusing on chronic diseases such as diabetes and cardiovascular conditions. The company combines pharmaceutical advances with targeted therapeutic development to provide effective treatment options, while fostering scientific collaboration to better understand regional healthcare challenges.
Building on these insights and in line with its commitment to providing effective solutions for type 2 diabetes, Cinfa works to optimize blood glucose regulation. In this context, the company has introduced vildagliptin, under the brand name Gliptacin, for the treatment of type 2 diabetes. It can be used as monotherapy - in patients inadequately controlled by diet and exercise alone and for whom metformin is inappropriate due to contraindications or intolerance- and as combination therapy as well.
In the coming months, this therapy will also be available in combination with metformin, under the brand name Gliptacin Met, providing an additional option for comprehensive diabetes management.
Dr. Alicia López de Ocáriz, Corporate Medical Director at Cinfa Group, stressed the importance of ensuring global access to high-quality treatments, highlighting that this requires continued R&D investment, accessibility, adherence support, and prevention programs. With 56 years of experience and a team of over 2,300 professionals, Cinfa provides accessible and quality pharmaceutical solutions, merging expertise, innovation, and a strong commitment to global well-being.
Cinfa approaches diabetes care as part of a broad, sustained
WE HAVE A RESPONSIBILITY TO IMPROVE GLOBAL ACCESS TO THE HIGHESTQUALITY TREATMENTS, AND THIS IS A CORE PART OF OUR MISSION— ESPECIALLY WHEN IT COMES TO HIGHLY PREVALENT CONDITIONS SUCH AS DIABETES.
effort involving multiple disciplines, and promoting continuous scientific dialogue and knowledge exchange through events and training programs. This approach underlines the importance of interdisciplinary collaboration, ongoing medical education, and incorporating patient experiences to inform clinical decisions and improve long-term outcomes.
Beyond therapeutic solutions, Cinfa actively participates in leading scientific congresses across the Middle East, strengthening partnerships with regional and international experts and reinforcing a long-term vision of collaboration to improve patient care.
As part of this commitment and given that cardiovascular diseases are the main complications of diabetes, Cinfa continues to actively engage in initiatives that foster closer collaboration with healthcare professionals, exemplified by its participation for the third consecutive year in the 36th Annual Conference of the Saudi Heart Association (SHA 2025). The company has shown its cardiovascular portfolio to healthcare professionals, demonstrating its ongoing commitment to providing accessible, high-quality, evidence-based therapies aimed at improving patient outcomes across the Middle East. By joining this international gathering, Cinfa contributes to the scientific exchange, driving advances in cardiovascular medicine and reinforcing its dedication to sustainable, patientfocused healthcare systems.Cinfa is the leading laboratory in units and values in the Spanish pharmaceutical market, present in over 100 countries.
References
1. Diabetes Atlas. https://diabetesatlas.org/data-by-location/ region/middle-east-and-north-africa/

Boehringer
INDIA — Boehringer Ingelheim has appointed Meenal Gauri as Managing Director for India, effective 12 January 2026, overseeing operations in India, Bangladesh, Nepal, Sri Lanka, and Maldives.
She also joins the Board of Boehringer Ingelheim India, taking charge of driving strategic initiatives across the region.
Gauri brings over 16 years of international healthcare experience across Europe, Asia, and the Middle East, with expertise in marketing, sales, corporate strategy, business development, and market access.
Her track record includes leading successful product launches and expanding Boehringer Ingelheim’s presence in complex markets.
A trained Co-Active coach, she emphasizes talent development, aligning with the company’s culture of collaboration and workforce growth.
In her new role, Gauri is committed to improving patient health, fostering innovation, and strengthening partnerships across the India region.
Evren Ozlu, Head of Human Pharma for India, Middle East, Turkey, and Africa, praised her industry expertise and people-first leadership approach, expressing confidence in her ability to advance Boehringer Ingelheim’s mission.
Holdings appoints Dr. Ali Bin Harmal Aldhaheri as new chairman
UAE — Amanat Holdings PJSC has appointed Dr. Ali Saeed Bin Harmal Aldhaheri as its new Chairman of the Board of Directors, effective following a board meeting on February 12, 2026.
He succeeds Dr. Shamsheer Vayalil, who stepped down to focus on other business interests.
Dr. Ali has been a board member since November 2020 and brings over 20 years of expertise in education and business development.
He has founded and managed ventures across education, tourism, and real estate, and contributed to high-level government strategies in investment, MICE, sports, and international trade.
As Chairman, he will guide Amanat Holdings through its next growth phase, leveraging his experience leading multiple organizations.
Dr. Ali praised Amanat’s achievements under Dr. Shamsheer, including the AED 453 million (USD123.35m) sale of an education real estate asset in 2025 and the listing of Almasar Alshamil Education on the Saudi Exchange, noting the company’s strong profitability, regional impact, and commitment to value creation.
He expressed confidence in sustaining these positive trends and advancing Amanat’s mission in healthcare and education across the GCC.


Dr. Anne Musuva named acting CEO of Kenya Healthcare Federation
KENYA — The Kenya Healthcare Federation (KHF) has appointed Dr. Anne Musuva, a current KHF Board Member, as Acting CEO effective February 9, 2026, following the completion of Dr. Tim Theuri’s fixed-term contract on February 8.
Dr. Musuva will lead the federation during the search for a permanent CEO, ensuring continuity of operations.
Dr. Theuri, reflecting on his threeyear tenure, highlighted his role in expanding KHF’s membership, shaping national health policy, and supporting the transition to the Social Health Authority under Kenya’s Universal Health Coverage agenda.
He credited the Board and Secretariat for their collaboration and emphasized the human connections and partnerships that underpinned the federation’s growth.
Dr. Musuva now assumes responsibility for steering KHF’s initiatives, including promoting private sector contributions to healthcare, advancing digital integration, and strengthening local manufacturing.
The Board affirmed that leadership arrangements are in place to maintain operational stability while positioning the federation for continued impact on Kenya’s health sector.
appoints Beryl Naiken as first Secretary of State for Mental Health Care
SEYCHELLES — The Office of the President has appointed Beryl Naiken as the country’s first Secretary of State for Mental Health Care, effective January 15, 2026.
Mrs. Naiken brings over 28 years of experience as a Senior Clinical Psychotherapist, with expertise spanning psychotherapy, counselling, crisis intervention, rehabilitation, and national policy development.
She holds a Master of Psychotherapy and Counselling from the University of Adelaide, a Postgraduate Diploma in Psychology from Edith Cowan University, and is pursuing a PhD in Relationship Dynamics.
Her career includes leadership roles such as Director General at the Ministry of Youth, Sports and Family, Advisor to the Minister of Family Affairs, CEO of the Agency for the Prevention of Drug Abuse and Rehabilitation (APDAR), and most recently CEO of the Seychelles Human Rights Commission.
In her new role, Mrs. Naiken will oversee the development and implementation of mental health policies and programs, focusing on clinical care, community wellbeing, and human rights-based approaches.
Her appointment signals a strategic national commitment to enhancing mental health services and addressing the complex psychological needs of Seychellois citizens.


King Faisal Specialist Hospital extends CEO Dr. Björn Zoëga’s term until 2030
SAUDI ARABIA — King Faisal Specialist Hospital & Research Centre (KFSHRC) has extended Dr. Björn Zoëga’s tenure as CEO of its Riyadh operations through 2030.
The Board of Directors cited strong confidence in Dr. Zoëga’s leadership and the broader executive team’s ability to execute the hospital’s strategic objectives.
Since his appointment, Dr. Zoëga has strengthened the Centers of Excellence model, focusing specialized expertise and resources on complex clinical cases, while improving operational efficiency across clinical care, education, and research.
His leadership has enhanced KFSHRC’s global standing as a tertiary and quaternary care institution, expanded academic impact through research publications and clinical trials, and fostered international collaborations.
The extension ensures leadership continuity, allowing multi-year initiatives to proceed without disruption.
Dr. Zoëga’s tenure will continue to guide KFSHRC in delivering worldclass healthcare, advancing medical innovation, and training healthcare professionals, reinforcing the hospital’s position as a premier institution in Saudi Arabia and the broader international medical community. serving patients across the region and internationally.
Proff. Wezile Chitha elected President of Health Professions Council of South Africa
SOUTH AFRICA — Professor Wezile Chitha has been elected President of the Health Professions Council of South Africa (HPCSA) for a five-year term running from 2025 to 2030.
Professor Chitha currently serves as Executive Dean of the Faculty of Medicine and Health Sciences and Associate Professor of Health Systems and Policy at Walter Sisulu University.
A qualified medical doctor, health economist, and award-winning public health manager, he brings extensive experience in strategic planning, academic innovation, and governance across South Africa’s health system.
His career spans work in primary, tertiary, and academic healthcare settings, with collaborations involving government institutions, NGOs, research organizations, and universities nationally and internationally.
Chitha holds a medical degree, a master’s in public health (health economics) from the University of Cape Town, and a PhD in public health.
In his new role, he will lead HPCSA’s efforts to protect the public, strengthen regulatory frameworks, and advance integrity and excellence across health professions.

