

CLICK CHEMISTRY
Empowering Peptide Innovation
With this guiding theme in mind, Iris Biotech’s mission is to support researchers by supplying
• innovative technologies,
• rare compounds,
• as well as a broad portfolio on standard consumables, available in flexible quantities from small scale to bulk quantities. To fulfill our dedication “Empowering Peptide Innovation”, we are attending various conferences, symposia, and exhibitions each year. This allows us to remain in direct contact with scientists all over the world, both from academia and industry, to exchange knowledge, and to gather new ideas to tackle your current challenges.
Guided by our dedication to provide
• competent service,
• as well as novel substances and latest technologies,
Iris Biotech is your trusted partner for the world of peptides, while having strong expertise in associated disciplines. Thus, our portfolio comprises reagents and tools for the synthesis and modification of peptides, e.g., amino acids, resins and solvents but also for related technologies such as drug delivery, linkerology® and life sciences.
Owed to the growing demand for tailor-made compounds, our portfolio is fine-tuned by our custom synthesis service at Iris Biotech Laboratories. Our skilled scientists offer profound expertise in de novo route development,
• upscaling towards larger scale production,
• as well as synthesis optimization for increased efficiency.
Examples are the synthesis of rare chiral building blocks, unnatural amino acid derivatives, sophisticated orthogonal protecting groups, heterocycles, building blocks for nucleotides, PEGs and PEG-analogs as well as specific linkers for controlled drug delivery and release.
Protecting Groups
Portfolio Overview
Peptide Synthesis and Modification
(Protected) Amino Acids
Standards such as Fmoc-D/L-AAA and Boc-D/L-AAA, Smoc amino acids for peptide synthesis in water, variety of protecting groups (e.g., Pbf, Trt, tBu, Bzl, Acm, Mob, SIT, Phacm, Allocam, Mmt), unusual amino acids, fluorinated derivatives, substituted prolines, arginine analogs
Building Blocks
Amino alcohols, amino aldehydes, diamines and hydrazines, (pseudoproline) dipeptides, polyamines and spermines, fatty acid derivatives, nucleic acid building blocks
Reagents
Coupling reagents, solvents and scavengers, protecting groups
Resins
Preloaded resins (e.g., based on Trityl, TCP, TentaGel, Methoxybenzhydryl, Merrifield, PAM, Rink, Sieber, Wang), scavenger resins, hydrazone resins, poly(acrylamide) resins, Cyclover
Linkerology® and Drug Delivery Life Sciences
Linkers for Solid Phase Peptide Synthesis
Cleavable Linkers
Val-Ala-based, Val-Cit-based, disulfide-based, Dde-helping hands, pH-sensitive linkers
Photo-Activatable Linkers
Functionalized Linkers
Clickable linkers, trifunctional linkers, linkers with maleimide function, cross-linkers, selective N-term acylation and biotinylation, 5HP2O, next generation maleimides
Lipids Fullerenes, Poly(2-oxazolines), Dextrans & Plant-Derived Cholesterol
Nanoparticles
Poly-Amino Acids
Poly-Arg, Poly-Glu, Poly-Lys, Poly-Orn, Poly-Sar
PEGylation
Branched PEGylating reagents, (amino-)PEG-acids, PEG-amines & hydrazides & guanidines, reagents for Click-conjugation, Biotin-PEG-reagents, PEG-thiols, PEG-maleimides, other PEGylating reagents
Biotinylation Reagents
Carbohydrates
Galactose, Glucose, Mannose, Xylose and others
Drug Metabolites
Peptides
Substrates & Inhibitors
E.g., protein kinase inhibitors, substrates for fusion (Halo/ Snap)-tagged proteins
Natural Products
Dyes and Fluorescent Labels
E.g., ICG, AMC, DAPI
Maillard & Amadori Reaction Products
Large portfolio of derivatives useful as standards for food, pharma and cosmetics industry
Vitamins
Custom Synthesis
Your project requires a compound not listed in our portfolio? Get in contact and inquire about our custom synthesis capabilities.
Our experienced scientists are excited to accept your synthetic challenge! In such cases, your request undergoes the following stages:
Step-by-Step Analysis
Customer’s demands
Process Evaluation
Det ailed literature review
Synthetic possibilities
Strategy Development
Protocol development
Method development and validation
• Customized synthesis
Our Service Promise
Quality Consistency
• Identity confirmation Purity verification
All our services are based on high standards, transparency & documentation, trust, honesty & confidentiality, as well as the required know-how.
High Standards
Values: sustainability & responsibility
State-of-the-art equipment & latest technologies
High quality standards
Qualified suppliers & regular audits
Trust, Honesty & Confidentiality
Intergenerational business valuing partnerships
• Meeting the customer‘s expectations
• Integrity towards our customers
Transparency & Documentation
Talk to our specialists – customer care
Certificates of analysis & origin
Impurity profiling
Safety data sheets
• Analytical and process reports
Our Know-How
One-step reactions & complex multi-step synthesis
• Scalability from mg to kg quantities
• Route scouting


1.2.
1.2.1.
1.2.2.
2.2.
6.1.
6.2.
6.3.
1. The Click Reaction
1.1. The 1st Generation Click Reaction: CuAAC
Alkynes and azides can undergo a Cu(I)-catalyzed azide-alkyne 1,3-dipolar cycloaddition (CuAAC) to afford 1,4-disubstituted 1,2,3-triazoles. Developed by K. Barry Sharpless and Morton Meldal, this type of chemical transformation was quickly dubbed “Click chemistry”. It has since become a widely used reaction that is orthogonal to many other types of chemical transformations and is used in various kinds of applications. Due to its high thermodynamic driving force, which is usually greater than 20 kcal/ mol, the Click reaction rapidly proceeds to completion in almost all cases. Moreover, while the thermal Huisgen 1,3-dipolar cycloaddition affords a mixture of both the 1,4-disubstituted and the 1,5-disubstituted regioisomers, the CuAAC is highly selective for the 1,4-disubstituted isomer only (Fig. 1). Worth noting is the fact that ruthenium is also able to catalyze a 1,3-dipolar cycloaddition between an azide and an alkyne affording the 1,5-disubstituted product instead.
Fig. 1: The copper-catalyzed azide-alkyne cycloaddition affords the 1,4-disubstituted isomers.
Cycloaddition reactions such as the [3+2] azide-alkyne and the [4+2] Diels-Alder reaction, have become common conjugation techniques. Applications range from imaging and drug design to the development of sensors, thereby covering such diverse fields as chemical biology, material science, surface and polymer chemistry.
Tris(benzyltriazolylmethyl)amine (TBTA; RL-2010 on page 87) is stabilizing copper(I) towards oxidation in solution by forming a complex and effectively catalyzes quantitative and regioselective Click cycloaddition reactions in a variety of aqueous and organic solvents. Among scientists, CuAAC has found widespread use as a biochemical tool for the site-specific labeling of peptides, proteins, and other biomolecules.
THPTA (RL-2210 on page 87) is a water-soluble alternative to TBTA (RL-2010 on page 87) and a highly efficient ligand for Click chemistry in partially organic and particularly in completely aqueous reactions. The benefits of a completely aqueous reaction include the biological labelling of live cells or the labelling of proteins without the concern of denaturing secondary structures. THPTA complexes Cu(I) and thus blocks its bioavailability. This mitigates potentially toxic effects while maintaining the catalytic effectiveness in Click conjugations. Successful Click reactions with oligonucleotides can be found in many publications.
A variety of azido and alkyne building blocks are available from Iris Biotech. Some of those compounds can be incorporated into peptides and proteins by recombinant syntheses, particularly by non-natural protein translation using the amber-suppression-based orthogonal system, while others are suitable for solid phase peptide synthesis. The presence of an azido or alkyne function at a particular position of a peptide sequence opens up the possibility for the site-selective conjugation of other biomolecules (e.g., carbohydrates), labels or APIs.
References:
→ A Stepwise Huisgen Cycloaddition Process: Copper(I)-Catalyzed Regioselective “Ligation” of Azides, Terminal Alkynes; V. V. Rostovtsev, L. G. Green, V. V. Fokin, K. B. Sharpless; Angew. Chem. Int. Ed. 2002; 41: 2596-2599. arrow-up-right-from-square https://doi.org/10.1002/1521-3773(20020715)41:14<2596::Aid-anie2596>3.0.Co;2-4
→ Peptidotriazoles on solid phase: [1,2,3]-triazoles by regiospecific copper(i)-catalyzed 1,3-dipolar cycloadditions of terminal alkynes to azides; C. W. Tornoe, C. Christensen, M. Meldal; J Org Chem 2002; 67: 3057-64. arrow-up-right-from-square https://doi.org/10.1021/jo011148j
→ Click Chemistry: Diverse Chemical Function from a Few Good Reactions; H. C. Kolb, M. G. Finn, K. B. Sharpless; Angew. Chem. Int. Ed. 2001; 40: 2004-2021. arrow-up-right-from-square https://doi.org/10.1002/1521-3773(20010601)40:11<2004::aid-anie2004>3.0.co;2-5
→ The growing impact of click chemistry on drug discovery; H. C. Kolb, K. B. Sharpless; Drug Discov Today 2003; 8: 1128-37. arrow-up-right-from-square https://doi.org/10.1016/S1359-6446(03)02933-7
→ Polytriazoles as copper(I)-stabilizing ligands in catalysis; T. R. Chan, R. Hilgraf, K. B. Sharpless, V. V. Fokin; Org Lett 2004; 6: 2853-5. arrow-up-right-from-square https://doi.org/10.1021/ol0493094
→ CuI-Catalyzed Alkyne-Azide “Click” Cycloadditions from a Mechanistic and Synthetic Perspective; V. D. Bock, H. Hiemstra, J. H. van Maarseveen; Eur. J. Org. Chem. 2006; 1: 51-68. arrow-up-right-from-square https://doi.org/10.1002/ejoc.200500483
→ A3-type star polymers via click chemistry; O. Altintas, B. Yankul, G. Hizal, U. Tunca; J. Polym. Sci. A. Polym. Chem. 2006; 44: 6458-6465. arrow-up-right-from-square https://doi.org/10.1002/pola.21728
→ Preparation of alumina supported copper nanoparticles and their application in the synthesis of 1,2,3-triazoles; M. L. Kantam, V. S. Jaya, B. Sreedhar, M. M. Rao, B. M. Choudary; J. Mol. Catal. A Chem. 2006; 256: 273-277. arrow-up-right-from-square https://doi.org/10.1016/j.molcata.2006.04.054
→ A rapid and versatile method to label receptor ligands using „click“ chemistry: Validation with the muscarinic M1 antagonist pirenzepine; D. Bonnet, B. Ilien, J. L. Galzi, S. Riche, C. Antheaune, M. Hibert; Bioconjug Chem 2006; 17: 1618-23. arrow-up-right-from-square https://doi.org/10.1021/bc060140j
→ Alkyne-azide click reaction catalyzed by metallic copper under ultrasound; P. Cintas, A. Barge, S. Tagliapietra, L. Boffa, G. Cravotto; Nat Protoc 2010; 5: 607-16. arrow-up-right-from-square https://doi.org/10.1038/nprot.2010.1
→ Synthesis of a DOTA--biotin conjugate for radionuclide chelation via Cu-free click chemistry; M. K. Schultz, S. G. Parameswarappa, F. C. Pigge; Org Lett 2010; 12: 2398-401. arrow-up-right-from-square https://doi.org/10.1021/ol100774p
→ ‚Click‘ cycloaddition catalysts: copper(I) and copper(II) tris(triazolylmethyl)amine complexes; P. S. Donnelly, S. D. Zanatta, S. C. Zammit, J. M. White, S. J. Williams; Chem Commun 2008: 2459-61. arrow-up-right-from-square https://doi.org/10.1039/b719724a
→ Click Chemistry and Radiochemistry: The First 10 Years; J. P. Meyer, P. Adumeau, J. S. Lewis, B. M. Zeglis; Bioconjug Chem 2016; 27: 2791-2807. arrow-up-right-from-square https://doi.org/10.1021/acs.bioconjchem.6b00561
1.2. Catalyst-free Click Reactions: 2nd and 3rd Generation Click Chemistry
Introduced in 2002, the copper-catalyzed variant of the azide-alkyne cycloaddition (CuAAC) reaction has found broad applicability in various fields and is as such currently the most widely used conjugation technique. The presence of copper, however, limits in vivo applications of this reaction for several reasons:
High cell toxicity
Undesired oxidation of proteins and
Inhibition of luminescence properties of nanocrystals
To allow for fast and efficient in vivo conjugations, new methodologies were developed that do not require the use of a metal catalyst while still making use of bioorthogonal functional groups. The most commonly used approaches can be classified into two categories.
1.2.1. 2nd Generation: Strain-Promoted Azide-Alkyne Cycloadditions (SPAAC)
As early as 1961, Wittig and Krebs noted the propensity of cyclooctyne to strongly react with phenyl azide via a 1,3-dipolar cycloaddition, forming a triazole product. This finding stood in stark contrast to previous research that found slow kinetics for Huisgen 1,3-dipolar cycloadditions of azides with unstrained, linear alkynes. The latter reaction can be drastically accelerated by copper catalysis. The use of this metal, however, is linked with several drawbacks as noted above.
This property of cyclooctynes was exploited by Bertozzi et al. in the design of SPAAC reagents for bioorthogonal couplings to azide-bearing biomolecules in live cells or organisms such as C. elegans, zebrafish or mice. By modifying the cyclooctyne core structure of SPAAC reagents with heteroatoms, fluorine substituents and fused rings, key properties such as cycloaddition kinetics, stability, solubility, and pharmacokinetics could be optimized.
strained cyclooctynes: DBCO
Fig. 2: Reaction of DBCO as an exemplary strained cyclooctyne with an azide.
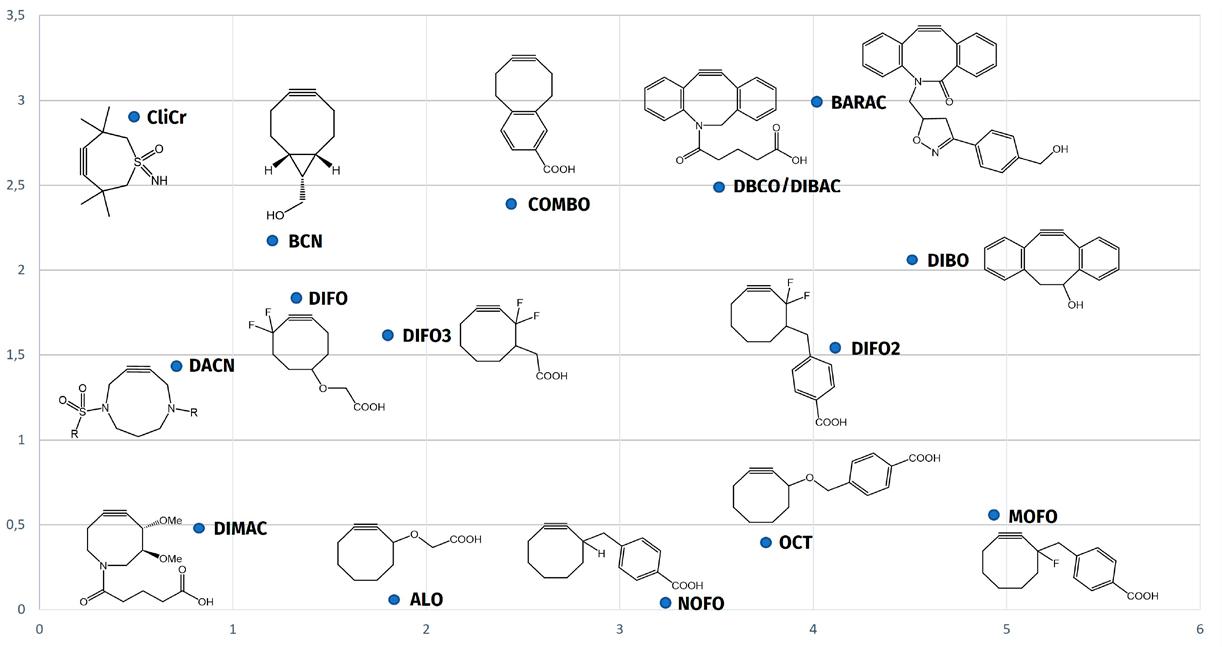
In Fig. 4, various strained cyclooctynes and cyclononynes are depicted with their corresponding reactivities, as determined by their reaction with benzyl azide as a model compound. In general, the presence of atoms with high electronegativity next to the alkyne function, i.e. good σ-acceptors, leads to increased reactivity. A higher reactivity also correlates with increased ring strain, as exemplified by dibenzo-fused cyclooctynes (DiBO, DBCO) and bicyclo[6.1.0]non-4-yne (BCN).
A relatively new addition to this ensemble is 4,8-diazacyclononyne (DACN). While exhibiting a reactivity twice as high as OCT, DACN is also more hydrophilic than most cyclooctynes, highly stable (both thermal and chemical stability), and highly selective towards ynophiles. Additionally, the two endocyclic nitrogens in DACN may serve as additional attachment points for further conjugation, rendering the compound functionally versatile.
CliCr (R) - an innovative Click reagent
CliCr® is based on the small-molecule TMTH-SulfoxImine (TMTHSI). It constitutes a superior class of reagents for metal-free click strain-promoted cycloaddition-conjugation with azides. The imine in the 7-membered CliCr® ring can be conveniently functionalized with a variety of linkers, e.g., via acylation, sulfonylation, N-alkylation, or carbamoylation. CliCr® reagents can be used in diverse applications, including the construction of antibody-drug conjugates (ADCs), small molecule-drug conjugates, oligonucleotide conjugates as well as for diagnostic labelling of a variety of nanoparticles and other agents. CliCr® can also be used in the conjugation of larger proteins (including optionality for native peptide release) or for ex vivo cell modification (e.g., glycocalyx).
3: Chemical structure of the CliCr® base compound and its derivatization possibilities.
Hydrophobicity (LogD)
Fig. 4: Reaction rate constants for different strained cycloheptyne (CliCr), cyclooctyne and cyclononyne (DACN) derivatives. arrow-up back to content
S O N L R
Fig.
Click Chemistry
Key benefits:
More attractive CoG via faster click reactions:
Shorter reaction times than all other marketed copper-free click reagents, providing greater chemical yield.
Generation of (biodegradable) bioconjugates:
Highly stable reagents yielding (biodegradable) linked drug products.
Greater variety of click reactions:
Due to a larger variation of linkers that can easily be attached to the seven-membered ring.
Broad applicability:
Next to straightforward bioconjugation, a plethora of additional important applications in biochemical, aqueous environments is envisioned, such as surface plasmon resonance (SPR) applications and conjugation of chelator moieties for radio active isotope incorporation in theragnostic applications.
CliCr® is shown to be > 5 times more reactive than BCN. The reaction progress of 5 mM CliCr® (blue line) or BCN-OH (red line), respectively, with 1.3 eq. of benzylazide in CDCl 3 at room temperature was monitored by MS. The reaction conversion was measured based on the increase of the triazole signals (see arrow-up-right-from-square https://doi.org/10.1039/d0sc03477k).




time
Fig. 5: Reaction progress with benzylazide in CDCl 3 – comparison of BCN-OH and TMTHSI.
The increased hydrophilicity as observed for antibody CliCr®-derived conjugates as compared to DBCO-linked constructs, translates into superior in vivo biodistribution, namely less liver and spleen uptake. Alike, also significantly faster and more cell labeling was detected upon using CliCr® as compared to DBCO.
Within our portfolio, we offer a selection of CliCr® derivatives. The CliCr® derivatives can be clicked to azide compounds with high efficiency and excellent stability. For further derivatives, large scale production or GMP grade, please get in contact!
CliCr® is provided under an intellectual property license from Cristal Therapeutics. The trademark CliCr® is the property of Cristal Therapeutics. For information on purchasing a license of CliCr® reagents, contact Cristal Therapeutics via Oxfordlaan 55, 6229 EV Maastricht (The Netherlands) or via info@cristaltherapeutics.com.
CliCr (R) Products
RL-4180 CliCr ® base compound
TMTH-Sulfoximine
CAS-No. 2408481-82-1
Formula C10 H17NOS Mol. weight 199,31 g/mol
RL-4190 CliCr ® -beta-Ala-NH2*TFA
TMTH-sulfoximine beta-alanine amide TFA salt
CAS-No. 3038495-92-7
Formula C13 H22N2O 2 S*CF 3 COOH Mol. weight 270,39*114,02 g/mol
RL-4200 CliCr ® -Suc
TMTH-sulfoximine succinic acid
CAS-No. 2479971-29-2
Formula C14H21NO4 S Mol. weight 299,39 g/mol
RL-4330 CliCr ® -OSu
TMTH-sulfoximine succinimidyl ester
CAS-No. 2408481-89-8
Formula C15H20 N2O 5 S Mol. weight 340,39 g/mol
RL-4205 CliCr ® Feasibility Service
This paid service package consists of 20 hours of consulting and advice by the CliCr ® experts of Cristal Therapeutics for the period of 1 year, paid in advance, no refunds. This Service Package will help to maximize your results when testing CliCr ® - the innovative click reagent. For more information, please get in contact!

