Europea n Biotechnolog y



OUTSOURCING Today, 90% of biotechs outsource manufacturing to CDMOs. Outsourcing decisions are no longer straightforward cost-benefit analyses. Geopolitical volatility and unprecedented demand for advanced modalities have made CDMO partnerships existential. Pharma and biotech leaders face fundamentally different pressures, tensions that take center stage at CDMO Live Europe 2026 (19–21 May, Rotterdam).
The European CDMO market is expanding at 7–7.6% CAGR, with valuations clustering around €35–€42 billion in 2025–2026 and forecasts reaching €60–€89 billion by 2034–2035. Germany leads with 30% market share, while France is the fastestgrowing market, driven by €1.87 billion in pharmaceutical investment for manufacturing capacity.
PharmaSource analysis of CDMO news announcements over the last 12 months shows that 30% were related to Europe, with US manufacturing at 46% as Amer-
ica-first policies encourage investment. This reflects Europe's critical strategic importance in global supply chain resilience. Growth varies significantly by modality:
› APIs command the highest share (between 42–64% of market revenue), reflecting sustained demand for generic and specialty chemicals.
› Biologics and monoclonal antibodies are expanding with new capacity additions, though competitive pricing has emerged as post-pandemic buildouts mature.
› Sterile fill-finish (vials, prefilled syringes, cartridges) is the most strategically

constrained segment, with explicit supply/ demand imbalances driven by biologics pipelines and the growth of GLP-1 medicines creating capacity pressures
› Highly potent APIs and bioconjugates (ADCs) are in high demand, with capacity expansions supported by long-term manufacturing contracts tied to GLP-1 peptide demand.
This expansion is underpinned by increasingly supportive regulatory frameworks. The EU's Critical Medicines Act aims to encourage supply security through manufacturing across multiple European sites. The EU Biotech Act is expected to accelerate biotech innovation and development timelines.
Manufacturing partnership decisions have become extraordinarily complex. Pharma external manufacturing leaders are seeking to move from transactional relationships toward strategic partnerships, driven by geopolitical volatility, capacity constraints, and the strategic imperatives of supply chain resilience. Long-term manufacturing contracts now underwrite capacity investment, while sponsors are embedding digital, sustainability, and resilience requirements into negotiations from the outset.


Tariffs, US sourcing pressure, and regionalization have moved from background to dominant strategic question. Multi-site, dual-sourced supply networks now essential for portfolio security.
Unexpected cost increases and renegotiations common. Balancing accountability with relationship preservation increasingly difficult.
Too many similar CDMO claims with insufficient comparison methods. Capability verification and differentiation remain challenging.
››› Recent PharmaSource research reveals divergent concerns shaping partnership dynamics. Large pharmaceutical companies prioritize geopolitical risk mitigation, cost stability, and portfolio optimization across fragmented CDMO landscapes. Early-stage biotech, conversely, struggles with capacity access, partner
Early-stage work deprioritized by large CDMOs. Critical need for CDMOs who take small companies seriously – the “true partner, not subcontractor” gap remains acute.
Runway anxiety reshapes decisions. Speed and cost efficiency prioritized over optimal tech selection and scale planning.
Need for specialized mAbs, bispecifics, ADCs, and CGT capability. Hard to verify claims reliably; reliance on references and due diligence remains high-friction.
prioritization, and rapid commercialization timelines.
These competing priorities are reshaping how CDMOs segment their customer base and design service offerings.
Sponsors now expect CDMOs to implement AI and digital manufacturing tools, yet a significant gap persists between


