

Find a home for your research with The Electrochemical Society
The Electrochemical Society, a prestigious nonprofit professional organization, has led the world in electrochemistry, solid state science and technology, and allied subjects since 1902, providing a rigorous and high-quality home for the whole community. The Society is dedicated to moving science forward by empowering researchers globally to leave their mark.






MKnock-on Effects
ost of us join scientific professional societies as places to give and receive knowledge. The older I get, the more I realize that the greatest value of these organizations is the “society” part. We come for the science, but we stay for the community. Below I provide an illustration of that knock-on effect that impacted my family a great deal. Spoiler alert: much of this article focuses on Bubba, unofficial Interface mascot and all-around good dog.
The summer and fall of 2025 were challenging in the Kelly household, but they were also filled with gratitude. In mid-July our dogs Bubba and Josie went streaking down the hill to the fence because another dog had the temerity to walk in front of our house, albeit 80’ away on the road. I saw that Bubba was not putting weight on his left rear leg. A chill ran down my spine despite the July heat as that was the leg in which he had a tumor removed and endured a month of daily radiation treatments. We rushed down the hill, and I scooped him up. Mind you, he had not made a sound; he just stopped using the leg. Heather did a quick inspection and realized he had badly broken his heel. The emergency vet confirmed the diagnosis and splinted him until he could have surgery. Ironically, he already had surgery scheduled the next week as Heather had found the tiniest recurrence of his sarcoma. Our wonderful vet surgeon, whose boat I think I have paid for, put in a plate with screws and sent us off to heal. Long story long, it didn’t. The radiation he received in 2021 had damaged the heel bone, and no new bone would grow there even after three months. The radiation damage likely embrittled the bone in that area. It was a knock-on effect that we did not see coming. Still a good deal; he had been cancer-free for well over 4 years—a rarity for the sarcoma he had.
We faced a terrible decision which was really no decision: he had to have that leg amputated. While we waited for the appointed day, we reached out to our dog friends who connected us to people owned by other whippets that had gone through the same surgery. They assured us that the people have a lot harder time adapting than the dogs do. It has now been nine weeks, and I am happy to report that they are right—tripods, as they are called, get around well and don’t seem all that bothered by the loss of a leg, although their cornering ability is nearly zero. Bubba is back to himself, including walking into my office and barking at me to feed him at exactly 5 pm every day. I have not yet found his watch, but he must have it somewhere. He is even running in the side yard albeit a bit slower than before, but with the same look of absolute joy on his face. A final knock-on effect is that we have now covered all flooring with throw rugs as without them he slips constantly.
So why gratitude? Beyond having our boy back to himself, there is now no issue of recurrence of the sarcoma as the new tumor went with the leg. His heel is no longer causing him the pain that must have been brutal, despite pain medications. In addition, because I can work from home on most days, I could watch over him, give him his medications, and make sure he didn’t do anything stupid. I would go into my study each morning and he would follow, lazing in his bed just behind my chair while I tapped away at the keyboard and had many, many Zoom meetings. So I am grateful for one of the very few good knock-on effects of the pandemic: the ability for many to work from home, despite all the management hysteria.
Most of all Heather and I are grateful for the outpouring of support and love we and Bubba have received through countless texts, emails, phone calls, Zooms, and in-person talks. Although we know it was the right thing to do, the guilt about his losing his leg on our watch can be hard to suppress. Having people reassure us has been a huge help. I have often referred to ECS as my scientific family without always thinking about the connectedness that forms among members. This knock-on effect manifests itself in many ways which can fly below the radar, but not this time.
Let me end with a repeat of my plea back in 2021; please make a habit of checking your pets for lumps once a month during one of your petting sprees. They rely on us to catch these lumps as early as possible. For the boundless, unconditional love they give us, it seems the least we can do.
Until next time, be safe and happy.
Rob Kelly Editor-in-Chief
Published by: The Electrochemical Society (ECS) 65 South Main Street Pennington, NJ 08534-2839, USA Tel 609.737.1902, Fax 609.737.2743 www.electrochem.org
Editor-in-Chief: Rob Kelly
Guest Editor: Patrick Keil
Contributing Editors: Christopher L. Alexander, Christopher G. Arges, Scott Cushing, Ahmet Kusoglu, Donald Pile, Alice Suroviec
Senior Director of Publications: Adrian Plummer
Production Editor: Kara McArthur
Graphic Design & Print Production Manager: Dinia Agrawala
Staff Contributors: Frances Chaves, Francesca Di Palo, Genevieve Goldy, Maggie Hohenadel, Mary Hojlo, Christopher J. Jannuzzi, John Lewis, Anna Olsen, Fern A. Oram, Jennifer Quartararo, JaneAnn Wormann
Advisory Board: Jie Xiao (Battery Division)
Eiji Tada (Corrosion Division)
Vaddiraju Sreeram (Dielectric Science and Technology Division)
Andreas Bund (Electrodeposition Division) Vidhya Chakrapani (Electronics and Photonics Division)
Minhua Shao (Energy Technology Division)
Xingbo Liu (High-Temperature Energy, Materials, & Processes Division)
Paul Kenis (Industrial Electrochemistry and Electrochemical Engineering Division)
Chong-Geng Ma (Luminescence and Display Materials Division)
Jeffrey L. Blackburn (Nanocarbons Division)
Ariel Furst (Organic and Biological Electrochemistry Division)
Anne Co (Physical and Analytical Electrochemistry Division)
Praveen Kumar Sekhar (Sensor Division)
Publications Subcommittee Chair: Robert Savinell
Society Officers: James (Jim) Fenton, President; Francis D'Souza, Senior Vice President; Robert Savinell, 2nd Vice President; Marca Doeff, 3rd Vice President; Gessie Brisard, Secretary; Elizabeth J. Podlaha-Murphy, Treasurer; Alice Suroviec, Community Inclusion Chair; Christopher J. Jannuzzi, Executive Director & CEO
Statements and opinions given in The Electrochemical Society Interface are those of the contributors, and ECS assumes no responsibility for them. Authorization to photocopy any article for internal or personal use beyond the fair use provisions of the Copyright Act of 1976 is granted by The Electrochemical Society to libraries and other users registered with the Copyright Clearance Center (CCC). Copying for other than internal or personal use without express permission of ECS is prohibited. The CCC Code for The Electrochemical Society Interface is 1064-8208/92.
ISSN : Print: 1064-8208 Online: 1944-8783
The Electrochemical Society Interface is published quarterly by The Electrochemical Society (ECS), at 65 South Main Street, Pennington, NJ 08534-2839, USA. Subscription to members is part of membership service. © Copyright 2026 by The Electrochemical Society. *“Save as otherwise expressly stated.”
The Electrochemical Society is an educational, nonprofit 501(c)(3) organization with more than 9,000 scientists and engineers in over 75 countries worldwide who hold individual membership. Founded in 1902, the Society has a long tradition in advancing the theory and practice of electrochemical and solid state science by dissemination of information through its publications and international meetings.


https://orcid.org/0000-0002-7354-0978


Sustainability and Digitalization
by Patrick Keil
Corrosion
by Ingrid Milošev

by Thomas R. Selmi, Craig Moore, Jake Kattelman, Tolin SkovBlack, Jonathan L. Ramos, and Dev Chidambaram
by Mikhail Zheludkevich and Daniela Jacob



Building on 2025 Successes to Meet 2026 Challenges
One of the most important, and genuinely rewarding, aspects of my role as Executive Director of The Electrochemical Society is the opportunity to engage regularly in one-on-one conversations with science and technology thought leaders from around the world. These kinds of conversations took place over the past year on-site at ECS meetings in the United States, Canada, and Sweden; during my presentations at partner and peer conferences in Britain, China, and Mexico; and at industry meetings across the global science and technology ecosystem. Together, these engagements connected me with colleagues around the world and provided invaluable insight into shared priorities and emerging concerns.
Despite differences in location, discipline, and perspectives, a clear theme emerged relative to the questions asked. Nearly everyone wanted to know about recent and proposed US policy changes affecting science, research funding, and immigration—and, critically, how ECS can advocate for science amid these challenges. Many expressed deep concerns about devastating research funding cuts, restrictions on international collaboration, and the growing influence of politically motivated, non-technical appointees on research oversight and decision-making. These concerns are neither abstract nor speculative. They are real and widely shared across our membership and the broader scientific community, with effects felt almost immediately in 2025. From disrupted research programs to uncertainty surrounding international partnerships and talent mobility, these policy changes have already begun to reshape the scientific landscape. There is little doubt that these pressures will continue to affect the ECS community throughout 2026 and beyond.
Against this backdrop, I want to reflect on 2025 and outline the key challenges and opportunities ahead. By understanding where we have been and the forces shaping our environment, we can better position ECS to support our members, advocate effectively for science, and navigate the complex landscape we face in 2026.
Despite the very real challenges that defined 2025, the resilience and strength of the ECS community not only sustained our core operations but also provided inspiration and steadiness amid an increasingly turbulent external environment. Time and again, our members demonstrated that scientific collaboration endures even in uncertain times.
While there were a number of last-minute speaker and attendee cancellations in 2025, overall ECS Meeting attendance remained strong throughout the year and, most importantly, continued to reflect our community’s truly global nature. More than 65 countries were represented at the 247th and 248th ECS Meetings, reaffirming our shared commitment to open exchange, international cooperation, and scientific excellence.
I am especially grateful to ECS Secretary Gessie Brisard for opening the spring ECS Meeting in Montréal, Canada, with warmth, energy, and unmistakable local flair. Addressing the audience in her native French, Gessie delivered a rousing and
deeply affirming message that underscored our community’s strength and optimism. Her remarks established an upbeat, forward-looking spirit that carried through the meeting.
That same spirit was powerfully displayed at our fall ECS Meeting in Chicago, IL. The strong international participation was particularly meaningful, offering a vivid counterpoint to the negative rhetoric and damaging imagery directed at the city in the months prior to the event. By convening colleagues from around the world there, we showcased Chicago’s beauty, vibrancy, and vitality—and reaffirmed global scientific gatherings’ essential role in fostering understanding and collaboration. We were truly honored to host the meeting in Chicago and share it with our community and the world.
ECS Publications continued thriving in 2025. With more than 15 million downloads—an all-time high—our publications sustained their unprecedented growth trajectory following the transition to the IOPscience platform in 2020. This continued momentum reflects the quality of the science our members produce and the enduring relevance of our journals within the global research ecosystem.
At the same time, recognizing the increasingly challenging political climate facing science in the United States, the Society significantly expanded its advocacy efforts. ECS leaders met with Congress members and staff from both major political parties to underscore the critical importance of sustaining science funding and continuing support for international students. Read more about this work here
In addition, I was honored to participate in a working group of scientific society executives that authored a multisociety response to the August 7 Executive Order, “Improving Oversight of Federal Grantmaking.” Endorsed by more than 55 learned societies, this collective effort aimed to ensure that the US federal grantmaking ecosystem continues to serve as the global gold standard for supporting cutting-edge research and driving technological innovation.
We are under no illusion that any single letter, meeting, or action can, by itself, alter the broader challenges we face. Advocacy is sustained work, and it will remain a major focus of ECS efforts in 2026 and beyond. Together, we will continue to identify new ways to convene and engage our community despite funding constraints and evolving immigration policies. We will expand our presence on Capitol Hill, deepen our advocacy outreach, and provide our members with practical tools and strategies for them to engage effectively in advocacy.
Looking ahead, I remain deeply confident in the collective strength of the ECS community. Even in challenging times, our shared commitment to science, collaboration, and global engagement endures. It is that commitment—reflected in our meetings, educational outreach, awards, publications, and advocacy—that will guide us forward and ensure that ECS remains a strong, principled voice for science in the years to come.

Christopher J. Jannuzzi ECS Executive Director/Chief Executive Officer https://orcid.org/0000-0002-7293-7404


Battery Developement
Innovation at every stage

M470
Scanning Electrochemical Workstation
• Customize up to 7 modules
• Differentiate local corrosion effects
• 110 mm range on all axis
Materials & Components

MTZ-35
Impedance Analyzer
• Up to 35 MHz
• Atmospheric, pressure & temperature control

VMP-300
Premium Potentiostat
• Up to 16 channels
• Up to 150 A
• EIS up to 11 MHz
Fundamental & Proof of Concept

VMP-3e
Essential Potentiostat
• Up to 16 channels
• Up to 800 A with booster
• EIS up to 1 MHz
A solution for each step of your battery development journey
Battery development spans from fundamental research to full-scale validation & screening. Whether you’re investigating new materials and chemistries, validating prototypes, or optimizing performance at scale, we can help you optimize each stage with dedicated instruments and methodologies by providing solutions tailored to your research needs.

MPG-2xx
Premium Research Grade
Battery Cyclers
• Fast CC-CV shift
• 3-electrode cells
• Negative voltage
BT-Lab® Suite
BCS Control & Analysis Software
• BT-Test™- Cycling control & monitoring
• BT-Analysis™ - Batch data processing
• BT-Clim™ - Climate chamber control


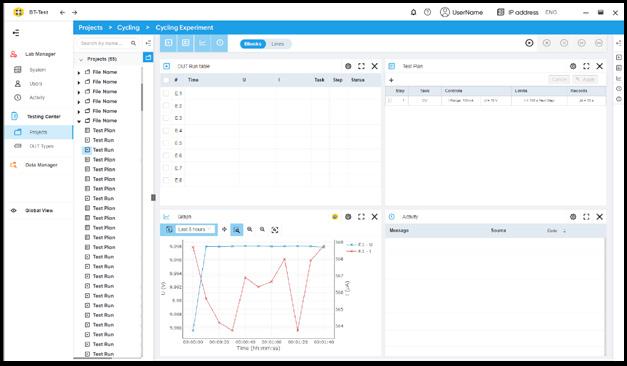
EC-Lab®
Potentiostat Control & Analysis
Software
• Battery dedicated techniques & analysis
• Climate chamber control
• EIS QI™
Scale up & Validation

BCS-900 series
Premium Battery Cyclers
• 4 cycler modules
• From nA to 300 A
• Native EIS 10 kHz to 10 mHz


Screening
BCS-1012
Essential Battery Cycler
• High density
• 6 A
• 32 channels in 8U
• Ideal for cylindrical cells



2025 Year in Review
n 2025, our members’ crucial role in addressing global challenges—establishing renewable energy sources, mitigating climate change, providing clean water, finding new and more efficient ways to store and utilize energy, inventing environmentally safe corrosion inhibitors, developing advanced sensors, and more—continued to expand in the face of significant local and global challenges. As the leading society in solid state and electrochemical science, The Electrochemical Society (ECS) is proud to fulfill our urgent mission of empowering our members at the forefront of discovery for the benefit of all humanity.
Key 2025 Society Achievements
• Accelerating scientific discovery at ECS meetings by convening record numbers of scientists worldwide and across career stages, with the 247th ECS Meeting the largest spring meeting in the Society’s 123-year history and the 248th the largest non-PRiME meeting during that time
• Disseminating groundbreaking research and discoveries broadly through ECS journals, achieving a record 21,541,651 downloads from the ECS Digital Library hosted on IOPscience
• Increasing the number of ECS Student Chapters to 163
• Expanding science advocacy efforts with a leadership visit to Capitol Hill and coauthoring a response to Executive Order “Improving Oversight of Federal Grantmaking”
• Affirming our commitment to DEI with the formation of the Community Inclusion Committee and appointment of Alice Suroviec as the Society’s first Community Inclusion Committee Chair
• Achieving strong financial results to sustain these initiatives
249th
ECS Meeting
May 24–28, 2026
Seattle, WA US
Seattle Convention Center –Arch and Sheraton Grand Seattle

250th ECS
Meeting
October 25–29, 2026
Calgary, Canada
BMO Centre
Total
52 9,493 3,688 163
New Student Chapters in China, Lithuania, Mexico, United Arab Emirates, US
Honors, Awards, Fellowships, and Grants
ECS Toyota Young Investigator Fellowship
• Total $ awarded $250,000 (5 x $50,000)
• Total # of 2025–2026 Fellowship recipients 5 (Wesley Chang, Jovan Kamcev, Jeffrey Lopez, Gioele Pagot, and Evan Wenbo Zhao)
• Grant per Fellowship recipient $50,000
• Total $ awarded since program inception $1.9 M
• Total # of awardees since program inception 38 (including 2025–2026 recipients)
ECS Toyota Young Investigator Fellowship expanded beyond the US, Canada, and Mexico to include researchers based in Europe
ECS Summer Fellowships
• Total $ awarded $20,000
• Total # of Fellowship recipients 4
• Amount of each Fellowship $5,000
• Total $ awarded for ECS Colin Garfield Fink Fellowship $5,000
• Total # of ECS Colin Garfield Fink Fellowship recipient(s) 1
ECS Travel Grants
• 247th ECS Meeting
o Total $ awarded $71,950
o Total # of recipients 134
• 248th ECS Meeting
o Total $ awarded $78,070
o Total # of recipients 134
Z01—General Student Poster Awards
• Total $ awarded $6,000
• Total # of Student Poster Award winners 7
Society, Division, and Section Awards
• Total $ awarded $82,500
• Total # of Society Award recipients 7
• Total # of Division Award recipients 27
• Total # of Section Award recipients 9
2025 Class of Fellows of The Electrochemical Society
Thierry Brousse, École polytechnique universitaire de Nantes Université
Scott Calabrese Barton, Michigan State University
Uroš Cvelbar, Jožef Stefan Institut
Avetik R. Harutyunyan, Honda Research Institute USA
Bing-Joe Hwang, National Taiwan University of Science and Technology
Ryoji Kanno, Institute of Integrated Research, Institute of Science Tokyo
Xingbo Liu, West Virginia University
Radenka Maric, University of Connecticut
Nosang Myung, University of Notre Dame
Colm O’Dwyer, University College Cork
Jeff Sakamoto, University of California, Santa Barbara
David Wilkinson, University of British Columbia
ECS Education

• Total # of webinars 10
o Total # of registrants 6,038
o Total # of speakers 12
• Total # of ECS Short Courses 6
o Total # of registrants 87
Total # of:
• Total # of Professional Development Workshops 3
o Total # of registrants 128
• Total # of Battery Courses 3
o Total # of registrants 44
Launch of third ECS Battery Course, “Manufacturing of Lithium-Ion Batteries and Pack Design.”
Meetings
247th ECS Meeting
May 18–22, 2025 | Montréal, Canada
• Total external symposium funding $56,920

• Registrants 2,454 (plus) Non-technical guests 20
• Exhibitors 38
• Technical sponsors 15
• Total Division symposium funding $30,200
• Total # of registration waivers 29

1,184


247th ECS Meeting: ECS United
• Largest spring meeting in the Society’s 123-year history
• ECS Lecture delivered by David P. Wilkinson, University of British Columbia
• Return of “It Could Be Verse,” an evening of international poetry and song
• Member reception had its own theme: “Celebrating 12 Million Digital Library Downloads”
• First “Whittingham Young Investigator and Student Slam” symposium (formerly the Battery Student Slam symposium)
248th ECS Meeting
October 12–16, 2025 | Chicago, IL US UNITED THROUGH SCIENCE & TECHNOLOGY
Total # of:
• Registrants 3,113 (plus) Non-technical guests 28
• Exhibitors 32
• Technical sponsors 28
• Total external symposium funding $96,385
• Total Division symposium funding $26,800
• Total # of registration waivers 63





ECS-Related Meeting
248th ECS Meeting: United Through Science & Technology
• Largest non-PRiME meeting in the Society’s 123-year history
• ECS Lecture delivered by Yang Shao-Horn, Massachusetts Institute of Technology
• Fifth meeting in a row (since fall 2023) to exceed 3,000 scheduled presentations
• Member reception had its own theme: “Celebrating Student Chapters”
• NEW for this meeting: Video Hub where meeting attendees could share their ECS stories
19th International Symposium on Solid Oxide Fuel Cells (SOFC-XIX)
July 13–July 18, 2025 | Stockholm, Sweden
Total # of:
• Participants 511 (plus) Non-technical guests 25
• Sessions 58
• Countries represented 40
• Abstracts 488
o Including # student abstracts 212
• Oral talks 300
o Including # student talks 125
o Invited talks 7
SOFC-XIX
• Posters 188
o Including # student posters 87
• Exhibitors 15
• Technical sponsors 2
• Total external symposium funding $5,000
• Total Division symposium funding $7,500
• Total # of registration waivers 0
• Award talk: High-Temperature Energy, Materials, & Processes Division Subhash Singhal Award winner Subhasish Mukerjee, Ceres Power Limited
• Keynote talks: Speakers from NEDO (New Energy and Industrial Technology Development Organization, Japan), Korea Institute of Energy Research (KIER), and Clean Hydrogen Partnership (Europe) gave plenary talks
• SOFC held their opening reception at the Stockholm Opera House, and a banquet at The Brewery (the main meeting venue)
Publications

Editorial Changes
Farewell to:
• Harshina Mukundan, Associate Editor, ECS Sensors Plus (ECSSP)
• Katja Frohlich, Associate Editor, ECSSP
• Olja Simoska, Associate Editor, ECSSP
• Krishnan Rajeshwar, Editor-in-Chief, Journal of Solid State Science (JSS)
• Stefano Cinti, Technical Editor, Journal of The Electrochemical Society (JES) & JSS Sensors
• Janine Mauseroll, Technical Editor, JES Organic and Bioelectrochemistry
• Aniruddh Khanna, Technical Editor, JSS Electronic Materials and Processing

• Dirk Guldi, Technical Editor, JSS Carbon Nanostructures and Devices
• Thomas Thundat, Associate Editor, ECSSP
• Nae-Lih (Nick) Wu, Associate Editor, JES
Welcome to:
• Tae-Yeon Seong, Technical Editor, JSS Electronic and Photonic Devices and Systems
• Jeffrey Bell, Associate Editor, ECSSP
• Keith Stevenson, Associate Editor, JES
• Thiagarajan Soundappan, Associate Editor, ECSSP
• Jihyun Kim, Technical Editor, JSS Carbon Nanostructures and Devices
Top ECS Reviewers
• Batteries and Energy Storage: Bingan Lu, Liqiang Mai, Simin Peng
• Carbon Nanostructures and Devices: Ghulam Dastgeer, Hanim Hussin, L. Bahmad
• Corrosion Science and Technology: Elena Romanovskaia, Gerald Frankel, Joey Kish
• Dielectric Science and Materials: Abhishek Rawat, Arun Banotra, Ritesh Verma
• Electrochemical Engineering: Ali Abbar, Arash Namaeighasemi, Geethapriyan Thangamani
• Electrochemical/Electroless Deposition: György Kaptay, Graham T. Cheek, Jianxun Song
• Electronic and Photonic Devices and Systems: Jian-Sian Li, Shreesha Rao, Stephen J. Pearton
• Electronic Materials and Processing: Jihoon Seo, Fatima Anwar, Qinzhi Xu
• Fuel Cells, Electrolyzers, and Energy Conversion: Jean St-Pierre, Nagappan Ramaswamy, Yudong Wang
• Luminescence and Display Materials, Devices, and Processing: Alok Srivastava, Milesh Ugemuge, A. N. Yerpude
• Organic and Bioelectrochemistry: Marta Jarczewska, Philippe Dauphin-Ducharme, Yuquan Feng
• Physical and Analytical Electrochemistry, Electrocatalysis, and Photoelectrochemistry: Cheolwoo Park, Mingyi Zhang, Paweł Józef Kulesza
• Sensors: Anushkannan Nedumaran Kalavathy, Pramod Kumar K. Gupta, Yi Hsing Liu
• JSS and JES now included in publishing agreements with the University of California Digital Library and the French higher education and research institutions’ Couperin Consortium.
• ECS Advances (ECSA) now officially indexed in Web of Science (WoS).
21,541,651 7,101 530 35%
Total # of ECS Digital Library articles and abstracts downloaded
Total # of journal articles published in the 2025 volume year (not including ECS Interface)

Free the Science Week downloads from JES, JSS, ECS Meeting Abstracts, ECS Interface, and ECS Transactions (ECST) jumped from 281,260 in 2024 to 518,132 in 2025—an 84% increase
Total # of open access papers published in 2025 (JES, JSS, ECSA, & ECSSP)
Percentage of articles published as open access in 2025 (JES, JSS, ECSA, & ECSSP only)




• ECS wins two 2025 APEX Awards for Publication Excellence.
Look Where ECS Was Represented in 2025!
ECS-Sponsored Meetings
• 2025 Workshop on Electrochemistry
April 28–30 | Allen J. Bard Center for Electrochemistry, University of Texas at Austin
• Electrochemical Engineering Workshop: From Fundamentals to Applications
June 2–4 | Case Western Reserve University
• 2025 Battery Safety Workshop
June 5–6 | University of North Carolina at Charlotte
• 19th International Symposium on Solid Oxide Fuel Cells (SOFC-XIX)
July 13–18 | Stockholm, Sweden
• LEC 2025 Lester Eastman Conference on High Performance Devices
August 11–13 | University of Florida
• Annual Workshop on Electrochemical Measurements
August 11–15 | Case Western Reserve University
• International Conference on Excited States of Transition Elements (ESTE2025)
August 24–29 | Wrocław, Poland
ECS-Exhibited Meeting
• International Battery Seminar & Exhibit (IBSE)
March 17–20 | Orlando, FL

249th ECS Meeting | May 24–28, 2026 |
Seattle Convention Center – Arch and Sheraton Grand Seattle
The 249th ECS Meeting takes place in Seattle, WA, from May 24 to 28, 2026, at the Seattle Convention Center. Anchored by the theme Sustainable Technologies, the meeting underscores the vital role of electrochemistry and solid state science in addressing today’s most pressing sustainability challenges. From clean energy and decarbonization to circular materials and climate-resilient systems, international researchers from academia, industry, and government come together to share breakthroughs that reduce emissions, conserve resources, and advance a more sustainable future. Through oral presentations, panel discussions, poster sessions, tutorial sessions, and exhibits, the 249th ECS Meeting spotlights cuttingedge innovations that drive climate action, resource efficiency, and long-term planetary health.






Plenary Session: ECS Lecture
Monday, May 25
Wearable Bioelectronic Platforms
Joseph Wang, University California San Diego
The ECS Lecture describes the Wang Group’s journey toward the introduction of skin-worn bioelectronic platforms, along with related energy and integration advances, and their latest efforts which are aimed at transforming these innovations into commercial products for the management of health conditions such as diabetes, Parkinson’s disease, and sepsis.
Wearable electronic systems hold tremendous promise for monitoring the wearer’s health and wellness. Inspired by continuous glucose monitoring, the landscape of wearable devices has substantially evolved over the past decade with the advent of epidermal electrochemical sensors capable of non-invasively and continuously capturing rich and dynamic molecular information.

Electrochemical sensors are particularly attractive for such on-body health monitoring owing to their high performance and inherent miniaturization. The recent development of multimodal (hybrid) wearable systems enables the simultaneous real-time monitoring of multiple key chemical and physical parameters and offers previously unattainable comprehensive temporospatial insights into a person’s health.
A major bottleneck that hampers the widespread use of advanced health monitoring systems is the need for a continuous power supply. Integrated energy-autonomous wearable microgrids can support the growing power demands of wearable health monitoring platforms. However, wearable microgrid systems require optimal energy management, tailored to changing environmental conditions and dynamic user demands.
Can’t Miss Events
(Consult the Online Program for days/times)
Opening Reception
Kick off an exciting week with fellow attendees over small bites and an open bar. Enjoy dedicated networking time with ECS leaders and

Division representatives—learn about Society initiatives, share your ideas and discuss the Society's future.


Student Mixer
Sponsored by Pine Research and Scribner
You’re out of the lab, so come have fun at the Student Mixer! After a full day of symposia, unwind your mind and meet up with friends old and new over snacks and beverages. Students and early-career researchers are welcome.

Blue & Green Day
Show off your ECS spirit by wearing blue and green—the official colors of ECS! Whether you’re presenting research, attending sessions, or networking, let’s visually celebrate the unity of our community!

Exhibit Hall
Discover the latest innovations in instruments, materials, systems, publications, and software as you explore the vibrant Exhibit Hall. Meet face-to-face with leading organizations and experts from across academia, industry, and government, all gathered to showcase what’s next in electrochemistry and solid state science.

General and Z01—Student Poster Sessions
Presenters meet and greet peers, professors, and industry representatives; field questions; make connections; and discuss compelling research questions. Browse the aisles and find the posters that draw you in!

Award Talks
Gain unique insights, inspiration, and motivation at Society, Division, and Section award winners’ talks throughout the week. Hear firsthand about their research innovations and how they achieved success. Network with the best and the brightest!

Video Hub & Professional Portrait Hub

Show off your passion, boost your professional presence, and help inspire the science and technology community—one smile and one great first impression at a time. Visit the Video Hub to step in front of the camera and record a quick video telling your ECS story. Then get your free professional headshot to glow-up your LinkedIn profile and give your job search—or next career move—a boost.
A Batteries and Energy Storage

Symposium Topics
A01 New Approaches and Advances in Electrochemical Energy Systems
Loraine Torres-Castro, Yuliya Preger, Golareh Jalilvand, Sanjeev Mukerjee, Duhan Zhang, Chockkalingam Karuppaiah, Yue Qi ECS Energy Technology; ECS Battery; ECS Industrial Electrochemistry and Electrochemical Engineering
A02 Whittingham Young Investigator and Student Slam 2
Chunmei Ban, Rana Mohtadi, Neil Dasgupta, Hui Zhou ECS Battery
A03 Li and Beyond Li Chemistries for Rechargeable Batteries: Electrodes, Electrolytes, and Interphase
Bishnu Prasad Thapaliya, Craig Bridges, Yan Yao, Charl Jafta, Kumar Raju, Bharatiraja Chokkalingam, Vito Di Noto ECS Battery; ECS Physical and Analytical Electrochemistry
A04 Materials Advancements for Beyond Li-ion Batteries from Industrial and Academic Perspectives
Rana Mohtadi, Zhirong Zhao-Karger, Kang Xu, Siwen Wang ECS Battery
A05 Commercialization of New Battery Technologies: Difficulties and Challenges
Yan Wang, Jason Croy, William Chueh, Xin Li ECS Battery
A06 Advancing Energy Storage Technology Development with DataDriven Methods
Yangang Liang, Steven Torrisi, Shijing Sun, Shoichi Matsuda, Chockkalingam Karuppaiah, Antoni Forner-Cuenca, Tzu-chen Liu ECS Battery
B Carbon Nanostructures and Devices
B01 Carbon Nanostructures for Energy Conversion and Storage
Andrew Ferguson, Jeffrey Blackburn, Uroš Cvelbar, Adam Boies, Avetik Harutyunyan, Eva Kovacevic, Albert Liu, Jennifer Lu, KyuYoung Park, Ming Lee Tang, Chunsheng Wang
ECS Nanocarbons; ECS Battery; ECS Energy Technology
B02 Carbon Nanostructures in Medicine and Biology
Delphine Bouilly, Daniel Heller, Ardemis Boghossian, Tatiana Da Ros, Markita Landry, Larry Nagahara, Jeffrey Halpern, Mekki Bayachou, Jessica Koehne, Anton Naumov, Nicole Iverson, Noe Alvarez
ECS Nanocarbons; ECS Organic and Biological Electrochemistry; ECS Sensor
B03 Carbon Nanotubes – From Fundamentals to Devices YuHuang Wang, R. Bruce Weisman, Slava Rotkin, Shigeo Maruyama, Yan Li, Benjamin Flavel, Ming Zheng, Jana Zaumseil, Sofie Cambre, Mijin Kim
ECS Nanocarbons
B04 NANO in Korea
Ji-Hyun Jang, Jae-Joon Lee, Yuanzhe Piao, Slava Rotkin, Kyu-Young Park, Changshin Jo
ECS Nanocarbons
B05 Fullerenes, Endohedral Fullerenes, and Molecular Carbon Yoko Yamakoshi, Alan Balch, Francis D’Souza, Luis Echegoyen, Dirk Guldi, Nazario Martín, Steven Stevenson, Akimitsu Narita, Vijay Krishna
ECS Nanocarbons
B06 2D Layered Materials from Fundamental Science to Applications
Hiroki Kondo, Stephen E. Creager, Michael Scott Arnold, Richard Martel, Eva Kovacevic, Uroš Cvelbar, Elisa M Miller-Link, Neelakandan Marath Santhosh, Slava V. Rotkin, Abdoulaye Djire, and Thomas Szkopek ECS Nanocarbons; ECS Dielectric Science and Technology
B07 Light Energy Conversion with Metal Halide Perovskites, Semiconductor and Nanostructures, Inorganic/Organic Hybrid Materials, and Dynamic Exciton Hiroshi Imahori, Prashant Kamat, Kei Murakoshi, Tsukasa Torimoto, Andrea Listorti
ECS Nanocarbons
B08 Porphyrins, Phthalocyanines, and Supramolecular Assemblies
Nathalie Solladie, Karl Kadish, Tomás Torres, Roberto Paolesse, Norbert Jux, Ángela Sastre-Santos
ECS Nanocarbons
B09 On-Surface Synthesis of Carbon Nanomaterials
David Ecija, Nazario Martín, Hiroshi Imahori, Wilhelm Auwärter
ECS Nanocarbons
B10 Nano for Industry
Mark Hersam, Slava Rotkin, Joseph Andrews, David Estrada, Sheng Xu, Michael Pomfret
ECS Nanocarbons
C Corrosion Science and Technology
C01 Corrosion General Session
Eiji Tada, Rebecca Schaller, Dev Chidambaram ECS Corrosion
D Dielectric Science and Materials
D01 Chemical Mechanical Polishing 18
Gul Bahar Basim, Yaw Obeng, Robert Rhoades, Gautam Banerjee, Jason Keleher, Taesung Kim
ECS Dielectric Science and Technology
D02 Dielectrics for Nanosystems 11: Materials Science, Processing, Reliability, and Manufacturing
Eva Kovacevic, Durga Misra, Toyohiro Chikyow, Yaw Obeng, Zhi
David Chen
ECS Dielectric Science and Technology
D03 Nanoscale Luminescent Materials 9
Peter Mascher, Dong-Kyun Ko, David Lockwood, Lluis Marsal
ECS Dielectric Science and Technology; ECS Electronics and Photonics; ECS Luminescenece and Display Materials
D04 Plasma Electrochemistry and Catalysis 5
Uroš Cvelbar, Davide Mariotti, Mohan Sankaran, Mahendra
Sunkara, Neelakandan Marath Santhosh
ECS Dielectric Science and Technology
E Electrochemical/Electroless Deposition
E01 Electrodeposition for Semiconductor Applications
Luca Magagnin, Philippe Vereecken, Harold Philipsen, Colm O’Dwyer
ECS Electrodeposition; ECS Dielectric Science and Technology; ECS Electronics and Photonics
E02 Electrodeposition of Multiphase Materials: Composites, Multilayers, and Graded Coatings
Elizabeth (Lisa) Podlaha-Murphy, Adriana Ispas, Luca Magagnin, Sudipta Roy, Qiang Huang
ECS Electrodeposition; ECS Physical and Analytical Electrochemistry
F Electrochemical Engineering
F01 Advances in Industrial Electrochemistry and Electrochemical Engineering: In Honor of Mark Orazem
Vincent Vivier, Douglas Riemer, Elizabeth Biddinger, Chockkalingam Karuppaiah, Paul Kenis, Venkateshkumar Prabhakaran, Samaneh Shahgaldi, Luca Magagnin, Masayuki Itagaki
ECS Industrial Electrochemistry and Electrochemical Engineering; ECS Corrosion; ECS Electrodeposition; ECS Energy Technology
F02 Highlighting Startups Pursuing Electrochemical Manufacturing 2 Juan Lopez-Ruiz, William Tarpeh, Miguel Modestino, Ashwin Ramanujam, Adam Weber
ECS Industrial Electrochemistry and Electrochemical Engineering
G Electronic Materials and Processing
G01 Silicon Compatible Emerging Materials, Processes, and Technologies for Advanced CMOS and Post-CMOS Applications 16 Hemanth Jagannathan, Zia Karim, Kuniyuki Kakushima, Paul Timans, Stefan De Gendt, Durga Misra, Yaw Obeng, Fred Roozeboom, Rishikesh Krishnan, Evgeni Gousev, James Kelly
ECS Electronics and Photonics; ECS Dielectric Science and Technology
Symposium Topics