Product
References:
→ TMTHSI, a superior 7-membered ring alkyne containing reagent for strain-promoted azide-alkyne cycloaddition reactions; J. Weterings, C. J. F. Rijcken, H. Veldhuis, T. Meulemans, D. Hadavi, M. Timmers, M. Honing, H. Ippel, R. M. J. Liskamp; Chem. Sci. 2020; 11: 9011-9016. arrow-up-right-from-square https://doi.org/10.1039/d0sc03477k
→ Exploring the Chemical Properties and Medicinal Applications of Tetramethylthiocycloheptyne Sulfoximine Used in Strain-Promoted Azide–Alkyne Cycloaddition Reactions; M. Timmers, A. Kipper, R. Frey, S. Notermans, M. Voievudskyi, C. Wilson, N. Hentzen, M. Ringle, C. Bovino, B. Stump, C. J. F. Rijcken, T. Vermonden, I. Dijkgraaf, R. Liskamp; Pharmaceuticals 2023; 16: 1155. arrow-up-right-from-square https://doi.org/10.3390/ph16081155
→ Specific N-terminal attachment of TMTHSI linkers to native peptides and proteins for strain-promoted azide alkyne cycloaddition; M. Timmers, W. Peeters, N. J. Hauwert, C. J. F. Rijcken, T. Vermonden, I. Dijkgraaf, R. M. J. Liskamp; Chem. Commun. 2023. arrow-up-right-from-square https://doi.org/10.1039/d3cc03397j
→ Versatile click linker enabling native peptide release from nanocarriers upon redox trigger; E. R. Hebels, S. Dietl, M. Timmers, J. Hak, A. van den Dikkenberg, C. J. F. Rijcken, W. E. Hennink, R. M. J. Liskamp, T. Vermonden; Bioconj. Chem. 2023, 34: 2375-2386. arrow-up-right-from-square https://doi.org/10.1021/acs.bioconjchem.3c00484
You want more details about CliCr (R)?
Watch the recording of our online workshop!
Products with DBCO
Product details
RL-2870 ICG-DBCO
Indocyanine green dibenzoazacyclooctyne
CAS-No. 3024705-52-7
Formula C 63 H 64N4O 5 S
Mol. weight 989,27 g/mol
PEG7465 Me-PEG(12)-DBCO
Methyl-12(ethylene glycol)-amido-dibenzoazacyclooctyne
Formula C 45H 68 N2O 14
Mol. weight 861,04 g/mol
RL-2120 DBCO-NH2
Dibenzocyclooctyne-amine
CAS-No. 1255942-06-3
Formula C18 H16 N2O Mol. weight 276,33 g/mol
RL-2430 DBCO-COOH
Dibenzoazacyclooctyne-carboxylic acid
CAS-No. 1425485-72-8
Formula C 21H19 NO 3 Mol. weight 333,38 g/mol
RL-2440 DBCO-NHS
Dibenzoazacyclooctyne-carboxylic acid succinimidyl ester
CAS-No. 1384870-47-6
Formula C 25H22N2O 5 Mol. weight 430,45 g/mol
RL-2490 DBCO-mal
Dibenzoazacyclooctyne-maleimide
CAS-No. 1395786-30-7
Formula C 25H21N3 O4 Mol. weight 427,45 g/mol
RL-4020 DBCO-C6-Alkyne
N-(propargylamidoadipoyl)-dibenzoazacyclooctyne
Formula C 24H22N2O 2 Mol. weight 370,45 g/mol
ADC1620 DBCO-cyclobutane-1,1-dicarboxamide-Ala-PAB dibenzoazacyclooctyne-cyclobutane-1,1-dicarboxamide-alanyl-(4-aminobenzyl alcohol)
CAS-No. 2576471-46-8
Formula C 34H34N4O 5 Mol. weight 578,66 g/mol
ADC1630 DBCO-cyclobutane-1,1-dicarboxamide-Ala-PAB-PNP
dibenzoazacyclooctyne-cyclobutane-1,1-dicarboxamide-alanyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
CAS-No. 2576471-43-5
Formula C 41H37N 5 O 9 Mol. weight 743,76 g/mol
ADC1520 DBCO-cyclobutane-1,1-dicarboxamide-Cit-PAB
dibenzoazacyclooctyne-cyclobutane-1,1-dicarboxamide-citrullyl-(4-aminobenzyl alcohol)
CAS-No. 2576471-51-5
Formula C 37H40 N 6 O 6 Mol. weight 664,75 g/mol
ADC1530 DBCO-cyclobutane-1,1-dicarboxamide-Cit-PAB-PNP
dibenzoazacyclooctyne-cyclobutane-1,1-dicarboxamide-citrullyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
CAS-No. 2576471-34-4
Formula C 44H43 N7 O 10 Mol. weight 829,85 g/mol
RL-2480 DBCO-PEG(3)-BisSulfonThiol-Linker
Dibenzoazacyclooctyne-PEG(3)-BisSulfon-Thiol-Linker
Formula
Mol. weight 1140,39 g/mol
RL-3670 Halo-DBCO
N-[2-[2-[(6-chlorohexyl)oxy]ethoxy]ethyl]-gamma-oxodibenz[b,f]azocine-5(6H)-butanamide
CAS-No. 1808119-16-5
Formula C 29 H35 ClN2O4 Mol. weight 511,06 g/mol
LS-4270 Biotin-DBCO
(3aS,4S,6aR)-N-[3-(11,12-Didehydrodibenz[b,f] azocin-5(6H)-yl)-3-oxopropyl]hexahydro-2-oxo-1Hthieno[3,4-d]imidazole-4-pentanamide
CAS-No. 1418217-95-4
Formula C 28 H30 N4O 3 S Mol. weight 502,63 g/mol
RL-2520 Biotin-PEG(4)-DBCO
Dibenzoazacyclooctyne-tetra(ethylene glycol)-biotin
CAS-No. 1255942-07-4
Formula C 39 H 51N 5 O 8 S Mol. weight 749,92 g/mol
PEG8140 Biotin-PEG(4)-Dde-DBCO
N-(15-(4,4-dimethyl-2,6-dioxocyclohexylidene)-19-oxo19-(azadibenzocyclooctyn-1-yl)-3,6,9,12-tetraoxa-16azanonadecyl)-5-((3aS,4S,6aR)-2-oxohexahydro-1Hthieno[3,4-d]imidazol-4-yl)pentanamide
CAS-No. 1807512-43-1
Formula C 47H 61N 5 O 9 S Mol. weight 872,08 g/mol
PEG8120 Biotin-PEG(4)-SS-DBCO
N-(2-((3-(3-(azadibenzocyclooctyn-1-yl)-3-oxopropylamino)-3-oxopropyl)disulfanyl)ethyl)-1-(5-((3aS,4S,6aR)2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamido)-3,6,9,12-tetraoxapentadecan-15-amide
Formula C 44H 60 N 6 O 9 S 3 Mol. weight 913,18 g/mol
RL-2420 DBCO-PEG(4)-NH2*TFA
Dibenzoazacyclooctyne-tetra(ethylene glycol)-amine trifluoro acetic acid salt
CAS-No. 1255942-08-5
Formula C 29 H37N3 O 6*C 2F 3 HO 2 Mol. weight 523,62*114,02 g/mol
RL-2421 DBCO-Sulfo-PEG(4)-NH2
Dibenzoazacyclooctyne-tetra(ethylene glycol)amine
CAS-No. 2055198-05-3
Formula C 32H42N4O 10 S Mol. weight 674,76 g/mol
RL-2510 DBCO-PEG(4)-OH
Dibenzoazacyclooctyne-tetra(ethylene glycol)
CAS-No. 1416711-60-8
Formula C 29 H36 N2O 6 Mol. weight 508,61 g/mol
Click Chemistry
PEG6790
Bromoacetamido-PEG(4)-DBCO
Bromoacetamido-tetra(ethylene glycol)-amido-dibenzoazacyclooctyne
CAS-No. 2735663-70-2
Formula C 31H38 BrN3 O 7 Mol. weight 644,55 g/mol
PEG6740 DBCO-PEG(4)-TFP
Dibenzoazacyclooctyne-tetra(ethylene glycol)-propionyl 2,3,5,6-tetrafluorophenol ester
CAS-No. 2247993-79-7
Formula C 37H38 F4N2O 8 Mol. weight 714,7 g/mol
RL-2500 DBCO-PEG(4)-mal
Dibenzoazacyclooctyne-tetra(ethylene glycol)-maleimide
CAS-No. 1480516-75-3
Formula C 36 H42N4O 9 Mol. weight 674,74 g/mol
RL-2450 DBCO-PEG(5)-COOH
Dibenzoazacyclooctyne-penta(ethylene glycol)-propanoic acid
CAS-No. 1870899-46-9
Formula C 32H40 N2O 9 Mol. weight 596,67 g/mol
RL-2460
DBCO-PEG(5)-NHS
Dibenzoazacyclooctyne-penta(ethylene glycol)-propanoic acid succinimidyl ester
CAS-No. 1378531-80-6
Formula C 36 H43 N3 O 11 Mol. weight 693,74 g/mol
PEG6800
Bromoacetamido-PEG(12)-DBCO
Bromoacetamido-dodeca(ethylene glycol)-amido-dibenzoazacyclooctyne
CAS-No. 2852742-40-4
Formula C 47H70 BrN3 O 17 Mol. weight 996,97 g/mol
PEG6810
Bromoacetamido-PEG(24)-DBCO
Bromoacetamido-24(ethylene glycol)-amido-dibenzoazacyclooctyne
CAS-No. 2852742-74-4
Formula C 71H118 BrN3 O 27 Mol. weight 1525,6 g/mol
PEG6750 DBCO-PEG(12)-TFP
Dibenzoazacyclooctyne-dodeca(ethylene glycol)-propionyl 2,3,5,6-tetrafluorophenol ester
CAS-No. 3038549-18-4
Formula C 53 H70 F4N2O 16 Mol. weight 1067,12 g/mol
PEG6770 DBCO-PEG(12)-MAL
Dibenzoazacyclooctyne-dodeca(ethylene glycol)-maleimide
CAS-No. 2011777-01-6
Formula C 52H74N4O 17 Mol. weight 1027,16 g/mol
PEG6830 DBCO-PEG(12)-(5)6-carboxyfluorescein
Dibenzoazacyclooctyne-dodeca(ethylene glycol)-(5)6-carboxyfluorescein
Formula C 46 H39 N3 O 10 Mol. weight 1234,34 g/mol
PEG7460 DBCO-PEG(24)-OMe
alpha-Methoxy-24(ethylene glycol)-amido-dibenzoazacyclooctyne
CAS-No. 3023098-71-4
Formula C 68 H114N2O 26 Mol. weight 1375,63 g/mol
PEG6760 DBCO-PEG(24)-TFP
Dibenzoazacyclooctyne-24(ethylene glycol)-propionyl 2,3,5,6-tetrafluorophenol ester
CAS-No. 2754372-40-0
Formula C 77H118 F4N2O 28 Mol. weight 1595,75 g/mol
Click Chemistry
PEG6780 DBCO-PEG(24)-MAL
Dibenzoazacyclooctyne-24(ethylene glycol)-maleimide
CAS-No. 2924872-84-2
Formula C 76 H122N4O 29 Mol. weight 1555,79 g/mol
PEG6765 DBCO-PEG(36)-TFP
Dibenzoazacyclooctyne-36(ethylene glycol)-propionyl 2,3,5,6-tetrafluorophenol ester
CAS-No. 2924873-16-3
Formula C101H166 F4N2O40 Mol. weight 2124,41 g/mol
RL-2530 DBCO-mPEG (5kDa)
alpha-Dibenzoazacyclooctyne-omega-methoxy-poly(ethylene glycol)
CAS-No. 2262541-53-5 Mol. weight 5000 Da
RL-2540 DBCO-mPEG (10kDa)
alpha-Dibenzoazacyclooctyne-omega-methoxy-poly(ethylene glycol)
CAS-No. 2262541-53-5 Mol. weight 10000 Da
RL-2550 DBCO-mPEG (20kDa)
alpha-Dibenzoazacyclooctyne-omega-methoxy-poly(ethylene glycol)
CAS-No. 2262541-53-5 Mol. weight 20000 Da
RL-2560 DBCO-mPEG (30kDa)
alpha-Dibenzoazacyclooctyne-omega-methoxy-poly(ethylene glycol)
CAS-No. 2262541-53-5 Mol. weight 30000 Da
RL-4110
DBCO-Suc-SS-COOH
CAS-No. 2749426-25-1
Formula C 24H24N2O4 S 2 Mol. weight 468,59 g/mol
RL-4310
DBCO-Suc-CBT
N1-(2-cyanobenzo[d]thiazol-6-yl)-N4-(3-(11,12-didehydro-5,6-dihydro-dibenzo[b,f]azocin-yl)-3-oxopropyl) succinamide
Formula C 30 H23 N 5 O 3 S Mol. weight 533,61 g/mol
PEG7070 Tetra(-PEG(11)-DBCO)pentaerythritol
Formula C193 H288 N12O 60 Mol. weight 3736,45 g/mol
PEG7075 Bis-PEG(11)-DBCO
Formula C 64H 82N4O 15 Mol. weight 1147,37 g/mol
PEG7095 DBCO-PEG(24)-amido-PEG(24)-DSPE
Formula C163 H299 N4O 60 P Mol. weight 3306,13 g/mol
RL-8755
DBCO-NH-SS-Bzl-OpNC
2-((2-(4-(11,12-didehydrodibenzo[b,f]azocin-5(6H)-yl)-4-oxobutanamido)ethyl)disulfaneyl)ethyl (4-nitrophenyl) carbonate
Formula C 35H29 N3 O 7S 2 Mol. weight 667,75 g/mol
RL-8760 DBCO-NH-SS-OpNC
4-((2-(4-(11,12-didehydrodibenzo[b,f]azocin-5(6H)-yl)-4-oxobutanamido)ethyl)disulfaneyl) benzyl (4-nitrophenyl) carbonate
Formula C 30 H27N3 O 7S 2
Mol. weight 605,68 g/mol
RL-8810
Dibromo-pyridazinedione-PEG(4)-DBCO
1-(3-(4,5-dibromo-2-methyl-3,6-dioxo-3,6-dihydropyridazin-1(2H)-yl)propanamido)-N-(3-(11,12-dihydrodibenzo[b,f]azocin-5(6H)-yl)-3-oxopropyl)-3,6,9,12-tetraoxapentadecan-15-amide
Formula C 37H43 Br2N 5 O 9
Mol. weight 861,58 g/mol
RL-8820 Monobromo-maleimido-PEG(4)-DBCO
1-(2-(3-dibromo-2,5-dioxo-2,5-dihydro-1H-pyrrol1-yl)acetamido)-N-(3-(11,12-didehydrodibenzo[b,f] azocin-5(6H)-yl)-3-oxopropyl)-3,6,9,12-tetraoxapentadecan-15-amide
Formula C 35H39 BrN4O 9 Mol. weight 739,62 g/mol
RL-8830 Monobromo-pyridazinedione-PEG(4)-DBCO
1-(3-(4/5-bromo-2-methyl-3,6-dioxo-3,6-dihydropyridazin-1(2H)-yl)propanamido)-N-(3-(11,12-didehydrodibenzo[b,f]azocin-5(6H)-yl)-3-oxopropyl)-3,6,9,12-tetraoxapentadecan-15-amide (mixture of regioisomers)
Formula C 37H44BrN 5 O 9 Mol. weight 782,69 g/mol
RL-8840 DBCO-Alkyne
N-(3-(11,12-didehydrodibenzo[b,f]azocin-5(6H)-yl)-3oxopropyl)pent-4-ynamide
CAS-No. 1887761-13-8
Formula C 23 H20 N2O 2 Mol. weight 356,43 g/mol
RL-8845 DBCO-PEG(4)-Alkyne
N-(3-(11,12-didehydrodibenzo[b,f]azocin-5(6H)-yl)-3-oxopropyl)-4,7,10,13-tetraoxahexadec-15-ynamide
Formula C 30 H34N2O 6 Mol. weight 518,61 g/mol
Products with DACN
RL-3600 DACN(Ms)*HCl
N-(Mesyl)-4,8-diazacyclononyne hydrochloride
CAS-No. 2331322-16-6
Formula C 8 H14N2O 2 S*HCl Mol. weight 202,27*36,46 g/mol
RL-2735 DACN(Tos)*HCl
N-( p-toluenesulfonyl)-4,8-diazacyclononyne hydrochloride
CAS-No. 2331322-18-8
Formula C14H18 N2O 2 S*HCl Mol. weight 278,37*36,46 g/mol
RL-2720 DACN(Tos,Suc-OH)
N-succinoyl-N‘-( p-toluenesulfonyl)-4,8-diazacyclononyne
CAS-No. 2109751-68-8
Formula C18 H22N2O 5 S Mol. weight 378,44 g/mol
RL-2710 DACN(Tos,Ns)
N-(o-nitrobenzenesulfonyl)-N‘-( p-toluenesulfonyl)-4,8-diazacyclononyne
CAS-No. 1797508-58-7
Formula C 20 H21N3 O 6 S 2 Mol. weight 463,53 g/mol
RL-2730 DACN(Tos2)
N,N‘-bis( p-toluenesulfonyl)-4,8-diazacyclononyne
CAS-No. 1797508-57-6
Formula C 21H24N2O4 S 2 Mol. weight 432,56 g/mol
RL-2737 DACN(Tos2,6-OH)
4,8-Bis( p-toluenesulfonyl)-4,8-diazacyclononyn-6-ol
CAS-No. 2109751-74-6
Formula C 21H24N2O 5 S 2 Mol. weight 448,55 g/mol
References:
→ A strain-promoted [3 + 2] azide-alkyne cycloaddition for covalent modification of biomolecules in living systems; N. J. Agard, J. A. Prescher, C. R. Bertozzi; J Am Chem Soc 2004; 126: 15046-7. arrow-up-right-from-square https://doi.org/10.1021/ja044996f
→ A hydrophilic azacyclooctyne for Cu-free click chemistry; E. M. Sletten, C. R. Bertozzi; Org Lett 2008; 10: 3097-9. arrow-up-right-from-square https://doi.org/10.1021/ol801141k
→ Live-cell imaging of cellular proteins by a strain-promoted azide-alkyne cycloaddition; K. E. Beatty, J. D. Fisk, B. P. Smart, Y. Y. Lu, J. Szychowski, M. J. Hangauer, J. M. Baskin, C. R. Bertozzi, D. A. Tirrell; Chembiochem 2010; 11: 2092-5. arrow-up-right-from-square https://doi.org/10.1002/cbic.201000419
→ Bioconjugation with strained alkenes and alkynes; M. F. Debets, S. S. van Berkel, J. Dommerholt, A. T. Dirks, F. P. Rutjes, F. L. van Delft; Acc Chem Res 2011; 44: 805-15. arrow-up-right-from-square https://doi.org/10.1021/ar200059z
→ Reactivity of biarylazacyclooctynones in copper-free click chemistry; C. G. Gordon, J. L. Mackey, J. C. Jewett, E. M. Sletten, K. N. Houk, C. R. Bertozzi; J Am Chem Soc 2012; 134: 9199-208. arrow-up-right-from-square https://doi.org/10.1021/ja3000936
→ Bioorthogonal labelling of biomolecules: new functional handles and ligation methods; M. F. Debets, J. C. van Hest, F. P. Rutjes; Org Biomol Chem 2013; 11: 6439-55. arrow-up-right-from-square https://doi.org/10.1039/c3ob41329b
→ 18F-labeling using click cycloadditions; K. Kettenbach, H. Schieferstein, T. L. Ross; Biomed Res Int 2014; 2014: 361329. arrow-up-right-from-square https://doi.org/10.1155/2014/361329
→ Click Chemistry and Radiochemistry: The First 10 Years; J. P. Meyer, P. Adumeau, J. S. Lewis, B. M. Zeglis; Bioconjug Chem 2016; 27: 2791-2807. arrow-up-right-from-square https://doi.org/10.1021/acs.bioconjchem.6b00561
→ Strain-Promoted 1,3-Dipolar Cycloaddition of Cycloalkynes and Organic Azides; J. Dommerholt, F. Rutjes, F. L. van Delft; Top Curr Chem 2016; 374: 16. arrow-up-right-from-square https://doi.org/10.1007/s41061-016-0016-4
→ A facile preparation of functional cycloalkynes via an azide-to-cycloalkyne switching approach; S. Yoshida, T. Kuribara, H. Ito, T. Meguro, Y. Nishiyama, F. Karaki, Y. Hatakeyama, Y. Koike, I. Kii, T. Hosoya; Chem Commun (Camb) 2019; 55: 3556-3559. arrow-up-right-from-square https://doi.org/10.1039/c9cc01113g
→ Selective strain-promoted azide-alkyne cycloadditions through transient protection of bicyclo[6.1.0] nonynes with silver or gold; K. Adachi, T. Meguro, Y. Sakata, K. Igawa, K. Tomooka, T. Hosoya, S. Yoshida; Chem Commun (Camb) 2020; 56: 9823-9826. arrow-up-right-from-square https://doi.org/10.1039/d0cc04606j
→ Facile assembly of three cycloalkyne-modules onto a platform compound bearing thiophene S,S-dioxide moiety and two azido groups; T. Meguro, Y. Sakata, T. Morita, T. Hosoya, S. Yoshida; Chem Commun (Camb) 2020; 56: 4720-4723. arrow-up-right-from-square https://doi.org/10.1039/d0cc01810d
→ Copper-Free Huisgen Cycloaddition for the 14-3-3-Templated Synthesis of Fusicoccin-Peptide Conjugates; R. Masuda, Y. Kawasaki, K. Igawa, Y. Manabe, H. Fujii, N. Kato, K. Tomooka, J. Ohkanda; Chem Asian J 2020; 15: 742-747. arrow-up-right-from-square https://doi.org/10.1002/asia.202000042
1.2.2.
3rd Generation:
Inverse Electron-Demand Diels-Alder (IEDDA) Reactions
Click chemistry is frequently the method of choice for site-selective labeling and crosslinking. However, in biological systems, the cytotoxicity of copper used for the classical Cu-promoted 1,3-dipolar cycloaddition may cause major problems. The copper-free strain-promoted alkyne-azide cycloaddition (SPAAC) utilizing cyclooctynes, on the other hand, is limited by its moderate reaction kinetics for the application in live cells, where the concentration of biomolecules is usually low. Another potential drawback of cyclooctynes is the extensive patent coverage of many variants.
Tetrazine ligation presents the option for a copper-free, rapid, and fully bioorthogonal type of Click chemistry. Mechanistically, this reaction proceeds via an inverse electron-demand Diels-Alder cycloaddition reaction between a tetrazine and a strained alkene such as trans-cyclooctene (TCO), cyclopropane or norbornene, followed by a retro-Diels-Alder reaction under elimination of N 2 , the latter rendering the reaction irreversible.
Fig. 6: Reaction between a trans-cyclooctene (TCO) and a tetrazine.
Stability vs. Faster Reaction Kinetics: 6-Me or 6-H Tetrazines
Fig. 7: Chemical structures of 6-Me and 6-H tetrazine.
There are two main types of tetrazines that are widely applied: 6-methyl-substituted tetrazines and 6-hydrogen-substituted tetrazines. Methyl-substituted tetrazines exhibit a high stability even when dissolved in aqueous media, while still offering faster reaction kinetics with TCO derivatives than any other bioorthogonal reaction pairs (approx. 1000 M-1s-1). Moreover, they tolerate a wide array of reaction conditions which renders them the prime choice for applications such as protein labeling. Hydrogen-substituted tetrazines, on the other hand, show lower stability and less tolerance to harsh reaction conditions, but offer extremely fast reaction kinetics (up to 30000 M-1s-1) for applications like in vivo imaging.
Click Chemistry
This method excels at very low concentrations (e.g., in biological systems) due to the extremely rapid second order reaction rate constants (between approx. 800 M-1s-1 and 30000 M-1s-1). Moreover, the tetrazine-TCO ligation can be performed in aqueous media and has been applied in live cell imaging. These properties make tetrazine Click chemistry the method of choice for labeling or crosslinking biomolecules in living cells.
Fig. 8: Common reaction partners for tetrazines.
Choice of Spacer: Alkyl or PEG?
Tetrazines equipped with alkyl spacers are suitable for reactions in organic solvents. For applications in aqueous media, however, PEG-spacers are usually the superior choice. Moreover, tetrazines equipped with PEG-spacers are ideal for the functionalization of proteins since PEGs are known to reduce the aggregation of labeled polypeptides.
In summary, the reaction between a tetrazine (Tz) and a trans-cyclooctene (TCO) is the innovative third generation Click reaction that proceeds without the use of copper or other catalysts. It is rapid, fully bioorthogonal, irreversible and excels at very low concentrations.
Products with Tetrazine
HAA9470 H-L-Phe(4-MeTz)-OH*TFA (S)-2-amino-3-(4-(6-methyl-1,2,4,5-tetrazin-3-yl)phenyl) propanoic acid trifluoroacetic acid salt (S) OH
CAS-No. 1698038-85-5
Formula C12H13 N 5 O 2*CF 3 COOH Mol. weight 259,27*114,02 g/mol
Product details
HAA9480 H-L-Phe(4-Azido-PrTz)-OH*TFA
(S)-2-amino-3-(4-(6-(3-azidopropyl)-1,2,4,5-tetrazin-3yl)phenyl)propanoic acid trifluoroacetic acid salt
Formula C14H16 N 8 O 2*CF 3 COOH Mol. weight 328,34*114,02 g/mol
HAA9490 H-L-Phe(3-MeTz)-OH*TFA
(S)-2-amino-3-(3-(6-methyl-1,2,4,5-tetrazin-3-yl)phenyl) propanoic acid trifluoroacetic acid salt
CAS-No. 2036323-75-6
Formula C12H13 N 5 O 2*CF 3 COOH Mol. weight 259,27*114,02 g/mol
HAA9500 H-L-Phe(3-BuTz)-OH*TFA
(S)-2-amino-3-(3-(6-butyl-1,2,4,5-tetrazin-3-yl)phenyl) propanoic acid trifluoroacetic acid salt
CAS-No. 2036323-83-6 net
Formula C15H19 N 5 O 2*CF 3 COOH Mol. weight 301,35*114,02 g/mol
HAA9510 H-L-Phe(3-iPrTz)-OH*TFA
(S)-2-amino-3-(3-(6-isopropyl-1,2,4,5-tetrazin-3-yl) phenyl)propanoic acid trifluoroacetic acid salt
CAS-No. 2421119-12-0 net
Formula C14H17N 5 O 2*CF 3 COOH Mol. weight 287,32*114,02 g/mol
HAA9170 H-L-Lys(MeTz-PhAc)-OH*TFA
N-(2-(4-(6-methyl-1,2,4,5-tetrazin-3-yl)phenyl)acetyl)-L-lysine TFA salt
CAS-No. 2578384-82-2 (net)
Formula C17H22N 6 O 3*CF 3 COOH Mol. weight 358,40*114,02 g/mol
RL-2140 (Me)Tz-butanoic acid
4-(6-methyl-1,2,4,5-tetrazin-3-yl)butanoic acid
CAS-No. 1923268-81-8
Formula C 7H10 N4O 2 Mol. weight 182,18 g/mol
Click Chemistry
RL-3905 MeTz-PEG(4)-STP
sodium 2,3,5,6-tetrafluoro-4-((1-(4-(6-methyl-1,2,4,5tetrazin-3-yl)phenoxy)-3,6,9,12-tetraoxapentadecan-15-oyl)oxy)benzenesulfonate
Formula C 26 H27F4N4NaO 10 S Mol. weight 686,56 g/mol
RL-2360 MeTz-Bzl-NH2*HCl
Methyltetrazine-benzylamine*HCl
CAS-No. 1345955-28-3
Formula C10 H11N 5*HCl
Mol. weight 201,23*36,46 g/mol
RL-2130 (Me)Tz-benzoic acid
4-(6-methyl-1,2,4,5-tetrazin-3-yl)benzoic acid
CAS-No. 1345866-66-1
Formula C10 H 8 N4O 2 Mol. weight 216,2 g/mol
RL-2580 Tz-benzoic acid
4-(1,2,4,5-tetrazin-3-yl)benzoic acid
CAS-No. 1345866-65-0
Formula C9 H 6 N4O 2 Mol. weight 202,17 g/mol
RL-2300 MeTz-PhAcOH
Methyltetrazine-phenylacetic acid
CAS-No. 1380500-88-8
Formula C11H10 N4O 2
Mol. weight 230,22 g/mol
RL-2320 MeTz-PhAc-NHS
Methyltetrazine-phenylacetyl succinimidyl ester
CAS-No. 1644644-96-1
Formula C15H13 N 5 O4 Mol. weight 327,29 g/mol
RL-3915 MeTz-PhAc-Sulfo-NHS
sodium 1-(2-(4-(6-methyl-1,2,4,5-tetrazin-3-yl)phenyl) acetoxy)-2,5-dioxopyrrolidine-3-sulfonate
CAS-No. 1821017-46-2
Formula C15H12N 5NaO 7 Mol. weight 429,34 g/mol
RL-2230 Bz-(Me)Tz-NHS
2,5-dioxopyrrolidin-1-yl 5-(4-(6-methyl-1,2,4,5-tetrazin3-yl)benzylamino)-5-oxopentanoate
CAS-No. 1454558-58-7 Mol. weight 412,41 g/mol
RL-2240 Bz-Tz-NHS
2,5-dioxopyrrolidin-1-yl 5-(4-(1,2,4,5-tetrazin-3-yl) benzylamino)-5-oxopentanoate
CAS-No. 1244040-64-9
Formula C18 H18 N 6 O 5 Mol. weight 398,37 g/mol
RL-2370
MeTz-PEG(4)-NH2*HCl
Methyltetrazine-PEG(4)-amine HCL salt
CAS-No. 1802908-05-9
Formula C17H25N 5 O4*HCl Mol. weight 363,41*HCl g/mol
RL-2310
MeTz-PEG(4)-COOH
Methyltetrazine-PEG(4)-acid
CAS-No. 1802907-91-0
Formula C 20 H28 N4O 7 Mol. weight 436,56 g/mol
RL-2330
MeTz-PEG(4)-NHS
Methyltetrazine-PEG(4)-propanoyl succinimidyl ester
CAS-No. 1802907-92-1
Formula C 24H31N 5 O 9 Mol. weight 533,53 g/mol
RL-2340 MeTz-PEG(4)-mal
Methyltetrazine-PEG(4)-maleimide
CAS-No. 1802908-02-6
Formula C 24H30 N 6 O 7 Mol. weight 514,53 g/mol
RL-2250 Bz-Tz-PEG(5)-NHS
2,5-dioxopyrrolidin-1-yl 1-(4-(1,2,4,5-tetrazin-3-yl) phenyl)-3-oxo-6,9,12,15,18-pentaoxa-2-azahenicosan21-oate
CAS-No. 1682653-80-0
Formula C 27H36 N 6 O 10 Mol. weight 604,61 g/mol
LS-4280 Biotin-MeTz
N-(4-(6-methyl-1,2,4,5-tetrazin-3-yl)benzyl)-5((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamide
CAS-No. 1802883-51-7
Formula C 20 H25N7 O 2 S Mol. weight 427,53 g/mol
LS-4290 Biotin-PEG(4)-MeTz
N-(4-(6-methyl-1,2,4,5-tetrazin-3-yl)benzyl)-1-(5((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d] imidazol-4-yl)pentanamido)-3,6,9,12-tetraoxapentadecan-15-amide
CAS-No. 1962919-31-8
Formula C 31H46 N 8 O 7S Mol. weight 674,82 g/mol
Products with TCO
TCO1070 TCO-PEG(3)-NH2*HCl
trans-Cyclooctene-PEG(3)-amine
CAS-No. 2028288-77-7
Formula C19 H36 N2O 5*HCl Mol. weight 372,51*36,46 g/mol
Product details
TCO1060 TCO-NH2*HCI
trans-Cyclooctene-amine hydrochloride
CAS-No. 1800507-94-1
Formula C12H22N2O 2*HCl Mol. weight 226,32*36,45 g/mol
TCO1000 TCO-NHS
trans-Cyclooctene succinimidyl carbonate
CAS-No. 1191901-33-3
Formula C13 H17NO 5 Mol. weight 267,28 g/mol
TCO1040 TCO-PEG(4)-COOH
trans-Cyclooctene-PEG(4)-Acid
CAS-No. 1802913-21-8
Formula C 20 H35NO 8 Mol. weight 417,49 g/mol
TCO1010 TCO-PEG(4)-NHS
trans-Cyclooctene-PEG(4)-carboxy succinimidyl ester
CAS-No. 1621096-79-4
Formula C 24H38 N2O 10 Mol. weight 514,57 g/mol
TCO1050 TCO-PEG(3)-mal
trans-Cyclooctene-PEG(3)-maleimide
CAS-No. 1809356-72-6
Formula C 26 H41N3 O 8 Mol. weight 523,62 g/mol
TCO1020 TCO-PEG(12)-NHS
trans-Cyclooctene-PEG(12)-carboxy succinimidyl ester
CAS-No. 2185016-39-9
Formula C 40 H70 N2O 18 Mol. weight 866,99 g/mol
Click Chemistry
Products with Norbornene
HAA9235 H-L-Lys(Norbornene-methoxycarbonyl)-OH*HCl
N-epsilon-(norbornene-methoxycarbonyl)-L-lysine hydrochloride
CAS-No. 1378916-76-7
Formula C15H24N2O4*HCl Mol. weight 296,37*36,46 g/mol
RL-2080 Norbornene-NHS
(Norbornene-2-yl)-N-hydroxysuccinimidylcarbonate
CAS-No. 1888335-48-5
Formula C12H13 NO 5 Mol. weight 251,24 g/mol
Product details
RL-2090 Norbornene-methyl-NHS (Norbornene-2-methyl)-N-hydroxysuccinimidylcarbonate
CAS-No. 1986791-87-0
Formula C13 H15NO 5 Mol. weight 265,26 g/mol
References:
→ Biomedical applications of tetrazine cycloadditions; N. K. Devaraj, R. Weissleder; Acc Chem Res 2011; 44: 816-27. arrow-up-right-from-square https://doi.org/10.1021/ar200037t
→ trans-Cyclooctene–a stable, voracious dienophile for bioorthogonal labeling; R. Selvaraj, J. M. Fox; Curr Opin Chem Biol 2013; 17: 753-60. arrow-up-right-from-square https://doi.org/10.1016/j.cbpa.2013.07.031
→ Inverse electron demand Diels-Alder (iEDDA)-initiated conjugation: a (high) potential click chemistry scheme; A. C. Knall, C. Slugovc; Chem Soc Rev 2013; 42: 5131-42. arrow-up-right-from-square https://doi.org/10.1039/c3cs60049a
→ The growing impact of bioorthogonal click chemistry on the development of radiopharmaceuticals; D. Zeng, B. M. Zeglis, J. S. Lewis, C. J. Anderson; J Nucl Med 2013; 54: 829-32. arrow-up-right-from-square https://doi.org/10.2967/jnumed.112.115550
→ Highly accelerated inverse electron-demand cycloaddition of electron-deficient azides with aliphatic cyclooctynes; J. Dommerholt, O. van Rooijen, A. Borrmann, C. F. Guerra, F. M. Bickelhaupt, F. L. van Delft; Nat Commun 2014; 5: 5378. arrow-up-right-from-square https://doi.org/10.1038/ncomms6378
→ The inverse electron demand Diels-Alder click reaction in radiochemistry; T. Reiner, B. M. Zeglis; J Labelled Comp Radiopharm 2014; 57: 285-90. arrow-up-right-from-square https://doi.org/10.1002/jlcr.3149
→ 3,6-Substituted-1,2,4,5-tetrazines: tuning reaction rates for staged labeling applications; D. Wang, W. Chen, Y. Zheng, C. Dai, K. Wang, B. Ke, B. Wang; Org Biomol Chem 2014; 12: 3950-5. arrow-up-right-from-square https://doi.org/10.1039/c4ob00280f
→ Click Chemistry and Radiochemistry: The First 10 Years; J. P. Meyer, P. Adumeau, J. S. Lewis, B. M. Zeglis; Bioconjug Chem 2016; 27: 2791-2807. arrow-up-right-from-square https://doi.org/10.1021/acs.bioconjchem.6b00561
→ Bio-orthogonal Fluorescent Labelling of Biopolymers through Inverse-Electron-Demand Diels-Alder Reactions; E. Kozma, O. Demeter, P. Kele; Chembiochem 2017; 18 : 486-501. arrow-up-right-from-square https://doi.org/10.1002/cbic.201600607
→ Bicyclo[6.1.0]nonyne and tetrazine amino acids for Diels–Alder reactions; X. Li, Z. Liu, S. Dong; RSC Advances 2017; 7: 44470-44473. arrow-up-right-from-square https://doi.org/10.1039/c7ra08136g
→ Inverse electron demand Diels-Alder reactions in chemical biology; B. L. Oliveira, Z. Guo, G. J. L. Bernardes; Chem Soc Rev 2017; 46: 4895-4950. arrow-up-right-from-square https://doi.org/10.1039/c7cs00184c
→ Advances in Tetrazine Bioorthogonal Chemistry Driven by the Synthesis of Novel Tetrazines and Dienophiles; H. Wu, N. K. Devaraj; Acc Chem Res 2018; 51: 1249-1259. arrow-up-right-from-square https://doi.org/10.1021/acs.accounts.8b00062
→ Inverse electron demand Diels-Alder (IEDDA) reactions in peptide chemistry; M. Pagel; J. Pept. Sci. 2019; 25: e3141. arrow-up-right-from-square https://doi.org/10.1002/psc.3141
→ Bioorthogonal Fluorescence Turn-On Labeling Based on Bicyclononyne-Tetrazine Cycloaddition Reactions that Form Pyridazine Products; S. J. Siegl, J. Galeta, R. Dzijak, M. Dracinsky, M. Vrabel; Chempluschem 2019; 84: 493497. arrow-up-right-from-square https://doi.org/10.1002/cplu.201900176
→ An Extended Approach for the Development of Fluorogenic trans-Cyclooctene-Tetrazine Cycloadditions; S. J. Siegl, J. Galeta, R. Dzijak, A. Vazquez, M. Del Rio-Villanueva, M. Dracinsky, M. Vrabel; Chembiochem 2019; 20: 886-890. arrow-up-right-from-square https://doi.org/10.1002/cbic.201800711
→ Structural insights into incorporation of norbornene amino acids for click modification of proteins; S. Schneider, M. J. Gattner, M. Vrabel, V. Fluegel, V. Lopez-Carrillo, S. Pril, T. Carell; ChemBioChem 2013; 14(16) : 2114-2118. arrow-up-right-from-square https://doi.org/10.1002/cbic.201300435
→ A need for speed: genetic encoding of rapid cycloaddition chemistries for protein labelling in living cells; M. J. Schmidt, D. Summerer; ChemBioChem 2012; 13(11) : 1553-1557. arrow-up-right-from-square https://doi.org/10.1002/cbic.201200321
→ A genetically encoded norbornene amino acid for the mild and selective modification of proteins in a copper-free click reaction; E. Kaya, M. Vrabel, C. Deiml, S. Prill, V. S. Fluxa, T. Carell; Angew. Chem. Int. Ed. 2012; 51(18): 4466-4469. arrow-up-right-from-square https://doi.org/10.1002/anie.201109252
→ Amino Acids for Diels-Alder Reactions in Living Cells; T. Plass, S. Milles, C. Koehler, J. Szymansk, R. Mueller, M. Wiessler, C. Schultz, E. A. Lemke; Angew. Chem. Int. Ed. 2012; 51(17) : 4166-4170. arrow-up-right-from-square https://doi.org/10.1002/anie.201108231
. circle-arrow-right
We offer custom synthesis of DBCO, DACN, TCO and tetrazine derivatives and related conjugations. Send us your inquiry to info@iris-biotech.de!
1.2.3. Custom Synthesis of DBCO, Tetrazine and TCO Derivatives
Table 1: A selection of derivatives of tetrazine, TCO and DBCO available by custom synthesis.
DBCO with Cleavable Linkers
e.g., ADC linkers, Dde-based linkers, disulfide-based linkers.
DBCO-PEG-Derivatives
DBCO-PEG-NHS
DBCO-PEG-mal
DBCO-PEG-Bis-Sulfone-Thiol
DBCO-PEG-COOH
DBCO-PEG-NH2
With both monodisperse/uniform and polydisperse PEG-spacers.
DBCO-Biotin
With both monodisperse/uniform and polydisperse PEG-spacers.
DBCO-Dye
With both monodisperse and polydisperse PEG-spacers.
With the dye of your choice, e.g., ICG, (5)6-carboxyfluorescein.
Tetrazine-PEG-Derivatives
Tetrazine-PEG-NHS
Tetrazine-PEG-mal
Tetrazine-PEG-COOH
Tetrazine-PEG-NH2
With both monodisperse/uniform and polydisperse PEG-spacers.
Tetrazine-Biotin
With both monodisperse/uniform and polydisperse PEG-spacers, or alkyl-spacers.
Tetrazine-Dye
With both monodisperse/uniform and polydisperse PEG-spacers. With the dye of your choice, e.g., ICG, (5)6-carboxyfluorescein.
TCO-PEG-Derivatives
TCO-PEG-NHS
TCO-PEG-mal
TCO-PEG-COOH
TCO-PEG-NH2
. circle-arrow-right
You want to know more about our custom synthesis capabilities?
Check out our brochure!

