Studying cells under pressure
Growth and biosynthesis decrease under pressure E. coli
S. cerevisiae
Cancer cells
Cell multiplication is typically constrained by physical boundaries leading to growth pressure increase, and there is a universal decay in proliferation rates as pressure increases. Dr Morgan Delarue is studying the common physical or biological response that leads to this decrease in cell proliferation, work which holds wider relevance to our understanding of cancer. The process of cell multiplication does not typically occur in free space, as a physical boundary usually limits the amount of space available, effectively confining cells. When cells have filled up all of the space available, further growth occurs at the expense of the surrounding cells, leading to the emergence of growthinduced pressure. “There are compressive forces on cells and their surroundings, in order that they can keep on increasing their volume and continue dividing,” explains Dr Morgan Delarue, a CNRS researcher based at the LAAS-CNRS laboratory in Toulouse. This growth in pressure occurs in both physiological and pathological conditions, in plants, cancer cells, fungi, and other organisms. “For example, think of the roots of a tree growing underground, at some point there will not be enough space for them to grow. So the roots will build up force, which can even break concrete,” outlines Dr Delarue. “With cancer cells, solid tumours grow at the expense of the surrounding tissue, and by doing so they develop this mechanical stress.”
Cells Under Pressure
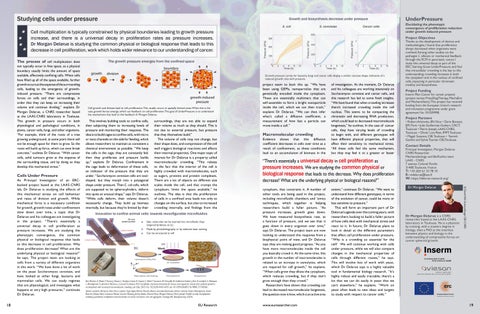
18
Growth pressure curves for bacteria, fungi and cancer cells display a similar concave shape, indicative of a reduced growth rate with pressure.
This involves building tools to confine cells, then subjecting them to increasing levels of pressure and monitoring their response. The idea is to build cages to confine cells, with micro or nano channels on the side for feeding, which allows researchers to maintain as consistent a chemical environment as possible. “We keep the cells in the cage, they are constantly fed, then they proliferate and pressure builds up,” explains Dr Delarue. Confinement in the cage leads to deformation of these cells, an indicator of the pressure that they are under. “Saccharomyces cerevisiae cells are ovalshaped, but they morph into a polyagonal shape under pressure. Then E. coli cells, which are supposed to be spherocylinders, deform into quite an unusual shape,” says Dr Delarue. “While cells deform, their volume doesn’t necessarily change. They build up biomass internally, but because they’re limited by their
surroundings, they are not able to expand their volume as much as they should. This is not due to external pressure, but pressure that they themselves build.” The volume of cells may not change, but their shape does, and compression of the cell wall triggers biological reactions and affects their internal properties. One major topic of interest for Dr Delarue is a property called macromolecular crowding. “This relates to the fact that the interior of the cell is highly crowded with macromolecules, such as sugars, proteins and protein complexes. There are a lot of objects on different size scales inside the cell, and that cramps the cytoplasm, limits the space available,” he outlines. The idea is that the proliferation of cells in a confined area leads not only to changes on the surface, but also to increased crowding internally, and findings from the
Innovation to confine animal cells: towards reconfigurable microfluidics Idea : sticks that can be inserted into microfluidic chips (inspired by Venzac et al.) Made by photolithography or by selective laser etching Can be structured at will
Ben Meriem Z, Mateo T, Faccini J, Denais C, Dusfour-Castan R, Guynet C, Merle T, Suzanne M, Di-Luoffo M, Guillermet-Guibert J, Alric B, Landiech S, Malaquin L, Mesnilgrente F, Laborde A, Mazenq L, Courson R, Delarue M. A microfluidic mechano-chemostat for tissues and organisms reveals that confined growth is accompanied with increased macromolecular crowding. Lab Chip. 2023 Oct 10;23(20):4445-4455. doi: 10.1039/d3lc00313b. PMID: 37740366. Sylvain Landiech, Marianne Elias, Pierre Lapèze, Hajar Ajiyel, Marine Plancke, Blanca González-Bermúdez, Adrian Laborde, Fabien Mesnilgrente, David Bourrier, Debora Berti, Costanza Montis, Laurent Mazenq, Jérémy Baldo, Clément Roux, Morgan Delarue, Pierre Joseph. Parallel on-chip micropipettes enabling quantitative multiplexed characterization of vesicle mechanics and cell aggregates rheology. APL Bioengineering (2024).
EU Research
project seem to back this up. “We have been using GEMs, nanoparticles that are genetically encoded inside the cytoplasm. These are essentially multimers that can self-assemble to form a bright nanoparticle inside the cell, which we can then track,” explains Dr Delarue. “We can then infer what’s called a diffusion coefficient, a measurement of how fast a particle can move inside a cell.”
