ALPHA- -TO-ONE
Practical advice, personal experiences and pertinent news for people touched by ALPHA-1
Special Issue:




Practical advice, personal experiences and pertinent news for people touched by ALPHA-1




Practical advice, personal experiences, and pertinent news for people touched by Alpha 1, their families, and friends.
VOL. 23, NO. 3 (Winter 2026)
Published by the Alpha 1 Foundation 3300 Ponce de Leon Blvd., Coral Gables, FL 33134 (877) 2 CURE A1 (228-7321) ALPHA1.ORG
2026 is off to a great start, and we are excited to meet you where you are this year! Our first A1F Virtual Education Day in January brought the entire Alpha‑1 community together on our new online platform for the A1F State of the Union, along with incredible presentations from Alpha‑1 experts. All sessions are now available on‑demand at alpha1.org/video library.
A1F strives to provide programming, resources, education, and services tailored to each patient’s unique journey. Whether you are newly diagnosed, newly in need, on treatment, wanting to improve your quality of life, or feeling grateful and want to give back, there is something at A1F for you.
Our continued success starts with the patient journey, and A1F is poised and ready to accompany you in the future. We gain incredible insight from your Alpha 1 stories, making us wiser and more knowledgeable about living with this rare condition. We have fueled innovation, encouraging both scientific and clinical experts to seek new solutions. We have invested significant resources in groundbreaking initiatives while building lasting and trusting partnerships. Most importantly, the impact of our collective efforts has been a measurable improvement in the lives of Alphas.
As a community, we are on the cusp of innovative breakthroughs, a good portion of which would not be possible without A1F’s entrepreneurial spirit, 30 years of resources, and the inspired resilience we derive from working with and assisting patients and their families.
Alpha-1 Foundation Board of Directors
Executive Committee
Jon Hagstrom *, Chair
Peg Iverson*, Vice Chair
Catherine Vernon*, Secretary
Kenneth Irvine+, Treasurer
Virginia Clark, MD, Physician Director
Members
Mark L. Brantly, MD
Erin Carr+
Jennifer Jopp*
Ann Knebel, PhD, RN
Darrell N. Kotton, MD
Tammy McGuinness*
James K. Stoller, MD, MS
Alice Turner, MBChB, PhD
Martin R. Zamora, MD, Director Emeritus
Alpha-1 Foundation Executive Staff
Scott Santarella, President & Chief Executive Officer
Andrew A. Wilson, MD, Scientific Director
Robert A. Sandhaus, MD, PhD, FCCP, Clinical Director
Jeanine D’Armiento, MD, PhD, Medical Liaison
Alpha-1-To-One Editorial Board
Erin Carr+; Catriona Garry*; Jon Hagstrom*; Andrew A. Wilson, MD; William J. Martin II, MD; Robert A. Sandhaus, MD, PhD, FCCP; Patricia Tew*; Bruce Trapnell, MS, MD
Managing Editor
Jeanne Kushner
The impact A1F has in advancing the cure for Alpha 1 is unparalleled, and this is highlighted in A1F’s current Impact Report, featured in this special issue of the Alpha 1 To One Magazine. This abridged version of the document can be found on page 6 and is also available in its entirety at alpha1.org. The investments that we have made, particularly over the last five years, in providing resources to build scientific infrastructure dedicated exclusively to Alpha 1 research, solidify A1F’s position as the global leader in funding Alpha 1 specific research projects. A1F has provided seed funding of over $100 million since inception and leveraged those dollars by nearly a factor of 8, resulting in more than $750 million in ancillary funding invested in Alpha 1 research.
We continue to advance the field on behalf of patients and their families by expediting discovery and approval of new therapies and treatments. It’s why the triad of the AIF Clinical Resource Center (CRC) network, A1F Research Registry, and A1F Therapeutic Development Network (TDN) are essential to achieving our mission. All three of these A1F created and supported initiatives help expedite clinical trials and hasten our mission to find a cure.
We were excited to launch the A1F Fred C. Walsh Speaker Bureau in December with a two day training (page 12). This incredible group of volunteers from all over the United States represents all segments of the Alpha 1 community, including individuals affected by lung and liver conditions, Alpha 1 kids, transplant recipients, and caregivers.
They will share their insights and experiences throughout the year with diverse audiences. Named for the late Fred C. Walsh, an active Alpha 1 patient voice on A1F’s Board of Directors as the Vice Chair and an AlphaNet Coordinator, Fred served as a key figure in the Alpha 1 community. We could not be prouder to have this fitting legacy to Fred, the quintessential patient advocate.
Congratulations to the educational scholarship recipients (page 28). We applaud the outstanding achievements of these young adults in pursuing their higher education. Be sure to learn more about how to apply for this year’s awards on page 30.
A1F is committed to making a cure a reality. We hope you will join us, support our efforts through increased and highly motivated engagement, donations, and the continuation of sharing your Alpha 1 stories. Where we are headed, we go together, and I am looking forward to engaging with each of you on the next part of our collective journey.
Sincerely,
Scott Santarella President & CEO Alpha 1 Foundation
Contributing Editors
Gennesis Corado; Cathey Henderson; Angela McBride; Shakira Molet; Miriam O’Day; Alexis Artiles Ojeda; Randel Plant; Linda Rodriguez; Scott Santarella
Alpha 1 To One is published by the Alpha 1 Foundation with the support of its advertisers. No part may be reproduced in any form by any means without prior written permission of A1F. The contents are not intended to provide medical advice, which should be obtained directly from a physician. A1F is not responsible for the accuracy of information expressed in advertisements in this publication.
Advertising: For advertising inquiries, please contact Angela McBride, Senior Director of Corporate Relations & Strategic Alliances at (877) 228 7321 ext. 233 or amcbride@alpha1.org
Letters to the Editor: Please send letters to the editor to Jeanne Kushner, Senior Director of Communications & Policy at jkushner@alpha1.org
The Alpha-1 Foundation is committed to finding a cure for Alpha-1 Antitrypsin Deficiency (Alpha-1) and to improving the lives of people affected by Alpha-1 worldwide.
*Diagnosed with Alpha 1 Antitrypsin Deficiency. +Diagnosed family member.













2025 marked the 30th anniversary of the Alpha 1 Foundation (A1F), a significant milestone and an opportunity to reflect on where we have come from, what we have accomplished, and the work that remains to be done. The purpose of this Impact Report is to carry out those tasks and to attempt to measure how effective we have been in accomplishing our goals. Since the last report five years ago, we have made significant progress as a community. We have navigated a pandemic, resumed in person activities, and overseen a resurgence of research interest in Alpha 1 Antitrypsin Deficiency (Alpha 1).
We can now cheer for multiple new therapeutic modalities being tested in clinical trials, an enviable and unique situation among genetic diseases, and a welcome departure from prior years when there were few new treatment options on the horizon. We feel a great urgency to make the most of this opportunity, and it is with that urgency in mind that we now take stock of our prior accomplishments and look ahead.
This report documents the output of a robust and outstanding community of researchers, physicians, A1F staff, and patients working together towards a common goal. I hope you will share in the optimism that I feel when reviewing our progress and the position we now occupy as we look ahead. As always, we are guided and inspired by lessons learned from John and Fred Walsh, who taught us to be impatient in our expectations for progress and to remember to “Keep the Faith.”
—Andrew Wilson, MD A1F Scientific Director