BY VINCENT MORANGA
The craze for weight loss among celebrities across Africa and the Middle East has spread to the general population, driving sales of GLP-1 medications like Ozempic, with or without expert input. Yet recent studies show that weight loss programs, pills, and injections often deliver only temporary results. Once patients stop the medication or complete the weight loss program, they tend to regain weight, making these options far costlier to maintain than alternatives like bariatric surgery.
Doctors regard bariatric surgery as the safest, most effective option for substantial, long-term weight loss and significant improvements in metabolic health. The procedure helps patients lose excess weight and lower risks of life-threatening conditions, including certain cancers (such as breast, endometrial, and prostate cancer); heart disease and stroke; high blood pressure; high cholesterol; nonalcoholic fatty liver disease (NAFLD) or nonalcoholic steatohepatitis (NASH); sleep apnoea; and type 2 diabetes.
The most common bariatric surgeries are Roux-en-Y gastric bypass and sleeve gastrectomy. , The procedures are recommended for the treatment of obesity for people with a body mass index (BMI) greater than 40 (class 3 obesity), or 35 to 39.9 (class 2 obesity) alongside a serious weight-related health problem, such as type 2 diabetes, high blood pressure, or severe sleep apnea. In certain cases, patients with a BMI of 30-34.9 may also qualify.
MAYO CLINIC'S PRESENCE IN THE REGION
In a recent interview with Healthcare Middle East and Africa, Dr. Omar Ghanem, M.D., Mayo Clinic's medical director for the Middle East, a metabolic surgeon and chair of the Division of Metabolic and Abdominal Wall Reconstructive Surgery at Mayo Clinic in Rochester, Minnesota, shared insights into his clinical practice. He discussed Mayo Clinic's journey in enhancing regional healthcare by connecting global expertise locally, along with Mayo Clinic's enduring commitment to patients in the region.
Mayo Clinic has been in practice for over 160 years, Dr. Ghanem noted that Mayo Clinic is marking the five-year anniversary of its patient appointment services office in Dubai and has relocated the office to Dubai Healthcare City for better accessibility. The new office is at Office 401, Al Jalila Foundation. The office staff may also be reached by email at dubaioffice@mayo.edu or by phone at +971-55-526-8899.


87%
PATIENTS WHO UNDERGO CORRECTIVE SURGERY
From there, Dr. Ghanem explained, patients and families seeking Mayo Clinic appointments can get free assistance with travel and lodging arrangements, help with questions about billing, insurance, general orientation, medical record reviews, and future scheduling. While the Dubai office doesn't provide medical care, it accepts direct patient appointment requests and physician referrals. "Interpreters are available at no cost to assist with communication between healthcare providers and patients whose primary language isn't English," he added.
The office also supports current and prospective Mayo Clinic Laboratories clients, healthcare providers, payers, and others interested in connecting with Mayo, including organizations interested in learning more about Mayo Clinic Platform, Mayo Clinic Care Network membership, or Mayo Clinic Global Consulting services.
Obesity poses a global challenge, with some patients facing significant stigma and barriers to care. Dr. Ghanem traces his motivation to his upbringing in rural Lebanon, in the mountains, where quality healthcare was scarce. "I was the first physician from my village," he says. "That experience shaped my purpose: to help people with limited healthcare options." It inspired him to specialize in metabolic and bariatric surgery.
He cited a JAMA Network study that projects that by 2050, half of the world's adults will live with obesity by 2050. Metabolic surgery can offer the most effective, durable treatment in some patients. "Beyond weight loss, it tackles related conditions like type 2 diabetes and hypertension," he said. "For many patients, the transformation is profound; some leave the hospital no longer needing medications for type 2 diabetes or high blood pressure."
"That's why I'm committed to this field," Dr.
Ghanem continued. "Metabolic surgery doesn't just change numbers on a scale; it restores health, improves quality of life, and gives patients their lives back. That's the impact we strive for every day."
Mayo Clinic is renowned for corrective surgeries after complications from procedures done elsewhere - such as hernias, fistulas, ulcers, or recurrent weight gain - often in patients with complex health issues. "Corrective surgery is highly complex, especially with multiple conditions," he said. "It requires expertise and coordination that few institutions can match. This is where Mayo Clinic excels, uniting specialists for truly multidisciplinary care."
"Sometimes, patients come to us needing weight loss before another critical procedure, like a hip or knee replacement, heart or kidney transplant, or hernia repair," he explained. "Collaboration is key. We work with cardiologists, anesthesiologists, gastroenterologists, endocrinologists, obesity medicine specialists and nutritionists to ensure safety and optimize outcomes."
For instance, a patient awaiting a heart transplant might need to reach a target BMI first. "I'll consult the cardiologist, then the anesthesiologist for assessing surgical risk, and endocrinology or obesity medicine for medication-supported weight loss if needed," Dr. Ghanem said. "Once the target is reached, we coordinate with the transplant team. During surgery, the transplant specialist is on hand if required, and vice versa."
Every year, patients turned away elsewhere find solutions at Mayo Clinic. "We bring the right people together and never
stop seeking a way forward," he said. "That's the power of multidisciplinary care. At Mayo Clinic, the patient's needs come first."
In his work, Dr. Omar has encountered many cases, but a few stand out as particularly memorable. "We had a malnourished patient from the MENA region with an end-organ disease," he recalled. "The patient had undergone surgery elsewhere and after losing excessive weight, was told they would likely need feeding tubes for the rest of their life." After reviewing the case, Dr. Ghanem determined it was reversible.
He referenced a Mayo Clinic study he authored on malnutrition after bariatric surgery. "It shows that if there's a correctable pathology, we can enable 87% of patients to stop feeding tubes or food by vein (also known as total parenteral nutrition) through corrective surgery." That proved true for this patient, who no longer needs feeding tubes and is back
home with their family, enjoying life. "This is the kind of story a surgeon remembers," Dr. Ghanem said, "because it changed someone's life."
While bariatric treatment can improve metabolic and longterm health, its role in abdominal wall reconstructive surgery is often overlooked. Dr. Ghanem explained that obesity and hernias are closely linked, as obesity is a major risk factor for developing hernias or experiencing recurrence after hernia repair. Mayo Clinic contributed to a Society of American Gastrointestinal and Endoscopic Surgeons (SAGES) study showing how obesity contributes to hernias and the best ways to address it.
"That often involves managing obesity through medications, endoscopic therapy, or bariatric surgery," he said. "Obesity can make hernia repair difficult because the bowels or stomach may protrude into the hernia." Mayo Clinic, he noted, specializes in these complex cases.
"Many of our surgeons and I perform both hernia and bariatric procedures," Dr. Ghanem added. "Hernia repairs have better outcomes when patients have lower weight, which is where bariatric surgery fits in. Most often, these

- Mayo Clinic Medical Director for middle East
procedures are done in several stages: first, the weight loss surgery, followed by hernia repair." Sometimes, he explained, they perform a temporary hernia repair until the patient loses weight, then complete the definitive repair when the patient is in optimal condition.
Looking at emerging innovations in bariatric surgery, Dr. Ghanem expressed excitement about combined therapy: anti-obesity medications followed by weight-loss surgery to help patients achieve health targets. "Integrating GLP-1 medications with surgery can be highly effective for patients with obesity. This approach to combining both medications and surgery is seen in the treatment of patients with cancer with chemotherapy and surgery."
He emphasized, however, that every patient with obesity is unique, allowing for personalized treatments. "Personalized medicine is very promising," he said. They have developed a calculator to predict weight loss and type 2 diabetes remission for patients who need bariatric surgery. "The Diabetes Remission Index (DRI) model uses a patient's baseline conditions and is available online for patients and providers worldwide."
Dr. Ghanem also led a study with Mayo Clinic, SAGES, and the American Society of Transplant Surgeons on the benefits of bariatric and metabolic surgery for patients who are candidates for transplantation. "Mayo Clinic research shows that simultaneous bariatric surgery and liver transplant can be safe for patients with severe obesity and can improve longterm outcomes. This approach addresses a critical gap, as many such patients do not qualify for lifesaving transplants due to their weight. Physicians from across the U.S. and around the world visit Mayo Clinic to learn our model."
At the end of the interview, Dr. Ghanem offered advice for patients in the Middle East considering bariatric surgery at Mayo Clinic. He recommended connecting with Mayo Clinic's Dubai office, which provides helpful information and links to their bariatric practice. Patients from the Middle East or Africa, or their providers, should reach out to the office, as should those seeking other Mayo Clinic services.
"If patients have Mayo Clinic appointments, the Dubai office can assist with travel arrangements and orientation," he said.
On a personal note, Dr. Ghanem highlighted Mayo Clinic's efforts to make international patients feel at home, including cultural sensitivity, international services, and free language assistance. "Many Mayo Clinic physicians hail from the Middle East, including surgeons."
"In Rochester, Minnesota, where I'm based, the city's culture welcomes people seeking care," he added. "You might even see Arabic signs as you walk around. For non-speakers, it may not seem like much, but for those who know the language, it's a powerful sign of welcome," he concluded. HCMEA







