Italiana La Metallurgia
International Journal of the Italian Association for Metallurgy
n.02 febbraio 2026
Organo ufficiale dell’Associazione Italiana di Metallurgia.
Rivista fondata nel 1909


International Journal of the Italian Association for Metallurgy
n.02 febbraio 2026
Organo ufficiale dell’Associazione Italiana di Metallurgia.
Rivista fondata nel 1909

International Journal of the Italian Association for Metallurgy
Organo ufficiale dell’Associazione Italiana di Metallurgia. HouseorganofAIMItalianAssociationforMetallurgy. Rivista fondata nel 1909
Direttore responsabile/Chiefeditor: Mario Cusolito
Direttore vicario/Deputydirector: Gianangelo Camona
Comitato scientifico/Editorialpanel: Marco Actis Grande, Silvia Barella, Paola Bassani, Christian Bernhard, Massimiliano Bestetti, Wolfgang Bleck, Franco Bonollo, Irene Calliari, Mariano Enrique Castrodeza, Emanuela Cerri, Vlatislav Deev, Andrea Di Schino, Donato Firrao, Bernd Kleimt, Carlo Mapelli, Denis Jean Mithieux, Roberto Montanari, Marco Ormellese, Mariapia Pedeferri, Massimo Pellizzari, Barbara Previtali, Evgeny S. Prusov, Dario Ripamonti, Dieter Senk
Segreteria di redazione/Editorialsecretary: Flynn Russo
Comitato di redazione/Editorialcommittee: Federica Bassani, Gianangelo Camona, Mario Cusolito, Carlo Mapelli, Federico Mazzolari, Flynn Russo, Silvano Panza
Direzione e redazione/Editorialandexecutiveoffice: AIM - Via F. Turati 8 - 20121 Milano tel. 02 76 02 11 32 - fax 02 76 02 05 51 met@aimnet.it - www.aimnet.it
Reg. Trib. Milano n. 499 del 18/9/1948. Sped. in abb. Post. - D.L.353/2003 (conv. L. 27/02/2004 n. 46) art. 1, comma 1, DCB UD
Immagine in copertina: Tiziano Bellezze

Gestione editoriale e pubblicità Publisher and marketing office: siderweb spa sb Via Don Milani, 5 - 25020 Flero (BS) tel. 030 25 400 06 commerciale@siderweb.com - www.siderweb.com
La riproduzione degli articoli e delle illustrazioni è permessa solo citando la fonte e previa autorizzazione della Direzione della rivista. Reproduction in whole or in part of articles and images is permitted only upon receipt of required permission and provided that the source is cited.
siderweb spa sb è iscritta al Roc con il num. 26116

n.02 febbraio 2026
Anno 117 - ISSN 0026-0843
Editoriale / Editorial a cura di Marina Cabrini Università degli studi di Bergamo . pag.05
Memorie scientifiche / Scientific papers
Corrosione / Corrosion
Studio del comportamento a corrosione dell’acciaio inossidabile AISI 316L prodotto mediante diverse tecnologie di additive manufacturing
D. Fiorona, L. Nani, S. Lorenzi, M. Cabrini, T. Pastore, M. Quarto, D. Manfredi, T. Tirelli ................................. pag.08
Validation of pitting engineering diagrams for stainless steel used in water applications: some recent case studies
S. Hägg Mameng, B. Helmersson, E. Johansson, S. Hosseinpour .......................................................... pag.18
Effect of Passivation Treatments on the Corrosion Resistance Properties of As-sintered 17-4 PH Additive-Manufactured by Binder Jetting Technology
P. Forcellese, W. A. Khan, G. Giuliani, T. Bellezze . pag.26
Corrosion behavior of steel reinforcements in sustainable low-resistive cement mortars
A. Mobili, T. Bellezze, F. Tittarelli . pag.36
Corrosion resistance properties of 304 stainless steel and nickel-titanium orthodontic wires in artificial saliva solution
P. Forcellese, W. A. Khan, G. Orilisi, G. Orsini, F. Luzi, A. Mazzoli, V. Corinaldesi, T. Bellezze . pag.44
Atti e notizie / AIM news
Aziende di AIM ........................................................................................................................ pag.52
Eventi AIM / AIM events ...................................................................................................... pag.55
Secondo classificato “La Metallurgia a Fumetti” ..................................................... pag.58
Normativa / Standards ....................................................................................................... pag.63
Milan
siderweb FORUM è l’appuntamento biennale di siderweb che riunisce i protagonisti dell’industria siderurgica italiana ed europea per discutere il presente e il futuro del settore.
Per la seconda edizione, l’evento cresce in format, durata e attrattività per il pubblico: si svolgerà in contemporanea con EEC 2026 (14th European Electric Steelmaking Conference) ed EMECR 2026 (5thInternational Conference on Energy and Material Efficiency and CO2
Reduction in the Steel Industry), due conferenze internazionali organizzate da AIM
Associazione italiana di Metallurgia
SIDERWEB FORUM
• Scenari geopolitici
• Mercati globali
• Transizione energetica
• Innovazione
EEC | EMECR
• Tecnologie per la decarbonizzazione
• Processi innovativi per EAFs
• Efficienza energetica e riduzione della CO2
Il programma è in fase di definizione.
L’agenda aggiornata dei convegni è disponibile su siderweb.com/siderweb-forum
Oltre ai convegni e alle conferenze, sarà presente anche un’area espositiva dedicata alle aziende del settore. Guarda gli espositori ALREADY ON BOARD
Diventa espositore!
I pacchetti sponsor sono pensati per garantire massima visibilità su tutti i partecipanti.


Diventa sponsor!

“L’ottenimento di leghe metalliche maggiormente resistenti al degrado e lo sviluppo di sistemi di protezione ecocompatibili e duraturi, sono requisiti fondamentali per aumentare la vita di esercizio in sicurezza di prodotti e infrastrutture.”
“The development of metallicalloyswith enhanced resistance to degradation,togetherwith durableandenvironmentally friendlyprotection systems, represents a fundamental requirement forincreasingthesafe service life of products and infrastructures.”
Questo numero raccoglie la seconda parte delle memorie presentate nella XVI edizione delle Giornate Nazionali sulla Corrosione e Protezione tenutesi ad Ancona dal 25 al 27 giugno 2025. La manifestazione si è confermata come un importante punto di incontro tra la realtà industriale e l’università, ulteriormente valorizzata dalla presenza di numerosi giovani. Le memorie pubblicate su questo numero e su quello di novembre/dicembre 2025 de La Metallurgia Italiana testimoniano l’alta qualità dei lavori presentati.
In questa nuova uscita sono presenti lavori incentrati su temi tradizionali, ma sempre attuali, come la failure analysis, la resistenza al pitting, lo studio della corrosione delle armature e delle leghe per impieghi biomedicali, ma è dato ampio spazio anche alla caratterizzazione del comportamento a corrosione di leghe ottenute tramite tecnologie additive.
Quest’ultimo argomento è di grande attualità poiché, attraverso la cosiddetta stampa 3D, è possibile realizzare geometrie complesse con costi contenuti e senza lavorazioni alle macchine utensili, con notevoli vantaggi di tempo, soprattutto per le leghe di difficile lavora-

This issue collects the second part of the papers presented at the 16th National Conference on Corrosion and Protection, held in Ancona from 25 to 27 June 2025. The event was confirmed as an important meeting point between industry and academia, further enhanced by the strong participation of young researchers. The quality of the contributions is attested by the papers published in the November/December issue and in the present one of La Metallurgia Italiana.
This new issue includes works focused on traditional yet continually relevant topics—such as failure analysis, pitting corrosion resistance, the study of corrosion of reinforcing steels, and alloys for biomedical applications—while also devoting substantial attention tothecharacterizationofthecorrosionbehaviorofalloys produced by additive manufacturing technologies. This latter topic is of great current interest, as socalled 3D printing enables the fabrication of complex geometries at reduced cost and without conventional machining operations, offering significant time advantages, especially for alloys that are difficult to machine. However, these technologies produce
zione. Tuttavia, queste tecnologie producono materiali con microstrutture differenti da quelli ottenibili tramite lavorazione a caldo, che ne modificano il comportamento meccanico e a corrosione. Da ciò nasce la crescente esigenza di studio delle loro caratteristiche in modo da poterne sfruttare a pieno i vantaggi e prevenire eventuali problemi.
Lo studio dell’interazione tra proprietà e microstruttura, che è alla base della scienza e tecnologia dei materiali, trova in questo settore la sua naturale connotazione e si propone come propulsore verso una manifattura sempre più circolare e sostenibile, ma allo stesso tempo al passo con le richieste sempre più specifiche dell’industria moderna. L’ottenimento di leghe metalliche maggiormente resistenti al degrado e lo sviluppo di sistemi di protezione ecocompatibili e duraturi, sono requisiti fondamentali per aumentare la vita di esercizio in sicurezza di prodotti e infrastrutture, abbattendo in questo modo i costi economico-ambientali di realizzazione e manutenzione.
materials with microstructures that differ from those obtained through hot working, thereby altering their mechanical and corrosion behavior. This has led to a growing need for systematic investigation of their characteristics in order to fully exploit their advantages and prevent potential issues.
The study of the interaction between properties and microstructure—which lies at the core of materials science and engineering—finds in this field a natural applicationandactsasadrivingforcetowardincreasingly circular and sustainable manufacturing, while remaining aligned with the ever more specific demands of modern industry. The development of metallic alloys with enhanced resistance to degradation, together with durable and environmentally friendly protection systems, represents a fundamental requirement for increasing the safe service life of products and infrastructures, thereby reducing the economic and environmental costs associated with their production and maintenance.




The 4th WCCM will take place on August 25-27, 2026, at FAST Congress Centre, Milan, Italy. The primary objective of the World Congress on Condition Monitoring (WCCM) is to facilitate the global exchange of research and applications in Condition Monitoring (CM) and related areas, with a strong emphasis on scientific and technological advancements, industrial applications and international cooperation. The Congress serves as a worldwide platform for scientists and professionals from both academia and industry to share knowledge and insights. The event is organized in collaboration with condition monitoring and NDT societies worldwide.
• 2017: WCCM 2017 London, UK
• 2019: WCCM 2019 Singapore
• 2024: WCCM 2024 Beijing, China
The congress technical programme includes:
• Plenary keynote presentations
• Distinguished overview presentations
• Invited and contributed presentations, including case study presentations
• Industrial sessions for major industrial sectors
• Workshops/tutorials on advanced topics in condition monitoring, led by recognised scientists and engineers
• Expert panel session on hot topics in condition monitoring and related areas
• Exhibition, vendor presentations and a plenary spotlight session for exhibitors and sponsors
• New deadline for abstract submission: February 25, 2026
• Notification of abstract acceptance: February 28, 2026
• Deadline for submission of full papers: April 20, 2026
• Deadline for Speakers registration & payment: ..........April 30, 2026
• Deadline for early bird registration & payment: June 10, 2026
• Standard Registration deadline: July 31, 2026
NH Milano Congress centre. Address: Strada 2, 7, Assago (MI) - Italy. NH Milano Congress Centre is located in Assago, Milan business district just 15 minutes away from the city center and nearby the vibrant Navigli area.
The detailed exhibiting and sponsorship packages will be available on the WCCM 2026 website.


