

SOUTHWESTRETORT
SEVENTY
-NINTH YEAR February 2026
Published for the advancement of Chemists, Chemical Engineers and Chemistry in this area published by
The Dallas-Fort Worth Section, with the cooperation of five other local sections of the American Chemical Society in the Southwest Region.
Vol. 79 (6) February 2026
Editorial and Business Offices: Contact the Editor for subscription and advertisement information.
Editor: Connie Hendrickson: retort@acsdfw.org
Copy and Layout Editor: Lance Hughes: hugla64@gmail.com
Business Manager: Martha Gilchrist: Martha.Gilchrist@tccd.edu
The Southwest Retort is published monthly, September through May, by the Dallas-Ft. Worth Section of the American Chemical Society, Inc., for the ACS Sections of the Southwest Region.







Data Entry and Editing
Specializes in:
• Company newsletters
• Confidential Data Entry
• Free Quotes Contact Lance at hugla64@gmail.com Or 214-356-9002
2026 ACS DFW Executive Committee
Chair: Jonathan Dannatt, PhD
Chair-elect: Rajani Srinivasan, PhD
Past Chair: Denise Lynn Merkle, PhD
Treasurer: Martha Gilchrist, MS
Secretary: Celyna Rackov, PhD Councilors:
Mary E. Anderson, PhD (2025-2026)
Kirby B. Drake, JD (2025-2027)
Linda D Schultz, PhD (2026-2028)
Rebecca Weber, PhD (2025-2027)
Alternate Councilors:
Daniela Hutanu, PhD (2025-2026)
Trey Putnam, PhD (2026-2028)
Daniel Tran, PhD (2025-2027)
Yunxiang Li, PhD (2025-2027)

From the ACS Press Room
Secondhand vape plumes could form lung-damaging radicals
“Interplay of Metals and Organics in E‑Cigarette Aerosols Enhances the Production of Reactive Oxygen Species within Ultrafine Particles: Implications for Passive Vaping Exposures”
Environmental Science & Technology
Electronic cigarettes or vapes can release puffs of vapor in aromatic clouds. The health risks of breathing in this secondhand or passive vapor aren’t fully understood. So, researchers reporting in ACS’ Environmental Science & Technology conducted a preliminary study on lingering vape plumes in indoor environments. They found that aged vapor contained fine particles with several metals and highly reactive compounds, which together produce radicals that might damage lung tissue if inhaled.
“Our study reveals that the chemical cocktail of metal nanoparticles and reactive peroxides in aged e-cigarette aerosols creates a unique profile of respiratory health risks, highlighting that secondhand vapor is something bystanders shouldn't have to breathe,” says Ying-Hsuan Lin, the corresponding author of the study from the University of California, Riverside.
Unlike traditional cigarettes, e-cigarettes don’t create smoke; they create vapors that could expose non-users to harmful substances. Early studies showed that these secondhand vape aerosols contain volatile organic compounds that react with indoor ozone, creating new compounds, such as peroxides. Additionally, vape liquids and puffs commonly contain heavy metals, as well as
other metals that could easily react with peroxides to produce potentially damaging compounds like free radicals.
In another step toward understanding the po-

The secondhand vapor from e-cigarettes can react with ozone in the air, forming particles that could negatively impact lung health, especially for people with asthma or other lung conditions.
Yta23/Shutterstock.com
tential health effects of secondhand vape plumes, Lin and colleagues examined how ozone indoors impacts the metal and peroxide composition of vape aerosols. They also wanted to see what happens when these substances react in wet environments, simulating what might happen inside the lungs.
For their experiments, the researchers created a simplified vape liquid with one flavoring ingredient (a floral-smelling terpene) and no nicotine, loaded it into two different vape pens with refillable cartridges, and puffed it into a chamber with ozone in the air. After 90 minutes, they collected the aged aerosols for analysis. The particles from both pens contained iron, aluminum and zinc ions, as well
Continued on page 15
From the ACS Press Room
Tiny gold spheres could improve solar energy harvesting
“Plasmonic Supraballs for Scalable Broadband Solar Energy Harvesting”
ACS Applied Materials & Interfaces
Sunbeams contain a lot of energy. But current technology for harvesting solar power doesn’t capture as much as it could. Now, in ACS Applied Materials & Interfaces, researchers report that gold nanospheres, named supraballs, can absorb nearly all wavelengths in sunlight including some that traditional photovoltaic materials miss. Applying a layer of supraballs onto a commercially available electricity converter demonstrated that the technology nearly doubled solar energy absorption compared to traditional materials.