H Electronic and Photonic Devices and Systems
H01 Wide-Bandgap Semiconductor Materials and Devices 27: In Memory of John C. Angus
Jennifer Hite, Vidhya Chakrapani, Qiliang Li, Travis Anderson, Marko Tadjer, Sunghwan Lee, Gautam Banerjee, Mahendra Sunkara, Sreeram Vaddiraju, Mohan Sankaran
ECS Electronics and Photonics; ECS Dielectric Science and Technology
H02 Solid State Electronics and Photonics in Biology and Medicine 12 Zong-Hong Lin, Yu-Lin Wang, Wenzhuo Wu, Chih-Ting Lin, Toshiya Sakata, Mark Ming-Cheng Cheng, Lluis Marsal, Bor-Ran Li, Yu-Jui Fan
ECS Electronics and Photonics
I Fuel Cells, Electrolyzers, and Energy Conversion
I01 Low-Temperature Water Electrolysis (LT-WE) for Hydrogen Production 4
Hui Xu, Peter Strasser, Svitlana Pylypenko, Fan Yang, Marcelo Carmo, Zhenhua Zeng
ECS Energy Technology; ECS Industrial Electrochemistry and Electrochemical Engineering; ECS Physical and Analytical Electrochemistry
I02 Renewable Fuels via Artificial Photosynthesis or Heterocatalysis 12 Nianqiang Wu, Frank Osterloh, Jae-Joon Lee, Heli Wang, Scott Cushing, Tsutomu Minegishi, Ali Seifitokaldani, Valentine Vullev, Tianquan (Tim) Lian, Gary Phillip Wiederrecht, and Haimei Zheng
ECS Energy Technology; ECS Physical and Analytical Electrochemistry; ECS Sensor
I03 Materials for Low-Temperature Electrochemical Systems 12 Minhua Shao, Paweł Kulesza, Iwona Rutkowska
ECS Energy Technology; ECS Battery; ECS Physical and Analytical Electrochemistry
I04 Mechano-Electro-Chemical Coupling in Energy Related Materials and Devices 6
Xinfang Jin, Nicola Perry, Paul Albertus, Jason Nicholas
ECS High-Temperature Energy, Materials, & Processes; ECS Battery
I05 Energy Conversion Based on N, P, and Other Nutrients 5
Damilola Daramola, William Tarpeh, Shiqiang Zou, Valentine Vullev, Thomas Schmidt
ECS Energy Technology; ECS Industrial Electrochemistry and Electrochemical Engineering; ECS Physical and Analytical Electrochemistry
I06 Heterogeneous Functional Materials for Energy Conversion and Storage 4
Wilson Chiu, Fanglin (Frank) Chen, Deryn Chu, Steven DeCaluwe, Vito Di Noto, Srikanth Gopalan, Andrew Herring, Nian Liu, Torsten Markus, Alice Suroviec, Jian Xie
ECS High-Temperature Energy, Materials, & Processes; ECS Battery; ECS Energy Technology; ECS Physical and Analytical Electrochemistry
I07 High-Temperature Hydrogen Production
Sean Bishop, Olga Marina, Dong Ding, Brandon Wood, S Elangovan
ECS High-Temperature Energy, Materials, & Processes; ECS Battery
I08—Flow Batteries: Conventional, Hybrid, and Beyond
James Saraidaridis, Thomas Zawodzinski, Antoni Forner-Cuenca, Joaquín Rodríguez-López
ECS Energy Technology; ECS Battery; ECS Physical and Analytical Electrochemistry
K Organic and Bioelectrochemistry
K01 Advances in Organic and Biological Electrochemistry and Division
Manuel M. Baizer Award
Ariel Furst, Jeffrey Halpern, David Hickey
ECS Organic and Biological Electrochemistry; ECS Physical and Analytical Electrochemistry
L Physical and Analytical Electrochemistry, Electrocatalysis, and Photoelectrochemistry
L01 Physical and Analytical Electrochemistry, Electrocatalysis, and Photoelectrochemistry General Session
Anne Co, Svitlana Pylypenko
ECS Physical and Analytical Electrochemistry
L02 Impedance Technologies, Diagnostics, and Sensing 6
David Cliffel, Petr Vanýsek, Jeffrey Halpern
ECS Physical and Analytical Electrochemistry; ECS Organic and Biological Electrochemistry
L03 Electroactive and Ion-Conducting Polymers
Stephen Paddison, Alice Suroviec, Shelley Minteer, Johna Leddy, A. Robert Hillman
ECS Physical and Analytical Electrochemistry; ECS Organic and Biological Electrochemistry
L04 Polynuclear Coordination Networks of Metal Clusters with Oxo, Cyano, Nitrogenous or Organic Linkers in Electrocatalysis, Electrochemical Energy Conversion, and Storage
Paweł Kulesza, Iwona Rutkowska, Nianqiang Wu, Marco Giorgetti, Andrew Herring, Vito Di Noto, Krishnan Rajeshwar
ECS Physical and Analytical Electrochemistry
M Sensors
M01 Recent Advances in Sensors Systems 7
Larry Nagahara, Dong-Joo Kim, Raluca Van Staden, Gary Hunter, Aida Ebrahimi
ECS Sensor
M02 Interface of Bioelectronics with Biology
Aida Ebrahimi, Vivek Kamat, Harshini Mukundan, Milad Navaei, Ryan Williams, Farnaz Lorestani, Daniel Heller
ECS Sensor; ECS Organic and Biological Electrochemistry; ECS Nanocarbons
Z General
Z01 General Student Poster Session
Alice Suroviec, Jennifer Hite, Venkateshkumar Prabhakaran, Taylor Garrick
All Divisions
Z02 Electrochemical Recovery, Recycling, and Sustainability of Critical and Value Added Materials 3
Chockkalingam Karuppaiah, Xiao Su, Santosh Vijapur, Shiqiang Zou, Luis Aldana, Taeyoung Kim, Damilola Daramola, Luca Magagnin, Benjamin Gould, Plamen Atanassov, Walter van Schalkwijk, Rojana Pornprasertsuk
ECS Industrial Electrochemistry and Electrochemical Engineering; ECS Battery; ECS Dielectric Science and Technology; ECS Electrodeposition; ECS Energy Technology; ECS High-Temperature Energy, Materials, & Processes; ECS Physical and Analytical Electrochemistry; Interdisciplinary Science and Technology Subcommittee
Z03 Electrochemistry for Chemical Manufacturing 3
Elizabeth Biddinger, Chockkalingam Karuppaiah, Wenzhen Li, Paul Kenis, James Fenton, Matthew Graaf, Abdoulaye Djire, Greg S. Jackson
ECS Industrial Electrochemistry and Electrochemical Engineering; ECS Energy Technology; ECS Organic and Biological Electrochemistry; ECS Physical and Analytical Electrochemistry; Interdisciplinary Science and Technology Subcommittee
Z05 Young Researchers in Western North America: A Special Symposium and Workshop April Li, Gao Liu
ECS Pacific Northwest Section; ECS San Francisco Section; ECS University of Washington Student Chapter; ECS University of California San Diego Student Chapter; ECS Canada Section
May 24–28, 2026| Seattle, WA
2 4 9 t h E C S M e e t i n g B Y T H E N U M B E R S

S e s s i o n s
S y m p o s i a 3 5 8 4 7
C o u n t r i e s R e p r e s e n t e d 6 2
I n v i t e d T a l k s
A w a r d /
K e y n o t e T a l k s 3 1 5 2 4
G r a n d T o t a l s
T o t a l
A b s t r a c t s 2 , 7 9 3

2 , 2 5 0
T o t a l O r a l
P r e s e n t a t i o n s
T o t a l P o s t e r
P r e s e n t a t i o n s 5 4 3
S t u d e n t T o t a l s

S t u d e n t A b s t r a c t s 7 6 4
S t u d e n t O r a l
P r e s e n t a t i o n s
S t u d e n t
P o s t e r
P r e s e n t a t i o n s 3 1 3 1 , 0 7 7
















Publications Update
Integrity in Action: Responsible Publishing with ECS
by Adrian T. Plummer, MPA, PMP, Senior Director of Publications
As we move further into 2026, the ECS Publications team welcomes the opportunity to pause and reconnect with our global community of authors, reviewers, editors, and readers. The early months of the year are often marked by momentum: new projects underway, manuscripts in preparation, and ideas moving swiftly toward submission. This moment offers a valuable reminder to slow the pace just enough to ensure that each step in the publication journey is taken with intention and care.
A Shared Commitment to Integrity in ECS Publishing
At ECS, scholarly publishing is understood as both a privilege and a responsibility. Every manuscript published in an ECS journal becomes part of a permanent scientific record, carrying implications that extend well beyond the point of acceptance or publication. Submission is not a provisional act; it is a deliberate and often irreversible commitment. For that reason, we encourage all authors— whether submitting for the first time or contributing as long-standing members of the ECS community—to approach submission with a clear understanding of the journal’s scope and of ECS’s ethical expectations and definition of author responsibilities.
Ethical Standards at the Core of ECS Publishing
ECS maintains robust policies to uphold the highest standards of ethical publishing. These policies address authorship and contributorship, conflicts of interest, data and image integrity, peer review conduct, and the responsible use of artificial intelligence tools. ECS and its publishing partner, IOP Publishing (IOPP), are aligned with the principles and best practices of the Committee on Publication Ethics (COPE), ensuring that ethical matters are handled consistently, transparently, and in accordance with globally recognized standards.
Ethical issues may arise at any stage of the publication lifecycle, sometimes inadvertently, and often are preventable through careful preparation and communication among coauthors. When concerns do arise, ECS is committed to safeguarding the integrity of the scholarly record while treating authors, reviewers, and editors with fairness, professionalism, and respect.
Aligning Research with ECS Topical Interest Areas
An essential early step in the submission process is confirming that a manuscript aligns with a journal’s scope. ECS journals are structured around clearly defined topical interest areas (TIAs), which reflect the scientific communities they serve and guide editorial and peer review workflows.
Authors are strongly encouraged to review the ECS TIAs before submission. Thoughtful alignment helps ensure that manuscripts are evaluated by editors and reviewers with appropriate subject-matter expertise and that the research reaches the audience most likely to benefit from it. Careful consideration of TIAs supports efficient peer review and strengthens the overall impact of the published work.
Author Guidelines: A Living Resource
Equally important is a thorough review of journal author guidelines prior to submission. These guidelines outline expectations related to manuscript preparation, ethical disclosures, authorship confirmation, data presentation, and technical requirements. They also describe policies governing revisions, author changes, and post-publication updates.
The ECS author guidelines are living documents and are updated regularly to reflect evolving best practices, policy refinements, and changes across the scholarly publishing ecosystem. Even experienced ECS authors are encouraged to review the guidelines with each new submission rather than relying on past experience alone. Doing so can prevent delays, reduce the likelihood of ethical complications, and support a smoother and more transparent peer review process.
Submission as a Deliberate Act
By the time a manuscript is submitted, all authors should have reviewed and approved the content, agreed on authorship order, and confirmed that the work meets journal and ethical requirements. Changes after submission, particularly to authorship, are often complex and, in some cases, not possible. These safeguards exist to protect authors, editors, reviewers, and the integrity of the scientific record.
Submission also represents a commitment from the broader community. Reviewers volunteer their expertise, editors apply careful judgment, and readers place trust in the rigor of the process. Recognizing the seriousness of submission honors these contributions and reinforces confidence in ECS publications.
A Collective Responsibility, Ongoing
The strength and reputation of ECS journals are sustained through shared responsibility. Authors, reviewers, editors, and staff all contribute to upholding ethical standards and to advancing electrochemical and solid state science. By thoughtfully engaging with Topical Interest Areas, reviewing current author guidelines, and approaching submission with care and deliberation, authors play a critical role in this collective effort.
As 2026 continues to unfold, we thank you for your continued engagement with ECS publications and for your commitment to responsible, high-quality scholarship. We look forward to another year of collaboration, discovery, and service to the global scientific community.

Dr. Alice Suroviec has accepted appointment as Interim Technical Editor for the Journal of The Electrochemical Society (JES) Organic and Bioelectrochemistry topical interest area for a sixmonth term of January 1, 2026–June 30, 2026 while the journal seeks nominees for the role.

Dr. Gang Wu has accepted appointment as Associate Editor for the Journal of The Electrochemical Society (JES) Fuel Cells, Electrolyzers, and Energy Conversion topical interest area for the 36-month term December 1, 2025–November 30, 2028.
Editorial Update

Dr. Rajeev Gupta has accepted appointment as Associate Editor for the Journal of The Electrochemical Society (JES) Corrosion Science and Technology topical interest area for the 12-month term January 1, 2026–December 31, 2026.

Dr. Gregory Jackson has accepted reappointment as Associate Editor for the Journal of The Electrochemical Society (JES) Fuel Cells, Electrolyzers, and Energy Conversion topical interest area for the 36-month term January 15, 2026–January 14, 2029.

Dr. Yu-Lin Wang has accepted reappointment as Technical Editor for the ECS Journal of Solid State Science and Technology (JSS) Sensors topical interest area for the 36-month term January 1, 2026–December 31, 2029.
ECS Board of Directors Report
The ECS Board of Directors held its fall gathering on Thursday, October 16, 2025, in conjunction with the 248th ECS Meeting in Chicago, IL, US. ECS President James Fenton called the Board to order and kicked off an agenda-packed meeting. In addition to the reports on the Society’s major initiatives, key motions were passed to finalize the 2026 operating budget, set the strategic direction for ECS’s operational units such as Meetings and Publications, and approve the first of ECS’s 2026 Society Award Winners.
ECS Secretary Gessie Brisard then presented the previous Board meeting minutes and had the pleasure of announcing the recently elected board members: Andreas Bund, Electrodeposition Division; Xingbo Liu, High-Temperature Energy, Materials, & Processes Division; and Chong-Geng Ma, Luminescence and Display Materials Division. Their two-year terms, which began immediately following the Board meeting in Montréal, Canada, end in October 2027. Congratulations and best of luck to our newly elected Board members!
Following the Secretary’s report, the Board heard committee updates. New Education Committee Chair Scott Calabrese Barton outlined changes to the enormously successful ECS Toyota Young Investigator Fellowship program. Individual Membership Committee Chair EJ Taylor, with help from former ECS Senior Director of Engagement Shannon Reed, called for the approval of two new ECS Student Chapters (Brown University, Rhode Island, US, and Khalifa University, Abu Dhabi, United Arab Emirates). Institutional Engagement Committee Chair Alex Peroff shared details of another sold-out exhibition floor at the Chicago meeting.
ECS Vice President and Technical Affairs Committee Chair Francis D’Souza, along with ECS Executive Director Chris Jannuzzi, then provided the Technical Affairs Committee report,
which included updates on the Society’s Meetings, Publications, and Interdisciplinary Science and Technology Subcommittees. Highlights included:
• More than 3,175 attendees at the Chicago meeting made it one of the largest meetings ever!
• We are on track to exceed fifteen million downloads from the ECS Digital Library in 2025.
• The Interdisciplinary Science and Technology Committee (ISTS) is sponsoring three symposia at the upcoming 249th ECS Meeting, including Z04—Materials, Devices, and Systems for Neuromorphic Computing and Artificial Intelligence Hardware
ECS Treasurer Lisa Podlaha-Murphy discussed the state of ECS’s finances, noting that despite the strong financial position ECS is in today, concerns over policy changes and disruptions to science funding, especially in the US, have resulted in a reduced operating budget for 2026. The goal of the 2026 budget is to ensure that ECS can continue advancing the Society’s mission and supporting our community, despite the potentially challenging days ahead.
The meeting concluded with the report from Honors and Awards Chair Adam Weber, who presented the motion to approve Robert Kelly (University of Virginia, Charlottesville) as the winner of the 2026 Henry B. Linford Award for Distinguished Teaching, and Thomas Zawodzinski (University of Tennessee) as the winner of the 2026 Vittorio de Nora Award. Congratulations to Robert and Thomas. We look forward to sharing the public recognition of these awards at the 249th ECS Meeting.
Last, members of the Board made, seconded, and unanimously approved a motion to close the meeting. The Board will reconvene in May 2026 during the 249th ECS Meeting in Seattle, WA, US.




ECS Division Contacts
H M
Battery
Jie Xiao, Chair
Pacific Northwest National Laboratory
Jagjit Nanda, Vice Chair
Xiaolin Li, Secretary
Neil Dasgupta, Treasurer
Doron Aurbach, Journals Editorial Board Representative
Cl–H+ Corrosion
Eiji Tada, Chair
Institute of Science Tokyo
Rebecca Schaller, Vice Chair
Yaiza Gonzalez-Garcia, Secretary/Treasurer
Sannakaisa Virtanen, Journals Editorial Board Representative
D T S Dielectric Science and Technology
Sreeram Vaddiraju, Chair
Texas A&M University
Eva Kovacevic, Vice Chair
Zhi David Chen, Secretary
Thorsten Lill, Treasurer
Peter Mascher, Journals Editorial Board Representative
Electrodeposition
Andreas Bund, Chair
Technische Universität Ilmenau
Rohan Akolkar, Vice Chair
Adriana Ispas, Secretary
Massimo Innocenti, Treasurer
Takayuki Homma, Journals Editorial Board Representative
Electronics and Photonics
Vidhya Chakrapani, Chair
Rensselaer Polytechnic Institute
Zia Karim, Vice Chair
Travis Anderson, 2nd Vice Chair
Jennifer Hite, Secretary
Helmut Baumgart, Treasurer
Travis Anderson, Journals Editorial Board Representative
Tae-Yeon Seong, Journals Editorial Board Representative
Energy Technology
Minhua Shao, Chair
Hong Kong University of Science and Technology
Hui Xu, Vice Chair
Iryna Zenyuk, Secretary
Ertan Agar, Treasurer
Minhua Shao, Journals Editorial Board Representative
High-Temperature Energy, Materials, & Processes
Xingbo Liu, Chair
West Virginia University
Teruhisa Horita, Vice Chair
Dong Ding, Junior Vice Chair
Xinfang Jin, Secretary/Treasurer
Minhua Shao, Journals Editorial Board Representative
Industrial Electrochemistry and Electrochemical Engineering
Paul Kenis, Chair
University of Illinois at Urbana-Champaign
Elizabeth Biddinger, Vice Chair
Chockalingam Karuppaiah, Secretary/Treasurer
Paul Kenis, Journals Editorial Board Representative
Luminescence and Display Materials
Chong-Geng Ma, Chair
Chongqing University of Posts and Telecommunications
William Cohen, Vice Chair
Luiz Jacobsohn, Secretary/Treasurer
Won Bin Im, Journals Editorial Board Representative
Nanocarbons
Jeffrey L. Blackburn, Chair
National Renewable Energy Laboratory
Ardemis Boghossian, Vice Chair
Yan Li, Secretary
Hiroshi Imahori, Treasurer
Jihyun Kim, Journals Editorial Board Representative
Organic and Biological Electrochemistry
Ariel Furst, Chair
Massachusetts Institute of Technology
Jeffrey Halpern, Vice Chair
David Hickey, Secretary/Treasurer
Alice Suroviec, Journals Editorial Board Representative
Physical and Analytical Electrochemistry
Anne Co, Chair
The Ohio State University
Svitlana Pylypenko, Vice Chair
Iwona Rutkowska, Secretary
Valentine Vullev, Treasurer
Andrew Hillier, Journals Editorial Board Representative
Sensor
Praveen Kumar Sekhar, Chair
Washington State University
Dong-Joo Kim, Vice Chair
Leyla Soleymani, Secretary
Harshini Mukundan, Treasurer
Yu-Lin Wang, Journals Editorial Board Representative
SOCIETY NEWS SOCIETY NEWS
Division News
ECS High-Temperature Energy, Materials, & Processes
H M The High-Temperature Energy, Materials, & Processes (H-TEMP) Division is pleased to introduce its new MidCareer Award, established in 2026 to recognize outstanding mid-career researchers whose work is reshaping the landscape of high-temperature electrochemistry, energy systems, materials, and processes. Generously sponsored by MTI Corporation and the Jiang Family Foundation, the award honors researchers 13–25 years into their careers who have demonstrated exceptional scientific or technological impact and show clear promise of continued leadership in the field.
Beginning with the inaugural award cycle in 2026, the nomination period will run from March 15 through June 15, 2026. Eligible candidates must be ECS members in good standing and present
Division
a compelling record of contributions, demonstrated through foundational research, influential publications (including at least one ECS publication), patents, or innovative technologies that advance high-temperature energy and materials science. The award includes a certificate; $1,000 honorarium; and support to attend the ECS spring meeting, where the recipient will deliver a keynote lecture at an H-TEMP–sponsored symposium.
Through this award, the H-TEMP Division seeks to spotlight and elevate mid-career scientists who are driving the next wave of breakthroughs in solid state electrochemistry, high-temperature processes, and energy conversion technologies. We warmly encourage the ECS community to nominate deserving colleagues and help recognize the rising leaders shaping the future of our discipline.
Division News
ECS Sensor Division
Dr. Dongmei Dong, Assistant Professor of Physics & Astronomy at Rowan University and 2024 ECS Sensor Division Early Career Award recipient, has received a National Science Foundation research grant to support the development of advanced sensor technologies for the detection of per- and polyfluoroalkyl substances (PFAS). Prof. Dong’s work aims to enable more rapid and reliable monitoring solutions for these persistent environmental contaminants which pose increasing risks to water safety and public health.
Her research will accelerate the translation of innovative sensing principles into practical systems for environmental applications. The project also supports student involvement and hands-on electrochemical research training, helping to develop the next generation of sensor scientists and engineers within the ECS community. Prof. Dong continues her strong contribution to the ECS Sensor Division’s mission through invited research talks, symposium leadership, and editorial activities across the Society.
Prof. Dong’s work significantly advances ECS’s mission by enabling emerging sensor technologies that address energy, environmental, and societal needs. A recent article in Rowan Today details the innovation and its broader implementation, reflecting strong institutional enthusiasm for impactful electrochemical sensing research.
Over the past months, the Ebrahimi Bioelectronics and Biosensor Engineering (E-Bio-E) Laboratory at Pennsylvania State University (Penn State), headed by Prof. Aida Ebrahimi, has actively contributed to the sensors community through research achievements, community engagement, and symposium leadership. Prof. Ebrahimi has received a new National Science Foundation award to advance CRISPRenabled multimodal electrochemical devices for ultrasensitive, preamplification-free nucleic acid testing. In December 2025, her group published “Portable Dynamic Laser Speckle Imaging for Rapid Antimicrobial Susceptibility Testing” in ECS Sensors Plus They also secured $6,500 in support from ECS and Penn State for the M02 symposium at the 249th ECS Meeting, for which 11 invited speakers are already confirmed. Also in 2025, Prof. Ebrahimi became an Associate Editor of IEEE Sensors, reflecting her expanding role in the professional community.
The E-Bio-E Group trainees have also achieved notable recognition. Vinay Kammarchedu received the 2025 Rustum and Della Roy Innovation in Materials Research Award and the J. Jeffery and Ann Marie Fox Graduate School Dissertation Completion Award. Graduate students Heshmat Asgharian and Hossein Chenani were awarded ECS Sensor Division Student Travel Awards for the 248th ECS Meeting in Chicago, IL.
The team is preparing for the M02 symposium at the 249th ECS Meeting, which will highlight advances in electrochemical and multimodal sensing technologies. They look forward to continued engagement with the Division and to showcasing the innovative work of their members and students.

ECS Sensor Division member Prof. Dongmei Dong, Rowan University, is the recipient of a National Science Foundation research grant to support the development of advanced sensor technologies for the detection of perand polyfluoroalkyl substances (PFAS); the grant also supports student involvement and hands-on electrochemical research training.
Photo: Michael DiGiorgio, Rowan University
Slate of Candidates for Division Officers – Spring Elections
The ECS Dielectric Science and Technology Division, ECS Industrial Electrochemistry and Electrochemical Engineering Division, and ECS Nanocarbons Division have nominated new officers and members at large for the spring 2026–spring 2028 terms. These nominations recognize members’ dedication and leadership in supporting the work and strategic direction of their divisions over the next two years. Elections take place April 1–30, 2026, with results published in the ECS Interface summer 2026 issue. All Division members are encouraged to participate in the elections.
D T S Dielectric Science and Technology
Chair
Eva Kovacevic, GREMI/Université d’Orléans
Vice Chair
Thorsten Lill, Lam Research Corporation
Secretary
Hiroki Kondo, Kyushu University
Treasurer
Neelakandan Marath Santhosh, Jožef Stefan Institute Members at Large
Gautam Banerjee, IBM Corporation Research Center
Vimal H. Chaitanya, New Mexico State University
Z. David Chen, University of Kentucky
Uroš Cvelbar, Jožef Stefan Institute
Stefan De Gendt, imec
Dennis Hess, Georgia Institute of Technology
Hemanth Jagannathan, IBM Corporation Research Center
Zia Karim, Yield Engineering Systems
Dong-Kyun Ko, New Jersey Institute of Technology
Oana Leonte, Berkeley Polymer Technologies, Inc.
Peter Mascher, McMaster University
Durga Misra, New Jersey Institute of Technology
Yaw Obeng, National Institute of Standards and Technology
Kalpathy Sundaram, University of Central Florida
Mahendra Sunkara, University of Louisville
Industrial Electrochemistry and Electrochemical Engineering
Mark E. Orazem, University of Florida
Tyler Petek, The Lubrizol Corporation
Doug Riemer, Niron Magnetics, Inc.
Shrihari Sankarasubramanian, University of Texas at San Antonio
Robert Savinell, Case Western Reserve University
John Staser, Ohio University
Xiao Su, University of Illinois at Urbana-Champaign
Venkat Subramanian, University of Texas at Austin
William Tarpeh, Stanford University
Santosh Vijapur, Faraday Technologies, Inc.
John Weidner, University of Cincinnati
Nanocarbons
Chair
Ardemis Boghossian, Independent Researcher
Vice Chair
Michael S. Arnold, University of Wisconsin–Madison
Secretary
Anton Naumov, Texas Christian University
Treasurer
Jeffrey Blackburn, National Renewable Energy Laboratory
Members at Large
Noe Alvarez, University of Cincinnati
Delphine Bouilly, Université de Montréal
Sophie Cambré, Universiteit Antwerpen
Tatiana Da Ros, Università degli Studi di Trieste
Francis D’Souza, University of North Texas
David Écija, Instituto Madrileño de Estudios Avanzados en Nanociencia
Chair
Elizabeth Biddinger, City College of New York
Vice Chair
Chockalingam Karuppaiah, Vetri Labs
Division Secretary/Treasurer
Shrihari Sankarasubramanian, University of Texas at San Antonio
William Tarpeh, Stanford University
Members at Large
Christopher Arges, Argonne National Laboratory
Saket Bhargava, Blueshift
Gerardine Botte, Texas Tech University
Fikile Brushett, Massachusetts Institute of Technology
Damilola Daramola, Northeastern University
Luis Diaz Aldana, Idaho National Laboratory
James Fenton, University of Central Florida
Taylor Garrick, General Motors Holdings, LLC
Matthew Graaf, Corteva Agriscience
John Harb, Brigham Young University
Marta Hatzell, Georgia Institute of Technology
Shrisudersan Jayaraman, Corning, Inc.
Wenzhen Li, Iowa State University
Juan Lopez-Ruiz, Pacific Northwest National Laboratory
Trung Van Nguyen, University of Kansas
Andrew Ferguson, National Renewable Energy Laboratory
Daniel Heller, Memorial Sloan Kettering Cancer Center
Mark Hersam, Northwestern University
Tetyana Ignatova, University of North Carolina at Greensboro
Hiroshi Imahori, Kyoto University
Nicole Iverson, University of Nebraska-Lincoln
Markita Landry, University of California, Berkeley
Yan Li, Peking University
Richard Martel, Université de Montréal
Nazario Martín, Universidad Complutense de Madrid
Shigeo Maruyama, University of Tokyo
Elisa Miller-Link, National Renewable Energy Laboratory
Roberto Paolesse, Università degli Studi di Roma Tor Vergata
Slava V. Rotkin, The Pennsylvania State University
Ángela Sastre-Santos, Universidad Miguel Hernández de Elche
Steve Stevenson, Purdue University
Tomás Torres, Universidad Autónoma de Madrid
YuHuang Wang, University of Maryland
R. Bruce Weisman, Rice University
Yoko Yamakoshi, Eidgenössische Technische Hochschule Zürich
Jana Zaumseil, University of Heidelberg
Ming Zheng, National Institute of Standards and Technology
Podcasts of Note
Selected
for you
by Alice H. Suroviec
MIT Learn

MIT Learn is a vast library of resources, including podcasts and video lectures, that introduce students to microelectronics, carbon nanotubes, and solid state chemistry topics. It is a great resource for anyone looking for a quick tutorial from reliable instructors.
https://learn.mit.edu
Navigating Nano

In the student-led and student-produced podcast Navigating Nano, the Materials Research Society at the Joint School of Nanoscience and Nanoengineering at the University of North Carolina Greensboro presents weekly conversations with experts in the vast field of nanotechnology.
https://podcasts.apple.com/us/podcast/ navigating-nano/id1647049733
The Superposition Guy’s Podcast

The Superposition Guy’s podcast, hosted by Yuval Boger, Chief Commercial Officer of QuEra, is a weekly podcast that invites leaders and developers of quantum computing and quantum sensing to discuss both the business and the technical aspects of their work.
https://podcast.yboger.com
SemiWiki
SemiWiki is a weekly podcast full of industry news, expert interviews, and technical discussions about a wide range of semiconductor topics. With podcasts of less than 20 minutes each you can be caught up with the latest news and insights quickly.
https://semiwiki.com/podcast/
About the Author

Alice Suroviec is a Professor of Bioanalytical Chemistry and Dean of the School of Mathematical and Natural Sciences at Berry College. She earned a BS in Chemistry from Allegheny College in 2000. She received her PhD from Virginia Tech in 2005 under the direction of Dr.
Mark R. Anderson. Her research focuses on enzymatically modified electrodes for use as biosensors. She is a Fellow of The Electrochemical Society, ECS Community Inclusion Chair, and Associate Editor of the PAE Technical Division for the Journal of The Electrochemical Society. She welcomes feedback from the ECS community.


https://orcid.org/0000-0002-9252-2468
ECS CAREER CENTER
jobs.electrochem.org

RESUME WRITING
Stand out to employers and step into your dream career with a professionally written resume, cover letter, LinkedIn profile, and interview prep with Job Seeker Premium Services.
INSIGHTS
Access to in-depth data on industry outlook, wages, qualifications, and more.
COACHING
Elevate your career with personalized guidance from an experienced and certified career coach.
JOB SEARCH
Discover new niche job opportunities tailored to your industry—you won’t find these anywhere else! OPEN 365 DAYS A YEAR FOR EMPLOYERS AND EMPLOYEES!
Create a profile today to take advantage of all the tools the ECS Career Center offers. Save time, increase exposure, and find exactly what you’re looking for in your professional career.
The summer issue of Interface, “Molecular Insight into the Electrical Double Layer (EDL),” guest edited Yue Qi, Associate Editor of JSS, and Stephen Paddison, Physical and Analytical Electrochemistry Division Past Chair, highlights how modeling and simulations have transformed our molecular-level understanding of this fundamental aspect of electrochemistry nearly 80 years after David C. Grahame’s seminal paper.
Tentatively scheduled topics include “Electrons Meet Confined Electrolytes: Redefining the Electrochemical Interface,” “A Tale of Two Double Layers: Atomistic Insights into Metallic vs. Semiconductor Electrodes,” and “The Nature of Ions in Hydrated Ionomers.”
Summer 2026 will also include a From the President column by the newly elected ECS President; results of the 2025 election of officers, along with the slate of officers for 2026; and our regular features and news from the Society, members, divisions, and sections.
UPCOMING ECS SPONSORED MEETINGS
Visit the ECS website for more information.
69th Annual SVC Technical Conference
April 25–30, 2026 | Long Beach, CA
Long Beach Convention Center
Electrochemical Engineering Workshop: From Fundamentals to Applications
June 8–10, 2026 | Cleveland, OH
Case Western Reserve University
21st International Meeting on Chemical Sensors
July 11–15, 2027 | Montréal, Canada
Concordia University

To learn more about what ECS sponsorship can do for your meeting or to request ECS sponsorship for your technical event, contact ecs@electrochem.org


249th ECS Meeting May 24–28, 2026 Seattle, WA US Seattle Convention Center Early Registration ends May 4, 2026 Register


Registration opens June 2026

Characterize Sulfide Solid-State Batteries

(up to 318 MPa at 6 mm / 114 MPa at 10 mm electrode diameter)
For aprotic chemistries with solid-state and liquid electrolytes
Gas pressure sensor and gas in- and outlet (optional)



















Solid-State Batteries!
UNITED THROUGH SCIENCE & TECHNOLOGY
HIGHLIGHTS OF THE 248th ECS MEETING
The 248th ECS Meeting convened in Chicago, IL, US, from October 12 to 16, 2025. A total of 3,141 registrants (including 28 guests) from 65 countries came together under the banner of United Through Science & Technology, making it one of ECS’s largest fall meetings!* The meeting included 51 symposia with 420 sessions. The total of 3,618 accepted abstracts included 2,526 oral talks and 1,092 posters. Of these, 1,763 abstracts were from students (1,050 oral and 713 posters). The meeting featured 493 invited talks and 58 ECS award and keynote talks.
United Through Science & Technology echoed throughout the program, highlighting how electrochemistry and solid state science intersect to drive innovation in energy, electronics, and sustainability. Scientists and engineers from across academia, industry, and government came together, accelerating progress, exchanging ideas, and advancing technologies that improve lives and protect the planet. A truly collaborative scientific community engaged with the latest scientific advancements.
*Meeting metrics as of January 2026.
ECS Member Reception Celebrating Student Chapters


Kiersten Smith, ECS Lewis University Student Chapter leader (left), shares the positive impact of her involvement with ECS after being introduced by Robert Savinell, ECS 2nd Vice President (right), at the ECS Member Reception Celebrating Student Chapters.
ECS welcomed more than 400 members from around the world to the sold-out ECS Member Reception Celebrating Student Chapters. In a room alive with energy, ideas, and warm fellowship, members enjoyed an animated celebration filled with memorable moments, news, proud achievements, and the alwaysanticipated ECS raffle.
Shannon Reed, ECS Senior Director of Engagement, introduced the importance of the Society’s Student Chapters. “The heartbeat of our future, where curiosity becomes discovery and community becomes impact, these global hubs of energy, innovation, and community are shaping the future of electrochemical and solid state science.” Robert Savinell, ECS 2nd Vice President and Case Western Reserve University Student Chapter Faculty Advisor, acknowledged the 2025 Outstanding Student Chapter Award winner Case Western Reserve University, represented by students Desiree Mae Prado and Yuanman Ma and Student Chapter of Excellence winner Lewis University, represented by Kiersten Smith. Desiree Mae, Yuanman, and Kiersten then spoke about how their chapters connect them to the larger ECS community and provide opportunities to network and collaborate with other emerging scientists in their fields. (The University of Washington, which was also named a 2025 Student Chapter of Excellence, will be acknowledged at the 249th ECS Meeting in Seattle, WA, in May 2026.)
These lucky ECS members received raffle prizes:
• Elizabeth Biddinger (Associate Professor, City College of New York), an ECS member since 2004, won a 5-Night Hotel Stay for the 250th ECS Meeting in Calgary, Canada (October 2026).
• Praduman Gupta (Senior Research Fellow, Indian Association for the Cultivation of Science), a new ECS Student Member, walked away with a complimentary ECS Lifetime Membership—a gift that keeps on giving!
• Dayi Chen (CEO, PowerOnMe LLC), a rejoined ECS member since 2022, scored complimentary registration for the 249th ECS Meeting in Seattle, WA (May 2026).
• Paul J. A. Kenis (Professor, University of Illinois at UrbanaChampaign), an ECS Member since 2003, and Abigail Paul (PhD Student, Ohio University), an ECS Student Member since 2022, each took home a $250 Amazon Gift Card!
Opening Reception

The 248th ECS Meeting Opening Reception hosted a full house with several hundred attendees enjoying food and an open bar. Members and guests reaffirmed old connections and made new ones.
ECS Award & Recognition Ceremony Highlights
ECS Executive Director and Chief Executive Officer Christopher J. Jannuzzi officially welcomed everyone to the 248th ECS Meeting in Chicago, IL, and previewed the agenda of symposia, abstracts, invited talks, and student presentations. He outlined some of the Society’s responses to difficulties faced in the year so far, including cowriting a response to an executive order on US federal grantmaking. He thanked ECS Advances supporters

Socializing and connecting at the sold-out ECS Member Reception Celebrating Student Chapters. All Photos © ECS/PaulSakuma.com 2025
Socializing at the Opening Reception.
ECS Executive Director and Chief Executive Officer Chris Jannuzzi welcomes attendees at the ECS Awards & Recognition Ceremony.
as it celebrates its third year of publication. An exciting upcoming milestone is the celebration of ECS’s 250th meeting in 2026, along with a special commemorative issue of Interface. He described the importance of unity in our community to meet the coming challenges. Chris encouraged non-members to join and lapsed members to rejoin. He thanked the Society’s Institutional Partners, exhibitors, and meeting and symposium sponsors—and the important contributions of ECS divisions and sections.