BY VINCENT MORANGA
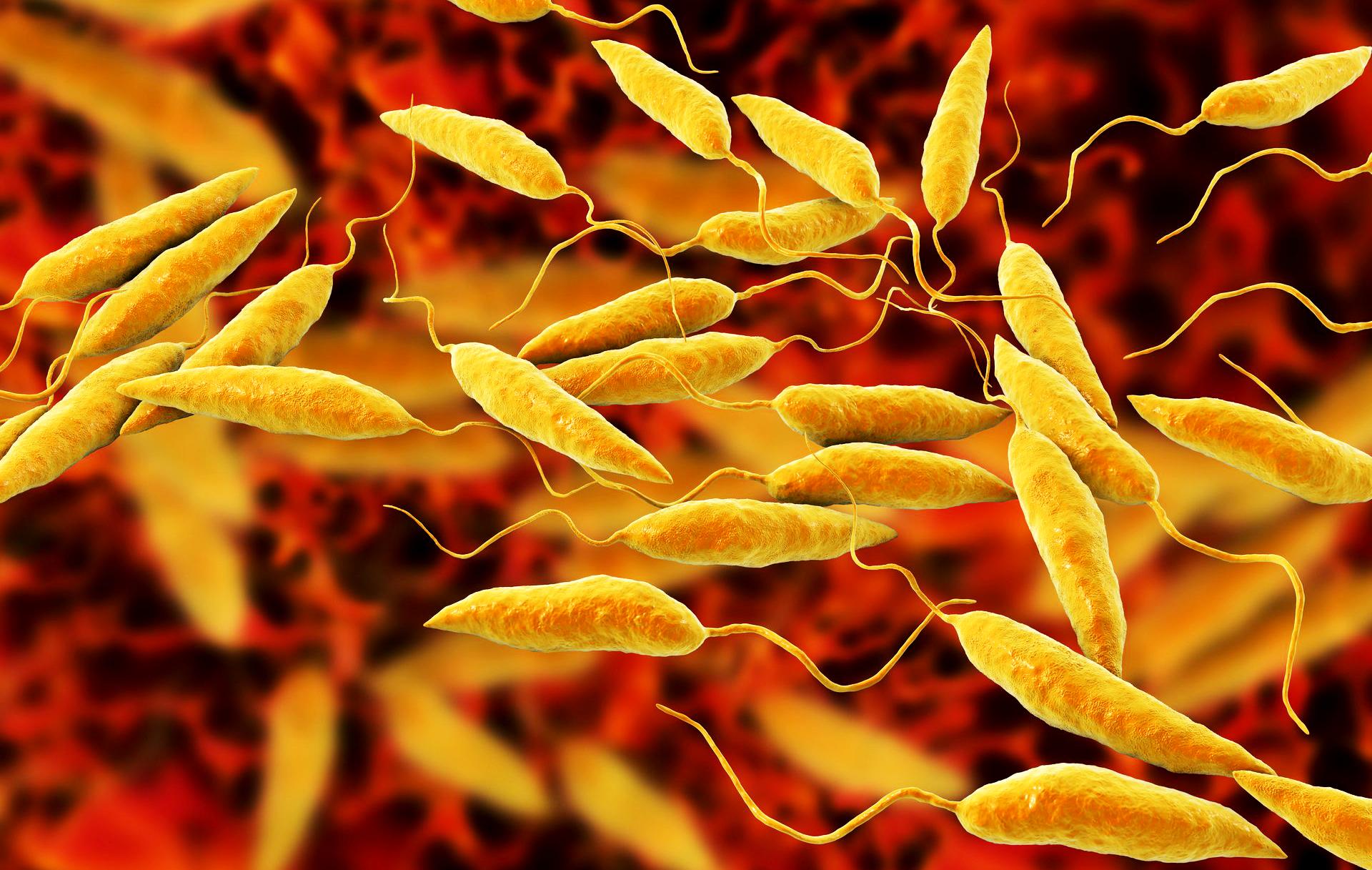
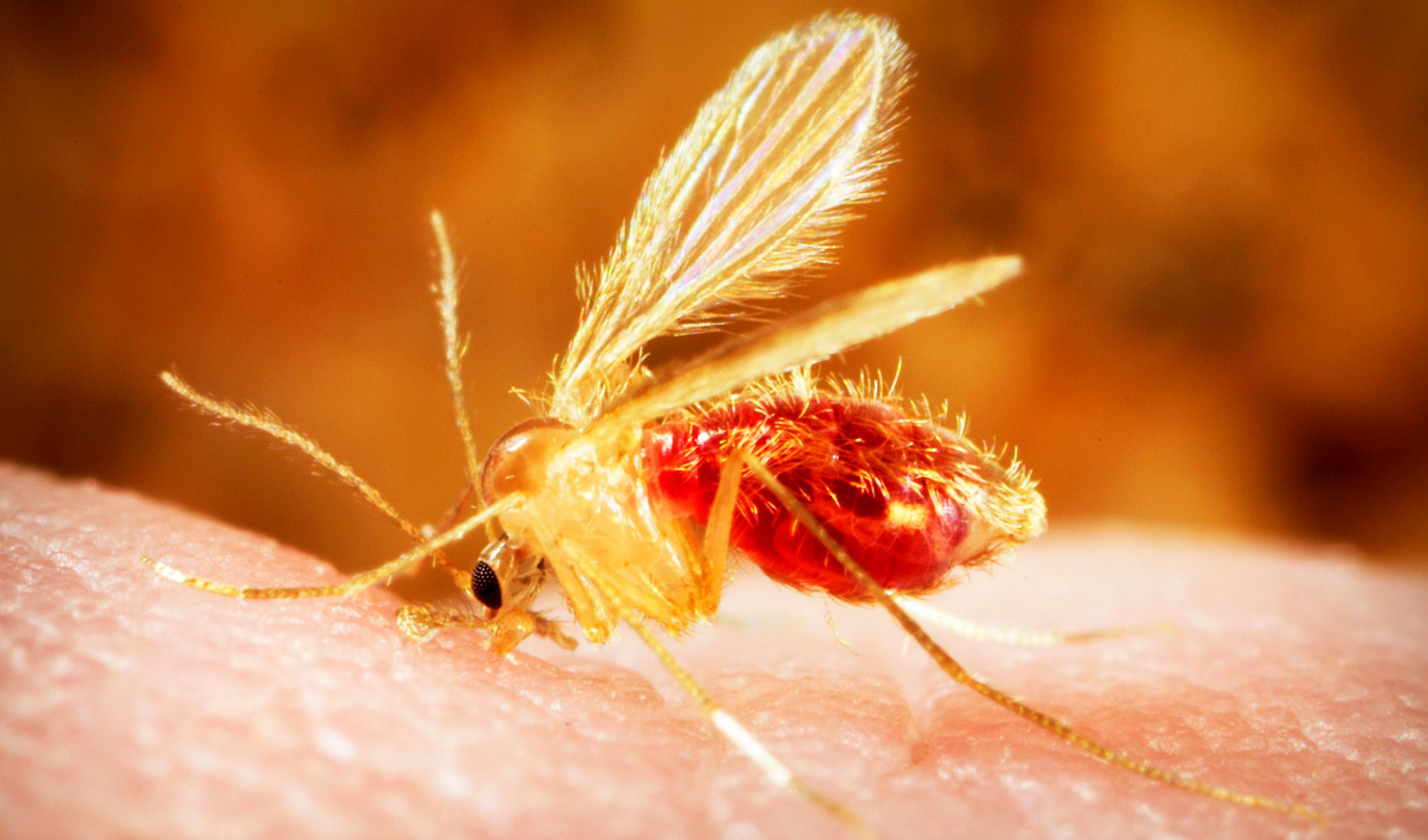
Kala-azar, medically known as visceral leishmaniasis (VL), remains one of the world’s most severe neglected tropical diseases. Caused by protozoan parasites of the Leishmania genus and transmitted through the bite of infected female sandflies, the disease attacks internal organs, primarily the spleen, liver and bone marrow.
Without treatment, visceral leishmaniasis is almost always fatal. Yet despite its high mortality rate, it is both preventable and treatable, making its continued burden a reflection of inequality, fragile health systems and limited access to care.
Globally, the World Health Organization estimates that between 700,000 and 1 million new cases of leishmaniasis occur each year, with visceral leishmaniasis accounting for an estimated 50,000 to 90,000 of those cases.
However, only about 25% to 45% of cases are officially reported, suggesting that the true burden is significantly higher. Kala-azar is endemic in more than 80 countries, primarily in Eastern Africa, South Asia, and Latin America. Approximately 85% of global VL cases occur in just seven countries: Brazil, Ethiopia, India, Kenya, Somalia, South Sudan and Sudan.
EPIDEMIOLOGY: WHY EASTERN AFRICA BEARS THE HEAVIEST BURDEN
Eastern Africa has emerged as the epicentre of the disease in recent years. In 2022, the region accounted for roughly 73% of global visceral leishmaniasis cases, with children under 15 years representing about half of those infections. The epidemiology of kala-azar in Africa is closely linked to poverty, malnutrition, population displacement, conflict and weak surveillance systems.
In Kenya, the Ministry of Health reports that kala-azar is now spreading to previously unaffected regions, becoming endemic with reported infections in Turkana, West Pokot, Baringo, Isiolo, Marsabit, and Wajir counties. According to the Kenya Health Information System (KHIS), Turkana County was the epicenter in 2025, with 2,043 cases, followed by West Pokot (1,913 cases) and Wajir (905 cases); Samburu recorded
just 4 cases.
On a global scale, there are between 50,000 and 90,000 new cases of kala-azar each year, with about 90 percent occurring in endemic regions such as Brazil, Ethiopia, India, Kenya, Somalia, South Sudan, and Sudan. Between 1989 and 2020, MSF teams treated nearly 150,000 people for the disease, with over a third of cases in South Sudan.
In arid and semi-arid areas, especially in border regions, seasonal migration and humanitarian crises exacerbate transmission by increasing exposure to sandflies and reducing access to timely diagnosis and treatment.
The disease thrives in environments where housing conditions allow sandflies to breed and where communities have limited protective measures. Climate variability and environmental changes are also influencing vector patterns, raising concerns that endemic zones could expand if preventive measures are not sustained.
Environmental and climate change have also led to increased drought, famine, and flooding, which drive people to migrate to areas with high parasite transmission. Climate change also affects the spread of leishmaniasis by affecting temperature and rainfall, which in turn influence the size and geographic distribution of sandfly populations. The breeding and biting rates of sandflies are also changing as climate and environmental conditions evolve.
"Climate change is expanding the range of sandflies and increasing the risk of outbreaks in new areas," says Dr Cherinet
Adera, a researcher at the Nairobi-based not-for-profit Drugs for Neglected Diseases initiative (DNDi) and its partners.
CLINICAL FORMS AND COMPLICATIONS
Leishmaniases are classified into three forms: visceral, cutaneous, and mucocutaneous. Visceral leishmaniasis is the deadliest, with a death rate of up to 95% if left untreated. Cutaneous leishmaniasis, the most prevalent type, typically causes skin ulcers, while mucocutaneous leishmaniasis affects the mouth, nose, and throat.
In 5–10% of cases, individuals treated for visceral leishmaniasis develop post-kala-azar dermal leishmaniasis (PKDL), a complication characterised by rash or skin lesions that may appear months to years after successful treatment.
Although not life-threatening, PKDL can be disfiguring and lead to stigma. Its lesions also serve as reservoirs for Leishmania parasites, thereby enabling ongoing transmission of kalaazar and complicating elimination.
COUNTRIES WITH LEISHMANIA-HIV COINFECTION
Kala-azar and HIV co-infection continue to be a significant challenge. The two diseases exacerbate each other in a vicious cycle, mutually attacking and weakening the immune system. Both suppress immunity, causing more severe morbidity, limited treatment options, higher relapse rates, exposure to toxic medicines, and increased mortality.
Médecins Sans Frontières (MSF) reports that mortality rates are up to 14 times higher in HIV co-infected and relapse patients, making timely diagnosis crucial for saving lives.
People living with HIV who contract leishmaniasis face a high risk of full-blown disease, relapse, and death. Antiretroviral therapy (ART) reduces disease progression, delays relapses, and improves survival. As of 2021, Leishmania-HIV coinfection had been reported in 45 countries. Coinfected patients are also vulnerable to comorbidities like tuberculosis and cryptococcal meningitis, as well as varying degrees of stigmatization and human rights violations.
Despite the scale of the challenge, important progress has been achieved, particularly in South Asia. Bangladesh has been validated for eliminating visceral leishmaniasis as a public health problem, while India has reported up to a 99% reduction in cases over the past decade in previously endemic districts.
These achievements were driven by sustained political commitment, aggressive case detection campaigns, widespread use of rapid diagnostic tests, and the scale-up of effective treatment regimens such as liposomal amphotericin B and combination therapies.
Integrated vector control strategies, including indoor residual spraying and the distribution of insecticide-treated bed nets, played a crucial role in interrupting transmission. The South Asian experience demonstrates that elimination is feasible when surveillance, treatment and prevention strategies are aligned and consistently funded.
In contrast, African countries are now
intensifying their efforts to replicate similar success. In 2023, eight Eastern African nations adopted the Nairobi Declaration, committing to eliminate visceral leishmaniasis as a public health problem by 2030. This political pledge was followed by the launch of a regional elimination framework targeting a 90% reduction in cases, bringing annual cases below 1,500 across the region by the end of the decade.
The framework also aims to ensure that at least 90% of patients receive treatment within 30 days of symptom onset and to eliminate deaths among children due to VL. Crossborder collaboration is central to this strategy, as migration between endemic areas often undermines isolated national interventions.
Countries such as Kenya and Ethiopia are expanding rapid diagnostic testing in remote health facilities, while fragile states including South Sudan and Somalia are strengthening partnerships with humanitarian agencies to improve access to treatment.
Scientific and operational innovation is reshaping the fight against visceral leishmaniasis (kala-azar), with pharmaceutical manufacturers, diagnostic developers and global health partnerships accelerating progress through improved therapies, rapid detection tools and smarter delivery systems.
A central driver of treatment innovation has been the Drugs for Neglected Diseases initiative (DNDi), which has worked with endemic-country governments and industry partners to develop safer, shorter and more effective combination therapies. These regimens are designed to reduce toxicity, shorten hospital stays and limit the emergence of drug resistance.
Among the most significant breakthroughs has been the expanded use of liposomal amphotericin B formulations, including products such as AmBisome. In South Asia, cure rates often exceed 95%, with far lower toxicity than older pentavalent antimonials.
Through negotiated pricing agreements and technology transfer arrangements with manufacturers, the cost of liposomal amphotericin B has fallen markedly over the past decade, improving affordability for national treatment programmes.
In Eastern Africa, where parasite strains and treatment responses differ, tailored combination regimens are being introduced to address emerging resistance. These are supported by strengthened pharmacovigilance systems that monitor safety and outcomes, signalling a broader shift from donor-dependent emergency responses toward more sustainable procurement and supply models integrated into national health systems.
Despite these advances, current therapies still present challenges. Pentavalent antimonials such as sodium stibogluconate and meglumine antimoniate require 30 days of injections. Miltefosine offers an oral option but must be taken twice daily for 28 days and is associated with tolerability
concerns.
Paromomycin involves three weeks of intramuscular injections. In many African settings, patients undergo painful daily injections for 17 days, with potential side effects affecting the heart, liver or pancreas.
Diagnostics innovation has progressed in parallel. Rapid tests such as the rK39 antigen-based assay now deliver results within 15–20 minutes at primary health facilities, enabling same-day treatment initiation and reducing loss to follow-up.
Operationally, digital surveillance platforms, geographic information systems and satellite-based environmental mapping are improving identification of sandfly habitats and transmission hotspots.
Meanwhile, manufacturers are refining long-lasting insecticide-treated nets and residual spraying formulations to enhance durability and vector control. Research into new oral therapies continues, with the goal of simplifying regimens further, enabling outpatient management and reducing hospitalization costs across endemic regions.
While momentum is building, major obstacles remain. Underreporting continues to obscure the true scale of the disease in parts of Africa. Climate change may alter sandfly distribution patterns, potentially expanding endemic areas. Conflict and displacement further complicate consistent delivery of health services in high-burden countries.
Nevertheless, kala-azar is increasingly regarded as eliminable as a public health problem. The dramatic decline in cases across South Asia provides a blueprint for success. If Eastern Africa sustains political will, strengthens surveillance systems, expands access to diagnostics and treatment, and maintains regional cooperation, a comparable reduction is within reach.
Ultimately, the fight against kala-azar is not merely a technical health intervention—it is a measure of global commitment to addressing diseases that disproportionately affect the poorest populations. With coordinated action and sustained investment, visceral leishmaniasis could shift from being one of the world’s deadliest neglected diseases to a public health success story by 2030. HCMEA
BY MARK ADAMS, CEO OF CMC HOSPITAL
Medicine is undergoing a pivotal shift, driven by technological innovation and a deepening respect for patient autonomy and quality of life. For much of modern medicine, progress has been defined by the ability to solve problems by removing the tumour, killing the disease, and extending survival. These goals have driven extraordinary advances in treatment and cure, and we marvel at them every day.
But something else is now happening alongside technological advancement. The focus has shifted from whether a problem is solvable to how it is solved, and whether the outcome preserves the patient’s quality of life. Recovery time, side effects, long-term quality of life and emotional wellbeing are no longer secondary considerations – they are central to what patients understand as good care.
This shift is being enabled by technology. Innovation has profoundly expanded what can be treated, and it is now expanding how care is provided. Precision treatments, such as imaging and less invasive options, mean clinicians can address disease while preserving what matters most to patients: dignity,