25 years of Alpha-1 Foundation- sponsored Research (1999 to 2024)
Promoting research with the vision of finding better treatments and ultimately a cure for Alpha 1 has been a major focus of A1F for the past 25 years. Investigator initiated grants have been the driving force of A1F’s research program since its inception in 1999, based on the time tested principle in biomedical discovery that innovative ideas typically arise in the research community. Between 1999 and 2024, over $100 million has been invested in research. Of those funds, approximately 50% was directed at the peer reviewed investigator initiated grants program, while the remainder was used to organize and publish the proceedings of scientific conferences, and to support the DNA & Tissue Bank and the Alpha 1 Research Registry, along with other programs as resources for basic and clinical researchers.
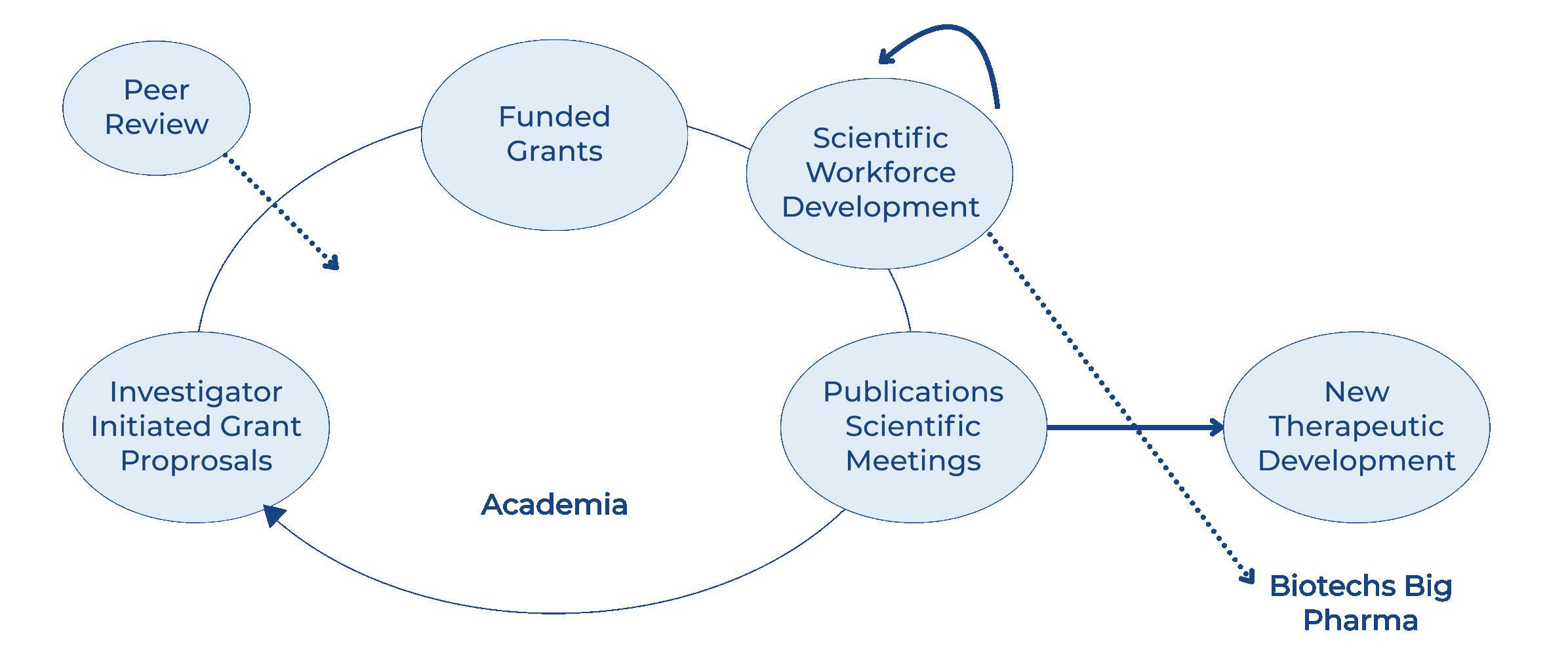
A1F sponsored research has had a significant impact on our understanding of the mechanisms leading to the clinical manifestations of Alpha 1, and most of this information has been obtained through investigations supported by investigator initiated grants (Figure 1).
The program is based on the concept that investigator initiated research leads to scientific publications and is the basis of scientific meetings, which facilitate cross pollination, amplifying the impact of new findings and informing the biotech and pharmaceutical companies in their quest for new therapeutic solutions. A1F support has the additional benefit of attracting and retaining the scientific workforce to focus on Alpha 1. This
document aims to summarize the scientific contributions of this research from 1999 to 2024.
Applicants come from a variety of institutions and countries: Cutting edge research that advances the field can happen in many locations around the world. To leverage worldwide expertise in the service of Alpha 1 patients, A1F receives and funds research proposals from many different countries, representing many institutions.
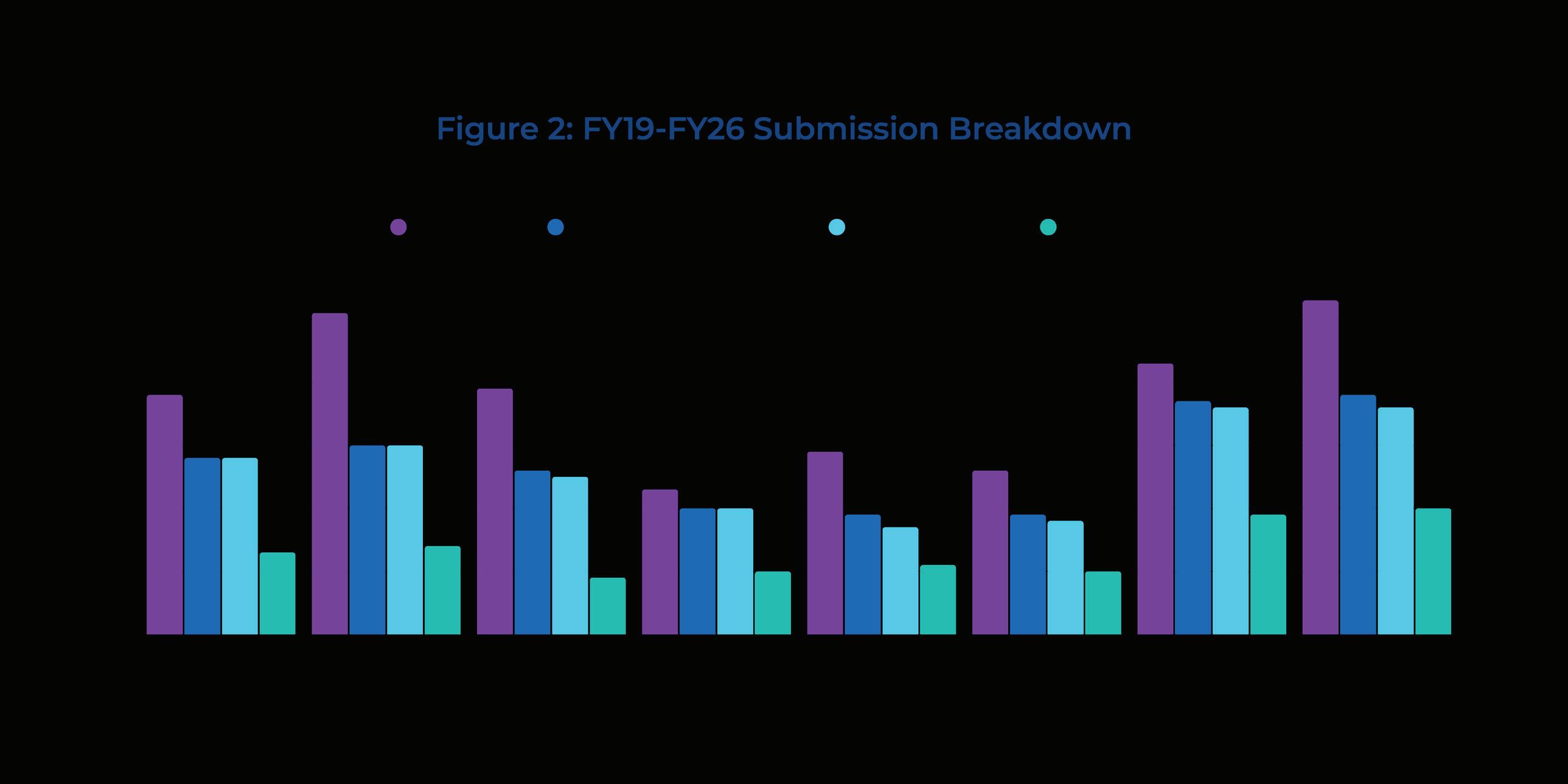
Applications for A1F funding are increasing. During the COVID 19 pandemic, applications for funding declined as research became more challenging to conduct and the time of physician researchers was occupied with patient care. Applications for funding were at their lowest in 2020 and remained low for several years but have subsequently rebounded, as indicated in (Figure 2). For the 2024 2025 cycle, we received the highest number of letters of intent (LOIs) to submit a grant proposal in the past 10 years.
A1F has developed a rigorous process, modeled on the National Institutes of Health (NIH), to vet grant applications and ensure that our funds support the highest quality research. It is important that we measure the impact of A1F support to validate the efficacy of this approach. We can measure this impact in a variety of ways, including the metrics below:
• NIH grant support resulting from A1F funding: NIH is historically the largest funder of biomedical research in the world. We encourage A1F funded researchers to build on the findings they make with A1F support to apply for additional
million in investigator initiated proposals from 1999 2025 has subsequently resulted in $732.8 million in funds from the federal government to our awardees, a return on investment of approximately $15 per dollar awarded.
• Publications resulting from A1F funding: Dissemination of research findings is critical to advance scientific knowledge. A1F funded investigators are strongly encouraged to publish findings they make with A1F support. From 2018 2025, A1F funded investigators published 153 articles in scientific journals on all aspects of Alpha 1.
• Patents resulting from A1F funding: Research findings have the potential to identify mechanisms or develop techniques with potential therapeutic value. Patent activity is thus an additional measure of impact. Between 2001 2025, A1F funded investigators have filed 48 patent applications.
A key aspect of our mission is to develop new treatments and ultimately a cure for patients with Alpha 1. In the 5 years since the last impact report, the Alpha 1 therapeutic landscape has changed dramatically, with numerous drugs targeting a variety of mechanisms advancing to preclinical development or clinical trials. This evolution reflects a combination of increased understanding of cellular disease mechanisms together with technological advances, particularly in the ability to manipulate gene architecture and expression through a variety of approaches. Our community is fortunate that seminal advances in genetic targeting (collectively including Nobel prize winning discoveries such as RNA interference and CRISPR, together with gene therapy and RNA editing) that can be applied to many genetic diseases are potentially therapeutically useful in Alpha 1. The development of small molecules to assist in proper folding or stabilize
promise as a novel class of potential therapeutics. Finally, alternative delivery approaches, dosing, and recombinant forms of the Alpha 1 protein are in ongoing testing and could result in improved efficacy and quality of life for patients.
The rapid progress made in the application of emerging therapies to Alpha 1 is in no small part the result of A1F funding. A1F support laid out the groundwork that made it possible to identify approaches that were likely to succeed, demonstrate efficacy in preclinical models, and is enabling their successful testing and application for patients suffering from Alpha 1. Over a 25 year period, A1F funded researchers have established a broad understanding of the disease, ranging from basic disease mechanisms to the clinical experience of affected patients.
This knowledge base is now being applied in many ways for trial design and implementation, from identifying which patients with Alpha 1 are most likely to benefit from specific therapeutic approaches to determining how to measure the success of drugs in development.
In addition to direct funding of research in the form of grant support, A1F has invested significantly in infrastructure designed to facilitate and promote the success of the aforementioned clinical trials. This support includes:
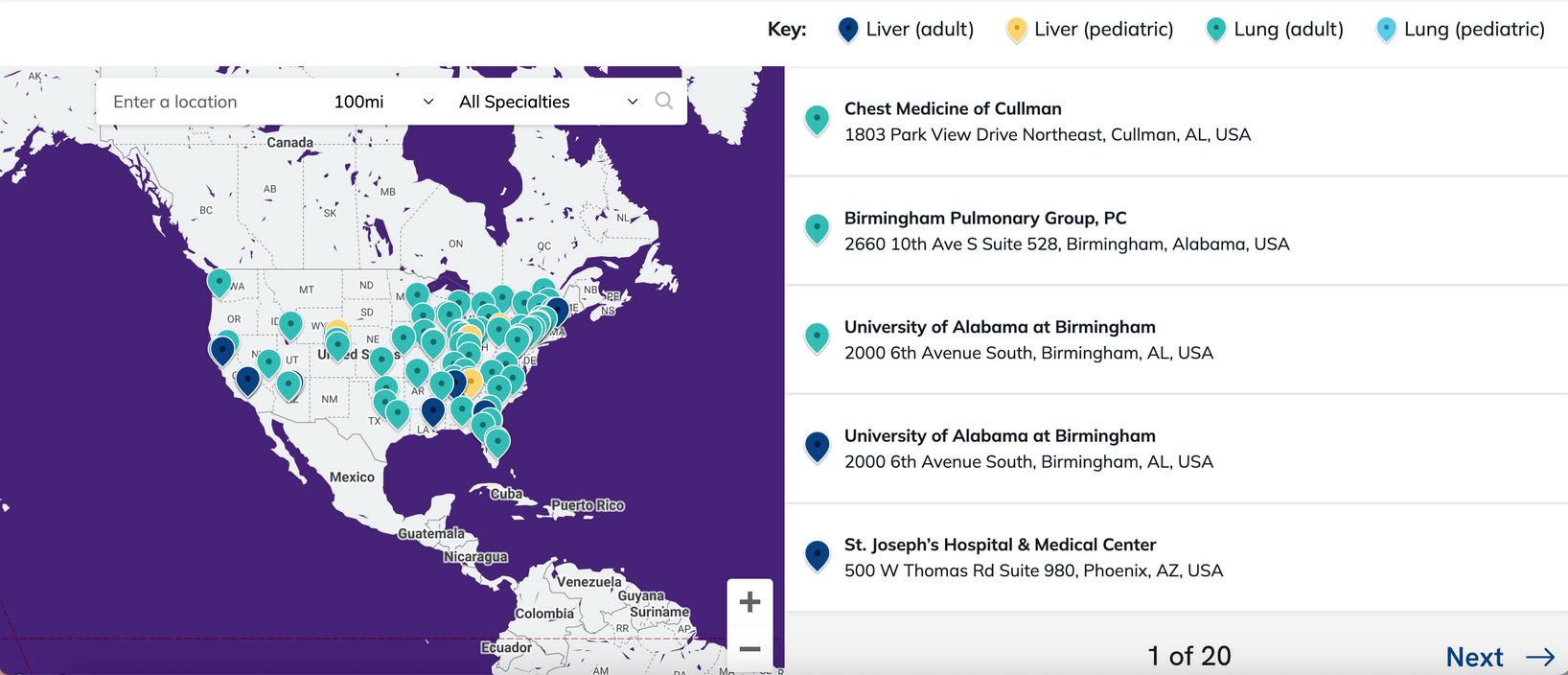
• The Alpha 1 Research Registry: The Alpha 1 Research Registry includes data from more than 4,000 individuals including >1,300 with severe Alpha 1. In addition to its utility as a tool for investigators to address questions of natural history, the registry helps to connect patients with clinical studies that they may qualify for based on their demographic and clinical characteristics. Registry staff have been successful in helping to recruit Alphas to participate in ongoing clinical trials of emerging therapeutics.
• The Therapeutic Development Network (TDN): To identify patients interested in participating in clinical trials and who live in proximity to clinical trial sites as a complement to the Alpha 1 Research Registry, the A1F created the TDN. The TDN consists of 20 centers across the US that have agreed to participate in upcoming clinical trials. Each site enrolls local patients and collects a set of data about each enrolled participant that includes the basic inclusion and exclusion criteria for most clinical trials. The resulting database is then a resource for identifying potential clinical trial participants across the country.
• The Alpha 1 Biomarkers Consortium (A1BC): In order to design and execute clinical trials, industry partners need biomarkers that are established and recognized by the FDA to correlate with disease severity and progression. The A1BC is a consortium of A1F Clinical Resource Centers (CRCs) funded by the NIH and A1F that has enrolled > 270 severely deficient (ZZ) patients. These centers are collecting longitudinal data and biosamples from this cohort to develop the biomarkers that can then be used as clinical trial endpoints.
• The DNA and Tissue Bank: Established in 2001, the bank includes a data repository together with plasma samples and genomic DNA from 2,849 participants, including 812 ZZ, 501 MZ, 88 SZ, 115 MS, and 342 rare alleles. This resource provides the ability to connect DNA sequences with disease phenotypes and clinical characteristics and is a tool for addressing research questions about the genetic basis of Alpha 1.
As we look to the future, we see many reasons for optimism. Much of this optimism relates to the clinical trials that have recently started and are anticipated to start in the upcoming 24 months. To ensure that the promise of this moment is realized, A1F will take the following actions in the next 5 years:
• Facilitate the success of clinical trials: To ensure the success of clinical trials, we will take several key steps: First, we will continue to support the A1BC and its efforts to identify radiologic and serum biomarkers associated with specific disease states that can similarly inform clinical trials. Second, we will continue to invest in the Alpha 1 Research Registry and the TDN. Finally, we will continue our partnership with the Critical Path Institute and our industry partners to establish endpoints that the FDA will embrace in clinical trials.
• Increase detection of Alphas: It is estimated that only 15% of severely deficient patients
with Alpha 1 have been identified. It is critical that we increase the detection of Alphas both to facilitate clinical trial enrollment and to identify severely deficient patients earlier in the course of disease when they are more likely to benefit from treatment. To increase detection, we will continue our recent investment in AlphaDetect, a new non profit subsidiary of A1F, that will lead a transformative initiative that unifies all community stakeholders – patients, healthcare providers, advocates, and industry partners – with a collective common purpose: to ensure no Alpha 1 patient is left undetected.
• Invest in research infrastructure: By developing tools that facilitate the ability of researchers to ask questions, we can increase their ability to make discoveries that will benefit Alphas in the years to come. To do so, we will continue to build the natural history component of the Alpha 1 Research Registry to enhance its value for research purposes. We will likewise continue to build genetic data capabilities to facilitate the ability to connect genotypes to phenotypes and continue to invest in human capital through support for the careers of early stage investigators to sustain a pipeline of scientific expertise in Alpha 1. Finally, we will continue to fund cutting edge research that will produce discoveries that will enable the development of the next generation of treatments for Alpha 1.
• Increase CRC availability to patients: To get the best care available, patients need access to physicians and allied health professionals with Alpha 1 expertise. To increase the availability of such expertise, we will continue to identify and cultivate physicians with Alpha 1 expertise, particularly those specializing in pediatric gastroenterology and adult gastroenterology. We will specifically work to increase the number of Clinical Resource Centers in areas of the country that are currently less well represented.
In summary, A1F is working on many fronts and will continue to do so to accomplish the best possible outcomes for Alphas to achieve our mission of finding a cure for Alpha 1. 1 ALPHA-1 FOU NDATION