2. Amino Acid Derivatives and Related Building Blocks for Click Chemistry
2.1. Recombinant Incorporation of Amino Acids into Proteins
Genetic code expansion is a powerful technology in proteomics, facilitating the site-specific incorporation of noncanonical amino acids (ncAAs) into proteins using the cellular machinery. A wide variety of ncAAs can be incorporated into proteins using this technology that relies on aminoacyl-tRNA synthetase/tRNA pairs.
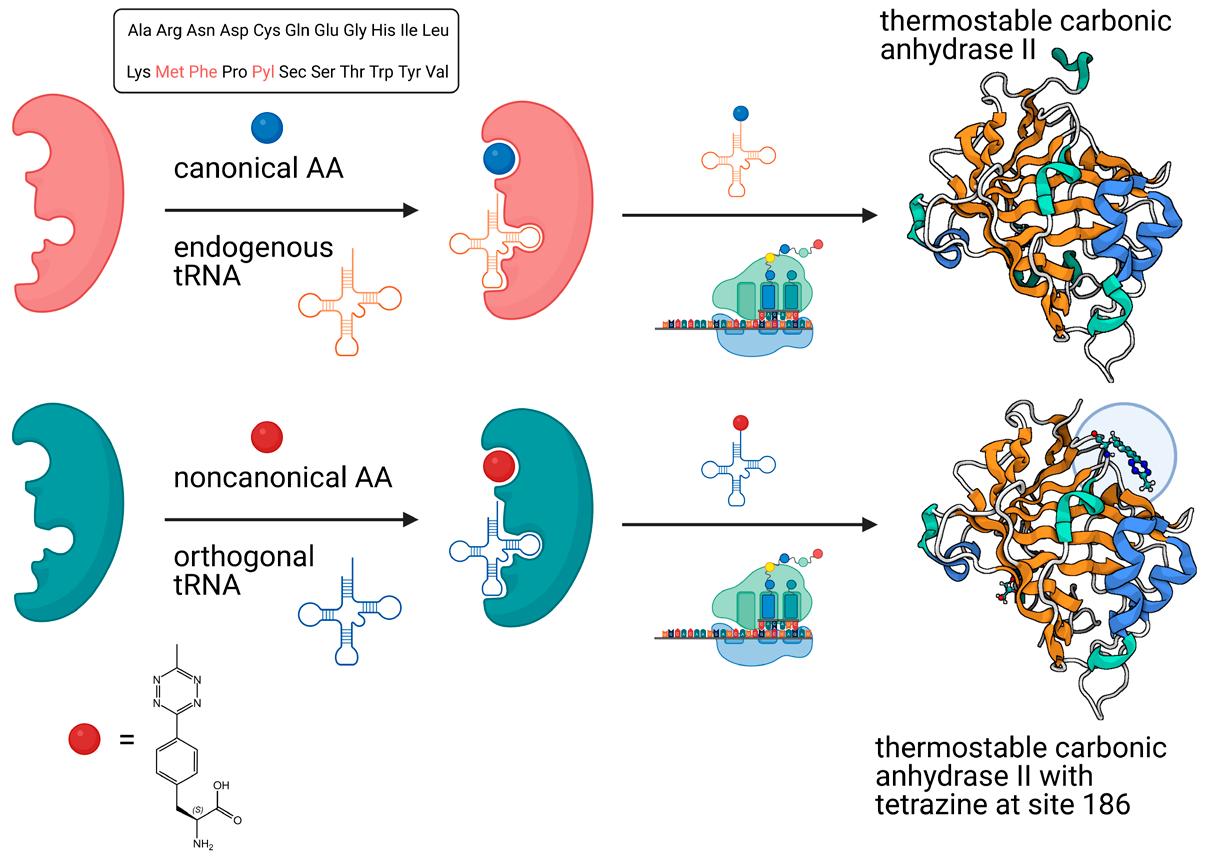
Fig. 9: Principle of genetic code expansion (protein structures adapted from Kean et al., Protein Sci. 2018 ; Bednar et al., ACS Appl. Mater. Interfaces 2019).
Certain amino acids such as azidohomoalanine (Aha) can be incorporated into proteins using the cell’s native translational apparatus. Aha is a structural analog of methionine (Met), and as such activated by the native methionyl-tRNA synthetase of Escherichia coli, replacing methionine in proteins expressed in methionine-depleted bacterial cultures. Aside from being a clickable amino acid, azidohomoalanine is an excellent conformationally sensitive IR probe to study protein folding and protein structure.
In most cases, researchers resort to engineering suitable aminoacyl-tRNA synthetase/tRNA pairs in order to incorporate ncAAs. For example, the usually promiscuous pyrrolysyl-tRNA synthetase (PylRS) machinery can be engineered to accommodate more than 100 ncAAs or α-hydroxy acids into proteins at amber codons, and can be reassigned to other codons such as ochre (UAA) or opal (UGA). Among the most prominent noncanonical amino acids that are routinely incorporated by engineered PylRS/ tRNAPyl pairs are azido and propargyl analogs of L-lysine, enabling the biochemist to site-specifically introduce an azido or alkyne group into a protein for further Click conjugation.
Recent developments in the field of genetic code expansion include the directed evolution of tRNA synthetases to improve substrate selectivity, as well as the reassignment of further codons to encode ncAAs. Once an azido or alkyne function has been built into the protein sequence, conjugation with a large number of diverse clickable compounds opens up a wide field of possibilities. Alternatively, a protein bearing an azido group can be selectively modified via Staudinger ligation (see Fig. 10). Many different applications from therapeutics to diagnostics can be addressed through conjugates with PEG-polymers, dyes, cofactors, antibodies, small molecules, toxins, additional proteins, and peptides.
alkyne-functionalized polymer, e.g., PEG or PAA
DBCO-functionalized payload
phosphine-functionalized dye
Fig. 10: Site-specific conjugation to an azido-functionalized IgG antibody.
Click Chemistry
One recent example of a sophisticated application of genetic code expansion is the site-directed incorporation of two different non-canonical amino acids into human erythropoietin via cell-free protein synthesis (Zemella et al., Sci. Rep. 2018). Either p-propargyloxyphenylalanine (pPa) or p-azido-L-phenyl-alanine (AzF) was incorporated into an erythropoietin amber-mutant (EPO-Amb) via amber suppression in a eukaryotic translationally active lysate. This eukaryotic system also facilitated the glycosylation of EPO which is known to be crucial for its pharmacokinetics. The recombinant EPO variants were subsequently labelled with various fluorophores, as well as functionalized with a PEG of 10 kDa. Similar to glycosylation, the attachment of PEGs has been shown to improve solubility, stability and activity of recombinantly produced EPO (Hoffmann et al., Mol. Biosyst. 2016).
The usefulness of the cell-free protein synthesis approach for the incorporation of ncAAs was further demonstrated by the preparation and ligand-free dimerization of functional human epidermal growth factor receptor (EGFR), a complex eukaryotic transmembrane protein (Quast et al., Sci. Rep. 2016). EGFR is a receptor tyrosine kinase that dimerizes and autophosphorylates upon binding to its ligand, thereby initiating an intracellular signal transduction cascade. In order to facilitate dimerization in the absence of a ligand, p-azido-L-phenylalanine (AzF) was site-selectively incorporated into EGFR, which was verified by Staudinger ligation of a phosphine dye to AzF. Two different EGFR amber mutants that incorporate AzF in the intracellular juxtamembrane domain were synthesized and reacted with a bis-COMBO Click-crosslinking reagent, thereby generating covalently linked receptor dimers.
References:
→ Genetic Code Expansion: Inception, Development, Commercialization; M. Manandhar, E. Chun, F. E. Romesberg; J Am Chem Soc 2021. arrow-up-right-from-square https://doi.org/10.1021/jacs.0c11938
→ Cell-free protein synthesis as a novel tool for directed glycoengineering of active erythropoietin; A. Zemella, L. Thoring, C. Hoffmeister, M. Samalikova, P. Ehren, D. A. Wustenhagen, S. Kubick; Sci Rep 2018; 8: 8514. arrow-up-right-from-square https://doi.org/10.1038/s41598-018-26936-x
→ Cell-free synthesis of functional human epidermal growth factor receptor: Investigation of ligand-independent dimerization in Sf21 microsomal membranes using non-canonical amino acids; R. B. Quast, B. Ballion, M. Stech, A. Sonnabend, B. R. Varga, D. A. Wustenhagen, P. Kele, S. M. Schiller, S. Kubick; Sci Rep 2016; 6: 34048. arrow-up-right-from-square https://doi.org/10.1038/srep34048
→ Cotranslational incorporation of non-standard amino acids using cell-free protein synthesis; R. B. Quast, D. Mrusek, C. Hoffmeister, A. Sonnabend, S. Kubick; FEBS Lett 2015; 589: 1703-12. arrow-up-right-from-square https://doi.org/10.1016/j.febslet.2015.04.041
→ Pyrrolysyl-tRNA synthetase: an ordinary enzyme but an outstanding genetic code expansion tool; W. Wan, J. M. Tharp, W. R. Liu; Biochim Biophys Acta 2014; 1844: 1059-70. arrow-up-right-from-square https://doi.org/10.1016/j.bbapap.2014.03.002
→ Genetic encoding and labeling of aliphatic azides and alkynes in recombinant proteins via a pyrrolysyl-tRNA Synthetase/tRNA(CUA) pair and click chemistry; D. P. Nguyen, H. Lusic, H. Neumann, P. B. Kapadnis, A. Deiters, J. W. Chin; J Am Chem Soc 2009; 131: 8720-1. arrow-up-right-from-square https://doi.org/10.1021/ja900553w
→ High-throughput screening for methionyl-tRNA synthetases that enable residue-specific incorporation of noncanonical amino acids into recombinant proteins in bacterial cells; T. H. Yoo, D. A. Tirrell; Angew. Chem. Int. Ed. 2007; 46: 5340-3. arrow-up-right-from-square https://doi.org/10.1002/anie.200700779
→ Direct charging of tRNACUA with pyrrolysine in vitro and in vivo; S. K. Blight, R. C. Larue, A. Mahapatra, D. G. Longstaff, E. Chang, G. Zhao, P. T. Kang, K. B. Green-Church, M. K. Chan, J. A. Krzycki; Nature 2004; 431: 333. arrow-up-right-from-square https://doi.org/10.1038/nature02895
→ Incorporation of azides into recombinant proteins for chemoselective modification by the Staudinger ligation; K. L. Kiick, E. Saxon, D. A. Tirrell, C. R. Bertozzi; Proc Natl Acad Sci U S A 2002; 99: 19-24. arrow-up-right-from-square https://doi.org/10.1073/pnas.012583299
→ Global replacement of tryptophan with aminotryptophans generates non-invasive protein-based optical pH sensors; N. Budisa, M. Rubini, J. H. Bae, E. Weyher, W. Wenger, R. Golbik, R. Huber, L. Moroder; Angew. Chem. Int. Ed. 2002; 41: 4066-9. arrow-up-right-from-square https://doi.org/10.1002/1521-3773(20021104)41:21<4066::AID-ANIE4066>3.0.CO;2-6
As part of daily lab procedures, proteins and peptides need to be kept in their reduced state, e.g., for protein analysis, to maintain their activity, to prevent denaturation, to couple them to carriers or payloads, or to inactivate RNAses.
For this purpose, DTT and BME are frequently used, however, they come with certain drawbacks: DTT and BME are easily oxidized by ambient air, they may react with heavy metal ions and interfere with metal ion affinity chromatography (IMAC), they are quenching thiol-reactive reagents like maleimides, and, in the case of BME, it is malodorous.
Discover TCEP as nearly odorless alternative!
Product details
LS-3405 TCEP*HCl
3-[bis(2-carboxyethyl)phosphanyl]propanoic acid, hydrochloride
CAS-No. 51805-45-9
Formula C9 H15 O 6 P*HCl
Mol. weight 250,19*36,45 g/mol
Reference:
→ A Comparison between the Sulfhydryl Reductants Tris(2-carboxyethyl)phosphine and Dithiothreitol for Use in Protein Biochemistry; E. B. Getz, M. Xiao, T. Chakrabarty, R. Cooke, P. R. Selvin; Anal. Biochem 1999; 273(1) : 73-80. arrow-up-right-from-square https://doi.org/10.1006/abio.1999.4203
2.2. Peptide Synthesis with Azido and Alkyne Amino Acids
Both Boc- and Fmoc-protected derivatives of azido and alkyne amino acids can be readily introduced into peptide sequences by standard SPPS protocols. Such building blocks have found widespread use in techniques such as peptide ligation, bioconjugation, labeling, immobilization and linkerology. Bioconjugation, which is defined as the joining of two biomolecules or the ligation of a synthetic molecule with a biomolecule, stands out in particular among those applications. Targets that are notoriously difficult to access such as glycopeptides and -proteins can be synthesized in a straightforward and chemoselective fashion via Click reaction to afford neoglycopeptides and -proteins. Peptides or proteins that aid in the translocation into cells or that facilitate the targeting to certain tissues or organelles may be conjugated to toxins, fluorophores, or oligonucleotides by means of the Click reaction.
Click Chemistry
Another potential application is the cyclization of peptides via Click chemistry. This technique is a wellknown approach to stabilize specific conformations in order to optimize peptide binding, and to increase resistance toward proteolytic degradation. If two clickable groups are placed at a suitable distance from each other in a peptide, they can undergo intramolecular cycloaddition with good yields and minimal side reactions. For example, this Click-mediated cyclization may be used to stabilize an α-helical secondary structure when azide and alkyne are located in side-chains at positions i and i+4, respectively.
Example for a Protocol for Click Reactions in Peptide Synthesis:
Successful protocols have been published applying to 3 μmol peptide in 4 mL tBuOH/H 2 O (1:2) with excess of ascorbic acid (40 μmol) and CuSO 4*5 H 2 O (40 μmol) generating Cu(I) in situ. Stirring at room temperature overnight is followed by appropriate chromatographic work up. [Le Chevalier-Isaad et al., Eur. J. Org. Chem. 2010]
References:
→ Synthesis and conformational analysis of a cyclic peptide obtained via i to i+4 intramolecular side-chain to side-chain azide-alkyne 1,3-dipolar cycloaddition; S. Cantel, C. Isaad Ale, M. Scrima, J. J. Levy, R. D. DiMarchi, P. Rovero, J. A. Halperin, A. M. D‘Ursi, A. M. Papini, M. Chorev; J Org Chem 2008; 73: 5663-74. arrow-up-right-from-square https://doi.org/10.1021/jo800142s
→ Side chain-to-side chain cyclization by click reaction; A. Le Chevalier Isaad, A. M. Papini, M. Chorev, P. Rovero; J. Pept. Sci. 2009; 15: 451-4. arrow-up-right-from-square https://doi.org/10.1002/psc.1141
→ CuI-Catalyzed Azide-Alkyne Intramolecular i-to-(i+4) Side-Chain-to-Side-Chain Cyclization Promotes the Formation of Helix-Like Secondary Structures; M. Scrima, A. Le Chevalier-Isaad, P. Rovero, A. M. Papini, M. Chorev, A. M. D‘Ursi; Eur. J. Org. Chem. 2010; 2010: 446-457. arrow-up-right-from-square https://doi.org/10.1002/ejoc.200901157
→ Improved synthesis and biological evaluation of chelator-modified alpha-MSH analogs prepared by copperfree click chemistry; N. J. Baumhover, M. E. Martin, S. G. Parameswarappa, K. C. Kloepping, M. S. O‘Dorisio, F. C. Pigge, M. K. Schultz; Bioorg Med Chem Lett 2011; 21: 5757-61. arrow-up-right-from-square https://doi.org/10.1016/j.bmcl.2011.08.017
→ „Click“-cyclized (68)Ga-labeled peptides for molecular imaging and therapy: synthesis and preliminary in vitro and in vivo evaluation in a melanoma model system; M. E. Martin, M. Sue O‘Dorisio, W. M. Leverich, K. C. Kloepping, S. A. Walsh, M. K. Schultz; Recent Results Cancer Res 2013; 194: 149-75. arrow-up-right-from-square https://doi.org/10.1007/978-3-642-27994-2_9
2.3. Azido Amino Acids and Related Derivatives
Azido-Alkyl/Aryl Acids and Alcohols
RL-4430 N3 -C7H14-COOH
8-azidooctanoic acid
CAS-No. 217180-76-2
Formula C 8 H15N3 O 2
Mol. weight 185,23 g/mol
Product details
BNN1370 N3 -EDA-Suc-OH
Azido-ethylenediamine-succinoyl-OH
CAS-No. 2225891-73-4
Formula C 6 H10 N4O 3
Mol. weight 186,17 g/mol
RL-4350 N3 -DAPr-Suc-OH
Azido-propylenediamine-succinoyl-OH
CAS-No. 929894-58-6
Formula C 7H12N4O 3 Mol. weight 200,20 g/mol
RL-4360 N3 -DABu-Suc-OH
Azido-butylenediamine-succinoyl-OH
CAS-No. 2226183-50-0
Formula C 8 H14N4O 3 Mol. weight 214,23 g/mol
HAA6990 N3 -Aca-Aca-OH
6-(6-azidohexanamido)hexanoic acid
CAS-No. 866363-71-5
Formula C12H22N4O 3 Mol. weight 270,33 g/mol
RL-3650 N3 -Phenylpropionic-OH
3-(4-azidophenyl)propanoic acid
CAS-No. 103489-31-2
Formula C9 H9 N3 O 2 Mol. weight 191,19 g/mol
AAA2190 DAPOA*DCHA
2-(1,3-diazidopropan-2-yloxy)acetic acid dicyclohexylamine
CAS-No. 2389064-43-9
Formula C 5H 8 N 6 O 3*C12H23 N Mol. weight 200,16*181,32 g/mol
Click Chemistry
HAA2230 N3 -1,4-cis-CHC-OH
cis-4-Azidocyclohexanecarboxylic acid
CAS-No. 863222-21-3
Formula C 7H11N3 O Mol. weight 169,18 g/mol
HAA2235 N3 -1,4-trans-CHC-OH
trans-4-Azidocyclohexanecarboxylic acid
CAS-No. 1931895-14-5
Formula C 7H11N3 O 2 Mol. weight 169,18 g/mol
HAA2240 N3 -trans-MCHC-OH
trans-4-(Azidomethyl)cyclohexanecarboxylic acid
CAS-No. 170811-10-6
Formula C 8 H13 N3 O 2 Mol. weight 183,21 g/mol
HAA3175 N3 -HABA*DCHA (2S)
(S)-4-azido-2-hydroxybutyric acid dicyclohexalamine
CAS-No. 959148-55-1
Formula C 4H7N3 O 3*C12H23 N Mol. weight 145,12*181,32 g/mol
HAA3365 N3 -IsoSer*DCHA (2S)
(S)-2-Hydroxy-3-azidopropanoic acid dicyclohexalamine
CAS-No. 1620171-65-4
Formula C 3 H 5N3 O 3*C12H23 N Mol. weight 131,09*181,32 g/mol
HAA2245 N3 -PhAc-OH
(4-Azidophenyl)acetic acid
CAS-No. 62893-37-2
Formula C 8 H7N3 O 2 Mol. weight 177,16 g/mol
AAA1970 N3 -Pen-OH
5-Azido-pentanoic acid
CAS-No. 79583-98-5
Formula C 5H9 N3 O 2
Mol. weight 143,14 g/mol
AAA1960 N3 -Hx-OH
6-Azido-hexanoic acid
CAS-No. 79598-53-1
Formula C 6 H11N3 O 2 Mol. weight 157,17 g/mol
RL-2980 N3 -Aca-OSu
6-Azidocaproic acid N-hydroxysuccinimidyl ester
CAS-No. 866363-70-4
Formula C10 H14N4O4 Mol. weight 254,24 g/mol
RL-3480 8-Azido-octanoyl-OSu
8-Azidodooctanoic acid N-hydroxysuccinimide ester
CAS-No. 2576471-56-0
Formula C12H18 N4O4 Mol. weight 282,30 g/mol

RL-3200 11-Azidoundecanoic acid
11-Azido-undecanoic acid
CAS-No. 118162-45-1
Formula C11H21N3 O 2 Mol. weight 227,30 g/mol

RL-3170 11-Azido-undecanoyl-OSu
11-Azidoundecanoic acid N-hydroxysuccinimide ester
CAS-No. 850080-13-6
Formula C15H24N4O4 Mol. weight 324,38 g/mol

Click Chemistry
RL-3210 12-Azidododecanoic acid
12-Azido-dodecanoic acid
CAS-No. 80667-36-3
Formula C12H23 N3 O 2 Mol. weight 241,33 g/mol

RL-3220 12-Azido-dodecanoyl-OSu
12-Azidododecanoic acid N-hydroxysuccinimide ester
CAS-No. 2489524-00-5
Formula C16 H26 N4O4 Mol. weight 338,40 g/mol

RL-3230 14-Azido-myristic acid
14-azidotetradecanoic acid
CAS-No. 176108-61-5
Formula C14H27N3 O 2 Mol. weight 269,38 g/mol

RL-3240 16-Azido-palmitic acid
16-azidohexadecanoic acid
CAS-No. 112668-54-9
Formula C16 H31N3 O 2 Mol. weight 297,44 g/mol

RL-3250 18-Azido-stearic acid
18-azidooctadecanoic acid
CAS-No. 1529763-58-3
Formula C18 H35N3 O 2 Mol. weight 325,49 g/mol

RL-2995 4-(Azidomethyl)benzoic acid
4-Azidomethylbenzoic acid
CAS-No. 79584-03-5
Formula C 8 H7N3 O 2 Mol. weight 177,16 g/mol

RL-2035 ATFB
4-Azido-2,3,5,6-tetrafluorobenzoic acid
CAS-No. 122590-77-6
Formula C 7HF4N3 O 2 Mol. weight 235,1 g/mol

RL-2045 ATFB-NHS
N-Succinimidyl 4-azido-2,3,5,6-tetrafluorobenzoate
CAS-No. 126695-58-7
Formula C11H4F4N4O4 Mol. weight 332,17 g/mol

PEG5000 ATFB-O2Oc
{2-[2-(4-Azido-2,3,5,6-tetrafluorobenzoyl-amino)ethoxy] ethoxy}acetic acid
CAS-No. 1993119-45-1
Formula C13 H12F4N4O 5 Mol. weight 380,25 g/mol
RL-2990 4-Azidobenzyl alcohol
(4-azidophenyl)methanol
CAS-No. 31499-54-4
Formula C 7H7N3 O Mol. weight 149,15 g/mol
RL-4070 2-Azidobenzyl alcohol
(2-azidophenyl)methanol
CAS-No. 20615-76-3
Formula C 7H7N3 O Mol. weight 149,15 g/mol

Click Chemistry
Azido-Alanine and Propionic Acid Derivatives
HAA1880 H-L-Aza-OH*HCl hydrate
(S)-2-Amino-3-azidopropanoic acid hydrochloride hydrate
CAS-No. 1620171-64-3
Formula C 3 H 6 N4O 2*HCl*nH2O Mol. weight 130,11*36,45 g/mol
HAA1885 H-D-Aza-OH*HCl hydrate
(R)-2-Amino-3-azidopropanoic acid hydrochloride hydrate
CAS-No. 1379690-01-3
Formula C 3 H 6 N4O 2*HCl*nH2O Mol. weight 130,11*36,45 g/mol
BAA1820 Boc-L-Aza-OH*CHA
(S)-2-t-Butyloxycarbonylamino-3-azidopropanoic acid cyclohexylamine
CAS-No. 2098496-88-7
Formula C 8 H14N4O4*C 6 H13 N Mol. weight 230,22*99,18 g/mol
BAA1825 Boc-D-Aza-OH*CHA
(R)-2-t-Butyloxycarbonylamino-3-azidopropanoic acid cyclohexylamine
CAS-No. 2098496-96-7
Formula C 8 H14N4O4*C 6 H13 N Mol. weight 230,22*99,18 g/mol
FAA1820 Fmoc-L-Aza-OH (solv.)
(S)-2-(9-Fluorenylmethyloxycarbonylamino)-3-azidopropanoic acid, solvate with DIPE
CAS-No. 684270-46-0
Formula C18 H16 N4O4 Mol. weight 352,34 g/mol
Product details





FAA6870 Fmoc-D-Aza-OH
(R)-2-(9-Fluorenylmethyloxycarbonylamino)-3-azidopropanoic acid
CAS-No. 1016163-79-3
Formula C18 H16 N4O4 Mol. weight 352,34 g/mol
FAA9420 Fmoc-L-MeDap(N3)-OH
(S)-2-((((9H-fluoren-9-yl)methoxy)carbonyl)(methyl) amino)-3-azidopropanoic acid
CAS-No. 1263721-08-9
Formula C19 H18 N4O4 Mol. weight 366,38 g/mol
HAA2130 N3 -L-Dap(Boc)-OH
(S)-2-Azido-3-((t-butyloxycarbonyl)amino)propanoic acid
CAS-No. 1932432-15-9
Formula C 8 H14N4O4 Mol. weight 230,22 g/mol
HAA2135 N3 -D-Dap(Boc)-OH
(R)-2-Azido-3-((t-butyloxycarbonyl)amino)propanoic acid
CAS-No. 1630044-08-4
Formula C 8 H14N4O4 Mol. weight 230,22 g/mol
HAA2140 N3 -L-Dap(Fmoc)-OH
(S)-2-Azido-3-[(9-fluorenylmethyloxycarbonyl)amino] propanoic acid
CAS-No. 880637-82-1
Formula C18 H16 N4O4 Mol. weight 352,34 g/mol
HAA2145 N3 -D-Dap(Fmoc)-OH
(R)-2-Azido-3-[(9-fluorenylmethyloxycarbonyl)amino] propanoic acid
CAS-No. 1807631-13-5
Formula C18 H16 N4O4 Mol. weight 352,34 g/mol




References:
→ Azidoalanine mutagenicity in Salmonella: effect of homologation and alpha-methyl substitution; J. B. Mangold, M. R. Mischke, J. M. LaVelle; Mutat Res 1989; 216: 27-33. arrow-up-right-from-square https://doi.org/10.1016/0165-1161(89)90020-4
→ „Click to chelate“: synthesis and installation of metal chelates into biomolecules in a single step; T. L. Mindt, H. Struthers, L. Brans, T. Anguelov, C. Schweinsberg, V. Maes, D. Tourwe, R. Schibli; J Am Chem Soc 2006; 128: 15096-7. arrow-up-right-from-square https://doi.org/10.1021/ja066779f
→ Design, synthesis, and biological activity of novel triazole amino acids used to probe binding interactions between ligand and neutral amino acid transport protein SN1; M. Gajewski, B. Seaver, C. S. Esslinger; Bioorg Med Chem Lett 2007; 17: 4163-6. arrow-up-right-from-square https://doi.org/10.1016/j.bmcl.2007.05.061
→ Peptide tertiary structure nucleation by side-chain crosslinking with metal complexation and double „click“ cycloaddition; O. Torres, D. Yuksel, M. Bernardina, K. Kumar, D. Bong; Chembiochem 2008; 9: 1701-5. arrow-up-right-from-square https://doi.org/10.1002/cbic.200800040
→ Maintaining biological activity by using triazoles as disulfide bond mimetics; K. Holland-Nell, M. Meldal; Angew. Chem. Int. Ed. 2011; 50: 5204-6. arrow-up-right-from-square https://doi.org/10.1002/anie.201005846
Azido-Homoalanine and Azido-Butanoic Acid Derivatives
HAA5730 H-L-Aha-OH*HCl
(S)-2-Amino-4-azidobutanoic acid hydrochloride
CAS-No. 942518-29-8
Formula C 4H 8 N4O 2*HCl Mol. weight 144,13*36,45 g/mol
Product details

HAA9280 H-L-Aha-OH
4-Azido-L-homoalanine
CAS-No. 120042-14-0
Formula C 4H 8 N4O 2 Mol. weight 144,13 g/mol

HAA1630 H-D-Aha-OH*HCl
(R)-2-Amino-4-azidobutanoic acid hydrochloride
CAS-No. 1858224-26-6
Formula C 4H 8 N4O 2*HCl Mol. weight 144,13*36,45 g/mol

BAA1800 Boc-L-Aha-OH*CHA
(S)-2-t-Butyloxycarbonylamino-4-azidobutanoic acid cyclohexylamine
CAS-No. 120042-08-2
Formula C9 H16 N4O4*C 6 H13 N Mol. weight 244,25*99,18 g/mol
BAA1805 Boc-D-Aha-OH*CHA
(R)-2-t-Butyloxycarbonylamino-4-azidobutanoic acid cyclohexylamine
CAS-No. 1609202-75-6 net
Formula C9 H16 N4O4*C 6 H13 N Mol. weight 244,25*99,18 g/mol
FAA6620 Fmoc-L-Aha-OH
(S)-2-(9-Fluorenylmethyloxycarbonylamino)-4-azidobutanoic acid
CAS-No. 942518-20-9
Formula C19 H18 N4O4 Mol. weight 366,41 g/mol
FAA6810 Fmoc-D-Aha-OH
(R)-2-(9-Fluorenylmethyloxycarbonylamino)-4-azidobutanoic acid
CAS-No. 1263047-53-5
Formula C19 H18 N4O4 Mol. weight 366,41 g/mol
ZAA5700 Z-L-Aha-OH*DCHA
(S)-2-Benzyloxycarbonylamino-4-azidobutanoic acid dicyclohexylamine
CAS-No. 1263047-43-3 net
Formula C12H14N4O4*C12H23 N Mol. weight 278,26*181,34 g/mol
HAA2150 N3 -L-Dab(Boc)-OH
(S)-2-Azido-4-((t-butyloxycarbonyl)amino)butanoic acid
CAS-No. 1932403-71-8
Formula C9 H16 N4O4 Mol. weight 244,25 g/mol






Click Chemistry
HAA2155 N3 -D-Dab(Boc)-OH
(R)-2-Azido-4-((t-butyloxycarbonyl)amino)butanoic acid
CAS-No. 1922891-74-4
Formula C9 H16 N4O4 Mol. weight 244,25 g/mol
HAA3170 N3 -L-Dab(Fmoc)-OH
(S)-2-Azido-4-[(9-fluorenylmethyloxycarbonyl)amino] butanoic acid
CAS-No. 2250436-44-1
Formula C9 H16 N4O4 Mol. weight 366,37 g/mol


References:
→ EP2190856 (A2), US2008096819 (A1), WO2009026393 (A2),WO2009026393 (A3), WO2006079364
→ Presentation and detection of azide functionality in bacterial cell surface proteins; A. J. Link, M. K. Vink, D. A. Tirrell; J Am Chem Soc 2004; 126: 10598-602. arrow-up-right-from-square https://doi.org/10.1021/ja047629c
→ Mild and chemoselective peptide-bond cleavage of peptides and proteins at azido homoalanine; J. W. Back, O. David, G. Kramer, G. Masson, P. T. Kasper, L. J. de Koning, L. de Jong, J. H. van Maarseveen, C. G. de Koster; Angew. Chem. Int. Ed. 2005; 44: 7946-50. arrow-up-right-from-square https://doi.org/10.1002/anie.200502431
→ Expanding the diversity of chemical protein modification allows post-translational mimicry; S. I. van Kasteren, H. B. Kramer, H. H. Jensen, S. J. Campbell, J. Kirkpatrick, N. J. Oldham, D. C. Anthony, B. G. Davis; Nature 2007; 446: 1105-9. arrow-up-right-from-square https://doi.org/10.1038/nature05757
→ Site-specific modification of Candida antarctica lipase B via residue-specific incorporation of a non-canonical amino acid; S. Schoffelen, M. H. Lambermon, M. B. van Eldijk, J. C. van Hest; Bioconjug Chem 2008; 19: 1127-31. arrow-up-right-from-square https://doi.org/10.1021/bc800019v
→ Unnatural amino acid incorporation into virus-like particles; E. Strable, D. E. Prasuhn, Jr., A. K. Udit, S. Brown, A. J. Link, J. T. Ngo, G. Lander, J. Quispe, C. S. Potter, B. Carragher, D. A. Tirrell, M. G. Finn; Bioconjug Chem 2008; 19: 866-75. arrow-up-right-from-square https://doi.org/10.1021/bc700390r
→ Selective enrichment of azide-containing peptides from complex mixtures; M. A. Nessen, G. Kramer, J. Back, J. M. Baskin, L. E. Smeenk, L. J. de Koning, J. H. van Maarseveen, L. de Jong, C. R. Bertozzi, H. Hiemstra, C. G. de Koster; J Proteome Res 2009; 8: 3702-11. arrow-up-right-from-square https://doi.org/10.1021/pr900257z → „Clickable“ elastins: elastin-like polypeptides functionalized with azide or alkyne groups; R. L. Teeuwen, S. S. van Berkel, T. H. van Dulmen, S. Schoffelen, S. A. Meeuwissen, H. Zuilhof, F. A. de Wolf, J. C. van Hest; Chem Commun (Camb) 2009; 4022-4. arrow-up-right-from-square https://doi.org/10.1039/b903903a
Azido-beta-Homoalanine
HAA3970 H-L-Dbu(N3)-OH*HCl
(S)-3-Amino-4-azidobutanoic acid hydrochloride
CAS-No. 2389078-78-6 net
Formula C 4H 8 N4O 2*HCl Mol. weight 144,13*36,45 g/mol
Product

FAA2035 Fmoc-L-Dbu(N3)-OH
(S)-3-(9-Fluorenylmethyloxycarbonyl)amino-4-azido-butanoic acid
CAS-No. 934502-72-4
Formula C19 H18 N4O4 Mol. weight 366,37 g/mol
FAA3650 Fmoc-D-Dbu(N3)-OH
(R)-3-(9-Fluorenylmethyloxycarbonyl)amino-4-azido-butanoic acid
CAS-No. 1932023-47-6
Formula C19 H18 N4O4 Mol. weight 366,37 g/mol
ZAA1290 Z-L-Dbu(N3)-OH
(S)-3-(Benzyloxycarbonyl)amino-4-azido-butanoic acid
CAS-No. 1932657-23-2
Formula C12H14N4O4 Mol. weight 278,26 g/mol



ZAA1285 Z-D-Dbu(N3)-OH
CAS-No. 1931958-82-5
(R)-3-(Benzyloxycarbonyl)amino-4-azido-butanoic acid (R)
Formula C12H14N4O4 Mol. weight 278,26 g/mol

2-Amino-3-Azido-Butanoic Acid
HAA3010 H-Abu(3-N3)-OH*HCl (2S,3S)
(2S,3S)-2-amino-3-azidobutanoic acid hydrochloride
CAS-No. 2737202-68-3
Formula C 4H 8 N4O 2*HCl Mol. weight 144,13*36,45 g/mol
Product details

HAA3020 H-Abu(3-N3)-OH*HCl (2S,3R)
(2S,3S)-2-amino-3-azidobutanoic acid hydrochloride
CAS-No. 2737202-63-8
Formula C 4H 8 N4O 2*HCl Mol. weight 144,13*36,45 g/mol

FAA2040 Fmoc-Abu(3-N3)-OH (2S,3S)
(2S,3S)-2-(9-Fluorenylmethyloxycarbonyl)amino-3-azido-butanoic acid
CAS-No. 131669-42-6
Formula C19 H18 N4O4 Mol. weight 366,37 g/mol
FAA3200 Fmoc-Abu(3-N3)-OH (2S,3R)
(2S,3R)-2-(9-Fluorenylmethyloxycarbonyl)amino-3-azido-butanoic acid
CAS-No. 146306-79-8
Formula C19 H18 N4O4 Mol. weight 366,37 g/mol
FAA3540 Fmoc-Abu(3-N3)-OH (2R,3S)
(2R,3S)-2-(9-Fluorenylmethyloxycarbonyl)amino-3-azido-butanoic acid
CAS-No. 1932349-21-7
Formula C19 H18 N4O4 Mol. weight 366,37 g/mol



FAA2095 Fmoc-Abu(3-N3)-OH (2R,3R)
(2R,3R)-2-(9-Fluorenylmethyloxycarbonyl)amino-3-azidobutanoic acid
CAS-No. 1229394-75-5
Formula C19 H18 N4O4
Mol. weight 366,37 g/mol

Interested in Click Chemistry and automated synthesis?
Watch the recording of our workshop!
Azido-Masked Amino Function
Azido groups located in amino acid side-chains can be used for various applications. 2-Amino-3-azidobutanoic acid is shown as an example below.
Staudinger conjugate
Fig. 11: Possible applications of amino acids bearing azide groups in the side-chain.
Click Chemistry
A) Azido groups can be used for any type of Click conjugation with any available alkynyl residue forming conjugates with peptides or any other organic molecule.
B) Azido groups may also be used for another prominent type of bioconjugation, namely the Staudinger ligation, which is a further development of the Staudinger reaction. The Staudinger ligation is characterized by high selectivity and a typically rapid and high-yielding turnover. As a biorthogonal reaction, it has been used for the semisynthesis of proteins, for installing posttranslational modifications such as glycosylations, and for DNA labeling.
C) The azido group can be reduced to an amino function and hereby serve as a masked amino group. Prominent methods for the reduction of azido groups include the Staudinger reaction as well as the reduction by DTT. Azido groups are stable towards treatment with piperidine (Fmoc deprotection), Pd(0) (Alloc removal) and acidic treatment (cleavage of Mtt, Trt or other acid-sensitive groups). However, as it is a pseudohalogenide, care must be taken during coupling steps, as HATU will cause a high degree of racemization. This can be avoided using collidine or other non-nucleophilic bases instead of DIPEA.
Chiral α,ꞵ-diamines and diamino acids have increasingly become motifs of interest in organic synthesis owing to their ubiquity in natural products and medicinal agents. For example, these motifs are found in biotin, penicillins, and the antiinfluenza neuraminidase inhibitor Tamiflu. Chiral vicinal diamines and their metal complexes have been employed in stereoselective organic synthesis, in particular as chiral auxiliaries and ligands in catalytic asymmetric synthesis.
Azido-Arginine
BAA6515
Boc-L-Arg(3-azidopropyl,Pbf)-OH
(E)-Nw-(3-azidopropyl)-N2-(tert-butoxycarbonyl)-Nw‘-((2,2,4,6,7-pentamethyl-2,3-dihydrobenzofuran-5-yl)sulfonyl)-L-arginine
Formula C 27H43 N7 O 7S Mol. weight 609,74 g/mol
FAA9730
Fmoc-L-Arg(3-azidopropyl,Pbf)-OH
(E)-N2-(((9H-fluoren-9-yl)methoxy)carbonyl)-Nw-(3-azidopropyl)-Nw‘-((2,2,4,6,7-pentamethyl-2,3-dihydrobenzofuran-5-yl)sulfonyl)-L-arginine
CAS-No. 1060769-51-8
Formula C 37H45N7 O 7S Mol. weight 731,87 g/mol
Product details
Azido-Asparagine
FAA8605 Fmoc-L-Asn(EDA-N3)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-N-gamma-(2-azidoethyl)-L-asparagine
CAS-No. 2616562-74-2
Formula C 21H21N 5 O 5 Mol. weight 423,43
Azido Aspartic Acid
FAA9665 Fmoc-L-Asp(4-N3 -OBzl)-OH
(S)-2-((((9H-fluoren-9-yl)methoxy)carbonyl) amino)-4-((4-azidobenzyl)oxy)-4-oxobutanoic acid
Formula C 26 H22N4O 6 Mol. weight 486,48 g/mol
Product details
Product details
References:
→ Über neue organische Phosphorverbindungen III. Phosphinmethylenderivate und Phosphinimine; H. Staudinger, J. Meyer; Helv. Chim. Acta 1919; 2: 635-646. arrow-up-right-from-square https://doi.org/10.1002/hlca.19190020164
→ Reduction of aryl azides by thiols: Implications for the use of photoaffinity reagents; J. V. Staros, H. Bayley, D. N. Standring, J. R. Knowles; Biochem. Biophys. Res. Commun. 1978; 80: 568-572. arrow-up-right-from-square https://doi.org/10.1016/0006-291X(78)91606-6
→ Sixty years of staudinger reaction; Y. G. Gololobov, I. N. Zhmurova, L. F. Kasukhin; Tetrahedron 1981; 37: 437-472. arrow-up-right-from-square https://doi.org/10.1016/s0040-4020(01)92417-2
→ Staudinger ligation: a peptide from a thioester and azide; B. L. Nilsson, L. L. Kiessling, R. T. Raines; Org Lett 2000; 2: 1939-41. arrow-up-right-from-square https://doi.org/10.1021/ol0060174
→ A „traceless“ Staudinger ligation for the chemoselective synthesis of amide bonds; E. Saxon, J. I. Armstrong, C. R. Bertozzi; Org Lett 2000; 2: 2141-3. arrow-up-right-from-square https://doi.org/10.1021/ol006054v
→ Alpha,beta-diamino acids: biological significance and synthetic approaches; A. Viso, R. Fernandez de la Pradilla, A. Garcia, A. Flores; Chem Rev 2005; 105: 3167-96. arrow-up-right-from-square https://doi.org/10.1021/cr0406561
→ Chemoselective peptide cyclization by traceless Staudinger ligation; R. Kleineweischede, C. P. Hackenberger; Angew. Chem. Int. Ed. 2008; 47: 5984-8. arrow-up-right-from-square https://doi.org/10.1002/anie.200801514
→ Protein engineering with the traceless Staudinger ligation; A. Tam, R. T. Raines; Methods Enzymol. 2009; 462: 25-44. arrow-up-right-from-square https://doi.org/10.1016/S0076-6879(09)62002-4
→ Bioconjugation via azide-Staudinger ligation: an overview; C. I. Schilling, N. Jung, M. Biskup, U. Schepers, S. Brase; Chem Soc Rev 2011; 40: 4840-71. arrow-up-right-from-square https://doi.org/10.1039/c0cs00123f
→ Update 1 of: alpha,beta-Diamino acids: biological significance and synthetic approaches; A. Viso, R. Fernandez de la Pradilla, M. Tortosa, A. Garcia, A. Flores; Chem Rev 2011; 111: PR1-42. arrow-up-right-from-square https://doi.org/10.1021/cr100127y
→ The Staudinger Ligation; C. Bednarek, I. Wehl, N. Jung, U. Schepers, S. Bräse; Chem Rev 2020; 120: 4301-4354. arrow-up-right-from-square https://doi.org/10.1021/acs.chemrev.9b00665 arrow-up back to content
Click Chemistry
→ Site-selective traceless Staudinger ligation for glycoprotein synthesis reveals scope and limitations; G. J. Bernardes, L. Linderoth, K. J. Doores, O. Boutureira, B. G. Davis; Chembiochem 2011; 12: 1383-6. arrow-up-right-from-square https://doi.org/10.1002/cbic.201100125
Azido-Citrulline
HAA4980 N3 -L-Cit-OH*DCHA
(S)-2-Azido-citrulline dicyclohexylamine
CAS-No. 1799421-66-1 net
Formula C 6 H11N 5 O 3*C12H23 N Mol. weight 201,18*181,32 g/mol
Product details

Azido-Cysteine
HAA2810 N3 -L-Cys(Trt)-OH*CHA
(R)-2-azido-3-(tritylthio)propanoic acid cyclohexylamine
CAS-No. 1286670-90-3
Formula C 22H19 N3 O 2 S*C 6 H13 N Mol. weight 389,47*99,17 g/mol
FAA9460 Fmoc-L-Cys(Azidoethyl)-OH
N-(((9H-fluoren-9-yl)methoxy)carbonyl)-S-(2-azidoethyl)-L-cysteine
CAS-No. 3029657-80-2
Formula C 20 H20 N4O4 S Mol. weight 412,46 g/mol
Product details