their capability and pharma/biotech expectations. Recent PharmaSource research reveals that 92% of sponsors ask for digital requirements in negotiations, while only 60% of CDMOs report operating at advanced maturity levels.
Regulatory pressure – particularly EU GMP Annex 1 standards – reinforces that European CDMOs must compete on specialized expertise and reliability, not price alone. Decarbonization is a strategic imperative: pharma companies pursuing netzero commitments are discovering that 70–80% of their carbon footprint sits in Scope 3 (their supply value chain), making CDMO manufacturing practices a material lever in emissions reduction.
At a time when drug supply chains are under unprecedented pressure, the need for strategic, trusted manufacturing partnerships has never been greater. CDMO Live Europe 2026 (19–21 May, Rotterdam) is firmly established as Europe's #1 event for external manufacturing.
600 external manufacturing professionals will convene across three days. The 40+ expert speaker roster includes manufacturing heads and CMC directors from MSD, Roche, Johnson & Johnson, Bayer, BMS, Daiichi Sankyo, Takeda, Sandoz, Polpharma, and others, bringing authentic, battle-tested perspectives on partnership strategy.
For organizations navigating Europe's complex manufacturing landscape, CDMO Live Europe 2026 (19-21 May, Rotterdam) is the must-attend event of the year. Secure your ticket at www.CDMOLive.com ■ Luke Bilton, Co-Founder, PharmaSource

Rezon Bio is a science-driven European CDMO specializing in the development and manufacturing of mammalian biologics. Headquartered in Poland, we provide fully integrated solutions from cell line development to commercial supply. With state-of-the-art facilities, world-class teams and a legacy of biosimilars development and global commercialization, Rezon Bio brings together deep scientific expertise, digital transparency and cost-efficiency to help clients move their biologics programs from concept to market with confidence.

Scan QR code to discover our capabilities

16
Years of operation

73 Batches produced in 2025
95%
Batch success rate achieved in 2025
35+
Successful audits (FDA, EMA, ANVISA)
MANUFACTURING Europe continues to play a pivotal role in the global biologics landscape, combining scientific excellence, regulatory maturity and a strong industrial foundation.
As biologic modalities grow more complex, and development timelines accelerate, the ability to transform innovation into robust, scalable, and compliant manufacturing has become a critical determinant of success.
Europe is at the heart of advanced biologics development
In this dynamic environment, CDMOs are evolving rapidly. They are increasingly expected to support drug development from the earlies stages, seamlessly integrating process development, analytics, quality and large-scale manufacturing considerations from the outset.
3PBIOVIAN was built around this integrated approach. As a pan European CDMO, the company supports biologics and advanced therapy programs from early development through clinical supply and commercial manufacturing, acting as a long-term partner rather than a transactional service provider.
Early decisions that have a lasting impact
For biotech and pharmaceutical companies across Europe, the objective is clear: to transform promising science into reliable products that can efficiently progress through clinical development into commercial supply. In this process, early decisions around a drug candidate’s manufacturability, scalability, and regulatory alignment often determine the long-term viability of the program.
3PBIOVIAN’s capabilities span a wide range of expression systems and modalities, including microbial and mammalian platforms for recombinant proteins, as well as viral vectors, plasmid DNA and advanced therapy products. This technological breadth allows development programs to evolve with flexibility, selecting the most appropriate manufacturing route as scientific and clinical needs change.
With GMP manufacturing sites in Spain (Pamplona) and Finland (Turku), 3PBIOVIAN combines industrial-scale capabilities with the agility demanded by Europe’s innovation driven biotech ecosystem. Its services cover both Drug Substance and Drug Product and are designed as a seamless pathway rather than a sequence of disconnected steps.
A strong quality culture forms the foundation of 3PBIOVIAN's organization, and is reinforced by multidisciplinary teams with extensive experience across development and manufacturing. In an industry where precision in execution is as vital as innovation, the expertise of people, the strength of collaboration, and the consistency of processes are what truly set organizations apart.
As biologics pipelines grow increasingly complex, European CDMOs are assuming