Your legacy can support the Alpha-1 Foundation (A1F), not just today, but for years to come.
Many A1F supporters have included a gift in their estate plans to make a lasting impact at no cost today.
To learn more about planned giving and leaving a legacy, visit: alpha1.org/planned-giving/
For more information about joining the Alpha-1 Legacy Society, visit: alpha1.org/alpha-1-legacy-society/
For additional information or to discuss planned gifts to A1F, please contact Angela McBride at amcbride@alpha1.org or call 1-877-228-7321, ext. 233.




Empowering Voices.
Inspiring Action.
Elevating Patient-Centered Work.
On December 5th 6th, 2025 members of the A1F Fred C. Walsh Speaker Bureau joined in Charleston, South Carolina for a two day training session. Alphas and family members from around the country came together to share their Alpha 1 journeys, to be a voice and a representative for the Alpha 1 community. At the informative training, speakers learned how to share their story with
clarity and impact in a way that will resonate, educate, and inspire others. During this coaching session, each speaker was given the opportunity to publicly share their story and work on fine tuning and tailoring messaging for future live speaking engagements.
This incredible group of volunteers from all over the United States represents all segments of the Alpha 1 community, including lung affected, liver affected, Alpha 1 kids, caregivers, and transplant recipients. They will share their insights and experiences throughout the year with a diverse audience including:

• Patients, caregivers and families at community events, educational sessions, and support groups who need encouragement and connection.
• Medical professionals attending conferences, hospital training sessions, and grand rounds to help them gain firsthand insights into what truly matters to the people they treat.
• Industry leaders and researchers at meetings, advisory boards, and innovation summits that are seeking to understand patient needs as they develop treatments and therapies and guide them to prioritize patient centric solutions.
• Government officials and agencies during policy discussions and awareness initiatives that shape policies impacting equity, quality, and access to better care.
• At plasma and collection center events, emphasizing the impact of lifesaving donations on Alpha 1 patients whose therapeutic treatments rely on them.
Their presence in these spaces ensures that the patient’s perspective is not only heard but
prioritized with the power to inspire hope, drive understanding, and spark change . The patient’s voice represents the lived experience that no statistic or clinical study can fully capture. This group of incredible patient speakers brings humanity to healthcare and ensures that decisions, treatments, and innovations are guided by those who experience them. The goal is to shape perspectives, influence decisions, and be a beacon of advocacy and education.
The A1F Fred C. Walsh Speaker Bureau is a new A1F program that serves to empower Alphas to be the voice of the Alpha 1 community. Named for the late Fred Walsh, an active Alpha 1 patient voice on A1F’s Board of Directors as the Vice Chair and an AlphaNet Coordinator, Fred served as a key figure in the Alpha 1 community. Since the establishment of both A1F and AlphaNet in 1995 by his twin brother John W. Walsh, Susan Stanley and Sandy Lindsey, Fred dedicated his life to supporting and educating his fellow Alphas. His belief in patient empowerment resonated deeply,



and he championed the idea that individuals must take an active role in their health and advocate for themselves. Fred remained hopeful for the continued growth and success of the Alpha 1 community, envisioning it as a beacon of hope for patients everywhere. His legacy, rooted in compassion, advocacy, and community, will undoubtedly inspire future generations of Alphas to connect, support, and strive for a brighter future.
To learn more about the A1F Fred C. Walsh Speaker Bureau and meet the speakers, visit https://alpha1.org/a1f fred c walsh speaker bureau/ or contact Chelsea Boring at cboring@alpha1.org