Azido-Glutamic Acid
FAA9675 Fmoc-L-Glu(4-N3 -OBzl)-OH
(S)-2-((((9H-fluoren-9-yl)methoxy)carbonyl) amino)-5-((4-azidobenzyl)oxy)-5-oxopentanoic acid
Formula C 27H24N4O 6 Mol. weight 500,51 g/mol
Azido-Glycine
BAA4895 Boc-Aeg(N3)-OK
N-(2-azidoethyl)-N-(tert-butoxycarbonyl)glycine potassium salt
CAS-No. 2172548-14-8 net
Formula C9 H15KN4O4 Mol. weight 282,34 g/mol
FAA4060 Fmoc-Aeg(N3)-OH
N-(9-Fluorenylmethyloxycarbonyl)-N-(2-azidoethyl) glycine
CAS-No. 1935981-35-3
Formula C19 H18 N4O4 Mol. weight 366,37 g/mol
FAA4055 Fmoc-Abg(N3)-OH
N-(9-Fluorenylmethyloxycarbonyl)-N-(4-azidobutyl) glycine
CAS-No. 2250433-81-7
Formula C 21H22N4O4 Mol. weight 394,42 g/mol
HAA2850 N3 -Gly-Gly-OH*DCHA
Azido-glycylglycine dicyclohexylamine
CAS-No. 855750-87-7
Formula C 4H 6 N4O 3*C12H23 N Mol. weight 158,12*181,32 g/mol
Product details
Product details



HAA2840 N3 -Gly-Gly-Gly-OH
Azido-glycylglycylglycine
CAS-No. 1993176-75-2
Formula C 6 H9 N 5 O4 Mol. weight 215,17 g/mol

HAA2860 N3 -Gly-Gly-Gly-Gly-Gly-OH
CAS-No. 2250433-77-1
Formula C10 H15N7 O 6 Mol. weight 329,27 g/mol

HAA9330 N3 -Gly-Aeg(Fmoc)-OH
N-(2-((((9H-fluoren-9-yl)methoxy)carbonyl)amino) ethyl)-N-(2-azidoacetyl)glycine
CAS-No. 2606227-07-8
Formula C 21H21N 5 O 5 Mol. weight 423,43 g/mol
FAA9835 Fmoc-Ahg(N3)-OH
N-(((9H-fluoren-9-yl)methoxy)carbonyl)-N-(6-azidohexyl)glycine
Formula C 23 H26 N4O4 Mol. weight 422,49 g/mol
Azido-Leucine
HAA3350 N3 -L-Leu-OH*BHA
(S)-2-azido-4-methylpentanoic acid benzhydrylamine salt
CAS-No. 79410-33-6
Formula C 6 H11N3 O 2*C13 H13 N Mol. weight 157,17*183,25 g/mol
Product details

HAA2820 N3 -L-Leu-OH*CHA
(S)-2-Azido-4-methylpentanoic acid cyclohexylamine
CAS-No. 1286670-79-8
Formula C 6 H11N3 O 2*C 6 H13 N Mol. weight 157,17*99,18 g/mol

Azido-Lysine
HAA9210 H-L-Lys(N3)-OH
N-epsilon-azido-L-lysine
CAS-No. 159610-92-1
Formula C 6 H12N4O 2 Mol. weight 172,19 g/mol

HAA1625 H-L-Lys(N3)-OH*HCl
N-epsilon-Azido-L-lysine hydrochloride
CAS-No. 1454334-76-9
Formula C 6 H12N4O 2*HCI Mol. weight 172,19*36,45 g/mol

HAA9340 H-L-Lys(N3 -Gly)-OH*HCl
Azidoacetyl-L-Lysine hydrochloride
CAS-No. 1198617-82-1 net
Formula C 8 H15N 5 O 3 Mol. weight 229,24 g/mol
FAA8855 Fmoc-L-Lys(N3 -Gly)-OH
Azidoacetyl-Fmoc-L-Lysine
CAS-No. 1198617-89-8
Formula C 23 H25N 5 O 5 Mol. weight 451,48 g/mol
Click Chemistry
HAA1890 H-D-Lys(N3)-OH*HCl
N-epsilon-Azido-D-lysine hydrochloride
CAS-No. 2098497-01-7
Formula C 6 H12N4O 2*HCl
Mol. weight 172,19*36,45 g/mol

HAA2080 H-L-Lys(EO-N3)-OH*HCl
(S)-2-amino-6-((2-azidoethoxy)carbonylamino)hexanoic acid hydrochloride
CAS-No. 1994331-17-7
Formula C9 H17N 5 O4*HCl Mol. weight 259,26*36,46 g/mol
HAA9295 H-L-Lys(COCF2 N3)-OH*HCl
N6-(2-azido-2,2-difluoroacetyl)-L-lysine
Formula C 8 H13 F 2N 5 O 3*HCl
Mol. weight 265,22*36,46 g/mol


HAA2870 H-(Gly)3 -Lys(N3)-OH*HCl
Triglycyl-epsilon-azido-L-lysine hydrochloride
CAS-No. 2250437-45-5
Formula C12H21N7 O 5*HCl Mol. weight 343,34*36,45 g/mol

BAA4900 Boc-L-Lys(N3)-OK
Boc-azidolysine potassium salt
CAS-No. 846549-33-5
Formula C11H19 KN4O4 Mol. weight 310,40 g/mol
BAA1810 Boc-L-Lys(N3)-OH*CHA
N-alpha-t-Butyloxycarbonyl-epsilon-azido-L-lysine cyclohexylamine
CAS-No. 2098497-30-2
Formula C11H20 N4O4*C 6 H13 N Mol. weight 272,30*99,18 g/mol
BAA1815 Boc-D-Lys(N3)-OH*CHA
N-alpha-t-Butyloxycarbonyl-epsilon-azido-D-lysine cyclohexylamine
CAS-No. 1858224-39-1
Formula C11H20 N4O4*C 6 H13 N Mol. weight 272,30*99,18 g/mol
FAA1793 Fmoc-L-Lys(N3)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-epsilon-azido-L-lysine
CAS-No. 159610-89-6
Formula C 21H22N4O4 Mol. weight 394,42 g/mol
FAA1835 Fmoc-D-Lys(N3)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-epsilon-azido-D-lysine
CAS-No. 1198791-53-5
Formula C 21H22N4O4 Mol. weight 394,42 g/mol
FAA9740 Fmoc-L-HLys(N3)-OH
(S)-2-((((9H-fluoren-9-yl)methoxy)carbonyl) amino)-7-azidoheptanoic acid
Formula C 22H24N4O4 Mol. weight 408,46 g/mol
FAA7925 Fmoc-L-Lys(N3 -AEEA)-OH
N2-(((9H-fluoren-9-yl)methoxy)carbonyl)-N6-(2-(2-(2-azidoethoxy)ethoxy)acetyl)-L-lysine
CAS-No. 1236293-83-6
Formula C 27H33 N 5 O 7 Mol. weight 539,59 g/mol
FAA8145 Fmoc-L-Lys(N3 -Aca-DIM)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-N-epsilon-[6-azido-1-(4,4-dimethyl-2,6- dioxocyclohexylidene)hexyl]-L-lysine
CAS-No. 2408993-39-3
Formula C 35H43 N 5 O 6 Mol. weight 629,76 g/mol



FAA8595 Fmoc-L-MeLys(N3)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-N-alpha-methyl-epsilon-azido-L-lysine
CAS-No. 1263721-14-7
Formula C 22H24N4O4
Mol. weight 408,46 g/mol
FAA9615 Fmoc-D-MeLys(N3)-OH
N2-(((9H-fluoren-9-yl)methoxy)carbonyl)-N6-diazo-N2-methyl-D-lysine
Formula C 22H24N4O4 Mol. weight 405,50 g/mol
HAA2170 N3 -L-Lys(Boc)-OH
(S)-2-Azido-6-[(t-butyloxycarbonyl)amino]hexanoic acid
CAS-No. 333366-32-8
Formula C11H20 N4O4 Mol. weight 272,3 g/mol


HAA2175 N3 -D-Lys(Boc)-OH
(R)-2-Azido-6-[(t-butyloxycarbonyl)amino]hexanoic acid
CAS-No. 1178899-92-7
Formula C11H20 N4O4 Mol. weight 272,3 g/mol

HAA2160 N3 -L-Lys(Fmoc)-OH
(S)-2-Azido-6-[(9-fluorenylmethyloxycarbonyl)amino] hexanoic acid
CAS-No. 473430-12-5
Formula C 21H22N4O4 Mol. weight 394,42 g/mol
HAA2165 N3 -D-Lys(Fmoc)-OH
(R)-2-Azido-6-[(9-fluorenylmethyloxycarbonyl)amino] hexanoic acid
CAS-No. 1994300-35-4
Formula C 21H22N4O4 Mol. weight 394,42 g/mol


HAA2880 N3 -L-Lys(Mtt)-OH
(S)-2-Azido-6-[(4-methyltrityl)amino]hexanoic acid
CAS-No. 1333231-26-7
Formula C 26 H28 N4O 2 Mol. weight 428,53 g/mol

FAA8825 Fmoc-L-Lys(COCF2N3)-OH
N2-(((9H-fluoren-9-yl)methoxy)carbonyl)-N6-(2-azido-2,2-difluoroacetyl)-L-lysine
Formula C 23 H23 F 2N 5 O 5 Mol. weight 487,46 g/mol

FAA8830 Fmoc-L-Lys(4-N3 -Z)-OH
N2-(((9H-fluoren-9-yl)methoxy)carbonyl)-N6-(((4-azidobenzyl)oxy)carbonyl)-L-lysine
CAS-No. 1446511-14-3
Formula C 29 H29 N 5 O 6 Mol. weight 543,58 g/mol
HAA9315 H-L-Lys(4-N3 -Z)-OH*HCl
(2S)-6-(4-Azido-benzyloxycarbonylamino)-2-amino-hexanoic acid hydrochloride
CAS-No. 2084913-49-3
Formula C14H19 N 5 O4*HCl Mol. weight 321,34*36,46 g/mol
HAA9380 H-L-Lys(2-N3 -Z)-OH
N6-(((2-azidobenzyl)oxy)carbonyl)-L-lysine
CAS-No. 1131963-69-3
Formula C14H19 N 5 O4
Mol. weight 321,34 g/mol
HAA9370 H-L-Lys(3-N3 -Z)-OH*HCl
N6-(((3-azidobenzyl)oxy)carbonyl)-L-lysine
CAS-No. 2084913-47-1
Formula C14H19 N 5 O4*HCl Mol. weight 321,34*36,45 g/mol


Click Chemistry
Product details
FAA8880 Fmoc-L-Lys(2-N3 -Z)-OH
(2S)-6-(2-Azido-benzyloxycarbonylamino)-2-(9H-fluoren-9-ylmethoxycarbonylamino)-hexanoic acid
CAS-No. 2714331-96-9
Formula C 29 H29 N 5 O 6 Mol. weight 543,58 g/mol
FAA8890 Fmoc-L-Lys(3-N3 -Z)-OH
(2S)-6-(3-Azido-benzyloxycarbonylamino)-2-(9H-fluoren-9-ylmethoxycarbonylamino)-hexanoic acid
CAS-No. 1836202-27-7
Formula C 29 H29 N 5 O 6 Mol. weight 543,58 g/mol
Azido-Ornithine
HAA1620 H-L-Orn(N3)-OH*HCl
N-delta-Azido-L-ornithine hydrochloride
CAS-No. 1782935-10-7
Formula C 5H10 N4O 2*HCI Mol. weight 158,16*36,45 g/mol
Product details

HAA1895 H-D-Orn(N3)-OH*HCl
N-delta-Azido-D-ornithine hydrochloride
CAS-No. 1858224-08-4
Formula C 5H10 N4O 2*HCI Mol. weight 158,16*36,45 g/mol

BAA1830 Boc-L-Orn(N3)-OH*CHA
N-alpha-t-Butyloxycarbonyl-delta-azido-L-ornithine cyclohexylamine
CAS-No. 2098497-17-5
Formula C10 H18 N4O4*C 6 H13 N Mol. weight 258,27*99,18 g/mol

BAA1835 Boc-D-Orn(N3)-OH*CHA
N-alpha-t-Butyloxycarbonyl-delta-azido-D-ornithine cyclohexylamine
CAS-No. 1858224-18-6
Formula C10 H18 N4O4*C 6 H13 N Mol. weight 258,27*99,18 g/mol
FAA6880 Fmoc-L-Orn(N3)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-delta-azido-L-ornithine
CAS-No. 1097192-04-5
Formula C 20 H20 N4O4 Mol. weight 380,4 g/mol
FAA6890 Fmoc-D-Orn(N3)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-delta-azido-D-ornithine
CAS-No. 1176270-25-9
Formula C 20 H20 N4O4 Mol. weight 380,4 g/mol
FAA8680 Fmoc-L-MeOrn(N3)-OH
(S)-2-((((9H-fluoren-9-yl)methoxy)carbonyl)(methyl) amino)-5-azidopentanoic acid
CAS-No. 2991255-09-3
Formula C 21H22N4O4 Mol. weight 394,43 g/mol
HAA2210 N3 -D-Orn(Boc)-OH*CHA
(R)-2-Azido-5-[(t-butyloxycarbonyl)amino]pentanoic acid cyclohexylamine
CAS-No. 2165877-62-1
Formula C10 H18 N4O4*C 6 H13 N Mol. weight 258,27*99,18 g/mol
HAA9485 N3 -L-Orn(Boc)-ONa
sodium (S)-2-azido-5-((tert-butoxycarbonyl)amino) pentanoate
CAS-No. 1639198-67-6
Formula C10 H17N4NaO4 Mol. weight 280,26 g/mol




HAA9495 N3 -L-Orn(Boc)-OK
potassium (S)-2-azido-5-((tert-butoxycarbonyl)amino) pentanoate
CAS-No. 1639198-67-6 net
Formula C10 H17N4KO4
Mol. weight 296,37 g/mol
HAA2220 N3 -L-Orn(Boc)-OH*CHA
(S)-2-Azido-5-[(t-butyloxycarbonyl)amino]pentanoic acid cyclohexylamine
CAS-No. 2301169-18-4
Formula C10 H18 N4O4*C 6 H13 N Mol. weight 258,27*99,18 g/mol
HAA2225 N3 -L-Orn(Fmoc)-OH
(S)-2-Azido-5-[(9-fluorenylmethyloxycarbonyl)amino] pentanoic acid
CAS-No. 1994267-98-9
Formula C 20 H20 N4O4 Mol. weight 380,4 g/mol

Reference:
→ Application of metal-free triazole formation in the synthesis of cyclic RGD-DTPA conjugates; S. S. van Berkel, A. Dirks, S. A. Meeuwissen, D. L. Pingen, O. C. Boerman, P. Laverman, F. L. van Delft, J. J. Cornelissen, F. P. Rutjes; Chembiochem 2008; 9: 1805-15. arrow-up-right-from-square https://doi.org/10.1002/cbic.200800074
Azido-Proline
HAA2125 H-L-Pro(4-N3)-OH*HCl (2S,4S)
(2S,4S)-4-Azidopyrrolidine-2-carboxylic acid hydrochloride
CAS-No. 1217823-31-8
Formula C 5H 8 N4O 2*HCl Mol. weight 156,14*36,45 g/mol
Product details

HAA3150 H-L-Pro(4-N3)-OH*HCl (2S,4R)
(2S,4R)-4-Azidopyrrolidine-2-carboxylic acid hydrochloride
CAS-No. 1019849-13-8
Formula C 5H 8 N4O 2*HCl
Mol. weight 156,14*36,45 g/mol
HAA3140 H-D-Pro(4-N3)-OH*HCl (2R,4S)
(2R,4S)-4-Azidopyrrolidine-2-carboxylic acid hydrochloride
CAS-No. 2137086-50-9
Formula C 5H 8 N4O 2*HCl Mol. weight 156,14*36,45 g/mol
HAA3190 H-D-Pro(4-N3)-OH*HCl (2R,4R)
(2R,4R)-4-Azidopyrrolidine-2-carboxylic acid hydrochloride
CAS-No. 2737202-69-4
Formula C 5H 8 N4O 2*HCl Mol. weight 156,14*36,45 g/mol
BAA1905
Boc-L-Pro(4-N3)-OH (2S,4S)
cis-N-alpha-(t-Butyloxycarbonyl)-4-azido-L-proline
CAS-No. 132622-65-2
Formula C10 H16 N4O4 Mol. weight 256,26 g/mol




BAA1930
Boc-L-Pro(4-N3)-OH*DCHA (2S,4R)
trans-N-alpha-(t-Butyloxycarbonyl)-4-azido-L-proline dicyclohexylamine
CAS-No. 132622-68-5 net
Formula C10 H16 N4O4*C12H23 N Mol. weight 256,26*181,32 g/mol
BAA3110
Boc-D-Pro(4-N3)-OH*DCHA (2R,4S)
trans-N-alpha-(t-Butyloxycarbonyl)-4-azido-D-proline dicyclohexylamine
CAS-No. 132622-77-6
Formula C10 H16 N4O4*C12H23 N Mol. weight 256,26*181,32 g/mol


BAA3120 Boc-D-Pro(4-N3)-OH (2R,4R)
cis-N-alpha-(t-Butyloxycarbonyl)-4-azido-D-proline
CAS-No. 650601-59-5
Formula C10 H16 N4O4 Mol. weight 256,26 g/mol

FAA2050 Fmoc-L-Pro(4-N3)-OH (2S,4S)
cis-N-alpha-(9-Fluorenylmethyloxycarbonyl)-4-azido-L-proline
CAS-No. 263847-08-1
Formula C 20 H18 N4O4 Mol. weight 378,38 g/mol
FAA3000 Fmoc-L-Pro(4-N3)-OH (2S,4R)
trans-N-alpha-(9-Fluorenylmethyloxycarbonyl)-4-azido-L-proline
CAS-No. 702679-55-8
Formula C 20 H18 N4O4 Mol. weight 378,38 g/mol
FAA4630 Fmoc-D-Pro(4-N3)-OH (2R,4S)
trans-N-alpha-(9-Fluorenylmethyloxycarbonyl)-4-azido-D-proline
CAS-No. 2137142-63-1
Formula C 20 H18 N4O4 Mol. weight 378,38 g/mol
FAA4720 Fmoc-D-Pro(4-N3)-OH (2R,4R)
cis-N-alpha-(9-Fluorenylmethyloxycarbonyl)-4-azido-D-proline
CAS-No. 1378847-51-8
Formula C 20 H18 N4O4 Mol. weight 378,38 g/mol




Azido-Phenylalanine
HAA9840 H-L-Phe(3-N3)-OH*HCl
3-Azido-L-phenylalanine hydrochloride
CAS-No. 1409940-46-0
Formula C9 H10 N4O 2*HCl
Mol. weight 206,20*36,46 g/mol
HAA1850 H-L-Phe(4-N3)-OH
4-Azido-L-phenylalanine
CAS-No. 33173-53-4
Formula C9 H10 N4O 2
Mol. weight 206,20 g/mol
Product

HAA2980 H-L-Phe(4-N3)-OH*HCl
4-Azido-L-phenylalanine hydrochloride
CAS-No. 34670-43-4
Formula C9 H10 N4O 2*HCl
Mol. weight 206,2*36,45 g/mol

HAA1855 H-D-Phe(4-N3)-OH
4-Azido-D-phenylalanine
CAS-No. 1241681-80-0
Formula C9 H10 N4O 2
Mol. weight 206,20 g/mol

HAA1856 H-D-Phe(4-N3)-OH*HCl
4-Azido-D-phenylalanine hydrochloride
CAS-No. 1241681-80-0
Formula C9 H10 N4O 2*HCl
Mol. weight 206,2*36,45 g/mol

HAA4090 H-L-Phe(4-CH2 -N3)*HCl
4-azidomethyl-L-phenylalanine hydrochloride
CAS-No. 1446772-80-0
Formula C10 H12N4O 2*HCl
Mol. weight 220,23*36,45 g/mol

HAA9480 H-L-Phe(4-Azido-PrTz)-OH*TFA
(S)-2-amino-3-(4-(6-(3-azidopropyl)-1,2,4,5-tetrazin-3yl)phenyl)propanoic acid trifluoroacetic acid salt
CAS-No. 2036323-79-0
Formula C14H16 N 8 O 2*CF 3 COOH Mol. weight 328,34*114,02 g/mol
BAA1850 Boc-L-Phe(4-N3)-OH
N-alpha-t-Butyloxycarbonyl-4-azido-L-phenylalanine
CAS-No. 33173-55-6
Formula C14H18 N4O4 Mol. weight 306,32 g/mol

BAA1855 Boc-D-Phe(4-N3)-OH
N-alpha-t-Butyloxycarbonyl-4-azido-D-phenylalanine
CAS-No. 214630-05-4
Formula C14H18 N4O4 Mol. weight 306,32 g/mol

BAA4660 Boc-L-Phe(4-CH2 -N3)-OH
N-alpha-t-Butyloxycarbonyl-4-azidomethyl-L-phenylalanine
CAS-No. 205127-59-9
Formula C15H20 N4O4
Mol. weight 320,35 g/mol
FAA1905 Fmoc-L-Phe(4-N3)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-4-azido-L-phenylalanine
CAS-No. 163217-43-4
Formula C 24H20 N4O4
Mol. weight 428,44 g/mol


FAA1910 Fmoc-D-Phe(4-N3)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-4-azido-D-phenylalanine
CAS-No. 1391586-30-3
Formula C 24H20 N4O4 Mol. weight 428,44 g/mol
FAA7740 Fmoc-L-Phe(4-CH2 -N3)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-4-azidomethyl-L-phenylalanine
CAS-No. 2375587-79-2
Formula C 25H22N4O4 Mol. weight 442,47 g/mol
HAA3360 N3 -L-Phe-OH*DCHA
(S)-2-Azido-3-phenylpropanoic acid dicyclohexylamine
CAS-No. 79410-36-9
Formula C9 H9 N3 O 2*C13 H23 N Mol. weight 191,19*181,32 g/mol
Azido-Tyrosine
HAA3940 H-L-Tyr(3-N3)-OH
3-Azido-L-tyrosine
CAS-No. 129960-90-3
Formula C9 H10 N4O 3 Mol. weight 222,2 g/mol



Product details

FAA9990 Fmoc-L-Tyr(3-N3)-OH
(S)-2-((((9H-fluoren-9-yl)methoxy)carbonyl)amino)-3-(3azido-4-hydroxyphenyl)propanoic acid
CAS-No. 2382114-46-5
Formula C 23 H20 N4O 5 Mol. weight 444,44 g/mol
HAA9310 H-L-Tyr(2-azidoethyl)-OH
O-2-azidoethyl-tyrosine
CAS-No. 1570523-47-5
Formula C11H14N4O 3 Mol. weight 250,26 g/mol

BAA4650 Boc-L-Tyr(2-azidoethyl)-OH
N-alpha-t-Butyloxycarbonyl-O-(2-azidoethyl)-L-tyrosine
CAS-No. 1434445-10-9
Formula C16 H22N4O 5 Mol. weight 350,38 g/mol
FAA8535 Fmoc-L-Tyr(2-azidoethyl)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-O-(2-azidoethyl)-L-tyrosine
CAS-No. 1454816-10-4
Formula C 26 H24N4O 5 Mol. weight 472,50 g/mol
HAA9215 H-L-Tyr(2-azidoethyl)-OH*HCl
O-(2-azidoethyl)-L-tyrosine hydrochloride
CAS-No. 1567845-62-8
Formula C11H14N4O 3*HCl Mol. weight 250,26*36,46 g/mol



Azido-Valine
HAA9285 N3 -L-Val-OH*CHA
Azido-L-valine cyclohexylammonium salt
CAS-No. 1217462-63-9
Formula C 5H9 N3 O 2*C 6 H13 N Mol. weight 143,15*99,17 g/mol
Product details

References:
→ Improved solid-phase peptide synthesis method utilizing alpha-azide-protected amino acids; J. T. t. Lundquist, J. C. Pelletier; Org Lett 2001; 3: 781-3. arrow-up-right-from-square https://doi.org/10.1021/ol0155485
→ Azide reduction during peptide cleavage from solid support-the choice of thioscavenger?; P. E. Schneggenburger, B. Worbs, U. Diederichsen; J. Pept. Sci. 2010; 16: 10-4. arrow-up-right-from-square https://doi.org/10.1002/psc.1202
→ A DOTA-peptide conjugate by copper-free click chemistry; M. E. Martin, S. G. Parameswarappa, M. S. O‘Dorisio, F. C. Pigge, M. K. Schultz; Bioorg Med Chem Lett 2010; 20: 4805-7. arrow-up-right-from-square https://doi.org/10.1016/j.bmcl.2010.06.111
→ CuI-Catalyzed Azide-Alkyne Intramolecular i-to-(i+4) Side-Chain-to-Side-Chain Cyclization Promotes the Formation of Helix-Like Secondary Structures; M. Scrima, A. Le Chevalier-Isaad, P. Rovero, A. M. Papini, M. Chorev, A. M. D‘Ursi; Eur. J. Org. Chem. 2010; 2010: 446-457. arrow-up-right-from-square https://doi.org/10.1002/ejoc.200901157
→ Synthesis and conformational analysis of a cyclic peptide obtained via i to i+4 intramolecular side-chain to side-chain azide-alkyne 1,3-dipolar cycloaddition; S. Cantel, C. Isaad Ale, M. Scrima, J. J. Levy, R. D. DiMarchi, P. Rovero, J. A. Halperin, A. M. D‘Ursi, A. M. Papini, M. Chorev; J Org Chem 2008; 73: 5663-74. arrow-up-right-from-square https://doi.org/10.1021/jo800142s
→ Side chain-to-side chain cyclization by click reaction; A. Le Chevalier Isaad, A. M. Papini, M. Chorev, P. Rovero; J. Pept. Sci. 2009; 15: 451-4. arrow-up-right-from-square https://doi.org/10.1002/psc.1141
→ An efficient peptide ligation using azido-protected peptides via the thioester method; H. Katayama, H. Hojo, T. Ohira, Y. Nakahara; Tetrahedron Lett. 2008; 49: 5492-5494. arrow-up-right-from-square https://doi.org/10.1016/j.tetlet.2008.07.037
Azido-Linkers and Azido-ADC Linkers
Product details
RL-4100
Azido-SS-COOH
3-((2-azidoethyl)disulfanyl)propanoic acid
CAS-No. 2228857-32-5
Formula C 5H9 N3 O 2 S 2 Mol. weight 207,27 g/mol
RL-3320
Azido-Pen-SS-COOH
3-((2-(5-azidopentanamido)ethyl)disulfanyl)propanoic acid
CAS-No. 2576471-47-9
Formula C10 H18 N4O 3 S 2 Mol. weight 306,40 g/mol
HNN1090 N3 -Cystamine*HCl
Azido-cystamine hydrochloride N+ N S S NH2 -N
CAS-No. 1807512-40-8
Formula C 4H10 N4 S 2*HCl Mol. weight 178,28*36,45 g/mol
Click Chemistry
RL-4120 Biotin-SS-N3
N-(2-((2-azidoethyl)disulfanyl)ethyl)-5-((3aS,4S,6aR)-2oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamide
CAS-No. 1620523-64-9
Formula C14H24N 6 O 2 S 3 Mol. weight 404,57 g/mol
RL-4150 Azido-SS-OpNC
2-((2-azidoethyl)disulfanyl)ethyl (4-nitrophenyl) carbonate
CAS-No. 2766027-28-3
Formula C11H12N4O 5 S 2 Mol. weight 344,36 g/mol
RL-3280 N3 -Pen-Dde
2-(5-azido-1-hydroxypentylidene)-5,5-dimethylcyclohexane-1,3-dione
CAS-No. 1867129-38-1
Formula C13 H19 N3 O 3 Mol. weight 265,31 g/mol
RL-3290 N3 -Pen-Dtpp
5-(5-azido-1-hydroxypentylidene)-1,3-dimethylpyrimidine-2,4,6(1H,3H,5H)-trione
CAS-No. 1867129-42-7
Formula C11H15N 5 O4 Mol. weight 281,27 g/mol
ADC1680 6-Azidohexanoyl-Val-Ala-PAB-Cl
6-Azidohexanoyl-valyl-alanyl-(4-aminobenzyl chloride)
CAS-No. 2706565-03-7
Formula C 21H31 ClN 6 O 3 Mol. weight 450,97 g/mol
ADC1710 Azido-PEG(4)-Val-Ala-PAB-Cl
Azido-tetraethyleneglycol-propanoyl-valyl-alanyl-(4-aminobenzyl chloride)
Formula C 26 H41 ClN 6 O 7 Mol. weight 585,10 g/mol

ADC1290 6-Azidohexanoyl-Val-Ala-PAB
6-azidohexanoyl-valyl-alanyl-(4-aminobenzyl alcohol)
CAS-No. 2706564-30-7
Formula C 21H32N 6 O4 Mol. weight 432,52 g/mol
ADC1300 6-Azidohexanoyl-Val-Ala-PAB-PNP
6-azidohexanoyl-valyl-alanyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
Formula C 28 H35N7 O 8 Mol. weight 597,62 g/mol

ADC1120 6-Azidohexanoyl-Val-Cit-PAB
6-azidohexanoyl-valyl-citrullyl-(4-aminobenzyl alcohol)
CAS-No. 1613321-02-0
Formula C 24H38 N 8 O 5 Mol. weight 518,61 g/mol
ADC1130 6-Azidohexanoyl-Val-Cit-PAB-PNP
6-azidohexanoyl-valyl-citrullyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
CAS-No. 1613321-01-9
Formula C 31H41N9 O 9 Mol. weight 683,71 g/mol
ADC1580
Azido-cyclobutane-1,1-dicarboxamide-Ala-PAB
3-azidopropyl-cyclobutane-1,1-dicarboxamide-alanyl-(4-aminobenzyl alcohol)
CAS-No. 2576471-45-7
Formula C19 H26 N 6 O4 Mol. weight 402,45 g/mol
ADC1590
Azido-cyclobutane-1,1-dicarboxamide-Ala-PAB-PNP
3-azidopropyl-cyclobutane-1,1-dicarboxamide-alanyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
CAS-No. 2576471-36-6
Formula C 26 H29 N7 O 8 Mol. weight 567,55 g/mol


ADC1480
Azido-cyclobutane-1,1-dicarboxamide-Cit-PAB
3-azidopropyl-cyclobutane-1,1-dicarboxamide-citrullyl-(4-aminobenzyl alcohol)
CAS-No. 2576471-33-3
Formula C 22H32N 8 O 5
Mol. weight 488,54 g/mol
ADC1490
Azido-cyclobutane-1,1-dicarboxamide-Cit-PAB-PNP
3-azidopropyl-cyclobutane-1,1-dicarboxamide-citrullyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
CAS-No. 2576471-44-6
Formula C 29 H35N9 O 9
Mol. weight 653,64 g/mol
ADC1330
Azido-PEG(4)-Val-Ala-PAB
azido-tetraethyleneglycol-propanoyl-valyl-alanyl-(4-aminobenzyl alcohol)
Formula C 26 H42N 6 O 8
Mol. weight 566,65 g/mol

ADC1340
Azido-PEG(4)-Val-Ala-PAB-PNP
azido-tetraethyleneglycol-propanoyl-valyl-alanyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
Formula C 33 H45N7 O 12
Mol. weight 731,75 g/mol

ADC1160
Azido-PEG(4)-Val-Cit-PAB
azido-tetraethyleneglycol-propanoyl-valyl-citrullyl-(4-aminobenzyl alcohol)
CAS-No. 2055024-64-9
Formula C 29 H48 N 8 O 9
Mol. weight 652,74 g/mol
ADC1170
Azido-PEG(4)-Val-Cit-PAB-PNP
azido-tetraethyleneglycol-propanoyl-valyl-citrullyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
CAS-No. 1869126-60-2
Formula C 36 H 51N9 O 13
Mol. weight 817,84 g/mol


Other Azido Building Blocks
HNN1110 N3 -TEMPO
4-Azido-2,2,6,6-tetramethyl-1-piperidinyloxy
CAS-No. 63697-61-0
Formula C9 H17N4O Mol. weight 197,26 g/mol

2.4. Alkyne Amino Acids and Related Derivatives
Propargylating Reagents
Product details
RL-8670 5HP2O-alkyne
5-hydroxy-5-methyl-1-(prop-2-yn-1-yl)-1,5-dihydro-2Hpyrrol-2-one
CAS-No. 2484704-61-0
Formula C 8 H9 NO 2 Mol. weight 151,16 g/mol
RL-3945 Mal-Alkyne
3-(2,5-dioxo-2,5-dihydro-1H-pyrrol-1-yl)-N-(prop-2-yn1-yl)propanamide
CAS-No. 548777-19-1
Formula C10 H10 N2O 3 Mol. weight 206,20 g/mol
PEG2755 Propargyl amine
CAS-No. 2450-71-7
Formula C 3 H 5N Mol. weight 55,08 g/mol

MAA1100 Mal-AMCHC-N-Propargylamide
trans-4-[(2,5-dioxo-2,5-dihydro-1H-pyrrol-1-yl)methyl]N-(prop-2-yn-1-yl)cyclohexane-1-carboxamide
CAS-No. 2027476-42-0
Formula C15H18 N2O 3 Mol. weight 274,32 g/mol
LS-4310 DecarboxyBiotin-Alkyne
(3aS,4S,6aR)- 4-(6-heptyn-1-yl)tetrahydro-1H-Thieno[3,4-d]imidazol-2(3H)-one
CAS-No. 887915-53-9
Formula C12H18 N2OS Mol. weight 238,35 g/mol
RL-3490 Biotin-Propargylamide
Biotinyl-N-propargylamide
CAS-No. 773888-45-2
Formula C13 H19 N3 O 2 S Mol. weight 281,37 g/mol
Alkyne-Alkyl Acids and Alkyne-Aryl Acids
RL-8660 Poc-Aoa
2-((((prop-2-yn-1-yloxy)carbonyl)amino)oxy)acetic acid
CAS-No. 1877011-25-0
Formula C 6 H7NO 5 Mol. weight 173,12 g/mol
RL-3330 Alkyne-SS-COOH
3-((2-pent-4-ynamidoethyl)disulfanyl)propanoic acid
CAS-No. 2279938-29-1
Formula C10 H15NO 3 S 2 Mol. weight 261,36 g/mol


Product
RL-4110 DBCO-Suc-SS-COOH
CAS-No. 2749426-25-1
Formula C 24H24N2O4 S 2 Mol. weight 468,59 g/mol
RL-3410
Photo-Click-Heptanoic acid
2-(3-(but-3-ynyl)-3H-diazirin-3-yl)acetic acid
CAS-No. 2049109-24-0
Formula C 7H 8 N2O 2 Mol. weight 152,15 g/mol

RL-2055 Alkyne-myristic acid
13-Tetradecynoic acid
CAS-No. 82909-47-5
Formula C14H24O 2 Mol. weight 224,34 g/mol

RL-2060 Alkyne-palmitic acid
15-Hexadecynoic acid
CAS-No. 99208-90-9
Formula C16 H28 O 2 Mol. weight 252,39 g/mol

