A peek inside the primate brain There are typically several folds on the surface of the human brain, which increases its surface area and supports higher cognitive abilities. While most primate species have some degree of folding in the brain there are some exceptions, now Dr Michael Heide and his colleagues in the Primazinc project aim to shed new light on the underlying reasons why. The human brain typically has many folds on its surface, which increases its surface area and means there is more space for neurons, supporting higher cognitive abilities. Most primate species have some degree of folding in the brain, but there are some exceptions. “The marmoset for example has a fairly smooth brain, it’s less complex than other primates,” explains Dr Michael Heide, a Junior Research Group Leader at the German Primate Center in Gőttingen. As part of his role in the ERCbacked Primazinc project, Dr Heide is investigating the underlying factors behind this variability. “We use brain tissue samples – from marmosets and from rhesus macaques – to essentially find out which genes are active in the neural progenitor cells (NPCs) of the brain,” he outlines. “We basically look for genes that control the behaviour of these cells, for example the rate at which they divide, and whether they produce different kinds of cells. We aim to effectively trace their lineage.”
Rhesus macaque brain organoids in culture medium in a cell culture dish. Photo: Nesil Esiyok
Zinc finger transcription factors This research is focused on a group of proteins called zinc finger transcription factors (ZF-TFs), which are strongly expressed in the primate lineage. The ZFTFs are believed to have formed part of a defence system against certain viruses, before they later on became involved in regulating gene expression. “There are for
example some viruses that exist within our genome, that basically lead to instability of the genome. The zinc finger transcription factors originally inhibited the activity of these internal viruses,” says Dr Heide. Over time however the ZF-TFs developed new features and started to play a wider role, now Dr Heide aims to dig deeper. “We think they could play an important role in brain development by regulating different genes and generating a network,” he continues. “We are now looking to functionally test these ZF-TFs, building on earlier research, which led to a list of primate-specific genes that are known to be enriched in the progenitor cells.” The brain tissue samples are first used to identify candidate genes, then Dr Heide and his team test these genes in brain organoids, 3-D tissue structures artificially grown in the laboratory. These organoids provide an effective model of the developing brain, allowing Dr Heide to then monitor how genes influence neural progenitor cells and
Section through part of an electroporated chimpanzee brain organoid. Green: electroporated cells; magenta: neural progenitor cells; blue: cell nuclei. Photo: Nesil Esiyok
12
EU Research
Microinjected neural progenitor cells. Note the differences in morphology and position. Photo: Michael Heide
the development of the brain. “We look to see what the candidate genes do in these organoids,” he says. Researchers are using an unguided organoid protocol in this work, which differs from guided protocols. “With unguided organoids, we essentially don’t have any signalling molecules or small molecules to direct brain organoids in a particular direction. We want to draw comparisons between different primate species, so we want them to use their own, endogenous programme, instead of directing them using external factors,” explains Dr Heide. “The organoids that we are using mainly form a cortical identity.” Researchers are now using these organoids to gain deeper insights into the role of the C2 H 2 family of ZF-TFs in the development and evolution of the neocortex in different species. A key first step was to modify the existing unguided protocol, so that it could be used across different primate species, enabling easier cross-comparisons. “Our unguided protocol was initially just for humans, but now we can use it for different primate species. We have a unified protocol which we can use not only for humans, but also for chimpanzees, rhesus macaques and marmosets,” outlines Dr Heide. A method called electroporation, which involves applying an electrical pulse to a cell and then monitoring the response, is being used to study the influence of specific genes. “In the Primazinc project we electroporated chimpanzee organoids and introduced the ARHGAP11B gene, then we checked if this affected the activity of the progenitor cells and neuronal output,” says Dr Heide. This particular gene plays an important role in neocortex folding in humans, and it is thought that defects in ARHGAP11B may also affect brain development. This may lead to a smaller brain or a less folded brain, characteristics which are associated with conditions called microcephaly and lissencephaly respectively, and Dr Heide
www.euresearcher.com
“Early/Young” human brain organoid, 7 days in culture. Photo: Lidiia Tynianskaia
says the project’s research could shed new light in this area. “Our research can maybe help in identifying new risk factors, or new candidates for genetic screenings,” he says. It may be possible in future to screen
marmoset could be useful for other researchers,” continues Dr Heide. “The marmoset brain has just a single fold, it’s much less complex than the human brain, which is a major topic of interest for us.”
“We use brain tissue samples – from marmosets and from rhesus macaques – to essentially find out which genes are active in the progenitor cells of the brain. We basically look for genes that control the behaviour of these cells.” people for genetic mutations related to cortical malformations, for example, while the transcriptome datasets gathered in the project also hold wider importance. “Transcriptome datasets on the common
Brain and tissue organoids Researchers are currently working to get access to new brain and tissue organoids, which would be highly valuable in this respect, and potentially help the project team gain
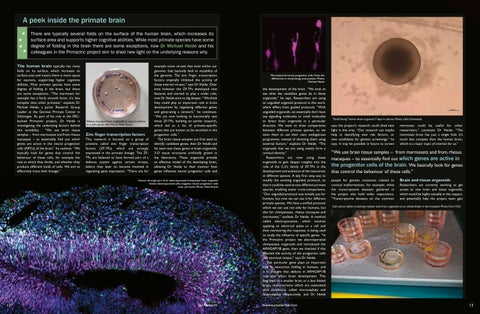
Cell culture dishes containing medium with brain organoids on an orbital shaker in the incubator. Photo: Karin Tilch
13