The 28th Gordon L. Snider (GLS) Critical Issues Workshop entitled, “AAT Polymers: Disease Pathogenesis, Detection, and Therapeutic Targeting” took place on Friday, October 3, 2025, in London, England. The overall goal of this workshop, chaired by Dr. Andrew Wilson and Dr. David Lomas, was to focus on the alpha 1 antitrypsin (AAT) polymer, the structure that is potentiated when AAT monomers misfold, and that is a significant
contributor to clinical disease in Alpha 1 Antitrypsin Deficiency (Alpha 1).
A1F welcomed 80 attendees from around the world who joined the workshop to contribute their Alpha 1 expertise on this topic. The goal of GLS topical workshops is to provide new information that speeds the journey to a cure for Alpha 1.
I was delighted with the success of the workshop which provided a platform to discuss the role of antitrypsin polymers in many aspects of Alpha-1 Antitrypsin Deficiency: their role in the pathogenesis of the liver and lung disease, as a biomarker of liver disease, and as a marker of the response to therapies that target the liver. I am hopeful that the sharing of information will stimulate new developments in this exciting field of research.
Dr. David Lomas, workshop co-chair
We are now fortunate to be entering a period in which multiple distinct therapeutic approaches are being developed and tested in patients for the first time. This GLS workshop will therefore survey the field, reviewing what is known about polymer structure and formation, how we can detect and quantify AAT polymers, what is known about their presence in organs affected in Alpha 1, how they contribute to cellular dysfunction, their prognostic significance in disease, how cells compensate to degrade polymers, and how we can contemplate targeting polymers as a therapeutic intervention.
“It has been nearly 40 years since David Lomas and his colleagues found that the “Z” mutation in alpha 1 antitrypsin facilitates the formation of AAT polymers. Since that time, we have learned that AAT polymers play a central role in cellular dysfunction and tissue injury that leads to disease in Alpha 1. The success of emerging therapeutic approaches now in development will likely hinge upon their ability to alter the formation, clearance, or destructive potential of AAT polymers, making it a good time to revisit the molecular basis of the disease. I was honored to plan this meeting with Dr. Lomas and to hear from the outstanding speakers about polymer structure and formation, how we can detect and quantify AAT polymers, what is known about their presence in organs affected in Alpha 1, their prognostic significance in disease,
how cells compensate to degrade polymers, and how we can contemplate targeting polymers as a therapeutic intervention. I am hopeful that data and discussion from the meeting will help to focus our priorities, as we work to enable the development of treatments and a cure for Alpha 1 patients,” said Dr. Andrew Wilson, A1F Scientific Director and workshop co chair.
A1F aims to accomplish the following actionable items from this robust workshop:
(i) to establish a standard protocol for measuring polymers in cohorts and clinical studies.
(ii) to share academic and industry data in a precompetitive space to assess whether we can validate circulating polymers as a biomarker for liver disease and response to therapy in alpha 1 antitrypsin deficiency.
(iii) to evaluate whether the measurement of circulating polymers is useful in detecting individuals with antitrypsin deficiency. This will be done with AlphaDetect, a new non profit subsidiary of A1F, to lead a centralized effort to identify individuals with Alpha 1.
An understanding of the mechanisms of antitrypsin polymerization is critical as it is underpins the accumulation of mutant protein as inclusions within the liver. It was this understanding that enabled the recognition that polymers may play a role in the associated emphysema, that their level in the circulation may predict individuals at risk of liver disease, and that their suppression forms the basis for novel therapies to treat the cirrhosis associated with Alpha 1.
“A1F was proud and honored to hold our first GLS conference in Europe. AAT polymers, as a topic, represent the innovative spirit of Alpha 1 Researchers and Scientists and their commitment to advancing the field on behalf of patients,” – Scott Santarella, A1F President and CEO.
A1F extends its gratitude to the workshop’s co chairs, Dr. Andrew Wilson and Professor David Lomas, for their invaluable contributions to shaping the meeting program and for inviting and securing the participation of this esteemed group of experts.
We are deeply grateful to the 28th GLS Critical Workshop sponsors AlphaNet, CSL, Grifols, Takeda, and Vertex Pharmaceuticals. 1 ALPHA-1 FOU NDATION

The role of the Alpha 1 Foundation (A1F) as a global leader in the Alpha 1 Antitrypsin Deficiency (Alpha 1) community was highlighted recently at an Alpha 1 Awareness Month Recognition Forum held in conjunction with European Parliament on November 20, in Brussels, Belgium.
In collaboration with the RPP Group and in partnership with the Alpha 1 Europe Alliance, A1F seized on an opportunity to build momentum for rare diseases, and specifically the Alpha 1 community as the EU unlocks key opportunities associated with the upcoming Biotech Act and their ongoing implementation of the Substances of Human Origin (SoHO) Regulation. A1F recognized the time is now to push for fair access to plasma derived therapies and better care for all rare disease patients.
A1F presented to EU Parliament: its longstanding and considerable investment in funding researchers at institutions based within the European Union. This includes our unwavering commitment to educating European Alphas through our Global Patient Congress and Research Conference which took place this past April in Lisbon, Portugal. Additionally, we highlighted our proactive our engagement with EU Parliamentarians and patients through our recent social media video awareness campaign, further strengthening our collaborative efforts.
While advances in medicine and European cross border collaboration have occurred, thousands of people living with Alpha 1 still face delayed diagnosis, limited treatment options, fragmented care pathways, and significant inequities in access to treatment among different member states. The disparities across European health systems remain profound.
A1F’s engagement with EU policymaking is core to advocacy efforts in Europe because the European Union sets up the overall strategic and regulatory framework for rare diseases in Europe. While health remains a national competence, the EU’s complementary role is indispensable for advancing rare disease care. By providing the necessary scale, coordination, and resources, the EU helps address key challenges such as the low prevalence of rare diseases, fragmented expertise across member states, and limited national resources.
A1F is proud of the global leadership role we have played for 30 years. We applaud and support the dedicated action plan presented by the Alpha-1 Europe Alliance to ensure equitable access to diagnosis, care, and treatment for all Alpha-1 patients across Europe. The promotion of clinical guidelines, awareness campaigns, research initiatives, and collaboration with healthcare stakeholders to reduce inequalities and strengthen care pathways are at the core of A1F’s mission, and we are committed to helping the Alpha-1 Europe Alliance and their 5-year strategic framework “Strategy 2030” for the Alpha-1 global community.
Scott Santarella A1F President & CEO
EU initiatives play a pivotal role in shaping how Member States develop and implement their national health systems for rare diseases. Key examples include the Clinical Trials Regulation (CTR), which has streamlined and harmonized the authorization process for multinational clinical trials, making Europe more attractive for research and accelerating patient access to innovative medicines. The EU also actively supports national rare disease strategies through initiatives like the Orphan Medicinal Products Regulation, the Cross Border Healthcare Directive, and the Pharmaceutical Package, alongside recommendations on screening and rare disease planning. Crucially, the establishment of European Reference Networks (ERNs), such as ERN LUNG, directly addresses fragmented expertise by facilitating cross border consultations and knowledge sharing, significantly improving diagnostic rates and treatment pathways for patients with complex conditions like Alpha 1.
Furthermore, the SoHO Regulation ensures high standards of safety and quality for therapeutic substances, facilitating their safe cross border exchange.
Looking ahead, the forthcoming Biotech Act represents a major strategic advance, aiming to establish a clear, predictable, and innovative regulatory framework to stimulate the development of new therapies, facilitate patient access, and strengthen Europe’s leadership in medical research.


Overall, when Alpha 1 is represented in EU debates, it directly influences the long term structures and legislative frameworks underpinning national decisions.
Watch the meeting here: https://alpha1.org/video library/alpha 1 awareness month event 2025 recording/
The second Latin American Alfa 1 Meeting titled “Consolidando avances, ampliando horizontes”, (“Consolidating Advances, Expanding Horizons”) was held on December 10, 2025, in Buenos wAires, Argentina.
Organized by Alfa 1 Argentina and supported by A1F, the meeting aimed to bring together specialists, healthcare professionals, and representatives from
patient associations from across Latin America to exchange experiences, share scientific advances, and discuss strategies for addressing Alpha 1.
Throughout the day, experts including pulmonologists, hepatologists, psychologists, physiotherapists, nurses, and nutritionists shared their knowledge and experience to shed light on the latest developments in treatments, research, and standards of care for Alpha 1.
A1F’s Global Program works tirelessly with patients and stakeholders to increase awareness, detection, and access to care for the Alphas around the world.
To learn more about the Alpha 1 Global program, please contact Nadine Nuchovich, Clinical Research & Global Outreach Manager at nnuchovich@alpha1.org 1 ALPHA-1 FOU NDATION

For patients with emphysema caused by severe Alpha-1 antitrypsin deficiency. Neither are their needs. No two alphas are alike.
GLASSIA ® offers the most infusion setting options, including at-home infusion, so you and your doctor can choose what’s best for you.
Available in 1-, 4-, and 5-gram vials.