RL-3720 Photo-Click-Palmitic acid
11-(3-(but-3-yn-1-yl)-3H-diazirin-3-yl)undecanoic acid
CAS-No. 2988174-40-7
Formula C16 H26 N2O 2 Mol. weight 278,40 g/mol
RL-2065 Alkyne-stearic acid
17-Octadecynoic acid
CAS-No. 34450-18-5
Formula C18 H32O 2 Mol. weight 280,45 g/mol

RL-3760 Photo-Click-Stearic acid
13-(3-(but-3-yn-1-yl)-3H-diazirin-3-yl)tridecanoic acid
CAS-No. 2989205-54-9
Formula C18 H30 N2O 2 Mol. weight 306,45 g/mol
PEG1935 Propargyl-NHS
3-(Prop-2-ynyloxy)propanoic acid succinimidyl ester
CAS-No. 1174157-65-3
Formula C10 H11NO 5 Mol. weight 225,2 g/mol
RL-3460 10-Undecynoyl-OSu
10-Undecynoic acid N-hydroxysuccinimide ester
CAS-No. 1006592-57-9
Formula C15H21NO4 Mol. weight 279,34 g/mol
RL-3780 Fmoc-N-propargyl-MPBA
4-(4-(((9-Fluorenylmethyloxycarbonyl)(propargyl)amino)methyl)-3-methoxyphenoxy)butanoic acid
CAS-No. 1009362-00-8
Formula C 30 H29 NO 6 Mol. weight 499,20 g/mol
Propargylalanine and Propargyl-Propionic Acid
Derivatives
FAA4170 Fmoc-L-Dap(Pentynoyl)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-N-beta-(4-pentynoyl)-L-2,3-diaminopropionic acid
CAS-No. 2250436-47-4
Formula C 23 H22N2O 5 Mol. weight 406,43 g/mol
Product details

FAA2080 Fmoc-alpha-Prg-D-Ala-OH
(S)-2-[(9-Fluorenylmethyloxycarbonyl)amino]-2-methyl4-pentynoic acid
CAS-No. 1198791-58-0
Formula C 21H19 NO4 Mol. weight 349,38 g/mol
FAA4230 Fmoc-L-Dap(Poc)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-N-beta-propargyloxycarbonyl-L-2,3-diaminopropionic acid
CAS-No. 2250437-44-4
Formula C 22H20 N2O 6 Mol. weight 408,41 g/mol


Product details
BAA6510 Boc-L-Arg(Propargyl,Pbf)-OH
(E)-N2-(tert-butoxycarbonyl)-Nw‘-((2,2,4,6,7-pentamethyl-2,3-dihydrobenzofuran-5-yl)sulfonyl)-Nw(prop-2-yn-1-yl)-L-arginine
Formula C 27H40 N4O 7S
Mol. weight 564,70 g/mol
FAA7400 Fmoc-L-Arg(Propargyl,Pbf)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-N‘-(2,2,4,6,7-pentamethyldihydrobenzofuran)-N‘ ‘-propargyl-5-sulfonyl-L-arginine
Formula C 37H42N4O 7S
Mol. weight 686,82 g/mol

Propargylarginine
Click Chemistry
Propargylcysteine
HAA2350 H-L-Cys(Propargyl)-OH*HCl
S-Propargyl-L-cysteine hydrochloride
CAS-No. 3262-64-4
Formula C 6 H9 NO 2 S*HCl Mol. weight 159,21*36,45 g/mol
Product details

HAA9650 H-L-HSec(Propargyl)-OH*HCl
(S)-2-amino-4-(prop-2-yn-1-ylselanyl)butanoic acid hydrochloride
CAS-No. 1601474-63-8
Formula C 7H11NO 2 Se*HCl Mol. weight 220,14*36,46 g/mol
BAA2250 Boc-L-Cys(Propargyl)-OH*DCHA
N-alpha-t-Butyloxycarbonyl-S-propargyl-L-cysteine dicyclohexylamine
CAS-No. 1260119-25-2 net
Formula C11H17NO4 S*C12H23 N Mol. weight 259,32*181,32 g/mol
FAA3810 Fmoc-L-Cys(Propargyl)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-S-propargyl-L-cysteine
CAS-No. 1354752-76-3
Formula C 21H19 NO4 S Mol. weight 381,44 g/mol
FAA9785 Fmoc-L-HCys(Propargyl)-OH
(R)-3-((((9H-fluoren-9-yl)methoxy)carbonyl)amino)-5(prop-2-yn-1-ylthio)pentanoic acid
Formula C 22H21NO4 S Mol. weight 395,47 g/mol


References:
→ Photoinduced addition of glycosyl thiols to alkynyl peptides: use of free-radical thiol-yne coupling for posttranslational double-glycosylation of peptides; M. Lo Conte, S. Pacifico, A. Chambery, A. Marra, A. Dondoni; J Org Chem 2010; 75: 4644-7. arrow-up-right-from-square https://doi.org/10.1021/jo1008178
→ Neoglycopeptides through direct functionalization of cysteine; C. Vala, F. Chrétien, E. Balentova, S. LamandéLangle and Y. Chapleur; Tetrahedron Letters 2011; 52: 17-20. arrow-up-right-from-square https://doi.org/10.1016/j.tetlet.2010.10.021
Propargylglycine
BAA3230 Boc-N-(propargyl)-glycine
N-alpha-t-Butyloxycarbonyl-N-alpha-propargyl-glycine
CAS-No. 158979-29-4
Formula C10 H12NO4 Mol. weight 213,23 g/mol
Product details

FAA4950 Fmoc-N-(propargyl)-glycine
N-alpha-(9-Fluorenylmethyloxycarbonyl)-N-alpha-propargyl-glycine
CAS-No. 1033622-38-6
Formula C 20 H17NO4 Mol. weight 335,35 g/mol
HAA7151 H-L-Pra-OH
L-Propargylglycine
CAS-No. 23235-01-0
Formula C 5H7NO 2 Mol. weight 113,11 g/mol


HAA6490 H-D-Pra-OH
D-Propargylglycine
CAS-No. 23235-03-2
Formula C 5H7NO 2 Mol. weight 113,11 g/mol

HAA9445 H-Sar-Sar-NH-Propargyl*HCl
Di-sarcosine-propargylamide hydrochloride
Formula C9 H15N3 O 2*HCl Mol. weight 197,24*36,45 g/mol
Click Chemistry
RL-8705 Alkynyl-Sar-Sar-OH*DCHA
N-methyl-N-(N-methyl-N-(pent-4-ynoyl)glycyl)glycine dicyclohexylamine
Formula C11H16 N2O4*C12H23 N Mol. weight 240,26*181,32 g/mol
BAA1434 Boc-L-Pra-OH*DCHA
N-alpha-(t-Butyloxycarbonyl)-L-propargylglycine dicyclohexylamine
CAS-No. 63039-49-6
Formula C10 H15NO4*C12H23 N Mol. weight 213,23*181,32 g/mol
BAA1377 Boc-D-Pra-OH*DCHA
N-alpha-t-Butyloxycarbonyl-D-propargylglycine dicyclohexylamine
CAS-No. 63039-47-4
Formula C10 H15NO4*C12H23 N Mol. weight 213,23*181,32 g/mol
FAA1589 Fmoc-L-Pra-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-L-propargylglycine
CAS-No. 198561-07-8
Formula C 20 H17NO4 Mol. weight 335,35 g/mol
FAA1690 Fmoc-D-Pra-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-D-propargylglycine
CAS-No. 220497-98-3
Formula C 20 H17NO4 Mol. weight 335,36 g/mol
WAA6025 Fmoc-L-Pra-Wang Resin
Fmoc-L-Propargylglycine-Wang Resin
Mesh Size 100-200 mesh
DVB 1% DVB





FAA9845 Fmoc-L-Photo-Click-ANon-OH
(S)-2-((((9H-fluoren-9-yl)methoxy)carbonyl)amino)-4(3-(but-3-yn-1-yl)-3H-diazirin-3-yl)butanoic acid
CAS-No. 2763841-90-1
Formula C 24H23 N3 O4
Mol. weight 417,47 g/mol
HAA9720 H-L-Photo-Click-ANon-OH*HCl
(S)-2-amino-4-(3-(but-3-yn-1-yl)-3H-diazirin-3-yl)butanoic acid hydrochloride
CAS-No. 2254648-53-6 net
Formula C9 H13 N3 O 2*HCl
Mol. weight 195,22*36,45 g/mol
Propargyllysine
HAA2085 H-L-Lys(Pentynoyl)-OH*HCl
(S)-2-Amino-6-(pent-4-ynamido)hexanoic acid hydrochloride
CAS-No. 1167421-22-8 net
Formula C11H18 N2O 3 *HCl
Mol. weight 226,27 *36,5 g/mol
HAA9440 H-L-Lys(Pentynoyl)-OH
N6-(pent-4-ynoyl)-L-lysine
CAS-No. 1167421-22-8
Formula C11H18 N2O 3 Mol. weight 226,28 g/mol
HAA2090 H-L-Lys(Poc)-OH*HCl
(S)-Amino-6-((prop-2-ynyloxy)carbonylamino)hexanoic acid hydrochloride
CAS-No. 1428330-91-9
Formula C10 H16 N2O4*HCl Mol. weight 228,25*36,45 g/mol

HAA9390 H-L-Lys(CO-Ethoxypropargyl)-OH*HCl
(2S)-2-amino-6-({[2-(prop-2-yn-1-yloxy)ethoxy]carbonyl}amino)hexanoic acid
Formula C12H20 N2O 5*HCl Mol. weight 272,30*36,45 g/mol
FAA4175 Fmoc-L-Lys(pentynoyl)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-N-epsilon-(4-pentynoyl)-L-lysine
CAS-No. 1159531-18-6
Formula C 26 H28 N2O 5 Mol. weight 448,51 g/mol
FAA8115 Fmoc-L-Lys(Pentynoyl-DIM)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-N-epsilon-[1-(4,4-dimethyl-2,6- dioxocyclohexylidene)pent-4yn-1-yl]-L-lysine
CAS-No. 2408993-33-7
Formula C 34H38 N2O 6 Mol. weight 570,69 g/mol
FAA8135 Fmoc-D-Lys(pentynoyl)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-N-epsilon-(4-pentynoyl)-D-lysine
CAS-No. 2576508-18-2
Formula C 26 H28 N2O 5 Mol. weight 448,51 g/mol
FAA3150 Fmoc-L-Lys(Pryoc)-OH
(S)-2-((9-Fluorenylmethyloxyl)amino)-6-((prop-2-ynyloxy)carbonylamino)hexanoic acid
CAS-No. 1584133-25-4
Formula C 25H26 N2O 6 Mol. weight 450,48 g/mol
FAA9660 Fmoc-L-MeLys(Poc)-OH
N2-(((9H-fluoren-9-yl)methoxy)carbonyl)-N2-methylN6-((prop-2-yn-1-yloxy)carbonyl)-L-lysine
Formula C 26 H28 N2O 6 Mol. weight 464,25 g/mol




FAA9565 Fmoc-D-Lys(Pryoc)-OH
N2-(((9H-fluoren-9-yl)methoxy)carbonyl)-N6-((prop-2yn-1-yloxy)carbonyl)-D-lysine
CAS-No. 2991236-42-9
Formula C 25H26 N2O 6 Mol. weight 450,49 g/mol
FAA8995 Fmoc-L-Lys(Hexynoyl)-OH
N2-(((9H-fluoren-9-yl)methoxy)carbonyl)-N6-(hex-5-ynoyl)-L-lysine
CAS-No. 1219440-73-9
Formula C 27H30 N2O 5 Mol. weight 462,55 g/mol
FAA8905 Fmoc-L-Lys(CO-Ethoxypropargyl)-OH
(2S)-2-({[(9H-fluoren-9-yl)methoxy]carbonyl}amino)6-({[2-(prop-2-yn-1-yloxy)ethoxy]carbonyl}amino) hexanoic acid
Formula C 27H30 N2O 7 Mol. weight 494,54 g/mol
Product details
HAA4970 H-L-Phe(4-NH-Poc)-OH*HCl
4-(Propargyloxycarbonyl)amino-L-phenylalanine hydrochloride
CAS-No. 2576508-14-8
Formula C13 H14N2O4*HCl Mol. weight 262,26*36,45 g/mol
HAA9220 H-L-Phe(4-Eth)-OH*HCl
4-Ethynyl-L-phenylalanine hydrochloride
CAS-No. 188640-63-3
Formula C11H11NO 2*HCl Mol. weight 189,21*36,46 g/mol

Propargylphenylalanine
Click Chemistry
BAA3980 Boc-L-Phe(4-NH-Poc)-OH
N-alpha-t-Butyloxycarbonyl-4-(propargyloxycarbonyl) amino-L-phenylalanine
CAS-No. 2576508-03-5
Formula C18 H22N2O 6 Mol. weight 362,38 g/mol
BAA4670 Boc-L-Phe(4-Eth)-OH
N-alpha-t-Butyloxycarbonyl-4-Ethynyl-L-phenylalanine
CAS-No. 169158-05-8
Formula C16 H19 NO4 Mol. weight 289,33 g/mol
FAA7720 Fmoc-L-Phe(4-NH-Poc)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-4-propargyloxycarbonylamino-L-phenylalanine
CAS-No. 2576508-07-9
Formula C 28 H24N2O 6 Mol. weight 484,5 g/mol
FAA8530 Fmoc-L-Phe(4-Eth)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-4-ethynyl-L-phenylalanine
CAS-No. 1228049-41-9
Formula C 26 H21NO4 Mol. weight 411,46 g/mol
Propargylproline
FAA7130 Fmoc-L-Pro(4-NHPoc)-OH (2S,4S) (2S,4S)-1-(9-Fluorenylmethyloxycarbonyl)-4-(propargyloxycarbonyl)amino-pyrrolidine-2-carboxylic acid
CAS-No. 2451202-17-6
Formula C 24H22N2O 6 Mol. weight 434,44 g/mol
Product details


Product details

Propargylserine
HAA2355 H-L-Ser(Propargyl)-OH*HCl
O-Propargyl-L-serine hydrochloride
CAS-No. 1379150-93-2
Formula C 6 H9 NO 3*HCl Mol. weight 143,14*36,45 g/mol

BAA2260 Boc-L-Ser(Propargyl)-OH*DCHA
N-alpha-t-Butyloxycarbonyl-O-propargyl-L-serine dicyclohexylamine
CAS-No. 145205-94-3 (net)
Formula C11H17NO 5*C12H23 N Mol. weight 243,26*181,32 g/mol
FAA3820 Fmoc-L-Ser(Propargyl)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-O-propargyl-L-serine
CAS-No. 1354752-75-2
Formula C 21H19 NO 5 Mol. weight 365,38 g/mol


References:
→ Isoxazolyl-serine-based agonists of peroxisome proliferator-activated receptor: design, synthesis, and effects on cardiomyocyte differentiation; Z. L. Wei, P. A. Petukhov, F. Bizik, J. C. Teixeira, M. Mercola, E. A. Volpe, R. I. Glazer, T. M. Willson, A. P. Kozikowski; J Am Chem Soc 2004; 126: 16714-5. arrow-up-right-from-square https://doi.org/10.1021/ja046386l
→ Lacosamide isothiocyanate-based agents: novel agents to target and identify lacosamide receptors; K. D. Park, P. Morieux, C. Salome, S. W. Cotten, O. Reamtong, C. Eyers, S. J. Gaskell, J. P. Stables, R. Liu, H. Kohn; J Med Chem 2009; 52: 6897-911. arrow-up-right-from-square https://doi.org/10.1021/jm9012054
Propargyltyrosine
HAA1971 H-L-Tyr(Propargyl)-OH*HCl
O-Propargyl-L-tyrosine hydrochloride
CAS-No. 1919043-11-0
Formula C12H13 NO 3*HCl Mol. weight 219,24*36,45 g/mol
Product details

BAA2265 Boc-L-Tyr(Propargyl)-OH*DCHA
N-alpha-t-Butyloxycarbonyl-O-propargyl-L-tyrosine dicyclohexylamine
CAS-No. 340732-79-8
Formula C17H21NO 5 Mol. weight 319,35 g/mol
FAA3830 Fmoc-L-Tyr(Propargyl)-OH
N-alpha-(9-Fluorenylmethyloxycarbonyl)-O-propargyl-L-tyrosine
CAS-No. 1204595-05-0
Formula C 27H23 NO 5 Mol. weight 441,48 g/mol
Alkyne-Linker and Alkyne-ADC Linker
RL-8850 Ynamine-Alkyne
N-(1-ethynyl-1H-benzo[d]imidazol-6-yl)hept-6-ynamide
Formula C16 H15N3 O Mol. weight 265,32 g/mol
RL-8855 Ynamine-PEG(4)-Alkyne
N-(1-ethynyl-1H-benzo[d]imidazol-6-yl)-4,7,10,13-tetraoxahexadec-15-ynamide
Formula C 21H25N3 O 5 Mol. weight 399,45 g/mol
RL-8860 Biotin-Ynamine
N-(1-ethynyl-1H-benzo[d]imidazol-6-yl)-5((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamide
Formula C19 H21N 5 O 2 S Mol. weight 383,47 g/mol
Product details


Product details
PAA1170 Poc-Cystamine*HCl
Prop-2-yn-1-yl (2-((2-aminoethyl)disulfaneyl)ethyl) carbamate
CAS-No. 1266354-28-2 net
Formula C 8 H14N2O 2 S 2*HCl
Mol. weight 234,33*36,45 g/mol
BNN1320 Boc-Cystamine-Poc
Tert-butyl (2-((2-(((prop-2-yn-1-yloxy)carbonyl)amino) ethyl)disulfaneyl)ethyl)carbamate
CAS-No. 2171512-56-2
Formula C13 H22N2O4 S 2 Mol. weight 334,45 g/mol
ADC1310 4-Pentynoyl-Val-Ala-PAB
4-pentynoyl-valyl-alanyl-(4-aminobenzyl alcohol)
CAS-No. 1956294-75-9
Formula C 20 H27N3 O4 Mol. weight 373,45 g/mol

ADC1690 4-Pentynoyl-Val-Ala-PAB-Cl
4-Penynoyl-valyl-alanyl-(4-aminobenzyl chloride)
Formula C 20 H26 ClN3 O 3 Mol. weight 391,90 g/mol
ADC1320 4-Pentynoyl-Val-Ala-PAB-PNP
4-pentynoyl-valyl-alanyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
CAS-No. 1956294-76-0
Formula C 27H30 N4O 8 Mol. weight 538,55 g/mol
ADC1140 4-Pentynoyl-Val-Cit-PAB
4-pentynoyl-valyl-citrullyl-(4-aminobenzyl alcohol)
CAS-No. 2708150-97-2
Formula C 23 H33 N 5 O 5 Mol. weight 459,54 g/mol

ADC1150 4-Pentynoyl-Val-Cit-PAB-PNP
4-pentynoyl-valyl-citrullyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
Formula C 30 H36 N 6 O 9 Mol. weight 624,64 g/mol

ADC1350 Alkyne-PEG(4)-Val-Ala-PAB
propargyl-tetraethyleneglycol-propanoyl-valyl-alanyl-(4-aminobenzyl alcohol)
CAS-No. 2348405-90-1
Formula C 29 H45N3 O 9 Mol. weight 579,68 g/mol
ADC1720 Alkyne-PEG(4)-Val-Ala-PAB-Cl
Propargyl-tetraethyleneglycol-propanoyl-valyl-alanyl-(4-aminobenzyl chloride)
Formula C 29 H44ClN3 O 8 Mol. weight 598,13 g/mol
ADC1360 Alkyne-PEG(4)-Val-Ala-PAB-PNP
propargyl-tetraethyleneglycol-propanoyl-valyl-alanyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
CAS-No. 2348405-91-2
Formula C 36 H48 N4O 13
Mol. weight 744,79 g/mol
ADC1180 Alkyne-PEG(5)-Val-Cit-PAB
propargyl-tetraethyleneglycol-propanoyl-valyl-citrullyl-(4-aminobenzyl alcohol)
Formula C 32H 51N 5 O 10
Mol. weight 665,77 g/mol

ADC1190 Alkyne-PEG(5)-Val-Cit-PAB-PNP
propargyl-tetraethyleneglycol-propanoyl-valyl-citrullyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
Formula C 39 H 54N 6 O 14
Mol. weight 830,88 g/mol

ADC1600
Propargyl-cyclobutane-1,1-dicarboxamide-Ala-PAB
propargyl-cyclobutane-1,1-dicarboxamide-alanyl-(4-aminobenzyl alcohol)
CAS-No. 2576471-28-6
Formula C19 H23 N3 O4 Mol. weight 357,40 g/mol
ADC1610
Propargyl-cyclobutane-1,1-dicarboxamide-Ala-PAB-PNP
propargyl-cyclobutane-1,1-dicarboxamide-alanyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
CAS-No. 2576471-42-4
Formula C 26 H26 N4O 8 Mol. weight 522,51 g/mol
ADC1500
Propargyl-cyclobutane-1,1-dicarboxamide-Cit-PAB
propargyl-cyclobutane-1,1-dicarboxamide-citrullyl-(4-aminobenzyl alcohol)
CAS-No. 2576471-27-5
Formula C 22H29 N 5 O 5 Mol. weight 443,50 g/mol
ADC1510
Propargyl-cyclobutane-1,1-dicarboxamide-Cit-PAB-PNP
propargyl-cyclobutane-1,1-dicarboxamide-citrullyl-(4-aminobenzyl)-(4-nitrophenyl)-carbonate
CAS-No. 2576471-40-2
Formula C 29 H32N 6 O 9 Mol. weight 608,60 g/mol
ADC1790
Alkyne-HMPO-OH
(2-ethoxy-7-(prop-2-yn-1-yloxy)benzo[d][1,3]dioxol-5-yl) methanol
CAS-No. 3028213-63-7
Formula C13 H14O 5 Mol. weight 250,25 g/mol
ADC1800
Alkyne-HMPO-PNP
(2-ethoxy-7-(prop-2-yn-1-yloxy)benzo[d][1,3]dioxol-5-yl) methyl (4-nitrophenyl) carbonate
CAS-No. 3028213-53-5
Formula C 20 H17NO 9 Mol. weight 415,35 g/mol
circle-arrow-right
Find more linkers and background on linker technology in our booklet Linkerology®!

Auxiliary Reagents
RL-2010 TBTA
Tris[(1-benzyl-1H-1,2,3-triazol-4-yl)methyl]amine
CAS-No. 510758-28-8
Formula C 30 H30 N10
Mol. weight 530,63 g/mol
Product details

RL-2210 THPTA
Tris(3-hydroxypropyltriazolylmethyl)amine
CAS-No. 760952-88-3
Formula C18 H30 N10 O 3
Mol. weight 434,51 g/mol

3. Spermines and Amines for Click Chemistry
Polyamines are aliphatic cations with multiple functions in cell proliferation and differentiation and are essential for normal cell growth and development in eukaryotes. These molecules carry positive charges at their primary and secondary amino groups at physiological pH. Consequently, polyamines bind to various anionic macromolecules including DNA, RNA, acidic phospholipids, and certain proteins. These polycationic alkylamines are involved in various critical cellular functions, such as maintaining chromatin structure, regulating ion-channels, maintaining membrane stability, modulating protein synthesis, and scavenging free radicals. Polyamines also serve as substrates for transglutaminase reactions and for the synthesis of the translational regulator hypusine.
Crucial parts of the biological function of polyamines are the regulation of gene expression by altering DNA structure, the modulation of protein synthesis by binding to RNA, and the modulation of signal transduction pathways. The binding of polyamines to both RNA and DNA leads to conformational changes of those nucleic acids. Polyamines cause the conformational transition of DNA from the B form to the Z form and also cause bending of DNA. Both structural alterations are known to influence transcription. Close to 80% of all polyamines in the cell are associated with RNA, while spermine in particular has been shown to stabilize tRNA structures. Binding of polyamines to RNA causes structural changes that increase the efficiency of protein synthesis.
Polyamines are also known to modulate DNA-protein interactions, e.g., by enhancing the binding of specific gene-regulatory proteins to certain regulatory sequences termed response elements. The poly-amine spermine has been reported to facilitate the binding of estrogen receptor and nuclear factor κB (NF-κB) to their respective response elements at 100 to 500 μM concentrations. Polyamines are also involved in modulating ligand-receptor interactions, for example N-methyl-D-aspartate (NMDA) receptors, which are important for the excitatory synaptic transmission in the brain and spinal cord.
Moreover, polyamines have been implicated as important molecules in virus-host interactions since many viruses utilize and manipulate polyamines for their own replication. Those pathogens depend on the presence of polyamines for numerous aspects of their replication cycles, such as DNA and RNA polymerization, genome packaging, and viral protein translation. Certain viruses even appear to stimulate polyamine synthesis upon infection, a fact that underlines the importance of this class of molecules for the viral life cycle.
The polyamine metabolic pathway and thus polyamine levels are strictly regulated in cells. However, dysregulation of polyamine metabolism is a frequently observed event in cancer. For example, elevated levels of polyamines have been associated with breast, colon, prostate, and skin cancers. Consequently, polyamine synthesis, metabolism, uptake, and function may be promising targets for cancer therapy.
References:
→ Structural analysis of DNA interactions with biogenic polyamines and cobalt(III)hexamine studied by Fourier transform infrared and capillary electrophoresis; A. A. Ouameur, H. A. Tajmir-Riahi; J Biol Chem 2004; 279: 42041-54. arrow-up-right-from-square https://doi.org/10.1074/jbc.M406053200
→ Polyamines and Their Role in Virus Infection; B. C. Mounce, M. E. Olsen, M. Vignuzzi, J. H. Connor; Microbiol. Mol. Biol. Rev. 2017; 81: e00029-17. arrow-up-right-from-square https://doi.org/10.1128/MMBR.00029-17
arrow-up back to content
Click Chemistry
→ Targeting polyamine metabolism for cancer therapy and prevention; T. R. Murray-Stewart, P. M. Woster, R. A. Casero, Jr.; Biochem J 2016; 473: 2937-53. arrow-up-right-from-square https://doi.org/10.1042/BCJ20160383
→ Polyamine catabolism in carcinogenesis: potential targets for chemotherapy and chemoprevention; V. Battaglia, C. DeStefano Shields, T. Murray-Stewart, R. A. Casero, Jr.; Amino Acids 2014; 46: 511-9. arrow-up-right-from-square https://doi.org/10.1007/s00726-013-1529-6
→ Targeting polyamine metabolism and function in cancer and other hyperproliferative diseases; R. A. Casero, Jr., L. J. Marton; Nat Rev Drug Discov 2007; 6: 373-90. arrow-up-right-from-square https://doi.org/10.1038/nrd2243
Product details
FNN1020 Fmoc-EDA-N3
1-[(9-Fluorenylmethyloxycarbonyl)amino]-2-azidoethane
CAS-No. 432507-62-5
Formula C17H16 N4O 2 Mol. weight 308,33 g/mol
FNN1030 Fmoc-DAP-N3
1-[(9-Fluorenylmethyloxycarbonyl)amino]-3-azidopropane
CAS-No. 1021422-85-4
Formula C18 H18 N4O 2 Mol. weight 322,36 g/mol
SNN1170 Spermine(HHHN3)*3HCl
N1-(3-Aminopropyl)-N4-(3-azidopropyl)butane-1,4-diamine trihydrochloride
CAS-No. 1190203-77-0
Formula C10 H24N 6*3HCl Mol. weight 228,34*109,38 g/mol
SNN1230 Spermine(N3 BBB)
tert-butyl (4-((3-azidopropyl)(tert-butoxycarbonyl) amino)butyl)(3-((tert-butoxycarbonyl)amino)propyl) carbamate
CAS-No. 1190203-80-5
Formula C 25H48 N 6 O 6 Mol. weight 528,70 g/mol
SNN1210 Spermine(N3 HHN3)*2TsOH
N1-(3-Aminopropyl)-N4-(3-azidopropyl)butane-1,4-diamine bistosylate
CAS-No. 2250433-79-3
Formula C10 H22N 8*C14H16 O 6 S 2 Mol. weight 254,34*344,40 g/mol





4. Click Reagents for Drug Delivery
4.1. Principles of
Polymer Therapeutics
Peptides, proteins, and other biomolecules have a high potential as drugs due to their usually high specificity and efficacy. However, they often show poor pharmacokinetic properties, with their low stability under physiological conditions being a major factor. Since synthetic biomolecules are similar to endogenous molecules found in the human body, they are quickly degraded by enzymes and cleared from the system. Especially peptides and small proteins are susceptible to renal clearance. Additionally, the immunogenic responses and side effects elicited by many drugs, in particular protein drugs, are exacerbated by their hydrophobicity. Conjugation to biocompatible polymers, such as PEG (poly(ethylene glycol)), PGA (poly(glutamic acid)) or POX (poly(2-oxazoline)), increases aqueous solubility of a drug and often drastically enhances its pharmacokinetics at both the whole organism and subcellular level. By using bi- or multifunctional polymers, a linkage between two compounds can be formed or multivalent conjugates generated.
Fig. 12: Synthesis of a PEGylated RGD-DOTA conjugate for PET imaging (adapted from Chen et al., J. Nucl. Med. 2004).
The main advantage of proteins, antibodies, siRNA, mRNA and other biomolecules when applied as drugs is their high specificity in combination with their low side effects, as they usually only interact with their dedicated target. A current focus is the study of targeted drug delivery systems for the controlled delivery and/or release of therapeutic agents. To this end, a biocompatible polymeric carrier is covalently linked to an active agent and a targeting moiety. This recognition part can be a peptide or protein that specifically binds to a certain cellular receptor, or an antibody against a specific antigen on the cell surface. Especially in the context of antibody-drug conjugate, the introduction of a PEG moiety can be very beneficial for pharmacokinetics by attenuating the frequently hydrophobic nature of payload molecules. After internalization of the whole conjugate, the active part (e.g., a nucleic acid or toxin) is released by e.g., variations in pH, temperature, or enzyme concentration. Consequently, the active agent is enabled to exert its function (e.g., the inhibition of a certain enzyme or the initiation of apoptosis) at a high local drug concentration, further increasing drug efficacy. If the active agent and/or the targeting entity are tailored to certain characteristics of an individual or a group of individuals (e.g., specific cancer cell antigens), this approach opens the door to individualized therapies (“personalized medicine”).
To enlarge the field of Polymer Therapeutics to new classes of drug molecules, there is a constant search for new types of polymers. One interesting group are homopolymers of amino acids. Typical examples are polylysine and poly-α-glutamic acid (PGA) - polymers that do not exist in nature, but which show good physiological properties due to their similarity to natural proteins. In particular, poly(glutamic acid) has been identified as suitable carrier system.
PGA shows the ability to conjugate with partners on its N- and C-termini, analogous to the α and ω derivatization of a PEG – poly(ethylene glycol). Additionally, the glutamic acid side-chains may be used for further decoration of the polymer. Therefore, a multivalent presentation of a specific molecule along the polymer chain is possible, which is especially interesting for small molecules (Fig. 13). It is theoretically also possible to PEGylate a small molecule. However, inactivation of the small drug molecule is often the consequence. PGA is hence an ideal carrier for low molecular weight APIs.
Fig. 13: Multivalent presentation of a drug on poly-α-(glutamic acid).
Fig. 14: A pentapeptide (green) conjugated to poly-α-(glutamic acid) (blue).
4.2. PEGylation Improves Drug Delivery and Pharmacokinetics
Small drug molecules, but also large biomolecules like antibodies suffer from rapid clearance, causing a sharp decrease in plasma concentration of the drug as it is removed from the body. Consequently, drug administration has to be repeated within relatively short time intervals in order to keep its plasma concentration over a certain threshold. Otherwise, immunogenic reactions may be triggered.
Fig. 15: Pharmacokinetic properties of a PEGylated drug in comparison to a non-PEGylated drug.
PEGylated drugs show decreased rates of renal clearance and reduced immunogenicity. Consequently, plasma half-life of the drug is significantly increased, extending the time intervals between applications of the drug over the course of the treatment. This is due to the following mechanisms:
1. Preventing Degradation and Reducing Immunogenicity:
PEG chains cover the surface of a biopharmaceutical and thus effectively shield it from recognition by the immune system. This PEG layer has characteristics that are rather similar to a solvent, preventing uptake by cells of the retinal endothelial system (macrophage system). Therefore, recognition by the immune system (antibodies, proteases, and other degradation enzymes) is significantly attenuated. The drug stays intact and is not degraded or metabolized during its presence in and journey through the body.
Poly(ethylene glycol) is a polymeric linear structure with repeating polyethylene oxide units.
Mass spectrum of a polyethylene glycol showing the typical signals with a difference of m/z = 44
Whenever there is a distribution of molecular weights, the weight average M w° is always greater than the number average M n° and the polydispersity Đ is greater than 1.
Fig. 16: Composition of poly(ethylene glycols), typical mass spectrum of a polydisperse PEG, and formula for the polydispersity Đ.
2. Preventing Excretion:
PEG is very hygroscopic by nature and surrounded by a large solvation sphere of water. Thus, the overall hydrodynamic radius of a biopharmaceutical may be increased by PEGylation by up to an order of magnitude, to a size larger than the diameter of the glomerular capillaries (6 to 12 nm). Consequently, a PEGylated drug can no longer be excreted through the kidneys, and pharmacologic half-life is significantly extended.
Chemical/Physical Properties and Quality Parameters of PEGs, Dispersity
Depending on whether a given PEG consists of a single molecular weight species (a defined number n of repeating units) or of a range of species with an average mass and a distribution of n around a mean value, PEG polymers are referred to as monodisperse/uniform or polydisperse, respectively. If the polymer is polydisperse, its mass spectrum will show a range of different molecular weights (Fig. 16).
A measure of the distribution of molecular weights in a polymer is given by the Dispersity Đ, which is defined as the ratio between the weight average molecular weight M w and the number average molecular weight M n . The weight average M w does not “count” species just by their number but takes into account the total weight of each species and is therefore a much more realistic indicator of the gross mechanical properties of a polymer.
In case of a homogeneous PEG, which consists only of one polymer species with a defined chain length, M w is equal to M n , thus the dispersity Đ equals 1 and the compound is referred to as monodisperse. Whenever there is a distribution of molecular weights, the weight average M w is always greater than the number average M n , and consequently the dispersity Đ is greater than 1. The dispersity of PEGs typically used in PEGylations ranges between 1.05 and 1.50.
However, whenever a PEGylated drug candidate needs to be approved by EMEA, FDA and other authorities, it is easier and faster if this compound is a defined species with a defined molecular weight. Therefore, the need for large but monodisperse PEGs is increasing.
Summary of Chemical and Physical Properties of PEGs:
• Good solubility in BOTH hydrophilic AND hydrophobic solvents as water, toluene, methylene chloride, and many other organic solvents.
• Insoluble in diethyl ether, hexane, ethylene glycol.
• Insoluble in water at elevated temperature.
• The solubility is influenced by forming derivatives.
• Highly mobile in water with high exclusion volume; large hydrodynamic radius.
• Complex formation with metal cations. Can be used to precipitate proteins and nucleic acids. Form two-phase system with aqueous solutions of other polymers. Non-toxic and FDA approved for use in drug products.
PEGylating Biopharmaceuticals and Small Molecules has the Following Effects:
Improves solubility of conjugated molecules. Renders proteins non-immunogenic and tolerogenic. Reduces the rate of renal clearance through the kidney and alters pharmacokinetics.
• Alters electroosmotic flow.
• Increases cell permeability.
References:
→ Structure of a PEGylated protein reveals a highly porous double-helical assembly; G. Cattani, L. Vogeley and P. B. Crowley; Nat Chem 2015; 7: 823-8. arrow-up-right-from-square https://doi.org/10.1038/nchem.2342
→ Peptide and Protein PEGylation III: Advances in Chemistry and Clinical Applications; F. M. Veronese, J. M. Harris; Adv. Drug Deliv. Rev. 2008; 60: 1-88.
→ PEGylation of native disulfide bonds in proteins; S. Brocchini, S. Balan, A. Godwin, J. W. Choi, M. Zloh, S. Shaunak; Nat Protoc 2006; 1: 2241-52. arrow-up-right-from-square https://doi.org/10.1038/nprot.2006.346
→ Pegylated Arg-Gly-Asp Peptide: 64Cu Labeling and PET Imaging of Brain Tumor αvβ3-Integrin Expression; X. Chen, Y. Hou, M. Tohme, R. Park, V. Khankaldyyan, I. Gonzales-Gomez, J. R. Bading, W. E. Laug, P. S. Conti; J Nucl Med 2004; 45: 1776.
→ Chemistry for peptide and protein PEGylation; M. J. Roberts, M. D. Bentley, J. M. Harris; Adv Drug Deliv Rev 2002; 54: 459-76. arrow-up-right-from-square https://doi.org/10.1016/S0169-409X(02)00022-4
→ Peptide and protein PEGylation: a review of problems and solutions; F. M. Veronese; Biomaterials 2001; 22: 405-17. arrow-up-right-from-square https://doi.org/10.1016/S0142-9612(00)00193-9
→ Functionalization of poly(ethylene glycol) and monomethoxy-poly(ethylene glycol); A. F. Bückmann, M. Morr, G. Johansson; Makromol. Chem. 1981; 182: 1379-1384. arrow-up-right-from-square https://doi.org/10.1002/macp.1981.021820509
Click Chemistry
→ New, easily removable poly(ethylene glycol) supports for the liquid-phase method of peptide synthesis; V. N. R. Pillai, M. Mutter, E. Bayer, I. Gatfield; J. Org. Chem. 1980; 45: 5364-5370. arrow-up-right-from-square https://doi.org/10.1021/jo01314a032
→ Synthesis and characterization of poly(ethylene glycol) derivatives; J. M. Harris, E. C. Struck, M. G. Case, M. S. Paley, M. Yalpani, J. M. Van Alstine, D. E. Brooks; J Polymer Sci Polymer Chem Ed 1984; 22: 341-352. arrow-up-right-from-square https://doi.org/10.1002/pol.1984.170220207
→ Attachment of drugs to polyethylene glycols; S. Zalipsky, C. Gilon, A. Zilkha; Eur. Polym. J. 1983; 19: 1177-1183. arrow-up-right-from-square https://doi.org/10.1016/0014-3057(83)90016-2
→ Effect of covalent attachment of polyethylene glycol on immunogenicity and circulating life of bovine liver catalase; A. Abuchowski, J. R. McCoy, N. C. Palczuk, T. van Es, F. F. Davis; J Biol Chem 1977; 252: 3582-6.
→ Process design for large-scale purification of formate dehydrogenase fromcandida boidinii by affinity partition; A. Cordes, M.-R. Kula; J Chromat 1986; 376: 375-384. arrow-up-right-from-square https://doi.org/10.1016/s0378-4347(00)80853-1
→ Chemical modification of horseradish peroxidase with ethanal-methoxypolyethylene glycol: Solubility in organic solvents, activity, and properties; P. Wirth, J. Souppe, D. Tritsch, J.-F. Biellmann; Bioorg. Chem. 1991; 19: 133-142. arrow-up-right-from-square https://doi.org/10.1016/0045-2068(91)90029-o
→ The Synthesis of Substituted Methoxy-Poly(Ethyleneglycol) Derivatives Suitable for Selective Protein Modification; T. P. Kogan; Synth. Commun. 1992; 22: 2417-2424. arrow-up-right-from-square https://doi.org/10.1080/00397919208019100
4.3. Azido-PEG Derivatives for Click Chemistry
RL-4030 PFB-mercaptopropionyl-PEG3-N3
Perfluorobiphenyl-mercaptopropionyl-PEG(3)-N3
Formula C 23 H21F9 N4O4 S Mol. weight 620,49 g/mol
PEG1088 N3 -PEG(8)-OH
alpha-Azido-omega-hydroxy octa(ethylene glycol)
CAS-No. 352439-36-2
Formula C16 H33 N3 O 8 Mol. weight 395,45 g/mol
Product details