Prof. Dr. Len Gelman
The University of Huddersfield, UK
Prof. Dr. Hamid Reza Karimi Politecnico di Milano, Italy
For inquiries, contact:
• E-mail: conference@aimnet.it
• Phone: +39 0276021132
• Website: www.aimnet.it/wccm2026
Follow us for updates:
10.36146/2026_02_08
D. Fiorona, L. Nani, S. Lorenzi, M. Cabrini, T. Pastore, M. Quarto, D. Manfredi, T. Tirelli
Lo scopo del presente lavoro è la valutazione del comportamento a corrosione localizzata di provini in acciaio AISI
316L ottenuti con diverse tecnologie di additive manufacturing, mettendoli a confronto con prodotti ottenuti per via tradizionale.
Sono state condotte delle prove di polarizzazione potenziodinamica e prove di critical pitting temperature (CTP): i risultati delle prove di polarizzazione potenziodinamica hanno mostrato differenze nei potenziali di breakdown tra le tecnologie additive e le tecniche produttive tradizionali, mentre quelle di CPT hanno confermato che il comportamento rispetto alla corrosione localizzata per pitting differisce in modo significativo in relazione alle diverse tecnologie di produzione.
L’articolo ribadisce l’importanza della definizione di protocolli idonei per la qualificazione a corrosione delle leghe ottenute per additive manufacturing che tengano conto della specificità del processo produttivo e dei trattamenti di post processo specifici per le leghe stesse.
INTRODUZIONE
L’acciaio inossidabile austenitico AISI 316L è tradizionalmente considerato un materiale strategico in diversi settori industriali, tra cui Oil&Gas, biomedicale, farmaceutico e alimentare per le sue caratteristiche meccaniche unite alla buona resistenza alla corrosione in ambienti aggressivi. Per queste ragioni, a oggi, questa lega è una delle più utilizzate ed è disponibile nel portafoglio materiali di diversi processi di additive manufacturing (AM). Tuttavia, non è ancora definito in maniera univoca come le proprietà meccaniche e a corrosione si modifichino in funzione della tecnologia impiegata, rendendo necessaria una caratterizzazione specifica per confrontare le prestazioni del materiale prodotto con diverse tecnologie AM rispetto ai prodotti commerciali tradizionali. Negli ultimi decenni, le tecnologie di manifattura additiva per la realizzazione di componenti metallici hanno subito una rapida diffusione in molti settori industriali per applicazioni ingegneristiche avanzate [1]. Il loro principale vantaggio è la possibilità di realizzare geometrie complesse con costi contenuti e in alcuni casi senza necessità di lavorazioni finali alle macchine utensili, con risparmio dei
Davide Fiorona, Lorenzo Nani, Sergio Lorenzi, Marina Cabrini, Tommaso Pastore
Dipartimento di Ingegneria e Scienze applicate, Università degli Studi di Bergamo, Italy
Mariangela Quarto
Dipartimento di Ingegneria gestionale, dell’informazione e della produzione, Università degli Studi di Bergamo, Italy
Diego Manfredi
Dipartimento di Scienza applicata e Tecnologia, Politecnico di Torino, Italy
Tommaso Tirelli
Aidro s.r.l., Taino, Varese, Italy
tempi di industrializzazione e di attrezzaggio, al quale si aggiunge il vantaggio di un’economia circolare legata al recupero del materiale non lavorato e alla riduzione degli scarti e degli sfridi [2]. I materiali prodotti per additive manufacturing hanno caratteristiche micro e macrostrutturali uniche, strettamente dipendenti dalla tecnica additiva adottata che ne determinano proprietà meccaniche differenti dai materiali prodotti con lavorazioni convenzionali [3, 4], ma possono influire anche sulla loro resistenza alla corrosione. La valutazione di come la tecnologia additiva possa influenzare il comportamento a corrosione della lega, può essere effettuata tramite l’esecuzione di test standardizzati e il confronto dei risultati ottenuti su provini ricavati da materiale proveniente da lavorazioni tradizionali, quali la deformazione plastica a caldo o a freddo, e quelli ottenuti tramite queste tecnologie innovative. Questo lavoro ha l’obiettivo ultimo di valutare l’effetto del processo produttivo sul comportamento a corrosione localizzata dell’acciaio AISI 316L, ottenuto mediante Laser Powder Bed Fusion (LPBF), Binder Jetting (BJ) e Material Extrusion (MEX).
La Laser Powder Bed Fusion (LPBF) rappresenta la tecnologia additiva più matura e diffusa in ambito industriale per materiali metallici. Il processo si basa sulla fusione selettiva di strati di polvere metallica mediante un fascio laser ad alta densità di potenza, all’interno di una camera in atmosfera inerte e controllata. Consente di ottenere componenti a basso indice di porosità (<0,5%) con proprietà meccaniche comparabili o superiori ai materiali lavorati per via convenzionale. Tra i principali vantaggi si annoverano l’elevata precisione dimensionale, la possibilità di realizzare geometrie complesse e di produrre leghe innovative dal punto di vista composizionale e quasi completamente esenti da alliganti indesiderati. Gli svantaggi includono costi elevati delle polveri, limitata produttività e necessità di supporti e trattamenti termici post-processo. È oggi ampiamente utilizzata nei settori aerospaziale, medicale e racing, con prospettive di automatizzazione e incremento della produttività per impieghi più estesi. Dal punto di vista della resistenza alla corrosione considerando l’ambiente aerato 0,6 M NaCl, la LPBF mostra una buona resistenza alla corrosione localizzata [5, 6]. Alcuni studi riportano inoltre che la resistenza sia persino migliore rispetto al 316L realizzato convenzionalmente [7]. In ogni caso, i meccanismi di corrosione della LPBF
sono dettati da aspetti microstrutturali. Infatti, il rapido processo di solidificazione induce una microstruttura unica, con presenza di melt-pool, grani epitassiali e una distribuzione non omogenea degli elementi in lega che, per esempio, può modificare il film di passività. Inoltre, la presenza di stress residui sembra avere un effetto benefico sull’aumento del potenziale di pitting, ma rende più difficile la ripassivazione del materiale. Invece, le ipotesi sull’effetto della struttura e della dimensione dei grani restano senza conclusione [8]. In aggiunta, sono stati presi in considerazione fattori macroscopici quali la porosità e la rugosità superficiale, senza tuttavia chiarire del tutto il loro impatto. In ogni caso, sembra che entrambe queste caratteristiche possano influire negativamente sulla resistenza a corrosione localizzata [8].
La tecnologia Binder Jetting (BJ) prevede la deposizione selettiva di un legante su un letto di polvere metallica, senza apporto di calore durante la fase di stampa. Il pezzo ottenuto (“green part”) viene successivamente sottoposto a debinding, per eliminare la parte polimerica, e sinterizzazione per raggiungere le proprietà meccaniche richieste. Il processo consente velocità di produzione molto superiori alla LPBF e l’assenza di tensioni residue, ma genera componenti con porosità maggiore e isotropia inferiore, richiedendo un’attenta gestione delle fasi di debinding e sinterizzazione. È una tecnologia promettente per la produzione di serie medio-alte di componenti strutturali e utensili, con un forte potenziale di industrializzazione in ambiti automotive e macchine utensili. Solo alcuni potenziali fattori di corrosione sono stati analizzati per i materiali prodotti con la tecnica BJ, rendendo necessaria un’attenta fase di ricerca per colmare l’incompletezza delle informazioni [9]. A questo riguardo, le porosità sembrano avere un ruolo chiave come sito di nucleazione favorevole, benché ancora indefinito. Analogamente, resta incerto l’impatto della presenza di inclusioni [10].
La tecnologia Material Extrusion (MEX) per metalli, evoluzione della Fused Filament Fabrication (FFF), impiega filamenti e/o granulato caricati a polvere metallica o paste composte da polveri metalliche e legante polimerico, estruse e successivamente sottoposte a debinding e sinterizzazione. Pur offrendo costi ridotti e semplicità di processo, presenta limitazioni in termini di accuratezza e porosità finale, rendendola più adatta a prototipi funzionali e piccole serie. Tuttavia, i recenti sviluppi nei materiali feed-
stock e nella gestione del sintering ne stanno ampliando l’applicazione in ambiti produttivi decentralizzati e per la manifattura on-demand. Lo studio del comportamento a corrosione della tecnica MEX si è finora focalizzato principalmente sia su alcuni aspetti microstrutturali, per esempio la morfologia dei grani e la presenza di fasi secondarie [11], sia soprattutto sui fattori macroscopici come la presenza di macroporosità e l’impatto della rugosità superficiale [9, 11, 12], lasciando comunque aperto il campo di ricerca legato a questa tecnologia.
Nel complesso, queste tecnologie definiscono un panorama complementare: la LPBF per componenti ad alte prestazioni e possibili materiali taylor made, la BJ per la produzione su scala industriale e la MEX come soluzione economica e flessibile per la produzione distribuita. I campioni ottenuti con le tecniche additive sono stati confrontati con la medesima lega realizzata con processi convenzionali per deformazione plastica attraverso lami-
nazione a caldo e a freddo, utilizzate come riferimento. Dal punto di vista dello studio del comportamento a corrosione, obiettivo di questo lavoro, è stata riscontrata una maggiore conoscenza sul comportamento della tecnica LPBF, al contrario per le tecniche BJ e MEX per le quali è necessaria un’approfondita ricerca.
METODOLOGIA SPERIMENTALE
Le prove sono state eseguite su provini di AISI 316L, la cui composizione e PREN sono indicati nella tabella 1, mettendo a confronto i dati ottenuti da analisi al quantometro rispetto alla composizione nominale da normativa UNI EN 10088. Le composizioni di tutte le tecnologie analizzate rientrano nella specifica fornita dalla normativa di riferimento, a meno del tenore di carbonio per la tecnologia BJ, il cui valore risulta eccedente, questo nonostante il tenore di carbonio dichiarato per le polveri di partenza risulti inferiore al valore limite di riferimento.
- Composizione e PREN dei campioni / Specimens composition and PREN.
I provini cilindrici ottenuti tramite tecnologie tradizionali (altezza 2-5 mm) sono stati ricavati da una barra di diametro 40 mm trafilata a caldo (Hot Rolled - HR), da una barra di diametro 16 mm trafilata a freddo (Cold Rolled - CR) e da un laminato (Sheet Metal - SM). I provini delle tecniche additive sono stati stampati in monete di 5 mm di altezza e 15 mm di diametro. Sono stati levigati con carte abrasive in SiC a granulometria decrescente fino a 4000 grit e successivamente con pasta abrasiva di granulometria 1 µ m.
La caratterizzazione elettrochimica è stata effettuata tramite prove di polarizzazione potenziodinamica ciclica per la determinazione del potenziale di breakdown e prove di critical pitting temperature. Le prove di polarizzazione potenziodinamica ciclica sono state svolte in singolo
mentre le prove di critical pitting temperature sono state condotte in doppio. Le prove di polarizzazione potenziodinamica ciclica sono state eseguite secondo la normativa ASTM G61, in soluzione di NaCl 0.6M, deaerata e termostatata a 25 ± 1 °C; il potenziale di corrosione libera è stato monitorato per 3600 s, fino al raggiungimento di un valore stabile, di seguito è stata eseguita una scansione di potenziale di 10 mV/min, da -20 mV vs Ecorr fino a 1200 mV vs SCE o fino al superamento di 5 mA/cm2 di corrente anodica, quindi la scansione del potenziale è stata invertita fino alla ripassivazione del campione. Tale normativa considera lo svolgimento di una polarizzazione potenziodinamica ciclica al fine di determinare la suscettibilità alla corrosione localizzata (morfologia di corrosione pitting o
crevice); inoltre un indice della suscettibilità all’innesco di un fenomeno di corrosione localizzata, in questo test, è da intendersi come il potenziale al quale si assiste un rapido incremento della corrente anodica. Ancorchè i risultati della prova possono essere condizionati dall’innesco dei ben noti fenomeni di corrosione per crevice sotto la guarnizione, nel presente lavoro le prove di polarizzazione ciclica sono da intendersi interpretate come indice di suscettibilità relativa all’innesco della corrosione localizzata. Il potenziale a cui fa riferimento la normativa è indicato come potenziale di break-down, ed è stato calcolato dalle curve di polarizzazione come potenziale al quale si registra un aumento della densità di corrente anodica oltre 10 µ A/cm2, il potenziale di ripassivazione è stato calcolato come il potenziale nel quale la curva di ritorno intercetta la zona di passività. Come densità di corrente nel tratto passivo è stato considerato il valore corrispondente al potenziale di 0 mV vs SCE. La considerazione sul potenziale di breakdown ha come scopo principale quello di qualificare il materiale in funzione della tecnica produttiva e mostrare l’effetto che questa ha sul comportamento a corrosione. Le prove di CPT sono state eseguite secondo la normativa ASTM G150 in soluzione aerata di NaCl 1M, e in soluzione con minore concentrazione di cloruri (NaCl 0.01M), sempre aerata, al fine di discriminare meglio l’effetto della tecnologia produttiva in un ambiente meno aggressivo rispetto a quello previsto dalla normativa. Le prove sono state eseguite dopo aver raggiunto un valo-
re stabile del potenziale di corrosione libera, monitorato per 600 s alla temperatura costante di 0°C. I campioni sono stati quindi polarizzati potenziostaticamente a +800 mV vs SCE, misurando la densità di corrente circolante. Il campione è riscaldato con una velocità pari a 1°C/minuto. La temperatura critica di pitting viene individuata come il valore al quale la densità di corrente supera i 100 µ A/cm2 senza avere abbassamenti per almeno 60 secondi. Al termine delle prove tutti i provini sono stati osservati al microscopio ottico per la determinazione delle zone di attacco.
Le curve di polarizzazione presentano degli andamenti differenti: per i campioni SM e LPBF si osserva un tratto passivo ben delineato fino al potenziale di breakdown (figura 1 e figura 3), l’innesco della corrosione localizzata è confermata dall’ampio ciclo di isteresi della curva una volta invertita la scansione del potenziale; un comportamento analogo è osservato anche per i campioni CR e HR, ma con potenziali più bassi. MEX e BJ mostrano delle curve con un tratto passivo meno marcato, con numerosi picchi di correnti riconducibili alla rottura e ripassivazione del metallo. Per questi provini, il potenziale è di poco superiore o coincidente con il potenziale di corrosione, indice di una maggiore suscettibilità all’innesco della corrosione localizzata.
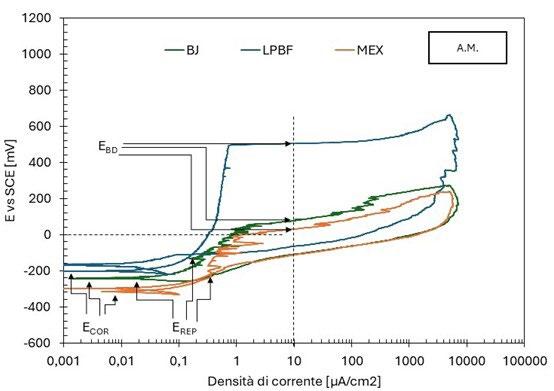
Fig.1 - Curve di polarizzazione potenziodinamica tecniche additive / Potentiodynamic polarization curves of additive techniques.
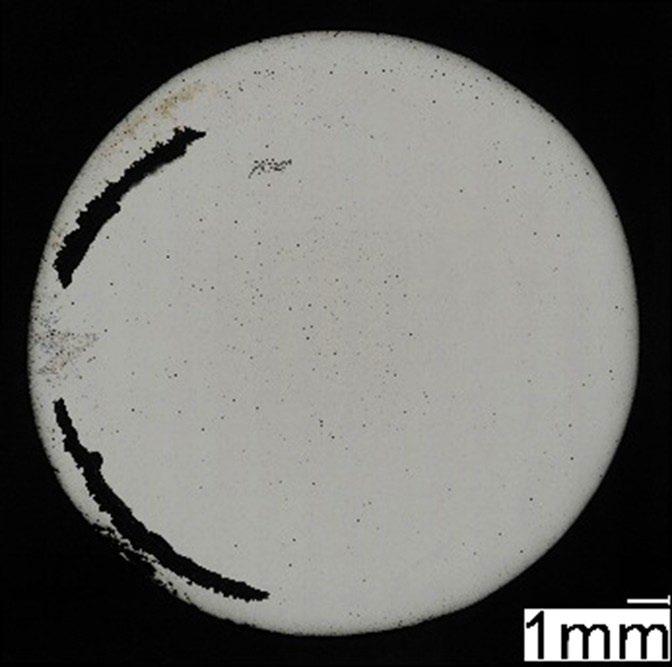
Fig.2 - Campione LPBF post prova di polarizzazione potenziodinamica / LPBF specimen after potentiodynamic polarization test.
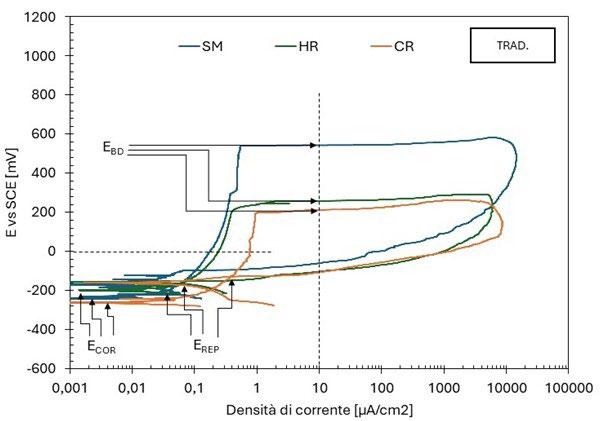
Fig.3 - Curve di polarizzazione potenziodinamica tecniche tradizionali / Potentiodynamic polarization curves of traditional techniques.
La tabella 2 riassume i risultati delle prove potenziodinamiche cicliche. I potenziali di corrosione libera e di break-down sono riassunti nel grafico in figura 5 in funzione del PREN della lega. I potenziali di corrosione (triangoli blu nel grafico) risultano compresi tra -200 e -300 mV vs SCE per tutti i provini, a indicare per tutti il mantenimento delle condizioni di passività durante il periodo di stabilizzazione prima dell’inizio della scansione di potenziale. Viceversa, i potenziali di break-down (quadrati rossi nel grafico) sono compresi in un intervallo di oltre 450 mV, a indicare delle significative differenze di comportamento tra un provino e l’altro. Tali differenze non sembrano correlate con il valore del PREN. Infatti, il provino LPBF e il tradizionale laminato a caldo (SM) hanno valori molto elevati, pari a 503 e 543 mV vs SCE rispettivamente, i provini provenienti dalle barre trafilate a caldo (HR) e a freddo (CR) hanno valori poco superiori a 200 mV vs SCE (258 e 212 mV vs SCE rispettivamente) mentre i provini BJ e MEX danno dei potenziali di break-down molto bassi, rispettivamente di 76 e 29 mV vs SCE. Questi valori sono naturalmente indicativi poiché ottenuti da prove in singolo e considerando la ben nota natura statistica del pitting [13, 14]. Tutti i provini osservati al termine della prova di polarizzazione ciclica, indipendentemente dalla tecnologia produttiva, hanno
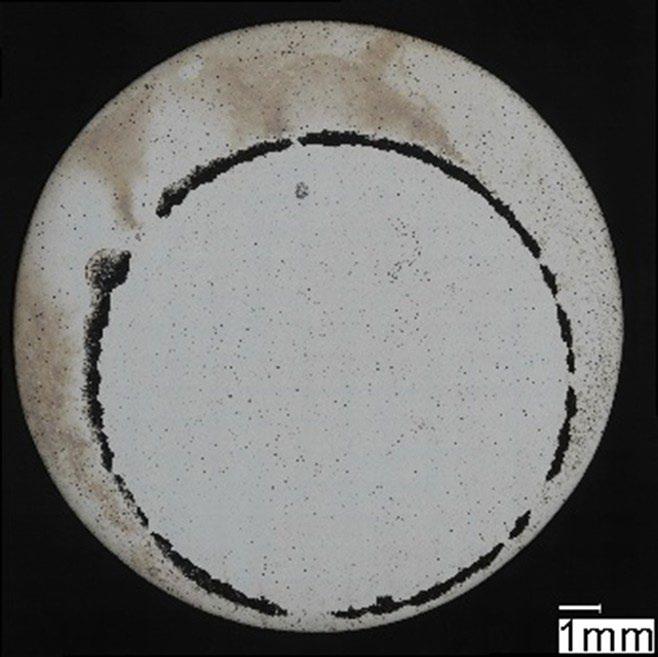
Fig.4 - Campione HR post prova di polarizzazione potenziodinamica / HR specimen after potentiodynamic polarization test.
mostrato corrosione localizzata per crevice nella zona della guarnizione (figura 2 e 4) che risulta enfatizzata dalla forte acidificazione anodica prodotta durante la prova stessa. I potenziali di ripassivazione sono notevolmente inferiori rispetto ai potenziali di break-down, indicando un intervallo di passività perfetta molto ridotto, pari a pochi millivolt per leghe prodotte con tecnologie additive e da 50 a 100 mV per le leghe prodotte con tecnologie tradizionali, indicando per le prima una maggiore difficoltà nel ripristinare il film passivo.
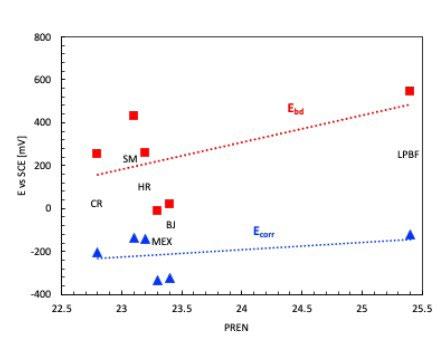
Fig.5 - Potenziali di breakdown, Potenziali di ripassivazione, Potenziali di corrosione libera / Breakdown potential, Repassivation potential, Corrosion potential.
Anche la densità di corrente nel tratto di passività (itp) varia di un ordine di grandezza tra un provino e l’altro, facendo registrare i valori inferiori per i provini caratterizzati da potenziali di break down più elevati, e alte densità di corrente per i provini BJ e, soprattutto, MEX.
Tab.2 - Potenziale di break-down, Potenziale di ripassivazione, Potenziale di corrosione libera, Densità di corrente nel tratto di passività / Break-down potential, Repassivation potential, Corrosion potential, Current density in passivity region.
TECNOLOGIA
La resistenza alla corrosione localizzata è stata valutata anche attraverso la determinazione della temperatura critica di pitting (CPT). Prima dell’inizio di queste prove, il potenziale di corrosione libera è stato monitorato fino alla stabilizzazione del valore, alla temperatura costante di 0°C. Come atteso, per tutti i provini si osserva la diminuzione del potenziale di corrosione al crescere del tenore
di cloruri in soluzione (figura 6); alla concentrazione di cloruri più bassa tutti i provini hanno valori di potenziale di corrosione simili, con l’eccezione del provino MEX, nettamente meno nobile.
Aumentando la concentrazione di cloruri a 1M (come previsto dalla normativa ASTM G-150) i provini LPBF, HR e SM mantengono potenziali di corrosione inferiori di circa
50 mV rispetto alla soluzione più diluita, mentre gli altri provini subiscono una netta snobilitazione (tabella 3). Inoltre, i provini MEX e BJ mostrano delle ampie oscillazioni del potenziale di corrosione, indice di instabilità del film passivo. La successiva polarizzazione al potenziale di +800 mV vs SCE, come previsto dalla normativa ASTM G-150, porta all’innesco immediato della corrosione localizzata per i provini CR, HR, MEX e BJ, per i quali la temperatura critica di pitting (CPT) risulta inferiore a 0°C. Un comportamento nettamente migliore è fatto registrare dai provini
LPBF e SM (tabella 3 e figura 7). I valori di CPT dei provini
SM e LPBF sono in accordo con i dati riportati da [15].
Le condizioni di prova della normativa ASTM G-150 sono molto severe; Nakhaie et al. [15] suggeriscono di abbas-
sare il potenziale applicato ai provini, poiché anche a potenziali inferiori si ha una frequenza di formazione di pit metastabili sufficientemente elevata da rendere statisticamente valido il test. Secondo gli stessi autori, infatti, il valore di CPT non dipende dall’innesco del pit, ma dall’attivazione del pit dalla condizione di metastabilità. Altri autori affermano invece che la CPT dipende sia dall’innesco sia dalla transizione, con il primo meccanismo prevalente nel caso di bassa concentrazione di cloruri [16, 17, 18]. Per cercare di discriminare meglio tra i provini prodotti con le diverse tecnologie, le prove CPT sono state ripetute in soluzione a minor concentrazione di cloruri.
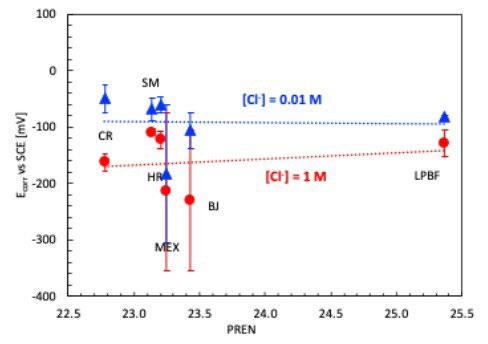
Fig.6 - Potenziale di corrosione prove CPT NaCl 1M e NaCl 0.01M / Corrosion potential in NaCl 1M and NaCl 0.01M solution at T 0°C.
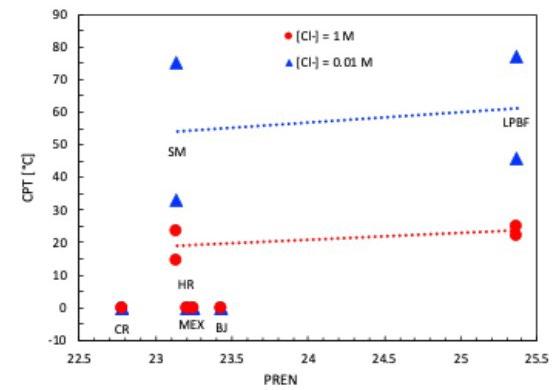