AT INFUSION CENTER AT CLINIC AT HOME SELF-INFUSION* AT HOME WITH NURSE
*If self-infusion is deemed appropriate, ensure that you receive detailed instructions and adequate training on how to infuse at home or other appropriate setting and have demonstrated the ability to independently administer GLASSIA.
Scan the code or visit glassialiquid.com to learn about which infusion setting option may be right for you.
What is GLASSIA?
GLASSIA is a medicine containing human Alpha₁-Proteinase
Inhibitor (Alpha₁-PI) that is used to treat adults with lung disease (emphysema) because of severe Alpha₁-antitrypsin (Alpha₁) deficiency. GLASSIA is not meant to be used as a therapy for lung disease other than severe Alpha₁ deficiency. Effects of GLASSIA on worsening lung function and emphysema progression have not been proven in clinical trials. Long-term effects of Alpha₁ replacement and maintenance therapy have not been studied.
IMPORTANT SAFETY INFORMATION
What is the most important information I need to know about GLASSIA?
• GLASSIA can cause severe allergic reactions including hives, swelling in the mouth or throat, itching, tightness in the chest, trouble breathing, wheezing, faintness or low blood pressure
• If you will be taking GLASSIA outside a healthcare setting, ask your healthcare provider (HCP) about an epinephrine pen and/or other supportive care for certain severe allergic reactions.
Who should not use GLASSIA?
Do not use GLASSIA if you:
• Have immunoglobulin A (IgA) deficiency with antibodies to IgA
• Have a severe allergic reaction to human Alpha₁-PI products.
When you’re prescribed GLASSIA, Takeda Patient Support is here for you. For more information, visit TakedaPatientSupport.com
IMPORTANT SAFETY INFORMATION (continued)
What are the possible or reasonably likely side effects of GLASSIA?
If any of the following problems occur contact your healthcare provider (HCP) or call emergency services right away:
• Worsening or flare-up of your chronic obstructive pulmonary disease (COPD)
• Hives, swelling in the mouth or throat, itching, chest tightness, trouble breathing, wheezing, fainting or dizziness. These could be signs of a serious allergic reaction.
The most common side effects that may occur are headache and upper respiratory tract infections.
Other possible side effects of GLASSIA include:
• Cough
• Sinus infection
• Chest discomfort
• Dizziness
• Increased liver enzymes
• Shortness of breath
• Nausea
• Fatigue
These are not all the possible side effects. Tell your HCP about any side effect that bothers you or that does not go away. You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.
Please see the Important Facts About GLASSIA on the next page.
©2025 Takeda
GLASSIA®
(Alpha1-Proteinase Inhibitor (Human)) Injection, For Intravenous Use
The following summarizes important information about GLASSIA (pronounced glass-see-ă). Please read it carefully before using this medicine. This information does not take the place of talking with your healthcare professional, and it does not include all of the important information about GLASSIA. If you have any questions after reading this, ask your healthcare professional.
What is GLASSIA?
GLASSIA is a liquid medicine containing human Alpha1Proteinase Inhibitor (Alpha1-PI) also known as alpha1antitrypsin (AAT), which is purified from human blood. The main purpose of infusing GLASSIA is to increase the levels of the AAT protein in your blood and lungs. AAT protein protects the lung tissue by blocking certain enzyme-caused damage. Such damage can lead to severe lung disease, such as emphysema.
Limitations of Use:
• The effects of increasing the AAT protein levels with GLASSIA or any other Alpha1-PI product on worsening pulmonary function and progression of emphysema have not been proven in clinical trials.
• The long-term effects of AAT replacement and maintenance therapy with GLASSIA have not been studied.
• GLASSIA is not intended as a therapy in individuals with lung disease other than severe Alpha1-PI deficiency. Who should not take GLASSIA?
You should not use GLASSIA if you:
• Have immunoglobulin A (IgA) deficiency with antibodies to IgA
• Have had a severe allergic reaction to human Alpha1-PI products
What is the most important information that I should know about GLASSIA?
Severe allergic reactions can occur with GLASSIA. Your doctor will inform you about signs of allergic reactions which include hives, swelling in the mouth or throat, itching, tightness in the chest, trouble breathing, wheezing, faintness, low blood pressure, or serious allergic reaction. If you have any of these reactions, discontinue use of the product and contact your physician and/or seek immediate emergency care, depending on the severity of the reaction. If you or your caregiver will be administering GLASSIA outside a healthcare setting, ask your doctor about an epinephrine pen and/or other supportive care for certain severe allergic reactions. Ask your doctor to make sure you receive training on how and when to use any prescribed supportive care medicine and keep it close at hand when administering GLASSIA.
How should I take GLASSIA?
• GLASSIA is given directly into the bloodstream.
• You can get GLASSIA at your healthcare professional’s office, clinic, hospital, or delivered directly to your home by a healthcare professional from a limited network of specialty pharmacy providers.
• Your healthcare professional will decide if self-infusion in your home is right for you. You should be trained on how to do infusions by your healthcare professional.
What should I tell my healthcare professional before I start using GLASSIA?
Before starting GLASSIA, tell your healthcare professional if you:
• Have IgA deficiency with antibodies to IgA.
• Have a history of severe allergic reactions to Alpha1-PI products.
What are the possible or reasonably likely side effects of GLASSIA?
• A possible side effect to GLASSIA is worsening or flare-up of your chronic obstructive pulmonary disease (COPD) in which your breathing gets worse than usual.
• Call your healthcare professional or go to your emergency department right away if you get: Hives, swelling in the mouth or throat, itching, chest tightness, trouble breathing, wheezing, fainting or dizziness. These could be signs of a serious allergic reaction.
• The most common side effects are headache and upper respiratory tract infections. Other possible side effects of GLASSIA include: cough, sinus infection, chest discomfort, dizziness, increased liver enzymes, shortness of breath, nausea, and fatigue.
These are not all of the possible side effects for GLASSIA. You can ask your healthcare professional for information that is provided to healthcare professionals. Talk to your healthcare professional about any side effects that bother you or that don’t go away.
How do I store GLASSIA?
Store GLASSIA refrigerated or at room temperature.
• You can store GLASSIA in the refrigerator (36°F to 46°F [2°C to 8°C]). Do not freeze.
• You can store GLASSIA at room temperature (up to 77°F [25°C]) for up to one month. You must use GLASSIA within one month once you remove it from the refrigerator. Do not re-refrigerate GLASSIA once the product has been stored at room temperature.
• Keep the GLASSIA vial in the box until you are ready to administer the product.
Check the expiration date on the carton and vial label. Do not use GLASSIA after the expiration date.
Manufactured by:
Takeda Pharmaceuticals U.S.A., Inc. Cambridge, MA 02142 USA
U.S. License No. 1898
GLASSIA® is a registered trademark of Kamada Ltd., used under license. Takeda and are registered trademarks of Takeda Pharmaceutical Company Limited.

©2025 Takeda Pharmaceutical Company Limited. All rights reserved. Revised: February 2025
GLA378 US-GLA-0568v1.0 03/25
We are recruiting for Alpha-1 related studies and need your participation!
We encourage you and your family members to join the Alpha-1 Research Registry to help advance Alpha-1 research, diagnosis, and treatment
Anyone diagnosed with Alpha-1 can join the Registry in three steps:
1. Complete a Registration Form
2 Sign the Informed Consent
3. Complete the Questionnaire
To enroll or for more information:



alpha1.org/join-the-alpha-1-research-registry











LEARN MORE TODAY! Visit alpha1.org/national-conference for more information and to register. For questions, call (877) 228-7321, ext. 323.
STAY
rates • Central location • Easy access
CONFERENCE AT A GLANCE
General Sessions • Tracks • Panels • Community • Networking
KEY DATES
Registration • Early bird • Conference days
OXYGEN TRAVEL FUND
Supporting access through travel assistance
AGENDA & FAQS
Schedules • Travel info • Planning details


November 2025 marked a defining chapter for the Alpha 1 Foundation (A1F) as the Alpha 1 community came together to celebrate Alpha 1 Awareness Month and A1F’s 30th anniversary. Over 30 days, shared stories, scientific milestones, fundraising, and advocacy reflected three decades of progress and a renewed commitment to what comes next.
This year’s campaign centered on four guiding pillars: INSIGHT, INNOVATION, INVESTMENT, and IMPACT. Each week highlighted a different pillar, weaving together research, education, community engagement, and advocacy into one coordinated national effort powered by patients, caregivers, clinicians, Alpha 1 support group leaders, industry partners, and advocates.
At the heart of the awareness campaign was the 2025 A1F Virtual Walk, A1F’s signature Building Friends for a Cure (BFC) fundraising initiative, where participants walked all over the United States for themselves, loved ones, and in memory of those lost.
By the end of November, the A1F Virtual Walk raised more than $91,000 with 82 teams participating nationwide, making it the most successful virtual walk campaign in A1F history.
Momentum built early in the month as nearly 40 walkers, team captains, and community leaders gathered for the A1F Virtual Walk Kickoff Zoom. Throughout the month, social media filled with Alpha 1 facts, personal stories, and calls to action
supported by A1F’s Digital Awareness Toolkit and unified by #Alpha1Awareness and #A1FVW25.
Teams brought Alpha 1 Awareness Month to life in meaningful and personal ways. In Nebraska, the Big Red Alphas accepted an official Alpha 1 Awareness Month proclamation from Governor Jim Pillen while celebrating growing bipartisan support for the John W. Walsh Alpha 1 Home Infusion Act (H.R. 2343), which reached 40 congressional co sponsors during the month.
In Nevada, the Battle Born Alphas, led by Norma Leising, joined a Sparks parade, distributing 500 Alpha 1 flyers and sharing a route wide awareness announcement in memory of Norma’s late husband, Steve Faucett.
Long standing teams returned alongside first time walkers. Team Harrison continued nearly a decade of walking together in Kentucky. Team Mitch, led by Lori and Dave Mitcheroney, turned a coastal Massachusetts route into a community fundraiser. In Massachusetts, Karin Pittsley hosted her first walk, while in Minnesota, newly diagnosed Daniel Ross became one of the top individual fundraisers after publicly sharing his Alpha 1 journey.
Families walked across generations, from Team Steve in Louisiana, marking ten years of honoring a loved one’s memory, to Richie Rocks Alpha 1 in Brooklyn, where 9 year old Richie celebrated the fifth anniversary of his life saving liver transplant as his entire school wore purple in support.