PEG2065 Biotin-TEG-ATFBA
Biotin-triethylenglycol-( p-azido-tetrafluorobenzamide)
CAS-No. 1264662-85-2
Formula C 27H37F4N7 O 6 S Mol. weight 663,68 g/mol

PEG4940
Biotin-PEG(3)-N3
11-[D(+)-Biotinylamino]-1-azido-3,6,9-trioxaundecane
CAS-No. 875770-34-6
Formula C18 H32N 6 O 5 S Mol. weight 444,55 g/mol
PEG6795
Biotin-PEG3-Ph(4-N3)
Biotin-PEG3-phenyl azide
CAS-No. 2088238-77-9
Formula C 25H37N7 O 6 S Mol. weight 563,67 g/mol
PEG7990
Biotin-PEG(4)-N3
(3aS,4S,6aR)-4-(28-azido-5,21-dioxo-9,12,15,18-tetraoxa-6,22-diazaoctacosyl)tetrahydro-1H-thieno[3,4-d]imidazol-2(3H)-one
CAS-No. 1006592-62-6
Formula C 27H49 N7 O 7S Mol. weight 615,79 g/mol
PEG7960
Biotin-PEG(4)-Dde-N3
N-(19-azido-15-(4,4-dimethyl-2,6-dioxocyclohexylidene)-3,6,9,12-tetraoxa-16-azanonadecyl)-5-((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamide
CAS-No. 1802907-93-2
Formula C 32H 53 N7 O 8 S Mol. weight 695,87 g/mol
PEG8000
Biotin-PEG(4)-Picolyl-N3
(3aS,4S,6aR)-4-(1-(6-(azidomethyl)pyridin-3-ylamino)1,17-dioxo-4,7,10,13-tetraoxa-16-azahenicosan-21-yl) tetrahydro-1H-thieno[3,4-d]imidazol-2(3H)-one
CAS-No. 2222687-71-8
Formula C 27H42N 8 O 7S Mol. weight 622,74 g/mol



PEG7970
Biotin-PEG(4)-Dde-Picolyl-N3
N-(6-(azidomethyl)pyridin-3-yl)-15-(4,4-dimethyl-2,6dioxocyclohexylidene)-1-(5-((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamido)-3,6,9,12-tetraoxa-16-azanonadecan-19-amide
CAS-No. 2055048-42-3
Formula C 38 H 57N9 O 9 S Mol. weight 815,98 g/mol
PEG8100
Biotin-PEG(4)-SS-Azide
N-(2-((3-((3-azidopropyl)amino)-3-oxopropyl)disulfaneyl)ethyl)-1-(5-((3aS,4S,6aR)-2-oxohexahydro-1Hthieno[3,4-d]imidazol-4-yl)pentanamido)-3,6,9,12-tetraoxapentadecan-15-amide
CAS-No. 1260247-52-6
Formula C 29 H 52N 8 O 8 S 3 Mol. weight 736,96 g/mol
PEG4330
Biotin-PEG(7)-N3
alpha-Biotin-omega-azido hepta(ethylene glycol)
CAS-No. 1334172-75-6
Formula C 26 H48 N 6 O 9 S Mol. weight 620,76 g/mol



PEG4340 Biotin-PEG(11)-N3
[2-(2-aminoethoxy)ethoxy]acetic acid tert-butyl ester*HCl
CAS-No. 956494-20-5
Formula C 34H 64N 6 O 13 S Mol. weight 796,97 g/mol
PEG4350
Biotin-PEG(23)-N3
alpha-Biotin-omega-azido 23(ethylene glycol)
CAS-No. 956494-20-5
Formula C 58 H112N 6 O 25 S Mol. weight 1325,6 g/mol


PEG7215 N3 -PEG2-NMe3 mesylate
2-(2-(2-azidoethoxy)ethoxy)-N,N,N-trimethylethan-1-aminium
CAS-No. 1447915-25-4
Formula C10 H24N4O 5 S Mol. weight 312,39 g/mol
PEG4980 H2 N-PEG(2)-N3*TosOH
2-[2-(2-Azidoethoxy)ethoxy]ethanaminium tosylat
CAS-No. 166388-57-4
Formula C 7H14N4O 2*C 7H 8 O 3 S Mol. weight 174,20*172,20 g/mol

PEG3060 H2 N-PEG(3)-N3
1-Amino-11-azido-3,6,9-trioxaundecane
CAS-No. 134179-38-7
Formula C 8 H18 N4O 3 Mol. weight 218,25 g/mol

RL-4320 N3 -PEG(3)-NH-Suc
1-azido-13-oxo-3,6,9-trioxa-12-azahexadecan-16-oic acid
CAS-No. 1202400-17-6
Formula C12H22N4O 6 Mol. weight 318,33 g/mol
BNN1150 N3 -TOTA
1-Azido-4,7,10-trioxa-13-tridecanamine
CAS-No. 1162336-72-2
Formula C10 H22N4O 3 Mol. weight 246,31 g/mol

PEG5320 N3 -PEG(4)-NH2
14-Azido-3,6,9,12-tetraoxatetradecan-1-amine
CAS-No. 951671-92-4
Formula C10 H22N4O4 Mol. weight 262,31 g/mol

Click Chemistry
PEG1087 H2 N-PEG(6)-N3
alpha-Amino-omega-azido hexa(ethylene glycol)
CAS-No. 957486-82-7
Formula C14H30 N4O 6 Mol. weight 350,42 g/mol

PEG2350 H2 N-PEG(7)-N3
alpha-Amino-omega-azido hepta(ethylene glycol)
CAS-No. 1333154-77-0
Formula C16 H34N4O 7 Mol. weight 394,46 g/mol

PEG3050 H2 N-PEG(8)-N3
alpha-Amino-omega-azido octa(ethylene glycol)
CAS-No. 857891-82-8
Formula C18 H38 N4O 8 Mol. weight 438,52 g/mol

PEG1081 H2 N-PEG(11)-N3
alpha-Amino-omega-azido undecae(ethylene glycol)
CAS-No. 1800414-71-4
Formula C 24H 50 N4O 11 Mol. weight 570,69 g/mol

PEG3070 H2 N-PEG(23)-N3
alpha-Azido-omega-amino 23(ethylene glycol)
CAS-No. 2172677-19-7
Formula C 48 H98 N4O 23 Mol. weight 1099,3 g/mol
23

PEG3080 H2 N-PEG(35)-N3
alpha-Azido-omega-amino 35(ethylene glycol)
CAS-No. 749244-38-0
Formula C 72H146 N4O 35 Mol. weight 1627,94 g/mol
N3
35

PEG3010 H2 N-PEG-N3 (3 kDa)
alpha-Amino-omega-azido poly(ethylene glycol)
Mol. weight 3000 Da

PEG3030 H2 N-PEG-N3 (5 kDa)
alpha-Amino-omega-azido poly(ethylene glycol)
Mol. weight 5000 Da

PEG3000 H2 N-PEG-N3 (10 kDa)
alpha-Amino-omega-azido poly(ethylene glycol)
Mol. weight 10000 Da

PEG3020 H2 N-PEG-N3 (20 kDa)
alpha-Amino-omega-azido poly(ethylene glycol)
Mol. weight 20000 Da

PEG4960 Boc-NH-PEG(2)-N3
1-(t-Butyloxycarbonyl-amino)-3,6-dioxa-8-octaneazide
CAS-No. 950683-55-3
Formula C11H22N4O4
Mol. weight 274,32 g/mol

BAA4920 Boc-Aoa-NH-PEG3-N3
tert-butyl ((14-azido-2-oxo-6,9,12-trioxa-3-azatetradecyl)oxy)carbamate
CAS-No. 1803191-52-7
Formula C15H29 N 5 O 7
Mol. weight 391,43 g/mol
Click Chemistry
PEG8160 N3 -PEG(3)-NH-Boc
t-Butyl N-(2-(2-(2-(2-azidoethoxy)ethoxy)ethoxy)ethyl) carbamate
CAS-No. 642091-68-7
Formula C13 H26 N4O 5 Mol. weight 318,37 g/mol
PEG4900 N3 -EEEt-OH
2-[2-(2-Azidoethoxy)ethoxy]ethanol
CAS-No. 86520-52-7
Formula C 6 H13 N3 O 3 Mol. weight 175,19 g/mol


PEG3760 N3 -PEG(3)-OH
alpha-Azido-omega-hydroxy tetra(ethylene glycol)
CAS-No. 86770-67-4
Formula C 8 H17N3 O4 Mol. weight 219,24 g/mol

PEG5300 N3 -PEG(4)-OH
2-(2-(2-(2-(2-Azidoethoxy)ethoxy)ethoxy)ethoxy) ethanol
CAS-No. 86770-68-5
Formula C10 H21N3 O 5 Mol. weight 263,29 g/mol
PEG6720 N3 -PEG(5)-OH
17-Azido-3,6,9,12,15-pentaoxaheptadecan-1-ol
CAS-No. 86770-69-6
Formula C12H25N3 O 6 Mol. weight 307,34 g/mol


PEG1390 N3 -PEG(12)-OH
35-Azido-3,6,9,12,15,18,21,24,27,30,33-undecaoxapentatriacontan-1-ol
CAS-No. 73342-16-2
Formula C 24H49 N3 O 12 Mol. weight 571,66 g/mol

PEG3770 N3 -PEG(24)-OH
alpha-Azido-omega-hydroxy 24(ethylene glycol)
CAS-No. 73342-16-2
Formula C 48 H97N3 O 24
Mol. weight 1100,29 g/mol

PEG3780 N3 -PEG(36)-OH
alpha-Azido-omega-hydroxy 36(ethylene glycol)
CAS-No. 73342-16-2
Formula C 72H145N3 O 36 Mol. weight 1628,92 g/mol

PEG5350 HO-PEG-N3 (3 kDa)
alpha-Hydroxy-omega-azido poly(ethylene glycol)
CAS-No. 73342-16-2
Mol. weight 3000 Da
PEG5360 HO-PEG-N3 (5 kDa)
alpha-Hydroxy-omega-azido poly(ethylene glycol)
CAS-No. 73342-16-2 Mol. weight 5000 Da
PEG5330 HO-PEG-N3 (10 kDa)
alpha-Hydroxy-omega-azido poly(ethylene glycol)
CAS-No. 73342-16-2
Mol. weight 10000 Da
PEG5340 HO-PEG-N3 (20 kDa)
alpha-Hydroxy-omega-azido poly(ethylene glycol)
CAS-No. 73342-16-2
Mol. weight 20000 Da
Click Chemistry
PEG5170 N3 -TOTA-Suc
1-Azido-4,7,10-trioxa-13-tridecaneamine succinamic acid
CAS-No. 1993176-74-1
Formula C14H26 N4O 6 Mol. weight 346,38 g/mol
PEG5400 N3 -AEEEA*CHA
11-Azido-3,6,9-trioxaundecanoic acid cyclohexylamine
CAS-No. 172531-37-2
Formula C 8 H15N3 O 5*C 6 H13 N Mol. weight 233,22*99,17 g/mol


PEG7950 N3 -AEEA-OK
Potassium 8-azido-3,6-dioxaoctanoate
CAS-No. 882518-90-3
Formula C 6 H10 KN3 O4 Mol. weight 39,10*188,16 g/mol

PEG5390 N3 -O2Oc-Ot Bu
8-Azido-3,6-dioxaoctanoic acid t-butyl ester
CAS-No. 251564-45-1
Formula C10 H19 N3 O4 Mol. weight 245,28 g/mol

PEG2780 N3 -O2Oc-OH*CHA
[2-(2-azidoethoxy)ethoxy]acetic acid cyclohexylamine salt
CAS-No. 2098500-94-6
Formula C 6 H11N3 O4*C 6 H13 N Mol. weight 189,17*99,17 g/mol
PEG2790 N3 -O2Oc-O2Oc-OH
8-(8-Azido-3,6-dioxaoctanoylamido)-3,6-dioxaoctanoic acid
CAS-No. 1254054-60-8
Formula C12H22N4O 7 Mol. weight 334,33 g/mol


RL-4370 N3 -PEG(3)-NH-DIG-OH
Diglycolic acid PEG3 azide
CAS-No. 239081-53-9
Formula C12H22N4O 7 Mol. weight 334,33 g/mol
RL-4380 Fmoc-NH-PEG(3)-N3
(9H-fluoren-9-yl)methyl (2-(2-(2-(2-azidoethoxy)ethoxy) ethoxy)ethyl)carbamate
CAS-No. 1172605-58-1
Formula C 23 H28 N4O 5 Mol. weight 440,50 g/mol
PEG2345 N3 -PEG(4)-COOH
15-Azido-4,7,10,13-tetraoxa-pentadecanoic acid
CAS-No. 1257063-35-6
Formula C11H21N3 O 6 Mol. weight 291,3 g/mol

PEG6915 N3 -PEG(4)-TFP
2,3,5,6-tetrafluorophenyl 1-azido-3,6,9,12-tetraoxapentadecan-15-oate
CAS-No. 1807505-33-4
Formula C17H21F4N3 O 6 Mol. weight 439,36 g/mol
PEG1400 N3 -PEG(4)-NHS
15-Azido-4,7,10,13-tetraoxa-pentadecanoic acid succinimidyl ester
CAS-No. 944251-24-5
Formula C15H24N4O 8 Mol. weight 388,37 g/mol
PEG4170 N3 -PEG(8)-COOH
alpha-Azido-omega-(propionic acid) octa(ethylene glycol)
CAS-No. 1214319-92-2
Formula C19 H37N3 O 10 Mol. weight 467,51 g/mol


Click Chemistry
PEG1405 N3 -PEG(8)-NHS
1-Azido-3,6,9,12,15,18,21,24-octaoxaheptacosan-27-oic acid succinimidyl ester
CAS-No. 1204834-00-3
Formula C 23 H40 N4O 12 Mol. weight 564,58 g/mol
PEG2015 N3 -PEG(9)-COOH
O-(2-Azidoethyl)-O-[2-(diglycolyl-amino)ethyl]heptaethylene glycol
CAS-No. 846549-37-9
Formula C 22H42N4O 12 Mol. weight 554,59 g/mol
PEG6895 Aminooxy-PEG(11)-N3*HCl
O-(35-azido-3,6,9,12,15,18,21,24,27,30,33-undecaoxapentatriacontyl)hydroxylamine hydrochloride
CAS-No. 2560602-21-1 net
Formula C 24H 50 N4O 12*HCl Mol. weight 586,68*36,45 g/mol
PEG4180 N3 -PEG(12)-COOH
alpha-Azido-omega-(propionic acid) dodeca(ethylene glycol)
CAS-No. 1167575-20-3
Formula C 27H 53 N3 O 14 Mol. weight 643,72 g/mol
PEG6925 N3 -PEG(12)-TFP
2,3,5,6-tetrafluorophenyl 1-azido-3,6,9,12,15,18,21,24,27,30,33,36-dodecaoxanonatriacontan-39-oate
CAS-No. 2735663-71-3
Formula C 33 H 53 F4N3 O 14 Mol. weight 791,79 g/mol
PEG1395 N3 -PEG(12)-NHS
1-Azido-3,6,9,12,15,18,21,24,27,30,33,36-dodecaoxanonatriacontan-39-oic acid succinimidyl ester
CAS-No. 1108750-59-9
Formula C 31H 56 N4O 16 Mol. weight 740,79 g/mol




PEG4190 N3 -PEG(24)-COOH
alpha-Azido-omega-(propionic acid) 24(ethylene glycol)
CAS-No. 1167575-20-3
Formula C 51H101N3 O 26 Mol. weight 1172,35 g/mol

PEG7650 N3 -PEG(24)-TFP
alpha-Azido-omega-(2,3,5,6-tetrafluorophenyl propionate) 24(ethylene glycol)
Formula C 57H101F4N3 O 26 Mol. weight 1320,41 g/mol

PEG7660 N3 -PEG(36)-TFP
alpha-Azido-omega-(2,3,5,6-tetrafluorophenyl propionate) 36(ethylene glycol)
CAS-No. 3017961-70-2
Formula C 81H149 F4N3 O 38 Mol. weight 1849,04 g/mol
Whenever free thiol groups (e.g., from cysteine) are used for conjugation, maleimides are typically the reaction partner of choice. However, maleimides also react with other functional groups, for example -COOH, -OH or -NH 2 which may lead to the formation of unwanted impurities. The iodo group reacts more specifically with thiols, resulting in much cleaner conjugates. Product details
PEG7190 Bromoacetamido-PEG(3)-N3
Bromoacetamido-tri(ethylene glycol)-azide
CAS-No. 940005-81-2
Formula C10 H19 BrN4O4 Mol. weight 339,19 g/mol

PEG7200
Bromoacetamido-PEG(11)-N3
Bromoacetamido-undeca(ethylene glycol)-azide
CAS-No. 2172677-17-5
Formula C 26 H 51BrN4O 12 Mol. weight 691,61 g/mol
Click Chemistry
PEG7210 Bromoacetamido-PEG(23)-N3
Bromoacetamido-23(ethylene glycol)-azide
CAS-No. 2735663-83-7
Formula C 50 H99 BrN4O 24
Mol. weight 1220,24 g/mol
PEG3130 I-PEG-N3 (10 kDa)
alpha-Iodo-omega-azido poly(ethylene glycol)
Mol. weight 10000 Da

PEG3140 I-PEG-N3 (20 kDa)
alpha-Iodo-omega-azido poly(ethylene glycol)
Mol. weight 20000 Da

PEG3150 I-PEG-N3 (3 kDa)
alpha-Iodo-omega-azido poly(ethylene glycol)
Mol. weight 3000 Da

PEG3160 I-PEG-N3 (5 kDa)
alpha-Iodo-omega-azido poly(ethylene glycol)
Mol. weight 5000 Da

Reference:
→ Quantitative reactivity profiling predicts functional cysteines in proteomes; E. Weerapana, C. Wang, G. M. Simon, F. Richter, S. Khare, M. B. Dillon, D. A. Bachovchin, K. Mowen, D. Baker, B. F. Cravatt; Nature 2010; 468 : 790-5. arrow-up-right-from-square https://doi.org/10.1038/nature09472
PEG1690
MeO-PEG(4)-N3
13-Azido-2,5,8,11-tetraoxa-tridecane
CAS-No. 606130-90-9
Formula C9 H19 N3 O4 Mol. weight 233,26 g/mol

PEG1705
MeO-PEG(8)-N3
25-Azido-2,5,8,11,14,17,20,23-octaoxapentacosane
CAS-No. 869718-80-9
Formula C17H35N3 O 8 Mol. weight 409,48 g/mol
PEG1660
MeO-PEG(12)-N3
37-Azido-2,5,8,11,14,17,20,23,26,29,32,35-dodecaoxaheptatriacontane
CAS-No. 2170098-29-8
Formula C 25H 51N3 O 12 Mol. weight 585,69 g/mol
PEG1710
MeO-PEG(24)-N3
alpha-Methoxy-omega-azido-24(ethylene glycol)
CAS-No. 89485-61-0
Formula C 49 H99 N3 O 24 Mol. weight 1114,34 g/mol


PEG3430
MeO-PEG(36)-N3
alpha-Methoxy-omega-azido-36(ethylene glycol)
CAS-No. 89485-61-0
Formula C 73 H147N3 O 36 Mol. weight 1642,95 g/mol

PEG1219
MeO-PEG-N3 (750 Da)
alpha-Methoxy-omega-azido poly(ethylene glycol)
Mol. weight 750 Da

Click Chemistry
PEG1225 MeO-PEG-N3 (2 kDa)
alpha-Methoxy-omega-azido poly(ethylene glycol)
Mol. weight 2000 Da

PEG2040 MeO-PEG-N3 (5 kDa)
alpha-Methoxy-omega-azido poly(ethylene glycol)
Mol. weight 5000 Da

PEG2045 MeO-PEG-N3 (10 kDa)
alpha-Methoxy-omega-azido poly(ethylene glycol)
Mol. weight 10000 Da

PEG2050 MeO-PEG-N3 (20 kDa)
alpha-Methoxy-omega-azido poly(ethylene glycol)
Mol. weight 20000 Da

PEG4830 Azido-PEG-Si(OMe)3 (3 kDa)
alpha-Azido-omega-trimethoxysilyl poly(ethylene glycol)
Mol. weight 3000 Da

PEG4835 Azido-PEG-Si(OMe)3 (5 kDa)
alpha-Azido-omega-trimethoxysilyl poly(ethylene glycol)
Mol. weight 5000 Da

PEG4840 Azido-PEG-Si(OMe)3 (10 kDa)
alpha-Azido-omega-trimethoxysilyl poly(ethylene glycol)
Mol. weight 10000 Da

PEG4845 Azido-PEG-Si(OMe)3 (20 kDa)
alpha-Azido-omega-trimethoxysilyl poly(ethylene glycol)
Mol. weight 20000 Da

RL-4420 N3 -PEG(3)-SQA
3-((2-(2-(2-(2-azidoethoxy)ethoxy)ethoxy)ethyl)amino)4-ethoxycyclobut-3-ene-1,2-dione
CAS-No. 2920089-51-4
Formula C14H22N4O 6 Mol. weight 342,35 g/mol
PEG6655 N3 -PEG-SQA (3 kDa)
alpha-Azido-omega-squaric acid ethyl ester poly(ethylene glycol)
Mol. weight 3000 Da

PEG6660 N3 -PEG-SQA (5 kDa)
alpha-Azido-omega-squaric acid ethyl ester poly(ethylene glycol)
Mol. weight 5000 Da

PEG6645 N3 -PEG-SQA (10 kDa)
alpha-Azido-omega-squaric acid ethyl ester poly(ethylene glycol)
Mol. weight 10000 Da

PEG6650 N3 -PEG-SQA (20 kDa)
alpha-Azido-omega-squaric acid ethyl ester poly(ethylene glycol)
Mol. weight 20000 Da

4.4. Alkyne-PEG Derivatives for Click Chemistry
RL-8865 Biotin-PEG(4)-Ynamine
N-(1-ethynyl-1H-benzo[d]imidazol-6-yl)-1-(5((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d] imidazol-4-yl)pentanamido)-3,6,9,12-tetraoxapentadecan-15-amide
Formula C 30 H42N 6 O 7S Mol. weight 630,76 g/mol
PEG4950 Biotin-PEG(4)-alkyne
15-[D(+)-Biotinylamino]-4,7,10,13-tetraoxapentadec-1-yne
CAS-No. 1262681-31-1
Formula C 21H35N3 O 6 S Mol. weight 457,58 g/mol
PEG7980 Biotin-PEG(4)-Dde-Alkyne
N-(15-(4,4-dimethyl-2,6-dioxocyclohexylidene)-3,6,9,12tetraoxa-16-azanonadec-18-ynyl)-5-((3aS,4S,6aR)-2oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamide
CAS-No. 1802908-00-4
Formula C 32H 50 N4O 8 S Mol. weight 650,83 g/mol
PEG8010 Biotin-PEG(4)-Alkyne
(3aS,4S,6aR)-4-(5,21-dioxo-8,11,14,17-tetraoxa-4,20diazapentacos-1-yn-25-yl)tetrahydro-1H-thieno[3,4-d] imidazol-2(3H)-one
CAS-No. 1006592-45-5
Formula C 24H40 N4O 7S Mol. weight 528,66 g/mol
Product details



PEG8110 Biotin-PEG(4)-SS-Alkyne
N-(2-((3-oxo-3-(prop-2-ynylamino)propyl)disulfanyl)ethyl)-1-(5-((3aS,4S,6aR)-2-oxohexahydro-1Hthieno[3,4-d]imidazol-4-yl)pentanamido)-3,6,9,12-tetraoxapentadecan-15-amide
CAS-No. 1260247-54-8
Formula C 29 H49 N 5 O 8 S 3 Mol. weight 691,92 g/mol
PEG6815 Alkyne-O2Oc-OH
Pentynoyl-8-amino-3,6-dioxaoctanoic acid
CAS-No. 2134622-80-1
Formula C11H17NO 5 Mol. weight 243,26 g/mol
PEG5430 Alkyne-PEG(4)-NH2
Alkyne-PEG(4)-amine
CAS-No. 1013921-36-2
Formula C11H21NO4 Mol. weight 231,29 g/mol
PEG2960 H2 N-PEG-alkyne (3 kDa)
alpha-Amino-omega-propargylacetamido poly(ethylene glycol)
Mol. weight 3000 Da



PEG2980 H2 N-PEG-alkyne (5 kDa)
alpha-Amino-omega-propargylacetamido poly(ethylene glycol)
Mol. weight 5000 Da

PEG2950 H2 N-PEG-alkyne (10 kDa)
alpha-Amino-omega-propargylacetamido poly(ethylene glycol)
Mol. weight 10000 Da

Click Chemistry
PEG2970 H2 N-PEG-alkyne (20 kDa)
alpha-Amino-omega-propargylacetamido poly(ethylene glycol)
Mol. weight 20000 Da

PAA1050 Poc-O2Oc-OH*DCHA
8-(Popargylyloxycarbonyl-amino)-3,6-dioxaoctanoic acid dicyclohexylamine
CAS-No. 2988660-57-5
Formula C10 H15NO 6*C12H23 N Mol. weight 245,23*181,32 g/mol
PEG8170 Propargyl-PEG(5)-COOH
4,7,10,13,16-pentaoxanonadec-18-ynoic acid
CAS-No. 1245823-51-1
Formula C14H24O 7 Mol. weight 304,34 g/mol

PEG5410 Alkyne-PEG(4)-NHS
Alkyne-PEG(4)-succinimidyl ester
CAS-No. 1393330-40-9
Formula C18 H27NO 9 Mol. weight 401,41 g/mol

PEG2860 NHS-PEG-alkyne (3 kDa)
alpha-Succinimidyl ester-omega-propargylacetamido poly(ethylene glycol)
Mol. weight 3000 Da

PEG2880 NHS-PEG-alkyne (5 kDa)
alpha-Succinimidyl ester-omega-propargylacetamido poly(ethylene glycol)
Mol. weight 5000 Da

PEG2850
NHS-PEG-alkyne (10 kDa)
alpha-Succinimidyl ester-omega-propargylacetamido poly(ethylene glycol)
Mol. weight 10000 Da

PEG2870
NHS-PEG-alkyne (20 kDa)
alpha-Succinimidyl ester-omega-propargylacetamido poly(ethylene glycol)
Mol. weight 20000 Da

PEG2910
NHS-PEG(NH-Boc)-alkyne (3 kDa)
alpha-Succinimidyl ester-omega-(N-t-Butyloxycarbonyl-L-propargyl-glycinyl) poly(ethylene glycol)
Mol. weight 3000 Da

PEG2930
NHS-PEG(NH-Boc)-alkyne (5 kDa)
alpha-Succinimidyl ester-omega-(N-t-Butyloxycarbonyl-L-propargyl-glycinyl) poly(ethylene glycol)
Mol. weight 5000 Da

PEG2900
NHS-PEG(NH-Boc)-alkyne (10 kDa)
alpha-Succinimidyl ester-omega-(N-t-Butyloxycarbonyl-L-propargyl-glycinyl) poly(ethylene glycol)
Mol. weight 10000 Da

PEG2920
NHS-PEG(NH-Boc)-alkyne (20 kDa)
alpha-Succinimidyl ester-omega-(N-t-Butyloxycarbonyl-L-propargyl-glycinyl) poly(ethylene glycol)
Mol. weight 20000 Da

Click Chemistry
PEG2915
NHS-PEG(NH-Fmoc)-alkyne (3 kDa)
alpha-Succinimidyl ester-omega-(N-(9-Fluorenylmethyloxycarbonyl)-L-propargyl-glycinyl) poly(ethylene glycol)
Mol. weight 3000 Da

PEG2935
NHS-PEG(NH-Fmoc)-alkyne (5 kDa)
alpha-Succinimidyl ester-omega-(N-(9-Fluorenylmethyloxycarbonyl)-L-propargyl-glycinyl) poly(ethylene glycol)
Mol. weight 5000 Da

PEG2905
NHS-PEG(NH-Fmoc)-alkyne (10 kDa)
alpha-Succinimidyl ester-omega-(N-(9-Fluorenylmethyloxycarbonyl)-L-propargyl-glycinyl) poly(ethylene glycol)
Mol. weight 10000 Da

PEG2925
NHS-PEG(NH-Fmoc)-alkyne (20 kDa)
alpha-Succinimidyl ester-omega-(N-(9-Fluorenylmethyloxycarbonyl)-L-propargyl-glycinyl) poly(ethylene glycol)
Mol. weight 20000 Da

PEG5440 Alkyne-PEG(4)-mal
Alkyne-PEG(4)-maleimide
CAS-No. 1609651-90-2
Formula C18 H26 N2O 7
Mol. weight 382,41 g/mol

PEG2840 MeO-PEG-alkyne (750 Da)
alpha-Methoxy-omega-propargylacetamido poly(ethylene glycol)
CAS-No. 1993176-75-2
Mol. weight 750 Da

PEG2810
MeO-PEG-alkyne (2 kDa)
alpha-Methoxy-omega-propargylacetamido poly(ethylene glycol)
CAS-No. 850180-69-7
Mol. weight 2000 Da
PEG2830
MeO-PEG-alkyne (5 kDa)
alpha-Methoxy-omega-propargylacetamido poly(ethylene glycol)
CAS-No. 850180-69-7
Mol. weight 5000 Da
PEG2800
MeO-PEG-alkyne (10 kDa)
alpha-Methoxy-omega-propargylacetamido poly(ethylene glycol)
CAS-No. 850180-69-7
Mol. weight 10000 Da
PEG2820
MeO-PEG-alkyne (20 kDa)
alpha-Methoxy-omega-propargylacetamido poly(ethylene glycol)
CAS-No. 850180-69-7
Mol. weight 20000 Da
PEG4810
Alkyne-PEG-Si(OMe)3 (3 kDa)
alpha-Propargylacetamido-omega-trimethoxysilyl poly(ethylene glycol)
Mol. weight 3000 Da

PEG4815
Alkyne-PEG-Si(OMe)3 (5 kDa)
alpha-Propargylacetamido-omega-trimethoxysilyl poly(ethylene glycol)
Mol. weight 5000 Da

Click Chemistry
PEG4820 Alkyne-PEG-Si(OMe)3 (10 kDa)
alpha-Propargylacetamido-omega-trimethoxysilyl poly(ethylene glycol)
Mol. weight 10000 Da

PEG4825 Alkyne-PEG-Si(OMe)3 (20 kDa)
alpha-Propargylacetamido-omega-trimethoxysilyl poly(ethylene glycol)
Mol. weight 20000 Da

PEG6570 Alkynyl-PEG-SQA (3 kDa)
alpha-Pentynyl-omega-squaric acid ethyl ester poly(ethylene glycol)
Mol. weight 3000 Da

PEG6575 Alkynyl-PEG-SQA (5 kDa)
alpha-Pentynyl-omega-squaric acid ethyl ester poly(ethylene glycol)
Mol. weight 5000 Da

PEG6560 Alkynyl-PEG-SQA (10 kDa)
alpha-Pentynyl-omega-squaric acid ethyl ester poly(ethylene glycol)
Mol. weight 10000 Da

PEG6565 Alkynyl-PEG-SQA (20 kDa)
alpha-Pentynyl-omega-squaric acid ethyl ester poly(ethylene glycol)
Mol. weight 20000 Da

Maleimides are frequently used for conjugation to free thiol groups (e.g., from cysteine). However, maleimides also react with other functional groups such as OH or NH 2 , potentially leading to the formation of impurities. The iodo group reacts more specifically with thiols, resulting in much cleaner conjugates.
Product details
PEG3110 I-PEG-alkyne (3 kDa)
alpha-Iodo-omega-propargylacetamido poly(ethylene glycol)
Mol. weight 3000 Da

PEG3120 I-PEG-alkyne (5 kDa)
alpha-Iodo-omega-propargylacetamido poly(ethylene glycol)
Mol. weight 5000 Da

PEG3090 I-PEG-alkyne (10 kDa)
alpha-Iodo-omega-propargylacetamido poly(ethylene glycol)
Mol. weight 10000 Da

PEG3100 I-PEG-alkyne (20 kDa)
alpha-Iodo-omega-propargylacetamido poly(ethylene glycol)
Mol. weight 20000 Da

4.5. Poly(Amino Acids) and Poly(Oxazolines) for Click Chemistry
PSR1870 N3 -PSar-Ac (5kDa)
N-alpha-(acetyl)-polysarcosine omega-azido propylamide
Mol. weight 5000 Da
POX1200 Me-PMeOx(50)-N3
alpha-Methyl-poly(2-methyl-2-oxazoline)-omega-azide
CAS-No. 26375-28-0
Formula CH3 (C 4H7NO)50N3 Mol. weight 4300 Da

POX1210 Me-PMeOx(100)-N3
alpha-Methyl-poly(2-methyl-2-oxazoline)-omega-azide
CAS-No. 26375-28-0
Formula CH3 (C 4H7NO)100N3 Mol. weight 8500 Da

POX2200 Me-PEtOx(50)-N3
alpha-Methyl-poly(2-ethyl-2-oxazoline)-omega-azide
CAS-No. 25805-17-8
Formula CH3 (C 5H9 NO)50N3 Mol. weight 5000 Da

POX2210 Me-PEtOx(100)-N3
alpha-Methyl-poly(2-ethyl-2-oxazoline)-omega-azide
CAS-No. 25805-17-8
Formula CH3 (C 5H9 NO)100N3 Mol. weight 5000 Da

POX2250 HOOC-PEtOx(50)-N3
alpha-Carboxymethyl-poly(2-ethyl-2-oxazoline)-omega-azide
CAS-No. 25805-17-8
Formula HOCOCH2 (C 5H9 NO)50N3 Mol. weight 5000 Da
POX1250 HOOC-PMeOx(50)-N3
alpha-Carboxymethyl-poly(2-methyl-2-oxazoline)-omega-azide
CAS-No. 26375-28-0
Formula HOCOCH2 (C 4H7NO)50N3 Mol. weight 4300 Da
circle-arrow-right
Interested in products and technologies for drug delivery?
Download our brochure Polymer Therapeutics!