This supraball (top) is 2100 nanometers in diameter and is made from hundreds of tiny gold nanoparticles (bottom) engineered to boost solar energy absorption. Adapted from ACSApplied Materials & Interfaces 2026, DOI: 10.1021/ acsami.5c23149
Scientists are exploring materials that absorb light across the solar spectrum to improve solar energy harvesting. Gold and silver nanoparticles (NPs) have been suggested as a solution because they’re easy and cost-effective to make, but current NPs’ light absorption is confined to visible wavelengths a fraction of the full solar spectrum. To capture additional wavelengths, including near -infrared light, Jaewon Lee, Seungwoo Lee and Kyung Hun Rho propose using self-
assembling gold supraballs. These structures consist of gold NPs that clump together and form tiny spheres. The diameter of the supraballs was adjusted to maximize the absorption of wavelengths present in sunlight.
The researchers first used computer simulations to optimize the design of individual supraballs and to predict the performance of supraball films. Results from the simulations showed that the supraballs should absorb more than 90% of wavelengths from sunlight.
Next, they created a film of gold supraballs by drying a liquid solution containing the structures on the surface of a commercially available thermoelectric generator (TEG), a device that converts light energy into electricity. The films were created in ambient room conditions no clean rooms or extreme temperatures required.
In demonstrations with an LED solar simulator, the supraball-coated TEG had an average solar absorption of about 89%, nearly twice that of a TEG with a conventional film made from single gold NPs (45%).
“Our plasmonic supraballs offer a simple route to harvesting the full solar spectrum,” says Seungwoo Lee. “Ultimately, this coating technology could significantly lower the barrier for high-efficiency solar-thermal and photothermal systems in real-world energy applications.”
The authors acknowledge funding from the National Research Foundation of Korea, the Korea Institute of Science and Technology Institutional Program, the Korea-US Collaborative Research Fund, and a Korea University grant.
From the ACS Press Room
Toothbrush-activated powder whitens, repairs and protects teeth
“Sr‑, Ca-Doped BaTiO3 with Synergistic Piezoelectric Catalysis and Microbial Balance Effects Enables Tooth Whitening for Home Oral Health”
ACS Nano
Even with regular brushing, teeth can become stained from genetic factors or consuming foods and drinks like tomatoes and coffee. Chemical whiteners can help, but they can also damage teeth in the process. So, researchers reporting in ACS Nano have designed a prototype teeth-whitening powder that is activated by the vibrations from an electric toothbrush. The system both brightened and protected teeth in lab demonstrations.
“This work offers a safe, at-home teeth whitening strategy integrating whitening, enamel repair and microbiome balance for long-term oral health,” says Min Xing, first author on the study.
Peroxide-based bleaching with strips, gels and mouth rinses is a popular option among consumers for whitening stained teeth. This type of chemical whitening generates reactive oxygen species (ROS), compounds that break apart stain-causing molecules. But in addition to removing stains, many whitening techniques can damage the tooth enamel, which might result in easier re-staining or other oral problems. Now, Xing, Wenhao Qian, Xuanyong Liu, Jiajun Qiu and colleagues are developing a tooth-whitening compound that generates ROS when activated by elec-
tric toothbrush vibrations while strengthening and repairing teeth at the same time. The researchers combined strontium and calcium ions with barium titanate in a solution and then heated and formed it into a ceramic powder, dubbed BSCT. When vibrated, the powder generates a small electric field a phenomenon known as the piezoelectric effect that causes ROS-generating chemical reactions.