Mid month, attention turned to scientific progress. A1F continued strengthening the connection between patients and research through its Therapeutic Development Network (TDN) and Alpha 1 Research Registry, prioritizing individuals ready to participate in clinical trials.
At the annual American Association for the Study of Liver Diseases (AASLD) Liver Meeting in Washington, DC, A1F engaged researchers and clinicians from around the world and hosted its annual Liver Grant Awards Reception, recognizing 21 in cycle liver grant awardees.
In total, A1F awarded more than $3.75 million in 2025 in cycle grants, with 64% focused on lung research and 36% on liver research, reflecting A1F’s comprehensive, patient centered approach to Alpha 1 science.
Awareness Month also advanced public policy and advocacy priorities. Supporters nationwide acted on:
• Permanent Medicare coverage for home infusion therapy through the John W. Walsh Alpha 1 Home Infusion Act (H.R. 2343)
• Improved access to supplemental and liquid oxygen through the Supplemental Oxygen Access Reform (SOAR) Act (H.R. 2902)
In New York, longtime advocate Richard Lovrich helped secure a New York State Senate Resolution recognizing Alpha 1 Awareness Month, presented by Senator Jim Tedisco and acknowledged by Governor Kathy Hochul and other state and federal leaders.

Internationally, A1F participated in the European Alliance Alpha 1 Awareness Month event at the European Parliament, where discussions on rare disease policy and access reinforced global alignment around Alpha 1 care.
Industry partners amplified Awareness Month efforts throughout November. Grifols led a nationwide walking challenge with 90+ participants. Takeda, CSL, Beam Therapeutics, and Sanofi expanded Alpha 1 awareness through employee engagement, social media outreach, and community participation, emphasizing the power of collaboration in advancing Alpha 1 care.
Thirty years in, the Alpha 1 community is not slowing down. With deeper insight, bolder innovation, targeted investment, and measurable impact, November 2025 reflected both how far the community has come, and how much possibility lies ahead. The steps taken, stories shared, funds raised, and policies advanced this month will continue shaping the future as A1F works toward earlier diagnosis, better treatments, stronger access to care, and one day, a cure.
To learn more about how to get involved, visit alpha1.org 1 ALPHA-1 FOU
Each year the Alpha-1 Foundation (A1F) awards Alpha-1 Educational Scholarships to Alphas and family members seeking to further their higher education. This year, ten scholarships were awarded to deserving individuals. Congratulations to these outstanding young adults.
Kali Cook and Andrew Flickenschild were awarded the James “Jim” Quill Memorial Scholarships. These scholarships honor the memory of Jim Quill. Jim Served as an AlphaNet Coordinator, and then Manager of AlphaNet. Additionally, he served on the A1F Board of Directors. He consistently solved problems for the entire Alpha 1 community. Not only was Jim the consummate mentor to all AlphaNet Coordinators and staff members, but he was always available to listen and offer counsel to any Alpha.

Kali Cook (Sterling Heights, Michigan) is a Freshman at Macomb Community College majoring in Elementary Education:
“Alpha 1 has not only taught me to be responsible but also helped me realize that I can create my own definition of fun focusing on genuine joy, good company, and a future I can look forward to with pride.”

Andrew Flickenschild (Ellington, Connecticut) is a Freshman at the University of Connecticut majoring in Biology:
“My dad’s illness and passing changed my perspective on life and made me realize how important your health is. It also gave me a strong desire to understand the human body and how I can help people who are facing similar struggles”
This year’s Peter Smith Scholarships have been awarded to four well deserving awardees, Amy Jageman, Jessalyn Rospert, John Dyer, and Alexus Calhoun. Peter Smith wrote, edited, and published Alpha 1 News from 1989 1992. Before his early death due to Alpha 1, he was spreading the word to over 1,200 households. His efforts encouraged, enlightened, and inspired those with Alpha 1 to reach out to one another and to learn from one another. The intent of the Peter Smith Scholarship is to continue this tradition by helping those with Alpha 1 and their families to learn and achieve.

Amy Jageman (Bridgeville, Pennsylvania) is in Graduate school at La Roche University for a Doctorate of Nurse Anesthesia Practice (DNAP):
“Living with Alpha 1 Antitrypsin Deficiency has shaped my life in profound ways, teaching me resilience, the power of advocacy, and the importance of compassionate healthcare. Diagnosed at birth, Alpha 1 has never been just a medical condition to me—it has been a defining part of my journey, influencing my perspective on life, my education, and my career aspirations.”

Jessalyn Rospert (Portland, Texas) is a Freshman at Texas State University majoring in Nursing:
“I will never let my Alpha 1 diagnosis define me. Instead, I strive to do my best in everything I do, every single day. It only makes me stronger.”

John Dyer (Bethel, Connecticut) is a Freshman at Wesleyan University majoring in Economics:
“Watching someone you admire struggle with an incurable disease is heartbreaking, but it has also strengthened my character in ways I never imagined.”

Alexus Calhoun (Bloomington, Illinois) is a Senior at Illinois State University majoring in Computer Science:
“Throughout the rest of my life, I will continue to advocate for spreading awareness about Alpha 1 to hopefully encourage other people to get tested and potentially save lives”
E. Lou Glenn Memorial Scholarship Recipient
This scholarship honors the memory of Lou Glenn, a dedicated caregiver of her daughter who was diagnosed with Alpha 1. An icon in the Alpha 1 community, she served on various committees, always supported programs for Alphas, and represented the voice of caregivers.

Ava Castillo (San Antonio, Texas) is a Freshman at Juniata College majoring in Biology:
“Observing the adversity my siblings face daily makes me want to follow their lead in living a healthier life.”
Terry L. Young Memorial Memorial Scholarship Recipient
This scholarship honors the memory of Terry Young, co founder of AlphaNet who also served as the very first AlphaNet Coordinator. An icon in the Alpha 1 community, he perpetuated the AlphaNet slogan, “Alphas serving Alphas.”

Mariella Cimadevilla (Flowery Branch, Georgia) is a Graduate student at Georgia State University studying Occupational Therapy:
“Alpha 1 taught me that my mom was bigger than the deficiency that tried to define her.”
John W. Walsh III (Jack) Memorial Scholarship Recipient
This scholarship honors the memory of Jack Walsh, affectionately known as “Coach”. Having celebrated 94 birthdays, Coach was an icon in the Alpha 1 community. Father to John, Fred, Judy and Sue, he perpetuated the importance of education and family within the entire Alpha 1 community.

Ashleigh Cameron (Lynchburg, Virginia) is a Sophomore at Liberty University majoring in Nursing:
“My uncle and grandmother’s experiences with Alpha 1 have not only shaped their lives but also ignited a passion for advocacy and education within our family.”
Robert (Bob) J. Haggerty Memorial Scholarship Recipient
This scholarship honors the memory of Bob Haggerty, who was the face and voice of the Alpah 1 Association for almost a decade. He served as Treasurer for many years, as well as Master of Ceremonies of the Alpha 1 National Conference.


McKinley Chittick (Harper, Iowa) is a Freshman at Iowa State University majoring in Pre-Veterinary Medicine:
“I haven’t used Alpha 1 as an excuse to stop living life to the fullest.” 1 ALPHA-1 FOU NDATION

The application will soon be available on the website The deadline to submit the completed application with all attachments is April 15, 2026.
Scholarships are available to Alphas and their immediate family members.
Scholarships can be used for study at an approved institution for post-high school education and career change/returning adult students.
To learn more about the award process and download an application, visit alpha1.org/alpha-1-educational-scholarships


The Redwood Study is looking for adults 18 to 75 years of age who have a confirmed or suspected diagnosis of Alpha-1 Liver Disease with PiZZ genotype. There is currently no approved treatment available for Alpha-1 Liver Disease. The investigational study drug aims to reduce the production of the abnormal Z-AAT protein and its buildup in the liver. To learn more and see if you may qualify, talk to your doctor and visit Alpha1.org/Redwood-Study-Prescreening today.