La diminuzione di concentrazione di NaCl a 0.01M porta al raggiungimento di temperature CPT più elevate per i provini LPBF e SM, anche se ne aumenta ulteriormente la variabilità, mentre i campioni HR, CR, MEX e BJ confermano i risultati ottenuti alla concentrazione 1M, con CPT inferiori a 0°C. Nel caso delle prove di CPT la morfologia di corrosione è da considerarsi in ogni caso di pitting sulla superficie esposta e non al di sotto della guarnizione come nel caso delle prove di polarizzazione potenziodinamica, in quanto è stato impiegato un setup di prova con una cella a flusso aperto, come suggerito nella normativa di riferimento ASTM G150, con lo scopo di eliminare il crevice nella zona di tenuta della guarnizione.
Vengono riportati in figura 8 e 9 due campioni a titolo rappresentativo della morfologia di corrosione, verificatasi anche nel caso delle altre tecnologie. I valori di CPT sono, analogamente ai potenziali di break-down, poco influenzati dal valore di PREN nell’intervallo di composizione delle leghe oggetto di questa sperimentazione. Infatti, nel caso dei provini SM, nonostante il PREN sia simile a quelli HR e inferiore ai BJ, si sono osservate delle temperature critiche di pitting più elevate, paragonabile a quelle ottenute per LPBF, che però presenta un PREN superiore di due punti.
Fig.3 - Comparison of training loss profiles vs. iteration number for the baseline (left) and the optimised model (right), trained on combined training and validation data.
I risultati delle prove di polarizzazione potenziodinamica in soluzione di NaCl deaerata e le prove CPT sono pertanto concordi nell’evidenziare il peggior comportamento nei confronti della corrosione localizzata dei provini ottenuti con le tecnologie BJ e MEX, malgrado i valori PREN molto simili. È quindi necessario un ulteriore approfondimento sperimentale, per valutare l’effetto della macrostruttura con porosità elevata e della microstruttura a grano grosso, oltre alla presenza di seconde fasi e inclusioni affioranti. In aggiunta si sottolinea come la morfologia della porosità della tecnologia MEX, continua e strutturata, direttamente correlata al processo produttivo del materiale stesso, risulti diversa rispetto a quella isolata e discontinua della tecnologia BJ, influenzando quindi la morfologia di corrosione. Questo condiziona inevitabilmente i risultati ottenuti, rendendo l’interpretazione degli stessi più complessa rispetto
a quanto possibile concludere per le leghe tradizionali di riferimento.
Al contrario, i provini realizzati tramite LPBF, grazie al PREN maggiore, alla macrostruttura con ridotta porosità e alla microstruttura a grano più fine con assenza di seconde fasi e inclusioni, hanno un comportamento a corrosione localizzata migliore di CR e HR, e confrontabile con il prodotto laminato SM.
I comportamenti osservati sulla lega 316L ottenuta mediante diverse tecnologie di additive manufacturing hanno confermato l’esigenza di definire e sviluppare protocolli idonei per la qualificazione a corrosione delle leghe ottenute con queste tecnologie, molto diverse l’una dall’altra, che tengano conto della specificità del processo produttivo e dei trattamenti di post processo specifici per le leghe stesse. In aggiunta, riveste particolare importanza lo sviluppo di trat-
tamenti superficiali idonei per la mitigazione di fenomeni di corrosione localizzata che si possono innescare a causa delle porosità affioranti che, specialmente per le tecnologie MEX e BJ, sono da considerare come difetti endogeni del materiale e fisiologici per il processo specifico.
Sono state condotte prove sperimentali sulla lega inossidabile AISI 316L, ottenuta mediante diverse tecnologie di additive manufacturing, al fine di evidenziare l’effetto sul comportamento a corrosione localizzata. I risultati delle prove confermano che:
• la lega inossidabile AISI 316L ottenuta mediante LPBF possiede una resistenza alla corrosione localizzata equivalente o perfino superiore a quella delle leghe tradizionali.
• Le tecnologie MEX e BJ mostrano una resistenza alla corrosione localizzata inferiore rispetto a SM e LPBF. Per queste tecnologie (MEX e BJ) è necessario valutare la messa a punto di trattamenti di post processo idonei per la mitigazione di fenomeni di corrosione localizzata ascrivibili alle specificità del processo produttivo stesso (network poroso, porosità isolate affioranti, seconde fasi).
• Si ribadisce l’esigenza di mettere a punto protocolli idonei per la qualificazione a corrosione delle leghe ottenute per additive manufacturing che tengano conto della specificità del processo produttivo e dei trattamenti di post processo specifici per le leghe stesse.
[1] A. Vafadar, F. Guzzomi, A. Rassau, K. Hayward, «Advances in Metal Additive Manufacturing: A Review of Common Processes, Industrial Applications, and Current Challenges», Appl. Sci., vol. 11, fasc. 3, p. 1213, 2021, doi: 10.3390/app11031213.
[2] T. M. Tavares, G. M. D. Ganga, M. Godinho Filho, V. P. Rodrigues, «The benefits and barriers of additive manufacturing for circular economy: A framework proposal», Sustain. Prod. Consum., vol. 37, pp. 369–388, mag. 2023, doi: 10.1016/j.spc.2023.03.006.
[3] A. Sola, A. Nouri, «Microstructural porosity in additive manufacturing: The formation and detection of pores in metal parts fabricated by powder bed fusion», J. Adv. Manuf. Process., 2019, doi: 10.1002/amp2.10021.
[4] M. Ziaee, N. B. Crane, «Binder jetting: A review of process, materials, and methods», Addit. Manuf., vol. 28, pp. 781–801, ago. 2019, doi: 10.1016/j.addma.2019.05.031.
[5] K. Sangoi, M. Nadimi, J. Song, Y. Fu, «Heat Treatment Effect on the Corrosion Resistance of 316L Stainless Steel Produced by Laser Powder Bed Fusion», Metals, vol. 15, fasc. 1, p. 41, gen. 2025, doi: 10.3390/met15010041.
[6] V. Salarvand et al., «Microstructure and corrosion evaluation of as-built and heat-treated 316L stainless steel manufactured by laser powder bed fusion», J. Mater. Res. Technol., vol. 18, pp. 4104–4113, mag. 2022, doi: 10.1016/j.jmrt.2022.03.156.
[7] J. I. Ahuir-Torres et al., «A Study of the Corrosion Resistance of 316L Stainless Steel Manufactured by Powder Bed Laser Additive Manufacturing», Appl. Sci., vol. 14, fasc. 17, p. 7471, gen. 2024, doi: 10.3390/app14177471.
[8] T. Voisin et al., «Pitting Corrosion in 316L Stainless Steel Fabricated by Laser Powder Bed Fusion Additive Manufacturing: A Review and Perspective», JOM, vol. 74, fasc. 4, pp. 1668–1689, apr. 2022, doi: 10.1007/s11837-022-05206-2.
[9] S. Mirzababaei, S. Pasebani, «A Review on Binder Jet Additive Manufacturing of 316L Stainless Steel», J. Manuf. Mater. Process, 2019, 3, 82. https://doi.org/10.3390/jmmp3030082.
[10] P. Kaae, E. Eikeland, «Corrosion Performance of Additively Manufactured Stainless Steel by Binder Jetting», IOP Conf. Ser. Mater. Sci. Eng., vol. 1249, p. 012051, lug. 2022, doi: 10.1088/1757-899X/1249/1/012051.
[11] R. Santamaria, K. Wang, M. Salasi, M. Iannuzzi, M. Y. Mendoza, M. Z. Quadir, «Stress Corrosion Cracking of 316L Stainless Steel Additively Manufactured with Sinter-Based Material Extrusion», Materials, vol. 16, fasc. 11, p. 4006, gen. 2023, doi: 10.3390/ ma16114006.
[12] S. Lorenzi, G. D’Urso, L. Nani, M. Quarto, S. Fest-Santini, T. Pastore, «Effect of the deposition strategy and endogenous defect pattern on the plastic deformability and the fracture mechanism of 316L stainless steel obtained using material extrusion», Eng. Fail. Anal., vol. 171, p. 109395, apr. 2025, doi: 10.1016/j.engfailanal.2025.109395.
[13] A. Racot, I. Aubert, M. Touzet, S. Thiebaut, M. Demesy, «Statistical analysis of the pitting corrosion induced by potentiostatic pulse tests of wrought and SLM 316L stainless steels», Corros. Sci., vol. 197, p. 110036, apr. 2022, doi: 10.1016/j.corsci.2021.110036.
[14] L. B. Coelho et al., «Estimating pitting descriptors of 316 L stainless steel by machine learning and statistical analysis», Npj Mater. Degrad., vol. 7, fasc. 1, p. 82, ott. 2023, doi: 10.1038/s41529-023-00403-z.
[15] D. Nakhaie, A. Imani, M. Autret, R. F. Schaller, E. Asselin, «Critical pitting temperature of selective laser melted 316L stainless steel: A mechanistic approach», Corros. Sci., vol. 185, p. 109302, giu. 2021, doi: 10.1016/j.corsci.2021.109302.
[16] T. Li, J. R. Scully, G. S. Frankel, «Localized Corrosion: Passive Film Breakdown vs Pit Growth Stability: Part V. Validation of a New Framework for Pit Growth Stability Using One-Dimensional Artificial Pit Electrodes», J. Electrochem. Soc., vol. 166, fasc. 11, p. C3341, giu. 2019, doi: 10.1149/2.0431911jes.
[17] J. S. Noh, N. J. Laycock, W. Gao, D. B. Wells, «Effects of nitric acid passivation on the pitting resistance of 316 stainless steel», Corros. Sci., vol. 42, fasc. 12, pp. 2069–2084, dic. 2000, doi: 10.1016/S0010-938X(00)00052-4.
[18] Y. Hou, Y. Nakamori, K. Kadoi, H. Inoue, H. Baba, «Initiation mechanism of pitting corrosion in weld heat affected zone of duplex stainless steel», Corros. Sci., vol. 201, p. 110278, giu. 2022, doi: 10.1016/j.corsci.2022.110278.
The aim of the present work is to evaluate the localized corrosion behaviour of an AISI 316L stainless steel alloy produced using different additive manufacturing technologies, comparing it with products manufactured through conventional way. Potentiodynamic polarization and critical pitting temperature (CPT) tests were carried out. The results of the potentiodynamic polarization tests showed differences in breakdown potentials between materials produced with additive technologies and those produced by traditional methods. The CPT tests yielded results consistent with those observed in the potentiodynamic tests. The results of CPT tests are in agreement with the potentiodynamic tests, confirming that the pitting corrosion behaviour is strictly dependent upon the additive manufacturing technologies. The article stresses once more the importance of defining suitable protocols for the corrosion qualification of alloys obtained by additive manufacturing that take into account the specificity of the production process and the treatments performed on the alloys themselves.
KEYWORDS: 316; ADDITIVE MANUFACTURING; PITTING; CREVICE; CORROSION;
S. Hägg Mameng, B. Helmersson, E. Johansson, S. Hosseinpour
Pitting engineering diagrams have been developed for various grades of stainless steel, informed by extensive longterm electrochemical testing, to assist in the selection of appropriate materials for a range of water applications. Key factors affecting pitting resistance of stainless steel include alloy composition, chloride ion concentration, temperature, and the oxidizing potential of the water, which can differ across contexts, such as sterile tap water versus slightly chlorinated environments.
This paper introduces pitting resistance engineering diagrams as stainless steel selection tools for water applications. The diagrams assist in the selection process of stainless steel and are further supported by relevant case studies that showcase its practical utility and applicability in real-world scenarios. The engineering diagrams provide a visual reference to define the pitting resistance of various stainless steel grades, while case studies demonstrate the performance of these materials in water-related environments. Overall, this selection tool aims to improve decisionmaking for engineers and designers involved in aquatic applications, ensuring the longevity and reliability of stainless steel components in challenging conditions.
KEYWORDS: PITTING ENGINEERING DIAGRAM; WATER APPLICATION; STAINLESS STEEL; PITTING CORROSION; CHLORIDE ION; TEMPERATURE; OXIDIZING POTENTIAL OF WATER SYSTEM;
Localized corrosion (e.g., pitting and crevice corrosion) is one of the most critical degradation mechanisms in stainless steel, as it can initiate locally, grow rapidly, and ultimately compromise structural integrity. The resistance of stainless steel to localized corrosion depends upon both the material’s characteristics, such as alloying elements and surface conditions, and the environment it is exposed to. In aquatic environments, factors such as chloride ion concentration, temperature, and solution pH are crucial factors to consider when determining the appropriate grade of stainless steel for a particular application [1-21]. However, the corrosivity is also significantly influenced by the oxidizing potential of the system. For instance, the open-circuit potential (OCP) in a sterile tap water system can vary widely, typically between 100 and 200 mV vs saturated calomel electrode (SCE), whereas in chlorinated water systems, the opencircuit potential increases with chlorine concentration, reaching approximately 700 mV vs SCE [2-11]. In natural water environments, the OCP may also rise, attributed
Sukanya Hägg Mameng, Björn Helmersson, Elisabeth Johansson
Avesta R&D Center, Outokumpu Stainless AB, Avesta, Sweden
Saman Hosseinpour
Krefeld R&D Center, Outokumpu GmbH, Krefeld, Germany
to more efficient cathodic reactions, and can reach levels around 400 mV vs SCE, depending on the water quality [2-5]. In this regard, pitting engineering diagrams serve as valuable tools that facilitate the selection of appropriate materials for specific environments and applications [1, 22-25].
A method for generating pitting engineering diagrams has been proposed based on electrochemical laboratory testing [12, 15, 19-25]. The various electrochemical approaches utilized in prior research have effectively established pitting diagrams that delineate the boundaries between pitting and non-pitting conditions with respect to chloride concentration, temperature, and the oxidizing potential of the system. Previous studies indicate that the electrochemical procedure can also be used for ranking different grades of stainless steel based on their resistance to localized corrosion. Long-term potentiostatic laboratory testing over 30 days yields the most conservative data, as the extended duration allows for a prolonged incubation period, making it the most representative method for simulating real-world applications. It was found that the incubation time for pitting initiation is a crucial factor to consider [12, 15]. Generally, lower temperatures correspond to longer incubation times before a pit can initiate and begin to grow. To thoroughly account for both the incubation time and the oxidative power of the system, the engineering diagrams in figure 2 were developed based on extensive laboratory testing conducted over a 30-day period at two
distinct potentials: 150 mV and 400 mV vs SCE. These oxidising potentials correspond to sterile tap water and slightly chlorinated water (or water with some bacterial activity), respectively [1, 2, 4, 5, 12, 16, 17]. Further details regarding the testing methodology can be found in references [12, 16, 17].
Briefly, after determining the open-circuit potential (OCP) in the corresponding electrolyte, the potential is swept from EOCP to the target potential (Eapp) of 150 mV or 400 mV vs SCE, while maintaining a constant electrolyte temperature throughout the test duration (i.e., 30 days).
All other parameters, such as material geometry (30 x 60 mm), exposed area (40 cm2), and surface condition (#320 mesh ground), are kept constant for better comparison. To establish such pitting engineering diagrams, the chloride concentration and temperature were varied to assess the boundary between pitting and non-pitting conditions. As shown in figure 1, an increase in potential enhances the aggressiveness of the water, leading to a downward shift in the pitting corrosion boundary on the diagrams. Figure 1 presents all the studied grades, where each diagram represents only one potential. Notably, the grades Ultra 254 SMO (EN 1.4547) and Forta SDX 2507 (EN 1.4410) are exclusively included in figure 1B, as they exhibit adequate pitting resistance in all tested conditions at the lower potential of 150 mV vs SCE (see figure 1A) [1, 12, 15, 17].
Table 1 presents details of the chemical composition and the pitting resistance equivalent number (PREN = %Cr + 3.3%Mo + 16%N).[1].
Tab.1 - Typical chemical compositions of the stainless steels investigated (wt%). The alloys are given in order of increasing PREN within each group.
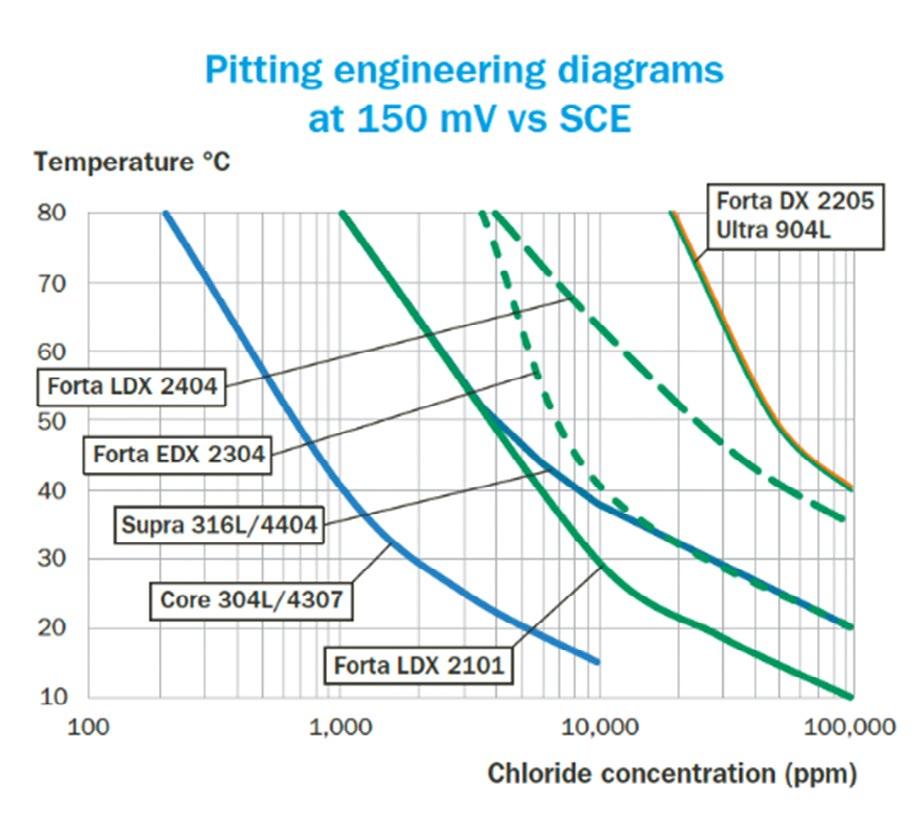
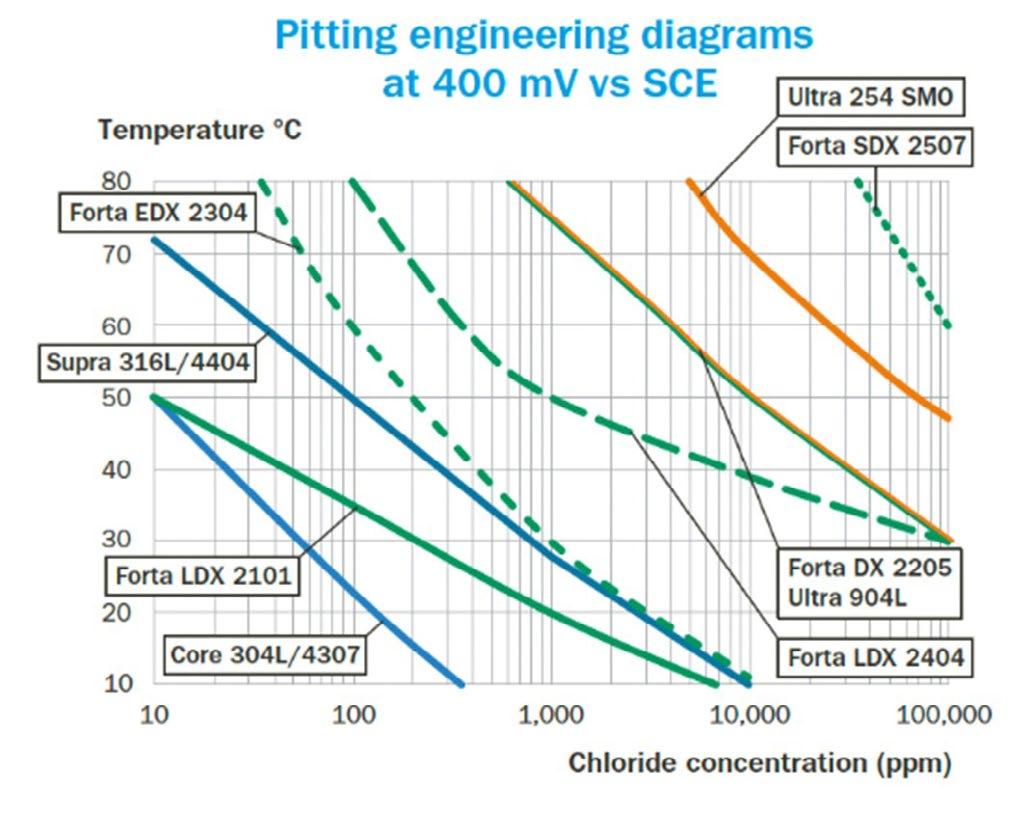
Fig.1 - Pitting engineering diagrams indicating the pitting resistance boundaries for various stainless steels in two different water systems. a) 150 mV vs SCE simulating sterile tap water, and b) 400 mV vs SCE representing slightly chlorinated water or water with some biological activity [1, 15].
Please be aware that these diagrams provide only approximate guidance regarding the resistance of the respective grades. The final selection will depend on several additional factors, many of which are discussed in this section. It is essential to recognise that actual service conditions may vary significantly from those used to create the diagram. For instance, the presence of crevices or weld oxides in the construction can impact the performance of stainless steel. Crevice corrosion typically initiates more readily than pitting corrosion, which is why the lines in the diagram are set to more conservative values in such cases [1, 4, 5, 22, 24]. Additionally, if present in the water, certain contaminants or substances may facilitate or inhibit the corrosion process, further influencing outcomes. In the following sections, case studies are presented to demonstrate the consistency between the results of the pitting engineering diagram and the materials’ performance in real applications or field tests.
Case 1: Long-term immersion test for domestic water heater application
Stainless steels are widely used in domestic water heaters due to their natural corrosion resistance in potable water,
eliminating the need for linings or cathodic protection. This study involved long-term corrosion tests conducted over a period of one year in water containing 250 ppm chlorides at a temperature of 75 °C. The materials evaluated included welded specimens of standard austenitic grades (Core 304L/4307 and Supra 316L/4404) as well as duplex grades (Forta LDX 2101, DX 2304, LDX 2404, and DX 2205).
The focus of the assessment was specifically on pitting corrosion [26].
The long-term laboratory results were compared with pitting engineering diagrams and real-world experiences involving stainless steel in domestic water heaters. A summary of the observations for the various specimens involved in this study can be found in figure 3. After one year of exposure, it was concluded that pitting corrosion is unlikely to occur on the tested grades in a 250 ppm Cl- environment at 75 °C, except for Core 304L/4307 (EN 1.4307). Over the one year, no significant weight loss was observed in any of the specimens; however, some discolouration was observed, as illustrated in figure 2. Overall, the results for this environment correlate well with the pitting engineering diagram at 150 mV vs. SCE, unless the potential is raised, e.g., through chlorination.






circle)
Fig.2 - The results and appearance from testing various welded specimens at a 250-ppm chloride ion concentration at 75ºC over the course of one year. The red circle shows the area with the most severe pitting corrosion.
Field testing in North Sea seawater (near Helgoland) has been conducted, focusing on four grades of duplex stainless steel: Forta EDX 2304 (EN 1.4362), Forta LDX 2404 (EN 1.4662), Forta DX 2205 (EN 1.4462), and Forta SDX 2507 (EN 1.4410). These materials were subjected to immersion tests in the seawater over a span of two years [27]. During the testing period, the average chloride concentration in the seawater was found to be 18.5 g/L. Additionally, documented annual fluctuations in sea temperature, particularly near Helgoland, ranged from 5 °C to 20 °C. At the end of the 24-month study, all grades demonstrated significant biofouling on their surfaces, as illustrated in figure 3A. The magnitude of biofouling, which often induces microbiologically influenced corrosion (MIC), increased steadily throughout the testing duration. Literature indicates that the OCP rises by approximately 300-400 mV vs. SCE after an initial incubation period in seawater under varying climatic conditions [1, 28]. After cleaning and the removal of the biofilm and marine organisms, only staining remained, with no pitting
observed (see figure 3B). This finding aligns well with the pitting engineering diagram, indicating that a 400-mV vs SCE increase in potential is not sufficient to cause pitting on such grades according to the pitting engineering diagram. The pitting - no pitting borderline in figure 1B for grade Forta EDX 2304 (EN 1.4362) is very close to the seawater chloride concentration and the mean temperature of 10 °C. The lack of pitting corrosion on this grade in the field exposure is related to the conservative approach in generating the pitting engineering diagrams and uncertainty in the boundary between pitting susceptibility and resistance (see figure 1).