independence, and day-to-day quality of life. Urology, particularly prostate cancer care, sits at the heart of this development.
In the past, success in urology has been closely tied to the procedure. Surgical skill, technical accuracy and complete disease removal have been the primary benchmarks of achievement. And there’s nothing wrong with that – after all, this approach has saved countless lives and set the standard for care.
However, prostate conditions also involve difficult trade-offs. Traditional treatments, while destroying the disease, can have significant impacts on continence, sexual function, and recovery time. Patients have had to choose between treating the disease decisively and protecting their quality of life. Having it both ways has not been an option before now.
It’s not that no one cared or that clinicians simply didn’t think about this; it was a limitation of the tools available at the time. When surgery or radiation were the most effective options, decision-making naturally revolved around them.
However, new and varied treatments have changed the game, allowing a broader reframing of what “success” means.
Research shows that, when compared to more invasive treatments such as surgery or radiotherapy, focal therapies for prostate cancer often offer the patient an improved quality of life.
High-Intensity Focused Ultrasound (HIFU) is the latest in a new generation of prostate cancer treatment is designed with precision and restraint in mind. It works by blasting concentrated ultrasound energy to a clearly defined area of the prostate, rapidly heating and destroying targeted cancerous tissue while minimising damage to the surrounding body.
HIFU is a focused therapy that impacts only the treated area. Plus, it doesn’t involve incisions or radiation and, in many cases, can be performed as a day procedure.
This level of accuracy matters. By treating only what needs to be treated, HIFU allows clinicians to preserve the parts of the body that support urinary and sexual function. For patients with localised, low- to intermediate-risk prostate cancer, this can mean effective cancer control with fewer long-term side effects. And because there are no surgical incisions, there’s a reduced risk of infection and blood loss, matched with shorter hospital stays and faster recovery times compared to traditional surgery.
While HIFU doesn’t necessarily replace established treatments, it expands the spectrum of care. It gives clinicians the ability to tailor treatment intensity to disease characteristics and to the person living with them. And this shift is making all the difference.
As treatment options diversify and increase, patient priorities are coming into sharper focus. People are able to take some power back and understand not only whether a cancer can be treated, but also how treatment will affect their independence, relationships, and daily routines.
The result has been a much more collaborative approach to care, involving the patient in their own treatment. This benefits patient and doctor. Now, treatment planning can involve shared decision-making, where clinical evidence and patient values are discussed and considered together. This involvement helps give the patient a sense of empowerment and hope over their treatment.
The clinical evidence for HIFU is clear. Studies of HIFU in localised prostate cancer have demonstrated encouraging cancer control at medium-term follow-up, alongside lower rates of urinary incontinence and erectile dysfunction in appropriately selected patients.
Recovery experiences are also affected. Many patients return home the same day and resume normal activities far sooner than with conventional surgery. All of this reinforces a simple truth: when treatment respects the patient’s life, recovery is not just about a cure, it’s about a return to normalcy.
Patient-centric care doesn’t always mean defaulting to the latest technology. HIFU is not suitable for every patient, and aggressive or advanced disease may still require more extensive intervention.
But the ability to choose more precisely has changed everything. Today’s urologist isn’t limited to a single way forward – they have options and can weigh innovation against evidence and match treatment to each patient’s condition, lifestyle, and priorities. This responsibility requires clinicians to master new technologies while explaining them clearly and empathetically to ensure patients make informed, confident decisions.
Around the world, medical institutions are responding to this shift by investing in technologies that support less invasive, patient-centred care. CMC Hospital in Dubai is one such example, integrating advanced HIFU systems into its urology services and committing the necessary time, training, and resources needed to use them responsibly and effectively.
These investments are not just about keeping pace with medical advances, although that matters. They reflect a belief that progress in healthcare should be felt by patients themselves through quicker recoveries, fewer side effects, and greater confidence about what lies ahead.
Innovation is the root of progress in the medical field. But it only becomes meaningful when embedded thoughtfully into clinical pathways and supported by experience, collaboration, and outcomes monitoring.
The future of urology will not be shaped by a single breakthrough or device. It will be shaped by a philosophy –one that values precision, compassion and restraint in equal measure.
As technology advances, medicine can finally deliver cures that prioritise care, resolving medical issues without compromising the patient's future – a hallmark of the patientcentric innovation that is quickly becoming the standard in urology. HCMEA
BY
The 2026 African Union Summit has come and gone, along with numerous meetings and side events united by a common theme: health financing, local manufacturing, global health architecture reform, and advancing Africa's health security and sovereignty.
Having sat through enough high-level meetings, I know that the distance between a well-crafted declaration and a functioning diagnostic test at a primary health clinic in rural Gambia, Kenya, or northern Nigeria can feel infinite. We have all become good at articulating the problem and remarkably adept at the language of reform.
Five years ago, the 2021 Lancet Commission on Diagnostics reported that almost half the global population had little or no access to diagnostics, and that over a million premature deaths in low- and middle-income countries could be avoided annually by closing the diagnostic gap for just six priority conditions.
The 2024 Commission update found that advancement was slowest on recommendations with substantial resource implications, specifically labour force, affordability, and diagnostics in fragile settings.
The 2025 Global Diagnostics Gap Assessment, produced jointly by the International Pandemic Preparedness Secretariat, the Brown University Pandemic Centre, and FIND, identifies six major interconnected barriers across the diagnostics ecosystem, including underinvestment, delayed access to pathogen data, and the absence of clearly defined target product profiles. These are not technical puzzles but systemic failures.
Africa has not been silent. For seventeen years, the Maputo Declaration of 2008 made the moral case for quality-assured laboratory services as a pillar of health system strengthening. The Lusaka Agenda calls for transitioning out of donor dependency and places domestically financed primary health care as the cornerstone of health sovereignty.