Chris introduced ECS President James (Jim) Fenton, Director of the Florida Solar Energy Center and Professor of Materials Science and Engineering at the University of Central Florida. Jim spoke on the meeting theme of United Through Science & Technology and its relevance to the scientists, engineers, students, teachers, and researchers from industry, government, and academia gathered for the meeting from every continent, bound by their shared commitment to advancing science and technology— not in isolation, but in unity. He described technologies—solid state batteries, fuel cell research, semiconductor manufacturing, wearable diagnostics—developed through collaboration among different institutions, industries, divisions, and technical interest areas. Jim encouraged attendees to carry this spirit forward at the meeting because science advances fastest when it’s united.
Jim acknowledged ECS’s receipt of 2025 APEX Awards for Publication Excellence by the Journal of The Electrochemical Society (JES) and ECS Interface, and congratulated JES Editor-inChief Dave Cliffel and ECS Interface Editor-in-Chief Robert Kelly. (Robert Savinell, ECS 2nd Vice President, accepted the Interface award on Prof. Kelly’s behalf.)
Jim presented a Leadership Circle Award to BASi, represented by Ritesh Vyas, in recognition and thanks for their 10-year silverlevel Institutional Partnership.
Jim congratulated Case Western University, the 2025 Outstanding Student Chapter, represented by student chapter members Desiree Mae Prado and Yuanman Ma, and Lewis University, the ECS Student Chapter of Excellence, represented by student chapter member Kiersten Smith.
Louis Hartmann of the Technische Universität München received the 2025 Norman Hackerman Young Author Award, which recognizes the best paper published in the Journal of The Electrochemical Society by a young author or coauthors for the volume year preceding the award.
Jim then introduced ECS Senior Vice President Francis D’Souza from the University of North Texas. Francis recognized and thanked the outgoing Division chairs:
• Maria Inman, Industrial Electrochemistry and Electrochemical Engineering Division (2022–2024)
• Dev Chidambaram, Corrosion Division (2022–2024)
• Larry Nagahara, Sensor Division (2022–2024)
• Kathy Ayers, Energy Technology Division (2023–2025)
• Shelley Minteer, Organic and Biological Electrochemistry Division (2023–2025)
Francis then introduced inductees into the 2021, 2024, and 2025 Class of Fellows of The Electrochemical Society, who received this recognition for their advanced individual technological contributions to electrochemical and solid state science and technology, as well as their active membership and involvement with the Society:
2021 Class of Fellows: Jihyun Kim, Seoul National University
2024 Class of Fellows:
• Felix N. Büchi, Paul Scherrer Institut
• Sossina M. Haile, Northwestern University
• Mark Hersam, Northwestern University
• Song Jin, University of Wisconsin–Madison

2025 Class of Fellows:
• Scott Calabrese Barton, Michigan State University
• Uroš Cvelbar, Jožef Stefan Institute
• Avetik R. Harutyunyan, Honda Research Institute USA, Inc.
• Ryoji Kanno, Institute of Science Tokyo
• Xingbo Liu, West Virginia University
• Radenka Maric, University of Connecticut
• Nosang Vincent Myung, University of Notre Dame
• Jeff Sakamoto, University of Santa California Santa Barbara Jim retook the stage, introducing Mindy Zhang, Vice President, Toyota Research Institute of North America. She presented 2023–2024 and 2024–2025 ECS Toyota Young Investigator Fellowship recipients, and stated that since the program’s founding, more than $1.9 million dollars in fellowships have been awarded to elevate talented researchers pushing the boundaries of green energy technology. With their $50,000 grants, the fellows advance groundbreaking work in electrochemical research. Mindy explained that the 2025–2026 fellows will present their work at the 250th ECS Meeting in Calgary, Canada. On behalf of Toyota, she thanked ECS for this valuable partnership and expressed excitement for continuing the program’s growth in the US, Canada, Mexico, Europe, and beyond, in the years to come.
2023–2024 ECS Toyota Fellowship recipient:
• Yaocai Bai, Oak Ridge National Laboratory, Solvent-Based Defluorination for Direct Electrode Recycling 2024–2025 ECS Toyota Fellowship recipients:
• Zheng Chen, University of California San Diego, Sustainable Binder Design for Aqueous Processing of Lithium-Ion Battery Electrodes
(continued on next page)


ECS President Jim Fenton takes the stage at the ECS Awards & Recognition Ceremony.
Chris Jannuzzi (far right) honors outgoing ECS Division Chairs (from left) Maria Inman (IE&EE), Dev Chidambaram (CORR), Larry Nagahara (SENS), Kathy Ayers (ETD), and Shelley Minteer (OBE).
(From left to right:) 2023–2024 ECS Toyota Young Investigator Fellow Yaocai Bai (Oak Ridge National Laboratory) and 2024–-2025 ECS Toyota Young Investigator Fellows Juner Zhu (Northeastern University), Zheng Chen (University of California San Diego), and Zhong Wang (The University of Alabama) display their trophies!
UNITED THROUGH SCIENCE & TECHNOLOGY
(continued from previous page)
• Zhongyang Wang, The University of Alabama, Synthesis and Assessment of HydrocarbonBased Proton Exchange Membranes for Advancing Fuel Cell Technology
• Juner Zhu, Northeastern University, Interpretable Pressure-Based Analytics for Battery Assessment Jim then announced the Society Award winners.
• Paul J. A. Kenis (University of Illinois Urbana-Champaign) won the Carl Wagner Memorial Award (est. 1980), which recognizes mid-career achievement, excellence in research areas of interest of the Society, and significant contributions in the teaching or guidance of students or colleagues in education, industry, or government. His talk was titled Electrochemical Approaches for Chemical Manufacturing.


• Arumugam Manthiram (University of Texas at Austin) won the Olin Palladium Award (est. 1950) for distinguished contributions to the field of electrochemical or corrosion science. His talk was titled The Triumph with Oxide Chemistry in Energy Storage
Plenary Session: The ECS Lecture

Following the Awards Ceremony, Jim introduced the highly anticipated ECS lecturer, Yang Shao-Horn (Massachusetts Institute of Technology) and her presentation, Unlocking Electrochemical Processes for Energy Abundance
Yang Shao-Horn is JR East Professor of Engineering and Professor of Mechanical Engineering, Materials Science and Engineering, and Chemistry, and Principal Investigator, Research Laboratory of Electronics, at the Massachusetts Institute of Technology (MIT). She is a Highly Cited Researcher in chemistry and materials science with 450+ publications in the fields of physical chemistry, electrochemistry, electrocatalysis, energy, batteries, fuel cells, and electrolyzers. Her research brings together diverse disciplines (from physics and chemistry to mechanical, materials, and chemical engineering) to tackle grand challenges in sustainable energy and the environment, bridging theory and practice to advance basic science, applied discoveries, and technology development.
Prof. Shao-Horn is a member of the National Academy of Engineering, and Fellow of The Electrochemical Society, the National Academy of Inventors, and the International Society of Electrochemistry. She has advised 150+ MIT students and postdocs who are now pursuing successful careers in academia, including 50+ with faculty positions at MIT, Cornell, and Yale; at national labs; and in the private sector.
Prof. Shao-Horn is an awarded ECS Life Member since 2013, and her ECS recognitions include:
• Physical and Analytical Electrochemistry Division David C. Grahame Award (2025)
• Fellow of The Electrochemical Society (2017)
• Battery Division Technology Award (2016)
• Charles W. Tobias Young Investigator Award (2008)
• Norman Hackerman Young Authors Award (1998)
• Battery Division Student Research Award sponsored by Mercedes-Benz Research & Development (1997)
In the ECS Lecture, Prof. Shao-Horn discussed challenges, opportunities, and recent advances in fundamental understanding of charge transfer processes, and accelerated discovery of the molecules underpinning electrochemical energy storage and the making of fuels/chemicals from water splitting, as well as the reduction of carbon dioxide and molecular nitrogen. Understanding and tuning charge transfer and dynamics at interfaces is key to enabling efficient electrochemical processes and sustainable energy transformation. She reviewed recent research advancing design principles of interfacial kinetics, ion mobility, and dynamics to enhance function and performance. She discussed how covalent and noncovalent interactions can be tuned to alter the catalytic activity of water reduction and oxidation. For example, increasing the metal-oxygen covalency of oxides and activating surface oxygen sites can weaken covalent interactions between reaction intermediates and surface sites, and can enhance the activity of water oxidation; however, increasing the activity beyond an optimal value reduces oxide stability. Unfortunately, increasing metal-oxygen covalency promotes dissociative adsorption and oxidative dehydrogenation of carbonate molecules on surface oxygen sites of positive electrode materials (e.g., nickel-rich) in Li-ion batteries, which generates protic species, degrades battery electrolytes, and reduces battery life. The concept is further supported by the design of new electrolytes with high resistance to oxidative dehydrogenation, which show stable cycling of nickel-rich positive electrodes. Beyond covalent interactions, tuning non-covalent interactions and solvation environments (such as water breaking and making ions) at the electrified interface can significantly alter the kinetic barriers and rates for concerted protonelectron transfer reactions. She gave examples of how such concepts can be used to greatly increase the catalytic activity of hydrogen evolution/oxidation and oxygen reduction as well as the activity and selectivity of carbon dioxide reduction to methanol.
Prof. Shao-Horn also discussed the use of automated experimentation with machine learning (ML) and generative AI to accelerate discovery of molecules and materials. The development of advanced battery technologies, with enhanced power, energy, and life, demands the design of new electrolytes, which need to meet many requirements, including high ion conductivity and chemical/electrochemical stability with electrode materials. The ion conductivity of state-of-the-art liquid electrolytes for Li-ion and Na-ion batteries (~10 mS/cm) has remained unchanged for decades, while that of solid state batteries has increased significantly in recent years, surpassing that of liquid/polymers.

Automated experimentation can be combined with machine learning (ML) to accelerate battery electrolyte discovery. The closedloop framework employs robotic platforms for high-throughput (HT) synthesis and characterization of liquid and polymer electrolyte formulations, generating large experimental datasets with minimal human intervention. These data are correlated with HT molecular dynamics to provide fundamental understanding of the molecular processes responsible for diffusivity and are used to train ML models that predict ionic conductivity and interfacial stability and inform subsequent experiments and computation through active learning. Electrolyte discovery can be expedited by focusing on electrolyte formulations for liquid electrolytes, guiding new strategies for novel electrolyte designs for advanced battery technologies. This research paradigm represents immense opportunities for accelerating fundamental understanding of molecular processes that dictate
Paul J. A. Kenis (University of Illinois UrbanaChampaign) wins the Carl Wagner Memorial Award.
Arumugam Manthiram (University of Texas at Austin) receives the Olin Palladium Award.
Yang Shao-Horn (Massachusetts Institute of Technology) presents the ECS Lecture.
functions and the optimization and discovery of new molecules and materials for sustainable energy.
After the ECS Lecture, Chris took the stage, thanking attendees and inviting them to join Prof. Shao-Horn at the ECS Booth, where she would be answering questions. He concluded by welcoming everyone and reminding them of all the meeting offered.
Division Awards
During the meeting, Divisions presented 17 awards (including six student awards).
• Battery Division Early Career Award: Michael Metzger, Replacing Inactive Components to Improve Li- and Na-ion Cells
• Battery Division Postdoctoral Associate Research Award Sponsored by MTI Corporation and the Jiang Family Foundation: Weilai Yu, Molecular LOGICS of Li Metal Battery: From Interface to Interphase
• Battery Division Research Award: Venkataraman Thangadurai, Advances in Lithium Metal Batteries
• Battery Division Student Research Award: Xintong Yuan, Multi-Faceted Roles of Lithium Metal in Batteries and Beyond
• Battery Division Student Research Award: Seungju Yu, Unveiling Lithium Superionic Conduction in Halide Solid Electrolytes
• Battery Division Technology Award: Gau Liu, Advancing High Energy Density Batteries Through Electrode Binder Innovation
• Battery Division Technology Award: Sang-Young Lee, Upscaling High-Energy Battery Electrodes
• Corrosion Division Early-Career Award: Rebecca Schaller, Localized Corrosion: Investigating the Controlling Environmental Variables
• Corrosion Division H. H. Uhlig Award: James Noël, Coupling Electrochemical Methods with Ion Beam or Neutron Beam Techniques in situ for Corrosion Studies
• Corrosion Division Morris Cohen Graduate Student Award: Jijo Christudasjustus, Corrosion Mechanisms and Passive Film Evolution in Nanostructured Al Alloys with Far-fromEquilibrium Compositions
• Corrosion Division Rusty Award for Mid-Career Excellence: Dev Chidambaram, Strategic Selection of Materials for Pyroprocessing of Used Nuclear Fuel: Understanding Corrosion Behavior in Molten Salt Systems
• Electrodeposition Division Early Career Award: Adam Maraschky, Seeking Stability in Electrodeposition: Understanding Dendrites, Degradation, and Delamination
• Electrodeposition Division Research Award: Philippe Vereecken, Morphology Control During Electrodeposition: An Interplay of Surface Chemistry and Interphase Formation
• Energy Technology Division Walter van Schalkwijk Award in Sustainable Energy Technology: Wenzhen Li, Integrated Electrocatalytic Strategies for CO₂ Capture, Conversion, and C–N Coupling
• Industrial Electrochemistry and Electrochemical Engineering Division Student Achievement Award: Monsuru Olatunji Dauda, Strategies for Sustainable Ethylene, Ethanol, and Acetate via Electrochemical CO₂ Reduction
• Industrial Electrochemistry and Electrochemical Engineering Division Ralph E. White Outstanding Student Award, Kaustubh Girish Naik, Mechanistic Interrogation of SolidState Electrode Architectures
• Sensor Division Student Research Award: Wonhyeong Kim, Development of Molecularly Imprinted Polymer (MIP)Based Sensors for the Sensitive and Selective Detection of Analytes
248th Z01—General Student Poster Session
Congratulations to the four students who won the 248th ECS Meeting Z01—General Student Poster Session Awards, out of a field of more than 90 posters.

(From left to right:) Andrew
and Amanda
University), Yongbeom Kwon (Columbia University), and
(Indian Institute of Technology Madras) are the 248th Z01— General Student Poster Session Award Winners.
1st Prize – $1,500
Yongbeom Kwon, Columbia University
Z01—3080 Operando NMR Observation of Li Metal Plating Morphology, SEI Formation, and Electrode Wetting in Li Metal Batteries
2nd Prize – $1,000
Nilesh Nilesh, Indian Institute of Technology Madras Z01—3168 A Fresh Approach for Edge Isolation in Silicon Based Carrier Selective Contact Solar Cells through As-ALD 3rd Prize – $500
Amanda K. Warfield and Andrew C. Murphy, Lewis University
Z01—3154 Optimizing Slurry Formulation Using Organometallic Complexes to Improve Surface Oxidation of Silicon Carbide Substrates
General Student Poster Session Judges
The Society thanks the ECS members who served as reviewers for the 248th ECS Meeting Z01—General Student Poster Session. In-person judges
• Christopher Arges, Argonne National Laboratory
• Damilola Daramola, Northeastern University
• Ahmed Farghaly, Argonne National Laboratory
• Maureen Tang, Drexel University
Virtual judges
• Christoph Baumer, Technische Universität Ilmenau

• Ardemis Boghossian, Independent Researcher, 1015 Lausanne, Switzerland
• Dongmei Dong, Rowan University
• Jeffrey Halpern, University of New Hampshire
• Peter Mascher, McMaster University
• Milad Navaei, Georgia Institute of Technology
• Mark Overberg, Sandia National Laboratories
• Eijii Tada, Institute of Science Tokyo
• Loraine Torres-Castro, Sandia National Laboratories
• Roseanne Warren, University of Utah
• Hui Yang, University of South Carolina
• Chen You, COMSOL, Inc.
ECS thanks Dr. Alice Suroviec, Berry College, for serving as the Z01—General Student Poster Awards symposium organizer.
(continued on next page)
Murphy
Warfield (Lewis
Nilesh Nilesh
UNITED THROUGH SCIENCE & TECHNOLOGY
(continued from previous page)
Symposia Best Presentation and Poster Awards
Some 248th ECS Meeting symposia presented much-appreciated awards for best posters, presentations, and papers. The symposia organizers thank the sponsors who generously supported these awards.
A05—Battery
Characterization and Diagnosis
Student Oral Presenter: $500
Marie-Chloé Michaud Paradis (A05—0631), Université de Montréal
Student Poster Presenter: $500
Mahla Bakhshi (A05—0534), Universitetet i Agder
A08—Interplay between Temperature and Battery Phenomenon 2
Best Presentation Award: $1,000
Catherine Folkson (A08—0830), Imperial College London
Best Presentation Award: $1,000
Kelsey Cavallaro (A08—0834), Georgia Institute of Technology
Best Presentation Award: $1,000
Jarom Sederholm (A08—0832), University of Illinois UrbanaChampaign
L07—New Horizons in Spectroelectrochemistry and Photoelectrochemistry
Student Oral Presenter: $200
Lahiru Pasikku Hannadige (L07—2749), Texas Tech University
Student Oral Presenter: $150
Yamini Kumaran (L07—2750), University at Albany, State University of New York
Student Oral Presenter: $150
Ranil Clement Temgoua Tonleu (L07—2720), Bundesanstalt für Materialforschung und -prüfung
Student Mixer

Mixing it up at the Student Mixer!
Nearly 200 students and early career professionals packed the sold-out Student Mixer, mingling, comparing notes, and enjoying refreshments. Everyone received Student Mixer t-shirts. Special thanks to Pine Research Instrumentation and Scribner for supporting our student members by sponsoring the event.
Notable Special Events
Blue & Green Day
Meeting attendees were encouraged to show off their ECS pride on Monday by wearing blue and green, ECS’s official colors. Lots of people got in on the action. Blue and green hair, clothes, and accessories were spotted during this visual celebration of the community’s unity.

Meet the Editors
Esteemed ECS journal editors answered questions about publishing, peer review, journal scope, their vision for the growth of ECS Publications, and more at the ECS Booth. Present were ECS Journals editors Kara McArthur (Managing Editor, ECS Interface), Peter Mascher (DS&T Technical Editor, JSS), Thiagarajan Soundappan (Associate Editor, ECSSP), Netz Arroyo (Editor-in-Chief, ECSSP), Rohan Alkolkar (Editor-in-Chief, ECSA), Jeffrey Bell (Associate Editor, ECSSP), and Pietro Papa Lopes (Associate Editor, ECSA).

Striking a pose with ECS journals, attendees claim their bona fides!


(From left to right:)ECS Journals editors Kara McArthur (Managing Editor, ECS Interface), Peter Mascher (DS&T Technical Editor, JSS), Thiagarajan Soundappan (Associate Editor, ECSSP), Netz Arroyo (Editor-in-Chief, ECSSP), Rohan Alkolkar (Editor-in-Chief, ECSA), Jeffrey Bell (Associate Editor, ECSSP), and Pietro Papa Lopes (Associate Editor, ECSA), meet with meeting attendees, answering their questions and updating them on journal happenings
Meet the Leaders
ECS Executive Committee members President James Fenton, Third Vice President Marca Doeff, Senior Vice President Francis D’Souza, 2nd Vice President Robert Savinell, Secretary Gessie Brisard, Community Inclusion Chair Alice Suroviec, and Member Christopher Jannuzzi met with meeting attendees at the ECS Booth. Conversations ranged from the Society’s priorities to new initiatives.
ECS United through Science & Technology fans show off their blue and green.

Meeting participants meet the leaders—members of the ECS Executive Committee.
ECS Video Hub
In a new initiative, ECS invited everyone to stop by the ECS Video Hub and record a brief video message. Nearly 100 people participated and shared experiences that ranged from how the Society enhanced their research, to connections made at meetings that propelled their work and careers, and to what made meeting participation an outstanding experience.

Sponsors and Exhibitors
ECS applauds the meeting sponsors and exhibitors whose support and participation contributed directly to the meeting’s success. Thank you for developing the tools and equipment driving scientific advancement, sharing your innovations with the electrochemical and solid state communities, and providing generous support for the 248th ECS Meeting!


248th ECS Meeting – General Meeting Sponsors
Thank you to the 248th ECS Meeting sponsors!
Thank you to our Meeting Sponsors!











248th ECS Meeting – Exhibitors
Thank you to the 248th ECS Meeting Exhibitors!
Thank you to our Exhibitors!































248th ECS Meeting – Symposia Sponsors
Thank you to our Symposia Sponsors!
Thank you to the 248th ECS Meeting symposia sponsors!








Meeting
ECS Benefactor Level Institutional Partner BioLogic brings its A Team to the Exhibition Hall (from left to right:) Florian Chapon, Matthew Matlock, Nick Hall, David Carey, and Bill Eggers
No scripts, no stress! Nearly 100 meeting attendees dropped by the ECS Video Hub to record short videos about their ECS experiences.
In Memoriam ... Stephen John Pearton
(1957–2025)

A Pillar of Semiconductor Science and a Beloved Colleague
The Electrochemical Society and the broader scientific community mourn the passing of Professor Stephen John Pearton, who died on December 7, 2025, at the age of 68. Steve, as he was known to friends and colleagues worldwide, was a towering figure in the fields of materials science and semiconductor engineering. His passing leaves a profound void, but his monumental legacy—forged over four decades of pioneering research, prolific writing, dedicated mentorship, and spirited collaboration— will continue to illuminate the path for future generations of scientists and engineers.
Born in Hobart, Tasmania, on January 15, 1957, Steve’s intellectual journey began in Australia. He earned his PhD in Physics from the University of Tasmania in 1981, supported by the highly selective Australian Institute of Nuclear Science and Engineering Postgraduate Studentship. This early recognition foreshadowed a career defined by excellence. After a brief stint at the Australian Atomic Energy Commission and a postdoctoral fellowship at the University of California, Berkeley, Steve’s career took a defining turn in 1984 when he joined the prestigious AT&T Bell Laboratories in Murray Hill, New Jersey. It was here, at the epicenter of materials innovation, that he began the work that would reshape compound semiconductor technology. It was also where he met his future wife and lifelong scientific partner, Cammy Abernathy
In 1993, Steve and Cammy moved to the University of Florida, where they both served as full professors in the Materials Science and Engineering Department. Steve was named a University of Florida Distinguished Professor in 2001 and continued his vigorous research program long after assuming emeritus status in 2020. His career was a master class in the impactful application of fundamental science to real-world technological problems. His research portfolio—spanning plasma etching, ion implantation, Ohmic and Schottky contacts, defect studies, and wide-bandgap semiconductors—was not merely academic; it formed the foundational processing toolkit for the devices that power our modern world.
Steve’s contributions were both deep and broad. He was instrumental in developing robust methods for ion implantation isolation in III-V semiconductors like GaAs, AlGaAs, and InP. This work, critical for achieving electrical isolation between devices on a chip, enabled larger-scale integration and improved manufacturability for the field-effect transistors (FETs), high-electron-mobility transistors (HEMTs), and heterojunction bipolar transistors (HBTs) that would become ubiquitous in cellular phones and wireless communication systems.
Perhaps even more transformative was his pioneering role in bringing high-density plasma etching to compound semiconductor fabrication. Steve and his collaborators systematically developed the plasma chemistries and processes necessary to etch intricate device structures with low damage and high precision. This work moved the field from wet chemical etching to the dry, anisotropic processes essential for modern device dimensions. His techniques became the industry standard, particularly for challenging materials like
gallium nitride (GaN), where they remain the only practical method for fabricating the mesas in light-emitting diodes (LEDs) and laser diodes. His more than 2000 publications in this area educated a generation of process engineers.
A third pillar of his legacy was his seminal work on the role of hydrogen and defects in semiconductors. He was among the first to comprehensively demonstrate how hydrogen could passivate the electrical activity of dopants and defects in silicon and compound semiconductors. His authoritative review and book on the subject are among the most cited works in the field, clarifying a phenomenon of great importance to both device performance and fundamental materials science.
Steve’s approach was inherently interdisciplinary—blending physics, chemistry, and engineering to solve complex problems. This was evident in his work at Bell Labs on novel laser geometries and single-electron transistors, which presaged the nano-electronics revolution, and later in his leadership in GaN research, where he developed new contact metallizations and surface passivation schemes. His ability to collaborate across disciplines is reflected in an astonishing publication record of more than 2,600 journal articles, 12 books, and 37 book chapters, which have garnered more than 128,000 citations. He was a sought-after speaker, delivering more than 250 invited talks that inspired audiences at major conferences all over the globe.
His service to the scientific community was exemplary. Steve was a dedicated member of The Electrochemical Society, publishing 139 articles in the Journal of The Electrochemical Society and ECS Journal of Solid State Science and Technology, organizing 11 symposia, and serving on the Electronics and Photonics Division executive committee. His contributions were recognized with one of ECS's highest honors, the Gordon E. Moore Medal for Outstanding Achievement in Solid State Science and Technology. This medal joined a glittering array of accolades, including the IEEE’s J. J. Ebers Award, the American Physical Society’s David Adler Lectureship Award in Materials Physics, and Fellow of six major professional societies: APS, AVS, IEEE, MRS, SPIE, TMS, and ECS. These honors were not merely personal achievements but affirmations of the centrality of his work to the advancement of solid state science.
Beyond the laboratory and podium, Steve was a man of immense vitality, warmth, and humor. Colleagues remember not just his sharp intellect, but his wicked sense of humor, his directness, and his unwavering support. He was a passionate mentor to countless graduate students and postdoctoral researchers, guiding them with a focus on rigorous science and clear communication. His competitive spirit, honed in his youth as a rugby and cricket player in Australia and as a Taekwondo black belt, found new outlets in his fervent support of Florida Gators athletics—especially volleyball and softball—and in his devoted encouragement of his son Max’s baseball pursuits. He was a voracious reader and a loving family man, finding great joy in his 32-year marriage to Cammy and in raising their son.
Steve Pearton’s life was a testament to the power of curiosity, dedication, and collaboration. He translated deep physical understanding into engineering practices that manufacture the technologies that connect our society. From the LEDs that light our homes and streets to the power electronics in electric vehicles and the RF chips in every smartphone, his fingerprints are embedded in the fabric of contemporary life.
He is survived by his wife, Professor Cammy Abernathy, and his son, Max. The Electrochemical Society extends its deepest condolences to his family, friends, and all who were fortunate enough to work with him. Stephen J. Pearton was a giant in his field, a cherished colleague, and a true gentleman of science. His legacy is not only in his vast body of work but in the thriving field of semiconductor processing he

helped to create and the countless scientists he inspired. He will be profoundly missed and forever remembered.
This notice was prepared by Dr. Fan Ren, Distinguished Professor of Chemical Engineering, University of Florida, and Fellow of The Electrochemical Society.
ECS EDUCATION
WEBINAR SERIES
SHOWCASING DISTINGUISHED SPEAKERS AND ECS COMMUNITY MEMBERS
Co-sponsored by ECS and partners at PhysicsWorld.com
Upcoming and past webinars are at
ELECTROCHEM.ORG/WEBINARS