(3A) Before cleaning

(3B) After cleaning
Fig.3 - An overview of the assembly of the samples after 24 months of field exposure, before (A) and after (B) cleaning. Samples from left to right in each panel are: Forta EDX 2304 (EN 1.4362), Forta LDX 2404 (EN 1.4662), Forta DX 2205 (EN 1.4462), and Forta SDX 2507 (EN 1.4410).
Case 3: Long-term immersion test for brine handling applications
As desalination technologies advance and the concentration of corrosive chlorides in rejected brine increases, a critical inquiry arises regarding the corrosivity limits of commonly utilized stainless steel grades, specifically duplex Forta SDX 2507 (EN 1.4410). This investigation aimed to assess the applicability of grade 2507 for brine environments characterized by high chloride levels.
In this study, welded tube samples of duplex Forta SDX 2507 (EN 1.4410) were exposed to sodium chloride solutions at 50,000 and 70,000 ppm chloride ion concentration at 40 °C
for one month. Measurements of open-circuit potential were recorded, yielding values ranging from 250 to 400 mV versus SCE (see figure 4) [4]. These findings are in alignment with the recognized threshold of 400 mV vs. SCE, as illustrated in figure 1B. Furthermore, the potential measurements were corroborated by visual inspections utilizing a light optical microscope, which revealed no traces of pitting corrosion across all tested specimens. The results indicate that grade Forta SDX 2507 is a promising option for applications in environments with elevated chloride concentrations, meeting the established 400 mV vs. SCE criteria.
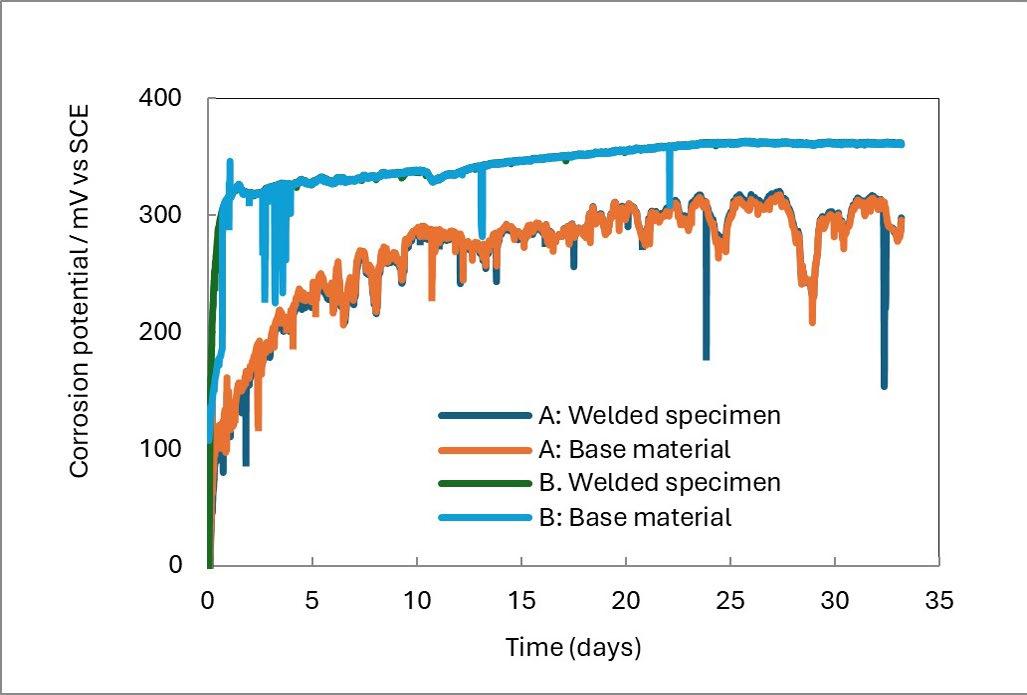
Fig.4 - Corrosion potentials of specimens in Forta SDX 2507 (EN 1.4410) in the experiment with 50000 mg/L of chlorides at 40 °C. [4]
The failure case of heat exchanger tubes made from austenitic stainless steel grade Supra 316L/4404 within the food industry underscores critical concerns regarding material performance under specific environmental conditions. Notably, these tubes exhibited failure within a mere four weeks of operation, predominantly manifesting as red spots and localized pitting on the outer walls, as depicted in figure 5.
During this operational period, the tubes were cooled with cooling water containing 100 mg/L (ppm) chloride ions, which had been treated with chlorine dioxide. The tube wall temperatures ranged from 60 °C to 65 °C.
Initial corrosion assessments indicated a low opencircuit potential in the tube heater system, which would indicate that the 150-mV vs SCE line (Dark blue) in figure 1A could be used. This suggested that the likelihood of pitting corrosion was minimal at that time. However, an alarming development occurred after four weeks of service: the open-circuit potential increased, attributed to the oxidative effects of the chlorination process, in

which case the 400-mV vs SCE line (Dark blue, figure 1B) would be more applicable, which would indicate a shift towards conditions conducive to pitting corrosion on Supra 316L/4404. In this system, figure 1B (400 mV vs SCE diagram) could be used to select a more appropriate material.
The primary factors influencing this corrosion scenario include elevated operational temperatures, the chloride concentration in the cooling water, and chlorination treatment, which collectively augment the risk of pitting.
To address these corrosion challenges, it is critical to improve water-quality management within the cooling system. Furthermore, transitioning to a higher-alloy material, such as duplex grade Forta LDX 2404 (EN 1.4662) or Forta DX 2205 (EN 1.4462), is advisable and would offer improved corrosion resistance.
In summary, this case highlights the importance of selecting suitable materials and maintaining optimal environmental conditions to prevent premature failure, e.g., in heat exchangers, particularly in sensitive industries such as food processing.
A pilot plant for condenser tubes utilizing duplex grade Forta DX 2304 (EN 1.4362) was constructed; however, this material experienced failure after merely nine months of operation within the wastewater system. Corrosion
damage was evident on both the interior and exterior surfaces of the outer tube walls, with the most severe deterioration occurring on the exterior surfaces, as depicted in figure 6.
The investigation revealed that the primary cause of the corrosion stemmed from a combination of high
temperatures (ranging from 65 °C to 125 °C) and water containing elevated concentrations of corrosive species, particularly chloride ions (ranging from 28,000 ppm to 48,000 ppm). These conditions exceed the corrosion resistance capabilities of Forta DX 2304, as indicated in figure 1A. The engineering diagram in figure 1A further illustrates that the operational conditions—specifically the chloride concentration and temperature—during

production surpassed the limiting thresholds beyond which Forta DX 2205 (1.4462) is prone to pitting corrosion. To mitigate the risk of localized corrosion, it may be prudent to consider utilizing a higher-alloyed material, such as Forta SDX 2507 (EN 1.4410) or Ultra 254 SMO (EN 1.4547), for this system.

Key factors influencing pitting resistance of stainless steel in water applications include alloy composition, chloride ion concentration, temperature, and the oxidizing potential of the water. In this regard, pitting engineering diagrams are valuable assets for the selection of the optimum stainless steel in corresponding applications. The accuracy of these diagrams has been validated through various case studies, which support their reliability and performance. These studies encompass investigations into failure analysis and long-term testing conducted in both controlled laboratory settings and real-world environments. The focus is on identifying the factors behind pitting failures and evaluating the applicability and reliability of engineering diagrams over extended periods. Findings from these investigations contribute to improved design practices and the optimization of material selection across diverse applications and
support the validation of pitting engineering diagrams for water applications. This selection tool can be a primary tool for facilitating decision-making for engineers and designers involved in water applications, ensuring the longevity and reliability of stainless steel components in challenging conditions. However, it should be noted that while these diagrams highlight factors such as chloride ion concentration, temperature, and oxidizing potential, they do not account for all variables affecting corrosion, including surface conditions, environmental fluctuations, pH and other contaminants.
[1] Corrosion Handbook, 12th Edition, 2023; Outokumpu Oyj, Finland.
[2] S. Hägg Mameng, R. Pettersson, “Localised corrosion of stainless steels depending on chlorine dosage in chlorinated water”, Proceedings Eurocorr 2011. Presented at the EUROCORR 2011, 4-8 September 2011, Stockholm, Sweden. Retrieved from https:// urn.kb.se/resolve?urn=urn:nbn:se:kth:diva-263815.
[3] S. Ahmad, A. U. Malik, “Corrosion behaviour of some stainless steels in chlorinated Gulf seawater”, Journal of Applied Electrochemistry, vol. 31 (9), pp. 1009–1016, 2001.
[4] E. Halling, S. Randström, “High alloyed stainless steels for brine handling applications”, in Singapore International Water Week (SIWW), 2010.
[5] S. Hägg Mameng, A. Bergquist, E. Johansson. “Corrosion of Stainless Steel in Sodium Chloride Brine Solutions”, CORROSION (2014)
[6] CP. Cutler, “Stainless steels and the water industry: From knowledge to applications”, in 6th European Stainless Steel Conference Science and Market, Helsinki, Finland, 2008, 395–400.
[7] G. Ventura, E. Traverso, A. Mollica, “Effect of NaClO Biocide additions in natural seawater on stainless steel corrosion resistance”, Corrosion, vol. 45 (4), pp. 319-325, 1989
[8] P. D. Goodman, “Effect of chlorination on materials for sea water cooling systems: a review of chemical reactions”, British Corrosion Journal, vol. 22 (1), pp. 56-62, 1987
[9] Y. C. Lu, J. L. Luo, M. B. Ives, “The influence of chlorination of saline environments on localized corrosion of stainless steels”, ISIJ International, vol. 31 (2), pp. 210-215, 1991.
[10] R. Franceis, J. B. Irwin, G. Byrne, “Repassivation of high alloy stainless steels in chlorinated sea water”, British Corrosion Journal, vol. 30 (3), pp. 237-242, 1995.
[11] M. H. Moayed, M. Golestanipour, “An investigation on the effect of bleaching environment on pitting corrosion and transpassive dissolution of 316 stainless steel”, Materials and Corrosion, vol. 56 (1), pp. 39-43, 2005.
[12] S. Hägg Mameng et al., “Methods for construction of pitting engineering diagrams of stainless steels used for water systems”, in AMPP Annual conference, Denver, Colorado, USA, 2023, Paper C2023-19246.
[13] C. O. Olsson, D. Landolt, “Passive films on stainless steels - chemistry, structure and growth,” Electrochimica Acta, vol. 48, (9), pp. 1093-1104, 2003.
[14] D. G. Li, J. D. Wang, D. R. Chen, “Influence of pH value on the structure and electronic property of the passive film on 316L SS in the simulated cathodic environment of proton exchange membrane fuel cell (PEMFC)”, International Journal of Hydrogen Energy, vol. 39, pp. 20105-20115, 2014.
[15] S. Hägg Mameng, L. Wegrelius, S. Hosseinpour, Stainless Steel Selection Tool for Water Application: Pitting Engineering Diagrams. Front. Mater. 2024, 11, doi:10.3389/fmats.2024.1353907.
[16] S.H. Mameng, R. Pettersson, “Limiting conditions of pitting corrosion of stainless steel EN 1.4404 (316L) in terms of temperature, potential and chloride concentration”, EUROCORR 2013, Estoril, Portugal, 2013.
[17] S. H. Mameng, R. Pettersson, “Limiting conditions of pitting corrosion for lean duplex stainless steel as a substitute for standard austenitic grades”, EUROCORR 2014, Pisa, Italy, 2014.
[18] E. A. Abd El Meguid, N. A. Mahmoud, V. K. Gouda. "Pitting corrosion behaviour of AISI 316L steel in chloride-containing solutions", Br. Corros. J., 33, 1 (1998): pp. 42-48.
[19] J. W. Fielder, D. R. Johns, "Pitting Corrosion Engineering Diagrams for Stainless Steels", British Steel Technical, (1989): pp. 9-14.
[20] N. J. Laycock, R. C. Newman, "Temperature dependence of pitting potentials for austenitic stainless steels above their critical pitting temperature", Corr. Sci., 40, 6 (1998): pp. 887-902.
[21] H. P. Leakie, H. H. Uhlig, "Environmental factors affecting the critical potential for pitting in 18-8 stainless steel", J. Electro. Soc., 113, 12 (1966), pp. 1262-1267.
[22] Outokumpu Corrosion Handbook, 11th edition; 2015.
[23] The Euro Inox Handbook of Stainless Steel, Euro Inox, Luxembourg, First Edition, 2002.
[24] ASM International. (2005). ASM Handbook, Volume 13B: Corrosion—Materials: Corrosion of Wrought Stainless Steels. ASM International. ISBN: 978-0-87170-707-9.
[25] S. M. Sharland, ¨A review of the theoretical modelling of crevice and pitting corrosion¨, Corrosion Science. vol. 27. pp. 289–323, 1987.
[26] E. Johansson, S. Hägg Mameng, “Long-term corrosion test of stainless steel for water heater applications”, EUROCORR 2014, Pisa, Italy, 2014.
[27] S. Hosseinpour, S. Hägg Mameng, M. Almen and M. Liimatainen, “Field exposure of duplex stainless steel in the marine environment: the impact of the exposure zone”, Corros. Mater. Degrad. 2025, 6(4), 63; https://doi.org/10.3390/cmd6040063
[28] N. Larché, E. Diler, D. Thierry, “Crevice and Pitting Corrosion of Stainless-Steel and Nickel-Based Alloys in Deep Sea Water”, CORROSION (2019).
P. Forcellese, W. A. Khan, G. Giuliani, T. Bellezze
Binder Jetting (BJ) is an additive manufacturing technology that can produce alloy components at a higher speed and resolution than other systems. However, the localized corrosion resistance properties of BJ stainless steel are lower than those obtained with conventional manufacturing processes. This study aims to evaluate the effects of passivation treatments on the corrosion behavior of as-sintered 17-4 PH samples fabricated through BJ. The samples were treated with four acidic solutions: 15 %, 20 %, 40 % HNO ₃ , and 40 % HNO ₃ + 1 % HF. The localized corrosion resistance properties were evaluated through Cyclic Potentiodynamic Polarization (CPP) tests in a neutral pH sodium chloride electrolyte. The treatments significantly enhanced the localized corrosion resistance properties of as-sintered BJ samples, determining the typical passive anodic behavior of the CPP curve, which was not shown in the untreated samples. Moreover, higher concentrations of HNO3 (40%) improved the pitting corrosion resistance, while adding HF was ineffective. The study paves the way for broader industrial applications of additive-manufactured stainless steel, emphasizing the critical role of improving localized corrosion resistance properties through passive treatments.
KEYWORDS: LOCALIZED CORROSION; PASSIVATION TREATMENTS; BINDER JETTING; CYCLIC POTENTIODYNAMIC POLARIZATION;
Additive Manufacturing (AM) technologies have gained significant attention in recent years as an innovative technology capable of printing complex metallic components with reduced fabrication time and cost [1]. Among the different AM technologies, Binder Jetting (BJ) has emerged as a promising technique due to its high printing speed of a wide range of alloys and the absence of residual stress, which is typically induced by high-energy beam-based processes [2–5]. In BJ, a liquid binder is selectively deposited into layers of metallic powder to generate a green part, which is subsequently cured, depowdered, debound, and finally sintered at high temperatures around 1300 °C [6,7]. Besides these benefits, one of the main limitations of BJ lies in the intrinsic heterogeneous microstructure produced during sintering, which directly affect both mechanical and corrosion resistance properties [8–11]. The precipitation hardened martensitic Stainless Steel (SS) 17-4 PH is widely used in aerospace, petrochemical, and biomedical industries because of its good balance
of mechanical and corrosion resistance properties [12]. Conventionally manufactured 17-4 PH exhibits a primarily martensitic microstructure which provides high mechanical strength and acceptable corrosion resistance in moderately aggressive environments. On the other hand, BJ-fabricated and as-sintered 17-4 PH typically shows a mixed ferritic-martensitic microstructure with microporosities, secondary phases, and inclusions [13,14]. The samples investigated in this study were fabricated with the Shop System™ (Desktop Metal, Burlington, MA, USA) using a layer thickness of 75 µ m and sintered at 1300 °C under an argon-hydrogen atmosphere, exhibit porosities up to 0.5% and compositional segregations at grain boundaries [14].
Localized corrosion is a significant aspect that arises from the breakdown of the passive oxide film, which typically provides protection to the metal surface. In BJ-fabricated alloys, porosities, inclusions, and secondary phases act as preferential sites for pit initiation. In particular, elemental segregation around inclusions and grain boundaries strongly influences local stability of the passive film. In a recent study [15] demonstrates that segregation of Cu and Sb at inclusion-matrix interfaces promotes localized corrosion during pit initiation, while co-segregation of Cr and solute atoms at grain boundaries can produce Cr-depleted zones, making the alloy highly susceptible to intergranular corrosion [16]. Moreover, limited oxygen diffusion in pore regions impairs repassivation, accelerating pit and crevice propagation. As a result, corrosion resistance properties of as-sintered samples fabricated by BJ technology are generally inferior to their wrought counterparts, as shown in previous studies [13,14,17,18].
Most of the passivation treatments commonly involve nitric acid (HNO3) or nitric (HNO3) and (HF) acids mixtures to restore and stabilize passive films on stainless steels, which enhance resistance against localized corrosion [19–22]. While these treatments are well established for conventionally manufactured stainless steels, their application to additive-manufactured stainless steels is marginally studied.
The goal of this study is to evaluate the effect of different passivation treatments on the localized corrosion resistance of as-sintered 17-4 PH fabricated by BJ technology. Comparisons are made between untreated
and passivated BJ samples and wrought counterparts through Cyclic Potentiodynamic Polarization (CPP) tests in neutral sodium chloride solution.
Two sets of stainless-steel specimens were employed in this study:
1. Binder Jetting (BJ) 17-4 PH samples manufactured by using the Shop System™ (Desktop Metal, Burlington, MA, USA). The printing process was carried out with a layer thickness of 75 µ m, and after curing and depowdering, the samples were sintered at 1300 °C under an atmosphere of 97% Argon and 3% Hydrogen. The samples obtained are square plates with dimensions of 25 × 25 × 10 mm as shown in figure 1.
2. Wrought (Wr) 17-4 PH samples, used as a reference.
Before conducting the corrosion test, the specimens were mounted in acrylic resin to have one flat exposed surface. Surfaces of the samples were ground by using Silicon Carbide (SiC) papers up to 1200 grit size and cleaned in an ultrasonic bath with deionized water for 5 minutes and then normal hexane for 15 minutes. Before performing an electrochemical test, the samples rested for 24 hours. Then the exposed area (2.01 cm2) was defined by using polyimide tape with a circular aperture.
Passivation treatments were performed using four chemical solutions prepared as follows:
P1: 15 vol.% HNO ₃ in deionized water
P2: 20 vol.% HNO ₃ in deionized water
P3: 40 vol.% HNO ₃ in deionized water
P4: 40 vol.% HNO ₃ + 1 vol.% HF in deionized water
The samples were immersed in each solution for 30 minutes at room temperature. After the passivation treatment they were rinsed in deionized water in a sonicator for 5 minutes, to remove the residual chemical, and then dried with compressed air and lastly stored in a desiccator for 24 hours.