The 2025 Accra Compact and the 2026 Addis Reckoning both call for moving from resilience to reliance, including prioritising regional manufacturing of diagnostics. The 2023 WHA Resolution on diagnostics, the first of its kind, formally recognised diagnostics as essential to universal health coverage. Ministers, presidents, and commissioners have all spoken with clarity and conviction.
Yet every day, a mother presents at a primary health clinic (PHC) with a febrile child and there is no rapid diagnostic test available. A clinician makes a presumptive diagnosis and prescribes accordingly, often incorrectly. This is not a hypothetical but the lived reality of hundreds of millions of people that no communiqué can mask.
In the WHO African Region, 40% of countries have no regulations for medical devices, and 60% lack national essential diagnostics lists that meet WHO recommendations. This is the backdrop against which we expect declarations to deliver change.
To move from rhetoric to action, we must acknowledge that a health system without a diagnostic backbone is not a system at all. The state of health in Africa is fundamentally a political decision. When a country allocates 4% of the national budget to health rather than the 15% pledged in Abuja, that is a choice.
When a Ministry of Health has an essential diagnostics list that has never been operationalised at the facility level, that is also a choice. Sovereignty without adequate domestic financing
is just a slogan. Health sovereignty cannot be donated, and it does not arrive with a declaration; it requires a budget line backed by law. So, what needs to be done?
National essential diagnostics lists must move from paper to practice. Too many countries have lists sitting in policy documents while primary health clinics operate without a single functioning rapid test. Implementation requires supply chain investment, training, quality assurance, and sustained procurement financing.
Communities need integrated testing at the primary health level for fever syndromes, maternal and neonatal conditions, and non-communicable diseases. While the technology exists, the political and financial commitment to deploy it consistently is lacking.
Perhaps the most significant silent barrier is the fragmented regulatory landscape. The newly operationalised African Medicines Agency holds immense potential to transform access to quality-assured diagnostics across 55 member states. Compared to medicines, the regulatory landscape for in vitro diagnostics is considerably weaker, and the AMA must prioritise IVDs as a core workstream.
The harmonised IVD registration pathways being built by Africa CDC's Diagnostic Advisory Committee and AUDANEPAD's AMRH initiative must also be accelerated, resourced, and politically protected. Third, market fragmentation must be addressed as a structural problem.
With 55 different procurement systems, African manufacturers face high entry costs and unpredictable demand. Pooled procurement, aligned with AfCFTA commitments and the Lusaka Agenda's single-plan approach, can create the demand certainty that incentivises investment. Without viable markets, local diagnostics manufacturing will remain aspirational.
Fourth, health financing must be treated as the emergency it is. The contraction of donor financing has exposed the
danger of aid-dependent health systems. African governments must urgently increase health budget allocations, protect diagnostic procurement specifically, and explore domestic financing mechanisms such as health levies, pooled insurance, debt swaps, and sovereign health funds capable of protecting essential services from external shocks.
The continental architecture, from the AMA to Africa CDC's Diagnostic Advisory Committee to the Global Diagnostics Coalition, is more coherent now than at any point in the past two decades. As the 2024 Lancet Commission update rightly observed, greater progress has occurred where political will is aligned with industrial policy goals.
We are not just approaching a diagnostics crisis in Africa. Truth be told, Africa has been living through one for generations. The mother with the febrile child at the PHC is not waiting for the next summit. She is waiting for a test.
At FIND, our commitment is to #DiagnosisForAall, not as a hashtag but as a measurable, deliverable outcome. FIND is here to support Africa's continental aspirations and to work alongside governments, regulators, manufacturers, and communities to turn the ambitions of the Lusaka Agenda, the Accra Compact, and the WHA Resolution into diagnostics that reach patients. Africa has waited long enough. HCMEA