(Turku).
a more strategic role in shaping development outcomes. Combining extensive experience and track record with technological breadth, regulatory expertise, and an integrated end to end offering, 3PBIOVIAN exemplifies how modern European CDMOs are helping transform promising concepts into therapies that ultimately reach patients.
■ Contact us: 3PBIOVIAN www.3pbiovian.com
BEST PRACTICE As advanced therapeutic medicinal products (ATMPs) and other next-generation biologics move toward approval, Chemistry, Manufacturing and Controls (CMC) continues to be the most frequent source of regulatory concern. This article highlights recurring CMC barriers and best practices to mitigate approval delays or refusals.
› Martin Mewies, Ph.D., is director, regulatory affairs, CMC, at Cencora
As more advanced therapeutic medicinal products (ATMPs) and other next-generation biologics seek regulatory approval, the development challenges these products encounter is gaining prominence. Assessments from regulatory authorities consistently show that Chemistry, Manufacturing and Controls (CMC) remains the area of greatest concern.
Last year, the US Food and Drug Administration (FDA) published more than 200 complete response letters (CRLs) that had been issued between 2002 and 2024 for products that have since been approved1, providing greater transparency into the agency’s decision-making process.
Additionally, FDA has rejected or delayed several new cell and gene therapy (CGT) biologics license application (BLA) submissions due to problems with manufacturing readiness. According to the manufacturers of these products, deficiencies included process control gaps, inadequate stability data, unvalidated analytical methods, and ongoing issues following Good Manufacturing Practice (GMP) inspections2
These CMC and GMP issues are welldocumented in Europe, where most biological products and all ATMPs go through the Centralised Procedure (CP) with the European Medicines Agency (EMA). The agency has a long-established documentation framework and each product is subject to European public assessment reports (EPARs)3, whether approved or denied authorization. The release of the CRLs from FDA now al -