The vibrations from an electric toothbrush can activate a compound that whitens, repairs and protects teeth. New Africa/Shutterstock.com
In initial lab tests using human teeth artificially stained with tea and coffee, four hours of brushing with BSCT and an electric toothbrush caused visible whitening. After 12 hours of brushing, treated teeth were almost 50% whiter than the control group stained in the same way but brushed with saline. For teeth with damaged enamel and dentin, BSCT brushing regenerated these structural components because the strontium, calcium and barium ions included in the powder formed deposits on the tooth surface. Next, researchers tested the powder on rats
Continued on page 15

From the ACS Press Room
Turning industrial exhaust into useful materials with a new electrode
“Integrated Capture and Conversion of Dilute CO2 Using an Oxygen Tolerant Porous Carbon Modified Gas Diffusion Electrode”
ACS Energy Letters
Flue gas is exhausted from home furnaces, fireplaces and even industrial plants, and it carries polluting carbon dioxide (CO2) into the atmosphere. To help mitigate these emissions, researchers reporting in ACS Energy Letters have designed a specialized electrode that captures airborne CO2 and directly converts it into a useful chemical material called formic acid. The system performed better than existing electrodes in tests with simulated flue gas and at ambient CO2 concentrations.

Carbon dioxide captured from industrial flue gases is efficiently converted into formic acid with a new, simple electrode.
Nomad_Soul/Shutterstock.com
“This work shows that carbon capture and conversion do not need to be treated as separate steps. By integrating both functions into a single electrode, we demonstrate a simpler pathway for CO2 utilization under realistic gas conditions,” explains Wonyong Choi, a corresponding author on the study.
Capturing CO2 from the air should be relatively simple after all, plants do it all the time. But converting the gas into something useful is difficult, and it is a crucial step in ensuring that carbon capture methods are widely implemented. In industrial emissions like flue gas, CO2 is often diluted amid other gases such as nitrogen and oxygen. However, existing conversion methods require highly concentrated CO2 that’s already separated from other gases to function efficiently. So, Donglai Pan, Myoung Hwan Oh, Wonyong Choi and colleagues wanted to design a carbon capture and conversion system that functioned in conditions consistent with real-world flue gas and could convert even small amounts of captured CO2 into a useful product.
The team constructed an electrode that allows gas to diffuse in, then catches and converts the airborne CO2. The electrode consists of three layers: a specialized carboncapturing material, gas-permeable carbon paper, and catalytic tin(IV) oxide. This design converted CO2 gas directly into formic acid, a valuable starting material for a variety of chemical applications, including fuel cells.
In tests with pure CO2 gas, the new electrode was around 40% more efficient than other existing carbon-converting electrodes under comparable laboratory conditions. More importantly, in tests with a simulated flue gas containing 15% CO2, 8% oxygen gas and
Continued on page 15
From the ACS Press Room
Why don’t antibiotic-making bacteria self-destruct?
“An Acetyltransferase Conferring SelfResistance of the Producer to Lasso Peptide
Antibiotic Lariocidin”
ACS
Infectious Diseases
Early in 2025, scientists discovered a promising new antibiotic in a soil sample from a lab technician’s backyard. The molecule, called lariocidin, is produced by the microbe Paenibacillus and shows broad activity against pathogenic bacteria, including several that are multi-drug-resistant. Now, the researchers report in ACS Infectious Diseases how Paenibacillus avoids harm by its own antibiotic information that is crucial for developing lariocidin or similar compounds into new drug candidates.
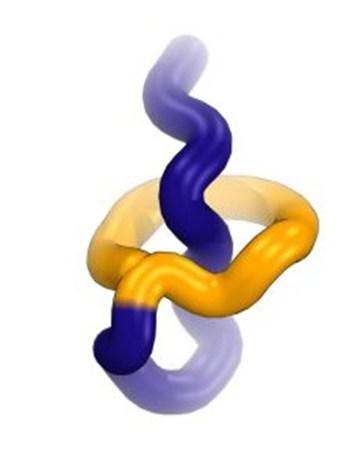
This computer simulation shows the recently discovered lariocidin, a lasso-shaped compound with its “lasso loop” shown in yellow and “tail” shown in purple.
Yury Polikanov and Dmitri Travin/University of Illinois, Chicago
The discovery of a new antibiotic is just the first step in advancing it toward clinical use. It is vital to understand potential mechanisms of resistance to pressure test the novelty and clinical potential of the original discovery,” says Gerry Wright, the lead investigator of the lariocidin project.
As bacteria evolve resistance to drugs, new antibiotics are urgently needed. In their initial breakthrough,
Wright and colleagues identified that a slowgrowing Paenibacillus strain produces lariocidin, which inactivates the bacteria against which it competes for resources in the soil. Expanding on that work, the researchers examined how the microbe resists its own powerful antibiotic.
Lariocidin deactivates bacteria by binding to ribosomal RNA and interfering with protein synthesis. In the current study, the researchers identified one enzyme (shortened to lrcE) produced by the Paenibacillus strain that modifies lariocidin. Their experiments revealed that lrcE adds a functional group onto the antibiotic molecule, which prevents it from binding to the bacterium’s ribosomal RNA, thereby protecting Paenibacillus. Additionally, the enzyme was specific to lariocidin and did not impact other antibiotic compounds such as aminoglycosides and streptothricins.
Then, from an analysis of the bacterium’s genome, the researchers characterized the gene that encodes the lariocidin-resistance enzyme. A small-scale search for similar genes in environmental bacteria and human pathogens revealed some in Bacillus genomes and environmental proteobacteria but none in the genomes of human pathogens. The researchers say that gene transfer from environmental bacteria to human pathogens is slow and rare, but if lariocidin or related compounds become pharmaceutical treatments, potential gene transfer should be monitored. These insights suggest to the team that lariocidin holds promise as a powerful next-generation
Continued on Page 16
From the ACS Press Room
Oil residues can travel over 5,000 miles on ocean debris, study finds
“Long-Range Transport of Oil by Marine Plastic Debris: Evidence from an 8500 km Journey”
Environmental
Science & Technology
When oily plastic and glass, as well as rubber, washed onto Florida beaches in 2020, a community group shared the mystery online, attracting scientists’ attention. Working together, they linked the black residue-coated debris to a 2019 oil slick along Brazil’s coastline. Using ocean current models and chemical analysis, the team explains in ACS’ Environmental Science & Technology how some of the oily material managed to travel over 5,200 miles (8,500 kilometers) by clinging to debris.