Fig.1 - Provini as-sintered di 17-4 PH ottenuti per Binder Jetting / The 17-4 PH BJ as-sintered specimens.
The CPP tests were carried out at room temperature in a neutral electrolyte having a concentration of 0.035 wt.% NaCl with the scan rate of 0.166 mVs-1
A typical three-electrode electrochemical cell setup was used in this study. The configuration included:
• Working electrode: the prepared stainless-steel sample
• Reference electrode: Saturated Calomel Electrode (SCE, +0.241 V vs SHE)
• Counter electrode: activated titanium wire
CPP scans were initiated 175 mV below the open circuit potential (OCP), sweeping towards positive values at 83 µ V/s until a current density of 0.1 mA·cm ² was reached.
The scan was then reversed until current density returned to near-passivation conditions.
The tests were repeated at least three times for each
sample type, and the most representative curve will be shown. Concerning the characteristic parameters obtained from the CPP curves, such as corrosion potential Ecorr, protection potential Eprot and pitting potential Epit, average values and the corresponding standard deviations were calculated.
After electrochemical measurement, post-corrosion surface morphology was examined by using optical microscopy (OM) to identify localized corrosion (pitting and crevice attack) associated with the samples submitted to different passivation treatments.
The average chemical composition (wt.%) of BJ-fabricated 17-4 PH and wrought sample measured by using Spark Analyzer (Spectrolab, Sylmar, CA, USA) at four different points is reported in table 1.
Tab.1 - Composizione chimica (% in peso) di un campione BJ as-sintered e wrought / Chemical composition (wt.%) of an as-sintered BJ sample and wrought.
Although, a detailed microstructural characterization of untreated samples has been extensively reported in previous work [13, 14], a summary of the main
microstructural features is provided to support the discussion of the electrochemical tests. The XRD diffractogram of the as-sintered sample reveals only
peaks commonly attributed to the body-centered cubic (BCC) phase, which is found in both ferrite and martensite [14], despite the latter having a distorted BCC structure in terms of a higher lattice cell parameter in the z-axis. The presence of hydrogen gas within the sintering chamber, along with pores and compositional heterogeneities at the grain boundaries, promotes ferrite stabilization during cooling rather than retention of austenite at room temperature [23]. In addition, OM and Scanning Electron Microscopy (SEM) revealed significant residual porosities together with elemental segregations, mainly involving Cu and Nb, which are preferentially located at the grain boundaries. These microstructural features are responsible for influencing electrochemical behavior by promoting localized passive film instability and corrosion initiation sites.
On the other hand, the wrought sample displays BCC peaks and Face-Centered Cubic (FCC) peaks, which correspond to retained austenite (FCC), representing a small fraction in volume, as observed in a previous study [13]. Consistently, the microstructure of wrought 17-4 PH is characterized by a finer and more homogeneous martensitic matrix, with a limited amount of retained austenite and δ -ferrite stringers. This comparatively dense and uniform microstructure, in contrast to the assintered BJ condition, is known to promote a more stable passive film formation and plays a key role in the different
electrochemical response observed between wrought and BJ samples.
The most representative CPP curves of wrought specimens before and after passivation are shown in figure 2: it can be observed that the passivation treatments did not improve their localized corrosion resistance, as the untreated material already exhibited a relatively stable passive behavior due to its dense and homogeneous martensitic microstructure as shown in figure 2a, similar results were found in other studies [18].This indicates that for wrought 17-4 PH, passivation treatments are not beneficial. The only beneficial effect of the passivation treatments that can be observed in figure 2 and 3 is the increase of the corrosion potential. Conversely, BJ as-sintered samples show a markedly different response. The untreated material does not display a well-defined passive region, owing to microstructural heterogeneities, residual porosities, and elemental segregation, other studies observed the similar results [14]. However, after passivation treatments, a well-defined passivation trait was observed in figure 3, especially at higher HNO3 concentrations, indicating that passivation effectively stabilizes the passive film by mitigating the effects of porosity and microstructural heterogeneities and thus improves pitting corrosion resistance. These results confirm that the effectiveness of passivation treatments is strongly governed by intrinsic microstructural characteristics of the material.
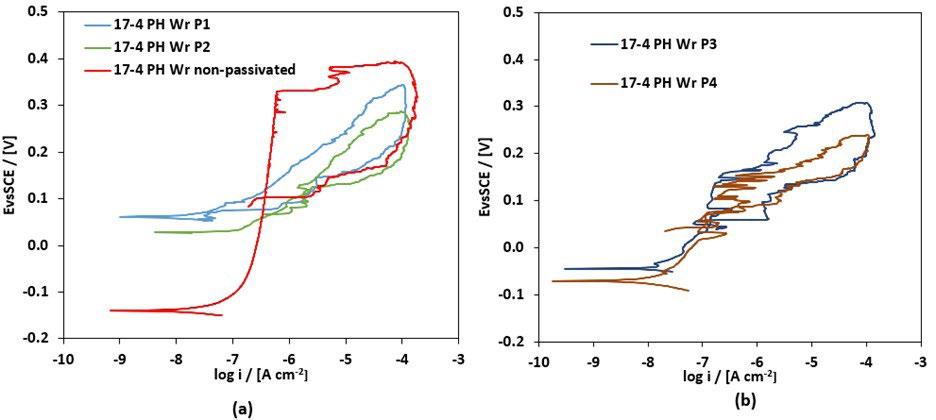
Fig.2 - Curve CPP dei campioni 17-4 PH wrought (Wr) dopo i trattamenti di passivazione: (a) campione non passivato, P1, P2; (b) P3, P4 / CPP curves of 17-4 PH Wr samples after passivation treatments: (a) non-passivated sample, P1, P2; (b) P3, P4.
All passivation treatments produced a well-defined passive region, which is more extended as the concentration of HNO3 increase from 15% (P1) to 40% (P3, P4). In particular, BJ samples passivated by P3 and P4 treatments supplied
comparable CPP curves as figure 3 shows, thus the same localized corrosion resistance of the obtained samples. This means that the addition of HF (P4) did not give any particular benefit to the passivation treatment.
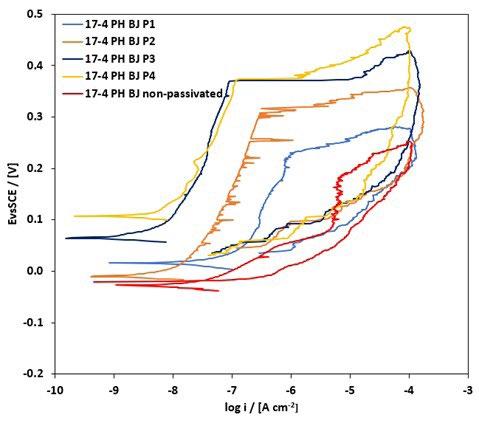
Fig.3 - Curve CPP dei campioni 17-4 PH ottenuti tramite Binder Jetting allo stato non passivato e dopo i trattamenti di passivazione / CPP curves of 17-4 PH BJ-fabricated samples in the non-passivated condition and after passivation treatments.
Figure 4 represents the characteristic potentials obtained from the CPP curves: Ecorr, Epit, and Eport. The passivation treatments produced a clear benefit in terms of corrosion resistance properties for BJ fabricated samples, particularly at high nitric acid concentrations, determining an increase of Epit. In figure 4, the perfect passivity region
corresponds to potentials less positive than Eprot, where the pitting corrosion cannot initiate and, furthermore, existing pits cannot propagate; the imperfect passivity region is defined by the potential range between E pit and Eprot, where pits cannot initiate but existing pits can propagate.
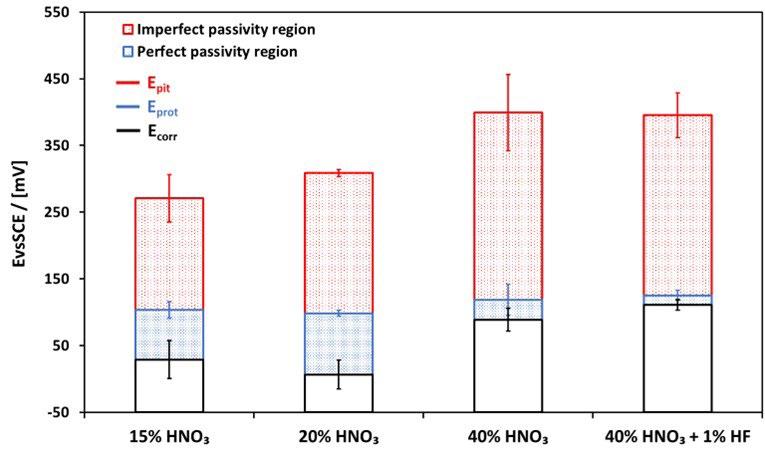
Fig.4 - Potenziali caratteristici (Epit, Ecorr ed Eprot) dei campioni ottenuti tramite Binder Jetting, dopo i trattamenti di passivazione / Characteristic potentials (Epit, Ecorr and Eprot) of BJ-fabricated samples after passivation treatments.
The OM was performed after the electrochemical tests. In a previous study [14], as-sintered BJ samples without passivation showed preferential initiation of localized corrosion, mainly in the form of pits and micro-crevice. However, after passivation treatments, the protective film significantly improved, resulting in a noticeable reduction in corrosion extension. Through OM observations, very few corrosion attacks were found like that shown in figure 5, for a sample treated with passivation P3, as an example, which is representative of the attacks found in the passivated samples: it affects a limited area of the sample and remains
very superficial because it does not seem to penetrate deeply, contrarily to what was previously observed in as-sintered samples. Although passivation effectively mitigates corrosion, based on these observations localized attacks may still occur at specific sites, in agreement with that is suggested by the anodic characteristics of the examined samples (figure 3).

Fig.5 - Immagini di microscopia ottica dopo prove CPP: campione BJ passivato P3 con attacco localizzato / Optical microscopy images after CPP tests: BJ sample passivated with P3 showing localized pitting.
From a previous study of the same authors, the as-sintered BJ-fabricated 17-4 PH samples were found to be characterized by a heterogeneous microstructure, which was considered responsible of a limited ability to form a stable passive film. Therefore, solutions to improve the localized corrosion resistance of these materials are of paramount importance. For this reason, this study has been done, where 17-4 PH in as-sintered condition was submitted to passivation treatments by different solutions of HNO3, adding in one case a small amount of HF. The present work demonstrated that passivation treatments significantly improve the localized
corrosion resistance of BJ-fabricated 17-4 PH stainless steel, while their effect on wrought counterparts is negligible or slightly detrimental. Electrochemical characterizations showed that, after passivation, a well-defined passive region appeared, particularly with 40% of HNO3 treatment. The addition of HF did not bring benefits. These results underline the novelty of applying passivation to BJ-fabricated stainless steels, proving it as an effective strategy to mitigate their intrinsic corrosion limitation and extend their potential for industrial use.
[1] A. Vafadar, F. Guzzomi, A. Rassau, K. Hayward, “Advances in Metal Additive Manufacturing: A Review of Common Processes, Industrial Applications, and Current Challenges”, Applied Sciences 2021, 11, doi:10.3390/app11031213.
[2] J. Kráľ, T. Dzuro, H. Debski, “Applying Binder Jetting Technology to 316L Stainless Steel Materials and Testing Its Mechanical and Dimensional Properties Depending on the Printing Method”, Materials 2024, 17, doi:10.3390/ma17174400.
[3] I. Bianchi, A. Forcellese, P. Forcellese, T. Mancia, C. Mignanelli, M. Simoncini, T. Verdini, “Effect of Printing Orientation Angle and Heat Treatment on the Mechanical Properties and Microstructure of Binder-Jetting-Printed Parts in 17-4 PH Stainless Steel”, Metals (Basel) 2024, 14, doi:10.3390/met14111220.
[4] B. Ibrahim, L. Lopez, S. Kulkarni, D. Jobes, M. Forgiarini, J. R. Barber, J. V. Gordon, “Increasing Strength Properties in Sinter-Based Additive Manufacturing of SS316L via Metal Material Jetting of Sub-Micron Powders”, Addit Manuf 2024, 89, 104268, doi:https://doi. org/10.1016/j.addma.2024.104268
[5] N. Lecis, R. Beltrami, M. Mariani, “Binder jetting 3D printing of 316 stainless steel: influence of process parameters on microstructural and mechanical properties”, La Metallurgia Italiana 2021, 2, 31-41.
[6] M. Li, W. Du, A. Elwany, Z. Pei, C. Ma, “Metal Binder Jetting Additive Manufacturing: A Literature Review”, Journal of Manufacturing Science and Engineering, Transactions of the ASME 2020, 142.
[7] F. Dini, S. A. Ghaffari, J. Javadpour, H. Rezaie, M. Shiri, “A Review of Binder Jet Process Parameters; Powder, Binder, Printing and Sintering Condition”, Metal Powder Report 2019, 75, doi:10.1016/j.mprp.2019.05.001.
[8] G. K. Meenashisundaram, Z. Xu, M. L. S. Nai, S. Lu, J. S. Ten, J. Wei, “Binder Jetting Additive Manufacturing of High Porosity 316L Stainless Steel Metal Foams”, Materials 2020, 13, doi:10.3390/ma13173744.
[9] P. Muthuswamy, “Additive Manufacturing of Tungsten Carbide Hardmetal Parts by Selective Laser Melting (SLM), Selective Laser Sintering (SLS) and Binder Jet 3D Printing (BJ3DP) Techniques”, Lasers in Manufacturing and Materials Processing 2020, 7, doi:10.1007/ s40516-020-00124-0.
[10] J. M. Alegre, A. Díaz, R. García, L. B. Peral, M. Lorenzo-Bañuelos, I. I. Cuesta, “Mechanical and Fatigue Properties of Ti-6Al-4V Alloy Fabricated Using Binder Jetting Process and Subjected to Hot Isostatic Pressing”, Materials 2024, 17, doi:10.3390/ma17153825.
[11] M. Xu, H. Guo, Y. Wang, Y. Hou, Z. Dong, L. Zhang, “Mechanical Properties and Microstructural Characteristics of 316L Stainless Steel Fabricated by Laser Powder Bed Fusion and Binder Jetting”, Journal of Materials Research and Technology 2023, 24, 4427–4439, doi:https://doi.org/10.1016/j.jmrt.2023.04.069
[12] H. Dong, M. Esfandiari, X. Y. Li, “On the Microstructure and Phase Identification of Plasma Nitrided 17-4PH Precipitation Hardening Stainless Steel”, Surf Coat Technol 2008, 202, 2969–2975, doi:https://doi.org/10.1016/j.surfcoat.2007.10.036.
[13] P. Forcellese, T. Mancia, M. Simoncini, T. Bellezze, “Characterization of Microstructure and Localized Corrosion Resistance of HeatTreated 17-4 PH Stainless Steel Fabricated by Material Extrusion”, Metals (Basel) 2025, 15, doi:10.3390/met15020137.
[14] P. Forcellese, W.A. Khan, T. Mancia, M. Simoncini, M. Reiser, M. Kouřil, T. Bellezze, “Corrosion Resistance Properties of As-Sintered 17-4 PH Samples Additive-Manufactured Through Binder Jetting”, Metals (Basel) 2025, 15, doi:10.3390/met15101082.
[15] Z. Li, J. Chen, W. Xue, C. Yin, J. Song, K. Xiao, “Role of Segregation Behavior of Cu and Sb in the Region of Inclusions on Initial Corrosion”, Npj Mater Degrad 2023, 7, doi:10.1038/s41529-023-00354-5.
[16] S. Hu, E. H. Han, X. Liu, “Atomic-Scale Evidence for the Intergranular Corrosion Mechanism Induced by Co-Segregation of LowChromium Ferritic Stainless Steel”, Corros. Sci. 2021, 189, doi:10.1016/j.corsci.2021.109588.
[17] P. Forcellese, S. Belegni, A. Silvi, T. Mancia, M. Simoncini, T. Bellezze, “Study of Corrosion Resistance of 17-4 PH Stainless Steel asSintered and Heat-Treated Samples Manufactured by Bound Metal DepositionTM”, La Metallurgia Italiana 2023, 11/12, 9-14.
[18] P. Forcellese, T. Mancia, M. Simoncini, T. Bellezze, “Investigation on Corrosion Resistance Properties of 17-4 PH Bound Metal Deposition As-Sintered Specimens with Different Build-Up Orientations”, Metals (Basel) 2022, 12, doi:10.3390/met12040588.
[19] M. Lara-Banda, C. Gaona-Tiburcio, P. Zambrano-Robledo, M. Delgado-Espino, J.A. Cabral-Miramontes, D. Nieves-Mendoza, E. Maldonado-Bandala, F. Estupiñan-López, J. Chacón-Nava, F. Almeraya-Calderón, “Alternative to Nitric Acid Passivation of 15-5 and 17-4PH Stainless Steel Using Electrochemical Techniques”, Materials 2020, 13, doi:10.3390/ma13122836.
[20] J. Noh, N. J. Laycock, W. Gao, D. B. Wells, “Effects of Nitric Acid Passivation on the Pitting Resistance of 316 Stainless Steel”, Corros Sci 2000, 42, 2069–2084, doi:10.1016/S0010-938X(00)00052-4.
[21] C. Zhang, Y. Wei, J. Yang, W. Emori, J. Li, “Effects of Nitric Acid Passivation on the Corrosion Behavior of ZG06Cr13Ni4Mo Stainless Steel in Simulated Marine Atmosphere”, Materials and Corrosion 2020, 71, doi:10.1002/maco.202011597.
[22] T. Bellezze, G. Roventi, A. Quaranta, R. Fratesi, “Improvement of Pitting Corrosion Resistance of AISI 444 Stainless Steel to Make It a Possible Substitute for AISI 304L and 316L in Hot Natural Waters”, Materials and Corrosion 2008, 59, 727–731, doi:10.1002/ maco.200804112.
[23] Y. Wu, D. Blaine, B. Marx, C. Schlaefer, R. M. German, “Sintering Densification and Microstructural Evolution of Injection Molding Grade 17-4 PH Stainless Steel Powder”, Metall. Mater. Trans. A 33, 2185–2194 (2002). https://doi.org/10.1007/s11661-002-0050-4
Il Binder Jetting (BJ) è una tecnologia di additive manufacturing che consente di produrre componenti in lega con una velocità e risoluzione superiori rispetto ad altri sistemi. Tuttavia, le proprietà di resistenza alla corrosione localizzata dell’acciaio inox prodotto tramite BJ sono inferiori a quelle ottenute con i processi di produzione convenzionali. Questo studio si propone di valutare gli effetti dei trattamenti di passivazione sul comportamento alla corrosione dei campioni 17-4 PH as-sintered realizzati tramite BJ. I campioni sono stati trattati con quattro soluzioni acide: 15% HNO 3, 20% HNO3, 40% HNO3 e 40% HNO3 + 1% HF. Le proprietà di resistenza alla corrosione localizzata sono state valutate tramite test di Polarizzazione Potenziodinamica Ciclica (CPP) in un elettrolita a base di cloruro di sodio a pH neutro. I trattamenti hanno migliorato significativamente le proprietà di resistenza alla corrosione localizzata dei campioni BJ assintered, determinando il tipico andamento anodico passivo della curva CPP, che non era stato osservato nei campioni non trattati. Inoltre, concentrazioni più elevate di HNO 3 (40%) hanno migliorato la resistenza alla corrosione per pitting, mentre l’aggiunta di HF si è rivelata inefficace. Lo studio svolto permette di rendere più ampia l’applicazione industriale dell’acciaio inox prodotto tramite manifattura additiva, grazie al miglioramento delle sue proprietà di resistenza a corrosione localizzata attraverso trattamenti di passivazione.