5. Click Chemistry Tools for Proteomics
5.1. Indocyanine Green Dyes for Click Chemistry
Indocyanine green (ICG) dye, a material approved by the FDA for various applications, is a powerful tool for imaging in live cells and tissues.
Iris Biotech offers a series of ICG dyes functionalized with various clickable moieties, such as tetrazine, alkyne, azide or DBCO. Moreover, we offer ICG-equipped with different popular functional groups for conjugation, e.g., maleimide, 2-cyanobenzothiazole (CBT), and NHS.
Fig. 17: Functional groups available for conjugation of indocyanine green.
ICG exhibits an absorption maximum in the near infrared region (NIR) at ca. 800 nm with a slight absorption in the visible range, resulting in a low auto-fluorescence. The emission maximum is at 810 nm. This absorption/emission profile allows for tissue-penetrating excitation without causing tissue damage. Consequently, ICG has found use in fields as diverse as angiography, detection of solid tumours and fluorescence image-guided surgery.
relative intensity (%)
18: Absorption and emission spectra of indocyanine green.
ICG Derivatives for Click Chemistry
RL-2840 ICG-azide
Indocyanine green azide
Formula C 48 H 56 N 6 O4 S Mol. weight 813,06 g/mol

RL-2880 ICG-alkyne
Indocyanine green alkyne
CAS-No. 1622335-41-4
Formula C 48 H 53 N3 O4 S Mol. weight 768,02 g/mol

RL-2870 ICG-DBCO
Indocyanine green dibenzoazacyclooctyne
Formula C 63 H 64N4O 5 S Mol. weight 989,27 g/mol

Fig.
RL-4270 ICG-CBT
4-(2-((1E,3E,5E,7E)-7-(3-(6-((2-((2-cyanobenzo[d] thiazol-6-yl)amino)-2-oxoethyl)amino)-6-oxohexyl)1,1-dimethyl-1,3-dihydro-2H-benzo[e]indol-2-ylidene) hepta-1,3,5-trien-1-yl)-1,1-dimethyl-1H-benzo[e]indol-3ium-3-yl)butane-1-sulfonate
Formula C 55
Mol. weight 945,21 g/mol
References:
→ Near-infrared fluorescence: application to in vivo molecular imaging; S. A. Hilderbrand, R. Weissleder; Curr Opin Chem Biol 2010; 14: 71-9. arrow-up-right-from-square https://doi.org/10.1016/j.cbpa.2009.09.029
→ NIR dyes for bioimaging applications; J. O. Escobedo, O. Rusin, S. Lim, R. M. Strongin; Curr Opin Chem Biol 2010; 14: 64-70. arrow-up-right-from-square https://doi.org/10.1016/j.cbpa.2009.10.022
→ In vivo molecular imaging of cancer with a quenching near-infrared fluorescent probe using conjugates of monoclonal antibodies and indocyanine green; M. Ogawa, N. Kosaka, P. L. Choyke, H. Kobayashi; Cancer Res 2009; 69: 1268-72. arrow-up-right-from-square https://doi.org/10.1158/0008-5472.CAN-08-3116
5.2. Clickable Linkers for Selective Protein Labeling
One way to selectively label a protein is to recombinantly express a modified version containing a sequence that selectively reacts with a specific linker type. Two examples of this approach are the His tag acylation and the HaloTag®.
In the His tag acylation approach, a N-terminal GlyHis 6 tag attached to a protein of interest selectively reacts with a 4-methoxyphenyl ester, generating an acylated N-terminus. While 4-methoxyphenyl esters are too unreactive to undergo acylation with any other primary amine, a proximal imidazole in the GlyHis 6 sequence acts as a catalyst to facilitate selective acylation of the N-terminal glycine.
For the acylation of lysine, Jensen et al. developed the peptide sequence His n-Lys-His m (Lys-His tag) that directs the acylation of the designated lysine N-ε amine under mild conditions and with high selectivity over native lysine residues.
Product details
RL-3010 N3 Ac-OPhOMe 4-Methoxyphenyl 2-azidoacetate
CAS-No. 2546513-31-7
Formula C9 H9 N3 O 3
Mol. weight 207,19 g/mol

The HaloTag® is a protein tag whose sequence can easily be fused to a gene coding for a protein of interest. Functionally, it is a haloalkane dehalogenase that binds and forms covalent bonds to specific halogenated ligands. Those ligands are composed of two parts: a chloroalkane linker that forms the bond with HaloTag® protein, and a functional group or affinity handle. A HaloTag®-containing fusion protein is thus able to selectively label itself with an appropriate haloalkane dehalogenase ligand.
Product details
RL-3640 Halo-PEG(5)-azide
1-azido-21-chloro-3,6,9,12,15-pentaoxahenicosane
CAS-No. 1261238-21-4
Formula C16 H32ClN3 O 5 Mol. weight 381,90 g/mol
RL-3670
Halo-DBCO
N-[2-[2-[(6-chlorohexyl)oxy]ethoxy]ethyl]-gamma-oxodibenz[b,f]azocine-5(6H)-butanamide
CAS-No. 1808119-16-5
Formula C 29 H35 ClN2O4 Mol. weight 511,06 g/mol
RL-3700
Halo-PEG(2)-Azide
1-Azido-12-chloro-3,6-dioxadodecane
CAS-No. 2568146-55-2
Formula C10 H20 ClN3 O 2 Mol. weight 249,74 g/mol
RL-3710
Halo-PEG(4)-Azide
1-Azido-18-chloro-3,6,9,12-tetraoxaoctadecane
CAS-No. 2989398-83-4
Formula C14H28 ClN3 O4 Mol. weight 337,85 g/mol
Find many more Halo derivatives in our webshop!
References:
→ Direct pH measurements by using subcellular targeting of 5(and 6-) carboxyseminaphthorhodafluor in mammalian cells; H. A. Benink, M. G. McDougall, D. H. Klaubert, G. V. Los; Biotechniques 2018; 47(3): 769-774. arrow-up-right-from-square https://doi.org/10.2144/000113220
→ HaloTag technology: a versatile platform for biomedical applications; C. G. England, H. Luo and W. Cai; Bioconjug Chem 2015; 26: 975-86. arrow-up-right-from-square https://doi.org/10.1021/acs.bioconjchem.5b00191
→ HaloTag: a novel protein labeling technology for cell imaging and protein analysis; G. V. Los, L. P. Encell, M. G. McDougall, D. D. Hartzell, N. Karassina, C. Zimprich, M. G. Wood, R. Learish, R. F. Ohana, M. Urh, D. Simpson, J. Mendez, K. Zimmerman, P. Otto, G. Vidugiris, J. Zhu, A. Darzins, D. H. Klaubert, R. F. Bulleit and K. V. Wood; ACS Chem Biol 2008; 3: 373-382. arrow-up-right-from-square https://doi.org/10.1021/cb800025k
Besides HaloTag®, we also offer clickable SNAP-tag® and CLIP-tagTM ligands. The SNAP-tag® is a 20 kDa self-labeling protein tag bearing a cysteine moiety that undergoes an irreversible reaction with synthetic O6-benzylguanine (BG) derivatives, resulting in a covalent thioether bond. The CLIP-tagTM is a modified version of the SNAP-tag (R) , engineered to react with benzylcytosine (BC) instead of benzylguanine (BG).
RL-3930 Alkyne-SNAP
N-(4-(((2-amino-9H-purin-6-yl)oxy)methyl)benzyl) pent-4-ynamide
CAS-No. 1104822-07-2
Formula C18 H18 N 6 O 2 Mol. weight 350,38 g/mol
RL-3940 Alkyne-PEG(5)-SNAP
N-(4-(((2-amino-9H-purin-6-yl)oxy)methyl)benzyl)-4,7,10,13,16-pentaoxanonadec-18-ynamide
Formula C 27H36 N 6 O 7
Mol. weight 556,62 g/mol
RL-3950 Azide-SNAP
N-(4-(((2-amino-9H-purin-6-yl)oxy)methyl)benzyl)-6-azidohexanamide
Formula C19 H23 N9 O 2
Mol. weight 409,45 g/mol
RL-3960 Azide-PEG(4)-SNAP
N-(4-(((2-amino-9H-purin-6-yl)oxy)methyl)benzyl)-1-azido-3,5,7,9-tetraoxadodecan-12-amide
Formula C 24H33 N9 O 6 Mol. weight 543,59 g/mol
RL-4010 DBCO-SNAP
N-(4-(((2-amino-9H-purin-6-yl)oxy)methyl) benzyl)-6-(11,12-didehydrodibenzo[b,f]azocin-5(6H)-yl)-6-oxohexanamide
Formula C 34H31N7 O 3 Mol. weight 585,67 g/mol
References:
→ Site-specific protein labeling with SNAP-Tags; N. B. Cole; Curr Protoc Protein Sci. 2013; 73(30): 1-30. arrow-up-right-from-square https://doi.org/10.1002/0471140864.ps3001s73
→ Directed evolution of O6-alkylguanine-DNA alkyltransferase for efficient labeling of fusion proteins with small molecules in vivo; A. Juillerat, T. Gronemeyer, A. Keppler, S. Gendreizig, H. Pick, H. Vogel, K. Johnsson; Chem. Biol. 2003; 10(4): 313-317. arrow-up-right-from-square https://doi.org/10.1016/s1074-5521(03)00068-1
→ Site-specific, Covalent Labeling of Recombinant Antibody Fragments via Fusion to an Engineered Version of 6-O-Alkylguanine DNA Alkyltransferase; F. Kampmeier, M. Ribbert, T. Nachreiner, S. Dembski, F. Beaufils, A. Brecht, S. Barth; Bioconjugate Chem. 2009; 20(5): 1010-1015. arrow-up-right-from-square https://doi.org/10.1021/bc9000257
→ SNAP-Tag Technology: A Useful Tool to Determine Affinity Constants and Other Functional Parameters of Novel Antibody Fragments; J. Niesen, M. Sack, M. Seidel, R. Fendel, S. Barth, R. Fischer, C. Stein; Bioconjugate Chem. 2016; 27(8): 1931-1941. arrow-up-right-from-square https://doi.org/10.1021/acs.bioconjchem.6b00315
→ Snap-, CLIP- and Halo-Tag Labelling of Budding Yeast Cells; F. Stagge, G. Y. Mitronova, V. N. Belov, C. A. Wurm, S. Jakobs; PLoS ONE 8(10): e78745. arrow-up-right-from-square https://doi.org/10.1371/journal.pone.0078745
In recent years, the so-called cyanobenzothiazole (CBT) click condensation has gained increased interest. It is highly biocompatible and controllable and runs under physiological conditions. In the CBT click reaction, the nitrile function of 2-cyanobenzothiazole (2-CBT) reacts with a β-aminothiol (1,2 aminothiol) and forms a 4,5-dihydro-1,3 thiazole. The β-aminothiol may originate from, e.g., an N-terminal D- or L-cysteine residue, which can be easily introduced recombinantly or during SPPS.
Fig. 19: Mechanism of the CBT click reaction: The electron pair of the sulfur attacks the carbon of the cyano group, the nitrogen forms an enamine. The cysteine’s nitrogen then attacks the carbon of the enamine, and a cyclic intermediate is formed. Finally, the lone electron pair of the amino nitrogen of the cysteine attacks the positively charged carbon to yield the product. The nitrogen which came from the nitrile is leaving the reaction as ammonia.
RL-4280 4-Pentynoyl-Gly-CBT
N-(2-((2-cyanobenzo[d]thiazol-6-yl)amino)-2-oxoethyl) pent-4-ynamide
Formula C15H12N4O 2 S
Mol. weight 312,35 g/mol
RL-4290 6-Azidopentanoyl-Gly-CBT
5-azido-N-(2-((2-cyanobenzo[d]thiazol-6-yl)amino)-2oxoethyl)pentanamide
Formula C15H15N7 O 2 S
Mol. weight 357,39 g/mol
Product details
RL-4310 DBCO-Suc-CBT
N1-(2-cyanobenzo[d]thiazol-6-yl)-N4-(3-(11,12-didehydro-5,6-dihydro-dibenzo[b,f]azocin-yl)-3-oxopropyl) succinamide
Formula C 30 H23 N 5 O 3 S Mol. weight 533,61 g/mol
RL-4300 Suc-CBT
4-((2-cyanobenzo[d]thiazol-6-yl)amino)-4-oxobutanoic acid
CAS-No. 1207457-37-1
Formula C12H9 N3 O 3 S Mol. weight 275,28 g/mol
RL-4270 ICG-CBT
4-(2-((1E,3E,5E,7E)-7-(3-(6-((2-((2-cyanobenzo[d] thiazol-6-yl)amino)-2-oxoethyl)amino)-6-oxohexyl)1,1-dimethyl-1,3-dihydro-2H-benzo[e]indol-2-ylidene) hepta-1,3,5-trien-1-yl)-1,1-dimethyl-1H-benzo[e]indol-3ium-3-yl)butane-1-sulfonate
Formula C 55H 56 N 6 O 5 S 2 Mol. weight 945,21 g/mol
RL-4230 Biotin-Gly-CBT
N-(2-((2-cyanobenzo[d]thiazol-6-yl)amino)-2-oxoethyl)biotin
Formula C 20 H22N 6 O 3 S 2 Mol. weight 458,56 g/mol
RL-4260 FITC-Gly-CBT
N-(2-cyanobenzo[d]thiazol-6-yl)-2-(3-(3‘,6‘-dihydroxy3-oxo-3H-spiro[isobenzofuran-1,9‘-xanthen]-5-yl) thioureido) acetamide
CAS-No. 1207457-29-1
Formula C 31H19 N 5 O 6 S 2 Mol. weight 621,64 g/mol
RL-4240 Biotin-Pen-CBT
N-(5-((2-cyano-6-benzothiazolyl)amino)-5-oxopentyl)biotin
CAS-No. 2550397-17-4
Formula C 23 H28 N 6 O 3 S 2 Mol. weight 500,64 g/mol
RL-4220 Gly-CBT*HCl
2-Amino-N-(2-cyano-6-benzothiazolyl)acetamide hydrochloride
CAS-No. 1207457-28-0 net
Formula C10 H 8 N4OS*HCl Mol. weight 232,26*36,45 g/mol
RL-4250 Biotin-PEG(4)-Gly-CBT
15-Biotinylamino-4,7,10,13-tetraoxa-pentadecanoyl-glycinyl-N-2-cyano-benzothiazole
CAS-No. 1907639-46-6
Formula C 31H43 N7 O 8 S 2 Mol. weight 705,85 g/mol
FAA9760 Fmoc-L-Lys-CBT*TFA
(9H-fluoren-9-yl)methyl (S)-(6-amino-1-((2-cyanobenzo[d]thiazol-6-yl)amino)-1-oxohexan-2-yl)carbamate trifluoroacetic acid
CAS-No. 2241547-00-0 (net)
Formula C 29 H27N 5 O 3 S*C 2HF 3 O 2 Mol. weight 525,62*114,02 g/mol
HAA9700 H-L-Lys(Fmoc)-CBT*TFA
(9H-fluoren-9-yl)methyl (S)-(5-amino-6-((2-cyanobenzo[d]thiazol-6-yl)amino)-6-oxohexyl)carbamate trifluoroacetic acid
CAS-No. 1315482-88-2 (net)
Formula C 29 H27N 5 O 3 S*C 2HF 3 O 2 Mol. weight 525,62*114,02 g/mol
RL-8790 NCBT
2-Cyano-6-aminobenzothiazole
CAS-No. 7224-12-1
Formula C 8 H 5N3 S Mol. weight 175,21 g/mol
References:
→ N, S-Double labeling of N-terminal cysteines via an alternative conjugation pathway with 2-cyanobenzothiazole; W. Wang, J. Gao; Org. Chem. 2020; 85: 1756-1763. arrow-up-right-from-square https://doi.org/10.1021/acs.joc.9b02959
→ Thiol-cyanobenzothiazole ligation for the efficient preparation of peptide-PNA conjugates; N. Patil, J. Karas, B. Turner, F. Shabanpoor; Bioconjugate Chem. 2019; 30: 73-799. arrow-up-right-from-square https://doi.org/10.1021/acs.bioconjchem.8b00908
→ CBT-Cys click reaction for optical bioimaging in vivo. X. Hu, R. Tang, L. Bai, S. Liu, G. Liang, X. Sun; View. 2023; 20220065. arrow-up-right-from-square https://doi.org/10.1002/VIW.20220065
While nitriles in general may react with sulfhydryl groups (e.g., from cysteines) in a rather unspecific way, the advantage of α-cyanopyridines (2-picolinonitrils) is their strong selectivity for 1,2-aminothiols (e.g., provided as N-terminal cysteine or internally as non-canonical amino acid). Thus, this reaction may be used for the synthesis of cyclic peptides or to fuse ligands to peptide chains.
H2O, pH 7.5
Fig. 20: The reaction between a 2-cyanopyridine and a 1,2-aminothiol forms a 2-thiazoline.
This click-like reaction is biorthogonal, biocompatible, catalyst-free, and selective; it proceeds readily in aqueous solutions at physiological pH and ambient temperature. The formation of the resulting 2-thiazoline (alternative name: 2,5-dihydrothiazole) is driven by the release of ammonia. The reaction product is stable at physiological conditions.
Product details
RL-8625
N3 -PEG(4)-CINA
N-(14-azido-3,6,9,12-tetraoxatetradecyl)-2-cyanoisonicotinamide
Formula C17H24N 6 O 5 Mol. weight 392,42 g/mol
RL-8630 Alkyne-PEG(4)-CINA
2-cyano-N-(3,6,9,12-tetraoxapentadec-14-yn-1-yl) isonicotinamide
Formula C18 H23 N3 O 5 Mol. weight 361,40 g/mol
RL-8640
DBCO-PEG(4)-CINA
Dibenzoazacyclooctyne-tetra(ethylene glycol)-cyanoisonicotinamide
Formula C 36 H39 N 5 O 7 Mol. weight 653,74 g/mol
References:
→ The Cyanopyridine-Aminothiol Click Reaction: Expanding Horizons in Chemical Biology; C. Nitsche; SynLett. 2024; 35: A-E. arrow-up-right-from-square https://dx.doi.org/10.1055/a-2214-7612
→ Biocompatible and Selective Generation of Bicyclic Peptides; S. Ullrich, J. George, A. Coram, R. Morewood, C. Nitsche; Angew. Chem Int. Ed. 2022; 61(43): e20228400. arrow-up-right-from-square https://doi.org/10.1002/anie.202208400
→ Tobacco Etch Virus protease: A shortcut across biotechnologies; F. Cesaratto, O. Burrone, G. Petris; J Biotechnol. 2016; 231(10) : 239-249. arrow-up-right-from-square https://doi.org/10.1016/j.jbiotec.2016.06.012
circle-arrow-right Find many more derivatives in our webshop!
Besides, we offer click-functionalized biotin derivatives. Biotinylation represents a versatile tool for purification, immobilization, and labeling. It is a rather small molecule compared to other labels, thus minimizing its steric impact on the biotinylated carrier itself.
LS-4660 Biotinyl-DAPe-N3
N-(5-azidopentyl)-5-((3aS,4S,6aR)-2-oxohexahydro-1Hthieno[3,4-d]imidazol-4-yl)pentanamide
CAS-No. 1349190-76-6
Formula C15H26 N 6 O 2 S Mol. weight 354,47 g/mol
LS-4210 Biotin-N3
N-(3-azidopropyl)-5-((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamide
CAS-No. 908007-17-0
Formula C13 H22N 6 O 2 S Mol. weight 326,42 g/mol
LS-4300 DecarboxyBiotin-N3
(3aS,4S,6aR)- 4-(5-Azidopentyl)tetrahydro-1H-Thieno[3,4-d]imidazol-2(3H)-one
CAS-No. 1260586-88-6
Formula C10 H17N 5 OS Mol. weight 255,34 g/mol
Product details
LS-4270 Biotin-DBCO
(3aS,4S,6aR)-N-[3-(11,12-Didehydrodibenz[b,f] azocin-5(6H)-yl)-3-oxopropyl]hexahydro-2-oxo-1Hthieno[3,4-d]imidazole-4-pentanamide
CAS-No. 1418217-95-4
Formula C 28 H30 N4O 3 S Mol. weight 502,63 g/mol
LS-4280 Biotin-MeTz
N-(4-(6-methyl-1,2,4,5-tetrazin-3-yl)benzyl)-5((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamide
CAS-No. 1802883-51-7
Formula C 20 H25N7 O 2 S Mol. weight 427,53 g/mol
LS-4290 Biotin-PEG(4)-MeTz
N-(4-(6-methyl-1,2,4,5-tetrazin-3-yl)benzyl)-1-(5((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d] imidazol-4-yl)pentanamido)-3,6,9,12-tetraoxapentadecan-15-amide
CAS-No. 1962919-31-8
Formula C 31H46 N 8 O 7S Mol. weight 674,82 g/mol
PEG8100
Biotin-PEG(4)-SS-Azide
N-(2-((3-((3-azidopropyl)amino)-3-oxopropyl)disulfaneyl)ethyl)-1-(5-((3aS,4S,6aR)-2-oxohexahydro-1Hthieno[3,4-d]imidazol-4-yl)pentanamido)-3,6,9,12-tetraoxapentadecan-15-amide
CAS-No. 1260247-52-6
Formula C 29 H 52N 8 O 8 S 3 Mol. weight 736,96 g/mol
PEG8140
Biotin-PEG(4)-Dde-DBCO
N-(15-(4,4-dimethyl-2,6-dioxocyclohexylidene)-19-oxo19-(azadibenzocyclooctyn-1-yl)-3,6,9,12-tetraoxa-16azanonadecyl)-5-((3aS,4S,6aR)-2-oxohexahydro-1Hthieno[3,4-d]imidazol-4-yl)pentanamide
CAS-No. 1807512-43-1
Formula C 47H 61N 5 O 9 S Mol. weight 872,08 g/mol
PEG8110
Biotin-PEG(4)-SS-Alkyne
N-(2-((3-oxo-3-(prop-2-ynylamino)propyl)disulfanyl)ethyl)-1-(5-((3aS,4S,6aR)-2-oxohexahydro-1Hthieno[3,4-d]imidazol-4-yl)pentanamido)-3,6,9,12-tetraoxapentadecan-15-amide
CAS-No. 1260247-54-8
Formula C 29 H49 N 5 O 8 S 3 Mol. weight 691,92 g/mol
PEG8120
Biotin-PEG(4)-SS-DBCO
N-(2-((3-(3-(azadibenzocyclooctyn-1-yl)-3-oxopropylamino)-3-oxopropyl)disulfanyl)ethyl)-1-(5-((3aS,4S,6aR)2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamido)-3,6,9,12-tetraoxapentadecan-15-amide
Formula C 44H 60 N 6 O 9 S 3 Mol. weight 913,18 g/mol
PEG7990 Biotin-PEG(4)-N3
(3aS,4S,6aR)-4-(28-azido-5,21-dioxo-9,12,15,18-tetraoxa-6,22-diazaoctacosyl)tetrahydro-1H-thieno[3,4-d]imidazol-2(3H)-one
CAS-No. 1006592-62-6
Formula C 27H49 N7 O 7S Mol. weight 615,79 g/mol
PEG7960
Biotin-PEG(4)-Dde-N3
N-(19-azido-15-(4,4-dimethyl-2,6-dioxocyclohexylidene)-3,6,9,12-tetraoxa-16-azanonadecyl)-5-((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamide
CAS-No. 1802907-93-2
Formula C 32H 53 N7 O 8 S Mol. weight 695,87 g/mol
PEG8000
Biotin-PEG(4)-Picolyl-N3
(3aS,4S,6aR)-4-(1-(6-(azidomethyl)pyridin-3-ylamino)1,17-dioxo-4,7,10,13-tetraoxa-16-azahenicosan-21-yl) tetrahydro-1H-thieno[3,4-d]imidazol-2(3H)-one
CAS-No. 2222687-71-8
Formula C 27H42N 8 O 7S Mol. weight 622,74 g/mol
PEG7970
Biotin-PEG(4)-Dde-Picolyl-N3
N-(6-(azidomethyl)pyridin-3-yl)-15-(4,4-dimethyl-2,6dioxocyclohexylidene)-1-(5-((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamido)-3,6,9,12-tetraoxa-16-azanonadecan-19-amide
CAS-No. 2055048-42-3
Formula C 38 H 57N9 O 9 S Mol. weight 815,98 g/mol
PEG4950
Biotin-PEG(4)-alkyne
15-[D(+)-Biotinylamino]-4,7,10,13-tetraoxapentadec-1-yne
CAS-No. 1262681-31-1
Formula C 21H35N3 O 6 S Mol. weight 457,58 g/mol
PEG8010 Biotin-PEG(4)-Alkyne
(3aS,4S,6aR)-4-(5,21-dioxo-8,11,14,17-tetraoxa-4,20diazapentacos-1-yn-25-yl)tetrahydro-1H-thieno[3,4-d] imidazol-2(3H)-one
CAS-No. 1006592-45-5
Formula C 24H40 N4O 7S Mol. weight 528,66 g/mol
PEG7980 Biotin-PEG(4)-Dde-Alkyne
N-(15-(4,4-dimethyl-2,6-dioxocyclohexylidene)-3,6,9,12tetraoxa-16-azanonadec-18-ynyl)-5-((3aS,4S,6aR)-2oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamide
CAS-No. 1802908-00-4
Formula C 32H 50 N4O 8 S Mol. weight 650,83 g/mol
PEG4330
Biotin-PEG(7)-N3
alpha-Biotin-omega-azido hepta(ethylene glycol)
CAS-No. 1334172-75-6
Formula C 26 H48 N 6 O 9 S Mol. weight 620,76 g/mol
Click Chemistry
PEG4340 Biotin-PEG(11)-N3
[2-(2-aminoethoxy)ethoxy]acetic acid tert-butyl ester*HCl
CAS-No. 956494-20-5
Formula C 34H 64N 6 O 13 S Mol. weight 796,97 g/mol
PEG4350 Biotin-PEG(23)-N3
alpha-Biotin-omega-azido 23(ethylene glycol)
CAS-No. 956494-20-5
Formula C 58 H112N 6 O 25 S Mol. weight 1325,6 g/mol
RL-3490 Biotin-Propargylamide
Biotinyl-N-propargylamide
CAS-No. 773888-45-2
Formula C13 H19 N3 O 2 S Mol. weight 281,37 g/mol
LS-4680 Biotin-PEG(3)-PyAz
6-(azidomethyl)-N-(13-oxo-17-((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)-3,6,9-trioxa-12-azaheptadecyl)nicotinamide
Formula C 25H38 N 8 O 6 S Mol. weight 578,69 g/mol . circle-arrow-right
Find many more biotin derivatives in our dedicated brochure!

In addition to the aforementioned options, we would like to introduce ynamines for Click chemistry. Ynamines are molecules in which an alkynyl group is attached to a saturated nitrogen. Aromatic ynamines are very reactive and readily undergo CuAAC Click reactions with azides much faster than conventional aliphatic alkynes, even if both types are present in the same molecule. This different reactivity allows for sequential, chemoselective Click reactions. Additionally, the ynamine-azide Click reaction requires only the presence of copper(II) ions, in contrast to the standard CuAAC Click reaction, where a copper ligand and ascorbate are also needed. This simplifies the process and offers further selectivity and reaction control.
Fig. 21: Sequential and highly chemoselective reaction of ynamine-alkyne (RL-8850) with two different azide-tagged molecules (red and yellow bubbles). First, the ynamine (alkynyl moiety in orange) is converted to the 1,2,3-triazole. Then, the aliphatic alkyne (blue) reacts. In this way, sequential Click reactions may be conducted.
By carefully selecting the solvent and reaction conditions, excellent chemoselectivity can be achieved and one-pot syntheses with equimolar amounts of the reactants may be set up – eliminating the need to isolate and purify the intermediates. The first CuAAC Click reaction is performed between the ynamine and the first azide in acetonitrile as solvent, using 5 mol% copper(II) acetate as the catalyst, at room temperature for 16 hours. For the second Click reaction, targeting the aliphatic alkyne, the conditions are adjusted to those of a standard CuAAC protocol: aqueous methanol is added, followed by the second azide, 10 mol% AMTC (1-(trans-2-hydroxycyclohexyl)-4-(N,N-dimethylaminomethyl)-1,2,3-triazole)) as a ligand, and 10 mol% sodium ascorbate. The reaction mixture is then stirred for additional 16 hours at room temperature.
1st Click reaction
2nd Click reaction
Click Chemistry
RL-8850 Ynamine-Alkyne
N-(1-ethynyl-1H-benzo[d]imidazol-6-yl)hept-6-ynamide
Formula C16 H15N3 O Mol. weight 265,32 g/mol
RL-8855 Ynamine-PEG(4)-Alkyne
N-(1-ethynyl-1H-benzo[d]imidazol-6-yl)-4,7,10,13-tetraoxahexadec-15-ynamide
Formula C 21H25N3 O 5 Mol. weight 399,45 g/mol
RL-8860 Biotin-Ynamine
N-(1-ethynyl-1H-benzo[d]imidazol-6-yl)-5((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamide
Formula C19 H21N 5 O 2 S Mol. weight 383,47 g/mol
RL-8865 Biotin-PEG(4)-Ynamine
N-(1-ethynyl-1H-benzo[d]imidazol-6-yl)-1-(5((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d] imidazol-4-yl)pentanamido)-3,6,9,12-tetraoxapentadecan-15-amide
Formula C 30 H42N 6 O 7S Mol. weight 630,76 g/mol
References:
→ Chemoselective Sequential Click Ligations Directed by Enhanced Reactivity of an Aromatic Ynamine; M. Z. C. Hatit, J. C. Sadler, L. A. McLean, B. C. Whitehurst, C. P. Seath, L. D. Humphreys, R. J. Young, A. J. B. Watson, G. A. Burley; Org. Lett. 2016; 18: 1694-1697. arrow-up-right-from-square https://doi.org/10.1021/acs.orglett.6b00635
→ Mechanistic Basis of the Cu(OAc)2 Catalyzed Azide-Ynamine (3 + 2) Cycloaddition Reaction; R. P. Bunschoten, F. Peschke, A. Taladriz-Sender, E. Alexander, M. J. Andrews, A. R. Kennedy, N. J. Fazakerley, G. C. L. Jones, A. J. B. Watson, G. A. Burley; J. Am. Chem. Soc. 2024; 146(19): 13558-3570. arrow-up-right-from-square https://doi.org/10.1021/jacs.4c03348
→ Reactivity Profiling for High-Yielding Ynamine-Tagged Oligonucleotide Click Chemistry Bioconjugations; F. Peschke, A. Taladriz-Sender, A. J. B. Watson, G. A. Burley; Bioconj. Chem. 2024; 35(11): 1788-1796. arrow-up-right-from-square https://doi.org/10.1021/acs.bioconjchem.4c00353
→ Strategy for Conditional Orthogonal Sequential CuAAC Reactions Using a Protected Aromatic Ynamine; M. Z. C. Hatit, C. P. Seath, A. J. B. Watson, G. A. Burley; J. Org Chem. 2017; 82(10) : 5461-5468. arrow-up-right-from-square https://doi.org/10.1021/acs.joc.7b00545
6. Carbohydrates for Click Chemistry
Glycoconjugates, i.e. glycans linked to proteins or lipids, are an essential part of all living organisms. In higher organisms, but also in lower eukaryotes and some bacteria and archaea, many proteins are posttranslationally modified by linking oligosaccharides to amino acid side-chains, forming glycoproteins. Glycosylation is the most complex posttranslational modification and can be observed on membrane proteins, secreted proteins and peptides, or proteins in the cytosol and nucleus.
Glycoconjugates display a multitude of biological effects from protein folding and stabilization, to cell surface interaction through molecular recognition motifs for cell-cell communication, and structural support and protection.
Abnormal glycosylation patterns can be observed in pathological conditions such as neurodegenerative diseases or tumor growth and metastasis. Moreover, glycosylation patterns play a decisive role in the infection pathways of and the immune response against many pathogens, further underlining the importance of this type of modification.
Synthetic glycoconjugates are interesting targets for the investigation of immunogenicity, infection pathways or structure activity relationships, and for the development of novel drugs and vaccines. Carbohydrates functionalized for Click chemistry provide mild and selective access to such glycoconjugates.
Fig. 22: Simplified representation of a eukaryotic cell and its cell surface glycans.
6.1. Galactose Derivatives
GBB1445 alpha-GalNAc-N3
1-O-(2-Azidoethoxy)-2-acetamido-2deoxy-alpha-D-galactopyranoside
CAS-No. 195384-42-0
Formula C10 H18 N4O 6 Mol. weight 290,27 g/mol
GBB1370 alpha-GalNAc-TEG-N3
1-O-(2-(2-(2-Azidoethoxy)ethoxy)ethoxy)-2-acetamido-2deoxy-alpha-D-galactopyranoside
CAS-No. 882873-70-3
Formula C14H26 N4O 8 Mol. weight 378,38 g/mol
GBB1430 beta-Gal-Et-N3
1-(2-Azidoethoxy)-beta-D-galactopyranose
CAS-No. 151651-54-6
Formula C 8 H15N3 O 6 Mol. weight 249,22 g/mol
GBB1380 beta-Gal-TEG-N3
1-O-(2-(2-(2-Azidoethoxy)ethoxy)ethoxy)-beta-D-galactopyranoside
CAS-No. 126765-27-3
Formula C12H23 N3 O 8 Mol. weight 337,33 g/mol
GBB1375 alpha-GalNAc-TEG-Alkyne
1-O-(2-(2-(2-(Prop-2-ynyloxy)ethoxy)ethoxy) ethoxy)-2-acetamido-2deoxy-alpha-D-galactopyranoside
Formula C17H29 NO 9 Mol. weight 391,41 g/mol