Adapted from Environmental Science & Technology 2026, DOI: 10.1021/acs.est.5c14571
“The research findings of our study would not have been possible without the dedication of the Friends of Palm Beach,” says Bryan James, lead author of the study and a researcher at Northeastern University.
“Their long-term knowledge of the local marine debris enabled them to notice when unique and interesting items like oily plas-
tic comes ashore. If they hadn’t been willing to investigate and share their observations, this discovery would still be lost at sea.”
Although some plastics can drift thousands of miles on ocean currents, crude oil or refined petroleum usually doesn’t. Instead, sunlight and microbes break down oil within a few hundred miles (300 kilometers) of where it entered the water. So, in 2020, the source of oily plastic bottles and glass containers along the shore in Palm Beach, Florida, was curious to the Friends of Palm Beach cleanup group. With no spills reported nearby, the group’s main clues about the oil’s source were the bottle labels in Portuguese, Spanish and English, and large chunks of rubber that had also washed up. The group became community scientists as some members teamed up with international researchers led by James and Christopher Reddy at Woods Hole Oceanographic Institution to find the origin of the oil and plastic pollution. James, Reddy and their colleagues hypothesized that the oily plastic and rubber littering the beach in Florida could have the same origin as similar pollution found on Brazil’s coast in late 2019. And the source of the oil and rubber might be the SS Rio Grande, a sunken World War II supply ship in the Atlantic Ocean. To test their hypothesis, the researchers conducted computer simulations and oil spill forensic analyses.
• Origin: Ocean current models traced the plastic bottles backwards in time, predicting origins spanning from the Gulf
Continued on page 16
In 2020, members of the Friends of Palm Beach found oily plastic and glass (left) and floating rubber chunks (right) on the beach, and researchers traced their origin offshore of Brazil.

DFWACS MEETING-IN-MINIATURE (MIM)
SATURDAYAPRIL18, 2026 9 a.m. - 5 p.m.
McFadden Science Center
Call forAbstracts!
Registration is Open! Registration for this meeting is free and will close on April 1.
Call forAbstracts!
Post-docs, grad, and undergrad students to submit abstracts for a 10 12-minute oral presentation•