Correlation Between Reversible Anionic Redox and OpenCircuit-Voltage Hysteresis in Co-Free Li-Rich Layered Oxides
Cathode active materials (CAMs) have the highest cost share among the components in a Li-ion battery pack, making their cost reduction crucial for more competitive battery electric vehicles. Li-rich nickel manganese oxides with no cobalt (LR-NMs) are a promising next-generation CAM, but they display a problematic open circuit voltage (OCV) hysteresis, causing low energy efficiency. A team led by the Technical University of Munich reported a study of the OCV hysteresis in materials in this class with formula Li1+δ[NixMny]1-δO2, in which the Ni/ Mn ratio x/y and the degree of overlithiation δ were systematically varied to study the effect on OCV hysteresis. The electrochemical responses in these materials have segments ascribed to transition metal redox and, at higher voltage, anionic redox. They found increasing Li and/or Mn content increased both the contribution of the reversible anionic redox and the OCV hysteresis. This demonstrated that the OCV hysteresis is predominantly governed by the reversible anionic redox. Ultimately, the researchers found a tradeoff in the materials between specific discharge energy and round-trip energy efficiency at OCV, recommending LRNMs with Ni/Mn ratios near 1/1 as promising materials that balance both metrics.
From: T. Kipfer, R. B. Berk, F. Riewald, et al., J Electrochem Soc, 172, 100505 (2025).
Effect of Liquid Film Thickness and Flow Velocity on the Cathodic Reaction of Carbon Steel Under Liquid Film Flow Conditions
The damaged Fukushima Daiichi Nuclear Power Station (1F) primary containment vessel (PCV), made of carbon steel, has undergone unique corrosion conditions. The emergency-injected seawater has been replaced with nitrogen-saturated freshwater and water circulation has been restored. A drone inspection of the PCV inner wall revealed a liquid film flowing down it. Upcoming debris removal work is anticipated to increase dissolved oxygen to an unknown concentration. Therefore, researchers at the Japan Atomic Energy Agency studied the combined effects of both water film thickness and flow velocity on the oxygen reduction rate on carbon steel and on silver electrodes (which do not form solid corrosion products). Specially developed liquid film flow equipment allowed a planar three-electrode cell to be in contact with the flowing solution (0.1 M NaCl), which thickness was controlled by adjusting a weir and flow speed by additionally managing the flow plane angle and reservoir height. The authors obtained polarization curves of pairs of specimens (WE and CE) to determine the limiting current density as a function of water film thickness and flow velocities. Experimental
TECH HIGHLIGHTS TECH HIGHLIGHTS
values were lower than theoretically calculated values, demonstrating the value of the liquid film flow equipment in cases of corrosion products.
From: K. Otani and T. Igarashi, J Electrochem Soc, 172, 091503 (2025).
Understanding the Cathodic Overpotential in LiMn2O4 Electrodes for Lithium Recovery from Brines in a Continuous Flow-by Electrochemical Reactor
Lithium’s status as a key strategic resource has stimulated the development of new direct lithium extraction (DLE) methods. Among these, electrochemical lithiumion pumping (ELIP) is notable for its low energy consumption without additional chemical inputs. Researchers in Germany have addressed the challenge of maintaining efficiency during the high brine depletion required for economic viability. Their study provides a detailed analysis of the cathodic overpotential in lithium manganese oxide electrodes within a continuous flow-by reactor. Key findings reveal that the largest contributions to potential loss during lithium depletion come from the shift in equilibrium potential, followed by concentration overpotential. The research demonstrates that electrodes with high mass loading of 30 mg cm⁻² suffer a severe capacity reduction of 43% in lithium-depleted brine due to an additional, unmodeled overpotential from the electrode’s porous structure. In contrast, low mass loading electrodes of 10 mg cm⁻² are significantly more resilient, with only an 11% capacity loss. These results experimentally validate the proposed design of a “zoned reactor,” of progressively lower mass loading electrodes to match the decreasing lithium concentration, thereby optimizing material utilization, reducing energy consumption, and decreasing the required electrode area by up to 30%.
From: C. Roggerone, F. La Mantia, J. Kowal, et al., J Electrochem Soc, 172, 093502 (2025).
CV Shape as a Tool for Predicting Memory Effect in WO3 Sputtered Electrochromic Films
We all recognize the classical “duck” shape of a cyclic voltammogram, but not all ducks are alike. In a recent study, researchers from the University of Bordeaux, France, mapped CV profiles of WO3 electrochromic films into different “bird” shapes in their investigation on the films’ memory effect—the ability to retain the electrically induced optical state after removing the applied potential. Along with their previous work (Adv Mater Interfaces, 10, 2300549 (2023)), the authors have explored multiple ways to fabricate WO3 films and studied their memory effect with CV, optical, and other surface analysis techniques. They classified CV shapes into three categories to qualitatively correlate with memory behavior: a thin, small “crane”
indicated irreversible behavior; a fat, large “mandarin duck” represented a highly reversible system with no memory effect; and a “barnacle goose” fell in between, showing partial memory retention. Beyond qualitative mapping, the authors fit CV data to extract key parameters and combine them with optical measurements to assign a global score for the memory effect. This approach offers a quick and simple way to predict memory behavior using routine CV measurements—without resorting to complex or time-consuming techniques.
From: P. Bras, M. Maglione, M. Ammari, et al., J Electrochem Soc, 172, 106507 (2025).
Electrochemical Performances of MetalDoped Activated Carbon Compounds
Derived from Shrimp Shell Waste
The conversion of biowaste into carbon materials enables the valorization of abundant, low-value residues into high-performance functional materials, reducing reliance on fossil-derived carbons and mitigating waste management and environmental burdens. In this study, researchers developed a method to prepare activated carbon (AC) from waste shrimp shells, which are naturally high in chitosan. Additionally, they present a facile method to prepare composites of activated carbons with metal nanoparticles (Ag, Li, Na). They tested AC/metal composites as working electrodes for supercapacitors and evaluated their electrochemical performance with cyclic voltammetry. At low scan rates (10 - 20 mV s-1), AC-Na exhibited the highest specific capacitance, indicating improved charge storage compared with pristine AC and the Li- and Ag-containing composites. At higher scan rates (≥ 60 mV s-1), AC-Ag and AC-Na demonstrated higher capacitance than pristine AC and AC-Li, indicating improved rate capability and faster charge-discharge kinetics in the Ag and Na composites. This study demonstrates that supercapacitor electrode materials can be prepared from biowaste and their electrochemical performance can be enhanced through the incorporation of various metals.
From: A. B. N, F. K, S. Kumar, et al., J. Solid State Sci Technol, 14, 091001 (2025).
Tech Highlights was prepared by Joshua Gallaway of Northeastern University, David McNulty of University of Limerick, Zenghe Liu of Abbott Diabetes Care, Chock Karuppaiah of Vetri Labs and Ohmium International, and Donald Pile of EnPower, Inc. Each article highlighted here is available free online. Go to the online version of Tech Highlights in each issue of Interface and click on the article summary to take you to the full-text version of the article.
Corrosion Science at the Nexus of Sustainability and Digitalization
by Patrick Keil
In modern materials science, corrosion has always been fundamentally linked to electrochemical processes, directly affecting the development and application of novel materials across different industries. Today, its impact extends far beyond being a steady economic and environmental factor shaping the way we build, maintain, and preserve the material world. Corrosion has evolved into a strategic topic for safety and sustainability, shaped by increasing regulatory pressure, advances in both digital and material innovation, and the drive for decarbonization. This edition of Interface showcases how corrosion science has evolved, considering sustainability awareness, regulatory alignment, novel material design, and digital technologies as key drivers of scientific development rather than external limitations.
The environmental impact of corrosion is often underestimated and typically becomes apparent only when a failure occurs. However, when infrastructure or products degrade prematurely, replacement materials must be produced. This replacement typically demands substantial amounts of raw materials and energy, resulting in significant carbon emissions. Every additional year of service life achieved through effective corrosion protection reduces the environmental and economic burden associated with producing replacements. This benefit is particularly significant in industrial sectors with high embodied energy; metals manufacturing, heavy industry, and large-scale infrastructure. By mitigating degradation, we preserve the value embedded in these materials, increase material integrity, and reduce the need for carbon-intensive production cycles.
As global demand for metals continues to surge, mitigating corrosion is vital for preserving critical resources. Prof. Ingrid Milošev’s article highlights how efficient corrosion management—utilizing design, protective coatings, monitoring, and lifecycle approaches—contributes to circular-economy objectives. At the same time, sustainability introduces new challenges for corrosion protection. Regulations increasingly restrict hazardous substances, improving the quality of our lives. Industries must now innovate within narrower chemical spaces while ensuring equal or even superior performance. We need a deeper integration of corrosion science within sustainability frameworks, with interdisciplinary collaboration being essential for addressing resource and environmental challenges.
The rapid progress of modern infrastructure is truly impressive. Technologies like offshore wind farms, hydrogen pipelines, electric vehicle components, and advanced energy generation and storage systems all face their own unique corrosion challenges. Understanding these processes and environments—often multiphase, variable, and harsh—requires integrating electrochemistry, materials science, surface engineering, and mechanistic understanding. This integration is showcased by the article from Prof. Dev Chidambaram and co-workers, guiding us though the corrosion challenges in high-temperature molten systems for energy applications. Corrosion is driven by multiple factors and complex mechanisms. These corrosion mechanisms threaten structural materials, anodes, and current collectors in applications ranging from nuclear fuel to liquid-metal batteries. Managing impurity chemistry, maintaining redox control, and making smart material choices are crucial for ensuring that advanced energy technologies can operate reliably over the long term. These broad, interdisciplinary insights are vital not only for making these systems durable but also for supporting the future sustainability of renewable energy solutions.
From a sustainability perspective, digitalization supports and enhances this transformation. Modern corrosion science is increasingly transforming into a data-driven discipline built on machine learning, AI, digital twins, high-throughput experimentation, and sensor-integrated monitoring. These tools move us from reactive mitigation to predictive lifecycle engineering.
Profs. Mikhail Zheludkevich and Daniela Jacob share their vision of a comprehensive framework for a climate-aware digital twin that
integrates high-resolution regional climate modeling with hybrid corrosion prediction approaches to improve predictive maintenance of metallic infrastructure. Their research highlights the influence of environmental changes on corrosion processes, as well as the reciprocal role corrosion plays in driving environmental transformation. As climate-driven changes accelerate corrosion rates, conventional empirical and static models become increasingly inadequate for forecasting future conditions. This digital twin approach incorporates physics-based simulations, machine learning, uncertainty calculations, and real-time sensing, enabling sitespecific risk assessments, dynamic updates, and scenario-based decisionmaking—bridging accelerated tests with real-world performance. With the support of robust data pipelines and validated models, this facilitates earlier identification of degradation pathways, more rapid optimization of protective systems, and the extension of service life with improved accuracy.
This edition of Interface brings together contributions that emphasize the importance of collaborative thinking, calling on corrosion scientists, electrochemists, environmental analysts, chemists, engineers, and data scientists to work together. Corrosion protection is not a peripheral topic— it is a foundational element for achieving durable, efficient, and sustainable technologies. The advances presented here demonstrate that a sustainable future is inseparable from our ability to understand and control corrosion processes.
Organizations such as The Electrochemical Society and the European Federation of Corrosion, among many others, play a crucial role in connecting diverse scientific fields, fostering interdisciplinary collaboration, and facilitating the dissemination of scientific research across disciplines. Scientific advancement is a collective achievement. It relies on researchers, but it is built on the coordinated efforts of countless individuals who ensure that scientific exchange can occur reliably, efficiently, and to the highest professional standards. And to everyone whose contributions take place quietly, reliably, and sometimes unnoticed: please know that your work is not invisible. Your dedication and support help us to push boundaries, generate impactful knowledge, and contribute meaningfully to society. I do believe that I am speaking for all of us in expressing our wholehearted gratitude to all staff members and supporters for their unwavering service to our scientific community!
Enjoy reading this edition, please stay safe and happy. © The Electrochemical Society. DOI:10.1149/2.F06261IF
About the Author

Patrick Keil, Senior Research FellowCorrosion Protection, DirectorCorrosion Protection Research & Electrochemistry, BASF Coatings GmbH Education: PhD in Physics (University of Wuppertal)
Research Interests: Patrick’s research interests include coatings, metal pre-treatment, and applied electrochemistry. He is dedicated to the development of innovative protection technologies by means of fundamental understanding of the impact of corrosion on material performance. This includes utilizing state-of-the-art analytical methods and AI/ML techniques, promoting innovation through collaboration with both academia and industry throughout the value chain.
Honors & Awards: Patrick is serving the European Federation of Corrosion (EFC) as Vice-President https://orcid.org/0000-0001-7899-0882


(continued
ECS TOYOTA YOUNG
INVESTIGATOR FELLOWSHIP
Submissions accepted from Europe, Canada, Mexico, and US

Application deadline: April 30, 2026
Corrosion Science and Metal Protection in Support of Sustainable Development
by Ingrid Milošev
The common perception of corrosion is an image of a heavily rusted piece of metal lying abandoned somewhere, maybe an old boat corroding in a bay, forgotten by everyone, or a fence waiting to be painted. See Figure 1. Usually, corroded objects are not a pleasant sight. Some exceptions are copper and bronze monuments, weathering steel objects that rust elegantly to be admired, and, for example, the patinated zinc roofs of Paris, which give the city its exceptional panorama. However, corrosion is much more than this common perception. It affects not only our everyday lives but also our planet and, yes, sustainable development. The term sustainable development has been used extensively in recent years, and there is an excellent reason for that. Growing concerns about the impact of modern lifestyles have driven many global activities in recent decades aimed at reducing the overuse of natural resources and harmful and unwanted emissions into the environment.
To put corrosion in the framework of sustainable development, let us first define it. The commonly used definition is that sustainable development is “development that meets the needs of the present without compromising the ability of future generations to meet their own needs.” This definition was proposed in 1987 by the United Nations Commission on Environment and Development in its report, “Our Common Future.”1 The report is known as the Brundtland Report, as the Commission was chaired by the then-Prime Minister of Norway, Ms Gro Harlem Brundtland. The Commission examined the causes of environmental degradation in relation to poverty, the global economy, population growth, ecosystems, energy, industry, and other factors. Today, almost 40 years later, the strategies outlined in the Brundtland report remain of interest. The Paris Agreement was adopted by 195 parties at the UN Climate Change Conference (COP21) in Paris in 2015 and signed as a legally binding international treaty on climate change.2 The Paris Agreement has a long-term temperature goal: to keep the rise in global surface temperature well below 2 °C (3.6 °F) above pre-industrial levels. In 2015, at the United Nations Summit in New York City, 17 Sustainable Development Goals were proposed to achieve a better, more sustainable future for all, by transforming our world through the 2030 Agenda for Sustainable Development.3 The Agenda addresses the global challenges we face, including poverty, inequality, climate change, and environmental

degradation. Among these 17 goals, at least two are closely related to metals and their corrosion protection. Goal 9, “Industry, innovation and infrastructure,” is focused on building resilient infrastructure, promoting inclusive and sustainable industrialization, and fostering innovation. To achieve this goal, investments in infrastructure, advanced technologies, and lower carbon emissions are required. The second goal of interest in this context is Goal 12: “Responsible consumption and production,” which aims to ensure sustainable development and production patterns. Achieving this goal requires setting a target for reducing waste generation, along with promotion of circular economy practices and support for sustainable procurement policies. These goals are essential to the corrosion community because they are interconnected with the consumption of metals used to build infrastructure, technological development, and the transition to green energies. In this context, strategies that ensure a long metal lifecycle and metal recycling play critical roles.
The corrosion community should put more effort into highlighting the importance of corrosion and corrosion protection, and into integrating it into the broader framework of societal development to help achieve the goals we set for preservation of metal resources, increased material performance and lifetime, and reduced environmental impact. As corrosion scientists, we are primarily focused on understanding how and why a given metal corrodes, under which environmental conditions, and how we can mitigate corrosion. Indeed, these processes are complex and require substantial research effort, as they depend on multiple factors involving the metal—its chemical composition, microstructure, and mechanical properties— as well as the environment, which can be atmospheric, soil, marine, or industrial, at various temperatures and pressures. Corrosion protection is another demanding subject. It starts with careful design and selection of the most appropriate metal for the intended application, based on physical-chemical characteristics, adequate performance, and affordability. The next step is to select suitable corrosion protection measures. These are either on the metal side, by choosing among various protective coatings, surface modifications, or, for example, imposed cathodic protection, or on the environment side, by changing its composition, temperature, pressure, and gas content. Once the system is set up, it requires monitoring, inspection, and maintenance to detect corrosion early, address potential issues, and respond quickly.
Corrosion affects processes beyond the apparent level of corroding metal. It is part of a broader process that begins with mining metal ore and proceeds through metal extraction and the production of metal objects. Once produced, these objects have a particular lifetime performing their function and are eventually either disposed of or, hopefully, recycled. Each of these levels is complex and depends on numerous factors that determine production efficiency, ensure reliable performance, and drive potential environmental impact through the release of harmful substances. Another critical point is the safety of metal devices, objects, and infrastructure, and, consequently, human lives. Device safety is vital to engineering systems, energy and fuel infrastructure, transportation, and security. In the past, we have already seen several catastrophic accidents due to unrecognized corrosion issues, inappropriate material selection, or irregular maintenance.
Finally, corrosion incurs enormous costs; the direct and indirect costs due to corrosion are estimated at 3−6% of global gross domestic product (GDP).4 In 2025, the global GDP totaled 117 trillion USD,5 translating to projected corrosion costs of 3.5−7 trillion USD. (continued on next page)
Fig. 1. The common perception of corrosion is an image of a heavily rusted piece of metal lying abandoned. Photo: Ingrid Milošev
(continued from previous page)
Considering these numbers, even a small percentage preserved and redirected to other purposes, such as education, societal growth, and environmental preservation, would be meaningful.
In our recent perspective article “Challenges for the Corrosion Science, Engineering, and Technology Community as a Consequence of Growing Demand and Consumption of Materials: A Sustainability Issue” written together with Prof. John R. Scully of the University of Virginia, we sought to raise awareness within the corrosion community that the growing demand for engineering metallic materials in our increasingly technological society is unsustainable and jeopardizes natural resources.6 Given continual modernization and the ever-increasing world population, consumption of a wide number metals is increasing despite the need to transition to a lowcarbon society. The diversity of metals can be observed across many industries and consumer sectors, as well as in our everyday lives.
In 2022, annual metal production was around 2.8 billion tonnes, most of which was industrial metals, primarily iron ore (around 2.6 billion tonnes), followed by other industrial metals such as aluminium (69 million tonnes), chromium (41 million), manganese (20 million), copper (22 million), and zinc (13 million tonnes).7 Data for 2024 further increased.8
These estimates imply that demand for the major metals (iron and steel, copper, aluminium, zinc, and nickel) will likely increase by 2−6 fold over this century. Depending on the models used (top-down or bottom-up), the largest median growth rate between 2010 and 2050 is observed for Al (215%), Cu (140%), Ni (140%), Fe (86%), and Zn (81%).9 These projected demands depend on many factors (e.g., methodological choices, socioeconomic variables, GDP, population
growth, etc.). Basic models can be divided into those driven by socioeconomic parameters and those driven by stock dynamics. Consequently, estimates vary depending on the global scenario chosen (Market First, Policy First, Security First, or Equity First).
Higher rates of demand for metal imply substantial new energy allocations for mining, leading to a 21% to 37% increase in global energy demand.10 These results suggest that extensively modified or new technological approaches, along with new governmental policies, are required to meet the rising demand for metal supply. The production of all materials, including metals, is closely related to CO2 emissions. Compared to the late 19th century and the start of the second industrial revolution, driven by the discovery of electricity, gas, and oil, CO2 emissions began to grow progressively during the third (digital) revolution in the last 50 years of the 20th century and the ongoing fourth (cyber) industrial revolution. Today, we produce over 35 billion tonnes of carbon dioxide, compared to 5 billion tonnes in 1950.11 See Figure 2. Over the past 50 years, the world population has more than doubled, the global economy has grown more than tenfold, the extraction of natural resources has tripled, and global CO2 emissions have more than doubled.12 The largest share of global carbon emissions comes from iron and steel (around 4 of 37 gigatons), followed by concrete, aluminium, plastic, paper, and other metals.
The impact of corrosion on overall CO2 emissions is undoubtedly an area where more data are needed. Recently, Mario Iannuzzi and Gerald S. Frankel estimated that the CO2 emissions associated with steelmaking required to replace corroded steel will be 4.1–9.1% of total emissions by 2030, considering the European Union and recent US greenhouse gas reduction targets.13 C. Atkins and P. Lambert presented a review of embodied energy expressed as carbon dioxide equivalent (CO2e) in current corrosion-prevention practices.14 Once a
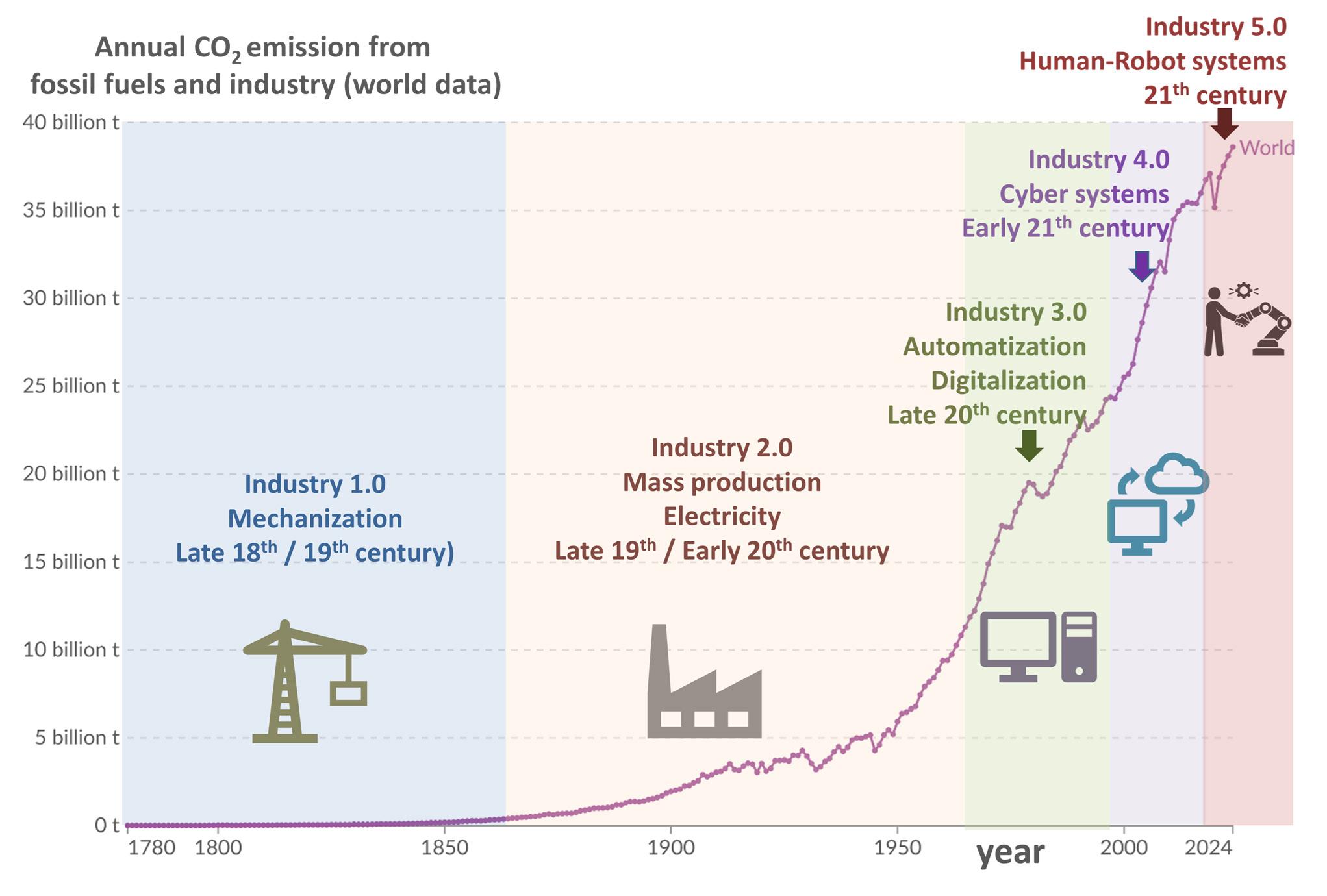
structure has been built, corrosion may occur, reducing its capacity. This is regarded as a waste of embodied carbon dioxide (so-called embodied carbon) directly related to the mass of material lost due to corrosion. Embodied carbon for coated steel was calculated based on corrosivity category (C). According to the ISO 12944-2 standard,15 there are five C categories, C1 to C5, where C1 represents very low corrosivity, and C5 represents extreme corrosivity. For example, for atmospherically exposed steel, in a C1 environment, coatings do not reduce CO2e over the life of the structure. However, in more corrosive categories, galvanizing and recoating result in progressively lower CO2e waste lifetimes. In a C5 environment, 85 µm of galvanizing, with recoating every 15 years, produces the lowest lifetime waste of CO2e. This means that the protection significantly benefits the environment in higher corrosive categories.
Further, the increased use of metals threatens natural resources and undermines their criticality, as we either deplete them or, for various geostrategic reasons, are unable to exploit them. The US Department of Energy has estimated the risk posed by individual metals.16 The analysis identifies seven metals, namely dysprosium, neodymium, gallium, graphite, cobalt, terbium, and iridium, as critical in the short term (2020–2025). These materials are used in contemporary applications that include magnets, batteries, and electronics. Others are classified as near-critical in the short term, such as lithium, uranium, electrical steel, nickel, and magnesium. Over the medium term (2025–2035), supply risk scores for specific materials are expected to shift. Specifically, nickel, platinum, and magnesium become critical, primarily due to their roles in batteries and vehicle lightweighting. Aluminium, copper, and silicon are becoming nearcritical due to increased demand for solar-energy technologies, global electrification, and vehicle lightweighting.
Today, preservation of metals is a balance among metal resources, mining, consumption, and recycling. With these facts in mind, strategies for promoting sustainable metal cycles must become increasingly effective. Society today is well aware that the classical linear-economy approach, “mine-make-use-dispose,” is not sustainable and is moving toward a circular economy approach, including the “3Rs” (reduce, reuse, recycle).17 The concept of a “circular economy” would close loops in industrial ecosystems by applying the reduce-reuse-recycle (3Rs) principle, preventing waste generation, and turning waste into resources. The circular economy originates from the “spaceship theory” introduced by the ecological economist K. E. Boulding (1966), who perceived the Earth as a circular system with no exchanges of matter with the outside environment.18 This circular development model ultimately seeks to decouple global economic development from finite resource consumption.
The introduction of 3Rs management into the Waste Framework Directive 2008/98/in the European Union. (WFD) is seen as a milestone of modern waste management in the EU.19 It is based on the 1975 Council Directive on Waste (75/442/EEC),20 in which methods for waste management were divided into (i) reduction in quantities of waste, and (ii) disposal via recycling and re-use, via recovery, and via storage and underground. In 1979, a Dutch politician, Ad Lansink, proposed a concept, the “Ladder of Lansink,” for waste hierarchy, prioritizing waste management and resource conservation options, with “reduce” at the top and “landfill” at the bottom.21
The European Union introduced the concept of a circular economy in 2014 and envisioned it as one of its most important goals to be achieved by 2035.22 Today, in the European Union, recovery rates are very high: around 90%. However, the recycled content is lower. The important fact is that the reduction in energy needed for primary production amounts to between 70 and 95% and these savings in energy are reflected in the reduced CO2 emissions. However, even in ideal scenarios with high recycling rates of 70 or 90%, the expected increase in demand would still fall short of meeting society’s high demand. The recycling rate (the percentage of remelted scrap used for product manufacture as a fraction of total scrap) for the major industrial metals is relatively high, over 50%. However, recycled content (the percentage or amount of scrap used for product manufacture relative to the total amount of material—primary material and scrap) is lower, between 30% and 40%, except for aluminium cans (80%) and lead (66% due to battery recycling). But for some metals it is as low as 1%.
We are currently in a phase of the reuse economy, a combination of the two models. Recently, ten levels of circularity were introduced by Prof. Jacqueline Cramer23 of Utrecht University: Refuse: prevent raw materials use, Reduce: decrease raw materials use, Renew: redesign product in view of circularity, Re-use: use product again (secondhand), Repair: maintain and repair product, Refurbish: revive product, Remanufacture: make new products from secondhand materials, Repurpose: re-use product but with another function, Recycle: salvage material streams with highest possible value, and Recover: incinerate waste with energy recovery. A concept of improved materials conservation through a 4D strategy: dematerialisation, durability, design for multiple lifecycles, and diversion of wastestreams through industrial symbiosis was presented as materials stewardship, as described in the article “Towards Materials Sustainability through Materials Stewardship” by Christopher Taylor et al.24
According to Prof. Dierk Raabe from the Max-Planck Institute for Sustainable Materials, even in the best-case scenario by the year 2060 only two-thirds of the greenhouse gas-intensive mass market for metals like iron, aluminium, and nickel will be circular and at least one-third will remain linear (that is based on mining, refining, and primary reduction) by the decades to come.25 Because of high growth in demand, at least one-third of the metal mass market will also, in the future, have to be supplied by primary synthesis, which creates a massive greenhouse effect through CO2 emissions. Today, globally, the situation is the opposite—only about one-third of the total mass market is based on the circular economy (through melting scrap), while two-thirds is linear (from newly mined feedstock).
In our perspective article,6 we proposed that science and engineering contribute to each step of the circular lifetime, from mining (by efficient element separation, better fabrication yields, and lower CO2 consumption), designing (materials informatics, advanced materials design modeling, machine learning, and inverse strategy methods), making (manufacturing efficiency, dematerialization, element substitution, leightweighting, fuel savings), using (better corrosion management, more intensive use, use of smart materials, lifetime prolongation, and improved post-treatments), reuse and repairing (better solvent extraction, solvometallurgy, increased recovery), and recycling (increased recycle share, electrochemical separation, selective leaching).
The connection between corrosion and sustainable development needs to be better recognized and more intensively promoted, as urged by Prof. Gerald S. Frankel of The Ohio State University, Fontana Corrosion Center, in an editorial in CORROSION Journal published in 2024.26 In 2022, Roman Bender et al. published the position paper “Corrosion Challenges Towards a Sustainable Society,” which presents a series of cases demonstrating the importance of protecting metals and alloys from corrosion for the development of energy production and a deeper understanding of corrosion science.27 He emphasized that a global transition toward more sustainable, affordable, and reliable energy systems poses a challenge for the corrosion industry, as the construction of climate‐resilient energy systems and infrastructure makes structural materials (mainly metals and alloys) a major factor.
Recently, Tomáš Prošek, Patrick Keil, and Kateryna Popova explored the intrinsic relationship between corrosion protection and sustainability, examining the economic costs, environmental impacts, and technological strategies involved in the article “Corrosion Protection and Sustainability: Why Are the Two Concepts Interwined?”28 They concluded that effective corrosion management can extend the service life of metallic components, thereby preserving resources and minimizing environmental impact. Approaches such as Total Cost of Ownership (TCO) and Life Cycle Analysis (LCA) can provide a framework for selecting the most cost-effective and environmentally friendly corrosion protection method based on the required lifetime. Best-practice guidelines and scientific societies should help to bridge the gaps between countries at different levels of development. Institutional organizations should promote extending the lifespan of structures and implementing safe, environmentally friendly practices, including lifecycle considerations, environmental compliance, low volatile organic compound content, and other
(continued on next page)
(continued from previous page)
measures. Further, scientific and expert organizations play an essential role in sharing knowledge and information on corrosion mitigation across different industries.
To conclude, new strategies from a metal lifecycle perspective are required to narrow the gap between the growth of high demand and the assurance of resources. If future consumption rates continue, the mining and production of metals must be balanced with novel engineering and technological methods that incorporate strategies for metal sustainability. So, corrosion protection, although necessary for prolonging the lifetime of a metal part, is not the only measure we should take to preserve metals. We are facing severe challenges related to global environmental pollution and potential resource constraints, not only within each research field but especially across and between disciplines, that hamper our ability to address the critical issues we need to solve. Above all, we must become aware of these challenges and bring the community together to do the best we can by using fewer resources, reusing materials, and converting waste into products. As engineers and scientists, we have a greater responsibility to contribute to a more sustainable society by inspecting and reconsidering each step of our work process, always seeking more environmentally friendly solutions.
© The Electrochemical Society. DOI:10.1149/2.F07261IF
About the Author

Ingrid Milošev, Professor and Head of the Department of Physical and Organic Chemistry at the Jožef Stefan Institute
Education: BSc in Chemical Technology (University of Zagreb), PhD in Chemistry (University of Ljubljana), Postdoctoral and research positions at the University of Duesseldorf, Germany, University of Sherbrooke, Canada and PSL University, CNRS, Chimie ParisTech, France
Research Interests: Mechanism of corrosion of metals and alloys; Electrochemical processes on metallic materials in various electrolytes and material characterization; Corrosion protection and functionalization: corrosion inhibitors, conversion coatings, sol-gel coatings, superhydrophobic coatings; Metallic materials for biomedical applications; Additive technologies for metal production Pubs + Patents: >260 peer-reviewed publications, >200 scientific conference publications, 9 book chapters, >15,000 citations (Google Scholar), h-index: 62
Awards: 2025 European Corrosion Metal, 2024 H. H. Uhlig Award of the Corrosion Division of The Electrochemical Society, 2016 Pregl Award of the National Institute of Chemistry for Exceptional Achievements, and 2011 Zois Certificate of Recognition for Significant Achievements in Slovenia
References
1. https://en.wikipedia.org/wiki/Brundtland_Commission
2. https://unfccc.int/process-and-meetings/the-paris-agreement
3. https://www.un.org/sustainabledevelopment/sustainabledevelopment-goals/
4. G. Koch, J. Varney, N. Thompson, O. Moghissi, M. Gould, and J. Payer. International Measures of Prevention, Application, and Economics of Corrosion Technologies Study. NACE International Impact (2016).
5. https://www.statista.com/gross-domestic-product-gdp/
6. I. Milošev, and J. R. Scully, CORROSION Journal, 79 (9), 988 (2023).
7. https://www.visualcapitalist.com/
8. https://www.world-mining-data.info/wmd/downloads/PDF/ WMD%202024.pdf
9. T. Watari, K. Nansai, and K. Nakajima, Resour Conserv Recycl, 164, 105107 (2021).
10. A. Elshkaki, T. E. Graedel, L. Ciacci, and B. K. Reck, Environ Sci Technol, 52 (5), 2491 (2018).
11. https://ourworldindata.org/co2-emissions
12. UN Environment Programme, “Making peace with Nature” (2021).
13. M. Iannuzzi and G. S. Frankel, npj Mater Degrad, 6, 101 (2022).
14. C. Atkins and P. Lambert, Proc Inst Civ Eng Eng Sustainability 175 (1), 22 (2022).
15. ISO 12944-2:2017: Paints and varnishes—Corrosion protection of steel structures by protective paint systems.
16. US Department of Energy, Critical Materials Assessment (2023).
17. C. Zhang, M. Hu, F. Di Maio, B. Sprecher, X. Yang, et al., Sci Total Environ, 803, 149892 (2022).
18. K. E. Boulding. The Economy of the Coming Spaceship Earth. In H. Jarrett (Ed.), Environmental Quality in a Growing Economy (pp. 3–14). Baltimore (1966).
19. EU Waste Framework Directive.
20. EU Council Directive.
21. https://www.eurowaste.be/en/knowledge-base/lansinks-laddera-sustainable-guide-to-effective-waste-management/
22. Recycling Europe. EURIC Metal Recycling Factsheet.
23. https://www.uu.nl/staff/JMCramer/Publications
24. C. D. Taylor, B. Gully, A. N. Sánchez, E. Rode, and A. S. Agarwal, Sustainability, 8 (10), 1001 (2016).
25. D. Raabe, Chem Rev, 123, 2436 (2023).
26. G. S. Frankel, CORROSION Journal, 80 (12), 1145 (2024).
27. R. Bender, D. Féron, D. Mills, S. Ritter, R. Bäßler, et al., Mater Corros, 73, 1730 (2022).
28. T. Prošek, P. Keil, and K. Popova, Corr Mater Degrad, 6, 38 (2025).