Presentazione di Memorie
Gli interessati a presentare memorie scientifiche (sia per le sessioni orali che per la sessione poster) dovranno inviare entro il 31 marzo 2026, il titolo della memoria, i nomi degli autori e la loro affiliazione ed un sommario di circa 300 parole. Ci sono due modi per sottoporre le proposte di memorie:
• compilando il form online presente sul sito dell’evento: www.aimnet.it/nazionaleaim
• inviando tutte le informazioni (titolo, autori con affiliazioni, recapiti del relatore e sommario) a mezzo e-mail: info@aimnet.it
Spazio aziende e Sponsorizzazione
È previsto uno spazio per l’esposizione di apparecchiature, per la presentazione dei servizi e per la distribuzione di materiale promozionale. Le aziende interessate potranno richiedere informazioni più dettagliate sullo spazio aziende e sulle diverse possibilità di sponsorizzazione dell’evento alla Segreteria AIM.
Aziende che sponsorizzano il Convegno: (lista in aggiornamento)




Brescia
Brescia, città riconosciuta dall’UNESCO per il suo ricco patrimonio architettonico, si presenta oggi anche come un simbolo del futuro della metallurgia. Con acciaierie all’avanguardia, distretti produttivi dinamici e una vibrante offerta culturale ed enogastronomica, rappresenta un perfetto connubio tra innovazione tecnologica e qualità della vita. Da sempre terra di progresso, oggi è protagonista della transizione ecologica e digitale che sta ridefinendo il settore industriale. Brescia, infatti, è uno dei principali poli europei nel settore della metallurgia e della produzione di acciaio. Il suo distretto industriale, tra i più avanzati d’Italia, integra imprese di eccellenza, centri di ricerca e istituzioni accademiche, configurandosi come un modello di sviluppo tecnologico e produttivo.
ASSOCIAZIONE ITALIANA DI METALLURGIA
Via Filippo Turati 8 - 20121 Milano MI
Tel. 02-76397770
E-mail: info@aimnet.it www.aimnet.it/nazionaleaim
Brescia 9-11 settembre 2026



A. Mobili, T. Bellezze, F. Tittarelli
This work was carried out within the H2020 EnDurCrete project (GA n° 760639). In this study, innovative cement mortars with low electrical impedance were manufactured with sustainable carbon-based additions, such as biochar (BCH) at 0.5 vol% and recycled carbon fibers (RCF) at 0.05 vol% on the total mix. Electrical impedance (Z Re) of mortar of cement mortars and corrosion behavior (Ecorr and Rp) of embedded steel reinforcements were evaluated during both the curing period (i.e., 28 days) and weekly wet/dry cycles in a 3.5% NaCl solution. Results show that the combination of BCH and RCF reduces by approximately 60% the ZRe value compared to the plain mortar. However, the lower electrical impedance of mortars with carbon-based additions with respect to the plain mortar does not impair negatively the corrosion behavior of embedded rebars, both during the curing and the exposure to wet/dry cycles in the chloriderich solution, since they seem to be even more protective for reinforcements during the whole experimentation.
Carbon-based materials in the form of particles and fibers are often added in cement-based composites to reduce electrical resistivity and enhance mechanical properties, making them suitable for technologically advanced products [1]. Several types of carbonaceous materials have been studied in technical and scientific literature like carbon fibers [2], carbon black [3], graphite [4] and graphene [5] particles, carbon nanotubes [6], gasification char [7], etc. Recently, the present authors have studied the properties of cement-based mortars and concretes prepared with sustainable carbon-based materials, proving that an optimized combination of biochar (BCH) and recycled carbon fibers (RCF) addition enhances both electrical and mechanical performance [8]. Including BCH in concrete at approximately 2% by mass enhances mechanical strength [9], and replacing part of cement with BCH contributes to reduce CO2 emissions [10] thanks also to the biochar capability of absorbing CO2 [11]. On the other hand, carbon fibers (CF) have been extensively used in self-sensing and low-resistive cement-based materials, enabling the monitoring of the compressive strain of concrete elements thanks to their physical and chemical properties [12].
delle Marche, Ancona, Italy
The durability of reinforced cement-based materials governs the service life of structures and it is well known that, the longer is the time between the first sign of degradation to the intervention, the higher the costs needed to repair a concrete structure [13]. Although in recent years many studies have been published on the use of carbon-based additions to enhance physical, mechanical, and electrical properties of cement-based materials, still there are very few works regarding the effect of these materials on the corrosion of embedded steel reinforcements. Previous studies highlighted that CF addition increases corrosion current density (Icorr) of steel reinforcements, probably due to the decreasing of electrical resistivity [14], and the higher the CF content, the higher is the Icorr value [15]. Del Carmen Camacho et al. [16] added carbon nanotubes (CNT) in reinforced cement paste specimens exposed to carbonation and chloride attacks. Results obtained showed that the increase in CNT content does not modify significantly the mechanical properties but increases steel corrosion rates. On the other hand, other authors added graphene nanoplatelets (GNP) to mortars at 0, 0.05, and 1% by cement mass and concluded that at low level GNP addition may enhance the passivity and resistance to chloride-induced corrosion of carbon steel rebars [17]. Therefore, given the few papers reported by literature on the effect of carbon based addition in cement based materials on corrosion of embedded steel reinforcements and even with contradictory results, in this study innovative cement mortars containing BCH and RCF were studied in terms of electrical impedance (ZRe) and corrosion behavior of embedded steel rebars during both
the curing period and weekly wet/dry cycles in a 3.5% NaCl solution.
MATERIALS AND METHODS
Mortars were prepared with a limestone cement CEM II A-LL 42.5R and calcareous sand (0/8 mm) in saturated surface dry (s.s.d.) condition (water absorption of 2% by mass). Recycled carbon fibers (RCF) with 6 mm length and 7 µ m diameter supplied by Procotex Belgium SA were used as fibrous addition. RCF have a specific surface area of 0.132 m2/g, a density of 1.85 g/cm3 and a carbon content of 94% by mass. BCH supplied by RES Italia was used in filler form (diameter lower than 75 µ m). BCH has a specific surface area of 46.2 m2/g, a density of 2.0 g/cm3 and it is composed by 100% amorphous carbon. BCH and RCF were added to mortars at 0.5 vol% and 0.05 vol% on the total, respectively. As superplasticizer (SP), an acrylic one (Dynamon SP1, Mapei S.p.A.) was used to manufacture mortars with plastic consistency (flow value between 140 and 210 mm according to the EN 1015:3 standard). In table 1, the mix design of mortars is reported. After mixing, mortars were poured into molds and placed inside a climatic chamber. Specimens were cured at temperature (T) = 20 ± 1 °C and relative humidity (RH) > 95% for 7 days inside molds covered by plastic sheets, then they were demolded, the plastic sheets were removed, and specimens were cured at T = 20 ± 1 °C and RH = 50 ± 5% for 56 days. Then, specimens were exposed to 6 weekly wet/dry cycles in a 3.5% NaCl solution in water (5 days dry and 2 days wet). During the drying period, specimens were left at room conditions.
The mechanical performance of mortars was studied after 1, 7, and 28 days of curing in terms of compressive strength (Rc) with a Galdabini hydraulic press (400 kN full scale and 1% precision). Three specimens (40 mm x 40 mm x 160 mm) per composition were tested and the average strength was calculated. The electrical properties of mortars were studied in terms of electrical impedance (ZRe) with a 4-electrode configuration according to the Wenner’s method [18] in alternating current at 10 kHz with a Gamry Reference 600 in galvanostatic mode. This configuration was chosen in order to avoid the polarization of both electrodes and the material itself [19]. Electrical impedance was studied on three prismatic specimens (40 mm x 40 mm x 160 mm) equipped with 4 stainless-steel rods (Ø 3 mm and 40 mm length) embedded for 20 mm inside the mortar with a spacing of 20 mm, as reported in [8]. The corrosion behavior of embedded rebars was studied in terms of E corr and R p with an Autolab PGSTAT 204 potentiostat/galvanostat. The Rp was measured with the potentiodynamic polarization method (scan rate = 0.167 mV/s; ΔV = ± 10 mV) by calculating the slope of the anodic branch, which can be indicated as Ranod. For corrosion measurements, cylindrical mortar specimens equipped with 4 corrugated carbon steel rebars (Ø 8 mm) acting as working electrode and a stainless-steel rebar (Ø 6 mm)
acting as counter-electrode partially embedded inside the specimen were used. The corrugated carbon steel rebars were embedded in the mortar specimens leaving an exposed surface area (As) of 16 cm2. The configuration of the cylindrical specimens is reported in [20]. As reference electrode, a Saturated Calomel Electrode (SCE, +0.241 V vs SHE) was used. The ZRe and corrosion behavior of embedded reinforcements was studied on mortar specimens during the curing period (i.e. up to 56 days) and during 6 weekly wet/dry cycles in a 3.5% NaCl solution in water (5 days dry and 2 days wet). In order to depurate the corrosion measurements from the mortar resistance, an additional measurement on the cylindrical specimens was performed. An electrochemical impedance spectroscopy measurement has been done in the frequency (f) range between 100 kHz and 10 Hz, then the value of impedance modulus in terms of log |Z| and the phase θ were reported as a function of log f (Bode plot). The minimum value of θ (very close to 0°) was found and a range around it was suitably selected. The values of log |Z| found in this range were used to calculate an average value that corresponds to the resistance of the mortar (Rm). This value was subtracted from the Ranod to find a resistance that becomes R p after multiplying it by the exposed area As of the sample:
The results of compressive strength tests of mortars during the first 28 days of curing are given in figure 1. During the first week, mortars show approximately the same R c values. With the progressive hydration of the matrix, the differences between the studied mixes increase. The REF mortar reaches a R c of 78 MPa; the highest obtained Rc value. When RCF or BCH are added to the mix alone, the Rc slightly diminishes, reaching 71 and 72 MPa for the two mixes, respectively. The combination of RCF and BCH further decreases the R c of the mortar, which obtains 68 MPa (13% less than the plain mortar). As previously found by the authors [8,21], the addition of RCF
or BCH can slightly decrease the mechanical properties of cement-based materials due to an increased porosity. In any case, all mortars reach very high R c values, suitable for reinforced structural applications.
The calculated values of electrical impedance of mortars during 56 days of curing are given in figure 2. As expected, electrical impedance increases in time [22]; this is due to water consumptions due both to cement hydration and evaporation [23]. The results obtained show that the plain mortar has the highest electrical impedance values during the whole testing period. Conversely, the inclusion of carbonaceous additions contributes to the reduction of the electrical impedance of mortars. With more details, when
RCF is used the ZRe values decrease from 8250 to 4570 Ω after 56 days, thus become 45% lower than the reference mixture. When BCH is added, ZRe lowers to 6692 Ω , which is 19% lower than REF mortar. After 56 days of curing, the highest reduction of electrical impedance was registered by the mortar containing the combined addition of RCF and BCH, with a ZRe value of 3700 Ω , which is 55% lower than that of plain mortar, confirming the results obtained by the same authors in a previous study [8].

In figure 3 Ecorr and R p values of steel reinforcements embedded in mortars during the curing period (56 days) are reported. Ecorr results highlighted that only steel reinforcements embedded in REF and RCF mortars reached a high probability of corrosion during the first days immediately after casting. However, the higher probability
of corrosion is found for rebars embedded in the reference mortar. After 10 days of curing all reinforcements seem to have reached passivation, given the increasing trend of E corr values. R p values are in agreement with corrosion potential trend, given that all rebars embedded in mortars manufactured with carbon-based additions show R p values higher than those embedded in the reference one. With more detail, steel reinforcements embedded in the RCF mix show slightly higher Rp values compared to the rebars embedded in REF mortar, with a R p reaching 2012 k Ω ·cm2 at the end of the test (+8% than REF). On the other hand, steel reinforcements contained in mortars
manufactured with BCH alone and together with RCF recorded 56% and 70% higher Rp than those in the REF mortar, respectively. This means that when mortars are not exposed to aggressive environments, the addition of carbon-based additions in the form of filler and fibers seems to be beneficial to protect reinforcements from corrosion, confirming the preliminary results found by Shakouri and Abraham [17] when GNP is added to mortars up to 1% by cement mass. Indeed, they concluded that GNP contributed to form a denser passive layer structure.
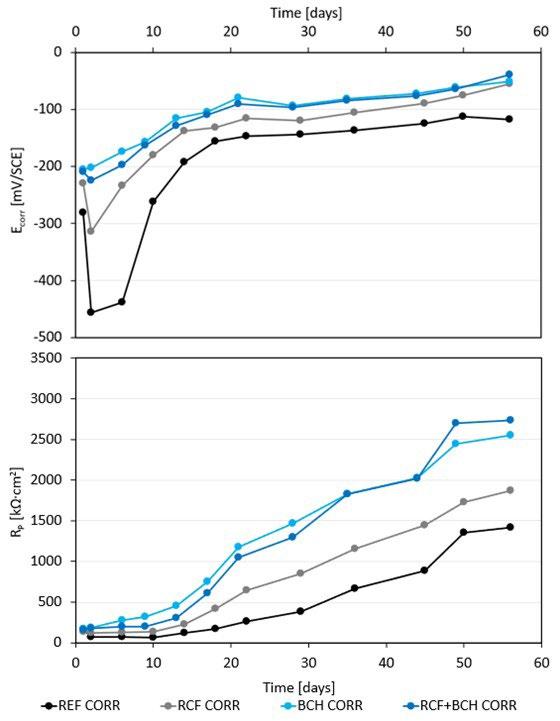
Fig.3 - E corr (sopra) e Rp (sotto) durante stagionatura / E corr (up) and Rp (down) during curing.
In figure 4 Ecorr and R p values of steel reinforcements embedded in mortars during wet/dry cycles in 3.5% NaCl solution are given. During the wet/dry cycles in the chloride-rich solution, steel rebars show and immediate drop of both Ecorr and R p due to the passage from air to a water-based solution. However, as for the curing period, steel reinforcements embedded in the matrix loaded with
BCH alone or coupled to RCF maintain their Rp higher than those in the REF mortars. Conversely, the addition of the sole RCF seems to be ineffective for corrosion resistance. In any case, all the studied steel reinforcements do not show a high probability of corrosion. Indeed, for carbon steel the probability of corrosion is high when Ecorr < −275 mV/SCE (as reported in the ASTM C876). Some authors
[24] have found that the addition of graphite powder (GP) to concrete can decrease the corrosion current density of steel rebars immersed in a 0.5 M NaCl solution only when GP is equal to 3% and 5% by cement mass, whereas lower contents seem to be less effective. A decrease of R p is expected with the additions of carbonaceous materials in contact with steel reinforcements, due to a coupling effect among them. On the contrary, in this work the Rp values increased in agreement with other authors [24]. Probably, oxygen binds to carbon particles, reducing its availability in the pore solution and therefore increasing the Rp values after immersion [24]. On the other hand, the contact between steel rebars and CF causes a more pronounced shift towards more positive Ecorr values, thus lower corrosion current density levels, only when the amount
of CF exceeds 1% by cement mass. Indeed, carbon has a more positive potential than steel, so a higher CF content implies a higher Ecorr shift [24]. Considering the present experimentation, BCH acts as GP, giving that the amount of BCH in the studied mixes is equal to 2% by cement mass. Probably, this percentage is enough to reduce the steel rebars corrosion exposed to chlorides. In the same way, the RCF content is not sufficient to determine the same corrosion reduction, given that RCF mortar contains only 0.2% of fibers by cement mass. Therefore, the combination of BCH and RCF slightly enhances the corrosion resistance of embedded steels in the 3.5% NaCl solution, given the increased quantity of carbon-based materials.
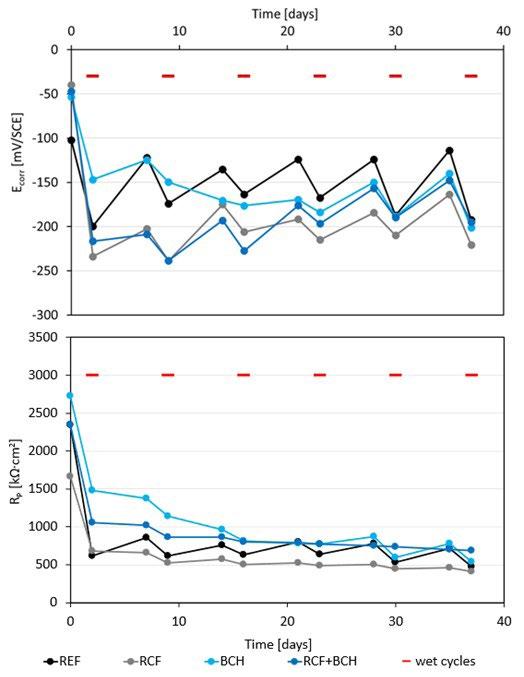
E corr
e Rp (sotto) durante cicli di bagnasciuga. Le linee rosse rappresentano il periodo in bagnato (2 giorni) / E corr (up) and Rp (down) during wet/dry cycles. Red lines represent the wet period (2 days).
Although, a decrease of Ecorr and R p values is registered during the wet/dry cycles, all the reinforcements can be considered in a passive state. Indeed, an estimation of
the corrosion current density (icorr) has been performed, according to the following equation:

and considering the coefficient B value equal to 52 mV, characteristic of rebars in a passive state (while B = 26 mV refers to steel in active state) [25]. Results demonstrate that before exposure (after 56 days of curing) the icorr ranges around 0.02 µ A/cm2 for reinforcements embedded in all mixes. At the end of curing, the icorr slightly increases reaching values close to 0.1 µ A/cm2. As reported in [25], these values represent a negligible corrosion, therefore all reinforcements remain passive also during accelerated exposure to wet/dry cycles in a 3.5% NaCl solution. Indeed, it should be noted that all R p values have been calculated after removing the mortar resistance (Rm) which negligibly affects the polarization curve of steel rebars.
This research was carried out to evaluate the mechanical and electrical behavior of cement mortars containing sustainable carbon-based additions, i.e., biochar (BCH) and recycled carbon fibers (RCF). Moreover, the corrosion behavior of embedded carbon steel rebars during the curing period and accelerated chloride contamination was investigated in terms of polarization resistance (Rp). It was found that the addition of BCH and RCF both alone and coupled together does not negatively impair the mechanical strength of mortars, since the highest R c reduction is only 13% for the BCH+RCF mixture, which
reaches a final compressive strength of 68 MPa after 28 days of curing. The obtained results demonstrate that the mortar with the lowest electrical impedance is the one manufactured with the combination of BCH and RCF, which shows a decreased electrical impedance of 55% compared to the reference one. Mortars containing BCH alone and coupled to RCF, despite being more conductive than the reference mixture, do not worsen the corrosion behavior of the embedded reinforcements since they seem to be even more protective for reinforcements both during curing and during exposure to the chloride-rich solution.
This research activity was carried out within the EnDurCrete (New Environmental friendly and Durable conCrete, integrating industrial by-products and hybrid systems, for civil, industrial and offshore applications) project, funded by the European Union’s Horizon 2020 research and innovation programme under grant agreement n° 760639.
[1] D. D. L. Chung, Composites Part B: Engineering, 31, 511–526 (2000).
[2] N. Banthia et al., Cement and Concrete Research, 22, 804–814 (1992).
[3] S. Gwon et al., Cement and Concrete Composites, 137, 104942 (2023).
[4] S. Sun et al., Construction and Building Materials, 136, 314–328 (2017).
[5] W. Li et al., Construction and Building Materials, 331, 127284 (2022).
[6] G. Xing et al., Journal of Building Engineering, 95, 110364 (2024).
[7] A. Mobili et al., Journal of Building Engineering, 50, 104237 (2022).
[8] A. Mobili et al., Construction and Building Materials, 376, 131051 (2023).
[9] H. Maljaee et al., Construction and Building Materials, 283, 122757 (2021).
[10] R. A. Mensah et al., Sustainability, 13(16), 9336 (2021).
[11] D. Winters et al., Sustainability, 14(8), 4633 (2022).
[12] B. Han et al., Self-Sensing Concr. Smart Struct., Ed. Butterworth-Heinemann, (2014).
[13] W. R. De Sitter, CEB-RILEM Workshop on Durability of Concrete Structures, Ed. Comité Euro-International du Béton, Copenhagen (1984).
[14] P. W. Chen, D. D. L. Chung, Journal of Electronic Materials, 24, 47–51 (1995).
[15] P. Garcés et al., Cement and Concrete Research, 35, 324–331 (2005).
[16] M. Del Carmen Camacho et al., Materials, 7(3), 1640–1651 (2014).
[17] M. Shakouri, O.F. Abraham, Construction and Building Materials, 452, 138948 (2024).
[18] F. Wenner, Journal of the Washington Academy of Sciences, 5, 561–563 (1915).
[19] G. Cosoli et al., Construction and Building Materials, 271, 121546 (2021).
[20] F. Tittarelli et al., Corrosion Science, 134, 64–77 (2018).
[21] A. Belli et al., Journal of Building Engineering, 73 106836, (2023).
[22] P. Azarsa, R. Gupta, Advances in Materials Science and Engineering, 2017, 8453095 (2017).
[23] A. Mobili et al., Sustainability, 14(3), 1775 (2022).
[24] P. Garcés et al., Corrosion Science, 49, 2557–2566 (2007).
[25] A. M. Aguirre-Guerrero et al., Applied Clay Science, 135, 437-446 (2017).
Comportamento
Questo lavoro è stato realizzato nell’ambito del progetto H2020 EnDurCrete (GA n° 760639). In questo studio, sono state create malte cementizie innovative a bassa impedenza elettrica contenenti aggiunte sostenibili a base di carbonio come biochar (BCH) allo 0.5% e fibre di carbonio riciclate (RCF) allo 0.05% sul volume totale. L’impedenza elettrica (ZRe) delle malte cementizie e il comportamento a corrosione (Ecorr e Rp) di rinforzi in acciaio sono stati valutati sia durante il periodo di stagionatura (28 giorni) sia durante cicli di bagnato/asciutto in una soluzione al 3.5% di NaCl. I risultati mostrano che l’aggiunta combinata di BCH e RCF riduce di circa il 60% il valore di ZRe della malta. Inoltre, le malte con aggiunte carboniose, nonostante più elettricamente conduttive della miscela di riferimento, non peggiorano il comportamento a corrosione delle armature inglobate sia durante la stagionatura sia durante l’esposizione ai cicli di bagnasciuga nella soluzione ricca di cloruri; anzi, sembrano addirittura essere più protettive per le armature durante l’intera sperimentazione.
P. Forcellese, W. A. Khan, G. Orilisi, G. Orsini, F. Luzi, A. Mazzoli, V. Corinaldesi, T. Bellezze
The most commonly used alloys in orthodontic wires are stainless steel and titanium due to their corrosion-resistant properties. While these alloys generally exhibit good corrosion behavior in mildly aggressive environments, unexpected breakages and allergic reactions can sometimes occur. This is mainly due to the degradation of the passive film of orthodontic wires, which is caused by the complex and variable conditions within the oral cavity. The composition of human saliva and the duration of exposure can differ significantly depending on the patient. This study evaluated the corrosion resistance properties of two commercially available orthodontic wires: 304 steel and a nickel-titanium alloy. Their electrochemical behavior was investigated using cyclic potentiodynamic polarization curves recorded in artificial saliva at 37 °C and a pH of 5.5. The results showed that 304 stainless steel orthodontic wires exhibit variable corrosion resistance properties, ranging from excellent to poor. Furthermore, when the passive film on 304 stainless steel breaks down, the repassivation potential is lower than the corrosion potential. This indicates that damage can easily propagate, leading to significant failure and the release of metal ions. This poses risk of allergies. On the other hand, the nickeltitanium orthodontic wires never showed localized corrosion behavior, demonstrating superior corrosion resistance properties compared to 304.
KEYWORDS: LOCALIZED CORROSION, ORTHODONTIC WIRES, ARTIFICIAL SALIVA, CYCLIC POTENTIODYNAMIC POLARIZATION;
Stainless steel and nickel-titanium (NiTi) alloys are among the most commonly used materials for orthodontic wires due to their combination of mechanical strength, formability, and corrosion resistance. However, despite their widespread use and general reliability, failures and allergic reactions have been reported in clinical practice, often associated with corrosion-induced degradation. These failures can compromise the mechanical integrity of the wire, leading to fracture or premature loss of functionality, and can also result in the release of metallic ions, such as nickel, iron, and chromium, which may cause biological complications, including local irritation or allergic responses [1, 2].
The corrosion behavior of orthodontic alloys is strongly influenced by the oral environment, which is highly variable and chemically complex. The composition of saliva differs among individuals and can fluctuate with diet, medication, or pathological conditions. These conditions create an aggressive environment in the oral cavity, capable of
Pietro Forcellese, Wasiq Ali Khan, Francesca Luzi, Alida Mazzoli, Valeria
Dipartimento di Scienze e Ingegneria della Materia, dell’Ambiente e Urbanistica, Università Politecnica delle Marche, Via Brecce Bianche, 60131, Ancona, Italy
Giulia Orilisi, Giovanna Orsini
Dipartimento di Scienze Cliniche Specialistiche e Odontostomatologiche, Università Politecnica delle Marche, Via Tronto 10A, 60126, Ancona, Italy
promoting localized corrosion phenomena, such as pitting or crevice corrosion, particularly in stainless steels.
To reproduce such complex in vivo conditions, laboratory investigations typically employ artificial saliva (AS) solutions with controlled composition, temperature, and pH. These tests enable the evaluation of corrosion resistance under reproducible and clinically relevant conditions. Nevertheless, before assessing the electrochemical performance of orthodontic materials, it is essential to verify that both the electrolyte and the experimental setup are appropriate and reliable.
In this context, the present study aimed to verify the suitability of the selected artificial saliva solution as a representative electrolyte for corrosion testing. Subsequently, the experimental setup was validated to ensure that it could effectively evaluate the corrosion resistance of wire-shaped metallic samples. Once these
preliminary steps were established, the research focused on investigating the localized corrosion resistance of commercial and orthodontic wires in artificial saliva at 37 °C and pH 5.5.
By following this approach, the study intends to provide a more reliable framework for the electrochemical evaluation of orthodontic alloys, linking methodological accuracy with clinically relevant insights into corrosion behavior.
The corrosion behavior of different stainless steel and NiTi wires was investigated through electrochemical testing in AS at 37 °C and pH 5.5. The AS composition shown in table 1 was derived from previous studies aimed at reproducing the ionic environment of the human oral cavity [3].
Tab.1 - Composizione chimica della saliva artificiale in % in peso / Chemical composition of the AS in wt.%.
Five different metallic materials were investigated in this study: wrought AISI 304 stainless steel (wrought 304), commercial AISI 304 stainless steel wire (wire 304), orthodontic AISI 304 stainless steel wire (orthodontic 304), commercial NiTi wire (nitinol), and orthodontic NiTi wire (orthodontic nitinol).
Two electrochemical cell configurations were employed depending on the geometry of the samples.
A conventional three-electrode cell was used for the plain surface of wrought 304, equipped with a saturated calomel electrode (SCE) as reference and a platinum wire as counter electrode. The exposed area of the wrought 304 specimen was 2 cm², and its surface roughness was the result of grinding using emery papers (1200 grit). This setup, shown in figure 1a and inspired by previously published works [4,5], was employed to validate the electrochemical measurements conducted in the AS solution (table 1), specifically formulated for testing the wire samples, ensuring that the modified wire cell configuration produced reliable and interpretable data for
the 304 stainless steel.
For wire samples, a modified cell setup was adopted from previously published works [6,7] for studying pitting corrosion and avoiding crevice corrosion: a bent wire, used as the working electrode, is immersed in the testing solution without using any specific sample holders or polymer coating to define the exposed surface. More specifically, this last parameter is defined considering the length of the wire portion immersed in the solution and its cross-section geometry. In this configuration, the counter electrode consisted of a platinum mesh surrounding the metallic wire to ensure a uniform current distribution along the cylindrical geometry (figure 1b), while the reference electrode (SCE) was the same as in the conventional setup. From this electrochemical cell configuration, the exposed areas were 1 cm² for wire 304, 0.8 cm² for orthodontic 304, 0.8 cm² for nitinol, and 0.64 cm² for orthodontic nitinol.
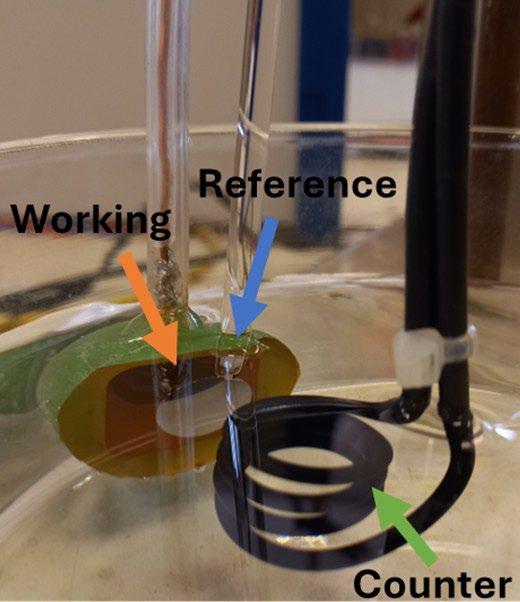
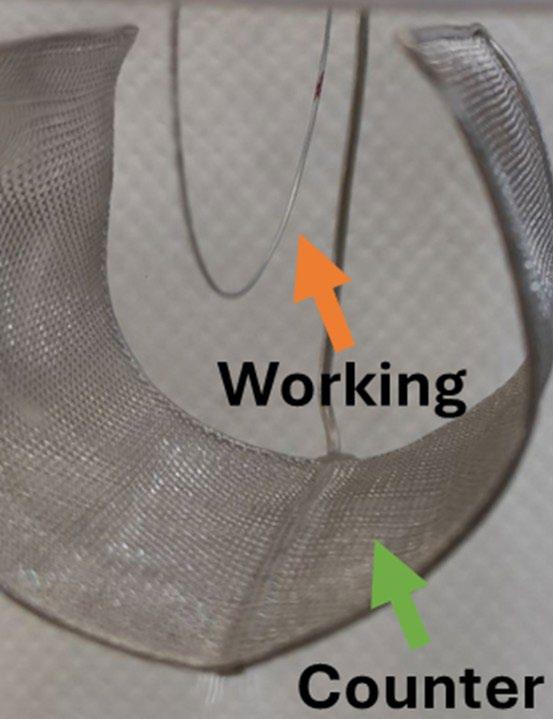
Fig.1 - Setup della cella: convenzionale per superfici piane (a) e per campioni di filo (b) / Cell setup: conventional plain samples (a) and wire samples (b).
Before testing, all samples were ultrasonically cleaned in n-hexane and deionized water to remove surface contaminants.
Electrochemical Impedance Spectroscopy (EIS) was carried out before Cyclic Potentiodynamic Polarization (CPP) measurements to determine the solution resistance and correct the polarization curves for the ohmic drop. The EIS spectra were collected in the frequency range from 100 kHz to 1 Hz, with 10 points per decade and a sinusoidal perturbation amplitude of 10 mV.
After EIS, CPP tests were performed to assess the localized corrosion resistance. The Open-Circuit Potential (OCP) was monitored for 30 minutes to ensure stabilization, after which the polarization scan was initiated 15 mV below the stationary OCP. The potential was swept in the anodic direction at a scan rate of 0.166 mV · s ¹, and then reversed using the same scan rate once the current density reached 0.01 mA · cm ². Each test was repeated at least five times to verify the reproducibility of the measurements. All electrochemical tests were conducted at 37 °C using a thermostatic bath to maintain a constant temperature during the experiments. The wrought 304 specimen was also tested at room temperature (RT).
The stability of the artificial saliva was preliminarily verified by monitoring its pH over several days. The pH
remained constant at approximately 5.5, ensuring the stability of the electrolyte and thus the reliability of the CPP measurements.
Figure 2 shows the CPP curves for the wrought 304 stainless steel tested at RT and at 37 °C, as well as for the wire 304 tested at 37 °C. The comparison between the wrought 304 curves at the two temperatures highlights the expected effect of temperature on the electrochemical behavior of stainless steels: the pitting and repassivation potentials decrease as the temperature increases, whereas the corrosion potential shifts slightly toward more positive values. This confirms that the electrolyte simulating the saliva supplies an expected anodic behavior of the material as typically occurs, thus validating the experimental setup. Additionally, figure 2 illustrates the comparison of the anodic curves of the wire 304 and the wrought 304 sample. The CPP curve of the wire 304 exhibits markedly different characteristic potentials: a significantly higher corrosion potential (around +0.1 V), a lower repassivation potential, and a higher pitting potential compared to the wrought 304. Moreover, the curve is shifted leftward by nearly two orders of magnitude in current density relative to the wrought sample, corresponding to a lower passivation current density and indicating a more protective passive film present on the wire. This result agrees with the significantly higher pitting potential of the wire 304, suggesting a less susceptibility to localized corrosion with
respect to wrought 304. Such differences can be partially attributed to the different geometry of the electrochemical cells (figure 1), but most likely to the smoother surface
of the wire as a result of the drawing process compared to the wrought 304, ground at 1200 grit. A low roughness improves the protective characteristic of the passive film.
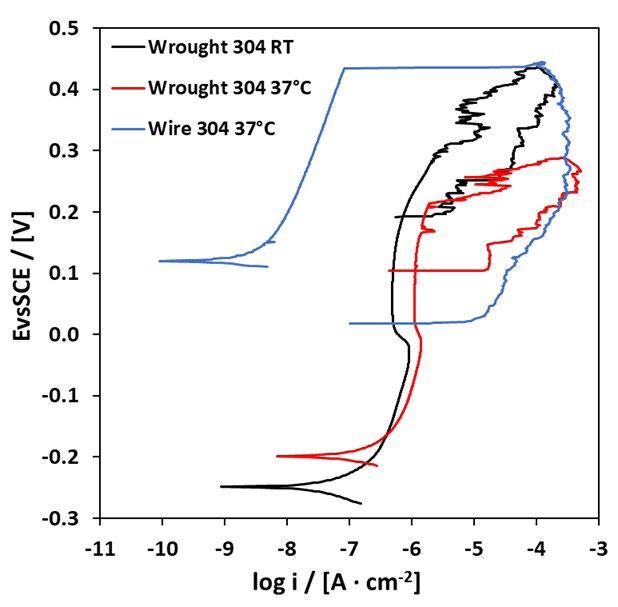
- Curve CPP più rappresentative dell’acciaio Wrought 304 e del filo 304 a temperatura ambiente e a 37 °C / Most representative CPP curves of Wrought 304 and wire 304 at RT and 37°C.
Representative CPP curves for the orthodontic 304 wires are shown in figure 3. The corrosion potential and the passivation current density are highly reproducible and close to that observed for the wire 304 in figure 2; in particular, the purple curve in figure 3 is comparable to that reported by other authors [7], who tested similar orthodontic wires in an AS solution with a composition close to that used in this work. However, the pitting potential exhibits significant variability: in some specimens, it exhibits low values (around +0.45 V), in others, it is higher (up to +0.95 V), and in certain cases, the anodic branch exhibits only an increase of current density,
most likely associated with oxygen evolution from water. Most of the orthodontic 304 wire curves recorded in this work fall into the “higher potential” category (blue curve in figure 3). Assuming this anodic behavior as the most representative of the orthodontic 304 wires examined, the corresponding pitting potential is considerably higher than that of the commercial wire (see blue curve in figure 2). This result can be attributed to the better surface finish of the orthodontic wires, which is deliberately diamond drawn to obtain a very smooth surface that reduces the adhesion of bacteria present in the oral cavity [8].
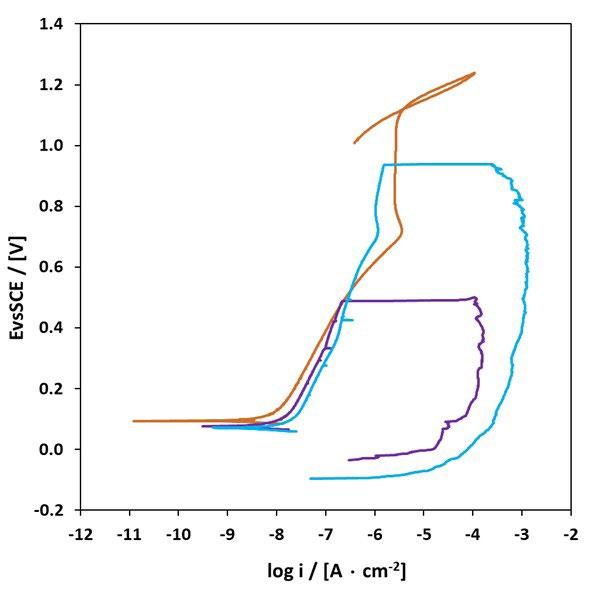
Fig.3 - Curve CPP più rappresentative del filo ortodontico in acciaio 304 a 37 °C / Most representative CPP curves of orthodontic 304 wire at 37°C.
The repassivation potential is found below the corrosion potential, indicating imperfect passive behavior of the tested orthodontic wires. This suggests that when these wires undergo localized corrosion, the process tends to propagate, potentially leading to mechanical failure and the release of significant amounts of metal ions. In several cases, the localized corrosion damage observed on orthodontic 304 wires was extensive. In the most severe instances, corrosion attack was so pronounced that it led to the complete fracture of the orthodontic wire, confirming that the wires operate in the critical condition
of the absence of the perfect passive behavior in the solution simulating the saliva.
Figure 4 displays the representative CPP curves of the NiTi commercial and orthodontic wires. Both materials exhibit very similar electrochemical behavior, showing no evidence of localized corrosion phenomena under the present test conditions. These results confirm the superior resistance to localized corrosion of NiTi alloys compared with the 304 stainless steel samples when exposed to this artificial saliva environment [7, 9, 10].
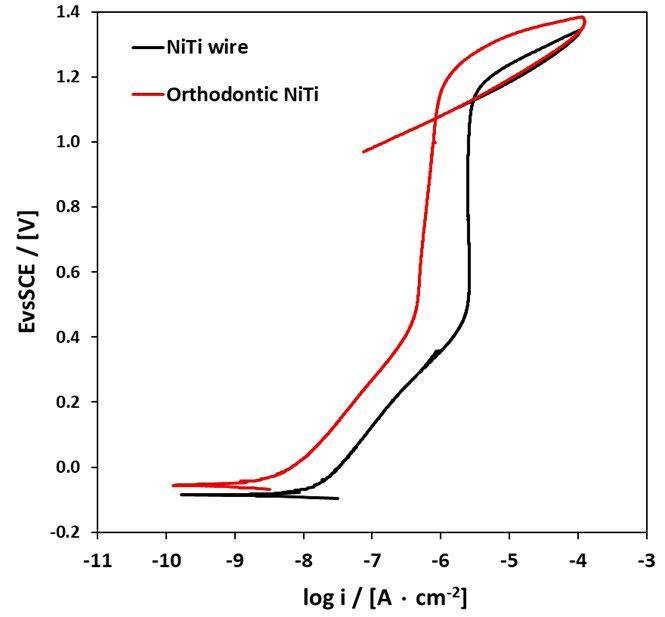
- Curve CPP più rappresentative del filo commerciale e ortodontico in NiTi a 37 °C / Most representative CPP curves of NiTi commercial and orthodontic NiTi wire at 37°C.
Finally, figure 5 summarizes the characteristic potentials (corrosion, pitting, and repassivation) for the 304-based samples. The reported values represent the average and standard deviation for each characteristic potential
obtained from the CPP curves. For the orthodontic 304 wires, only the specimens that exhibited measurable pitting potentials were considered in this analysis.
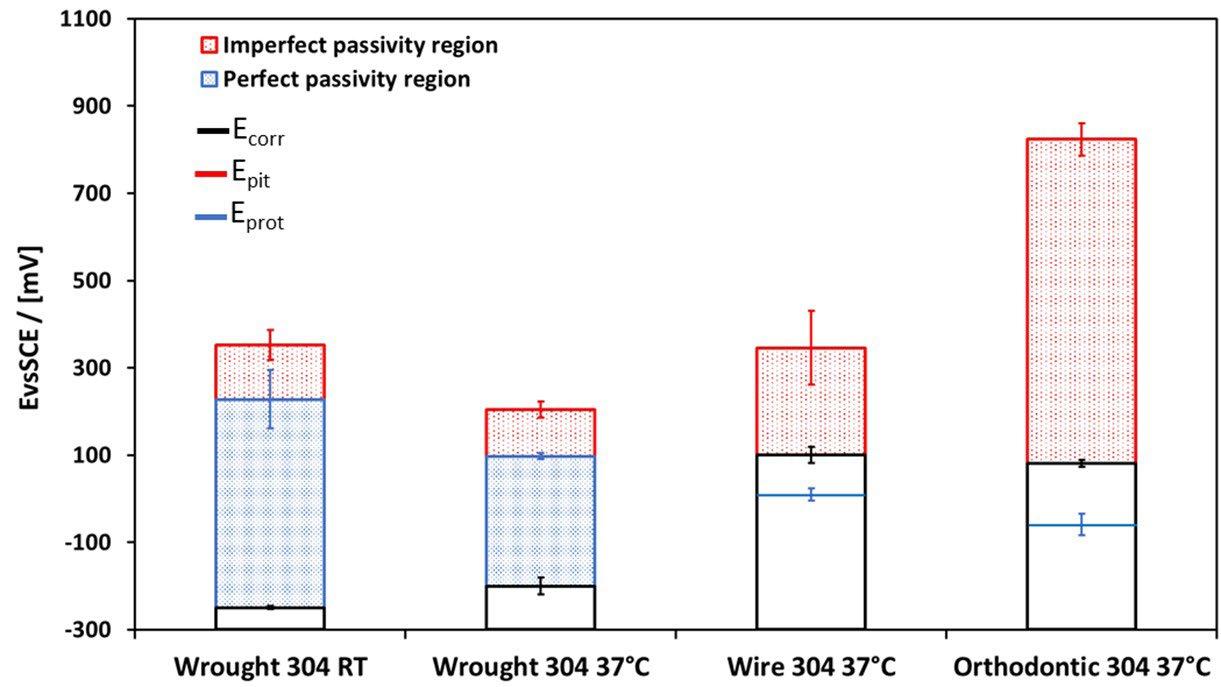
- Medie e deviazioni standard dei potenziali caratteristici delle curve CPP / Averages and standard deviations of characteristic potentials of the CPP curves.
This study examined the corrosion behavior of stainless steel and nickel-titanium orthodontic wires in artificial saliva at 37 °C and pH 5.5, assessing the electrolyte stability, cell setup reliability, and localized corrosion resistance. The artificial saliva remained stable over time, ensuring reproducible CPP measurements.
The comparison between wrought and wire 304 stainless steel confirmed that temperature, geometry of the electrochemical cell and the surface finish of metal significantly affect corrosion behavior. Orthodontic 304 wires showed variable resistance to localized corrosion,
with most samples exhibiting high pitting potentials (up to +0.95 V) and imperfect passivity. In severe cases, localized attack was significant and even caused wire fractures, indicating a potential risk of mechanical failure and metal ion release.
In contrast, NiTi and orthodontic NiTi wires showed no evidence of localized corrosion, confirming the superior stability of their passive film. Overall, while 304 stainless steel may be suitable for short-term orthodontic applications, NiTi wires represent a more reliable and durable option for long-term use.
[1] M.Conradi, P. M. Schön, A. Kocijan, M. Jenko, G. J. Vancso, “Surface analysis of localized corrosion of austenitic 316L and duplex 2205 stainless steels in simulated body solutions”, Mater. Chem. Phys. 2011, 130, 708–713, doi:10.1016/j.matchemphys.2011.07.049.
[2] A. Kocijan, D. K. Merl, M. Jenko, “The corrosion behaviour of austenitic and duplex stainless steels in artificial saliva with the addition of fluoride”, Corros. Sci. 2011, 53, 776–783, doi:10.1016/j.corsci.2010.11.010.
[3] G.S. Duffó, E. Q. Castillo, “Development of an Artificial Saliva Solution for Studying the Corrosion Behavior of Dental Alloys”, Corrosion 2004, 60, 594–602, doi:10.5006/1.3287764.
[4] P. Forcellese, T. Mancia, M. Simoncini, T. Bellezze, “Characterization of Microstructure and Localized Corrosion Resistance of HeatTreated 17-4 PH Stainless Steel Fabricated by Material Extrusion”, Metals (Basel) 2025, 15, doi:10.3390/met15020137.
[5] P. Forcellese, W.A. Khan, T. Mancia, M. Simoncini, M. Reiser, M. Kouřil, T. Bellezze, “Corrosion Resistance Properties of As-Sintered 17-4 PH Samples Additive-Manufactured Through Binder Jetting”, Metals (Basel) 2025, 15, 1082, doi:10.3390/met15101082.
[6] L. Stockert, F. Hunkeler, H. Bohni, “Technical Note: A Crevice-Free Measurement Technique to Determine Reproducible Pitting Potentials”, Corrosion 1985, 41, 676–677, doi:10.5006/1.3583002.
[7] M. Mirjalili, M. Momeni, N. Ebrahimi, M.H. Moayed, “Comparative study on corrosion behaviour of Nitinol and stainless steel orthodontic wires in simulated saliva solution in presence of fluoride ions”, Mater. Sci. Eng. C 2013, 33, 2084–2093, doi:10.1016/j. msec.2013.01.026.
[8] S. Ashique Abdulhameed, N. N. Mude, D. Chandrasekaran, D. S. Goswami, O. N. Almutairi, P. Manimegalan, “Comparison of Bacterial Adherence on Different Archwires after Clinical Use”, J Pharm Bioallied Sci. 2024, 16(4), S3778-S3780, doi: 10.4103/jpbs.jpbs_974_24.
[9] G. Rondelli, “Localized corrosion behaviour in simulated human body fluids of commercial Ni-Ti orthodontic wires”, Biomaterials 1999, 20, 785–792, doi:10.1016/S0142-9612(98)90233-2.
[10] S. A. Shabalovskaya, G. C. Rondelli, A. L. Undisz, J. W. Anderegg, T. D. Burleigh, M. E. Rettenmayr, “The electrochemical characteristics of native Nitinol surfaces”. Biomaterials 2009, 30, 3662–3671, doi:10.1016/j.biomaterials.2009.03.034.
Le leghe più comunemente utilizzate negli archi ortodontici sono l’acciaio inossidabile e le leghe di titanio per via delle loro proprietà di resistenza alla corrosione. Queste leghe sono generalmente caratterizzate da un buon comportamento alla corrosione in ambienti debolmente aggressivi, sebbene talvolta si verifichino rotture impreviste e reazioni allergiche. Le principali cause possono essere attribuite al degrado del film passivo degli archi ortodontici, dovuto alle condizioni complesse e variabili che si verificano all’interno del cavo orale, poiché la composizione della saliva umana e la durata dell’esposizione possono differire significativamente a seconda del paziente. In questo studio è stata considerata la valutazione delle proprietà di resistenza alla corrosione per due archi ortodontici disponibili in commercio: acciaio 304 e una lega nichel-titanio. Il comportamento elettrochimico è stato indagato mediante curve di polarizzazione potenziodinamica ciclica registrate in saliva artificiale a 37 °C e pH 5,5. I risultati hanno dimostrato che gli archi ortodontici in acciaio 304 presentano proprietà di resistenza alla corrosione variabili, da eccellenti a scarse. Inoltre, quando si verifica la rottura del film passivo nell’acciaio 304, il potenziale di ripassivazione risulta inferiore al potenziale di corrosione, indicando che i danni possono propagarsi facilmente, portando a guasti significativi e al rilascio di ioni metallici, con conseguente rischio di allergie. Al contrario, gli archi ortodontici in nichel-titanio non hanno mai mostrato corrosione localizzata, dimostrando proprietà di resistenza alla corrosione superiori rispetto al 304.
L’Associazione Italiana di Metallurgia, per onorare la memoria del prof. Felice De Carli, ex Presidente dell’AIM, istituì nel 1968 un premio da assegnare ad un giovane ricercatore di cittadinanza italiana, che non avesse superato il 32° anno di età al momento della data di presentazione della domanda di concorso al premio e che avesse dimostrato di possedere un’adeguata maturità nel settore della ricerca metallurgica fondamentale e applicata.
L’Associazione ha deciso di bandire nuovamente il concorso per l’assegnazione del premio, consistente in una somma dell’importo di 1500 Euro e in una medaglia di ricordo recante l’effige del prof. Felice De Carli.
Per concorrere al premio occorre presentare domanda, anche a mezzo e-mail, con il testo di uno o più articoli originali del concorrente oltre che il curriculum vitae e l’elenco dei lavori già pubblicati o in corso di stampa.
La domanda va inviata, entro il 30 giugno 2026, alla Segreteria AIM, e-mail: info@aimnet.it.
La Commissione Giudicatrice, nominata dal Consiglio Direttivo AIM, a suo insindacabile giudizio, sceglierà l’Autore meritevole del premio, sia in base all’esame del lavoro inedito che dei titoli presentati.
La consegna del premio avverrà il 9 settembre 2026 a Brescia, in occasione del 41° Convegno Nazionale AIM.
Milano, 12 febbraio 2026