Register or Submit Abstracts
From the ACS Press Room
Frozen hydrogen cyanide ‘cobwebs’ offer clues to origin of life
“Electric Fields Can Assist Prebiotic Reactivity on Hydrogen Cyanide Surfaces”
ACS Central Science
A substance poisonous to humans hydrogen cyanide may have helped create the seeds of life on Earth. At cold temperatures, hydrogen cyanide forms crystals. And, according to computer models reported in ACS Central Science, some of the facets on these crystals are highly reactive, enabling chemical reactions that are otherwise not possible at low temperatures. The researchers say these reactions could have started a cascade that gave rise to several building blocks of life.
“We may never know precisely how life began, but understanding how some of its ingredients take shape is within reach. Hydrogen cyanide is likely one source of this chemical complexity, and we show that it can react surprisingly quickly in cold places,” says Martin Rahm, the corresponding author of the study.
Hydrogen cyanide is abundant in many environments beyond Earth, including on comets and the atmosphere of planets and moons (e.g., Saturn’s moon Titan). Additionally, when combined with water, hydrogen cyanide can form polymers, amino acids and nucleobases (components of proteins and DNA strands, respectively). To further examine its chemical reactivity, Marco Capelletti, Hilda Sandström and Martin Rahm conducted computer simulations of frozen hydrogen cyanide.
The researchers simulated a stable hydrogen cyanide crystal as a 450-nanometer-long cylinder with a rounded base and a multifaceted top shaped like a cut gemstone. They say this shape matches prior observations of crystal “cobwebs” branching out from a central point, where the multifaceted ends come together.

In cold environments, hydrogen cyanide forms solid crystals, which computer simulations predict have multifaceted tips that attract other crystals to form the cobweb-like structures scientists observe in experiments.
Adapted from ACS Central Science 2026, DOI: 10.1021/ acscentsci.5c01497
Calculations revealed that the crystals have the potential to trigger chemical reactions that don’t usually occur in extreme-cold environments. Using the chemical properties of the crystal surface, the researchers identified two pathways that could convert hydrogen cyanide into the more-reactive hydrogen isocyanide within minutes to days, depending on the temperature. They say that the presence of hydrogen isocyanide at the crystals’ surface suggests that other, more complex prebiotic precursors could form there.
The team now hopes that other scientists will follow up on these predictions by performing
experiments, in which hydrogen cyanide crystals are crushed in the presence of substances like water, to see if the exposed crystal surfaces can indeed encourage the production of complex molecules at extremely cold temperatures.
The authors acknowledge funding from the Swedish Research Council and the National Academic Infrastructure for Supercomputing in Sweden.