Work with ECS: Vice President (2024–2025) and current President, ECS Europe Section; Associate editor, JES Website: https://www.ijs.si/ijsw/K3-en/Milosev https://orcid.org/0000-0002-7633-9954

Challenges and Corrosion in HighTemperature Molten Systems for Energy Applications
by Thomas R. Selmi, Craig Moore, Jake Kattelman, Tolin Skov-Black, Jonathan L. Ramos, and Dev Chidambaram
Increasing energy demand and a desire for energy security have spurred interest in next-generation high-temperature energy technologies. These technologies include new nuclear reactors and reprocessing of used fuel, concentrated solar power, high-temperature energy storage, and grid-scale batteries. All of these, except grid-scale batteries and reprocessing of used nuclear fuel, require the use of a heat engine operating with the Rankine (steam) or Brayton (supercritical CO2) cycles to convert thermal energy to electrical energy. The efficiency of a heat engine is described by the Carnot Theorem:
where ηmax is the maximum achievable efficiency, TC is the temperature of the cold leg of the cycle, and TH is the temperature at the hot leg. Since the cold temperature is largely a fixed value at ambient temperatures, the only way to increase the Carnot efficiency is to increase the temperature of the hot leg of the cycle. Next-generation energy technologies use high-temperature heat transfer fluids in the hot leg. Due to their high temperature stability, low vapor pressures, and high heat capacities, both halide salts and molten metals are attractive heat transfer materials. A major barrier to the implementation of these fluids is their corrosivity to structural materials in their application. This article discusses the corrosion mechanisms in molten salts and metals in their use in energy applications.
Corrosion in Molten Halide Salts
Molten fluoride and chloride salts are candidate materials for next-generation energy technologies. Molten halide salts can be used as heat-transfer fluids or thermal-energy storage materials in concentrated solar power.1 In Gen IV nuclear reactors, halide salts can be used as a coolant or a fuel-bearing medium.2 Molten chlorides are also used as the electrolyte for the pyroprocessing of used nuclear fuel.3 A primary barrier to the wider implementation of molten halide salts is their high corrosivity to materials, which is primarily driven by the presence of impurities in the salt.4 The presence of these impurities in the salt is essential for the electrochemical corrosion of alloys used for containing the salt, as without them, there is no energetically favorable cathodic reaction.5,6 In a pure salt, oxidation of metals may occur through the reduction of the cationic constituents of the melt: Li(I), Na(I), K(I), Be(II), Mg(II), Ca(II). The reduction potentials of these cations are far below the oxidation potential of any alloying metals—meaning that the expected corrosion in a pure salt is expected to be negligible compared to an impure salt.7 This scenario is now accepted by an aggregation of molten salt corrosion data which show that alloys exposed to purified molten salts exhibit significantly less mass loss compared to those exposed to salts without purification.4
Many chloride salts are highly hygroscopic and will take up moisture from the atmosphere in both the solid and the fused states.8–12 Moisture impurities present in the salt can hydrolyze to form corrosive impurities such as OH-, O2-, and HCl/HF when molten or during heating.7,13 HCl and HF have both been shown to have measurable solubilities in their respective molten halide salts.14,15
HF and HCl are also both highly corrosive in the salt.7 In molten fluorides, HF has been shown to associate with F- to form HF2- The reduction of HF through the hydrogen evolution reaction is limited by the rate at which the formed HF2- decomposes back into HF and F- at the surface of a metal.7,16
HF eH F 1 2 2
HF FHF2
HCl instead fully dissociates into H+ and Cl- where H+ is then reduced to form hydrogen, similar to the formation of hydrogen from HF.
He H 1 2 2
These reactions allow for a change in the cathodic reaction from the reduction of salt cations to the hydrogen evolution reaction, which is a more thermodynamically favorable cathodic reaction for the oxidation of structural alloys. The presence of OH- in the salt has also been shown to greatly increase the corrosivity of molten salts. Potentiodynamic polarization of SS316L samples show a change in the cathodic branch of the plot when OH- is present.13 This observation implies that OH- is changing the cathodic reaction which occurs during oxidation of the metal; possibly due to the reduction of OH- to form H2 and O2- 13,17
OH eH O 1 2 2 2
The oxide ion is also corrosive to structural materials due to the formation of nonprotective oxide on the surface of structural alloys. The stability of these metal oxides is thought to depend on the basicity or pO2- of the salt.18,19 Highly basic salts, which have a high O2- concentration, attack surface oxide layers to create soluble oxometallates.18–22
Metal impurities in the salt, either formed as corrosion products or present in as-received salt, are also known to increase the corrosivity of molten halide salts.7,23 Less noble metals are selectively leached from homogenous alloys due to differences in reduction potentials among metallic constituents. Dissolved Ni(II) and Fe(II) have been shown to be highly oxidizing to Cr. Cr(III) and Fe(III) are both oxidizing to Cr, Fe, and Ni metal.7 The selective dissolution of these metals creates suboxide voids, leading to spallation of any formed oxide layer.24 This spallation exposes the metal to further oxidation and attack. Proper redox control of the salt can mitigate the presence of these oxidizing metallic impurities, through the consumption of active metals, such as Be metal, or by the use of an H2 cover gas.6 Thermal gradients are also a cause of corrosion in molten salts. Because these salts are often used as heat transfer fluids, substantial temperature gradients are expected across a given system. It has been shown that in a flowing thermal convection loop, mass loss is experienced in the hot leg and mass gain is observed in the cold leg.25–27 This observation has been attributed to a local temperature-dependent equilibrium between a metal and its dissolved cation. Cr dissolves into the salt as Cr(II) which can then spontaneous (continued on next page)
(continued from previous page)
disproportionate to Cr(III) and Cr(0). The disproportionation reaction is also seen for Fe(II). These relations are temperature dependent and are thought to be responsible for depositing metals, particularly Fe, in the cold leg of a thermal loop.27
Corrosion in Materials for Pyroprocessing
As mentioned, molten chloride salts are used for reprocessing of used nuclear fuel (UNF) through pyroprocessing. Originally developed by Argonne National Laboratory and demonstrated on a pilot scale at Idaho National Laboratory to reprocess metallic fuel from sodium-cooled fast reactors, pyroprocessing uses molten chlorides to electrochemically dissolve and deposit elements present in the UNF, allowing uranium and transuranic elements to be separated from fission products.28 Recovery of uranium and plutonium will allow for their reuse in nuclear reactors, thereby increasing the energy utilization of the original fuel. To accommodate ceramic fuels used in light water reactors, the ceramic spent fuel must first be converted into a metallic state. This can be accomplished through the electrochemical reduction of the UO2 through the following reaction in a molten LiCl salt with 1–2 wt% of added Li2O.29
UO eU O 2 2 42
To counterbalance the reduction of UO2, O2- is oxidized at an inert anode to form oxygen gas through the following reaction:
24 2 2OO e
The development of a long-lived, inert anode material in the LiClLi2O molten salt has been a significant challenge for pyroprocessing due to corrosion that occurs at the anode. Platinum, the most commonly used anode, corrodes through the formation of a nonprotective Li2PtO3 oxide layer that spalls into the salt over time.30 Due to the high cost of platinum and the need for periodic replacement, corrosion of the platinum anode represents a large material and time cost for the oxide reduction unit, requiring the identification of alternative anode materials. Because of the high anodic potential required to initiate the oxygen evolution reaction, metallic anode materials, including platinum group metals, will oxidize before oxygen evolution occurs.31,32 Therefore, the ability of a metallic anode to operate in the molten salt is dependent on the formation of a stable, passivating, and electrically conductive oxide on the surface.
Of the metallic anode materials tested, iridium and ruthenium appear to be the most promising anode materials, even though the nature of the formed oxide layers on the surface of these metals remains unknown.33,34
An alternative material class to metal anodes is conductive ceramics. Already present in an oxidized form, ceramic anodes can avoid the anodic dissolution that often occurs in metallic anodes. The ability of ceramic anodes to continuously evolve oxygen is dependent on the chemical stability of the ceramic in the molten LiCl-Li2O. Due to the low electrical conductivity of ceramics compared to metals, ceramic anodes are often tested in the form of a stable ceramic coating that is developed on the surface of a metal before being used in the LiCl-Li2O. A LiFe5O8 coated iron nickel alloy produced through oxidation in a molten carbonate salt and a NiO coated nickel electrode produced through the thermal oxidation of nickel have both been shown to be promising oxygen-evolving anode materials.35,36
In addition to corrosion at the anode, corrosion of container materials (including the cathode basket) in the LiCl-Li2O salt can be caused by metallic lithium that is produced at the cathode. UO2 reduction occurs at a potential only 70mV greater than lithium reduction as shown through the following reaction 37
Li eLi
While metallic lithium will chemically react with UO2 to produce metallic uranium, metallic lithium is soluble in the molten salt and can form clusters that allow for its accumulation in the salt bath.38 As a strong reducing agent, metallic lithium limits the formation of oxide layers on the surface of container materials.39 Despite the reducing environment imposed by the presence of metallic lithium, severe corrosion through dealloying and void formation has been observed for multiple candidate container materials when high concentrations of metallic lithium are present (>0.6 wt%).21,40–42 For example, in LiCl-Li2O with added lithium metal, 316L stainless steel undergoes preferential oxidation and carburization of chromium and manganese along the grain boundaries of the container material. Lithium metal has been shown to destabilize these oxides, resulting in the dissolution of chromium and manganese, leaving voids at the grain boundaries. Due to the high temperature of the LiCl-Li2O salt, diffusion of chromium and manganese from grains occurs over time, resulting in continued material depletion and void formation within the grains. A schematic detailing the corrosion mechanism of 316L in the LiCl-Li2O with metallic lithium is presented in Fig. 1.43
Selmi
Fig. 1. Corrosion mechanism of 316L in LiCl-Li2O with added lithium. A) Preferential oxidation and carburization of Cr and Mn at grain boundaries, B) Li metal dissolves formed oxides and carbides leaving voids at grain boundaries, C) Continued corrosion results in further intergranular dealloying. Diffusion in grains produces voids in bulk metal. Reproduced with copyright permissions.43
Liquid Alkali Metal Corrosion
Four primary liquid alkali metal (LAM) compositions are considered for high-temperature energy applications: Na, K, NaK, and Li. These LAMs offer melting temperatures below 200 °C, boiling points above 750 °C, and relatively high thermal conductivity, making them ideal for use in high-temperature systems such as nuclear reactor coolants and liquid metal battery (LMB) electrodes. However, due to their high reactivity with moisture and air, leakage of LAMs from containment is of particular concern.44
The primary mode of corrosion in pure LAMs is the selective dissolution of elements from the surface of the exposed structural metals, which will continue until the LAM becomes saturated.45 While saturation limits corrosion under static conditions, steadystate corrosion conditions are established in flowing systems because the liquid across the surface rarely reaches saturation.46,47 In flowing systems with a temperature or a concentration gradient, the chemical activity of the constituent elements varies, leading to the dissolution of constituents in the low concentration, or high temperature, side and their deposition in the high concentration, or low temperature, side.48 Due to the varying solubility of elements in the exposed alloy, the high-temperature process, and the continual loss and deposition of elements, phase changes become possible at the surface of the exposed metal, modifying the surface’s mechanical properties.49 As these LAM systems operate at temperatures that can exceed 500 °C, thermal aging processes can also occur in the metal. Thermal aging can lead to increased areas of nonuniform elemental distribution, such as Cr23C6 on grain boundaries in austenitic steels.50 Due to the relatively small size of LAMs, especially lithium, preferential dissolution may occur at the grain boundaries, leading to intergranular corrosion.43,51
Impurities, particularly oxygen, are of keen interest in terms of corrosion in molten alkali systems. Oxygen impurities impact LAM corrosion in two ways: (i) through the formation of soluble and insoluble binary and ternary oxide compounds due to the high affinity of alkali metals for oxygen, leading to increased depletion of alloying elements and (ii) by increasing the solubility of elements directly.45,46,52 Both of these processes exacerbate the dissolution conditions explained previously. The increase in solubility varies by element with Fe and Cr increasing with the amount of dissolved oxygen and Ni solubility being independent of oxygen concentration.46 Carbon impurities may also impact surface conditions of the metal and aid in phase transitions at the metal surface. Depending on the carbon activity in the LAM and the exposed metal, carburizing or decarburizing conditions may be created which can lead to surface softening of high temperature areas and hardening within low temperature areas.53 The simplest method for ensuring minimal impurities within flowing LAM systems is to use a cold and hot trap. Carbon, oxygen, and hydrogen decrease in solubility with decreasing temperature; therefore, LAM systems, particularly in nuclear reactors, use a cooled loop with a mesh filter to remove solidified impurities.54 The cooled LAM is then passed through a high-temperature region with an oxygen getter such as Zr that further reduces impurity concentration.55 In these purified conditions the LAM corrosion processes have relatively slow kinetics and therefore are not a major concern over most system lifetimes, especially for heavier LAMs.56
Liquid Heavy Metal Corrosion
In addition to LAMs being considered for high-temperature energy systems, liquid heavy metals (LHM) such as Pb and Sb alloys are promising for use as high-temperature coolants and electrodes for liquid metal battery (LMB) energy storage systems.57,58 These LHMs offer higher boiling points than alkali metals, allowing greater operating temperatures.59 Similar to the LAM systems, liquid metal corrosion (LMC) of structural materials is a major barrier for implementation of LHMs in high-temperature energy systems. Chemical activity acts as the driving force for dissolution of elements from the surface of the alloy into the liquid metal.60 This dissolution,
in conjunction with erosion from the flow of dense liquid metals, can lead to degradation of structural materials over time.61 Dissolved elements can form intermetallics which can increase erosion when solidified.57
Unlike in LAMs, dissolved oxygen alleviates the effects of corrosion in LHM systems by creating protective oxide films on alloy surfaces, as seen in Fig. 2A. The formation of heavy metal oxides is less favorable than the formation of oxides of common alloy components, leading to stable layers that protect the base structural metal.62 The formation of oxide films on steel has been proposed as a corrosion mitigation technique in these systems, with FeCrAl alloys and manufactured coatings being studied.63,64 Fe, Cr, and Al form sturdy oxides that are more stable than the oxide of the Pb or lead-bismuth eutectic. Ensuring continuity is of crucial importance for the long-term health of the structural material. LMC can occur where the oxide is not present causing liquid metal embrittlement, where the wetting of the structural alloy by the liquid metal leads to crack initiation, propagation, and inevitably failure as seen in Figure 2B.65,66 Of importance for oxide formation, especially continuous oxide layers, is the dissolved oxygen concentration within the liquid metal. Oxygen sensors that can operate within liquid metals are being developed to monitor the amount of dissolved oxygen and ensure healthy oxide formation on the structural material.67
For LMB applications, oxide formation on the positive current collector (PCC) becomes difficult to control and maintain. LMBs are built with two molten metal electrodes with respective current collectors and an electrolyte that can operate within the hightemperature conditions, typically a molten salt or a conductive ceramic.68 The negative electrode is commonly an alkali metal due to their high reactivity and low density, and elements like Sb and Pb are attractive options for LMB positive electrodes because of their lower cost and high expected discharge potentials with favored negative electrode candidates. However, the introduction of alkali metals into the LHM electrode disrupts the oxide film that forms on the PCC materials due to alkali metal affinity for oxygen.69 Immersion tests have shown that lithiated Sb-alloys commonly form intermetallic phases with PCC compositional components, such as Fe-Sb, Fe-NiCr-Sb, Fe-Cr-Sb, and Ni-Sb alloys with a failure to form a continuous oxide layer.69,70 These intermetallic phases not only threaten the structural integrity of the PCC, but may also lead to the consumption of Sb due to the formation of a Ni-Sb alloy that limits availability for lithiation and causes a drop in capacity over the cycle life. Different PCC compositions are being investigated to minimize intermetallic phase effects and consumption of the active Sb by lowering Ni content in the alloys.69–71 Additionally, low and intermediate-temperature LMB systems are being investigated to minimize LMC by lowering the operating temperature of the system to minimize solubility of PCC components into the liquid metals.72
Conclusions
Many next-generation energy technologies rely on high temperatures to increase efficiency and/or reprocess the fuel, requiring the use of molten materials for energy conversion. These molten media include molten halide salts, molten alkali metals, and molten heavy metals. Each of these systems has varying impurities that present unique challenges with differing corrosion mechanisms. Some applications require exposed metals to have uniform protective oxide layers, such as in an LHM, or anodes for pyroprocessing. In other applications, such as those with LAMs, continuous and protective oxide layers usually cannot form, and structural materials are continuously attacked. The same is true for structural materials in molten salt systems. Because of the instability of oxide layers in these systems, they require different forms of corrosion protection and mitigation. Understanding and preventing corrosion in these energy systems are key to enabling their commercial reliability.
© The Electrochemical Society. DOI:10.1149/2.F08261IF
(continued on next page)
About the Authors References

The authors (from left) Tolin Skov-Black, Craig Moore, Jonathan Ramos, Dev Chidambaram, Jake Kattelman, and Thomas Selmi) are from the Materials and Electrochemical Research (MER) laboratory in Materials Science and Engineering at the University of Nevada, Reno (UNR).
This article is written by Professor Dev Chidambaram and graduate student researchers Craig Moore, Jake Kattelman, Thomas Selmi, Tolin Skov-Black, and Jonathan Ramos. All the students are native Nevadans who completed their bachelor’s degrees at the University of Nevada, Reno, and are continuing their graduate training in various nuclear-related molten systems (salts and metals) in Materials Science and Engineering at UNR. Selmi, Kattelman, Moore, and Ramos are pursuing PhD degrees, while Skov-Black is completing an MS degree; Skov-Black is also a US Navy veteran. Moore, Selmi, and Skov-Black are Department of Energy Nuclear Energy Graduate Fellows.
Craig Moore is developing new stable anodes for pyroprocessing of used nuclear fuel. Thomas Selmi is developing new methods to identify and quantify moisture and related impurities in molten salts, while Jonathan Ramos is studying how those impurities affect corrosion behavior. Tolin Skov-Black is studying the effect of oxygen impurity on corrosion of materials in liquid sodium. Jake Kattelman is developing liquid metal batteries using liquid sodium. All group members use a complementary suite of techniques to characterize their systems.
1. W. Ding, A. Bonk, and T. Bauer, Front Chem Sci Eng, 12(3), 564 (2018).
2. G. S. Was, D. Petti, S. Ukai, and S. Zinkle, J Nucl Mater, 527, 151837 (2019).
3. M. A. Williamson and J. L. Willit, Nucl Eng Technol, 43 (4), 329 (2011).
4. S. S. Raiman and S. Lee, J Nucl Mater, 511, 523 (2018).
5. T. Ghaznavi, S. Y. Persaud, and R. C . Newman, J Electrochem Soc, 169 (6), 061502 (2022).
6. J. Zhang, C. W. Forsberg, M. F. Simpson, S. Guo, S. T. Lam, et al., Corros Sci, 144, 44 (2018).
7. S. Guo, J. Zhang, W. Wu, and W. Zhou, Prog Mater Sci, 97, 448 (2018).
8. W. J. Burkhard and J. D. Corbett, J Am Chem Soc, 79(24), 6361 (1957).
9. S. Adams, Analysis of Process Monitoring to Support Safety and Safeguards Requirements for a Spent Nuclear Fuel Electrorefiner, University of Utah, 2017 (accessed 2025-10-23).
10. A. Schneider, D. Andersson, and Y. Zhang, Commun Mater, 5(1), 91 (2024).
11. M. Takeuchi, R. Kurosawa, J. Ryu, and M. Matsuoka, ACS Omega, 6(48), 33075 (2021).
12. R. V. Gough, K. M. Primm, E. G. Rivera-Valentín, G. M. and M. A. Tolbert, Icarus, 321, 1 (2019).
13. T. R. Selmi and D. Chidambaram, J Radioanal Nucl Chem, 2025.
14. P. E. Field and J. H. Shaffer, J Phys Chem, 71(10), 3218 (1967).
15. J. D. Van Norman and R. J. Tivers, J Electrochem Soc, 118(2), 258 (1971).
16. S. Pizzini and R. Morlotti, Electrochimica Acta, 10(10), 1033 (1965).
17. Y. Kanzaki and M. Takahashi, J Electroanal Chem Interfacial Electrochem, 58(2), 349 (1975).
18. L. Sharpless, J. Moon, and D. Chidambaram, J Mol Liq, 394, 123803 (2024).
19. A. Merwin and D. Chidambaram, Corros Sci, 126, 1 (2017).
20. Y. Peng, K. K. Bawane, X. Liu, X. Zheng, M. Ge et al., ACS Appl Mater Interfaces, 17, 28764 (2025).
21. W. Phillips, A. Merwin, and D. Chidambaram, Metall Mater Trans A, 49(6), 2384 (2018).
22. A. Merwin and D. Chidambaram, Nucl Technol, 195(2), 204 (2016).
23. Y. Wang, S. Zhang, X. Ji, P. Wang, and W. Li, Int J Electrochem Sci, 13(5), 4891 (2018).
24. G. Salinas-Solano, J. Porcayo-Calderon, J. G. GonzalezRodriguez, V. M. Salinas-Bravo, J. A. Ascencio-Gutierrez et al., Adv Mater Sci Eng, 2014(1), 696081 (2014).
25. B. A. Pint, Y.-F. Su, D. Sulejmanovic, and R. Pillai, Mater High Temp, 40(4), 360 (2023).
Fig. 2. A) The formation of a protective oxide layer on an FeCrAl alloy exposed to oxygenated LHM preventing further crack propagation. B) Dissolution of an FeCrAl alloy exposed to deoxygenated LHM causing crack initiation and propagation due to no protective oxide formation.
26. B. A. Pint, J. W. McMurray, A. W. Willoughby, J. M. Kurley, S. R. Pearson et al., Mater Corros, 70(8), 1439 (2019).
27. S. S. Raiman, J. M. Kurley, D. Sulejmanovic, A. Willoughby, S. Nelson et al., J Nucl Mater, 561, 153551 (2022).
28. J. J. Laidler, J. E. Battles, W. E. Miller, J. P. Ackerman, and E. L. Carls, Prog Nucl Energy, 31(1–2), 131 (1997).
29. E.-Y. Choi and S. M. Jeong, Prog Nat Sci: Mater Int, 25(6), 572 (2015).
30. S.-K. Lee, M. K. Jeon, S.-W. Kim, E.-Y. Choi, J. Lee, J Radioanal Nucl Chem, 316(3), 1053 (2018).
31. S.-W. Kim, S.-K. Lee, H. W. Kang, E.-Y. Choi, W. Park et al., J Radioanal Nucl Chem, 311(1), 809 (2017).
32. A. Merwin and D. Chidambaram, Metall Mater Trans A, 46(1), 536 (2015).
33. S. D. Herrmann, P. K. Tripathy, S. M. Frank, and J. A. King, J Appl Electrochem, 49(4), 379 (2019).
34. D. C. Horvath, J. A. King, R. O. Hoover, S. A. Warmann, K. C. Marsden et al., J Nucl Fuel Cycle Waste Technol, 20, (2022)
35. K. Du, E. Gao, C. Zhang, Y. Ma, P. Wang et al., Nat Commun, 14(1), 253 (2023).
36. Y. Sakamura, T. Murakami, M. Iizuka, and H. Kofuji, J Electrochem Soc, 169(6), 063504 (2022).
37. A. Merwin, M. A. Williamson, J. L. Willit, and D. Chidambaram, J Electrochem Soc, 164(8), H5236 (2017).
38. A. Merwin, W. C. Phillips, M. A. Williamson, J. L. Willit, P. N. Motsegood et al., Sci Rep, 6(1), 25435 (2016).
39. W. Phillips and D. Chidambaram, J Electrochem Soc, 166(11), C3193 (2019).
40. A. Merwin and D. Chidambaram, Corros Sci, 126, 1 (2017).
41. W. Phillips, Z. Karmiol, and D. Chidambaram, J Electrochem Soc, 166(6), C162 (2019).
42. A. Merwin and D. Chidambaram, Nucl Technol, 195(2), 204 (2016).
43. J. T. Moon, W. Phillips, W. Chuirazzi, J. Kane, and D. Chidambaram, Corros Sci, 228, 111836 (2024).
44. L. Zhang, C. Deng, X. Ji, and X. Liu, The Innovation, 6(9), 100959 (2025).
45. D. Féron, Nuclear Corrosion Science and Engineering; Woodhead publishing series in energy; Woodhead Publ.: Cambridge, 2012.
46. J. R. Weeks and H. S. Isaacs, In Advances in Corrosion Science and Technology; Fontana, M. G., Staehle, R. W., Eds.; Springer US: Boston, MA, 1973; pp 1–66.
47. T. Suzuki, I. Mutoh, T. Yagi, and Y. Ikenaga, J Nucl Mater, 139(2), 97 (1986).
48. E. Yoshida, S. Kato, and Y. Wada, In Liquid Metal Systems: Material Behavior and Physical Chemistry in Liquid Metal Systems 2; Borgstedt, H. U., Frees, G., Eds.; Springer US: Boston, MA, 1995; pp 55–66.
49. V. Ganesan and V. Ganesan, J Nucl Mater, 256(1), 69 (1998).
50. C. Sudha, N. Sivai Bharasi, R. Anand, H. Shaikh, R. K. Dayal et al., J Nucl Mater, 402(2–3), 186 (2010).
51. X. Meng, G. Zuo, J. Ren, W. Xu, Z. Sun et al., J Nucl Mater, 480, 25 (2016).
52. H. U. Borgstedt, Material Behavior and Physical Chemistry in Liquid Metal Systems; Springer US: Boston, MA, 1982.
53. J. R. Gwyther, M. R. Hobdell, and A. J. Hooper, Metals Technol, 1(1), 406 (1974).
54. F. Zhao and X. Ren, Nucl Eng Des, 239(3), 490 (2009).
55. F. A. Kozlov, V. V. Alexeev, Yu. P. Kovalev, V. Ya. Kumaev, V. V. Matyuchin et al. At Energy, 112(1), 21 (2012).
56. R. S. Fidler and M. J. Collins, At Energy Rev, 13(1), 3 (1975)
57. C. Fazio, V. Sobolev, A. Aerts, S. Gavrilov, K. Lambrinou et al., Handbook on Lead-Bismuth Eutectic Alloy and Lead Properties-2015 Edition; Organisation for Economic CoOperation and Development, Nuclear Energy, 2015.
58. J. Kim, D. Shin, Y. Jung, S. M. Hwang, T. Song et al., J Power Sources, 377, 87 (2018).
59. T. R. Allen, D. C. Crawford, Sci Technol Nucl Install, 2007, 1 (2007).
60. OECD; Nuclear Energy Agency. Handbook on Lead-Bismuth Eutectic Alloy and Lead Properties, Materials Compatibility, Thermalhydraulics and Technologies; Nuclear Science; OECD, 2015.
61. J. Zhang and N. Li, J Nucl Mater, 373 (1), 351 (2008).
62. F. Barbier and A. Rusanov, J Nucl Mater, 296(1), 231 (2001).
63. J. Lim, I. S. Hwang, and J. H. Kim, J Nucl Mater, 441(1), 650 (2013).
64. Y. Zhang, X. Pan, X. Cao, H. Jiang, D. Yan et al., J Nucl Mater, 615, 155931 (2025).
65. X. Gong, J. Chen, C. Xiang, Z. Yu, H. Gong et al., Corros Sci, 183, 109346 (2021).
66. J. E. Norkett, M. D. Dickey, and V. M. Miller, Metall Mater Trans A, 52(6), 2158 (2021).
67. S. N. Orlov, N. A. Bogachev, A. S. Mereshchenko, A. A. Zmitrodan, M. Skripkin et al., Sensors, 23(2), 812 (2023).
68. H. Kim, D. A. Boysen, J. M. Newhouse, B. L. Spatocco, B. Chung et al., Chem Rev, 113(3), 2075 (2013).
69. K. Cui, F. An, W. Zhao, P. Li, S. Li et al., J Phys Chem C, 125(1), 237 (2021).
70. T. Ouchi and D. R. Sadoway, J Power Sources, 357, 158 (2017).
71. R. Fetzer, T. Zhang, A. Heinzel, A. Weisenburger, and G. Müller, J Power Sources, 623, 235473 (2024).
72. J. Kattelman, J. Moon, and D. Chidambaram, J Electrochem Soc, 172(2), 020528 (2025).