Per informazioni e candidature:
Per Ecotre Valente, l’innovazione è una questione di tecnologie, di processi, e, soprattutto, una questione di conoscenza.
Comprendere i fenomeni alla base della produzione industriale significa poterli governare, migliorare e rendere più sostenibili nel tempo; per questo motivo, in un contesto industriale sempre più complesso, la formazione tecnica evoluta rappresenta
un fattore strategico. È da questa convinzione che nasce la decisione di Ecotre Valente di acquisire i diritti del Solidification Course, uno dei percorsi formativi più longevi e riconosciuti a livello internazionale nel campo della solidificazione e della metallurgia, per continuare a diffondere una cultura tecnica solida, basata sulla comprensione profonda dei fenomeni fisici che governano i
processi produttivi, creando un linguaggio comune tra modelli teorici, esperienza industriale e osservazione diretta dei fenomeni.
Il Solidification Course offre l’opportunità di un confronto tra professionisti, ricercatori e tecnici, provenienti da settori e ruoli diversi, ma con la stessa esigenza: comprendere il comportamento dei metalli.

Nel tempo, il Solidification Course è cresciuto fino a diventare un punto di riferimento globale. Con 32 edizioni, oltre 1.100 partecipanti e la presenza di professionisti provenienti da più di 40 Paesi, ha coinvolto oltre 380 aziende attive nei settori della fusione, della metallurgia e del manufacturing. Il corso si concentra su ciò che accade durante la solidificazione dei metalli. Tra i principali temi affrontati:
• i fondamenti della solidificazione e i meccanismi di formazione dei difetti;
• l’evoluzione della microstruttura e il suo controllo.
Accanto a questi contenuti consolidati, questa 33° edizione del Solidification Course, sotto la guida di Ecotre Valente, amplia il proprio raggio d’azione, includendo ambiti emergenti come Additive manufacturing e Intelligenza artificiale, strumenti sempre più rilevanti nei processi manufatturieri.
Ecotre Valente inaugura una nuova fase di continuità e sviluppo per questo progetto: un percorso che guarda al futuro, mantenendo salda la propria vocazione formativa e internazionale.
“Ringrazio i fondatori del corso Prof. Rappaz e Dr. Gremaud, docenti nell’edizione 2001 a cui io stesso ho partecipato, che sono oggi preziosi collaboratori con cui garantire e condividere il tesoro di conoscenze che il corso offre” così Ing. Lorenzo Valente, CEO di Ecotre Valente, esprime con soddisfazione l’acquisizione del Solidification Course.

Il programma dettagliato, il corpo docente e i contenuti tecnici sono disponibili nella brochure ufficiale del Solidification Course (www.solidificationcourse.com).

As an integral element of the event, the Conference will feature an exhibition, that will enable excellent exposure for products, technologies, innovative solutions or services. At this opportunity the Organizers will set an area strategically located as regards the main Conference rooms. Companies will be able to reinforce their participation and enhance their corporate identification by taking advantage of benefits offered to them as Contributing Sponsors of the Conference. More information will be soon available at the Conference website. For any further information please contact Siderweb - The Italian Steel Community: commerciale@siderweb.com tel. +39 030 2540006 Organised by








Corso di base modulare Corrosione webinar (zoom) - 5-19 marzo 2026
>> SCOPRI DI PIÙ
Master
Master in Progettazione stampi ITINERANTE, MARZO-LUGLIO 2026
>> SCOPRI DI PIÙ
Corso
Gli acciai inossidabili XII edizione Milano - 3-4-10-11-17-18 marzo 2026
>> SCOPRI DI PIÙ
International Journal of the Italian Association for Metallurgy
Giornata di Studio Innovazione, efficienza e sostenibilità negli impianti di laminazione a caldo Legnano/Cuggiono (MI) - 6 marzo 2026
>> SCOPRI DI PIÙ
Corso Il disegno dei getti in leghe di alluminio Codesign e definizione dei sistemi di colata Kilometro Rosso Innovation District (Bergamo) - 10 marzo 2026
>> SCOPRI DI PIÙ
n. 6 giugno 2020 Organo ufficiale dell’Associazione Italiana di Metallurgia. Rivista fondata nel 1909





Giornata di Studio
Tra resistenza a fatica e tenacità. La risposta delle leghe metalliche agli sforzi dinamici
Milano c/o Centro Congressi FAST - 12 marzo 2026
>> SCOPRI DI PIÙ
EEC 2026 - EMECR 2026 Conferences - siderweb FORUM
4th European Electric Steelmaking conference, 5th International Conference on Energy and Material Efficiency and CO2 Reduction in the Steel Industry and the 2nd edition of the biennial event organised by siderweb to discuss the present and future of Italian and European steel Milano - 11-13 May 2026
>> MORE INFO
Giornata di Studio
Caratterizzazione di prodotti da processi additive manufacturing Casi pratici e studi di difettologie Rovereto c/o Trentino Sviluppo - 21 maggio 2026
>> SCOPRI DI PIÙ
Giornata di Studio ECNDT 2026 | Failure Analysis & NDT Verona - 18 giugno 2026
>> SCOPRI DI PIÙ
Tribologia c/o Laboratorio Te.Si. dell'Università degli Studi di Padova Rovigo - 24-25 giugno 2026
>> SCOPRI DI PIÙ





Summer School Digitalization & AI in Metallurgy Udine – 28-29-30 giugno – 1 luglio 2026
>> MORE INFO
Summer school “Environmental Assisted Cracking” Milazzo (ME) - 5-9 July 2026
>> MORE INFO
WCCM 2026
The 4th World Congress on Condition Monitoring Milano, Italy - 25-27 August 2026
>> MORE INFO
41° Convegno Nazionale AIM Progettiamo il futuro tra ricerca e innovazione Brescia - 9-11 settembre 2026
>> SCOPRI DI PIÙ
5th International Conference on INGOT CASTING, ROLLING & FORGING
Bardolino, Verona - 13-15 October 2026
>> MORE INFO
di Claudio Tegoni
Secondo classificato della prima edizione del concorso “Metallurgia a fumetti” 2025





Norme pubblicate e progetti in inchiesta (aggiornamento al 31 gennaio 2026)
Norme UNSIDER pubblicate da UNI nel mese di gennaio 2026
UNI EN ISO 15363:2026
Materiali metallici – Prova di espansione idraulica su anello tubolare
Norme UNSIDER ritirate con sostituzione da UNI nel mese di gennaio 2026
UNI EN 10275:2000
Materiali metallici – Prova di espansione idraulica su anello tubolare
Norme UNSIDER pubblicate da CEN e ISO nel mese di gennaio 2026
ISO 18203:2026
Steel – Determination of the thickness of surface-hardened layers
ISO 8458-1:2026
Steel wire for mechanical springs – Part 1: General requirements
ISO 8458-2:2026
Steel wire for mechanical springs – Part 2: Patented cold-drawn non-alloy steel wire
ISO 8458-3:2026
Steel wire for mechanical springs – Part 3: Oil-hardened and tempered wire
ISO 629:2026
Steel and cast iron – Determination of manganese content – Spectrophotometric method
Progetti UNSIDER messi allo studio dal CEN (Stage 10.99) – febbraio 2026
prEN ISO 10700 rev
Steel and iron – Determination of manganese content – Flame atomic absorption spectrometric method
prEN 10351 rev
Chemical analysis of ferrous materials – Inductively coupled plasma optical emission spectrometric analysis of unalloyed and low alloyed steels – Determination of Mn, P, Cu, Ni, Cr, Mo, V, Co, Al (total) and Sn
prEN 10355 rev
Chemical analysis of ferrous materials – Inductively coupled plasma optical emission spectrometric analysis of unalloyed and low alloyed steels – Determination of Si, Mn, P, Cu, Ni, Cr, Mo and Sn, following dissolution with nitric and sulphuric acids
prEN 10213 rev
Steel castings for pressure purposes
Progetti UNSIDER in inchiesta prEN e ISO/DIS – febbraio 2026
prEN – progetti di norma europei
prEN ISO 19277-1
Oil and gas industries including lower carbon energy – Qualification testing and acceptance criteria for protective coating systems under insulation – Part 1: Liquid applied coatings (ISO/DIS 19277-1:2026)
prEN 17248
District heating and district cooling pipe systems – Terms and definitions
prEN 10160
Non-destructive testing – Ultrasonic testing of steel flat product of thickness equal or greater than 5 mm (reflection method)
ISO/DIS – progetti di norma internazionali
ISO/DIS 29502
Operating management systems – Conformity assessment guidelines and criteria
ISO/DIS 29501
Operating management systems – Requirements with guidance for use
ISO/DIS 24826
Oil and gas industries including lower carbon energy – Drilling, production and injection equipment – Mud pumps
ISO/DIS 19277-1
Oil and gas industries including lower carbon energy – Qualification testing and acceptance criteria for protective coating systems under insulation – Part 1: Liquid applied coatings
ISO/DIS 16440
Oil and gas industries including lower carbon energy – Pipeline transportation systems –Design, construction and maintenance of steel cased pipelines
ISO/DIS 14250
Steel – Metallographic characterization of duplex grain size and distributions
ISO/DIS 10701
Steel and iron – Determination of sulfur content – Methylene blue spectrophotometric method
ISO/DIS 9681
Manganese ores and concentrates – Determination of iron content – Flame atomic absorption spectrometric method
Progetti UNSIDER al voto FprEN e ISO/ FDIS – febbraio 2026
FprEN – progetti di norma europei
FprEN ISO 14577-2
Metallic materials – Instrumented indentation test for hardness and materials parameters –
Part 2: Verification and calibration of testing machines (ISO/FDIS 14577-2:2026)
FprEN ISO 14577-3
Metallic materials – Instrumented indentation test for hardness and materials parameters–
Part 3: Calibration of reference blocks (ISO/ FDIS 14577-3:2026)
FprEN 10365
Hot rolled steel channels, I and H sections –Dimensions and masses
ISO/FDIS – progetti di norma internazionali
ISO/FDIS 14577-2
Metallic materials – Instrumented indentation test for hardness and materials parameters –
Part 2: Verification and calibration of testing machines
ISO/FDIS 14577-3
Metallic materials – Instrumented indentation test for hardness and materials parameters –
Part 3: Calibration of reference blocks
ISO/FDIS 13503-9
Oil and gas industries including lower carbon energy – Completion fluids and materials –Part 9: Methods for evaluating performance of acidizing fluids
ISO/FDIS 11951
Cold-reduced tinmill products – Blackplate
ISO/FDIS 11950
Cold-reduced tinmill products – Electrolytic chromium/chromium oxide-coated steel
ISO/FDIS 11949
Cold-reduced tinmill products – Electrolytic tinplate
ISO/FDIS 148-4
Metallic materials – Charpy pendulum impact test – Part 4: Testing of miniature Charpy-type V-notch test pieces



SAVE THE DATE
www.aimnet.it/eec2026

EEC 2026 & EMECR 2026 will be jointly held by AIM, Italian Association for Metallurgy, in Milan on 11-13 May 2026, together with siderweb FORUM!
The EEC 2026 (14th European Electric Steelmaking conference) will cover a wide range of topics related to the production of steel using electric arc furnaces (EAFs) and other electric-based processes.
The EMECR (International Conference on Energy and Material Efficiency and CO2 Reduction in the Steel Industry) has become a recognized forum for high level discussions on environmental related topics such as CO2 reduction, materials efficiency and product life cycles in the steel industry worldwide.
EEC 2026 & EMECR 2026 will provide opportunities for networking with industry leaders, researchers, and policymakers, discussing collaborative projects and partnerships.
Registration fees and participation details are available at www.aimnet.it/eec2026/register/
siderweb FORUM is the 2nd edition of the biennial event organised by siderweb to discuss the present and future of Italian and European steel.
The Conferences will be enriched with an exhibition, where sponsors will be able to display new technologies and equipment. The detailed exhibiting and sponsorship packages are available at www.aimnet.it/eec2026/exhibition-sponsorship/