BY MARK ADAMS, CEO OF CMC
In Dubai's competitive private healthcare market, where local and international patients choose between, what are essentially, clinically equivalent facilities, the differentiator is no longer what care is delivered, it's how it feels to receive it.
Among the most frequent and emotionally charged touchpoints in any hospital stay isn’t doctor care or comfort, it’s food. Not as surprising as it first seems. Meals arrive multiple times daily and is one of the few moments of personal agency in an otherwise controlled and restricted clinical environment. Despite this, food service remains undervalued: treated as operational overhead rather than what it actually is — a powerful, recurring signal of whether an institution sees patients as people or as cases.
The evidence is unambiguous. Poor food quality isn’t just about a disappointing dinner, it impacts the entire care process. In a healthcare landscape where patient choice drives revenue and reputation, catering is integral to the experience of care, and to the business model that depends on it.
Hospital food is typically written off as overhead — something to be managed, not improved. But the data tells a different story. When patients are unhappy with their meals, they're also more likely to report problems with their care overall, experience complications, and stay in hospital longer. A multi-hospital study published in Clinical Nutrition ESPEN found that
patients who didn’t like their food were more likely to report negative outcomes across the board compared to those who said the food was at least acceptable.
There's a clinical dimension too. Poor food service has been linked to higher rates of malnutrition during hospital stays, which slows recovery and extends length of stay. Patients who were more satisfied with what they were eating and how it was served, showed lower malnutrition risk while admitted.
What this suggests is straightforward: food doesn't just affect mood. It shapes how patients experience and recover from care.
What makes hospital food "good" isn't just whether it tastes acceptable. Research using service-quality frameworks shows that satisfaction is driven by a cluster of factors: food quality, yes, but also perceived value, variety, and, critically, how staff deliver it. Empathy during meal service is as strong a predictor of satisfaction as the food itself.
This matters because these aren't really food issues. They're dignity issues. When a patient is offered a choice, even one limited by dietary restrictions or clinical safety, it reminds them of something important: that they're still a person with opinions and preferences, not just a condition being managed. In an environment where nearly everything else is decided for them, the question "what would you like for lunch?" becomes a small but meaningful restoration of agency.
Hospitals that treat catering as purely logistical are missing the point. The tray isn't just delivering nutrition. It's delivering a message.
Patient satisfaction with hospital catering is multi-factorial. Research using established service-quality frameworks shows that food quality, perceived value, meal variety and staff empathy are all significant predictors of satisfaction.
Importantly, these factors mirror broader expectations about dignity and respect. Choice — even when limited by clinical safety — signals that patients are being listened to at a time when control over many other aspects of life has been surrendered.
As we talk about experience, let's be clear: in healthcare catering, safety isn't negotiable. Hospitals serve medically vulnerable people with specific nutritional needs — allergies, texture-modified diets, chronic disease management. Get any of that wrong and you're not just disappointing someone, you're putting them at risk.
UAE healthcare accreditation, standards require hospitals to maintain documented systems for safe food preparation, hygiene, balanced nutrition, allergy management and integration with dietetic services. Dubai Health Authority, facility guidelines go further, emphasising that catering must address diverse dietary, cultural and religious needs while remaining clinically appropriate.
When these fundamentals fail and meals are incorrect, unsafe or unsuitable, patient confidence collapses, no matter how good the clinical care.
In hospital kitchens, mistakes aren't just inconvenient. They can be dangerous. Allergy management, food consistency and condition-specific diets are among the most operationally complex and high-risk areas of non-clinical hospital services.
Take allergies. If someone in the kitchen misses a note that should have come from the ward, a patient could end up in serious trouble — and any trust in the system evaporates immediately. This isn't something you can manage with good intentions. It requires clear protocols, proper training and workflows that actually connect clinical and catering teams.
Texture-modified diets are another high-stakes area. Patients who've had strokes, swallowing difficulties or certain surgeries depend on food being prepared to an exact consistency — too thick or too thin and they risk choking or aspiration. Get it wrong once and the consequences are real.
Then there's diabetes. Meals need to arrive on time, and portions need to be consistent, or blood sugar becomes unstable. Miss a meal window or send the wrong portion and you're not just inconveniencing someone, you're potentially slowing their recovery and keeping them in hospital longer.
In Dubai, where healthcare institutions serve international, multilingual populations with vastly different food expectations, managing these risks while delivering culturally appropriate meals requires robust systems and tight clinical alignment. Catering here isn't hospitality. It's part of the care pathway.
And while all of this should be baseline, the reality is that building genuine trust and dependability in this area can still set an institution apart in a fiercely competitive market.
Dubai’s inpatient population is unusually diverse. Emirati nationals, expatriate residents and international medical travellers bring distinct cultural, religious and personal food expectations. Religious considerations such as halal
compliance, pork-free preparation and ingredient transparency are baseline expectations within UAE hospitals.
Hospital catering providers in the UAE routinely incorporate cultural preferences, allergen controls and menu diversity into their operational models to ensure meals feel familiar and supportive during recovery.
Dubai’s private healthcare sector increasingly serves highermiddle-income and affluent patients who actively choose where to receive care. For this cohort, clinical competence is assumed. Decision-making is instead shaped by trust, reassurance and alignment with the service standards they experience in luxury hospitality, international travel and premium outpatient care.
Research into private healthcare choice shows that affluent patients use non-clinical cues — environment, service consistency and attention to detail — as proxies for underlying quality, particularly in moments of uncertainty such as hospital admission or surgery. Food service sits squarely within this evaluative framework because it is frequent, personal and highly visible.
In more and more premium institutions, catering is not positioned as an amenity but as an extension of the brand’s clinical philosophy. Menu design, service interaction and presentation are deliberately aligned with values of precision, empathy and reassurance. This may include culturally familiar comfort foods prepared within strict clinical parameters, clearly communicated nutritional information, and hospitality cues such as afternoon tea or enhanced family meal options for long-stay patients.
These touches are not about indulgence. They are about reducing anxiety. Studies in patient experience consistently show that when non-clinical services feel organized and thoughtful, patients infer that the same discipline applies to clinical care. Food becomes a daily reinforcement of competence.
Family members play a critical role in this perception. In private healthcare, families often act as decision-makers, advocates and reviewers. Relatives interpret food quality and meal service as indicators of how well the hospital is managing invisible aspects of care (source). A poorly delivered meal can undermine confidence disproportionately; a well-executed one reassures.
Premium hospitals therefore invest in catering systems that deliver predictability and respect rather than novelty: meals that arrive on time, are clearly appropriate for the patient’s condition, reflect cultural norms, and are served with professionalism. In Dubai, where many patients are far from home, familiar flavours and considerate service can provide emotional grounding during recovery.
In this segment of the market, catering becomes part of the hospital’s brand architecture. It signals organisational maturity, reinforces trust among patients and families, and
quietly supports the perception that the hospital is worthy of both confidence and cost.
Digital meal-ordering platforms are increasingly deployed in Middle Eastern healthcare settings to embed dietary safety and patient preferences directly into catering workflows. When integrated with clinical records, these systems reduce errors, improve compliance and increase patient engagement.
Technology is not a luxury in this context, it is a service multiplier that enables consistency at scale.
Differentiation in healthcare catering is rarely about spectacle or culinary indulgence. It's about frictionless, reliable service design that respects the patient's dignity throughout the day.
An experience-oriented catering system is designed around how patients actually live their hospital routine. That means clear communication about when meals will arrive. Predictable delivery that aligns with clinical schedules, not kitchen convenience. Respectful interaction during service — not just polite, but attuned. And menu options that reflect clinical needs, cultural expectations and personal preferences, not just what's easiest to batch-produce.
These aren't marketing gestures. They're operational choices. And research into hospital food service consistently shows that predictability, empathy and perceived value influence how patients judge not just their meals, but the organisation delivering them.
Patients may not consciously articulate these details, but they feel them, and they carry those impressions into their overall assessment of care.
When talking about differentiation in catering, it’s easy to think of sophisticated menus, lavish service or unique ingredients, but that is simply not the case. In healthcare markets where clinical competence is assumed, differentiation lives in small, repeated moments that patients remember long after discharge. Catering is an opportunity to show the customer that their agency still matters, that their mental wellbeing is being considered as well as their physical health, and that they are seen as individuals with individual needs.
When hospitals understand meals as part of care, rather than a line item, they reinforce trust, confidence and brand credibility. In Dubai’s private healthcare sector, where patients actively compare experiences and expectations are high, these details matter.
In markets where outcomes converge, it is experience, delivered unobtrusively and consistently, that wins loyalty, reputation and long-term advantage. HCMEA



BY ALPHONSE OKOTH
In every tablet swallowed, capsule dispensed or suspension measured, there is an invisible foundation that determines whether a medicine performs as intended. Excipients, often overlooked by patients and even policymakers, are essential to drug safety, stability and efficacy. As East Africa’s pharmaceutical sector expands and matures, the quality, reliability and regulatory compliance of these inputs have never been more critical.
Positioned at this crucial intersection is Scivion Limited, a pharmaceutical excipient distribution and technical support company quietly shaping how medicines are manufactured across the region. With a clear focus on compliance, trusted partnerships and long-term ecosystem development, Scivion is helping local manufacturers meet global standards while
keeping patient safety firmly in focus.
Scivion Limited was founded with a defined purpose: to bridge global excipient innovation with the practical needs of East Africa’s pharmaceutical manufacturers. The company’s origins lie in a recognition that inconsistent access to compliant excipients posed a systemic risk, not only to manufacturers, but ultimately to patients.
“Scivion was established to ensure a compliant, reliable and sustainable supply of excipients for manufacturers and patients across East Africa,” the company explains. “Our mission is to provide industry-leading excipients supported by technical expertise, regulatory alignment and authentic

partnerships.”
From the outset, Scivion positioned itself as more than a distributor. Its operating model reflects an understanding that excipients are not interchangeable commodities; they are regulated pharmaceutical inputs that demand technical knowledge, documentation and accountability.
What distinguishes Scivion in a competitive and often fragmented market is its integrated approach. The company pairs excipient distribution with technical support and regulatory guidance, ensuring that manufacturers receive both materials and the expertise required to use them effectively.
“Our differentiation lies in combining product supply with technical and regulatory support,” Scivion notes. “Clients gain access not only to high-quality excipients, but also to formulation guidance and compliance assurance.”
This strategy has underpinned the company’s growth. Since inception, Scivion has evolved from a local distributor into a regional partner serving manufacturers across East Africa. Its expansion has been marked by portfolio diversification, enhanced logistics capabilities and the integration of technical services into its core operations.
Today, Scivion operates with a regional outlook, emphasizing scale, efficiency and regulatory consistency, key
requirements as East African manufacturers increasingly target both domestic and export markets.
Central to Scivion’s value proposition is its collaboration with JRS Pharma, a globally recognized leader in excipient innovation. Through this partnership, Scivion provides East African manufacturers with access to internationally validated excipients supported by local technical and regulatory expertise.
Scivion’s portfolio includes microcrystalline cellulose grades, superdisintegrants, co-processed excipients, functional fillers, lubricants and coatings. These materials are designed to enhance formulation performance, stability and patient safety across a range of dosage forms.
“Our collaboration with JRS Pharma ensures manufacturers benefit from globally recognized innovation while remaining compliant with regional regulatory requirements,” the company says.
Partner selection is governed by strict criteria. Scivion works exclusively with suppliers that demonstrate robust compliance, consistent quality, proven innovation and a commitment to sustainability.
“We prioritize partners whose products enhance patient safety, ensure regulatory compliance and support longterm trust in the regional pharmaceutical market,” Scivion emphasizes.
Recent additions to Scivion’s portfolio include advanced coprocessed excipients and functional fillers from JRS Pharma. These solutions address persistent manufacturing challenges by improving flowability, compressibility and overall tablet performance.
“These innovations simplify manufacturing processes while improving dosage form consistency and patient adherence,” Scivion explains.
For manufacturers operating in cost-sensitive and highly regulated environments, such efficiencies are significant. Simplified processing reduces production variability, supports
scale-up and enhances compliance, while improved dosage forms contribute directly to patient confidence and therapeutic outcomes.
Quality assurance underpins every product supplied. By sourcing exclusively from JRS Pharma, Scivion ensures excipients are supported by rigorous audits, comprehensive regulatory documentation and continuous technical support aligned with international and local standards.
Ensuring consistent supply across multiple East African markets requires careful coordination. Scivion has implemented streamlined logistics planning, digital order tracking and regional distribution hubs to support timely and reliable delivery.
“Proactive inventory management and close coordination with our manufacturing partner are essential to minimizing disruptions,” the company notes. “Manufacturers depend on consistency to maintain compliant production schedules.”
Despite these systems, structural challenges remain. One significant barrier is the absence of adequate HS code listings for certain pharmaceutical excipients within the East African Community.
“Inadequate HS code classification hinders the trade of essential excipients,” Scivion states. “We avoid distributing products without definitive HS codes and actively engage regulators to address these gaps.”
Counterfeit excipients represent another serious risk to patient safety and market integrity.
“Products falsely represented as branded excipients undermine trust and pose direct risks to patients,” the company cautions. “We enforce strict compliance, collaborate closely with JRS Pharma and advocate for stronger regulatory action against counterfeiting.”