Around the Area

UT Dallas

The Department of Chemistry and Biochemistry welcomes Dr. Shruti Hariyani to the faculty as an Assistant Professor. Following the awarding of her Ph.D. in Chemistry from the University of Houston, Dr. Hariyani was a Visiting Research Scientist at the Paul Scherrer Institute/ETH Zürich and an Arnold O. Beckman Postdoctoral Fellow at TAMU; her research concerns Designing Dynamically-Responsive Inorganic Materials for Energy Storage and Information Processing. Professor Jeremiah J. Gassensmith was awarded a$75K American Cancer Society grant titled, Reactivating Innate Immunity in cGAS -Deficient HER2+ Ovarian
Tumors Using a Virus-Like Particle-Based cGAS Delivery Platform. Poster Session Awardees from the 2025 Welch Foundation Conference on Chemical Research included Graduate Student Sachin Giri (1st Place) and Postdoctoral Trainee Dr. Sidhant Wagulde (2nd Place), both from the Group of Robert A. Welch Distinguished Chair in Chemistry Vladimir Gevorgyan. Doctoral student Shelby Phelps received the Jess Hay Chancellor’s Graduate Student Research Fellowship from the UT System, and will continue her research in the Group of Associate Professor Sheel C. Dodani.
From the ACS Press Room
continued
Secondhand vape plumes
Continued from page 5
as trace amounts of heavy metals such as lead, arsenic and tin. And the two sets of aged aerosols had similar levels of peroxides. The smallest particles, classified as ultrafine particles, contained higher percentages of metals and peroxide compounds compared to larger aerosols.
To preliminarily understand how aged aerosols react with lung fluids, the researchers placed the samples in a water-based solution. The aged aerosols created radicals with the ultrafine particles producing 100 times more radicals relative to their weight as compared to larger particles. Because ultrafine particles can get deep into humans’ lungs and enter the sensitive, fluid-lined alveoli, the researchers say the results indicate these particles’ potential to damage lung tissues and lower respiratory function.
The researchers acknowledge that the study was done under controlled laboratory conditions, and more research is needed using real -world indoor environments and commercially available e-cigarette liquids. Regardless, these results suggest that repeated exposure to aged vape plumes could negatively impact lung health, especially for individuals with pre-existing lung conditions such as asthma or chronic obstructive pulmonary disease, commonly referred to as COPD.
The authors acknowledge funding from the University of California, the Office of the President Tobacco-Related Disease Research Program and a NRSA T32 training grant.
Toothbrush-activated powder
Continued from page 7
fed high-sugar diets. Daily brushing for one minute over four weeks using BSCT helped restore the rats’ oral microbiome, killing periodontitis-causing Porphyromonas gingivalis and Staphylococcus aureus bacteria and reducing inflammation.
Though current tests haven’t yet incorporated the BSCT powder into a toothpaste formula, researchers say that this study is a step toward a new effective, at-home treatment for safely whitening teeth and promoting oral health.
The authors acknowledge funding from the Youth Innovation Promotion Association of the Chinese Academy of Sciences, the Science and Technology Commission of Shanghai Municipality, the Shanghai Medical Key Specialty, the Medical Key Subject of Xuhui District, and the Opening Project of the State Key Laboratory of High Performance Ceramics.
Turning industrial exhaust
Continued from page 9
77% nitrogen gas, it continued to produce a substantial amount of formic acid, with the other systems producing a negligible amount. Finally, the new electrode system captured CO2 at concentrations similar to current atmospheric levels, demonstrating its utility to operate in ambient air conditions. The re-
From the ACS Press Room
continued
searchers say that this work offers a promising strategy for integrating CO2 capture into practical industrial applications, and they hope that it can lead to similar systems to capture other greenhouse gases like methane.
The authors acknowledge funding from the National Research Foundation of Korea.
Why don’t antibiotic-making bacteria self-destruct?
Continued from page 10
antibiotic treatment for humans with minimal risk of resistance, making it a promising candidate for preclinical development.
The authors acknowledge funding from the Canadian Institutes for Health Research, the National Institute of General Medical Sciences of the National Institutes of Health, and Illinois State startup funds.
Oil residues can travel over 5,000 miles
Continued from page 11 of Mexico, CentralAmerica and Brazil.
• Travel time: Additional models estimated that the oily debris drifted for 240 days, which is a timeframe consistent with currents carrying material from the 2019 Brazil oil spill to the Florida coast.
• Chemical analyses: Several of the oily residues collected from the Florida debris showed evidence of refining, and the “chemical fingerprints” of the oily
plastic matched those collected from the Brazil oil spill.
Reddy concludes that this work demonstrates an additive contaminant effect where plastic pollution can transport oil pollution far beyond its origin, and it expands on the current understanding of “petroplastic” a recently recognized form of plastic pollution from humans.
The authors acknowledge collaboration from the Friends of Palm Beach and the Brazilian National Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico, CNPq) Research Productivity Fellowship and PQ-1 grant, the Long Term Ecological Research Program-Semi-Arid Coast of Brazil (Programa de Pesquisa Ecológico de Longa Duração Costa Semiárida do Brasil, PELD-CSB), the Alexander Von Humboldt Foundation-CAPES Coordenação de Aperfeiçoamento de Pessoal de Nível Superior) fellowships, the I-plastics Consortium within the Joint Programming Initiative Healthy and Productive Seas and Oceans Cearense Foundation for Scientific and Technological Development Support (Fundação Cearense de Apoio ao Desenvolvimento Cientifico e Tecnológico, FUNCAP), the Ceará Foundation for Scientific and Technological Development Support (FUNCAP) Chief Scientist Program, the Coordination for the Improvement of Higher Education Personnel within the Ministry of Higher Education in Brazil, the U.S. National Science Foundation, the State of Florida, the Woods Hole Oceanographic Institution’ s Independent Research and Development Program, and the Natural Sciences and Engineering Research Council of Canada.
From the Editor
It’s almost spring, and that means it’s time for the Meeting-in-Miniature. This year it will be on Saturday,April 18, at Texas Wesleyan in Fort Worth. When I entered graduate school, I had rarely spoken in front of a class. We did have a required course called Speech, and we had to give very short talks on pre-determined subjects. But at LSU, the biochemistry department had seminar every semester, including summer semester, and we were required to take it twice a year. I was pretty shaky about it at first...but it became natural after one or two runs. I cannot emphasize enough that undergraduate as well as graduate students should get up and talk in front of a class as often as possible.
So, point being, present your work at the M-i-M!