MARTIN MEWIES, PH.D., is director, regulatory affairs, CMC, at Cencora. He has nearly 30 years of experience in protein biochemistry, with over 20 years in biologics CMC / regulatory.
lows observers to more rigorously assess areas of respective interest and concern between the health authorities.
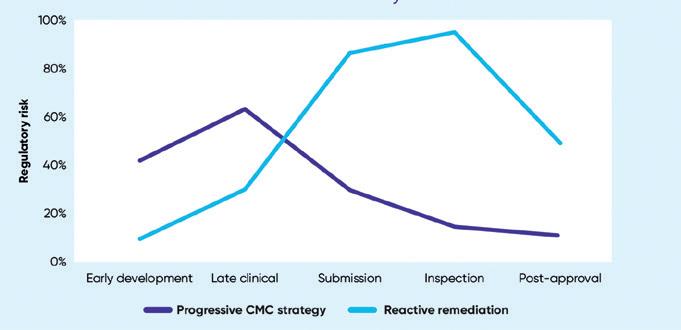
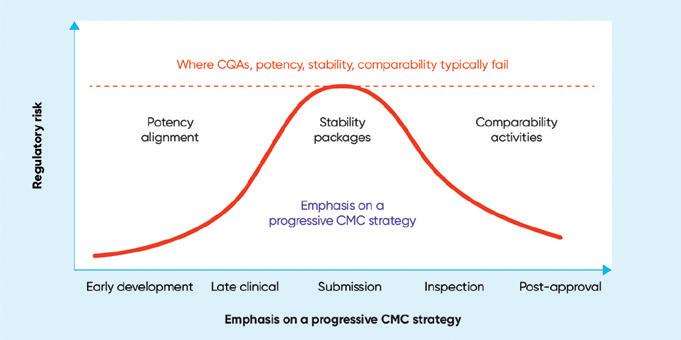
One of the core challenges with developing an ATMP is characterizing the process and getting well-understood critical quality attributes (CQAs.) Having those robustly understood and justified is key to unlocking a lot of the other CMC challenges4. A frequently identified CMC issue is poten-
cy, particularly getting a relevant potency assay that links to a meaningful CQA that accurately reflects the mode of action of the product. While potency issues are not new, the more complex the therapy, the harder it can be for manufacturers to convince regulators that their CQAs are meaningful and justified and will link back into the clinical efficacy that needs to be demonstrated in clinical trials.
This is relevant not only for potency but also for comparability – another issue highlighted in the CRLs. This refers to the pre- or post-change data when scaling, transferring sites, or changing materials. These changes inevitably occur during product development, particularly with biological products. Unless comparability is linked to meaningful CQAs it is hard to make a compelling case to support those changes.
The same is true for stability. Without stability data linking back to meaningful attributes of the product, it is hard to demonstrate that within reasonable parameters the product is stable over its shelf life and during transfer and transport. Steps to avoid negative findings from the regulators include generating real-time data early, carrying out degradation mapping, and establishing an interim shelf-life supported by a well-justified extension plan.
The CRLs also identify challenges with regards to manufacturers’ control strategies – another issue commonly highlighted in EPARs. They also reinforce the need to have an appropriate manufacturing environment, ensuring that all
facilities comply with standard GMP requirements, including sterility assurance. ATMPs are often made in small facilities using novel manufacturing technologies, nonetheless, the same requirements for GMP sterility assurance still apply. Positioning CPP/CQA controls within a riskbased quality framework, such as ICH Q9(R1) 5 , can demonstrate proactive compliance and inspection readiness.
Manufacturers must ensure they build in all the compendial requirements governed by the European Pharmacopoeia that control sterility and appropriate control and characterization of the process6.
ATMPs are inherently complex products, and small personalized products present even greater challenges. But having the appropriate quality control and processes in place is fundamental to product safety and should be built in from the outset. Embedding a holistic approach or best practices into the organization from concept through development and product submission can help to mitigate the CMC issues that have resulted in CRLs in the U.S. and marketing application refusals in Europe. Some best practices manufacturers can put in place include:
› Work cross-functionally to evaluate assays and guide decisions across the full development and commercialization lifecycle.
› Engage the health authorities early to seek scientific advice, leveraging programs like PRIME (where relevant) in Europe, the Innovative Licensing and Access Pathway (ILAP) in the UK, and the Support for Clinical Trials Advancing Rare Disease Therapeutics (START) pilot in the U.S. Raise as many CMC hotspot topics as possible during these interactions.
› Consult key opinion leaders and thirdparty experts to gain diverse therapeutic-area perspectives and to help build robust plans for monitoring and managing process changes.
› Future-proof the development process to pre-empt evolving regulatory expectations by focusing on data integration
(a priority for health authorities) and taking a forward-looking approach to the indicators regulators are looking for before and after inspections.
The CMC challenges facing ATMPs are inherent to the nature of the products and their novelty in the market. As guidelines evolve and as new interpretations of the guidelines emerge, the requirements will become more defined, which will make it easier to identify areas of discrepancy.
By understanding what regulators expect – through assessment of CRLs and EPARs and seeking good scientific advice – manufacturers can better understand this complex and dynamic environment and mitigate risk with their CMC processes. ■
1 FDA Embraces Radical Transparency by Publishing Complete Response Letters, FDA, July 2025. https:// www.fda.gov/news-events/press-announcements/fda-
embraces-radical-transparency-publishing-completeresponse-letters
2 Why gene and cell therapies are stalling at the FDA, Drug Discovery News, Aug 2025. https://www.drugdiscoverynews.com/why-gene-and-cell-therapies-arestalling-at-the-fda-16527
3 EMA. European public assessment reports: background and context. https://www.ema.europa.eu/en/ medicines/what-we-publish-medicines-when/european-public-assessment-reports-background-context
4 Johanna I, Daudeij A, Devina F, et al. Basics of advanced therapy medicinal product development in academic pharma and the role of a GMP simulation unit. Immuno-Oncology and Technology, October 2023. https://pmc.ncbi.nlm.nih.gov/articles/PMC10772236/
5 Q9(R1) Quality Risk Management, FDA, May 2023. https://www.fda.gov/regulatory-information/searchfda-guidance-documents/q9r1-quality-risk-management
6 European Pharmacopoeia (Ph. Eur.) 11th Edition, European Directorate for the Quality of Medicines & HealthCare. https://www.edqm.eu/en/european-pharmacopoeia-ph.-eur.-11th-edition
Disclaimer:
The information provided in this article does not constitute legal advice. Cencora strongly encourages readers to review the references provided with this article and all available information related to the topics mentioned herein and to rely on their own experience and expertise in making decisions related thereto.