In an environment of increasing regulatory scrutiny, Scivion treats compliance as a strategic priority rather than a procedural obligation. Every excipient supplied is supported by


comprehensive documentation, quality audits and alignment with both EAC and international pharmaceutical guidelines.
Beyond compliance, the company is actively engaged in advancing technical capability within the region. In collaboration with JRS Pharma, Scivion supports ongoing R&D initiatives focused on multifunctional excipients and digital tools for excipient selection.
Manufacturers also benefit from access to a specialized global technical team providing formulation advice, excipient application support and regulatory guidance across development and production stages.
Scivion anticipates continued transformation in East Africa’s pharmaceutical distribution landscape. Regulatory harmonization within the EAC, increased digitalization and heightened enforcement against counterfeits are reshaping operational expectations.
“Growth in generics and specialty excipients will drive demand,” the company observes. “However, success will depend on stronger regulatory frameworks, standardized classifications and enhanced supply chain transparency.”
Emerging technologies, including blockchain-based traceability and advanced excipient platforms, are expected to further influence procurement, compliance and quality
CLIENTS
SCIVION LIMITED EXPLAINS
assurance practices.
Scivion’s response is rooted in agility and partnership. “We remain competitive by enforcing compliance, collaborating with trusted global partners and adapting quickly to regulatory and technological change,” it says.
Sustainability is embedded in Scivion’s sourcing and advocacy strategy. By working exclusively with compliant global manufacturers and promoting responsible supply chains, the company aligns environmental stewardship with patient safety and regulatory integrity.
“Responsible sourcing and long-term market integrity guide our operations,” Scivion explains. “Sustainability, for us, is inseparable from compliance and quality.”
Looking ahead, Scivion’s strategic priorities for the next three to five years include expanding excipient distribution across East Africa and into Francophone West Africa. This growth will be supported by deeper technical collaboration with global partners, enhanced digital tools and continued regulatory advocacy.
Planned initiatives include advanced excipient solutions, expanded technical support services, digital excipient selection platforms and blockchain-based traceability systems to improve transparency and efficiency.
“Our objective is to support manufacturers with reliable access to compliant, innovative excipients while strengthening regulatory alignment across emerging markets,” the company says.
While excipients rarely attract public attention, their role in healthcare delivery is fundamental. Through its focus on quality, compliance and partnership, Scivion Limited is reinforcing the foundations of pharmaceutical manufacturing in East Africa.
By connecting global innovation with regional expertise, the company is contributing to safer medicines, stronger manufacturers and a more resilient healthcare ecosystem, one formulation at a time. HCMEA

BY ALPHONSE OKOTH
When a hospital pharmacy runs out of a critical antibiotic or a cancer center delays treatment because of a drug shortage, the consequences are immediate and deeply personal. Behind every stockout is a supply chain that failed to absorb a shock. In pharmaceutical manufacturing, supply chain resilience is not just a business strategy, it is a matter of patient safety and public trust.
Over the past few years, the industry has been tested like never before. The COVID-19 pandemic, geopolitical tensions, trade restrictions, energy crises, and extreme weather events have all exposed the fragility of a supply model built for efficiency rather than disruption.
According to a 2023 survey by the Parenteral Drug Association (PDA), more than 75% of pharmaceutical manufacturers reported moderate to severe supply chain
disruptions during the pandemic, while 60% experienced raw material shortages that directly affected production schedules. What was once considered a lean, optimized system revealed hidden vulnerabilities, especially when demand surged or transportation routes stalled.
Today, resilience has moved from boardroom theory to frontline priority. A 2024 Deloitte survey of life sciences executives found that nearly 80% ranked supply chain resilience and risk visibility among their top three operational priorities. The message is clear: continuity of medicine supply is as critical as innovation in drug discovery.
One of the most pressing challenges lies in the heavy geographic concentration of active pharmaceutical ingredient (API) production. Data from the U.S. FDA indicate that
approximately 70% of API manufacturing facilities supplying the U.S. market are located outside the country, with China and India accounting for a significant share.
The European Medicines Agency has highlighted similar dependencies across EU supply chains. While this model reduced expenses, it also created vulnerability.
When factories shut down during lockdowns or export restrictions were imposed, supply bottlenecks rippled across continents. The American Society of Health-System Pharmacists recorded hundreds of active drug shortages at the peak of pandemic disruption, many involving sterile injectables and essential generics. The problem was not lack of scientific capability, it was structural overreliance on limited sources.
In response, companies are rethinking sourcing strategies. McKinsey’s 2023 pharma operations survey found that over 65% of manufacturers are actively qualifying secondary API suppliers, and nearly half are investing in regional manufacturing footprints.
The global pharmaceutical CDMO market, projected to exceed US$200 billion by 2030, is seeing increased investment in North America, Europe, and parts of the Middle East. For many leaders, paying slightly more for redundancy is preferable to risking prolonged shortages.
For many manufacturers, resilience is no longer theoretical, it is measurable. Pfizer accelerated rollout of its Trade Exchange Management Platform and Operations (TEMPO), integrating data across suppliers, manufacturing sites, and logistics partners. Industry case analyses report a 25% reduction in supply chain disruptions, a 15% reduction in inventory holding costs, and a 30% improvement in supplier compliance following implementation.
Merck redesigned its global network after experiencing major operational disruption from a cyberattack several years ago. By diversifying production across multiple regions and deploying AI-driven forecasting tools, Merck reduced future vulnerability by an estimated 50–60% and improved planning accuracy, lowering emergency logistics expenses.
GSK invested in IoT-enabled monitoring systems and enhanced end-to-end visibility tools across its manufacturing footprint. Internal reporting shared in industry forums suggests measurable reductions in disruption response times and improved compliance performance following digital integration.
Meanwhile, Novartis has outlined more than US$20 billion in U.S. manufacturing expansion over five years to reduce geographic concentration risk, while AstraZeneca, Eli Lilly, Johnson & Johnson, and Roche have collectively announced multi-billion-dollar regional manufacturing investments to strengthen supply continuity.
Distribution partners are reinforcing the network as well. Cencora has committed approximately US$1 billion to expanding U.S. distribution and cold-chain infrastructure,
supporting the growing volume of temperature-sensitive biologics.
Biotechnology leaders such as Amgen are leveraging AI-driven demand forecasting and digital twins to enhance manufacturing responsiveness. Industry benchmarks suggest AI-enabled planning can reduce logistics costs by up to 10% and improve forecast accuracy by 20%, directly strengthening resilience during volatility.
Resilience does not stop at sourcing. Manufacturing itself must be adaptable. An ISPE survey found that nearly 40% of manufacturers cited limited production flexibility as a constraint during emergency demand spikes.
Continuous manufacturing offers a more flexible alternative. By enabling real-time monitoring and faster scale adjustments, it reduces downtime and improves quality oversight. The FDA has encouraged adoption of continuous processes, citing improved reliability and reduced batch failure risks.
Digital twins allow manufacturers to simulate disruptions before they occur. Deloitte reports that predictive maintenance enabled by AI can reduce equipment downtime by 30–50%. Automation and robotics further reduce dependency on manual labor during workforce shortages.
The pharmaceutical manufacturing technology market is projected to grow at a CAGR exceeding 8% through 2030, fueled by investments in smart factories and advanced analytics. Increasingly, resilience is tied directly to digital maturity.
A resilient supply chain must also be secure and transparent. The World Health Organization estimates that one in ten medical products in low- and middle-income countries may be substandard or falsified. Regulatory frameworks such as the U.S. Drug Supply Chain Security Act and the EU Falsified Medicines Directive mandate serialization and traceability.
According to industry data, more than 90% of U.S. manufacturers have completed serialization upgrades to meet DSCSA milestones. Blockchain pilots and enhanced digital verification tools are strengthening end-to-end visibility.

Cybersecurity is equally critical. IBM’s Cost of a Data Breach Report consistently ranks healthcare as the most targeted sector, with average breach costs exceeding US$10 million. For pharmaceutical manufacturers, protecting operational technology systems is essential to maintaining uninterrupted production.
A 2023 EY life sciences report found that 55% of pharma companies are revising inventory policies to increase safety stock for critical medicines. Advanced forecasting tools are improving demand accuracy by up to 20%, according to Gartner.
The pharmaceutical supply chain management market is projected to expand at 7–9% CAGR through 2030, reflecting rising investment in digital control towers that provide end-to-end visibility. McKinsey estimates companies implementing such systems can reduce disruption response times by up to 40%.
Collaboration is also expanding. Regulatory
agencies are strengthening early-warning systems for shortages, while public-private partnerships are building regional production capacity, particularly for vaccines and essential generics.
Drug shortages are not abstract metrics, they are delayed treatments and anxious patients. Healthcare providers report spending hours each week managing supply gaps and identifying alternatives.
The cost of inaction is measured in health outcomes and trust. Conversely, measurable gains, reduced disruption rates, improved compliance, stronger forecasting, and diversified production, translate into more reliable access to medicines.
As volatility becomes the norm, resilience is no longer optional. It is a strategic investment and an ethical responsibility. In pharmaceutical manufacturing, safeguarding the supply chain means safeguarding lives. HCMEA