Digital Twin Framework for Predictive Maintenance and Materials Selection Facing Climate Change
by Mikhail Zheludkevich and Daniela Jacob
The resilience and longevity of metallic infrastructures are increasingly challenged by the growing impacts of climate change, which manifest through complex and evolving environmental stressors. Traditional approaches to corrosion management, which have historically relied on empirical models, periodic inspections, and the extrapolation of historical climate data, are proving insufficient in the context of climate change, including rapidly changing weather conditions. This paper articulates a scientific vision for the future of predictive maintenance and materials selection for metallic structures exposed to dynamic climate regimes. Central to this vision is the development of an integrated digital twin framework that couples high-resolution regional-to-local climate modeling with advanced corrosion prediction methodologies, encompassing
Metallic infrastructure forms the backbone of modern civilization, underpinning critical sectors such as transportation, energy, water supply, and housing.
The durability and operational reliability of these assets are increasingly threatened by the multifaceted impacts of climate change, which introduce novel and complex environmental stressors. These include, but are not limited to, rising global-to-local temperatures, altered humidity and precipitation patterns, increased frequency and severity of extreme weather events, and elevated levels of atmospheric pollutants and salts. Collectively, these factors contribute to the accelerated degradation of metallic structures, with profound economic implications. The annual global cost of corrosion is estimated to exceed $2.5 trillion, and climate change is projected to intensify these losses by increasing atmospheric corrosivity and reducing the service life of critical infrastructure.1 For example, projections for coastal regions in the United States indicate that the useful life of steel structures may decrease by up to 16% by the end of the 21st century under high-emission scenarios, with direct economic losses exceeding $100 per square meter for bridges alone.2 This is consistent with global trends, with infrastructure in Europe, Asia, and Australia facing heightened risks due to changing patterns of temperature, humidity, and airborne pollutants.3,4
Traditional approaches to corrosion management are predominantly reactive, relying on historical climate data, empirical models, and periodic inspections. These methods are often static, calibrated for a narrow range of materials and environmental conditions, and typically neglect the effects of protective coatings, multi-material assemblies, or the impact of extreme events.5 The limitations of current corrosion prediction models are well known: many widely used models are empirical, lacking mechanisms for real-time updating or validation, and are thus unable to accommodate the dynamic and complex challenges posed by climate change. As a result, maintenance strategies are frequently suboptimal, failing to address the evolving risks associated with changing environmental exposures.
A paradigm shift is urgently required, moving from reactive maintenance and generic materials selection to predictive, data-driven, and location-specific strategies. This vision is enabled by recent advances in several key areas. High-resolution climate modeling, facilitated by regional climate models and downscaling techniques,
both physics-based and data-driven approaches. The digital twin is dynamically updated through an active feedback loop incorporating real-time data from environmental and corrosion sensors, as well as periodic inspection records. In addition, the digital twin framework will be used to understand the consequences of future climate conditions. Designed to operate at spatial resolutions down to a few kilometers, the system enables site-specific risk assessments and supports informed decision-making for both maintenance scheduling and the selection of materials for new infrastructure. The framework further integrates scenario analysis, uncertainty quantification, and risk-based decision-making, thereby supporting the transition from reactive to predictive and adaptive asset management strategies.
now provides detailed projections of temperature, humidity, wind, salt, and pollutant concentrations at spatial resolutions down to a few kilometers.6 The integration of physics-based corrosion models with data-driven approaches, including machine learning and artificial intelligence, allows for the assimilation of large, heterogeneous datasets from sensors, inspections, and historical records.7,8 Advances in sensor technology and in the Internet of Things (IoT) enable the deployment of distributed networks of atmospheric and corrosion sensors, providing continuous, high-resolution data for model calibration, uncertainty reduction, and early warning of accelerated degradation.
The concept of a digital twin is based on a dynamic, virtual replica of a physical asset. It offers a powerful framework for integrating climate projections, corrosion models, sensor data, and decisionsupport tools.9 Through active feedback loops, the digital twin continuously updates its predictions, supports scenario analysis, and recommends optimal maintenance actions and materials selection.
Climate Change and Atmospheric Corrosivity
Climate change is exerting a profound influence on the corrosion of metallic and reinforced concrete structures worldwide, with significant implications for infrastructure durability, safety, and economic sustainability. The primary mechanisms by which climate change affects corrosion are through alterations in environmental parameters such as temperature, relative humidity, atmospheric CO₂ concentration, precipitation patterns, and the frequency and intensity of extreme weather events. These changes interact with material properties and local exposure conditions, resulting in complex, often nonlinear effects on corrosion rates and structural degradation. The relationship between corrosion rate and relative humidity (RH) is nonlinear, and the presence of pollutants such as NaCl (from marine aerosols) or SO₂ (from industrial emissions) can further accelerate corrosion, especially in coastal or urban environments. For example, Mansfeld’s classic experiments showed that the corrosion rate of rust-covered steel increases exponentially with RH above the critical threshold, and that NaCl contamination can amplify this effect by an order of magnitude.10
(continued on next page)
Zheludkevich and Jacob
(continued from previous page)
Modeling local climate conditions in the context of global warming is a complex task that relies on ensembles of climate projections, combining global and regional climate models to capture both large-scale trends and local variability. The process begins with representative concentration pathways (RCPs), which are assumptions for the development of future greenhouse gas concentrations based on socio-economic trajectories, which in turn are dependent on human activities in mitigating greenhouse gases. The RCPs drive global Earth system models (ESMs) that simulate atmosphere, land, and ocean dynamics as well as their interactions at global-to-regional scales. However, due to computational restrictions, ESMs perform on coarse spatial resolutions, which limits their ability to represent local phenomena, such as the influence of mountains, proximity to water bodies, and land use. To address this, regional climate models (RCMs) are developed within ESMs, providing higher spatial resolution (down to approx. 3 km in the EURO-CORDEX initiative) and enabling more accurate projections of, for example, temperature, precipitation, and humidity at the local level,6,7 see Figure 1.
The robustness of these projections is assessed using climate signal maps, which condense the ensemble output to highlight regions where model agreement and statistical significance indicate reliable trends. For Germany and much of Europe, these models consistently project increases in winter precipitation and temperature, with the magnitude and spatial pattern depending on the scenario. Under high-emission scenarios such as RCP8.5, temperature increases of up to 6–8°C in southern Europe during summer are expected, while northern Europe may see more pronounced changes in winter.6 Relative humidity, a key driver of corrosion processes, is projected to remain roughly constant at the planetary scale, but with regional decreases in southern Europe and increases in the north, reflecting changes in soil moisture. Salt deposition, primarily from sea spray, is another critical factor, especially for coastal infrastructure. Changes in wind patterns and oceanic conditions under global warming can alter the production and transport of salt aerosols, potentially increasing corrosion rates in some regions while decreasing them in others. The interplay between rainfall and salt deposition is complex: less frequent rainfall can lead to higher concentrations of corrosive agents on metal surfaces, exacerbating degradation.11 Studies in Australia have shown that, under future climate scenarios, corrosion rates in coastal cities could increase by 18–415% by 2070, primarily due to increased salt retention from less frequent rain events, while inland locations may see a moderate decrease in corrosion due to reduced RH and less condensation.12
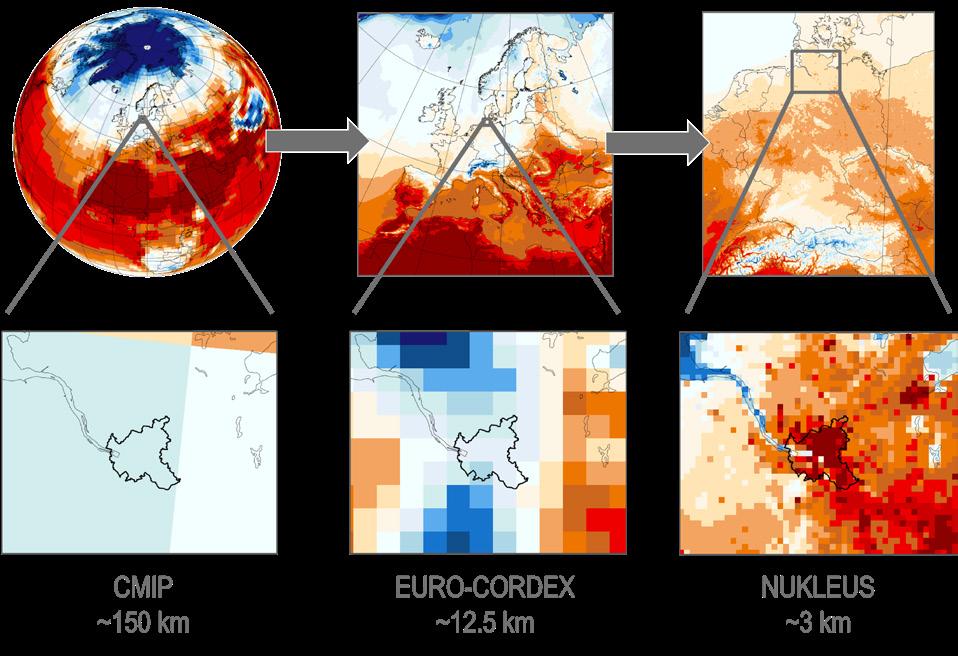
Corrosive Degradation of Metallic Structures
For metallic structures, atmospheric corrosion is fundamentally governed by the presence of electrolyte layers on the metal surface, which enable electrochemical reactions. The time-of-wetness (TOW), defined as the period during which the surface remains sufficiently moist for corrosion to occur, is a critical parameter. Laboratory and field studies demonstrate that the corrosion rate of metals such as steel increases sharply once relative humidity exceeds a critical threshold, typically around 40–60%, with the maximum corrosion rate often observed at 60–90% RH. The relationship between corrosion rate and RH is nonlinear, and the presence of pollutants such as NaCl (from marine aerosols) or SO₂ (from industrial emissions) can further accelerate corrosion, especially in coastal or urban environments.10
Climate change is expected to alter both the average and the variability of temperature and humidity in many regions. Recent climate projections for Europe and other regions indicate that by 2100, average temperatures could rise by 2–4°C under moderate scenarios (RCP4.5) and by up to 6–10°C under extreme scenarios (RCP8.5), with relative humidity changes generally smaller and locally very variable but still significant in some areas.5 Rising temperatures generally increase the rate of electrochemical reactions, enhancing corrosion rates, but the effect is modulated by humidity. In humid climates, higher temperatures can keep surfaces wet for longer periods, increasing TOW and thus corrosion. In contrast, in dry climates, increased temperatures reduce surface moisture slowing down corrosion. The combined effect of temperature and humidity is particularly important for reinforced concrete structures, where carbonation-induced corrosion is a major concern. Carbonation occurs when atmospheric CO₂ diffuses into concrete, reacting with calcium hydroxide and reducing the alkalinity that protects steel reinforcement. The rate of carbonation, and thus the risk of corrosion initiation, increases with higher CO₂ concentrations and temperatures, and is maximized at intermediate RH (typically 60–70%).5 These effects are more severe in older concrete buildings, particularly those constructed before the adoption of modern durability standards. For example, a pan-European assessment by the Joint European Commission's Research Centre found that, under RCP8.5, carbonation depths in old buildings could increase by 58–89% by 2085 compared to a no-climate-change baseline, triggering corrosion initiation decades earlier than previously expected. The associated repair costs for the EU-27 could reach €883 billion by 2100 under the extreme scenario.5
Extreme weather events, such as storms, floods, and heatwaves, are expected to become more frequent and severe under climate change, posing additional risks to infrastructure. Flooding can cause immediate structural damage and deposit salts and pollutants that lead to long-term corrosion, while heatwaves can accelerate chemical reactions and, in some cases, cause thermal cracking that exposes fresh surfaces to attack. The effect of climate change on corrosion is not uniform across regions or materials. In Europe, for example, the greatest increases in carbonation depth and corrosion risk are projected for northern and eastern cities, where temperature rises are expected to be largest and where older buildings often have lower concrete cover and less durable materials. In southern Europe, despite higher absolute temperatures, the effect may be mitigated by lower RH or higher cement content in concrete.13
Air pollution remains a significant co-factor in climate-driven corrosion. While SO₂ levels have declined in many regions due to emission controls, other pollutants such as NOₓ, ozone, and particulate matter continue to contribute to material degradation. Dose–response functions (DRFs) developed in Europe and China have shown that the combined effects of multiple pollutants and climate variables can be quantitatively linked to corrosion rates, enabling the mapping of high-risk areas and informing policy interventions. However, these models are empirical and must be regularly updated to reflect changing environmental conditions and material properties.13
Fig. 1. Regional and local climate prediction maps with resolution down to 3 kilometers.
Predictive Corrosion Modeling Approaches
Corrosion modeling has evolved from empirical and physicsbased approaches to increasingly sophisticated data-driven and hybrid frameworks, each with distinct strengths and limitations. Traditional physics-based models, such as those grounded in electrochemical theory, finite element analysis (FEA), and firstprinciples calculations (e.g., density functional theory, DFT), have long been the foundation for understanding and predicting corrosion processes. These models are built on established physical laws and mechanistic understanding, allowing for the simulation of corrosion phenomena under controlled conditions and the exploration of the effects of variables such as temperature, humidity, alloy composition, and environmental chemistry.14
However, the limitations of current physics-based models are increasingly apparent in real-world applications. Firstly, these models often require simplifying assumptions to make the problem manageable, such as idealized geometries, homogeneous materials, or simplified or constant environmental conditions. As a result, they may not capture the full complexity of corrosion processes in heterogeneous, dynamic, or multi-scale environments. For example, DFT and molecular dynamics (MD) simulations are powerful for elucidating atomic-scale mechanisms, but are computationally expensive and limited to small model systems and short timescales. FEM and phase-field models can simulate larger-scale phenomena, such as pit growth or stress corrosion cracking, but require extensive parameterization and may not account for all relevant microstructural or environmental factors. Furthermore, the accuracy of these models is often constrained by the availability and quality of input data, such as reaction rate constants, diffusion coefficients, and environmental exposure histories.15
Data-driven models, particularly those based on machine learning (ML) and artificial intelligence (AI), have emerged as powerful tools for predicting corrosion rates, defect growth, and material lifetimes from large, complex datasets. These models excel at capturing nonlinear relationships and high-dimensional interactions among variables, often outperforming traditional regression or empirical models in predictive accuracy. For example, random forests, support vector machines, and neural networks have been successfully applied to predict atmospheric corrosion rates, pitting behavior, and inhibitor performance using features such as alloy composition, environmental parameters, molecular descriptors, and sensor data. Deep learning approaches, including convolutional neural networks (CNNs) and recurrent neural networks (RNNs), have shown promise in imagebased corrosion detection and time-series prediction of degradation.8
Despite their strengths, data-driven models also have significant limitations. Their predictive power is highly dependent on the availability of large, high-quality, and representative datasets. In many corrosion applications, such datasets are scarce, noisy, or biased, limiting the generalizability and robustness of purely data-driven models. Moreover, these models often function as “black boxes” lacking interpretability and physical insight, which can undermine trust and hinder their adoption in safety-critical or regulatory contexts. Without explicit incorporation of physical laws, data-driven models may produce predictions that violate known constraints or fail to extrapolate reliably to new conditions.16
To address these challenges, there is a growing need for advanced hybrid models that integrate physics-based understanding with datadriven learning. Hybrid approaches seek to combine the mechanistic fidelity and interpretability of physics-based models with the flexibility and pattern-recognition capabilities of ML/AI. One promising direction is the development of physics-informed machine learning (PIML) frameworks, where physical laws are embedded as constraints, inductive biases, or custom loss functions within neural networks. For example, physics-constrained neural networks can be trained to respect conservation laws, irreversibility of corrosion, or known boundary conditions, improving generalization and reducing the risk of unphysical predictions. Bayesian networks and
probabilistic graphical models offer another avenue for integrating prior knowledge, experimental data, and uncertainty quantification in a unified framework.17,18
Recent studies have demonstrated the effectiveness of hybrid approaches in corrosion modeling. For instance, a physics-informed, data-driven model for atmospheric corrosion of carbon steel was developed by calibrating a physics-based simulation with real-world monitoring data using a Bayesian network. This approach allowed for the updating of model parameters and uncertainty reduction, resulting in improved predictive accuracy and applicability to specific environments.17 Similarly, in the context of pitting corrosion in aluminium alloys, integrating finite element modeling (FEM) with machine learning (random forests) enabled more accurate prediction of pit area and depth distributions, outperforming conventional datadriven models and providing probabilistic assessments of localized corrosion19 as schematically shown in Figure 2. Physics-informed deep learning frameworks have also been proposed to guide neural network predictions with discovered or known physical relationships, reducing data requirements and enhancing interpretability.16
A critical aspect of advanced corrosion modeling is uncertainty reduction. Both physics-based and data-driven models are subject to uncertainties arising from model structure, parameter estimation, measurement noise, and environmental variability.20 Hybrid approaches can mitigate these uncertainties by leveraging complementary strengths: physics-based models provide structure and constraints, while data-driven components can learn residual patterns, correct for model bias, and adapt to new data. Techniques such as Bayesian inference, Gaussian process surrogate modeling, and adaptive sampling have been used to quantify and reduce uncertainty in corrosion predictions, enabling more reliable risk assessments and decision-making. Feature importance analysis, such as SHAP values in neural networks, can further enhance interpretability and guide data collection by identifying the most influential environmental or material factors.21
Nevertheless, challenges remain. The integration of physics and data-driven methods requires careful design to avoid overfitting, ensure consistency, and balance computational cost. The development of standardized, high-quality corrosion datasets and open datasharing practices is essential to support the training and validation of advanced models. Moreover, the field would benefit from continued research into explainable AI, transfer learning, and active learning strategies to improve model robustness and applicability across diverse materials, environments, and timescales.
From Accelerated Test to in-Service Performance
Accelerated corrosion tests are widely employed as practical tools to predict the long-term atmospheric corrosion behavior of materials and coatings, especially when real-world exposures are prohibitively time-consuming and resource-intensive. These tests, such as cyclic corrosion tests, salt spray, and humidity chamber exposures, are designed to simulate the environmental stresses materials will face in service, but within a compressed timeframe. Their value lies in enabling rapid screening of materials, comparative ranking of protective systems, and supporting the qualification of new alloys or coatings, particularly in sectors like automotive, marine, and aerospace. However, the translation of accelerated test results to realworld performance is associated with challenges and limitations that stem from the inherent complexity and variability of atmospheric corrosion processes.22
One of the principal challenges is the representativeness of accelerated tests, especially for weather conditions in future climates. While these tests can mimic certain aspects of service environments, such as wet/dry cycling, salt deposition, and temperature fluctuations, they often fail to capture the full spectrum of environmental variables and their dynamic interactions. For example, the relative humidity (RH) and temperature cycles in laboratory tests are typically more regular and less extreme than those experienced in the field.
(continued on next page)
(continued from previous page)
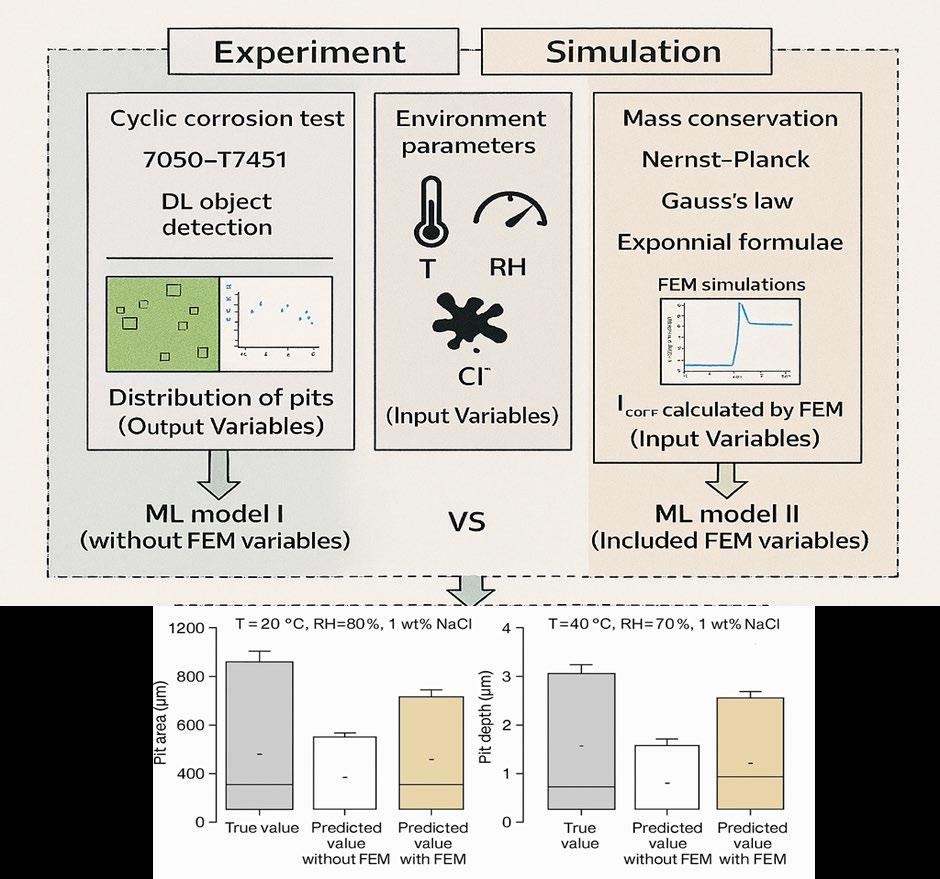
Moreover, the presence of pollutants, UV radiation, and microclimatic effects (such as those found on operating aircraft or in sheltered locations) are difficult to replicate accurately. As a result, the corrosion mechanisms and morphologies observed in accelerated tests may differ significantly from those in natural exposures. For
instance, certain tests may promote blistering or filiform corrosion that is not prevalent in service, or conversely, may underestimate the risk of underfilm corrosion or pitting that develops over longer timescales.23
A further complication arises from the role of drying phases and the hygroscopicity of corrosion products and salts. Recent research has highlighted that RH during the dry phases of cyclic tests can have a profound effect on corrosion rates and mechanisms. For example, drying at 70% RH may not be sufficient to halt corrosion on steel or zinc surfaces due to the persistence of thin electrolyte films, while lower RH levels (e.g., 30–50%) are more effective in interrupting corrosion cycles and better reflect real-world drying conditions.24
Given these challenges, there is a growing consensus that coupling accelerated corrosion tests with advanced modeling approaches is essential for achieving robust, predictive connections between laboratory data and service performance. Modeling can take several forms, from empirical dose–response functions and power-law kinetics to more sophisticated machine learning and transfer learning frameworks. By integrating environmental data (such as temperature, RH, pollutant levels) and mechanistic understanding of corrosion processes, models can help interpret accelerated test results in the context of real-world exposures, identify key variables driving
corrosion, and extrapolate short-term laboratory data to long-term
Recent advances in machine learning, particularly transfer learning, have shown promise in bridging the gap between controlled accelerated tests and unpredictable outdoor environments. For
example, models trained on rich datasets from accelerated tests can be fine-tuned with limited field data to improve their generalizability and predictive accuracy. This approach is especially valuable when field data are scarce or when environmental conditions differ substantially between laboratory and service settings. However, the success of such models depends on the quality and representativeness of both the test data and the environmental descriptors used in modeling.26
Real-time monitoring is pivotal for transitioning from reactive, schedule-based maintenance to proactive, condition-based strategies that are sensitive to local microclimates and evolving exposure. A robust sensing strategy for metallic structures draws on three complementary modalities: atmospheric sensors that characterize the driving environment; corrosion-specific sensors that quantify degradation kinetics; and structural health monitoring (SHM) sensors that capture the mechanical response and integrity of the asset. Atmospheric packages typically include calibrated temperature and relative humidity probes, barometers, wind speed/direction anemometers, rainfall gauges, and targeted pollutant analyzers for SO₂, NOₓ, O₃, and particulate matter. In coastal zones, salt deposition is measured using dry/wet collectors, wet candle methods, or electrochemical chloride sensors to estimate deposition fluxes and retention, which are critical when rainfall frequency decreases and
Jacob
Fig. 2. Schematic illustration of hybrid modeling workflow and its outcomes for prediction of pitting corrosion of aluminum alloy (stylized from ref. 19).
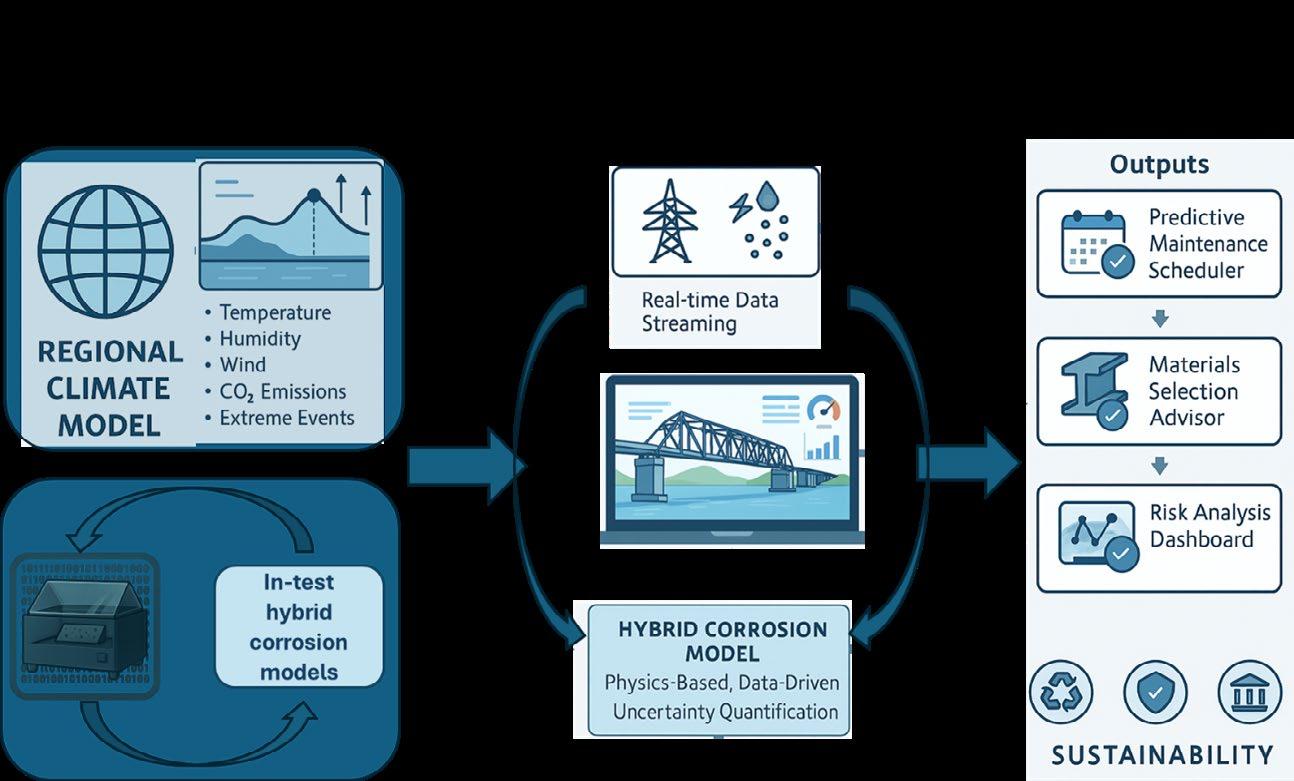
natural surface cleaning diminishes. TOW can be approximated by thresholding RH and temperature, but direct TOW sensors, based on conductive tracks or impedance films, provide superior fidelity by integrating surface wetness, contamination, and microcondensation phenomena.27
From Models Coupling to Digital Twins
A digital twin framework for corrosion–prone metallic infrastructure is a sophisticated virtual model that should act as a living digital copy of a real-world asset, such as a bridge, pipeline, or tower. This digital twin brings together information from many sources, helping owners and engineers understand the current state of the structure and predict how it might change in the future, especially as the climate and environment evolve. The architecture of the digital twin can be organized into six closely connected layers, each playing a specific role in the system, as shown in Figure 3.
At the foundation is the data layer, which is responsible for collecting and organizing all the information needed to understand the asset. This layer includes data from sensors that measure weather conditions, corrosivity, and the structural health of the asset. It also incorporates inspection reports, maintenance records, and details about the asset’s materials, coatings, and construction history. All this information is carefully managed to ensure that it is accurate, upto-date, and linked to the correct part of the structure, with digital mapping tools and building information models helping to keep everything organized and connected to the asset’s real-world location and design.
An additional layer in the digital twin should focus on digitalized accelerated corrosion tests that replicate complex cyclic environmental conditions in a virtual environment. This layer captures the protocols of tests such as cyclic corrosion, salt spray, and humidity exposure, encoding their parameters and outcomes into structured digital workflows. It is linked in a closed loop with hybrid models, combining physics-based simulations and machine learning to calibrate predictions and extrapolate short-term laboratory data to long-term service performance. By integrating these tests, the digital twin can simulate extreme scenarios, validate corrosion mechanisms, and refine uncertainty estimates through adaptive learning.
A specialized layer for local climate models provides essential input for digital twins by delivering high-resolution, region, and
site-specific projections. It combines real-time sensor data, historical trends, and advanced algorithms to generate accurate localized climate projections. Crucially, it incorporates different climate change scenarios, enabling simulations of long-term impacts and extreme events under varying conditions. By feeding dynamic climatechange data into the digital twin, the system remains responsive to environmental variability. This approach ensures that the digital twins reflect realistic, evolving climate conditions rather than static assumptions.
The corrosion modeling layer uses this wealth of information to simulate how the environment and corrosion affect the asset over time. It combines physics-based models, which use scientific formulas and standards to predict corrosion, with data-driven models that use artificial intelligence and machine learning to find patterns in large amounts of data. By integrating these approaches, the digital twin can provide more accurate forecasts and also keep track of uncertainties, showing where predictions are more or less reliable.
The learning and inference layer allows the digital twin to improve with new data. Advanced statistical methods help the system update its predictions whenever new sensor readings or inspection results are available. The digital twin can also run “what-if” scenarios, such as predicting the impact of extreme weather events or changes in maintenance strategies, ensuring the model stays relevant and accurate over time.
The applications layer is where the digital twin turns its insights into practical advice and decisions. It can suggest the best times for inspections, cleaning, or repairs, recommend which materials or coatings will last longest in a specific environment, and help owners and engineers understand the risks and make informed choices about maintenance and upgrades. The information is presented in clear, user-friendly dashboards, with options for virtual inspections or training using augmented or virtual reality.
Using a digital twin framework, infrastructure owners and engineers can move from simply reacting to problems as they arise, to anticipating and preventing them. This approach leads to safer, longer-lasting structures, lower maintenance costs, and better use of resources. It also helps ensure that critical infrastructure remains reliable even as climate conditions change, supporting both sustainability and public safety.
(continued on next page)
Fig. 3. Schematic representation of complex digital twin concept for predictive maintenance and materials selection facing climate change.
Zheludkevich and Jacob
(continued from previous page)
Effective risk analysis is essential to bridge predictive modeling and sound decision-making for climate-driven corrosion. Uncertainty arises from variable climate inputs, model limitations, and measurement errors. The suggested framework will aim at quantifying these uncertainties using ensemble climate projections, probabilistic corrosion models, and reliability methods (e.g., Monte Carlo, Bayesian networks). Uncertainty can be reduced through targeted monitoring, value-of-information analysis, and adaptive experiments. Accelerated testing and field validation are the ways to calibrate models, while robust optimization can ensure that maintenance and materials choices remain effective across a range of scenarios. Risk dashboards can visualize probabilities and confidence bands, linking thresholds to actionable plans. Risk-based maintenance scheduling can prioritize interventions by asset criticality and exposure, adapting as new evidence emerges.
A digital twin approach will enhance sustainability by extending asset life, reducing material and energy use, and minimizing waste from unnecessary maintenance. Materials selection will be optimized for local corrosivity, favoring durable, low-impact alloys and coatings. Smart polymers and self-healing technologies can be deployed where monitoring shows clear benefit. Digital twins can support circularity by documenting materials and conditions, aiding selective deconstruction and recycling. The suggested approach can also contribute to further development of policies and standards to require climate-resilient design, with digital twins providing transparent evidence for compliance and funding. The result will be more resilient infrastructure, predictable costs, and greater public safety and trust.
Acknowledgements
The authors used Copilot (Microsoft, USA) to assist with language editing and phrasing. All ideas, interpretations, and the final text were developed and verified by the authors.
© The Electrochemical Society. DOI:10.1149/2.F09261IF
About the Authors

Mikhail Zheludkevich, Director, Institute of Surface Science at Helmholtz-Zentrum Hereon and Full Professor, University of Kiel Education: PhD in Physical Chemistry (Belarusian State University) where he specialized in the interaction of atomic gases with metallic surfaces
Brief Work Experience: After completing his PhD, Prof. Zheludkevich spent a decade at the University of Aveiro in Portugal, progressing from postdoctoral researcher to group leader. In 2014 he joined Helmholtz-Zentrum Geesthacht (now Hereon) as Head of the Corrosion and Surface Technology Department. Continuing to expand the field, he established the Institute of Surface Science in 2021, which he has led since its founding, while simultaneously holding a full professorship at the University of Kiel. Research Interests: His research interests include electrochemistry, multi-functional and smart surfaces, and the active protection of lightweight materials and multi-material systems, with a focus on corrosion processes and advanced protective strategies.
Publications + Patents: He has authored over 400 SCI scientific papers with an h-index of 89 (Scopus 01.2026); delivered more than 60 invited, keynote, and plenary talks; and edited two books. Awards: His scientific achievements have been recognized with major distinctions, including the Tajima Prize from the International Society of Electrochemistry in 2016, the Cavallaro Medal from the European Federation of Corrosion in 2024, and the Person of the Year 2023 and Special Contribution to Innovations Award 2025 from the International Magnesium Society.
Website: https://www.hereon.de/institutes/surface_science/ team/098938/index.php.en https://orcid.org/0000-0002-9658-9619



Daniela Jacob, Climate Service Center Germany (GERICS), Helmholtz-Zentrum Hereon GmbH
Education: Prof Dr Daniela Jacob studied meteorology in Darmstadt and obtained her doctorate in Hamburg. Since 2015, Daniela Jacob has been Director of the Climate Service Center Germany (GERICS), an institution of the Helmholtz-Zentrum Hereon. She is also honorary professor at the Leuphana University of Lüneburg.
Brief Work experience: Before joining the Climate Service Center, she worked at the Max-Planck-Institute for Meteorology, Hamburg (1993–2015) and as Adjunct Professor for Regional Climate Change, University of Bergen, Norway (Professor II, Secondary employment, 2009–2013).
Research Interests: Her research focuses on regional climate modeling, climate services and adaptation to climate change, climate resilient development, and sustainability, as well as scientific support for the societal transformation toward a sustainable and climate-resilient 1.5°C lifestyle.
Publications + Patents: With an h-index of 66 (Scopus 01/2026) and through more than 300 publications, including her contribution as coordinating lead author of the IPCC Special Report on the impacts of global warming of 1.5 °C above pre-industrial levels and as one of the lead authors of the IPCC Fifth Assessment Report (WG 2), she has received high recognition worldwide. Until 2024, She was editor-in-chief of the journal Climate Services, a scientific journal she co-founded with Elsevier.
Awards: Her scientific achievements have been recognized by her participation in numerous national and international committees, such as the Climate Council of the Free and Hanseatic City of Hamburg, the German Committee for Sustainability Research in Future Earth (DKN), WPN2030, European Commission’s Mission Board for “Adaptation to Climate Change including Societal Transformation,” the WCRP-CORDEX Science Advisory Team, the Earth League, and the Scientific Advisory Board of the European Destination Earth Initiative. From 2021 to 2023 she was an ECMWF Fellow. In 2023, she was elected as Fellow of the International Science Council (ISC). In 2022 she received the German Renewables Award, category lifetime achievements, from the Cluster Erneuerbare Energien Hamburg. Website: https://www.gerics.de/index.php.en https://orcid.org/0000-0002-5249-4044


References
1. G. H. Koch, M. P. H. Brongers, N. G. Thompson, Y. P. Virmani, and J. H. Payer, Corrosion Costs and Preventive Strategies in the United States. NACE International (2016).
2. Y. Zhang, B. Ayyub, and J. Fung, Resilient Cities and Structures, 1, 98 (2022).
3. M. G. Stewart, X. Wang, and M. N. Nguyen, Eng Struct, 33(4), 1326 (2011).
4. M. F. Tamimi , A. A. Alshannaq, and M. I. AbuQamar, Corros Eng, Sci Technol, 59, 273 (2024).
5. M. L. Sousa, S. Dimova, A. Athanasopoulou, G. Rianna, P. Mercogliano, et al.. Expected implications of climate change on the corrosion of structures. Joint Research Centre, hal-04544233 (2020).
6. S. Pfeifer, K. Bülow, A. Gobiet, A. Hänsler, M. Mudelsee, et al., Atmosphere, 6, 677 (2015).
7. D. Jacob, C. Teichmann, S. Sobolowski, E. Katragkou, I. Anders, et al., Reg Environ Change, 20, 2 (2020).
8. L. B. Coelho, D. Zhang, Y. Van Ingelgem, D. Steckelmacher, A. Nowé, et al., npj Mater Degrad, 6, 8 (2022).
9. A. Hagen and T. M. Andersen, Struct Infrastruct Eng, 20, 1242 (2024).
10. F. Mansfeld, Werkstoffe und Korrosion, 30, 38 (1979).
11. I.S. Cole, T.H. Muster, N.S. Azmat, M.S. Venkatraman, and A. Cook, Electrochim Acta, 56, 1856 (2011).
12. N. S. Trivedi, M. S. Venkatraman, C. Chu, and I. S. Cole, Clim Change, 124, 133 (2014).
13. Z. Bai and Y. Yan, Buildings, 15, 2271 (2025).
14. S. Li, C. Li, and F. Wang, Mater Today Chem, 37, 101986 (2024).
15. D. Xu, Z. Pei, X. Yang, Q. Li, F. Zhang, et al., Materials, 16, 3396 (2023).
16. Hamidreza Habibollahi Najaf Abadi, Physics-Informed Deep Learning-Based Approach for Probabilistic Modeling of Degradation. Annual Conference of the PHM Society, 15(1), (2023).
17. T. Choi, and D. Lee, Materials, 16, 5326 (2023).
18. D. A. Najera-Flores, G. Qian, Z. Hu, and M. D. Todd, Mech Sys Signal Process, 200, 110515 (2023).
19. Z. Xu, X. Li, B. Cai, G. Liu, L. Yan, et al., Prog Nat Sci: Mater Int, 35, 701 (2025).
20. P. Bansal, Z. Zheng, C. Shao, J. Li, M. Banu, et al., Reliab Eng Syst Saf, 227, 108711 (2022).
21. Hamidreza Habibollahi Najaf Abadi, Dissertation: Physicsinformed deep learning framework for probabilistic modeling of environmentally induced degradation (2024).
22. F. Peltier, and D. Thierry, Corros Mater Degrad, 5, 427 (2024).
23. K. Pélissier, N. Le Bozec, D. Thierry, and N. Larché, Coatings, 12, 1758 (2022).
24. K. Popova and T. Prošek, Mater Corros, 76, 979 (2025).
25. V. Vangrunderbeek, L. B. Coelho, D. Zhang, Y. Li, Y. Van Ingelgem, et al., Corros Sci, 225, 111619 (2023).
26. M. Komary, S. Komarizadehasl, N. Tošic, I. Segura, J. A. Lozano-Galant, et al., Sensors, 23, 1309 (2023).



ECS Europe Section
As of January 1, 2026, the ECS Europe Section welcomed a new Executive Committee. Prof. Ingrid Milošev (Jožef Stefan Institute) has assumed the role of Chair, with Prof. Thierry Djenizian (École nationale supérieure des mines de Saint-Étienne) as Vice Chair. Dr. Iwona Rutkowska (Uniwersytet Warszawski) serves as Secretary, and Dr. Andrea Bourke (Technological University of the Shannon) as Treasurer. Prof. Jan Macák (Univerzita Pardubice) continues his involvement as Immediate Past Chair. The Executive Committee is further supported by Members at Large: Profs. Philippe Marcus, Paweł Kulesza, Noel Buckley, Petr Vanýsek, Giuseppe Barillaro, Geir Martin Haarberg, Stefan de Gendt, Uroš Cvelbar, and Dr. Robert Lynch
The ECS Europe Section sincerely thanks the previous Executive Committee, led by Chair Prof. Jan Macák with Dr. Adriana Ispas

Chair ECS Europe Section

Vice Chair ECS Europe Section
as Secretary and Dr. Robert Lynch as Treasurer, for their dedication and service.
Today, the section comprises more than 600 members. Founded to advance science, technology, and human understanding, it is committed to encouraging young scientists and fostering the exchange of ideas between fundamental and applied research across Europe. The section supports and promotes activities in electrochemistry and solid state science, provides networking opportunities for researchers at all career stages, and works to strengthen collaboration among and the visibility of European ECS members within the Society and internationally. The new Executive Committee is dedicated to fostering cooperation, welcoming new ideas, and supporting the organization of events that promote electrochemical science and technology across Europe and beyond.

Secretary





Ingrid Milošev
Thierry Djenizian
Iwona Rutkowska
ECS Europe Section
Andrea Bourke Treasurer ECS Europe Section
Section Leadership
Connect with Local Scientists and Engineers
ECS Sections introduce and support activities in electrochemistry and solid state science within specific regions and bring technical news and activities to those not able to attend ECS meetings. Sections participate in overall ECS affairs, work to increase ECS membership, and help create awareness for science. Getting involved with a section
is an excellent networking opportunity for those new to the field or advanced in their careers!
For more information on your region’s section, go to https://www. electrochem.org/sections. For more information on joining, contact ECS Section & Chapter Engagement Specialist Maggie Hohenadel
Section Name
Section Chair
Arizona Section Candace K. Chan
Brazil Section Raphael Nagao
Canada Section Christian Kuss
Chile Section José H. Zagal
China Section Open
Detroit Section Tobias Glossmann
Europe Section Ingrid Milošev
Georgia Section Faisal Alamgir
India Section Sinthai Ilangovan
Israel Section Eran Edri
Japan Section Seiichiro Higashi
Korea Section Won-Sub Yoon
Mexico Section Norberto Casillas Santana
Mid-America Section Ahmed Farghaly
National Capital Section Alexander Zestos
New England Section Joshua Gallaway
Pacific Northwest Section April Li
Pittsburgh Section Open
San Francisco Section Xiong Peng
Singapore Section Qingyu Yan
Taiwan Section Chi-Chang Hu
Texas Section Yan Yao
Thailand Section Soorathep Kheawhom
Twin Cities Section Lifeng Dong
Learn more about ECS sections at www.electrochem.org/sections.