Macromolecular crowding Evidence shows that this diffusion coefficient decreases in cells over time as a result of confinement, as these conditions lead to an accumulation of biomass in the
of investigation. At the moment, Dr Delarue and his colleagues are working intensively on Saccharomyces cerevisiae and cancer cells, and this research has led to some fresh insights. “We have found that when crowding increases there’s increased crowding inside the cell nucleus. This seems to be compacting the chromatin and decreasing RNA production, which could lead to decreased macromolecule biogenesis,” he outlines. In the case of cancer cells, they have varying levels of crowding to begin with, and different genotypes will have different physical properties, that will affect their sensitivity to mechanical stress. “All these cells feel the same mechanism, but they may feel it to a greater or lesser
“There’s essentially a universal decay in cell proliferation as pressure increases. We are studying the common physical or biological response that leads to this decrease. Why does proliferation decrease? What are the underlying physical or biological reasons?” cytoplasm, that constrains it. A number of other tools are being used in the project, including microfluidic chambers and ‘omics techniques, which together is helping researchers build a fuller picture. “As pressure increases, growth goes down. We have measured biosynthesis rate, as a function of pressure, and we see that it goes down in every organism over time,” says Dr Delarue. The project team are now looking to understand this response from a biophysical point of view, and Dr Delarue says they are making good progress. “As you have more macromolecules inside the cell you basically crowd it. At the same time, this growth in the number of macromolecules is related to an increase in osmolytes, which are required for cell growth,” he explains. “When cells grow they dilute the cytoplasm, which reduces crowding, but if they don’t grow enough then they crowd.” Researchers have shown that crowding can lead to decreased macromolecular biogenesis, the question now is how, which is an active area
www.euresearcher.com
extent,” continues Dr Delarue. “We want to understand how different genotypes, in terms of the evolution of cancer, could be more or less sensitive to pressure.” This will form an important part of Dr Delarue’s agenda over the coming years, with researchers looking to build a fuller picture of how cells deal with mechanical stress and react to it. In future, Dr Delarue plans to look in detail at the different parameters that affect cell proliferation under pressure. “Why is a crowding so essential for the cell? We will continue working with cells under pressure, while we will also compare changes in the mechanical properties of cells through different routes,” he says. This will involve lots of work with yeast, which Dr Delarue says is a highly valuable tool in fundamental biology research. “It’s highly robust and easily tractable; there’s a lot that we can do easily in yeast that we can’t elsewhere,” he explains. “Work on yeast often leads to new ideas and targets to study with respect to cancer cells.”
Thanks to the development of devices and methodologies, I found that proliferation always decreased when organisms were confined. Among other studies on the pathogen C. albicans or mechanical feedback through the ECM in pancreatic cancer, I study this universal decay as part of the ERC Starting Grant UnderPressure, and find that intracellular crowding is the key to this understanding: crowding increases in both the cytoplasm and in the nucleus of confined cells, impacting in particular chromatin motility and biosynthesis.
Project Funding
Inserm Plan Cancer for cancer projects (project names: PressDiagTherapy, MechaEvo and MechanoStem). This project has received funding from the European Union’s research and innovation programme under grant agreement No 101039998.
Project Partners
• Robert Arkowitz, iBV, Nice • Daria Bonazzi, IJM, Paris • Julie Guillermet-Guibert, CRCT, Toulouse • Pierre Joseph, LAAS-CNRS, Toulouse • Olivier Liot-Petit, IMFT, Toulouse • Magali Suzanne, CBI, Toulouse • Yannick Gachet and Sylvie Tounier, CBI, Toulouse
Contact Details
Principal Investigator, Morgan Delarue CNRS Researcher Mechanobiology and Biofluidics team LAAS - CNRS 7 avenue Colonel Roche 31400, Toulouse, France T: +33 (0)5 61 33 78 10 E: mdelarue@laas.fr W: https://delarue-research.org/
Dr Morgan Delarue
Dr Morgan Delarue is a CNRS researcher based at the LAAS-CNRS laboratory in Toulouse. He is a physicist by training, with a masters degree in biology, then a PhD at the interface between physics and biology in the understanding of compressive forces on cancer spheroid growth.
© David Villa ScienceImage CBI CNRS
As Principal Investigator of an ERCbacked project based at the LAAS-CNRS lab, Dr Delarue is studying the effects of this mechanical stress on cell behaviour and rates of division and growth. While mechanical force is a necessary condition for growth, growth rates under confinement slow down over time, a topic that Dr Delarue and his colleagues are investigating in the project. “There’s essentially a universal decay in cell proliferation as pressure increases. We are studying the phenotypic convergence, the common physical or biological response that leads to this decrease in cell proliferation. Why does proliferation decrease? What are the underlying physical or biological reasons?” he says. The project team are looking at cells from a variety of different organisms in this work. “We have done a lot of work on the yeast Saccharomyces cerevisiae, and have looked at other fungi, bacteria and mammalian cells. We can study regimes that are physiological, and investigate what happens at very high pressures,” continues Dr Delarue.
Project Objectives
The growth pressure emerges from the confined space
Cell growth and division lead to cell proliferation. This usually occurs in spatially limited areas. When this is the case, growth forces emerge, which can feedback on cell proliferation. The goal of UnderPressure is to understand the mechanisms that lead to this feedback. © Morgan Delarue
UnderPressure Elucidating the phenotypic convergence of proliferation reduction under growth induced pressure
19