A dominant multinational player, GSK leads through strong vaccine, respiratory, and chronic disease portfolios. Backed by global R&D muscle and a robust regional distribution network, it remains a benchmark for pharmaceutical excellence in Kenya.
With roots in Boots International and now part of the Shelys Group, Beta Healthcare is a regional manufacturing powerhouse. Its Nairobi-based facility produces a wide range of prescription and OTC medicines serving East and Central Africa.
Cosmos has built its reputation on accessibility. By prioritizing essential medicines and nationwide availability, it continues to play a critical role in bridging healthcare gaps across both urban and rural Kenya.
A key generics and antibiotic producer, Dawa operates advanced penicillin manufacturing facilities and maintains a growing footprint across multiple African markets, reinforcing its regional growth trajectory.
5
Founded in 1981, Regal Pharmaceuticals brings over four decades of manufacturing expertise. Its focus on high-quality essential medicines has cemented its credibility across Eastern and Central Africa.
Distinguished by its integrated model, Laboratory & Allied combines pharmaceutical manufacturing with diagnostics and laboratory services, offering comprehensive solutions to hospitals and research institutions.
Universal Corporation’s diversified portfolio spans prescription and OTC medicines. Its broad therapeutic reach and nationwide distribution underpin its resilience in a competitive market.
Specializing in complex formulations, Elys Chemical merges chemical and pharmaceutical manufacturing expertise to serve niche therapeutic demands within Kenya’s healthcare system.
Representing the industry’s newer generation, Nextgen emphasizes modern manufacturing practices and innovation-driven product development tailored to evolving healthcare needs.
With a specialized focus on orthopedic and medical solutions, Orthomedics strengthens healthcare delivery in Western Kenya and supports niche treatment segments often underserved by larger manufacturers.
UAE — FUJIFILM Middle East FZE has unveiled its Interventional Cardiology Business Line across the Middle East and Africa (MEA), ahead of WHX Dubai 2026.
The initiative aims to expand access to advanced cardiovascular care in a region facing escalating heart disease rates.
The launch coincides with a strategic partnership with Innvolution Healthcare, a specialist in precision diagnostics
and personalized cardiovascular treatments.
Together, they will equip hospitals and clinicians with cutting-edge interventional cardiology technologies, including cardiac catheterization labs, angiography systems, Optical Coherence Tomography (OCT) devices, balloon catheters, drug-eluting stents, and a range of interventional accessories. FUJIFILM will integrate these offerings with its existing cardiovascular IT solutions, cardiac CT scanners, and ultrasound systems, ensuring comprehensive and accurate patient care.
Shin Tagawa, Managing Director of FUJIFILM Middle East, highlighted the urgency of addressing cardiovascular disease, which claims over 1.4 million lives annually in the Middle East and North Africa, a 48% rise over three decades, and over 1 million lives in Africa each year.
The partnership with Innvolution Healthcare seeks to fill this critical gap by delivering reliable, advanced interventional solutions to clinicians and patients.
Gaurav Agarwal, MD of Innvolution Healthcare, emphasized that the collaboration will elevate cardiac care standards across MEA, supporting better patient outcomes through cuttingedge imaging, interventional solutions, and clinical support.
The portfolio features industry-leading technologies, including compact, floor-mounted interventional imaging systems with AI-enhanced stent visualization, high-resolution OCT systems for precise vascular imaging, and Everolimus drug-eluting stents with innovative 60-micron strut designs that optimize insertion, blood flow, and vessel conformity.
UAE — Zoya Technologies has unveiled ZoyeMed 3.0, an edgenative clinical terminal that provides autonomous primary and acute care with human oversight, at WHX Dubai 2026.
The system delivers high-fidelity diagnostics without constant cloud connectivity, combining on-device artificial intelligence, multimodal sensing, point-of-care testing, and longitudinal patient modeling.
This allows healthcare providers to maintain consistent, protocol-driven care even in settings with unreliable internet access.
ZoyeMed 3.0 represents a major architectural advancement over previous models. Its closed-loop, edge-first design separates real-time safety and triage functions from more complex reasoning processes.
This enables the system to track patient health over time, identifying patterns and changes that might otherwise go unnoticed, while functioning effectively amid bandwidth limitations or staffing shortages.
Since 2025, Zoya Technologies has shipped 44 ZoyeMed 1.0 units, with active deployments in Mexico and Colombia
under multi-year contracts. The first ZoyeMed 3.0 unit has already been delivered to Bogotá ahead of pilot deployment.
Unlike conventional telemedicine platforms or AI copilots, ZoyeMed 3.0 is designed as physical healthcare infrastructure.
It integrates diagnostics, decision support, and follow-up care into a compact, on-site unit, reducing clinicians’ cognitive burden and enabling standardized, evidence-based care across multiple locations.
Dr. Syed Sabahat Azim, CEO of Zoya Technologies, highlighted that the edge-based design brings clinical intelligence closer to patients while building a longitudinal view of their health.
Following the WHX Dubai launch, the company plans staged deployments across Latin America, the Middle East, and South Asia, targeting regions with limited healthcare access and developing connectivity infrastructure.
ZoyeMed 3.0 positions Zoya Technologies at the forefront of edge-based clinical intelligence, promising consistent, high-quality care in challenging healthcare environments worldwide.
UAE — Insulet Corporation, a global leader in tubeless insulin pumps, has introduced its Omnipod 5 Automated Insulin Delivery (AID) System across the Middle East.
Pat Crannell, Insulet’s Senior VP and International GM, highlighted the launch as a milestone for a region with substantial unmet diabetes care needs.
The introduction coincides with Omnipod Discover, a new data analytics platform that simplifies glucose and insulin management for patients, caregivers, and healthcare providers. Together, the systems aim to reduce the daily burden of diabetes.
Omnipod 5 automatically adjusts insulin every five minutes, helping improve time in range and lower HbA1c levels.
The tubeless, waterproof, and discreet device communicates directly with a sensor to prevent high and low blood sugar levels throughout the day and night. Patients can wear it during normal activities, including swimming and exercise.
The system is available for individuals aged two and above with type 1 diabetes and integrates with Abbott FreeStyle Libre 2 Plus and Dexcom G7 sensors in Saudi Arabia, Kuwait, and the UAE; in Qatar, it pairs only with Dexcom G7.
Omnipod Discover converts complex glucose and insulin data into actionable insights through an intuitive web-based platform, helping patients and providers make informed decisions.
The Middle East is the first region to receive this platform, with plans to expand globally.
The launch expands Omnipod 5’s presence to 19 countries, with Spain set for 2026, followed by Greece and Croatia in early 2027.
Medical experts, including Dr. Mohammed Almehthel of King Fahad Medical City, hailed the system as transformative for diabetes care in the region.

ISRAEL — Bendit Technologies has secured U.S. Food and Drug Administration (FDA) clearance for its Bendit17 steerable microcatheter, expanding options for clinicians performing complex neurovascular and peripheral endovascular procedures.
The device features proprietary real-time 3D steering technology and can be used with or without a guidewire, enhancing precision and navigation through challenging vascular pathways. Preclinical studies demonstrated successful aneurysm access and vessel recanalization.
Bendit plans to begin U.S. clinical use in January 2026 while pursuing additional global approvals.
The neuro microcatheter market is projected to grow at a 5.4% CAGR, reaching US$792 million by 2034, reflecting rising demand for advanced interventional devices.
USA — Roche has received U.S. Food and Drug Administration (FDA) 510(k) clearance for a rapid point-of-care PCR test to detect pertussis (whooping cough) and other Bordetella infections.
The test also secured CE IVDR certification in Europe and a CLIA waiver in the U.S., broadening access across healthcare settings.
Run on Roche’s cobas liat system, the assay delivers results in 15 minutes, enabling samevisit diagnosis and timely antibiotic treatment.
It differentiates between B. pertussis, B. parapertussis, and B. holmesii, supporting accurate clinical management.
The launch strengthens Roche’s expanding rapid diagnostics portfolio, which includes respiratory and infectious disease assays.
UK — BD has launched its Surgiphor surgical wound irrigation system in Europe following CE approval, making it available in selected markets.
Developed by Becton, Dickinson and Company (BD), the pre-mixed, sterile solution is designed to clear surgical wounds of debris and foreign material during procedures, eliminating the need for on-site preparation.
The formulation includes an antiseptic preservative to reduce bacterial contamination and lower the risk of surgical site infections.
Its ergonomic design enables quick and efficient application. The launch aligns with BD’s broader strategy to enhance surgical safety and expand manufacturing and diagnostic capabilities globally.
USA — TECLens has treated its first patient in a clinical trial evaluating its quantitative corneal crosslinking (qCXL) technology, a non-invasive approach to vision correction.
The proprietary CXLens system uses patterned ultraviolet light to create targeted molecular bonds that strengthen and reshape the cornea without removing tissue or disrupting the epithelium.
Guided by a customized treatment engine and real-time ultrasound monitoring, the procedure can be performed in-office.
The trial targets refractive errors, particularly presbyopia, which affects around two billion people globally.
Backed by investors including Johnson & Johnson Innovation, TECLens aims to shift vision correction from surgical theatres to examination rooms.
NETHERLANDS — Royal Philips, in collaboration with InkSpace Imaging, has launched the Snuggle™ pediatric body array coil, designed to make MRI scans more comfortable and less intimidating for children while maintaining high-quality imaging.
Compatible with Philips 3.0T MRI systems, the coil wraps around young patients like a soft blanket, replacing traditional rigid coils that can feel cold and uncomfortable.
The lightweight, flexible design helps reduce anxiety in children, enabling them to remain still during scans and minimizing the need for repeat imaging.
At the same time, the high-density array captures sharp, detailed images across various pediatric anatomies, ensuring clinicians receive the diagnostic clarity they require without compromising patient comfort.
For medical technologists, the Snuggle coil simplifies patient positioning, shortens setup times, and reduces overall scan durations, allowing hospitals to serve more patients efficiently.
Dr. Ioannis Panagiotelis of Philips emphasized that the device represents a successful integration of patient-centered design with advanced MRI technology, while Dr. Peter Fischer, CEO of InkSpace Imaging, noted that the coil balances comfort and clinical performance without compromise.
The device recently received FDA 510(k) clearance, validating its safety and effectiveness. Hospitals and imaging centers across the United States can now deploy the Snuggle coil, with plans to expand availability to additional regions in the near future.
Philips and InkSpace Imaging aim to continue innovating in pediatric imaging, combining technological excellence with thoughtful design to support radiologists, technologists, and families alike.
The Snuggle coil promises a safer, faster, and more positive MRI experience for children while maintaining the highest standards of diagnostic accuracy.




Please complete the following details
Telephone: Website:
Email:
What best describes your main business activity (e.g.
What best describes your job:
Chairman, CEO, Director General Management
Sales/Marketing
QA/R&D
Others (specify)
Applicant’s Signature & Stamp:
Date:
Purchasing/Supply chain
Process Management