ECS Awards, Fellowships, and Grants
The ECS Honors & Awards Program celebrates exceptional technical achievement in electrochemical and solid state science and technology, and acknowledges exceptional service to the Society. Awards opportunities are provided in the categories of Society Awards, Division Awards, Section Awards, and Student Awards. As today’s emerging scientists will drive our field’s future, ECS offers competitive fellowships and grants supporting students and early-career professionals pursuing new discoveries and advancing science for years to come.
The nomination period for all awards is March 15–June 15, 2026, unless otherwise indicated.
Society Awards
Allen J. Bard Award (est. 2013) recognizes distinguished contributions to electrochemical science with a plaque; $7,500*; complimentary meeting registration for the award recipient and a companion; dinner in the recipient’s honor; and ECS Life Membership.
Nominate a Candidate*
Gordon E. Moore Medal for Outstanding Achievement in Solid State Science and Technology (est. 1971) acknowledges distinguished contributions to the field of solid state science and technology with a silver medal; plaque; $7,500; complimentary meeting registration for the award recipient and a companion; dinner in the recipient’s honor; and ECS Life Membership.
Nominate a Candidate
John B. Goodenough Award (est. 2022) recognizes distinguished contributions to fundamental and technological aspects of electrochemical materials science and engineering (especially in batteries, solid ion conductors, fuel cells, transition-metal oxides, and/or magnetic materials) with a gold medal; plaque; $10,000; complimentary meeting registration for the award recipient and a companion; dinner in the recipient’s honor; and ECS Life Membership.
Nominate a Candidate
Leadership Circle Awards (est. 2002) honor and thank our electrochemistry and solid state science institutional partners when they reach milestone levels, with commemorative plaques and ECS website and ECS Interface recognition.
Nominations not accepted
Division Awards
Battery Division M. Stanley Whittingham Mid-Career Award (est. 2024) honors M. Stanley Whittingham (long-term ECS member and Nobel laureate) and recognizes mid-career achievements and contributions to electrochemical energy storage. Awardees receive a certificate and $3,000.
Nominate a Candidate
Dielectric Science & Technology Early-Career Award (est. 2025) encourages excellence by early-career scientists in dielectric investigations, the preparation of high-quality science and technology papers and patents, and publication in ECS journals. Awardees receive a certificate and $1,000.
Nominate a Candidate
Dielectric Science and Technology Division Thomas D. Callinan Award (est. 1967) supports excellence in dielectric investigations, the preparation of high-quality science and technology papers and patents, and publication in ECS journals Awardees receive a certificate and $2,000.
Nominate a Candidate
Electronics and Photonics Division Award (est. 1969) encourages excellence in electronics research and outstanding technical contributions by recognizing authors whose impactful papers advance electronics science and enhance ECS’s stature. The award includes a certificate; $1,500; and ECS Life Membership or up to $1,000 in travel support.
Nominate a Candidate
Energy Technology Division Research Award (est. 1992) recognizes outstanding, original contributions to energy-related science and technology within the Division’s technical interest areas and encourages excellence in energy research and publication in JES The award includes a certificate; $2,000; and ETD membership while the recipient maintains ECS membership.
Nominate a Candidate
Energy Technology Division Supramaniam Srinivasan EarlyCareer Investigator Award (est. 2011) recognizes and rewards outstanding early-career researchers in the energy technology field and includes a certificate; $1,000; and complimentary meeting registration.
Nominate a Candidate
Industrial Electrochemistry and Electrochemical Engineering Division New Electrochemical Technology (NET) Award est. 1997) is endowed by the Dow Company Foundation. The award recognizes significant advances in industrial electrochemistry and promotes high-quality applied electrochemical research and development. The recipient organization receives a commemorative plaque and team members receive certificates and IE&EE Division Business Luncheon tickets. The winner presents a paper on its technology development at a Division-sponsored symposium.
Nominate a Candidate
Nanocarbons Division Robert C. Haddon Research Award (est. 2006) encourages excellence in fullerenes, nanotubes, and carbon nanostructures research, and recognizes researchers making outstanding contributions to the understanding and applications of fullerenes. Awardees receive a certificate; $1,000; and up to $1,500 in travel assistance.
Nominate a Candidate
Fellowships
ECS Toyota Young Investigator Fellowships (est. 2015 in partnership with the Toyota Research Institute of North America) encourage young professionals and scholars to pursue research into innovative electrochemical research in green energy technology (batteries, fuel cells and hydrogen, and future sustainable technologies). Fellows receive $50,000 to conduct their proposed research within one year; complimentary oneyear ECS membership; and the opportunity to present their research at a Society meeting and publish in an ECS journal.
Materials deadline: April 30, annually
Colin Garfield Fink Fellowship (est. 1962) provides postdoctoral scientists/researchers with $5,000 to pursue research from June through September in a field of interest to the Society, and the opportunity to publish a report in ECS Interface
Materials deadline: January 15, annually
ECS Summer Fellowships (est. 1928) provide students with $5,000 to pursue research from June through August in a field of interest to ECS and to publish a report in ECS Interface. Support is through the Edward G. Weston Fellowship, Joseph W. Richards Fellowship, F. M. Becket Fellowship, and H. H. Uhlig Fellowship.
Materials deadline: January 15, annually
Section Awards
Mid-America Section Ernest B. Yeager Electrochemistry Award acknowledges significant contributions to the advancement of electrochemistry in Indiana, Illinois, Ohio, and Kentucky. Awardees receive a certificate and $1,000.
Nominate a Candidate

Pacific Northwest Section Electrochemistry Research Award Sponsored by Gamry Instruments (est. 2021) recognizes excellence in electrochemistry and solid state science and technology research by independent scientists or engineers working in Washington, Oregon, or Idaho, and includes a certificate and $1,000.
Nominate a Candidate
Student Awards
ECS General Student Poster Session Awards (est. 1993) recognize outstanding research of general interest to the Society and aim to promote graduate and undergraduate work in electrochemical and solid state science and technology while encouraging active student interest and participation in ECS. Posters accepted for presentation are eligible for awards of $1,500 (1st place); $1,000 (2nd place); and $500 (3rd place). Eligibility requires submission and acceptance of an abstract into the Z01—General Student Poster Session, upload of a digital poster, and the author’s in-person presence during live judging.
Materials are due by the relevant ECS Meeting abstract deadline (250th ECS Meeting abstract submission deadline: March 27, 2026)
ECS Outstanding Student Chapter Award (est. 2012) acknowledges student chapters that actively engage in Society technical activities, conduct electrochemical and solid state science and engineering education and outreach, and build strong membership. The Outstanding Student Chapter receives a plaque; certificates; $1,000; and ECS Interface and electronic communications recognition. Up to two Chapters of Excellence are also awarded.
Materials deadline: April 15, 2026
Energy Technology Division Graduate Student Award Sponsored by BioLogic (est. 2012) acknowledges promising young engineers and scientists in fields pertaining to the division, and consists of a certificate; $1,000; complimentary meeting registration; and division business meeting admission.
Nominate a Candidate
Industrial Electrochemistry and Electrochemical Engineering Division Student Achievement Awards (est. 1989) recognize promising young engineers and scientists in electrochemical engineering. Recipients receive certificates; $1,000; one year of ECS membership; and complimentary IE&EE Division Business Luncheon tickets. Two winners may be selected, one of whom receives the H. H. Dow Memorial Student Award (est.1990).
Nominate a Candidate
Industrial Electrochemistry and Electrochemical Engineering Division Ralph E. White Outstanding Student Award (est. 2022) honors promising young engineers and scientists researching modeling and simulation of electrochemical systems. Recipients receive a certificate and $1,000 and are encouraged to continue careers in electrochemical engineering or applied electrochemistry.
Nominate a Candidate
Travel Grants
Biannual Meeting Travel Grants, offered by many ECS divisions and sections to undergraduates, graduate students, postdoctoral researchers, and young professionals and faculty presenting ECS biannual meeting papers, range from complimentary meeting registration to luncheon/reception tickets, travel expenses, and more. Application requirements vary by Division and section.
250th ECS Meeting Travel Grant application period: March 27–July 6, 2026
*Award amounts are in US dollars.
**The nomination period for all awards is March 15–June 15, 2026, unless otherwise indicated.
Society, Division, and Section Award Winners
Join us in congratulating ECS community members recognized for outstanding technical achievements and contributions to electrochemical and solid state science and technology through the ECS Honors & Awards Program.
Society Awards
Henry B. Linford Award for Distinguished Teaching

Robert G. Kelly is the Associate Chair for Operations, Thomas Goodwin Digges Professor of Materials Science, Professor of Materials Science and Engineering, and CoDirector of the Center for Electrochemical Science and Engineering at the University of Virginia (UVA). For 40 years, he has conducted research on the corrosion of metals. In the past, he worked on the corrosion of metals and alloys in marine environments, non-aqueous and mixed solvents, as well as stresscorrosion cracking and localized corrosion. Now he studies conditions inside localized corrosion sites in various alloy systems, corrosion of additive manufacturing (AM) materials, and modeling of corrosion processes.
After completing his PhD at Johns Hopkins University in 1989, Prof. Kelly was a Fulbright Scholar and NSF/NATO Postdoctoral Fellow at the University of Manchester Corrosion and Protection Centre. He joined the UVA faculty in 1990. The coauthor of more than 150 papers, he has presented 100 invited talks. A Fellow of The Electrochemical Society and a National Association of Corrosion Engineers (NACE) International Fellow, Prof. Kelly received the 2021 NACE W. R. Whitney Award, 2016 ECS Corrosion Division H. H. Uhlig Award, 1999 NACE H. H. Uhlig Award, and 1997 NACE A. B. Campbell Award. UVA has honored Prof. Kelly with teaching awards that include the 2024 Research Collaboration Award. He has rendered technical assistance to the US Nuclear Regulatory Commission and Department of Energy concerning the Yucca Mountain Project, USAF Aging Aircraft Program, NASA Safety and Engineering Center, and 9/11 Pentagon Memorial design team.
Prof. Kelly joined ECS in 1984. His service to the Society includes Editor-in-Chief of ECS Interface since 2018, ECS Corrosion Division Chair (2004–2006), and multiple ECS Corrosion Division and Society committee memberships.
Vittorio de Nora Award

Thomas Zawodzinski holds the University of Tennessee-Knoxville (UT)-Oak Ridge National Laboratory (ORNL) Governor’s Chair in Electrical Energy Conversion and Storage, with dual appointments in UT’s Department of Chemical and Biomolecular Engineering and ORNL’s Division of Materials Science and Technology. He has made significant contributions to electrocatalysis, gas diffusion media, and fuel cell durability, as well as to research on redox flow and metal-air batteries, electrochemical sensors, electro-responsive polymers, electroseparations, and related electrochemical technologies. His work most frequently focused on ionic systems, particularly polymer electrolytes, for which he developed multiple innovative analytical approaches. Throughout his career, he collaborated extensively with industry partners, translating fundamental science into practical technologies.
A current emphasis in Prof. Zawodzinski’s lab is adapting fuel cell advances to a range of other “open” electrochemical systems relevant to an electron economy powered by renewable electricity. Through an ARPA-E project, his team achieved a tenfold reduction in the amount of platinum needed to run a fuel cell system, and the technology was licensed in 2016 by the University of Tennessee Research Foundation (UTRF).
After completing his PhD at The State University of New York (SUNY) at Buffalo under the guidance of Profs. Robert Osteryoung and Robert Kurland, Prof. Zawodzinski spent 13 years at Los Alamos National Laboratory researching polymer electrolyte fuel cells. From there, he went to Case Western Reserve University, where he served as the F. Alex Nason Professor of Engineering, Director of the Case Advanced Power Institute, and Ohio Eminent Scholar in Fuel Cells. He was the founding Director of the Wright Fuel Cell Group. He joined UT in September 2009.
Prof. Zawodzinski received the 2016 ECS Energy Technology Division Research Award and is a Fellow of The Electrochemical Society and the American Chemical Society Polymer Division. He has published more than 200 refereed papers and several book chapters, and holds a dozen patents. He joined the Society in 2001.
Division Awards
Battery Division M. Stanley Whittingham Mid-Career Award

Yi-Chun Lu is Professor at The Chinese University of Hong Kong (CUHK). Her research focuses on advancing clean energy storage and conversion through fundamental insights and innovative material design. Her group investigates electrode and electrolyte design for high-energy metal-air and metalsulfur batteries, redox-active components and solution chemistry for redox-flow batteries, high-voltage aqueous battery development, and mechanistic studies of interfacial phenomena in electrochemical energy systems. Prof. Lu is the co-Founder and Chief Scientist of Luquos Energy Limited, a company dedicated to commercializing polysulfide flow battery technology for longduration energy storage.
In 2012, Prof. Lu earned a PhD in Materials Science & Engineering from the Massachusetts Institute of Technology, then conducted postdoctoral research at the Technische Universität München in 2013. She joined CUHK in 2013 as an assistant professor, receiving promotion to Associate Professor in 2018 and to Professor in 2022.
Prof. Lu is a Fellow of the Royal Society of Chemistry and Founding Member of the Young Academy of Science of Hong Kong. Among the recognition and awards she has received are the 2024 ISE Tajima Prize, 2023 Hong Kong Engineering Science and Technology Award, 2021 Xplorer Prize, 2021 IBA Early Career Award, and 2020 Top 10 Falling Walls Science Breakthroughs of the Year. She serves as an Associate Editor and Board Member for Journal of Materials Chemistry A and as an Editorial Board/Advisory Member for Energy & Environmental Science, Materials Today, and Chemistry of Materials. Prof. Lu joined ECS in 2010.
Photo:
Tom Daly
Dielectric Science & Technology Division
Thomas D. Callinan Award

Mahendra Sunkara is the Founding Director of the Conn Center for Renewable Energy Research, Professor of Chemical Engineering, and University Scholar at the University of Louisville (UofL). His research spans the development of plasma-catalytic processes for the synthesis of nanowire materials, energy conversion and storage technologies, and advanced plasma catalysis for chemical production.
Prof. Sunkara received his PhD in Chemical Engineering from Case Western Reserve University in 1993. He has published more than 175 peer-reviewed journal articles and four book chapters, and holds more than 35 US patents. He is coauthor of the book Inorganic Nanowires: Applications, Properties, and Characterization. His work has been cited more than 14,800 times with an h-index of 57. Under his mentorship, Prof. Sunkara has graduated more than 20 MS/ MEng and 30 PhD students, many of whom have successful careers in academia—serving as faculty at institutions such as RPI, Texas A&M, University of Arkansas, IIT Hyderabad, and Oklahoma State University—and in industry at companies that include Intel, HP, Lam Research, Veeco, Applied Materials, and Lexmark. Since founding the Conn Center for Renewable Energy Research in 2009, Prof. Sunkara has built it into a thriving hub with a dedicated full-time research staff and over 30,000 square feet of state-of-the-art laboratory space. He currently leads the Regional Energy Business Education Commercialization Convergence Accelerator (REBECCA)—a consortium of industry, educational institutions, and government agencies fostering an innovation ecosystem across Kentucky and Southern Indiana. In addition to his academic leadership, Prof. Sunkara founded ADEM Technologies, Inc. (formerly Advanced Energy Materials, LLC) in 2011. The company has successfully scaled up nanowire production for use in catalysts and sorbents targeting desulfurization, carbon capture, and reforming applications.
Among the awards Prof. Sunkara has received are, in 2009, the UofL President’s Distinguished Faculty Award for Research and the Indian Institute of Chemical Engineers United Phosphorus CDS Award, and, in 1999, the Ralph E. Powe Junior Faculty in Engineering Award and the National Science Foundation CAREER grant. He joined ECS in 2004.
Electronics and Photonics Division Award

Fred Roozeboom is Guest (Emeritus) Professor at the Universiteit Twente (UT). His research interests are rooted in selective atomic layer deposition (ALD), atomic layer etching (ALE), Li-ion batteries, the longevity of extreme ultraviolet (EUV) optics, and CO2 capture on active coal.
Prof. Roozeboom holds a PhD in Technical Sciences from UT with a specialization in inorganic chemistry and catalysis. After working in catalysis for three years at ExxonMobil R&D Labs, he joined Philips Research (known from 2007 as NXP) in 1983. His research there concentrated on thin-film technology and plasma processing. From 1997 to 2009, he led a team that focused on applications in 3D passive and heterogeneous integration for system-in-package devices for wireless communication and power management. He was a Research Fellow and full Professor at Technische Universiteit Eindhoven from 2007 to 2021, working on atomic layer deposition—including area-selective—and etching. From 2009 to 2021 he specialized in spatial atomic layer process and
reactor design for photovoltaics and displays at TNO Holst Centre. In 2021, he joined UT. His high-tech industry consultancies since 2021 have included applications of thin-film processing for atomic layer etching–based patterning, extending the lifetime of EUV optics, 3D Li-ion batteries, CO2 capture, and reduction of greenhouse gas emission.
Prof. Roozeboom has published more than 200 papers in journals (h-index of 44) and holds more than 50 granted and pending patents. He is a Fellow of The Electrochemical Society and of AVS. Prof. Roozeboom joined ECS in 1991 and received the 2023 Gordon E. Moore Medal for Outstanding Achievement in Solid State Science and Technology. Over the past 30 years, he has organized many conferences and workshops on thin-film processing, most recently as Program Chair of the 11th International Atomic Layer Etching Workshop (ALE 2024), part of ALD2024
Energy Technology Division Research Award

Minhua Shao is the Cheong Ying Chan Professor of Energy Engineering and Environment, and Head and Chair Professor in the Department of Chemical and Biological Engineering at the Hong Kong University of Science and Technology (HKUST). He is also the Director of the HKUST Energy Institute and Director of the Joint Laboratory for Hydrogen Energy.
After completing his PhD in Materials Science and Engineering at the State University of New York at Stony Brook in 2006, Prof. Shao joined UTC Power in 2007. There he led the development of advanced electrocatalyts for fuel cells and was promoted to UTC Technical Fellow in 2012. He conducted research on lithium-ion batteries at the Ford Motor Company in 2013, then joined HKUST in 2014.
His research has been recognized with numerous awards that include the 2022 International Outstanding Young Chemical Engineer Award and 2014 ECS Energy Technology Division Supramaniam Srinivasan Young Investigator Award. He is a founding member of The Hong Kong Young Academy of Sciences and is author of more than 350 peer-reviewed papers.
Prof. Shao joined ECS in 2005; he is a Fellow of The Electrochemical Society, currently chairs the Energy Technology Division, and serves as a Technical Editor for the Journal of The Electrochemical Society
Energy Technology Division Supramaniam Srinivasan Early-Career Investigator Award

Antoni Forner-Cuenca is Full Professor at the Technische Universiteit Eindhoven (TU/e), where he founded and leads the Electrochemical Materials and Systems Group. His research focuses on advancing transformative electrochemical technologies for real-world energy applications. By leveraging principles from (electro)chemical engineering, materials science, and physical chemistry, his team designs, synthesizes, characterizes, and models innovative materials and electrochemical reactors. Key applications include large-scale energy storage with flow batteries, energy conversion via hydrogen fuel cells and electrolyzers, and decarbonization of the chemical industry through efficient molecular synthesis and separation processes.
Prof. Forner-Cuenca earned his PhD at the Eidgenössische Technische Hochschule (ETH) Zürich Paul Scherrer Institut in 2016.
(continued on next page)
Photo: Ashly Cecil
Photo: Princess of Girona Foundation
(continued from previous page)
AWARDS PROGRAM AWARDS PROGRAM
His pioneering work on gas diffusion layers with patterned wettability for polymer electrolyte fuel cells received the ETH Zürich Medal for Outstanding Thesis. As a Postdoctoral Fellow at the Massachusetts Institute of Technology from 2017 to 2018, he advanced the science and engineering of redox flow batteries for large-scale energy storage. Prof. Forner-Cuenca joined the TU/e faculty in 2019 and became Full Professor in 2025.
His research has earned numerous honors, including the 2025 Princess of Girona Award, 2024 Royal Netherlands Academy of Arts and Sciences Early Career Award, 2024 Dutch Research Council (NWO) Vidi Grant, 2022 European Research Council Starting Grant, 2021 Hydrogen Europe Young Scientist Award, 2019 Dutch Research Council Division of Applied & Engineering Sciences Veni Grant, and 2017 ECS Energy Technology Division Graduate Student Award Sponsored by BioLogic. The TU/e Master’s degree program in the Department of Chemical Engineering and Chemistry voted him Best Teacher in 2022 and 2023. He joined ECS in 2015.
Nanocarbons Division
Richard E. Smalley Research Award

Jeffrey Blackburn is a Senior Research Fellow at the National Renewable Energy Laboratory (NREL). He is the lead investigator of NREL’s BES Solar Photochemistry Program and a Principal Investigator in two DOE Energy Frontier Research Centers (EFRCs)—the Center for Hybrid Organic/Inorganic Semiconductors for Energy (CHOISE) and Reconfigurable Electronic Materials Inspired by Nonlinear Neuron Dynamics (reMIND). His research on emergent properties and dynamic processes in low-dimensional materials for energy conversion and storage focuses on the transfer, transport, and transduction of charges, excitons, and spins. His research interests include solar photoconversion, energy-efficient information processing, microelectronic devices, thermoelectric energy conversion, and (photo)catalytic processes.
Dr. Blackburn received a PhD in Chemistry from Colorado University, Boulder under the guidance of Prof. Art Nozik, and completed postdoctoral research at NREL with Dr. Mike Heben. A member of ECS for almost twenty years, he was named Fellow of The Electrochemical Society in 2023 and currently serves as Chair of the ECS Nanocarbons Division.
Nanocarbons Division
SES Research Young Investigator Award

Kyu-Young Park is Associate Professor with joint appointments in the Department of Battery Engineering and Department of Materials Science and Engineering at Pohang University of Science and Technology (POSTECH). His research focuses on developing next-generation batteries. He is interested in cathode materials and strives to realize high-capacity, high-safety, and highenergy-density chemistries. Specializing in electrochemical energy storage materials and their translation from lab discovery to manufacturable technologies, his research integrates quantum-mechanical modeling (DFT) with multimodal structural probes—X-ray absorption, X-ray and neutron diffraction, and imaging—to reveal electron/ion/mass-transfer bottlenecks that govern battery performance and reliability. Recognizing that practical
performance is decided at the electrode scale, Park focuses on carbon. His group endows conventional conductive agents such as CNTs and graphene with additional functionalities beyond electron transport. These include suppressing oxygen release in cathodes, absorbing chemo-mechanical deformation energy to preserve percolation, and mediating heat dissipation to improve thermal stability. By treating carbon as an active component rather than a passive additive, the team creates pathways toward state-of-the-art battery technologies. Building on these insights, Park’s team translates design rules into outcomes: ultra-high-energy-density thick electrodes, long-life highnickel cathodes, and thermally stable electrodes with engineered carbon–binder domains and robust electrolyte wetting.
Prof. Park received his PhD in 2016 from the Seoul National University School of Engineering, followed by postdoctoral fellowships at Seoul National University from 2016 to 2018 and at Northwestern University with Prof. Mark C. Hersam from 2018 to 2021. His 80 peer-reviewed papers are published in journals that include Advanced Materials, Advanced Energy Materials, and ACS Nano. His awarded honors include the 2017 Korean Synchrotron Radiation Users Association Young Scientist Award and the 2017 Korean Conference on Neutron Scattering Best Researcher Award. His group collaborates with academia and industry to couple operando diagnostics with manufacturing–centric process design. He joined ECS in 2023. Outside the lab, he enjoys walks, values conversations with students, and hopes to hasten society’s move toward a carbonneutral future.
Section Awards
PNW Section Electrochemistry Research Award Sponsored by Gamry Instruments

Xiulei (David) Ji is the Bert and Emelyn Christensen Professor of Chemistry at Oregon State University. His research focuses on developing new charge-storage chemistries for aqueous batteries and alkalimetal-ion batteries.
Prof. Ji earned his PhD in Materials Chemistry from the University of Waterloo in 2009. From 2010 to 2012, he was an NSERC (Natural Sciences and Engineering Research Council of Canada) Postdoctoral Fellow at the University of California, Santa Barbara. Prof. Ji has been a Clarivate Highly Cited Researcher every year since 2019. A Fellow of the Royal Society of Chemistry, he received NSF CAREER Awards from 2016 to 2021, and a 2017 Research Corporation for Science Advancement Scialog Fellowship. He is a founding Associate Editor of Carbon Energy. Dr. Ji joined ECS in 2019.

Wu Xu is Chief Scientist in the Electrochemical Materials and Systems Group in the Energy Processes and Materials Division under the Energy and Environment Directorate at Pacific Northwest National Laboratory (PNNL). His research includes developing novel electrolytes and electrodes materials and investigating electrode/ electrolyte interphases for various advanced energy storage systems, including lithium batteries (with intercalation cathodes and conversion cathodes), sodium batteries, zinc batteries, and organic redox flow batteries.
Dr. Xu completed his PhD in Chemistry at the National University of Singapore in 2000. Before joining PNNL in 2008, he worked as a Faculty Research Associate in the Department of Chemistry and Biochemistry at Arizona State University and as a Senior Scientist in
Photo: Andrea Starr-PNNL
the Technical Center of Ferro Corporation. Dr. Xu has more than 250 peer-reviewed publications and 39 granted US patents. He received the 2023 DOE EERE Vehicle Technologies Office Distinguished Achievement Award, 2020 Battelle Distinguished Inventor Award, 2013 PNNL Inventor-of-the-Year Award, and the 2012 R&D 100 Award. Clarivate listed Dr. Xu as a Highly Cited Researcher in 2018 and every year from 2020 through 2025. He joined ECS in 1999.
Student Awards
Energy Technology Division Graduate Student Award Sponsored by Bio-Logic

Siddharth Rajupet is a fifth-year PhD student at the University of California, Berkeley, working in the Energy Conversion Group with Dr. Adam Weber and Prof. Clay Radke. His thesis explores interactions between ion-conducting polymers (ionomers) and catalyst particles in inks deposited to form fuel-cell electrodes. His work investigates the driving forces for and factors limiting the extent of ionomer adsorption to catalyst particles, which governs the ionomer distribution and structure of the final fuel-cell electrode. His research combines fundamentals of colloid science and electrochemistry to address these questions.
Sidd earned his BS and MS in Chemical Engineering from Case Western Reserve University in 2021 where he worked with Prof. Daniel Lacks investigating forces governing the dynamics of fine particles adhered to surfaces, suspended in air, and dispersed in solution. He won an AIChE Student Poster Competition Award in 2019.

S. Avery Vigil is a fourth-year PhD candidate and NSF Graduate Research Fellow in the Department of Chemistry at Duke University, advised by Prof. Ivan A. Moreno-Hernandez. Avery’s doctoral work is centered on using in situ transmission electron microscopy to understand the nanoscale dynamics of electrocatalyst materials. They leveraged this technique to investigate the degradation of model nanocrystalline electrocatalysts for the oxygen evolution reaction, revealing complex dissolution phenomena. Avery’s research has led to the synthesizing of next-generation electrocatalyst materials for the oxygen evolution reaction. These materials are designed with a focus on reduced noble metal loadings and high stability at the device scale. Avery has collaborated closely with researchers at the University of Pennsylvania, Columbia University, Brookhaven National Laboratory, and Lawrence Livermore National Laboratory.
Avery earned a BS in Chemistry and BA in Linguistics from the University of Oklahoma in 2020, with a research focus on optoelectronic material synthesis for light emission and language preservation, respectively.
Outside of the lab, they serve the community through the Materials Research Society, Duke Materials Initiative, and Graduate Chemistry Council at Duke. They have published their work in the Journal of the American Chemical Society, Energy & Environmental Science, and Matter, among other journals.
Georgia Section Outstanding Student Award

Abiram Krishnan is a fifth-year PhD candidate in the School of Materials Science and Engineering at the Georgia Institute of Technology (Georgia Tech), advised by Dr. Faisal Alamgir. His research explores the role of anion redox in metal-oxide cathodes for lithium-ion batteries. In particular, he investigates how anions influence the stability and energy density of these materials.
Abiram serves as President of the ECS Georgia Institute of Technology Student Chapter, helping to organize lectures, poster competitions, and social events. Under his leadership, the chapter’s monthly Energy Seminar Series has hosted more than 12 talks by leading electrochemistry and spectroscopy researchers. He is a member of Georgia Tech’s Tech-to-Teaching program, which trains current and future faculty in different pedagogical approaches to teaching. In his free time, Abiram enjoys traveling, playing tennis, and listening to music.
Industrial Electrochemistry and Electrochemical Engineering Division
H. H. Dow Memorial Student Achievement Award

Simon Rufer is CEO & Co-Founder of CoFlo Medical and a PhD candidate in Mechanical Engineering at the Massachusetts Institute of Technology (MIT). His electrochemical research approach spans length scales and systems—from deploying novel micro-electrode architectures at the micro level, to developing new electrochemical cell schema at a macro level and establishing processes that more efficiently incorporate electrochemistry at a system level. Throughout, his work focuses on addressing mass-transport limitations which are often limiting factors to the industrial scalability of electrochemical processes, while using techno-economic analyses to contextualize these improvements. Simon has explored and optimized electrochemical approaches for environmental CO2 removal (batterybased ocean carbon dioxide removal and direct air capture), point source capture, electrochemical CO2 conversion to value-added chemicals, and hydrogen electrolysis. He is passionate about entrepreneurship and bringing technologies from the lab to the market.
Simon completed an MS in Mechanical Engineering at MIT in 2022 before pursuing his PhD as a Presidential Fellow and National Science Foundation Graduate Research Fellow in the Varanasi Research Group. CoFlo Medical, the company Simon founded with MIT Prof. Kripa Varanasi and Vishnu Jayaprakash, won the 2025 MIT $100,000 Entrepreneurship Competition. Simon received the 2025 Wunsch Foundation Silent Hoist and Crane Award and won the 2025 De Florez Competition.
A California native and appreciator of his state’s beautiful nature, Simon enjoys all things outdoors.
(continued on next page)
(continued from previous page)
AWARDS PROGRAM AWARDS PROGRAM
Industrial Electrochemistry and Electrochemical Engineering Division Student Achievement Award

Xintong Yuan recently completed her PhD in Chemical Engineering at the University of California, Los Angeles (UCLA), working with Prof. Yuzhang Li. Xintong worked on next-generation batteries, exploring the fundamentals of lithium-metal deposition and the formation of solid electrolyte interphase using cryo-EM. Specifically, Dr. Yuan’s research tackled a decades-old question in battery research: what is the intrinsic morphology of lithium deposits in the absence of a solid electrolyte interphase layer? She revealed it to be that of a nondendritic rhombic dodecahedron that is independent of electrolyte chemistry or substrate composition. Her unexpected findings, which transformed how the field thinks about lithium-metal electrodeposition, provide new insights for avoiding key failure modes during fast charging of lithium-metal batteries.
Dr. Yuan is a member of the 2025 class of next-generation researchers pursuing postdoctoral studies with support from the Schmidt Science Fellowship. In 2025, she received the ECS Battery Division Student Research Award, Materials Research Society Graduate Student Gold Award, and American Institute of Chemical Engineers Women in Chemical Engineering Travel Award. In 2023, Dr. Yuan received recognition as a Massachusetts Institute of Technology (MIT) Chemical Engineering Rising Star and Materials Science and Engineering Rising Star (co-organized by MIT, Stanford University, Carnegie Mellon University, and the University of Illinois Urbana-Champaign). The author of 21 journal publications, including five as first author, she joined ECS as a student in 2024.

Industrial Electrochemistry and Electrochemical Engineering Division Ralph E. White Outstanding Student Award

Tushar Khemraj Telmasre is a PhD candidate in Materials Science and Engineering at The University of Texas at Austin (UT Austin) with Prof. Venkat Subramanian as advisor. His research seeks to bring greater coherence among electrochemical theory, simulation, and practice by focusing on experimental design and validation that directly support improved battery performance and the precise estimation of states and model parameters. He has developed advanced physics–based electrochemical models in both the time and the frequency domains for lithium-ion and next-generation battery systems with the goal of enabling fast and accurate models for battery management systems (BMS). His work has led to nine publications spanning charging optimization, impedance modeling, pack modeling, and FMU–based battery digital twins, each informed by the need for models that can withstand experimental scrutiny.
Tushar completed his MS in Metallurgical and Materials Engineering at the Colorado School of Mines; MS at the Indian Institute of Technology Kanpur, where his MS thesis received the Gold Medal; and BTech at the Visvesvaraya National Institute of Technology. His received awards include the 2024 Allen J. Bard Center for Electrochemistry Student Scholar Fellowship.
Since 2022, Tushar has served as President of the ECS UT Austin Student Chapter, leading initiatives that include collaborations with other ECS student chapters in Texas.
Nominations March 15 through June 15 (approved in the fall)
Nominations October 31 through January 31 (approved in the spring)
someone making a big impact? Plan accordingly to submit your nomination within the new window! www.electrochem.org/society-awards