In November, the Alpha 1 Adult Liver A1F Virtual Support Group met online for a presentation by Dr. Virginia Clark, Associate Professor of Medicine at the University of Florida. Dr. Clark is the GI Fellowship training program director. Her clinical research focuses on metabolic and genetic liver diseases with a focus on Alpha 1 Antitrypsin Deficiency (Alpha 1). She is the principal investigator or co investigator on several active clinical trials for therapies in AAT liver disease as well as non alcoholic steatohepatitis. Dr. Clark is part of the Alpha 1 Foundation Clinical Resource Center network and provides care to patients with Alpha 1.
What is the best diet for a liveraffected Alpha?
Dietary recommendations must be individualized based on the severity of the liver condition. In general, I recommend avoiding sugar sweetened beverages and ultra processed foods. Black coffee (2 cups per day) may protect against liver fibrosis. If you have cirrhosis, the recommended protein intake is 1.2 1.5 g/kg of ideal body weight. For example, this would equal 98 123 grams of protein a day for a 180 pound person.
Does the new drug for bronchiectasis (brensocatib) have any consequences for the liver?
Some mild liver enzyme elevations were seen during the clinical trials, but these were not considered to be clinically meaningful. It has been tested in a few patients with cirrhosis, and no safety signals have been identified. It is important to know that individuals with Alpha 1 were excluded from the clinical trial.
How often should an Alpha with moderate scarring and fatty liver have elastography (FibroScan®) or other imaging done?
Monitoring for fibrosis (scarring) worsening should be done every 1 2 years if you already have moderate scarring in the liver. If you have cirrhosis, imaging of the liver should be done every six months.
To follow up, is an MRI better than a FibroScan®?
A FibroScan® is a good test to evaluate for liver stiffness (scarring). MR elastography (MRE) is another imaging technique that can measure the same thing. It is more precise than a FibroScan®, and the results are more reliable in certain circumstances. However, MRE is much more expensive than FibroScan®. For this reason, I use it when the FibroScan® result is equivocal or considered unreliable. I often use the MRE in place of a liver biopsy when I want to know if cirrhosis is present.
Is there any reason to have a FibroScan® after a diagnosis of cirrhosis of the liver?
Yes, it can be used annually to estimate how severe the liver disease is. The FibroScan® result is reported as a kilopascal (kPa). If your kPa score is greater than 20 and your platelet count is <150, an endoscopy to evaluate for esophageal varices is recommended. If the kPa is <20, you can avoid having an endoscopy. This is an evolving area of liver disease management.
What is your take on cholesterol medication and liver disease? Doesn’t cholesterol medication affect the liver?
The most common medications for treating high cholesterol are statins. These have been used for years without major safety concerns for patients. Studies have shown that taking a statin can decrease the chance of heart disease and stroke if you have certain risks, and you can live longer. Some people do not tolerate it due to side effects, but making liver disease worse is uncommon.
Are two alcoholic drinks per month okay for an Alpha-1 patient, or should the liver have no alcohol?
If you have been diagnosed with cirrhosis, you should not drink alcohol. It is less clear what the answer is when you have fibrosis or other risks for liver disease, such as Alpha 1. I recommend avoiding alcohol in most circumstances because there is a lot of evidence that the use of alcohol can make the progression of any liver disease worse.
What vaccines should a liver patient receive?
If liver cirrhosis is present, hepatitis A and B vaccines are recommended, if you are not immune. It is always best to check with your physician.
What are the important signs to look for to determine if your liver is getting sicker?
Early liver disease is asymptomatic. You may see fatty liver on an ultrasound, and the liver enzymes may or may not be elevated. You can detect cirrhosis through imaging and still not have symptoms. Once signs of advanced liver disease occur (ascites, jaundice, varices), you should be under specialty care with a GI or liver doctor.
Check the A1F calendar for the upcoming Spring Alpha-1 Adult Liver A1F Virtual Support Group

Every February, the rare disease community comes together with a shared purpose: to raise awareness, inspire action, and amplify the voices of millions of individuals and families living with rare conditions. For the Alpha 1 Foundation (A1F), Rare Disease Day is not just a single day of recognition, but a month long commitment to education, advocacy, and connection, ensuring that Alpha 1 Antitrypsin Deficiency (Alpha 1) remains part of the global rare disease conversation.
Rare Disease Day is observed on the last day of February each year and serves as a powerful reminder of the scale and impact of rare diseases worldwide. More than 300 million people across the globe are living with a rare disease, and Alpha 1 is one of more than 7,000 rare conditions currently identified. Nearly 72% of rare diseases are genetic, and for many individuals, the path to diagnosis, treatment, and long term care can be long and uncertain. Through awareness and advocacy, Rare Disease Day works to create visibility for these challenges while pushing progress forward.
Throughout February 2026, A1F was is proud to collaborate with RareDiseaseDay.org and the National
Organization for Rare Disorders (NORD) to raise awareness of Alpha 1 and support the broader rare disease community. Patients, caregivers, clinicians, researchers, and advocates are encouraged to take part by sharing educational resources, engaging in events, and telling their personal Alpha 1 stories. By downloading the Rare Disease Day Digital Toolkit and sharing official A1F content on social media using the hashtags #Alpha1Awareness and #RareDiseaseDay, supporters amplified awareness and helped extend the impact of Rare Disease Day far beyond a single day.
Rare Disease Day 2026 was marked by a series of events that brought the community together across education, research, and policy. On February 21st, Rare Disease Day was celebrated at the A1F’s Austin Education Day, where Alphas, family members, caregivers, friends, and healthcare providers gathered to learn about the latest Alpha 1 research, lung and liver disease, and practical strategies for living well with Alpha 1. On February 23rd, the U.S. Food and Drug Administration hosted a virtual Rare Disease Day public meeting focused on engaging patients and communities to advance rare disease medical
product development and regulation. That same week, from February 23rd through 27th, the EveryLife Foundation for Rare Diseases convened advocates from across the country for Rare Disease Week, empowering participants to engage directly with policymakers and advocate for meaningful change.
On Monday, February 23rd, at the Health and Human Services Rare Disease Announcement, the U.S. Food and Drug Administration (FDA) issued draft guidance for sponsors seeking approval for targeted individualized therapies by generating
substantial evidence of effectiveness and safety when randomized controlled trials are not feasible due to small patient populations. “President Trump promised to accelerate cures for American families — and we are delivering, especially for children with ultra rare diseases who cannot afford to wait,” said Health and Human Services Secretary Robert F. Kennedy, Jr. “We are cutting unnecessary red tape, aligning regulation with modern biology, and clearing a path for breakthrough treatments to reach the patients who need them most.”

Miriam O’Day, Senior Policy Advisor, welcomed the opportunity to directly engage FDA leadership during the announcement. “I was excited to ask FDA Commissioner, Dr. Makary, about the flexibility of clinical trial design for rare disease therapies, including treatments for Alpha 1 Antitrypsin Deficiency. For small patient populations, traditional randomized controlled trials are often not feasible. FDA’s draft guidance recognizing alternative pathways to generate substantial evidence is an important step toward aligning regulatory science with the realities faced by rare disease communities. Families living with Alpha 1 cannot afford unnecessary delays — thoughtful flexibility in trial design can help bring safe, effective therapies forward more efficiently while maintaining rigorous standards.”
The week culminated on February 27th with Rare Disease Day at the National Institutes of Health (NIH), where a full day of in person and virtual programming will center on lived experiences, scientific discovery, and collaboration. The event featured panel
discussions, rare disease stories, scientific posters, exhibits, and an art exhibition, highlighting both the human and scientific dimensions of rare disease research. On February 24th, the National Organization for Rare Disorders (NORD) hosted a public congressional briefing at the U.S. Capitol Visitor Center titled Patient Advocacy Driving Innovation for People Living with Rare Diseases, underscoring the critical role of advocacy and federal policy in advancing research and treatment. Today, an estimated 30 million Americans are living with a rare disease, yet fewer than five percent of known rare diseases have an FDA approved treatment, a gap that continues to drive urgent advocacy and innovation.
At its core, Rare Disease Day is about people, sharing stories, building community, and reminding those affected by rare diseases that they are not alone. By participating in Rare Disease Day, raising awareness, attending events, and speaking out, the Alpha 1 community helps drive progress not only for Alpha 1 but for all rare diseases. 1 ALPHA-1





ZEMAIRA®, Alpha1-Proteinase Inhibitor (Human) lyophilized powder for reconstitution for intravenous use
Initial U.S. Approval: 2003
BRIEF SUMMARY OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ZEMAIRA safely and effectively. See full prescribing information for ZEMAIRA.
-----------------------------------INDICATIONS AND USAGE----------------------------------
• ZEMAIRA is an alpha1-proteinase inhibitor (A1-PI) indicated for chronic augmentation and maintenance therapy in adults with A1-PI deficiency and clinical evidence of emphysema.
• The effect of augmentation therapy with ZEMAIRA or any A1-PI product on pulmonary exacerbations and on the progression of emphysema in A1-PI deficiency has not been demonstrated in randomized, controlled clinical studies.
• ZEMAIRA is not indicated as therapy for lung disease patients in whom severe A1-PI deficiency has not been established.
-------------------------------DOSAGE AND ADMINISTRATION------------------------------
For intravenous use after reconstitution only
• The recommended weekly dose of ZEMAIRA is 60 mg/kg body weight. Dose ranging studies using efficacy endpoints have not been performed with ZEMAIRA or any A1-PI product.
• Administer through a suitable 5 micron infusion filter (not supplied) at room temperature within 3 hours after reconstitution.
• Do not mix with other medicinal products. Administer through a separate dedicated infusion line.
• Administer at a rate of approximately 0.08 mL/kg/min as determined by the response and comfort of the patient.
• Monitor closely the infusion rate and the patient’s clinical state, including vital signs, throughout the infusion. Slow or stop the infusion if adverse reactions occur. If symptoms subside promptly, the infusion may be resumed at a lower rate that is comfortable for the patient.
--------------------------------DOSAGE FORMS AND STRENGTHS---------------------------
ZEMAIRA is supplied in a single-dose vial containing approximately 1000 mg, 4000 mg, or 5000 mg of functionally active A1-PI as a white to off-white lyophilized powder for reconstitution with 20 mL, 76 mL, or 95 mL of Sterile Water for Injection, USP. The amount of functional A1-PI is printed on the vial label and carton.
---------------------------------------CONTRAINDICATIONS -----------------------------------
• History of anaphylaxis or severe systemic reactions to ZEMAIRA or A1-PI protein.
• Immunoglobulin A (IgA)-deficient patients with antibodies against IgA, due to the risk of severe hypersensitivity.
• Observe any signs of hypersensitivity such as tachycardia, hypotension, confusion, syncope, oxygen consumption decrease, and pharyngeal edema when administering ZEMAIRA to patients with known hypersensitivity to an A1-PI product.
• Patients with selective or severe IgA deficiency can develop antibodies to IgA and, therefore, have a greater risk of developing potentially severe hypersensitivity and anaphylactic reactions. If anaphylactic or severe anaphylactoid reactions occur, discontinue the infusion immediately.
• Because ZEMAIRA is made from human blood, it may carry a risk of transmitting infectious agents, e.g., viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent and, theoretically, the Creutzfeldt-Jakob disease (CJD) agent.
• Serious adverse reactions reported following administration of ZEMAIRA in prelicensure clinical trials included one event each in separate subjects of bronchitis and dyspnea, and one event each in a single subject of chest pain, cerebral ischemia and convulsion.
• The most common adverse reactions occurring in at least 5% of subjects receiving ZEMAIRA in all pre-licensure clinical trials were headache, sinusitis, upper respiratory infection, bronchitis, asthenia, cough increased, fever, injection site hemorrhage, rhinitis, sore throat, and vasodilation.
To report SUSPECTED ADVERSE REACTIONS, contact CSL Behring Pharmacovigilance at 1-866-915-6958 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Based on January 2024 revision
Influenza (Flu) • COVID-19 • Respirator y Syncy tial Virus
Seasonal respiratory viruses are more common during the winter months. For people with Alpha-1 Antitrypsin Def iciency (Alpha-1), especially those with lung disease, these infections can increase the risk of severe illness. Staying up to date with recommended vaccines is an important way to help protect lung health.
Reduce the risk of infection
Lessen the severity of illness
Protect those who are lung-affected
Impor tant Vaccines to Consider:

Updated every year
Protects against A & B strains
Those who are 65+ should get a high-dose option
Updated vaccines for recent strains
Still widely circulating
Especially important for people with lung disease
One-time vaccine (not yearly)
Recommended for those who are 60+ with risk factors like Alpha-1
Strongly encouraged for those who are 75+
Yes, Flu, COVID-19, and RSV vaccines can be given during the same visit. Mild side effects may be possible including arm pain, fatigue, and headache. There is no waiting period needed between vaccines.




When Brian and Kara Hodges learned their daughter Malia had Alpha‑1 Antitrypsin Deficiency (Alpha‑1), their lives changed immediately. What began as a search for medical answers quickly became a journey defined by determination, community support, and a commitment to securing the best future for their daughter. From their home in League City, Texas, they entered an unfamiliar world of genetic testing, new relationships, and a growing network of families and clinicians who would become essential to their support system. Today, they are not only parents managing their child’s rare condition but active advocates for other families facing the same challenges.
Their path forward began with Dr. Sanjiv “Sonny” Harpavat of Texas Children’s Hospital, a respected
pediatric liver specialist who diagnosed Malia at 3 weeks old and guided them through the early stages of her care. Brian describes him as an invaluable resource and the person who first connected them to the Alpha‑1 Foundation (A1F).
Prior to Malia’s diagnosis, the family had no awareness of Alpha‑1. Subsequent testing revealed that Brian and Kara were MZ carriers. Further testing showed that both of their mothers were also MZ carriers, while their fathers tested as MS carriers. What had been unknown quickly became a clearer picture of their genetic risk and its implications for their family’s health.
The Hodges family first engaged with A1F through virtual walks in November of 2020 and 2021. Their

deeper involvement began after attending the Celtic Connection event in Boston, MA, in 2024, their first in‑person event following the pandemic. Brian and Kara were invited to attend the Building Friends for a Cure (BFC) training that weekend. Surrounded by others who understood their concerns, they realized they wanted to take a more active role. Since then, they have attended two A1F National Conferences, joined the A1F Speaker Bureau and the Advocacy & Public Policy Group, continued participating in fundraising walks, and began organizing additional awareness events. What started as information‑gathering has become a sustained commitment to the Alpha‑1 community.
They credit A1F and the Alpha 1 community as their greatest source of support. Kara notes that having others to rely on significantly eased their adjustment. They speak highly of the friendships they have formed, the clinicians who have given them hope, and the Parents of Alpha 1 Kids (PAK) Virtual Support Group, where families share experiences and guidance. Brian describes it as “an amazing family of Alphas,” many of whom they expect to remain connected with for life.
Their advice to other Alpha‑1 parents reflects their own experience: ask questions, stay engaged, and rely on the community. They also advocate

for extended family testing, emphasizing the potential of early diagnosis to positively influence long‑term health outcomes. In some cases, it can correct misdiagnosed conditions, such as COPD, as identifying Alpha 1 status enables individuals to access appropriate treatments, including augmentation therapy through infusions.
Looking ahead, the Hodges family believes progress for the Alpha‑1 community depends on continued support for clinicians, researchers, government, and industry partners working toward improved therapies and ultimately a cure. They emphasize the need to expand testing, grow the network of Alpha 1 experts or Clinical Resource Centers (CRCs), and maintain active involvement in policy discussions to advance healthcare access and coverage.
At the heart of their efforts is their incredible 6 year old daughter, whose resilience inspires them. Their hope for Malia is simple yet profound: a cure, a life free from the limitations of her Alpha‑1 diagnosis, and the strength to use her voice to inspire and mentor other pediatric Alphas as she grows. They call her their warrior, and in many ways, she has made them warriors as well, dedicated to advancing awareness, research, raising funds and community support for families affected by Alpha‑1.

— Heather, actual patient since 2018
Trusted by patients and their doctors for more than 35 years.
• The #1 prescribed augmentation therapy for alpha1antitrypsin deficiency, or alpha-1*
• Proven to effectively raise the alpha1 antitrypsin protein levels in patients with severe alpha-1
• The PROLASTIN DIRECT® program gives you the confidence of ongoing personalized support
Visit PROLASTIN.com to learn more.
*Data on file, Executive Summary, Grifols

PROLASTIN ® -C LIQUID is an alpha1-proteinase inhibitor (human) (alpha1-PI) indicated for chronic augmentation and maintenance therapy in adults with clinical evidence of emphysema due to severe hereditary deficiency of alpha1-PI (alpha1-antitrypsin deficiency).
Limitations of Use
• The effect of augmentation therapy with any alpha1-PI, including PROLASTIN-C LIQUID, on pulmonary exacerbations and on the progression of emphysema in alpha1-PI deficiency has not been conclusively demonstrated in randomized, controlled clinical trials
• Clinical data demonstrating the long-term effects of chronic augmentation or maintenance therapy with PROLASTIN-C LIQUID are not available
• PROLASTIN-C LIQUID is not indicated as therapy or lung disease in patients in whom severe alpha1-PI deficiency has not been established
PROLASTIN-C LIQUID is contraindicated in immunoglubin A (IgA)-deficient patients with antibodies against IgA or patients with a history of anaphylaxis or other severe systemic reaction to alpha1-PI products.
Hypersensitivity reactions, including anaphylaxis, may occur. Monitor vital signs and observe the patient carefully throughout the infusion. If hypersensitivity symptoms occur, promptly stop PROLASTIN-C LIQUID infusion and begin appropriate therapy.
Because PROLASTIN-C LIQUID is made from human plasma, it may carry a risk of transmitting infectious agents, eg, viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent, and, theoretically, the Creutzfeldt-Jakob disease (CJD) agent. This also applies to unknown or emerging viruses and other pathogens.
The most common adverse reactions during PROLASTIN-C LIQUID clinical trials in >5% of subjects were diarrhea and fatigue, each of which occured in 2 subjects (6%).
Please see brief summary of the full Prescribing Information for PROLASTIN-C LIQUID on adjacent page.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088.
The impact of A1F, described in the A1F Impact Report (page 8), would not be possible without the support and contributions from the Alpha-1 community including donors, patients, family, friends, industry, researchers, and clinicians.
A1F’s $100 million investment in research has resulted in:
• $733 million in leveraged support
• A 10 fold increase in the number of clinical trials conducted
• Funding for 130 researchers worldwide
Every gift makes a difference. Together, these contributions fuel scientific progress, strengthen critical services, and advance the search for better therapies and, ultimately, a cure for Alpha‑1. No matter how supporters choose to give, their commitment continues to move the mission forward.
Learn more about ways to give to A1F. Please contact Linda Rodriguez, Senior Director of Development at lrodriguez@alpha1.org with any questions. 1

February 20-21, 2026
Austin, TX

March 7, 2026
Boston, MA
April 10-11, 2026
Fort Lee, NJ
April 17, 2026
Miami, FL

Barcelona, Spain on May 27 -30, 2026 Alpha-1 Global
May 13, 2026
Greenwich, CT
May 18, 2026
Orlando, FL
June 4-7, 2026
San Diego, CA
y September 18-19, 2026
Boise, ID
September 25-27, 2026
Cape Cod, MA
October 1-4, 2026
Miami, FL A
November 6-7, 2026
Kansas City, KS
December 4-5, 2026 Nashville, TN



Are you receiving our emails?
Do you receive the monthly e newsletter “A1F Community Currents”? This is a good time to check that you are on our e mail list. Update your contact information by visiting ALPHA1.ORG
March 21, 2026
SF Bay Area Alphas Pleasant Hill, CA
April 4, 2026
SoCal Roadrunners/ San Diego Alphas Virtual
April 10-11, 2026
A1F Education Day Fort Lee, NJ
December 4-5, 2026
A1F Education Day Nashville, TN
April 17, 2026
29th A1F Gordon L. Snider Critical Issues Workshop Miami, FL
March 22, 2026
Georgia Alphas Tucker, GA
April 25, 2026
Alpha 1 Opportunities Phoenixville, PA
March 28, 2026
NEPA Alphas Mount Bethel, PA
For additional Alpha 1 Support Group meetings or to register to attend an upcoming meeting, please check the A1F Events Calendar: alpha1.org/calendar
March 28, 2026 Charleston, SC Alphas Charleston, SC
June 4-7, 2026
2026 A1F National Conference San Diego, CA
September 18-19, 2026
A1F Education Day Boise, ID
November 6-7, 2026
A1F Education Day Kansas City, KS
May 13, 2026
Bettina B. Irvine Invitational Classic Greenwich, CT
May 18, 2026
A1F Research Grants Award Reception Orlando, FL
September 25-27, 2026
A1F Escape to the Cape Cape Cod, MA


Do you receive Alpha-1 Foundation email blasts, event reminders, or fundraising updates?
Be sure to update your e-mail address and contact information by visiting: alpha1.org/subscribe