GBB1385 beta-Gal-TEG-Alkyne
1-O-(2-(2-(2-(Prop-2-ynyloxy)ethoxy)ethoxy) ethoxy)-beta-D-galactopyranoside
CAS-No. 1072903-79-7
Formula C15H26 O 9 Mol. weight 350,36 g/mol
6.2. Glucose Derivatives
GBB1435 beta-Glc-N3
1-(2-Azidoethoxy)-beta-D-glucopyranose
CAS-No. 165331-08-8
Formula C 8 H15N3 O 6 Mol. weight 249,22 g/mol
GBB1390 beta-Glc-TEG-N3
1-O-(2-(2-(2-Azidoethoxy)ethoxy)ethoxy)-beta-D-glucopyranoside
CAS-No. 156058-83-2
Formula C12H23 N3 O 8 Mol. weight 337,33 g/mol
GBB1395 beta-Glc-TEG-Alkyne
1-O-(2-(2-(2-(Prop-2-ynyloxy)ethoxy)ethoxy) ethoxy)-beta-D-glucopyranoside
CAS-No. 1072903-76-4
Formula C15H26 O 9 Mol. weight 350,36 g/mol
GBB1400 beta-GlcNAc-TEG-N3
1-O-(2-(2-(2-Azidoethoxy)ethoxy)ethoxy)-2-acetamido-2deoxy-beta-D-glucopyranoside
CAS-No. 86520-54-9
Formula C14H26 N4O 8 Mol. weight 378,73 g/mol
Product details

GBB1405 beta-GlcNAc-TEG-Alkyne
1-O-(2-(2-(2-(Prop-2-ynyloxy)ethoxy)ethoxy) ethoxy)-2-acetamido-2deoxy-beta-D-glucopyranoside
CAS-No. 1884184-44-4
Formula C17H29 NO 9 Mol. weight 391,41 g/mol
6.3. Mannose Derivatives
GBB1440 alpha-Man-N3
1-(2-Azidoethoxy)-alpha-D-mannopyranose
CAS-No. 155196-97-7
Formula C 8 H15N3 O 6 Mol. weight 249,22 g/mol
GBB1420 alpha-Man-TEG-N3
1-O-(2-(2-(2-Azidoethoxy)ethoxy)ethoxy)-alpha-D-mannopyranoside
CAS-No. 246855-76-5
Formula C12H23 N3 O 8 Mol. weight 337,33 g/mol
GBB1425 alpha-Man-TEG-Alkyne
1-O-(2-(2-(2-(Prop-2-ynyloxy)ethoxy)ethoxy)ethoxy)-alpha-D-mannopyranoside
CAS-No. 1072903-80-0
Formula C15H26 O 9 Mol. weight 350,36 g/mol
6.4. Lactose Derivatives
GBB1455 beta-Lac-EO-N3
2-Azidoethyl 4-O-beta-D-galactopyranosyl-beta-(1->4)-D-glucopyranoside
CAS-No. 230286-11-0
Formula C14H25N3 O 11
Mol. weight 411,36 g/mol
GBB1410 beta-Lac-TEG-N3
(2-(2-(2-Azidoethoxy)ethoxy)ethyl) 4-O-beta-D-galactopyranosyl-beta-(1->4)-D-glucopyranoside
CAS-No. 246855-74-3
Formula C18 H33 N3 O 13 Mol. weight 499,47 g/mol
GBB1415 beta-Lac-TEG-Alkyne
2-[2-[2-(2-propyn-1-yloxy)ethoxy]ethoxy]ethyl 4-O-beta-D-galactopyranosyl-beta-(1->4)-D-glucopyranoside
CAS-No. 1442747-66-1
Formula C 21H36 O 14 Mol. weight 512,5 g/mol


References:
→ Nanovesicles displaying functional linear and branched oligomannose self-assembled from sequence-defined Janus glycodendrimers; Q. Xiao, M. Delbianco, S. E. Sherman, A. M. Reveron Perez, P. Bharate, A. Pardo-Vargas, C. Rodriguez-Emmenegger, N. Y. Kostina, K. Rahimi, D. Soder, M. Moller, M. L. Klein, P. H. Seeberger, V. Percec; Proc Natl Acad Sci U S A 2020: 202003938. arrow-up-right-from-square https://doi.org/10.1073/pnas.2003938117
→ Glycans in drug discovery; P. Valverde, A. Ardá, N.-C. Reichardt, J. Jiménez-Barbero, A. Gimeno; MedChemComm 2019. arrow-up-right-from-square https://doi.org/10.1039/c9md00292h
→ Stepwise orthogonal click chemistry toward fabrication of paclitaxel/galactose functionalized fluorescent nanoparticles for HepG2 cell targeting and delivery; C. H. Lai, T. C. Chang, Y. J. Chuang, D. L. Tzou, C. C. Lin; Bioconjug Chem 2013; 24: 1698-709. arrow-up-right-from-square https://doi.org/10.1021/bc400219t
→ Design of multivalent galactosyl carborane as a targeting specific agent for potential application to boron neutron capture therapy; C. H. Lai, Y. C. Lin, F. I. Chou, C. F. Liang, E. W. Lin, Y. J. Chuang, C. C. Lin; Chem Commun (Camb) 2012; 48 : 612-4. arrow-up-right-from-square https://doi.org/10.1039/c1cc14447b
→ Essentials of Glycobiology; A. Varki, R. Schauer, R. D. Cummings, J. D. Esko, H. H. Freeze, P. Stanley, C. R. Bertozzi, G. W. Hart, M. E. Etzler; 2009.
→ Role of glycosylation in development; R. S. Haltiwanger, J. B. Lowe; Annu. Rev. Biochem. 2004; 73: 491-537. arrow-up-right-from-square https://doi.org/10.1146/annurev.biochem.73.011303.074043
→ Glycosylation and the immune system; P. M. Rudd, T. Elliott, P. Cresswell, I. A. Wilson, R. A. Dwek; Science 2001; 291: 2370-6. arrow-up-right-from-square https://doi.org/10.1126/science.291.5512.2370
→ Glycosylation in cellular mechanisms of health and disease; K. Ohtsubo, J. D. Marth; Cell 2006; 126: 855-67. arrow-up-right-from-square https://doi.org/10.1016/j.cell.2006.08.019
→ The role of selectins in inflammation and disease; K. Ley; Trends Mol Med 2003; 9: 263-8.
→ Carbohydrate diversity: synthesis of glycoconjugates and complex carbohydrates; A. Holemann, P. H. Seeberger; Curr Opin Biotechnol 2004; 15: 615-22. arrow-up-right-from-square https://doi.org/10.1016/j.copbio.2004.10.001
→ Post-translational modifications in the context of therapeutic proteins; G. Walsh, R. Jefferis; Nat. biotechnol. 2006; 24: 1241-52. arrow-up-right-from-square https://doi.org/10.1038/nbt1252
→ Glycomics: a pathway to a class of new and improved therapeutics; Z. Shriver, S. Raguram, R. Sasisekharan; Nat Rev Drug Discov 2004; 3: 863-73. arrow-up-right-from-square https://doi.org/10.1038/nrd1521
→ The bittersweet promise of glycobiology; A. Dove; Nat. biotechnol. 2001; 19: 913-7. arrow-up-right-from-square https://doi.org/10.1038/nbt1001-913
→ Carbohydrate and protein immobilization onto solid surfaces by sequential Diels-Alder and azide-alkyne cycloadditions; X. L. Sun, C. L. Stabler, C. S. Cazalis, E. L. Chaikof; Bioconjug Chem 2006; 17: 52-7. arrow-up-right-from-square https://doi.org/10.1021/bc0502311
→ Covalent display of oligosaccharide arrays in microtiter plates; M. C. Bryan, F. Fazio, H. K. Lee, C. Y. Huang, A. Chang, M. D. Best, D. A. Calarese, O. Blixt, J. C. Paulson, D. Burton, I. A. Wilson, C. H. Wong; J Am Chem Soc 2004; 126: 8640-1. arrow-up-right-from-square https://doi.org/10.1021/ja048433f
→ Multivalent, bifunctional dendrimers prepared by click chemistry; P. Wu, M. Malkoch, J. N. Hunt, R. Vestberg, E. Kaltgrad, M. G. Finn, V. V. Fokin, K. B. Sharpless, C. J. Hawker; Chem Commun (Camb) 2005: 5775-7. arrow-up-right-from-square https://doi.org/10.1039/b512021g
→ A chemoenzymatic approach to glycopeptide antibiotics; H. Lin, C. T. Walsh; J Am Chem Soc 2004; 126: 139984003. arrow-up-right-from-square https://doi.org/10.1021/ja045147v
. circle-arrow-right
You cannot find the derivative you are looking for? Get in contact for a custom synthesis!

RL-3240
4-Pentynoyl-Val-Ala-PAB-PNP
RL-8670 5HP2O-alkyne
ADC1290 6-Azidohexanoyl-Val-Ala-PAB
ADC1680 6-Azidohexanoyl-Val-Ala-PAB-Cl
ADC1300 6-Azidohexanoyl-Val-Ala-PAB-PNP
ADC1120 6-Azidohexanoyl-Val-Cit-PAB
ADC1130 6-Azidohexanoyl-Val-Cit-PAB-PNP
RL-4290 6-Azidopentanoyl-Gly-CBT
RL-3480 8-Azido-octanoyl-OSu
ADC1790 Alkyne-HMPO-OH
ADC1800 Alkyne-HMPO-PNP
RL-2055 Alkyne-myristic acid
PEG6815 Alkyne-O2Oc-OH
RL-2060 Alkyne-palmitic acid
RL-8630 Alkyne-PEG(4)-CINA
PEG5440 Alkyne-PEG(4)-mal
PEG5430 Alkyne-PEG(4)-NH2 112
PEG5410 Alkyne-PEG(4)-NHS 113
ADC1350 Alkyne-PEG(4)-Val-Ala-PAB 85
ADC1720 Alkyne-PEG(4)-Val-Ala-PAB-Cl 85
ADC1360 Alkyne-PEG(4)-Val-Ala-PAB-PNP 85
RL-3940 Alkyne-PEG(5)-SNAP 125
ADC1180 Alkyne-PEG(5)-Val-Cit-PAB 85
ADC1190 Alkyne-PEG(5)-Val-Cit-PAB-PNP 85
PEG4820 Alkyne-PEG-Si(OMe)3 (10 kDa) 117
PEG4825 Alkyne-PEG-Si(OMe)3 (20 kDa) 117
PEG4810 Alkyne-PEG-Si(OMe)3 (3 kDa) 116
PEG4815 Alkyne-PEG-Si(OMe)3 (5 kDa)
PEG6565 Alkynyl-PEG-SQA (20 kDa)
PEG6570 Alkynyl-PEG-SQA (3 kDa) 117
PEG6575 Alkynyl-PEG-SQA (5 kDa)
ADC1580
ADC1590
ADC1480
ADC1490
ADC1330
ADC1710
ADC1340
Azido-cyclobutane-1,1-dicarboxamide-Ala-PAB 68
Azido-cyclobutane-1,1-dicarboxamide-Ala-PAB-PNP 68
Azido-cyclobutane-1,1-dicarboxamide-Cit-PAB 69
Azido-cyclobutane-1,1-dicarboxamide-Cit-PAB-PNP 69
Azido-PEG(4)-Val-Ala-PAB 69
Azido-PEG(4)-Val-Ala-PAB-Cl 67
Azido-PEG(4)-Val-Ala-PAB-PNP 69
ADC1160 Azido-PEG(4)-Val-Cit-PAB 69
ADC1170
Azido-PEG(4)-Val-Cit-PAB-PNP 69
PEG4840 Azido-PEG-Si(OMe)3 (10 kDa) 110
Click Chemistry
PEG4940
LS-4680 Biotin-PEG(3)-PyAz
PEG4950 Biotin-PEG(4)-alkyne
PEG8010 Biotin-PEG(4)-Alkyne
RL-2520 Biotin-PEG(4)-DBCO
PEG7980 Biotin-PEG(4)-Dde-Alkyne
PEG8140 Biotin-PEG(4)-Dde-DBCO
PEG7960 Biotin-PEG(4)-Dde-N3
PEG7970 Biotin-PEG(4)-Dde-Picolyl-N3
RL-4250 Biotin-PEG(4)-Gly-CBT 129
LS-4290 Biotin-PEG(4)-MeTz
PEG7990 Biotin-PEG(4)-N3
PEG8000 Biotin-PEG(4)-Picolyl-N3
PEG8110 Biotin-PEG(4)-SS-Alkyne
PEG8100 Biotin-PEG(4)-SS-Azide
PEG8120 Biotin-PEG(4)-SS-DBCO
RL-8865 Biotin-PEG(4)-Ynamine
PEG4330 Biotin-PEG(7)-N3
Product code Product name Page
PEG6795 Biotin-PEG3-Ph(4-N3) 96
RL-4240 Biotin-Pen-CBT 128
RL-3490 Biotin-Propargylamide 71, 135
RL-4120 Biotin-SS-N3 67
PEG2065 Biotin-TEG-ATFBA 95
RL-8860 Biotin-Ynamine 83, 137
LS-4660 Biotinyl-DAPe-N3 131
PEG7075 Bis-PEG(11)-DBCO 14
BAA4895 Boc-Aeg(N3)-OK 50
BAA4920 Boc-Aoa-NH-PEG3-N3 100
BNN1320 Boc-Cystamine-Poc 84
(2R,4R)
Boc-L-Cys(Propargyl)-OH*DCHA 75
BAA1810 Boc-L-Lys(N3)-OH*CHA 53
BAA4900 Boc-L-Lys(N3)-OK 53
BAA1830 Boc-L-Orn(N3)-OH*CHA 57
BAA4660 Boc-L-Phe(4-CH2-N3)-OH 63
BAA4670 Boc-L-Phe(4-Eth)-OH 81
BAA1850 Boc-L-Phe(4-N3)-OH 63
BAA3980 Boc-L-Phe(4-NH-Poc)-OH 81
BAA1434 Boc-L-Pra-OH*DCHA 77
BAA1905 Boc-L-Pro(4-N3)-OH (2S,4S) 60
BAA1930 Boc-L-Pro(4-N3)-OH*DCHA (2S,4R) 60
BAA2260 Boc-L-Ser(Propargyl)-OH*DCHA 82
BAA4650 Boc-L-Tyr(2-azidoethyl)-OH 65
BAA2265 Boc-L-Tyr(Propargyl)-OH*DCHA 83
BAA3230 Boc-N-(propargyl)-glycine 76
PEG4960 Boc-NH-PEG(2)-N3 100
PEG7200 Bromoacetamido-PEG(11)-N3 106
PEG6800 Bromoacetamido-PEG(12)-DBCO 11
ADC1620 DBCO-cyclobutane-1,1-dicarboxamide-Ala-PAB
ADC1630 DBCO-cyclobutane-1,1-dicarboxamide-Ala-PAB-PNP
ADC1520 DBCO-cyclobutane-1,1-dicarboxamide-Cit-PAB
(10kDa)
(5kDa)
DBCO-PEG(12)-(5)6-carboxyfluorescein
RL-2480 DBCO-PEG(3)-BisSulfonThiol-Linker
DBCO-PEG(4)-mal
DBCO-PEG(4)-NH2*TFA
DBCO-PEG(4)-TFP
(2R,4R)
Click Chemistry
FAA7400
FAA8605
FAA9665
FAA1820
(solv.)
FAA2035
FAA9675
FAA8855
FAA3150 Fmoc-L-Lys(Pryoc)-OH
FAA9760 Fmoc-L-Lys-CBT*TFA
Fmoc-L-MeDap(N3)-OH
FAA9660
HAA2090 H-L-Lys(Poc)-OH*HCl 78
HAA1620 H-L-Orn(N3)-OH*HCl 57
HAA9500 H-L-Phe(3-BuTz)-OH*TFA 20
HAA9510 H-L-Phe(3-iPrTz)-OH*TFA 20
HAA9490 H-L-Phe(3-MeTz)-OH*TFA 20
HAA9840 H-L-Phe(3-N3)-OH*HCl 62
HAA9480 H-L-Phe(4-Azido-PrTz)-OH*TFA 20, 63
HAA4090 H-L-Phe(4-CH2-N3)*HCl 63
HAA9220 H-L-Phe(4-Eth)-OH*HCl 80
HAA9470
PEG2950 H2N-PEG-alkyne (10 kDa)
PEG2970
PEG2960
(20 kDa)
(3 kDa)
H2N-PEG-alkyne (5 kDa)
(5 kDa)
PEG2810
(3 kDa)
(5 kDa)
I-PEG-alkyne (10 kDa)
PEG2050
(10 kDa)
(2 kDa)
(20 kDa)
(5 kDa)
(750 Da)
(10 kDa)
(2 kDa)
(20 kDa)
(5 kDa)
Click Chemistry
(750 Da)
RL-3915 MeTz-PhAc-Sulfo-NHS
RL-8820 Monobromo-maleimido-PEG(4)-DBCO 15
RL-8830 Monobromo-pyridazinedione-PEG(4)-DBCO 15
PEG2920 NHS-PEG(NH-Boc)-alkyne (20 kDa)
PEG2910 NHS-PEG(NH-Boc)-alkyne (3 kDa)
PEG2930 NHS-PEG(NH-Boc)-alkyne (5 kDa)
PEG2905 NHS-PEG(NH-Fmoc)-alkyne (10 kDa)
PEG2925 NHS-PEG(NH-Fmoc)-alkyne (20 kDa)
PEG2915 NHS-PEG(NH-Fmoc)-alkyne (3 kDa) 115
PEG2935 NHS-PEG(NH-Fmoc)-alkyne (5 kDa)
PEG2850 NHS-PEG-alkyne (10 kDa)
PEG2870 NHS-PEG-alkyne (20 kDa)
PEG2860 NHS-PEG-alkyne (3 kDa)
PEG2880 NHS-PEG-alkyne (5 kDa)
RL-2090 Norbornene-methyl-NHS
RL-2080 Norbornene-NHS
RL-4030 PFB-mercaptopropionyl-PEG3-N3
Photo-Click-Palmitic acid
RL-3760 Photo-Click-Stearic acid
ADC1600 Propargyl-cyclobutane-1,1-dicarboxamide-Ala-PAB
ADC1500 Propargyl-cyclobutane-1,1-dicarboxamide-Cit-PAB 86 ADC1510 Propargyl-cyclobutane-1,1-dicarboxamide-Cit-PAB-PNP 86
Code of Conduct
As business activity of Iris Biotech GmbH impacts people’s lives and health, it must be operated in ethical and correct manner and act with integrity and responsibility. To ensure high ethical standards and fair business practices, Iris Biotech GmbH applies an integrated policy known as its Code of Conduct.
In 2001 Iris Biotech GmbH was founded just at the beginning of the Biotech movement and the first remarkable breakthrough of biotech pharma products. Although the biotech field is rather young compared to other industries we believe on long-term business, a good partnership between our business partners and Iris Biotech GmbH and a good reputation. It is our duty as well as our responsibility to maintain and to extend this over the next generations – based on the principles of an honourable and prudent tradesman which based upon the concept of honourable entrepreneurship.
This Code of Conduct has been developed following the “Voluntary Guidelines for Manufacturers of Fine Chemical Intermediates and Active Ingredients” issued by AIME (Agrochemical & Intermediates Manufacturers in Europe) and the requirements of some of our business associates.
Iris Biotech GmbH commits to hold this Code of Conduct and to include and apply its principles in the management system and the company policies.
Ethics
Iris Biotech GmbH undertakes business in an ethical manner and acts with integrity. All corruption, extortion and embezzlement are prohibited. We do not pay or accept bribes or participate in other illegal inducements in business or government relationships. We conduct our business in compliance with all applicable anti-trust laws. Employees are encouraged to report concerns or illegal activities in the workplace, without threat of reprisal, intimidation or harassment.
Labour
Iris Biotech GmbH is committed to uphold the human rights of workers and to treat them with dignity and respect. Child labour, workplace harassment, discrimination, and harsh and inhumane treatment are prohibited. Iris Biotech GmbH respects the rights of the employees to associate freely, join or not join labour unions, seek representation and join workers’ councils. Employees are paid and their working timetable is established according to applicable wage and labour laws. Employees are able to communicate openly with management regarding working conditions without threat of reprisal, intimidation or harassment.
General Policies
Contracts and Secrecy Agreements are binding and the confidential information received is only used for intended purposes. Clear management and organizational structures exist to provide efficient normal working and to address problems quickly. Know-how is protected and intellectual property isrespected.
Health and Safety
Iris Biotech GmbH provides a safe and healthy working environment to the employees and protects them from overexposure to chemical and physical hazards. Products are produced, stored and shipped under the guidelines of the relevant chemical and safety legislation. Risks and emergency scenarios are identified and evaluated, and their possible impact is minimized by implementing emergency plans and written procedures. Safety information regarding hazardous materials is available to educate, train and protect workers from hazards. Preventive equipment and facilities maintenance is performed at suitable periods to reduce potential hazards. Employees are regularly trained in health and safety matters and are informed about product properties and risk classification when it is required.
Environment
Iris Biotech GmbH operates in an environmentally responsible and efficient manner, minimizing adverse impacts on the environment. Waste streams are managed to ensure a safe handling, movement, storage, recycling and reuse, before and after being generated. Systems to prevent and mitigate accidental spills and releases to the environment are in place. All required environmental permits and licenses are obtained and their operational and reporting requirements are complied with.
Production and Quality Management
A quality management system following the Good Distribution Practices (GDP rules) of Active Pharmaceutical Ingredients is established covering all the aspects of the worldwide distribution of products. Regular audits are performed to evaluate the efficiency and fulfilling of the quality system. Process controls to provide reproducible product quality are established. There are preventive maintenance procedures to ensure plant reliability and the lowest risk of failure. Staff is trained periodically about GMP and GDP rules. Procedures are established and installations are designed to avoid cross contamination. Batch and analytical records are kept for inspection and audit purposes for suitable periods according guidelines.
Research and Development
Research and development staff education is appropriate to their functional activity and they are trained to develop, optimize and scale-up the processes. Intellectual property is respected and knowhow protected. Development of manufacturing processes reflects the principles of the Green Chemistry according to the American Chemical Society Green Chemistry Institute. Animal testing is not used unless alternatives are not scientifically valid or accepted by regulators. If animal testing is carried out, animals are treated so that pain and stress are minimized.
Terms and Conditions of Sales
All orders placed by a buyer are accepted and all contracts are made subject to the terms which shall prevail and be effective notwithstanding any variations or additions contained in any order or other document submitted by the buyer. No modification of these terms shall be binding upon Iris Biotech GmbH unless made in writing by an authorised representative of Iris Biotech GmbH.
Placing of Orders
Every order made by the buyer shall be deemed an offer by the buyer to purchase products from Iris Biotech GmbH and will not be binding on Iris Biotech GmbH until a duly authorised representative of Iris Biotech GmbH has accepted the offer made by the buyer. Iris Biotech GmbH may accept orders from commercial, educational or government organisations, but not from private individuals and Iris Biotech GmbH reserves the right to insist on a written order and/or references from the buyer before proceeding.
There is no minimum order value. At the time of acceptance of an order Iris Biotech GmbH will either arrange prompt despatch from stock or the manufacture/acquisition of material to satisfy the order. In the event of the latter Iris Biotech GmbH will indicate an estimated delivery date. In addition to all its other rights Iris Biotech GmbH reserves the right to refuse the subsequent cancellation of the order if Iris Biotech GmbH expects to deliver theproduct on or prior to the estimated delivery date. Time shall not be of the essence in respect of delivery of the products. If Iris Biotech GmbH is unable to deliver any products by reason of any circumstances beyond its reasonable control („Force Majeure“) then the period for delivery shall be extended by the time lost due to such Force Majeure. Details of Force Majeure will be forwarded by Iris Biotech GmbH to the buyer as soon as reasonably practicable.
Prices, Quotations and Payments
Prices are subject to change. For the avoidance of doubt, the price advised by Iris Biotech GmbH at the time of the buyer placing the order shall supersede any previous price indications. The buyer must contact the local office of Iris Biotech GmbH before ordering if further information is required. Unless otherwise agreed by the buyer and Iris Biotech GmbH, the price shall be for delivery ex-works. In the event that the buyer requires delivery of the products otherwise than ex-works the buyer should contact the local office of Iris Biotech GmbH in order to detail its requirements. Iris Biotech GmbH shall, at its discretion, arrange the buyer‘s delivery requirements including, without limitation, transit insurance, the mode of transit (Iris Biotech GmbH reserves the right to vary the mode of transit if any regulations or other relevant considerations so require) and any special packaging requirements (including cylinders). For the avoidance of doubt all costs of delivery and packaging in accordance with the buyer‘s requests over and above that of delivery in standard packaging ex-works shall be for the buyer‘s account unless otherwise agreed by both parties. Incoterms 2020 shall apply. Any tax, duty or charge imposed by governmental authority or otherwise and any other applicable taxes, duties or charges shall be for the buyer‘s account. Iris Biotech GmbH may, on request and where possible, provide quotations for multiple packs or bulk quantities, and non-listed items. Irrespective of the type of request or means of response all quotations must be accepted by the buyer without condition and in writing before an order will be accepted by Iris Biotech GmbH. Unless agreed in writing on different terms, quotations are valid for 30 days from the date thereof. Payment terms are net 30 days from invoice date unless otherwise agreed in writing. Iris Biotech GmbH reserves the right to request advance payment at its discretion. For overseas transactions the buyer shall pay all the banking charges of Iris Biotech GmbH. The buyer shall not
be entitled to withhold or set-off payment for the products for any reason whatsoever. Government/ Corporate Visa and MasterCard (and other such credit cards) may be accepted on approved accounts for payment of the products. Personal credit cards are not acceptable. Failure to comply with the terms of payment of Iris Biotech GmbH shall constitute default without reminder. In these circumstances Iris Biotech GmbH may (without prejudice to any other of its rights under these terms) charge interest to accrue on a daily basis at the rate of 2% per month from the date upon which payment falls due to the actual date of payment (such interest shall be paid monthly). If the buyer shall fail to fulfil the payment terms in respect of any invoice of Iris Biotech GmbH Iris Biotech GmbH may demand payment of all outstanding balances from the buyer whether due or not and/or cancel all outstanding orders and/or decline to make further deliveries or provision of services except upon receipt of cash or satisfactory securities. Until payment by the buyer in full of the price and any other monies due to Iris Biotech GmbH in respect of all other products or services supplied or agreed to be supplied by Iris Biotech GmbH to the buyer (including but without limitation any costs of delivery) the property in the products shall remain vested in Iris Biotech GmbH.
Shipping, Packaging and Returns
The buyer shall inspect goods immediately on receipt and inform Iris Biotech GmbH of any shortage or damage within five days. Quality problems must be notified within ten days of receipt. Goods must not be returned without prior written authorisation of Iris Biotech GmbH. Iris Biotech GmbH shall at its sole discretion replace the defective products (or parts thereof) free of charge or refund the price (or proportionate price) to buyer. Opened or damaged containers cannot be returned by the buyer without the written prior agreement of Iris Biotech GmbH. In the case of agreed damaged containers which cannot be so returned, the buyer assumes responsibility for the safe disposal of such containers in accordance with all applicable laws.
Product Quality, Specifications and Technical Information
Products are analysed in the Quality Control laboratories of Iris Biotech GmbH’s production partners by methods and procedures which Iris Biotech GmbH considers appropriate. In the event of any dispute concerning reported discrepancies arising from the buyer’s analytical results, determined by the buyer’s own analytical procedures, Iris Biotech GmbH reserves the right to rely on the results of own analytical methods of Iris Biotech GmbH. Certificates of Analysis or Certificates of Conformity are available at the discretion of Iris Biotech GmbH for bulk orders but not normally for prepack orders. Iris Biotech GmbH reserves the right to make a charge for such certification. Specifications may change and reasonable variation from any value listed should not form the basis of a dispute. Any supply by Iris Biotech GmbH of bespoke or custom product for a buyer shall be to a specification agreed by both parties in writing. Technical information, provided orally, in writing, or by electronic means by or on behalf of Iris Biotech GmbH, including any descriptions, references, illustrations or diagrams in any catalogue or brochure, is provided for guidance purposes only and is subject to change.
Safety
All chemicals should be handled only by competent, suitably trained persons, familiar with laboratory procedures and potential chemical hazards. The burden of safe use of the products of Iris Biotech GmbH vests in the buyer. The buyer assumes all responsibility for warning his employees, and any persons who might reasonably be expected to come into contact with the products, of all risks to person and property in any way connected with the products and for instructing them in their safe handling and use. The buyer also assumes the responsibility for the safe disposal of all products in accordance with all applicable laws.
Uses, Warranties and Liabilities
All products of Iris Biotech GmbH are intended for laboratory research purposes and unless otherwise stated on product labels, in the catalogue and product information sheet of Iris Biotech GmbH or in other literature furnished to the buyer, are not to be used for any other purposes, including but not limited to use as or as components in drugs for human or animal use, medical devices, cosmetics, food additives, household chemicals, agricultural or horticultural products or pesticides. Iris Biotech GmbH offers no warranty regarding the fitness of any product for a particular purpose and shall not be responsible for any loss or damage whatsoever arising there from. No warranty or representation is given by Iris Biotech GmbH that the products do not infringe any letters patent, trademarks, registered designs or other industrial rights. The buyer further warrants to Iris Biotech GmbH that any use of the products in the United States of America shall not result in the products becoming adulterated or misbranded within the meaning of the Federal Food, Drug and Cosmetic Act (or such equivalent legislation in force in the buyer‘s jurisdiction) and shall not be materials which may not, under sections 404, 505 or 512 of the Act, be introduced into interstate commerce. The buyer acknowledges that, since the products of Iris Biotech GmbH are intended for research purposes, they may not be on the Toxic Substances Control Act 1976 („TSCA“) inventory. The buyer warrants that it shall ensure that the products are approved for use under the TSCA (or such other equivalent legislation in force in the buyer‘s jurisdiction), if applicable. The buyer shall be responsible for complying with any legislation or regulations governing the use of the products and their importation into the country of destination (for the avoidance of doubt to include, without limitation, the TSCA and all its amendments, all EINECS, ELINCS and NONS regulations). If any licence or consent of any government or other authority shall be required for the acquisition, carriage or use of the products by the buyer the buyer shall obtain the same at its own expense and if necessary produce evidence of the same to Iris Biotech GmbH on demand. Failure to do so shall not entitle the buyer to withhold or delay payment. Any additional expenses or charges incurred by Iris Biotech GmbH resulting from such failure shall be for the buyer‘s account. Save for death or personal injury caused by negligence of Iris Biotech GmbH, sole obligation of Iris Biotech GmbH and buyer‘s exclusive remedy with respect to the products proved to the satisfaction of Iris Biotech GmbH to be defective or products incorrectly supplied shall be to accept the return of said products to Iris Biotech GmbH for refund of the actual purchase price paid by the buyer (or proportionate part thereof), or replacement of the defective product (or part thereof) with alternative product. Iris Biotech GmbH shall have no liability to the buyer under or arising directly or indirectly out of or otherwise in connection with the supply of products by Iris Biotech GmbH to the buyer and/or their re-sale or use by the buyer or for any product, process or services of the buyer which in any way comprises the product in contract tort (including negligence or breach of statutory duty) or otherwise for pure economic loss, loss of profit, business, reputation, depletion of brand, contracts, revenues or anticipated savings or for any special indirect or consequential damage or loss of any nature except as may otherwise be expressly provided for in these terms. All implied warranties, terms and representations in respect of the products (whether implied by statute or otherwise) are excluded to the fullest extent permitted by law. The buyer shall indemnify Iris Biotech GmbH for and against any and all losses, damages and expenses, including legal fees and other costs of defending any action, that Iris Biotech GmbH may sustain or incur as a result of any act or omission by the buyer, its officers, agents or employees, its successors or assignees, its customers or all other third parties, whether direct or indirect, in connection with the use of any product. For the avoidance of doubt and in the event that Iris Biotech GmbH supplies bespoke or custom product to the buyer‘s design or specification, this indemnity shall extend to include any claim by a third party that the manufacture of the product for the buyer or the use of the product by the buyer infringes the intellectual property rights of any third party.
General
Iris Biotech GmbH shall be entitled to assign or sub-contract all or any of its rights and obligations hereunder. The buyer shall not be entitled to assign, transfer, sub-contract or otherwise delegate any of its rights or obligations hereunder. Any delay or forbearance by Iris Biotech GmbH in exercising any right or remedy under these terms shall not constitute a waiver of such right or remedy. If any provision of these terms is held by any competent authority to be invalid or unenforceable in whole or in part the validity of the other provisions of these terms and the remainder of the provision in question shall not be affected. These terms shall be governed by German Law and the German Courts shall have exclusive jurisdiction for the hearing of any dispute between the parties save in relation to enforcement where the jurisdiction of the German Courts shall be non-exclusive.

Get in Contact Distribution Partners
Iris Biotech GmbH
Adalbert-Zoellner-Str. 1 95615 Marktredwitz Germany




+49 (0) 9231 97121-0
+49 (0) 9231 97121-99 info@iris-biotech.de www.iris-biotech.de
The list contains the current distributors of Iris Biotech in different regions of the world. The latest list of distribution partners and contact details is available at: arrow-up-right-from-square www.iris-biotech.de/distribution-partner
China: Chengdu Yoo Technology Co., Ltd.
Japan:
BizCom Japan, Inc.
Shigematsu & Co., Ltd
Cosmo Bio Co., Ltd.
USA & Canada: Peptide Solutions LLC
India, Bangladesh, Oman, Sri Lanka, United Arab Emirates: Sumit Biosciences Pvt Ltd.
Argentina, Brazil, Colombia: Seafront Biotech GmbH