ECS is proud to announce the new members for October, November, and December 2025. Members are listed alphabetically by family/last name.
Members w A
Aeshah Alamri, Dammam, El Hasa, Saudi Arabia
Soujanya Reddy Annapareddy, Lake Forest, CA, US
B
Dominika Buchberger, Warsaw, Mazovia, Poland C
Peng Chen, Ithaca, NY, US
Rebecca Ciez, West Lafayette, IN, US
George Cintra, Jamestown, RI, US
D
Jesús Alejandro De Sousa Rodriguez, Antwerp, Antwerp, Belgium
Nicolas Duarte, Cheswick, PA, US
F
Wasif Farooq, Dhahran, El Hasa, Saudi Arabia
Colin Fischer, Essen, NRW, Germany
Jihyeon Gim, Naperville, IL, US
Ksenija Glusac, Chicago, IL, US I
Naoko Isobe, Hagagun, Tochigi, Japan
Piotr Kowalski, Jülich, NRW, Germany
Castro Laicer, Acton, MA, US
Ju Hun Lee, Ansan, Gyeonggi-do, ROK
Tao Li, Naperville, IL, US
Carlos Lopez, Houston, TX, US N
Kensaku Nagasawa, Koriyama, Fukushima, Japan
Louis Noodleman, San Diego, CA, US
Oleg Prezhdo, Albuquerque, NM, US
R Rannow, Colorado Springs, CO, US
Ken Sakaushi, Tsukuba, Ibaraki Prefecture, Japan
Muhammad Nawaz Tahir, Dhahran, El Hasa, Saudi Arabia
Tsutomu Takayama, Chiyoda-ku, Tokyo, Japan
Andrew van Bommel, Bedford, NS, CA
Andres Villa, Indianapolis, IN, US
Dung Vu, Deajeon, Chungcheongnam-do, ROK
Matthias Waegele, Chestnut Hill, MA, US
Guanyi Wang, Lemont, IL, US
Hung-Ta Wang, Shanghai, China
Magdalena Winkowska-Struzik, Warsaw, Mazovia, Poland
Gento Yamahata, Atsugi-shi, Kanagawa, Japan
Jiayi Yu, Los Angeles, CA, US
ZLingshi Zhang, Beijing, Beijing, China
Student Members
AWaliu Abdulrasheed, Dammam, El Hasa, Saudi Arabia
Ademola Adeniji, Mulhouse, Grand Est, France
Samuel Adigbo, Varennes, QC, CA
Bilal Ahmad, Buner, KPK, Pakistan
Zainab Al Badarin, Dhahran, El Hasa, Saudi Arabia
Mai Algarni, Dhahran, El Hasa, Saudi Arabia
Maryam Al-Harazi, Dhahran, El Hasa, Saudi Arabia
Joud Aljohani, Dharan, El Hasa, Saudi Arabia
Shakira Allah Baksh, Dhahran, El Hasa, Saudi Arabia
Shathar Alobeidat, Halifax, NS, CA
Talal Al-Qahtani, Zahran, El Hasa, Saudi Arabia
Hussa Alwohaibi, Dhahran, El Hasa, Saudi Arabia
Khakim Asadov, College Park, MD, US
Muhammad Muzamal Ashfaq, Fargo, ND, US
Saffena Aziz, Srinagar, JK, India
BTrent Badrak, University Park, MD, US
Surendar Balu, Chennai, TN, India
Ayesha Bano, Dammam, El Hasa, Saudi Arabia
Jaxiry Barroso Martínez, Bellaterra, CAT, Spain
Amina Bhatti, Lahore, Punjab, Pakistan
Muhammad Bilal, Dhahran, El Hasa, Saudi Arabia
Maryem Boughanmi, Dhahran, El Hasa, Saudi Arabia
Svenas Burba, Rykene, Aust Agder, Norway
CBianca Camacho Rodriguez, Lubbock, TX, US
Linyu Cao, Chongqing, Jiangsu, China
Sherry Cao, Richmond Hill, ON, CA
Xifei Cao, Ann Arbor, MI, US
Lingtong Chen, Chongqing, Jiangsu, China
Xi Chen, Chongqing, Jiangsu, China
Jiajun Cui, Chongqing, Jiangsu, China
DXi Liang Deng, Chongqing, Jiangsu, China
Abhinandan Dhar, Joynagar Majilpur, WB, India
Mohamed Draou, Dhahran, El Hasa, Saudi Arabia
Markus Driller, Garching, BY, Germany
Yuting Du, Chongqing, Jiangsu, China
Xingpei Duan, Chongqing, Jiangsu, China
EJosephine Esposto, Peterborough, ON, CA
FWeitong Fu, Chongqing, Jiangsu, China
Yuqiao Fu, Chongqing, Jiangsu, China
G
Yaser Gadelhak, Dammam, Riyadh, Saudi Arabia
José Gallo, San Carlos, CA, US
Srividhya Ganesan, Chennai, TN, India
Pengju Gao, Toronto, ON, CA
Eduarda Gibbert, Calgary, AB, CA
Irene Gómez Berenguer, Madrid, MAD, Spain
Wenchao Gong, Chongqing, Jiangsu, China
Zachary Griffith, Venetia, PA, US Antonia Gyger, München, BY, Germany
HTasneem Hammami, Dhahran, El Hasa, Saudi Arabia
Alexander Harrison, Calgary, AB, CA
Rahma Hasan, Jidhafs, Capital Governorate, Bahrain
James Ho, Evanston, IL, US
Robert Hoye, Atlanta, GA, US
Xin Hu, Chongqing, Jiangsu, China
Junyu Huang, Chongqing, Jiangsu, China
Qiyu Huang, Chongqing, Jiangsu, China
Zayneb Hussain, Toronto, ON, CA
J
Juhee Jang, Evanston, IL, US
Aqsa Javaid, Dhahran, El Hasa, Saudi Arabia
KDavid Keisar, Cambridge, MA, US
Justin Kelley, Plainview, TX, US
Abbas Khanmohammadi, Potsdam, NY, US
Ryo Kitamura, Clemson, SC, US
Anil Kumar, Delhi, DL, India
Himanshu Kumar, Hong Kong, Hong Kong, China
Rohini Kumari, Varanasi, UP, India
LJessica Lam, Calgary, AB, CA
Wing-chi Lam, Chicago, IL, US
Carinna Lapson, Richland, WA, US
Kevin Larkin, Pittsburgh, PA, US
Destiny Law, Tallahassee, FL, US
Evelyn Li, Toronto, ON, CA
Frank Li, Toronto, ON, CA
Honghong Li, Chongqing, Jiangsu, China
Huan Li, Chongqing, Jiangsu, China
Jiaqi Li, Chongqing, Jiangsu, China
Jinghan Li, Waterloo, ON, CA
Qifan Li, Victoria, BC, CA
Yixiu Li, Chongqing, Jiangsu, China
Yunlong Li, Chongqing, Jiangsu, China
Qian Liu, Chongqing, Jiangsu, China
Wentao Liu, Chongqing, Jiangsu, China
Xuan Liu, Chongqing, Jiangsu, China
Yongchao Liu, Chongqing, Jiangsu, China
Devon Louwe, Calgary, AB, CA
Qiyue Luo, Providence, RI, US
August Luure, Tartu, Tartu, Estonia
Xinzhuo Lv, Chongqing, Jiangsu, China
MZeyu Ma, Cambridge, MA, US
Justin Mainville, Sainte-Julie, QC, CA
Mahdiyeh Mallahi Karai, Hamilton, ON, CA
P. Manju, Mumbai, MH, India
Robert Marshall, College Station, TX, US
Laura Martinčič, Ljubljana, Osrednjeslovenska, Slovenia
Katia McCallum, Calgary, AB, CA
Shima Mehregan, Columbia, MO, US
Karol Viviana Mejía Centeno, Barcelona, CT, Spain
Casey Mezerkor, Richland, WA, US
MD Sumon Miah, Lubbock, TX, US
Jessica Moras, Calgary, AB, CA
Vikas Muralidharan, Broomfield, CO, US
Preethi Muruganandam, Tiruchirappalli, TN, India
Umair Mushtaq, Dhahran, El Hasa, Saudi Arabia
Pooja Nehra, Hyderabad, TG, India
Lidia Nemtsov, Toronto, ON, CA
OWonkyoung Oh, München, BY, Germany
Yuki Ono, Sendai-City Taihaku-ku, Miyagi, Japan
Skerxho Osmani, Casale Monferrato, AL, Italy
PPeter Pajka, Commerce City, CO, US
Dhananjaya Panda, New Delhi, DL, India
Gaurav Pande, Chennai, TN, India
Rupali Pandey, North East Delhi, DL, India
Junze Pang, Chongqing, Jiangsu, China
Samuel Parker, Goleta, CA, US
Yusuf Parvez, Coventry, England, UK
Avery Pritchard, La Jolla, CA, US
QXiaoxiao Qi, Chongqing, Jiangsu, China
Lucie Quincke, Garching, BY, Germany
RHaley Redfern, Wallsend, NSW, Australia
Wilma Rishko, Berkeley, CA, US
Constantin Ruess, Garching, BY, Germany
SKiana Sabzehei, Vancouver, BC, CA
Ebenezer Sam, College Park, MD, US
Kabery Sarker, Lubbock, TX, US
Tehzeeb Sawaira, Narowal, Punjab, Pakistan
Shahide Sayadian, Columbia, SC, US
Jordan Scalia, Buffalo, NY, US
Ishani Senevirathna, Chicago, IL, US
Reham Shams-Eldin, Chicago, IL, US
Xiaorui Shi, Cambridge, England, UK
Michael Shola David, Melbourne, Victoria, Australia
Sachin Sigdel, Dallas, TX, US
Winne Silva, Columbia, MO, US
Evan Stern, Eugene, OR, US
Natalie Strom, Columbia, SC, US
Shuguo Sun, Newark, DE, US
TZehang Tan, Chongqing, Jiangsu, China
Ans Bin Tariq, Dhahran, El Hasa, Saudi Arabia
Qanita Tayyaba, Sainte-Foy, QC, CA
Serena Tenhumberg, München, BY, Germany
Jiajia Tian, Chongqing, Jiangsu, China
Michael Tomechko, Berkeley, CA, US
UJulian Ulrich, Karlsruhe, BW, Germany
VVeronika Vachenauer, München, BY, Germany
WXu Wang, Chongqing, Jiangsu, China
Yonglin Wang, Waterloo, ON, CA
Shaoqing Wen, Chongqing, Jiangsu, China
Oskar Wittmann, Oberschleißheim, BY, Germany
Krzysztof Wojno, Purda, Warmian-Masurian Voivodeship, Poland
XYaxin Xia, Chongqing, Jiangsu, China
Yuntao Xie, Chongqing, Jiangsu, China
YPaifeng Yang, Chongqing, Jiangsu, China
Patrick Yang, Cypress, CA, US
ZMohammad Asaf Zaki, Minneapolis, MN, US
Muhammad Zeeshan, Dhahran, El Hasa, Saudi Arabia
Taylor Zeman, Tucson, AZ, US
Chenyang Zhang, Chongqing, Jiangsu, China
Jing Zhang, Chongqing, Jiangsu, China
Ling Zhang, Chongqing, Jiangsu, China
Taifang Zhang, Chongqing, Jiangsu, China
Weijian Zhang, Chongqing, Jiangsu, China
Xinyuan Zhang, Chongqing, Jiangsu, China
Yuzhen Zhang, Chongqing, Jiangsu, China
Zichen Zhang, Chongqing, Jiangsu, China
Yuewen Zhong, Chongqing, Jiangsu, China
Bao Zhan Zhou, Chongqing, Jiangsu, China
Lin Zhou, Chongqing, Jiangsu, China
Xinyi Zhu, Chongqing, Jiangsu, China
Youheng Zhu, Chongqing, Jiangsu, China
Ziyang Zhuang, München, BY, Germany
Lisa Zimprich, München, BY, Germany
MEMBER ANNIVERSARIES
Member Anniversaries 2026
It is with great pleasure that we recognize the following ECS members who reached their 30th, 40th, 50th, or 60th anniversary milestones with the Society in 2026. Congratulations to you all!
60-Year Members
Herbert D. Barber
David James Curran
Arabinda N. Dey
William G. Howard
Dieter Landolt
Herbert J. Moltzan
R.Winston Revie
50-Year Members
Robert D. Braun
Madhav Datta
Gilbert J. Declerck
Karl W. Frese
David S. Ginley
Richard D. Goodin
William R. Heineman
Henry G. Hughes
Theodore I. Kamins
Martin W. Kendig
Donald W. Kirk
Tyler X. Mahy
Kimon Papadopoulos
Patricia Paulette
Ronald A. Rizzo
Michael D. Ryan
Rajendra Singh
Subhash C. Singhal
Lawrence A. Tinker
Masamichi Yamashita
José H. Zagal
40-Year Members
Jennifer A. Bardwell
José-María Bastidas
Christopher M. A. Brett
Gessie Brisard
Arturo Bronson
Steve Crouch-Baker
Alanah Fitch
Neal Golovin
Arnold Z. Gordon
Steven G. Greenbaum
Hans Aage Hjuler
Michael A. Kepros
Robert E. Kraska
Rodney M. Lafollette
Keryn K. Lian
Boryann (Bor Yann) Liaw
Cortney K. Mittelsteadt
Tatsuo Nakato
David Ofer
Shi-Woo Rhee
Toshio Shibata
Joseph Stockel
W.Stephen Tait
Francisco A. Uribe
Petr Vanýsek
30-Year Members
Carmel B. Breslin
Thierry Brousse
Alicia B. Brune
Ravi R. Chandran
Shaowei Chen
Yet-Ming Chiang
Dev Chidambaram
David E. Cliffel
David J. Duquette
Grant M. Ehrlich
Henry F. Erk
Hiroyuki Fujimori
Naoko Fujiwara
Yutaka Fujiwara
Pedro Gómez-Romero
Fernando González
Gregory L. Griffin
Hitoshi Habuka
Yutaka Hiratsuka
Minoru Inaba
Lars-Gunnar Johansson
Katsumi Katakura
Do-Heyoung Kim
Shinichi Komaba
Chowdary R. Koripella
Prashant N. Kumta
Claude Lévy-Clément
Jing Li
Vladimir G. Litovchenko
Jiří Ludvík
Kazuo Matsuzaki
Duane J. Mazur
Chris Menictas
Yuriy Mikhaylik
Shelley D. Minteer
Shigenori Mitsushima
Suzanne E. Mohney
Toshiyuki Momma
Masayuki Morita
Nosang Vincent Myung
Makoto Nanko
James J. Noel
Toshiaki Ohtsuka
Yasuhisa Omura
Sachiko Ono
M.Rosa Palacín
Kim L. Pamplin
Aleksandr Y. Pikelny
Mikko K. Ritala
R.Ekwal E. Sah
Chariclea A. Scordilis-Kelley
Yasuhiro Shimizu
Yukihiro Shimogaki
Eiichi Shoji
Parthasarathi Sridhar
I-Wen Sun
Scott L. Swartz
Keiichi Takahashi
George G. Totir
Wei-Tsu Tseng
Hiroyuki Uchida
Mikito Ueda
CH Wang
Adam H. Whitehead
David L. Wood
Weibing Xing
Kang Xu
Atsuo Yamada
Takeshi Yao
Jim P. Zheng
Andreas R. Zielonka
STUDENT NEWS STUDENT NEWS
ECS lImenau Student Chapter
The second ECS Ilmenau Student Chapter meeting of 2025 was held at Ostbayerische Technische Hochschule (OTH) Regensburg with thirteen online and in-person participants. The event brought together students and early-career researchers to exchange experiences, research insights, and professional perspectives in a collegial setting.
The meeting opened with a presentation by Dr. Martin Leimbach, who shared his experience transitioning from academia to industry following his postdoc. He provided valuable insight into navigating the industrial environment and offered practical advice for young researchers considering a similar path. This was followed by a talk from Dr. Marius Grad, who reflected on his journey toward a PhD, discussing his motivation, challenges, and key milestones along the way.
Suvetha Logeswaran introduced her newly launched industrial PhD project, which focuses on novel electroplating approaches for through glass vias (TGV) in next-generation interposers for semiconductor applications. She outlined her research objectives and provided an insightful perspective on applied electrochemical research in an industrial context. Jessica Kloiber then presented her work on the bioactivation of magnesium, emphasizing the influence of electropolishing and corrosion behavior, and highlighting its relevance for biomedical applications.
The program continued with a joint presentation by Jan Zentgraf and Lukas Esper, who showcased current developments in 3D printing of biomaterials and metals across several OTH Regensburg laboratories. Their contribution provided exciting insights into innovative manufacturing techniques and post-processing methods.
The event concluded with a locally guided tour of the UNESCO World Heritage City of Regensburg, followed by a group dinner. These activities provided participants with excellent opportunities for informal professional and personal exchanges.
The ECS Ilmenau Student Chapter extends its sincere gratitude to all meeting speakers and participants for their contributions and engagement, as well as to the organizers for making the meeting a success. The next chapter meeting is planned for spring 2026.

Participants at the fall 2025 ECS Ilmenau Student Chapter meeting, held in conjunction with the Technische Universität Ilmenau biannual Electrochemistry and Electroplating Group PhD student meeting.
Florian Erzinger
ECS Montréal Student Chapter
The Montréal Student Chapter’s 13th Annual Symposium on November 14, 2025, brought together more than 65 students, professors, and researchers from nine universities. The event featured highly engaging oral and poster presentations, along with outstanding keynote talks by Dr. Samantha Gateman from Western University and Dr. Ana Tavares from the Institut national de la recherche scientifique.
The chapter also organized a Sip of Science evening on September 22, during which students shared their research through a Draw Your Thesis activity. This event provided an opportunity to network and exchange ideas while enjoying delicious food and drinks. Another in-person event was the November 26 industry tour of the Institut des communications graphiques et de l’imprimabilité, where students visited laboratories and learned about the Institute’s diverse activities.
(continued on next page)

Photo:
Participants at the ECS Montréal Student Chapter’s 13th Annual Symposium.
(continued from previous page)
STUDENT NEWS STUDENT NEWS


Two webinars were hosted by the chapter. On July 28, Prof. Xia Li of Concordia University delivered an innovative talk on the development of sustainable cathode materials for all-solid-state
batteries, and on December 11, Dr. Alex Peroff from Pine Research presented an introductory course on cyclic voltammetry and iR compensation.
ECS Navajo Technical University Student Chapter
The chapter held its first in-person meeting in fall 2025, establishing the first Native American ECS Student Chapter. The student leaders introduced themselves and chapter activities and timelines, and the advisors encouraged members to expand outreach in the areas of electrochemistry and sensor research.
As part of their fall 2025 outreach, on October 27, 2025, chapter members visited Chinle High School in Chinle, AZ, an unincorporated area of Apache County. In spring 2026, the chapter plans to expand outreach and recruit members from across the Navajo Nation, which spans parts of Arizona, New Mexico, and Utah.

ECS North Carolina State University Student Chapter
As one of the fastest growing industries in the Raleigh-DurhamChapel Hill Research Triangle, the battery and electrochemistry sector offers tremendous opportunities. However, students often have limited access to industry professionals. The ECS North Carolina State University (NC State) Student Chapter aims to bridge this gap by facilitating meaningful connections between students and leaders in the field. During the fall of 2025, the chapter hosted three signature events designed to connect students with cutting-edge electrochemical research, foster industry partnerships, and strengthen collaborative networks across the Research Triangle.
The chapter visited Pine Research Instrumentation in Durham, NC, where members gained insight into the collaborative design process behind electrochemical testing equipment. The Pine Research team described how electrochemists, electrical engineers, and software engineers work together to design rotators, workstations, and electrodes. Students also learned about instrument calibration procedures and marketing strategies for engaging clients within the

ECS Navajo Technical University Student Chapter President Wayant Billey and chapter member Karen Dan promote electrochemistry research activities at Chinle High School, AZ.
ECS North Carolina State University Student Chapter members tour the Pine Research facilities.
Photo: Frank Dalton
The ECS Montréal Student Chapter brings students together to Draw Your Thesis during the Sip of Science event.
Dr. Alex Peroff from Pine Research presents an introductory course on cyclic voltammetry and iR compensation during an ECS Montréal Student Chapter webinar.
electrochemical research community. It was especially meaningful to have Pine Research serve as the chapter’s first site tour, given the company’s long-standing impact on the electrochemistry community and its role in the chapter’s growth.
The chapter hosted its inaugural NC State Electrochemistry Open House, welcoming local students and chapter members from Duke University and the University of North Carolina at Chapel Hill. The event showcased state-of-the-art electrochemistry labs, facilities, and cutting-edge research across multiple departments at NC State. Visitors toured the labs of Prof. Veronica Augustyn (Department of Materials Science and Engineering and ECS NC State Student Chapter Faculty Advisor) and Prof. Susmita Sarkar (Department of Mechanical and Aerospace Engineering), gaining firsthand exposure to the diverse electrochemical research underway at NC State. The open house created a vibrant forum for inter-university exchange and strengthened connections across the Research Triangle’s electrochemistry community.

The keynote speaker was Jennifer McConnell, Product Manager at Protochips, a company specializing in in situ TEM holders and sample supports. Ms. McConnell discussed collaborative innovation at Protochips, outlining how cross-functional teams communicate at every stage from early brainstorming through final testing to product. Her presentation gave students valuable insight into the role of interdisciplinary teamwork in developing cutting-edge microscopy technology for electrochemical research.
The chapter is proud to further ECS’s mission by engaging the university and broader communities in advancing electrochemical research and providing opportunities for collaboration. Looking ahead, the chapter plans to build on this momentum by further strengthening connections between academia and industry.

ECS Ohio University Student Chapter

On October 27, 2025, the ECS Ohio University Student Chapter hosted Eric Knauf for a seminar talk and professional development workshop. Eric is the founder of BelongHQ, author of The 56% Solution: How Belonging Infrastructure Transforms Performance, and a Senior Fellow with The Conference Board’s Human Capital Practice. He has previously served as global head of talent at high-growth companies that include SAP, WorkRise, Tradeshift, Commvault, and Goodyear.
Eric’s presentation, “Belonging Infrastructure: Your Career Catalyst – A Reality Check for Chemical Engineering PhD Students,” gave students valuable insight into the five pillars of belonging and how to identify and evaluate them within organizations. Eric also shared practical advice to help students develop into successful and impactful leaders in industry.
(continued on next page)

Members of Ohio University’s Tau Beta Pi, American Institute of Chemical Engineers (AIChE), and ECS Ohio University Student Chapter host a December 4 bingo night, including periodic table bingo.
Photo by Abigail Paul
From left to right: Pine Research Electrical Engineering Manager John Castellucci (far right) demonstrates device manufacturing, testing, and calibration to (from left to right) Chapter Vice President Matthew Ayoola, Chapter Secretary Tony Lavelle, and Colton King
Photo: Alex Peroff
From left to right: Chapter Secretary Tony Lavelle, Chapter Treasurer Adriana Lara, Protochips Marketing and Product Manager Jennifer McConnell, Chapter President Alan Ferris, and Prof. Ray Unocic gather after the ECS Industry Speaker Series Protochips presentation.
Photo: Matthew Ayoola
ECS Ohio University Student Chapter’s invited speaker Eric Knauf of BelongHQ hosts an interactive workshop for PhD students
Photo by Abigail Paul
(continued from previous page)
STUDENT NEWS STUDENT NEWS
In the evening, Eric led an interactive workshop titled “The 56% Solution: Engineering Belonging Through Collaboration.” Students worked collaboratively to discuss small, actionable steps they could take in their daily lives to foster a stronger sense of belonging within their organizations and communities.
To close out the fall semester, on December 4, the chapter cohosted a bingo night with the Ohio University American Institute of Chemical Engineers (AIChE) and Tau Beta Pi student organizations. Attendees enjoyed multiple rounds of bingo—including periodic table bingo—and won festive prizes!

ECS Ontario Tech University and Trent University Student Chapter
On September 27, 2025, the chapter hosted its first symposium—a fullday event that brought together student researchers and faculty with shared interests in electrochemistry. The program featured insightful oral presentations by undergraduate and graduate students on topics that included electro-organic chemistry, materials for electrocatalysis and energy storage, and the development of electrochromic films. For many participants, the symposium provided a valuable opportunity to foster new connections and engage in meaningful discussion with a broader audience. A highlight of the symposium’s program was a guided tour of Ontario Tech University’s Electrochemical Materials Lab, which is led by Prof. Brad Easton. Attendees learned about the group’s innovative electrochemical research.
Building on the inaugural event’s success, the chapter looks forward to continuing this momentum by creating future opportunities for networking, collaboration, and community building among its members.

ECS Texas A&M University Student Chapter
The chapter leads in encouraging, teaching, and supporting students of all levels in understanding and pursuing the science and engineering of electrochemistry at Texas A&M University (TAMU). During the fall 2025 semester, the chapter participated in many events supporting these endeavors.
Semester activities started on October 11, 2025, with an Engineering Tailgate at the Zachary Engineering Education Complex. Students presented a redox battery demonstration to an audience of all ages, demonstrating the incredible nature of electrolytes and how they function as charge carriers. Utilizing zinc and copper plates which worked as anodes and cathodes respectively, they built a lowvoltage battery that could power an LED light. Using many different hydration-focused drinks, they showed how the different electrolytes inside them allowed for a larger magnitude of charge to pass through the LED, which in turn increased the brightness. The presentation was a huge success. Adults and children learned a great deal about how batteries used every day rely on electrolytes to function, much as we do. Participating in the event increased awareness of the science of electrochemistry and also was a great learning experience for chapter members.
On October 18, the Department of Chemistry and College of Arts and Sciences hosted the 2025 Chemistry Open House as part of National Chemistry Week. Thousands of visitors gathered for the event, themed “The Hidden Life of Spices.” The chapter played an active role in engaging the community through a handson electrochemistry demonstration focused on saltwater batteries. The outreach activity explored how different salts and spice-based seasonings influence electrochemical performance. Visitors compared Creole seasoning, Himalayan pink salt, lava salt, potassium chloride,

and sea salt, learning how ionic composition and sodium content affect voltage generation. By connecting everyday kitchen materials to fundamental electrochemical principles, the experiment sparked curiosity among K–12 students, their families, and educators. Through interactive demonstrations and clear scientific explanations, the chapter highlighted the relevance of electrochemistry in daily life while supporting large-scale science outreach. The event aligned with ECS values in showcasing the chapter’s commitment to education, public engagement, and interdisciplinary outreach. Following this fun event, the chapter concluded its outreach activities for the semester and shifted its focus toward learning and professional development.
ECS Ohio University Student Chapter members interact with guest speaker Eric Knauf during his interactive workshop on October 27.
Photo by Abigail Paul
Attendees gather for the ECS Ontario Tech University and Trent University Student Chapter’s first symposium.
ECS Texas A&M University Student Chapter members participate in the 2025 Chemistry Open House
Photo: Laura Hoagland
STUDENT
On November 10th, the chapter invited TAMU graduate Dr. Denis Johnson to present “Applications of Electrochemistry Techniques in Aqueous and Molten Salt Environments.” Dr. Johnson presented research from his doctoral thesis on ammonia production in titanium nitride MXenes and provided guidance on interpreting electrochemical data, offering valuable instruction for newer chapter members. The second part of his talk highlighted his ongoing work at Argonne National Laboratory, demonstrating how similar electrochemistry techniques can be applied to the analysis of nuclear and molten salt materials. The chapter extends its sincere thanks to Dr. Johnson for returning to TAMU to share his expertise and insight into both academic and national laboratory research. Inspired by electrochemistry work at the national level, the group then transitioned to hands-on learning within academic labs.
Student chapters also reconnected this past fall through interchapter engagement! Our chapter visited the ECS University of Texas at Austin Student Chapter to explore potential collaborations, strengthen new and old friendships, and learn more about ongoing electrochemistry and solid-state research. The day began with travel from College Station to Austin, followed by presentations from the UT Austin chapter highlighting research from professors’ groups, including the David Mitlin, Arumugam Manthiram, C. Buddie Mullins, and Venkat Subramanian groups. The visit continued with lab tours, including the Hang Ren and Michael Aubrey Labs. A standout moment was observing a synthetic muscle in the Aubrey Lab, where applied voltage controlled system movement!
After a brief excursion across campus and lunch, the group continued touring laboratories and learning about research connected to John B. Goodenough’s Nobel Prize–winning work on lithium-ion batteries. The day ended with miniature golf, dinner, and live music at a local restaurant. After a full and inspiring day, the TAMU students returned home, energized by the experience and looking forward to future visits!
The Texas A&M Electrochemical Society had a remarkable fall 2025 semester. Through outreach, invited seminars, and interchapter collaboration, the chapter continued to uphold the ECS mission of advancing electrochemical and solid state science. Chapter leadership thanks all the chapter members and supporters and looks forward to continued growth and engagement in the years ahead.


ECS Texas Tech University Student Chapter
The ECS Texas Tech University (TTU) Student Chapter continued its commitment to professional development, technical exchange, and student engagement through a series of academic and industryfocused activities during the year.
One of the chapter’s key learning initiatives was an industrial site visit to X-FAB, where members gained first hand exposure to semiconductor manufacturing processes and clean room operations. The tour provided valuable insight into microfabrication technologies, industrial electrochemistry applications, and semiconductor sector career pathways. Students engaged with industry professionals and learned how electrochemical principles are applied in large-scale manufacturing environments.
The chapter also hosted three ECS TTU Student Chapter Online Seminars during fall 2025, featuring distinguished researchers from academia: Professors Joseph Gauthier, Texas Tech University, and Abdoulaye Djire, Texas A&M University, and Postdoctoral Researcher Raul Marquez, University of Texas at Austin. These seminars provided opportunities to interact with leading experts, ask technical questions, and gain exposure to diverse research directions within electrochemical science and engineering.
(continued on next page)

ECS Texas Tech University Student Chapter members explore semiconductor fabrication and industrial electrochemical applications at X-FAB.
Texas A&M University graduate Dr. Denis Johnson presents “Applications of Electrochemistry Techniques in Aqueous and Molten Salt Environments” to ECS Texas A&M University Student Chapter members.
Photo: Laura Hoagland
Touring the Michael Aubrey Lab was a highlight of the ECS Texas A&M University Student Chapter’s visit with the ECS University of Texas at Austin Chapter.
Photo: Laura Hoagland
(continued from previous page)
STUDENT NEWS STUDENT NEWS
The chapter elected 2026 officers, ensuring continuity of leadership and student involvement. The newly elected officers are President Sergio A. Arias, Vice President Jessica Alexandra Ortega Ramos, Secretary Sergio A. Arango, Treasurer Daniela Ferreira Garcia, and Undergraduate Chair Bianca Camacho Rodriguez. With new leadership in place, the chapter remains committed to its ongoing efforts to support students, encourage collaboration, and engage the broader electrochemical community.

ECS University of Calgary Student Chapter
In response to the success of CHEM Connect, the chemistry department networking mixer held last fall, the chapter has received significant interest from students and faculty in establishing this event as an annual program during the fall semester.
In November, graduate students also had the opportunity to tour two facilities operated by one of Calgary’s oldest chemical manufacturers, Nova Chemicals. This visit provided valuable exposure to industrialscale research environments, including laboratories dedicated to materials discovery, product development and testing, and pilot plant operations.
The chapter is planning several initiatives to further support graduate student development in 2026. These include hosting a seminar speaker on the application of machine learning to materials discovery in electrochemical applications. A second workshop series is planned to strengthen graduate students’ familiarity with the commonly used materials characterization instruments that are available on campus. These workshops feature example datasets to build proficiency in data processing, interpretation, and best practices. Together, these activities aim to enhance both technical skills and professional engagement within the community.

ECS University of Chicago Student Chapter
Although it’s new, the chapter has started out strong. Officially formed in fall 2024, the chapter’s inaugural executive team—co-Presidents Avi Gargye and Emily Doyle, Communications & Outreach Director Ashley Lam, Finance Director Hrishikesh Srinivasan, Secretary Andrea Diaz, and Undergraduate Representative Sammy Alderden—aims to foster a strong sense of community among students involved in and interested in electrochemistry research. To that end, the chapter has been actively organizing technical talks, outreach events, and social events.
Notably, ECS hosted its 248th Meeting in October 2025 in Chicago. The University of Chicago was well represented, with many undergraduates, PhD students, and postdocs presenting their groundbreaking research. On the penultimate evening of the meeting, the chapter organized a student mixer at the Chicago Athletic Association to celebrate the meeting’s successful conclusion. Chapter members were joined by students and postdocs from institutions across the country, including the University of Michigan; Washington University in St. Louis; Montana State University; Massachusetts Institute of Technology; University of California, Berkeley; Dartmouth College; Argonne National Laboratory; and the National Laboratory of the Rockies.

Students participate in the ECS Texas Tech University Student Chapter fall 2025 kickoff meeting.
Photo: Alejandro Gutierrez
ECS University of Calgary Student Chapter members tour Nova Chemicals with a guide (center).
Photo: Dr. Rohen Prinsloo
ECS Student Members from around the country celebrate the success of the 248th ECS Meeting in Chicago.

Back on campus, the chapter has been busy planning a variety of events for members and for the broader university community. They include a series of invited speaker seminars featuring experts in the lithium-ion battery field, cohosted with Prof. Y. Shirley Meng’s Laboratory for Energy Storage and Conversion (LESC) group. In August, the chapter hosted Dr. Stephen Harris (Lawrence Berkeley National Laboratory) who delivered an insightful presentation titled “Predicting Durability with Machine Learning: How to Improve Our Usefulness to the Battery Industry and End Users.” The seminar was followed by a small group breakout session, giving students the opportunity to engage more deeply with Dr. Harris through questions about his presentation and their own research, as well as receiving career advice.
On Halloween (October 31), the chapter welcomed Prof. Zheng Chen (University of California San Diego) who presented a compelling seminar on his group’s recent advances in battery recycling and solid state battery manufacturing. These talks are part of an ongoing effort to regularly host speakers whose work is directly relevant to chapter members’ research, while also highlighting emerging frontiers in electrochemistry. Previous invited speakers include Prof. Jordi Cabana (University of Illinois Chicago), who provided an educational primer on synchrotron X-ray techniques for studying battery materials, and Dr. Brian Ingram and Dr. Justin Connell (Argonne National Laboratory), who discussed their recent efforts to electrify steelmaking.
Beyond technical programming, the chapter is committed to outreach and community building. In October, the members volunteered at the University of Chicago’s annual Southside Science Festival, which attracted approximately 4,300 kids and parents from across Chicago. Using hands-on demonstrations such as lemon batteries and thermoelectric generators, chapter volunteers introduced elementary and middle school students to fundamental concepts of electrochemistry.
The chapter also regularly organized socials to strengthen connections among the university’s electrochemistry research groups. Activities included a summer barbeque on the shores of Lake Michigan, where members cooked burgers, hot dogs, and s’mores, and tossed around a rotating disk electrode (Frisbee). To cap off a great first year, the chapter hosted a holiday party with students competing in a lively electrochemistry-themed Jeopardy game.
In 2026, the chapter plans to expand its programming to create additional opportunities for students to further their professional development and engage with leaders in the field. Chapter leadership also aims to broaden outreach efforts throughout the Chicagoland area by strengthening partnerships with industry and universities.

Professor Zheng Chen poses with attendees at his seminar, “Towards Sustainable Battery Recycling and Solid-State Battery Manufacturing.”
ECS University of Chicago Student Chapter members take a break from the lab to enjoy a barbeque at Promontory Point.
STUDENT NEWS STUDENT NEWS
ECS University of Michigan Student Chapter
Over the course of the fall 2025 semester, the chapter brought together students and researchers from across campus for a dynamic program of technical talks, interactive networking opportunities, and community-building social events designed to promote academic enrichment and collaboration in electrochemistry.
The chapter welcomed professors and industry professionals from Michigan and beyond to deliver a series of talks. Dr. Chi-Hao Chang from Dow Chemical Company presented “Solutions for Battery Cell/ Module/Pack Safety Improvements,” highlighting how siliconebased materials enhance fire protection and safety in lithium-ion battery packs. Prof. Chengcheng Fang of Michigan State University followed with “Closing the Scale-Up Gap: Advanced Materials and Manufacturing for High-Performance Lithium Batteries,” sharing recent advances from her lab that integrate materials design with scalable manufacturing approaches to accelerate lithium-battery deployment.
Later in the term, the chapter hosted Prof. Gerald Meyer, Arey Distinguished Professor at the University of North Carolina at Chapel Hill and Deputy Director of CHASE (Center for Hybrid


Approaches in Solar Energy to Liquid Fuels). His talk, “Solar Energy Conversion with Molecular-Semiconductor Hybrid Photoelectrodes,” provided mechanistic insights into interfacial electron transfer and proton-coupled electron transfer reactions in semiconductor-based photoelectrochemical systems. To cap off the fall seminar series, Prof. Brian McCloskey, Chair of Chemical and Biomolecular Engineering, Warren and Katharine Schlinger Distinguished Professor of Chemical Engineering, University of California, Berkeley, and Lawrence Berkeley National Laboratory, delivered a virtual talk titled “Quantifying Li Plating on Graphite during Fast Charging of Li-Ion Batteries.” His presentation detailed chemical and electrochemical methods for detecting and quantifying lithium plating on graphite anodes, providing new insight into the mechanisms underlying lithium plating onset and capacity fade during fast charging of lithium-ion batteries.
In addition to technical seminars, the chapter emphasized professional development by organizing opportunities for students to visit diverse research environments and engage with external professors and industry professionals. Members participated in a guided tour of the Michigan Center for Materials Characterization (MC)², gaining firsthand insight into state-of-the-art instrumentation and experimental techniques.
The chapter also hosted numerous networking dinners that allowed students to connect informally with peers, professionals, and invited speakers. One such event featured two battery industry leaders— Celina Mikolajczak (Chief Battery Technology Officer, Lyten) and Dr. Said Al-Hallaj (CEO, AllCell Technologies, LLC). Members discussed career pathways, industry trends, and perspectives on careers in industry and academia with both visitors. The chapter continues to foster community and camaraderie through a series of social events, including a game night co-hosted with the Graduate Society of Women Engineers (GradSWE) and a holiday-themed movie night. These events provided relaxed settings for members to connect and strengthen relationships beyond the classroom and laboratory.

Prof. Gerald Meyer (University of North Carolina at Chapel Hill) presents “Solar Energy Conversion with Molecular-Semiconductor Hybrid Photoelectrodes,” as part of the ECS University of Michigan Student Chapter seminar series.
Photo: Fernando Villavicencio.
The ECS University of Michigan Student Chapter seminar series features “Closing the Scale-Up Gap: Advanced Materials and Manufacturing for High-Performance Lithium Batteries” by Prof. Chengcheng Fang (Michigan State University).
Photo: Fernando Villavicencio.
The ECS University of Michigan Student Chapter hosts a networking dinner with Celina Mikolajczak (Chief Battery Technology Officer, Lyten) and Dr. Said Al-Hallaj (CEO, AllCell Technologies, LLC).
Photo: Fernando Villavicencio.
ECS INSTITUTIONAL PARTNERS
BENEFACTOR PARTNERS
BioLogic, Knoxville, TN US
Duracell US Operations, Inc., Bethel, CT US
Gamry Instruments, Warminster, PA US
Hydro-Québec, Varennes, QC Canada
PalmSens BV, Houten, Netherlands
Pine Research Instrumentation, Durham, NC US
SPONSORING PARTNERS
BASi, West Lafayette, IN US
Center for Solar Energy and Hydrogen Research Baden-WÜrttemberg (ZSW), Germany
Central Electrochemical Research Institute, Tamil Nadu, India
Corteva Agriscience, Indianapolis, IN US
DLR – Institute of Engineering Thermodynamics , Oldenburg, Germany
EL-CELL GmbH, Hamburg, Germany
Ford Motor Company, Dearborn, MI US
GS Yuasa International Ltd., Kyoto, Japan
Honda R&D Co., Ltd., Tochigi, Japan
Medtronic, Inc., Minneapolis, MN US
Nel Hydrogen, Wallingford, CT US
Nissan Motor Co., Ltd., Yokosuka, Japan
NSF Center for Synthetic Organic Electrochemistry, Salt Lake City, UT US
Pacific Northwest National Laboratory (PNNL), Richland, WA US
Panasonic Energy Corporation, Osaka, Japan
Permascand AB , Ljungaverk, Sweden
Plug Power, Inc., Latham, NY US
Teledyne Energy Systems, Inc., Hunt Valley, MD US
UL Research Institutes, Northbrook, IL US
PATRON PARTNERS
easyXAFS, LLC , Renton, WA US
Energizer Battery, Westlake, OH US
Faraday Technology, Inc., Clayton, OH US
GE Aerospace Research, Niskayuna, NY US
Lawrence Berkeley National Laboratory (LBNL), Berkeley, CA US
Scribner, LLC , Southern Pines, NC US
Toyota Research Institute of North America (TRINA), Ann Arbor, MI US
SUSTAINING PARTNERS
BMW Group, München, Germany
Current Chemicals, Cleveland, OH US
Electrosynthesis Company, Inc., Lancaster, NY US
General Motors Holdings LLC , Warren, MI US
Giner, Inc., Newton, MA US
Ion Power, Inc., New Castle, DE US
Littelfuse, Inc., Wiltshire, United Kingdom
Los Alamos National Laboratory (LANL), Los Alamos, NM US
Metrohm USA, Inc., Riverview, FL US
Microsoft Corporation, Redmond, WA US
next Machinery Group I nextCoatema Technologies GmbH , Chadds Ford, PA US
Occidental Petroleum Corporation, Houston, TX US
Sandia National Laboratories, Albuquerque, NM US
Sandisk GK , Tokyo, Japan
Sensolytics GmbH, Bochum, Germany
Sherwin-Williams, Minneapolis, MN US
Spectro Inlets ApS , Copenhagen, Denmark
Technic, Inc., Providence, RI US
United Mineral & Chemical Corporation, Lyndhurst, NJ US
Westlake Corporation, Monroeville, PA US
Support the vital work of ECS by joining as an institutional partner. Contact Anna Olsen, Senior Manager, Corporate Programs, at sponsorship@ electrochem.org, to renew, join, or discuss program options
UPCOMING MEETINGS

249th ECS Meeting
May 24–28, 2026
Seattle, WA US
Seattle Convention Center
250th ECS Meeting
October 25–29, 2026
Calgary, Canada BMO Centre
251st ECS Meeting
May 30–June 3, 2027
Washington, DC US
Walter E. Washington Convention Center and Marriott Marquis
252nd ECS Meeting
October 17–21, 2027
Detroit, MI US Huntington Place Convention Center

253rd ECS Meeting
May 21–25, 2028
Gothenburg, Sweden
Swedish Exhibition & Congress Centre
